Nov. 20, 1973 G. GERGELY 3,773,922 METHOD FOR THE MANUFACTURE OF EFFERVESCENT TABLETS Filed April 6, 1972
|
|
|
- Curtis Morton
- 6 years ago
- Views:
Transcription
1 Nov. 20, 1973 G. GERGELY 3,773,922 METHD FR THE MANUFACTURE F EFFERVESCENT TABLETS Filed April 6, 1972
2 United States Patent ffice 3,773,922 Patented Nov. 20, ,773,922 METHD FR THE MANUFACTURE F EFFERVESCENT TABLETS Gerhard Gergely, Vienna, Austria, assignor to Societe Francaise de Developpement et de Recherche Sofrader, Paris, France Filed Apr. 6, 1972, Ser. No. 241,772 Claims priority, application France, Apr. 6, 1971, 12,17; Sept. 29, 1971, U.S. C nt. C. A61k 9/00 8 Claims ABSTRACT F THE DISCLSURE Method for the manufacture of effervescent tablets con taining on the one hand at least one medicine, on the other hand an effervescent mixture comprising the granulation of the effervescent mixture possibly containing the med icine in three stages: (1) the humidification of the effervescent mixture by means of a quantity of water less than 1% by weight with respect to the alkaline bicarbonate; (2) the pre-drying of the humidified effervescent mixture in a fluidized bed, by hot air, the moisture content of which does not exceed g./m3 and the temperature of which does not exceed 60 C., this operation taking place by a sequence of short drying operations so as to stop the chemical reaction; and (3) the final drying of the granulated mixture by hot air, the moisture content of which does not exceed g/m3, and the temperature of which does not exceed 60 C., in a fluidized bed, until the residual content of water in the granules is less than 0.2% by weight and, preferably comprised between 0.0 and 0.1%. The present invention relates to a method for the manu facture of effervescent tablets containing: (a) a medicine, for example acetylsalicylic acid or paracetamol, possibly in combination with absorbic acid, (b) an effervescent mixture contituted, on the one hand, by an alkaline carbonate or bicarbonate, on the other hand, by an aliphatic polycarboxylic acid or an acid alka line salt of such an acid, (c) possibly customary ingredients such as binding agents, Sweetening agents, flavouring agents, colouring agents. The customary manufacture of effervescent tablets sub stantially comprises the following operations: (a) the preparation of the effervescent mixture; (b) the preparation of the active mixture containing the medicine; (c) the combination of these two mixtures in a final mixture and the production of tablets of the latter. More particularly, in this standard manufacture, the effervescent mixture is prepared by mixing the alkaline bicarbonate in the dry state with the aliphatic polycar boxylic acid in the form of a powder and a variable amount of water is vaporized onto the mixture obtained in order to granulate it; the grains obtained are dried in a thin layer on sieves in an oven for a period of time varying from 2 to 3 hours according to the reltaive moisture con tent inside this oven. The effervescent mixture obtained is then added to the active mixture and again dried in a thin layer on sieves in an oven for 11 hours and finally made into tablets. Now the applicant has ascertained that in the course of the granulation of the effervescent mixture by humidifi cation, a chemical reaction takes place in this mixture between the acid and the bicarbonate with the liberation of carbon dioxide gas leading to the swelling of the grains and with the formation of water which further accelerates Said reaction. Due to this, it is absolutely necessary to stop this chemical reaction as soon as possible in order to limit the deterioration of the effervescent mixture. The method according to the invention comprises the granulation of the effervescent mixture, possibly contain ing the medicine, in three main stages: (1) The humidification of the effervescent mixture pos sibly containing the medicine, or of one of the constituents of the said mixture by a quantity of water less than 1% by Weight with respect to the alkaline bicarbonate, in order to initiate the chemical reaction between the alkaline bicar bonate and the citric acid in the presence of moisture with the liberation of carbon dioxide gas and the formation of water; (2) The predrying of the humidified mixture, in a fluid ized bed, by hot air, the temperature of which does not exceed 60 C. and the moisture content of which does not exceed g. per m.8, by a sequence of short drying opera tions each of 4 to 8 mins. by varying the speed of flow of the drying air in each operation from a maximum of approximately 6-8 m./s. to 0 m./s. at the end of said operation, and this is an order to stop the chemical re action; (3) The final drying of the granulated mixture, in a fluidized bed, by hot air, the temperature of which does not exceed 60 C, and the moisture content of which does not exceed g./m3 at the maximum speed of flow of the drying air until the residual content of water of the gran ules is less than 0.2% by weight, preferably comprised between 0.0 and 0.1%. Generally, the overall duration of the predrying and of the final drying is from 2 to 30 minutes approximately (whereas, in the customary process, the duration of drying of the effervescent mixture is approximately 11 hours) such that the chemical reaction is quickly stopped. The effervescent mixture thus obtained is then combined with the active mixture prepared separately and the final mixture is immediately made into tablets, without any ne cessity to dry it again. The tablet obained dissolves in water very quickly ( secs. to 1 min.). The invention is illustrated by the following nonlimiting examples. EXAMPLE 1. In this example, the manufacture of effervescent tablets of vitaminized aspirin having the following formula is described: Formula of an effervescent tablet weighing 3.0 g. G. Aceylsalicylic acid AScorbic acid Glycocol Sodium bicarbonate Anhydrous citric acid Sodium benzoate The effervescent mixture is firstly prepared as follows, from the following ingredients: Glycocol (binding agent) Sodium bicarbonate Anhydrous citric acid ,92 these ingredients having been previously passed through a sieve having a mesh size of 2 mm. The sodium bicarbonate is introduced into a mixture of the "Lodige FKM 600 D' type equipped with anti-deposit blades and the apparatus is started for 1 min. then 1,12 ml. of demineralized water is sprayed in; then, the mixer is stopped and the citric acid and glycocol are poured in, then the mixer is re-tarted for 3 mins. The powdery mix
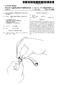
3 3 ture becomes compact. It is removed into a carrier and dried. DESCRIPTIN F THE DRAWING The drier used for ths purpose is shown diagrammati cally in the single figure of the accompanying drawing. The operating process of such a drier consists of drying the granulated material in a fluidized bed by passing through it a current of hot air treated with regard to the moisture content in such a way that the product forms a suspension in the vessel. The necessary current of air is produced by a turbine 1 incorporated in the upper part of the apparatus and directly connected to a drive motor 2. The air drawn in from outside enters at 3, the dust is removed therefrom by a preliminary filter 4 then heated by a heater to the desired temperature (60. C.). The current of air circulating in an upwards direction according to the arrows F, F, and F through the carrier 6 containing the product to be dried, dries this granulated material in a very short time; to this end the base of the carrier is constituted by perforated sheet metal to which is fixed a stainless steel, fine mesh gauze. By regulating the speed of flow of the air it is possible to reduce or increase the turbulence of the material to be dried. A Nylon filter 7 covers the carriers and pre vents the escape of any fine particles of material. After the drying operation, the filter is simply shaken, either manually or by means of an automatic device so that these particles fall into the carrier. The air saturated with moisture passes to the outside. A part of this air is recycled by means of a recycling valve 8 via a distributor 9 and a filter 10, is mixed with fresh air supplied by a blower 11; the mixture is dried in a dehy drator 12 and is supplied at 3. The operating procedure is as follows: (1) The pre-drying with hot air at 60 C., in a fluidized bed, comprises: (a) a first operation lasting 8 mins. with a speed of flow of the air decreasing continuously from 8 m./s. to 0 m./s. from the beginning to the end of the operation, with a flow of air decreasing continuously by degrees from 29 m.8/min. to 0 m.8/min.; (b) a second operation lasting 7 minutes, with a speed of flow of the air decreasing continuously from 6 m./s. to 0 m./s. and with a flow of air decreasing continuously from 22 m.8/min. to 0 m.8/min.; (c) a third operation lasting 4 mins., with a speed of flow of the air decreasing continuously from. m/s. to 0 m./s. and with a flow of air decreasing continu ously from 20 m.3/min. to 0 m.8/min. The filter 7 is shaken at the end of each operation. (2) The drying with hot air at 60 C. in a fluidized bed lasts 6 mins. with a speed of flow of the drying air of 6 m./s, and with a flow of air of 22 m2/min. The total pre-drying and drying time of the granulated material is 2 mins. After drying, the effervescent mixture is granulated by means of a granulator having a mesh size of 1.4 mm. The granulated material is recovered in a container. Its water content is from 0.0 to 0.1%. In addition, the active mixture is prepared separately from the following ingredients: Acetylsalicylic acid Ascorbic acid ,000 Sodium benzoate (binding agent) The acetylsalicylic acid and the ascorbic acid are passed through a sieve having a mesh size of 2 mm. The products are then weighed and poured into a mixer. The previously sieved sodium benzoate is weighed then poured into the mixer. The mixing lasts 12 minutes. The effervescent mixture and the active mixture are 3,773, combined in a mixer and mixed for 20 min.; then the final mixture is guided towards the compression chains. Tablets are thus obtained having a diameter of 23 mm., an average weight of 3.0 g., an average thickness of.61 mm., a breakage pressure of 00 to 6000 kg. and a dissolving time of -60 secs. EXAMPLE 2 For the manufacture of effervescent tablets of aspirin without vitamin C according to one of the following for mulae, one proceeds as in Example 1: Formula of an effervescent tablet weighing 3.2g. Formula Formula without with NaIK potassium equilibrium, ion, g. Acetylsalicylic acid Sodium bicarbonate Potassium bicarbonate Anhydrous citric acid Glycocol a -- as Sodium benzoate Formula of an effervescent tablet weighing 3.7g: 2 Formula Formula without with Nafs potassium equilibrium, ion, g. Acetylsalicylic acid Sodium bicarbonate Potassium bicarbonate Citric acid Glycocol EXAMPLE 3 In this example the manufacture of effervescent tablets of vitaminized paracetamol according to the following formula is described: Formula for an effervescent tablet weighing 3 g. G. Paracetamol Vitamin C Sodium saccharinate Monosodium carbonate Monopotassium carbonate Monosodium citrate Sodium benzoate Anhydrous citric acid Firstly, one proceeds to the granulation of the para cetamol from the following ingredients: Paracetamol Monosodium carbonate Sodium saccharinate Purified water The sodium saccharinate is dissolved in water to ob tain a moist granulated material. The monosodium carbonate and the paracetamol are sieved separately on a sieve having a mesh size of 2 mm. The sieved monosodium carbonate is poured into a mixer equipped with anti-deposit blades, it is wetted with the aqueous saccharinate solution and mixed for 2 min utes; the paracetamol is added to it and it is mixed for minutes then the emptying flap is opened whilst the mixer rotates; the product is discharged into a carrier and dried in a fluidized bed in a drier having a current of hot air according to the procedure given in Example 1, for ap proximately half an hour at 60 C. The air used for the drying is previously treated in an appropriate device, so as to reduce its humidity rate to a value of less than g/m3. When the mixture is dry, it is calibrated by means of a granulator using a grid having a mesh size of 1. mm. Thus the mixture No. 1 is obtained ( kg).
4 ne then proceeds to the granulation of the effer vescent mixture and of the paracetamol with the follow ing ingredients: Monosodium carbonate Monopotassium carbonate Monosodium citrate Anhydrous citric acid Mixture No Distilled Water ethanol The monosodium and monopotassium carbonates are sieved through a sieve having a mesh size of 2 mm. They are placed in a high capacity mixer equipped with anti-deposit blades. The mixer is allowed to turn, then the distilled water and ethanol are added, it is allowed to turn for minutes. Mixture No. 1 is then added and mixed for two minutes. The previously sieved monoso dium citrate and anhydrous citric acid are poured in and briefly mixed. The mixer is operated for 2 minutes. The mixture is poured into a carrier and is dried in a fluidized bed, in a drier having a current of hot air according to the procedure described in Example 1 at 60 C. for approximately 4 hour until the quantity of water is no greater than 0.20%. The mixture is thus calibrated as previously by means of a granulator using a grid with a mesh size of 1.4 mm., 19 kg. of mixture No. 2 are obtained. The final mixture is thus prepared as follows: 1 kg. of vitamin C and 1 kg. of sodium benzoate lubricant previously sieved on a sieve with a mesh size of 1 mm. are poured into a cubic mixer, mixture No. 2 is then added and the mixer operates for 20 mins.; 22 kg. of the final mixture are obtained. The final mixture is distributed in the hoppers of com pressing machines to give tablets weighing 3.00 g. (7.000 tablets) of 23 mm. diameter,.10 mm. average thickness, having a breaking pressure greater than 3 kg. and a dissolving time of less than 2 mins. EXAMPLE 4 In order to manufacture effervescent tablets of parace tamol (without vitamin C) according to the following formula, on proceeds as in Example 3: Formula of an effervescent paracetamol tablet without vitamin C 9. Paracetamol Potassium bicarbonate Sodium bicarbonate Anhydrous citric acid Anhydrous monosodium citrate Sodium Saccharinate Sodium benzoate ,778,922 2,704 What is claimed is: 1. Method for the manufacture of effervescent tablets 60 containing at least one medicine and an effervescent mix ture constituted by at least one alkaline bicarbonate and one aliphatic polycarboxylic acid or one alkaline metal acid salt of said acid and optionally containing ingredi ents such as binding agents, sweetening agents, compres sion lubricants, said process being characterised in that it comprises the granulation of the effervescent mixture possibly containing the medicine in three stages: (1) the humidification of the effervescent mixture by means of a quantity of water less than 1% by weight with respect to the alkaline bicarbonate in order to initiate the chemical reaction between said bicar bonate and said acid in the presence of moisture, with the liberation of carbon dioxide gas and the formation of water; (2) the predrying of the humidifying effervescent mix ture, in a fluidized bed, by hot air, the moisture con tent of which does not exceed g/m3 and the tem perature of which does not exceed 60 C, this op eration taking place by a sequence of short drying operations in order to stop the chemical reaction; and (3) the final drying of the granulated mixture by hot air, the moisture content of which does not exceed g/m3, and the temperature of which does not exceed 60 C., in a fluidized bed, until the residual water content of the granules is less than 0.2% by weight and, preferably, comprised between 0.0 and 0.1%. 2. Method according to claim 1, characterised in that the predrying of the humidified, effervescent mixture, in a fluidized bed, by hot air comprises a sequence of brief fluidization operations each of 4-8 mins, the speed of flow of the drying air varying in each operation from a maximum of 6-8 m./s, approximately to 0 m./s, at the end of said operation. 3. Method according to claim 1, characterised in that the final drying of the granulated mixture by hot air at a temperature not exceeding 60 C., in a fluidized bed, is carried out at the maximum speed of flow of the dry ing air of 6-8 m./s, approximately. 4. Method according to claim 1, characterised in that the total duration of the predrying and of the final dry ing is from 2 to 30 mins. for an initial grain size of the effervescent mixture not exceeding 2 mm.. Method according to claim 1, characterised in that the medicine is acetylsalicylic acid or acetylsalicylic acid, combined with ascorbic acid. 6. Method according to claim 1, characterised in that the medicine is paracetamol or paracetamol combined With ascorbic acid. 7. Method according to claim 6, characterised in that it comprises the following stages: (1) the granulation of the paracetamol with at the most half the alkaline bicarbonate (a) by humdifi cation by means of a quantity of water less than 3.% by weight with respect to the carbonate, and this by moistening and firstly mixing the carbonate then the paracetamol with an aqueous solution of sodium saccharinate, (b) by predrying and drying the humidified mixture, in a fluidized bed, by a cur rent of hot air at a temperature not exceeding 60 C., and having a moisture content not exceeding g/m., for approximately 2 hour. (2) the granulation of the paracetamol thus treated with the effervescent mixture (a) by humdification of the remaining alkaline bicarbonate mixture with a quantity of a wetting liquid less than % by weight with respect to the bicarbonate, mixing of the moistened mixture firstly with the granulated para cetamol, then with the monosodium citrate and the anhydrous citric acid, the wetting liquid may be either water, or a mixture of ethanol or acetone with water in proportions ranging from 30 to 70% and (b) by predrying and drying of the mixture in a fluidized bed, by a current of hot air at a tem perature not exceeding 60 C. and having a moisture content not exceeding g/m3 for 2 hour approx imately until the water content is no greater than 0.2%. (3) the incorporation of sodium benzoate or vitamin C and mixtures thereof with the granulated paracet amol and effervescent mixture to form the final mix ture which is made directly into tablets. 8. Method according to claim, characterised in that it comprises the following stages: (1) the granulation of the effervescent mixture (a) by humidification of said mixture by a quantity of water less than 1% by weight with respect to the
5 3,773, alkaline bicarbonate, (b) by predrying and drying References Cited the humidified mixture in a fluidized bed, by a Current UNITED STATES PATENTS of hot air at a temperature not exceeding 60 C. and having a moisture content not exceeding 1,,86 4/1923 Pelic X g./m. for approximately 2 hour, 3,63,914 4/1972 Schmitt (2) the preparation of an active mixture in the dry state containing acetylsalicylic acid and possibly SHEP K. RSE, Primary Examiner ascorbic acid, (3) the combination of the granulated effervescent U.S. C1. X.R. mixture and of the active mixture in a final mix ; 99-78; ; ; , 280 ture and the making of tablets from said final mixture.
EUROPEAN QUALIFYING EXAMINATION 2006
 EUROPEAN QUALIFYING EXAMINATION 2006 PAPER A CHEMISTRY This paper comprises: * Letter from the applicant 2006/A(Ch)/e/1-6 * Document 1 2006/A(Ch)/e/7 * Document 2 2006/A(Ch)/e/8-10 2006/A(Ch)/e - 1 - LETTER
EUROPEAN QUALIFYING EXAMINATION 2006 PAPER A CHEMISTRY This paper comprises: * Letter from the applicant 2006/A(Ch)/e/1-6 * Document 1 2006/A(Ch)/e/7 * Document 2 2006/A(Ch)/e/8-10 2006/A(Ch)/e - 1 - LETTER
US A United States Patent (19) 11 Patent Number: 5,635,209 Groenewoud et al. 45 Date of Patent: Jun. 3, 1997
 US005635209A United States Patent (19) 11 Patent Number: Groenewoud et al. 45 Date of Patent: Jun. 3, 1997 (54) STABILIZED COMPOSITION OF LEVOTHYROXNE SODIUM MEDICATION 4,585,652 4,818,531 4/1986 Miller
US005635209A United States Patent (19) 11 Patent Number: Groenewoud et al. 45 Date of Patent: Jun. 3, 1997 (54) STABILIZED COMPOSITION OF LEVOTHYROXNE SODIUM MEDICATION 4,585,652 4,818,531 4/1986 Miller
DEVELOPMENT OF NON SODIUM EFFERVESCENT TABLET OF PARACETAMOL USING ARGININE CARBONATE
 IJPSR (2013), Vol. 4, Issue 5 (Research Article) Received on 17 July, 2012; received in revised form, 23 February, 2013; accepted, 14 April, 2013 DEVELOPMENT OF NON SODIUM EFFERVESCENT TABLET OF PARACETAMOL
IJPSR (2013), Vol. 4, Issue 5 (Research Article) Received on 17 July, 2012; received in revised form, 23 February, 2013; accepted, 14 April, 2013 DEVELOPMENT OF NON SODIUM EFFERVESCENT TABLET OF PARACETAMOL
INTERNATIONAL ŒNOLOGICAL CODEX. BENTONITES Bentonita N SIN: 558 (Oeno 11/2003 modified Oeno )
 BENTONITES Bentonita N SIN: 558 (Oeno 11/2003 modified Oeno 441-2011) 1. OBJECT, ORIGIN AND FIELD OF APPLICATION Bentonites are hydrous aluminium silicates belonging to the montmorillonite group. The brute
BENTONITES Bentonita N SIN: 558 (Oeno 11/2003 modified Oeno 441-2011) 1. OBJECT, ORIGIN AND FIELD OF APPLICATION Bentonites are hydrous aluminium silicates belonging to the montmorillonite group. The brute
(12) Patent Application Publication (10) Pub. No.: US 2003/ A1
 US 2003.01.18647A1 (19) United States (12) Patent Application Publication (10) Pub. No.: US 2003/0118647 A1 Seth (43) Pub. Date: (54) EXTENDED RELEASE TABLET OF Publication Classification METFORMIN (51)
US 2003.01.18647A1 (19) United States (12) Patent Application Publication (10) Pub. No.: US 2003/0118647 A1 Seth (43) Pub. Date: (54) EXTENDED RELEASE TABLET OF Publication Classification METFORMIN (51)
15. Mixed fertilizers sources preparations- their compatibility advantages
 15. Mixed fertilizers sources preparations- their compatibility advantages Mixed fertilizers For over hundred years the mixed fertilizers are in use besides straight fertilizers. Many fertilizer mixtures
15. Mixed fertilizers sources preparations- their compatibility advantages Mixed fertilizers For over hundred years the mixed fertilizers are in use besides straight fertilizers. Many fertilizer mixtures
Formulation and Evaluation
 Chapter-5 Formulation and Evaluation 5.1 OBJECTIVE After successful taste masking and solubility enhancement of drugs in preliminary studies, by using Mannitol Solid Dispersion, next step includes the
Chapter-5 Formulation and Evaluation 5.1 OBJECTIVE After successful taste masking and solubility enhancement of drugs in preliminary studies, by using Mannitol Solid Dispersion, next step includes the
UNITED STATES PATENT OFFICE
 Patented Aug. 18, 1942 2,293,676 UNITED STATES PATENT OFFICE 2,293,676 METHOD OF SEPARATING FATTY ACDS 8 Claims. This invention relates to a method of separat ing mixed higher fatty acids one from another.
Patented Aug. 18, 1942 2,293,676 UNITED STATES PATENT OFFICE 2,293,676 METHOD OF SEPARATING FATTY ACDS 8 Claims. This invention relates to a method of separat ing mixed higher fatty acids one from another.
Daher 45 Date of Patent: Sep. 20, U.S. C /66, 424/484 containing Sodium, potassium or mixed sodium potas
 I United States Patent (19) 11 USOO534-87A Patent Number: Daher Date of Patent: Sep., 1994 54 AQUEOUS GRANULATIONSOLUTION AND 4,704,269 11/1987 Korab... 424/44 A METHOD OF TABLET GRANULATION 4,716,046
I United States Patent (19) 11 USOO534-87A Patent Number: Daher Date of Patent: Sep., 1994 54 AQUEOUS GRANULATIONSOLUTION AND 4,704,269 11/1987 Korab... 424/44 A METHOD OF TABLET GRANULATION 4,716,046
High5 Effervescent Tablets Zero X treme 80g/20 tablets Berry + Caffeine
 720022 - High5 Effervescent Tablets Zero X treme 80g/20 tablets Berry + Caffeine Food supplement with sweeteners and caffeine/berry flavour. Acidity regulators (E330, E296), sodium carbonate, sweeteners
720022 - High5 Effervescent Tablets Zero X treme 80g/20 tablets Berry + Caffeine Food supplement with sweeteners and caffeine/berry flavour. Acidity regulators (E330, E296), sodium carbonate, sweeteners
High5 Effervescent Tablets Zero 160g/40 Berry + caffeine
 2139148 - High5 Effervescent Tablets Zero 160g/40 Berry + caffeine Food supplement with sweeteners and caffeine/berry flavour. Acidity regulators (E330, E296), sodium carbonate, sweeteners (E420, E955),
2139148 - High5 Effervescent Tablets Zero 160g/40 Berry + caffeine Food supplement with sweeteners and caffeine/berry flavour. Acidity regulators (E330, E296), sodium carbonate, sweeteners (E420, E955),
III. United States Patent (19) Carlsson et al. 11 Patent Number: 5, Date of Patent: Apr. 30, 1996
 United States Patent (19) Carlsson et al. 54) 75) 73) 21) 22) 63) 51) 52) (58 56 SMOKING SUBSTITUTE Inventors: Thommy Carlsson, Helsingborg; Sven B. Andersson, Odakra, both of Sweden Assignee: Pharmica
United States Patent (19) Carlsson et al. 54) 75) 73) 21) 22) 63) 51) 52) (58 56 SMOKING SUBSTITUTE Inventors: Thommy Carlsson, Helsingborg; Sven B. Andersson, Odakra, both of Sweden Assignee: Pharmica
United States Patent (19) Ashmead et al.
 United States Patent (19) Ashmead et al. 11 Patent Number: Date of Patent: 4,7,427 Feb. 16, 1988 54 EFFERVESCENT VITAMIN-MINERAL GRANULE PREPARATION 75) Inventors: H. DeWayne Ashmead, Fruit Heights; Robert
United States Patent (19) Ashmead et al. 11 Patent Number: Date of Patent: 4,7,427 Feb. 16, 1988 54 EFFERVESCENT VITAMIN-MINERAL GRANULE PREPARATION 75) Inventors: H. DeWayne Ashmead, Fruit Heights; Robert
FAO SPECIFICATIONS FAO PLANT PROTECTION PRODUCTS SULPHUR. FOOD AND AGRICULTURE ORGANIZATION OF THE UNITED NATIONS Rome, 1973
 AGP: CP/58 FAO SPECIFICATIONS FAO PLANT PROTECTION PRODUCTS SULPHUR FOOD AND AGRICULTURE ORGANIZATION OF THE UNITED NATIONS Rome, 1973 DISCLAIMER 1 FAO specifications are developed with the basic objective
AGP: CP/58 FAO SPECIFICATIONS FAO PLANT PROTECTION PRODUCTS SULPHUR FOOD AND AGRICULTURE ORGANIZATION OF THE UNITED NATIONS Rome, 1973 DISCLAIMER 1 FAO specifications are developed with the basic objective
Purity Tests for Modified Starches
 Residue Monograph prepared by the meeting of the Joint FAO/WHO Expert Committee on Food Additives (JECFA), 82 nd meeting 2016 Purity Tests for Modified Starches This monograph was also published in: Compendium
Residue Monograph prepared by the meeting of the Joint FAO/WHO Expert Committee on Food Additives (JECFA), 82 nd meeting 2016 Purity Tests for Modified Starches This monograph was also published in: Compendium
FAO SPECIFICATIONS FAO PLANT PROTECTION PRODUCTS. ZINEB Zinc ethylenebisdithiocarbamate
 AGP: CP/82 FAO SPECIFICATIONS FAO PLANT PROTECTION PRODUCTS ZINEB Zinc ethylenebisdithiocarbamate FOOD AND AGRICULTURE ORGANIZATION OF THE UNITED NATIONS Rome, 1979 DISCLAIMER 1 FAO specifications are
AGP: CP/82 FAO SPECIFICATIONS FAO PLANT PROTECTION PRODUCTS ZINEB Zinc ethylenebisdithiocarbamate FOOD AND AGRICULTURE ORGANIZATION OF THE UNITED NATIONS Rome, 1979 DISCLAIMER 1 FAO specifications are
LIQUID PREPARATIONS FOR ORAL USE. Final text for addition to The International Pharmacopoeia (November 2007)
 November 2007 LIQUID PREPARATIONS FOR ORAL USE Final text for addition to The International Pharmacopoeia (November 2007) This monograph was adopted at the Forty-second WHO Expert Committee on Specifications
November 2007 LIQUID PREPARATIONS FOR ORAL USE Final text for addition to The International Pharmacopoeia (November 2007) This monograph was adopted at the Forty-second WHO Expert Committee on Specifications
(12) United States Patent
 USOO9034382B2 (12) United States Patent Li et al. (54) OSELTAMIVIR PHOSPHATEGRANULE AND PREPARATION METHOD THEREOF (75) Inventors: Song Li, Beijing (CN); Wu Zhong, Beijing (CN); Junhai Xiao, Beijing (CN);
USOO9034382B2 (12) United States Patent Li et al. (54) OSELTAMIVIR PHOSPHATEGRANULE AND PREPARATION METHOD THEREOF (75) Inventors: Song Li, Beijing (CN); Wu Zhong, Beijing (CN); Junhai Xiao, Beijing (CN);
III. USOO A United States Patent (19) 11. Patent Number: 5,589,191 Ukigaya et al. 45) Date of Patent: Dec. 31, 1996
 III USOO5589191A United States Patent (19) 11. Patent Number: 5,589,191 Ukigaya et al. 45) Date of Patent: Dec. 31, 1996 54) SLOW-RELEASE SODIUM VALPROATE 5,019,398 5/1991 Daste... 424/480 TABLETS 5,055,306
III USOO5589191A United States Patent (19) 11. Patent Number: 5,589,191 Ukigaya et al. 45) Date of Patent: Dec. 31, 1996 54) SLOW-RELEASE SODIUM VALPROATE 5,019,398 5/1991 Daste... 424/480 TABLETS 5,055,306
INTERMEDIATE 1 1 Food and Diet. These elements are present in compounds - not as free elements.
 INTERMEDIATE 1 1 Food and Diet FOOD AND DIET The main elements present in the human body are: Hydrogen Oxygen Nitrogen Carbon These elements are present in compounds - not as free elements. Unlike plants,
INTERMEDIATE 1 1 Food and Diet FOOD AND DIET The main elements present in the human body are: Hydrogen Oxygen Nitrogen Carbon These elements are present in compounds - not as free elements. Unlike plants,
AGP: CP/89 FAO SPECIFICATIONS FOR PLANT PROTECTION PRODUCTS. PHENTHOATE S - a - ethoxycarbonylbenzyl 00-dimethyl phosphorodithioate
 AGP: CP/89 FAO SPECIFICATIONS FOR PLANT PROTECTION PRODUCTS PHENTHOATE S - a - ethoxycarbonylbenzyl 00-dimethyl phosphorodithioate FOOD AND AGRICULTURE ORGANIZATION OF THE UNITED NATIONS Rome, 1980 DISCLAIMER
AGP: CP/89 FAO SPECIFICATIONS FOR PLANT PROTECTION PRODUCTS PHENTHOATE S - a - ethoxycarbonylbenzyl 00-dimethyl phosphorodithioate FOOD AND AGRICULTURE ORGANIZATION OF THE UNITED NATIONS Rome, 1980 DISCLAIMER
(12) United States Patent (10) Patent No.: US 6,365,596 B1
 USOO63696B1 (12) United States Patent (10) Patent No.: US 6,365,596 B1 Valenti (45) Date of Patent: Apr. 2, 2002 (54) ORAL PHARMACEUTICAL COMPOSITIONS (51) Int. Cl."... A61K 31/44 CONTAINING BUPRENORPHIN
USOO63696B1 (12) United States Patent (10) Patent No.: US 6,365,596 B1 Valenti (45) Date of Patent: Apr. 2, 2002 (54) ORAL PHARMACEUTICAL COMPOSITIONS (51) Int. Cl."... A61K 31/44 CONTAINING BUPRENORPHIN
Plastcompactors Series HV
 Plastcompactors Series HV Plastcompactor HV 50 Agglomeration of powders, fibres, film and foam materials Drying of powders, fibres, film and foam materials Re-crystallisation of PET flakes Compounding
Plastcompactors Series HV Plastcompactor HV 50 Agglomeration of powders, fibres, film and foam materials Drying of powders, fibres, film and foam materials Re-crystallisation of PET flakes Compounding
(12) United States Patent
 USOO7022364B1 (12) United States Patent De Meuter et al. (10) Patent No.: (45) Date of Patent: Apr. 4, 2006 (54) SUGAR-FREE HARD COATINGS PREPARED FROM LIQUID MIXTURES OF ERYTHRTOL AND SORBITOL (75) Inventors:
USOO7022364B1 (12) United States Patent De Meuter et al. (10) Patent No.: (45) Date of Patent: Apr. 4, 2006 (54) SUGAR-FREE HARD COATINGS PREPARED FROM LIQUID MIXTURES OF ERYTHRTOL AND SORBITOL (75) Inventors:
LECTURE 10. PACKAGING TECHNOLOGY
 LECTURE 10. PACKAGING TECHNOLOGY The increasing demand for fresh and quality packaged food, consumer convenience and manufacturers concern for longer shelf life of the food products is driving the market
LECTURE 10. PACKAGING TECHNOLOGY The increasing demand for fresh and quality packaged food, consumer convenience and manufacturers concern for longer shelf life of the food products is driving the market
Compliance. Should you have any questions, please contact Behnaz Almasi, Associate Scientific Liaison ( or
 Extended-Release Tablets Type of Posting Revision Bulletin Posting Date 30 Mar 2018 Official Date 01 Apr 2018 Expert Committee Chemical Medicines Monographs 3 Reason for Revision Compliance In accordance
Extended-Release Tablets Type of Posting Revision Bulletin Posting Date 30 Mar 2018 Official Date 01 Apr 2018 Expert Committee Chemical Medicines Monographs 3 Reason for Revision Compliance In accordance
(51) Int Cl.: A61K 9/20 ( ) A61K 31/41 ( )
 (19) TEPZZ 94677_A_T (11) (12) EUROPEAN PATENT APPLICATION (43) Date of publication: 2.11.1 Bulletin 1/48 (1) Int Cl.: A61K 9/ (06.01) A61K 31/41 (06.01) (21) Application number: 116790.3 (22) Date of
(19) TEPZZ 94677_A_T (11) (12) EUROPEAN PATENT APPLICATION (43) Date of publication: 2.11.1 Bulletin 1/48 (1) Int Cl.: A61K 9/ (06.01) A61K 31/41 (06.01) (21) Application number: 116790.3 (22) Date of
E55A GELATIN, GELLING GRADE Gelatina
 00-0PDG.pdf 0 0 0 0 EA GELATIN, GELLING GRADE Gelatina DEFINITION Purified protein obtained from collagen of animals (including fish and poultry) by partial alkaline and/or acid hydrolysis, by enzymatic
00-0PDG.pdf 0 0 0 0 EA GELATIN, GELLING GRADE Gelatina DEFINITION Purified protein obtained from collagen of animals (including fish and poultry) by partial alkaline and/or acid hydrolysis, by enzymatic
Most of the ethanol that is used as a biofuel in this country is produced from corn.
 Chem 251 Ethanol from Corn Most of the ethanol that is used as a biofuel in this country is produced from corn. In this experiment you will make ethanol from frozen corn kernels using a process similar
Chem 251 Ethanol from Corn Most of the ethanol that is used as a biofuel in this country is produced from corn. In this experiment you will make ethanol from frozen corn kernels using a process similar
6.02 Uniformity of Dosage Units
 6.02 Uniformity of Dosage Units Change 1. Content Uniformity, 3. Criteria and Table 6.02-2 as follows: 1. Content Uniformity Select not less than 30 units, and proceed as follows for the dosage form designated.
6.02 Uniformity of Dosage Units Change 1. Content Uniformity, 3. Criteria and Table 6.02-2 as follows: 1. Content Uniformity Select not less than 30 units, and proceed as follows for the dosage form designated.
Contract Manufacturing Fluid Bed Technology. Drying. Spray granulation. Coating. Agglomeration. Instantization.
 Contract Manufacturing Fluid Bed Technology Drying. Spray granulation. Coating. Agglomeration. Instantization. Multifunctional fluid bed processor State-of-the-art fluid bed technology for a diversity
Contract Manufacturing Fluid Bed Technology Drying. Spray granulation. Coating. Agglomeration. Instantization. Multifunctional fluid bed processor State-of-the-art fluid bed technology for a diversity
ASHXX ASH (Residue on Ignition)
 ASHXX.01-1 ASH (Residue on Ignition) PRINCIPLE SCOPE Corn, corn wet milling by-products, or other products made from corn contain small amounts of inorganic materials which may vary in concentration and
ASHXX.01-1 ASH (Residue on Ignition) PRINCIPLE SCOPE Corn, corn wet milling by-products, or other products made from corn contain small amounts of inorganic materials which may vary in concentration and
Petrolatum. Stage 4, Revision 1. Petrolatum is a purified semi solid mixture of hydrocarbons obtained from petroleum.
 1 001-1208PDG.pdf Petrolatum Stage 4, Revision 1 Definition Petrolatum is a purified semi solid mixture of hydrocarbons obtained from petroleum. It may contain a suitable antioxidant. Description and Solubility
1 001-1208PDG.pdf Petrolatum Stage 4, Revision 1 Definition Petrolatum is a purified semi solid mixture of hydrocarbons obtained from petroleum. It may contain a suitable antioxidant. Description and Solubility
Addendum to the 2016 FAO/WHO Manual on pesticide specifications: revised Tablet Specifications for DT, ST and WT
 Addendum to the 2016 FAO/WHO Manual on pesticide specifications: revised Tablet Specifications for DT, ST and WT Introductory Note: The revised model specifications for tablets (ST, WT, DT) have been jointly
Addendum to the 2016 FAO/WHO Manual on pesticide specifications: revised Tablet Specifications for DT, ST and WT Introductory Note: The revised model specifications for tablets (ST, WT, DT) have been jointly
Food supplement manufacture
 Food supplement manufacture Nick Bennett BSc. RNutr April 2018 1 Introduction There are many different product formats available Liquids, powders, tablets, capsules etc Many different types of machine
Food supplement manufacture Nick Bennett BSc. RNutr April 2018 1 Introduction There are many different product formats available Liquids, powders, tablets, capsules etc Many different types of machine
DRY SYRUPS SWAPNA.M. Ist semester DEPARTMENT OF PHARMACEUTICS UNIVERSITY COLLEGE OF PHARMACEUTICAL SCIENCES KAKATIYA UNIVERSITY, WARANGAL SEMINAR BY
 DRY SYRUPS SEMINAR BY SWAPNA.M M.PHARMACY Ist semester DEPARTMENT OF PHARMACEUTICS UNIVERSITY COLLEGE OF PHARMACEUTICAL SCIENCES KAKATIYA UNIVERSITY, WARANGAL CONTENTS DEFINITION CHARACTERISTICS OF SUSPENSIONS
DRY SYRUPS SEMINAR BY SWAPNA.M M.PHARMACY Ist semester DEPARTMENT OF PHARMACEUTICS UNIVERSITY COLLEGE OF PHARMACEUTICAL SCIENCES KAKATIYA UNIVERSITY, WARANGAL CONTENTS DEFINITION CHARACTERISTICS OF SUSPENSIONS
EXPERIMENT #5 Water of Hydration
 OBJECTIVES: EXPERIMENT #5 Water of Hydration Observe changes in color and form of a hydrate after heating strongly Accurately weigh reaction mixtures before and after heating Calculate the mass of water
OBJECTIVES: EXPERIMENT #5 Water of Hydration Observe changes in color and form of a hydrate after heating strongly Accurately weigh reaction mixtures before and after heating Calculate the mass of water
REVISION OF MONOGRAPH ON TABLETS. Tablets
 March 2011 REVISION OF MONOGRAPH ON TABLETS Final text for addition to The International Pharmacopoeia This monograph was adopted by the Forty-fourth WHO Expert Committee on Specifications for Pharmaceutical
March 2011 REVISION OF MONOGRAPH ON TABLETS Final text for addition to The International Pharmacopoeia This monograph was adopted by the Forty-fourth WHO Expert Committee on Specifications for Pharmaceutical
Frying. PRO Ch. 17 of Fellows
 Frying PRO Ch. 17 of Fellows a unit operation mainly to alter the eating quality of a food, to preserve thermal process & reduction in Aw at the surface or throughout the food. Shelf life of fried foods
Frying PRO Ch. 17 of Fellows a unit operation mainly to alter the eating quality of a food, to preserve thermal process & reduction in Aw at the surface or throughout the food. Shelf life of fried foods
Sticky Business - Chewing Gum
 Sticky Business - Chewing Gum With close to one million metric tonnes of chewing gum expected to be produced worldwide in 2010, it s clear that it hasn t lost its flavour with consumers and retailers.
Sticky Business - Chewing Gum With close to one million metric tonnes of chewing gum expected to be produced worldwide in 2010, it s clear that it hasn t lost its flavour with consumers and retailers.
(51) Int Cl.: A61K 9/00 ( ) A61K 31/198 ( ) A61K 47/26 ( )
 (19) TEPZZ 8Z6B_T (11) EP 2 8 06 B1 (12) EUROPEAN PATENT SPECIFICATION (4) Date of publication and mention of the grant of the patent: 08.06.16 Bulletin 16/23 (21) Application number: 1171684.2 (22) Date
(19) TEPZZ 8Z6B_T (11) EP 2 8 06 B1 (12) EUROPEAN PATENT SPECIFICATION (4) Date of publication and mention of the grant of the patent: 08.06.16 Bulletin 16/23 (21) Application number: 1171684.2 (22) Date
(12) Patent Application Publication (10) Pub. No.: US 2009/ A1
 (19) United States US 200901236O2A1 (12) Patent Application Publication (10) Pub. No.: US 2009/0123602 A1 YAN et al. (43) Pub. Date: (54) METHOD FOR MAKING (30) Foreign Application Priority Data LACTOSE-REMOVING
(19) United States US 200901236O2A1 (12) Patent Application Publication (10) Pub. No.: US 2009/0123602 A1 YAN et al. (43) Pub. Date: (54) METHOD FOR MAKING (30) Foreign Application Priority Data LACTOSE-REMOVING
METHOD OF TEST FOR DETERMINATION OF INSOLUBLE RESIDUE OF CARBONATE AGGREGATES
 Laboratory Testing Manual Date: 96 05 01 Page 1 of 6 METHOD OF TEST FOR DETERMINATION OF INSOLUBLE RESIDUE OF CARBONATE AGGREGATES 1. SCOPE 1.1 This method covers the determination of the percentage of
Laboratory Testing Manual Date: 96 05 01 Page 1 of 6 METHOD OF TEST FOR DETERMINATION OF INSOLUBLE RESIDUE OF CARBONATE AGGREGATES 1. SCOPE 1.1 This method covers the determination of the percentage of
TEPZZ _7584 A_T EP A1 (19) (11) EP A1 (12) EUROPEAN PATENT APPLICATION. (51) Int Cl.: A61K 9/00 ( ) A61K 31/439 (2006.
 (19) TEPZZ _784 A_T (11) EP 3 17 842 A1 (12) EUROPEAN PATENT APPLICATION (43) Date of publication: 07.06.17 Bulletin 17/23 (1) Int Cl.: A61K 9/00 (06.01) A61K 31/439 (06.01) (21) Application number: 1197874.9
(19) TEPZZ _784 A_T (11) EP 3 17 842 A1 (12) EUROPEAN PATENT APPLICATION (43) Date of publication: 07.06.17 Bulletin 17/23 (1) Int Cl.: A61K 9/00 (06.01) A61K 31/439 (06.01) (21) Application number: 1197874.9
Pectins. Residue Monograph prepared by the meeting of the Joint FAO/WHO Expert Committee on Food Additives (JECFA), 82 nd meeting 2016
 Residue Monograph prepared by the meeting of the Joint FAO/WHO Expert Committee on Food Additives (JECFA), 82 nd meeting 2016 Pectins This monograph was also published in: Compendium of Food Additive Specifications.
Residue Monograph prepared by the meeting of the Joint FAO/WHO Expert Committee on Food Additives (JECFA), 82 nd meeting 2016 Pectins This monograph was also published in: Compendium of Food Additive Specifications.
FOR PLANT PROTECTION PRODUCTS FOOD AND AGRICULTURE ORGANIZATION OF THE UNITED NATIONS
 FAO SPECIFICATIONS FOR PLANT PROTECTION PRODUCTS CARBOSULFAN (AGP:CP/315) FOOD AND AGRICULTURE ORGANIZATION OF THE UNITED NATIONS Rome, 1995 2 CONTENTS DISCLAIMER... 3 INTRODUCTION... 4 SUBMISSION OF DRAFT
FAO SPECIFICATIONS FOR PLANT PROTECTION PRODUCTS CARBOSULFAN (AGP:CP/315) FOOD AND AGRICULTURE ORGANIZATION OF THE UNITED NATIONS Rome, 1995 2 CONTENTS DISCLAIMER... 3 INTRODUCTION... 4 SUBMISSION OF DRAFT
Pharmaceutical Preparation For Internal Use
 Pharmaceutical Preparation For Internal Use 1. Solid Preparations (Tablet, Capsule, Pill) 2. Liquid Preparations (Aqua, Syrup, Elixir, Extract, Liquor, Emulsion, Mixture, Infusion, Decoction). 3. Powder
Pharmaceutical Preparation For Internal Use 1. Solid Preparations (Tablet, Capsule, Pill) 2. Liquid Preparations (Aqua, Syrup, Elixir, Extract, Liquor, Emulsion, Mixture, Infusion, Decoction). 3. Powder
By Authority Of THE UNITED STATES OF AMERICA Legally Binding Document
 By Authority Of THE UNITED STATES OF AMERICA Legally Binding Document By the Authority Vested By Part 5 of the United States Code 552(a) and Part 1 of the Code of Regulations 51 the attached document has
By Authority Of THE UNITED STATES OF AMERICA Legally Binding Document By the Authority Vested By Part 5 of the United States Code 552(a) and Part 1 of the Code of Regulations 51 the attached document has
STUDY OF THE DETERIORATION OF ASPIRIN IN THE PRESENCE OF VARIOUS EXCIPIENTS
 STUDY OF THE DETERIORATION OF ASPIRIN IN THE PRESENCE OF VARIOUS EXCIPIENTS D.L.D.A.N DAHANAYAKA, A. MUNISINGHE, D.T.U. ABEYTUNGA DEPARTMENT OF CHEMISTRY, UNIVERSITY OF COLOMBO Abstract Aspirin is a non
STUDY OF THE DETERIORATION OF ASPIRIN IN THE PRESENCE OF VARIOUS EXCIPIENTS D.L.D.A.N DAHANAYAKA, A. MUNISINGHE, D.T.U. ABEYTUNGA DEPARTMENT OF CHEMISTRY, UNIVERSITY OF COLOMBO Abstract Aspirin is a non
T 676 cm-97 SUGGESTED METHOD 1979 CLASSICAL METHOD 1985 REVISED TAPPI. Viscosity of starch and starch products. 1. Scope
 T 676 cm-97 SUGGESTED METHOD 1979 CLASSICAL METHOD 1985 REVISED 1997 1997 TAPPI The information and data contained in this document were prepared by a technical committee of the Association. The committee
T 676 cm-97 SUGGESTED METHOD 1979 CLASSICAL METHOD 1985 REVISED 1997 1997 TAPPI The information and data contained in this document were prepared by a technical committee of the Association. The committee
Laboratory Pesticide Formulations, Labels, and Safety
 1 Laboratory Pesticide Formulations, Labels, and Safety I. Pesticide Formulations Pesticides are rarely used in their pure form (technical grade). They are processed into a usable form for direct application.
1 Laboratory Pesticide Formulations, Labels, and Safety I. Pesticide Formulations Pesticides are rarely used in their pure form (technical grade). They are processed into a usable form for direct application.
United States Patent Smith, Jr.
 United States Patent Smith, Jr. 4 (72) 73) 22 (21) 2) 1 8 VANADYLOXALATE COMPOUNDS AND PROCESS FOR PRODUCING SAME Inventor: William Novis Smith, Jr., Exton, w Pa. 1934 Assignee: Foote Mineral Company,
United States Patent Smith, Jr. 4 (72) 73) 22 (21) 2) 1 8 VANADYLOXALATE COMPOUNDS AND PROCESS FOR PRODUCING SAME Inventor: William Novis Smith, Jr., Exton, w Pa. 1934 Assignee: Foote Mineral Company,
Residue Monograph prepared by the meeting of the Joint FAO/WHO Expert Committee on Food Additives (JECFA), 82 nd meeting 2016.
 Residue Monograph prepared by the meeting of the Joint FAO/WHO Expert Committee on Food Additives (JECFA), 82 nd meeting 2016 Aspartame This monograph was also published in: Compendium of Food Additive
Residue Monograph prepared by the meeting of the Joint FAO/WHO Expert Committee on Food Additives (JECFA), 82 nd meeting 2016 Aspartame This monograph was also published in: Compendium of Food Additive
LAB.2. Tablet Production Methods
 LAB.2 Tablet Production Methods Dry methods Direct compression Dry granulation Wet methods Wet granulation Regardless whether tablets are made by direct compression or granulation, the first step, milling
LAB.2 Tablet Production Methods Dry methods Direct compression Dry granulation Wet methods Wet granulation Regardless whether tablets are made by direct compression or granulation, the first step, milling
FAO SPECIFICATIONS FOR PLANT PROTECTION PRODUCTS (MERCURIAL SEED TREATMENTS) ETHOXYETHLYMERCURY CHLORIDE
 AGP : CP / 86 FAO SPECIFICATIONS FOR PLANT PROTECTION PRODUCTS (MERCURIAL SEED TREATMENTS) ETHOXYETHLYMERCURY CHLORIDE FOOD AND AGRICULTURE ORGANIZATION OF THE UNITED NATIONS Rome, 1971 1 TABLE OF CONTENTS
AGP : CP / 86 FAO SPECIFICATIONS FOR PLANT PROTECTION PRODUCTS (MERCURIAL SEED TREATMENTS) ETHOXYETHLYMERCURY CHLORIDE FOOD AND AGRICULTURE ORGANIZATION OF THE UNITED NATIONS Rome, 1971 1 TABLE OF CONTENTS
7.2.7 Analytical Techniques
 7.2.7 Analytical Techniques 55 minutes 59 marks Page 1 of 20 Q1. A company making sweets uses different colour additives in different countries. In some countries the company uses Allura Red to colour
7.2.7 Analytical Techniques 55 minutes 59 marks Page 1 of 20 Q1. A company making sweets uses different colour additives in different countries. In some countries the company uses Allura Red to colour
Revision Bulletin 28 Jul Aug 2017 Chemical Medicines Monographs 3
 Oxybutynin Chloride Extended-Release Tablets Type of Posting Posting Date Official Date Expert Committee Reason for Revision Revision Bulletin 28 Jul 2017 01 Aug 2017 Chemical Medicines Monographs 3 Compliance
Oxybutynin Chloride Extended-Release Tablets Type of Posting Posting Date Official Date Expert Committee Reason for Revision Revision Bulletin 28 Jul 2017 01 Aug 2017 Chemical Medicines Monographs 3 Compliance
United States Patent (19)
 United States Patent (19) Gazzaniga et al. 11 Patent Number: () Date of Patent: * May, 1989 54 PHARMACEUTICAL COMPOSITION WITH ANALGESIC ACTIVITY 75 Inventors: Annibale Gazzaniga, Rescaldina, Italy; Walter
United States Patent (19) Gazzaniga et al. 11 Patent Number: () Date of Patent: * May, 1989 54 PHARMACEUTICAL COMPOSITION WITH ANALGESIC ACTIVITY 75 Inventors: Annibale Gazzaniga, Rescaldina, Italy; Walter
GENERAL STANDARD FOR EDIBLE FUNGI AND FUNGUS PRODUCTS 1 CODEX STAN
 CODEX STAN 38 Page 1 of 11 1. SCOPE GENERAL STANDARD FOR EDIBLE FUNGI AND FUNGUS PRODUCTS 1 CODEX STAN 38-1981 This standard contains general requirements applicable to all edible fungi, whether fresh
CODEX STAN 38 Page 1 of 11 1. SCOPE GENERAL STANDARD FOR EDIBLE FUNGI AND FUNGUS PRODUCTS 1 CODEX STAN 38-1981 This standard contains general requirements applicable to all edible fungi, whether fresh
*EP A1* EP A1 (19) (11) EP A1. (12) EUROPEAN PATENT APPLICATION published in accordance with Art.
 (19) Europäisches Patentamt European Patent Office Office européen des brevets *EP00147089A1* (11) EP 1 47 089 A1 (12) EUROPEAN PATENT APPLICATION published in accordance with Art. 18(3) EPC (43) Date
(19) Europäisches Patentamt European Patent Office Office européen des brevets *EP00147089A1* (11) EP 1 47 089 A1 (12) EUROPEAN PATENT APPLICATION published in accordance with Art. 18(3) EPC (43) Date
FAO SPECIFICATIONS FAO PLANT PROTECTION PRODUCTS. BROMOPHOS 4-bromo-2,5-dichloropheny1 dimethy1 phosphorothioate
 AGP: CP/70 FAO SPECIFICATIONS FAO PLANT PROTECTION PRODUCTS BROMOPHOS 4-bromo-2,5-dichloropheny1 dimethy1 phosphorothioate FOOD AND AGRICULTURE ORGANIZATION OF THE UNITED NATIONS Rome, 1977 DISCLAIMER
AGP: CP/70 FAO SPECIFICATIONS FAO PLANT PROTECTION PRODUCTS BROMOPHOS 4-bromo-2,5-dichloropheny1 dimethy1 phosphorothioate FOOD AND AGRICULTURE ORGANIZATION OF THE UNITED NATIONS Rome, 1977 DISCLAIMER
CONTENTS PAGE. Please note: Preface Matrix system Selection of METOLOSE grades Specifications
 Hypromellose CONTENTS PAGE 2 Preface Matrix system Selection of METOLOSE grades Specifications Properties Powder Solution Application Related Patents 3 4-5 6 8 10 13 14 17 Please note: The information
Hypromellose CONTENTS PAGE 2 Preface Matrix system Selection of METOLOSE grades Specifications Properties Powder Solution Application Related Patents 3 4-5 6 8 10 13 14 17 Please note: The information
MATERIAL SAFETY DATA SHEET (MSDS) Information about stainless steel for Surgical/Dental/Orthodontic Instruments.
 MATERIAL SAFETY DATA SHEET (MSDS) THE INFORMATION BELOW IS BELIEVED TO BE ACCURATE AND REPRESENTS THE BEST INFORMATION CURRENTYLY AVAILABLE TO US. HOWEVER, WE MAKE NO WARRANTY OF MERCHANTABILITY OR ANY
MATERIAL SAFETY DATA SHEET (MSDS) THE INFORMATION BELOW IS BELIEVED TO BE ACCURATE AND REPRESENTS THE BEST INFORMATION CURRENTYLY AVAILABLE TO US. HOWEVER, WE MAKE NO WARRANTY OF MERCHANTABILITY OR ANY
Fluoxetine Hydrochloride 5.6 mg Oral Chewable Treats (Solid Suspension, 60 x 1 ml Chewable Treats)
 4/11/2015; Page 1 Note: Fluoxetine Hydrochloride 5.6 mg is equivalent to Fluoxetine 5 mg. SUGGESTED FORMULATION Ingredient Listing Qty. Unit NDC # Supplier Fluoxetine Hydrochloride, USP 0.336 g Glycerin
4/11/2015; Page 1 Note: Fluoxetine Hydrochloride 5.6 mg is equivalent to Fluoxetine 5 mg. SUGGESTED FORMULATION Ingredient Listing Qty. Unit NDC # Supplier Fluoxetine Hydrochloride, USP 0.336 g Glycerin
GB Translated English of Chinese Standard: GB NATIONAL STANDARD
 Translated English of Chinese Standard: GB5009.5-2016 www.chinesestandard.net Sales@ChineseStandard.net GB NATIONAL STANDARD OF THE PEOPLE S REPUBLIC OF CHINA GB 5009.5-2016 National food safety standard
Translated English of Chinese Standard: GB5009.5-2016 www.chinesestandard.net Sales@ChineseStandard.net GB NATIONAL STANDARD OF THE PEOPLE S REPUBLIC OF CHINA GB 5009.5-2016 National food safety standard
FAO SPECIFICATIONS FOR PLANT PROTECTION PRODUCTS ALACHLOR (AGP: CP/300) FOOD AND AGRICULTURE ORGANIZATION OF THE UNITED NATIONS.
 1 FAO SPECIFICATIONS FOR PLANT PROTECTION PRODUCTS ALACHLOR (AGP: CP/300) FOOD AND AGRICULTURE ORGANIZATION OF THE UNITED NATIONS Rome, 1992 2 DISCLAIMER 1 FAO specifications are developed with the basic
1 FAO SPECIFICATIONS FOR PLANT PROTECTION PRODUCTS ALACHLOR (AGP: CP/300) FOOD AND AGRICULTURE ORGANIZATION OF THE UNITED NATIONS Rome, 1992 2 DISCLAIMER 1 FAO specifications are developed with the basic
CORESTA Recommended Method No. 85
 Cooperation Centre for Scientific Research Relative to Tobacco Routine Analytical Chemistry Sub-Group CORESTA Recommended Method No. 85 TOBACCO - DETERMINATION OF THE CONTENT OF TOTAL ALKALOIDS AS NICOTINE
Cooperation Centre for Scientific Research Relative to Tobacco Routine Analytical Chemistry Sub-Group CORESTA Recommended Method No. 85 TOBACCO - DETERMINATION OF THE CONTENT OF TOTAL ALKALOIDS AS NICOTINE
Paper No.: 13 Paper Title: Food Additives Module 2. Functional Classification of Food Additives
 Paper No.: 13 Paper Title: Food Additives Module 2. Functional Classification of Food Additives 2.1 Introduction According to the Food Protection Committee of the Food and Nutrition Board, food additives
Paper No.: 13 Paper Title: Food Additives Module 2. Functional Classification of Food Additives 2.1 Introduction According to the Food Protection Committee of the Food and Nutrition Board, food additives
Dehydration and Oral Rehydration Solutions
 Health Professional Training Guide Up to 75% less sugar and 4x more electrolytes vs. the leading sports drinks Wide range of innovate formats and great tasting flavors Lasts for 30 days in the refrigerator
Health Professional Training Guide Up to 75% less sugar and 4x more electrolytes vs. the leading sports drinks Wide range of innovate formats and great tasting flavors Lasts for 30 days in the refrigerator
CAKING OF WHITE SUGAR AND HOW TO PREVENT IT
 CAKING OF WHITE SUGAR AND HOW TO PREVENT IT M MATHLOUTHI 1,2 and B ROGÉ 2 1 Association Andrew VanHook, Maison des Agriculteurs, 2 Rue Léon Patoux F-51664 Reims Cedex 2, France 2 Laboratoire de Chimie
CAKING OF WHITE SUGAR AND HOW TO PREVENT IT M MATHLOUTHI 1,2 and B ROGÉ 2 1 Association Andrew VanHook, Maison des Agriculteurs, 2 Rue Léon Patoux F-51664 Reims Cedex 2, France 2 Laboratoire de Chimie
Viscosity of starch and starch products (WITHDRAWAL of T 676 cm-08 No changes from Draft 01)
 NOTICE: This is a DRAFT of a TAPPI Standard in ballot. Although available for public viewing, it is still under TAPPI s copyright and may not be reproduced or distributed without permission of TAPPI. This
NOTICE: This is a DRAFT of a TAPPI Standard in ballot. Although available for public viewing, it is still under TAPPI s copyright and may not be reproduced or distributed without permission of TAPPI. This
ON TEA TANNIN ISOLATED FROM GREEN TEA.
 70 [Vol. 6 ON TEA TANNIN ISOLATED FROM GREEN TEA. By MICHIYO TSUJIMIIRA. (Received September 8th., 1930). The author(1) has recently isolated Tea catechin from green tea and pro posed the following formula
70 [Vol. 6 ON TEA TANNIN ISOLATED FROM GREEN TEA. By MICHIYO TSUJIMIIRA. (Received September 8th., 1930). The author(1) has recently isolated Tea catechin from green tea and pro posed the following formula
Inspirational chemistry 97. Index sheets. Rhubarb contains oxalic acid, which has the formula C H 3. O + 2 Mn CO 2
 Inspirational chemistry 97 Rates and rhubarb Index 4.1.3 3 sheets Rhubarb contains oxalic acid, which has the formula C 2 H 2 O 4 : O OH C C HO Figure 1 Structural formula of oxalic acid Oxalic acid reacts
Inspirational chemistry 97 Rates and rhubarb Index 4.1.3 3 sheets Rhubarb contains oxalic acid, which has the formula C 2 H 2 O 4 : O OH C C HO Figure 1 Structural formula of oxalic acid Oxalic acid reacts
TEST. Directions: Circle the letter indicating whether the following statements are either true ("T") or false ("F").
 TEST Directions: Circle the letter indicating whether the following statements are either true ("T") or false ("F"). T F 1. A closed glass jar of pickles is a closed system. T F 2. Cellular respiration
TEST Directions: Circle the letter indicating whether the following statements are either true ("T") or false ("F"). T F 1. A closed glass jar of pickles is a closed system. T F 2. Cellular respiration
ANIMALS OILS AND FATS CHAPTER 2 PREPARATION AND SAPONIFICATION OF SPERMACETI 1. PREPARATION OF SPERMACETI
 182 ANIMALS OILS AND FATS CHAPTER 2 PREPARATION AND SAPONIFICATION OF SPERMACETI 1. PREPARATION OF SPERMACETI 690. The spermaceti I examined was separated as follows from a yellow colored oil which commercial
182 ANIMALS OILS AND FATS CHAPTER 2 PREPARATION AND SAPONIFICATION OF SPERMACETI 1. PREPARATION OF SPERMACETI 690. The spermaceti I examined was separated as follows from a yellow colored oil which commercial
United States Patent (19) Vollmer et al.
 United States Patent (19) Vollmer et al. (54 EXTRACTION OF PROTEIN FROM EDIBLE BEEF BONES AND PRODUCT 75 Inventors: Arthur N. Vollmer; George E. Rainey, both of Canton, Ohio (73) Assignee: Sugardale Foods,
United States Patent (19) Vollmer et al. (54 EXTRACTION OF PROTEIN FROM EDIBLE BEEF BONES AND PRODUCT 75 Inventors: Arthur N. Vollmer; George E. Rainey, both of Canton, Ohio (73) Assignee: Sugardale Foods,
EXPERIMENT 4 DETERMINATION OF REDUCING SUGARS, TOTAL REDUCING SUGARS, SUCROSE AND STARCH
 Practical Manual Food Chemistry and Physiology EXPERIMENT 4 DETERMINATION OF REDUCING SUGARS, TOTAL REDUCING SUGARS, SUCROSE AND STARCH Structure 4.1 Introduction Objectives 4.2 Experiment 4a: Reducing
Practical Manual Food Chemistry and Physiology EXPERIMENT 4 DETERMINATION OF REDUCING SUGARS, TOTAL REDUCING SUGARS, SUCROSE AND STARCH Structure 4.1 Introduction Objectives 4.2 Experiment 4a: Reducing
(12) United States Patent (10) Patent No.: US 6,692,756 B2
 USOO6692756B2 (12) United States Patent (10) Patent No.: US 6,692,756 B2 Chou (45) Date of Patent: *Feb. 17, 2004 (54) ALOE VERA GLOVE AND 5,679.399 A 10/1997 Shlenker et al. MANUFACTURING METHOD 5,682,617.
USOO6692756B2 (12) United States Patent (10) Patent No.: US 6,692,756 B2 Chou (45) Date of Patent: *Feb. 17, 2004 (54) ALOE VERA GLOVE AND 5,679.399 A 10/1997 Shlenker et al. MANUFACTURING METHOD 5,682,617.
The professionals choice.
 The professionals choice. Soft water worth its weight in money. EN 973 type A EN 14805 type 1 Codex Alimentarius NF standard AXAL Pro Salt Tablets The many benefits of soft water Soft water is not only
The professionals choice. Soft water worth its weight in money. EN 973 type A EN 14805 type 1 Codex Alimentarius NF standard AXAL Pro Salt Tablets The many benefits of soft water Soft water is not only
Mechanical Calibration
 Mechanical Calibration Distance between the inside bottom of the vessel and the bottom of the basket or paddle is maintained at 25 + 2 mm during the test use a spacing gauge or ball Temperature control
Mechanical Calibration Distance between the inside bottom of the vessel and the bottom of the basket or paddle is maintained at 25 + 2 mm during the test use a spacing gauge or ball Temperature control
LubriTose Mannitol Michael Crowley, Director of R&D, Excipients
 LubriTose Mannitol Michael Crowley, Director of R&D, Excipients Introduction Michael Crowley Director of R&D Excipients 158 St. Highway 320 Norwich, NY 13815 PH 315-802-5970 Michael.Crowley@Kerry.com 2
LubriTose Mannitol Michael Crowley, Director of R&D, Excipients Introduction Michael Crowley Director of R&D Excipients 158 St. Highway 320 Norwich, NY 13815 PH 315-802-5970 Michael.Crowley@Kerry.com 2
PREMIXES FOR MEDICATED FEEDING STUFFS FOR VETERINARY USE. Praeadmixta ad alimenta medicata ad usum veterinarium. Effervescent powders
 Preparations for inhalation administered in or with water or another suitable liquid. They may also be swallowed directly. They are presented as single-dose or multidose preparations. Where applicable,
Preparations for inhalation administered in or with water or another suitable liquid. They may also be swallowed directly. They are presented as single-dose or multidose preparations. Where applicable,
BIOCHEMICAL STUDIES ON CARBOHYDRATES. XL. Preparation of Mucoitin* from Umbilical Cords.
 The Journal of Biochemistry, Vol. 28, No. 3. BIOCHEMICAL STUDIES ON CARBOHYDRATES. XL. Preparation of Mucoitin* from Umbilical Cords. MASAMI BY SUZUKI. (From the Medico-Chemical Institute, Hokkaido Imperial
The Journal of Biochemistry, Vol. 28, No. 3. BIOCHEMICAL STUDIES ON CARBOHYDRATES. XL. Preparation of Mucoitin* from Umbilical Cords. MASAMI BY SUZUKI. (From the Medico-Chemical Institute, Hokkaido Imperial
Available Online through Research Article
 ISSN: 0975-766X Available Online through Research Article www.ijptonline.com DESIGN AND EVALUATION OF GASTRORETENTIVE TABLETS FOR CONTROLLED DELIVERY OF NORFLOXOCIN Ganesh Kumar Gudas*, Subal Debnath,
ISSN: 0975-766X Available Online through Research Article www.ijptonline.com DESIGN AND EVALUATION OF GASTRORETENTIVE TABLETS FOR CONTROLLED DELIVERY OF NORFLOXOCIN Ganesh Kumar Gudas*, Subal Debnath,
PIL. ParaCet Flu 500 mg+250 mg/sachet Powder for oral solution paracetamol + ascorbic acid
 PIL ParaCet Flu 500 mg+250 mg/sachet Powder for oral solution paracetamol + ascorbic acid Please, carefully read this PIL before you start using the medicine. This medicine is available without prescription,
PIL ParaCet Flu 500 mg+250 mg/sachet Powder for oral solution paracetamol + ascorbic acid Please, carefully read this PIL before you start using the medicine. This medicine is available without prescription,
Microwave-assisted esterification of cassava starch
 Microwave-assisted esterification of cassava starch 1 Central Tuber Crops Research Institute,Kerala, India 2 Dept. of Chemistry, University of Kerala, India email:sreejyothi in@yahoo.com Introduction Cassava
Microwave-assisted esterification of cassava starch 1 Central Tuber Crops Research Institute,Kerala, India 2 Dept. of Chemistry, University of Kerala, India email:sreejyothi in@yahoo.com Introduction Cassava
(12) Patent Application Publication (10) Pub. No.: US 2006/ A1
 (19) United States US 20060105287A1 (12) Patent Application Publication (10) Pub. No.: Wilburn (43) Pub. Date: May 18, 2006 (54) DO-IT-YOURSELF ORTHODONTIC KIT AND Publication Classification METHOD (51)
(19) United States US 20060105287A1 (12) Patent Application Publication (10) Pub. No.: Wilburn (43) Pub. Date: May 18, 2006 (54) DO-IT-YOURSELF ORTHODONTIC KIT AND Publication Classification METHOD (51)
GRAPEVINE DISEASES. Formulation Information & Adjuvants. Online Guide To. Virginia Tech
 Online Guide To GRAPEVINE DISEASES Virginia Tech Formulation Information & Adjuvants Ashley L. Myers Grape Pathology Extension Specialist, Department of Plant Pathology, Physiology, and Weed Sciences,
Online Guide To GRAPEVINE DISEASES Virginia Tech Formulation Information & Adjuvants Ashley L. Myers Grape Pathology Extension Specialist, Department of Plant Pathology, Physiology, and Weed Sciences,
STABILITY STUDIES OF FORMULATED CONTROLLED RELEASE ACECLOFENAC TABLETS
 Int. J. Chem. Sci.: 8(1), 2010, 405-414 STABILITY STUDIES OF FORMULATED CONTROLLED RELEASE ACECLOFENAC TABLETS V. L. NARASAIAH, T. KARTHIK KUMAR, D. SRINIVAS, K. SOWMYA, P. L. PRAVALLIKA and Sk. Md. MOBEEN
Int. J. Chem. Sci.: 8(1), 2010, 405-414 STABILITY STUDIES OF FORMULATED CONTROLLED RELEASE ACECLOFENAC TABLETS V. L. NARASAIAH, T. KARTHIK KUMAR, D. SRINIVAS, K. SOWMYA, P. L. PRAVALLIKA and Sk. Md. MOBEEN
Global College of Pharmacy, Kahnpur Khui, Tehsil Anandpur Sahib, Distt.- Ropar, Punjab, India
 IJPSR (2012), Vol. 3, Issue 09 (Research Article) Received on 19 May, 2012; received in revised form 25 June, 2012; accepted 27 August, 2012 IN-VITRO EVALUATION OF TWO MARKETED BRANDS OF PARACETAMOL TABLETS
IJPSR (2012), Vol. 3, Issue 09 (Research Article) Received on 19 May, 2012; received in revised form 25 June, 2012; accepted 27 August, 2012 IN-VITRO EVALUATION OF TWO MARKETED BRANDS OF PARACETAMOL TABLETS
The Cannabis Workflow and the IMPORTANCE OF TEMPERATURE CONTROL
 WHITEPAPER The Cannabis Workflow and the IMPORTANCE OF TEMPERATURE CONTROL As more states legalize the recreational and medicinal use of cannabis in the United States and Canada, the number of processors
WHITEPAPER The Cannabis Workflow and the IMPORTANCE OF TEMPERATURE CONTROL As more states legalize the recreational and medicinal use of cannabis in the United States and Canada, the number of processors
PHARMACEUTICAL AID PREPARED BY B.KIRUTHIGA LECTURER DEPT OF PHARMACEUTICAL CHEMISTRY
 PHARMACEUTICAL AID PREPARED BY B.KIRUTHIGA LECTURER DEPT OF PHARMACEUTICAL CHEMISTRY Excipients are inactive ingredients used as carriers for the active ingredients in a pharmaceutical product. These may
PHARMACEUTICAL AID PREPARED BY B.KIRUTHIGA LECTURER DEPT OF PHARMACEUTICAL CHEMISTRY Excipients are inactive ingredients used as carriers for the active ingredients in a pharmaceutical product. These may
Name GLOSSARY. Carrier protein a molecule in the cell membrane that allows materials to pass through it
 2 GLOSSARY Active transport movement of matter into, or out of, a cell which requires energy from the cell; usually this involves movement against the concentration gradient Adenosine triphosphate (ATP)
2 GLOSSARY Active transport movement of matter into, or out of, a cell which requires energy from the cell; usually this involves movement against the concentration gradient Adenosine triphosphate (ATP)
UNDERSTANDING YOUR WATER PROFILE PRESENTED BY POULTRY PARTNERS AND AHPD
 UNDERSTANDING YOUR WATER PROFILE PRESENTED BY POULTRY PARTNERS AND AHPD WHY DOES IT MATTER? Water intake for commercial poultry breeds is 1.5-2x greater than feed intake Commercial birds drink more now
UNDERSTANDING YOUR WATER PROFILE PRESENTED BY POULTRY PARTNERS AND AHPD WHY DOES IT MATTER? Water intake for commercial poultry breeds is 1.5-2x greater than feed intake Commercial birds drink more now
Title Revision n date
 A. THIN LAYER CHROMATOGRAPHIC TECHNIQUE (TLC) 1. SCOPE The method describes the identification of hydrocortisone acetate, dexamethasone, betamethasone, betamethasone 17-valerate and triamcinolone acetonide
A. THIN LAYER CHROMATOGRAPHIC TECHNIQUE (TLC) 1. SCOPE The method describes the identification of hydrocortisone acetate, dexamethasone, betamethasone, betamethasone 17-valerate and triamcinolone acetonide
United States Patent Office 3,506,582
 United States Patent Office Patented Apr. 14, 19 DRAIN CLEANER COMPOSITON AND PROCESS Donald Paul Gertzman, Elkhart, Ind., assignor to Miles Laboratories, Inc., Elkhart, Ind., a corporation of Indiana
United States Patent Office Patented Apr. 14, 19 DRAIN CLEANER COMPOSITON AND PROCESS Donald Paul Gertzman, Elkhart, Ind., assignor to Miles Laboratories, Inc., Elkhart, Ind., a corporation of Indiana
ETHYLENE GLYCOL. Table 1.1 Physical properties of Ethylene glycol
 ETHYLENE GLYCOL Introduction [1]: Glycols are dihydric alcohols having an aliphatic carbon chain. They have the general chemical formula C n H 2n (OH) 2. is the simplest and the most important of the glycols.
ETHYLENE GLYCOL Introduction [1]: Glycols are dihydric alcohols having an aliphatic carbon chain. They have the general chemical formula C n H 2n (OH) 2. is the simplest and the most important of the glycols.
Ingredient Listing Qty. Unit NDC # Supplier g. Sterile Preparation
 Note: Glucosamine Sulfate Potassium Chloride 1267 mg is equivalent to Glucosamine 750 mg. 11/8/2016; Page 1 SUGGESTED FORMULATION Ingredient Listing Qty. Unit NDC # Supplier Chondroitin Sulfate Sodium,
Note: Glucosamine Sulfate Potassium Chloride 1267 mg is equivalent to Glucosamine 750 mg. 11/8/2016; Page 1 SUGGESTED FORMULATION Ingredient Listing Qty. Unit NDC # Supplier Chondroitin Sulfate Sodium,
Nitrate and Nitrite Key Words: 1. Introduction 1.1. Nature, Mechanism of Action, and Biological Effects (Fig. 1)
 7 Nitrate and Nitrite Key Words: Nitrate; nitrite; methemoglobin; blood pressure; asphyxia; spinach; spongy cadmium column; zinc metal; sodium nitrate; sodium nitrite; ammonia buffer solution; Jones reductor.
7 Nitrate and Nitrite Key Words: Nitrate; nitrite; methemoglobin; blood pressure; asphyxia; spinach; spongy cadmium column; zinc metal; sodium nitrate; sodium nitrite; ammonia buffer solution; Jones reductor.
(12) Patent Application Publication (10) Pub. No.: US 2016/ A1
 US 2016O1 OO694A1 (19) United States (12) Patent Application Publication (10) Pub. No.: US 2016/0100694 A1 PELLETER (43) Pub. Date: Apr. 14, 2016 (54) KNEE PILLOW Publication Classification (71) Applicant:
US 2016O1 OO694A1 (19) United States (12) Patent Application Publication (10) Pub. No.: US 2016/0100694 A1 PELLETER (43) Pub. Date: Apr. 14, 2016 (54) KNEE PILLOW Publication Classification (71) Applicant:
