Characterization of a Single-Cycle Rabies Virus-Based Vaccine Vector
|
|
|
- Kelly Horn
- 5 years ago
- Views:
Transcription
1 JOURNAL OF VIROLOGY, Mar. 2010, p Vol. 84, No X/10/$12.00 doi: /jvi Copyright 2010, American Society for Microbiology. All Rights Reserved. Characterization of a Single-Cycle Rabies Virus-Based Vaccine Vector Emily A. Gomme, 1 Elizabeth J. Faul, 1 Phyllis Flomenberg, 2,3 James P. McGettigan, 1,3 and Matthias J. Schnell 1,2 * Departments of Microbiology and Immunology 1 and of Medicine, 2 and Jefferson Vaccine Center, 3 Jefferson Medical College, Thomas Jefferson University, Philadelphia, Pennsylvania Received 3 September 2009/Accepted 24 December 2009 Recombinant rabies virus (RV)-based vectors have demonstrated their efficacy in generating long-term, antigen-specific immune responses in murine and monkey models. However, replication-competent viral vectors pose significant safety concerns due to vector pathogenicity. RV pathogenicity is largely attributed to its glycoprotein (RV-G), which facilitates the attachment and entry of RV into host cells. We have developed a live, single-cycle RV by deletion of the G gene from an RV vaccine vector expressing HIV-1 Gag (SPBN- G- Gag). Passage of SPBN- G-Gag on cells stably expressing RV-G allowed efficient propagation of the G-deleted RV. The in vivo immunogenicity data comparing single-cycle RV to a replication-competent control (BNSP- Gag) showed lower RV-specific antibodies; however, the overall isotype profiles (IgG2a/IgG1) were similar for the two vaccine vectors. Despite this difference, mice immunized with SPBN- G-Gag and BNSP-Gag mounted similar levels of Gag-specific CD8 T-cell responses as measured by major histocompatibility complex class I Gag-tetramer staining, gamma interferon enzyme-linked immunospot assay, and cytotoxic T-cell assay. Moreover, these cellular responses were maintained equally at immunization titers as low as 10 3 focus-forming units for both RV vaccine vectors. CD8 T-cell responses were significantly enhanced by a boost with a single-cycle RV complemented with a heterologous vesicular stomatitis virus glycoprotein. These findings demonstrate that single-cycle RV is an effective alternative to replication-competent RV vectors for future development of vaccines for HIV-1 and other infectious diseases. * Corresponding author. Mailing address: Department of Microbiology and Immunology, Jefferson Medical College, Thomas Jefferson University, 233 South 10th St., 531 BLSB, Philadelphia, PA Phone: (215) Fax: (215) Published ahead of print on 6 January The global spread of HIV-1 represents one of the most significant pandemics to afflict humans (22). Despite tremendous efforts to increase HIV awareness in the general population, UNAIDS reports that fewer than one in five people has access to HIV prevention strategies and many are subject to cultural stigmas thwarting such efforts (43). As such, an HIV vaccine is paramount for preventing disease transmission. It is not yet clear precisely what characteristics are critical for an effective HIV vaccine, yet evidence suggests one would need to induce both antibody and CD8 T-cell-mediated immunity (reviewed in reference 25). Live viruses are at the forefront of HIV vaccine development (7) because they are powerful inducers of both of these arms of immunity. We previously demonstrated that replication-competent rabies virus (RV)-based vectors can induce long-lasting antigen-specific immune responses in both murine and monkey models, as well as protect rhesus macaques from an AIDS-like disease (23, 24, 26 29, 42). However, there are safety concerns with the use of any replication-competent virus for widespread immunization. To address this, we sought to develop and evaluate the immunogenicity of a safer alternative: a single-cycle RV expressing HIV-1 Gag as a model antigen. Single-cycle viral vectors are defective in certain viral components that are required for infectious particle assembly (reviewed in reference 12). As such, the virus undergoes one complete cycle of replication in the primary infected cell and produces progeny virions that are unable to spread to a second round of cells. The progeny are noninfectious and provide inert antigen that may or may not be immunogenic (12). In contrast, so-called replication-deficient viruses do not complete that initial round of replication. These two attenuation strategies have been adopted for use with many different viruses including, but not limited to, adenovirus (Ad), vaccinia virus (VV), canarypox virus (CPV), herpes simplex virus (HSV), vesicular stomatitis virus (VSV), and, more recently, RV (4, 6, 9, 18, 21, 33, 35, 36, 38). However, the results regarding the immunogenicity of such vectors are mixed. For example, both the replication-deficient Ad5 vector and modified vaccinia Ankara (MVA) showed reduced humoral and cellular immunogenicity compared to their replication-competent counterparts, but the use of higher titers and multiple immunizations did increase such responses (18, 33, 35). In the case of CPV, the replication-deficient vector provided poor HIV-specific cellular responses, causing the termination of phase II HIV-1 vaccine trials (38). In contrast, single-cycle VSV, a rhabdovirus closely related to RV, has been shown to induce HIV-1 Env-specific CD8 T-cell responses equivalent to full-length VSV when administered intramuscularly (36). However, protection of rhesus macaques against highly pathogenic simian immunodeficiency virus (SIV) challenge by both replication-competent and single-cycle VSV needs to be shown. In the study described here, we generated a single-cycle RV vector expressing HIV-1 Gag (SPBN- G-Gag) by deletion of 2820
2 VOL. 84, 2010 SINGLE-CYCLE RV VECTOR 2821 FIG. 1. Construction of the single-cycle RV, SPBN- G-Gag. Shown are the negative-sense RNA genomes of the recombinant RV utilized in subsequent experiments. The cdna copy of full-length SPBN (A) has a deletion of RV-G, and HIV-1-Gag is inserted between RV-M and RV-L to make single-cycle SPBN- G-Gag (B). A replication-competent control was generated by inserting HIV-1 Gag between RV-N and RV-P of the full-length BNSP cdna (C) to make BNSP-Gag (D). the entire RV glycoprotein (RV-G) from the RV genome. RV-G was chosen due to its critical role in the attachment and entry of RV into host cells, which makes RV-G one of the most important determinants of viral pathogenicity (10, 11, 37). RV particles lacking G are unable to spread, as evidenced by intracranial infection with a G-deleted RV that remains restricted to the primary infected neurons (13, 44). It must be noted that in the absence of RV-G, virions are still capable of budding though at a 30-fold lower efficiency (32). These virions, however, are incapable of attachment and entry into a secondary host cell. Because of this, SPBN- G-Gag was propagated on a trans-complementing cell line induced to express RV-G (or VSV RV-G), effectively facilitating virus spread. To evaluate the immunogenicity of the single-cycle vector, we immunized mice and compared the humoral and cellular responses to responses generated by replication-competent RV. Our results indicate that single-cycle RV generates reduced vector-specific antibody responses but similar HIV-1 Gag-specific CD8 T-cell responses. Moreover, these responses can be significantly enhanced by a heterologous boost with a singlecycle RV complemented with a VSV glycoprotein. Taken together, the results presented here show evidence that singlecycle RV is a promising platform for a safe, live viral vaccine for use against HIV-1 and other applications. MATERIALS AND METHODS Construction of recombinant RV. The plasmid encoding SPBN-Gag, described previously (27), was used to synthesize pspbn- G-Gag. pspbn-gag was digested with SmaI and PacI to remove the entire RV-G gene. Incubation with Klenow fragment was followed by blunt end ligation, yielding pspbn- G-Gag (Fig. 1). The construction of pbnsp-gag has been described previously (26) (Fig. 1). Recovery of infectious virus from cdna. Recombinant RV was recovered as previously described (15) with the following modifications for single-cycle RV recovery. Briefly, BSR cells were transfected using FuGENE 6 (Roche Diagnostics) with 5 g of pspbn- G-Gag cdna, 1.5 g of T7, 2.5 g of ptit-n, 1.25 g of ptit-p, 1.25 g of ptit-l, and 1 g of ptit-g (per six-well plate). Four days posttransfection the supernatant was transferred to induced BSR RV-G cells or BSR VSV RV-G cells for passage to higher titers in serum-free medium. These viruses are complemented with RV-G or VSV RV-G from the cell lines on which they are passaged and are referred to as SPBN- G-Gag RVG and SPBN- G-Gag VSV-RVG, respectively. Infectious virus titers were determined by focus-forming assay, as previously described (37). Titers of stocks of single-cycle virus were determined in parallel on noncomplementing BSR cells to verify that they were restricted to single cell infections. Generation of cell lines stably expressing RV-G or VSV-G. (i) BSR-RVG cells. We used the Tet-off inducible gene expression system (Clontech Inc.) to generate a BSR cell line (a BHK-21 cell clone) stably expressing RV-G (BSR-RVG cells). Briefly, RV-G was cloned into the ptre2 response plasmid (Clontech Inc.) utilizing the MluI and NheI restriction enzyme sites, yielding ptre2-rvg. BSR-RVG cells were generated by cotransfection of BSR cells using FuGENE 6 (Roche Diagnostics) with the ptet-off transcriptional activator plasmid (Clontech Inc.) and the ptre2-rvg plasmid. Cell clones resistant to G418 were selected and maintained in Dulbecco s modified Eagle s medium (DMEM) containing 1 g/ml doxycycline to suppress RV-G transcription and treated with 1 mg/ml G418 at every third passage. (ii) BSR-VSV-RVG cells. Similarly, VSV-RVG cells were generated by cotransfection of BSR cells with ptet-off and ptre2hyg (Clontech Inc.) encoding a VSV RV-G chimera (ptre2hyg-vsvrvg). The chimeric VSV RV-G is composed of the ectodomain (ED) and transmembrane domain (TM) of VSV-G, Indiana strain, fused to the RV-G cytoplasmic domain (CD), as described previously (17, 29). Cell clones resistant to G418 and hygromycin were selected and maintained in DMEM containing 1 g/ml doxycycline and treated with 1 mg/ml G418 and 250 g/ml hygromycin selection agents at every third passage. (iii) Expression of RV-G or VSV-G. Anti-RV-G surface staining was used to confirm RV-G expression by the inducible BSR-RVG cell line. Cells were washed three times in PBS to remove all traces of doxycycline, cultured in doxycycline-free medium for 48 h, and fixed with 4% paraformaldehyde (Cytofix; BD Biosciences) for 30 min. Fixed cells were washed with 10 mm glycine phosphate-buffered saline (PBS), blocked with 1% bovine serum albumin for 1 h, and stained with polyclonal rabbit anti-rv-g, followed by secondary staining with Cy-2-conjugated donkey anti-rabbit IgG (Jackson Immunoresearch). Similarly, cell surface expression of VSV-G was confirmed by staining with two monoclonal mouse anti-vsv-g antibodies (I1 and I14 [39]), followed by secondary staining with Cy-2-conjugated donkey anti-mouse IgG (Jackson Immunoresearch). p24 antigen ELISA. To quantify the relative production of p24 by each viral vector, BSR cells were infected at a multiplicity of infection (MOI) of 10. Seventy-two hours later, lysate was collected as previously described (27), and p24 was quantified using a p24 antigen enzyme-linked immunosorbent assay (ELISA) kit (ZeptoMetrix, Inc.), as described by the manufacturer. Virus spread assay. Spread assays were conducted to analyze the capacity of the single-cycle virus to multiply on trans-complementing BSR-RVG or BSR- VSV-RVG cells, as well as on noncomplementing BSR cells. The complementing cell lines were induced by removal of doxycycline, followed by infection with the respective RV at an MOI of After 2 h, the virus was removed, cells were washed one time in PBS, and either doxycycline-containing or doxycyclinefree medium was replenished. After incubation for 72 h at 34 C, cells were fixed with 4% paraformaldehyde (Cytofix; BD Biosciences) for 30 min, and viral antigen was detected with fluorescein isothiocyanate (FITC)-conjugated anti-rv nucleoprotein (RV-N) monoclonal antibody (Centocor) using a fluorescence microscope. Expression of HIV-1 Gag was confirmed by focus-forming assay, as previously described (37). Briefly, cells were fixed with 80% acetone for 30 min and stained with monoclonal human anti-p24 antibody (71-31; NIH AIDS Research and Reference Reagent Program) at room temperature for 1 h, followed by secondary staining with Cy-2-conjugated donkey anti-human IgG (Jackson Immunoresearch). Multistep growth curves. The BSR-RVG or BSR-VSV-RVG cell line, induced, as described above, in parallel with BSR cells, was infected at an MOI of
3 2822 GOMME ET AL. J. VIROL After 2 h the virus was removed, cells were washed one time in PBS, and doxycycline-free medium was replenished. Supernatants were sampled at the time points indicated in the figures and virus titers were determined as described above. Mouse immunization, boost, and challenge. Mice were maintained at the Thomas Jefferson University Animal Facilities, and all experimental procedures were approved by the Institutional Animal Care and Use Committee. In vivo studies were conducted using 6- to 8-week-old female BALB/c mice immunized (for prime or prime-boost experiments) intramuscularly (i.m.) in the leg muscle at the time points and concentrations of virus indicated in the figures. To study the recall immune responses, mice rested at least 4 weeks postimmunization were inoculated intraperitoneal (i.p.) with PFU recombinant vaccinia virus expressing HIV-1 Gag (VV-Gag) (NIH AIDS Research and Reference Reagent Program). The induced immune responses were studied by sacrificing the mice at the time points indicated in the figures postimmunization/challenge and aseptically harvesting and homogenizing the spleens into a single-cell suspension in splenocyte medium (RPMI 1640 medium supplemented with 10% fetal bovine serum [FBS], penicillin-streptomycin, 10 mm HEPES, and 50 M -mercaptoethanol). Red blood cells were lysed using ACK lysing buffer (Gibco), and splenocytes were counted by trypan blue exclusion. Spleen samples were kept separate for analysis. Serum was collected at the time points indicated in the figures postimmunization for antibody analysis by ELISA. Anti-RV-G and anti-rnp antibody ELISA assays. Mouse serum was analyzed for anti-rv-g and anti-rnp total IgG, IgG1, and IgG2a antibodies by ELISA as described previously (15, 24, 26). Flow cytometry. (i) Surface staining. Isolated splenocytes were blocked for 1 h at 4 C with Fc Block (rat anti-mouse CD16/CD32; BD Biosciences) and unconjugated streptavidin. Cells were washed with fluorescence-activated cell sorting (FACS) buffer (2% bovine serum albumin [BSA] PBS) and stained for surface markers for 30 min at room temperature. Cells were washed with FACS buffer and fixed with 4% paraformaldehyde (Cytofix; BD Biosciences) for 20 min at 4 C. Surface markers were detected by phycoerythrin (PE)-Gag tetramer (H- 2K d -restricted AMQMLKETI epitope; Beckman Coulter), and antibodies FITC-CD44, peridinin chlorophyll protein (PerCP)-Cy5.5-CD8, and allophycocyanin (APC)-CD62L (BD Biosciences). Stained samples were analyzed on a BD FACSCalibur, which collected data on 200,000 events per sample. Live cells were gated by forward scatter/side scatter. Within the CD8 population, the percentage of activated (CD62L lo ) and Gag tetramer-specific (Gag-tetramer ) cells were detected. (ii) Internal cytokine staining (ICS). Splenocytes were plated into roundbottom plates with or without a stimulation cocktail of 10 g/ml AMQMLKETI Gag peptide (New England Peptide LLC), 1 g/ml CD49d, and 1 g/ml CD28 (both, BD Biosciences). After incubation for 2hat37 C in 5% CO 2, all samples were treated with 1 l/ml GolgiPlug (BD Biosciences) and allowed to incubate for four additional hours. Cells were blocked, stained for surface CD8, and fixed as described above. Cells were washed with FACS buffer and stained for internal molecules in the presence of BD Perm/Wash Buffer (BD Biosciences) for 30 min at room temperature. Antibodies used were PE-tumor necrosis factor (TNF- ), FITC-gamma interferon (IFN- ), and APC interleukin-2 (IL-2) (BD Biosciences). Cells were subsequently fixed with 4% paraformaldehyde (Cytofix; BD Biosciences) for 20 min at 4 C. Stained samples were analyzed on a BD FACSCalibur, which collected data on 400,000 events per sample. The percentage of cytokine-producing cells was calculated as the difference between Gag-stimulated and unstimulated samples [(Gag-stimulated percentage) (unstimulated percentage)]. IFN- ELISPOT assay. The 96-well enzyme-linked immunospot (ELISPOT) assay plates (MultiScreen-IP hydrophobic PVDF; Millipore) were coated with 10 g/ml rat anti-mouse IFN- antibody (BD Biosciences) and incubated overnight at 4 C. Plates were washed in 0.25% Tween 20 PBS, blocked with 5% BSA PBS for 1 h at 37 C, and incubated in splenocyte medium for 2 h. Splenocytes were plated in triplicate at three cell densities appropriate for the experiment (ranging from 10 4 to 10 6 cells/well). These cells were incubated with or without a stimulation cocktail of 10 g/ml AMQMLKETI Gag peptide (New England Peptide LLC), 1 g/ml CD49d, and 1 g/ml CD28 (both, BD Biosciences) for 18 h at 37 C in 5% CO 2. Plates were washed with 0.25% Tween 20 PBS followed by sterile distilled water to lyse residual cells. Wells were incubated with 5 g/ml biotinylated rat anti-mouse IFN- antibody (BD Biosciences) in 1% BSA PBS for2hat37 C in 5% CO 2. Following a washing step with 0.25% Tween 20 PBS, wells were incubated with 0.2 g/ml horseradish peroxidase-conjugated streptavidin (Jackson ImmunoResearch) in 1% BSA PBS for 2hat37 C in 5% CO 2. IFN- -secreting cells were detected by developing the plates with a solution of 3,3 -diaminobenzidine and 4-chloro-1-naphthol in methanol. The plates were scanned using an ImmunoSpot reader (Cellular Technology Ltd.), and the spots were counted using ImmunoSpot software (version 4.0; Cellular Technology Ltd.). Cytotoxic T-cell assay. Splenocyte lytic activity was measured by a nonradioactive calcein release assay, described previously (3, 34), with the following modifications. Briefly, P815 target cells (ATCC) cultured in DMEM supplemented with 5% FBS and penicillin-streptomycin were pulsed with either 10 g/ml AMQMLKETI HIV-1 Gag peptide (H2-K d restricted; New England Peptide LLC) or 10 g/ml IGPGRAFYAR HIV-1 Env peptide (H2-D d restricted; New England Peptide LLC), both immunodominant major histocompatibility complex class I (MHC-I)-restricted epitopes, for 18 h at 37 C in 5% CO 2. These P815 targets were washed and resuspended in HBSSF medium (Hanks balanced salt solution without Ca 2,Mg 2, or phenol red [Gibco], supplemented with 5% FBS, 10 mm HEPES, and 4 mm L-glutamine), labeled with 10 M calcein-am (Molecular Probes) for 30 min in a 37 C water bath, and washed with HBSSF medium. Target cells were then plated into round-bottom plates in triplicate at 10 4 cells/well with various concentrations of splenocytes (effectors) to reach final effector/target (E/T) ratios of 100, 33, 11, and 3.7. Plates were incubated for 3 h at 37 C in 5% CO 2. Cells were pelleted by spinning at 700 g for 5 min, and 100 l of the supernatant was transferred to a 96-well flat-bottom plate. Fluorescence was measured on a Victor multilabel counter (Wallac) scanning 1.0 s/well at 485-nm/535-nm wavelengths. Spontaneous release (SR) was measured by incubation of targets with HBSSF medium alone, and maximum release (MR) was measured by incubation of targets with lysis buffer (0.1% Triton X-100 in 50 mm sodium borate, ph 9.0). Percent lysis was calculated as follows: [(sample fluorescence SR fluorescence)/(mr fluorescence SR fluorescence)] 100. Statistical analysis. All data were analyzed by Prism software (GraphPad). Unpaired two-tailed t tests were used with the addition of Welch s correction if the variances were unequal. One-way analysis of variance (ANOVA) with Bonferroni correction was used for between-group comparisons, and a two-way ANOVA was used to deduce the total effect of virus verses treatment. RESULTS Construction and recovery of SPBN- G-Gag virus. The goal of this study was to establish a single-cycle RV as a vaccine vector using HIV-1 Gag as a model antigen. For this approach, we constructed a novel RV vector whereby the entire RV-G gene was deleted from pspbn-gag by utilizing restriction cites flanking the RV-G gene. This vector was designated SPBN- G-Gag (Fig. 1B), and infectious virus was recovered. We used BNSP-Gag (Fig. 1D), which encodes all five of the RV genes, in addition to Gag, as a replication-competent control virus for comparative analysis. While SPBN- G-Gag encodes Gag between the RV-M and RV-L genes, BNSP-Gag encodes Gag the between RV-N and RV-P genes. Because RV-G is required for virus spread (10, 13, 37, 44), we propagated SPBN- G-Gag on a BSR-RVG trans-complementing cell line. Virions collected from these harvests do not encode RV-G in their genome but are complemented with RV-G from the cell line. Virus produced in this manner is hereafter referred to as SPBN- G-Gag RVG. To circumvent loss of expression of RV-G (14), the complementing cell line was generated using the Tet-off inducible gene expression system whereby RV-G gene expression is off unless conditionally turned on by the removal of doxycycline (Fig. 2A). Though we have previously shown that the relative position of Gag within the RV genome does not impact either virus growth kinetics or Gag-specific immunogenicity (26), we sought to quantify the relative production of HIV-1 p24 by SPBN- G-Gag and BNSP-Gag viruses. Cell lysates collected from BSR cells infected at an MOI of 10 for 72 h showed that SPBN- G-Gag produced slightly less p24 than BNSP-Gag (15 and 20 ng/ml, respectively). SPBN- G-Gag RVG is a single-cycle RV in vitro. To evaluate the replication of SPBN- G-Gag RVG, we analyzed virus
4 VOL. 84, 2010 SINGLE-CYCLE RV VECTOR 2823 FIG. 2. SPBN- G-Gag is limited to single-cell infection in vitro. (A) Anti-RV-G surface staining of BSR-RVG trans-complementing cells induced to express RV-G in the absence of doxycycline (top) and not induced in the presence of doxycycline (bottom). (B) Virus spread assay on BSR or induced BSR-RVG cells infected with SPBN- G-Gag or BNSP-Gag virus at an MOI of for 72 h and stained for intracellular RV-N. Images in panels A and B are shown at 10 fluorescent microscope magnification. (C) Viral growth kinetics were demonstrated by multistep growth curves in which BSR or BSR-RVG cells are infected at an MOI of 0.01 with either BNSP-Gag or SPBN- G-Gag virus, and viral titers were determined from samples taken at 0, 24, 48, and 72 h postinfection. spread on BSR and trans-complementing BSR-RVG cells. Both cell lines were inoculated at an MOI of and 72 h later immunostained for the presence of RV-N (Fig. 2B). When SPBN- G-Gag RVG was grown on trans-complementing BSR-RVG cells, we detected the formation of foci similar in size and morphology to those generated by replication-competent BNSP-Gag. However, when SPBN- G-Gag RVG was used to infect BSR cells, only single infected cells were observed. These results demonstrated that SPBN- G-Gag RVG can infect cells, but the infection is restricted to the initially infected cell. To further characterize the growth kinetics, we conducted multistep growth curve assays in which BSR or BSR-RVG cells were infected at an MOI of 0.01, and the presence of infectious virus was determined at various time points by focus-forming assay. The results, shown in Fig. 2C, demonstrate that when SPBN- G-Gag RVG is grown on BSR cells, no infectious virus is detected in the supernatants. However, when propagated on the trans-complementing BSR-RVG cells, SPBN- G-Gag RVG grew to high titers of 10 7 to 10 8 focus-forming units (FFU)/ml, similar to replication-competent BNSP-Gag. Antibody profiles are comparable between single-cycle and replication-competent RVs. Replication-competent vectors are believed to have an immunogenic advantage over alternative vaccine strategies, including inactivated virions, due to their inherent ability to replicate and spread within the immunized host (12). It has previously been shown that replication-deficient RV generated by deletion of the RV-P retained the same balanced IgG2a/IgG1 antibody isotype profile as was observed for live replicating RV while inactivated RV induced a lower humoral response with an IgG1 bias (6). To determine if a G-deleted, single-cycle SPBN- G-Gag RVG virus induces antibody responses similar to replication-competent RV, we immunized groups of five BALB/c mice each with FFU of SPBN- G-Gag RVG or BNSP-Gag. Blood was collected at 15 days postimmunization, and an ELISA for detection of total IgG, IgG1, and IgG2a antibody against RV-G was performed (Fig. 3A and B). As expected, we detected significantly less (P 0.05) total IgG and IgG2a antibody for the SPBN- G- Gag RVG -immunized mice than for the BNSP-Gag-immunized mice (Fig. 3A). There was no detectable difference in IgG1 levels between the immunization groups. Despite this general decrease in anti-rv-g antibodies, the ratios of IgG2a/IgG1 were not statistically different between the two groups (Fig. 3B). Of note, SPBN- G-Gag RVG does not encode genomic RV-G. It was therefore important to measure the antibody responses against a protein encoded by both RVs. Thus, we conducted an anti-rnp ELISA for detection of antibodies against RV-N (Fig. 3C and D). Again, we detected significantly less (P 0.001) total IgG for SPBN- G-Gag RVG -immunized mice than for the BNSP-Gag-immunized mice (Fig. 3C), but no differences in either the IgG2a or IgG1 levels were observed. Likewise, the IgG2a/IgG1 ratios were not statistically different (Fig. 3D). To determine the humoral response to different doses of these vectors, blood was collected from mice 1 month after i.m. immunization with 10 5,10 4,10 3,10 2,or10 1 FFU of BNSP-Gag or SPBN- G-Gag RVG. Serum was analyzed for anti-rv-g total IgG antibodies by ELISA (Fig. 3E). Similar to what was initially observed (Fig. 3A), SPBN- G-Gag RVG induces fewer antibodies than BNSP-Gag at titers of 10 5,10 4, and 10 3 FFU (Fig. 3E). Results show that the number of anti-rv-g antibodies is dose dependent as they decrease linearly with decreasing immunization titers. No antibodies were detected for immunizations with 10 2 and 10 1 FFU. Lastly, analysis of the onset of anti-rv-g total IgG antibodies over time was conducted on serum collected from mice immunized with FFU of BNSP-Gag or SPBN- G-Gag RVG at 7, 16, 30, and 55 days postimmunization. Though lower, the overall kinetics of antibody development by SPBN- G-Gag RVG is comparable to that of BNSP-Gag (Fig. 3F). Together, these data demonstrate that while SPBN- G- Gag RVG induces lower antibody responses than BNSP-Gag, the response has the same balanced IgG isotype profile and kinetics as the replication-competent RV. Primary T-cell responses are equivalent between single-cycle and replication-competent RVs. CD8 T-cell responses are thought to be an important characteristic of a potential HIV
5 2824 GOMME ET AL. J. VIROL. Downloaded from FIG. 3. Single-cycle RV induces antibody profiles comparable to replication-competent RV. (A and C) Relative serum concentrations of anti-rv-g and anti-rnp total IgG, IgG2a, and IgG1 antibodies from serum diluted 1:60 and their respective IgG2a/IgG1 isotype ratios (B and D) from mice 15 days postimmunization with FFU of SPBN- G-Gag RVG or BNSP-Gag. (E) Mice immunized i.m. with different titers of SPBN- G-Gag RVG or BNSP-Gag were assayed at 1 month postimmunization for anti-rv-g total IgG antibody. (F) A time course showing total anti-rv-g IgG antibody levels from mice immunized i.m. with FFU of SPBN- G-Gag RVG or BNSP-Gag. (E and F) Results shown are for sera diluted 1:50. Significance between virus groups was measured by a t test. Values are the means standard errors. OD, optical density. *, P 0.05; **, P 0.01; and ***, P vaccine (25). Thus, to determine such cellular responses generated by a single-cycle RV, we compared the HIV-1 Gagspecific CD8 T-cell responses induced by priming with SPBN- G-Gag RVG to those induced by BNSP-Gag. For this approach, BALB/c mice immunized i.m. with FFU of BNSP-Gag or SPBN- G-Gag RVG were sacrificed at 7, 10, 15, and 20 days postimmunization (Fig. 4A). The splenocytes were then analyzed by flow cytometry (Fig. 4B) and an IFN- -ELISPOT assay (Fig. 4C) at each time point. In mice immunized with BNSP-Gag, we measured a significant increase, using one-way ANOVA with a Bonferroni correction, in both activated Gag tetramer-positive T cells and IFN- producing cells at day 10 postprime relative to naïve mice. Additionally, we compared the T-cell responses of the immunized mice averaged across all time points to the responses of the naïve mice using a two-way analysis of variance comparing virus type (BNSP-Gag or SPBN- G-Gag RVG ) to immunization (immunized or unimmunized). We found that the type of virus (replication-competent or single-cycle RV) had no effect on the response (for Gag tetramer-positive cells, P ; for IFN- production, P ), while the immunization did have a significant effect (Gag tetramer-positive cells, P ; IFN- production, P ). Together, these results indicate that priming with BNSP-Gag or SPBN- G-Gag RVG induces a significant number of Gag-specific CD8 T cells and that the magnitude of this primary response is equivalent between replication-competent and single-cycle RV vectors. To analyze whether the observed T-cell responses were de- on November 18, 2018 by guest
6 VOL. 84, 2010 SINGLE-CYCLE RV VECTOR 2825 FIG. 4. The magnitude and kinetics of primary T-cell responses are comparable for single-cycle and replication-competent RVs. (A) Schematic of mouse experiments for analysis of primary immune responses. BALB/c mice immunized i.m. with FFU of SPBN- G-Gag RVG or BNSP-Gag were sacrificed at 7, 10, 15, or 20 days postimmunization. Splenocytes were analyzed for activated Gag tetramer-positive T cells (Gag-tetramer CD62L lo ) (B) and IFN- -producing cells by ELISPOT assay (C). Sample groups were composed of 8 to 13 mice at each time point for each virus, with the exception of the naïve group and day 20 time point, which had 4 to 5 mice for each virus. Significance between time points within viral immunization groups was measured by one-way ANOVA with a Bonferroni correction. (D) Mice immunized i.m. with different titers of SPBN- G-Gag RVG or BNSP-Gag were assayed at 10 days postimmunization, and splenocytes were analyzed for IFN- -producing cells by ELISPOT assay. *, P 0.05; **, P pendent on the initial titers used for immunization, we performed a dose-response analysis. Groups of BALB/c mice were immunized i.m. with 10 6,10 5,or10 4 FFU of BNSP-Gag or SPBN- G-Gag RVG. At 10 days postprime, spleens were harvested, and splenocytes were analyzed by IFN- -ELISPOT assay (Fig. 4D). Interestingly, for both viruses, the number of IFN- -producing T cells remained consistently high in mice across all three immunization titers. Though there is a decrease in IFN- -producing cells in mice immunized with 10 6 and 10 5 FFU of SPBN- G-Gag RVG, there is no significant difference either between the 10 6 and 10 4 FFU groups or the 10 5 and 10 4 FFU groups, suggesting that there is no dose response. In contrast to the antibody responses, low immunization titers (10 4 FFU) with either virus were sufficient to induce T-cell responses equal to titers 100-fold higher (Fig. 4D). Therefore, RV-induced antibody responses are dose dependent while CD8 T-cell responses are not. Cytotoxic T lymphocyte (CTL) recall responses to challenge virus are equivalent in mice primed with single-cycle and replication-competent RV. As priming by both single-cycle and replication-competent RVs induce the formation of HIV-1 Gag-specific CD8 T cells, we next sought to determine the capacity of these T cells to expand and assume a cytolytic phenotype during a recall response to the HIV-1 Gag antigen. Groups of five BALB/c mice were immunized i.m. with FFU of either virus, rested 10 weeks, and then challenged i.p. with PFU of vaccinia virus expressing HIV-1 Gag (VV-Gag) (Fig. 5A). Spleens were harvested 4.5 days postchallenge, and splenocytes were analyzed by flow cytometry and IFN- -ELISPOT assay. Immunization by BNSP-Gag and SPBN- G-Gag RVG induced similar numbers of activated Gag tetramer-positive T cells, as depicted in Fig. 5B for single representative mice and in Fig. 5C for all mice analyzed (P ). As with the tetramer staining, we obtained similar numbers of IFN- -producing cells (Fig. 5D) (P ). Unstimulated splenocytes had no positive responders, demonstrating the specificity of the assay (data not shown). This indicates that single-cycle RV induces antigen-specific T cells that are functional in cytokine secretion. However, CD8 T-cell antiviral activity is mediated by both cytokine secretion and lytic activity. These two effector functions can be differentially regulated (1, 5, 19) yielding T cells that are antigen specific yet not functionally cytolytic. Thus, to determine the cytolytic capacity of the T cells generated by the
7 2826 GOMME ET AL. J. VIROL. Downloaded from FIG. 5. CTL recall responses are equal in magnitude and functionality in mice primed with single-cycle and replication-competent RVs. (A) Schematic of prime-challenge schedule. BALB/c mice were immunized i.m. with FFU of SPBN- G-Gag or BNSP-Gag and challenged i.p. with PFU of VV-Gag 10 weeks postimmunization. Splenocytes were analyzed at 4.5 days postchallenge for activated Gag-tetramerpositive T cells (Gag-tetramer CD62L lo ), as shown in a representative experiment on individual mice (B) and in summary for five mice per group (C). IFN- -producing cells were assayed by ELISPOT assay (D). (E and F) BALB/c mice were immunized i.m. with FFU of SPBN- G-Gag RVG or BNSP-Gag and challenged i.p. with PFU of VV-Gag 4 weeks postimmunization. Naïve mice were challenged as controls. After 4.5 days splenocytes were harvested and cultured with Gag-stimulated targets or irrelevant antigen Env-stimulated targets and assayed for lytic activity by a nonradioactive CTL assay. (E) The percent specific lysis was evaluated on individual samples and is shown as a mean standard error. (F) The same splenocytes used in the nonradioactive CTL assay were analyzed for activated Gag-tetramer-positive T cells, and a positive correlation between cytolytic activity and tetramer-positive staining was observed. Cytolytic data are representative of three independent experiments. single-cycle RV, BALB/c mice were immunized i.m. with FFU of SPBN- G-Gag RVG or BNSP-Gag, rested 4 weeks, and challenged i.p. with PFU of VV-Gag. Spleens were harvested 4.5 days postchallenge, and splenocytes were assayed for cytolytic activity by a nonradioactive CTL assay (calcein release assay). Splenocytes from both immunization groups exhibited lytic activity when cultured with Gag-stimulated target cells (Fig. 5E) and remained unresponsive when cultured with irrelevant Env-stimulated target cells (Fig. 5E). Naïve mice challenged with VV-Gag exhibited no detectable Gagspecific cytolytic activity. The percent specific lysis detected from BNSP-Gag immunization was equal to that of SPBN- G- Gag RVG (P ) at an E/T ratio of 100. Data are representative of three independent experiments. Of note, we found that the percent specific lysis positively correlated to the percent activated Gag tetramer-positive T cells (r ) (Fig. 5F). To evaluate the proliferative capacity of T cells generated from low-dose RV immunizations, we immunized groups of BALB/c mice i.m. with 10 5,10 4,10 3,10 2,or10 1 FFU of BNSP-Gag or SPBN- G-Gag RVG. Five weeks postimmunization, mice were challenged i.p. with PFU of VV-Gag, and spleens were harvested 4.5 days postchallenge. Splenocytes were analyzed by flow cytometry (Fig. 6A) and IFN- ELISPOT assay (Fig. 6B). Interestingly, for both viruses, the number of activated Gag tetramer-positive T cells remained consistently high in mice immunized with 10 5, 10 4, and 10 3 FFU and then dropped to background levels, as seen in naïve mice, with the titers 10 2 and 10 1 (Fig. 6A). Similar results were found for the number of IFN- - on November 18, 2018 by guest
8 VOL. 84, 2010 SINGLE-CYCLE RV VECTOR 2827 FIG. 6. Robust proliferation of antigen-specific T-cell responses following low-titer immunization with SPBN- G-Gag RVG. (A and B) Mice were immunized i.m. with different titers of SPBN- G-Gag RVG or BNSP-Gag and challenged i.p. with PFU of VV-Gag at 5 weeks postimmunization. Splenocytes were analyzed at 4.5 days postchallenge for activated Gag tetramer-positive T cells (Gag-tetramer CD62L lo ) (A) and IFN- -producing cells by ELISPOT assay (B). *, P producing cells (Fig. 6B). Statistical analysis showed a significant difference in IFN- -producing cells in mice immunized with 10 5 verses 10 3 FFU of SPBN- G-Gag RVG (Fig. 6B). However, direct comparison of BNSP-Gag-immunized mice with SPBN- G-Gag RVG -immunized mice at each specific titer (i.e., 10 5 versus 10 5 ) showed no significant differences, indicating that the single-cycle vector is just as potent as the replication-competent vector, even at very low titers. The complete lack of a cellular response in animals immunized with 10 2 or 10 1 FFU may be due to the fact that titers determined in vitro do not translate into infectivity in vivo. This correlates to the absence of detectable antibodies at these low titers (Fig. 3E). This dose response analysis demonstrates that, upon VV-Gag challenge, low-titer immunizations with single-cycle RV induce CD8 T-cell responses capable of expanding to levels as robust as high-dose replication-competent immunizations. In summary, our results demonstrate that single-cycle RVs induce CD8 T cells with effector functions, cytokine secretion and lytic activity, equally as potent and proliferative as those observed for the replication-competent vector, BNSP-Gag. This further illustrates the potential of single-cycle RV as a vaccine vector. Heterologous prime-boost with single-cycle RV enhances antigen-specific T-cell response. Vaccination strategies commonly include multiple inoculations, or boosters, to increase the antigen-specific immune responses and better protect the immunized host. In this next step, we sought to examine the capacity of our single-cycle RV as a booster vaccine. As RV-G is highly immunogenic, mice previously immunized with an RV-G expressing RV are likely to avoid reinfection with the same vector, as previously shown for replication-competent RV in mice and monkeys (29, 42). Thus, we sought to generate single-cycle RV for boosting that is complemented with a heterologous glycoprotein. For this we used a VSV-RVG chimera in which the ectodomain and transmembrane domains of the VSV-G (Indiana strain) are fused to the RV-G cytoplasmic domain (CD) (17). Previously, it was shown that replacing the CD of a foreign glycoprotein with that of RV-G promotes its incorporation into the RV envelope (30). To produce a vector containing this heterologous glycoprotein, we propagated SPBN- G-Gag on a BSR-VSV-RVG trans-complementing cell line. Like the BSR-RVG cells (Fig. 2), this cell line was generated using the Tet-off inducible gene expression system whereby VSV-RVG gene expression is off unless conditionally turned on by the removal of doxycycline (Fig. 7A). Virus produced in this manner was designated SPBN- G-Gag VSV-RVG. To evaluate replication of SPBN- G-Gag VSV-RVG on BSR- VSV-RVG cells, we analyzed virus spread (Fig. 7B) on induced or uninduced cells by inoculating them at an MOI of and 72 h later immunostaining the cells for the presence of RV-N. Foci were detected only when SPBN- G-Gag was grown on induced BSR-VSV-RVG cells (Fig. 7B, top). This demonstrates that SPBN- G-Gag VSV-RVG is a single-cycle virus like SPBN- G-Gag RVG. These findings were confirmed by multistep growth curve assays (Fig. 7C), which demonstrate that when SPBN- G-Gag is grown on BSR cells, no infectious virus is produced (Fig. 7C); however, when propagated on trans-complementing BSR-VSV-RVG cells, SPBN- G-Gag grew to titers of 10 6 to 10 7 FFU /ml, approximately 10-fold lower than growth of BNSP-Gag (Fig. 7C). This 10-fold decrease in viral titer may be due to less efficient budding due to the chimeric VSV RV-G, as was previously observed (17). To evaluate the capacity of heterologous single-cycle RV to increase HIV-1 Gag-specific immune responses, groups of five BALB/c mice were immunized i.m. with FFU of SPBN- G-Gag RVG and boosted i.m. at 6 weeks postimmunization with FFU of SPBN- G-Gag VSV-RVG (heterologous boost), FFU of SPBN- G-Gag RVG (homologous boost), or PBS (mock boost) (Fig. 8A). At 0, 5, 10, and 15 days postboost, mice were sacrificed, and splenocytes were assayed for IFN- production by ELISPOT assay (Fig. 8B), for activated Gag tetramer-positive T cells by flow cytometry (Fig. 8C), and for generally activated CD8 T cells (Fig. 8D). Additionally, cells were assayed for cytokine production by ICS (Fig. 8E to H). In all assays, we found that both the homologous boost and mock boost did not increase the numbers of Gag-specific CD8 T cells. This suggests that primary immunization by the single-cycle virus induces neutralizing antibodies that prevent a successful homologous boost, as shown previously (29, 42). However, the heterologous boost induced a significantly higher number of IFN- -producing cells (Fig. 8B), activated Gag tetramer-positive T cells (Fig. 8C), and generally activated CD8 T cells (Fig. 8D) than the homologously
9 2828 GOMME ET AL. J. VIROL. FIG. 7. Production of SPBN- G-Gag VSV-RVG complemented with chimeric VSV-RVG for heterologous prime-boost immunization. (A) Anti- VSV-G surface staining of BSR-VSV-RVG trans-complementing cells induced to express VSV-RVG in the absence of doxycycline (top) and not induced in the presence of doxycycline (bottom). (B) Virus spread assay on induced (top) or uninduced (bottom) BSR-VSV-RVG cells infected with SPBN- G-Gag at an MOI of for 72 h and stained for intracellular RV-N. Images in panels A and B are shown at 20 fluorescent microscope magnification. (C) Virus growth kinetics were demonstrated by multistep growth curves in which BSR or BSR-VSV-RVG cells were infected at an MOI of 0.01 with either BNSP-Gag or SPBN- G-Gag virus, and virus titers were determined from samples taken at 0, 24, 48, and 72 h postinfection. boosted mice. Importantly, the Gag-specific cells are also functional, as evidenced by their capacity to produce IFN-, IL-2, TNF-, and the dual expression of both TNF- and IFN- (Fig. 8B and E to H). Together, this demonstrates that the immune response generated by single-cycle RV can be significantly enhanced by boosting with a single-cycle RV complemented with a heterologous glycoprotein. DISCUSSION The need for an effective HIV-1 vaccine is greater than ever. Live viral vectors are at the forefront of vaccine development due to their capacity to generate strong humoral and cellular immune responses. However, the safety concerns associated with live, replicating vectors have proven to be a major obstacle in their approval for use in humans. As such, recent vaccine development has focused on making substantially attenuated vectors by truncation or deletion of viral genes directly involved with virus replication and spread (8, 18, 26, 33, 35, 36, 38). Here, we describe the development of a single-cycle RV, attenuated by deletion of RV-G and expressing HIV-1 Gag as a model antigen. Our immunogenicity studies demonstrate that single-cycle RV induced equally potent CD8 T-cell responses but lower vector-specific antibody responses than replication-competent RV following i.m. injection. Even though the induced humoral responses were lower, they were still potent enough to block a second infection with the same vector, indicating the potential of such vectors to induce neutralizing responses against other pathogens. The decision to delete the RV-G gene was made based on what is known about its critical role in the attachment and entry of RV into host cells. RV-G is one of the most important determinants of viral pathogenicity (10, 11, 37), and without it, RV particles are unable to spread in vitro or in vivo (13, 44). However, because RV-G has little known involvement in genome transcription and replication, a G-deleted vector would theoretically infect a cell and express its gene products just as efficiently as a fully replication-competent RV. This intracellular gene expression would provide ample antigen for presentation to both MHC-I and MHC-II pathways thought to contribute to live viral vector immunogenicity. Therefore, we hypothesized that a G-deleted, single-cycle RV would provide a safe, yet potent, alternative to our prototypic RV vector. The safety benefits are enhanced by the fact that, unlike viruses that are attenuated by point mutations, a single-cycle virus missing a large section of the genome is unlikely to revert back to its virulent form. This is especially true for negative-stranded viruses, including RV, where recombination is extremely rare and has been described for only respiratory syncytial virus (RSV) (41). In order to establish the relative immunogenicity of a singlecycle RV vector, we had to choose an appropriate replicationcompetent control virus for comparative analysis. VSV, a closely related rhabdovirus, has documented transcriptional attenuation of gene products in the 3 to 5 direction (20), and a similar mechanism of transcriptional attenuation has been suggested for RV (16). In order to utilize the most stringent control with equal or greater expression of the model antigen, HIV-1 Gag, we chose BNSP-Gag as our replication control virus. Indeed, when infected cell lysate was assayed for p24, we confirmed that SPBN- G-Gag produces just slightly less p24 than BNSP-Gag (15 and 20 ng/ml, respectively). This decrease may be attributed to changes in virus transcription that occur from removal of an entire gene segment. However, we have previously shown that the relative position of Gag within the RV genome does not impact virus growth kinetics (26), and this was confirmed in growth curves for SPBN- G-Gag and BNSP-Gag (Fig. 2C). Vector-specific antibody responses were lower for the singlecycle vector than for the replication-competent vector. However, unlike inactivated RV (20), the isotype profiles generated by single-cycle vectors were identical to those of replicationcompetent vectors, indicating the same balanced Th1/Th2 immune responses. Th1 responses are particularly important for
10 VOL. 84, 2010 SINGLE-CYCLE RV VECTOR 2829 Downloaded from FIG. 8. Heterologous boost with single-cycle RV generates enhanced Gag-specific immune responses. (A) Schematic of prime-boost schedule. Five BALB/c mice per group were immunized i.m. with FFU of SPBN- G-Gag RVG and boosted i.m. at 6 weeks postimmunization with FFU of SPBN- G-Gag VSV-RVG (heterologous boost), FFU of SPBN- G-Gag RVG (homologous boost), or PBS (mock boost). Splenocytes were analyzed by ICS at 0, 5, 10, and 15 days postboost for IFN- -producing cells by ELISPOT assay (B), for activated Gag-tetramerpositive T cells (Gag-tetramer CD62L lo ) (C), for generally activated CD8 T cells (D), and for cytokine production (E to H). Significant differences between heterologous and homologous boosting groups at a single time point were measured by a t test (indicated by an asterisk above a time point). *, P 0.05; **, P 0.01; and ***, P Significant differences between time points for heterologously boosted mice only were measured by one-way ANOVA with a Bonferroni correction (indicated by pound signs spanning two time points). #, P 0.05; ##, P 0.01; ###, P the efficacy of a vaccine targeted for intracellular pathogens due to their known cytokine-mediated antiviral activities and macrophage activation. We showed that anti-rv-g antibodies are dose dependent (Fig. 3E), suggesting that higher immunization titers could be used if high antibody levels are sought, as is done for other replication-deficient vectors (18, 33, 35). It is still interesting that a G-deleted RV induces such substantial anti-rv-g antibodies, and based on the ineffective homologous boost, these antibodies appear to be neutralizing. Considering that the same was observed for anti-rnp antibodies, this would suggest that comparable humoral responses may be generated against other vector-encoded proteins, including foreign antigens. Despite the lower humoral response to single-cycle RV, the CD8 T-cell responses were equal in quality and quantity to those of replication-competent RV. While the primary T-cell response is relatively low, it is clear that these Gag-specific T-cell precursors effectively expand in a robust secondary recall response in the event of a VV-Gag challenge, yielding highly functional, cytokine-secreting CTLs. Of note, these recall responses were reached at titers as low as 10 3 FFU of single-cycle or replication-competent RV. This is in contrast to immunization titers in the order of 10 9 PFU used for recombinant VV (rvv) and its replication-deficient counterpart, rmva, as well as the replication-competent and -deficient Ad5 vectors (18, 33, 35). The CD8 T cells induced by single-cycle RV exhibited potent CTL function as well as the capacity to expand to heterologous boost or challenge. This is significant because other RNA viruses, including lymphocytic choriomeningitis virus (LCMV) and hepatitis C virus (HCV), are known to induce functionally unresponsive T cells, usually a consequence of low-level or persistent infections (1, 5, 19). Furthermore, because rhabdoviruses can incorporate foreign glycoproteins into viral particles (17, 30, 31), we were able to develop a single-cycle booster virus complemented with VSV-RVG that significantly enhances the cellular immune response. We showed that boosting with a single-cycle RV enhances frequency and functionality of antigen-specific T cells. Enhanced on November 18, 2018 by guest
Supporting Information
 Supporting Information Valkenburg et al. 10.1073/pnas.1403684111 SI Materials and Methods ELISA and Microneutralization. Sera were treated with Receptor Destroying Enzyme II (RDE II, Accurate) before ELISA
Supporting Information Valkenburg et al. 10.1073/pnas.1403684111 SI Materials and Methods ELISA and Microneutralization. Sera were treated with Receptor Destroying Enzyme II (RDE II, Accurate) before ELISA
Gladstone Institutes, University of California (UCSF), San Francisco, USA
 Fluorescence-linked Antigen Quantification (FLAQ) Assay for Fast Quantification of HIV-1 p24 Gag Marianne Gesner, Mekhala Maiti, Robert Grant and Marielle Cavrois * Gladstone Institutes, University of
Fluorescence-linked Antigen Quantification (FLAQ) Assay for Fast Quantification of HIV-1 p24 Gag Marianne Gesner, Mekhala Maiti, Robert Grant and Marielle Cavrois * Gladstone Institutes, University of
SUPPLEMENTARY INFORMATION
 Complete but curtailed T-cell response to very-low-affinity antigen Dietmar Zehn, Sarah Y. Lee & Michael J. Bevan Supp. Fig. 1: TCR chain usage among endogenous K b /Ova reactive T cells. C57BL/6 mice
Complete but curtailed T-cell response to very-low-affinity antigen Dietmar Zehn, Sarah Y. Lee & Michael J. Bevan Supp. Fig. 1: TCR chain usage among endogenous K b /Ova reactive T cells. C57BL/6 mice
GOVX-B11: A Clade B HIV Vaccine for the Developed World
 GeoVax Labs, Inc. 19 Lake Park Drive Suite 3 Atlanta, GA 3 (678) 384-72 GOVX-B11: A Clade B HIV Vaccine for the Developed World Executive summary: GOVX-B11 is a Clade B HIV vaccine targeted for use in
GeoVax Labs, Inc. 19 Lake Park Drive Suite 3 Atlanta, GA 3 (678) 384-72 GOVX-B11: A Clade B HIV Vaccine for the Developed World Executive summary: GOVX-B11 is a Clade B HIV vaccine targeted for use in
Blocking antibodies and peptides. Rat anti-mouse PD-1 (29F.1A12, rat IgG2a, k), PD-
 Supplementary Methods Blocking antibodies and peptides. Rat anti-mouse PD-1 (29F.1A12, rat IgG2a, k), PD- L1 (10F.9G2, rat IgG2b, k), and PD-L2 (3.2, mouse IgG1) have been described (24). Anti-CTLA-4 (clone
Supplementary Methods Blocking antibodies and peptides. Rat anti-mouse PD-1 (29F.1A12, rat IgG2a, k), PD- L1 (10F.9G2, rat IgG2b, k), and PD-L2 (3.2, mouse IgG1) have been described (24). Anti-CTLA-4 (clone
Current Strategies in HIV-1 Vaccine Development Using Replication-Defective Adenovirus as a Case Study
 Note: I have added some clarifying comments to the slides -- please click on Comments under View to see them. Current Strategies in HIV-1 Vaccine Development Using Replication-Defective Adenovirus as a
Note: I have added some clarifying comments to the slides -- please click on Comments under View to see them. Current Strategies in HIV-1 Vaccine Development Using Replication-Defective Adenovirus as a
Recombinant Vaccinia Virus-Induced T-Cell Immunity: Quantitation of the Response to the Virus Vector and the Foreign Epitope
 JOURNAL OF VIROLOGY, Apr. 2002, p. 3329 3337 Vol. 76, No. 7 0022-538X/02/$04.00 0 DOI: 10.1128/JVI.76.7.3329 3337.2002 Copyright 2002, American Society for Microbiology. All Rights Reserved. Recombinant
JOURNAL OF VIROLOGY, Apr. 2002, p. 3329 3337 Vol. 76, No. 7 0022-538X/02/$04.00 0 DOI: 10.1128/JVI.76.7.3329 3337.2002 Copyright 2002, American Society for Microbiology. All Rights Reserved. Recombinant
Critical Role for Alpha/Beta and Gamma Interferons in Persistence of Lymphocytic Choriomeningitis Virus by Clonal Exhaustion of Cytotoxic T Cells
 JOURNAL OF VIROLOGY, Sept. 2001, p. 8407 8423 Vol. 75, No. 18 0022-538X/01/$04.00 0 DOI: 10.1128/JVI.75.18.8407 8423.2001 Copyright 2001, American Society for Microbiology. All Rights Reserved. Critical
JOURNAL OF VIROLOGY, Sept. 2001, p. 8407 8423 Vol. 75, No. 18 0022-538X/01/$04.00 0 DOI: 10.1128/JVI.75.18.8407 8423.2001 Copyright 2001, American Society for Microbiology. All Rights Reserved. Critical
Fayth K. Yoshimura, Ph.D. September 7, of 7 HIV - BASIC PROPERTIES
 1 of 7 I. Viral Origin. A. Retrovirus - animal lentiviruses. HIV - BASIC PROPERTIES 1. HIV is a member of the Retrovirus family and more specifically it is a member of the Lentivirus genus of this family.
1 of 7 I. Viral Origin. A. Retrovirus - animal lentiviruses. HIV - BASIC PROPERTIES 1. HIV is a member of the Retrovirus family and more specifically it is a member of the Lentivirus genus of this family.
Supplementary Figures
 Inhibition of Pulmonary Anti Bacterial Defense by IFN γ During Recovery from Influenza Infection By Keer Sun and Dennis W. Metzger Supplementary Figures d a Ly6G Percentage survival f 1 75 5 1 25 1 5 1
Inhibition of Pulmonary Anti Bacterial Defense by IFN γ During Recovery from Influenza Infection By Keer Sun and Dennis W. Metzger Supplementary Figures d a Ly6G Percentage survival f 1 75 5 1 25 1 5 1
Detailed step-by-step operating procedures for NK cell and CTL degranulation assays
 Supplemental methods Detailed step-by-step operating procedures for NK cell and CTL degranulation assays Materials PBMC isolated from patients, relatives and healthy donors as control K562 cells (ATCC,
Supplemental methods Detailed step-by-step operating procedures for NK cell and CTL degranulation assays Materials PBMC isolated from patients, relatives and healthy donors as control K562 cells (ATCC,
Supplementary Materials for
 immunology.sciencemag.org/cgi/content/full/2/16/eaan6049/dc1 Supplementary Materials for Enzymatic synthesis of core 2 O-glycans governs the tissue-trafficking potential of memory CD8 + T cells Jossef
immunology.sciencemag.org/cgi/content/full/2/16/eaan6049/dc1 Supplementary Materials for Enzymatic synthesis of core 2 O-glycans governs the tissue-trafficking potential of memory CD8 + T cells Jossef
Ex vivo Human Antigen-specific T Cell Proliferation and Degranulation Willemijn Hobo 1, Wieger Norde 1 and Harry Dolstra 2*
 Ex vivo Human Antigen-specific T Cell Proliferation and Degranulation Willemijn Hobo 1, Wieger Norde 1 and Harry Dolstra 2* 1 Department of Laboratory Medicine - Laboratory of Hematology, Radboud University
Ex vivo Human Antigen-specific T Cell Proliferation and Degranulation Willemijn Hobo 1, Wieger Norde 1 and Harry Dolstra 2* 1 Department of Laboratory Medicine - Laboratory of Hematology, Radboud University
MHC Tetramers and Monomers for Immuno-Oncology and Autoimmunity Drug Discovery
 MHC Tetramers and Monomers for Immuno-Oncology and Autoimmunity Drug Discovery Your Partner in Drug Discovery and Research MHC Tetramer Background T-Cell Receptors recognize and bind to complexes composed
MHC Tetramers and Monomers for Immuno-Oncology and Autoimmunity Drug Discovery Your Partner in Drug Discovery and Research MHC Tetramer Background T-Cell Receptors recognize and bind to complexes composed
Viral Persistence Alters CD8 T-Cell Immunodominance and Tissue Distribution and Results in Distinct Stages of Functional Impairment
 JOURNAL OF VIROLOGY, Apr. 2003, p. 4911 4927 Vol. 77, No. 8 0022-538X/03/$08.00 0 DOI: 10.1128/JVI.77.8.4911 4927.2003 Copyright 2003, American Society for Microbiology. All Rights Reserved. Viral Persistence
JOURNAL OF VIROLOGY, Apr. 2003, p. 4911 4927 Vol. 77, No. 8 0022-538X/03/$08.00 0 DOI: 10.1128/JVI.77.8.4911 4927.2003 Copyright 2003, American Society for Microbiology. All Rights Reserved. Viral Persistence
The Adaptive Immune Responses
 The Adaptive Immune Responses The two arms of the immune responses are; 1) the cell mediated, and 2) the humoral responses. In this chapter we will discuss the two responses in detail and we will start
The Adaptive Immune Responses The two arms of the immune responses are; 1) the cell mediated, and 2) the humoral responses. In this chapter we will discuss the two responses in detail and we will start
The humoral immune responses to IBV proteins.
 The humoral immune responses to IBV proteins. E. Dan Heller and Rosa Meir The Hebrew University of Jerusalem, Israel COST FA1207 meeting WG2 + WG3, Budapest, Jan. 2015 1 IBV encodes four major structural
The humoral immune responses to IBV proteins. E. Dan Heller and Rosa Meir The Hebrew University of Jerusalem, Israel COST FA1207 meeting WG2 + WG3, Budapest, Jan. 2015 1 IBV encodes four major structural
VIROLOGY. Engineering Viral Genomes: Retrovirus Vectors
 VIROLOGY Engineering Viral Genomes: Retrovirus Vectors Viral vectors Retrovirus replicative cycle Most mammalian retroviruses use trna PRO, trna Lys3, trna Lys1,2 The partially unfolded trna is annealed
VIROLOGY Engineering Viral Genomes: Retrovirus Vectors Viral vectors Retrovirus replicative cycle Most mammalian retroviruses use trna PRO, trna Lys3, trna Lys1,2 The partially unfolded trna is annealed
HIV-1 Virus-like Particle Budding Assay Nathan H Vande Burgt, Luis J Cocka * and Paul Bates
 HIV-1 Virus-like Particle Budding Assay Nathan H Vande Burgt, Luis J Cocka * and Paul Bates Department of Microbiology, Perelman School of Medicine at the University of Pennsylvania, Philadelphia, USA
HIV-1 Virus-like Particle Budding Assay Nathan H Vande Burgt, Luis J Cocka * and Paul Bates Department of Microbiology, Perelman School of Medicine at the University of Pennsylvania, Philadelphia, USA
Third line of Defense
 Chapter 15 Specific Immunity and Immunization Topics -3 rd of Defense - B cells - T cells - Specific Immunities Third line of Defense Specific immunity is a complex interaction of immune cells (leukocytes)
Chapter 15 Specific Immunity and Immunization Topics -3 rd of Defense - B cells - T cells - Specific Immunities Third line of Defense Specific immunity is a complex interaction of immune cells (leukocytes)
Recombinant adeno-associated virus vectors induce functionally impaired transgene product specific CD8 + T cells in mice
 Recombinant adeno-associated virus vectors induce functionally impaired transgene product specific CD8 + T cells in mice Shih-Wen Lin,, Marcio O. Lasaro, Hildegund C.J. Ertl J Clin Invest. 2007;117(12):3958-3970.
Recombinant adeno-associated virus vectors induce functionally impaired transgene product specific CD8 + T cells in mice Shih-Wen Lin,, Marcio O. Lasaro, Hildegund C.J. Ertl J Clin Invest. 2007;117(12):3958-3970.
Supplementary Data Table of Contents:
 Supplementary Data Table of Contents: - Supplementary Methods - Supplementary Figures S1(A-B) - Supplementary Figures S2 (A-B) - Supplementary Figures S3 - Supplementary Figures S4(A-B) - Supplementary
Supplementary Data Table of Contents: - Supplementary Methods - Supplementary Figures S1(A-B) - Supplementary Figures S2 (A-B) - Supplementary Figures S3 - Supplementary Figures S4(A-B) - Supplementary
Supporting Online Material for
 www.sciencemag.org/cgi/content/full/1175194/dc1 Supporting Online Material for A Vital Role for Interleukin-21 in the Control of a Chronic Viral Infection John S. Yi, Ming Du, Allan J. Zajac* *To whom
www.sciencemag.org/cgi/content/full/1175194/dc1 Supporting Online Material for A Vital Role for Interleukin-21 in the Control of a Chronic Viral Infection John S. Yi, Ming Du, Allan J. Zajac* *To whom
Rabies virus infection induces type I interferon production in an IPS-1 dependent manner while dendritic cell activation relies on IFNAR signaling.
 Thomas Jefferson University Jefferson Digital Commons Department of Microbiology and Immunology Faculty Papers Department of Microbiology and Immunology 7-1-2010 Rabies virus infection induces type I interferon
Thomas Jefferson University Jefferson Digital Commons Department of Microbiology and Immunology Faculty Papers Department of Microbiology and Immunology 7-1-2010 Rabies virus infection induces type I interferon
Medical Virology Immunology. Dr. Sameer Naji, MB, BCh, PhD (UK) Head of Basic Medical Sciences Dept. Faculty of Medicine The Hashemite University
 Medical Virology Immunology Dr. Sameer Naji, MB, BCh, PhD (UK) Head of Basic Medical Sciences Dept. Faculty of Medicine The Hashemite University Human blood cells Phases of immune responses Microbe Naïve
Medical Virology Immunology Dr. Sameer Naji, MB, BCh, PhD (UK) Head of Basic Medical Sciences Dept. Faculty of Medicine The Hashemite University Human blood cells Phases of immune responses Microbe Naïve
Optimizing Intracellular Flow Cytometry:
 Optimizing Intracellular Flow Cytometry: Simultaneous Detection of Cytokines and Transcription Factors An encore presentation by Jurg Rohrer, PhD, BD Biosciences 10.26.10 Outline Introduction Cytokines
Optimizing Intracellular Flow Cytometry: Simultaneous Detection of Cytokines and Transcription Factors An encore presentation by Jurg Rohrer, PhD, BD Biosciences 10.26.10 Outline Introduction Cytokines
HIV-1 p24 ELISA Pair Set Cat#: orb54951 (ELISA Manual)
 HIV-1 p24 ELISA Pair Set Cat#: orb54951 (ELISA Manual) BACKGROUND Human Immunodeficiency Virus ( HIV ) can be divided into two major types, HIV type 1 (HIV-1) and HIV type 2 (HIV-2). HIV-1 is related to
HIV-1 p24 ELISA Pair Set Cat#: orb54951 (ELISA Manual) BACKGROUND Human Immunodeficiency Virus ( HIV ) can be divided into two major types, HIV type 1 (HIV-1) and HIV type 2 (HIV-2). HIV-1 is related to
Increased IL-12 induced STAT-4 signaling in CD8 T cells. from aged mice
 Increased IL-2 induced STAT-4 signaling in CD8 T cells from aged mice Erin Rottinghaus * Abstract: Aging is associated with poor immune function leading to increased susceptibility to infectious diseases
Increased IL-2 induced STAT-4 signaling in CD8 T cells from aged mice Erin Rottinghaus * Abstract: Aging is associated with poor immune function leading to increased susceptibility to infectious diseases
Recombinant adeno-associated virus vectors induce functionally impaired transgene product specific CD8 + T cells in mice
 Research article Recombinant adeno-associated virus vectors induce functionally impaired transgene product specific CD8 + T cells in mice Shih-Wen Lin, 1,2 Scott E. Hensley, 1,2 Nia Tatsis, 2 Marcio O.
Research article Recombinant adeno-associated virus vectors induce functionally impaired transgene product specific CD8 + T cells in mice Shih-Wen Lin, 1,2 Scott E. Hensley, 1,2 Nia Tatsis, 2 Marcio O.
A VACCINE FOR HIV BIOE 301 LECTURE 10 MITALI BANERJEE HAART
 BIOE 301 LECTURE 10 MITALI BANERJEE A VACCINE FOR HIV HIV HAART Visit wikipedia.org and learn the mechanism of action of the five classes of antiretroviral drugs. (1) Reverse transcriptase inhibitors (RTIs)
BIOE 301 LECTURE 10 MITALI BANERJEE A VACCINE FOR HIV HIV HAART Visit wikipedia.org and learn the mechanism of action of the five classes of antiretroviral drugs. (1) Reverse transcriptase inhibitors (RTIs)
Immunization with Single-Cycle SIV Significantly Reduces Viral Loads After an Intravenous Challenge with SIV mac 239
 Immunization with Single-Cycle SIV Significantly Reduces Viral Loads After an Intravenous Challenge with SIV mac 239 Bin Jia 1, Sharon K. Ng 1, M. Quinn DeGottardi 1, Michael Piatak Jr. 2, Eloísa Yuste
Immunization with Single-Cycle SIV Significantly Reduces Viral Loads After an Intravenous Challenge with SIV mac 239 Bin Jia 1, Sharon K. Ng 1, M. Quinn DeGottardi 1, Michael Piatak Jr. 2, Eloísa Yuste
Supplementary Data 1. Alanine substitutions and position variants of APNCYGNIPL. Applied in
 Supplementary Data 1. Alanine substitutions and position variants of APNCYGNIPL. Applied in Supplementary Fig. 2 Substitution Sequence Position variant Sequence original APNCYGNIPL original APNCYGNIPL
Supplementary Data 1. Alanine substitutions and position variants of APNCYGNIPL. Applied in Supplementary Fig. 2 Substitution Sequence Position variant Sequence original APNCYGNIPL original APNCYGNIPL
Application of μmacs Streptavidin MicroBeads for the analysis of HIV-1 directly from patient plasma
 Excerpt from MACS&more Vol 8 1/2004 Application of μmacs Streptavidin MicroBeads for the analysis of HIV-1 directly from patient plasma L. Davis Lupo and Salvatore T. Butera HIV and Retrovirology Branch,
Excerpt from MACS&more Vol 8 1/2004 Application of μmacs Streptavidin MicroBeads for the analysis of HIV-1 directly from patient plasma L. Davis Lupo and Salvatore T. Butera HIV and Retrovirology Branch,
Received 3 September 2002/Accepted 15 January 2003
 JOURNAL OF VIROLOGY, Apr. 2003, p. 4646 4657 Vol. 77, No. 8 0022-538X/03/$08.00 0 DOI: 10.1128/JVI.77.8.4646 4657.2003 Copyright 2003, American Society for Microbiology. All Rights Reserved. Ability of
JOURNAL OF VIROLOGY, Apr. 2003, p. 4646 4657 Vol. 77, No. 8 0022-538X/03/$08.00 0 DOI: 10.1128/JVI.77.8.4646 4657.2003 Copyright 2003, American Society for Microbiology. All Rights Reserved. Ability of
Zheng, BJ; Du, LY; Zhao, GY; Lin, YP; Sui, HY; Chan, C; Ma, S; Guan, Y; Yuen, KY. Citation Hong Kong Medical Journal, 2008, v. 14 suppl. 4, p.
 Title Studies of SARS virus vaccines Author(s) Zheng, BJ; Du, LY; Zhao, GY; Lin, YP; Sui, HY; Chan, C; Ma, S; Guan, Y; Yuen, KY Citation Hong Kong Medical Journal, 2008, v. 14 suppl. 4, p. 39-43 Issued
Title Studies of SARS virus vaccines Author(s) Zheng, BJ; Du, LY; Zhao, GY; Lin, YP; Sui, HY; Chan, C; Ma, S; Guan, Y; Yuen, KY Citation Hong Kong Medical Journal, 2008, v. 14 suppl. 4, p. 39-43 Issued
Human Immunodeficiency Virus type 1 (HIV-1) p24 / Capsid Protein p24 ELISA Pair Set
 Human Immunodeficiency Virus type 1 (HIV-1) p24 / Capsid Protein p24 ELISA Pair Set Catalog Number : SEK11695 To achieve the best assay results, this manual must be read carefully before using this product
Human Immunodeficiency Virus type 1 (HIV-1) p24 / Capsid Protein p24 ELISA Pair Set Catalog Number : SEK11695 To achieve the best assay results, this manual must be read carefully before using this product
Interferon γ regulates idiopathic pneumonia syndrome, a. Th17 + CD4 + T-cell-mediated GvH disease
 Interferon γ regulates idiopathic pneumonia syndrome, a Th17 + CD4 + T-cell-mediated GvH disease Nora Mauermann, Julia Burian, Christophe von Garnier, Stefan Dirnhofer, Davide Germano, Christine Schuett,
Interferon γ regulates idiopathic pneumonia syndrome, a Th17 + CD4 + T-cell-mediated GvH disease Nora Mauermann, Julia Burian, Christophe von Garnier, Stefan Dirnhofer, Davide Germano, Christine Schuett,
Nature Medicine: doi: /nm.4322
 1 2 3 4 5 6 7 8 9 10 11 Supplementary Figure 1. Predicted RNA structure of 3 UTR and sequence alignment of deleted nucleotides. (a) Predicted RNA secondary structure of ZIKV 3 UTR. The stem-loop structure
1 2 3 4 5 6 7 8 9 10 11 Supplementary Figure 1. Predicted RNA structure of 3 UTR and sequence alignment of deleted nucleotides. (a) Predicted RNA secondary structure of ZIKV 3 UTR. The stem-loop structure
CHAPTER 4 IMMUNOLOGICAL TECHNIQUES
 CHAPTER 4 IMMUNOLOGICAL TECHNIQUES Nitroblue Tetrazolium Chloride (NBT) Reduction test NBT reduction test was evaluated by employing the method described by Hudson and Hay,1989 based upon principle that
CHAPTER 4 IMMUNOLOGICAL TECHNIQUES Nitroblue Tetrazolium Chloride (NBT) Reduction test NBT reduction test was evaluated by employing the method described by Hudson and Hay,1989 based upon principle that
Optimizing Intracellular Flow Cytometry:
 Optimizing Intracellular Flow Cytometry: Simultaneous Detection of Cytokines and Transcription Factors Presented by Jurg Rohrer, PhD, BD Biosciences 23-10780-00 Outline Introduction Cytokines Transcription
Optimizing Intracellular Flow Cytometry: Simultaneous Detection of Cytokines and Transcription Factors Presented by Jurg Rohrer, PhD, BD Biosciences 23-10780-00 Outline Introduction Cytokines Transcription
Commercially available HLA Class II tetramers (Beckman Coulter) conjugated to
 Class II tetramer staining Commercially available HLA Class II tetramers (Beckman Coulter) conjugated to PE were combined with dominant HIV epitopes (DRB1*0101-DRFYKTLRAEQASQEV, DRB1*0301- PEKEVLVWKFDSRLAFHH,
Class II tetramer staining Commercially available HLA Class II tetramers (Beckman Coulter) conjugated to PE were combined with dominant HIV epitopes (DRB1*0101-DRFYKTLRAEQASQEV, DRB1*0301- PEKEVLVWKFDSRLAFHH,
Immunization with single-cycle SIV significantly reduces viral loads after an intravenous challenge with SIV(mac)239
 University of Massachusetts Medical School escholarship@umms Preventive and Behavioral Medicine Publications and Presentations Preventive and Behavioral Medicine 1-23-2009 Immunization with single-cycle
University of Massachusetts Medical School escholarship@umms Preventive and Behavioral Medicine Publications and Presentations Preventive and Behavioral Medicine 1-23-2009 Immunization with single-cycle
Gene Vaccine Dr. Sina Soleimani
 Gene Vaccine Dr. Sina Soleimani Human Viral Vaccines Quality Control Laboratory (HVVQC) Titles 1. A short Introduction of Vaccine History 2. First Lineage of Vaccines 3. Second Lineage of Vaccines 3. New
Gene Vaccine Dr. Sina Soleimani Human Viral Vaccines Quality Control Laboratory (HVVQC) Titles 1. A short Introduction of Vaccine History 2. First Lineage of Vaccines 3. Second Lineage of Vaccines 3. New
Human Immunodeficiency Virus Type-1 Myeloid Derived Suppressor Cells Inhibit Cytomegalovirus Inflammation through Interleukin-27 and B7-H4
 Human Immunodeficiency Virus Type-1 Myeloid Derived Suppressor Cells Inhibit Cytomegalovirus Inflammation through Interleukin-27 and B7-H4 Ankita Garg, Rodney Trout and Stephen A. Spector,,* Department
Human Immunodeficiency Virus Type-1 Myeloid Derived Suppressor Cells Inhibit Cytomegalovirus Inflammation through Interleukin-27 and B7-H4 Ankita Garg, Rodney Trout and Stephen A. Spector,,* Department
Supplementary information. MARCH8 inhibits HIV-1 infection by reducing virion incorporation of envelope glycoproteins
 Supplementary information inhibits HIV-1 infection by reducing virion incorporation of envelope glycoproteins Takuya Tada, Yanzhao Zhang, Takayoshi Koyama, Minoru Tobiume, Yasuko Tsunetsugu-Yokota, Shoji
Supplementary information inhibits HIV-1 infection by reducing virion incorporation of envelope glycoproteins Takuya Tada, Yanzhao Zhang, Takayoshi Koyama, Minoru Tobiume, Yasuko Tsunetsugu-Yokota, Shoji
Supplementary Figures
 Supplementary Figures Supplementary Fig. 1. Surface thiol groups and reduction of activated T cells. (a) Activated CD8 + T-cells have high expression levels of free thiol groups on cell surface proteins.
Supplementary Figures Supplementary Fig. 1. Surface thiol groups and reduction of activated T cells. (a) Activated CD8 + T-cells have high expression levels of free thiol groups on cell surface proteins.
7.012 Quiz 3 Answers
 MIT Biology Department 7.012: Introductory Biology - Fall 2004 Instructors: Professor Eric Lander, Professor Robert A. Weinberg, Dr. Claudette Gardel Friday 11/12/04 7.012 Quiz 3 Answers A > 85 B 72-84
MIT Biology Department 7.012: Introductory Biology - Fall 2004 Instructors: Professor Eric Lander, Professor Robert A. Weinberg, Dr. Claudette Gardel Friday 11/12/04 7.012 Quiz 3 Answers A > 85 B 72-84
L-selectin Is Essential for Delivery of Activated CD8 + T Cells to Virus-Infected Organs for Protective Immunity
 Cell Reports Supplemental Information L-selectin Is Essential for Delivery of Activated CD8 + T Cells to Virus-Infected Organs for Protective Immunity Rebar N. Mohammed, H. Angharad Watson, Miriam Vigar,
Cell Reports Supplemental Information L-selectin Is Essential for Delivery of Activated CD8 + T Cells to Virus-Infected Organs for Protective Immunity Rebar N. Mohammed, H. Angharad Watson, Miriam Vigar,
Lecture 11. Immunology and disease: parasite antigenic diversity
 Lecture 11 Immunology and disease: parasite antigenic diversity RNAi interference video and tutorial (you are responsible for this material, so check it out.) http://www.pbs.org/wgbh/nova/sciencenow/3210/02.html
Lecture 11 Immunology and disease: parasite antigenic diversity RNAi interference video and tutorial (you are responsible for this material, so check it out.) http://www.pbs.org/wgbh/nova/sciencenow/3210/02.html
Shenghua Zhou, Rong Ou, Lei Huang, and Demetrius Moskophidis*
 JOURNAL OF VIROLOGY, Jan. 2002, p. 829 840 Vol. 76, No. 2 0022-538X/02/$04.00 0 DOI: 10.1128/JVI.76.2.829 840.2002 Copyright 2002, American Society for Microbiology. All Rights Reserved. Critical Role
JOURNAL OF VIROLOGY, Jan. 2002, p. 829 840 Vol. 76, No. 2 0022-538X/02/$04.00 0 DOI: 10.1128/JVI.76.2.829 840.2002 Copyright 2002, American Society for Microbiology. All Rights Reserved. Critical Role
~Lentivirus production~
 ~Lentivirus production~ May 30, 2008 RNAi core R&D group member Lentivirus Production Session Lentivirus!!! Is it health threatening to lab technician? What s so good about this RNAi library? How to produce
~Lentivirus production~ May 30, 2008 RNAi core R&D group member Lentivirus Production Session Lentivirus!!! Is it health threatening to lab technician? What s so good about this RNAi library? How to produce
LAB 1, Immunology. Laboratory manual Immunology and Infection Biology Biomedicine course Autumn 2007
 LAB 1, Immunology Laboratory manual Immunology and Infection Biology Biomedicine course Autumn 2007 QUANTIFICATION OF CYTOTOXIC T LYMPHOCYTE ACTIVITY AND DETECTION OF FAS/TRAILR INDUCED APOPTOSIS RESPONSIBLE:
LAB 1, Immunology Laboratory manual Immunology and Infection Biology Biomedicine course Autumn 2007 QUANTIFICATION OF CYTOTOXIC T LYMPHOCYTE ACTIVITY AND DETECTION OF FAS/TRAILR INDUCED APOPTOSIS RESPONSIBLE:
Memory CD4 T Cells Enhance Primary CD8 T-Cell Responses
 INFECTION AND IMMUNITY, July 2007, p. 3556 3560 Vol. 75, No. 7 0019-9567/07/$08.00 0 doi:10.1128/iai.00086-07 Copyright 2007, American Society for Microbiology. All Rights Reserved. Memory CD4 T Cells
INFECTION AND IMMUNITY, July 2007, p. 3556 3560 Vol. 75, No. 7 0019-9567/07/$08.00 0 doi:10.1128/iai.00086-07 Copyright 2007, American Society for Microbiology. All Rights Reserved. Memory CD4 T Cells
Figure S1. Generation of inducible PTEN deficient mice and the BMMCs (A) B6.129 Pten loxp/loxp mice were mated with B6.
 Figure S1. Generation of inducible PTEN deficient mice and the BMMCs (A) B6.129 Pten loxp/loxp mice were mated with B6.129-Gt(ROSA)26Sor tm1(cre/ert2)tyj /J mice. To induce deletion of the Pten locus,
Figure S1. Generation of inducible PTEN deficient mice and the BMMCs (A) B6.129 Pten loxp/loxp mice were mated with B6.129-Gt(ROSA)26Sor tm1(cre/ert2)tyj /J mice. To induce deletion of the Pten locus,
MATERIALS AND METHODS. Neutralizing antibodies specific to mouse Dll1, Dll4, J1 and J2 were prepared as described. 1,2 All
 MATERIALS AND METHODS Antibodies (Abs), flow cytometry analysis and cell lines Neutralizing antibodies specific to mouse Dll1, Dll4, J1 and J2 were prepared as described. 1,2 All other antibodies used
MATERIALS AND METHODS Antibodies (Abs), flow cytometry analysis and cell lines Neutralizing antibodies specific to mouse Dll1, Dll4, J1 and J2 were prepared as described. 1,2 All other antibodies used
Therapeutic PD L1 and LAG 3 blockade rapidly clears established blood stage Plasmodium infection
 Supplementary Information Therapeutic PD L1 and LAG 3 blockade rapidly clears established blood stage Plasmodium infection Noah S. Butler, Jacqueline Moebius, Lecia L. Pewe, Boubacar Traore, Ogobara K.
Supplementary Information Therapeutic PD L1 and LAG 3 blockade rapidly clears established blood stage Plasmodium infection Noah S. Butler, Jacqueline Moebius, Lecia L. Pewe, Boubacar Traore, Ogobara K.
Chapter 22: The Lymphatic System and Immunity
 Bio40C schedule Lecture Immune system Lab Quiz 2 this week; bring a scantron! Study guide on my website (see lab assignments) Extra credit Critical thinking questions at end of chapters 5 pts/chapter Due
Bio40C schedule Lecture Immune system Lab Quiz 2 this week; bring a scantron! Study guide on my website (see lab assignments) Extra credit Critical thinking questions at end of chapters 5 pts/chapter Due
SIV p27 ANTIGEN CAPTURE ASSAY
 SIV p27 ANTIGEN CAPTURE ASSAY Enzyme Immunoassay for the detection of Simian Immunodeficiency Virus (SIV) p27 in tissue culture media Catalog #5436 and #5450 Version 6; 12/2012 ABL PRODUCTS AND SERVICES
SIV p27 ANTIGEN CAPTURE ASSAY Enzyme Immunoassay for the detection of Simian Immunodeficiency Virus (SIV) p27 in tissue culture media Catalog #5436 and #5450 Version 6; 12/2012 ABL PRODUCTS AND SERVICES
Bead Based Assays for Cytokine Detection
 Bead Based Assays for Cytokine Detection September 27, 2014 6 th EFIS-EJI South East European Immunology School SEEIS 2014 Timisoara, Romania The Cells of the Immune System The Immune Reaction (Th2) (Th1)
Bead Based Assays for Cytokine Detection September 27, 2014 6 th EFIS-EJI South East European Immunology School SEEIS 2014 Timisoara, Romania The Cells of the Immune System The Immune Reaction (Th2) (Th1)
Blocking Interhost Transmission of Influenza Virus by Vaccination in the Guinea Pig Model
 JOURNAL OF VIROLOGY, Apr. 2009, p. 2803 2818 Vol. 83, No. 7 0022-538X/09/$08.00 0 doi:10.1128/jvi.02424-08 Copyright 2009, American Society for Microbiology. All Rights Reserved. Blocking Interhost Transmission
JOURNAL OF VIROLOGY, Apr. 2009, p. 2803 2818 Vol. 83, No. 7 0022-538X/09/$08.00 0 doi:10.1128/jvi.02424-08 Copyright 2009, American Society for Microbiology. All Rights Reserved. Blocking Interhost Transmission
Electron micrograph of phosphotungstanic acid-stained exosomes derived from murine
 1 SUPPLEMENTARY INFORMATION SUPPLEMENTARY FIGURES Supplementary Figure 1. Physical properties of murine DC-derived exosomes. a, Electron micrograph of phosphotungstanic acid-stained exosomes derived from
1 SUPPLEMENTARY INFORMATION SUPPLEMENTARY FIGURES Supplementary Figure 1. Physical properties of murine DC-derived exosomes. a, Electron micrograph of phosphotungstanic acid-stained exosomes derived from
Figure S1. Schematic presentation of genomic replication of idsiv after transfection and infection. After transfection of idsiv plasmid DNA into 293T
 Figure S1. Schematic presentation of genomic replication of idsiv after transfection and infection. After transfection of idsiv plasmid DNA into 293T cells, the RNA genomes with all modifications are generated
Figure S1. Schematic presentation of genomic replication of idsiv after transfection and infection. After transfection of idsiv plasmid DNA into 293T cells, the RNA genomes with all modifications are generated
Resolution of a chronic viral infection after interleukin-10 receptor blockade
 ARTICLE Resolution of a chronic viral infection after interleukin-10 receptor blockade Mette Ejrnaes, 1 Christophe M. Filippi, 1 Marianne M. Martinic, 1 Eleanor M. Ling, 1 Lisa M. Togher, 1 Shane Crotty,
ARTICLE Resolution of a chronic viral infection after interleukin-10 receptor blockade Mette Ejrnaes, 1 Christophe M. Filippi, 1 Marianne M. Martinic, 1 Eleanor M. Ling, 1 Lisa M. Togher, 1 Shane Crotty,
Sarah A. Kopecky and Douglas S. Lyles*
 JOURNAL OF VIROLOGY, Apr. 2003, p. 4658 4669 Vol. 77, No. 8 0022-538X/03/$08.00 0 DOI: 10.1128/JVI.77.8.4658 4669.2003 Copyright 2003, American Society for Microbiology. All Rights Reserved. Contrasting
JOURNAL OF VIROLOGY, Apr. 2003, p. 4658 4669 Vol. 77, No. 8 0022-538X/03/$08.00 0 DOI: 10.1128/JVI.77.8.4658 4669.2003 Copyright 2003, American Society for Microbiology. All Rights Reserved. Contrasting
Optimizing Intracellular Flow Cytometry
 Optimizing Intracellular Flow Cytometry Detection of Cytokines, Transcription Factors, and Phosphoprotein by Flow Cytometry Presented by Erika O Donnell, PhD, BD Biosciences 23-14876-00 Outline Basic principles
Optimizing Intracellular Flow Cytometry Detection of Cytokines, Transcription Factors, and Phosphoprotein by Flow Cytometry Presented by Erika O Donnell, PhD, BD Biosciences 23-14876-00 Outline Basic principles
Oncolytic Immunotherapy: A Local and Systemic Antitumor Approach
 Oncolytic Immunotherapy: A Local and Systemic Antitumor Approach Oncolytic immunotherapy Oncolytic immunotherapy the use of a genetically modified virus to attack tumors and induce a systemic immune response
Oncolytic Immunotherapy: A Local and Systemic Antitumor Approach Oncolytic immunotherapy Oncolytic immunotherapy the use of a genetically modified virus to attack tumors and induce a systemic immune response
Cover Page. The handle holds various files of this Leiden University dissertation.
 Cover Page The handle http://hdl.handle.net/1887/23854 holds various files of this Leiden University dissertation. Author: Marel, Sander van der Title: Gene and cell therapy based treatment strategies
Cover Page The handle http://hdl.handle.net/1887/23854 holds various files of this Leiden University dissertation. Author: Marel, Sander van der Title: Gene and cell therapy based treatment strategies
Plasmid-Driven Formation of Influenza Virus-Like Particles
 JOURNAL OF VIROLOGY, Jan. 2000, p. 547 551 Vol. 74, No. 1 0022-538X/00/$04.00 0 Copyright 2000, American Society for Microbiology. All Rights Reserved. Plasmid-Driven Formation of Influenza Virus-Like
JOURNAL OF VIROLOGY, Jan. 2000, p. 547 551 Vol. 74, No. 1 0022-538X/00/$04.00 0 Copyright 2000, American Society for Microbiology. All Rights Reserved. Plasmid-Driven Formation of Influenza Virus-Like
COMPONENT NAME COMPONENT # QUANTITY STORAGE SHELF LIFE FORMAT. Store at 2-8 C. Do not freeze. Store at 2-8 C. Do not freeze.
 This document is available at www.stemcell.com/pis Catalog #18765 EasySep Mouse CD4+CD62L+ T Cell Isolation Kit For processing 1x 10^9 cells Description Isolate highly purified naïve CD4+ T cells (CD4+CD62L+)
This document is available at www.stemcell.com/pis Catalog #18765 EasySep Mouse CD4+CD62L+ T Cell Isolation Kit For processing 1x 10^9 cells Description Isolate highly purified naïve CD4+ T cells (CD4+CD62L+)
HCC1937 is the HCC1937-pcDNA3 cell line, which was derived from a breast cancer with a mutation
 SUPPLEMENTARY INFORMATION Materials and Methods Human cell lines and culture conditions HCC1937 is the HCC1937-pcDNA3 cell line, which was derived from a breast cancer with a mutation in exon 20 of BRCA1
SUPPLEMENTARY INFORMATION Materials and Methods Human cell lines and culture conditions HCC1937 is the HCC1937-pcDNA3 cell line, which was derived from a breast cancer with a mutation in exon 20 of BRCA1
Third line of Defense. Topic 8 Specific Immunity (adaptive) (18) 3 rd Line = Prophylaxis via Immunization!
 Topic 8 Specific Immunity (adaptive) (18) Topics - 3 rd Line of Defense - B cells - T cells - Specific Immunities 1 3 rd Line = Prophylaxis via Immunization! (a) A painting of Edward Jenner depicts a cow
Topic 8 Specific Immunity (adaptive) (18) Topics - 3 rd Line of Defense - B cells - T cells - Specific Immunities 1 3 rd Line = Prophylaxis via Immunization! (a) A painting of Edward Jenner depicts a cow
Influenza A H1N1 HA ELISA Pair Set
 Influenza A H1N1 HA ELISA Pair Set for H1N1 ( A/Puerto Rico/8/1934 ) HA Catalog Number : SEK11684 To achieve the best assay results, this manual must be read carefully before using this product and the
Influenza A H1N1 HA ELISA Pair Set for H1N1 ( A/Puerto Rico/8/1934 ) HA Catalog Number : SEK11684 To achieve the best assay results, this manual must be read carefully before using this product and the
PBMC from each patient were suspended in AIM V medium (Invitrogen) with 5% human
 Anti-CD19-CAR transduced T-cell preparation PBMC from each patient were suspended in AIM V medium (Invitrogen) with 5% human AB serum (Gemini) and 300 international units/ml IL-2 (Novartis). T cell proliferation
Anti-CD19-CAR transduced T-cell preparation PBMC from each patient were suspended in AIM V medium (Invitrogen) with 5% human AB serum (Gemini) and 300 international units/ml IL-2 (Novartis). T cell proliferation
Helminth worm, Schistosomiasis Trypanosomes, sleeping sickness Pneumocystis carinii. Ringworm fungus HIV Influenza
 Helminth worm, Schistosomiasis Trypanosomes, sleeping sickness Pneumocystis carinii Ringworm fungus HIV Influenza Candida Staph aureus Mycobacterium tuberculosis Listeria Salmonella Streptococcus Levels
Helminth worm, Schistosomiasis Trypanosomes, sleeping sickness Pneumocystis carinii Ringworm fungus HIV Influenza Candida Staph aureus Mycobacterium tuberculosis Listeria Salmonella Streptococcus Levels
Department of Surgery, Duke University, Durham, North Carolina; and 7 Southern Research Institute, Frederick, Maryland
 MAJOR ARTICLE Highly Attenuated Rabies Virus Based Vaccine Vectors Expressing Simian-Human Immunodeficiency Virus 89.6P Env and Simian Immunodeficiency Virus mac239 Gag Are Safe in Rhesus Macaques and
MAJOR ARTICLE Highly Attenuated Rabies Virus Based Vaccine Vectors Expressing Simian-Human Immunodeficiency Virus 89.6P Env and Simian Immunodeficiency Virus mac239 Gag Are Safe in Rhesus Macaques and
General information. Cell mediated immunity. 455 LSA, Tuesday 11 to noon. Anytime after class.
 General information Cell mediated immunity 455 LSA, Tuesday 11 to noon Anytime after class T-cell precursors Thymus Naive T-cells (CD8 or CD4) email: lcoscoy@berkeley.edu edu Use MCB150 as subject line
General information Cell mediated immunity 455 LSA, Tuesday 11 to noon Anytime after class T-cell precursors Thymus Naive T-cells (CD8 or CD4) email: lcoscoy@berkeley.edu edu Use MCB150 as subject line
Supplementary Table; Supplementary Figures and legends S1-S21; Supplementary Materials and Methods
 Silva et al. PTEN posttranslational inactivation and hyperactivation of the PI3K/Akt pathway sustain primary T cell leukemia viability Supplementary Table; Supplementary Figures and legends S1-S21; Supplementary
Silva et al. PTEN posttranslational inactivation and hyperactivation of the PI3K/Akt pathway sustain primary T cell leukemia viability Supplementary Table; Supplementary Figures and legends S1-S21; Supplementary
Received 30 January 2002/Accepted 18 December 2002
 JOURNAL OF VIROLOGY, Mar. 2003, p. 3634 3646 Vol. 77, No. 6 0022-538X/03/$08.00 0 DOI: 10.1128/JVI.77.6.3634 3646.2003 Copyright 2003, American Society for Microbiology. All Rights Reserved. Rational Site-Directed
JOURNAL OF VIROLOGY, Mar. 2003, p. 3634 3646 Vol. 77, No. 6 0022-538X/03/$08.00 0 DOI: 10.1128/JVI.77.6.3634 3646.2003 Copyright 2003, American Society for Microbiology. All Rights Reserved. Rational Site-Directed
and follicular helper T cells is Egr2-dependent. (a) Diagrammatic representation of the
 Supplementary Figure 1. LAG3 + Treg-mediated regulation of germinal center B cells and follicular helper T cells is Egr2-dependent. (a) Diagrammatic representation of the experimental protocol for the
Supplementary Figure 1. LAG3 + Treg-mediated regulation of germinal center B cells and follicular helper T cells is Egr2-dependent. (a) Diagrammatic representation of the experimental protocol for the
ADCC Assay Protocol Vikram Srivastava 1, Zheng Yang 1, Ivan Fan Ngai Hung 2, Jianqing Xu 3, Bojian Zheng 3 and Mei- Yun Zhang 3*
 ADCC Assay Protocol Vikram Srivastava 1, Zheng Yang 1, Ivan Fan Ngai Hung 2, Jianqing Xu 3, Bojian Zheng 3 and Mei- Yun Zhang 3* 1 Department of Microbiology, Li Ka Shing Faculty of Medicine, University
ADCC Assay Protocol Vikram Srivastava 1, Zheng Yang 1, Ivan Fan Ngai Hung 2, Jianqing Xu 3, Bojian Zheng 3 and Mei- Yun Zhang 3* 1 Department of Microbiology, Li Ka Shing Faculty of Medicine, University
Essential Medium, containing 10% fetal bovine serum, 100 U/ml penicillin and 100 µg/ml streptomycin. Huvec were cultured in
 Supplemental data Methods Cell culture media formulations A-431 and U-87 MG cells were maintained in Dulbecco s Modified Eagle s Medium. FaDu cells were cultured in Eagle's Minimum Essential Medium, containing
Supplemental data Methods Cell culture media formulations A-431 and U-87 MG cells were maintained in Dulbecco s Modified Eagle s Medium. FaDu cells were cultured in Eagle's Minimum Essential Medium, containing
Rabies virus-like particles expressed in HEK293 cells
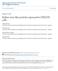 Engineering Conferences International ECI Digital Archives Vaccine Technology IV Proceedings Spring 5-21-2012 Rabies virus-like particles expressed in HEK293 cells Diego Fontana Cell Culture Laboratory
Engineering Conferences International ECI Digital Archives Vaccine Technology IV Proceedings Spring 5-21-2012 Rabies virus-like particles expressed in HEK293 cells Diego Fontana Cell Culture Laboratory
Micro 204. Cytotoxic T Lymphocytes (CTL) Lewis Lanier
 Micro 204 Cytotoxic T Lymphocytes (CTL) Lewis Lanier Lewis.Lanier@ucsf.edu Lymphocyte-mediated Cytotoxicity CD8 + αβ-tcr + T cells CD4 + αβ-tcr + T cells γδ-tcr + T cells Natural Killer cells CD8 + αβ-tcr
Micro 204 Cytotoxic T Lymphocytes (CTL) Lewis Lanier Lewis.Lanier@ucsf.edu Lymphocyte-mediated Cytotoxicity CD8 + αβ-tcr + T cells CD4 + αβ-tcr + T cells γδ-tcr + T cells Natural Killer cells CD8 + αβ-tcr
Patricia Fitzgerald-Bocarsly
 FLU Patricia Fitzgerald-Bocarsly October 23, 2008 Orthomyxoviruses Orthomyxo virus (ortho = true or correct ) Negative-sense RNA virus (complementary to mrna) Five different genera Influenza A, B, C Thogotovirus
FLU Patricia Fitzgerald-Bocarsly October 23, 2008 Orthomyxoviruses Orthomyxo virus (ortho = true or correct ) Negative-sense RNA virus (complementary to mrna) Five different genera Influenza A, B, C Thogotovirus
Challenges in Development and Validation of an Intracellular Cytokine Staining assay
 Challenges in Development and Validation of an Intracellular Cytokine Staining assay Jenny Hendriks, Crucell Hatching @ EBF, Brussels, June 202 www.crucell.com Vaccines vs Protein therapeutics Protein
Challenges in Development and Validation of an Intracellular Cytokine Staining assay Jenny Hendriks, Crucell Hatching @ EBF, Brussels, June 202 www.crucell.com Vaccines vs Protein therapeutics Protein
COMPONENT NAME COMPONENT # QUANTITY STORAGE SHELF LIFE FORMAT. Store at 2-8 C. Do not freeze. Store at 2-8 C. Do not freeze.
 This document is available at www.stemcell.com/pis Catalog #18765 EasySep Mouse CD4+CD62L+ T Cell Isolation Kit For processing 1x 10^9 cells Description Isolate highly purified naïve CD4+ T cells (CD4+CD62L+)
This document is available at www.stemcell.com/pis Catalog #18765 EasySep Mouse CD4+CD62L+ T Cell Isolation Kit For processing 1x 10^9 cells Description Isolate highly purified naïve CD4+ T cells (CD4+CD62L+)
Chapter 15 Adaptive, Specific Immunity and Immunization
 Chapter 15 Adaptive, Specific Immunity and Immunization Adaptive Immunity: The third line of defense Third line of defense acquired and specific. Dual System of B and T lymphocytes- Immunocompetence Antigen
Chapter 15 Adaptive, Specific Immunity and Immunization Adaptive Immunity: The third line of defense Third line of defense acquired and specific. Dual System of B and T lymphocytes- Immunocompetence Antigen
QuickTiter Lentivirus Titer Kit (Lentivirus-Associated HIV p24)
 Product Manual QuickTiter Lentivirus Titer Kit (Lentivirus-Associated HIV p24) Catalog Number VPK-107 VPK-107-5 96 assays 5 x 96 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction
Product Manual QuickTiter Lentivirus Titer Kit (Lentivirus-Associated HIV p24) Catalog Number VPK-107 VPK-107-5 96 assays 5 x 96 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction
AGAINST VIRAL INFECTIONS. Identify the types of immunity involve in the mechanisms of protection against viral infections.
 LECTURE: 02 Title: THE IMMUNOLOGICAL PROTECTIVE MECHANISMS AGAINST VIRAL INFECTIONS LEARNING OBJECTIVES: The student should be able to: Identify the types of immunity involve in the mechanisms of protection
LECTURE: 02 Title: THE IMMUNOLOGICAL PROTECTIVE MECHANISMS AGAINST VIRAL INFECTIONS LEARNING OBJECTIVES: The student should be able to: Identify the types of immunity involve in the mechanisms of protection
Overview: Chapter 19 Viruses: A Borrowed Life
 Overview: Chapter 19 Viruses: A Borrowed Life Viruses called bacteriophages can infect and set in motion a genetic takeover of bacteria, such as Escherichia coli Viruses lead a kind of borrowed life between
Overview: Chapter 19 Viruses: A Borrowed Life Viruses called bacteriophages can infect and set in motion a genetic takeover of bacteria, such as Escherichia coli Viruses lead a kind of borrowed life between
For questions 1-5, match the following with their correct descriptions. (24-39) A. Class I B. Class II C. Class III D. TH1 E. TH2
 Questions Made by SI ATTENDEES!! :) Page 1 of 6 Student-Made Practice Exam Activity All questions, answers, and slide numbers are based off of Monday s SI activity, where students/attendees created possible
Questions Made by SI ATTENDEES!! :) Page 1 of 6 Student-Made Practice Exam Activity All questions, answers, and slide numbers are based off of Monday s SI activity, where students/attendees created possible
Madhav V. Dhodapkar, Joseph Krasovsky, Ralph M. Steinman, and Nina Bhardwaj
 Mature dendritic cells boost functionally superior CD8 + T-cell in humans without foreign helper epitopes Rapid PUBLICATION Madhav V. Dhodapkar, Joseph Krasovsky, Ralph M. Steinman, and Nina Bhardwaj Laboratory
Mature dendritic cells boost functionally superior CD8 + T-cell in humans without foreign helper epitopes Rapid PUBLICATION Madhav V. Dhodapkar, Joseph Krasovsky, Ralph M. Steinman, and Nina Bhardwaj Laboratory
xcelligence Real-Time Cell Analyzers
 xcelligence Real-Time Cell Analyzers Application Note No. 9 A New Way to Monitor Virus-Mediated Cytopathogenicity Introduction One of the most important procedures in virology is the measurement of viral
xcelligence Real-Time Cell Analyzers Application Note No. 9 A New Way to Monitor Virus-Mediated Cytopathogenicity Introduction One of the most important procedures in virology is the measurement of viral
Hepatitis B Antiviral Drug Development Multi-Marker Screening Assay
 Hepatitis B Antiviral Drug Development Multi-Marker Screening Assay Background ImQuest BioSciences has developed and qualified a single-plate method to expedite the screening of antiviral agents against
Hepatitis B Antiviral Drug Development Multi-Marker Screening Assay Background ImQuest BioSciences has developed and qualified a single-plate method to expedite the screening of antiviral agents against
PERSISTENT INFECTIONS WITH HUMAN PARAINFLUENZAVIRUS TYPE 3 IN TWO CELL LINES
 71 PERSISTENT INFECTIONS WITH HUMAN PARAINFLUENZAVIRUS TYPE 3 IN TWO CELL LINES Harold G. Jensen, Alan J. Parkinson, and L. Vernon Scott* Department of Microbiology & Immunology, University of Oklahoma
71 PERSISTENT INFECTIONS WITH HUMAN PARAINFLUENZAVIRUS TYPE 3 IN TWO CELL LINES Harold G. Jensen, Alan J. Parkinson, and L. Vernon Scott* Department of Microbiology & Immunology, University of Oklahoma
Chapter 19: The Genetics of Viruses and Bacteria
 Chapter 19: The Genetics of Viruses and Bacteria What is Microbiology? Microbiology is the science that studies microorganisms = living things that are too small to be seen with the naked eye Microorganisms
Chapter 19: The Genetics of Viruses and Bacteria What is Microbiology? Microbiology is the science that studies microorganisms = living things that are too small to be seen with the naked eye Microorganisms
Received 24 March 2007/Accepted 4 May 2007
 JOURNAL OF VIROLOGY, July 2007, p. 7766 7775 Vol. 81, No. 14 0022-538X/07/$08.00 0 doi:10.1128/jvi.00633-07 Copyright 2007, American Society for Microbiology. All Rights Reserved. DNA Immunization Using
JOURNAL OF VIROLOGY, July 2007, p. 7766 7775 Vol. 81, No. 14 0022-538X/07/$08.00 0 doi:10.1128/jvi.00633-07 Copyright 2007, American Society for Microbiology. All Rights Reserved. DNA Immunization Using
PREPARED FOR: U.S. Army Medical Research and Materiel Command Fort Detrick, Maryland Approved for public release; distribution unlimited
 AD (Leave blank) Award Number: W81XWH-07-1-0345 TITLE: Second-Generation Therapeutic DNA Lymphoma Vaccines PRINCIPAL INVESTIGATOR: Larry W. Kwak, M.D., Ph.D. CONTRACTING ORGANIZATION: University of Texas
AD (Leave blank) Award Number: W81XWH-07-1-0345 TITLE: Second-Generation Therapeutic DNA Lymphoma Vaccines PRINCIPAL INVESTIGATOR: Larry W. Kwak, M.D., Ph.D. CONTRACTING ORGANIZATION: University of Texas
Supplemental Materials for. Effects of sphingosine-1-phosphate receptor 1 phosphorylation in response to. FTY720 during neuroinflammation
 Supplemental Materials for Effects of sphingosine-1-phosphate receptor 1 phosphorylation in response to FTY7 during neuroinflammation This file includes: Supplemental Table 1. EAE clinical parameters of
Supplemental Materials for Effects of sphingosine-1-phosphate receptor 1 phosphorylation in response to FTY7 during neuroinflammation This file includes: Supplemental Table 1. EAE clinical parameters of
