The organic matrix of gallstones
|
|
|
- Elizabeth Underwood
- 5 years ago
- Views:
Transcription
1 Gut, 1974, 15, The organic matrix of gallstones D. JUNE SUTOR AND SUSAN E. WOOLEY From the Department of Chemistry, University College, London SUMMARY Dissolution of gallstones consisting of cholesterol, calcium carbonate, or calcium phosphate in different solvents left an amorphous organic gel-like substance (the matrix). Matrix from cholesterol stones could be colourless but was usually orange, yellow, or brown while that from calcium carbonate and calcium phosphate stones was almost invariably coloured black or dark brown. These pigments were also shown to be organic and amorphous. The amount of matrix present and its structure varied with the texture of the crystalline material. Irrespective of their composition, laminated pieces of material yielded compact laminated matrix of the same shape as the original piece and areas of loose crystalline material gave small pieces of non-cohesive matrix. Only large cholesterol crystals which usually radiate from the stone nucleus had no associated matrix. In common with urinary stones and other pathological concretions investigated, gallstones consisting entirely or predominantly of cholesterol contain an organic matrix of mucopolysaccharide. King and Boyce (1959) dissolved out cholesterol and some pigment from pooled gallstones and found the overall composition of mucopolysaccharide present in the remaining material different from that in both urinary and salivary calculi. The intact gallstones were not analysed but dissolution studies suggested that the pool was 60% cholesterol. The presence of mucopolysaccharide in cholesterol gallstones was confirmed by Womack, Zeppa, and Irvin (1963) who stained polished sections of them. Matrix has not been specifically sought in gallstones other than those of cholesterol nor has it been investigated for structural features. Studies on this material are necessary because it is an integral part of cholesterol gallstones at least. Its properties are also of importance since Danzinger, Hofmann, Schoenfield, and Thistle (1972) dissolved cholesterol stones in vivo by giving orally chenodeoxycholic acid, a compound unlikely to attack mucopolysaccharide. These could help to assess the likelihood of any residual matrix from 'stone dissolution' either being expelled by the gallbladder or remaining in that organ. In the latter case, such material could act as a nidus for further stone formation if the bile were in a supersaturated state. To help to solve these problems, we have undertaken a study of any matrix in gallstones of different composition and texture. Received for publication 18 March M_aterial and General Techniques At our disposal were several hundred gallstones whose crystalline composition had been thoroughly investigated by the x-ray powder method (Sutor and Wooley, 1971 and 1973). Each stone or piece of stone selected for study consisted entirely of one of the main constituents of gallstones, namely, cholesterol, calcium carbonate, calcium phosphate, calcium palmitate, or calcium bilirubinate. Pigment was, however, present in most samples. In different experiments different solvents were used to remove the crystalline material. The presence or absence of matrix could be quickly established by dissolving cholesterol gallstones in organic solvents such as chloroform, and calcium carbonate and calcium phosphate stones in normal hydrochloric acid. These reactions were usually so quick or vigorous that the matrix often broke up and structural studies on this substance required much slower dissolution of the crystalline material. To this end cholesterol gallstones varying in weight from 11 to 26 mg were each put in 12 ml of a 4% aqueous solution of the sodium salt of chenodeoxycholic acid (ph = 8.1) and the solutions changed every six weeks, while calcium carbonate or calcium phosphate stones (5-41 mg in weight) were each placed in 12 ml of a 5 % solution of the disodium salt of ethylenediaminetetra-acetic acid (EDTA) containing 0.4% w/v of formalin (ph = 4 5) and the solutions changed every 10 days. Suitable solvents for calcium palmitate and calcium bilirubinate stones could not be found. Most of the calcium stones contained dark 487
2 488 pigment some of which was soluble in water and some in a 5 % solution of the tetrasodium salt of EDTA (ph adjusted to 7.9), but solvents could not be found which removed all the colouring matter, particularly the black pigment. These stones were alternated between the depigmenting and decalcifying solutions. The majority of experiments were carried out in a refrigerator (1 C) but dissolution of cholesterol stones was also studied at room temperature (23-27 C) and at 37 C. Some stones were weighed before and after dissolution in order to ascertain the amount of matrix present. All matrix was well washed in water and freeze dried before being weighed, analysed for carbon, hydrogen, nitrogen, and calcium, ashed or photographed by x-ray diffraction to determine whether it was amorphous or crystalline. Detailed Experiments and Results CHOLESTEROL GALLSTONES Preliminary dissolution studies of cholesterol gallstones in chloroform showed that a gel-like substance (the matrix) was present in many stones and its structure varied with that of the crystalline material with which it was associated. The matrix could be colourless but it was usually orange, yellow, brown, or sometimes black. These observations were confirmed in the following experiments. Five small gallstones from the same patient, each weighing mg and having a thin surface band of fine-grained yellow material round a central area of almost colourless crystals radiating from the stone nucleus, were put in separate sodium chenodeoxycholate solutions in a refrigerator. Diffuseness of the outline of the stone, which was the first indication of the presence of matrix and resulted from the dissolution of the crystalline material leaving behind any associated gel, appeared within two weeks. The matrix as it was exposed flaked off and gradually broke into small yellow or colourless pieces. After about six months when the laminated surface bands and their associated matrix had gone, the stones took on a 'spiky' appearance. These spikes, which were crystals of cholesterol monohydrate, became more prominent as dissolution proceeded and finally broke up into a heap of single crystals which gradually dissolved. No matrix was present in this area. Dissolution of the stones took from seven to nine months. To confirm the absence of matrix in areas consisting of radiating crystals of cholesterol, a few such pieces having linear dimensions of 1 cm were placed in chloroform and their dissolution was followed under the microscope. Matrix was only found in a D. June Sutor and Susan E. Wooley small pigmented section of one corresponding to the original stone nucleus (magnification used = 150). Gallstones frequently consist of aggregates of crystals surrounded by laminated material. Small yellow-brown cholesterol stones (11-21 mg in weight) having these textures and from the same patient were placed in sodium chenodeoxycholate solution at 37 C, room temperature, and 1 C. A cohesive area of almost colourless matrix was associated with the laminated bands (fig 1) and this sometimes separated from the stone in one piece but in all cases it gradually broke up (fig 2) eventually into small pieces. Dissolution of the central crystalline aggregates yielded only small incohesive pieces of brown matrix. The stones took two and a quarter to six months to dissolve. The solubility of cholesterol appeared to increase with temperature but because of variations in the weight of individual stones and the texture distribution in them, a quantitative estimation of this was not possible. Fig 1 Cholesterol gallstone kept in sodium chenodeoxycholate for six weeks. Outer stone areas have dissolved leaving a colourless matrix but hard central core of cholesterol remains. Overall dimensions of material 6 x 4 mm. Approximate magnification in the figure xlo. A completely laminated brown cholesterol stone was cut into pieces weighing mg which were dissolved in sodium chenodeoxycholate at 37 C, room temperature, and 1 C. In all cases an exact replica in matrix of the original piece was obtained which was pigmented brown and tended not to disintegrate. The time for complete dissolution of the pieces was two-and-a-half to five months. The rate of dissolution appeared to increase with temperature
3 The organic matrix ofgallstones... because *:., : :present but they were not unambi uously identified ;. not enough material was available to take the 1 anecessary type of x-ray photographs. Apatite (calcium phosphate) and calcium carbonate were -...iablso imes found in these matrices which had not X-ray diffraction photographs showed that both the colourless and pigmented matrixwere amorphous. GALLSTONES OF CALCIUM SALTS When calcium carbonate or calcium phosphate i - w :forgallstones O~~~~~~~~~~~~beenr in were Ucur dissolved afeaowrgso decalcifyingsoluion in normal hydrochloric acid, a matrix was obtained which was almost ~~~~~~~invariably pigmented black and whose structure was related to that of the associated crystalline material. If, however, the gallstones contained much black... : rigment this tended to hold the matrix together and obscure structural features. These results were confirmed by the following slow decalcification (bottom) kept io cnexperiments. Six patients provided the calcium carbonate and..p.ec...n.sfour the calcium phosphate gallstones which were selected so that different textures and crystalline.e..d... forms of the calcium salts were studied. The stones gave... wereblack except for a group of brown calcium carbonate ones from one patient. All specimens were kept win the tetrasodium salt of EDTA for two months to,,~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~...:...,... remove a dark green pigmnent. They were then matrix... transferred to disodium EDTA and left to decalcify for nine and a half months. During this period no different samples matrix gav onl small pigment was removed. When they were returned to...tetrasodium EDTA, a further loss of green pigment of... ash suggesting thatany metals we c coccurred in many which was usually negligible after..e.n...s... three months. The remaining material was then well...washed in water for several weeks to remove the Fig 2 Shell of matrix (top) from cholesterol gallstone (bottom) kept in sodium chenodeoxycholate for 12 weeks. The matrix peeled off the stone and then broke up. Note diffuse areas of matrix on stone surface. Gallstone dimensions 4 x 2.7 mm. Approximate magnification in the figure x JO. but lack of uniformity in size and surface area of the specimens made comparisons difficult. In the laminated pieces, the brown matrix accounted for 1 % of their original weight and on heating gave a negligible amount of ash which was found to be calcium oxide by x-ray diffraction. Analysis figures for this material were C =58.O1 %,H= 7.39%, N %, but there was insufficient = matrix from this and the other experiments to make further quantitative estimations. However, heating Fig 3 Cross section of laminated matrix from a different samples of matrix gave only smiall amounts laminated cakcium carbonate gallstone. Dimensions of ash suggesting that any metals were trace constitu- 7 x 4.7 mm. Approximate magnification in the ents. Several metals as well as calcium could be figure x
4 490 Fig 4 Surface detail of the matrix in figure 3. Dimensions 7 x 4-8 mm. Approximate magnification in the figure xj05. EDTA solution and yellow and green pigments which were soluble in water. The brown laminated calcium carbonate stones contained a yellow-brown laminated matrix (figs 3 and 4) which accounted for 9.3 % of the original stone weight. Analysis figures for this material were C = 56-81%, H = 6.04%, N = 8.89%. The material obtained from the dissolution of the black calcium carbonate and calcium phosphate D. June Sutor and Susan E. Wooley stones was usually gel-like and almost invariably pigmented black by an apparently insoluble compound (fig 5). Occasionally, firm areas remained in the matrix suggesting a localization of insoluble pigment or other material. In general, laminated stone areas gave laminated matrix which retained its cohesiveness and shape, while granular areas broke up in the decalcifying solution and yielded many small pieces of matrix. If, however, much black pigment was present in the stone, it obscured the underlying matrix structure (fig 5). The matrix and black pigment accounted for 12-27% of the original weight of gallstones from which a predominantly soft but pigmented matrix was obtained. Carbon, hydrogen, and nitrogen analysis figures for 10 different specimens of pigmented matrix varied considerably: carbon = %, hydrogen = %, nitrogen= %. Calcium was found in four out of six matrices analysed, but accounted for only 0-3-2% of their weight. X-ray powder photographs of ashed matrices showed that other metals (not identified) could also be present in very small amounts. These pigmented matrices gave amorphous x-ray diffraction patterns. Discussion A gel-like substance, the matrix, appears to be an essential constituent of all the gallstones studied irrespective of their composition. X-ray diffraction photographs showed that it was amorphous and analysis figures suggested it was organic, since calcium and other metals which were often present Fig 5 Black pigmented matrix from gallstones containing 40 % pigment and 60% calcium phosphate. Approximate magnification in the figure x 56.
5 The organic matrix ofgallstones 491 occurred only in trace amounts. The relative proportions of carbon, hydrogen, and nitrogen could not be determined exactly because the matrix was usually pigmented when isolated under the conditions described here. Matrix from cholesterol stones was yellow, orange, or brown but very rarely black, while that from calcium carbonate and calcium phosphate varieties was black or dark brown. The colour of the matrix generally correlated with that of the stone but not with the presence or absence of trace elements. The pigments were not studied but analysis figures suggested that they too were organic compounds. This conflicts with Suzuki's view (1966) that the black insoluble pigment of gallstones is a metal complex of bilirubin but supports his earlier suggestion that it could be a high molecular weight polymer of bilirubin (Suzuki, 1965). This pigment is of particular interest since its occurrence would indicate a connexion between its formation and that of calcium phosphate and calcium carbonate. Ashing matrix showed the presence of trace amounts of apatite and calcium carbonate in some from cholesterol gallstones which had not been in a decalcifying solution. Since neither of these compounds form during this process, they must be original stone constituents bound to the matrix otherwise they would have been removed as the cholesterol dissolved, but as trace constituents, they would not be detected when the stones were analysed by x-ray diffraction (Sutor and Wooley, 1971). The amount and structure of the matrix present in the gallstones varied with the texture of the crystalline material. Matrix was associated with all crystalline textures except areas of large radiating crystals of cholesterol. Laminated stone areas gave a compact gel whose shape was an almost exact replica of the original material but loose crystalline aggregates yielded many small pieces of matrix. Estimations of the amount of matrix varied considerably, probably mainly because of contamination of this material by unknown amounts of pigments. The close structural relationship between crystalline material and matrix in gallstones would indicate that their deposition is in some way connected, but the relative roles played by each in the nucleation and growth of a stone cannot usually be deduced. The exception is stone areas consisting of radiating cholesterol crystals in which the absence of observable matrix shows that such crystal growth occurred independently of this substance. On the other hand, Womack (1971) found that in Syrian hamsters fed a diet which resulted in cholesterol gallstone formation, mucus collected in viscous masses before the appearance of crystals and the earliest stones were soft and later ones more compact. It is likely then that crystal growth from supersaturated bile can occur in the presence or absence of a matrix. Most cholesterol gallstones contain some matrix since very few consist entirely of the large radiating crystals. Although in most of our experiments this matrix tended to break up into small pieces as the crystalline material dissolved, it was not destroyed. After cholesterol gallstones have been dissolved in vivo by oral administration of chenodeoxycholic acid, should any of these pieces remain in the gallbladder, they can act as a nidus for crystal growth if the bile becomes supersaturated with respect to any compound. Furthermore, crystal growth on such a nidus will occur at a much lower level of supersaturation than is required for homogeneous nucleation and stone formation is therefore more likely to recur. A study of any matrix in gallstones formed after chenodeoxycholic acid therapy would be of great interest. We thank the Medical Research Council for financial support, Mr J. Cresswell for taking the photographs and Weddel Pharmaceuticals Ltd for providing the chenodeoxycholic acid. References Danzinger, R. G., Hofmann, A. F., Schoenfield, L. J., and Thistle, J.L. (1972). Dissolution ofcholesterol gallstones by chenodeoxycholic acid. New Engl. J. Med., 286, 1-8. King, J. S., Jr., and Boyce, W. H. (1959). Analysis of renal calculous matrix compared with some other matrix materials and with uromucoid. Arch. Biochem., 82, Sutor, D. J., and Wooley, S. E. (1971). A statistical survey of the composition of gallstones in eight countries. Gut, 12, Sutor, D. J., and Wooley, S. E. (1973). The nature and incidence of gallstones containing calcium. Gut, 14, Suzuki, N. (1965). On black pigment of gallstones with special reference to comparison with bilirubin derivatives. Tohoku J. exp. Med., 85, Suzuki, N. (1966). On bilirubin-metal complex compounds in relation to black pigments of gallstones. Tohoku J. exp. Med., 90, Womack, N. A. (1971). The development of gallstones. Surg. Gynec. Obstet., 133, Womack, N. A., Zeppa, R., and Irvin, G. L. (1963). The anatomy of gallstones. Ann. Surg., 157,
Decalcification and Clearing of Bone eg. Fibula. Salem Kharwa Clinical Anatomy Medical School
 Decalcification and Clearing of Bone eg. Fibula Salem Kharwa Clinical Anatomy Medical School Bone composition Bone consists of cells (osteocytes) surrounded by a calcified matrix containing collagen fibres.
Decalcification and Clearing of Bone eg. Fibula Salem Kharwa Clinical Anatomy Medical School Bone composition Bone consists of cells (osteocytes) surrounded by a calcified matrix containing collagen fibres.
Etiology Bacteria Sand particles Particles of ingesta / intestinal contents Desquamated epithelium
 10 CONCRETIONS Concretions Calculi o Urinary Calculi o Biliary Calculi o Salivary Calculi o Pancreatic Calculi o Enteric Calculi Piliconcretions Phytoconcretions Polyconcretions Model Questions CONCRETIONS
10 CONCRETIONS Concretions Calculi o Urinary Calculi o Biliary Calculi o Salivary Calculi o Pancreatic Calculi o Enteric Calculi Piliconcretions Phytoconcretions Polyconcretions Model Questions CONCRETIONS
Calcium carbonate in human gallstones and
 Gut, 1978, 19, 22-224 Calcium carbonate in human gallstones and total CO2 in bile D. JUNE SUTOR AND LYNETTE I. WILKIE From the Department of Chemistry, University College London, London SUMMARY Measurement
Gut, 1978, 19, 22-224 Calcium carbonate in human gallstones and total CO2 in bile D. JUNE SUTOR AND LYNETTE I. WILKIE From the Department of Chemistry, University College London, London SUMMARY Measurement
Identification and qualitative Analysis. of Renal Calculi
 Identification and qualitative Analysis of Renal Calculi 1 -Renal Calculi: Kidney stones, renal calculi or renal lithiasis (stone formation) are small, hard deposits that form inside your kidneys. The
Identification and qualitative Analysis of Renal Calculi 1 -Renal Calculi: Kidney stones, renal calculi or renal lithiasis (stone formation) are small, hard deposits that form inside your kidneys. The
Formulation and Evaluation
 Chapter-5 Formulation and Evaluation 5.1 OBJECTIVE After successful taste masking and solubility enhancement of drugs in preliminary studies, by using Mannitol Solid Dispersion, next step includes the
Chapter-5 Formulation and Evaluation 5.1 OBJECTIVE After successful taste masking and solubility enhancement of drugs in preliminary studies, by using Mannitol Solid Dispersion, next step includes the
EUROPEAN QUALIFYING EXAMINATION 2006
 EUROPEAN QUALIFYING EXAMINATION 2006 PAPER A CHEMISTRY This paper comprises: * Letter from the applicant 2006/A(Ch)/e/1-6 * Document 1 2006/A(Ch)/e/7 * Document 2 2006/A(Ch)/e/8-10 2006/A(Ch)/e - 1 - LETTER
EUROPEAN QUALIFYING EXAMINATION 2006 PAPER A CHEMISTRY This paper comprises: * Letter from the applicant 2006/A(Ch)/e/1-6 * Document 1 2006/A(Ch)/e/7 * Document 2 2006/A(Ch)/e/8-10 2006/A(Ch)/e - 1 - LETTER
What Are Gallstones? GALLSTONES. Gallstones are pieces of hard, solid matter that form over time in. the gallbladder of some people.
 What Are Gallstones? Gallstones are pieces of hard, solid matter that form over time in the gallbladder of some people. The gallbladder sits under the liver and stores bile (a key digestive juice ). Gallstones
What Are Gallstones? Gallstones are pieces of hard, solid matter that form over time in the gallbladder of some people. The gallbladder sits under the liver and stores bile (a key digestive juice ). Gallstones
Urinalysis and Body Fluids CRg CAUTION. Abnormal Crystals of Metabolic Origin. Unit 2; Session 5. Abnormal crystals
 Urinalysis and Body Fluids CRg Unit 2; Session 5 Crystals Found in the Urine Microscopic Examination - Part D, Abnormal Crystals CAUTION Abnormal crystals should not be reported haphazardly are rarely
Urinalysis and Body Fluids CRg Unit 2; Session 5 Crystals Found in the Urine Microscopic Examination - Part D, Abnormal Crystals CAUTION Abnormal crystals should not be reported haphazardly are rarely
Hydroxypropyl Starch (Tentative)
 Residue Monograph prepared by the meeting of the Joint FAO/WHO Expert Committee on Food Additives (JECFA), 82 nd meeting 2016 Hydroxypropyl Starch (Tentative) This monograph was also published in: Compendium
Residue Monograph prepared by the meeting of the Joint FAO/WHO Expert Committee on Food Additives (JECFA), 82 nd meeting 2016 Hydroxypropyl Starch (Tentative) This monograph was also published in: Compendium
points raised, and the following is an account of what I have done under touched, but my work has fallen under two main heads:
 NOTES ON CREATININE. BY P. C. COLLS, late Assistant Demonstrator in Physiology, King's College, London. (From the Physiological Laboratory, King's College, London.) ABOUT two years ago, a lengthy correspondence
NOTES ON CREATININE. BY P. C. COLLS, late Assistant Demonstrator in Physiology, King's College, London. (From the Physiological Laboratory, King's College, London.) ABOUT two years ago, a lengthy correspondence
XXVI. STUDIES ON THE INTERACTION. OF AMINO-COMPOUNDS AND CARBOHYDRATES.
 XXVI. STUDIES ON THE INTERACTION. OF AMINO-COMPOUNDS AND CARBOHYDRATES. II. THE PREPARATION OF GLUCOSE UREIDE. BY ALEXANDER HYND. From the Department of Physiology, University of St Andrews. (Received
XXVI. STUDIES ON THE INTERACTION. OF AMINO-COMPOUNDS AND CARBOHYDRATES. II. THE PREPARATION OF GLUCOSE UREIDE. BY ALEXANDER HYND. From the Department of Physiology, University of St Andrews. (Received
There are enzymes in biological washing powders. Biological washing powder has to be used at temperatures below 45 C.
 There are enzymes in biological washing powders. Biological washing powder has to be used at temperatures below 45 C. The enzymes in biological washing powders do not work on the stains on clothes at temperatures
There are enzymes in biological washing powders. Biological washing powder has to be used at temperatures below 45 C. The enzymes in biological washing powders do not work on the stains on clothes at temperatures
چهارمین کنگره مهندسی نفت ایران. Lab Investigation of Inorganic Scale Removal Using Chelating Agents and Hydrochloric Acid Solutions.
 Lab Investigation of Inorganic Scale Removal Using Chelating Agents and Hydrochloric Acid Solutions Abstract One of the most important damages to the oil and gas reservoirs, are inorganic scales resulted
Lab Investigation of Inorganic Scale Removal Using Chelating Agents and Hydrochloric Acid Solutions Abstract One of the most important damages to the oil and gas reservoirs, are inorganic scales resulted
Sokalan PA types. Sokalan PA 15 Sokalan PA 40. Sokalan PA 20 Sokalan PA 50. Sokalan PA 25 PN Granules Sokalan PA 80 S. Technical Information
 Technical Information Sokalan PA types TI/ES 1151 e July 1992 (DFC) Supersedes TI/P 3037 e dated May 1988 Sokalan PA 13 PN Sokalan PA 30 CL Sokalan PA 15 Sokalan PA 40 Sokalan PA 20 PN Sokalan PA 40 Powder
Technical Information Sokalan PA types TI/ES 1151 e July 1992 (DFC) Supersedes TI/P 3037 e dated May 1988 Sokalan PA 13 PN Sokalan PA 30 CL Sokalan PA 15 Sokalan PA 40 Sokalan PA 20 PN Sokalan PA 40 Powder
ON TEA TANNIN ISOLATED FROM GREEN TEA.
 70 [Vol. 6 ON TEA TANNIN ISOLATED FROM GREEN TEA. By MICHIYO TSUJIMIIRA. (Received September 8th., 1930). The author(1) has recently isolated Tea catechin from green tea and pro posed the following formula
70 [Vol. 6 ON TEA TANNIN ISOLATED FROM GREEN TEA. By MICHIYO TSUJIMIIRA. (Received September 8th., 1930). The author(1) has recently isolated Tea catechin from green tea and pro posed the following formula
TECHNICAL METHODS GASTRIC CONTENTS AND URINE THE IDENTIFICATION OF BARBITURATE DRUGS IN. aspirated and examined as well.
 J. clin. Path. (1950), 3, 152. TECHNICAL METHODS THE IDENTIFICATION OF BARBITURATE DRUGS IN GASTRIC CONTENTS AND URINE BY From the Pathological Depariment, Salisbury General Infirmary (RECEIVED FOR PUBLICATION
J. clin. Path. (1950), 3, 152. TECHNICAL METHODS THE IDENTIFICATION OF BARBITURATE DRUGS IN GASTRIC CONTENTS AND URINE BY From the Pathological Depariment, Salisbury General Infirmary (RECEIVED FOR PUBLICATION
Chemical Pharmaceutical Quality Control. Prof.Dr.Joumaa Al- Zehouri Damascus university Faculty of Pharmacy
 Chemical Pharmaceutical Quality Control Prof.Dr.Joumaa Al- Zehouri Damascus university Faculty of Pharmacy COMPLEXOMETRIC REACTIONS AND TITRATIONS Prof.J.Al-Zehouri 1 ml of 0.1Msodium edetate is equivalent
Chemical Pharmaceutical Quality Control Prof.Dr.Joumaa Al- Zehouri Damascus university Faculty of Pharmacy COMPLEXOMETRIC REACTIONS AND TITRATIONS Prof.J.Al-Zehouri 1 ml of 0.1Msodium edetate is equivalent
Topic Objectives. Physical Examination LECTURE MODULE 3; PHYSICAL EXAMINATION OF URINE. Appearance. Odor Specific Gravity Volume
 LECTURE MODULE 3; PHYSICAL EXAMINATION OF URINE Topic Objectives 1. Identify the colors which commonly associated with abnormal urine. 2. State two possible causes for urine turbidity in a sample that
LECTURE MODULE 3; PHYSICAL EXAMINATION OF URINE Topic Objectives 1. Identify the colors which commonly associated with abnormal urine. 2. State two possible causes for urine turbidity in a sample that
Unit 1, Section C.1. In which you will learn about: Solutions Electrolytes Saturation Solubility curves
 Unit 1, Section C.1 In which you will learn about: Solutions Electrolytes Saturation Solubility curves Some Definitions A solution is a homogeneous mixture of 2 or more substances in a single phase. One
Unit 1, Section C.1 In which you will learn about: Solutions Electrolytes Saturation Solubility curves Some Definitions A solution is a homogeneous mixture of 2 or more substances in a single phase. One
E55A GELATIN, GELLING GRADE Gelatina
 00-0PDG.pdf 0 0 0 0 EA GELATIN, GELLING GRADE Gelatina DEFINITION Purified protein obtained from collagen of animals (including fish and poultry) by partial alkaline and/or acid hydrolysis, by enzymatic
00-0PDG.pdf 0 0 0 0 EA GELATIN, GELLING GRADE Gelatina DEFINITION Purified protein obtained from collagen of animals (including fish and poultry) by partial alkaline and/or acid hydrolysis, by enzymatic
CRYSTALLINE PEPSIN V. ISOLATION OF CRYSTALLINE PEPSIN FROM BOVINE GASTRIC JUICE BY JOHN H. NORTHROP
 CRYSTALLINE PEPSIN V. ISOLATION OF CRYSTALLINE PEPSIN FROM BOVINE GASTRIC JUICE BY JOHN H. NORTHROP (From the Laboratories of The Rockefeller Institute for Medical Research, Princeton, N. J.) (Accepted
CRYSTALLINE PEPSIN V. ISOLATION OF CRYSTALLINE PEPSIN FROM BOVINE GASTRIC JUICE BY JOHN H. NORTHROP (From the Laboratories of The Rockefeller Institute for Medical Research, Princeton, N. J.) (Accepted
LAB.2. Tablet Production Methods
 LAB.2 Tablet Production Methods Dry methods Direct compression Dry granulation Wet methods Wet granulation Regardless whether tablets are made by direct compression or granulation, the first step, milling
LAB.2 Tablet Production Methods Dry methods Direct compression Dry granulation Wet methods Wet granulation Regardless whether tablets are made by direct compression or granulation, the first step, milling
Petrolatum. Stage 4, Revision 1. Petrolatum is a purified semi solid mixture of hydrocarbons obtained from petroleum.
 1 001-1208PDG.pdf Petrolatum Stage 4, Revision 1 Definition Petrolatum is a purified semi solid mixture of hydrocarbons obtained from petroleum. It may contain a suitable antioxidant. Description and Solubility
1 001-1208PDG.pdf Petrolatum Stage 4, Revision 1 Definition Petrolatum is a purified semi solid mixture of hydrocarbons obtained from petroleum. It may contain a suitable antioxidant. Description and Solubility
Experiment 2: Melting Points and the Identification of an Unknown and Cholesterol from Human Gallstones
 1 Experiment 2: Melting Points and the Identification of an Unknown and Cholesterol from Human Gallstones Part 1. Melting Points and the Identification of an Unknown Read pp 211-220, Chapter 14, (especially
1 Experiment 2: Melting Points and the Identification of an Unknown and Cholesterol from Human Gallstones Part 1. Melting Points and the Identification of an Unknown Read pp 211-220, Chapter 14, (especially
MORPHOLOGICAL AND CHEMICAL STUDY OF URINARY CALCULI
 MORPHOLOGICAL AND CHEMICAL STUDY OF URINARY CALCULI Pages with reference to book, From 300 To 303 F.D. Khan, M.S. Memon, A.F. Ansari ( Department of Biochemistry, Institute of Chemistry, University of
MORPHOLOGICAL AND CHEMICAL STUDY OF URINARY CALCULI Pages with reference to book, From 300 To 303 F.D. Khan, M.S. Memon, A.F. Ansari ( Department of Biochemistry, Institute of Chemistry, University of
URINARY CRYSTALS. by Geoffrey K. Dube and Robert S. Brown
 URINARY CRYSTALS by Geoffrey K. Dube and Robert S. Brown A 26 year-old man presents with a fever and weakness. His WBC is 133,000, with 83% blasts. Creatinine is 2.0 mg/dl and serum uric acid is 15.4 mg/dl.
URINARY CRYSTALS by Geoffrey K. Dube and Robert S. Brown A 26 year-old man presents with a fever and weakness. His WBC is 133,000, with 83% blasts. Creatinine is 2.0 mg/dl and serum uric acid is 15.4 mg/dl.
» Croscarmellose Sodium is a cross linked polymer of carboxymethylcellulose sodium.
 BRIEFING Croscarmellose Sodium, NF 22 page 2856 and page 702 of PF 30(2) [Mar. Apr. 2004]. A modification is made in the test for Degree of substitution to correct the endpoint color to agree with the
BRIEFING Croscarmellose Sodium, NF 22 page 2856 and page 702 of PF 30(2) [Mar. Apr. 2004]. A modification is made in the test for Degree of substitution to correct the endpoint color to agree with the
Biopharmaceutics Dosage form factors influencing bioavailability Lec:5
 Biopharmaceutics Dosage form factors influencing bioavailability Lec:5 Ali Y Ali BSc Pharmacy MSc Industrial Pharmaceutical Sciences Dept. of Pharmaceutics School of Pharmacy University of Sulaimani 09/01/2019
Biopharmaceutics Dosage form factors influencing bioavailability Lec:5 Ali Y Ali BSc Pharmacy MSc Industrial Pharmaceutical Sciences Dept. of Pharmaceutics School of Pharmacy University of Sulaimani 09/01/2019
UNIVERSITY OF CAMBRIDGE INTERNATIONAL EXAMINATIONS International General Certificate of Secondary Education
 UNIVERSITY OF CAMBRIDGE INTERNATIONAL EXAMINATIONS International General Certificate of Secondary Education *6062662112* CO-ORDINATED SCIENCES 0654/52 Paper 5 Practical Test October/November 2011 2 hours
UNIVERSITY OF CAMBRIDGE INTERNATIONAL EXAMINATIONS International General Certificate of Secondary Education *6062662112* CO-ORDINATED SCIENCES 0654/52 Paper 5 Practical Test October/November 2011 2 hours
A study of the histochemical demonstration of cytochrome oxidase
 497 A study of the histochemical demonstration of cytochrome oxidase By R. G. BUTCHER, J. V. DIENGDOH, and J. CHAYEN (From the Department of Pathology, Royal College of Surgeons of England, Lincoln's Inn
497 A study of the histochemical demonstration of cytochrome oxidase By R. G. BUTCHER, J. V. DIENGDOH, and J. CHAYEN (From the Department of Pathology, Royal College of Surgeons of England, Lincoln's Inn
People who have a zinc deficiency can take hydrated zinc sulfate (ZnSO 4.xH 2O) as a dietary supplement.
 Q1.Zinc forms many different salts including zinc sulfate, zinc chloride and zinc fluoride. (a) People who have a zinc deficiency can take hydrated zinc sulfate (ZnSO 4.xH 2O) as a dietary supplement.
Q1.Zinc forms many different salts including zinc sulfate, zinc chloride and zinc fluoride. (a) People who have a zinc deficiency can take hydrated zinc sulfate (ZnSO 4.xH 2O) as a dietary supplement.
Ultrastructure of Mycoplasmatales Virus laidlawii x
 J. gen. Virol. (1972), I6, 215-22I Printed in Great Britain 2I 5 Ultrastructure of Mycoplasmatales Virus laidlawii x By JUDY BRUCE, R. N. GOURLAY, AND D. J. GARWES R. HULL* Agricultural Research Council,
J. gen. Virol. (1972), I6, 215-22I Printed in Great Britain 2I 5 Ultrastructure of Mycoplasmatales Virus laidlawii x By JUDY BRUCE, R. N. GOURLAY, AND D. J. GARWES R. HULL* Agricultural Research Council,
The Amendment of Standards for Specification, Scope, Application and Limitation of Food Additives
 G/SPS/N/TPKM/136Add1 G/SPS/N/TPKM/139Add1 The Amendment of Standards for Specification, Scope, Application and Limitation of Food Additives DOH Food No. 0980400197, January 22, 2009 Appendix 1: Standards
G/SPS/N/TPKM/136Add1 G/SPS/N/TPKM/139Add1 The Amendment of Standards for Specification, Scope, Application and Limitation of Food Additives DOH Food No. 0980400197, January 22, 2009 Appendix 1: Standards
Pectins. Residue Monograph prepared by the meeting of the Joint FAO/WHO Expert Committee on Food Additives (JECFA), 82 nd meeting 2016
 Residue Monograph prepared by the meeting of the Joint FAO/WHO Expert Committee on Food Additives (JECFA), 82 nd meeting 2016 Pectins This monograph was also published in: Compendium of Food Additive Specifications.
Residue Monograph prepared by the meeting of the Joint FAO/WHO Expert Committee on Food Additives (JECFA), 82 nd meeting 2016 Pectins This monograph was also published in: Compendium of Food Additive Specifications.
PHYSICAL CHARACTERISTICS, QUALITATIVE AND QUANTITATIVE ANALYSIS OF URINARY STONES (PATHARI)
 Int. J. Chem. Sci.: 11(1), 2013, 457463 ISSN 0972768X www.sadgurupublications.com PHYSICAL CHARACTERISTICS, QUALITATIVE AND QUANTITATIVE ANALYSIS OF URINARY STONES (PATHARI) SUMAN PARIHAR a, PRASOON HADA
Int. J. Chem. Sci.: 11(1), 2013, 457463 ISSN 0972768X www.sadgurupublications.com PHYSICAL CHARACTERISTICS, QUALITATIVE AND QUANTITATIVE ANALYSIS OF URINARY STONES (PATHARI) SUMAN PARIHAR a, PRASOON HADA
Residue Monograph prepared by the meeting of the Joint FAO/WHO Expert Committee on Food Additives (JECFA), 82 nd meeting 2016.
 Residue Monograph prepared by the meeting of the Joint FAO/WHO Expert Committee on Food Additives (JECFA), 82 nd meeting 2016 Aspartame This monograph was also published in: Compendium of Food Additive
Residue Monograph prepared by the meeting of the Joint FAO/WHO Expert Committee on Food Additives (JECFA), 82 nd meeting 2016 Aspartame This monograph was also published in: Compendium of Food Additive
SUMMATIVE ASSESSMENT I, Class IX
 SUMMATIVE ASSESSMENT I, 0-6 SCIENCE Class IX Time Allowed : hours Maximum Marks : 90 General Instructions :. The question paper comprises of two Sections, A and B. You are to attempt both the sections..
SUMMATIVE ASSESSMENT I, 0-6 SCIENCE Class IX Time Allowed : hours Maximum Marks : 90 General Instructions :. The question paper comprises of two Sections, A and B. You are to attempt both the sections..
human gall bladder bile
 Gut, 1986, 27, 1382-1386 Effect of binding of ionised calcium on the in vitro nucleation of cholesterol and calcium bilirubinate in human gall bladder bile S GALLINGER, P R C HARVEY, C N PETRUNKA, AND
Gut, 1986, 27, 1382-1386 Effect of binding of ionised calcium on the in vitro nucleation of cholesterol and calcium bilirubinate in human gall bladder bile S GALLINGER, P R C HARVEY, C N PETRUNKA, AND
Minerals composition of different types of gallstones in Sudanese population
 Open Science Journal of Analytical Chemistry 2014; 1(1): 1-5 Published online January 30, 2014 (http://www.openscienceonline.com/journal/osjac) Minerals composition of different types of gallstones in
Open Science Journal of Analytical Chemistry 2014; 1(1): 1-5 Published online January 30, 2014 (http://www.openscienceonline.com/journal/osjac) Minerals composition of different types of gallstones in
Official Journal of the European Communities. (Acts whose publication is obligatory) COMMISSION DIRECTIVE 2002/82/EC. of 15 October 2002
 28.10.2002 L 292/1 I (Acts whose publication is obligatory) COMMISSION DIRECTIVE 2002/82/EC of 15 October 2002 amending Directive 96/77/EC laying down specific purity criteria on food additives other than
28.10.2002 L 292/1 I (Acts whose publication is obligatory) COMMISSION DIRECTIVE 2002/82/EC of 15 October 2002 amending Directive 96/77/EC laying down specific purity criteria on food additives other than
A Comparative Evaluation of Cross Linked Starch Urea-A New Polymer and Other Known Polymers for Controlled Release of Diclofenac
 Asian Journal of Chemistry Vol. 22, No. 6 (2010), 4239-4244 A Comparative Evaluation of Cross Linked Starch Urea-A New Polymer and Other Known Polymers for Controlled Release of Diclofenac K.P.R. CHOWDARY*
Asian Journal of Chemistry Vol. 22, No. 6 (2010), 4239-4244 A Comparative Evaluation of Cross Linked Starch Urea-A New Polymer and Other Known Polymers for Controlled Release of Diclofenac K.P.R. CHOWDARY*
Gallstones Information Leaflet THE DIGESTIVE SYSTEM. Gutscharity.org.uk
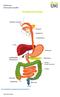 THE DIGESTIVE SYSTEM http://healthfavo.com/digestive-system-for-kids.html This factsheet is about gallstones Gall is an old-fashioned word for bile, a liquid made in the liver and stored in the gall bladder
THE DIGESTIVE SYSTEM http://healthfavo.com/digestive-system-for-kids.html This factsheet is about gallstones Gall is an old-fashioned word for bile, a liquid made in the liver and stored in the gall bladder
International Journal of Advanced Research in Biological Sciences ISSN: Research Article
 Int. J. Adv. Res. Biol.Sci. 1(7): (2014): 16 21 International Journal of Advanced Research in Biological Sciences ISSN: 2348-8069 www.ijarbs.com Research Article Microencapsulation of Vitamin C through
Int. J. Adv. Res. Biol.Sci. 1(7): (2014): 16 21 International Journal of Advanced Research in Biological Sciences ISSN: 2348-8069 www.ijarbs.com Research Article Microencapsulation of Vitamin C through
Clinical and Nutritional Study on Gallstone. Disease in Japan. Haruo Kameda, MD, Fumitake Ishihara, MD, Koji Shibata, MDand Eiichi Tsukie, MD
 Original Article Clinical and Nutritional Study on Gallstone Haruo Kameda, MD, Fumitake Ishihara, MD, Koji Shibata, MDand Eiichi Tsukie, MD The dietary factors, as a cause on increasing incidence of gallstones
Original Article Clinical and Nutritional Study on Gallstone Haruo Kameda, MD, Fumitake Ishihara, MD, Koji Shibata, MDand Eiichi Tsukie, MD The dietary factors, as a cause on increasing incidence of gallstones
Paper No.: 13 Paper Title: Food Additives Module 2. Functional Classification of Food Additives
 Paper No.: 13 Paper Title: Food Additives Module 2. Functional Classification of Food Additives 2.1 Introduction According to the Food Protection Committee of the Food and Nutrition Board, food additives
Paper No.: 13 Paper Title: Food Additives Module 2. Functional Classification of Food Additives 2.1 Introduction According to the Food Protection Committee of the Food and Nutrition Board, food additives
Most of the ethanol that is used as a biofuel in this country is produced from corn.
 Chem 251 Ethanol from Corn Most of the ethanol that is used as a biofuel in this country is produced from corn. In this experiment you will make ethanol from frozen corn kernels using a process similar
Chem 251 Ethanol from Corn Most of the ethanol that is used as a biofuel in this country is produced from corn. In this experiment you will make ethanol from frozen corn kernels using a process similar
Antacid therapy: Antacids side effects:
 Antacids Lec:5 When the general public ask why it takes antacids, the answer will include: 1.that uncomfortable feeling from overeating. 2. heart burn. 3. a growing hungry feeling between meals. Antacids
Antacids Lec:5 When the general public ask why it takes antacids, the answer will include: 1.that uncomfortable feeling from overeating. 2. heart burn. 3. a growing hungry feeling between meals. Antacids
A. History Urinalysis is the oldest lab test still being performed today
 III. THE ROUTINE URINALYSIS A. History Urinalysis is the oldest lab test still being performed today 1. Cave man noted change in urine properties associated with disease 2. Babylonians and Egyptians noted
III. THE ROUTINE URINALYSIS A. History Urinalysis is the oldest lab test still being performed today 1. Cave man noted change in urine properties associated with disease 2. Babylonians and Egyptians noted
Heparin Sodium ヘパリンナトリウム
 Heparin Sodium ヘパリンナトリウム Add the following next to Description: Identification Dissolve 1 mg each of Heparin Sodium and Heparin Sodium Reference Standard for physicochemical test in 1 ml of water, and
Heparin Sodium ヘパリンナトリウム Add the following next to Description: Identification Dissolve 1 mg each of Heparin Sodium and Heparin Sodium Reference Standard for physicochemical test in 1 ml of water, and
Practical 3 Extemporaneous Preparations Solutions. Name: Group: Date:
 Practical 3 Extemporaneous Preparations Solutions Name: Group: Date: FILTRATION Unwanted particulate matter may be removed from solutions by the process of filtration. Filtration may be either fine or
Practical 3 Extemporaneous Preparations Solutions Name: Group: Date: FILTRATION Unwanted particulate matter may be removed from solutions by the process of filtration. Filtration may be either fine or
Paper No.: 07 Paper Title: TECHNOLOGY OF MILK AND MILK PRODUCTS Module 32: Technology of dairy byproducts-1: Caseinates
 Paper No.: 07 Paper Title: TECHNOLOGY OF MILK AND MILK PRODUCTS Module 32: Technology of dairy byproducts-1: Caseinates INTRODUCTION With a content of 0.7-0.9% phosphorus, covalently bound to the casein
Paper No.: 07 Paper Title: TECHNOLOGY OF MILK AND MILK PRODUCTS Module 32: Technology of dairy byproducts-1: Caseinates INTRODUCTION With a content of 0.7-0.9% phosphorus, covalently bound to the casein
Dr.N Damodharan Professor and Head. Department of Pharmaceutics SRM College of Pharmacy
 Dr.N Damodharan Professor and Head Department of Pharmaceutics SRM College of Pharmacy 1 Definition: - Medicines prepared according to the formulae of Galen. - A medicinal preparation composed mainly of
Dr.N Damodharan Professor and Head Department of Pharmaceutics SRM College of Pharmacy 1 Definition: - Medicines prepared according to the formulae of Galen. - A medicinal preparation composed mainly of
THERMALLY OXIDIZED SOYA BEAN OIL
 THERMALLY OXIDIZED SOYA BEAN OIL Prepared at the 39th JECFA (1992), published in FNP 52 Add 1 (1992). Metals and arsenic specifications revised at the 55th JECFA (2000). An ADI of 0-3 mg/kg bw was established
THERMALLY OXIDIZED SOYA BEAN OIL Prepared at the 39th JECFA (1992), published in FNP 52 Add 1 (1992). Metals and arsenic specifications revised at the 55th JECFA (2000). An ADI of 0-3 mg/kg bw was established
CHEMO-IMMUNOLOGICAL STUDIES ON CONJUGATED CARBOHYDRATE-PROTEINS IV. Tm~ SX~TI~SlS of Tm~ p-amn~obenzx~ ETm~R OF THE SOLUBLE
 Published Online: 1 September, 1931 Supp Info: http://doi.org/10.1084/jem.54.3.431 Downloaded from jem.rupress.org on October 31, 2018 CHEMO-IMMUNOLOGICAL STUDIES ON CONJUGATED CARBOHYDRATE-PROTEINS IV.
Published Online: 1 September, 1931 Supp Info: http://doi.org/10.1084/jem.54.3.431 Downloaded from jem.rupress.org on October 31, 2018 CHEMO-IMMUNOLOGICAL STUDIES ON CONJUGATED CARBOHYDRATE-PROTEINS IV.
COMMISSION DIRECTIVE 2011/3/EU
 18.1.2011 Official Journal of the European Union L 13/59 DIRECTIVES COMMISSION DIRECTIVE 2011/3/EU of 17 January 2011 amending Directive 2008/128/EC laying down specific purity criteria on colours for
18.1.2011 Official Journal of the European Union L 13/59 DIRECTIVES COMMISSION DIRECTIVE 2011/3/EU of 17 January 2011 amending Directive 2008/128/EC laying down specific purity criteria on colours for
INTERMEDIATE 1 1 Food and Diet. These elements are present in compounds - not as free elements.
 INTERMEDIATE 1 1 Food and Diet FOOD AND DIET The main elements present in the human body are: Hydrogen Oxygen Nitrogen Carbon These elements are present in compounds - not as free elements. Unlike plants,
INTERMEDIATE 1 1 Food and Diet FOOD AND DIET The main elements present in the human body are: Hydrogen Oxygen Nitrogen Carbon These elements are present in compounds - not as free elements. Unlike plants,
CHAPTER 5 CRYSTALLISATION OF HYDRATED AND ANHYDROUS URIC ACID AND AMMONIUM ACID URATE CRYSTALS IN GEL
 61 CHAPTER 5 CRYSTALLISATION OF HYDRATED AND ANHYDROUS URIC ACID AND AMMONIUM ACID URATE CRYSTALS IN GEL 5.1 INTRODUCTION Uric acid is an important constituent of human stones. Under normal circumstances
61 CHAPTER 5 CRYSTALLISATION OF HYDRATED AND ANHYDROUS URIC ACID AND AMMONIUM ACID URATE CRYSTALS IN GEL 5.1 INTRODUCTION Uric acid is an important constituent of human stones. Under normal circumstances
INTERNATIONAL ŒNOLOGICAL CODEX. BENTONITES Bentonita N SIN: 558 (Oeno 11/2003 modified Oeno )
 BENTONITES Bentonita N SIN: 558 (Oeno 11/2003 modified Oeno 441-2011) 1. OBJECT, ORIGIN AND FIELD OF APPLICATION Bentonites are hydrous aluminium silicates belonging to the montmorillonite group. The brute
BENTONITES Bentonita N SIN: 558 (Oeno 11/2003 modified Oeno 441-2011) 1. OBJECT, ORIGIN AND FIELD OF APPLICATION Bentonites are hydrous aluminium silicates belonging to the montmorillonite group. The brute
COLOUR REACTIONS ATTRIBUTED
 LXIV. COLOUR REACTIONS ATTRIBUTED TO VITAMIN A. BY FRANCIS HOWARD CARR AND ERNEST ARTHUR PRICE. From the Laboratories of The British Drug Houses, Ltd., Graham Street, City Road, N. 1. (Received March 17th,
LXIV. COLOUR REACTIONS ATTRIBUTED TO VITAMIN A. BY FRANCIS HOWARD CARR AND ERNEST ARTHUR PRICE. From the Laboratories of The British Drug Houses, Ltd., Graham Street, City Road, N. 1. (Received March 17th,
Excretory System. Biology 2201
 Excretory System Biology 2201 Excretory System How does the excretory system maintain homeostasis? It regulates: Body heat Water-salt concentrations Acid-base concentrations Metabolite concentrations ORGANS
Excretory System Biology 2201 Excretory System How does the excretory system maintain homeostasis? It regulates: Body heat Water-salt concentrations Acid-base concentrations Metabolite concentrations ORGANS
Excretory System. Excretory System
 Excretory System Biology 2201 Excretory System How does the excretory system maintain homeostasis? It regulates: Body heat Water-salt concentrations Acid-base concentrations Metabolite concentrations 1
Excretory System Biology 2201 Excretory System How does the excretory system maintain homeostasis? It regulates: Body heat Water-salt concentrations Acid-base concentrations Metabolite concentrations 1
Organic Molecule Composition of Milk: Lab Investigation
 Name: Organic Molecule Composition of Milk: Lab Investigation Introduction & Background Milk & milk products have been a major food source from earliest recorded history. Milk is a natural, nutritionally
Name: Organic Molecule Composition of Milk: Lab Investigation Introduction & Background Milk & milk products have been a major food source from earliest recorded history. Milk is a natural, nutritionally
EXPERIMENT 13 QUALITATIVE ANALYSIS
 PURPOSE: 1. To separate and identify each cation in a mixture of Ag +, Cu 2+, Zn 2+, and Ca 2+ cations. 2. To identify the cations present in three individually assigned unknown mixtures that may contain
PURPOSE: 1. To separate and identify each cation in a mixture of Ag +, Cu 2+, Zn 2+, and Ca 2+ cations. 2. To identify the cations present in three individually assigned unknown mixtures that may contain
Name: Enzymes. Class: Date: 101 minutes. Time: 100 marks. Marks: Level 1,2 and 3. Increasing demand. Comments:
 Enzymes Name: Class: Date: Time: 0 minutes Marks: 00 marks Comments: Level,2 and 3. Increasing demand Q. Catalase is an enzyme. Catalase controls the following reaction: hydrogen peroxide water + oxygen
Enzymes Name: Class: Date: Time: 0 minutes Marks: 00 marks Comments: Level,2 and 3. Increasing demand Q. Catalase is an enzyme. Catalase controls the following reaction: hydrogen peroxide water + oxygen
ELECTRON MICROSCOPY OF A SMALL PIGMENTED CUTANEOUS LESION*
 ELECTRON MICROSCOPY OF A SMALL PIGMENTED CUTANEOUS LESION* The description of the lesion in the title of this rcport is intentionally non-committal. Diagnosed clinically as a lentigo, it was removed as
ELECTRON MICROSCOPY OF A SMALL PIGMENTED CUTANEOUS LESION* The description of the lesion in the title of this rcport is intentionally non-committal. Diagnosed clinically as a lentigo, it was removed as
DESIGN AND EVALUATION OF CONTROLLED RELEASE MATRIX TABLETS OF FLURBIPROFEN
 Int. J. Chem. Sci.: 10(4), 2012, 2199-2208 ISSN 0972-768X www.sadgurupublications.com DESIGN AND EVALUATION OF CONTROLLED RELEASE MATRIX TABLETS OF FLURBIPROFEN K. V. R. N. S. RAMESH *, B. HEMA KIRNAMAYI
Int. J. Chem. Sci.: 10(4), 2012, 2199-2208 ISSN 0972-768X www.sadgurupublications.com DESIGN AND EVALUATION OF CONTROLLED RELEASE MATRIX TABLETS OF FLURBIPROFEN K. V. R. N. S. RAMESH *, B. HEMA KIRNAMAYI
REVISION OF MONOGRAPH ON TABLETS. Tablets
 March 2011 REVISION OF MONOGRAPH ON TABLETS Final text for addition to The International Pharmacopoeia This monograph was adopted by the Forty-fourth WHO Expert Committee on Specifications for Pharmaceutical
March 2011 REVISION OF MONOGRAPH ON TABLETS Final text for addition to The International Pharmacopoeia This monograph was adopted by the Forty-fourth WHO Expert Committee on Specifications for Pharmaceutical
7. SUMMARY, CONCLUSION AND RECOMMENDATIONS
 211 7. SUMMARY, CONCLUSION AND RECOMMENDATIONS Drug absorption from the gastro intestinal tract can be limited by various factors with the most common one being poor aqueous solubility and poor permeability
211 7. SUMMARY, CONCLUSION AND RECOMMENDATIONS Drug absorption from the gastro intestinal tract can be limited by various factors with the most common one being poor aqueous solubility and poor permeability
College of Medicine, Newcastle-upon-Tyne.)
 GLUCOSE ABSORPTION IN THE RENAL TUBULES OF THE FROG. BY G. A. CLARK. (From the Physiological Laboratory of the University of Durham College of Medicine, Newcastle-upon-Tyne.) OPINION is divided on the
GLUCOSE ABSORPTION IN THE RENAL TUBULES OF THE FROG. BY G. A. CLARK. (From the Physiological Laboratory of the University of Durham College of Medicine, Newcastle-upon-Tyne.) OPINION is divided on the
KIDNEY STONE ANALYSIS
 KIDNEY STONE ANALYSIS Lesson aim To familiarise students with the use of chemical analysis in a healthcare related context, and to give students an idea of how the results can be used in diagnosis. Lesson
KIDNEY STONE ANALYSIS Lesson aim To familiarise students with the use of chemical analysis in a healthcare related context, and to give students an idea of how the results can be used in diagnosis. Lesson
RBM dental implants by CST Process Technical Release September 2012
 Page 1 of 7 RBM dental implants by CST Process Technical Release September 2012 Summary Finish Line operates technologically validated RBM (Resorbable Blasting Media) Process, based on its CST (Comprehensive
Page 1 of 7 RBM dental implants by CST Process Technical Release September 2012 Summary Finish Line operates technologically validated RBM (Resorbable Blasting Media) Process, based on its CST (Comprehensive
Corn/Maize Starch. Speci cations
 Corn/Maize Starch Maize starch also known as Corn starch. It is extracted from the endosperm of the corn kernel and has a distinctive appearance. Maize starch in natural, modified and dextrinised forms
Corn/Maize Starch Maize starch also known as Corn starch. It is extracted from the endosperm of the corn kernel and has a distinctive appearance. Maize starch in natural, modified and dextrinised forms
ANIMAL OILS AND FATS CHAPTER 9 CHOLESTEROL 1. COMPOSITION. Oxygen Carbon Hydrogen
 120 ANIMAL OILS AND FATS CHAPTER 9 CHOLESTEROL 1. COMPOSITION 455. BY WEIGHT BY VOLUME 1 Oxygen 3.025 100 1.00 Carbon 85.095 2813 36.77 Hydrogen 11.880 392.7 63.03 2. PHYSICAL PROPERTIES 456. It is a solid
120 ANIMAL OILS AND FATS CHAPTER 9 CHOLESTEROL 1. COMPOSITION 455. BY WEIGHT BY VOLUME 1 Oxygen 3.025 100 1.00 Carbon 85.095 2813 36.77 Hydrogen 11.880 392.7 63.03 2. PHYSICAL PROPERTIES 456. It is a solid
PRINCIPLE OF URINALYSIS
 PRINCIPLE OF URINALYSIS Vanngarm Gonggetyai Objective Can explain : the abnormalities detected in urine Can perform : routine urinalysis Can interprete : the results of urinalysis Examination of urine
PRINCIPLE OF URINALYSIS Vanngarm Gonggetyai Objective Can explain : the abnormalities detected in urine Can perform : routine urinalysis Can interprete : the results of urinalysis Examination of urine
Discussion CHAPTER - 5
 CHAPTER - 5 Discussion The chapter deals with discussion of the results. The thesis ends with this chapter. The chapter interpretates and discusses the results of the investigation on the physical properties
CHAPTER - 5 Discussion The chapter deals with discussion of the results. The thesis ends with this chapter. The chapter interpretates and discusses the results of the investigation on the physical properties
UNIVERSITY OF CAMBRIDGE INTERNATIONAL EXAMINATIONS International General Certificate of Secondary Education CO-ORDINATED SCIENCES
 Centre Number Candidate Number Name UNIVERSITY OF CAMBRIDGE INTERNATIONAL EXAMINATIONS International General Certificate of Secondary Education CO-ORDINATED SCIENCES 0654/05 Paper 5 Practical Test October/November
Centre Number Candidate Number Name UNIVERSITY OF CAMBRIDGE INTERNATIONAL EXAMINATIONS International General Certificate of Secondary Education CO-ORDINATED SCIENCES 0654/05 Paper 5 Practical Test October/November
ANIMAL OILS AND FATS CHAPTER 10 ÉTHAL 1 1. COMPOSITION 479. BY WEIGHT BY VOLUME 2
 126 ANIMAL OILS AND FATS CHAPTER 10 ÉTHAL 1 1. COMPOSITION 479. BY WEIGHT BY VOLUME 2 Oxygen 6.2888 100.00 1.00 Carbon 79.7660 1268.40 16.60 Hydrogen 13.9452 221.74 35.54 which is equivalent to: Ethylene..
126 ANIMAL OILS AND FATS CHAPTER 10 ÉTHAL 1 1. COMPOSITION 479. BY WEIGHT BY VOLUME 2 Oxygen 6.2888 100.00 1.00 Carbon 79.7660 1268.40 16.60 Hydrogen 13.9452 221.74 35.54 which is equivalent to: Ethylene..
6.02 Uniformity of Dosage Units
 6.02 Uniformity of Dosage Units Change 1. Content Uniformity, 3. Criteria and Table 6.02-2 as follows: 1. Content Uniformity Select not less than 30 units, and proceed as follows for the dosage form designated.
6.02 Uniformity of Dosage Units Change 1. Content Uniformity, 3. Criteria and Table 6.02-2 as follows: 1. Content Uniformity Select not less than 30 units, and proceed as follows for the dosage form designated.
C 6 H 12 O 6 + 6O 2 6CO 2 + 6H 2 O
 Sample Questions for the Individual Round Exam 1. Glucose is the most basic sugar involved in human metabolism. Its structure is provided below: a. The overall reaction of glucose metabolism is given below.
Sample Questions for the Individual Round Exam 1. Glucose is the most basic sugar involved in human metabolism. Its structure is provided below: a. The overall reaction of glucose metabolism is given below.
Measurement of cholesterol gallstone growth in vitro
 Measurement of cholesterol gallstone growth in vitro A. Andre van den Berg, 1 Jaap D. van Buul, J. Donald Ostrow, 2 and Albert K. Groen Department of Gastrointestinal and Liver Diseases, Academic Medical
Measurement of cholesterol gallstone growth in vitro A. Andre van den Berg, 1 Jaap D. van Buul, J. Donald Ostrow, 2 and Albert K. Groen Department of Gastrointestinal and Liver Diseases, Academic Medical
ANIMALS OILS AND FATS CHAPTER 2 PREPARATION AND SAPONIFICATION OF SPERMACETI 1. PREPARATION OF SPERMACETI
 182 ANIMALS OILS AND FATS CHAPTER 2 PREPARATION AND SAPONIFICATION OF SPERMACETI 1. PREPARATION OF SPERMACETI 690. The spermaceti I examined was separated as follows from a yellow colored oil which commercial
182 ANIMALS OILS AND FATS CHAPTER 2 PREPARATION AND SAPONIFICATION OF SPERMACETI 1. PREPARATION OF SPERMACETI 690. The spermaceti I examined was separated as follows from a yellow colored oil which commercial
Amendment of Standards for Specification, Scope, Application and Limitation of Food Additives
 G/SPS/N/TPKM/147Add.1 Amendment of Standards for Specification, Scope, Application and Limitation of Food Additives DOH Food No. 0980403340, April 24, 2009 Appendix 1: Standards for Scope, Application
G/SPS/N/TPKM/147Add.1 Amendment of Standards for Specification, Scope, Application and Limitation of Food Additives DOH Food No. 0980403340, April 24, 2009 Appendix 1: Standards for Scope, Application
Water, Vitamins, & Minerals
 Water, Vitamins, & Minerals Intro to Water, Vitamins & Minerals 1. What is the main function of vitamins and minerals? To regulate BODY FUNCTIONS 2. What foods are many of the vitamins and minerals we
Water, Vitamins, & Minerals Intro to Water, Vitamins & Minerals 1. What is the main function of vitamins and minerals? To regulate BODY FUNCTIONS 2. What foods are many of the vitamins and minerals we
CIE Biology GCSE 7: Human nutrition
 CIE Biology GCSE 7: Human nutrition Notes Humans need many different nutrients to survive. To receive these nutrients in the correct quantities, a balanced diet must be eaten. A balanced diet includes
CIE Biology GCSE 7: Human nutrition Notes Humans need many different nutrients to survive. To receive these nutrients in the correct quantities, a balanced diet must be eaten. A balanced diet includes
Chapter 2 Part 3: Organic and Inorganic Compounds
 Chapter 2 Part 3: Organic and Inorganic Compounds Objectives: 1) List the major groups of inorganic chemicals common in cells. 2) Describe the functions of various types of inorganic chemicals in cells.
Chapter 2 Part 3: Organic and Inorganic Compounds Objectives: 1) List the major groups of inorganic chemicals common in cells. 2) Describe the functions of various types of inorganic chemicals in cells.
Final text for addition to The International Pharmacopoeia (June 2010)
 June 2010 KANAMYCIN ACID SULFATE: Final text for addition to The International Pharmacopoeia (June 2010) This monograph was adopted at the Forty-fourth WH Expert Committee on Specifications for Pharmaceutical
June 2010 KANAMYCIN ACID SULFATE: Final text for addition to The International Pharmacopoeia (June 2010) This monograph was adopted at the Forty-fourth WH Expert Committee on Specifications for Pharmaceutical
THERMALLY OXIDIZED SOYA BEAN OIL interacted with MONO- and DIGLYCERIDES of FATTY ACIDS
 THERMALLY OXIDIZED SOYA BEAN OIL interacted with MONO- and DIGLYCERIDES of FATTY ACIDS Prepared at the 39th JECFA (1992), published in FNP 52 Add 1 (1992). Metals and arsenic specifications revised at
THERMALLY OXIDIZED SOYA BEAN OIL interacted with MONO- and DIGLYCERIDES of FATTY ACIDS Prepared at the 39th JECFA (1992), published in FNP 52 Add 1 (1992). Metals and arsenic specifications revised at
TEACHER/LECTURER GUIDE
 TEACHER/LECTURER GUIDE Type and purpose of activity This experiment can be used to: provide evidence for assessment of Outcome 3 (For advice on marking Outcome 3 report, please contact the SAPS Scotland
TEACHER/LECTURER GUIDE Type and purpose of activity This experiment can be used to: provide evidence for assessment of Outcome 3 (For advice on marking Outcome 3 report, please contact the SAPS Scotland
THE RING STRUCTURE OF THYMIDINE
 THE RING STRUCTURE OF THYMIDINE BY P. A. LEVENE AND R. STUART TIPSON (From the Laboratories of The Rockefeller Institute for Medical Research, New York) (Received for publication, March 13, 1935) The 2-desoxy-ribose
THE RING STRUCTURE OF THYMIDINE BY P. A. LEVENE AND R. STUART TIPSON (From the Laboratories of The Rockefeller Institute for Medical Research, New York) (Received for publication, March 13, 1935) The 2-desoxy-ribose
Describing and interpreting gross lesions. Prepared for VPM 4600, May 2018; Shannon Martinson
 Describing and interpreting gross lesions Prepared for VPM 4600, May 2018; Shannon Martinson How to Describe (and Interpret) Lesions Step 1 Step 2 Step 3 Step 4 Look at the specimen: Is it normal or abnormal
Describing and interpreting gross lesions Prepared for VPM 4600, May 2018; Shannon Martinson How to Describe (and Interpret) Lesions Step 1 Step 2 Step 3 Step 4 Look at the specimen: Is it normal or abnormal
Metabolism of echitamine and plumbagin in rats
 J. Biosci., Vol. 3, Number 4, December 1981, pp. 395-400. Printed in India. Metabolism of echitamine and plumbagin in rats B. CHANDRASEKARAN and B. NAGARAJAN Microbiology Division, Cancer Institute, Madras
J. Biosci., Vol. 3, Number 4, December 1981, pp. 395-400. Printed in India. Metabolism of echitamine and plumbagin in rats B. CHANDRASEKARAN and B. NAGARAJAN Microbiology Division, Cancer Institute, Madras
1. Gastric Emptying Time Anatomically, a swallowed drug rapidly reaches the stomach. Eventually, the stomach empties its content in the small
 Lecture-5 1. Gastric Emptying Time Anatomically, a swallowed drug rapidly reaches the stomach. Eventually, the stomach empties its content in the small intestine. Because the duodenum has the greatest
Lecture-5 1. Gastric Emptying Time Anatomically, a swallowed drug rapidly reaches the stomach. Eventually, the stomach empties its content in the small intestine. Because the duodenum has the greatest
Core practical 14: Investigate the effect of gibberellin on the production of amylase in germinating cereals using a starch agar assay
 Core practical 14 Teacher sheet Core practical 14: Investigate the effect of gibberellin on the production of amylase in germinating cereals using a starch agar assay Objectives To investigate the effect
Core practical 14 Teacher sheet Core practical 14: Investigate the effect of gibberellin on the production of amylase in germinating cereals using a starch agar assay Objectives To investigate the effect
Evaluation of in-vitro anti-urolithiasis activity of Nerunjil kudineer
 INTERNATIONAL JOURNAL OF CURRENT RESEARCH IN BIOLOGY AND MEDICINE ISSN: 2455-944X www.darshanpublishers.com DOI:10.22192/ijcrbm Volume 3, Issue 5-2018 Original Research Article DOI: http://dx.doi.org/10.22192/ijcrbm.2018.03.05.020
INTERNATIONAL JOURNAL OF CURRENT RESEARCH IN BIOLOGY AND MEDICINE ISSN: 2455-944X www.darshanpublishers.com DOI:10.22192/ijcrbm Volume 3, Issue 5-2018 Original Research Article DOI: http://dx.doi.org/10.22192/ijcrbm.2018.03.05.020
Proteins their functions and uses revision 3
 Proteins their functions and uses revision 3 59 minutes 59 marks Page of 23 Q. The diagram gives information about some parts of the human digestive system. (a) (i) Name the organ which makes bile. Label
Proteins their functions and uses revision 3 59 minutes 59 marks Page of 23 Q. The diagram gives information about some parts of the human digestive system. (a) (i) Name the organ which makes bile. Label
Making a plastic from potato starch
 Making a plastic from potato starch In this activity students make a plastic from potato starch and investigate the effect that adding a plasticiser has on the properties of the polymer that they make.
Making a plastic from potato starch In this activity students make a plastic from potato starch and investigate the effect that adding a plasticiser has on the properties of the polymer that they make.
RICINOLEATE UPON BACTERIA
 A COMPARATIVE STUDY OF THE ACTION OF SODIUM RICINOLEATE UPON BACTERIA From the Division of Laboratories and Research, New York State Department of Health, Albany Received for publication, May 14, 1928
A COMPARATIVE STUDY OF THE ACTION OF SODIUM RICINOLEATE UPON BACTERIA From the Division of Laboratories and Research, New York State Department of Health, Albany Received for publication, May 14, 1928
Wettable Magnesium Stearate. What Are Customers Looking for in Selecting Pharmaceutical Lubricants?
 Wettable Magnesium Stearate Presented By: Richard Pudlo P.E. Principal Chemical Engineer April 29 th, 2015 What Are Customers Looking for in Selecting Pharmaceutical Lubricants? Meet USP/.NF monograph
Wettable Magnesium Stearate Presented By: Richard Pudlo P.E. Principal Chemical Engineer April 29 th, 2015 What Are Customers Looking for in Selecting Pharmaceutical Lubricants? Meet USP/.NF monograph
Inhibitive properties of some chelates and their ligands in acid corrosion of zinc
 Int. J. Corros. Scale Inhib., 2014, 3, no. 3, 204 214 Inhibitive properties of some chelates and their ligands in acid corrosion of zinc V. P. Grigor ev, 1. A. Belousova, 1 E. V. Plekhanova 1 and A.S.
Int. J. Corros. Scale Inhib., 2014, 3, no. 3, 204 214 Inhibitive properties of some chelates and their ligands in acid corrosion of zinc V. P. Grigor ev, 1. A. Belousova, 1 E. V. Plekhanova 1 and A.S.
