Migraine is an important clinical problem in
|
|
|
- Rudolf Clark
- 6 years ago
- Views:
Transcription
1 REVIEW ARTICLE Symptomatic Treatment of Migraine in Children: A Systematic Review of Medication Trials Léonie Damen, PhD*; Jacques K. J. Bruijn, MD ; Arianne P. Verhagen, PhD*; Marjolein Y. Berger, MD PhD*; Jan Passchier, PhD ; and Bart W Koes, PhD* ABSTRACT. Objective. Treatment of pediatric migraine includes an individually tailored regimen of both nonpharmacologic and pharmacologic measures. The mainstay of symptomatic treatment in children with migraine is intermittent oral or suppository analgesics, but there is no coherent body of evidence on symptomatic treatment of childhood migraine available. The objective of this review is to describe and assess the evidence from randomized and clinical controlled trials concerning the efficacy and tolerability of symptomatic treatment of migraine in children. Design. Systematic review according to the standards of the Cochrane Collaboration. Methods. Databases were searched from inception to June Additional reference checking was performed. Two authors independently selected randomized and controlled trials evaluating the effects of symptomatic treatment in children (<18 years old) with migraine, using headache (HA) clinical improvement as an outcome measure. Two authors assessed trial quality independently by using the Delphi list, and data were extracted from the original reports by using standardized forms. Quantitative and qualitative analysis was conducted according to type of intervention. Results. A total of 10 trials were included in this review, of which 6 studies were considered to be of high quality. The number of included participants in each trial ranged from 14 to 653, with a total of 1575 patients included in this review. Mean dropout rate was 19.8% (range: %), and the mean age of participants was years (range: 4 18 years). All studies used HA diaries to assess outcomes. In most studies, a measure of clinical improvement was calculated by using these diaries. Improvement often was regarded as being clinically relevant when the patients HA declined by >50%. Regarding oral analgesic treatment, the effectiveness of acetaminophen, ibuprofen, and nimesulide were evaluated. When compared with placebo, acetaminophen (relative risk [RR]: 1.5; 95% confidence interval [CI]: ) and ibuprofen (pooled RR: 1.5; 95% CI: ) significantly reduced HAs. We conclude that there is moderate evidence that both acetaminophen and ibuprofen are more effective in reduction of symptoms 1 and 2 From the Departments of *General Practice, Pediatric Neurology, and Medical Psychology and Psychotherapy, Erasmus Medical Centre, Rotterdam, Netherlands; and Department of Pediatrics, Vlietland Hospital, Vlaardingen, Netherlands. Accepted for publication Feb 15, doi: /peds No conflict of interest declared. Address correspondence to Arianne P. Verhagen, PhD, Department of General Practice, Erasmus Medical Centre, PO Box 1738, 3000 DR, Rotterdam, Netherlands. a.verhagen@erasmusmc.nl PEDIATRICS (ISSN ). Copyright 2005 by the American Academy of Pediatrics. hours after intake than placebo with minor adverse effects. No clear differences in effect were found between acetaminophen and ibuprofen or nimesulide. Regarding the nonanalgesic interventions, nasal-spray sumatriptan, oral sumatriptan, oral rizatriptan, oral dihydroergotamine, intravenous prochlorperazine, and ketorolac were evaluated. When compared with placebo, nasal-spray sumatriptan (pooled RR: 1.4; 95% CI: ) seemed to significantly reduce HAs. We conclude that there is moderate evidence that nasal-spray sumatriptan is more effective in reduction of symptoms than placebo but with significantly more adverse events. No differences in effect were found between oral triptans and placebo. All medications were well tolerated, but significantly more adverse events were reported for nasalspray sumatriptan compared with placebo. We also conclude that there is moderate evidence that intravenous prochlorperazine is more effective than intravenous ketorolac in the reduction of symptoms 1 hour after intake. No differences in effect were found between oral dihydroergotamine and placebo. Conclusions. Acetaminophen, ibuprofen, and nasalspray sumatriptan are all effective symptomatic pharmacologic treatments for episodes of migraine in children. The new frontier for symptomatic treatment is likely to be the development of triptan agents for use in children. Most treatments have only been evaluated in 1 or 2 studies, which limits the generalizability of the findings. We strongly recommend performing a large, highquality randomized, controlled trial evaluating different symptomatic medications compared with each other or to placebo treatment. Favorable high-quality studies should be performed and reported according to the CONSORT statement. Clinical improvement of HA should be used as the primary outcome measure, but quality of life, days missed at school, and satisfaction of child or parents should also be used as an outcome measure in future studies. Pediatrics 2005;116:e295 e302. URL: www. pediatrics.org/cgi/doi/ /peds ; migraine, children, symptomatic treatment, systematic review, clinical trials, medication. ABBREVIATIONS. RCT, randomized, controlled trial; CCT, clinical controlled trial; HA, headache; IHS, International Headache Society; RR, relative risk; CI, confidence interval; IV, intravenous. Migraine is an important clinical problem in school-aged children, with an estimated prevalence of 2.7% to 10%. 1,2 It is characterized by attacks of intense, throbbing, unilateral head pain, often accompanied by nausea, vomiting, photophobia, and phonophobia. 3,4 Treatment of pediatric migraine includes an individually tailored regimen of both nonpharmacologic PEDIATRICS Vol. 116 No. 2 August 2005 e295
2 and pharmacologic measures. Nonpharmacologic modalities include lifestyle adjustments (ie, sleep hygiene, dietary adjustment, or exercise program), reassurance, stress management, biofeedback, and other biobehavioral therapies. Pharmacologic interventions include the use of symptomatic medication such as analgesics (acetaminophen, ibuprofen), triptans (sumatriptan), and antiemetics (prochlorperazine) and the use of prophylactic medication. The mainstay of symptomatic treatment in children with migraine is intermittent oral or suppository analgesics, but there is no coherent body of evidence on symptomatic treatment of childhood migraine available. Good-quality controlled trials preferably summarized in a systematic review form the basis for evidence-based treatment guidelines, which may improve the management and treatment of individual patients. One systematic review on triptans only for pediatric migraine has been performed. 5 Based on 4 randomized, controlled trials (RCTs), they found effectiveness with nasal-spray sumatriptan compared with placebo in acute pediatric migraine, whereas oral sumatriptan and rizatriptan were not clearly beneficial. 5 To study adverse events, open-label studies were also included. 5 The objective of this review was to describe and assess the evidence from RCTs and clinical controlled trials (CCTs) concerning the efficacy and tolerability of symptomatic treatment of migraine in children. METHODS Search Strategy We searched Medline, Embase, PsychInfo, Web of Science, and Cinahl from inception to June 2004 using the terms migraine, headache, cephalgia, cephalalgia, child*, infant, teenage, adolescent, or p(a)ediatric together with the search strategy for identifying clinical trials described by Robinson and Dickerson. 6 The Cochrane Controlled Trials Register was searched using the words migraine, headache, cephalgia, cephalalgia, child*, infant, teenage, adolescent, or p(a)ediatric. Additional strategies for identifying trials included searching the reference lists of review articles and included studies. Study Selection We selected only RCTs and CCTs including symptomatic pharmacologic medicine used in the management of migraine in children ( 18 years old), with criteria designed to distinguish migraine from other types of headache (HA). The use of a specific set of diagnostic criteria (eg, International Headache Society [IHS] 3 and Ad Hoc Committee on the Classification of Headache 7 ) was not required, but migraine diagnoses had to be based on at least some of the distinctive features of migraine. 3,4 Our outcome measures of interest were: HA intensity, frequency or duration, HA index, and overall improvement. Rescue medication was defined as additional medications (other than study medication) permitted in nonresponders, usually limited to the habitual medications a person uses for their migraine HA. When a protocol permitted the use of rescue medication before outcome measurement (2 6 hours), then the latest outcome assessment not confounded by the use of rescue medication was extracted. No language restriction was applied. Two authors (L.D. and J.K.J.B.) independently screened titles and abstracts of studies identified by the literature search for eligibility. All potentially relevant studies were retrieved as full papers and then again independently reviewed by 2 authors (L.D. and J.K.J.B.). Any disagreements were resolved through consensus, when possible, or by arbitration of a third author (A.P.V.). Methodologic Quality and Data Extraction Two authors (L.D. and J.K.J.B. or A.P.V.) independently assessed the methodologic quality of the included trials using the Delphi list. 8 The Delphi list is a generic criteria list developed by international consensus and consists of (1) randomization, (2) adequate allocation concealment, (3) groups similar at baseline, (4) specification of eligibility criteria, (5) blinding of outcome assessor, (6) blinding of care provider, (7) blinding of patient, (8) presentation of point estimates and measures of variability, and (9) intention-to-treat analysis. One extra item was added because it was found to be relevant for these studies: (10) withdrawal/ dropout rate ( 20% or selective dropout) unlikely to cause bias. The methodologic criteria were scored as yes (1), no (0), or don t know (0). A quality score of each trial was computed by counting the number of positive scores. Any disagreements were resolved through consensus, when possible, or by arbitration of a third author (J.K.J.B. or A.P.V.). Data extraction was performed by 1 author (L.D.) and checked by a second author (A.P.V.). Disagreements were resolved by consensus. Extracted information included (if available) demographic data, detailed description of the intervention and control (ie, dose given, study duration), outcome measures, and information on adverse effects. Data Analysis We calculated relative risks (RRs) with 95% confidence intervals (CIs). We performed an available-case analysis (data reported), and if sufficient data were available, we also performed a worst-case analysis (ie, all dropouts are assumed to be nonresponders). Data are presented as treatment success, indicating that arr 1represents a better outcome for the first mentioned intervention group. In case of a crossover trial, we ideally would like to restrict our analysis to first-period data only or, in case of a sufficient washout period and no carryover effect, data of both periods could be combined. In this review we analyzed the crossover trials as if they were parallel-group trials, because none of them provided separate data of each treatment period or data concerning a carryover effect, which would otherwise lead to exclusion of most crossover trials. Statistical pooling was performed in the case of clinical homogeneity. In parallel studies, when 1 comparison from the same study (ie, sumatriptan 5, 10, and 20 mg versus placebo) was used for the statistical pooling of sumatriptan versus placebo, the results from the placebo group were evenly spread out over the 3 comparisons, and the number of patients in the placebo group was divided by 3 to prevent triple counting (R.J.P.M. Scholten, PhD, Dutch Cochrane Centre, verbal communication, 2004). A qualitative analysis was performed by using a rating system with levels of evidence. 9 The evidence was judged to be strong when multiple ( 2) high-quality RCTs produced generally consistent findings. Results were considered consistent if 75% of the studies reported similar results on the same outcome measure. It was judged to be moderate when 1 high-quality RCT or multiple ( 2) low-quality RCTs or CCTs produced generally consistent findings. Evidence was considered to be limited when only 1 low-quality RCT or CCT existed and conflicting if the findings of existing trials were inconsistent. No evidence was considered when no RCTs or CCTs were found or when the authors provide no sufficient data for analysis. We arbitrarily regarded trials with methodologic quality scores of 6 as of high quality. 9 RESULTS Search Results The results of our search strategy are presented in Fig 1. In total, 9 RCTs and 1 CCT are included in this review. Description of Studies Full details of the included studies are presented in Table 1. e296 SYMPTOMATIC TREATMENT OF MIGRAINE IN CHILDREN
3 Fig 1. Flowchart showing results of the search strategy. Participants The number of included participants in each trial ranged from 14 to 653 (mean patients), with a total of 1575 patients included in this review. The mean percentage of participants who dropped out from the trials was 19.8% (range: %). Mean age of participants was years (range: 4 18 years). Overall, the percentage of boys was generally the same as girls (mean: 55%; range: 42 82%). Nine of 10 trials used the criteria of the IHS to classify migraine, and 1 trial 10 used the criteria of Prensky and Sommer. 11 Six studies used a crossover design. Interventions The pharmacologic interventions 10,12 20 used for symptomatic treatment could be divided into analgesic treatment and nonanalgesic treatment. Regarding oral analgesic treatment, the effectiveness of acetaminophen, ibuprofen, and nimesulide was evaluated. Regarding the nonanalgesic interventions, nasal-spray sumatriptan, oral sumatriptan, oral rizatriptan, oral dihydroergotamine, intravenous (IV) prochlorperazine, and IV ketorolac were evaluated. Eight studies included a placebo comparison. Outcome Measures All studies used HA diaries to assess outcomes. Using this diary, among others, the frequency, intensity, and duration of HAs were scored on a Likert scale. In most studies a measure of clinical improvement was calculated. Improvement often was regarded as being clinically relevant when the patients HAs decline by 50%. This score is presented as HA clinical improvement in Table 1 and is our primary outcome measure. Other outcome measures were HA index, frequency, intensity or duration, rescue medication, and adverse events posttreatment and at follow-up. Methodologic Quality Quality scores (with positive items in parentheses) are presented in the study quality section of Table 1. The median score for methodologic quality was 6 (range: 3 9). Using a cutoff point of 6 of 10 criteria, 6 studies (60.0%) were considered to be of high quality. 10,12,13,16,18 20 Most prevalent methodologic shortcomings were a concealed randomization method (unclear: 80%), the intention-to-treat analysis (unclear: 50%; negative: 20%), and comparability of groups at baseline (unclear: 70%; negative: 10%). Effectiveness of Symptomatic Treatment Table 2 gives an overview of effect estimates of the different treatment modalities described below. Oral Analgesic Treatment Acetaminophen Versus Placebo One high-quality study compared 15 mg/kg acetaminophen with placebo. 12 was significantly higher for acetaminophen, although the use of rescue medication as well as the number of adverse events were not significantly different between the 2 groups. Adverse events mentioned for acetaminophen versus placebo were nausea (2.4% vs 3.7%) and vomiting (2.4% vs 7.4%). Ibuprofen Versus Placebo Two studies compared 10 mg/kg ibuprofen with placebo, including 1 high-quality study 12 and 1 low- e297
4 TABLE 1. Study Characteristics of Included Studies (Available From Authors or Published Electronically on Web Site of the Journal) Study Migraine Criteria Set Participants Interventions Outcome Measures Results* Ahonen et al 16 ; RCTcrossover; QS: 6 (items 1 and 4 8) Brousseau et al 10 ; RCTparallel group; QS: 9 (items 1 8 and 10) Hämäläinen et al 12 ; RCT-crossover; QS: 7 (items 1 and 4 9) Hämäläinen et al 18 ; RCT-crossover; QS: 6 (items 1 and 4 8) Ham al aïnen et al 20 ; RCT-crossover; QS: 7 (items 1, 4 8, and 10) IHS 45.7% female; mean age: 12.4 y (SD: 2.4 y); n 129; 42 dropouts; time since onset of HAs: 3.8 y (range: y) Prensky and Sommer 58.1% female; mean age: 13.7 y (range: y); n 62; no dropouts; time since onset of HAs: not stated IHS 50% female; mean age: 10.7 y (range: y); n 106; 28 dropouts; median time since onset of HAs: 2.83 y (range: y) IHS 52.2% female; median age: 12.3 y (range: y); n 31; 8 dropouts; median time since onset of HAs: 3.4 y (range: y) IHS 38.5% female; mean age: 10.2 y (SD: 3.2 y); n 16; 4 dropouts; median time since onset of HAs: 3.4 y I: sumatriptan nasal spray (10 mg with kg N 31 and 20 mg with 40 kg body weight N 63 for 1 attack); N 129; 39 dropouts C: placebo nasal spray (1 attack); N 129; 42 dropouts MT: before and 15, 30, and 60 min thereafter, continuing hourly for up to 7 h unless Rescue medication: nonsteroidal antiinflammatory drugs or acetaminophen 2 h after intake of study drug I: IV prochlorperazine (0.15 mg/kg max: 10 mg 10 ml/kg bolus of normal saline solution for 1 attack); N 33 C: IV ketorolac (0.5 mg/kg max: 30 mg 10 ml/kg bolus of normal saline solution for 1 attack); N 29 HA diary (5-point Likert HA clinical improvement: 1 h I vs C RR: 1.86 (1.27, 2.72); 2hIvs C RR: 1.76 (1.29, 2.39) Rescue medication Rescue medication: I vs C RR: 0.64 (0.44, 0.92) Adverse events Adverse events: I vs C RR: 4.83 (2.27, 10.29) HA diary (Faces pain HA clinical improvement: 60 min; I vs C RR: 1.54 (1.07, 2.20) MT: before and 30 and 60 min thereafter Adverse events Adverse events: I vs C RR: 0.88 (0.06, 13.4) I: acetaminophen (15 mg/kg for 1 attack); N 106; 26 dropouts C1: ibuprofen (10 mg/kg for 1 attack); N 106; 28 dropouts C2: placebo medication (cellulose for 1 attack); N 106; 28 dropouts MT: before and 30 and 60 min thereafter, continuing hourly for up to 5 h unless Rescue medication: usual treatment within 2hor 2 h after intake of study drug I: sumatriptan tablet (50 mg per m 2 body surface area 6 12 y or 100 mg for 1.5 m 2 body surface area 12 y for 1 attack); N 31; 8 dropouts C: placebo medication. 1 attack, N 31, 8 dropouts MT: before and 30 and 60 min thereafter, continuing hourly for up to 5 h unless Rescue medication: usual treatment after intake of study drug I: dihydroergotamine (20 g/kg [or 40 g/ kg by no response for 1 attack); N 16; 4 dropouts C: placebo medication (1 attack); N 16; 3 dropouts HA diary (5-point Likert scale or visual analogue Rescue medication C1 RR: 0.93 (0.70, 1.24); I vs C2 RR: 1.46 (1.02, 2.10); C1 vs C2 RR: 1.57 (1.10, 2.24) Rescue medication: 2 hivsc1 RR: 0.98 (0.33, 2.89); I vs C2 RR: 0.73 (0.27, 2.01); C1 vs C2 RR: 0.75 (0.27, 2.06); 2 hivsc1 RR: 3.41 (0.73, 15.9); I vs C2 RR: 0.68 (0.27, 1.70); C1 vs C2 RR: 0.20 (0.05, 0.88) Adverse events Adverse events: I vs C1 RR: 0.49 (0.15, 1.56); I vs C2 RR: 0.43 (0.14, 1.35); C1 vs C2 RR: 0.89 (0.36, 2.19) HA diary (5-point Likert scale or visual analogue, resulting in pain intensity difference and summed pain intensity C RR: 1.40 (0.52, 3.77) Rescue medication Rescue medication: 1 2 h I vs C RR: 2.00 (0.81, 4.94) Adverse events Adverse events: I vs C RR: 2.67 (0.81, 8.80) HA diary (5-point Likert C RR: 3.79 (0.97, 14.8) e298 SYMPTOMATIC TREATMENT OF MIGRAINE IN CHILDREN
5 TABLE 1. Continued Study Migraine Criteria Set Participants Interventions Outcome Measures Results* Lewis et al 14 ; RCTparallel group; QS: 4 (items 1, 5, 7, and 8) Soriani et al 15 ; CCTcrossover; QS: 3 (items 4, 8, and 10) Ueberall and Wenzel 17 ; RCT-crossover; QS: 5 (items 1, 4, and 8 10) Winner et al 13 ; RCTparallel group; QS: 8 IHS 44% (I) and 38.5% (C) female; mean age: 9.1 y (range: 6 12 y); n 138; 54 dropouts; time since onset of HAs: not stated IHS 50% female; mean age: 12.8 y (SD: 3.1 y); n 66; 6 dropouts; time since onset of HAs: not stated IHS 50% female; mean age: 8.1 (I) and 8.3 (C) y (range: y); n 14; no dropouts; median time since onset of HAs: 1.1 y (range: y) IHS 51.5% female; mean age: 14.1 y (SD: 1.6 y); n (items 1 8) 653; 143 dropouts; time since onset of HAs: not stated MT: before and 30 and 60 min thereafter, continuing hourly for up to 5 h unless Rescue medication: usual treatment after intake of study drug I: ibuprofen (7.5 mg/kg liquid for 1 attack); N 45 C: placebo medication (1 attack); N 39 MT: before and 30, 60, and 90 min thereafter, continuing hourly for up to 4 h unless Rescue medication: usual treatment after intake of study drug I: acetaminophen (15 mg/kg for 10 attacks); N 66; 6 dropouts (300 attacks) C: nimesulide (2.5 mg/kg for 10 attacks); N 66; 6 dropouts (300 attacks) MT: before and 90 min thereafter unless I: sumatriptan nasal spray (20 mg per dose for 1 attack); N 14 C: placebo nasal spray (0.9% sodium chloride for 1 attack); N 14 MT: before and 30 and 60 min thereafter, continuing hourly for up to 5 h unless I: sumatriptan nasal spray (5 mg per dose for 1 attack); N 128 C1: sumatriptan nasal spray (10 mg per dose for 1 attack); N 133 C2: sumatriptan nasal spray (20 mg per dose for 1 attack); N 118 Rescue medication Rescue medication: 1 2 h I vs C RR: 1.08 (0.41, 2.83) Adverse events Adverse events: I vs C RR: 2.17 (0.22, 20.94) HA diary (4-point Likert Rescue medication Adverse events HA diary (4-point Likert Rescue medication HA diary (4-point Likert C RR: 1.40 (1.00, 1.96) Rescue medication: 2 h I vs C RR: 0.06 (0.01, 0.42) HA clinical improvement: 1.5 h I vs C RR: 0.97 (0.89, 1.06) Adverse events: I vs C RR: 1.25 (0.35, 4.43) C RR: 2.00 (1.05, 3.80) Rescue medication: 1 2 h I vs C RR: 2.00 (0.81, 4.94) C1 RR: (0.87, 1.24); I vs C2 RR: 1.05 (0.87, 1.26); I vs C3 RR: 1.26 (1.02, 1.54); C1 vs C2 RR: 1.01 (0.84, 1.22); C1 vs C3 RR: 1.21 (0.99, 1.49); C2 vs C3 RR: 1.20 (0.97, 1.49) Rescue medication Rescue medication: I vs C1 RR: 0.97 (0.61, 1.54); I vs C2 RR: 0.80 (0.51, 1.26); I vs C3 RR: 0.64 (0.42, 0.97); C1 vs C2 RR: 0.83 (0.53, 1.29); C1 vs C3 RR: 0.66 (0.44, 1.00); C2 vs C3 RR: 0.80 (0.54, 1.18) C3: placebo nasal spray (1 attack); N 131 Adverse events Adverse events: I vs C1 RR: 0.92 MT: before and 15, 30, 60, and 120 min thereafter (0.67, 1.26); I vs C2 RR: 0.88 (0.64, 1.22); I vs C3 RR: 1.96 (1.26, 3.03); C1 vs C2 RR: 0.96 (0.71, 1.31); C1 vs C3 RR: 2.13 (1.39, 3.28); C2 vs C3 RR: 2.22 (1.44, 3.41) e299
6 TABLE 1. Continued Participants Interventions Outcome Measures Results* Study Migraine Criteria Set C RR: 1.18 (0.98, 1.42) HA diary (4-point Likert I: rizatriptan (5 mg per dose for 3 attacks); N 149 N 360; N completed: 296 Winner et al 19 ; RCTparallel group; QS: 5 (items 1, 3, and 8 10) IHS 18.2% female; mean age: C: placebo medication (3 attacks); N 147 Rescue medication Rescue medication: I vs C RR: 0.84 (0.64, 1.10) MT: before and 30, 60, and 90 min thereafter, continuing hourly for up to 4 h unless 14 y; time since onset of HAs: not stated Adverse events Adverse events: I vs C RR: 0.95 (0.69, 1.30) Rescue medication: usual treatment 2 h after intake of study drug C1 indicates control 1; C2, control 2; C3, control 3; I, intervention; MT, measure time; N, number of subjects who completed the trial; ns, not significant; QS, quality score on Delphi list, with positive items in parentheses. * Shown are standard mean differences with 95% confidence intervals (in parentheses). quality study. 14 In the pooled analysis, was significantly higher for ibuprofen, and the use of rescue medication was significantly higher for placebo. No difference in the number of adverse events was found (vomiting: 4.9% vs 7.4%; nausea: 3.7% for both; gastric pain: 1.2% [ibuprofen only]). 12 Acetaminophen Versus Ibuprofen One high-quality study compared 15 mg/kg acetaminophen with 10 mg/kg ibuprofen and found no significant differences for any outcome measures. 12 Acetaminophen Versus Nimesulide One low-quality study compared 15 mg/kg acetaminophen with 2.5 mg/kg nimesulide and found no significant differences. 15 Adverse events (mild abdominal discomfort and nausea) accompanied 9% of the treatments with acetaminophen and 6% of treatments with nimesulide. We conclude that there is moderate evidence that both acetaminophen and ibuprofen are more effective in reduction of symptoms 1 and 2 hours after intake than placebo, with minor adverse effects. No clear differences in effect were found between acetaminophen and ibuprofen or nimesulide. Triptans Sumatriptan Versus Placebo Four studies compared sumatriptan and placebo, including 3 high-quality studies 13,16,18 and 1 lowquality study. 17 Three studies evaluated intranasal sumatriptan. 13,16,17 Ahonen et al 16 did not present outcomes for the doses separately. At 1 and 2 hours after administration we found significant differences concerning in favor of sumatriptan. The use of rescue medication was significantly more for placebo, whereas the number of adverse events was significantly more for sumatriptan. Adverse events mentioned for sumatriptan versus placebo included bad taste (25.7% vs 3.1%), nausea (7.9% vs 8.3%), vomiting (3.9% vs 4.4%), triptan sensation (temperature [warmth], burning/stinging sensations, or paresthesia) (2.4% vs 1.6%), feeling of lightheadedness (2.2%, sumatriptan only), and stiffness in the jaw (1.1%, sumatriptan only). One high-quality study evaluated oral sumatriptan and found no significant differences for any outcome measures. 18 Adverse events mentioned for sumatriptan were vomiting (17.4%), nausea (8.7%), dizziness (4.3%), and very strong photophobia (4.3%). For placebo, the adverse events were vomiting (8.7%) and nausea (4.3%). Rizatriptan Versus Placebo One low-quality study 19 compared oral rizatriptan with placebo and found no significant differences for clinical improvement of HA, rescue medication, or the number of adverse events. Adverse events mentioned for rizatriptan versus placebo were dry mouth (4.7% vs 3.4%), dizziness (4.7% vs 4.8%), asthenia (3.4% vs 2.0%), nausea (2.7% vs 8.2%), and somnolence (2.7% vs 8.2%). We conclude that there is moderate evidence that nasal-spray sumatriptan is more effective in the reduction of symptoms than placebo, but with signifi- e300 SYMPTOMATIC TREATMENT OF MIGRAINE IN CHILDREN
7 TABLE 2. Overview: Results of Symptomatic Treatment Comparisons Total Trials, n HA Clinical Improvement 2 h After Intake, RR (95% CI) Rescue Medication 2 h After Intake, RR (95% CI) Number of Adverse Events, RR (95% CI) Analgesic treatment Acetaminophen vs placebo 1 H 1.5 (1.0, 2.1)* 0.7 (0.3, 1.7) 0.4 (0.1, 1.4) Ibuprofen vs placebo 1 H, 1 L 1.5 (1.2, 1.9)* 0.1 (0.0, 0.4)* 0.9 (0.4, 2.2) Acetaminophen vs ibuprofen 1 H 0.9 (0.7, 1.2) 3.4 (0.7, 15.9) 0.5 (0.2, 1.6) Acetaminophen vs nimesulide 1 L 1.0 (0.9, 1.1) 1.3 (0.4, 4.4) Triptans treatment Sumatriptan nasal spray vs placebo 2 H, 1 L 1.4 (1.2, 1.7)* 0.7 (0.5, 0.8)* 2.5 (1.7, 3.7)* Sumatriptan tablets vs placebo 1 H 1.4 (0.5, 3.8) 2.0 (0.8, 4.9) 2.7 (0.8, 8.8) Rizatriptan vs placebo 1 L 1.2 (1.0, 1.4) 0.8 (0.6, 1.1) 1.0 (0.7, 1.3) Other treatment Dihydroergotamine vs placebo 1 H 3.8 (1.0, 14.8) 1.1 (0.4, 2.8) 2.2 (0.2, 20.9) Prochlorperazine (IV) vs ketorolac (IV) 1 H 1.5 (1.1, 2.2)* 0.9 (0.1, 13.4) H indicates high-quality trial; L, low-quality trial; n, number of trials, which can have 1 comparison;, no data available. * Significant difference. cantly more adverse events. No differences in effect were found between oral triptans and placebo. Other Medications Dihydroergotamine Versus Placebo One high-quality study compared oral dihydroergotamine with placebo. 20 We found no significant differences for clinical improvement of HA, rescue medication, or the number of adverse events, probably because of statistically low power (smallest treatment group: 25 patients). The 1 adverse event mentioned for dihydroergotamine versus placebo was vomiting (16.7% vs 7.7%). Prochlorperazine Versus Ketorolac One high-quality study compared IV prochlorperazine with IV ketorolac. 10 After 1 hour, prochlorperazine worked significantly better compared with ketorolac. The number of adverse events was not significantly different between the 2 groups. We conclude that there is moderate evidence that IV prochlorperazine is more effective than IV ketorolac in the reduction of symptoms 1 hour after intake. No differences in effect were found between oral dihydroergotamine and placebo. Worst-Case Analysis When sufficient data were available, a worst-case analysis was conducted in which all dropouts were assumed to be nonresponders. Only 4 studies provided sufficient data to allow a worst-case analysis, but the overall conclusions did not change. 12,16,18,20 DISCUSSION The most effective therapies for migraine are those that can be given quickly at the beginning of an attack and have a rapid onset of action. Based on the available literature, we conclude that over-thecounter analgesics such as acetaminophen and ibuprofen are relatively safe and effective. Most RRs were at 1.5, which means that there is 50% more chance (risk) to recover when using, for example, ibuprofen instead of placebo. No clear differences in effect were found between the different analgesics. After finishing our study, another systematic review on treatments of pediatric migraine was published with similar conclusions. 21 This review included just 5 controlled trials on pharmacologic treatment, and no statistical pooling resulting in an overall effect estimate was performed. The new frontier for symptomatic treatment is likely to be the development of triptan agents for use in children. We found moderate evidence that nasalspray sumatriptan is more effective than placebo, but no differences in effect were found between oral sumatriptan and rizatriptan compared with placebo. It is possible that oral triptans may not be effective in children during acute migraine attacks because of nausea and vomiting. In adults, sumatriptan pharmacokinetic data have been collected after oral administration in patients suffering from migraine: the absorption from an oral tablet was delayed during a migraine attack because of gastric stasis, nausea, and vomiting. 22 It is likely that this also takes place in children. Also, migraine attacks in children tend to be shorter than those in adults and may spontaneously remit within 2 hours. Medications, which would be anticipated to provide maximum benefit close to 2 hours after administration, may not show efficacy for shorter-duration attacks, and results may be comparable to those when using placebos. Nasal administration offers an advantage over the tablet administration, because it provides faster absorption immediately after dosing, which results in faster initial relief of migraine symptoms. The only disadvantage of the use of nasal-spray sumatriptan compared with the other investigated medications is the adverse events. Although all reported adverse events were minor for all reported medications, the use of nasal-spray sumatriptan resulted in significantly more adverse events than the use of placebo. Although systematic reviews offer the least biased method of summarizing research literature, our results must be interpreted with consideration of the quality of evidence from which they were obtained. None of the crossover trials provided separate data of each treatment period or data concerning the existence of a carryover effect. Positive results of analgesics and triptans in children and adolescents have frequently emerged from open-label studies, which might be biased. Most treatments have been evaluated in only 1 or e301
8 2 studies, which limits the generalizability of the findings. There are no clinical trials that tested the efficacy of different triptans with each other. Studies comparing the efficacy of triptans to analgesic drugs with known efficacy are also needed. Clinical improvement of HA and adverse events are 2 outcome measures frequently used in the included trials for symptomatic treatment of migraine attacks. Although most studies described the adverse events as mild and safe, the simple description of the kind and number of adverse events often gives insufficient insight into the severity and appreciation of the adverse events for a child. Outcome measures such as quality of life, satisfaction of child or parents, and days missed at school are not used. We strongly recommend performing a large highquality RCT that evaluates different symptomatic medications compared with each other or to placebo treatment. Favorable high-quality studies should be performed and reported according to the CONSORT statement (see Clinical improvement of HA should be used as the primary outcome measure, but quality of life, days missed at school, and satisfaction of child or parents should also be used as outcome measures in future studies. CONCLUSIONS There is moderate evidence that analgesics (acetaminophen and ibuprofen) and nasal-spray sumatriptan are more effective than placebo treatment. Based on the available literature, no differences in effect were found between the different analgesics. ACKNOWLEDGMENT We thank the Netherlands Organization for Health Research and Development for funding this research. REFERENCES 1. Annequine D, Tourniaire B, Massiou H. Migraine and headache in children and adolescence. Pediatr Clin North Am. 2000;37: Guidetti V, Galli F, Fabrizi P, et al. Headache and psychiatric comorbidity: clinical aspects and outcome in an 8-year follow-up study. Cephalalgia. 1998;18: Headache Classification Committee of the International Headache Society. Classification and diagnostic criteria for headache disorders, cranial neuralgias and facial pain. Cephalalgia. 1988;8(suppl 7): Headache Classification Committee of the International Headache Society. The international classification of headache disorders. Cephalalgia. 2004;24(suppl 1): Major PW, Grubisa HIS, Thie NMR. Triptans for treatment of acute pediatric migraine: a systematic literature review. Pediatr Neurol. 2003; 29: Robinson KA, Dickerson K. Development of a highly sensitive search strategy for the retrieval of reports of controlled trials using PubMed. Int J Epidemiol. 2002;31: Ad Hoc Committee on the Classification of Headache of the National Institute of Neurological Diseases and Blindness. Classification of headache. JAMA. 1962;179: Verhagen AP, de Vet HCW, de Bie RA, et al. The Delphi list: a criteria list for quality assessment of randomised clinical trials for conducting systematic reviews developed by Delphi consensus. J Clin Epidemiol. 1998;51: van Tulder M, Furlan A, Bombardier C, Bouter L; Editorial Board of the Cochrane Collaboration Back Review Group. Updated method guidelines for systematic reviews in the Cochrane Collaboration Back Review Group. Spine. 2003;28: Brousseau DC, Duffy SJ, Anderson AC, Linakis JG. Treatment of pediatric migraine headaches: a randomized, double-blind trial of prochlorperazine versus ketorolac. Ann Emerg Med. 2004;43: Prensky AL, Sommer D. Diagnosis and treatment of migraine in children. Neurology. 1979;29: Hämäläinen ML, Hoppu K, Valkeila E, Santavuori P. Ibuprofen or acetaminophen for the acute treatment of migraine in children: a double-blind, randomized, placebo-controlled, crossover study. Neurology. 1997;48: Winner P, Rothner AD, Saper J, et al. A randomized, double-blind, placebo-controlled study of sumatriptan nasal spray in the treatment of acute migraine in adolescents. Pediatrics. 2000;106: Lewis DW, Kellstein D, Dahl G, et al. Children s ibuprofen suspension for the acute treatment of pediatric migraine. Headache. 2002;42: Soriani S, Battistella PA, Naccarella C, Tozzi E, Fiumana E, Fanaro S. Nimesulide and acetaminophen for the treatment of juvenile migraine: a study for comparison of efficacy, safety, and tolerability. Headache Q. 2001;12: Ahonen K, Hämäläinen ML, Rantala H, Hoppu K. Nasal sumatriptan is effective in treatment of migraine attacks in children: a randomized trial. Neurology. 2004;62: Ueberall MA, Wenzel D. Intranasal sumatriptan for the acute treatment of migraine in children. Neurology. 1999;52: Hämäläinen ML, Hoppu K, Santavuori P. Sumatriptan for migraine attacks in children: a randomized placebo-controlled study. Do children with migraine respond to oral sumatriptan differently from adults? Neurology. 1997;48: Winner P, Lewis D, Visser WH, Jiang K, Ahrens S, Evans JK; Rizatriptan Adolescent Study Group. Rizatriptan 5 mg for the acute treatment of migraine in adolescents: a randomized, double-blind, placebocontrolled study. Headache. 2002;42: Hämäläinen ML, Hoppu K, Santavuori PR. Oral dihydroergotamine for therapy-resistant migraine attacks in children. Pediatr Neurol. 1997;16: Lewis D, Ashwal S, Hershey A, et al. Practice parameter: pharmacological treatment of migraine headache in children and adolescents: report of the American Academy of Neurology Quality Standards Subcommittee and the Practice Committee of the Child Neurology Society. Neurology. 2004;63: Culter NR, Hussey EK, Sramek JJ, et al. Pharmacokinetics of oral sumatriptan in migraine patients during an attack and while pain free. Biol Psychiatry. 1992;31:180A e302 SYMPTOMATIC TREATMENT OF MIGRAINE IN CHILDREN
9 Symptomatic Treatment of Migraine in Children: A Systematic Review of Medication Trials Léonie Damen, Jacques K.J. Bruijn, Arianne P. Verhagen, Marjolein Y. Berger, Jan Passchier and Bart W Koes Pediatrics 2005;116;e295 DOI: /peds Updated Information & Services References Permissions & Licensing Reprints including high resolution figures, can be found at: This article cites 22 articles, 7 of which you can access for free at: 1 Information about reproducing this article in parts (figures, tables) or in its entirety can be found online at: Information about ordering reprints can be found online: Pediatrics is the official journal of the American Academy of Pediatrics. A monthly publication, it has been published continuously since. Pediatrics is owned, published, and trademarked by the American Academy of Pediatrics, 141 Northwest Point Boulevard, Elk Grove Village, Illinois, Copyright 2005 by the American Academy of Pediatrics. All rights reserved. Print ISSN:.
10 Symptomatic Treatment of Migraine in Children: A Systematic Review of Medication Trials Léonie Damen, Jacques K.J. Bruijn, Arianne P. Verhagen, Marjolein Y. Berger, Jan Passchier and Bart W Koes Pediatrics 2005;116;e295 DOI: /peds The online version of this article, along with updated information and services, is located on the World Wide Web at: Pediatrics is the official journal of the American Academy of Pediatrics. A monthly publication, it has been published continuously since. Pediatrics is owned, published, and trademarked by the American Academy of Pediatrics, 141 Northwest Point Boulevard, Elk Grove Village, Illinois, Copyright 2005 by the American Academy of Pediatrics. All rights reserved. Print ISSN:.
Pharmacological treatment of attacks in juvenile migraine
 J Headache Pain (2004) 5:S62 S66 DOI 10.1007/s10194-004-0110-9 Beatrice Gallai Giovanni Mazzotta Paola Sarchielli Pharmacological treatment of attacks in juvenile migraine Received: Accepted in revised
J Headache Pain (2004) 5:S62 S66 DOI 10.1007/s10194-004-0110-9 Beatrice Gallai Giovanni Mazzotta Paola Sarchielli Pharmacological treatment of attacks in juvenile migraine Received: Accepted in revised
Disclosures. Triptans for Kids 5/16/13
 5/16/13 Disclosures Triptans for Kids Amy A. Gelfand, MD GelfandA@neuropeds.ucsf.edu Departments of Neurology and Pediatrics UCSF Child Neurology and Headache Center I receive grant funding from: NIH/NINDS
5/16/13 Disclosures Triptans for Kids Amy A. Gelfand, MD GelfandA@neuropeds.ucsf.edu Departments of Neurology and Pediatrics UCSF Child Neurology and Headache Center I receive grant funding from: NIH/NINDS
Specific Care Question : Question Originator: Plain Language Summary from The Office of Evidence Based Practice: Conditional Recommendation
 Ketorolac for Refractory Migraine in the ED Specific Care Question : In the pediatric patient diagnosed with refractory migraine is ketorolac an effective treatment? Question Originator: Migraine Therapy
Ketorolac for Refractory Migraine in the ED Specific Care Question : In the pediatric patient diagnosed with refractory migraine is ketorolac an effective treatment? Question Originator: Migraine Therapy
ISSN doi: /head VC 2015 American Headache Society Published by Wiley Periodicals, Inc.
 ISSN 0017-8748 Headache doi: 10.1111/head.12746 VC 2015 American Headache Society Published by Wiley Periodicals, Inc. Review Article Acute Treatment Therapies for Pediatric Migraine: A Qualitative Systematic
ISSN 0017-8748 Headache doi: 10.1111/head.12746 VC 2015 American Headache Society Published by Wiley Periodicals, Inc. Review Article Acute Treatment Therapies for Pediatric Migraine: A Qualitative Systematic
I have no financial relationships to disclose. I will not discuss investigational use of medication in my presentation.
 I have no financial relationships to disclose. I will not discuss investigational use of medication in my presentation. In 1962, Bille published landmark epidemiologic survey of headache among 9,000 school
I have no financial relationships to disclose. I will not discuss investigational use of medication in my presentation. In 1962, Bille published landmark epidemiologic survey of headache among 9,000 school
Study No.: Title: Rationale: Phase: Study Period: Study Design: Centres: Indication: Treatment: Objectives: Primary Outcome/Efficacy Variable:
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
ARTICLE. Migraine Therapeutics in Adolescents. A Systematic Analysis and Historic Perspectives of Triptan Trials in Adolescents
 COMPARATIVE EFFECTIVENESS RESEARCH ARTICLE Migraine Therapeutics in Adolescents A Systematic Analysis and Historic Perspectives of Triptan Trials in Adolescents Haihao Sun, MD, PhD; Eric Bastings, MD;
COMPARATIVE EFFECTIVENESS RESEARCH ARTICLE Migraine Therapeutics in Adolescents A Systematic Analysis and Historic Perspectives of Triptan Trials in Adolescents Haihao Sun, MD, PhD; Eric Bastings, MD;
5 HT receptor agonists (triptans) and Pediatric Migraine Critically Appraised Topic (CAT)
 5 HT receptor agonists (triptans) and Pediatric Migraine Critically Appraised Topic (CAT) PICOT Question: In children and adolescents does use of 5 HT receptor agonists (triptans) versus no treatment treat
5 HT receptor agonists (triptans) and Pediatric Migraine Critically Appraised Topic (CAT) PICOT Question: In children and adolescents does use of 5 HT receptor agonists (triptans) versus no treatment treat
Rizatriptan vs. ibuprofen in migraine: a randomised placebo-controlled trial
 J Headache Pain (2007) 8:175 179 DOI 10.1007/s10194-007-0386-7 ORIGINAL Usha Kant Misra Jayantee Kalita Rama Kant Yadav Rizatriptan vs. ibuprofen in migraine: a randomised placebo-controlled trial Received:
J Headache Pain (2007) 8:175 179 DOI 10.1007/s10194-007-0386-7 ORIGINAL Usha Kant Misra Jayantee Kalita Rama Kant Yadav Rizatriptan vs. ibuprofen in migraine: a randomised placebo-controlled trial Received:
Adult & Pediatric Patients. Stanford Health Care, Division Pain Medicine
 Acute Treatment Strategies in Adult & Pediatric Patients Theresa Mallick Searle, MS, RN BC, ANP BC Disclosures Speakers Bureau: Allergan, Depomed Acute Treatment Strategies in Adult & Pediatric Patients
Acute Treatment Strategies in Adult & Pediatric Patients Theresa Mallick Searle, MS, RN BC, ANP BC Disclosures Speakers Bureau: Allergan, Depomed Acute Treatment Strategies in Adult & Pediatric Patients
Study No.: Title: Rationale: Phase: Study Period: Study Design: Centers: Indication: Treatment: Objectives: Primary Outcome/Efficacy Variable:
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
Faculty Disclosures. Learning Objectives. Acute Treatment Strategies
 WWW.AMERICANHEADACHESOCIETY.ORG Acute Treatment Strategies Content developed by: Lawrence C. Newman, MD, FAHS Donna Gutterman, PharmD Faculty Disclosures LAWRENCE C. NEWMAN, MD, FAHS Dr. Newman has received
WWW.AMERICANHEADACHESOCIETY.ORG Acute Treatment Strategies Content developed by: Lawrence C. Newman, MD, FAHS Donna Gutterman, PharmD Faculty Disclosures LAWRENCE C. NEWMAN, MD, FAHS Dr. Newman has received
Review Article Pediatric Migraine
 International Pediatrics Volume 2009, Article ID 424192, 7 pages doi:10.1155/2009/424192 Review Article Pediatric Migraine Ubaid Hameed Shah and Veena Kalra Apollo Centre for Advanced Pediatrics, Indraprastha
International Pediatrics Volume 2009, Article ID 424192, 7 pages doi:10.1155/2009/424192 Review Article Pediatric Migraine Ubaid Hameed Shah and Veena Kalra Apollo Centre for Advanced Pediatrics, Indraprastha
HMFP Comprehensive Headache Center Department of Anesthesia, Critical Care and Pain Medicine Beth Israel Deaconess Medical Center Instructor in
 HMFP Comprehensive Headache Center Department of Anesthesia, Critical Care and Pain Medicine Beth Israel Deaconess Medical Center Instructor in Anesthesia and Neurology Harvard Medical School Limited time
HMFP Comprehensive Headache Center Department of Anesthesia, Critical Care and Pain Medicine Beth Israel Deaconess Medical Center Instructor in Anesthesia and Neurology Harvard Medical School Limited time
Çiçek Wöber-Bingöl HEADACHE UNIT FOR CHILDREN AND ADOLESCENCE
 Headache in children and adolescents Çiçek Wöber-Bingöl HEADACHE UNIT FOR CHILDREN AND ADOLESCENCE Dept. of Psychiatry of Childhood and Adolescence Medical University of Vienna, Vienna, Austria Impact
Headache in children and adolescents Çiçek Wöber-Bingöl HEADACHE UNIT FOR CHILDREN AND ADOLESCENCE Dept. of Psychiatry of Childhood and Adolescence Medical University of Vienna, Vienna, Austria Impact
Dose: Metoclopramide -0.1 mg/kg (max 10 mg) IV, over 15 minutes
 Metoclopramide for Refractory Migraine in the ED Specific Care Question : In the pediatric patient diagnosed with refractory migraine, is metoclopramide an effective treatment? Question Originator: Migraine
Metoclopramide for Refractory Migraine in the ED Specific Care Question : In the pediatric patient diagnosed with refractory migraine, is metoclopramide an effective treatment? Question Originator: Migraine
Extended Abstracts: Naproxen
 Primary Literature 1) Prior MJ, Cooper KM, May LG, Bowen DL. Efficacy and safety of acetaminophen and naproxen in the treatment of tension-type headache. A randomized, double-blind, placebo-controlled
Primary Literature 1) Prior MJ, Cooper KM, May LG, Bowen DL. Efficacy and safety of acetaminophen and naproxen in the treatment of tension-type headache. A randomized, double-blind, placebo-controlled
Acute treatment of migraine in children and adolescents
 Acute treatment of migraine in children and adolescents Cristiano Termine, MD a,b Matteo Ferri, MD a,b Umberto Balottin, MD b,c a Child Neuropsychiatry Unit, Department of Clinical and Biological Sciences,
Acute treatment of migraine in children and adolescents Cristiano Termine, MD a,b Matteo Ferri, MD a,b Umberto Balottin, MD b,c a Child Neuropsychiatry Unit, Department of Clinical and Biological Sciences,
Is Topiramate Effective in Preventing Pediatric Migraines?
 Philadelphia College of Osteopathic Medicine DigitalCommons@PCOM PCOM Physician Assistant Studies Student Scholarship Student Dissertations, Theses and Papers 2013 Is Topiramate Effective in Preventing
Philadelphia College of Osteopathic Medicine DigitalCommons@PCOM PCOM Physician Assistant Studies Student Scholarship Student Dissertations, Theses and Papers 2013 Is Topiramate Effective in Preventing
Migraine: Developing Drugs for Acute Treatment Guidance for Industry
 Migraine: Developing Drugs for Acute Treatment Guidance for Industry U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation and Research (CDER) February 2018
Migraine: Developing Drugs for Acute Treatment Guidance for Industry U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation and Research (CDER) February 2018
MEASURE #1: MEDICATION PRESCRIBED FOR ACUTE MIGRAINE ATTACK Headache
 MEASURE #1: MEDICATION PRESCRIBED FOR ACUTE MIGRAINE ATTACK Headache Measure Description Percentage of patients age 12 years and older with a diagnosis of migraine who were prescribed a guideline recommended
MEASURE #1: MEDICATION PRESCRIBED FOR ACUTE MIGRAINE ATTACK Headache Measure Description Percentage of patients age 12 years and older with a diagnosis of migraine who were prescribed a guideline recommended
5.9. Rehabilitation to Improve Central Pain
 5.9. Rehabilitation to Improve Central Pain Evidence Tables and References Canadian Best Practice Recommendations for Stroke Care 2011-2013 Update Last Updated: June 25 th, 2013 Contents Search Strategy...
5.9. Rehabilitation to Improve Central Pain Evidence Tables and References Canadian Best Practice Recommendations for Stroke Care 2011-2013 Update Last Updated: June 25 th, 2013 Contents Search Strategy...
The best defense is a good offense. Optimizing the Acute Treatment of Migraine. Disclosures 11/10/2017
 Optimizing the Acute Treatment of Migraine Brian M. Plato, DO, FAHS Norton Neuroscience Institute Louisville, KY Disclosures Speakers Bureau (personal): Allergan, Depomed, Avanir Research Funding (paid
Optimizing the Acute Treatment of Migraine Brian M. Plato, DO, FAHS Norton Neuroscience Institute Louisville, KY Disclosures Speakers Bureau (personal): Allergan, Depomed, Avanir Research Funding (paid
Despite the widespread use of triptans ... REPORTS... Almotriptan: A Review of Pharmacology, Clinical Efficacy, and Tolerability
 ... REPORTS... Almotriptan: A Review of Pharmacology, Clinical Efficacy, and Tolerability Randal L. Von Seggern, PharmD, BCPS Abstract Objective: This article summarizes preclinical and clinical data for
... REPORTS... Almotriptan: A Review of Pharmacology, Clinical Efficacy, and Tolerability Randal L. Von Seggern, PharmD, BCPS Abstract Objective: This article summarizes preclinical and clinical data for
How do we treat migraine? New SIGN Guidelines
 How do we treat migraine? New SIGN Guidelines Managing your migraine Migraine Trust, Edinburgh 2018 Callum Duncan Consultant Neurologist Aberdeen Royal Infirmary Chair SIGN Guideline 155 Premonitory Mood
How do we treat migraine? New SIGN Guidelines Managing your migraine Migraine Trust, Edinburgh 2018 Callum Duncan Consultant Neurologist Aberdeen Royal Infirmary Chair SIGN Guideline 155 Premonitory Mood
Biofeedback as a Treatment of Headache
 Biofeedback as a Treatment of Headache Policy Number: 2.01.29 Last Review: 7/2018 Origination: 7/2008 Next Review: 7/2019 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) may provide coverage
Biofeedback as a Treatment of Headache Policy Number: 2.01.29 Last Review: 7/2018 Origination: 7/2008 Next Review: 7/2019 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) may provide coverage
A new questionnaire for assessment of adverse events associated with triptans: methods of assessment influence the results. Preliminary results
 J Headache Pain (2004) 5:S112 S116 DOI 10.1007/s10194-004-0123-4 Michele Feleppa Fred D. Sheftell Luciana Ciannella Amedeo D Alessio Giancarlo Apice Nino N. Capobianco Donato M.T. Saracino Walter Di Iorio
J Headache Pain (2004) 5:S112 S116 DOI 10.1007/s10194-004-0123-4 Michele Feleppa Fred D. Sheftell Luciana Ciannella Amedeo D Alessio Giancarlo Apice Nino N. Capobianco Donato M.T. Saracino Walter Di Iorio
...SELECTED ABSTRACTS...
 The following abstracts, from medical journals containing literature on migraine management, were selected for their relevance to this Special Report supplement. Two Sumatriptan Studies Two double-blind
The following abstracts, from medical journals containing literature on migraine management, were selected for their relevance to this Special Report supplement. Two Sumatriptan Studies Two double-blind
Migraine treatment in developmental age: guidelines update
 J Headache Pain (2010) 11:267 276 DOI 10.1007/s10194-010-0205-4 TUTORIAL Migraine treatment in developmental age: guidelines update Laura Papetti Alberto Spalice Francesco Nicita Maria Chiara Paolino Rosa
J Headache Pain (2010) 11:267 276 DOI 10.1007/s10194-010-0205-4 TUTORIAL Migraine treatment in developmental age: guidelines update Laura Papetti Alberto Spalice Francesco Nicita Maria Chiara Paolino Rosa
Neurological disorders. Headache (chronic tension-type) Search date December 2013 Mona Ghadiri-Sani and Nicholas Silver ...
 Headache (chronic tensiontype) Search date December 213 Mona GhadiriSani and Nicholas Silver.................................................. ABSTRACT INTRODUCTION: Chronic tensiontype headache (CTTH)
Headache (chronic tensiontype) Search date December 213 Mona GhadiriSani and Nicholas Silver.................................................. ABSTRACT INTRODUCTION: Chronic tensiontype headache (CTTH)
Neurologic Disorders: Headache in Children
 1 of 12 17/06/2014 7:57 PM Print Close Neurologic Disorders: Headache in Children Sharon Whiting, MBBS, FRCPC Date of revision: February 2014 Headaches occur commonly in children and adolescents. They
1 of 12 17/06/2014 7:57 PM Print Close Neurologic Disorders: Headache in Children Sharon Whiting, MBBS, FRCPC Date of revision: February 2014 Headaches occur commonly in children and adolescents. They
Systematic reviews and meta-analyses of observational studies (MOOSE): Checklist.
 Systematic reviews and meta-analyses of observational studies (MOOSE): Checklist. MOOSE Checklist Infliximab reduces hospitalizations and surgery interventions in patients with inflammatory bowel disease:
Systematic reviews and meta-analyses of observational studies (MOOSE): Checklist. MOOSE Checklist Infliximab reduces hospitalizations and surgery interventions in patients with inflammatory bowel disease:
Headaches in the Pediatric Emergency Dept
 Headaches in Children February 23, 2011 Jinny Tavee, MD Associate Professor Neuromuscular Center Cleveland Clinic Foundation Cleveland, OH 1 Headaches in the Pediatric Emergency Dept Burton Gutierrez Kan
Headaches in Children February 23, 2011 Jinny Tavee, MD Associate Professor Neuromuscular Center Cleveland Clinic Foundation Cleveland, OH 1 Headaches in the Pediatric Emergency Dept Burton Gutierrez Kan
Outpatient Headache Care Guideline
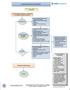 1 Outpatient Care Guideline Inclusion criteria: children > 3 yrs with headaches Is urgent emergency department, neuroimaging, or Neurology consultation indicated? Referral to ED if: New severe headache
1 Outpatient Care Guideline Inclusion criteria: children > 3 yrs with headaches Is urgent emergency department, neuroimaging, or Neurology consultation indicated? Referral to ED if: New severe headache
Headaches in Children and Adolescents. Paul Shillito
 Headaches in Children and Adolescents Paul Shillito Topics For Discussion What s different about childhood migraine Chronic daily headache (CDH) Tumours and other things to worry about Management of childhood
Headaches in Children and Adolescents Paul Shillito Topics For Discussion What s different about childhood migraine Chronic daily headache (CDH) Tumours and other things to worry about Management of childhood
Study No.: Title: Rationale: Phase: Study Period: Study Design: Centres: Indication: Treatment: Objectives: Primary Outcome/Efficacy Variable:
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
Individual Study Table Referring to Part of the Dossier. Page:
 2. SYNOPSIS Title of Study: A comparative study of the pharmacokinetic/pharmacodynamic and safety profiles of extended release, regular release and placebo during a 12 hour observation in post-extraction
2. SYNOPSIS Title of Study: A comparative study of the pharmacokinetic/pharmacodynamic and safety profiles of extended release, regular release and placebo during a 12 hour observation in post-extraction
PROSPERO International prospective register of systematic reviews
 PROSPERO International prospective register of systematic reviews High-dose chemotherapy followed by autologous haematopoietic cell transplantation for children, adolescents and young adults with first
PROSPERO International prospective register of systematic reviews High-dose chemotherapy followed by autologous haematopoietic cell transplantation for children, adolescents and young adults with first
Headache. Karen Thaxter
 Headache Karen Thaxter An eight year old girl is taken to her paediatrician because she has been complaining of almost daily pain at the back of her head for the past 4 months. She states that each headache
Headache Karen Thaxter An eight year old girl is taken to her paediatrician because she has been complaining of almost daily pain at the back of her head for the past 4 months. She states that each headache
Downloaded from:
 Arnup, SJ; Forbes, AB; Kahan, BC; Morgan, KE; McKenzie, JE (2016) The quality of reporting in cluster randomised crossover trials: proposal for reporting items and an assessment of reporting quality. Trials,
Arnup, SJ; Forbes, AB; Kahan, BC; Morgan, KE; McKenzie, JE (2016) The quality of reporting in cluster randomised crossover trials: proposal for reporting items and an assessment of reporting quality. Trials,
Guidance for Industry Migraine: Developing Drugs for Acute Treatment
 Guidance for Industry Migraine: Developing Drugs for Acute Treatment DRAFT GUIDANCE This guidance document is being distributed for comment purposes only. Comments and suggestions regarding this draft
Guidance for Industry Migraine: Developing Drugs for Acute Treatment DRAFT GUIDANCE This guidance document is being distributed for comment purposes only. Comments and suggestions regarding this draft
Chronic Migraine in Primary Care. December 11 th, 2017 Werner J. Becker University of Calgary
 Chronic Migraine in Primary Care December 11 th, 2017 Werner J. Becker University of Calgary Disclosures Faculty: Werner J. Becker Relationships with commercial interests: Grants/Research Support: Clinical
Chronic Migraine in Primary Care December 11 th, 2017 Werner J. Becker University of Calgary Disclosures Faculty: Werner J. Becker Relationships with commercial interests: Grants/Research Support: Clinical
Drug Class Review on Triptans
 Drug Class Review on UPDATED FINAL REPORT #1 December 2003 Mark Helfand, MD, MPH Kim Peterson, MS Oregon Evidence-based Practice Center Oregon Health & Science University Table of Contents Introduction
Drug Class Review on UPDATED FINAL REPORT #1 December 2003 Mark Helfand, MD, MPH Kim Peterson, MS Oregon Evidence-based Practice Center Oregon Health & Science University Table of Contents Introduction
Efficacy of Levetiracetam: A Review of Three Pivotal Clinical Trials
 Epilepsia, 42(Suppl. 4):31 35, 2001 Blackwell Science, Inc. International League Against Epilepsy Efficacy of : A Review of Three Pivotal Clinical Trials Michael Privitera University of Cincinnati Medical
Epilepsia, 42(Suppl. 4):31 35, 2001 Blackwell Science, Inc. International League Against Epilepsy Efficacy of : A Review of Three Pivotal Clinical Trials Michael Privitera University of Cincinnati Medical
6/2/2017. Objectives. Statement of Problem: Migraine Headaches Are Common. Chronic Headache In Pediatrics, Botox and Beyond
 Chronic Headache In Pediatrics, Botox and Beyond Ken Mack MD PhD Mayo Clinic 2015 MFMER slide-1 Objectives Understand pediatric chronic headache presentations Review evidence for the treatment of chronic
Chronic Headache In Pediatrics, Botox and Beyond Ken Mack MD PhD Mayo Clinic 2015 MFMER slide-1 Objectives Understand pediatric chronic headache presentations Review evidence for the treatment of chronic
OVERVIEW OF COMMENTS RECEIVED ON DRAFT GUIDELINE ON CLINICAL INVESTIGATION OF MEDICINAL PRODUCTS FOR THE TREATMENT OF MIGRAINE
 European Medicines Agency London, 24 January 2007 Doc. Ref. EMEA/CHMP/EWP/21477/2007 OVERVIEW OF COMMENTS RECEIVED ON DRAFT GUIDELINE ON CLINICAL INVESTIGATION OF MEDICINAL PRODUCTS FOR THE TREATMENT OF
European Medicines Agency London, 24 January 2007 Doc. Ref. EMEA/CHMP/EWP/21477/2007 OVERVIEW OF COMMENTS RECEIVED ON DRAFT GUIDELINE ON CLINICAL INVESTIGATION OF MEDICINAL PRODUCTS FOR THE TREATMENT OF
Drug Class Review on the Triptans
 Drug Class Review on the Final Report March 7, 2003 Expires October 31, 2003 Mark Helfand, MD, MPH Kim Peterson, MS Produced by Oregon Health & Science University 3181 SW Sam Jackson Park Road Mailcode:
Drug Class Review on the Final Report March 7, 2003 Expires October 31, 2003 Mark Helfand, MD, MPH Kim Peterson, MS Produced by Oregon Health & Science University 3181 SW Sam Jackson Park Road Mailcode:
Cochrane Breast Cancer Group
 Cochrane Breast Cancer Group Version and date: V3.2, September 2013 Intervention Cochrane Protocol checklist for authors This checklist is designed to help you (the authors) complete your Cochrane Protocol.
Cochrane Breast Cancer Group Version and date: V3.2, September 2013 Intervention Cochrane Protocol checklist for authors This checklist is designed to help you (the authors) complete your Cochrane Protocol.
Individual Study Table Referring to Part of the Dossier. Page:
 2. SYNOPSIS Title of Study: A phase III comparative study of the pharmacokinetic/pharmacodynamic and safety profiles of extended release, regular release and placebo during a 12 hour observation in post-extraction
2. SYNOPSIS Title of Study: A phase III comparative study of the pharmacokinetic/pharmacodynamic and safety profiles of extended release, regular release and placebo during a 12 hour observation in post-extraction
MorphiDex (MS:DM) Double-Blind, Multiple-Dose Studies In Chronic Pain Patients
 Vol. 19 No. 1(Suppl.) January 2000 Journal of Pain and Symptom Management S37 Proceedings Supplement NMDA-Receptor Antagonists: Evolving Role in Analgesia MorphiDex (MS:DM) Double-Blind, Multiple-Dose
Vol. 19 No. 1(Suppl.) January 2000 Journal of Pain and Symptom Management S37 Proceedings Supplement NMDA-Receptor Antagonists: Evolving Role in Analgesia MorphiDex (MS:DM) Double-Blind, Multiple-Dose
Pharmacy Medical Necessity Guidelines: Migraine Medications
 Pharmacy Medical Necessity Guidelines: Effective: September 18, 2017 Prior Authorization Required Type of Review Care Management Not Covered Type of Review Clinical Review Pharmacy (RX) or Medical (MED)
Pharmacy Medical Necessity Guidelines: Effective: September 18, 2017 Prior Authorization Required Type of Review Care Management Not Covered Type of Review Clinical Review Pharmacy (RX) or Medical (MED)
Eletriptan vs sumatriptan: A double-blind, placebo-controlled, multiple migraine attack study
 Eletriptan vs sumatriptan: A double-blind, placebo-controlled, multiple migraine attack study G. Sandrini, M. Färkkilä, G. Burgess, et al. Neurology 2002;59;1210-1217 DOI 10.1212/WNL.59.8.1210 This information
Eletriptan vs sumatriptan: A double-blind, placebo-controlled, multiple migraine attack study G. Sandrini, M. Färkkilä, G. Burgess, et al. Neurology 2002;59;1210-1217 DOI 10.1212/WNL.59.8.1210 This information
Drug Class Review on Triptans
 Drug Class Review on Triptans Final Report Update 3 November 2005 Original Report Date: March 2003 Update 1 Report Date: December 2003 Update 2 Report Date: September 2004 A literature scan of this topic
Drug Class Review on Triptans Final Report Update 3 November 2005 Original Report Date: March 2003 Update 1 Report Date: December 2003 Update 2 Report Date: September 2004 A literature scan of this topic
Critical Appraisal Practicum. Fabio Di Bello Medical Implementation Manager
 Critical Appraisal Practicum Fabio Di Bello Medical Implementation Manager fdibello@ebsco.com What we ll talk about today: DynaMed process for appraising randomized trials and writing evidence summaries
Critical Appraisal Practicum Fabio Di Bello Medical Implementation Manager fdibello@ebsco.com What we ll talk about today: DynaMed process for appraising randomized trials and writing evidence summaries
Cinnarizine versus Topiramate in Prophylaxis of Migraines among Children and Adolescents: A Randomized, Double-Blind Clinical Trial
 original ARTICLE Cinnarizine versus Topiramate in Prophylaxis of Migraines among Children and Adolescents: A Randomized, Double-Blind Clinical Trial How to Cite This Article: Ashrafi MR, Najafi Z, Shafiei
original ARTICLE Cinnarizine versus Topiramate in Prophylaxis of Migraines among Children and Adolescents: A Randomized, Double-Blind Clinical Trial How to Cite This Article: Ashrafi MR, Najafi Z, Shafiei
TITLE: Montelukast for Sleep Apnea: A Review of the Clinical Effectiveness, Cost Effectiveness, and Guidelines
 TITLE: Montelukast for Sleep Apnea: A Review of the Clinical Effectiveness, Cost Effectiveness, and Guidelines DATE: 17 January 2014 CONTEXT AND POLICY ISSUES Obstructive sleep apnea (OSA) is a common
TITLE: Montelukast for Sleep Apnea: A Review of the Clinical Effectiveness, Cost Effectiveness, and Guidelines DATE: 17 January 2014 CONTEXT AND POLICY ISSUES Obstructive sleep apnea (OSA) is a common
This information is current as of April 28, 2010
 Practice Parameter: Pharmacological treatment of migraine headache in children and adolescents: Report of the American Academy of Neurology Quality Standards Subcommittee and the Practice Committee of
Practice Parameter: Pharmacological treatment of migraine headache in children and adolescents: Report of the American Academy of Neurology Quality Standards Subcommittee and the Practice Committee of
Guidelines for controlled trials of drugs in migraine: second edition
 Guidelines for controlled trials of drugs in migraine: second edition International Headache Society Clinical Trials Subcommittee. Committee members: P Tfelt-Hansen (Chairman) (Denmark), G Block (USA),
Guidelines for controlled trials of drugs in migraine: second edition International Headache Society Clinical Trials Subcommittee. Committee members: P Tfelt-Hansen (Chairman) (Denmark), G Block (USA),
Migraine Types and Triggering Factors in Children
 original ARTICLE Migraine Types and Triggering Factors in Children How to Cite this Article: Nejad Biglari H, Karimzadeh P, Mohammadi Kord-kheyli M, Hashemi SM. Migraine Types and Triggering Factors in
original ARTICLE Migraine Types and Triggering Factors in Children How to Cite this Article: Nejad Biglari H, Karimzadeh P, Mohammadi Kord-kheyli M, Hashemi SM. Migraine Types and Triggering Factors in
Study No.:MPX Title: Rationale: Phase: IIB Study Period: Study Design: Centres: Indication: Treatment: Objectives:
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
CANADIAN STROKE BEST PRACTICE RECOMMENDATIONS. Stroke Rehabilitation Evidence Tables Rehabilitation to Improve Central Pain
 CANADIAN STROKE BEST PRACTICE RECOMMENDATIONS Rehabilitation to Improve Central Pain Hebert, D, Teasell, R (Writing Group Chairs) on Behalf of the STROKE REHABILITATION Writing Group 2015 December 2015
CANADIAN STROKE BEST PRACTICE RECOMMENDATIONS Rehabilitation to Improve Central Pain Hebert, D, Teasell, R (Writing Group Chairs) on Behalf of the STROKE REHABILITATION Writing Group 2015 December 2015
What A Headache! Theresa Biesiada March 8, 2012
 What A Headache! Theresa Biesiada March 8, 2012 Objectives Describe the EM relevance of headaches and migraines Discuss the rationale for steroid therapy Review the evidence Conclusions My inspiration
What A Headache! Theresa Biesiada March 8, 2012 Objectives Describe the EM relevance of headaches and migraines Discuss the rationale for steroid therapy Review the evidence Conclusions My inspiration
Immediate-release Hydrocodone/Acetaminophen M Abbreviated Clinical Study Report R&D/08/1020
 2.0 Synopsis Abbott Laboratories Individual Study Table Referring to Part of Dossier: (For National Authority Use Only) Name of Study Drug: Volume: Hydrocodone Bitartrate- Acetaminophen (NORCO ) Name of
2.0 Synopsis Abbott Laboratories Individual Study Table Referring to Part of Dossier: (For National Authority Use Only) Name of Study Drug: Volume: Hydrocodone Bitartrate- Acetaminophen (NORCO ) Name of
10/17/2017 CHRONIC MIGRAINES BOTOX: TO INJECT OR NOT INJECT? IN CHRONIC MIGRAINE PROPHYLAXIS OBJECTIVES PATIENT CASE EPIDEMIOLOGY EPIDEMIOLOGY
 BOTOX: TO INJECT OR NOT INJECT? IN CHRONIC MIGRAINE PROPHYLAXIS OBJECTIVES JENNIFER SHIN, PHARMD PGY2 AMBULATORY CARE PHARMACY RESIDENT COMMUNITYCARE HEALTH CENTERS PHARMACOTHERAPY ROUNDS OCTOBER 20, 2017
BOTOX: TO INJECT OR NOT INJECT? IN CHRONIC MIGRAINE PROPHYLAXIS OBJECTIVES JENNIFER SHIN, PHARMD PGY2 AMBULATORY CARE PHARMACY RESIDENT COMMUNITYCARE HEALTH CENTERS PHARMACOTHERAPY ROUNDS OCTOBER 20, 2017
ORIGINAL ARTICLE INTRODUCTION
 ORIGINAL ARTICLE The Efficacy of Propofol vs. Subcutaneous Sumatriptan for Treatment of Acute Migraine Headaches in the Emergency Department: A Double-Blinded Clinical Trial Hossein Moshtaghion, MD*; Najmeh
ORIGINAL ARTICLE The Efficacy of Propofol vs. Subcutaneous Sumatriptan for Treatment of Acute Migraine Headaches in the Emergency Department: A Double-Blinded Clinical Trial Hossein Moshtaghion, MD*; Najmeh
PICC or peripheral intravenous catheter?
 PICC or peripheral intravenous catheter? B.S. Niël-Weise 1, P.J. van den Broek 2 1 Dutch Infection Prevention Working Party, Leiden, The Netherlands 2 Department of Infectious Diseases, Leiden University
PICC or peripheral intravenous catheter? B.S. Niël-Weise 1, P.J. van den Broek 2 1 Dutch Infection Prevention Working Party, Leiden, The Netherlands 2 Department of Infectious Diseases, Leiden University
Inpatient Treatment of Status Migraine With Dihydroergotamine in Children and Adolescents
 Headache 2008 the Authors Journal compilation 2008 American Headache Society ISSN 0017-8748 doi: 10.1111/j.1526-4610.2008.01293.x Published by Wiley Periodicals, Inc. Brief Communication Inpatient Treatment
Headache 2008 the Authors Journal compilation 2008 American Headache Society ISSN 0017-8748 doi: 10.1111/j.1526-4610.2008.01293.x Published by Wiley Periodicals, Inc. Brief Communication Inpatient Treatment
Antidepressants (Tricyclic Antidepressants, Selective Serotonin Reuptake Inhibitors) in children 6-12 years of age with depressive episode/disorder
 updated 2012 Antidepressants (Tricyclic Antidepressants, Selective Serotonin Reuptake Inhibitors) in children 6-12 years of age with depressive episode/disorder Q10: Are antidepressants (Tricyclic antidepressants
updated 2012 Antidepressants (Tricyclic Antidepressants, Selective Serotonin Reuptake Inhibitors) in children 6-12 years of age with depressive episode/disorder Q10: Are antidepressants (Tricyclic antidepressants
Up to date on the use of triptans for child and adolescent migraine: state of the art
 J Headache Pain (2001) 2:S97 S101 Springer-Verlag 2001 THERAPY Dario Calderoni Federica Galli Vincenzo Guidetti Up to date on the use of triptans for child and adolescent migraine: state of the art D.
J Headache Pain (2001) 2:S97 S101 Springer-Verlag 2001 THERAPY Dario Calderoni Federica Galli Vincenzo Guidetti Up to date on the use of triptans for child and adolescent migraine: state of the art D.
Treatment of Headache in the ED
 Treatment of Headache in the ED Benjamin W. Friedman, MD, MS, FAAEM Associate professor of Emergency Medicine Albert Einstein College of Medicine Montefiore Medical Center Disclosure Topics of Discussion
Treatment of Headache in the ED Benjamin W. Friedman, MD, MS, FAAEM Associate professor of Emergency Medicine Albert Einstein College of Medicine Montefiore Medical Center Disclosure Topics of Discussion
Headache Master School Japan-Osaka 2016 (HMSJ-Osaka2016) October 23, II. Management of Refractory Headaches
 Headache Master School Japan-Osaka 2016 (HMSJ-Osaka2016) October 23, 2016 II. Management of Refractory Headaches Case presentation 1: A case of intractable daily-persistent headache Keio University School
Headache Master School Japan-Osaka 2016 (HMSJ-Osaka2016) October 23, 2016 II. Management of Refractory Headaches Case presentation 1: A case of intractable daily-persistent headache Keio University School
Understanding. Migraine. Amy, diagnosed in 1989, with her family.
 Understanding Migraine Amy, diagnosed in 1989, with her family. What Is a Migraine? A migraine is a recurring moderate to severe headache. The pain usually occurs on one side of the head. It is typically
Understanding Migraine Amy, diagnosed in 1989, with her family. What Is a Migraine? A migraine is a recurring moderate to severe headache. The pain usually occurs on one side of the head. It is typically
5-ASA for the treatment of Crohn s disease DR. STEPHEN HANAUER FEINBERG SCHOOL OF MEDICINE, NORTHWESTERN UNIVERSITY, CHICAGO, IL, USA
 5-ASA for the treatment of Crohn s disease DR. STEPHEN HANAUER FEINBERG SCHOOL OF MEDICINE, NORTHWESTERN UNIVERSITY, CHICAGO, IL, USA Background RCTs investigating the efficacy of aminosalicylates for
5-ASA for the treatment of Crohn s disease DR. STEPHEN HANAUER FEINBERG SCHOOL OF MEDICINE, NORTHWESTERN UNIVERSITY, CHICAGO, IL, USA Background RCTs investigating the efficacy of aminosalicylates for
The comparison or control group may be allocated a placebo intervention, an alternative real intervention or no intervention at all.
 1. RANDOMISED CONTROLLED TRIALS (Treatment studies) (Relevant JAMA User s Guides, Numbers IIA & B: references (3,4) Introduction: The most valid study design for assessing the effectiveness (both the benefits
1. RANDOMISED CONTROLLED TRIALS (Treatment studies) (Relevant JAMA User s Guides, Numbers IIA & B: references (3,4) Introduction: The most valid study design for assessing the effectiveness (both the benefits
Influence of blinding on treatment effect size estimate in randomized controlled trials of oral health interventions
 Saltaji et al. BMC Medical Research Methodology (218) 18:42 https://doi.org/1.1186/s12874-18-491- RESEARCH ARTICLE Open Access Influence of blinding on treatment effect size estimate in randomized controlled
Saltaji et al. BMC Medical Research Methodology (218) 18:42 https://doi.org/1.1186/s12874-18-491- RESEARCH ARTICLE Open Access Influence of blinding on treatment effect size estimate in randomized controlled
Value of postmarketing surveillance studies in achieving a complete picture of antimigraine agents: using almotriptan as an example
 J Headache Pain (2006) 7:27 33 DOI 10.1007/s10194-006-0266-6 ORIGINAL Julio Pascual Hans-Christoph Diener Hélène Massiou Value of postmarketing surveillance studies in achieving a complete picture of antimigraine
J Headache Pain (2006) 7:27 33 DOI 10.1007/s10194-006-0266-6 ORIGINAL Julio Pascual Hans-Christoph Diener Hélène Massiou Value of postmarketing surveillance studies in achieving a complete picture of antimigraine
Clinical Trial Results Summary Study EN3409-BUP-305
 Title of Study: A 52-Week, Open-Label, Long-Term Treatment Evaluation of the Safety and Efficacy of BEMA Buprenorphine in Subjects with Moderate to Severe Chronic Pain Coordinating Investigator: Martin
Title of Study: A 52-Week, Open-Label, Long-Term Treatment Evaluation of the Safety and Efficacy of BEMA Buprenorphine in Subjects with Moderate to Severe Chronic Pain Coordinating Investigator: Martin
OBSTETRICS & GYNECOLOGY
 DECEMBER 2011 NORLAND AVENUE PHARMACY PRESCRIPTION COMPOUNDING N ORLANDA VENUEP HARMACY. COM We customize individual prescriptions for the specific needs of our patients. INSIDE THIS ISSUE: Migraine Headache
DECEMBER 2011 NORLAND AVENUE PHARMACY PRESCRIPTION COMPOUNDING N ORLANDA VENUEP HARMACY. COM We customize individual prescriptions for the specific needs of our patients. INSIDE THIS ISSUE: Migraine Headache
Drug Class Review on Newer Antiemetics
 Drug Class Review on Newer Antiemetics Final Report January 2006 A literature scan of this topic is done periodically The purpose of this report is to make available information regarding the comparative
Drug Class Review on Newer Antiemetics Final Report January 2006 A literature scan of this topic is done periodically The purpose of this report is to make available information regarding the comparative
Combined spinalepidural. epidural analgesia in labour (review) By Neda Taghizadeh
 Combined spinalepidural versus epidural analgesia in labour (review) By Neda Taghizadeh Cochrane review Cochrane collaboration was founded in 1993 and is named after Archie Cochrane (1909-1988), British
Combined spinalepidural versus epidural analgesia in labour (review) By Neda Taghizadeh Cochrane review Cochrane collaboration was founded in 1993 and is named after Archie Cochrane (1909-1988), British
PFIZER INC. THERAPEUTIC AREA AND FDA APPROVED INDICATIONS: See United States Package Insert (USPI)
 PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
medications. This was an openlabel study consisting of patients with migraines who historically failed to respond to oral triptan
 J Headache Pain (2007) 8:13 18 DOI 10.1007/s10194-007-0354-7 ORIGINAL Seymour Diamond Fred G. Freitag Alexander Feoktistov George Nissan Sumatriptan 6 mg subcutaneous as an effective migraine treatment
J Headache Pain (2007) 8:13 18 DOI 10.1007/s10194-007-0354-7 ORIGINAL Seymour Diamond Fred G. Freitag Alexander Feoktistov George Nissan Sumatriptan 6 mg subcutaneous as an effective migraine treatment
SYNOPSIS 2/198 CSR_BDY-EFC5825-EN-E02. Name of company: TABULAR FORMAT (For National Authority Use only)
 SYNOPSIS Title of the study: A randomized, double-blind, placebo-controlled, parallel-group, fixed-dose (rimonabant 20 mg) multicenter study of long-term glycemic control with rimonabant in treatment-naïve
SYNOPSIS Title of the study: A randomized, double-blind, placebo-controlled, parallel-group, fixed-dose (rimonabant 20 mg) multicenter study of long-term glycemic control with rimonabant in treatment-naïve
ipad Increasing Nickel Exposure in Children
 ipad Increasing Nickel Exposure in Children abstract We discuss allergic contact dermatitis to the ipad to highlight a potential source of nickel exposure in children. Pediatrics 2014;134:e580 e582 AUTHORS:
ipad Increasing Nickel Exposure in Children abstract We discuss allergic contact dermatitis to the ipad to highlight a potential source of nickel exposure in children. Pediatrics 2014;134:e580 e582 AUTHORS:
Drug Class Literature Scan: Pancreatic Enzymes
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Drug Class Review. Pharmacologic Treatments for Attention Deficit Hyperactivity Disorder
 Drug Class Review Pharmacologic Treatments for Attention Deficit Hyperactivity Disorder Final Update 5 Report July 2015 The purpose of reports is to make available information regarding the comparative
Drug Class Review Pharmacologic Treatments for Attention Deficit Hyperactivity Disorder Final Update 5 Report July 2015 The purpose of reports is to make available information regarding the comparative
Zolmitriptan is effective and well tolerated in Japanese patients with migraine: a dose response study
 is effective and well tolerated in Japanese patients with migraine: a dose response study F Sakai 1, M Iwata 2, K Tashiro 3, Y Itoyama 4, S Tsuji 5, Y Fukuuchi 6, G Sobue 7, K Nakashima 8 & M Morimatsu
is effective and well tolerated in Japanese patients with migraine: a dose response study F Sakai 1, M Iwata 2, K Tashiro 3, Y Itoyama 4, S Tsuji 5, Y Fukuuchi 6, G Sobue 7, K Nakashima 8 & M Morimatsu
NSAIDs and Tonsillectomy: Efficacy for Pain Relief
 NSAIDs and Tonsillectomy: Efficacy for Pain Relief Natasha Cohen, Sarah Lapner, Jayant Ramakrishna, Lehana Thabane, Doron Sommer, Gideon Koren McMaster OTL HNS Resident Research Day October 25 th 2013
NSAIDs and Tonsillectomy: Efficacy for Pain Relief Natasha Cohen, Sarah Lapner, Jayant Ramakrishna, Lehana Thabane, Doron Sommer, Gideon Koren McMaster OTL HNS Resident Research Day October 25 th 2013
2018 American Academy of Neurology
 Practice Guideline Update Efficacy and Tolerability of the New Antiepileptic Drugs II: Treatment-Resistant Epilepsy Report by: Guideline Development, Dissemination, and Implementation Subcommittee of the
Practice Guideline Update Efficacy and Tolerability of the New Antiepileptic Drugs II: Treatment-Resistant Epilepsy Report by: Guideline Development, Dissemination, and Implementation Subcommittee of the
Clinical Study Synopsis
 Clinical Study Synopsis This document is not intended to replace the advice of a healthcare professional and should not be considered as a recommendation. Patients should always seek medical advice before
Clinical Study Synopsis This document is not intended to replace the advice of a healthcare professional and should not be considered as a recommendation. Patients should always seek medical advice before
Disclosures. Objectives 6/2/2017
 Classification: Migraine and Trigeminal Autonomic Cephalalgias Lauren Doyle Strauss, DO, FAHS Assistant Professor, Child Neurology Assistant Director, Child Neurology Residency @StraussHeadache No disclosures
Classification: Migraine and Trigeminal Autonomic Cephalalgias Lauren Doyle Strauss, DO, FAHS Assistant Professor, Child Neurology Assistant Director, Child Neurology Residency @StraussHeadache No disclosures
A Benefit-Risk Assessment of Agomelatine in the Treatment of Major Depression
 REVIEW ARTICLE Drug Saf 2011; 34 (9): 709-731 0114-5916/11/0009-0709/$49.95/0 ª 2011 Adis Data Information BV. All rights reserved. A Benefit-Risk Assessment of Agomelatine in the Treatment of Major Depression
REVIEW ARTICLE Drug Saf 2011; 34 (9): 709-731 0114-5916/11/0009-0709/$49.95/0 ª 2011 Adis Data Information BV. All rights reserved. A Benefit-Risk Assessment of Agomelatine in the Treatment of Major Depression
United Recommended Childhood and Adolescent Immunization Schedule States, 2013
 Recommended Childhood and Adolescent Immunization Schedule United States, 2013 COMMITTEE ON INFECTIOUS DISEASES Pediatrics; originally published online January 28, 2013; DOI: 10.1542/peds.2012-3706 The
Recommended Childhood and Adolescent Immunization Schedule United States, 2013 COMMITTEE ON INFECTIOUS DISEASES Pediatrics; originally published online January 28, 2013; DOI: 10.1542/peds.2012-3706 The
Comparative Efficacy and Safety of Long-Acting Oral Opioids for Chronic Non-Cancer Pain: A Systematic Review
 1026 Journal of Pain and Symptom Management Vol. 26 No. 5 November 2003 Review Article Comparative Efficacy and Safety of Long-Acting Oral Opioids for Chronic Non-Cancer Pain: A Systematic Review Roger
1026 Journal of Pain and Symptom Management Vol. 26 No. 5 November 2003 Review Article Comparative Efficacy and Safety of Long-Acting Oral Opioids for Chronic Non-Cancer Pain: A Systematic Review Roger
Drug Class Review on Newer Drugs for Insomnia
 Drug Class Review on Newer Drugs for Insomnia Final Report July 00 The Agency for Healthcare Research and Quality has not yet seen or approved this report The purpose of this report is to make available
Drug Class Review on Newer Drugs for Insomnia Final Report July 00 The Agency for Healthcare Research and Quality has not yet seen or approved this report The purpose of this report is to make available
TITLE: Fusidic Acid for Ophthalmic Infections: A Review of Clinical and Cost Effectiveness and Safety
 TITLE: Fusidic Acid for Ophthalmic Infections: A Review of Clinical and Cost Effectiveness and Safety DATE: 22 February 2013 CONTEXT AND POLICY ISSUES The antibiotic fusidic acid is available in a 1% suspension
TITLE: Fusidic Acid for Ophthalmic Infections: A Review of Clinical and Cost Effectiveness and Safety DATE: 22 February 2013 CONTEXT AND POLICY ISSUES The antibiotic fusidic acid is available in a 1% suspension
Outline. Understanding Placebo Response in Psychiatry: The Good, The Bad, and The Ugly. Definitions
 Outline Understanding Placebo Response in Psychiatry: The Good, The Bad, and The Ugly Michael E. Thase, MD Professor of Psychiatry Perelman School of Medicine University of Pennsylvania and Philadelphia
Outline Understanding Placebo Response in Psychiatry: The Good, The Bad, and The Ugly Michael E. Thase, MD Professor of Psychiatry Perelman School of Medicine University of Pennsylvania and Philadelphia
Specific features of migraine syndrome in children
 J Headache Pain (2006) 7:206 210 DOI 10.1007/s10194-006-0312-4 RAPID COMMUNICATION Marija Knezevic-Pogancev Specific features of migraine syndrome in children Received: 16 April 2006 Accepted in revised
J Headache Pain (2006) 7:206 210 DOI 10.1007/s10194-006-0312-4 RAPID COMMUNICATION Marija Knezevic-Pogancev Specific features of migraine syndrome in children Received: 16 April 2006 Accepted in revised
Progress in the Control of Childhood Obesity
 William H. Dietz, MD, PhD a, Christina D. Economos, PhD b Two recent reports from the Centers for Disease Control and Prevention and reports from a number of states and municipalities suggest that we are
William H. Dietz, MD, PhD a, Christina D. Economos, PhD b Two recent reports from the Centers for Disease Control and Prevention and reports from a number of states and municipalities suggest that we are
PROSPERO International prospective register of systematic reviews
 PROSPERO International prospective register of systematic reviews Closed reduction methods for acute anterior shoulder dislocation [Cochrane Protocol] Kanthan Theivendran, Raj Thakrar, Subodh Deshmukh,
PROSPERO International prospective register of systematic reviews Closed reduction methods for acute anterior shoulder dislocation [Cochrane Protocol] Kanthan Theivendran, Raj Thakrar, Subodh Deshmukh,
