Kasper Huus, Principal Scientist Peptide and Protein Formulation Injectable Drug Product Global Research Novo Nordisk A/S
|
|
|
- Darrell Watson
- 5 years ago
- Views:
Transcription
1 Insulin thermostability Paris, 26th September Insulin thermostability, ligand interactions, and correlation of DSC data with other stability indicating methods 2nd European Microcal Symposium Institut Pasteur, Paris 26th-27th September, 2016 Kasper Huus, Principal Scientist Peptide and Protein Formulation Injectable Drug Product Global Research Novo Nordisk A/S
2 Insulin thermostability Paris, 26th September Outline Insulin structure and selfassociation forms DSC interpretation of insulin thermal unfolding Correlation between DSC results and other methods for chemical and physical stability Thermostability of insulin analogues
3 Insulin thermostability Paris, 26th September Human Insulin structure Primary structure of human insulin Secondary and tertiary structure A21 B1 A1 B30 Insulin monomer from T 6 crystal structure Brange et al., Diabetes Care, 1990
4 Insulin thermostability Paris, 26th September Human Insulin self-association forms Monomer Dimer T 6 hexamer 2 Zn 2+ /6 insulin R 6 hexamer 2 Zn 2+ /6 insulin 6 phenol/6 insulin B1-B8 convert to α-helix A21 B1 A1 B30 Circulation in blood stream Receptor binding Building block for hexamer Crystalline storage in β- cells granula in vivo Pharmaceutical formulation (with additional excipients)
5 Insulin thermostability Paris, 26th September Stability of insulin formulations Physical stability issues: Primarily fibrillation, precipitation, crystallization Chemical stability Main degradation pathways in neutral, soluble insulin formulations: Asn B3 deamidation Dimerization Polymerization
6 Insulin thermostability Paris, 26th September Biphasic thermal denaturation of human insulin DSC thermogram obtained under formulation relevant conditions Phenolic ligand Buffer Zn 2+ /hexamer 3.0 Anionic ligand ph 7.4 Human insulin 0.6 mm Phenol 20 mm Phosphate 7 mm Sodium chloride 50 mm
7 Insulin thermostability Paris, 26th September Biphasic thermal denaturation of human insulin Known origins of biphasic thermal denaturation Unfolding of separate domains at different temperatures Larger proteins and antibodies Oligomeric proteins: Dissociation of oligomer followed by unfolding of monomer Ligand induced stabilization of native protein when the ligand binding is not saturated
8 Thermal Denaturation of Human Insulin Parameters effecting the biphasic properties Insulin thermostability Paris, 26th September Zinc concentration effect Insulin concentration effect Monomeric analogues zinc-free Fixed ratio Zn 2+ ratio (2 Zn 2+ /hexamer). Insulin concentrations 0.1, 0.3, 0.6 mm (left to right) 2 Zn 2+ /hex Fixed insulin concentration at 0.6 mm. Zn 2+ concentrations are indicated in the figure. Huus et al.,biochemistry, 2005 Human insulin Insulin Asp B28 Insulin Asp B9,Glu B27
9 Thermal Denaturation of Human Insulin NUV-CD studies Insulin thermostability Paris, 26th September NUV-CD thermal unfolding NUV-CD. From top to bottom: Insulin Asp B9,Glu B27, Insulin Asp B28, and HI - all zinc-free Bottom: HI 3 Zn 2+ /hexamer 22 C -400 CD 275 nm Zn/hexamer Biphasic denaturation is caused by zinc redistribution during heating Huus et al.,biochemistry, 2005
10 Insulin thermostability Paris, 26th September Formation of amyloid fibrils Amyloid fibrils Insoluble, supramolecular with cross β-sheet structure formed of peptide/protein monomers Physical change no change of covalent bonds (but hydrogen bonds, van der Waals interactions, hydrophobic effect) Stochastic process Requires build-up (lag time) of critical mass (seed) May occur in bulk solution (no physical stress) or dependent on surface/interface interaction Physical stress (e.g. shear forces) required Interaction with primary packaging crucial Unacceptable in a pharmaceutical drug product Cryo Electron Microscopy (Top) Model (Below) [Jimeniz et al., 2001] Partially unfolded structure Denatured peptide Nucleus
11 Insulin thermostability Paris, 26th September Insulin formulations: An unexpected effect of phenol ThT assay:45h, 30 C, 960 rpm shake 8 replica, 1 µm ThT Phenol binds to the T 6 insulin hexamer Conformational change to R 6 insulin hexamer with improved chemical and physical stability Pulls the equilibrium further away from the monomeric state Lag time (h) Lagtime (h) Recovery (% ) Insulin (no zinc) Insulin + 2 Zn/6 ins Recovery (%) mm phenol + 10 mm phenol + 15 mm phenol + 30 mm phenol mm phenol + 10 mm phenol + 15 mm phenol + 30 mm phenol 6x phenol No effect of phenol on insulin lag time Shorter lag times of insulin+zn with increasing phenol! Morten Schlein, unpublished data
12 Insulin thermostability Paris, 26th September Phenolic ligands influence on insulin thermostability 20 mm phenolic ligand 80 mm phenol 20 mm phenol Without phenol 100 mm phenolic ligand Destabilization of zinc-free insulin Stabilization of zinc hexamers Huus et al.,biochemistry, 2006
13 Insulin thermostability Paris, 26th September DSC vs physical stability of formulations in vials DSC data Sample Tm,onset ( C) Tm,hexamer ( C) A B Ref Sample A Start 28 days Huus, unpublished data
14 Covalently linked insulin dimer - Extremly stable Insulin thermostability Paris, 26th September Disulfide linked monomers (B25C) Dimer and hexamer structure equivalent to self-association forms of human insulin Does not bind to the insulin receptor DSC Thioflavin T fibrillation assay Vinther et al, PLoS One, 2012
15 Insulin thermostability Paris, 26th September Insulin analog with additional disulfide bond CysA10, CysB4, desb30 insulin Self-association similar to human insulin Increased affinity to the insulin receptor Vinther et al, Protein Science, 2013 Does not adopt R-confirmation, stays in T-conformation DSC in zinc-free condition 4SS-insulin monomers are more stable than HI zinc hexamers Also strongly reduced propensity to fibrillate
16 Insulin thermostability Paris, 26th September Ligands and insulin chemical stability Effect of zinc on AsnB3 deamidation and covalent dimer formation Without phenol 20 mm phenol Zinc-hexamer formation stabilizes slightly against deamidation. Contradictory to the effect of zinc on thermostability, additional zinc increases deamidation in T 6 hexamer In R 6 hexameric insulin this effect is not seen. Possibly a specific interaction in the C-terminal B-chain Huus et al., Pharm. Res., 2006
17 Insulin thermostability Paris, 26th September Ligands and insulin chemical stability Effect of zinc on other chemical degradation Increased stability against fragmentation and formation of other related substances with increased zinc levels: Correlates to thermostability Without phenol 20 mm phenol Huus et al., Pharm. Res., 2006
18 Insulin thermostability Paris, 26th September Ligands and insulin chemical stability Effect of phenolic and anionic ligands (with 3 Zn 2+ /hexamer) Effect on the biphasic insulin denaturation Effect on Asn B3 deamidation correlates with thermostability 3 Zn 2+ /hexamer 3 Zn 2+ /hexamer + 20 mm phenol 3 Zn 2+ /hexamer + 20 mm phenol + 50 mm NaCl Huus et al., Pharm. Res., 2006
19 Insulin thermostability Paris, 26th September DSC for stability screening of new insulin analogues Different insulin analogues formulated similarly Data from DSC screening: T m,onset T m, hexamer unfolding Huus, unpublished data
20 Insulin thermostability Paris, 26th September HMWP formation of various insulin analogues Good correlation with T m,onset data HMWP formation rate (%/month) HMWP formation rate (%/month) T m, hexamer ( C) T m, onset ( C) Huus, unpublished data
21 Insulin thermostability Paris, 26th September Purity loss of various insulin analogues Slight correlation with T m,onset data Purity loss rel. to reference Purity loss rel. to reference T m, hexamer ( C) T m, onset ( C) Huus, unpublished data
22 Insulin thermostability Paris, 26th September Conclusions and applications - I Zinc-free vs. zinchexamers Biphasic denaturation at low concentrations of zinc Understanding of the mechanism provides more rational use of DSC in insulin formulation development
23 Insulin thermostability Paris, 26th September Conclusions and applications - II DSC as a method to study ligand binding to the insulin hexamer Correlation between thermal and chemical stability studied at formulation-relevant zinc concentrations Can be used for studying both phenolic and anionic ligands Useful in examination of zinc binding and self-association properties of insulin analogues Clear correlation for B3 deamidation but not for dimerization/other Low zinc results (biphasic denaturation) correlate with high-zinc results DSC is useful for studying insulin formulations and identifying new stabilizing ligands Limitations: Binding is studied at high temperatures may not correlate to lower temperatures Interactions with the unfolded state Limitations: DSC may give false positive or false negative results (examples: KSCN and resorcinol) DSC results are exclusively related to the structural stability of the protein
24 Insulin thermostability Paris, 26th September Acknowledgements University of Copenhagen Sven Frøkjær Marco van de Weert Bent W. Sigurskjold Novo Nordisk Helle B. Olsen Svend Havelund Morten Schlein Tine Vinther Ida Boldsen and many more
Organic Molecules: Proteins
 Organic Molecules: Proteins Proteins Most structurally & functionally diverse group Function: involved in almost everything enzymes (pepsin, DNA polymerase) structure (keratin, collagen) carriers & transport
Organic Molecules: Proteins Proteins Most structurally & functionally diverse group Function: involved in almost everything enzymes (pepsin, DNA polymerase) structure (keratin, collagen) carriers & transport
BIO 311C Spring Lecture 15 Friday 26 Feb. 1
 BIO 311C Spring 2010 Lecture 15 Friday 26 Feb. 1 Illustration of a Polypeptide amino acids peptide bonds Review Polypeptide (chain) See textbook, Fig 5.21, p. 82 for a more clear illustration Folding and
BIO 311C Spring 2010 Lecture 15 Friday 26 Feb. 1 Illustration of a Polypeptide amino acids peptide bonds Review Polypeptide (chain) See textbook, Fig 5.21, p. 82 for a more clear illustration Folding and
Sheet #5 Dr. Mamoun Ahram 8/7/2014
 P a g e 1 Protein Structure Quick revision - Levels of protein structure: primary, secondary, tertiary & quaternary. - Primary structure is the sequence of amino acids residues. It determines the other
P a g e 1 Protein Structure Quick revision - Levels of protein structure: primary, secondary, tertiary & quaternary. - Primary structure is the sequence of amino acids residues. It determines the other
Chapter 3. Structure of Enzymes. Enzyme Engineering
 Chapter 3. Structure of Enzymes Enzyme Engineering 3.1 Introduction With purified protein, Determining M r of the protein Determining composition of amino acids and the primary structure Determining the
Chapter 3. Structure of Enzymes Enzyme Engineering 3.1 Introduction With purified protein, Determining M r of the protein Determining composition of amino acids and the primary structure Determining the
(12) Patent Application Publication (10) Pub. No.: US 2015/ A1
 (19) United States (12) Patent Application Publication (10) Pub. No.: US 2015/0126442 A1 Naver et al. US 2015O126442A1 (43) Pub. Date: (54) (71) (72) (73) (21) (22) (86) NSULIN FORMULATIONS Applicant:
(19) United States (12) Patent Application Publication (10) Pub. No.: US 2015/0126442 A1 Naver et al. US 2015O126442A1 (43) Pub. Date: (54) (71) (72) (73) (21) (22) (86) NSULIN FORMULATIONS Applicant:
Understand how protein is formed by amino acids
 Identify between fibrous and globular proteins Understand how protein is formed by amino acids Describe the structure of proteins using specific examples Functions of proteins Fibrous proteins Globular
Identify between fibrous and globular proteins Understand how protein is formed by amino acids Describe the structure of proteins using specific examples Functions of proteins Fibrous proteins Globular
Chapter 5 THE STRUCTURE AND FUNCTION OF LARGE BIOLOGICAL MOLECULES
 Chapter 5 THE STRUCTURE AND FUNCTION OF LARGE BIOLOGICAL MOLECULES You Must Know The role of dehydration synthesis in the formation of organic compounds and hydrolysis in the digestion of organic compounds.
Chapter 5 THE STRUCTURE AND FUNCTION OF LARGE BIOLOGICAL MOLECULES You Must Know The role of dehydration synthesis in the formation of organic compounds and hydrolysis in the digestion of organic compounds.
Protein Structure and Function
 Protein Structure and Function Protein Structure Classification of Proteins Based on Components Simple proteins - Proteins containing only polypeptides Conjugated proteins - Proteins containing nonpolypeptide
Protein Structure and Function Protein Structure Classification of Proteins Based on Components Simple proteins - Proteins containing only polypeptides Conjugated proteins - Proteins containing nonpolypeptide
The building blocks of life.
 The building blocks of life. The 4 Major Organic Biomolecules The large molecules (biomolecules OR polymers) are formed when smaller building blocks (monomers) bond covalently. via anabolism Small molecules
The building blocks of life. The 4 Major Organic Biomolecules The large molecules (biomolecules OR polymers) are formed when smaller building blocks (monomers) bond covalently. via anabolism Small molecules
BIOCHEMISTRY 460 FIRST HOUR EXAMINATION FORM A (yellow) ANSWER KEY February 11, 2008
 WRITE YOUR AND I.D. NUMBER LEGIBLY ON EVERY PAGE PAGES WILL BE SEPARATED FOR GRADING! CHECK TO BE SURE YOU HAVE 6 PAGES, (print): ANSWERS INCLUDING COVER PAGE. I swear/affirm that I have neither given
WRITE YOUR AND I.D. NUMBER LEGIBLY ON EVERY PAGE PAGES WILL BE SEPARATED FOR GRADING! CHECK TO BE SURE YOU HAVE 6 PAGES, (print): ANSWERS INCLUDING COVER PAGE. I swear/affirm that I have neither given
Objective: You will be able to explain how the subcomponents of
 Objective: You will be able to explain how the subcomponents of nucleic acids determine the properties of that polymer. Do Now: Read the first two paragraphs from enduring understanding 4.A Essential knowledge:
Objective: You will be able to explain how the subcomponents of nucleic acids determine the properties of that polymer. Do Now: Read the first two paragraphs from enduring understanding 4.A Essential knowledge:
Macromolecules Structure and Function
 Macromolecules Structure and Function Within cells, small organic molecules (monomers) are joined together to form larger molecules (polymers). Macromolecules are large molecules composed of thousands
Macromolecules Structure and Function Within cells, small organic molecules (monomers) are joined together to form larger molecules (polymers). Macromolecules are large molecules composed of thousands
The Structure and Function of Large Biological Molecules Part 4: Proteins Chapter 5
 Key Concepts: The Structure and Function of Large Biological Molecules Part 4: Proteins Chapter 5 Proteins include a diversity of structures, resulting in a wide range of functions Proteins Enzymatic s
Key Concepts: The Structure and Function of Large Biological Molecules Part 4: Proteins Chapter 5 Proteins include a diversity of structures, resulting in a wide range of functions Proteins Enzymatic s
Biology 12 - Biochemistry Practice Exam
 Biology 12 - Biochemistry Practice Exam Name: Water: 1. The bond between water molecules is a (n) a. ionic bond b. covalent bond c. polar covalent bond d. hydrogen bond 2. The water properties: good solvent,
Biology 12 - Biochemistry Practice Exam Name: Water: 1. The bond between water molecules is a (n) a. ionic bond b. covalent bond c. polar covalent bond d. hydrogen bond 2. The water properties: good solvent,
Copyright 2008 Pearson Education, Inc., publishing as Pearson Benjamin Cummings
 Concept 5.4: Proteins have many structures, resulting in a wide range of functions Proteins account for more than 50% of the dry mass of most cells Protein functions include structural support, storage,
Concept 5.4: Proteins have many structures, resulting in a wide range of functions Proteins account for more than 50% of the dry mass of most cells Protein functions include structural support, storage,
Characterizing Protein Structure by DSC
 Characterizing Protein Structure by DSC Christin T. Choma TA Instruments, 109 Lukens Drive, New Castle, DE 19720, USA The non-covalent interactions dictating the three dimensional structures of proteins
Characterizing Protein Structure by DSC Christin T. Choma TA Instruments, 109 Lukens Drive, New Castle, DE 19720, USA The non-covalent interactions dictating the three dimensional structures of proteins
Biology 12. Biochemistry. Water - a polar molecule Water (H 2 O) is held together by covalent bonds.
 Biology 12 Biochemistry Water - a polar molecule Water (H 2 O) is held together by covalent bonds. Electrons in these bonds spend more time circulating around the larger Oxygen atom than the smaller Hydrogen
Biology 12 Biochemistry Water - a polar molecule Water (H 2 O) is held together by covalent bonds. Electrons in these bonds spend more time circulating around the larger Oxygen atom than the smaller Hydrogen
Amino Acids and Proteins (2) Professor Dr. Raid M. H. Al-Salih
 Amino Acids and Proteins (2) Professor Dr. Raid M. H. Al-Salih 1 Some important biologically active peptides 2 Proteins The word protein is derived from Greek word, proteios which means primary. As the
Amino Acids and Proteins (2) Professor Dr. Raid M. H. Al-Salih 1 Some important biologically active peptides 2 Proteins The word protein is derived from Greek word, proteios which means primary. As the
KEY NAME (printed very legibly) UT-EID
 BIOLOGY 311C - Brand Spring 2007 KEY NAME (printed very legibly) UT-EID EXAMINATION II Before beginning, check to be sure that this exam contains 7 pages (including front and back) numbered consecutively,
BIOLOGY 311C - Brand Spring 2007 KEY NAME (printed very legibly) UT-EID EXAMINATION II Before beginning, check to be sure that this exam contains 7 pages (including front and back) numbered consecutively,
Protein structure. Dr. Mamoun Ahram Summer semester,
 Protein structure Dr. Mamoun Ahram Summer semester, 2017-2018 Overview of proteins Proteins have different structures and some have repeating inner structures, other do not. A protein may have gazillion
Protein structure Dr. Mamoun Ahram Summer semester, 2017-2018 Overview of proteins Proteins have different structures and some have repeating inner structures, other do not. A protein may have gazillion
Proteins. Proteins. Proteins. Proteins. Effect of different R groups: Nonpolar amino acids. Amino acids H C OH H R. Multipurpose molecules.
 Multipurpose molecules 2008-2009 Most structurally & functionally diverse group Function: involved in almost everything enzymes (pepsin, DNA polymerase) structure (keratin, collagen) carriers & transport
Multipurpose molecules 2008-2009 Most structurally & functionally diverse group Function: involved in almost everything enzymes (pepsin, DNA polymerase) structure (keratin, collagen) carriers & transport
Structure of proteins
 Structure of proteins Presented by Dr. Mohammad Saadeh The requirements for the Pharmaceutical Biochemistry I Philadelphia University Faculty of pharmacy Structure of proteins The 20 a.a commonly found
Structure of proteins Presented by Dr. Mohammad Saadeh The requirements for the Pharmaceutical Biochemistry I Philadelphia University Faculty of pharmacy Structure of proteins The 20 a.a commonly found
MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question.
 Practice Quiz 1 AP Bio Sept 2016 Name MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1) The element present in all organic molecules is A) hydrogen.
Practice Quiz 1 AP Bio Sept 2016 Name MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1) The element present in all organic molecules is A) hydrogen.
The Effects of Induced Aggregation of IgG and Insulin Preparations on In Vitro T Cell Stimulation
 Website home page picture here The Effects of Induced Aggregation of IgG and Insulin Preparations on In Vitro T Cell Stimulation 2 nd EIP Symposium 17 th -19 th November 29 Immune Responses to Protein
Website home page picture here The Effects of Induced Aggregation of IgG and Insulin Preparations on In Vitro T Cell Stimulation 2 nd EIP Symposium 17 th -19 th November 29 Immune Responses to Protein
Ch5: Macromolecules. Proteins
 Ch5: Macromolecules Proteins Essential Knowledge 4.A.1 The subcomponents of biological molecules and their sequence determine the properties of that molecule A. Structure and function of polymers are derived
Ch5: Macromolecules Proteins Essential Knowledge 4.A.1 The subcomponents of biological molecules and their sequence determine the properties of that molecule A. Structure and function of polymers are derived
Biological Molecules B Lipids, Proteins and Enzymes. Triglycerides. Glycerol
 Glycerol www.biologymicro.wordpress.com Biological Molecules B Lipids, Proteins and Enzymes Lipids - Lipids are fats/oils and are present in all cells- they have different properties for different functions
Glycerol www.biologymicro.wordpress.com Biological Molecules B Lipids, Proteins and Enzymes Lipids - Lipids are fats/oils and are present in all cells- they have different properties for different functions
Water: 1. The bond between water molecules is a(n) a. ionic bond b. covalent bond c. polar covalent bond d. hydrogen bond
 Biology 12 - Biochemistry Practice Exam KEY Water: 1. The bond between water molecules is a(n) a. ionic bond b. covalent bond c. polar covalent bond d. hydrogen bond 2. The water properties: good solvent,
Biology 12 - Biochemistry Practice Exam KEY Water: 1. The bond between water molecules is a(n) a. ionic bond b. covalent bond c. polar covalent bond d. hydrogen bond 2. The water properties: good solvent,
NOTE: For studying for the final, you only have to worry about those with an asterix (*)
 NOTE: For studying for the final, you only have to worry about those with an asterix (*) (*)1. An organic compound is one that: a. contains carbon b. is slightly acidic c. forms long chains d. is soluble
NOTE: For studying for the final, you only have to worry about those with an asterix (*) (*)1. An organic compound is one that: a. contains carbon b. is slightly acidic c. forms long chains d. is soluble
a) The statement is true for X = 400, but false for X = 300; b) The statement is true for X = 300, but false for X = 200;
 1. Consider the following statement. To produce one molecule of each possible kind of polypeptide chain, X amino acids in length, would require more atoms than exist in the universe. Given the size of
1. Consider the following statement. To produce one molecule of each possible kind of polypeptide chain, X amino acids in length, would require more atoms than exist in the universe. Given the size of
! Proteins are involved functionally in almost everything: " Receptor Proteins - Respond to external stimuli. " Storage Proteins - Storing amino acids
 Proteins Most structurally & functionally diverse group! Proteins are involved functionally in almost everything: Proteins Multi-purpose molecules 2007-2008 Enzymatic proteins - Speed up chemical reactions!
Proteins Most structurally & functionally diverse group! Proteins are involved functionally in almost everything: Proteins Multi-purpose molecules 2007-2008 Enzymatic proteins - Speed up chemical reactions!
The Structure and Func.on of Macromolecules Proteins GRU1L6
 The Structure and Func.on of Macromolecules Proteins GRU1L6 Proteins Proteins Most structurally & functionally diverse group Function: involved in almost everything enzymes (pepsin, DNA polymerase) structure
The Structure and Func.on of Macromolecules Proteins GRU1L6 Proteins Proteins Most structurally & functionally diverse group Function: involved in almost everything enzymes (pepsin, DNA polymerase) structure
Chapter 1-2 Review Assignment
 Class: Date: Chapter 1-2 Review Assignment Multiple Choice dentify the choice that best completes the statement or answers the question. Corn seedlings A student wanted to design an investigation to see
Class: Date: Chapter 1-2 Review Assignment Multiple Choice dentify the choice that best completes the statement or answers the question. Corn seedlings A student wanted to design an investigation to see
The Basics: A general review of molecular biology:
 The Basics: A general review of molecular biology: DNA Transcription RNA Translation Proteins DNA (deoxy-ribonucleic acid) is the genetic material It is an informational super polymer -think of it as the
The Basics: A general review of molecular biology: DNA Transcription RNA Translation Proteins DNA (deoxy-ribonucleic acid) is the genetic material It is an informational super polymer -think of it as the
Biology 5A Fall 2010 Macromolecules Chapter 5
 Learning Outcomes: Macromolecules List and describe the four major classes of molecules Describe the formation of a glycosidic linkage and distinguish between monosaccharides, disaccharides, and polysaccharides
Learning Outcomes: Macromolecules List and describe the four major classes of molecules Describe the formation of a glycosidic linkage and distinguish between monosaccharides, disaccharides, and polysaccharides
Biological Molecules
 The Chemical Building Blocks of Life Chapter 3 Biological molecules consist primarily of -carbon bonded to carbon, or -carbon bonded to other molecules. Carbon can form up to 4 covalent bonds. Carbon may
The Chemical Building Blocks of Life Chapter 3 Biological molecules consist primarily of -carbon bonded to carbon, or -carbon bonded to other molecules. Carbon can form up to 4 covalent bonds. Carbon may
The Chemical Building Blocks of Life. Chapter 3
 The Chemical Building Blocks of Life Chapter 3 Biological Molecules Biological molecules consist primarily of -carbon bonded to carbon, or -carbon bonded to other molecules. Carbon can form up to 4 covalent
The Chemical Building Blocks of Life Chapter 3 Biological Molecules Biological molecules consist primarily of -carbon bonded to carbon, or -carbon bonded to other molecules. Carbon can form up to 4 covalent
Study Guide Chapter 5 MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question
 Study Guide Chapter 5 MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question 1) What type of covalent bond between amino acid side chains (R groups) functions
Study Guide Chapter 5 MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question 1) What type of covalent bond between amino acid side chains (R groups) functions
Q1: Circle the best correct answer: (15 marks)
 Q1: Circle the best correct answer: (15 marks) 1. Which one of the following incorrectly pairs an amino acid with a valid chemical characteristic a. Glycine, is chiral b. Tyrosine and tryptophan; at neutral
Q1: Circle the best correct answer: (15 marks) 1. Which one of the following incorrectly pairs an amino acid with a valid chemical characteristic a. Glycine, is chiral b. Tyrosine and tryptophan; at neutral
Additional problems: 1. Match and label the conjugate acid and base pairs in the following reactions. Which one of these systems is a good buffer?
 1 ESA Biochemistry Fall 2001 eview problems for the first exam Acid/Base chemistry Sections to review: 10.2,.3,.4,.6.9,.12,.13 omplete the following sentences 1. 2 3 is a acid. 2. The anion of a weak acid
1 ESA Biochemistry Fall 2001 eview problems for the first exam Acid/Base chemistry Sections to review: 10.2,.3,.4,.6.9,.12,.13 omplete the following sentences 1. 2 3 is a acid. 2. The anion of a weak acid
Proteins and their structure
 Proteins and their structure Proteins are the most abundant biological macromolecules, occurring in all cells and all parts of cells. Proteins also occur in great variety; thousands of different kinds,
Proteins and their structure Proteins are the most abundant biological macromolecules, occurring in all cells and all parts of cells. Proteins also occur in great variety; thousands of different kinds,
Biological systems interact, and these systems and their interactions possess complex properties. STOP at enduring understanding 4A
 Biological systems interact, and these systems and their interactions possess complex properties. STOP at enduring understanding 4A Homework Watch the Bozeman video called, Biological Molecules Objective:
Biological systems interact, and these systems and their interactions possess complex properties. STOP at enduring understanding 4A Homework Watch the Bozeman video called, Biological Molecules Objective:
University of Bradford ethesis
 University of Bradford ethesis This thesis is hosted in Bradford Scholars The University of Bradford Open Access repository. Visit the repository for full metadata or to contact the repository team University
University of Bradford ethesis This thesis is hosted in Bradford Scholars The University of Bradford Open Access repository. Visit the repository for full metadata or to contact the repository team University
Polymers 2015, 7, ; doi: /polym Article. Chandrasekar Manoharan 1,2 and Jagdish Singh 1, *
 Polymers 215, 7, 836-85; doi:1.339/polym75836 Article OPEN ACCESS polymers ISSN 273-436 www.mdpi.com/journal/polymers Addition of Zinc Improves the Physical Stability of Insulin in the Primary Emulsification
Polymers 215, 7, 836-85; doi:1.339/polym75836 Article OPEN ACCESS polymers ISSN 273-436 www.mdpi.com/journal/polymers Addition of Zinc Improves the Physical Stability of Insulin in the Primary Emulsification
Short polymer. Dehydration removes a water molecule, forming a new bond. Longer polymer (a) Dehydration reaction in the synthesis of a polymer
 HO 1 2 3 H HO H Short polymer Dehydration removes a water molecule, forming a new bond Unlinked monomer H 2 O HO 1 2 3 4 H Longer polymer (a) Dehydration reaction in the synthesis of a polymer HO 1 2 3
HO 1 2 3 H HO H Short polymer Dehydration removes a water molecule, forming a new bond Unlinked monomer H 2 O HO 1 2 3 4 H Longer polymer (a) Dehydration reaction in the synthesis of a polymer HO 1 2 3
The Structure and Function of Macromolecules
 The Structure and Function of Macromolecules Macromolecules are polymers Polymer long molecule consisting of many similar building blocks. Monomer the small building block molecules. Carbohydrates, proteins
The Structure and Function of Macromolecules Macromolecules are polymers Polymer long molecule consisting of many similar building blocks. Monomer the small building block molecules. Carbohydrates, proteins
Lecture Series 2 Macromolecules: Their Structure and Function
 Lecture Series 2 Macromolecules: Their Structure and Function Reading Assignments Read Chapter 4 (Protein structure & Function) Biological Substances found in Living Tissues The big four in terms of macromolecules
Lecture Series 2 Macromolecules: Their Structure and Function Reading Assignments Read Chapter 4 (Protein structure & Function) Biological Substances found in Living Tissues The big four in terms of macromolecules
Nafith Abu Tarboush DDS, MSc, PhD
 Nafith Abu Tarboush DDS, MSc, PhD natarboush@ju.edu.jo www.facebook.com/natarboush Protein conformation Many conformations are possible for proteins due to flexibility of amino acids linked by peptide
Nafith Abu Tarboush DDS, MSc, PhD natarboush@ju.edu.jo www.facebook.com/natarboush Protein conformation Many conformations are possible for proteins due to flexibility of amino acids linked by peptide
the nature and importance of biomacromolecules in the chemistry of the cell: synthesis of biomacromolecules through the condensation reaction lipids
 the nature and importance of biomacromolecules in the chemistry of the cell: synthesis of biomacromolecules through the condensation reaction lipids and their sub-units; the role of lipids in the plasma
the nature and importance of biomacromolecules in the chemistry of the cell: synthesis of biomacromolecules through the condensation reaction lipids and their sub-units; the role of lipids in the plasma
Lecture Series 2 Macromolecules: Their Structure and Function
 Lecture Series 2 Macromolecules: Their Structure and Function Reading Assignments Read Chapter 4 (Protein structure & Function) Biological Substances found in Living Tissues The big four in terms of macromolecules
Lecture Series 2 Macromolecules: Their Structure and Function Reading Assignments Read Chapter 4 (Protein structure & Function) Biological Substances found in Living Tissues The big four in terms of macromolecules
AP Biology. Proteins. Proteins. Proteins. Amino acids H C OH H R. Effect of different R groups: Polar amino acids polar or charged & hydrophilic
 Most structurally & functionally diverse group : involved in almost everything enzymes (pepsin, DNA polymerase) structure (keratin, collagen) carriers & transport (, aquaporin) cell communication signals
Most structurally & functionally diverse group : involved in almost everything enzymes (pepsin, DNA polymerase) structure (keratin, collagen) carriers & transport (, aquaporin) cell communication signals
This exam consists of two parts. Part I is multiple choice. Each of these 25 questions is worth 2 points.
 MBB 407/511 Molecular Biology and Biochemistry First Examination - October 1, 2002 Name Social Security Number This exam consists of two parts. Part I is multiple choice. Each of these 25 questions is
MBB 407/511 Molecular Biology and Biochemistry First Examination - October 1, 2002 Name Social Security Number This exam consists of two parts. Part I is multiple choice. Each of these 25 questions is
Metal ions and amyloid fiber formation in neurodegenerative diseases. Copper, zinc and iron in Alzheimer's, Parkinson's and prion diseases
 Metal ions and amyloid fiber formation in neurodegenerative diseases. Copper, zinc and iron in Alzheimer's, Parkinson's and prion diseases Viles, JH 2012 Elsevier B.V. All rights reserved. For additional
Metal ions and amyloid fiber formation in neurodegenerative diseases. Copper, zinc and iron in Alzheimer's, Parkinson's and prion diseases Viles, JH 2012 Elsevier B.V. All rights reserved. For additional
A. Lipids: Water-Insoluble Molecules
 Biological Substances found in Living Tissues Lecture Series 3 Macromolecules: Their Structure and Function A. Lipids: Water-Insoluble Lipids can form large biological molecules, but these aggregations
Biological Substances found in Living Tissues Lecture Series 3 Macromolecules: Their Structure and Function A. Lipids: Water-Insoluble Lipids can form large biological molecules, but these aggregations
Qualitative test of protein-lab2
 1- Qualitative chemical reactions of amino acid protein functional groups: Certain functional groups in proteins can react to produce characteristically colored products. The color intensity of the product
1- Qualitative chemical reactions of amino acid protein functional groups: Certain functional groups in proteins can react to produce characteristically colored products. The color intensity of the product
Protein Structure Danilo V. Rogayan Jr.
 Protein Structure Danilo V. Rogayan Jr. RMTU San Marcelino Outline I Categories of Proteins Fibrous proteins Globular proteins II Protein Denaturation & Renaturation III Functions of Proteins IV Journal
Protein Structure Danilo V. Rogayan Jr. RMTU San Marcelino Outline I Categories of Proteins Fibrous proteins Globular proteins II Protein Denaturation & Renaturation III Functions of Proteins IV Journal
AP Bio. Protiens Chapter 5 1
 Concept.4: Proteins have many structures, resulting in a wide range of functions Proteins account for more than 0% of the dry mass of most cells Protein functions include structural support, storage, transport,
Concept.4: Proteins have many structures, resulting in a wide range of functions Proteins account for more than 0% of the dry mass of most cells Protein functions include structural support, storage, transport,
Chapter 5 Structure and Function Of Large Biomolecules
 Formation of Macromolecules Monomers Polymers Macromolecules Smaller larger Chapter 5 Structure and Function Of Large Biomolecules monomer: single unit dimer: two monomers polymer: three or more monomers
Formation of Macromolecules Monomers Polymers Macromolecules Smaller larger Chapter 5 Structure and Function Of Large Biomolecules monomer: single unit dimer: two monomers polymer: three or more monomers
130327SCH4U_biochem April 09, 2013
 Option B: B1.1 ENERGY Human Biochemistry If more energy is taken in from food than is used up, weight gain will follow. Similarly if more energy is used than we supply our body with, weight loss will occur.
Option B: B1.1 ENERGY Human Biochemistry If more energy is taken in from food than is used up, weight gain will follow. Similarly if more energy is used than we supply our body with, weight loss will occur.
Bioinformatics for molecular biology
 Bioinformatics for molecular biology Structural bioinformatics tools, predictors, and 3D modeling Structural Biology Review Dr Research Scientist Department of Microbiology, Oslo University Hospital -
Bioinformatics for molecular biology Structural bioinformatics tools, predictors, and 3D modeling Structural Biology Review Dr Research Scientist Department of Microbiology, Oslo University Hospital -
Role of C-terminal B-chain residues in insulin assembly: the structure of hexameric LysB 28 ProB 29 -human insulin
 Role of C-terminal B-chain residues in insulin assembly: the structure of hexameric LysB 28 ProB 29 -human insulin Ewa Ciszak l, John M Beals 2, Bruce H Frank 2, Jeffrey C Baker 2, Nancy D Carter 2 and
Role of C-terminal B-chain residues in insulin assembly: the structure of hexameric LysB 28 ProB 29 -human insulin Ewa Ciszak l, John M Beals 2, Bruce H Frank 2, Jeffrey C Baker 2, Nancy D Carter 2 and
WHY IS THIS IMPORTANT?
 CHAPTER 2 FUNDAMENTAL CHEMISTRY FOR MICROBIOLOGY WHY IS THIS IMPORTANT? An understanding of chemistry is essential to understand cellular structure and function, which are paramount for your understanding
CHAPTER 2 FUNDAMENTAL CHEMISTRY FOR MICROBIOLOGY WHY IS THIS IMPORTANT? An understanding of chemistry is essential to understand cellular structure and function, which are paramount for your understanding
Chemistry and Biochemistry 153A Spring Exam 2
 hemistry and Biochemistry 153A Spring 2011 Exam 2 Instructions: You will have 1 hour 45 minutes to complete the exam. You may use a pencil (recommended) or blue or black ink pen to write your answers.
hemistry and Biochemistry 153A Spring 2011 Exam 2 Instructions: You will have 1 hour 45 minutes to complete the exam. You may use a pencil (recommended) or blue or black ink pen to write your answers.
Multiple-Choice Questions Answer ALL 20 multiple-choice questions on the Scantron Card in PENCIL
 Multiple-Choice Questions Answer ALL 20 multiple-choice questions on the Scantron Card in PENCIL For Questions 1-10 choose ONE INCORRECT answer. 1. Which ONE of the following statements concerning the
Multiple-Choice Questions Answer ALL 20 multiple-choice questions on the Scantron Card in PENCIL For Questions 1-10 choose ONE INCORRECT answer. 1. Which ONE of the following statements concerning the
Chapter 9. Protein Folding, Dynamics, and Structural Evolution
 Chapter 9 Protein Folding, Dynamics, and Structural Evolution Proteins folding Self-assembly Primary sequence dictates three dimensional structure Folding accessory proteins Protein disulfide isomerases
Chapter 9 Protein Folding, Dynamics, and Structural Evolution Proteins folding Self-assembly Primary sequence dictates three dimensional structure Folding accessory proteins Protein disulfide isomerases
Proteins. Amino acids, structure and function. The Nobel Prize in Chemistry 2012 Robert J. Lefkowitz Brian K. Kobilka
 Proteins Amino acids, structure and function The Nobel Prize in Chemistry 2012 Robert J. Lefkowitz Brian K. Kobilka O O HO N N HN OH Ser65-Tyr66-Gly67 The Nobel prize in chemistry 2008 Osamu Shimomura,
Proteins Amino acids, structure and function The Nobel Prize in Chemistry 2012 Robert J. Lefkowitz Brian K. Kobilka O O HO N N HN OH Ser65-Tyr66-Gly67 The Nobel prize in chemistry 2008 Osamu Shimomura,
Chemistry of Carbon. All living things rely on one particular type of molecule: carbon
 Ach Chemistry of Carbon All living things rely on one particular type of molecule: carbon Carbon atom with an outer shell of four electrons can form covalent bonds with four atoms. In organic molecules,
Ach Chemistry of Carbon All living things rely on one particular type of molecule: carbon Carbon atom with an outer shell of four electrons can form covalent bonds with four atoms. In organic molecules,
Spectroscopic analysis of amyloid fibril formation in SH3-domains
 Spectroscopy 17 (2003) 647 652 647 IOS Press Spectroscopic analysis of amyloid fibril formation in SH3-domains Salvador Ventura and Luis Serrano European Molecular Biology Laboratory, Meyerhofstrasse 1,
Spectroscopy 17 (2003) 647 652 647 IOS Press Spectroscopic analysis of amyloid fibril formation in SH3-domains Salvador Ventura and Luis Serrano European Molecular Biology Laboratory, Meyerhofstrasse 1,
Biological molecules
 Biological molecules 04-04-16 Announcements Your lab report 1 is due now Quiz 1 is on Wednesday at the beginning of class, so don t be late Review Macromolecues are large molecules necessary for life made
Biological molecules 04-04-16 Announcements Your lab report 1 is due now Quiz 1 is on Wednesday at the beginning of class, so don t be late Review Macromolecues are large molecules necessary for life made
Macromolecules (Learning Objectives)
 Macromolecules (Learning Objectives) Recognize the role of water in synthesis and breakdown of polymers Name &recognize the monomer and the chemical bond that holds the polymeric structure of all biomolecules
Macromolecules (Learning Objectives) Recognize the role of water in synthesis and breakdown of polymers Name &recognize the monomer and the chemical bond that holds the polymeric structure of all biomolecules
Biology: Life on Earth Chapter 3 Molecules of life
 Biology: Life on Earth Chapter 3 Molecules of life Chapter 3 Outline 3.1 Why Is Carbon So Important in Biological Molecules? p. 38 3.2 How Are Organic Molecules Synthesized? p. 38 3.3 What Are Carbohydrates?
Biology: Life on Earth Chapter 3 Molecules of life Chapter 3 Outline 3.1 Why Is Carbon So Important in Biological Molecules? p. 38 3.2 How Are Organic Molecules Synthesized? p. 38 3.3 What Are Carbohydrates?
Defense Antibodies, interferons produced in response to infection Coordination and growth (signaling) Hormones (e.g. insulin, growth hormone) Communic
 Proteins Chapter 3 An Introduction to Organic Compounds Most varied of the biomolecules Also called polypeptides Make up more than half the dry weight of cells Categorized by function Lecture 3: Proteins
Proteins Chapter 3 An Introduction to Organic Compounds Most varied of the biomolecules Also called polypeptides Make up more than half the dry weight of cells Categorized by function Lecture 3: Proteins
CS612 - Algorithms in Bioinformatics
 Spring 2016 Protein Structure February 7, 2016 Introduction to Protein Structure A protein is a linear chain of organic molecular building blocks called amino acids. Introduction to Protein Structure Amine
Spring 2016 Protein Structure February 7, 2016 Introduction to Protein Structure A protein is a linear chain of organic molecular building blocks called amino acids. Introduction to Protein Structure Amine
We are going to talk about two classifications of proteins: fibrous & globular.
 Slide # 13 (fibrous proteins) : We are going to talk about two classifications of proteins: fibrous & globular. *fibrous proteins: (dense fibers) *Their structures are mainly formed of the secondary structure
Slide # 13 (fibrous proteins) : We are going to talk about two classifications of proteins: fibrous & globular. *fibrous proteins: (dense fibers) *Their structures are mainly formed of the secondary structure
Lecture Series 2 Macromolecules: Their Structure and Function
 Lecture Series 2 Macromolecules: Their Structure and Function Reading Assignments Read Chapter 4 (Protein structure & Function) Biological Substances found in Living Tissues The big four in terms of macromolecules
Lecture Series 2 Macromolecules: Their Structure and Function Reading Assignments Read Chapter 4 (Protein structure & Function) Biological Substances found in Living Tissues The big four in terms of macromolecules
CALCIUM CASEINATE. What Is Casein?
 CALCIUM CASEINATE A high quality milk protein that is Calcium Rich, manufactured from fresh pasteurized skimmed milk through precipitation of casein followed by neutralization and natural drying, which
CALCIUM CASEINATE A high quality milk protein that is Calcium Rich, manufactured from fresh pasteurized skimmed milk through precipitation of casein followed by neutralization and natural drying, which
Nafith Abu Tarboush DDS, MSc, PhD
 Nafith Abu Tarboush DDS, MSc, PhD natarboush@ju.edu.jo www.facebook.com/natarboush Types of proteins Proteins can be divided into two groups according to structure: Fibrous (fiber-like with a uniform secondary-structure
Nafith Abu Tarboush DDS, MSc, PhD natarboush@ju.edu.jo www.facebook.com/natarboush Types of proteins Proteins can be divided into two groups according to structure: Fibrous (fiber-like with a uniform secondary-structure
Paper No. 01. Paper Title: Food Chemistry. Module-16: Protein Structure & Denaturation
 Paper No. 01 Paper Title: Food Chemistry Module-16: Protein Structure & Denaturation The order of amino acids in a protein molecule is genetically determined. This primary sequence of amino acids must
Paper No. 01 Paper Title: Food Chemistry Module-16: Protein Structure & Denaturation The order of amino acids in a protein molecule is genetically determined. This primary sequence of amino acids must
The three important structural features of proteins:
 The three important structural features of proteins: a. Primary (1 o ) The amino acid sequence (coded by genes) b. Secondary (2 o ) The interaction of amino acids that are close together or far apart in
The three important structural features of proteins: a. Primary (1 o ) The amino acid sequence (coded by genes) b. Secondary (2 o ) The interaction of amino acids that are close together or far apart in
Macromolecules. Note: If you have not taken Chemistry 11 (or if you ve forgotten some of it), read the Chemistry Review Notes on your own.
 Macromolecules Note: If you have not taken Chemistry 11 (or if you ve forgotten some of it), read the Chemistry Review Notes on your own. Macromolecules are giant molecules made up of thousands or hundreds
Macromolecules Note: If you have not taken Chemistry 11 (or if you ve forgotten some of it), read the Chemistry Review Notes on your own. Macromolecules are giant molecules made up of thousands or hundreds
Supplementary Materials for
 advances.sciencemag.org/cgi/content/full/4/3/eaaq0762/dc1 Supplementary Materials for Structures of monomeric and oligomeric forms of the Toxoplasma gondii perforin-like protein 1 Tao Ni, Sophie I. Williams,
advances.sciencemag.org/cgi/content/full/4/3/eaaq0762/dc1 Supplementary Materials for Structures of monomeric and oligomeric forms of the Toxoplasma gondii perforin-like protein 1 Tao Ni, Sophie I. Williams,
BIOLOGICAL MOLECULES REVIEW-UNIT 1 1. The factor being tested in an experiment is the A. data. B. variable. C. conclusion. D. observation. 2.
 BIOLOGICAL MOLECULES REVIEW-UNIT 1 1. The factor being tested in an experiment is the A. data. B. variable. C. conclusion. D. observation. 2. A possible explanation for an event that occurs in nature is
BIOLOGICAL MOLECULES REVIEW-UNIT 1 1. The factor being tested in an experiment is the A. data. B. variable. C. conclusion. D. observation. 2. A possible explanation for an event that occurs in nature is
The Molecular Chaperone α-crystallin as an Excipient in an Insulin Formulation
 Pharm Res (2010) 27:1337 1347 DOI 10.1007/s11095-010-0116-8 RESEARCH PAPER The Molecular Chaperone α-crystallin as an Excipient in an Insulin Formulation Tue Rasmussen & Ruedeeporn Tantipolphan & Marco
Pharm Res (2010) 27:1337 1347 DOI 10.1007/s11095-010-0116-8 RESEARCH PAPER The Molecular Chaperone α-crystallin as an Excipient in an Insulin Formulation Tue Rasmussen & Ruedeeporn Tantipolphan & Marco
Biomolecules: amino acids
 Biomolecules: amino acids Amino acids Amino acids are the building blocks of proteins They are also part of hormones, neurotransmitters and metabolic intermediates There are 20 different amino acids in
Biomolecules: amino acids Amino acids Amino acids are the building blocks of proteins They are also part of hormones, neurotransmitters and metabolic intermediates There are 20 different amino acids in
Organic Compounds. Compounds that contain CARBON are called organic. Macromolecules are large organic molecules.
 Macromolecules 1 Organic Compounds Compounds that contain CARBON are called organic. Macromolecules are large organic molecules. 2 Carbon (C) Carbon has 4 electrons in outer shell. Carbon can form covalent
Macromolecules 1 Organic Compounds Compounds that contain CARBON are called organic. Macromolecules are large organic molecules. 2 Carbon (C) Carbon has 4 electrons in outer shell. Carbon can form covalent
4. THE THREE-DIMENSIONAL STRUCTURE OF PROTEINS
 4. THE THREE-DIMENSIONAL STRUCTURE OF PROTEINS 4.1 Proteins Structures and Function Levels of Structure in Proteins Native conformation - Biological activity - Random structure: no obvious regular repeating
4. THE THREE-DIMENSIONAL STRUCTURE OF PROTEINS 4.1 Proteins Structures and Function Levels of Structure in Proteins Native conformation - Biological activity - Random structure: no obvious regular repeating
Biological Molecules
 Chemical Building Blocks of Life Chapter 3 Biological Molecules Biological molecules consist primarily of -carbon bonded to carbon, or -carbon bonded to other molecules. Carbon can form up to 4 covalent
Chemical Building Blocks of Life Chapter 3 Biological Molecules Biological molecules consist primarily of -carbon bonded to carbon, or -carbon bonded to other molecules. Carbon can form up to 4 covalent
AP BIOLOGY: READING ASSIGNMENT FOR CHAPTER 5
 1) Complete the following table: Class Monomer Functions Carbohydrates 1. 3. Lipids 1. 3. Proteins 1. 3. 4. 5. 6. Nucleic Acids 1. 2) Circle the atoms of these two glucose molecules that will be removed
1) Complete the following table: Class Monomer Functions Carbohydrates 1. 3. Lipids 1. 3. Proteins 1. 3. 4. 5. 6. Nucleic Acids 1. 2) Circle the atoms of these two glucose molecules that will be removed
Bio 12 Chapter 2 Test Review
 Bio 12 Chapter 2 Test Review 1.Know the difference between ionic and covalent bonds In order to complete outer shells in electrons bonds can be Ionic; one atom donates or receives electrons Covalent; atoms
Bio 12 Chapter 2 Test Review 1.Know the difference between ionic and covalent bonds In order to complete outer shells in electrons bonds can be Ionic; one atom donates or receives electrons Covalent; atoms
SRTUCTURE OF PROTEINS DR. A. TARAB DEPT. OF BIOCHEMISTRY HKMU
 SRTUCTURE OF PROTEINS DR. A. TARAB DEPT. OF BIOCHEMISTRY HKMU I. OVERVIEW The twenty amino acids commonly found in proteins are joined together by peptide bonds The linear sequence of the linked amino
SRTUCTURE OF PROTEINS DR. A. TARAB DEPT. OF BIOCHEMISTRY HKMU I. OVERVIEW The twenty amino acids commonly found in proteins are joined together by peptide bonds The linear sequence of the linked amino
Macromolecules. copyright cmassengale
 Macromolecules 1 Organic Compounds Compounds that contain CARBON are called organic. Macromolecules are large organic molecules. 2 Carbon (C) Carbon has 4 electrons in outer shell. Carbon can form covalent
Macromolecules 1 Organic Compounds Compounds that contain CARBON are called organic. Macromolecules are large organic molecules. 2 Carbon (C) Carbon has 4 electrons in outer shell. Carbon can form covalent
The Structure and Function of Large Biological Molecules
 Chapter 5 The Structure and Function of Large Biological Molecules PowerPoint Lecture Presentations for Biology Eighth Edition Neil Campbell and Jane Reece Lectures by Chris Romero, updated by Erin Barley
Chapter 5 The Structure and Function of Large Biological Molecules PowerPoint Lecture Presentations for Biology Eighth Edition Neil Campbell and Jane Reece Lectures by Chris Romero, updated by Erin Barley
Organic Compounds. Compounds that contain CARBON are called organic. Macromolecules are large organic molecules.
 Macromolecules Organic Compounds Compounds that contain CARBON are called organic. Macromolecules are large organic molecules. Carbon (C) Carbon has 4 electrons in outer shell. Carbon can form covalent
Macromolecules Organic Compounds Compounds that contain CARBON are called organic. Macromolecules are large organic molecules. Carbon (C) Carbon has 4 electrons in outer shell. Carbon can form covalent
Proteins. (b) Protein Structure and Conformational Change
 Proteins (b) Protein Structure and Conformational Change Protein Structure and Conformational Change Proteins contain the elements carbon (C), hydrogen (H), oxygen (O2) and nitrogen (N2) Some may also
Proteins (b) Protein Structure and Conformational Change Protein Structure and Conformational Change Proteins contain the elements carbon (C), hydrogen (H), oxygen (O2) and nitrogen (N2) Some may also
OPTION GROUP: BIOLOGICAL MOLECULES 3 PROTEINS WORKBOOK. Tyrone R.L. John, Chartered Biologist
 NAME: OPTION GROUP: BIOLOGICAL MOLECULES 3 PROTEINS WORKBOOK Tyrone R.L. John, Chartered Biologist 1 Tyrone R.L. John, Chartered Biologist 2 Instructions REVISION CHECKLIST AND ASSESSMENT OBJECTIVES Regular
NAME: OPTION GROUP: BIOLOGICAL MOLECULES 3 PROTEINS WORKBOOK Tyrone R.L. John, Chartered Biologist 1 Tyrone R.L. John, Chartered Biologist 2 Instructions REVISION CHECKLIST AND ASSESSMENT OBJECTIVES Regular
INTERACTION DRUG BODY
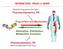 INTERACTION DRUG BODY What the drug does to the body What the body does to the drug Receptors - intracellular receptors - membrane receptors - Channel receptors - G protein-coupled receptors - Tyrosine-kinase
INTERACTION DRUG BODY What the drug does to the body What the body does to the drug Receptors - intracellular receptors - membrane receptors - Channel receptors - G protein-coupled receptors - Tyrosine-kinase
Introduction to proteins and protein structure
 Introduction to proteins and protein structure The questions and answers below constitute an introduction to the fundamental principles of protein structure. They are all available at [link]. What are
Introduction to proteins and protein structure The questions and answers below constitute an introduction to the fundamental principles of protein structure. They are all available at [link]. What are
Proteins. These are polymers of 20 common amino acids linked in various sequences. Proteins differ in molecular mass, molecular structure and shape
 Proteins These are polymers of 20 common amino acids linked in various sequences. Proteins differ in molecular mass, molecular structure and shape Characteristics if protein Amino acids are linked by covalent
Proteins These are polymers of 20 common amino acids linked in various sequences. Proteins differ in molecular mass, molecular structure and shape Characteristics if protein Amino acids are linked by covalent
Structure and Function of Macromolecules Chapter 5 Macromolecules Macromolecules Multiple Units Synthesis of Dimers and Polymers
 Structure and Function of Macromolecules Chapter 5 Macromolecules Giant molecules weighing over 100,000 daltons Emergent properties not found in component parts Macromolecules Multiple Units meris = one
Structure and Function of Macromolecules Chapter 5 Macromolecules Giant molecules weighing over 100,000 daltons Emergent properties not found in component parts Macromolecules Multiple Units meris = one
Biomolecules. Biomolecules. Carbohydrates. Biol 219 Lec 3 Fall Polysaccharides. Function: Glucose storage Fig. 2.2
 Biomolecules Biomolecules Monomers Polymers Carbohydrates monosaccharides polysaccharides fatty acids triglycerides Proteins amino acids polypeptides Nucleic Acids nucleotides DNA, RNA Carbohydrates Carbohydrates
Biomolecules Biomolecules Monomers Polymers Carbohydrates monosaccharides polysaccharides fatty acids triglycerides Proteins amino acids polypeptides Nucleic Acids nucleotides DNA, RNA Carbohydrates Carbohydrates
Scantron Instructions
 BIOLOGY 1A MIDTERM # 1 February 17 th, 2012 NAME SECTION # DISCUSSION GSI 1. Sit every other seat and sit by section number. Place all books and paper on the floor. Turn off all phones, pagers, etc. and
BIOLOGY 1A MIDTERM # 1 February 17 th, 2012 NAME SECTION # DISCUSSION GSI 1. Sit every other seat and sit by section number. Place all books and paper on the floor. Turn off all phones, pagers, etc. and
