Payments For Vaccines Guidance.
|
|
|
- Myrtle Bell
- 6 years ago
- Views:
Transcription
1 FINAL VERSION Payments For Vaccines Guidance. GP Practices in Blackburn with Darwen, Blackpool, Central Lancashire, Cumbria, East Lancashire and North Lancashire Primary Care Trusts Prepared by the Cumbria and Lancashire PCTs Vaccine Patient Group Direction Sub-group June
2 Patient Group Direction Sub-group Members: Zena Baxter Blackpool PCT Jackie Bolton - North Lancashire Teaching PCT Kate Brierley Health Protection Agency (Chair) Kath Darroch - North Lancashire Teaching PCT Marion Fleming Blackpool PCT Melanie Greenall - North Lancashire Teaching PCT Catherine Harding East Lancashire PCT Lindsay Holden Blackburn with Darwen PCT Andrea Loudon Cumbria PCT Diane McGinn North Lancashire Teaching PCT Jane Morphet Cumbria PCT Susan Perry NHS Central Lancashire Melanie Preston Blackpool PCT Nicola Schaffel - NHS Central Lancashire Lisa Vallente-Osborne- Blackpool PCT Fiona Ward University Hospitals Morecambe Bay Acknowledgement: This document is based on the original document produced by Kent Local Medical Committee April 2008 version 2.6 This document may be copied freely so long as the original authorship is recognised and the Kent LMC does not take any responsibility for any reliance on the document outside of Kent. Queries: If there are any queries relating to this document please contact your local Immunisation & Vaccination Co-ordinator or Kate Brierley, Chair of the Group. kate.brierley@hpa.org.uk 2
3 Glossary PPA DES LES NES DH FP34D FP10 LMC PCT GMS HPA Prescription Pricing Authority Directly Enhanced Service Locally Enhanced Service Nationally Enhanced Service Department of Health Claims for vaccines purchased and personally administered Prescription dispensed by community pharmacy Local Medical Committee Primary Care Trust General Medical Service Health Protection Agency 3
4 Introduction How to use the booklet The vaccinations and immunisations listed in this Guide are divided into 3 sections with respect to payments: 1. vaccinations and immunisations that are now part of the Global sum and where NO charges may be made to patients. The vaccine can be obtained in bulk by the practice and charge to the Prescription Pricing Authority (PPA) on FP10 or FP34D for Flu, typhoid, Hepatitis A, Hepatitis B, Pneumococcal, Meningococcal or combination of these vaccines e.g. Twinrix. Some childhood vaccines are bought centrally by the Department of Health (DH) and provided free of charge by a DH contracted storage and distribution company 2. Enhanced service vaccinations and immunisations where the item of service has been transferred to the NES or where a LES is in place and has been agreed between the local Primary Care Trust (PCT) and the Local Medical Committee (LMC). 3. Private service where the practice may charge the patient directly for the service. But Please Note: a. You CANNOT charge for advice b. You CANNOT charge if the service is available on the NHS c. You CANNOT mix NHS and non NHS d. The level of charges is for the practice to determine. It is advisable to develop a practice protocol available to patients in the form of a leaflet or section of the practice leaflet. e. You cannot provide occupational health services to your own patients and charge either the patient or the employer. You must refer patients to another practice. This is particularly relevant to Hepatitis B vaccination. f. Practices may charge the patient directly for immunisations for travel where noted in these guidelines but are not compelled to do so. In some cases for example with Hepatitis B or meningitis the practice has the option to prescribe the vaccine on an FP10 or claim it back through reimbursement. This is a matter for the practice to decide. Practitioners should also refer to the current edition of the Green Book and the National Travel Health Network and Centre (NaTHNaC) for advice on appropriate immunisation and the BNF for detailed advice for each preparation. Details of the at risk groups have been mainly taken from the headings in the Green Book and are not a full explanation. If in doubt please consult the Green Book for full details. Please check with your PCT that either the DES or the LES has been approved before claiming. 4
5 Links to useful sources of information NHS Immunisation Information or The National Travel Health Network and Centre (NaTHNaC) is funded by the Department of Health to promote clinical standards in travel medicine. NaTHNaC advice line for health professionals: Telephone: The Green Book Immunisation against Infectious Disease (2006): On line version with all current updates NTENT_ID= &chk=isTfGX Health Advice for travellers Health Information for Overseas Travel (Yellow Book) Clinical Knowledge Summaries Prescription Pricing Authority For a copy of FP34PD and FP34D Appendix NB High volume drugs are: Influenza, Typhoid, Hepatitis A, Hepatitis B, Pneumococcal, Meningococcal or combinations of the vaccines listed e.g. Hepatitis A and B (Twinrix) and Hepatitis A and Typhoid (Hepatyrix). 5
6 Childhood vaccinations and immunisations Vaccinations and Immunisations that are part of full childhood schedule are all part of the and are not covered in detail in this Guide. The outline of the schedule is as follows Age What is given Vaccine 2 months Diphtheria, tetanus, pertussis, polio and haemophilus influenzae type b (DTaP/IPV/Hib) Pediacel Pneumococcal (PCV) Prevenar13 3 months Diphtheria, tetanus, pertussis, polio and haemophilus influenzae type b (DTaP/IPV/Hib) Meningitis C (Men C) 4 months Diphtheria, tetanus, pertussis, polio and haemophilus influenzae type b (DTaP/IPV/Hib) Pneumococcal (PCV) Meningitis C (Men C) months Haemophilus influenzae type b, meningitis C (Hib/Men C) Measles, mumps and rubella (MMR) Pneumococcal (PCV) Pediacel Neisvac C or Meningitec or Menjugate Kit Pediacel Prevenar13 Neisvac C or Meningitec or Menjugate Kit Menitorix Priorix or MMRvaxPro Prevenar13 3 years 4 months to 5 years Diphtheria, tetanus, pertussis and polio (dtap/ipv or DTaP/IPV) Measles, Mumps and Rubella (MMR) Repevax or Infanrix-IPV Priorix or MMRvaxPro 13 to 18 years Tetanus, diphtheria and polio (Td/IPV) Human Papilloma Virus vaccine Revaxis Cervarix 6
7 Contents and vaccine index Glossary...3 Links to useful sources of information...5 Childhood vaccinations and immunisations...6 Contents and vaccine index 7 BCG vaccine...8 Cholera vaccine...9 Low dose Diphtheria/tetanus/polio Haemophilus influenza type b (Hib) vaccine Hepatitis A vaccination Hepatitis B vaccination Hepatitis A and B combined vaccine Hepatitis A & typhoid combined vaccine Influenza vaccine Japanese B encephalitis vaccine Measles, Mumps & Rubella (MMR) combined vaccine Meningococcal A,C,W135 & Y vaccine Meningococcal C vaccine (Hib/MenC combined vaccine) Pneumococcal polysaccharide vaccine (PPV) Polio vaccine (as part of Td/IPV) Rabies vaccine Tetanus vaccine (as part of Td/IPV) Tick-borne encephalitis vaccine Typhoid polysaccharide vaccine Varicella (chickenpox) vaccine
8 BCG vaccine Where a BCG vaccination is requested in connection with travel abroad, and the patient has not been vaccinated as part of the childhood immunisation schedule then refer the patient to your local Specialist TB Service. 8
9 Cholera vaccine a) Aid workers assisting in disaster relief of refugee camps b) More adventurous backpackers travelling to remote regions with limited access to medical care c) In addition the vaccine may be considered for at risk travellers with underlying gastrointestinal illness or immune suppression in whom cholera would have serious adverse consequences FP10 Private Service a) Travellers requesting vaccination as a personal preference Issue a private prescription or charge patients from stock and charge for the administration of vaccine 9
10 Low dose Diphtheria/tetanus/polio a) Children over 10 years and adults requiring either a primary course or a booster should be given a vaccine containing low dose diphtheria b) Children aged 6 and over that have had the basic course of immunisation but not a reinforcing dose. c) Staff in hospital considered being at risk of infection of Diphtheria. d) Unimmunised travellers (over 10 years) require a full course of three doses at monthly intervals. e) Previously immunised travellers requiring a booster dose if they are to live or work with local residents and their primary immunisation was more than 10 years ago. For categories a), and b) - the vaccine is centrally purchased and distributed free by DH as part of the childhood schedule. For categories c), d) and e) the vaccines is purchased directly from the manufacturers with an FP10 issue to cover the costs. 10
11 Haemophilus influenza type b (Hib) vaccine a) Asplenic children and adults, irrespective of age or the interval from splenectomy, should receive a single dose of Hib vaccine if not already given. - the vaccine is centrally purchased by DH and distributed free by Movianto UK Ltd for children and adults. There is no single Hib vaccine available, therefore Hib/MenC (Menitorix) should be used. 11
12 Hepatitis A vaccination a) Patients with chronic liver disease b) Haemophiliacs c) Men who have sex with men d) Persons in institution who are exposed to a high risk of infection and for whom vaccination is recommended e) Persons (particularly those going to reside for 3 months or longer or who, if infected, might be less resistant because of pre-existing disease) travelling outside Northern Europe, Australia or New Zealand to areas e.g. of poor sanitation, where the degree of exposure to infections is likely to be high f) Injecting drug users FP34D Locally Enhanced Service a) Vaccine administered as part of outbreak control on the advice of Health Protection Agency/Consultant in Communicable Disease Control FP34D Private Service a) Occupational Exposure (refer to employer to undertake or to another practice) b) Travellers to areas that do not qualify for GMS Issue a private prescription or charge patients from your stock and charge for the administration of vaccine 12
13 Hepatitis B vaccination a) Babies born to mothers who are chronic carriers of hepatitis B virus or to mothers who have had acute hepatitis b during pregnancy b) Parental drug misusers c) Individuals who change sexual partners frequently d) Close family/household/sexual contacts of a case or carrier e) Families adopting children from countries with a high or intermediate prevalence of hepatitis B f) Foster carers g) Individuals receiving regular blood or blood products and their carers h) Patients with chronic liver disease i) Patients with chronic renal failure j) Children born outside the UK and who have received a primary dose in their country of origin and who are now domiciled in the UK should have their course of the vaccine completed under GMS k) Travel see comments below l) Staff and residents in accommodation for those with learning difficulties m) Inmates of custodial institutions FP34D Local Enhanced Service a) Post-exposure prophylaxis with hepatitis B immunoglobulin (HBIG) Private Service a) Occupational Health. Advise the employer to carry out a risk assessment. If hepatitis B is required, inform the employer that this service is not covered by GMS and the employer will need to make private arrangements with and alternative provider. 13
14 If requested by the employer to carry out this private service for a patient not on your NHS list, bill employer for payment for administration and cost of drug (+ VAT) + on cost and dispensing and administration fee. As part of the service blood test for hepatitis B antibodies are indicated. Bill the employer for the cost of phlebotomy and the test. Practices should determine an appropriate fee taking into account the cost of the drugs, blood tests and the work involved. b) Travel. If the vaccination is requested in connection with travel abroad, and the patient does not fall into a risk group for a GMS service then a private service is an option. Refer to the Green Book recommendations. Issue a private prescription or charge patients from your stock and charge for the administration of vaccine. MAKE THE PATIENT AWARE OF THE NEED FOR 3 INJECTIONS OF HEPATITIS B VACCINE, AND IF GIVEN FOR OCCUPATIONAL HEALTH REASONS, THE BLOOD TEST TO CONFIRM IMMUNITY 14
15 Hepatitis A and B combined vaccine a) There are circumstances where the combined A & B will be required and there is the option to use the combined vaccine which offers a reduced number of injections and may help compliance. Hepatitis A combined with Typhoid is also a useful combination where the patient is travelling to a risk area. Where this latter combination is helpful and hepatitis B is also indicated it is possible to administer this separately. b) For travel, combined vaccine should be used for the group of travellers who it is felt are at risk of hepatitis A and B. It can be used for those who present up to 21 days before travel if the rapid schedule is used. The potential benefit to these travellers is the reduction in the total number of injections from five to three. The cost of the combined vaccine primary course is the same as a primary course using the separate vaccines in pre-filled syringes. The primary course of the combined vaccine should give protection against hepatitis A for ten years and hepatitis B for five years, the same as for the vaccines given separately. It is important to remember to use hepatitis B alone as a booster if required after 5 years. c) Children under 16. Where combined hepatitis A and B are indicated this may be given in the paediatric two dose combined vaccine (Ambirix) which reduces the number of injections still further from 5 to just two. These are given 6 months apart and so this is unsuitable for rapid immunisation. Any booster can be given with single dose hepatitis B. FP34D or issue an FP10 Private Service Where not covered by GMS Notes Persons requiring only hepatitis A in connection with travel abroad (see page 10 for entitlement to NHS service) should receive hepatitis A vaccine (Havrix, Avaxim ). 15
16 Persons requesting only hepatitis B vaccination in connection with travel abroad should receive hepatitis B vaccine. A charge may be made to the patient for administering the vaccine unless the patient falls into the risk group as set out under global sum on page 11. Patients requiring combined hepatitis A & B cannot be charged a private fee. The vaccine should be claimed on FP34D or issue an FP10. 16
17 Hepatitis A & typhoid combined vaccine a) Persons (particularly those going to reside for 3 months or longer or who, if infected, might be less resistant because of pre-existing disease) travelling outside Northern Europe, Australia or New Zealand to areas e.g. of poor sanitation, where the degree of exposure to infections is likely to be high. The above requirement is the same for both hepatitis A and typhoid. The combined vaccine is presented as a convenience to travellers, in that hepatitis A and typhoid vaccinations can be administered with one injection. Primary course of the combined vaccine should give protection against hepatitis A for up to 1 year, and typhoid for 3 years. This is the same as for the vaccines given separately. This may lead to confusion because at 6 months to 1 year patients will need a booster dose of hepatitis A not the combined vaccine booster. After boosting, patient s protection for Hepatitis A is extended to 20 years, but the typhoid component only gives protection for 3 years. FP34D Private Service a) Where the patient requests vaccination but does not qualify for a GMS service. Issue a private prescription or charge patients from stock 17
18 Influenza vaccine a) Where the doctor considers the vaccine is indicated but the patient does not qualify for the enhanced service, no fee can be charged from the patient FP34D Enhanced Service a) Where the patient qualifies under the terms of the DES FP34D Private Sector a) Where the doctor does not consider the vaccine is clinically indicated it should not be given b) Where the doctor considers the vaccine is indicated but the patient does not qualify for the enhanced service the patient should be referred to another practice for a private service c) Patients who are not registered with the practice Issue a private prescription or charge patients from stock and charge for the administration of vaccine 18
19 Japanese B encephalitis vaccine Private Service a) In connection with travel abroad. Japanese encephalitis vaccine is recommended for those going to reside in areas where Japanese encephalitis is endemic or epidemic. All travellers should undergo a risk assessment considering their itinerary, season of travel, duration of stay and planned activities. The risk of Japanese encephalitis should then be balanced against their risk of adverse events from vaccination. Issue a private prescription or charge patients for the administration of vaccine 19
20 Measles, Mumps & Rubella (MMR) combined vaccine Enhanced Service a) Individuals over the age of 15 years if not previously been immunised with an MMR combined vaccine b) Young adults with outstanding MMR vaccinations- offered opportunistically. c) Sero-negative women of child bearing age d) Any patient presenting with incomplete or unknown MMR vaccine history. The vaccine is centrally purchased and distributed free by DH. For occupational health purposes the vaccine should be sourced privately either direct from the supplier or via private prescription. For people moving abroad or travel purposes the vaccine can be sourced privately and charged for. However GPs can choose not to charge and claim the cost of the vaccine from the PCT 20
21 Meningococcal A,C,W135 & Y vaccine a) Asplenic children and adults irrespective of age or the interval from splenectomy should receive a single dose of vaccine before travelling to area where there is increased risk of Group A infection under GMS or via FP34D The vaccine is centrally purchased by DH and distributed free by Movianto UK for children Enhanced Service a) Meningococcal vaccination of contacts of cases Where a case is confirmed as infected with a Group A or C or W135 strain, the Health Protection Agency will advise the doctor to offer vaccination as additional protection to those who have already received prophylaxis with rifampicin/ciprofloxacin The vaccine is available free of charge from the HPA Private Service a) If the vaccination is requested in connection with travel abroad, there is no item of service fee payable under the SFA. You may wish to offer the service under GMS and claim reimbursement of the vaccine or you may wish to charge the patient for a private service. Issue a private prescription or charge patients from stock and charge for the administration of vaccine 21
22 Meningococcal C vaccine a) Children and adults under 25 years who have not completed their primary course The vaccine is centrally purchased and distributed free by DH for children and adults Enhanced Service a) Adults (25 yrs and over) entering full-time education at university or college of higher education FP34D 22
23 Pneumococcal polysaccharide vaccine (PPV) a) Pneumococcal vaccine is recommended for adults over 65 years, and all those aged 2 years or older in whom pneumococcal infection is likely to be more common and/or dangerous i.e. those with: a. Asplenia or dysfunction of the spleen including homogenous sickle cell disease and coeliac syndrome b. Chronic respiratory disease c. Chronic heart disease d. Chronic kidney disease e. Chronic liver disease f. Diabetes mellitus g. Immunodeficiency or immunosuppression due to disease or treatment, including HIV at all stages h. Cochlear implants i. Cerebrospinal fluid leaks FP34D 23
24 Polio vaccine (IPV) a) Infants and children under ten years of age who have not completed their primary course b) Children aged ten years or over and adults who have not completed their full primary course and/or boosters. It is recommended that the Td/IPV is given where a booster of any element is required c) For travellers (children aged ten years or over and adults) it is recommended that the combined Td/IPV is given where a booster of any element is required For categories a) and b) the vaccines is provided free of charge from DH For category c) the vaccine is purchased directly from the manufacturer with an FP10 issue to cover the costs 24
25 Rabies vaccine These occupational groups are in the global sum because an item of service fee was payable under the SFA a) Vaccination of at risk groups e.g. a. laboratory workers handling the virus b. those who, in the course of their work, regularly handle imported animals, for example: at animal quarantine centres at zoos at research and acclimatisation centres where primates and other imported animals are housed at ports, e.g. certain HM Revenue and Customs officers at the premises of carrying agents authorised to carry imported animals c. veterinary and technical staff in the State Veterinary Service; the Department for Environment, Food and Rural Affairs; the Scottish Executive Environment and Rural Affairs Department; the Welsh Assembly Government Environment, Planning and Countryside Department; and the Northern Ireland Department of Agriculture and Rural Development d. inspectors appointed by local authorities under the Animal Health Act (2002). This only includes those local authority dog wardens who are also inspectors. Other dog wardens have a low risk of exposure, and post-exposure prophylaxis in the event of an incident is appropriate e. people who regularly handle bats in the UK f. those working abroad (e.g. veterinary staff or zoologists) who by the nature of their work are at risk of contact with rabid animals g. health workers who are about to be at risk of direct exposure to body fluids or other tissue from a patient with probable or confirmed rabies. For the occupational groups listed above vaccine is supplied free of charge by DH and distributed by the Health Protection Agency. Enhanced Service a) Post exposure treatment including travellers 25
26 For post-exposure use and the occupational groups listed above vaccine is supplied free of charge by DH and distributed by the Health Protection Agency. Private Service Travellers seeking vaccination Pre-exposure immunisation is also recommended for some travellers, including: a) those living in or travelling for more than one month to rabies-enzootic areas, unless there is reliable access to prompt, safe medical care b) those travelling for less than one month to enzootic areas but who may be exposed to rabies due to their travel activities, or those who may have limited access to post-exposure medical care. Issue a private prescription or charge patients from stock and charge for the administration of vaccine. 26
27 Tetanus vaccine a) Infants and children under ten years of age who have not completed their primary course. b) Children aged ten years and over, and adults who have not completed their primary course and/or boosters. c) Travellers requiring vaccination or booster For categories a) and b) the vaccine is centrally purchased and distributed free by DH as part of the childhood schedule For category c) the vaccine is purchased directly from the manufacturers with an FP10 issued to cover the costs. 27
28 Tick-borne encephalitis vaccine Private Service a) Travellers or those going to live/work in endemic forested areas, during spring and summer months particularly those involved in hiking, camping, hunting, forestry, wood cutting, farming and the military. Issue a private prescription or charge patients from stock and charge for the administration of vaccine 28
29 Typhoid polysaccharide vaccine a) Travel to countries where typhoid is endemic (e.g. South Asia, parts of South East Asia, Middle East or Central and South America) especially if staying with or visiting the local population, or will have frequent and/or prolonged exposure to conditions where sanitation and food hygiene are likely to be poor. To cover the cost of service FP34D Private Service a) If the patient is travelling to an area that does not qualify for an item of service Issue a private prescription or charge patients from your stock and charge for the administration of vaccine 29
30 Varicella (chickenpox) vaccine a) Public sector front line Health Care Workers should be covered by an appropriate Occupational Health Service. Refer staff to Occupational Health Service. Private Service a) All front line Health Care Workers in the private sector For patients on your list, refer to another practice For patients not on your list, issue a private prescription or charge patients from your stock and charge for the administration of vaccine 30
31 Yellow fever vaccine Yellow fever vaccine is only supplied to and administered by designated centres Private Service a) Persons aged nine months and over travelling through or living in infected areas and those travelling outside urban areas of countries in the yellow fever endemic zone (see maps in Health Information for Overseas Travel ), This applies even if these countries have not officially reported the disease and do not require evidence of immunisation on entry. Immunisation under nine months is not recommended but may be performed if exposure to the risk of infection cannot be avoided b) Travellers requiring an International Certificate of Vaccination for entry to a country Issue a private prescription or charge patients from your stock and charge for the administration of vaccine THOSE PRACTICES WISHING TO APPLY FOR DESIGNATION SHOULD DOWNLOAD APPLICATION FORM FROM Yellow Fever Administration Office National Travel Health Network and Centre UCLH NHS Foundation Trust 5 th Floor West 250 Euston Road London NW1 2PG YELLOW FEVER VACCINE AND INTERNATIONAL CERTIFICATES ARE SUPPLIED ONLY TO SUCH CENTRES 31
CORNWALL AND ISLES OF SCILLY L O C A L M E D I C A L C O M M I T T E E GUIDANCE ON PAYMENTS FOR VACCINES
 CORNWALL AND ISLES OF SCILLY L O C A L M E D I C A L C O M M I T T E E Chairman: Dr P G Dommett John Keay House Vice-Chairman: Dr R A Harvey Tregonissey Road LMC Secretary: Rosalind Winter ST AUSTELL PL25
CORNWALL AND ISLES OF SCILLY L O C A L M E D I C A L C O M M I T T E E Chairman: Dr P G Dommett John Keay House Vice-Chairman: Dr R A Harvey Tregonissey Road LMC Secretary: Rosalind Winter ST AUSTELL PL25
2017/18 Immunisation programmes list of additional and enhanced services
 2017/18 Immunisation programmes list of additional and enhanced services 2017/18 Vaccination and Immunisation list of additional and enhanced services Version number: 1 First published: April 2017 Prepared
2017/18 Immunisation programmes list of additional and enhanced services 2017/18 Vaccination and Immunisation list of additional and enhanced services Version number: 1 First published: April 2017 Prepared
2018/19 Immunisation programmes list of additional and enhanced services
 2018/19 Immunisation programmes list of additional and enhanced services 2018/19 Vaccination and Immunisation list of additional and enhanced services Version number: 1 First published: April 2018 Prepared
2018/19 Immunisation programmes list of additional and enhanced services 2018/19 Vaccination and Immunisation list of additional and enhanced services Version number: 1 First published: April 2018 Prepared
2016/17 Vaccination and Immunisation list of additional services and enhanced services
 2016/17 Vaccination and Immunisation list of additional services and enhanced services 2016/17 Vaccination and Immunisation list of additional services and enhanced services Version number: 1 First published:
2016/17 Vaccination and Immunisation list of additional services and enhanced services 2016/17 Vaccination and Immunisation list of additional services and enhanced services Version number: 1 First published:
GUIDANCE ON PAYMENTS FOR VACCINATIONS AND IMMUNISATIONS
 GUIDANCE ON PAYMENTS FOR VACCINATIONS AND IMMUNISATIONS Version: Ratified by: Date ratified: Date issued: Expiry date: (Document is not valid after this date) Review date: Lead Executive/Director: Name
GUIDANCE ON PAYMENTS FOR VACCINATIONS AND IMMUNISATIONS Version: Ratified by: Date ratified: Date issued: Expiry date: (Document is not valid after this date) Review date: Lead Executive/Director: Name
UNSCHEDULED VACCINATION OF CHILDREN AND YOUNG PEOPLE WHO HAVE OUTSTANDING ROUTINE IMMUNISATIONS. Service Specification
 UNSCHEDULED VACCINATION OF CHILDREN AND YOUNG PEOPLE WHO HAVE OUTSTANDING ROUTINE IMMUNISATIONS Service Specification National Enhanced Service Specification For The Unscheduled Vaccination Of Children
UNSCHEDULED VACCINATION OF CHILDREN AND YOUNG PEOPLE WHO HAVE OUTSTANDING ROUTINE IMMUNISATIONS Service Specification National Enhanced Service Specification For The Unscheduled Vaccination Of Children
GUIDANCE ON PRESCRIBING VACCINATIONS ON THE NHS & PRIVATELY (INCLUDING TRAVEL VACCINATIONS)
 GUIDANCE ON PRESCRIBING VACCINATIONS ON THE NHS & PRIVATELY (INCLUDING TRAVEL VACCINATIONS) Under the new GMS contract (from 2004), vaccinations and immunisations are paid for through various mechanisms
GUIDANCE ON PRESCRIBING VACCINATIONS ON THE NHS & PRIVATELY (INCLUDING TRAVEL VACCINATIONS) Under the new GMS contract (from 2004), vaccinations and immunisations are paid for through various mechanisms
Family and Travel Vaccinations
 Family and Travel Vaccinations We offer the full range of baby, child and family vaccinations. We are able to tailor schedules to your child s needs or international schedule. We have a suggested vaccination
Family and Travel Vaccinations We offer the full range of baby, child and family vaccinations. We are able to tailor schedules to your child s needs or international schedule. We have a suggested vaccination
Introduction. Infections acquired by travellers
 Introduction The number of Australians who travel overseas has increased steadily over recent years and now between 3.5 and 4.5 million exits are made annually. Although many of these trips are to countries
Introduction The number of Australians who travel overseas has increased steadily over recent years and now between 3.5 and 4.5 million exits are made annually. Although many of these trips are to countries
Childhood Immunisations Template Guide 2016
 Childhood Immunisations Template Guide 2016 1 Template Control Page Childhood Immunisations Template Guide Title Childhood Immunisations Author CEG Version 9 Description Supports the schedule of Childhood
Childhood Immunisations Template Guide 2016 1 Template Control Page Childhood Immunisations Template Guide Title Childhood Immunisations Author CEG Version 9 Description Supports the schedule of Childhood
THE KEATS GROUP PRACTICE REGISTRATION FORM PLEASE COMPLETE IN BLOCK CAPITALS PERSONAL BACKGROUND INFORMATION
 Registration checked by: Date: 11 & Under THE KEATS GROUP PRACTICE REGISTRATION FORM PLEASE COMPLETE IN BLOCK CAPITALS PERSONAL BACKGROUND INFORMATION Male Female Has your child ever been treated at this
Registration checked by: Date: 11 & Under THE KEATS GROUP PRACTICE REGISTRATION FORM PLEASE COMPLETE IN BLOCK CAPITALS PERSONAL BACKGROUND INFORMATION Male Female Has your child ever been treated at this
14: Immunological products and vaccines
 File name: Immunological Products and s Formulary BNF Original Date of issue: 30/10/2005 Last Reviewed: 23/11/2012 Version:4 Page 1 of 6 14: Immunological products and vaccines 14.1 Active immunity Live
File name: Immunological Products and s Formulary BNF Original Date of issue: 30/10/2005 Last Reviewed: 23/11/2012 Version:4 Page 1 of 6 14: Immunological products and vaccines 14.1 Active immunity Live
Immunization Guidelines for the Use of State Supplied Vaccine April 18, 2013
 DTaP / DT DTaP/IPV/Hep B Combination (Pediarix ) Children from 6 weeks of age up to the 7 th birthday Children from 2 months of age up to the 7th birthday: Indicated for the primary doses of DTaP, IPV,
DTaP / DT DTaP/IPV/Hep B Combination (Pediarix ) Children from 6 weeks of age up to the 7 th birthday Children from 2 months of age up to the 7th birthday: Indicated for the primary doses of DTaP, IPV,
Childhood Immunisations Template Guide 2017
 Childhood Immunisations Template Guide 2017 1 Template Control Page Childhood Immunisations Template Guide Title Childhood Immunisations Author CEG Description Supports the schedule of Childhood Immunisations
Childhood Immunisations Template Guide 2017 1 Template Control Page Childhood Immunisations Template Guide Title Childhood Immunisations Author CEG Description Supports the schedule of Childhood Immunisations
THIS PATIENT GROUP DIRECTION HAS BEEN AGREED BY THE FOLLOWING ORGANISATIONS:
 Patient Group Direction for Pneumococcal polysaccharide vaccine VERSION: PNEUMOCOCCAL POLY-2013.1 (Pneumovax II ) Start 1 st May 2013 Expiry 30 th April 2015 THIS PATIENT GROUP DIRECTION HAS BEEN AGREED
Patient Group Direction for Pneumococcal polysaccharide vaccine VERSION: PNEUMOCOCCAL POLY-2013.1 (Pneumovax II ) Start 1 st May 2013 Expiry 30 th April 2015 THIS PATIENT GROUP DIRECTION HAS BEEN AGREED
National Immunisation News
 National Immunisation News The newsletter of the HSE National Immunisation Office July 2016 Changes to the Primary Childhood Immunisation Programme The National Immunisation Advisory Committee (NIAC) has
National Immunisation News The newsletter of the HSE National Immunisation Office July 2016 Changes to the Primary Childhood Immunisation Programme The National Immunisation Advisory Committee (NIAC) has
Immunization Guidelines for the Use of State Supplied Vaccine May 17, 2015
 DTaP / DT DTaP/IPV/Hep B Combination (Pediarix ) Children from 6 weeks of age up to the 7 th birthday Children from 2 months of age up to the 7th birthday: Indicated for the primary doses of DTaP, IPV,
DTaP / DT DTaP/IPV/Hep B Combination (Pediarix ) Children from 6 weeks of age up to the 7 th birthday Children from 2 months of age up to the 7th birthday: Indicated for the primary doses of DTaP, IPV,
What are the new active vaccine recommendations in the Canadian Immunization Guide?
 154 CCDR 17 April 2014 Volume 40-8 https://doi.org/10.14745/ccdr.v40i08a03 1 What are the new active vaccine recommendations in the Canadian Immunization Guide? Warshawsky B 1 and Gemmill I 2 on behalf
154 CCDR 17 April 2014 Volume 40-8 https://doi.org/10.14745/ccdr.v40i08a03 1 What are the new active vaccine recommendations in the Canadian Immunization Guide? Warshawsky B 1 and Gemmill I 2 on behalf
Guidelines for the immunisation of children following treatment with Standard-Dose Chemotherapy
 Guidelines for the immunisation of children following treatment with Standard-Dose Chemotherapy Version 2.0 Approved by Haem / Onc Senior Clinical Management team Date Approved March 2015 Ratified by:
Guidelines for the immunisation of children following treatment with Standard-Dose Chemotherapy Version 2.0 Approved by Haem / Onc Senior Clinical Management team Date Approved March 2015 Ratified by:
National Immunisation News
 July 2015 National Immunisation News The newsletter of the HSE National Immunisation Office CONTENTS Changes to the Primary Childhood Immunisation Programme BCG Shortage Pertussis Vaccine Mumps Order Vaccines
July 2015 National Immunisation News The newsletter of the HSE National Immunisation Office CONTENTS Changes to the Primary Childhood Immunisation Programme BCG Shortage Pertussis Vaccine Mumps Order Vaccines
Immunization Guidelines For the Use of State Supplied Vaccine July 1, 2011
 DTaP / DT DTaP/IPV/Hep B Combination (Pediarix ) Children from 6 weeks of age up to the 7 th birthday Children from 2 months of age up to the 7th birthday: Indicated for the primary doses of DTaP, IPV,
DTaP / DT DTaP/IPV/Hep B Combination (Pediarix ) Children from 6 weeks of age up to the 7 th birthday Children from 2 months of age up to the 7th birthday: Indicated for the primary doses of DTaP, IPV,
CLINICAL CONTENT OF PATIENT GROUP DIRECTION FOR PNEUMOCOCCAL CONJUGATE VACCINE (Prevenar 13 )
 Patient Group Direction for Pneumococcal Conjugate Vaccine (Prevenar 13 ) Version: PNEUMOCOCCAL CONJUGATE 2014.1 Start 1 st February 2014 Expiry 31 st January 2017 THIS PATIENT GROUP DIRECTION HAS BEEN
Patient Group Direction for Pneumococcal Conjugate Vaccine (Prevenar 13 ) Version: PNEUMOCOCCAL CONJUGATE 2014.1 Start 1 st February 2014 Expiry 31 st January 2017 THIS PATIENT GROUP DIRECTION HAS BEEN
GENERAL PRACTITIONER DATA PACK GUIDANCE
 RESTRICTED For CQC internal use only This should NOT be shared outside CQC GENERAL PRACTITIONER DATA PACK GUIDANCE PRIMARY CARE DATA PACKS AND INSPECTION TEAM - November 2015 - Table of Contents Introduction...
RESTRICTED For CQC internal use only This should NOT be shared outside CQC GENERAL PRACTITIONER DATA PACK GUIDANCE PRIMARY CARE DATA PACKS AND INSPECTION TEAM - November 2015 - Table of Contents Introduction...
Change history Version number Change details Date Updated for National ACWY programme to be implemented August June 2015.
 Patient Group Direction for Conjugate Meningococcal ACWY vaccine Version: MEN ACWY CONJUGATE 2015.1 Start 1 st August 2015 Expiry 31 st July 2018 THIS PATIENT GROUP DIRECTION HAS BEEN AGREED BY THE FOLLOWING
Patient Group Direction for Conjugate Meningococcal ACWY vaccine Version: MEN ACWY CONJUGATE 2015.1 Start 1 st August 2015 Expiry 31 st July 2018 THIS PATIENT GROUP DIRECTION HAS BEEN AGREED BY THE FOLLOWING
National Immunisation News The newsletter of the HSE National Immunisation Office
 February 2015 National Immunisation News The newsletter of the HSE National Immunisation Office CONTENTS Changes to the PCI programme Uptake statistics Pneumovax name change Flu season Common queries School
February 2015 National Immunisation News The newsletter of the HSE National Immunisation Office CONTENTS Changes to the PCI programme Uptake statistics Pneumovax name change Flu season Common queries School
abcdefghijklm abcde abc a eé~äíü=aéé~êíãéåí=
 abcdefghijklm eé~äíü=aéé~êíãéåí= Dear Colleague IMPORTANT CHANGES TO THE CHILDHOOD IMMUNISATION PROGRAMME We are writing to you with further information about the changes to the routine childhood immunisation
abcdefghijklm eé~äíü=aéé~êíãéåí= Dear Colleague IMPORTANT CHANGES TO THE CHILDHOOD IMMUNISATION PROGRAMME We are writing to you with further information about the changes to the routine childhood immunisation
Staff Immunisation Policy
 Policy No: IC05 Version: 8.0 Name of Policy: Staff Immunisation Policy Effective From: 18/08/2015 Date Ratified 15/07/2015 Ratified Infection Prevention & Control Committee Review Date 01/07/2017 Sponsor
Policy No: IC05 Version: 8.0 Name of Policy: Staff Immunisation Policy Effective From: 18/08/2015 Date Ratified 15/07/2015 Ratified Infection Prevention & Control Committee Review Date 01/07/2017 Sponsor
INFECTION PREVENTION AND CONTROL POLICY AND PROCEDURES Sussex Partnership NHS Foundation Trust (The Trust)
 A member of: Association of UK University Hospitals INFECTION PREVENTION AND CONTROL POLICY AND PROCEDURES Sussex Partnership NHS Foundation Trust (The Trust) IPC20 VACCINATION PROGRAMME FOR STAFF AND
A member of: Association of UK University Hospitals INFECTION PREVENTION AND CONTROL POLICY AND PROCEDURES Sussex Partnership NHS Foundation Trust (The Trust) IPC20 VACCINATION PROGRAMME FOR STAFF AND
Adolescent vaccination strategies
 Adolescent vaccination strategies Gregory Hussey Vaccines for Africa Initiative Institute of Infectious Diseases & Molecular Medicine University of Cape Town www.vacfa.uct.ac.za gregory.hussey@uct.ac.za
Adolescent vaccination strategies Gregory Hussey Vaccines for Africa Initiative Institute of Infectious Diseases & Molecular Medicine University of Cape Town www.vacfa.uct.ac.za gregory.hussey@uct.ac.za
Immunisation Policy. Country Children s Early Learning Ph: M:
 Immunisation Policy Published June 2018 Review Date December 2019 Sources Education and Care Services National Regulations, October 2017 Guide to the Education and Care Services National Law and the Education
Immunisation Policy Published June 2018 Review Date December 2019 Sources Education and Care Services National Regulations, October 2017 Guide to the Education and Care Services National Law and the Education
Series of 2 doses, 6-12 months apart. One dose is 720 Elu/0.5ml (GSK) or 25 u/0.5 ml (Merck)
 UTAH PREVENTIVE CARE RECOMMENDATIONS Adult - Ages 19 and Above IMMUNIZATIONS CONTENTS: General Instructions Hepatitis A Hepatitis B Human Papilloma Virus Influenza Meningococcal A, C, Y, W (MCV4) Meningococcal
UTAH PREVENTIVE CARE RECOMMENDATIONS Adult - Ages 19 and Above IMMUNIZATIONS CONTENTS: General Instructions Hepatitis A Hepatitis B Human Papilloma Virus Influenza Meningococcal A, C, Y, W (MCV4) Meningococcal
The schedule for childhood vaccination is:(web link to NHS Childhood Immunisation Schedule for 2008
 Immunisations and vaccinations Immunisation is an effective public health intervention for promoting good health and protecting individuals and populations against serious disease and infection through
Immunisations and vaccinations Immunisation is an effective public health intervention for promoting good health and protecting individuals and populations against serious disease and infection through
WELSH HEALTH CIRCULAR
 WELSH HEALTH CIRCULAR WHC (2017) 022 Issue Date: 12 May 2017 STATUS: ACTION CATEGORY: PUBLIC HEALTH Title: Change of vaccine for the routine primary infant immunisation Date of Expiry / Review N/A For
WELSH HEALTH CIRCULAR WHC (2017) 022 Issue Date: 12 May 2017 STATUS: ACTION CATEGORY: PUBLIC HEALTH Title: Change of vaccine for the routine primary infant immunisation Date of Expiry / Review N/A For
Public Statement: Medical Policy. Effective Date: 01/01/2012 Revision Date: 03/24/2014 Code(s): Many. Document: ARB0454:04.
 ARBenefits Approval: 01/01/2012 Effective Date: 01/01/2012 Revision Date: 03/24/2014 Code(s): Many Medical Policy Title: Immunization Coverage Document: ARB0454:04 Administered by: Public Statement: 1.
ARBenefits Approval: 01/01/2012 Effective Date: 01/01/2012 Revision Date: 03/24/2014 Code(s): Many Medical Policy Title: Immunization Coverage Document: ARB0454:04 Administered by: Public Statement: 1.
Worcestershire 2011/12 Childhood Immunisation Action Plan
 Worcestershire /12 Childhood Immunisation Action Plan 1. INTRODUCTION 1.1. NHS Worcestershire (NHSW) has been set aspirational immunisation childhood immunisation targets for /12 that are unlikely to be
Worcestershire /12 Childhood Immunisation Action Plan 1. INTRODUCTION 1.1. NHS Worcestershire (NHSW) has been set aspirational immunisation childhood immunisation targets for /12 that are unlikely to be
Below you will find information about diseases, the risk of contagion, and preventive vaccinations.
 Vaccinations Below you will find information about diseases, the risk of contagion, and preventive vaccinations. DTP - Diphtheria Tetanus Polio Yellow fever Hepatitis A Typhoid fever Cerebrospinal meningitis
Vaccinations Below you will find information about diseases, the risk of contagion, and preventive vaccinations. DTP - Diphtheria Tetanus Polio Yellow fever Hepatitis A Typhoid fever Cerebrospinal meningitis
Patient Group Direction for the Administration of Haemophilus Influenzae Type b and Meningococcal C conjugated vaccine (Hib/MenC - Menitorex )
 Patient Group Direction for the Administration of Haemophilus Influenzae Type b and Meningococcal C conjugated vaccine (Hib/MenC - Menitorex ) This PGD must be read in conjunction with the core PGD (Reference:
Patient Group Direction for the Administration of Haemophilus Influenzae Type b and Meningococcal C conjugated vaccine (Hib/MenC - Menitorex ) This PGD must be read in conjunction with the core PGD (Reference:
Vaccinations for Adults
 Case: Vaccinations for Adults Lisa Winston, MD University of California, San Francisco San Francisco General Hospital A 30-year old healthy woman comes for a routine visit. She is recently married and
Case: Vaccinations for Adults Lisa Winston, MD University of California, San Francisco San Francisco General Hospital A 30-year old healthy woman comes for a routine visit. She is recently married and
Deaths/yr Efficacy Use Prev Deaths/yr. Influenza 36,000 70% 60% 18,000. Pneumonia 40,000 60% 40% 20,000 HBV 6,000 90% 30% 4,000
 Tetanus, Diptheria, Pertussis,! Measles, Mumps, Rubella, Varicella, HPV, Polio Meningococcus, Pneumococcus,! Influenza, Hepatitis B, Hepatitis A,! H influenza, Rabies, Typhoid,! Yellow Fever, Japanese
Tetanus, Diptheria, Pertussis,! Measles, Mumps, Rubella, Varicella, HPV, Polio Meningococcus, Pneumococcus,! Influenza, Hepatitis B, Hepatitis A,! H influenza, Rabies, Typhoid,! Yellow Fever, Japanese
Decision to amend access criteria for some vaccines
 3 July 2015 Decision to amend access criteria for some vaccines PHARMAC is pleased to announce the following changes to the funding access criteria for a number of vaccines. The changes in criteria follow
3 July 2015 Decision to amend access criteria for some vaccines PHARMAC is pleased to announce the following changes to the funding access criteria for a number of vaccines. The changes in criteria follow
Syrian Programme Refugees Advice on assessment of immunisation status and recommendations for additional immunisation
 Syrian Programme Refugees Advice on assessment of immunisation status and recommendations for additional immunisation Background Information Immunisation services in Syria Syria had good immunisation services,
Syrian Programme Refugees Advice on assessment of immunisation status and recommendations for additional immunisation Background Information Immunisation services in Syria Syria had good immunisation services,
Changes to the Meningococcal C conjugate (MenC) vaccine schedule. Questions and Answers
 Changes to the Meningococcal C conjugate (MenC) vaccine schedule Questions and Answers Background The meningococcal C (MenC) vaccination programme was first introduced into the UK routine immunisation
Changes to the Meningococcal C conjugate (MenC) vaccine schedule Questions and Answers Background The meningococcal C (MenC) vaccination programme was first introduced into the UK routine immunisation
Patient Group Direction For the supply and administration of
 Patient Group Direction For the supply and administration of HAEMOPHILUS INFLUENZAE TYPE B AND MENINGOCOCCAL C CONJUGATE VACCINE (HIB-MenC) MENITORIX Vaccine By Registered Nurse/Midwife/Health Visitor
Patient Group Direction For the supply and administration of HAEMOPHILUS INFLUENZAE TYPE B AND MENINGOCOCCAL C CONJUGATE VACCINE (HIB-MenC) MENITORIX Vaccine By Registered Nurse/Midwife/Health Visitor
Early Learning Centre Immunisation Policy Legislation ACT Public Health Regulations (2000)
 Early Learning Centre Immunisation Policy Legislation ACT Public Health Regulations (2000) National Quality Standard / Education and Care Services National Regulations Standard 2.1 Each child s health
Early Learning Centre Immunisation Policy Legislation ACT Public Health Regulations (2000) National Quality Standard / Education and Care Services National Regulations Standard 2.1 Each child s health
Enhanced immunisation schedule Victoria
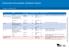 Children from March 2016 Age Disease Vaccine brand Birth Hepatitis B H-B-Vax-II Paediatric 2 months - from 6 weeks Diphtheria-tetanus-pertussis, poliomyelitis-hepatitis B- Reconstitute Site given Route
Children from March 2016 Age Disease Vaccine brand Birth Hepatitis B H-B-Vax-II Paediatric 2 months - from 6 weeks Diphtheria-tetanus-pertussis, poliomyelitis-hepatitis B- Reconstitute Site given Route
Patient Group Direction for Combined Hepatitis A & B vaccine Version: Hep A + B Start Date: 1 st January 2014 Expiry Date: 31 st December 2015
 Patient Group Direction for Combined Hepatitis A & B vaccine Version: Hep A + B 2014.1 Start 1 st January 2014 Expiry 31 st December 2015 THIS PATIENT GROUP DIRECTION HAS BEEN AGREED BY THE FOLLOWING ORGANISATIONS:
Patient Group Direction for Combined Hepatitis A & B vaccine Version: Hep A + B 2014.1 Start 1 st January 2014 Expiry 31 st December 2015 THIS PATIENT GROUP DIRECTION HAS BEEN AGREED BY THE FOLLOWING ORGANISATIONS:
National Immunisation News The Newsletter of the HSE National Immunisation Office
 December 2017 National Immunisation News The Newsletter of the HSE National Immunisation Office Contents Flu Vaccine Campaign 2017/2018 Flu vaccine campaign 2017/2018 Updated NIAC Guidelines for influenza
December 2017 National Immunisation News The Newsletter of the HSE National Immunisation Office Contents Flu Vaccine Campaign 2017/2018 Flu vaccine campaign 2017/2018 Updated NIAC Guidelines for influenza
The National Immunisation Schedule Update and Current issues. Dr Brenda Corcoran National Immunisation Office.
 The National Immunisation Schedule Update and Current issues Dr Brenda Corcoran National Immunisation Office : Dates vaccines introduced into the Irish immunisation schedule Vaccine 1937-1999 Date introduced
The National Immunisation Schedule Update and Current issues Dr Brenda Corcoran National Immunisation Office : Dates vaccines introduced into the Irish immunisation schedule Vaccine 1937-1999 Date introduced
INFECTIOUS DISEASES PROCEDURE
 INFECTIOUS DISEASES PROCEDURE Policy Hierarchy link Responsible Officer Contact Officer Superseded Documents Associated Documents Children (Education and Care Services National Law Application) Act 2010
INFECTIOUS DISEASES PROCEDURE Policy Hierarchy link Responsible Officer Contact Officer Superseded Documents Associated Documents Children (Education and Care Services National Law Application) Act 2010
CONJUGATE MENINGOCOCCAL (ACWY) VACCINE
 Cumbria & Lancashire Vaccine PGD Group ADVISORY INFORMATION FOR USE WITH PATIENT SPECIFIC DIRECTION OR INDIVIDUAL PRESCRIPTION SIGNED BY GP OR NON-MEDICAL PRESCRIBER. CONJUGATE MENINGOCOCCAL (ACWY) VACCINE
Cumbria & Lancashire Vaccine PGD Group ADVISORY INFORMATION FOR USE WITH PATIENT SPECIFIC DIRECTION OR INDIVIDUAL PRESCRIPTION SIGNED BY GP OR NON-MEDICAL PRESCRIBER. CONJUGATE MENINGOCOCCAL (ACWY) VACCINE
The hexavalent DTaP/IPV/Hib/HepB combination vaccine
 The hexavalent DTaP/IPV/Hib/HepB combination vaccine Factsheet for healthcare professionals about the neonatal selective immunisation programme for babies at risk of hepatitis B Background All babies born
The hexavalent DTaP/IPV/Hib/HepB combination vaccine Factsheet for healthcare professionals about the neonatal selective immunisation programme for babies at risk of hepatitis B Background All babies born
History and aims of immunisation. Dr Anna Clarke Department of Public Health Dr. Steevens Hospital Dublin 8
 History and aims of immunisation Dr Anna Clarke Department of Public Health Dr. Steevens Hospital Dublin 8 Objectives To examine the history of immunisation To explain the aim of immunisation To develop
History and aims of immunisation Dr Anna Clarke Department of Public Health Dr. Steevens Hospital Dublin 8 Objectives To examine the history of immunisation To explain the aim of immunisation To develop
Changes to the National Immunisation Schedule
 17 December 2013 Changes to the National Immunisation Schedule PHARMAC is pleased to announce decisions related to the National Immunisation Schedule (NIS) that will take effect from 1 July 2014. This
17 December 2013 Changes to the National Immunisation Schedule PHARMAC is pleased to announce decisions related to the National Immunisation Schedule (NIS) that will take effect from 1 July 2014. This
SCIMP GP Quick Guide to Immunisation changes for
 SCIMP GP Quick Guide to Immunisation changes for 2013-14 Version 1.0 31.7.13 This document summarises the changes to the immunisation schedules that will impact on General Practices for 2013-14, particularly
SCIMP GP Quick Guide to Immunisation changes for 2013-14 Version 1.0 31.7.13 This document summarises the changes to the immunisation schedules that will impact on General Practices for 2013-14, particularly
Communicable Disease Update; Vol. 16 (1), February 2017
 Communicable Disease Update; Vol. 16 (1), February 017 Item Type Other Authors Health Service Executive (HSE) South (South East), Department of Public Health Publisher Health Service Executive (HSE) South
Communicable Disease Update; Vol. 16 (1), February 017 Item Type Other Authors Health Service Executive (HSE) South (South East), Department of Public Health Publisher Health Service Executive (HSE) South
Young Adults (Ages 18 26)
 Young Adults (Ages 18 26) Vaccines help prevent many diseases. Some new vaccines are available today that were not in use just a few years ago. By protecting yourself, you help protect everyone around
Young Adults (Ages 18 26) Vaccines help prevent many diseases. Some new vaccines are available today that were not in use just a few years ago. By protecting yourself, you help protect everyone around
SAMPLE. PGD reviewed by: Dr Tim Patterson, Chris Faldon, John Maloney, Adrian Mackenzie
 Patient Group Direction for the Supply or Administration of combined Diphtheria, Tetanus, acellular Pertussis, inactivated Polio vaccine and Haemophilus type b conjugate Vaccine (Infanrix-IPV-HIB) to children
Patient Group Direction for the Supply or Administration of combined Diphtheria, Tetanus, acellular Pertussis, inactivated Polio vaccine and Haemophilus type b conjugate Vaccine (Infanrix-IPV-HIB) to children
BODY DEFENCES AGAINST DISEASE AND THE ROLE OF VACCINES
 BODY DEFENCES AGAINST DISEASE AND THE ROLE OF VACCINES Topic 3 This topic links in with MICROBES (from unit 1) 1. What are the 3 types of microbes? 2. Which microbe do antibiotics destroy? 3. What microbe
BODY DEFENCES AGAINST DISEASE AND THE ROLE OF VACCINES Topic 3 This topic links in with MICROBES (from unit 1) 1. What are the 3 types of microbes? 2. Which microbe do antibiotics destroy? 3. What microbe
Travel. Program Management. Schedule
 Program Management 69_4 YF (yellow fever) vaccine should be offered to all travellers to and from at-risk areas, unless they belong to the group of individuals for whom YF vaccination is contraindicated.
Program Management 69_4 YF (yellow fever) vaccine should be offered to all travellers to and from at-risk areas, unless they belong to the group of individuals for whom YF vaccination is contraindicated.
NHS Greater Glasgow & Clyde SOP No. BMT Haemopoietic Stem Cell Transplantation Services Vaccination Policy
 This policy was written with advice from Dr S Ahmed, Consultant in Public Health, Greater Glasgow & Clyde. We would like to thank him for his guidance. 1. Background It is recommended by EBMT and CDC that
This policy was written with advice from Dr S Ahmed, Consultant in Public Health, Greater Glasgow & Clyde. We would like to thank him for his guidance. 1. Background It is recommended by EBMT and CDC that
Our Mission. To promote healthy and safe travel by providing medicines, preventive vaccines and health counseling to a diverse group of travelers
 Our Mission To promote healthy and safe travel by providing medicines, preventive vaccines and health counseling to a diverse group of travelers The Travel Clinic utilizes national and international travel
Our Mission To promote healthy and safe travel by providing medicines, preventive vaccines and health counseling to a diverse group of travelers The Travel Clinic utilizes national and international travel
National Immunisation NEWS The newsletter of the National Immunisation Office Directorate of Population Health
 Page 1 of 8 National Immunisation News February 2008 National Immunisation NEWS The newsletter of the National Immunisation Office Directorate of Population Health 2008: Issue 5 HSE National Cold Chain
Page 1 of 8 National Immunisation News February 2008 National Immunisation NEWS The newsletter of the National Immunisation Office Directorate of Population Health 2008: Issue 5 HSE National Cold Chain
Kadina Child Care Centre Enrolment Form
 Court Orders Are there any court orders (including Parenting Orders and Parent Plans) relating to the powers and responsibilites of the parents in relation to the child or access to the child? No Yes go
Court Orders Are there any court orders (including Parenting Orders and Parent Plans) relating to the powers and responsibilites of the parents in relation to the child or access to the child? No Yes go
immunisation in New Zealand
 This appendix details the history of. Section A1.1 is a brief summary of when each vaccine was introduced to the National Immunisation Schedule (the Schedule). This summary includes vaccines which were
This appendix details the history of. Section A1.1 is a brief summary of when each vaccine was introduced to the National Immunisation Schedule (the Schedule). This summary includes vaccines which were
PGD for Td/IPV. Version number: 1.1
 Patient Group Direction (PGD) for the Administration of Combined Diphtheria (low dose), Tetanus, and Inactivated Poliomyelitis Vaccine (Td/IPV REVAXIS ) This PGD must be read in conjunction with the core
Patient Group Direction (PGD) for the Administration of Combined Diphtheria (low dose), Tetanus, and Inactivated Poliomyelitis Vaccine (Td/IPV REVAXIS ) This PGD must be read in conjunction with the core
3 rd dose. 3 rd or 4 th dose, see footnote 5. see footnote 13. for certain high-risk groups
 Figure 1. Recommended immunization schedule for persons aged 0 through 18 years 2013. (FOR THOSE WHO FALL BEHIND OR START LATE, SEE THE CATCH-UP SCHEDULE [FIGURE 2]). These recommendations must be read
Figure 1. Recommended immunization schedule for persons aged 0 through 18 years 2013. (FOR THOSE WHO FALL BEHIND OR START LATE, SEE THE CATCH-UP SCHEDULE [FIGURE 2]). These recommendations must be read
GOVERNING BOARD. Date of Meeting 15 May 2013 Agenda Item No 13. Title Immunisation and Vaccination Report 2012/13
 GOVERNING BOARD Date of Meeting 15 May 2013 Agenda Item No 13 Title Immunisation and Vaccination Report 2012/13 Purpose of Paper To update the CCG on the performance and quality of vaccination programmes
GOVERNING BOARD Date of Meeting 15 May 2013 Agenda Item No 13 Title Immunisation and Vaccination Report 2012/13 Purpose of Paper To update the CCG on the performance and quality of vaccination programmes
Guideline for the immunization of HIV infected persons in Sri Lanka
 DOI: http://doi.org/10.4038/joshhm.v3i0.64 Guideline for the immunization of HIV infected persons in Sri Lanka Dr. M. K. Darshanie Mallikarachchi, Consultant Venereologist, Provincial General Hospital
DOI: http://doi.org/10.4038/joshhm.v3i0.64 Guideline for the immunization of HIV infected persons in Sri Lanka Dr. M. K. Darshanie Mallikarachchi, Consultant Venereologist, Provincial General Hospital
NHS GRAMPIAN IMMUNISATION PROGRAMMES ANNUAL REPORT 2010/11
 NHS GRAMPIAN IMMUNISATION PROGRAMMES ANNUAL REPORT 2010/11 NHS GRAMPIAN IMMUNISATION STEERING GROUP March 2012 1 Table of Content 1. Purpose of this report.3 2. Uptake of immunisation: key points during
NHS GRAMPIAN IMMUNISATION PROGRAMMES ANNUAL REPORT 2010/11 NHS GRAMPIAN IMMUNISATION STEERING GROUP March 2012 1 Table of Content 1. Purpose of this report.3 2. Uptake of immunisation: key points during
PLACEMENT OPERATIONS - FREQUENTLY ASKED QUESTIONS
 PLACEMENT OPERATIONS - FREQUENTLY ASKED QUESTIONS IMMUNISATION AND HEALTH RECORDS Q: When should I commence my immunisations? A: All immunisation requirements must be completed prior to placement. It is
PLACEMENT OPERATIONS - FREQUENTLY ASKED QUESTIONS IMMUNISATION AND HEALTH RECORDS Q: When should I commence my immunisations? A: All immunisation requirements must be completed prior to placement. It is
SAMPLE. PGD Reviewed by: Chris Faldon, John Maloney, Tim Patterson, Adrian MacKenzie, Claire Stein
 Patient Group Direction for the supply or administration of haemophilus influenzae type B and meningococcal conjugate vaccine to children requiring immunisation as part of the national childhood vaccination
Patient Group Direction for the supply or administration of haemophilus influenzae type B and meningococcal conjugate vaccine to children requiring immunisation as part of the national childhood vaccination
Immunizations are among the most cost effective and widely used public health interventions.
 Focused Issue of This Month Recommended by the Korean Pediatric Society, 2008 Hoan Jong Lee, MD Department of Pediatrics, Seoul National University College of Medicine E mail : hoanlee@snu.ac.kr J Korean
Focused Issue of This Month Recommended by the Korean Pediatric Society, 2008 Hoan Jong Lee, MD Department of Pediatrics, Seoul National University College of Medicine E mail : hoanlee@snu.ac.kr J Korean
The hexavalent DTaP/IPV/Hib/HepB combination vaccine
 The hexavalent combination vaccine Information for registered healthcare practitioners about the neonatal selective immunisation programme for babies at high risk (babies born to hepatitis B positive mothers)
The hexavalent combination vaccine Information for registered healthcare practitioners about the neonatal selective immunisation programme for babies at high risk (babies born to hepatitis B positive mothers)
301 W. Alder, Missoula, MT or
 301 W. Alder, Missoula, MT 59802 406-258-4745 or 406-258-3363 Routine Immunizations are available on a walk-in basis: Mondays, Tuesdays, Thursdays, and Fridays from 9:00am to 4:30pm Wednesdays, 10:00am-4:30pm
301 W. Alder, Missoula, MT 59802 406-258-4745 or 406-258-3363 Routine Immunizations are available on a walk-in basis: Mondays, Tuesdays, Thursdays, and Fridays from 9:00am to 4:30pm Wednesdays, 10:00am-4:30pm
Pneumococcal vaccination in UK: an update. Dr Richard Pebody Immunisation Department Health Protection Agency Centre for Infections
 Pneumococcal vaccination in UK: an update Dr Richard Pebody Immunisation Department Health Protection Agency Centre for Infections Leading infectious causes of mortality, 2000 WHO estimates 3.5 Deaths
Pneumococcal vaccination in UK: an update Dr Richard Pebody Immunisation Department Health Protection Agency Centre for Infections Leading infectious causes of mortality, 2000 WHO estimates 3.5 Deaths
Thank you for offering the opportunity to comment on the proposed amendments to the Pharmacy Act (Proposal #16-HLTC005).
 Administration & Office of the Medical Officer of Health Telephone: 519-383-8331 160 Exmouth Street Toll-free: 1-800-667-1839 Point Edward, ON N7T 7Z6 Fax: 519-383-7092 www.lambtonhealth.on.ca Ontario
Administration & Office of the Medical Officer of Health Telephone: 519-383-8331 160 Exmouth Street Toll-free: 1-800-667-1839 Point Edward, ON N7T 7Z6 Fax: 519-383-7092 www.lambtonhealth.on.ca Ontario
Guidelines for immunisation of children following treatment with high dose chemotherapy and Haematopoietic Stem Cell Transplantation (HSCT)
 Guidelines for immunisation of children following treatment with high dose chemotherapy and Haematopoietic Stem Cell Transplantation (HSCT) Version 2.0 Approved by Haem / Onc Senior Clinical Management
Guidelines for immunisation of children following treatment with high dose chemotherapy and Haematopoietic Stem Cell Transplantation (HSCT) Version 2.0 Approved by Haem / Onc Senior Clinical Management
How to use our country pages when researching country related travel health risks
 Country pages guide How to use our country pages when researching country related travel health risks Overview We hope you will like the new layout for our country information pages; we have listened to
Country pages guide How to use our country pages when researching country related travel health risks Overview We hope you will like the new layout for our country information pages; we have listened to
Patient Group Direction Hepatitis A vaccine Version: Hepatitis A 2015 Start Date: 1 st July 2015 Expiry Date:30 th June 2018
 Patient Group Direction Hepatitis A vaccine Version: Hepatitis A 2015 Start 1 st July 2015 Expiry 30 th June 2018 THIS PATIENT GROUP DIRECTION HAS BEEN AGREED BY THE FOLLOWING ORGANISATIONS: BLACKPOOL
Patient Group Direction Hepatitis A vaccine Version: Hepatitis A 2015 Start 1 st July 2015 Expiry 30 th June 2018 THIS PATIENT GROUP DIRECTION HAS BEEN AGREED BY THE FOLLOWING ORGANISATIONS: BLACKPOOL
OVERVIEW OF THE NATIONAL CHILDHOOD IMMUNISATION PROGRAMME IN SINGAPORE
 OVERVIEW OF THE NATIONAL CHILDHOOD IMMUNISATION PROGRAMME IN SINGAPORE Dr Tiong Wei Wei, MD, MPH Senior Assistant Director Policy and Control Branch, Communicable Diseases Division Ministry of Health 9
OVERVIEW OF THE NATIONAL CHILDHOOD IMMUNISATION PROGRAMME IN SINGAPORE Dr Tiong Wei Wei, MD, MPH Senior Assistant Director Policy and Control Branch, Communicable Diseases Division Ministry of Health 9
National Immunisation News The newsletter of the HSE National Immunisation Office
 February 2014 National Immunisation News The newsletter of the HSE National Immunisation Office CONTENTS Adverse Local Reactions following 4 in 1 booster 2013 Immunisation Guidelines for Ireland Flu Season
February 2014 National Immunisation News The newsletter of the HSE National Immunisation Office CONTENTS Adverse Local Reactions following 4 in 1 booster 2013 Immunisation Guidelines for Ireland Flu Season
Enrollment Application
 Enrollment Application www.ladybirdnursery.com Child s Full Name: Date of Birth: / / Sex: Male Female Address: Postcode Home Telephone No: Mobile No: Email Address: Parent Details: If parents live separately,
Enrollment Application www.ladybirdnursery.com Child s Full Name: Date of Birth: / / Sex: Male Female Address: Postcode Home Telephone No: Mobile No: Email Address: Parent Details: If parents live separately,
National Pandemic Vaccination Programme
 March 2010 National Pandemic Vaccination Programme Swine Flu Vaccination Programme Pneumococcal Catch-up Campaign Measles Outbreak National Immunisation Guidelines NIO Website HPV Vaccine Campaign Vaccine
March 2010 National Pandemic Vaccination Programme Swine Flu Vaccination Programme Pneumococcal Catch-up Campaign Measles Outbreak National Immunisation Guidelines NIO Website HPV Vaccine Campaign Vaccine
Patient Group Direction for Varicella vaccine Version: Varicella Start Date: 1 st May 2014 Expiry Date: 30 th April 2017
 Patient Group Direction for Varicella vaccine Version: Varicella 2014.1 Start Date: 1 st May 2014 Expiry Date: 30 th April 2017 THIS PATIENT GROUP DIRECTION HAS BEEN AGREED BY THE FOLLOWING ORGANISATIONS:
Patient Group Direction for Varicella vaccine Version: Varicella 2014.1 Start Date: 1 st May 2014 Expiry Date: 30 th April 2017 THIS PATIENT GROUP DIRECTION HAS BEEN AGREED BY THE FOLLOWING ORGANISATIONS:
P L E A S E N O T E - There will be a charge for each person, even if your travel has been discussed previously with your own GP.
 I M P O R T A N T - It is preferable that your Travel Consultation appointment is made one month prior to your departure. Please return this record this form promptly. You will be advised of an appointment
I M P O R T A N T - It is preferable that your Travel Consultation appointment is made one month prior to your departure. Please return this record this form promptly. You will be advised of an appointment
Utah s Immunization Rule Individual Vaccine Requirements
 Utah s Immunization Rule Individual Vaccine Requirements Which vaccines are required for school entry in Utah? Grades K-6: 5 doses DTaP (4 doses if the 4 th dose was given after the 4 th birthday) 4 doses
Utah s Immunization Rule Individual Vaccine Requirements Which vaccines are required for school entry in Utah? Grades K-6: 5 doses DTaP (4 doses if the 4 th dose was given after the 4 th birthday) 4 doses
Immunisation Subcommittee of PTAC Meeting held 23 April (minutes for web publishing)
 Immunisation Subcommittee of PTAC Meeting held 23 April 2013 (minutes for web publishing) Immunisation Subcommittee minutes are published in accordance with the Terms of Reference for the Pharmacology
Immunisation Subcommittee of PTAC Meeting held 23 April 2013 (minutes for web publishing) Immunisation Subcommittee minutes are published in accordance with the Terms of Reference for the Pharmacology
Student Health Services 881 Commonwealth Ave, West / Student Information (To be completed by the student) Student Name Last First Middle
 Medical Clearance The following information must be completed on the medical history form, if any information is missing the form will be considered incomplete and will not be processed. If you have questions,
Medical Clearance The following information must be completed on the medical history form, if any information is missing the form will be considered incomplete and will not be processed. If you have questions,
Student Immunisation Record Faculty of Medicine. Section 1: Information. Notes
 Student Immunisation Record Faculty of Section 1: Information Students enrolled in programs offered by the Faculty of are REQUIRED to provide evidence of their immunisation status for the diseases listed
Student Immunisation Record Faculty of Section 1: Information Students enrolled in programs offered by the Faculty of are REQUIRED to provide evidence of their immunisation status for the diseases listed
UNIVERSITY OF UTAH INTERNATIONAL TRAVEL CLINIC Phone: Fax: N Medical Dr. Salt Lake City, UT 84132
 Welcome to UNIVERSITY OF UTAH INTERNATIONAL TRAVEL CLINIC Phone: 801-801-581-2898 Fax: 801-585-7315 50 N Medical Dr. Salt Lake City, UT 84132 Our goal is to help you have a safe and enjoyable experience
Welcome to UNIVERSITY OF UTAH INTERNATIONAL TRAVEL CLINIC Phone: 801-801-581-2898 Fax: 801-585-7315 50 N Medical Dr. Salt Lake City, UT 84132 Our goal is to help you have a safe and enjoyable experience
Vaccination Workshop for Immigrants
 National Center for Emerging and Zoonotic Infectious Diseases Vaccination Workshop for Immigrants Zachary White, MPH Public Health Advisor March 12, 2018 Learning Objectives After this workshop, you should
National Center for Emerging and Zoonotic Infectious Diseases Vaccination Workshop for Immigrants Zachary White, MPH Public Health Advisor March 12, 2018 Learning Objectives After this workshop, you should
Vaccination Cribsheet for Borders Health Professionals Childhood immunisations and general vaccination information July 2007
 Vaccination Cribsheet for Health Professionals Childhood immunisations and general vaccination information July 2007 SECTION CONTENT 1 Timetable of routine immunisations 2 Important immunisation policies
Vaccination Cribsheet for Health Professionals Childhood immunisations and general vaccination information July 2007 SECTION CONTENT 1 Timetable of routine immunisations 2 Important immunisation policies
Risk Consultation and Assessment
 Risk Consultation and Assessment Please find product Prescribing Information at the end of this presentation UK/VAC/0047/16b Date of preparation: July 2016 The pre-travel risk consultation When to do it?
Risk Consultation and Assessment Please find product Prescribing Information at the end of this presentation UK/VAC/0047/16b Date of preparation: July 2016 The pre-travel risk consultation When to do it?
TRAVEL VACCINATION. Acknowledgements: Dr Conrad Moreira
 TRAVEL VACCINATION Acknowledgements: Dr Conrad Moreira www.who.int www.travelvaccinationcentre.com.a www.traveldoctor.com.au WWW.TRAVELDOCTOR.COM. AU Measles Home grown measles eliminated in Australia
TRAVEL VACCINATION Acknowledgements: Dr Conrad Moreira www.who.int www.travelvaccinationcentre.com.a www.traveldoctor.com.au WWW.TRAVELDOCTOR.COM. AU Measles Home grown measles eliminated in Australia
A list of useful resources including advice on how to reduce the risk of certain health problems is available below.
 Honduras Capital City : "Tegucigalpa" Official Language: "Spanish" Monetary Unit: "lempira (L)" General Information The information on these pages should be used to research health risks and to inform
Honduras Capital City : "Tegucigalpa" Official Language: "Spanish" Monetary Unit: "lempira (L)" General Information The information on these pages should be used to research health risks and to inform
Immunisation Subcommittee of PTAC Meeting held 10 February (minutes for web publishing)
 Immunisation Subcommittee of PTAC Meeting held 10 February 2014 (minutes for web publishing) Immunisation Subcommittee minutes are published in accordance with the Terms of Reference for the Pharmacology
Immunisation Subcommittee of PTAC Meeting held 10 February 2014 (minutes for web publishing) Immunisation Subcommittee minutes are published in accordance with the Terms of Reference for the Pharmacology
Manitoba Annual Immunization Surveillance Report
 Annual Immunization Surveillance Report January 1 to December 31, 2014 Epidemiology & Surveillance Public Branch Public and Primary Care Division, y Living and Seniors Released: January 2016 TABLE OF CONTENTS
Annual Immunization Surveillance Report January 1 to December 31, 2014 Epidemiology & Surveillance Public Branch Public and Primary Care Division, y Living and Seniors Released: January 2016 TABLE OF CONTENTS
Immunise your child on time. It s their best protection
 Immunise your child on time It s their best protection If you are a parent or caregiver, this pamphlet is for you. It tells you about the recommended ages for your baby to receive their immunisations.
Immunise your child on time It s their best protection If you are a parent or caregiver, this pamphlet is for you. It tells you about the recommended ages for your baby to receive their immunisations.
Vaccinating special risk groups/responding to outbreaks. VACCSline
 Vaccinating special risk groups/responding to outbreaks 2016 Protecting vulnerable persons Raise awareness of recommendations for certain patients Apply this to own workplace when developing immunisation
Vaccinating special risk groups/responding to outbreaks 2016 Protecting vulnerable persons Raise awareness of recommendations for certain patients Apply this to own workplace when developing immunisation
Immunisation and health screening
 TOPIC 4 THEME 2 Immunisation and health screening LEARNING OUTCOMES To understand the purpose of immunisation and screening programmes To be aware of the immunisation and screening services that are available
TOPIC 4 THEME 2 Immunisation and health screening LEARNING OUTCOMES To understand the purpose of immunisation and screening programmes To be aware of the immunisation and screening services that are available
