(2) Phase-lll / Phase-ll (2/3) ASP7487 (OSI-906) linsitinib IGF-1R/IR tyrosine kinase inhibitor Adrenocortical carcinoma Ovarian cancer, Non-small cel
|
|
|
- Bryce Johnson
- 5 years ago
- Views:
Transcription
1 R&D Pipeline (February 2012) 1. Global Development (1) Filed YM178 mirabegron Beta 3 receptor agonist Overactive bladder associated with symptoms of urgency, urinary frequency, and urge urinary incontinece US Filed (Aug. 2011) Europe Filed (Aug. 2011) (2) Phase-lll / Phase-ll (1/3) Stage in the most advanced territory EC905 solifenacin/ tamsulosin Fixed dose combination of solifenacin and tamsulosin Lower urinary tract symptoms associated with benign prostatic hyperplasia, with storage symptoms I MDV3100 Androgen antagonist Prostate cancer I I II Asia Phase-III Medivation ASP1941 ipragliflozin SGLT2 inhibitor Type 2 diabetes II (co-development with Kotobuki) isavuconazole Azole antifungal Invasive aspergillosis Candidemia / Invasive candidiasis US/I US/I Basilea erlotinib (Tarceva) HER1/EGFR tyrosine kinase inhibitor Non-small cell lung cancer (First line for patients with EGFR mutation, adjuvant), Hepatocellular carcinoma I (OSI) 22
2 (2) Phase-lll / Phase-ll (2/3) ASP7487 (OSI-906) linsitinib IGF-1R/IR tyrosine kinase inhibitor Adrenocortical carcinoma Ovarian cancer, Non-small cell lung cancer I Stage in the most advanced territory (OSI) ASP4130 tivozanib Triple VEGF receptors inhibitor Renal cell carcinoma Breast cancer, Colorectal cancer US/I US/ AVEO ASP0113 (VCL-CB01) DNA vaccine for cytomegalovirus Cytomegalovirus reactivation in hematopoietic stem cell transplant recipients Cytomegalovirus infection or reactivation in solid organ transplant recipients US/I US/ Vical YM905 solifenacin Muscarine M 3 receptor antagonist Neurogenic detrusor overactivity and idiopathic overactive bladder in pediatric patients US/I YM155 Survivin suppressant Breast cancer, Non-Hodgkin's lymphoma AC220 quizartinib FLT3 kinase inhibitor Acute myeloid leukemia Ambit ASP1517 (FG-4592) HIF stabilizer Renal anemia FibroGen YM311 (FG-2216) HIF stabilizer Renal anemia FibroGen AGS-1C4D4 Antibody (Prostate stem cell antigen) Pancreatic cancer US/ (Agensys) 23
3 (2) Phase-lll / Phase-ll (3/3) US/ Rheumatoid arthritis I ASP015K JAK inhibitor Prevention of organ transplant rejection Stage in the most advanced territory ASKP1240 Anti-CD40 antagonist Prevention of organ transplant rejection Kyowa Hakko Kirin OSI-027 mtor kinase inhibitor Renal cell cancer PSN821 GPR119 agonist Type 2 diabetes, Obesity (OSI) (OSI) ASP8597 diannexin Inhibition of monocyte and platelet binding to phosphatidylserine Prevention of delayed graft function in kidney transplantation Alavita EB178 solifenacin/ mirabegron Concomitant use of solifenacin and mirabegron Urinary frequency, urinary incontinence or urgency associated with overactive bladder ASP3652 Inhibition of afferent nerve activity Chronic prostatitis / Chronic pelvic pain syndrome Bladder pain syndrome / Interstitial cystitis 24
4 2. Local Development: Japan (1) Approved Product Name (Approval Date) Classification Therapeutic Target Area YM177 celecoxib Celecox (Dec. 2011) Cyclooxygenase-II inhibitor Anti-inflammatory and analgesic effects in post-operation, posttrauma, and post-tooth extraction Japan Pfizer ASP8825 (XP13512) gabapentin enacarbil Regnite (Jan. 2012) Prodrug of gabapentin Moderate-to-severe primary restless legs syndrome Japan XenoPort (2) Filed The most advanced stage YM443 acotiamide ASP3550 degarelix Acetylcholine esterase inhibitor GnRH receptor antagonist Functional dyspepsia Prostate cancer (One-month formulation) Prostate cancer (Three-month formulation) Japan Filed (Sep. 2010) Japan Filed (Oct. 2010) I Zeria Ferring New formulation ASP1585 (AMG223) bixalomer Amine-functional phosphate-binding polymer Hyperphosphatemia in patients with chronic kidney disease on dialysis Japan Filed (Mar. 2011) Ilypsa/Amgen amoxicillin Penicillin antibiotic Pediatric infectious diseases (Changes in the maximum dosages) Japan Filed (Aug. 2011) Additional dosage and administration 25
5 (3) Phase-lll / Phase-ll YM533 beraprost sodium Prostacyclin receptor stimulator Chronic renal failure (primary, nephrosclerosis) Japan/Asia Phase-III Stage in the most advanced territory Toray New formulation FK949E quetiapine Serotonin/dopamine antagonist Depressive episode in bipolar disorders Major depressive disorder II I AstraZeneca New formulation YM060 ramosetron 5-HT3 receptor antagonist Irritable bowel syndrome Female patients Irritable bowel syndrome (ly-disintegrating tablet) I Japan Bio-equivalent study New formulation ASP7373 Influenza vaccine Prophylaxis of H5N1 influenza I UMN Pharma ASP7374 Influenza vaccine Prophylaxis of seasonal influenza I UMN Pharma 3. Local Development: Europe (1) Approved Product Name (Approval Date) Classification Therapeutic Target Area fidaxomicin DIFICLIR (Dec. 2011) Macrocyclic antibiotic Treatment of clostridium difficile infection Europe Optimer 26
6 4. Phase-l Therapeutic Target AGS-16M8F Cancer (ADC technology) (Agensys) ASG-5ME Cancer (ADC technology) (Agensys) (co-development with Seattle Genetics) ASP7035 Nocturia ASP0777 Alzheimer s disease [Dementia] ASP3291 Ulcerative colitis ASP5034 Type 2 diabetes ASP1707 Prostate cancer, Endometriosis ASP0306 Lower urinary tract symptoms associated with benign prostatic hyperplasia ASP4058 Multiple sclerosis ASP0456 linaclotide ASP4901 (AKP-002) Irritable bowel syndrome Ironwood Lower urinary tract symptoms associated with benign prostatic hyperplasia ASKA ASP8477 Neuropathic pain ASP2408 Rheumatoid arthritis (Perseid) ASP3026 Cancer ASP9521 Prostate cancer AGS-22M6E Cancer (ADC technology) (Agensys) (co-development with Seattle Genetics) ASP7147 Irritable bowel syndrome ASP9226 Neuropathic pain ASP9603 Prostate cancer ASP7991 Secondary hyperparathyroidism ASP2409 Prevention of organ transplant rejection (Perseid) 27
7 5. Project Discontinued Area / Phase Therapeutic Target Reason telavancin Europe Approved US Filed * Treatment of adults with nosocomial pneumonia (hospital-acquired), including ventilator associated pneumonia, known or suspected to be caused by MRSA when other alternatives are not suitable Nosocomial pneumonia Development was discontinued because we have terminated the global License, Development and Commercialization Agreement for telavancin. MRSA infections ASP4178 Phase-I Type 2 diabetes We have decided to discontinue the in-house development and to seek a partner, considering comprehensively our focus area and pipeline prioritization. *Received a Complete Response letter from the FDA in November 2009, and the second Complete Response letter in December
8 <Changes from the Previous Announcement on November 1, 2011> Approved -YM177: Approved in Japan for new indication. Product Name (Approval Date) Classification Therapeutic Target Area YM177 celecoxib Celecox (Dec. 2011) Cyclooxygenase-II inhibitor Anti-inflammatory and analgesic effects in postoperation, post-trauma, and post-tooth extraction Japan Pfizer -ASP8825: Approved in Japan. Product Name (Approval Date) Classification Therapeutic Target Area ASP8825 (XP13512) gabapentin enacarbil Regnite (Jan. 2012) Prodrug of gabapentin Moderate-to-severe primary restless legs syndrome Japan XenoPort -Fidaxomicin: Approved in Europe. Product Name (Approval Date) Classification Therapeutic Target Area fidaxomicin DIFICLIR (Dec. 2011) Macrocyclic antibiotic Treatment of clostridium difficile infection Europe Optimer 29
9 Phase-III / Phase-II -EC905: Changed the description of classification and therapeutic target. (Underlined) EC905 solifenacin/ tamsulosin Fixed dose combination of solifenacin and tamsulosin Lower urinary tract symptoms associated with benign prostatic hyperplasia, with storage symptoms I -YM905: Entered into phase-iii. (Underlined) YM905 solifenacin Muscarine M 3 receptor antagonist Neurogenic detrusor overactivity and idiopathic overactive bladder in pediatric patients US/Europe Phase-III -ASP015K: Entered into phase-ii for rheumatoid arthritis in US/Europe and Japan, and changed the classification from immunosuppressant to JAK inhibitor. (Underlined) US/Europe Rheumatoid arthritis Phase-II ASP015K JAK inhibitor I Prevention of organ transplant rejection 30
10 -ASP3652: Entered into phase-ii for bladder pain syndrome / interstitial cystitis in Europe. (Underlined) ASP3652 Inhibition of afferent nerve activity Chronic prostatitis / Chronic pelvic pain syndrome Bladder pain syndrome / Interstitial cystitis -ASP7487 (OSI-906): Deleted hepatocellular carcinoma from the therapeutic target because we discontinued the development in it, which was in phase-ii. (Changed underlined part) ASP7487 (OSI-906) linsitinib IGF-1R/IR tyrosine kinase inhibitor Adrenocortical carcinoma Ovarian cancer, Non-small cell lung cancer I (OSI) Phase-I (Newly Developed) -Added ASP9603 [Prostate cancer] to the Phase-I list. -Added ASP7991 [Secondary hyperparathyroidism] to the Phase-I list. -Added ASP2409 [Prevention of organ transplant rejection] to the Phase-I list. Project Discontinued -Deleted telavancin from the list because we terminated the license agreement with Theravance and discontinued the development. -Deleted ASP4178 due to the development discontinuation. 31
(3) Phase-lll / Phase-ll (1/3) Stage in the most advanced territory EC905 solifenacin/ tamsulosin Concomitant use of solifenacin and tamsulosin Lower
 R&D Pipeline (November 2011) 1. Global Development (1) Approved telavancin Product Name (Approval Date) VIBATIV (Sep. 2011) Classification Lipoglycopeptide antibiotic Therapeutic Target Treatment of adults
R&D Pipeline (November 2011) 1. Global Development (1) Approved telavancin Product Name (Approval Date) VIBATIV (Sep. 2011) Classification Lipoglycopeptide antibiotic Therapeutic Target Treatment of adults
R&D Pipeline (May 2010)
 R&D Pipeline (May 2010) 1 Global development (1) Filed Stage in the Most Advanced Territory YM617 tamsulosin Alpha-1 receptor antagonist Lower urinary tract syndrome in male patients (June 2007) New indication
R&D Pipeline (May 2010) 1 Global development (1) Filed Stage in the Most Advanced Territory YM617 tamsulosin Alpha-1 receptor antagonist Lower urinary tract syndrome in male patients (June 2007) New indication
R&D Pipeline (May 2014) 1. Global Development (1) Approved Product Name (Approval Date) Classification Target Disease Area * MDV3100 enzalutamide XTAN
 R&D Pipeline (May 2014) 1. Global Development (1) Approved Product Name (Approval Date) Classification Target Disease Area * MDV3100 enzalutamide XTANDI (Mar. 2014) Androgen receptor inhibitor
R&D Pipeline (May 2014) 1. Global Development (1) Approved Product Name (Approval Date) Classification Target Disease Area * MDV3100 enzalutamide XTANDI (Mar. 2014) Androgen receptor inhibitor
R&D Pipeline (As of July 2016) 1. Global Development Underlined items indicate for changes from the previous announcement on May 11, (1) Regulat
 R&D Pipeline (As of July 2016) 1. Global Development Underlined items indicate for changes from the previous announcement on May 11, 2016. (1) Regulatory decision *Compounds with "" in this column include
R&D Pipeline (As of July 2016) 1. Global Development Underlined items indicate for changes from the previous announcement on May 11, 2016. (1) Regulatory decision *Compounds with "" in this column include
R&D Pipeline (As of April 2017)
 R&D Pipeline (As of April 2017) Underlined items indicate changes from the previous announcement on January 31, 2017. Oncology (1/2) MDV3100 enzalutamide Androgen receptor inhibitor Metastatic castration-resistant
R&D Pipeline (As of April 2017) Underlined items indicate changes from the previous announcement on January 31, 2017. Oncology (1/2) MDV3100 enzalutamide Androgen receptor inhibitor Metastatic castration-resistant
*Compounds with "In-house" in this column include ones discovered by collaborative research.
 AGS67E Lymphoid malignancies P-I Injection In-house (ADC technology in-licensed from Seattle Genetics) AGS62P1 Acute myeloid leukemia P-I Injection In-house (ADC technology, EuCODE license from Ambrx)
AGS67E Lymphoid malignancies P-I Injection In-house (ADC technology in-licensed from Seattle Genetics) AGS62P1 Acute myeloid leukemia P-I Injection In-house (ADC technology, EuCODE license from Ambrx)
R&D Pipeline (As of Apr 2018) Underlined items indicate changes from the previous announcement on Jan 31, 2018.
 R&D Pipeline (As of Apr 2018) Underlined items indicate changes from the previous announcement on Jan 31, 2018. Oncology (1/2) MDV3100 enzalutamide Androgen receptor inhibitor Castration-resistant prostate
R&D Pipeline (As of Apr 2018) Underlined items indicate changes from the previous announcement on Jan 31, 2018. Oncology (1/2) MDV3100 enzalutamide Androgen receptor inhibitor Castration-resistant prostate
R&D Pipeline (As of Oct 2018) Underlined items indicate changes from the previous announcement on July 27, 2018.
 R&D Pipeline (As of Oct 2018) Underlined items indicate changes from the previous announcement on July 27, 2018. Oncology (1/2) enzalutamide MDV3100 (XTANDI ) Androgen receptor inhibitor Non-metastatic
R&D Pipeline (As of Oct 2018) Underlined items indicate changes from the previous announcement on July 27, 2018. Oncology (1/2) enzalutamide MDV3100 (XTANDI ) Androgen receptor inhibitor Non-metastatic
R&D Pipeline (As of Jul 2018) Underlined items indicate changes from the previous announcement on Apr 26, 2018.
 R&D Pipeline (As of Jul 2018) Underlined items indicate changes from the previous announcement on Apr 26, 2018. Oncology (1/2) MDV3100 enzalutamide (XTANDI ) Androgen receptor inhibitor Non-metastatic
R&D Pipeline (As of Jul 2018) Underlined items indicate changes from the previous announcement on Apr 26, 2018. Oncology (1/2) MDV3100 enzalutamide (XTANDI ) Androgen receptor inhibitor Non-metastatic
R&D Pipeline (May 2006)
 R&D Pipeline (May 2006) 1Japan (1)Approved Product Name (Code No) Vesicare (YM905) Funguard (FK463) (Approval Date ) solifenacin (April 2006) micafungin (April 2006) Urinary frequency, urinary incontinence
R&D Pipeline (May 2006) 1Japan (1)Approved Product Name (Code No) Vesicare (YM905) Funguard (FK463) (Approval Date ) solifenacin (April 2006) micafungin (April 2006) Urinary frequency, urinary incontinence
R&D Pipeline (November 2006)
 R&D Pipeline (November 2006) 1Japan (1)Filed Code No Generic Name (Filing Date ) Therapeutic Target Classification YM177 celecoxib (Dec 2002) Rheumatoid arthritis, osteoarthritis, low back pain, etc Cyclooxygenase-II
R&D Pipeline (November 2006) 1Japan (1)Filed Code No Generic Name (Filing Date ) Therapeutic Target Classification YM177 celecoxib (Dec 2002) Rheumatoid arthritis, osteoarthritis, low back pain, etc Cyclooxygenase-II
R&D Pipeline (November 2005)
 R&D Pipeline (November 2005) 1Japan (1)Filed Code No YM905 YM177 YM152 FK463 Generic Name (Filing Date ) solifenacin (Aug 2004) celecoxib (Dec 2002) finasteride (Dec1994) micafungin (July 2004) (Oct 2005)
R&D Pipeline (November 2005) 1Japan (1)Filed Code No YM905 YM177 YM152 FK463 Generic Name (Filing Date ) solifenacin (Aug 2004) celecoxib (Dec 2002) finasteride (Dec1994) micafungin (July 2004) (Oct 2005)
Changes from Third Quarter Flash Report for the Fiscal Year ending March 2015 announced on February 3, 2015 *1: Marketing authorization of Onoact 150
 Annual Flash Report (unaudited) Fiscal Year ended March 31, 2015 Supplemental Information Status of Development Pipeline as of May 12, 2015 I. Main Pipelines Other than ONO-4538 ⅰ. Developments Status
Annual Flash Report (unaudited) Fiscal Year ended March 31, 2015 Supplemental Information Status of Development Pipeline as of May 12, 2015 I. Main Pipelines Other than ONO-4538 ⅰ. Developments Status
Overview of Product Pipeline
 Overview of Product Pipeline November 4, 2004 Yamanouchi Pharmaceutical Co., Ltd. Shinji Usuda,, Ph. D. Corporate Vice President Director of Drug Development Division Cautionary statement regarding forward-looking
Overview of Product Pipeline November 4, 2004 Yamanouchi Pharmaceutical Co., Ltd. Shinji Usuda,, Ph. D. Corporate Vice President Director of Drug Development Division Cautionary statement regarding forward-looking
Maximizing the Product Value
 Business Review Review of Operations by Therapeutic Area Astellas is working to steadily grow and maximize the value of products developed through its investments over the years, including its growth drivers
Business Review Review of Operations by Therapeutic Area Astellas is working to steadily grow and maximize the value of products developed through its investments over the years, including its growth drivers
Summary of Strategic Competitive Analysis and Publication Planning
 Summary of Strategic Competitive Analysis and Publication Planning Carole Alison Chrvala, PhD 919.545.2149 (Work) 919.951.5230 (Mobile) cchrvala@centurylink.net www.healthmattersmedwriting.com 1 Briefing
Summary of Strategic Competitive Analysis and Publication Planning Carole Alison Chrvala, PhD 919.545.2149 (Work) 919.951.5230 (Mobile) cchrvala@centurylink.net www.healthmattersmedwriting.com 1 Briefing
Annual Flash Report (unaudited) Fiscal Year ended March 31, 2017
 Annual Flash Report (unaudited) Fiscal Year ended March 31, 2017 Supplemental nation Status of Development Pipeline as of May 8, 2017. Main Status of Development Pipelines (Oncology) 1. Development Status
Annual Flash Report (unaudited) Fiscal Year ended March 31, 2017 Supplemental nation Status of Development Pipeline as of May 8, 2017. Main Status of Development Pipelines (Oncology) 1. Development Status
Astellas Growth Strategy
 Press Conference Astellas Growth Strategy November 29,2010 Masafumi Nogimori, President&CEO Astellas Pharma Inc..1 Cautionary Statement Regarding Forward-Looking Information This material includes forward-looking
Press Conference Astellas Growth Strategy November 29,2010 Masafumi Nogimori, President&CEO Astellas Pharma Inc..1 Cautionary Statement Regarding Forward-Looking Information This material includes forward-looking
Financial Results for FY2012 Year ended March 31, 2013
 0 Financial Results for FY2012 Year ended March 31, 2013 May 14, 2013 Yoshihiko Hatanaka President and CEO Astellas Pharma Inc. 1 Cautionary Statement Regarding Forward-Looking Information This material
0 Financial Results for FY2012 Year ended March 31, 2013 May 14, 2013 Yoshihiko Hatanaka President and CEO Astellas Pharma Inc. 1 Cautionary Statement Regarding Forward-Looking Information This material
Overactive Bladder Syndrome
 Overactive Bladder Syndrome behavioural modifications to pharmacological and surgical treatments Dr Jos Jayarajan Urologist Austin Health, Eastern Health Warringal Private, Northpark Private, Epworth Overactive
Overactive Bladder Syndrome behavioural modifications to pharmacological and surgical treatments Dr Jos Jayarajan Urologist Austin Health, Eastern Health Warringal Private, Northpark Private, Epworth Overactive
Development Pipeline Progress Status. February 1, 2019
 Development Pipeline Progress Status February 1, 2019 /9 Development status of OPDIVO (nivolumab) (1) Target disease JAPAN US/EU KR/TW Melanoma (1 st ) Approved Approved Approved Melanoma (Adjuvant Therapy)
Development Pipeline Progress Status February 1, 2019 /9 Development status of OPDIVO (nivolumab) (1) Target disease JAPAN US/EU KR/TW Melanoma (1 st ) Approved Approved Approved Melanoma (Adjuvant Therapy)
URGE MOTOR INCONTINENCE
 URGE MOTOR INCONTINENCE URGE INCONTINENCE COMMONEST TYPE IN ELDERLY WOMEN Causes: 1 - Defects in CNS regulation Stroke Parkinson s disease Dementia (Alzheimer s and other types) Normopressure hydrocephalus
URGE MOTOR INCONTINENCE URGE INCONTINENCE COMMONEST TYPE IN ELDERLY WOMEN Causes: 1 - Defects in CNS regulation Stroke Parkinson s disease Dementia (Alzheimer s and other types) Normopressure hydrocephalus
Major R&D Pipeline As of January 2016
 Major R&D Pipeline As of January 2016 Therapeutic area Cardiovascular- Metabolics Phase 1 Phase 2 Phase 3 Application DS-1040 (Acute ischemic stroke / TAFIa DS-2330 (Hyperphosphatemia) DS-9231/TS23 (Thrombosis
Major R&D Pipeline As of January 2016 Therapeutic area Cardiovascular- Metabolics Phase 1 Phase 2 Phase 3 Application DS-1040 (Acute ischemic stroke / TAFIa DS-2330 (Hyperphosphatemia) DS-9231/TS23 (Thrombosis
Financial Results for the 2Q/FY 2009 Ending March 31, November 6, 2009 Masafumi Nogimori President and CEO Astellas Pharma Inc.
 Financial Results for the 2Q/FY 2009 Ending March 31, 2010 November 6, 2009 Masafumi Nogimori President and CEO Astellas Pharma Inc. Cautionary Statement Regarding Forward-Looking Information This material
Financial Results for the 2Q/FY 2009 Ending March 31, 2010 November 6, 2009 Masafumi Nogimori President and CEO Astellas Pharma Inc. Cautionary Statement Regarding Forward-Looking Information This material
Development status of ONO-4538(nivolumab)1
 Development status of ONO-4538(nivolumab)1 Development Code Target Disease JAPAN US/EU KOR/TAI ONO-4538 Melanoma(2 nd ~) Launched Launched (US) Filing (EU) Approved(KOR) Filing(TAI) ONO-4538 Melanoma(1
Development status of ONO-4538(nivolumab)1 Development Code Target Disease JAPAN US/EU KOR/TAI ONO-4538 Melanoma(2 nd ~) Launched Launched (US) Filing (EU) Approved(KOR) Filing(TAI) ONO-4538 Melanoma(1
Merck Pipeline. August 1, 2018
 Merck Pipeline August 1, 2018 Lead-in Language The chart below reflects the Company s research pipeline as of August 1, 2018. Candidates shown in Phase 3 include specific products and the date such candidate
Merck Pipeline August 1, 2018 Lead-in Language The chart below reflects the Company s research pipeline as of August 1, 2018. Candidates shown in Phase 3 include specific products and the date such candidate
Financial Results for FY2014 Ended March 31, 2015
 0 Financial Results for FY2014 Ended March 31, 2015 May 11, 2015 Yoshihiko Hatanaka President and CEO Astellas Pharma Inc. 1 Cautionary Statement Regarding Forward-Looking Information This material includes
0 Financial Results for FY2014 Ended March 31, 2015 May 11, 2015 Yoshihiko Hatanaka President and CEO Astellas Pharma Inc. 1 Cautionary Statement Regarding Forward-Looking Information This material includes
Development status of OPDIVO (nivolumab) 1
 November 2, 2018 Development status of OPDIVO (nivolumab) 1 Target disease JAPAN US/EU KR/TW Melanoma (1 st ) Approved Approved Approved Melanoma (Adjuvant Therapy) Approved Approved(EU) Non-small cell
November 2, 2018 Development status of OPDIVO (nivolumab) 1 Target disease JAPAN US/EU KR/TW Melanoma (1 st ) Approved Approved Approved Melanoma (Adjuvant Therapy) Approved Approved(EU) Non-small cell
Financial Results for FY2013 Ended March 31, May 12, 2014 Yoshihiko Hatanaka President and CEO Astellas Pharma Inc.
 0 Financial Results for Ended March 31, 2014 May 12, 2014 Yoshihiko Hatanaka President and CEO Astellas Pharma Inc. 1 Cautionary Statement Regarding Forward-Looking Information This material includes forward-looking
0 Financial Results for Ended March 31, 2014 May 12, 2014 Yoshihiko Hatanaka President and CEO Astellas Pharma Inc. 1 Cautionary Statement Regarding Forward-Looking Information This material includes forward-looking
Development status of ONO-4538(nivolumab)1
 Development status of ONO-4538(nivolumab)1 Development Code Target Disease JAPAN US/EU KOR/TAI ONO-4538 Melanoma(2 nd ~) Launched Launched (US) Filing (EU) Approved(KOR) Filing(TAI) ONO-4538 Melanoma(1
Development status of ONO-4538(nivolumab)1 Development Code Target Disease JAPAN US/EU KOR/TAI ONO-4538 Melanoma(2 nd ~) Launched Launched (US) Filing (EU) Approved(KOR) Filing(TAI) ONO-4538 Melanoma(1
Financial Results for 1Q/FY2015
 Financial Results for 1Q/FY2015 Ending March 31, 2016 July 31, 2015 Yasumasa Masuda Senior Corporate Executive, Chief Financial Officer Astellas Pharma Inc. 0 Cautionary Statement Regarding Forward-Looking
Financial Results for 1Q/FY2015 Ending March 31, 2016 July 31, 2015 Yasumasa Masuda Senior Corporate Executive, Chief Financial Officer Astellas Pharma Inc. 0 Cautionary Statement Regarding Forward-Looking
Pfizer Pipeline. August 9, 2012
 Pfizer Pipeline August 9, 2012 Disclaimer As some programs are still confidential, some candidates may not be identified in this list. In these materials, Pfizer discloses Mechanism of Action (MOA) information
Pfizer Pipeline August 9, 2012 Disclaimer As some programs are still confidential, some candidates may not be identified in this list. In these materials, Pfizer discloses Mechanism of Action (MOA) information
Rayos Prior Authorization Program Summary
 Rayos Prior Authorization Program Summary FDA APPROVED INDICATIONS AND DOSAGE FDA-Approved Indications: 1 Agent Indication Dosage Rayos (prednisone delayedrelease tablet) as an anti-inflammatory or immunosuppressive
Rayos Prior Authorization Program Summary FDA APPROVED INDICATIONS AND DOSAGE FDA-Approved Indications: 1 Agent Indication Dosage Rayos (prednisone delayedrelease tablet) as an anti-inflammatory or immunosuppressive
Pfizer Pipeline. November 8, 2012
 Pfizer Pipeline November 8, 2012 Disclaimer As some programs are still confidential, some candidates may not be identified in this list. In these materials, Pfizer discloses Mechanism of Action (MOA) information
Pfizer Pipeline November 8, 2012 Disclaimer As some programs are still confidential, some candidates may not be identified in this list. In these materials, Pfizer discloses Mechanism of Action (MOA) information
ICD-9-CM CODING FUNDAMENTALS CODING EXERCISES
 Steps to Accurate Coding Underline the main term, then locate code: Stenosis of Carotid Artery Transient Ischemic Attack Gastrointestinal hemorrhage Degenerative Joint Disease Coronary Artery Disease Alcoholic
Steps to Accurate Coding Underline the main term, then locate code: Stenosis of Carotid Artery Transient Ischemic Attack Gastrointestinal hemorrhage Degenerative Joint Disease Coronary Artery Disease Alcoholic
Pfizer Independent Grants for Learning & Change Track 2- Annual Meetings: Submission Deadlines, Areas of Interest & Educational Goals
 In order to submit a request for general meeting support through Track 2- Annual Meetings the answer to the following questions must be Yes : Does the activity align with Pfizer s areas of interest? (listed
In order to submit a request for general meeting support through Track 2- Annual Meetings the answer to the following questions must be Yes : Does the activity align with Pfizer s areas of interest? (listed
Anticholinergic medication use for female overactive bladder in the ambulatory setting in the United States.
 Página 1 de 6 PubMed darifenacin vs solifenacin Display Settings:, Sorted by Recently Added Results: 5 1. Int Urogynecol J. 2013 Oct 25. [Epub ahead of print] Anticholinergic medication use for female
Página 1 de 6 PubMed darifenacin vs solifenacin Display Settings:, Sorted by Recently Added Results: 5 1. Int Urogynecol J. 2013 Oct 25. [Epub ahead of print] Anticholinergic medication use for female
HEALTH MATTERS, INC. SUMMARY OF PROJECTS AND KEY ACCOMPLISHMENTS TO 2016
 Selected 2016 Highlights Epidemiologic research on chronic kidney disease and fistula patency, solid and hematologic malignancies Health economics and outcomes research on beta3-agonist treatment for overactive
Selected 2016 Highlights Epidemiologic research on chronic kidney disease and fistula patency, solid and hematologic malignancies Health economics and outcomes research on beta3-agonist treatment for overactive
Pfizer Pipeline. May 10, 2012
 Pfizer Pipeline May 10, 2012 Disclaimer As some programs are still confidential, some candidates may not be identified in this list. In these materials, Pfizer discloses Mechanism of Action (MOA) information
Pfizer Pipeline May 10, 2012 Disclaimer As some programs are still confidential, some candidates may not be identified in this list. In these materials, Pfizer discloses Mechanism of Action (MOA) information
2Q/FY2015 Financial Results
 2Q/FY2015 Financial Results Ended September 30, 2015 October 30, 2015 Yoshihiko Hatanaka President and CEO Astellas Pharma Inc. 0 Cautionary Statement Regarding Forward-Looking Information This material
2Q/FY2015 Financial Results Ended September 30, 2015 October 30, 2015 Yoshihiko Hatanaka President and CEO Astellas Pharma Inc. 0 Cautionary Statement Regarding Forward-Looking Information This material
Development status of OPDIVO (nivolumab) 1
 August 1, 2018 Development status of OPDIVO (nivolumab) 1 Target disease JAPAN US/EU KR/TW Melanoma (1 st ) Approved Approved Approved Melanoma (Adjuvant Therapy) Filing Approved (US) Non-small cell lung
August 1, 2018 Development status of OPDIVO (nivolumab) 1 Target disease JAPAN US/EU KR/TW Melanoma (1 st ) Approved Approved Approved Melanoma (Adjuvant Therapy) Filing Approved (US) Non-small cell lung
Third Quarter Consolidated Financial Results
 Third Quarter Consolidated Financial Results for the Fiscal Year Ending March 31, 2014 1. Overview of consolidated results 2. Highlights of Business Performance 3. Actual and Forecast of Main Subsidiary
Third Quarter Consolidated Financial Results for the Fiscal Year Ending March 31, 2014 1. Overview of consolidated results 2. Highlights of Business Performance 3. Actual and Forecast of Main Subsidiary
(U / Anti-HER3 antibody) n Pexidartinib (US) (PLX3397 / CSF-1R/KIT/FLT3-ITD. inhibitor)
 Red: Major changes after the FY2015 Q3 financial announcement on January 29, 2016 Major R&D Pipeline Therapeutic area Oncology Cardiovascular- Metabolics Phase 1 Phase 2 Phase 3 Application n DS-3032 (US/JP)
Red: Major changes after the FY2015 Q3 financial announcement on January 29, 2016 Major R&D Pipeline Therapeutic area Oncology Cardiovascular- Metabolics Phase 1 Phase 2 Phase 3 Application n DS-3032 (US/JP)
Merck Pipeline. November 1, 2017
 Merck Pipeline November 1, 2017 Lead-in Language The chart below reflects the Company s research pipeline as of November 1, 2017. Candidates shown in Phase 3 include specific products and the date such
Merck Pipeline November 1, 2017 Lead-in Language The chart below reflects the Company s research pipeline as of November 1, 2017. Candidates shown in Phase 3 include specific products and the date such
Patient Information. Basic Information on Overactive Bladder Symptoms. pubic bone. urethra. scrotum. bladder. vaginal canal
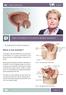 Patient Information English Basic Information on Overactive Bladder Symptoms The underlined terms are listed in the glossary. What is the bladder? pubic bone bladder seminal vesicles prostate rectum The
Patient Information English Basic Information on Overactive Bladder Symptoms The underlined terms are listed in the glossary. What is the bladder? pubic bone bladder seminal vesicles prostate rectum The
Pfizer Global Medical Grants Annual Meetings: Submission Deadlines, Primary Areas of Interest, & Educational Goals
 In order to submit a request for general meeting support for an Annual Meeting, the answers to the following questions must be Yes : Does the activity align with Pfizer s Primary Areas of Interest? (listed
In order to submit a request for general meeting support for an Annual Meeting, the answers to the following questions must be Yes : Does the activity align with Pfizer s Primary Areas of Interest? (listed
Antimicrobial prophylaxis in liver transplant A multicenter survey endorsed by the European Liver and Intestine Transplant Association
 Antimicrobial prophylaxis in liver transplant A multicenter survey endorsed by the European Liver and Intestine Transplant Association Els Vandecasteele, Jan De Waele, Dominique Vandijck, Stijn Blot, Dirk
Antimicrobial prophylaxis in liver transplant A multicenter survey endorsed by the European Liver and Intestine Transplant Association Els Vandecasteele, Jan De Waele, Dominique Vandijck, Stijn Blot, Dirk
Pharmacotherapy Handbook
 Pharmacotherapy Handbook Eighth Edition Barbara G. Wells, PharmD, HP, FCCP, BCPP Dean and Professor Executive Director, Research Institute of Pharmaceutical Sciences School of Pharmacy, The University
Pharmacotherapy Handbook Eighth Edition Barbara G. Wells, PharmD, HP, FCCP, BCPP Dean and Professor Executive Director, Research Institute of Pharmaceutical Sciences School of Pharmacy, The University
Figure 1. Comparison of Cancer Incidence Rates 1 of Individual Census Tracts with Louisiana, All Cancers Combined,
 Figure. Comparison of Cancer Incidence Rates of Individual Census Tracts with Louisiana, All Cancers Combined, 006-04 The rate is statistically significantly lower than Louisiana The rate is not statistically
Figure. Comparison of Cancer Incidence Rates of Individual Census Tracts with Louisiana, All Cancers Combined, 006-04 The rate is statistically significantly lower than Louisiana The rate is not statistically
UNC Cancer Epidemiology Seminar: Cancer Risk in New Users of Overactive Bladder Drugs
 February 19, 2016 UNC Cancer Epidemiology Seminar: Cancer Risk in New Users of Overactive Bladder Drugs James A. Kaye, MD, DrPH Senior Director, Epidemiology, RTI Health Solutions Collaborators: Andrea
February 19, 2016 UNC Cancer Epidemiology Seminar: Cancer Risk in New Users of Overactive Bladder Drugs James A. Kaye, MD, DrPH Senior Director, Epidemiology, RTI Health Solutions Collaborators: Andrea
Overactive bladder (OAB) affects approximately 15% of the adult population. Diagnosis is based
 Overactive bladder (OAB) affects approximately 15% of the adult population. Diagnosis is based upon a medical history, and includes a focused physical exam (abdominal, neurological, pelvic in females and
Overactive bladder (OAB) affects approximately 15% of the adult population. Diagnosis is based upon a medical history, and includes a focused physical exam (abdominal, neurological, pelvic in females and
Management of OAB. Lynsey McHugh. Consultant Urological Surgeon. Lancashire Teaching Hospitals
 Management of OAB Lynsey McHugh Consultant Urological Surgeon Lancashire Teaching Hospitals Summary Physiology Epidemiology Definitions NICE guidelines Evaluation Conservative management Medical management
Management of OAB Lynsey McHugh Consultant Urological Surgeon Lancashire Teaching Hospitals Summary Physiology Epidemiology Definitions NICE guidelines Evaluation Conservative management Medical management
Influenza Backgrounder
 Influenza Backgrounder Influenza Overview Influenza causes an average of 36,000 deaths and 200,000 hospitalizations in the U.S. every year. 1,2 Combined with pneumonia, influenza is the seventh leading
Influenza Backgrounder Influenza Overview Influenza causes an average of 36,000 deaths and 200,000 hospitalizations in the U.S. every year. 1,2 Combined with pneumonia, influenza is the seventh leading
Presentation Goals 4/14/2015. Pharmacology for Urinary Incontinence in Women. Medications Review anti muscarinic medications Focus on newer meds
 Presentation Goals Pharmacology for Urinary Incontinence in Women Medications Review anti muscarinic medications Focus on newer meds Introduce beta adrenergic medications Current Concepts in Drug Therapy
Presentation Goals Pharmacology for Urinary Incontinence in Women Medications Review anti muscarinic medications Focus on newer meds Introduce beta adrenergic medications Current Concepts in Drug Therapy
Merck Pipeline. As of August 3, 2015
 Merck Pipeline As of August 3, 2015 Merck Pipeline as of August 3, 2015 Phase 2 Phase 2 Phase 3 Phase 3 Phase 3 Alzheimer s Disease MK-7622 Contraception, Next Generation Ring MK-8342B Allergy, House Dust
Merck Pipeline As of August 3, 2015 Merck Pipeline as of August 3, 2015 Phase 2 Phase 2 Phase 3 Phase 3 Phase 3 Alzheimer s Disease MK-7622 Contraception, Next Generation Ring MK-8342B Allergy, House Dust
Premium Specialty: Pediatrics
 Premium Specialty: Pediatrics Credentialed Specialties include: Adolescent Medicine, Pediatric Adolescent, and Pediatrics This document is designed to be used in conjunction with the UnitedHealth Premium
Premium Specialty: Pediatrics Credentialed Specialties include: Adolescent Medicine, Pediatric Adolescent, and Pediatrics This document is designed to be used in conjunction with the UnitedHealth Premium
Tarceva. Tarceva (erlotinib) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.21.82 Subject: Tarceva Page: 1 of 5 Last Review Date: June 22, 2018 Tarceva Description Tarceva (erlotinib)
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.21.82 Subject: Tarceva Page: 1 of 5 Last Review Date: June 22, 2018 Tarceva Description Tarceva (erlotinib)
TUMOR M ARKERS MARKERS
 TUMOR MARKERS M.Shekarabi IUMS Definition Many cancers are associated with the abnormal production of some molecules l which h can be measured in plasma. These molecules are known as tumor markers. A good
TUMOR MARKERS M.Shekarabi IUMS Definition Many cancers are associated with the abnormal production of some molecules l which h can be measured in plasma. These molecules are known as tumor markers. A good
Development pipeline (as of February 1, 2017)
 Chugai Pharmaceutical Co., Ltd. (4519) Supplementary Materials for Consolidated Financial Results for FY2016 (IFRS) 13 Development pipeline (as of February 1, 2017) Oncology Additional indication name
Chugai Pharmaceutical Co., Ltd. (4519) Supplementary Materials for Consolidated Financial Results for FY2016 (IFRS) 13 Development pipeline (as of February 1, 2017) Oncology Additional indication name
HELPING YOU AND YOUR PATIENTS TALK OPENLY ABOUT MODERATELY TO SEVERELY ACTIVE RA
 SIMPONI ARIA (golimumab) is indicated for the treatment of adults with moderately to severely active rheumatoid arthritis (RA) in combination with MTX, active psoriatic arthritis, and active ankylosing
SIMPONI ARIA (golimumab) is indicated for the treatment of adults with moderately to severely active rheumatoid arthritis (RA) in combination with MTX, active psoriatic arthritis, and active ankylosing
First Quarter Consolidated Financial Results Main Product Sales Update Main R&D Activities Reference. KYORIN Co., Ltd.
 First Quarter Consolidated Financial Results for the Fiscal Year Ending March 31, 2010 Consolidated Financial Results Main Product Sales Update Main R&D Activities Reference Consolidated Financial Results
First Quarter Consolidated Financial Results for the Fiscal Year Ending March 31, 2010 Consolidated Financial Results Main Product Sales Update Main R&D Activities Reference Consolidated Financial Results
Office based non-oncology urology trials Richard W. Casey, MD, 1 Jack Barkin, MD 2
 Office based non-oncology urology trials Richard W. Casey, MD, 1 Jack Barkin, MD 2 1 The Male Health Centre, Oakville, Ontario, Canada 2 Humber River Regional Hospital, University of Toronto, Toronto,
Office based non-oncology urology trials Richard W. Casey, MD, 1 Jack Barkin, MD 2 1 The Male Health Centre, Oakville, Ontario, Canada 2 Humber River Regional Hospital, University of Toronto, Toronto,
Lower Urinary Tract Symptoms (LUTS) and Nurse-Led Clinics. Sean Diver Urology Advanced Nurse Practitioner candidate Letterkenny University Hospital
 Lower Urinary Tract Symptoms (LUTS) and Nurse-Led Clinics Sean Diver Urology Advanced Nurse Practitioner candidate Letterkenny University Hospital 01/02/2018 Lower Urinary Tract Symptoms LUTS - one of
Lower Urinary Tract Symptoms (LUTS) and Nurse-Led Clinics Sean Diver Urology Advanced Nurse Practitioner candidate Letterkenny University Hospital 01/02/2018 Lower Urinary Tract Symptoms LUTS - one of
Regulatory Status FDA-approved indications: Valcyte is a deoxynucleoside analogue cytomegalovirus (CMV) DNA polymerase inhibitor indicated for: (1)
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.01.22 Subject: Valcyte Page: 1 of 5 Last Review Date: December 8, 2017 Valcyte Description Valcyte (valganciclovir)
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.01.22 Subject: Valcyte Page: 1 of 5 Last Review Date: December 8, 2017 Valcyte Description Valcyte (valganciclovir)
Barbara G. Wells, PharmD, FASHP, FCCP, BCPP Dean and Professor School of Pharmacy, The University of Mississippi Oxford, Mississippi
 Barbara G. Wells, PharmD, FASHP, FCCP, BCPP Dean and Professor School of Pharmacy, The University of Mississippi Oxford, Mississippi Joseph T. DiPiro, PharmD, FCCP Panoz Professor of Pharmacy, College
Barbara G. Wells, PharmD, FASHP, FCCP, BCPP Dean and Professor School of Pharmacy, The University of Mississippi Oxford, Mississippi Joseph T. DiPiro, PharmD, FCCP Panoz Professor of Pharmacy, College
EMERGING FUNGAL INFECTIONS IN IMMUNOCOMPROMISED PATIENTS
 EMERGING FUNGAL INFECTIONS IN IMMUNOCOMPROMISED PATIENTS DR LOW CHIAN YONG MBBS, MRCP(UK), MMed(Int Med), FAMS Consultant, Dept of Infectious Diseases, SGH Introduction The incidence of invasive fungal
EMERGING FUNGAL INFECTIONS IN IMMUNOCOMPROMISED PATIENTS DR LOW CHIAN YONG MBBS, MRCP(UK), MMed(Int Med), FAMS Consultant, Dept of Infectious Diseases, SGH Introduction The incidence of invasive fungal
Cardiff & Vale (C&V) UHB Corporate Medicines Management Group (c MMG) SHARED CARE. Drug: TACROLIMUS Protocol number: CV 43
 Cardiff & Vale (C&V) UHB Corporate Medicines Management Group (c MMG) SHARED CARE Drug: TACROLIMUS Protocol number: CV 43 Indications: RENAL, PANCREAS OR COMBINED RENAL PANCREAS TRANSPLANTATION IN ADULTS.
Cardiff & Vale (C&V) UHB Corporate Medicines Management Group (c MMG) SHARED CARE Drug: TACROLIMUS Protocol number: CV 43 Indications: RENAL, PANCREAS OR COMBINED RENAL PANCREAS TRANSPLANTATION IN ADULTS.
Pfizer Announces Modification To Ongoing Tofacitinib FDA Post- Marketing Requirement Study In Patients With Rheumatoid Arthritis
 February 19, 2019 Media Contact: Neha Wadhwa M: +1 212-733-2835 E: Neha.Wadhwa@pfizer.com Investor Contact: Charles Triano O: +1 212-733-3901 E: Charles.E.Triano@pfizer.com Pfizer Announces Modification
February 19, 2019 Media Contact: Neha Wadhwa M: +1 212-733-2835 E: Neha.Wadhwa@pfizer.com Investor Contact: Charles Triano O: +1 212-733-3901 E: Charles.E.Triano@pfizer.com Pfizer Announces Modification
AVEO and Astellas Announce TAURUS Patient Preference Clinical Study Comparing Tivozanib with Sunitinib in First-Line Kidney Cancer
 FOR IMMEDIATE RELEASE AVEO and Astellas Announce TAURUS Patient Preference Clinical Study Comparing Tivozanib with Sunitinib in First-Line Kidney Cancer Study designed to build upon safety profile demonstrated
FOR IMMEDIATE RELEASE AVEO and Astellas Announce TAURUS Patient Preference Clinical Study Comparing Tivozanib with Sunitinib in First-Line Kidney Cancer Study designed to build upon safety profile demonstrated
Lahey Clinic Internal Medicine Residency Program: Curriculum for Infectious Disease
 Lahey Clinic Internal Medicine Residency Program: Curriculum for Infectious Disease Faculty representative: Eva Piessens, MD, MPH Resident representative: Karen Ganz, MD Revision date: February 1, 2006
Lahey Clinic Internal Medicine Residency Program: Curriculum for Infectious Disease Faculty representative: Eva Piessens, MD, MPH Resident representative: Karen Ganz, MD Revision date: February 1, 2006
Urinary Incontinence. Vibhash Mishra Consultant Urological Surgeon Royal Free Hospital
 Urinary Incontinence Vibhash Mishra Consultant Urological Surgeon Royal Free Hospital Affects women of all ages Impacts physical, psychological & social wellbeing Impact on families & carers Costs the
Urinary Incontinence Vibhash Mishra Consultant Urological Surgeon Royal Free Hospital Affects women of all ages Impacts physical, psychological & social wellbeing Impact on families & carers Costs the
Urinary Incontinence for the Primary Care Provider
 Urinary Incontinence for the Primary Care Provider Diana J Scott FNP-BC https://youtu.be/gmzaue1ojn4 1 Assessment of Urinary Incontinence Urge Stress Mixed Other overflow, postural, continuous, insensible,
Urinary Incontinence for the Primary Care Provider Diana J Scott FNP-BC https://youtu.be/gmzaue1ojn4 1 Assessment of Urinary Incontinence Urge Stress Mixed Other overflow, postural, continuous, insensible,
3Q/FY2015 Financial Results
 3Q/FY2015 Financial Results Ended December 31, 2015 January 29, 2016 Yasumasa Masuda Senior Corporate Executive, Chief Financial Officer Astellas Pharma Inc. Copyright 2015 Astellas Pharma Inc. 0 Cautionary
3Q/FY2015 Financial Results Ended December 31, 2015 January 29, 2016 Yasumasa Masuda Senior Corporate Executive, Chief Financial Officer Astellas Pharma Inc. Copyright 2015 Astellas Pharma Inc. 0 Cautionary
Tamiflu. Tamiflu (oseltamivir) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.01.19 Subject: Tamiflu Page: 1 of 5 Last Review Date: March 18, 2016 Tamiflu Description Tamiflu (oseltamivir)
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.01.19 Subject: Tamiflu Page: 1 of 5 Last Review Date: March 18, 2016 Tamiflu Description Tamiflu (oseltamivir)
Immune Checkpoint Inhibitors: The New Breakout Stars in Cancer Treatment
 Immune Checkpoint Inhibitors: The New Breakout Stars in Cancer Treatment 1 Introductions Peter Langecker, MD, PhD Executive Medical Director, Global Oncology Clinipace Worldwide Mark Shapiro Vice President
Immune Checkpoint Inhibitors: The New Breakout Stars in Cancer Treatment 1 Introductions Peter Langecker, MD, PhD Executive Medical Director, Global Oncology Clinipace Worldwide Mark Shapiro Vice President
Pfizer Independent Grants for Learning & Change Track 2- Annual Meetings: Submission Deadlines, Areas of Interest & Educational Goals
 In order to submit a request for general meeting support through Track 2- Annual Meetings the answer to the following questions must be Yes : Does the activity align with Pfizer s areas of interest? (listed
In order to submit a request for general meeting support through Track 2- Annual Meetings the answer to the following questions must be Yes : Does the activity align with Pfizer s areas of interest? (listed
Male Lower Urinary Tract Symptoms: Management in primary care and beyond. Daniel Cohen PhD FRCS(Urol) Consultant Urological Surgeon
 Male Lower Urinary Tract Symptoms: Management in primary care and beyond Daniel Cohen PhD FRCS(Urol) Consultant Urological Surgeon 1 LUTS Very common: 1/3 men over age of 50 have moderate to severe LUTS
Male Lower Urinary Tract Symptoms: Management in primary care and beyond Daniel Cohen PhD FRCS(Urol) Consultant Urological Surgeon 1 LUTS Very common: 1/3 men over age of 50 have moderate to severe LUTS
Summary of late stage clinical trials. As of Jul. 22, 2016
 Summary of late stage clinical trials As of Jul. 22, 2016 List of abbreviations AE BID DLT GFR iv MTD ORR OS PD PFS PK po PPK Q2W Q3W Q4W QD QW sc TID Adverse Events Twice daily Dose Limiting Toxicity
Summary of late stage clinical trials As of Jul. 22, 2016 List of abbreviations AE BID DLT GFR iv MTD ORR OS PD PFS PK po PPK Q2W Q3W Q4W QD QW sc TID Adverse Events Twice daily Dose Limiting Toxicity
ONO PHARMACEUTICAL CO., LTD.
 ONO PHARMACEUTICAL CO., LTD. February 6, 2006 Ono Pharmaceutical Co., Ltd. has announced its consolidated financial results for nine months ended December 31,. This First-Third Quarter Flash Report 2006
ONO PHARMACEUTICAL CO., LTD. February 6, 2006 Ono Pharmaceutical Co., Ltd. has announced its consolidated financial results for nine months ended December 31,. This First-Third Quarter Flash Report 2006
Overview of consolidated results Main Product Sales Update Development pipelines. KYORIN Holdings, Inc.
 First Quarter Consolidated Financial Results for the Fiscal Year Ending March 31, 2012 Overview of consolidated results Main Product Sales Update Development pipelines Reference Segment information for
First Quarter Consolidated Financial Results for the Fiscal Year Ending March 31, 2012 Overview of consolidated results Main Product Sales Update Development pipelines Reference Segment information for
Table E1. Standardized Mortality Ratios for Total and Specific Causes of Death Parameter Radiologists Psychiatrists No. of Deaths
 RSNA, 2016 10.1148/radiol.2016152472 Table E1. Standardized Mortality Ratios for Total and Specific Causes of Death Parameter Radiologists Psychiatrists No. of Deaths Observed/Expected No. of Deaths Observed/Expected
RSNA, 2016 10.1148/radiol.2016152472 Table E1. Standardized Mortality Ratios for Total and Specific Causes of Death Parameter Radiologists Psychiatrists No. of Deaths Observed/Expected No. of Deaths Observed/Expected
APPENDIX "C" COMPANY DEVELOPMENT PIPELINE
 Horse Horse APPENDIX "C" COMPANY DEVELOPMENT PIPELINE Animal Cancer Vaccines AUTOLOGOUS Preclinical Approval ALLOGENEIC Preclinical Approval DIRECT INJECTION Preclinical Approval Horse Q4 2016 Human Cancer
Horse Horse APPENDIX "C" COMPANY DEVELOPMENT PIPELINE Animal Cancer Vaccines AUTOLOGOUS Preclinical Approval ALLOGENEIC Preclinical Approval DIRECT INJECTION Preclinical Approval Horse Q4 2016 Human Cancer
Q1/FY2016 FINANCIAL RESULTS
 Q1/FY2016 FINANCIAL RESULTS ENDED JUNE 30, 2016 Yasumasa Masuda Senior Corporate Executive, Chief Financial Officer Astellas Pharma Inc. July 29, 2016 CAUTIONARY STATEMENT REGARDING FORWARD-LOOKING INFORMATION
Q1/FY2016 FINANCIAL RESULTS ENDED JUNE 30, 2016 Yasumasa Masuda Senior Corporate Executive, Chief Financial Officer Astellas Pharma Inc. July 29, 2016 CAUTIONARY STATEMENT REGARDING FORWARD-LOOKING INFORMATION
Dr. Melissa Kagarise, PA C
 Dr. Melissa Kagarise, PA C This program has been supported by an educational grant from Pfizer Pharmaceuticals PharmCon is accredited by the Accreditation Council for Pharmacy Education as a provider of
Dr. Melissa Kagarise, PA C This program has been supported by an educational grant from Pfizer Pharmaceuticals PharmCon is accredited by the Accreditation Council for Pharmacy Education as a provider of
All-Cause Mortality, Waterloo Region and Ontario, 2010, 2011 and 2012
 All-Cause Mortality, Waterloo Region and Ontario, 2010, 2011 and 2012 Table 1. Number of deaths and age-standardized mortality rate per 100,000 population, by place of residence, Waterloo Region and Ontario,
All-Cause Mortality, Waterloo Region and Ontario, 2010, 2011 and 2012 Table 1. Number of deaths and age-standardized mortality rate per 100,000 population, by place of residence, Waterloo Region and Ontario,
January Intravenous Nurse Day ALL MONTH LONG SUN MON TUE WED THU FRI SAT. Blood Donor Month. Glaucoma Awareness Month
 January 1 2 3 4 5 6 Blood Donor Month 7 8 9 10 11 12 13 Glaucoma Awareness Month Volunteer Blood Donor Month Cervical Health Awareness Month 14 15 16 17 18 19 20 Thyroid Awareness Month Birth Defects Prevention
January 1 2 3 4 5 6 Blood Donor Month 7 8 9 10 11 12 13 Glaucoma Awareness Month Volunteer Blood Donor Month Cervical Health Awareness Month 14 15 16 17 18 19 20 Thyroid Awareness Month Birth Defects Prevention
Management of Female Stress Incontinence
 Management of Female Stress Incontinence Dr. Arvind Goyal Associate Professor (Urology& Renal Transplant) Dayanand Medical College & Hospital, Ludhiana, Punjab, India Stress Incontinence Involuntary loss
Management of Female Stress Incontinence Dr. Arvind Goyal Associate Professor (Urology& Renal Transplant) Dayanand Medical College & Hospital, Ludhiana, Punjab, India Stress Incontinence Involuntary loss
H&HD ANTINEOPLASTIC DRUG CARD ASSEMBLY INSTRUCTIONS
 H&HD ANTINEOPLASTIC DRUG CARD ASSEMBLY INSTRUCTIONS Each of you should have 37 new cards: 7 orange cards for antimetabolites 11 white cards for miscellaneous drugs (2 DNA synthesis inhibitors, 1 enzyme,
H&HD ANTINEOPLASTIC DRUG CARD ASSEMBLY INSTRUCTIONS Each of you should have 37 new cards: 7 orange cards for antimetabolites 11 white cards for miscellaneous drugs (2 DNA synthesis inhibitors, 1 enzyme,
What is on the Horizon in Drug Therapy for OAB?
 What is on the Horizon in Drug Therapy for OAB? K-E Andersson, MD, PhD Wake Forest Institute for Regenerative Medicine Wake Forest University School of Medicine Winston Salem, North Carolina Disclosures
What is on the Horizon in Drug Therapy for OAB? K-E Andersson, MD, PhD Wake Forest Institute for Regenerative Medicine Wake Forest University School of Medicine Winston Salem, North Carolina Disclosures
Executive Summary. Reproduction prohibited v
 Kinases are a large family of proteins that have now become firmly established as a major class of drug targets for the pharmaceutical industry. The sequencing of the Human Genome has led to the identification
Kinases are a large family of proteins that have now become firmly established as a major class of drug targets for the pharmaceutical industry. The sequencing of the Human Genome has led to the identification
David Henry Jablonski, M.D North Mills Avenue Orlando, FL 32803
 David Henry Jablonski, M.D. 1812 North Mills Avenue Orlando, FL 32803 EDUCATION: University of Florida College of Medicine, Gainesville, Florida Division of Urology Residency Training Program July 1994-June
David Henry Jablonski, M.D. 1812 North Mills Avenue Orlando, FL 32803 EDUCATION: University of Florida College of Medicine, Gainesville, Florida Division of Urology Residency Training Program July 1994-June
In your own words, please write the reason you are here. Please be specific, putting in dates as necessary. Use the back of the form if needed.
 Name: SS# In your own words, please write the reason you are here. Please be specific, putting in dates as necessary. Use the back of the form if needed. Patient Medical, Surgical and Family History Review
Name: SS# In your own words, please write the reason you are here. Please be specific, putting in dates as necessary. Use the back of the form if needed. Patient Medical, Surgical and Family History Review
Summary of Risk Minimization Measures
 Table 6.1.4-1: Summary of Risk Minimization Measures Safety Concern Vaccination Hepatic and renal impairment Combination therapy Elderly Routine Risk Minimization Measures Specific subsection on vaccination
Table 6.1.4-1: Summary of Risk Minimization Measures Safety Concern Vaccination Hepatic and renal impairment Combination therapy Elderly Routine Risk Minimization Measures Specific subsection on vaccination
KEYTRUDA is also indicated in combination with pemetrexed and platinum chemotherapy for the
 FDA-Approved Indication for KEYTRUDA (pembrolizumab) in Combination With Carboplatin and Either Paclitaxel or Nab-paclitaxel for the Firstline Treatment of Patients With Metastatic Squamous Non Small Cell
FDA-Approved Indication for KEYTRUDA (pembrolizumab) in Combination With Carboplatin and Either Paclitaxel or Nab-paclitaxel for the Firstline Treatment of Patients With Metastatic Squamous Non Small Cell
Pfizer Pipeline. February 28, 2012
 Pfizer Pipeline February 28, 2012 Disclaimer As some programs are still confidential, some candidates may not be identified in this list. In these materials, Pfizer discloses Mechanism of Action (MOA)
Pfizer Pipeline February 28, 2012 Disclaimer As some programs are still confidential, some candidates may not be identified in this list. In these materials, Pfizer discloses Mechanism of Action (MOA)
INDEX OF HEALTH AND WELLNESS NEWS ITEMS January 2013-December 2014
 INDEX OF HEALTH AND WELLNESS NEWS ITEMS January 2013-December 2014 Topic (followed by title, month and page number) (Many issues can be found on the KORA website . Complete file is in
INDEX OF HEALTH AND WELLNESS NEWS ITEMS January 2013-December 2014 Topic (followed by title, month and page number) (Many issues can be found on the KORA website . Complete file is in
ENROLLMENT : Line of Business Summary
 ENROLLMENT : Line of Business Summary Date Range : JAN 2017 through DEC 2017 COMPREHENSIVE MAJOR MEDICAL Print Date : 1/19/2018 9:43:49AM Page 1 of 1 Month Year Single 2 Person : Emp/Spouse 2 Person :
ENROLLMENT : Line of Business Summary Date Range : JAN 2017 through DEC 2017 COMPREHENSIVE MAJOR MEDICAL Print Date : 1/19/2018 9:43:49AM Page 1 of 1 Month Year Single 2 Person : Emp/Spouse 2 Person :
Ontario s Referral and Listing Criteria for Adult Pancreas-After- Kidney Transplantation
 Ontario s Referral and Listing Criteria for Adult Pancreas-After- Kidney Transplantation Version 2.0 Trillium Gift of Life Network Adult Pancreas-After-Kidney Transplantation Referral & Listing Criteria
Ontario s Referral and Listing Criteria for Adult Pancreas-After- Kidney Transplantation Version 2.0 Trillium Gift of Life Network Adult Pancreas-After-Kidney Transplantation Referral & Listing Criteria
