It is advisable to refer to the publisher s version if you intend to cite from the work.
|
|
|
- Bernadette Shaw
- 5 years ago
- Views:
Transcription
1 Beta event related desynchronization as an index of individual differences in processing human facial expression: further investigations of autistic traits in typically developing adults Article Accepted Version Open Access Cooper, N. R., Simpson, A., Till, A., Simmons, K. and Puzzo, I. (2013) Beta event related desynchronization as an index of individual differences in processing human facial expression: further investigations of autistic traits in typically developing adults. Frontiers in Human Neuroscience, ISSN doi: Available at It is advisable to refer to the publisher s version if you intend to cite from the work. To link to this article DOI: Publisher: Frontiers All outputs in CentAUR are protected by Intellectual Property Rights law, including copyright law. Copyright and IPR is retained by the creators or other
2 copyright holders. Terms and conditions for use of this material are defined in the End User Agreement. CentAUR Central Archive at the University of Reading Reading s research outputs online
3 IN HUMAN NEUROSCIENCE Beta event-related desynchronization as an index of individual differences in processing human facial expression: further investigations of autistic traits in typically developing adults. Nicholas Robert Cooper, Andrew Simpson, Amy Till, Kelly Simmons and Ignazio Puzzo Journal Name: Frontiers in Human Neuroscience ISSN: Article type: Original Research Article Received on: 13 Feb 2013 Accepted on: 10 Apr 2013 Provisional PDF published on: 10 Apr 2013 Frontiers website link: Citation: Article URL: Cooper NR, Simpson A, Till A, Simmons K and Puzzo I(2013) Beta event-related desynchronization as an index of individual differences in processing human facial expression: further investigations of autistic traits in typically developing adults.. Front. Hum. Neurosci. 7:159. doi: /fnhum name=human%20neuroscience&art_doi= /fnhum (If clicking on the link doesn't work, try copying and pasting it into your browser.) Copyright statement: 2013 Cooper, Simpson, Till, Simmons and Puzzo. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits use, distribution and reproduction in other forums, provided the original authors and source are credited and subject to any copyright notices concerning any third-party graphics etc. This Provisional PDF corresponds to the article as it appeared upon acceptance, after rigorous peer-review. Fully formatted PDF and full text (HTML) versions will be made available soon.
4 Beta event-related desynchronization as an index of individual differences in processing human facial expression: further investigations of autistic traits in typically developing adults. Nicholas R Cooper 1, 2, Andrew Simpson 2, Amy Till 2, Kelly Simmons 2 and Ignazio Puzzo 3 1 Corresponding author 2 Centre for Brain Science, Department of Psychology, University of Essex, Wivenhoe Park, Colchester CO4 3SQ, United Kingdom 3 Centre for Integrative Neuroscience and Neurodynamics, University of Reading, Whiteknights Campus (Earley Gate), Reading RG6 6AH, United Kingdom Key words: alpha, beta, mu, EEG, event-related desynchronization, ERD, autism, facial expression, emotion. Running title: mu-suppression to facial expression
5 Abstract The human mirror neuron system (hmns) has been associated with various forms of social cognition and affective processing including vicarious experience. It has also been proposed that a faulty hmns may underlie some of the deficits seen in the autism spectrum disorders. In the present study we set out to investigate whether emotional facial expressions could modulate a putative EEG index of hmns activation (mu suppression) and if so, would this differ according to the individual level of autistic traits (high versus low AQ score). Participants were presented with 3 second films of actors opening and closing their hands (classic hmns mu-suppression protocol) while simultaneously wearing happy, angry or neutral expressions. Musuppression was measured in the alpha and low beta bands. The low AQ group displayed greater low beta ERD to both angry and neutral expressions. The high AQ group displayed greater low beta ERD to angry than to happy expressions. There was also significantly more low beta ERD to happy faces for the low than for the high AQ group. In conclusion, an interesting interaction between AQ group and emotional expression revealed that hmns activation can be modulated by emotional facial expressions and that this is differentiated according to individual differences in the level of autistic traits. The EEG index of hmns activation (mu suppression) seems to be a sensitive measure of the variability in facial processing in typically developing individuals with high and low self reported traits of autism.
6 Introduction The study presented here was undertaken in order to examine the usefulness of measuring EEG sensorimotor reactivity to examine individual differences in emotional facial processing. For half a century, it has been known that suppression of the dominant resting rhythm in the EEG over sensorimotor areas accompanies not only movement execution but also movement observation (Gastaut, 1952; Gastaut & Bert, 1954). This rhythm, most commonly known as mu (but also referred to as the Rolandic or wicket rhythm) has two contributing bandwidths: an 8-12Hz component oscillating at alpha frequencies and a 12-20Hz low beta band component, perhaps reflecting contributions from primary somatosensory cortex and motor cortex respectively (Avanzini et al., 2012; Hari, 2006). A substantial amount of experimental work has established that movement execution is associated with suppression of the mu oscillatory activity over the sensorimotor cortex: at rest, the mu bandwidths show a synchronised activity, leading to high-amplitude oscillations. This synchronized activity is functionally distinguishable from the dominant occipital alpha activity. When a movement is executed, this synchronised activity is suppressed and this suppression is thought to reflect active processing in sensorimotor areas (Pfurtscheller & Lopes da Silva, 1999). Such suppression is often referred to as desynchronization or event-related desynchronization (ERD), particularly when it is measured in relation to a pre-stimulus baseline (or reference) period (Pfurtscheller & Aranibar, 1977). Gastaut and colleagues investigation of mu activity demonstrated that not only did mu desynchronise to movement execution but also to imagining and observing movements (Gastaut, 1952; Gastaut & Bert, 1954). The findings pertaining to movement observation were under-explored for several decades until the discovery of so-called mirror neurons in monkeys in the 1990s (Di Pellegrino, Fadiga, Fogassi, Gallese, & Rizzolatti, 1992; Rizzolatti, Fadiga, Gallese, & Fogassi, 1996). Research then turned to looking for human analogues of mirror neurons using various neuroimaging and other psychophysiological techniques. Mirror neurons were originally described as cells in monkey area F5 (an analog of the inferior frontal gyrus in humans and also later in parietal lobule) that fire not only when the
7 animal makes a specific movement but also when it observes that movement (Rizzolatti & Craighero, 2004). Work in humans using fmri (e.g. Iacoboni et al., 2005; Iacoboni et al., 1999; Molnar-Szakacs, Kaplan, Greenfield, & Iacoboni, 2006), transcranial magnetic stimulation (TMS; Enticott, Kennedy, Bradshaw, Rinehart, & Fitzgerald, 2010; Fadiga, Fogassi, Pavesi, & Rizzolatti, 1995; Sartori, Bucchioni, & Castiello, 2012), depth electrode recording (Mukamel, Ekstrom, Kaplan, Iacoboni, & Fried, 2010) and EEG/MEG (e.g. Hari et al., 1998; Kilner, Marchant, & Frith, 2009; Muthukumaraswamy & Johnson, 2004a; Muthukumaraswamy, Johnson, & McNair, 2004; Nishitani & Hari, 2000) have since shown the existence of a similar observation-execution matching system that has been labeled the human mirror neuron system (hmns) as this does not necessitate the existence of mirror neurons per se in humans, just a functionally similar mechanism. In this context, it is the EEG/MEG research that has drawn on the work of Gastaut and colleagues to explore the links between mu suppression and the hmns. Not only has mu-suppression been shown to be a useful indicator of action-observation pattern matching (in that suppression accompanies both action-execution and action-observation) but that it also closely matches other measures of the putative hmns. For instance, mu-suppression to the observation of hand movements has been shown to closely mirror fmri BOLD activation in areas analogous in humans to mirror neuron areas in primate studies (Perry & Bentin, 2009). In this context, mu-suppression has also been shown to be modulated by the laterality of the presentation stimulus (i.e. it is driven by the side of the screen on which an observed movement occurs), to be consistent with the reactivity of mirror neurons in area F5 in monkeys (Kilner et al., 2009) and to be dynamically modulated similarly in both action observation and action performance (Press, Cook, Blakemore, & Kilner, 2011). Accordingly, musuppression during action observation is interpreted as an index of activity in the hmns (Kilner et al., 2009; Pineda, 2005, 2008). Indeed, whereas until recently, mu-suppression during action-observation has been thought to result from post-synaptic modulation from mirror neurons in premotor cortex (Pineda, 2008; Rizzolatti & Craighero, 2004), recent evidence of so-called M1 view cells in primary motor cortex with mirror neuron-like properties (Dushanova & Donoghue, 2010) suggests that mu-suppression
8 may be a more direct measure of hmns than was previously believed, as M1 may itself be a part of the hmns (Press et al., 2011). The notion of a human mirror neuron system (hmns) has been used as an argument for the biological mechanisms underlying theories of embodied cognition such as simulation theory. Simulation theory posits that we understand the behaviours and emotions of others by activating similar neural processes in ourselves to those at play in the person observed (Gallese, 2009; Gallese & Goldman, 1998). This has been particularly investigated in relation to how we understand the facial expressions of others. Many studies have found fmri evidence for common neural activation during both the execution and perception of facial expressions, particularly in areas associated with the hmns (e.g. Carr, Iacoboni, Dubeau, Mazziotta, & Lenzi, 2003; Hennenlotter et al., 2005; Leslie, Johnson-Frey, & Grafton, 2004; van der Gaag, Minderaa, & Keysers, 2007). This has been strengthened by TMS studies showing that performance on a facial emotion processing task correlates with TMS-induced motor evoked potentials (thought to be an index of hmns activity; Enticott, Johnston, Herring, Hoy, & Fitzgerald, 2008) and that disrupting pre-sma activity with TMS impairs the recognition of happy faces (Rochas et al., 2012). To date, although it has been known for some time that mu suppression is sensitive to oro-facial movements (Muthukumaraswamy, Johnson, Gaetz, & Cheyne, 2004), little work has been carried out using EEG to gauge mu reactivity to facial emotion processing. However, a handful of studies report findings that suggest that the use of mu suppression may be useful in this context. For instance, Moore, Gorodnitsky and Pineda (2012) report mu ERD to both happy and disgusted faces, with an earlier response to disgust and a longer, more extensive response to happy faces. Similarly, decreased beta power (akin to increased beta ERD) has been observed to painful stimuli during the observation of emotional compared to neutral expressions (Senkowski, Kautz, Hauck, Zimmermann, & Engel, 2011). One other study has also reported a difference between beta reactivity over central electrodes (sensorimotor areas) to angry and happy faces; with increased beta power in the angry condition (Guntekin & Basar, 2007). In addition, Pineda & Hecht have shown that mu suppression is positively correlated with a social-perception task (matching facial
9 expressions based on the eye region alone) but not with a social-cognitive task (judging intentions and beliefs of others), suggesting that the hmns may be involved in the former behaviour but not the latter (Pineda & Hecht, 2009). With regard to action observation, the use of EEG to measure mu suppression has been useful in terms of discovering clinical and individual differences in sensorimotor (and possible hmns) activation. Clinically both schizophrenia (McCormick et al., 2012) and autism (Bernier, Dawson, Webb, & Murias, 2007; Oberman et al., 2005) have been associated with abnormal mu reactivity, although much debate remains regarding the robustness and interpretation of these results (Fan, Decety, Yang, Liu, & Cheng, 2010; Puzzo, Cooper, Cantarella, & Russo, 2011; Raymaekers, Wiersema, & Roeyers, 2009). In terms of individual differences, the level of expertise (Behmer & Jantzen), amount of learning (Marshall, Bouquet, Shipley, & Young, 2009) and degree of habituation (e.g. in smokers; Pineda & Oberman, 2006) have been shown to affect mu suppression. Sex differences have also been observed (Cheng et al., 2008; Silas, Levy, Nielsen, Slade, & Holmes, 2010), along with altered mu reactivity according to the degree of empathy (Cooper et al., 2012; Perry, Troje, & Bentin, 2010; Woodruff, Martin, & Bilyk, 2011) and the level of autistic traits (Puzzo, Cooper, Vetter, & Russo, 2010). However, to date, no studies looking at mu reactivity to facial emotion processing have found any individual differences. Of the three studies to look in this area, two did not investigate individual differences (Guntekin & Basar, 2007; Senkowski et al., 2011) and one, investigating the influence of the level of empathic traits, found no differences between those scoring high and low for empathy (Moore et al., 2012). Given the lack of research in this area and the evidence for the usefulness of mu suppression as an index of individual differences in action observation mechanisms, we undertook to explore its application for investigating the neural mechanisms of facial emotion processing. Specifically, we were interested in examining whether emotionally charged facial expressions (positive, negative and neutral) modulate the sensorimotor reactivity induced by hand movement observation. In addition, given the debate in the autism literature, we were interested in testing whether or not this reactivity would vary according to the level of self-reported autistic traits in typically
10 developing adults. The benefits of using such a population include, the availability of larger numbers of potential participants, the lack of certain possible confounds such as medication and the potential to gain insight into the boundaries of the disorder (Hirsch & Weinberger, 2003). Indeed, in the last decade, autism spectrum disorder (ASD) classifications have changed, so that now, facets of autism are seen as an extreme end of the behavioural traits observed in the normal population (Baron-Cohen, Wheelwright, Skinner, Martin, & Clubley, 2001; Constantino & Todd, 2003; Constantino & Todd, 2005). Thus, investigating autistic traits in a typically developing population is useful both for the insight it may provide into autism per se and also into how these traits are manifest in the population as a whole. Method Participants Initially, 80 participants completed the Autism Spectrum Quotient (Baron-Cohen et al., 2001). From this sample, 2 groups were formed comprising of 10 high scorers (high AQ group; 7 female) and 10 low scorers (low AQ group; 6 females). The high AQ group was comprised of those scoring >= 22 and the low AQ group scoring < 11 (Almeida, Dickinson, Maybery, Badcock, & Badcock, 2010). Thus the number of participants in the EEG part of the study was 20 (mean age = 25.4 years). The mean AQ score was 23.9 (sd = 2.28) for the high group and 7.6 (sd = 1.43) for the low group. All participants gave written informed consent and the study was approved by the University of Essex Ethics Committee. Materials The Autism Spectrum Quotient (AQ) was used to assess the degree to which adults from a normal population have traits typically associated with ASD (Baron-Cohen et al., 2001). The questionnaire comprises of 50 questions, each item in the AQ scores 1 point if the participant s answer is an autistic-like
11 answer. Participants scores can range from 0 to 50, with higher scores associated with high traits of autism. This experiment was part of a larger study looking at social gestures, and for the purposes of this experiment, videos containing actors opening and closing their right hands with three different facial expressions were used (see figure 1). For each condition (happy, neutral, angry), 4 actors were filmed (2 female) wearing dark clothes against a dark back-drop and seated in the centre of the screen. The actors hands were held in front of their chests so that both the hand movement and the facial expression were clearly visible. The actors opened and closed their hands at a rate of 1Hz, holding their fingers and thumbs straight. Thus in total, there were 12 different video clips that constituted 1 block. 6 blocks were run in total with the presentation of the video clips randomly ordered at the start of each block. Each video lasted 3 seconds with a 3 second inter-trial interval. Stimuli were presented using Superlab software (Cedrus Corporation, San Pedro, Ca.) on an Apple PowerMac (2GHz PowerPC G5; Apple Inc., Cupertino, Ca.). Insert Figure 1 about here EEG Data Acquisition EEG data were recorded with Neuroscan 4.4 acquisition software and SynampsII amplifiers using a 64 channel Quick-Cap arranged according to the international system (Compumedics, Melbourne, Australia). Eye movements were recorded using 2 facial electrodes - above and below the left eye. Impedances for all electrodes were reduced to below 10 kohm before the start of each session. All data were continuously sampled at 1000 Hz with a bandpass filter of Hz and a 50Hz notch filter. Online EEG data were referenced to a point midway between Cz and CPz, and grounded midway between Fz and FPz.
12 EEG data preparation Following visual inspection of the data, noisy data blocks were rejected. Bad electrodes were excluded on a participant by participant basis (electrode C2 was excluded from one high AQ participant and one low AQ participant; electrode Oz was excluded from 3 high AQ participants). Ocular artifact rejection was carried out using the Neuroscan Edit transform (derived from Semlitsch, Anderer, Schuster, & Presslich, 1986) followed by a second, automatic artifact rejection sweep, with exclusion parameters set at +/- 75mV. In order to calculate ERD/S, the data were epoched from to 3500ms around the start of each video clip and the following steps were performed using the event-related band-power transform in Neuroscan Edit 4.4 (Compumedics, Melbourne, Australia): the data underwent complex demodulation and concurrent filtering (zero phase-shift, 24dB roll-off, envelope computed) into the EEG bandwidths of interest: alpha (8-12Hz) and low beta (12-20Hz). It was trimmed (1000ms from each end, to remove filter warm-up artefacts) and averaged. A reference interval of ms was used to calculate the percentage change between the active period ( ms) and it, using the classic method adapted from Pfurtscheller and colleagues (e.g. Pfurtscheller & Aranibar, 1977; Pfurtscheller & Lopes da Silva, 1999): ERD% = (R-A)/R*100, where R = power in the reference interval and A = power in the active or task phase. Thus, desynchronization and synchronization are expressed as a percentage of activity relative to the reference interval (NB, using this formula ERD produces positive scores and ERS negative). In order to reduce the number of multiple comparisons, the electrodes were collapsed within each hemisphere, resulting in 2 variables: left central (C5, C3, C1) and right central (C6, C4, C2). Design This experiment was a mixed factor design with two repeated-measures factors: emotional expression (happy, neutral, angry) and hemisphere (left, right) and one between-subjects factor: AQ group (high AQ, low AQ). In order to check that our findings were due to mu activity (i.e., deriving from sensorimotor areas) and not related to occipital alpha we also employed Oz as a control site. For Oz data, there was
13 only one repeated measures factor (emotional expression). The dependent variables for all ANOVAs were the event-related desynchronization/synchronization (ERD/S) values in the alpha and low beta bandwidths. Thus two mixed measures ANOVAs were carried out for each scalp location (central alpha, central low beta, occipital alpha and occipital low beta). In order to explore interactions, planned comparisons used one-way ANOVAs to examine between subjects differences and paired students t-tests for repeated measures differences. Results Central sites (C5, C3, C1, C2, C4, C6) Low beta band: No main effects for emotion, hemisphere or group were observed (ps >.187). A strong interaction was observed between emotion and group (F (2,36) = 9.38; p =.001; η 2 p =.343). As can be seen in figure 2, this was driven by greater low beta ERD to happy than both angry and neutral expressions in the low AQ group (t (9) = 2.867; p =.019; 95% CI = 2.83 to and t (9) = 3.327: p =.009; 95% CI = 2.22 to respectively) and by greater low beta ERD to angry than to happy expressions in the high AQ group (t (9) = 2.497; p =.034). There was also significantly more low beta ERD to happy faces for the low than for the high AQ group (t (18) = 2.221; p =.039; 95% CI =.94 to 34.02). No other two- or three-way interactions were significant (ps >.154) Insert figure 2 about here Alpha band: No main effects for emotion, hemisphere or group were observed (ps >.459) but there was a significant interaction between emotion and hemisphere (F (2,36) = 3.492; p =.041; η p 2 =.162). As can
14 be seen in figure 3, greater alpha ERD was observed for happy than for angry expressions in the left hemisphere (t (19) = 2.847; p =.01; 95% CI = 3.57 to 23.4). Also, for happy expressions, alpha ERD was greater in the left than in the right hemisphere (t (19) = 2.51; p =.021; 95% CI = 2.28 to 25.26). Insert figure 3 about here Occipital site (Oz) Data from 3 participants (all high AQ group) were omitted due to noise on the Oz electrode. No main effects or interactions were observed in either bandwidth (ps >.071). This suggests that our findings for the central sites were indeed due to mu activity and not to occipital alpha. Discussion This study sought to examine the usefulness of mu suppression when investigating individual differences in emotional facial processing. Specifically, we investigated whether alpha and low beta ERD over sensorimotor areas would differ according to both the degree of autistic traits of the observer and the facial expression of the observed subject (i.e., the person doing the actions). Our main finding was that in the low beta band from central sites (overlying primary motor areas), whereas those scoring high in autistic traits (high AQ group) showed greater low beta ERD to angry compared to happy expressions, those with low AQ scores showed the opposite effect (greater ERD to happy than either angry or neutral expressions). Also, the low AQ group had greater low beta ERD to happy faces than the high AQ group. In the context of action observation, mu suppression is regarded as a reliable index of hmns activation (Kilner et al., 2009; Muthukumaraswamy & Johnson, 2004b; Pineda, 2005, 2008). In the present study,
15 mu suppression to action observation was modulated by the facial expression of the actor making the hand movement. Consequently, our results suggest that those with higher levels of autistic traits have greater hmns activation to negative facial expressions (anger) and those with low levels have greater hmns activation to positive ones (happy). Additionally, when viewing happy expressions, the low AQ group showed greater hmns activation than the high AQ group. This differentiation according to the level of autistic traits may also help to explain the discrepancy in findings in the previous studies examining mu reactivity in facial processing (Guntekin & Basar, 2007; Moore et al., 2012) as such individual differences were not taken into account in these studies. It is interesting, and perhaps surprising, that we did not find any differences between AQ groups in the alpha bandwidth or indeed, much in the way of alpha ERD to the stimuli presented, regardless of AQ group. Many previous studies, investigating action observation have shown alpha to be suppressed during the observation of movement (e.g. Muthukumaraswamy & Johnson, 2004a; Muthukumaraswamy, Johnson, & McNair, 2004; Oberman, Pineda, & Ramachandran, 2007; Perry & Bentin, 2009) and some have reported differences in this suppression between people with autism and control groups in alpha (Bernier et al., 2007; Oberman et al., 2005). This alpha suppression is typically interpreted in terms of the internal simulation of the movement in the observer. The reason for our lack of findings in this bandwidth is unclear. It is possible that the nature of the stimuli presented may have altered the response (e.g. the relatively small area of the visual scene taken up by the moving hand). Also, with the inclusion of the emotional faces, there is more to take in and potentially more to simulate. It may be that the addition of faces to the stimuli usually presented in such protocols (i.e. moving hands) has a differential modulating effect on the two mu components (alpha and low beta) and that would suggest a different functional role for them both in the simulation process. For instance, it has been suggested that changes in alpha may reflect activation of primary somatosensory cortex, whereas those in beta might indicate motor cortex activity (Avanzini et al., 2012; Hari, 2006) and therefore the results from the current study might reflect relatively greater motor cortex and less somatosensory activation in response to the stimuli.
16 The differential functions of the mu bandwidths in action observation and emotional recognition is an interesting question that merits further investigation. Returning to our main results in the lower beta band, a superficial interpretation might lead one to expect that those scoring high for autistic traits should be worse at recognising happy faces (possibly as a result of less emotional resonance with positive emotions). However, a recent meta-analysis of emotional facial processing in autism suggests that while there may be a difficulty in recognising emotions in autism, recognition of happiness is only marginally impaired (Uljarevic & Hamilton). However, it should be noted there were problems in this analysis resulting from a lack of viable control stimuli (e.g. neutral faces) and that much of the studies analysed used still images as opposed to more ecologically valid moving images. In contrast, and in line with our results, recent psychophysiological findings do show an atypical response to happy faces in adolescents with autism and their siblings (Spencer et al., 2011) and individuals scoring highly on autism spectrum personality traits (Gayle, Gal, & Kieffaber, 2012). Specifically, Gayle and colleagues found a reduced EEG mismatch negativity response to happy but not sad images in those scoring highly on the AQ. Spencer s group found that fmri BOLD responses to happy faces were significantly reduced compared to neutral expressions in both those with autism and their siblings but that this effect was not seen for fearful expressions; this BOLD response was observed in the fusiform face area and putative social brain areas, particularly the superior temporal sulcus (STS). These findings were interpreted in terms of impaired emotional reactivity in autism (Spencer et al., 2011) and argued to be consistent with diminished approach motivation and positive affect and to underlie the general negative experience of social interactions in ASD (Gayle et al., 2012). Additionally, Gayle and colleagues suggested that a reduced response to positive expression is not surprising (as it is consistent with negative social interaction), but that reduced response to negative expressions would be (as it would be consistent with positive social interaction). Our results of both decreased reactivity to happy expressions and increased reactivity to angry faces in the high AQ group fit well with this interpretation and provide even more rationale for negative social experience in ASD. The finding of increased
17 reactivity to angry faces is also compatible with previous reports of preserved anger superiority effect in Asperger s syndrome (Ashwin, Wheelwright, & Baron-Cohen, 2006). The previous findings of decreased STS BOLD response to happy faces in ASD (Spencer et al., 2011) is interesting in relation to our present findings of decreased mu desynchronization for the high AQ group for happy faces. There is a question as to whether previous findings of decreased mu suppression to action observation in ASD reflect a problem with the core hmns or whether it is a reflection of inefficient upstream modulation by a faulty STS (Puzzo, Cooper, Vetter, Russo, & Fitzgerald, 2009). The STS can be included in descriptions of an extended hmns (e.g. Pineda, 2008) and has been shown to be involved in several mentalizing tasks and biological motion processing (Allison, Puce, & McCarthy, 2000; Spencer et al., 2011). Given that individuals with ASD show an impairment in motion perception (Dakin & Frith, 2005) and that the level of autistic traits correlates with STS structure and function (von dem Hagen et al., 2011) it is plausible to suggest that observed problems in core hmns areas (and their associated behaviours) might stem from abnormal input from the STS (information passes from the STS to the inferior parietal lobe and then on to the inferior frontal gyrus; Pineda, 2008). This is an issue that needs to be addressed in future research. Another issue that warrants further investigation is that of how an individual with average levels of autistic traits would react to the protocol used in this experiment. In this paper we have reported the cortical reactivity (in the form a mu ERD) of both high and low AQ scorers. We have found a strong interaction between emotional expression and AQ group, with opposite effects according to group. However, it is unknown as to whether the mu-erd of an average AQ scorer would more resemble that of a high or low scorer or be intermediate between the two. Common sense might suggest that average scorers will be like low scorers but given that the anger superiority effect is also seen in typically developing individuals (e.g. Ohman, Lundqvist, & Esteves, 2001) it is entirely plausible that the mu ERD of average scorers might resemble the pattern of results shown by high AQ scorers. In such a scenario, the findings presented here of low AQ scorers increased mu reactivity to happy expressions and
18 decreased reactivity (indeed ERS: event-related synchronization) to angry faces could be viewed as the more atypical reaction and might be indicative of increased empathic ability in this group. However a recent review paper has suggested that the findings of an anger superiority effect in the general population may be an artefact of the stimuli used and that in fact, there is a tendency towards a happiness superiority effect (Becker, Anderson, Mortensen, Neufeld, & Neel, 2011), in which case, it is arguable that it is the low AQ group who are producing more typical responses. Clearly more work is warranted in this field, both in terms of typical and atypical development. Another issue and possible limitation of the present study, was our use of only three emotional expressions (anger, happiness and neutrality) with two of these (anger and happiness) being somewhat extreme. We chose not to explore other, arguably more subtle, emotions as we were primarily interested in testing the usefulness of mu-erd in detecting individual differences in responses to emotional facial expressions. The data presented in this study goes some way to establish its value and sets the scene for further investigations into the more subtle aspects of facial processing, particularly in ASD. Other issues to be explored include, did our use of somewhat fixed facial expressions (albeit, on a moving person), influen ce the results. There is some evidence, for example, that individuals with ASD do better on tasks with slow dynamic facial expressions rather than static images (e.g. Gepner, Deruelle, & Grynfeltt, 2001; Tardif, Laine, Rodriguez, & Gepner, 2007). The potential for high temporal resolution in ERD/S measures puts it in a good position to answer such questions. Also, the degree to which different facial muscles are involved in different facial expressions may also have had an effect on our findings. If (as in ASD), our high AQ group was only focusing on certain parts of the faces they were presented with, then this may have had an effect on the amount of beta ERD elicited. Future work needs to investigate this possibility through the use of isolating various aspects of the expressions whilst measuring musuppression, preferably with the concomitant use of eye-tracking techniques. Although not directly related to the main aims of the present study, it is also interesting to note the findings pertaining to the interaction between emotion and hemisphere in the alpha band. To recap, we
19 found ERD to happy faces over the left hemisphere in contrast to ERS (alpha synchronisation) in the right hemisphere. Additionally, we found that this ERD to happy faces in the left hemisphere was significantly different to the left hemisphere alpha activation to the angry faces (which also took the form of ERS). This suggests that hmns activation is greater in the left hemisphere to happy faces and is intriguingly consistent with theories of hemispheric laterality in approach-avoidance actions (e.g. Maxwell & Davidson, 2007). However, at present it is unclear what alpha ERS represents in this context. It is plausible that, as in other contexts (e.g. memory and attention), alpha ERS may represent an active inhibition of cortical processing (Cooper, Croft, Dominey, Burgess, & Gruzelier, 2003; Klimesch, Sauseng, & Hanslmayr, 2007) but at present this remains speculative and much more work is needed in this area to understand the possible balance between activation and inhibition in the hmns and how this may be reflected in oscillatory activity in the mu bandwidths. What can be seen from our results as a whole, is that low beta activation may be a more sensitive index of hmns activation than alpha. This is consistent with previous work from our lab with regard to biological motion (Puzzo et al., 2011) and extends the usefulness of this approach to the measurement of individual differences in emotional facial processing In summary, we sought to examine the usefulness of measuring mu reactivity (changes in alpha and low beta oscillations over sensorimotor cortex) to examine individual differences in emotional facial processing. We found that those scoring highly for autistic traits had greater low beta ERD to angry than to happy faces. Those with low AQ scores exhibited the opposite pattern (greater low beta ERD to happy than angry faces) and also showed greater low beta ERD to happy faces than high scorers did. We interpret these findings in the context of the general negative experience of social interactions in ASD and propose that the measurement of mu reactivity in emotional face processing is a useful tool that facilitates the differentiation of both affective stimuli and individual differences in the level of autistic traits.
20 Acknowledgements The authors would like to thank Roger Deeble and Roger Grace for their technical assistance during this study.
21 Figure legends Figure 1: Stills taken from stimulus video of one actor portraying from left to right: happy, neutral and angry facial expressions. Figure 2: Low beta ERD percentage-change over Central sites for low and high AQ groups during angry, neutral and happy conditions (positive values indicate ERD, negative scores indicate ERS). Figure 3: Alpha ERD percentage-change over Central sites for left and right hemispheres during angry, neutral and happy conditions (positive values indicate ERD, negative scores indicate ERS).
22 References Allison, T., Puce, A., & McCarthy, G. (2000). Social perception from visual cues: role of the STS region. Trends in Cognitive Sciences, 4, Almeida, R. A., Dickinson, J. E., Maybery, M. T., Badcock, J. C., & Badcock, D. R. (2010). Visual search performance in the autism spectrum II: the radial frequency search task with additional segmentation cues. Neuropsychologia, 48(14), Ashwin, C., Wheelwright, S., & Baron Cohen, S. (2006). Finding a face in the crowd: testing the anger superiority effect in Asperger's syndrome. Brain and Cognition, 61(1), Avanzini, P., Fabbri Destro, M., Dalla Volta, R., Daprati, E., Rizzolatti, G., & Cantalupo, G. (2012). The dynamics of sensorimotor cortical oscillations during the observation of hand movements: an EEG study. PloS One, 7(5), e Baron Cohen, S., Wheelwright, S., Skinner, R., Martin, J., & Clubley, E. (2001). The autism spectrum quotient (AQ): evidence from Asperger syndrome/high functioning autism, males and females, scientists and mathematicians. J Autism Dev Disord, 31(1), Becker, D. V., Anderson, U. S., Mortensen, C. R., Neufeld, S. L., & Neel, R. (2011). The face in the crowd effect unconfounded: happy faces, not angry faces, are more efficiently detected in single and multiple target visual search tasks. J Exp Psychol Gen, 140(4), Behmer, L. P., Jr., & Jantzen, K. J. (2011). Reading sheet music facilitates sensorimotor mudesynchronization in musicians. Clin Neurophysiol, 122(7), Bernier, R., Dawson, G., Webb, S., & Murias, M. (2007). EEG mu rhythm and imitation impairments in individuals with autism spectrum disorder. Brain Cogn, 64(3), Carr, L., Iacoboni, M., Dubeau, M. C., Mazziotta, J. C., & Lenzi, G. L. (2003). Neural mechanisms of empathy in humans: a relay from neural systems for imitation to limbic areas. Proc Natl Acad Sci U S A, 100(9), Cheng, Y., Lee, P. L., Yang, C. Y., Lin, C. P., Hung, D., & Decety, J. (2008). Gender differences in the mu rhythm of the human mirror neuron system. PLoS One, 3(5), e2113. Constantino, J. N., & Todd, R. D. (2003). Autistic traits in the general population: a twin study. Archives of General Psychiatry, 60, Constantino, J. N., & Todd, R. D. (2005). Intergenerational transmission of subthreshold autistic traits in the general population. Biological Psychiatry, 57, Cooper, N. R., Croft, R. J., Dominey, S. J. J., Burgess, A. P., & Gruzelier, J. H. (2003). Paradox lost? Exploring the role of alpha oscillations during externally vs. internally directed attention and the implications for idling and inhibition hypotheses. International Journal of Psychophysiology, 47(1), Cooper, N. R., Puzzo, I., Pawley, A. D., Bowes Mulligan, R. A., Kirkpatrick, E. V., Antoniou, P. A., et al. (2012). Bridging a yawning chasm: EEG investigations into the debate concerning the role of the human mirror neuron system in contagious yawning. Cogn Affect Behav Neurosci, 12(2), Dakin, S., & Frith, U. (2005). Vagaries of visual perception in autism. Neuron 48, Di Pellegrino, G., Fadiga, L., Fogassi, L., Gallese, V., & Rizzolatti, G. (1992). Understanding motor events: a neurophysiological study. Experimental Brain Research, 91, Dushanova, J., & Donoghue, J. (2010). Neurons in primary motor cortex engaged during action observation. Eur J Neurosci, 31(2),
23 Enticott, P. G., Johnston, P. J., Herring, S. E., Hoy, K. E., & Fitzgerald, P. B. (2008). Mirror neuron activation is associated with facial emotion processing. Neuropsychologia, 46(11), Enticott, P. G., Kennedy, H. A., Bradshaw, J. L., Rinehart, N. J., & Fitzgerald, P. B. (2010). Understanding mirror neurons: evidence for enhanced corticospinal excitability during the observation of transitive but not intransitive hand gestures. Neuropsychologia, 48(9), Fadiga, L., Fogassi, L., Pavesi, G., & Rizzolatti, G. (1995). Motor facilitation during action observation: a magnetic stimulation study. J Neurophysiol, 73(6), Fan, Y. T., Decety, J., Yang, C. Y., Liu, J. L., & Cheng, Y. (2010). Unbroken mirror neurons in autism spectrum disorders. J Child Psychol Psychiatry, 51(9), Gallese, V. (2009). Mirror Neurons, Embodied Simulation, and the Neural Basis of Social Identification. Psychoanalytic Dialogues, 19(5), Gallese, V., & Goldman, A. (1998). Mirror neurons and the simulation theory of mind reading. Trends in Cognitive Sciences, 2(12), Gastaut, H. J. (1952). Étude electrocorticographique de la réactivité des rythmes rolandiques. Rev. Neurol., 87, Gastaut, H. J., & Bert, J. (1954). EEG changes during cinematographic presentation; moving picture activation of the EEG. Electroencephalogr Clin Neurophysiol, 6(3), Gayle, L. C., Gal, D. E., & Kieffaber, P. D. (2012). Measuring affective reactivity in individuals with autism spectrum personality traits using the visual mismatch negativity event related brain potential. Front Hum Neurosci, 6, 334. Gepner, B., Deruelle, C., & Grynfeltt, S. (2001). Motion and emotion: a novel approach to the study of face processing by young autistic children. J Autism Dev Disord, 31(1), Guntekin, B., & Basar, E. (2007). Emotional face expressions are differentiated with brain oscillations. Int J Psychophysiol, 64(1), Hari, R. (2006). Action perception connection and the cortical mu rhythm. Prog Brain Res, 159, Hari, R., Forss, N., Avikainen, S., Kirveskari, E., Salenius, S., & Rizzolatti, G. (1998). Activation of human primary motor cortex during action observation: a neuromagnetic study. Proc Natl Acad Sci U S A, 95(25), Hennenlotter, A., Schroeder, U., Erhard, P., Castrop, F., Haslinger, B., Stoecker, D., et al. (2005). A common neural basis for receptive and expressive communication of pleasant facial affect. Neuroimage, 26(2), Hirsch, S. R., & Weinberger, D. R. (Eds.). (2003). Schizophrenia. (2nd ed.). Oxford: Blackwell Science Ltd. Iacoboni, M., Molnar Szakacs, I., Gallese, V., Buccino, G., Mazziotta, J. C., & Rizzolatti, G. (2005). Grasping the intentions of others with one's own mirror neuron system. PLoS Biol, 3(3), e79. Iacoboni, M., Woods, R. P., Brass, M., Bekkering, H., Mazziotta, J. C., & Rizzolatti, G. (1999). Cortical mechanisms of human imitation. Science, 286(5449), Kilner, J. M., Marchant, J. L., & Frith, C. D. (2009). Relationship between activity in human primary motor cortex during action observation and the mirror neuron system. PLoS One, 4(3), e4925. Klimesch, W., Sauseng, P., & Hanslmayr, S. (2007). EEG alpha oscillations: the inhibition timing hypothesis. Brain Res Rev, 53(1), Leslie, K. R., Johnson Frey, S. H., & Grafton, S. T. (2004). Functional imaging of face and hand imitation: towards a motor theory of empathy. Neuroimage, 21(2), Marshall, P. J., Bouquet, C. A., Shipley, T. F., & Young, T. (2009). Effects of brief imitative experience on EEG desynchronization during action observation. Neuropsychologia, 47(10), Maxwell, J. S., & Davidson, R. J. (2007). Emotion as motion: asymmetries in approach and avoidant actions. Psychol Sci, 18(12), McCormick, L. M., Brumm, M. C., Beadle, J. N., Paradiso, S., Yamada, T., & Andreasen, N. (2012). Mirror neuron function, psychosis, and empathy in schizophrenia. Psychiatry Res, 201(3),
24 Molnar Szakacs, I., Kaplan, J., Greenfield, P. M., & Iacoboni, M. (2006). Observing complex action sequences: The role of the fronto parietal mirror neuron system. Neuroimage, 33(3), Moore, A., Gorodnitsky, I., & Pineda, J. (2012). EEG mu component responses to viewing emotional faces. Behav Brain Res, 226(1), Mukamel, R., Ekstrom, A. D., Kaplan, J., Iacoboni, M., & Fried, I. (2010). Single Neuron Responses in Humans during Execution and Observation of Actions. Curr Biol. Muthukumaraswamy, S. D., & Johnson, B. W. (2004a). Changes in rolandic mu rhythm during observation of a precision grip. Psychophysiology, 41(1), Muthukumaraswamy, S. D., & Johnson, B. W. (2004b). Primary motor cortex activation during action observation revealed by wavelet analysis of the EEG. Clin Neurophysiol, 115(8), Muthukumaraswamy, S. D., Johnson, B. W., Gaetz, W. C., & Cheyne, D. O. (2004). Modulation of neuromagnetic oscillatory activity during the observation of oro facial movements. Neurol Clin Neurophysiol, 2004, 2. Muthukumaraswamy, S. D., Johnson, B. W., & McNair, N. A. (2004). Mu rhythm modulation during observation of an object directed grasp. Brain Res Cogn Brain Res, 19(2), Nishitani, N., & Hari, R. (2000). Temporal dynamics of cortical representation for action. Proc Natl Acad Sci U S A, 97(2), Oberman, L. M., Hubbard, E. M., McCleery, J. P., Altschuler, E. L., Ramachandran, V. S., & Pineda, J. A. (2005). EEG evidence for mirror neuron dysfunction in autism spectrum disorders. Brain Res Cogn Brain Res, 24(2), Oberman, L. M., Pineda, J. A., & Ramachandran, V. S. (2007). The human mirror neuron system: a link between action observation and social skills. Soc Cogn Affect Neurosci, 2(1), Ohman, A., Lundqvist, D., & Esteves, F. (2001). The face in the crowd revisited: a threat advantage with schematic stimuli. J Pers Soc Psychol, 80(3), Perry, A., & Bentin, S. (2009). Mirror activity in the human brain while observing hand movements: A comparison between EEG desynchronization in the mu range and previous fmri results. Brain Research, 1282, Perry, A., Troje, N. F., & Bentin, S. (2010). Exploring motor system contributions to the perception of social information: Evidence from EEG activity in the mu/alpha frequency range. Soc Neurosci, 5(3), Pfurtscheller, G., & Aranibar, A. (1977). Event related cortical desynchronization detected by power measurements of scalp EEG. Electroencephalogr Clin Neurophysiol, 42(6), Pfurtscheller, G., & Lopes da Silva, F. H. (1999). Event related EEG/MEG synchronization and desynchronization: basic principles. Clin Neurophysiol, 110(11), Pineda, J. A. (2005). The functional significance of mu rhythms: translating "seeing" and "hearing" into "doing". Brain Res Brain Res Rev, 50(1), Pineda, J. A. (2008). Sensorimotor cortex as a critical component of an 'extended' mirror neuron system: Does it solve the development, correspondence, and control problems in mirroring? Behav Brain Funct, 4, 47. Pineda, J. A., & Hecht, E. (2009). Mirroring and mu rhythm involvement in social cognition: are there dissociable subcomponents of theory of mind? Biol Psychol, 80(3), Pineda, J. A., & Oberman, L. M. (2006). What goads cigarette smokers to smoke? Neural adaptation and the mirror neuron system. Brain Res, 1121(1), Press, C., Cook, J., Blakemore, S. J., & Kilner, J. (2011). Dynamic modulation of human motor activity when observing actions. J Neurosci, 31(8), Puzzo, I., Cooper, N. R., Cantarella, S., & Russo, R. (2011). Measuring the effects of manipulating stimulus presentation time on sensorimotor alpha and low beta reactivity during hand movement observation. NeuroImage, 57(4),
25 Puzzo, I., Cooper, N. R., Vetter, P., & Russo, R. (2010). EEG activation differences in the pre motor cortex and supplementary motor area between normal individuals with high and low traits of autism. Brain Res, 1342, Puzzo, I., Cooper, N. R., Vetter, P., Russo, R., & Fitzgerald, P. B. (2009). Reduced cortico motor facilitation in a normal sample with high traits of autism. Neurosci Lett, 467(2), Raymaekers, R., Wiersema, J. R., & Roeyers, H. (2009). EEG study of the mirror neuron system in children with high functioning autism. Brain Res, 1304, Rizzolatti, G., & Craighero, L. (2004). The mirror neuron system. Annu Rev Neurosci, 27, Rizzolatti, G., Fadiga, L., Gallese, V., & Fogassi, L. (1996). Premotor cortex and the recognition of motor actions. Brain Res Cogn Brain Res, 3(2), Rochas, V., Gelmini, L., Krolak Salmon, P., Poulet, E., Saoud, M., Brunelin, J., et al. (2012). Disrupting Pre SMA Activity Impairs Facial Happiness Recognition: An Event Related TMS Study. Cereb Cortex. Sartori, L., Bucchioni, G., & Castiello, U. (2012). Motor cortex excitability is tightly coupled to observed movements. Neuropsychologia, 50(9), Semlitsch, H. V., Anderer, P., Schuster, P., & Presslich, O. (1986). A solution for reliable and valid reduction of ocular artifacts, applied to the P300 ERP. Psychophysiology, 23(6), Senkowski, D., Kautz, J., Hauck, M., Zimmermann, R., & Engel, A. K. (2011). Emotional facial expressions modulate pain induced beta and gamma oscillations in sensorimotor cortex. J Neurosci, 31(41), Silas, J., Levy, J. P., Nielsen, M. K., Slade, L., & Holmes, A. (2010). Sex and individual differences in induced and evoked EEG measures of action observation. Neuropsychologia, 48(9), Spencer, M. D., Holt, R. J., Chura, L. R., Suckling, J., Calder, A. J., Bullmore, E. T., et al. (2011). A novel functional brain imaging endophenotype of autism: the neural response to facial expression of emotion. Transl Psychiatry, 1, e19. Tardif, C., Laine, F., Rodriguez, M., & Gepner, B. (2007). Slowing down presentation of facial movements and vocal sounds enhances facial expression recognition and induces facial vocal imitation in children with autism. J Autism Dev Disord, 37(8), Uljarevic, M., & Hamilton, A. (2012). Recognition of Emotions in Autism: A Formal Meta Analysis. J Autism Dev Disord. van der Gaag, C., Minderaa, R. B., & Keysers, C. (2007). Facial expressions: what the mirror neuron system can and cannot tell us. Soc Neurosci, 2(3 4), von dem Hagen, E. A., Nummenmaa, L., Yu, R., Engell, A. D., Ewbank, M. P., & Calder, A. J. (2011). Autism spectrum traits in the typical population predict structure and function in the posterior superior temporal sulcus. Cereb Cortex, 21(3), Woodruff, C. C., Martin, T., & Bilyk, N. (2011). Differences in self and other induced Mu suppression are correlated with empathic abilities. Brain Res, 1405,
26 Figure 1.JPEG
The Change of Mu Rhythm during Action Observation in People with Stroke. Tae-won Yun, PT, MSc, Moon-Kyu Lee, PT, PhD
 1) 동작관찰시뇌졸중환자의뮤리듬변화 The Change of Mu Rhythm during Action Observation in People with Stroke Tae-won Yun PT MSc Moon-Kyu Lee PT PhD Department of Rehab Center Gwangju City Rehabilitation Hospital
1) 동작관찰시뇌졸중환자의뮤리듬변화 The Change of Mu Rhythm during Action Observation in People with Stroke Tae-won Yun PT MSc Moon-Kyu Lee PT PhD Department of Rehab Center Gwangju City Rehabilitation Hospital
Social Cognition and the Mirror Neuron System of the Brain
 Motivating Questions Social Cognition and the Mirror Neuron System of the Brain Jaime A. Pineda, Ph.D. Cognitive Neuroscience Laboratory COGS1 class How do our brains perceive the mental states of others
Motivating Questions Social Cognition and the Mirror Neuron System of the Brain Jaime A. Pineda, Ph.D. Cognitive Neuroscience Laboratory COGS1 class How do our brains perceive the mental states of others
Mirror Neurons in Primates, Humans, and Implications for Neuropsychiatric Disorders
 Mirror Neurons in Primates, Humans, and Implications for Neuropsychiatric Disorders Fiza Singh, M.D. H.S. Assistant Clinical Professor of Psychiatry UCSD School of Medicine VA San Diego Healthcare System
Mirror Neurons in Primates, Humans, and Implications for Neuropsychiatric Disorders Fiza Singh, M.D. H.S. Assistant Clinical Professor of Psychiatry UCSD School of Medicine VA San Diego Healthcare System
Running head: EMPATHY, MIRROR NEURONS, and HEART RATE
 Relationship Between Empathy 1 Running head: EMPATHY, MIRROR NEURONS, and HEART RATE Relationship Between Empathy, Mirror Neuron Activity, and Heart Rate Matt Novakovic, Eric Miller, Timothy Robinson Gustavus
Relationship Between Empathy 1 Running head: EMPATHY, MIRROR NEURONS, and HEART RATE Relationship Between Empathy, Mirror Neuron Activity, and Heart Rate Matt Novakovic, Eric Miller, Timothy Robinson Gustavus
Novel single trial movement classification based on temporal dynamics of EEG
 Novel single trial movement classification based on temporal dynamics of EEG Conference or Workshop Item Accepted Version Wairagkar, M., Daly, I., Hayashi, Y. and Nasuto, S. (2014) Novel single trial movement
Novel single trial movement classification based on temporal dynamics of EEG Conference or Workshop Item Accepted Version Wairagkar, M., Daly, I., Hayashi, Y. and Nasuto, S. (2014) Novel single trial movement
We are IntechOpen, the world s leading publisher of Open Access books Built by scientists, for scientists. International authors and editors
 We are IntechOpen, the world s leading publisher of Open Access books Built by scientists, for scientists 4,000 116,000 120M Open access books available International authors and editors Downloads Our
We are IntechOpen, the world s leading publisher of Open Access books Built by scientists, for scientists 4,000 116,000 120M Open access books available International authors and editors Downloads Our
Developmental changes in mu suppression to observed and executed actions in autism spectrum disorders
 Social Cognitive and Affective Neuroscience Advance Access published February 1, 2012 doi:10.1093/scan/nsr097 SCAN (2012) 1 of 5 Developmental changes in mu suppression to observed and executed actions
Social Cognitive and Affective Neuroscience Advance Access published February 1, 2012 doi:10.1093/scan/nsr097 SCAN (2012) 1 of 5 Developmental changes in mu suppression to observed and executed actions
Mirror neurons. Romana Umrianova
 Mirror neurons Romana Umrianova The functional role of the parieto-frontal mirror circuit: interpretations and misinterpretations Giacomo Rizzolatti and Corrado Sinigaglia Mechanism that unifies action
Mirror neurons Romana Umrianova The functional role of the parieto-frontal mirror circuit: interpretations and misinterpretations Giacomo Rizzolatti and Corrado Sinigaglia Mechanism that unifies action
Giacomo Rizzolatti - selected references
 Giacomo Rizzolatti - selected references 1 Rizzolatti, G., Semi, A. A., & Fabbri-Destro, M. (2014). Linking psychoanalysis with neuroscience: the concept of ego. Neuropsychologia, 55, 143-148. Notes: Through
Giacomo Rizzolatti - selected references 1 Rizzolatti, G., Semi, A. A., & Fabbri-Destro, M. (2014). Linking psychoanalysis with neuroscience: the concept of ego. Neuropsychologia, 55, 143-148. Notes: Through
The old adage I feel your pain may be closer to reality
 16 Health Psychology Volume 6 No. 1, 2009 HEALTH PSYCHOLOGY I Feel Your Pain: Mirror Neurons and Empathy Lindsey MacGillivray, BSc ABSTRACT The old adage I feel your pain may be closer to reality than
16 Health Psychology Volume 6 No. 1, 2009 HEALTH PSYCHOLOGY I Feel Your Pain: Mirror Neurons and Empathy Lindsey MacGillivray, BSc ABSTRACT The old adage I feel your pain may be closer to reality than
MANIPULATION OF HAND MOVEMENT OBSERVATION AND EXECUTION ON MU SUPPRESSION MEASURED BY ELECTROENCEPHALOGRAPHY. By Adrian Andelin.
 MANIPULATION OF HAND MOVEMENT OBSERVATION AND EXECUTION ON MU SUPPRESSION MEASURED BY ELECTROENCEPHALOGRAPHY By Adrian Andelin A Thesis Submitted in Partial Fulfillment of the Requirements for the Degree
MANIPULATION OF HAND MOVEMENT OBSERVATION AND EXECUTION ON MU SUPPRESSION MEASURED BY ELECTROENCEPHALOGRAPHY By Adrian Andelin A Thesis Submitted in Partial Fulfillment of the Requirements for the Degree
Oriental Painting and Cortical Motor Activation: An EEG Oscillation Study
 1 Oriental Painting and Cortical Motor Activation: An EEG Oscillation Study Joung A Eom 1,3, Sung-Eun Lee 2,3* Department of Aesthetics, College of Humanities, Seoul National University, Seoul, Korea 1
1 Oriental Painting and Cortical Motor Activation: An EEG Oscillation Study Joung A Eom 1,3, Sung-Eun Lee 2,3* Department of Aesthetics, College of Humanities, Seoul National University, Seoul, Korea 1
Neuropsychologia 51 (2013) Contents lists available at SciVerse ScienceDirect. Neuropsychologia
 Neuropsychologia 51 (2013) 52 58 Contents lists available at SciVerse ScienceDirect Neuropsychologia journal homepage: www.elsevier.com/locate/neuropsychologia Modulation of motor cortex activity when
Neuropsychologia 51 (2013) 52 58 Contents lists available at SciVerse ScienceDirect Neuropsychologia journal homepage: www.elsevier.com/locate/neuropsychologia Modulation of motor cortex activity when
Modulation of motor cortex activity when observing. rewarding and punishing actions
 1 2 3 Modulation of motor cortex activity when observing rewarding and punishing actions 4 5 6 7 Elliot Clayton Brown 1,2 *, Jan Roelf Wiersema 3, Gilles Pourtois 3, Martin Brüne 1,2 8 9 10 11 12 13 14
1 2 3 Modulation of motor cortex activity when observing rewarding and punishing actions 4 5 6 7 Elliot Clayton Brown 1,2 *, Jan Roelf Wiersema 3, Gilles Pourtois 3, Martin Brüne 1,2 8 9 10 11 12 13 14
Mirror neuron functioning: an explanation for gender differences in empathy?
 Author: Marja Nab, 314483 Supervisor: Drs. A. H. M. van Boxtel Bachelor thesis in cognitive Neuroscience Department Psychology and Health, Cognitive Neuroscience, Tilburg University June, 2010 Abstract
Author: Marja Nab, 314483 Supervisor: Drs. A. H. M. van Boxtel Bachelor thesis in cognitive Neuroscience Department Psychology and Health, Cognitive Neuroscience, Tilburg University June, 2010 Abstract
Gender di erences in the human mirror system: a magnetoencephalography study
 COGNITIVE NEUROSCIENCE AND NEUROPSYCHOLOGY Gender di erences in the human mirror system: a magnetoencephalography study Ya-Wei Cheng a,c,ovidj.l.tzeng b,d,jeandecety e,toshiakiimada f and Jen-Chuen Hsieh
COGNITIVE NEUROSCIENCE AND NEUROPSYCHOLOGY Gender di erences in the human mirror system: a magnetoencephalography study Ya-Wei Cheng a,c,ovidj.l.tzeng b,d,jeandecety e,toshiakiimada f and Jen-Chuen Hsieh
EEG evidence for the presence of an action observation execution matching system in children
 European Journal of Neuroscience, Vol. 23, pp. 2505 2510, 2006 doi:10.1111/j.1460-9568.2006.04769.x EEG evidence for the presence of an action observation execution matching system in children Jean-François
European Journal of Neuroscience, Vol. 23, pp. 2505 2510, 2006 doi:10.1111/j.1460-9568.2006.04769.x EEG evidence for the presence of an action observation execution matching system in children Jean-François
Manuscript. Do not cite. 1. Mirror neurons or emulator neurons? Gergely Csibra Birkbeck, University of London
 Manuscript. Do not cite. 1 Mirror neurons or emulator neurons? Gergely Csibra Birkbeck, University of London Mirror neurons are cells in the macaque brain that discharge both when the monkey performs a
Manuscript. Do not cite. 1 Mirror neurons or emulator neurons? Gergely Csibra Birkbeck, University of London Mirror neurons are cells in the macaque brain that discharge both when the monkey performs a
This article appeared in a journal published by Elsevier. The attached copy is furnished to the author for internal non-commercial research and
 This article appeared in a journal published by Elsevier. The attached copy is furnished to the author for internal non-commercial research and education use, including for instruction at the authors institution
This article appeared in a journal published by Elsevier. The attached copy is furnished to the author for internal non-commercial research and education use, including for instruction at the authors institution
Distinct EEG Amplitude Suppression to Facial Gestures as Evidence for a Mirror Mechanism in Newborn Monkeys
 Distinct EEG Amplitude Suppression to Facial Gestures as Evidence for a Mirror Mechanism in Newborn Monkeys Pier Francesco Ferrari 1,2 *, Ross E. Vanderwert 3 *, Annika Paukner 4, Seth Bower 4, Stephen
Distinct EEG Amplitude Suppression to Facial Gestures as Evidence for a Mirror Mechanism in Newborn Monkeys Pier Francesco Ferrari 1,2 *, Ross E. Vanderwert 3 *, Annika Paukner 4, Seth Bower 4, Stephen
Modulation of the mirror system by social relevance
 doi:1.193/scan/nsl17 SCAN (26) 1,143 148 Modulation of the mirror system by social relevance James M. Kilner, Jennifer L. Marchant, and Chris D. Frith The Wellcome Department of Imaging Neuroscience, Queen
doi:1.193/scan/nsl17 SCAN (26) 1,143 148 Modulation of the mirror system by social relevance James M. Kilner, Jennifer L. Marchant, and Chris D. Frith The Wellcome Department of Imaging Neuroscience, Queen
Exploring motor system contributions to the perception of social information: Evidence from EEG activity in the mu/alpha frequency range
 Social Neuroscience ISSN: 1747-0919 (Print) 1747-0927 (Online) Journal homepage: http://www.tandfonline.com/loi/psns20 Exploring motor system contributions to the perception of social information: Evidence
Social Neuroscience ISSN: 1747-0919 (Print) 1747-0927 (Online) Journal homepage: http://www.tandfonline.com/loi/psns20 Exploring motor system contributions to the perception of social information: Evidence
Hierarchically Organized Mirroring Processes in Social Cognition: The Functional Neuroanatomy of Empathy
 Hierarchically Organized Mirroring Processes in Social Cognition: The Functional Neuroanatomy of Empathy Jaime A. Pineda, A. Roxanne Moore, Hanie Elfenbeinand, and Roy Cox Motivation Review the complex
Hierarchically Organized Mirroring Processes in Social Cognition: The Functional Neuroanatomy of Empathy Jaime A. Pineda, A. Roxanne Moore, Hanie Elfenbeinand, and Roy Cox Motivation Review the complex
The Integration of Features in Visual Awareness : The Binding Problem. By Andrew Laguna, S.J.
 The Integration of Features in Visual Awareness : The Binding Problem By Andrew Laguna, S.J. Outline I. Introduction II. The Visual System III. What is the Binding Problem? IV. Possible Theoretical Solutions
The Integration of Features in Visual Awareness : The Binding Problem By Andrew Laguna, S.J. Outline I. Introduction II. The Visual System III. What is the Binding Problem? IV. Possible Theoretical Solutions
The EEG Analysis of Auditory Emotional Stimuli Perception in TBI Patients with Different SCG Score
 Open Journal of Modern Neurosurgery, 2014, 4, 81-96 Published Online April 2014 in SciRes. http://www.scirp.org/journal/ojmn http://dx.doi.org/10.4236/ojmn.2014.42017 The EEG Analysis of Auditory Emotional
Open Journal of Modern Neurosurgery, 2014, 4, 81-96 Published Online April 2014 in SciRes. http://www.scirp.org/journal/ojmn http://dx.doi.org/10.4236/ojmn.2014.42017 The EEG Analysis of Auditory Emotional
Published 21 May 2009 Cite this as: BMJ Case Reports 2009 [doi: /bcr ] Copyright 2009 by the BMJ Publishing Group Ltd.
![Published 21 May 2009 Cite this as: BMJ Case Reports 2009 [doi: /bcr ] Copyright 2009 by the BMJ Publishing Group Ltd. Published 21 May 2009 Cite this as: BMJ Case Reports 2009 [doi: /bcr ] Copyright 2009 by the BMJ Publishing Group Ltd.](/thumbs/75/72410127.jpg) Published 21 May 2009 Cite this as: BMJ Case Reports 2009 [doi:10.1136/bcr.07.2008.0593] Copyright 2009 by the BMJ Publishing Group Ltd. Right hemisphere dominance for understanding the intentions of others:
Published 21 May 2009 Cite this as: BMJ Case Reports 2009 [doi:10.1136/bcr.07.2008.0593] Copyright 2009 by the BMJ Publishing Group Ltd. Right hemisphere dominance for understanding the intentions of others:
Genetic Conclusions. Layers of the ASD Onion. The notion of finding a simple genetic cause to help define ASD will not likely occur.
 Genetic Conclusions The notion of finding a simple genetic cause to help define ASD will not likely occur. There are likely over 1,000 genes that have various roles in ASD which replicates the huge clinical
Genetic Conclusions The notion of finding a simple genetic cause to help define ASD will not likely occur. There are likely over 1,000 genes that have various roles in ASD which replicates the huge clinical
Assessing Human Mirror Activity With EEG Mu Rhythm: A Meta-Analysis
 Psychological Bulletin 2015 American Psychological Association 2016, Vol. 142, No. 3, 291 313 0033-2909/16/$12.00 http://dx.doi.org/10.1037/bul0000031 Assessing Human Mirror Activity With EEG Mu Rhythm:
Psychological Bulletin 2015 American Psychological Association 2016, Vol. 142, No. 3, 291 313 0033-2909/16/$12.00 http://dx.doi.org/10.1037/bul0000031 Assessing Human Mirror Activity With EEG Mu Rhythm:
Abstract. Introduction. European Journal of Neuroscience, pp. 1 12, doi: /ejn USA 2 Washington State University, Pullman, WA, USA
 European Journal of Neuroscience, pp. 1 12, 2016 doi:10.1111/ejn.13389 Mirror neuron activation as a function of explicit learning: changes in mu-event-related power after learning novel responses to ideomotor
European Journal of Neuroscience, pp. 1 12, 2016 doi:10.1111/ejn.13389 Mirror neuron activation as a function of explicit learning: changes in mu-event-related power after learning novel responses to ideomotor
EEG-Rhythm Dynamics during a 2-back Working Memory Task and Performance
 EEG-Rhythm Dynamics during a 2-back Working Memory Task and Performance Tsvetomira Tsoneva, Davide Baldo, Victor Lema and Gary Garcia-Molina Abstract Working memory is an essential component of human cognition
EEG-Rhythm Dynamics during a 2-back Working Memory Task and Performance Tsvetomira Tsoneva, Davide Baldo, Victor Lema and Gary Garcia-Molina Abstract Working memory is an essential component of human cognition
Oscillations: From Neuron to MEG
 Oscillations: From Neuron to MEG Educational Symposium, MEG UK 2014, Nottingham, Jan 8th 2014 Krish Singh CUBRIC, School of Psychology Cardiff University What are we trying to achieve? Bridge the gap from
Oscillations: From Neuron to MEG Educational Symposium, MEG UK 2014, Nottingham, Jan 8th 2014 Krish Singh CUBRIC, School of Psychology Cardiff University What are we trying to achieve? Bridge the gap from
Dynamic functional integration of distinct neural empathy systems
 Social Cognitive and Affective Neuroscience Advance Access published August 16, 2013 Dynamic functional integration of distinct neural empathy systems Shamay-Tsoory, Simone G. Department of Psychology,
Social Cognitive and Affective Neuroscience Advance Access published August 16, 2013 Dynamic functional integration of distinct neural empathy systems Shamay-Tsoory, Simone G. Department of Psychology,
Infants mu suppression during the observation of real and mimicked goal-directed actions. Herbert Roeyers 1
 Infants mu suppression during the observation of real and mimicked goal-directed actions Petra Warreyn 1, Lieselot Ruysschaert 1,Jan R. Wiersema 1,Andrea Handl 2,Griet Pattyn 1, & Herbert Roeyers 1 1 Department
Infants mu suppression during the observation of real and mimicked goal-directed actions Petra Warreyn 1, Lieselot Ruysschaert 1,Jan R. Wiersema 1,Andrea Handl 2,Griet Pattyn 1, & Herbert Roeyers 1 1 Department
The role of motor contagion in the prediction of action
 Neuropsychologia 43 (2005) 260 267 Review The role of motor contagion in the prediction of action Sarah-Jayne Blakemore a,, Chris Frith b,1 a Institute of Cognitive Neuroscience, 17 Queen Square, London
Neuropsychologia 43 (2005) 260 267 Review The role of motor contagion in the prediction of action Sarah-Jayne Blakemore a,, Chris Frith b,1 a Institute of Cognitive Neuroscience, 17 Queen Square, London
A Brain Computer Interface System For Auto Piloting Wheelchair
 A Brain Computer Interface System For Auto Piloting Wheelchair Reshmi G, N. Kumaravel & M. Sasikala Centre for Medical Electronics, Dept. of Electronics and Communication Engineering, College of Engineering,
A Brain Computer Interface System For Auto Piloting Wheelchair Reshmi G, N. Kumaravel & M. Sasikala Centre for Medical Electronics, Dept. of Electronics and Communication Engineering, College of Engineering,
Men fear other men most: Gender specific brain activations in. perceiving threat from dynamic faces and bodies. An fmri. study.
 Men fear other men most: Gender specific brain activations in perceiving threat from dynamic faces and bodies. An fmri study. Kret, ME, Pichon, S 2,4, Grèzes, J 2, & de Gelder, B,3 Cognitive and Affective
Men fear other men most: Gender specific brain activations in perceiving threat from dynamic faces and bodies. An fmri study. Kret, ME, Pichon, S 2,4, Grèzes, J 2, & de Gelder, B,3 Cognitive and Affective
Sensorimotor learning configures the human mirror system
 1 Sensorimotor learning configures the human mirror system Caroline Catmur 1, Vincent Walsh 1,2 & Cecilia Heyes 1 1 Department of Psychology, University College London, London WC1H 0AP, UK. 2 Institute
1 Sensorimotor learning configures the human mirror system Caroline Catmur 1, Vincent Walsh 1,2 & Cecilia Heyes 1 1 Department of Psychology, University College London, London WC1H 0AP, UK. 2 Institute
Mu rhythm desynchronization by tongue thrust observation
 ORIGINAL RESEARCH published: 11 September 2015 doi: 10.3389/fnhum.2015.00501 Mu rhythm desynchronization by tongue thrust observation Kotoe Sakihara 1,2 * and Masumi Inagaki 1 1 Department of Developmental
ORIGINAL RESEARCH published: 11 September 2015 doi: 10.3389/fnhum.2015.00501 Mu rhythm desynchronization by tongue thrust observation Kotoe Sakihara 1,2 * and Masumi Inagaki 1 1 Department of Developmental
Unbroken mirrors: challenging a theory of Autism
 Opinion Unbroken mirrors: challenging a theory of Autism Victoria Southgate 1 and Antonia F. de C. Hamilton 2 1 Centre for Brain and Cognitive Development, School of Psychology, Birkbeck College, London,
Opinion Unbroken mirrors: challenging a theory of Autism Victoria Southgate 1 and Antonia F. de C. Hamilton 2 1 Centre for Brain and Cognitive Development, School of Psychology, Birkbeck College, London,
Report. Sensorimotor Learning Configures the Human Mirror System. Caroline Catmur, 1, * Vincent Walsh, 1,2 and Cecilia Heyes 1 1
 Current Biology 17, 1527 1531, September 4, 2007 ª2007 Elsevier Ltd All rights reserved DOI 10.1016/j.cub.2007.08.006 Sensorimotor Learning Configures the Human Mirror System Report Caroline Catmur, 1,
Current Biology 17, 1527 1531, September 4, 2007 ª2007 Elsevier Ltd All rights reserved DOI 10.1016/j.cub.2007.08.006 Sensorimotor Learning Configures the Human Mirror System Report Caroline Catmur, 1,
Perception of Faces and Bodies
 CURRENT DIRECTIONS IN PSYCHOLOGICAL SCIENCE Perception of Faces and Bodies Similar or Different? Virginia Slaughter, 1 Valerie E. Stone, 2 and Catherine Reed 3 1 Early Cognitive Development Unit and 2
CURRENT DIRECTIONS IN PSYCHOLOGICAL SCIENCE Perception of Faces and Bodies Similar or Different? Virginia Slaughter, 1 Valerie E. Stone, 2 and Catherine Reed 3 1 Early Cognitive Development Unit and 2
Action and Emotion Understanding
 Action and Emotion Understanding How do we grasp what other people are doing and feeling? Why does it seem so intuitive? Why do you have a visceral reaction when you see a wound or someone in a physically
Action and Emotion Understanding How do we grasp what other people are doing and feeling? Why does it seem so intuitive? Why do you have a visceral reaction when you see a wound or someone in a physically
CS/NEUR125 Brains, Minds, and Machines. Due: Friday, April 14
 CS/NEUR125 Brains, Minds, and Machines Assignment 5: Neural mechanisms of object-based attention Due: Friday, April 14 This Assignment is a guided reading of the 2014 paper, Neural Mechanisms of Object-Based
CS/NEUR125 Brains, Minds, and Machines Assignment 5: Neural mechanisms of object-based attention Due: Friday, April 14 This Assignment is a guided reading of the 2014 paper, Neural Mechanisms of Object-Based
Simultaneous Real-Time Detection of Motor Imagery and Error-Related Potentials for Improved BCI Accuracy
 Simultaneous Real-Time Detection of Motor Imagery and Error-Related Potentials for Improved BCI Accuracy P. W. Ferrez 1,2 and J. del R. Millán 1,2 1 IDIAP Research Institute, Martigny, Switzerland 2 Ecole
Simultaneous Real-Time Detection of Motor Imagery and Error-Related Potentials for Improved BCI Accuracy P. W. Ferrez 1,2 and J. del R. Millán 1,2 1 IDIAP Research Institute, Martigny, Switzerland 2 Ecole
AUXILIARIES AND NEUROPLASTICITY
 AUXILIARIES AND NEUROPLASTICITY Claudio Babiloni, Ph.D. Department of Biomedical Sciences, University of Foggia (UNIFG), Italy UNIFG structured personnel involved Prof. Claudio Babiloni (Coordinator),
AUXILIARIES AND NEUROPLASTICITY Claudio Babiloni, Ph.D. Department of Biomedical Sciences, University of Foggia (UNIFG), Italy UNIFG structured personnel involved Prof. Claudio Babiloni (Coordinator),
Dr. Mark Ashton Smith, Department of Psychology, Bilkent University
 UMAN CONSCIOUSNESS some leads based on findings in neuropsychology Dr. Mark Ashton Smith, Department of Psychology, Bilkent University nattentional Blindness Simons and Levin, 1998 Not Detected Detected
UMAN CONSCIOUSNESS some leads based on findings in neuropsychology Dr. Mark Ashton Smith, Department of Psychology, Bilkent University nattentional Blindness Simons and Levin, 1998 Not Detected Detected
EEG Analysis on Brain.fm (Focus)
 EEG Analysis on Brain.fm (Focus) Introduction 17 subjects were tested to measure effects of a Brain.fm focus session on cognition. With 4 additional subjects, we recorded EEG data during baseline and while
EEG Analysis on Brain.fm (Focus) Introduction 17 subjects were tested to measure effects of a Brain.fm focus session on cognition. With 4 additional subjects, we recorded EEG data during baseline and while
Figure 1. Source localization results for the No Go N2 component. (a) Dipole modeling
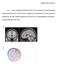 Supplementary materials 1 Figure 1. Source localization results for the No Go N2 component. (a) Dipole modeling analyses placed the source of the No Go N2 component in the dorsal ACC, near the ACC source
Supplementary materials 1 Figure 1. Source localization results for the No Go N2 component. (a) Dipole modeling analyses placed the source of the No Go N2 component in the dorsal ACC, near the ACC source
The neurolinguistic toolbox Jonathan R. Brennan. Introduction to Neurolinguistics, LSA2017 1
 The neurolinguistic toolbox Jonathan R. Brennan Introduction to Neurolinguistics, LSA2017 1 Psycholinguistics / Neurolinguistics Happy Hour!!! Tuesdays 7/11, 7/18, 7/25 5:30-6:30 PM @ the Boone Center
The neurolinguistic toolbox Jonathan R. Brennan Introduction to Neurolinguistics, LSA2017 1 Psycholinguistics / Neurolinguistics Happy Hour!!! Tuesdays 7/11, 7/18, 7/25 5:30-6:30 PM @ the Boone Center
Developmental Social Cognition Cognitive Development
 Developmental Social Cognition Cognitive Development People Concepts Do we think about people in the same way that we think about inanimate objects? What if your brother was... a zombie? Walk slowly About
Developmental Social Cognition Cognitive Development People Concepts Do we think about people in the same way that we think about inanimate objects? What if your brother was... a zombie? Walk slowly About
REHEARSAL PROCESSES IN WORKING MEMORY AND SYNCHRONIZATION OF BRAIN AREAS
 REHEARSAL PROCESSES IN WORKING MEMORY AND SYNCHRONIZATION OF BRAIN AREAS Franziska Kopp* #, Erich Schröger* and Sigrid Lipka # *University of Leipzig, Institute of General Psychology # University of Leipzig,
REHEARSAL PROCESSES IN WORKING MEMORY AND SYNCHRONIZATION OF BRAIN AREAS Franziska Kopp* #, Erich Schröger* and Sigrid Lipka # *University of Leipzig, Institute of General Psychology # University of Leipzig,
Imitation and action understanding in autistic spectrum disorders: How valid is the hypothesis of a deficit in the mirror neuron system?
 Neuropsychologia 45 (2007) 1859 1868 Imitation and action understanding in autistic spectrum disorders: How valid is the hypothesis of a deficit in the mirror neuron system? Antonia F. de C. Hamilton a,,
Neuropsychologia 45 (2007) 1859 1868 Imitation and action understanding in autistic spectrum disorders: How valid is the hypothesis of a deficit in the mirror neuron system? Antonia F. de C. Hamilton a,,
AUTOCORRELATION AND CROSS-CORRELARION ANALYSES OF ALPHA WAVES IN RELATION TO SUBJECTIVE PREFERENCE OF A FLICKERING LIGHT
 AUTOCORRELATION AND CROSS-CORRELARION ANALYSES OF ALPHA WAVES IN RELATION TO SUBJECTIVE PREFERENCE OF A FLICKERING LIGHT Y. Soeta, S. Uetani, and Y. Ando Graduate School of Science and Technology, Kobe
AUTOCORRELATION AND CROSS-CORRELARION ANALYSES OF ALPHA WAVES IN RELATION TO SUBJECTIVE PREFERENCE OF A FLICKERING LIGHT Y. Soeta, S. Uetani, and Y. Ando Graduate School of Science and Technology, Kobe
Seeing and hearing speech excites the motor system involved in speech production
 Neuropsychologia 41 (2003) 989 994 Seeing and hearing speech excites the motor system involved in speech production K.E. Watkins, A.P. Strafella, T. Paus Cognitive Neuroscience Unit, Montreal Neurological
Neuropsychologia 41 (2003) 989 994 Seeing and hearing speech excites the motor system involved in speech production K.E. Watkins, A.P. Strafella, T. Paus Cognitive Neuroscience Unit, Montreal Neurological
Social and Pragmatic Language in Autistic Children
 Parkland College A with Honors Projects Honors Program 2015 Social and Pragmatic Language in Autistic Children Hannah Li Parkland College Recommended Citation Li, Hannah, "Social and Pragmatic Language
Parkland College A with Honors Projects Honors Program 2015 Social and Pragmatic Language in Autistic Children Hannah Li Parkland College Recommended Citation Li, Hannah, "Social and Pragmatic Language
EEG evidence for mirror neuron dysfunc1on in au1sm spectrum disorders. Oberman, Hubbard, McCleery, Altschuler, Ramachandran, Pineda
 EEG evidence for mirror neuron dysfunc1on in au1sm spectrum disorders Oberman, Hubbard, McCleery, Altschuler, Ramachandran, Pineda Au9sm Spectrum Disorders Characterized by deficits in social and communica9ve
EEG evidence for mirror neuron dysfunc1on in au1sm spectrum disorders Oberman, Hubbard, McCleery, Altschuler, Ramachandran, Pineda Au9sm Spectrum Disorders Characterized by deficits in social and communica9ve
Biomedical Research 2013; 24 (3): ISSN X
 Biomedical Research 2013; 24 (3): 359-364 ISSN 0970-938X http://www.biomedres.info Investigating relative strengths and positions of electrical activity in the left and right hemispheres of the human brain
Biomedical Research 2013; 24 (3): 359-364 ISSN 0970-938X http://www.biomedres.info Investigating relative strengths and positions of electrical activity in the left and right hemispheres of the human brain
Observational Learning of New Movement Sequences Is Reflected in Fronto-Parietal Coherence
 of New Movement Sequences Is Reflected in Fronto-Parietal Coherence Jurjen van der Helden 1,2 *., Hein T. van Schie 3., Christiaan Rombouts 2 1 Donders Institute for Brain, Cognition and Behaviour, Radboud
of New Movement Sequences Is Reflected in Fronto-Parietal Coherence Jurjen van der Helden 1,2 *., Hein T. van Schie 3., Christiaan Rombouts 2 1 Donders Institute for Brain, Cognition and Behaviour, Radboud
Selective bias in temporal bisection task by number exposition
 Selective bias in temporal bisection task by number exposition Carmelo M. Vicario¹ ¹ Dipartimento di Psicologia, Università Roma la Sapienza, via dei Marsi 78, Roma, Italy Key words: number- time- spatial
Selective bias in temporal bisection task by number exposition Carmelo M. Vicario¹ ¹ Dipartimento di Psicologia, Università Roma la Sapienza, via dei Marsi 78, Roma, Italy Key words: number- time- spatial
The role of amplitude, phase, and rhythmicity of neural oscillations in top-down control of cognition
 The role of amplitude, phase, and rhythmicity of neural oscillations in top-down control of cognition Chair: Jason Samaha, University of Wisconsin-Madison Co-Chair: Ali Mazaheri, University of Birmingham
The role of amplitude, phase, and rhythmicity of neural oscillations in top-down control of cognition Chair: Jason Samaha, University of Wisconsin-Madison Co-Chair: Ali Mazaheri, University of Birmingham
MULTI-CHANNEL COMMUNICATION
 INTRODUCTION Research on the Deaf Brain is beginning to provide a new evidence base for policy and practice in relation to intervention with deaf children. This talk outlines the multi-channel nature of
INTRODUCTION Research on the Deaf Brain is beginning to provide a new evidence base for policy and practice in relation to intervention with deaf children. This talk outlines the multi-channel nature of
Supporting Online Material for
 www.sciencemag.org/cgi/content/full/1176170/dc1 Supporting Online Material for Mindblind Eyes: An Absence of Spontaneous Theory of Mind in Asperger Syndrome Atsushi Senju,* Victoria Southgate, Sarah White,
www.sciencemag.org/cgi/content/full/1176170/dc1 Supporting Online Material for Mindblind Eyes: An Absence of Spontaneous Theory of Mind in Asperger Syndrome Atsushi Senju,* Victoria Southgate, Sarah White,
(This is a sample cover image for this issue. The actual cover is not yet available at this time.)
 (This is a sample cover image for this issue. The actual cover is not yet available at this time.) This article appeared in a journal published by Elsevier. The attached copy is furnished to the author
(This is a sample cover image for this issue. The actual cover is not yet available at this time.) This article appeared in a journal published by Elsevier. The attached copy is furnished to the author
Crossmodal classification of mu rhythm activity during. action observation and execution suggests specificity to. somatosensory features of actions.
 Mu rhythm crossmodal multivariate classification - 1 ABBREVIATED TITLE: Mu rhythm crossmodal multivariate classification Crossmodal classification of mu rhythm activity during action observation and execution
Mu rhythm crossmodal multivariate classification - 1 ABBREVIATED TITLE: Mu rhythm crossmodal multivariate classification Crossmodal classification of mu rhythm activity during action observation and execution
FAILURES OF OBJECT RECOGNITION. Dr. Walter S. Marcantoni
 FAILURES OF OBJECT RECOGNITION Dr. Walter S. Marcantoni VISUAL AGNOSIA -damage to the extrastriate visual regions (occipital, parietal and temporal lobes) disrupts recognition of complex visual stimuli
FAILURES OF OBJECT RECOGNITION Dr. Walter S. Marcantoni VISUAL AGNOSIA -damage to the extrastriate visual regions (occipital, parietal and temporal lobes) disrupts recognition of complex visual stimuli
Functional connectivity during real vs imagined visuomotor tasks: an EEG study
 MOTOR SYSTEMS Functional connectivity during real vs imagined visuomotor tasks: an EEG study James M. Kilner, 1,CA Yves Paulignan and Driss Boussaoud Institut des Sciences Cognitives, 67 Boulevard Pinel,
MOTOR SYSTEMS Functional connectivity during real vs imagined visuomotor tasks: an EEG study James M. Kilner, 1,CA Yves Paulignan and Driss Boussaoud Institut des Sciences Cognitives, 67 Boulevard Pinel,
Gender Differences in the Mu Rhythm of the Human Mirror-Neuron System
 Gender Differences in the Mu Rhythm of the Human Mirror-Neuron System Yawei Cheng 1,2, Po-Lei Lee 3, Chia-Yen Yang 4, Ching-Po Lin 1, Daisy Hung 5, Jean Decety 6,7 * 1 Institute of Neuroscience, School
Gender Differences in the Mu Rhythm of the Human Mirror-Neuron System Yawei Cheng 1,2, Po-Lei Lee 3, Chia-Yen Yang 4, Ching-Po Lin 1, Daisy Hung 5, Jean Decety 6,7 * 1 Institute of Neuroscience, School
Mirror Neuron System Differentially Activated by Facial Expressions and Social Hand Gestures: A Functional Magnetic Resonance Imaging Study
 Mirror Neuron System Differentially Activated by Facial Expressions and Social Hand Gestures: A Functional Magnetic Resonance Imaging Study Kimberly J. Montgomery and James V. Haxby Abstract & Facial expressions
Mirror Neuron System Differentially Activated by Facial Expressions and Social Hand Gestures: A Functional Magnetic Resonance Imaging Study Kimberly J. Montgomery and James V. Haxby Abstract & Facial expressions
Journal of Personality Assesment. Mirroring Activity in the Brain and Movement Determinant in the Rorschach Test
 Mirroring Activity in the Brain and Movement Determinant in the Rorschach Test Journal: Journal of Personality Assessment Manuscript ID: Draft Manuscript Type: General Submission Keywords: electroencephalography,
Mirroring Activity in the Brain and Movement Determinant in the Rorschach Test Journal: Journal of Personality Assessment Manuscript ID: Draft Manuscript Type: General Submission Keywords: electroencephalography,
When Do We Stop Calling Them Mirror Neurons?
 When Do We Stop Calling Them Mirror Neurons? Sebo Uithol (S.Uithol@nici.ru.nl) Willem F. G. Haselager (W.Haselager@nici.ru.nl) Harold Bekkering (H.Bekkering@nici.ru.nl) Nijmegen Institute for Cognition
When Do We Stop Calling Them Mirror Neurons? Sebo Uithol (S.Uithol@nici.ru.nl) Willem F. G. Haselager (W.Haselager@nici.ru.nl) Harold Bekkering (H.Bekkering@nici.ru.nl) Nijmegen Institute for Cognition
WHAT AUTISM CAN TELL US ABOUT THE LINK BETWEEN EMPATHY AND MORAL REASONING? 1
 FLAVIA FELLETTI University of Barcelona Flavia.felletti@gmail.com WHAT AUTISM CAN TELL US ABOUT THE LINK BETWEEN EMPATHY AND MORAL REASONING? 1 abstract I will discuss the relationship between empathy
FLAVIA FELLETTI University of Barcelona Flavia.felletti@gmail.com WHAT AUTISM CAN TELL US ABOUT THE LINK BETWEEN EMPATHY AND MORAL REASONING? 1 abstract I will discuss the relationship between empathy
Emotion Detection Using Physiological Signals. M.A.Sc. Thesis Proposal Haiyan Xu Supervisor: Prof. K.N. Plataniotis
 Emotion Detection Using Physiological Signals M.A.Sc. Thesis Proposal Haiyan Xu Supervisor: Prof. K.N. Plataniotis May 10 th, 2011 Outline Emotion Detection Overview EEG for Emotion Detection Previous
Emotion Detection Using Physiological Signals M.A.Sc. Thesis Proposal Haiyan Xu Supervisor: Prof. K.N. Plataniotis May 10 th, 2011 Outline Emotion Detection Overview EEG for Emotion Detection Previous
ArteSImit: Artefact Structural Learning through Imitation
 ArteSImit: Artefact Structural Learning through Imitation (TU München, U Parma, U Tübingen, U Minho, KU Nijmegen) Goals Methodology Intermediate goals achieved so far Motivation Living artefacts will critically
ArteSImit: Artefact Structural Learning through Imitation (TU München, U Parma, U Tübingen, U Minho, KU Nijmegen) Goals Methodology Intermediate goals achieved so far Motivation Living artefacts will critically
THE SCIENCE of WEB PSYCHOLOGY ONLINE THE WEB PSYCHOLOGIST LTD.
 WEB PSYCHOLOGY THE SCIENCE of ONLINE PERSUASION NATHALIE NAHAI @NathalieNahai TheWebPsychologist.com WEB PSYCHOLOGY The empirical study of how our online environments influence our attitudes and behaviours
WEB PSYCHOLOGY THE SCIENCE of ONLINE PERSUASION NATHALIE NAHAI @NathalieNahai TheWebPsychologist.com WEB PSYCHOLOGY The empirical study of how our online environments influence our attitudes and behaviours
PAPER Baby steps: investigating the development of perceptual motor couplings in infancy
 Developmental Science 18:2 (2015), pp 270 280 DOI: 10.1111/desc.12226 PAPER Baby steps: investigating the development of perceptual motor couplings in infancy Carina C.J.M. de Klerk, 1 Mark H. Johnson,
Developmental Science 18:2 (2015), pp 270 280 DOI: 10.1111/desc.12226 PAPER Baby steps: investigating the development of perceptual motor couplings in infancy Carina C.J.M. de Klerk, 1 Mark H. Johnson,
Temporal Context and the Recognition of Emotion from Facial Expression
 Temporal Context and the Recognition of Emotion from Facial Expression Rana El Kaliouby 1, Peter Robinson 1, Simeon Keates 2 1 Computer Laboratory University of Cambridge Cambridge CB3 0FD, U.K. {rana.el-kaliouby,
Temporal Context and the Recognition of Emotion from Facial Expression Rana El Kaliouby 1, Peter Robinson 1, Simeon Keates 2 1 Computer Laboratory University of Cambridge Cambridge CB3 0FD, U.K. {rana.el-kaliouby,
Behavioural Brain Research
 Behavioural Brain Research 226 (2012) 309 316 Contents lists available at ScienceDirect Behavioural Brain Research j ourna l ho mepage: www.elsevier.com/locate/bbr Research report EEG mu component responses
Behavioural Brain Research 226 (2012) 309 316 Contents lists available at ScienceDirect Behavioural Brain Research j ourna l ho mepage: www.elsevier.com/locate/bbr Research report EEG mu component responses
Music-induced Emotions and Musical Regulation and Emotion Improvement Based on EEG Technology
 Music-induced Emotions and Musical Regulation and Emotion Improvement Based on EEG Technology Xiaoling Wu 1*, Guodong Sun 2 ABSTRACT Musical stimulation can induce emotions as well as adjust and improve
Music-induced Emotions and Musical Regulation and Emotion Improvement Based on EEG Technology Xiaoling Wu 1*, Guodong Sun 2 ABSTRACT Musical stimulation can induce emotions as well as adjust and improve
Cognitive Modelling Themes in Neural Computation. Tom Hartley
 Cognitive Modelling Themes in Neural Computation Tom Hartley t.hartley@psychology.york.ac.uk Typical Model Neuron x i w ij x j =f(σw ij x j ) w jk x k McCulloch & Pitts (1943), Rosenblatt (1957) Net input:
Cognitive Modelling Themes in Neural Computation Tom Hartley t.hartley@psychology.york.ac.uk Typical Model Neuron x i w ij x j =f(σw ij x j ) w jk x k McCulloch & Pitts (1943), Rosenblatt (1957) Net input:
Supplementary Motor Area exerts Proactive and Reactive Control of Arm Movements
 Supplementary Material Supplementary Motor Area exerts Proactive and Reactive Control of Arm Movements Xiaomo Chen, Katherine Wilson Scangos 2 and Veit Stuphorn,2 Department of Psychological and Brain
Supplementary Material Supplementary Motor Area exerts Proactive and Reactive Control of Arm Movements Xiaomo Chen, Katherine Wilson Scangos 2 and Veit Stuphorn,2 Department of Psychological and Brain
Positive effects of neurofeedback on autism symptoms correlate with brain activation during imitation and observation
 European Journal of Neuroscience, Vol. 47, pp. 579 591, 2018 doi:10.1111/ejn.13551 Positive effects of neurofeedback on autism symptoms correlate with brain activation during imitation and observation
European Journal of Neuroscience, Vol. 47, pp. 579 591, 2018 doi:10.1111/ejn.13551 Positive effects of neurofeedback on autism symptoms correlate with brain activation during imitation and observation
EEG MU RHYTHMS DURING ACTION
 RESEARCH ARTICLE Received: 18.12.2015 Accepted: 28.06.2016 A Study Design B Data Collection C Statistical Analysis D Data Interpretation E Manuscript Preparation F Literature Search G Funds Collection
RESEARCH ARTICLE Received: 18.12.2015 Accepted: 28.06.2016 A Study Design B Data Collection C Statistical Analysis D Data Interpretation E Manuscript Preparation F Literature Search G Funds Collection
This article appeared in a journal published by Elsevier. The attached copy is furnished to the author for internal non-commercial research and
 This article appeared in a journal published by Elsevier. The attached copy is furnished to the author for internal non-commercial research and education use, including for instruction at the authors institution
This article appeared in a journal published by Elsevier. The attached copy is furnished to the author for internal non-commercial research and education use, including for instruction at the authors institution
Top-down Modulation of Motor Priming by Belief about Animacy
 Top-down Modulation of Motor Priming by Belief about Animacy ROMAN LIEPELT 1, & MARCEL BRASS 1,2 1 Max Planck Institute for Human Cognitive and Brain Sciences, Department of Psychology, Leipzig, Germany
Top-down Modulation of Motor Priming by Belief about Animacy ROMAN LIEPELT 1, & MARCEL BRASS 1,2 1 Max Planck Institute for Human Cognitive and Brain Sciences, Department of Psychology, Leipzig, Germany
Mirror neurons in the infant brain are formed by the interactions between self and other. M. Iacoboni SELF AND OTHER. J. A. Pineda COGS171 UCSD
 Mirror neurons in the infant brain are formed by the interactions between self and other. M. Iacoboni SELF AND OTHER J. A. Pineda COGS171 UCSD What is the SELF? There are multiple attributes of SELF including
Mirror neurons in the infant brain are formed by the interactions between self and other. M. Iacoboni SELF AND OTHER J. A. Pineda COGS171 UCSD What is the SELF? There are multiple attributes of SELF including
To point a finger: Attentional and motor consequences of observing pointing movements
 Available online at www.sciencedirect.com Acta Psychologica 128 (2008) 56 62 www.elsevier.com/locate/actpsy To point a finger: Attentional and motor consequences of observing pointing movements Artem V.
Available online at www.sciencedirect.com Acta Psychologica 128 (2008) 56 62 www.elsevier.com/locate/actpsy To point a finger: Attentional and motor consequences of observing pointing movements Artem V.
Neuropsychologia 50 (2012) Contents lists available at SciVerse ScienceDirect. Neuropsychologia
 Neuropsychologia 50 (2012) 2745 2751 Contents lists available at SciVerse ScienceDirect Neuropsychologia journal homepage: www.elsevier.com/locate/neuropsychologia Sensitivity of alpha and beta oscillations
Neuropsychologia 50 (2012) 2745 2751 Contents lists available at SciVerse ScienceDirect Neuropsychologia journal homepage: www.elsevier.com/locate/neuropsychologia Sensitivity of alpha and beta oscillations
Kinematic cues in perceptual weight judgement and their origins in box lifting
 Psychological Research (2007) 71: 13 21 DOI 10.1007/s00426-005-0032-4 ORIGINAL ARTICLE A. F. de C. Hamilton Æ D. W. Joyce Æ J. R. Flanagan C. D. Frith Æ D. M. Wolpert Kinematic cues in perceptual weight
Psychological Research (2007) 71: 13 21 DOI 10.1007/s00426-005-0032-4 ORIGINAL ARTICLE A. F. de C. Hamilton Æ D. W. Joyce Æ J. R. Flanagan C. D. Frith Æ D. M. Wolpert Kinematic cues in perceptual weight
Physiology Unit 2 CONSCIOUSNESS, THE BRAIN AND BEHAVIOR
 Physiology Unit 2 CONSCIOUSNESS, THE BRAIN AND BEHAVIOR In Physiology Today What the Brain Does The nervous system determines states of consciousness and produces complex behaviors Any given neuron may
Physiology Unit 2 CONSCIOUSNESS, THE BRAIN AND BEHAVIOR In Physiology Today What the Brain Does The nervous system determines states of consciousness and produces complex behaviors Any given neuron may
fmri Activities in the Emotional Cerebellum: A Preference for Negative Stimuli and Goal-Directed Behavior
 DOI 10.1007/s12311-011-0301-2 fmri Activities in the Emotional Cerebellum: A Preference for Negative Stimuli and Goal-Directed Behavior Caroline K. L. Schraa-Tam & Willem J. R. Rietdijk & Willem J. M.
DOI 10.1007/s12311-011-0301-2 fmri Activities in the Emotional Cerebellum: A Preference for Negative Stimuli and Goal-Directed Behavior Caroline K. L. Schraa-Tam & Willem J. R. Rietdijk & Willem J. M.
Imitation: is cognitive neuroscience solving the correspondence problem?
 Imitation: is cognitive neuroscience solving the correspondence problem? Marcel Brass 1 and Cecilia Heyes 2 1 Department of Cognitive Neurology, Max Planck Institute for Human Cognitive and Brain Sciences,
Imitation: is cognitive neuroscience solving the correspondence problem? Marcel Brass 1 and Cecilia Heyes 2 1 Department of Cognitive Neurology, Max Planck Institute for Human Cognitive and Brain Sciences,
Brain Computer Interface. Mina Mikhail
 Brain Computer Interface Mina Mikhail minamohebn@gmail.com Introduction Ways for controlling computers Keyboard Mouse Voice Gestures Ways for communicating with people Talking Writing Gestures Problem
Brain Computer Interface Mina Mikhail minamohebn@gmail.com Introduction Ways for controlling computers Keyboard Mouse Voice Gestures Ways for communicating with people Talking Writing Gestures Problem
INTRODUCTION TO MIRROR NEURONS MARY ET BOYLE, PH.D. DEPARTMENT OF COGNITIVE SCIENCE UCSD
 INTRODUCTION TO MIRROR NEURONS MARY ET BOYLE, PH.D. DEPARTMENT OF COGNITIVE SCIENCE UCSD Announcements Midterm 1 Review Friday during Lecture Midterm 1 Exam February 5 Monday! During lecture come prepared
INTRODUCTION TO MIRROR NEURONS MARY ET BOYLE, PH.D. DEPARTMENT OF COGNITIVE SCIENCE UCSD Announcements Midterm 1 Review Friday during Lecture Midterm 1 Exam February 5 Monday! During lecture come prepared
Your support makes a vast difference to the success of our research and the health of our community.
 STEWARDSHIP REPORT THE 256-CHANNEL ELECTROENCEPHALOGRAM SYSTEM FOR SCHIZOPHRENIA RESEARCH 2015-2016 PROGRESS REPORT Prepared for the BC Schizophrenia Society Foundation By the UBC Faculty of Medicine May
STEWARDSHIP REPORT THE 256-CHANNEL ELECTROENCEPHALOGRAM SYSTEM FOR SCHIZOPHRENIA RESEARCH 2015-2016 PROGRESS REPORT Prepared for the BC Schizophrenia Society Foundation By the UBC Faculty of Medicine May
Mental representation of number in different numerical forms
 Submitted to Current Biology Mental representation of number in different numerical forms Anna Plodowski, Rachel Swainson, Georgina M. Jackson, Chris Rorden and Stephen R. Jackson School of Psychology
Submitted to Current Biology Mental representation of number in different numerical forms Anna Plodowski, Rachel Swainson, Georgina M. Jackson, Chris Rorden and Stephen R. Jackson School of Psychology
Attention Response Functions: Characterizing Brain Areas Using fmri Activation during Parametric Variations of Attentional Load
 Attention Response Functions: Characterizing Brain Areas Using fmri Activation during Parametric Variations of Attentional Load Intro Examine attention response functions Compare an attention-demanding
Attention Response Functions: Characterizing Brain Areas Using fmri Activation during Parametric Variations of Attentional Load Intro Examine attention response functions Compare an attention-demanding
Motor Theories of Cognition
 Motor Theories of Cognition In his Essay Towards a New Theory of Vision (1709) Berkeley uses a motor representation to describe depth perception. Motor theory in Movement and Mental Imagery (1916) by Margeret
Motor Theories of Cognition In his Essay Towards a New Theory of Vision (1709) Berkeley uses a motor representation to describe depth perception. Motor theory in Movement and Mental Imagery (1916) by Margeret
The Effect of Motor Learning of Serial Reaction Time Task (SRTT) Through Action Observation on Mu Rhythm and Improvement of Behavior Abilities
 Elmer Original Article ress The Effect of Motor Learning of Serial Reaction Time Task (SRTT) Through Action Observation on Mu Rhythm and Improvement of Behavior Abilities Sang-Yeol Lee a, f, Sung-Soo Bae
Elmer Original Article ress The Effect of Motor Learning of Serial Reaction Time Task (SRTT) Through Action Observation on Mu Rhythm and Improvement of Behavior Abilities Sang-Yeol Lee a, f, Sung-Soo Bae
Reading Minds. Mary ET Boyle, Ph.D. Department of Cognitive Science, UCSD
 Reading Minds g Mary ET Boyle, Ph.D. Department of Cognitive Science, UCSD Complexity of Social Cognition orrelaon across primate species between the size of their social group and the relative volume
Reading Minds g Mary ET Boyle, Ph.D. Department of Cognitive Science, UCSD Complexity of Social Cognition orrelaon across primate species between the size of their social group and the relative volume
