Von Willebrand factor (VWF) is a multimeric plasma sialoglycoprotein
|
|
|
- Nigel Harper
- 5 years ago
- Views:
Transcription
1 Sialyltransferase ST3Gal-IV operates as a dominant modifier of hemostasis by concealing asialoglycoprotein receptor ligands Lesley G. Ellies*, David Ditto, Gallia G. Levy, Mark Wahrenbrock, David Ginsburg, Ajit Varki **, Dzung T. Le **, and Jamey D. Marth* ** *Howard Hughes Medical Institute, **Glycobiology Research and Training Center, and Departments of Cellular and Molecular Medicine, Medicine, and Pathology, 9500 Gilman Drive 0625, University of California San Diego, La Jolla, CA 92093; and Howard Hughes Medical Institute, Departments of Internal Medicine and Human Genetics, University of Michigan Medical School, Ann Arbor, MI Edited by Stuart A. Kornfeld, Washington University School of Medicine, St. Louis, MO, and approved May 6, 2002 (received for review January 4, 2002) A number of poorly characterized genetic modifiers contribute to the extensive variability of von Willebrand disease, the most prevalent bleeding disorder in humans. We find that a genetic lesion inactivating the murine ST3Gal-IV sialyltransferase causes a bleeding disorder associated with an autosomal dominant reduction in plasma von Willebrand factor (VWF) and an autosomal recessive thrombocytopenia. Although both ST3Gal-IV and ST6Gal-I sialyltransferases mask galactose linkages implicated as asialoglycoprotein receptor ligands, only ST3Gal-IV deficiency promotes asialoglycoprotein clearance mechanisms with a reduction in plasma levels of VWF and platelets. Exposed galactose on VWF was also found in a subpopulation of humans with abnormally low VWF levels. Oligosaccharide branch-specific sialylation by the ST3Gal-IV sialyltransferase is required to sustain the physiologic half-life of murine hemostatic components and may be an important modifier of plasma VWF level in humans. Von Willebrand factor (VWF) is a multimeric plasma sialoglycoprotein that stabilizes coagulation factor VIII and plays an essential role in hemostasis by mediating platelet aggregation to subendothelium at sites of vascular injury (reviewed in refs. 1 and 2). VWF levels in plasma vary widely among the human population, with particularly low VWF levels or dysfunctional VWF resulting in von Willebrand disease (VWD). VWD is the most common inherited bleeding disorder in humans. Inheritance is typically autosomal dominant with incomplete penetrance and highly variable expressivity. There are also a number of distinct variants of the disease, each with a specific qualitative functional defect, leading to a complex classification scheme (reviewed in ref. 3). A large number of mutations within the VWF gene have been identified in VWD individuals, particularly in patients with qualitative variants of VWD; however, the genetic basis for the wide variation in plasma VWF levels among both normal and VWD individuals remains largely unknown. VWF function appears conserved among mammalian species, indicating that animal models may be useful in identifying the molecular mechanisms and underlying genetic factors modifying VWF homeostasis and the severity of VWD (reviewed in ref. 4). The glycosylation of VWF comprises a posttranslational mechanism capable of modulating VWF stability and function in circulation. Enzymatic elimination of multiple glycan structures, including sialic acid linkages, has been shown to alter VWF proteolytic degradation, platelet aggregation function, multimerization, and half-life in plasma (5 10). These findings suggest the possibility that inherited genetic lesions in the pathways of glycan synthesis could modify the severity of VWD and may be directly responsible for VWD in some cases. However, such lesions would likely be less severe in altering glycan structure, otherwise viability could be affected in mammalian gestation and postnatal development (11). Some of the variance in normal plasma VWF levels has been associated with the ABO blood group determinants, because humans with blood type O due to homozygosity for null alleles in the H-locus glycosyltransferase generally have reduced VWF levels (12, 13). The remarkable finding of an inherited genetic mutation altering cell typespecific expression of the Galgt2 glycosyltransferase in the RIIIS J mouse, which resulted in an autosomal dominant VWF deficiency, revealed that enzymes in the glycosylation pathway could also play a dominant role in VWF homeostasis (14). Sialic acids are negatively charged molecules existing in either 2 3, -6, or -8 linkages on various glycan types and are generated by a family of 18 sialyltransferase genes differentially expressed among tissues (15 18). Sialic acids are most commonly linked to the penultimate galactose (Gal) or N-acetylgalactosamine (GalNAc) on glycan branches and can be essential in the formation of ligands for endogenous sialic acid-specific lectins such as the Siglecs (reviewed in ref. 19), the Selectins (reviewed in ref. 20), or pathogen receptors (21). However, they can also mask ligands involving Gal or GalNAc recognition by the galectins and asialoglycoprotein receptors (ASGPRs) (reviewed in refs. 22 and 23). The majority of sialic acids attached to plasma components are 2 3 linked and produced by up to six different ST3Gal genes encoding sialyltransferases ST3Gal-I VI. Only the ST6Gal-I sialyltransferase has been implicated in hemostatic regulation, by producing 2 6 sialic acid linkages that can block recognition of VWF by ASGPRs (9). However, ST6Gal-I-deficient mice exhibit a B lymphocyte defect but otherwise appear normal (24). Studies herein of mice bearing distinct sialyltransferase lesions identify ST3Gal-IV as a modulator of hematologic components and provide mechanistic insights regarding sialic acid linkage-specific function in hemostasis. Methods Gene Targeting and Mutant Mouse Production. Mouse ST3Gal-IV genomic DNA was isolated for use in gene-targeted mutagenesis procedures as previously described (25). In screening for homologous recombinants, the wild-type allele was detected by using PCR primers adjacent to the deleted region (W5 : 5 -GACGCCATC- CACCTATGAG and W3 : 5 -GGCTGCTCCCATTCCACT-3 ), resulting in a 260-bp fragment. The mutant allele was detected by using W5 and a primer from the loxp region (M3 : 5 -GGCTCTTTGTGGGACCATCAG-3 ), yielding a 450-bp fragment. RNA Analysis. Total RNA from indicated tissues was isolated and analyzed with a mouse ST3Gal-IV cdna probe containing the entire protein-coding sequence, by procedures described (25). This paper was submitted directly (Track II) to the PNAS office. Abbreviations: VWF, von Willebrand factor; VWD, von Willebrand disease; ASGP, asialoglycoprotein; ASGPR, ASGP receptor; Gal, galactose. Present address: University of California at San Diego Cancer Center, 9500 Gilman Drive, La Jolla, CA To whom reprint requests should be addressed. jmarth@ucsd.edu PNAS July 23, 2002 vol. 99 no cgi doi pnas
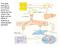
2 Hematology. Blood from the tail vein of methoxyfluoraneanethesized mice was collected into EDTA-containing polypropylene microtubes (Becton Dickinson). Analyses were carried out with a CELL-DYN 3500 (Abbott Labs, Abbott Park, IL) calibrated with normal mouse blood and microscopic examination with Wright Giemsa stain. For clotting assays, plasma samples were prepared from whole blood collected by cardiac puncture in one-tenth of volume buffered citrate anticoagulant (0.06 mol liter of sodium citrate 0.04 mol liter of citric acid, ph 7.4). Bleeding Time. Mice were anesthetized and restricted horizontally. The tail was severed 2 mm from the tip with a razor blade and immersed vertically 1 cm below the surface of 37 C saline. Time until bleeding stopped was recorded. The tail was cauterized when bleeding times exceeded 10 min. Coagulation Factor Analyses. VWF levels were measured by an Elisa approach by using the anti-vwf antibody (DAKO), as previously described (14). Factor VIII and other coagulation factors were assayed as described (26). Multimer Analysis. Mouse plasma samples were diluted 1:25 and separated by SDS-agarose electrophoresis in standard SDS PAGE running buffer by using a miniprotean II apparatus (BioRad). Gel and loading buffer conditions were as described (27). Gels were blotted onto Immobilon P membranes (Millipore) by capillary transfer overnight in PBS. Multimers were detected by incubating the membrane with horseradish peroxidase labeled anti-human VWF antibody (DAKO) at a 1:500 dilution and visualized by enhanced chemiluminescence (Amersham Pharmacia). Lectin Binding. Plates coated with anti-vwf antibodies (DAKO) and blocked with BSA were prepared. Plasma was added at 1:8 to 1:100 dilution. After washing, biotinylated Ricinus communis agglutinin-1 (RCA-I), Erythrina cristagalli (ECA), Sambucus nigra (SNA), or peanut agglutinin (PNA) lectins (Vector Laboratories) at 1 g ml was added. For VWF antigen determination in parallel anti-vwf, horseradish peroxidase-conjugated antibodies were added. Lectin binding was detected by using an avidin and biotinylated-horseradish peroxidase (ABC) detection system and developed with 3,3,5,5 tetramethylbenzidine (TMB, Bio-Rad). Binding was analyzed at 655 nm on a spectrophotometric plate reader (Molecular Devices) with SOFTMAX software. For VWF desialylation and lectin-binding analysis, whole plasma pooled from more than 20 wild-type mice was treated with 0.3 units Arthrobacter ureafaciens neuraminidase (Sigma) in the supplied buffer, ph 6.0, at 37 C for 90 min. Serial dilutions of 100 l were analyzed as above. VWF Clearance Measurements. Blood was obtained by cardiac puncture into citrate from three to six wild-type or ST3Gal-IV / mice. Plasma proteins were biotinylated in fresh EDTAanticoagulated plasma by using biotin-n-hydroxylsuccinimide (Calbiochem), followed by i.v. injection of 200 l per mouse. Blood was collected from the tail vein at 0, 2, 4, and 6 h. VWF in these fractions was captured on anti-vwf antibody-coated plates, and biotinylated VWF was detected by using ABC TMB as above. In experiments to detect ASGPR involvement, plasma obtained from ST3Gal-IV / mice was similarly injected with or without tissue-culture grade pyrogen-free fetuin preparations. Asialofetuin was prepared by mild acid hydrolysis as described (28). All fetuin preparations were tested for potential pyrogenic activity and contamination by a modified version of a blood mononuclear cell IL-6 secretion assay (29). Fig. 1. ST3Gal-IV mutation in the germline of mice ablates the large sialyl motif and results in loss of RNA. (A) An ST3Gal-IV genomic isolate in germline configuration was used in conjunction with the pflox vector to construct a targeting vector in which exons 5 7 containing the large sialyl motif (16) were flanked by loxp sites for deletion (ST3Gal-IV F[tkneo] ). Restriction enzyme sites indicated are BamHI (B) AvrII (A), EcoRI (E), Hind III (H), KpnI (K), NotI (N), SalI (Sa), and SpeI (Sp). (B) Transient Cre expression in ST3Gal-IV gene-targeted R1 embryonic stem cells produces subclones heterozygous for the (systemic deleted) or F (conditional) mutations illustrated. (C) Southern blot analysis of AvrII- and SpeI-digested embryonic stem cell DNA probed with a loxp probe confirmed the expected genomic structures. Three loxp sites are present in a targeted parental clone (21 6), whereas one loxp site is present in each of two ST3Gal-IV /wt subclones (21-F1 and 21-D1), and two loxp sites are present in the ST3Gal-IV F/wt subclones (21-A3 and 21-E1). (D) Tail DNA derived from offspring of parents heterozygous for the allele was digested with HindIII and probed with the indicated genomic probe to reveal the 6.8-kb wild-type allele and the 5.3-kb deleted ( ) allele. (E) ST3Gal-IV RNA expression in normal wild-type mouse tissues. (F) ST3Gal-IV RNA expression is deficient among ST3Gal-IV / mice. Platelet Cytometry and Clearance. Whole blood from the tail vein was collected in EDTA microtubes and diluted in Tyrode s buffer. Platelets were stained with anti-cd41 FITC (Becton Dickinson) and RCA-I biotin (Vector Laboratories) followed by streptavidincytochrome (Becton Dickinson). Data from 10,000 platelet events determined by anti-cd41 binding as well as forward- and sidescatter were analyzed on a FACScan flow cytometer by using CELLQUEST software (Becton Dickinson). In clearance studies, ST3Gal-IV / mice were injected i.p. with 10 mg of fetuin or asialofetuin at 0, 4, and 8 h. Blood was collected from the tail vein at 0, 4, 8, and 12 h into EDTA tubes and analyzed for platelet levels as above. Statistical Analysis. Data were analyzed by ANOVA and Scheffé s t test for unpaired samples. Results ST3Gal-IV Sialyltransferase Structure and Mutagenesis. The murine ST3Gal-IV sialyltransferase is a type II transmembrane protein localized to the Golgi apparatus and encoded by a gene spanning 10 kb containing 10 exons (16). A genomic clone encompassing the first 9 exons was isolated and characterized for producing a mutant allele in embryonic stems cells by Cre-loxP gene-targeting approaches (Fig. 1A). All sialyltransferases share structural features MEDICAL SCIENCES Ellies et al. PNAS July 23, 2002 vol. 99 no
3 Fig. 2. Increased bleeding time with decreased levels of VWF and Factor VIII occurs with ST3Gal-IV deficiency but not with ST6Gal-I deficiency. (A) Bleeding times were analyzed among 6- to 12-wk-old littermates of the indicated genotypes. Results are from over 30 mice of each ST3Gal-IV genotype and 11 of each ST6Gal-I genotype are expressed as means SEM. Significantly increased bleeding times for ST3Gal-IV heterozygotes and homozygotes were observed (P 0.05 and P 0.001, respectively). (B) VWF levels in plasma were measured from over 30 mice of each genotype and plotted as a percentage of the mean value obtained from ST3Gal-IV wild-type littermates. A significant reduction in plasma VWF was observed in both heterozygous and homozygous ST3Gal-IV-deficient mice to a mean of 57 and 50%, respectively (P 0.001). No decrease in plasma VWF levels was observed in mice lacking ST6Gal-I. (C) Factor VIII (F VIII) levels were also reduced in mice analyzed in B (P 0.001). ST6Gal-I deficiency had no effect on F VIII levels in circulation (not shown). (D) Levels of blood coagulation factors were analyzed from 40 mice of indicated genotypes. No alterations of hemostatic significance were observed. that include the large sialyl motif shown to be essential for nucleotide sugar binding and enzymatic activity (30); therefore, we mutagenized ST3Gal-IV by flanking exons 5 7 containing the large sialyl motif with loxp sites (F allele), resulting in the deletion of these sequences on Cre recombination ( allele) (Fig. 1B) and a translational frameshift within exon 8 (data not shown). Targeted embryonic stem cells bearing the expected allelic structures (Fig. 1C) were used to generate mice as described (25). Mice homozygous for the allele were produced in Mendelian ratios among heterozygous matings with both male and female mice fully fertile and appearing normal (Fig. 1D and data not shown). ST3Gal-IV RNA expression is widespread among tissues and cell types, with highest levels in the small intestine and colon (Fig. 1E). In mice homozygous for the allele, ST3Gal-IV RNA levels were below detection by Northern blot analysis, indicating that the deletion of genomic sequences including exons 5 7 destabilizes ST3Gal-IV RNA (Fig. 1F). The ST3Gal-IV allele was bred into the C57BL 6 strain for more than five generations for the following studies. An Autosomal Dominant Bleeding Disorder Associated with Reduced VWF and Factor VIII. A determination of bleeding time after tail transection indicated a significant increase among mice bearing the ST3Gal-IV allele (Fig. 2A). In contrast, mice lacking the ST6Gal-I sialyltransferase (24) exhibited normal bleeding times (Fig. 2A). Measurements of blood coagulation factor levels revealed a deficiency in plasma VWF in mice heterozygous and homozygous for the ST3Gal-IV allele (Fig. 2B). VWF mrna expression among various tissues was unaltered compared with wild-type littermates (data not shown). Fig. 3. VWF glycosylation deficiencies and multimer formation. Alterations in lectin binding are compared with results obtained by using purified sialidase treatment (*; see Methods). (A) RCA-I lectin binding to plasma-derived VWF from wild-type and ST3Gal-IV mutant mice indicates increased exposure of terminal -linked Gal on VWF from both heterozygous and homozygous mutant samples. Reduced Ricin binding to VWF from ST6Gal-I-deficient mice was observed. (B) ECA-I lectin binding to Gal 1 4GlcNAc- termini on VWF is increased in mice heterozygous or homozygous for ST3Gal-IV gene mutation. Absence of ST6Gal-I also results as expected in increased ECA binding to VWF. (C) SNA lectin binding to VWF is eliminated in ST6Gal-I deficiency, as expected, but increased in ST3Gal-IV deficiency. (D) PNA lectin binding to Gal 1 3GalNAc- is unaltered on VWF glycans from ST3Gal-IV mutant mice. (E) Possible N-glycan branch termini structures of wild-type and mutant VWF. Wild-type VWF bears highly sialylated N-glycan branches with both 3- and 6-linked sialic acids. Absence of ST6Gal-I eliminates only 6-linked sialic acid, although a partial compensation by ST3Gal-IV may occur (lightly shaded sialic acid). Separately, ST3Gal-IV deficiency exposes ECA-reactive Gal 1 4 GlcNAc termini and may also expose the Gal 1 3GlcNAc- disaccharide. Partial compensation (shaded sialic acid linkage) by ST6Gal-I occurs, whereas no sialyltransferase exists that can add 6 linkages to Gal 1 3GlcNAc-. However, some degree of 3 sialylation of Gal 1 3GlcNAc- may occur (shaded sialic acid). No changes occur in the sialylation of the Core 1 O-glycan Gal 1 3GalNAc-. (F) Multimer analysis of plasma VWF from wild-type and ST3Gal-IV mutants indicates normal multimer formation in mutant samples. Data are representative of three separate experiments. Lectin binding in A C was quantitated by ELISA and plotted as the ratio of lectin binding per unit of VWF antigen (see Methods). Data in A C are means SEM as derived from analyses of 12 or more mice of each genotype in each assay. Saccharide symbols: GlcNAc, black square; GalNAc; open square; Gal, black circle; sialic acid, black diamond. Factor VIII levels in plasma were reduced similarly compared with its carrier molecule VWF (Fig. 2C). Other coagulation factors were, however, present at normal levels in plasma (Fig. 2D), and analyses of serum chemistry indicated normal hepatic and renal function (data not shown). The cause of the increased bleeding times is not known but is not likely due to the degree of VWF deficiency observed, and analysis of VWF function in a collagenbinding assay indicated no change in this activity (data not shown). Deficient Sialylation of VWF in ST3Gal-IV and ST6Gal-I Deficiency with Normal VWF Multimer Formation. Loss of either ST3Gal-IV or ST6Gal-I sialyltransferase activity would be expected to increase exposure of subterminal -linked Gal on various glycan branches. Reactivity of the -Gal-binding lectin RCA-I (ref. 31) to VWF was increased in ST3Gal-IV mutant plasma samples (Fig. 3A). Unexpectedly, reduced RCA-1 binding to VWF was observed in ST6Gal-I deficiency (Fig. 3A). The reason for this reduction is unclear but may reflect uncharacterized influences of other terminal glycan structures in RCA-1 binding. Analyses after neuraminidase (sialidase) treatment to remove all 2 3 and -6 sialic acids linkages indicated that sialic acids remained on VWF in the absence of either ST3Gal-IV or ST6Gal-I (Fig. 3A) cgi doi pnas Ellies et al.
4 Gal exposure on the type II branch structure Gal 1 4GlcNAcwas detected by using ECA-I (refs. 32 and 33). An increase in ECA-I binding to VWF was observed among plasma samples from both ST3Gal-IV- and ST6Gal-I-deficient mice (Fig. 3B). ECA binding after sialidase treatment was greatly increased, suggesting that sialic acid linkages to the type II glycan branch structure remained with either sialyltransferase deficiency. Further evidence of ST6Gal-I sialylation of VWF was obtained by using the SNA lectin, which specifically binds to the sialylated type II glycan (Sia 2 6Gal 1 4GlcNAc-; ref. 34). Complete loss of SNA binding to VWF was observed in mice lacking the ST6Gal-I sialyltransferase or VWF treated with sialidase. However, increased SNA binding to VWF was evident among ST3Gal-IV-deficient mice suggesting compensation by ST6Gal-I (Fig. 3C). PNA lectin binds specifically to the unsialylated O-glycan Core 1 branch structure: Gal 1 3GalNAc- (35). No change in PNA binding to VWF occurred in mice deficient in either ST3Gal-IV or ST6Gal-I (Fig. 3D). Lectin-binding profiles to VWF are consistent with the presence of glycan terminal branch sequences indicated in Fig. 3E (Left). These branches may also be modified in some cases by 1 2-linked fucose on the penultimate Gal, by 1 3-linked fucose on the N-acetylglucosamine proximal to the -linked Gal, or in the mouse by terminal 1 3 Gal (not shown). In ST6Gal-I-deficient mice, a fraction of type II branch termini may be sialylated by other sialyltransferases bearing overlap in substrate specificity, such as ST3Gal-III, -IV, or -VI (Fig. 3E Center). However, such compensation would be incomplete as ECA-I binding to VWF is increased in ST6Gal-I-deficient mice. In contrast, VWF in ST3Gal-IVdeficient mice may have exposed Gal on types II and I (Gal 1 3GlcNAc-) glycans, should both chains be present on mouse VWF (Fig. 3E Right). Compensation by ST6Gal-I is evident from the increased SNA binding to VWF. However, this compensation is incomplete, as indicated by increased ECA-I binding. These alterations in glycan sialylation in ST3Gal-IV deficiency do not, however, result in a defect in VWF multimerization (Fig. 3F). ST3Gal-IV Mutation Alters VWF Trafficking and Half-Life by ASGP Recognition and Clearance Mechanisms. Elimination of sialic acids can reduce the half-life of some glycoproteins in circulation by ASGP clearance mechanisms (reviewed in ref. 36). ASGPRs exist among hepatocytes and macrophages in organs including the liver and spleen. We observed a significant increase in VWF antigen levels in the liver of ST3Gal-IV mutant mice (data not shown) and investigated whether the reduction in plasma VWF was due to ASGPR recognition and clearance. Mouse plasma samples were biotinylated before i.v. injection into wild-type mice in the presence or absence of coinjected fetuin glycoprotein preparations. The levels of biotinylated VWF in plasma were measured over a time course to obtain the half-life of VWF in circulation (Fig. 4A). Wild-type VWF has a half-life of 4.5 h, whereas VWF from ST3Gal-IV homozygous mutant mice exhibits a half-life of 1.9 h. A half-life of 2.7 h was observed for heterozygote-derived VWF (data not shown). No change in VWF half-life was observed among ST6Gal-I-deficient plasma samples (Fig. 4A). Injection of native sialylated fetuin did not alter the rate of glycoprotein clearance from plasma (data not shown); however, coinjection of asialofetuin, which saturates ASPGRs, resulted in the restoration of normal half-life in circulation for VWF in ST3Gal-IV deficiency. Autosomal Recessive Thrombocytopenia Corrected by Blocking ASGPR in Vivo. Both ST3Gal-IV and ST6Gal-I mutant mice were subjected to an extensive hematologic examination. No remarkable findings were noted among ST6Gal-I null mice; however, ST3Gal-IV deficiency resulted in a marked decrease in platelets to 30% of normal with a substantial increase in platelet volume among homozygotes (Table 1). This could reflect sequestration and degradation of platelets in the spleen; however, splenomegaly was not present, and Fig. 4. Decreased VWF half-life and platelet homeostasis corrected by blocking ASGPR. (A) Plasma fractions from mice of indicated genotypes were biotinylated and injected into wild-type mice in the presence or absence of 10 mg of pyrogenfree asialofetuin. Biotinylated VWF levels in blood were analyzed at the indicated times. Data are derived from three mice per treatment group and are representative of two separate experiments. In additional experiments that included native fetuin, only asialofetuin was able to block ASGP clearance and increase half-life (data not shown). (B) RCA lectin binding to platelets was increased among blood samples from mice homozygous for the ST3Gal-IV mutation as detected by flow cytometric analysis. A slight increase in RCA-I binding to platelets is seen in heterozygous samples. Data are representative of three separate experiments. (C) Mice homozygous for the ST3Gal-IV gene deletion were injected with asialofetuin or fetuin at 0, 4, and 8 h. Platelet levels were analyzed by flow cytometry of whole blood at the timepoints indicated. Data are the means SEM from 10 mice per group. no increase in platelet numbers occurred on splenectomy (data not shown). Megakaryocytes were not diminished among the bone marrow and spleen; in fact, there was a trend toward an increase in Table 1. Hematology Wild type ST3Gal-IV wt/ ST3Gal-IV / ST6Gal-I / WBC, cells per l 6, , , , RBC, m l HGB, g dl HCT, % PLT, k l 1, , * 1, MPV, fl * Sample size, n Values are presented as means SEM. WBC, white blood cells; RBC, red blood cells; HGB, hemoglobin; HCT, hematocrit; PLT, platelet; MPV, mean platelet volume. *, P between wild-type and genotypes. MEDICAL SCIENCES Ellies et al. PNAS July 23, 2002 vol. 99 no
5 mean value. Moreover, four of those five patients had the lowest VWF levels recorded among all 136 patient samples (Fig. 5B). In no case did an increase in RCA-I VWF ratio above 2 SD from the mean occur in human plasma samples bearing 50% or more of the normal level of VWF. These data reveal that a subpopulation of humans with abnormally low plasma levels of VWF have unusually high -linked Gal or GalNAc exposure that may target VWF for ASGPR recognition and clearance. Fig. 5. RCA VWF ratios in ST3Gal-IV deficiency are similar to those in a subpopulation of humans bearing abnormally low plasma VWF levels. (A) Ratios of RCA-I binding to VWF antigen level in mouse plasma consist of low values in wild-type littermates. In ST3Gal-IV mutant mice, ratios are almost always increased to greater than 2 SD above the mean, as indicated by the dotted line. Data reflect more than 15 mice of each genotype. (B) A subpopulation of humans bearing abnormally low VWF levels shows increased RCA VWF ratios more than 2 SD from the mean. No examples of similarly increased RCA VWF ratios were found among samples bearing more than 50% the normal level of VWF. megakaryocyte numbers with normal cellular morphology (data not shown). We suspected that the thrombocytopenia results from an increase in platelet clearance by ASGPRs expressed among nonsplenic tissue. A significant increase in -linked Gal exposure was observed on the platelet cell surface of homozygous mutants in ST3Gal-IV measured by RCA-I binding in flow cytometric analyses (Fig. 4B). Platelets from heterozygotes had very slightly increased levels of -linked Gal exposure. To determine whether ASGPRs were involved, i.p. injections of asialofetuin or sialylated fetuin were administered. ST3Gal-IV mutant mice receiving native sialylated fetuin continued to exhibit thrombocytopenia, whereas those receiving asialofetuin responded with an increase in circulating platelets to normal levels within 12 h (Fig. 4C). Gal Exposure on VWF in a Patient Subpopulation with Abnormally Low VWF Levels. RCA-I lectin binds to VWF antigen from wild-type mice with an RCA-I VWF ratio of 1.25:1. A substantial increase in this ratio to more than 2 SD above the mean occurs in mice lacking ST3Gal-IV (Fig. 5A). However, variation remained from 3:1 to 11:1, suggesting the presence of other genetic and stress modifiers that may contribute to VWF plasma levels. Plasma from patients referred to the Special Coagulation Laboratory at the University of California at San Diego Hillcrest Hematology Clinic over a 2-yr period for a real or suspected bleeding disorder were analyzed for RCA-I VWF ratios. VWF levels were expressed as a percentage of the value obtained from a pooled normal human plasma standard. Of 136 human patient samples analyzed, 117 contained VWF levels at 50% or more of the pooled normal human plasma standard. However, 19 patients were deficient in VWF by 50% or more. The VWF from 5 of these 19 patients had RCA-I VWF ratios of more than 2 8 SD above the Discussion The ST3Gal-IV sialyltransferase is distinct from ST6Gal-I in the ability to modulate hemostasis by differential sialylation of VWF in the mouse. Previous studies of the stabilizing role of sialic acids in VWF homeostasis used global desialylation approaches in vitro followed by recombinant ST6Gal-I expression. Although sialic acids contributed by sialyltransferases other than ST3Gal-IV may also be involved in modulating VWF homeostasis, ST6Gal-I does not perform this function independently. Further, it is unlikely that asialo-vwf molecules with half-lives of a few minutes are produced in vivo, because this would require the absence of multiple sialyltransferases, a deficit of sialic acid in the Golgi that can be lethal, or perhaps abnormal induction of an endogenous or pathogen-derived sialidase. For these reasons, the physiologic properties ascribed to sialic acid linkages and their regulation by sialyltransferases cannot be empirically investigated by available desialylation schemes. By using a genetic approach to investigate sialyltransferase function in vivo, a high degree of functional specificity can be observed that identifies the ST3Gal-IV sialyltransferase as a discrete modifier of hemostasis that acts by concealing ASGPR ligands and thereby contributing to VWF homeostasis. Most naturally occurring or experimentally derived genetic defects in the mouse yield autosomal recessive phenotypes. ST3Gal-IV mutations, however, result in an autosomal dominant phenotype with reductions in plasma VWF levels apparently due to haploinsufficiency. It is possible that the ST3Gal-IV mutation produced herein leads to the production of a truncated polypeptide bearing a dominant-negative function. This appears unlikely, however, as the catalytic deletion engineered destabilizes the RNA to the extent that no expression is observed. In addition, the thrombocytopenia is an autosomal recessive phenotype due to similar ASGPR clearance mechanisms without a significant increase in -linked Gal exposure or a decrease in platelet levels in the heterozygote. Sialyltransferases in Hemostasis. VWF is a highly glycosylated and sialylated protein. Analyses in humans show that VWF contains approximately 24 glycosylation sites with 14 involved in N-glycan modification leading to various multiantennary N-glycan branch structures (37). In addition, VWF contains O-glycans that have sialic acid linked to the Core 1 Gal and the peptide-proximal GalNAc (38). Together, this glycan diversity encompasses two sialic acid linkage types ( 2 3 and -6) distributed on at least five different glycan branch structures. Our data indicate that multiple sialyltransferases modify VWF in vivo. Besides ST6Gal-I and ST3Gal- IV, seven others may act, including ST3Gal-I, -III, -VI, and ST6GalNAc-I-IV. Discerning which may modulate VWF homeostasis has been unclear as all these exhibit substrate specificities for Gal and GalNAc linkages detected on human VWF, and their expression may overlap in VWF-producing endothelium. A reproducible, quantitative, and low RCA VWF ratio exists among most human plasma samples. However, a small subpopulation of individuals was identified with both abnormally low VWF levels and abnormally high Gal exposure detected by RCA-I lectin binding. Aberrantly low VWF levels in the presence of high RCA-I binding suggest the possibility of a defect in a sialyltransferase such as ST3Gal-IV, or perhaps a mutation in VWF that alters conformation and attenuates sialylation of VWF in the Golgi. The degree cgi doi pnas Ellies et al.
6 of plasma VWF deficiency in four of the five cases found was severe and only slightly higher than the VWF levels characteristic of type 3 VWD. However, the thrombocytopenia noted among ST3Gal-IV homozygous null mice is not a feature of human type 3 VWD. Although thrombocytopenia is characteristic of type 2B VWD, this variant exhibits enhanced VWF affinity for platelets leading to spontaneous platelet aggregation and a defect in circulating VWF multimers (39). Systemic glycosyltransferase deficiencies may be expected to affect many different glycoproteins. Indeed, the inherited glycosyltransferase deficiencies responsible for the severe childhood syndromes termed the Congenital Defects in Glycosylation (CDG) include diagnostic blood coagulopathies involving multiple glycoproteins with reduced levels of antithrombin-iii, Factors VII, XI, XII, and protein C (40). CDG type I is due to abnormal processing of N-glycans in the endoplasmic reticulum, which can widely attenuate glycoprotein folding, maturation, and expression. In a mouse model of CDG type II, the presence of a typical blood coagulopathy likely reflects widespread and essential roles involving multiple N-glycan branches on glycoproteins that are produced early in the biosynthetic process in the Golgi apparatus (26). In contrast, sialyltransferase deficiencies yield less extensive structural changes and may involve fewer substrates dictated by expression patterns, substrate specificity, access to substrate, and access to substrate in the Golgi glycan biosynthetic pathway. In fact, the steady-state plasma levels of blood coagulation factors produced in the liver were unaltered in ST3Gal-IV deficiency. These glycoproteins may not be modified by ST3Gal-IV. Alternatively, Gal exposure per se may be insufficient to comprise an ASGPR ligand in vivo. The Genetic Basis of ASGPR Ligand Masking in Vivo. Multivalency and conformation appear as important elements of ASGPR function and lectin binding in general (reviewed in ref. 41). Although both ST6Gal-I and ST3Gal-IV sialylate VWF in vivo, ST6Gal-I prefers type II glycan branches residing on the 1 3 linked mannose of N-glycans (42). ST3Gal-IV, however, can sialylate both types II and I glycan branch termini without such preference for a subset of type II branches. Therefore, ST6Gal-I deficiency may not result in the exposure of multiple closely spaced -linked Gal residues, which may be more important than the molecular abundance of types I or II branches. In these considerations, the different branch substrate specificity of ST3Gal-IV in vivo may effectively mask multivalent Gal-bearing glycan branches that comprise an ASGPR ligand. ST3Gal-IV conceals ligands for multiple ASGPRs in altering ASGPR-dependent clearance of both VWF and platelets. The hepatic ASGPR can bind and initiate the phagocytosis of particles less than 8 nm in diameter (43). Large particles such as platelets could be bound and processed by the macrophage ASGPR, which differs in molecular weight, membrane anchorage, and receptor arrangement (43, 44). In particular, the arrangement of macrophage ASGPRs in clusters on the cell surface is thought to be critical for the binding and endocytosis of particulate ligands such as platelets. The identification of ASGPRs that modulate VWF and platelet levels in ST3Gal-IV deficiency should provide information useful in understanding further this endogenous hemostatic lectinligand system. Plasma VWF levels are highly variable in normal mammalian physiology, as well as among human patients with VWD. This variability appears to be a major determinant of bleeding severity in VWD. We find that ST3Gal-IV acts as a limiting component in the masking of ASGPR ligands, achieving an equilibrium involving VWF and platelet levels in circulation that is susceptible to modulation. Intriguingly, reductions in sialic acid abundance on VWF and Factor VIII have been reported in some cases of VWD (45 47). Endogenous mechanisms that limit plasma expression of VWF may also be vital. High levels of VWF and Factor VIII appear to be a major risk factor for thrombosis in humans (48). Our findings suggest that complex interactions among glycosyltransferases are a critical factor in regulating the steady-state level of plasma constituents, including VWF and platelets, and identify the ST3Gal-IV sialyltransferase as an attractive candidate for a major VWF modifier gene in humans. This research was funded by the National Institutes of Health program project Grant PO1-HL57345, Fellowship F32CA79130 (L.G.E.), and Grant R01-HL39693 (D.G.). J.D.M. and D.G. are supported as Investigators of the Howard Hughes Medical Institute. 1. Ruggeri, Z. M., Ware, J. & Ginsberg, D. (1998) in Thrombosis and Hemorrhage, eds. Loscalzo, J. & Schafer, A. I. (Williams & Wilkins, Baltimore), pp Sadler, J. E. (1998) Annu. Rev. Biochem. 67, Nichols, W. C. & Ginsburg, D. (1997) Medicine (Baltimore) 76, Denis, C. V. & Wagner, D. D. (1999) Cell Mol. Life Sci. 56, Berkowitz, S. D. & Federici, A. B. (1988) Blood 72, Carew, J. A., Quinn, S. M., Stoddart, J. H. & Lynch, D. C. (1992) J. Clin. Invest. 90, Federici, A. B., Elder, J. H., De Marco, L., Ruggeri, Z. M. & Zimmerman, T. S. (1984) J. Clin. Invest. 74, Sodetz, J. M., Pizzo, S. V. & McKee, P. A. (1977) J. Biol. Chem. 252, Sodetz, J. M., Paulson, J. C., Pizzo, S. V. & McKee, P. A. (1978) J. Biol. Chem. 253, Wagner, D. D., Mayadas, T. & Marder, V. J. (1986) J. Cell Biol. 102, Marth, J. D. (1999) in Essentials of Glycobiology, eds. Varki, A., Cummings, R., Esko, H., Freeze, H., Hart, G. & Marth, J. (Cold Spring Harbor Lab. Press, Plainview, NY), pp Gill, J. C., Endres-Brooks, J., Bauer, P. J., Marks, W. J., Jr., & Montgomery, R. R. (1987) Blood 69, Orstavik, K. H., Kornstad, L., Reisner, H. & Berg, K. (1989) Blood 73, Mohlke, K. L., Purkayastha, A. A., Westrick, R. J., Smith, P. L., Petryniak, B., Lowe, J. B. & Ginsburg, D. (1999) Cell 96, Kitagawa, H. & Paulson, J. C. (1994) J. Biol. Chem. 269, Takashima, S. & Tsuji, S. (2000) Cytogenet. Cell Genet. 89, Lowe, J. B. & Marth, J. D. (1999) in Essentials of Glycobiology, eds. Varki, A., Cummings, R., Esko, H., Freeze, H., Hart, G. & Marth, J. (Cold Spring Harbor Lab. Press, Plainview, NY), pp Harduin-Lepers, A., Vallejo-Ruiz, V., Krzewinski-Recchi, M., Samyn-Petit, B., Julien, S. & Delannoy, P. (2001) Biochimie 83, Crocker, P. R. & Varki, A. (2001) Trends Immunol. 22, Lowe, J. B. (1997) Kidney Int. 51, Gagneux, P. & Varki, A. (1999) Glycobiology 9, Stockert, R. J., Morell, A. G. & Ashwell, G. (1991) Targeted Diagn. Ther. 4, Cooper, D. N. & Barondes, S. H. (1999) Glycobiology 9, Hennet, T., Chui, D., Paulson, J. C. & Marth, J. D. (1998) Proc. Natl. Acad. Sci. USA 95, Shafi, R., Iyer, S. P., Ellies, L. G., O Donnell, N., Marek, K. W., Chui, D., Hart, G. W. & Marth, J. D. (2000) Proc. Natl. Acad. Sci. USA 97, Wang, Y., Tan, J., Sutton-Smith, M., Ditto, D., Panico, M., Campbell, R. M., Varki, N. M., Long, J. M., Jaeken, J., Levinson, S. R., et al. (2001) Glycobiology 11, Lawrie, A. S., Hoser, M. J. & Savidge, G. F. (1990) Thromb. Res. 59, Varki, A. & Diaz, S. (1984) Anal. Biochem. 137, Taktak, Y. S., Selkirk, S., Bristow, A. F., Carpenter, A., Ball, C., Rafferty, B. & Poole, S. (1991) J. Pharm. Pharmacol. 43, Datta, A. K. & Paulson, J. C. (1995) J. Biol. Chem. 270, Baenziger, J. U. & Fiete, D. (1979) J. Biol. Chem. 254, Debray, H., Montreuil, J., Lis, H. & Sharon, N. (1986) Carbohydr. Res. 151, Moreno, E., Teneberg, S., Adar, R., Sharon, N., Karlsson, K. A. & Angstrom, J. (1997) Biochemistry 36, Shibuya, N., Goldstein, I. J., Broekaert, W. F., Nsimba-Lubaki, M., Peeters, B. & Peumans, W. J. (1987) J. Biol. Chem. 262, Lotan, R., Skutelsky, E., Danon, D. & Sharon, N. (1975) J. Biol. Chem. 250, Weigel, P. H. (1994) BioEssays 16, Matsui, T., Titani, K. & Mizuochi, T. (1992) J. Biol. Chem. 267, Samor, B., Michalski, J. C., Mazurier, C., Goudemand, M., De Waard, P., Vliegenthart, J. F., Strecker, G. & Montreuil, J. (1989) Glycoconj. J. 6, Ruggeri, Z. M. & Zimmerman, T. S. (1980) J. Clin. Invest. 65, Jaeken, J., Matthijs, G., Caarchon, J. & Van Schaftingen, E. (2001) in The Metabolic and Molecular Bases of Inherited Disease, eds. Scriver, C. R., Beaudet, A. L., Sly, W. S. & Vallee, D. (McGraw Hill, New York), pp Rice, K. G. & Lee, Y. C. (1993) Adv. Enzymol. Relat. Areas Mol. Biol. 66, Joziasse, D. H., Schiphorst, W. E., van den Eijnden, D. H., van Kuik, J. A., van Halbeek, H. & Vliegenthart, J. F. (1985) J. Biol. Chem. 260, Schlepper-Schafer, J., Hulsmann, D., Djovkar, A., Meyer, H. E., Herbertz, L., Kolb, H. & Kolb-Bachofen, V. (1986) Exp. Cell Res. 165, Roos, P. H., Hartman, H. J., Schlepper-Schafer, J., Kolb, H. & Kolb-Bachofen, V. (1985) Biochim. Biophys. Acta 847, Gralnick, H. R., Coller, B. S. & Sultan, Y. (1976) Science 192, Gralnick, H. R., Sultan, Y. & Coller, B. S. (1977) N. Engl. J. Med. 296, Gralnick, H. R., Cregger, M. C. & Williams, S. B. (1982) Blood 59, Rosendaal, F. R. (2000) Thromb. Haemostasis 83, 1 2. MEDICAL SCIENCES Ellies et al. PNAS July 23, 2002 vol. 99 no
BIOSYNTHESIS OF CANCER-RELATED CARBOHYDRATE ANTIGENS. Fabio Dall Olio Department of Experimental Pathology University of Bologna, Italy
 BIOSYNTHESIS OF CANCER-RELATED CARBOHYDRATE ANTIGENS Fabio Dall Olio Department of Experimental Pathology University of Bologna, Italy TOPICS OF THE LECTURE 1. Structure and function of some representative
BIOSYNTHESIS OF CANCER-RELATED CARBOHYDRATE ANTIGENS Fabio Dall Olio Department of Experimental Pathology University of Bologna, Italy TOPICS OF THE LECTURE 1. Structure and function of some representative
Biosynthesis of N and O Glycans
 TechNote #TNGL101 Biosynthesis of N and O Glycans These suggestions and data are based on information we believe to be reliable. They are offered in good faith, but without guarantee, as conditions and
TechNote #TNGL101 Biosynthesis of N and O Glycans These suggestions and data are based on information we believe to be reliable. They are offered in good faith, but without guarantee, as conditions and
The addition of sugar moiety determines the blood group
 The addition of sugar moiety determines the blood group Sugars attached to glycoproteins and glycolipids on the surfaces of red blood cells determine the blood group termed A, B, and O. The A and B antigens
The addition of sugar moiety determines the blood group Sugars attached to glycoproteins and glycolipids on the surfaces of red blood cells determine the blood group termed A, B, and O. The A and B antigens
Influenza A H1N1 HA ELISA Pair Set
 Influenza A H1N1 HA ELISA Pair Set for H1N1 ( A/Puerto Rico/8/1934 ) HA Catalog Number : SEK11684 To achieve the best assay results, this manual must be read carefully before using this product and the
Influenza A H1N1 HA ELISA Pair Set for H1N1 ( A/Puerto Rico/8/1934 ) HA Catalog Number : SEK11684 To achieve the best assay results, this manual must be read carefully before using this product and the
Supplementary figure 1 Supplementary figure 2
 Supplementary figure 1 Schematic overview of the Fc-glycan of IgG. The glycan is composed of a constant core domain (highlighted in red) composed of mannose (Man) and N-acetylglucosamine (GlcNAc) residues
Supplementary figure 1 Schematic overview of the Fc-glycan of IgG. The glycan is composed of a constant core domain (highlighted in red) composed of mannose (Man) and N-acetylglucosamine (GlcNAc) residues
Significance and Functions of Carbohydrates. Bacterial Cell Walls
 Biochemistry 462a - Carbohydrate Function Reading - Chapter 9 Practice problems - Chapter 9: 2, 4a, 4b, 6, 9, 10, 13, 14, 15, 16a, 17; Carbohydrate extra problems Significance and Functions of Carbohydrates
Biochemistry 462a - Carbohydrate Function Reading - Chapter 9 Practice problems - Chapter 9: 2, 4a, 4b, 6, 9, 10, 13, 14, 15, 16a, 17; Carbohydrate extra problems Significance and Functions of Carbohydrates
Human LDL Receptor / LDLR ELISA Pair Set
 Human LDL Receptor / LDLR ELISA Pair Set Catalog Number : SEK10231 To achieve the best assay results, this manual must be read carefully before using this product and the assay is run as summarized in
Human LDL Receptor / LDLR ELISA Pair Set Catalog Number : SEK10231 To achieve the best assay results, this manual must be read carefully before using this product and the assay is run as summarized in
Human Cathepsin D ELISA Kit
 GenWay Biotech, Inc. 6777 Nancy Ridge Drive San Diego, CA 92121 Phone: 858.458.0866 Fax: 858.458.0833 Email: techline@genwaybio.com http://www.genwaybio.com Human Cathepsin D ELISA Kit Catalog No. GWB-J4JVV9
GenWay Biotech, Inc. 6777 Nancy Ridge Drive San Diego, CA 92121 Phone: 858.458.0866 Fax: 858.458.0833 Email: techline@genwaybio.com http://www.genwaybio.com Human Cathepsin D ELISA Kit Catalog No. GWB-J4JVV9
Supplementary Figures
 Supplementary Figures Supplementary Fig. 1. Galectin-3 is present within tumors. (A) mrna expression levels of Lgals3 (galectin-3) and Lgals8 (galectin-8) in the four classes of cell lines as determined
Supplementary Figures Supplementary Fig. 1. Galectin-3 is present within tumors. (A) mrna expression levels of Lgals3 (galectin-3) and Lgals8 (galectin-8) in the four classes of cell lines as determined
The autoimmune disease-associated PTPN22 variant promotes calpain-mediated Lyp/Pep
 SUPPLEMENTARY INFORMATION The autoimmune disease-associated PTPN22 variant promotes calpain-mediated Lyp/Pep degradation associated with lymphocyte and dendritic cell hyperresponsiveness Jinyi Zhang, Naima
SUPPLEMENTARY INFORMATION The autoimmune disease-associated PTPN22 variant promotes calpain-mediated Lyp/Pep degradation associated with lymphocyte and dendritic cell hyperresponsiveness Jinyi Zhang, Naima
Supplementary Figure 1. Generation of knockin mice expressing L-selectinN138G. (a) Schematics of the Sellg allele (top), the targeting vector, the
 Supplementary Figure 1. Generation of knockin mice expressing L-selectinN138G. (a) Schematics of the Sellg allele (top), the targeting vector, the targeted allele in ES cells, and the mutant allele in
Supplementary Figure 1. Generation of knockin mice expressing L-selectinN138G. (a) Schematics of the Sellg allele (top), the targeting vector, the targeted allele in ES cells, and the mutant allele in
Influenza A H1N1 (Swine Flu 2009) Hemagglutinin / HA ELISA Pair Set
 Influenza A H1N1 (Swine Flu 2009) Hemagglutinin / HA ELISA Pair Set Catalog Number : SEK001 To achieve the best assay results, this manual must be read carefully before using this product and the assay
Influenza A H1N1 (Swine Flu 2009) Hemagglutinin / HA ELISA Pair Set Catalog Number : SEK001 To achieve the best assay results, this manual must be read carefully before using this product and the assay
Ch 8 Practice Questions
 Ch 8 Practice Questions Multiple Choice Identify the choice that best completes the statement or answers the question. 1. What fraction of offspring of the cross Aa Aa is homozygous for the dominant allele?
Ch 8 Practice Questions Multiple Choice Identify the choice that best completes the statement or answers the question. 1. What fraction of offspring of the cross Aa Aa is homozygous for the dominant allele?
(Stratagene, La Jolla, CA) (Supplemental Fig. 1A). A 5.4-kb EcoRI fragment
 SUPPLEMENTAL INFORMATION Supplemental Methods Generation of RyR2-S2808D Mice Murine genomic RyR2 clones were isolated from a 129/SvEvTacfBR λ-phage library (Stratagene, La Jolla, CA) (Supplemental Fig.
SUPPLEMENTAL INFORMATION Supplemental Methods Generation of RyR2-S2808D Mice Murine genomic RyR2 clones were isolated from a 129/SvEvTacfBR λ-phage library (Stratagene, La Jolla, CA) (Supplemental Fig.
Human Urokinase / PLAU / UPA ELISA Pair Set
 Human Urokinase / PLAU / UPA ELISA Pair Set Catalog Number : SEK10815 To achieve the best assay results, this manual must be read carefully before using this product and the assay is run as summarized
Human Urokinase / PLAU / UPA ELISA Pair Set Catalog Number : SEK10815 To achieve the best assay results, this manual must be read carefully before using this product and the assay is run as summarized
Laura T. Martin, Jamey D. Marth, Ajit Varki, and Nissi M. Varki
 THE JOURNAL OF BIOLOGICAL CHEMISTRY Vol. 277, No. 36, Issue of September 6, pp. 32930 32938, 2002 2002 by The American Society for Biochemistry and Molecular Biology, Inc. Printed in U.S.A. Genetically
THE JOURNAL OF BIOLOGICAL CHEMISTRY Vol. 277, No. 36, Issue of September 6, pp. 32930 32938, 2002 2002 by The American Society for Biochemistry and Molecular Biology, Inc. Printed in U.S.A. Genetically
Influenza B Hemagglutinin / HA ELISA Pair Set
 Influenza B Hemagglutinin / HA ELISA Pair Set Catalog Number : SEK11053 To achieve the best assay results, this manual must be read carefully before using this product and the assay is run as summarized
Influenza B Hemagglutinin / HA ELISA Pair Set Catalog Number : SEK11053 To achieve the best assay results, this manual must be read carefully before using this product and the assay is run as summarized
Glycoprotein Deglycosylation Kit Cat. No
 Visit our interactive pathways at /pathways User Protocol 362280 Rev. 23 February 2006 RFH Page 1 of 5 Glycoprotein Deglycosylation Kit Cat. No. 362280 Note that this user protocol is not lot-specific
Visit our interactive pathways at /pathways User Protocol 362280 Rev. 23 February 2006 RFH Page 1 of 5 Glycoprotein Deglycosylation Kit Cat. No. 362280 Note that this user protocol is not lot-specific
Mouse Cathepsin B ELISA Kit
 GenWay Biotech, Inc. 6777 Nancy Ridge Drive San Diego, CA 92121 Phone: 858.458.0866 Fax: 858.458.0833 Email: techline@genwaybio.com http://www.genwaybio.com Mouse Cathepsin B ELISA Kit Catalog No. GWB-ZZD154
GenWay Biotech, Inc. 6777 Nancy Ridge Drive San Diego, CA 92121 Phone: 858.458.0866 Fax: 858.458.0833 Email: techline@genwaybio.com http://www.genwaybio.com Mouse Cathepsin B ELISA Kit Catalog No. GWB-ZZD154
Coagulation Disorders. Dr. Muhammad Shamim Assistant Professor, BMU
 Coagulation Disorders Dr. Muhammad Shamim Assistant Professor, BMU 1 Introduction Local Vs. General Hematoma & Joint bleed Coagulation Skin/Mucosal Petechiae & Purpura PLT wound / surgical bleeding Immediate
Coagulation Disorders Dr. Muhammad Shamim Assistant Professor, BMU 1 Introduction Local Vs. General Hematoma & Joint bleed Coagulation Skin/Mucosal Petechiae & Purpura PLT wound / surgical bleeding Immediate
Human Immunodeficiency Virus type 1 (HIV-1) gp120 / Glycoprotein 120 ELISA Pair Set
 Human Immunodeficiency Virus type 1 (HIV-1) gp120 / Glycoprotein 120 ELISA Pair Set Catalog Number : SEK11233 To achieve the best assay results, this manual must be read carefully before using this product
Human Immunodeficiency Virus type 1 (HIV-1) gp120 / Glycoprotein 120 ELISA Pair Set Catalog Number : SEK11233 To achieve the best assay results, this manual must be read carefully before using this product
Supplementary Data 1. Alanine substitutions and position variants of APNCYGNIPL. Applied in
 Supplementary Data 1. Alanine substitutions and position variants of APNCYGNIPL. Applied in Supplementary Fig. 2 Substitution Sequence Position variant Sequence original APNCYGNIPL original APNCYGNIPL
Supplementary Data 1. Alanine substitutions and position variants of APNCYGNIPL. Applied in Supplementary Fig. 2 Substitution Sequence Position variant Sequence original APNCYGNIPL original APNCYGNIPL
Human Immunodeficiency Virus type 1 (HIV-1) p24 / Capsid Protein p24 ELISA Pair Set
 Human Immunodeficiency Virus type 1 (HIV-1) p24 / Capsid Protein p24 ELISA Pair Set Catalog Number : SEK11695 To achieve the best assay results, this manual must be read carefully before using this product
Human Immunodeficiency Virus type 1 (HIV-1) p24 / Capsid Protein p24 ELISA Pair Set Catalog Number : SEK11695 To achieve the best assay results, this manual must be read carefully before using this product
New Tools to Study and Perturb the Glycocalyx
 New Tools to Study and Perturb the Glycocalyx Glycobiology 2015 10-12 August, 2015 Thomas Boltje Institute for Molecules and Materials Radboud University The Netherlands Sialic Acid Structure and Function
New Tools to Study and Perturb the Glycocalyx Glycobiology 2015 10-12 August, 2015 Thomas Boltje Institute for Molecules and Materials Radboud University The Netherlands Sialic Acid Structure and Function
Lectins: selected topics 3/2/17
 Lectins: selected topics 3/2/17 Selected topics Regulation of T-cell receptor signaling Thymic selection of self vs. non-self T-cells Essentials of Glycobiology Second Edition Signaling pathways associated
Lectins: selected topics 3/2/17 Selected topics Regulation of T-cell receptor signaling Thymic selection of self vs. non-self T-cells Essentials of Glycobiology Second Edition Signaling pathways associated
Nature Biotechnology: doi: /nbt Supplementary Figure 1. RNAseq expression profiling of selected glycosyltransferase genes in CHO.
 Supplementary Figure 1 RNAseq expression profiling of selected glycosyltransferase genes in CHO. RNAseq analysis was performed on two common CHO lines (CHO-K1, CHO-GS) and two independent CHO-GS triple
Supplementary Figure 1 RNAseq expression profiling of selected glycosyltransferase genes in CHO. RNAseq analysis was performed on two common CHO lines (CHO-K1, CHO-GS) and two independent CHO-GS triple
Problem Set 8 Key 1 of 8
 7.06 2003 Problem Set 8 Key 1 of 8 7.06 2003 Problem Set 8 Key 1. As a bright MD/PhD, you are interested in questions about the control of cell number in the body. Recently, you've seen three patients
7.06 2003 Problem Set 8 Key 1 of 8 7.06 2003 Problem Set 8 Key 1. As a bright MD/PhD, you are interested in questions about the control of cell number in the body. Recently, you've seen three patients
TECHNICAL BULLETIN. Enzymatic Protein Deglycosylation Kit. Catalog Number EDEGLY Storage Temperature 2 8 C
 Enzymatic Protein Deglycosylation Kit Catalog Number EDEGLY Storage Temperature 2 8 C TECHNICAL BULLETIN Product Description The EDEGLY kit contains all the enzymes and reagents needed to completely remove
Enzymatic Protein Deglycosylation Kit Catalog Number EDEGLY Storage Temperature 2 8 C TECHNICAL BULLETIN Product Description The EDEGLY kit contains all the enzymes and reagents needed to completely remove
2009 H1N1 Influenza ( Swine Flu ) Hemagglutinin ELISA kit
 2009 H1N1 Influenza ( Swine Flu ) Hemagglutinin ELISA kit Catalog Number : SEK001 To achieve the best assay results, this manual must be read carefully before using this product and the assay is run as
2009 H1N1 Influenza ( Swine Flu ) Hemagglutinin ELISA kit Catalog Number : SEK001 To achieve the best assay results, this manual must be read carefully before using this product and the assay is run as
Figure S1. Generation of inducible PTEN deficient mice and the BMMCs (A) B6.129 Pten loxp/loxp mice were mated with B6.
 Figure S1. Generation of inducible PTEN deficient mice and the BMMCs (A) B6.129 Pten loxp/loxp mice were mated with B6.129-Gt(ROSA)26Sor tm1(cre/ert2)tyj /J mice. To induce deletion of the Pten locus,
Figure S1. Generation of inducible PTEN deficient mice and the BMMCs (A) B6.129 Pten loxp/loxp mice were mated with B6.129-Gt(ROSA)26Sor tm1(cre/ert2)tyj /J mice. To induce deletion of the Pten locus,
7.012 Quiz 3 Answers
 MIT Biology Department 7.012: Introductory Biology - Fall 2004 Instructors: Professor Eric Lander, Professor Robert A. Weinberg, Dr. Claudette Gardel Friday 11/12/04 7.012 Quiz 3 Answers A > 85 B 72-84
MIT Biology Department 7.012: Introductory Biology - Fall 2004 Instructors: Professor Eric Lander, Professor Robert A. Weinberg, Dr. Claudette Gardel Friday 11/12/04 7.012 Quiz 3 Answers A > 85 B 72-84
Influenza A H7N9 (A/Anhui/1/2013) Hemagglutinin / HA ELISA Pair Set
 Influenza A H7N9 (A/Anhui/1/2013) Hemagglutinin / HA ELISA Pair Set Catalog Number : SEK40103 To achieve the best assay results, this manual must be read carefully before using this product and the assay
Influenza A H7N9 (A/Anhui/1/2013) Hemagglutinin / HA ELISA Pair Set Catalog Number : SEK40103 To achieve the best assay results, this manual must be read carefully before using this product and the assay
Human HBcAb IgM ELISA kit
 Human HBcAb IgM ELISA kit Catalog number: NR-R10163 (96 wells) The kit is designed to qualitatively detect HBcAb IgM in human serum or plasma. FOR RESEARCH USE ONLY. NOT FOR DIAGNOSTIC OR THERAPEUTIC PURPOSES
Human HBcAb IgM ELISA kit Catalog number: NR-R10163 (96 wells) The kit is designed to qualitatively detect HBcAb IgM in human serum or plasma. FOR RESEARCH USE ONLY. NOT FOR DIAGNOSTIC OR THERAPEUTIC PURPOSES
Fungal Mannoproteins: the Sweet Path to Immunodominance
 Fungal Mannoproteins: the Sweet Path to Immunodominance Mannose residues on glycoproteins trigger key host immune responses that, if better harnessed, could protect against damage and disease Michael K.
Fungal Mannoproteins: the Sweet Path to Immunodominance Mannose residues on glycoproteins trigger key host immune responses that, if better harnessed, could protect against damage and disease Michael K.
Mouse Leptin ELISA Kit (mleptin-elisa)
 Mouse Leptin ELISA Kit (mleptin-elisa) Cat. No. EK0438 96 Tests in 8 x 12 divisible strips Background Leptin (or obese, OB) is a circulating hormone that is expressed abundantly and specifically in the
Mouse Leptin ELISA Kit (mleptin-elisa) Cat. No. EK0438 96 Tests in 8 x 12 divisible strips Background Leptin (or obese, OB) is a circulating hormone that is expressed abundantly and specifically in the
ABO and H Blood Groups. Terry Kotrla, MS, MT(ASCP)BB 2010
 ABO and H Blood Groups Terry Kotrla, MS, MT(ASCP)BB 2010 History Discovered in 1900 by Karl Landsteiner and remains the most important blood group system Mixed blood of colleagues (serum from one, cells
ABO and H Blood Groups Terry Kotrla, MS, MT(ASCP)BB 2010 History Discovered in 1900 by Karl Landsteiner and remains the most important blood group system Mixed blood of colleagues (serum from one, cells
Doctor of Philosophy
 Regulation of Gene Expression of the 25-Hydroxyvitamin D la-hydroxylase (CYP27BI) Promoter: Study of A Transgenic Mouse Model Ivanka Hendrix School of Molecular and Biomedical Science The University of
Regulation of Gene Expression of the 25-Hydroxyvitamin D la-hydroxylase (CYP27BI) Promoter: Study of A Transgenic Mouse Model Ivanka Hendrix School of Molecular and Biomedical Science The University of
SUPPLEMENTARY FIGURES
 SUPPLEMENTARY FIGURES Supplementary Figure 1: Chemokine receptor expression profiles of CCR6 + and CCR6 - CD4 + IL-17A +/ex and Treg cells. Quantitative PCR analysis of chemokine receptor transcript abundance
SUPPLEMENTARY FIGURES Supplementary Figure 1: Chemokine receptor expression profiles of CCR6 + and CCR6 - CD4 + IL-17A +/ex and Treg cells. Quantitative PCR analysis of chemokine receptor transcript abundance
Welcome Back! 2/6/18. A. GGSS B. ggss C. ggss D. GgSs E. Ggss. 1. A species of mice can have gray or black fur
 Welcome Back! 2/6/18 1. A species of mice can have gray or black fur and long or short tails. A cross between blackfurred, long-tailed mice and gray-furred, shorttailed mice produce all black-furred, long-tailed
Welcome Back! 2/6/18 1. A species of mice can have gray or black fur and long or short tails. A cross between blackfurred, long-tailed mice and gray-furred, shorttailed mice produce all black-furred, long-tailed
TECHNICAL BULLETIN. Sialic Acid Quantitation Kit. Catalog Number SIALICQ Storage Temperature 2 8 C
 Sialic Acid Quantitation Kit Catalog Number SIALICQ Storage Temperature 2 8 C TECHNICAL BULLETIN Product Description The Sialic Acid Quantitation Kit provides a rapid and accurate determination of total
Sialic Acid Quantitation Kit Catalog Number SIALICQ Storage Temperature 2 8 C TECHNICAL BULLETIN Product Description The Sialic Acid Quantitation Kit provides a rapid and accurate determination of total
Variations in Chromosome Structure & Function. Ch. 8
 Variations in Chromosome Structure & Function Ch. 8 1 INTRODUCTION! Genetic variation refers to differences between members of the same species or those of different species Allelic variations are due
Variations in Chromosome Structure & Function Ch. 8 1 INTRODUCTION! Genetic variation refers to differences between members of the same species or those of different species Allelic variations are due
Controversies in the Diagnosis of Type 1 VWD. Paula James MD, FRCPC ISLH Honolulu, Hawaii Friday, May 5, 2017
 Controversies in the Diagnosis of Type 1 VWD Paula James MD, FRCPC ISLH Honolulu, Hawaii Friday, May 5, 2017 Disclosures for Paula James Research Support/P.I. Employee Consultant Major Stockholder Speakers
Controversies in the Diagnosis of Type 1 VWD Paula James MD, FRCPC ISLH Honolulu, Hawaii Friday, May 5, 2017 Disclosures for Paula James Research Support/P.I. Employee Consultant Major Stockholder Speakers
7.06 Cell Biology EXAM #3 April 24, 2003
 7.06 Spring 2003 Exam 3 Name 1 of 8 7.06 Cell Biology EXAM #3 April 24, 2003 This is an open book exam, and you are allowed access to books and notes. Please write your answers to the questions in the
7.06 Spring 2003 Exam 3 Name 1 of 8 7.06 Cell Biology EXAM #3 April 24, 2003 This is an open book exam, and you are allowed access to books and notes. Please write your answers to the questions in the
Human inherited diseases
 Human inherited diseases A genetic disorder that is caused by abnormality in an individual's DNA. Abnormalities can range from small mutation in a single gene to the addition or subtraction of a whole
Human inherited diseases A genetic disorder that is caused by abnormality in an individual's DNA. Abnormalities can range from small mutation in a single gene to the addition or subtraction of a whole
Platelet Disorders. By : Saja Al-Oran
 Platelet Disorders By : Saja Al-Oran Introduction The platelet arise from the fragmentation of the cytoplasm of megakaryocyte in the bone marrow. circulate in the blood as disc-shaped anucleate particles
Platelet Disorders By : Saja Al-Oran Introduction The platelet arise from the fragmentation of the cytoplasm of megakaryocyte in the bone marrow. circulate in the blood as disc-shaped anucleate particles
Enzymatic Removal of N- and O-glycans using PNGase F or the Protein Deglycosylation Mix
 be INSPIRED drive DISCOVERY stay GENUINE APPLICATION NOTE Enzymatic Removal of N- and O-glycans using PNGase F or the Protein Deglycosylation Mix Alicia Bielik and Paula Magnelli, New England Biolabs,
be INSPIRED drive DISCOVERY stay GENUINE APPLICATION NOTE Enzymatic Removal of N- and O-glycans using PNGase F or the Protein Deglycosylation Mix Alicia Bielik and Paula Magnelli, New England Biolabs,
Human LDL ELISA Kit. Innovative Research, Inc.
 Human LDL ELISA Kit Catalog No: IRKTAH2582 Lot No: SAMPLE INTRODUCTION Human low-density lipoprotein (LDL) transports cholesterol from the liver to tissues where it is incorporated into cell membranes.
Human LDL ELISA Kit Catalog No: IRKTAH2582 Lot No: SAMPLE INTRODUCTION Human low-density lipoprotein (LDL) transports cholesterol from the liver to tissues where it is incorporated into cell membranes.
Von Willebrand Disease. Alison Street Malaysia April 2010
 Von Willebrand Disease Alison Street Malaysia April 2010 Physiology of VWF OUTLINE Clinical presentation of VWD Classification of VWD with emphases on Type 1, 2B and 2N disease Testing for VWD Treatment
Von Willebrand Disease Alison Street Malaysia April 2010 Physiology of VWF OUTLINE Clinical presentation of VWD Classification of VWD with emphases on Type 1, 2B and 2N disease Testing for VWD Treatment
Most mammalian cells are located in tissues where they are surrounded by a complex extracellular matrix (ECM) often referred to as connective tissue.
 GLYCOSAMINOGLYCANS Most mammalian cells are located in tissues where they are surrounded by a complex extracellular matrix (ECM) often referred to as connective tissue. The ECM contains three major classes
GLYCOSAMINOGLYCANS Most mammalian cells are located in tissues where they are surrounded by a complex extracellular matrix (ECM) often referred to as connective tissue. The ECM contains three major classes
Supplementary Figure 1: Co-localization of reconstituted L-PTC and dendritic cells
 a CD11c Na + K + ATPase Na + K + ATPase CD11c x-y CD11c Na + K + ATPase Na + K + ATPase CD11c x-z c b x-y view BoNT NAPs CD11c BoNT CD11c NAPs BoNT NAPs CD11c 90 x-z view Apical Basolateral Supplementary
a CD11c Na + K + ATPase Na + K + ATPase CD11c x-y CD11c Na + K + ATPase Na + K + ATPase CD11c x-z c b x-y view BoNT NAPs CD11c BoNT CD11c NAPs BoNT NAPs CD11c 90 x-z view Apical Basolateral Supplementary
Name: Multiple choice questions. Pick the BEST answer (2 pts ea)
 Exam 1 202 Oct. 5, 1999 Multiple choice questions. Pick the BEST answer (2 pts ea) 1. The lipids of a red blood cell membrane are all a. phospholipids b. amphipathic c. glycolipids d. unsaturated 2. The
Exam 1 202 Oct. 5, 1999 Multiple choice questions. Pick the BEST answer (2 pts ea) 1. The lipids of a red blood cell membrane are all a. phospholipids b. amphipathic c. glycolipids d. unsaturated 2. The
HIV-1 p24 ELISA Pair Set Cat#: orb54951 (ELISA Manual)
 HIV-1 p24 ELISA Pair Set Cat#: orb54951 (ELISA Manual) BACKGROUND Human Immunodeficiency Virus ( HIV ) can be divided into two major types, HIV type 1 (HIV-1) and HIV type 2 (HIV-2). HIV-1 is related to
HIV-1 p24 ELISA Pair Set Cat#: orb54951 (ELISA Manual) BACKGROUND Human Immunodeficiency Virus ( HIV ) can be divided into two major types, HIV type 1 (HIV-1) and HIV type 2 (HIV-2). HIV-1 is related to
Nature Immunology: doi: /ni Supplementary Figure 1. Huwe1 has high expression in HSCs and is necessary for quiescence.
 Supplementary Figure 1 Huwe1 has high expression in HSCs and is necessary for quiescence. (a) Heat map visualizing expression of genes with a known function in ubiquitin-mediated proteolysis (KEGG: Ubiquitin
Supplementary Figure 1 Huwe1 has high expression in HSCs and is necessary for quiescence. (a) Heat map visualizing expression of genes with a known function in ubiquitin-mediated proteolysis (KEGG: Ubiquitin
(A) PCR primers (arrows) designed to distinguish wild type (P1+P2), targeted (P1+P2) and excised (P1+P3)14-
 1 Supplemental Figure Legends Figure S1. Mammary tumors of ErbB2 KI mice with 14-3-3σ ablation have elevated ErbB2 transcript levels and cell proliferation (A) PCR primers (arrows) designed to distinguish
1 Supplemental Figure Legends Figure S1. Mammary tumors of ErbB2 KI mice with 14-3-3σ ablation have elevated ErbB2 transcript levels and cell proliferation (A) PCR primers (arrows) designed to distinguish
H5N1 ( Avian Flu ) Hemagglutinin ELISA Pair Set
 H5N1 ( Avian Flu ) Hemagglutinin ELISA Pair Set Catalog Number : SEK002 To achieve the best assay results, this manual must be read carefully before using this product and the assay is run as summarized
H5N1 ( Avian Flu ) Hemagglutinin ELISA Pair Set Catalog Number : SEK002 To achieve the best assay results, this manual must be read carefully before using this product and the assay is run as summarized
Pedigree Construction Notes
 Name Date Pedigree Construction Notes GO TO à Mendelian Inheritance (http://www.uic.edu/classes/bms/bms655/lesson3.html) When human geneticists first began to publish family studies, they used a variety
Name Date Pedigree Construction Notes GO TO à Mendelian Inheritance (http://www.uic.edu/classes/bms/bms655/lesson3.html) When human geneticists first began to publish family studies, they used a variety
SUPPLEMENTARY INFORMATION
 Complete but curtailed T-cell response to very-low-affinity antigen Dietmar Zehn, Sarah Y. Lee & Michael J. Bevan Supp. Fig. 1: TCR chain usage among endogenous K b /Ova reactive T cells. C57BL/6 mice
Complete but curtailed T-cell response to very-low-affinity antigen Dietmar Zehn, Sarah Y. Lee & Michael J. Bevan Supp. Fig. 1: TCR chain usage among endogenous K b /Ova reactive T cells. C57BL/6 mice
CHAPTER 10 BLOOD GROUPS: ABO AND Rh
 CHAPTER 10 BLOOD GROUPS: ABO AND Rh The success of human blood transfusions requires compatibility for the two major blood group antigen systems, namely ABO and Rh. The ABO system is defined by two red
CHAPTER 10 BLOOD GROUPS: ABO AND Rh The success of human blood transfusions requires compatibility for the two major blood group antigen systems, namely ABO and Rh. The ABO system is defined by two red
Human Leptin ELISA Kit
 Product Manual Human Leptin ELISA Kit Catalog Numbers MET-5057 MET-5057-5 96 assays 5 x 96 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Leptin is a polypeptide hormone
Product Manual Human Leptin ELISA Kit Catalog Numbers MET-5057 MET-5057-5 96 assays 5 x 96 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Leptin is a polypeptide hormone
VI. ABO and H Blood Groups
 VI. ABO and H Blood Groups A. History of ABO System. Discovered in 900 by Karl Landsteiner and remains the most important of the blood group systems as far as the transfusion of blood is concerned. 2.
VI. ABO and H Blood Groups A. History of ABO System. Discovered in 900 by Karl Landsteiner and remains the most important of the blood group systems as far as the transfusion of blood is concerned. 2.
Oligosaccharide structure determination of glycoconjugates using lectins
 J. Biosci., Vol. 11, Numbers 1 4, March 1987, pp. 41 46. Printed in India. Oligosaccharide structure determination of glycoconjugates using lectins DEBKUMAR BASU*, JYOTI V. NAIR and P. S. APPUKUTTAN Neurochemistry
J. Biosci., Vol. 11, Numbers 1 4, March 1987, pp. 41 46. Printed in India. Oligosaccharide structure determination of glycoconjugates using lectins DEBKUMAR BASU*, JYOTI V. NAIR and P. S. APPUKUTTAN Neurochemistry
Congenital bleeding disorders
 Congenital bleeding disorders Overview Factor VIII von Willebrand Factor Complex factor VIII von Willebrand factor (vwf) complex circulate as a complex + factor IX intrinsic pathway Platelets bind via
Congenital bleeding disorders Overview Factor VIII von Willebrand Factor Complex factor VIII von Willebrand factor (vwf) complex circulate as a complex + factor IX intrinsic pathway Platelets bind via
Insulin Resistance. Biol 405 Molecular Medicine
 Insulin Resistance Biol 405 Molecular Medicine Insulin resistance: a subnormal biological response to insulin. Defects of either insulin secretion or insulin action can cause diabetes mellitus. Insulin-dependent
Insulin Resistance Biol 405 Molecular Medicine Insulin resistance: a subnormal biological response to insulin. Defects of either insulin secretion or insulin action can cause diabetes mellitus. Insulin-dependent
29th Annual Meeting of the Glomerular Disease Collaborative Network
 29th Annual Meeting of the Glomerular Disease Collaborative Network Updates on the Pathogenesis IgA Nephropathy and IgA Vasculitis (HSP) J. Charles Jennette, M.D. Brinkhous Distinguished Professor and
29th Annual Meeting of the Glomerular Disease Collaborative Network Updates on the Pathogenesis IgA Nephropathy and IgA Vasculitis (HSP) J. Charles Jennette, M.D. Brinkhous Distinguished Professor and
Bleeding and Thrombotic Disorders. Kristine Krafts, M.D.
 Bleeding and Thrombotic Disorders Kristine Krafts, M.D. Bleeding and Thrombotic Disorders Bleeding disorders von Willebrand disease Hemophilia A and B DIC TTP/HUS ITP Thrombotic disorders Factor V Leiden
Bleeding and Thrombotic Disorders Kristine Krafts, M.D. Bleeding and Thrombotic Disorders Bleeding disorders von Willebrand disease Hemophilia A and B DIC TTP/HUS ITP Thrombotic disorders Factor V Leiden
ACTG Laboratory Technologist Committee Revised Version 2.0 ACTG Lab Man Coulter HIV-1 p24 ELISA May 21, 2004
 Coulter HIV p24 1. PRINCIPLE The Human Immunodeficiency Virus Type 1 (HIV-1) is recognized as the etiologic agent of acquired immunodeficiency syndrome (AIDS). The virus is transmitted by sexual contact,
Coulter HIV p24 1. PRINCIPLE The Human Immunodeficiency Virus Type 1 (HIV-1) is recognized as the etiologic agent of acquired immunodeficiency syndrome (AIDS). The virus is transmitted by sexual contact,
1) DNA unzips - hydrogen bonds between base pairs are broken by special enzymes.
 Biology 12 Cell Cycle To divide, a cell must complete several important tasks: it must grow, during which it performs protein synthesis (G1 phase) replicate its genetic material /DNA (S phase), and physically
Biology 12 Cell Cycle To divide, a cell must complete several important tasks: it must grow, during which it performs protein synthesis (G1 phase) replicate its genetic material /DNA (S phase), and physically
APPENDIX -1 LIST OF TABLES. 01 The blood group systems Blood group collections
 APPENDIX -1 LIST OF TABLES Table 01 The blood group systems. 014 005 02 Blood group collections. 016 008 03 Low frequency antigens: the 700 series. 017 009 04 Frequencies of low frequency antigens. 017
APPENDIX -1 LIST OF TABLES Table 01 The blood group systems. 014 005 02 Blood group collections. 016 008 03 Low frequency antigens: the 700 series. 017 009 04 Frequencies of low frequency antigens. 017
Suppl Video: Tumor cells (green) and monocytes (white) are seeded on a confluent endothelial
 Supplementary Information Häuselmann et al. Monocyte induction of E-selectin-mediated endothelial activation releases VE-cadherin junctions to promote tumor cell extravasation in the metastasis cascade
Supplementary Information Häuselmann et al. Monocyte induction of E-selectin-mediated endothelial activation releases VE-cadherin junctions to promote tumor cell extravasation in the metastasis cascade
This slide belongs to iron lecture and it is to clarify the iron cycle in the body and the effect of hypoxia on erythropoitein secretion
 This slide belongs to iron lecture and it is to clarify the iron cycle in the body and the effect of hypoxia on erythropoitein secretion Topics of today lectures: Hemostasis Meaning of hemostasis Mechanisms
This slide belongs to iron lecture and it is to clarify the iron cycle in the body and the effect of hypoxia on erythropoitein secretion Topics of today lectures: Hemostasis Meaning of hemostasis Mechanisms
The Diagnosis of VWD Interpreting laboratory testing for a complex genetic disorder
 The Diagnosis of VWD Interpreting laboratory testing for a complex genetic disorder David Lillicrap Department of Pathology and Molecular Medicine Queen's University, Kingston, Canada Bangkok, November
The Diagnosis of VWD Interpreting laboratory testing for a complex genetic disorder David Lillicrap Department of Pathology and Molecular Medicine Queen's University, Kingston, Canada Bangkok, November
The major histocompatibility complex (MHC) is a group of genes that governs tumor and tissue transplantation between individuals of a species.
 Immunology Dr. John J. Haddad Chapter 7 Major Histocompatibility Complex The major histocompatibility complex (MHC) is a group of genes that governs tumor and tissue transplantation between individuals
Immunology Dr. John J. Haddad Chapter 7 Major Histocompatibility Complex The major histocompatibility complex (MHC) is a group of genes that governs tumor and tissue transplantation between individuals
Balancing intestinal and systemic inflammation through cell type-specific expression of
 Supplementary Information Balancing intestinal and systemic inflammation through cell type-specific expression of the aryl hydrocarbon receptor repressor Olga Brandstätter 1,2,6, Oliver Schanz 1,6, Julia
Supplementary Information Balancing intestinal and systemic inflammation through cell type-specific expression of the aryl hydrocarbon receptor repressor Olga Brandstätter 1,2,6, Oliver Schanz 1,6, Julia
Supplemental Figure 1 ELISA scheme to measure plasma total, mature and furin-cleaved
 1 Supplemental Figure Legends Supplemental Figure 1 ELISA scheme to measure plasma total, mature and furin-cleaved PCSK9 concentrations. 4 Plasma mature and furin-cleaved PCSK9s were measured by a sandwich
1 Supplemental Figure Legends Supplemental Figure 1 ELISA scheme to measure plasma total, mature and furin-cleaved PCSK9 concentrations. 4 Plasma mature and furin-cleaved PCSK9s were measured by a sandwich
Supporting Information Table of Contents
 Supporting Information Table of Contents Supporting Information Figure 1 Page 2 Supporting Information Figure 2 Page 4 Supporting Information Figure 3 Page 5 Supporting Information Figure 4 Page 6 Supporting
Supporting Information Table of Contents Supporting Information Figure 1 Page 2 Supporting Information Figure 2 Page 4 Supporting Information Figure 3 Page 5 Supporting Information Figure 4 Page 6 Supporting
MRC-Holland MLPA. Description version 19;
 SALSA MLPA probemix P6-B2 SMA Lot B2-712, B2-312, B2-111, B2-511: As compared to the previous version B1 (lot B1-11), the 88 and 96 nt DNA Denaturation control fragments have been replaced (QDX2). SPINAL
SALSA MLPA probemix P6-B2 SMA Lot B2-712, B2-312, B2-111, B2-511: As compared to the previous version B1 (lot B1-11), the 88 and 96 nt DNA Denaturation control fragments have been replaced (QDX2). SPINAL
Thyroid Function. Thyroglobulin Analyte Information
 Thyroid Function Thyroglobulin Analyte Information - 1-2011-01-11 Thyroglobulin Introduction Thyroglobulin (Tg) is a big dimeric protein consisting of two identical subunits. It has 2,748 amino acids in
Thyroid Function Thyroglobulin Analyte Information - 1-2011-01-11 Thyroglobulin Introduction Thyroglobulin (Tg) is a big dimeric protein consisting of two identical subunits. It has 2,748 amino acids in
Molecular Cell Biology Problem Drill 16: Intracellular Compartment and Protein Sorting
 Molecular Cell Biology Problem Drill 16: Intracellular Compartment and Protein Sorting Question No. 1 of 10 Question 1. Which of the following statements about the nucleus is correct? Question #01 A. The
Molecular Cell Biology Problem Drill 16: Intracellular Compartment and Protein Sorting Question No. 1 of 10 Question 1. Which of the following statements about the nucleus is correct? Question #01 A. The
The ABO and Rh system. Dr U. La Rocca 03 th Novembre 2017
 The ABO and Rh system Dr U. La Rocca 03 th Novembre 2017 Main learning endpoints! ü Chemical structure ü Inheritance ü AB0 and Rh antibodies and their importance in transfusion ü Principles of AB0 and
The ABO and Rh system Dr U. La Rocca 03 th Novembre 2017 Main learning endpoints! ü Chemical structure ü Inheritance ü AB0 and Rh antibodies and their importance in transfusion ü Principles of AB0 and
Human Obestatin ELISA
 K-ASSAY Human Obestatin ELISA For the quantitative determination of obestatin in human serum and plasma Cat. No. KT-495 For Research Use Only. 1 Rev. 081309 K-ASSAY PRODUCT INFORMATION Human Obestatin
K-ASSAY Human Obestatin ELISA For the quantitative determination of obestatin in human serum and plasma Cat. No. KT-495 For Research Use Only. 1 Rev. 081309 K-ASSAY PRODUCT INFORMATION Human Obestatin
Glycosaminoglycans: Anionic polysaccharide chains made of repeating disaccharide units
 Glycosaminoglycans: Anionic polysaccharide chains made of repeating disaccharide units Glycosaminoglycans present on the animal cell surface and in the extracellular matrix. Glycoseaminoglycans (mucopolysaccharides)
Glycosaminoglycans: Anionic polysaccharide chains made of repeating disaccharide units Glycosaminoglycans present on the animal cell surface and in the extracellular matrix. Glycoseaminoglycans (mucopolysaccharides)
SALSA MLPA KIT P060-B2 SMA
 SALSA MLPA KIT P6-B2 SMA Lot 111, 511: As compared to the previous version B1 (lot 11), the 88 and 96 nt DNA Denaturation control fragments have been replaced (QDX2). Please note that, in contrast to the
SALSA MLPA KIT P6-B2 SMA Lot 111, 511: As compared to the previous version B1 (lot 11), the 88 and 96 nt DNA Denaturation control fragments have been replaced (QDX2). Please note that, in contrast to the
GLP-2 ELISA. For the quantitative determination of GLP-2 in human serum and plasma samples.
 GLP-2 ELISA For the quantitative determination of GLP-2 in human serum and plasma samples. For Research Use Only. Not For Use In Diagnostic Procedures. Catalog Number: 48-GP2HU-E01.1 Size: 96 wells Version:
GLP-2 ELISA For the quantitative determination of GLP-2 in human serum and plasma samples. For Research Use Only. Not For Use In Diagnostic Procedures. Catalog Number: 48-GP2HU-E01.1 Size: 96 wells Version:
human Total Cathepsin B Catalog Number: DY2176
 human Total Cathepsin B Catalog Number: DY2176 This DuoSet ELISA Development kit contains the basic components required for the development of sandwich ELISAs to measure natural and recombinant human Total
human Total Cathepsin B Catalog Number: DY2176 This DuoSet ELISA Development kit contains the basic components required for the development of sandwich ELISAs to measure natural and recombinant human Total
Probe. Hind III Q,!?R'!! /0!!!!D1"?R'! vector. Homologous recombination
 Supple-Zhang Page 1 Wild-type locus Targeting construct Targeted allele Exon Exon3 Exon Probe P1 P P3 FRT FRT loxp loxp neo vector amh I Homologous recombination neo P1 P P3 FLPe recombination Q,!?R'!!
Supple-Zhang Page 1 Wild-type locus Targeting construct Targeted allele Exon Exon3 Exon Probe P1 P P3 FRT FRT loxp loxp neo vector amh I Homologous recombination neo P1 P P3 FLPe recombination Q,!?R'!!
Biochemistry: A Short Course
 Tymoczko Berg Stryer Biochemistry: A Short Course Second Edition CHAPTER 10 Carbohydrates 2013 W. H. Freeman and Company Chapter 10 Outline Monosaccharides are aldehydes or ketones that contain two or
Tymoczko Berg Stryer Biochemistry: A Short Course Second Edition CHAPTER 10 Carbohydrates 2013 W. H. Freeman and Company Chapter 10 Outline Monosaccharides are aldehydes or ketones that contain two or
BIOL2005 WORKSHEET 2008
 BIOL2005 WORKSHEET 2008 Answer all 6 questions in the space provided using additional sheets where necessary. Hand your completed answers in to the Biology office by 3 p.m. Friday 8th February. 1. Your
BIOL2005 WORKSHEET 2008 Answer all 6 questions in the space provided using additional sheets where necessary. Hand your completed answers in to the Biology office by 3 p.m. Friday 8th February. 1. Your
Supporting Online Material for
 www.sciencemag.org/cgi/content/full/1175194/dc1 Supporting Online Material for A Vital Role for Interleukin-21 in the Control of a Chronic Viral Infection John S. Yi, Ming Du, Allan J. Zajac* *To whom
www.sciencemag.org/cgi/content/full/1175194/dc1 Supporting Online Material for A Vital Role for Interleukin-21 in the Control of a Chronic Viral Infection John S. Yi, Ming Du, Allan J. Zajac* *To whom
Y. Helen Zhang, MD Andy Nguyen, MD 10/28/2012
 Y. Helen Zhang, MD Andy Nguyen, MD 10/28/2012 Clinical History Patient: 23-year-old female Clinical course: status-post cholecystectomy, complicated by retained common bile duct stones. Following three
Y. Helen Zhang, MD Andy Nguyen, MD 10/28/2012 Clinical History Patient: 23-year-old female Clinical course: status-post cholecystectomy, complicated by retained common bile duct stones. Following three
Mitochondrial Trifunctional Protein (TFP) Protein Quantity Microplate Assay Kit
 PROTOCOL Mitochondrial Trifunctional Protein (TFP) Protein Quantity Microplate Assay Kit DESCRIPTION Mitochondrial Trifunctional Protein (TFP) Protein Quantity Microplate Assay Kit Sufficient materials
PROTOCOL Mitochondrial Trifunctional Protein (TFP) Protein Quantity Microplate Assay Kit DESCRIPTION Mitochondrial Trifunctional Protein (TFP) Protein Quantity Microplate Assay Kit Sufficient materials
von Willebrand Disease
 von Willebrand Disease Jeremy Robertson Paediatric Haematologist Royal Children s s Hospital & Pathology Queensland Foglo,, April 1924: the journey begins Oskar and Augusta sail to Helsinki... ...to o
von Willebrand Disease Jeremy Robertson Paediatric Haematologist Royal Children s s Hospital & Pathology Queensland Foglo,, April 1924: the journey begins Oskar and Augusta sail to Helsinki... ...to o
Instructions for Use. APO-AB Annexin V-Biotin Apoptosis Detection Kit 100 tests
 3URGXFW,QIRUPDWLRQ Sigma TACS Annexin V Apoptosis Detection Kits Instructions for Use APO-AB Annexin V-Biotin Apoptosis Detection Kit 100 tests For Research Use Only. Not for use in diagnostic procedures.
3URGXFW,QIRUPDWLRQ Sigma TACS Annexin V Apoptosis Detection Kits Instructions for Use APO-AB Annexin V-Biotin Apoptosis Detection Kit 100 tests For Research Use Only. Not for use in diagnostic procedures.
Mouse Myeloperoxidase/MPO ELISA Kit
 OriGene Technologies, Inc 9620 Medical Center Dr., Suite 200, Rockville, MD 20850 Phone: 1.888.267.4436 Fax: 301-340-9254 Email: techsupport@origene.com Web: Mouse Myeloperoxidase/MPO ELISA Kit Catalog
OriGene Technologies, Inc 9620 Medical Center Dr., Suite 200, Rockville, MD 20850 Phone: 1.888.267.4436 Fax: 301-340-9254 Email: techsupport@origene.com Web: Mouse Myeloperoxidase/MPO ELISA Kit Catalog
Mercodia Glucagon ELISA
 Mercodia Glucagon ELISA Directions for Use 10-1271-01 REAGENTS FOR 96 DETERMINATIONS For Research Use Only Manufactured by Mercodia AB, Sylveniusgatan 8A, SE-754 50 Uppsala, Sweden EXPLANATION OF SYMBOLS
Mercodia Glucagon ELISA Directions for Use 10-1271-01 REAGENTS FOR 96 DETERMINATIONS For Research Use Only Manufactured by Mercodia AB, Sylveniusgatan 8A, SE-754 50 Uppsala, Sweden EXPLANATION OF SYMBOLS
Single Gene (Monogenic) Disorders. Mendelian Inheritance: Definitions. Mendelian Inheritance: Definitions
 Single Gene (Monogenic) Disorders Mendelian Inheritance: Definitions A genetic locus is a specific position or location on a chromosome. Frequently, locus is used to refer to a specific gene. Alleles are
Single Gene (Monogenic) Disorders Mendelian Inheritance: Definitions A genetic locus is a specific position or location on a chromosome. Frequently, locus is used to refer to a specific gene. Alleles are
Mouse TrkB ELISA Kit
 Mouse TrkB ELISA Kit CATALOG NO: IRKTAH5472 LOT NO: SAMPLE INTENDED USE For quantitative detection of mouse TrkB in cell culture supernates, cell lysates and tissue homogenates. BACKGROUND TrkB receptor
Mouse TrkB ELISA Kit CATALOG NO: IRKTAH5472 LOT NO: SAMPLE INTENDED USE For quantitative detection of mouse TrkB in cell culture supernates, cell lysates and tissue homogenates. BACKGROUND TrkB receptor
Human Leptin ELISA Kit
 OriGene Technologies, Inc 9620 Medical Center Dr., Suite 200, Rockville, MD 20850 Phone: 1.888.267.4436 Fax: 301-340-9254 Email: techsupport@origene.com Web: www.origene.com Human Leptin ELISA Kit Catalog
OriGene Technologies, Inc 9620 Medical Center Dr., Suite 200, Rockville, MD 20850 Phone: 1.888.267.4436 Fax: 301-340-9254 Email: techsupport@origene.com Web: www.origene.com Human Leptin ELISA Kit Catalog
GLP-2 (Rat) ELISA. For the quantitative determination of glucagon-like peptide 2 (GLP-2) in rat serum and plasma
 GLP-2 (Rat) ELISA For the quantitative determination of glucagon-like peptide 2 (GLP-2) in rat serum and plasma For Research Use Only. Not For Use In Diagnostic Procedures. Catalog Number: 48-GP2RT-E01
GLP-2 (Rat) ELISA For the quantitative determination of glucagon-like peptide 2 (GLP-2) in rat serum and plasma For Research Use Only. Not For Use In Diagnostic Procedures. Catalog Number: 48-GP2RT-E01
