Treatment of Mild to Moderate Psoriasis with Reliéva, a Mahonia aquifolium Extract A Double-Blind, Placebo-Controlled Study
|
|
|
- Shana Singleton
- 5 years ago
- Views:
Transcription
1 American Journal of Therapeutics 13, (2006) Treatment of Mild to Moderate Psoriasis with Reliéva, a Mahonia aquifolium Extract A Double-Blind, Placebo-Controlled Study Steve Bernstein, Howard Donsky,* Wayne Gulliver, Douglas Hamilton, Sion Nobel, and Robert Norman Psoriasis is usually treated with local and systemic medications that have varying degrees of efficacy and safety profiles. We investigated the efficacy and safety of an alternative treatment from natural sources, Mahonia aquifolium, for the management of mild to moderate psoriasis. Two hundred subjects participated in a randomized, double-blind, placebo-controlled study using either the topical cream Reliéva (a homeopathic product containing a proprietary M. aquifolium extract) or control (placebo) twice a day for 12 weeks. Efficacy and safety were assessed using the Psoriasis Area Severity Index (PASI) and the Quality of Life Index (QLI) questionnaires at different times throughout the 12-week study. The PASI was evaluated by the physician at the beginning (week 0) and end (week 12) of the study. The QLI was assessed by patients at weeks 0, 4, 8, and 12. The results indicate statistically significant (P < 0.05) improvements in PASI and QLI in the Mahoniatreated group, compared with the control group. The side effects reported were infrequent, < 1% and minor; the most frequent side effects were rash, a burning sensation when applying the cream, and clothing stain. These data indicate that Reliéva, a proprietary form of M. aquifolium, is effective and well tolerated in patients with mild to moderate psoriasis. Keywords: psoriasis, Mahonia aquifolium, clinical trial, efficacy, safety INTRODUCTION Psoriasis is a common skin disorder that affects more than 4.5 million people in North America. 1 Treatment of psoriasis is problematic because the severity and distribution of psoriatic plaques varies immensely. 2 Localized psoriasis can often respond to topical medications, of which steroid and vitamin D analogue creams such as calcipotriol, anthralin, coal tar, and ultraviolet light treatments are the most common. More generalized involvement of the skin may require systemic treatments with retinoids, immunosuppressives, PUVA, or biologic medications. 3 Dermatology and Cosmetic Center, 1665 East Ridge Road, Rochester, New York. Supported in part by Apollo Pharmaceuticals Inc. *Address for correspondence: Dermatology and Cosmetic Center, 1665 East Ridge Road, Rochester, NY MDSkin- Care@aol.com M. aquifolium (Barberry, Oregon grape, Berberis) belongs to the berberidaceae family and grows wild in Europe, North and South America. M. aquifolium was initially used in American folk medicine as an oral medication for inflammatory skin diseases, including psoriasis and syphilis. 4 The root and wood of M. aquifolium contain many isoquinoline alkaloids, of which berberine is the best characterized. The mechanism of action of the alkaloids in suppressing the inflammatory response is poorly understood; however, laboratory studies have suggested that the alkaloids may 1 inhibit DNA synthesis by blocking reverse transcriptase, 2 inhibit lipoxygenase and lipid peroxidation, and/or 3 inhibit the cyclooxygenase-2 pathway through the reduction of prostaglandin E2. Topical application of M. aquifolium has been shown by immunohistochemistry to reduce the inflammatory and keratinocyte hyperproliferation markers typical of psoriasis. Several open and placebo-controlled clinical trials have supported the effectiveness of M. aquifolium in the treatment of psoriasis. 5 In these r 2006 Lippincott Williams & Wilkins
2 122 Bernstein et al trials, between 70% and 81% of patients improved on the basis of physician and patient assessment. Adverse events upon application of M. aquifolium ointment included burning and itching sensations. The purpose of this 12-week randomized, doubleblind, placebo-controlled clinical study was to assess the efficacy and safety of the topical cream, M. aquifolium (Reliéva), in patients with mild to moderate plaque psoriasis. We hypothesized that patients with mild to moderate psoriasis who were treated with M. aquifolium topical cream would achieve a greater improvement in the signs and symptoms of psoriasis compared with the control (placebo) group applying the vehicle for the same period of time. The results indicate a beneficial response in those patients using M. aquifolium topical cream for the treatment of psoriasis. MATERIALS AND METHODS Study design A randomized, placebo-controlled, double-blind, clinical trial of Reliéva (containing a proprietary M. aquifolium extract known as Psorberine) was conducted at 6 sites in the United States and Canada between August 2004 and February Patient eligibility was assessed through a telephone prescreening visit before entry into the study. During the initial visit (week 0), subjects were evaluated for eligibility, and informed consent and bill of rights forms were obtained. In addition, all subjects completed a screening health questionnaire and a brief physical examination. Qualified participants (n = 200) were randomized into 1 of 2 treatment groups: active (M. aquifolium, 10%; n = 100) or placebo (control excipients, n = 100). The active product group received active ingredients (M. aquifolium, 10%), and the placebo product group received the same formulation without active ingredient. Subjects in both groups applied the product twice a day to the selected area. The selected area was a 4.0 cm 4.0 cm area of skin that typified the patient s psoriasis. This area was selected by the investigator, using a precut template. Study preparations The active group was treated with a study preparation that contained Reliéva (IGI, Inc, NJ; Canadian Custom Packaging, Canada), a homeopathic product. Its active ingredient is a highly concentrated, proprietary extract of M. aquifolium 10% known as Psorberine, in Novasome, a patented liposome preparation from IGI, Inc, and formulated in an emulsion cream base for topical administration. Matching placebo preparations were prepared with excipients but no active components (IGI, Inc, NJ; Canadian Custom Packaging, Canada). Study protocol The study protocol required 4 patient visits, including the initial visit. At the initial visit (week 0), a Psoriasis Area Severity Index (PASI) evaluation was performed by the physician and each patient completed the Quality of Life Index (QLI) Questionnaire. Participants then returned at 4, 8, and 12 weeks after the start of treatment. At each visit, patients completed the QLI. The physician conducted a second PASI evaluation at week 12. Study medications were provided at the time of visits to the study site, and compliance was monitored via telephone calls. Assistance was provided as needed. PASI The PASI evaluation is used to indicate the severity of psoriasis and was selected as the primary efficacy end point. A higher score indicates more active disease. 6 PASI was evaluated using a 4.0 cm 4.0 cm template on a selected area of skin that was typical of the patient s psoriasis involvement. The selected area was evaluated on the basis of the amount of skin in the template affected (0% to 100%). Erythema (redness), infiltration (thickness), and desquamation (scaliness) were assessed as none, minimal, mild, moderate, or severe and translated into a number (0 to 4) for each time point. The PASI score was calculated using the following equation: PASI = % affected (erythema + infiltration + desquamation). Maximum PASI = 100% ( ) = 12. QLI questionnaire The QLI questionnaire was selected as a secondary end point and used to assess the patient s quality of life and adverse events. It included 12 questions that reflect impairment in the quality of life of the patient during the previous month. Each question was quantified from not at all to very much using a number from 0 to 10. The maximum possible score of 120 is correlated with very active disease, and the minimum score of zero indicated no quality of life impact. The change in QLI was calculated at the end of study by subtracting the week 12 score from the QLI
3 Treatment of Psoriasis 123 score at baseline. A decreasing score indicated a positive treatment benefit. Study participants All male or female participants between the ages of 18 to 80 years, in good overall health with current mild to moderate plaque psoriasis covering less than 10% to 15% of the body, were eligible for this study. Subjects were recruited from the clinical private practice setting, local college student population, local newspaper ads, and word of mouth. Exclusion criteria included painful or inflamed lesions, intertriginous psoriasis, extremely hypertrophic lesions, and severe psoriasis. Patients using topical psoriasis medications within the past 2 weeks, and those taking systemic (oral, intravenous, intramuscular, or intradermal) medications for psoriasis in the past 28 days, those using steroids, immunosuppressive medications, and cyclooxygenase-2 antiinflammatory drugs, and those using any medication conflicting with the product ingredients were also excluded from this study. Women planning to become pregnant within 90 days of the start of the study and pregnant or lactating women or women not taking medically approved birth control were also excluded. Statistical analysis Mean and standard deviations (SD), median, and minimum and maximum changes (min and max) were reported for continuous variables. Frequencies and percentages were reported for categorical variables. Independent sample t tests were used to compare differences between groups for continuous variables, and Fisher exact tests were used to test between group differences for categorical variables. The difference in PASI and QLI scores, pretreatment and posttreatment, was performed by subtracting the score determined posttreatment from the score at baseline. The change in score was analyzed using the Wilcoxon rank sum test. The accepted level of significance for all tests was a = All analyses were performed using SAS (SAS Institute, Cary, NC). A per protocol analysis and an intent-to-treat analysis were performed. RESULTS Patient demographics and baseline disease characteristics The 200 patients (100 active patients, 100 control patients) who entered the study constituted the intent-to-treat population. Of these, 171 patients (97 active group, 74 control group) completed the study, had no significant protocol deviation, and constituted an evaluable population used for the per-protocol analysis. Intent-to-treat population The demographics and patient characteristics (age, sex, PASI, QLI) of the intent-to-treat population indicate no differences among the 2 treatment groups (Table 1). Mean PASI scores (6.93 ± 2.6 in the active group; 6.85 ± 2.9 in the control group) and QLI scores (60.9 ± 32 in the active group; 56.6 ± 31 in the control group) were comparable among the 2 treatment groups. Eighty-one patients (41%) had a PASI score between 0 and 6, and 119 (59%) had a PASI > 6. Discontinuation of treatment The intent-to-treat population that entered the study consisted of 100 patients in the active group and 100 Table 1. Patient demographics and baseline disease characteristics. Active (N = 100) Placebo (N = 100) *P Mean age, years (SD) 48.3 ( ± 13.7) 48.3 ( ± 14.0) 0.49 w Sex Males, N (%) 51 (51%) 42 (42%) 0.26 z Females, N (%) 49 (49%) 58 (58%) Psoriasis area severity index at baseline (mean ± ) 6.93 ( ± 2.6) 6.85 ( ± 2.9) > 0.1 y Quality of life index at baseline (mean ± ) 60.9 ( ± 32) ( ± 31) > 0.1 y w Independent samples t test P value. z Fisher exact test P value. y Wilcoxon rank sum P value. 8 N = 99; 1 patient in the active treatment group did not have baseline scores available for analysis and was excluded from all analyses.
4 124 Bernstein et al Table 2. PASI scores intent-to-treat population.* N Active N Placebo P w PASI Baseline ( ± 2.6) ( ± 2.9) Average z (3.59 SD) (4.85 SD) Median z ( 11/3 min/max) ( 12/12 min/max) *Using all randomized patients; 3 in the active and 26 in the placebo group received worst-case scores. w Wilcoxon rank sum P value. z Reduction from baseline at week 12. Table 3. QLI intent-to-treat population.* N Active N Placebo P w QLI Baseline > 0.1 Average z (31.3 SD) (41.71 SD) Median z ( 98/98 min/max) ( 113/81 min/max) *Using all randomized patients; 1 patient in the active treatment group did not have baseline scores available for analysis and was excluded from all analyses. w Wilcoxon rank sum P value. z Reduction from baseline. patients in the control group. The most frequent causes of discontinuation were no response, which occurred in 2 patients in the active group and 11 in the control group; product caused burning, which occurred in 1 patient in the control group; rash developed in 1 patient in the control group. Two patients in the control group moved out of state and 11 patients in the control group discontinued therapy because of noncompliance. One subject in the active group died for non-treatment related reasons. Efficacy analysis Intent-to-treat population An intent-to-treat analysis was performed on all patients who entered the study. For PASI, a score of 12 (worst case score) was imputed at the 12-week visit for all patients who dropped from the study. Of the 100 patients in each group who began the study, 3 patients in the active group and 26 patients in the placebo group received worst-case scores. In this analysis, a statistically (P = ) significant improvement from baseline was observed between the active and control groups for PASI (Table 2). A similar analysis was performed on the quality of life assessments (QLI) measured at baseline and at the end of study (week 12). One patient in the active treatment group did not have baseline scores and was excluded. The average QLI change score in the active group was 25.5 (SD = 28.8) and 15.1 in the placebo group (SD = 22.5). Statistical analysis of the data indicates a statistically (P = ) significant improvement from baseline for the active group compared with the control group for QLI. This indicates that the active group and the placebo group were not similar with respect to the change in QLI from the beginning to the end of the study (Table 3). Taken together, the PASI and QLI data clearly indicate a greater clinical improvement in patients receiving Reliéva compared with patients receiving placebo. Evaluable population (per-protocol analysis) Ninety-seven active patients and 74 placebo patients were included in the per-protocol analysis. The average PASI change in the active group was 3.58 (SD = 3.47, median = 3, and min = 11, max = 3) and 2.22 in the placebo group (SD = 3.25, median = 1.6, and min = 11.9, max = 4). Testing the median PASI change between active and placebo patients gives a significant P value of using a 2-sided Wilcoxon rank sum test, indicating that the active group and the placebo group are not similar with respect to the outcome. A similar analysis was performed on the quality of life assessments (QLI) measured at baseline and at the end of study (week 12). The average QLI change score in the active group was 25.5 (SD = 28.76, median = 21, and min = 41, max = 98) and 15.1 in the placebo group (SD = 22.45, median = 12.5, and min = 31,
5 Treatment of Psoriasis 125 max = 81). Testing the median change scores between active and placebo patients gives a significant P value of using a 2-sided Wilcoxon rank sum test, indicating that the active group and the placebo group are not similar with respect to the change in QLI score from baseline to end of study. Side effects No significant side effects were reported by either group. One patient in the active group complained of staining from the cream. In the control (placebo) group, 1 patient reported a rash and 2 others reported a burning sensation when applying the cream. DISCUSSION Psoriasis is a difficult disorder to treat because of the severity and distribution of psoriatic plaques varying greatly, and some of the side effects of the current medications. The present study was conducted to determine if the use of Reliéva, a proprietary formulation of M. aquifolium, a natural preparation from a plant source, could provide effective treatment for psoriasis without serious side effects. The study found that patients with mild to moderate psoriasis who were treated with Reliéva topical cream achieved a significantly greater improvement in the signs and symptoms of psoriasis compared with the control group. One reason for the effectiveness of Reliéva may be found from the study by Augustin. 7 This author used immunohistochemistry to stain psoriatic plaques treated with either topical application of M. aquifolium or conventional treatment (dithranol). The study demonstrated marked reduction in the inflammatory and keratinocyte hyperproliferation markers typically seen in psoriasis. This author 7 reported that both treatments were effective, although greater reductions were seen in dithranol-treated plaques. Similar reports of the clinical effectiveness and safety of M. aquifolium for mild to severe psoriasis are found in the literature in placebo-controlled, open clinical trials and observational studies. 5,7 11 All of these studies reported improvement in psoriatic plaques treated with M. aquifolium cream. A recent review of all of these studies was conducted by Gulliver and Donsky. 5 Our study reports similar results from a doubleblind, placebo-controlled clinical trial. The efficacy of Reliéva was measured by changes in the PASI and QLI scores. For the PASI score, the Reliéva-treated active group showed a significantly greater reduction in the PASI score compared with the control (placebo) group. A similar greater reduction was reported for the QLI scores for the active compared with the control group. The significant improvements of the active group might also be reflected in the dropout rate of the study. More patients in the control (placebo) group dropped out of the study because of lack of response and noncompliance than the active treatment group (control group: 22 patients; active group: 3 patients). The tolerability of the active M. aquifolium (Reliéva) topical cream was excellent. The side effects reported were minor and reported in few patients. The most frequent side effects were rash, a burning sensation when applying the cream, and clothing stain. Clothing stains were removed with a simple wash. Similar side effects were reported by Weisenauer and Ludtke, 10 who evaluated the safety of M. aquifolium cream and concluded that M. aquifolium was a potent and safe therapy for moderately severe cases of psoriasis. A low side effect profile was also reported by others. 7,9 CONCLUSIONS This randomized, double-blind, placebo-controlled study demonstrated that Reliéva topical cream is a safe and effective treatment for mild to moderate plaque psoriasis. This was demonstrated by the statistically significant improvement of the signs and symptoms of mild to moderate plaque psoriasis compared with patients receiving placebo. Reliéva is well tolerated when applied to the affected areas twice a day for 12 weeks. ACKNOWLEDGMENTS The authors thank Global Clinicals, Inc ( Clinicals.com) and its staff for performing this study, Missy Magill for the statistical evaluation, and Andrea Gwosdow (PhD) for preparing this manuscript. REFERENCES 1. MedlinePlus: What is Psoriasis? Available at: Accessed September 21, Gotlieb AB. Psoriasis: emerging therapeutic strategies. Nat Rev/Drug Dis. 2005;4: Greaves MW, Weinstein GD. Treatment of psoriasisreview. New Engl J Med. 2005;332:
6 126 Bernstein et al 4. Duke JA. Medicinal plants. Science. 1985;229: Gulliver WP, Donsky HJ. A report on three recent clinical trials utilizing Mahonia aquifolium 10% topical cream and a review of the worldwide clinical experience with Mahonia aquifolium for the treatment of plaque psoriasis. Am J Ther In press. 6. Frederikkson T, Pettersson U. Severe psoriasis. A questionnaire survey of 2144 patients. Dermatology. 1978;157: Augustin M. Mahonia aquifolium in the treatment of psoriasis, a comparative study with dithranol. Z Phytother. 1996;17: Weisenauer M. Mahonia aquifolium-salbe bei psoriasis vulgaris. Z Allg Med. 1992;16: Gieler U, Von der Weth A, Heger M. Mahonia aquifoliuma new type of topical treatment for psoriasis. J Dermatol Treat (United Kingdom). 1995;6: Wiesenauer M, Ludtke R. Mahonia aquifolium patients with psoriasis vulgaris an intraindividual study. Phytomedicine. 1996;3: Augustin M, Andrees U, Grimme H, et al. Effects of Mahonia aquifolium ointment on the expression of adhesion, proliferation, and activation markers in the skin of patients with psoriasis. Forsch Komplementarmed. 1999;6(Suppl 2):19 21.
INTRODUCTION. Wayne P. Gulliver 1 * and Howard J. Donsky 2
 American Journal of Therapeutics 12, 398 406 (2005) A Report on Three Recent Clinical Trials Using Mahonia aquifolium 10% Topical Cream and a Review of the Worldwide Clinical Experience With Mahonia aquifolium
American Journal of Therapeutics 12, 398 406 (2005) A Report on Three Recent Clinical Trials Using Mahonia aquifolium 10% Topical Cream and a Review of the Worldwide Clinical Experience With Mahonia aquifolium
D E R M A T O L O G Y
 We customize individual prescriptions for the specific needs of our patients. F E B R U A R Y 2 0 1 3 I N S I D E T H I S I S S U E : Psoriasis Vulgaris 2 Atopic Dermatitis 3 P R E S C R I P T I O N C
We customize individual prescriptions for the specific needs of our patients. F E B R U A R Y 2 0 1 3 I N S I D E T H I S I S S U E : Psoriasis Vulgaris 2 Atopic Dermatitis 3 P R E S C R I P T I O N C
This PDF is available for free download from a site hosted by Medknow Publications
 Net Study Comparison of clinical efficacy of topical tazarotene.1% cream with topical clobetasol propionate.5% cream in chronic plaque psoriasis: A double-blind, randomized, right-left comparison study
Net Study Comparison of clinical efficacy of topical tazarotene.1% cream with topical clobetasol propionate.5% cream in chronic plaque psoriasis: A double-blind, randomized, right-left comparison study
2 SYNOPSIS. Study code : MC 9308 FR.
 MC9308 FR Study 19 December 2000 Page 15 of142 2 SYNOPSIS Study code : MC 9308 FR. Title: A comparative study of calcipotriol ointment in combination with narrow-band UVB (TL-01) phototherapy and placebo
MC9308 FR Study 19 December 2000 Page 15 of142 2 SYNOPSIS Study code : MC 9308 FR. Title: A comparative study of calcipotriol ointment in combination with narrow-band UVB (TL-01) phototherapy and placebo
The role of the practice nurse in managing psoriasis in primary care
 The role of the practice nurse in managing psoriasis in primary care Item type Authors Publisher Journal Article Buckley, David Nursing in General Practice Nursing in general practice Downloaded 16-Sep-2016
The role of the practice nurse in managing psoriasis in primary care Item type Authors Publisher Journal Article Buckley, David Nursing in General Practice Nursing in general practice Downloaded 16-Sep-2016
Psoriasis is a lifelong condition, with onset
 THERAPEUTICS FOR THE CLINICIAN Clobetasol Propionate Lotion in the Treatment of Moderate to Severe Plaque-Type Psoriasis Jacques Decroix, MD; Henrik Pres, MD; Nicolaï Tsankov, MD; Michel Poncet, PhD; Stéphanie
THERAPEUTICS FOR THE CLINICIAN Clobetasol Propionate Lotion in the Treatment of Moderate to Severe Plaque-Type Psoriasis Jacques Decroix, MD; Henrik Pres, MD; Nicolaï Tsankov, MD; Michel Poncet, PhD; Stéphanie
Clinical Study Synopsis for Public Disclosure
 Clinical Study Synopsis for Public Disclosure These results are supplied for informational purposes only in the interest of scientific disclosure. The synopsis may include approved and non-approved uses,
Clinical Study Synopsis for Public Disclosure These results are supplied for informational purposes only in the interest of scientific disclosure. The synopsis may include approved and non-approved uses,
A Pilot Study. Name of investigational product:
 An Open Label, Multi Center Clinical Study to Evaluate the Efficacy and Safety of a New Topical Cosmeceutical in Relieving the Redness, Scaling and Flaking Associated with Severe Skin Conditions A Pilot
An Open Label, Multi Center Clinical Study to Evaluate the Efficacy and Safety of a New Topical Cosmeceutical in Relieving the Redness, Scaling and Flaking Associated with Severe Skin Conditions A Pilot
Evaluation of Imuspora Tablets and Ointment (Multi-Ingredient Herbal Formulation) In the Management of Psoriasis: An Open Trial
 Evaluation of Imuspora Tablets and Ointment (Multi-Ingredient Herbal Formulation) In the Management of Psoriasis: An Open Trial Background: D G Saple*, S Medhekar** & M Patil*** *Professor & Head, Department
Evaluation of Imuspora Tablets and Ointment (Multi-Ingredient Herbal Formulation) In the Management of Psoriasis: An Open Trial Background: D G Saple*, S Medhekar** & M Patil*** *Professor & Head, Department
Follow this and additional works at: Part of the Medicine and Health Sciences Commons
 Philadelphia College of Osteopathic Medicine DigitalCommons@PCOM PCOM Physician Assistant Studies Student Scholarship Student Dissertations, Theses and Papers 2012 Is High Dose Intramuscular Alefacept
Philadelphia College of Osteopathic Medicine DigitalCommons@PCOM PCOM Physician Assistant Studies Student Scholarship Student Dissertations, Theses and Papers 2012 Is High Dose Intramuscular Alefacept
50 microgram/g Calcipotriol and 500 microgram/g betamethasone (as dipropionate).
 DUPISOR Composition Gel 50 microgram/g Calcipotriol and 500 microgram/g betamethasone (as dipropionate). Action Calcipotriol is a non-steroidal antipsoriatic agent, derived from vitamin D. Calcipotriol
DUPISOR Composition Gel 50 microgram/g Calcipotriol and 500 microgram/g betamethasone (as dipropionate). Action Calcipotriol is a non-steroidal antipsoriatic agent, derived from vitamin D. Calcipotriol
Location of study report in Regulatory Dossier for authorities
 This document has been downloaded from www.leo-pharma.com subject to the terms of use state on the website. It contains data and results regarding approved and non-approved uses, formulations or treatment
This document has been downloaded from www.leo-pharma.com subject to the terms of use state on the website. It contains data and results regarding approved and non-approved uses, formulations or treatment
IMO 3100, an antagonist of Toll like receptor (TLR) 7 and TLR9, demonstrates clinical activity in psoriasis patients
 IMO 3100, an antagonist of Toll like receptor (TLR) 7 and TLR9, demonstrates clinical activity in psoriasis patients with 4 weeks of treatment in a Phase 2a trial A. B. Kimball 1, J. Krueger 2, T. Sullivan
IMO 3100, an antagonist of Toll like receptor (TLR) 7 and TLR9, demonstrates clinical activity in psoriasis patients with 4 weeks of treatment in a Phase 2a trial A. B. Kimball 1, J. Krueger 2, T. Sullivan
Clinical Trial Report Synopsis
 Clinical Trial Report Synopsis A phase 2a, proof of concept trial, testing twice daily application of LEO 124249 ointment 30 mg/g in the treatment of mild to moderate inverse psoriasis Design of trial:
Clinical Trial Report Synopsis A phase 2a, proof of concept trial, testing twice daily application of LEO 124249 ointment 30 mg/g in the treatment of mild to moderate inverse psoriasis Design of trial:
MC 590 ABSTRACT. PageS
 This docwnent has OOen dov,nloaded from 'W'W'\VJ eo-pharma.c-om subject to the terms of use state on the website. It contains data and results regarding approved and non-approved uses, formulations or
This docwnent has OOen dov,nloaded from 'W'W'\VJ eo-pharma.c-om subject to the terms of use state on the website. It contains data and results regarding approved and non-approved uses, formulations or
STUDY. 1% Pimecrolimus, 0.005% Calcipotriol, and 0.1% Betamethasone in the Treatment of Intertriginous Psoriasis
 STUDY 1% Pimecrolimus, 0.005% Calcipotriol, and 0.1% Betamethasone in the Treatment of Intertriginous Psoriasis A Double-blind, Randomized Controlled Study Alexander Kreuter, MD; Anna Sommer, MD; Julia
STUDY 1% Pimecrolimus, 0.005% Calcipotriol, and 0.1% Betamethasone in the Treatment of Intertriginous Psoriasis A Double-blind, Randomized Controlled Study Alexander Kreuter, MD; Anna Sommer, MD; Julia
Etanercept: a new option in paediatric plaque psoriasis
 : a new option in paediatric plaque psoriasis Steve Chaplin MSc, MRPharmS, Medical Writer, Dr David Atherton MA, MB, BChir, FRCP, Honorary Consultant in Paediatric Dermatology, Great Ormond Street Hospital
: a new option in paediatric plaque psoriasis Steve Chaplin MSc, MRPharmS, Medical Writer, Dr David Atherton MA, MB, BChir, FRCP, Honorary Consultant in Paediatric Dermatology, Great Ormond Street Hospital
Clinical Trial Report Synopsis. Patient insights following use of LEO aerosol foam and Daivobet gel in subjects with psoriasis vulgaris
 This document has been downloaded from W\vw.leo-pharma.com subject to the ten:n.s of use state on the website. It contains data and results regarding approved and non-approved uses, formulations or treatment
This document has been downloaded from W\vw.leo-pharma.com subject to the ten:n.s of use state on the website. It contains data and results regarding approved and non-approved uses, formulations or treatment
During the last 20 years, the number of topical
 THERAPEUTICS FOR THE CLINICIAN Cumulative Irritation Potential of Adapalene 0.1% Cream and Gel Compared With Tretinoin Microsphere 0.04% and 0.1% Jonathan S. Dosik, MD; Kenneth Homer, MS; Stéphanie Arsonnaud
THERAPEUTICS FOR THE CLINICIAN Cumulative Irritation Potential of Adapalene 0.1% Cream and Gel Compared With Tretinoin Microsphere 0.04% and 0.1% Jonathan S. Dosik, MD; Kenneth Homer, MS; Stéphanie Arsonnaud
It is estimated that about 26,000 new cases of
 Focus on CME at Dalhousie University Set On Soothing Psoriasis A. H. Murray, MD, FRCP(C) Presented at the 76th Annual Dalhousie Refresher Course It is estimated that about 26,000 new cases of psoriasis
Focus on CME at Dalhousie University Set On Soothing Psoriasis A. H. Murray, MD, FRCP(C) Presented at the 76th Annual Dalhousie Refresher Course It is estimated that about 26,000 new cases of psoriasis
Prurisol: A New Small Molecule under investigation for the treatment of Psoriasis
 Prurisol: A New Small Molecule under investigation for the treatment of Psoriasis Krishna Menon, RCM, PhD, VMD, President of Research and Chief Scientific Officer MEDICAL DERMATOLOGY THERAPEUTIC R&D AND
Prurisol: A New Small Molecule under investigation for the treatment of Psoriasis Krishna Menon, RCM, PhD, VMD, President of Research and Chief Scientific Officer MEDICAL DERMATOLOGY THERAPEUTIC R&D AND
Psoriasis: Causes, Symptoms, And Treatment
 Psoriasis: Causes, Symptoms, And Treatment We all know that a healthy immune system is good. But, do you know that an overactive immune system can cause certain conditions like Psoriasis? Read on to find
Psoriasis: Causes, Symptoms, And Treatment We all know that a healthy immune system is good. But, do you know that an overactive immune system can cause certain conditions like Psoriasis? Read on to find
Individual Study Table Referring to Part of the Dossier. Volume: Page:
 2 Synopsis Title of study: An Investigator-blind, Controlled Study to Assess the Efficacy and Safety of Different Formulations of Compared to Placebo and to Active Control in a Psoriasis Plaque Test Investigators
2 Synopsis Title of study: An Investigator-blind, Controlled Study to Assess the Efficacy and Safety of Different Formulations of Compared to Placebo and to Active Control in a Psoriasis Plaque Test Investigators
CLINICAL STUDY REPORT Efficacy of Psorioderm cream in treatment of mild to moderate plaque psoriasis which does not require biological therapy
 DEBRECENI EGYETEM ORVOS- ÉS EGÉSZSÉGTUDOMÁNYI CENTRUM Bőrgyógyászati Klinika Igazgató: Dr. Remenyik Éva egyetemi tanár UNIVERSITY OF DEBRECEN MEDICAL AND HEALTH SCIENCE CENTRE Department of Dermatology
DEBRECENI EGYETEM ORVOS- ÉS EGÉSZSÉGTUDOMÁNYI CENTRUM Bőrgyógyászati Klinika Igazgató: Dr. Remenyik Éva egyetemi tanár UNIVERSITY OF DEBRECEN MEDICAL AND HEALTH SCIENCE CENTRE Department of Dermatology
EFFICACY AND SAFETY OF CLOBETASOL PROPIONATE SHAMPOO IN THERAPY OF PSORIASIS OF THE SCALP
 WORLD JOURNAL OF PHARMACY AND PHARMACEUTICAL SCIENCES Platonova et al. SJIF Impact Factor 2.786 Volume 4, Issue 04, 238-246. Research Article ISSN 2278 4357 EFFICACY AND SAFETY OF CLOBETASOL PROPIONATE
WORLD JOURNAL OF PHARMACY AND PHARMACEUTICAL SCIENCES Platonova et al. SJIF Impact Factor 2.786 Volume 4, Issue 04, 238-246. Research Article ISSN 2278 4357 EFFICACY AND SAFETY OF CLOBETASOL PROPIONATE
Comparative Efficacy of Topical Calcipotriol (0.005%) Versus Topical Corticosteroid (Betamethasone 0.1%) in Treating Plaque Type Psoriasis
 Original Article Comparative Efficacy of Topical Calcipotriol (0.005%) Versus Topical Corticosteroid (Betamethasone 0.1%) in Treating Plaque Type Psoriasis Admed GKA 1, Khondker L 2, Nessa M 3, Alam MN
Original Article Comparative Efficacy of Topical Calcipotriol (0.005%) Versus Topical Corticosteroid (Betamethasone 0.1%) in Treating Plaque Type Psoriasis Admed GKA 1, Khondker L 2, Nessa M 3, Alam MN
KEY MESSAGES. Psoriasis patients are more prone to cardiovascular diseases, stroke, lymphoma and non-melanoma skin cancers, and increased mortality.
 KEY MESSAGES Psoriasis is a genetically determined, systemic immune-mediated chronic inflammatory disease that affects primarily the skin and joints. Psoriasis Vulgaris is characterised by well-demarcated
KEY MESSAGES Psoriasis is a genetically determined, systemic immune-mediated chronic inflammatory disease that affects primarily the skin and joints. Psoriasis Vulgaris is characterised by well-demarcated
Psoriasis. What is Psoriasis? What causes psoriasis? Medical Topics Psoriasis
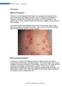 1 Psoriasis What is Psoriasis? Psoriasis is a long standing inflammatory non-contagious skin disease which waxes and wanes with triggering factors. There is a genetic predisposition in psoriasis. Internationally,
1 Psoriasis What is Psoriasis? Psoriasis is a long standing inflammatory non-contagious skin disease which waxes and wanes with triggering factors. There is a genetic predisposition in psoriasis. Internationally,
A systematic review of treatments for severe psoriasis Griffiths C E, Clark C M, Chalmers R J, Li Wan Po A, Williams H C
 A systematic review of treatments for severe psoriasis Griffiths C E, Clark C M, Chalmers R J, Li Wan Po A, Williams H C Authors' objectives To compare the effectiveness of currently available treatments
A systematic review of treatments for severe psoriasis Griffiths C E, Clark C M, Chalmers R J, Li Wan Po A, Williams H C Authors' objectives To compare the effectiveness of currently available treatments
Month/Year of Review: January 2015 Date of Last Review: January 2010
 Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35, Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119 Copyright 2012 Oregon State University. All Rights
Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35, Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119 Copyright 2012 Oregon State University. All Rights
Topical Calcipotriol Algorithm
 Topical Calcipotriol Algorithm Is this patient an adult previously diagnosed with psoriasis by a doctor? Do the skin patches look the same as those diagnosed as psoriasis? Is this psoriasis covering an
Topical Calcipotriol Algorithm Is this patient an adult previously diagnosed with psoriasis by a doctor? Do the skin patches look the same as those diagnosed as psoriasis? Is this psoriasis covering an
Patients who achieved the primary criterion for response i.e.: complete clearance or a reduction
 MC 9101 F Study Page3 ABSTRACT Background: Cyclosporin A has been shown to be an effective systemic treatment in severe psoriasis but with the disadvantage of dose-dependent toxic effects particularly
MC 9101 F Study Page3 ABSTRACT Background: Cyclosporin A has been shown to be an effective systemic treatment in severe psoriasis but with the disadvantage of dose-dependent toxic effects particularly
This PDF is available for free download from a site hosted by Medknow Publications
 Original Article Niosomal methotrexate gel in the treatment of localized psoriasis: Phase I and phase II studies P. K. Lakshmi, Gayathri S. Devi*, Shyamala Bhaskaran**, S. Sacchidanand*** Director-Drug
Original Article Niosomal methotrexate gel in the treatment of localized psoriasis: Phase I and phase II studies P. K. Lakshmi, Gayathri S. Devi*, Shyamala Bhaskaran**, S. Sacchidanand*** Director-Drug
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE. Proposed Health Technology Appraisal
 NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Proposed Health Technology Appraisal Dimethyl fumarate for treating moderate to severe Draft scope (pre-referral) Draft remit/appraisal objective To appraise
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Proposed Health Technology Appraisal Dimethyl fumarate for treating moderate to severe Draft scope (pre-referral) Draft remit/appraisal objective To appraise
Chemical structure of calcipotriol
 PRODUCT INFORMATION DAIVONEX CREAM AUST R 57354 Calcipotriol 50 microgram/g NAME OF THE MEDICINE: CALCIPOTRIOL DESCRIPTION Calcipotriol is a white or almost white crystalline substance. It is a vitamin
PRODUCT INFORMATION DAIVONEX CREAM AUST R 57354 Calcipotriol 50 microgram/g NAME OF THE MEDICINE: CALCIPOTRIOL DESCRIPTION Calcipotriol is a white or almost white crystalline substance. It is a vitamin
SKIN-CAP IN TREATMENT OF PATIENTS WITH PSORIASIS
 SKIN-CAP IN TREATMENT OF PATIENTS WITH PSORIASIS Mavlyanova Sh., Z., Abidova Z.M., Luk yanova A.S., Ismagilov A.I., Azimova F.A. Scientific-Research Institute of Dermatology and Venerology, Public Health
SKIN-CAP IN TREATMENT OF PATIENTS WITH PSORIASIS Mavlyanova Sh., Z., Abidova Z.M., Luk yanova A.S., Ismagilov A.I., Azimova F.A. Scientific-Research Institute of Dermatology and Venerology, Public Health
2.0 Synopsis. Adalimumab R&D/04/118. (For National Authority Use Only) Referring to Part of Dossier: Volume:
 2.0 Synopsis Abbott Laboratories Name of Study Drug: Adalimumab Name of Active Ingredient: Adalimumab Title of Study: Individual Study Table Referring to Part of Dossier: Volume: Page: (For National Authority
2.0 Synopsis Abbott Laboratories Name of Study Drug: Adalimumab Name of Active Ingredient: Adalimumab Title of Study: Individual Study Table Referring to Part of Dossier: Volume: Page: (For National Authority
Topical treatment of psoriasis in difficult to treat areas: Genital, folds and palmoplantar
 Topical treatment of psoriasis in difficult to treat areas: Genital, folds and palmoplantar Dr. Steven Feldman 1 Genital psoriasis: a questionnaire-based survey on a concealed skin disease in the Netherlands
Topical treatment of psoriasis in difficult to treat areas: Genital, folds and palmoplantar Dr. Steven Feldman 1 Genital psoriasis: a questionnaire-based survey on a concealed skin disease in the Netherlands
Novan Announces Promising Clinical Results with SB414
 Novan Announces Promising Clinical Results with SB414 In the recently completed Phase 1b trial for atopic dermatitis, clinical efficacy measures were highly correlated with critical and disease-relevant
Novan Announces Promising Clinical Results with SB414 In the recently completed Phase 1b trial for atopic dermatitis, clinical efficacy measures were highly correlated with critical and disease-relevant
Adalimumab M Clinical Study Report Final R&D/14/1263. Page:
 Synopsis AbbVie Inc. Name of Study Drug: Adalimumab Name of Active Ingredient: Adalimumab Individual Study Table Referring to Part of Dossier: Volume: Page: (For National Authority Use Only) Title of Study:
Synopsis AbbVie Inc. Name of Study Drug: Adalimumab Name of Active Ingredient: Adalimumab Individual Study Table Referring to Part of Dossier: Volume: Page: (For National Authority Use Only) Title of Study:
2.0 Synopsis. Adalimumab M Clinical Study Report R&D/04/900. (For National Authority Use Only) Referring to Part of Dossier: Volume:
 2. Synopsis Abbott Laboratories Name of Study Drug: Name of Active Ingredient: Title of Study: Individual Study Table Referring to Part of Dossier: Volume: Page: (For National Authority Use Only) Phase
2. Synopsis Abbott Laboratories Name of Study Drug: Name of Active Ingredient: Title of Study: Individual Study Table Referring to Part of Dossier: Volume: Page: (For National Authority Use Only) Phase
Type of intervention Treatment. Economic study type Cost-effectiveness analysis.
 Cost-effectiveness of once-daily treatment with calcipotriol/betamethasone dipropionate followed by calcipotriol alone compared with tacalcitol in the treatment of psoriasis vulgaris Peeters P, Ortonne
Cost-effectiveness of once-daily treatment with calcipotriol/betamethasone dipropionate followed by calcipotriol alone compared with tacalcitol in the treatment of psoriasis vulgaris Peeters P, Ortonne
2 Synopsis. Name of Sponsor/Company: Volume: Page: (For National Authority Use Only) Almirall Hermal GmbH. Name of Finished Product: LAS 41004
 2 Synopsis Title of study: An Investigator-blind, Controlled Exploratory Study to Assess the Efficacy and Safety of Different Concentrations of Active Ingredients in the project Formulations of Compared
2 Synopsis Title of study: An Investigator-blind, Controlled Exploratory Study to Assess the Efficacy and Safety of Different Concentrations of Active Ingredients in the project Formulations of Compared
To order reprints or e-prints of JDD articles please contact JDD
 June 2017 534 VOLUME 16 ISSUE 6 Copyright 2017 ORIGINAL ARTICLE Journal of Drugs in Dermatology The Efficacy and Safety of Azelaic Acid 15% Foam in the Treatment of Truncal Acne Vulgaris Lauren K. Hoffman
June 2017 534 VOLUME 16 ISSUE 6 Copyright 2017 ORIGINAL ARTICLE Journal of Drugs in Dermatology The Efficacy and Safety of Azelaic Acid 15% Foam in the Treatment of Truncal Acne Vulgaris Lauren K. Hoffman
P4081 Secukinumab skin clearance is associated with greater improvements in patient-reported pain, itching, and scaling
 P4081 Secukinumab skin clearance is associated with greater improvements in patient-reported pain, itching, and scaling Mark Lebwohl, 1 Andrew Blauvelt, 2 Matthias Augustin, 3 Yang Zhao, 4 Isabelle Gilloteau,
P4081 Secukinumab skin clearance is associated with greater improvements in patient-reported pain, itching, and scaling Mark Lebwohl, 1 Andrew Blauvelt, 2 Matthias Augustin, 3 Yang Zhao, 4 Isabelle Gilloteau,
Public Assessment Report Scientific discussion. Ovixan (mometasone furoate) SE/H/1088/01/DC
 Public Assessment Report Scientific discussion Ovixan (mometasone furoate) SE/H/1088/01/DC This module reflects the scientific discussion for the approval of Ovixan. The procedure was finalised at 2012-12-14.
Public Assessment Report Scientific discussion Ovixan (mometasone furoate) SE/H/1088/01/DC This module reflects the scientific discussion for the approval of Ovixan. The procedure was finalised at 2012-12-14.
Follow this and additional works at: Part of the Skin and Connective Tissue Diseases Commons
 Philadelphia College of Osteopathic Medicine DigitalCommons@PCOM PCOM Physician Assistant Studies Student Scholarship Student Dissertations, Theses and Papers 2014 Is the Addition of a Topical Agent to
Philadelphia College of Osteopathic Medicine DigitalCommons@PCOM PCOM Physician Assistant Studies Student Scholarship Student Dissertations, Theses and Papers 2014 Is the Addition of a Topical Agent to
Management of psoriasis with Aloe Vera extract in a hydrophilic cream: a placebo-controlled, double-blind study*
 Tropical Medicine and International Health VOLUME I NO. 4 PP 505-509 AUGUST 1996 Management of psoriasis with Aloe Vera extract in a hydrophilic cream: a placebo-controlled, double-blind study* Tanweer
Tropical Medicine and International Health VOLUME I NO. 4 PP 505-509 AUGUST 1996 Management of psoriasis with Aloe Vera extract in a hydrophilic cream: a placebo-controlled, double-blind study* Tanweer
Keywords: Psoriasis vulgaris Zinc pyrithione Betamethasone dipropionate
 CLINICAL EFFICACY AND SAFETY OF A COMBINED FORMULATION OF ZINC PYRITHIONE 0.25% AND BETAMETHASONE DIPROPIONATE MICRONIZED 0.05% IN THE TREATMENT OF MILD TO MODERATE PLAQUE PSORIASIS. Abstract Background
CLINICAL EFFICACY AND SAFETY OF A COMBINED FORMULATION OF ZINC PYRITHIONE 0.25% AND BETAMETHASONE DIPROPIONATE MICRONIZED 0.05% IN THE TREATMENT OF MILD TO MODERATE PLAQUE PSORIASIS. Abstract Background
Is Apremilast (Otezla) Effective in Reducing Pruritus in Adults over 18 Years Old with Plaque Psoriasis?
 Philadelphia College of Osteopathic Medicine DigitalCommons@PCOM PCOM Physician Assistant Studies Student Scholarship Student Dissertations, Theses and Papers 3-2017 Is Apremilast (Otezla) Effective in
Philadelphia College of Osteopathic Medicine DigitalCommons@PCOM PCOM Physician Assistant Studies Student Scholarship Student Dissertations, Theses and Papers 3-2017 Is Apremilast (Otezla) Effective in
Psoriasis, Incidence, Quality of Life, Psoriatic Arthritis, Prevalence
 1.0 Abstract Title Prevalence and Incidence of Articular Symptoms and Signs Related to Psoriatic Arthritis in Patients with Psoriasis Severe or Moderate with Adalimumab Treatment (TOGETHER). Keywords Psoriasis,
1.0 Abstract Title Prevalence and Incidence of Articular Symptoms and Signs Related to Psoriatic Arthritis in Patients with Psoriasis Severe or Moderate with Adalimumab Treatment (TOGETHER). Keywords Psoriasis,
Enstilar , Version 3 PUBLIC SUMMARY OF RISK MANAGEMENT PLAN
 Enstilar 21.1.2016, Version 3 PUBLIC SUMMARY OF RISK MANAGEMENT PLAN VI.2.1 Overview of disease epidemiology Psoriasis is a common skin disease where parts of the skin develop into thick, red and scaly
Enstilar 21.1.2016, Version 3 PUBLIC SUMMARY OF RISK MANAGEMENT PLAN VI.2.1 Overview of disease epidemiology Psoriasis is a common skin disease where parts of the skin develop into thick, red and scaly
COS DERM. Do Not Copy
 Study Use of a Bovine-Based Collagen Ointment in the Treatment of Plaque Psoriasis: An Open-Label, Pilot, Observational Clinical Study of 12 Patients Montserrat Pérez, MD, PhD; Marta Alegre Fernández,
Study Use of a Bovine-Based Collagen Ointment in the Treatment of Plaque Psoriasis: An Open-Label, Pilot, Observational Clinical Study of 12 Patients Montserrat Pérez, MD, PhD; Marta Alegre Fernández,
COMPARATIVE STUDY OF THE EFFICACY OF VARIOUS TOPICAL TREATMENT MODALITIES AND PHOTOTHERAPY FOR PSORIASIS VULGARIS: A REVIEW
 COMPARATIVE STUDY OF THE EFFICACY OF VARIOUS TOPICAL TREATMENT MODALITIES AND PHOTOTHERAPY FOR PSORIASIS VULGARIS: A REVIEW Dr. Shweta Aryal *, Prof. Dr. Liu Jin Xian, Dr. Gong Shao Zhi and Dr. Jyoti Karki
COMPARATIVE STUDY OF THE EFFICACY OF VARIOUS TOPICAL TREATMENT MODALITIES AND PHOTOTHERAPY FOR PSORIASIS VULGARIS: A REVIEW Dr. Shweta Aryal *, Prof. Dr. Liu Jin Xian, Dr. Gong Shao Zhi and Dr. Jyoti Karki
Clinical Study Synopsis
 Clinical Study Synopsis This Clinical Study Synopsis is provided for patients and healthcare professionals to increase the transparency of Bayer's clinical research. This document is not intended to replace
Clinical Study Synopsis This Clinical Study Synopsis is provided for patients and healthcare professionals to increase the transparency of Bayer's clinical research. This document is not intended to replace
Kim A. Papp, MD PhD Probity Medical Research, Waterloo, ON, Canada
 Apremilast, an Oral Phosphodiesterase 4 Inhibitor, in Patients With Moderate to Severe Plaque Psoriasis: Pooled Efficacy Results from the Subgroup of Canadian Patients in Two Phase III Randomized Controlled
Apremilast, an Oral Phosphodiesterase 4 Inhibitor, in Patients With Moderate to Severe Plaque Psoriasis: Pooled Efficacy Results from the Subgroup of Canadian Patients in Two Phase III Randomized Controlled
Topical betamethasone for the prevention of acute radiation dermatitis in breast cancer patients
 Iran. J. Radiat. Res., 23; (2): 5 - Topical betamethasone for the prevention of acute radiation dermatitis in breast cancer patients F. Farhan, A. Kazemian, H. Alagheband Radiotherapy-Oncology Department,
Iran. J. Radiat. Res., 23; (2): 5 - Topical betamethasone for the prevention of acute radiation dermatitis in breast cancer patients F. Farhan, A. Kazemian, H. Alagheband Radiotherapy-Oncology Department,
Clearance in vulvar lichen sclerosus: a realistic treatment endpoint or a chimera?
 DOI: 10.1111/jdv.14516 JEADV SHORT REPORT Clearance in vulvar lichen sclerosus: a realistic treatment endpoint or a chimera? A. Borghi,* A. Virgili, S. Minghetti, G. Toni, M. Corazza Dipartimento di Scienze
DOI: 10.1111/jdv.14516 JEADV SHORT REPORT Clearance in vulvar lichen sclerosus: a realistic treatment endpoint or a chimera? A. Borghi,* A. Virgili, S. Minghetti, G. Toni, M. Corazza Dipartimento di Scienze
Original Policy Date
 MP 2.01.07 Psoralens with Ultraviolet A (PUVA) Medical Policy Section Medicine Issue 12:2013 Original Policy Date 12:2013 Last Review Status/Date Reviewed by consensus/12:2013 Return to Medical Policy
MP 2.01.07 Psoralens with Ultraviolet A (PUVA) Medical Policy Section Medicine Issue 12:2013 Original Policy Date 12:2013 Last Review Status/Date Reviewed by consensus/12:2013 Return to Medical Policy
Phototherapy and Photochemotherapy Treatment (Ultraviolet A [PUVA] and B [UBV])
![Phototherapy and Photochemotherapy Treatment (Ultraviolet A [PUVA] and B [UBV]) Phototherapy and Photochemotherapy Treatment (Ultraviolet A [PUVA] and B [UBV])](/thumbs/89/99382632.jpg) Origination: 09/27/07 Revised: 08/2/17 Annual Review: 11/2/17 Purpose: To provide Phototherapy and Photochemotherapy Treatment (PUVA and UBV) guidelines for the Medical Department staff to reference when
Origination: 09/27/07 Revised: 08/2/17 Annual Review: 11/2/17 Purpose: To provide Phototherapy and Photochemotherapy Treatment (PUVA and UBV) guidelines for the Medical Department staff to reference when
Drug Class Literature Scan: Topical Antipsoriatics
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Clinical Trial Report Synopsis
 This document has been do\vnloaded from \v ww.leo-pharma.com subject to the terms of use state on the website. It contains data and results regarding approved and non-approved uses, formulations or treatment
This document has been do\vnloaded from \v ww.leo-pharma.com subject to the terms of use state on the website. It contains data and results regarding approved and non-approved uses, formulations or treatment
Psoriasis is an inflammatory condition of the skin that
 Mayo Clin Proc, September 2001, Vol 76 Topical Therapies for Localized Psoriasis 943 Concise Review for Clinicians Concise Review for Clinicians Topical Therapies for Localized Psoriasis PATRICIA M. WITMAN,
Mayo Clin Proc, September 2001, Vol 76 Topical Therapies for Localized Psoriasis 943 Concise Review for Clinicians Concise Review for Clinicians Topical Therapies for Localized Psoriasis PATRICIA M. WITMAN,
Comparison of the efficacy of PUVA versus BBUVB in the treatment of psoriasis vulgaris
 IJMAMR 5 (2017) 1-6 ISSN 2053-1834 Comparison of the efficacy of PUVA versus BBUVB in the treatment of psoriasis vulgaris Tran Hau Khang* and Le Huu Doanh National Hospital of Dermatology and Venereology,
IJMAMR 5 (2017) 1-6 ISSN 2053-1834 Comparison of the efficacy of PUVA versus BBUVB in the treatment of psoriasis vulgaris Tran Hau Khang* and Le Huu Doanh National Hospital of Dermatology and Venereology,
The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION. 18 January 2012
 The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 18 January 2012 EPIDUO, gel Tube of 30 g (CIP code: 383 814-6) Tube of 60 g (CIP code: 383 816-9) Applicant: GALDERMA
The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 18 January 2012 EPIDUO, gel Tube of 30 g (CIP code: 383 814-6) Tube of 60 g (CIP code: 383 816-9) Applicant: GALDERMA
TRANSPARENCY COMMITTEE OPINION. 26 April 2006
 TRANSPARENCY COMMITTEE OPINION 26 April 2006 REMICADE 100 mg powder for concentrate for solution for infusion Box of 1 (CIP code: 562 070.1) Applicant : laboratoires Schering Plough List I Drug for hospital
TRANSPARENCY COMMITTEE OPINION 26 April 2006 REMICADE 100 mg powder for concentrate for solution for infusion Box of 1 (CIP code: 562 070.1) Applicant : laboratoires Schering Plough List I Drug for hospital
NORLAND AVENUE PHARMACY PRESCRIPTION COMPOUNDING FOR GENERAL PRACTICE
 AUGUST 2011 PRESCRIPTION COMPOUNDING N ORLANDA VENUEP HARMACY. COM We customize individual prescriptions for the specific needs of our patients. INSIDE THIS ISSUE: Hemorrhoids 2 Celiac Disease 3 PRESCRIPTION
AUGUST 2011 PRESCRIPTION COMPOUNDING N ORLANDA VENUEP HARMACY. COM We customize individual prescriptions for the specific needs of our patients. INSIDE THIS ISSUE: Hemorrhoids 2 Celiac Disease 3 PRESCRIPTION
Predicting the Response to Phototherapy for Psoriasis Patients
 A*STAR-NHG-NTU Skin Research Grant Joint Workshop 17 October 2015 Predicting the Response to Phototherapy for Psoriasis Patients Is it possible? Dr Eugene Tan Consultant Dermatologist National Skin Centre
A*STAR-NHG-NTU Skin Research Grant Joint Workshop 17 October 2015 Predicting the Response to Phototherapy for Psoriasis Patients Is it possible? Dr Eugene Tan Consultant Dermatologist National Skin Centre
A study of treatment modalities in psoriasis in dermatology outpatient department of a tertiary care teaching hospital
 Original article A study of treatment modalities in psoriasis in dermatology outpatient department of a tertiary care teaching hospital 1Y Roja Ramani, 2 Benu Panigrahy, 3 Sailenkumar Mishra, 4 BTPS Singh
Original article A study of treatment modalities in psoriasis in dermatology outpatient department of a tertiary care teaching hospital 1Y Roja Ramani, 2 Benu Panigrahy, 3 Sailenkumar Mishra, 4 BTPS Singh
TREATMENT OPTIONS FOR PSORIASIS. Sandra Hanlon Dermatology Senior Charge Nurse NHS Ayrshire and Arran 07/03/17
 TREATMENT OPTIONS FOR PSORIASIS Sandra Hanlon Dermatology Senior Charge Nurse NHS Ayrshire and Arran 07/03/17 PSORIASIS A chronic, non-infectious inflammatory skin condition that has no cure Characterised
TREATMENT OPTIONS FOR PSORIASIS Sandra Hanlon Dermatology Senior Charge Nurse NHS Ayrshire and Arran 07/03/17 PSORIASIS A chronic, non-infectious inflammatory skin condition that has no cure Characterised
Objectives The objectives of the study were to compare the efficacy and safety of three different strengths
 MC 392 Study 18 May 1998 PageS ABSTRACT Objectives The objectives of the study were to compare the efficacy and safety of three different strengths of caldpotriol cream (10, 25, 50 f!g/ g) and placebo
MC 392 Study 18 May 1998 PageS ABSTRACT Objectives The objectives of the study were to compare the efficacy and safety of three different strengths of caldpotriol cream (10, 25, 50 f!g/ g) and placebo
Vulval dermatoses. Dr Fiona Lewis, Consultant Dermatologist St John s Institute of Dermatology, London & Heatherwood & Wexham Park Hospital, Slough
 Vulval dermatoses Dr Fiona Lewis, Consultant Dermatologist St John s Institute of Dermatology, London & Heatherwood & Wexham Park Hospital, Slough Pigmentation Vulvodynia Ulcers Genetic Pruritus VULVAL
Vulval dermatoses Dr Fiona Lewis, Consultant Dermatologist St John s Institute of Dermatology, London & Heatherwood & Wexham Park Hospital, Slough Pigmentation Vulvodynia Ulcers Genetic Pruritus VULVAL
PFIZER INC. Study Initiation Date and Completion Dates: Information not available (Date of Statistical Report: 16 May 2004)
 PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
Details of UNCOVER-2 and UNCOVER-3 Study Results Published in The Lancet
 June 10, 2015 Eli Lilly and Company Lilly Corporate Center Indianapolis, Indiana 46285 U.S.A. +1.317.276.2000 www.lilly.com For Release: Refer to: Immediately Tim Coulom; tim.coulom@lilly.com; 317-771-2241
June 10, 2015 Eli Lilly and Company Lilly Corporate Center Indianapolis, Indiana 46285 U.S.A. +1.317.276.2000 www.lilly.com For Release: Refer to: Immediately Tim Coulom; tim.coulom@lilly.com; 317-771-2241
The Ointment is back!
 Now Available for Plaque Psoriasis The Ointment is back! Bioequivalent to Dovonex * Ointment Before prescribing, please see attached full Prescribing Information. The same Delivers the reliability you
Now Available for Plaque Psoriasis The Ointment is back! Bioequivalent to Dovonex * Ointment Before prescribing, please see attached full Prescribing Information. The same Delivers the reliability you
The Treatment Toolbox for Severe Pediatric Psoriasis
 The Treatment Toolbox for Severe Pediatric Psoriasis Dr. Kim A. Papp, MD, PhD, FRCPC, FAAD K Papp Clinical Research and Probity Medical Research Objectives: Treating severe pediatric psoriasis 1. Challenges
The Treatment Toolbox for Severe Pediatric Psoriasis Dr. Kim A. Papp, MD, PhD, FRCPC, FAAD K Papp Clinical Research and Probity Medical Research Objectives: Treating severe pediatric psoriasis 1. Challenges
Effects of Acupuncture on Chinese Adult Patients with Psoriatic Arthritis: A Prospective Cohort Study
 Effects of Acupuncture on Chinese Adult Patients with Psoriatic Arthritis: A Prospective Cohort Study Peiyi Chen 1, Tiantian Xin 2* and Yingchun Zeng 3,4 1 School of Nursing, Guangzhou University of Chinese
Effects of Acupuncture on Chinese Adult Patients with Psoriatic Arthritis: A Prospective Cohort Study Peiyi Chen 1, Tiantian Xin 2* and Yingchun Zeng 3,4 1 School of Nursing, Guangzhou University of Chinese
This study may not be disseminated, reproduced in whole or in part without the written permission of Roduve Healthcare Solutions.
 This study may not be disseminated, reproduced in whole or in part without the written permission of Roduve Healthcare Solutions. A Randomized Controlled Trial for Roduve Healthcare Solutions on the Efficacy
This study may not be disseminated, reproduced in whole or in part without the written permission of Roduve Healthcare Solutions. A Randomized Controlled Trial for Roduve Healthcare Solutions on the Efficacy
Novan Provides Update on SB414 Inflammatory Skin Disease Development Program
 Novan Provides Update on SB414 Inflammatory Skin Disease Development Program SB414 Nitric Oxide-Releasing Cream Safe and Well-Tolerated in Psoriasis Phase 1b Trial Preclinical Data with SB414 Targeting
Novan Provides Update on SB414 Inflammatory Skin Disease Development Program SB414 Nitric Oxide-Releasing Cream Safe and Well-Tolerated in Psoriasis Phase 1b Trial Preclinical Data with SB414 Targeting
Prescribing Information
 Prescribing Information Pr DERMOVATE Cream (clobetasol propionate cream, USP) Pr DERMOVATE Ointment (clobetasol propionate ointment, USP) Topical corticosteroid TaroPharma Preparation Date: A Division
Prescribing Information Pr DERMOVATE Cream (clobetasol propionate cream, USP) Pr DERMOVATE Ointment (clobetasol propionate ointment, USP) Topical corticosteroid TaroPharma Preparation Date: A Division
The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION. 13 May 2009
 The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 13 May 2009 STELARA 45 mg, solution for injection B/1 x 0.5 ml vial (CIP code: 392 586-2) JANSSEN-CILAG Ustekinumab
The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 13 May 2009 STELARA 45 mg, solution for injection B/1 x 0.5 ml vial (CIP code: 392 586-2) JANSSEN-CILAG Ustekinumab
Prescribing Information. Taro-Clobetasol. Taro-Clobetasol
 Prescribing Information Pr Taro-Clobetasol Clobetasol Propionate Cream USP, 0.05% w/w Pr Taro-Clobetasol Clobetasol Propionate Ointment USP, 0.05% w/w Therapeutic Classification Topical corticosteroid
Prescribing Information Pr Taro-Clobetasol Clobetasol Propionate Cream USP, 0.05% w/w Pr Taro-Clobetasol Clobetasol Propionate Ointment USP, 0.05% w/w Therapeutic Classification Topical corticosteroid
Psoriasis Disease Severity Affects Patient Satisfaction With Therapy
 82 Psoriasis Disease Severity Affects Patient Satisfaction With Therapy Korman NJ 1, Zhao Y 2, Tran MH 2, Lu J 2 (Please see authors affiliations on the last panel) Background Psoriasis is a chronic, immune
82 Psoriasis Disease Severity Affects Patient Satisfaction With Therapy Korman NJ 1, Zhao Y 2, Tran MH 2, Lu J 2 (Please see authors affiliations on the last panel) Background Psoriasis is a chronic, immune
The reliability of three psoriasis assessment tools: Psoriasis area and severity index, body surface area and physician global assessment
 Original papers The reliability of three psoriasis assessment tools: Psoriasis area and severity index, body surface area and physician global assessment Agnieszka Bożek A F, Adam Reich A F Department
Original papers The reliability of three psoriasis assessment tools: Psoriasis area and severity index, body surface area and physician global assessment Agnieszka Bożek A F, Adam Reich A F Department
Trial ID: LP May-2016 Page 2 of 5
 Trial ID: LP0053-1227 31-May-2016 Page 2 of 5 Clinical Trial Report Synopsis Statement Approval Statement, LEO Pharma A/S The following persons have approved this clinical study report synopsis using electronic
Trial ID: LP0053-1227 31-May-2016 Page 2 of 5 Clinical Trial Report Synopsis Statement Approval Statement, LEO Pharma A/S The following persons have approved this clinical study report synopsis using electronic
Individual Study Table Referring to Part of the Dossier. Volume:
 2 Synopsis Title of study: An Investigator-blind, Controlled Study to Assess the Efficacy of Five Distinct Combinations of in Different Concentrations Compared to Placebo and to Two Active Controls in
2 Synopsis Title of study: An Investigator-blind, Controlled Study to Assess the Efficacy of Five Distinct Combinations of in Different Concentrations Compared to Placebo and to Two Active Controls in
Clinical Management Of Psoriasis Using 0.25% Niosomal Methotrexate Gel: A Placebo Controlled Double Blind Study
 ISPUB.COM The Internet Journal of Dermatology Volume 3 Number 1 Clinical Management Of Psoriasis Using 0.25% Niosomal Methotrexate Gel: A Placebo Controlled Double P Lakshmi, S Devi, S Bhaskaran, S Sacchidananda,
ISPUB.COM The Internet Journal of Dermatology Volume 3 Number 1 Clinical Management Of Psoriasis Using 0.25% Niosomal Methotrexate Gel: A Placebo Controlled Double P Lakshmi, S Devi, S Bhaskaran, S Sacchidananda,
Clinico Pathological Test SCPA605-Essential Pathology
 Clinico Pathological Test SCPA605-Essential Pathology Somphong Narkpinit, M.D. Department of Pathogbiology, Faculty of Science, Mahidol University e-mail : somphong.nar@mahidol.ac.th Pathogenesis of allergic
Clinico Pathological Test SCPA605-Essential Pathology Somphong Narkpinit, M.D. Department of Pathogbiology, Faculty of Science, Mahidol University e-mail : somphong.nar@mahidol.ac.th Pathogenesis of allergic
Instructions concerning lifestyle and concomitant medications
 Appendix S1. Supporting information STUDY 1: Double-blind, vehicle-controlled, single centre, two-period, Phase I study to investigate pharmacokinetics, safety and tolerability of a single topical dose
Appendix S1. Supporting information STUDY 1: Double-blind, vehicle-controlled, single centre, two-period, Phase I study to investigate pharmacokinetics, safety and tolerability of a single topical dose
0BCore Safety Profile. Pharmaceutical form(s)/strength: Cream 1% DK/H/PSUR/0009/005 Date of FAR:
 0BCore Safety Profile Active substance: Pimecrolimus Pharmaceutical form(s)/strength: Cream 1% P-RMS: DK/H/PSUR/0009/005 Date of FAR: 06.06.2013 4.3 Contraindications Hypersensitivity to pimecrolimus,
0BCore Safety Profile Active substance: Pimecrolimus Pharmaceutical form(s)/strength: Cream 1% P-RMS: DK/H/PSUR/0009/005 Date of FAR: 06.06.2013 4.3 Contraindications Hypersensitivity to pimecrolimus,
Bioavailability 55% - 77%
 Brand Name: Cosentyx Generic Name: secukinumab Manufacturer 1 : Novartis Pharmaceuticals Corporation Drug Class 2,3 : Antipsoriatic Agent, Interleukin-17A Receptor Antagonist Uses: Labeled Uses 1,2,3 :
Brand Name: Cosentyx Generic Name: secukinumab Manufacturer 1 : Novartis Pharmaceuticals Corporation Drug Class 2,3 : Antipsoriatic Agent, Interleukin-17A Receptor Antagonist Uses: Labeled Uses 1,2,3 :
NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE SCOPE
 NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE 1 Guideline title SCOPE Psoriasis: the management of psoriasis 1.1 Short title Psoriasis 2 The remit The Department of Health has asked NICE: 'to produce
NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE 1 Guideline title SCOPE Psoriasis: the management of psoriasis 1.1 Short title Psoriasis 2 The remit The Department of Health has asked NICE: 'to produce
Supplementary Figure 1 IMQ-Induced Mouse Model of Psoriasis. IMQ cream was
 Supplementary Figure 1 IMQ-Induced Mouse Model of Psoriasis. IMQ cream was painted on the shaved back skin of CBL/J and BALB/c mice for consecutive days. (a, b) Phenotypic presentation of mouse back skin
Supplementary Figure 1 IMQ-Induced Mouse Model of Psoriasis. IMQ cream was painted on the shaved back skin of CBL/J and BALB/c mice for consecutive days. (a, b) Phenotypic presentation of mouse back skin
Horizon Scanning Centre March Tildrakizumab for moderate to severe plaque psoriasis SUMMARY NIHR HSC ID: 6798
 Horizon Scanning Centre March 2015 Tildrakizumab for moderate to severe plaque psoriasis SUMMARY NIHR HSC ID: 6798 This briefing is based on information available at the time of research and a limited
Horizon Scanning Centre March 2015 Tildrakizumab for moderate to severe plaque psoriasis SUMMARY NIHR HSC ID: 6798 This briefing is based on information available at the time of research and a limited
PDF hosted at the Radboud Repository of the Radboud University Nijmegen
 PDF hosted at the Radboud Repository of the Radboud University Nijmegen he following full text is a publisher's version. For additional information about this publication click this link. http://hdl.handle.net/2066/24322
PDF hosted at the Radboud Repository of the Radboud University Nijmegen he following full text is a publisher's version. For additional information about this publication click this link. http://hdl.handle.net/2066/24322
PFIZER INC. GENERIC DRUG NAME and/or COMPOUND NUMBER: 5% Spironolactone Cream/ PF
 PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
Pravit Asawanonda, MD, DSc, and Yaowalak Nateetongrungsak, MD Bangkok, Thailand
 Methotrexate plus narrowband UVB phototherapy versus narrowband UVB phototherapy alone in the treatment of plaque-type psoriasis: A randomized, placebo-controlled study Pravit Asawanonda, MD, DSc, and
Methotrexate plus narrowband UVB phototherapy versus narrowband UVB phototherapy alone in the treatment of plaque-type psoriasis: A randomized, placebo-controlled study Pravit Asawanonda, MD, DSc, and
PFIZER INC. Study Initiation Date and Completion Dates: 09 March 2000 to 09 August 2001.
 PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
Blue light therapy. Blue light therapy has shown to have a positive effect on the following: Why to choose blue light?
 PURE COMPLEXiON Blue light therapy Blue light therapy is an alternative form of therapy, where the narrow spectrum of the organic blue LED light is used to heal, treat and prevent skin diseases and other
PURE COMPLEXiON Blue light therapy Blue light therapy is an alternative form of therapy, where the narrow spectrum of the organic blue LED light is used to heal, treat and prevent skin diseases and other
