Domain 2: Matter. 2.1: Organisms must exchange matter with the environment to grow, reproduce and maintain organization. (EK2.A.3) 1.
|
|
|
- Ashlie Wiggins
- 6 years ago
- Views:
Transcription
1 Domain 2: Matter 2.1: Organisms must exchange matter with the environment to grow, reproduce and maintain organization. (EK2.A.3) 1. Matter exchange The atoms in living systems are common on this planet. Living systems are mostly made of 6 elements and a handful of other trace elements. Living systems cycle matter from the environment, turning it in to biologically useful molecules before returning it back to the environment through life processes. Carbon: Carbon is the major structural atom in all organic molecules. The major nonliving source of carbon is the atmosphere (as CO 2 ). Carbon is incorporated in to producers through photosynthesis and from there in to living systems through the food chain, and returned back to the environment through cellular respiration, and decomposition. Oxygen: Oxygen is also found in most organic molecules. The major non-living source of oxygen is the atmosphere (as O 2 ) and water (also the major non-living source of hydrogen). Oxygen is incorporated in to cellular respiration and through the food chain. Oxygen is returned back to the environment through photosynthesis. 47
2 Nitrogen: Nitrogen is found in all proteins and nucleic acids (present in all living systems). The major non-living source of Nitrogen is the atmosphere (as N 2 ). Nitrogen is incorporated in to the food chain through the action of nitrogen-fixing bacteria, which convert N 2 in to molecules that can be used by producers (NO - 3 ), and from producers through the food chain. Nitrogen is returned back to the environment through decomposition and the action of denitrifying bacteria. Phosphorous and Sulfur: Phosphorous is found in all nucleic acids. Phosphate groups are used to quickly store and release free energy in cells. Sulfur is found in all proteins. The major non-living source of these elements is rocks. Weathering processes release them in to the soil/water, where they are available for producers to incorporate and then move through the food chain. Decomposition returns them back to the environment. Hydrogen: Hydrogen is a major component of all organic molecules. It is the most common atom in the Universe. It enters biological systems largely bonded to oxygen in water, and is returned to the environment by decomposition and water release. 2. Properties of Water Water is a major component of all living systems. Water molecules occupy most of a cell s volume. 48
3 Water has major properties that living systems require. These properties are due to water s polarity (unequal sharing of electrons, leading to a partially positive and partially negative charge). Hydrogen Bonds: the polarity of water results in attractive forces between the oxygen and hydrogen atoms of neighboring water molecules. This makes water cohesive (attracted to itself) and adhesive (attractive to anything else that has positive or negative charges). Uses: Transpiration pull, dissolving of substances. Temperature buffering: The presence of hydrogen bonds requires more energy to be absorbed or released by a given amount of water to change the temperature of that water. This is known as the specific heat of a substance. Water has a very high specific heat. Water can absorb or release a comparatively large amount of energy before its temperature changes. Water will keep the environment warmer when the air cools (because it has a larger store of energy to release), and keep the environment cooler when the air warms (because it can absorb a larger amount of energy without changing temperature). Uses: Insulating the ocean. Keeping living systems within homeostatic temperature ranges. 3. MATH Skills: ph 49
4 Water dissociates. A water molecule can be pulled apart into a proton (H + ) and a Hydroxide ion (OH - ) by other water molecules. This happens ~ once for every 10,000,000 (10 7 ) water molecules. The products of the H + and OH - ion concentrations in an aqueous solution is a physical constant (10-14 ). As the concentration of one ion increases, the other must decrease. In pure water, the concentrations of H + and OH - are each 10-7 (one in 10 million). Acids: substances that increase the amount of H + ions in an aqueous solution. This will cause the concentration of OH - ions to decrease. Bases: Substances that decrease the amount of H + ions in an aqueous solution. This will cause the concentration of H + ions to increase. ph: a measurement of the concentration of H+ ions in a solution. ph = - log [H + ] This is a complicated way of saying that we can take the exponent of the ph concentration and negate it (make it positive) to state the ph. Acids have a ph lower than 7. Bases have a ph higher than 7. Each whole number on the ph represents a power of 10. A solution with a ph of 5 has a ph that is 100 times more acidic than a solution with a ph of Constraints on Cell Size Living systems are constrained by the need to exchange matter with the environment. 50
5 Cellular life has an upper and lower limit on the size a cell can be: Lower limit: Constrained by the need to have a certain amount of matter inside the cell in order to keep functioning. Upper limit: Constrained by efficiency. Surface area: Volume ratio- as a three-dimensonal object s volume increases, so does its surface area. However, volume increases as a cubic function of size, while surface area increases as a squared function. As volume increases, the efficiency of a cell s material exchange with the environment decreases. Surface Area Increase Adaptations: A variety of structures have evolved to maximize surface area in cells and increase material exchange efficiency Examples: root hairs: Plant root cells Microvilli: Vertebrate intestines 5. MATH Skills: Surface Area:Volume Ratio Being able to calculate the surface area and volume of an object, if provided with its physical dimensions. Use the formula sheet to find relevant equations. 51
6 Sample problem: Determine the relative efficiency of material exchange for a spherical cell with a radius of 10 µm, and a cubic cell with a side length of 10µm. Determine the surface area:volume ratio Surface Area of cube: 6xarea = 6 x 100um 2 = 600um 2 Volume of cube: l*w*h = 10 x 10 x 10 = 1000um 3 SA:V ratio of cube: 600:1000 =.6 Surface Area of sphere: 4 pi r 2 = 4 x 3.14 x 100 = ~1257 um 2 Volume of a cube: 4/3 pi r 3 = 4/3 * 3.14 * 1000 = ~4189 um 3 SA:V ratio of sphere: 1257:4189 = ~.3 Divide the surface area by the volume. Smaller numbers mean more volume per unit of surface area (less efficiency in transport. 2.2: The subcomponents of biological molecules and their sequence determine the properties of that molecule. (EK4.A.1) 1. Biological Molecules Living systems are made of four major types of Macromolecule: Carbohydrates- Sugars: function in short-term energy storage and structural support. Lipids- Fats, oils, waxes: function in long-term energy storage, cell membrane structure, and cell signaling. 52
7 Proteins- function in all cellular processes. Nucleic Acids- DNA and RNA: function in information storage and expression of that information by determining protein sequences. All macromolecules are synthesized via condensation reactions ( dehydration synthesis - the removal of water) and decomposed via hydrolysis reactions (the addition of water). Except for lipids, Macromolecules are polymers, made of repeating subunits (monomers). Carbohydrates: Monomer: Monosaccharide \ Polymer: polysaccharide Monosaccharides exist as ring structures. Lipids: Steroids, triglycerides, and phospholipids. Proteins: Monomer: Amino acid \ Polymer: polypeptide There are 20 different types of amino acids used in biological systems. They all have the same Backbone structure, but differ based on the composition of the variable group that is bonded to the central carbon. The bond between two amino acids is referred to as a peptide bond. Nucleic Acids: Monomer: nucleotides \ Polymer: nucleic acid 53
8 There are two types of nucleic acids in biological systems (DNA and RNA). Each one is made of four types of nucleotides, which differ from each other based on the type of nitrogenous base that they contain (DNA: ACTG / RNA: ACUG) 2.3: Variation in molecular units provides cells with a wider range of functions. (EK4.C.1) 1. Variation in Biological Molecules- Carbohydrates and Lipids The structure of carbohydrates and lipids determines their function: Ex: Cellulose vs. Amylose ( starch ): Both are polysaccharide polymers of glucose monomers, but they vary in the connections between monomers. As a result, cellulose is not easily digested, while amylose is very easily digested. Ex: Lipid membrane composition: Cells will vary the lipid composition of their membranes in order to respond to changes in temperature. One examplechanges in cholesterol concentration to resist increasing and decreasing membrane fluidity. 2. Variation in Biological Molecules- Proteins The structure of proteins determines their function: Proteins are functionally responsible for all cellular processes. They have a wide and diverse variety of functions, which are determined by their structure. Protein structure is discussed at four levels of organization: 54
9 Primary structure: the sequence of amino acids in a polypeptide. This is determined by DNA. Each amino acid is joined to the next via a peptide bond. Secondary structure: Regular 3D structures that arise in polypeptide chains due to hydrogen bonding between the oxygen, nitrogen, and hydrogen of neighboring amino acids. Since all polypeptides are made of amino acids, these secondary structures are found in all proteins. Tertiary structure: The unique 3D structure ( conformation ) of a particular polypeptide chain, due to the variety of interactions between the variable groups of the amino acids in the polypeptide and the influence of the environment that the polypeptide chain is in. Quarternary structure: Refers to the overall 3D shape of any protein that is composed of more than one polypeptide chain. Not all proteins have this level of structure. Ex: Adult hemoglobin vs. fetal hemoglobin: Differences in the polypeptide chains that comprise fetal hemoglobin give the molecule a higher binding affinity to oxygen, which is necessary since the fetus needs to get its oxygen from its mothers hemoglobin. Ex: Antibody structure: Differences in the variable regions of the proteins that comprise antibodies makes it possible to create millions of different possible antibodies. 55
10 Denaturation: The disruption of the structure of a macromolecule leading to a disruption of its function. Caused by changes to the environment of the molecule. 3. Variation in Biological Molecules- Nucleic Acids Genetic variation leads to molecular variation. DNA sequence determines protein sequence. Protein sequence contributes to protein structure. Protein structure determines protein function. Protein function determines cellular function. Cellular function determines organism function. A change in DNA sequence can affect all levels of function in a living system. Ex: Sickle cell anemia- one nucleotide change leads to one amino acid change. This change alters the function of hemoglobin, and leads to major disease effects in the organism. New functions can arise via genetic duplications. The duplication of genetic information allows for new variations and adaptations. Ex. Antifreeze gene in fish- allows fish to live in freezing temperatures. Evolved from pre-existing genes. 2.4: Cell membranes are selectively permeable due to their structure. (EK2.B.1) 56
11 1. Cell Membrane Structure Cell membrane structure and function: The cell membrane is the boundary between the cell and the environment. It also allows the cell to control which substances pass in to and out of the cell. The cell membrane is composed of a phospholipid bilayer with embedded proteins (the Fluid mosaic model). Phospholipid structure causes them to have a hydrophilic phosphate group head, and hydrophobic fatty acid tails. This leads to spontaneous arrangements in water with the phosphate heads pointing toward the water molecules and the fatty acid tails pointing away. One such arrangement is the bi-layer seen in cell membranes. The membrane proteins have a variety of functions: transport, cell-cell contact and recognition, anchorage, enzymatic functions, and others. Selective Permeability: Selective permeability is controlled through control of the proteins present in the cell membrane. Only small, non-polar molecules are able to move through the phospholipid bi-layer. All other substances must move through proteins Cell Wall structure and function: Many cells have a cell wall external to the cell membrane (animals and animal-like cells are the only exception). The cell wall is a structure made of structural polysaccharides (cellulose in plants and plant-like cells) that provides structural support, but no dynamic control of transport. 57
12 2.5: Growth and dynamic homeostasis are maintained by the constant movement of molecules across membranes. (EK2.B.2) 1. Mechanisms of Cellular Transport Passive Transport: Refers to the diffusion of material, moving from an area of high concentration to an area of low concentration across a membrane. No energy is required by the cell to enable this process. Simple/Facilitated Diffusion: Simple diffusion: movement through the bi-layer. Only small non-polar molecules can do this. Facilitated diffusion: movement through a protein channel. Any molecule that is not small and nonpolar must diffuse through a protein pore in the membrane. Ex: aquaporins. Active Transport: Refers to the movement of material from an area of low concentration to an area of high concentration. This requires energy, in order to work against the natural tendency for molecules to diffuse. Cells always use 58
13 Ex: Na/K pump: In order to move sodium out of the cell and potassium in to the cell to establish specific concentrations. ATP is used to provide the energy needed to modulate the shape of the pump proteins. Bulk Transport: The movement of large molecules in to or out of the cell. Requires the use of a vesicle. Endocytosis: Movement of large particles in to the cell. Exocytosis: Movement of large particles out of the cell. 2. Analyzing Transport Tonicity: A measurement of the relative concentrations of solute and solvent in two different solutions. Used to be able to determine how a cell (its internal solution) will respond when placed in to different aqueous environments (the external solution). Three types of tonicity relationships: Hypertonic: The solution has more solute (and less solvent) than the one being compared to. Hypotonic: The solution has less solute (and more solvent) than the one being compared to. 59
14 Isotonic: The solution has the same amount of solute (and solvent) as the one being compared to. Tonicity relationships and effects on cells: Without the expenditure of energy, material will always diffuse from high concentration to low concentration, if it is able to move across the membrane. Solute will always move from hypertonic solutions to hypotonic solutions. Solvent will always move from hypotonic solutions to hypertonic solutions. As long as it is able to. Cells placed in hypertonic solutions will gain solute and lose solvent (water). Plasmolysis: The loss of water Cells placed in hypotonic solutions will lose solute and gain solvent (water). Lysis: The bursting of the cell membrane. Animal-like cells are adapted to exist in isotonic solutions. Plant-like cells are adapted to exist in hypotonic solutions (the presence of the cell wall prevents lysis). 3. MATH Skills: Water Potential and Solute Potential 60
15 Water Potential: A measurement of how likely water is to diffuse ( osmosis ) in to an area. Water will move from areas of higher water potential to areas of lower water potential. Pure water is assigned a water potential of 0 (no net diffusion) Water potential depends on several different factors, but we only focus on the pressure difference (the pressure potential ) and the tonicity (the solute potential ) as they are the only significant factors in biological systems. Water potential equation: Ψ Water Potential Ψ p pressure potential Ψ s solute potential The typical units of water potential are bars aka torr aka mmhg in a barometer. In any open air system with no active pressure generation, the pressure potential will be zero, and the water potential will depend entirely upon the solute potential. This is the typical case when investigating diffusion and osmosis in the lab. Solute potential calculation 61
16 i = ionization constant for the solute (1.0 for sucrose, 2.0 for NaCl, etc.) C = molar concentration of the solute R= pressure constant liter bars/mole K T= temperature in Kelvin (C + 273) Sample Problem: Determine which of the following solutions will gain the most water if placed in to a sample of pure water in a piece of dialysis tubing at the temperature indicated. Assume all samples are at atmospheric pressure: Solution: Solute: Tonicity: Temperature A Sucrose 2M 298K B NaCl 1M 290K C Glucose 1M 300K Water Potential Solution A = -1.0 * 2M *.0831 * 298K = ~ bars Water Potential Solution B = -2.0 * 1M *.0831 * 290K = ~ bars Water Potential Solution C = -1.0 * 1M *.0831 * 300K = ~ bars Expect C to gain the most water, since water potential is most negative. 2.6: Eukaryotic cells maintain internal membranes that partition the cell into specialized regions. (EK2.B.3) 62
17 1. Cellular Compartmentalization Compartmentalization: Increased compartmentalization allows for increased control of life processes, and increased efficiency of those processes, as different components of the cell can function in different environmental conditions. Different areas of the cell can be specialized to perform different processes. Eukaryotes vs. Prokaryotes: The major difference between the organization of eukaryotes and prokaryotes is the amount of internal compartmentalization. Prokaryotes: The vast majority of life on earth. Possess no internal compartments ( organelles ). Unicellular and smaller than eukaryotic cells. Eukaryotes: Contain many different internal compartments separated from the rest of the cell by membrane ( organelles ). Most notably, the nucleus, but many others. Two major types: plant-like and animal-like, according to how they process energy. Most eukaryotes are unicellular, too, but all multicellular organisms (fungi, animals, and plants) are eukaryotes. Plant-like vs. Animal-like differences: Plant-like cells contain chloroplasts (for photosynthesis), a large central vacuole, and an external cell wall. Animals do not have any of these organelles. 63
18 2. Major Eukaryotic Organelles Major Eukaryotic Organelles: Nuclear membrane: Surrounds and separates the DNA from the rest of the cell. Contains pores to allow material to enter and exit the nucleus and interact with the DNA. Golgi Apparatus: A series of flattened membranous disks. Receive vesicles of membrane and protein from the endoplasmic reticulum and modify those proteins prior to routing them to their final destinations. Also the site of polysaccharide production for the cell. Endoplasmic Reticulum: A series of membranous channels that run throughout the cell. Responsible for producing all membrane used by the cell and lot of roles in intercellular transport. Divided in to two parts: smooth and rough Rough ER: Named because it is covered in bound ribosomes. The ribosomes are producing proteins that will be embedded in membrane or exported from the cell. Smooth ER: Not covered in ribosomes. Involved in toxin detoxification. Mitochondria: The site of aerobic cellular respiration. A double-membrane structure, with a highly-folded inner membrane (a surface area adaptation). Chloroplasts: The site of photosynthesis. A double-membrane structure with a highly-folded inner membrane (a surface area adaptation). 64
19 2.7: The structure and function of subcellular components, and their interactions, provide essential cellular processes. (EK4.A.2) 1. Organelle structure and function- information processing Ribosome structure and function: All cells contain ribosomes. Ribosomes are made of two subunits of RNA and protein. They are able to assemble and disassemble as required by the cell. In eukaryotes they are able to associate and disassociate with the endoplasmic reticulum. Endomembrane System: The cellular system by which the information in DNA is expressed and incorporated in to cellular processes: Nucleus! ER! Golgi! final destination (membrane or export). 2. Organelle structure and function- matter and energy processing Mitochondria: Mitochondria have a double membrane which allows for separation of different processes that take place in the mitochondria. The highly folded inner membrane (the cristae ) contains many copies of the enzymes needed to produce ATP by the cell, with maximized surface area. Chloroplasts: Chloroplasts have a double outer membrane with inner membranous stacks called thylakoids. 65
20 The membrane of the thylakoid contains many copies of the enzymes and chlorophyll needed to produce chemical energy from solar radiation during the first part of photosynthesis (the light reactions ). The inside of the thylakoids (the stroma ) contain the enzymes needed to produce chemical compounds during the second part of photosynthesis (Carbon fixation). Lysosomes: Lysosomes are membrane-enclosed sacs that contain collections of digestive, hydrolytic enzymes. Lysosomes serve roles in digestion of molecules, recycling a cell s damaged components, and programmed cell death. Vacuoles: A vacuole is a membrane-bound sac that stores material. Plants have a large central vacuole that increases the cells surface area: volume ratio by decreasing the active volume of the cell. 66
2.1: Organisms must exchange matter with the environment to grow, reproduce and maintain organization. 1. MATTER EXCHANGE
 Domain 2: Matter 2.1: Organisms must exchange matter with the environment to grow, reproduce and maintain organization. 1. MATTER EXCHANGE Matter Cycles The atoms in living systems are common on this planet.
Domain 2: Matter 2.1: Organisms must exchange matter with the environment to grow, reproduce and maintain organization. 1. MATTER EXCHANGE Matter Cycles The atoms in living systems are common on this planet.
CELLS and TRANSPORT Student Packet SUMMARY CELL MEMBRANES ARE SELECTIVELY PERMEABLE DUE TO THEIR STRUCTURE Hydrophilic head
 CELLS and TRANSPORT Student Packet SUMMARY CELL MEMBRANES ARE SELECTIVELY PERMEABLE DUE TO THEIR STRUCTURE Hydrophilic head Hydrophobic tail Hydrophobic regions of protein Hydrophilic regions of protein
CELLS and TRANSPORT Student Packet SUMMARY CELL MEMBRANES ARE SELECTIVELY PERMEABLE DUE TO THEIR STRUCTURE Hydrophilic head Hydrophobic tail Hydrophobic regions of protein Hydrophilic regions of protein
BIOLOGY 111. CHAPTER 3: The Cell: The Fundamental Unit of Life
 BIOLOGY 111 CHAPTER 3: The Cell: The Fundamental Unit of Life The Cell: The Fundamental Unit of Life Learning Outcomes 3.1 Explain the similarities and differences between prokaryotic and eukaryotic cells
BIOLOGY 111 CHAPTER 3: The Cell: The Fundamental Unit of Life The Cell: The Fundamental Unit of Life Learning Outcomes 3.1 Explain the similarities and differences between prokaryotic and eukaryotic cells
LIFE IS CELLULAR. Cell Theory. Cells Are Small. Prokaryotic Cell 10/4/15. Chapter 7 Cell Structure and Function
 Chapter 7 Cell Structure and Function The cell basic unit of life, all living things are made of a cell (unicellular) or more than one cell (multicellular). LIFE IS CELLULAR The invention of the microscope
Chapter 7 Cell Structure and Function The cell basic unit of life, all living things are made of a cell (unicellular) or more than one cell (multicellular). LIFE IS CELLULAR The invention of the microscope
/ The following functional group is a. Aldehyde c. Carboxyl b. Ketone d. Amino
 Section A: Multiple Choice Select the answer that best answers the following questions. Please write your selected choice on the line provided, in addition to circling the answer. /25 1. The following
Section A: Multiple Choice Select the answer that best answers the following questions. Please write your selected choice on the line provided, in addition to circling the answer. /25 1. The following
Endomembrane system, *Chloroplasts, *Mitochondria. *Learn these from text/connect1. Fertilization of a human cell
 Key Concepts: - Cells are the Basic Unit of Life Cell Theory, Surface to Volume - 2 Cell Types Prokaryotic, Eukaryotic - Cell Membrane Membrane Structure - Cell Organelles Endomembrane system, *Chloroplasts,
Key Concepts: - Cells are the Basic Unit of Life Cell Theory, Surface to Volume - 2 Cell Types Prokaryotic, Eukaryotic - Cell Membrane Membrane Structure - Cell Organelles Endomembrane system, *Chloroplasts,
Unit 2 Warm Ups. Equilibrium
 Unit 2 Warm Ups Equilibrium 1. Cell wall 2. Mitochondria 3. Chloroplast 4. Vesicle 5. Vacuole 6. Rough Endoplasmic Reticulum 7. Smooth Endoplasmic Reticulum 8. Cytoskeleton 9. Lysosomes 10.Cell Membrane
Unit 2 Warm Ups Equilibrium 1. Cell wall 2. Mitochondria 3. Chloroplast 4. Vesicle 5. Vacuole 6. Rough Endoplasmic Reticulum 7. Smooth Endoplasmic Reticulum 8. Cytoskeleton 9. Lysosomes 10.Cell Membrane
Organelles. copyright cmassengale 1
 Organelles copyright cmassengale 1 Organelles Very small (Microscopic) Perform various functions for a cell Found in the cytoplasm May or may not be membrane-bound 2 Animal Cell Organelles Nucleolus Nucleus
Organelles copyright cmassengale 1 Organelles Very small (Microscopic) Perform various functions for a cell Found in the cytoplasm May or may not be membrane-bound 2 Animal Cell Organelles Nucleolus Nucleus
Look at the following images, what are some similarities and differences between the cells?
 Look at the following images, what are some similarities and differences between the cells? Name the two different types of cells 1. Prokaryotic Cells 2. Eukaryotic Cells Unit 3: Cells Objective: To
Look at the following images, what are some similarities and differences between the cells? Name the two different types of cells 1. Prokaryotic Cells 2. Eukaryotic Cells Unit 3: Cells Objective: To
The further from the nucleus, the higher the electron s energy Valence shell electrons participate in biological reactions
 Chemistry of Life Revision: The further from the nucleus, the higher the electron s energy Valence shell electrons participate in biological reactions Atoms exchange electrons with other elements to form
Chemistry of Life Revision: The further from the nucleus, the higher the electron s energy Valence shell electrons participate in biological reactions Atoms exchange electrons with other elements to form
Chapter 3 Review Assignment
 Class: Date: Chapter 3 Review Assignment Multiple Choice 40 MC = 40 Marks Identify the choice that best completes the statement or answers the question. 1. Which of the following organelles produces transport
Class: Date: Chapter 3 Review Assignment Multiple Choice 40 MC = 40 Marks Identify the choice that best completes the statement or answers the question. 1. Which of the following organelles produces transport
WELCOME TO BIOLOGY 11. Mr. Gandha
 WELCOME TO BIOLOGY 11 Mr. Gandha TOPICS OF BIOLOGY 11 Chemicals of life Cells Evolution Taxonomy Microbio Plants Animals BIOLOGY THIS SEMESTER Review of Biology and Processes Adaptation and Evolution:
WELCOME TO BIOLOGY 11 Mr. Gandha TOPICS OF BIOLOGY 11 Chemicals of life Cells Evolution Taxonomy Microbio Plants Animals BIOLOGY THIS SEMESTER Review of Biology and Processes Adaptation and Evolution:
MY BIOLOGY FINAL EXAM WORKBOOK
 NAME PER DATE MY BIOLOGY FINAL EXAM WORKBOOK DIRECTIONS: This study work book is due on the day of your final exam. Start now! After you have completed this study guide, you need to memorize it! 1. Look
NAME PER DATE MY BIOLOGY FINAL EXAM WORKBOOK DIRECTIONS: This study work book is due on the day of your final exam. Start now! After you have completed this study guide, you need to memorize it! 1. Look
Biology 12 Cell Structure and Function. Typical Animal Cell
 Biology 12 Cell Structure and Function Typical Animal Cell Vacuoles: storage of materials and water Golgi body: a series of stacked disk shaped sacs. Repackaging centre stores, modifies, and packages proteins
Biology 12 Cell Structure and Function Typical Animal Cell Vacuoles: storage of materials and water Golgi body: a series of stacked disk shaped sacs. Repackaging centre stores, modifies, and packages proteins
History of the Cell. History of the Cell 10/24/2013. Unit 3: Cellular Structure and Function. Robert Hooke (1665) Robert Hooke (1665)
 Unit 3: Cellular Structure and Function Mr. Hulse BVHS 2013-2014 Unit 3: Learning Targets 1-9 History of the Cell Robert Hooke (1665) 1 st person to see a cell Observed a piece of cork using a microscope
Unit 3: Cellular Structure and Function Mr. Hulse BVHS 2013-2014 Unit 3: Learning Targets 1-9 History of the Cell Robert Hooke (1665) 1 st person to see a cell Observed a piece of cork using a microscope
Cytosol the fluid Cytoplasm cell interior, everything outside the nucleus but within the cell membrane, includes the organelles, cytosol, and
 Cell Organelles Plasma Membrane comprised of a phospholipid bilayer and embedded proteins Outer surface has oligosaccharides separates the cells s contents from its surroundings Cytosol the fluid Cytoplasm
Cell Organelles Plasma Membrane comprised of a phospholipid bilayer and embedded proteins Outer surface has oligosaccharides separates the cells s contents from its surroundings Cytosol the fluid Cytoplasm
Study Guide for Biology Chapter 5
 Class: Date: Study Guide for Biology Chapter 5 Multiple Choice Identify the choice that best completes the statement or answers the question. 1. Which of the following led to the discovery of cells? a.
Class: Date: Study Guide for Biology Chapter 5 Multiple Choice Identify the choice that best completes the statement or answers the question. 1. Which of the following led to the discovery of cells? a.
Unit 2: More on Matter & Energy in Ecosystems. Macromolecules to Organelles to Cells
 IN: Unit 2: More on Matter & Energy in Ecosystems Macromolecules to Organelles to Cells Where are cells on the biological scale? Sub-Atomic Particles Atoms Molecules Macromolecules (proteins, lipids, nucleic
IN: Unit 2: More on Matter & Energy in Ecosystems Macromolecules to Organelles to Cells Where are cells on the biological scale? Sub-Atomic Particles Atoms Molecules Macromolecules (proteins, lipids, nucleic
First discovered in 1665 since then every organism observed with microscopes shows cells
 The Cell Cell theory (1838): 1. All organisms are composed of one or more cells, and the life processes of metabolism and heredity occur within these cells. 2. Cells are the smallest living things, the
The Cell Cell theory (1838): 1. All organisms are composed of one or more cells, and the life processes of metabolism and heredity occur within these cells. 2. Cells are the smallest living things, the
The Cell and Cellular transport
 Cell theory (1838): The Cell 1. All organisms are composed of one or more cells, and the life processes of metabolism and heredity occur within these cells. 2. Cells are the smallest living things, the
Cell theory (1838): The Cell 1. All organisms are composed of one or more cells, and the life processes of metabolism and heredity occur within these cells. 2. Cells are the smallest living things, the
Cell Theory. Cells are the basic unit of life.
 3.1 7.1 Cell Theory Cells are the basic unit of life. 3.1 7.1 Cell Theory The cell theory grew out of the work of many scientists Galileo (1610) made the first microscope Hooke (1665) made up the term
3.1 7.1 Cell Theory Cells are the basic unit of life. 3.1 7.1 Cell Theory The cell theory grew out of the work of many scientists Galileo (1610) made the first microscope Hooke (1665) made up the term
Lesson 1. Cell Theory - Statements - Exceptions. Categorizing Cells - Prokaryotes vs Eukaryotes
 Lesson 1 Cell Theory - Statements - Exceptions Categorizing Cells - Prokaryotes vs Eukaryotes The Cell Theory The discovery of cells and their structure is linked to the development of the magnifying lenses,
Lesson 1 Cell Theory - Statements - Exceptions Categorizing Cells - Prokaryotes vs Eukaryotes The Cell Theory The discovery of cells and their structure is linked to the development of the magnifying lenses,
The Cell. Biology 105 Lecture 4 Reading: Chapter 3 (pages 47 62)
 The Cell Biology 105 Lecture 4 Reading: Chapter 3 (pages 47 62) Outline I. Prokaryotic vs. Eukaryotic II. Eukaryotic A. Plasma membrane transport across B. Main features of animal cells and their functions
The Cell Biology 105 Lecture 4 Reading: Chapter 3 (pages 47 62) Outline I. Prokaryotic vs. Eukaryotic II. Eukaryotic A. Plasma membrane transport across B. Main features of animal cells and their functions
Sample Questions BSC1010C Chapters 5-7
 Sample Questions BSC1010C Chapters 5-7 1. Which type of lipid is most important in biological membranes? a. oils b. fats c. wax d. phospholipids e. triglycerides 2. Which type of interaction stabilizes
Sample Questions BSC1010C Chapters 5-7 1. Which type of lipid is most important in biological membranes? a. oils b. fats c. wax d. phospholipids e. triglycerides 2. Which type of interaction stabilizes
Chapter 1 Plasma membranes
 1 of 5 TEXTBOOK ANSWERS Chapter 1 Plasma membranes Recap 1.1 1 The plasma membrane: keeps internal contents of the cell confined to one area keeps out foreign molecules that damage or destroy the cell
1 of 5 TEXTBOOK ANSWERS Chapter 1 Plasma membranes Recap 1.1 1 The plasma membrane: keeps internal contents of the cell confined to one area keeps out foreign molecules that damage or destroy the cell
Don t Freak Out. Test on cell organelle on Friday!
 Cell Structure 1 Don t Freak Out Test on cell organelle on Friday! This test should be a buffer test and help raise your overall test score. All information will come from this week! 2 Cells Provide Compartments
Cell Structure 1 Don t Freak Out Test on cell organelle on Friday! This test should be a buffer test and help raise your overall test score. All information will come from this week! 2 Cells Provide Compartments
The Nature of a Cell. A cell is a compartment containing a variety of controlled chemical reactions. All organisms are made of cells.
 The Nature of a Cell A cell is a compartment containing a variety of controlled chemical reactions. All organisms are made of cells. Intracellular Aqueous Environment Extracellular Aqueous Environment
The Nature of a Cell A cell is a compartment containing a variety of controlled chemical reactions. All organisms are made of cells. Intracellular Aqueous Environment Extracellular Aqueous Environment
Think About it. Prokaryotic v. Eukaryotic Cells. The Discovery of the Cell. The cell theory states: Exploring the Cell 10/10/2016
 Think About it What s the smallest part of any living thing that still counts as being alive? Prokaryotic v. Eukaryotic Cells Can we just keep dividing living things into smaller and smaller parts, or
Think About it What s the smallest part of any living thing that still counts as being alive? Prokaryotic v. Eukaryotic Cells Can we just keep dividing living things into smaller and smaller parts, or
Name: Date: Block: Biology 12
 Name: Date: Block: Biology 12 Provincial Exam Review: Cell Processes and Applications January 2003 Use the following diagram to answer questions 1 and 2. 1. Which labelled organelle produces most of the
Name: Date: Block: Biology 12 Provincial Exam Review: Cell Processes and Applications January 2003 Use the following diagram to answer questions 1 and 2. 1. Which labelled organelle produces most of the
Objectives. By the end of the lesson you should be able to: State the 2 types of cells Relate the structure to function for all the organelles
 Biology 11 THE Cell Objectives By the end of the lesson you should be able to: State the 2 types of cells Relate the structure to function for all the organelles Types of Cells There are two types of cells:
Biology 11 THE Cell Objectives By the end of the lesson you should be able to: State the 2 types of cells Relate the structure to function for all the organelles Types of Cells There are two types of cells:
Cells & Transport. Chapter 7.1, 7.2, & 7.4
 Cells & Transport Chapter 7.1, 7.2, & 7.4 Do Now How big is a cell? How many cells are we made of? How many cells is the smallest living organism made of? Objectives Describe how cells were discovered
Cells & Transport Chapter 7.1, 7.2, & 7.4 Do Now How big is a cell? How many cells are we made of? How many cells is the smallest living organism made of? Objectives Describe how cells were discovered
Chapter 8 Cells and Their Environment
 Chapter Outline Chapter 8 Cells and Their Environment Section 1: Cell Membrane KEY IDEAS > How does the cell membrane help a cell maintain homeostasis? > How does the cell membrane restrict the exchange
Chapter Outline Chapter 8 Cells and Their Environment Section 1: Cell Membrane KEY IDEAS > How does the cell membrane help a cell maintain homeostasis? > How does the cell membrane restrict the exchange
In the space provided, write the letter of the term or phrase that best completes each statement or best answers each question.
 CHAPTER 3 TEST Cell Structure Circle T if the statement is true or F if it is false. T F 1. Small cells can transport materials and information more quickly than larger cells can. T F 2. Newly made proteins
CHAPTER 3 TEST Cell Structure Circle T if the statement is true or F if it is false. T F 1. Small cells can transport materials and information more quickly than larger cells can. T F 2. Newly made proteins
Exam 2 Practice Problems
 Exam 2 Practice Problems Cell Structure and Function Practice Questions 1. One of the relationships that exists between ribosomes and lysosomes is that a. ribosomes produce enzymes that could be stored
Exam 2 Practice Problems Cell Structure and Function Practice Questions 1. One of the relationships that exists between ribosomes and lysosomes is that a. ribosomes produce enzymes that could be stored
Cell Structure and Function
 Cell Structure and Function Agre and cells in the news Cells Smallest living unit Most are microscopic Discovery of Cells Robert Hooke (mid-1600s) Observed sliver of cork Saw row of empty boxes Coined
Cell Structure and Function Agre and cells in the news Cells Smallest living unit Most are microscopic Discovery of Cells Robert Hooke (mid-1600s) Observed sliver of cork Saw row of empty boxes Coined
Cells. Prokaryotic vs. Eukaryotic Euakryotic cells are generally one to one hundred times bigger than prokaryotic cells
 Cell Theory Cells 1. All living things are composed of one or more cell 2. Cell is the basic unit of life 3. All cells come from the division of pre-existing cells Cells are divided into 2 categories:
Cell Theory Cells 1. All living things are composed of one or more cell 2. Cell is the basic unit of life 3. All cells come from the division of pre-existing cells Cells are divided into 2 categories:
A. Major parts 1. Nucleus 2. Cytoplasm a. Contain organelles (see below) 3. Plasma membrane (To be discussed in Cellular Transport Lecture)
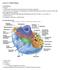 Lecture 5: Cellular Biology I. Cell Theory Concepts: 1. Cells are the functional and structural units of living organisms 2. The activity of an organism is dependent on both the individual and collective
Lecture 5: Cellular Biology I. Cell Theory Concepts: 1. Cells are the functional and structural units of living organisms 2. The activity of an organism is dependent on both the individual and collective
Basic Structure of a Cell
 Basic Structure of a Cell 1 Introduction to Cells Cells are the basic units of organisms Cells can only be observed under microscope Basic types of cells: Animal Cell Plant Cell Bacterial Cell 2 Number
Basic Structure of a Cell 1 Introduction to Cells Cells are the basic units of organisms Cells can only be observed under microscope Basic types of cells: Animal Cell Plant Cell Bacterial Cell 2 Number
Mock Exam 1 Biology 123 SI 1. Sodium and Lithium are two different elements, yet they react very similarly. What is the best explanation for this. a.
 Mock Exam 1 Biology 123 SI 1. Sodium and Lithium are two different elements, yet they react very similarly. What is the best explanation for this. a. They have the same number of electron shells. b. They
Mock Exam 1 Biology 123 SI 1. Sodium and Lithium are two different elements, yet they react very similarly. What is the best explanation for this. a. They have the same number of electron shells. b. They
Cellular Structure and Function. Chapter 7
 Cellular Structure and Function. Chapter 7 Cell Discovery and Theory. A cell is the basic structural and functional unit of all living organisms. The human body is made of trillions of cells that are too
Cellular Structure and Function. Chapter 7 Cell Discovery and Theory. A cell is the basic structural and functional unit of all living organisms. The human body is made of trillions of cells that are too
Renaissance Biology Midterm Study Guide Answers
 Renaissance Biology Midterm Study Guide Answers 2016-2017 LEARNING TARGET 1: List the characteristics of life Made of one or more cells Organization cells -> tissues -> organs -> organ systems -> organisms
Renaissance Biology Midterm Study Guide Answers 2016-2017 LEARNING TARGET 1: List the characteristics of life Made of one or more cells Organization cells -> tissues -> organs -> organ systems -> organisms
Chapter 1: Studying Life
 Chapter 1: Studying Life 1. Most living organisms share key characteristics: consist of one or more cells, contain genetic information, reproduce themselves, have evolved, and carry out metabolism. 2.
Chapter 1: Studying Life 1. Most living organisms share key characteristics: consist of one or more cells, contain genetic information, reproduce themselves, have evolved, and carry out metabolism. 2.
SBI3U7 Cell Structure & Organelles. 2.2 Prokaryotic Cells 2.3 Eukaryotic Cells
 SBI3U7 Cell Structure & Organelles 2.2 Prokaryotic Cells 2.3 Eukaryotic Cells No nucleus Prokaryotic Cells No membrane bound organelles Has a nucleus Eukaryotic Cells Membrane bound organelles Unicellular
SBI3U7 Cell Structure & Organelles 2.2 Prokaryotic Cells 2.3 Eukaryotic Cells No nucleus Prokaryotic Cells No membrane bound organelles Has a nucleus Eukaryotic Cells Membrane bound organelles Unicellular
What are the parts of a eukaryotic cell? What is the function of each part of a eukaryotic cell?
 CHAPTER 3 SECTION 2 Cells: The Basic Units of Life Eukaryotic Cells BEFORE YOU READ After you read this section, you should be able to answer these questions: What are the parts of a eukaryotic cell? What
CHAPTER 3 SECTION 2 Cells: The Basic Units of Life Eukaryotic Cells BEFORE YOU READ After you read this section, you should be able to answer these questions: What are the parts of a eukaryotic cell? What
Cell Structure & Function. Source:
 Cell Structure & Function Source: http://koning.ecsu.ctstateu.edu/cell/cell.html Definition of Cell A cell is the smallest unit that is capable of performing life functions. http://web.jjay.cuny.edu/~acarpi/nsc/images/cell.gif
Cell Structure & Function Source: http://koning.ecsu.ctstateu.edu/cell/cell.html Definition of Cell A cell is the smallest unit that is capable of performing life functions. http://web.jjay.cuny.edu/~acarpi/nsc/images/cell.gif
Chapter 4 Organization of the Cell
 Chapter 4 Organization of the Cell Cell basic unit of life o Small o Self-sufficient o Self-replicating Cell Theory organisms are composed of cells and all cells come from the division of other cells Cells
Chapter 4 Organization of the Cell Cell basic unit of life o Small o Self-sufficient o Self-replicating Cell Theory organisms are composed of cells and all cells come from the division of other cells Cells
Delve AP Biology Lecture 4: 10/9/11 Melissa Ko and Anne Huang
 Today s Agenda: I. Review of organelles II. More important organelles III. Plasma membrane structure IV. Diffusion and transport Delve AP Biology Lecture 4: 10/9/11 Melissa Ko and Anne Huang I. Review
Today s Agenda: I. Review of organelles II. More important organelles III. Plasma membrane structure IV. Diffusion and transport Delve AP Biology Lecture 4: 10/9/11 Melissa Ko and Anne Huang I. Review
3. Describe the study in mimicry, using king snakes and coral snakes. Identify the control in the experiment.
 Biology Semester 1 Exam Review Guide Chapter 1 Biology in the 21 st Century 1. Distinguish between the following key terms: Biology Name : Pd: Hypothesis Variable Controlled experiment Theory Model Technology
Biology Semester 1 Exam Review Guide Chapter 1 Biology in the 21 st Century 1. Distinguish between the following key terms: Biology Name : Pd: Hypothesis Variable Controlled experiment Theory Model Technology
BIOLOGICAL MOLECULES REVIEW-UNIT 1 1. The factor being tested in an experiment is the A. data. B. variable. C. conclusion. D. observation. 2.
 BIOLOGICAL MOLECULES REVIEW-UNIT 1 1. The factor being tested in an experiment is the A. data. B. variable. C. conclusion. D. observation. 2. A possible explanation for an event that occurs in nature is
BIOLOGICAL MOLECULES REVIEW-UNIT 1 1. The factor being tested in an experiment is the A. data. B. variable. C. conclusion. D. observation. 2. A possible explanation for an event that occurs in nature is
Lab 3: Cellular Structure and Function
 Lab 3: Cellular Structure and Function What is the basic unit of life? The simplest form of life is the cell! All living things are either: unicellular (only one cell) multicellular (many cells make one
Lab 3: Cellular Structure and Function What is the basic unit of life? The simplest form of life is the cell! All living things are either: unicellular (only one cell) multicellular (many cells make one
Cells. Unit 3 Cell Structure and Function. Cells. Plasma Membrane
 Unit 3 Cell Structure and Function Cells Cell theory The cell is the basic unit of life The cells of all living things exhibit the seven characteristics of life All living things are made of cells Cells
Unit 3 Cell Structure and Function Cells Cell theory The cell is the basic unit of life The cells of all living things exhibit the seven characteristics of life All living things are made of cells Cells
A Tour of the Cell Lecture 2, Part 1 Fall 2008
 Cell Theory 1 A Tour of the Cell Lecture 2, Part 1 Fall 2008 Cells are the basic unit of structure and function The lowest level of structure that can perform all activities required for life Reproduction
Cell Theory 1 A Tour of the Cell Lecture 2, Part 1 Fall 2008 Cells are the basic unit of structure and function The lowest level of structure that can perform all activities required for life Reproduction
Notes Chapter 7 Cell Structure and Function Hooke looked at cork under a simple microscope and found tiny chambers he named cells.
 Notes Chapter 7 Cell Structure and Function 7.1 Cell discovery and Theory 1665 Hooke looked at cork under a simple microscope and found tiny chambers he named cells. Cells are the basic structural and
Notes Chapter 7 Cell Structure and Function 7.1 Cell discovery and Theory 1665 Hooke looked at cork under a simple microscope and found tiny chambers he named cells. Cells are the basic structural and
Cytoskeleton. Provide shape and support for the cell. Other functions of the cytoskeleton. Nucleolus. Nucleus
 Chapter 4: Cell Structure and Function Cytoskeleton The cytoskeleton is a network of fibers that organizes structures and activities in the cell. Microtubules (the largest) Intermediate fibers Microfilaments
Chapter 4: Cell Structure and Function Cytoskeleton The cytoskeleton is a network of fibers that organizes structures and activities in the cell. Microtubules (the largest) Intermediate fibers Microfilaments
Unit 2:The Cell. Section 3: Organelle Structure and Function Mrs. McNamara Biology
 Unit 2:The Cell Section 3: Organelle Structure and Function Mrs. McNamara Biology Organelle-cell part that performs a specific function for the cell Most are surrounded by a membrane Each helps to maintain
Unit 2:The Cell Section 3: Organelle Structure and Function Mrs. McNamara Biology Organelle-cell part that performs a specific function for the cell Most are surrounded by a membrane Each helps to maintain
Standard 2 Exam Biology. 2. This macromolecule is responsible for short term energy storage and structural support in plants
 1. This macromolecule is responsible for structural support, movement, enzymatic activity, cell communication, and is made of amino acids. a. Lipids b. Carbohydrates c. Proteins d. Nucleic Acids e. ATP
1. This macromolecule is responsible for structural support, movement, enzymatic activity, cell communication, and is made of amino acids. a. Lipids b. Carbohydrates c. Proteins d. Nucleic Acids e. ATP
Cell Category? Prokaryote
 CELLS Cell Category? Prokaryote Prokaryote Eukaryote Cell Category? Cell Type? Cell Category? Cell Type? Endosymbiosis eukaryotic cells were formed from simpler prokaryotes Endo within Symbiosis together
CELLS Cell Category? Prokaryote Prokaryote Eukaryote Cell Category? Cell Type? Cell Category? Cell Type? Endosymbiosis eukaryotic cells were formed from simpler prokaryotes Endo within Symbiosis together
Transport: Cell Membrane Structure and Function. Biology 12 Chapter 4
 Transport: Cell Membrane Structure and Function Biology 12 Chapter 4 FLUID-MOSAIC MODEL OF MEMBRANE STRUCTURE The cell membrane (plasma membrane) is made of two layers of phospholipid molecules (bilayer)
Transport: Cell Membrane Structure and Function Biology 12 Chapter 4 FLUID-MOSAIC MODEL OF MEMBRANE STRUCTURE The cell membrane (plasma membrane) is made of two layers of phospholipid molecules (bilayer)
Basic Structure of a Cell. copyright cmassengale
 Basic Structure of a Cell 1 Review Facts About Living Things 2 What Are the Main Characteristics of organisms? 1. Made of CELLS 2. Require ENERGY (food) 3. REPRODUCE (species) 4. Maintain HOMEOSTASIS 5.
Basic Structure of a Cell 1 Review Facts About Living Things 2 What Are the Main Characteristics of organisms? 1. Made of CELLS 2. Require ENERGY (food) 3. REPRODUCE (species) 4. Maintain HOMEOSTASIS 5.
8/7/18. UNIT 2: Cells Chapter 3: Cell Structure and Function. I. Cell Theory (3.1) A. Early studies led to the development of the cell theory
 8/7/18 UNIT 2: Cells Chapter 3: Cell Structure and Function I. Cell Theory (3.1) A. Early studies led to the development of the cell theory 1. Discovery of Cells a. Robert Hooke (1665)-Used compound microscope
8/7/18 UNIT 2: Cells Chapter 3: Cell Structure and Function I. Cell Theory (3.1) A. Early studies led to the development of the cell theory 1. Discovery of Cells a. Robert Hooke (1665)-Used compound microscope
Eukaryotic Cell Structure
 Eukaryotic Cell Structure Vocabulary listed for Chapter 7.3: cell wall, chromatin, nucleolus, ribosome, cytoplasm, endoplasmic reticulum, Golgi apparatus, vacuole, lysosome, chloroplast, plastid, chlorophyll,
Eukaryotic Cell Structure Vocabulary listed for Chapter 7.3: cell wall, chromatin, nucleolus, ribosome, cytoplasm, endoplasmic reticulum, Golgi apparatus, vacuole, lysosome, chloroplast, plastid, chlorophyll,
Lesson Overview. 7.2 Cell Structure
 7.2 Cell Organization The eukaryotic cell can be divided into two major parts: the nucleus and the cytoplasm. The cytoplasm is the fluid portion of the cell outside the nucleus. Prokaryotes do not have
7.2 Cell Organization The eukaryotic cell can be divided into two major parts: the nucleus and the cytoplasm. The cytoplasm is the fluid portion of the cell outside the nucleus. Prokaryotes do not have
Chapter Seven. A View of the Cell
 Chapter Seven A View of the Cell Cellular Organization Cell Tissue group of cells functioning together. Organ group of tissues functioning together. Organ System group of organs functioning together. Organism
Chapter Seven A View of the Cell Cellular Organization Cell Tissue group of cells functioning together. Organ group of tissues functioning together. Organ System group of organs functioning together. Organism
BIOCHEMISTRY. How Are Macromolecules Formed? Dehydration Synthesis or condensation reaction Polymers formed by combining monomers and removing water.
 BIOCHEMISTRY Organic compounds Compounds that contain carbon are called organic. Inorganic compounds do not contain carbon. Carbon has 4 electrons in outer shell. Carbon can form covalent bonds with as
BIOCHEMISTRY Organic compounds Compounds that contain carbon are called organic. Inorganic compounds do not contain carbon. Carbon has 4 electrons in outer shell. Carbon can form covalent bonds with as
From Atoms to Cells: Fundamental Building Blocks. Models of atoms. A chemical connection
 From Atoms to Cells: A chemical connection Fundamental Building Blocks Matter - all materials that occupy space & have mass Matter is composed of atoms Atom simplest form of matter not divisible into simpler
From Atoms to Cells: A chemical connection Fundamental Building Blocks Matter - all materials that occupy space & have mass Matter is composed of atoms Atom simplest form of matter not divisible into simpler
CELL PART OF THE DAY. Chapter 7: Cell Structure and Function
 CELL PART OF THE DAY Chapter 7: Cell Structure and Function Cell Membrane Cell membranes are composed of two phospholipid layers. Cell membrane is flexible, not rigid The cell membrane has two major functions.
CELL PART OF THE DAY Chapter 7: Cell Structure and Function Cell Membrane Cell membranes are composed of two phospholipid layers. Cell membrane is flexible, not rigid The cell membrane has two major functions.
3UNIT. Photosynthesis and. Cellular Respiration. Unit PreQuiz? General Outcomes. Unit 3 Contents. Focussing Questions
 3UNIT Photosynthesis and Cellular Respiration General Outcomes In this unit, you will relate photosynthesis to the storage of energy in organic compounds explain the role of cellular respiration in releasing
3UNIT Photosynthesis and Cellular Respiration General Outcomes In this unit, you will relate photosynthesis to the storage of energy in organic compounds explain the role of cellular respiration in releasing
Nucleic acids. Nucleic acids are information-rich polymers of nucleotides
 Nucleic acids Nucleic acids are information-rich polymers of nucleotides DNA and RNA Serve as the blueprints for proteins and thus control the life of a cell RNA and DNA are made up of very similar nucleotides.
Nucleic acids Nucleic acids are information-rich polymers of nucleotides DNA and RNA Serve as the blueprints for proteins and thus control the life of a cell RNA and DNA are made up of very similar nucleotides.
First to View Cells. copyright cmassengale
 CELL THEORY All living things are made of cells Cells are the basic unit of structure and function in an organism (basic unit of life) Cells come from the reproduction of existing cells (cell division)
CELL THEORY All living things are made of cells Cells are the basic unit of structure and function in an organism (basic unit of life) Cells come from the reproduction of existing cells (cell division)
Unit 2 Cell Structure and Function
 Unit 2 Cell Structure and Function Biology 30 Mr. Oosterom Development of the Cell Theory People have known about the existence of cells for approximately 300 yrs Early microscopes allowed scientists to
Unit 2 Cell Structure and Function Biology 30 Mr. Oosterom Development of the Cell Theory People have known about the existence of cells for approximately 300 yrs Early microscopes allowed scientists to
CELL STRUCTURE AND FUNCTION. Chapter 7
 CELL STRUCTURE AND FUNCTION Chapter 7 WARM UP EXERCISE Please complete the pretest that you picked up as you came in. LIFE IS CELLULAR Robert Hooke- coined the term cells The Cell Theory All living things
CELL STRUCTURE AND FUNCTION Chapter 7 WARM UP EXERCISE Please complete the pretest that you picked up as you came in. LIFE IS CELLULAR Robert Hooke- coined the term cells The Cell Theory All living things
Introduction. Biochemistry: It is the chemistry of living things (matters).
 Introduction Biochemistry: It is the chemistry of living things (matters). Biochemistry provides fundamental understanding of the molecular basis for the function and malfunction of living things. Biochemistry
Introduction Biochemistry: It is the chemistry of living things (matters). Biochemistry provides fundamental understanding of the molecular basis for the function and malfunction of living things. Biochemistry
A Tour of the Cell. Ch. 7
 A Tour of the Cell Ch. 7 Cell Theory O All organisms are composed of one or more cells. O The cell is the basic unit of structure and organization of organisms. O All cells come from preexisting cells.
A Tour of the Cell Ch. 7 Cell Theory O All organisms are composed of one or more cells. O The cell is the basic unit of structure and organization of organisms. O All cells come from preexisting cells.
2.2 Properties of Water
 2.2 Properties of Water I. Water s unique properties allow life to exist on Earth. A. Life depends on hydrogen bonds in water. B. Water is a polar molecule. 1. Polar molecules have slightly charged regions
2.2 Properties of Water I. Water s unique properties allow life to exist on Earth. A. Life depends on hydrogen bonds in water. B. Water is a polar molecule. 1. Polar molecules have slightly charged regions
Cell Structure and Function Practice Exam - KEY
 Biology 12 Name: Cell Structure and Function Practice Exam - KEY Cell parts and Function 1. Identify each part of the cell indicated and give one role for each structure in the secretion and/or synthesis
Biology 12 Name: Cell Structure and Function Practice Exam - KEY Cell parts and Function 1. Identify each part of the cell indicated and give one role for each structure in the secretion and/or synthesis
Chapter 3 Cell Structures & Functions
 Biology 12 Name: Cell Biology Per: Date: Chapter 3 Cell Structures & Functions Complete using BC Biology 12, pages 62-107 Diagnostic Questions (mark using the answer key on page 527) 1. 2. 3. 4. 9. What
Biology 12 Name: Cell Biology Per: Date: Chapter 3 Cell Structures & Functions Complete using BC Biology 12, pages 62-107 Diagnostic Questions (mark using the answer key on page 527) 1. 2. 3. 4. 9. What
Biomolecules. Unit 3
 Biomolecules Unit 3 Atoms Elements Compounds Periodic Table What are biomolecules? Monomers vs Polymers Carbohydrates Lipids Proteins Nucleic Acids Minerals Vitamins Enzymes Triglycerides Chemical Reactions
Biomolecules Unit 3 Atoms Elements Compounds Periodic Table What are biomolecules? Monomers vs Polymers Carbohydrates Lipids Proteins Nucleic Acids Minerals Vitamins Enzymes Triglycerides Chemical Reactions
Modern Cell Theory. Plasma Membrane. Generalized Cell Structures. Cellular Form and Function. Three principle parts of a cell
 Cellular Form and Function Concepts of cellular structure Cell surface Membrane transport Cytoplasm Modern Cell Theory All living organisms are composed of cells. the simplest structural and functional
Cellular Form and Function Concepts of cellular structure Cell surface Membrane transport Cytoplasm Modern Cell Theory All living organisms are composed of cells. the simplest structural and functional
Cell and Cell Membrane Structure and Function
 Lesson 3 Cell and Cell Membrane Structure and Function Introduction to Life Processes - SCI 102 1 The Cell Theory Three principles comprise the cell theory 1) Every living organism is made up of one or
Lesson 3 Cell and Cell Membrane Structure and Function Introduction to Life Processes - SCI 102 1 The Cell Theory Three principles comprise the cell theory 1) Every living organism is made up of one or
Membranes. Chapter 5
 Membranes Chapter 5 Membrane Structure The fluid mosaic model of membrane structure contends that membranes consist of: -phospholipids arranged in a bilayer -globular proteins inserted in the lipid bilayer
Membranes Chapter 5 Membrane Structure The fluid mosaic model of membrane structure contends that membranes consist of: -phospholipids arranged in a bilayer -globular proteins inserted in the lipid bilayer
Cells. Variation and Function of Cells
 Cells Variation and Function of Cells Cell Theory states that: 1. All living things are made of cells 2. Cells are the basic unit of structure and function in living things 3. New cells are produced from
Cells Variation and Function of Cells Cell Theory states that: 1. All living things are made of cells 2. Cells are the basic unit of structure and function in living things 3. New cells are produced from
AP Biology Exam Review Part I: Biochemistry, Cells and Transport. A. Chemistry of Life 1. CHNOPS- most common elements in all living matter
 AP Biology Exam Review Part I: Biochemistry, Cells and Transport 2A3: Organisms must exchange matter with the environment to grow, reproduce, and maintain organization. 2B1: Cell Membranes are selectively
AP Biology Exam Review Part I: Biochemistry, Cells and Transport 2A3: Organisms must exchange matter with the environment to grow, reproduce, and maintain organization. 2B1: Cell Membranes are selectively
Cell Structure and Function. Biology 12 Unit 1 Cell Structure and Function Inquiry into Life pages and 68-69
 Cell Structure and Function Biology 12 Unit 1 Cell Structure and Function Inquiry into Life pages 45 59 and 68-69 Assignments for this Unit Pick up the notes/worksheet for this unit and the project There
Cell Structure and Function Biology 12 Unit 1 Cell Structure and Function Inquiry into Life pages 45 59 and 68-69 Assignments for this Unit Pick up the notes/worksheet for this unit and the project There
Membrane Structure. Membrane Structure. Membrane Structure. Membranes
 Membrane Structure Membranes Chapter 5 The fluid mosaic model of membrane structure contends that membranes consist of: -phospholipids arranged in a bilayer -globular proteins inserted in the lipid bilayer
Membrane Structure Membranes Chapter 5 The fluid mosaic model of membrane structure contends that membranes consist of: -phospholipids arranged in a bilayer -globular proteins inserted in the lipid bilayer
Exam 1 SC135 spring 2011 Page 1
 xam 1 S135 spring 2011 Page 1 Name: ate: 1 Which other item is worth the same (has the same weight) as your lecture exams toward your final grade? quizzes writing assignments participation presentation
xam 1 S135 spring 2011 Page 1 Name: ate: 1 Which other item is worth the same (has the same weight) as your lecture exams toward your final grade? quizzes writing assignments participation presentation
Biology 12. Biochemistry. Water - a polar molecule Water (H 2 O) is held together by covalent bonds.
 Biology 12 Biochemistry Water - a polar molecule Water (H 2 O) is held together by covalent bonds. Electrons in these bonds spend more time circulating around the larger Oxygen atom than the smaller Hydrogen
Biology 12 Biochemistry Water - a polar molecule Water (H 2 O) is held together by covalent bonds. Electrons in these bonds spend more time circulating around the larger Oxygen atom than the smaller Hydrogen
Answer Key for Exam 2 Practice Problems
 Answer Key for Exam 2 Practice Problems Biology 100 Revised Spring 2014 Cell Structure and Function Practice Questions 1. One of the relationships that exists between ribosomes and lysosomes is that a.
Answer Key for Exam 2 Practice Problems Biology 100 Revised Spring 2014 Cell Structure and Function Practice Questions 1. One of the relationships that exists between ribosomes and lysosomes is that a.
Cells are the smallest units of life CH 3
 Cells are the smallest units of life CH 3 The Cell Theory 1. All living things are composed of one or more cells. 2. Cells are the basic units of structure and function. 3. Cells are produced only from
Cells are the smallest units of life CH 3 The Cell Theory 1. All living things are composed of one or more cells. 2. Cells are the basic units of structure and function. 3. Cells are produced only from
(A) Cell membrane (B) Ribosome (C) DNA (D) Nucleus (E) Plasmids. A. Incorrect! Both prokaryotic and eukaryotic cells have cell membranes.
 High School Biology - Problem Drill 03: The Cell No. 1 of 10 1. Which of the following is NOT found in prokaryotic cells? #01 (A) Cell membrane (B) Ribosome (C) DNA (D) Nucleus (E) Plasmids Both prokaryotic
High School Biology - Problem Drill 03: The Cell No. 1 of 10 1. Which of the following is NOT found in prokaryotic cells? #01 (A) Cell membrane (B) Ribosome (C) DNA (D) Nucleus (E) Plasmids Both prokaryotic
Test Review Worksheet 1 Name: Per:
 Test Review Worksheet 1 Name: Per: 1. Put the following in order according to blood flow through the body, starting with the lungs: Lungs, right atrium, left atrium, right ventricle, left ventricle, aorta,
Test Review Worksheet 1 Name: Per: 1. Put the following in order according to blood flow through the body, starting with the lungs: Lungs, right atrium, left atrium, right ventricle, left ventricle, aorta,
Human Epithelial Cells
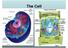 The Cell Human Epithelial Cells Plant Cells Cells have an internal structure Eukaryotic cells are organized Protective membrane around them that communicates with other cells Organelles have specific jobs
The Cell Human Epithelial Cells Plant Cells Cells have an internal structure Eukaryotic cells are organized Protective membrane around them that communicates with other cells Organelles have specific jobs
Chapters 2 and 3. Pages and Pages Prayer Attendance Homework
 Chapters 2 and 3 Pages 44-45 and Pages 59-62 Prayer Attendance Homework The Cell The cell is the basic unit of life on Earth, separated from its environment by a membrane and sometimes an outer wall. Prokaryotic
Chapters 2 and 3 Pages 44-45 and Pages 59-62 Prayer Attendance Homework The Cell The cell is the basic unit of life on Earth, separated from its environment by a membrane and sometimes an outer wall. Prokaryotic
Plasma Membrane Function
 Plasma Membrane Function Cells have to maintain homeostasis, they do this by controlling what moves across their membranes Structure Double Layer of phospholipids Head (polar) hydrophiliclikes water -
Plasma Membrane Function Cells have to maintain homeostasis, they do this by controlling what moves across their membranes Structure Double Layer of phospholipids Head (polar) hydrophiliclikes water -
10/5/2015. Cell Size. Relative Rate of Reaction
 The Cell Biology 102 Fundamental unit of life Smallest unit that displays all the basic elements of life Lecture 5: Cells Cell Theory 1. All living things are made of one or more cells Cell Theory 2. The
The Cell Biology 102 Fundamental unit of life Smallest unit that displays all the basic elements of life Lecture 5: Cells Cell Theory 1. All living things are made of one or more cells Cell Theory 2. The
CELL MEMBRANES. CELL MEMBRANE- Structure and Function
 BIOLOGY 12 CELL MEMBRANES NAME: INTRODUCTION 1. The cell membrane the passage of molecules into and out of the cell. 2. Some types of molecules, particularly molecules, pass freely across the cell membrane
BIOLOGY 12 CELL MEMBRANES NAME: INTRODUCTION 1. The cell membrane the passage of molecules into and out of the cell. 2. Some types of molecules, particularly molecules, pass freely across the cell membrane
Bio 12 Chapter 2 Test Review
 Bio 12 Chapter 2 Test Review 1.Know the difference between ionic and covalent bonds In order to complete outer shells in electrons bonds can be Ionic; one atom donates or receives electrons Covalent; atoms
Bio 12 Chapter 2 Test Review 1.Know the difference between ionic and covalent bonds In order to complete outer shells in electrons bonds can be Ionic; one atom donates or receives electrons Covalent; atoms
Chapter 3: Cell Structure and Function Assignment
 Chapter 3: Cell Structure and Function Assignment Provide full detail and be specific for full marks. 1. How do these organelles work together? 2 marks each = 10 marks a. lysosomes and vacuoles When material
Chapter 3: Cell Structure and Function Assignment Provide full detail and be specific for full marks. 1. How do these organelles work together? 2 marks each = 10 marks a. lysosomes and vacuoles When material
Membranes. Chapter 5. Membrane Structure
 Membranes Chapter 5 Membrane Structure Lipid Bilayer model: - double phospholipid layer - Gorter & Grendel: 1925 Fluid Mosaic model: consist of -phospholipids arranged in a bilayer -globular proteins inserted
Membranes Chapter 5 Membrane Structure Lipid Bilayer model: - double phospholipid layer - Gorter & Grendel: 1925 Fluid Mosaic model: consist of -phospholipids arranged in a bilayer -globular proteins inserted
Cell Transport Unit Test
 Cell Transport Unit Test ~Please DO NOT write on the test~ I CAN describe the parts of cells. 1. The ideas that all living things are composed of cells come from other cells defines: a. Central dogma b.
Cell Transport Unit Test ~Please DO NOT write on the test~ I CAN describe the parts of cells. 1. The ideas that all living things are composed of cells come from other cells defines: a. Central dogma b.
