Topic: Balloon Catheter Dilation for Treatment of Sinusitis Date of Origin: August 8, Section: Surgery Last Reviewed Date: February 2014
|
|
|
- Bartholomew Jenkins
- 5 years ago
- Views:
Transcription
1 Medical Policy Manual Topic: Balloon Catheter Dilation for Treatment of Sinusitis Date of Origin: August 8, 2006 Section: Surgery Last Reviewed Date: February 2014 Policy No: 153 Effective Date: April 1, 2014 IMPORTANT REMINDER Medical Policies are developed to provide guidance for members and providers regarding coverage in accordance with contract terms. Benefit determinations are based in all cases on the applicable contract language. To the extent there may be any conflict between the Medical Policy and contract language, the contract language takes precedence. PLEASE NOTE: Contracts exclude from coverage, among other things, services or procedures that are considered investigational or cosmetic. Providers may bill members for services or procedures that are considered investigational or cosmetic. Providers are encouraged to inform members before rendering such services that the members are likely to be financially responsible for the cost of these services. DESCRIPTION Balloon catheter dilation (also known as balloon sinuplasty) for the treatment of sinusitis involves placement and inflation of a balloon catheter within an obstructed frontal, sphenoid, or maxillary sinus ostium. The balloon catheter is placed using transnasal endoscopy, or a transantral approach may be used for direct access to the maxillary sinus. Inflation of the balloon is intended to enlarge the sinus ostium by compressing mucosa and displacing local bony structures. This technique has been used as an alternative or adjunct to conventional endoscopic sinus surgery which involves surgical excision of the mucosa and bone. When performed in combination with FESS, it is sometimes referred to as a hybrid procedure. Regulatory Status In March 2008, the device Relieva Sinus Balloon Catheter (Acclarent, Menlo Park, CA) was cleared for marketing by the U.S. Food and Drug Administration (FDA) through the 510(k) process. The FDA determined that this device was substantially equivalent to existing devices for use in dilating the sinus ostia and paranasal spaces in adults and maxillary sinus spaces in children. Subsequent devices developed by Acclarent have also been granted 510(k) approval. These include the Relieva Spin Sinus Dilation System, approved in August 2011, and the Relieva Seeker Balloon Sinuplasty System, approved in November SUR153
2 In June 2008, the device, FinESS TM Sinus Treatment (Entellus Medical, Inc, Maple Grove, MN) was cleared for marketing by the FDA through the 510(k) process. The indication noted is to access and treat the maxillary ostia/ethmoid infundibulum in adults using a transantral approach. The bony sinus outflow tracts are remodeled by balloon displacement of adjacent bone and paranasal sinus structures. Two other balloon sinuplasty devices by Entellus Medical, Inc. also received 510(k) approval in August, These are the ENTrigue Sinus Dilation System, and the XprESS Multi-Sinus Dilation Tool. MEDICAL POLICY CRITERIA Use of a catheter-based inflatable device in the treatment of sinusitis is considered investigational. SCIENTIFIC EVIDENCE In order to determine the benefits and harms of balloon catheter dilation as a stand-alone procedure for the treatment of sinusitis, it must be compared with standard functional endoscopic sinus surgery (FESS) which involves excision of ostial tissues. Well-designed prospective comparative studies, preferably randomized controlled trials (RCTs), are needed to compare health outcomes between the two procedures and determine whether balloon dilation is as effective and durable as excision. The most important clinical outcomes to compare for treatment of sinusitis are: Symptom relief Durability of any beneficial effects Adverse event rate and severity Rate and type of reoperations including repeat dilation procedures Literature Appraisal The focus of this evidence review is on systematic reviews, randomized controlled trials, and nonrandomized comparative trials. Technology Assessment and Systematic Reviews A BlueCross BlueShield Association Technology Evaluation Center (TEC) Assessment was completed in 2012 titled Balloon Ostial Dilation for Treatment of Chronic Rhinosinusitis. [1] This Assessment reviewed evidence from one RCT, three non-randomized comparative studies, and nine case series. The following conclusions were made concerning the adequacy of this evidence for determining the effect of balloon sinuplasty on health outcomes: The evidence is insufficient to determine the effect of the technology on health outcomes. One randomized clinical trial comparing balloon sinuplasty to FESS was inadequately powered and did not evaluate differences in outcomes between the two treatments. While most nonrandomized comparative studies of balloon sinuplasty and FESS show no difference in health outcomes between the two treatments, confounding factors may bias the comparison of the two treatments. Several case series show improvement in symptoms of rhinosinusitis over baseline measures, and such 2 SUR153
3 improvement appears durable up to 2 years. Case series do not allow conclusions regarding the comparative efficacy of balloon sinuplasty to FESS. A 2011 Cochrane systematic review on balloon sinuplasty for chronic rhinosinusitis concentrated on RCTs. [2] One small RCT [3] met the inclusion criteria. Patients were randomized to a hybrid approach that included balloon sinuplasty of the affected frontal recess along with traditional FESS of other paranasal sinuses (n=16), or to traditional FESS (n=16). At 12 months follow=up, both groups reported improvements in symptoms, but there were no significant differences between the two groups. The authors of the Cochrane review rated this study as having a low risk for bias for most parameters, but a high risk for bias in reporting of the outcomes. Specifically, symptom scores were not presented systematically and details of statistical testing were not reported. The overall conclusion of this review was that there is no convincing evidence supporting the use of balloon sinuplasty in chronic rhinosinusitis (CRS). In 2010, Batra and colleagues performed a comprehensive review of the literature regarding balloon catheter technology (BCT) in rhinology. [4] The authors noted the following significant study design flaws that prevented the pooling of effectiveness data and precluded conclusions: o The lack of a comparator group significantly limited interpretation of the results or any efficacy claims relative to the FESS paradigm. o The single comparative study [5] was a retrospective study in which the comparison group was not randomized or matched. In addition, the treatment modality was selected by the patients, which was likely to result in biased symptom and satisfaction scores. o It is unclear if the current data can be extrapolated to the general population with sinusitis because the selection criteria in most studies were not clearly defined. o The use of patient-reported symptom improvement was subject to recall bias and could not reliably quantify disease improvement after surgery. o Data on children was difficult to interpret because some patients also had adenoidectomy which is known to have a positive effect on sinusitis in children. The authors reached the following conclusions: The accrued data attests to its safety, whereas the largest published observational cohort studies have demonstrated the ability to achieve ostia patency for up to 2 years. However, because the selection criteria for these studies were not clearly defined, it is unclear if this data can be extrapolated to the general population with chronic rhinosinusitis (CRS). Is BCT superior or equivalent to the existing devices employed in FESS for the management of CRS? Will the use of BCT translate into improvements in patient outcomes, overall health, and/or quality of life? The many unsettled questions will be best answered by prospective randomized trials that directly compare FESS to BCT, or directly compare medical to surgical treatment. Randomized Controlled Trials The REMODEL study was an industry-sponsored RCT that compared balloon ostial dilation as a stand-alone procedure with FESS. [6] A total of 105 patients with recurrent acute sinusitis or chronic sinusitis and failure of medical therapy were randomized to balloon ostial dilation or FESS. Balloon ostial dilation was performed with the Entellus device, which is labeled for a transantral approach. FESS consisted of maxillary antrostomy and uncinectomy with or without 3 SUR153
4 anterior ethmoidectomy. Thirteen patients withdrew consent prior to treatment, 11 in the FESS group (21%) and 2 in the balloon ostial dilation group (4%). The primary outcomes were the change in the Sino-Nasal Outcome Test (SNOT-20) score at 6-month follow-up, and the mean number of débridements performed postoperatively. Secondary outcomes included recovery time, complication rates, and rates of revision surgery. Both superiority and noninferiority analyses were performed on these outcomes. A total of 91 patients were available at 6-month follow-up. The improvement in the SNOT-20 score was 1.67±1.10 in the balloon dilation group and 1.60±0.96 in the FESS arm (p=0.001 for noninferiority). Postoperative débridements were more common in the FESS group compared with balloon dilation (1.2±1.0 vs 0.1±0.6 in the FESS arm, p<0.001 for superiority). Patients in the balloon dilation arm returned to normal daily activities earlier (1.6 days vs 4.8 days, p=0.002 for superiority), and required fewer days of prescription pain medications (0.9 days vs 2.8 days, p=0.002 for superiority). There were no major complications in either group, and 1 patient in each group required revision surgery. This study was likely to have adequate power to detect group differences; however there were some methodologic limitations. The study was unblinded and did not have blinded outcome assessment for the symptom-based outcomes or the secondary clinical outcomes. There was also some evidence of differential dropout, with larger numbers of patients withdrawing from the FESS group following randomization (21% vs 4%). A small RCT was published in 2011 that reported on physiologic outcomes. [7] Twenty patients were randomly assigned to removal of the uncinate process via FESS or balloon sinus ostial dilation as a stand-alone procedure. The main outcome measures were CO 2 concentration in the sinuses and maximum sinus pressure, both intended to be surrogate measures for sinus ventilation. The CO 2 concentration decreased in both study arms to a similar degree. The mean maxillary sinus pressure on inspiration decreased in the FESS group but did not change in the balloon sinus ostial dilation group. Another small RCT was published by Achar et al in [8] This trial enrolled 24 patients with chronic sinusitis who had failed medical therapy and were scheduled for surgery. Patients were randomized to balloon dilation or FESS and followed for a total of 24 weeks. The primary outcome measures were changes in the SNOT-20 score and the saccharine clearance time (SCT) test. Both groups improved significantly on both outcome measures. The degree of improvement was greater for the functional endoscopic dilatation sinus surgery group compared to the FESS group on both the SNOT-20 score (43.8±15.2 vs 29.7±12.3, p<0.03) and on the SCT score (7.5±5.1 vs 3.5±4.3, p=0.03). Adverse events were not reported. Bozdemir et al published a small study of 10 patients with nasal polyposis, in which one side was treated with FESS and the other with balloon sinus ostial dilation. [9] All procedures were performed by the same surgeon, and polypectomy was performed prior to FESS or balloon sinus ostial dilation in all patients. Outcome measures included sinus patency, as measured by computed tomography (CT) scan (Lund-McKay classification) or repeat endoscopy (McKay grading). At 10 days following the procedure, there were improvements in both groups on measures of patency, but there were no differences between groups. Nonrandomized Comparative Trials 4 SUR153
5 A number of nonrandomized studies have been identified which do not allow conclusions concerning the impact of BSD on primary health outcomes compared with FESS. These studies have methodological limitations such as a limited number of patients, [10,11 ] a heterogenous study population, [12] no primary health outcomes reported, [13] limited follow-up, [10-12,14] retrospective study design [14,15,16], or implementation of self-reported questionnaires. [13,15] Retrospective studies are limited by the accuracy of the medical records reviewed or the recall ability of patients when filling out a study questionnaire. In addition there is no randomization or blinding in a retrospective study design and therefore it is difficult to control for bias and confounders. Clinical Practice Guidelines and Position Statements The published position statements noted below are not evidence-based and did not involve systematic reviews and critical appraisals of the scientific literature. Both statements are based on opinion and consensus. American Rhinologic Society (ARS) The current opinion of the ARS is as follows: o Endoscopic balloon dilation technology, alone or in combination with conventional endoscopic techniques, is acceptable and safe for use in the management of sinus disease. o Endoscopic balloon dilation technology is a tool, not a procedure. o As a tool, balloon catheter technology should not be viewed as investigational or experimental. This position was not based on a systematic review of the published evidence. Three references were provided to support this change in the ARS position. One study was a preclinical cadaver study. One was the preliminary study by Brown and colleagues which included only ten patients. [18] The third was the six-month data from the CLEAR study. [19] The opinion that the procedure is acceptable and safe appears to be based primarily on the ARS position that this technique is a not a distinct procedure. American Academy of Otolaryngology-Head and Neck Surgery (AAO-HNS) The AAO-HNS states that sinus ostial dilation (e.g. balloon ostial dilation) is an appropriate therapeutic option for selected patients with sinusitis. This approach may be used alone to dilate a sinus ostium (frontal, maxillary, or sphenoid) or in conjunction with other instruments No references are cited to support this opinion. Summary There is insufficient evidence to determine the impact on health outcomes of balloon sinuplasty as a minimally invasive alternative to functional endoscopic sinus surgery. The role of this technique as a stand-alone procedure in patients with chronic sinus disease remains uncertain. Prospective comparative studies with larger patient populations that include relevant outcomes with sufficient follow-up duration are needed to determine the outcomes for this treatment compared with surgical alternatives. Relevant outcomes data include symptom improvement, the durability of any treatment effects, and the need for subsequent revision. In addition, more information is needed to determine which patients might be 5 SUR153
6 treated with the balloon technique and which require the more standard approaches. Therefore, balloon sinuplasty as a stand-alone treatment of any sinus is considered investigational. REFERENCES 1. TEC Assessment "Balloon Ostial Dilation for Treatment of Chronic Rhinosinusitis." BlueCross BlueShield Association Technology Evaluation Center, Vol. 27, Tab Ahmed, J, Pal, S, Hopkins, C, Jayaraj, S. Functional endoscopic balloon dilation of sinus ostia for chronic rhinosinusitis. Cochrane Database Syst Rev. 2011(7):CD PMID: Plaza, G, Eisenberg, G, Montojo, J, Onrubia, T, Urbasos, M, O'Connor, C. Balloon dilation of the frontal recess: a randomized clinical trial. Ann Otol Rhinol Laryngol Aug;120(8): PMID: Batra, PS, Ryan, MW, Sindwani, R, Marple, BF. Balloon catheter technology in rhinology: Reviewing the evidence. Laryngoscope Jan;121(1): PMID: Friedman, M, Schalch, P, Lin, HC, Mazloom, N, Neidich, M, Joseph, NJ. Functional endoscopic dilatation of the sinuses: patient satisfaction, postoperative pain, and cost. Am J Rhinol Mar-Apr;22(2): PMID: Cutler, J, Bikhazi, N, Light, J, Truitt, T, Schwartz, M, Investigators, AT. Standalone balloon dilation versus sinus surgery for chronic rhinosinusitis: A prospective, multicenter, randomized, controlled trial. Am J Rhinol Allergy Aug 5. PMID: Kutluhan, A, Salviz, M, Bozdemir, K, et al. The effects of uncinectomy and natural ostial dilatation on maxillary sinus ventilation: a clinical experimental study. Eur Arch Otorhinolaryngol Apr;268(4): PMID: Achar, P, Duvvi, S, Kumar, BN. Endoscopic dilatation sinus surgery (FEDS) versus functional endoscopic sinus surgery (FESS) for treatment of chronic rhinosinusitis: a pilot study. Acta otorhinolaryngologica Italica : organo ufficiale della Societa italiana di otorinolaringologia e chirurgia cervico-facciale Oct;32(5): PMID: Bozdemir, K, Kutluhan, A, Cetin, H, Yalciner, G, Bilgen, AS. Comparison of outcomes of simple polypectomy plus balloon catheter dilatation versus functional endoscopic sinus surgery in nasal polyposis: a preliminary study. Am J Rhinol Allergy May-Jun;25(3): PMID: Albritton, FDt, Casiano, RR, Sillers, MJ. Feasibility of in-office endoscopic sinus surgery with balloon sinus dilation. Am J Rhinol Allergy May-Jun;26(3): PMID: Stankiewicz, J, Truitt, T, Atkins, J, Jr. One-year results: Transantral balloon dilation of the ethmoid infundibulum. Ear Nose Throat J Feb;89(2):72-7. PMID: Stankiewicz, J, Tami, T, Truitt, T, Atkins, J, Liepert, D, Winegar, B. Transantral, endoscopically guided balloon dilatation of the ostiomeatal complex for chronic rhinosinusitis under local anesthesia. Am J Rhinol Allergy May-Jun;23(3): PMID: Stankiewicz, J, Tami, T, Truitt, T, et al. Impact of chronic rhinosinusitis on work productivity through one-year follow-up after balloon dilation of the ethmoid infundibulum. International forum of allergy & rhinology Jan-Feb;1(1): PMID: Levine, HL. Functional endoscopic sinus surgery: evaluation, surgery, and follow-up of 250 patients. Laryngoscope Jan;100(1): PMID: Koskinen, A, Penttila, M, Myller, J, et al. Endoscopic sinus surgery might reduce exacerbations and symptoms more than balloon sinuplasty. Am J Rhinol Allergy Nov-Dec;26(6):e PMID: Matthews, BL, Smith, LE, Jones, R, Miller, C, Brookschmidt, JK. Endoscopic sinus surgery: outcome in 155 cases. Otolaryngol Head Neck Surg Feb;104(2): PMID: SUR153
7 17. American Rhinologic Society (ARS) Position Statement. [cited 03/17/2014]; Available from: Brown, CL, Bolger, WE. Safety and feasibility of balloon catheter dilation of paranasal sinus ostia: a preliminary investigation. Ann Otol Rhinol Laryngol Apr;115(4):293-9; discussion PMID: Bolger, WE, Brown, CL, Church, CA, et al. Safety and outcomes of balloon catheter sinusotomy: a multicenter 24-week analysis in 115 patients. Otolaryngol Head Neck Surg Jul;137(1): PMID: American Academy of Otolaryngology-Head and Neck Surgery. Statement on Balloon Dilation. [cited 03/17/2014]; Available from: CROSS REFERENCES None CODES NUMBER DESCRIPTION CPT Nasal/sinus endoscopy, surgical; with dilation of maxillary sinus ostium (eg, balloon dilation), transnasal or via canine fossa (Do not report in conjunction with 31233, 31256, and when performed on the same sinus) Nasal/sinus endoscopy, surgical; with dilation of frontal sinus ostium (eg, balloon dilation) (Do not report in conjunction with 31233, 31256, and when performed on the same sinus) Nasal/sinus endoscopy, surgical; with dilation of sphenoid sinus ostium (eg, balloon dilation) (Do not report in conjunction with 31233, 31256, and when performed on the same sinus) HCPCS None 7 SUR153
Description. Section: Surgery Effective Date: April 15, 2014 Subsection: Surgery Original Policy Date: June 7, 2012 Subject: Page: 1 of 12
 Last Review Status/Date: March 2014 Page: 1 of 12 Description Balloon ostial dilation (also known as balloon sinuplasty ) is proposed as an alternative to traditional endoscopic sinus surgery for patients
Last Review Status/Date: March 2014 Page: 1 of 12 Description Balloon ostial dilation (also known as balloon sinuplasty ) is proposed as an alternative to traditional endoscopic sinus surgery for patients
Protocol. Balloon Ostial Dilation for Treatment of Chronic Rhinosinusitis
 Protocol Balloon Ostial Dilation for Treatment of Chronic Rhinosinusitis Medical Benefit Effective Date: 04/01/15 Next Review Date: 11/19 Preauthorization No Review Dates: 07/07, 07/08, 11/08, 05/09, 01/10,
Protocol Balloon Ostial Dilation for Treatment of Chronic Rhinosinusitis Medical Benefit Effective Date: 04/01/15 Next Review Date: 11/19 Preauthorization No Review Dates: 07/07, 07/08, 11/08, 05/09, 01/10,
Description. Section: Surgery Effective Date: April 15, 2017 Subsection: Surgery Original Policy Date: June 7, 2012 Subject: Page: 1 of 13
 Last Review Status/Date: March 2017 Page: 1 of 13 Description Balloon ostial dilation (also known as balloon sinuplasty ) is proposed as an alternative to traditional endoscopic sinus surgery for patients
Last Review Status/Date: March 2017 Page: 1 of 13 Description Balloon ostial dilation (also known as balloon sinuplasty ) is proposed as an alternative to traditional endoscopic sinus surgery for patients
MEDICAL POLICY EFFECTIVE DATE: 04/21/11 REVISED DATE: 06/21/12, 05/23/13, 06/19/14, 06/18/15, 06/16/16, 06/15/17
 MEDICAL POLICY SUBJECT: SINUS OSTIAL DILATION FOR PAGE: 1 OF: 6 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial product, including
MEDICAL POLICY SUBJECT: SINUS OSTIAL DILATION FOR PAGE: 1 OF: 6 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial product, including
Balloon Ostial Dilation for Treatment of Chronic Rhinosinusitis
 Balloon Ostial Dilation for Treatment of Chronic Rhinosinusitis Policy Number: 7.01.105 Last Review: 3/2018 Origination: 3/2007 Next Review: 9/2018 Policy Blue Cross and Blue Shield of Kansas City (Blue
Balloon Ostial Dilation for Treatment of Chronic Rhinosinusitis Policy Number: 7.01.105 Last Review: 3/2018 Origination: 3/2007 Next Review: 9/2018 Policy Blue Cross and Blue Shield of Kansas City (Blue
Medicare Advantage Medical Policy. Title: Balloon Ostial Dilation for Treatment of Chronic Rhinosinusitis
 Medicare Advantage Medical Policy Current Policy Effective Date: 1/1/18 Title: Balloon Ostial Dilation for Treatment of Chronic Rhinosinusitis Description/Background CHRONIC RHINOSINUSITIS Chronic rhinosinusitis
Medicare Advantage Medical Policy Current Policy Effective Date: 1/1/18 Title: Balloon Ostial Dilation for Treatment of Chronic Rhinosinusitis Description/Background CHRONIC RHINOSINUSITIS Chronic rhinosinusitis
Medical Policy An independent licensee of the Blue Cross Blue Shield Association
 Balloon Sinuplasty for Treatment of Chronic Sinusitis Page 1 of 17 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Balloon Sinuplasty for Treatment of Chronic Sinusitis
Balloon Sinuplasty for Treatment of Chronic Sinusitis Page 1 of 17 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Balloon Sinuplasty for Treatment of Chronic Sinusitis
Medical Policy An independent licensee of the Blue Cross Blue Shield Association
 Balloon Sinuplasty for Treatment of Chronic Sinusitis Page 1 of 18 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Balloon Sinuplasty for Treatment of Chronic Sinusitis
Balloon Sinuplasty for Treatment of Chronic Sinusitis Page 1 of 18 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Balloon Sinuplasty for Treatment of Chronic Sinusitis
Patency of Maxillary Sinus Ostia Following Dilation with a Novel Osmotic Expansion Device
 WHITE PAPER Patency of Maxillary Sinus Ostia Following Dilation with a Novel Osmotic Expansion Device Six-Month Results from a Multi-Center Prospective Study Jerome Hester, MD California Sleep Institute,
WHITE PAPER Patency of Maxillary Sinus Ostia Following Dilation with a Novel Osmotic Expansion Device Six-Month Results from a Multi-Center Prospective Study Jerome Hester, MD California Sleep Institute,
FRONTAL SINUPLASTY P R E P A R E D A N D P R E S E N T E D B Y : D R. Y A H Y A F A G E E H R 4 16/ 12/ 2013
 FRONTAL SINUPLASTY P R E P A R E D A N D P R E S E N T E D B Y : D R. Y A H Y A F A G E E H R 4 16/ 12/ 2013 ANATOMY: FRONTAL SINUS Not present at birth Starts developing at 4 years Radiographically visualized
FRONTAL SINUPLASTY P R E P A R E D A N D P R E S E N T E D B Y : D R. Y A H Y A F A G E E H R 4 16/ 12/ 2013 ANATOMY: FRONTAL SINUS Not present at birth Starts developing at 4 years Radiographically visualized
Balloon Sinuplasty: Indications and Cost-Effectiveness
 Balloon Sinuplasty: Indications and Cost-Effectiveness Marilene B. Wang, MD Professor, Department of Head and Neck Surgery David Geffen School of Medicine at UCLA Chief of Otolaryngology VA Greater Los
Balloon Sinuplasty: Indications and Cost-Effectiveness Marilene B. Wang, MD Professor, Department of Head and Neck Surgery David Geffen School of Medicine at UCLA Chief of Otolaryngology VA Greater Los
Chronic rhinosinusitis (CRS) is a significant health
 ORIGINAL ARTICLE The safety and performance of a maxillary sinus ostium self-dilation device: a pilot study Iain Hathorn, MBChB, FRCSEd (ORL-HNS), Al-Rahim Habib, MSc, Rachelle Dar Santos, BSc, CCRP and
ORIGINAL ARTICLE The safety and performance of a maxillary sinus ostium self-dilation device: a pilot study Iain Hathorn, MBChB, FRCSEd (ORL-HNS), Al-Rahim Habib, MSc, Rachelle Dar Santos, BSc, CCRP and
BALLOON OSTIAL DILATION FOR TREATMENT OF CHRONIC SINUSITIS
 BALLOON OSTIAL DILATION FOR TREATMENT OF CHRONIC SINUSITIS Non-Discrimination Statement and Multi-Language Interpreter Services information are located at the end of this document. Coverage for services,
BALLOON OSTIAL DILATION FOR TREATMENT OF CHRONIC SINUSITIS Non-Discrimination Statement and Multi-Language Interpreter Services information are located at the end of this document. Coverage for services,
Implantable Sinus Stents for Postoperative Use Following Endoscopic Sinus Surgery and for Recurrent Sinonasal Polyposis
 Medical Policy Manual Surgery, Policy No. 198 Implantable Sinus Stents for Postoperative Use Following Endoscopic Sinus Surgery and for Recurrent Sinonasal Polyposis Next Review: August 2019 Last Review:
Medical Policy Manual Surgery, Policy No. 198 Implantable Sinus Stents for Postoperative Use Following Endoscopic Sinus Surgery and for Recurrent Sinonasal Polyposis Next Review: August 2019 Last Review:
Learner Objectives. Overview. Minimally Invasive Sinus Surgery: What is the data for Balloon Catheter Dilation 7/31/2017
 Minimally Invasive Sinus Surgery: What is the data for Balloon Catheter Dilation David M. Poetker, MD Christopher M. Long, MD Rhinology and Sinus Surgery Medical College of Wisconsin Thank you to Susan
Minimally Invasive Sinus Surgery: What is the data for Balloon Catheter Dilation David M. Poetker, MD Christopher M. Long, MD Rhinology and Sinus Surgery Medical College of Wisconsin Thank you to Susan
Clinical Policy: Balloon Sinus Ostial Dilation for Treatment of Chronic Sinusitis
 Clinical Policy: Balloon Sinus Ostial Dilation for Treatment of Chronic Sinusitis Reference Number: CP.MP.119 Last Review Date: 11/17 See Important Reminder at the end of this policy for important regulatory
Clinical Policy: Balloon Sinus Ostial Dilation for Treatment of Chronic Sinusitis Reference Number: CP.MP.119 Last Review Date: 11/17 See Important Reminder at the end of this policy for important regulatory
The ORIOS 2 Study: Office-Based Balloon Sinus Dilation A prospective multi-center study of 203 patients
 The ORIOS 2 Study: Office-Based Balloon Sinus Dilation A prospective multi-center study of 203 patients Boris Karanfilov, MD, Ohio Sinus Institute, Dublin, OH 1 Stacey Silvers, MD, Madison ENT & Facial
The ORIOS 2 Study: Office-Based Balloon Sinus Dilation A prospective multi-center study of 203 patients Boris Karanfilov, MD, Ohio Sinus Institute, Dublin, OH 1 Stacey Silvers, MD, Madison ENT & Facial
BALLOON SINUS OSTIAL DILATION
 BALLOON SINUS OSTIAL DILATION UnitedHealthcare Oxford Clinical Policy Policy Number: ENT 021.6 T2 Effective Date: April 1, 2018 Table of Contents Page INSTRUCTIONS FOR USE... 1 CONDITIONS OF COVERAGE...
BALLOON SINUS OSTIAL DILATION UnitedHealthcare Oxford Clinical Policy Policy Number: ENT 021.6 T2 Effective Date: April 1, 2018 Table of Contents Page INSTRUCTIONS FOR USE... 1 CONDITIONS OF COVERAGE...
In-office, multisinus balloon dilation: 1-Year outcomes from a prospective, multicenter, open label trial DO NOT COPY METHODS.
 In-office, multisinus balloon dilation: 1-Year outcomes from a prospective, multicenter, open label trial James Gould, M.D., 1 Ian Alexander, M.D., 2 Edward Tomkin, D.O., 3 and David Brodner, M.D. 4 ABSTRACT
In-office, multisinus balloon dilation: 1-Year outcomes from a prospective, multicenter, open label trial James Gould, M.D., 1 Ian Alexander, M.D., 2 Edward Tomkin, D.O., 3 and David Brodner, M.D. 4 ABSTRACT
BALLOON SINUS OSTIAL DILATION
 UnitedHealthcare of California (HMO) UnitedHealthcare Benefits Plan of California (EPO/POS) UnitedHealthcare of Oklahoma, Inc. UnitedHealthcare of Oregon, Inc. UnitedHealthcare Benefits of Texas, Inc.
UnitedHealthcare of California (HMO) UnitedHealthcare Benefits Plan of California (EPO/POS) UnitedHealthcare of Oklahoma, Inc. UnitedHealthcare of Oregon, Inc. UnitedHealthcare Benefits of Texas, Inc.
White Paper: Balloon Sinuplasty for Chronic Sinusitis, The Latest Recommendations
 White Paper: Balloon Sinuplasty for Chronic Sinusitis, The Latest Recommendations For Health Plans, Medical Management Organizations and TPAs Executive Summary Despite recent advances in instrumentation
White Paper: Balloon Sinuplasty for Chronic Sinusitis, The Latest Recommendations For Health Plans, Medical Management Organizations and TPAs Executive Summary Despite recent advances in instrumentation
Site of Service - Select Surgical Procedures. Select a hyperlink below to be directed to that section.
 MEDICAL POLICY 7.01.559 Sinus Surgery BCBSA Ref. Policies: 7.01.105, 7.01.155 Effective Date: June 1, 2018* RELATED MEDICAL POLICIES: Last Revised: May 1, 2018 7.01.558 Rhinoplasty Replaces: N/A 11.01.524
MEDICAL POLICY 7.01.559 Sinus Surgery BCBSA Ref. Policies: 7.01.105, 7.01.155 Effective Date: June 1, 2018* RELATED MEDICAL POLICIES: Last Revised: May 1, 2018 7.01.558 Rhinoplasty Replaces: N/A 11.01.524
Treatment Options for Chronic Sinusitis
 Treatment Options for Chronic Sinusitis Jesse Ryan, M.D. Assistant Professor Head and Neck Surgery & Reconstruction Department of Otolaryngology January 17, 2019 Disclosures I have no financial relationship
Treatment Options for Chronic Sinusitis Jesse Ryan, M.D. Assistant Professor Head and Neck Surgery & Reconstruction Department of Otolaryngology January 17, 2019 Disclosures I have no financial relationship
Sinusitis is a highly prevalent condition, affecting approximately
 ORIGINAL ARTICLE Office-based balloon sinus dilation: a prospective, multicenter study of 203 patients Boris Karanfilov, MD 1, Stacey Silvers, MD 2, Raza Pasha, MD 3, Ashley Sikand, MD 4, Alan Shikani,
ORIGINAL ARTICLE Office-based balloon sinus dilation: a prospective, multicenter study of 203 patients Boris Karanfilov, MD 1, Stacey Silvers, MD 2, Raza Pasha, MD 3, Ashley Sikand, MD 4, Alan Shikani,
Sinus Surgery (Functional Endoscopic Sinus Surgery and Balloon Sinuplasty)
 Sinus Surgery (Functional Endoscopic Sinus Surgery and Balloon Sinuplasty) Date of Origin: 10/2015 Last Review Date: 06/28/2017 Effective Date: 07/01/2017 Dates Reviewed: 10/2015, 06/2017 Developed By:
Sinus Surgery (Functional Endoscopic Sinus Surgery and Balloon Sinuplasty) Date of Origin: 10/2015 Last Review Date: 06/28/2017 Effective Date: 07/01/2017 Dates Reviewed: 10/2015, 06/2017 Developed By:
Site of Service: Select Surgical Procedures. Select a hyperlink below to be directed to that section.
 MEDICAL POLICY 7.01.559 Sinus Surgery BCBSA Ref. Policies: 7.01.105, 7.01.155 Effective Date: June 1, 2018 RELATED MEDICAL POLICIES: Last Revised: Sept. 21, 2018 7.01.558 Rhinoplasty Replaces: N/A 11.01.524
MEDICAL POLICY 7.01.559 Sinus Surgery BCBSA Ref. Policies: 7.01.105, 7.01.155 Effective Date: June 1, 2018 RELATED MEDICAL POLICIES: Last Revised: Sept. 21, 2018 7.01.558 Rhinoplasty Replaces: N/A 11.01.524
Protocol. Implantable Sinus Stents for Postoperative Use Following Endoscopic Sinus Surgery and for Recurrent Sinus Disease
 (701134) Medical Benefit Effective Date: 04/01/17 Next Review Date: 01/19 Preauthorization No Review Dates: 01/13, 01/14, 01/15, 01/16, 01/17, 01/18 This protocol considers this test or procedure investigational.
(701134) Medical Benefit Effective Date: 04/01/17 Next Review Date: 01/19 Preauthorization No Review Dates: 01/13, 01/14, 01/15, 01/16, 01/17, 01/18 This protocol considers this test or procedure investigational.
Implantable Sinus Stents for Postoperative Use Following Endoscopic Sinus Surgery
 Last Review Status/Date: March 2014 Page: 1 of 7 Use Following Endoscopic Sinus Surgery Description Sinus stents are devices that are used postoperatively following endoscopic sinus surgery (ESS). The
Last Review Status/Date: March 2014 Page: 1 of 7 Use Following Endoscopic Sinus Surgery Description Sinus stents are devices that are used postoperatively following endoscopic sinus surgery (ESS). The
Implantable Sinus Stents for Postoperative Use Following Endoscopic Sinus Surgery
 Page: 1 of 8 Last Review Status/Date: March 2015 Use Following Endoscopic Sinus Surgery Description Sinus stents are devices that are used postoperatively following endoscopic sinus surgery (ESS). The
Page: 1 of 8 Last Review Status/Date: March 2015 Use Following Endoscopic Sinus Surgery Description Sinus stents are devices that are used postoperatively following endoscopic sinus surgery (ESS). The
Implantable Sinus Stents for Postoperative Use Following Endoscopic Sinus Surgery and for Recurrent Sinus Disease
 Page: 1 of 8 Last Review Status/Date: March 2017 Implantable Sinus Stents for Postoperative Use Following Endoscopic Sinus Surgery and Description Sinus stents are devices used postoperatively following
Page: 1 of 8 Last Review Status/Date: March 2017 Implantable Sinus Stents for Postoperative Use Following Endoscopic Sinus Surgery and Description Sinus stents are devices used postoperatively following
MP Implantable Sinus Stents for Postoperative Use Following Endoscopic Sinus Surgery and for Recurrent Sinus Disease. Related Policies None
 Medical Policy MP 7.01.134 Implantable Sinus Stents for Postoperative Use Following Endoscopic Sinus Surgery and for Recurrent Sinus BCBSA Ref. Policy: 7.01.134 Last Review: 02/26/2018 Effective Date:
Medical Policy MP 7.01.134 Implantable Sinus Stents for Postoperative Use Following Endoscopic Sinus Surgery and for Recurrent Sinus BCBSA Ref. Policy: 7.01.134 Last Review: 02/26/2018 Effective Date:
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE
 NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Medical technology guidance SCOPE The XprESS Multi-Sinus Dilation System for the treatment of 1 Technology 1.1 Description of the technology The XprESS
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Medical technology guidance SCOPE The XprESS Multi-Sinus Dilation System for the treatment of 1 Technology 1.1 Description of the technology The XprESS
Corporate Medical Policy
 Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: surgical_treatment_of_sinus_disease 2/2010 8/2017 8/2018 8/2017 Description of Procedure or Service Chronic
Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: surgical_treatment_of_sinus_disease 2/2010 8/2017 8/2018 8/2017 Description of Procedure or Service Chronic
Corporate Medical Policy
 Corporate Medical Policy Surgical Treatment of Sinus Disease Description of Procedure or Service Sinusitis refers to infection or inflammation of the sinuses, which are small openings in the bones of the
Corporate Medical Policy Surgical Treatment of Sinus Disease Description of Procedure or Service Sinusitis refers to infection or inflammation of the sinuses, which are small openings in the bones of the
ORIGINAL ARTICLE. Computed Tomographic Staging and the Fate of the Dependent Sinuses in Revision Endoscopic Sinus Surgery
 Computed Tomographic Staging and the Fate of the Dependent es in Revision Endoscopic Surgery Neil Bhattacharyya, MD ORIGINAL ARTICLE Objectives: To determine the patterns of disease recurrence in chronic
Computed Tomographic Staging and the Fate of the Dependent es in Revision Endoscopic Surgery Neil Bhattacharyya, MD ORIGINAL ARTICLE Objectives: To determine the patterns of disease recurrence in chronic
Implantable Sinus Stents for Postoperative Use Following Endoscopic Sinus Surgery and for Recurrent Sinus Disease
 Implantable Sinus Stents for Postoperative Use Following Endoscopic Sinus Surgery and for Recurrent Sinus Disease Policy Number: 7.01.134 Last Review: 8/2018 Origination: 12/2015 Next Review: 12/2018 Policy
Implantable Sinus Stents for Postoperative Use Following Endoscopic Sinus Surgery and for Recurrent Sinus Disease Policy Number: 7.01.134 Last Review: 8/2018 Origination: 12/2015 Next Review: 12/2018 Policy
Medical technologies guidance Published: 13 December 2016 nice.org.uk/guidance/mtg30
 XprESS multi sinus dilation system for treating chronic sinusitis Medical technologies guidance Published: 13 December 2016 nice.org.uk/guidance/mtg30 NICE 2018. All rights reserved. Subject to Notice
XprESS multi sinus dilation system for treating chronic sinusitis Medical technologies guidance Published: 13 December 2016 nice.org.uk/guidance/mtg30 NICE 2018. All rights reserved. Subject to Notice
FUNCTIONAL ENDOSCOPIC SINUS SURGERY (FESS)
 FUNCTIONAL ENDOSCOPIC SINUS SURGERY (FESS) Protocol: SUR055 Effective Date: June 1, 2018 Table of Contents Page COMMERCIAL, MEDICAID AND MEDICARE COVERAGE RATIONALE... 1 DEFINITIONS... 2 DESCRIPTION OF
FUNCTIONAL ENDOSCOPIC SINUS SURGERY (FESS) Protocol: SUR055 Effective Date: June 1, 2018 Table of Contents Page COMMERCIAL, MEDICAID AND MEDICARE COVERAGE RATIONALE... 1 DEFINITIONS... 2 DESCRIPTION OF
FUNCTIONAL ENDOSCOPIC SINUS SURGERY (FESS)
 UnitedHealthcare Community Plan Medical Policy FUNCTIONAL ENDOSCOPIC SINUS SURGERY (FESS) Policy Number: CS144.D Effective Date: January 1, 2018 Table of Contents Page INSTRUCTIONS FOR USE... 1 BENEFIT
UnitedHealthcare Community Plan Medical Policy FUNCTIONAL ENDOSCOPIC SINUS SURGERY (FESS) Policy Number: CS144.D Effective Date: January 1, 2018 Table of Contents Page INSTRUCTIONS FOR USE... 1 BENEFIT
2017 FACILITY AND PHYSICIAN REIMBURSEMENT GUIDE
 2017 AND PHYSICIAN REIMBURSEMENT GUIDE NASAL/SINUS ENDOSCOPIC SURGERY Some of the Current Procedure Terminology (CPT ) Codes for endoscopic nasal/sinus surgery are listed below. CPT codes 31295, 31296
2017 AND PHYSICIAN REIMBURSEMENT GUIDE NASAL/SINUS ENDOSCOPIC SURGERY Some of the Current Procedure Terminology (CPT ) Codes for endoscopic nasal/sinus surgery are listed below. CPT codes 31295, 31296
Evaluation of the Change in Recent Diagnostic Criteria of Chronic Rhinosinusitis: A Cross-sectional Study
 Abhishek Ramadhin REVIEW ARTICLE 10.5005/jp-journals-10013-1246 Evaluation of the Change in Recent Diagnostic Criteria of Chronic Rhinosinusitis: A Cross-sectional Study Abhishek Ramadhin ABSTRACT There
Abhishek Ramadhin REVIEW ARTICLE 10.5005/jp-journals-10013-1246 Evaluation of the Change in Recent Diagnostic Criteria of Chronic Rhinosinusitis: A Cross-sectional Study Abhishek Ramadhin ABSTRACT There
Steroid-Eluting Ethmoidal Stent Versus Antero- -Posterior Ethmoidectomy: Comparison Of Efficacy And Safety In Allergic Patients
 Steroid-Eluting Ethmoidal Stent Versus Antero- -Posterior Ethmoidectomy: Comparison Of Efficacy And Safety In Allergic Patients Authors Contribution: A Study Design B Data Collection C Statistical Analysis
Steroid-Eluting Ethmoidal Stent Versus Antero- -Posterior Ethmoidectomy: Comparison Of Efficacy And Safety In Allergic Patients Authors Contribution: A Study Design B Data Collection C Statistical Analysis
Sphenopalatine Ganglion Block for Headache and Pain
 Medical Policy Manual Medicine, Policy No. 160 Sphenopalatine Ganglion Block for Headache and Pain Next Review: May 2019 Last Review: January 2019 Effective: February 1, 2019 IMPORTANT REMINDER Medical
Medical Policy Manual Medicine, Policy No. 160 Sphenopalatine Ganglion Block for Headache and Pain Next Review: May 2019 Last Review: January 2019 Effective: February 1, 2019 IMPORTANT REMINDER Medical
POLICY PRODUCT VARIATIONS DESCRIPTION/BACKGROUND RATIONALE DEFINITIONS BENEFIT VARIATIONS DISCLAIMER CODING INFORMATION REFERENCES POLICY HISTORY
 Original Issue Date (Created): July 30, 2013 Most Recent Review Date (Revised): May 20, 2014 Effective Date: August 1, 2014 POLICY PRODUCT VARIATIONS DESCRIPTION/BACKGROUND RATIONALE DEFINITIONS BENEFIT
Original Issue Date (Created): July 30, 2013 Most Recent Review Date (Revised): May 20, 2014 Effective Date: August 1, 2014 POLICY PRODUCT VARIATIONS DESCRIPTION/BACKGROUND RATIONALE DEFINITIONS BENEFIT
Section: Durable Medical Equipment Last Reviewed Date: June Policy No: 82 Effective Date: September 1, 2014
 Medical Policy Manual Topic: Bioimpedance Devices for Detection and Management of Lymphedema Date of Origin: April 2011 Section: Durable Medical Equipment Last Reviewed Date: June 2014 Policy No: 82 Effective
Medical Policy Manual Topic: Bioimpedance Devices for Detection and Management of Lymphedema Date of Origin: April 2011 Section: Durable Medical Equipment Last Reviewed Date: June 2014 Policy No: 82 Effective
MEDICAL POLICY I. POLICY FUNCTIONAL ENDOSCOPIC SINUS SURGERY FOR CHRONIC RHINOSINUSITIS MP POLICY TITLE POLICY NUMBER
 Original Issue Date (Created): 8/1/2018 Most Recent Review Date (Revised): 1/30/2018 Effective Date: 8/1/2018 POLICY PRODUCT VARIATIONS DESCRIPTION/BACKGROUND RATIONALE DEFINITIONS BENEFIT VARIATIONS DISCLAIMER
Original Issue Date (Created): 8/1/2018 Most Recent Review Date (Revised): 1/30/2018 Effective Date: 8/1/2018 POLICY PRODUCT VARIATIONS DESCRIPTION/BACKGROUND RATIONALE DEFINITIONS BENEFIT VARIATIONS DISCLAIMER
Dose-dependent effects of tobramycin in an animal model of Pseudomonas sinusitis Am J Rhino Jul-Aug; 21(4):423-7
 AMINOGLYCOSIDES Dose-dependent effects of tobramycin in an animal model of Pseudomonas sinusitis Am J Rhino. 2007 Jul-Aug; 21(4):423-7 http://www.ncbi.nlm.nih.gov/pubmed/17882910 Evaluation of the in-vivo
AMINOGLYCOSIDES Dose-dependent effects of tobramycin in an animal model of Pseudomonas sinusitis Am J Rhino. 2007 Jul-Aug; 21(4):423-7 http://www.ncbi.nlm.nih.gov/pubmed/17882910 Evaluation of the in-vivo
An Innovative Treatment Option for Patients with Recurrent Nasal Polyps
 An Innovative Treatment Option for Patients with Recurrent Nasal Polyps Burden of illness and management of Chronic Sinusitis with Nasal Polyps Continuum of care and polyp recurrence Clinical and health
An Innovative Treatment Option for Patients with Recurrent Nasal Polyps Burden of illness and management of Chronic Sinusitis with Nasal Polyps Continuum of care and polyp recurrence Clinical and health
PATIENT SELECTION AND MANAGEMENT OF IN-OFFICE BALLOON SINUS DILATION UNDER LOCAL ANESTHESIA IN ADOLESCENT PATIENTS
 WHITE PAPER PATIENT SELECTION AND MANAGEMENT OF IN-OFFICE BALLOON SINUS DILATION UNDER LOCAL ANESTHESIA IN ADOLESCENT PATIENTS Jeffrey S. Rosenbloom, MD Jeffrey Holland, AAS, CRC Alamo ENT Associates,
WHITE PAPER PATIENT SELECTION AND MANAGEMENT OF IN-OFFICE BALLOON SINUS DILATION UNDER LOCAL ANESTHESIA IN ADOLESCENT PATIENTS Jeffrey S. Rosenbloom, MD Jeffrey Holland, AAS, CRC Alamo ENT Associates,
FUNCTIONAL ENDOSCOPIC SINUS SURGERY (FESS)
 UnitedHealthcare Oxford Clinical Policy FUNCTIONAL ENDOSCOPIC SINUS SURGERY (FESS) Policy Number: ENT 022.5 T2 Effective Date: April 1, 2018 Table of Contents Page INSTRUCTIONS FOR USE... 1 CONDITIONS
UnitedHealthcare Oxford Clinical Policy FUNCTIONAL ENDOSCOPIC SINUS SURGERY (FESS) Policy Number: ENT 022.5 T2 Effective Date: April 1, 2018 Table of Contents Page INSTRUCTIONS FOR USE... 1 CONDITIONS
Reasons for Failure and Surgical Revisions. Stil Kountakis, MD, PhD Professor and Chief, Division of Rhinology
 Reasons for Failure and Surgical Revisions Stil Kountakis, MD, PhD Professor and Chief, Division of Rhinology Medical College of Georgia of Georgia Regents University Department of Otolaryngology / Head
Reasons for Failure and Surgical Revisions Stil Kountakis, MD, PhD Professor and Chief, Division of Rhinology Medical College of Georgia of Georgia Regents University Department of Otolaryngology / Head
Delivering Innovation. Where It s Needed. August 2018
 Delivering Innovation. Where It s Needed. August 2018 Forward-Looking Statements Certain statements in this presentation constitute forward-looking statements within the meaning of the Securities Act of
Delivering Innovation. Where It s Needed. August 2018 Forward-Looking Statements Certain statements in this presentation constitute forward-looking statements within the meaning of the Securities Act of
Limitations of balloon sinuplasty in frontal sinus surgery
 Eur Arch Otorhinolaryngol (2011) 268:1463 1467 DOI 10.1007/s00405-011-1626-7 RHINOLOGY Limitations of balloon sinuplasty in frontal sinus surgery S. Heimgartner J. Eckardt D. Simmen H. R. Briner A. Leunig
Eur Arch Otorhinolaryngol (2011) 268:1463 1467 DOI 10.1007/s00405-011-1626-7 RHINOLOGY Limitations of balloon sinuplasty in frontal sinus surgery S. Heimgartner J. Eckardt D. Simmen H. R. Briner A. Leunig
Clinical Policy Title: Propel (drug eluting devices after sinus surgery)
 Clinical Policy Title: Propel (drug eluting devices after sinus surgery) Clinical Policy Number: 10.03.07 Effective Date: July 1, 2017 Initial Review Date: May 19, 2017 Most Recent Review Date: June 22,
Clinical Policy Title: Propel (drug eluting devices after sinus surgery) Clinical Policy Number: 10.03.07 Effective Date: July 1, 2017 Initial Review Date: May 19, 2017 Most Recent Review Date: June 22,
Clinical Policy Title: Propel (drug eluting devices after sinus surgery)
 Clinical Policy Title: Propel (drug eluting devices after sinus surgery) Clinical Policy Number: 10.03.07 Effective Date: July 1, 2017 Initial Review Date: May 19, 2017 Most Recent Review Date: May 1,
Clinical Policy Title: Propel (drug eluting devices after sinus surgery) Clinical Policy Number: 10.03.07 Effective Date: July 1, 2017 Initial Review Date: May 19, 2017 Most Recent Review Date: May 1,
Topic: Percutaneous Axial Anterior Lumbar Fusion Date of Origin: June Section: Surgery Last Reviewed Date: June 2013
 Medical Policy Manual Topic: Percutaneous Axial Anterior Lumbar Fusion Date of Origin: June 2007 Section: Surgery Last Reviewed Date: June 2013 Policy No: 157 Effective Date: August 1, 2013 IMPORTANT REMINDER
Medical Policy Manual Topic: Percutaneous Axial Anterior Lumbar Fusion Date of Origin: June 2007 Section: Surgery Last Reviewed Date: June 2013 Policy No: 157 Effective Date: August 1, 2013 IMPORTANT REMINDER
Surgical Management of Sinusitis (What About Balloons?) Relative Indications for Sinus Surgery in Children
 Surgical Management of Sinusitis (What About Balloons?) Andrew N. Goldberg M.D. Andrew H. Murr M.D. Michael J. Cunningham, M.D. Department of Otolaryngology and Communication Enhancement Children s Hospital
Surgical Management of Sinusitis (What About Balloons?) Andrew N. Goldberg M.D. Andrew H. Murr M.D. Michael J. Cunningham, M.D. Department of Otolaryngology and Communication Enhancement Children s Hospital
Functional Endoscopic Sinus Surgery (FESS)
 Functional Endoscopic Sinus Surgery (FESS) Last Review Date: December 12, 2017 Number: MG.MM.SU.56C2 Medical Guideline Disclaimer Property of EmblemHealth. All rights reserved. The treating physician or
Functional Endoscopic Sinus Surgery (FESS) Last Review Date: December 12, 2017 Number: MG.MM.SU.56C2 Medical Guideline Disclaimer Property of EmblemHealth. All rights reserved. The treating physician or
Policy Effective January 29, 2019
 Corporate Medical Policy Surgical Treatment of Sinus Disease File Name: Origination: Last CAP Review: Next CAP Review: Last Review: surgical_treatment_of_sinus_disease 2/2010 8/2018 8/2019 8/2018 Policy
Corporate Medical Policy Surgical Treatment of Sinus Disease File Name: Origination: Last CAP Review: Next CAP Review: Last Review: surgical_treatment_of_sinus_disease 2/2010 8/2018 8/2019 8/2018 Policy
DIFFICULT-TO-TREAT CHRONIC
 MANAGEMENT STRATEGIES FOR DIFFICULT-TO-TREAT CHRONIC RHINOSINUSITIS DR ZULKEFLI HUSSEIN CONSULTANT EAR NOSE & THROAT SURGEON PANTAI HOSPITAL PENANG DISCLAIMER Nothing to disclose PENANG ISLAND, MALAYSIA
MANAGEMENT STRATEGIES FOR DIFFICULT-TO-TREAT CHRONIC RHINOSINUSITIS DR ZULKEFLI HUSSEIN CONSULTANT EAR NOSE & THROAT SURGEON PANTAI HOSPITAL PENANG DISCLAIMER Nothing to disclose PENANG ISLAND, MALAYSIA
FUNCTIONAL ENDOSCOPIC SINUS SURGERY (FESS)
 UnitedHealthcare Oxford Clinical Policy FUNCTIONAL ENDOSCOPIC SINUS SURGERY (FESS) Policy Number: ENT 022.3 T2 Effective Date: October 1, 2017 Table of Contents Page INSTRUCTIONS FOR USE... 1 CONDITIONS
UnitedHealthcare Oxford Clinical Policy FUNCTIONAL ENDOSCOPIC SINUS SURGERY (FESS) Policy Number: ENT 022.3 T2 Effective Date: October 1, 2017 Table of Contents Page INSTRUCTIONS FOR USE... 1 CONDITIONS
Absorbable Nasal Implant for Treatment of Nasal Valve Collapse
 NOTE: This policy is not effective until March 1, 2019. Medical Policy Manual Surgery, Policy No. 209 Absorbable Nasal Implant for Treatment of Nasal Valve Collapse Next Review: November 2019 Last Review:
NOTE: This policy is not effective until March 1, 2019. Medical Policy Manual Surgery, Policy No. 209 Absorbable Nasal Implant for Treatment of Nasal Valve Collapse Next Review: November 2019 Last Review:
Medical Policy Manual. Topic: Peripheral Subcutaneous Field Stimulation Date of Origin: April Section: Surgery Last Reviewed Date: April 2014
 Medical Policy Manual Topic: Peripheral Subcutaneous Field Stimulation Date of Origin: April 2013 Section: Surgery Last Reviewed Date: April 2014 Policy No: 188 Effective Date: July 1, 2014 IMPORTANT REMINDER
Medical Policy Manual Topic: Peripheral Subcutaneous Field Stimulation Date of Origin: April 2013 Section: Surgery Last Reviewed Date: April 2014 Policy No: 188 Effective Date: July 1, 2014 IMPORTANT REMINDER
Testing with currently available computerized 2-lead resting ECG analysis devices consists of four steps:
 Medical Policy Manual Topic: Computerized 2-lead Resting Electrocardiogram Analysis for the Diagnosis of Coronary Artery Disease Date of Origin: December 2011 Section: Medicine Last Reviewed Date: September
Medical Policy Manual Topic: Computerized 2-lead Resting Electrocardiogram Analysis for the Diagnosis of Coronary Artery Disease Date of Origin: December 2011 Section: Medicine Last Reviewed Date: September
The Honrubia Technique of Balloon Sinuplasty for the Improvement of Symptoms in Chronic Sinusitis. Patients. Vincent Honrubia MD, FACS
 The Honrubia Technique of Balloon Sinuplasty for the Improvement of Symptoms in Chronic Sinusitis Patients Vincent Honrubia MD, FACS Allyssa Cantu, PA-S Sharon Gelman, MEd, PA-S Rachel Tsai, BS Director,
The Honrubia Technique of Balloon Sinuplasty for the Improvement of Symptoms in Chronic Sinusitis Patients Vincent Honrubia MD, FACS Allyssa Cantu, PA-S Sharon Gelman, MEd, PA-S Rachel Tsai, BS Director,
Conventional Sinus Surgery Vs Fess
 IOSR Journal of Dental and Medical Sciences (IOSR-JDMS) e-issn: 2279-0853, p-issn: 2279-0861.Volume 16, Issue 7 Ver. III (July. 2017), PP 44-51 www.iosrjournals.org Conventional Sinus Surgery Vs Fess *
IOSR Journal of Dental and Medical Sciences (IOSR-JDMS) e-issn: 2279-0853, p-issn: 2279-0861.Volume 16, Issue 7 Ver. III (July. 2017), PP 44-51 www.iosrjournals.org Conventional Sinus Surgery Vs Fess *
Prefabricated Oral Appliances for Obstructive Sleep Apnea
 Medical Policy Manual Allied Health, Policy No. 36 Prefabricated Oral Appliances for Obstructive Sleep Apnea Next Review: May 2019 Last Review: April 2018 Effective: May 1, 2018 IMPORTANT REMINDER Medical
Medical Policy Manual Allied Health, Policy No. 36 Prefabricated Oral Appliances for Obstructive Sleep Apnea Next Review: May 2019 Last Review: April 2018 Effective: May 1, 2018 IMPORTANT REMINDER Medical
2019 ACCLARENT REIMBURSEMENT GUIDE. Physician and Facility
 2019 ACCLARENT REIMBURSEMENT GUIDE Physician and Facility TABLE OF CONTENTS This guide has been developed to assist you in obtaining physician and facility reimbursement for: Nasal/Sinus Endoscopic Surgery
2019 ACCLARENT REIMBURSEMENT GUIDE Physician and Facility TABLE OF CONTENTS This guide has been developed to assist you in obtaining physician and facility reimbursement for: Nasal/Sinus Endoscopic Surgery
David C. Brodner, M.D.
 David C. Brodner, M.D. 8794 Boynton Beach Boulevard Suite 208 Boynton Beach, FL 33472 Office: (561) 735-8750 Cell: (561) 374-3308 Fax: (561) 735-8785 www.brodnermd.com DavidBrodnerMD@alumni.duke.edu SUMMARY:
David C. Brodner, M.D. 8794 Boynton Beach Boulevard Suite 208 Boynton Beach, FL 33472 Office: (561) 735-8750 Cell: (561) 374-3308 Fax: (561) 735-8785 www.brodnermd.com DavidBrodnerMD@alumni.duke.edu SUMMARY:
Pediatric Endoscopic Sinus Surgery in a Tertiary Government Hospital: Patient Profile and Surgical Indications
 Philippine Journal Of Otolaryngology-Head And Neck Surgery Vol. 4 No. January June ichael Joseph C. David, D Gil. Vicente, D, Antonio H. Chua, D, Jose R. Reyes emorial edical Center St. Luke s edical Center
Philippine Journal Of Otolaryngology-Head And Neck Surgery Vol. 4 No. January June ichael Joseph C. David, D Gil. Vicente, D, Antonio H. Chua, D, Jose R. Reyes emorial edical Center St. Luke s edical Center
Frontal Sinus Drillout (Modified Lothrop Procedure): Long-Term Results in 204 Patients
 The Laryngoscope VC 2013 The American Laryngological, Rhinological and Otological Society, Inc. Frontal Sinus Drillout (Modified Lothrop Procedure): Long-Term Results in 204 Patients Jonathan Y. Ting,
The Laryngoscope VC 2013 The American Laryngological, Rhinological and Otological Society, Inc. Frontal Sinus Drillout (Modified Lothrop Procedure): Long-Term Results in 204 Patients Jonathan Y. Ting,
Single Multi-Functional Device
 Treat Multiple Sinuses with a Single Multi-Functional Device BALLOON DILATION SEEKER CONFIRMATION SUCTION IRRIGATION Treat the Frontal, Sphenoid and Maxillary Sinuses with a Single Multi-Functional Device
Treat Multiple Sinuses with a Single Multi-Functional Device BALLOON DILATION SEEKER CONFIRMATION SUCTION IRRIGATION Treat the Frontal, Sphenoid and Maxillary Sinuses with a Single Multi-Functional Device
Section: Medicine Last Reviewed Date: June Policy No: 130 Effective Date: September 1, 2014
 Medical Policy Manual Topic: Manipulation Under Anesthesia for the Treatment of Pain Date of Origin: April 2009 Section: Medicine Last Reviewed Date: June 2014 Policy No: 130 Effective Date: September
Medical Policy Manual Topic: Manipulation Under Anesthesia for the Treatment of Pain Date of Origin: April 2009 Section: Medicine Last Reviewed Date: June 2014 Policy No: 130 Effective Date: September
Cigna Medical Coverage Policy
 Cigna Medical Coverage Policy Subject Drug-Eluting Devices for Use Following Endoscopic Sinus Surgery Table of Contents Coverage Policy... 1 General Background... 1 Coding/Billing Information... 6 References...
Cigna Medical Coverage Policy Subject Drug-Eluting Devices for Use Following Endoscopic Sinus Surgery Table of Contents Coverage Policy... 1 General Background... 1 Coding/Billing Information... 6 References...
FUNCTIONAL ENDOSCOPIC SINUS SURGERY (FESS)
 Oxford UnitedHealthcare Oxford Clinical Policy FUNCTIONAL ENDOSCOPIC SINUS SURGERY (FESS) Policy Number: ENT 022.6 T2 Effective Date: October 1, 2018 Table of Contents Page INSTRUCTIONS FOR USE... 1 CONDITIONS
Oxford UnitedHealthcare Oxford Clinical Policy FUNCTIONAL ENDOSCOPIC SINUS SURGERY (FESS) Policy Number: ENT 022.6 T2 Effective Date: October 1, 2018 Table of Contents Page INSTRUCTIONS FOR USE... 1 CONDITIONS
Handheld Radiofrequency Spectroscopy for Intraoperative Assessment of Surgical Margins During Breast-Conserving Surgery
 7.01.140 Handheld Radiofrequency Spectroscopy for Intraoperative Assessment of Surgical Margins During Breast-Conserving Surgery Section 7.0 Surgery Subsection Description Effective Date November 26, 2014
7.01.140 Handheld Radiofrequency Spectroscopy for Intraoperative Assessment of Surgical Margins During Breast-Conserving Surgery Section 7.0 Surgery Subsection Description Effective Date November 26, 2014
Incidence of accessory ostia in patients with chronic maxillary sinusitis
 International Journal of Otorhinolaryngology and Head and Neck Surgery Ghosh P et al. Int J Otorhinolaryngol Head Neck Surg. 2018 Mar;4(2):443-447 http://www.ijorl.com pissn 2454-5929 eissn 2454-5937 Original
International Journal of Otorhinolaryngology and Head and Neck Surgery Ghosh P et al. Int J Otorhinolaryngol Head Neck Surg. 2018 Mar;4(2):443-447 http://www.ijorl.com pissn 2454-5929 eissn 2454-5937 Original
The advent of high-resolution computerized tomography
 An anatomic classification of the ethmoidal bulla REUBEN C. SETLIFF, III, MD, PETER J. CATALANO, MD, FACS, LISA A. CATALANO, MPH, and CHAD FRANCIS, BA, Sioux Falls, South Dakota, and Burlington, Massachusetts
An anatomic classification of the ethmoidal bulla REUBEN C. SETLIFF, III, MD, PETER J. CATALANO, MD, FACS, LISA A. CATALANO, MPH, and CHAD FRANCIS, BA, Sioux Falls, South Dakota, and Burlington, Massachusetts
National Institute for Health and Care Excellence Medical Technologies Evaluation Programme
 National Institute for Health and Care Excellence Medical Technologies Evaluation Programme Consultation Comments table MTAC date: 23 September 2016 There were 25 consultation comments from 8 consultees
National Institute for Health and Care Excellence Medical Technologies Evaluation Programme Consultation Comments table MTAC date: 23 September 2016 There were 25 consultation comments from 8 consultees
If looking for a book by Norra MacReady The patient with a sinus headache may actually have a migraine.(more than 3,000 Patients Studied): An article
 The Patient With A Sinus Headache May Actually Have A Migraine.(More Than 3,000 Patients Studied): An Article From: Internal Medicine News [HTML] [Digital] By Norra MacReady READ ONLINE If looking for
The Patient With A Sinus Headache May Actually Have A Migraine.(More Than 3,000 Patients Studied): An Article From: Internal Medicine News [HTML] [Digital] By Norra MacReady READ ONLINE If looking for
US Drug Eluting Sinus Stent (DESS) Market: Size, Trends & Forecasts ( ) September 2016
 US Drug Eluting Sinus Stent (DESS) Market: Size, Trends & Forecasts (2016-2021) September 2016 US Drug Eluting Sinus Stent Market Report Scope of the Report The report entitled US Drug Eluting Sinus Stent
US Drug Eluting Sinus Stent (DESS) Market: Size, Trends & Forecasts (2016-2021) September 2016 US Drug Eluting Sinus Stent Market Report Scope of the Report The report entitled US Drug Eluting Sinus Stent
Endoscopic Management Of A Giant Ethmoid Mucocele
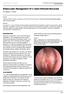 ISPUB.COM The Internet Journal of Otorhinolaryngology Volume 6 Number 1 S Ceylan, F Bora Citation S Ceylan, F Bora.. The Internet Journal of Otorhinolaryngology. 2006 Volume 6 Number 1. Abstract We present
ISPUB.COM The Internet Journal of Otorhinolaryngology Volume 6 Number 1 S Ceylan, F Bora Citation S Ceylan, F Bora.. The Internet Journal of Otorhinolaryngology. 2006 Volume 6 Number 1. Abstract We present
Hitting the Jackpot Sinuses, Scopes, and Surgery
 Hitting the Jackpot Sinuses, Scopes, and Surgery Candice Fenildo holds an Associates Degree in Health Sciences and is Certified in Healthcare Compliance (CHC), a Certified Professional Coder (CPC), a Certified
Hitting the Jackpot Sinuses, Scopes, and Surgery Candice Fenildo holds an Associates Degree in Health Sciences and is Certified in Healthcare Compliance (CHC), a Certified Professional Coder (CPC), a Certified
Tomographical Findings in Adult Patients Undergoing Endoscopic Sinus Surgery Revision
 THIEME Original Research 73 Tomographical Findings in Adult Patients Undergoing Endoscopic Sinus Surgery Revision Jan Alessandro Socher 1 Jonas Mello 2 Barbara Batista Baltha 2 1 Department of Otorhinolaryngology,
THIEME Original Research 73 Tomographical Findings in Adult Patients Undergoing Endoscopic Sinus Surgery Revision Jan Alessandro Socher 1 Jonas Mello 2 Barbara Batista Baltha 2 1 Department of Otorhinolaryngology,
1. BRIEF DESCRIPTION OF TRAINING
 RHINOLOGY 1. BRIEF DESCRIPTION OF TRAINING Exposure to clinical rhinology is provided in each of the four ORL years over the course of several rotations in a graduated approach. MEE General Otolaryngology
RHINOLOGY 1. BRIEF DESCRIPTION OF TRAINING Exposure to clinical rhinology is provided in each of the four ORL years over the course of several rotations in a graduated approach. MEE General Otolaryngology
Page: 1 of 6. Transtympanic Micropressure Applications as a Treatment of Meniere's Disease
 Page: 1 of 6 Last Review Status/Date: December 2013 as a Treatment of Meniere's Disease Description Transtympanic micropressure treatment for Meniere s disease involves use of a hand-held air pressure
Page: 1 of 6 Last Review Status/Date: December 2013 as a Treatment of Meniere's Disease Description Transtympanic micropressure treatment for Meniere s disease involves use of a hand-held air pressure
ISSN X (Print) Research Article. *Corresponding author Dr.V. Krishna Chaitanya
 Scholars Journal of Applied Medical Sciences (SJAMS) Sch. J. App. Med. Sci., 2015; 3(1G):508-513 Scholars Academic and Scientific Publisher (An International Publisher for Academic and Scientific Resources)
Scholars Journal of Applied Medical Sciences (SJAMS) Sch. J. App. Med. Sci., 2015; 3(1G):508-513 Scholars Academic and Scientific Publisher (An International Publisher for Academic and Scientific Resources)
Handheld Radiofrequency Spectroscopy for Intraoperative Assessment of Surgical Margins During Breast-Conserving Surgery
 Last Review Status/Date: December 2016 Page: 1 of 6 Intraoperative Assessment of Surgical Description Breast-conserving surgery as part of the treatment of localized breast cancer is optimally achieved
Last Review Status/Date: December 2016 Page: 1 of 6 Intraoperative Assessment of Surgical Description Breast-conserving surgery as part of the treatment of localized breast cancer is optimally achieved
Balloon Dilation of the Eustachian Tube
 Applies to all products administered or underwritten by Blue Cross and Blue Shield of Louisiana and its subsidiary, HMO Louisiana, Inc.(collectively referred to as the Company ), unless otherwise provided
Applies to all products administered or underwritten by Blue Cross and Blue Shield of Louisiana and its subsidiary, HMO Louisiana, Inc.(collectively referred to as the Company ), unless otherwise provided
2017 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY. MEASURE TYPE: Efficiency
 Measure #334: Adult Sinusitis: More than One Computerized Tomography (CT) Scan Within 90 Days for Chronic Sinusitis (Overuse) National Quality Strategy Domain: Efficiency and Cost Reduction 2017 OPTIONS
Measure #334: Adult Sinusitis: More than One Computerized Tomography (CT) Scan Within 90 Days for Chronic Sinusitis (Overuse) National Quality Strategy Domain: Efficiency and Cost Reduction 2017 OPTIONS
ENT NAVIGATION SIMPLE AND INTUITIVE
 ENT NAVIGATION SIMPLE AND INTUITIVE REVOLUTIONARY NAVIGATION TECHNOLOGY FOR ROUTINE AND COMPLEX ENT PROCEDURES We at Fiagon believe patient care should be centered around the patient. For us, patient-centered-care
ENT NAVIGATION SIMPLE AND INTUITIVE REVOLUTIONARY NAVIGATION TECHNOLOGY FOR ROUTINE AND COMPLEX ENT PROCEDURES We at Fiagon believe patient care should be centered around the patient. For us, patient-centered-care
Original Article Comparison of different surgical approaches of functional endoscopic sinus surgery on patients with chronic rhinosinusitis
 Int J Clin Exp Med 2014;7(6):1585-1591 www.ijcem.com /ISSN:1940-5901/IJCEM0000315 Original Article Comparison of different surgical approaches of functional endoscopic sinus surgery on patients with chronic
Int J Clin Exp Med 2014;7(6):1585-1591 www.ijcem.com /ISSN:1940-5901/IJCEM0000315 Original Article Comparison of different surgical approaches of functional endoscopic sinus surgery on patients with chronic
By: Abdulrahman A. Al-Humaizi Rhinology Fellow, F2
 20.03.2017 By: Abdulrahman A. Al-Humaizi Rhinology Fellow, F2 Objectives Introduction Materials and methods Results Discussion Literature Review Conclusion Introduction Topical delivery to the paranasal
20.03.2017 By: Abdulrahman A. Al-Humaizi Rhinology Fellow, F2 Objectives Introduction Materials and methods Results Discussion Literature Review Conclusion Introduction Topical delivery to the paranasal
CHRONIC RHINOSINUSITIS IN ADULTS
 CHRONIC RHINOSINUSITIS IN ADULTS SCOPE OF THE PRACTICE GUIDELINE This clinical practice guideline is for use by the Philippine Society of Otolaryngology-Head and Neck Surgery. It covers the diagnosis and
CHRONIC RHINOSINUSITIS IN ADULTS SCOPE OF THE PRACTICE GUIDELINE This clinical practice guideline is for use by the Philippine Society of Otolaryngology-Head and Neck Surgery. It covers the diagnosis and
Canine Fossa Puncture for Severe Maxillary Disease in Unilateral Chronic Sinusitis With Nasal Polyp
 The Laryngoscope VC 2013 The American Laryngological, Rhinological and Otological Society, Inc. Canine Fossa Puncture for Severe Maxillary Disease in Unilateral Chronic Sinusitis With Nasal Polyp Jang
The Laryngoscope VC 2013 The American Laryngological, Rhinological and Otological Society, Inc. Canine Fossa Puncture for Severe Maxillary Disease in Unilateral Chronic Sinusitis With Nasal Polyp Jang
Eosinophilic Rhinosinusitis is Not a Disease of Ostiomeatal Occlusion
 The Laryngoscope VC 2013 The American Laryngological, Rhinological and Otological Society, Inc. Eosinophilic Rhinosinusitis is Not a Disease of Ostiomeatal Occlusion Kornkiat Snidvongs, MD; David Chin,
The Laryngoscope VC 2013 The American Laryngological, Rhinological and Otological Society, Inc. Eosinophilic Rhinosinusitis is Not a Disease of Ostiomeatal Occlusion Kornkiat Snidvongs, MD; David Chin,
Sinusitis: Medical and Surgical Management. R. Jonathan Lara, DO, FAOCO. Sonoran Ear Nose & Throat
 Sinusitis: Medical and Surgical Management R. Jonathan Lara, DO, FAOCO Sonoran Ear Nose & Throat 27 th Annual Southwestern Conference on Medicine April 26, 2018 What are sinuses? Air filled cavities of
Sinusitis: Medical and Surgical Management R. Jonathan Lara, DO, FAOCO Sonoran Ear Nose & Throat 27 th Annual Southwestern Conference on Medicine April 26, 2018 What are sinuses? Air filled cavities of
If looking for a ebook by Norra MacReady The patient with a sinus headache may actually have a migraine.(more than 3,000 Patients Studied): An
 The Patient With A Sinus Headache May Actually Have A Migraine.(More Than 3,000 Patients Studied): An Article From: Internal Medicine News [HTML] [Digital] By Norra MacReady READ ONLINE If looking for
The Patient With A Sinus Headache May Actually Have A Migraine.(More Than 3,000 Patients Studied): An Article From: Internal Medicine News [HTML] [Digital] By Norra MacReady READ ONLINE If looking for
treating chronic sinusitis
 NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Medical technology consultation document XprESS multi-sinus dilation system for The National Institute for Health and Care Excellence (NICE) is producing
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Medical technology consultation document XprESS multi-sinus dilation system for The National Institute for Health and Care Excellence (NICE) is producing
