|
|
|
- Piers Perry
- 5 years ago
- Views:
Transcription
1 COMPANY PROFILE
2 Table of Content 1. Cellvax Presentation Cellvax History Management team Cellvax Preclinical Studies Services Collaboration and Business Development Why contact us? Contact Information... 10
3 1. Cellvax Presentation Cellvax, founded in 2001, is a French preclinical CRO which provides complete innovating drug validation studies both in vitro and in vivo allowing to accelerate the drug development process for unmet needs related to severe human diseases, mainly in Cancer, Diabetes and Osteoarthritis (OA). Oncology Leading cause of death worldwide - Test in vitro; -Primary Tumor cells; -Nodule syste; Animal models, S.C, orthotopic tumor models; -Metastasis; Angiogenesis. Diabetes Huge market with continuous growth -Elisa Test (Glucose, insulin); -Diet-induced animal models; -Chemical-induced animal models; -Genetic-induced animal models. Osteoarthritis Affecting large number of population -Animal models; -Induced OA, ACLT model; -Spontaneous OA model. Toxicology, PK/PD, Biomarkers and other customized models Today, Cellvax directly employs motivated and differently skilled scientists and engineers, working in interdisciplinary teams on several innovative products. Cellvax positioning in drug discovery process: Candidate Molecule Identification Preclinical Trials Phase I Phase II Phase III Phase IV Commercialization Drug Surveillance Cellvax Services Our services allow speeding up the development of your drug candidates, valorizing drug potential and decreasing the cost of your drug development. Cellvax, SAS, France, Tel : + 33 (0) , contact@cellvax-pharma.com, Web: 2
4 2. Cellvax History Year Event 2001 Foundation of Cellvax 2006 French Government Grant OSEO 2007 First Eureka Project (Cancer Field) 2015 New facility allowing multiple animal models 2015 Eurostars Program (Breast Cancer) 2015 Official Partner with Biobanques INSERM 2016 EURONANOMED projects in cancer and OA fields large EU Grants and 2 large French Grants Bpifrance 2017 Eurostars Project (Cancer) 3. Management team Dr. Mingxing WEI Founder and Chief Executive Officer Ph.D. in molecular and cellular biology, University of Lyon I. HDR University Paris XIII. Researcher at «Harvard Medical School», USA. Training Officer in animal experimentation in France. Marketing & Finance training. Head of project in Biotech. Pr. Michel CREPIN Co-founder Ph.D. Professor University Paris XIII. Director of Institut d oncologie cellulaire et moléculaire humaine ( ). Director of university laboratory EA445 ( ). Director of INSERM team (U553), Saint- Louis hospital, Paris (since 2000). Cellvax, SAS, France, Tel : + 33 (0) , contact@cellvax-pharma.com, Web: 3
5 Dr. Christian SCHATZ Co-founder MD, Doctor of Pharmacy, Ph.D., HDR University of Strasbourg, Aptitude certificate in enterprise administration. Medical director, former member of executive committee of Transgène. Mr. Olivier-Noël MARTIN Co-founder Graduate of E.S.C.P. & DECS (1969), President of General Biscuit / Danone USA, President of KAYSERBERG society. He has created and sold to GLAXOSMITHKLINE in 2002 the STERILYO laboratories. Investment in 6 start-up biotech. 4. Cellvax Preclinical Studies Services Anti-proliferative tests in vitro Cellvax proposes in their services to realize different tests in vitro on established cell lines or primary cultures: anti-proliferative tests, PK study, cell cycle, apoptosis, etc. Animal cancer models Cellvax propose not only subcutaneous tumor models but also original orthotopic tumor models in animal in which we induce lung cancer, colon cancer, pancreas cancer, prostate cancer, breast cancer, gastric cancer, brain tumor, leukemia. Angiogenic tests in vitro & in vivo Angiogenesis plays an important role in the tumoral development. Cellvax proposes pro or anti-angiogenic tests. An in vivo original model is also available and consists on a Matrigel implantation in mouse and on a measure of the drug candidates effects on the microvessels formation. Cellvax, SAS, France, Tel : + 33 (0) , contact@cellvax-pharma.com, Web: 4
6 Tumoral invasion tests in vitro Cellvax proposes in vitro tests which allow measuring the anti-metastatic potential of drug candidates. These techniques allow studying the intravasation and extravasation potentials of tumor cells through endothelium. Orthotopic tumor models Cellvax proposes to realise orthotopic models for drug validation studies in breast cancer, colon cancer, lung cancer, pancreas cancer, kidney cancer, prostate cancer, melanoma, glioblastoma, etc. Syngeneic tumor models in animals It is extremely usual to employ syngeneic immuno-competent animal tumor models for the preclinical evaluation of test agents. For this purpose, Cellvax have developed different syngeneic tumor models for Leukemia, Melanoma, Bladder cancer, Prostate cancer, Pancreatic tumor, Kidney cancer, Fibrosarcoma, Breast Cancer, Brain tumor, Colon cancer, Pancreatic tumor, Mastocytoma, Hepatic Cancer, Lung cancer. Original drug-resistant tumor models in vitro & in vivo Intrinsic or acquired tumor-mediated drug resistance is a major clinical problem that can result in the lack of tumor response to chemotherapy. In order to continuously improve the quality and the originality of Cellvax s oncology services, we have successfully established several drug-resistant tumor models: lung cancer, pancreatic tumor, ovary cancer, etc. In vivo imaging in alive animals For these studies, we can propose several imaging technologies: PET Scan, Scanner, X-ray, MRI, Bioluminescence. The new technology, PET (Positron Emission Tomography) Scan, by labeling with 18FDG (Fluoro-Deoxy-d- Glucose), allows to observe phenomenon (micro-metastasis formation, drug bio-distribution, etc) in the same alive animal at different time points without sacrificing animals. Cellvax, SAS, France, Tel : + 33 (0) , contact@cellvax-pharma.com, Web: 5
7 Primary tumor cells Testing your promising compounds by using human primary tumor cells is the closest way to the real clinical situation. These data would be very useful for your future clinical trials. The established tumor cell lines, after numerous passages in vitro, might lose their initial biological characteristics. In contrast, these primary tumor cells (freshly established from human biopsies) conserve these biological characteristics and could be an ideal tool in anti-cancer studies. More relevant tumor models Direct implantation of human tumor biopsies in mice To reconstitute tumor micro-environment; To be as close as possible to human tumors; Tumors are maintained from mice to mice, without using cell culture system. Non-rodent cancer models, spontaneous tumors Osteoarthritis animal models For this disease, Cellvax can propose two types of animal models: Induced Osteoarthritis (OA) Animal Models: Anterior Cruciate Ligament Transection (ACLT) in rabbits, rats, dogs...; Meniscectomy; Patellectomy; Immobilization; Ovariectomy: Postmenopausal OA; Enzymatic / chemically induced OA; Others. Spontaneous Osteoarthritis (OA) Animal Models: Mouse (DBA/1, STR/ORT, C57BL/6, C57); Guinea pig (Hartley, Duncan-Hartley); Others. Cellvax, SAS, France, Tel : + 33 (0) , contact@cellvax-pharma.com, Web: 6
8 Toxicity studies in vivo A complete toxicity experiment can be proposed at Cellvax in both rodent and non-rodents: Acute toxicity in mice, animals; Sub-acute toxicity in animals; Chronic toxicity in animals; MTD (Maximum Tolerated Dose) with single dose and/or repeated doses; Abnormal toxicity in two animal species according to European guidelines; Complete blood count (CBC). (Total blood analysis: Red cells; White cells; Platelets; Hemoglobin) and Chemistry profile (Alanine aminotransferase (ALT); Aspartate aminotransferase (AST); Alkaline phosphatase (ALP); Creatinine kinase; Glucose); Monitoring: animal body weight, behavior, eventual death; Hematological and biochemical analysis; Macroscopic observation and histological analysis on main organs. Bio-distribution and PK studies in vivo In order to understand the pharmacokinetics (PK) of your innovating drugs, we propose a bio-distribution and PK studies in mice, rats, beagle dogs, mini-pigs. At different time points (Typical time points: 30 Min; 1h; 3h; 6h; 12h; 24h; 32h; 48h and 96h), the presence of your drug or its metabolites will be searched and analyzed in different organs (Liver; Spleen; Lung; Heart; Brain; Intestine; Adipose tissues; Skin; Bone; Serum; Urine, etc.). Mechanisms of actions studies Topoisomerases I & II, required during DNA replication, transcription and homologous recombination; Cell cycle analysis Cells, stained with BrdU (Bromodeoxyuridin) and IP (iodide propidium); Specific biomarkers by ICH Proliferation marker, Ki-67; angiogenesis marker, CD-31, T cell infiltrations in tumors (CD4+ or CD8+), hypoxia, etc.; Cellvax, SAS, France, Tel : + 33 (0) , contact@cellvax-pharma.com, Web: 7
9 Apoptosis analysis: Cells labeled with Annexin V and IP. Doublestained cells analyzed by FACScan; ELISA tests; Western blot analysis; Enzymes assays. Complete histological analysis and/or with experts interpretations Tissues fixation in 4% Formalin; Frozen tissues; Paraffin-embedded or frozen sections; H&E and/or immuno-staining. Other specific studies upon request Cellvax is also able to provide customized studies according to your specific needs in order to fully satisfy the specific needs of our customers. HSV-1 and HSV-2 infection models in mice. Diabetic models: In vitro models; In vivo model for Type I diabetes; In vivo model for Type II diabetes. Parkinson diseases: In vitro models; In vivo models. Ascite production: Ascite production in mice by administration of the incomplete Freund's adjuvant. Cellvax, SAS, France, Tel : + 33 (0) , contact@cellvax-pharma.com, Web: 8
10 5. Collaboration and Business Development The will to collaborate Cellvax is a preclinical service company with strong expertise mainly in Oncology and Osteoarthritis fields. Our interactive collaborations allow accelerating the development of your drug candidates. Cellvax has a strong will to collaborate with public laboratories, biotech and/or with pharmaceutical companies which develop novel compounds. These collaborations could be very enriching and will allow speeding up the development of drug candidates. Partnering strategy Cellvax is also seeking to establish close collaboration and/or partnership to develop or co-develop innovating compounds with a greater speed and less cost. 6. Why contact us? Expertise of our founder and operational teams; Complementary and extensive biological tests; Reactivity and efficiency of services; Confidentiality and experimental procedures with Quality Insurance manager; State of the art animal facility with rodents and non-rodents; Access to a network of public and private laboratories; Continuous improvement of our tests in order to be more competitive; Our services allow to speed up the development of your drug candidates; Our services allow to valorize drug potential in several pathologies; Our services allow decreasing your cost of drug development; Support of external marketing; A Window company or a local partner in France and in Europe. Cellvax, SAS, France, Tel : + 33 (0) , contact@cellvax-pharma.com, Web: 9
11 7. Contact Information Adress: Parc BIOCITECH 102, Avenue Gaston Roussel 93230, Romainville France Fax: + 33 (0) Tel: Mobile: + 33 (0) (0)
EDUCATIONAL SEMINAR. Humanized Mouse Models in Pre- Clinical Efficacy Studies in Oncology. BIOCOM CRO Event January 23, 2018
 EDUCATIONAL SEMINAR Humanized Mouse Models in Pre- Clinical Efficacy Studies in Oncology BIOCOM CRO Event January 23, 2018 Agenda 8.00 8.30 Registration and networking 8.30 8.45 Introduction, by Jussi
EDUCATIONAL SEMINAR Humanized Mouse Models in Pre- Clinical Efficacy Studies in Oncology BIOCOM CRO Event January 23, 2018 Agenda 8.00 8.30 Registration and networking 8.30 8.45 Introduction, by Jussi
SU2C TOP SCIENCE ACCOMPLISHMENTS
 SU2C TOP SCIENCE ACCOMPLISHMENTS INTRODUCTION Since our founding in 2008, Stand Up To Cancer has funded 87 team science projects, with budgets ranging from $250,000 for a short research project to over
SU2C TOP SCIENCE ACCOMPLISHMENTS INTRODUCTION Since our founding in 2008, Stand Up To Cancer has funded 87 team science projects, with budgets ranging from $250,000 for a short research project to over
Innovative Nuclear Receptor Modulation Medicine
 Innovative Nuclear Receptor Modulation Medicine Dale C Leitman, M.D., Ph.D. Isaac Cohen, O.M.D., Ph.D. October 2015 Corporate Mission Iaterion, Inc. is Developing Novel Pharmaceutical Nuclear Receptor
Innovative Nuclear Receptor Modulation Medicine Dale C Leitman, M.D., Ph.D. Isaac Cohen, O.M.D., Ph.D. October 2015 Corporate Mission Iaterion, Inc. is Developing Novel Pharmaceutical Nuclear Receptor
Melanoma Research Alliance-Pfizer Academic-Industry Partnership Awards
 Melanoma Research Alliance-Pfizer Academic-Industry Partnership Awards Wendy K.D. Selig, President & CEO, MRA Julia Perkins, M.D. Medical Director, Oncology, Pfizer Melanoma Research Alliance Mission:
Melanoma Research Alliance-Pfizer Academic-Industry Partnership Awards Wendy K.D. Selig, President & CEO, MRA Julia Perkins, M.D. Medical Director, Oncology, Pfizer Melanoma Research Alliance Mission:
numares at a glance company overview 2014 Discover, understand and use biological systems high level NMR analytics
 numares at a glance company overview 2014 Discover, understand and use biological systems high level NMR analytics Three benefits of numares 1 Supporting patients to receive better cure based on effective
numares at a glance company overview 2014 Discover, understand and use biological systems high level NMR analytics Three benefits of numares 1 Supporting patients to receive better cure based on effective
Human TNFα Transgenic Mouse Model of Spontaneous Arthritis
 Human TNFα Transgenic Mouse Model of Spontaneous Arthritis HEIDELBERG PHARMA AT A GLANCE CRO situated in Ladenburg, near Heidelberg, Germany 45 employees, 2000 m 2 of lab space Core competence: pre-clinical
Human TNFα Transgenic Mouse Model of Spontaneous Arthritis HEIDELBERG PHARMA AT A GLANCE CRO situated in Ladenburg, near Heidelberg, Germany 45 employees, 2000 m 2 of lab space Core competence: pre-clinical
NEWS RELEASE Media Contact: Krysta Pellegrino (650) Investor Contact: Sue Morris (650) Advocacy Contact: Kristin Reed (650)
 NEWS RELEASE Media Contact: Krysta Pellegrino (650) 225-8226 Investor Contact: Sue Morris (650) 225-6523 Advocacy Contact: Kristin Reed (650) 467-9831 FDA APPROVES AVASTIN IN COMBINATION WITH CHEMOTHERAPY
NEWS RELEASE Media Contact: Krysta Pellegrino (650) 225-8226 Investor Contact: Sue Morris (650) 225-6523 Advocacy Contact: Kristin Reed (650) 467-9831 FDA APPROVES AVASTIN IN COMBINATION WITH CHEMOTHERAPY
Revolutionizing the Treatment of Cancer
 Revolutionizing the Treatment of Cancer June 2014 Safe Harbor Statement The statements that follow (including projections and business trends) are forward looking statements. Rexahn's actual results may
Revolutionizing the Treatment of Cancer June 2014 Safe Harbor Statement The statements that follow (including projections and business trends) are forward looking statements. Rexahn's actual results may
Bayer to Present New Data on Advancing Oncology Portfolio. New Preclinical and Early Clinical Data Evaluating Innovative Compounds in Drug Development
 Investor News Not intended for U.S. and UK Media Bayer AG Investor Relations 51368 Leverkusen Germany www.investor.bayer.com 105 th Annual Meeting American Association for Cancer Research Bayer to Present
Investor News Not intended for U.S. and UK Media Bayer AG Investor Relations 51368 Leverkusen Germany www.investor.bayer.com 105 th Annual Meeting American Association for Cancer Research Bayer to Present
Your partner for Medical Research and Development
 Your partner for Medical Research and Development Overview Integrated place for clinical research SERVICES AND RESOURCES DNA Sequencing Proteomics Imaging Histology Flow Cytometry Sample Processing Cryogenics
Your partner for Medical Research and Development Overview Integrated place for clinical research SERVICES AND RESOURCES DNA Sequencing Proteomics Imaging Histology Flow Cytometry Sample Processing Cryogenics
IMIDIA IMPROVING BETA-CELL FUNCTION AND IDENTIFICATION OF DIAGNOSTIC BIOMARKERS FOR TREATMENT MONITORING IN DIABETES. A. Ktorza, B.
 IMIDIA IMPROVING BETA-CELL FUNCTION AND IDENTIFICATION OF DIAGNOSTIC BIOMARKERS FOR TREATMENT MONITORING IN DIABETES A. Ktorza, B. Thorens DIABETES Definition Diabetes is a metabolic disease characterized
IMIDIA IMPROVING BETA-CELL FUNCTION AND IDENTIFICATION OF DIAGNOSTIC BIOMARKERS FOR TREATMENT MONITORING IN DIABETES A. Ktorza, B. Thorens DIABETES Definition Diabetes is a metabolic disease characterized
C A N C É R O P Ô L E L Y O N A U V E R G N E R H Ô N E - A L P E S
 Cancer Research Cluster to Facilitate and Structure Oncology Research C A N C É R O P Ô L E L Y O N A U V E R G N E R H Ô N E - A L P E S s P E E D I N G U P P R O G R E S S CANCÉROPÔLE LYON AUVERGNE RHÔNE-ALPES
Cancer Research Cluster to Facilitate and Structure Oncology Research C A N C É R O P Ô L E L Y O N A U V E R G N E R H Ô N E - A L P E S s P E E D I N G U P P R O G R E S S CANCÉROPÔLE LYON AUVERGNE RHÔNE-ALPES
POSITRON EMISSION TOMOGRAPHY (PET)
 Status Active Medical and Behavioral Health Policy Section: Radiology Policy Number: V-27 Effective Date: 08/27/2014 Blue Cross and Blue Shield of Minnesota medical policies do not imply that members should
Status Active Medical and Behavioral Health Policy Section: Radiology Policy Number: V-27 Effective Date: 08/27/2014 Blue Cross and Blue Shield of Minnesota medical policies do not imply that members should
Supplementary Materials. for Garmy-Susini, et al, Integrin 4 1 signaling is required for lymphangiogenesis and tumor metastasis
 Supplementary Materials for Garmy-Susini, et al, Integrin 4 1 signaling is required for lymphangiogenesis and tumor metastasis 1 Supplementary Figure Legends Supplementary Figure 1: Integrin expression
Supplementary Materials for Garmy-Susini, et al, Integrin 4 1 signaling is required for lymphangiogenesis and tumor metastasis 1 Supplementary Figure Legends Supplementary Figure 1: Integrin expression
EXCELLENCE AND INNOVATION
 EXCELLENCE AND INNOVATION FOR CANCER TREATMENT THE CENTRE LÉON BÉRARD Non-profit private health care facility of public interest (ESPIC), affiliated with the French National Cancer Center Federation UNICANCER
EXCELLENCE AND INNOVATION FOR CANCER TREATMENT THE CENTRE LÉON BÉRARD Non-profit private health care facility of public interest (ESPIC), affiliated with the French National Cancer Center Federation UNICANCER
Revolutionizing the Treatment of Cancer
 Revolutionizing the Treatment of Cancer January 2014 Safe Harbor Statement The statements that follow (including projections and business trends) are forward looking statements. Rexahn's actual results
Revolutionizing the Treatment of Cancer January 2014 Safe Harbor Statement The statements that follow (including projections and business trends) are forward looking statements. Rexahn's actual results
GLG Pharma, LLC. A Specialty Pharmaceutical Company developing the next generation of targeted drugs
 GLG Pharma, LLC A Specialty Pharmaceutical Company developing the next generation of targeted drugs Privately Held Company Summary Experienced Management Team Development stage full spectrum Marketed Products:
GLG Pharma, LLC A Specialty Pharmaceutical Company developing the next generation of targeted drugs Privately Held Company Summary Experienced Management Team Development stage full spectrum Marketed Products:
VeriStrat Poor Patients Show Encouraging Overall Survival and Progression Free Survival Signal; Confirmatory Phase 2 Study Planned by Year-End
 AVEO and Biodesix Announce Exploratory Analysis of VeriStrat-Selected Patients with Non-Small Cell Lung Cancer in Phase 2 Study of Ficlatuzumab Presented at ESMO 2014 Congress VeriStrat Poor Patients Show
AVEO and Biodesix Announce Exploratory Analysis of VeriStrat-Selected Patients with Non-Small Cell Lung Cancer in Phase 2 Study of Ficlatuzumab Presented at ESMO 2014 Congress VeriStrat Poor Patients Show
PRESS RELEASE FOR IMMEDIATE RELEASE SINGAPORE INSTITUTES COLLABORATE WITH SAMSUNG MEDICAL CENTER TO IMPROVE TREATMENT OF LIVER CANCER
 PRESS RELEASE FOR IMMEDIATE RELEASE SINGAPORE INSTITUTES COLLABORATE WITH SAMSUNG MEDICAL CENTER TO IMPROVE TREATMENT OF LIVER CANCER This collaboration creates the world s first clinically reliable and
PRESS RELEASE FOR IMMEDIATE RELEASE SINGAPORE INSTITUTES COLLABORATE WITH SAMSUNG MEDICAL CENTER TO IMPROVE TREATMENT OF LIVER CANCER This collaboration creates the world s first clinically reliable and
CTA Strengths. Organisational Structure Board. CTA Board CEO. Background to Cancer Trials Australia What we do well An increasing struggle Conclusions
 Models for Clinical Research: Cancer Trials Australia Professor Mark Rosenthal Chairman; Cancer Trials Australia and Director of Medical Oncology, Royal Melbourne Hospital For discussion: Background to
Models for Clinical Research: Cancer Trials Australia Professor Mark Rosenthal Chairman; Cancer Trials Australia and Director of Medical Oncology, Royal Melbourne Hospital For discussion: Background to
Revolutionizing the Treatment of Cancer
 Revolutionizing the Treatment of Cancer March 2014 Safe Harbor Statement The statements that follow (including projections and business trends) are forward-looking statements. Rexahn's actual results may
Revolutionizing the Treatment of Cancer March 2014 Safe Harbor Statement The statements that follow (including projections and business trends) are forward-looking statements. Rexahn's actual results may
28, rue de Londres Paris -France Tel March-16
 28, rue de Londres 75009 Paris -France Tel +33 178 42 35 04 contact@o-ameisen.org @O_Ameisen_assoc www.o-ameisen.org March-16 Olivier Ameisen s medical discovery Baclofen improves efficacy in therapy-resistant
28, rue de Londres 75009 Paris -France Tel +33 178 42 35 04 contact@o-ameisen.org @O_Ameisen_assoc www.o-ameisen.org March-16 Olivier Ameisen s medical discovery Baclofen improves efficacy in therapy-resistant
microrna Therapeutics Harnessing the power of micrornas to target multiple pathways of disease
 microrna Therapeutics Harnessing the power of micrornas to target multiple pathways of disease January 2018 Safe Harbor Statement Statements contained in this presentation regarding matters that are not
microrna Therapeutics Harnessing the power of micrornas to target multiple pathways of disease January 2018 Safe Harbor Statement Statements contained in this presentation regarding matters that are not
Safe Harbor Statement
 Safe Harbor Statement This document may include statements that constitute forward looking statements, which are often characterized by the terms may, believes, expects or anticipates and do not reflect
Safe Harbor Statement This document may include statements that constitute forward looking statements, which are often characterized by the terms may, believes, expects or anticipates and do not reflect
Inflammatory Cells and Metastasis
 Inflammatory Cells and Metastasis Experimentelle Krebsforschung SS 07 Gerhard Christofori Institute of Biochemistry and Genetics Department of Clinical-Biological Sciences Center of Biomedicine University
Inflammatory Cells and Metastasis Experimentelle Krebsforschung SS 07 Gerhard Christofori Institute of Biochemistry and Genetics Department of Clinical-Biological Sciences Center of Biomedicine University
Novel RCC Targets from Immuno-Oncology and Antibody-Drug Conjugates
 Novel RCC Targets from Immuno-Oncology and Antibody-Drug Conjugates Christopher Turner, MD Vice President, Clinical Science 04 November 2016 Uveal Melanoma Celldex Pipeline CANDIDATE INDICATION Preclinical
Novel RCC Targets from Immuno-Oncology and Antibody-Drug Conjugates Christopher Turner, MD Vice President, Clinical Science 04 November 2016 Uveal Melanoma Celldex Pipeline CANDIDATE INDICATION Preclinical
TIME IS THE KEY IN CANCER DIAGNOSTICS. THIS IS WHY WE HAVE CREATED INPROBE MINIMALLY INVASIVE MEDICAL DEVICE CAPABLE OF REAL-TIME DIAGNOSTICS OF HER2
 TIME IS THE KEY IN CANCER DIAGNOSTICS. THIS IS WHY WE HAVE CREATED INPROBE MINIMALLY INVASIVE MEDICAL DEVICE CAPABLE OF REAL-TIME DIAGNOSTICS OF HER2 BREAST CANCER AND HER2 STOMACH CANCER BIOMARKERS. GLOBAL
TIME IS THE KEY IN CANCER DIAGNOSTICS. THIS IS WHY WE HAVE CREATED INPROBE MINIMALLY INVASIVE MEDICAL DEVICE CAPABLE OF REAL-TIME DIAGNOSTICS OF HER2 BREAST CANCER AND HER2 STOMACH CANCER BIOMARKERS. GLOBAL
Decipher Bladder Predicts Which Patients May Benefit from Neoadjuvant Chemotherapy Prior to Radical Cystectomy
 Decipher Bladder Predicts Which Patients May Benefit from Neoadjuvant Chemotherapy Prior to Cystectomy Contact the GenomeDx Customer Support Team 1.888.792.1601 (toll-free) customersupport@genomedx.com
Decipher Bladder Predicts Which Patients May Benefit from Neoadjuvant Chemotherapy Prior to Cystectomy Contact the GenomeDx Customer Support Team 1.888.792.1601 (toll-free) customersupport@genomedx.com
Research For the Future. Len Lichtenfeld, MD, MACP Interim Chief Medical and Scientific Officer
 Research For the Future Len Lichtenfeld, MD, MACP Interim Chief Medical and Scientific Officer 1 2 ATTACKING CANCER FROM EVERY ANGLE 3 CANCER PREVENTION STUDIES (CPS) Intramural Research Smoking Obesity
Research For the Future Len Lichtenfeld, MD, MACP Interim Chief Medical and Scientific Officer 1 2 ATTACKING CANCER FROM EVERY ANGLE 3 CANCER PREVENTION STUDIES (CPS) Intramural Research Smoking Obesity
Approval based on the successful BFORE Phase 3 study conducted by Avillion under a collaborative development agreement with Pfizer
 Avillion Announces US Approval of Pfizer s BOSULIF (bosutinib) for the Treatment of Patients with Newly-Diagnosed Ph+ Chronic Myelogenous Leukemia (CML) Approval based on the successful BFORE Phase 3 study
Avillion Announces US Approval of Pfizer s BOSULIF (bosutinib) for the Treatment of Patients with Newly-Diagnosed Ph+ Chronic Myelogenous Leukemia (CML) Approval based on the successful BFORE Phase 3 study
BioArctic announces positive topline results of BAN2401 Phase 2b at 18 months in early Alzheimer s Disease
 Press release BioArctic announces positive topline results of BAN2401 Phase 2b at 18 months in early Alzheimer s Disease The full 18 month analysis of the 856 patient BAN2401 Phase 2b clinical study in
Press release BioArctic announces positive topline results of BAN2401 Phase 2b at 18 months in early Alzheimer s Disease The full 18 month analysis of the 856 patient BAN2401 Phase 2b clinical study in
Founded in 2016 to develop life-changing therapies against debilitating aggressive cancers that have limited treatment options
 Founded in 2016 to develop life-changing therapies against debilitating aggressive cancers that have limited treatment options Integrated platform technology for discovery of novel molecular targets and
Founded in 2016 to develop life-changing therapies against debilitating aggressive cancers that have limited treatment options Integrated platform technology for discovery of novel molecular targets and
Index. Surg Oncol Clin N Am 16 (2007) Note: Page numbers of article titles are in boldface type.
 Surg Oncol Clin N Am 16 (2007) 465 469 Index Note: Page numbers of article titles are in boldface type. A Adjuvant therapy, preoperative for gastric cancer, staging and, 339 B Breast cancer, metabolic
Surg Oncol Clin N Am 16 (2007) 465 469 Index Note: Page numbers of article titles are in boldface type. A Adjuvant therapy, preoperative for gastric cancer, staging and, 339 B Breast cancer, metabolic
NEWS RELEASE Media Contact: Megan Pace Investor Contact: Kathee Littrell Patient Inquiries: Ajanta Horan
 NEWS RELEASE Media Contact: Megan Pace 650-467-7334 Investor Contact: Kathee Littrell 650-225-1034 Patient Inquiries: Ajanta Horan 650-467-1741 GENENTECH RECEIVES COMPLETE RESPONSE LETTER FROM FDA FOR
NEWS RELEASE Media Contact: Megan Pace 650-467-7334 Investor Contact: Kathee Littrell 650-225-1034 Patient Inquiries: Ajanta Horan 650-467-1741 GENENTECH RECEIVES COMPLETE RESPONSE LETTER FROM FDA FOR
The Galectin-3 Inhibitor GR-MD-02 for Combination Cancer Immunotherapy
 The Galectin-3 Inhibitor GR-MD-02 for Combination Cancer Immunotherapy Supplemental Information to Corporate Presentation February 6, 2018 NASDAQ: GALT www.galectintherapeutics.com 2018 2017 Galectin Therapeutics
The Galectin-3 Inhibitor GR-MD-02 for Combination Cancer Immunotherapy Supplemental Information to Corporate Presentation February 6, 2018 NASDAQ: GALT www.galectintherapeutics.com 2018 2017 Galectin Therapeutics
Dicerna Pharmaceuticals Overview. Delivering RNAi-Based Breakthrough Therapies
 Pharmaceuticals Overview Delivering RNAi-Based Breakthrough Therapies Forward-Looking Statements This information may contain projections and other forward looking statements regarding future events, including
Pharmaceuticals Overview Delivering RNAi-Based Breakthrough Therapies Forward-Looking Statements This information may contain projections and other forward looking statements regarding future events, including
37 th ANNUAL JP MORGAN HEALTHCARE CONFERENCE
 37 th ANNUAL JP MORGAN HEALTHCARE CONFERENCE January 2019 Safe Harbor Statement This presentation contains forward-looking statements within the meaning of Section 27A of the Securities Act of 1933, as
37 th ANNUAL JP MORGAN HEALTHCARE CONFERENCE January 2019 Safe Harbor Statement This presentation contains forward-looking statements within the meaning of Section 27A of the Securities Act of 1933, as
Accelerate Your Research with Conversant Bio
 Imagination has given us the steam engine, the telephone, the talkingmachine and the automobile, for these things had to be dreamed of before they became realities. So I believe that dreams... are likely
Imagination has given us the steam engine, the telephone, the talkingmachine and the automobile, for these things had to be dreamed of before they became realities. So I believe that dreams... are likely
Dr. Bernard Huber CEO. Bio-Europe
 Dr. Bernard Huber CEO Bio-Europe 2015 1 ORYX Overview ORYX GmbH & Co. KG (ORYX) is a privately held company for translational oncology founded in 2007 and located in Baldham/Munich, Germany ORYX bridges
Dr. Bernard Huber CEO Bio-Europe 2015 1 ORYX Overview ORYX GmbH & Co. KG (ORYX) is a privately held company for translational oncology founded in 2007 and located in Baldham/Munich, Germany ORYX bridges
Novogen Limited. An emerging oncology drug developer with two clinical-stage programs NRT NVGN. April 2017
 Novogen Limited An emerging oncology drug developer with two clinical-stage programs NRT April 2017 NVGN Forward-Looking Statements This presentation contains forward-looking statements within the meaning
Novogen Limited An emerging oncology drug developer with two clinical-stage programs NRT April 2017 NVGN Forward-Looking Statements This presentation contains forward-looking statements within the meaning
APPENDIX "C" COMPANY DEVELOPMENT PIPELINE
 Horse Horse APPENDIX "C" COMPANY DEVELOPMENT PIPELINE Animal Cancer Vaccines AUTOLOGOUS Preclinical Approval ALLOGENEIC Preclinical Approval DIRECT INJECTION Preclinical Approval Horse Q4 2016 Human Cancer
Horse Horse APPENDIX "C" COMPANY DEVELOPMENT PIPELINE Animal Cancer Vaccines AUTOLOGOUS Preclinical Approval ALLOGENEIC Preclinical Approval DIRECT INJECTION Preclinical Approval Horse Q4 2016 Human Cancer
Bayer and Loxo Oncology to develop and commercialize two novel oncology therapies selectively targeting genetic drivers of cancer
 Investor News Not intended for U.S. and UK Media Bayer AG Investor Relations 51368 Leverkusen Germany www.investor.bayer.com Bayer and Loxo Oncology to develop and commercialize two novel oncology therapies
Investor News Not intended for U.S. and UK Media Bayer AG Investor Relations 51368 Leverkusen Germany www.investor.bayer.com Bayer and Loxo Oncology to develop and commercialize two novel oncology therapies
Curriculum Vitae Hui Yuan, MD
 Curriculum Vitae Hui Yuan, MD Current Position: Associate Professor Department of Saint Louis University Health Sciences Center Education: 1/1978 12/1982 M.D. in Medicine Postgraduate Training: 6/1984
Curriculum Vitae Hui Yuan, MD Current Position: Associate Professor Department of Saint Louis University Health Sciences Center Education: 1/1978 12/1982 M.D. in Medicine Postgraduate Training: 6/1984
Business Update & Financial Results for Q1 2018
 Business Update & Financial Results for Q1 2018 May 15, 2018 Disclaimer The statements made in this presentation may include forward-looking statements regarding the future operations of ERYTECH Pharma
Business Update & Financial Results for Q1 2018 May 15, 2018 Disclaimer The statements made in this presentation may include forward-looking statements regarding the future operations of ERYTECH Pharma
Inhibitors of Methionine Aminopeptidase-2 2 in the Treatment of Non-Hodgkin
 Inhibitors of Methionine Aminopeptidase-2 2 in the Treatment of Non-Hodgkin Hodgkin s s Lymphoma Targeted Cancer Therapies William Westlin, Ph.D. Vice President, Preclinical Research Discovery of Fumagillin
Inhibitors of Methionine Aminopeptidase-2 2 in the Treatment of Non-Hodgkin Hodgkin s s Lymphoma Targeted Cancer Therapies William Westlin, Ph.D. Vice President, Preclinical Research Discovery of Fumagillin
Claudia Adams Barr Program in Innovative Cancer Research Dana-Farber Cancer Institute BARR PROGRAM IMPACT STATEMENTS
 Claudia Adams Barr Program in Innovative Cancer Research Dana-Farber Cancer Institute BARR PROGRAM IMPACT STATEMENTS Brain Cancer New Treatment Opportunities - Discovery of new pathways in brain cancers
Claudia Adams Barr Program in Innovative Cancer Research Dana-Farber Cancer Institute BARR PROGRAM IMPACT STATEMENTS Brain Cancer New Treatment Opportunities - Discovery of new pathways in brain cancers
Metabolic Disease drug discovery at Evotec
 Metabolic Disease drug discovery at Evotec Evotec, Metabolic Disease Drug Discovery, 2016 Evotec, an ideal partner in metabolic disease drug discovery The different ways to work with us On your specific
Metabolic Disease drug discovery at Evotec Evotec, Metabolic Disease Drug Discovery, 2016 Evotec, an ideal partner in metabolic disease drug discovery The different ways to work with us On your specific
Best possible treatment from day 1. January 2019
 Best possible treatment from day 1 January 2019 1 Biovica develops and commercializes blood based assays for prognosis and monitoring of effect of targeted cancer treatments Our objective is to enable
Best possible treatment from day 1 January 2019 1 Biovica develops and commercializes blood based assays for prognosis and monitoring of effect of targeted cancer treatments Our objective is to enable
Corporate Update. NASDAQ: GALT April 9, 2018
 Corporate Update April 9, 2018 NASDAQ: GALT www.galectintherapeutics.com Forward-Looking Statements This presentation contains, in addition to historical information, forward-looking statements within
Corporate Update April 9, 2018 NASDAQ: GALT www.galectintherapeutics.com Forward-Looking Statements This presentation contains, in addition to historical information, forward-looking statements within
Parkinson s Research and the New Centre for Brain Health at UBC. Case for Support The University of British Columbia Faculty of Medicine
 Parkinson s Research and the New Centre for Brain Health at UBC Case for Support The University of British Columbia Faculty of Medicine THE VALUE OF A HEALTHY BRAIN By 2020, brain disease will overtake
Parkinson s Research and the New Centre for Brain Health at UBC Case for Support The University of British Columbia Faculty of Medicine THE VALUE OF A HEALTHY BRAIN By 2020, brain disease will overtake
Can we prevent metastasis?
 Can we prevent metastasis? A research example to translate from the bench to the bedside Diane Palmieri, Ph.D. Women s Cancers Section Laboratory of Molecular Pharmacology CCR, NCI Some Basic Truths Most
Can we prevent metastasis? A research example to translate from the bench to the bedside Diane Palmieri, Ph.D. Women s Cancers Section Laboratory of Molecular Pharmacology CCR, NCI Some Basic Truths Most
Immunocore and MedImmune announce new collaboration to conduct immuno-oncology combination trials in melanoma
 PRESS RELEASE IMMUNOCORE LIMITED Immunocore and MedImmune announce new collaboration to conduct immuno-oncology combination trials in melanoma (Oxford, UK, 16 April 2015) Immunocore Limited, a world-leading
PRESS RELEASE IMMUNOCORE LIMITED Immunocore and MedImmune announce new collaboration to conduct immuno-oncology combination trials in melanoma (Oxford, UK, 16 April 2015) Immunocore Limited, a world-leading
NEWSLETTER June Summer greetings from Follicum
 NEWSLETTER June 2018 Follicum hopes that this newsletter reaches as many of the company's owners and stakeholders as possible. If you wish to follow our continued development, please register your e-mail
NEWSLETTER June 2018 Follicum hopes that this newsletter reaches as many of the company's owners and stakeholders as possible. If you wish to follow our continued development, please register your e-mail
Partnering in Oncology Sharing a Vision to Help Prolong and Improve Patients Lives. Oncology Therapeutic Area Janssen Research & Development, LLC
 Partnering in Oncology Sharing a Vision to Help Prolong and Improve Patients Lives Oncology Therapeutic Area Janssen Research & Development, LLC The patients are waiting. Dr. Paul Janssen To Our Potential
Partnering in Oncology Sharing a Vision to Help Prolong and Improve Patients Lives Oncology Therapeutic Area Janssen Research & Development, LLC The patients are waiting. Dr. Paul Janssen To Our Potential
NKTR-255: Accessing The Immunotherapeutic Potential Of IL-15 for NK Cell Therapies
 NKTR-255: Accessing The Immunotherapeutic Potential Of IL-15 for NK Cell Therapies Saul Kivimäe Senior Scientist, Research Biology Nektar Therapeutics NK Cell-Based Cancer Immunotherapy, September 26-27,
NKTR-255: Accessing The Immunotherapeutic Potential Of IL-15 for NK Cell Therapies Saul Kivimäe Senior Scientist, Research Biology Nektar Therapeutics NK Cell-Based Cancer Immunotherapy, September 26-27,
A NOVEL CANCER STEM CELL (CSC) DRUG DISCOVERY PLATFORM. Collaborative Opportunity
 A NOVEL CANCER STEM CELL (CSC) DRUG DISCOVERY PLATFORM Collaborative Opportunity March 2014 PURPOSE Cancer Research UK funded Investigators have established in vitro and in vivo models of CSCs for use
A NOVEL CANCER STEM CELL (CSC) DRUG DISCOVERY PLATFORM Collaborative Opportunity March 2014 PURPOSE Cancer Research UK funded Investigators have established in vitro and in vivo models of CSCs for use
Juyoun Jin, D.V.M., Ph.D. Institute for Refractory Cancer Research, Samsung Medical Center
 Juyoun Jin, D.V.M., Ph.D. Institute for Refractory Cancer Research, Samsung Medical Center Overview of Anticancer Drug Development Discovery Non-clinical development Clinical Trial Target Identification
Juyoun Jin, D.V.M., Ph.D. Institute for Refractory Cancer Research, Samsung Medical Center Overview of Anticancer Drug Development Discovery Non-clinical development Clinical Trial Target Identification
Figure 1: PALLAS Study Schema. Endocrine adjuvant therapy may have started before randomization and be ongoing at that time.
 Figure 1: PALLAS Study Schema Endocrine adjuvant therapy may have started before randomization and be ongoing at that time. Approximately 4600 patients from approximately 500 global sites will be randomized
Figure 1: PALLAS Study Schema Endocrine adjuvant therapy may have started before randomization and be ongoing at that time. Approximately 4600 patients from approximately 500 global sites will be randomized
What EU research policy can do for conditions such as chronic pain?
 The EU Framework Programme for Research and Innovation HORIZON 2020 What EU research policy can do for conditions such as chronic pain? K Berkouk, PhD Deputy Head of Unit, Non-communicable diseases and
The EU Framework Programme for Research and Innovation HORIZON 2020 What EU research policy can do for conditions such as chronic pain? K Berkouk, PhD Deputy Head of Unit, Non-communicable diseases and
Understanding Tumor Markers for Breast and Colorectal Cancers. A Patient s Guide. Recommendations of the American Society of Clinical Oncology
 Cover Layout 1 A Patient s Guide Understanding Tumor Markers for Breast and Colorectal Cancers Recommendations of the American Society of Clinical Oncology ... Cover Layout 2 A Patient s Guide Understanding
Cover Layout 1 A Patient s Guide Understanding Tumor Markers for Breast and Colorectal Cancers Recommendations of the American Society of Clinical Oncology ... Cover Layout 2 A Patient s Guide Understanding
CAELUM BIOSCIENCES. Corporate Overview May, 2017
 CAELUM BIOSCIENCES Corporate Overview May, 2017 Forward Looking Statements Statements in this presentation that are not descriptions of historical facts are forward looking statements within the meaning
CAELUM BIOSCIENCES Corporate Overview May, 2017 Forward Looking Statements Statements in this presentation that are not descriptions of historical facts are forward looking statements within the meaning
Convergent and Divergent Mechanisms in Aging and Cancer
 Convergent and Divergent Mechanisms in Aging and Cancer Mariana S. De Lorenzo, PhD Department of Cell Biology & Molecular Medicine delorems@umdnj.edu LEARNING OBJECTIVES 1. To identify convergent and divergent
Convergent and Divergent Mechanisms in Aging and Cancer Mariana S. De Lorenzo, PhD Department of Cell Biology & Molecular Medicine delorems@umdnj.edu LEARNING OBJECTIVES 1. To identify convergent and divergent
IMMUNOMEDICS, INC. Advanced Antibody-Based Therapeutics
 IMMUNOMEDICS, INC. Advanced Antibody-Based Therapeutics Oncology Autoimmune Diseases Rodman & Renshaw 19 th Annual Global Investment Conference Michael R. Garone, Principal Executive Officer and CFO Forward-Looking
IMMUNOMEDICS, INC. Advanced Antibody-Based Therapeutics Oncology Autoimmune Diseases Rodman & Renshaw 19 th Annual Global Investment Conference Michael R. Garone, Principal Executive Officer and CFO Forward-Looking
(+1) Matti Ojanen (+44) Investor Contact: Jennifer M. Davis (+1)
 For immediate release: October 24, 2012 Media Contact: Victoria Davis (+1) 347-558-3455 Matti Ojanen (+44) 7557-202-394 Investor Contact: Jennifer M. Davis (+1) 212-733-0717 Pfizer s XALKORI Receives Conditional
For immediate release: October 24, 2012 Media Contact: Victoria Davis (+1) 347-558-3455 Matti Ojanen (+44) 7557-202-394 Investor Contact: Jennifer M. Davis (+1) 212-733-0717 Pfizer s XALKORI Receives Conditional
(212) Investors Contact: Ryan Crowe (212)
 For immediate release: February 5, 2014 Media Contact: Sally Beatty (212) 733-6566 Investors Contact: Ryan Crowe (212) 733-8160 Pfizer And Merck To Collaborate On Innovative Anti-Cancer Combination Studies
For immediate release: February 5, 2014 Media Contact: Sally Beatty (212) 733-6566 Investors Contact: Ryan Crowe (212) 733-8160 Pfizer And Merck To Collaborate On Innovative Anti-Cancer Combination Studies
2017 ANNUAL RESULTS AND UPDATE ON TANGO
 Press release 2017 ANNUAL RESULTS AND UPDATE ON TANGO Solid cash position of 16.3 million at December 31, 2017 TANGO timing update: results postponed, full 12 month results expected in Q4 2018 per protocol
Press release 2017 ANNUAL RESULTS AND UPDATE ON TANGO Solid cash position of 16.3 million at December 31, 2017 TANGO timing update: results postponed, full 12 month results expected in Q4 2018 per protocol
Team Progress Updates. SU2C Pancreatic Dream Team: Cutting Off the Fuel Supply: A New Approach in the Treatment of Pancreatic Cancer
 SU2C Pancreatic Dream Team: Cutting Off the Fuel Supply: A New Approach in the Treatment of Pancreatic Cancer Cancer of the pancreas is one of the deadliest forms of cancer, with a five-year survival rate
SU2C Pancreatic Dream Team: Cutting Off the Fuel Supply: A New Approach in the Treatment of Pancreatic Cancer Cancer of the pancreas is one of the deadliest forms of cancer, with a five-year survival rate
Introducing A New Platform In Cancer Care Robotically Assisted Sonic Therapy (RAST)
 Introducing A New Platform In Cancer Care Robotically Assisted Sonic Therapy (RAST) CED Life Science Conference February 27, 2018 Almost 40% of us will be diagnosed with cancer at some point in our lives
Introducing A New Platform In Cancer Care Robotically Assisted Sonic Therapy (RAST) CED Life Science Conference February 27, 2018 Almost 40% of us will be diagnosed with cancer at some point in our lives
Antitumor Activity of CUDC-305, a Novel Oral HSP90 Inhibitor, in Solid and Hematological Tumor Xenograft Models
 Antitumor Activity of CUDC-5, a Novel Oral HSP Inhibitor, in Solid and Hematological Tumor Xenograft Models Rudi Bao, MD/PhD April 1, 2 AACR 1th Annual Meeting 2 Experimental and Molecular Therapeutics
Antitumor Activity of CUDC-5, a Novel Oral HSP Inhibitor, in Solid and Hematological Tumor Xenograft Models Rudi Bao, MD/PhD April 1, 2 AACR 1th Annual Meeting 2 Experimental and Molecular Therapeutics
Conflict of Interest Statement
 Specific Aspects and Approaches for Regulatory Evaluation of Pharmaceuticals in Two-Year Rodent Carcinogenicity Studies James A. Popp Stratoxon LLC Morgantown, PA Tel: 610.286.7592 popp@stratoxon.com Conflict
Specific Aspects and Approaches for Regulatory Evaluation of Pharmaceuticals in Two-Year Rodent Carcinogenicity Studies James A. Popp Stratoxon LLC Morgantown, PA Tel: 610.286.7592 popp@stratoxon.com Conflict
PHASE 1 CLINICAL TRIALS: DESIGNS AND CONSIDERATIONS. Sarit Assouline, MD, MSc, FRCPC Associate professor, McGill University Jewish General Hospital
 PHASE 1 CLINICAL TRIALS: DESIGNS AND CONSIDERATIONS Sarit Assouline, MD, MSc, FRCPC Associate professor, McGill University Jewish General Hospital Outline Introduction Goal of Phase I studies in oncology
PHASE 1 CLINICAL TRIALS: DESIGNS AND CONSIDERATIONS Sarit Assouline, MD, MSc, FRCPC Associate professor, McGill University Jewish General Hospital Outline Introduction Goal of Phase I studies in oncology
About OMICS International
 About OMICS International OMICS International through its Open Access Initiative is committed to make genuine and reliable contributions to the scientific community. OMICS International hosts over 700
About OMICS International OMICS International through its Open Access Initiative is committed to make genuine and reliable contributions to the scientific community. OMICS International hosts over 700
BRANCHED-CHAIN AMINO ACIDS FOUND TO REGULATE THE DEVELOPMENT AND PROGRESSION OF CANCER
 SCIENTIFIC ADVISORY BRANCHED-CHAIN AMINO ACIDS FOUND TO REGULATE THE DEVELOPMENT AND PROGRESSION OF CANCER Researchers from A*STAR s SBIC have discovered how changes in BCAA metabolism influence the development
SCIENTIFIC ADVISORY BRANCHED-CHAIN AMINO ACIDS FOUND TO REGULATE THE DEVELOPMENT AND PROGRESSION OF CANCER Researchers from A*STAR s SBIC have discovered how changes in BCAA metabolism influence the development
Oncology Program. Chlorotoxin Technology Platform. Corporate Partnering Opportunities
 TransMolecular, Inc. A Neuroscience Biotechnology Company Oncology Program Chlorotoxin Technology Platform Corporate Partnering Opportunities Background TransMolecular, Inc. (TMI) is a neuroscience biotechnology
TransMolecular, Inc. A Neuroscience Biotechnology Company Oncology Program Chlorotoxin Technology Platform Corporate Partnering Opportunities Background TransMolecular, Inc. (TMI) is a neuroscience biotechnology
PPD S EXPERT HEMATOLOGY AND ONCOLOGY TEAM
 HEMATOLOGY ONCOLOGY PPD S EXPERT HEMATOLOGY AND ONCOLOGY TEAM COMMITTED TO ADVANCING DRUG DEVELOPMENT IN ONCOLOGY $ MARKETPLACE COMPLEXITIES increasingly competitive marketplace and rising cost pressures
HEMATOLOGY ONCOLOGY PPD S EXPERT HEMATOLOGY AND ONCOLOGY TEAM COMMITTED TO ADVANCING DRUG DEVELOPMENT IN ONCOLOGY $ MARKETPLACE COMPLEXITIES increasingly competitive marketplace and rising cost pressures
ModiQuant Technology Innovative assessment of modified proteins. Massimo Papale CEO & Co-Founder
 ModiQuant Technology Innovative assessment of modified proteins Massimo Papale CEO & Co-Founder Our Milestones January 2016 Patent application for R/pR test June 2016 Keep Calm & Startup Foggia Prize October
ModiQuant Technology Innovative assessment of modified proteins Massimo Papale CEO & Co-Founder Our Milestones January 2016 Patent application for R/pR test June 2016 Keep Calm & Startup Foggia Prize October
A company developing innovative, high-impact drugs for cancer
 A company developing innovative, high-impact drugs for cancer Investor Briefing March 2019 ASX: KZA NASDAQ : KZIA Twitter: @KaziaTx Forward-Looking Statements This presentation contains forward-looking
A company developing innovative, high-impact drugs for cancer Investor Briefing March 2019 ASX: KZA NASDAQ : KZIA Twitter: @KaziaTx Forward-Looking Statements This presentation contains forward-looking
Molecular Imaging and Cancer
 Molecular Imaging and Cancer Cancer causes one in every four deaths in the United States, second only to heart disease. According to the U.S. Department of Health and Human Services, more than 512,000
Molecular Imaging and Cancer Cancer causes one in every four deaths in the United States, second only to heart disease. According to the U.S. Department of Health and Human Services, more than 512,000
Phoenix Molecular Designs
 Phoenix Molecular Designs DESIGNING PRECISE THERAPEUTICS TO REVOLUTIONIZE TREATMENT Our Team Dr. Sandra Dunn, Ph.D. Dr. Anna Stratford, Ph.D. Dr. Aarthi Jayanthan, Ph.D. Dr. Zaihui Zhang CEO, CSO UBC Professor
Phoenix Molecular Designs DESIGNING PRECISE THERAPEUTICS TO REVOLUTIONIZE TREATMENT Our Team Dr. Sandra Dunn, Ph.D. Dr. Anna Stratford, Ph.D. Dr. Aarthi Jayanthan, Ph.D. Dr. Zaihui Zhang CEO, CSO UBC Professor
The Current Status of Immune Checkpoint Inhibitors: Arvin Yang, MD PhD Oncology Global Clinical Research Bristol-Myers Squibb
 The Current Status of Immune Checkpoint Inhibitors: A Global Overview of the Field Arvin Yang, MD PhD Oncology Global Clinical Research Bristol-Myers Squibb Immune Checkpoint Inhibitors Conference, March
The Current Status of Immune Checkpoint Inhibitors: A Global Overview of the Field Arvin Yang, MD PhD Oncology Global Clinical Research Bristol-Myers Squibb Immune Checkpoint Inhibitors Conference, March
PRESENTATION. AGM 27 th OCTOBER 2015 ASX: PAA ACN
 PRESENTATION AGM 27 th OCTOBER 2015 ASX: PAA ACN 094 006 023 Disclaimer This presentation does not constitute investment advice. Neither this presentation nor the information contained in it constitutes
PRESENTATION AGM 27 th OCTOBER 2015 ASX: PAA ACN 094 006 023 Disclaimer This presentation does not constitute investment advice. Neither this presentation nor the information contained in it constitutes
CML TREATMENT GUIDELINES
 CML TREATMENT GUIDELINES INITIAL INVESTIGATION Propose enrolment in the CML Registry of the CML-MPN Quebec Research Group. Medical history : Question for cardio-respiratory disorders, diabetes, pancreatitis,
CML TREATMENT GUIDELINES INITIAL INVESTIGATION Propose enrolment in the CML Registry of the CML-MPN Quebec Research Group. Medical history : Question for cardio-respiratory disorders, diabetes, pancreatitis,
NKTR-255: Accessing IL-15 Therapeutic Potential through Robust and Sustained Engagement of Innate and Adaptive Immunity
 NKTR-255: Accessing IL-15 Therapeutic Potential through Robust and Sustained Engagement of Innate and Adaptive Immunity Peiwen Kuo Scientist, Research Biology Nektar Therapeutics August 31 st, 2018 Emerging
NKTR-255: Accessing IL-15 Therapeutic Potential through Robust and Sustained Engagement of Innate and Adaptive Immunity Peiwen Kuo Scientist, Research Biology Nektar Therapeutics August 31 st, 2018 Emerging
Revolutionizing the Treatment of Cancer
 Revolutionizing the Treatment of Cancer September 2013 Safe Harbor Statement The statements that follow (including projections and business trends) are forward looking statements. Rexahn's actual results
Revolutionizing the Treatment of Cancer September 2013 Safe Harbor Statement The statements that follow (including projections and business trends) are forward looking statements. Rexahn's actual results
A263 A352 A204. Pan CK. pstat STAT3 pstat3 STAT3 pstat3. Columns Columns 1-6 Positive control. Omentum. Rectosigmoid A195.
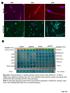 pstat3 75 Pan CK A A263 A352 A24 B Columns 1-6 Positive control A195 A22 A24 A183 Rectal Nodule STAT3 pstat3 STAT3 pstat3 Columns 7-12 Omentum Rectosigmoid Left Ovary Right Ovary Omentum Uterus Uterus
pstat3 75 Pan CK A A263 A352 A24 B Columns 1-6 Positive control A195 A22 A24 A183 Rectal Nodule STAT3 pstat3 STAT3 pstat3 Columns 7-12 Omentum Rectosigmoid Left Ovary Right Ovary Omentum Uterus Uterus
A NEW FRONTIER IN IMMUNO-ONCOLOGY
 A NEW FRONTIER IN IMMUNO-ONCOLOGY Proactive One2One Forum London, March 8 th 2018 Dr Richard Goodfellow LSE: SCLP.L CLINICAL STAGE IMMUNO-ONCOLOGY COMPANY Scancell is developing innovative immunotherapies
A NEW FRONTIER IN IMMUNO-ONCOLOGY Proactive One2One Forum London, March 8 th 2018 Dr Richard Goodfellow LSE: SCLP.L CLINICAL STAGE IMMUNO-ONCOLOGY COMPANY Scancell is developing innovative immunotherapies
(your) future starts now
 -- (your) future starts now PhD in oncology www.phd-in-oncology.com the best PhD students need the best center The course of excellence in oncology Fondation Philanthropia / 2 nd graduation class With
-- (your) future starts now PhD in oncology www.phd-in-oncology.com the best PhD students need the best center The course of excellence in oncology Fondation Philanthropia / 2 nd graduation class With
performed to help sway the clinician in what the appropriate diagnosis is, which can substantially alter the treatment of management.
 Hello, I am Maura Polansky at the University of Texas MD Anderson Cancer Center. I am a Physician Assistant in the Department of Gastrointestinal Medical Oncology and the Program Director for Physician
Hello, I am Maura Polansky at the University of Texas MD Anderson Cancer Center. I am a Physician Assistant in the Department of Gastrointestinal Medical Oncology and the Program Director for Physician
Corporate Presentation October 2018 Nasdaq: ADXS
 Innovations in Immuno-Oncology Corporate Presentation October 2018 Nasdaq: ADXS Forward-Looking Statements This presentation contains forward-looking statements, including, but not limited to, statements
Innovations in Immuno-Oncology Corporate Presentation October 2018 Nasdaq: ADXS Forward-Looking Statements This presentation contains forward-looking statements, including, but not limited to, statements
Since the year 2000 a significant amount of investment has been made in the private healthcare sector in Turkey.
 Background Since the year 2000 a significant amount of investment has been made in the private healthcare sector in Turkey. In the year 2000, nearly 70% of the healthcare investments were made solely in
Background Since the year 2000 a significant amount of investment has been made in the private healthcare sector in Turkey. In the year 2000, nearly 70% of the healthcare investments were made solely in
Pioneering vaccines that transform lives.
 Pioneering vaccines that transform lives. Immunomic Therapeutics, Inc. LAMP-Vax for General Oncology Presentation Outline 1. Immunomic Therapeutics: corporate overview 2. Unmet need in cancer immunotherapy
Pioneering vaccines that transform lives. Immunomic Therapeutics, Inc. LAMP-Vax for General Oncology Presentation Outline 1. Immunomic Therapeutics: corporate overview 2. Unmet need in cancer immunotherapy
SUBSTANCE USE DISORDERS
 THE POWER OFx SUBSTANCE USE DISORDERS Experts. Experience. Execution. Deep Dive: Substance Use Disorder Clinical Research Accelerate your next substance use disorder study with Medpace s noted medical
THE POWER OFx SUBSTANCE USE DISORDERS Experts. Experience. Execution. Deep Dive: Substance Use Disorder Clinical Research Accelerate your next substance use disorder study with Medpace s noted medical
CERTIFICATE OF ACCREDITATION
 CERTIFICATE OF ACCREDITATION In terms of section 22(2) (b) of the Accreditation for Conformity Assessment, Calibration and Good Laboratory Practice Act, 2006 (Act 19 of 2006), read with sections 23(1),
CERTIFICATE OF ACCREDITATION In terms of section 22(2) (b) of the Accreditation for Conformity Assessment, Calibration and Good Laboratory Practice Act, 2006 (Act 19 of 2006), read with sections 23(1),
DISCLOSURES. This activity is jointly provided by Northwest Portland Area Indian Health Board and Cardea
 DISCLOSURES This activity is jointly provided by Northwest Portland Area Indian Health Board and Cardea Cardea Services is approved as a provider of continuing nursing education by Montana Nurses Association,
DISCLOSURES This activity is jointly provided by Northwest Portland Area Indian Health Board and Cardea Cardea Services is approved as a provider of continuing nursing education by Montana Nurses Association,
REDECTANE Preliminary data of the Phase III trial 30 November 2009
 REDECTANE Preliminary data of the Phase III trial 30 November 2009 Forward looking statements This communication contains certain forward-looking statements, relating to the Company s business, which can
REDECTANE Preliminary data of the Phase III trial 30 November 2009 Forward looking statements This communication contains certain forward-looking statements, relating to the Company s business, which can
Supplementary Figure 1. Deletion of Smad3 prevents B16F10 melanoma invasion and metastasis in a mouse s.c. tumor model.
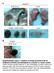 A B16F1 s.c. Lung LN Distant lymph nodes Colon B B16F1 s.c. Supplementary Figure 1. Deletion of Smad3 prevents B16F1 melanoma invasion and metastasis in a mouse s.c. tumor model. Highly invasive growth
A B16F1 s.c. Lung LN Distant lymph nodes Colon B B16F1 s.c. Supplementary Figure 1. Deletion of Smad3 prevents B16F1 melanoma invasion and metastasis in a mouse s.c. tumor model. Highly invasive growth
JP Morgan Healthcare Conference
 JP Morgan Healthcare Conference Lamberto Andreotti Chief Executive Officer January 14, 2014 Forward-Looking Information During this meeting, we will make statements about the Company s future plans and
JP Morgan Healthcare Conference Lamberto Andreotti Chief Executive Officer January 14, 2014 Forward-Looking Information During this meeting, we will make statements about the Company s future plans and
Lung Cancer Screening
 Lung Cancer Screening v Lung Cancer: America s leading cancer killer Annual US cancer mortality 158,040 221,000 new diagnoses in US 4,100 14,180 27,540 Lung 40,560 40,730 49,700 Cervix Ovary Prostate Pancreas
Lung Cancer Screening v Lung Cancer: America s leading cancer killer Annual US cancer mortality 158,040 221,000 new diagnoses in US 4,100 14,180 27,540 Lung 40,560 40,730 49,700 Cervix Ovary Prostate Pancreas
Oragenics Shareholder Update
 January 4, 2017 Oragenics Shareholder Update Advances Drug Development Programs Focused on Conditions with Significant Unmet Medical Needs TAMPA, Fla.--(BUSINESS WIRE)-- Oragenics, Inc. (NYSE MKT:OGEN.BC),
January 4, 2017 Oragenics Shareholder Update Advances Drug Development Programs Focused on Conditions with Significant Unmet Medical Needs TAMPA, Fla.--(BUSINESS WIRE)-- Oragenics, Inc. (NYSE MKT:OGEN.BC),
