Heterogeneous pathogenesis of LGMD2: consequences for therapy Abstract Key words
|
|
|
- Ella Terry
- 5 years ago
- Views:
Transcription
1 Heterogeneous pathogenesis of LGMD2: consequences for therapy Corrado Angelini, Lucia Nardetto, Marina Fanin, Anna Chiara Nascimbeni, Elisabetta Tasca Department of Neurosciences, University of Padova and Venetian Institute of Molecular Medicine, Padova, Italy Abstract Autosomal recessive limb girdle muscular dystrophies (LGMD type 2) are a clinically and genetically heterogeneous group of disorders, including at least 13 different genetic entities, which are characterized by progressive involvement and wasting of proximal limb girdle muscles. All LGMD2 seem to be present worldwide, though the relative frequency of the different types is variable. The gold standard for LGMD diagnosis is the study of protein involved either by immunoblot or immunohistochemistry on the basis of clinical phenotypes, followed by a mutation study in candidate genes. Pathogenetic mechanisms are various; a different pathophysiology implies different clinical muscle involvement and might require a differential therapeutical approach. Key words: LGMD2, phenotype, therapy. The diagnosis of recessive LGMD is complicated: the variability of clinical phenotype, the wide effort required in the identification of point mutations in relatively large genes, makes this target a hard task. The molecular diagnosis of LGMD type 2, which has become available only recently due to the progress of molecular genetics and the availability of antibodies for protein testing, is the necessary step to select patients for future clinical and experimental trials with drugs or gene therapy. The wide genetic heterogeneity of recessive LGMD implies a number of different pathogenetic mechanisms, which characterize each different genetic disease. Until a genetic characterization of LGMD patients is obtained, any therapeutic intervention can only be targeted to counter-act some of the general aspects of the muscular dystrophy rather than to the specific defective pathway. LGMD2A or calpainopathy LGMD type 2A (LGMD2A) is the most common form of LGMD in European countries, (Table 1) where it represents the 40% of LGMD and affects about 1:100,000 inhabitants (21, 37, 41, 44, 50). LGMD2A is caused by mutations in the CAPN3 gene, encoding for a muscle-specific member of a family of Ca ++ -activated neutral proteases, and binds to titin (30, 43, 46, 47). However, the search for the specific substrates of calpain-3 has been so far unsuccessful. Several interesting lines of research have investigated the pathogenesis of calpainopathy: calpain-3 could be involved in the regulation of transcription factors controlling survival genes and apoptosis (4), or in the degradation and disassembly of cytoskeletal or myofibrillar proteins (sarcomere remodeling) (28, 30, 31, 42, 48). In calpainopathy a marked clinical heterogeneity has been observed considering the age and the phenotype at onset and the disease progression, even in patients with the same gene mutation. On this basis, according to the original clinical descriptions of LGMDs, clinical phenotype of LGMD2A can be subdivided in the pelvifemoral form of Leyden-Moebius (with prevalent proximal muscle weakness in lower girdle) and the scapulo-humeral form of Erb (with prevalent proximal muscle weakness in upper girdle) (51); the onset may be early (< 12 years old), typical (between 12 and 30 years old) and late (> 30 years old). The loss of ambulation occurs usually years after the onset. The variability of both the clinical phenotype and disease course may be only partly attributable to the genotype. While null type gene mutations are usually associated with absent calpain-3 protein in muscle and severe phenotype, missense type mutations (which account for about 70% of mutations in this gene) are associated with an extreme unpredictability of their effect at both the protein and the phenotype levels (20, 41, 45), suggesting that additional and still unknown genetic or environmental factors may be playing a role in modulating the phenotype
2 Table 1: Autosomal recessive limb girdle muscular dystrophies TYPE LOCUS PROTEIN PROTEIN FUNCTION LGMD2A 15q15.1 calpain-3 Protease LGMD2B 2p13 dysferlin Membrane repair LGMD2C 13q12 γ-sarcoglycan SG complex LGMD2D 17q21 α-sarcoglycan SG complex LGMD2E 4q12 β-sarcoglycan SG complex LGMD2F 5q33 δ-sarcoglycan SG complex LGMD2G 17q11-12 telethonin Cytoskeleton organization LGMD2H 9q31-33 TRIM-32 Ubiquitin ligase LGMD2I 19q13.3 FKRP Glycosyl Transferase LGMD2J 2q31 titin Structural protein LGMD2K 9q34.1 POMT1 LGMD 2B or dysferlinopathy Limb-girdle muscular dystrophy type 2B (LGMD2B), and the distal muscular dystrophy named Miyoshi myopathy (MM), are caused by mutations in dysferlin gene, mapped to chromosome region 2p13 (34). Dysferlin immunolocalizes to the sarcolemma similarly to dystrophin, but it does not associate with dystrophinglycoprotein complex. This protein presents a high degree of sequence homology with the protein Fer-1 in C. elegans. Since the mutant worm has an abnormal spermatogenesis, it was argued that mutant fer-1 may be involved in the failure of membrane fusion between the organelles and plasmalemma. By analogy, the pathogenetic mechanism in dysferlinopathies is correlated to the abnormal trafficking of vesicles, which repair muscle fiber membrane (6, 12). The absence of dysferlin causes a failure of damaged muscle fibers repair, possibly impairing the recovery from exercise induced damage. In many patients with LGMD2B the initial symptoms occur abruptly after age 20, sometimes following a regular heavy exercise, which damages especially the biarticular muscles. The clinician should discourage these patients from performing heavy exercise, which might exacerbate muscle breakdown. Creatine kinase is usually markedly elevated at presentation (often times or even more above the normal range) (35). The anterior distal leg muscles and the distal arm muscles are relatively spared even in the later stages of the disease, and, in contrast with Glycosyl Transferase Glycosyl LGMD2L 9q31 fukutin Transferase LGMD2M 11p13-p12?? calpainopathy, scapular involvement is mild and absent at the onset (10). Other important clinical clues are the early inability to walk on tiptoes and the early involvement of the medial gastrocnemius muscle. Despite the clinical features of LGMD2B and MM are quite different, both phenotypes can be detected among patients belonging to the same family, thus sharing the same mutations (6). This clinical heterogeneity might be attributed to additional epigenetic factors resulting in variable dysferlin expression. Gene expression profiling has been recently used to investigate modifier genes in dysferlinopathy (11). Several studies reported a prominent inflammatory response in dysferlinopathy patients, but the origin of this feature and its role in the development of muscle pathology is still under investigation. Patients treated with steroids or immunosuppressant drugs do not show benefit on the long term, therefore it seems advisable to try alternative drugs, since this muscular dystrophy develops in relation to a defective sarcolemmal repair. LGMD 2C 2F or sarcoglycanopathies The sarcoglycanopathies are due to mutations in the genes encoding for the components of the sarcoglycan (SG) complex. The SG complex, composed of 5 glycoproteins (α-, β-, γ-, δ-, ε-sg), is a member of the dystrophin-associated glycoprotein (DAG) complex localized to the sarcolemma of muscle fibers, which acts as a link between the extracellular matrix and the cytoskeleton, confers structural stability and protects the sarcolemma from mechanical stress developed during muscle contraction. This molecular complex can also participate in the transmission of various signals from the inside to the outside of the cell and vice-versa. The pathogenetic mechanism underlying sarcolemmal damage and fiber necrosis in sarcoglycanopathies is related to the fact that the deficiency in any SG subunit results in a loss of the whole SG complex, causing the disruption of the DAG complex and the inability to counteract the mechanical stress of contractile activity. One hypothesis is that SG complex masks a metalloproteinase cleavage site of the β-dystroglycan in normal muscle: the loss of SG in sarcoglycanopathies would lead to activation of the cleavage site of β- dystroglycan, causing the disruption of the link between the sarcolemma and the basement membrane. The clinical phenotype of sarcoglycanopathies ranges from a severe Duchenne-like dystrophy to a relatively mild LGMD (2, 3). α- and γ-sarcoglycanopathy are characterized by presentation with proximal lower limb weakness, predominance of childhood onset, frequent calf hypertrophy, usually rare cardiac complications and normal intelligence. In the early-onset cases, loss of ambulation usually occurs within the second decade. β- sarcoglycanopathy is characterized by broad range of age at onset and clinical severity, while δ-sarcoglycanopathy usually presents a very severe course. The
3 cardiac involvement, which represents an important and sometimes life-threatening clinical complication, is often observed in association with mutations of β-sg (19) and δ-sg genes, whereas patients with mutations in the α-sg gene only rarely display cardiomyopathy. Recent studies demonstrate that β-sg and δ-sg are both expressed in striated muscle and smooth muscle of coronary arteries, suggesting that a perturbation of coronary vascular function might represent a common mechanism in the pathogenesis of the cardiomyopathy. LGMD2G LGMD2G results from mutation in the gene for the sarcomeric protein telethonin (38). Telethonin is a protein associated with the Z-disk region that seems to be involved in the reorganization of the cytoskeleton during myofibrillogenesis and in the inhibition of the secretion of myostatin, a negative regulator of skeletal muscle growth (36, 39). Patients with telethoninopathy usually present a severe involvement of the proximal muscles of the upper and lower limbs but also a marked weakness of the distal muscles of legs: tibialis anterior muscle is early affected, leading to foot drop and to difficulty with walking on the heels. CK levels are mildly elevated and muscle biopsy shows rimmed vacuoles (38). LGMD2H LGMD2H is a mild muscular dystrophy firstly identified in the Manitoba Hutterite community of Canada that is caused by mutations in the gene encoding TRIM32 (23). TRIM32 is a member of the tripartite motif (TRIM) family, characterized by the presence of three motifs: a RING-finger domain, a B1 box and a coiled coil domain. TRIM32 is an E3-ubiquitin ligase, which marks proteins for degradation by the proteasome pathway and contains six NHL domains that could be involved in protein-protein interactions and could confer specifity in protein selection. The missense mutation in TRIM32 identified in LGMD2H does not abolish its E3 ubiquitin ligase activity but seems to change its interaction with a yet unknown substrate (the wrong protein target or aberrant accumulation of a protein normally degraded) (14, 32, 33). LGMD2I-2K-2L or dystroglycanopathies This group of LGMD is due to mutations in the genes encoding various glycosyl-transferases that are believed to be Golgi resident. They share the same defective glycosylation of α-dystroglycan, causing an abnormal interaction with laminin-α2 and other extracellular matrix proteins. Dystroglycanopathies are a group of disorders whose phenotype range from severe congenital muscular dystrophy to relatively mild LGMD. LGMD2I is caused by mutation in FKRP gene (15) and it is a milder allelic variant of congenital muscular dystrophy type 1C (MDC1C) (9). Recent studies suggest that the severe phenotype in MDC1C is due to the ER-retention of mutant FKRP, while in LGMD2I the protein is still able to reach the Golgi complex (17, 18). The spectrum of LGMD2I phenotypes ranges from Duchenne-like forms with cardiomyopathy, to milder forms with a relatively benign course. LGMD2K is caused by mutations in POMT1 gene, which have originally been identified in patients with Walker-Warburg syndrome, the most severe form of MDC. Recently, a distinct LGMD phenotype, resulting from a common mutation, has been described in Turkish population (5). LGMD2K patients present LGMD, mental retardation, microcephaly, but without eye abnormalities (13). LGMD2L is caused by mutation in fukutin gene (24) and it is considered a milder variant of Fukuyama congenital muscular dystrophy. LGMD2L is characterized by an acute and severe onset of muscle weakness, and drugs response. LGMD2J LGMD2J is caused by missense mutation in the titin gene and it is associated with secondary calpain-3 defects (26, 27). It has been so far reported only in the Finnish population, and when a single allele is mutated, the disease manifests itself as a dominant disorder, the Tibial Muscular Dystrophy. Conclusions and future prospectives for treatment Since different pathogenetic mechanisms are at play in the different types of LGMD, it is conceivable that different types of therapeutic intervention(s) should be required to slow down muscle destruction (Table 2). Nowadays the main importance of establishing an exact molecular diagnosis in individual patients with dystrophy is genetic counseling, but in the future the discovery of different pathogenetic mechanism might suggest differential therapeutic strategies. Up to now, several preclinical studies have been conducted: Experimental gene transfer: gene replacement has been tried in sarcoglycanopathies and in calpainopathy using non viral and viral vectors (adenoviruses and AAV) (1, 7, 22). Even if several experiments have reached preclinical proof of efficiency and persistency of gene transfer in muscle tissues, for gene transfer in humans there are many difficulties to be overcome, like the host immune response against the vector or the gene (53), the necessity of performing the vector injection before the onset of the dystrophic pattern (1), and the difficulty to obtain an efficient delivery in such a large amount of tissue. Cell therapy: several sources of stem cells or early precursor cells have been identified, and the possibility of restoring muscle function by providing cells capable of regenerating damaged tissues has been tested in sarcoglycanopathies and in dysferlinopathies (25)
4 Table 2. Pathogenesis of LGMD2: consequences for therapy TYPE PATHOGENETIC MECHANISM EXPERIMENTED THERAPEUTIC INTERVENTIONS LGMD2A Loss of proteolytic activity Gene replacement/ inhibitors of myostatin LGMD2B Defective membrane repair Cell therapy/ to inhibit the complement membrane attack complex LGMD2C Defective membrane integrity Gene replacement/ Cell therapy/ Drugs to inhibit the proteasome LGMD2D Defective membrane integrity Gene replacement/ Cell therapy / Steroids/ inhibitors of myostatin/ up regulation of other proteins/ Drugs to inhibit the proteasome LGMD2E Defective membrane integrity Gene replacement/ Cell therapy/ Drugs to inhibit the proteasome LGMD2F Defective membrane integrity Gene replacement/ Cell therapy/ Drugs to inhibit the proteasome LGMD2G Abnormal Z-line assembly? LGMD2H Abnormal ubiquitin-proteasome system? LGMD2I Defects in proteins glycosylation? LGMD2J Abnormal sarcomeric assembly? LGMD2K Defects in proteins glycosylation? LGMD2L Defects in proteins glycosylation Steroids LGMD2M Loss of proteolytic activity? Pharmacological therapies: a recent study reported the beneficial effect of steroids on LGMD2L, and there is evidence of a beneficial effect on LGMD2D (16). Other possible therapeutic approaches are focused to: 1) to counteract muscle wasting enhancing muscle mass, using inhibitors of myostatin in LGMD2A and in LGMD2D (8, 40); 2) to up-regulate of proteins that can compensate the deficiency of the mutated ones (for example ε- sarcoglycan for α-sarcoglycan (49); 3) to inhibit the down regulation of SG-complex into the proteasome or blocking Calcium with the consequent activation of ubiquitous calpains (29); 4) to inhibit the complement membrane attack complex in dysferlinopathies, where decay-accelerating factor is down-regulated (52). Acknowledgments The research in this study was funded by a grant from the Telethon-Italy (GUP to M.F.). We also acknowledge the support from the AFM - Association Française contre le Myopathies ( /12926 to M.F. and /12988 to C.A.), the Telethon-Italy (GTF05003 to C.A.), the Eurobiobank network (QLRT to C.A.) and the Italian Ministry for University and Research (MIUR, project COFIN 2006/ to C.A.). Address correspondence to: Prof. C. Angelini, Department of Neurosciences, via Giustiniani 5, Padova, Italy. Tel Fax corrado.angelini@unipd.it References [1] Allamand V, Donahue KM, Straub V, Davisson RL, Davidson BL, Campbell KP: Early adenovirus-mediated gene transfer effectively prevents muscular dystrophy in alpha-sarcoglycan-deficient mice. Gene Ther. 2000; 7: [2] Angelini C, Fanin M, Menegazzo E, Freda MP, Duggan DJ, Hoffman EP: Homozygous α- sarcoglycan mutation in two siblings: one asymptomatic and one steroid responsive mild limb-girdle muscular dystrophy patient. Muscle & Nerve 1998; 21: [3] Angelini C, Fanin M, Freda MP, Duggan DJ, Siciliano G, Hoffman EP: The clinical spectrum of sarcoglycanopathies. Neurology 1999; 52: [4] Baghdiguian S, Martin M, Richard I, Pons F, Astier C, Bourg N, Hay RT, Chemaly R, Halaby G, Loiselet J, Anderson LVB, de Munain AL, Fardeau M, Mangeat P, Beckmann JS, Lefranc G: Calpain 3 deficiency is associated with myonuclear apoptosis and profound perturbation of the IkBα/NF-kB pathway in LGMD type 2A. Nature Med. 1999; 5: [5] Balci B, Uyanik G, Dincer P, Gross C, Willer T, Talim B, Haliloglu G, Kale G, Hehr U, Winkler J, Topaloglu H: An autosomal recessive limb girdle muscular dystrophy (LGMD2) with mild mental retardation is allelic to Walker-Warburg
5 syndrome (WWS) caused by a mutation in the POMT1 gene. Neuromusc. Disord. 2005; 15: [6] Bansal D, Miyake K, Vogel SS, Groh S, Chen CC, Williamson R, McNeil PL, Campbell KP: Defective membrane repair in dysferlin-deficient muscular dystrophy. Nature 2003; 423: [7] Bartoli M, Roudaut C, Martin S, Fougerousse F, Suel L, Puopiot J, Gicquel E, Noulet F, Danos O, Richard I: Safety and efficacy of AAV-mediated calpain 3 gene transfer in a mouse model of limb girdle muscular dystrophy type 2A. Mol. Ther. 2006; 13: [8] Bartoli M, Poupiot J, Vulin A, Fougerousse F, Arandel L, Daniele N, Roudaut C, Noulet F, Garcia L, Danos O, Richard I: AAV-mediated delivery of a mutated myostatin propeptide ameliorates calpain 3 but not alpha-sarcoglycan deficiency. Gene Ther (in press). [9] Brockington M, Yuva Y, Prandini P, Brown SC, Torelli S, Benson MA, Hermann R, Anderson LVB, Bashir R, Burgunder JM, Fallet S, Romero N, Fardeau M, Straub V, Storey G, Pollit C, Richard I, Sewry CA, Bushby K, Voit T, Blake DJ, Muntoni F: Mutations in the fukutin-related protein gene (FKRP) identify limb-girdle muscular dystrophy 2I as a milder allelic variant of congenital muscular dystrophy MDC1C. Hum. Mol. Genet. 2001; 10: [10] Bushby KMD: Making sense of the limb-girdle muscular dystrophies. Brain 1999; 122: [11] Campanaro S, Romualdi C, Fanin M, Celegato B, Pacchioni B, Trevisan S, Laveder P, De Pittà C, Pegoraro E, Hayashi YK, Valle G, Angelini C, Lanfranchi G: Gene expression profiling in dysferlinopathies using a dedicated muscle microarray. Hum. Mol. Genet. 2002; 26: [12] Cenacchi G, Fanin M, De Giorgi L, Angelini C: Ultrastructural changes in dysferlinopathy support detective membrane repair mechanism. J.Clin. Pathol. 2005; 58(2): [13] D Amico A, Tessa A, Bruno C, Petrini S, Biancheri R, Pane M, Pedemonte M, Ricci E, Falace A, Rossi A, Mercuri E, Santorelli FM, Bertini E: Expanding the clinical spectrum of POMT1 phenotype. Neurology 2006; 66: [14] Daniele N, Richard I, Bartoli M: Ins and outs of therapy in limb-girdle muscular dystrophies. Intl J. Biochem. Cell Biol (in press). [15] Driss A, Amouri R, Ben Hamida C, Souilem S, Gouidier-Khhouja N, Hentati L: A new locus for autosomal recessive limb-girdle muscular dystrophy in a large consanguineous Tunisian family maps to chromosome 19q13.3. Neuromusc. Disord 2000; 10: [16] Dubrovsky AL, Angelini C, Bonifati DM, Pegoraro E, Mesa L: Steroids in muscular dystrophy: where do we stand? Neuromusc. Disord. 1998; 8: [17] Esapa CT, Benson MA, Schroder JE, Martin- Rendon E, Brockington M, Brown SC, Muntoni F, Kroger S, Blake DJ: Functional requirements for fukutin-related protein in the Golgi apparatus. Hum. Mol. Genet. 2002; 11: [18] Esapa CT, McIhinney RA, Blake DJ: Fukutinrelated protein mutations that causes congenital muscular dystrophy result in ER-retention of the mutant protein in cultured cells. Hum. Mol. Genet. 2005; 14: [19] Fanin M., Melacini P., Boito C., Pegoraro E., Angelini C.: LGMD2E patients risk developing dilated cardiomyopathy. Neuromusc. Disord. 2003; 13: [20] Fanin M, Fulizio L, Nascimbeni AC, Spinazzi M, Piluso G, Ventriglia VM, Ruzza G, Siciliano G, Trevisan CP, Politano L, Nigro V, Angelini C. Molecular diagnosis in LGMD2A: mutation analysis or protein testing? Hum. Mut. 2004; 24: [21] Fanin M, Nascimbeni AC, Fulizio L, Angelici C: The frequency of limb girdle muscular dystrophy 2A in northeastern Italy. Neuromusc. Disord. 2005; 15: [22] Fougerousse F, Bartoli M, Poupiot J, Arandel L, Durand M, Guerchet N, Gicquel E, Danos O, Richard I: Phenotypic correlation of alphasarcoglycan deficiency by intra-arterial injection of a muscle-specific serotype 1 raav vector. Mol. Ther. 2007; 15: [23] Frosk P, Weiler T, Nylen E, Sudha T, Greenberg CR, Morgan K, Fujiwara TM, Wrogemann K: Limb-girdle muscular dystrophy type 2H associated mutation in TRIM 32, a putative E- ubiquitim ligase gene. Am. J. Hum. Genet. 2002; 70: [24] Godfrey C, Escolar D, Brockington M, Clement EM, Mein R. Jimenez-Mallebrera C, Torelli S, Feng L, Brown SC, Sewry CA, Rutherford M, Shapira Y, Abbs S, Muntoni F: Fukutin gene mutations in steroid-responsive limb girdle muscular dystrophy. Ann. Neurol. 2006; 60: [25] Guttinger M, Tafi E, Battaglia M, Coletta M, Cossu G: Allogeneic mesoangioblasts give rise to alpha-sarcolgycan expressing fibers when transplantated into dystrophic mice. Exp. Cell Res. 2006; 312:
6 [26] Hackman P, Viola A, Haravuori H, Marchand S, Sarparanta J, de Seze J. Labeit S, Witt C, Peltonen L, Richard I, Udd B: Tibial Muscular Dystrophy is a titinopathy caused by mutations in TTN, the gene encoding the giant skeletalmuscle protein titin. Am. J. Hum. Genet. 2002; 71: [27] Haravuori H, Vihola A, Straub V, Auranen M, Richard I, Marchand S, Voit T, Labeit S, Somer H, Peltonen L, Beckmann JS, Udd B: Secondary Calpain3 deficiency in 2q-linked muscular dystrophy: titin is the candidate gene. Neurology 2001; 56: [28] Huang J, Fosberg NE : Role of calpain in skeletal-muscle protein degradation. Proc. Natl. Acad. Sci. 1998; 95: [29] Iwata Y, Katanosaka Y, Shijun Z, Kobayashi Y, Hanada H, Shigekawa M, Wakabayashi S: Protective effects of Ca2+ handling drugs against abnormal Ca2+ homeostasis and cell damage in myopathic skeletal muscle cells. Biochem. Pharmacol. 2005; 70: [30] Keira Y, Noguchi S, Minami N, Hayashi YK, Nishino I. Localization of calpain 3 in human skeletal muscle and its alterations in limb-girdle muscular dystrophy 2A muscle. J. Biochem. 2003; 133: [31] Kramerova I, Kudryashova E, Venkatraman G, Spencer MJ: Calpain 3 partecipates in sarcomere remodeling by acting upstream of the ubiquitinproteasome pathway. Hum. Mol. Genet. 2005; 14: [32] Kudryashova E, Kudryashov D, Kramerova I, Spencer MJ: Trim32 is a ubiquitin ligase mutated in limb-girdle muscular dystrophy type 2H that binds to skeletal muscle myosin and ubiquitinates actin. J. Mol. Biol. 2005; 354: [33] Laval SH, Bushby KMD: Limb- girdle muscular dystrophies- from genetics to molecular pathology. Neuropathol. Appl. Neurobiol. 2004; 30: [34] Liu J, Aoki M, Illa I, Wu CY, Fardeau M, Angelini C, Serrano C, Urtizberea JA, Hentati F, BenHamida M, Bohlega S, Culper EJ, Amato AA, Bossie K, Oeltjen T, Bejaoui K, McKennaYasek D, Hosler BA, Schurr E, Arahata K, dejong PJ, Brown RH: Dysferlin, a novel skeletal muscle gene, is mutated in Miyoshi myopathy and limb girdle muscular dystrophy. Nature Genet. 1998; 20: [35] Mahjneh I, Passos-Bueno MR, Zatz M, Vainzof M, Marconi G, Nashef L, Bashir R, Bushby K: The phenotype of chromosome 2p-linked limbgirdle muscular dystrophy. Neuromusc. Disord. 1996; 6: [36] Mayans O, van der Ven PF, Wilm M, Mues A, Young P, Furst DO, Wilmanns M, Gautel M: Structural basis for activation of the titin kinase domain during myofibrillogenesis. Nature 1998; 395: [37] Minami N, Nishino I, Kobayashi O, Ikezoe K, Goto Y, Nonaka I: Mutations of calpain 3 gene in patients with sporadic limb-girdle muscular dystrophy in Japan. J. Neurol. Sci. 1999; 171: [38] Moreira ES, Vainzof M, Marie SK, Sertiè AL, Zatz M, Passos-Bueno MR: The seventh form of autosomal recessive limb-girdle muscular dystrophy is mapped to 17q Am. J. Hum. Genet. 1997; 61: [39] Nicholas G, Thomas M, Langley B, Somers W, Patel K, Kemp CF, Sharma M, Kambadur R: Titin-cap associates with, and regulates secretion of, Myostatin. J. Cell. Physiol. 2002; 193: [40] Parsons SA, Millay DP, Sargent MA, McNally EM, Molkentin JD: Age-dependent effect of myostatin blockade on disease severity in a murine model of limb-girdle muscular dystrophy. Am. J. Pathol 2006; 168: [41] Piluso G, Politano L, Aurino S. Fanin M, Ricci E, Ventriglia VM, Belsito A, totaro A, Saccone V, Topaloglu H, Nascimbeni AC, Fulizio L, Broccolini A, Canki-Klain N, Ines Comi L, Nigro G, Angelini C, Nigro V: Extensive scanning of the calpain-3 gene broadens the spectrum of LGMD2A phenotypes. J. Med. Genet. 2005; 42: [42] Poussard S, Duvert M, Balcerzak D, Ramassay S, Brustis JJ, Cottin P, Ducastaing A : Evidence for implication of muscle-specific calpain (p94) in myofibrillar integrity. Cell Growth Differ. 1996; 7: [43] Richard I, Broux O, Allamand V, Fougerousse F, ChianniKulchai N, Bourg N, Brenquier L, Devaud C, Pasturaud P, Roudat C: Mutations in the proteolytic calpain 3 cause limb-girdle muscular dystrophy type 2A. Cell 1995; 81: [44] Richard I, Brenguier L, Dincer P, Roudaut C, Bady B, Burgunder JM, Chemaly R, Garcia CA, Halaby G, Jackson CE, Kurnit DM, Lefranc G, Legum C, Loiselet J, Merlini L, Nivelon- Chevallier A, Ollagnon-Roman E, restagno G, Topaloglu H, Beckmann JS: Multiple independent molecular etiology for limb-girdle muscular dystrophy type 2A patients from
7 various geographical origins. Am. J. Hum. Genet. 1997; 60: [45] Saenz A, Leturcq F, Cobo AM, Poza JJ, Ferrer X, Otaegui D, Camano P, Urtasun M, Vilchez J, Gutierrez-Rivas E, Emparanza J, Merlini L, Paisan C, Goicoechea M, Blazquez L, Eymard B, Lochmuller H, Walter M, Bonnemann C, Figarella-Branger D, Kaplan JC, Urtizberea JA, Mart-Masso JF, Lopez de Munain A: LGMD2A: genotype-phenotype correlations based on a large mutational survey on the calpain-3 gene. Brain 2005; 128: [46] Sorimachi H, Imajoh-Ohmi S, Emori Y, et al. Molecular cloning of a novel mammalian calcium-dependent protease distinct from both m- and μ-types: specific expression of the mrna in skeletal muscle. J. Biol. Chem. 1989; 264: [47] Sorimachi H, Kinbara K, Kimura S, Takahashi M, Ishiura S, Sasagawa N, Sorimachi N, Shimada H, Tagawa K, Maruyama K, Suzuki K: Muscle-specific calpain, p94, responsible for limb girdle muscular dystrophy type 2A, associates with connectin through IS2, a p94- specific sequence. J. Biol. Chem. 1995; 270: [48] Spencer MJ, Guyon JR, Sorimachi H, Potts A, Richard I, Herasse M, Chamberlain J, Dalkilic I, Kunkel LM, Beckmann JS : Stable expression of calpain-3 from a muscle transgene in vivo: immature muscle in transgenic mice suggests a role for calpain-3 in muscle maturation. Proc. Natl. Acad. Sci. USA 2002; 99: [49] Straub V, Ettinger AJ, Durbeej M, Venzke DP, Cutshall S, Sanes JR, Campbell KP: Epsilonsarcoglycan replaces alpha-sarcoglycan in smooth muscle to form a unique dystrophinglycoprotein complex. J. Biol. Chem. 1999; 274: [50] Topaloglu H, Dincer P, Richard I,Akcoren Z, Alehan D, Ozme S, Caglar M, Karaduman A, Urtizberea JA, Beckmann JS: Calpain-3 deficiency causes a mild muscular dystrophy in childhood. Neuropediatrics 1997; 28: [51] Walton JN, Nattrass FJ: On the classification, natural history and treatment of the myopathies. Brain 1954; 77: [52] Wenzel K, Zabojszcza J, Carl M, Taubert S, Lass A, Harris CL, Ho M, Schulz H, Hummel O, Hubner N, Osterziel KJ, Spuler S: Increased susceptibility to complement attack due to downregulation of decay-accelerating factor/cd55 in dysferlin-deficient nuscular dystrophy. J. Immunol. 2005; 175: [53] Zaiss AK, Muruve DA: Immune response to adeno-associated virus vectors. Curr. Gene Ther. 2005; 5:
A rare case of muscular dystrophy with POMT2 and FKRP gene mutation. Present by : Ghasem Khazaei Supervisor :Dr Mina Mohammadi Sarband
 A rare case of muscular dystrophy with POMT2 and FKRP gene mutation Present by : Ghasem Khazaei Supervisor :Dr Mina Mohammadi Sarband Index : Congenital muscular dystrophy (CMD) Dystroglycanopathies Walker-Warburg
A rare case of muscular dystrophy with POMT2 and FKRP gene mutation Present by : Ghasem Khazaei Supervisor :Dr Mina Mohammadi Sarband Index : Congenital muscular dystrophy (CMD) Dystroglycanopathies Walker-Warburg
Corporate Medical Policy
 Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: mutation_testing_for_limb_girdle_muscular_dystrophies 01/01/2019 N/A 01/01/2020 01/01/2019 Description of
Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: mutation_testing_for_limb_girdle_muscular_dystrophies 01/01/2019 N/A 01/01/2020 01/01/2019 Description of
Limb Girdle Muscular Dystrophy
 Limb Girdle Muscular Dystrophy Reza Shervin Badv MD, Pediatric Neurologist Children s Medical Center Pediatrics Center of Excellence Tehran University of Medical Sciences Limb-girdle muscular dystrophies(lgmd)
Limb Girdle Muscular Dystrophy Reza Shervin Badv MD, Pediatric Neurologist Children s Medical Center Pediatrics Center of Excellence Tehran University of Medical Sciences Limb-girdle muscular dystrophies(lgmd)
Dysferlinopathies. LGMD2B, Miyoshi & Others. 2B Empowered Conference
 Dysferlinopathies LGMD2B, Miyoshi & Others 2B Empowered Conference Matthew P. Wicklund, MD, FAAN Professor of Neurology and Pediatrics Penn State Health May 24, 2015 Outline A. Empower you with knowledge
Dysferlinopathies LGMD2B, Miyoshi & Others 2B Empowered Conference Matthew P. Wicklund, MD, FAAN Professor of Neurology and Pediatrics Penn State Health May 24, 2015 Outline A. Empower you with knowledge
Hutterite brothers both affected with two forms of limb girdle muscular dystrophy: LGMD2H and LGMD2I
 (2005) 13, 978 982 & 2005 Nature Publishing Group All rights reserved 1018-4813/05 $30.00 ARTICLE www.nature.com/ejhg Hutterite brothers both with two forms of limb girdle muscular dystrophy: LGMD2H and
(2005) 13, 978 982 & 2005 Nature Publishing Group All rights reserved 1018-4813/05 $30.00 ARTICLE www.nature.com/ejhg Hutterite brothers both with two forms of limb girdle muscular dystrophy: LGMD2H and
Immunohistochemical Study of Dystrophin Associated Glycoproteins in Limb-girdle Muscular Dystrophies
 Dystrophin Immunohistochemical Study of Dystrophin Associated Glycoproteins in Limb-girdle Muscular Dystrophies NSC 89-2314-B-002-111 88 8 1 89 7 31 ( Peroxidase -AntiPeroxidase Immnofluorescence) Abstract
Dystrophin Immunohistochemical Study of Dystrophin Associated Glycoproteins in Limb-girdle Muscular Dystrophies NSC 89-2314-B-002-111 88 8 1 89 7 31 ( Peroxidase -AntiPeroxidase Immnofluorescence) Abstract
Asymptomatic carriers for homozygous novel mutations in the FKRP gene: the other end of the spectrum
 (2003) 11, 923 930 & 2003 Nature Publishing Group All rights reserved 1018-4813/03 $25.00 www.nature.com/ejhg ARTICLE Asymptomatic carriers for homozygous novel mutations in the FKRP gene: the other end
(2003) 11, 923 930 & 2003 Nature Publishing Group All rights reserved 1018-4813/03 $25.00 www.nature.com/ejhg ARTICLE Asymptomatic carriers for homozygous novel mutations in the FKRP gene: the other end
Myopathies of Unknown Etiology (Western Blot)
 Myopathies of Unknown Etiology (Western Blot) April 2013 DISCLAIMER: This document was originally drafted in French by the Institut national d'excellence en santé et en services sociaux (INESSS), and that
Myopathies of Unknown Etiology (Western Blot) April 2013 DISCLAIMER: This document was originally drafted in French by the Institut national d'excellence en santé et en services sociaux (INESSS), and that
Diagnosis of limb-girdle muscular dystrophy 2A by immunohistochemical techniques
 Neuropathology 2008; 28, 264 268 doi:10.1111/j.1440-1789.2007.00871.x Original Article Diagnosis of limb-girdle muscular dystrophy 2A by immunohistochemical techniques Hanna K. Kolski, 1 Cynthia Hawkins,
Neuropathology 2008; 28, 264 268 doi:10.1111/j.1440-1789.2007.00871.x Original Article Diagnosis of limb-girdle muscular dystrophy 2A by immunohistochemical techniques Hanna K. Kolski, 1 Cynthia Hawkins,
Muscle fatigue, nnos and muscle fiber atrophy in limb girdle muscular dystrophy
 Acta Myologica 2014; XXXIII: p. 119-126 invited review Muscle fatigue, nnos and muscle fiber atrophy in limb girdle muscular dystrophy Corrado Angelini 1, Elisabetta Tasca 1, Anna Chiara Nascimbeni 2 and
Acta Myologica 2014; XXXIII: p. 119-126 invited review Muscle fatigue, nnos and muscle fiber atrophy in limb girdle muscular dystrophy Corrado Angelini 1, Elisabetta Tasca 1, Anna Chiara Nascimbeni 2 and
READ ORPHA.NET WEBSITE ABOUT BETA-SARCOGLYOCANOPATHY LIMB-GIRDLE MUSCULAR DYSTROPHIES
 READ ORPHA.NET WEBSITE ABOUT BETA-SARCOGLYOCANOPATHY LIMB-GIRDLE MUSCULAR DYSTROPHIES (LGMD) Limb-girdle muscular dystrophies (LGMD) are a heterogeneous group of genetically determined disorders with a
READ ORPHA.NET WEBSITE ABOUT BETA-SARCOGLYOCANOPATHY LIMB-GIRDLE MUSCULAR DYSTROPHIES (LGMD) Limb-girdle muscular dystrophies (LGMD) are a heterogeneous group of genetically determined disorders with a
Clinical and Pathological Characteristics of Four Korean Patients with Limb-Girdle Muscular Dystrophy type 2B
 J Korean Med Sci 2004; 19: 447-52 ISSN 1011-8934 Copyright The Korean Academy of Medical Sciences Clinical and Pathological Characteristics of Four Korean Patients with Limb-Girdle Muscular Dystrophy type
J Korean Med Sci 2004; 19: 447-52 ISSN 1011-8934 Copyright The Korean Academy of Medical Sciences Clinical and Pathological Characteristics of Four Korean Patients with Limb-Girdle Muscular Dystrophy type
Mutations of CAPN3 in Korean Patients with Limb-Girdle Muscular Dystrophy
 J Korean Med Sci 2007; 22: 463-9 ISSN 1011-8934 Copyright The Korean Academy of Medical Sciences Mutations of CAPN3 in Korean Patients with Limb-Girdle Muscular Dystrophy The limb-girdle muscular dystrophy
J Korean Med Sci 2007; 22: 463-9 ISSN 1011-8934 Copyright The Korean Academy of Medical Sciences Mutations of CAPN3 in Korean Patients with Limb-Girdle Muscular Dystrophy The limb-girdle muscular dystrophy
Candidate-gene testing for orphan limb-girdle muscular dystrophies
 Acta Myologica 2008; XXVII: p. 90-97 Candidate-gene testing for orphan limb-girdle muscular dystrophies S. Aurino 1, 2, G. Piluso 1, V. Saccone 2, M. Cacciottolo 2, F. D Amico 1, M. Dionisi 2, A. Totaro
Acta Myologica 2008; XXVII: p. 90-97 Candidate-gene testing for orphan limb-girdle muscular dystrophies S. Aurino 1, 2, G. Piluso 1, V. Saccone 2, M. Cacciottolo 2, F. D Amico 1, M. Dionisi 2, A. Totaro
The limb girdle muscular dystrophies (LGMDs)
 The limb gird muscular dystrophies (LGMDs) This factsheet is for all peop for whom a diagnosis of limb gird muscular dystrophy (LGMD) has been suggested. This is a complicated subject since there are many
The limb gird muscular dystrophies (LGMDs) This factsheet is for all peop for whom a diagnosis of limb gird muscular dystrophy (LGMD) has been suggested. This is a complicated subject since there are many
RECESSIVELY INHERITED LIMBgirdle
 ORIGINAL CONTRIBUTION Cardiac Involvement in Patients With Limb-Girdle Muscular Dystrophy Type 2 and Becker Muscular Dystrophy Marie-Louise Sveen, MD; Jens Jakob Thune, MD; Lars Køber, MD, DMSci; John
ORIGINAL CONTRIBUTION Cardiac Involvement in Patients With Limb-Girdle Muscular Dystrophy Type 2 and Becker Muscular Dystrophy Marie-Louise Sveen, MD; Jens Jakob Thune, MD; Lars Køber, MD, DMSci; John
MP Genetic Testing for Limb-Girdle Muscular Dystrophies
 BCBSA Ref. Policy: 2.04.132 Last Review: 04/30/2018 Effective Date: 04/30/2018 Section: Medicine Related Policies 2.04.86 Genetic Testing for Duchenne and Becker Muscular Dystrophy 2.04.105 Genetic Testing
BCBSA Ref. Policy: 2.04.132 Last Review: 04/30/2018 Effective Date: 04/30/2018 Section: Medicine Related Policies 2.04.86 Genetic Testing for Duchenne and Becker Muscular Dystrophy 2.04.105 Genetic Testing
Genetic Testing for Limb-Girdle Muscular Dystrophies
 Applies to all products administered or underwritten by Blue Cross and Blue Shield of Louisiana and its subsidiary, HMO Louisiana, Inc.(collectively referred to as the Company ), unless otherwise provided
Applies to all products administered or underwritten by Blue Cross and Blue Shield of Louisiana and its subsidiary, HMO Louisiana, Inc.(collectively referred to as the Company ), unless otherwise provided
Genetic diagnosis of limb girdle muscular dystrophy type 2A, A Case Report
 Genetic diagnosis of limb girdle muscular dystrophy type 2A, A Case Report Roshanak Jazayeri, MD, PhD Assistant Professor of Medical Genetics Faculty of Medicine, Alborz University of Medical Sciences
Genetic diagnosis of limb girdle muscular dystrophy type 2A, A Case Report Roshanak Jazayeri, MD, PhD Assistant Professor of Medical Genetics Faculty of Medicine, Alborz University of Medical Sciences
Advances and Perspectives in Muscular Dystrophies Abstract Key words X-Linked Muscular Dystrophies
 Advances and Perspectives in Muscular Dystrophies Corrado Angelini Department of Neurological and Psychiatric Sciences, University of Padova, Padova, Italy Abstract The muscular dystrophies are inherited
Advances and Perspectives in Muscular Dystrophies Corrado Angelini Department of Neurological and Psychiatric Sciences, University of Padova, Padova, Italy Abstract The muscular dystrophies are inherited
Muscular Dystrophy. Biol 405 Molecular Medicine
 Muscular Dystrophy Biol 405 Molecular Medicine Duchenne muscular dystrophy Duchenne muscular dystrophy is a neuromuscular disease that occurs in ~ 1/3,500 male births. The disease causes developmental
Muscular Dystrophy Biol 405 Molecular Medicine Duchenne muscular dystrophy Duchenne muscular dystrophy is a neuromuscular disease that occurs in ~ 1/3,500 male births. The disease causes developmental
Disease taxonomy - monogenic muscular dystrophy
 Disease taxonomy - monogenic muscular dystrophy Jacques S Beckmann URA CNRS 1922 - Genethon, Evry, France The field of the autosomal recessive progressive muscular dystrophies has clarified significantly
Disease taxonomy - monogenic muscular dystrophy Jacques S Beckmann URA CNRS 1922 - Genethon, Evry, France The field of the autosomal recessive progressive muscular dystrophies has clarified significantly
Sarcoglycanopathies: A Multiplex Molecular Analysis for the Most Common Mutations
 ORIGINAL ARTICLE Sarcoglycanopathies: A Multiplex Molecular Analysis for the Most Common Mutations Telma L.F. Gouveia, MS,* Julia F.O. Paim, MD, PhD,w Rita C. Pavanello, MD,* Mayana Zatz, PhD,* and Mariz
ORIGINAL ARTICLE Sarcoglycanopathies: A Multiplex Molecular Analysis for the Most Common Mutations Telma L.F. Gouveia, MS,* Julia F.O. Paim, MD, PhD,w Rita C. Pavanello, MD,* Mayana Zatz, PhD,* and Mariz
T he sarcoglycanopathies are a group of autosomal
 1of7 ONLINE MUTATION REPORT Novel sarcoglycan gene mutations in a large cohort of Italian patients C Boito, M Fanin, G Siciliano, C Angelini, E Pegoraro... T he sarcoglycanopathies are a group of autosomal
1of7 ONLINE MUTATION REPORT Novel sarcoglycan gene mutations in a large cohort of Italian patients C Boito, M Fanin, G Siciliano, C Angelini, E Pegoraro... T he sarcoglycanopathies are a group of autosomal
The extensive scanning of the calpain-3 gene broadens the spectrum of LGMD2A phenotypes
 Piluso et al., 1 The extensive scanning of the calpain-3 gene broadens the spectrum of LGMD2A phenotypes Authors: Giulio Piluso, MS 1,2,X, Luisa Politano, MD 2,3,X, Stefania Aurino, MS 1,4, Marina Fanin,
Piluso et al., 1 The extensive scanning of the calpain-3 gene broadens the spectrum of LGMD2A phenotypes Authors: Giulio Piluso, MS 1,2,X, Luisa Politano, MD 2,3,X, Stefania Aurino, MS 1,4, Marina Fanin,
Clinical, Molecular, and Protein Correlations in a Large Sample of Genetically Diagnosed Italian Limb Girdle Muscular Dystrophy Patients
 HUMAN MUTATION 29(2), 258^266, 2008 RESEARCH ARTICLE Clinical, Molecular, and Protein Correlations in a Large Sample of Genetically Diagnosed Italian Limb Girdle Muscular Dystrophy Patients Michela Guglieri,
HUMAN MUTATION 29(2), 258^266, 2008 RESEARCH ARTICLE Clinical, Molecular, and Protein Correlations in a Large Sample of Genetically Diagnosed Italian Limb Girdle Muscular Dystrophy Patients Michela Guglieri,
L imb-girdle muscular dystrophies (LGMD) and congenital
 1of7 ONLINE MUTATION REPORT FKRP (826C.A) frequently causes limb-girdle muscular dystrophy in German patients M C Walter, J A Petersen, R Stucka, D Fischer, R Schröder, M Vorgerd, A Schroers, H Schreiber,
1of7 ONLINE MUTATION REPORT FKRP (826C.A) frequently causes limb-girdle muscular dystrophy in German patients M C Walter, J A Petersen, R Stucka, D Fischer, R Schröder, M Vorgerd, A Schroers, H Schreiber,
A novel POMT2 mutation causes mild congenital muscular dystrophy with normal brain MRI
 Brain & Development 31 (2009) 465 468 Case report A novel POMT2 mutation causes mild congenital muscular dystrophy with normal brain MRI Terumi Murakami a,b, Yukiko K. Hayashi a, *, Megumu Ogawa a, Satoru
Brain & Development 31 (2009) 465 468 Case report A novel POMT2 mutation causes mild congenital muscular dystrophy with normal brain MRI Terumi Murakami a,b, Yukiko K. Hayashi a, *, Megumu Ogawa a, Satoru
18 (2), DOI: /bjmg
 18 (2), 2015 71-76 DOI: 10.1515/bjmg-2015-0088 CASE REPORT SARCOLEMMAL DEFICIENCY OF SARCOGLYCAN COMPLEX IN AN 18-MONTH-OLD TURKISH BOY WITH A LARGE DELETION IN THE BETA SARCOGLYCAN GENE Diniz G 1,*, Tekgul
18 (2), 2015 71-76 DOI: 10.1515/bjmg-2015-0088 CASE REPORT SARCOLEMMAL DEFICIENCY OF SARCOGLYCAN COMPLEX IN AN 18-MONTH-OLD TURKISH BOY WITH A LARGE DELETION IN THE BETA SARCOGLYCAN GENE Diniz G 1,*, Tekgul
CONTENT ANATOMIC LOCI OF NM DISEASE ASSOCIATED FEATURES FUNCTIONAL DIFFICULTIES. CLINICAL HISTORY IN NEUROMUSCULAR DISEASES Weakness 06/11/60
 CONTENT HOW TO APPROACH LIMB GIRDLE AND NON-LIMB GIRDLE WEAKNESS Kongkiat Kulkantrakorn, M.D. Professor Thammasat University Clinical approach in NM disease and phenotype Common and uncommon LGMDs Common
CONTENT HOW TO APPROACH LIMB GIRDLE AND NON-LIMB GIRDLE WEAKNESS Kongkiat Kulkantrakorn, M.D. Professor Thammasat University Clinical approach in NM disease and phenotype Common and uncommon LGMDs Common
T he limb-girdle muscular dystrophies (LGMDs) are genetically
 1of8 ELECTRONIC LETTER Genotype-phenotype correlations in 35 Brazilian families with sarcoglycanopathies including the description of three novel mutations E S Moreira, M Vainzof, O T Suzuki, RCMPavanello,
1of8 ELECTRONIC LETTER Genotype-phenotype correlations in 35 Brazilian families with sarcoglycanopathies including the description of three novel mutations E S Moreira, M Vainzof, O T Suzuki, RCMPavanello,
FEP Medical Policy Manual
 FEP Medical Policy Manual FEP 2.04.132 Genetic Testing for Limb-Girdle Muscular Dystrophies Effective Date: July 15, 2018 Related Policies: 2.04.86 Genetic Testing for Duchenne and Becker Muscular Dystrophy
FEP Medical Policy Manual FEP 2.04.132 Genetic Testing for Limb-Girdle Muscular Dystrophies Effective Date: July 15, 2018 Related Policies: 2.04.86 Genetic Testing for Duchenne and Becker Muscular Dystrophy
근이양증의분자적병리학적진단 Molecular and Pathological Diagnosis of Muscular Dystrophies
 HANYANG MEDICAL REVIEWS Vol. 26, No. 1, 2006 근이양증의분자적병리학적진단 Molecular and Pathological Diagnosis of Muscular Dystrophies 최영철연세대학교의과대학신경과교실 Young-Chul Choi, M.D., Ph.D. Department of Neurology, Yonsei University
HANYANG MEDICAL REVIEWS Vol. 26, No. 1, 2006 근이양증의분자적병리학적진단 Molecular and Pathological Diagnosis of Muscular Dystrophies 최영철연세대학교의과대학신경과교실 Young-Chul Choi, M.D., Ph.D. Department of Neurology, Yonsei University
Disruption of heart sarcoglycan complex and severe cardiomyopathy caused by β sarcoglycan mutations
 102 Department of Neuromuscular Diseases, Istituto Nazionale Neurologico C Besta, Via Celoria 11, 20133 Milano, Italy R Barresi C Di Blasi T Negri R Brugnoni S Daniel F Cornelio L Morandi M Mora Department
102 Department of Neuromuscular Diseases, Istituto Nazionale Neurologico C Besta, Via Celoria 11, 20133 Milano, Italy R Barresi C Di Blasi T Negri R Brugnoni S Daniel F Cornelio L Morandi M Mora Department
We are IntechOpen, the world s leading publisher of Open Access books Built by scientists, for scientists. International authors and editors
 We are IntechOpen, the world s leading publisher of Open Access books Built by scientists, for scientists 4,000 116,000 120M Open access books available International authors and editors Downloads Our
We are IntechOpen, the world s leading publisher of Open Access books Built by scientists, for scientists 4,000 116,000 120M Open access books available International authors and editors Downloads Our
The New England Journal of Medicine MUTATIONS IN THE SARCOGLYCAN GENES IN PATIENTS WITH MYOPATHY
 MUTATIONS IN THE SARCOGLYCAN GENES IN PATIENTS WITH MYOPATHY DAVID J. DUGGAN, B.S., J. RAFAEL GOROSPE, M.D., PH.D., MARINA FANIN, M.S., ERIC P. HOFFMAN, PH.D., A CORRADO ANGELINI, M.D. ABSTRACT Background
MUTATIONS IN THE SARCOGLYCAN GENES IN PATIENTS WITH MYOPATHY DAVID J. DUGGAN, B.S., J. RAFAEL GOROSPE, M.D., PH.D., MARINA FANIN, M.S., ERIC P. HOFFMAN, PH.D., A CORRADO ANGELINI, M.D. ABSTRACT Background
Three Muscular Dystrophies: Loss of Cytoskeleton-Extracellular Matrix Linkage
 Cell, Vol. 80, 675-679, March 10, 1995, Copyright 1995 by Cell Press Three Muscular Dystrophies: Loss of Cytoskeleton-Extracellular Matrix Linkage Review Kevin P. Campbell Howard Hughes Medical Institute
Cell, Vol. 80, 675-679, March 10, 1995, Copyright 1995 by Cell Press Three Muscular Dystrophies: Loss of Cytoskeleton-Extracellular Matrix Linkage Review Kevin P. Campbell Howard Hughes Medical Institute
Author Manuscript Faculty of Biology and Medicine Publication
 Serveur Académique Lausannois SERVAL serval.unil.ch Author Manuscript Faculty of Biology and Medicine Publication This paper has been peer-reviewed but dos not include the final publisher proof-corrections
Serveur Académique Lausannois SERVAL serval.unil.ch Author Manuscript Faculty of Biology and Medicine Publication This paper has been peer-reviewed but dos not include the final publisher proof-corrections
Dysferlinopathy: A clinical and histopathological study of 28 patients from India
 Original Article Dysferlinopathy: A clinical and histopathological study of 28 patients from India A. Nalini, N. Gayathri 1 Departments of Neurology and 1 Neuropathology, National Institute of Mental Health
Original Article Dysferlinopathy: A clinical and histopathological study of 28 patients from India A. Nalini, N. Gayathri 1 Departments of Neurology and 1 Neuropathology, National Institute of Mental Health
Limb-girdle muscular dystrophies: Where next after six decades from the first proposal (Review)
 MOLECULAR MEDICINE REPORTS 9: 1515-1532, 2014 Limb-girdle muscular dystrophies: Where next after six decades from the first proposal (Review) OMAR A. MAHMOOD 1,2 and XIN MEI JIANG 1 1 Department of Neurology,
MOLECULAR MEDICINE REPORTS 9: 1515-1532, 2014 Limb-girdle muscular dystrophies: Where next after six decades from the first proposal (Review) OMAR A. MAHMOOD 1,2 and XIN MEI JIANG 1 1 Department of Neurology,
Original Article CD4 + cells, macrophages, MHC-I and C5b-9 involve the pathogenesis of dysferlinopathy
 Int J Clin Exp Pathol 2015;8(3):3069-3075 www.ijcep.com /ISSN:1936-2625/IJCEP0004885 Original Article CD4 + cells, macrophages, MHC-I and C5b-9 involve the pathogenesis of dysferlinopathy Xi Yin 1*, Qian
Int J Clin Exp Pathol 2015;8(3):3069-3075 www.ijcep.com /ISSN:1936-2625/IJCEP0004885 Original Article CD4 + cells, macrophages, MHC-I and C5b-9 involve the pathogenesis of dysferlinopathy Xi Yin 1*, Qian
Genetic Testing for Muscular Dystrophies
 MEDICAL POLICY 12.04.86 Genetic Testing for Muscular Dystrophies BCBSA Ref. Policies: 2.04.86*, 2.04.105*, 2.04.132* Effective Date: June 1, 2018 RELATED MEDICAL POLICIES: Last Revised: May 3, 2018 None
MEDICAL POLICY 12.04.86 Genetic Testing for Muscular Dystrophies BCBSA Ref. Policies: 2.04.86*, 2.04.105*, 2.04.132* Effective Date: June 1, 2018 RELATED MEDICAL POLICIES: Last Revised: May 3, 2018 None
Novel pharmacological approaches to treat protein-misfolding diseases
 IRCCS E. Medea, Bosisio Parini (LC) 14 November 2014 Distrofia dei cingoli da deficit di Calpaina 3 Dal gene al paziente Novel pharmacological approaches to treat protein-misfolding diseases Dorianna Sandonà
IRCCS E. Medea, Bosisio Parini (LC) 14 November 2014 Distrofia dei cingoli da deficit di Calpaina 3 Dal gene al paziente Novel pharmacological approaches to treat protein-misfolding diseases Dorianna Sandonà
Atypical Miyoshi distal myopathy: A case report
 3068 Atypical Miyoshi distal myopathy: A case report MEILING WANG 1, YUJIE GUO 1, YONG FU 1, RUI JIA 1 and GANG CHEN 2 Departments of 1 Neurology and 2 Interventional Radiology, The Affiliated Hospital
3068 Atypical Miyoshi distal myopathy: A case report MEILING WANG 1, YUJIE GUO 1, YONG FU 1, RUI JIA 1 and GANG CHEN 2 Departments of 1 Neurology and 2 Interventional Radiology, The Affiliated Hospital
Calpain-3 is autolyzed and hence activated in human skeletal muscle 24 h following a single bout of eccentric exercise
 J Appl Physiol 103: 926 931, 2007. First published June 21, 2007; doi:10.1152/japplphysiol.01422.2006. Calpain-3 is autolyzed and hence activated in human skeletal muscle 24 h following a single bout of
J Appl Physiol 103: 926 931, 2007. First published June 21, 2007; doi:10.1152/japplphysiol.01422.2006. Calpain-3 is autolyzed and hence activated in human skeletal muscle 24 h following a single bout of
Muscular Dystrophies. Pinki Munot Consultant Paediatric Neurologist Great Ormond Street Hospital Practical Neurology Study days April 2018
 Muscular Dystrophies Pinki Munot Consultant Paediatric Neurologist Great Ormond Street Hospital Practical Neurology Study days April 2018 Definition and classification Clinical guide to recognize muscular
Muscular Dystrophies Pinki Munot Consultant Paediatric Neurologist Great Ormond Street Hospital Practical Neurology Study days April 2018 Definition and classification Clinical guide to recognize muscular
Amsterdam, The Netherlands, June 24 27, Teaching Course 18. How to diagnose a muscle disorder - Level 1. Muscle imaging
 3 rd Congress of the European Academy of Neurology Amsterdam, The Netherlands, June 24 27, 2017 Teaching Course 18 How to diagnose a muscle disorder - Level 1 Muscle imaging Volker Straub Newcastle upon
3 rd Congress of the European Academy of Neurology Amsterdam, The Netherlands, June 24 27, 2017 Teaching Course 18 How to diagnose a muscle disorder - Level 1 Muscle imaging Volker Straub Newcastle upon
Assignment of the Tibial Muscular Dystrophy Locus to Chromosome 2q31
 Am. J. Hum. Genet. 62:620 626, 1998 Assignment of the Tibial Muscular Dystrophy Locus to Chromosome 2q31 Henna Haravuori, 1 Päivi Mäkelä-Bengs, 1 Bjarne Udd, 3 Juhani Partanen, 4 Leena Pulkkinen, 4 Hannu
Am. J. Hum. Genet. 62:620 626, 1998 Assignment of the Tibial Muscular Dystrophy Locus to Chromosome 2q31 Henna Haravuori, 1 Päivi Mäkelä-Bengs, 1 Bjarne Udd, 3 Juhani Partanen, 4 Leena Pulkkinen, 4 Hannu
The Limb-Girdle Muscular Dystrophies and the Dystrophinopathies Stanley Jones P. Iyadurai, MSc, PhD, MD; John T. Kissel, MD, FAAN
 Review Article Downloaded from https://journals.lww.com/continuum by maxwo3znzwrcfjddvmduzvysskax4mzb8eymgwvspgpjoz9l+mqfwgfuplwvy+jmyqlpqmifewtrhxj7jpeo+505hdqh14pdzv4lwky42mcrzqckilw0d1o4yvrwmuvvhuyo4rrbviuuwr5dqytbtk/icsrdbt0hfryk7+zagvaltkgnudxdohhaxffu/7kno26hifzu/+bcy16w7w1bdw==
Review Article Downloaded from https://journals.lww.com/continuum by maxwo3znzwrcfjddvmduzvysskax4mzb8eymgwvspgpjoz9l+mqfwgfuplwvy+jmyqlpqmifewtrhxj7jpeo+505hdqh14pdzv4lwky42mcrzqckilw0d1o4yvrwmuvvhuyo4rrbviuuwr5dqytbtk/icsrdbt0hfryk7+zagvaltkgnudxdohhaxffu/7kno26hifzu/+bcy16w7w1bdw==
A novel mutation in the DYSF gene in a patient with a presumed inflammatory myopathy
 2018; 38, 433 437 doi:10.1111/neup.12474 Case Report A novel mutation in the DYSF gene in a patient with a presumed inflammatory myopathy Jin Tang, Xueqin Song, Guang Ji, Hongran Wu, Shuyan Sun, Shan Lu,
2018; 38, 433 437 doi:10.1111/neup.12474 Case Report A novel mutation in the DYSF gene in a patient with a presumed inflammatory myopathy Jin Tang, Xueqin Song, Guang Ji, Hongran Wu, Shuyan Sun, Shan Lu,
Dubowitz Neuromuscular Centre, Institute of Child Health and Great Ormond Street Hospital for Children, UCL, London, UK. 2
 Brain Pathology ISSN 1015-6305 RESEARCH ARTICLE A Comparative Study of a-dystroglycan Glycosylation in Dystroglycanopathies Suggests that the Hypoglycosylation of a-dystroglycan Does Not Consistently Correlate
Brain Pathology ISSN 1015-6305 RESEARCH ARTICLE A Comparative Study of a-dystroglycan Glycosylation in Dystroglycanopathies Suggests that the Hypoglycosylation of a-dystroglycan Does Not Consistently Correlate
Muscle Protein Alterations in LGMD2I Patients With Different Mutations in the Fukutin-related Protein Gene
 Volume 56(11): 995 1001, 2008 Journal of Histochemistry & Cytochemistry http://www.jhc.org ARTICLE Muscle Protein Alterations in LGMD2I Patients With Different Mutations in the Fukutin-related Protein
Volume 56(11): 995 1001, 2008 Journal of Histochemistry & Cytochemistry http://www.jhc.org ARTICLE Muscle Protein Alterations in LGMD2I Patients With Different Mutations in the Fukutin-related Protein
The Seventh Form of Autosomal Recessive Limb-Girdle Muscular Dystrophy Is Mapped to 17q11-12
 Am. J. Hum. Genet. 61:151 159, 1997 The Seventh Form of Autosomal Recessive Limb-Girdle Muscular Dystrophy Is Mapped to 17q11-12 Eloisa S. Moreira, Mariz Vainzof, Sueli K. Marie, Andrea L. Sertié, Mayana
Am. J. Hum. Genet. 61:151 159, 1997 The Seventh Form of Autosomal Recessive Limb-Girdle Muscular Dystrophy Is Mapped to 17q11-12 Eloisa S. Moreira, Mariz Vainzof, Sueli K. Marie, Andrea L. Sertié, Mayana
Title: 233Rd ENMC international workshop: clinical trial readiness for calpainopathies, september 15 17, 2017 naarden, The Netherlands
 Accepted Manuscript Title: 233Rd ENMC international workshop: clinical trial readiness for calpainopathies, september 15 17, 2017 naarden, The Netherlands Author: William Lostal, J. Andoni Urtizberea,
Accepted Manuscript Title: 233Rd ENMC international workshop: clinical trial readiness for calpainopathies, september 15 17, 2017 naarden, The Netherlands Author: William Lostal, J. Andoni Urtizberea,
Congenital Muscular Dystrophy: Hospital Based Study in Egyptian Pediatric Patients
 World Journal of Medical Sciences 10 (4): 503-507, 2014 ISSN 1817-3055 IDOSI Publications, 2014 DOI: 10.5829/idosi.wjms.2014.10.4.953 Congenital Muscular Dystrophy: Hospital Based Study in Egyptian Pediatric
World Journal of Medical Sciences 10 (4): 503-507, 2014 ISSN 1817-3055 IDOSI Publications, 2014 DOI: 10.5829/idosi.wjms.2014.10.4.953 Congenital Muscular Dystrophy: Hospital Based Study in Egyptian Pediatric
Muscular Dystrophies in Adulthood Matthew P. Wicklund, MD, FAAN Professor of Neurology University of Colorado School of Medicine
 Disclosure Information Disclosure of Relevant Financial Relationships Muscular Dystrophies in Adulthood Matthew P. Wicklund, MD, FAAN Professor of Neurology University of Colorado School of Medicine I
Disclosure Information Disclosure of Relevant Financial Relationships Muscular Dystrophies in Adulthood Matthew P. Wicklund, MD, FAAN Professor of Neurology University of Colorado School of Medicine I
Muscle Diseases: The Muscular Dystrophies
 Annu. Rev. Pathol. Mech. Dis. 2007. 2:87 109 The Annual Review of Pathology: Mechanisms of Disease is online at pathmechdis.annualreviews.org This article s doi: 10.1146/annurev.pathol.2.010506.091936
Annu. Rev. Pathol. Mech. Dis. 2007. 2:87 109 The Annual Review of Pathology: Mechanisms of Disease is online at pathmechdis.annualreviews.org This article s doi: 10.1146/annurev.pathol.2.010506.091936
MRI and proteomic studies in normal and
 MRI and proteomic studies in normal and pathological muscles. Isabel ILLA. Catedrática Medicina de la Universitat Autònoma de Barcelona. Chief Neuromuscular Diseases Unit. Hospital Santa Creu i Sant Pau.
MRI and proteomic studies in normal and pathological muscles. Isabel ILLA. Catedrática Medicina de la Universitat Autònoma de Barcelona. Chief Neuromuscular Diseases Unit. Hospital Santa Creu i Sant Pau.
The Italian LGMD registry: relative frequency, clinical features, and differential diagnosis
 The Italian LGMD registry: relative frequency, clinical features, and differential diagnosis Authors: Francesca Magri MD 1, Vincenzo Nigro MD 2,3,, Corrado Angelini MD 4, Tiziana Mongini MD 5, Marina Mora
The Italian LGMD registry: relative frequency, clinical features, and differential diagnosis Authors: Francesca Magri MD 1, Vincenzo Nigro MD 2,3,, Corrado Angelini MD 4, Tiziana Mongini MD 5, Marina Mora
ACTA MYOLOGICA. Established in 1982 as Cardiomyology. (Myopathies, Cardiomyopathies and Neuromyopathies) Vol. XXXIII - May 2014
 ISSN 28-2460 Established in 982 as Cardiomyology POSTE ITALIANE SPA - Spedizione in Abbonamento Postale - D.L. 353/2003 conv. in L. 27/02/2004 n 46 art., comma, DCB PISA Vol. XXXIII - May 204 ACTA MYOLOGICA
ISSN 28-2460 Established in 982 as Cardiomyology POSTE ITALIANE SPA - Spedizione in Abbonamento Postale - D.L. 353/2003 conv. in L. 27/02/2004 n 46 art., comma, DCB PISA Vol. XXXIII - May 204 ACTA MYOLOGICA
Iowa Wellstone Center Muscle Tissue and Cell Culture Repository
 Iowa Wellstone Center Muscle Tissue and Cell Culture Repository Steven A. Moore, M.D., Ph.D. The University of Iowa Department of Pathology and Iowa Wellstone Muscular Dystrophy Cooperative Research Center
Iowa Wellstone Center Muscle Tissue and Cell Culture Repository Steven A. Moore, M.D., Ph.D. The University of Iowa Department of Pathology and Iowa Wellstone Muscular Dystrophy Cooperative Research Center
Introduction. Case report. Abstract
 Case report Case report of an adolescent girl with limb-girdle muscular dystrophy type 2B the usefulness of muscle protein immunostaining in the diagnosis of dysferlinopathies Sylwia Szymanska 1, Dariusz
Case report Case report of an adolescent girl with limb-girdle muscular dystrophy type 2B the usefulness of muscle protein immunostaining in the diagnosis of dysferlinopathies Sylwia Szymanska 1, Dariusz
Facts About Limb-Girdle. Muscular Dystrophies
 Facts About Limb-Girdle Muscular Dystrophies Updated December 2009 Mandy Van Benthuysen Dear Friends: When I was 4 years old, my parents took me to a specialist to find out why I walked with an unusual
Facts About Limb-Girdle Muscular Dystrophies Updated December 2009 Mandy Van Benthuysen Dear Friends: When I was 4 years old, my parents took me to a specialist to find out why I walked with an unusual
Gene therapy and genome editing technologies for the study and potential treatment of :
 WORKSHOP ON GENOME EDITING Gene therapy and genome editing technologies for the study and potential treatment of : Duchenne Muscular Dystrophy by Dr France Piétri-Rouxel, Institut de Myologie Centre de
WORKSHOP ON GENOME EDITING Gene therapy and genome editing technologies for the study and potential treatment of : Duchenne Muscular Dystrophy by Dr France Piétri-Rouxel, Institut de Myologie Centre de
Corporate Medical Policy
 Corporate Medical Policy Genetic Testing for Duchenne and Becker Muscular Dystrophy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: genetic_testing_for_duchenne_and_becker_muscular_dystrophy
Corporate Medical Policy Genetic Testing for Duchenne and Becker Muscular Dystrophy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: genetic_testing_for_duchenne_and_becker_muscular_dystrophy
Experimental Physiology
 Exp Physiol 99.4 (2014) pp 621 626 621 Symposium Report Symposium Report Modifiers of heart and muscle function: where genetics meets physiology Kayleigh A. Swaggart 1 and Elizabeth M. McNally 1,2 1 Departments
Exp Physiol 99.4 (2014) pp 621 626 621 Symposium Report Symposium Report Modifiers of heart and muscle function: where genetics meets physiology Kayleigh A. Swaggart 1 and Elizabeth M. McNally 1,2 1 Departments
Molecular mechanisms of muscular dystrophies: old and new players
 MECHANISMS OF DISEASE Molecular mechanisms of muscular dystrophies: old and new players Kay E Davies* and Kristen J Nowak Abstract The study of the muscle cell in the muscular dystrophies (MDs) has shown
MECHANISMS OF DISEASE Molecular mechanisms of muscular dystrophies: old and new players Kay E Davies* and Kristen J Nowak Abstract The study of the muscle cell in the muscular dystrophies (MDs) has shown
LARGE expression in different types of muscular dystrophies other than dystroglycanopathy
 Balci-Hayta et al. BMC Neurology (2018) 18:207 https://doi.org/10.1186/s12883-018-1207-0 RESEARCH ARTICLE LARGE expression in different types of muscular dystrophies other than dystroglycanopathy Burcu
Balci-Hayta et al. BMC Neurology (2018) 18:207 https://doi.org/10.1186/s12883-018-1207-0 RESEARCH ARTICLE LARGE expression in different types of muscular dystrophies other than dystroglycanopathy Burcu
Limb-girdle Muscular Dystrophy with New Mutation in Sarcoglycan Beta Gene: A Case Report
 Iran J Public Health, Vol. 47, No.12, Dec 2018, pp.1953-1957 Case Report Limb-girdle Muscular Dystrophy with New Mutation in Sarcoglycan Beta Gene: A Case Report Eskandar TAGHIZADEH 1,2, Hamed ABDOLKARIMI
Iran J Public Health, Vol. 47, No.12, Dec 2018, pp.1953-1957 Case Report Limb-girdle Muscular Dystrophy with New Mutation in Sarcoglycan Beta Gene: A Case Report Eskandar TAGHIZADEH 1,2, Hamed ABDOLKARIMI
Seminar. Muscular dystrophies
 Muscular dystrophies Eugenio Mercuri, Francesco Muntoni Muscular dystrophies are a heterogeneous group of inherited disorders that share similar clinical features and dystrophic changes on muscle biopsy.
Muscular dystrophies Eugenio Mercuri, Francesco Muntoni Muscular dystrophies are a heterogeneous group of inherited disorders that share similar clinical features and dystrophic changes on muscle biopsy.
Clinical and Pathological Heterogeneity of Korean Patients with CAPN3 Mutations
 Original Article Yonsei Med J 2016 Jan;57(1):173-179 pissn: 0513-5796 eissn: 1976-2437 Clinical and Pathological Heterogeneity of Korean Patients with CAPN3 Mutations Hyung Jun Park 1,2, Hoon Jang 3, Jung
Original Article Yonsei Med J 2016 Jan;57(1):173-179 pissn: 0513-5796 eissn: 1976-2437 Clinical and Pathological Heterogeneity of Korean Patients with CAPN3 Mutations Hyung Jun Park 1,2, Hoon Jang 3, Jung
MUSCULAR DYSTROPHY encompasses
 CASE REPORTS Loss of Podocyte Dysferlin Expression Is Associated With Minimal Change Nephropathy Hassane Izzedine, MD, PhD, Isabelle Brocheriou, MD, PhD, Bruno Eymard, MD, PhD, Monique Le Charpentier,
CASE REPORTS Loss of Podocyte Dysferlin Expression Is Associated With Minimal Change Nephropathy Hassane Izzedine, MD, PhD, Isabelle Brocheriou, MD, PhD, Bruno Eymard, MD, PhD, Monique Le Charpentier,
MUSCLE DISEASE ANTIBODIES NOVOCASTRA ADVANCING MUSCLE DISEASE DIAGNOSIS, MANAGEMENT AND RESEARCH RESULTS YOU CAN RELY ON
 MUSCLE DISEASE ANTIBODIES ADVANCING MUSCLE DISEASE DIAGNOSIS, MANAGEMENT AND RESEARCH NOVOCASTRA RESULTS YOU CAN RELY ON Novocastra Muscle Disease Antibodies The Novocastra muscle disease portfolio comprises
MUSCLE DISEASE ANTIBODIES ADVANCING MUSCLE DISEASE DIAGNOSIS, MANAGEMENT AND RESEARCH NOVOCASTRA RESULTS YOU CAN RELY ON Novocastra Muscle Disease Antibodies The Novocastra muscle disease portfolio comprises
Electron microscopy in the investigation and diagnosis of muscle disease
 Electron microscopy in the investigation and diagnosis of muscle disease Roy Weller Clinical Neurosciences University of Southampton School of Medicine Normal Muscle Normal Muscle The Sarcomere The names
Electron microscopy in the investigation and diagnosis of muscle disease Roy Weller Clinical Neurosciences University of Southampton School of Medicine Normal Muscle Normal Muscle The Sarcomere The names
Anesthesia recommendations for patients suffering from Welander distal myopathy
 orphananesthesia Anesthesia recommendations for patients suffering from Welander distal myopathy Disease name: Welander distal myopathy ICD 10: G71.0 Synonyms: late adult onset type 1 distal myopathy;
orphananesthesia Anesthesia recommendations for patients suffering from Welander distal myopathy Disease name: Welander distal myopathy ICD 10: G71.0 Synonyms: late adult onset type 1 distal myopathy;
Revised spectrum of mutations in sarcoglycanopathies
 (2008) 16, 793 803 & 2008 Nature Publishing Group All rights reserved 1018-4813/08 $30.00 www.nature.com/ejhg ARTICLE Revised spectrum of mutations in sarcoglycanopathies Madiha Trabelsi 1,3,4, Niloufar
(2008) 16, 793 803 & 2008 Nature Publishing Group All rights reserved 1018-4813/08 $30.00 www.nature.com/ejhg ARTICLE Revised spectrum of mutations in sarcoglycanopathies Madiha Trabelsi 1,3,4, Niloufar
Genetic modifiers of muscular dystrophy: Implications for therapy
 Biochimica et Biophysica Acta 1772 (2007) 216 228 www.elsevier.com/locate/bbadis Review Genetic modifiers of muscular dystrophy: Implications for therapy Ahlke Heydemann a, Katherine R. Doherty b, Elizabeth
Biochimica et Biophysica Acta 1772 (2007) 216 228 www.elsevier.com/locate/bbadis Review Genetic modifiers of muscular dystrophy: Implications for therapy Ahlke Heydemann a, Katherine R. Doherty b, Elizabeth
The relative frequency of common neuromuscular diagnoses in a reference center
 https://doi.org/10.1590/0004-282x20170151 ARTICLE The relative frequency of common neuromuscular diagnoses in a reference center Frequência relativa de diagnósticos neuromusculares comuns em um serviço
https://doi.org/10.1590/0004-282x20170151 ARTICLE The relative frequency of common neuromuscular diagnoses in a reference center Frequência relativa de diagnósticos neuromusculares comuns em um serviço
Current Research Strategies and Therapeutic Approaches in Duchenne Muscular Dystrophy
 Current Research Strategies and Therapeutic Approaches in Duchenne Muscular Dystrophy H. Lee Sweeney, Ph.D. Department of Physiology University of Pennsylvania Perelman School of Medicine Current Research
Current Research Strategies and Therapeutic Approaches in Duchenne Muscular Dystrophy H. Lee Sweeney, Ph.D. Department of Physiology University of Pennsylvania Perelman School of Medicine Current Research
CONTRIBUTION OF DYSFERLIN-CONTAINING MEMBRANES TO MEMBRANE REPAIR IN SKELETAL MUSCLE. Joel Ryan McDade
 CONTRIBUTION OF DYSFERLIN-CONTAINING MEMBRANES TO MEMBRANE REPAIR IN SKELETAL MUSCLE by Joel Ryan McDade A dissertation submitted in partial fulfillment of the requirements for the degree of Doctor of
CONTRIBUTION OF DYSFERLIN-CONTAINING MEMBRANES TO MEMBRANE REPAIR IN SKELETAL MUSCLE by Joel Ryan McDade A dissertation submitted in partial fulfillment of the requirements for the degree of Doctor of
ORIGINAL CONTRIBUTION
 ORIGINAL CONTRIBUTION Phenotypic Study in 40 Patients With Dysferlin Gene Mutations High Frequency of Atypical Phenotypes Karine Nguyen, MD; Guillaume Bassez, MD; Martin Krahn, MD; Rafaelle Bernard, MD;
ORIGINAL CONTRIBUTION Phenotypic Study in 40 Patients With Dysferlin Gene Mutations High Frequency of Atypical Phenotypes Karine Nguyen, MD; Guillaume Bassez, MD; Martin Krahn, MD; Rafaelle Bernard, MD;
Whole Exome Sequencing Reveals DYSF, FKTN, and ISPD Mutations in Congenital Muscular Dystrophy Without Brain or Eye Involvement
 Whole Exome Sequencing Reveals DYSF, FKTN, and ISPD Mutations in Congenital Muscular Dystrophy Without Brain or Eye Involvement The Harvard community has made this article openly available. Please share
Whole Exome Sequencing Reveals DYSF, FKTN, and ISPD Mutations in Congenital Muscular Dystrophy Without Brain or Eye Involvement The Harvard community has made this article openly available. Please share
Ubiquitous calpains 1 and 2 and calpastatin: mouse models
 Some Calpain History- Part 2: GENETICS and EVOLUTION The information for calpain genetics is organized by genes for component subunits of calpain-1 (Capn1 and Capns1), calpain-2 (Capn2 and Capns1) and
Some Calpain History- Part 2: GENETICS and EVOLUTION The information for calpain genetics is organized by genes for component subunits of calpain-1 (Capn1 and Capns1), calpain-2 (Capn2 and Capns1) and
Fukutin Gene Mutations Cause Dilated Cardiomyopathy with Minimal Muscle Weakness
 Fukutin Gene Mutations Cause Dilated Cardiomyopathy with Minimal Muscle Weakness Terumi Murakami, MD, 1,2 Yukiko K. Hayashi MD, PhD, 1 Satoru Noguchi, PhD 1,2 Megumu Ogawa, BS 1 Ikuya Nonaka, MD, PhD,
Fukutin Gene Mutations Cause Dilated Cardiomyopathy with Minimal Muscle Weakness Terumi Murakami, MD, 1,2 Yukiko K. Hayashi MD, PhD, 1 Satoru Noguchi, PhD 1,2 Megumu Ogawa, BS 1 Ikuya Nonaka, MD, PhD,
Petri Net Representation and Analysis of Mannose Type O-Glycan Biosynthesis
 Institute of Experimental Morphology, Pathology and Anthropology with Museum Bulgarian Anatomical Society Acta morphologica et anthropologica, 25 (1-2) Sofia 2018 Petri Net Representation and Analysis
Institute of Experimental Morphology, Pathology and Anthropology with Museum Bulgarian Anatomical Society Acta morphologica et anthropologica, 25 (1-2) Sofia 2018 Petri Net Representation and Analysis
PROCEEDINGS OF LGMD days MEETING: Prognosis and Treatment in LGMD. October 15-17, IRCCS S. Camillo, Lido di Venezia (VE), Italy
 Acta Myologica 204; XXXIII: p. 55-65 PROCEEDINGS OF LGMD days MEETING: Prognosis and Treatment in LGMD October 5-7, 204 IRCCS S. Camillo, Lido di Venezia (VE), Italy 55 56 WEDNESDAY OCTOBER 5 th 9.5 Registration
Acta Myologica 204; XXXIII: p. 55-65 PROCEEDINGS OF LGMD days MEETING: Prognosis and Treatment in LGMD October 5-7, 204 IRCCS S. Camillo, Lido di Venezia (VE), Italy 55 56 WEDNESDAY OCTOBER 5 th 9.5 Registration
Early-Onset LMNA-Associated Muscular Dystrophy with Later Involvement of Contracture
 JCN Open Access pissn 1738-6586 / eissn 2005-5013 / J Clin Neurol 2017;13(4):405-410 / https://doi.org/10.3988/jcn.2017.13.4.405 ORIGINAL ARTICLE Early-Onset LMNA-Associated Muscular Dystrophy with Later
JCN Open Access pissn 1738-6586 / eissn 2005-5013 / J Clin Neurol 2017;13(4):405-410 / https://doi.org/10.3988/jcn.2017.13.4.405 ORIGINAL ARTICLE Early-Onset LMNA-Associated Muscular Dystrophy with Later
Introduction. Overview
 Congenital muscular dystrophies Emma Clement MD ( Dr. Clement of Great Ormond Street Children's Hospital has no relevant financial relationships to disclose. ) Heinz Jungbluth MD PhD ( Dr. Jungbluth of
Congenital muscular dystrophies Emma Clement MD ( Dr. Clement of Great Ormond Street Children's Hospital has no relevant financial relationships to disclose. ) Heinz Jungbluth MD PhD ( Dr. Jungbluth of
Genetics of Inclusion Body Myositis
 Genetics of Inclusion Body Myositis Thomas Lloyd, MD, PhD Associate Professor of Neurology and Neuroscience Co-director, Johns Hopkins Myositis Center Sporadic IBM (IBM) Age at onset usually > 50 Prevalence
Genetics of Inclusion Body Myositis Thomas Lloyd, MD, PhD Associate Professor of Neurology and Neuroscience Co-director, Johns Hopkins Myositis Center Sporadic IBM (IBM) Age at onset usually > 50 Prevalence
The enigma of 7q36-linked autosomal dominant limb-girdle muscular dystrophy
 The enigma of 7q36-linked autosomal dominant limb-girdle muscular dystrophy Satu M Sandell, Sanna Huovinen, Jaakko Sarparanta, Helena Luque, Olayinka Raheem, Hannu Haapasalo, Peter Hackman, Bjarne Udd
The enigma of 7q36-linked autosomal dominant limb-girdle muscular dystrophy Satu M Sandell, Sanna Huovinen, Jaakko Sarparanta, Helena Luque, Olayinka Raheem, Hannu Haapasalo, Peter Hackman, Bjarne Udd
K. P. Loss of sarcolemma nnos in sarcoglycandeficient
 Loss of sarcolemma nnos in sarcoglycan-deficient muscle RACHELLE H. CROSBIE 1,2, RITA BARRESI 1, AND KEVIN P. CAMPBELL 1 Howard Hughes Medical Institute, Department of Physiology and Biophysics, Department
Loss of sarcolemma nnos in sarcoglycan-deficient muscle RACHELLE H. CROSBIE 1,2, RITA BARRESI 1, AND KEVIN P. CAMPBELL 1 Howard Hughes Medical Institute, Department of Physiology and Biophysics, Department
Pathognomonic muscle imaging findings in DNAJB6 mutated LGMD1D
 European Journal of Neurology 2013, 20: 1553 1559 doi:10.1111/ene.12239 Pathognomonic muscle imaging findings in DNAJB6 mutated LGMD1D S. M. Sandell a,b, I. Mahjneh c,d, J. Palmio b, G. Tasca e, E. Ricci
European Journal of Neurology 2013, 20: 1553 1559 doi:10.1111/ene.12239 Pathognomonic muscle imaging findings in DNAJB6 mutated LGMD1D S. M. Sandell a,b, I. Mahjneh c,d, J. Palmio b, G. Tasca e, E. Ricci
Female Carrier of Duchenne Muscular Dystrophy Presenting With Secondary Dilated Cardiomyopathy : A Case Report
 J Cardiol 2001 ; 38: 35 40 Duchenne 1 Female Carrier of Duchenne Muscular Dystrophy Presenting With Secondary Dilated Cardiomyopathy : A Case Report Shigeki Kiyoaki Toru Kenji Kojiro HIRAMATSU, MD MAEKAWA,
J Cardiol 2001 ; 38: 35 40 Duchenne 1 Female Carrier of Duchenne Muscular Dystrophy Presenting With Secondary Dilated Cardiomyopathy : A Case Report Shigeki Kiyoaki Toru Kenji Kojiro HIRAMATSU, MD MAEKAWA,
Progress in human genetics has identified a number. -Sarcoglycan Deficiency Leads to Muscle Membrane Defects and Apoptosis Independent of Dystrophin
 -Sarcoglycan Deficiency Leads to Muscle Membrane Defects and Apoptosis Independent of Dystrophin Andrew A. Hack,* Chantal T. Ly, Fang Jiang, Cynthia J. Clendenin, Kirsten S. Sigrist, Robert L. Wollmann,
-Sarcoglycan Deficiency Leads to Muscle Membrane Defects and Apoptosis Independent of Dystrophin Andrew A. Hack,* Chantal T. Ly, Fang Jiang, Cynthia J. Clendenin, Kirsten S. Sigrist, Robert L. Wollmann,
Refining genotype ^phenotype correlations in muscular dystrophies with defective glycosylation of dystroglycan
 Brain Advance Access published September 18, 2007 doi:10.1093/brain/awm212 Brain (2007)Page1of11 Refining genotype ^phenotype correlations in muscular dystrophies with defective glycosylation of dystroglycan
Brain Advance Access published September 18, 2007 doi:10.1093/brain/awm212 Brain (2007)Page1of11 Refining genotype ^phenotype correlations in muscular dystrophies with defective glycosylation of dystroglycan
CARDIAC muscle is commonly affected in muscular
 362 THE NEW ENGLAND JOURNAL OF MEDICINE Feb. 8, 1996 BRIEF REPORT: DEFICIENCY OF A DYSTROPHIN-ASSOCIATED GLYCOPROTEIN (ADHALIN) IN A PATIENT WITH MUSCULAR DYSTROPHY AND CARDIOMYOPATHY RICARDO FADIC, M.D.,
362 THE NEW ENGLAND JOURNAL OF MEDICINE Feb. 8, 1996 BRIEF REPORT: DEFICIENCY OF A DYSTROPHIN-ASSOCIATED GLYCOPROTEIN (ADHALIN) IN A PATIENT WITH MUSCULAR DYSTROPHY AND CARDIOMYOPATHY RICARDO FADIC, M.D.,
Animal Models for Genetic Neuromuscular Diseases
 J Mol Neurosci (2008) 34:241 248 DOI 10.1007/s12031-007-9023-9 Animal Models for Genetic Neuromuscular Diseases Mariz Vainzof & Danielle Ayub-Guerrieri & Paula C. G. Onofre & Poliana C. M. Martins & Vanessa
J Mol Neurosci (2008) 34:241 248 DOI 10.1007/s12031-007-9023-9 Animal Models for Genetic Neuromuscular Diseases Mariz Vainzof & Danielle Ayub-Guerrieri & Paula C. G. Onofre & Poliana C. M. Martins & Vanessa
Familial DilatedCardiomyopathy Georgios K Efthimiadis, MD
 Familial DilatedCardiomyopathy Georgios K Efthimiadis, MD Dilated Cardiomyopathy Dilated LV/RV, reduced EF, in the absence of CAD valvulopathy pericardial disease Prevalence:40/100.000 persons Natural
Familial DilatedCardiomyopathy Georgios K Efthimiadis, MD Dilated Cardiomyopathy Dilated LV/RV, reduced EF, in the absence of CAD valvulopathy pericardial disease Prevalence:40/100.000 persons Natural
Abnormalities of dystrophin, the sarcoglycans, and laminin a2 in the muscular dystrophies
 JMed Genet 1998;35:379-386 Department of Clinical Genetics, Royal Alexandra Hospital for Children, Westmead, Sydney, Australia K J Jones Neurogenetics Research Unit, Clinical Sciences Building, Royal Alexandra
JMed Genet 1998;35:379-386 Department of Clinical Genetics, Royal Alexandra Hospital for Children, Westmead, Sydney, Australia K J Jones Neurogenetics Research Unit, Clinical Sciences Building, Royal Alexandra
THE JOURNAL OF CELL BIOLOGY
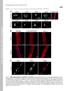 Supplemental Material Mejat et al., http://www.jcb.org/cgi/content/full/jcb.200811035/dc1 THE JOURNAL OF CELL BIOLOGY Figure S1. SUN1, SUN2, and Nesprin-1 localization in muscle fibers. (A) SUN1 and SUN2
Supplemental Material Mejat et al., http://www.jcb.org/cgi/content/full/jcb.200811035/dc1 THE JOURNAL OF CELL BIOLOGY Figure S1. SUN1, SUN2, and Nesprin-1 localization in muscle fibers. (A) SUN1 and SUN2
