TOXIC EPIDERMAL NECROLYSIS AND CLARITHROMYCIN
|
|
|
- Lydia Townsend
- 6 years ago
- Views:
Transcription
1 TOXIC EPIDERMAL NECROLYSIS AND CLARITHROMYCIN Nadia Khaldi 1, Alain Miras 1, Sophie Gromb 1,2 1 Forensic Science Laboratory, Pellegrin Hospital, University Teaching Hospital Bordeaux, France, 2 Forensic Medicine and Medical Law at the Medicine University, Bordeaux, France Corresponding Author: sophie.gromb@chu-bordeaux.fr ABSTRACT Toxic epidermal necrolysis almost always occurs after taking a medication. Despite spectacular clinical signs, it is mainly diagnosed with pathologic techniques. The identification of a drug as the cause for the immune related cytotoxic reaction can be difficult if the molecule is not generally known to be a classical cause of this reaction. The present study describes a female patient who rapidly developed a severe bullous skin disease after taking clarithromycin for tonsillitis. The case illustrates the process involved in attributing causality to a molecule using an established imputability assessment framework. Key Words: Toxic epidermal necrolysis, clarithromycin, causality, macrolide A lthough rare, the unforeseen effects of medication are potentially dangerous and even life-threatening, so they must be documented in order to assess their frequency and circumstances of occurrence. 1,2 Toxic epidermal necrolysis (TEN) is a severe bullous skin disease inducing massive epidermal peeling with painful ulceration of all mucosal tissue. It rapidly extends and may cover the whole cutaneous-mucosal surface. Although it is rare, with an estimates incidence of one case per million inhabitants per year, it is fatal in approximately 30 % of cases. 1 In 90 % of cases it is triggered by an immunocytotoxic reaction to medication. 2,3,4 The most frequently incriminated molecules are anticonvulsants, non steroidal anti inflammatory drugs, allopurinol, and sulfonamide antibiotics. 5,6 Other antibiotics known to induce this syndrome are the aminopenicillins, the cephalosporins and the quinolones, while macrolides were thought to induce a specific allergic cutaneous reaction rather than TEN. A number of years ago an amendment was made to the pharmaceutical dictionary (VIDAL, 2001) mentioning the possible but exceptional occurrence of TEN with macrolides. We report a case in which clarithromycin (Naxy ), a semi-synthetic derivative of erythromycin A, triggered TEN. Case report While suffering from fever accompanied by laryngo-pharyngeal pain and diffuse myalgia, a 29-year-old woman without significant past medical history consulted her general practitioner. Tonsillitis was diagnosed and 10 days of treatment were given with clarithromycin (Naxy 250 ), paracetamol (Doliprane 500 ), acetylsalicylic acid (Aspegic 1000 ) and erdosteine (Vectrine ). Treatment commenced on the day of diagnosis, except for the aspirin which the patient did not use. The patient had previously received antibiotics on several occasions for upper respiratory tract infections, particularly the macrolide roxithromycin (Rulid ). Forty-eight hours after commencing treatment, a pruritic vesicular rash appeared on her face and thighs. Her general practitioner discontinued the clarithromycin and replaced it with amoxicillin (Clamoxyl ). Her state worsened during the night with the appearance of bullous lesions on her abdomen and a fever of 40 o C. She was hospitalized the following day and Stevens-Johnson syndrome was diagnosed die to the presence of peeling of the ocular, buccal, and vulvo-vaginal muscosa. On day 4, the peeling affected 70% of the body surface, and severe involvement of all mucosal tissue required her to be transferred to a specialized burn unit. Intensive e264
2 care was provided and included antibiotics (Vancomycin, Vancocine, Gentamycin, Gentalline, doxycyclin, doxicline ) for a cutaneous staphylococcal superinfection and pneumonia of the right lung. The patient also received paracetamol for several days owing to the persistence of fever. All complementary tests pointed to a diagnosis of TEN, particularly a skin biopsy that evidenced sub-epidermal bullous peeling accompanied by numerous necrotic keratinocytes and negative immunofluorescence. All bacteriological tests including search for mycoplasma and staphylococci were negative. No hypereosinophilia was found. The patient remained in hospital for one month with satisfactory re-epithelilization occurring in over 10 days. After one year, sequelae included dyschromic scars that persist on her skin and ophthalmologic lesions with decreased visual acuity, diffuse keratitis and a palpebral symblepharon. DISCUSSION For many years TEN was attributed to particularly slow acetylating genotypes, leading to the accumulation of toxic metabolites or to immune mechanisms involving reactive metabolites behaving as highly immunogenic haptenes. At present, several studies 7,8,9 point to the role of T lymphocytes directly reacting with a molecule and triggering the synthesis of cytokines, leading to the apoptosis of the keratinocytes and to epidermal necrolysis. Particular profiles favour the occurrence of TEN although the precise mechanism remains 3-4, 7-9 unknown. These include HIV infection, systemic lupus erythematesus, radiotherapy, bone marrow allograft and certain immunologic phenotypes such as HLA B12 and HLA DR4. 1 However, these factors were not relevant in this patient. The clinical presentation was characteristic of TEN except that the onset of clinical signs was faster after exposure than is usually described (7 to 21 days). The complications were classical, as pneumonia due to the peeling of the bronchial mucosa occurs in about 30 % of cases and ocular sequelae occur in 40 % of cases. 10,11 The diagnosis of TEN due to a medication is quite likely in this case but it is difficult to decide which molecule is to be incriminated since the severity of the symptoms ruled out any attempt at a cutaneous test or re-introduction. We therefore used the drug imputability criteria established by the French pharmacovigilance centers 12 which allowed us to establish a very strong suspicion of clarythromycin (Table 1). These criteria take into account both the chronology of events from the moment the drug is taken until the appearance of the lesions (intrinsic chronological imputability), the type of clinical sign (intrinsic semiologic imputability) and the data in the literature (extrinsic imputability). Clarithromycin would appear to be the most likely imputable drug since the patient was not taking any medication prior to the appearance of the bullous lesions, apart from a contraceptive pill (which she had taken for ten years); Paracetamol and erdosteine have been re-administered and tolerated since the event (semiologic criterion: S3).Even though TEN symptoms usually occur between days 7 and 21 after exposure to the precipitating molecule, there are rare cases where re-introduction of a molecule previously prescribed has triggered TEN in a much shorter time (< 3 days). For this reason, the previous administration of the macrolide roxithromycin (Rulid ) may be considered as a prior source of sensitisation and would explain the acceleration of the immune response (chronological criterion: C3). The overall intrinsic imputability score (I4) strongly suggests the involvement of clarithromycin. Finally, although there is very little published on macrolides causing TEN, the rareness of adverse event and the very low degree of involvement of antibiotics in this syndrome would have a limiting effect on the extrinsic imputability score (B1) with respect to the overall causality assessment. CONCLUSION TEN is a life-threatening condition causing serious ocular and cutaneous sequelae. A number of drugs are known to induce TEN and the list should include macrolides. Because clarithromycin is widely used it is important to bring the present case to the attention of the medical community so that prescribers can be aware of the risk. e265
3 REFERENCES 1. Wolkenstein P, Roujeau JC, Revuz J. Drug induced Toxic Epidermal Necrolysis. Clinics in dermatology 1998;16: Roujeau JC, Stern R. Severe adverse cutaneous reactions to drugs. The New England Journal of Medicine 1994;331: Knowles SR, Uetrecht J, Shear NH. Idiosyncratic drug reactions: the reactive metabolite syndromes. The Lancet 2000;356: Roujeau JC. Toxidermies médicamenteuses: quelques nouveautés. Revue Française d'allergologie immunologie clinique 2002;42: Paquet P. Drugs involved in Toxic Epidermal Necrolysis. Thérapie 1993;48(2): Roujeau JC, Kelly JP, Naldi L, et al. Medication use and risk of Steven-Johnson syndrome or Toxic Epidermal Necrolysis. The New England Journal of Medicine, 1995;333(24): Viard I, Wehrli P, Bullani R, et al. Inhibition of toxic epidermal necrolysis by blockade of CD95 with human intravenous immunoglobulin. Science 1998;282: Schnyder B, Burkhart C, Schnyder-Frutig K, et al. Recognition of sulfomethoxazole and its reactive metabolites by drug-specific CD4+T cells from allergic individuals. J Immunol 2000; 164: Yawallkar N, et al. Evidence for a role for IL-5 and eotaxin in activating and recruiting eosinophils in drug-induced cutaneous eruptions. Allergy Clinic Immunol 2000;106: Hans C, Paquet C, Maréchal-Courtois C. Le traitement ophtalmologique du Syndrome de Lyell. Revue Médicale de Liège, XLVIII; Becker SD. Toxic epidermal necrolysis. The Lancet 1998;351: Bégaud B, Evreux JC, Jouglard J, Lagier G. Imputabilité des effets inattendus ou toxiques des médicaments. Actualisation de la méthode utilisées en France. Thérapie 1985;40: e266
4 TABLE 1 Criteria used for imputability Chronologic criteria (C) Intrinsic imputability score Evolution when drug is curtailed Suggestive Inconclusive Non-suggestive Time to appearance of event Highly suggestive Compatibility Incompatibility C3 C3 C1 C3 C2 C1 C0 C3 C2 C1 C3 C1 C1 C0 C1 C1 C1 C1 C1 C1 C0 positive not made negative positive not made negative not made Relapse during involuntary re-administration of drug Highly suggestive: e.g., anaphylactic shock a few minutes after injection; fetal malformation for which administration of suspect drug coincides with the precise period during which involved organ is formed Compatibility: eczema-type reaction occurring several days after taking incompatible drug Incompatibility: fixed pigmented erythema occurring several weeks after taking drug Inconclusive: regression which is spontaneous or induced by non-specific dermocorticoid anti-histaminic symptomatic treatment, irreversible lesions, insufficient follow- up, drug not curtailed because essential Non-suggestive absence of regression of reversible type of event or complete regression despite continuation of drug C0 excluded ; C1 doubtful ; C2 plausible ; C3 probable Can J Clin Pharmacol Vol 12(3) Fall 2005:e264-e268; Nov. 8, 2005 e267
5 TABLE 1 Cont d Semiologic criteria (S) Intrinsic imputability score Semiology (clinical or para-clinical) Evocative of role of drug (and / or highly suggestive factor) Other semiologic possibilities Other explanation not attributable to drug Absent (after appropriate work-up) Possible S3 (present or not sought) S1 doubtful ; S2 plausible ; S3 probable positive S3 S3 S1 S3 S2 S1 S2 S1 S3 S1 S1 0 negative positive 0 negative Reliable specific complementary test ( 0 = test not available ) Chronologic criteria (C) Intrinsic imputability score (I) I0 excluded ; I1 doubtful ; I2 plausible ; I3 probable ; I4 very probable Semiologic criterion (S) S1 doubtful S2 plausible S3 probable C0 excluded I 0 I 0 I 0 C1 doubtful I 1 I 1 I 2 C2 plausible I 1 I 2 I 3 C3 probable I 3 I 3 I 4 Extrinsic imputability score (B) B0 Effect never published in international reference documents, even after exhaustive bibliographic search B1 B2 B3 Not well-known effect published once or twice only, different semiology or similar drug Effect not described in international reference works Well-known effect described in the VIDAL dictionary or in reference works e268 Can J Clin Pharmacol Vol 12(3) Fall 2005:e264-e268; Nov. 8, 2005
New product information wording Extracts from PRAC recommendations on signals
 12 October 2017 EMA/PRAC/610988/2017 Pharmacovigilance Risk Assessment Committee (PRAC) New product information wording Extracts from PRAC recommendations on signals Adopted at the 25-29 September 2017
12 October 2017 EMA/PRAC/610988/2017 Pharmacovigilance Risk Assessment Committee (PRAC) New product information wording Extracts from PRAC recommendations on signals Adopted at the 25-29 September 2017
Drug Allergy A Guide to Diagnosis and Management
 Drug Allergy A Guide to Diagnosis and Management (Version 1 April 2015 updated April 2018) Author: Jed Hewitt Chief Pharmacist, Governance & Professional Practice Date of Preparation: April 2015 Updated:
Drug Allergy A Guide to Diagnosis and Management (Version 1 April 2015 updated April 2018) Author: Jed Hewitt Chief Pharmacist, Governance & Professional Practice Date of Preparation: April 2015 Updated:
Correspondence should be addressed to Wanjarus Roongpisuthipong; rr
 Dermatology Research and Practice, Article ID 237821, 5 pages http://dx.doi.org/10.1155/2014/237821 Research Article Retrospective Analysis of Corticosteroid Treatment in Stevens-Johnson Syndrome and/or
Dermatology Research and Practice, Article ID 237821, 5 pages http://dx.doi.org/10.1155/2014/237821 Research Article Retrospective Analysis of Corticosteroid Treatment in Stevens-Johnson Syndrome and/or
Dilantin (phenytoin) ROBERT A. SCHWARTZ
 Dilantin (phenytoin) ROBERT A. SCHWARTZ Bailey & Galyen Attorney in Charge, Mass Tort Litigation Managing Attorney, Houston 18333 Egret Bay Blvd., Suite 120 Houston, Texas 77058 Toll Free: (866) 715-1529
Dilantin (phenytoin) ROBERT A. SCHWARTZ Bailey & Galyen Attorney in Charge, Mass Tort Litigation Managing Attorney, Houston 18333 Egret Bay Blvd., Suite 120 Houston, Texas 77058 Toll Free: (866) 715-1529
Emergency Dermatology Dr Melissa Barkham
 Emergency Dermatology Dr Melissa Barkham Spotlight Seminar 30 th September 2010 Why is this important? Urgent recognition and treatment of dermatologic emergencies can be life saving and prevent long term
Emergency Dermatology Dr Melissa Barkham Spotlight Seminar 30 th September 2010 Why is this important? Urgent recognition and treatment of dermatologic emergencies can be life saving and prevent long term
Skin Manifestations of Drug Reactions
 Skin Manifestations of Drug Reactions Dr Carol Hlela, Division of Dermatology Department of Medicine, University of Cape Town and Red Cross Children s Hospital What are the Skin Manifestations of Drug
Skin Manifestations of Drug Reactions Dr Carol Hlela, Division of Dermatology Department of Medicine, University of Cape Town and Red Cross Children s Hospital What are the Skin Manifestations of Drug
Case Report Cephazolin-Induced Toxic Epidermal Necrolysis Treated with Intravenous Immunoglobulin and N-Acetylcysteine
 Case Reports in Immunology Volume 2012, Article ID 931528, 4 pages doi:10.1155/2012/931528 Case Report Cephazolin-Induced Toxic Epidermal Necrolysis Treated with Intravenous Immunoglobulin and N-Acetylcysteine
Case Reports in Immunology Volume 2012, Article ID 931528, 4 pages doi:10.1155/2012/931528 Case Report Cephazolin-Induced Toxic Epidermal Necrolysis Treated with Intravenous Immunoglobulin and N-Acetylcysteine
Adverse Drug Reactions (ADRs) Outline
 Adverse Drug Reactions (ADRs) Outline 1. What are Adverse Drug Reactions (ADRs)? WHAT WHY HOW 2. How important are ADRs and are they preventable? 3. What are the classifications and mechanisms of ADRs?
Adverse Drug Reactions (ADRs) Outline 1. What are Adverse Drug Reactions (ADRs)? WHAT WHY HOW 2. How important are ADRs and are they preventable? 3. What are the classifications and mechanisms of ADRs?
They are updated regularly as new NICE guidance is published. To view the latest version of this NICE Pathway see:
 bring together everything NICE says on a topic in an interactive flowchart. are interactive and designed to be used online. They are updated regularly as new NICE guidance is published. To view the latest
bring together everything NICE says on a topic in an interactive flowchart. are interactive and designed to be used online. They are updated regularly as new NICE guidance is published. To view the latest
SKIN REACTIONS WITH PSYCHOTROPICS: A SYSTEMATIC REVIEW
 SKIN REACTIONS WITH PSYCHOTROPICS: A SYSTEMATIC REVIEW *Anderson Isaac, PharmD Candidate, 2019 Pooja Patel, PharmD Candidate, 2019 Katelyn Thomasson, PharmD Candidate, 2019 Erika Tillery, PharmD, BCPP,
SKIN REACTIONS WITH PSYCHOTROPICS: A SYSTEMATIC REVIEW *Anderson Isaac, PharmD Candidate, 2019 Pooja Patel, PharmD Candidate, 2019 Katelyn Thomasson, PharmD Candidate, 2019 Erika Tillery, PharmD, BCPP,
DERMATOLOGICAL EMERGENCIES. DR. Ian Hoyle MBBS DIP IMC RCS (Ed), DA (UK),FRACGP,FACRRM,DIP DERM(Wales) TASMANIAN SKIN AND BODY CENTRE
 DERMATOLOGICAL EMERGENCIES DR. Ian Hoyle MBBS DIP IMC RCS (Ed), DA (UK),FRACGP,FACRRM,DIP DERM(Wales) TASMANIAN SKIN AND BODY CENTRE Dermatological Emergencies INFECTIONS ERYTHRODERMA DRUG ERUPTIONS STEVENS-JOHNSON
DERMATOLOGICAL EMERGENCIES DR. Ian Hoyle MBBS DIP IMC RCS (Ed), DA (UK),FRACGP,FACRRM,DIP DERM(Wales) TASMANIAN SKIN AND BODY CENTRE Dermatological Emergencies INFECTIONS ERYTHRODERMA DRUG ERUPTIONS STEVENS-JOHNSON
allergy Asia Pacific Stevens-Johnson syndrome and toxic epidermal necrolysis in Dr. Hasan Sadikin General Hospital Bandung, Indonesia from
 pissn -876 eissn -868 Original Article Asia Pac Allergy 6;6:-7 Stevens-Johnson syndrome and toxic epidermal necrolysis in Dr. Hasan Sadikin General Hospital Bandung, Indonesia from 9 Oki Suwarsa *, Wulan
pissn -876 eissn -868 Original Article Asia Pac Allergy 6;6:-7 Stevens-Johnson syndrome and toxic epidermal necrolysis in Dr. Hasan Sadikin General Hospital Bandung, Indonesia from 9 Oki Suwarsa *, Wulan
Cutaneous Conditions Associated with Systemic Disease
 Cutaneous Conditions Associated with Systemic Disease Johnnie M Woodson, M.D., F.A.A.D. Assistant Professor of Dermatology University of Nevada School of Medicine Director of J. Woodson Dermatology & Associates,
Cutaneous Conditions Associated with Systemic Disease Johnnie M Woodson, M.D., F.A.A.D. Assistant Professor of Dermatology University of Nevada School of Medicine Director of J. Woodson Dermatology & Associates,
EFAVIRENZ ASSOCIATED STEVENS-JOHNSON SYNDROME
 EFAVIRENZ ASSOCIATED STEVENS-JOHNSON SYNDROME To the Editor: Persons with human immunodeficiency virus (HIV) infection are highly susceptible to adverse dermatological reactions to specific medications
EFAVIRENZ ASSOCIATED STEVENS-JOHNSON SYNDROME To the Editor: Persons with human immunodeficiency virus (HIV) infection are highly susceptible to adverse dermatological reactions to specific medications
REGISTRY OF SEVERE CUTANEOUS ADVERSE REACTIONS TO DRUGS AND COLLECTION OF BIOLOGICAL SAMPLES. R e g i S C A R PATIENT'S DATA. Age country of birth
 REGISTRY OF SEVERE CUTANEOUS ADVERSE REACTIONS TO DRUGS AND COLLECTION OF BIOLOGICAL SAMPLES R e g i S C A R PATIENT'S DATA Initials of the patient date of birth Age country of birth Gender male female
REGISTRY OF SEVERE CUTANEOUS ADVERSE REACTIONS TO DRUGS AND COLLECTION OF BIOLOGICAL SAMPLES R e g i S C A R PATIENT'S DATA Initials of the patient date of birth Age country of birth Gender male female
A Case Report on Amoxicillin Induced Stevens- Johnson Syndrome
 Open Journal of Clinical & Medical Case Reports Volume 2 (2016) Issue 11 A Case Report on Amoxicillin Induced Stevens- Johnson Syndrome Rajendra Singh Airee*; Aastha Rawal; Binu Mathew; H. Doddayya Abstract
Open Journal of Clinical & Medical Case Reports Volume 2 (2016) Issue 11 A Case Report on Amoxicillin Induced Stevens- Johnson Syndrome Rajendra Singh Airee*; Aastha Rawal; Binu Mathew; H. Doddayya Abstract
Adverse drug reactions
 Medications William Smith Adverse drug reactions Allergy? Side-effect? Intolerance? Background Adverse drug reactions (ADRs) vary from life-threatening anaphylaxis to minor common side-effects. Objective
Medications William Smith Adverse drug reactions Allergy? Side-effect? Intolerance? Background Adverse drug reactions (ADRs) vary from life-threatening anaphylaxis to minor common side-effects. Objective
Cutaneous Adverse Drug Reactions in Domestic Animals. Katherine Doerr, DVM, Dip. ACVD. Veterinary Dermatology Center
 Cutaneous Adverse Drug Reactions in Domestic Animals Katherine Doerr, DVM, Dip. ACVD Veterinary Dermatology Center Maitland, Rockledge, Waterford Lakes, FL Not highly studied in veterinary medicine Unknown
Cutaneous Adverse Drug Reactions in Domestic Animals Katherine Doerr, DVM, Dip. ACVD Veterinary Dermatology Center Maitland, Rockledge, Waterford Lakes, FL Not highly studied in veterinary medicine Unknown
A Middle-Aged Man with Newly Diagnosed HIV Infection and Rash
 CLINICAL CASE OF THE MONTH A Middle-Aged Man with Newly Diagnosed HIV Infection and Rash Patrick Njoku, MD; Temeka Tate, MD; Sousan Zadeh, MD; Erin Hauck, MD; Anila Chaudhry, MD; Betty Lo-Blais, MD; Lee
CLINICAL CASE OF THE MONTH A Middle-Aged Man with Newly Diagnosed HIV Infection and Rash Patrick Njoku, MD; Temeka Tate, MD; Sousan Zadeh, MD; Erin Hauck, MD; Anila Chaudhry, MD; Betty Lo-Blais, MD; Lee
Multiple Drug Allergies
 Management of Patients with Multiple Drug Allergies Roland Solensky, MD, FAAAAI The Corvallis Clinic Corvallis, OR roland.solensky@corvallisclinic.com Conflict of Interest Financial: None Research: Merck,
Management of Patients with Multiple Drug Allergies Roland Solensky, MD, FAAAAI The Corvallis Clinic Corvallis, OR roland.solensky@corvallisclinic.com Conflict of Interest Financial: None Research: Merck,
DOSING GUIDE. Indications. Important Safety Information. Enable the immune system. RECOGNIZE. RESPOND.
 DOSING GUIDE For patients with unresectable Stage III NSCLC following concurrent CRT For patients with locally advanced or metastatic UC previously treated with platinum-based therapy Enable the immune
DOSING GUIDE For patients with unresectable Stage III NSCLC following concurrent CRT For patients with locally advanced or metastatic UC previously treated with platinum-based therapy Enable the immune
PedsCases Podcast Scripts
 PedsCases Podcast Scripts This is a text version of a podcast from Pedscases.com on Drug Allergy. These podcasts are designed to give medical students an overview of key topics in pediatrics. The audio
PedsCases Podcast Scripts This is a text version of a podcast from Pedscases.com on Drug Allergy. These podcasts are designed to give medical students an overview of key topics in pediatrics. The audio
An unpredictable, dose-independent adverse drug reaction which is immunologically or IgEmediated.
 R H E U M A T I S M D I S O R D E R S A N D A L L E R G I E S APPROACH TO DRUG ALLERGY Dr Bernard Thong DEFINITION OF DRUG ALLERGY An unpredictable, dose-independent adverse drug reaction which is immunologically
R H E U M A T I S M D I S O R D E R S A N D A L L E R G I E S APPROACH TO DRUG ALLERGY Dr Bernard Thong DEFINITION OF DRUG ALLERGY An unpredictable, dose-independent adverse drug reaction which is immunologically
PHM142 Autoimmune Disorders + Idiosyncratic Drug Reactions
 PHM142 Autoimmune Disorders + Idiosyncratic Drug Reactions 1 Autoimmune Disorders Auto-reactivity: low physiological levels (e.g. tolerance) vs. pathogenic levels 80+ types of autoimmune diseases affect
PHM142 Autoimmune Disorders + Idiosyncratic Drug Reactions 1 Autoimmune Disorders Auto-reactivity: low physiological levels (e.g. tolerance) vs. pathogenic levels 80+ types of autoimmune diseases affect
Non-Beta-lactam Antibiotic: Testing and Desensitization
 Non-Beta-lactam Antibiotic: Testing and Desensitization David A. Khan, MD Professor of Medicine Allergy & Immunology Program Director Division of Allergy & Immunology 1 Disclosures n Research Grants n
Non-Beta-lactam Antibiotic: Testing and Desensitization David A. Khan, MD Professor of Medicine Allergy & Immunology Program Director Division of Allergy & Immunology 1 Disclosures n Research Grants n
Managing Penicillin Allergy
 Managing Penicillin Allergy Brian T. Kelly, MD MA April 12, 2019 Objectives Review penicillin allergy prevalence, morbidity, and management Describe the penicillin testing and oral challenge process Provide
Managing Penicillin Allergy Brian T. Kelly, MD MA April 12, 2019 Objectives Review penicillin allergy prevalence, morbidity, and management Describe the penicillin testing and oral challenge process Provide
DOSING AND INFORMATION GUIDE LEAPS AHEAD
 DOSING AND INFORMATION GUIDE In patients with WT RAS* mcrc 1 VECTIBIX (panitumumab) LEAPS AHEAD 5.6-month increase in median OS with FOLFOX vs FOLFOX alone 1 Spot the difference. CHOOSE VECTIBIX PRIME
DOSING AND INFORMATION GUIDE In patients with WT RAS* mcrc 1 VECTIBIX (panitumumab) LEAPS AHEAD 5.6-month increase in median OS with FOLFOX vs FOLFOX alone 1 Spot the difference. CHOOSE VECTIBIX PRIME
R-BAC-500 (Rituximab, Bendamustine, Cytarabine) for Mantle Cell Lymphoma
 R-BAC-500 (Rituximab, Bendamustine, Cytarabine) for Mantle Cell Lymphoma Not routinely commissioned, each case requires prior documented approval before offering & commencing therapy from NHS England Cancer
R-BAC-500 (Rituximab, Bendamustine, Cytarabine) for Mantle Cell Lymphoma Not routinely commissioned, each case requires prior documented approval before offering & commencing therapy from NHS England Cancer
Department of Dermatology, Christian Medical College and Hospital, Ludhiana, Punjab, India.
 Bullous pemphigoid mimicking granulomatous inflammation Abhilasha Williams, Emy Abi Thomas. Department of Dermatology, Christian Medical College and Hospital, Ludhiana, Punjab, India. Egyptian Dermatology
Bullous pemphigoid mimicking granulomatous inflammation Abhilasha Williams, Emy Abi Thomas. Department of Dermatology, Christian Medical College and Hospital, Ludhiana, Punjab, India. Egyptian Dermatology
Risk Factors and Management of Mood Stabilizer-associated Stevens-Johnson Syndrome/Toxic Epidermal Necrolysis: A Mini-review
 140 Taiwanese Journal of Psychiatry (Taipei) Vol. 25 No. 3 2011 Overview Risk Factors and Management of Mood Stabilizer-associated Stevens-Johnson Syndrome/Toxic Epidermal Necrolysis: A Mini-review Gen-Tang
140 Taiwanese Journal of Psychiatry (Taipei) Vol. 25 No. 3 2011 Overview Risk Factors and Management of Mood Stabilizer-associated Stevens-Johnson Syndrome/Toxic Epidermal Necrolysis: A Mini-review Gen-Tang
Endocarditis. By : Mehrnoush. dianatkhah
 Endocarditis By : Mehrnoush. dianatkhah Case 5.31, 31 years old woman CC : Fever, dyspnea, 3 days postpartum PMH : Mitral prolapse Fever 38.5 WBC : 8900 ESR : 84 CRP : 10.4 Cr : 0.6 NT Pro BNP: 5469 Physical
Endocarditis By : Mehrnoush. dianatkhah Case 5.31, 31 years old woman CC : Fever, dyspnea, 3 days postpartum PMH : Mitral prolapse Fever 38.5 WBC : 8900 ESR : 84 CRP : 10.4 Cr : 0.6 NT Pro BNP: 5469 Physical
EMEA PUBLIC STATEMENT ON LEFLUNOMIDE (ARAVA) - PANCYTOPENIA AND SERIOUS SKIN REACTIONS
 The European Agency for the Evaluation of Medicinal Products Evaluation of Medicines for Human Use London, 25 October 1999 EMEA/31637/99 EMEA PUBLIC STATEMENT ON LEFLUNOMIDE (ARAVA) - PANCYTOPENIA AND
The European Agency for the Evaluation of Medicinal Products Evaluation of Medicines for Human Use London, 25 October 1999 EMEA/31637/99 EMEA PUBLIC STATEMENT ON LEFLUNOMIDE (ARAVA) - PANCYTOPENIA AND
Package leaflet: Information for the user. Piperacillin/Tazobactam Actavis 4 g / 0.5 g powder for solution for infusion. piperacillin / tazobactam
 Package leaflet: Information for the user Piperacillin/Tazobactam Actavis 2 g / 0.25 g powder for solution for infusion Piperacillin/Tazobactam Actavis 4 g / 0.5 g powder for solution for infusion piperacillin
Package leaflet: Information for the user Piperacillin/Tazobactam Actavis 2 g / 0.25 g powder for solution for infusion Piperacillin/Tazobactam Actavis 4 g / 0.5 g powder for solution for infusion piperacillin
Definitions of Current SHOT Categories & What to Report
 Definitions of Current SHOT Categories & What to Report Revised March 2011 1 ADVERSE EVENTS TERM DEFINITION WHAT TO REPORT IBCT - Wrong Blood Transfused (Incorrect Blood Component Transfused) Where a patient
Definitions of Current SHOT Categories & What to Report Revised March 2011 1 ADVERSE EVENTS TERM DEFINITION WHAT TO REPORT IBCT - Wrong Blood Transfused (Incorrect Blood Component Transfused) Where a patient
פורמט עלון זה נקבע ע"י משרד הבריאות ותוכנו נבדק ואושר SUMMARY OF PRODUCT CHARACTERISTICS
 פורמט עלון זה נקבע ע"י משרד הבריאות ותוכנו נבדק ואושר SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT FELDENE GEL 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each gram contains 5mg
פורמט עלון זה נקבע ע"י משרד הבריאות ותוכנו נבדק ואושר SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT FELDENE GEL 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each gram contains 5mg
STEVENS-JOHNSON SYNDROME: A CASE REPORT
 STEVENS-JOHNSON SYNDROME: A CASE REPORT Castana O., Rempelos G., Anagiotos G., Apostolopoulou C., Dimitrouli A., Alexakis D. Department of Plastic and Reconstructive Surgery, Evangelismos General Hospital,
STEVENS-JOHNSON SYNDROME: A CASE REPORT Castana O., Rempelos G., Anagiotos G., Apostolopoulou C., Dimitrouli A., Alexakis D. Department of Plastic and Reconstructive Surgery, Evangelismos General Hospital,
Pathophysiology of autoimmune and allergic diseases. Hypersensitivity reactions type II type IV
 Pathophysiology of autoimmune and allergic diseases Hypersensitivity reactions type II type IV Audrey NOSBAUM, Florence HACARD, Marc VOCANSON, Frédéric BERARD, Jean François NICOLAS Allergologie et Immunologie
Pathophysiology of autoimmune and allergic diseases Hypersensitivity reactions type II type IV Audrey NOSBAUM, Florence HACARD, Marc VOCANSON, Frédéric BERARD, Jean François NICOLAS Allergologie et Immunologie
Scientific conclusions
 Annex II Scientific conclusions and grounds for variation to the terms of the marketing authorisations or suspension of the marketing authorisations, as applicable, taking into consideration the approved
Annex II Scientific conclusions and grounds for variation to the terms of the marketing authorisations or suspension of the marketing authorisations, as applicable, taking into consideration the approved
A Retrospective Study of Spectrum of Nevirapine Induced Cutaneous Drug Reactions in HIV Positive Patients
 Journal of US-China Medical Science 12 (2015) 85-89 doi: 10.17265/1548-6648/2015.02.008 D DAVID PUBLISHING A Retrospective Study of Spectrum of Nevirapine Induced Cutaneous Drug Reactions in HIV Positive
Journal of US-China Medical Science 12 (2015) 85-89 doi: 10.17265/1548-6648/2015.02.008 D DAVID PUBLISHING A Retrospective Study of Spectrum of Nevirapine Induced Cutaneous Drug Reactions in HIV Positive
Elements for a Public Summary
 VI.2 VI.2.1 Elements for a Public Summary Overview of Disease Epidemiology Non-Small Cell Lung Cancer (NSCLC): Non-small cell lung cancer accounts for 85 90% of lung cancers [Reck, 2014] and is the leading
VI.2 VI.2.1 Elements for a Public Summary Overview of Disease Epidemiology Non-Small Cell Lung Cancer (NSCLC): Non-small cell lung cancer accounts for 85 90% of lung cancers [Reck, 2014] and is the leading
Diagnostic evaluation of suspected Drug-Induced Liver Injury
 Diagnostic evaluation of suspected Drug-Induced Liver Injury Ynto de Boer, MD Department of Gastroenterology and Hepatology Disclosure No disclosures Drug-induced liver injury? Outline What is DILI? What,
Diagnostic evaluation of suspected Drug-Induced Liver Injury Ynto de Boer, MD Department of Gastroenterology and Hepatology Disclosure No disclosures Drug-induced liver injury? Outline What is DILI? What,
Warfarin-induced toxic epidermal necrolysis in combination therapy of Henoch- Schönlein purpura nephritis: a case report
 Kasahara et al. BMC Nephrology (2017) 18:237 DOI 10.1186/s12882-017-0648-9 CASE REPORT Open Access Warfarin-induced toxic epidermal necrolysis in combination therapy of Henoch- Schönlein purpura nephritis:
Kasahara et al. BMC Nephrology (2017) 18:237 DOI 10.1186/s12882-017-0648-9 CASE REPORT Open Access Warfarin-induced toxic epidermal necrolysis in combination therapy of Henoch- Schönlein purpura nephritis:
SUMMARY OF PRODUCT CHARACTERISTICS
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Flavamed Hustensaft 15 mg/5 ml oral solution 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 1 ml of oral solution contains 3 mg ambroxol
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Flavamed Hustensaft 15 mg/5 ml oral solution 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 1 ml of oral solution contains 3 mg ambroxol
Overview. Barriers help animals defend against many dangerous pathogens they encounter.
 Immunity Overview Barriers help animals defend against many dangerous pathogens they encounter. The immune system recognizes foreign bodies and responds with the production of immune cells and proteins.
Immunity Overview Barriers help animals defend against many dangerous pathogens they encounter. The immune system recognizes foreign bodies and responds with the production of immune cells and proteins.
Future of Pediatrics: Blisters, Hives and Other Tales from the Emergency Room June 14 th, 2016
 A. Yasmine Kirkorian MD Assistant Professor of Dermatology & Pediatrics Children s National Health System George Washington University School of Medicine & Health Sciences Future of Pediatrics: Blisters,
A. Yasmine Kirkorian MD Assistant Professor of Dermatology & Pediatrics Children s National Health System George Washington University School of Medicine & Health Sciences Future of Pediatrics: Blisters,
MabThera. SC. The wait is over. MabThera delivered in just 5 minutes. SC= subcutaneous injection
 MabThera SC. The wait is over. MabThera delivered in just 5 minutes Abbreviated Prescribing Information MabThera 1400 mg solution for subcutaneous (SC) injection (Rituximab) Indications: Indicated in adults
MabThera SC. The wait is over. MabThera delivered in just 5 minutes Abbreviated Prescribing Information MabThera 1400 mg solution for subcutaneous (SC) injection (Rituximab) Indications: Indicated in adults
Objectives 8/30/2012. How Do I Deal with a Person s Multiple (and Single) Drug Allergies? Adverse Drug Reactions
 How Do I Deal with a Person s Multiple (and Single) Drug Allergies? Faoud Ishmael, MD, PhD Assistant Professor of Medicine Section of Allergy and Immunology Penn State College of Medicine I have no conflicts
How Do I Deal with a Person s Multiple (and Single) Drug Allergies? Faoud Ishmael, MD, PhD Assistant Professor of Medicine Section of Allergy and Immunology Penn State College of Medicine I have no conflicts
Diagnosis and management of suspected drug allergies
 Diagnosis and management of suspected drug allergies SPL Sophie Farooque MRCP Allergic reactions can be caused by commonly prescribed drugs and can lead to fatal anaphylaxis. Here, the author describes
Diagnosis and management of suspected drug allergies SPL Sophie Farooque MRCP Allergic reactions can be caused by commonly prescribed drugs and can lead to fatal anaphylaxis. Here, the author describes
Presumed anaphylaxis to Hydrochlorothiazide in a 67 year old female with known sulphonamide allergy. By: Cody Clovechok
 Presumed anaphylaxis to Hydrochlorothiazide in a 67 year old female with known sulphonamide allergy. By: Cody Clovechok Home for the Summer Program - June and July, 2017 Lac du Bonnet, Manitoba Supervisor:
Presumed anaphylaxis to Hydrochlorothiazide in a 67 year old female with known sulphonamide allergy. By: Cody Clovechok Home for the Summer Program - June and July, 2017 Lac du Bonnet, Manitoba Supervisor:
OPTIMAL MANAGEMENT OF IMMUNE- RELATED ADVERSE EVENTS ASSOCIATED WITH CHECKPOINT INHIBITORS
 OPTIMAL MANAGEMENT OF IMMUNE- RELATED ADVERSE EVENTS ASSOCIATED WITH CHECKPOINT INHIBITORS Alberto Fusi Charité Comprehensive Cancer Centre Berlin, Germany 1 Immune check point blockade with CTLA-4, anti-pd-1
OPTIMAL MANAGEMENT OF IMMUNE- RELATED ADVERSE EVENTS ASSOCIATED WITH CHECKPOINT INHIBITORS Alberto Fusi Charité Comprehensive Cancer Centre Berlin, Germany 1 Immune check point blockade with CTLA-4, anti-pd-1
Cases of Adverse Drug Events in Pediatrics: Prospective Study at a Hospital in Rabat (Morocco)
 J Young Pharm, 2018; 10(3): 362-366 A multifaceted peer reviewed journal in the field of Pharmacy www.jyoungpharm.org www.phcog.net Case Report Cases of Adverse Drug Events in Pediatrics: Prospective Study
J Young Pharm, 2018; 10(3): 362-366 A multifaceted peer reviewed journal in the field of Pharmacy www.jyoungpharm.org www.phcog.net Case Report Cases of Adverse Drug Events in Pediatrics: Prospective Study
Pediatric Dermatology
 Pediatric Dermatology --------- Emergencies & Urgencies Nicholas V. Nguyen, M.D. Director, Pediatric Dermatology Disclosures In the past 12 months, I have had the following financial relationships with
Pediatric Dermatology --------- Emergencies & Urgencies Nicholas V. Nguyen, M.D. Director, Pediatric Dermatology Disclosures In the past 12 months, I have had the following financial relationships with
Package leaflet: Information for the patient. / / 30 mg/5 ml syrup. Ambroxol hydrochloride
 Package leaflet: Information for the patient / / 30 mg/5 ml syrup Ambroxol hydrochloride Read all of this leaflet carefully before you start taking this medicine because it contains important information
Package leaflet: Information for the patient / / 30 mg/5 ml syrup Ambroxol hydrochloride Read all of this leaflet carefully before you start taking this medicine because it contains important information
CARBAMAZEPINE INDUCED STEVENS JOHNSON SYNDROME- A CASE STUDY
 WORLD JOURNAL OF PHARMACY AND PHARMACEUTICAL SCIENCES Ragesh SJIF Impact Factor 2.786 Volume 3, Issue 6, 1599-1604. Case Study ISSN 2278 4357 CARBAMAZEPINE INDUCED STEVENS JOHNSON SYNDROME- A CASE STUDY
WORLD JOURNAL OF PHARMACY AND PHARMACEUTICAL SCIENCES Ragesh SJIF Impact Factor 2.786 Volume 3, Issue 6, 1599-1604. Case Study ISSN 2278 4357 CARBAMAZEPINE INDUCED STEVENS JOHNSON SYNDROME- A CASE STUDY
ESVD Conference Budapest Lucky s history. Lucky s history. Lucky s history. Lucky s history. MVDr. Lucia Panakova, DipECVD
 Lucky, Posavacki gonic, ca. 2y, castrated male ESVD Conference Budapest 2016 MVDr. Lucia Panakova, DipECVD Veterinärmedizinische Universität Wien 2 Lucky s history Lucky s history Referred to the dermatology
Lucky, Posavacki gonic, ca. 2y, castrated male ESVD Conference Budapest 2016 MVDr. Lucia Panakova, DipECVD Veterinärmedizinische Universität Wien 2 Lucky s history Lucky s history Referred to the dermatology
, version 1.1 PUBLIC SUMMARY OF THE RISK MANAGEMENT PLAN
 Docetaxel Orion 20 mg/1 ml concentrate for solution for infusion Docetaxel Orion 80 mg/4 ml concentrate for solution for infusion Docetaxel Orion 160 mg/8 ml concentrate for solution for infusion 11.8.2014,
Docetaxel Orion 20 mg/1 ml concentrate for solution for infusion Docetaxel Orion 80 mg/4 ml concentrate for solution for infusion Docetaxel Orion 160 mg/8 ml concentrate for solution for infusion 11.8.2014,
Local Natalizumab Treatment Protocol
 Local Natalizumab Treatment Protocol 1. New medicine name: Natalizumab 300mg concentrate for solution for infusion (Natalizumab ) 2. Licensed indication(s): Natalizumab is indicated for single disease
Local Natalizumab Treatment Protocol 1. New medicine name: Natalizumab 300mg concentrate for solution for infusion (Natalizumab ) 2. Licensed indication(s): Natalizumab is indicated for single disease
PRAC recommendations on signals
 12 October 2017 EMA/PRAC/610975/2017 Pharmacovigilance Risk Assessment Committee (PRAC) Adopted at the 25-29 September 2017 PRAC meeting This document provides an overview of the recommendations adopted
12 October 2017 EMA/PRAC/610975/2017 Pharmacovigilance Risk Assessment Committee (PRAC) Adopted at the 25-29 September 2017 PRAC meeting This document provides an overview of the recommendations adopted
Prevention of severe cutaneous adverse drug reactions: the emerging value of pharmacogenetic screening
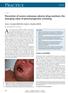 CMAJ Cases Prevention of severe cutaneous adverse drug reactions: the emerging value of pharmacogenetic screening Suran L. Fernando MB BS PhD, Andrew J. Broadfoot MB BS Previously published at www.cmaj.ca
CMAJ Cases Prevention of severe cutaneous adverse drug reactions: the emerging value of pharmacogenetic screening Suran L. Fernando MB BS PhD, Andrew J. Broadfoot MB BS Previously published at www.cmaj.ca
NEW ZEALAND DATA SHEET
 1 PRODUCT NAME BISOLVON CHESTY FORTE Bromhexine hydrochloride tablets Bromhexine hydrochloride Oral Solution 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each tablet of Bisolvon Chesty Forte contains bromhexine
1 PRODUCT NAME BISOLVON CHESTY FORTE Bromhexine hydrochloride tablets Bromhexine hydrochloride Oral Solution 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each tablet of Bisolvon Chesty Forte contains bromhexine
Stevens Johnson Syndrome: How Diagnosis Impacts Disease Course
 Southern Adventist Univeristy KnowledgeExchange@Southern Graduate Research Projects Nursing 12-4-2015 Stevens Johnson Syndrome: How Diagnosis Impacts Disease Course Sharon K. Hart Southern Adventist University,
Southern Adventist Univeristy KnowledgeExchange@Southern Graduate Research Projects Nursing 12-4-2015 Stevens Johnson Syndrome: How Diagnosis Impacts Disease Course Sharon K. Hart Southern Adventist University,
Diagnosis, classification, and management of erythema multiforme and Stevens Johnson syndrome
 Arch Dis Child 2000;83:347 352 347 Unité de Dermatologie Pédiatrique, Hôpital Pellegrin-Enfants, Place Amélie Raba-Leon, 33 076 Bordeaux Cedex, France C Léauté-Labrèze J Maleville A Taïeb Service de Pédiatrie,
Arch Dis Child 2000;83:347 352 347 Unité de Dermatologie Pédiatrique, Hôpital Pellegrin-Enfants, Place Amélie Raba-Leon, 33 076 Bordeaux Cedex, France C Léauté-Labrèze J Maleville A Taïeb Service de Pédiatrie,
Syndrome de Lyell Approche diagnostique. seminaires iris. Veronique del Marmol Alexandre Chamoun Service de Dermatologie Hôpital Erasme.
 Syndrome de Lyell Approche diagnostique Veronique del Marmol Alexandre Chamoun Service de Dermatologie Hôpital Erasme Serge Jennes Hôpital Militaire Rash benign Pustulose exanthematique Aigue et généralisée
Syndrome de Lyell Approche diagnostique Veronique del Marmol Alexandre Chamoun Service de Dermatologie Hôpital Erasme Serge Jennes Hôpital Militaire Rash benign Pustulose exanthematique Aigue et généralisée
OXCARBAZEPINE-INDUCED STEVENS-JOHNSON SYNDROME: A CASE REPORT
 OXCARBAZEPINE-INDUCED STEVENS-JOHNSON SYNDROME: A CASE REPORT Lung-Chang Lin, 1,2 Ping-Chin Lai, 3 Sheau-Fang Yang, 4 and Rei-Cheng Yang 1,5 Departments of 1 Pediatrics and 4 Pathology, Kaohsiung Medical
OXCARBAZEPINE-INDUCED STEVENS-JOHNSON SYNDROME: A CASE REPORT Lung-Chang Lin, 1,2 Ping-Chin Lai, 3 Sheau-Fang Yang, 4 and Rei-Cheng Yang 1,5 Departments of 1 Pediatrics and 4 Pathology, Kaohsiung Medical
Vancomycin. A bactericidal drug which acts by inhibiting cell wall synthesis. Active only against gram-positive bacteria, particularly staphylococci.
 Vancomycin A bactericidal drug which acts by inhibiting cell wall synthesis. Active only against gram-positive bacteria, particularly staphylococci. Used IV in treating endocarditis caused by methicillin-resistant
Vancomycin A bactericidal drug which acts by inhibiting cell wall synthesis. Active only against gram-positive bacteria, particularly staphylococci. Used IV in treating endocarditis caused by methicillin-resistant
The Case-Population Study Design
 ORIGINAL RESEARCH ARTICLE Drug Saf 2011; 34 (10): 861-868 0114-5916/11/0010-0861/$49.95/0 ª 2011 Adis Data Information BV. All rights reserved. The Case-Population Study Design An Analysis of its Application
ORIGINAL RESEARCH ARTICLE Drug Saf 2011; 34 (10): 861-868 0114-5916/11/0010-0861/$49.95/0 ª 2011 Adis Data Information BV. All rights reserved. The Case-Population Study Design An Analysis of its Application
Respiratory tract infections. Krzysztof Buczkowski
 Respiratory tract infections Krzysztof Buczkowski Etiology Viruses Rhinoviruses Adenoviruses Coronaviruses Influenza and Parainfluenza Viruses Respiratory Syncitial Viruses Enteroviruses Etiology Bacteria
Respiratory tract infections Krzysztof Buczkowski Etiology Viruses Rhinoviruses Adenoviruses Coronaviruses Influenza and Parainfluenza Viruses Respiratory Syncitial Viruses Enteroviruses Etiology Bacteria
Clinical Case Scenario. HIVeEducation Workshop, Sint Maarten 2009
 Clinical Case Scenario HIVeEducation Workshop, Sint Maarten 2009 Background Mrs. S is a 34 year-old woman who was referred from the VCT center after testing HIV positive three weeks ago. Her husband recently
Clinical Case Scenario HIVeEducation Workshop, Sint Maarten 2009 Background Mrs. S is a 34 year-old woman who was referred from the VCT center after testing HIV positive three weeks ago. Her husband recently
Disruptions in the Immune
 Disruptions in the Immune System Bởi: OpenStaxCollege A functioning immune system is essential for survival, but even the sophisticated cellular and molecular defenses of the mammalian immune response
Disruptions in the Immune System Bởi: OpenStaxCollege A functioning immune system is essential for survival, but even the sophisticated cellular and molecular defenses of the mammalian immune response
RiTUXimab 375 mg/m 2 Therapy-7 day
 RiTUXimab 375 mg/m 2 Therapy-7 day This regimen supercedes NCCP Regimen 00208 rituximab 375mg/m2 therapy-follicular lymphoma as of February 2019 INDICATIONS FOR USE: Regimen *Reimbursement INDICATION ICD10
RiTUXimab 375 mg/m 2 Therapy-7 day This regimen supercedes NCCP Regimen 00208 rituximab 375mg/m2 therapy-follicular lymphoma as of February 2019 INDICATIONS FOR USE: Regimen *Reimbursement INDICATION ICD10
nevirapine, zidovudine and lamivudine
 nevirapine, zidovudine and lamivudine a fixed dose ART combination for HIV treatment http://www.aidsmap.com/hatip nevirapine. zidovudine and lamivudine (non-fixed dose) 2 Information on starting treatment
nevirapine, zidovudine and lamivudine a fixed dose ART combination for HIV treatment http://www.aidsmap.com/hatip nevirapine. zidovudine and lamivudine (non-fixed dose) 2 Information on starting treatment
Drug induced allergy and hypersensitivity
 Drug induced allergy and hypersensitivity Yunita Sari Pane, Aznan Lelo Dept. Pharmacology & Therapeutic School of Medicine Universitas Sumatera Utara 13 Mei 2009, KBK-FK USU, Medan Drug Allergy Adverse
Drug induced allergy and hypersensitivity Yunita Sari Pane, Aznan Lelo Dept. Pharmacology & Therapeutic School of Medicine Universitas Sumatera Utara 13 Mei 2009, KBK-FK USU, Medan Drug Allergy Adverse
Cutaneous Drug Reactions
 Cutaneous Drug Reactions Andrei Metelitsa, MD, FRCPC, FAAD Co-Director, Institute for Skin Advancement Clinical Associate Professor, Dermatology University of Calgary, Canada Copyright 2017 by Sea Courses
Cutaneous Drug Reactions Andrei Metelitsa, MD, FRCPC, FAAD Co-Director, Institute for Skin Advancement Clinical Associate Professor, Dermatology University of Calgary, Canada Copyright 2017 by Sea Courses
A Case Report on Paracetamol Induced Generalized Fixed Drug Eruptions
 Human Journals Case report November 2016 Vol.:7, Issue:4 All rights are reserved by Dr. C Sugunakar Raju et al. A Case Report on Paracetamol Induced Generalized Fixed Drug Eruptions Keywords: Skin Eruptions,
Human Journals Case report November 2016 Vol.:7, Issue:4 All rights are reserved by Dr. C Sugunakar Raju et al. A Case Report on Paracetamol Induced Generalized Fixed Drug Eruptions Keywords: Skin Eruptions,
Emergency Dermatology. Emergency Dermatology
 Emergency Dermatology These are rapidly progressive skin conditions and some are potentially lifethreatening. Early recognition is important to implement prompt supportive care and therapy. Some are drug
Emergency Dermatology These are rapidly progressive skin conditions and some are potentially lifethreatening. Early recognition is important to implement prompt supportive care and therapy. Some are drug
Setting The setting was secondary care. The economic study was carried out in the USA.
 Cost-effectiveness of IV-to-oral switch therapy: azithromycin vs cefuroxime with or without erythromycin for the treatment of community-acquired pneumonia Paladino J A, Gudgel L D, Forrest A, Niederman
Cost-effectiveness of IV-to-oral switch therapy: azithromycin vs cefuroxime with or without erythromycin for the treatment of community-acquired pneumonia Paladino J A, Gudgel L D, Forrest A, Niederman
Breast Pathway Group EC x 4 Docetaxel x 4: Epirubicin & Cyclophosphamide followed by Docetaxel in Early Breast Cancer
 Breast Pathway Group EC x 4 Docetaxel x 4: Epirubicin & Cyclophosphamide followed by Docetaxel in Early Breast Indication: Neoadjuvant therapy for high risk and fit breast cancer patients suitable for
Breast Pathway Group EC x 4 Docetaxel x 4: Epirubicin & Cyclophosphamide followed by Docetaxel in Early Breast Indication: Neoadjuvant therapy for high risk and fit breast cancer patients suitable for
Annex II. Scientific conclusions and grounds for the maintenance of the marketing authorisations presented by the EMA
 Annex II Scientific conclusions and grounds for the maintenance of the marketing authorisations presented by the EMA 22 Scientific conclusions Overall summary of the scientific evaluation of pholcodine-containing
Annex II Scientific conclusions and grounds for the maintenance of the marketing authorisations presented by the EMA 22 Scientific conclusions Overall summary of the scientific evaluation of pholcodine-containing
Summary of Product Characteristics
 1 NAME OF THE MEDICINAL PRODUCT Feldene 5mg/g Gel Summary of Product Characteristics 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each gram contains 5 mg piroxicam (0.5% w/w). Excipient with known effect:
1 NAME OF THE MEDICINAL PRODUCT Feldene 5mg/g Gel Summary of Product Characteristics 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each gram contains 5 mg piroxicam (0.5% w/w). Excipient with known effect:
Oral Acetazolamide after Boston Keratoprosthesis in Stevens Johnson Syndrome
 Oral Acetazolamide after Boston Keratoprosthesis in Stevens Johnson Syndrome The Harvard community has made this article openly available. Please share how this access benefits you. Your story matters
Oral Acetazolamide after Boston Keratoprosthesis in Stevens Johnson Syndrome The Harvard community has made this article openly available. Please share how this access benefits you. Your story matters
AOU Ospedali Riuniti - Ancona
 AOU Ospedali Riuniti - Ancona Ospedale Materno-Infantile di Alta Specializzazione G. Salesi UOC Pediatria Allergia a farmaci e infezioni: tra coesistenza e casualità fabrizio franceschini Drug Hypersensitivity
AOU Ospedali Riuniti - Ancona Ospedale Materno-Infantile di Alta Specializzazione G. Salesi UOC Pediatria Allergia a farmaci e infezioni: tra coesistenza e casualità fabrizio franceschini Drug Hypersensitivity
Lecture 03: Drug allergy
 Lecture 03: Drug allergy 1. Basic Information Author: Netherlands Pharmacovigilance Centre Lareb Version date: 17 Nov 2017 Content: This hand-out describes Drug allergy and provides background information
Lecture 03: Drug allergy 1. Basic Information Author: Netherlands Pharmacovigilance Centre Lareb Version date: 17 Nov 2017 Content: This hand-out describes Drug allergy and provides background information
Transfusion Reactions. Directed by M-azad March 2012
 Transfusion Reactions Directed by M-azad March 2012 Transfusion Reactions are Adverse reactions associated with the transfusion of blood and its components Transfusion reactions Non-threatening to fatal
Transfusion Reactions Directed by M-azad March 2012 Transfusion Reactions are Adverse reactions associated with the transfusion of blood and its components Transfusion reactions Non-threatening to fatal
A. Erythema multiforme and related diseases
 Go Back to the Top To Order, Visit the Purchasing Page for Details Chapter Erythema, Erythroderma (Exfoliative Dermatitis) Erythema is caused by telangiectasia or hyperemia in the papillary and reticular
Go Back to the Top To Order, Visit the Purchasing Page for Details Chapter Erythema, Erythroderma (Exfoliative Dermatitis) Erythema is caused by telangiectasia or hyperemia in the papillary and reticular
SEVERE CUTANEOUS ADVERSE DRUG REACTIONS: STEVENS-JOHNSON SYNDROME AND TOXIC EPIDERMAL NECROLYSISA, A REPORT OF 4 CASES SEEN AT UMMC
 SEVERE CUTANEOUS ADVERSE DRUG REACTIONS: STEVENS-JOHNSON SYNDROME AND TOXIC EPIDERMAL NECROLYSISA, A REPORT OF 4 CASES SEEN AT UMMC Shasha Khairullah, Rokiah Che Ismail Department of Medicine, Faculty
SEVERE CUTANEOUS ADVERSE DRUG REACTIONS: STEVENS-JOHNSON SYNDROME AND TOXIC EPIDERMAL NECROLYSISA, A REPORT OF 4 CASES SEEN AT UMMC Shasha Khairullah, Rokiah Che Ismail Department of Medicine, Faculty
PRAC recommendations on signals
 25 November 2013 EMA/PRAC/693228/2013 Pharmacovigilance Risk Assessment Committee Adopted at the PRAC meeting of 4-7 November 2013 This document provides an overview of the recommendations adopted by the
25 November 2013 EMA/PRAC/693228/2013 Pharmacovigilance Risk Assessment Committee Adopted at the PRAC meeting of 4-7 November 2013 This document provides an overview of the recommendations adopted by the
Beta-Lactam Use in Penicillin Allergic Patients Clinical Guideline
 RECOMMENDATIONS: Beta-Lactam Use in Penicillin Allergic Patients Clinical Guideline 1. Penicillin Anaphylaxis (Type-1 IgE Mediated hypersensitivity) a. May use Cephalosporins and Carbapenems 2. Penicillin
RECOMMENDATIONS: Beta-Lactam Use in Penicillin Allergic Patients Clinical Guideline 1. Penicillin Anaphylaxis (Type-1 IgE Mediated hypersensitivity) a. May use Cephalosporins and Carbapenems 2. Penicillin
Toxic Epidermal Necrolysis and SJS : Case Reports & Brief Review
 Case Report Toxic Epidermal Necrolysis and SJS : Case Reports & Brief Review Deepali P. Mohite*, Satyajitraje Tekade*, Amol Gadbail*, M. S. Chaudhary** Abstract : Toxic epidermal necrolysis (TEN) and Stevens
Case Report Toxic Epidermal Necrolysis and SJS : Case Reports & Brief Review Deepali P. Mohite*, Satyajitraje Tekade*, Amol Gadbail*, M. S. Chaudhary** Abstract : Toxic epidermal necrolysis (TEN) and Stevens
Blood and Immune system Acquired Immunity
 Blood and Immune system Acquired Immunity Immunity Acquired (Adaptive) Immunity Defensive mechanisms include : 1) Innate immunity (Natural or Non specific) 2) Acquired immunity (Adaptive or Specific) Cell-mediated
Blood and Immune system Acquired Immunity Immunity Acquired (Adaptive) Immunity Defensive mechanisms include : 1) Innate immunity (Natural or Non specific) 2) Acquired immunity (Adaptive or Specific) Cell-mediated
Prof Dr Najlaa Fawzi
 1 Prof Dr Najlaa Fawzi is an acute highly infectious disease, characterized by vesicular rash, mild fever and mild constitutional symptoms. is a local manifestation of reactivation of latent varicella
1 Prof Dr Najlaa Fawzi is an acute highly infectious disease, characterized by vesicular rash, mild fever and mild constitutional symptoms. is a local manifestation of reactivation of latent varicella
New product information wording Extracts from PRAC recommendations on signals
 20 July 2017 EMA/PRAC/406976/2017 Pharmacovigilance Risk Assessment Committee (PRAC) New product information wording Extracts from PRAC recommendations on signals Adopted at the 3-6 July 2017 PRAC The
20 July 2017 EMA/PRAC/406976/2017 Pharmacovigilance Risk Assessment Committee (PRAC) New product information wording Extracts from PRAC recommendations on signals Adopted at the 3-6 July 2017 PRAC The
VANLID Capsules (Vancomycin hydrochloride)
 Published on: 22 Sep 2014 VANLID Capsules (Vancomycin hydrochloride) Composition VANLID Capsules Each capsule contains: Vancomycin Hydrochloride IP equivalent to Vancomycin.. 250 mg Approved colours used
Published on: 22 Sep 2014 VANLID Capsules (Vancomycin hydrochloride) Composition VANLID Capsules Each capsule contains: Vancomycin Hydrochloride IP equivalent to Vancomycin.. 250 mg Approved colours used
Elements for a public summary
 VI.2 Elements for a public summary VI.2.1 Overview of disease epidemiology Nevirapine is used for antiretroviral combination therapy of Human Immunodeficiency Virus (HIV) infection. Human immunodeficiency
VI.2 Elements for a public summary VI.2.1 Overview of disease epidemiology Nevirapine is used for antiretroviral combination therapy of Human Immunodeficiency Virus (HIV) infection. Human immunodeficiency
DECISION SUPPORT TOOL FOR RN SANES DISPENSING PROPHYLAXIS FOR SEXUALLY TRANSMITTED INFECTION AFTER SEXUAL ASSAULT
 BC WOMEN S SEXUAL ASSAULT SERVICE PURPOSE DECISION SUPPORT TOOL FOR RN SANES DISPENSING PROPHYLAXIS FOR SEXUALLY TRANSMITTED INFECTION AFTER SEXUAL ASSAULT This decision support tool (DST) and accompanying
BC WOMEN S SEXUAL ASSAULT SERVICE PURPOSE DECISION SUPPORT TOOL FOR RN SANES DISPENSING PROPHYLAXIS FOR SEXUALLY TRANSMITTED INFECTION AFTER SEXUAL ASSAULT This decision support tool (DST) and accompanying
O-CVP with maintenance Obinutuzumab
 with maintenance Obinutuzumab INDICATION Follicular Lymphoma: 1 st line treatment in advanced symptomatic patients (NICE TA513 for FLIPI score 2 or higher - BLUETEQ required) TREATMENT INTENT Disease modification.
with maintenance Obinutuzumab INDICATION Follicular Lymphoma: 1 st line treatment in advanced symptomatic patients (NICE TA513 for FLIPI score 2 or higher - BLUETEQ required) TREATMENT INTENT Disease modification.
Skin Pathway Group Alemtuzumab in Cutaneous Lymphoma
 Skin Pathway Group Alemtuzumab in Cutaneous Lymphoma Indication: Treatment of patients with Cutaneous Lymphoma (Unlicensed use) Disease control prior to Reduced Intensity Conditioning Stem Cell Transplant
Skin Pathway Group Alemtuzumab in Cutaneous Lymphoma Indication: Treatment of patients with Cutaneous Lymphoma (Unlicensed use) Disease control prior to Reduced Intensity Conditioning Stem Cell Transplant
PRODUCT INFORMATION TARGOCID
 NAME OF THE MEDICINE Non-proprietary Name teicoplanin PRODUCT INFORMATION TARGOCID DESCRIPTION Teicoplanin is a glycopeptide-antibiotic produced by Actinoplanes teichomyceticus. It is presented as a sterile,
NAME OF THE MEDICINE Non-proprietary Name teicoplanin PRODUCT INFORMATION TARGOCID DESCRIPTION Teicoplanin is a glycopeptide-antibiotic produced by Actinoplanes teichomyceticus. It is presented as a sterile,
1-844-FAX-A360 ( )
 1-844-ASK-A360 (1-844-275-2360) 1-844-FAX-A360 (1-844-329-2360) www.myaccess360.com For more information, call AstraZeneca Access 360 at 1-844-ASK-A360, Monday through Friday, 8 am to 8 pm ET. IMFINZI
1-844-ASK-A360 (1-844-275-2360) 1-844-FAX-A360 (1-844-329-2360) www.myaccess360.com For more information, call AstraZeneca Access 360 at 1-844-ASK-A360, Monday through Friday, 8 am to 8 pm ET. IMFINZI
Chapter 38- Immune System
 Chapter 38- Immune System First Line of Defense: Barriers Nonspecific defenses, such as the skin and mucous membranes, are barriers to potential pathogens. In addition to being a physical barrier to pathogens,
Chapter 38- Immune System First Line of Defense: Barriers Nonspecific defenses, such as the skin and mucous membranes, are barriers to potential pathogens. In addition to being a physical barrier to pathogens,
RABEPRAZOL 10mg and 20mg Gastro-resistant Tablets
 PACKAGE LEAFLET: INFORMATION FOR THE USER RABEPRAZOL 10mg and 20mg Gastro-resistant Tablets RABEPRAZOLE This leaflet is a copy of the Summary of Product Characteristics and Patient Information Leaflet
PACKAGE LEAFLET: INFORMATION FOR THE USER RABEPRAZOL 10mg and 20mg Gastro-resistant Tablets RABEPRAZOLE This leaflet is a copy of the Summary of Product Characteristics and Patient Information Leaflet
