Corporate Medical Policy
|
|
|
- Magdalen Greene
- 5 years ago
- Views:
Transcription
1 Corporate Medical Policy Surgical Treatment of Sinus Disease Description of Procedure or Service Sinusitis refers to infection or inflammation of the sinuses, which are small openings in the bones of the face. Symptoms include a stuffy nose, facial pain and discharge from the nose. Most sinus infections will get better without any specific treatment. For some people, sinus symptoms may last for months, and this is called chronic sinusitis. Standard therapy to treat chronic sinusitis may include decongestants, antibiotics, antihistamines, saline irrigation, and the use of nasal spray containing steroids. When chronic sinusitis does not respond to standard medical treatments, then surgery may be the next step. In some cases of chronic sinusitis, surgical drainage may be necessary when symptoms have not responded to medications or other conservative methods. Traditional functional endoscopic sinus surgery is generally considered the clinical standard; however balloon sinus ostial dilation is a recently emerged option that is appropriate for certain clinical circumstances. In both procedures, the sinuses are opened wider to allow for natural drainage of mucus resulting in movement of air through the nasal and sinus pathways. Background Chronic sinusitis is one of the most frequently diagnosed chronic medical conditions, even more so than hypertension and arthritis. Chronic sinusitis is characterized by purulent nasal discharge, usually without fever, that persists for weeks to months. Symptoms of congestion often accompany the nasal discharge. There also may be mild pain and/or headache. Thickening of mucosa may restrict or close natural openings between sinus cavities and the nasal fossae. Considerable variation exists in the location and shape of these sinus ostia. A course of conservative medical therapy is attempted initially to resolve the symptoms; this treatment may include antibiotics, nasal irrigation, decongestants, antihistamines, and steroids. Functional endoscopic sinus surgery (FESS) is a commonly used surgical technique to treat medically unresponsive chronic sinusitis and other serious conditions of the nasal sinuses that result in impaired sinus drainage. The use of the endoscope permits a better view of the surgical field. Goals of FESS are to allow for maximum preservation of mucosa, and to open and enlarge the sinus passageways allowing for proper drainage. FESS is performed using a rigid endoscope to view the structures of the nose and sinuses. The endoscope is inserted through the nose, as are the tiny surgical instruments the surgeon uses
2 to perform the surgery. The procedure can be performed under general or local anesthesia on an outpatient basis, and patients usually experience minimal discomfort. The use of FESS allows for a much less invasive and traumatic procedure than conventional sinus surgery. There are shorter surgery and healing times, less postoperative discomfort, and fewer surgical complications with FESS. However, because of the proximity of sinus structures to the eyes and the brain, it is not risk free. Balloon ostial dilation is considered an alternative to endoscopic sinus surgery for those with chronic sinusitis of the frontal, maxillary, or sphenoid sinuses. The procedure involves placing a guidewire in the sinus ostium, advancing a balloon over the guidewire, and then stretching the opening by inflating the balloon. The guidewire location is confirmed with fluoroscopy or with direct trans illumination of the targeted sinus cavity. General anesthesia may be needed for this procedure to minimize patient movement. This technique is said to allow improved sinus drainage. In March 2008, the device Relieva Sinus Balloon Catheter was cleared for marketing by the U.S. Food and Drug Administration (FDA). The FDA determined that this device was substantially equivalent to existing devices for use in dilating the sinus ostia and paranasal spaces in adults and maxillary sinus spaces in children. Subsequent devices developed have also been granted clearance. These include the Relieva Spin Sinus Dilation System, and the Relieva Seeker Balloon Sinuplasty System, FinESS Sinus Treatment, ENTrigue Sinus Dilation System, and the XprESS Multi-Sinus Dilation Tool Some form of sinus packing is generally performed postoperatively. Simple dressings moistened with saline can be inserted manually following surgery. Foam dressings are polysaccharide substances that form a gel when hydrated and can be used as nasal packs for a variety of indications. Middle meatal spacers are splint-like devices that prop open the sinus cavities post- FESS, but are not capable of drug delivery. Implantable drug-eluting sinus implants are another option for postoperative management following FESS and other sinus procedures. These implants are inserted under endoscopic guidance to stabilize the sinus openings and the turbinates, reduce edema, and/or prevent obstruction by adhesions. They also deliver medications (e.g. steroids) topically over an extended period of time (e.g. 30 days), and this local delivery of medications may be superior to other treatment options in the postoperative setting. The only U.S. Food and Drug Administration approved drug-eluting sinus implant at this time is Propel (mometasone furoate sinus implant). There are numerous other stenting and packing materials commonly used in sinus surgery. Two RCTs have compared the Propel device with steroids to the same device without steroids, and reported that the steroid-eluting device reduced postoperative inflammation, reduced the need for oral steroids, and reduced the need for postoperative re-interventions. These trials evaluate the benefit of local steroid delivery in addition to standard care. The improvements reported in these trials reflect the impact of local steroid delivery, which was withheld in the control arm, as well as the impact of the stent device itself.
3 Benefit Application This medical policy relates only to the services or supplies described herein. Please refer to the member s benefit booklet for availability of benefits. The clinical coverage policy for rhinoplasty can be found at geha.com. GEHA considers any surgical procedure (or any portion of a procedure) performed primarily to improve physical appearance through change in bodily form, except repair of accidental injury if repair is initiated promptly or as soon as the member s condition permits, as cosmetic and therefore not a covered benefit. Regulatory Status Sinus surgeries are procedures and, as such, are not subject to regulation by the FDA. However, the FDA does regulate manufacturing and dispensing practices and use of devices and drugs for such procedures. Policy Statement GEHA will cover surgical treatment of sinus disease when determined to be medical necessary because the medical criteria and guidelines shown below are met. When surgical treatment of sinus disease is covered Functional endoscopic sinus surgery (FESS) is considered medically necessary for the treatment of sinusitis, polyposis, or sinus tumor when any one of the following circumstances is present: 1. Suspected tumor seen on preferred imaging, physical examination, or endoscopy; or 2. Supportive complications, including but are not limited to: A. Sub periosteal abscess; or B. Brain abscess; or 3. Chronic polyposis with symptoms unresponsive to medical therapy, including, but not limited to the following: A. Oral steroids (unless contraindicated); and B. Oral antibiotics (unless contraindicated); and C. Topical nasal steroid sprays; and D. Allergy evaluation and treatment (where indicated). 4. Allergic fungal sinusitis, as indicated by all of the following: A. Specific diagnostic criteria including: 1. Nasal polyposis; and 2. Positive CT findings; and 3. Eosinophilic mucus; and AND B. Allergy evaluation and treatment that fails to adequately manage the condition
4 5. Mucocele causing chronic sinusitis; or 6. Recurrent sinusitis that triggers or exacerbates comorbidities such as asthma or cystic fibrosis; or 7. Uncomplicated sinusitis (for example, sinusitis confined to the paranasal sinuses without adjacent involvement of neurologic, soft tissue, or bony structures) and all (A, B, and C) of the following: A. Either of the following: 1. Four or more documented episodes of acute rhinosinusitis (less than 4 weeks duration) in one year; or 2. Chronic sinusitis (greater than 12 weeks continuously) associated with current functional deficits; and B. Maximal medical therapy has been attempted, as indicated by all of the following: 1. Antibiotic therapy for at least 4 consecutive weeks with appropriate antimicrobial therapy; and 2. Trial of antihistamines and steroids and/or topical intranasal steriods; and 3. Nasal lavage; and 4. Allergy testing where indicated; and C. Abnormal findings from diagnostic workup, as indicated by any one of the following: 1. CT findings suggestive of obstruction or infection for example, but not limited to, air fluid levels, air bubbles, significant mucosal thickening, pansinusitis, or diffuse opacification; or 2. Nasal endoscopy findings suggestive of significant disease; or 8. Fungal mycetoma; or 9. Cerebrospinal fluid rhinorrhea; or 10. Encephalocele; or 11. Posterior epistaxis (refractory to conservative treatment); or 12. Persistent facial pain with identified anatomic pathology in the sinuses confirmed by preferred imaging; or 13. Cavernous sinus thrombosis caused by chronic sinusitis. When nasal or sinus cavity debridement is covered Nasal or sinus cavity debridement following FESS is considered medically necessary for any of the following circumstances: 1. Nasal or sinus cavity debridement is allowed three times during the first 30 days postoperatively; or
5 2. Postoperative loss of vision or double vision; or 3. Evidence of cerebrospinal fluid leak such as rhinorrhea; or 4. When prompted by physical obstruction of the sinus opening related to: A. Nasal polyps unresponsive to oral or nasal steroids; or B. Documented presence of papilloma, carcinoma or other neoplasm; or C. Allergic fungal sinusitis. When surgical treatment of sinus disease is not covered: 1. Functional endoscopic sinus surgery is considered not medically necessary for the treatment of sinusitis, polyposis, sinus tumor, or any other condition when the criteria above are not met. 2. Nasal or sinus cavity debridement following FESS is considered not medically necessary when criteria above are not met, including additional post-surgical debridement beyond 30 days post-procedure. When Balloon Sinus Ostial Dilation is covered: Balloon sinus ostial dilation is medically necessary for treating chronic rhinosinusitis (defined as rhinosinusitis lasting longer than 12 weeks) when the following are met: 1. Chronic rhinosinusitis of the sinus to be dilated is confirmed on computed tomography scan. CT scan findings of chronic rhinosinusitis include one or more of the following: significant mucosal thickening, periosteal remodeling, periosteal thickening, and or obstruction of the ostiomeatal complex. 2. Balloon sinus ostial dilation is limited to the frontal, maxillary and/or sphenoid sinuses 3. Balloon sinus ostial dilation is performed either as a stand-alone procedure or as part of functional endoscopic sinus surgery 4. Balloon sinus ostial dilation is performed in persons older than 12 years of age whose symptoms persist despite medical therapy, as indicated, including all of the following: A. Oral steroids and/or nasal steroid sprays (unless contraindicated); and B. Oral antibiotics (unless contraindicated); and C. Allergy evaluation and treatment (where indicated). i. Nasal lavage; and ii. Allergy testing where indicated; and D. Abnormal findings from diagnostic workup, as indicated by any one of the following: i. CT findings suggestive of obstruction or infection for example, but not limited to, air fluid levels, air bubbles, significant mucosal thickening, pansinusitis, or diffuse opacification; or
6 ii. Nasal endoscopy findings suggestive of significant disease. When Balloon Dilation is not covered: 1. Balloon sinus ostial dilation is considered investigational for treating nasal polyps or tumors. There is insufficient published clinical evidence to conclude that balloon sinus ostial dilation is safe and effective for treating nasal polyps or tumors. 2. Balloon sinus ostial dilation is considered investigational in children 12 years of age or younger. There is insufficient evidence to support the use of balloon sinus ostial dilation in the management of rhinosinusitis in children. Long-term, well-designed studies using appropriate controls are needed to determine the effectiveness of balloon sinus ostial dilation in this population. When use of a mometasone furoate sinus implant (Propel ) is covered: 1. Patient is > 18 years of age; and 2. Ethmoid sinus surgery is planned; and 3. Patient has one or more of the following conditions: A. Polypoid disease B. Failed prior surgery and/or restenosis C. Absolute or relative contraindication to systemic steroids; and 4. The functional endoscopic sinus surgery (FESS) or other primary sinus procedure is A. Considered medically necessary. Policy Guidelines Functional endoscopic sinus surgery (FESS) and balloon ostial dilation should be reserved for use in patients in whom optimal medical treatment has failed. The majority of patients with sinusitis do not require surgery. Their sinus symptoms can usually be successfully treated medically, including antibiotic therapy and other medications, treatment of allergy, and environmental control. Optimal medical treatment consists of the following: 1. Oral antibiotics of 2-4 weeks duration for patients with chronic rhinosinusitis (directed by culture is preferred). 2. Oral antibiotics with multiple 1-3 week courses for patients with recurrent acute rhinosinusitis. 3. Systemic and/or topical steroids. 4. Saline irrigations. 5. Topical and/or systemic decongestants (if not contraindicated).
7 6. Treatment of concomitant allergic rhinitis, including avoidance measures, pharmacotherapy, and/or immunotherapy. Note: Imaging studies should be generally obtained after maximal medical therapy. Based on clinical situation (i.e. concern for extra sinus complications or neoplasm), early or emergent imaging may be required to confirm a diagnosis. Prior to performing endoscopic sinus surgery in the pediatric population, the physician must consider the following specific to the pediatric patient: 1. A comprehensive historical assessment that evaluates chronic rhinosinusitis or recurrent acute rhinosinusitis that is collateral to or in addition to one of the following (this list is not all inclusive): a) Allergy b) Day care exposure c) Gastroesophageal reflux contributing to rhinosinusitis d) Adenoiditis and/or obstructive adenoid hypertrophy e) Cystic fibrosis f) Immune deficiency disorders g) Ciliary dysfunction/dyskinesia h) Progressively worsening asthma with opaque sinus(es) i) Nasal polyposis with airway obstruction and/or sinusitis j) Suspected neoplasm (e.g., juvenile nasopharyngeal angiofibroma) k) Adenoidectomy should be strongly considered a minimum of three months prior to performing pediatric sinus surgery for any of the above indications l) Intracranial complications m) Cavernous sinus thrombosis n) Mucocoeles and mucopyocoeles o) Subperiosteal or orbital abscess/periorbital cellulitis p) Traumatic injury to optic canal (decompression) q) Dacryocystitis from rhinosinusitis r) Meningocephaloceles s) Cerebrospinal fluid leaks t) Tumors of the nasal cavity, paranasal sinuses, orbit or skull base u) Recurrent acute rhinosinusitis (RARS) 2. Other tests, including for surgical planning, a coronal CT scan following medical therapy is required. A complete axial CT scan is recommended in cases with complex disease. MRI, culture and sensitivity, and allergy testing are optional. 3. Optimal medical therapy, including
8 A. Management for any of the identified medical conditions listed above in number 1. B. Treatment of rhinitis medicamentosa, when present. C. Parental education of environmental factors including allergens, irritants, or secondhand tobacco smoke. D. Antibiotic therapy consisting of four to six consecutive weeks of appropriate antibiotic drugs. E. Appropriate topical and/or systemic steroids when indicated. Physician documentation The following documentation must be provided for medical necessity review: Completed GEHA Nasal Surgeries Authorization form (can be found on geha.com) A history and physical within 12 months from request of authorization Relative office notes to include pre-procedure examination including the extent of the member s symptoms, pathology report and testing reports not already listed For post procedure submissions, include operative and/or procedure reports Imaging report(s) A letter of medical necessity including statement of unilateral or bilateral procedure. If unilateral, indicate right or left. Clinical documentation of medical management for the last 12 months with the name of the pharmacological interventions, date prescribed, and length of time each has been taken. Additional documentation must be provided for pediatric patients: Documentation of evaluation and results for the conditions listed within the guidelines of this policy. Documentation of pre-procedure anterior and posterior nasal examination (rhinoscopy after mucosal decongestion) as possible for patient s age. A coronal CT scan performed following medical therapy. Applicable codes include but are not limited to: S1090 Mometasone furoate sinus implant, 370 micrograms Excision inferior turbinate, partial or complete, any method Submucous resection inferior turbinate, partial or complete, any method Septoplasty or submucous resection, with or without cartilage scoring, contouring or replacement with graft
9 30620 Septal or other intranasal dermatoplasty (does not include obtaining graft) Nasal endoscopy, diagnostic, unilateral or bilateral (separate procedure) Nasal/sinus endoscopy, diagnostic with maxillary sinusoscopy (via inferior meatus or canine fossa puncture) Nasal/sinus endoscopy, diagnostic with sphenoid sinusoscopy (via puncture of sphenoidal face or cannulation of ostium) Nasal/sinus endoscopy, surgical; with biopsy, polypectomy or debridement (separate procedure) Nasal/sinus endoscopy, surgical; with control of nasal hemorrhage Nasal/sinus endoscopy, surgical; with dacryocystorhinostomy Nasal/sinus endoscopy, surgical; with concha bullosa resection Nasal/sinus endoscopy, surgical; with ligation of sphenopalatine artery Nasal/sinus endoscopy, surgical with ethmoidectomy; partial (anterior) Nasal/sinus endoscopy, surgical with ethmoidectomy; total (anterior and posterior) Nasal/sinus endoscopy, surgical, with sphenoidotomy; with removal of tissue from the sphenoid sinus Nasal/sinus endoscopy, surgical with ethmoidectomy; total (anterior and posterior), including sphenoidotomy, with removal of tissue from the sphenoid sinus Nasal/sinus endoscopy, surgical, with maxillary antrostomy; with removal of tissue from maxillary sinus Nasal/sinus endoscopy, surgical, with frontal sinus exploration, including removal of tissue from frontal sinus, when performed Nasal/sinus endoscopy, surgical, with sphenoidotomy Nasal/sinus endoscopy, surgical, with sphenoidotomy; with removal of tissue from the sphenoid sinus Nasal/sinus endoscopy, surgical, with repair of cerebrospinal fluid leak; ethmoid region Nasal/sinus endoscopy, surgical, with repair of cerebrospinal fluid leak; sphenoid region Nasal/sinus endoscopy, surgical; with medial or inferior orbital wall decompression Nasal/sinus endoscopy, surgical; with medial orbital wall and inferior orbital wall decompression
10 31295 Nasal/sinus endoscopy, surgical; with dilation of maxillary sinus ostium (eg, balloon dilation), transnasal or via canine fossa Nasal/sinus endoscopy, surgical; with dilation of frontal sinus ostium (eg, balloon dilation) Nasal/sinus endoscopy, surgical; with dilation of sphenoid sinus ostium (eg, balloon dilation) Nasal/sinus endoscopy, surgical; with dilation of frontal and sphenoid sinus ostia (eg, balloon dilation) 0406T Nasal endoscopy, surgical, ethmoid sinus, placement of drug eluting implant 0407T Nasal endoscopy, surgical, ethmoid sinus, placement of drug eluting implant; with biopsy, polypectomy or debridement Scientific references Berlucchi M, Castelnuovo P, Vincenzi A et al. Endoscopic outcomes of resorbable nasal packing after functional endoscopic sinus surgery: a multicenter prospective randomized controlled study. Eur Arch Otorhinolaryngol 2009; 266(6): Cote DW, Wright ED. Triamcinolone-impregnated nasal dressing following endoscopic sinus surgery: a randomized, double-blind, placebo-controlled study. Laryngoscope 2010; 120(6): Freeman SR, Sivayoham ES, Jepson K et al. A preliminary randomised controlled trial evaluating the efficacy of saline douching following endoscopic sinus surgery. Clin Otolaryngol 2008; 33(5): Rotenberg BW, Zhang I, Arra I et al. Postoperative care for Samter's triad patients undergoing endoscopic sinus surgery: a double-blinded, randomized controlled trial. Laryngoscope 2011; 121(12): Lee JM, Grewal A. Middle meatal spacers for the prevention of synechiae following endoscopic sinus surgery: a systematic review and meta-analysis of randomized controlled trials. Int Forum Allergy Rhinol 2012 Marple BF, Smith TL, Han JK et al. Advance II: a prospective, randomized study assessing safety and efficacy of bioabsorbable steroid-releasing sinus implants. Otolaryngol. Head Neck Surg. 2012; 146(6): Murr AH, Smith TL, Hwang PH et al. Safety and efficacy of a novel bioabsorbable, steroid-eluting sinus stent. Int Forum Allergy Rhinol 2011; 1(1): Forwith KD, Chandra RK, Yun PT et al. ADVANCE: a multisite trial of bioabsorbable steroid-eluting sinus implants. Laryngoscope 2011; 121(11):
11 Catalona PJ, Thong M, Weiss R, Rimash T. The MicroFlow Spacer: a drug-eluting stent for the ethmoid sinus. Indian J Otolaryngol Head Neck Surg 2011; 63(3): Rudmik L, Soler ZM, Orlandi RR et al. Early postoperative care following endoscopic sinus surgery: an evidence-based review with recommendations. Int Forum Allergy Rhinol 2011; 1(6): Han JK, Marple BF, Smith TL. Effect of steroid-releasing sinus implants on postoperative medical and surgical interventions: an efficacy meta-analysis. Int Forum Allergy Rhinol 2012; 2(4): Gould J, Alexander I, Tomkin E, Brodner D. In-office, multisinus balloon dilation: 1-Year outcomes from a prospective, multicenter, open label trial. Am J Rhinol Allergy 2014; 28(2): Bikhazi N, Light J, Truitt T, et.al. Standalone balloon dilation versus sinus surgery for chronic rhinosinusitis: A prospective, multicenter, randomized, controlled trial with 1- year follow-up. Am J Rhinol Allergy 2014; 28(4): Rosenfeld RM, Piccirillo JF, Chandrasekhar SS, et al. Clinical practice guideline (update): adult sinusitis. Otolaryngol Head Neck Surg. Apr 2015;152(2 Suppl):S1-S39. PMID American Academy of Otolaryngology-Head and Neck Surgery. Clinical Indicators: Endoscopic Sinus Surgery, Pediatric Rosenfeld RM, Andes D, Bhattacharyya N, et al. (June 2007). Clinical practice guidelines: Adult sinusitis. Otolarygol Head Neck Surg, 2007;137(3S):S1-S31. American Academy of Otolaryngology-Head and Neck Surgery. Policy statement for Sinus Endoscopy. Slack R, Bates G. (Sept 1998). Functional Endoscopic Sinus Surgery Rudmik L, Mace J, Mechor B. Effect of a dexamethasone SinuFoam middle meatal spacer on endoscopic sinus surgery outcomes: A randomized, double-blind, placebo-controlled trial. Int Forum Allergy Rhinol 2012; 2(3): Policy implementation and updates 02/2019 New policy issued. GEHA will cover sinus surgery when determined to be medically necessary because the medical criteria and guidelines outlined in the policy are met.
Implantable Sinus Stents for Postoperative Use Following Endoscopic Sinus Surgery
 Last Review Status/Date: March 2014 Page: 1 of 7 Use Following Endoscopic Sinus Surgery Description Sinus stents are devices that are used postoperatively following endoscopic sinus surgery (ESS). The
Last Review Status/Date: March 2014 Page: 1 of 7 Use Following Endoscopic Sinus Surgery Description Sinus stents are devices that are used postoperatively following endoscopic sinus surgery (ESS). The
Corporate Medical Policy
 Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: surgical_treatment_of_sinus_disease 2/2010 8/2017 8/2018 8/2017 Description of Procedure or Service Chronic
Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: surgical_treatment_of_sinus_disease 2/2010 8/2017 8/2018 8/2017 Description of Procedure or Service Chronic
Protocol. Implantable Sinus Stents for Postoperative Use Following Endoscopic Sinus Surgery and for Recurrent Sinus Disease
 (701134) Medical Benefit Effective Date: 04/01/17 Next Review Date: 01/19 Preauthorization No Review Dates: 01/13, 01/14, 01/15, 01/16, 01/17, 01/18 This protocol considers this test or procedure investigational.
(701134) Medical Benefit Effective Date: 04/01/17 Next Review Date: 01/19 Preauthorization No Review Dates: 01/13, 01/14, 01/15, 01/16, 01/17, 01/18 This protocol considers this test or procedure investigational.
Implantable Sinus Stents for Postoperative Use Following Endoscopic Sinus Surgery
 Page: 1 of 8 Last Review Status/Date: March 2015 Use Following Endoscopic Sinus Surgery Description Sinus stents are devices that are used postoperatively following endoscopic sinus surgery (ESS). The
Page: 1 of 8 Last Review Status/Date: March 2015 Use Following Endoscopic Sinus Surgery Description Sinus stents are devices that are used postoperatively following endoscopic sinus surgery (ESS). The
POLICY PRODUCT VARIATIONS DESCRIPTION/BACKGROUND RATIONALE DEFINITIONS BENEFIT VARIATIONS DISCLAIMER CODING INFORMATION REFERENCES POLICY HISTORY
 Original Issue Date (Created): July 30, 2013 Most Recent Review Date (Revised): May 20, 2014 Effective Date: August 1, 2014 POLICY PRODUCT VARIATIONS DESCRIPTION/BACKGROUND RATIONALE DEFINITIONS BENEFIT
Original Issue Date (Created): July 30, 2013 Most Recent Review Date (Revised): May 20, 2014 Effective Date: August 1, 2014 POLICY PRODUCT VARIATIONS DESCRIPTION/BACKGROUND RATIONALE DEFINITIONS BENEFIT
Policy Effective January 29, 2019
 Corporate Medical Policy Surgical Treatment of Sinus Disease File Name: Origination: Last CAP Review: Next CAP Review: Last Review: surgical_treatment_of_sinus_disease 2/2010 8/2018 8/2019 8/2018 Policy
Corporate Medical Policy Surgical Treatment of Sinus Disease File Name: Origination: Last CAP Review: Next CAP Review: Last Review: surgical_treatment_of_sinus_disease 2/2010 8/2018 8/2019 8/2018 Policy
Implantable Sinus Stents for Postoperative Use Following Endoscopic Sinus Surgery and for Recurrent Sinonasal Polyposis
 Medical Policy Manual Surgery, Policy No. 198 Implantable Sinus Stents for Postoperative Use Following Endoscopic Sinus Surgery and for Recurrent Sinonasal Polyposis Next Review: August 2019 Last Review:
Medical Policy Manual Surgery, Policy No. 198 Implantable Sinus Stents for Postoperative Use Following Endoscopic Sinus Surgery and for Recurrent Sinonasal Polyposis Next Review: August 2019 Last Review:
Implantable Sinus Stents for Postoperative Use Following Endoscopic Sinus Surgery and for Recurrent Sinus Disease
 Page: 1 of 8 Last Review Status/Date: March 2017 Implantable Sinus Stents for Postoperative Use Following Endoscopic Sinus Surgery and Description Sinus stents are devices used postoperatively following
Page: 1 of 8 Last Review Status/Date: March 2017 Implantable Sinus Stents for Postoperative Use Following Endoscopic Sinus Surgery and Description Sinus stents are devices used postoperatively following
Implantable Sinus Stents for Postoperative Use Following Endoscopic Sinus Surgery and for Recurrent Sinus Disease
 Implantable Sinus Stents for Postoperative Use Following Endoscopic Sinus Surgery and for Recurrent Sinus Disease Policy Number: 7.01.134 Last Review: 8/2018 Origination: 12/2015 Next Review: 12/2018 Policy
Implantable Sinus Stents for Postoperative Use Following Endoscopic Sinus Surgery and for Recurrent Sinus Disease Policy Number: 7.01.134 Last Review: 8/2018 Origination: 12/2015 Next Review: 12/2018 Policy
MP Implantable Sinus Stents for Postoperative Use Following Endoscopic Sinus Surgery and for Recurrent Sinus Disease. Related Policies None
 Medical Policy MP 7.01.134 Implantable Sinus Stents for Postoperative Use Following Endoscopic Sinus Surgery and for Recurrent Sinus BCBSA Ref. Policy: 7.01.134 Last Review: 02/26/2018 Effective Date:
Medical Policy MP 7.01.134 Implantable Sinus Stents for Postoperative Use Following Endoscopic Sinus Surgery and for Recurrent Sinus BCBSA Ref. Policy: 7.01.134 Last Review: 02/26/2018 Effective Date:
Functional Endoscopic Sinus Surgery (FESS)
 Functional Endoscopic Sinus Surgery (FESS) Last Review Date: December 12, 2017 Number: MG.MM.SU.56C2 Medical Guideline Disclaimer Property of EmblemHealth. All rights reserved. The treating physician or
Functional Endoscopic Sinus Surgery (FESS) Last Review Date: December 12, 2017 Number: MG.MM.SU.56C2 Medical Guideline Disclaimer Property of EmblemHealth. All rights reserved. The treating physician or
Treatment Options for Chronic Sinusitis
 Treatment Options for Chronic Sinusitis Jesse Ryan, M.D. Assistant Professor Head and Neck Surgery & Reconstruction Department of Otolaryngology January 17, 2019 Disclosures I have no financial relationship
Treatment Options for Chronic Sinusitis Jesse Ryan, M.D. Assistant Professor Head and Neck Surgery & Reconstruction Department of Otolaryngology January 17, 2019 Disclosures I have no financial relationship
White Paper: Balloon Sinuplasty for Chronic Sinusitis, The Latest Recommendations
 White Paper: Balloon Sinuplasty for Chronic Sinusitis, The Latest Recommendations For Health Plans, Medical Management Organizations and TPAs Executive Summary Despite recent advances in instrumentation
White Paper: Balloon Sinuplasty for Chronic Sinusitis, The Latest Recommendations For Health Plans, Medical Management Organizations and TPAs Executive Summary Despite recent advances in instrumentation
FRONTAL SINUPLASTY P R E P A R E D A N D P R E S E N T E D B Y : D R. Y A H Y A F A G E E H R 4 16/ 12/ 2013
 FRONTAL SINUPLASTY P R E P A R E D A N D P R E S E N T E D B Y : D R. Y A H Y A F A G E E H R 4 16/ 12/ 2013 ANATOMY: FRONTAL SINUS Not present at birth Starts developing at 4 years Radiographically visualized
FRONTAL SINUPLASTY P R E P A R E D A N D P R E S E N T E D B Y : D R. Y A H Y A F A G E E H R 4 16/ 12/ 2013 ANATOMY: FRONTAL SINUS Not present at birth Starts developing at 4 years Radiographically visualized
Clinical Policy Title: Propel (drug eluting devices after sinus surgery)
 Clinical Policy Title: Propel (drug eluting devices after sinus surgery) Clinical Policy Number: 10.03.07 Effective Date: July 1, 2017 Initial Review Date: May 19, 2017 Most Recent Review Date: May 1,
Clinical Policy Title: Propel (drug eluting devices after sinus surgery) Clinical Policy Number: 10.03.07 Effective Date: July 1, 2017 Initial Review Date: May 19, 2017 Most Recent Review Date: May 1,
2017 FACILITY AND PHYSICIAN REIMBURSEMENT GUIDE
 2017 AND PHYSICIAN REIMBURSEMENT GUIDE NASAL/SINUS ENDOSCOPIC SURGERY Some of the Current Procedure Terminology (CPT ) Codes for endoscopic nasal/sinus surgery are listed below. CPT codes 31295, 31296
2017 AND PHYSICIAN REIMBURSEMENT GUIDE NASAL/SINUS ENDOSCOPIC SURGERY Some of the Current Procedure Terminology (CPT ) Codes for endoscopic nasal/sinus surgery are listed below. CPT codes 31295, 31296
Clinical Policy Title: Propel (drug eluting devices after sinus surgery)
 Clinical Policy Title: Propel (drug eluting devices after sinus surgery) Clinical Policy Number: 10.03.07 Effective Date: July 1, 2017 Initial Review Date: May 19, 2017 Most Recent Review Date: June 22,
Clinical Policy Title: Propel (drug eluting devices after sinus surgery) Clinical Policy Number: 10.03.07 Effective Date: July 1, 2017 Initial Review Date: May 19, 2017 Most Recent Review Date: June 22,
MEDICAL POLICY EFFECTIVE DATE: 04/21/11 REVISED DATE: 06/21/12, 05/23/13, 06/19/14, 06/18/15, 06/16/16, 06/15/17
 MEDICAL POLICY SUBJECT: SINUS OSTIAL DILATION FOR PAGE: 1 OF: 6 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial product, including
MEDICAL POLICY SUBJECT: SINUS OSTIAL DILATION FOR PAGE: 1 OF: 6 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial product, including
Rhinosinusitis. John Ramey, MD Joseph Russell, MD
 Rhinosinusitis John Ramey, MD Joseph Russell, MD Disclosure Statement RSFH as a continuing medical education provider, accredited by the South Carolina Medical Association, it is the policy of RSFH to
Rhinosinusitis John Ramey, MD Joseph Russell, MD Disclosure Statement RSFH as a continuing medical education provider, accredited by the South Carolina Medical Association, it is the policy of RSFH to
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE
 NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Medical technology guidance SCOPE The XprESS Multi-Sinus Dilation System for the treatment of 1 Technology 1.1 Description of the technology The XprESS
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Medical technology guidance SCOPE The XprESS Multi-Sinus Dilation System for the treatment of 1 Technology 1.1 Description of the technology The XprESS
DIFFICULT-TO-TREAT CHRONIC
 MANAGEMENT STRATEGIES FOR DIFFICULT-TO-TREAT CHRONIC RHINOSINUSITIS DR ZULKEFLI HUSSEIN CONSULTANT EAR NOSE & THROAT SURGEON PANTAI HOSPITAL PENANG DISCLAIMER Nothing to disclose PENANG ISLAND, MALAYSIA
MANAGEMENT STRATEGIES FOR DIFFICULT-TO-TREAT CHRONIC RHINOSINUSITIS DR ZULKEFLI HUSSEIN CONSULTANT EAR NOSE & THROAT SURGEON PANTAI HOSPITAL PENANG DISCLAIMER Nothing to disclose PENANG ISLAND, MALAYSIA
Surgical Management of Sinusitis (What About Balloons?) Relative Indications for Sinus Surgery in Children
 Surgical Management of Sinusitis (What About Balloons?) Andrew N. Goldberg M.D. Andrew H. Murr M.D. Michael J. Cunningham, M.D. Department of Otolaryngology and Communication Enhancement Children s Hospital
Surgical Management of Sinusitis (What About Balloons?) Andrew N. Goldberg M.D. Andrew H. Murr M.D. Michael J. Cunningham, M.D. Department of Otolaryngology and Communication Enhancement Children s Hospital
FUNCTIONAL ENDOSCOPIC SINUS SURGERY (FESS)
 FUNCTIONAL ENDOSCOPIC SINUS SURGERY (FESS) Protocol: SUR055 Effective Date: June 1, 2018 Table of Contents Page COMMERCIAL, MEDICAID AND MEDICARE COVERAGE RATIONALE... 1 DEFINITIONS... 2 DESCRIPTION OF
FUNCTIONAL ENDOSCOPIC SINUS SURGERY (FESS) Protocol: SUR055 Effective Date: June 1, 2018 Table of Contents Page COMMERCIAL, MEDICAID AND MEDICARE COVERAGE RATIONALE... 1 DEFINITIONS... 2 DESCRIPTION OF
FUNCTIONAL ENDOSCOPIC SINUS SURGERY (FESS)
 UnitedHealthcare Community Plan Medical Policy FUNCTIONAL ENDOSCOPIC SINUS SURGERY (FESS) Policy Number: CS144.D Effective Date: January 1, 2018 Table of Contents Page INSTRUCTIONS FOR USE... 1 BENEFIT
UnitedHealthcare Community Plan Medical Policy FUNCTIONAL ENDOSCOPIC SINUS SURGERY (FESS) Policy Number: CS144.D Effective Date: January 1, 2018 Table of Contents Page INSTRUCTIONS FOR USE... 1 BENEFIT
Medical Policy An independent licensee of the Blue Cross Blue Shield Association
 Balloon Sinuplasty for Treatment of Chronic Sinusitis Page 1 of 18 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Balloon Sinuplasty for Treatment of Chronic Sinusitis
Balloon Sinuplasty for Treatment of Chronic Sinusitis Page 1 of 18 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Balloon Sinuplasty for Treatment of Chronic Sinusitis
Cigna Medical Coverage Policy
 Cigna Medical Coverage Policy Subject Drug-Eluting Devices for Use Following Endoscopic Sinus Surgery Table of Contents Coverage Policy... 1 General Background... 1 Coding/Billing Information... 6 References...
Cigna Medical Coverage Policy Subject Drug-Eluting Devices for Use Following Endoscopic Sinus Surgery Table of Contents Coverage Policy... 1 General Background... 1 Coding/Billing Information... 6 References...
Protocol. Balloon Ostial Dilation for Treatment of Chronic Rhinosinusitis
 Protocol Balloon Ostial Dilation for Treatment of Chronic Rhinosinusitis Medical Benefit Effective Date: 04/01/15 Next Review Date: 11/19 Preauthorization No Review Dates: 07/07, 07/08, 11/08, 05/09, 01/10,
Protocol Balloon Ostial Dilation for Treatment of Chronic Rhinosinusitis Medical Benefit Effective Date: 04/01/15 Next Review Date: 11/19 Preauthorization No Review Dates: 07/07, 07/08, 11/08, 05/09, 01/10,
FUNCTIONAL ENDOSCOPIC SINUS SURGERY (FESS)
 UnitedHealthcare Oxford Clinical Policy FUNCTIONAL ENDOSCOPIC SINUS SURGERY (FESS) Policy Number: ENT 022.5 T2 Effective Date: April 1, 2018 Table of Contents Page INSTRUCTIONS FOR USE... 1 CONDITIONS
UnitedHealthcare Oxford Clinical Policy FUNCTIONAL ENDOSCOPIC SINUS SURGERY (FESS) Policy Number: ENT 022.5 T2 Effective Date: April 1, 2018 Table of Contents Page INSTRUCTIONS FOR USE... 1 CONDITIONS
An Innovative Treatment Option for Patients with Recurrent Nasal Polyps
 An Innovative Treatment Option for Patients with Recurrent Nasal Polyps Burden of illness and management of Chronic Sinusitis with Nasal Polyps Continuum of care and polyp recurrence Clinical and health
An Innovative Treatment Option for Patients with Recurrent Nasal Polyps Burden of illness and management of Chronic Sinusitis with Nasal Polyps Continuum of care and polyp recurrence Clinical and health
FUNCTIONAL ENDOSCOPIC SINUS SURGERY (FESS)
 UnitedHealthcare Oxford Clinical Policy FUNCTIONAL ENDOSCOPIC SINUS SURGERY (FESS) Policy Number: ENT 022.3 T2 Effective Date: October 1, 2017 Table of Contents Page INSTRUCTIONS FOR USE... 1 CONDITIONS
UnitedHealthcare Oxford Clinical Policy FUNCTIONAL ENDOSCOPIC SINUS SURGERY (FESS) Policy Number: ENT 022.3 T2 Effective Date: October 1, 2017 Table of Contents Page INSTRUCTIONS FOR USE... 1 CONDITIONS
Clinical Policy: Balloon Sinus Ostial Dilation for Treatment of Chronic Sinusitis
 Clinical Policy: Balloon Sinus Ostial Dilation for Treatment of Chronic Sinusitis Reference Number: CP.MP.119 Last Review Date: 11/17 See Important Reminder at the end of this policy for important regulatory
Clinical Policy: Balloon Sinus Ostial Dilation for Treatment of Chronic Sinusitis Reference Number: CP.MP.119 Last Review Date: 11/17 See Important Reminder at the end of this policy for important regulatory
Nasal Polyposis. DEPARTMENT OF ENT K.S.Hegde Medical Academy Deralakatte, Mangalore
 Nasal Polyposis DEPARTMENT OF ENT K.S.Hegde Medical Academy Deralakatte, Mangalore Def: INTRODUCTION Chronic inflammatory disease of the mucous membrane in the nose & PNS, presenting as pedunculated smooth
Nasal Polyposis DEPARTMENT OF ENT K.S.Hegde Medical Academy Deralakatte, Mangalore Def: INTRODUCTION Chronic inflammatory disease of the mucous membrane in the nose & PNS, presenting as pedunculated smooth
Medical Policy An independent licensee of the Blue Cross Blue Shield Association
 Balloon Sinuplasty for Treatment of Chronic Sinusitis Page 1 of 17 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Balloon Sinuplasty for Treatment of Chronic Sinusitis
Balloon Sinuplasty for Treatment of Chronic Sinusitis Page 1 of 17 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Balloon Sinuplasty for Treatment of Chronic Sinusitis
9/18/2018. Disclosures. Objectives
 Is It Really Acute Bacterial Rhinosinusitis? Assessment, Differential Diagnosis and Management of Common Sinonasal Symptoms Kristina Haralambides, MS, RN, FNP-C Disclosures The content of this presentation
Is It Really Acute Bacterial Rhinosinusitis? Assessment, Differential Diagnosis and Management of Common Sinonasal Symptoms Kristina Haralambides, MS, RN, FNP-C Disclosures The content of this presentation
Description. Section: Surgery Effective Date: April 15, 2014 Subsection: Surgery Original Policy Date: June 7, 2012 Subject: Page: 1 of 12
 Last Review Status/Date: March 2014 Page: 1 of 12 Description Balloon ostial dilation (also known as balloon sinuplasty ) is proposed as an alternative to traditional endoscopic sinus surgery for patients
Last Review Status/Date: March 2014 Page: 1 of 12 Description Balloon ostial dilation (also known as balloon sinuplasty ) is proposed as an alternative to traditional endoscopic sinus surgery for patients
Hitting the Jackpot Sinuses, Scopes, and Surgery
 Hitting the Jackpot Sinuses, Scopes, and Surgery Candice Fenildo holds an Associates Degree in Health Sciences and is Certified in Healthcare Compliance (CHC), a Certified Professional Coder (CPC), a Certified
Hitting the Jackpot Sinuses, Scopes, and Surgery Candice Fenildo holds an Associates Degree in Health Sciences and is Certified in Healthcare Compliance (CHC), a Certified Professional Coder (CPC), a Certified
Description. Section: Surgery Effective Date: April 15, 2017 Subsection: Surgery Original Policy Date: June 7, 2012 Subject: Page: 1 of 13
 Last Review Status/Date: March 2017 Page: 1 of 13 Description Balloon ostial dilation (also known as balloon sinuplasty ) is proposed as an alternative to traditional endoscopic sinus surgery for patients
Last Review Status/Date: March 2017 Page: 1 of 13 Description Balloon ostial dilation (also known as balloon sinuplasty ) is proposed as an alternative to traditional endoscopic sinus surgery for patients
Delivering Innovation. Where It s Needed. August 2018
 Delivering Innovation. Where It s Needed. August 2018 Forward-Looking Statements Certain statements in this presentation constitute forward-looking statements within the meaning of the Securities Act of
Delivering Innovation. Where It s Needed. August 2018 Forward-Looking Statements Certain statements in this presentation constitute forward-looking statements within the meaning of the Securities Act of
Functional Endoscopic Sinus Surgery
 WHAT IS FUNCTIONAL ENDOSCOPIC SINUS SURGERY (FESS)? The nasal telescope has greatly changes the evaluation and treatment of rhino-sinusitis. This instrument, which provides a view of the structures in
WHAT IS FUNCTIONAL ENDOSCOPIC SINUS SURGERY (FESS)? The nasal telescope has greatly changes the evaluation and treatment of rhino-sinusitis. This instrument, which provides a view of the structures in
Topic: Balloon Catheter Dilation for Treatment of Sinusitis Date of Origin: August 8, Section: Surgery Last Reviewed Date: February 2014
 Medical Policy Manual Topic: Balloon Catheter Dilation for Treatment of Sinusitis Date of Origin: August 8, 2006 Section: Surgery Last Reviewed Date: February 2014 Policy No: 153 Effective Date: April
Medical Policy Manual Topic: Balloon Catheter Dilation for Treatment of Sinusitis Date of Origin: August 8, 2006 Section: Surgery Last Reviewed Date: February 2014 Policy No: 153 Effective Date: April
In-office, multisinus balloon dilation: 1-Year outcomes from a prospective, multicenter, open label trial DO NOT COPY METHODS.
 In-office, multisinus balloon dilation: 1-Year outcomes from a prospective, multicenter, open label trial James Gould, M.D., 1 Ian Alexander, M.D., 2 Edward Tomkin, D.O., 3 and David Brodner, M.D. 4 ABSTRACT
In-office, multisinus balloon dilation: 1-Year outcomes from a prospective, multicenter, open label trial James Gould, M.D., 1 Ian Alexander, M.D., 2 Edward Tomkin, D.O., 3 and David Brodner, M.D. 4 ABSTRACT
Corporate Medical Policy Septoplasty
 Corporate Medical Policy Septoplasty File Name: Origination: Last CAP Review: Next CAP Review: Last Review: septoplasty 4/1999 8/2018 8/2019 8/2018 Description of Procedure or Service There are many potential
Corporate Medical Policy Septoplasty File Name: Origination: Last CAP Review: Next CAP Review: Last Review: septoplasty 4/1999 8/2018 8/2019 8/2018 Description of Procedure or Service There are many potential
1. BRIEF DESCRIPTION OF TRAINING
 RHINOLOGY 1. BRIEF DESCRIPTION OF TRAINING Exposure to clinical rhinology is provided in each of the four ORL years over the course of several rotations in a graduated approach. MEE General Otolaryngology
RHINOLOGY 1. BRIEF DESCRIPTION OF TRAINING Exposure to clinical rhinology is provided in each of the four ORL years over the course of several rotations in a graduated approach. MEE General Otolaryngology
Sinusitis: Medical and Surgical Management. R. Jonathan Lara, DO, FAOCO. Sonoran Ear Nose & Throat
 Sinusitis: Medical and Surgical Management R. Jonathan Lara, DO, FAOCO Sonoran Ear Nose & Throat 27 th Annual Southwestern Conference on Medicine April 26, 2018 What are sinuses? Air filled cavities of
Sinusitis: Medical and Surgical Management R. Jonathan Lara, DO, FAOCO Sonoran Ear Nose & Throat 27 th Annual Southwestern Conference on Medicine April 26, 2018 What are sinuses? Air filled cavities of
MEDICAL POLICY I. POLICY FUNCTIONAL ENDOSCOPIC SINUS SURGERY FOR CHRONIC RHINOSINUSITIS MP POLICY TITLE POLICY NUMBER
 Original Issue Date (Created): 8/1/2018 Most Recent Review Date (Revised): 1/30/2018 Effective Date: 8/1/2018 POLICY PRODUCT VARIATIONS DESCRIPTION/BACKGROUND RATIONALE DEFINITIONS BENEFIT VARIATIONS DISCLAIMER
Original Issue Date (Created): 8/1/2018 Most Recent Review Date (Revised): 1/30/2018 Effective Date: 8/1/2018 POLICY PRODUCT VARIATIONS DESCRIPTION/BACKGROUND RATIONALE DEFINITIONS BENEFIT VARIATIONS DISCLAIMER
Sinusitis: Medical and Surgical Management. R. Jonathan Lara, DO, FAOCO. Sonoran Ear Nose & Throat
 Sinusitis: Medical and Surgical Management R. Jonathan Lara, DO, FAOCO Sonoran Ear Nose & Throat 27 th Annual Southwestern Conference on Medicine April 26, 2018 What are sinuses? Air filled cavities of
Sinusitis: Medical and Surgical Management R. Jonathan Lara, DO, FAOCO Sonoran Ear Nose & Throat 27 th Annual Southwestern Conference on Medicine April 26, 2018 What are sinuses? Air filled cavities of
Medicare Advantage Medical Policy. Title: Balloon Ostial Dilation for Treatment of Chronic Rhinosinusitis
 Medicare Advantage Medical Policy Current Policy Effective Date: 1/1/18 Title: Balloon Ostial Dilation for Treatment of Chronic Rhinosinusitis Description/Background CHRONIC RHINOSINUSITIS Chronic rhinosinusitis
Medicare Advantage Medical Policy Current Policy Effective Date: 1/1/18 Title: Balloon Ostial Dilation for Treatment of Chronic Rhinosinusitis Description/Background CHRONIC RHINOSINUSITIS Chronic rhinosinusitis
Archives of Otolaryngology and Rhinology
 v Clinical Group Archives of Otolaryngology and Rhinology ISSN: 2455-1759 DOI CC By Michael Schlewet* and Peter Catalano # Department of Otolaryngology, Head and Neck Surgery, St Elizabeth s Medical Center,
v Clinical Group Archives of Otolaryngology and Rhinology ISSN: 2455-1759 DOI CC By Michael Schlewet* and Peter Catalano # Department of Otolaryngology, Head and Neck Surgery, St Elizabeth s Medical Center,
2019 ACCLARENT REIMBURSEMENT GUIDE. Physician and Facility
 2019 ACCLARENT REIMBURSEMENT GUIDE Physician and Facility TABLE OF CONTENTS This guide has been developed to assist you in obtaining physician and facility reimbursement for: Nasal/Sinus Endoscopic Surgery
2019 ACCLARENT REIMBURSEMENT GUIDE Physician and Facility TABLE OF CONTENTS This guide has been developed to assist you in obtaining physician and facility reimbursement for: Nasal/Sinus Endoscopic Surgery
Conventional Sinus Surgery Vs Fess
 IOSR Journal of Dental and Medical Sciences (IOSR-JDMS) e-issn: 2279-0853, p-issn: 2279-0861.Volume 16, Issue 7 Ver. III (July. 2017), PP 44-51 www.iosrjournals.org Conventional Sinus Surgery Vs Fess *
IOSR Journal of Dental and Medical Sciences (IOSR-JDMS) e-issn: 2279-0853, p-issn: 2279-0861.Volume 16, Issue 7 Ver. III (July. 2017), PP 44-51 www.iosrjournals.org Conventional Sinus Surgery Vs Fess *
FUNCTIONAL ENDOSCOPIC SINUS SURGERY (FESS)
 Oxford UnitedHealthcare Oxford Clinical Policy FUNCTIONAL ENDOSCOPIC SINUS SURGERY (FESS) Policy Number: ENT 022.6 T2 Effective Date: October 1, 2018 Table of Contents Page INSTRUCTIONS FOR USE... 1 CONDITIONS
Oxford UnitedHealthcare Oxford Clinical Policy FUNCTIONAL ENDOSCOPIC SINUS SURGERY (FESS) Policy Number: ENT 022.6 T2 Effective Date: October 1, 2018 Table of Contents Page INSTRUCTIONS FOR USE... 1 CONDITIONS
Sinus Surgery (Functional Endoscopic Sinus Surgery and Balloon Sinuplasty)
 Sinus Surgery (Functional Endoscopic Sinus Surgery and Balloon Sinuplasty) Date of Origin: 10/2015 Last Review Date: 06/28/2017 Effective Date: 07/01/2017 Dates Reviewed: 10/2015, 06/2017 Developed By:
Sinus Surgery (Functional Endoscopic Sinus Surgery and Balloon Sinuplasty) Date of Origin: 10/2015 Last Review Date: 06/28/2017 Effective Date: 07/01/2017 Dates Reviewed: 10/2015, 06/2017 Developed By:
Site of Service: Select Surgical Procedures. Select a hyperlink below to be directed to that section.
 MEDICAL POLICY 7.01.559 Sinus Surgery BCBSA Ref. Policies: 7.01.105, 7.01.155 Effective Date: June 1, 2018 RELATED MEDICAL POLICIES: Last Revised: Sept. 21, 2018 7.01.558 Rhinoplasty Replaces: N/A 11.01.524
MEDICAL POLICY 7.01.559 Sinus Surgery BCBSA Ref. Policies: 7.01.105, 7.01.155 Effective Date: June 1, 2018 RELATED MEDICAL POLICIES: Last Revised: Sept. 21, 2018 7.01.558 Rhinoplasty Replaces: N/A 11.01.524
SINUS SURGERY. Dr Zenia Chow MBBS(hons), FRACS
 SINUS SURGERY Dr Zenia Chow MBBS(hons), FRACS Facial Plastic & Reconstructive Surgeon Otolaryngology, Head and Neck Surgeon ENDOSCOPIC SINUS SURGERY/FESS What are sinuses The sinuses are a connected system
SINUS SURGERY Dr Zenia Chow MBBS(hons), FRACS Facial Plastic & Reconstructive Surgeon Otolaryngology, Head and Neck Surgeon ENDOSCOPIC SINUS SURGERY/FESS What are sinuses The sinuses are a connected system
Patency of Maxillary Sinus Ostia Following Dilation with a Novel Osmotic Expansion Device
 WHITE PAPER Patency of Maxillary Sinus Ostia Following Dilation with a Novel Osmotic Expansion Device Six-Month Results from a Multi-Center Prospective Study Jerome Hester, MD California Sleep Institute,
WHITE PAPER Patency of Maxillary Sinus Ostia Following Dilation with a Novel Osmotic Expansion Device Six-Month Results from a Multi-Center Prospective Study Jerome Hester, MD California Sleep Institute,
Disclaimers. Topical Therapy. The Problem. Topical Therapy for Chronic Rhinosinusitis No Disclosures
 Topical Therapy for Chronic Rhinosinusitis No Disclosures Disclaimers Off-label use of multiple steroid and antibiotic medications Large talk, limited time Steven D. Pletcher MD University of California,
Topical Therapy for Chronic Rhinosinusitis No Disclosures Disclaimers Off-label use of multiple steroid and antibiotic medications Large talk, limited time Steven D. Pletcher MD University of California,
Commen Nose Diseases
 Commen Nose Diseases Symptoms List: Nasal obstruction. Nasal discharge: Anterior (Rhinorrhea). Posterior (Postnasal discharge). Epistaxis. Hyposmia and Anosmia. Headache. Snoring. Nasal Obstruction Definition:
Commen Nose Diseases Symptoms List: Nasal obstruction. Nasal discharge: Anterior (Rhinorrhea). Posterior (Postnasal discharge). Epistaxis. Hyposmia and Anosmia. Headache. Snoring. Nasal Obstruction Definition:
The Nose and Sinuses. Ophir Ilan, MD, PhD Department of Otolaryngology/Head&Neck surgery Hadassah University Hospital
 The Nose and Sinuses Ophir Ilan, MD, PhD Department of Otolaryngology/Head&Neck surgery Hadassah University Hospital Nasal Mucociliary System Function of the Nasal Mucosa warming and humidifying the
The Nose and Sinuses Ophir Ilan, MD, PhD Department of Otolaryngology/Head&Neck surgery Hadassah University Hospital Nasal Mucociliary System Function of the Nasal Mucosa warming and humidifying the
Sphenoid rhinosinusitis associated with abducens nerve palsy Case report
 Romanian Journal of Rhinology, Volume 8, No. 30, April-June 2018 CASE REPORT Sphenoid rhinosinusitis associated with abducens nerve palsy Case report Lucian Lapusneanu 1, Marlena Radulescu 1, Florin Ghita
Romanian Journal of Rhinology, Volume 8, No. 30, April-June 2018 CASE REPORT Sphenoid rhinosinusitis associated with abducens nerve palsy Case report Lucian Lapusneanu 1, Marlena Radulescu 1, Florin Ghita
FOR QUESTIONS PLEASE CONTACT US AT
 NASAL AND SINUS ENDOSCOPY PROCEDURES EFFECTIVE JANUARY 2018 Medtronic provides this information for your convenience only. It does not constitute legal advice or a recommendation regarding clinical practice.
NASAL AND SINUS ENDOSCOPY PROCEDURES EFFECTIVE JANUARY 2018 Medtronic provides this information for your convenience only. It does not constitute legal advice or a recommendation regarding clinical practice.
MANAGEMENT OF RHINOSINUSITIS IN ADOLESCENTS AND ADULTS
 MANAGEMENT OF RHINOSINUSITIS IN ADOLESCENTS AND ADULTS Ministry of Health Malaysia Malaysian Society of Otorhinolaryngologist - Head & Neck Surgeons (MS)-HNS) Academy of Medicine Malaysia KEY MESSAGES
MANAGEMENT OF RHINOSINUSITIS IN ADOLESCENTS AND ADULTS Ministry of Health Malaysia Malaysian Society of Otorhinolaryngologist - Head & Neck Surgeons (MS)-HNS) Academy of Medicine Malaysia KEY MESSAGES
Medical technologies guidance Published: 13 December 2016 nice.org.uk/guidance/mtg30
 XprESS multi sinus dilation system for treating chronic sinusitis Medical technologies guidance Published: 13 December 2016 nice.org.uk/guidance/mtg30 NICE 2018. All rights reserved. Subject to Notice
XprESS multi sinus dilation system for treating chronic sinusitis Medical technologies guidance Published: 13 December 2016 nice.org.uk/guidance/mtg30 NICE 2018. All rights reserved. Subject to Notice
7/28/2017. Disclaimer. AOA Practice Builder Session: Coding & Coverage of Drug eluting Sinus Implants. Introduction and Agenda
 AOA Practice Builder Session: Coding & Coverage of Drug eluting Sinus Implants Betsy Gross, RN, BSN, CPC, CENTC Vice President, Health Economics & Reimbursement DELIVERING INNOVATION. Intersect ENT WHERE
AOA Practice Builder Session: Coding & Coverage of Drug eluting Sinus Implants Betsy Gross, RN, BSN, CPC, CENTC Vice President, Health Economics & Reimbursement DELIVERING INNOVATION. Intersect ENT WHERE
MANAGEMENT OF RHINOSINUSITIS IN ADULTS IN PRIMARY CARE
 PROFESSOR DR SALINA HUSAIN DEPUTY HEAD DEPARTMENT OF OTORHINOLARYNGOLOGY-HEAD NECK SURGERY UKM MEDICAL CENTRE MANAGEMENT OF RHINOSINUSITIS IN ADULTS IN PRIMARY CARE CLINICAL PRACTICE GUIDELINES ON MANAGEMENT
PROFESSOR DR SALINA HUSAIN DEPUTY HEAD DEPARTMENT OF OTORHINOLARYNGOLOGY-HEAD NECK SURGERY UKM MEDICAL CENTRE MANAGEMENT OF RHINOSINUSITIS IN ADULTS IN PRIMARY CARE CLINICAL PRACTICE GUIDELINES ON MANAGEMENT
Balloon Sinuplasty: Indications and Cost-Effectiveness
 Balloon Sinuplasty: Indications and Cost-Effectiveness Marilene B. Wang, MD Professor, Department of Head and Neck Surgery David Geffen School of Medicine at UCLA Chief of Otolaryngology VA Greater Los
Balloon Sinuplasty: Indications and Cost-Effectiveness Marilene B. Wang, MD Professor, Department of Head and Neck Surgery David Geffen School of Medicine at UCLA Chief of Otolaryngology VA Greater Los
Chronic Sinusitis. Acute Sinusitis. Sinusitis. Anatomy of the Paranasal Sinuses. Sinusitis. Medical Topics - Sinusitis
 1 Acute Chronic is the inflammation of the inner lining of the parnasal sinuses due to infection or non-infectious causes such as allergies or environmental pullutants. If the inflammation lasts more than
1 Acute Chronic is the inflammation of the inner lining of the parnasal sinuses due to infection or non-infectious causes such as allergies or environmental pullutants. If the inflammation lasts more than
BALLOON SINUS OSTIAL DILATION
 BALLOON SINUS OSTIAL DILATION UnitedHealthcare Oxford Clinical Policy Policy Number: ENT 021.6 T2 Effective Date: April 1, 2018 Table of Contents Page INSTRUCTIONS FOR USE... 1 CONDITIONS OF COVERAGE...
BALLOON SINUS OSTIAL DILATION UnitedHealthcare Oxford Clinical Policy Policy Number: ENT 021.6 T2 Effective Date: April 1, 2018 Table of Contents Page INSTRUCTIONS FOR USE... 1 CONDITIONS OF COVERAGE...
MedStar Health considers Septoplasty-Rhinoplasty medically necessary for the following indications:
 MedStar Health, Inc. POLICY AND PROCEDURE MANUAL MP.038.MH Septoplasty-Rhinoplasty This policy applies to the following lines of business: MedStar Employee (Select) MedStar MA DSNP CSNP MedStar CareFirst
MedStar Health, Inc. POLICY AND PROCEDURE MANUAL MP.038.MH Septoplasty-Rhinoplasty This policy applies to the following lines of business: MedStar Employee (Select) MedStar MA DSNP CSNP MedStar CareFirst
Balloon Ostial Dilation for Treatment of Chronic Rhinosinusitis
 Balloon Ostial Dilation for Treatment of Chronic Rhinosinusitis Policy Number: 7.01.105 Last Review: 3/2018 Origination: 3/2007 Next Review: 9/2018 Policy Blue Cross and Blue Shield of Kansas City (Blue
Balloon Ostial Dilation for Treatment of Chronic Rhinosinusitis Policy Number: 7.01.105 Last Review: 3/2018 Origination: 3/2007 Next Review: 9/2018 Policy Blue Cross and Blue Shield of Kansas City (Blue
Congestion, headache, recurrent infection, post-nasal drip, smell problems? We can find the source and offer solutions for relief.
 Sinus Sinus Congestion, headache, recurrent infection, post-nasal drip, smell problems? We can find the source and offer solutions for relief. So what is sinusitis? Although many individuals interpret
Sinus Sinus Congestion, headache, recurrent infection, post-nasal drip, smell problems? We can find the source and offer solutions for relief. So what is sinusitis? Although many individuals interpret
+ Conflict of interest. + Sinus and Nasal Anatomy. + What is your diagnosis? 1) Allergic Rhinitis. 2) Non-Allergic rhinitis. 3) Chronic Rhinosinusitis
 Rhinitis & Sinusitis Conflict of interest I have no conflict of interest to declare for this lecture Al Chiodo, MD FRCSC Assistant Professor Director of Undergraduate Medical Education Department of Otolaryngology-Head
Rhinitis & Sinusitis Conflict of interest I have no conflict of interest to declare for this lecture Al Chiodo, MD FRCSC Assistant Professor Director of Undergraduate Medical Education Department of Otolaryngology-Head
Nasal Polyps. Multimedia Health Education. Disclaimer
 Disclaimer This movie is an educational resource only and should not be used to manage your health. All decisions about the management of Nasal Polyps must be made in conjunction with your Physician or
Disclaimer This movie is an educational resource only and should not be used to manage your health. All decisions about the management of Nasal Polyps must be made in conjunction with your Physician or
Reasons for Failure and Surgical Revisions. Stil Kountakis, MD, PhD Professor and Chief, Division of Rhinology
 Reasons for Failure and Surgical Revisions Stil Kountakis, MD, PhD Professor and Chief, Division of Rhinology Medical College of Georgia of Georgia Regents University Department of Otolaryngology / Head
Reasons for Failure and Surgical Revisions Stil Kountakis, MD, PhD Professor and Chief, Division of Rhinology Medical College of Georgia of Georgia Regents University Department of Otolaryngology / Head
National Imaging Associates, Inc. Clinical guidelines/considerations SINUS & MAXILLOFACIAL AREA CT 70486, 70487, 70488
 National Imaging Associates, Inc. Clinical guidelines/considerations SINUS & MAXILLOFACIAL AREA CT 70486, 70487, 70488 Date: September 1997 Page 1 of 5 LIMITED OR LOCALIZED FOLLOW UP - SINUS CT 76380 Guideline
National Imaging Associates, Inc. Clinical guidelines/considerations SINUS & MAXILLOFACIAL AREA CT 70486, 70487, 70488 Date: September 1997 Page 1 of 5 LIMITED OR LOCALIZED FOLLOW UP - SINUS CT 76380 Guideline
CHRONIC RHINOSINUSITIS IN ADULTS
 CHRONIC RHINOSINUSITIS IN ADULTS SCOPE OF THE PRACTICE GUIDELINE This clinical practice guideline is for use by the Philippine Society of Otolaryngology-Head and Neck Surgery. It covers the diagnosis and
CHRONIC RHINOSINUSITIS IN ADULTS SCOPE OF THE PRACTICE GUIDELINE This clinical practice guideline is for use by the Philippine Society of Otolaryngology-Head and Neck Surgery. It covers the diagnosis and
Advances in the Surgical Management of Chronic Rhinosinusitis
 Rhinitis and Sinusitis dvances in the Surgical Management of Chronic Rhinosinusitis Erin D. Wright, MDCM, MEd, FRCSC; Saul Frenkiel, MDCM, FRCSC bstract The surgical management of chronic rhinosinusitis
Rhinitis and Sinusitis dvances in the Surgical Management of Chronic Rhinosinusitis Erin D. Wright, MDCM, MEd, FRCSC; Saul Frenkiel, MDCM, FRCSC bstract The surgical management of chronic rhinosinusitis
Site of Service - Select Surgical Procedures. Select a hyperlink below to be directed to that section.
 MEDICAL POLICY 7.01.559 Sinus Surgery BCBSA Ref. Policies: 7.01.105, 7.01.155 Effective Date: June 1, 2018* RELATED MEDICAL POLICIES: Last Revised: May 1, 2018 7.01.558 Rhinoplasty Replaces: N/A 11.01.524
MEDICAL POLICY 7.01.559 Sinus Surgery BCBSA Ref. Policies: 7.01.105, 7.01.155 Effective Date: June 1, 2018* RELATED MEDICAL POLICIES: Last Revised: May 1, 2018 7.01.558 Rhinoplasty Replaces: N/A 11.01.524
BALLOON OSTIAL DILATION FOR TREATMENT OF CHRONIC SINUSITIS
 BALLOON OSTIAL DILATION FOR TREATMENT OF CHRONIC SINUSITIS Non-Discrimination Statement and Multi-Language Interpreter Services information are located at the end of this document. Coverage for services,
BALLOON OSTIAL DILATION FOR TREATMENT OF CHRONIC SINUSITIS Non-Discrimination Statement and Multi-Language Interpreter Services information are located at the end of this document. Coverage for services,
The Honrubia Technique of Balloon Sinuplasty for the Improvement of Symptoms in Chronic Sinusitis. Patients. Vincent Honrubia MD, FACS
 The Honrubia Technique of Balloon Sinuplasty for the Improvement of Symptoms in Chronic Sinusitis Patients Vincent Honrubia MD, FACS Allyssa Cantu, PA-S Sharon Gelman, MEd, PA-S Rachel Tsai, BS Director,
The Honrubia Technique of Balloon Sinuplasty for the Improvement of Symptoms in Chronic Sinusitis Patients Vincent Honrubia MD, FACS Allyssa Cantu, PA-S Sharon Gelman, MEd, PA-S Rachel Tsai, BS Director,
INFORMATION REGARDING YOUR NASAL SURGERY
 INFORMATION REGARDING YOUR NASAL SURGERY This document contains information about the following aspects of nasal surgery: Pre-op information: How to prepare for surgery. Procedure: Wat is done during surgery.
INFORMATION REGARDING YOUR NASAL SURGERY This document contains information about the following aspects of nasal surgery: Pre-op information: How to prepare for surgery. Procedure: Wat is done during surgery.
BALLOON SINUS OSTIAL DILATION
 UnitedHealthcare of California (HMO) UnitedHealthcare Benefits Plan of California (EPO/POS) UnitedHealthcare of Oklahoma, Inc. UnitedHealthcare of Oregon, Inc. UnitedHealthcare Benefits of Texas, Inc.
UnitedHealthcare of California (HMO) UnitedHealthcare Benefits Plan of California (EPO/POS) UnitedHealthcare of Oklahoma, Inc. UnitedHealthcare of Oregon, Inc. UnitedHealthcare Benefits of Texas, Inc.
Pathophysiology and Etiology
 Sinusitis Pathophysiology and Etiology Sinusitis is inflammation of the mucosa of one or more sinuses. It can be either acute chronic. Chronic sinusitis is diagnosed if symptoms are present for more than
Sinusitis Pathophysiology and Etiology Sinusitis is inflammation of the mucosa of one or more sinuses. It can be either acute chronic. Chronic sinusitis is diagnosed if symptoms are present for more than
Incidence of accessory ostia in patients with chronic maxillary sinusitis
 International Journal of Otorhinolaryngology and Head and Neck Surgery Ghosh P et al. Int J Otorhinolaryngol Head Neck Surg. 2018 Mar;4(2):443-447 http://www.ijorl.com pissn 2454-5929 eissn 2454-5937 Original
International Journal of Otorhinolaryngology and Head and Neck Surgery Ghosh P et al. Int J Otorhinolaryngol Head Neck Surg. 2018 Mar;4(2):443-447 http://www.ijorl.com pissn 2454-5929 eissn 2454-5937 Original
SINUSITIS. HAVAS ENT CLINICS Excellence in otolaryngology
 JULY 2015 SINUSITIS WHAT IS IT? WHAT SHOULD YOU DO? WHAT WORKS? THOMAS E HAVAS MBBS (SYD) MD (UNSW) FRCSE, FRACS, FACS CONJOINT ASSOCIATE PROFESSOR UNSW OTOLARNGOLOGY HEAD AND NECK SURGERY HAVAS ENT CLINICS
JULY 2015 SINUSITIS WHAT IS IT? WHAT SHOULD YOU DO? WHAT WORKS? THOMAS E HAVAS MBBS (SYD) MD (UNSW) FRCSE, FRACS, FACS CONJOINT ASSOCIATE PROFESSOR UNSW OTOLARNGOLOGY HEAD AND NECK SURGERY HAVAS ENT CLINICS
A comprehensive study on complications of endoscopic sinus surgery
 International Journal of Otorhinolaryngology and Head and Neck Surgery Shyras JAD et al. Int J Otorhinolaryngol Head Neck Surg. 2017 Jul;3(3):472-477 http://www.ijorl.com pissn 2454-5929 eissn 2454-5937
International Journal of Otorhinolaryngology and Head and Neck Surgery Shyras JAD et al. Int J Otorhinolaryngol Head Neck Surg. 2017 Jul;3(3):472-477 http://www.ijorl.com pissn 2454-5929 eissn 2454-5937
EVALUATION OF PATIENT'S OUTCOME AFTER ENDOSCOPIC SINUS SURGERY
 Basrah Journal Of Surgery Bas J Surg, September, 16, 2010 EVALUATION OF PATIENT'S OUTCOME AFTER ENDOSCOPIC SINUS SURGERY Hiwa A Abdulkareem MB.ChB, FICMS, CABS(ENT); Teaching Hospital; University of Sulaimania.
Basrah Journal Of Surgery Bas J Surg, September, 16, 2010 EVALUATION OF PATIENT'S OUTCOME AFTER ENDOSCOPIC SINUS SURGERY Hiwa A Abdulkareem MB.ChB, FICMS, CABS(ENT); Teaching Hospital; University of Sulaimania.
Original Article Comparison of different surgical approaches of functional endoscopic sinus surgery on patients with chronic rhinosinusitis
 Int J Clin Exp Med 2014;7(6):1585-1591 www.ijcem.com /ISSN:1940-5901/IJCEM0000315 Original Article Comparison of different surgical approaches of functional endoscopic sinus surgery on patients with chronic
Int J Clin Exp Med 2014;7(6):1585-1591 www.ijcem.com /ISSN:1940-5901/IJCEM0000315 Original Article Comparison of different surgical approaches of functional endoscopic sinus surgery on patients with chronic
Canine Fossa Puncture for Severe Maxillary Disease in Unilateral Chronic Sinusitis With Nasal Polyp
 The Laryngoscope VC 2013 The American Laryngological, Rhinological and Otological Society, Inc. Canine Fossa Puncture for Severe Maxillary Disease in Unilateral Chronic Sinusitis With Nasal Polyp Jang
The Laryngoscope VC 2013 The American Laryngological, Rhinological and Otological Society, Inc. Canine Fossa Puncture for Severe Maxillary Disease in Unilateral Chronic Sinusitis With Nasal Polyp Jang
Endoscopic Management Of A Giant Ethmoid Mucocele
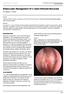 ISPUB.COM The Internet Journal of Otorhinolaryngology Volume 6 Number 1 S Ceylan, F Bora Citation S Ceylan, F Bora.. The Internet Journal of Otorhinolaryngology. 2006 Volume 6 Number 1. Abstract We present
ISPUB.COM The Internet Journal of Otorhinolaryngology Volume 6 Number 1 S Ceylan, F Bora Citation S Ceylan, F Bora.. The Internet Journal of Otorhinolaryngology. 2006 Volume 6 Number 1. Abstract We present
ORIGINAL ARTICLE. Computed Tomographic Staging and the Fate of the Dependent Sinuses in Revision Endoscopic Sinus Surgery
 Computed Tomographic Staging and the Fate of the Dependent es in Revision Endoscopic Surgery Neil Bhattacharyya, MD ORIGINAL ARTICLE Objectives: To determine the patterns of disease recurrence in chronic
Computed Tomographic Staging and the Fate of the Dependent es in Revision Endoscopic Surgery Neil Bhattacharyya, MD ORIGINAL ARTICLE Objectives: To determine the patterns of disease recurrence in chronic
Imaging of the Paranasal Sinuses
 14. Sommerschule Imaging of the Paranasal Sinuses Bettlach 24.08.2018 Christoph Schlegel Conventional Radiology NNH-Status: okzipito-frontal: frontal sinus, anterior ethmoid okzipito-nasal : maxillary
14. Sommerschule Imaging of the Paranasal Sinuses Bettlach 24.08.2018 Christoph Schlegel Conventional Radiology NNH-Status: okzipito-frontal: frontal sinus, anterior ethmoid okzipito-nasal : maxillary
Chronic rhinosinusitis (CRS) is a significant health
 ORIGINAL ARTICLE The safety and performance of a maxillary sinus ostium self-dilation device: a pilot study Iain Hathorn, MBChB, FRCSEd (ORL-HNS), Al-Rahim Habib, MSc, Rachelle Dar Santos, BSc, CCRP and
ORIGINAL ARTICLE The safety and performance of a maxillary sinus ostium self-dilation device: a pilot study Iain Hathorn, MBChB, FRCSEd (ORL-HNS), Al-Rahim Habib, MSc, Rachelle Dar Santos, BSc, CCRP and
Sinusitis & its complication. MOHAMMED ALESSA MBBS,FRCSC Assistant Professor,Consultant Otolaryngology, Head & Neck Surgery King Saud University
 Sinusitis & its complication MOHAMMED ALESSA MBBS,FRCSC Assistant Professor,Consultant Otolaryngology, Head & Neck Surgery King Saud University Definition Types Clinical manifestation Complications Diagnosis
Sinusitis & its complication MOHAMMED ALESSA MBBS,FRCSC Assistant Professor,Consultant Otolaryngology, Head & Neck Surgery King Saud University Definition Types Clinical manifestation Complications Diagnosis
Clinical Outcome of Endoscopic Surgery for Frontal Sinusitis
 Clinical Outcome of Endoscopic Surgery for Frontal Sinusitis Ralph Metson, MD; Richard E. Gliklich, MD ORIGINAL ARTICLE Objective: To determine the efficacy of endoscopic surgery for chronic frontal sinusitis.
Clinical Outcome of Endoscopic Surgery for Frontal Sinusitis Ralph Metson, MD; Richard E. Gliklich, MD ORIGINAL ARTICLE Objective: To determine the efficacy of endoscopic surgery for chronic frontal sinusitis.
ISSN X (Print) Research Article. *Corresponding author Dr.V. Krishna Chaitanya
 Scholars Journal of Applied Medical Sciences (SJAMS) Sch. J. App. Med. Sci., 2015; 3(1G):508-513 Scholars Academic and Scientific Publisher (An International Publisher for Academic and Scientific Resources)
Scholars Journal of Applied Medical Sciences (SJAMS) Sch. J. App. Med. Sci., 2015; 3(1G):508-513 Scholars Academic and Scientific Publisher (An International Publisher for Academic and Scientific Resources)
Sinusitis. Disclosures. What are sinuses? STEVEN E DAVIS, MD. Consultant and speaker, Novartis Research: Intersect ENT
 Sinusitis STEVEN E DAVIS, MD Disclosures Consultant and speaker, Novartis Research: Intersect ENT What are sinuses? Stedman s medical dictionary 25 th edition 1. a channel for the passage of blood or lymph,
Sinusitis STEVEN E DAVIS, MD Disclosures Consultant and speaker, Novartis Research: Intersect ENT What are sinuses? Stedman s medical dictionary 25 th edition 1. a channel for the passage of blood or lymph,
Sinusitis what it is and what is SNOT: Updates on therapies and other assorted snacks. Shane Gailushas, MD Mercy Ear, Nose, and Throat Clinic
 Sinusitis what it is and what is SNOT: Updates on therapies and other assorted snacks Shane Gailushas, MD Mercy Ear, Nose, and Throat Clinic ANATOMY & PHYSIOLOGY Factors that predispose patients to Rhinosinusitis:
Sinusitis what it is and what is SNOT: Updates on therapies and other assorted snacks Shane Gailushas, MD Mercy Ear, Nose, and Throat Clinic ANATOMY & PHYSIOLOGY Factors that predispose patients to Rhinosinusitis:
US Drug Eluting Sinus Stent (DESS) Market: Size, Trends & Forecasts ( ) September 2016
 US Drug Eluting Sinus Stent (DESS) Market: Size, Trends & Forecasts (2016-2021) September 2016 US Drug Eluting Sinus Stent Market Report Scope of the Report The report entitled US Drug Eluting Sinus Stent
US Drug Eluting Sinus Stent (DESS) Market: Size, Trends & Forecasts (2016-2021) September 2016 US Drug Eluting Sinus Stent Market Report Scope of the Report The report entitled US Drug Eluting Sinus Stent
Functional endoscopic sinus surgery (FESS) is an effective
 ORIGINAL ARTICLE Evaluation of patient nasal saline irrigation practices following endoscopic sinus surgery Frederick Yoo, MD, Elisabeth H. Ference, MD, MPH, Edward C. Kuan, MD, MBA, Jivianne T. Lee, MD,
ORIGINAL ARTICLE Evaluation of patient nasal saline irrigation practices following endoscopic sinus surgery Frederick Yoo, MD, Elisabeth H. Ference, MD, MPH, Edward C. Kuan, MD, MBA, Jivianne T. Lee, MD,
