Prevalence of Theileria equi and Babesia caballi
|
|
|
- Evan Chase
- 6 years ago
- Views:
Transcription
1 Onderstepoort Journal of Veterinary Research, 75: (2008) Prevalence of Theileria equi and Babesia caballi infections in horses belonging to resource-poor farmers in the north-eastern Free State Province, South Africa M.Y. MOTLOANG 1, 2 *, O.M.M. THEKISOE 3, A. ALHASSAN 3, M. BAKHEIT 1, M.P. MOTHEO 1, F.E.S. MASANGANE 1, M.L. THIBEDI 1, N. INOUE 3, I. IGARASHI 3, C. SUGIMOTO 4 and P.A. MBATI 1 ABSTRACT MOTLOANG, M.Y., THEKISOE, M.M.O., ALHASSAN, A., BAKHEIT, M., MOTHEO, M.P., MASAN- GANE, F.E.S., THIBEDI, M.L., INOUE, N., IGARASHI, I., SUGIMOTO, C. & MBATI, P.A Prevalence of Theileria equi and Babesia caballi infections in horses belonging to resource-poor farmers in the north-eastern Free State, South Africa. Onderstepoort Journal of Veterinary Research, 75: The prevalence of Theileria equi and Babesia caballi infections in the north-eastern Free State Province of South Africa was determined by examination of thin and thick Giemsa-stained blood smears, IFAT and PCR. No parasites were detected by microscopy from any blood samples collected at five study sites, Qwaqwa, Kestell, Harrismith, Vrede and Warden. Of the tested serum samples, 28/29 (96.5 %), 20/21 (95.2 %) and 42/42 (100 %) were positive by IFAT for T. equi infections in Harrismith, Kestell and Qwaqwa, respectively, and 5/29 (17.2 %), 13/21 (61.9 %) and 30/42 (71.4 %) were sero-positive for B. caballi infections in Harrismith, Kestell and Qwaqwa, respectively. All DNA samples from the study sites were negative for B. caballi infections by PCR, but five samples, two from each of Kestell and Warden and one from Vrede, were PCR positive for T. equi infections. The high prevalence of antibodies against T. equi and B. caballi in the sampled horses indicates that the animals had been exposed to T. equi and B. caballi infections but the absence of parasitaemia and very low number of positive PCR samples, however, imply that T. equi and B. caballi are endemically stable in the north-eastern Free State Province. Keywords: Babesia caballi, Free State, Theileria equi INTRODUCTION Equine piroplasmosis is a tick-borne disease affecting horses, mules, donkeys and zebras worldwide * Author to whom correspondence is to be directed. motloangm@arc.agric.za 1 Parasitology Research Programme, University of the Free State, Qwaqwa Campus, Phuthaditjhaba, 9866 South Africa 2 ARC-Onderstepoort Veterinary Institute, Parasitology Division, Onderstepoort, 0110 South Africa 3 National Research Center for Protozoan Diseases, Obihiro University of Agriculture and Veterinary Medicine, Obihiro, Hokkaido, , Japan 4 Center for Zoonosis Control, Hokkaido University, Sapporo, Hokkaido, Japan Accepted for publication 14 April 2008 Editor (De Waal 2000) caused by two haemoprotozoan parasites, Theileria equi and Babesia caballi. The disease can be acute, sub acute or chronic and is characterized by fever, anaemia, icterus, hepatomegally, splenomegally and, in some cases, death (Friedhoff & Soule 1996; Knowles 1996). Most clinical cases of equine piroplasmosis in southern Africa are caused by T. equi, which usually shows a high prevalence, while B. caballi infection is usually not clinically apparent and usually has a lower prevalence (De Waal 1995, 2000; Zweygarth, Lopez- Rebollar, Nurton & Guthrie 2002). Horses infected with T. equi and/or B. caballi may remain carriers for long periods and act as sources of infection for ticks, which in turn act as vectors of the disease (De Waal 2000; Zweygarth et al. 2002). In South Africa, the 141
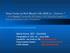
2 Theileria equi and Babesia caballi infections in horses in north-eastern Free State Province, South Africa Vrede Free State Province Thabo Mofutsanyane District Warden Kestell Harrismith Lesotho N Qwaqwa N FIG. 1 Maps of the Free State Province and the Thabo Mofutsanyane District Municipality showing the location of the five study sites: Harrismith, Kestell, Qwaqwa, Vrede and Warden. The maps were obtained from the website of the Municipality Demarcation Board of the Republic of South Africa ( ixodid tick, Rhipicephalus evertsi evertsi is the main vector of T. equi and B. caballi (De Waal & Potgieter 1987). Theileria equi and B. caballi infections are usually diagnosed by microscopic examination of Giemsaor Wright-stained blood films (Saal 1964), but these methods have poor sensitivity during very low parasitaemia. Serological assays, such as enzyme-linked immunosorbent assay (ELISA) and immunofluorescent antibody test (IFAT) are also commonly used for detection of T. equi and B. caballi infections (Bose, Jorgensen, Dalgleish, Friedhoff & De Vos 1995; Bruning 1996; Weiland 1986) but cannot distinguish between current and past infections due to persistence of antibodies even when parasites are cleared from the animal. PCR is the commonly used molecular technique for the diagnosis of T. equi and B. caballi and has been reported to be highly specific and sensitive (Bashiruddin, Camma & Rebelo 1999; Rampersad, Cesar, Campbell, Samlal & Ammons 2003). The prevalence of some of the tick-borne diseases, poultry diseases and helminth infections in domestic livestock of resource-poor farmers in the northeastern Free State Province has recently been reported, as well as the ticks occurring in the region (Hlatshwayo, Mbati & Dipeolu 2002; Mbati, Hlatshwayo, Mtshali, Mogaswane, De Waal & Dipeolu 2002; Nyaile, Thekisoe, Bisschop & Mbati 2003; Thekisoe, Mbati & Bisschop 2003; Tsotetsi & Mbati 2003; Mtshali, De Waal & Mbati 2004). The current study was aimed at documenting T. equi and B. caballi infections in horses in the north-eastern Free State Province by conventional parasitological methods, IFAT and DNA amplification by PCR. A questionnaire survey in order to assess the perceptions, attitudes and expectations of farmers in the study area with regard to T. equi and B. caballi infections was also conducted. MATERIALS AND METHODS Description of the study area The north-eastern Free State Province includes towns situated in the Thabo Mofutsanyane district municipality which borders Lesotho on the south, Motheo, Lejweleputswa and northern Free State district municipalities on the south-west, west and north, respectively, while Mpumalanga and KwaZulu- Natal provinces are on the north-east and eastern borders, respectively. Blood samples were collected between August 2003 and October 2005 from horses of 38 resource-poor farmers located in five study sites within the north-eastern Free State: Harrismith (four farms), Kestell (three farms), Qwa qwa (16 villages), Vrede (11 farms) and Warden (four farms). Fig. 1 shows the exact locations of the study sites within Thabo Mofutsanyane district. Collection of blood samples A total of 122 blood samples were collected from horses at the five localities, viz. 29 from Harrismith, 21 from Kestell, 42 from Qwaqwa, 19 from Vrede 142
3 M.Y. MOTLOANG et al. and 11 from Warden. Ten milliliters of blood were collected in both silicone and EDTA-coated vacutainers by venipuncture of the jugular vein, and were kept in a cooler-box before transport to the laboratory. Blood samples were also collected from seven horses known to be infected with T. equi parasites from Kaalplaas farm of the Onderstepoort Veterinary Institute (OVI), Pretoria, and were used as controls for T. equi. Preparation of thin and thick blood smears Thin and thick blood smears were prepared from the unclotted blood in the EDTA-coated vacutainers and stained with Giemsa as described by De Waal (1999). The parasitaemia was estimated by counting parasitized red blood cells (RBC) in eight different fields consisting of an average of 800 red blood cells per field using a formula in which the number of infected RBC is divided by the total number of uninfected RBC x 100. A smear was considered negative based on the criterion described by De Waal & Potgieter (1987), i.e. smears that reveal no parasites after 5 min examination are judged to be negative. Serum preparation and DNA extraction The blood in silicone-coated vacutainers was left for 24 h at room temperature and then centrifuged at 200 x g for 5 min (De Waal & Potgieter 1987). The resulting sera were then placed in cryogenic vials and stored at 35 C until used. Genomic DNA was extracted from horse blood by the phenol-chloroform method (Sambrook & Russel 2001) with minor modifications. Briefly, 400 μl of blood was treated with five units of TE (10 mm Tris-HCl [ph 7.4], 1 mm EDTA) buffer and then centrifuged at x g for 2 min. The resulting pellet was washed five times in TE, removing the erythrocyte ghost layer with every wash, and re-suspended in 400 μl of distilled water. After incubation at 37 C for 1 h with 100 μg/ml of proteinase K, 1 % SDS and 1 % sarcosine, DNA was extracted with phenol-chloroform and precipitated with ethanol at 34 C, and was finally dissolved in 20 μl distilled water. Indirect immunofluorescent test (IFAT) Antigen slides, conjugate and control sera were obtained from the OVI. Batches of antigen slides were removed from storage at 20 C, incubated at 37 C for 10 min and then fixed in cold acetone at 20 C. Two-fold dilutions, of the test and control sera, starting at 1/40, 1/80 and 1/160, were prepared using PBS (Tenter & Friedhoff 1986). A drop (~ 4 μl) of each diluted serum was pipetted into each respective circle of the antigen slide, incubated at 37 C for 30 min and then washed off with PBS. A second wash was done using distilled water and the slides were allowed to air dry, after which 2 μl of conjugate dilution was added and incubated at 37 C for 30 min. The conjugate was washed in PBS for 10 min and after air-drying, mounting fluid consisting of 50 % glycerin and 50 % PBS was placed on each slide and covered with 24 x 50 mm cover slip. The slides were observed under a fluorescent microscope using a 50x water objective (Tenter & Fried hoff 1986). Test readings were ranked as negative for slides with no specific fluorescence when observed under the fluorescent microscope, positive for those that presented strong fluorescence at a serum dilution of 1/80 and positive/negative for those that showed a weak fluorescence (OVI 1999). Polymerase chain reaction (PCR) A primer pair targeting the EMA1 gene (Accession no. AY058899) of T. equi with a product of 743 bp was used for amplification of T. equi DNA, and a primer pair designed from the Bc48 gene (Accession no. AB017700) with a product of 179 bp were used for amplification of B. caballi DNA (Table 1). The PCR was conducted in a total volume of 50 μl containing PCR buffer (10 mm Tris-HCl (ph 8.3), 50 mm KCl, 1.5 mm MgCl 2 ), 2 mm each of the 4 dntps, 5 pmol of each primer and five units of AmpliTaq Gold DNA polymerase (Applied Biosystems Japan Ltd., Tokyo, Japan). The reaction mixture was heated at 94 C for 10 min (denaturation step) and subjected to 35 cycles each at 94 C for 45 s, 60 C for TABLE 1 PCR primers used for amplification of T. equi and B. caballi DNA Parasite Target gene Primer sequence Theileria equi EMA1 F: 5 GCATCCATTGCCATTTCGAG3 R: 5 TGCGCCATAGACGGAGAAGC3 Babesia caballi Bc48 F: 5 CGGCTGCTATGGTTATTCAG3 R: 5 AGAGTGCAACCGAGCAATGC3 143
4 Theileria equi and Babesia caballi infections in horses in north-eastern Free State Province, South Africa 1 min (annealing), and 2 min at 72 C with a final extension at 72 C for 7 min. The PCR products were electrophoresed on a 1 % agarose gel, stained with ethidium bromide and visualized under UV light. The positive PCR products were purified using the QIAquick gel extraction Kit (Qiagen, USA). The nucleic acid sequence was determined with the BigDye terminator cycle sequencing kit (Applied Biosystems, Japan). Questionnaire survey Questionnaires were completed by a total of 23 farmers in Harrismith, Kestell and Qwaqwa. The aim of the survey was to determine the level of knowledge of resource poor farmers in the north-eastern Free State Province concerning equine piroplasmosis, ranging from knowledge of its cause and transmission, whether they could recognize the clinical signs and whether they had knowledge on control measures for the disease. RESULTS Gene amplification by PCR All the blood samples from Kaalplaas were PCR pos itive for T. equi, while only five samples (two from Kestell, two from Warden and one from Vrede) were PCR positive for this parasite (Fig. 2). These PCRpositive products were sequenced and the sequence fragments were identical ranging from % nu cleic acid sequence identities, with other EMA1 genes in the NCBI GenBank database determined by BLASTn search (data not shown) ( nlm.nih.gov/blast). All the north-eastern Free State Province and Kaalplaas DNA samples were PCR neg ative for B. caballi using primers targeting the Bc48 gene. Questionnaire survey None of the farmers in the study sites who completed questionnaires had any scientific knowledge on equine piroplasmosis. Forty-eight percent of them admitted observing clinical signs such as nasal discharge where affected animals appeared to suffocate from accumulation of mucus with occasional Microscopy and IFAT Neither T. equi nor B. caballi was detected in the thick and thin blood smears made from the blood on samples from Harrismith, Kestell, Qwaqwa, Vrede and Warden (Table 2). However, antibodies against T. equi and B. caballi were detected by IFAT in the blood of some of the horses in Harrismith, Kestell and Qwaqwa. Out of a total of 99 samples from these three study sites, 98 % were T. equi positive and 48 % were B. caballi positive (Table 2), while 58 % had mixed infections. Theileria equi was detected in three of the seven horses from Kaalplaas in both thick and thin smears, which were used as a positive control group for T. equi, while serology showed the presence of antibodies against T. equi in all of them (Table 2). 700 bp 500 bp 100 bp FIG. 2 M Five DNA samples positive by PCR for Theileria equi using EMA1 primers. Lane M: 100 bp marker; Lanes 1 2: Kestell samples; Lane 3: Vrede sample; Lanes 4 5: Warden samples; Lane +: Positive control (T. equi USDA strain); and Lane : Negative control (B. caballi USDA strain) TABLE 2 Microscopic, IFAT and PCR results for detection of T. equi and B. caballi infections Study site No. of samples Microscopy (%) IFAT (%) PCR (%) T. equi B. caballi T. equi B. caballi T. equi B. caballi Harrismith Kestell Qwaqwa Vrede Warden Kaalplaas (43) 28 (96.5) 20 (95.2) 42 (100) ND* ND 7 (100) 5 (17.2) 13 (61.9) 30 (71.4) ND ND 3 (43) 2 (9.5) 1 (5.2) 2 (18.1) 7 (100) ND* Not done 144
5 M.Y. MOTLOANG et al. sneezing, but these were signs which could not necessarily be linked to equine piroplasmosis. All of them indicated that they believed ticks did have an effect on their livestock s health although they could not specifically single out the disease(s) they transmitted. A common inexpensive method of tick control used by those who owned a relatively small number of livestock included burning the grass of the grazing area in winter while dipping in an acaricide was done by those who owned a larger numbers of animals. DISCUSSION In this study, neither T. equi nor B. caballi was detected by microscopy in the samples from Qwaqwa, Harrismith, Kestell, Vrede and Warden. Beaver, Jung & Cupp (1984) stated that blood smears are useful for studying the morphological changes of blood cells and blood parasites. However, the main disadvantage of this method is that the volume of blood used for preparing the smears is small, making the detection of a low parasitaemia and of carrier animals difficult (Ambrosio & De Waal 1990). Demon stration of parasites by microscopy was only successful in three of the seven control horses from Kaalplaas which had a known history of T. equi infections. Indirect immunofluorescent test is the most widely used serological diagnostic test. As shown in Table 2, the prevalence of antibodies against T. equi in the samples from Kestell, Harrismith and Qwaqwa was higher than that of B. caballi. Results of the current study correspond to those of a previous seroepidemiological survey of T. equi and B. caballi in the Northern and Eastern Cape provinces in which it was found that the prevalence of T. equi infections was relatively higher than that of B. caballi (Gummow, De Wet & De Waal 1996). Moreover, T. equi parasites are reported to propagate faster than B. caballi (Holman, Frerichs, Chieves & Wagner 1993), which also explains why the prevalence and pathogenicity of T. equi is higher than those of B. caballi in endemic areas (Schein 1988; De Waal 2000; Alhas san, Pumidonming, Okamura, Hirata, Battse t- seg, Fujisaki, Yokoyama & Igarashi 2005). All the DNA samples from the study areas were negative for B. caballi infections by PCR. Babesia caballi generally produces a low parasitaemia (Potgieter, De Waal & Posnett 1992; Holman et al. 1993) which makes it extremely difficult to detect in blood smears and even DNA probes have been reported to detect B. caballi-specific DNA at irregular intervals during the clinical course of an infection (Posnett & Ambrosio 1991; Holman et al. 1993). The respective sequences of T. equi were identical to other EMA1 genes of T. equi in the Genbank database, thereby confirming that our results are not falsely positive. The two PCR-positive animals from Kestell also showed the presence of antibodies by IFAT but parasitaemia was not within detectable levels by microscopic examination. The current study has demonstrated a high prevalence of antibodies against T. equi and B. caballi in the horses by IFAT. Although the presence of antibodies did not distinguish current from previous infections, these results do, however, indicate that the horses in the study region are exposed to T. equi and B. caballi infections. However, it can be concluded that five T. equi PCR-positive animals were still harbouring the parasites, although at a very low parasitaemia level, while the remainder might have recovered from the infections before the sampling period. Furthermore, the questionnaire survey revealed that these animals had never been treated for equine piroplasmosis, which suggests that they are possibly randomly re-exposed to T. equi and B. caballi infections several times during their lifetime as the vector tick (R. evertsi evertsi) is ever present in the region (Hlatshwayo et al. 2002; Mbati et al. 2002). The PCR method, with its high specificity, can be used in confirmatory diagnosis while IFAT is useful in large-scale epidemiological surveys. In conclusion, this study has demonstrated the existence of T. equi and B. caballi in the north-eastern Free State Province which confirms previous reports that this tick-borne disease is widespread in South Africa (De Waal 1995; Gummow et al. 1996). However, the negative Giemsa-stained blood smears, high incidence of positive serological assay, presence of the tick vector (Hlatshwayo et al. 2002; Mbati et al. 2002) and absence of clinical signs from the horses imply that the study region is endemically stable for T. equi and B. caballi infections. Data from this study can serve as a basis for future largescale epidemiological studies on equine piroplasmosis in this region. There is a need for scientific education for resource-poor farmers on diseases of economic and veterinary importance as this can improve their overall control. ACKNOWLEDGEMENTS The authors are grateful to the horse owners in the study sites for their co-operation. We thank Mr J. Komape; Ms M. Sentle; Mr A. Spickett and Mrs A. Spickett from the OVI and Dr B. Visser, University of 145
6 Theileria equi and Babesia caballi infections in horses in north-eastern Free State Province, South Africa the Free State, for their technical support and training in diagnostic assays. This study received financial support from two grants made available to Prof. P.A. Mbati (University of the Free State, Qwaqwa Campus [Senate Research Grant ] and The National Research Foundation [GUN ]) as well as one grant, the Japanese Society for the Promotion of Science Asia-Africa Platform grant made available to Prof. C. Sugimoto (Hokkaido University) and Prof. N. Inoue (Obihiro University). REFERENCES ALHASSAN, A., PUMIDONMING, W., OKAMURA, M., HIRATA, H., BATTSETSEG, B., FUJISAKI, K., YOKOYAMA, N. & IGARASHI, I Development of a single-round and multiplex PCR method for the simultaneous detection of Babesia caballi and Babesia equi in horse blood. Veterinary Parasitology, 129: AMBROSIO, R.E. & DE WAAL, D.T Diagnosis of parasitic diseases. Revue Scientific et Technique Office Internationale des Épizooties, 9: BASHIRUDDIN, J.B., CAMMA, C. & REBELO, E Molecular detection of Babesia equi and Babesia caballi in horse blood by PCR amplification of parts of the 16S rrna gene. Veterinary Parasitology, 84: BEAVER, P.C., JUNG, R.C. & CUPP, E.W Technical appendix, in Clinical Parasitology 9 th ed., Philadelphia: Lea and Febiger. BOSE, R., JORGENSEN, W.K., DALGLEISH, R.J., FRIEDHOFF, K.T. & DE VOS, A.J Current status and future trends in the diagnosis of babesiosis. Veterinary Parasitology, 57: BRUNING, A Equine piroplasmosis: an update on diagnosis, treatment and prevention. British Veterinary Journal, 152: DE WAAL, D.T. & POTGIETER, F.T The transtadial transmission of Babesia caballi by Rhipicephalus evertsi evertsi. Onderstepoort Journal of Veterinary Research, 54: DE WAAL, D.T Distribution, transmission and serodiagnosis of Babesia equi and Babesia caballi in South Africa. Ph.D. thesis, University of Pretoria. DE WAAL, D.T Indirect Fluorescent Antibody Test for Babesia caballi and Theileria equi. Standard Operating Procedure, Laboratory Test Methods. Onderstepoort Veterinary Institute, Division of Parasitology. DE WAAL, D.T Global importance of piroplasmosis. Journal of Protozoology Research, 10: FRIEDHOFF, K.T. & SOULE, C An account on equine babesiosis. Review Scientific et Technique Office Inter nationale des Épizooties, 15: GUMMOW, B., DE WET, C.S. & DE WAAL, D.T A seroepidemiological survey of equine piroplasmosis in the northern and eastern Cape Provinces of South Africa. Journal of the South African Veterinary Association, 67: HLATSHWAYO, M., MBATI, P.A. & DIPEOLU, O.O Seasonal abundace of adult ixodid ticks infesting cattle belonging to resource-limited farmers in the north-eastern Free State Province of South Africa. Onderstepoort Journal of Vet erinary Research, 69:1 6. HOLMAN, P.J., FRERICHS, W.M., CHIEVES, L. & WAGNER, G.G Culture confirmation of the carrier status of Babesia caballi-infected horses. Journal of Clinical Microbiology, 31: KNOWLES, D.P Control of Babesia equi parasitemia. Parasitology Today, 12: MBATI, P.A., HLATSHWAYO, M., MTSHALI, M.S., MOGA SWA- NE, K.R., DE WAAL, D.T. & DIPEOLU, O.O Tick and tick-borne diseases of livestock belonging to resource-poor farmers in the eastern Free State. Experimental and Applied Acarology, 28: MTSHALI, M.S., DE WAAL, D.T. & MBATI, P.A A seroepidemiological survey of blood parasites in cattle in the north-eastern Free State, South Africa. Onderstepoort Journal of Veterinary Research, 71: NYAILE, S.F.C., THEKISOE, M.M.O., BISSCHOP, S.P.R. & MBATI, P.A A diagnostic survey of avian parasitic infections from village poultry in Qwa-Qwa, South Africa. Journal of Protozoology Research, 13: POSNETT, E.S. & AMBROSIO, R.E DNA probes for the detection of Babesia caballi. Parasitology, 102: POTGIETER, F.T., DE WAAL, D.T. & POSNETT, E.S Transmission and diagnosis of equine babesiosis in South Africa. Memorias do Instituto Oswaldo Cruz, 87: RAMPERSAD, J., CESAR, E., CAMPBELL, M.D., SAMLAL, M. & AMMONS, D A field evaluation of PCR for the routine detection of Babesia equi in horses. Veterinary Parasitology, 114: SAAL, J.R Giemsa stain for the diagnosis of bovine babesiosis. II. Changes in erythrocytes infected with Babesia bigemina and B. argentina. Journal of Protozoology, 11: SAMBROOK, J. & RUSSELL, D.W Preparation and analysis of eukaryotic genomic DNA, in Molecular cloning, 3 rd ed., edited by J. Sambrook & D.W. Russell. Cold Spring Harbor, New York: Cold Spring Harbor Laboratory Press. SCHEIN, E Equine babesiosis, in Babesiosis of domestic animals and man, edited by M. Ristic. Boca Raton: CRC Press. TENTER, A.M. & FRIEDHOFF, K.T Serodiagnosis of experimental and natural Babesia equi and B. caballi infections. Veterinary Parasitology, 20: THEKISOE, M.M.O., MBATI, P.A. & BISSCHOP, S.P.R Diseases of free ranging chickens in the Qwa-Qwa district of the north-eastern Free State province of South Africa. Journal of the South African Veterinary Association, 74: TSOTETSI, A.M. & MBATI, P.A Parasitic helminths of veterinary importance in cattle, sheep and goats on communal farms in the northeastern Free State, South Africa. Journal of the South African Veterinary Association, 72: WEILAND, G Species-specific serodiagnosis of equine piroplasma infections by means of complement fixation test (CFT), immunofluorescence (IIF), and enzyme-linked immunosorbent assay (ELISA). Veterinary Parasitology, 20: ZWEYGARTH, E., LOPEZ-REBOLLAR, L., NURTON, J. & GUTH RIE, A.J Culture, isolation and propagation of Babesia caballi from naturally infected horses. Parasitology Research, 88:
Comparison of light microscopy and nested PCR assay in detecting of malaria mixed species infections in an endemic area of Iran
 Comparison of light microscopy and nested PCR assay in detecting of malaria mixed species infections in an endemic area of Iran Aliehsan Heidari, Manizheh Nourian, Hossein Keshavarz Associate Prof. Dept.
Comparison of light microscopy and nested PCR assay in detecting of malaria mixed species infections in an endemic area of Iran Aliehsan Heidari, Manizheh Nourian, Hossein Keshavarz Associate Prof. Dept.
Effect of diminazene block treatment on live redwater vaccine reactions
 Onderstepoort Journal of Veterinary Research, 71:113 117 (2004) Effect of diminazene block treatment on live redwater vaccine reactions M.P. COMBRINK 1 and P.C. TROSKIE Parasitology Division, Onderstepoort
Onderstepoort Journal of Veterinary Research, 71:113 117 (2004) Effect of diminazene block treatment on live redwater vaccine reactions M.P. COMBRINK 1 and P.C. TROSKIE Parasitology Division, Onderstepoort
Laboratory diagnosis of congenital infections
 Laboratory diagnosis of congenital infections Laboratory diagnosis of HSV Direct staining Tzanck test Immunostaining HSV isolation Serology PCR Tzanck test Cell scrape from base of the lesion smear on
Laboratory diagnosis of congenital infections Laboratory diagnosis of HSV Direct staining Tzanck test Immunostaining HSV isolation Serology PCR Tzanck test Cell scrape from base of the lesion smear on
Serodiagnosis of Canine Babesiosis
 Serodiagnosis of Canine Babesiosis Xuenan XUAN, DVM, PhD National Research Center for Protozoan Diseases Obihiro University of Agriculture and Veterinary Medicine Obihiro, Hokkaido 080-8555, Japan Tel.:+81-155-495648;
Serodiagnosis of Canine Babesiosis Xuenan XUAN, DVM, PhD National Research Center for Protozoan Diseases Obihiro University of Agriculture and Veterinary Medicine Obihiro, Hokkaido 080-8555, Japan Tel.:+81-155-495648;
Kakekhani et al / Archives of Razi Institute, Vol. 72, No. 1 (2017) INTRODUCTION
 52 Kakekhani et al / Archives of Razi Institute, Vol. 72, No. 1 (2017) 51-55 INTRODUCTION Equine piroplasmosis, caused by Theileria equi (recently re-classified from Babesia equi (Mehlhorn and Schein,
52 Kakekhani et al / Archives of Razi Institute, Vol. 72, No. 1 (2017) 51-55 INTRODUCTION Equine piroplasmosis, caused by Theileria equi (recently re-classified from Babesia equi (Mehlhorn and Schein,
. MONITORING THEILERIA ON THE BASIS OF MICROSCOPIC EXAMINATION
 . MONITORING THEILERIA ON THE BASIS OF MICROSCOPIC EXAMINATION 3.1 Introduction Theileria is a vector borne protozoan parasite that causes theileriosis which is a fatal disease for cows. It is a disease
. MONITORING THEILERIA ON THE BASIS OF MICROSCOPIC EXAMINATION 3.1 Introduction Theileria is a vector borne protozoan parasite that causes theileriosis which is a fatal disease for cows. It is a disease
EVALUATION OF AN ENZYME LINKED IMMUNOSORBENT ASSAY-KIT FOR THE DETECTION OF BABESIA BOVIS-ANTIBODIES IN CATTLE IN ARGENTINA
 EVALUATION OF AN ENZYME LINKED IMMUNOSORBENT ASSAY-KIT FOR THE DETECTION OF BABESIA BOVIS-ANTIBODIES IN CATTLE IN ARGENTINA S.T. DE ECHAIDE*, I.E. ECHAIDE*, A.B. GAIDO**, A.J. MANGOLD*, C.I. LUGARESI*,
EVALUATION OF AN ENZYME LINKED IMMUNOSORBENT ASSAY-KIT FOR THE DETECTION OF BABESIA BOVIS-ANTIBODIES IN CATTLE IN ARGENTINA S.T. DE ECHAIDE*, I.E. ECHAIDE*, A.B. GAIDO**, A.J. MANGOLD*, C.I. LUGARESI*,
XUENAN XUAN, ALEJANDRA LARSEN, HIROMI IKADAI, TETSUYA TANAKA, IKUO IGARASHI, HIDEYUKI NAGASAWA, KOZO FUJISAKI, YUTAKA TOYODA, NAOYOSHI SUZUKI,
 JOURNAL OF CLINICAL MICROBIOLOGY, Feb. 2001, p. 705 709 Vol. 39, No. 2 0095-1137/01/$04.00 0 DOI: 10.1128/JCM.39.2.705 709.2001 Copyright 2001, American Society for Microbiology. All Rights Reserved. Expression
JOURNAL OF CLINICAL MICROBIOLOGY, Feb. 2001, p. 705 709 Vol. 39, No. 2 0095-1137/01/$04.00 0 DOI: 10.1128/JCM.39.2.705 709.2001 Copyright 2001, American Society for Microbiology. All Rights Reserved. Expression
The introduction of carrier animals into areas where tick vectors are prevalent can lead to an epizootic spread of the disease.
 Equine piroplasmosis is a tick-borne protozoal disease of horses, mules, donkeys and zebra. The aetiological agents are blood parasites named Theileria equi and Babesia caballi. Theileria equi was previously
Equine piroplasmosis is a tick-borne protozoal disease of horses, mules, donkeys and zebra. The aetiological agents are blood parasites named Theileria equi and Babesia caballi. Theileria equi was previously
Human Immunodeficiency Virus-1 (HIV-1) Genemer. Primer Pair for amplification of HIV-1 Specific DNA Fragment
 Product Manual Human Immunodeficiency Virus-1 (HIV-1) Genemer Primer Pair for amplification of HIV-1 Specific DNA Fragment Catalog No.: 60-2002-10 Store at 20 o C For research use only. Not for use in
Product Manual Human Immunodeficiency Virus-1 (HIV-1) Genemer Primer Pair for amplification of HIV-1 Specific DNA Fragment Catalog No.: 60-2002-10 Store at 20 o C For research use only. Not for use in
Recommended laboratory tests to identify influenza A/H5 virus in specimens from patients with an influenza-like illness
 World Health Organization Recommended laboratory tests to identify influenza A/H5 virus in specimens from patients with an influenza-like illness General information Highly pathogenic avian influenza (HPAI)
World Health Organization Recommended laboratory tests to identify influenza A/H5 virus in specimens from patients with an influenza-like illness General information Highly pathogenic avian influenza (HPAI)
Identification of Microbes Lecture: 12
 Diagnostic Microbiology Identification of Microbes Lecture: 12 Electron Microscopy 106 virus particles per ml required for visualization, 50,000-60,000 magnification normally used. Viruses may be detected
Diagnostic Microbiology Identification of Microbes Lecture: 12 Electron Microscopy 106 virus particles per ml required for visualization, 50,000-60,000 magnification normally used. Viruses may be detected
Animal Disease Reporting for the month August 2017
 Animal Disease Reporting for the month August 2017 Sub-Directorate Epidemiology Directorate Animal Health 01 November 2017 Introduction According to the Animal Diseases Act, 1984 (Act 35 of 1984) Controlled
Animal Disease Reporting for the month August 2017 Sub-Directorate Epidemiology Directorate Animal Health 01 November 2017 Introduction According to the Animal Diseases Act, 1984 (Act 35 of 1984) Controlled
HIV-1 Genemer Detection Kit Ready to Use Amplification Kit for HIV-1 Specific DNA Fragment Analysis
 Product Manual HIV-1 Genemer Detection Kit Ready to Use Amplification Kit for HIV-1 Specific DNA Fragment Analysis For research use only. Not for use in diagnostic procedures for clinical purposes Catalog
Product Manual HIV-1 Genemer Detection Kit Ready to Use Amplification Kit for HIV-1 Specific DNA Fragment Analysis For research use only. Not for use in diagnostic procedures for clinical purposes Catalog
Isolation and identification of Mycoplasma gallisepticum in chickensbn from industrial farms in Kerman province
 Available online at http://www.ijabbr.com International journal of Advanced Biological and Biomedical Research Volume 2, Issue 1, 2014: 100-104 Isolation and identification of Mycoplasma gallisepticum
Available online at http://www.ijabbr.com International journal of Advanced Biological and Biomedical Research Volume 2, Issue 1, 2014: 100-104 Isolation and identification of Mycoplasma gallisepticum
Received 23 March 2005/Accepted 19 April 2005
 JOURNAL OF CLINICAL MICROBIOLOGY, Aug. 2005, p. 3755 3759 Vol. 43, No. 8 0095-1137/05/$08.00 0 doi:10.1128/jcm.43.8.3755 3759.2005 Copyright 2005, American Society for Microbiology. All Rights Reserved.
JOURNAL OF CLINICAL MICROBIOLOGY, Aug. 2005, p. 3755 3759 Vol. 43, No. 8 0095-1137/05/$08.00 0 doi:10.1128/jcm.43.8.3755 3759.2005 Copyright 2005, American Society for Microbiology. All Rights Reserved.
Role of Paired Box9 (PAX9) (rs ) and Muscle Segment Homeobox1 (MSX1) (581C>T) Gene Polymorphisms in Tooth Agenesis
 EC Dental Science Special Issue - 2017 Role of Paired Box9 (PAX9) (rs2073245) and Muscle Segment Homeobox1 (MSX1) (581C>T) Gene Polymorphisms in Tooth Agenesis Research Article Dr. Sonam Sethi 1, Dr. Anmol
EC Dental Science Special Issue - 2017 Role of Paired Box9 (PAX9) (rs2073245) and Muscle Segment Homeobox1 (MSX1) (581C>T) Gene Polymorphisms in Tooth Agenesis Research Article Dr. Sonam Sethi 1, Dr. Anmol
Chromatin IP (Isw2) Fix soln: 11% formaldehyde, 0.1 M NaCl, 1 mm EDTA, 50 mm Hepes-KOH ph 7.6. Freshly prepared. Do not store in glass bottles.
 Chromatin IP (Isw2) 7/01 Toshi last update: 06/15 Reagents Fix soln: 11% formaldehyde, 0.1 M NaCl, 1 mm EDTA, 50 mm Hepes-KOH ph 7.6. Freshly prepared. Do not store in glass bottles. 2.5 M glycine. TBS:
Chromatin IP (Isw2) 7/01 Toshi last update: 06/15 Reagents Fix soln: 11% formaldehyde, 0.1 M NaCl, 1 mm EDTA, 50 mm Hepes-KOH ph 7.6. Freshly prepared. Do not store in glass bottles. 2.5 M glycine. TBS:
Equine Importance Piroplasmosis Last Updated: Etiology Species Affected Geographic Distribution Transmission page 1 of 5
 Equine Piroplasmosis Equine Babesiosis, Equine Theileriosis, Biliary Fever Last Updated: August 29, 2008 Importance Equine piroplasmosis is a tick-borne protozoal infection of horses. Piroplasmosis may
Equine Piroplasmosis Equine Babesiosis, Equine Theileriosis, Biliary Fever Last Updated: August 29, 2008 Importance Equine piroplasmosis is a tick-borne protozoal infection of horses. Piroplasmosis may
Hepatitis B Virus Genemer
 Product Manual Hepatitis B Virus Genemer Primer Pair for amplification of HBV Viral Specific Fragment Catalog No.: 60-2007-10 Store at 20 o C For research use only. Not for use in diagnostic procedures
Product Manual Hepatitis B Virus Genemer Primer Pair for amplification of HBV Viral Specific Fragment Catalog No.: 60-2007-10 Store at 20 o C For research use only. Not for use in diagnostic procedures
Mitochondrial DNA Isolation Kit
 Mitochondrial DNA Isolation Kit Catalog Number KA0895 50 assays Version: 01 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Background... 3 General Information... 4 Materials
Mitochondrial DNA Isolation Kit Catalog Number KA0895 50 assays Version: 01 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Background... 3 General Information... 4 Materials
IDENTIFICATION OF CRYPTOSPORIDIUM PARVUM GENOTYPE FROM HIV AND NON-HIV FECAL SAMPLES BY PCR
 IDENTIFICATION OF CRYPTOSPORIDIUM PARVUM GENOTYPE FROM HIV AND NON-HIV FECAL SAMPLES BY PCR Zulhainan Hamzah 1, Songsak Petmitr 2, Mathirut Mungthin 3 and Porntip Chavalitshewinkoon-Petmitr 1 1 Department
IDENTIFICATION OF CRYPTOSPORIDIUM PARVUM GENOTYPE FROM HIV AND NON-HIV FECAL SAMPLES BY PCR Zulhainan Hamzah 1, Songsak Petmitr 2, Mathirut Mungthin 3 and Porntip Chavalitshewinkoon-Petmitr 1 1 Department
Aggregation of Sphingosine-DNA and cell construction using components from egg white
 Integrative Molecular Medicine Research Article ISSN: 2056-6360 Aggregation of Sphingosine-DNA and cell construction using components from egg white Shoshi Inooka* The Institute of Japan Applied Food Materials
Integrative Molecular Medicine Research Article ISSN: 2056-6360 Aggregation of Sphingosine-DNA and cell construction using components from egg white Shoshi Inooka* The Institute of Japan Applied Food Materials
PARASITOLOGY CASE HISTORY #14 (BLOOD PARASITES) (Lynne S. Garcia)
 PARASITOLOGY CASE HISTORY #14 (BLOOD PARASITES) (Lynne S. Garcia) A 37-year-old woman, who had traveled to New Guinea for several weeks, presented to the medical clinic with fever, chills, and rigors within
PARASITOLOGY CASE HISTORY #14 (BLOOD PARASITES) (Lynne S. Garcia) A 37-year-old woman, who had traveled to New Guinea for several weeks, presented to the medical clinic with fever, chills, and rigors within
Product # Kit Components
 3430 Schmon Parkway Thorold, ON, Canada L2V 4Y6 Phone: (905) 227-8848 Fax: (905) 227-1061 Email: techsupport@norgenbiotek.com Pneumocystis jirovecii PCR Kit Product # 42820 Product Insert Background Information
3430 Schmon Parkway Thorold, ON, Canada L2V 4Y6 Phone: (905) 227-8848 Fax: (905) 227-1061 Email: techsupport@norgenbiotek.com Pneumocystis jirovecii PCR Kit Product # 42820 Product Insert Background Information
Parasitology. Helminthology (Helminths)
 Parasitology Protozoology (Protozoa) Helminthology (Helminths) Entomology (Arthropodes) Platyhelminthes (flat worms) Nematheminthes (round worms) Trematodes Nematodes Cestodes Collection of the specimens
Parasitology Protozoology (Protozoa) Helminthology (Helminths) Entomology (Arthropodes) Platyhelminthes (flat worms) Nematheminthes (round worms) Trematodes Nematodes Cestodes Collection of the specimens
VETERINARSKI ARHIV 80 (6), , Vet. arhiv 80, , 2010.
 VETERINARSKI ARHIV 80 (6), 715-722, 2010 Immunochromatographic assay of Babesia caballi and Babesia equi Laveran 1901 (Theileria equi Mehlhorn and Schein, 1998) (Phylum Apicomplexa) infection in Philippine
VETERINARSKI ARHIV 80 (6), 715-722, 2010 Immunochromatographic assay of Babesia caballi and Babesia equi Laveran 1901 (Theileria equi Mehlhorn and Schein, 1998) (Phylum Apicomplexa) infection in Philippine
Better Training for Safer Food BTSF
 Better Training for Safer Food BTSF Importation of vector-borne infectious diseases Tanguy Marcotty Institute of Tropical Medicine Importation routes Trade Infected live animals (e.g. H5N1 avian influenza)
Better Training for Safer Food BTSF Importation of vector-borne infectious diseases Tanguy Marcotty Institute of Tropical Medicine Importation routes Trade Infected live animals (e.g. H5N1 avian influenza)
Report on the Foot and Mouth Disease outbreak in KwaZulu Natal. 20 May 2011
 Directorate of Animal Health, Private Bag X138, Pretoria,0001. Tel: (012) 319 7470, Fax: (012) 329 7218, E-mail: DAH@daff.gov.za Report on the Foot and Mouth Disease outbreak in KwaZulu Natal 1). History:
Directorate of Animal Health, Private Bag X138, Pretoria,0001. Tel: (012) 319 7470, Fax: (012) 329 7218, E-mail: DAH@daff.gov.za Report on the Foot and Mouth Disease outbreak in KwaZulu Natal 1). History:
E.Z.N.A. SQ Blood DNA Kit II. Table of Contents
 E.Z.N.A. SQ Blood DNA Kit II Table of Contents Introduction and Overview...2 Kit Contents/Storage and Stability...3 Blood Storage and DNA Yield...4 Preparing Reagents...5 100-500 μl Whole Blood Protocol...6
E.Z.N.A. SQ Blood DNA Kit II Table of Contents Introduction and Overview...2 Kit Contents/Storage and Stability...3 Blood Storage and DNA Yield...4 Preparing Reagents...5 100-500 μl Whole Blood Protocol...6
Rapid differentiation of mycobacterium tuberculosis and mycobacterium leprae from sputum by polymerase chain reaction
 Original Article Nepal Medical College Journal 27; 9(1): Rapid differentiation of mycobacterium tuberculosis and mycobacterium leprae from sputum by polymerase chain reaction Bishwa Raj Sapkota, Chaman
Original Article Nepal Medical College Journal 27; 9(1): Rapid differentiation of mycobacterium tuberculosis and mycobacterium leprae from sputum by polymerase chain reaction Bishwa Raj Sapkota, Chaman
Title. CitationJapanese Journal of Veterinary Research, 63(3): 129- Issue Date DOI. Doc URL. Type. File Information. specific antibodies
 Title Diagnostic application of recombinant equine merozoi specific antibodies Author(s)Kumar, Sandeep; Rakha, Naresh K.; Goyal, Liza; Goel, CitationJapanese Journal of Veterinary Research, 63(3): 129-
Title Diagnostic application of recombinant equine merozoi specific antibodies Author(s)Kumar, Sandeep; Rakha, Naresh K.; Goyal, Liza; Goel, CitationJapanese Journal of Veterinary Research, 63(3): 129-
Detection of Toxoplasma Parasitemia by PCR: Does it Correlate with IgG and IgM Antibody Titers?
 Detection of Toxoplasma Parasitemia by : Does it Correlate with IgG and IgM Antibody Titers? Behzad Haghpanah 1, Mansoor Salehi 2, Shahram Sadri 1 1 Department of Parasitology and Mycology, 2 Department
Detection of Toxoplasma Parasitemia by : Does it Correlate with IgG and IgM Antibody Titers? Behzad Haghpanah 1, Mansoor Salehi 2, Shahram Sadri 1 1 Department of Parasitology and Mycology, 2 Department
Validation Report: VERSA Mini PCR Workstation Reverse Transcription of Avian Flu RNA and Amplification of cdna & Detection of H5N1
 I. Objectives Validation Report: VERSA Mini PCR Workstation Reverse Transcription of Avian Flu RNA and Amplification of cdna & Detection of H5N1 1. To ensure stability of RNA (highly thermolabile and degradatively
I. Objectives Validation Report: VERSA Mini PCR Workstation Reverse Transcription of Avian Flu RNA and Amplification of cdna & Detection of H5N1 1. To ensure stability of RNA (highly thermolabile and degradatively
Why to vaccinate? Lumpy skin disease prevention, control, and awareness workshop Budapest, Hungary, 7-9 March 2017
 1 Vaccination against Lumpy skin disease virus Eeva Tuppurainen, DVM, MSc, PhD, MRCVS Lumpy skin disease scientific expert FAO Regional Office for Europe and Central Asia 2 Why to vaccinate? Feasible control
1 Vaccination against Lumpy skin disease virus Eeva Tuppurainen, DVM, MSc, PhD, MRCVS Lumpy skin disease scientific expert FAO Regional Office for Europe and Central Asia 2 Why to vaccinate? Feasible control
More than 98 countries affected by Leishmaniasis
 More than 98 countries affected by Leishmaniasis ACL In many urban & sub urban areas (8 provinces) Tehran, Mashhad, Neishabur, Shiraz, Kerman, Bam (Sharifi et al. 2011) ZCL In 2012, totally ( ZCL & ACL)
More than 98 countries affected by Leishmaniasis ACL In many urban & sub urban areas (8 provinces) Tehran, Mashhad, Neishabur, Shiraz, Kerman, Bam (Sharifi et al. 2011) ZCL In 2012, totally ( ZCL & ACL)
Animal Disease Reporting for the month March 2018
 Animal Disease Reporting for the month March 2018 Sub-Directorate Epidemiology Directorate Animal Health 23 May 2018 Introduction According to the Animal Diseases Act, 1984 (Act 35 of 1984) Controlled
Animal Disease Reporting for the month March 2018 Sub-Directorate Epidemiology Directorate Animal Health 23 May 2018 Introduction According to the Animal Diseases Act, 1984 (Act 35 of 1984) Controlled
Recognizing African swine fever 23. Diagnosis of ASF
 Recognizing African swine fever 23 Diagnosis of ASF When large numbers of pigs of all ages die and the clinical signs and post mortem lesions look like those of ASF, that is the first disease that should
Recognizing African swine fever 23 Diagnosis of ASF When large numbers of pigs of all ages die and the clinical signs and post mortem lesions look like those of ASF, that is the first disease that should
(A) PCR primers (arrows) designed to distinguish wild type (P1+P2), targeted (P1+P2) and excised (P1+P3)14-
 1 Supplemental Figure Legends Figure S1. Mammary tumors of ErbB2 KI mice with 14-3-3σ ablation have elevated ErbB2 transcript levels and cell proliferation (A) PCR primers (arrows) designed to distinguish
1 Supplemental Figure Legends Figure S1. Mammary tumors of ErbB2 KI mice with 14-3-3σ ablation have elevated ErbB2 transcript levels and cell proliferation (A) PCR primers (arrows) designed to distinguish
Animal Disease Reporting for the month June 2018
 Animal Disease Reporting for the month June 2018 Sub-Directorate Epidemiology Directorate Animal Health 17 August 2018 Introduction According to the Animal Diseases Act, 1984 (Act 35 of 1984) Controlled
Animal Disease Reporting for the month June 2018 Sub-Directorate Epidemiology Directorate Animal Health 17 August 2018 Introduction According to the Animal Diseases Act, 1984 (Act 35 of 1984) Controlled
For in vitro Veterinary Diagnostics only. Kylt Rotavirus A. Real-Time RT-PCR Detection.
 For in vitro Veterinary Diagnostics only. Kylt Rotavirus A Real-Time RT-PCR Detection www.kylt.eu DIRECTION FOR USE Kylt Rotavirus A Real-Time RT-PCR Detection A. General Kylt Rotavirus A products are
For in vitro Veterinary Diagnostics only. Kylt Rotavirus A Real-Time RT-PCR Detection www.kylt.eu DIRECTION FOR USE Kylt Rotavirus A Real-Time RT-PCR Detection A. General Kylt Rotavirus A products are
Animal Disease Reporting for the month May 2016
 Animal Disease Reporting for the month May 2016 Sub-Directorate Epidemiology Directorate Animal Health 05 August 2016 Introduction According to the Animal Diseases Act (Act 35 of 1984) Controlled and Notifiable
Animal Disease Reporting for the month May 2016 Sub-Directorate Epidemiology Directorate Animal Health 05 August 2016 Introduction According to the Animal Diseases Act (Act 35 of 1984) Controlled and Notifiable
BRIEF COMMUNICATION ANTIGENIC ANALYSIS OF JAPANESE ENCEPHALITIS VIRUS ISOLATED IN HOKKAIDO WITH MONOCLONAL ANTIBODIES
 Title ANTIGENIC ANALYSIS OF JAPANESE ENCEPHALITIS VIRUS IS MONOCLONAL ANTIBODIES Author(s)OCHIAI, Kenichi; TAKASHIMA, Ikuo; HASHIMOTO, Nobuo CitationJapanese Journal of Veterinary Research, 37(1): 21-2
Title ANTIGENIC ANALYSIS OF JAPANESE ENCEPHALITIS VIRUS IS MONOCLONAL ANTIBODIES Author(s)OCHIAI, Kenichi; TAKASHIMA, Ikuo; HASHIMOTO, Nobuo CitationJapanese Journal of Veterinary Research, 37(1): 21-2
Observations on the use of Anaplasma centrale for immunization of cattle against anaplasmosis in Zimbabwe
 Onderstepoort Journal of Veterinary Research, 65:81-86 (1998) Observations on the use of Anaplasma centrale for immunization of cattle against anaplasmosis in Zimbabwe J.A. TURTON 1 2, T.C. KATSANDE 1,
Onderstepoort Journal of Veterinary Research, 65:81-86 (1998) Observations on the use of Anaplasma centrale for immunization of cattle against anaplasmosis in Zimbabwe J.A. TURTON 1 2, T.C. KATSANDE 1,
The Gribbles Veterinary laboratory received an EDTA blood sample from a cow with a specific request to look for this disease.
 Case Submission (with diagnosis) Submitter Laura Brandt and Harold Tvedten Employer Gribbles Veterinary 35 O Rorke Road PO Box 12049 Penrose, Auckland New Zealand 1642 Phone 64 9 5744709 Fax 64 7 8500770
Case Submission (with diagnosis) Submitter Laura Brandt and Harold Tvedten Employer Gribbles Veterinary 35 O Rorke Road PO Box 12049 Penrose, Auckland New Zealand 1642 Phone 64 9 5744709 Fax 64 7 8500770
Whole Mount Drosophila Embryo In Situ Hybridization with RNA probes 2/5/2001 Leslie Vosshall
 Whole Mount Drosophila Embryo In Situ Hybridization with RNA probes 2/5/2001 Leslie Vosshall DAY ONE All incubations are done at room temperature unless otherwise noted. All solutions and all containers
Whole Mount Drosophila Embryo In Situ Hybridization with RNA probes 2/5/2001 Leslie Vosshall DAY ONE All incubations are done at room temperature unless otherwise noted. All solutions and all containers
Animal Disease Reporting for the month September 2017
 Animal Disease Reporting for the month September 2017 Sub-Directorate Epidemiology Directorate Animal Health 12 December 2017 Introduction According to the Animal Diseases Act, 1984 (Act 35 of 1984) Controlled
Animal Disease Reporting for the month September 2017 Sub-Directorate Epidemiology Directorate Animal Health 12 December 2017 Introduction According to the Animal Diseases Act, 1984 (Act 35 of 1984) Controlled
Vaccination against Lumpy skin disease virus
 1 Vaccination against Lumpy skin disease virus Eeva Tuppurainen, DVM, MSc, PhD, MRCVS Lumpy skin disease scientific expert FAO Regional Office for Europe and Central Asia, Hungary Vaccination of the whole
1 Vaccination against Lumpy skin disease virus Eeva Tuppurainen, DVM, MSc, PhD, MRCVS Lumpy skin disease scientific expert FAO Regional Office for Europe and Central Asia, Hungary Vaccination of the whole
 Malaria parasites Malaria parasites are micro-organisms that belong to the genus Plasmodium. There are more than 100 species of Plasmodium, which can infect many animal species such as reptiles, birds,
Malaria parasites Malaria parasites are micro-organisms that belong to the genus Plasmodium. There are more than 100 species of Plasmodium, which can infect many animal species such as reptiles, birds,
Instructions for Use. APO-AB Annexin V-Biotin Apoptosis Detection Kit 100 tests
 3URGXFW,QIRUPDWLRQ Sigma TACS Annexin V Apoptosis Detection Kits Instructions for Use APO-AB Annexin V-Biotin Apoptosis Detection Kit 100 tests For Research Use Only. Not for use in diagnostic procedures.
3URGXFW,QIRUPDWLRQ Sigma TACS Annexin V Apoptosis Detection Kits Instructions for Use APO-AB Annexin V-Biotin Apoptosis Detection Kit 100 tests For Research Use Only. Not for use in diagnostic procedures.
LEGIONELLA PNEUMOPHILA IFA SLIDE
 1 LEGIONELLA PNEUMOPHILA IFA SLIDE SLEPN: Slides kit for the diagnosis of Legionella pneumophila antibodies in human serum by indirect immunofluorescent assay (IFA). INTRODUCTION: More than 30 species
1 LEGIONELLA PNEUMOPHILA IFA SLIDE SLEPN: Slides kit for the diagnosis of Legionella pneumophila antibodies in human serum by indirect immunofluorescent assay (IFA). INTRODUCTION: More than 30 species
Product Manual. Omni-Array Sense Strand mrna Amplification Kit, 2 ng to 100 ng Version Catalog No.: Reactions
 Genetic Tools and Reagents Universal mrna amplification, sense strand amplification, antisense amplification, cdna synthesis, micro arrays, gene expression, human, mouse, rat, guinea pig, cloning Omni-Array
Genetic Tools and Reagents Universal mrna amplification, sense strand amplification, antisense amplification, cdna synthesis, micro arrays, gene expression, human, mouse, rat, guinea pig, cloning Omni-Array
In vitro cultivation of Plasmodium falciparum
 Chapter 2 In vitro cultivation of Plasmodium falciparum In vitro cultivation of Plasmodium falciparum 2.1 INTRODUCTION Malaria represents the world s greatest public health problem in terms of number of
Chapter 2 In vitro cultivation of Plasmodium falciparum In vitro cultivation of Plasmodium falciparum 2.1 INTRODUCTION Malaria represents the world s greatest public health problem in terms of number of
ASF DIAGNOSIS UPDATE. oie - ASF REFERENCE LABORATORY, MADRID. Prof. José M. Sánchez- Vizcaíno
 ASF DIAGNOSIS UPDATE oie - ASF REFERENCE LABORATORY, MADRID Prof. José M. Sánchez- Vizcaíno jmvizcaino@visavet.ucm.es www.sanidadanimal.info BSL-2 LABORATORIES BSL-3 LABORATORIES BSL-3 BOXES MONITORING
ASF DIAGNOSIS UPDATE oie - ASF REFERENCE LABORATORY, MADRID Prof. José M. Sánchez- Vizcaíno jmvizcaino@visavet.ucm.es www.sanidadanimal.info BSL-2 LABORATORIES BSL-3 LABORATORIES BSL-3 BOXES MONITORING
Q Fever among Dairy Cattle in Chiang Mai Province, Thailand, 2012: A Preliminary Study
 Q Fever among Dairy Cattle in Chiang Mai Province, Thailand, 2012: A Preliminary Study Pranee Rodtian 1, *, Nuamjit M 1, Srijan M 2, Opaschaitat P 3, Ekgatat M 3 1 Fifth Regional Livestock Office, Chiang
Q Fever among Dairy Cattle in Chiang Mai Province, Thailand, 2012: A Preliminary Study Pranee Rodtian 1, *, Nuamjit M 1, Srijan M 2, Opaschaitat P 3, Ekgatat M 3 1 Fifth Regional Livestock Office, Chiang
The diagnostic value of gyrb RFLP PCR. Mycobacteria in patients with clinical. in Mazandaran
 Mazandaran University of Medical Sciences The diagnostic value of gyrb RFLP PCR test t in differentiation between pathogenic Mycobacteria in patients with clinical suspicions spicions of tuberculosis in
Mazandaran University of Medical Sciences The diagnostic value of gyrb RFLP PCR test t in differentiation between pathogenic Mycobacteria in patients with clinical suspicions spicions of tuberculosis in
Blood Smears Only 19 May Sample Preparation and Quality Control
 NEW YORK STATE Parasitology Proficiency Testing Program Blood Smears Only 9 May 205 The purpose of the New York State Proficiency Testing Program in the category of Parasitology - Blood Smears Only is
NEW YORK STATE Parasitology Proficiency Testing Program Blood Smears Only 9 May 205 The purpose of the New York State Proficiency Testing Program in the category of Parasitology - Blood Smears Only is
Study of Virulence and Vector Transmission of Babesia bovis by Use of Cloned Parasite Lines
 INFECTION AND IMMUNITY, JUlY 1990, p. 2171-2176 0019-9567/90/072171-06$02.00/0 Copyright X) 1990, American Society for Microbiology Vol. 58, No. 7 Study of Virulence and Vector Transmission of Babesia
INFECTION AND IMMUNITY, JUlY 1990, p. 2171-2176 0019-9567/90/072171-06$02.00/0 Copyright X) 1990, American Society for Microbiology Vol. 58, No. 7 Study of Virulence and Vector Transmission of Babesia
in the Gastrointestinal and Reproductive Tracts of Quarter Horse Mares
 Influence of Probiotics on Microflora in the Gastrointestinal and Reproductive Tracts of Quarter Horse Mares Katie Barnhart Research Advisors: Dr. Kimberly Cole and Dr. John Mark Reddish Department of
Influence of Probiotics on Microflora in the Gastrointestinal and Reproductive Tracts of Quarter Horse Mares Katie Barnhart Research Advisors: Dr. Kimberly Cole and Dr. John Mark Reddish Department of
West Nile Virus. Family: Flaviviridae
 West Nile Virus 1 Family: Flaviviridae West Nile Virus Genus: Flavivirus Japanese Encephalitis Antigenic Complex Complex Includes: Alfuy, Cacipacore, Japanese encephalitis, koutango, Kunjin, Murray Valley
West Nile Virus 1 Family: Flaviviridae West Nile Virus Genus: Flavivirus Japanese Encephalitis Antigenic Complex Complex Includes: Alfuy, Cacipacore, Japanese encephalitis, koutango, Kunjin, Murray Valley
Instructions for Use. RealStar Influenza S&T RT-PCR Kit /2017 EN
 Instructions for Use RealStar Influenza S&T RT-PCR Kit 3.0 01/2017 EN RealStar Influenza S&T RT-PCR Kit 3.0 For research use only! (RUO) 163003 INS-163000-EN-S02 96 01 2017 altona Diagnostics GmbH Mörkenstr.
Instructions for Use RealStar Influenza S&T RT-PCR Kit 3.0 01/2017 EN RealStar Influenza S&T RT-PCR Kit 3.0 For research use only! (RUO) 163003 INS-163000-EN-S02 96 01 2017 altona Diagnostics GmbH Mörkenstr.
Fluorescence Based Methods for Rapid Diagnosis of Malaria
 International Journal of Current Microbiology and Applied Sciences ISSN: 2319-7706 Volume 4 Number 8 (2015) pp. 832-839 http://www.ijcmas.com Original Research Article Fluorescence Based Methods for Rapid
International Journal of Current Microbiology and Applied Sciences ISSN: 2319-7706 Volume 4 Number 8 (2015) pp. 832-839 http://www.ijcmas.com Original Research Article Fluorescence Based Methods for Rapid
Distribution of bluetongue in the United States of America,
 Vet. Ital., 40 (3), 83-88 Distribution of bluetongue in the United States of America, 1991-2002 E.N. Ostlund (1), K.M. Moser (1), D.J. Johnson (1), J.E. Pearson (2) & B.J. Schmitt (1) (1) Diagnostic Virology
Vet. Ital., 40 (3), 83-88 Distribution of bluetongue in the United States of America, 1991-2002 E.N. Ostlund (1), K.M. Moser (1), D.J. Johnson (1), J.E. Pearson (2) & B.J. Schmitt (1) (1) Diagnostic Virology
Detection of equine infectious anemia nucleic acid in asymptomatic carrier horses by nested PCR
 Asian Biomedicine Vol. 4 No. 6 December 2010; 971-975 Brief communication (Original) Detection of equine infectious anemia nucleic acid in asymptomatic carrier horses by nested PCR Sunutcha Suntrarachun
Asian Biomedicine Vol. 4 No. 6 December 2010; 971-975 Brief communication (Original) Detection of equine infectious anemia nucleic acid in asymptomatic carrier horses by nested PCR Sunutcha Suntrarachun
Identification of vaccine candidate antigens for a Babesia bovis transmission blocking vaccine
 Identification of vaccine candidate antigens for a Babesia bovis transmission blocking vaccine Carlos E. Suarez Animal Disease Research Unit ARS-USDA Department of Veterinary Microbiology and Pathology
Identification of vaccine candidate antigens for a Babesia bovis transmission blocking vaccine Carlos E. Suarez Animal Disease Research Unit ARS-USDA Department of Veterinary Microbiology and Pathology
Epidemiology, diagnosis and control of Toxoplasma gondii in animals and food stuff
 Epidemiology, diagnosis and control of Toxoplasma gondii in animals and food stuff Aize Kijlstra Rome 2009 Toxoplasmosis is a neglected disease entity Disease burden is similar to salmonellosis and campylobacteriosis
Epidemiology, diagnosis and control of Toxoplasma gondii in animals and food stuff Aize Kijlstra Rome 2009 Toxoplasmosis is a neglected disease entity Disease burden is similar to salmonellosis and campylobacteriosis
EPIDEMIOLOGY REPORT. Buffer Zones. Disease reporting Outbreaks vs. Cases
 EPIDEMIOLOGY REPORT Volume 1, Issue November 009 WESTERN CAPE DEPARTMENT OF AGRICULTURE VETERINARY SERVICES Buffer Zones Buffer zones are calculated around any disease outbreak which, according to legislation,
EPIDEMIOLOGY REPORT Volume 1, Issue November 009 WESTERN CAPE DEPARTMENT OF AGRICULTURE VETERINARY SERVICES Buffer Zones Buffer zones are calculated around any disease outbreak which, according to legislation,
Serological response to early vaccination against. Babesia bovis and Babesia bigemina in dairy. calves
 Serological response to early vaccination against Babesia bovis and Babesia bigemina in dairy calves by Anthony J. Davis Supervisor: Prof B L Penzhorn Co-supervisor: Prof A A Latif Co-supervisor: Dr J
Serological response to early vaccination against Babesia bovis and Babesia bigemina in dairy calves by Anthony J. Davis Supervisor: Prof B L Penzhorn Co-supervisor: Prof A A Latif Co-supervisor: Dr J
certificate, the horses will be returned or destroyed by AQSIQ. Article 2 Article 3 Article 4 Article 5
 PROTOCOL BETWEEN THE FEDERAL AGENCY FOR THE SAFETY OF FOOD CHAIN OF THE KINGDOM OF BELGIUM AND THE GENERAL ADMINISTRATION OF QUALITY SUPERVISION, INSPECTION AND QUARANTINE OF THE PEOPLE S REPUBLIC OF CHINA
PROTOCOL BETWEEN THE FEDERAL AGENCY FOR THE SAFETY OF FOOD CHAIN OF THE KINGDOM OF BELGIUM AND THE GENERAL ADMINISTRATION OF QUALITY SUPERVISION, INSPECTION AND QUARANTINE OF THE PEOPLE S REPUBLIC OF CHINA
Outbreak Investigation Guidance for Vectorborne Diseases
 COMMUNICABLE DISEASE OUTBREAK MANUAL New Jersey s Public Health Response APPENDIX T3: EXTENDED GUIDANCE Outbreak Investigation Guidance for Vectorborne Diseases As per N.J.A.C. 8:57, viruses that are transmitted
COMMUNICABLE DISEASE OUTBREAK MANUAL New Jersey s Public Health Response APPENDIX T3: EXTENDED GUIDANCE Outbreak Investigation Guidance for Vectorborne Diseases As per N.J.A.C. 8:57, viruses that are transmitted
Pinpoint Slide RNA Isolation System II Catalog No. R1007
 INSTRUCTION MANUAL Pinpoint Slide RNA Isolation System II Catalog No. R1007 Highlights Allows for the isolation of total RNA from paraffin-embedded tissue sections on glass slides Simple procedure combines
INSTRUCTION MANUAL Pinpoint Slide RNA Isolation System II Catalog No. R1007 Highlights Allows for the isolation of total RNA from paraffin-embedded tissue sections on glass slides Simple procedure combines
Norgen s HIV proviral DNA PCR Kit was developed and validated to be used with the following PCR instruments: Qiagen Rotor-Gene Q BioRad icycler
 3430 Schmon Parkway Thorold, ON, Canada L2V 4Y6 Phone: (905) 227-8848 Fax: (905) 227-1061 Email: techsupport@norgenbiotek.com HIV Proviral DNA PCR Kit Product # 33840 Product Insert Background Information
3430 Schmon Parkway Thorold, ON, Canada L2V 4Y6 Phone: (905) 227-8848 Fax: (905) 227-1061 Email: techsupport@norgenbiotek.com HIV Proviral DNA PCR Kit Product # 33840 Product Insert Background Information
Situation Report. 1. Highlights. 2. Background
 Situation Report Outbreak name Listeriosis Country affected South Africa Date & Time of report 26 July 2018 Investigation start date August 2017 Prepared by National Listeria Incident Management Team A
Situation Report Outbreak name Listeriosis Country affected South Africa Date & Time of report 26 July 2018 Investigation start date August 2017 Prepared by National Listeria Incident Management Team A
Cattle vaccination studies using novel anti-cattle tick antigens developed during Beef CRC research
 final report Project code: Prepared by: B.AHE.0212 Alicia Tabor, Manuel Rodriguez Valle, Michael McGowan (UQ), David Mayer, Elizabeth Fowler, Catherin Minchin and Bing Zhang (QDAF) The University of Queensland;
final report Project code: Prepared by: B.AHE.0212 Alicia Tabor, Manuel Rodriguez Valle, Michael McGowan (UQ), David Mayer, Elizabeth Fowler, Catherin Minchin and Bing Zhang (QDAF) The University of Queensland;
Supplementary Information
 Supplementary Information Supplementary Figure 1. CD4 + T cell activation and lack of apoptosis after crosslinking with anti-cd3 + anti-cd28 + anti-cd160. (a) Flow cytometry of anti-cd160 (5D.10A11) binding
Supplementary Information Supplementary Figure 1. CD4 + T cell activation and lack of apoptosis after crosslinking with anti-cd3 + anti-cd28 + anti-cd160. (a) Flow cytometry of anti-cd160 (5D.10A11) binding
Absence of Kaposi s Sarcoma-Associated Herpesvirus in Patients with Pulmonary
 Online Data Supplement Absence of Kaposi s Sarcoma-Associated Herpesvirus in Patients with Pulmonary Arterial Hypertension Cornelia Henke-Gendo, Michael Mengel, Marius M. Hoeper, Khaled Alkharsah, Thomas
Online Data Supplement Absence of Kaposi s Sarcoma-Associated Herpesvirus in Patients with Pulmonary Arterial Hypertension Cornelia Henke-Gendo, Michael Mengel, Marius M. Hoeper, Khaled Alkharsah, Thomas
Ring Forms in Red Blood Cells (RBCs) Babesia? from Danish Chronically Ill Patients, All Clinically Suspect of Having Persistent Active Borreliosis!
 Ring Forms in Red Blood Cells (RBCs) Babesia? from Danish Chronically Ill Patients, All Clinically Suspect of Having Persistent Active Borreliosis! Marie Kroun, MD Denmark Presentation in York, UK June
Ring Forms in Red Blood Cells (RBCs) Babesia? from Danish Chronically Ill Patients, All Clinically Suspect of Having Persistent Active Borreliosis! Marie Kroun, MD Denmark Presentation in York, UK June
Odyssean malaria outbreak at a bush lodge in Madikwe Game Reserve, North West Province, October-November 2015
 Odyssean malaria outbreak at a bush lodge in Madikwe Game Reserve, North West Province, October-November 2015 Genevie Ntshoe, Andrew Tlagadi, Thejane Motladiile, Kerrigan McCarthy, Oleteng Mokate, John
Odyssean malaria outbreak at a bush lodge in Madikwe Game Reserve, North West Province, October-November 2015 Genevie Ntshoe, Andrew Tlagadi, Thejane Motladiile, Kerrigan McCarthy, Oleteng Mokate, John
Frequency of seropositive equines for Theileria equi in the Southern Rio Grande do Sul State, Brazil
 Parasitol Latinoam 63: 46-50, 2008 FLAP ARTÍCULO ORIGINAL Frequency of seropositive equines for Theileria equi in the Southern Rio Grande do Sul State, Brazil LEANDRO QUINTANA NIZOLI*, MARCELO MENDES GÖTZE*,
Parasitol Latinoam 63: 46-50, 2008 FLAP ARTÍCULO ORIGINAL Frequency of seropositive equines for Theileria equi in the Southern Rio Grande do Sul State, Brazil LEANDRO QUINTANA NIZOLI*, MARCELO MENDES GÖTZE*,
Instructions for Use. RealStar Influenza Screen & Type RT-PCR Kit /2017 EN
 Instructions for Use RealStar Influenza Screen & Type RT-PCR Kit 4.0 05/2017 EN RealStar Influenza Screen & Type RT-PCR Kit 4.0 For research use only! (RUO) 164003 INS-164000-EN-S01 96 05 2017 altona
Instructions for Use RealStar Influenza Screen & Type RT-PCR Kit 4.0 05/2017 EN RealStar Influenza Screen & Type RT-PCR Kit 4.0 For research use only! (RUO) 164003 INS-164000-EN-S01 96 05 2017 altona
Elements of the FMD control problem in Southern Africa: 2
 Elements of the FMD control problem in Southern Africa: 2 Gavin Thomson Commodity-based trade of beef and enhanced market access: The vital role of the Department of Veterinary Services Gaborone; 6-7 January
Elements of the FMD control problem in Southern Africa: 2 Gavin Thomson Commodity-based trade of beef and enhanced market access: The vital role of the Department of Veterinary Services Gaborone; 6-7 January
P. Dandapat. Eastern Regional Station ICAR-Indian Veterinary Research Institute 37 Belgachia Road, Kolkata
 Evaluation of the Sensitivity and Specificity of Gamma Interferon Assay, Single Intradermal Tuberculin and Comparative Intradermal Tuberculin Tests in Naturally Infected Cattle Herds P. Dandapat Eastern
Evaluation of the Sensitivity and Specificity of Gamma Interferon Assay, Single Intradermal Tuberculin and Comparative Intradermal Tuberculin Tests in Naturally Infected Cattle Herds P. Dandapat Eastern
Poultry Disease Manual Characteristics And
 Poultry Disease Manual Characteristics And Control Of Infections Written by: Dr. Jacquie Jacob, University of Kentucky Pullorum disease, also called Infection by Salmonella pullorum has also been reported
Poultry Disease Manual Characteristics And Control Of Infections Written by: Dr. Jacquie Jacob, University of Kentucky Pullorum disease, also called Infection by Salmonella pullorum has also been reported
RIFT VALLEY FEVER AN EVALUATION OF THE OUTBREAKS IN SOUTH AFRICA
 RIFT VALLEY FEVER AN EVALUATION OF THE OUTBREAKS IN SOUTH AFRICA RIFT VALLEY FEVER (RVF) IN SOUTH AFRICA: Introduction Mosquito-borne viral disease which affects livestock, some game and Humans The disease
RIFT VALLEY FEVER AN EVALUATION OF THE OUTBREAKS IN SOUTH AFRICA RIFT VALLEY FEVER (RVF) IN SOUTH AFRICA: Introduction Mosquito-borne viral disease which affects livestock, some game and Humans The disease
PRODUCT: RNAzol BD for Blood May 2014 Catalog No: RB 192 Storage: Store at room temperature
 PRODUCT: RNAzol BD for Blood May 2014 Catalog No: RB 192 Storage: Store at room temperature PRODUCT DESCRIPTION. RNAzol BD is a reagent for isolation of total RNA from whole blood, plasma or serum of human
PRODUCT: RNAzol BD for Blood May 2014 Catalog No: RB 192 Storage: Store at room temperature PRODUCT DESCRIPTION. RNAzol BD is a reagent for isolation of total RNA from whole blood, plasma or serum of human
ANTIGEN SANDWICH ELISA PREDICTS RT-PCR DETECTION OF DENGUE VIRUS GENOME IN INFECTED CULTURE FLUIDS OF AEDES ALBOPICTUS C6/36 CELLS
 ANTIGEN SANDWICH ELISA PREDICTS RT-PCR DETECTION OF DENGUE VIRUS GENOME IN INFECTED CULTURE FLUIDS OF AEDES ALBOPICTUS C6/36 CELLS Corazon C Buerano 1,2, Filipinas F Natividad 2, Rodolfo C Contreras 3,
ANTIGEN SANDWICH ELISA PREDICTS RT-PCR DETECTION OF DENGUE VIRUS GENOME IN INFECTED CULTURE FLUIDS OF AEDES ALBOPICTUS C6/36 CELLS Corazon C Buerano 1,2, Filipinas F Natividad 2, Rodolfo C Contreras 3,
Midi Plant Genomic DNA Purification Kit
 Midi Plant Genomic DNA Purification Kit Cat #:DP022MD/ DP022MD-50 Size:10/50 reactions Store at RT For research use only 1 Description: The Midi Plant Genomic DNA Purification Kit provides a rapid, simple
Midi Plant Genomic DNA Purification Kit Cat #:DP022MD/ DP022MD-50 Size:10/50 reactions Store at RT For research use only 1 Description: The Midi Plant Genomic DNA Purification Kit provides a rapid, simple
PUBLIC HEALTH SIGNIFICANCE SEASONAL INFLUENZA AVIAN INFLUENZA SWINE INFLUENZA
 INFLUENZA DEFINITION Influenza is an acute highly infectious viral disease characterized by fever, general and respiratory tract catarrhal manifestations. Influenza has 3 Types Seasonal Influenza Avian
INFLUENZA DEFINITION Influenza is an acute highly infectious viral disease characterized by fever, general and respiratory tract catarrhal manifestations. Influenza has 3 Types Seasonal Influenza Avian
CHAPTER 7: REAGENTS AND SOLUTIONS
 7.1. ANALYSIS OF MODULATION OF SOD ENZYME Acetic acid (cat. no. 11007, Glaxo Qualigen, India): Bovine Serum Albumin stock solution (BSA, 1mg/ml): 1 mg of standard bovine serum albumin (cat. no. A2153,
7.1. ANALYSIS OF MODULATION OF SOD ENZYME Acetic acid (cat. no. 11007, Glaxo Qualigen, India): Bovine Serum Albumin stock solution (BSA, 1mg/ml): 1 mg of standard bovine serum albumin (cat. no. A2153,
Annexure III SOLUTIONS AND REAGENTS
 Annexure III SOLUTIONS AND REAGENTS A. STOCK SOLUTIONS FOR DNA ISOLATION 0.5M Ethylene-diamine tetra acetic acid (EDTA) (ph=8.0) 1M Tris-Cl (ph=8.0) 5M NaCl solution Red cell lysis buffer (10X) White cell
Annexure III SOLUTIONS AND REAGENTS A. STOCK SOLUTIONS FOR DNA ISOLATION 0.5M Ethylene-diamine tetra acetic acid (EDTA) (ph=8.0) 1M Tris-Cl (ph=8.0) 5M NaCl solution Red cell lysis buffer (10X) White cell
IMPORTANT INFORMATION REGARDING VACCINATION AGAINST AFRICAN HORSE SICKNESS (AHS) IN SOUTH AFRICA
 IMPORTANT INFORMATION REGARDING VACCINATION AGAINST AFRICAN HORSE SICKNESS (AHS) IN SOUTH AFRICA AHS is a controlled animal disease in terms of the Animal Diseases Act 1984 (Act no 35 of 84) and certain
IMPORTANT INFORMATION REGARDING VACCINATION AGAINST AFRICAN HORSE SICKNESS (AHS) IN SOUTH AFRICA AHS is a controlled animal disease in terms of the Animal Diseases Act 1984 (Act no 35 of 84) and certain
Meeting of the Ministers o Agriculture and Food From South Eastern Europe in relation to Bluetongue disease
 Meeting of the Ministers o Agriculture and Food From South Eastern Europe in relation to Bluetongue disease Epidemiological situation in Republic of Kosovo Dr. Valdet Gjinovci Chief Executive Officer Food
Meeting of the Ministers o Agriculture and Food From South Eastern Europe in relation to Bluetongue disease Epidemiological situation in Republic of Kosovo Dr. Valdet Gjinovci Chief Executive Officer Food
THE ROLE OF LIVESTOCK IN THE EPIDEMIOLOGY OF SLEEPING SICKNESS IN TANZANIA
 THE ROLE OF LIVESTOCK IN THE EPIDEMIOLOGY OF SLEEPING SICKNESS IN TANZANIA Imna Malele, Hamisi Nyingilili, Eugen Lyaruu, Winston Kitwika Tsetse & Trypanosomiasis Research Institute Majani Mapana, Off Korogwe
THE ROLE OF LIVESTOCK IN THE EPIDEMIOLOGY OF SLEEPING SICKNESS IN TANZANIA Imna Malele, Hamisi Nyingilili, Eugen Lyaruu, Winston Kitwika Tsetse & Trypanosomiasis Research Institute Majani Mapana, Off Korogwe
Pakistan Journal of Life and Social Sciences
 Pak. j. life soc. sci. (2007), 5(1-2): 1-5 Pakistan Journal of Life and Social Sciences Comparison of Conventional Bacterial isolation, Rapid Slide Agglutination and Polymerase Chain Reaction for Detection
Pak. j. life soc. sci. (2007), 5(1-2): 1-5 Pakistan Journal of Life and Social Sciences Comparison of Conventional Bacterial isolation, Rapid Slide Agglutination and Polymerase Chain Reaction for Detection
Confirmation of Trypanosome Parasitaemia in Previously Serologically Positive Individuals in the Abraka Area of Delta State, Nigeria
 JMBR: A Peer-review Journal of Biomedical Sciences December 2006 Vol. 5 No.2 pp-28-32 Confirmation of Trypanosome Parasitaemia in Previously Serologically Positive Individuals in the Abraka Area of Delta
JMBR: A Peer-review Journal of Biomedical Sciences December 2006 Vol. 5 No.2 pp-28-32 Confirmation of Trypanosome Parasitaemia in Previously Serologically Positive Individuals in the Abraka Area of Delta
Norgen s HIV Proviral DNA PCR Kit was developed and validated to be used with the following PCR instruments: Qiagen Rotor-Gene Q BioRad T1000 Cycler
 3430 Schmon Parkway Thorold, ON, Canada L2V 4Y6 Phone: 866-667-4362 (905) 227-8848 Fax: (905) 227-1061 Email: techsupport@norgenbiotek.com HIV Proviral DNA PCR Kit Product# 33840 Product Insert Intended
3430 Schmon Parkway Thorold, ON, Canada L2V 4Y6 Phone: 866-667-4362 (905) 227-8848 Fax: (905) 227-1061 Email: techsupport@norgenbiotek.com HIV Proviral DNA PCR Kit Product# 33840 Product Insert Intended
Study of the One-Step Growth Curve of Equine Infectious Anemia Virus by Immunofluorescence
 INFECTION AND IMMUNITY, June 1972, p. 89-895 Copyright 1972 American Society for Microbiology Vol. 5, No. 6 Printed in U.S.A Study of the One-Step Growth Curve of Equine Infectious Anemia Virus by Immunofluorescence
INFECTION AND IMMUNITY, June 1972, p. 89-895 Copyright 1972 American Society for Microbiology Vol. 5, No. 6 Printed in U.S.A Study of the One-Step Growth Curve of Equine Infectious Anemia Virus by Immunofluorescence
J07 Titer dynamics, complement fixation test and neutralization tests
 avllm0421c (spring 2017) J07 Titer dynamics, complement fixation test and neutralization tests Outline titer, antibody titer dynamics complement, complement fixation reaction neutralization tests 2/35
avllm0421c (spring 2017) J07 Titer dynamics, complement fixation test and neutralization tests Outline titer, antibody titer dynamics complement, complement fixation reaction neutralization tests 2/35
Blood Smears Only 5 February Sample Preparation and Quality Control 13B A
 NEW YORK STATE Parasitology Proficiency Testing Program Blood Smears Only 5 February 2013 The purpose of the New York State Proficiency Testing Program in the category of Parasitology Blood Smears Only
NEW YORK STATE Parasitology Proficiency Testing Program Blood Smears Only 5 February 2013 The purpose of the New York State Proficiency Testing Program in the category of Parasitology Blood Smears Only
IMMUNOGENICITY OF FORMALDYDE INACTIVATED NEWCASTLE DISEASE VIRUS FIELD ISOLATE IN MATERNAL ANTIBODY FREE CHICKENS
 IMMUNOGENICITY OF FORMALDYDE INACTIVATED NEWCASTLE DISEASE VIRUS FIELD ISOLATE IN MATERNAL ANTIBODY FREE CHICKENS Anak Agung Ayu Mirah Adi 1 *, IGusti Agung Arta Putra 2, Nyoman Mantik Astawa 3, I Made
IMMUNOGENICITY OF FORMALDYDE INACTIVATED NEWCASTLE DISEASE VIRUS FIELD ISOLATE IN MATERNAL ANTIBODY FREE CHICKENS Anak Agung Ayu Mirah Adi 1 *, IGusti Agung Arta Putra 2, Nyoman Mantik Astawa 3, I Made
