August 7, Dear Ms. Verma: RE: 2018 Preliminary Gapfill Payment Determinations for CY2019. Dear Ms. Verma:
|
|
|
- Myron Scott
- 5 years ago
- Views:
Transcription
1 ASSOCIATION FOR MOLECULAR PATHOLOGY Education. Innovation & Improved Patient Care. Advocacy Rockville Pike. Bethesda, Maryland Tel: Fax: August 7, 2018 Seema Verma, CMS Administrator Centers for Medicare & Medicaid Services Department of Health and Human Services Hubert H. Humphrey Building, Room 445-G 200 Independence Avenue, SW Washington, DC Dear Ms. Verma: RE: 2018 Payment Determinations for CY2019 Dear Ms. Verma: On behalf of the Association of Molecular Pathology (AMP), thank you for this opportunity to submit comments on the 2018 preliminary gapfill determinations for CY2019. AMP is an international medical and professional association representing approximately 2,400 physicians, doctoral scientists, and medical technologists who perform or are involved with laboratory testing based on knowledge derived from molecular biology, genetics, and genomics. Membership includes professionals from the government, academic medicine, private and hospital-based clinical laboratories, and the in vitro diagnostics industry. Many of the codes that are going through the gapfill pricing exercise are not new codes but existing Clinical Laboratory Fee Schedule (CLFS) codes for which the Centers for Medicare and Medicaid Services (CMS) received no (i.e., values of zero) and/or insufficient data to calculate a weighted median private payor rate during the 2017 Protecting Access to Medicare Act (PAMA) pricing process. The PAMA final rule states that for a CDLT for which CMS receives no applicable information, payment is made based on the crosswalking or gapfilling methods described in (b)(1) and (2). However, the pricing process employed by CMS to solicit stakeholder input on these codes provided stakeholders and the Advisory Panel on CDLTs little time to provide meaningful and comprehensive input to CMS. We recommend that in the future the agency include these codes on the agenda for the CLFS public meeting that immediately follows the reporting period. We believe this will allow all interested stakeholders the opportunity to provide meaningful input on the re-pricing of these codes. Since CMS began utilizing the gapfill process to price services on the CLFS, AMP has expressed concerns about the lack of transparency. It remains difficult to constructively respond to preliminary gapfill values without increased transparency and no discernible rationale as to how the MACs determined preliminary pricing. We hoped that the implementation of PAMA would ultimately improve this process. In the PAMA final rule, CMS noted that although it generally provided its rationale for the basis of payment and for crosswalks, it did not typically provide explanations of final payment amounts for gapfilled tests in the past. CMS explained that it
2 Page 2 of 5 was adding 42 CFR (d)(3) and (4) to provide an explanation of gapfilled payment amounts and how we took into account the Panel s recommendations. However, the preliminary gapfill rates posted by CMS do not include explanations. AMP plans to follow-up with CMS on this issue to get a better understanding of how the agency plans to implement this and improve the process. Based on the limited information released by CMS, we are extremely concerned that the preliminary national limitation amounts (NLAs) do not accurately reflect the value of these procedures. We urge CMS to seriously consider the comments provided by AMP and other stakeholders during the preliminary determination comment period. Undervaluation of these services threatens patient access to care if laboratories can no longer afford to provide these procedures. Genome CPT codes (81425, 81426, and 81427) Whole genome sequencing procedures are clinically actionable, most often for patients with severe genetic disorders. These patients have no alternatives: either no existing diagnostic panel is appropriate or the cost of multiple panels exceeds the cost of either whole exome or whole genome sequencing. There is now a significant body of peer-reviewed literature that shows the value of whole genome sequencing in clinical practice. In July 2018, a literature review of exome and genome sequencing was published that examined the role of genome sequencing in clinical care of select patients 1. AMP appreciates that all MACs recommended a price for each code undergoing gapfill, however we remain very concerned about many of the gapfill values submitted. Under 42 C.F.R (b)(1), Medicare regulations state that MACs are required to consider the following criteria when establishing gapfill rates: (b) Gapfilling. Gapfilling is used when no comparable existing test is available. (1) In the first year, carrier-specific amounts are established for the new test code using the following sources of information to determine gapfill amounts, if available: (i) Charges for the test and routine discounts to charges; (ii) Resources required to perform the test; (iii) Payment amounts determined by other payers; and (iv) Charges, payment amounts, and resources required for other tests that may be comparable or otherwise relevant. The preliminary determinations for genome sequencing (CPT codes 81425, 81426, and 81427) fall significantly below the actual costs to perform these procedures. A recent publication reports that it costs $8,482 per patient to perform a clinical genome sequencing on a patient. 2 Further, in investigating charges for these services, a search of whole genome sequencing services using the Concert Genetics database, laboratories with prices available currently charge prices that range from $7,395 to $7,500 3 for Clark, M. M. (2018). "Meta-analysis of the diagnostic and clinical utility of genome and exome sequencing and chromosomal microarray in children with suspected genetic diseases." Genome Med EPUB. 2 Farnaes, L., et al. (2018). "Rapid whole-genome sequencing decreases infant morbidity and cost of hospitalization." NPJ Genom Med 3:
3 Page 3 of 5 AMP cannot support pricing of the genome codes at this level and urges CMS not to finalize the preliminary pricing determinations for these codes. Instead, CMS should consider crosswalks for these services based on their relationship to CPT codes currently on the CLFS to set an appropriate price. Due to the PAMA reporting process, comparable codes are now priced on the CLFS, namely the whole exome sequencing codes (81415, 81416, and 81417). Below, AMP offers a method for establishing a relationship between the priced exome codes to the genome codes. The exome codes are comparable for a number of reasons. Whole genome sequencing and whole exome sequencing are similar in methodology, both in terms of sequencing and informatics pipelines, as well as interpretation requirements. However, genome sequencing procedures require sequencing and interpretation of more DNA content than exome sequencing procedures. Therefore, the exome sequencing procedures codes are comparable to the genome sequencing procedure codes, but the work and resources required for genome sequencing are approximately twice that of the exome sequencing procedure codes. AMP recommends that CMS reconsider the preliminary determinations for the genome sequencing procedure codes and consider a value that is twice the value of the exome sequencing codes. Comparator s (Exome codes 81415, 81416, 81417) 2018 NLA syndrome); sequence analysis $4, syndrome); sequence analysis, each comparator exome (eg, parents, siblings) (List separately in addition to code for primary procedure) syndrome); re-evaluation of previously obtained exome sequence (eg, updated knowledge or unrelated condition/syndrome) $12, $ s for Insufficiently-priced 2018 Gapfill Genome CPT codes heritable disorder or syndrome); sequence analysis $ X 2 = $4780 X 2 = $ heritable disorder or syndrome); sequence analysis, each comparator genome (eg, parents, siblings) (List separately in addition to code for primary procedure) $ X 2 = $4780 X 2 = $9560
4 Page 4 of heritable disorder or syndrome); reevaluation of previously obtained genome sequence (eg, updated knowledge or unrelated condition/syndrome) $ X 2 = $640 If CMS intends on gapfilling these codes, AMP urges the agency and the MACs to evaluate charges, payment amounts, and resources for these services as directed by the regulation cited above. We have provided some preliminary information in these comments, but would welcome the opportunity to work with CMS and the MACs to identify additional information required to develop accurate gapfill amounts. X-linked Intellectual Disability s (81425, 81426, and 81427) Again, AMP appreciates that all MACs recommended a price for each code undergoing gapfill, however we remain concerned about many of the gapfill values submitted. In accordance with 42 C.F.R (b)(1), CMS should instead evaluate charges, payment amounts, and resources required for other tests that may be comparable or otherwise relevant. CPT detects pathogenic variants (e.g., single nucleotide variants, small indels) in genes known to be causative of X-linked intellectual disability (XLID). This procedure is performed by next generation sequencing and bioinformatics analysis followed by professional interpretation. Similar methodologies are employed to comparable code 81432, but a larger number of required genes exist in the XLID panel. Therefore, is roughly equivalent to twice the resources required for Further, in investigating charges for these services, a search of XLID panel testing using the Concert Genetics database, laboratories with prices available currently charge prices that range from $1,700 to $3, Similarly, CPT code detects pathogenic deletion and duplication variants in genes known to be causative of XLID. This procedure is performed most commonly using a targeted array for the X chromosome. AMP urges CMS and the MACs not to finalize the preliminary determinations for these codes. Instead, CMS should consider the relationship of these codes to CPT codes currently on the CLFS. The preliminary gapfill determinations are insufficient when one examines comparable tests and current charges for the tests. Below, AMP offers a method for establishing more accurate pricing for and using comparable codes that exist on the CLFS. Comparator s (81432 and 81436) 2018 NLA Hereditary breast cancer-related disorders (eg, hereditary breast cancer, hereditary ovarian cancer, hereditary endometrial cancer); genomic sequence analysis panel, must include sequencing of at least 10 genes, always including BRCA1, BRCA2, CDH1, MLH1, MSH2, MSH6, PALB2, PTEN, STK11, and TP53 Hereditary colon cancer disorders (eg, Lynch syndrome, PTEN hamartoma syndrome, Cowden syndrome, familial adenomatosis polyposis); duplication/deletion analysis panel, must include analysis of at least 5 genes, including MLH1, MSH2, EPCAM, SMAD4, and STK11 $ $
5 Page 5 of 5 s for Insufficiently-priced 2018 Gapfill Genome CPT codes X-linked intellectual disability (XLID) (eg, syndromic and non-syndromic XLID); genomic sequence analysis panel, must include sequencing of at least 60 genes, including ARX, ATRX, CDKL5, FGD1, FMR1, HUWE1, IL1RAPL, KDM5C, L1CAM, MECP2, MED12, MID1, OCRL, RPS6KA3, and SLC16A2 $ X 2 = $ X 2 = $ X-linked intellectual disability (XLID) (eg, syndromic and non-syndromic XLID); duplication/deletion gene analysis, must include analysis of at least 60 genes, including ARX, ATRX, CDKL5, FGD1, FMR1, HUWE1, IL1RAPL, KDM5C, L1CAM, MECP2, MED12, MID1, OCRL, RPS6KA3, and SLC16A2 $ X 2 = $ X 2 = $ We thank you for the opportunity to submit these comments on the preliminary gapfill recommendations. We believe that the rationale, data, and recommendations provided above will result in more accurate and equitable pricing for these services. We are happy to answer any questions about our recommendations and provide follow up information. Please direct your correspondence to Tara Burke, Director of Public Policy and Advocacy, at tburke@amp.org. Sincerely, Samuel K. Caughron, MD Chair, Economics Affairs Committee Association for Molecular Pathology
Code CPT Descriptor Test Purpose and Method Crosswalk Recommendation SEPT9 (Septin9) (e.g., colorectal cancer) methylation analysis
 ASSOCIATION FOR MOLECULAR PATHOLOGY Education. Innovation & Improved Patient Care. Advocacy. 9650 Rockville Pike. Bethesda, Maryland 20814 Tel: 301-634-7939 Fax: 301-634-7995 amp@amp.org www.amp.org August
ASSOCIATION FOR MOLECULAR PATHOLOGY Education. Innovation & Improved Patient Care. Advocacy. 9650 Rockville Pike. Bethesda, Maryland 20814 Tel: 301-634-7939 Fax: 301-634-7995 amp@amp.org www.amp.org August
Mr. Glenn McGuirk Centers for Medicare and Medicaid Services 7500 Security Boulevard Baltimore, MD 21244
 August 6, 2018 Mr. Glenn McGuirk Centers for Medicare and Medicaid Services 7500 Security Boulevard Baltimore, MD 21244 Submitted via email: glenn.mcguirk@cms.hhs.gov Dear Mr. McGuirk, The American Clinical
August 6, 2018 Mr. Glenn McGuirk Centers for Medicare and Medicaid Services 7500 Security Boulevard Baltimore, MD 21244 Submitted via email: glenn.mcguirk@cms.hhs.gov Dear Mr. McGuirk, The American Clinical
Prior Authorization. Additional Information:
 Genetic Testing for Lynch Syndrome MP9487 Covered Service: Prior Authorization Required: Additional Information: Yes when meets criteria below Yes-as shown below Pre and post test genetic counseling is
Genetic Testing for Lynch Syndrome MP9487 Covered Service: Prior Authorization Required: Additional Information: Yes when meets criteria below Yes-as shown below Pre and post test genetic counseling is
Genetic Testing for Lynch Syndrome
 Genetic Testing for Lynch Syndrome MP9487 Covered Service: Prior Authorization Required: Additional Information: Yes when meets criteria below Yes-as shown below Pre and post-test genetic counseling is
Genetic Testing for Lynch Syndrome MP9487 Covered Service: Prior Authorization Required: Additional Information: Yes when meets criteria below Yes-as shown below Pre and post-test genetic counseling is
Yes when meets criteria below. Dean Health Plan covers when Medicare also covers the benefit.
 Genetic Testing for Lynch Syndrome MP9487 Covered Service: Prior Authorization Required: Additional Information: Yes when meets criteria below Yes-as shown below Pre and post test genetic counseling is
Genetic Testing for Lynch Syndrome MP9487 Covered Service: Prior Authorization Required: Additional Information: Yes when meets criteria below Yes-as shown below Pre and post test genetic counseling is
Calendar Year (CY) 2017 Clinical Laboratory Fee Schedule (CLFS) Final Determinations
 Calendar Year (CY) 2017 Clinical Laboratory Fee Schedule (CLFS) Final Determinations A. Reconsidered Tests For 2016, CMS implemented four new HCPCS G codes for definitive drug testing: G0480 (Drug test(s),
Calendar Year (CY) 2017 Clinical Laboratory Fee Schedule (CLFS) Final Determinations A. Reconsidered Tests For 2016, CMS implemented four new HCPCS G codes for definitive drug testing: G0480 (Drug test(s),
WHAT IS A GENE? CHROMOSOME DNA PROTEIN. A gene is made up of DNA. It carries instructions to make proteins.
 WHAT IS A GENE? CHROMOSOME GENE DNA A gene is made up of DNA. It carries instructions to make proteins. The proteins have specific jobs that help your body work normally. PROTEIN 1 WHAT HAPPENS WHEN THERE
WHAT IS A GENE? CHROMOSOME GENE DNA A gene is made up of DNA. It carries instructions to make proteins. The proteins have specific jobs that help your body work normally. PROTEIN 1 WHAT HAPPENS WHEN THERE
Clinical Laboratory Fee Schedule CY 2015 Updates June 9, 2014
 Clinical Laboratory Fee Schedule CY 2015 Updates June 9, 2014 Please note that this listing includes the most recent codes provided by the American Medical Association (AMA), and that it is subject to
Clinical Laboratory Fee Schedule CY 2015 Updates June 9, 2014 Please note that this listing includes the most recent codes provided by the American Medical Association (AMA), and that it is subject to
WHAT IS A GENE? CHROMOSOME DNA PROTEIN. A gene is made up of DNA. It carries instructions to make proteins.
 WHAT IS A GENE? CHROMOSOME E GEN DNA A gene is made up of DNA. It carries instructions to make proteins. The proteins have specific jobs that help your body work normally. PROTEIN 1 WHAT HAPPENS WHEN THERE
WHAT IS A GENE? CHROMOSOME E GEN DNA A gene is made up of DNA. It carries instructions to make proteins. The proteins have specific jobs that help your body work normally. PROTEIN 1 WHAT HAPPENS WHEN THERE
CPT Codes for Pharmacogenomic Tests
 CPT s for Pharmacogenomic Tests The table below lists CPT codes and lab fee information for pharmacogenomic tests as established by the Centers for Medicare and Medicaid Services. It was compiled by the
CPT s for Pharmacogenomic Tests The table below lists CPT codes and lab fee information for pharmacogenomic tests as established by the Centers for Medicare and Medicaid Services. It was compiled by the
Prior Authorization. Additional Information:
 Genetic Testing for Cowden Syndrome - PTEN Gene MP9488 Covered Service: Prior Authorization Required: Additional Information: Yes when meets criteria below Yes as shown below Pre and post-test genetic
Genetic Testing for Cowden Syndrome - PTEN Gene MP9488 Covered Service: Prior Authorization Required: Additional Information: Yes when meets criteria below Yes as shown below Pre and post-test genetic
NGS for Cancer Predisposition
 NGS for Cancer Predisposition Colin Pritchard MD, PhD University of Washington Dept. of Lab Medicine AMP Companion Society Meeting USCAP Boston March 22, 2015 Disclosures I am an employee of the University
NGS for Cancer Predisposition Colin Pritchard MD, PhD University of Washington Dept. of Lab Medicine AMP Companion Society Meeting USCAP Boston March 22, 2015 Disclosures I am an employee of the University
GYNplus: A Genetic Test for Hereditary Ovarian and/or Uterine Cancer
 GYNplus: A Genetic Test for Hereditary Ovarian and/or Uterine Cancer Causes of Hereditary Ovarian and Uterine Cancer uterine cancer ovarian cancer Sporadic 75-90% Sporadic 70-80% Hereditary, 5% Lynch syndrome
GYNplus: A Genetic Test for Hereditary Ovarian and/or Uterine Cancer Causes of Hereditary Ovarian and Uterine Cancer uterine cancer ovarian cancer Sporadic 75-90% Sporadic 70-80% Hereditary, 5% Lynch syndrome
The benefit of. knowing. Genetic testing for hereditary cancer. A patient support guide
 The benefit of knowing Genetic testing for hereditary cancer A patient support guide Does cancer run in your family? Cancer is more common in some families. Sometimes cancer is caused by a change in a
The benefit of knowing Genetic testing for hereditary cancer A patient support guide Does cancer run in your family? Cancer is more common in some families. Sometimes cancer is caused by a change in a
Medical Policy Update
 Medical Policy Update Summer 2017 Highlights of recent medical policy revisions as well as any new medical policies approved by Prevea360 Health Plan s Medical Policy Committee are shown below. The Medical
Medical Policy Update Summer 2017 Highlights of recent medical policy revisions as well as any new medical policies approved by Prevea360 Health Plan s Medical Policy Committee are shown below. The Medical
GYNplus. genetic testing for hereditary ovarian and/or uterine cancer
 GYNplus genetic testing for hereditary ovarian and/or uterine cancer What Are the Causes of Hereditary Ovarian and Uterine Cancer? uterine cancer ovarian cancer sporadic 70-80% hereditary 5% Lynch syndrome
GYNplus genetic testing for hereditary ovarian and/or uterine cancer What Are the Causes of Hereditary Ovarian and Uterine Cancer? uterine cancer ovarian cancer sporadic 70-80% hereditary 5% Lynch syndrome
MEDICAL POLICY Genetic Testing for Breast and Ovarian Cancers
 POLICY: PG0067 ORIGINAL EFFECTIVE: 07/30/02 LAST REVIEW: 01/25/18 MEDICAL POLICY Genetic Testing for Breast and Ovarian Cancers GUIDELINES This policy does not certify benefits or authorization of benefits,
POLICY: PG0067 ORIGINAL EFFECTIVE: 07/30/02 LAST REVIEW: 01/25/18 MEDICAL POLICY Genetic Testing for Breast and Ovarian Cancers GUIDELINES This policy does not certify benefits or authorization of benefits,
Genetic Testing for BRCA1 and BRCA2 Genes
 Genetic Testing for BRCA1 and BRCA2 Genes MP9478 Covered Service: Prior Authorization Required: Additional Information: Yes when meets criteria below Yes as shown below Pre and post-test genetic counseling
Genetic Testing for BRCA1 and BRCA2 Genes MP9478 Covered Service: Prior Authorization Required: Additional Information: Yes when meets criteria below Yes as shown below Pre and post-test genetic counseling
CentoCancer STRIVE FOR THE MOST COMPLETE INFORMATION
 CentoCancer STRIVE FOR THE MOST COMPLETE INFORMATION CentoCancer our most comprehensive oncogenetics panel for hereditary mutations Hereditary pathogenic variants confer an increased risk of developing
CentoCancer STRIVE FOR THE MOST COMPLETE INFORMATION CentoCancer our most comprehensive oncogenetics panel for hereditary mutations Hereditary pathogenic variants confer an increased risk of developing
Learn your genetic risk for the most common hereditary cancers.
 Learn your genetic risk for the most common hereditary cancers. color.com Color analyzes 30 genes including BRCA1 and BRCA2 to help women and men understand their risk for the most common hereditary cancers,
Learn your genetic risk for the most common hereditary cancers. color.com Color analyzes 30 genes including BRCA1 and BRCA2 to help women and men understand their risk for the most common hereditary cancers,
Re: NC Medicaid and NC Health Choice Program coverage of genetic testing for susceptibility to breast and ovarian cancer
 March 23, 2018 North Carolina (NC) Division of Medical Assistance (DMA), Clinical Policy Section C/O Pam Greeson, BSN, RN-C, pam.greeson@dhhs.nc.gov Nurse Consultant, Division of Medical Assistance Practitioners,
March 23, 2018 North Carolina (NC) Division of Medical Assistance (DMA), Clinical Policy Section C/O Pam Greeson, BSN, RN-C, pam.greeson@dhhs.nc.gov Nurse Consultant, Division of Medical Assistance Practitioners,
PROVIDER POLICIES & PROCEDURES
 PROVIDER POLICIES & PROCEDURES GENETIC CANCER SUSCEPTIBILITY PANELS USING NEXT GENERATION SEQUENCING The purpose of this document is to assist providers enrolled in the Connecticut Medical Assistance Program
PROVIDER POLICIES & PROCEDURES GENETIC CANCER SUSCEPTIBILITY PANELS USING NEXT GENERATION SEQUENCING The purpose of this document is to assist providers enrolled in the Connecticut Medical Assistance Program
Genetic Testing for BRCA1 and BRCA2 Genes
 Genetic Testing f BRCA1 and BRCA2 Genes MP9478 Covered Service: Pri Authization Required: Additional Infmation: Yes when meets criteria below Yes--as shown below Pre and post-test genetic counseling is
Genetic Testing f BRCA1 and BRCA2 Genes MP9478 Covered Service: Pri Authization Required: Additional Infmation: Yes when meets criteria below Yes--as shown below Pre and post-test genetic counseling is
The Next Generation of Hereditary Cancer Testing
 The Next Generation of Hereditary Cancer Testing Why Genetic Testing? Cancers can appear to run in families. Often this is due to shared environmental or lifestyle patterns, such as tobacco use. However,
The Next Generation of Hereditary Cancer Testing Why Genetic Testing? Cancers can appear to run in families. Often this is due to shared environmental or lifestyle patterns, such as tobacco use. However,
10/21/ New Release, Quest Diagnostics Nichols Institute, Valencia
 NEW TESTS Please Note: Not all test codes assigned to each assay are listed in the table of contents. Please refer to the complete listing on the page numbers indicated. Test Code Test Name Effective Date
NEW TESTS Please Note: Not all test codes assigned to each assay are listed in the table of contents. Please refer to the complete listing on the page numbers indicated. Test Code Test Name Effective Date
Genetic Testing for Inherited Conditions
 Genetic Testing for Inherited Conditions Policy Number: 2018-101 Effective Date: January 26, 2018 Review Date: January 26, 2018 Next Review Date: January 26, 2019 Important Information - Please Read Before
Genetic Testing for Inherited Conditions Policy Number: 2018-101 Effective Date: January 26, 2018 Review Date: January 26, 2018 Next Review Date: January 26, 2019 Important Information - Please Read Before
Association for Molecular Pathology Promoting Clinical Practice, Basic Research, and Education in Molecular Pathology
 Association for Molecular Pathology Promoting Clinical Practice, Basic Research, and Education in Molecular Pathology 9650 Rockville Pike, Bethesda, Maryland 20814 Tel: 301-634-7939 Fax: 301-634-7990 Email:
Association for Molecular Pathology Promoting Clinical Practice, Basic Research, and Education in Molecular Pathology 9650 Rockville Pike, Bethesda, Maryland 20814 Tel: 301-634-7939 Fax: 301-634-7990 Email:
Submitted to: Re: Comments on CMS Proposals for Patient Condition Groups and Care Episode Groups
 April 24, 2017 The Honorable Seema Verma Administrator Centers for Medicare & Medicaid Services Hubert H. Humphrey Building 200 Independence Avenue SW Washington, D.C. 20201 Submitted to: macra-episode-based-cost-measures-info@acumenllc.com
April 24, 2017 The Honorable Seema Verma Administrator Centers for Medicare & Medicaid Services Hubert H. Humphrey Building 200 Independence Avenue SW Washington, D.C. 20201 Submitted to: macra-episode-based-cost-measures-info@acumenllc.com
January 16, Dear Administrator Verma:
 Ms. Seema Verma Administrator Centers for Medicare and Medicaid Services Department of Health and Human Services Attention: CMMI New Direction P.O. Box 8011 Baltimore, MD 21244-1850 Re: (CMS-4182-P) Medicare
Ms. Seema Verma Administrator Centers for Medicare and Medicaid Services Department of Health and Human Services Attention: CMMI New Direction P.O. Box 8011 Baltimore, MD 21244-1850 Re: (CMS-4182-P) Medicare
Assessment and Management of Genetic Predisposition to Breast Cancer. Dr Munaza Ahmed Consultant Clinical Geneticist 2/7/18
 Assessment and Management of Genetic Predisposition to Breast Cancer Dr Munaza Ahmed Consultant Clinical Geneticist 2/7/18 Overview The role of the Cancer Genetics team NICE guidelines for Familial Breast
Assessment and Management of Genetic Predisposition to Breast Cancer Dr Munaza Ahmed Consultant Clinical Geneticist 2/7/18 Overview The role of the Cancer Genetics team NICE guidelines for Familial Breast
Calendar Year (CY) 2016 Clinical Laboratory Fee Schedule (CLFS) Preliminary Determinations
 Calendar Year (CY) 2016 Clinical Laboratory Fee Schedule (CLFS) Preliminary Determinations A. Drug Testing Current coding for testing for drugs of abuse relies on a structure of screening (known as presumptive
Calendar Year (CY) 2016 Clinical Laboratory Fee Schedule (CLFS) Preliminary Determinations A. Drug Testing Current coding for testing for drugs of abuse relies on a structure of screening (known as presumptive
November 19, Dear Messrs. Holdren and Lander:
 John P. Holdren, Co-Chair Eric Lander, Co-Chair President s Council of Advisors on Science and Technology Executive Office of the President 1650 Pennsylvania Avenue, NW Washington, DC 20504 Dear Messrs.
John P. Holdren, Co-Chair Eric Lander, Co-Chair President s Council of Advisors on Science and Technology Executive Office of the President 1650 Pennsylvania Avenue, NW Washington, DC 20504 Dear Messrs.
Genetic testing for hereditary cancer
 Genetic testing for hereditary cancer THE GENETICS OF HEREDITARY CANCER About half of all men and one-third of all women in the US will develop cancer during their lifetimes. Approximately 5% to 10% of
Genetic testing for hereditary cancer THE GENETICS OF HEREDITARY CANCER About half of all men and one-third of all women in the US will develop cancer during their lifetimes. Approximately 5% to 10% of
Invasive Prenatal (Fetal) Diagnostic Testing
 Protocol Invasive Prenatal (Fetal) Diagnostic Testing (204116) Medical Benefit Effective Date: 04/01/18 Next Review Date: 01/19 Preauthorization Yes Review Dates: 01/15, 01/16, 01/17, 01/18 Preauthorization
Protocol Invasive Prenatal (Fetal) Diagnostic Testing (204116) Medical Benefit Effective Date: 04/01/18 Next Review Date: 01/19 Preauthorization Yes Review Dates: 01/15, 01/16, 01/17, 01/18 Preauthorization
COLON CANCER & GENETICS VERMONT COLORECTAL CANCER SUMMIT NOVEMBER 15, 2014
 COLON CANCER & GENETICS VERMONT COLORECTAL CANCER SUMMIT NOVEMBER 15, 2014 WENDY MCKINNON, MS, CGC CERTIFIED GENETIC COUNSELOR FAMILIAL CANCER PROGRAM UNIVERSIT Y OF VERMONT MEDICAL CENTER 1 CHARACTERISTICS
COLON CANCER & GENETICS VERMONT COLORECTAL CANCER SUMMIT NOVEMBER 15, 2014 WENDY MCKINNON, MS, CGC CERTIFIED GENETIC COUNSELOR FAMILIAL CANCER PROGRAM UNIVERSIT Y OF VERMONT MEDICAL CENTER 1 CHARACTERISTICS
August 17, Dear Valued Client:
 August 7, 08 Re: CMS Announces 6-Month Period of Enforcement Discretion for Laboratory Date of Service Exception Policy Under the Medicare Clinical Laboratory Fee Schedule (the 4 Day Rule ) Dear Valued
August 7, 08 Re: CMS Announces 6-Month Period of Enforcement Discretion for Laboratory Date of Service Exception Policy Under the Medicare Clinical Laboratory Fee Schedule (the 4 Day Rule ) Dear Valued
CASE STUDY. Germline Cancer Testing in A Proband: Extension of Benefits to Unaffected Family Members. Introduction. Patient Profile.
 CASE STUDY Germline Cancer Testing in A Proband: Extension of Benefits to Unaffected Family Members Introduction Most cancers are caused by genetic damage resulting from random mutations and exposure to
CASE STUDY Germline Cancer Testing in A Proband: Extension of Benefits to Unaffected Family Members Introduction Most cancers are caused by genetic damage resulting from random mutations and exposure to
Inaccuracies in CMS Interim Final RVUs for Intracranial Endovascular Intervention Codes
 Andy Slavitt, Acting Administrator Centers for Medicare & Medicaid Services Department of Health and Human Services Attention: Room 445-G, Hubert H. Humphrey Building 200 Independence Avenue, SW Washington,
Andy Slavitt, Acting Administrator Centers for Medicare & Medicaid Services Department of Health and Human Services Attention: Room 445-G, Hubert H. Humphrey Building 200 Independence Avenue, SW Washington,
Medical Policy. MP Invasive Prenatal (Fetal) Diagnostic Testing
 Medical Policy MP 2.04.116 BCBSA Ref. Policy: 2.04.116 Last Review: 04/30/2018 Effective Date: 04/30/2018 Section: Medicine Related Policies 2.04.59 Genetic Testing for Developmental Delay/Intellectual
Medical Policy MP 2.04.116 BCBSA Ref. Policy: 2.04.116 Last Review: 04/30/2018 Effective Date: 04/30/2018 Section: Medicine Related Policies 2.04.59 Genetic Testing for Developmental Delay/Intellectual
A guide to genetic testing for hereditary cancers
 Cancer Testing Solutions A guide to genetic testing for hereditary cancers The benefit of knowing TM Hereditary cancer genetic testing can play a critical role in managing health Cancer touches millions
Cancer Testing Solutions A guide to genetic testing for hereditary cancers The benefit of knowing TM Hereditary cancer genetic testing can play a critical role in managing health Cancer touches millions
Re: HIPAA Administrative Simplification: Modification to Medical Data Code Set Standards to Adopt ICD-10-CM and ICD-10-PCS
 October 20, 2008 The Honorable Michael O. Leavitt Secretary U.S. Department of Health and Human Services Room 445-G Hubert H. Humphrey Building 200 Independence Avenue, SW Washington, DC 20201 Re: HIPAA
October 20, 2008 The Honorable Michael O. Leavitt Secretary U.S. Department of Health and Human Services Room 445-G Hubert H. Humphrey Building 200 Independence Avenue, SW Washington, DC 20201 Re: HIPAA
Germline Testing for Hereditary Cancer with Multigene Panel
 Germline Testing for Hereditary Cancer with Multigene Panel Po-Han Lin, MD Department of Medical Genetics National Taiwan University Hospital 2017-04-20 Disclosure No relevant financial relationships with
Germline Testing for Hereditary Cancer with Multigene Panel Po-Han Lin, MD Department of Medical Genetics National Taiwan University Hospital 2017-04-20 Disclosure No relevant financial relationships with
Expert Interview: Inherited Susceptibility to Cancer with Dr. Nicoleta Voian
 Expert Interview: Inherited Susceptibility to Cancer with Dr. Nicoleta Voian ANNOUNCER OPEN: Welcome to CME on ReachMD. This segment, entitled Inherited Susceptibility to Cancer: What Do Primary Care Providers
Expert Interview: Inherited Susceptibility to Cancer with Dr. Nicoleta Voian ANNOUNCER OPEN: Welcome to CME on ReachMD. This segment, entitled Inherited Susceptibility to Cancer: What Do Primary Care Providers
Re: Docket No. FDA D Presenting Risk Information in Prescription Drug and Medical Device Promotion
 1201 Maryland Avenue SW, Suite 900, Washington, DC 20024 202-962-9200, www.bio.org August 25, 2009 Dockets Management Branch (HFA-305) Food and Drug Administration 5600 Fishers Lane, Rm. 1061 Rockville,
1201 Maryland Avenue SW, Suite 900, Washington, DC 20024 202-962-9200, www.bio.org August 25, 2009 Dockets Management Branch (HFA-305) Food and Drug Administration 5600 Fishers Lane, Rm. 1061 Rockville,
Information for You and Your Family
 Information for You and Your Family What is Prevention? Cancer prevention is action taken to lower the chance of getting cancer. In 2017, more than 1.6 million people will be diagnosed with cancer in the
Information for You and Your Family What is Prevention? Cancer prevention is action taken to lower the chance of getting cancer. In 2017, more than 1.6 million people will be diagnosed with cancer in the
Proactive Patient Paves the Way for Genetic Testing of Eight Family Members
 CASE STUDY Proactive Patient Paves the Way for Genetic Testing of Eight Family Members Quick Summary Samar Mohite * was diagnosed with colon adenocarcinoma at the age of 49 years. Genetic counselling was
CASE STUDY Proactive Patient Paves the Way for Genetic Testing of Eight Family Members Quick Summary Samar Mohite * was diagnosed with colon adenocarcinoma at the age of 49 years. Genetic counselling was
Genomics in Public Health Vision and Goals for the Population Screening Working Group
 Genomics in Public Health Vision and Goals for the Population Screening Working Group Mike Murray Jim Evans Co-chairs; IOM Action Collaborative 24 February 2017 Washington DC Intent of IOM Action Collaborative
Genomics in Public Health Vision and Goals for the Population Screening Working Group Mike Murray Jim Evans Co-chairs; IOM Action Collaborative 24 February 2017 Washington DC Intent of IOM Action Collaborative
American Imaging Management, Inc.
 STRIDE sm (HMO) MEDICARE ADVANTAGE Molecular Diagnostic Testing Prior Authorization Information in this policy only applies to Harvard Pilgrim Stride (HMO) and Stride of New Hampshire (HMO) members. Overview
STRIDE sm (HMO) MEDICARE ADVANTAGE Molecular Diagnostic Testing Prior Authorization Information in this policy only applies to Harvard Pilgrim Stride (HMO) and Stride of New Hampshire (HMO) members. Overview
2018 Edition The Current Landscape of Genetic Testing
 2018 Edition The Current Landscape of Genetic Testing Market growth, reimbursement trends, challenges and opportunities November April 20182017 EXECUTIVE SUMMARY Concert Genetics is a software and managed
2018 Edition The Current Landscape of Genetic Testing Market growth, reimbursement trends, challenges and opportunities November April 20182017 EXECUTIVE SUMMARY Concert Genetics is a software and managed
PROVIDER PROCEDURE CODES EVICORE
 CARDIOLOGY/RADIOLOGY PROVIDER PROCEDURE CODES EVICORE CPT CODE CPT CODE DESCRIPTION 33206 Insertion of new or replacement of permanent pacemaker with transvenous electrode(s); atrial 33207 Insertion of
CARDIOLOGY/RADIOLOGY PROVIDER PROCEDURE CODES EVICORE CPT CODE CPT CODE DESCRIPTION 33206 Insertion of new or replacement of permanent pacemaker with transvenous electrode(s); atrial 33207 Insertion of
Cancer Survivorship Symposium Cancer and Heredity January 16, Jeanne P. Homer, MS Licensed Certified Genetic Counselor
 Cancer Survivorship Symposium Cancer and Heredity January 16, 2017 Jeanne P. Homer, MS Licensed Certified Genetic Counselor Outline Cancer and Heredity Hereditary Cancer Risk Assessment & Genetic testing
Cancer Survivorship Symposium Cancer and Heredity January 16, 2017 Jeanne P. Homer, MS Licensed Certified Genetic Counselor Outline Cancer and Heredity Hereditary Cancer Risk Assessment & Genetic testing
PROVIDER PROCEDURE CODES EVICORE
 CARDIOLOGY/RADIOLOGY PROVIDER PROCEDURE CODES EVICORE CPT CODE CPT CODE DESCRIPTION 33206 Insertion of new or replacement of permanent pacemaker with transvenous electrode(s); atrial 33207 Insertion of
CARDIOLOGY/RADIOLOGY PROVIDER PROCEDURE CODES EVICORE CPT CODE CPT CODE DESCRIPTION 33206 Insertion of new or replacement of permanent pacemaker with transvenous electrode(s); atrial 33207 Insertion of
Why Test for Hereditary Cancer in Preventive Care?
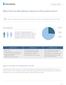 Why Test for Hereditary Cancer in Preventive Care? Millions of people are sidelined by cancer. Wouldn't it be worth it for your patients to know their risk? background HEREDITARY (5-10%) More than 1 in
Why Test for Hereditary Cancer in Preventive Care? Millions of people are sidelined by cancer. Wouldn't it be worth it for your patients to know their risk? background HEREDITARY (5-10%) More than 1 in
Using the Bravo Liquid-Handling System for Next Generation Sequencing Sample Prep
 Using the Bravo Liquid-Handling System for Next Generation Sequencing Sample Prep Tom Walsh, PhD Division of Medical Genetics University of Washington Next generation sequencing Sanger sequencing gold
Using the Bravo Liquid-Handling System for Next Generation Sequencing Sample Prep Tom Walsh, PhD Division of Medical Genetics University of Washington Next generation sequencing Sanger sequencing gold
Genetics and Cancer Care. Cynthia Forster-Gibson, MD, PhD and Loren Mackay- Loder, MSc Genetics Program, THP
 Genetics and Cancer Care Cynthia Forster-Gibson, MD, PhD and Loren Mackay- Loder, MSc Genetics Program, THP Faculty/Presenter Disclosure Faculty: Cynthia Forster-Gibson Relationships with commercial interests:
Genetics and Cancer Care Cynthia Forster-Gibson, MD, PhD and Loren Mackay- Loder, MSc Genetics Program, THP Faculty/Presenter Disclosure Faculty: Cynthia Forster-Gibson Relationships with commercial interests:
GENERAL COMMENTS. The Task Force Process Should be Fully Open, Balanced and Transparent
 December 9, 2013 Submitted Electronically United States Preventive Services Task Force c/o Dr. Robert Cosby Agency for Healthcare Research and Quality 540 Gaither Road Rockville, MD 20850 RE: USPSTF Draft
December 9, 2013 Submitted Electronically United States Preventive Services Task Force c/o Dr. Robert Cosby Agency for Healthcare Research and Quality 540 Gaither Road Rockville, MD 20850 RE: USPSTF Draft
Re: Episode-Based Cost Measure Development for the Quality Payment Program
 April 24 th, 2017 SUBMITTED VIA EMAIL macra-episode-based-cost-measuresinfo@acumenllc.com Seema Verma Administrator Centers for Medicare and Medicaid Services U.S. Department of Health and Human Services
April 24 th, 2017 SUBMITTED VIA EMAIL macra-episode-based-cost-measuresinfo@acumenllc.com Seema Verma Administrator Centers for Medicare and Medicaid Services U.S. Department of Health and Human Services
The following codes are under management for members who have health benefits covered by WellCare, administered by evicore healthcare.
 WellCare The following codes are under management for members who have health benefits covered by WellCare, administered by evicore healthcare. 81162 81163 81164 cancer) gene analysis; full sequence analysis
WellCare The following codes are under management for members who have health benefits covered by WellCare, administered by evicore healthcare. 81162 81163 81164 cancer) gene analysis; full sequence analysis
Hereditary Gastric Cancer
 Hereditary Gastric Cancer Dr Bastiaan de Boer Consultant Pathologist Department of Anatomical Pathology PathWest Laboratory Medicine, QE II Medical Centre Clinical Associate Professor School of Pathology
Hereditary Gastric Cancer Dr Bastiaan de Boer Consultant Pathologist Department of Anatomical Pathology PathWest Laboratory Medicine, QE II Medical Centre Clinical Associate Professor School of Pathology
Be Ready Pack Learn more about how Myriad myrisk is revolutionizing hereditary cancer testing.
 Be Ready Pack Learn more about how Myriad myrisk is revolutionizing hereditary cancer testing. Hereditary cancer and you Approximately 5% to 10% of all cancers develop because a person inherited a genetic
Be Ready Pack Learn more about how Myriad myrisk is revolutionizing hereditary cancer testing. Hereditary cancer and you Approximately 5% to 10% of all cancers develop because a person inherited a genetic
GENETIC TESTING FOR PREDICTING RISK OF NONFAMILIAL BREAST CANCER
 Non-Discrimination Statement and Multi-Language Interpreter Services information are located at the end of this document. Coverage for services, procedures, medical devices and drugs are dependent upon
Non-Discrimination Statement and Multi-Language Interpreter Services information are located at the end of this document. Coverage for services, procedures, medical devices and drugs are dependent upon
Genetic testing and pancreatic disease
 Genetic testing and pancreatic disease February 2 d, 2018 Yale Pancreas Symposium 2018: Multidisciplinary Management of Pancreatic Cancer Xavier Llor, M.D., PhD. Associate Professor of Medicine Co-Director,
Genetic testing and pancreatic disease February 2 d, 2018 Yale Pancreas Symposium 2018: Multidisciplinary Management of Pancreatic Cancer Xavier Llor, M.D., PhD. Associate Professor of Medicine Co-Director,
Genetic Determinants, Risk Assessment and Management
 Genetic Determinants, Risk Assessment and Management Rachel Rando, MS, CGC Genetic Counselor Hunterdon Regional Cancer Center Flemington, NJ I have no disclosures. Acknowledgements: Staff of Hunterdon
Genetic Determinants, Risk Assessment and Management Rachel Rando, MS, CGC Genetic Counselor Hunterdon Regional Cancer Center Flemington, NJ I have no disclosures. Acknowledgements: Staff of Hunterdon
Molecular Diagnostic Testing Prior Authorization
 Referral, Notification, and Authorization Authorization Molecular Diagnostic Testing Prior Authorization Unless otherwise specified, information in this policy does not apply to members with the Choice
Referral, Notification, and Authorization Authorization Molecular Diagnostic Testing Prior Authorization Unless otherwise specified, information in this policy does not apply to members with the Choice
May 16, Division of Dockets Management (HFA-305) Food and Drug Administration 5630 Fishers Lane, Room 1061 Rockville, MD 20852
 701 Pennsylvania Avenue, NW Suite 800 Washington, D.C. 20004 2654 Tel: 202 783 8700 Fax: 202 783 8750 www.advamed.org May 16, 2014 Division of Dockets Management (HFA-305) Food and Drug Administration
701 Pennsylvania Avenue, NW Suite 800 Washington, D.C. 20004 2654 Tel: 202 783 8700 Fax: 202 783 8750 www.advamed.org May 16, 2014 Division of Dockets Management (HFA-305) Food and Drug Administration
Hereditary Cancer Products
 Hereditary Products Integrated BRACAnalysis (BRCA1 and BRCA2 sequencing and large rearrangement testing (BART)), covered when: PERSONAL HISTORY of Breast (must meet at least 1) Diagnosed
Hereditary Products Integrated BRACAnalysis (BRCA1 and BRCA2 sequencing and large rearrangement testing (BART)), covered when: PERSONAL HISTORY of Breast (must meet at least 1) Diagnosed
Clinical Cancer Genetics
 Clinical Cancer Genetics Lisen Axell, MS, CGC University of Colorado Cancer Center Individuals with cancer Making surgical decisions (lump vs. mast) Making treatment decisions (XRT) Concerns for additional
Clinical Cancer Genetics Lisen Axell, MS, CGC University of Colorado Cancer Center Individuals with cancer Making surgical decisions (lump vs. mast) Making treatment decisions (XRT) Concerns for additional
PROVIDER POLICIES & PROCEDURES
 PROVIDER POLICIES & PROCEDURES BRCA GENETIC TESTING The purpose of this document is to assist providers enrolled in the Connecticut Medical Assistance Program (CMAP) with the information needed to support
PROVIDER POLICIES & PROCEDURES BRCA GENETIC TESTING The purpose of this document is to assist providers enrolled in the Connecticut Medical Assistance Program (CMAP) with the information needed to support
HHA Medical Products and Services Requiring Preauthorization
 12 21 31 34 51 53 54 55 56 61 Place of Service Place of Service Place of Service Place of Service Place of Service Place of Service Place of Service Place of Service Place of Service Place of Service 43644
12 21 31 34 51 53 54 55 56 61 Place of Service Place of Service Place of Service Place of Service Place of Service Place of Service Place of Service Place of Service Place of Service Place of Service 43644
A Patient s Guide to Hereditary Cancer. Is Hereditary Cancer Testing Right for You?
 A Patient s Guide to Hereditary Cancer Is Hereditary Cancer Testing Right for You? What is Hereditary Cancer? Most cancers occur in people who do not have a strong family history of that cancer. This is
A Patient s Guide to Hereditary Cancer Is Hereditary Cancer Testing Right for You? What is Hereditary Cancer? Most cancers occur in people who do not have a strong family history of that cancer. This is
CANCER GENETICS PROVIDER SURVEY
 Dear Participant, Previously you agreed to participate in an evaluation of an education program we developed for primary care providers on the topic of cancer genetics. This is an IRB-approved, CDCfunded
Dear Participant, Previously you agreed to participate in an evaluation of an education program we developed for primary care providers on the topic of cancer genetics. This is an IRB-approved, CDCfunded
Lynch Syndrome. Angie Strang, PGY2
 Lynch Syndrome Angie Strang, PGY2 Background Previously hereditary nonpolyposis colorectal cancer Autosomal dominant inherited cancer susceptibility syndrome Caused by defects in the mismatch repair system
Lynch Syndrome Angie Strang, PGY2 Background Previously hereditary nonpolyposis colorectal cancer Autosomal dominant inherited cancer susceptibility syndrome Caused by defects in the mismatch repair system
December 18, Submitted Electronically
 December 18, 2017 Seema Verma, Administrator Centers for Medicare & Medicaid Services Department of Health and Human Services Attn: CMS-4182-P PO Box 8013 Baltimore, MD 21244-8013 Submitted Electronically
December 18, 2017 Seema Verma, Administrator Centers for Medicare & Medicaid Services Department of Health and Human Services Attn: CMS-4182-P PO Box 8013 Baltimore, MD 21244-8013 Submitted Electronically
Myriad Financial Assistance Program (MFAP)
 Myriad Financial Assistance Program (MFAP) MEDICAL CRITERIA Hereditary Cancer Products The Myriad Financial Assistance Program offers aid to patients who meet specific financial and medical requirements.
Myriad Financial Assistance Program (MFAP) MEDICAL CRITERIA Hereditary Cancer Products The Myriad Financial Assistance Program offers aid to patients who meet specific financial and medical requirements.
CMS Issues New Draft Guidance on Coverage with Evidence Development Policy for National Coverage Determinations
 December 6, 2012 CMS Issues New Draft Guidance on Coverage with Evidence Development Policy for National Coverage Determinations For more information, contact: Seth H. Lundy +1 202 626 2924 slundy@kslaw.com
December 6, 2012 CMS Issues New Draft Guidance on Coverage with Evidence Development Policy for National Coverage Determinations For more information, contact: Seth H. Lundy +1 202 626 2924 slundy@kslaw.com
GENETIC TESTING FOR HEREDITARY CANCER
 UnitedHealthcare Oxford Clinical Policy GENETIC TESTING FOR HEREDITARY CANCER Policy Number: DIAGNOSTIC 004.27 T2 Effective Date: November 1, 2017 Table of Contents Page INSTRUCTIONS FOR USE... 1 CONDITIONS
UnitedHealthcare Oxford Clinical Policy GENETIC TESTING FOR HEREDITARY CANCER Policy Number: DIAGNOSTIC 004.27 T2 Effective Date: November 1, 2017 Table of Contents Page INSTRUCTIONS FOR USE... 1 CONDITIONS
December 13, The Future Reimbursement Environment for NGS for Oncology
 WELCOME! Today s Speaker Mr. Mathews has worked on a variety of diagnostic reimbursement projects involving the analysis, development, and implementation of coding, coverage, and payment strategies for
WELCOME! Today s Speaker Mr. Mathews has worked on a variety of diagnostic reimbursement projects involving the analysis, development, and implementation of coding, coverage, and payment strategies for
MP Genetic Testing for Developmental Delay/Intellectual Disability, Autism Spectrum Disorder, and Congenital Anomalies
 Medical Policy Genetic Testing for Developmental Delay/Intellectual Disability, Autism Spectrum Disorder, and Congenital BCBSA Ref. Policy: 2.04.59 Last Review: 10/18/2018 Effective Date: 10/18/2018 Section:
Medical Policy Genetic Testing for Developmental Delay/Intellectual Disability, Autism Spectrum Disorder, and Congenital BCBSA Ref. Policy: 2.04.59 Last Review: 10/18/2018 Effective Date: 10/18/2018 Section:
TGL clinical User Guide
 TGL clinical User Guide The Institute of Cancer Research Brookes Lawley Building 15 Cotswold Road Sutton, Surrey SM2 5NG Page 1 of 7 TGL clinical This document describes the services provided by TGL clinical
TGL clinical User Guide The Institute of Cancer Research Brookes Lawley Building 15 Cotswold Road Sutton, Surrey SM2 5NG Page 1 of 7 TGL clinical This document describes the services provided by TGL clinical
DNA Basics. We are all made up of cells. Cells contain DNA, or instructions to tell our bodies how to work.
 DNA Basics We are all made up of cells. Cells contain DNA, or instructions to tell our bodies how to work. DNA is packaged into structures called chromosomes. Each chromosome contains many genes and each
DNA Basics We are all made up of cells. Cells contain DNA, or instructions to tell our bodies how to work. DNA is packaged into structures called chromosomes. Each chromosome contains many genes and each
Genetic Testing for Familial Gastrointestinal Cancer Syndromes. C. Richard Boland, MD La Jolla, CA January 21, 2017
 Genetic Testing for Familial Gastrointestinal Cancer Syndromes C. Richard Boland, MD La Jolla, CA January 21, 2017 Disclosure Information C. Richard Boland, MD I have no financial relationships to disclose.
Genetic Testing for Familial Gastrointestinal Cancer Syndromes C. Richard Boland, MD La Jolla, CA January 21, 2017 Disclosure Information C. Richard Boland, MD I have no financial relationships to disclose.
HIGHLIGHTS OF THE 2017 PROPOSED MEDICARE PHYSICIAN FEE SCHEDULE RULE
 HIGHLIGHTS OF THE 2017 PROPOSED MEDICARE PHYSICIAN FEE SCHEDULE RULE July 13, 2016 INTRODUCTION The Medicare Physician Fee Schedule proposed rule released by the Centers for Medicare and Medicaid Services
HIGHLIGHTS OF THE 2017 PROPOSED MEDICARE PHYSICIAN FEE SCHEDULE RULE July 13, 2016 INTRODUCTION The Medicare Physician Fee Schedule proposed rule released by the Centers for Medicare and Medicaid Services
Precision Medicine. Wendy Chung, MD PhD Director of Clinical Genetics Columbia University
 Precision Medicine Wendy Chung, MD PhD Director of Clinical Genetics Columbia University What is precision medicine? Providing the right treatment for the right patient at the right time and engaging the
Precision Medicine Wendy Chung, MD PhD Director of Clinical Genetics Columbia University What is precision medicine? Providing the right treatment for the right patient at the right time and engaging the
Hereditary Breast and Ovarian Cancer Rebecca Sutphen, MD, FACMG
 Hereditary Breast and Ovarian Cancer 2015 Rebecca Sutphen, MD, FACMG Among a consecutive series of 11,159 women requesting BRCA testing over one year, 3874 responded to a mailed survey. Most respondents
Hereditary Breast and Ovarian Cancer 2015 Rebecca Sutphen, MD, FACMG Among a consecutive series of 11,159 women requesting BRCA testing over one year, 3874 responded to a mailed survey. Most respondents
The Role of Genetics in Ovarian Cancer Screening. Dawn DeLozier, Ph.D. Medical Geneticist, SAMC Associate Professor UCSF-Fresno
 The Role of Genetics in Ovarian Cancer Screening Dawn DeLozier, Ph.D. Medical Geneticist, SAMC Associate Professor UCSF-Fresno How Much Breast and Ovarian Cancer Is Hereditary? 15%-20% 5%-10% Breast Cancer
The Role of Genetics in Ovarian Cancer Screening Dawn DeLozier, Ph.D. Medical Geneticist, SAMC Associate Professor UCSF-Fresno How Much Breast and Ovarian Cancer Is Hereditary? 15%-20% 5%-10% Breast Cancer
GENETIC TESTING FOR HEREDITARY BREAST AND OVARIAN CANCER SYNDROME BRCA1 BRCA2
 SYNDROME BRCA1 BRCA2 Non-Discrimination Statement and Multi-Language Interpreter Services information are located at the end of this document. Coverage for services, procedures, medical devices and drugs
SYNDROME BRCA1 BRCA2 Non-Discrimination Statement and Multi-Language Interpreter Services information are located at the end of this document. Coverage for services, procedures, medical devices and drugs
TumorNext-HRD with OvaNext: Paired Germline and Tumor Analyses of Genes Involved in
 SAMPLE REPORT Ordered By Contact ID:1251298 Example, Doctor, MD MOCKORG44 (10829) 123 Somewhere LaneSuite 4 Heaven NV 78872 US Ph:123-123-1234 Fx:123-123-1223 Org ID:8141 Normal Specimen Accession #: 00-086947
SAMPLE REPORT Ordered By Contact ID:1251298 Example, Doctor, MD MOCKORG44 (10829) 123 Somewhere LaneSuite 4 Heaven NV 78872 US Ph:123-123-1234 Fx:123-123-1223 Org ID:8141 Normal Specimen Accession #: 00-086947
Medicare Program: Changes to Hospital Outpatient Prospective Payment and Ambulatory Surgical Center Payment Systems and Quality Reporting Programs
 Ms. Seema Verma Administrator Centers for Medicare and Medicaid Services US Department of Health and Human Services Attention: CMS-1695-FC P.O. Box 8013, 7500 Security Boulevard Baltimore, MD 21244-1850
Ms. Seema Verma Administrator Centers for Medicare and Medicaid Services US Department of Health and Human Services Attention: CMS-1695-FC P.O. Box 8013, 7500 Security Boulevard Baltimore, MD 21244-1850
Background USPSTF Guideline
 July 26, 2016 The Honorable Sylvia M. Burwell The Honorable Tom Perez Secretary, Department of Health and Human Services Secretary, Department of Labor 200 Independence Ave SW 200 Constitution Avenue,
July 26, 2016 The Honorable Sylvia M. Burwell The Honorable Tom Perez Secretary, Department of Health and Human Services Secretary, Department of Labor 200 Independence Ave SW 200 Constitution Avenue,
Policy Specific Section: Medical Necessity and Investigational / Experimental. October 14, 1998 March 28, 2014
 Medical Policy Genetic Testing for Colorectal Cancer Type: Medical Necessity and Investigational / Experimental Policy Specific Section: Laboratory/Pathology Original Policy Date: Effective Date: October
Medical Policy Genetic Testing for Colorectal Cancer Type: Medical Necessity and Investigational / Experimental Policy Specific Section: Laboratory/Pathology Original Policy Date: Effective Date: October
Recommendation 1: Promote Kidney Disease Prevention Research
 April 30, 2013 David M. Murray, PhD Office of Disease Prevention National Institutes of Health 6100 Executive Blvd., Room 2B03, MSC 7523 Bethesda, MD 20892-7523 Dear Dr. Murray: On behalf of the American
April 30, 2013 David M. Murray, PhD Office of Disease Prevention National Institutes of Health 6100 Executive Blvd., Room 2B03, MSC 7523 Bethesda, MD 20892-7523 Dear Dr. Murray: On behalf of the American
1201 Maryland Avenue SW Suite 900 Washington, DC
 June 14, 2012 Lonnie King, D.V.M. Chair Committee on Identifying and Prioritizing New Preventive Vaccines for Development Institute of Medicine 500 Fifth Street, NW Washington, DC 20001 Re: Ranking Vaccines:
June 14, 2012 Lonnie King, D.V.M. Chair Committee on Identifying and Prioritizing New Preventive Vaccines for Development Institute of Medicine 500 Fifth Street, NW Washington, DC 20001 Re: Ranking Vaccines:
Personalised cancer care Information for Medical Specialists. A new way to unlock treatment options for your patients
 Personalised cancer care Information for Medical Specialists A new way to unlock treatment options for your patients Contents Optimised for clinical benefit 4 Development history 4 Full FIND IT panel vs
Personalised cancer care Information for Medical Specialists A new way to unlock treatment options for your patients Contents Optimised for clinical benefit 4 Development history 4 Full FIND IT panel vs
1-Appropriate Use Criteria for Advanced Diagnostic Imaging Services
 The Honorable Seema Verma, Administrator Centers for Medicare and Medicaid Services Attention: CMS 1676 P P.O. Box 8016 Baltimore, MD 21244 1850 Dear Administrator Verma: September 10, 2017 On behalf of
The Honorable Seema Verma, Administrator Centers for Medicare and Medicaid Services Attention: CMS 1676 P P.O. Box 8016 Baltimore, MD 21244 1850 Dear Administrator Verma: September 10, 2017 On behalf of
Hereditary Cancer Update Strengthening Linkages Workshop April 22, 2017
 Hereditary Cancer Update Strengthening Linkages Workshop April 22, 2017 Renée Perrier, MD MSc FRCPC Clinical Assistant Professor University of Calgary, Department of Medical Genetics Medical Director,
Hereditary Cancer Update Strengthening Linkages Workshop April 22, 2017 Renée Perrier, MD MSc FRCPC Clinical Assistant Professor University of Calgary, Department of Medical Genetics Medical Director,
OvaNext. patient guide. genetic testing for hereditary breast, ovarian, and uterine cancer
 patient guide OvaNext genetic testing for hereditary breast, ovarian, and uterine cancer Because knowing your risk can mean early detection and prevention About half of the women diagnosed with uterine
patient guide OvaNext genetic testing for hereditary breast, ovarian, and uterine cancer Because knowing your risk can mean early detection and prevention About half of the women diagnosed with uterine
Carol Christianson, MS, CGC Genetic Counselor West Michigan Cancer Center
 Carol Christianson, MS, CGC Genetic Counselor West Michigan Cancer Center Following this presentation you will be able to: Identify cancer survivors in your practice who might benefit from genetic counseling
Carol Christianson, MS, CGC Genetic Counselor West Michigan Cancer Center Following this presentation you will be able to: Identify cancer survivors in your practice who might benefit from genetic counseling
Cervicocerebral Angiography Codes CPT Code Shortened Descriptor RUC Rec RVU CMS Interim RVU
 AMERICAN ASSOCIATION OF NEUROLOGICAL SURGEONS THOMAS A. MARSHALL, Executive Director 5550 Meadowbrook Drive Rolling Meadows, IL 60008 Phone: 888-566-AANS Fax: 847-378-0600 info@aans.org President MITCHEL
AMERICAN ASSOCIATION OF NEUROLOGICAL SURGEONS THOMAS A. MARSHALL, Executive Director 5550 Meadowbrook Drive Rolling Meadows, IL 60008 Phone: 888-566-AANS Fax: 847-378-0600 info@aans.org President MITCHEL
Genetics/Genomics: role of genes in diagnosis and/risk and in personalised medicine
 Genetics/Genomics: role of genes in diagnosis and/risk and in personalised medicine Lynn Greenhalgh, Macmillan Cancer and General Clinical Geneticist Cancer Genetics Service Cancer is common 1 in
Genetics/Genomics: role of genes in diagnosis and/risk and in personalised medicine Lynn Greenhalgh, Macmillan Cancer and General Clinical Geneticist Cancer Genetics Service Cancer is common 1 in
