Resistance to Antiangiogenic Therapies by Metabolic Symbiosis in Renal Cell Carcinoma PDX Models and Patients
|
|
|
- Tyler Melton
- 5 years ago
- Views:
Transcription
1 Cell Reports Report Resistance to Antiangiogenic Therapies by Metabolic Symbiosis in Renal Cell Carcinoma PDX Models and Patients Gabriela Jiménez-Valerio, 1 Mar Martínez-Lozano, 1 Nicklas Bassani, 1 August Vidal, 2 María Ochoa-de-Olza, 3,4 Cristina Suárez, 4 Xavier García-del-Muro, 3 Joan Carles, 4 Francesc Viñals, 1,5 Mariona Graupera, 6 Stefano Indraccolo, 7 and Oriol Casanovas 1, * 1 Tumor Angiogenesis Group, ProCURE, Catalan Institute of Oncology - IDIBELL, Barcelona, Spain 2 Anatomopathology Service, Bellvitge University Hospital, Barcelona, Spain 3 Medical Oncology Service, Catalan Institute of Oncology, Barcelona, Spain 4 Vall d Hebron Institute of Oncology, Vall d Hebron University Hospital, Barcelona, Spain 5 Departament de Ciències Fisiològiques II, Campus de Bellvitge, Universitat de Barcelona, Barcelona, Spain 6 Tumor Angiogenesis Group, Bellvitge Biomedical Research Institute (IDIBELL), Barcelona, Spain 7 Istituto Oncologico Veneto - IRCCS, Padova, Italy *Correspondence: ocasanovas@iconcologia.net SUMMARY Antiangiogenic drugs are used clinically for treatment of renal cell carcinoma (RCC) as a standard first-line treatment. Nevertheless, these agents primarily serve to stabilize disease, and resistance eventually develops concomitant with progression. Here, we implicate metabolic symbiosis between tumor cells distal and proximal to remaining vessels as a mechanism of resistance to antiangiogenic therapies in patient-derived RCC orthoxenograft (PDX) models and in clinical samples. This metabolic patterning is regulated by the mtor pathway, and its inhibition effectively blocks metabolic symbiosis in PDX models. Clinically, patients treated with antiangiogenics consistently present with histologic signatures of metabolic symbiosis that are exacerbated in resistant tumors. Furthermore, the mtor pathway is also associated in clinical samples, and its inhibition eliminates symbiotic patterning in patient samples. Overall, these data support a mechanism of resistance to antiangiogenics involving metabolic compartmentalization of tumor cells that can be inhibited by mtor-targeted drugs. INTRODUCTION Angiogenesis is a crucial step in neoplastic progression that provides vascular support to the growing cancer mass while allowing for tumor cell dissemination and metastasis. A number of anti-angiogenic drugs are approved for clinical use in several types of cancers (Folkman, 2007). Nevertheless, antiangiogenics are typically not able to eliminate all tumor cells, giving raise to tumor relapse. Therefore, clinical responses achieved with these types of agents are moderate increases in survival, and are limited by therapeutic resistance that produces long-term failure of these treatments (Kerbel and Folkman, 2002). In the case of renal cell carcinoma (RCC), antiangiogenic drugs that block the vascular endothelial growth factor (VEGF)/VEGF receptor (VEGFR) pathway are now standard first-line treatment in metastatic RCC and are used sequentially to prolong clinical benefit in patients with recurrent disease (Rini, 2009). Nevertheless, resistance to therapy ultimately emerges in most patients, and further understanding of the underlying biology and potential therapeutic targets are urgently needed in the clinic (Rini and Atkins, 2009). Several different mechanisms of resistance to antiangiogenic therapies have been described (Bergers and Hanahan, 2008). Tumors may activate alternative pro-angiogenic signals that produce revascularization and facilitate tumor regrowth (Casanovas et al., 2005). Less is known about alternative modes of resistance to antiangiogenic therapies that do not involve revascularization, where tumors adapt to hypoxic conditions for survival and tumor growth. Indeed, intratumor hypoxia induced by antiangiogenic factors produces an accumulation of tumor intrinsic and tumor microenvironmental modifications that enhance survival, including metabolic adaptations to survive in low oxygen and low nutrient conditions (De Bock et al., 2011). Tumor cells have demanding metabolic requirements for their high proliferative rate in an acidic and nutrient-depleted environment. This lack of proper oxygen and nutrient supply affects tumor cell metabolism, and nutrient sensing pathways and transporters are altered to support tumor cell survival (Schulze and Harris, 2012). A particular form of adaptation to hypoxia is metabolic symbiosis, where there is a coordinated compartmentalization of tumor cells and their use of glucose and lactate (Sonveaux et al., 2008). In severely hypoxic regions tumor cells import and metabolize glucose by anaerobic glycolysis with upregulation of glucose transporter GLUT1 and lactate transporter MCT4 (Ullah et al., 2006). On the other hand, in normoxic regions, oxidative metabolism is permitted and these tumor cells are typically lactate- and glutamine-avid, and express lactate 1134 Cell Reports 15, , May 10, 2016 ª 2016 The Authors This is an open access article under the CC BY-NC-ND license (
2 (legend on next page) Cell Reports 15, , May 10,
3 transporters such as MCT1 (Sonveaux et al., 2008). This allows for the mutual survival of these two tumor regions by compartmentalizing glycolysis and oxidative lactate metabolism at different areas that exchange glucose and lactate for their metabolic symbiosis. Metabolic symbiosis has recently been described in several mouse models of cancer in response to potent angiogenesis inhibitors (Pisarsky et al., 2016, and Allen et al., 2016, in this issue of Cell Reports) but there has not been confirmatory assessment in the clinical setting with human patient samples. Such validation will be necessary to establish this biological phenomenon as a bona fide mode of adaptive resistance to anti-angiogenic therapy, the mechanism of which might suggest new therapeutic targets aimed to circumvent it. Here, we describe a mechanism of resistance to antiangiogenics in patient-derived orthoxenograft models of RCC that implicates metabolic symbiosis between tumor cells that can be blocked by mtor inhibitors. Importantly, these results are supported by analysis of clinical biopsies from patients with treated RCC, where antiangiogenics produce a metabolic symbiosis pattern that is similarly suggested to be mediated by mtor pathway. RESULTS Resistance to Antiangiogenics in Patient-Derived Renal Cell Carcinoma Orthoxenograft Models To investigate tumor adaptation to anti-angiogenic treatments, we developed a clinically relevant mouse model based on the orthotopic implantation of primary biopsies of human clear-cell RCC tumors, in which we evaluated response and acquired resistance after VEGFR signaling inhibition with sunitinib. All orthoxenograft tumors derived from primary biopsies initially responded to sunitinib antiangiogenic therapy, but eventually the tumors rebounded, having adapted to treatment with sunitinib (Figure 1A). Notably, each of these PDX models demonstrated a distinctive efficacy of sunitinib at inhibiting tumor growth, but this therapeutic phase was always followed by tumor regrowth and resistance. Furthermore, these impairments of tumor growth translated into extensions both of time-to-progression and lifespan (overall survival) in treated animals (Figure 1B). To explore the mechanism underlying this resistance, we first evaluated the histological features typically altered by antiangiogenic treatment. In resistant tumors, microvessel density was diminished, and both tumor hypoxia (Pimonidazole adducts and GLUT1 membrane accumulation) and tumor necrosis were consistently increased (Figures 1C 1E). Nevertheless, tumor proliferation was maintained in the viable areas of these resistant tumors, indicating that tumor cells circumvented the therapeutic pressure to continue proliferating (Figure 1C). These results coordinately describe a mechanism of resistance with reduced vascular density, without evident revascularization (Figure S1), wherein tumor cells adapted to treatment-induced hypoxia and were able to maintain proliferation and continuation of growth. Antiangiogenic Resistance Is Associated with a Metabolic Symbiosis Phenotype RCC typically presents with von Hippel Lindau (VHL) mutations that produce chronic hypoxic signaling cascades due to HIF1a accumulation (Cancer Genome Atlas Research Network, 2013). Nevertheless, these tumors further respond to antiangiogenic therapies with an enhanced hypoxia response transcriptional program. Indeed, VHL-mutated RCC tumor cells further elevate the transcriptional hypoxia response program when cultured under hypoxia and partial nutrient depletion conditions that mimic antiangiogenic therapy in full tumors (Figures S2A S2C). Furthermore, these cells increase glucose consumption and lactate production under hypoxic conditions (Figure S2D). Consistently, in our orthoxenograft models Ren28 and Ren96, the expression of two hypoxia response transporters, glucose transporter GLUT1 and the lactate exporter MCT4, were increased by antiangiogenic treatment and accumulated distinctively in peri-necrotic areas where the maximal hypoxia is present (Figure 2A; Figure S3). Provocatively, the lactate importer MCT1 was expressed in other non-overlapping regions of sunitinib-treated tumors, separate from the hypoxic and necrotic regions (Figure 2A; Figure S3). This mutually exclusive localization of MCT1 and MCT4 is reminiscent of metabolic symbiosis, a form of coordinated compartmentalization of tumor cells and their use of glucose and lactate (Sonveaux et al., 2008). Indeed, sunitinib-resistant tumors showed a statistically significant increase in expression area of both MCT1 and MCT4, but more importantly, their pattern of expression changed from a low co-expression in controls to a mutually exclusive compartmentalized symbiosis pattern (Figure 2B). Specifically, MCT4-positive regions were far from blood vessels (>40 mm in both Ren28 and Ren96 models), but MCT1-positive regions were close to vessels (mostly <30 mm in both models) (Figure S4). Quantification of the incidence of this symbiosis pattern further demonstrated the significant adaptive change of the compartmentalization in resistant tumors. While in control tumors the metabolic symbiosis pattern was completely absent, the majority of sunitinib-resistant tumors showed a clearly defined compartmentalized pattering (87%) (Figure 2C). In a related paper in this issue of Cell Reports, Allen et al. (2016) describe the association of mtor signaling pathway activity in analogous symbiotic clusters in a mouse model of pancreatic neuroendocrine cancer (PanNET). Thus, we evaluated mtor pathway activity by means of phospho-s6 and phospho-s6k in the resistant RCC tumors. Immunofluorescence staining showed these mtor activity markers accumulated specifically in tumor regions close to vessels, co-localized with MCT1 and Figure 1. Sunitinib Treatment Effects in Patient-Derived RCC Orthoxenografts (A) Tumor progression with sunitinib treatment in Ren28 and Ren96 RCC PDX tumors starting at 1,000 mm 3, and (B) corresponding Kaplan-Meier survival curves. (C) Immunohistological staining of control and sunitinib-treated mice for CD31 (403), pimonidazole adducts in green (PIMO) counterstained with DAPI (203), GLUT1 (203), and Ki67 (203). (D and E) Quantification of microvessel density (D) and H&E and necrosis (E) (percentage of area) quantification graphed as box plots from four animals per treatment group, four images each Cell Reports 15, , May 10, 2016
4 (legend on next page) Cell Reports 15, , May 10,
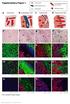
5 were mutually exclusive with MCT4 areas (Figure 2D; data not shown). These data clearly demonstrate that mtor activity was restricted to the MCT1-expressing normoxic compartment in sunitinib-resistant tumors. Blockade of Metabolic Symbiosis by Inhibition of mtor pathway To determine the functional implication of mtor signaling in the metabolic symbiosis pattern, sunitinib-resistant tumors were treated with a clinically approved mtor inhibitor, everolimus. The mtor inhibitor demonstrated antitumor efficacy in this second-line treatment, effectively blocking the adaptive resistance to antiangiogenic therapy in Ren28, but not in the Ren96 PDX model (Figure 3A). Similarly to the clinical setting, second-line everolimus in patients has a variable efficacy, with a proportion of patients not responding to this therapy (upfront refractory or intrinsic resistant) (Motzer et al., 2014). To confirm mtor pathway inhibition in everolimus-treated PDX tumors, immunofluorescent staining for the p-s6 and p-s6k markers demonstrated that everolimus was indeed effectively inhibiting mtor activity in Ren28, but it was not active in Ren96 (Figure 3B). This suggests that Ren96 PDX model could have a signaling misregulation or mutation that renders it insensitive to everolimus treatment. When the metabolic symbiosis phenotype was evaluated in both tumor models, everolimus treatment produced a substantial alteration in the expression area of the lactate transporters MCT1 and MCT4 only in Ren28, with widespread accumulation of MCT4, concomitant with reductions in MCT1- positive regions (Figure 3B). Quantification of these regions confirmed that everolimus treatment in Ren28 served to unbalance the MCT1/MCT4 symbiotic equilibrium in favor of MCT4-expressing areas, reducing MCT1-positive tumor areas (Figure 3C). On the contrary, everolimus treatment did not disrupt the sunitinib-resistance symbiotic pattern in the insensitive Ren96 tumors (Figure 3D). This correlation suggests that everolimus-induced disruption of the metabolic symbiosis phenotype is fully dependent on everolimus efficacy in inhibiting mtor pathway. To pinpoint the mechanism of everolimus decrease in MCT1-positive areas, an in vitro study was performed with RCC cells. Everolimus treatment decreased tumor cell viability by 53% (Figure S5), indicating that cell survival rather than a transcriptional downregulation of MCT1 was occurring in these cells. Thus, metabolic symbiosis abolishment by everolimus is associated to decreased viability of this particular tumor cell subpopulation. Overall, the data implicate the mtor pathway in this sunitinib resistance mechanism by aiding the induction of the perivascular p-s6/mct1-positive subcompartment. Furthermore, when mtor is effectively inhibited, sunitinib resistance is blocked, evidently disrupting the metabolic symbiosis phenotype via elimination of the ps6/mct1-positive subpopulation in favor of the GLUT1/MCT4-positive subpopulation. Metabolic Symbiosis in RCC Patients after Antiangiogenics To further assess the metabolic symbiosis phenotype observed in orthoxenograft models, we evaluated a series of 15 patients with clear-cell RCC tumors who had been treated with antiangiogenic therapy blocking the VEGF/VEGFR pathway (Figure S6A). This series is unique because we obtained paired FFPE tumor specimens before and after antiangiogenic treatment (either in response or resistance), therefore allowing determination of histopathological changes in each patient s tumor pre- and post-antiangiogenic treatment. Immunofluorescence staining of lactate transporters in samples before and after antiangiogenic treatment demonstrated that area with expression of both MCT1 and MCT4 were significantly increased upon treatment (Figure 4A, left; Figure S6B), suggestive of a metabolic adaptation to treatment involving upregulation of lactate transporters, similar to that observed in the tumor models. Importantly, localization of these transporters also occurred in a compartmental, mutually exclusive pattern, with MCT1 expressed in well-oxygenated areas close to vessels, and MCT4 expressed in more hypoxic (GLUT1 in the membrane) areas distal to tumor vessels (Figure S6C). Upon quantification, this patterning was observed in 71% of the post-treatment samples but completely absent in the pre-treatment samples, indicating the strong adaptive nature of this event (Figure 4A, right). This patterning strongly suggests a metabolic symbiosis phenotype in patients, and is fully consistent with findings in orthoxenograft tumor models. Due to the differential clinical responses to treatment in each of the 15 patients in study, they were grouped according to their response or resistance to treatment (clinically stable disease, SD versus progressive disease, PD) at the end of antiangiogenic therapy, when the post- sample was obtained. Tumors from responsive (n = 6) and resistant (n = 8) patients to antiangiogenic treatment both expressed MCT1 and MCT4 after treatment (Figure 4B). Notably, however, only tumors from patients who progressed (having acquired a resistant phenotype) had a clearly defined compartmentalization of MCT1 and MCT4, consistent with metabolic symbiosis patterning (Figure 4B). Quantification revealed that metabolic symbiosis patterning was observed in 100% of the post-samples from resistant tumors, whereas in patients still in the response phase (SD) the incidence was 33% (Figure 4C). Thus, this singular series of treated and biopsied patients supports the proposition that antiangiogenic resistance involves the formation of two mutually exclusive tumor cell Figure 2. Metabolic Symbiosis of Sunitinib-Treated Tumors (A) Immunohistofluorescence staining of Ren28 and Ren96 RCC PDX tumors showing representative images of MCT1, GLUT1, MCT4, and CD31, counterstained with DAPI (203 magnifications). (B) Quantification of expression of MCT1 and MCT4 in Ren28 and Ren96 as a percentage of positive area per field, from four animals per treatment group, four images each. (C) Quantification of metabolic symbiosis pattern in Ren28- and Ren96-treated tumors as presence of MCT1/MCT4 mutually exclusive pattern (symbiosis pattern) versus homogeneous, co-staining (no symbiosis pattern). (D) Representative images of ps6, MCT1, and MCT4 immunohistofluorescence staining in RCC PDX tumors Cell Reports 15, , May 10, 2016
6 (legend on next page) Cell Reports 15, , May 10,
7 compartments, a perivascular region and MCT1 expression, and another peri-necrotic region expressing MCT4 and a GLUT1/ hypoxia response. Motivated by the orthoxenograft results, mtor pathway involvement was also evaluated. The phospho-s6 mtor activity marker was also compartmentalized in post-treatment samples and, similar to orthoxenograft results, it was expressed in a mutually exclusive pattern with MCT4 lactate exporter (Figure 4D). Whereas our series of pre- and post-samples were from antiangiogenic-treated patients, we obtained samples from one patient with RCC that was effectively treated with mtor inhibitor (second line) just after antiangiogenics (first line). In this patient (patient no. 12), only MCT4 expression was detected after progression to mtor inhibitor treatment, and there was no compartmentalization patterning (Figure 4E). Although these data are from one individual, the results are fully consistent with animal models where mtor inhibition selectively eliminated the MCT1 compartment, leaving the MCT4-positive tumor compartment comparatively intact. Collectively, this singular set of sunitinib-treated and successively biopsied patients supports the hypothesis that antiangiogenic treatment induces a metabolic symbiosis patterning in RCC that is further exacerbated in progressive (resistant) tumors. Furthermore, the mtor pathway is also implicated as involved in inducing and maintaining the MCT1-positive compartment in the metabolic symbiosis. DISCUSSION Our study, together with Pisarsky et al. (2016) and Allen et al. (2016), establish in mouse models of three different tumor types a new mode of resistance to antiangiogenic therapies, involving metabolic symbiosis among differential subcompartments of tumor cells. Indeed, metabolic symbiosis has been described as a mode of adaptation to tumor hypoxia (Sonveaux et al., 2008), but here we coordinately describe this phenomenon as an active mechanism of resistance to potent antiangiogenic therapies in three different tumor types (pancreatic neuroendocrine tumor, breast cancer, and RCC). Several different modes of resistance to antiangiogenic therapies have been described, including the induction of revascularization and reoxygenation of tumors by different mechanisms (Bergers and Hanahan, 2008; Casanovas et al., 2005). Nevertheless, here we describe a mechanism of resistance in the context of continuing suppression of tumor neo-vascularization, whereby tumor cells adapt to treatment-induced hypoxia to enable continuing proliferation and growth. This adaptation involves a functional compartmentalization of energy metabolism previously described as metabolic symbiosis by Dewhirst and colleagues (Sonveaux et al., 2008). This is a form of coordinated compartmentalization of tumor cells and their differential use of glucose and lactate that produces a mutually exclusive patterning of MCT1-expressing cells and MCT4. This is functionally relevant as the hypoxic and peri-necrotic region upregulate hypoxia-response genes (GLUT1, PDK1, GAPDH, etc.) to activate anaerobic glycolysis and export accumulated lactate to the extracellular space via MCT4 lactate/h+ symporter. On the other hand, the perivascular compartment is better oxygenated and permits oxidative phosphorylation, for which it imports accumulated lactate to be further metabolized aerobically to obtain energy (Sonveaux et al., 2008). With these data, a more clinically relevant question emerges: Is there a way to block this metabolic symbiosis phenotype to therapeutically attack these antiangiogenic-resistant tumors? Consistent with Allen et al. (2016), we have produced evidence that the mtor signaling pathway has an important role in this symbiotic compartmentalization, and it can be therapeutically targeted. mtor activity markers reveal that this pathway is restricted to better oxygenated MCT1-positive areas, close to vessels, consistent with the report by Allen et al. (2016) Thus, therapeutic inhibition of this pathway with mtor (mtorc1) inhibitors such as everolimus and rapamycin disrupts the symbiotic pattern and unbalances this equilibrium by eliminating the MCT1-positive areas, accumulating glycolytic MCT4-positive tumor cells, and increasing necrosis by oxygen and nutrient depletion. The result is anti-tumor therapeutic benefit, and suggests everolimus as a useful second-line treatment after antiangiogenic therapy. Indeed, this is a typical sequence of treatment in metastatic RCC, where the standard first-line treatment is with antiangiogenic drugs and, upon resistance (disease progression), follow-up treatment with everolimus (RECORD trials) (Motzer et al., 2014). An important shortcoming in the concurrent metabolic symbiosis studies is that they are restricted to animal model studies, such that it remained to be determined whether this adaptive resistance mechanism was operative in human cancer treated with anti-angiogenic drugs. Here, we present the first validation of metabolic symbiosis in a series of 15 patients with clear cell RCC tumors treated with antiangiogenic therapies. By obtaining a set of uniquely paired FFPE tumor specimens before and after antiangiogenic treatment, we detected compartmentalization upon treatment, with mutually exclusive patterns of perivascular MCT1 and peri-necrotic/hypoxic MCT4 areas, strongly suggestive of a metabolic symbiosis phenotype in patients. Furthermore, this patterning was most prominent in tumors that had adapted and progressed on therapy, consistent with metabolic Figure 3. Everolimus Effects as a Second-Line Treatment in Patient-Derived RCC Orthoxenografts (A) Tumor progression of treatment with sunitinib or sunitinib followed by everolimus in Ren28 and Ren96 RCC PDX tumors starting at 1,000 mm 3, and Corresponding Kaplan-Meier survival curves. (B) Representative images of ps6 and S6kinase immunohistochemistry staining, and ps6, MCT4, MCT1, and MCT4 immunohistofluorescence staining, counterstained with DAPI (203 magnifications). (C) Quantification of expression MCT1 and MCT4 in Ren28 and Ren96 in the different treatments as percentage of positive area per field, from four animals per treatment group, four images each. (D) Quantification of metabolic symbiosis pattern in Ren28- and Ren96-treated tumors as presence of MCT1/MCT4 mutually exclusive pattern (symbiosis pattern) versus homogeneous, co-staining (no symbiosis pattern) Cell Reports 15, , May 10, 2016
8 (legend on next page) Cell Reports 15, , May 10,
9 symbiosis patterning occurring in and contributing to the resumed progression in resistant tumors. Moreover, we have implicated involvement of the mtor pathway in the adaptive resistance via metabolic compartmentalization in patient tumors, where the perivascular subcompartment shows mtor signaling activity and MCT1 expression. Furthermore, mtor inhibition in a patient further substantiates the promising value of targeting mtor to block metabolic symbiosis and overall resistance to antiangiogenics in patients. In summary, our study describes a mode of resistance to antiangiogenic therapies in orthotopic RCC PDX mouse models and in human patients, involving metabolic symbiosis compartmentalization of tumors. This resistance mechanism can be blocked therapeutically with mtor inhibitors that disrupt the symbiosis by eliminating one of the symbiotic compartments, which may be important for the benefits of this drug when used second line after resistance to angiogenesis inhibitors. EXPERIMENTAL PROCEDURES Patient-Derived Orthoxenograft Models from RCC Human Biopsies Fresh surgical specimens of RCC were obtained from the Bellvitge and Vall d Hebron Hospitals under local ethics committee s approved protocols (CEIC approvals ref. PR322/11 and PR[AG]240/2013). Their surgical implantation was done orthotopically in the kidney (original neoplastic organ) in male athymic nude mice (Harlan Laboratories). All animal experiments were developed according to our Institute s Animal Research Committee acceptance, and following Spanish laws and European directives on ethical usage of rodents for animal research (approval DARP-#4899). Treatment Schedule Once the tumor was palpable (1,000 mm 3 of volume), five to six animals were randomized to receive treatments. Sunitinib (LC Labs) was prepared and dosed at 40 mg/kg/day orally as described (Pàez-Ribes et al., 2009). Everolimus (LC Labs) was prepared and dosed at 10 mg/kg/day orally. At the end of treatment, mice were killed, and the tumor tissue was weighed and processed for histology. Immunohistology and Quantifications Frozen or FFPE tumor sections were stained with H&E and immunohistology techniques as described (Pàez-Ribes et al., 2009). Primary antibodies were rabbit anti-glut1 (Abcam, ab652), mouse anti-cd31 (Abcam, ab28364), rabbit anti-ki67 (Thermo Sci., RM9106 S-1), anti-ps6 (Cell Signaling, 4857), antips6k (Abcam, ab32359), rabbit anti-glut1 (Abcam ab652), rabbit anti-ps6 (Cell Signaling, 4857), rabbit anti-mct1 (H-70) (SCB, SC-50324), rabbit anti- MCT4 (H-90) (SCB, SC-50329), and anti-pimonidazole (NPI, ). Double or triple stainings with same-species antibodies were done with tyramide followed by fixation as described. To quantify CD31, Ki67, and GLUT1 staining, four hotspot fields in viable tissue zones at 4003 magnification were captured for each tumor. MCT1/MCT4 area density quantification was done with positive-pixel-area/total-imagepixel-area, using ImageJ software. Hypoxia was detected with pimonidazole (intraperitoneal 60 mg/kg) 1 hr before heart perfusion, and immunodetected with histological techniques as described above. Patient Series and Clinical Data The series of pre- and post-samples from patients with RCC was gathered prospectively and retrospectively between 2007 and 2014 under clinical ethics committee approval (IDIBELL CEIC approval ref. PR322/11, and VHIO CEIC approval ref. PR[AG]240/2013), and acceptance by the Spanish Government (AEMPS). A total of 15 clear-cell RCC samples from previously untreated patients were selected, and pre-treatment and post-treatment biopsies and/or surgical specimens were collected (including four post-treatment autopsy samples). These patients were treated with antiangiogenic therapy (mostly sunitinib and a few bevacizumab at standard dose) and the response was evaluated by RECIST guidelines (tumor progression PD, resistance; or tumor response SD, in efficacy) at the end of antiangiogenic treatment. Statistical Analysis Statistical comparisons between treatment groups were done using the Mann-Whitney U test (2-tailed), performed in GraphPad Prism. Differences were considered statistically significant at p < Statistics coding: ****p < ; ***p < 0.001; **p < 0.01, and *p < SUPPLEMENTAL INFORMATION Supplemental Information includes Supplemental Experimental Procedures and six figures and can be found with this article online at /j.celrep AUTHOR CONTRIBUTIONS G.J.V., M.M.L., and N.B. conducted experiments and processed data. A.V., M.O., C.S., X.G.M., J.C., and S.I. contributed clinical material and processed data. F.V., M.G., and S.I. processed data. O.C. conceived the study, and O.C. and G.J.V. wrote the paper. ACKNOWLEDGMENTS We are immensely grateful to Douglas Hanahan (ISREC) and Lidia Moserle (IOV) for their insightful comments on the manuscript. We also thank Roser Pons and Elizabeth Allen for valuable technical assistance and comments. The research leading to these results has received funding from the European Research Council under the FP7/ ERC grant agreement no (STROMALIGN), Ministerio de Economía y Competitividad-Spain (SAF ) co-funded by FEDER / ERDF funds a way to build Europe, ISCIII-Spain (PIE13/ ), and AGAUR-Generalitat de Catalunya (SGR ). S.I. was supported by AIRC (project IG14295). Received: December 1, 2015 Revised: January 19, 2016 Accepted: March 29, 2016 Published: April 28, 2016 Figure 4. Metabolic Symbiosis after Antiangiogenic Treatment in RCC (A) Left, Quantification of expression MCT1 and MCT4 in RCC in basal levels (pre-treatment samples) versus application of the antiangiogenic therapy (posttreatment samples), represented as percentage of positive area per field. Right, quantification of metabolic symbiosis pattern in the same samples as described above. (B) Representative images of MCT1, MCT4, and CD31 immunohistofluorescence staining in RCC according clinical response, 203 DAPI counterstained. (C) Quantification of metabolic symbiosis pattern in RCC according to clinical response, basal levels (pre-treatment samples) versus post-antiangiogenic-treated samples. (D) Representative images of GLUT1, MCT1, ps6, and MCT4 immunohistofluorescence staining in RCC according to clinical response. (E) Schematic diagram of treatment schedule for a patient treatment sequence and immunofluorescence show a representative staining of MCT1, MCT4, and CD31 before and after antiangiogenic-positive mtor inhibition treatments Cell Reports 15, , May 10, 2016
10 REFERENCES Allen, E., Miéville, P., Warren, C.M., Saghafinia, S., Li, L., Peng, M.W., and Hanahan, D. (2016). Metabolic symbiosis enables adaptive resistance to antiangiogenic therapy that is dependent on mtor signaling. Cell Rep. 15, this issue, Bergers, G., and Hanahan, D. (2008). Modes of resistance to anti-angiogenic therapy. Nat. Rev. Cancer 8, Cancer Genome Atlas Research Network (2013). Comprehensive molecular characterization of clear cell renal cell carcinoma. Nature 499, Casanovas, O., Hicklin, D.J., Bergers, G., and Hanahan, D. (2005). Drug resistance by evasion of antiangiogenic targeting of VEGF signaling in late-stage pancreatic islet tumors. Cancer Cell 8, De Bock, K., Mazzone, M., and Carmeliet, P. (2011). Antiangiogenic therapy, hypoxia, and metastasis: risky liaisons, or not? Nat. Rev. Clin. Oncol. 8, Folkman, J. (2007). Angiogenesis: an organizing principle for drug discovery? Nat. Rev. Drug Discov. 6, Kerbel, R., and Folkman, J. (2002). Clinical translation of angiogenesis inhibitors. Nat. Rev. Cancer 2, Motzer, R.J., Barrios, C.H., Kim, T.M., Falcon, S., Cosgriff, T., Harker, W.G., Srimuninnimit, V., Pittman, K., Sabbatini, R., Rha, S.Y., et al. (2014). Phase II randomized trial comparing sequential first-line everolimus and second-line sunitinib versus first-line sunitinib and second-line everolimus in patients with metastatic renal cell carcinoma. J. Clin. Oncol. 32, Pàez-Ribes, M., Allen, E., Hudock, J., Takeda, T., Okuyama, H., Viñals, F., Inoue, M., Bergers, G., Hanahan, D., and Casanovas, O. (2009). Antiangiogenic therapy elicits malignant progression of tumors to increased local invasion and distant metastasis. Cancer Cell 15, Pisarsky, L., Bill, R., Fagiani, E., Dimeloe, S., Goosen, R.W., Hagmann, J., Hess, C., and Christofori, G. (2016). Targeting metabolic symbiosis to overcome resistance to anti-angiogenic therapy. Cell Rep. 15, this issue, Rini, B.I. (2009). Vascular endothelial growth factor-targeted therapy in metastatic renal cell carcinoma. Cancer 115 (10, Suppl), Rini, B.I., and Atkins, M.B. (2009). Resistance to targeted therapy in renal-cell carcinoma. Lancet Oncol. 10, Schulze, A., and Harris, A.L. (2012). How cancer metabolism is tuned for proliferation and vulnerable to disruption. Nature 491, Sonveaux, P., Végran, F., Schroeder, T., Wergin, M.C., Verrax, J., Rabbani, Z.N., De Saedeleer, C.J., Kennedy, K.M., Diepart, C., Jordan, B.F., et al. (2008). Targeting lactate-fueled respiration selectively kills hypoxic tumor cells in mice. J. Clin. Invest. 118, Ullah, M.S., Davies, A.J., and Halestrap, A.P. (2006). The plasma membrane lactate transporter MCT4, but not MCT1, is up-regulated by hypoxia through a HIF-1alpha-dependent mechanism. J. Biol. Chem. 281, Cell Reports 15, , May 10,
RIP-Tag2 mouse model as a Paradigm for Target. Search in NETs
 RIP-Tag2 mouse model as a Paradigm for Target Search in NETs Oriol Casanovas, Ph.D. Tumor Angiogenesis Group INSTITUT CATALÀ d ONCOLOGIA IDIBELL Barcelona (SPAIN) Therapeutic Targeting of the Tumor Stroma
RIP-Tag2 mouse model as a Paradigm for Target Search in NETs Oriol Casanovas, Ph.D. Tumor Angiogenesis Group INSTITUT CATALÀ d ONCOLOGIA IDIBELL Barcelona (SPAIN) Therapeutic Targeting of the Tumor Stroma
Antiangiogenic Therapy Elicits Malignant Progression of Tumors to Increased Local Invasion and Distant Metastasis
 Article Antiangiogenic Therapy Elicits Malignant Progression of Tumors to Increased Local Invasion and Distant Metastasis Marta Pàez-Ribes, 1,6 Elizabeth Allen, 2,6 James Hudock, 3 Takaaki Takeda, 4 Hiroaki
Article Antiangiogenic Therapy Elicits Malignant Progression of Tumors to Increased Local Invasion and Distant Metastasis Marta Pàez-Ribes, 1,6 Elizabeth Allen, 2,6 James Hudock, 3 Takaaki Takeda, 4 Hiroaki
Evidenze cliniche nel trattamento del RCC
 Criteri di scelta nel trattamento sistemico del carcinoma renale Evidenze cliniche nel trattamento del RCC Alessandro Morabito Unità Sperimentazioni Cliniche Istituto Nazionale Tumori di Napoli Napoli,
Criteri di scelta nel trattamento sistemico del carcinoma renale Evidenze cliniche nel trattamento del RCC Alessandro Morabito Unità Sperimentazioni Cliniche Istituto Nazionale Tumori di Napoli Napoli,
Mechanisms of Resistance to Antiangiogenic. Martin J. Edelman, MD University of Maryland Greenebaum Cancer Center Dresden, 2012
 Mechanisms of Resistance to Antiangiogenic Agents Martin J. Edelman, MD University of Maryland Greenebaum Cancer Center Dresden, 2012 Angiogenesis: A fundamental attribute of cancer Premise of Anti-angiogenic
Mechanisms of Resistance to Antiangiogenic Agents Martin J. Edelman, MD University of Maryland Greenebaum Cancer Center Dresden, 2012 Angiogenesis: A fundamental attribute of cancer Premise of Anti-angiogenic
Targeted and immunotherapy in RCC
 Targeted and immunotherapy in RCC Treatment options Surgery (radical VS partial nephrectomy) Thermal ablation therapy Surveillance Immunotherapy Molecular targeted therapy Molecular targeted therapy Targeted
Targeted and immunotherapy in RCC Treatment options Surgery (radical VS partial nephrectomy) Thermal ablation therapy Surveillance Immunotherapy Molecular targeted therapy Molecular targeted therapy Targeted
Immunotherapy versus targeted treatments in metastatic renal cell carcinoma: The return game?
 Immunotherapy versus targeted treatments in metastatic renal cell carcinoma: The return game? Sylvie NEGRIER MD, PhD Centre Léon Bérard, Lyon Université Lyon I IMMUNOTHERAPY: A LONG AND WIDING ROAD! WHERE
Immunotherapy versus targeted treatments in metastatic renal cell carcinoma: The return game? Sylvie NEGRIER MD, PhD Centre Léon Bérard, Lyon Université Lyon I IMMUNOTHERAPY: A LONG AND WIDING ROAD! WHERE
CLINICAL POLICY Department: Medical Management Document Name: Inlyta Reference Number: NH.PHAR.100 Effective Date: 05/12
 Page: 1 of 5 IMPORTANT REMINDER This Clinical Policy has been developed by appropriately experienced and licensed health care professionals based on a thorough review and consideration of generally accepted
Page: 1 of 5 IMPORTANT REMINDER This Clinical Policy has been developed by appropriately experienced and licensed health care professionals based on a thorough review and consideration of generally accepted
David N. Robinson, MD
 David N. Robinson, MD Background and Treatment of mrcc Background ~ 64,770 new cases of kidney/renal pelvis cancers will be diagnosed in the US in 2012 with an estimated 13,570 deaths [1] ~ 75% are clear-cell
David N. Robinson, MD Background and Treatment of mrcc Background ~ 64,770 new cases of kidney/renal pelvis cancers will be diagnosed in the US in 2012 with an estimated 13,570 deaths [1] ~ 75% are clear-cell
Clinical Biomarker in Kidney Cancer. Maria Nirvana Formiga, M.D., Ph.D.
 Clinical Biomarker in Kidney Cancer Maria Nirvana Formiga, M.D., Ph.D. Disclosures I am on the Speaker s Bureau with Pfizer and Bayer Clinical trials of BMS and Pfizer Kidney Cancer 70% new cases in developed
Clinical Biomarker in Kidney Cancer Maria Nirvana Formiga, M.D., Ph.D. Disclosures I am on the Speaker s Bureau with Pfizer and Bayer Clinical trials of BMS and Pfizer Kidney Cancer 70% new cases in developed
Sequential Therapy in Renal Cell Carcinoma*
 Sequential Therapy in Renal Cell Carcinoma* Bernard Escudier, MD, Marine Gross Goupil, MD, Christophe Massard, MD, and Karim Fizazi, MD, PhD Because of the recent approval of several drugs for the treatment
Sequential Therapy in Renal Cell Carcinoma* Bernard Escudier, MD, Marine Gross Goupil, MD, Christophe Massard, MD, and Karim Fizazi, MD, PhD Because of the recent approval of several drugs for the treatment
The Process of Angiogenesis & Inhibition of Angiogenesis and/or Lymphangiogenesis
 The Process of Angiogenesis & Inhibition of Angiogenesis and/or Lymphangiogenesis Nam Deuk Kim, Ph.D. Pusan National University Contents Part 1. The process of angiogenesis Part 2. The role of angiopoietins
The Process of Angiogenesis & Inhibition of Angiogenesis and/or Lymphangiogenesis Nam Deuk Kim, Ph.D. Pusan National University Contents Part 1. The process of angiogenesis Part 2. The role of angiopoietins
Have Results of Recent Randomized Trials Changed the Role of mtor Inhibitors?
 Have Results of Recent Randomized Trials Changed the Role of mtor Inhibitors? Bernard Escudier Institut Gustave Roussy Villejuif, France EIKCS Lyon April 2015 What is the current role of mtor inhibitors?
Have Results of Recent Randomized Trials Changed the Role of mtor Inhibitors? Bernard Escudier Institut Gustave Roussy Villejuif, France EIKCS Lyon April 2015 What is the current role of mtor inhibitors?
Biologics Effects of Targeted Therapeutics
 Report on the isbtc Mini-symposium on Biologics Effects of Targeted Therapeutics Michael B. Atkins, MD Beth Israel Deaconess Medical Center Louis Weiner, M.D. Fox Chase Cancer Center Report on the isbtc
Report on the isbtc Mini-symposium on Biologics Effects of Targeted Therapeutics Michael B. Atkins, MD Beth Israel Deaconess Medical Center Louis Weiner, M.D. Fox Chase Cancer Center Report on the isbtc
Supplementary Materials. for Garmy-Susini, et al, Integrin 4 1 signaling is required for lymphangiogenesis and tumor metastasis
 Supplementary Materials for Garmy-Susini, et al, Integrin 4 1 signaling is required for lymphangiogenesis and tumor metastasis 1 Supplementary Figure Legends Supplementary Figure 1: Integrin expression
Supplementary Materials for Garmy-Susini, et al, Integrin 4 1 signaling is required for lymphangiogenesis and tumor metastasis 1 Supplementary Figure Legends Supplementary Figure 1: Integrin expression
Supplemental Figure 1. Isolation and characterization of CD133+ neurosphere-like
 SUPPLEMENTL FIGURE LEGENDS Supplemental Figure 1. Isolation and characterization of CD133+ neurosphere-like spheroids from a human brain tumor sample or glioma xenograft. () CD133+ tumor cells isolated
SUPPLEMENTL FIGURE LEGENDS Supplemental Figure 1. Isolation and characterization of CD133+ neurosphere-like spheroids from a human brain tumor sample or glioma xenograft. () CD133+ tumor cells isolated
The Angiopoietin Axis in Cancer
 Ang2 Ang1 The Angiopoietin Axis in Cancer Tie2 An Overview: The Angiopoietin Axis Plays an Essential Role in the Regulation of Tumor Angiogenesis Growth of a tumor beyond a limiting size is dependent upon
Ang2 Ang1 The Angiopoietin Axis in Cancer Tie2 An Overview: The Angiopoietin Axis Plays an Essential Role in the Regulation of Tumor Angiogenesis Growth of a tumor beyond a limiting size is dependent upon
When to Treat Beyond Progression with Systemic Therapies? Manuela Schmidinger Medical University of Vienna, Austria
 When to Treat Beyond Progression with Systemic Therapies? Manuela Schmidinger Medical University of Vienna, Austria Is Treatment Beyond Progression a Valid Strategy? 1) NO YES? Is Treatment Beyond Progression
When to Treat Beyond Progression with Systemic Therapies? Manuela Schmidinger Medical University of Vienna, Austria Is Treatment Beyond Progression a Valid Strategy? 1) NO YES? Is Treatment Beyond Progression
Antiangiogenic Agents in NSCLC Where are we? Which biomarkers? VEGF Is the Only Angiogenic Factor Present Throughout the Tumor Life Cycle
 Antiangiogenic Agents in NSCLC Where are we? Which biomarkers? Martin Reck Department e t of Thoracic c Oncology ogy Hospital Grosshansdorf Germany VEGF Is the Only Angiogenic Factor Present Throughout
Antiangiogenic Agents in NSCLC Where are we? Which biomarkers? Martin Reck Department e t of Thoracic c Oncology ogy Hospital Grosshansdorf Germany VEGF Is the Only Angiogenic Factor Present Throughout
Supplementary Figure 1. Deletion of Smad3 prevents B16F10 melanoma invasion and metastasis in a mouse s.c. tumor model.
 A B16F1 s.c. Lung LN Distant lymph nodes Colon B B16F1 s.c. Supplementary Figure 1. Deletion of Smad3 prevents B16F1 melanoma invasion and metastasis in a mouse s.c. tumor model. Highly invasive growth
A B16F1 s.c. Lung LN Distant lymph nodes Colon B B16F1 s.c. Supplementary Figure 1. Deletion of Smad3 prevents B16F1 melanoma invasion and metastasis in a mouse s.c. tumor model. Highly invasive growth
Catechin s anti-angiogenic effects in epithelial ovarian cancer
 Catechin s anti-angiogenic effects in epithelial ovarian cancer Brian Krug Background Epithelial ovarian cancer (EOC) is a common and lethal malignancy of the female reproductive tract (2). Often detected
Catechin s anti-angiogenic effects in epithelial ovarian cancer Brian Krug Background Epithelial ovarian cancer (EOC) is a common and lethal malignancy of the female reproductive tract (2). Often detected
The Met Pathway as a Target in RCC
 The Met Pathway as a Target in RCC Harriet Kluger, M.D. Associate Professor Yale Cancer Center Disclosures pertinent to this presentation - none c-met Pathway (Biocarta) Rationale for c-met targeting in
The Met Pathway as a Target in RCC Harriet Kluger, M.D. Associate Professor Yale Cancer Center Disclosures pertinent to this presentation - none c-met Pathway (Biocarta) Rationale for c-met targeting in
The Therapeutic Landscape in Advanced Renal Cell Carcinoma
 The Therapeutic Landscape in Advanced Renal Cell Carcinoma Cora Sternberg, MD, FACP Chairman, Department of Medical Oncology San Camillo-Forlanini Hospital Rome, Italy What best describes the change in
The Therapeutic Landscape in Advanced Renal Cell Carcinoma Cora Sternberg, MD, FACP Chairman, Department of Medical Oncology San Camillo-Forlanini Hospital Rome, Italy What best describes the change in
Targeted Therapy in Advanced Renal Cell Carcinoma
 Targeted Therapy in Advanced Renal Cell Carcinoma Brian I. Rini, M.D. Department of Solid Tumor Oncology Glickman Urologic and Kidney Institute Cleveland Clinic Taussig Cancer Institute Cleveland, Ohio
Targeted Therapy in Advanced Renal Cell Carcinoma Brian I. Rini, M.D. Department of Solid Tumor Oncology Glickman Urologic and Kidney Institute Cleveland Clinic Taussig Cancer Institute Cleveland, Ohio
Oncolytic Virotherapy: Targeting Cancer Stem Cells
 Oncolytic Virotherapy: Targeting Cancer Stem Cells Cancer Stem Cells (CSCs) or Cancer Initiating Cells (CICs) A consensus of five defining criteria has been established to affirm the existence of CICs:
Oncolytic Virotherapy: Targeting Cancer Stem Cells Cancer Stem Cells (CSCs) or Cancer Initiating Cells (CICs) A consensus of five defining criteria has been established to affirm the existence of CICs:
TAT meeting Paris Can the tumor genome help us to better select patients for antiangiogenic therapy?
 TAT meeting Paris 2011 Can the tumor genome help us to better select patients for antiangiogenic therapy? Emile E Voest Department of Medical Oncology UMC Utrecht Cancer Center Modest succes of bevacizumab
TAT meeting Paris 2011 Can the tumor genome help us to better select patients for antiangiogenic therapy? Emile E Voest Department of Medical Oncology UMC Utrecht Cancer Center Modest succes of bevacizumab
EPIGENETIC RE-EXPRESSION OF HIF-2α SUPPRESSES SOFT TISSUE SARCOMA GROWTH
 EPIGENETIC RE-EXPRESSION OF HIF-2α SUPPRESSES SOFT TISSUE SARCOMA GROWTH Supplementary Figure 1. Supplementary Figure 1. Characterization of KP and KPH2 autochthonous UPS tumors. a) Genotyping of KPH2
EPIGENETIC RE-EXPRESSION OF HIF-2α SUPPRESSES SOFT TISSUE SARCOMA GROWTH Supplementary Figure 1. Supplementary Figure 1. Characterization of KP and KPH2 autochthonous UPS tumors. a) Genotyping of KPH2
Reviewers' comments: Reviewer #1 (Remarks to the Author):
 Reviewers' comments: Reviewer #1 (Remarks to the Author): This is a well written and well executed study describing a novel mechanism of pro-angiogenic signalling which may, potentially, help to explain
Reviewers' comments: Reviewer #1 (Remarks to the Author): This is a well written and well executed study describing a novel mechanism of pro-angiogenic signalling which may, potentially, help to explain
Mechanisms of Gene Regulation and Signal! Transduction in Hypoxia!
 Mechanisms of Gene Regulation and Signal! Transduction in Hypoxia! Lorenz Poellinger! Dept. of Cell and Molecular Biology! Karolinska Institutet, Stockholm, Sweden! Normoxia - O 2 availability is in balance
Mechanisms of Gene Regulation and Signal! Transduction in Hypoxia! Lorenz Poellinger! Dept. of Cell and Molecular Biology! Karolinska Institutet, Stockholm, Sweden! Normoxia - O 2 availability is in balance
Sergio Bracarda MD. Head, Medical Oncology Department of Oncology AUSL-8 Istituto Toscano Tumori (ITT) San Donato Hospital Arezzo, Italy
 Sergio Bracarda MD Head, Medical Oncology Department of Oncology AUSL-8 Istituto Toscano Tumori (ITT) San Donato Hospital Arezzo, Italy Ninth European International Kidney Cancer Symposium Dublin 25-26
Sergio Bracarda MD Head, Medical Oncology Department of Oncology AUSL-8 Istituto Toscano Tumori (ITT) San Donato Hospital Arezzo, Italy Ninth European International Kidney Cancer Symposium Dublin 25-26
Winship Cancer Institute of Emory University Neoadjuvant Systemic Therapy in Metastatic Renal Cell Carcinoma Patients
 Winship Cancer Institute of Emory University Neoadjuvant Systemic Therapy in Metastatic Renal Cell Carcinoma Patients Bradley Carthon, MD, PhD Assistant Professor, Genitourinary Medical Oncology Winship
Winship Cancer Institute of Emory University Neoadjuvant Systemic Therapy in Metastatic Renal Cell Carcinoma Patients Bradley Carthon, MD, PhD Assistant Professor, Genitourinary Medical Oncology Winship
Background. Capdevila J, et al. Ann Oncol. 2018;29(Suppl 8): Abstract 1307O. 1. Dasari A, et al. JAMA Oncol. 2017;3(10):
 Efficacy of Lenvatinib in Patients With Advanced Pancreatic (pannets) and Gastrointestinal (ginets) WHO Grade 1/2 (G1/G2) Neuroendocrine Tumors: Results of the International Phase II TALENT Trial (GETNE
Efficacy of Lenvatinib in Patients With Advanced Pancreatic (pannets) and Gastrointestinal (ginets) WHO Grade 1/2 (G1/G2) Neuroendocrine Tumors: Results of the International Phase II TALENT Trial (GETNE
Emerging Biomarkers of VEGF and mtor Inhibitors in 2015
 Emerging Biomarkers of VEGF and mtor Inhibitors in 2015 Laurence Albiges Institut Gustave Roussy, France Fourteenth International Kidney Cancer Symposium Miami, Florida, USA November 6-7, 2015 www.kidneycancersymposium.com
Emerging Biomarkers of VEGF and mtor Inhibitors in 2015 Laurence Albiges Institut Gustave Roussy, France Fourteenth International Kidney Cancer Symposium Miami, Florida, USA November 6-7, 2015 www.kidneycancersymposium.com
Heterotypy and Angiogenesis
 Heterotypy and Angiogenesis Tumors are perpetual wounds 1. Normally stroma and epithelia converse at a distance. 2. Juxtaposition of stroma and epithelia is indicative of tissue damage. 4. Activate strategies
Heterotypy and Angiogenesis Tumors are perpetual wounds 1. Normally stroma and epithelia converse at a distance. 2. Juxtaposition of stroma and epithelia is indicative of tissue damage. 4. Activate strategies
Hypoxia inducible factor-1α (HIF-1α) and matrix metalloproteinase 9 (MMP-9) expression in invasive breast cancer
 Hypoxia inducible factor-1α (HIF-1α) and matrix metalloproteinase 9 (MMP-9) expression in invasive breast cancer Anastasios D. Papanastasiou 1, Haralabos Papatheodorou 2, Natassa Alexandrou 3, Foteini
Hypoxia inducible factor-1α (HIF-1α) and matrix metalloproteinase 9 (MMP-9) expression in invasive breast cancer Anastasios D. Papanastasiou 1, Haralabos Papatheodorou 2, Natassa Alexandrou 3, Foteini
Theralase Therapeutic Laser Technology Enhances Cancer Destruction
 Theralase Therapeutic Laser Technology Enhances Cancer Destruction Toronto, Ontario November 27, 2017, Theralase Technologies Inc. ( Theralase or the Company ) (TSXV: TLT) (OTCQX: TLTFF), a leading biotech
Theralase Therapeutic Laser Technology Enhances Cancer Destruction Toronto, Ontario November 27, 2017, Theralase Technologies Inc. ( Theralase or the Company ) (TSXV: TLT) (OTCQX: TLTFF), a leading biotech
Interleukin-8 Mediates Resistance to Antiangiogenic Agent Sunitinib in Renal Cell Carcinoma
 Therapeutics, Targets, and Chemical Biology Interleukin-8 Mediates Resistance to Antiangiogenic Agent Sunitinib in Renal Cell Carcinoma Cancer Research Dan Huang 1, Yan Ding 1, Ming Zhou 3, Brian I. Rini
Therapeutics, Targets, and Chemical Biology Interleukin-8 Mediates Resistance to Antiangiogenic Agent Sunitinib in Renal Cell Carcinoma Cancer Research Dan Huang 1, Yan Ding 1, Ming Zhou 3, Brian I. Rini
Quantification of early stage lesions for loss of p53 should be shown in the main figures.
 Reviewer #1 (Remarks to the Author): Expert in prostate cancer The manuscript "Clonal dynamics following p53 loss of heterozygosity in Kras-driven cancers" uses a number of novel genetically engineered
Reviewer #1 (Remarks to the Author): Expert in prostate cancer The manuscript "Clonal dynamics following p53 loss of heterozygosity in Kras-driven cancers" uses a number of novel genetically engineered
Cancer Metronomic Therapy Milan, February 26, 2016
 Cancer Metronomic Therapy Milan, February 26, 2016 Metronomic Chemotherapy: Evolution and Development of the Concept Robert S. Kerbel, PhD Senior Scientist Sunnybrook Research Institute Professor, Dept.
Cancer Metronomic Therapy Milan, February 26, 2016 Metronomic Chemotherapy: Evolution and Development of the Concept Robert S. Kerbel, PhD Senior Scientist Sunnybrook Research Institute Professor, Dept.
Can we prevent metastasis?
 Can we prevent metastasis? A research example to translate from the bench to the bedside Diane Palmieri, Ph.D. Women s Cancers Section Laboratory of Molecular Pharmacology CCR, NCI Some Basic Truths Most
Can we prevent metastasis? A research example to translate from the bench to the bedside Diane Palmieri, Ph.D. Women s Cancers Section Laboratory of Molecular Pharmacology CCR, NCI Some Basic Truths Most
* * * * Supplementary Figure 1. DS Lv CK HSA CK HSA. CK Col-3. CK Col-3. See overleaf for figure legend. Cancer cells
 Supplementary Figure 1 Cancer cells Desmoplastic stroma Hepatocytes Pre-existing sinusoidal blood vessel New blood vessel a Normal liver b Desmoplastic HGP c Pushing HGP d Replacement HGP e f g h i DS
Supplementary Figure 1 Cancer cells Desmoplastic stroma Hepatocytes Pre-existing sinusoidal blood vessel New blood vessel a Normal liver b Desmoplastic HGP c Pushing HGP d Replacement HGP e f g h i DS
Prognostic Factors: Does It Really Matter if New Drugs for Targeted Therapy Will Be Used?
 european urology supplements 8 (2009) 478 482 available at www.sciencedirect.com journal homepage: www.europeanurology.com Prognostic Factors: Does It Really Matter if New Drugs for Targeted Therapy Will
european urology supplements 8 (2009) 478 482 available at www.sciencedirect.com journal homepage: www.europeanurology.com Prognostic Factors: Does It Really Matter if New Drugs for Targeted Therapy Will
Novel RCC Targets from Immuno-Oncology and Antibody-Drug Conjugates
 Novel RCC Targets from Immuno-Oncology and Antibody-Drug Conjugates Christopher Turner, MD Vice President, Clinical Science 04 November 2016 Uveal Melanoma Celldex Pipeline CANDIDATE INDICATION Preclinical
Novel RCC Targets from Immuno-Oncology and Antibody-Drug Conjugates Christopher Turner, MD Vice President, Clinical Science 04 November 2016 Uveal Melanoma Celldex Pipeline CANDIDATE INDICATION Preclinical
A Phase II Study of Atezolizumab With or Without Bevacizumab vs Sunitinib in Untreated Metastatic Renal Cell Carcinoma Patients
 A Phase II Study of With or Without Bevacizumab vs in Untreated Metastatic Renal Cell Carcinoma Patients David McDermott, 1 Michael Atkins, 2 Robert Motzer, 3 Brian Rini, 4 Bernard Escudier, 5 Lawrence
A Phase II Study of With or Without Bevacizumab vs in Untreated Metastatic Renal Cell Carcinoma Patients David McDermott, 1 Michael Atkins, 2 Robert Motzer, 3 Brian Rini, 4 Bernard Escudier, 5 Lawrence
Correlation between expression and significance of δ-catenin, CD31, and VEGF of non-small cell lung cancer
 Correlation between expression and significance of δ-catenin, CD31, and VEGF of non-small cell lung cancer X.L. Liu 1, L.D. Liu 2, S.G. Zhang 1, S.D. Dai 3, W.Y. Li 1 and L. Zhang 1 1 Thoracic Surgery,
Correlation between expression and significance of δ-catenin, CD31, and VEGF of non-small cell lung cancer X.L. Liu 1, L.D. Liu 2, S.G. Zhang 1, S.D. Dai 3, W.Y. Li 1 and L. Zhang 1 1 Thoracic Surgery,
AperTO - Archivio Istituzionale Open Access dell'università di Torino
 AperTO - Archivio Istituzionale Open Access dell'università di Torino From the nucleus to the mitochondria and backthe odyssey of a multitask STAT3 This is the author's manuscript Original Citation: From
AperTO - Archivio Istituzionale Open Access dell'università di Torino From the nucleus to the mitochondria and backthe odyssey of a multitask STAT3 This is the author's manuscript Original Citation: From
SUPPLEMENTARY INFORMATION
 DOI: 1.138/ncb3355 a S1A8 + cells/ total.1.8.6.4.2 b S1A8/?-Actin c % T-cell proliferation 3 25 2 15 1 5 T cells Supplementary Figure 1 Inter-tumoral heterogeneity of MDSC accumulation in mammary tumor
DOI: 1.138/ncb3355 a S1A8 + cells/ total.1.8.6.4.2 b S1A8/?-Actin c % T-cell proliferation 3 25 2 15 1 5 T cells Supplementary Figure 1 Inter-tumoral heterogeneity of MDSC accumulation in mammary tumor
UNIVERSITY OF MEDICINE AND PHARMACY CRAIOVA PhD SCHOOL. PhD THESIS
 UNIVERSITY OF MEDICINE AND PHARMACY CRAIOVA PhD SCHOOL PhD THESIS THE IMPORTANCE OF TUMOR ANGIOGENESIS IN CEREBRAL TUMOR DIAGNOSIS AND THERAPY ABSTRACT PhD COORDINATOR: Prof. univ. dr. DRICU Anica PhD
UNIVERSITY OF MEDICINE AND PHARMACY CRAIOVA PhD SCHOOL PhD THESIS THE IMPORTANCE OF TUMOR ANGIOGENESIS IN CEREBRAL TUMOR DIAGNOSIS AND THERAPY ABSTRACT PhD COORDINATOR: Prof. univ. dr. DRICU Anica PhD
TITLE: Overcoming Resistance to Inhibitors of the Akt Protein Kinase by Modulation of the Pim Kinase Pathway
 AWARD NUMBER: W81XWH-12-1-0560 TITLE: Overcoming Resistance to Inhibitors of the Akt Protein Kinase by Modulation of the Pim Kinase Pathway PRINCIPAL INVESTIGATOR: Andrew S. Kraft, MD CONTRACTING ORGANIZATION:
AWARD NUMBER: W81XWH-12-1-0560 TITLE: Overcoming Resistance to Inhibitors of the Akt Protein Kinase by Modulation of the Pim Kinase Pathway PRINCIPAL INVESTIGATOR: Andrew S. Kraft, MD CONTRACTING ORGANIZATION:
Multiparametric imaging in oncology
 Multiparametric imaging in oncology p1 T p2 p2 T T p3 p1 p3 T Marco Ravanelli Roberto Maroldi The goal of traditional imaging is high spatial and contrast resolution diagnosis, tumor extent treatment planning,
Multiparametric imaging in oncology p1 T p2 p2 T T p3 p1 p3 T Marco Ravanelli Roberto Maroldi The goal of traditional imaging is high spatial and contrast resolution diagnosis, tumor extent treatment planning,
IVC History, Cancer Research
 Riordan Clinic IVC Academy 5 IVC History, Cancer Research O (slides 1 40) Riordan Clinic 2018 High Dose Vitamin C Adjunctive Care for Cancer Patients IVC and Cancer Research Overview History & Research
Riordan Clinic IVC Academy 5 IVC History, Cancer Research O (slides 1 40) Riordan Clinic 2018 High Dose Vitamin C Adjunctive Care for Cancer Patients IVC and Cancer Research Overview History & Research
The PI3K/AKT axis. Dr. Lucio Crinò Medical Oncology Division Azienda Ospedaliera-Perugia. Introduction
 The PI3K/AKT axis Dr. Lucio Crinò Medical Oncology Division Azienda Ospedaliera-Perugia Introduction Phosphoinositide 3-kinase (PI3K) pathway are a family of lipid kinases discovered in 1980s. They have
The PI3K/AKT axis Dr. Lucio Crinò Medical Oncology Division Azienda Ospedaliera-Perugia Introduction Phosphoinositide 3-kinase (PI3K) pathway are a family of lipid kinases discovered in 1980s. They have
High expression of fibroblast activation protein is an adverse prognosticator in gastric cancer.
 Biomedical Research 2017; 28 (18): 7779-7783 ISSN 0970-938X www.biomedres.info High expression of fibroblast activation protein is an adverse prognosticator in gastric cancer. Hu Song 1, Qi-yu Liu 2, Zhi-wei
Biomedical Research 2017; 28 (18): 7779-7783 ISSN 0970-938X www.biomedres.info High expression of fibroblast activation protein is an adverse prognosticator in gastric cancer. Hu Song 1, Qi-yu Liu 2, Zhi-wei
Neoplasia 18 lecture 8. Dr Heyam Awad MD, FRCPath
 Neoplasia 18 lecture 8 Dr Heyam Awad MD, FRCPath ILOS 1. understand the angiogenic switch in tumors and factors that stimulate and inhibit angiogenesis. 2. list the steps important for tumor metastasis
Neoplasia 18 lecture 8 Dr Heyam Awad MD, FRCPath ILOS 1. understand the angiogenic switch in tumors and factors that stimulate and inhibit angiogenesis. 2. list the steps important for tumor metastasis
Identifying Metabolic Phenotype Switches in Cancer Cells Using the Agilent Seahorse XF Analyzer in an Hypoxic Environment
 Identifying Metabolic Phenotype Switches in Cancer Cells Using the Agilent Seahorse XF Analyzer in an Hypoxic Environment Application Note Introduction Decreased oxygen levels, or hypoxia, and hypoxic-mediated
Identifying Metabolic Phenotype Switches in Cancer Cells Using the Agilent Seahorse XF Analyzer in an Hypoxic Environment Application Note Introduction Decreased oxygen levels, or hypoxia, and hypoxic-mediated
Mechanisms of hormone drug resistance
 Mechanisms of hormone drug resistance Ljiljana Stamatović Institute for Oncology and Radiology of Serbia Tenth UMOS Conference, Belgrade, 16-17 th May 2015. Hormone receptor-positive breast cancer (HR+
Mechanisms of hormone drug resistance Ljiljana Stamatović Institute for Oncology and Radiology of Serbia Tenth UMOS Conference, Belgrade, 16-17 th May 2015. Hormone receptor-positive breast cancer (HR+
SSM signature genes are highly expressed in residual scar tissues after preoperative radiotherapy of rectal cancer.
 Supplementary Figure 1 SSM signature genes are highly expressed in residual scar tissues after preoperative radiotherapy of rectal cancer. Scatter plots comparing expression profiles of matched pretreatment
Supplementary Figure 1 SSM signature genes are highly expressed in residual scar tissues after preoperative radiotherapy of rectal cancer. Scatter plots comparing expression profiles of matched pretreatment
PREPARED FOR: U.S. Army Medical Research and Materiel Command Fort Detrick, Maryland
 AD Award Number: W81XWH-04-1-0618 TITLE: Are Breast Tumor Stem Cells Responsible for Metastasis and Angiogenesis PRINCIPAL INVESTIGATOR: Quintin Pan, Ph.D. CONTRACTING ORGANIZATION: University of Michigan
AD Award Number: W81XWH-04-1-0618 TITLE: Are Breast Tumor Stem Cells Responsible for Metastasis and Angiogenesis PRINCIPAL INVESTIGATOR: Quintin Pan, Ph.D. CONTRACTING ORGANIZATION: University of Michigan
SHREE ET AL, SUPPLEMENTAL MATERIALS. (A) Workflow for tumor cell line derivation and orthotopic implantation.
 SHREE ET AL, SUPPLEMENTAL MATERIALS SUPPLEMENTAL FIGURE AND TABLE LEGENDS Supplemental Figure 1. Derivation and characterization of TS1-TGL and TS2-TGL PyMT cell lines and development of an orthotopic
SHREE ET AL, SUPPLEMENTAL MATERIALS SUPPLEMENTAL FIGURE AND TABLE LEGENDS Supplemental Figure 1. Derivation and characterization of TS1-TGL and TS2-TGL PyMT cell lines and development of an orthotopic
Relevant Disclosures. Targeting VHL Tumors with RTK Inhibitors. VHL Gene and Protein. VHL Mutation Replicates the Hypoxic State. HIF Non HIF Proteins
 HIF Non HIF Proteins HIF Targets Targeting Tumors with RTK Inhibitors Relevant Disclosures Consultant: Peloton, Pfizer, Novartis Research Funding: Pfizer, Novartis Eric Jonasch, MD UT MD Anderson Cancer
HIF Non HIF Proteins HIF Targets Targeting Tumors with RTK Inhibitors Relevant Disclosures Consultant: Peloton, Pfizer, Novartis Research Funding: Pfizer, Novartis Eric Jonasch, MD UT MD Anderson Cancer
Kidney Cancer Session
 New Frontiers in Urologic Oncology September 12 th, 2015 Kidney Cancer Session Moderator: Philippe E. Spiess, M.D. Invited Faculty Members: Wade J. Sexton, MD Jeremiah J. Morrissey, PhD Agenda for Session
New Frontiers in Urologic Oncology September 12 th, 2015 Kidney Cancer Session Moderator: Philippe E. Spiess, M.D. Invited Faculty Members: Wade J. Sexton, MD Jeremiah J. Morrissey, PhD Agenda for Session
UPDATE FROM ASCO GU FEBRUARY 2018, SAN FRANCISCO, USA. Prof. David Pfister University Hospital of Cologne Germany RENAL CELL CARCINOMA
 UPDATE FROM ASCO GU FEBRUARY 2018, SAN FRANCISCO, USA Prof. David Pfister University Hospital of Cologne Germany RENAL CELL CARCINOMA DISCLAIMER Please note: The views expressed within this presentation
UPDATE FROM ASCO GU FEBRUARY 2018, SAN FRANCISCO, USA Prof. David Pfister University Hospital of Cologne Germany RENAL CELL CARCINOMA DISCLAIMER Please note: The views expressed within this presentation
BY Mrs. K.SHAILAJA., M. PHARM., LECTURER DEPT OF PHARMACY PRACTICE, SRM COLLEGE OF PHARMACY
 BY Mrs. K.SHAILAJA., M. PHARM., LECTURER DEPT OF PHARMACY PRACTICE, SRM COLLEGE OF PHARMACY Cancer is a group of more than 100 different diseases that are characterized by uncontrolled cellular growth,
BY Mrs. K.SHAILAJA., M. PHARM., LECTURER DEPT OF PHARMACY PRACTICE, SRM COLLEGE OF PHARMACY Cancer is a group of more than 100 different diseases that are characterized by uncontrolled cellular growth,
Future outlooks in NET: The pathologists point of view. Aurel Perren, Institute of Pathology University of Bern
 Future outlooks in NET: The pathologists point of view Aurel Perren, Institute of Pathology University of Bern ESMO Preceptorship on GI neuroendocrine tumors (NETs) Leuven, November 2014 Near Future >
Future outlooks in NET: The pathologists point of view Aurel Perren, Institute of Pathology University of Bern ESMO Preceptorship on GI neuroendocrine tumors (NETs) Leuven, November 2014 Near Future >
Neuroendocrine Tumors: Just the Basics. George Fisher, MD PhD
 Neuroendocrine Tumors: Just the Basics George Fisher, MD PhD Topics that we will not discuss Some types of lung cancer: Small cell neuroendocrine lung cancer Large cell neuroendocrine lung cancer Some
Neuroendocrine Tumors: Just the Basics George Fisher, MD PhD Topics that we will not discuss Some types of lung cancer: Small cell neuroendocrine lung cancer Large cell neuroendocrine lung cancer Some
2015 EUROPEAN CANCER CONGRESS
 2015 EUROPEAN CANCER CONGRESS 25-29 September 2015 Vienna, Austria SUMMARY The European Cancer Congress (ECC 2015) combined the 40th European Society for Medical Oncology (ESMO) congress with the 18th
2015 EUROPEAN CANCER CONGRESS 25-29 September 2015 Vienna, Austria SUMMARY The European Cancer Congress (ECC 2015) combined the 40th European Society for Medical Oncology (ESMO) congress with the 18th
Targeting fibroblast growth factor receptor (FGFR) pathway in renal cell carcinoma
 Targeting fibroblast growth factor receptor (FGFR) pathway in renal cell carcinoma Francesco Massari, Chiara Ciccarese, Matteo Santoni, Antonio Lopez-Beltran, Marina Scarpelli, Rodolfo Montironi & Liang
Targeting fibroblast growth factor receptor (FGFR) pathway in renal cell carcinoma Francesco Massari, Chiara Ciccarese, Matteo Santoni, Antonio Lopez-Beltran, Marina Scarpelli, Rodolfo Montironi & Liang
C. Data & methodology: validity of approach, quality of data, quality of presentation Excellent.
 Reviewers' comments: Reviewer #1 (Immunotherapy expert) (Remarks to the Author): A. Summary of the key results This study elegantly demonstrates the sequential effects of bevacizumab followed by bevacizumab
Reviewers' comments: Reviewer #1 (Immunotherapy expert) (Remarks to the Author): A. Summary of the key results This study elegantly demonstrates the sequential effects of bevacizumab followed by bevacizumab
Biomarkers in Imunotherapy: RNA Signatures as predictive biomarker
 Biomarkers in Imunotherapy: RNA Signatures as predictive biomarker Joan Carles, MD PhD Director GU, CNS and Sarcoma Program Department of Medical Oncology Vall d'hebron University Hospital Outline Introduction
Biomarkers in Imunotherapy: RNA Signatures as predictive biomarker Joan Carles, MD PhD Director GU, CNS and Sarcoma Program Department of Medical Oncology Vall d'hebron University Hospital Outline Introduction
Cancer Cell Research 14 (2017)
 Available at http:// www.cancercellresearch.org ISSN 2161-2609 Efficacy and safety of bevacizumab for patients with advanced non-small cell lung cancer Ping Xu, Hongmei Li*, Xiaoyan Zhang Department of
Available at http:// www.cancercellresearch.org ISSN 2161-2609 Efficacy and safety of bevacizumab for patients with advanced non-small cell lung cancer Ping Xu, Hongmei Li*, Xiaoyan Zhang Department of
VHL Syndrome and Hemangioblastoma Management
 VHL Syndrome and Hemangioblastoma Management Eric Jonasch Professor of Medicine UT MD Anderson Cancer Center Fourteenth International Kidney Cancer Symposium Miami, Florida, USA November 6-7, 2015 www.kidneycancersymposium.com
VHL Syndrome and Hemangioblastoma Management Eric Jonasch Professor of Medicine UT MD Anderson Cancer Center Fourteenth International Kidney Cancer Symposium Miami, Florida, USA November 6-7, 2015 www.kidneycancersymposium.com
Second - Line Debate: Axitinib
 Second - Line Debate: Axitinib Alain Ravaud, MD PhD Bordeaux, France DISCLOSURES Member of Global, European and/or French advisory board in RCC and/or GU tumors for Pfizer, Novartis, GSK, Roche, BMS, Merck.
Second - Line Debate: Axitinib Alain Ravaud, MD PhD Bordeaux, France DISCLOSURES Member of Global, European and/or French advisory board in RCC and/or GU tumors for Pfizer, Novartis, GSK, Roche, BMS, Merck.
Angiogenesis and tumor growth
 Anti-angiogenic agents: where we are? Martin Reck Department of Thoracic Oncology Hospital Grosshansdorf Germany Angiogenesis and tumor growth Journal of experimental Medicine 1972; 133: 275-88 1 Angiogenesis
Anti-angiogenic agents: where we are? Martin Reck Department of Thoracic Oncology Hospital Grosshansdorf Germany Angiogenesis and tumor growth Journal of experimental Medicine 1972; 133: 275-88 1 Angiogenesis
Supplemental Material
 Supplemental Material Supplementary Fig. 1. EETs stimulate primary tumor growth. a) Schematic presentation of genetic and pharmacological tools used to manipulate endogenous EET levels. b) Endothelial
Supplemental Material Supplementary Fig. 1. EETs stimulate primary tumor growth. a) Schematic presentation of genetic and pharmacological tools used to manipulate endogenous EET levels. b) Endothelial
Metastatic Renal Cancer Medical Treatment
 Metastatic Renal Cancer Medical Treatment Bohuslav Melichar, M.D., Ph.D. Professor and Head Department of Oncology Palacký University Medical School and Teaching Hospital Olomouc, Czech Republic Peculiarities
Metastatic Renal Cancer Medical Treatment Bohuslav Melichar, M.D., Ph.D. Professor and Head Department of Oncology Palacký University Medical School and Teaching Hospital Olomouc, Czech Republic Peculiarities
Sequencing of therapies in mrcc. Ari Hakimi MD Assistant Professor Urology Service, Department of Surgery MSKCC
 Sequencing of therapies in mrcc Ari Hakimi MD Assistant Professor Urology Service, Department of Surgery MSKCC Old Paradigm Sequencing approved agents VEGF TKI Sunitinib Pazopanib Axitinib TKI TKI MTORi
Sequencing of therapies in mrcc Ari Hakimi MD Assistant Professor Urology Service, Department of Surgery MSKCC Old Paradigm Sequencing approved agents VEGF TKI Sunitinib Pazopanib Axitinib TKI TKI MTORi
IVC History, Cancer Research
 Riordan Clinic IVC Academy 5 IVC History, Cancer Research O (slides 41-80) Pharmacokinetics of Oral Vitamin C using Liposomal Form* To test whether plasma vitamin C levels, following oral doses in supplemented
Riordan Clinic IVC Academy 5 IVC History, Cancer Research O (slides 41-80) Pharmacokinetics of Oral Vitamin C using Liposomal Form* To test whether plasma vitamin C levels, following oral doses in supplemented
Cancer as a Metabolic Disease
 Cancer as a Metabolic Disease On the Origin, Management and Prevention of Cancer Thomas N. Seyfried @WILEY "" Forword Preface xiii xv 1. Images of Cancer 1 How Cancer is Viewed 2 References 13 2. Confusion
Cancer as a Metabolic Disease On the Origin, Management and Prevention of Cancer Thomas N. Seyfried @WILEY "" Forword Preface xiii xv 1. Images of Cancer 1 How Cancer is Viewed 2 References 13 2. Confusion
Medical Management of Renal Cell Carcinoma
 Medical Management of Renal Cell Carcinoma Lin Mei, MD Hematology-Oncology Fellow Hematology, Oncology and Palliative Care Virginia Commonwealth University Educational Objectives Background of RCC (epidemiology,
Medical Management of Renal Cell Carcinoma Lin Mei, MD Hematology-Oncology Fellow Hematology, Oncology and Palliative Care Virginia Commonwealth University Educational Objectives Background of RCC (epidemiology,
Supplemental Table 1. Primer sequences for transcript analysis
 Supplemental Table 1. Primer sequences for transcript analysis Primer Sequence (5 3 ) Primer Sequence (5 3 ) Mmp2 Forward CCCGTGTGGCCCTC Mmp15 Forward CGGGGCTGGCT Reverse GCTCTCCCGGTTTC Reverse CCTGGTGTGCCTGCTC
Supplemental Table 1. Primer sequences for transcript analysis Primer Sequence (5 3 ) Primer Sequence (5 3 ) Mmp2 Forward CCCGTGTGGCCCTC Mmp15 Forward CGGGGCTGGCT Reverse GCTCTCCCGGTTTC Reverse CCTGGTGTGCCTGCTC
Breast cancer as a systemic disease: a view of metastasis
 Review Click here for more articles from the symposium doi: 10.1111/joim.12084 Breast cancer as a systemic disease: a view of metastasis A. J. Redig 1 & S. S. McAllister 1,2,3 From the 1 Division of Hematology,
Review Click here for more articles from the symposium doi: 10.1111/joim.12084 Breast cancer as a systemic disease: a view of metastasis A. J. Redig 1 & S. S. McAllister 1,2,3 From the 1 Division of Hematology,
PT2385: HIF 2α Antagonist for the Treatment of. Peloton Therapeutics, Inc. 5/4/ th International VHL Medical Symposium April 8, 2016
 5/4/216 : Antagonist for the Treatment of VHL Mutant ccrcc 12th International VHL Medical Symposium Eli Wallace, Ph.D. Vice President of Chemistry Disclosure Information Eli Wallace I have the following
5/4/216 : Antagonist for the Treatment of VHL Mutant ccrcc 12th International VHL Medical Symposium Eli Wallace, Ph.D. Vice President of Chemistry Disclosure Information Eli Wallace I have the following
Nintedanib in Oncology Backgrounder
 For media outside the US, UK and Canada only Nintedanib in Oncology Backgrounder 1. What is nintedanib? 2. How does nintedanib work? 3. Data overview 4. Additional clinical data 5. Nintedanib approval
For media outside the US, UK and Canada only Nintedanib in Oncology Backgrounder 1. What is nintedanib? 2. How does nintedanib work? 3. Data overview 4. Additional clinical data 5. Nintedanib approval
breast and OVARIAN cancer
 breast and OVARIAN cancer DR DAVID FENNELLY CONSULTANT MEDICAL ONCOLOGIST ST VINCENT S UNIVERSITY HOSPITAL DUBLIN HOW RELEVANT IS ONCOLOGY IN MEDICINE TODAY? Cancer is the second leading cause of death
breast and OVARIAN cancer DR DAVID FENNELLY CONSULTANT MEDICAL ONCOLOGIST ST VINCENT S UNIVERSITY HOSPITAL DUBLIN HOW RELEVANT IS ONCOLOGY IN MEDICINE TODAY? Cancer is the second leading cause of death
Molecular and Cell Biology of Cancer. Code: ECTS Credits: 6. Degree Type Year Semester Biomedical Sciences OT 4 0
 2017/2018 Molecular and Cell Biology of Cancer Code: 101897 ECTS Credits: 6 Degree Type Year Semester 2501230 Biomedical Sciences OT 4 0 Contact Name: Carles Arús Caralto Email: Carles.Arus@uab.cat Other
2017/2018 Molecular and Cell Biology of Cancer Code: 101897 ECTS Credits: 6 Degree Type Year Semester 2501230 Biomedical Sciences OT 4 0 Contact Name: Carles Arús Caralto Email: Carles.Arus@uab.cat Other
INTRODUCTION. This material is quoted from below PDF and modified.
 INTRODUCTION While drug response profiling of cancer cells in two-dimensional culture has been a mainstay of predictive biomarker discovery and anti-cancer drug development, there are aspects of tumor
INTRODUCTION While drug response profiling of cancer cells in two-dimensional culture has been a mainstay of predictive biomarker discovery and anti-cancer drug development, there are aspects of tumor
Integrin v 3 targeted therapy for Kaposi s sarcoma with an in vitro evolved antibody 1
 Integrin v 3 targeted therapy for Kaposi s sarcoma with an in vitro evolved antibody 1 CHRISTOPH RADER, 2 MIKHAIL POPKOV, JOHN A. NEVES, AND CARLOS F. BARBAS III 2 Department of Molecular Biology and The
Integrin v 3 targeted therapy for Kaposi s sarcoma with an in vitro evolved antibody 1 CHRISTOPH RADER, 2 MIKHAIL POPKOV, JOHN A. NEVES, AND CARLOS F. BARBAS III 2 Department of Molecular Biology and The
Fifteenth International Kidney Cancer Symposium
 The following presentation should not be regarded as an endorsement of a particular product/drug/technique by the speaker. The presentation topics were assigned to the speakers by the scientific committee
The following presentation should not be regarded as an endorsement of a particular product/drug/technique by the speaker. The presentation topics were assigned to the speakers by the scientific committee
Oberndofer 1907 Illeal Serotonin Secreting Tumor Carcinoid (Karzinoide)
 GEP-NET Adel K. El-Naggar, M.D., Ph.D. The University of Texas MD Anderson Cancer Center, Houston, Texas Oberndofer 1907 Illeal Serotonin Secreting Tumor Carcinoid (Karzinoide) 1 Histogenesis 16 different
GEP-NET Adel K. El-Naggar, M.D., Ph.D. The University of Texas MD Anderson Cancer Center, Houston, Texas Oberndofer 1907 Illeal Serotonin Secreting Tumor Carcinoid (Karzinoide) 1 Histogenesis 16 different
Camillo Porta S.C. di Oncologia Medica Università degli Studi di Pavia & I.R.C.C.S. Fondazione Policlinico San Matteo di Pavia
 Keynote Lecture: Immunotherapy in GU cancer: where are we, and where are we going? Camillo Porta S.C. di Oncologia Medica Università degli Studi di Pavia & I.R.C.C.S. Fondazione Policlinico San Matteo
Keynote Lecture: Immunotherapy in GU cancer: where are we, and where are we going? Camillo Porta S.C. di Oncologia Medica Università degli Studi di Pavia & I.R.C.C.S. Fondazione Policlinico San Matteo
Vascular Endothelial Growth Factor Expression in Oral Epithelial Dysplasia and Oral Squamous Cell Carcinoma
 OMPJ Vascular Endothelial Growth Factor Expression in Oral Epithelial Dysplasia 10.5005/jp-journals-10037-1003 and Oral Squamous Cell Carcinoma ORIGINAL RESEARCH Vascular Endothelial Growth Factor Expression
OMPJ Vascular Endothelial Growth Factor Expression in Oral Epithelial Dysplasia 10.5005/jp-journals-10037-1003 and Oral Squamous Cell Carcinoma ORIGINAL RESEARCH Vascular Endothelial Growth Factor Expression
(a) Significant biological processes (upper panel) and disease biomarkers (lower panel)
 Supplementary Figure 1. Functional enrichment analyses of secretomic proteins. (a) Significant biological processes (upper panel) and disease biomarkers (lower panel) 2 involved by hrab37-mediated secretory
Supplementary Figure 1. Functional enrichment analyses of secretomic proteins. (a) Significant biological processes (upper panel) and disease biomarkers (lower panel) 2 involved by hrab37-mediated secretory
Prognostic Impact of Hyperglycemia in Patients with Locally Advanced Squamous Cell Carcinoma of Cervix Receiving Definite Radiotherapy
 Prognostic Impact of Hyperglycemia in Patients with Locally Advanced Squamous Cell Carcinoma of Cervix Receiving Definite Radiotherapy 2016.04.08 KCCH 김문홍 DM and prediabetes in cancer Negative impact on
Prognostic Impact of Hyperglycemia in Patients with Locally Advanced Squamous Cell Carcinoma of Cervix Receiving Definite Radiotherapy 2016.04.08 KCCH 김문홍 DM and prediabetes in cancer Negative impact on
Critical Clinical Updates
 Critical Clinical Updates ASTRO Spring Refresher Course JW Marriott Hotel Ramesh Rengan MD PhD Department of Radiation Oncology Friday March 22, 2013 Learning Objectives At the conclusion of this activity,
Critical Clinical Updates ASTRO Spring Refresher Course JW Marriott Hotel Ramesh Rengan MD PhD Department of Radiation Oncology Friday March 22, 2013 Learning Objectives At the conclusion of this activity,
REPROGRAMING IMMUNITY IN RENAL CELL CARCINOMA
 REPROGRAMING IMMUNITY IN RENAL CELL CARCINOMA RETHINKING TYROSINE KINASE INHIBITORS Dr. L.M. Antón Aparicio. Complejo Universitario de La Coruña INTRODUCTION Angiogenesis, which is regulated by a fine
REPROGRAMING IMMUNITY IN RENAL CELL CARCINOMA RETHINKING TYROSINE KINASE INHIBITORS Dr. L.M. Antón Aparicio. Complejo Universitario de La Coruña INTRODUCTION Angiogenesis, which is regulated by a fine
Sunitinib Treatment for Metastatic Renal Cell Carcinoma in Patients with Von Hippel-Lindau Disease
 pissn 1598-2998, eissn 2005-9256 Cancer Res Treat. 2013;45(4):349-353 Case Report http://dx.doi.org/10.4143/crt.2013.45.4.349 Open Access Sunitinib Treatment for Metastatic Renal Cell Carcinoma in Patients
pissn 1598-2998, eissn 2005-9256 Cancer Res Treat. 2013;45(4):349-353 Case Report http://dx.doi.org/10.4143/crt.2013.45.4.349 Open Access Sunitinib Treatment for Metastatic Renal Cell Carcinoma in Patients
Novel Molecular Molecular Therapies In Hepatocarcinoma Prof Eric
 Novel Molecular Therapies In Hepatocarcinoma Prof. Eric Raymond Department of Médical Oncology Hôpital Beaujon, Clichy Université Paris 7 Denis Diderot INSERM-U728 eric.raymond@bjn.aphp.fr HCC is a highly
Novel Molecular Therapies In Hepatocarcinoma Prof. Eric Raymond Department of Médical Oncology Hôpital Beaujon, Clichy Université Paris 7 Denis Diderot INSERM-U728 eric.raymond@bjn.aphp.fr HCC is a highly
Molecular and Cell Biology of Cancer. Code: ECTS Credits: 6. Degree Type Year Semester Biomedical Sciences OT 4 0
 2018/2019 Molecular and Cell Biology of Cancer Code: 101897 ECTS Credits: 6 Degree Type Year Semester 2501230 Biomedical Sciences OT 4 0 Contact Name: Carles Arús Caralto Email: Carles.Arus@uab.cat Other
2018/2019 Molecular and Cell Biology of Cancer Code: 101897 ECTS Credits: 6 Degree Type Year Semester 2501230 Biomedical Sciences OT 4 0 Contact Name: Carles Arús Caralto Email: Carles.Arus@uab.cat Other
Molecular Biology of Cancer. Code: ECTS Credits: 6. Degree Type Year Semester
 2017/2018 Molecular Biology of Cancer Code: 100863 ECTS Credits: 6 Degree Type Year Semester 2500252 Biochemistry OT 4 0 Contact Name: Carles Arús Caralto Email: Carles.Arus@uab.cat Other comments on languages
2017/2018 Molecular Biology of Cancer Code: 100863 ECTS Credits: 6 Degree Type Year Semester 2500252 Biochemistry OT 4 0 Contact Name: Carles Arús Caralto Email: Carles.Arus@uab.cat Other comments on languages
Complete Remission is a Reachable Goal in mrcc L. Albiges Institut Gustave Roussy
 Complete Remission is a Reachable Goal in mrcc L. Albiges Institut Gustave Roussy Is complete remission an achievable goal in mrcc? Lessons from observation Lessons from immunotherapy Current status in
Complete Remission is a Reachable Goal in mrcc L. Albiges Institut Gustave Roussy Is complete remission an achievable goal in mrcc? Lessons from observation Lessons from immunotherapy Current status in
A263 A352 A204. Pan CK. pstat STAT3 pstat3 STAT3 pstat3. Columns Columns 1-6 Positive control. Omentum. Rectosigmoid A195.
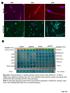 pstat3 75 Pan CK A A263 A352 A24 B Columns 1-6 Positive control A195 A22 A24 A183 Rectal Nodule STAT3 pstat3 STAT3 pstat3 Columns 7-12 Omentum Rectosigmoid Left Ovary Right Ovary Omentum Uterus Uterus
pstat3 75 Pan CK A A263 A352 A24 B Columns 1-6 Positive control A195 A22 A24 A183 Rectal Nodule STAT3 pstat3 STAT3 pstat3 Columns 7-12 Omentum Rectosigmoid Left Ovary Right Ovary Omentum Uterus Uterus
