Novitas Medicare Policy Primer
|
|
|
- Hubert Clinton Simpson
- 6 years ago
- Views:
Transcription
1 Novitas Medicare Policy Primer Medicare Jurisdiction (JL and JH) AR, CO, LA, MS, NM, OK, TX, DC, DE, MD, NJ, & PA Counties of Arlington and Fairfax in Virginia and the city of Alexandria in Virginia Application of Bioengineered Skin Substitutes to Lower Extremity Chronic Non-Healing Wounds #L35041 Indications Therefore, all products with FDA clearance/approval or designated 361 HCT/P exemption used in accordance with that product s individualized application guidelines will be equally considered for the purpose of this LCD and may be considered reasonable and necessary. Presence of neuropathic diabetic foot ulcer(s) having failed to respond to documented conservative wound-care measures of greater than four weeks, during which the patient is compliant with recommendations, and without evidence of underlying osteomyelitis or nidus of infection. Presence of a venous stasis ulcer for at least 3 months but unresponsive to appropriate wound care for at least 30 days with documented compliance. Presence of a full-thickness skin loss ulcer that is the result of abscess, injury, or trauma that has failed to respond to appropriate control of infection, foreign body, tumor resection, or other disease process for a period of 4 weeks or longer. Response from Apligraf reconsideration letter from Novitas JH June 2015 Novitas only requires 1 month of compliant wound therapy prior to placement on a venous stasis (VSU), though it is suggested that the wounds be present for at least 3 months; this is not a requirement for placement. It is standard therapy for all ulcers for 30 days and would not need to meet medical necessity requirements. Medicare covers application of skin substitutes to Ulcers or Wounds with Failed Response that are: Partial- or full-thickness ulcers, not involving tendon, muscle, joint capsule, or exhibiting exposed bone or sinus tracts, with a clean granular base; Skin deficit at least 1.0 cm² in size; Clean and free of necrotic debris or exudate; Have adequate circulation/oxygenation to support tissue growth/wound healing as evidenced by physical examination (e.g., Ankle-Brachial Index (ABI) of no less than 0.60, toe pressure > 30mm Hg);
2 For diabetic foot ulcers, the patient s medical record reflects a diagnosis of Type 1 or Type 2 Diabetes and also reflects medical management for this condition. Limitations Skin substitute grafts will be allowed for the episode of wound care in compliance with FDA guidelines for the specific product (see utilization guidelines) not to exceed 10 applications or treatments. In situations where more than one specific product is used, it is expected that the number of applications or treatments will still not exceed 10. Simultaneous use of more than one product for the episode of wound is not covered. Product change within the episode of wound is allowed, not to exceed the 10 application limit per wound per 12 week period of care. Treatment of any chronic skin wound will typically last no more than twelve (12) weeks. Repeat or alternative applications of skin substitute grafts are not considered medically reasonable and necessary when a previous full course of applications was unsuccessful. Unsuccessful treatment is defined as increase in size or depth of an ulcer or no change in baseline size or depth and no sign of improvement or indication that improvement is likely (such as granulation, epithelialization, or progress towards closing) for a period of 4 weeks past start of therapy. Retreatment of healed ulcers, those showing greater than 75% size reduction and smaller than.5 sq.cm, is not considered medically reasonable and necessary. Skin substitute grafts are contraindicated and are not considered reasonable and necessary in patients with inadequate control of underlying conditions or exacerbating factors (e.g., uncontrolled diabetes, active infection, and active Charcot arthropathy of the ulcer extremity, vasculitis, or continued tobacco smoking without physician attempt to effect smoking cessation). Repeat use of surgical preparation services (CPT codes 15002, 15003, 15004, and 15005) in conjunction with skin substitute application codes will be considered not reasonable and necessary. It is expected that each wound will require the use of appropriate wound preparation code at least once at initiation of care prior to placement of the skin substitute graft. Re-treatment within one (1) year of any given course of skin substitute treatment for a venous stasis ulcer or (diabetic) neuropathic foot ulcer is considered treatment failure and does not meet reasonable and necessary criteria for re-treatment of that ulcer with a skin substitute procedure.
3 Documentation Every page of the record must be legible and include appropriate patient identification information (e.g., complete name, dates of service(s)). The documentation must include the legible signature of the physician or non-physician practitioner responsible for and providing the care to the patient. The submitted medical record must support the use of the selected ICD-10-CM code(s). The submitted CPT/HCPCS code must describe the service performed. Medical record documentation must support the medical necessity of the services as directed in this policy. The documentation must support that the service was performed and must be included in the patient s medical record. This information is normally found in the history and physical, office/progress notes, hospital notes, and/or procedure report. The medical record must clearly show that the criteria listed under the Indications and Limitations of Coverage and/or Medical Necessity sections have been met, as well as, the appropriate diagnosis and response to treatment. A description of the wound(s) must be documented at baseline (prior to beginning conservative treatment) relative to size, location, stage, duration, and presence of infection, in addition to type of treatment given and response. Wound description must also be documented pre and post treatment with the skin substitute graft being used. If obvious signs of worsening or lack of treatment response is noted, continuing treatment with the skin substitute would not be considered medically reasonable and necessary without documentation of a reasonable rationale for doing so. Documentation of smoking history, and that the patient has received counseling on the effects of smoking on surgical outcomes and treatment for smoking cessation (if applicable) as well as outcome of counselling must be in the medical record. The amount of utilized and wasted skin substitute must be clearly documented in the procedure note. Coding HCPCS Codes: Q4101: Apligraf, per square centimeter Q4106: Dermagraft, per square centimeter Q4172: PuraPly, PuraPly Antimicrobial per square centimeter Q4159: Affinity, per square centimeter Q4160: NuShield, per square centimeter
4 JW Modifier: Effective January 1, 2017 in Physician Office Setting (Place of service 11): Claims for discarded drug or biological amount not administered to any patient shall be submitted using the JW modifier. This modifier, billed on a separate line, will provide payment for the amount of discarded drug or biological. Providers must document the discarded drugs or biologicals in patient's medical record. CPT Codes: Application Codes for Leg, Arm or Trunk: 15271: Application of skin substitute graft to trunk, arms, legs, total wound surface area up to 100 sq cm; first 25 sq cm or less wound surface area 15272: Application of skin substitute graft to trunk, arms, legs, total wound surface area up to 100 sq cm; each additional 25 sq cm wound surface area, or part thereof (List separately in addition to code for primary procedure) 15273: Application of skin substitute graft to trunk, arms, legs, total wound surface area greater than or equal to 100 sq cm; first 100 sq cm wound surface area 15274: Application of skin substitute graft to trunk, arms, legs, total wound surface area greater than or equal to 100 sq cm; each additional 100 sq cm wound surface area, or part thereof (List separately in addition to code for primary procedure) Application Codes for Foot, Face, Scalp, etc.: 15275: Application of skin substitute graft to face, scalp, eyelids, mouth, neck, ears, orbits, genitalia, hands, feet, and/or multiple digits, total wound surface area up to 100 sq cm; first 25 sq cm or less wound surface area 15276: Application of skin substitute graft to face, scalp, eyelids, mouth, neck, ears, orbits, genitalia, hands, feet, and/or multiple digits, total wound surface area up to 100 sq cm; each additional 25 sq cm wound surface area, or part thereof (List separately in addition to code for primary procedure) 15277: Application of skin substitute graft to face, scalp, eyelids, mouth, neck, ears, orbits, genitalia, hands, feet, and/or multiple digits, total wound surface area greater than or equal to 100 sq cm; first 100 sq cm wound surface area 15278: Application of skin substitute graft to face, scalp, eyelids, mouth, neck, ears, orbits, genitalia, hands, feet, and/or multiple digits, total wound surface area greater than or equal to 100 sq cm; each additional 100 sq cm wound surface area, or part thereof (List separately in addition to code for primary procedure)
5 Novitas Medicare Sample Model Documentation Medicare Jurisdiction (JL and JH) AR, CO, LA, MS, NM, OK, TX, DC, DE, MD, NJ, & PA Counties of Arlington and Fairfax in Virginia and the city of Alexandria in Virginia Pre-Treatment 1. Duration of ulcer: weeks 2. Exact location of ulcer 3. Describe adequate treatment of the underlying disease process contributing to the ulcer. 4. Diagnosis of patient Treatment 5. Document measurement of ulcer (width and length or circumference and depth) immediately prior to application of the skin substitute sq cm 6. Document whether this is an initial application of skin substitute or a reapplication. 7. For skin substitute reapplications, document that applications have been successful (e.g. decrease in size or depth, increase in granulation tissue). 8. Document the wound dressing changes and the standard conservative measures accompanying the wound treatment with the skin substitute. 9. Document how the wound site was prepared, and how the skin substitute was fixated on the wound. 10. Product Wastage Documentation Requirements: Date and time Location of ulcer Approximate amount of product unit used Approximate amount of product unit discarded Reason for the wastage Manufacture s serial/lot/batch number Modifiers JW: Skin substitute not applied to wound, wastage
CGS Medicare Policy Primer
 CGS Medicare Policy Primer Medicare Jurisdiction (J15) OH & KY Wound Application of Cellular and/or Tissue Based Products (CTPs) Lower Extremities #L36690 Indications Application of a CTP graft for lower
CGS Medicare Policy Primer Medicare Jurisdiction (J15) OH & KY Wound Application of Cellular and/or Tissue Based Products (CTPs) Lower Extremities #L36690 Indications Application of a CTP graft for lower
Palmetto Medicare Policy Primer
 Palmetto Medicare Policy Primer Medicare Jurisdiction (JM) NC, SC, WV & VA Application of Skin Substitutes LCD #L36466 Indications Presence of neuropathic diabetic foot ulcer(s) having failed to respond
Palmetto Medicare Policy Primer Medicare Jurisdiction (JM) NC, SC, WV & VA Application of Skin Substitutes LCD #L36466 Indications Presence of neuropathic diabetic foot ulcer(s) having failed to respond
First Coast Service Options (FCSO) Medicare Policy Primer
 First Coast Service Options (FCSO) Medicare Policy Primer Medicare Jurisdiction (JN) Florida, Puerto Rico and U.S. Virgin Islands Application of Skin Substitute Grafts for the treatment of DFU and VLU
First Coast Service Options (FCSO) Medicare Policy Primer Medicare Jurisdiction (JN) Florida, Puerto Rico and U.S. Virgin Islands Application of Skin Substitute Grafts for the treatment of DFU and VLU
WPS Medicare Policy Primer
 WPS Medicare Policy Primer Medicare Jurisdiction (J5 & J8) NE, KS, IA, MO, IN, & MI Retired skin substitute LCD 3/2016 Indications Apligraf Non-infected partial and full-thickness skin ulcers due to VSU
WPS Medicare Policy Primer Medicare Jurisdiction (J5 & J8) NE, KS, IA, MO, IN, & MI Retired skin substitute LCD 3/2016 Indications Apligraf Non-infected partial and full-thickness skin ulcers due to VSU
Summary of Package Insert 1 for PuraPly Wound Matrix
 Summary of Package Insert 1 for PuraPly Wound Matrix For NGS Indications Indicated for the management of wounds including: Partial and full-thickness wounds Venous ulcers Diabetic ulcers Drainage wounds
Summary of Package Insert 1 for PuraPly Wound Matrix For NGS Indications Indicated for the management of wounds including: Partial and full-thickness wounds Venous ulcers Diabetic ulcers Drainage wounds
Summary of Package Insert 1 for PuraPly Antimicrobial Wound Matrix
 Summary of Package Insert 1 for PuraPly Antimicrobial Wound Matrix For States with Non-Published Policies-Novitas Indications Indicated for the management of wounds as an effective barrier to resist microbial
Summary of Package Insert 1 for PuraPly Antimicrobial Wound Matrix For States with Non-Published Policies-Novitas Indications Indicated for the management of wounds as an effective barrier to resist microbial
Cahaba Medicare Policy Primer 1,2 for Apligraf
 Cahaba Medicare Policy Primer 1,2 for Apligraf MAC A: AL, GA & TN MAC B: AL, GA, & TN LCD# 31428 Indications Applied to partial- or full-thickness ulcers of the lower extremities (see individual product
Cahaba Medicare Policy Primer 1,2 for Apligraf MAC A: AL, GA & TN MAC B: AL, GA, & TN LCD# 31428 Indications Applied to partial- or full-thickness ulcers of the lower extremities (see individual product
Coding for Wound Care
 Coding for Wound Care ****IMPORTANT*** Disclaimer ***Information provided is to the best of our knowledge and as current as possible. ***Please verify all policy and reimbursement information with your
Coding for Wound Care ****IMPORTANT*** Disclaimer ***Information provided is to the best of our knowledge and as current as possible. ***Please verify all policy and reimbursement information with your
Wound & Burn. Reimbursement & Coding Guide
 Wound & Burn Reimbursement & Coding Guide Wound & Burn Reimbursement and Coding Guide MicroMatrix and Cytal devices facilitate the remodeling of functional, site-appropriate tissue. Comprised of ACell
Wound & Burn Reimbursement & Coding Guide Wound & Burn Reimbursement and Coding Guide MicroMatrix and Cytal devices facilitate the remodeling of functional, site-appropriate tissue. Comprised of ACell
Surgical Preparation Codes for Skin Replacement Surgery** Hospital Outpatient/Ambulatory Surgical Center Setting
 2018 National Medicare Reimbursement Rate Summary* for Integra Dermal Regeneration Template, & Office Settings Integra LifeSciences Corporation compiles this summary of Medicare payment rates to provide
2018 National Medicare Reimbursement Rate Summary* for Integra Dermal Regeneration Template, & Office Settings Integra LifeSciences Corporation compiles this summary of Medicare payment rates to provide
MP.083.MH Skin Substitutes- Human Skin Equivalents
 MedStar Health, Inc. POLICY AND PROCEDURE MANUAL MP.083.MH Skin Substitutes- Human Skin Equivalents This policy applies to the following lines of business: MedStar Employee (Select) MedStar MA DSNP CSNP
MedStar Health, Inc. POLICY AND PROCEDURE MANUAL MP.083.MH Skin Substitutes- Human Skin Equivalents This policy applies to the following lines of business: MedStar Employee (Select) MedStar MA DSNP CSNP
Clinical Policy: EpiFix Wound Treatment
 Clinical Policy: Reference Number: PA.CP.MP.140 Effective Date: 03/18 Last Review Date: 04/18 Coding Implications Revision Log Description EpiFix (MiMedx Group) is dehydrated human amniotic tissue that
Clinical Policy: Reference Number: PA.CP.MP.140 Effective Date: 03/18 Last Review Date: 04/18 Coding Implications Revision Log Description EpiFix (MiMedx Group) is dehydrated human amniotic tissue that
DermACELL AWM Comprehensive Reimbursement Resource Guide. Prepared by Musculoskeletal Clinical Regulatory Advisers, LLC. Version 01/2018.
 2018 Comprehensive Reimbursement Resource Guide Prepared by Musculoskeletal Clinical Regulatory Advisers, LLC. Version 01/2018. DermACELL AWM Disclaimer: This information is for educational/informational
2018 Comprehensive Reimbursement Resource Guide Prepared by Musculoskeletal Clinical Regulatory Advisers, LLC. Version 01/2018. DermACELL AWM Disclaimer: This information is for educational/informational
MedStar Health, Inc. POLICY AND PROCEDURE MANUAL Policy Number: MP.083.MH Last Review Date: 11/03/2016 Effective Date: 01/01/2017
 MedStar Health, Inc. POLICY AND PROCEDURE MANUAL MP.083.MH Skin Substitutes- Human Skin Equivalents This policy applies to the following lines of business: MedStar Employee (Select) MedStar MA DSNP CSNP
MedStar Health, Inc. POLICY AND PROCEDURE MANUAL MP.083.MH Skin Substitutes- Human Skin Equivalents This policy applies to the following lines of business: MedStar Employee (Select) MedStar MA DSNP CSNP
CLINICAL MEDICAL POLICY
 Policy Name: Policy Number: Responsible Department(s): CLINICAL MEDICAL POLICY Skin Replacement Therapy for Chronic Non healing Wounds in the Outpatient Setting MP-032-MD-DE Medical Management Provider
Policy Name: Policy Number: Responsible Department(s): CLINICAL MEDICAL POLICY Skin Replacement Therapy for Chronic Non healing Wounds in the Outpatient Setting MP-032-MD-DE Medical Management Provider
Coverage Summary. Wound Treatments
 Coverage Summary Wound Treatments Policy Number: W-001 Products: UnitedHealthcare Medicare Advantage Plans Original Approval Date: 02/18/2009 Approved by: UnitedHealthcare Medicare Benefit Interpretation
Coverage Summary Wound Treatments Policy Number: W-001 Products: UnitedHealthcare Medicare Advantage Plans Original Approval Date: 02/18/2009 Approved by: UnitedHealthcare Medicare Benefit Interpretation
CYGNUS REIMBURSEMENT GUIDE
 CYGNUS REIMBURSEMENT GUIDE The CYGNUS amnion patch is an immune-privileged tissue containing the natural regenerative factors responsible for creating a wound healing environment conducive to tissue regeneration.
CYGNUS REIMBURSEMENT GUIDE The CYGNUS amnion patch is an immune-privileged tissue containing the natural regenerative factors responsible for creating a wound healing environment conducive to tissue regeneration.
Skin Deep. Agenda. Burns Wounds Debridement Evaluation and Management Services. Presented by: Mike Strong, SFM The Work Comp Experts.
 Presented by: Mike Strong, SFM The Work Comp Experts Agenda Wounds Debridement Evaluation and Management Services 2 1 Types of First Degree Second Degree Third Degree Rule of 9 Adults Infants Burn Coding
Presented by: Mike Strong, SFM The Work Comp Experts Agenda Wounds Debridement Evaluation and Management Services 2 1 Types of First Degree Second Degree Third Degree Rule of 9 Adults Infants Burn Coding
Topical Oxygen Wound Therapy (MEDICAID)
 Topical Oxygen Wound Therapy (MEDICAID) Last Review Date: September 8, 2017 Number: MG.MM.DM.15C8v2 Medical Guideline Disclaimer Property of EmblemHealth. All rights reserved. The treating physician or
Topical Oxygen Wound Therapy (MEDICAID) Last Review Date: September 8, 2017 Number: MG.MM.DM.15C8v2 Medical Guideline Disclaimer Property of EmblemHealth. All rights reserved. The treating physician or
Premier Health Plan considers Negative Pressure Wound Therapy (NPWT) in the home setting medically necessary for the following indications:
 Premier Health Plan POLICY AND PROCEDURE MANUAL PA.009.PH Negative Pressure Wound Therapy This policy applies to the following lines of business: Premier Commercial Premier Employee Premier Health Plan
Premier Health Plan POLICY AND PROCEDURE MANUAL PA.009.PH Negative Pressure Wound Therapy This policy applies to the following lines of business: Premier Commercial Premier Employee Premier Health Plan
Negative Pressure Wound Therapy (NPWT)
 Negative Pressure Wound Therapy (NPWT) Policy Number: Original Effective Date: MM.01.005 11/19/1999 Line(s) of Business: Current Effective Date: HMO; PPO; QUEST 01/01/2015 Section: DME Place(s) of Service:
Negative Pressure Wound Therapy (NPWT) Policy Number: Original Effective Date: MM.01.005 11/19/1999 Line(s) of Business: Current Effective Date: HMO; PPO; QUEST 01/01/2015 Section: DME Place(s) of Service:
Negative Pressure Wound Therapy
 Origination: 6/29/04 Revised: 8/24/16 Annual Review: 11/10/16 Purpose: To provide Negative Pressure Wound Therapy (wound care treatment) guidelines for the Medical Department staff to reference when making
Origination: 6/29/04 Revised: 8/24/16 Annual Review: 11/10/16 Purpose: To provide Negative Pressure Wound Therapy (wound care treatment) guidelines for the Medical Department staff to reference when making
National Imaging Associates, Inc. Clinical guidelines CHIROPRACTIC SERVICES. Original Date: Page 1 of FOR CMS (MEDICARE) MEMBERS ONLY
 National Imaging Associates, Inc. Clinical guidelines CHIROPRACTIC SERVICES CPT4 Codes: Please refer to pages 2-10 LCD ID Number: L35424 Novitas: J-H: DC, DE, MD, NJ, PA J-L: AR, CO, LA, MS, NM, OK, TX
National Imaging Associates, Inc. Clinical guidelines CHIROPRACTIC SERVICES CPT4 Codes: Please refer to pages 2-10 LCD ID Number: L35424 Novitas: J-H: DC, DE, MD, NJ, PA J-L: AR, CO, LA, MS, NM, OK, TX
SAMPLE. Relative Values for Dentists Relative values based on survey data from Relative Value Studies, Inc. ICD-10
 www.optumcoding.com Relative Values for Dentists Relative values based on survey data from Relative Value Studies, Inc. 2017 a ICD-10 A full suite of resources including the latest code set, mapping products,
www.optumcoding.com Relative Values for Dentists Relative values based on survey data from Relative Value Studies, Inc. 2017 a ICD-10 A full suite of resources including the latest code set, mapping products,
Assisted Living Resident Assessment (To be used when yes is indicated for skin issues under Section 5 of Assisted Living Resident Assessment)
 Skin Assessment Current open skin areas: Yes No Current pressure ulcer: Yes No A. Stage 1 Ulcers Report based on highest stage of existing ulcers at its worst; do not reverse stage. Number of existing
Skin Assessment Current open skin areas: Yes No Current pressure ulcer: Yes No A. Stage 1 Ulcers Report based on highest stage of existing ulcers at its worst; do not reverse stage. Number of existing
Orthopedic Coding Changes for 2012
 Orthopedic Coding Changes for Lynn M. Anderanin, CPC,CPC-I, COSC Vertebroplasty 22520- Percutaneous vertebroplasty, 1 vertebral body, unilateral or bilateral injection; thoracic 22520- Percutaneous vertebroplasty,
Orthopedic Coding Changes for Lynn M. Anderanin, CPC,CPC-I, COSC Vertebroplasty 22520- Percutaneous vertebroplasty, 1 vertebral body, unilateral or bilateral injection; thoracic 22520- Percutaneous vertebroplasty,
ALLIANCE OF WOUND CARE STAKEHOLDERS. Palmetto GBA Public Meeting Draft LCD on Application of Skin Substitutes (DL36466) October 13, 2015
 ALLIANCE OF WOUND CARE STAKEHOLDERS Palmetto GBA Public Meeting Draft LCD on Application of Skin Substitutes (DL36466) October 13, 2015 1 ALLIANCE OF WOUND CARE Who is the Alliance? STAKEHOLDERS A non-profit
ALLIANCE OF WOUND CARE STAKEHOLDERS Palmetto GBA Public Meeting Draft LCD on Application of Skin Substitutes (DL36466) October 13, 2015 1 ALLIANCE OF WOUND CARE Who is the Alliance? STAKEHOLDERS A non-profit
2017 Coding and Reimbursement Survival Guide
 2017 Coding and Reimbursement Survival Guide Chapter 8: General Surgery Integumentary Procedures: 4 Questions Focus Your Skin Substitute Graft Coding Hint: Graft size doesn t matter. If your surgeon treats
2017 Coding and Reimbursement Survival Guide Chapter 8: General Surgery Integumentary Procedures: 4 Questions Focus Your Skin Substitute Graft Coding Hint: Graft size doesn t matter. If your surgeon treats
Durable Medical Equipment Providers
 August 2009 Provider Bulletin Number 974 Durable Medical Equipment Providers Vacuum Assisted Wound Closure Therapy Negative pressure wound therapy (NPWT) must be requested and supplied by an enrolled durable
August 2009 Provider Bulletin Number 974 Durable Medical Equipment Providers Vacuum Assisted Wound Closure Therapy Negative pressure wound therapy (NPWT) must be requested and supplied by an enrolled durable
Arkansas Medicaid Health Care Providers Podiatrist Provider Manual Update Transmittal #99 Page 2
 Division of Medical Services Program Planning & Development P.O. Box 1437, Slot S-295 Little Rock, AR 72203-1437 501-682-8368 Fax: 501-682-2480 TDD: 501-682-6789 TO: Arkansas Medicaid Health Care Providers
Division of Medical Services Program Planning & Development P.O. Box 1437, Slot S-295 Little Rock, AR 72203-1437 501-682-8368 Fax: 501-682-2480 TDD: 501-682-6789 TO: Arkansas Medicaid Health Care Providers
Negative Pressure Wound Therapy Pumps
 Negative Pressure Wound Therapy Pumps Adopted from the National Government Services website. For any item to be covered by The Health Plan, it must: 1. Be eligible for a defined Medicare or The Health
Negative Pressure Wound Therapy Pumps Adopted from the National Government Services website. For any item to be covered by The Health Plan, it must: 1. Be eligible for a defined Medicare or The Health
Inpatient ICD-9-CM Mapping to ICD-10 PCS Procedures Involving the Application of PriMatrix AG Antimicrobial Dermal Repair Scaffold
 Inpatient ICD-9-CM Mapping to ICD-10 PCS Procedures Involving the Application of PriMatrix AG Antimicrobial Dermal Repair Scaffold Effective October 1, 2015, the Centers for & Medicaid Services (CMS) is
Inpatient ICD-9-CM Mapping to ICD-10 PCS Procedures Involving the Application of PriMatrix AG Antimicrobial Dermal Repair Scaffold Effective October 1, 2015, the Centers for & Medicaid Services (CMS) is
Coding Hot Topics. Lawrence A. Santi, DPM, FASPS Member, APMA Coding Committee
 Coding Hot Topics Lawrence A. Santi, DPM, FASPS Member, APMA Coding Committee 1 Disclaimer Participants in the APMA Coding Webinar are reminded that CPT code descriptors and coding policies do not reflect
Coding Hot Topics Lawrence A. Santi, DPM, FASPS Member, APMA Coding Committee 1 Disclaimer Participants in the APMA Coding Webinar are reminded that CPT code descriptors and coding policies do not reflect
Inpatient ICD-9-CM Mapping to ICD-10 PCS Procedures Involving the Application of Integra Bilayer Wound Matrix
 Inpatient ICD-9-CM Mapping to ICD-10 PCS Procedures Involving the Application of Integra Bilayer Wound Matrix Effective October 1, 2015, the Centers for & Medicaid Services (CMS) is implementing International
Inpatient ICD-9-CM Mapping to ICD-10 PCS Procedures Involving the Application of Integra Bilayer Wound Matrix Effective October 1, 2015, the Centers for & Medicaid Services (CMS) is implementing International
Apligraf A Reimbursement Case Study Antonio S. Montecalvo, CPA Director of Customer Support Services ISCT San Diego - May 5th, 2009
 Apligraf A Reimbursement Case Study Antonio S. Montecalvo, CPA Director of Customer Support Services ISCT San Diego - May 5th, 2009 Key Points Current Business Reimbursement Landscape FDA Approval Hospital
Apligraf A Reimbursement Case Study Antonio S. Montecalvo, CPA Director of Customer Support Services ISCT San Diego - May 5th, 2009 Key Points Current Business Reimbursement Landscape FDA Approval Hospital
2012 Head and Neck Reconstruction/ENT Repair Coding Observations
 Health Policy, Economics & Reimbursement Reimbursement Hotline Tel: 888.543.3656 Fax: 866.262.6977 reimbursement@lifecell.com www.lifecell.com 2012 Head and Neck Reconstruction/ENT Repair Coding Observations
Health Policy, Economics & Reimbursement Reimbursement Hotline Tel: 888.543.3656 Fax: 866.262.6977 reimbursement@lifecell.com www.lifecell.com 2012 Head and Neck Reconstruction/ENT Repair Coding Observations
FUTURE Local Coverage Determination (LCD): Application of Bioengineered Skin Substitutes to Lower Extremity Chronic Non-Healing Wounds (L35041)
 FUTURE Local Coverage Determination (LCD): Application of Bioengineered Skin Substitutes to Lower Extremity Chronic Non-Healing Wounds (L35041) Links in PDF documents are not guaranteed to work. To follow
FUTURE Local Coverage Determination (LCD): Application of Bioengineered Skin Substitutes to Lower Extremity Chronic Non-Healing Wounds (L35041) Links in PDF documents are not guaranteed to work. To follow
Coding for Ulcer Debridement
 Coding for Ulcer Debridement Faculty Jeffrey D. Lehrman, DPM, FASPS, MAPWCA Adjunct Clinical Professor Temple University School of Podiatric Medicine Panel Physician Crozer Center for Wound Healing and
Coding for Ulcer Debridement Faculty Jeffrey D. Lehrman, DPM, FASPS, MAPWCA Adjunct Clinical Professor Temple University School of Podiatric Medicine Panel Physician Crozer Center for Wound Healing and
Wound Assessment Report
 Wound Assessment Report Single Assessment, Single Wound Mary Taylor Assessment Patient ID MT4367147 Date of Birth 1939-4-18 Left Foot, Sole: Wound A Image taken 16-45-43 Area 1.7cm2 Perimeter 48mm Maximum
Wound Assessment Report Single Assessment, Single Wound Mary Taylor Assessment Patient ID MT4367147 Date of Birth 1939-4-18 Left Foot, Sole: Wound A Image taken 16-45-43 Area 1.7cm2 Perimeter 48mm Maximum
OF WOUNDS SENIOR AUDITOR CAROLINAS HEALTHCARE SYSTEM. AHIA 32 nd Annual Conference August 25-28, 2013 Chicago, Illinois
 1 THE WACKY WORLD OF WOUNDS ERIN RYDELL SENIOR AUDITOR CAROLINAS HEALTHCARE SYSTEM AHIA 32 nd Annual Conference August 25-28, 2013 Chicago, Illinois www.ahia.org Carolinas HealthCare System 2 Carolinas
1 THE WACKY WORLD OF WOUNDS ERIN RYDELL SENIOR AUDITOR CAROLINAS HEALTHCARE SYSTEM AHIA 32 nd Annual Conference August 25-28, 2013 Chicago, Illinois www.ahia.org Carolinas HealthCare System 2 Carolinas
Krystexxa (pegloticase) Document Number: IC-0158
 Krystexxa (pegloticase) Document Number: IC-0158 Last Review Date: 06/27/2017 Date of Origin: 02/07/20103 Dates Reviewed: 11/2013, 08/2014, 07/2015, 07/2016, 09/2016, 12/2016, 03/2017, 06/2017 I. Length
Krystexxa (pegloticase) Document Number: IC-0158 Last Review Date: 06/27/2017 Date of Origin: 02/07/20103 Dates Reviewed: 11/2013, 08/2014, 07/2015, 07/2016, 09/2016, 12/2016, 03/2017, 06/2017 I. Length
POLICIES AND PROCEDURE MANUAL
 POLICIES AND PROCEDURE MANUAL Policy: MP058 Section: Medical Benefit Policy Subject: Negative Pressure Wound Therapy I. Policy: Negative Pressure Wound Therapy II. Purpose/Objective: To provide a policy
POLICIES AND PROCEDURE MANUAL Policy: MP058 Section: Medical Benefit Policy Subject: Negative Pressure Wound Therapy I. Policy: Negative Pressure Wound Therapy II. Purpose/Objective: To provide a policy
End Diastolic Pneumatic Compression Boot as a Treatment of Peripheral Vascular Disease or Lymphedema. Original Policy Date
 MP 2.02.12 End Diastolic Pneumatic Compression Boot as a Treatment of Peripheral Vascular Disease or Lymphedema Medical Policy Section Medicine Issue 12:2013 Original Policy Date 12:2013 Last Review Status/Date
MP 2.02.12 End Diastolic Pneumatic Compression Boot as a Treatment of Peripheral Vascular Disease or Lymphedema Medical Policy Section Medicine Issue 12:2013 Original Policy Date 12:2013 Last Review Status/Date
Diabetic Foot Ulcer Treatment and Prevention
 Diabetic Foot Ulcer Treatment and Prevention Alexander Reyzelman DPM, FACFAS Associate Professor California School of Podiatric Medicine at Samuel Merritt University Diabetic Foot Ulcers One of the most
Diabetic Foot Ulcer Treatment and Prevention Alexander Reyzelman DPM, FACFAS Associate Professor California School of Podiatric Medicine at Samuel Merritt University Diabetic Foot Ulcers One of the most
Diabetic Foot Ulcers. Care for Patients in All Settings
 Diabetic Foot Ulcers Care for Patients in All Settings Summary This quality standard focuses on care for people who have developed or are at risk of developing a diabetic foot ulcer. The scope of the standard
Diabetic Foot Ulcers Care for Patients in All Settings Summary This quality standard focuses on care for people who have developed or are at risk of developing a diabetic foot ulcer. The scope of the standard
SAMPLE. Home Health Reference Tool For Nurses
 Home Health Reference Tool For Nurses Foundation Management Services, Inc. 2010. All rights reserved. Unauthorized reproduction is strictly prohibited. (10/09) Table of Contents 2 Page Medicare Eligibility
Home Health Reference Tool For Nurses Foundation Management Services, Inc. 2010. All rights reserved. Unauthorized reproduction is strictly prohibited. (10/09) Table of Contents 2 Page Medicare Eligibility
See Policy CPT/HCPCS CODE section below for any prior authorization requirements
 Effective Date: 1/1/2019 Section: MED Policy No: 378 Medical Officer 1/1/2019 Date Technology Assessment Committee Approved Date: 3/09; 10/14; 1/16 Medical Policy Committee Approved Date: 4/02; 4/03; 9/04;
Effective Date: 1/1/2019 Section: MED Policy No: 378 Medical Officer 1/1/2019 Date Technology Assessment Committee Approved Date: 3/09; 10/14; 1/16 Medical Policy Committee Approved Date: 4/02; 4/03; 9/04;
IHCP banner page. IHCP to cover CPT code IHCP to cover CPT code INDIANA HEALTH COVERAGE PROGRAMS BR JULY 3, 2018
 IHCP banner page INDIANA HEALTH COVERAGE PROGRAMS BR201827 JULY 3, 2018 IHCP to cover CPT 93050 Effective August 3, 2018, the Indiana Health Coverage Programs (IHCP) will cover Current Procedural Terminology
IHCP banner page INDIANA HEALTH COVERAGE PROGRAMS BR201827 JULY 3, 2018 IHCP to cover CPT 93050 Effective August 3, 2018, the Indiana Health Coverage Programs (IHCP) will cover Current Procedural Terminology
Determining Wound Diagnosis and Documentation Tips Job Aid
 Determining Wound Diagnosis and Job Aid 1 Coding Is this a traumatic injury from an accident? 800 Codes - Injury Section of the Coding Manual Code by specific site of injury. Only use for accidents or
Determining Wound Diagnosis and Job Aid 1 Coding Is this a traumatic injury from an accident? 800 Codes - Injury Section of the Coding Manual Code by specific site of injury. Only use for accidents or
See Policy CPT/HCPCS CODE section below for any prior authorization requirements
 Effective Date: 7/1/2018 Section: DME Policy No: 377 Medical Officer 7/1/18 Date Technology Assessment Committee Approved Date: 10/10; 10/13; 9/14: 9/15; 4/16 Medical Policy Committee Approved Date: 3/03;
Effective Date: 7/1/2018 Section: DME Policy No: 377 Medical Officer 7/1/18 Date Technology Assessment Committee Approved Date: 10/10; 10/13; 9/14: 9/15; 4/16 Medical Policy Committee Approved Date: 3/03;
VACUUM ASSISTED CLOSURE (V.A.C.) THERAPY: Mr. Ismazizi Zaharudin Jabatan pembedahan Am Hospital Kuala Lumpur
 VACUUM ASSISTED CLOSURE (V.A.C.) THERAPY: Mr. Ismazizi Zaharudin Jabatan pembedahan Am Hospital Kuala Lumpur Learning Objectives Define Negative Pressure Wound Therapy (NPWT) Discuss guidelines for the
VACUUM ASSISTED CLOSURE (V.A.C.) THERAPY: Mr. Ismazizi Zaharudin Jabatan pembedahan Am Hospital Kuala Lumpur Learning Objectives Define Negative Pressure Wound Therapy (NPWT) Discuss guidelines for the
ULCERS 1/12/ million diabetics in the US (2012) Reamputation Rate 26.7% at 1 year 48.3% at 3 years 60.7% at 5 years
 Jay Christensen D.P.M Advanced Foot and Ankle of Wisconsin 2-4% of the population at any given time will have ulcers 0.06-0.20% of the total population Average age of patients 70 years increased as more
Jay Christensen D.P.M Advanced Foot and Ankle of Wisconsin 2-4% of the population at any given time will have ulcers 0.06-0.20% of the total population Average age of patients 70 years increased as more
CASE 1: TYPE-II DIABETIC FOOT ULCER
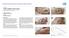 CASE 1: TYPE-II DIABETIC FOOT ULCER DIABETIC FOOT ULCER 48 YEAR-OLD MALE Mr. C., was a 48-year old man with a history of Type-II diabetes over the past 6 years. The current foot ulcer with corresponding
CASE 1: TYPE-II DIABETIC FOOT ULCER DIABETIC FOOT ULCER 48 YEAR-OLD MALE Mr. C., was a 48-year old man with a history of Type-II diabetes over the past 6 years. The current foot ulcer with corresponding
Diabetic Foot Ulcers. Alex Khan APRN ACNS-BC MSN CWCN CFCN WCN-C. Advanced Practice Nurse / Adult Clinical Nurse Specialist
 Diabetic Foot Ulcers Alex Khan APRN ACNS-BC MSN CWCN CFCN WCN-C Advanced Practice Nurse / Adult Clinical Nurse Specialist Organization of Wound Care Nurses www.woundcarenurses.org Objectives Identify Diabetic/Neuropathic
Diabetic Foot Ulcers Alex Khan APRN ACNS-BC MSN CWCN CFCN WCN-C Advanced Practice Nurse / Adult Clinical Nurse Specialist Organization of Wound Care Nurses www.woundcarenurses.org Objectives Identify Diabetic/Neuropathic
Venous Leg Ulcers. Care for Patients in All Settings
 Venous Leg Ulcers Care for Patients in All Settings Summary This quality standard focuses on care for people who have developed or are at risk of developing a venous leg ulcer. The scope of the standard
Venous Leg Ulcers Care for Patients in All Settings Summary This quality standard focuses on care for people who have developed or are at risk of developing a venous leg ulcer. The scope of the standard
2017 Physician Coding Survival Guide
 2017 Physician Coding Survival Guide Chapter 3: Dermatology Melanoma: Stop Melanoma Coding Errors Before They Spread If the dermatologist gets down to the fascia, would you still stick with an integumentary
2017 Physician Coding Survival Guide Chapter 3: Dermatology Melanoma: Stop Melanoma Coding Errors Before They Spread If the dermatologist gets down to the fascia, would you still stick with an integumentary
Imfinzi (durvalumab) (Intravenous)
 Imfinzi (durvalumab) (Intravenous) Last Review Date: 09/05/2018 Date of Origin: 05/30/2017 Dates Reviewed: 05/2017, 08/2017, 11/2017, 02/2018, 05/2018, 09/2018 Document Number: IC-0301 I. Length of Authorization
Imfinzi (durvalumab) (Intravenous) Last Review Date: 09/05/2018 Date of Origin: 05/30/2017 Dates Reviewed: 05/2017, 08/2017, 11/2017, 02/2018, 05/2018, 09/2018 Document Number: IC-0301 I. Length of Authorization
Final Rule CMS-1676-F was released on November 2, 2017 and finalized policies first proposed
 November 8, 2017 Subject: (CMS 1676 F) Summary of the Centers for Medicare and Medicaid Services Revisions to Payment Policies under the Physician Fee Schedule and Other Revisions to Part B for CY 2018
November 8, 2017 Subject: (CMS 1676 F) Summary of the Centers for Medicare and Medicaid Services Revisions to Payment Policies under the Physician Fee Schedule and Other Revisions to Part B for CY 2018
Local Coverage Article for Chiropractic Services (A47798) Contractor Information. Article Information. Contractor Name. Contractor Numbers
 Local Coverage Article for Chiropractic Services (A47798) Print Contractor Information Contractor Name Novitas Solutions, Inc. Contractor Numbers 12501, 12502, 12101, 12102, 12201, 12202, 12301, 12302,
Local Coverage Article for Chiropractic Services (A47798) Print Contractor Information Contractor Name Novitas Solutions, Inc. Contractor Numbers 12501, 12502, 12101, 12102, 12201, 12202, 12301, 12302,
The Practical Use of LIGASANO white in Plastic Surgery
 Practical experience 3 The Practical Use of LIGASANO white in Plastic Surgery Emergency Hospital of Mureş County, Romania Reports of practical experience from the burn center and plastic surgery department
Practical experience 3 The Practical Use of LIGASANO white in Plastic Surgery Emergency Hospital of Mureş County, Romania Reports of practical experience from the burn center and plastic surgery department
Contractor Name: Novitas Solutions, Inc. Contractor Number: Contractor Type: MAC B. LCD ID Number: L34834 Status: A-Approved
 LCD for Blood Glucose Monitoring in a Skilled Nursing Facility (SNF) (L34834) Contractor Name: Novitas Solutions, Inc. Contractor Number: 12502 Contractor Type: MAC B LCD ID Number: L34834 Status: A-Approved
LCD for Blood Glucose Monitoring in a Skilled Nursing Facility (SNF) (L34834) Contractor Name: Novitas Solutions, Inc. Contractor Number: 12502 Contractor Type: MAC B LCD ID Number: L34834 Status: A-Approved
LCD L B-type Natriuretic Peptide (BNP) Assays
 LCD L30559 - B-type Natriuretic Peptide (BNP) Assays Contractor Information Contractor Name: Novitas Solutions, Inc. Contractor Number(s): 12501, 12502, 12101, 12102, 12201, 12202, 12301, 12302, 12401,
LCD L30559 - B-type Natriuretic Peptide (BNP) Assays Contractor Information Contractor Name: Novitas Solutions, Inc. Contractor Number(s): 12501, 12502, 12101, 12102, 12201, 12202, 12301, 12302, 12401,
Successful Wound Management Strategies : An Introduction. Alex Khan, APRN ACNS-BC. Organization of Wound Care Nurses
 Successful Wound Management Strategies : An Introduction Alex Khan, APRN ACNS-BC Organization of Wound Care Nurses www.woundcarenurses.org Goals & Objectives The role and importance of wound care management
Successful Wound Management Strategies : An Introduction Alex Khan, APRN ACNS-BC Organization of Wound Care Nurses www.woundcarenurses.org Goals & Objectives The role and importance of wound care management
! " " # $ " " # $ " % " # $ # $
 ! " "#$ " "#$ " % "# $ #$ Skin Replacement Surgery Grafts 15040 Harvest of skin for cultured autograft 100 sq cm or less 15110 Epidermal autograft, trunk, arms, legs; first 100 sq cm or 1% of children
! " "#$ " "#$ " % "# $ #$ Skin Replacement Surgery Grafts 15040 Harvest of skin for cultured autograft 100 sq cm or less 15110 Epidermal autograft, trunk, arms, legs; first 100 sq cm or 1% of children
Nanogen Aktiv. Naz Wahab MD, FAAFP, FAPWCA Nexderma
 Nanogen Aktiv Naz Wahab MD, FAAFP, FAPWCA Nexderma Patient BM 75 y.o female with a history of Type 2 Diabetes, HTN, Hypercholesterolemia, Renal insufficiency, Chronic back Pain, who had undergone a L3-L4
Nanogen Aktiv Naz Wahab MD, FAAFP, FAPWCA Nexderma Patient BM 75 y.o female with a history of Type 2 Diabetes, HTN, Hypercholesterolemia, Renal insufficiency, Chronic back Pain, who had undergone a L3-L4
Understanding Your Costs and Coverage
 Understanding Your Costs and Coverage Thank you for choosing UW. We know that understanding your healthcare costs can be a challenge we re here to help. Your healthcare costs depend on many factors such
Understanding Your Costs and Coverage Thank you for choosing UW. We know that understanding your healthcare costs can be a challenge we re here to help. Your healthcare costs depend on many factors such
WOUND CARE UPDATE. -Commonly Used Skin Substitute Products For Wound. -Total Contact Casting. Jack W. Hutter DPM, FACFAS, C. ped.
 WOUND CARE UPDATE -Commonly Used Skin Substitute Products For Wound Closure -Total Contact Casting Jack W. Hutter DPM, FACFAS, C. ped. Commonly Used Skin Substitute Products for Wound Closure why are they
WOUND CARE UPDATE -Commonly Used Skin Substitute Products For Wound Closure -Total Contact Casting Jack W. Hutter DPM, FACFAS, C. ped. Commonly Used Skin Substitute Products for Wound Closure why are they
End-Diastolic Pneumatic Compression Boot as a Treatment of Peripheral Vascular Disease or Lymphedema
 End-Diastolic Pneumatic Compression Boot as a Treatment of Peripheral Vascular Disease or Applies to all products administered or underwritten by Blue Cross and Blue Shield of Louisiana and its subsidiary,
End-Diastolic Pneumatic Compression Boot as a Treatment of Peripheral Vascular Disease or Applies to all products administered or underwritten by Blue Cross and Blue Shield of Louisiana and its subsidiary,
Links in PDF documents are not guaranteed to work. To follow a web link, please use the MCD Website. Jurisdiction Texas. Retirement Date N/A
 Local Coverage Determination (LCD): Chiropractic Services (L35424) Links in PDF documents are not guaranteed to work. To follow a web link, please use the MCD Website. Contractor Information Contractor
Local Coverage Determination (LCD): Chiropractic Services (L35424) Links in PDF documents are not guaranteed to work. To follow a web link, please use the MCD Website. Contractor Information Contractor
Acute and Chronic WOUND ASSESSMENT. Wound Assessment OBJECTIVES ITEMS TO CONSIDER
 WOUND ASSESSMENT Acute and Chronic OBJECTIVES Discuss classification systems and testing methods for pressure ulcers, venous, arterial and diabetic wounds List at least five items to be assessed and documented
WOUND ASSESSMENT Acute and Chronic OBJECTIVES Discuss classification systems and testing methods for pressure ulcers, venous, arterial and diabetic wounds List at least five items to be assessed and documented
NPWT Case Series EXPERIENCES WITH INVIA MOTION. Precious life Progressive care. Invia Motion Negative Pressure Wound Therapy
 NPWT Case Series EXPERIENCES WITH INVIA MOTION Invia Motion Negative Pressure Wound Therapy Precious life Progressive care npwt_case_booklet_a4.indd 1 18.12.13 13:17 Chronic sacral pressure ulcer Case
NPWT Case Series EXPERIENCES WITH INVIA MOTION Invia Motion Negative Pressure Wound Therapy Precious life Progressive care npwt_case_booklet_a4.indd 1 18.12.13 13:17 Chronic sacral pressure ulcer Case
Negative Pressure Wound Therapy (NPWT) (Vacuum-Assisted Wound Closure)
 Negative Pressure Wound Therapy (NPWT) (Vacuum-Assisted Wound Closure) Date of Origin: 03/2018 Last Review Date: 03/28/2018 Effective Date: 03/28/2018 Dates Reviewed: 03/2018 Developed By: Medical Necessity
Negative Pressure Wound Therapy (NPWT) (Vacuum-Assisted Wound Closure) Date of Origin: 03/2018 Last Review Date: 03/28/2018 Effective Date: 03/28/2018 Dates Reviewed: 03/2018 Developed By: Medical Necessity
Jurisdiction New Mexico. Retirement Date N/A
 Local Coverage Determination (LCD): Chiropractic Services (L34816) Contractor Information Contractor Name Novitas Solutions, Inc. opens in new Contract Number 04212 Contract Type A and B MAC J - H LCD
Local Coverage Determination (LCD): Chiropractic Services (L34816) Contractor Information Contractor Name Novitas Solutions, Inc. opens in new Contract Number 04212 Contract Type A and B MAC J - H LCD
12501, 12502, 12101, 12102, 12201, 12202, 12301, 12302, 12401, 12402, 12901
 https://www.novitas-solutions.com/policy/mac-ab/l27547-r16.html LCD - Wound CarePrint Contractor Information Contractor Name: Novitas Solutions, Inc. Contractor Number(s): 12501, 12502, 12101, 12102, 12201,
https://www.novitas-solutions.com/policy/mac-ab/l27547-r16.html LCD - Wound CarePrint Contractor Information Contractor Name: Novitas Solutions, Inc. Contractor Number(s): 12501, 12502, 12101, 12102, 12201,
Use of an Acellular Regenerative Tissue Matrix Over Chronic Wounds
 Use of an Acellular Regenerative Tissue Matrix Over Chronic Wounds D. Heath Stacey, MD Northwest Arkansas Center for Plastic Surgery, Fayetteville, Ark Correspondence: dheathstacey@gmail.com Keywords:
Use of an Acellular Regenerative Tissue Matrix Over Chronic Wounds D. Heath Stacey, MD Northwest Arkansas Center for Plastic Surgery, Fayetteville, Ark Correspondence: dheathstacey@gmail.com Keywords:
Amputation is the removal of a limb by trauma, medical illness, or surgery. As a surgical measure, it is used to control pain or a disease process in
 Amputation is the removal of a limb by trauma, medical illness, or surgery. As a surgical measure, it is used to control pain or a disease process in the affected limb, such as malignancy or gangrene.
Amputation is the removal of a limb by trauma, medical illness, or surgery. As a surgical measure, it is used to control pain or a disease process in the affected limb, such as malignancy or gangrene.
2017 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY. MEASURE TYPE: Process
 Measure #127 (NQF 0416): Diabetes Mellitus: Diabetic Foot and Ankle Care, Ulcer Prevention Evaluation of Footwear National Quality Strategy Domain: Effective Clinical Care 2017 OPTIONS FOR INDIVIDUAL MEASURES:
Measure #127 (NQF 0416): Diabetes Mellitus: Diabetic Foot and Ankle Care, Ulcer Prevention Evaluation of Footwear National Quality Strategy Domain: Effective Clinical Care 2017 OPTIONS FOR INDIVIDUAL MEASURES:
HUSKY Health Benefits and Prior Authorization Requirements Grid* Behavioral Health Partnership Effective: January 1, 2012
 Behavioral Health Health and Behavior Assessments (CPT 96150-96155) When Performed by Psychologists Mental Health Inpatient 100% covered under medical benefit for members with diagnoses outside the range
Behavioral Health Health and Behavior Assessments (CPT 96150-96155) When Performed by Psychologists Mental Health Inpatient 100% covered under medical benefit for members with diagnoses outside the range
Local Coverage Determination (LCD) for Chiropractic Services (L34816) (Posted for Notice)
 Local Coverage Determination (LCD) for Chiropractic Services (L34816) (Posted for Notice) Print Contractor Information Contractor Name Novitas Solutions, Inc. Contractor Numbers 04911, 07101, 07102, 07201,
Local Coverage Determination (LCD) for Chiropractic Services (L34816) (Posted for Notice) Print Contractor Information Contractor Name Novitas Solutions, Inc. Contractor Numbers 04911, 07101, 07102, 07201,
NEGATIVE PRESURE WOUND THERAPY PROGRAM
 NEGATIVE PRESURE WOUND THERAPY PROGRAM WWW.USTOMWOUNDCARE.COM 866-802-0006/901-619-8897 SUPPORT@USTOM.COM TABLE OF CONTENTS 1. 2. 3. 4. To place an order: 1. Please visit our website 2. Please email to
NEGATIVE PRESURE WOUND THERAPY PROGRAM WWW.USTOMWOUNDCARE.COM 866-802-0006/901-619-8897 SUPPORT@USTOM.COM TABLE OF CONTENTS 1. 2. 3. 4. To place an order: 1. Please visit our website 2. Please email to
code it ACTISHIELD HCPCS Device Codes Amniotic Barrier Membrane HCPCS Code C9399 Description Q4100 C5271
 code it HCPCS Device Codes 2015 Reimbursement Codes The following codes contained within this document are representative of possible services or diagnoses that may be associated with use of Wright products.
code it HCPCS Device Codes 2015 Reimbursement Codes The following codes contained within this document are representative of possible services or diagnoses that may be associated with use of Wright products.
Local Coverage Determination (LCD): RAST Type Tests ( L30524 )
 Page 2 of 6 Local Coverage Determination (LCD): RAST Type Tests ( L30524 ) Contractor Information Contractor Name Novitas Solutions, Inc. Contract Number 12502 Contract Type A and B MAC LCD Information
Page 2 of 6 Local Coverage Determination (LCD): RAST Type Tests ( L30524 ) Contractor Information Contractor Name Novitas Solutions, Inc. Contract Number 12502 Contract Type A and B MAC LCD Information
Wound Care Evaluation by Kris Dalseg MS PT CWS CLT
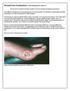 Wound Care Evaluation by Kris Dalseg MS PT CWS CLT This document is intended to describe a standard wound care evaluation for healthcare practitioners. In healthcare, all aspects of our treatment have
Wound Care Evaluation by Kris Dalseg MS PT CWS CLT This document is intended to describe a standard wound care evaluation for healthcare practitioners. In healthcare, all aspects of our treatment have
Original Date: October 2015 LUMBAR SPINAL FUSION FOR
 National Imaging Associates, Inc. Clinical guidelines Original Date: October 2015 LUMBAR SPINAL FUSION FOR Page 1 of 9 INSTABILITY AND DEGENERATIVE DISC CONDITIONS FOR CMS (MEDICARE) MEMBERS ONLY CPT4
National Imaging Associates, Inc. Clinical guidelines Original Date: October 2015 LUMBAR SPINAL FUSION FOR Page 1 of 9 INSTABILITY AND DEGENERATIVE DISC CONDITIONS FOR CMS (MEDICARE) MEMBERS ONLY CPT4
The Use of the. in Clinical Practice
 The Use of the SNAP Therapy System in Clinical Practice It s an ultraportable, mechanically-powered disposable NPWT. By Animesh Bhatia, DPM, CWS This article is written exclusively for PM and appears courtesy
The Use of the SNAP Therapy System in Clinical Practice It s an ultraportable, mechanically-powered disposable NPWT. By Animesh Bhatia, DPM, CWS This article is written exclusively for PM and appears courtesy
Discussion Topics. Calcium Alginates. DME For the Diabetic Foot 1/25/2017. Jeffrey D. Lehrman, DPM, FASPS, FACFAS, MAPWCA
 DME For the Diabetic Foot Jeffrey D. Lehrman, DPM, FASPS, FACFAS, MAPWCA Editorial Advisory Board, WOUNDS Board of Directors, American Society of Podiatric Surgeons Board of Directors, American Professional
DME For the Diabetic Foot Jeffrey D. Lehrman, DPM, FASPS, FACFAS, MAPWCA Editorial Advisory Board, WOUNDS Board of Directors, American Society of Podiatric Surgeons Board of Directors, American Professional
Intravitreal Avastin (Bevacizumab)
 Intravitreal Avastin (Bevacizumab) Date of Origin: 10/18/2018 Last Review Date: 10/18/2018 Effective Date: 10/18/2018 Dates Reviewed: 10/2018 Developed By: Medical Criteria Committee I. Length of Authorization
Intravitreal Avastin (Bevacizumab) Date of Origin: 10/18/2018 Last Review Date: 10/18/2018 Effective Date: 10/18/2018 Dates Reviewed: 10/2018 Developed By: Medical Criteria Committee I. Length of Authorization
Tissue-Engineered Skin Substitutes
 Tissue-Engineered Skin Substitutes Policy Number: Original Effective Date: MM.06.018 06/01/2012 Line(s) of Business: Current Effective Date: HMO; PPO 06/01/2012 Section: Surgery Place(s) of Service: Outpatient/Office
Tissue-Engineered Skin Substitutes Policy Number: Original Effective Date: MM.06.018 06/01/2012 Line(s) of Business: Current Effective Date: HMO; PPO 06/01/2012 Section: Surgery Place(s) of Service: Outpatient/Office
Tecentriq (atezolizumab) (Intravenous)
 Tecentriq (atezolizumab) (Intravenous) Last Review Date: 06/01/2018 Date of Origin: 06/28/2016 Document Number: IC-0278 Dates Reviewed: 06/2016, 08/2016, 10/2016, 02/2017, 04/2017, 08/2017, 11/2017, 02/2018,
Tecentriq (atezolizumab) (Intravenous) Last Review Date: 06/01/2018 Date of Origin: 06/28/2016 Document Number: IC-0278 Dates Reviewed: 06/2016, 08/2016, 10/2016, 02/2017, 04/2017, 08/2017, 11/2017, 02/2018,
XOLAIR (omalizumab) Prior Authorization
 MP9309 Covered Service: Prior Authorization Required: Additional Information: Yes when meets criteria below Yes as shown below May only be prescribed by Allergy, Pulmonary, Immunology or Dermatology specialists
MP9309 Covered Service: Prior Authorization Required: Additional Information: Yes when meets criteria below Yes as shown below May only be prescribed by Allergy, Pulmonary, Immunology or Dermatology specialists
Eylea (aflibercept) Document Number: IC-0026
 Eylea (aflibercept) Document Number: IC-0026 Last Review Date: 3/1/2018 Date of Origin: 02/07/2013 Dates Reviewed: 03/07/2013, 06/2013, 09/2013, 12/2013, 03/2014, 06/2014, 09/2014, 12/2014, 03/2015, 04/2015,
Eylea (aflibercept) Document Number: IC-0026 Last Review Date: 3/1/2018 Date of Origin: 02/07/2013 Dates Reviewed: 03/07/2013, 06/2013, 09/2013, 12/2013, 03/2014, 06/2014, 09/2014, 12/2014, 03/2015, 04/2015,
2018 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY. MEASURE TYPE: Process
 Quality ID #127 (NQF 0416): Diabetes Mellitus: Diabetic Foot and Ankle Care, Ulcer Prevention Evaluation of Footwear National Quality Strategy Domain: Effective Clinical Care 2018 OPTIONS FOR INDIVIDUAL
Quality ID #127 (NQF 0416): Diabetes Mellitus: Diabetic Foot and Ankle Care, Ulcer Prevention Evaluation of Footwear National Quality Strategy Domain: Effective Clinical Care 2018 OPTIONS FOR INDIVIDUAL
Medical Policy Original Effective Date: 01/23/2019
 Page 1 of 12 Disclaimer Refer to the member s specific benefit plan and Schedule of Benefits to determine coverage. This may not be a benefit on all plans or the plan may have broader or more limited benefits
Page 1 of 12 Disclaimer Refer to the member s specific benefit plan and Schedule of Benefits to determine coverage. This may not be a benefit on all plans or the plan may have broader or more limited benefits
OP-14: SIMULTANEOUS USE OF BRAIN COMPUTED TOMOGRAPHY (CT) AND SINUS COMPUTED TOMOGRAPHY (CT)
 OP-14: SIMULTANEOUS USE OF BRAIN COMPUTED TOMOGRAPHY (CT) AND SINUS COMPUTED TOMOGRAPHY (CT) Description of Measure This measure calculates the percentage of Brain CT studies with a simultaneous Sinus
OP-14: SIMULTANEOUS USE OF BRAIN COMPUTED TOMOGRAPHY (CT) AND SINUS COMPUTED TOMOGRAPHY (CT) Description of Measure This measure calculates the percentage of Brain CT studies with a simultaneous Sinus
PALLIATIVE WOUND CARE
 PALLIATIVE WOUND CARE Scott Bolhack, MD, FACP President/CEO TLC HealthCare Companies Learning Objectives: Describe the aspects of palliative wound care.. Demonstrate palliative practices used in the care
PALLIATIVE WOUND CARE Scott Bolhack, MD, FACP President/CEO TLC HealthCare Companies Learning Objectives: Describe the aspects of palliative wound care.. Demonstrate palliative practices used in the care
o Venous edema o Stasis ulcers o Varicose veins (not including spider veins) o Lipodermatosclerosis
 Wound Care Equipment and Supply Benefits to Change for Texas Medicaid July 1, 2018 Effective for dates of service on or after July 1, 2018, wound care equipment and supply benefits will change for Texas
Wound Care Equipment and Supply Benefits to Change for Texas Medicaid July 1, 2018 Effective for dates of service on or after July 1, 2018, wound care equipment and supply benefits will change for Texas
Contractor Information
 Local Coverage Determination (LCD): Chiropractic Services (L35424) Links in PDF documents are not guaranteed to work. To follow a web link, please use the MCD Website. Contractor Information CONTRACTOR
Local Coverage Determination (LCD): Chiropractic Services (L35424) Links in PDF documents are not guaranteed to work. To follow a web link, please use the MCD Website. Contractor Information CONTRACTOR
Description. Section: Medicine Effective Date: April 15, 2015 Subsection: Medicine Original Policy Date: September 13, 2012 Subject:
 Last Review Status/Date: March 2015 Description Page: 1 of 6 Low-frequency ultrasound (US) in the kilohertz (KHz) range may improve wound healing. Several noncontact devices have received regulatory approval
Last Review Status/Date: March 2015 Description Page: 1 of 6 Low-frequency ultrasound (US) in the kilohertz (KHz) range may improve wound healing. Several noncontact devices have received regulatory approval
Corporate Medical Policy
 Corporate Medical Policy Topical Negative Pressure Therapy for Wounds File Name: Origination: Last CAP Review: Next CAP Review: Last Review: topical_negative_pressure_therapy_for_wounds 12/1998 5/2018
Corporate Medical Policy Topical Negative Pressure Therapy for Wounds File Name: Origination: Last CAP Review: Next CAP Review: Last Review: topical_negative_pressure_therapy_for_wounds 12/1998 5/2018
