Molecular epidemiology and the evolution of human coxsackievirus A6
|
|
|
- Wilfrid Bailey
- 6 years ago
- Views:
Transcription
1 Journal of General Virology (2016), 97, DOI /jgv Short Communication Correspondence Yong Poovorawan Molecular epidemiology and the evolution of human coxsackievirus A6 Jiratchaya Puenpa, 1 Sompong Vongpunsawad, 1 Riikka Österback, 2,3 Matti Waris, 2,3 Eva Eriksson, 4,5 Jan Albert, 4,5 Sofie Midgley, 6 Thea K. Fischer, 6 Anna M. Eis-Hübinger, 7 María Cabrerizo, 8 Eleanor Gaunt, 9 Peter Simmonds 9,10 and Yong Poovorawan 1 1 Center of Excellence in Clinical Virology, Faculty of Medicine, Chulalongkorn University, Bangkok, Thailand 2 Department of Virology, University of Turku, Turku, Finland 3 Department of Clinical Virology, Turku University Hospital, Turku, Finland 4 Department of Microbiology, Tumor and Cell Biology, Karolinska Institute, Stockholm, Sweden 5 Department of Clinical Microbiology, Karolinska University Hospital, Stockholm, Sweden 6 Department of Microbiological Diagnostics & Virology, Artillerivej 5, DK-2300 Copenhagen, Denmark 7 Institute for Virology, University of Bonn Medical Center, Sigmund-Freud-Str. 25, D Bonn, Germany 8 Instituto de Salud Carlos III, Majadahonda, Madrid, Spain 9 Infection and Immunity Division, Roslin Institute, University of Edinburgh, Easter Bush, Edinburgh EH25 9RG, UK 10 Nuffield Department of Medicine, University of Oxford, Oxford OX1 3SY, UK Received 24 May 2016 Accepted 28 September 2016 Coxsackievirus A6 (CV-A6) is a major aetiologic agent for hand, foot and mouth disease (HFMD) in recent years. HFMD outbreaks associated with CV-A6 resulted from the evolutionary dynamics of CV-A6 and the appearance of novel recombinant forms (RFs). To examine this, 151 variants collected in 2013 and 2014 from Germany, Spain, Sweden, Denmark and Thailand were genotyped for the VP1 capsid and 3Dpol genes. Analysis of the VP1 gene showed an increasing correspondence between CV-A6 genome recombination and sequence divergence (estimated substitution rate of substitutions site 1 year 1 and RF half-life of 3.1 years). Bayesian phylogenetic analysis showed that recent recombination groups (RF-E, -F, -H, -J and -K) shared a common ancestor (RF-A). Thirty-nine full-length genomes of different RFs revealed recombination breakpoints between the 2A 2C and the 5 UTRs. The emergence of new CV-A6 recombination groups has become widespread in Europe and Asia within the last 8 years. Human enteroviruses are genetically diverse RNA viruses within the genus Enterovirus genus in the family Picornaviridae and are responsible for a wide spectrum of clinical manifestations (Knowles et al., 2012). Human enteroviruses are The GenBank/EMBL/DDBJ accession numbers for the new nucleotide sequences of the coxsackievirus A6 VP1, 3Dpol genes and complete genome investigated in this study are KX KX Three supplementary tables and four supplementary figures are available with the online Supplementary Material. divided into four species (A D). Species A, of which coxsackievirus A6 (CV-A6) is a member, currently comprises 20 types (Pons-Salort et al., 2015). CV-A6 possesses a positive-stranded RNA genome of approximately 7400 nucleotides encapsidated by a highly structured icosahedral capsid. The viral genome is translated into a large polyprotein that is subsequently cleaved into structural (VP1 VP4) and non-structural (NS) (2A 2C and 3A 3D) proteins (Whitton et al., 2005). The degree of similarity of nucleotides and amino acid sequences of the VP1 region provides the primary tool for the identification and assignment of new types ã 2016 The Authors Printed in Great Britain 3225
2 J. Puenpa and others within a species, in which novel variants showing less than 75 % nucleotide sequence identity are classified as new types (Oberste et al., 1999). CV-A6 infections are typically mild and asymptomatic, whereas enterovirus A71 and coxsackievirus A6 are most often implicated in causing hand, foot and mouth disease (HFMD), a disease characterized by vesicular exanthema on the hands, feet, and oral mucosa (Puenpa et al., 2011; Schuffenecker et al., 2011; Wu et al., 2010). In 2014, atypical HFMD was linked to CV-A6 infection in children with erythematous papular rash resembling eczema herpeticum (Sinclair et al., 2014) as a result of newly emerging variants of CV-A6. Such novel recombinant forms (RFs) of the virus have been assigned into RF-A H based on the 3D polymerase (3Dpol) phylogeny (Gaunt et al., 2015; McWilliam Leitch et al., 2012). It was subsequently determined that RF-H, which possessed phylogenetically distinct 3Dpol region sequences likely acquired from other human enterovirus species A serotypes through recombination, was largely responsible for the clinically unusual HFMD in Edinburgh, UK (Gaunt et al., 2015). Here, we further defined how recently emerged RFs including RF-H and its predecessor RF-A are circulating more widely and contributing to the increased incidence of HFMD elsewhere around the world. We initially examined the VP1 sequence divergence and 3Dpol sequence grouping by analysing 151 CV-A6 strains from Denmark (n=22), Germany (n=4), Spain (n=14), Sweden (n=6) and Thailand (n=105) collected in 2013 and 2014 (Table S1, available in the online Supplementary Material). Nested reverse transcription PCR (RT-PCR) was performed using newly designed primers to amplify the VP1 and 3Dpol genes (Table S2), followed by sequencing. The reverse transcription and first-round PCR utilized the Superscript III One-Step RT-PCR system with Platinum Taq High Fidelity (Invitrogen), while the second-round PCR utilized GoTaq DNA Polymerase (Promega). Sequences were analysed using the SSE 1.2 sequence editor package ( org) (Simmonds, 2012) and phylogenetic tree reconstruction was performed with the MEGA program (v6) using the best-fit models and the maximum-likelihood method (Tamura et al., 2013). All newly generated sequences were deposited in the GenBank database under the accession numbers KX KX The VP1 sequences from the CV-A6 identified in Denmark and Spain clustered within lineage I, the very same group of CV-A6 responsible for eczema herpeticum in Edinburgh in 2014 (Fig. S1). They were distinct from CV-A6 from Taiwan (lineage II), Thailand (lineage III), China (lineage IV) and Finland (lineage V). Examination of the sequences from the 3Dpol region enabled the designation of bootstrap-supported clades comprising groups A, B, C, D, E, F, G, H, I, J and K (Fig. 1, Table S3) in agreement with previous analysis (McWilliam Leitch et al., 2009, 2010, 2012). The majority of the CV-A6 strains clustered within two of the previously assigned recombinant forms RF-A (105/151) and RF-F (37/ 151). Meanwhile, four variants from Denmark grouped with RF-H, as did two variants from Spain. The additional three variants from Denmark and Spain comprised RF-G. Strong evidence of recombination was demonstrated by the observed differences in the phylogenetic trees based on the 3Dpol region, for which RF groups were assigned, and VP1 region for several strains. For example, the TW/00141/E/ 2007 strain was assigned to RF-E by the 3Dpol phylogeny, but grouped with the two RF-B strains (FI/TS3FinTu81042/ B/2008 and JP/Kyoto1/B/1999) in the VP1 tree. The 3Dpolassigned RF-J strain CN/P143/J/2013 grouped with some of the RF-A strains. Finally, the CN/CC13/K/2013 is an RF-K strain, but instead appeared next to an RF-D strain CN/ HN421/D/2011 in the VP1 phylogeny. To enable examination of the sequence relationships in other parts of the genome, we obtained 39 nearly complete genome sequences of CV-A6 variants (Germany=3, Spain=3, Denmark=8 and Thailand=25) representing RF-A (n=21), RF-F (n=10), RF-G (n=2) and RF-H (n=6). Comparison of the phylogenetic trees of the VP1, 5 UTR and VP4/2 regions yielded broadly similar groupings of CV-A6 variants (Figs 1 and S2). However, several instances of discordance in the trees provided additional evidence for potential CV-A6 recombination. For example, RF-E variants grouped with RF-A and -H in the VP1, but not in the 5 UTR and VP4/2 regions. RF-A strains were interspersed in the 5 UTR and VP4/2 trees with RF-J, which contrasts with their consistent grouping in the VP1 region. All of RF- K was monophyletic in the VP1, VP4/2 and the 5 UTR regions except one which grouped with RF-D. Therefore, the patterns of phylogenetic discordance were consistent with recombination in CV-A6 and in agreement with previous findings for other enteroviruses (Cabrerizo et al., 2014; Calvert et al., 2010; McIntyre et al., 2010; McWilliam Leitch et al., 2009, 2010, 2012). We estimated the rates of evolution and molecular clock phylogeny (Drummond & Rambaut, 2007) from VP1 gene sequences using the Bayesian Markov Chain Monte Carlo (MCMC) method implemented in BEAST (v1.8.0) (Drummond et al., 2012). Two independent runs involved constant and exponential growth as priors with a chain length of 100 million and a relaxed log-normal molecular clock model were analysed using the SRD06 model of substitution. All other parameters were optimized during the burn-in period. Convergence of the chains and effective sample sizes of the estimates were checked using Tracer ( Tracer). Since sequence divergence in VP1 provides a proxy measure for the time of divergence of CV-A6 variants from which estimates of the life spans of individual RFs can be derived as described previously for echovirus type 30 isolates (McWilliam Leitch et al., 2009), we evaluated the VP1 evolutionary divergence and the proportion of recombinant comparisons for variants with different 3Dpol groups. Pairwise comparison among CV-A6 variants showed precise correlation between VP1 sequence distances and 3226 Journal of General Virology 97
3 Evolution of human coxsackievirus A6 (a) (b) VP1 3Dpol Recombination group A J C B D E K H G F Fig. 1. Phylogenetic analysis of CV-A6 variants in different genome regions. Maximum-likelihood trees of (a) VP1 (positions 2485 and 3816 numbered based on the Gdula prototype strain, GenBank accession number AY421764), (b) 3Dpol (positions 5061 and 6364 numbered based on the Gdula prototype strain, GenBank accession number AY421764) and previously determined reference groups (RF-B E). The optimal substitution models were Kimura two-parameter K2P with invariant sites (I) for VP1 and Kimura two-parameter with I and gamma distribution (I ) for 3Dpol. Each sequence is identified by the country of origin, sample code, 3Dpol clade assignment and year of collection. Dot colours indicate the recombination group assignments based on 3Dpol phylogeny. Bars denote the evolutionary distance according to the number of nucleotide substitutions per site. Bootstrap consensus was inferred from 1000 replicates
4 J. Puenpa and others Table 1. Rates of sequence change and tmrcas by MCMC analysis RF n* Divergence d N /d S MCMC (BEAST) RF group and geographic set Nucleotide Amino acid Substitution rate (10 3 ) tmrca VP1 3Dpol VP1 3Dpol VP1 3Dpol VP1 3Dpol VP1 3Dpol Whole data set All All ND ND ND 8.1 ( ) ND 68.2 ( ) ND Individual RF group All RF-A ( ) 11.0 ( ) 15.9 ( ) 12.9 ( ) All RF-F ( ) 6.5 ( ) 17.1 ( ) 3.5 ( ) ND, Not determined. *Number of sequences in each set analysed. Mean pairwise P distances. Mean value with the HPD interval in parentheses. Times to the most recent common ancestor. assignment to different RF group by the 3Dpol region (Fig. S3). Furthermore, an estimate of the approximate RF halflives of CV-A6 lineages was calculated by combining the mean sequence divergence in VP1 at the 50 % recombination frequency threshold (estimated at 0.05) with the substitution rate in VP1 ( substitutions site 1 year 1 ) (Table 1). This corresponds to a period of 6.17 years (0.05/ ) of divergent evolution, or approximately 3.1 years from a common ancestor. This RF half-life of CV-A6 was very similar to that estimated previously for echovirus type 30, although higher than E6 and E9 but lower than EV71 and E11 (Cabrerizo et al., 2014; McWilliam Leitch et al., 2009, 2010, 2012). To determine when CV-A6 recombination groups first appeared, we reconstructed the temporal phylogeny from VP1 sequences. Since variability of the VP1 sequence was restricted primarily to synonymous sites, most sequence changes likely occurred through neutral drift. Using molecular clock analysis, we estimated the nucleotide substitution rate and times to the most recent common ancestor (tmrca) of different regions and the assignment of 3Dpol clades of the greatest RFs (RF-A and -F). The substitution rate of the whole data set of all VP1 sequences was estimated to be substitutions site 1 year 1 [highprobability distribution (HPD) range, ]. The MRCA of all CV-A6 clusters likely first appeared in 1947 (HPD, ), while that of RF-A probably emerged in 1999 (HPD, ). The diversity was low for RF-F variants as demonstrated by their tight clustering at the top of the tree (Fig. 1, Table 1), whereas RF-A variants dispersed into other branches and therefore implied that they possessed higher substitution rate. The number of RF-F variants was only one-third that of RF-A and therefore might have contributed to the bias due to sampling size. To determine more precisely the timescale of recombination events underlying the appearance of each RF, datasets of VP1 gene sequences were further analysed using the Bayesian MCMC method to generate time-correlated phylogeny (Fig. 2). While earlier recombination events could not be reconstructed in any detail due to inadequate sampling of CV-A6 before 2008, variants collected after this date were monophyletic and fell into three further lineages with estimated dates of splitting between 2004 and The oldest lineage was composed purely of RF-G samples, which first appeared in 2011, and thereafter underwent the recombination event that exchanged the 3Dpol region sequences between 2004 and The other two lineages contained samples belonging to RF-A. One was composed solely of RF-A and persisted for at least 11 years ( ). The other lineage contained RF-A sequences and samples isolated subsequently, which belonged to other RF groups (E, F, H, J and K). In lineage 1, the oldest variants were those originally described in Asia (Thailand and Japan) before 2012 and the lineage variants then spread into Europe and Asia between 2013 and Within this lineage, CV-A6 variants belonged to RF-F with the 3228 Journal of General Virology 97
5 Evolution of human coxsackievirus A6 RF-A RF-F RF-B RF-G RF-C RF-H RF-D RF-J RF-E RF-K 2009 ( ) Undetermined ( ) ( ) 2001 ( ) 1994 ( ) Fig. 2. Temporal phylogeny of VP1 sequences of CV-A6 variants in this study and published sequences. Branch colours denote recombination groups in each clade. Two RF-A lineages (1 and 2) are noted
6 J. Puenpa and others recombination event and dated between 2009 and The most recent group, RF-H, which appeared in 2013, probably recombined between 2011 and The RFs (F, G, H and K) were monophyletic and likely originated from single recombination events, unlike RF-E and -J which were detected in more than one VP1 lineage. With the exception of RF-F, which was isolated from both Europe and Asia, other RF groups were detected only from Europe or Asia: G and H (Europe), E (Taiwan) and J and K (China). Variants within lineage 2 (RF-A) were those detected in the first HFMD outbreak in Finland in 2008 along with variants detected subsequently in Europe and Asia over the following 1 5 years. Having identified the likely time course and direction of the recombination events, divergence scan analyses were performed between the ancestral RF-A sequences with complete genome sequences generated in the current study (RF-F H) and the RFs from previous study (RF-J and -K) to identify recombination breakpoints (Fig. S4). The sharp increase in sequence divergence at various points in the P2 region provided evidence for the occurrence of separate, individual recombination events for each RF. The first breakpoint was found at the 2A protein-encoding region around nucleotide position 3500 (RF-G). The next breakpoints were located in 2B and at the border between 2B and 2C regions (RF-F, -J and -H). The last breakpoint of RF-K can be recognized at nucleotide position 5000 and included the 3 part of the 2C region. This study reports a detailed, multi-centre investigation of the emergence of an enterovirus serotype associated with epidemics of HFMD. It catalogues the complexity of the evolutionary processes associated with its geographical expansion and the occurrence of a number of recombination events each involving replacement of close to complete NS gene blocks at varying times since the founder recombinant form RF-A was first described in 2008 (Osterback et al., 2009). NS region sequences of most RFs have not been described in association with other species A serotypes including any of the RFs described for enterovirus A71 (McWilliam Leitch et al., 2012). However, there was some evidence for a limited degree of re-circulation within the recombination pool of NS region sequences; RF-E and -K appeared at two different positions in the VP1 phylogenetic tree. Furthermore, a limited degree of sharing of NS region sequences between different species B serotypes has been documented (Bailly et al., 2009). Phylogenetic reconstruction was used to analyse trait evolution such as temporal and geographical correlates of individual recombination events, which was previously incomplete. The most frequently detected recombinant form, RF-A, showed decades-long circulation and was the ancestor of five separate recombination groups (RF-E, -F, -H, -K and -J) that have emerged in the past 5 10 years. The HFMD outbreak in Finland in 2008 was associated with RF-A with the subsequent appearance of this RF across Europe and Asia between 2013 and The more recent emergence of RFs originated from descendants of multiple VP1 lineages that have diverged from RF-A variants circulating in Asia (Thailand and Japan) between 2008 and Enterovirus recombination events can play a significant role in the evolution, and breakpoints detected in this study (2A 2C regions) are well-known recombination hotspots (Lukashev et al., 2005). CV-A6 recombination breakpoints within VP3 and between 5 UTR and VP1 have been detected in the genomes of RF-E variants in Taiwan (Gaunt et al., 2015). In summary, the typical pattern for an RF was its rapid emergence, variable penetrance into the sampled virus population and relatively rapid extinction, within years rather than decades, based on the average recombination half-lives documented for CV-A6 and other EV types. These patterns are well-attested in the turnover of RFs of CV-A6. While we remain relatively ignorant of the reasons for RF turnover, whether driven by immunological, host adaptive factors or transmissibility, or alternatively whether it occurs as a consequence of population bottlenecks and replacements without a fitness component, molecular epidemiological studies will be of value in understanding the nature of enterovirus evolution and the clinical outcomes. Acknowledgements This study was funded by the National Research Council of Thailand, the Research Chair Grant from the National Science and Technology Development Agency, the Center of Excellence in Clinical Virology of Chulalongkorn University (GCE ), King Chulalongkorn Memorial Hospital and the RGJ PhD program (PHD/0087/ 2554). This study was also supported by the Wellcome Trust (WT103767MA) and the Biotechnology and Biological Sciences Research Council (BB/J004324/1). References Bailly, J.-L., Mirand, A., Henquell, C., Archimbaud, C., Chambon, M., Charbonne, F., Traore, O. & Peigue-Lafeuille, H. (2009). Phylogeography of circulating populations of human echovirus 30 over 50 years: nucleotide polymorphism and signature of purifying selection in the VP1 capsid protein gene. Infect Genet Evol 9, Cabrerizo, M., Trallero, G. & Simmonds, P. (2014). Recombination and evolutionary dynamics of human echovirus 6. J Med Virol 86, Calvert, J., Chieochansin, T., Benschop, K. S., McWilliam Leitch, E. C., Drexler, J. F., Grywna, K., da Costa Ribeiro, H., Drosten, C., Harvala, H. & other authors (2010). Recombination dynamics of human parechoviruses: investigation of type-specific differences in frequency and epidemiological correlates. J Gen Virol 91, Drummond, A. J. & Rambaut, A. (2007). BEAST: Bayesian evolutionary analysis by sampling trees. BMC Evol Biol 7, 214. Drummond, A. J., Suchard, M. A., Xie, D. & Rambaut, A. (2012). Bayesian phylogenetics with BEAUTI and the BEAST 1.7. Mol Biol Evol 29, Gaunt, E., Harvala, H., Österback, R., Sreenu, V. B., Thomson, E., Waris, M. & Simmonds, P. (2015). Genetic characterization of human coxsackievirus A6 variants associated with atypical hand, foot and mouth disease: a potential role of recombination in emergence and pathogenicity. J Gen Virol 96, Knowles, N. J., Hovi, T., Hyypia, T., King, A. M. Q., Lindberg, A. M., Pallansch, M. A., Palmenberg, A. C., Simmonds, P. & Skern, T. & other authors (2012). Family Picornaviridae. In Virus Taxonomy: Ninth Report of the International Committee on Taxonomy of Viruses, pp Journal of General Virology 97
7 Evolution of human coxsackievirus A6 Edited by M. J. Adams, E. B. Carstens & E. J. Lefkowitz. London: Academic Press. Lukashev, A. N., Lashkevich, V. A., Ivanova, O. E., Koroleva, G. A., Hinkkanen, A. E. & Ilonen, J. (2005). Recombination in circulating human enterovirus B: independent evolution of structural and non-structural genome regions. J Gen Virol 86, McIntyre, C. L., McWilliam Leitch, E. C., Savolainen-Kopra, C., Hovi, T. & Simmonds, P. (2010). Analysis of genetic diversity and sites of recombination in human rhinovirus species C. J Virol 84, McWilliam Leitch, E. C., Bendig, J., Cabrerizo, M., Cardosa, J., Hyypi a, T., Ivanova, O. E., Kelly, A., Kroes, A. C., Lukashev, A. & other authors (2009). Transmission networks and population turnover of echovirus 30. J Virol 83, McWilliam Leitch, E. C., Cabrerizo, M., Cardosa, J., Harvala, H., Ivanova, O. E., Kroes, A. C., Lukashev, A., Muir, P., Odoom, J. & other authors (2010). Evolutionary dynamics and temporal/geographical correlates of recombination in the human enterovirus echovirus types 9, 11, and 30. J Virol 84, McWilliam Leitch, E. C., Cabrerizo, M., Cardosa, J., Harvala, H., Ivanova, O. E., Koike, S., Kroes, A. C., Lukashev, A., Perera, D. & other authors (2012). The association of recombination events in the founding and emergence of subgenogroup evolutionary lineages of human enterovirus 71. J Virol 86, Oberste, M. S., Maher, K., Kilpatrick, D. R. & Pallansch, M. A. (1999). Molecular evolution of the human enteroviruses: correlation of serotype with VP1 sequence and application to picornavirus classification. J Virol 73, Osterback, R., Vuorinen, T., Linna, M., Susi, P., Hyypi a, T. & Waris, M. (2009). Coxsackievirus A6 and hand, foot, and mouth disease, Finland. Emerg Infect Dis 15, Pons-Salort, M., Parker, E. P. & Grassly, N. C. (2015). The epidemiology of non-polio enteroviruses: recent advances and outstanding questions. Curr Opin Infect Dis 28, Puenpa, J., Theamboonlers, A., Korkong, S., Linsuwanon, P., Thongmee, C., Chatproedprai, S. & Poovorawan, Y. (2011). Molecular characterization and complete genome analysis of human enterovirus 71 and coxsackievirus A16 from children with hand, foot and mouth disease in Thailand during Arch Virol 156, Schuffenecker, I., Mirand, A., Antona, D., Henquell, C., Chomel, J. J., Archimbaud, C., Billaud, G., Peigue-Lafeuille, H., Lina, B. & Bailly, J. L. (2011). Epidemiology of human enterovirus 71 infections in France, J Clin Virol 50, Simmonds, P. (2012). SSE: a nucleotide and amino acid sequence analysis platform. BMC Res Notes 5, 50. Sinclair, C., Gaunt, E., Simmonds, P., Broomfield, D., Nwafor, N., Wellington, L., Templeton, K., Willocks, L., Schofield, O. & Harvala, H. (2014). Atypical hand, foot, and mouth disease associated with coxsackievirus A6 infection, Edinburgh, United Kingdom, January to February Euro Surveill 19, Tamura, K., Stecher, G., Peterson, D., Filipski, A. & Kumar, S. (2013). MEGA6: Molecular evolutionary genetics analysis version 6.0. Mol Biol Evol 30, Whitton, J. L., Cornell, C. T. & Feuer, R. (2005). Host and virus determinants of picornavirus pathogenesis and tropism. Nat Rev Microbiol 3, Wu, Y., Yeo, A., Phoon, M. C., Tan, E. L., Poh, C. L., Quak, S. H. & Chow, V. T. (2010). The largest outbreak of hand; foot and mouth disease in Singapore in 2008: the role of enterovirus 71 and coxsackievirus A strains. Int J Infect Dis 14, e1076 e
The BLAST search on NCBI ( and GISAID
 Supplemental materials and methods The BLAST search on NCBI (http:// www.ncbi.nlm.nih.gov) and GISAID (http://www.platform.gisaid.org) showed that hemagglutinin (HA) gene of North American H5N1, H5N2 and
Supplemental materials and methods The BLAST search on NCBI (http:// www.ncbi.nlm.nih.gov) and GISAID (http://www.platform.gisaid.org) showed that hemagglutinin (HA) gene of North American H5N1, H5N2 and
Epididymitis caused by coxsackievirus A6 in association of hand, foot and mouth disease
 JCM Accepts, published online ahead of print on 17 September 2014 J. Clin. Microbiol. doi:10.1128/jcm.02441-14 Copyright 2014, American Society for Microbiology. All Rights Reserved. 1 1 2 3 4 5 6 7 8
JCM Accepts, published online ahead of print on 17 September 2014 J. Clin. Microbiol. doi:10.1128/jcm.02441-14 Copyright 2014, American Society for Microbiology. All Rights Reserved. 1 1 2 3 4 5 6 7 8
PREVALENCE OF HUMAN ENTEROVIRUS AMONG PATIENTS WITH HAND, FOOT, AND MOUTH DISEASE AND HERPANGINA IN THAILAND, 2013
 Prevalence of enterovirus in Thailand PREVALENCE OF HUMAN ENTEROVIRUS AMONG PATIENTS WITH HAND, FOOT, AND MOUTH DISEASE AND HERPANGINA IN THAILAND, 2013 John Mauleekoonphairoj, Jiratchaya Puenpa, Sumeth
Prevalence of enterovirus in Thailand PREVALENCE OF HUMAN ENTEROVIRUS AMONG PATIENTS WITH HAND, FOOT, AND MOUTH DISEASE AND HERPANGINA IN THAILAND, 2013 John Mauleekoonphairoj, Jiratchaya Puenpa, Sumeth
Genetic Diversity of Coxsackievirus A16 Associated with Hand, Foot, and Mouth Disease Epidemics in Japan from 1983 to 2003
 JOURNAL OF CLINICAL MICROBIOLOGY, Jan. 2007, p. 112 120 Vol. 45, No. 1 0095-1137/07/$08.00 0 doi:10.1128/jcm.00718-06 Copyright 2007, American Society for Microbiology. All Rights Reserved. Genetic Diversity
JOURNAL OF CLINICAL MICROBIOLOGY, Jan. 2007, p. 112 120 Vol. 45, No. 1 0095-1137/07/$08.00 0 doi:10.1128/jcm.00718-06 Copyright 2007, American Society for Microbiology. All Rights Reserved. Genetic Diversity
IgM. (Polioviruses) 71 (EV71) B (Coxsackievirus B) (Virus isolation/ifa, VI-IFA) 7~14 [1,2] (Centers for Disease Control and Prevention, CDC) 1.
![IgM. (Polioviruses) 71 (EV71) B (Coxsackievirus B) (Virus isolation/ifa, VI-IFA) 7~14 [1,2] (Centers for Disease Control and Prevention, CDC) 1. IgM. (Polioviruses) 71 (EV71) B (Coxsackievirus B) (Virus isolation/ifa, VI-IFA) 7~14 [1,2] (Centers for Disease Control and Prevention, CDC) 1.](/thumbs/93/111050187.jpg) 267 DOI: 10.6526/ICJ.2017.603 (Polioviruses) 71 (EV71) B (Coxsackievirus B) [1,2] 1998 EV71 (Centers for Disease Control and Prevention, CDC) 1. (Hand, foot and mouth disease, HFMD) (herpangina) [3,4]
267 DOI: 10.6526/ICJ.2017.603 (Polioviruses) 71 (EV71) B (Coxsackievirus B) [1,2] 1998 EV71 (Centers for Disease Control and Prevention, CDC) 1. (Hand, foot and mouth disease, HFMD) (herpangina) [3,4]
Unusual skin manifestation of hand, foot and mouth disease associated with coxsackievirus A6: cases report
 DOI 10.1186/s40064-015-1143-z CASE STUDY Open Access Unusual skin manifestation of hand, foot and mouth disease associated with coxsackievirus A6: cases report Susheera Chatproedprai, Therdpong Tempark,
DOI 10.1186/s40064-015-1143-z CASE STUDY Open Access Unusual skin manifestation of hand, foot and mouth disease associated with coxsackievirus A6: cases report Susheera Chatproedprai, Therdpong Tempark,
Research Note. Causative agents of hand, foot and mouth disease in University Malaya Medical Centre, Kuala Lumpur, Malaysia in
 Tropical Biomedicine 34(1): 240 248 (2017) Research Note Causative agents of hand, foot and mouth disease in University Malaya Medical Centre, Kuala Lumpur, Malaysia in 2012-2013 Aw-Yong, K.L. 1, Sam,
Tropical Biomedicine 34(1): 240 248 (2017) Research Note Causative agents of hand, foot and mouth disease in University Malaya Medical Centre, Kuala Lumpur, Malaysia in 2012-2013 Aw-Yong, K.L. 1, Sam,
Discovery of Enterovirus A71-like nonstructural genomes in recent circulating viruses of the Enterovirus A species
 Lee et al. Emerging Microbes & Infections (2018) 7:111 DOI 10.1038/s41426-018-0107-0 Emerging Microbes & Infections www.nature.com/emi ARTICLE Discovery of Enterovirus A71-like nonstructural genomes in
Lee et al. Emerging Microbes & Infections (2018) 7:111 DOI 10.1038/s41426-018-0107-0 Emerging Microbes & Infections www.nature.com/emi ARTICLE Discovery of Enterovirus A71-like nonstructural genomes in
Molecular Detection of Human Enterovirus in Raw and Treated Wastewater from a Sewage Treatment Plant in Mahdia, Tunisia
 Molecular Detection of Human Enterovirus in Raw and Treated Wastewater from a Sewage Treatment Plant in Mahdia, Tunisia Ines Othman 1*, Ichrak Slama 1, Jean-Luc Bailly 2, and Mahjoub Aouni 1 1 Faculty
Molecular Detection of Human Enterovirus in Raw and Treated Wastewater from a Sewage Treatment Plant in Mahdia, Tunisia Ines Othman 1*, Ichrak Slama 1, Jean-Luc Bailly 2, and Mahjoub Aouni 1 1 Faculty
Change of Major Genotype of Enterovirus 71 in Outbreaks of Hand-Foot-and-Mouth Disease in Taiwan between 1998 and 2000
 JOURNAL OF CLINICAL MICROBIOLOGY, Jan. 2002, p. 10 15 Vol. 40, No. 1 0095-1137/02/$04.00 0 DOI: 10.1128/JCM.40.1.10 15.2002 Copyright 2002, American Society for Microbiology. All Rights Reserved. Change
JOURNAL OF CLINICAL MICROBIOLOGY, Jan. 2002, p. 10 15 Vol. 40, No. 1 0095-1137/02/$04.00 0 DOI: 10.1128/JCM.40.1.10 15.2002 Copyright 2002, American Society for Microbiology. All Rights Reserved. Change
(ii) The effective population size may be lower than expected due to variability between individuals in infectiousness.
 Supplementary methods Details of timepoints Caió sequences were derived from: HIV-2 gag (n = 86) 16 sequences from 1996, 10 from 2003, 45 from 2006, 13 from 2007 and two from 2008. HIV-2 env (n = 70) 21
Supplementary methods Details of timepoints Caió sequences were derived from: HIV-2 gag (n = 86) 16 sequences from 1996, 10 from 2003, 45 from 2006, 13 from 2007 and two from 2008. HIV-2 env (n = 70) 21
Midgley, Sofie E; Nielsen, Astrid G; Trebbien, Ramona; Poulsen, Mille Weismann; Andersen, Peter H; Fischer, Thea Kølsen
 Syddansk Universitet Co-circulation of multiple subtypes of enterovirus A71 (EV- A71) genotype C, including novel recombinants characterised by use of whole genome sequencing (WGS), Denmark 216 Midgley,
Syddansk Universitet Co-circulation of multiple subtypes of enterovirus A71 (EV- A71) genotype C, including novel recombinants characterised by use of whole genome sequencing (WGS), Denmark 216 Midgley,
Sequence analysis for VP4 of enterovirus 71 isolated in Beijing during 2007 to 2008
 16 2009 3 4 1 Journal of Microbes and Infection, March 2009, Vol. 4, No. 1 2007 2008 71 VP4 1, 2, 2, 2, 1, 2, 2, 2, 1, 2 1., 100730; 2., 100020 : 2007 2008 71 ( EV71), 2007 3 EV71( 1, 2 ) 2008 5 EV71(
16 2009 3 4 1 Journal of Microbes and Infection, March 2009, Vol. 4, No. 1 2007 2008 71 VP4 1, 2, 2, 2, 1, 2, 2, 2, 1, 2 1., 100730; 2., 100020 : 2007 2008 71 ( EV71), 2007 3 EV71( 1, 2 ) 2008 5 EV71(
Enterovirus 71 Outbreak in P. R. China, 2008
 JCM Accepts, published online ahead of print on 13 May 2009 J. Clin. Microbiol. doi:10.1128/jcm.00563-09 Copyright 2009, American Society for Microbiology and/or the Listed Authors/Institutions. All Rights
JCM Accepts, published online ahead of print on 13 May 2009 J. Clin. Microbiol. doi:10.1128/jcm.00563-09 Copyright 2009, American Society for Microbiology and/or the Listed Authors/Institutions. All Rights
Cyril C. Y. Yip Susanna K. P. Lau Boping Zhou Ming-Xia Zhang Hoi-Wah Tsoi Kwok-Hung Chan Xin-Chun Chen Patrick C. Y. Woo Kwok-Yung Yuen
 rch Virol DOI 10.7/s00705-010-0722-0 ORIGINL RTICLE Emergence of enterovirus 71 double-recombinant strains belonging to a novel genotype D originating from southern China: first evidence for combination
rch Virol DOI 10.7/s00705-010-0722-0 ORIGINL RTICLE Emergence of enterovirus 71 double-recombinant strains belonging to a novel genotype D originating from southern China: first evidence for combination
Evidence for enteroviral persistence in humans
 Journal of General Virology (1997), 78, 307 312. Printed in Great Britain...... SHORT COMMUNICATION Evidence for enteroviral persistence in humans Daniel N. Galbraith, Carron Nairn and Geoffrey B. Clements
Journal of General Virology (1997), 78, 307 312. Printed in Great Britain...... SHORT COMMUNICATION Evidence for enteroviral persistence in humans Daniel N. Galbraith, Carron Nairn and Geoffrey B. Clements
Downloaded from:
 Ruis, C; Roy, S; Brown, JR; Allen, DJ; Goldstein, RA; Breuer, J (2017) The emerging GII.P16-GII.4 Sydney 2012 norovirus lineage is circulating worldwide, arose by late-2014 and contains polymerase changes
Ruis, C; Roy, S; Brown, JR; Allen, DJ; Goldstein, RA; Breuer, J (2017) The emerging GII.P16-GII.4 Sydney 2012 norovirus lineage is circulating worldwide, arose by late-2014 and contains polymerase changes
Avian Influenza Virus H7N9. Dr. Di Liu Network Information Center Institute of Microbiology Chinese Academy of Sciences
 Avian Influenza Virus H7N9 Dr. Di Liu Network Information Center Institute of Microbiology Chinese Academy of Sciences Avian Influenza Virus RNA virus, Orthomyxoviruses Influenza A virus Eight Gene segments
Avian Influenza Virus H7N9 Dr. Di Liu Network Information Center Institute of Microbiology Chinese Academy of Sciences Avian Influenza Virus RNA virus, Orthomyxoviruses Influenza A virus Eight Gene segments
Enterovirus genotypes causing hand foot and mouth disease in Shanghai, China: a molecular epidemiological analysis
 Xu et al. BMC Infectious Diseases 2013, 13:489 RESEARCH ARTICLE Open Access Enterovirus genotypes causing hand foot and mouth disease in Shanghai, China: a molecular epidemiological analysis Menghua Xu,
Xu et al. BMC Infectious Diseases 2013, 13:489 RESEARCH ARTICLE Open Access Enterovirus genotypes causing hand foot and mouth disease in Shanghai, China: a molecular epidemiological analysis Menghua Xu,
Evidence for Other Non-Poliovirus Enteroviruses Multiplies in L20B Cells. ACCEPTED LUIS SARMIENTO*, PEDRO MÁS, ROSA PALOMERA, LUIS MORIER, MAGILÉ
 CVI Accepts, published online ahead of print on 14 March 2007 Clin. Vaccine Immunol. doi:10.1128/cvi.00017-06 Copyright 2007, American Society for Microbiology and/or the Listed Authors/Institutions. All
CVI Accepts, published online ahead of print on 14 March 2007 Clin. Vaccine Immunol. doi:10.1128/cvi.00017-06 Copyright 2007, American Society for Microbiology and/or the Listed Authors/Institutions. All
Enterovirus D68-US Outbreak 2014 Laboratory Perspective
 Enterovirus D68-US Outbreak 2014 Laboratory Perspective Allan Nix Team Lead Picornavirus Laboratory Polio and Picornavirus Laboratory Branch PAHO / SARInet Webinar November 6, 2014 National Center for
Enterovirus D68-US Outbreak 2014 Laboratory Perspective Allan Nix Team Lead Picornavirus Laboratory Polio and Picornavirus Laboratory Branch PAHO / SARInet Webinar November 6, 2014 National Center for
Molecular epidemiology of enterovirus 71, coxsackievirus A16 and A6 associated with hand, foot and mouth disease in Spain
 ORIGINAL ARTICLE VIROLOGY Molecular epidemiology of enterovirus 71, coxsackievirus A16 and A6 associated with hand, foot and mouth disease in Spain M. Cabrerizo 1, D. Tarrago 2,C.Mu~noz-Almagro 3, E. del
ORIGINAL ARTICLE VIROLOGY Molecular epidemiology of enterovirus 71, coxsackievirus A16 and A6 associated with hand, foot and mouth disease in Spain M. Cabrerizo 1, D. Tarrago 2,C.Mu~noz-Almagro 3, E. del
Evolution of influenza
 Evolution of influenza Today: 1. Global health impact of flu - why should we care? 2. - what are the components of the virus and how do they change? 3. Where does influenza come from? - are there animal
Evolution of influenza Today: 1. Global health impact of flu - why should we care? 2. - what are the components of the virus and how do they change? 3. Where does influenza come from? - are there animal
Evolution of hepatitis C virus in blood donors and their respective recipients
 Journal of General Virology (2003), 84, 441 446 DOI 10.1099/vir.0.18642-0 Short Communication Correspondence Jean-François Cantaloube jfc-ets-ap@gulliver.fr Evolution of hepatitis C virus in blood donors
Journal of General Virology (2003), 84, 441 446 DOI 10.1099/vir.0.18642-0 Short Communication Correspondence Jean-François Cantaloube jfc-ets-ap@gulliver.fr Evolution of hepatitis C virus in blood donors
Prevalence of human parechovirus in Chinese children hospitalized for acute gastroenteritis
 ORIGINAL ARTICLE VIROLOGY Prevalence of human parechovirus in Chinese children hospitalized for acute gastroenteritis D.-L. Zhang 1,2, Y. Jin 3, D.-D. Li 1, W.-X. Cheng 1,3, Z.-Q. Xu 1, J.-M. Yu 1, M.
ORIGINAL ARTICLE VIROLOGY Prevalence of human parechovirus in Chinese children hospitalized for acute gastroenteritis D.-L. Zhang 1,2, Y. Jin 3, D.-D. Li 1, W.-X. Cheng 1,3, Z.-Q. Xu 1, J.-M. Yu 1, M.
Evolutionary interactions between haemagglutinin and neuraminidase in avian influenza
 Ward et al. BMC Evolutionary Biology 2013, 13:222 RESEARCH ARTICLE Open Access Evolutionary interactions between haemagglutinin and neuraminidase in avian influenza Melissa J Ward 1*, Samantha J Lycett
Ward et al. BMC Evolutionary Biology 2013, 13:222 RESEARCH ARTICLE Open Access Evolutionary interactions between haemagglutinin and neuraminidase in avian influenza Melissa J Ward 1*, Samantha J Lycett
Envelope e_ :------, Envelope glycoproteins e_ ~ Single-stranded RNA ----, Nucleocapsid
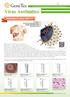 Virus Antib(tdies Your Expertise, Our Antibodies, Accelerated Discovery. Envelope e_---------:------, Envelope glycoproteins e_-------1~ Single-stranded RNA ----, Nucleocapsid First identified in 1989,
Virus Antib(tdies Your Expertise, Our Antibodies, Accelerated Discovery. Envelope e_---------:------, Envelope glycoproteins e_-------1~ Single-stranded RNA ----, Nucleocapsid First identified in 1989,
Author's response to reviews
 Author's response to reviews Title: Sentinel surveillance for human enterovirus 71 in Sarawak, Malaysia: Lessons from the first 7 years. Authors: Yuwana Podin (ypodin@yahoo.com) Edna LM Gias (amberr74@yahoo.com)
Author's response to reviews Title: Sentinel surveillance for human enterovirus 71 in Sarawak, Malaysia: Lessons from the first 7 years. Authors: Yuwana Podin (ypodin@yahoo.com) Edna LM Gias (amberr74@yahoo.com)
Human parechovirus type 1, 3, 4, 5 and 6 detection in picornavirus cultures ACCEPTED. Running title: HPeV type 1, 3, 4, 5 and 6 in virus cultures
 JCM Accepts, published online ahead of print on 12 December 2007 J. Clin. Microbiol. doi:10.1128/jcm.02009-07 Copyright 2007, American Society for Microbiology and/or the Listed Authors/Institutions. All
JCM Accepts, published online ahead of print on 12 December 2007 J. Clin. Microbiol. doi:10.1128/jcm.02009-07 Copyright 2007, American Society for Microbiology and/or the Listed Authors/Institutions. All
Noronet report, April 2014
 Noronet report, April 2014 Janko van Beek, Annelies Kroneman, Harry Vennema, Marion Koopmans A. van Leeuwenhoeklaan 9 3721 MA Bilthoven Postbus 1 3720 BA Bilthoven www.rivm.nl T 030 274 91 11 F 030 274
Noronet report, April 2014 Janko van Beek, Annelies Kroneman, Harry Vennema, Marion Koopmans A. van Leeuwenhoeklaan 9 3721 MA Bilthoven Postbus 1 3720 BA Bilthoven www.rivm.nl T 030 274 91 11 F 030 274
Xiang Yan, 1,2 Zhen-Zhen Zhang, 1 Zhen-Hua Yang, 2 Chao-Min Zhu, 1 Yun-Ge Hu, 1 and Quan-Bo Liu Introduction
 BioMed Research International Volume 2015, Article ID 802046, 8 pages http://dx.doi.org/10.1155/2015/802046 Research Article Clinical and Etiological Characteristics of Atypical Hand-Foot-and-Mouth Disease
BioMed Research International Volume 2015, Article ID 802046, 8 pages http://dx.doi.org/10.1155/2015/802046 Research Article Clinical and Etiological Characteristics of Atypical Hand-Foot-and-Mouth Disease
Enteroviruses Associated with Aseptic Meningitis in Poland,
 Polish Journal of Microbiology 2016, Vol. 65, No 2, 231 235 SHORT COMMUNICATION Enteroviruses Associated with Aseptic Meningitis in Poland, 2011 2014 MAGDALENA WIECZOREK*, AGNIESZKA FIGAS and ARLETA KRZYSZTOSZEK
Polish Journal of Microbiology 2016, Vol. 65, No 2, 231 235 SHORT COMMUNICATION Enteroviruses Associated with Aseptic Meningitis in Poland, 2011 2014 MAGDALENA WIECZOREK*, AGNIESZKA FIGAS and ARLETA KRZYSZTOSZEK
From Mosquitos to Humans: Genetic evolution of Zika Virus
 Article: From Mosquitos to Humans: Genetic evolution of Zika Virus Renata Pellegrino, PhD Director, Sequencing lab Center for Applied Genomics The Children s Hospital of Philadelphia Journal Club Clinical
Article: From Mosquitos to Humans: Genetic evolution of Zika Virus Renata Pellegrino, PhD Director, Sequencing lab Center for Applied Genomics The Children s Hospital of Philadelphia Journal Club Clinical
The molecular clock of HIV-1 unveiled through analysis of a known transmission history
 Proc. Natl. Acad. Sci. USA Vol. 96, pp. 10752 10757, September 1999 Evolution The molecular clock of HIV-1 unveiled through analysis of a known transmission history THOMAS LEITNER, AND JAN ALBERT Theoretical
Proc. Natl. Acad. Sci. USA Vol. 96, pp. 10752 10757, September 1999 Evolution The molecular clock of HIV-1 unveiled through analysis of a known transmission history THOMAS LEITNER, AND JAN ALBERT Theoretical
Arevir meeting Lize Cuypers
 Arevir meeting 2017 Reconstructing the HCV migration history to support public health efforts Lize Cuypers KU Leuven University of Leuven, Department of Microbiology and Immunology, Rega Institute for
Arevir meeting 2017 Reconstructing the HCV migration history to support public health efforts Lize Cuypers KU Leuven University of Leuven, Department of Microbiology and Immunology, Rega Institute for
Enterovirus surveillance reveals proposed new serotypes and provides new insight into enterovirus 59-untranslated region evolution
 Journal of General Virology (2007), 88, 2520 2526 DOI 10.1099/vir.0.82866-0 Short Communication Correspondence Merja Roivainen Merja.Roivainen@ktl.fi Enterovirus surveillance reveals proposed new serotypes
Journal of General Virology (2007), 88, 2520 2526 DOI 10.1099/vir.0.82866-0 Short Communication Correspondence Merja Roivainen Merja.Roivainen@ktl.fi Enterovirus surveillance reveals proposed new serotypes
NHS Lothian, NHS Fife
 Investigation of a cluster of cases of acute flaccid paralysis associated with Enterovirus D68 infection (EVD68) in children in South East Scotland, 2016 Catriona Waugh, Dr Kate Mark, Dr Janet Stevenson,
Investigation of a cluster of cases of acute flaccid paralysis associated with Enterovirus D68 infection (EVD68) in children in South East Scotland, 2016 Catriona Waugh, Dr Kate Mark, Dr Janet Stevenson,
tion, the parents of all patients were informed as to the purpose of the study and written consent was obtained. Study population: The subjects were r
 Jpn. J. Infect. Dis., 63, 229 233, 2010 Original Article Clinical and Molecular Characterization of Hand Foot and Mouth Disease in Thailand, 2008 2009 Susheera Chatproedprai, Apiradee Theanboonlers 1,
Jpn. J. Infect. Dis., 63, 229 233, 2010 Original Article Clinical and Molecular Characterization of Hand Foot and Mouth Disease in Thailand, 2008 2009 Susheera Chatproedprai, Apiradee Theanboonlers 1,
An update on the laboratory detection and epidemiology of astrovirus, adenovirus, sapovirus, and enterovirus in gastrointestinal disease
 An update on the laboratory detection and epidemiology of astrovirus, adenovirus, sapovirus, and enterovirus in gastrointestinal disease Christopher McIver, Principal Hospital Scientist, Microbiology Department
An update on the laboratory detection and epidemiology of astrovirus, adenovirus, sapovirus, and enterovirus in gastrointestinal disease Christopher McIver, Principal Hospital Scientist, Microbiology Department
Supplementary Online Content
 Supplementary Online Content Rodger AJ, Cambiano V, Bruun T, et al; PARTNER Study Group. Sexual activity without condoms and risk of HIV transmission in serodifferent couples when the HIVpositive partner
Supplementary Online Content Rodger AJ, Cambiano V, Bruun T, et al; PARTNER Study Group. Sexual activity without condoms and risk of HIV transmission in serodifferent couples when the HIVpositive partner
Circulation of multiple enterovirus serotypes causing hand, foot and mouth disease in India
 Journal of Medical Microbiology (2012), 61, 420 425 DOI 10.1099/jmm.0.036400-0 Circulation of multiple enterovirus serotypes causing hand, foot and mouth disease in India V. Gopalkrishna, Pooja R. Patil,
Journal of Medical Microbiology (2012), 61, 420 425 DOI 10.1099/jmm.0.036400-0 Circulation of multiple enterovirus serotypes causing hand, foot and mouth disease in India V. Gopalkrishna, Pooja R. Patil,
R E V I E W SUMMARY. Reviews in Medical Virology
 Reviews in Medical Virology R E V I E W Rev. Med. Virol. 2010; 20: 327 337. Published online 14 July 2010 in Wiley Online Library (wileyonlinelibrary.com)..660 Recombination among picornaviruses A.N. Lukashev
Reviews in Medical Virology R E V I E W Rev. Med. Virol. 2010; 20: 327 337. Published online 14 July 2010 in Wiley Online Library (wileyonlinelibrary.com)..660 Recombination among picornaviruses A.N. Lukashev
Bioinformation by Biomedical Informatics Publishing Group
 Predicted RNA secondary structures for the conserved regions in dengue virus Pallavi Somvanshi*, Prahlad Kishore Seth Bioinformatics Centre, Biotech Park, Sector G, Jankipuram, Lucknow 226021, Uttar Pradesh,
Predicted RNA secondary structures for the conserved regions in dengue virus Pallavi Somvanshi*, Prahlad Kishore Seth Bioinformatics Centre, Biotech Park, Sector G, Jankipuram, Lucknow 226021, Uttar Pradesh,
To test the possible source of the HBV infection outside the study family, we searched the Genbank
 Supplementary Discussion The source of hepatitis B virus infection To test the possible source of the HBV infection outside the study family, we searched the Genbank and HBV Database (http://hbvdb.ibcp.fr),
Supplementary Discussion The source of hepatitis B virus infection To test the possible source of the HBV infection outside the study family, we searched the Genbank and HBV Database (http://hbvdb.ibcp.fr),
Atypical hand-foot-mouth disease in children: a hospital-based prospective cohort study
 Huang et al. Virology Journal 2013, 10:209 RESEARCH Open Access Atypical hand-foot-mouth disease in children: a hospital-based prospective cohort study Wen-Chan Huang, Li-Min Huang, Chun-Yi Lu, Ai-Ling
Huang et al. Virology Journal 2013, 10:209 RESEARCH Open Access Atypical hand-foot-mouth disease in children: a hospital-based prospective cohort study Wen-Chan Huang, Li-Min Huang, Chun-Yi Lu, Ai-Ling
Control and Management of EV 71 Associated HFMD
 Control and Management of EV 71 Associated HFMD Amr Ismail MBBCh, MSc, MD, MACP, FISQua, FABAM, CHPI Consultant Dermatologist Objectives To overview EV 71 associated HFMD outbreaks. To describe prevention
Control and Management of EV 71 Associated HFMD Amr Ismail MBBCh, MSc, MD, MACP, FISQua, FABAM, CHPI Consultant Dermatologist Objectives To overview EV 71 associated HFMD outbreaks. To describe prevention
Distinguishing epidemiological dependent from treatment (resistance) dependent HIV mutations: Problem Statement
 Distinguishing epidemiological dependent from treatment (resistance) dependent HIV mutations: Problem Statement Leander Schietgat 1, Kristof Theys 2, Jan Ramon 1, Hendrik Blockeel 1, and Anne-Mieke Vandamme
Distinguishing epidemiological dependent from treatment (resistance) dependent HIV mutations: Problem Statement Leander Schietgat 1, Kristof Theys 2, Jan Ramon 1, Hendrik Blockeel 1, and Anne-Mieke Vandamme
Enterovirus 71 infection in children with hand, foot, and mouth disease in Shanghai, China: epidemiology, clinical feature and diagnosis
 Wang et al. Virology Journal (2015) 12:83 DOI 10.1186/s12985-015-0308-2 RESEARCH Enterovirus 71 infection in children with hand, foot, and mouth disease in Shanghai, China: epidemiology, clinical feature
Wang et al. Virology Journal (2015) 12:83 DOI 10.1186/s12985-015-0308-2 RESEARCH Enterovirus 71 infection in children with hand, foot, and mouth disease in Shanghai, China: epidemiology, clinical feature
Intercity Spread of Echovirus 6 in Shandong Province, China: Application of Environmental Surveillance in Tracing Circulating
 AEM Accepts, published online ahead of print on 27 July 2012 Appl. Environ. Microbiol. doi:10.1128/aem.01861-12 Copyright 2012, American Society for Microbiology. All Rights Reserved. 1 2 3 Intercity Spread
AEM Accepts, published online ahead of print on 27 July 2012 Appl. Environ. Microbiol. doi:10.1128/aem.01861-12 Copyright 2012, American Society for Microbiology. All Rights Reserved. 1 2 3 Intercity Spread
DOI: /ICJ poliovirus. [14] (Hand-Foot-Mouth Disease, HFMD) (Herpangina) 71 [26] [17,18,27] 71 picornaviridae
![DOI: /ICJ poliovirus. [14] (Hand-Foot-Mouth Disease, HFMD) (Herpangina) 71 [26] [17,18,27] 71 picornaviridae DOI: /ICJ poliovirus. [14] (Hand-Foot-Mouth Disease, HFMD) (Herpangina) 71 [26] [17,18,27] 71 picornaviridae](/thumbs/93/112612467.jpg) 174 DOI: 10.6526/ICJ.2016.404 71 71 [14] (Hand-Foot-Mouth Disease, HFMD) (Herpangina) 71 71 picornaviridae poliovirus / 1990 71 [26] [17,18,27] 71 175 [31] ICR NOD/SCID AG129 hpsgl-1 hscarb2 (MP4) (MP4)
174 DOI: 10.6526/ICJ.2016.404 71 71 [14] (Hand-Foot-Mouth Disease, HFMD) (Herpangina) 71 71 picornaviridae poliovirus / 1990 71 [26] [17,18,27] 71 175 [31] ICR NOD/SCID AG129 hpsgl-1 hscarb2 (MP4) (MP4)
Rajesh Kannangai Phone: ; Fax: ; *Corresponding author
 Amino acid sequence divergence of Tat protein (exon1) of subtype B and C HIV-1 strains: Does it have implications for vaccine development? Abraham Joseph Kandathil 1, Rajesh Kannangai 1, *, Oriapadickal
Amino acid sequence divergence of Tat protein (exon1) of subtype B and C HIV-1 strains: Does it have implications for vaccine development? Abraham Joseph Kandathil 1, Rajesh Kannangai 1, *, Oriapadickal
Received 19 May 2009/Returned for modification 7 August 2009/Accepted 2 October 2009
 JOURNAL OF CLINICAL MICROBIOLOGY, Dec. 2009, p. 3958 37 Vol. 47, No. 12 0095-1137/09/$12.00 doi:10.1128/jcm.003-09 Copyright 2009, American Society for Microbiology. All Rights Reserved. Screening Respiratory
JOURNAL OF CLINICAL MICROBIOLOGY, Dec. 2009, p. 3958 37 Vol. 47, No. 12 0095-1137/09/$12.00 doi:10.1128/jcm.003-09 Copyright 2009, American Society for Microbiology. All Rights Reserved. Screening Respiratory
Rapid Simultaneous Detection of Enterovirus and Parechovirus RNAs in Clinical Samples by One-Step Real-Time Reverse Transcription-PCR Assay
 JOURNAL OF CLINICAL MICROBIOLOGY, July 2011, p. 2620 2624 Vol. 49, No. 7 0095-1137/11/$12.00 doi:10.1128/jcm.02445-10 Copyright 2011, American Society for Microbiology. All Rights Reserved. Rapid Simultaneous
JOURNAL OF CLINICAL MICROBIOLOGY, July 2011, p. 2620 2624 Vol. 49, No. 7 0095-1137/11/$12.00 doi:10.1128/jcm.02445-10 Copyright 2011, American Society for Microbiology. All Rights Reserved. Rapid Simultaneous
The epidemiology of non-polio enteroviruses: recent advances and outstanding questions
 The epidemiology of non-polio enteroviruses: recent advances and outstanding questions Margarita Pons-Salort, Edward P. K. Parker, Nicholas C. Grassly Department of Infectious Disease Epidemiology, St
The epidemiology of non-polio enteroviruses: recent advances and outstanding questions Margarita Pons-Salort, Edward P. K. Parker, Nicholas C. Grassly Department of Infectious Disease Epidemiology, St
Host Dependent Evolutionary Patterns and the Origin of 2009 H1N1 Pandemic Influenza
 Host Dependent Evolutionary Patterns and the Origin of 2009 H1N1 Pandemic Influenza The origin of H1N1pdm constitutes an unresolved mystery, as its most recently observed ancestors were isolated in pigs
Host Dependent Evolutionary Patterns and the Origin of 2009 H1N1 Pandemic Influenza The origin of H1N1pdm constitutes an unresolved mystery, as its most recently observed ancestors were isolated in pigs
Full-Genome Sequence Analysis of a Multirecombinant Echovirus 3 Strain Isolated from Sewage in Greece
 JOURNAL OF CLINICAL MICROBIOLOGY, May 2010, p. 1513 1519 Vol. 48, No. 5 0095-1137/10/$12.00 doi:10.1128/jcm.00475-09 Copyright 2010, American Society for Microbiology. All Rights Reserved. Full-Genome
JOURNAL OF CLINICAL MICROBIOLOGY, May 2010, p. 1513 1519 Vol. 48, No. 5 0095-1137/10/$12.00 doi:10.1128/jcm.00475-09 Copyright 2010, American Society for Microbiology. All Rights Reserved. Full-Genome
Abstract. Introduction
 ORIGINAL ARTICLE VIROLOGY Outbreak of hand, foot and mouth disease/herpangina associated with coxsackievirus A6 and A10 infections in 2010, France: a large citywide, prospective observational study A.
ORIGINAL ARTICLE VIROLOGY Outbreak of hand, foot and mouth disease/herpangina associated with coxsackievirus A6 and A10 infections in 2010, France: a large citywide, prospective observational study A.
Phylodynamic Reconstruction Reveals Norovirus GII.4 Epidemic Expansions and their Molecular Determinants
 Phylodynamic Reconstruction Reveals Norovirus GII.4 Epidemic Expansions and their Molecular Determinants J. Joukje Siebenga 1,2 *., Philippe Lemey 3., Sergei L. Kosakovsky Pond 4, Andrew Rambaut 5, Harry
Phylodynamic Reconstruction Reveals Norovirus GII.4 Epidemic Expansions and their Molecular Determinants J. Joukje Siebenga 1,2 *., Philippe Lemey 3., Sergei L. Kosakovsky Pond 4, Andrew Rambaut 5, Harry
Modeling the Antigenic Evolution of Influenza Viruses from Sequences
 Modeling the Antigenic Evolution of Influenza Viruses from Sequences Taijiao Jiang Center of Systems Medicine, Chinese Academy of Medical Sciences Suzhou Institute of Systems Medicine October 8-10, 2015.
Modeling the Antigenic Evolution of Influenza Viruses from Sequences Taijiao Jiang Center of Systems Medicine, Chinese Academy of Medical Sciences Suzhou Institute of Systems Medicine October 8-10, 2015.
Phylogenetic Analysis of Enterovirus 71 Circulating in Beijing, China from 2007 to 2009
 Phylogenetic Analysis of Enterovirus 71 Circulating in Beijing, China from 2007 to 2009 Junping Zhu 1, Zhen Luo 2, Juan Wang 1, Zigang Xu 2, Hui Chen 1, Dongying Fan 1, Na Gao 1, Guoling Ping 1, Zhen Zhou
Phylogenetic Analysis of Enterovirus 71 Circulating in Beijing, China from 2007 to 2009 Junping Zhu 1, Zhen Luo 2, Juan Wang 1, Zigang Xu 2, Hui Chen 1, Dongying Fan 1, Na Gao 1, Guoling Ping 1, Zhen Zhou
Principles of phylogenetic analysis
 Principles of phylogenetic analysis Arne Holst-Jensen, NVI, Norway. Fusarium course, Ås, Norway, June 22 nd 2008 Distance based methods Compare C OTUs and characters X A + D = Pairwise: A and B; X characters
Principles of phylogenetic analysis Arne Holst-Jensen, NVI, Norway. Fusarium course, Ås, Norway, June 22 nd 2008 Distance based methods Compare C OTUs and characters X A + D = Pairwise: A and B; X characters
Epidemiology of childhood enterovirus infections in Hangzhou, China
 Li et al. Virology Journal (2015) 12:58 DOI 10.1186/s12985-015-0294-4 RESEARCH Open Access Epidemiology of childhood enterovirus infections in Hangzhou, China Wei Li, Xiao Zhang, Xi Chen, Yu-Ping Cheng,
Li et al. Virology Journal (2015) 12:58 DOI 10.1186/s12985-015-0294-4 RESEARCH Open Access Epidemiology of childhood enterovirus infections in Hangzhou, China Wei Li, Xiao Zhang, Xi Chen, Yu-Ping Cheng,
Enterovirus Co-infections and Onychomadesis after Hand, Foot, and Mouth Disease, Spain, 2008
 Enterovirus Co-infections and Onychomadesis after Hand, Foot, and Mouth Disease, Spain, 2008 Maria A. Bracho, Fernando González-Candelas, Ana Valero, Juan Córdoba, and Antonio Salazar Hand, foot, and mouth
Enterovirus Co-infections and Onychomadesis after Hand, Foot, and Mouth Disease, Spain, 2008 Maria A. Bracho, Fernando González-Candelas, Ana Valero, Juan Córdoba, and Antonio Salazar Hand, foot, and mouth
NEXT GENERATION SEQUENCING OPENS NEW VIEWS ON VIRUS EVOLUTION AND EPIDEMIOLOGY. 16th International WAVLD symposium, 10th OIE Seminar
 NEXT GENERATION SEQUENCING OPENS NEW VIEWS ON VIRUS EVOLUTION AND EPIDEMIOLOGY S. Van Borm, I. Monne, D. King and T. Rosseel 16th International WAVLD symposium, 10th OIE Seminar 07.06.2013 Viral livestock
NEXT GENERATION SEQUENCING OPENS NEW VIEWS ON VIRUS EVOLUTION AND EPIDEMIOLOGY S. Van Borm, I. Monne, D. King and T. Rosseel 16th International WAVLD symposium, 10th OIE Seminar 07.06.2013 Viral livestock
Phylogenetic analysis of the major causative agents of hand, foot and mouth disease in Suzhou city, Jiangsu province, China, in
 OPEN (1) 4, e12; doi:.38/emi.1.12 ß 1 SSCC. All rights reserved 2222-171/1 www.nature.com/emi ORIGINAL ARTICLE Phylogenetic analysis of the major causative agents of hand, foot and mouth disease in Suzhou
OPEN (1) 4, e12; doi:.38/emi.1.12 ß 1 SSCC. All rights reserved 2222-171/1 www.nature.com/emi ORIGINAL ARTICLE Phylogenetic analysis of the major causative agents of hand, foot and mouth disease in Suzhou
Tutorial using BEAST v2.4.6 Prior-selection Veronika Bošková, Venelin Mitov and Louis du Plessis
 Tutorial using BEAST v2.4.6 Prior-selection Veronika Bošková, Venelin Mitov and Louis du Plessis Prior selection and clock calibration using Influenza A data. 1 Background In the Bayesian analysis of sequence
Tutorial using BEAST v2.4.6 Prior-selection Veronika Bošková, Venelin Mitov and Louis du Plessis Prior selection and clock calibration using Influenza A data. 1 Background In the Bayesian analysis of sequence
Contents. lessmore. 1 of 11 6/2/ :06 PM
 1 of 11 6/2/2010 12:06 PM Background Since its initial detection in April 2009, the A/H1N1pdm influenza virus has spread rapidly in humans, with over 5,700 human deaths. However, little is known about
1 of 11 6/2/2010 12:06 PM Background Since its initial detection in April 2009, the A/H1N1pdm influenza virus has spread rapidly in humans, with over 5,700 human deaths. However, little is known about
Molecular Epidemiology of HBV Genotypes Circulating In Acute Hepatitis B Patients In The Campania Region
 Molecular Epidemiology of HBV Genotypes Circulating In Acute Hepatitis B Patients In The Campania Region Caterina Sagnelli 1, Massimo Ciccozzi 2,3, Mariantonietta Pisaturo 4, Gianguglielmo Zehender 5,
Molecular Epidemiology of HBV Genotypes Circulating In Acute Hepatitis B Patients In The Campania Region Caterina Sagnelli 1, Massimo Ciccozzi 2,3, Mariantonietta Pisaturo 4, Gianguglielmo Zehender 5,
Rotavirus Genotyping and Enhanced Annotation in the Virus Pathogen Resource (ViPR) Yun Zhang J. Craig Venter Institute ASV 2016 June 19, 2016
 Rotavirus Genotyping and Enhanced Annotation in the Virus Pathogen Resource (ViPR) Yun Zhang J. Craig Venter Institute ASV 2016 June 19, 2016 Loading Virus Pathogen Database and Analysis About Resource
Rotavirus Genotyping and Enhanced Annotation in the Virus Pathogen Resource (ViPR) Yun Zhang J. Craig Venter Institute ASV 2016 June 19, 2016 Loading Virus Pathogen Database and Analysis About Resource
Noronet report, April 2013
 Noronet report, April 2013 Janko van Beek, Annelies Kroneman, Harry Vennema, Marion Koopmans National Institute for Public Health and the Environment, Bilthoven, The Netherlands The major aim of Noronet
Noronet report, April 2013 Janko van Beek, Annelies Kroneman, Harry Vennema, Marion Koopmans National Institute for Public Health and the Environment, Bilthoven, The Netherlands The major aim of Noronet
Recombination and Selection in the Evolution of Picornaviruses and Other Mammalian Positive-Stranded RNA Viruses
 JOURNAL OF VIROLOGY, Nov. 2006, p. 11124 11140 Vol. 80, No. 22 0022-538X/06/$08.00 0 doi:10.1128/jvi.01076-06 Copyright 2006, American Society for Microbiology. All Rights Reserved. Recombination and Selection
JOURNAL OF VIROLOGY, Nov. 2006, p. 11124 11140 Vol. 80, No. 22 0022-538X/06/$08.00 0 doi:10.1128/jvi.01076-06 Copyright 2006, American Society for Microbiology. All Rights Reserved. Recombination and Selection
Characterizing intra-host influenza virus populations to predict emergence
 Characterizing intra-host influenza virus populations to predict emergence June 12, 2012 Forum on Microbial Threats Washington, DC Elodie Ghedin Center for Vaccine Research Dept. Computational & Systems
Characterizing intra-host influenza virus populations to predict emergence June 12, 2012 Forum on Microbial Threats Washington, DC Elodie Ghedin Center for Vaccine Research Dept. Computational & Systems
Supplementary Appendix
 Supplementary Appendix This appendix has been provided by the authors to give readers additional information about their work. Supplement to: Knox DC, Anderson PL, Harrigan PR, Tan DHS. Multidrug-resistant
Supplementary Appendix This appendix has been provided by the authors to give readers additional information about their work. Supplement to: Knox DC, Anderson PL, Harrigan PR, Tan DHS. Multidrug-resistant
The use of nonhuman primates in biomedical research has led to the isolation of many
 JVI Accepts, published online ahead of print on 29 September 2010 J. Virol. doi:10.1128/jvi.01928-10 Copyright 2010, American Society for Microbiology and/or the Listed Authors/Institutions. All Rights
JVI Accepts, published online ahead of print on 29 September 2010 J. Virol. doi:10.1128/jvi.01928-10 Copyright 2010, American Society for Microbiology and/or the Listed Authors/Institutions. All Rights
Coronaviruses. Virion. Genome. Genes and proteins. Viruses and hosts. Diseases. Distinctive characteristics
 Coronaviruses Virion Genome Genes and proteins Viruses and hosts Diseases Distinctive characteristics Virion Spherical enveloped particles studded with clubbed spikes Diameter 120-160 nm Coiled helical
Coronaviruses Virion Genome Genes and proteins Viruses and hosts Diseases Distinctive characteristics Virion Spherical enveloped particles studded with clubbed spikes Diameter 120-160 nm Coiled helical
Risk assessment of seasonal influenza - Update, EU/EEA, January 2017
 RAPID RISK ASSESSMENT Risk assessment of seasonal influenza - Update, EU/EEA, 2016-2017 24 January 2017 Conclusions and options for response Most countries with high influenza activity have reported severe
RAPID RISK ASSESSMENT Risk assessment of seasonal influenza - Update, EU/EEA, 2016-2017 24 January 2017 Conclusions and options for response Most countries with high influenza activity have reported severe
Whole Genomic Sequence and Replication Kinetics of a New Enterovirus C96 Isolated from Guangdong, China with a Different Cell Tropism
 Whole Genomic Sequence and Replication Kinetics of a New Enterovirus C96 Isolated from Guangdong, China with a Different Cell Tropism Jing Lu 1,2, Huanying Zheng 1 *, Yong Zhang 3, Xue Guo 1,DeWu 1, Hui
Whole Genomic Sequence and Replication Kinetics of a New Enterovirus C96 Isolated from Guangdong, China with a Different Cell Tropism Jing Lu 1,2, Huanying Zheng 1 *, Yong Zhang 3, Xue Guo 1,DeWu 1, Hui
Exploring the evolution of MRSA with Whole Genome Sequencing
 Exploring the evolution of MRSA with Whole Genome Sequencing PhD student: Zheng WANG Supervisor: Professor Margaret IP Department of Microbiology, CUHK Joint Graduate Seminar Department of Microbiology,
Exploring the evolution of MRSA with Whole Genome Sequencing PhD student: Zheng WANG Supervisor: Professor Margaret IP Department of Microbiology, CUHK Joint Graduate Seminar Department of Microbiology,
Patterns of hemagglutinin evolution and the epidemiology of influenza
 2 8 US Annual Mortality Rate All causes Infectious Disease Patterns of hemagglutinin evolution and the epidemiology of influenza DIMACS Working Group on Genetics and Evolution of Pathogens, 25 Nov 3 Deaths
2 8 US Annual Mortality Rate All causes Infectious Disease Patterns of hemagglutinin evolution and the epidemiology of influenza DIMACS Working Group on Genetics and Evolution of Pathogens, 25 Nov 3 Deaths
It is well known that some pathogenic microbes undergo
 Colloquium Effects of passage history and sampling bias on phylogenetic reconstruction of human influenza A evolution Robin M. Bush, Catherine B. Smith, Nancy J. Cox, and Walter M. Fitch Department of
Colloquium Effects of passage history and sampling bias on phylogenetic reconstruction of human influenza A evolution Robin M. Bush, Catherine B. Smith, Nancy J. Cox, and Walter M. Fitch Department of
Replication Defective Enterovirus Infections: Implications for Type I Diabetes
 Replication Defective Enterovirus Infections: Implications for Type I Diabetes N. M. Chapman Department of Pathology & Microbiology University of Nebraska Medical Center Enterovirus Genome and 2 Capsid
Replication Defective Enterovirus Infections: Implications for Type I Diabetes N. M. Chapman Department of Pathology & Microbiology University of Nebraska Medical Center Enterovirus Genome and 2 Capsid
TITLE: Influenza A (H7N9) virus evolution: Which genetic mutations are antigenically important?
 TITLE: Influenza A (H7N9) virus evolution: Which genetic mutations are antigenically important? AUTHORS: Joshua G. Petrie 1, Adam S. Lauring 2,3 AFFILIATIONS: 1 Department of Epidemiology, University of
TITLE: Influenza A (H7N9) virus evolution: Which genetic mutations are antigenically important? AUTHORS: Joshua G. Petrie 1, Adam S. Lauring 2,3 AFFILIATIONS: 1 Department of Epidemiology, University of
Sabin vaccine reversion in the field: a comprehensive analysis of Sabin-like poliovirus
 JVI Accepted Manuscript Posted Online 14 October 2015 J. Virol. doi:10.1128/jvi.01532-15 Copyright 2015, American Society for Microbiology. All Rights Reserved. 1 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16
JVI Accepted Manuscript Posted Online 14 October 2015 J. Virol. doi:10.1128/jvi.01532-15 Copyright 2015, American Society for Microbiology. All Rights Reserved. 1 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16
BEAST Bayesian Evolutionary Analysis Sampling Trees
 BEAST Bayesian Evolutionary Analysis Sampling Trees Introduction Revealing the evolutionary dynamics of influenza This tutorial provides a step-by-step explanation on how to reconstruct the evolutionary
BEAST Bayesian Evolutionary Analysis Sampling Trees Introduction Revealing the evolutionary dynamics of influenza This tutorial provides a step-by-step explanation on how to reconstruct the evolutionary
Recent outbreaks of chikungunya in Sri Lanka and the role of Asian Tigers
 Recent outbreaks of chikungunya in Sri Lanka and the role of Asian Tigers Introduction CHIK Virus Classification: An ARBOVIRUS Family - Togaviridae Genus Alphavirus ** Enveloped, positive- strand RNA virus.
Recent outbreaks of chikungunya in Sri Lanka and the role of Asian Tigers Introduction CHIK Virus Classification: An ARBOVIRUS Family - Togaviridae Genus Alphavirus ** Enveloped, positive- strand RNA virus.
Multi-clonal origin of macrolide-resistant Mycoplasma pneumoniae isolates. determined by multiple-locus variable-number tandem-repeat analysis
 JCM Accepts, published online ahead of print on 30 May 2012 J. Clin. Microbiol. doi:10.1128/jcm.00678-12 Copyright 2012, American Society for Microbiology. All Rights Reserved. 1 2 Multi-clonal origin
JCM Accepts, published online ahead of print on 30 May 2012 J. Clin. Microbiol. doi:10.1128/jcm.00678-12 Copyright 2012, American Society for Microbiology. All Rights Reserved. 1 2 Multi-clonal origin
Chronic HIV-1 Infection Frequently Fails to Protect against Superinfection
 Chronic HIV-1 Infection Frequently Fails to Protect against Superinfection Anne Piantadosi 1,2[, Bhavna Chohan 1,2[, Vrasha Chohan 3, R. Scott McClelland 3,4,5, Julie Overbaugh 1,2* 1 Division of Human
Chronic HIV-1 Infection Frequently Fails to Protect against Superinfection Anne Piantadosi 1,2[, Bhavna Chohan 1,2[, Vrasha Chohan 3, R. Scott McClelland 3,4,5, Julie Overbaugh 1,2* 1 Division of Human
Molecular Evolution of the SARS Coronavirus During the Course of the SARS Epidemic in China
 Molecular Evolution of the SARS Coronavirus During the Course of the SARS Epidemic in China The Chinese SARS Molecular Epidemiology Consortium* *The complete list of authors and their affiliated institutions
Molecular Evolution of the SARS Coronavirus During the Course of the SARS Epidemic in China The Chinese SARS Molecular Epidemiology Consortium* *The complete list of authors and their affiliated institutions
Outbreak of febrile illness caused by coxsackievirus A4 in a nursery school in Beijing, China
 Li et al. Virology Journal (2015) 12:92 DOI 10.1186/s12985-015-0325-1 SHORT REPORT Open Access Outbreak of febrile illness caused by coxsackievirus A4 in a nursery school in Beijing, China Jin-Song Li
Li et al. Virology Journal (2015) 12:92 DOI 10.1186/s12985-015-0325-1 SHORT REPORT Open Access Outbreak of febrile illness caused by coxsackievirus A4 in a nursery school in Beijing, China Jin-Song Li
Mutations in the non-structural protein region contribute to intra-genotypic evolution of enterovirus 71
 Huang et al. Journal of Biomedical Science 2014, 21:33 RESEARCH Open Access Mutations in the non-structural protein region contribute to intra-genotypic evolution of enterovirus 71 Sheng-Wen Huang 1, Hui-Li
Huang et al. Journal of Biomedical Science 2014, 21:33 RESEARCH Open Access Mutations in the non-structural protein region contribute to intra-genotypic evolution of enterovirus 71 Sheng-Wen Huang 1, Hui-Li
Molecular Epidemiology of the Human Rhinovirus Infection in Mongolia during
 Jpn. J. Infect. Dis., 68, 280 287, 2015 Original Article Molecular Epidemiology of the Human Rhinovirus Infection in Mongolia during 2008 2013 Sosorbaram Tsatsral 1, Zichun Xiang 4,NaokoFuji 5, Chulunbatar
Jpn. J. Infect. Dis., 68, 280 287, 2015 Original Article Molecular Epidemiology of the Human Rhinovirus Infection in Mongolia during 2008 2013 Sosorbaram Tsatsral 1, Zichun Xiang 4,NaokoFuji 5, Chulunbatar
Genomic characterization of two new enterovirus types, EV-A114 and EV-A121
 Journal of Medical Microbiology (2016), 65, 1465 1471 DOI 10.10/jmm.0.000380 Genomic characterization of two new enterovirus types, EV-A114 and EV-A121 Jagadish M. Deshpande, Deepa K. Sharma, Vinay K.
Journal of Medical Microbiology (2016), 65, 1465 1471 DOI 10.10/jmm.0.000380 Genomic characterization of two new enterovirus types, EV-A114 and EV-A121 Jagadish M. Deshpande, Deepa K. Sharma, Vinay K.
SURVEILLANCE TECHNICAL
 CHAPTER 5 SURVEILLANCE TECHNICAL ASPECTS 55 Protect - detect - protect Polio eradication strategies can be summed up as protect and detect protect children against polio by vaccinating them, and detect
CHAPTER 5 SURVEILLANCE TECHNICAL ASPECTS 55 Protect - detect - protect Polio eradication strategies can be summed up as protect and detect protect children against polio by vaccinating them, and detect
Hand Foot and Mouth Disease Due to Enterovirus 71 in Malaysia
 VIROLOGICA SINICA, August 2011, 26 (4): 221-228 DOI 10.1007/s12250-011-3195-8 Wuhan Institute of Virology, CAS and Springer-Verlag Berlin Heidelberg 2011 Hand Foot and Mouth Disease Due to Enterovirus
VIROLOGICA SINICA, August 2011, 26 (4): 221-228 DOI 10.1007/s12250-011-3195-8 Wuhan Institute of Virology, CAS and Springer-Verlag Berlin Heidelberg 2011 Hand Foot and Mouth Disease Due to Enterovirus
Existence of reassortant A (H1N2) swine influenza viruses in Saitama Prefecture, Japan
 International Congress Series 1263 (2004) 749 753 Existence of reassortant A (H1N2) swine influenza viruses in Saitama Prefecture, Japan Shin ichi Shimada a, *, Takayasu Ohtsuka b, Masayuki Tanaka b, Munehito
International Congress Series 1263 (2004) 749 753 Existence of reassortant A (H1N2) swine influenza viruses in Saitama Prefecture, Japan Shin ichi Shimada a, *, Takayasu Ohtsuka b, Masayuki Tanaka b, Munehito
Genetic Changes of Coxsackievirus A16 and Enterovirus 71 Isolated from Hand, Foot, and Mouth Disease Patients in Toyama, Japan between 1981 and 2007
 Jpn. J. Infect. Dis., 62, 254-259, 2009 Original Article Genetic Changes of Coxsackievirus A16 and Enterovirus 71 Isolated from Hand, Foot, and Mouth Disease Patients in Toyama, Japan between 1981 and
Jpn. J. Infect. Dis., 62, 254-259, 2009 Original Article Genetic Changes of Coxsackievirus A16 and Enterovirus 71 Isolated from Hand, Foot, and Mouth Disease Patients in Toyama, Japan between 1981 and
The Genealogical Population Dynamics of HIV-1 in a Large Transmission Chain: Bridging within and among Host Evolutionary Rates
 The Genealogical Population Dynamics of HIV-1 in a Large Transmission Chain: Bridging within and among Host Evolutionary Rates Bram Vrancken 1 *, Andrew Rambaut 2,3, Marc A. Suchard 4,5,6, Alexei Drummond
The Genealogical Population Dynamics of HIV-1 in a Large Transmission Chain: Bridging within and among Host Evolutionary Rates Bram Vrancken 1 *, Andrew Rambaut 2,3, Marc A. Suchard 4,5,6, Alexei Drummond
Summary. Primary care data. Week 49/2014. Season
 Summary Week 49/2014 In week 49/2014, influenza activity remained low across the WHO European Region. Twenty countries reported sporadic influenza activity and nine reported increasing trends in consultations
Summary Week 49/2014 In week 49/2014, influenza activity remained low across the WHO European Region. Twenty countries reported sporadic influenza activity and nine reported increasing trends in consultations
