A Panel of Serum Biomarkers Differentiates IgA Nephropathy from Other Renal Diseases
|
|
|
- Carmel Jackson
- 6 years ago
- Views:
Transcription
1 A Panel of Serum Biomarkers Differentiates IgA Nephropathy from Other Renal Diseases Hiroyuki Yanagawa 1., Hitoshi Suzuki 1., Yusuke Suzuki 1, Krzysztof Kiryluk 2, Ali G. Gharavi 2, Kiyoshi Matsuoka 3, Yuko Makita 1, Bruce A. Julian 4,5, Jan Novak 5, Yasuhiko Tomino 1 * 1 Division of Nephrology, Department of Internal Medicine, Juntendo University Faculty of Medicine, Tokyo, Japan, 2 Division of Nephrology, Department of Medicine, Columbia University College of Physicians and Surgeons, New York, New York, United States of America, 3 Clinical Research Center, Juntendo University Faculty of Medicine, Tokyo, Japan, 4 Department of Medicine, University of Alabama at Birmingham, Birmingham, Alabama, United States of America, 5 Department of Microbiology, University of Alabama at Birmingham, Birmingham, Alabama, United States of America Abstract Background and Objectives: There is increasing evidence that galactose-deficient IgA1 (Gd-IgA1) and Gd-IgA1-containing immune complexes are important for the pathogenesis of IgA nephropathy (IgAN). In the present study, we assessed a novel noninvasive multi-biomarker approach in the diagnostic test for IgAN. Materials and Methods: We compared serum levels of IgA, IgG, Gd-IgA1, Gd-IgA1-specific IgG and Gd-IgA1-specific IgA in 135 IgAN patients, 79 patients with non-igan chronic kidney disease (CKD) controls and 106 healthy controls. Serum was collected at the time of kidney biopsy from all IgAN and CKD patients. Results: Each serum marker was significantly elevated in IgAN patients compared to CKD (P,0.001) and healthy controls (P,0.001). While 41% of IgAN patients had elevated serum Gd-IgA1 levels, 91% of these patients exhibited Gd-IgA1-specific IgG levels above the 90th percentile for healthy controls (sensitivity 89%, specificity 92%). Although up to 25% of CKD controls, particularly those with immune-mediated glomerular diseases including lupus nephritis, also had elevated serum levels of Gd-IgA1-specific IgG, most IgAN patients had elevated levels of Gd-IgA1-specific antibody of both isotypes. Serum levels of Gd-IgA1-specific IgG were associated with renal histological grading. Furthermore, there was a trend toward higher serum levels of Gd-IgA1-specific IgG in IgAN patients with at least moderate proteinuria ($1.0 g/g), compared to patients with less proteinuria. Conclusions: Serum levels of Gd-IgA1-specific antibodies are elevated in most IgAN patients, and their assessment, together with serum levels of Gd-IgA1, improves the specificity of the assays. Our observations suggest that a panel of serum biomarkers may be helpful in differentiating IgAN from other glomerular diseases. Citation: Yanagawa H, Suzuki H, Suzuki Y, Kiryluk K, Gharavi AG, et al. (2014) A Panel of Serum Biomarkers Differentiates IgA Nephropathy from Other Renal Diseases. PLoS ONE 9(5): e doi: /journal.pone Editor: Sunil K Ahuja, South Texas Veterans Health Care System and University Health Science Center San Antonio, United States of America Received January 6, 2014; Accepted April 28, 2014; Published May 23, 2014 Copyright: ß 2014 Yanagawa et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. Funding: This study was supported, in part, by a research grant from the Study Group on IgA Nephropathy, a Grant-in-Aid for Progressive Renal Disease Research, Research on Intractable Disease from the Ministry of Health, Labour and Welfare of Japan, and by KAKENHI ( ). BAJ and JN were supported, in part, by National Institutes of Health grants DK078244, DK082753, DK083663, DK075868, and GM KK was supported by the Glomerular Center at Columbia University and the National Institute of Health grant K23DK and R03-DK The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript. Competing Interests: The authors have declared that no competing interests exist. * yasu@juntendo.ac.jp. These authors contributed equally to this work. Introduction IgA nephropathy (IgAN) is the most common type of primary glomerulonephritis worldwide [1,2]. IgAN has a significant morbidity, culminating in end-stage kidney disease in about 40% of patients within 20 years of diagnosis [3]. Renal biopsy is required for the diagnosis of IgAN. Typical histological features include granular mesangial deposits of IgA, usually accompanied by C3, a variable presence of IgG and/or IgM, and diverse degrees of mesangial cellular proliferation and expansion of the extracellular matrix [4]. Several recent studies suggest that aberrant O-glycosylation of circulatory IgA1 is vital in the pathogenesis of IgAN. The O-linked glycans in the hinge region of IgA1 are generally composed of N- acetylgalactosamine (GalNAc) and galactose; sialic acid may be attached to either or both sugars. IgA1-producing cells secrete a mixture of IgA1 O-glycofoms. Studies in different populations have shown that IgAN patients have significantly higher levels of circulating IgA1 with galactose-deficient, O-linked, hinge-region glycans [5 9]. Depending on the population studied, 50 75% of IgAN patients have levels above the 90 th percentile for healthy controls. In addition, IgA1 eluted from renal tissues of IgAN patients also exhibits a galactose deficiency in the O-linked glycans in the hinge-region [10,11]. The serum level of IgA1-containing circulating immune complexes is elevated in patients with IgAN [12 14]. These PLOS ONE 1 May 2014 Volume 9 Issue 5 e98081
2 complexes contain galactose-deficient IgA1 (Gd-IgA1) bound by IgG and/or IgA antibodies [14,15]. Recently, we have shown that the IgG auto-antibodies that recognize glycan-containing epitopes on Gd-IgA1 exhibit unique features in the complementaritydetermining region 3 of the variable region of their heavy chains [16]. Furthermore, the serum levels of IgG autoantibodies specific for Gd-IgA1 correlated with disease severity, as assessed by magnitude of proteinuria. However, the serum levels of Gd-IgA1- containing circulating immune complexes may differ widely among IgAN patients [15]. Furthermore, some IgAN patients do not show glomerular deposition of IgG, but rather only IgA. Therefore, it is difficult to explain the pathogenesis of IgAN by an elevated serum level of glycan-specific antibodies of only the IgG isotype. These latter features may be explained by our observation that some patients with IgAN have complexes generated by glycan-specific antibodies of the IgA1 isotype [15]. Whereas the serum levels of IgA, Gd-IgA1 and glycan-specific IgG are higher in patients with IgAN compared to healthy controls, the levels of these parameters have not been systematically studied in patients with other forms renal disease with clinical features similar to those of IgAN. We therefore examined the prevalence of elevated serum levels of IgA, Gd-IgA1 and glycan-specific IgG and IgA in IgAN patients and a large cohort of CKD controls to assess the utility of these biomarkers for the non-invasive diagnosis of IgAN. Our data revealed that this panel of biomarkers is helpful in differentiating patients with IgAN from patients with other glomerular diseases. Materials and Methods Ethics Statement This study was performed according to the Declaration of Helsinki and approved by the Ethics Review Committee of Juntendo University Faculty of Medicine. All study participants provided written informed consent. Patients and controls A cross-sectional study was performed using serum samples collected at Juntendo University Hospital in Japan from 2006 to 2010 at the time of renal biopsy from 135 patients with IgAN and 79 patients with other renal diseases as shown in Table 1. We collected serum samples from 106 healthy volunteers who had never exhibited any abnormality by urinalysis in medical examinations from 2009 to All patients and healthy volunteers were Japanese, and the demographic and clinical features are summarized in Table 2. Histological prognostic stages of IgAN were assessed by the Clinical Guidelines for IgA Nephropathy in Japan (second version) [17], and are as follows: Group I: good prognosis, Group II: relatively good prognosis, Group III: relatively poor prognosis, and Group IV: poor prognosis. Subgroups of disease controls As the urinary protein/creatinine ratio in disease controls was higher than that in IgAN patients, we divided CKD controls into two subgroups: one with urinary protein/creatinine ratio $2.5 g/ g (high-proteinuria) and the second with urinary protein/ creatinine ratio,2.5 g/g (low-proteinuria). Then, we compared the levels of each biomarker between the two subgroups. Measurement of serum biomarkers Serum level of Gd-IgA1. The serum level of Gd-IgA1 was measured by lectin ELISA using GalNAc-specific lectin from Helix aspersa (HAA; Sigma, St. Louis, MO) as previously reported [9,18,19]. Diluted sera were added 100 ng per well of serum IgA. The captured IgA was treated with 10 mu/ml neuraminidase (Roche Diagnostic Corp. Indianapolis, IN) to remove terminal sialic acid residues [9,19]. The desialylated IgA1 was then reacted with biotin-labeled HAA and subsequently developed; absorbance was measured at 490 nm. The HAA reactivity of IgA1 in each sample was then calculated as OD units/100 ng of serum IgA. Naturally galactose-deficient IgA1 (Ale) myeloma protein [9] treated with neuraminidase and was used as the standard. Serum level of total Gd-IgA1 was expressed in relative Units, calculated by multiplying the normalized HAA reactivity by the amount of IgA in the serum sample (mg/ml). Serum level of Gd-IgA1-specific IgG. ELISA plates were coated with the Fab fragment of Gd-IgA1 myeloma protein (Ste) generated with an IgA-specific protease from Haemophilus influenzae HK50 [15]. The amount of total IgG used for the analyses was normalized in all samples and added to each well. Captured IgG Table 1. Renal diseases in chronic kidney disease (CKD) controls. Renal disease N Diabetic nephropathy 33 Membranous nephropathy 9 Non-IgA proliferative nephropathy 9 Lupus nephritis 8 Minimal change nephrotic syndrome 6 Nephrosclerosis 4 Interstitial nephritis 2 Focal segmental glomerular sclerosis 2 ANCA-related nephritis 1 Scleroderma renal crisis 1 HCV-related nephritis 1 Fabry s disease 1 Acute post-streptococcal nephritis 1 Minor glomerular abnormality 1 doi: /journal.pone t001 PLOS ONE 2 May 2014 Volume 9 Issue 5 e98081
3 Table 2. Characteristics of patients with IgAN and CKD controls. IgAN patients CKD controls Healthy controls Numbers Age (years) Sex (male/female) 56/79 42/37 61/45 Serum creatinine (mg/dl) ND egfr (ml/min/1.73 m 2 ) ND Urine P/C (g/g) ND Hematuria ND Values are mean 6SD. egfr, estimated glomerular filtration rate; P/C, protein/creatinine ratio; ND, not determined. Hematuria: Assessed by assigning scores according to number of red blood cells per high-power field (RBC/HPF). #5 RBC/HPF = 0, 6 10 RBC/HPF = 1, RBC/HPF = 2, RBC/HPF = 3, RBC/HPF = 4, RBC/HPF = 5,.30 RBC/HPF = 6. doi: /journal.pone t002 was detected with a biotin-labeled F(ab ) 2 fragment of goat IgG anti-human IgG antibody (BioSource; Invitrogen, San Diego, CA). Avidin horseradish peroxidase conjugate (ExtrAvidin; Sigma- Aldrich) was then added, and the reaction was developed [16]. Serum levels of Gd-IgA1-specific IgG were expressed in Units (1 unit as OD 1.0 measured at 490 nm). Serum level of Gd-IgA1-specific IgA. ELISA plates were coated with Fab fragment of Gd-IgA1 (Ste) described above [15]. Captured antibodies were detected by incubation with mouse monoclonal antibody to human IgA (Fc-specific) (Applied Biological Materials Inc., Richmond, BC) and detected by Peroxidaseconjugated AffiniPure Goat Anti-Mouse IgG (H+L) (Jackson Immuno Research, West Grove, PA). Serum levels of Gd-IgA1- specific IgA were expressed in Units (1 unit as OD 1.0 measured at 490 nm). Statistical analysis Data are expressed as means 6 SD. Comparison of groups was performed using univariate ANOVA, and post hoc Bonferroni correction was used for multiple comparisons. Correlation between two groups was performed by regression analysis. P, 0.05 was considered significant. These statistical analyses were performed using the Prism software (GraphPad Software Inc., La Jolla, CA). The discriminatory ability of the biomarkers and clinical data combination was estimated with the area under the receiver operating characteristic curve (AUC-ROC). We additionally used Akaike s Information Criterion (AIC) to select the best predictive model [20]. The sensitivity, specificity, and positive and negative predictive values were calculated based on the ROC curves with the cut-off selected at the 90 th percentile biomarker value for healthy controls. We interpreted the model with the lowest AIC as the most useful in differentiating cases from controls. These analyses were performed with IBM SPSS Statistics release Results Clinical profiles, serum IgA, IgG and Gd-IgA1 The present study was comprised of 135 IgAN patients, 79 CKD controls and 106 healthy controls (Table 2). Age, serum creatinine concentration, and the urinary protein/creatinine ratio were higher for the CKD controls than for IgAN patients. The serum IgA concentration was significantly higher for the IgAN patients ( mg/ml) compared to those of the CKD controls ( mg/ml, P,0.001) and healthy controls ( mg/ml, P,0.001). Half of the patients with IgAN had a serum IgA concentration higher than the 90 th percentile for healthy controls (3.06 mg/ml), consistent with a prior publication [21]. There was no significant difference in serum IgG concentration between the different groups. Also consistent with prior reports, the serum level of Gd-IgA1 for the IgAN patients was significantly higher compared to that for the CKD controls (P,0.001) and healthy controls (P,0.001) (Figure 1A). Fifty-six of 135 IgAN patients (41%) but only eight nineof 79 (11%) CKD controls had a serum Gd-IgA1 level higher than the 90 th percentile for healthy controls (P = for differences in distribution in IgAN patients vs. CKD controls). Serum levels of IgG and IgA antibodies against Gd-IgA1 IgAN patients had significantly higher levels of serum Gd-IgA1- specific IgG compared with those of the CKD controls (P,0.001) and healthy controls (P,0.001) (Figure 1B). Most IgAN patients (123/135, 91%) had a serum level of Gd-IgA1-specific IgG higher than the 90 th percentile for healthy controls (1.48 Units). The differences in the distribution of Gd-IgA1-specific IgG in IgAN patients vs. disease controls were highly significant (P = ). Serum levels of Gd-IgA1-specific IgA were elevated in IgAN patients compared with CKD controls (P,0.001) and healthy controls (P,0.001) ( Units for IgAN patients, Units for CKD controls and Units for healthy controls; Figure 1C). The serum level of Gd-IgA1-specific IgA was higher than the 90 th percentile for healthy controls (0.611 Units) for 43% of IgAN patients and 14% of CKD controls. While the serum levels of Gd-IgA1-specific IgG or IgA were significantly higher in IgAN patients, about 25% of CKD controls also had a serum level of Gd-IgA1-specific IgG higher than the 90 th percentile for healthy controls. Many of IgAN patients (56/ 135, 41%) had elevated levels of both Gd-IgA1-specific IgG and IgA. In contrast, elevated levels of both IgG and IgA antibody against Gd-IgA1 were seen in only 5 of 79 CKD controls who had an immune- mediated glomerulonephritis, such as lupus nephritis or membranous nephropathy (Table 3). Twenty-three of 38 CKD controls with an immune-mediated kidney disease had an elevated serum level of Gd-IgA1-specific IgG or IgA. In contrast, only 8 of 41 CKD controls with non-immune-mediated renal disease had an elevated serum level of Gd-IgA1-specific IgG or IgA (p = for differences in distribution between the two groups). PLOS ONE 3 May 2014 Volume 9 Issue 5 e98081
4 Figure 1. Distribution of serum levels of (A) Gd-IgA1, (B) Gd-IgA1-specific IgG and (C) Gd-IgA1-specific IgA in patients with IgAN (n = 135), CKD controls (n = 79) and healthy controls (n = 106). Each biomarker was measured by capture ELISA. The serum levels of Gd-IgA, Gd-IgA1-specific IgG and Gd-IgA1-specific IgA were higher in IgAN patients compared with those of the CKD controls (*P,0.001) and healthy controls (**P,0.0001). doi: /journal.pone g001 Relationship between Gd-IgA1 and Gd-IgA1-specific IgG The serum levels of Gd-IgA1-specific IgG were significantly higher in IgAN patients with elevated Gd-IgA1 levels (P,0.001). However, about 91% of IgAN patients with a normal serum Gd- IgA1 level also had an elevated level of Gd-IgA1-specific IgG, suggesting that auto-antibody production may occur independently of serum Gd-IgA1 levels (Figure 2). Comparison of biomarker levels with clinical and pathological findings We assessed each biomarker for possible correlation with histological findings and the clinical manifestations at the time of renal biopsy. Proteinuria was inversely correlated with serum immunoglobulin levels (IgA, and Gd-IgA1-specific IgG and IgA autoantibodies) in CKD controls (r = to 20.28). This Table 3. Serum levels of Gd-IgA1-specific antibodies in patients with IgAN, CKD controls and healthy controls. Gd-IgA1-specific IgG (Units) Gd-IgA1-specific IgA (Units) IgAN (N = 135) Lupus nephritis WHO class III (N = 2) Lupus nephritis WHO class IV (N = 4) Lupus nephritis WHO class V (N = 2) Membranous nephropathy (N = 9) Non-IgA proliferative glomerulonephritis (N = 9) Diabetic nephropathy (N = 33) Nephrosclerosis (N = 4) Minimal-change nephrotic syndrome (N = 6) Interstitial nephritis (N = 2) Other immune mediated renal diseases (N = 6) Other non-immune mediated renal diseases (N = 2) Healthy controls (N = 106) WHO, World Health Organization. doi: /journal.pone t003 PLOS ONE 4 May 2014 Volume 9 Issue 5 e98081
5 Figure 2. Serum levels of Gd-IgA1-specific IgG in IgAN patients with high-gd-iga1 or normal-gd-iga1 in comparison to CKD and healthy controls. We divided IgAN patients into two subgroups: patients with serum levels of Gd-IgA1 $ the 90 th percentile for healthy controls (high-gd-iga1 group; n = 56) and patients with levels Gd-IgA1, the 90 th percentile for healthy controls (normal-gd-iga1 group; n = 79). Although serum levels of Gd-IgA1-specific IgG were significantly higher in IgAN patients with high Gd-IgA1 levels (vs. CKD controls; *P, , vs. healthy controls; **P,0.0001), IgAN patients with normal Gd- IgA1 levels also had elevated Gd-IgA1-specific IgG (vs. CKD controls; *P,0.0001, vs. healthy controls; **P,0.0001). doi: /journal.pone g002 association was most strongly driven by the CKD controls, who had significantly more proteinuria than did the IgAN patients. To assess whether the serum levels of these immunogloubulins differed based on the magnitude of proteinuria, we divided the CKD disease controls into high-proteinuria and low-proteinuria subgroups, and compared the levels of each biomarker between the two subgroups. There was no significant difference in serum levels of IgA, Gd-IgA1, Gd-IgA1-specific IgG, and Gd-IgA1-specific IgA between the two subgroups (Table S1). The serum level of Gd-IgA1-specific IgA correlated with degree of mesangial IgA deposits in patients with IgAN (R 2 = 0.61) (Figure 3A). In addition, the histological prognostic stage according to the clinical guidelines for IgAN in Japan [17] correlated with proteinuria (R 2 = 0.71) (Figure 3B) and the percentage of glomeruli with crescents (R 2 = 0.69) (Figure 3C). As we previously reported [16], there was no significant correlation between the serum levels of Gd-IgA1-specific IgG and the degree of mesangial IgG deposition. However, there was a trend for the serum levels of Gd-IgA1-specific IgG to be higher in patients with greater mesangial IgG deposition (2+) ( Units, n = 9) than in patients with no mesangial IgG deposition ( Units, n = 13). Furthermore, the mean serum level of Gd-IgA1-specific IgG in the histological prognostic group 4 (n = 35, Units) was high, compared to that in the histological prognostic group 1 (n = 8, Units). Utility of biomarkers for non-invasive diagnosis of IgAN We estimated the sensitivity and specificity of each biomarker for prediction of IgAN, using the cut-off value of the 90 th percentile for healthy controls. The serum level of Gd-IgA1- specific IgG performed best, providing a sensitivity of 89% and specificity of 92% for diagnosis of IgAN. The ROC curves showed excellent performance in comparison of IgAN vs. healthy controls (Figure 4, with corresponding statistics summarized in Tables 4 and 5). The areas under the ROC curve (C-statistics) for anti-gd- IgA1 IgG were (95%CI: ) for discriminating IgAN cases vs. healthy controls, (95%CI: ) for non-immune-mediated CKD controls, and (95%CI: ) for immune-mediated CKD controls. In addition, the AIC for models testing Gd-IgA1-specific IgG were 57.4 for discriminating IgAN cases vs. healthy controls, 75.3 for non-immunemediated CKD controls, and for immune-mediated CKD controls. These data suggest that although serum levels of Gd-IgA1- specific autoantibodies have excellent predictive properties to discriminate IgAN patients from healthy controls and patients with non-immune-mediated kidney disease, the specificity is lower for discriminating IgAN patients from patients with other types of immune-mediated kidney disease. Altogether, elevated Gd-IgA1- specific IgG provided positive predictive values of 92%, 96%, and 72%, and negative predictive values of 89%, 90%, and 82% for healthy controls and patients with non-immune kidney disease, and immune-mediated kidney disease, respectively. Discussion IgAN is the most common type of primary glomerulonephritis worldwide and is often detected by urinary abnormalities, such as microscopic hematuria and low-grade proteinuria. The clinical features of IgAN vary between patients with IgAN. In biopsy specimens, glomerular deposits of IgA1 are sometimes accompanied by IgG and/or IgM. Although microscopic hematuria and low-grade proteinuria are frequent laboratory findings in patients with IgAN, some cases manifest a rapidly progressive clinical course with crescentic glomerulonephritis while others exhibit acute nephritic syndrome or nephrotic syndrome. The long-term prognosis is not benign; about 40% of IgAN patients progress to end-stage kidney disease within 20 years [3]. Thus, early diagnosis and treatment are necessary to prevent or slow disease progression. A noninvasive approach for the diagnosis of IgAN has been sought worldwide, especially in countries where the routine immunohistological processing of renal biopsy tissue is prohibitively expensive. A pathway of multiple events has been proposed for the pathogenesis of IgAN [16,22]. Immune complexes formed by the binding of Gd-IgA1 by Gd-IgA1-specific IgG or IgA in the circulation deposit in the mesangium and activate resident cells [12,23 26]. Gd-IgA1-containing immune complexes from sera of patients with IgAN induce proliferation of human mesangial cells in culture whereas Gd-IgA1 alone does not [27,28]. Activated mesangial cells produce inflammatory cytokines that cause glomerular injury [29,30]. These reports suggest that Gd-IgA1- containing immune complexes play an essential role in the pathogenesis of IgAN. Therefore, we measured serum levels of the key biomarkers, IgA, Gd-IgA1, Gd-IgA1-specific IgG and Gd- IgA1-specific IgA to assess their utility in distinguishing patients with IgAN from patients with other forms of kidney disease and healthy controls. The present study showed that serum levels of IgA, Gd-IgA1, Gd-IgA1-specific IgG and Gd-IgA1-specific IgA were elevated in patients with IgAN compared with those of healthy and CKD controls, suggesting that these parameters may be useful for the diagnosis of IgAN. However, several interesting limitations of these tests have emerged. First, elevated serum levels of Gd-IgA1 have been found in healthy relatives of individuals with IgAN, suggesting that such levels alone are not sufficient to cause the disease [31]. Among patients with an established diagnosis of IgAN, the serum levels of Gd-IgA1 are variable [9]. Notably, longitudinal trajectories of serum Gd-IgA1 levels have not yet been established in prospective PLOS ONE 5 May 2014 Volume 9 Issue 5 e98081
6 Figure 3. Correlation between biomarkers, histological findings and clinical findings. The strength of correlation between biomarkers, histological findings and clinical findings was measured by the Spearman s correlation coefficient. The serum level of Gd-IgA1-specific IgA correlated with the amount of mesangial IgA deposits (A). Histological prognostic stage (Clinical Guidelines for IgA Nephropathy in Japan, second version) [17] correlated with the urinary protein/creatinine ratio (B), and percentage of glomeruli with a crescent (C). doi: /journal.pone g003 Figure 4. Receiver operating characteristic (ROC) curves. (A) Discrimination between IgAN versus healthy and CKD controls for serum Gd-IgA1 and Gd-IgA1-specific IgG levels; (B) Discrimination between IgAN versus CKD controls with immune-mediated renal disease and non-immunemediated renal disease for serum Gd-IgA1-specific IgG levels. doi: /journal.pone g004 PLOS ONE 6 May 2014 Volume 9 Issue 5 e98081
7 Table 4. Statistics summarized data; Discrimination between IgAN versus healthy and CKD controls for serum Gd-IgA1 and Gd- IgA1-specific IgG levels. Area under the ROC curve (C-statistic) Asymptotic 95% Confidence Interval for the C-statistic Asymptotic Significance (P-value) AIC*** Lower Bound Upper Bound Gd-IgA1-specific IgG (IgAN vs. healthy controls) E Gd-IgA1-specific IgG (IgAN vs. CKD controls) E Gd-IgA1-specific IgG (IgAN vs. CKD non-immunemediated E renal disease*) Gd-IgA1-specific IgG (IgAN vs. CKD immunemediated E renal disease**) Gd-IgA1 (IgAN vs. healthy controls) E Gd-IgA1 (IgAN vs. CKD controls) E Gd-IgA1-specific IgA (IgAN vs. healthy controls) E Gd-IgA1-specific IgA (IgAN vs. CKD controls) E * CKD non-immune-mediated renal disease includes diabetic nephropathy, nephrosclerosis, interstitial nephritis and Fabry s disease. ** CKD immune-mediated renal disease includes lupus nephritis, membranous nephropathy, minimal change disease, membranoproliferative glomerulonephritis, other types of non-igan glomerulonephritis. ***AIC: Akaike s Information Criterion. doi: /journal.pone t004 cohorts of IgAN cases, and some variability may also be expected when the diagnosis (the time of renal biopsy and serum sampling) occurs at different stages of the disease process. Second, in this study, we frequently observed elevated levels of Gd-IgA1-specific autoantibodies in IgAN patients with normal serum Gd-IgA1 levels. One potential explanation of these data is that even when the circulating level of Gd-IgA1 is normal, there may be a sufficient number of galactose-deficient residues on these molecules to induce formation of pathogenic immune complexes in the setting of elevated Gd-IgA1-specific antibodies. Furthermore, because the O-glycosylation defects may occur preferentially at specific amino acid positions, [32] and the lectin-based assay is not able to discriminate which sites are involved, some pathogenic glycosylation defects may exist below the detection level of our lectin-based method. We also found that serum levels of Gd-IgA1-specific IgA correlated with the intensity of mesangial IgA1 deposits, implying that Gd-IgA1-specific IgA plays a role in glomerular deposition of IgA1-containing immune complexes. Lastly, serum levels of Gd- IgA1-specific IgG associated with histological grading, and we detected a trend for higher serum levels of Gd-IgA1-specific IgG in IgAN patients with at least moderate proteinuria ($1.0 g/g), when compared to patients with less proteinuria. These data suggest that Gd-IgA1-specific IgG may represent a marker of disease severity, but its prognostic utility will require validation in independent prospective cohorts. We noted a substantial overlap in serum levels of individual biomarkers between patients with IgAN, CKD controls, and healthy controls. Consequently, no single biomarker was sufficiently specific for IgAN. The levels of Gd-IgA1-specific IgG and IgA were particularly elevated in patients with non-igan immunemediated kidney disease, such as lupus nephritis in which increased propensity to auto-antibody production is well established. Notably, some of these autoantibodies may be polyreactive, i.e., binding to several autoantigens [33,34], thus complicating the assay for IgAN. Our data raise a question as to whether the anti- Gd-IgA1 autoantibodies in patients with lupus nephritis are detected as an artifact of polyreactivity or constitute pathogenic mediators of kidney injury in non-igan kidney disease. Additional studies are necessary to examine this aspect of non-igan glomerular diseases. Another limitation of our study was that the CKD control group did not contain sufficient numbers of patients with glomerular disorders that are clinically difficult to differentiate from IgAN, such as Alport syndrome, thin basement membrane disease, postinfectious glomerulonephritis or membranoproliferative glomeru- Table 5. Statistics summarized data; Discrimination between IgAN versus immune-mediated CKD and non-immune-mediated CKD controls for serum Gd-IgA1-specific IgG levels. Sensitivity Specificity PPV NPV Gd-IgA1-specific IgG (IgAN vs. healthy controls) 89% 92% 92% 89% Gd-IgA1-specific IgG (IgAN vs. all CKD controls) 89% 81% 82% 88% Gd-IgA1-specific IgG (IgAN vs. non-immune-mediated CKD) 89% 96% 96% 90% Gd-IgA1-specific IgG (IgAN vs. immune-mediated CKD) 85% 67% 72% 82% Gd-IgA1 (IgAN vs. healthy controls) 41% 91% 82% 61% Gd-IgA1-specific IgA (IgAN vs. healthy controls) 34% 92% 81% 58% PPV, Positive Predictive Value; NPV, Negative Predictive Value. doi: /journal.pone t005 PLOS ONE 7 May 2014 Volume 9 Issue 5 e98081
8 lonephritis. Because of the relative rarity of these conditions, large multicenter studies will be required to validate our results in these settings. Conclusions Serum levels of Gd-IgA1-specific antibodies are elevated in most IgAN patients, even in the setting of normal serum level of Gd- IgA1. Our study suggests that a panel of serum biomarkers may be helpful in differentiating IgAN from other glomerular diseases. References 1. D Amico G (1987) The commonest glomerulonephritis in the world: IgA nephropathy. Q J Med 64: Julian BA, Waldo FB, Rifai A, Mestecky J (1988) IgA nephropathy, the most common glomerulonephritis worldwide. A neglected disease in the United States? Am J Med 84: D Amico G (2000) Natural history of idiopathic IgA nephropathy: role of clinical and histological prognostic factors. Am J Kidney Dis 36: Russell MW, Mestecky J, Julian BA, Galla JH (1986) IgA-associated renal diseases: antibodies to environmental antigens in sera and deposition of immunoglobulins and antigens in glomeruli. J Clin Immunol 6: Mestecky J, Tomana M, Crowley-Nowick PA, Moldoveanu Z, Julian BA, et al. (1993) Defective galactosylation and clearance of IgA1 molecules as a possible etiopathogenic factor in IgA nephropathy. Contrib Nephrol 104: Allen AC, Harper SJ, Feehally J (1995) Galactosylation of N- and O-linked carbohydrate moieties of IgA1 and IgG in IgA nephropathy. Clin Exp Immunol 100: Novak J, Julian BA, Tomana M, Mestecky J (2008) IgA glycosylation and IgA immune complexes in the pathogenesis of IgA nephropathy. Semin Nephrol 28: Hiki Y, Horii A, Iwase H, Tanaka A, Toda Y, et al. (1995) O-linked oligosaccharide on IgA1 hinge region in IgA nephropathy. Fundamental study for precise structure and possible role. Contrib Nephrol 111: Moldoveanu Z, Wyatt RJ, Lee JY, Tomana M, Julian BA, et al. (2007) Patients with IgA nephropathy have increased serum galactose-deficient IgA1 levels. Kidney Int 71: Hiki Y, Odani H, Takahashi M, Yasuda Y, Nishimoto A, et al. (2001) Mass spectrometry proves under-o-glycosylation of glomerular IgA1 in IgA nephropathy. Kidney Int 59: Allen AC, Bailey EM, Brenchley PE, Buck KS, Barratt J, et al. (2001) Mesangial IgA1 in IgA nephropathy exhibits aberrant O-glycosylation: observations in three patients. Kidney Int 60: Czerkinsky C, Koopman WJ, Jackson S, Collins JE, Crago SS, et al. (1986) Circulating immune complexes and immunoglobulin A rheumatoid factor in patients with mesangial immunoglobulin A nephropathies. J Clin Invest 77: Coppo R, Basolo B, Piccoli G, Mazzucco G, Bulzomì MR, et al. (1984) IgA1 and IgA2 immune complexes in primary IgA nephropathy and Henoch- Schönlein nephritis. Clin Exp Immunol 57: Tomana M, Matousovic K, Julian BA, Radl J, Konecny K, et al. (1997) Galactose-deficient IgA1 in sera of IgA nephropathy patients is present in complexes with IgG. Kidney Int 52: Tomana M, Novak J, Julian BA, Matousovic K, Konecny K, et al. (1999) Circulating immune complexes in IgA nephropathy consist of IgA1 with galactose-deficient hinge region and antiglycan antibodies. J Clin Invest 104: Suzuki H, Fan R, Zhang Z, Brown R, Hall S, et al. (2009) Aberrantly glycosylated IgA1 in IgA nephropathy patients is recognized by IgG antibodies with restricted heterogeneity. J Clin Invest 119: Supporting Information Table S1 Serum levels of biomarkers in high-proteinuria and low-proteinuria CKD control subgroups. (PDF) Author Contributions Conceived and designed the experiments: HS YT. Performed the experiments: HY HS. Analyzed the data: KM KK AGG. Contributed reagents/materials/analysis tools: BAJ JN. Wrote the paper: HY HS BAJ JN. Collected serum samples: YS. Collected clinical data: YM. 17. Tomino Y, Sakai H (2003) Special Study Group (IgA Nephropathy) on Progressive Glomerular Disease: Clinical guidelines for immunoglobulin A (IgA) nephropathy in Japan, second version. Clin Exp Nephrol 7: Moore JS, Kulhavy R, Tomana M, Moldoveanu Z, Suzuki H, et al. (2007) Reactivities of N-acetylgalactosamine-specific lectins with human IgA1 proteins. Mol Immunol 44: Suzuki H, Moldoveanu Z, Hall S, Brown R, Vu HL, et al. (2008) IgA1-secreting cell lines from patients with IgA nephropathy produce aberrantly glycosylated IgA1. J Clin Invest 118: Akaike H (1974) A new look at the statistical model identification. IEEE Trans Automat Contr 19: van der Boog PJ, van Kooten C, van Seggelen A, Mallat M, Klar-Mohamad N, et al. (2004) An increased polymeric IgA level is not a prognostic marker for progressive IgA nephropathy. Nephrol Dial Transplant 19: Suzuki H, Kiryluk K, Novak J, Moldoveanu Z, Herr AB, et al. (2011) The pathophysiology of IgA nephropathy. J Am Soc Nephrol 22: Coppo R, Amore A (2004) Aberrant glycosylation in IgA nephropathy (IgAN). Kidney Int 65: Barratt J, Feehally J (2005) IgA nephropathy. J Am Soc Nephrol 16: Julian BA, Novak J (2004) IgA nephropathy: an update. Curr Opin Nephrol Hypertens 13: Novak J, Julian BA, Tomana M, Mesteck J (2001) Progress in molecular and genetic studies of IgA nephropathy. J Clin Immunol 21: Novak J, Vu HL, Novak L, Julian BA, Mestecky J, et al. (2002) Interactions of human mesangial cells with IgA and IgA-containing immune complexes. Kidney Int 62: Novak J, Tomana M, Matousovic K, Brown R, Hall S, et al. (2005) IgA1- containing immune complexes in IgA nephropathy differentially affect proliferation of mesangial cells. Kidney Int 67: Lai KN, Leung JC, Chan LY, Saleem MA, Mathieson PW, et al. (2008) Activation of podocytes by mesangial-derived TNF-alpha: glomerulo-podocytic communication in IgA nephropathy. Am J Physiol Renal Physiol 294: F Lai KN, Leung JC, Chan LY, Saleem MA, Mathieson PW, et al. (2009) Podocyte injury induced by mesangial-derived cytokines in IgA nephropathy. Nephrol Dial Transplant 24: Gharavi AG, Moldoveanu Z, Wyatt RJ, Barker CV, Woodford SY, et al. (2008) Aberrant IgA1 glycosylation is inherited in familial and sporadic IgA nephropathy. J Am Soc Nephrol 19: Takahashi K, Wall SB, Suzuki H, Smith AD 4th, Hall S, et al. (2010) Clustered O-glycans of IgA1: defining macro- and microheterogeneity by use of electron capture/transfer dissociation. Mol Cell Proteomics 9: Zhang J, Jacobi AM, Wang T, Berlin R, Volpe BT, et al. (2009) Polyreactive autoantibodies in systemic lupus erythematosus have pathogenic potential. J Autoimmun 33: Mietzner B, Tsuiji M, Scheid J, Velinzon K, Tiller T, et al. (2008) Autoreactive IgG memory antibodies in patients with systemic lupus erythematosus arise from nonreactive and polyreactive precursors. Proc Natl Acad Sci USA 105: PLOS ONE 8 May 2014 Volume 9 Issue 5 e98081
Sugars and immune complex formation in IgA
 Glomerular disease Sugars and immune complex formation in IgA nephropathy Jonathan Barratt and Frank Eitner In vitro evidence suggests that immune complex formation in IgA nephropathy is determined by
Glomerular disease Sugars and immune complex formation in IgA nephropathy Jonathan Barratt and Frank Eitner In vitro evidence suggests that immune complex formation in IgA nephropathy is determined by
29th Annual Meeting of the Glomerular Disease Collaborative Network
 29th Annual Meeting of the Glomerular Disease Collaborative Network Updates on the Pathogenesis IgA Nephropathy and IgA Vasculitis (HSP) J. Charles Jennette, M.D. Brinkhous Distinguished Professor and
29th Annual Meeting of the Glomerular Disease Collaborative Network Updates on the Pathogenesis IgA Nephropathy and IgA Vasculitis (HSP) J. Charles Jennette, M.D. Brinkhous Distinguished Professor and
Immune complex formation in IgA nephropathy: A case of the right antibodies in the wrong place at. the wrong time?
 Immune complex formation in IgA nephropathy: A case of the right antibodies in the wrong place at the wrong time? Comment on: Suzuki H, Fan R, Zhang Z, Brown R, Hall S, Julian BA, Chatham WW, Suzuki Y,
Immune complex formation in IgA nephropathy: A case of the right antibodies in the wrong place at the wrong time? Comment on: Suzuki H, Fan R, Zhang Z, Brown R, Hall S, Julian BA, Chatham WW, Suzuki Y,
Original Article Serum galactose-deficient IgA1 levels in children with IgA nephropathy
 Int J Clin Exp Med 2015;8(5):7861-7866 www.ijcem.com /ISSN:1940-5901/IJCEM0006215 Original Article Serum galactose-deficient IgA1 levels in children with IgA nephropathy Mengjie Jiang 1*, Xiaoyun Jiang
Int J Clin Exp Med 2015;8(5):7861-7866 www.ijcem.com /ISSN:1940-5901/IJCEM0006215 Original Article Serum galactose-deficient IgA1 levels in children with IgA nephropathy Mengjie Jiang 1*, Xiaoyun Jiang
Research Article Serum Galactose-Deficient IgA1 Level Is Not Associated with Proteinuria in Children with IgA Nephropathy
 International Nephrology Volume 212, Article ID 315467, 7 pages doi:1.1155/212/315467 Research Article Serum Galactose-Deficient IgA1 Level Is Not Associated with Proteinuria in Children with IgA Nephropathy
International Nephrology Volume 212, Article ID 315467, 7 pages doi:1.1155/212/315467 Research Article Serum Galactose-Deficient IgA1 Level Is Not Associated with Proteinuria in Children with IgA Nephropathy
Analysis of regulatory T cell subsets in the peripheral blood of immunoglobulin A nephropathy (IgAN) patients
 Analysis of regulatory T cell subsets in the peripheral blood of immunoglobulin A nephropathy (IgAN) patients S. Yang, B. Chen, J. Shi, F. Chen, J. Zhang and Z. Sun Department of Nephrology, Huaihe Hospital
Analysis of regulatory T cell subsets in the peripheral blood of immunoglobulin A nephropathy (IgAN) patients S. Yang, B. Chen, J. Shi, F. Chen, J. Zhang and Z. Sun Department of Nephrology, Huaihe Hospital
A clinical syndrome, composed mainly of:
 Nephritic syndrome We will discuss: 1)Nephritic syndrome: -Acute postinfectious (poststreptococcal) GN -IgA nephropathy -Hereditary nephritis 2)Rapidly progressive GN (RPGN) A clinical syndrome, composed
Nephritic syndrome We will discuss: 1)Nephritic syndrome: -Acute postinfectious (poststreptococcal) GN -IgA nephropathy -Hereditary nephritis 2)Rapidly progressive GN (RPGN) A clinical syndrome, composed
Mass spectrometry in glycomics research: Application to IgA nephropathy
 Mass spectrometry in glycomics research: Application to IgA nephropathy Part I Jan Novak, Ph.D. and Matthew B. Renfrow, Ph.D. In: Proteomics and mass spectrometry 2007 March 9, 2007 IgA Nephropathy The
Mass spectrometry in glycomics research: Application to IgA nephropathy Part I Jan Novak, Ph.D. and Matthew B. Renfrow, Ph.D. In: Proteomics and mass spectrometry 2007 March 9, 2007 IgA Nephropathy The
Pathogenesis of IgA Nephropathy. Shokoufeh Savaj MD Associate Professor of Medicine Firoozgar hospital- IUMS
 Pathogenesis of IgA Nephropathy Shokoufeh Savaj MD Associate Professor of Medicine Firoozgar hospital- IUMS History Immunoglobin A nephropathy was first described by Berger and Hinglais in 1968 in Paris
Pathogenesis of IgA Nephropathy Shokoufeh Savaj MD Associate Professor of Medicine Firoozgar hospital- IUMS History Immunoglobin A nephropathy was first described by Berger and Hinglais in 1968 in Paris
Hemizygous Fabry disease associated with IgA nephropathy: A case report
 1 Hemizygous Fabry disease associated with IgA nephropathy: A case report Fabry disease and IgA nephropathy Homare Shimohata 1, 3, Keigyou Yoh 1, Kenji Takada 2, Hiroaki Tanaka 2, Joichi Usui 1, Kouichi
1 Hemizygous Fabry disease associated with IgA nephropathy: A case report Fabry disease and IgA nephropathy Homare Shimohata 1, 3, Keigyou Yoh 1, Kenji Takada 2, Hiroaki Tanaka 2, Joichi Usui 1, Kouichi
Mass spectrometry in glycomics research: Application to IgA nephropathy
 Mass spectrometry in glycomics research: Application to IgA nephropathy Part I Jan Novak, Ph.D. and Matthew B. Renfrow, Ph.D. In: Proteomics and mass spectrometry 2009 March 13, 2009 IgA Nephropathy The
Mass spectrometry in glycomics research: Application to IgA nephropathy Part I Jan Novak, Ph.D. and Matthew B. Renfrow, Ph.D. In: Proteomics and mass spectrometry 2009 March 13, 2009 IgA Nephropathy The
What s hiding behind IgA nephropathy?
 What s hiding behind IgA nephropathy? Bauerova L. Department of Pathology, the First Faculty of Medicine and General Hospital, Charles University Prague (nephropathology training: Department of Clinical
What s hiding behind IgA nephropathy? Bauerova L. Department of Pathology, the First Faculty of Medicine and General Hospital, Charles University Prague (nephropathology training: Department of Clinical
Biomarkers for IgA nephropathy on the basis of multi-hit pathogenesis
 https://doi.org/10.1007/s10157-018-1582-2 INVITED REVIEW ARTICLE Biomarkers for IgA nephropathy on the basis of multi-hit pathogenesis Hitoshi Suzuki 1 Received: 11 January 2018 / Accepted: 26 March 2018
https://doi.org/10.1007/s10157-018-1582-2 INVITED REVIEW ARTICLE Biomarkers for IgA nephropathy on the basis of multi-hit pathogenesis Hitoshi Suzuki 1 Received: 11 January 2018 / Accepted: 26 March 2018
Galactose-Deficient IgA1 in African Americans with IgA Nephropathy: Serum Levels and Heritability
 Galactose-Deficient IgA1 in African Americans with IgA Nephropathy: Serum Levels and Heritability M. Colleen Hastings,* Zina Moldoveanu, Bruce A. Julian, Jan Novak, John T. Sanders,* Kim R. McGlothan,
Galactose-Deficient IgA1 in African Americans with IgA Nephropathy: Serum Levels and Heritability M. Colleen Hastings,* Zina Moldoveanu, Bruce A. Julian, Jan Novak, John T. Sanders,* Kim R. McGlothan,
Does Electron Microscopy Change the View of the Diagnosis of IgA Nephropathy?
 Prague Medical Report / Vol. 106 (2005) No. 3, p. 283 290 283) Does Electron Microscopy Change the View of the Diagnosis of IgA Nephropathy? Maixnerová D. 1, Honsová E. 2, Merta M. 1, Reiterová J. 1, Ryšavá
Prague Medical Report / Vol. 106 (2005) No. 3, p. 283 290 283) Does Electron Microscopy Change the View of the Diagnosis of IgA Nephropathy? Maixnerová D. 1, Honsová E. 2, Merta M. 1, Reiterová J. 1, Ryšavá
Aberrant IgA1 Glycosylation Is Inherited in Familial and Sporadic IgA Nephropathy
 Aberrant IgA1 Glycosylation Is Inherited in Familial and Sporadic IgA Nephropathy Ali G. Gharavi,* Zina Moldoveanu, Robert J. Wyatt, Catherine V. Barker, Susan Y. Woodford, Richard P. Lifton, Jiri Mestecky,
Aberrant IgA1 Glycosylation Is Inherited in Familial and Sporadic IgA Nephropathy Ali G. Gharavi,* Zina Moldoveanu, Robert J. Wyatt, Catherine V. Barker, Susan Y. Woodford, Richard P. Lifton, Jiri Mestecky,
Glomerular pathology in systemic disease
 Glomerular pathology in systemic disease Lecture outline Lupus nephritis Diabetic nephropathy Glomerulonephritis Associated with Bacterial Endocarditis and Other Systemic Infections Henoch-Schonlein Purpura
Glomerular pathology in systemic disease Lecture outline Lupus nephritis Diabetic nephropathy Glomerulonephritis Associated with Bacterial Endocarditis and Other Systemic Infections Henoch-Schonlein Purpura
premature IgA1 sialylation hold the key to the pathogenic changes in
 Is sialylation of IgA the agent provocateur of IgA nephropathy?does Formatted: Font: Italic premature IgA1 sialylation hold the key to the pathogenic changes in IgA1 O-glycosylation in IgAN? Alice Smith,
Is sialylation of IgA the agent provocateur of IgA nephropathy?does Formatted: Font: Italic premature IgA1 sialylation hold the key to the pathogenic changes in IgA1 O-glycosylation in IgAN? Alice Smith,
C1q nephropathy the Diverse Disease
 C1q nephropathy the Diverse Disease Danica Galešić Ljubanović School of Medicine, University of Zagreb Dubrava University Hospital Zagreb, Croatia Definition Dominant or codominant ( 2+), mesangial staining
C1q nephropathy the Diverse Disease Danica Galešić Ljubanović School of Medicine, University of Zagreb Dubrava University Hospital Zagreb, Croatia Definition Dominant or codominant ( 2+), mesangial staining
Dr Ian Roberts Oxford. Oxford Pathology Course 2010 for FRCPath Illustration-Cellular Pathology. Oxford Radcliffe NHS Trust
 Dr Ian Roberts Oxford Oxford Pathology Course 2010 for FRCPath Present the basic diagnostic features of the commonest conditions causing proteinuria & haematuria Highlight diagnostic pitfalls Nephrotic
Dr Ian Roberts Oxford Oxford Pathology Course 2010 for FRCPath Present the basic diagnostic features of the commonest conditions causing proteinuria & haematuria Highlight diagnostic pitfalls Nephrotic
Atypical IgA Nephropathy
 Atypical IgA Nephropathy Richard J. Glassock, MD, MACP Geffen School of Medicine at UCLA XXXIII Chilean Congress of Nephrology, Hypertension and Transplantation Puerto Varas, Chile October 6, 2016 IgA
Atypical IgA Nephropathy Richard J. Glassock, MD, MACP Geffen School of Medicine at UCLA XXXIII Chilean Congress of Nephrology, Hypertension and Transplantation Puerto Varas, Chile October 6, 2016 IgA
Overview of glomerular diseases
 Overview of glomerular diseases *Endothelial cells are fenestrated each fenestra: 70-100nm in diameter Contractile, capable of proliferation, makes ECM & releases mediators *Glomerular basement membrane
Overview of glomerular diseases *Endothelial cells are fenestrated each fenestra: 70-100nm in diameter Contractile, capable of proliferation, makes ECM & releases mediators *Glomerular basement membrane
Renal Pathology 1: Glomerulus. With many thanks to Elizabeth Angus PhD for EM photographs
 Renal Pathology 1: Glomerulus With many thanks to Elizabeth Angus PhD for EM photographs Anatomy of the Kidney http://www.yalemedicalgroup.org/stw/page.asp?pageid=stw028980 The Nephron http://www.beltina.org/health-dictionary/nephron-function-kidney-definition.html
Renal Pathology 1: Glomerulus With many thanks to Elizabeth Angus PhD for EM photographs Anatomy of the Kidney http://www.yalemedicalgroup.org/stw/page.asp?pageid=stw028980 The Nephron http://www.beltina.org/health-dictionary/nephron-function-kidney-definition.html
Immune profile of IgA-dominant diffuse proliferative glomerulonephritis
 Clin Kidney J (2014) 7: 479 483 doi: 10.1093/ckj/sfu090 Exceptional Case Immune profile of IgA-dominant diffuse proliferative glomerulonephritis Eric Wallace 1, Nicolas Maillard 2, Hiroyuki Ueda 2, Stacy
Clin Kidney J (2014) 7: 479 483 doi: 10.1093/ckj/sfu090 Exceptional Case Immune profile of IgA-dominant diffuse proliferative glomerulonephritis Eric Wallace 1, Nicolas Maillard 2, Hiroyuki Ueda 2, Stacy
Case Presentation Turki Al-Hussain, MD
 Case Presentation Turki Al-Hussain, MD Director, Renal Pathology Chapter Saudi Society of Nephrology & Transplantation Consultant Nephropathologist & Urological Pathologist Department of Pathology & Laboratory
Case Presentation Turki Al-Hussain, MD Director, Renal Pathology Chapter Saudi Society of Nephrology & Transplantation Consultant Nephropathologist & Urological Pathologist Department of Pathology & Laboratory
Glomerular diseases mostly presenting with Nephritic syndrome
 Glomerular diseases mostly presenting with Nephritic syndrome 1 The Nephritic Syndrome Pathogenesis: proliferation of the cells in glomeruli & leukocytic infiltrate Injured capillary walls escape of RBCs
Glomerular diseases mostly presenting with Nephritic syndrome 1 The Nephritic Syndrome Pathogenesis: proliferation of the cells in glomeruli & leukocytic infiltrate Injured capillary walls escape of RBCs
Secondary IgA Nephropathy & HSP
 Secondary IgA Nephropathy & HSP Anjali Gupta, MD 1/11/11 AKI sec to Hematuria? 65 cases of ARF after an episode of macroscopic hematuria have been reported in the literature in patients with GN. The main
Secondary IgA Nephropathy & HSP Anjali Gupta, MD 1/11/11 AKI sec to Hematuria? 65 cases of ARF after an episode of macroscopic hematuria have been reported in the literature in patients with GN. The main
Relationship between Serum IgA/C3 Ratio and Progression of IgA Nephropathy
 ORIGINAL ARTICLE Relationship between Serum IgA/C3 Ratio and Progression of IgA Nephropathy Hiroyuki KOMATSU, Shouichi FUJIMOTO, Seiichiro HARA, Yuji SATO, Kazuhiro YAMADA and Tanenao ETO Abstract Objective
ORIGINAL ARTICLE Relationship between Serum IgA/C3 Ratio and Progression of IgA Nephropathy Hiroyuki KOMATSU, Shouichi FUJIMOTO, Seiichiro HARA, Yuji SATO, Kazuhiro YAMADA and Tanenao ETO Abstract Objective
CHAPTER 2. Primary Glomerulonephritis
 2nd Report of the PRIMARY GLOMERULONEPHRITIS CHAPTER 2 Primary Glomerulonephritis Sunita Bavanandan Lee Han Wei Lim Soo Kun 21 PRIMARY GLOMERULONEPHRITIS 2nd Report of the 2.1 Introduction This chapter
2nd Report of the PRIMARY GLOMERULONEPHRITIS CHAPTER 2 Primary Glomerulonephritis Sunita Bavanandan Lee Han Wei Lim Soo Kun 21 PRIMARY GLOMERULONEPHRITIS 2nd Report of the 2.1 Introduction This chapter
Dr. Ghadeer Mokhtar Consultant pathologists and nephropathologist, KAU
 Dr. Ghadeer Mokhtar Consultant pathologists and nephropathologist, KAU CLINICAL HISTORY A 4 year old Saudi girl presented to the ER with generalized body swelling, decrease urine output with passing dark
Dr. Ghadeer Mokhtar Consultant pathologists and nephropathologist, KAU CLINICAL HISTORY A 4 year old Saudi girl presented to the ER with generalized body swelling, decrease urine output with passing dark
Nephrology Grand Rounds. Mansi Mehta November 24, 2015
 Nephrology Grand Rounds Mansi Mehta November 24, 2015 Case 51yo F with PMH significant for Hypertension referred to renal clinic for evaluation of elevated Cr. no known history of CKD; baseline creatinine
Nephrology Grand Rounds Mansi Mehta November 24, 2015 Case 51yo F with PMH significant for Hypertension referred to renal clinic for evaluation of elevated Cr. no known history of CKD; baseline creatinine
The CARI Guidelines Caring for Australasians with Renal Impairment. Specific management of IgA nephropathy: role of steroid therapy GUIDELINES
 Specific management of IgA nephropathy: role of steroid therapy Date written: July 2005 Final submission: September 2005 Author: Merlin Thomas GUIDELINES Steroid therapy may protect against progressive
Specific management of IgA nephropathy: role of steroid therapy Date written: July 2005 Final submission: September 2005 Author: Merlin Thomas GUIDELINES Steroid therapy may protect against progressive
Histopathology: Glomerulonephritis and other renal pathology
 Histopathology: Glomerulonephritis and other renal pathology These presentations are to help you identify basic histopathological features. They do not contain the additional factual information that you
Histopathology: Glomerulonephritis and other renal pathology These presentations are to help you identify basic histopathological features. They do not contain the additional factual information that you
Case Presentation Turki Al-Hussain, MD
 Case Presentation Turki Al-Hussain, MD Director, Renal Pathology Chapter Saudi Society of Nephrology & Transplantation Consultant Nephropathologist & Urological Pathologist Department of Pathology & Laboratory
Case Presentation Turki Al-Hussain, MD Director, Renal Pathology Chapter Saudi Society of Nephrology & Transplantation Consultant Nephropathologist & Urological Pathologist Department of Pathology & Laboratory
Clinicopathologic Characteristics of IgA Nephropathy with Steroid-responsive Nephrotic Syndrome
 J Korean Med Sci 2009; 24 (Suppl 1): S44-9 ISSN 1011-8934 DOI: 10.3346/jkms.2009.24.S1.S44 Copyright The Korean Academy of Medical Sciences Clinicopathologic Characteristics of IgA Nephropathy with Steroid-responsive
J Korean Med Sci 2009; 24 (Suppl 1): S44-9 ISSN 1011-8934 DOI: 10.3346/jkms.2009.24.S1.S44 Copyright The Korean Academy of Medical Sciences Clinicopathologic Characteristics of IgA Nephropathy with Steroid-responsive
RENAL HISTOPATHOLOGY
 RENAL HISTOPATHOLOGY Peter McCue, M.D. Department of Pathology, Anatomy & Cell Biology Sidney Kimmel Medical College There are no conflicts of interest. 1 Goals and Objectives! Goals Provide introduction
RENAL HISTOPATHOLOGY Peter McCue, M.D. Department of Pathology, Anatomy & Cell Biology Sidney Kimmel Medical College There are no conflicts of interest. 1 Goals and Objectives! Goals Provide introduction
IgA nephropathy (IgAN) is one of the most common patterns
 O-Glycosylation of Serum IgA1 Antibodies against Mucosal and Systemic Antigens in IgA Nephropathy Alice C. Smith, Karen Molyneux, John Feehally, and Jonathan Barratt University of Leicester Department
O-Glycosylation of Serum IgA1 Antibodies against Mucosal and Systemic Antigens in IgA Nephropathy Alice C. Smith, Karen Molyneux, John Feehally, and Jonathan Barratt University of Leicester Department
Original. IgAN. Key words : IgA nephropathy, IgM deposition, proteinuria, tonsillectomy, steroid pulse therapy. Introduction
 Showa Univ J Med Sci 27 3, 167 174, September 2015 Original Prominent IgM Deposition in Glomerulus Is Associated with Severe Proteinuria and Reduced after Combined Treatment of Tonsillectomy with Steroid
Showa Univ J Med Sci 27 3, 167 174, September 2015 Original Prominent IgM Deposition in Glomerulus Is Associated with Severe Proteinuria and Reduced after Combined Treatment of Tonsillectomy with Steroid
Dense deposit disease with steroid pulse therapy
 Case Report Dense deposit disease with steroid pulse therapy Jun Odaka, Takahiro Kanai, Takane Ito, Takashi Saito, Jun Aoyagi, and Mariko Y Momoi Abstract Treatment of dense deposit disease DDD has not
Case Report Dense deposit disease with steroid pulse therapy Jun Odaka, Takahiro Kanai, Takane Ito, Takashi Saito, Jun Aoyagi, and Mariko Y Momoi Abstract Treatment of dense deposit disease DDD has not
Familial DDD associated with a gain-of-function mutation in complement C3.
 Familial DDD associated with a gain-of-function mutation in complement C3. Santiago Rodríguez de Córdoba, Centro de investigaciones Biológicas, Madrid Valdés Cañedo F. and Vázquez- Martul E., Complejo
Familial DDD associated with a gain-of-function mutation in complement C3. Santiago Rodríguez de Córdoba, Centro de investigaciones Biológicas, Madrid Valdés Cañedo F. and Vázquez- Martul E., Complejo
Title: A novel differential diagnostic model based on multiple biological parameters for immunoglobulin A nephropathy
 Author's response to reviews Title: A novel differential diagnostic model based on multiple biological parameters for immunoglobulin A nephropathy Authors: Nan Zhen Dong (dongzn@301hospital.com.cn) Yong
Author's response to reviews Title: A novel differential diagnostic model based on multiple biological parameters for immunoglobulin A nephropathy Authors: Nan Zhen Dong (dongzn@301hospital.com.cn) Yong
Year 2004 Paper one: Questions supplied by Megan
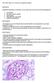 QUESTION 53 Endothelial cell pathology on renal biopsy is most characteristic of which one of the following diagnoses? A. Pre-eclampsia B. Haemolytic uraemic syndrome C. Lupus nephritis D. Immunoglobulin
QUESTION 53 Endothelial cell pathology on renal biopsy is most characteristic of which one of the following diagnoses? A. Pre-eclampsia B. Haemolytic uraemic syndrome C. Lupus nephritis D. Immunoglobulin
CHAPTER 2 PRIMARY GLOMERULONEPHRITIS
 CHAPTER 2 Sunita Bavanandan Lim Soo Kun 19 5th Report of the 2.1: Introduction This chapter covers the main primary glomerulonephritis that were reported to the MRRB from the years 2005-2012. Minimal change
CHAPTER 2 Sunita Bavanandan Lim Soo Kun 19 5th Report of the 2.1: Introduction This chapter covers the main primary glomerulonephritis that were reported to the MRRB from the years 2005-2012. Minimal change
Glomerular pathology-2 Nephritic syndrome. Dr. Nisreen Abu Shahin
 Glomerular pathology-2 Nephritic syndrome Dr. Nisreen Abu Shahin 1 The Nephritic Syndrome Pathogenesis: inflammation proliferation of the cells in glomeruli & leukocytic infiltrate Injured capillary walls
Glomerular pathology-2 Nephritic syndrome Dr. Nisreen Abu Shahin 1 The Nephritic Syndrome Pathogenesis: inflammation proliferation of the cells in glomeruli & leukocytic infiltrate Injured capillary walls
Case # 2 3/27/2017. Disclosure of Relevant Financial Relationships. Clinical history. Clinical history. Laboratory findings
 Case # 2 Christopher Larsen, MD Arkana Laboratories Disclosure of Relevant Financial Relationships USCAP requires that all planners (Education Committee) in a position to influence or control the content
Case # 2 Christopher Larsen, MD Arkana Laboratories Disclosure of Relevant Financial Relationships USCAP requires that all planners (Education Committee) in a position to influence or control the content
Monoclonal Gammopathies and the Kidney. Tibor Nádasdy, MD The Ohio State University, Columbus, OH
 Monoclonal Gammopathies and the Kidney Tibor Nádasdy, MD The Ohio State University, Columbus, OH Monoclonal gammopathy of renal significance (MGRS) Biopsies at OSU (n=475) between 2007 and 2016 AL or AH
Monoclonal Gammopathies and the Kidney Tibor Nádasdy, MD The Ohio State University, Columbus, OH Monoclonal gammopathy of renal significance (MGRS) Biopsies at OSU (n=475) between 2007 and 2016 AL or AH
Pathology of Complement Mediated Renal Disease
 Pathology of Complement Mediated Renal Disease Mariam Priya Alexander, MD Associate Professor of Pathology GN Symposium Hong Kong Society of Nephrology July 8 th, 2017 2017 MFMER slide-1 The complement
Pathology of Complement Mediated Renal Disease Mariam Priya Alexander, MD Associate Professor of Pathology GN Symposium Hong Kong Society of Nephrology July 8 th, 2017 2017 MFMER slide-1 The complement
REVIEW ARTICLE NEWER BIOMARKERS IN EARLY DIABETIC NEPHROPATHY
 JCD REVIEW ARTICLE NEWER BIOMARKERS IN EARLY DIABETIC NEPHROPATHY SAPTARSHI MUKHOPADHYAY* *FACULTY (DEPARTMENT OF MEDICINE), B R SINGH HOSPITAL (EASTERN RAILWAY), KOLKATA. INTRODUCTION renal disease. It
JCD REVIEW ARTICLE NEWER BIOMARKERS IN EARLY DIABETIC NEPHROPATHY SAPTARSHI MUKHOPADHYAY* *FACULTY (DEPARTMENT OF MEDICINE), B R SINGH HOSPITAL (EASTERN RAILWAY), KOLKATA. INTRODUCTION renal disease. It
Clinical and pathological characteristics of patients with glomerular diseases at a university teaching hospital: 5-year prospective review
 Clinical and pathological characteristics of patients with glomerular diseases at a university teaching hospital: 5-year prospective review KW Chan, TM Chan, IKP Cheng Objective. To examine the prevalence
Clinical and pathological characteristics of patients with glomerular diseases at a university teaching hospital: 5-year prospective review KW Chan, TM Chan, IKP Cheng Objective. To examine the prevalence
Pathogenesis and Treatment in IgA Nephropathy
 Pathogenesis and Treatment in IgA Nephropathy ThiS is a FM Blank Page Yasuhiko Tomino Editor Pathogenesis and Treatment in IgA Nephropathy An International Comparison Editor Yasuhiko Tomino Medical Corporation
Pathogenesis and Treatment in IgA Nephropathy ThiS is a FM Blank Page Yasuhiko Tomino Editor Pathogenesis and Treatment in IgA Nephropathy An International Comparison Editor Yasuhiko Tomino Medical Corporation
ESRD Dialysis Prevalence - One Year Statistics
 Age Group IL Other Total 00-04 12 1 13 05-09 5 2 7 10-14 15 1 16 15-19 55 2 57 20-24 170 10 180 25-29 269 14 283 30-34 381 9 390 35-39 583 14 597 40-44 871 20 891 45-49 1,119 20 1,139 50-54 1,505 35 1,540
Age Group IL Other Total 00-04 12 1 13 05-09 5 2 7 10-14 15 1 16 15-19 55 2 57 20-24 170 10 180 25-29 269 14 283 30-34 381 9 390 35-39 583 14 597 40-44 871 20 891 45-49 1,119 20 1,139 50-54 1,505 35 1,540
IgA Nephropathy - «Maladie de Berger»
 IgA Nephropathy - «Maladie de Berger» B. Vogt, Division de Néphrologie/Consultation d Hypertension CHUV, Lausanne 2011 Montreux CME SGN-SSN IgA Nephropathy 1. Introduction 2. Etiology and Pathogenesis
IgA Nephropathy - «Maladie de Berger» B. Vogt, Division de Néphrologie/Consultation d Hypertension CHUV, Lausanne 2011 Montreux CME SGN-SSN IgA Nephropathy 1. Introduction 2. Etiology and Pathogenesis
C3 GLOMERULOPATHIES. Budapest Nephrology School Zoltan Laszik
 C3 GLOMERULOPATHIES Budapest Nephrology School 8.30.2018. Zoltan Laszik 1 Learning Objectives Familiarize with the pathogenetic mechanisms of glomerular diseases Learn the pathologic landscape and clinical
C3 GLOMERULOPATHIES Budapest Nephrology School 8.30.2018. Zoltan Laszik 1 Learning Objectives Familiarize with the pathogenetic mechanisms of glomerular diseases Learn the pathologic landscape and clinical
Random forest can accurately predict the development of end-stage renal disease in immunoglobulin a nephropathy patients
 Original Article Page 1 of 8 Random forest can accurately predict the development of end-stage renal disease in immunoglobulin a nephropathy patients Xin Han 1#, Xiaonan Zheng 2#, Ying Wang 3, Xiaoru Sun
Original Article Page 1 of 8 Random forest can accurately predict the development of end-stage renal disease in immunoglobulin a nephropathy patients Xin Han 1#, Xiaonan Zheng 2#, Ying Wang 3, Xiaoru Sun
Surgical Pathology Report
 Louisiana State University Health Sciences Center Department of Pathology Shreveport, Louisiana Accession #: Collected: Received: Reported: 6/1/2012 09:18 6/2/2012 09:02 6/2/2012 Patient Name: Med. Rec.
Louisiana State University Health Sciences Center Department of Pathology Shreveport, Louisiana Accession #: Collected: Received: Reported: 6/1/2012 09:18 6/2/2012 09:02 6/2/2012 Patient Name: Med. Rec.
Glomerular Pathology- 1 Nephrotic Syndrome. Dr. Nisreen Abu Shahin
 Glomerular Pathology- 1 Nephrotic Syndrome Dr. Nisreen Abu Shahin The Nephrotic Syndrome a clinical complex resulting from glomerular disease & includes the following: (1) massive proteinuria (3.5 gm /day
Glomerular Pathology- 1 Nephrotic Syndrome Dr. Nisreen Abu Shahin The Nephrotic Syndrome a clinical complex resulting from glomerular disease & includes the following: (1) massive proteinuria (3.5 gm /day
Current treatment recommendations in children with IgA nephropathy Selçuk Yüksel
 Current treatment recommendations in children with IgA nephropathy Selçuk Yüksel Department of Pediatric Nephrology Pamukkale University School of Medicine IgA Nephropathy The most common cause of primary
Current treatment recommendations in children with IgA nephropathy Selçuk Yüksel Department of Pediatric Nephrology Pamukkale University School of Medicine IgA Nephropathy The most common cause of primary
Mayo Clinic/ RPS Consensus Report on Classification, Diagnosis, and Reporting of Glomerulonephritis
 Mayo Clinic/ RPS Consensus Report on Classification, Diagnosis, and Reporting of Glomerulonephritis Sanjeev Sethi, MD, PhD Department of Laboratory Medicine and Pathology Disclosure Relevant Financial
Mayo Clinic/ RPS Consensus Report on Classification, Diagnosis, and Reporting of Glomerulonephritis Sanjeev Sethi, MD, PhD Department of Laboratory Medicine and Pathology Disclosure Relevant Financial
Classification of Glomerular Diseases and Defining Individual Glomerular Lesions: Developing International Consensus
 Classification of Glomerular Diseases and Defining Individual Glomerular Lesions: Developing International Consensus Mark Haas MD, PhD Department of Pathology & Laboratory Medicine Cedars-Sinai Medical
Classification of Glomerular Diseases and Defining Individual Glomerular Lesions: Developing International Consensus Mark Haas MD, PhD Department of Pathology & Laboratory Medicine Cedars-Sinai Medical
H enoch-schönlein purpura is a vasculitis with IgA
 1147 ORIGINAL ARTICLE Role of mesangial fibrinogen deposition in the pathogenesis of crescentic Henoch-Schönlein nephritis in children J I Shin, J M Park, Y H Shin, J S Lee, H J Jeong... See end of article
1147 ORIGINAL ARTICLE Role of mesangial fibrinogen deposition in the pathogenesis of crescentic Henoch-Schönlein nephritis in children J I Shin, J M Park, Y H Shin, J S Lee, H J Jeong... See end of article
Case Studies: Renal and Urologic Impairments Workshop
 Case Studies: Renal and Urologic Impairments Workshop Justine Lee, MD, DBIM New York Life Insurance Co. Gina Guzman, MD, DBIM, FALU, ALMI Munich Re AAIM Triennial October, 2012 The Company You Keep 1 Case
Case Studies: Renal and Urologic Impairments Workshop Justine Lee, MD, DBIM New York Life Insurance Co. Gina Guzman, MD, DBIM, FALU, ALMI Munich Re AAIM Triennial October, 2012 The Company You Keep 1 Case
An update on the pathogenesis and treatment of IgA nephropathy
 http://www.kidney-international.org & 2012 International Society of Nephrology An update on the pathogenesis and treatment of IgA nephropathy Joanna K. Boyd 1,2, Chee K. Cheung 1,2, Karen Molyneux 1,2,
http://www.kidney-international.org & 2012 International Society of Nephrology An update on the pathogenesis and treatment of IgA nephropathy Joanna K. Boyd 1,2, Chee K. Cheung 1,2, Karen Molyneux 1,2,
Management and treatment of glomerular diseases KDIGO Controversies Conference Part 1
 Management and treatment of glomerular diseases KDIGO Controversies Conference Part 1 Dr.M.Matinfar Assistant Professor of Internal Medicine & Nephrology IUMS -IKRC GENERAL PRINCIPLES IN THE MANAGEMENT
Management and treatment of glomerular diseases KDIGO Controversies Conference Part 1 Dr.M.Matinfar Assistant Professor of Internal Medicine & Nephrology IUMS -IKRC GENERAL PRINCIPLES IN THE MANAGEMENT
IgA nephropathy (IgAN) is among the most frequent glomerulonephritides
 O-Glycosylation of Serum IgD in IgA Nephropathy Alice C. Smith,* Jacob F. de Wolff, Karen Molyneux,* John Feehally,* and Jonathan Barratt* *John Walls Renal Unit, Leicester General Hospital and Department
O-Glycosylation of Serum IgD in IgA Nephropathy Alice C. Smith,* Jacob F. de Wolff, Karen Molyneux,* John Feehally,* and Jonathan Barratt* *John Walls Renal Unit, Leicester General Hospital and Department
Membranoproliferative Glomerulonephritis
 Membranoproliferative Glomerulonephritis MPGN is characterizedby alterations in the GBM and mesangium and by proliferation of glomerular cells. 5% to 10% of cases of 1ry nephrotic syndrome in children
Membranoproliferative Glomerulonephritis MPGN is characterizedby alterations in the GBM and mesangium and by proliferation of glomerular cells. 5% to 10% of cases of 1ry nephrotic syndrome in children
Demonstration of secretory IgA in kidneys of patients with IgA nephropathy
 Nephrol Dial Transplant (2007) 22: 3191 3195 doi:10.1093/ndt/gfm346 Advance Access publication 2 June 2007 Original Article Demonstration of secretory IgA in kidneys of patients with IgA nephropathy Beatrijs
Nephrol Dial Transplant (2007) 22: 3191 3195 doi:10.1093/ndt/gfm346 Advance Access publication 2 June 2007 Original Article Demonstration of secretory IgA in kidneys of patients with IgA nephropathy Beatrijs
IgA Nephropathy: Morphologic Findings Associated with Disease Progression and Therapeutic Response A Working Group Approach
 I IgA Nephropathy: Morphologic Findings Associated with Disease Progression and Therapeutic Response A Working Group Approach Mark Haas Department of Pathology & Lab Medicine Cedars-Sinai Medical Center
I IgA Nephropathy: Morphologic Findings Associated with Disease Progression and Therapeutic Response A Working Group Approach Mark Haas Department of Pathology & Lab Medicine Cedars-Sinai Medical Center
The application of machine learning to the diagnosis of glomerular disease
 Pre-publication draft of a paper which appeared in Sarmeinto, C. (Ed.), Proceedings of the IJCAI Workshop W.15: Representing Knowledge in Medical Decision Support Systems, pages 8.1-8.8. The application
Pre-publication draft of a paper which appeared in Sarmeinto, C. (Ed.), Proceedings of the IJCAI Workshop W.15: Representing Knowledge in Medical Decision Support Systems, pages 8.1-8.8. The application
The gut kidney axis in IgA nephropathy. Rosanna Coppo. Fondazione Ricerca Molinette Torino
 The gut kidney axis in IgA nephropathy Rosanna Coppo Turin, Italy Fondazione Ricerca Molinette Torino IgA nephropathy (IgAN): a disease originated from mucosal immunity dysregulation IgA: most prevalent
The gut kidney axis in IgA nephropathy Rosanna Coppo Turin, Italy Fondazione Ricerca Molinette Torino IgA nephropathy (IgAN): a disease originated from mucosal immunity dysregulation IgA: most prevalent
FIBRILLARY GLOMERULONEPHRITIS DIAGNOSTIC CRITERIA, PITFALLS, AND DIFFERENTIAL DIAGNOSIS
 FIBRILLARY GLOMERULONEPHRITIS DIAGNOSTIC CRITERIA, PITFALLS, AND DIFFERENTIAL DIAGNOSIS Guillermo A. Herrera MD Louisiana State University, Shreveport Fibrils in bundles 10-20 nm d Diabetic fibrillosis
FIBRILLARY GLOMERULONEPHRITIS DIAGNOSTIC CRITERIA, PITFALLS, AND DIFFERENTIAL DIAGNOSIS Guillermo A. Herrera MD Louisiana State University, Shreveport Fibrils in bundles 10-20 nm d Diabetic fibrillosis
Approach to Glomerular Diseases: Clinical Presentation Nephrotic Syndrome Nephritis
 GLOMERULONEPHRITIDES Vivette D Agati Jai Radhakrishnan Approach to Glomerular Diseases: Clinical Presentation Nephrotic Syndrome Nephritis Heavy Proteinuria Renal failure Low serum Albumin Hypertension
GLOMERULONEPHRITIDES Vivette D Agati Jai Radhakrishnan Approach to Glomerular Diseases: Clinical Presentation Nephrotic Syndrome Nephritis Heavy Proteinuria Renal failure Low serum Albumin Hypertension
Epidemiological Profile, Clinicopathological Correlation and Treatment response in adult
 aaaasasasss Shakar P et al.: Epidemiological Profile, Treatment response in adult patients with IgA Nephropathy Epidemiological Profile, Clinicopathological Correlation and Treatment response in adult
aaaasasasss Shakar P et al.: Epidemiological Profile, Treatment response in adult patients with IgA Nephropathy Epidemiological Profile, Clinicopathological Correlation and Treatment response in adult
RECURRENT AND DE NOVO RENAL DISEASES IN THE ALLOGRAFT. J. H. Helderman,MD,FACP,FAST
 RECURRENT AND DE NOVO RENAL DISEASES IN THE ALLOGRAFT J. H. Helderman,MD,FACP,FAST Vanderbilt University Medical Center Professor of Medicine, Pathology and Immunology Medical Director, Vanderbilt Transplant
RECURRENT AND DE NOVO RENAL DISEASES IN THE ALLOGRAFT J. H. Helderman,MD,FACP,FAST Vanderbilt University Medical Center Professor of Medicine, Pathology and Immunology Medical Director, Vanderbilt Transplant
Kengo Furuichi, Miho Shimizu, Akinori Hara, Tadashi Toyama and Takashi Wada
 doi: 10.2169/internalmedicine.1132-18 http://internmed.jp REVIEW ARTICLE Diabetic Nephropathy: A Comparison of the Clinical and Pathological Features between the CKD Risk Classification and the Classification
doi: 10.2169/internalmedicine.1132-18 http://internmed.jp REVIEW ARTICLE Diabetic Nephropathy: A Comparison of the Clinical and Pathological Features between the CKD Risk Classification and the Classification
Nephrology Dialysis Transplantation
 Nephrol Dial Transplant (1998) 13: 1984 1990 Original Article Nephrology Dialysis Transplantation Glomerular deposition of mannose-binding lectin (MBL) indicates a novel mechanism of complement activation
Nephrol Dial Transplant (1998) 13: 1984 1990 Original Article Nephrology Dialysis Transplantation Glomerular deposition of mannose-binding lectin (MBL) indicates a novel mechanism of complement activation
Renal biopsy findings and clinical indicators of patients with hematuria without overt proteinuria
 Clin Exp Nephrol (2015) 19:918 924 DOI 10.1007/s10157-015-1090-6 ORIGINAL ARTICLE Renal biopsy findings and clinical indicators of patients with hematuria without overt proteinuria Yoshie Hoshino Toshie
Clin Exp Nephrol (2015) 19:918 924 DOI 10.1007/s10157-015-1090-6 ORIGINAL ARTICLE Renal biopsy findings and clinical indicators of patients with hematuria without overt proteinuria Yoshie Hoshino Toshie
News Release. Title Development of a novel invasive test using leukocyte-derived proteins in urine for the diagnosis of glomerulonephritis
 News Release Title Development of a novel invasive test using leukocyte-derived proteins in urine for the diagnosis of glomerulonephritis Key Points Because histological examination for the diagnosis of
News Release Title Development of a novel invasive test using leukocyte-derived proteins in urine for the diagnosis of glomerulonephritis Key Points Because histological examination for the diagnosis of
29 Glomerular disease: an overview
 29 Glomerular : an overview Renal Extra-renal Neurological changes Clinical syndromes pressure Sore throat (streptococcal) Rash Cardiac valve lesions Hemoptysis Asymptomatic or Acute Glomerulonephritis
29 Glomerular : an overview Renal Extra-renal Neurological changes Clinical syndromes pressure Sore throat (streptococcal) Rash Cardiac valve lesions Hemoptysis Asymptomatic or Acute Glomerulonephritis
Nephrotic syndrome minimal change disease vs. IgA nephropathy. Hadar Meringer Internal medicine B Sheba
 Nephrotic syndrome minimal change disease vs. IgA nephropathy Hadar Meringer Internal medicine B Sheba The Case 29 year old man diagnosed with nephrotic syndrome 2 weeks ago and complaining now about Lt.flank
Nephrotic syndrome minimal change disease vs. IgA nephropathy Hadar Meringer Internal medicine B Sheba The Case 29 year old man diagnosed with nephrotic syndrome 2 weeks ago and complaining now about Lt.flank
Association of anti-mcv autoantibodies with SLE (Systemic Lupus Erythematosus) overlapping with various syndromes
 International Journal of Medicine and Medical Sciences Vol. () pp. 21-214, June 211 Available online http://www.academicjournals.org/ijmms ISSN 2-972 211 Academic Journals Full Length Research Paper Association
International Journal of Medicine and Medical Sciences Vol. () pp. 21-214, June 211 Available online http://www.academicjournals.org/ijmms ISSN 2-972 211 Academic Journals Full Length Research Paper Association
Internal Medicine ( ) 51 巻 11 号 :1323~1328.
 Internal Medicine (2012.01) 51 巻 11 号 :1323~1328. Retrospective Comparison of the Efficacy of Tonsillectomy with and without Steroid-pulse Therapy in IgA Nephropathy Patients. Nakagawa Naoki, Kabara Maki,
Internal Medicine (2012.01) 51 巻 11 号 :1323~1328. Retrospective Comparison of the Efficacy of Tonsillectomy with and without Steroid-pulse Therapy in IgA Nephropathy Patients. Nakagawa Naoki, Kabara Maki,
GOODPASTURE'S SYNDROME WITH CONCOMITANT IMMUNE COMPLEX MIXED MEMBRANOUS AND PROLIFERATIVE GLOMERULONEFRITIS
 GOODPASTURE'S SYNDROME WITH CONCOMITANT IMMUNE COMPLEX MIXED MEMBRANOUS AND PROLIFERATIVE GLOMERULONEFRITIS VESNA JURČIĆ 1, ANDREJA ALEŠ RIGLER 2, INSTITUTE OF PATHOLOGY, FACULTY OF MEDICINE, UNIVERSITY
GOODPASTURE'S SYNDROME WITH CONCOMITANT IMMUNE COMPLEX MIXED MEMBRANOUS AND PROLIFERATIVE GLOMERULONEFRITIS VESNA JURČIĆ 1, ANDREJA ALEŠ RIGLER 2, INSTITUTE OF PATHOLOGY, FACULTY OF MEDICINE, UNIVERSITY
Masatoshi Kawashima 1, Koji Wada 2, Hiroshi Ohta 2, Rika Moriya 3 and Yoshiharu Aizawa 1. Journal of Occupational Health
 176 J Occup Health, Vol. 54, 2012 J Occup Health 2012; 54: 176 180 Journal of Occupational Health Evaluation of Validity of the Urine Dipstick Test for Identification of Reduced Glomerular Filtration Rate
176 J Occup Health, Vol. 54, 2012 J Occup Health 2012; 54: 176 180 Journal of Occupational Health Evaluation of Validity of the Urine Dipstick Test for Identification of Reduced Glomerular Filtration Rate
Expression of P311, a transforming growth factor beta latency-associated protein-binding protein, in human kidneys with IgA nephropathy
 Int Urol Nephrol (2010) 42:811 819 DOI 10.1007/s11255-009-9681-3 NEPHROLOGY - ORIGINAL PAPER Expression of P311, a transforming growth factor beta latency-associated protein-binding protein, in human kidneys
Int Urol Nephrol (2010) 42:811 819 DOI 10.1007/s11255-009-9681-3 NEPHROLOGY - ORIGINAL PAPER Expression of P311, a transforming growth factor beta latency-associated protein-binding protein, in human kidneys
Index. electron microscopy, 81 immunofluorescence microscopy, 80 light microscopy, 80 Amyloidosis clinical setting, 185 etiology/pathogenesis,
 A Acute antibody-mediated rejection (Acute AMR) clinical features, 203 clinicopathologic correlations, 206 pathogenesis, 205 206 204 205 light microscopy, 203 204 Acute cellular rejection (ACR) clinical
A Acute antibody-mediated rejection (Acute AMR) clinical features, 203 clinicopathologic correlations, 206 pathogenesis, 205 206 204 205 light microscopy, 203 204 Acute cellular rejection (ACR) clinical
Enterprise Interest Nothing to declare
 Enterprise Interest Nothing to declare Minimal change disease (MCD) related new electron microscopy findings in a patient on Levothyroxine sodium (LT) for hypothyroidism: A case report Dr. Ali Al-Omari
Enterprise Interest Nothing to declare Minimal change disease (MCD) related new electron microscopy findings in a patient on Levothyroxine sodium (LT) for hypothyroidism: A case report Dr. Ali Al-Omari
PATTERNS OF RENAL INJURY
 PATTERNS OF RENAL INJURY Normal glomerulus podocyte Glomerular capillaries electron micrograph THE CLINICAL SYNDROMES 1. The Nephrotic Syndrome 2. The Acute Nephritic Syndrome 3. Rapidly Progressive Glomerulonephritis
PATTERNS OF RENAL INJURY Normal glomerulus podocyte Glomerular capillaries electron micrograph THE CLINICAL SYNDROMES 1. The Nephrotic Syndrome 2. The Acute Nephritic Syndrome 3. Rapidly Progressive Glomerulonephritis
Dr Ian Roberts Oxford. Oxford Pathology Course 2010 for FRCPath Illustration-Cellular Pathology. Oxford Radcliffe NHS Trust
 Dr Ian Roberts Oxford Oxford Pathology Course 2010 for FRCPath Plan of attack: Diagnostic approach to the renal biopsy Differential diagnosis of the clinical syndromes of renal disease Microscopy Step
Dr Ian Roberts Oxford Oxford Pathology Course 2010 for FRCPath Plan of attack: Diagnostic approach to the renal biopsy Differential diagnosis of the clinical syndromes of renal disease Microscopy Step
Case 3. ACCME/Disclosure. Laboratory results. Clinical history 4/13/2016
 Case 3 Lynn D. Cornell, M.D. Mayo Clinic, Rochester, MN Cornell.Lynn@mayo.edu USCAP Renal Case Conference March 13, 2016 ACCME/Disclosure Dr. Cornell has nothing to disclose Clinical history 57-year-old
Case 3 Lynn D. Cornell, M.D. Mayo Clinic, Rochester, MN Cornell.Lynn@mayo.edu USCAP Renal Case Conference March 13, 2016 ACCME/Disclosure Dr. Cornell has nothing to disclose Clinical history 57-year-old
Supplementary Appendix
 Supplementary Appendix This appendix has been provided by the authors to give readers additional information about their work. Supplement to: Debiec H, Lefeu F, Kemper MJ, et al. Early-childhood membranous
Supplementary Appendix This appendix has been provided by the authors to give readers additional information about their work. Supplement to: Debiec H, Lefeu F, Kemper MJ, et al. Early-childhood membranous
Am J Nephrol 2015;41: DOI: /
 American Journal of Nephrology Original Report: Patient-Oriented, Translational Research Received: September 1, 214 Accepted: March 2, 215 Published online: April 9, 215 Addition of egfr and Age Improves
American Journal of Nephrology Original Report: Patient-Oriented, Translational Research Received: September 1, 214 Accepted: March 2, 215 Published online: April 9, 215 Addition of egfr and Age Improves
Disorders of the kidney. Urine analysis. Nephrotic and nephritic syndrome.
 Disorders of the kidney. Urine analysis. Nephrotic and nephritic syndrome. Azotemia and Urinary Abnormalities Disturbances in urine volume oliguria, anuria, polyuria Abnormalities of urine sediment red
Disorders of the kidney. Urine analysis. Nephrotic and nephritic syndrome. Azotemia and Urinary Abnormalities Disturbances in urine volume oliguria, anuria, polyuria Abnormalities of urine sediment red
Dr. Rai Muhammad Asghar Head of Paediatric Department BBH Rawalpindi
 Dr. Rai Muhammad Asghar Head of Paediatric Department BBH Rawalpindi Acute Post streptococcal Glomerulonephritis Sudden onset of Gross hematuria Edema Hypertension Renal insufficiency Cause of AGN Post
Dr. Rai Muhammad Asghar Head of Paediatric Department BBH Rawalpindi Acute Post streptococcal Glomerulonephritis Sudden onset of Gross hematuria Edema Hypertension Renal insufficiency Cause of AGN Post
Urinary CD80 as a Replacement for Renal Biopsy for Diagnosis of Pediatric Minimal Change Disease
 KIDNEY DISEASES Urinary CD80 as a Replacement for Renal Biopsy for Diagnosis of Pediatric Minimal Change Disease Heba Mostafa Ahmed, 1 Dina Ahmed Ezzat, 1 Noha A Doudar, 2 Mai Adel 1 1 Departement of Pediatrics,
KIDNEY DISEASES Urinary CD80 as a Replacement for Renal Biopsy for Diagnosis of Pediatric Minimal Change Disease Heba Mostafa Ahmed, 1 Dina Ahmed Ezzat, 1 Noha A Doudar, 2 Mai Adel 1 1 Departement of Pediatrics,
Elevated Serum Creatinine, a simplified approach
 Elevated Serum Creatinine, a simplified approach Primary Care Update Creighton University School of Medicine. April 27 th, 2018 Disclosure Slide I have no disclosures and have no conflicts with this presentation.
Elevated Serum Creatinine, a simplified approach Primary Care Update Creighton University School of Medicine. April 27 th, 2018 Disclosure Slide I have no disclosures and have no conflicts with this presentation.
Journal of Nephropathology
 www.nephropathol.com DOI: 10.15171/jnp.2018.24 J Nephropathol. 2018;7(3):101-105 Journal of Nephropathology Relationship of CD147 kidney expression with various pathologic lesions, biochemical and demographic
www.nephropathol.com DOI: 10.15171/jnp.2018.24 J Nephropathol. 2018;7(3):101-105 Journal of Nephropathology Relationship of CD147 kidney expression with various pathologic lesions, biochemical and demographic
Xi Yang, Ri-Bao Wei, Ping Li, Yue Yang, Ting-Yu Su, Yu-Wei Gao, Qing-Ping Li, Xue-Guang Zhang, Xiang-Mei Chen
 Int J Clin Exp Pathol 2016;9(9):9401-9407 www.ijcep.com /ISSN:1936-2625/IJCEP0028098 Original Article Correlation between serum hepatitis B virus DNA replication level and clinicopathology in 235 patients
Int J Clin Exp Pathol 2016;9(9):9401-9407 www.ijcep.com /ISSN:1936-2625/IJCEP0028098 Original Article Correlation between serum hepatitis B virus DNA replication level and clinicopathology in 235 patients
Article. The Use of the Oxford Classification of IgA Nephropathy to Predict Renal Survival
 Article The Use of the Oxford Classification of IgA Nephropathy to Predict Renal Survival Eric Alamartine,* Catherine Sauron,* Blandine Laurent, Aurore Sury,* Aline Seffert,* and Christophe Mariat* Summary
Article The Use of the Oxford Classification of IgA Nephropathy to Predict Renal Survival Eric Alamartine,* Catherine Sauron,* Blandine Laurent, Aurore Sury,* Aline Seffert,* and Christophe Mariat* Summary
Citation Acta medica Nagasakiensia. 2003, 48
 NAOSITE: Nagasaki University's Ac Title Author(s) Renal Outcome of Immunoglobulin A N Horita, Yoshio; Tadokoro, Masato; T Miyazaki, Masanobu; Taguchi, Takash Yoshiyuki; Kohno, Shigeru Citation Acta medica
NAOSITE: Nagasaki University's Ac Title Author(s) Renal Outcome of Immunoglobulin A N Horita, Yoshio; Tadokoro, Masato; T Miyazaki, Masanobu; Taguchi, Takash Yoshiyuki; Kohno, Shigeru Citation Acta medica
Thin basement membrane syndrome in adults
 J Clin Pathol 1987;40:318-322 Thin basement membrane syndrome in adults S ABE, Y AMAGASAKI,* S IYORI,t K KONISHI, E KATO, H SAKAGUCHI,: K SHIMOYAMA** From the Department of Internal Medicine and tpathology,
J Clin Pathol 1987;40:318-322 Thin basement membrane syndrome in adults S ABE, Y AMAGASAKI,* S IYORI,t K KONISHI, E KATO, H SAKAGUCHI,: K SHIMOYAMA** From the Department of Internal Medicine and tpathology,
