UCRF Strategy Refresh
|
|
|
- Josephine Dorsey
- 5 years ago
- Views:
Transcription
1 UCRF Strategy Refresh Clinical Caner Gene5cs (RAM lab / UNCseq / gpath) 4/16/2014
2 Cancer is a gene5c disease * Germline familial cancer syndromes pharmacogenomics Soma5c single nucleo5de variants ( muta5on ) short inser5on / dele5ons intragenic dele5ons large dele5ons / amplifica5ons structural variants (transloca5ons) DNA RNA Protein * A disease of DNA
3
4
5 Timeline 2008 present RAM lab planning and preliminary studies UNCSeq 2012 gpath 2012 UNCSeq
6 Rapid Adop5on Molecular Lab (RAM Lab) Provides resources directly in CLIA molecular pathology lab sewng Staff Equipment Reagents Assay development / delivery in CLIA cer5fied space Mature assays Mature clinical use
7 RAM LAB Projects 1. Wash U (Ellis): PDXs and WGS Luminal tumors with AI s (n=100) 2. DFCI (Krop): Neoadjuvant Inflammatory Breast Cancers (n=80) 3. Edinborough, Scotland (Dixon): recurrence series (n=10 patent series) 4. UNC (Carey): trametnib window study with before and awer (n=15 patents) 5. Madrid, Spain (MarTn) : TNBC Neoadjuvant docetaxel/carboplatn (n=85) 6. CooperaTve Groups 1. CALGB/ALLIANCE x2 (40601 (300 HER2+) and 40603(450 TNBC)) = (~900 total samples, also two ASCO oral presentatons by Lisa Carey 2013 and 2014) 2. GEICAM (Spain): Neoadjuvant # (n=90, ASCO Oral by Aleix Prat in 2012) 3. pilot with FDA and GBG (von Minckwitz) (n=20, if good, then >1500 more) 4. pilot for TAILORx/ECOG (n=20, if good, then >2500 more) 5. pilot for TNT (metastatc TNBC 1 st line) (UK, Tuh) (n=350) 6. ECOG1199 adjuvant taxane scheduling trial (Leland- Jones) = (>2000) Nanostring work to be done (600 cases, 100 done and working on this now) 2. CBCS3 (~2000 cases to be done) 3. clinical PAM50/Prosigna offering 4. development with others (Rathmell (Renal), Kim (Bladder), Gulley (Gastric)) 5. general ncounter maintenance and miscellaneous experiments (many external PAM50 RUO requests) 6. PAM50Prosigna offering in CLIA selng for ECOG TNBC study in planning phase to enrich for Basals
8 Limita5ons Earlier stage projects Sample sets not available Technology less mature More preliminary data needed No project management or informa5cs
9 Technology Clinical Care Rapid changes in understanding of clinical cancer genetics Clinical research of Cancer Patients Ethics and regulatory considerations Tissue collecton Tissue quality Tissue type Project management Assay development
10 LCCC 1108: Development of a Tumor Molecular Analyses Program and Its Use to Support Treatment Decisions 1. To provide a mechanism for: 1. associaton of molecular alteratons (broadly defined) 2. with clinical outcome (broadly defined) in oncology 3. via genetc profiling of patent specimens (technology independent / sequencing) 2. To report results back to patent 1. Ethically compliant 2. Regulatory compliant (FDA and other) 3. Project management 1. ConsenTng and Tssue collecton 2. Tracking 3. Core transfer 4. InformaTcs
11 LCCC 1108 Execu5ve Commi^ee (Earp, Hayes, Sharpless, Grilley- Olson) Tech. Development: Hayes Chair Perou, Chiang, Sharpless, Mieczkowski, Auman, W. Kim Clinical (CCGR): Billy Kim Chair O Neil, Richards, Carey, Lee, Shores, HJ Kim, W Kim, Wu, Bae- Jump, Wang, Evans, Berg, Thomas, Ollila Earp and Grilley- Olson (ex officio) Pathology: Eberhard/Weck Chair Sharpless (ex officio) Planorm Development: Gene list for capture, reagent and plakorm needs, LIMS, muta5on calling. Clinical Use: Pa5ent lists of genes that dictate which results are reported for a clinical scenario, ethical issues related to germline analysis Technology ImplementaTon: Orthogonal tes5ng, sample collec5on and QC, billing, results repor5ng, SOPs, eventual CLIA approval
12 Produc5on Protocol office, Study staff, Tissue procurement, Clinical staff, Biospecimens processing, Pre- clinical genomics core, Genomics core, Sequencing Core, Bioinforma5cs core, Pathology
13 Production Summary Report Tue Apr 15 23:39: requested by dnhayes UNCseq Status Summary 300 active 113 sample failure Tumor 1064 consented Sample Distribution 436 completed 248 mutations reported 188 no reportable mutations Normal 215/300 distributed 227/300 distributed 35/300 pending distribution 4/300 pending distribution 49/300 not yet collected 26/300 not yet collected 0/300 not initiated 43/300 not initiated
14 Ideal Schema5c Produc5on Timeline Time to Completion Stats Early Study Total patients = 400 Physician Perception Consent to Phone Message time (344) Range:.4-16months Average: 6months Consent to Discussion (344) Range: months Average: 4.6months Sample Collection to Phone Message time (344) Range: months Average: 4.6months Processing Time Sample Collection to Discussion (344) Range: months Average: 3.3months Active Study Total patients = 539 Physician Perception Consent to Phone Message time (180) Range: months Average: 3.6months Consent to Discussion (180) Range: months Average: 2.9months Sample Collection to Phone Message time (180) Range: months Average: 2.8months Processing Time Sample Collection to Discussion (180) Range:.5-4months Average: 2.1months
15
16 Future RNA gene signature and structural rearrangements DNA from blood resistance tes5ng, personalized tumor markers Clinical trials: Targeted trials referrals
17 Required Slides for Faculty Presenta5ons (I) Papers A compact VEGF signature associated with distant metastases and poor outcomes. BMC Med :9. PMCID: (PATENT LICENSED) Grants Duke-UNC-Washington University Partnership for Early Phase Clinical Trials in Cancer, (Duke PI, Herb Hurwitz, UNC PI, Claire Dees) Top three examples Total attributable to UCRF Supervised risk predictor of breast cancer based on intrinsic subtypes. J Clin Oncol Mar 10;27(8): PMCID: (PATENT LICENSED) Prediction of lung cancer histological types by RT-qPCR gene expression in FFPE specimens. J Mol Diagn Jul;15(4): PMCID: (PATENT LICENSED) > 70 % 100% UNC NCTN Integrated Translational Science (UNITS) Center. (UNC PI, Neil Hayes, Chuck Perou) Modular phase II study to link targeted therapy to patients with pathway activated tumors: Novartis Signature Trial. (UNC PI, Juneko Grilley)
18 Required Slides for Faculty Presenta5ons (II) Impact of UCRF Investment to Date Nirali Patel, MD, molecular pathology Recruitments David Eberhard, MD, molecular pathology Key UNC collaborators UNC Molecular Pathology UNC Molecular Tumor Board UNC Clinical Genetics Key external collaborators NCTN Network (ALLIANCE, NRG) Duke Washington University
19 Acknowledgements UCRF Shelley Earp Ned Sharpless
UCRF Re- evalua,on. UNC Health Registry/Cancer Survivorship Cohort. April 16, 2014
 UCRF Re- evalua,on UNC Health Registry/Cancer Survivorship Cohort April 16, 2014 Goals Create a cancer survivorship cohort of 10,000 pa,ents diagnosed and/or treated at UNC that focuses on cancers demonstra,ng
UCRF Re- evalua,on UNC Health Registry/Cancer Survivorship Cohort April 16, 2014 Goals Create a cancer survivorship cohort of 10,000 pa,ents diagnosed and/or treated at UNC that focuses on cancers demonstra,ng
Molecular Classification of Triple-Negative Breast Cancer (TNBC) Aleix Prat, MD
 Molecular Classification of Triple-Negative Breast Cancer (TNBC) Emerging genomic and clinical data Aleix Prat, MD Postdoctoral Research Associate at Perou Lab Lineberger Comprehensive Cancer Center University
Molecular Classification of Triple-Negative Breast Cancer (TNBC) Emerging genomic and clinical data Aleix Prat, MD Postdoctoral Research Associate at Perou Lab Lineberger Comprehensive Cancer Center University
Breast cancer: Molecular STAGING classification and testing. Korourian A : AP,CP ; MD,PHD(Molecular medicine)
 Breast cancer: Molecular STAGING classification and testing Korourian A : AP,CP ; MD,PHD(Molecular medicine) Breast Cancer Theory: Halsted Operative breast cancer is a local-regional disease The positive
Breast cancer: Molecular STAGING classification and testing Korourian A : AP,CP ; MD,PHD(Molecular medicine) Breast Cancer Theory: Halsted Operative breast cancer is a local-regional disease The positive
10/15/2012. Inflammatory Breast Cancer vs. LABC: Different Biology yet Subtypes Exist
 Triple-Negative Breast Cancer: Optimizing Treatment for Locally Advanced Breast Cancer Beth Overmoyer MD Director, Inflammatory Breast Cancer Program Dana Farber Cancer Institute Overview Inflammatory
Triple-Negative Breast Cancer: Optimizing Treatment for Locally Advanced Breast Cancer Beth Overmoyer MD Director, Inflammatory Breast Cancer Program Dana Farber Cancer Institute Overview Inflammatory
OVERVIEW OF GENE EXPRESSION-BASED TESTS IN EARLY BREAST CANCER
 OVERVIEW OF GENE EXPRESSION-BASED TESTS IN EARLY BREAST CANCER Aleix Prat, MD PhD Medical Oncology Department Hospital Clínic of Barcelona University of Barcelona esmo.org Disclosures Advisory role for
OVERVIEW OF GENE EXPRESSION-BASED TESTS IN EARLY BREAST CANCER Aleix Prat, MD PhD Medical Oncology Department Hospital Clínic of Barcelona University of Barcelona esmo.org Disclosures Advisory role for
Personalized Medicine Disruptive Technology? David Logan Senior Vice President, Commercial Genomic Health Inc
 1 Personalized Medicine Disruptive Technology? David Logan Senior Vice President, Commercial Genomic Health Inc Safe Harbor Statement This presentation contains forward-looking statements within the meaning
1 Personalized Medicine Disruptive Technology? David Logan Senior Vice President, Commercial Genomic Health Inc Safe Harbor Statement This presentation contains forward-looking statements within the meaning
Predicting outcome in metastatic breast cancer
 Predicting outcome in metastatic breast cancer Aleix Prat, MD, PhD Medical Oncology Department Translational Genomics and Targeted Therapeutics in Solid Tumors Monday, 15 th January, Manchester, UK Disclosures
Predicting outcome in metastatic breast cancer Aleix Prat, MD, PhD Medical Oncology Department Translational Genomics and Targeted Therapeutics in Solid Tumors Monday, 15 th January, Manchester, UK Disclosures
UNC Board of Trustees March 25, 2010
 UNC Board of Trustees March 25, 2010 UNC Lineberger: The Public Comprehensive Cancer Center Basic research: Molecular discovery Clinical Research: Diagnostics & Therapeutics Clinical Care: Patient-centered
UNC Board of Trustees March 25, 2010 UNC Lineberger: The Public Comprehensive Cancer Center Basic research: Molecular discovery Clinical Research: Diagnostics & Therapeutics Clinical Care: Patient-centered
Accelerate Your Research with Conversant Bio
 Imagination has given us the steam engine, the telephone, the talkingmachine and the automobile, for these things had to be dreamed of before they became realities. So I believe that dreams... are likely
Imagination has given us the steam engine, the telephone, the talkingmachine and the automobile, for these things had to be dreamed of before they became realities. So I believe that dreams... are likely
Relevancia práctica de la clasificación de subtipos intrínsecos en cáncer de mama Miguel Martín Instituto de Investigación Sanitaria Gregorio Marañón
 Relevancia práctica de la clasificación de subtipos intrínsecos en cáncer de mama Miguel Martín Instituto de Investigación Sanitaria Gregorio Marañón Universidad Complutense Madrid The new technologies
Relevancia práctica de la clasificación de subtipos intrínsecos en cáncer de mama Miguel Martín Instituto de Investigación Sanitaria Gregorio Marañón Universidad Complutense Madrid The new technologies
Session II: Academic Research in Breast Cancer: Challenges and Opportunities Robert L. Comis, MD ECOG-ACRIN Group Co-Chair
 Session II: Academic Research in Breast Cancer: Challenges and Opportunities Robert L. Comis, MD ECOG-ACRIN Group Co-Chair Symposium: Innovation in Breast Cancer 2014 Madrid, Spain February 21, 2014 Cancer
Session II: Academic Research in Breast Cancer: Challenges and Opportunities Robert L. Comis, MD ECOG-ACRIN Group Co-Chair Symposium: Innovation in Breast Cancer 2014 Madrid, Spain February 21, 2014 Cancer
Contemporary Classification of Breast Cancer
 Contemporary Classification of Breast Cancer Laura C. Collins, M.D. Vice Chair of Anatomic Pathology Professor of Pathology Beth Israel Deaconess Medical Center and Harvard Medical School Boston, MA Outline
Contemporary Classification of Breast Cancer Laura C. Collins, M.D. Vice Chair of Anatomic Pathology Professor of Pathology Beth Israel Deaconess Medical Center and Harvard Medical School Boston, MA Outline
The use of diagnostic FFPE material in cancer epidemiology research
 The use of diagnostic FFPE material in cancer epidemiology research Neil O Callaghan Genetic Epidemiology Laboratory Department of Pathology The University of Melbourne www.pedigree.org.au Overview Who
The use of diagnostic FFPE material in cancer epidemiology research Neil O Callaghan Genetic Epidemiology Laboratory Department of Pathology The University of Melbourne www.pedigree.org.au Overview Who
Genomic landscape of breast cancer
 Genomic landscape of breast cancer Aleix Prat MD PhD Head of the Translational Genomics Group Attending Physician at the Breast Cancer Unit VHIO, Barcelona, Spain Major research advance for breast cancer
Genomic landscape of breast cancer Aleix Prat MD PhD Head of the Translational Genomics Group Attending Physician at the Breast Cancer Unit VHIO, Barcelona, Spain Major research advance for breast cancer
Breast cancer classification: beyond the intrinsic molecular subtypes
 Breast cancer classification: beyond the intrinsic molecular subtypes Britta Weigelt, PhD Signal Transduction Laboratory CRUK London Research Institute Summary Breast cancer heterogeneity Molecular classification
Breast cancer classification: beyond the intrinsic molecular subtypes Britta Weigelt, PhD Signal Transduction Laboratory CRUK London Research Institute Summary Breast cancer heterogeneity Molecular classification
Prosigna BREAST CANCER PROGNOSTIC GENE SIGNATURE ASSAY
 Prosigna BREAST CANCER PROGNOSTIC GENE SIGNATURE ASSAY Methodology The test is based on the reported 50-gene classifier algorithm originally named PAM50 and is performed on the ncounter Dx Analysis System
Prosigna BREAST CANCER PROGNOSTIC GENE SIGNATURE ASSAY Methodology The test is based on the reported 50-gene classifier algorithm originally named PAM50 and is performed on the ncounter Dx Analysis System
Prosigna BREAST CANCER PROGNOSTIC GENE SIGNATURE ASSAY
 Prosigna BREAST CANCER PROGNOSTIC GENE SIGNATURE ASSAY GENE EXPRESSION PROFILING WITH PROSIGNA What is Prosigna? Prosigna Breast Cancer Prognostic Gene Signature Assay is an FDA-approved assay which provides
Prosigna BREAST CANCER PROGNOSTIC GENE SIGNATURE ASSAY GENE EXPRESSION PROFILING WITH PROSIGNA What is Prosigna? Prosigna Breast Cancer Prognostic Gene Signature Assay is an FDA-approved assay which provides
A new way of looking at breast cancer tumour biology
 A new way of looking at breast cancer tumour biology Contents Intrinsic subtypes of breast cancer 3 Gene expression assays 3 Basis of the Prosigna test 4 Information provided by Prosigna 5 The accuracy
A new way of looking at breast cancer tumour biology Contents Intrinsic subtypes of breast cancer 3 Gene expression assays 3 Basis of the Prosigna test 4 Information provided by Prosigna 5 The accuracy
Circulating Tumor DNA in GIST and its Implications on Treatment
 Circulating Tumor DNA in GIST and its Implications on Treatment October 2 nd 2017 Dr. Ciara Kelly Assistant Attending Physician Sarcoma Medical Oncology Service Objectives Background Liquid biopsy & ctdna
Circulating Tumor DNA in GIST and its Implications on Treatment October 2 nd 2017 Dr. Ciara Kelly Assistant Attending Physician Sarcoma Medical Oncology Service Objectives Background Liquid biopsy & ctdna
Accelerating Innovation in Statistical Design
 Implementing a National Cancer Clinical Trials System for the 21 st Century, Workshop #2 Session #5: Accelerating Innovation Through Effective Partnerships Accelerating Innovation in Statistical Design
Implementing a National Cancer Clinical Trials System for the 21 st Century, Workshop #2 Session #5: Accelerating Innovation Through Effective Partnerships Accelerating Innovation in Statistical Design
SYSTEMIC TREATMENT OF TRIPLE NEGATIVE BREAST CANCER
 SYSTEMIC TREATMENT OF TRIPLE NEGATIVE BREAST CANCER Sunil Shrestha 1*, Ji Yuan Yang, Li Shuang and Deepika Dhakal Clinical School of Medicine, Yangtze University, Jingzhou, Hubei Province, PR. China Department
SYSTEMIC TREATMENT OF TRIPLE NEGATIVE BREAST CANCER Sunil Shrestha 1*, Ji Yuan Yang, Li Shuang and Deepika Dhakal Clinical School of Medicine, Yangtze University, Jingzhou, Hubei Province, PR. China Department
SWOG Plenary I Translational Medicine Science as the Foundation for Rapid and Meaningful Advances in Oncology
 SWOG Plenary I Translational Medicine Science as the Foundation for Rapid and Meaningful Advances in Oncology Lee M. Ellis, MD SWOG Vice Chair, Translational Medicine Departments of Surgical Oncology and
SWOG Plenary I Translational Medicine Science as the Foundation for Rapid and Meaningful Advances in Oncology Lee M. Ellis, MD SWOG Vice Chair, Translational Medicine Departments of Surgical Oncology and
10/15/2012. Biologic Subtypes of TNBC. Topics. Topics. Histopathology Molecular pathology Clinical relevance
 Biologic Subtypes of TNBC Andrea L. Richardson M.D. Ph.D. Brigham and Women s Hospital Dana-Farber Cancer Institute Harvard Medical School Boston, MA Topics Histopathology Molecular pathology Clinical
Biologic Subtypes of TNBC Andrea L. Richardson M.D. Ph.D. Brigham and Women s Hospital Dana-Farber Cancer Institute Harvard Medical School Boston, MA Topics Histopathology Molecular pathology Clinical
Importance of Methodology Certification and Accreditations to Perform Assays. Stan Hamilton, MD Head, Pathology and Laboratory Medicine
 Importance of Methodology Certification and Accreditations to Perform Assays Stan Hamilton, MD Head, Pathology and Laboratory Medicine 1 Disclosures No disclosures relevant to this presentation 2 A bad
Importance of Methodology Certification and Accreditations to Perform Assays Stan Hamilton, MD Head, Pathology and Laboratory Medicine 1 Disclosures No disclosures relevant to this presentation 2 A bad
Molecular Tes,ng on Cytology Specimens
 Molecular Tes,ng on Cytology Specimens Sco6 Boerner MD FRCPC Medical Director & Head of Cytopathology University Health Network Associate Professor, University of Toronto sco6.boerner@uhn.ca UNIVERSITY
Molecular Tes,ng on Cytology Specimens Sco6 Boerner MD FRCPC Medical Director & Head of Cytopathology University Health Network Associate Professor, University of Toronto sco6.boerner@uhn.ca UNIVERSITY
A biomarker-driven approach for the development of the ICOS agonist antibody, JTX-2011 Heather A. Hirsch
 A biomarker-driven approach for the development of the ICOS agonist antibody, JTX-2011 Heather A. Hirsch On behalf of Jounce Therapeutics JTX-2011 team Immuno-Oncology Biomarkers: Today s Imperatives for
A biomarker-driven approach for the development of the ICOS agonist antibody, JTX-2011 Heather A. Hirsch On behalf of Jounce Therapeutics JTX-2011 team Immuno-Oncology Biomarkers: Today s Imperatives for
Analysis of circulating free DNA in peripheral blood. Piotr Mieczkowski University of North Carolina at Chapel Hill
 Analysis of circulating free DNA in peripheral blood Piotr Mieczkowski University of North Carolina at Chapel Hill Genomics2014 Next Generation Sequencing for Clinical Care of Cancer Patients - UNCseq
Analysis of circulating free DNA in peripheral blood Piotr Mieczkowski University of North Carolina at Chapel Hill Genomics2014 Next Generation Sequencing for Clinical Care of Cancer Patients - UNCseq
Study Finds Lung Cancer Subtype Test from UNC, Startup GeneCentric to be Accurate, Reproducible
 Study Finds Lung Cancer Subtype Test from UNC, Startup GeneCentric to be Accurate, Reproducible May 30, 2013 By Ben Butkus A team led by clinical researchers from the University of North Carolina, Chapel
Study Finds Lung Cancer Subtype Test from UNC, Startup GeneCentric to be Accurate, Reproducible May 30, 2013 By Ben Butkus A team led by clinical researchers from the University of North Carolina, Chapel
Terapia sistemica neoadiuvante: in quali tumori? Quali risultati? Dott. Giacomo Pelizzari
 Terapia sistemica neoadiuvante: in quali tumori? Quali risultati? Dott. Giacomo Pelizzari Neoadjuvant Treatment A window of opportunity Rational: Historically proposed to enable breast-conserving surgery
Terapia sistemica neoadiuvante: in quali tumori? Quali risultati? Dott. Giacomo Pelizzari Neoadjuvant Treatment A window of opportunity Rational: Historically proposed to enable breast-conserving surgery
Triple-Negative Breast Cancer
 June 2017 Triple-Negative Breast Cancer Amir Sonnenblick, MD, PhD Sharett institute of oncology Hadassah-Hebrew university medical center, Jerusalem, Israel This presentation is the intellectual property
June 2017 Triple-Negative Breast Cancer Amir Sonnenblick, MD, PhD Sharett institute of oncology Hadassah-Hebrew university medical center, Jerusalem, Israel This presentation is the intellectual property
Biomarkers for HER2-directed Therapies : Past Failures and Future Perspectives
 Biomarkers for HER2-directed Therapies : Past Failures and Future Perspectives Ian Krop Dana-Farber Cancer Institute Harvard Medical School Inchon 2018 Adjuvant Trastuzumab Improves Outcomes in HER2+ Breast
Biomarkers for HER2-directed Therapies : Past Failures and Future Perspectives Ian Krop Dana-Farber Cancer Institute Harvard Medical School Inchon 2018 Adjuvant Trastuzumab Improves Outcomes in HER2+ Breast
Basket and Umbrella Trial Designs in Oncology
 Basket and Umbrella Trial Designs in Oncology Eric Polley Biomedical Statistics and Informatics Mayo Clinic Polley.Eric@mayo.edu Dose Selection for Cancer Treatment Drugs Stanford Medicine May 2017 1 /
Basket and Umbrella Trial Designs in Oncology Eric Polley Biomedical Statistics and Informatics Mayo Clinic Polley.Eric@mayo.edu Dose Selection for Cancer Treatment Drugs Stanford Medicine May 2017 1 /
A view from the Oncology Clinic. Andrew Tu7 Consultant Clinical Oncologist
 A view from the Oncology Clinic Andrew Tu7 Consultant Clinical Oncologist Our pa'ents have become much more aware and are driving a changing demand Before surgery: MRI for contralateral BC; neoadjuvant
A view from the Oncology Clinic Andrew Tu7 Consultant Clinical Oncologist Our pa'ents have become much more aware and are driving a changing demand Before surgery: MRI for contralateral BC; neoadjuvant
Adjuvant Endocrine Therapy: How Long is Long Enough?
 Adjuvant Endocrine Therapy: How Long is Long Enough? Harold J. Burstein, MD, PhD Dana-Farber Cancer Institute Harvard Medical School Boston, Massachusetts hburstein@partners.org I have no conflicts to
Adjuvant Endocrine Therapy: How Long is Long Enough? Harold J. Burstein, MD, PhD Dana-Farber Cancer Institute Harvard Medical School Boston, Massachusetts hburstein@partners.org I have no conflicts to
Key points about expression profile intrinsic subtypes. Analytically robust expression profiles from FFPE sections using Nanostring technology
 Analytically robust expression profiles from FFPE sections using Nanostring technology Torsten O. Nielsen, MD/PhD, FRCPC Professor of Pathology and Laboratory Medicine University of British Columbia, Vancouver,
Analytically robust expression profiles from FFPE sections using Nanostring technology Torsten O. Nielsen, MD/PhD, FRCPC Professor of Pathology and Laboratory Medicine University of British Columbia, Vancouver,
Role of chemotherapy in BRCA and Triple negative breast cancer. Fernando Moreno Servicio de Oncología Médica Hospital Clinico San Carlos
 Role of chemotherapy in BRCA and Triple negative breast cancer Fernando Moreno Servicio de Oncología Médica Hospital Clinico San Carlos Association between TNBC & germline mutations in BRCA 1/2 TNBC is
Role of chemotherapy in BRCA and Triple negative breast cancer Fernando Moreno Servicio de Oncología Médica Hospital Clinico San Carlos Association between TNBC & germline mutations in BRCA 1/2 TNBC is
Molecular Heterogeneity of Triple Negative Breast Cancer
 Molecular Heterogeneity of Triple Negative Breast Cancer Katherine A. Hoadley Department of Genetics Lineberger Comprehensive Cancer Center The University of North Carolina at Chapel Hill Estimated Cancer
Molecular Heterogeneity of Triple Negative Breast Cancer Katherine A. Hoadley Department of Genetics Lineberger Comprehensive Cancer Center The University of North Carolina at Chapel Hill Estimated Cancer
ALCHEMIST. Adjuvant Lung Cancer Enrichment Marker Identification And Sequencing Trials
 ALCHEMIST Adjuvant Lung Cancer Enrichment Marker Identification And Sequencing Trials What is ALCHEMIST? ALCHEMIST is 3 integrated trials testing targeted therapy in early stage lung cancer: l A151216:
ALCHEMIST Adjuvant Lung Cancer Enrichment Marker Identification And Sequencing Trials What is ALCHEMIST? ALCHEMIST is 3 integrated trials testing targeted therapy in early stage lung cancer: l A151216:
Triple-Negative Breast Cancer Time to Slice and Dice? Carsten Denkert, MD Charité University Hospital Berlin, Germany
 Triple-Negative Breast Cancer Time to Slice and Dice? Carsten Denkert, MD Charité University Hospital Berlin, Germany Triple-Negative Breast Cancer (TNBC) 2018 Presentation Outline The molecular heterogeneity
Triple-Negative Breast Cancer Time to Slice and Dice? Carsten Denkert, MD Charité University Hospital Berlin, Germany Triple-Negative Breast Cancer (TNBC) 2018 Presentation Outline The molecular heterogeneity
Impressions of a New NCI Director: Big Data
 Impressions of a New NCI Director: Big Data Norman E. Sharpless, M.D. Cancer Informatics for Cancer Centers April 3, 2018 www.cancer.gov www.cancer.gov/espanol October 17, 2018 NCI: Leading the National
Impressions of a New NCI Director: Big Data Norman E. Sharpless, M.D. Cancer Informatics for Cancer Centers April 3, 2018 www.cancer.gov www.cancer.gov/espanol October 17, 2018 NCI: Leading the National
Cancer Genomics and Personalized Oncology. Dirce M Carraro, PhD Scien0st, Head of Genomics and Biology Molecular Group
 Cancer Genomics and Personalized Oncology Dirce M Carraro, PhD Scien0st, Head of Genomics and Biology Molecular Group Cancer in Brazil 600,000 new cases/year Popula0on: 210,867,959 Number of new cases:
Cancer Genomics and Personalized Oncology Dirce M Carraro, PhD Scien0st, Head of Genomics and Biology Molecular Group Cancer in Brazil 600,000 new cases/year Popula0on: 210,867,959 Number of new cases:
FAQs for UK Pathology Departments
 FAQs for UK Pathology Departments This is an educational piece written for Healthcare Professionals FAQs for UK Pathology Departments If you would like to discuss any of the listed FAQs further, or have
FAQs for UK Pathology Departments This is an educational piece written for Healthcare Professionals FAQs for UK Pathology Departments If you would like to discuss any of the listed FAQs further, or have
Controversies in Breast Pathology ELENA PROVENZANO ADDENBROOKES HOSPITAL, CAMBRIDGE
 Controversies in Breast Pathology ELENA PROVENZANO ADDENBROOKES HOSPITAL, CAMBRIDGE Neoadjuvant Chemotherapy Indications: Management of locally advanced invasive breast cancers including inflammatory breast
Controversies in Breast Pathology ELENA PROVENZANO ADDENBROOKES HOSPITAL, CAMBRIDGE Neoadjuvant Chemotherapy Indications: Management of locally advanced invasive breast cancers including inflammatory breast
Biologic Subtypes and Prognos5c Factors. Claudine Isaacs, MD Georgetown University
 Biologic Subtypes and Prognos5c Factors Claudine Isaacs, MD Georgetown University Prognos5c Factor Defini5on Predicts outcome in absence of systemic therapy Thus tell us when (not how) to treat a pa5ent
Biologic Subtypes and Prognos5c Factors Claudine Isaacs, MD Georgetown University Prognos5c Factor Defini5on Predicts outcome in absence of systemic therapy Thus tell us when (not how) to treat a pa5ent
Gene Signatures in Breast Cancer: Moving Beyond ER, PR, and HER2? Lisa A. Carey, M.D. University of North Carolina USA
 Gene Signatures in Breast Cancer: Moving Beyond ER, PR, and HER2? Lisa A. Carey, M.D. University of North Carolina USA When Are Biomarkers Ready To Use? Same Rules for Gene Expression Panels Key elements
Gene Signatures in Breast Cancer: Moving Beyond ER, PR, and HER2? Lisa A. Carey, M.D. University of North Carolina USA When Are Biomarkers Ready To Use? Same Rules for Gene Expression Panels Key elements
Alternate Gene Signatures, or Not
 Charles M. Perou, Ph.D. Departments of Genetics and Pathology Lineberger Comprehensive Cancer Center University of North Carolina Chapel Hill, North Carolina, USA Alternate Gene Signatures, or Not Charles
Charles M. Perou, Ph.D. Departments of Genetics and Pathology Lineberger Comprehensive Cancer Center University of North Carolina Chapel Hill, North Carolina, USA Alternate Gene Signatures, or Not Charles
RNA preparation from extracted paraffin cores:
 Supplementary methods, Nielsen et al., A comparison of PAM50 intrinsic subtyping with immunohistochemistry and clinical prognostic factors in tamoxifen-treated estrogen receptor positive breast cancer.
Supplementary methods, Nielsen et al., A comparison of PAM50 intrinsic subtyping with immunohistochemistry and clinical prognostic factors in tamoxifen-treated estrogen receptor positive breast cancer.
THE SEARCH FOR BIOMARKERS IN BLADDER CANCER
 THE SEARCH FOR BIOMARKERS IN BLADDER CANCER CDDP and IO WORLD ALEJO RODRÍGUEZ-VIDA MD PhD Consultant Medical Oncologist Associate Professor Hospital del Mar, Barcelona November 23 rd 2018 DISCLOSURE OF
THE SEARCH FOR BIOMARKERS IN BLADDER CANCER CDDP and IO WORLD ALEJO RODRÍGUEZ-VIDA MD PhD Consultant Medical Oncologist Associate Professor Hospital del Mar, Barcelona November 23 rd 2018 DISCLOSURE OF
IOM-ASCO Workshop. Cooperative Group Chairs Report. March 21, Jan Buckner, MD. Mayo Clinic Rochester, MN
 IOM-ASCO Workshop March 21, 2011 Cooperative Group Chairs Report Jan Buckner, MD Mayo Clinic Rochester, MN Therapeutic Advances Cooperative Groups have been successfully collaborating to conduct high-quality
IOM-ASCO Workshop March 21, 2011 Cooperative Group Chairs Report Jan Buckner, MD Mayo Clinic Rochester, MN Therapeutic Advances Cooperative Groups have been successfully collaborating to conduct high-quality
Follow-up Visit Procedures MTN-009
 Follow-up Visit Procedures MTN-009 General Overview MTN-009 follow-up visits are only required for participants who are found to be HIV infected These participants will have a minimum of two follow-up
Follow-up Visit Procedures MTN-009 General Overview MTN-009 follow-up visits are only required for participants who are found to be HIV infected These participants will have a minimum of two follow-up
The Neoadjuvant Model as a Translational Tool for Drug and Biomarker Development in Breast Cancer
 The Neoadjuvant Model as a Translational Tool for Drug and Biomarker Development in Breast Cancer Laura Spring, MD Breast Medical Oncology Massachusetts General Hospital Primary Mentor: Dr. Aditya Bardia
The Neoadjuvant Model as a Translational Tool for Drug and Biomarker Development in Breast Cancer Laura Spring, MD Breast Medical Oncology Massachusetts General Hospital Primary Mentor: Dr. Aditya Bardia
Decipher Bladder Predicts Which Patients May Benefit from Neoadjuvant Chemotherapy Prior to Radical Cystectomy
 Decipher Bladder Predicts Which Patients May Benefit from Neoadjuvant Chemotherapy Prior to Cystectomy Contact the GenomeDx Customer Support Team 1.888.792.1601 (toll-free) customersupport@genomedx.com
Decipher Bladder Predicts Which Patients May Benefit from Neoadjuvant Chemotherapy Prior to Cystectomy Contact the GenomeDx Customer Support Team 1.888.792.1601 (toll-free) customersupport@genomedx.com
Nielsen et al. BMC Cancer 2014, 14:177
 Nielsen et al. BMC Cancer 2014, 14:177 TECHNICAL ADVANCE Open Access Analytical validation of the PAM50-based Prosigna Breast Cancer Prognostic Gene Signature Assay and ncounter Analysis System using formalin-fixed
Nielsen et al. BMC Cancer 2014, 14:177 TECHNICAL ADVANCE Open Access Analytical validation of the PAM50-based Prosigna Breast Cancer Prognostic Gene Signature Assay and ncounter Analysis System using formalin-fixed
Genetically Engineered Murine Models of RCC William Y. Kim, M.D.
 Genetically Engineered Murine Models of RCC William Y. Kim, M.D. Co-director, Mouse Phase 1 Unit (MP1U) Departments of Medicine, Genetics, and Urology Lineberger Comprehensive Cancer Center University
Genetically Engineered Murine Models of RCC William Y. Kim, M.D. Co-director, Mouse Phase 1 Unit (MP1U) Departments of Medicine, Genetics, and Urology Lineberger Comprehensive Cancer Center University
Comparison of Triple Negative Breast Cancer between Asian and Western Data Sets
 2010 IEEE International Conference on Bioinformatics and Biomedicine Workshops Comparison of Triple Negative Breast Cancer between Asian and Western Data Sets Lee H. Chen Bioinformatics and Biostatistics
2010 IEEE International Conference on Bioinformatics and Biomedicine Workshops Comparison of Triple Negative Breast Cancer between Asian and Western Data Sets Lee H. Chen Bioinformatics and Biostatistics
Triple Negative Breast Cancer
 Triple Negative Breast Cancer Prof. Dr. Pornchai O-charoenrat Division of Head-Neck & Breast Surgery Department of Surgery Faculty of Medicine Siriraj Hospital Breast Cancer Classification Traditional
Triple Negative Breast Cancer Prof. Dr. Pornchai O-charoenrat Division of Head-Neck & Breast Surgery Department of Surgery Faculty of Medicine Siriraj Hospital Breast Cancer Classification Traditional
Therapeutic Targets for Triple- Negative Breast Cancer: Focus on Platinums and EGFR Inhibition
 Therapeutic Targets for Triple- Negative Breast Cancer: Focus on Platinums and EGFR Inhibition Lisa A Carey, MD Disclosures for Lisa A Carey, MD No real or apparent conflicts of interest to disclose Basal-Like
Therapeutic Targets for Triple- Negative Breast Cancer: Focus on Platinums and EGFR Inhibition Lisa A Carey, MD Disclosures for Lisa A Carey, MD No real or apparent conflicts of interest to disclose Basal-Like
Conflicts of Interest and Entrepreneurship
 Conflicts of Interest and Entrepreneurship Amita Verma (av234) Guilaine Senecal (gds64) June 16, 2015 7/8/2015 1 Cornell Policies on Conflict of Interest Cornell Policy 1.7, Financial Conflicts of Interest
Conflicts of Interest and Entrepreneurship Amita Verma (av234) Guilaine Senecal (gds64) June 16, 2015 7/8/2015 1 Cornell Policies on Conflict of Interest Cornell Policy 1.7, Financial Conflicts of Interest
Advances in Breast Cancer Therapeutics in the Adjuvant and Metastatic Settings. Eve Rodler, MD University of California at Davis October 2016
 Advances in Breast Cancer Therapeutics in the Adjuvant and Metastatic Settings Eve Rodler, MD University of California at Davis October 2016 17th Annual Advances in Oncology September 30-October 1, 2016
Advances in Breast Cancer Therapeutics in the Adjuvant and Metastatic Settings Eve Rodler, MD University of California at Davis October 2016 17th Annual Advances in Oncology September 30-October 1, 2016
Liquid Biopsies. Next Generation Cancer Molecular Diagnostics
 Liquid Biopsies Next Generation Cancer Molecular Diagnostics Forward Looking Statements 2 Statements pertaining to future financial and/or operating results, future research, diagnostic tests and technology
Liquid Biopsies Next Generation Cancer Molecular Diagnostics Forward Looking Statements 2 Statements pertaining to future financial and/or operating results, future research, diagnostic tests and technology
Updates in Molecular Classification of Breast Cancer. Emmanuel Agosto-Arroyo, MD Assistant Member Department of Anatomic Pathology
 Updates in Molecular Classification of Breast Cancer Emmanuel Agosto-Arroyo, MD Assistant Member Department of Anatomic Pathology 3/3/2018 Disclosure There are no conflicts of interest. Objectives Describe
Updates in Molecular Classification of Breast Cancer Emmanuel Agosto-Arroyo, MD Assistant Member Department of Anatomic Pathology 3/3/2018 Disclosure There are no conflicts of interest. Objectives Describe
Life Sciences Consortium Task Force Report to The National Cancer Policy Forum Workshop
 Life Sciences Consortium Task Force Report to The National Cancer Policy Forum Workshop Gregory A. Curt, MD, Chair U.S. Medical Science Lead, Emerging Products AstraZeneca -Oncology CEO Roundtable-I May
Life Sciences Consortium Task Force Report to The National Cancer Policy Forum Workshop Gregory A. Curt, MD, Chair U.S. Medical Science Lead, Emerging Products AstraZeneca -Oncology CEO Roundtable-I May
FEP Medical Policy Manual
 FEP Medical Policy Manual Effective Date: October 15, 2018 Related Policies: 2.04.38 Cytochrome P450 Genotype-Guided Treatment Strategy Genotype-Guided Tamoxifen Treatment Description Tamoxifen is prescribed
FEP Medical Policy Manual Effective Date: October 15, 2018 Related Policies: 2.04.38 Cytochrome P450 Genotype-Guided Treatment Strategy Genotype-Guided Tamoxifen Treatment Description Tamoxifen is prescribed
Clinical Utility of Diagnostic Tests
 Clinical Utility of Diagnostic Tests David A. Eberhard MD, PhD Director, Pre-Clinical Genomic Pathology, Lineberger Comprehensive Cancer Center Associate Professor, Depts. of Pathology and Pharmacology
Clinical Utility of Diagnostic Tests David A. Eberhard MD, PhD Director, Pre-Clinical Genomic Pathology, Lineberger Comprehensive Cancer Center Associate Professor, Depts. of Pathology and Pharmacology
Adjuvant endocrine therapy (essentials in ER positive early breast cancer)
 Adjuvant endocrine therapy (essentials in ER positive early breast cancer) Giuseppe Curigliano MD, PhD Breast Cancer Program Division of Experimental Therapeutics Outline Picking optimal adjuvant endocrine
Adjuvant endocrine therapy (essentials in ER positive early breast cancer) Giuseppe Curigliano MD, PhD Breast Cancer Program Division of Experimental Therapeutics Outline Picking optimal adjuvant endocrine
When is Chemotherapy indicated in Advanced Luminal Breast Cancer?
 When is Chemotherapy indicated in Advanced Luminal Breast Cancer? Soo-Chin Lee Head & Senior Consultant Department of Haematology-Oncology Clinical Care National University Cancer Institute, Singapore
When is Chemotherapy indicated in Advanced Luminal Breast Cancer? Soo-Chin Lee Head & Senior Consultant Department of Haematology-Oncology Clinical Care National University Cancer Institute, Singapore
Breast Cancer Assays of Genetic Expression in Tumor Tissue
 Breast Cancer Assays of Genetic Expression in Tumor Tissue Policy Number: Original Effective Date: MM.12.009 12/02/2008 Line(s) of Business: Current Effective Date Section: 05/25/2018 Other Miscellaneous
Breast Cancer Assays of Genetic Expression in Tumor Tissue Policy Number: Original Effective Date: MM.12.009 12/02/2008 Line(s) of Business: Current Effective Date Section: 05/25/2018 Other Miscellaneous
Early Stage Disease. Hope S. Rugo, MD Professor of Medicine Director Breast Oncology and Clinical Trials Education UCSF Comprehensive Cancer Center
 SABCS 2014: Early Stage Disease Hope S. Rugo, MD Professor of Medicine Director Breast Oncology and Clinical Trials Education UCSF Comprehensive Cancer Center Topics for Discussion Chemotherapy plus 10
SABCS 2014: Early Stage Disease Hope S. Rugo, MD Professor of Medicine Director Breast Oncology and Clinical Trials Education UCSF Comprehensive Cancer Center Topics for Discussion Chemotherapy plus 10
The TAILORx Trial: A review of the data and implications for practice
 The TAILORx Trial: A review of the data and implications for practice Angela DeMichele, MD, MSCE Jill & Alan Miller Endowed Chair in Breast Cancer Excellence Professor of Medicine and Epidemiology University
The TAILORx Trial: A review of the data and implications for practice Angela DeMichele, MD, MSCE Jill & Alan Miller Endowed Chair in Breast Cancer Excellence Professor of Medicine and Epidemiology University
ncounter Assay Automated Process Immobilize and align reporter for image collecting and barcode counting ncounter Prep Station
 ncounter Assay ncounter Prep Station Automated Process Hybridize Reporter to RNA Remove excess reporters Bind reporter to surface Immobilize and align reporter Image surface Count codes Immobilize and
ncounter Assay ncounter Prep Station Automated Process Hybridize Reporter to RNA Remove excess reporters Bind reporter to surface Immobilize and align reporter Image surface Count codes Immobilize and
Emerging Tissue and Serum Markers
 Emerging Tissue and Serum Markers for Immune Checkpoint Inhibitors Kyong Hwa Park MD, PhD Medical Oncology Korea University College of Medicine Contents Immune checkpoint inhibitors in clinical practice
Emerging Tissue and Serum Markers for Immune Checkpoint Inhibitors Kyong Hwa Park MD, PhD Medical Oncology Korea University College of Medicine Contents Immune checkpoint inhibitors in clinical practice
INDIVIDUALIZED MEDICINE
 CENTER FOR INDIVIDUALIZED MEDICINE Precision Oncology: Current Applications of omics ACP Arizona Chapter Scientific Meeting, 2014 Arizona State University in Tempe, Arizona Alan Bryce, MD 2012 MFMER slide-1
CENTER FOR INDIVIDUALIZED MEDICINE Precision Oncology: Current Applications of omics ACP Arizona Chapter Scientific Meeting, 2014 Arizona State University in Tempe, Arizona Alan Bryce, MD 2012 MFMER slide-1
Understanding and Improving Quality of Cancer Care in Diverse Underserved Populations
 Understanding and Improving Quality of Cancer Care in Diverse Underserved Populations Stephanie B Wheeler, PhD MPH Department of Health Policy & Management Overview Research Interests CDC-Special Interest
Understanding and Improving Quality of Cancer Care in Diverse Underserved Populations Stephanie B Wheeler, PhD MPH Department of Health Policy & Management Overview Research Interests CDC-Special Interest
Role of Genomic Profiling in (Minimally) Node Positive Breast Cancer
 Role of Genomic Profiling in (Minimally) Node Positive Breast Cancer Kathy S. Albain, MD, FACP Professor of Medicine Dean s Scholar Loyola University Chicago Stritch School of Medicine Cardinal Bernardin
Role of Genomic Profiling in (Minimally) Node Positive Breast Cancer Kathy S. Albain, MD, FACP Professor of Medicine Dean s Scholar Loyola University Chicago Stritch School of Medicine Cardinal Bernardin
PRECISION INSIGHTS. GPS Cancer. Molecular Insights You Can Rely On. Tumor-normal sequencing of DNA + RNA expression.
 PRECISION INSIGHTS GPS Cancer Molecular Insights You Can Rely On Tumor-normal sequencing of DNA + RNA expression www.nanthealth.com Cancer Care is Evolving Oncologists use all the information available
PRECISION INSIGHTS GPS Cancer Molecular Insights You Can Rely On Tumor-normal sequencing of DNA + RNA expression www.nanthealth.com Cancer Care is Evolving Oncologists use all the information available
Biology of Young Breast Cancer and Management in Pregnant Women
 19 th BSMO Annual Meeting 2017 Breast Cancer Task Force Biology of Young Breast Cancer and Management in Pregnant Women Matteo Lambertini, MD ESMO Fellow Institut Jules Bordet, Brussels Diegem, Belgium
19 th BSMO Annual Meeting 2017 Breast Cancer Task Force Biology of Young Breast Cancer and Management in Pregnant Women Matteo Lambertini, MD ESMO Fellow Institut Jules Bordet, Brussels Diegem, Belgium
Design considerations for Phase II trials incorporating biomarkers
 Design considerations for Phase II trials incorporating biomarkers Sumithra J. Mandrekar Professor of Biostatistics, Mayo Clinic Pre-Meeting Workshop Enhancing the Design and Conduct of Phase II Studies
Design considerations for Phase II trials incorporating biomarkers Sumithra J. Mandrekar Professor of Biostatistics, Mayo Clinic Pre-Meeting Workshop Enhancing the Design and Conduct of Phase II Studies
Circulating tumor cells/dna/etc for Radiation Oncologists
 Circulating tumor cells/dna/etc for Radiation Oncologists Andrew Z. Wang, M.D. Associate Professor Director of Clinical and Translational Research Department of Radiation Oncology Carolina Center for Cancer
Circulating tumor cells/dna/etc for Radiation Oncologists Andrew Z. Wang, M.D. Associate Professor Director of Clinical and Translational Research Department of Radiation Oncology Carolina Center for Cancer
SKCC Protocol Review Committee New Study Application
 Instructions: Submit the following documents to prc@jefferson.edu - Completed New Study Application (aka MCSF) - Protocol - Protocol Facilitation Committee Approval (if applicable) - MDG Priority Score
Instructions: Submit the following documents to prc@jefferson.edu - Completed New Study Application (aka MCSF) - Protocol - Protocol Facilitation Committee Approval (if applicable) - MDG Priority Score
Bayesian hierarchical models for adaptive randomization in biomarker-driven studies: Umbrella and platform trials
 Bayesian hierarchical models for adaptive randomization in biomarker-driven studies: Umbrella and platform trials William T. Barry, PhD Nancy and Morris John Lurie Investigator Biostatistics and Computational
Bayesian hierarchical models for adaptive randomization in biomarker-driven studies: Umbrella and platform trials William T. Barry, PhD Nancy and Morris John Lurie Investigator Biostatistics and Computational
CHARLES M. PEROU. The University of North Carolina at Chapel Hill, Chapel Hill, North Carolina, USA
 The Oncologist Molecular Stratification of Triple-Negative Breast Cancers CHARLES M. PEROU The University of North Carolina at Chapel Hill, Chapel Hill, North Carolina, USA Key Words. Triple-negative breast
The Oncologist Molecular Stratification of Triple-Negative Breast Cancers CHARLES M. PEROU The University of North Carolina at Chapel Hill, Chapel Hill, North Carolina, USA Key Words. Triple-negative breast
TABLE OF CONTENTS. Executive Summary The EndoPredict Test Intended Use Population Breast Cancer Clinical Dilemma. Analytical Validity
 Clinical Dossier TABLE OF CONTENTS Executive Summary The EndoPredict Test Intended Use Population Breast Cancer Clinical Dilemma 1 2 3 Analytical Validity 4 Clinical Validity 4 Clinical Utility 7 Medicare
Clinical Dossier TABLE OF CONTENTS Executive Summary The EndoPredict Test Intended Use Population Breast Cancer Clinical Dilemma 1 2 3 Analytical Validity 4 Clinical Validity 4 Clinical Utility 7 Medicare
NeoadjuvantTreatment In BC When, How, Who?
 NeoadjuvantTreatment In BC When, How, Who? Clifford Hudis, M.D. Chief, Breast Cancer Medicine Service, MSKCC Professor of Medicine, Weill Cornell Medical College President, ASCO 15 Potential Benefits Of
NeoadjuvantTreatment In BC When, How, Who? Clifford Hudis, M.D. Chief, Breast Cancer Medicine Service, MSKCC Professor of Medicine, Weill Cornell Medical College President, ASCO 15 Potential Benefits Of
Reliable Evaluation of Prognostic & Predictive Genomic Tests
 Reliable Evaluation of Prognostic & Predictive Genomic Tests Richard Simon, D.Sc. Chief, Biometric Research Branch National Cancer Institute http://brb.nci.nih.gov Different Kinds of Biomarkers Prognostic
Reliable Evaluation of Prognostic & Predictive Genomic Tests Richard Simon, D.Sc. Chief, Biometric Research Branch National Cancer Institute http://brb.nci.nih.gov Different Kinds of Biomarkers Prognostic
NSABP B-55/BIG Lynne Suhayda, RN, MSEd. Director, Clinical Coordinating Department. NRG Oncology - Pittsburgh, Pennsylvania
 NSABP B-55/BIG 6-13 Lynne Suhayda, RN, MSEd. Director, Clinical Coordinating Department NRG Oncology - Pittsburgh, Pennsylvania NRG Oncology Meeting July 15, 2016 Lynne Suhayda, RN, MSEd. No Financial
NSABP B-55/BIG 6-13 Lynne Suhayda, RN, MSEd. Director, Clinical Coordinating Department NRG Oncology - Pittsburgh, Pennsylvania NRG Oncology Meeting July 15, 2016 Lynne Suhayda, RN, MSEd. No Financial
A case of a BRCA2-mutated ER+/HER2 breast cancer during pregnancy
 ESMO Preceptorship Programme Breast Cancer Lisbon 16,17 September 2016 Emanuela Risi Sandro Pitigliani Medical Oncology Department Hospital of Prato, Istituto Toscano Tumori, Prato, Italy A case of a BRCA2-mutated
ESMO Preceptorship Programme Breast Cancer Lisbon 16,17 September 2016 Emanuela Risi Sandro Pitigliani Medical Oncology Department Hospital of Prato, Istituto Toscano Tumori, Prato, Italy A case of a BRCA2-mutated
Breast Cancer: ASCO Poster Review
 Breast Cancer: ASCO Poster Review Carmen Criscitiello, MD, PhD Istituto Europeo di Oncologia Milano HER2+ SUBTYPE Research questions in early HER2+ BC De-escalation of toxicity without compromising efficacy
Breast Cancer: ASCO Poster Review Carmen Criscitiello, MD, PhD Istituto Europeo di Oncologia Milano HER2+ SUBTYPE Research questions in early HER2+ BC De-escalation of toxicity without compromising efficacy
Next Generation Cancer Diagnostics For First Time Right Therapy Choice. Anja van de Stolpe
 Next Generation Cancer Diagnostics For First Time Right Therapy Choice Anja van de Stolpe Paradigm shift in cancer treatment towards personalized treatment Chemotherapy for all therapy targeting cancer
Next Generation Cancer Diagnostics For First Time Right Therapy Choice Anja van de Stolpe Paradigm shift in cancer treatment towards personalized treatment Chemotherapy for all therapy targeting cancer
ncounter Assay Automated Process Capture & Reporter Probes Bind reporter to surface Remove excess reporters Hybridize CodeSet to RNA
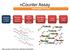 ncounter Assay Automated Process Hybridize CodeSet to RNA Remove excess reporters Bind reporter to surface Immobilize and align reporter Image surface Count codes mrna Capture & Reporter Probes slides
ncounter Assay Automated Process Hybridize CodeSet to RNA Remove excess reporters Bind reporter to surface Immobilize and align reporter Image surface Count codes mrna Capture & Reporter Probes slides
U.S. Army Medical Research and Materiel Command Fort Detrick, Maryland
 AWARD NUMBER: DoD Award W81XWH-13-1-0451 TITLE: The Genomic Evolution of Prostate Cancer PRINCIPAL INVESTIGATOR: David VanderWeele, M.D., Ph.D. CONTRACTING ORGANIZATION: University of Chicago, Chicago,
AWARD NUMBER: DoD Award W81XWH-13-1-0451 TITLE: The Genomic Evolution of Prostate Cancer PRINCIPAL INVESTIGATOR: David VanderWeele, M.D., Ph.D. CONTRACTING ORGANIZATION: University of Chicago, Chicago,
What to do after pcr in different subtypes?
 What to do after pcr in different subtypes? Luca Moscetti Breast Unit Università degli Studi di Modena e Reggio Emilia Policlinico di Modena, Italy Aims of neoadjuvant therapy in breast cancer Primary
What to do after pcr in different subtypes? Luca Moscetti Breast Unit Università degli Studi di Modena e Reggio Emilia Policlinico di Modena, Italy Aims of neoadjuvant therapy in breast cancer Primary
The document history table was updated to include Amendment 6.
 For Protocol Amendment #6 to: RTOG 0913, Phase I/II Trial of Concurrent RAD001 (Everolimus) With Temozolomide/Radiation Followed by Adjuvant RAD001/Temozolomide in Newly Diagnosed Glioblastoma NCI Protocol
For Protocol Amendment #6 to: RTOG 0913, Phase I/II Trial of Concurrent RAD001 (Everolimus) With Temozolomide/Radiation Followed by Adjuvant RAD001/Temozolomide in Newly Diagnosed Glioblastoma NCI Protocol
Subtype-directed therapy of TNBC Global Breast Cancer Conference 2015 & 4th International Breast Cancer Symposium Jeju Island, Korea, April 2015
 Subtype-directed therapy of TNBC Global Breast Cancer Conference 2015 & 4th International Breast Cancer Symposium Jeju Island, Korea, April 2015 Ruth M. O Regan, MD Visiting Professor and Division Chief
Subtype-directed therapy of TNBC Global Breast Cancer Conference 2015 & 4th International Breast Cancer Symposium Jeju Island, Korea, April 2015 Ruth M. O Regan, MD Visiting Professor and Division Chief
Triple Negative Breast Cancer: Part 2 A Medical Update
 Triple Negative Breast Cancer: Part 2 A Medical Update April 29, 2015 Tiffany A. Traina, MD Breast Medicine Service Memorial Sloan Kettering Cancer Center Weill Cornell Medical College Overview What is
Triple Negative Breast Cancer: Part 2 A Medical Update April 29, 2015 Tiffany A. Traina, MD Breast Medicine Service Memorial Sloan Kettering Cancer Center Weill Cornell Medical College Overview What is
Prostate Cancer Clinical Trials - PHEN s Rally Update and Next Steps
 Prostate Cancer Clinical Trials - PHEN s Rally Update and Next Steps 13 th Annual African- American Prostate Cancer Disparity Summit PHEN: Prostate Health Educa?on Network Bernard W. Parker MD, FACP CAPT,
Prostate Cancer Clinical Trials - PHEN s Rally Update and Next Steps 13 th Annual African- American Prostate Cancer Disparity Summit PHEN: Prostate Health Educa?on Network Bernard W. Parker MD, FACP CAPT,
NSABP: FB-11. Shannon Puhalla, MD
 NSABP: FB-11 Phase II Randomized Study Evaluating the Biological and Clinical Effects of the Combination of Palbociclib with Letrozole as Neoadjuvant Therapy in Post- Menopausal Women with Estrogen Receptor
NSABP: FB-11 Phase II Randomized Study Evaluating the Biological and Clinical Effects of the Combination of Palbociclib with Letrozole as Neoadjuvant Therapy in Post- Menopausal Women with Estrogen Receptor
Reporting of Breast Cancer Do s and Don ts
 Reporting of Breast Cancer Do s and Don ts 7 th SGH Annual Breast Pathology Course Professor Michael Bilous Conjoint Professor Western Sydney University Consultant Pathologist, Australian Clinical Labs,
Reporting of Breast Cancer Do s and Don ts 7 th SGH Annual Breast Pathology Course Professor Michael Bilous Conjoint Professor Western Sydney University Consultant Pathologist, Australian Clinical Labs,
Enterprise Interest Speaker and consultant for Astrazeneca, MSD, BMS, Roche, Pfizer, Novartis, Sanofi Resarch grants from BMS, Roche, MSD, Novartis
 Enterprise Interest Speaker and consultant for Astrazeneca, MSD, BMS, Roche, Pfizer, Novartis, Sanofi Resarch grants from BMS, Roche, MSD, Novartis Centre Jean Perrin Centre de Lutte contre le Cancer d'auvergne
Enterprise Interest Speaker and consultant for Astrazeneca, MSD, BMS, Roche, Pfizer, Novartis, Sanofi Resarch grants from BMS, Roche, MSD, Novartis Centre Jean Perrin Centre de Lutte contre le Cancer d'auvergne
TNBC: What s new Déjà vu All Over Again? Lucy R. Langer, MD MSHS Compass Oncology - SABCS 2016 Review February 21, 2017
 TNBC: What s new Déjà vu All Over Again? Lucy R. Langer, MD MSHS Compass Oncology - SABCS 2016 Review February 21, 2017 The problem with TNBC 1. Generally more aggressive 2. ONLY chemotherapy 3. No other
TNBC: What s new Déjà vu All Over Again? Lucy R. Langer, MD MSHS Compass Oncology - SABCS 2016 Review February 21, 2017 The problem with TNBC 1. Generally more aggressive 2. ONLY chemotherapy 3. No other
Gene Expression Profiling for Managing Breast Cancer Treatment. Policy Specific Section: Medical Necessity and Investigational / Experimental
 Medical Policy Gene Expression Profiling for Managing Breast Cancer Treatment Type: Medical Necessity and Investigational / Experimental Policy Specific Section: Laboratory/Pathology Original Policy Date:
Medical Policy Gene Expression Profiling for Managing Breast Cancer Treatment Type: Medical Necessity and Investigational / Experimental Policy Specific Section: Laboratory/Pathology Original Policy Date:
