Distal and proximal RTA. Detlef Bockenhauer
|
|
|
- Kimberly Stevens
- 5 years ago
- Views:
Transcription
1 Distal and proximal RTA Detlef Bockenhauer
2 acidosis: Is it real?
3 H + + HCO 3 - <=>> H2 O + CO 2
4 What do you need to assess an acid-base disorder Blood: blood gas, electrolytes including bicarbonate (measured) Urine: ph, electrolytes The bicarbonate (or base excess) on a gas is calculated => less reliable
5 Then you just look it up!
6 Renal tubular acidosis Acid-base homoeostasis is maintained by the kidney Excretion of acid should be equal to acid ingested (acid load)
7 Renal Acid Load Depends on diet: animal proteins are high in sulfur-containing AA (cystine, methionine) and thus acidproducing. Vegetable fruit, tubers, roots and leafy green vegetables contain salts of organic acids, which are metabolised to bicarbonate. Typical western diet presents a net acid load of ~50 mmol to the kidney
8 Renal Bicarbonate Handling In order to excrete acid, the kidney first needs to reclaim the filtered HCO 3 - Approximately 4 Mol of HCO 3 - is filtered each day % is reclaimed in proximal tubule % in distal nephron
9 Proximal tubule CA4 CA2 NBCe1/SLC4A4 NHE3/SLC9A3
10 Distal Tubule Principal cell K + ROMK t ATPV0A4 ATPV1B1 2K + ENaC t Na + CA2 AE1/SLC4A1 Na-K-ATPase 3Na +
11 And now to the clinical approach ph<7.37, HCO 3- <20 yes Diarrhoea? GI-fistula? Stoma? Intestinal Bicarbonate losses Fanconi Syndrome yes normal no <5.5 Urine ph Glucosuria? Aaciduria? Low TmP? no Isolated prta invalid data Anion gap >6.5 Serum K + low high high Inherited drta Methanol Uraemia DKA Paraldehyde Iron Lactate Ethylene glycol/ethanol Salicylic acid Hyperkalaemic RTA Aldosterone insufficiency/ resistance Renal failure Obstructive uropathy
12 The anion gap Na + - [Cl - +HCO 3 - ] = AG Normal: 8-12
13 What is the anion gap Unmeasured anions, mostly proteins Therefore: in hypoalbuminaemia expect low anion gap Correction: anion gap decreases by 2.5 for each 10g/l of serum albumin below 40 g/l
14 Renal tubular acidosis = non-anion gap acidosis Non-anion gap = bicarbonate loss Bicarbonate is lost through gastrointestinal tract (diarrhoea) or the kidney (RTA) Because of electroneutrality, bicarbonate loss must be accompanied by cation loss, typically sodium Na + - [Cl - + HCO 3- ] = AG
15 The difference between anion gap and non-anion gap acidosis Anion gap = acid gain. Addition of acid, where the corresponding base is an unmeasured anion, such as lactate or ketones Thus, the added proton is buffered by bicarbonate, decreasing the serum level, but the added anion is unmeasured. Na + - [Cl - + HCO 3 - ] = AG
16 Clinical consequences of renal Failure-to-thrive tubular acidosis Protons replace calcium ions in the bone, resulting in Rickets Hypercalciuria/nephrocalcinosis
17 Case 1 6 weeks old baby referred for failure to thrive. Born FT with a birth weight of 3.22 kg. Failed newborn hearing screen Examination: weight 3.0 kg, otherwise unremarkable
18 Laboratory Investigations Blood Na 147 mmol/l K 2.3 mmol/l Cl 127 mmol/l HCO 3 10 mmol/l ph 7.20 Urine Na 60 mmol/l K 36 mmol/l Cl 62 mmol/l ph 8.0
19 Renal Ultrasound
20 ph=7.20, HCO 3- =10 Anion gap AG=147-(127+10)=10=normal
21 ph<7.37, HCO 3- <20 Diarrhoea? GI-fistula? Stoma? normal Anion gap
22 What is usually used to distingiush between renal and non renal acidosis?
23 Urine anion gap? Urine Cation gap! Cl - Na + K = UCG
24 ph<7.37, HCO 3- <20 Diarrhoea? GI-fistula? Stoma? normal Anion gap no Urine ph Urine ph = 8.0
25 ph<7.37, HCO 3- <20 Diarrhoea? GI-fistula? Stoma? normal Anion gap no Urine ph >6.5 Serum K + K = 2.3 mmol/l
26 ph<7.37, HCO 3- <20 Diarrhoea? GI-fistula? Stoma? normal Anion gap no Urine ph >6.5 Serum K + low Inherited drta
27 Distal Tubule Principal cell MRCR K + ROMK t ATPV0A4 ATPV1B1 2K + ENaC t Na + CA2 AE1/SLC4A1 Na-K-ATPase 3Na +
28 Case 2 A 7 day old baby is brought to the A&E department because of weight loss, vomiting and being listless. Birth weight was 2.75 kg and on admission she weighs 2.32 kg with poor perfusion and unmeasurable blood pressure
29 Laboratory Investigations Blood Na 126 mmol/l K 8.8 mmol/l Cl 107 mmol/l HCO 3 10 mmol/l ph 7.16 Urine Na 196 mmol/l K <1 mmol/l Cl 168 mmol/l ph 7.5
30 ph=7.20, HCO 3- =10 Anion gap AG=126-(107+10)=11=normal
31 ph<7.37, HCO 3- <20 Diarrhoea? GI-fistula? Stoma? normal Anion gap
32 ph<7.37, HCO 3- <20 Diarrhoea? GI-fistula? Stoma? normal Anion gap no Urine ph Urine ph =7.5
33 ph<7.37, HCO 3- <20 Diarrhoea? GI-fistula? Stoma? normal Anion gap no Urine ph >6.5 Serum K + K=8.8 mmol/l
34 ph<7.37, HCO 3- <20 Diarrhoea? GI-fistula? Stoma? normal Anion gap no Urine ph >6.5 Serum K + high Hyperkalaemic RTA Aldosterone insufficiency/ resistance Renal failure Obstructive uropathy
35 Distal Tubule Principal cell MRCR K + ROMK t ATPV0A4 ATPV1B1 2K + ENaC t Na + CA2 AE1/SLC4A1 Na-K-ATPase 3Na +
36 Case 3 A 2 year old boy is brought to clinic because of failure to thrive. His weight has fallen below the 0.4 th %ile. His examination is remarkable for widening of chosto-chondral junctions and wrists.
37 Laboratory Investigations Blood Na 137 mmol/l K 2.6 mmol/l Cl 110 mmol/l HCO 3 14 mmol/l ph 7.24 Urine Na 24 mmol/l K 13 mmol/l Cl <50 mmol/l ph 6.0
38 ph=7.20, HCO 3- =10 Anion gap AG=137-(110+14)=13=? normal
39 ph<7.37, HCO 3- <20 Diarrhoea? GI-fistula? Stoma? normal Anion gap
40 Diarrhoea? GI-fistula? Stoma? ph<7.37, HCO 3- <20 normal no <5.5 Urine ph Anion gap Urine ph = 6.0 >6.5
41 ph<7.37, HCO 3- <20 Diarrhoea? GI-fistula? Stoma? Fanconi Syndrome yes normal no Urine ph <5.5 Glucosuria? Aaciduria? Low TmP? Anion gap >6.5 TRP: 58% ( ) (TmP/GFR: 0.56) Urine RBP: +glucosuria
42 Case 4 A 1 year old girl is referred to clinic because of her family history of dominant drta (mother and maternal grandmother). She has been well, thriving and developing. Her examination is unremarkable (height and weight on the second percentile).
43 Laboratory Investigations Blood Na 144 mmol/l K 4.3 mmol/l Cl 108 mmol/l HCO 3 20 mmol/l ph 7.34 Urine Na 14 mmol/l K 54 mmol/l Cl 45mmol/l ph 7.5 Calcium/creatinine ratio 0.36 ( )
44 Ultrasound
45 Is she affected?
46 Follow-up Admitted locally age 17 months with diarrhoea and found to have low plasma bicarbonate (15 mmol) Seen again in clinic age 18 month Blood Na 140 mmol/l K 4.2 mmol/l Cl 108 mmol/l HCO 3 16 mmol/l ph 7.29 Urine Na 47 mmol/l K 191 mmol/l Cl 100 mmol/l ph 7.5 Calcium/creatinine ratio 0.34 ( )
47 Case 5 3-y old girl referred as family moved to the UK History of cystic/dilated left kidney, initially noted on antenatal scans and confirmed postnatally s/p left nephrectomy age 6 months Noted to be acidotic at the time and commences bicarb supplementation, currently 20 mmol four times a day History of excessive tearing. Eye examination reportedly showed band keratopathy and glaucoma History of congenital hip dysplasia Family history: twin brother, reportedly healthy, except for vision problems. Parents are first cousins
48 Examination Well appearing, normal examination, except for opaqueness in eyes Weight: kg (<0.4 th %ile) Height: 84.7 cm (0.4the %ile) BP: 102 mmhg systolic Urine dip: neg. protein, neg. blood, ph 9, SG: 1.010
49 Look into my eyes
50 Laboratory Investigations Blood Na [mmol/l] 140 K [mmol/l] 3.8 Cl [mmol/l] 109 HCO3 [mmol/l] 17 Creatinine [mcmol/l] 42 ph 7.32
51 Diagnosis? Proximal RTA with eye findings Mutation in SLC4A4
52 SLC4A4
53 Treatment Alkali supplementation, now on 18ml tricitrate four times daily (equivalent to 144 mmol bicarb/day Proximal tubule normally reabsorbs ~5000 mmol/d..
54 The brother Because of vision problems also seen by ophthalmologist: same diagnosis: band keratopathy and glaucoma Weight: 0.4 th %ile Blood tests show acidosis.
55 Questions?
3/19/2009. The task of the kidney in acid-base balance Excretion of the daily acid load. Buffering of an acid load. A o B - + H + B - A o +OH - C +
 The task of the kidney in acid-base balance Excretion of the daily acid load Buffering of an acid load Oxidation of amino acids, fats and carbohydrates often lead to acid production. On an average American
The task of the kidney in acid-base balance Excretion of the daily acid load Buffering of an acid load Oxidation of amino acids, fats and carbohydrates often lead to acid production. On an average American
RENAL TUBULAR ACIDOSIS An Overview
 RENAL TUBULAR ACIDOSIS An Overview UNIVERSITY OF PNG SCHOOL OF MEDICINE AND HEALTH SCIENCES DISCIPLINE OF BIOCHEMISTRY & MOLECULAR BIOLOGY CLINICAL BIOCHEMISTRY PBL MBBS IV VJ. Temple 1 What is Renal Tubular
RENAL TUBULAR ACIDOSIS An Overview UNIVERSITY OF PNG SCHOOL OF MEDICINE AND HEALTH SCIENCES DISCIPLINE OF BIOCHEMISTRY & MOLECULAR BIOLOGY CLINICAL BIOCHEMISTRY PBL MBBS IV VJ. Temple 1 What is Renal Tubular
Acid-Base Tutorial 2/10/2014. Overview. Physiology (2) Physiology (1)
 Overview Acid-Base Tutorial Nicola Barlow Physiology Buffering systems Control mechanisms Laboratory assessment of acid-base Disorders of H + ion homeostasis Respiratory acidosis Metabolic acidosis Respiratory
Overview Acid-Base Tutorial Nicola Barlow Physiology Buffering systems Control mechanisms Laboratory assessment of acid-base Disorders of H + ion homeostasis Respiratory acidosis Metabolic acidosis Respiratory
Acid Base Balance. Professor Dr. Raid M. H. Al-Salih. Clinical Chemistry Professor Dr. Raid M. H. Al-Salih
 Acid Base Balance 1 HYDROGEN ION CONCENTRATION and CONCEPT OF ph Blood hydrogen ion concentration (abbreviated [H + ]) is maintained within tight limits in health, with the normal concentration being between
Acid Base Balance 1 HYDROGEN ION CONCENTRATION and CONCEPT OF ph Blood hydrogen ion concentration (abbreviated [H + ]) is maintained within tight limits in health, with the normal concentration being between
Anna Vinnikova, M.D. Division of Nephrology Virginia Commonwealth University
 Metabolic Acidosis Anna Vinnikova, M.D. Division of Nephrology Virginia Commonwealth University Graphics by permission from The Fluid, Electrolyte and Acid-Base Companion, S. Faubel and J. Topf, http://www.pbfluids.com
Metabolic Acidosis Anna Vinnikova, M.D. Division of Nephrology Virginia Commonwealth University Graphics by permission from The Fluid, Electrolyte and Acid-Base Companion, S. Faubel and J. Topf, http://www.pbfluids.com
Guidelines for approach to a child with
 Guidelines for approach to a child with Metabolic acidosis (including RTA) Children s Kidney Centre University Hospital of Wales Cardiff CF14 4XW DISCLAIMER: These guidelines were produced in good faith
Guidelines for approach to a child with Metabolic acidosis (including RTA) Children s Kidney Centre University Hospital of Wales Cardiff CF14 4XW DISCLAIMER: These guidelines were produced in good faith
Renal Tubular Acidosis
 1 Renal Tubular Acidosis Mohammad Tariq Ibrahim 6 th Grade Diyala College Of Medicine supervisor DR. Sabah Almaamoory 2 *Renal Tubular Acidosis:- RTA:- is a disease state characterized by a normal anion
1 Renal Tubular Acidosis Mohammad Tariq Ibrahim 6 th Grade Diyala College Of Medicine supervisor DR. Sabah Almaamoory 2 *Renal Tubular Acidosis:- RTA:- is a disease state characterized by a normal anion
PedsCases Podcast Scripts
 PedsCases Podcast Scripts This is a text version of a podcast from Pedscases.com on Approach to Acid-Base Disturbance. These podcasts are designed to give medical students an overview of key topics in
PedsCases Podcast Scripts This is a text version of a podcast from Pedscases.com on Approach to Acid-Base Disturbance. These podcasts are designed to give medical students an overview of key topics in
Slide 1. Slide 2. Slide 3. Learning Outcomes. Acid base terminology ARTERIAL BLOOD GAS INTERPRETATION
 Slide 1 ARTERIAL BLOOD GAS INTERPRETATION David O Neill MSc BSc RN NMP FHEA Associate Lecturer (Non Medical Prescribing) Cardiff University Advanced Nurse Practitioner Respiratory Medicine Slide 2 Learning
Slide 1 ARTERIAL BLOOD GAS INTERPRETATION David O Neill MSc BSc RN NMP FHEA Associate Lecturer (Non Medical Prescribing) Cardiff University Advanced Nurse Practitioner Respiratory Medicine Slide 2 Learning
ACID-BASE BALANCE URINE BLOOD AIR
 ACIDBASE BALANCE URINE BLOOD AIR H 2 PO 4 NH 4 HCO 3 KIDNEY H H HCO 3 CELLS Hb H LUNG H 2 CO 3 HHb CO 2 H 2 O ph = 7.4 [HCO 3 ] = 24 meq/l PCO 2 = 40 mm Hg CO 2 PRIMARY RENAL MECHANISMS INVOLVED IN ACIDBASE
ACIDBASE BALANCE URINE BLOOD AIR H 2 PO 4 NH 4 HCO 3 KIDNEY H H HCO 3 CELLS Hb H LUNG H 2 CO 3 HHb CO 2 H 2 O ph = 7.4 [HCO 3 ] = 24 meq/l PCO 2 = 40 mm Hg CO 2 PRIMARY RENAL MECHANISMS INVOLVED IN ACIDBASE
Hyponatraemia. Detlef Bockenhauer
 Hyponatraemia Detlef Bockenhauer Key message Plasma sodium can be low due to either excess water or deficiency of salt In clinical practice, dysnatraemias almost always reflect an abnormality of water
Hyponatraemia Detlef Bockenhauer Key message Plasma sodium can be low due to either excess water or deficiency of salt In clinical practice, dysnatraemias almost always reflect an abnormality of water
9/14/2017. Acid-Base Disturbances. Goal. Provide an approach to determine complex acid-base disorders
 Acid-Base Disturbances NCNP October 10, 2017 Eric Magaña, M.D. Presbyterian Medical Center Department of Pulmonary and Critical Care Medicine Goal Provide an approach to determine complex acid-base disorders
Acid-Base Disturbances NCNP October 10, 2017 Eric Magaña, M.D. Presbyterian Medical Center Department of Pulmonary and Critical Care Medicine Goal Provide an approach to determine complex acid-base disorders
Acid-Base Imbalance-2 Lecture 9 (12/4/2015) Yanal A. Shafagoj MD. PhD
 AcidBase Imbalance2 Lecture 9 (12/4/2015) Yanal A. Shafagoj MD. PhD Introduction Disturbance in acidbase balance are common clinical problem that range in severity from mild to life threatening, the acute
AcidBase Imbalance2 Lecture 9 (12/4/2015) Yanal A. Shafagoj MD. PhD Introduction Disturbance in acidbase balance are common clinical problem that range in severity from mild to life threatening, the acute
RENAL FUNCTION An Overview
 RENAL FUNCTION An Overview UNIVERSITY OF PNG SCHOOL OF MEDICINE AND HEALTH SCIENCES DIVISION OF BASIC MEDICAL SCIENCES DISCIPLINE OF BIOCHEMISTRY & MOLECULAR BIOLOGY PBL MBBS II SEMINAR VJ. Temple 1 Kidneys
RENAL FUNCTION An Overview UNIVERSITY OF PNG SCHOOL OF MEDICINE AND HEALTH SCIENCES DIVISION OF BASIC MEDICAL SCIENCES DISCIPLINE OF BIOCHEMISTRY & MOLECULAR BIOLOGY PBL MBBS II SEMINAR VJ. Temple 1 Kidneys
MS1 Physiology Review of Na+, K+, H + /HCO 3. /Acid-base, Ca+² and PO 4 physiology
 MS1 Physiology Review of,, / /Acidbase, Ca+² and PO 4 physiology I. David Weiner, M.D. Professor of Medicine and Physiology University of Florida College of Medicine Basic principles Proximal tubule Majority
MS1 Physiology Review of,, / /Acidbase, Ca+² and PO 4 physiology I. David Weiner, M.D. Professor of Medicine and Physiology University of Florida College of Medicine Basic principles Proximal tubule Majority
Physiology week 16 Renal 2 (volume/buffers)
 Physiology week 16 Renal 2 (volume/buffers) Defense of Tonicity and Volume Defense of tonicity Tonicity = osmolality of a solution relative to plasma Osmolality measures [ ] all particles in solution,
Physiology week 16 Renal 2 (volume/buffers) Defense of Tonicity and Volume Defense of tonicity Tonicity = osmolality of a solution relative to plasma Osmolality measures [ ] all particles in solution,
The kidney. (Pseudo) Practical questions. The kidneys are all about keeping the body s homeostasis. for questions Ella
 The kidney (Pseudo) Practical questions for questions Ella (striemit@gmail.com) The kidneys are all about keeping the body s homeostasis Ingestion Product of metabolism H 2 O Ca ++ Cl - K + Na + H 2 O
The kidney (Pseudo) Practical questions for questions Ella (striemit@gmail.com) The kidneys are all about keeping the body s homeostasis Ingestion Product of metabolism H 2 O Ca ++ Cl - K + Na + H 2 O
3/17/2017. Acid-Base Disturbances. Goal. Eric Magaña, M.D. Presbyterian Medical Center Department of Pulmonary and Critical Care Medicine
 Acid-Base Disturbances Eric Magaña, M.D. Presbyterian Medical Center Department of Pulmonary and Critical Care Medicine Goal Provide an approach to determine complex acid-base disorders Discuss the approach
Acid-Base Disturbances Eric Magaña, M.D. Presbyterian Medical Center Department of Pulmonary and Critical Care Medicine Goal Provide an approach to determine complex acid-base disorders Discuss the approach
Acid and Base Balance
 Acid and Base Balance 1 2 The Body and ph Homeostasis of ph is tightly controlled Extracellular fluid = 7.4 Blood = 7.35 7.45 < 7.35: Acidosis (acidemia) > 7.45: Alkalosis (alkalemia) < 6.8 or > 8.0: death
Acid and Base Balance 1 2 The Body and ph Homeostasis of ph is tightly controlled Extracellular fluid = 7.4 Blood = 7.35 7.45 < 7.35: Acidosis (acidemia) > 7.45: Alkalosis (alkalemia) < 6.8 or > 8.0: death
Non-Anion Gap Metabolic Acidosis. App.GoSoapbox.com then Join Now. Joel M. Topf, M.D.
 Non-Anion Gap Metabolic Acidosis App.GoSoapbox.com 665-971-584 then Join Now Joel M. Topf, M.D. http://pbfluids.com @kidney_boy App.GoSoapbox.com 665-971-584 32 y.o. female with fatigue, weakness and muscle
Non-Anion Gap Metabolic Acidosis App.GoSoapbox.com 665-971-584 then Join Now Joel M. Topf, M.D. http://pbfluids.com @kidney_boy App.GoSoapbox.com 665-971-584 32 y.o. female with fatigue, weakness and muscle
[Chapter 25] The Liver and the Gut. John A Kellum
![[Chapter 25] The Liver and the Gut. John A Kellum [Chapter 25] The Liver and the Gut. John A Kellum](/thumbs/85/92127350.jpg) [Chapter 25] The Liver and the Gut John A Kellum 25.1 Introduction Throughout this book we have seen that the Stewart formulation can be used to help understand the mechanisms of acid-base equilibrium
[Chapter 25] The Liver and the Gut John A Kellum 25.1 Introduction Throughout this book we have seen that the Stewart formulation can be used to help understand the mechanisms of acid-base equilibrium
SIMPLY Arterial Blood Gases Interpretation. Week 4 Dr William Dooley
 SIMPLY Arterial Blood Gases Interpretation Week 4 Dr William Dooley Plan Structure for interpretation 5-step approach Works for majority of cases Case scenarios Some common concerns A-a gradient BE Anion
SIMPLY Arterial Blood Gases Interpretation Week 4 Dr William Dooley Plan Structure for interpretation 5-step approach Works for majority of cases Case scenarios Some common concerns A-a gradient BE Anion
Acid Base Disorders: Key Core Concepts. Thomas DuBose M.D., MACP, FASN ASN Board Review Course Online Resource Material 2014
 Acid Base Disorders: Key Core Concepts Thomas DuBose M.D., MACP, FASN ASN Board Review Course Online Resource Material 2014 Speaker Disclosure I, Thomas DuBose, M.D., have no financial relationships or
Acid Base Disorders: Key Core Concepts Thomas DuBose M.D., MACP, FASN ASN Board Review Course Online Resource Material 2014 Speaker Disclosure I, Thomas DuBose, M.D., have no financial relationships or
CASE 27. What is the response of the kidney to metabolic acidosis? What is the response of the kidney to a respiratory alkalosis?
 CASE 27 A 21-year-old man with insulin-dependent diabetes presents to the emergency center with mental status changes, nausea, vomiting, abdominal pain, and rapid respirations. On examination, the patient
CASE 27 A 21-year-old man with insulin-dependent diabetes presents to the emergency center with mental status changes, nausea, vomiting, abdominal pain, and rapid respirations. On examination, the patient
Distal renal tubular acidosis: genetic and clinical spectrum
 Distal renal tubular acidosis: genetic and clinical spectrum Sabrina Giglio Medical Genetics Unit, Meyer Children s University Hospital, University of Florence sabrina.giglio@meyer.it sabrinarita.giglio@unifi.it
Distal renal tubular acidosis: genetic and clinical spectrum Sabrina Giglio Medical Genetics Unit, Meyer Children s University Hospital, University of Florence sabrina.giglio@meyer.it sabrinarita.giglio@unifi.it
Module : Clinical correlates of disorders of metabolism Block 3, Week 2
 Module : Clinical correlates of disorders of metabolism Block 3, Week 2 Department of Paediatrics and Child Health University of Pretoria Tutor : Prof DF Wittenberg : dwittenb@medic.up.ac.za Aim of this
Module : Clinical correlates of disorders of metabolism Block 3, Week 2 Department of Paediatrics and Child Health University of Pretoria Tutor : Prof DF Wittenberg : dwittenb@medic.up.ac.za Aim of this
Biochemistry of acid-base disorders. Alice Skoumalová
 Biochemistry of acid-base disorders Alice Skoumalová Main topics of the lecture: Measurement of acid-base dysbalance Classification of the acid-base disorders 4 basic acid-base disorders and their compensaiton
Biochemistry of acid-base disorders Alice Skoumalová Main topics of the lecture: Measurement of acid-base dysbalance Classification of the acid-base disorders 4 basic acid-base disorders and their compensaiton
Acids and Bases their definitions and meanings
 Acids and Bases their definitions and meanings Molecules containing hydrogen atoms that can release hydrogen ions in solutions are referred to as acids. (HCl H + Cl ) (H 2 CO 3 H + HCO 3 ) A base is an
Acids and Bases their definitions and meanings Molecules containing hydrogen atoms that can release hydrogen ions in solutions are referred to as acids. (HCl H + Cl ) (H 2 CO 3 H + HCO 3 ) A base is an
Chapter 27: WATER, ELECTROLYTES, AND ACID-BASE BALANCE
 Chapter 27: WATER, ELECTROLYTES, AND ACID-BASE BALANCE I. RELATED TOPICS Integumentary system Cerebrospinal fluid Aqueous humor Digestive juices Feces Capillary dynamics Lymph circulation Edema Osmosis
Chapter 27: WATER, ELECTROLYTES, AND ACID-BASE BALANCE I. RELATED TOPICS Integumentary system Cerebrospinal fluid Aqueous humor Digestive juices Feces Capillary dynamics Lymph circulation Edema Osmosis
1 st JAMAICAN PAEDIATRIC NEPHROLOGY CONFERENCE
 in association with 1 st JAMAICAN PAEDIATRIC NEPHROLOGY CONFERENCE Jamaica Conference Centre Kingston Jamaica October 4 th 2014 Demys&fying Renal Tubular Acidosis Rulan S. Parekh Professor of Pediatrics
in association with 1 st JAMAICAN PAEDIATRIC NEPHROLOGY CONFERENCE Jamaica Conference Centre Kingston Jamaica October 4 th 2014 Demys&fying Renal Tubular Acidosis Rulan S. Parekh Professor of Pediatrics
Disorders o f of water water Detlef Bockenhauer
 Disorders of water Detlef Bockenhauer How do we measure water? How do we measure water? Not directly! Reflected best in Na concentration Water overload => Hyponatraemia Water deficiency => Hypernatraemia
Disorders of water Detlef Bockenhauer How do we measure water? How do we measure water? Not directly! Reflected best in Na concentration Water overload => Hyponatraemia Water deficiency => Hypernatraemia
Metabolic Alkalosis: Vomiting
 RENAL ANL) ACID-BASE PHYSIOLOGY 213 Case 37 Metabolic Alkalosis: Vomiting Maria Cuervo is a 20-year-old philosophy major at a state university. When the "24-hour" stomach flu went around campus during
RENAL ANL) ACID-BASE PHYSIOLOGY 213 Case 37 Metabolic Alkalosis: Vomiting Maria Cuervo is a 20-year-old philosophy major at a state university. When the "24-hour" stomach flu went around campus during
Arterial Blood Gas Analysis
 Arterial Blood Gas Analysis L Lester www.3bv.org Bones, Brains & Blood Vessels Drawn from radial or femoral arteries. Invasive procedure Caution must be taken with patient on anticoagulants ph: 7.35-7.45
Arterial Blood Gas Analysis L Lester www.3bv.org Bones, Brains & Blood Vessels Drawn from radial or femoral arteries. Invasive procedure Caution must be taken with patient on anticoagulants ph: 7.35-7.45
INTRAVENOUS FLUIDS PRINCIPLES
 INTRAVENOUS FLUIDS PRINCIPLES Postnatal physiological weight loss is approximately 5-10% Postnatal diuresis is delayed in Respiratory Distress Syndrome (RDS) Preterm babies have limited capacity to excrete
INTRAVENOUS FLUIDS PRINCIPLES Postnatal physiological weight loss is approximately 5-10% Postnatal diuresis is delayed in Respiratory Distress Syndrome (RDS) Preterm babies have limited capacity to excrete
Physio 12 -Summer 02 - Renal Physiology - Page 1
 Physiology 12 Kidney and Fluid regulation Guyton Ch 20, 21,22,23 Roles of the Kidney Regulation of body fluid osmolarity and electrolytes Regulation of acid-base balance (ph) Excretion of natural wastes
Physiology 12 Kidney and Fluid regulation Guyton Ch 20, 21,22,23 Roles of the Kidney Regulation of body fluid osmolarity and electrolytes Regulation of acid-base balance (ph) Excretion of natural wastes
Acid-Base Balance Workshop. Dr. Najla Al Kuwaiti Dr. Abdullah Al Ameri Dr. Amar Al Shibli
 Acid-Base Balance Workshop Dr. Najla Al Kuwaiti Dr. Abdullah Al Ameri Dr. Amar Al Shibli Objectives Normal Acid-Base Physiology Simple Acid-Base Disorders Compensations and Disorders The Anion Gap Mixed
Acid-Base Balance Workshop Dr. Najla Al Kuwaiti Dr. Abdullah Al Ameri Dr. Amar Al Shibli Objectives Normal Acid-Base Physiology Simple Acid-Base Disorders Compensations and Disorders The Anion Gap Mixed
A case of DYSELECTROLYTEMIA. Dr. Prathyusha Dr. Lalitha janakiraman s unit
 A case of DYSELECTROLYTEMIA Dr. Prathyusha Dr. Lalitha janakiraman s unit CASE SUMMARY 4 month old, female infant 1 st born to NC parents, term, b.wt: 3.25kg No neonatal hospitalization Attained head control
A case of DYSELECTROLYTEMIA Dr. Prathyusha Dr. Lalitha janakiraman s unit CASE SUMMARY 4 month old, female infant 1 st born to NC parents, term, b.wt: 3.25kg No neonatal hospitalization Attained head control
BUFFERING OF HYDROGEN LOAD
 BUFFERING OF HYDROGEN LOAD 1. Extracellular space minutes 2. Intracellular space minutes to hours 3. Respiratory compensation 6 to 12 hours 4. Renal compensation hours, up to 2-3 days RENAL HYDROGEN SECRETION
BUFFERING OF HYDROGEN LOAD 1. Extracellular space minutes 2. Intracellular space minutes to hours 3. Respiratory compensation 6 to 12 hours 4. Renal compensation hours, up to 2-3 days RENAL HYDROGEN SECRETION
Arterial Blood Gas Interpretation: The Basics
 http://www.medicine-on-line.com ABG Basics: Page 1/10 Arterial Blood Gas Interpretation: The Basics Author: David C Chung MD, FRCPC Affiliation: The Chinese University of Hong Kong Sampling of arterial
http://www.medicine-on-line.com ABG Basics: Page 1/10 Arterial Blood Gas Interpretation: The Basics Author: David C Chung MD, FRCPC Affiliation: The Chinese University of Hong Kong Sampling of arterial
1. What is the acid-base disturbance in this patient?
 /ABG QUIZ QUIZ 1. What is the acid-base disturbance in this patient? Presenting complaint: pneumonia 1 point Uncompensated metabolic alkalosis Partially compensated respiratory alkalosis Mixed alkalosis
/ABG QUIZ QUIZ 1. What is the acid-base disturbance in this patient? Presenting complaint: pneumonia 1 point Uncompensated metabolic alkalosis Partially compensated respiratory alkalosis Mixed alkalosis
D fini n tion: p = = -log [H+] ph=7 me m an s 10-7 Mol M H+ + (100 nmol m /l); ) p ; H=8 me m an s 10-8 Mol M H+ + (10 (10 n nmol m /l) Nor
![D fini n tion: p = = -log [H+] ph=7 me m an s 10-7 Mol M H+ + (100 nmol m /l); ) p ; H=8 me m an s 10-8 Mol M H+ + (10 (10 n nmol m /l) Nor D fini n tion: p = = -log [H+] ph=7 me m an s 10-7 Mol M H+ + (100 nmol m /l); ) p ; H=8 me m an s 10-8 Mol M H+ + (10 (10 n nmol m /l) Nor](/thumbs/75/72756397.jpg) Definition: ph regulation ph = -log [H + ] ph=7 means 10-7 Mol H + (100 nmol/l); ph=8 means 10 Normal plasma value: 7.35-7.45; 7.45; (H Acidosis: ph7.45 Intracellular ph = 7.1-7.3
Definition: ph regulation ph = -log [H + ] ph=7 means 10-7 Mol H + (100 nmol/l); ph=8 means 10 Normal plasma value: 7.35-7.45; 7.45; (H Acidosis: ph7.45 Intracellular ph = 7.1-7.3
Ricky Bell Renal/ICM Registrar
 Ricky Bell Renal/ICM Registrar Objectives When to call renal How to manage the patient with AKI How the manage the patient with CKD (HD/PD) Special AKI situations What do the guidelines say? My referral
Ricky Bell Renal/ICM Registrar Objectives When to call renal How to manage the patient with AKI How the manage the patient with CKD (HD/PD) Special AKI situations What do the guidelines say? My referral
Fluid therapy in children
 Fluid therapy in children TJ Neuhaus and G Reusz Lucerne and Budapest ESPN 2012 Kraków Parenteral maintenance in children Daily requirements - Holliday and Segar, 1957 Fluid Patient weight: 25 kg 1-10
Fluid therapy in children TJ Neuhaus and G Reusz Lucerne and Budapest ESPN 2012 Kraków Parenteral maintenance in children Daily requirements - Holliday and Segar, 1957 Fluid Patient weight: 25 kg 1-10
DBL MAGNESIUM SULFATE CONCENTRATED INJECTION
 DBL MAGNESIUM SULFATE CONCENTRATED INJECTION NAME OF MEDICINE Magnesium Sulfate BP DESCRIPTION DBL Magnesium Sulfate Concentrated Injection is a clear, colourless, sterile solution. Each ampoule contains
DBL MAGNESIUM SULFATE CONCENTRATED INJECTION NAME OF MEDICINE Magnesium Sulfate BP DESCRIPTION DBL Magnesium Sulfate Concentrated Injection is a clear, colourless, sterile solution. Each ampoule contains
There are number of parameters which are measured: ph Oxygen (O 2 ) Carbon Dioxide (CO 2 ) Bicarbonate (HCO 3 -) AaDO 2 O 2 Content O 2 Saturation
 Arterial Blood Gases (ABG) A blood gas is exactly that...it measures the dissolved gases in your bloodstream. This provides one of the best measurements of what is known as the acid-base balance. The body
Arterial Blood Gases (ABG) A blood gas is exactly that...it measures the dissolved gases in your bloodstream. This provides one of the best measurements of what is known as the acid-base balance. The body
Inter Inter Pretation of Acid Base Disturbance in Critically ill Patients. By :-: Dr. Vinay Bhomia M.D.
 Inter Inter Pretation of Acid Base Disturbance in Critically ill Patients. By :-: Dr. Vinay Bhomia M.D. Normal Blood PH 7.35 to 7.45 Crucial importance to maintain homeostatic function of Body. Any Significant
Inter Inter Pretation of Acid Base Disturbance in Critically ill Patients. By :-: Dr. Vinay Bhomia M.D. Normal Blood PH 7.35 to 7.45 Crucial importance to maintain homeostatic function of Body. Any Significant
INTRAVENOUS FLUID THERAPY
 INTRAVENOUS FLUID THERAPY PRINCIPLES Postnatal physiological weight loss is approximately 5 10% in first week of life Preterm neonates have more total body water and may lose 10 15% of their weight in
INTRAVENOUS FLUID THERAPY PRINCIPLES Postnatal physiological weight loss is approximately 5 10% in first week of life Preterm neonates have more total body water and may lose 10 15% of their weight in
NORMAL POTASSIUM DISTRIBUTION AND BALANCE
 NORMAL POTASSIUM DISTRIBUTION AND BALANCE 98% of body potassium is contained within cells, principally muscle cells, and is readily exchangeable. Only 2% is in ECF. Daily intake exceeds the amount in ECF.
NORMAL POTASSIUM DISTRIBUTION AND BALANCE 98% of body potassium is contained within cells, principally muscle cells, and is readily exchangeable. Only 2% is in ECF. Daily intake exceeds the amount in ECF.
UNIT 9 INVESTIGATION OF ACID-BASE DISTURBANCES
 UNIT 9 INVESTIGATION OF ACIDBASE DISTURBANCES LEARNING OBJECTIVES At the end of this chapter, students must be able to: 1. Describe the main parametres that define the acidbase equilibrium 2. Identify
UNIT 9 INVESTIGATION OF ACIDBASE DISTURBANCES LEARNING OBJECTIVES At the end of this chapter, students must be able to: 1. Describe the main parametres that define the acidbase equilibrium 2. Identify
Water, Electrolytes, and Acid-Base Balance
 Chapter 27 Water, Electrolytes, and Acid-Base Balance 1 Body Fluids Intracellular fluid compartment All fluids inside cells of body About 40% of total body weight Extracellular fluid compartment All fluids
Chapter 27 Water, Electrolytes, and Acid-Base Balance 1 Body Fluids Intracellular fluid compartment All fluids inside cells of body About 40% of total body weight Extracellular fluid compartment All fluids
Dr. Suzana Voiculescu
 Dr. Suzana Voiculescu AB balance parameters Extracellular ph (plasmatic ph)= 7.35-7.45 < 7.35= acidosis >7.45= alkalosis Kassirer-Bleich equation [H+] = 24 PCO2/ [HCO3-] predicts that the ratio of dissolved
Dr. Suzana Voiculescu AB balance parameters Extracellular ph (plasmatic ph)= 7.35-7.45 < 7.35= acidosis >7.45= alkalosis Kassirer-Bleich equation [H+] = 24 PCO2/ [HCO3-] predicts that the ratio of dissolved
There are many buffers in the kidney, but the main one is the phosphate buffer.
 9 Yanal Obada Zalat Renal Control of AcidBase Balance The kidneys play three major roles in the maintenance of normal acidbase balance: 1excretion of H+ (fixed _non volatile H+) 2Reabsorption of filtrated
9 Yanal Obada Zalat Renal Control of AcidBase Balance The kidneys play three major roles in the maintenance of normal acidbase balance: 1excretion of H+ (fixed _non volatile H+) 2Reabsorption of filtrated
Renal Physiology - Lectures
 Renal Physiology - Lectures Physiology of Body Fluids PROBLEM SET, RESEARCH ARTICLE Structure & Function of the Kidneys Renal Clearance & Glomerular Filtration PROBLEM SET Regulation of Renal Blood Flow
Renal Physiology - Lectures Physiology of Body Fluids PROBLEM SET, RESEARCH ARTICLE Structure & Function of the Kidneys Renal Clearance & Glomerular Filtration PROBLEM SET Regulation of Renal Blood Flow
Chapter 21. Diuretic Agents. Mosby items and derived items 2008, 2002 by Mosby, Inc., an affiliate of Elsevier Inc.
 Chapter 21 Diuretic Agents Renal Structure and Function Kidneys at level of umbilicus Each weighs 160 to 175 g and is 10 to 12 cm long Most blood flow per gram of weight in body 22% of cardiac output (CO)
Chapter 21 Diuretic Agents Renal Structure and Function Kidneys at level of umbilicus Each weighs 160 to 175 g and is 10 to 12 cm long Most blood flow per gram of weight in body 22% of cardiac output (CO)
Important aspects of acid-base disorders
 Important aspects of acid-base disorders I. David Weiner, M.D. Co-holder, C. Craig and Audrae Tisher Chair in Nephrology Division of Nephrology, Hypertension and Transplantation University of Florida College
Important aspects of acid-base disorders I. David Weiner, M.D. Co-holder, C. Craig and Audrae Tisher Chair in Nephrology Division of Nephrology, Hypertension and Transplantation University of Florida College
Na + Transport 1 and 2 Linda Costanzo, Ph.D.
 Na + Transport 1 and 2 Linda Costanzo, Ph.D. OBJECTIVES: After studying this lecture, the student should understand: 1. The terminology applied to single nephron function, including the meaning of TF/P
Na + Transport 1 and 2 Linda Costanzo, Ph.D. OBJECTIVES: After studying this lecture, the student should understand: 1. The terminology applied to single nephron function, including the meaning of TF/P
Dr. Suzana Voiculescu Discipline of Physiology and Fundamental Neurosciences Carol Davila Univ. of Medicine and Pharmacy
 Dr. Suzana Voiculescu Discipline of Physiology and Fundamental Neurosciences Carol Davila Univ. of Medicine and Pharmacy AB balance parameters Extracellular ph (plasmatic ph)= 7.35-7.45 < 7.35= acidosis
Dr. Suzana Voiculescu Discipline of Physiology and Fundamental Neurosciences Carol Davila Univ. of Medicine and Pharmacy AB balance parameters Extracellular ph (plasmatic ph)= 7.35-7.45 < 7.35= acidosis
WATER, SODIUM AND POTASSIUM
 WATER, SODIUM AND POTASSIUM Attila Miseta Tamás Kőszegi Department of Laboratory Medicine, 2016 1 Average daily water intake and output of a normal adult 2 Approximate contributions to plasma osmolality
WATER, SODIUM AND POTASSIUM Attila Miseta Tamás Kőszegi Department of Laboratory Medicine, 2016 1 Average daily water intake and output of a normal adult 2 Approximate contributions to plasma osmolality
RTA) Tubular. Unravelling Renal. Acidosis (RTA( Robert Unwin. (Centre for Nephrology London Epithelial Group University College London)
 Unravelling Renal Tubular Acidosis (RTA( RTA) Robert Unwin (Centre for Nephrology London Epithelial Group University College London) An outline of my talk A definition of RTA Clinical aspects of RTA Underlying
Unravelling Renal Tubular Acidosis (RTA( RTA) Robert Unwin (Centre for Nephrology London Epithelial Group University College London) An outline of my talk A definition of RTA Clinical aspects of RTA Underlying
What location in the gastrointestinal (GI) tract has tight, or impermeable, junctions between the epithelial cells?
 CASE 32 A 17-year-old boy presents to his primary care physician with complaints of diarrhea for the last 2 days. The patient states that he just returned to the United States after visiting relatives
CASE 32 A 17-year-old boy presents to his primary care physician with complaints of diarrhea for the last 2 days. The patient states that he just returned to the United States after visiting relatives
Acid-Base Balance 11/18/2011. Regulation of Potassium Balance. Regulation of Potassium Balance. Regulatory Site: Cortical Collecting Ducts.
 Influence of Other Hormones on Sodium Balance Acid-Base Balance Estrogens: Enhance NaCl reabsorption by renal tubules May cause water retention during menstrual cycles Are responsible for edema during
Influence of Other Hormones on Sodium Balance Acid-Base Balance Estrogens: Enhance NaCl reabsorption by renal tubules May cause water retention during menstrual cycles Are responsible for edema during
The principal functions of the kidneys
 Renal physiology The principal functions of the kidneys Formation and excretion of urine Excretion of waste products, drugs, and toxins Regulation of body water and mineral content of the body Maintenance
Renal physiology The principal functions of the kidneys Formation and excretion of urine Excretion of waste products, drugs, and toxins Regulation of body water and mineral content of the body Maintenance
Electrolytes by case examples. Graham Bilbrough, European Medical Affairs Manager
 Electrolytes by case examples Graham Bilbrough, European Medical Affairs Manager 1 Acid-bases disturbances Generally result from one of the following: 1. damage to an organ such as the kidneys or lungs
Electrolytes by case examples Graham Bilbrough, European Medical Affairs Manager 1 Acid-bases disturbances Generally result from one of the following: 1. damage to an organ such as the kidneys or lungs
Renal Physiology Part II. Bio 219 Napa Valley College Dr. Adam Ross
 Renal Physiology Part II Bio 219 Napa Valley College Dr. Adam Ross Fluid and Electrolyte balance As we know from our previous studies: Water and ions need to be balanced in order to maintain proper homeostatic
Renal Physiology Part II Bio 219 Napa Valley College Dr. Adam Ross Fluid and Electrolyte balance As we know from our previous studies: Water and ions need to be balanced in order to maintain proper homeostatic
New aspects of acid-base disorders
 New aspects of acid-base disorders I. David Weiner, M.D. C. Craig and Audrae Tisher Chair in Nephrology Division of Nephrology, Hypertension and Transplantation University of Florida College of Medicine
New aspects of acid-base disorders I. David Weiner, M.D. C. Craig and Audrae Tisher Chair in Nephrology Division of Nephrology, Hypertension and Transplantation University of Florida College of Medicine
RENAL SYSTEM 2 TRANSPORT PROPERTIES OF NEPHRON SEGMENTS Emma Jakoi, Ph.D.
 RENAL SYSTEM 2 TRANSPORT PROPERTIES OF NEPHRON SEGMENTS Emma Jakoi, Ph.D. Learning Objectives 1. Identify the region of the renal tubule in which reabsorption and secretion occur. 2. Describe the cellular
RENAL SYSTEM 2 TRANSPORT PROPERTIES OF NEPHRON SEGMENTS Emma Jakoi, Ph.D. Learning Objectives 1. Identify the region of the renal tubule in which reabsorption and secretion occur. 2. Describe the cellular
UNIT VI: ACID BASE IMBALANCE
 UNIT VI: ACID BASE IMBALANCE 1 Objectives: Review the physiological mechanism responsible to regulate acid base balance in the body i.e.: Buffers (phosphate, hemoglobin, carbonate) Renal mechanism Respiratory
UNIT VI: ACID BASE IMBALANCE 1 Objectives: Review the physiological mechanism responsible to regulate acid base balance in the body i.e.: Buffers (phosphate, hemoglobin, carbonate) Renal mechanism Respiratory
Functions of Proximal Convoluted Tubules
 1. Proximal tubule Solute reabsorption in the proximal tubule is isosmotic (water follows solute osmotically and tubular fluid osmolality remains similar to that of plasma) 60-70% of water and solute reabsorption
1. Proximal tubule Solute reabsorption in the proximal tubule is isosmotic (water follows solute osmotically and tubular fluid osmolality remains similar to that of plasma) 60-70% of water and solute reabsorption
S.M. Gorbatkin MD, PhD Emory Board Review in Internal Medicine July Renal Division Department of Veterans Affairs Medical Center Atlanta
 S.M. Gorbatkin MD, PhD Emory Board Review in Internal Medicine July 2016 External Industry Relationships * Equity, stock, or options in biomedical industry companies or publishers Board of Directors or
S.M. Gorbatkin MD, PhD Emory Board Review in Internal Medicine July 2016 External Industry Relationships * Equity, stock, or options in biomedical industry companies or publishers Board of Directors or
Interpretation of ABG. Chandra Shekhar Bala, FCPS( Medicine) Junior Consultant NINS and Hospital, Dhaka
 Interpretation of ABG Chandra Shekhar Bala, FCPS( Medicine) Junior Consultant NINS and Hospital, Dhaka ABG analysis of Ms Rubi Ms. Rubi, 20 year-old lady PH 7.29 presented with breathlessness. She had
Interpretation of ABG Chandra Shekhar Bala, FCPS( Medicine) Junior Consultant NINS and Hospital, Dhaka ABG analysis of Ms Rubi Ms. Rubi, 20 year-old lady PH 7.29 presented with breathlessness. She had
Chapter 19 The Urinary System Fluid and Electrolyte Balance
 Chapter 19 The Urinary System Fluid and Electrolyte Balance Chapter Outline The Concept of Balance Water Balance Sodium Balance Potassium Balance Calcium Balance Interactions between Fluid and Electrolyte
Chapter 19 The Urinary System Fluid and Electrolyte Balance Chapter Outline The Concept of Balance Water Balance Sodium Balance Potassium Balance Calcium Balance Interactions between Fluid and Electrolyte
Oxygen and ABG. Dr Will Dooley
 Oxygen and ABG G Dr Will Dooley Oxygen and ABGs Simply in 10 cases Recap of: ABG interpretation Oxygen management Some common concerns A-a gradient Base Excess Anion Gap COPD patients CPAP/BiPAP First
Oxygen and ABG G Dr Will Dooley Oxygen and ABGs Simply in 10 cases Recap of: ABG interpretation Oxygen management Some common concerns A-a gradient Base Excess Anion Gap COPD patients CPAP/BiPAP First
RENAL PHYSIOLOGY, HOMEOSTASIS OF FLUID COMPARTMENTS
 RENAL PHYSIOLOGY, HOMEOSTASIS OF FLUID COMPARTMENTS (2) Dr. Attila Nagy 2017 TUBULAR FUNCTIONS (Learning objectives 54-57) 1 Tubular Transport About 99% of filtrated water and more than 90% of the filtrated
RENAL PHYSIOLOGY, HOMEOSTASIS OF FLUID COMPARTMENTS (2) Dr. Attila Nagy 2017 TUBULAR FUNCTIONS (Learning objectives 54-57) 1 Tubular Transport About 99% of filtrated water and more than 90% of the filtrated
Managing Acid Base and Electrolyte Disturbances with RRT
 Managing Acid Base and Electrolyte Disturbances with RRT John R Prowle MA MSc MD MRCP FFICM Consultant in Intensive Care & Renal Medicine RRT for Regulation of Acid-base and Electrolyte Acid base load
Managing Acid Base and Electrolyte Disturbances with RRT John R Prowle MA MSc MD MRCP FFICM Consultant in Intensive Care & Renal Medicine RRT for Regulation of Acid-base and Electrolyte Acid base load
BIOL 2402 Fluid/Electrolyte Regulation
 Dr. Chris Doumen Collin County Community College BIOL 2402 Fluid/Electrolyte Regulation 1 Body Water Content On average, we are 50-60 % water For a 70 kg male = 40 liters water This water is divided into
Dr. Chris Doumen Collin County Community College BIOL 2402 Fluid/Electrolyte Regulation 1 Body Water Content On average, we are 50-60 % water For a 70 kg male = 40 liters water This water is divided into
Urinary System. Dr. ZHANG Xiong. Dept. of Physiology. ZJU School of Medicine. QUESTION 6
 Urinary System Dr. ZHANG Xiong Dept. of Physiology ZJU School of Medicine http://10.71.121.158 Copyright@ Xiong Zhang QUESTION 6 How is the filtrate reabsorbed in tubular system? Copyright@ Xiong Zhang
Urinary System Dr. ZHANG Xiong Dept. of Physiology ZJU School of Medicine http://10.71.121.158 Copyright@ Xiong Zhang QUESTION 6 How is the filtrate reabsorbed in tubular system? Copyright@ Xiong Zhang
Instrumental determination of electrolytes in urine. Amal Alamri
 Instrumental determination of electrolytes in urine Amal Alamri What is the Electrolytes? Electrolytes are positively and negatively chargedions, Found in Within body's cells extracellular fluids, including
Instrumental determination of electrolytes in urine Amal Alamri What is the Electrolytes? Electrolytes are positively and negatively chargedions, Found in Within body's cells extracellular fluids, including
Introduction to the kidney: regulation of sodium & glucose. Dr Nick Ashton Senior Lecturer in Renal Physiology Faculty of Biology, Medicine & Health
 Introduction to the kidney: regulation of sodium & glucose Dr Nick Ashton Senior Lecturer in Renal Physiology Faculty of Biology, Medicine & Health Objectives Overview of kidney structure & function Glomerular
Introduction to the kidney: regulation of sodium & glucose Dr Nick Ashton Senior Lecturer in Renal Physiology Faculty of Biology, Medicine & Health Objectives Overview of kidney structure & function Glomerular
Acute Kidney Injury. Eleanor Haskey BSc(hons) RVN VTS(ECC) VPAC A1
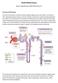 Acute Kidney Injury Eleanor Haskey BSc(hons) RVN VTS(ECC) VPAC A1 Anatomy and Physiology The role of the kidneys is to filter the blood through the glomerulus to form filtrate. The filtrate is then reabsorbed
Acute Kidney Injury Eleanor Haskey BSc(hons) RVN VTS(ECC) VPAC A1 Anatomy and Physiology The role of the kidneys is to filter the blood through the glomerulus to form filtrate. The filtrate is then reabsorbed
Acid-base mysteries explained. Friedrich Luft Experimental and Clinical Research Center, Berlin-Buch
 Acid-base mysteries explained Friedrich Luft Experimental and Clinical Research Center, Berlin-Buch Hydrogen ion concentration ph 7.4 In plasma 40 nmol/l (compared to mmol for other electrolytes M, mmol,
Acid-base mysteries explained Friedrich Luft Experimental and Clinical Research Center, Berlin-Buch Hydrogen ion concentration ph 7.4 In plasma 40 nmol/l (compared to mmol for other electrolytes M, mmol,
Martin Konrad has documented that he has no relevant financial relationships to disclose or conflict of interest to resolve.
 Martin Konrad has documented that he has no relevant financial relationships to disclose or conflict of interest to resolve. Nephrocalcinosis Clinical / Genetic Work-up and Outcome Martin Konrad University
Martin Konrad has documented that he has no relevant financial relationships to disclose or conflict of interest to resolve. Nephrocalcinosis Clinical / Genetic Work-up and Outcome Martin Konrad University
11/05/1431. Urine Formation by the Kidneys Tubular Processing of the Glomerular Filtrate
 Urine Formation by the Kidneys Tubular Processing of the Glomerular Filtrate Chapter 27 pages 327 347 1 OBJECTIVES At the end of this lecture you should be able to describe: Absorptive Characteristics
Urine Formation by the Kidneys Tubular Processing of the Glomerular Filtrate Chapter 27 pages 327 347 1 OBJECTIVES At the end of this lecture you should be able to describe: Absorptive Characteristics
Chapter 26 Fluid, Electrolyte, and Acid- Base Balance
 Chapter 26 Fluid, Electrolyte, and Acid- Base Balance 1 Body Water Content Infants: 73% or more water (low body fat, low bone mass) Adult males: ~60% water Adult females: ~50% water (higher fat content,
Chapter 26 Fluid, Electrolyte, and Acid- Base Balance 1 Body Water Content Infants: 73% or more water (low body fat, low bone mass) Adult males: ~60% water Adult females: ~50% water (higher fat content,
Interpretation of. No Da Vinci Code
 Interpretation of Mixed Acid Base Disorders- No Da Vinci Code by Surg Capt Aamir Ijaz, Consultant Pathologist / Assoc Prof of Pathology, PNS SHIFA ARTERIAL BLOOD GASES Measured on a Blood Gas Analyser.
Interpretation of Mixed Acid Base Disorders- No Da Vinci Code by Surg Capt Aamir Ijaz, Consultant Pathologist / Assoc Prof of Pathology, PNS SHIFA ARTERIAL BLOOD GASES Measured on a Blood Gas Analyser.
Acid-base balance is one of the most important of the body s homeostatic mechanisms Acid-base balance refers to regulation of hydrogen ion (H + )
 Acid-base balance is one of the most important of the body s homeostatic mechanisms Acid-base balance refers to regulation of hydrogen ion (H + ) concentration in body fluids Precise regulation of ph at
Acid-base balance is one of the most important of the body s homeostatic mechanisms Acid-base balance refers to regulation of hydrogen ion (H + ) concentration in body fluids Precise regulation of ph at
Nephron Structure inside Kidney:
 In-Depth on Kidney Nephron Structure inside Kidney: - Each nephron has two capillary regions in close proximity to the nephron tubule, the first capillary bed for fluid exchange is called the glomerulus,
In-Depth on Kidney Nephron Structure inside Kidney: - Each nephron has two capillary regions in close proximity to the nephron tubule, the first capillary bed for fluid exchange is called the glomerulus,
Introduction. Acids, Bases and ph; a review
 0 P a g e Introduction In this sheet, we discuss acidbase balance in our body and the role of kidneys in its establishment. Arrangement of topics is different from that of the lecture, to assure consistency
0 P a g e Introduction In this sheet, we discuss acidbase balance in our body and the role of kidneys in its establishment. Arrangement of topics is different from that of the lecture, to assure consistency
Note: During any ONE run the ph remains constant. It may be at any one of the above levels but it never change during a single run.
 1 BGYC34 (2007) PhysioEx Lab 10 AcidBase Balance Marking Scheme Part 1 Complete PhysioEx lab #10. Handin all of the pages associated with the lab. Note that there are 9 activities to be completed. You
1 BGYC34 (2007) PhysioEx Lab 10 AcidBase Balance Marking Scheme Part 1 Complete PhysioEx lab #10. Handin all of the pages associated with the lab. Note that there are 9 activities to be completed. You
Acid-Base Physiology. Dr. Tamás Bense Dr. Alexandra Turi
 Acid-Base Physiology Dr. Tamás Bense Dr. Alexandra Turi What is a blood gas assessment? We get it from an arterial sample (a.radialis, a. brachialis, a. femoralis) Invasive technique If the patient is
Acid-Base Physiology Dr. Tamás Bense Dr. Alexandra Turi What is a blood gas assessment? We get it from an arterial sample (a.radialis, a. brachialis, a. femoralis) Invasive technique If the patient is
Disorders of Acid-Base
 Disorders of Acid-Base Balance Bởi: OpenStaxCollege Normal arterial blood ph is restricted to a very narrow range of 7.35 to 7.45. A person who has a blood ph below 7.35 is considered to be in acidosis
Disorders of Acid-Base Balance Bởi: OpenStaxCollege Normal arterial blood ph is restricted to a very narrow range of 7.35 to 7.45. A person who has a blood ph below 7.35 is considered to be in acidosis
The Urinary S. (Chp. 10) & Excretion. What are the functions of the urinary system? Maintenance of water-salt and acidbase
 10.1 Urinary system The Urinary S. (Chp. 10) & Excretion 10.1 Urinary system What are the functions of the urinary system? 1. Excretion of metabolic wastes (urea, uric acid & creatinine) 1. Maintenance
10.1 Urinary system The Urinary S. (Chp. 10) & Excretion 10.1 Urinary system What are the functions of the urinary system? 1. Excretion of metabolic wastes (urea, uric acid & creatinine) 1. Maintenance
The relationship between H+,PaCO₂ and HCO₃ are expressed in the equation of:
 [Acid-Base Balance] [Dr. Bashir Khasawneh] [5 th February 2012] Acid-Base Basic Concepts: The relationship between H+,PaCO₂ and HCO₃ are expressed in the equation of: Which is modified from Henderson-Hasselbach
[Acid-Base Balance] [Dr. Bashir Khasawneh] [5 th February 2012] Acid-Base Basic Concepts: The relationship between H+,PaCO₂ and HCO₃ are expressed in the equation of: Which is modified from Henderson-Hasselbach
Principles of Anatomy and Physiology
 Principles of Anatomy and Physiology 14 th Edition CHAPTER 27 Fluid, Electrolyte, and Acid Base Fluid Compartments and Fluid In adults, body fluids make up between 55% and 65% of total body mass. Body
Principles of Anatomy and Physiology 14 th Edition CHAPTER 27 Fluid, Electrolyte, and Acid Base Fluid Compartments and Fluid In adults, body fluids make up between 55% and 65% of total body mass. Body
CHAPTER 27 LECTURE OUTLINE
 CHAPTER 27 LECTURE OUTLINE I. INTRODUCTION A. Body fluid refers to body water and its dissolved substances. B. Regulatory mechanisms insure homeostasis of body fluids since their malfunction may seriously
CHAPTER 27 LECTURE OUTLINE I. INTRODUCTION A. Body fluid refers to body water and its dissolved substances. B. Regulatory mechanisms insure homeostasis of body fluids since their malfunction may seriously
ARTERIAL BLOOD GASES PART 1 BACK TO BASICS SSR OLIVIA ELSWORTH SEPT 2017
 ARTERIAL BLOOD GASES PART 1 BACK TO BASICS SSR OLIVIA ELSWORTH SEPT 2017 WHAT INFORMATION DOES AN ABG GIVE US? ph = measure of hydrogen ion concentration (acidity or alkalinity) PaCO2 = partial pressure
ARTERIAL BLOOD GASES PART 1 BACK TO BASICS SSR OLIVIA ELSWORTH SEPT 2017 WHAT INFORMATION DOES AN ABG GIVE US? ph = measure of hydrogen ion concentration (acidity or alkalinity) PaCO2 = partial pressure
Acid-Base Balance Dr. Gary Mumaugh
 Acid-Base Balance Dr. Gary Mumaugh Introduction Acid-base balance is one of the most important of the body s homeostatic mechanisms Acid-base balance refers to regulation of hydrogen ion (H + ) concentration
Acid-Base Balance Dr. Gary Mumaugh Introduction Acid-base balance is one of the most important of the body s homeostatic mechanisms Acid-base balance refers to regulation of hydrogen ion (H + ) concentration
Dept. of Physiology. ZJU School of Medicine.
 Urinary System Dr. ZHANG Xiong Dept. of Physiology ZJU School of Medicine Http://10.10.10.151/Able.Acc2.Web/Template/View.aspx?action =view&coursetype=0&courseid=26519 QUESTION 6 How is the filtrate reabsorbed
Urinary System Dr. ZHANG Xiong Dept. of Physiology ZJU School of Medicine Http://10.10.10.151/Able.Acc2.Web/Template/View.aspx?action =view&coursetype=0&courseid=26519 QUESTION 6 How is the filtrate reabsorbed
Renal physiology V. Regulation of acid-base balance. Dr Alida Koorts BMS
 Renal physiology V Regulation of acidbase balance Dr Alida Koorts BMS 712 012 319 2921 akoorts@medic.up.ac.za Hydrogen ions (H + ): Concentration and origin Concentration in arterial blood, resting: [H
Renal physiology V Regulation of acidbase balance Dr Alida Koorts BMS 712 012 319 2921 akoorts@medic.up.ac.za Hydrogen ions (H + ): Concentration and origin Concentration in arterial blood, resting: [H
Clinical and molecular aspects of distal renal tubular acidosis in children
 Clinical and molecular aspects of distal renal tubular acidosis in children Martine TP Besouw 1, Marc Bienias 2, Patrick Walsh 3, Robert Kleta 1,4, William G van t Hoff 1, Emma Ashton 4, Lucy Jenkins,
Clinical and molecular aspects of distal renal tubular acidosis in children Martine TP Besouw 1, Marc Bienias 2, Patrick Walsh 3, Robert Kleta 1,4, William G van t Hoff 1, Emma Ashton 4, Lucy Jenkins,
ACID BASE BALANCE AND REGULATION OF ph
 ACID BASE BALANCE AND REGULATION OF ph 4 Chapter objectives After studying this chapter you should be able to: b Define the normal range for plasma ph. c Explain the role of the kidney in the steady state
ACID BASE BALANCE AND REGULATION OF ph 4 Chapter objectives After studying this chapter you should be able to: b Define the normal range for plasma ph. c Explain the role of the kidney in the steady state
