Intraocular Radiotherapy for Age- Related Macular Degeneration
|
|
|
- Leslie Doyle
- 5 years ago
- Views:
Transcription
1 Intraocular Radiotherapy for Age- Related Macular Degeneration Policy Number: Last Review: 9/2017 Origination: 9/2008 Next Review: 9/2018 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will not provide coverage for intraocular radiation therapy for age-related macular degeneration. This is considered investigational. When Policy Topic is covered Not Applicable When Policy Topic is not covered Intraocular placement of a radiation source (brachytherapy) for the treatment of choroidal neovascularization is considered investigational. Proton beam therapy for the treatment of choroidal neovascularization is considered investigational. Stereotactic radiation therapy for the treatment of choroidal neovascularization is considered investigational. Description of Procedure or Service Populations Interventions Comparators Outcomes Interventions of Brachytherapy Individuals: With choroidal neovascularization associated with agerelated macular degeneration Individuals: With choroidal neovascularization associated with agerelated macular degeneration Interventions of Proton beam therapy Comparators of Vascular endothelial growth factor injections Photodynamic therapy Comparators of Vascular endothelial growth factor injections Photodynamic therapy Relevant outcomes include: Change in disease status Morbid events Functional outcomes Quality of life Medication use Treatment-related morbidity Relevant outcomes include: Change in disease status Morbid events Functional outcomes Quality of life Medication use Treatment-related morbidity
2 Individuals: With choroidal neovascularization associated with agerelated macular degeneration Interventions of Stereotactic radiotherapy Comparators of Vascular endothelial growth factor injections Photodynamic therapy Relevant outcomes include: Change in disease status Morbid events Functional outcomes Quality of life Medication use Treatment-related morbidity Intraocular radiation, including brachytherapy, proton beam therapy, and stereotactic radiotherapy, are being evaluated to treat choroidal neovascularization (CNV) associated with age-related macular degeneration (AMD). For individuals who have CNV due to AMD who receive brachytherapy, the evidence includes 2 randomized controlled trials (RCTs) comparing brachytherapy plus vascular endothelial growth factor (VEGF) to VEGF monotherapy as well as phase 1/2 trials and case series on the use of brachytherapy. Relevant outcomes are change in disease status, morbid events, functional outcomes, quality of life, medication use, and treatment-related morbidity. Both RCTs showed that brachytherapy did not attain noninferiority for visual acuity outcomes and was associated with a higher proportion of adverse events. The evidence is insufficient to determine the effects of the technology on health outcomes. For individuals who have CNV due to AMD who receive proton beam therapy, the evidence includes a randomized, prospective, sham-controlled trial and a pilot study. Relevant outcomes are change in disease status, morbid events, functional outcomes, quality of life, medication use, and treatment-related morbidity. Recruitment into the RCT was halted for ethical concerns, and available results did not show statistically significant stabilization of visual acuity. The evidence is insufficient to determine the effects of the technology on health outcomes. For individuals who have CNV due to AMD who receive stereotactic radiotherapy, the evidence includes an RCT with sham control. Relevant outcomes are change in disease status, morbid events, functional outcomes, quality of life, medication use, and treatment-related morbidity. The RCT showed a reduction in the number of VEGF treatments at 12- and 24-month intervals, but no significant differences versus controls for changes in visual acuity. The evidence is insufficient to determine the effects of the technology on health outcomes. Background Age-Related Macular Degeneration Age-related macular degeneration (AMD) is the leading cause of legal blindness in individuals older than age 60 in developed nations. AMD is characterized in its earliest stages by minimal visual impairment and the presence of large drusen and other pigmentary abnormalities on ophthalmoscopic examination. Two distinctive forms of degeneration may be observed. The first, called the atrophic or areolar or dry form, evolves slowly. Atrophic AMD is the most common form of degeneration and may be a precursor of the more visually impairing exudative neovascular form, also referred to as disciform or wet AMD. The wet form is distinguished from
3 the atrophic form by the development of choroidal neovascularization (CNV) and serous or hemorrhagic detachment of the retinal pigment epithelium. Risk of developing severe irreversible loss of vision is greatly increased by the presence of CNV. Standard Clinical Management Usual care for neovascular AMD includes intravitreal agents that target vascular endothelial growth factor (VEGF), including pegaptanib, ranibizumab, bevacizumab, and aflibercept. Photodynamic therapy is an older method that has been largely replaced by anti-vegf therapies. The intravitreal therapies may necessitate repeated intravitreal injections. Hence, alternative treatments, such as intraocular radiation, including brachytherapy, proton beam therapy (PBT), and stereotactic radiotherapy, are being investigated. The NeoVista Epi-Rad90 Ophthalmic System, a brachytherapy device, treats CNV by delivering focal radiation to a subfoveal choroidal neovascular lesion. Using a standard vitrectomy procedure, the cannula tip of a handheld (pipette-like) surgical device is inserted into the vitreous cavity and positioned under visual guidance over the target lesion. The radiation source (strontium 90) is advanced down the cannula until it reaches the tip, which is then held in place over the lesion for a prescribed time to deliver focused radiation. The system is designed to deliver a 1-time peak dose of beta particle energy (24 gray) for a target area 3 mm in depth and up to 5.4 mm in diameter. This dose is believed to be below that toxic to the retina and optic nerve. Radiation exposure outside of the target area is expected to be minimal. PBT is a type of external radiation that uses charged atomic particles (protons or helium ions) to target a given area. PBT differs from conventional electromagnetic (photon) radiotherapy in that, with PBT, there is less scatter as the particle beams pass through tissue to deposit ionizing energy at precise depths (Bragg peak). The theoretical advantage of PBT over photon therapy is the ability to deliver higher radiation doses to the target without harm to adjacent normal tissue. Stereotactic radiotherapy is a nonsurgical procedure performed in an office setting. It uses a robotically controlled device to deliver radiation beams through the inferior sclera to overlap at the macula. Other Treatments Other available therapeutic options for AMD not addressed in this evidence review include photodynamic therapy (evidence review ) and VEGF antagonists or angiostatics (evidence review in separate policy). For those whose visual loss impairs their ability to perform daily tasks, low-vision rehabilitative services offer resources to compensate for deficits. Regulatory Status No devices are specifically approved by the U.S. Food and Drug Administration (FDA) for intraocular radiation. An investigational device exemption was granted
4 by FDA for a phase 3 multicenter trial of the EPI-RAD90 (now known as Vidion Anti-Neovascular Epimacular Brachytherapy [EMBT] System; NeoVista) to provide data for a device application to FDA. This is a category B procedure. Rationale This evidence review was originally created in July 2008 and has been updated regularly with literature searches of the MEDLINE database. The most recent literature review was performed through January 25, A search of MEDLINE when this evidence review was created did not identify any peer-reviewed publications on epiretinal radiation. The original search identified older randomized trials using external beam radiotherapy for age-related macular degeneration (AMD) associated choroidal neovascularization (CNV). Little to no benefit in visual acuity was observed following repeated single treatments of 2 gray (Gy) to a total of 12 to 20 Gy. 1,2 Intraocular Radiotherapy Brachytherapy In 2016, Jackson et al reported the results of a phase 3 randomized controlled trial (RCT) comparing epimacular brachytherapy (EMB) plus as-needed ranibizumab (n=224) with as-needed ranibizumab alone (n=119) in patients with neovascular AMD, already receiving ranibizumab (MERLOT). 3 It was not feasible to mask patients to their surgical group (EMB), but visual acuity testing and macular imaging were evaluated by masked assessors. The trial was powered to test the hypothesis that EMB would reduce the number of anti-vascular endothelial growth factor (anti-vegf) treatments, with a noninferior visual outcome (with a margin of 5 letters of visual acuity). Over 12 months of follow-up, the mean number of asneeded ranibizumab injections did not differ significantly between the EMB arm (4.8 treatments) and the ranibizumab monotherapy arm (4.1 treatments; p=0.068). From baseline to month 12, the mean change in best-corrected visual acuity (BCVA) was -4.8 letters in the EMB arm compared with -0.9 letters in the ranibizumab monotherapy arm (between-group difference 95% confidence interval [CI], -6.6 to -1.8, which did not demonstrate inferiority at the prespecified 5-letter margin). In contrast to the null hypothesis, ranibizumab monotherapy patients had superior outcomes for visual acuity. Adverse effects were more common in the EMB arm. Overall, these results do not support the use of EMB over ranibizumab monotherapy for neovascular AMD. CABERNET (NCT ), a phase 3 multicenter RCT, enrolled 494 subjects with AMD-related wet CNV from 42 sites. 4,5 The safety and efficacy of EMB combined with 2 loading injections of ranibizumab (Lucentis) were compared to ranibizumab monotherapy (2 loading doses and then quarterly). Patients in both arms of the trial could receive monthly treatment with ranibizumab as needed. At 24 months, 77% of the patients in the EBM group lost fewer than 15 letters compared with 90% in the control group. This result did not meet the prespecified noninferiority margin. EMB treatment also did not meet the superiority end point, which was the
5 proportion of participants gaining more than 15 letters (16% vs 26% for the ranibizumab group). The most common serious adverse event was cataract surgery (known to be associated with vitrectomy), which occurred in 40% of the EMB group compared with 11% of the ranibizumab monotherapy group. Mild radiation retinopathy occurred in 3% of the patients who received EMB treatment. This trial did not support the use of epiretinal radiotherapy. Twelve- and 24-month results from the multicenter MERITAGE study (NCT ) were reported between 2012 and MERITAGE was a phase 1/2 study of EMB for the treatment of subfoveal CNV associated with wet AMD in patients requiring continued anti-vegf therapy to maintain an adequate response. Following a single 24-gray (Gy) dose, the 53 patients in the study received retreatment with ranibizumab administered monthly (as needed). In the 12 months before the study, participants received 0.45 injections per month. At the 12-month follow-up, 81% (43/53) of patients maintained stable vision (loss of <15 letters), with a mean of 3.49 anti-vegf injections (0.29 per month). Over 24 months, the durability of the application diminished, with 68% (32/47) of patients maintaining stable vision at a mean of 8.7 anti-vegf injections (0.72 per month). Three publications from 2 studies have been reported by Avila et al on EMB using the EPI-RAD90 System One report (2009) described 12-month safety and visual acuity results of a feasibility study in 34 treatment-naive patients from Turkey, Mexico, and Brazil, recruited between February 2005 and February The second report (2009) described 12-month safety and visual acuity results for 24-Gy EMB combined with bevacizumab in 34 treatment-naive patients enrolled between June 2006 and April Adverse events related to the device or procedure included subretinal hemorrhage (n=1), retinal tear (n=1), subretinal fibrosis (n=2), epiretinal membrane (n=1), and cataract (n=6/24; 24 patients were phakic at baseline). All occurrences of cataracts were deemed to be related to the vitrectomy procedure. Two- and 3-year results from this trial were published in All 34 subjects were followed for 24 months; 1 site that enrolled 19 patients agreed to re-consent and follow patients for 3 years. On average, the cohort followed for 36 months received 3.0 bevacizumab injections. Twelve (50%) of the 24 phakic patients developed cataracts, and 4 had phacoemulsification with intraocular lens implantation. Mean change in visual acuity at 36 months was +3.9 letters. Seven (54%) of 13 phakic patients developed cataracts, and 4 had phacoemulsification with intraocular lens implantation. One case of nonproliferative radiation retinopathy was observed at 36 months. Section Summary: Brachytherapy At least 2 RCTs, which have been supported by additional nonrandomized studies, have found that EMB is inferior to local treatment with ranibizumab for the treatment of wet AMB. Proton Beam Therapy In 2012, Park et al reported on 12- to 36-month follow-up of a pilot study of ranibizumab combined with proton beam therapy (PBT) for AMD. 12 Six eyes (6
6 patients) were treated with 4 monthly ranibizumab plus 24-Gy proton beam treatments, followed by ranibizumab if needed. No radiation retinopathy was observed at follow-up. In 2002, Ciulla et al reported results from a randomized, prospective, shamcontrolled, double-masked treatment trial that examined the effect of PBT on subfoveal choroidal neovascular membranes associated with AMD. 13 Thirty-seven subjects were randomized to 16-Gy proton irradiation delivered in 2 fractions 24 hours apart or to sham control treatment. Recruitment was halted at 37 subjects for ethical reasons related to randomization to sham treatment when U.S. Food and Drug Administration approval of verteporfin (Visudyne; a light-activated drug used with photodynamic therapy) was anticipated. PBT was associated with a trend toward stabilization of visual acuity, but this association was not statistically significant. Section Summary: Proton Beam Therapy There is currently no available clinical trial evidence suggesting that PBT is noninferior to available treatment alternatives for AMD. Stereotactic Radiotherapy INTREPID was a randomized, sham-controlled, double-masked trial (2013) with 230 patients that assessed the efficacy and safety of stereotactic radiotherapy (SRT) to treat neovascular AMD. 14 The primary outcome measure was the number of ranibizumab injections needed over 52 weeks. Both SRT and sham-control patients received ranibizumab as needed. After 1 year, treatment with 16- or 24- Gy SRT reduced the number of ranibizumab treatments (median, 2 vs 3.5 for sham controls) with no significant differences from controls in changes in visual acuity over the 1-year follow-up. No safety concerns were identified in the first 12 months. In 2015, year 2 safety and efficacy results from the INTREPID trial were published. 15 Participants received 16- or 24-Gy SRT plus ranibizumab or sham SRT plus ranibizumab for 12 months, with bevacizumab or ranibizumab thereafter as needed. At year 2, the 16- and 24-Gy arms received fewer as needed bevacizumab (mean, 4.5; p=0.008) or ranibizumab (mean, 5.4; p=0.09) treatments compared with sham (mean, 6.6). Changes in mean best-corrected visual acuity were -10.0, -7.5, and -6.7 letters, respectively, with 68%, 75%, and 79% losing fewer than 15 letters, respectively. Differences for visual acuity were not statistically significant. Microvascular abnormalities were detected in 6 control eyes and 29 SRT eyes, of which 18 were attributed to radiation, with only 2 possibly affecting vision. The authors concluded that a single dose of SRT significantly reduced intravitreal injections over 2 years, and that, although radiotherapy can induce microvascular changes, only in 1% of eyes did this seem to affect vision. In 2016, Ranjbar et al reported results from an observational study of 32 patients (32 eyes) with neovascular AMD who met criteria for best responders in the INTREPID trial and were treated with SRT (16 Gy) along with aflibercept or
7 ranibizumab. 16 For the study s primary outcome (the number of anti-vegf treatments in the 12 months post-srt), significantly fewer intravitreal injections were given (3.47) compared with the year preceding SRT (6.81; p<0.001). No ocular or systemic adverse events occurred. Section Summary: Stereotactic Radiotherapy Evidence from a double-blind, randomized trial comparing SRT with ranibizumab for neovascular AMD has suggested that SRT can reduce the number or ranibizumab injections, but was associated with radiation retinopathy leading to microvascular changes. Summary of Evidence For individuals who have choroidal neovascularization (CNV) due to age-related macular degeneration (AMD) who receive brachytherapy, the evidence includes 2 randomized controlled trials (RCTs) comparing brachytherapy plus vascular endothelial growth factor (VEGF) to VEGF monotherapy as well as phase 1/2 trials and case series on the use of brachytherapy. Relevant outcomes are change in disease status, morbid events, functional outcomes, quality of life, medication use, and treatment-related morbidity. Both RCTs showed that brachytherapy did not attain noninferiority for visual acuity outcomes and was associated with a higher proportion of adverse events. The evidence is insufficient to determine the effects of the technology on health outcomes. For individuals who have CNV due to AMD who receive proton beam therapy, the evidence includes a randomized, prospective, sham-controlled trial and a pilot study. Relevant outcomes are change in disease status, morbid events, functional outcomes, quality of life, medication use, and treatment-related morbidity. Recruitment into the RCT was halted for ethical concerns, and available results did not show statistically significant stabilization of visual acuity. The evidence is insufficient to determine the effects of the technology on health outcomes. For individuals who have CNV due to AMD who receive stereotactic radiotherapy, the evidence includes an RCT with sham control. Relevant outcomes are change in disease status, morbid events, functional outcomes, quality of life, medication use, and treatment-related morbidity. The RCT showed a reduction in the number of VEGF treatments at 12- and 24-month intervals, but no significant differences versus controls for changes in visual acuity. The evidence is insufficient to determine the effects of the technology on health outcomes. Supplemental Information Practice Guidelines and Position Statements American Academy of Ophthalmology In 2015, the American Academy of Ophthalmology (AAO) updated its evidencedbased preferred practice pattern on age-related macular degeneration (AMD). 17 For extrafoveal choroidal neovascularization, radiotherapy was not recommended
8 (SIGN grade: III; GRADE assessment: moderate level of evidence, strong recommendation). National Institute for Health and Care Excellence The 2011 guidance from the U.K. s National Institute for Health and Care Excellence (NICE) stated that current evidence on the efficacy of epiretinal brachytherapy for wet AMD is inadequate and limited to small numbers of patients. For safety, vitrectomy has well-recognised complications and there is a possibility of subsequent radiation retinopathy. NICE concluded that wet AMD should only be used for research. 18 U.S. Preventive Services Task Force Recommendations Not applicable. Medicare National Coverage There is no national coverage determination (NCD). In the absence of an NCD, coverage decisions are left to the discretion of local Medicare carriers. Ongoing and Unpublished Clinical Trials Some currently unpublished trials that might influence this review are listed in Table 1. Table 1. Summary of Key Trials NCT No. Trial Name Planned Enrollment Unpublished NCT a A Feasibility Study to Evaluate the Safety And Tolerability of the EPI-RAD90 Ophthalmic System for the Treatment of Subfoveal Choroidal Neovascularization (CNV) in Patients With Age- Related Macular Degeneration (AMD) That Have Failed Primary Anti-VEGF Therapy NCT: national clinical trial. a Denotes industry-sponsored or cosponsored trial. Completion Date 20 Jun 2011 (completed) References 1. A prospective, randomized, double-masked trial on radiation therapy for neovascular agerelated macular degeneration (RAD Study). Radiation therapy for age-related macular degeneration. Ophthalmology. Dec 1999;106(12): PMID Stevenson MR, Hart PM, Chakravarthy U, et al. Visual functioning and quality of life in the SubFoveal Radiotherapy Study (SFRADS): SFRADS report 2. Br J Ophthalmol. Aug 2005;89(8): PMID Jackson TL, Desai R, Simpson A, et al. Epimacular brachytherapy for previously treated neovascular age-related macular degeneration (MERLOT): a phase 3 randomized controlled trial. Ophthalmology. Jun 2016;123(6): PMID Dugel PU, Bebchuk JD, Nau J, et al. Epimacular brachytherapy for neovascular age-related macular degeneration: a randomized, controlled trial (CABERNET). Ophthalmology. Feb 2013;120(2): PMID Jackson TL, Dugel PU, Bebchuk JD, et al. Epimacular brachytherapy for neovascular age-related macular degeneration (CABERNET): fluorescein angiography and optical coherence tomography. Ophthalmology. Aug 2013;120(8): PMID
9 6. Dugel PU, Petrarca R, Bennett M, et al. Macular epiretinal brachytherapy in treated age-related macular degeneration: MERITAGE study: twelve-month safety and efficacy results. Ophthalmology. Jul 2012;119(7): PMID Petrarca R, Dugel PU, Nau J, et al. Macular epiretinal brachytherapy in treated age-related macular degeneration (MERITAGE): month 12 optical coherence tomography and fluorescein angiography. Ophthalmology. Feb 2013;120(2): PMID Petrarca R, Dugel PU, Bennett M, et al. Macular epiretinal brachytherapy in treated age-related macular degeneration (MERITAGE): month 24 safety and efficacy results. Retina. May 2014;34(5): PMID Avila MP, Farah ME, Santos A, et al. Twelve-month safety and visual acuity results from a feasibility study of intraocular, epiretinal radiation therapy for the treatment of subfoveal CNV secondary to AMD. Retina. Feb 2009;29(2): PMID Avila MP, Farah ME, Santos A, et al. Twelve-month short-term safety and visual-acuity results from a multicentre prospective study of epiretinal strontium-90 brachytherapy with bevacizumab for the treatment of subfoveal choroidal neovascularisation secondary to agerelated macular degeneration. Br J Ophthalmol. Mar 2009;93(3): PMID Avila MP, Farah ME, Santos A, et al. Three-year safety and visual acuity results of epimacular 90strontium/90yttrium brachytherapy with bevacizumab for the treatment of subfoveal choroidal neovascularization secondary to age-related macular degeneration. Retina. Jan 2012;32(1): PMID Park SS, Daftari I, Phillips T, et al. Three-year follow-up of a pilot study of ranibizumab combined with proton beam irradiation as treatment for exudative age-related macular degeneration. Retina. May 2012;32(5): PMID Ciulla TA, Danis RP, Klein SB, et al. Proton therapy for exudative age-related macular degeneration: a randomized, sham-controlled clinical trial. Am J Ophthalmol. Dec 2002;134(6): PMID Jackson TL, Chakravarthy U, Kaiser PK, et al. Stereotactic radiotherapy for neovascular agerelated macular degeneration: 52-week safety and efficacy results of the INTREPID study. Ophthalmology. Sep 2013;120(9): PMID Jackson TL, Chakravarthy U, Slakter JS, et al. Stereotactic radiotherapy for neovascular agerelated macular degeneration: year 2 results of the INTREPID study. Ophthalmology. Jan 2015;122(1): PMID Ranjbar M, Kurz M, Holzhey A, et al. Stereotactic radiotherapy in neovascular age-related macular degeneration: Real-life efficacy and morphological evaluation of the outer retinachoroid complex. Medicine (Baltimore). Dec 2016;95(52):e5729. PMID American Academy of Ophthalmology Retina/Vitreous Panel. Preferred Practice Pattern: Age- Related Macular Degeneration. 2015; Accessed February 28, National Institute for Health and Clinical Excellence. Epiretinal brachytherapy for wet agerelated macular degeneration [IPG415]. 2011; Accessed March 8, Billing Coding/Physician Documentation Information 0190T Placement of intraocular radiation source applicator (List separately in addition to primary procedure) Vitrectomy, mechanical, pars plana approach; ICD-10 Codes H35.30 Unspecified macular degeneration (age-related) H35.31 Nonexudative age-related macular degeneration H35.32 Exudative age-related macular degeneration CPT code 0190T is to be used in conjunction with (Vitrectomy, mechanical, pars plana approach).
10 CPT code 0190T differs from code (destruction of localized lesion of the retina (e.g., macular edema, tumors), one or more sessions; radiation by implantation of source) because the radiation source is not implanted. Additional Policy Key Words N/A Policy Implementation/Update Information 9/1/08 New policy; considered investigational. 9/1/09 No policy statement changes. 9/1/10 No policy statement changes. 9/1/11 No policy statement changes. 9/1/12 No policy statement changes. 9/1/13 Investigational statement added on proton beam therapy and title changed from Epiretinal Radiation Therapy to Intraocular Radiation Therapy 9/1/14 Investigational statement added regarding stereotactic radiation therapy. 9/1/15 Title revised to say Radiotherapy. No policy statement changes. 9/1/16 Policy statements clarified as to type of radiation therapy used, but intent unchanged. 9/1/17 No policy statement changes. State and Federal mandates and health plan contract language, including specific provisions/exclusions, take precedence over Medical Policy and must be considered first in determining eligibility for coverage. The medical policies contained herein are for informational purposes. The medical policies do not constitute medical advice or medical care. Treating health care providers are independent contractors and are neither employees nor agents Blue KC and are solely responsible for diagnosis, treatment and medical advice. No part of this publication may be reproduced, stored in a retrieval system or transmitted, in any form or by any means, electronic, photocopying, or otherwise, without permission from Blue KC.
FEP Medical Policy Manual
 FEP Medical Policy Manual Last Review: September 2016 Next Review: September 2017 Related Policies 6.01.10 Stereotactic Radiosurgery and Stereotactic Body Radiotherapy 8.01.10 Charged-Particle (Proton
FEP Medical Policy Manual Last Review: September 2016 Next Review: September 2017 Related Policies 6.01.10 Stereotactic Radiosurgery and Stereotactic Body Radiotherapy 8.01.10 Charged-Particle (Proton
Intraocular Radiation Therapy for Age-Related Macular Degeneration
 Medical Policy Manual Medicine, Policy No. 134 Intraocular Radiation Therapy for Age-Related Macular Degeneration Next Review: April 2019 Last Review: June 2018 Effective: August 1, 2018 IMPORTANT REMINDER
Medical Policy Manual Medicine, Policy No. 134 Intraocular Radiation Therapy for Age-Related Macular Degeneration Next Review: April 2019 Last Review: June 2018 Effective: August 1, 2018 IMPORTANT REMINDER
Intraocular Radiation Therapy for Age-Related Macular Degeneration
 Intraocular Radiation Therapy for Age-Related Macular Degeneration Policy Number: 9.03.20 Last Review: 9/2014 Origination: 9/2008 Next Review: 9/2015 Policy Blue Cross and Blue Shield of Kansas City (Blue
Intraocular Radiation Therapy for Age-Related Macular Degeneration Policy Number: 9.03.20 Last Review: 9/2014 Origination: 9/2008 Next Review: 9/2015 Policy Blue Cross and Blue Shield of Kansas City (Blue
A Little Physics. How Does It Work? Radiation Therapy for Choroidal Neovascularisation in AMD A Review. => cell death
 Radiation Therapy for Choroidal Neovascularisation in AMD A Review Jen Anikina ST4 Hillingdon Hospital A Little Physics One gray is the absorption of one joule of ionizing radiation energy per kilogram
Radiation Therapy for Choroidal Neovascularisation in AMD A Review Jen Anikina ST4 Hillingdon Hospital A Little Physics One gray is the absorption of one joule of ionizing radiation energy per kilogram
Coding Implications Revision Log. See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: Verteporfin (Visudyne) Reference Number: CP.PHAR.187 Effective Date: 03.16 Last Review Date: 02.18 Line of Business: Commercial, Medicaid Coding Implications Revision Log See Important
Clinical Policy: Verteporfin (Visudyne) Reference Number: CP.PHAR.187 Effective Date: 03.16 Last Review Date: 02.18 Line of Business: Commercial, Medicaid Coding Implications Revision Log See Important
Clinical Policy: Implantable Miniature Telescope for Age Related Macular Degeneration Reference Number: CP.MP.517
 Clinical Policy: Implantable Miniature Telescope for Age Related Macular Reference Number: CP.MP.517 Effective Date: 11/16 Last Review Date: 11/17 See Important Reminder at the end of this policy for important
Clinical Policy: Implantable Miniature Telescope for Age Related Macular Reference Number: CP.MP.517 Effective Date: 11/16 Last Review Date: 11/17 See Important Reminder at the end of this policy for important
Clinical Policy: Neovascular (WET) Macular Degeneration Treatment Reference Number: CP.MP.283
 Clinical Policy: Neovascular (WET) Macular Degeneration Treatment Reference Number: CP.MP.283 Effective Date: 11/16 Last Review Date: 11/17 See Important Reminder at the end of this policy for important
Clinical Policy: Neovascular (WET) Macular Degeneration Treatment Reference Number: CP.MP.283 Effective Date: 11/16 Last Review Date: 11/17 See Important Reminder at the end of this policy for important
Coding Implications Revision Log. See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: (Eylea) Reference Number: CP.PHAR.184 Effective Date: 03.16 Last Review Date: 02.18 Line of Business: Commercial, Medicaid Coding Implications Revision Log See Important Reminder at the
Clinical Policy: (Eylea) Reference Number: CP.PHAR.184 Effective Date: 03.16 Last Review Date: 02.18 Line of Business: Commercial, Medicaid Coding Implications Revision Log See Important Reminder at the
CENTENE PHARMACY AND THERAPEUTICS NEW DRUG REVIEW 2Q17 April May
 BRAND NAME Lucentis GENERIC NAME ranibizumab MANUFACTURER Genentech, Inc. DATE OF APPROVAL June 30, 2006 PRODUCT LAUNCH DATE July 13, 2006 REVIEW TYPE Review type 1 (RT1): New Drug Review Full review of
BRAND NAME Lucentis GENERIC NAME ranibizumab MANUFACTURER Genentech, Inc. DATE OF APPROVAL June 30, 2006 PRODUCT LAUNCH DATE July 13, 2006 REVIEW TYPE Review type 1 (RT1): New Drug Review Full review of
Clinical Policy: Verteporfin (Visudyne) Reference Number: CP.PHAR.187
 Clinical Policy: (Visudyne) Reference Number: CP.PHAR.187 Effective Date: 03/16 Last Review Date: 03/17 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory
Clinical Policy: (Visudyne) Reference Number: CP.PHAR.187 Effective Date: 03/16 Last Review Date: 03/17 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory
Facet Arthroplasty. Policy Number: Last Review: 9/2018 Origination: 9/2009 Next Review: 3/2019
 Facet Arthroplasty Policy Number: 7.01.120 Last Review: 9/2018 Origination: 9/2009 Next Review: 3/2019 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will not provide coverage for total facet
Facet Arthroplasty Policy Number: 7.01.120 Last Review: 9/2018 Origination: 9/2009 Next Review: 3/2019 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will not provide coverage for total facet
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: (Lucentis) Reference Number: CP.PHAR.186 Effective Date: 03.16 Last Review Date: 02.18 Line of Business: Commercial, Medicaid Coding Implications Revision Log See Important Reminder at
Clinical Policy: (Lucentis) Reference Number: CP.PHAR.186 Effective Date: 03.16 Last Review Date: 02.18 Line of Business: Commercial, Medicaid Coding Implications Revision Log See Important Reminder at
Peripheral Subcutaneous Field Stimulation
 Peripheral Subcutaneous Field Stimulation Policy Number: 7.01.139 Last Review: 3/2018 Origination: 7/2013 Next Review: 9/2018 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will not provide
Peripheral Subcutaneous Field Stimulation Policy Number: 7.01.139 Last Review: 3/2018 Origination: 7/2013 Next Review: 9/2018 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will not provide
SUMMARY. Heather Casparis, MD,* and Neil M. Bressler, MD MARINA AND ANCHOR
 The following are summaries of selected presentations and posters from the American Society of Retina Specialists and European VitreoRetinal Society Annual Meeting held September 9 13, 2006, in Cannes,
The following are summaries of selected presentations and posters from the American Society of Retina Specialists and European VitreoRetinal Society Annual Meeting held September 9 13, 2006, in Cannes,
Avastin (bevacizumab)
 Avastin (bevacizumab) Policy Number: 5.02.502 Last Review: 04/2018 Origination: 03/2017 Next Review: 04/2019 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will provide coverage for Avastin
Avastin (bevacizumab) Policy Number: 5.02.502 Last Review: 04/2018 Origination: 03/2017 Next Review: 04/2019 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will provide coverage for Avastin
Clinical Policy: Aflibercept (Eylea) Reference Number: CP.PHAR.184
 Clinical Policy: (Eylea) Reference Number: CP.PHAR.184 Effective Date: 03/16 Last Review Date: 03/17 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory
Clinical Policy: (Eylea) Reference Number: CP.PHAR.184 Effective Date: 03/16 Last Review Date: 03/17 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory
FEP Medical Policy Manual
 FEP Medical Policy Manual Last Review: September 2016 Next Review: September 2017 Related Policies 9.03.20 Intraocular Radiation Therapy for Age-Related Macular Degeneration Photodynamic Therapy for Choroidal
FEP Medical Policy Manual Last Review: September 2016 Next Review: September 2017 Related Policies 9.03.20 Intraocular Radiation Therapy for Age-Related Macular Degeneration Photodynamic Therapy for Choroidal
Original Policy Date
 MP 9.03.08 Photocoagulation of Macular Drusen Medical Policy Section Miscellaneous Policies Issue 12/2013 Original Policy Date 12/2013 Last Review Status/Date Reviewed with literature search/12/2013 Return
MP 9.03.08 Photocoagulation of Macular Drusen Medical Policy Section Miscellaneous Policies Issue 12/2013 Original Policy Date 12/2013 Last Review Status/Date Reviewed with literature search/12/2013 Return
Clinical Trials Related to Age Related Macular Degeneration
 Clinical Trials Related to Age Related Macular Degeneration Kirti Singh MD, DNB, FRCS Kirti Singh MD, DNB, FRCS, Pooja Jain MBBS, Nitasha Ahir MBBS, Divya Jain MD, DNB Guru Nanak Eye Centre, Maulana Azad
Clinical Trials Related to Age Related Macular Degeneration Kirti Singh MD, DNB, FRCS Kirti Singh MD, DNB, FRCS, Pooja Jain MBBS, Nitasha Ahir MBBS, Divya Jain MD, DNB Guru Nanak Eye Centre, Maulana Azad
Photodynamic Therapy for Choroidal Neovascularization
 Photodynamic Therapy for Choroidal Neovascularization Policy Number: 9.03.08 Last Review: 10/2014 Origination: 10/2000 Next Review: 10/2015 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will
Photodynamic Therapy for Choroidal Neovascularization Policy Number: 9.03.08 Last Review: 10/2014 Origination: 10/2000 Next Review: 10/2015 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will
Vertebral Axial Decompression
 Vertebral Axial Decompression Policy Number: 8.03.09 Last Review: 11/2017 Origination: 11/2005 Next Review: 11/2018 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will not provide coverage
Vertebral Axial Decompression Policy Number: 8.03.09 Last Review: 11/2017 Origination: 11/2005 Next Review: 11/2018 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will not provide coverage
Clinical Policy Title: Ocular photodynamic therapy (OPDT) with Visudyne (verteporfin) for macular degeneration treatment
 Clinical Policy Title: Ocular photodynamic therapy (OPDT) with Visudyne (verteporfin) for macular degeneration treatment Clinical Policy Number: 10.02.04 Effective Date: January 1, 2016 Initial Review
Clinical Policy Title: Ocular photodynamic therapy (OPDT) with Visudyne (verteporfin) for macular degeneration treatment Clinical Policy Number: 10.02.04 Effective Date: January 1, 2016 Initial Review
FEP Medical Policy Manual
 FEP Medical Policy Manual Last Review: September 2016 Next Review: September 2017 Related Policies 9.03.08 Photodynamic Therapy for Choroidal Neovascularization 9.03.20 Intraocular Radiation Therapy for
FEP Medical Policy Manual Last Review: September 2016 Next Review: September 2017 Related Policies 9.03.08 Photodynamic Therapy for Choroidal Neovascularization 9.03.20 Intraocular Radiation Therapy for
Although photocoagulation and photodynamic PROCEEDINGS PEGAPTANIB SODIUM FOR THE TREATMENT OF AGE-RELATED MACULAR DEGENERATION *
 PEGAPTANIB SODIUM FOR THE TREATMENT OF AGE-RELATED MACULAR DEGENERATION Evangelos S. Gragoudas, MD ABSTRACT In December 24, the US Food and Drug Administration (FDA) approved pegaptanib sodium. Pegaptanib
PEGAPTANIB SODIUM FOR THE TREATMENT OF AGE-RELATED MACULAR DEGENERATION Evangelos S. Gragoudas, MD ABSTRACT In December 24, the US Food and Drug Administration (FDA) approved pegaptanib sodium. Pegaptanib
Bioimpedance Devices for Detection and Management of Lymphedema
 Bioimpedance Devices for Detection and Management of Lymphedema Policy Number: 2.01.82 Last Review: 5/2017 Origination: 1/2011 Next Review: 5/2018 Policy Blue Cross and Blue Shield of Kansas City (Blue
Bioimpedance Devices for Detection and Management of Lymphedema Policy Number: 2.01.82 Last Review: 5/2017 Origination: 1/2011 Next Review: 5/2018 Policy Blue Cross and Blue Shield of Kansas City (Blue
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE. Health Technology Appraisal. Aflibercept for treating diabetic macular oedema.
 NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Health Technology Appraisal Aflibercept for treating diabetic macular oedema Final scope Final remit/appraisal objective To appraise the clinical and cost
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Health Technology Appraisal Aflibercept for treating diabetic macular oedema Final scope Final remit/appraisal objective To appraise the clinical and cost
Description. Section: Other Effective Date: July 15, Subsection: Vision Original Policy Date: December 7, 2011 Subject: Page: 1 of 23
 Last Review Status/Date: June 2015 Page: 1 of 23 Description Photodynamic therapy (PDT) is a treatment modality designed to selectively occlude ocular choroidal neovascular tissue. The therapy is a 2-step
Last Review Status/Date: June 2015 Page: 1 of 23 Description Photodynamic therapy (PDT) is a treatment modality designed to selectively occlude ocular choroidal neovascular tissue. The therapy is a 2-step
There are no published randomized, double-blind trials comparing aflibercept to other therapies in neovascular AMD.
 Subject: Eylea (aflibercept) Original Effective Date: 7/11/2014 Policy Number: MCP-191 Revision Date(s): 10/11/2016 Review Date(s): 12/16/2015; 10/11/2016, 6/22/2017, 7/10/2018 DISCLAIMER This Medical
Subject: Eylea (aflibercept) Original Effective Date: 7/11/2014 Policy Number: MCP-191 Revision Date(s): 10/11/2016 Review Date(s): 12/16/2015; 10/11/2016, 6/22/2017, 7/10/2018 DISCLAIMER This Medical
Drug Class Update: Vascular Endothelial Growth Factors
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Peripheral Subcutaneous Field Stimulation
 Peripheral Subcutaneous Field Stimulation Policy Number: 7.01.139 Last Review: 9/2018 Origination: 7/2013 Next Review: 3/2019 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will not provide
Peripheral Subcutaneous Field Stimulation Policy Number: 7.01.139 Last Review: 9/2018 Origination: 7/2013 Next Review: 3/2019 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will not provide
MEDICAL POLICY SUBJECT: PHOTODYNAMIC THERAPY FOR EFFECTIVE DATE: 07/20/00 SUBFOVEAL CHOROIDAL NEOVASCULARIZATION
 MEDICAL POLICY SUBJECT: PHOTODYNAMIC THERAPY FOR PAGE: 1 OF: 8 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial product, including
MEDICAL POLICY SUBJECT: PHOTODYNAMIC THERAPY FOR PAGE: 1 OF: 8 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial product, including
Clinical Study Synopsis
 Clinical Study Synopsis This Clinical Study Synopsis is provided for patients and healthcare professionals to increase the transparency of Bayer's clinical research. This document is not intended to replace
Clinical Study Synopsis This Clinical Study Synopsis is provided for patients and healthcare professionals to increase the transparency of Bayer's clinical research. This document is not intended to replace
Ophthalmic VEGF Inhibitors. Eylea (aflibercept), Macugen (pegaptanib) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Subject: Ophthalmic VEGF Inhibitors Page: 1 of 5 Last Review Date: September 20, 2018 Ophthalmic VEGF Inhibitors
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Subject: Ophthalmic VEGF Inhibitors Page: 1 of 5 Last Review Date: September 20, 2018 Ophthalmic VEGF Inhibitors
London Medicines Evaluation Network Review
 London Medicines Evaluation Network Review Evidence for initiating intravitreal bevacizumab for the management of wet age-related macular degeneration (wet-amd) in eyes with vision better than 6/12 November
London Medicines Evaluation Network Review Evidence for initiating intravitreal bevacizumab for the management of wet age-related macular degeneration (wet-amd) in eyes with vision better than 6/12 November
Description. Section: Other Effective Date: July 15, Subsection: Vision Original Policy Date: September 13, 2011 Subject: Page: 1 of 18
 Last Review Status/Date: June 2014 Page: 1 of 18 Description Angiogenesis inhibitors (e.g., ranibizumab, bevacizumab, pegaptanib, aflibercept) are being evaluated for the treatment of disorders of choroidal
Last Review Status/Date: June 2014 Page: 1 of 18 Description Angiogenesis inhibitors (e.g., ranibizumab, bevacizumab, pegaptanib, aflibercept) are being evaluated for the treatment of disorders of choroidal
Vertebral Axial Decompression
 Vertebral Axial Decompression Policy Number: 8.03.09 Last Review: 11/2018 Origination: 11/2005 Next Review: 11/2019 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will not provide coverage
Vertebral Axial Decompression Policy Number: 8.03.09 Last Review: 11/2018 Origination: 11/2005 Next Review: 11/2019 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will not provide coverage
Corporate Medical Policy
 Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: charged_particle_radiotherapy 3/12/96 5/2017 5/2018 5/2017 Description of Procedure or Service Charged-particle
Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: charged_particle_radiotherapy 3/12/96 5/2017 5/2018 5/2017 Description of Procedure or Service Charged-particle
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE GUIDANCE EXECUTIVE (GE)
 NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE GUIDANCE EXECUTIVE (GE) Review of TA68; The clinical and cost effectiveness of photodynamic therapy for age-related macular degeneration TA68 guidance
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE GUIDANCE EXECUTIVE (GE) Review of TA68; The clinical and cost effectiveness of photodynamic therapy for age-related macular degeneration TA68 guidance
Instudies of vascular endothelial growth factor
 MONITORING THERAPEUTIC EFFICACY IN THE ANTI-VEGF ERA* SriniVas R. Sadda, MD ABSTRACT One of the most important questions with vascular endothelial growth factor (VEGF) inhibitors for age-related macular
MONITORING THERAPEUTIC EFFICACY IN THE ANTI-VEGF ERA* SriniVas R. Sadda, MD ABSTRACT One of the most important questions with vascular endothelial growth factor (VEGF) inhibitors for age-related macular
Class Update: Vascular Endothelial Growth Factors
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Bioimpedance Devices for Detection and Management of Lymphedema
 Bioimpedance Devices for Detection and Management of Lymphedema Policy Number: 2.01.82 Last Review: 5/2018 Origination: 1/2011 Next Review: 5/2019 Policy Blue Cross and Blue Shield of Kansas City (Blue
Bioimpedance Devices for Detection and Management of Lymphedema Policy Number: 2.01.82 Last Review: 5/2018 Origination: 1/2011 Next Review: 5/2019 Policy Blue Cross and Blue Shield of Kansas City (Blue
2018 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY. MEASURE TYPE: Process
 Quality ID #14 (NQF 0087): Age-Related Macular Degeneration (AMD): Dilated Macular Examination National Quality Strategy Domain: Effective Clinical Care 2018 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY
Quality ID #14 (NQF 0087): Age-Related Macular Degeneration (AMD): Dilated Macular Examination National Quality Strategy Domain: Effective Clinical Care 2018 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY
Clinical Policy Title: Ocular photodynamic therapy (OPDT) with Visudyne (verteporfin) for macular degeneration treatment
 Clinical Policy Title: Ocular photodynamic therapy (OPDT) with Visudyne (verteporfin) for macular degeneration treatment Clinical Policy Number: 10.02.04 Effective Date: January 1, 2016 Initial Review
Clinical Policy Title: Ocular photodynamic therapy (OPDT) with Visudyne (verteporfin) for macular degeneration treatment Clinical Policy Number: 10.02.04 Effective Date: January 1, 2016 Initial Review
Clinical Policy Title: Ocular photodynamic therapy with Visudyne (verteporfin) for macular degeneration treatment
 Clinical Policy Title: Ocular photodynamic therapy with Visudyne (verteporfin) for macular degeneration treatment Clinical Policy Number: 10.02.04 Effective Date: January 1, 2016 Initial Review Date: August
Clinical Policy Title: Ocular photodynamic therapy with Visudyne (verteporfin) for macular degeneration treatment Clinical Policy Number: 10.02.04 Effective Date: January 1, 2016 Initial Review Date: August
Nutrient/Nutritional Panel Testing
 Nutrient/Nutritional Panel Testing Policy Number: 2.04.136 Last Review: 10/2017 Origination: 10/2015 Next Review: 10/2018 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will not provide coverage
Nutrient/Nutritional Panel Testing Policy Number: 2.04.136 Last Review: 10/2017 Origination: 10/2015 Next Review: 10/2018 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will not provide coverage
Clinical Study Fixed Monthly versus Less Frequent Ranibizumab Dosing and Predictors of Visual Response in Exudative Age-Related Macular Degeneration
 Ophthalmology Volume 12, Article ID 69641, 8 pages doi:1.1155/12/69641 Clinical Study Fixed Monthly versus Less Frequent Ranibizumab Dosing and Predictors of Visual Response in Exudative Age-Related Macular
Ophthalmology Volume 12, Article ID 69641, 8 pages doi:1.1155/12/69641 Clinical Study Fixed Monthly versus Less Frequent Ranibizumab Dosing and Predictors of Visual Response in Exudative Age-Related Macular
Optical Coherence Tomography Monitoring Strategies for A-VEGF Treated Age-Related Macular Degeneration: An Evidence-Based Analysis
 Optical Coherence Tomography Monitoring Strategies for A-VEGF Treated Age-Related Macular Degeneration: An Evidence-Based Analysis G Pron August 2014 Ontario Health Technology Assessment Series; Vol. 14:
Optical Coherence Tomography Monitoring Strategies for A-VEGF Treated Age-Related Macular Degeneration: An Evidence-Based Analysis G Pron August 2014 Ontario Health Technology Assessment Series; Vol. 14:
A Treat and Extend Regimen Using Ranibizumab for Neovascular Age-Related Macular Degeneration
 A Treat and Extend Regimen Using Ranibizumab for Neovascular Age-Related Macular Degeneration Clinical and Economic Impact Omesh P. Gupta, MD, MBA, Gary Shienbaum, MD, Avni H. Patel, MD, Christopher Fecarotta,
A Treat and Extend Regimen Using Ranibizumab for Neovascular Age-Related Macular Degeneration Clinical and Economic Impact Omesh P. Gupta, MD, MBA, Gary Shienbaum, MD, Avni H. Patel, MD, Christopher Fecarotta,
The limited number of currently approved
 CLINICAL TRIALS OF VERTEPORFIN AND PEGAPTANIB: WHAT ARE THE RESULTS? * William F. Mieler, MD ABSTRACT Currently available treatment options for the management of choroidal neovascularization (CNV) in age-related
CLINICAL TRIALS OF VERTEPORFIN AND PEGAPTANIB: WHAT ARE THE RESULTS? * William F. Mieler, MD ABSTRACT Currently available treatment options for the management of choroidal neovascularization (CNV) in age-related
Management of Neovascular AMD
 Kapusta AMD Part 1 Management of Neovascular AMD Dr. Michael A. Kapusta, MD, FRCSC Ophthalmologist in Chief Jewish General Hospital Vitreoretinal Surgeon 1 FINANCIAL DISCLOSURES Consulting honoraria Bayer,
Kapusta AMD Part 1 Management of Neovascular AMD Dr. Michael A. Kapusta, MD, FRCSC Ophthalmologist in Chief Jewish General Hospital Vitreoretinal Surgeon 1 FINANCIAL DISCLOSURES Consulting honoraria Bayer,
Electronic Brachytherapy for Nonmelanoma Skin Cancer
 Electronic Brachytherapy for Nonmelanoma Skin Cancer Policy Number: 8.01.62 Last Review: 1/2018 Origination: 01/2016 Next Review: 1/2019 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will
Electronic Brachytherapy for Nonmelanoma Skin Cancer Policy Number: 8.01.62 Last Review: 1/2018 Origination: 01/2016 Next Review: 1/2019 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will
2018 OPTIONS FOR INDIVIDUAL MEASURES: CLAIMS ONLY. MEASURE TYPE: Process
 Quality ID #14 (NQF 0087): Age-Related Macular Degeneration (AMD): Dilated Macular Examination National Quality Strategy Domain: Effective Clinical Care 2018 OPTIONS FOR INDIVIDUAL MEASURES: CLAIMS ONLY
Quality ID #14 (NQF 0087): Age-Related Macular Degeneration (AMD): Dilated Macular Examination National Quality Strategy Domain: Effective Clinical Care 2018 OPTIONS FOR INDIVIDUAL MEASURES: CLAIMS ONLY
Dynamic Spinal Visualization and Vertebral Motion Analysis
 Dynamic Spinal Visualization and Vertebral Motion Analysis Policy Number: 6.01.46 Last Review: 2/2019 Origination: 2/2006 Next Review: 2/2020 Policy Blue Cross and Blue Shield of Kansas City (Blue KC)
Dynamic Spinal Visualization and Vertebral Motion Analysis Policy Number: 6.01.46 Last Review: 2/2019 Origination: 2/2006 Next Review: 2/2020 Policy Blue Cross and Blue Shield of Kansas City (Blue KC)
Eyelid Thermal Pulsation for the Treatment of Dry Eye Syndrome
 Eyelid Thermal Pulsation for the Treatment of Dry Eye Syndrome Policy Number: 9.03.29 Last Review: 12/2017 Origination: 9/2013 Next Review: 5/2018 Policy Blue Cross and Blue Shield of Kansas City (Blue
Eyelid Thermal Pulsation for the Treatment of Dry Eye Syndrome Policy Number: 9.03.29 Last Review: 12/2017 Origination: 9/2013 Next Review: 5/2018 Policy Blue Cross and Blue Shield of Kansas City (Blue
CDEC FINAL RECOMMENDATION
 CDEC FINAL RECOMMENDATION AFLIBERCEPT (Eylea Bayer Inc.) Indication: Wet Age-related Macular Degeneration Recommendation: The Canadian Drug Expert Committee (CDEC) recommends that aflibercept be listed
CDEC FINAL RECOMMENDATION AFLIBERCEPT (Eylea Bayer Inc.) Indication: Wet Age-related Macular Degeneration Recommendation: The Canadian Drug Expert Committee (CDEC) recommends that aflibercept be listed
These issues are covered in more detail below.
 26.3.07 Comments from Novartis on the Assessment Report for the Health Technology Appraisal of Pegaptinib and Ranibizumab for the treatment of age-related macular degeneration In general we feel that the
26.3.07 Comments from Novartis on the Assessment Report for the Health Technology Appraisal of Pegaptinib and Ranibizumab for the treatment of age-related macular degeneration In general we feel that the
Facts About Diabetic Eye Disease
 Facts About Diabetic Eye Disease Points to Remember 1. Diabetic eye disease comprises a group of eye conditions that affect people with diabetes. These conditions include diabetic retinopathy, diabetic
Facts About Diabetic Eye Disease Points to Remember 1. Diabetic eye disease comprises a group of eye conditions that affect people with diabetes. These conditions include diabetic retinopathy, diabetic
Peripheral Subcutaneous Field Stimulation
 Peripheral Subcutaneous Field Stimulation Policy Number: 7.01.139 Last Review: 9/2014 Origination: 7/2013 Next Review: 1/2015 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will not provide
Peripheral Subcutaneous Field Stimulation Policy Number: 7.01.139 Last Review: 9/2014 Origination: 7/2013 Next Review: 1/2015 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will not provide
Intracellular Micronutrient Analysis
 Intracellular Micronutrient Analysis Policy Number: 2.04.73 Last Review: 1/2019 Origination: 1/2013 Next Review: 1/2020 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will not provide coverage
Intracellular Micronutrient Analysis Policy Number: 2.04.73 Last Review: 1/2019 Origination: 1/2013 Next Review: 1/2020 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will not provide coverage
Despite our growing knowledge of the
 OVERVIEW OF AVAILABLE TREATMENTS FOR NEOVASCULAR AGE-RELATED MACULAR DEGENERATION * Carl D. Regillo, MD ABSTRACT Although potential treatments for neovascular age-related macular degeneration represent
OVERVIEW OF AVAILABLE TREATMENTS FOR NEOVASCULAR AGE-RELATED MACULAR DEGENERATION * Carl D. Regillo, MD ABSTRACT Although potential treatments for neovascular age-related macular degeneration represent
International Journal of Health Sciences and Research ISSN:
 International Journal of Health Sciences and Research www.ijhsr.org ISSN: 2249-9571 Original Research Article A Multivariate Analysis of Intravitreal Injection of Anti-VEGF Bevacizumab in the Treatment
International Journal of Health Sciences and Research www.ijhsr.org ISSN: 2249-9571 Original Research Article A Multivariate Analysis of Intravitreal Injection of Anti-VEGF Bevacizumab in the Treatment
The College of Optometrists - Learning outcomes for the Professional Certificate in Medical Retina
 Learning outcomes for the Professional Certificate in Medical Retina, incorporating diabetic retinopathy screening and age related macular degeneration The professional certificate is a prerequisite to
Learning outcomes for the Professional Certificate in Medical Retina, incorporating diabetic retinopathy screening and age related macular degeneration The professional certificate is a prerequisite to
Ophthalmology Macular Pathways
 Ophthalmology Macular Pathways Age related Macular Degeneration Diabetic Macular Oedema Macular Oedema secondary to Central Retinal Macular Oedema secondary to Branch Retinal CNV associated with pathological
Ophthalmology Macular Pathways Age related Macular Degeneration Diabetic Macular Oedema Macular Oedema secondary to Central Retinal Macular Oedema secondary to Branch Retinal CNV associated with pathological
Opthea Initiates OPT-302 Diabetic Macular Edema Clinical Trial
 ASX and Media Release 3 January 2018 Opthea Initiates OPT-302 Diabetic Macular Edema Clinical Trial Melbourne, Australia; January 3 2018 Opthea Limited (ASX:OPT), a late stage biopharmaceutical company
ASX and Media Release 3 January 2018 Opthea Initiates OPT-302 Diabetic Macular Edema Clinical Trial Melbourne, Australia; January 3 2018 Opthea Limited (ASX:OPT), a late stage biopharmaceutical company
Biofeedback as a Treatment of Headache
 Biofeedback as a Treatment of Headache Policy Number: 2.01.29 Last Review: 7/2018 Origination: 7/2008 Next Review: 7/2019 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) may provide coverage
Biofeedback as a Treatment of Headache Policy Number: 2.01.29 Last Review: 7/2018 Origination: 7/2008 Next Review: 7/2019 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) may provide coverage
Diagnosis and treatment of diabetic retinopathy. Blake Cooper MD Ophthalmologist Vitreoretinal Surgeon Retina Associates Kansas City
 Diagnosis and treatment of diabetic retinopathy Blake Cooper MD Ophthalmologist Vitreoretinal Surgeon Retina Associates Kansas City Disclosures Consulted for Novo Nordisk 2017,2018. Will be discussing
Diagnosis and treatment of diabetic retinopathy Blake Cooper MD Ophthalmologist Vitreoretinal Surgeon Retina Associates Kansas City Disclosures Consulted for Novo Nordisk 2017,2018. Will be discussing
Abicipar pegol for wet age related macular degeneration
 EVIDENCE BRIEFING AUGUST 2018 Abicipar pegol for wet age related macular degeneration NIHRIO ID 7084 NICE ID 9724 Developer/Company Allergan Ltd and Molecular Partners UKPS ID 646978 Licensing and market
EVIDENCE BRIEFING AUGUST 2018 Abicipar pegol for wet age related macular degeneration NIHRIO ID 7084 NICE ID 9724 Developer/Company Allergan Ltd and Molecular Partners UKPS ID 646978 Licensing and market
EU Regulatory workshop Ophthalmology clinical development and scientific advice. Industry view on DME and macular edema secondary to RVO
 EU Regulatory workshop Ophthalmology clinical development and scientific advice. Industry view on DME and macular edema secondary to RVO Yehia Hashad, M.D. Vice President and Global Therapeutic Area Head
EU Regulatory workshop Ophthalmology clinical development and scientific advice. Industry view on DME and macular edema secondary to RVO Yehia Hashad, M.D. Vice President and Global Therapeutic Area Head
Eyelid Thermal Pulsation for the Treatment of Dry Eye Syndrome
 Eyelid Thermal Pulsation for the Treatment of Dry Eye Syndrome Policy Number: 9.03.29 Last Review: 5/2018 Origination: 9/2013 Next Review: 11/2018 Policy Blue Cross and Blue Shield of Kansas City (Blue
Eyelid Thermal Pulsation for the Treatment of Dry Eye Syndrome Policy Number: 9.03.29 Last Review: 5/2018 Origination: 9/2013 Next Review: 11/2018 Policy Blue Cross and Blue Shield of Kansas City (Blue
Subgroup Analysis of the MARINA Study of Ranibizumab in Neovascular Age-Related Macular Degeneration
 Subgroup Analysis of the MARINA Study of in Neovascular Age-Related Macular Degeneration David S. Boyer, MD, 1 Andrew N. Antoszyk, MD, 2 Carl C. Awh, MD, 3 Robert B. Bhisitkul, MD, PhD, 4 Howard Shapiro,
Subgroup Analysis of the MARINA Study of in Neovascular Age-Related Macular Degeneration David S. Boyer, MD, 1 Andrew N. Antoszyk, MD, 2 Carl C. Awh, MD, 3 Robert B. Bhisitkul, MD, PhD, 4 Howard Shapiro,
Michael P. Blair, MD Retina Consultants, Ltd Libertyville/Des Plaines, Illinois Clinical Associate University of Chicago 17 October 2015
 Michael P. Blair, MD Retina Consultants, Ltd Libertyville/Des Plaines, Illinois Clinical Associate University of Chicago 17 October 2015 So What Parts of the Eye Retina are Affected by VHL Neural tissue
Michael P. Blair, MD Retina Consultants, Ltd Libertyville/Des Plaines, Illinois Clinical Associate University of Chicago 17 October 2015 So What Parts of the Eye Retina are Affected by VHL Neural tissue
Fundamentals of Retina Coding
 Fundamentals of Retina Coding Presented by: Joy Woodke, COE, OCS Sunday, April 2, 2017 ASRS Business of Retina Meeting Dallas, TX American Academy of Ophthalmic Executives Financial Disclosure Joy Woodke,
Fundamentals of Retina Coding Presented by: Joy Woodke, COE, OCS Sunday, April 2, 2017 ASRS Business of Retina Meeting Dallas, TX American Academy of Ophthalmic Executives Financial Disclosure Joy Woodke,
Surgical Treatment of Bilateral Gynecomastia
 Surgical Treatment of Bilateral Gynecomastia Policy Number: 7.01.13 Last Review: 4/2018 Origination: 4/2006 Next Review: 4/2019 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will provide coverage
Surgical Treatment of Bilateral Gynecomastia Policy Number: 7.01.13 Last Review: 4/2018 Origination: 4/2006 Next Review: 4/2019 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will provide coverage
VERTEPORFIN IN PHOTODYNAMIC THERAPY STUDY GROUP
 Verteporfin Therapy of Subfoveal Choroidal Neovascularization in Age-related Macular Degeneration: Two-year Results of a Randomized Clinical Trial Including Lesions With Occult With No Classic Choroidal
Verteporfin Therapy of Subfoveal Choroidal Neovascularization in Age-related Macular Degeneration: Two-year Results of a Randomized Clinical Trial Including Lesions With Occult With No Classic Choroidal
Vascular Endothelial Growth Factor (VEGF) Inhibitors Ocular Use Drug Class Monograph (Medical Benefit)
 Vascular Endothelial Growth Factor (VEGF) Inhibitors Ocular Use Drug Class Monograph (Medical Benefit) Line of Business: Medi-Cal Effective Date: May 17, 2017 Revision Date: May 17, 2017 This policy has
Vascular Endothelial Growth Factor (VEGF) Inhibitors Ocular Use Drug Class Monograph (Medical Benefit) Line of Business: Medi-Cal Effective Date: May 17, 2017 Revision Date: May 17, 2017 This policy has
Intravitreal Corticosteroid Implants
 Intravitreal Corticosteroid Implants Policy Number: 9.03.23 Last Review: 4/2018 Origination: 07/2015 Next Review: 4/2019 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will provide coverage
Intravitreal Corticosteroid Implants Policy Number: 9.03.23 Last Review: 4/2018 Origination: 07/2015 Next Review: 4/2019 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will provide coverage
Age-related macular degeneration (AMD) and RANIBIZUMAB (Lucentis)
 Aggiornamento biotecnologico: MoAb anti VEGF Age-related macular degeneration (AMD) and RANIBIZUMAB (Lucentis) Giulia Germena AMD DRY (non-neovascular) Atrophic cell death in the macula No leakage WET
Aggiornamento biotecnologico: MoAb anti VEGF Age-related macular degeneration (AMD) and RANIBIZUMAB (Lucentis) Giulia Germena AMD DRY (non-neovascular) Atrophic cell death in the macula No leakage WET
ANTIVASCULAR ENDOTHELIAL GROWTH FACTOR
 THE ROLE OF RANIBIZUMAB * Michael L. Klein, MD ABSTRACT The positive 1-year results of ranibizumab trials reported at the 23rd Annual Meeting of the American Society of Retina Specialists have generated
THE ROLE OF RANIBIZUMAB * Michael L. Klein, MD ABSTRACT The positive 1-year results of ranibizumab trials reported at the 23rd Annual Meeting of the American Society of Retina Specialists have generated
Optical Coherence Tomography: Pearls for the Anterior Segment Surgeon Basic Science Michael Stewart, M.D.
 Optical Coherence Tomography: Pearls for the Anterior Segment Surgeon Basic Science Michael Stewart, M.D. Disclosure OCT Optical Coherence Tomography No relevant financial relationships I will refer to
Optical Coherence Tomography: Pearls for the Anterior Segment Surgeon Basic Science Michael Stewart, M.D. Disclosure OCT Optical Coherence Tomography No relevant financial relationships I will refer to
Antigen Leukocyte Antibody Test
 Antigen Leukocyte Antibody Test Policy Number: 2.01.93 Last Review: 4/2018 Origination: 4/2014 Next Review: 4/2019 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will not provide coverage for
Antigen Leukocyte Antibody Test Policy Number: 2.01.93 Last Review: 4/2018 Origination: 4/2014 Next Review: 4/2019 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will not provide coverage for
Eylea (aflibercept) Document Number: IC-0026
 Eylea (aflibercept) Document Number: IC-0026 Last Review Date: 3/1/2018 Date of Origin: 02/07/2013 Dates Reviewed: 03/07/2013, 06/2013, 09/2013, 12/2013, 03/2014, 06/2014, 09/2014, 12/2014, 03/2015, 04/2015,
Eylea (aflibercept) Document Number: IC-0026 Last Review Date: 3/1/2018 Date of Origin: 02/07/2013 Dates Reviewed: 03/07/2013, 06/2013, 09/2013, 12/2013, 03/2014, 06/2014, 09/2014, 12/2014, 03/2015, 04/2015,
Vosevi (sofosbuvir/velpatasvir/voxilaprevir)
 Vosevi (sofosbuvir/velpatasvir/voxilaprevir) Policy Number: 5.01.646 Last Review: 10/2017 Origination: 10/2017 Next Review: 11/2018 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will provide
Vosevi (sofosbuvir/velpatasvir/voxilaprevir) Policy Number: 5.01.646 Last Review: 10/2017 Origination: 10/2017 Next Review: 11/2018 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will provide
Corporate Medical Policy
 Corporate Medical Policy Retinal Prosthesis File Name: Origination: Last CAP Review: Next CAP Review: Last Review: retinal_prosthesis 6/2011 6/2018 6/2019 6/2018 Description of Procedure or Service A retinal
Corporate Medical Policy Retinal Prosthesis File Name: Origination: Last CAP Review: Next CAP Review: Last Review: retinal_prosthesis 6/2011 6/2018 6/2019 6/2018 Description of Procedure or Service A retinal
Transtympanic Micropressure Applications as a Treatment of Meniere s Disease
 Transtympanic Micropressure Applications as a Treatment of Meniere s Disease Policy Number: 1.01.23 Last Review: 8/2017 Origination: 2/2006 Next Review: 8/2018 Policy Blue Cross and Blue Shield of Kansas
Transtympanic Micropressure Applications as a Treatment of Meniere s Disease Policy Number: 1.01.23 Last Review: 8/2017 Origination: 2/2006 Next Review: 8/2018 Policy Blue Cross and Blue Shield of Kansas
 measure of your overall performance. An isolated glucose test is helpful to let you know what your sugar level is at one moment, but it doesn t tell you whether or not your diabetes is under adequate control
measure of your overall performance. An isolated glucose test is helpful to let you know what your sugar level is at one moment, but it doesn t tell you whether or not your diabetes is under adequate control
Ophthalmologic Policy. Vascular Endothelial Growth Factor (VEGF) Inhibitors
 Ophthalmologic Policy UnitedHealthcare Commercial Drug Policy Vascular Endothelial Growth Factor (VEGF) Inhibitors Policy Number: 2016D0042H Effective Date: October 1, 2016 Table of Contents Page INSTRUCTIONS
Ophthalmologic Policy UnitedHealthcare Commercial Drug Policy Vascular Endothelial Growth Factor (VEGF) Inhibitors Policy Number: 2016D0042H Effective Date: October 1, 2016 Table of Contents Page INSTRUCTIONS
CDEC FINAL RECOMMENDATION
 CDEC FINAL RECOMMENDATION RANIBIZUMAB (Lucentis Novartis Pharmaceuticals Canada Inc.) New Indication: Macular Edema Secondary to Retinal Vein Occlusion Recommendation: The Canadian Drug Expert Committee
CDEC FINAL RECOMMENDATION RANIBIZUMAB (Lucentis Novartis Pharmaceuticals Canada Inc.) New Indication: Macular Edema Secondary to Retinal Vein Occlusion Recommendation: The Canadian Drug Expert Committee
Ranibizumab and pegaptanib for the treatment of agerelated macular degeneration
 Ranibizumab and pegaptanib for the treatment of agerelated Issued: August 2008 last modified: May 2012 guidance.nice.org.uk/ta155 NHS Evidence has accredited the process used by the Centre for Health Technology
Ranibizumab and pegaptanib for the treatment of agerelated Issued: August 2008 last modified: May 2012 guidance.nice.org.uk/ta155 NHS Evidence has accredited the process used by the Centre for Health Technology
CMS Manual System Pub Medicare National Coverage Determinations
 CMS Manual System Pub. 100-03 Medicare National Coverage Determinations Department of Health & Human Services (DHHS) Centers for Medicare & Medicaid Services (CMS) Transmittal 9 Date: APRIL 1, 2004 CHANGE
CMS Manual System Pub. 100-03 Medicare National Coverage Determinations Department of Health & Human Services (DHHS) Centers for Medicare & Medicaid Services (CMS) Transmittal 9 Date: APRIL 1, 2004 CHANGE
Clinically Significant Macular Edema (CSME)
 Clinically Significant Macular Edema (CSME) 1 Clinically Significant Macular Edema (CSME) Sadrina T. Shaw OMT I Student July 26, 2014 Advisor: Dr. Uwaydat Clinically Significant Macular Edema (CSME) 2
Clinically Significant Macular Edema (CSME) 1 Clinically Significant Macular Edema (CSME) Sadrina T. Shaw OMT I Student July 26, 2014 Advisor: Dr. Uwaydat Clinically Significant Macular Edema (CSME) 2
VASCULAR ENDOTHELIAL GROWTH FACTOR INHIBITORS
 VASCULAR ENDOTHELIAL GROWTH FACTOR INHIBITORS FOR OPHTHALMIC USE Policy Number: 2016D0001B Effective Date: January 1, 2016 Table of Contents: Page: Cross Reference Policy: POLICY DESCRIPTION 2 ZALTRAP
VASCULAR ENDOTHELIAL GROWTH FACTOR INHIBITORS FOR OPHTHALMIC USE Policy Number: 2016D0001B Effective Date: January 1, 2016 Table of Contents: Page: Cross Reference Policy: POLICY DESCRIPTION 2 ZALTRAP
Digital Journal of Ophthalmology Digital Journal of Ophthalmology. Abstract
 Original Articles Combined intravitreal ranibizumab and verteporfin photodynamic therapy versus ranibizumab alone for the treatment of age-related macular degeneration Rosalia Giustolisi, MD, Nicoletta
Original Articles Combined intravitreal ranibizumab and verteporfin photodynamic therapy versus ranibizumab alone for the treatment of age-related macular degeneration Rosalia Giustolisi, MD, Nicoletta
Corporate Medical Policy
 Corporate Medical Policy Intravitreal Implant File Name: Origination: Last CAP Review: Next CAP Review: Last Review: intravitreal_implant 11/2010 6/2017 6/2018 6/2017 Description of Procedure or Service
Corporate Medical Policy Intravitreal Implant File Name: Origination: Last CAP Review: Next CAP Review: Last Review: intravitreal_implant 11/2010 6/2017 6/2018 6/2017 Description of Procedure or Service
NEOVASCULAR AGE-RELATED MACULAR DEGENERation
 Randomized, Double-Masked, -Controlled Trial of Ranibizumab for Neovascular Age-Related Macular Degeneration: PIER Study Year 2 PREMA ABRAHAM, HUIBIN YUE, AND LAURA WILSON PURPOSE: To evaluate efficacy
Randomized, Double-Masked, -Controlled Trial of Ranibizumab for Neovascular Age-Related Macular Degeneration: PIER Study Year 2 PREMA ABRAHAM, HUIBIN YUE, AND LAURA WILSON PURPOSE: To evaluate efficacy
Vision loss in elderly
 Vision loss in elderly Erica Weir, February 2016 1 Burden Enter nursing homes 3 years earlier Twice the risk of falling 4x the risk of hip fracture Independent risk factor for delirium What are the leading
Vision loss in elderly Erica Weir, February 2016 1 Burden Enter nursing homes 3 years earlier Twice the risk of falling 4x the risk of hip fracture Independent risk factor for delirium What are the leading
The Foundation WHAT IS THE RETINA?
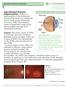 Age-Related Macular Degeneration (AMD) is a deterioration of the retina and choroid that leads to a substantial loss in visual acuity (sharpness of vision). AMD is the leading cause of significant visual
Age-Related Macular Degeneration (AMD) is a deterioration of the retina and choroid that leads to a substantial loss in visual acuity (sharpness of vision). AMD is the leading cause of significant visual
Prevention and treatment of agerelated macular degeneration
 THERAPY REVIEW Prevention and treatment of agerelated macular degeneration SARAH HORTON AND CATHERINE GULY Age-related macular degeneration (AMD) is a common cause of visual loss in older people and GPs
THERAPY REVIEW Prevention and treatment of agerelated macular degeneration SARAH HORTON AND CATHERINE GULY Age-related macular degeneration (AMD) is a common cause of visual loss in older people and GPs
The Natural History of Occult Choroidal Neovascularisation Associated With Age-related Macular Degeneration. A Systematic Review
 Review Article 145 The Natural History of Occult Choroidal Neovascularisation Associated With Age-related Macular Degeneration. A Systematic Review Antonio Polito, 1 MD, Miriam Isola, 2 MHS, Paolo Lanzetta,
Review Article 145 The Natural History of Occult Choroidal Neovascularisation Associated With Age-related Macular Degeneration. A Systematic Review Antonio Polito, 1 MD, Miriam Isola, 2 MHS, Paolo Lanzetta,
Use of Aflibercept in cases of recurrent or refractory neovascular AMD previously treated with other antiangiogenic drug: anatomo-functional outcomes
 Use of Aflibercept in cases of recurrent or refractory neovascular AMD previously treated with other antiangiogenic drug: anatomo-functional outcomes Daniel E. Charles, MD, Martin Charles, MD, Gisela Jelusich,
Use of Aflibercept in cases of recurrent or refractory neovascular AMD previously treated with other antiangiogenic drug: anatomo-functional outcomes Daniel E. Charles, MD, Martin Charles, MD, Gisela Jelusich,
