MEDICAL POLICY EFFECTIVE DATE: 04/21/11 REVISED DATE: 06/21/12, 05/23/13, 06/19/14, 06/18/15, 06/16/16, 06/15/17
|
|
|
- Letitia Shauna Lucas
- 6 years ago
- Views:
Transcription
1 MEDICAL POLICY SUBJECT: SINUS OSTIAL DILATION FOR PAGE: 1 OF: 6 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial product, including an Essential Plan product, covers a specific service, medical policy criteria apply to the benefit. If a Medicare product covers a specific service, and there is no national or local Medicare coverage decision for the service, medical policy criteria apply to the benefit. POLICY STATEMENT: Based upon our criteria and assessment of peer-reviewed literature, balloon sinuplasty or any other catheter-based inflatable device, as a stand-alone procedure has been medically proven to be effective and is considered medically appropriate for the treatment of chronic sinusitis when ALL the following criteria have been met: I. Documentation of persistent sinusitis for greater than three months; AND II. Failure of medical therapy greater than three months (e.g., antibiotics, steroid nasal spray, antihistamines); AND III. Radiologic evidence of chronic sinusitis by either air fluid levels, mucosal thickening greater than two mm, or opacification. POLICY GUIDELINES: If balloon sinuplasty is performed in conjunction with another sinus surgery in the same sinus, the balloon dilation would be considered inclusive/incidental to the primary procedure, and therefore, would not be allowed separate reimbursement. DESCRIPTION: Chronic rhinosinusitis (CRS) is characterized by purulent nasal discharge, usually without fever, that persists for weeks to months. Symptoms of congestion often accompany the nasal discharge. There also may be mild pain and/or headache. In some cases of chronic sinusitis, surgical drainage may be necessary. Functional endoscopic sinus surgery (FESS, used when patients fail to respond to aggressive medical management, has become an important aspect for surgical management of chronic sinusitis. For this procedure, a fiberoptic nasal endoscope is used to visualize the sinus ostia, and any obstruction found is corrected. This procedure restores patency and allows air and mucous transport through the natural ostium. A new procedure, balloon sinuplasty, has been proposed as an alternative to endoscopic sinus surgery for those with chronic sinusitis. The procedure involves placing a guidewire in the sinus ostium (confirmed with fluoroscopy, or with direct transillumination of the targeted sinus cavity), advancing a balloon over the guidewire, and then stretching the opening by inflating the balloon. Balloon sinuplasty aims to restore sinus drainage and function without damaging the sinus mucosa. Pressure caused by the inflated balloon restructures and widens the ostium by creating microfractures in the surrounding bone. General anesthesia is usually needed for this procedure to minimize patient movement. However, an increasing number of ENT doctors perform the procedure in the office, under local anesthesia. Newer devices have been developed that do not utilize balloon dilation, but allow for a more gradual dilation through an osmotic process using the body s natural mucosal fluids to expand the insert before removal. This technique is referred to as self-expanding absorptive sinus ostial dilation. This procedure involves the intranasal insertion of a pluglike device into the sinus ostia. Once inserted, the device absorbs moisture from the surrounding tissue and begins to expand, providing low-pressure, gradual dilation of the sinus ostia. Once the device has been given enough time to fully expand, it is removed. This type of device is proposed to maximize patient tolerability of the procedure. A nonprofit independent licensee of the BlueCross BlueShield Association
2 PAGE: 2 OF: 6 RATIONALE: In March 2008, the device Relieva Sinus Balloon Catheter (Acclarent, Menlo Park, CA) was cleared for marketing by the FDA through the 510(k) process. The FDA determined that this device was substantially equivalent to existing devices for use in dilating the sinus ostia and paranasal spaces in adults and maxillary sinus spaces in children. Subsequent devices developed by Acclarent have also been granted 510(k) marketing clearance. These include the Relieva Spin Sinus Dilation System cleared in August 2011, and the Relieva Seeker Balloon Sinuplasty System cleared in November In June 2008, the device FinESS Sinus Treatment (Entellus Medical, Inc., Maple Grove, MN) was cleared for marketing by the FDA through the 510(k) process. The indication noted is to access and treat the maxillary ostia/ethmoid infundibula in adults using a transantral approach. The bony sinus outflow tracts are remodeled by balloon displacement of adjacent bone and paranasal sinus structures. Two other balloon sinus ostial dilation devices by Entellus Medical Inc. also received 510(k) approval in August, These are the ENTrigue Sinus Dilation System, and the XprESS Multi-Sinus Dilation Tool. The NuVent EM Balloon Sinus Dilation System (Medtronic) received FDA 510(k) clearance in late It features a built-in electromagnetic surgical navigation technology. The Vent-Os-Gentle Sinus Dilation System (SinuSys Corporation) received FDA clearance in January Unlike balloon dilation devices that use rapid, high-pressure inflation, the Vent-Os sinus dilation system is a small, lowpressure, self-expanding insert designed to gently and gradually open the maxillary ostia. The Vent-Os System incorporates the Company s proprietary osmotic technology, which utilizes the body s natural mucosal fluids to expand the insert before removal. The FDA approval for the Vent-Os System was based on comparison to predicate devices. The Vent-Os System achieved post-procedural patency in 95 percent of the sinus ostia treated in a multi-center study and submitted as part of the Company s FDA application; five percent of the treated ostia could not be visualized. In the study, the Vent-Os device was inserted into the maxillary sinus opening at the beginning of the procedure and removed after 60 minutes. No adverse events occurred during insertion or removal of the device. Three-month follow-up was completed in 33 patients (55 ostia), with 93 percent of the treated ostia remaining patent and seven percent that could not be visualized. No ostia were reported to be occluded. Fifteen percent of patients in the study were treated in an office setting after pre-procedural injection of anesthesia; no additional anesthesia or medication was required for these patients for the duration of the procedure. This is in contrast to balloon dilation devices, which often require administration of anxiolytics, analgesics and/or additional local injections of anesthetics during the procedure to increase patient tolerability. The remaining patients were treated in the operating room adjunctive to functional endoscopic sinus surgery (FESS). While longer-term clinical outcomes are still required, there is short-term evidence that this technique can be performed successfully and safely in adult patients with chronic rhinosinusitis. Small, randomized controlled trials, including the largest study (REMODEL) of 105 patients, report short-term improvement in symptoms that are similar to FESS, and potential advantages for balloon ostial dilation on postoperative recovery time and pain medication use. CODES: Number Description Eligibility for reimbursement is based upon the benefits set forth in the member s subscriber contract. CODES MAY NOT BE COVERED UNDER ALL CIRCUMSTANCES. PLEASE READ THE POLICY AND GUIDELINES STATEMENTS CAREFULLY. Codes may not be all inclusive as the AMA and CMS code updates may occur more frequently than policy updates. CPT: Nasal/sinus endoscopy, surgical; with dilation of maxillary sinus ostium (eg, balloon dilation), transnasal or via canine fossa Nasal/sinus endoscopy, surgical; with dilation of frontal sinus ostium (eg, balloon dilation)
3 PAGE: 3 OF: Nasal/sinus endoscopy, surgical; with dilation of sphenoid sinus ostium (eg, balloon dilation) Copyright 2017 American Medical Association, Chicago, IL HCPCS: C1726 Catheter, balloon dilatation, non-vascular ICD9: Chronic sinusitis (code range) ICD10: J32.0-J32.9 Chronic sinusitis (code range) REFERENCES: Achar P, et al. Endoscopic dilatation sinus surgery (FEDS) versus functional endoscopic sinus surgery (FESS) for treatment of chronic rhinosinusitis: a pilot study. Acta Otorhinolaryngol Ital 2012 Oct;32(5): Albritton FD 4 th, et al. Feasibility of in office endoscopic sinus surgery with balloon sinus dilation. Am J Rhinol Allergy 2012 May-Jun;26(3): *American Academy of Otolaryngology-Head and Neck Surgery. Statement on balloon dilation Jun [ accessed 5/17/17. American Rhinologic Society. Revised position statement on endoscopic balloon catheter sinus dilation technology. [ accessed 5/17/17. Batra PS. Evidenced-based practice: balloon catheter dilation in rhinology. Otolaryngol Clin North Am 2012 Oct;45(5): Bikhazi N, et al. Standalone balloon dilation versus sinus surgery for chronic rhinosinusitis: A prospective, multicenter, randomized, controlled trial with 1-year follow-up. Am J Rhinol Allergy 2014 Jul-Aug;28(4): Bizaki AJ, et al. Quality of life after endoscopic sinus surgery or balloon sinuplasty: a randomized clinical study. Rhinology 2014 Dec;52(4): Bizaki AJ, et al. Decrease of nasal airway resistance and alleviations of symptoms after balloon sinuplasty in patients with isolated chronic rhinosinusitis: a prospective, randomized clinical study. Clin Otolaryngol 2016 Dec;41(6): BlueCross BlueShield Association. Balloon Sinuplasty for Treatment of Chronic Sinusitis. Medical Policy Reference Manual Medical Policy # May 8. BlueCross BlueShield Association. Technology Evaluation Center (TEC). Balloon sinus ostial dilation for treatment of chronic rhinosinusitis Apr;27(9). *Bolger WE, et al. Safety and outcomes of balloon catheter sinuostomy: a multicenter 24-week analysis in 115 patients. Otolaryngol Head Neck Surg 2007 Jul;137(1): Bowles PF, et al. Efficacy of balloon sinuplasty in treatment of frontal rhinosinusitis: a prospective study in sixty patients. Clin Otolaryngol 2016 Dec 22.[Epub ahead of print]. Brodner D, et al. Safety and outcomes following hybrid balloon and balloon-only procedures using multifunction, multisinus balloon dilation tool. Int Forum Allergy Rhinol 2013 Aug;3(8): Catalano PJ. Balloon dilation technology: let the truth be told. Curr Allergy Asthma Rep 2013 Apr;13(2): Chandra RK, et al. REMODEL larger cohort with long-term outcomes and meta-analysis of standalone balloon dilation studies. Laryngoscope 2016 Jan;126(1): *Cutler J, et al. First clinic experience: patient selection and outcomes for ostial dilation for chronic rhinosinusitis. Int Forum Allergy Rhinol 2011 Nov-Dec;1(6):460-5.
4 PAGE: 4 OF: 6 Cutler J, et al. Standalone balloon dilation versus sinus surgery for chronic rhinosinusitis: a prospective, multicenter, randomized, controlled trial. Am J Rhinol Allergy 2013 Sep-Oct;27(5): Dass K, et al. Diagnosis and management of rhinosinusitis: Highlights from the 2015 practice parameter. Curr Allergy Asthma Rep 2016 Apr;16(4):29. *Friedman M, et al. Functional endoscopic dilatation of the sinuses: patient satisfaction, postoperative pain, and cost. Am J Rhinology 2008 Mar-Apr;22(2): Gould J, et al. In-office, multisinus balloon dilation: 1-year outcomes from a prospective, multicenter, open label trial. Am J Rhinol Allergy 2014 Mar-Apr;28(2): Hathorn I, et al. The safety and performance of a maxillary sinus ostium self-dilation device: a pilot study. Int Forum Allergy Rhinol 2014 Aug;4(8): Hathorn IF, et al. Randomized controlled trial: hybrid technique using balloon dilation of the frontal sinus drainage pathway. Int Forum Allergy Rhinol 2015 Feb;5(2): Karanfilov B, et al. Office-based balloon sinus dilation: a prospective, multicenter study of 203 patients. Int Forum Allergy Rhinol 2013 May;3(5): *Kim E, et al. Balloon dilatation of the paranasal sinuses: a tool in sinus surgery. Otolaryngol Clin North Am 2009 Oct;42(5): *Kizhner V, et al. Transantral balloon dilation plus posterior ethmoidectomy. Laryngoscope 2011 May;121(5): Koskinen A, et al. Endoscopic sinus surgery might reduce exacerbations and symptoms more than balloon sinuplasty. Am J Rhinol Allergy 2012 Nov-Dec;26(6):e Koskinen A, et al. Long-term follow-up after ESS and balloon sinuplasty: comparison of symptom reduction and patient satisfaction. Acta Otolaryngol 2016;136(5): Koskinen A, et al. Comparison of intra-operative characteristics and early post-operative outcomes between endoscopic sinus surgery and balloon sinuplasty. Acta Otolaryngol 2017 Feb;137(2): *Kuhn FA, et al. Balloon catheter sinusotomy: one year follow-up- outcomes and role in functional endoscopic sinus surgery. Otolaryngol Head Neck Surg 2008 Sep;139(3 Suppl 3):S *Kutluhan A, et al. Endoscopic balloon dilation sinuplasty including ethmoidal air cells in chronic rhinosinusitis. Ann Oto Rhinol Laryngol 2009 Dec;118(12): Levine SB, et al. In-office stand-alone balloon dilation of maxillary sinus ostia and ethmoid infundibula in adults with chronic or recurrent acute rhinosinusitis: a prospective, multi-institutional study with 1-year follow-up. Ann Oto Rhinol Laryngol 2013 Nov;122(11): Levy JM, et al. Paranasal sinus balloon catheter dilation for treatment of chronic rhinosinusitis: a systematic review and meta-analysis. Otolaryngol Head Neck Surg 2016 Jan;154(1): Mazetti A, et al. The role of balloon sinuplasty in the treatment of sinus headache. Otolaryngol Pol 2014 Jan- Feb;68(1):15-9. *National Institute for Health and Clinical Excellence (NICE). Balloon catheter dilation of paranasal sinus ostia for chronic sinusitis Sep [ accessed 5/17/17. Orlandi RR, et al. International consensus statement on allergy and rhinology: rhinosinusitis executive summary. Int Forum Allergy Rhinol 2016 Feb;6 Suppl 1:S3-S21.
5 PAGE: 5 OF: 6 Payne SC, et al. Medical therapy versus sinus surgery by using balloon sinus dilation technology: A prospective multicenter study. Am J Rhinol Allergy. Jul 2016;30(4): *Plaza G, et al. Balloon dilation of the frontal recess: a randomized clinical trial. Ann Otol Rhinol Laryngol 2011 Aug;120(8): *Ramadan HH. Safety and feasibility of balloon sinuplasty for treatment of chronic rhinosinusitis in children. Ann Otol Rhinol Laryngol 2009 Mar;118(3): *Ramadan HH, et al. Balloon catheter sinuplasty in young children. Am J Rhinol Allergy 2010 Jan-Feb;24(1):e54-6. *Ramadan HH, et al. Balloon catheter sinuplasty and adenoidectomy in children with chronic rhinosinusitis. Ann Oto Rhinol Laryngol 2010 Sep;119(9): Ramadan HH, et al. Sinus balloon catheter dilation after adenoidectomy failure for children with chronic rhinosinusitis. Arch Otolaryngol Head Neck Surg 2012 Jul;138(7): Rosenfeld RM, et al. Clinical practice guideline (update): Adult sinusitis. Otolaryngol Head Neck Surg 2015 Apr;152(2S):S1-39. Sikand A, et al. Office-based balloon sinus dilation: 1-year follow-up of a prospective multicenter study. Ann Otol Rhinol Laryngol 2015 Aug;124(8): Sillers MJ, et al. In-office functional endoscopic sinus surgery for chronic rhinosinusitis utilizing balloon catheter dilation technology. Curr Opin Otolaryngol Head Neck Surg 2013 Feb;21(1): Soler ZM, et al. Prospective, multicenter evaluation of balloon sinus dilation for treatment of pediatric chronic rhinosinusitis. Int Forum Allergy Rhinol 2017 March;7(3): *Stankiewicz J, et al. Transantral, endoscopically guided balloon dilatation of the local ostiomeatal complex for chronic rhinosinusitis under local anesthesia. Am J Rhinol 2009 May-Jun;23(3): Stankiewicz J, et al. Two-year results: transantral balloon dilation of the ethmoid infundibulum. Int Forum Allergy Rhinol 2012 May-Jun;2(3): *Stewart AE, et al. Balloon sinuplasty vs surgical management of chronic rhinosinusitis. Curr Allergy Asthma Rep 2010 May;10(3): Szczygielski K, et al. A six-month analysis of frontal sinus drainage pathway in patients with frontal sinus after balloon sinuplasty. Acta Otolaryngol 2017 March 16:1-7. *Taghi AS, et al. Balloon sinuplasty: balloon catheter dilation of paranasal sinus ostia for chronic rhinosinusitis. Expert Rev Med Devices 2009 Jul;6(4): Thottam PJ, et al. Functional endoscopic sinus surgery (FESS) alone versus balloon catheter sinuplasty (BCS) and ethmoidectomy: a comparative outcome analysis in pediatric chronic rhinosinusitis. Int J Pediatr Otorhinolaryngol 2012 Sep;76(9): Thottam PJ, et al. Functional endoscopic sinus surgery versus balloon sinuplasty with ethmoidectomy: a 2-year analysis in pediatric chronic rhinosinusitis. Indian J Otolaryngol Head Neck Surg 2016 Sept;68(3): Tomazic PV, et al. Feasibility of balloon sinuplasty in patients with chronic rhinosinusitis: the Graz experience. Rhinology 2013 Jun;51(2): *Vaughan WC. Review of balloon sinuplasty. Curr Opin Otolaryngol Head Neck Surg 2008 Feb;16(1):2-9. Wang F, et al. Sinus balloon catheter dilation in pediatric chronic rhinosinusitis resistant to medical therapy. JAMA Otolaryngol Head Neck Surg 2015 Jun;141(6):
6 PAGE: 6 OF: 6 *Weiss RL, et al. Long-term outcome analysis of balloon catheter sinusotomy: two-year follow-up. Otolaryngol Head Neck Surg 2008 Sep;139(3 Suppl 3):S *Wittkopf ML, et al. Balloon sinuplasty for the surgical management of immuncompromised and critically ill patients with acute rhinosinusitis. Otolaryngol Head Neck Surg 2009 Apr;140(4): *key article KEY WORDS: Balloon sinuplasty, balloon dilation, catheter sinusotomy CMS COVERAGE FOR MEDICARE PRODUCT MEMBERS Based upon our review, sinus ostial dilation/balloon sinuplasty is not addressed in National or Regional CMS coverage determinations or policies.
Protocol. Balloon Ostial Dilation for Treatment of Chronic Rhinosinusitis
 Protocol Balloon Ostial Dilation for Treatment of Chronic Rhinosinusitis Medical Benefit Effective Date: 04/01/15 Next Review Date: 11/19 Preauthorization No Review Dates: 07/07, 07/08, 11/08, 05/09, 01/10,
Protocol Balloon Ostial Dilation for Treatment of Chronic Rhinosinusitis Medical Benefit Effective Date: 04/01/15 Next Review Date: 11/19 Preauthorization No Review Dates: 07/07, 07/08, 11/08, 05/09, 01/10,
Topic: Balloon Catheter Dilation for Treatment of Sinusitis Date of Origin: August 8, Section: Surgery Last Reviewed Date: February 2014
 Medical Policy Manual Topic: Balloon Catheter Dilation for Treatment of Sinusitis Date of Origin: August 8, 2006 Section: Surgery Last Reviewed Date: February 2014 Policy No: 153 Effective Date: April
Medical Policy Manual Topic: Balloon Catheter Dilation for Treatment of Sinusitis Date of Origin: August 8, 2006 Section: Surgery Last Reviewed Date: February 2014 Policy No: 153 Effective Date: April
Description. Section: Surgery Effective Date: April 15, 2017 Subsection: Surgery Original Policy Date: June 7, 2012 Subject: Page: 1 of 13
 Last Review Status/Date: March 2017 Page: 1 of 13 Description Balloon ostial dilation (also known as balloon sinuplasty ) is proposed as an alternative to traditional endoscopic sinus surgery for patients
Last Review Status/Date: March 2017 Page: 1 of 13 Description Balloon ostial dilation (also known as balloon sinuplasty ) is proposed as an alternative to traditional endoscopic sinus surgery for patients
Description. Section: Surgery Effective Date: April 15, 2014 Subsection: Surgery Original Policy Date: June 7, 2012 Subject: Page: 1 of 12
 Last Review Status/Date: March 2014 Page: 1 of 12 Description Balloon ostial dilation (also known as balloon sinuplasty ) is proposed as an alternative to traditional endoscopic sinus surgery for patients
Last Review Status/Date: March 2014 Page: 1 of 12 Description Balloon ostial dilation (also known as balloon sinuplasty ) is proposed as an alternative to traditional endoscopic sinus surgery for patients
Patency of Maxillary Sinus Ostia Following Dilation with a Novel Osmotic Expansion Device
 WHITE PAPER Patency of Maxillary Sinus Ostia Following Dilation with a Novel Osmotic Expansion Device Six-Month Results from a Multi-Center Prospective Study Jerome Hester, MD California Sleep Institute,
WHITE PAPER Patency of Maxillary Sinus Ostia Following Dilation with a Novel Osmotic Expansion Device Six-Month Results from a Multi-Center Prospective Study Jerome Hester, MD California Sleep Institute,
Medicare Advantage Medical Policy. Title: Balloon Ostial Dilation for Treatment of Chronic Rhinosinusitis
 Medicare Advantage Medical Policy Current Policy Effective Date: 1/1/18 Title: Balloon Ostial Dilation for Treatment of Chronic Rhinosinusitis Description/Background CHRONIC RHINOSINUSITIS Chronic rhinosinusitis
Medicare Advantage Medical Policy Current Policy Effective Date: 1/1/18 Title: Balloon Ostial Dilation for Treatment of Chronic Rhinosinusitis Description/Background CHRONIC RHINOSINUSITIS Chronic rhinosinusitis
Balloon Ostial Dilation for Treatment of Chronic Rhinosinusitis
 Balloon Ostial Dilation for Treatment of Chronic Rhinosinusitis Policy Number: 7.01.105 Last Review: 3/2018 Origination: 3/2007 Next Review: 9/2018 Policy Blue Cross and Blue Shield of Kansas City (Blue
Balloon Ostial Dilation for Treatment of Chronic Rhinosinusitis Policy Number: 7.01.105 Last Review: 3/2018 Origination: 3/2007 Next Review: 9/2018 Policy Blue Cross and Blue Shield of Kansas City (Blue
Medical Policy An independent licensee of the Blue Cross Blue Shield Association
 Balloon Sinuplasty for Treatment of Chronic Sinusitis Page 1 of 17 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Balloon Sinuplasty for Treatment of Chronic Sinusitis
Balloon Sinuplasty for Treatment of Chronic Sinusitis Page 1 of 17 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Balloon Sinuplasty for Treatment of Chronic Sinusitis
Medical Policy An independent licensee of the Blue Cross Blue Shield Association
 Balloon Sinuplasty for Treatment of Chronic Sinusitis Page 1 of 18 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Balloon Sinuplasty for Treatment of Chronic Sinusitis
Balloon Sinuplasty for Treatment of Chronic Sinusitis Page 1 of 18 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Balloon Sinuplasty for Treatment of Chronic Sinusitis
Clinical Policy: Balloon Sinus Ostial Dilation for Treatment of Chronic Sinusitis
 Clinical Policy: Balloon Sinus Ostial Dilation for Treatment of Chronic Sinusitis Reference Number: CP.MP.119 Last Review Date: 11/17 See Important Reminder at the end of this policy for important regulatory
Clinical Policy: Balloon Sinus Ostial Dilation for Treatment of Chronic Sinusitis Reference Number: CP.MP.119 Last Review Date: 11/17 See Important Reminder at the end of this policy for important regulatory
Treatment Options for Chronic Sinusitis
 Treatment Options for Chronic Sinusitis Jesse Ryan, M.D. Assistant Professor Head and Neck Surgery & Reconstruction Department of Otolaryngology January 17, 2019 Disclosures I have no financial relationship
Treatment Options for Chronic Sinusitis Jesse Ryan, M.D. Assistant Professor Head and Neck Surgery & Reconstruction Department of Otolaryngology January 17, 2019 Disclosures I have no financial relationship
FRONTAL SINUPLASTY P R E P A R E D A N D P R E S E N T E D B Y : D R. Y A H Y A F A G E E H R 4 16/ 12/ 2013
 FRONTAL SINUPLASTY P R E P A R E D A N D P R E S E N T E D B Y : D R. Y A H Y A F A G E E H R 4 16/ 12/ 2013 ANATOMY: FRONTAL SINUS Not present at birth Starts developing at 4 years Radiographically visualized
FRONTAL SINUPLASTY P R E P A R E D A N D P R E S E N T E D B Y : D R. Y A H Y A F A G E E H R 4 16/ 12/ 2013 ANATOMY: FRONTAL SINUS Not present at birth Starts developing at 4 years Radiographically visualized
BALLOON OSTIAL DILATION FOR TREATMENT OF CHRONIC SINUSITIS
 BALLOON OSTIAL DILATION FOR TREATMENT OF CHRONIC SINUSITIS Non-Discrimination Statement and Multi-Language Interpreter Services information are located at the end of this document. Coverage for services,
BALLOON OSTIAL DILATION FOR TREATMENT OF CHRONIC SINUSITIS Non-Discrimination Statement and Multi-Language Interpreter Services information are located at the end of this document. Coverage for services,
Learner Objectives. Overview. Minimally Invasive Sinus Surgery: What is the data for Balloon Catheter Dilation 7/31/2017
 Minimally Invasive Sinus Surgery: What is the data for Balloon Catheter Dilation David M. Poetker, MD Christopher M. Long, MD Rhinology and Sinus Surgery Medical College of Wisconsin Thank you to Susan
Minimally Invasive Sinus Surgery: What is the data for Balloon Catheter Dilation David M. Poetker, MD Christopher M. Long, MD Rhinology and Sinus Surgery Medical College of Wisconsin Thank you to Susan
Balloon Sinuplasty: Indications and Cost-Effectiveness
 Balloon Sinuplasty: Indications and Cost-Effectiveness Marilene B. Wang, MD Professor, Department of Head and Neck Surgery David Geffen School of Medicine at UCLA Chief of Otolaryngology VA Greater Los
Balloon Sinuplasty: Indications and Cost-Effectiveness Marilene B. Wang, MD Professor, Department of Head and Neck Surgery David Geffen School of Medicine at UCLA Chief of Otolaryngology VA Greater Los
BALLOON SINUS OSTIAL DILATION
 BALLOON SINUS OSTIAL DILATION UnitedHealthcare Oxford Clinical Policy Policy Number: ENT 021.6 T2 Effective Date: April 1, 2018 Table of Contents Page INSTRUCTIONS FOR USE... 1 CONDITIONS OF COVERAGE...
BALLOON SINUS OSTIAL DILATION UnitedHealthcare Oxford Clinical Policy Policy Number: ENT 021.6 T2 Effective Date: April 1, 2018 Table of Contents Page INSTRUCTIONS FOR USE... 1 CONDITIONS OF COVERAGE...
White Paper: Balloon Sinuplasty for Chronic Sinusitis, The Latest Recommendations
 White Paper: Balloon Sinuplasty for Chronic Sinusitis, The Latest Recommendations For Health Plans, Medical Management Organizations and TPAs Executive Summary Despite recent advances in instrumentation
White Paper: Balloon Sinuplasty for Chronic Sinusitis, The Latest Recommendations For Health Plans, Medical Management Organizations and TPAs Executive Summary Despite recent advances in instrumentation
Chronic rhinosinusitis (CRS) is a significant health
 ORIGINAL ARTICLE The safety and performance of a maxillary sinus ostium self-dilation device: a pilot study Iain Hathorn, MBChB, FRCSEd (ORL-HNS), Al-Rahim Habib, MSc, Rachelle Dar Santos, BSc, CCRP and
ORIGINAL ARTICLE The safety and performance of a maxillary sinus ostium self-dilation device: a pilot study Iain Hathorn, MBChB, FRCSEd (ORL-HNS), Al-Rahim Habib, MSc, Rachelle Dar Santos, BSc, CCRP and
BALLOON SINUS OSTIAL DILATION
 UnitedHealthcare of California (HMO) UnitedHealthcare Benefits Plan of California (EPO/POS) UnitedHealthcare of Oklahoma, Inc. UnitedHealthcare of Oregon, Inc. UnitedHealthcare Benefits of Texas, Inc.
UnitedHealthcare of California (HMO) UnitedHealthcare Benefits Plan of California (EPO/POS) UnitedHealthcare of Oklahoma, Inc. UnitedHealthcare of Oregon, Inc. UnitedHealthcare Benefits of Texas, Inc.
Expand your in-office treatment options for sinusitis. The Vent-Os Sinus Dilation System is simple and cost-effective. Vent-Os. Sinus Dilation System
 Expand your in-office treatment options for sinusitis The Vent-Os Sinus Dilation System is simple and cost-effective Vent-Os Sinus Dilation System Vent-Os Sinus Dilation System A Fast and Effective In-Office
Expand your in-office treatment options for sinusitis The Vent-Os Sinus Dilation System is simple and cost-effective Vent-Os Sinus Dilation System Vent-Os Sinus Dilation System A Fast and Effective In-Office
Corporate Medical Policy
 Corporate Medical Policy Surgical Treatment of Sinus Disease Description of Procedure or Service Sinusitis refers to infection or inflammation of the sinuses, which are small openings in the bones of the
Corporate Medical Policy Surgical Treatment of Sinus Disease Description of Procedure or Service Sinusitis refers to infection or inflammation of the sinuses, which are small openings in the bones of the
The ORIOS 2 Study: Office-Based Balloon Sinus Dilation A prospective multi-center study of 203 patients
 The ORIOS 2 Study: Office-Based Balloon Sinus Dilation A prospective multi-center study of 203 patients Boris Karanfilov, MD, Ohio Sinus Institute, Dublin, OH 1 Stacey Silvers, MD, Madison ENT & Facial
The ORIOS 2 Study: Office-Based Balloon Sinus Dilation A prospective multi-center study of 203 patients Boris Karanfilov, MD, Ohio Sinus Institute, Dublin, OH 1 Stacey Silvers, MD, Madison ENT & Facial
Implantable Sinus Stents for Postoperative Use Following Endoscopic Sinus Surgery and for Recurrent Sinonasal Polyposis
 Medical Policy Manual Surgery, Policy No. 198 Implantable Sinus Stents for Postoperative Use Following Endoscopic Sinus Surgery and for Recurrent Sinonasal Polyposis Next Review: August 2019 Last Review:
Medical Policy Manual Surgery, Policy No. 198 Implantable Sinus Stents for Postoperative Use Following Endoscopic Sinus Surgery and for Recurrent Sinonasal Polyposis Next Review: August 2019 Last Review:
In-office, multisinus balloon dilation: 1-Year outcomes from a prospective, multicenter, open label trial DO NOT COPY METHODS.
 In-office, multisinus balloon dilation: 1-Year outcomes from a prospective, multicenter, open label trial James Gould, M.D., 1 Ian Alexander, M.D., 2 Edward Tomkin, D.O., 3 and David Brodner, M.D. 4 ABSTRACT
In-office, multisinus balloon dilation: 1-Year outcomes from a prospective, multicenter, open label trial James Gould, M.D., 1 Ian Alexander, M.D., 2 Edward Tomkin, D.O., 3 and David Brodner, M.D. 4 ABSTRACT
Site of Service: Select Surgical Procedures. Select a hyperlink below to be directed to that section.
 MEDICAL POLICY 7.01.559 Sinus Surgery BCBSA Ref. Policies: 7.01.105, 7.01.155 Effective Date: June 1, 2018 RELATED MEDICAL POLICIES: Last Revised: Sept. 21, 2018 7.01.558 Rhinoplasty Replaces: N/A 11.01.524
MEDICAL POLICY 7.01.559 Sinus Surgery BCBSA Ref. Policies: 7.01.105, 7.01.155 Effective Date: June 1, 2018 RELATED MEDICAL POLICIES: Last Revised: Sept. 21, 2018 7.01.558 Rhinoplasty Replaces: N/A 11.01.524
Corporate Medical Policy
 Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: surgical_treatment_of_sinus_disease 2/2010 8/2017 8/2018 8/2017 Description of Procedure or Service Chronic
Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: surgical_treatment_of_sinus_disease 2/2010 8/2017 8/2018 8/2017 Description of Procedure or Service Chronic
Site of Service - Select Surgical Procedures. Select a hyperlink below to be directed to that section.
 MEDICAL POLICY 7.01.559 Sinus Surgery BCBSA Ref. Policies: 7.01.105, 7.01.155 Effective Date: June 1, 2018* RELATED MEDICAL POLICIES: Last Revised: May 1, 2018 7.01.558 Rhinoplasty Replaces: N/A 11.01.524
MEDICAL POLICY 7.01.559 Sinus Surgery BCBSA Ref. Policies: 7.01.105, 7.01.155 Effective Date: June 1, 2018* RELATED MEDICAL POLICIES: Last Revised: May 1, 2018 7.01.558 Rhinoplasty Replaces: N/A 11.01.524
Sinus Surgery (Functional Endoscopic Sinus Surgery and Balloon Sinuplasty)
 Sinus Surgery (Functional Endoscopic Sinus Surgery and Balloon Sinuplasty) Date of Origin: 10/2015 Last Review Date: 06/28/2017 Effective Date: 07/01/2017 Dates Reviewed: 10/2015, 06/2017 Developed By:
Sinus Surgery (Functional Endoscopic Sinus Surgery and Balloon Sinuplasty) Date of Origin: 10/2015 Last Review Date: 06/28/2017 Effective Date: 07/01/2017 Dates Reviewed: 10/2015, 06/2017 Developed By:
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE
 NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Medical technology guidance SCOPE The XprESS Multi-Sinus Dilation System for the treatment of 1 Technology 1.1 Description of the technology The XprESS
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Medical technology guidance SCOPE The XprESS Multi-Sinus Dilation System for the treatment of 1 Technology 1.1 Description of the technology The XprESS
Protocol. Implantable Sinus Stents for Postoperative Use Following Endoscopic Sinus Surgery and for Recurrent Sinus Disease
 (701134) Medical Benefit Effective Date: 04/01/17 Next Review Date: 01/19 Preauthorization No Review Dates: 01/13, 01/14, 01/15, 01/16, 01/17, 01/18 This protocol considers this test or procedure investigational.
(701134) Medical Benefit Effective Date: 04/01/17 Next Review Date: 01/19 Preauthorization No Review Dates: 01/13, 01/14, 01/15, 01/16, 01/17, 01/18 This protocol considers this test or procedure investigational.
Dose-dependent effects of tobramycin in an animal model of Pseudomonas sinusitis Am J Rhino Jul-Aug; 21(4):423-7
 AMINOGLYCOSIDES Dose-dependent effects of tobramycin in an animal model of Pseudomonas sinusitis Am J Rhino. 2007 Jul-Aug; 21(4):423-7 http://www.ncbi.nlm.nih.gov/pubmed/17882910 Evaluation of the in-vivo
AMINOGLYCOSIDES Dose-dependent effects of tobramycin in an animal model of Pseudomonas sinusitis Am J Rhino. 2007 Jul-Aug; 21(4):423-7 http://www.ncbi.nlm.nih.gov/pubmed/17882910 Evaluation of the in-vivo
Surgical Management of Sinusitis (What About Balloons?) Relative Indications for Sinus Surgery in Children
 Surgical Management of Sinusitis (What About Balloons?) Andrew N. Goldberg M.D. Andrew H. Murr M.D. Michael J. Cunningham, M.D. Department of Otolaryngology and Communication Enhancement Children s Hospital
Surgical Management of Sinusitis (What About Balloons?) Andrew N. Goldberg M.D. Andrew H. Murr M.D. Michael J. Cunningham, M.D. Department of Otolaryngology and Communication Enhancement Children s Hospital
Implantable Sinus Stents for Postoperative Use Following Endoscopic Sinus Surgery
 Last Review Status/Date: March 2014 Page: 1 of 7 Use Following Endoscopic Sinus Surgery Description Sinus stents are devices that are used postoperatively following endoscopic sinus surgery (ESS). The
Last Review Status/Date: March 2014 Page: 1 of 7 Use Following Endoscopic Sinus Surgery Description Sinus stents are devices that are used postoperatively following endoscopic sinus surgery (ESS). The
US Drug Eluting Sinus Stent (DESS) Market: Size, Trends & Forecasts ( ) September 2016
 US Drug Eluting Sinus Stent (DESS) Market: Size, Trends & Forecasts (2016-2021) September 2016 US Drug Eluting Sinus Stent Market Report Scope of the Report The report entitled US Drug Eluting Sinus Stent
US Drug Eluting Sinus Stent (DESS) Market: Size, Trends & Forecasts (2016-2021) September 2016 US Drug Eluting Sinus Stent Market Report Scope of the Report The report entitled US Drug Eluting Sinus Stent
Implantable Sinus Stents for Postoperative Use Following Endoscopic Sinus Surgery
 Page: 1 of 8 Last Review Status/Date: March 2015 Use Following Endoscopic Sinus Surgery Description Sinus stents are devices that are used postoperatively following endoscopic sinus surgery (ESS). The
Page: 1 of 8 Last Review Status/Date: March 2015 Use Following Endoscopic Sinus Surgery Description Sinus stents are devices that are used postoperatively following endoscopic sinus surgery (ESS). The
POLICY PRODUCT VARIATIONS DESCRIPTION/BACKGROUND RATIONALE DEFINITIONS BENEFIT VARIATIONS DISCLAIMER CODING INFORMATION REFERENCES POLICY HISTORY
 Original Issue Date (Created): July 30, 2013 Most Recent Review Date (Revised): May 20, 2014 Effective Date: August 1, 2014 POLICY PRODUCT VARIATIONS DESCRIPTION/BACKGROUND RATIONALE DEFINITIONS BENEFIT
Original Issue Date (Created): July 30, 2013 Most Recent Review Date (Revised): May 20, 2014 Effective Date: August 1, 2014 POLICY PRODUCT VARIATIONS DESCRIPTION/BACKGROUND RATIONALE DEFINITIONS BENEFIT
Treatment of Maxillary Sinusitis Using the SinuSys Vent-Os Sinus Dilation System
 WHITE PAPER Treatment of Maxillary Sinusitis Using the SinuSys Vent-Os Sinus Dilation System Jerome Hester, MD Chief Medical Officer SinuSys Corporation Palo Alto, California USA Introduction Establishment
WHITE PAPER Treatment of Maxillary Sinusitis Using the SinuSys Vent-Os Sinus Dilation System Jerome Hester, MD Chief Medical Officer SinuSys Corporation Palo Alto, California USA Introduction Establishment
An Innovative Treatment Option for Patients with Recurrent Nasal Polyps
 An Innovative Treatment Option for Patients with Recurrent Nasal Polyps Burden of illness and management of Chronic Sinusitis with Nasal Polyps Continuum of care and polyp recurrence Clinical and health
An Innovative Treatment Option for Patients with Recurrent Nasal Polyps Burden of illness and management of Chronic Sinusitis with Nasal Polyps Continuum of care and polyp recurrence Clinical and health
Policy Effective January 29, 2019
 Corporate Medical Policy Surgical Treatment of Sinus Disease File Name: Origination: Last CAP Review: Next CAP Review: Last Review: surgical_treatment_of_sinus_disease 2/2010 8/2018 8/2019 8/2018 Policy
Corporate Medical Policy Surgical Treatment of Sinus Disease File Name: Origination: Last CAP Review: Next CAP Review: Last Review: surgical_treatment_of_sinus_disease 2/2010 8/2018 8/2019 8/2018 Policy
DIFFICULT-TO-TREAT CHRONIC
 MANAGEMENT STRATEGIES FOR DIFFICULT-TO-TREAT CHRONIC RHINOSINUSITIS DR ZULKEFLI HUSSEIN CONSULTANT EAR NOSE & THROAT SURGEON PANTAI HOSPITAL PENANG DISCLAIMER Nothing to disclose PENANG ISLAND, MALAYSIA
MANAGEMENT STRATEGIES FOR DIFFICULT-TO-TREAT CHRONIC RHINOSINUSITIS DR ZULKEFLI HUSSEIN CONSULTANT EAR NOSE & THROAT SURGEON PANTAI HOSPITAL PENANG DISCLAIMER Nothing to disclose PENANG ISLAND, MALAYSIA
2017 FACILITY AND PHYSICIAN REIMBURSEMENT GUIDE
 2017 AND PHYSICIAN REIMBURSEMENT GUIDE NASAL/SINUS ENDOSCOPIC SURGERY Some of the Current Procedure Terminology (CPT ) Codes for endoscopic nasal/sinus surgery are listed below. CPT codes 31295, 31296
2017 AND PHYSICIAN REIMBURSEMENT GUIDE NASAL/SINUS ENDOSCOPIC SURGERY Some of the Current Procedure Terminology (CPT ) Codes for endoscopic nasal/sinus surgery are listed below. CPT codes 31295, 31296
Sinusitis is a highly prevalent condition, affecting approximately
 ORIGINAL ARTICLE Office-based balloon sinus dilation: a prospective, multicenter study of 203 patients Boris Karanfilov, MD 1, Stacey Silvers, MD 2, Raza Pasha, MD 3, Ashley Sikand, MD 4, Alan Shikani,
ORIGINAL ARTICLE Office-based balloon sinus dilation: a prospective, multicenter study of 203 patients Boris Karanfilov, MD 1, Stacey Silvers, MD 2, Raza Pasha, MD 3, Ashley Sikand, MD 4, Alan Shikani,
FUNCTIONAL ENDOSCOPIC SINUS SURGERY (FESS)
 UnitedHealthcare Community Plan Medical Policy FUNCTIONAL ENDOSCOPIC SINUS SURGERY (FESS) Policy Number: CS144.D Effective Date: January 1, 2018 Table of Contents Page INSTRUCTIONS FOR USE... 1 BENEFIT
UnitedHealthcare Community Plan Medical Policy FUNCTIONAL ENDOSCOPIC SINUS SURGERY (FESS) Policy Number: CS144.D Effective Date: January 1, 2018 Table of Contents Page INSTRUCTIONS FOR USE... 1 BENEFIT
FUNCTIONAL ENDOSCOPIC SINUS SURGERY (FESS)
 FUNCTIONAL ENDOSCOPIC SINUS SURGERY (FESS) Protocol: SUR055 Effective Date: June 1, 2018 Table of Contents Page COMMERCIAL, MEDICAID AND MEDICARE COVERAGE RATIONALE... 1 DEFINITIONS... 2 DESCRIPTION OF
FUNCTIONAL ENDOSCOPIC SINUS SURGERY (FESS) Protocol: SUR055 Effective Date: June 1, 2018 Table of Contents Page COMMERCIAL, MEDICAID AND MEDICARE COVERAGE RATIONALE... 1 DEFINITIONS... 2 DESCRIPTION OF
Functional Endoscopic Sinus Surgery (FESS)
 Functional Endoscopic Sinus Surgery (FESS) Last Review Date: December 12, 2017 Number: MG.MM.SU.56C2 Medical Guideline Disclaimer Property of EmblemHealth. All rights reserved. The treating physician or
Functional Endoscopic Sinus Surgery (FESS) Last Review Date: December 12, 2017 Number: MG.MM.SU.56C2 Medical Guideline Disclaimer Property of EmblemHealth. All rights reserved. The treating physician or
PATIENT SELECTION AND MANAGEMENT OF IN-OFFICE BALLOON SINUS DILATION UNDER LOCAL ANESTHESIA IN ADOLESCENT PATIENTS
 WHITE PAPER PATIENT SELECTION AND MANAGEMENT OF IN-OFFICE BALLOON SINUS DILATION UNDER LOCAL ANESTHESIA IN ADOLESCENT PATIENTS Jeffrey S. Rosenbloom, MD Jeffrey Holland, AAS, CRC Alamo ENT Associates,
WHITE PAPER PATIENT SELECTION AND MANAGEMENT OF IN-OFFICE BALLOON SINUS DILATION UNDER LOCAL ANESTHESIA IN ADOLESCENT PATIENTS Jeffrey S. Rosenbloom, MD Jeffrey Holland, AAS, CRC Alamo ENT Associates,
Medical technologies guidance Published: 13 December 2016 nice.org.uk/guidance/mtg30
 XprESS multi sinus dilation system for treating chronic sinusitis Medical technologies guidance Published: 13 December 2016 nice.org.uk/guidance/mtg30 NICE 2018. All rights reserved. Subject to Notice
XprESS multi sinus dilation system for treating chronic sinusitis Medical technologies guidance Published: 13 December 2016 nice.org.uk/guidance/mtg30 NICE 2018. All rights reserved. Subject to Notice
Steroid-Eluting Ethmoidal Stent Versus Antero- -Posterior Ethmoidectomy: Comparison Of Efficacy And Safety In Allergic Patients
 Steroid-Eluting Ethmoidal Stent Versus Antero- -Posterior Ethmoidectomy: Comparison Of Efficacy And Safety In Allergic Patients Authors Contribution: A Study Design B Data Collection C Statistical Analysis
Steroid-Eluting Ethmoidal Stent Versus Antero- -Posterior Ethmoidectomy: Comparison Of Efficacy And Safety In Allergic Patients Authors Contribution: A Study Design B Data Collection C Statistical Analysis
MEDICAL POLICY SUBJECT: MAMMOGRAPHY: COMPUTER- AIDED DETECTION (CAD) POLICY NUMBER: CATEGORY: Technology Assessment
 MEDICAL POLICY SUBJECT: MAMMOGRAPHY: COMPUTER- PAGE: 1 OF: 5 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial product, including
MEDICAL POLICY SUBJECT: MAMMOGRAPHY: COMPUTER- PAGE: 1 OF: 5 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial product, including
FUNCTIONAL ENDOSCOPIC SINUS SURGERY (FESS)
 UnitedHealthcare Oxford Clinical Policy FUNCTIONAL ENDOSCOPIC SINUS SURGERY (FESS) Policy Number: ENT 022.5 T2 Effective Date: April 1, 2018 Table of Contents Page INSTRUCTIONS FOR USE... 1 CONDITIONS
UnitedHealthcare Oxford Clinical Policy FUNCTIONAL ENDOSCOPIC SINUS SURGERY (FESS) Policy Number: ENT 022.5 T2 Effective Date: April 1, 2018 Table of Contents Page INSTRUCTIONS FOR USE... 1 CONDITIONS
Implantable Sinus Stents for Postoperative Use Following Endoscopic Sinus Surgery and for Recurrent Sinus Disease
 Page: 1 of 8 Last Review Status/Date: March 2017 Implantable Sinus Stents for Postoperative Use Following Endoscopic Sinus Surgery and Description Sinus stents are devices used postoperatively following
Page: 1 of 8 Last Review Status/Date: March 2017 Implantable Sinus Stents for Postoperative Use Following Endoscopic Sinus Surgery and Description Sinus stents are devices used postoperatively following
MEDICAL POLICY I. POLICY FUNCTIONAL ENDOSCOPIC SINUS SURGERY FOR CHRONIC RHINOSINUSITIS MP POLICY TITLE POLICY NUMBER
 Original Issue Date (Created): 8/1/2018 Most Recent Review Date (Revised): 1/30/2018 Effective Date: 8/1/2018 POLICY PRODUCT VARIATIONS DESCRIPTION/BACKGROUND RATIONALE DEFINITIONS BENEFIT VARIATIONS DISCLAIMER
Original Issue Date (Created): 8/1/2018 Most Recent Review Date (Revised): 1/30/2018 Effective Date: 8/1/2018 POLICY PRODUCT VARIATIONS DESCRIPTION/BACKGROUND RATIONALE DEFINITIONS BENEFIT VARIATIONS DISCLAIMER
MEDICAL POLICY SUBJECT: COGNITIVE REHABILITATION. POLICY NUMBER: CATEGORY: Therapy/Rehabilitation
 MEDICAL POLICY SUBJECT: COGNITIVE REHABILITATION PAGE: 1 OF: 5 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial product (including
MEDICAL POLICY SUBJECT: COGNITIVE REHABILITATION PAGE: 1 OF: 5 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial product (including
Evaluation of the Change in Recent Diagnostic Criteria of Chronic Rhinosinusitis: A Cross-sectional Study
 Abhishek Ramadhin REVIEW ARTICLE 10.5005/jp-journals-10013-1246 Evaluation of the Change in Recent Diagnostic Criteria of Chronic Rhinosinusitis: A Cross-sectional Study Abhishek Ramadhin ABSTRACT There
Abhishek Ramadhin REVIEW ARTICLE 10.5005/jp-journals-10013-1246 Evaluation of the Change in Recent Diagnostic Criteria of Chronic Rhinosinusitis: A Cross-sectional Study Abhishek Ramadhin ABSTRACT There
MEDICAL POLICY SUBJECT: MAGNETIC ESOPHAGEAL RING/ MAGNETIC SPHINCTER AUGMENTATION FOR THE TREATMENT OF GASTROESOPHAGEAL REFLUX DISEASE (GERD)
 MEDICAL POLICY SUBJECT: MAGNETIC ESOPHAGEAL RING/ MAGNETIC SPHINCTER PAGE: 1 OF: 5 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial
MEDICAL POLICY SUBJECT: MAGNETIC ESOPHAGEAL RING/ MAGNETIC SPHINCTER PAGE: 1 OF: 5 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial
MP Implantable Sinus Stents for Postoperative Use Following Endoscopic Sinus Surgery and for Recurrent Sinus Disease. Related Policies None
 Medical Policy MP 7.01.134 Implantable Sinus Stents for Postoperative Use Following Endoscopic Sinus Surgery and for Recurrent Sinus BCBSA Ref. Policy: 7.01.134 Last Review: 02/26/2018 Effective Date:
Medical Policy MP 7.01.134 Implantable Sinus Stents for Postoperative Use Following Endoscopic Sinus Surgery and for Recurrent Sinus BCBSA Ref. Policy: 7.01.134 Last Review: 02/26/2018 Effective Date:
Clinical Policy Title: Propel (drug eluting devices after sinus surgery)
 Clinical Policy Title: Propel (drug eluting devices after sinus surgery) Clinical Policy Number: 10.03.07 Effective Date: July 1, 2017 Initial Review Date: May 19, 2017 Most Recent Review Date: May 1,
Clinical Policy Title: Propel (drug eluting devices after sinus surgery) Clinical Policy Number: 10.03.07 Effective Date: July 1, 2017 Initial Review Date: May 19, 2017 Most Recent Review Date: May 1,
Limitations of balloon sinuplasty in frontal sinus surgery
 Eur Arch Otorhinolaryngol (2011) 268:1463 1467 DOI 10.1007/s00405-011-1626-7 RHINOLOGY Limitations of balloon sinuplasty in frontal sinus surgery S. Heimgartner J. Eckardt D. Simmen H. R. Briner A. Leunig
Eur Arch Otorhinolaryngol (2011) 268:1463 1467 DOI 10.1007/s00405-011-1626-7 RHINOLOGY Limitations of balloon sinuplasty in frontal sinus surgery S. Heimgartner J. Eckardt D. Simmen H. R. Briner A. Leunig
MEDICAL POLICY SUBJECT: COCHLEAR IMPLANTS AND AUDITORY BRAINSTEM IMPLANTS. POLICY NUMBER: CATEGORY: Technology Assessment
 MEDICAL POLICY PAGE: 1 OF: 5 If the member's subscriber contract excludes coverage for a specific service it is not covered under that contract. In such cases, medical policy criteria are not applied.
MEDICAL POLICY PAGE: 1 OF: 5 If the member's subscriber contract excludes coverage for a specific service it is not covered under that contract. In such cases, medical policy criteria are not applied.
Cigna Medical Coverage Policy
 Cigna Medical Coverage Policy Subject Drug-Eluting Devices for Use Following Endoscopic Sinus Surgery Table of Contents Coverage Policy... 1 General Background... 1 Coding/Billing Information... 6 References...
Cigna Medical Coverage Policy Subject Drug-Eluting Devices for Use Following Endoscopic Sinus Surgery Table of Contents Coverage Policy... 1 General Background... 1 Coding/Billing Information... 6 References...
Single Multi-Functional Device
 Treat Multiple Sinuses with a Single Multi-Functional Device BALLOON DILATION SEEKER CONFIRMATION SUCTION IRRIGATION Treat the Frontal, Sphenoid and Maxillary Sinuses with a Single Multi-Functional Device
Treat Multiple Sinuses with a Single Multi-Functional Device BALLOON DILATION SEEKER CONFIRMATION SUCTION IRRIGATION Treat the Frontal, Sphenoid and Maxillary Sinuses with a Single Multi-Functional Device
Clinical Policy Title: Propel (drug eluting devices after sinus surgery)
 Clinical Policy Title: Propel (drug eluting devices after sinus surgery) Clinical Policy Number: 10.03.07 Effective Date: July 1, 2017 Initial Review Date: May 19, 2017 Most Recent Review Date: June 22,
Clinical Policy Title: Propel (drug eluting devices after sinus surgery) Clinical Policy Number: 10.03.07 Effective Date: July 1, 2017 Initial Review Date: May 19, 2017 Most Recent Review Date: June 22,
Eosinophilic Rhinosinusitis is Not a Disease of Ostiomeatal Occlusion
 The Laryngoscope VC 2013 The American Laryngological, Rhinological and Otological Society, Inc. Eosinophilic Rhinosinusitis is Not a Disease of Ostiomeatal Occlusion Kornkiat Snidvongs, MD; David Chin,
The Laryngoscope VC 2013 The American Laryngological, Rhinological and Otological Society, Inc. Eosinophilic Rhinosinusitis is Not a Disease of Ostiomeatal Occlusion Kornkiat Snidvongs, MD; David Chin,
Sphenopalatine Ganglion Block for Headache and Pain
 Medical Policy Manual Medicine, Policy No. 160 Sphenopalatine Ganglion Block for Headache and Pain Next Review: May 2019 Last Review: January 2019 Effective: February 1, 2019 IMPORTANT REMINDER Medical
Medical Policy Manual Medicine, Policy No. 160 Sphenopalatine Ganglion Block for Headache and Pain Next Review: May 2019 Last Review: January 2019 Effective: February 1, 2019 IMPORTANT REMINDER Medical
MEDICAL POLICY SUBJECT: RADIOFREQUENCY JOINT ABLATION / DENERVATION
 MEDICAL POLICY SUBJECT: RADIOFREQUENCY JOINT PAGE: 1 OF: 5 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial product (including
MEDICAL POLICY SUBJECT: RADIOFREQUENCY JOINT PAGE: 1 OF: 5 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial product (including
Implantable Sinus Stents for Postoperative Use Following Endoscopic Sinus Surgery and for Recurrent Sinus Disease
 Implantable Sinus Stents for Postoperative Use Following Endoscopic Sinus Surgery and for Recurrent Sinus Disease Policy Number: 7.01.134 Last Review: 8/2018 Origination: 12/2015 Next Review: 12/2018 Policy
Implantable Sinus Stents for Postoperative Use Following Endoscopic Sinus Surgery and for Recurrent Sinus Disease Policy Number: 7.01.134 Last Review: 8/2018 Origination: 12/2015 Next Review: 12/2018 Policy
Reasons for Failure and Surgical Revisions. Stil Kountakis, MD, PhD Professor and Chief, Division of Rhinology
 Reasons for Failure and Surgical Revisions Stil Kountakis, MD, PhD Professor and Chief, Division of Rhinology Medical College of Georgia of Georgia Regents University Department of Otolaryngology / Head
Reasons for Failure and Surgical Revisions Stil Kountakis, MD, PhD Professor and Chief, Division of Rhinology Medical College of Georgia of Georgia Regents University Department of Otolaryngology / Head
MEDICAL POLICY SUBJECT: TRANSRECTAL ULTRASOUND (TRUS)
 MEDICAL POLICY SUBJECT: TRANSRECTAL ULTRASOUND 06/16/05, 05/18/06, 03/15/07, 02/21/08 PAGE: 1 OF: 5 If the member's subscriber contract excludes coverage for a specific service it is not covered under
MEDICAL POLICY SUBJECT: TRANSRECTAL ULTRASOUND 06/16/05, 05/18/06, 03/15/07, 02/21/08 PAGE: 1 OF: 5 If the member's subscriber contract excludes coverage for a specific service it is not covered under
Corporate Medical Policy Septoplasty
 Corporate Medical Policy Septoplasty File Name: Origination: Last CAP Review: Next CAP Review: Last Review: septoplasty 4/1999 8/2018 8/2019 8/2018 Description of Procedure or Service There are many potential
Corporate Medical Policy Septoplasty File Name: Origination: Last CAP Review: Next CAP Review: Last Review: septoplasty 4/1999 8/2018 8/2019 8/2018 Description of Procedure or Service There are many potential
National Institute for Health and Care Excellence Medical Technologies Evaluation Programme
 National Institute for Health and Care Excellence Medical Technologies Evaluation Programme Consultation Comments table MTAC date: 23 September 2016 There were 25 consultation comments from 8 consultees
National Institute for Health and Care Excellence Medical Technologies Evaluation Programme Consultation Comments table MTAC date: 23 September 2016 There were 25 consultation comments from 8 consultees
2019 ACCLARENT REIMBURSEMENT GUIDE. Physician and Facility
 2019 ACCLARENT REIMBURSEMENT GUIDE Physician and Facility TABLE OF CONTENTS This guide has been developed to assist you in obtaining physician and facility reimbursement for: Nasal/Sinus Endoscopic Surgery
2019 ACCLARENT REIMBURSEMENT GUIDE Physician and Facility TABLE OF CONTENTS This guide has been developed to assist you in obtaining physician and facility reimbursement for: Nasal/Sinus Endoscopic Surgery
ORIGINAL ARTICLE. Computed Tomographic Staging and the Fate of the Dependent Sinuses in Revision Endoscopic Sinus Surgery
 Computed Tomographic Staging and the Fate of the Dependent es in Revision Endoscopic Surgery Neil Bhattacharyya, MD ORIGINAL ARTICLE Objectives: To determine the patterns of disease recurrence in chronic
Computed Tomographic Staging and the Fate of the Dependent es in Revision Endoscopic Surgery Neil Bhattacharyya, MD ORIGINAL ARTICLE Objectives: To determine the patterns of disease recurrence in chronic
CHRONIC RHINOSINUSITIS IN ADULTS
 CHRONIC RHINOSINUSITIS IN ADULTS SCOPE OF THE PRACTICE GUIDELINE This clinical practice guideline is for use by the Philippine Society of Otolaryngology-Head and Neck Surgery. It covers the diagnosis and
CHRONIC RHINOSINUSITIS IN ADULTS SCOPE OF THE PRACTICE GUIDELINE This clinical practice guideline is for use by the Philippine Society of Otolaryngology-Head and Neck Surgery. It covers the diagnosis and
FUNCTIONAL ENDOSCOPIC SINUS SURGERY (FESS)
 Oxford UnitedHealthcare Oxford Clinical Policy FUNCTIONAL ENDOSCOPIC SINUS SURGERY (FESS) Policy Number: ENT 022.6 T2 Effective Date: October 1, 2018 Table of Contents Page INSTRUCTIONS FOR USE... 1 CONDITIONS
Oxford UnitedHealthcare Oxford Clinical Policy FUNCTIONAL ENDOSCOPIC SINUS SURGERY (FESS) Policy Number: ENT 022.6 T2 Effective Date: October 1, 2018 Table of Contents Page INSTRUCTIONS FOR USE... 1 CONDITIONS
Incidence of accessory ostia in patients with chronic maxillary sinusitis
 International Journal of Otorhinolaryngology and Head and Neck Surgery Ghosh P et al. Int J Otorhinolaryngol Head Neck Surg. 2018 Mar;4(2):443-447 http://www.ijorl.com pissn 2454-5929 eissn 2454-5937 Original
International Journal of Otorhinolaryngology and Head and Neck Surgery Ghosh P et al. Int J Otorhinolaryngol Head Neck Surg. 2018 Mar;4(2):443-447 http://www.ijorl.com pissn 2454-5929 eissn 2454-5937 Original
Clinical Outcome of Endoscopic Surgery for Frontal Sinusitis
 Clinical Outcome of Endoscopic Surgery for Frontal Sinusitis Ralph Metson, MD; Richard E. Gliklich, MD ORIGINAL ARTICLE Objective: To determine the efficacy of endoscopic surgery for chronic frontal sinusitis.
Clinical Outcome of Endoscopic Surgery for Frontal Sinusitis Ralph Metson, MD; Richard E. Gliklich, MD ORIGINAL ARTICLE Objective: To determine the efficacy of endoscopic surgery for chronic frontal sinusitis.
FUNCTIONAL ENDOSCOPIC SINUS SURGERY (FESS)
 UnitedHealthcare Oxford Clinical Policy FUNCTIONAL ENDOSCOPIC SINUS SURGERY (FESS) Policy Number: ENT 022.3 T2 Effective Date: October 1, 2017 Table of Contents Page INSTRUCTIONS FOR USE... 1 CONDITIONS
UnitedHealthcare Oxford Clinical Policy FUNCTIONAL ENDOSCOPIC SINUS SURGERY (FESS) Policy Number: ENT 022.3 T2 Effective Date: October 1, 2017 Table of Contents Page INSTRUCTIONS FOR USE... 1 CONDITIONS
Frontal Sinus Drillout (Modified Lothrop Procedure): Long-Term Results in 204 Patients
 The Laryngoscope VC 2013 The American Laryngological, Rhinological and Otological Society, Inc. Frontal Sinus Drillout (Modified Lothrop Procedure): Long-Term Results in 204 Patients Jonathan Y. Ting,
The Laryngoscope VC 2013 The American Laryngological, Rhinological and Otological Society, Inc. Frontal Sinus Drillout (Modified Lothrop Procedure): Long-Term Results in 204 Patients Jonathan Y. Ting,
Delivering Innovation. Where It s Needed. August 2018
 Delivering Innovation. Where It s Needed. August 2018 Forward-Looking Statements Certain statements in this presentation constitute forward-looking statements within the meaning of the Securities Act of
Delivering Innovation. Where It s Needed. August 2018 Forward-Looking Statements Certain statements in this presentation constitute forward-looking statements within the meaning of the Securities Act of
Conventional Sinus Surgery Vs Fess
 IOSR Journal of Dental and Medical Sciences (IOSR-JDMS) e-issn: 2279-0853, p-issn: 2279-0861.Volume 16, Issue 7 Ver. III (July. 2017), PP 44-51 www.iosrjournals.org Conventional Sinus Surgery Vs Fess *
IOSR Journal of Dental and Medical Sciences (IOSR-JDMS) e-issn: 2279-0853, p-issn: 2279-0861.Volume 16, Issue 7 Ver. III (July. 2017), PP 44-51 www.iosrjournals.org Conventional Sinus Surgery Vs Fess *
2017 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY. MEASURE TYPE: Efficiency
 Measure #334: Adult Sinusitis: More than One Computerized Tomography (CT) Scan Within 90 Days for Chronic Sinusitis (Overuse) National Quality Strategy Domain: Efficiency and Cost Reduction 2017 OPTIONS
Measure #334: Adult Sinusitis: More than One Computerized Tomography (CT) Scan Within 90 Days for Chronic Sinusitis (Overuse) National Quality Strategy Domain: Efficiency and Cost Reduction 2017 OPTIONS
David C. Brodner, M.D.
 David C. Brodner, M.D. 8794 Boynton Beach Boulevard Suite 208 Boynton Beach, FL 33472 Office: (561) 735-8750 Cell: (561) 374-3308 Fax: (561) 735-8785 www.brodnermd.com DavidBrodnerMD@alumni.duke.edu SUMMARY:
David C. Brodner, M.D. 8794 Boynton Beach Boulevard Suite 208 Boynton Beach, FL 33472 Office: (561) 735-8750 Cell: (561) 374-3308 Fax: (561) 735-8785 www.brodnermd.com DavidBrodnerMD@alumni.duke.edu SUMMARY:
Rhinosinusitis. John Ramey, MD Joseph Russell, MD
 Rhinosinusitis John Ramey, MD Joseph Russell, MD Disclosure Statement RSFH as a continuing medical education provider, accredited by the South Carolina Medical Association, it is the policy of RSFH to
Rhinosinusitis John Ramey, MD Joseph Russell, MD Disclosure Statement RSFH as a continuing medical education provider, accredited by the South Carolina Medical Association, it is the policy of RSFH to
2016 PQRS OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY
 Measure #334: Adult Sinusitis: More than One Computerized Tomography (CT) Scan Within 90 Days for Chronic Sinusitis (Overuse) National Quality Strategy Domain: Efficiency and Cost Reduction 2016 PQRS OPTIONS
Measure #334: Adult Sinusitis: More than One Computerized Tomography (CT) Scan Within 90 Days for Chronic Sinusitis (Overuse) National Quality Strategy Domain: Efficiency and Cost Reduction 2016 PQRS OPTIONS
The ROLE OF ALPHINTERN IN TREATMENT OF NASAL POLYPOSIS
 The ROLE OF ALPHINTERN IN TREATMENT OF NASAL POLYPOSIS Prospective study was performed on patients diagnosed as chronic nasalpolyposis who referred to ENT clinic, Benghazi medical center, Benghazi- Libya
The ROLE OF ALPHINTERN IN TREATMENT OF NASAL POLYPOSIS Prospective study was performed on patients diagnosed as chronic nasalpolyposis who referred to ENT clinic, Benghazi medical center, Benghazi- Libya
Endoscopic Management Of A Giant Ethmoid Mucocele
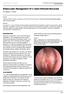 ISPUB.COM The Internet Journal of Otorhinolaryngology Volume 6 Number 1 S Ceylan, F Bora Citation S Ceylan, F Bora.. The Internet Journal of Otorhinolaryngology. 2006 Volume 6 Number 1. Abstract We present
ISPUB.COM The Internet Journal of Otorhinolaryngology Volume 6 Number 1 S Ceylan, F Bora Citation S Ceylan, F Bora.. The Internet Journal of Otorhinolaryngology. 2006 Volume 6 Number 1. Abstract We present
MEDICAL POLICY SUBJECT: TUMOR CHEMORESISTANCE AND CHEMOSENSITIVITY ASSAYS. POLICY NUMBER: CATEGORY: Laboratory Test
 MEDICAL POLICY SUBJECT: TUMOR CHEMORESISTANCE AND PAGE: 1 OF: 5 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial product, including
MEDICAL POLICY SUBJECT: TUMOR CHEMORESISTANCE AND PAGE: 1 OF: 5 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial product, including
OSTEITIS IN CRS. Rhinology Chair Meeting presented by Amal Binhazza a
 OSTEITIS IN CRS Rhinology Chair Meeting presented by Amal Binhazza a ROAD MAP Definition. pathophysiology. Diagnosis. Grading systems. Clinical implications. Management. OSTEITIS Presence of new bone formation,
OSTEITIS IN CRS Rhinology Chair Meeting presented by Amal Binhazza a ROAD MAP Definition. pathophysiology. Diagnosis. Grading systems. Clinical implications. Management. OSTEITIS Presence of new bone formation,
A comprehensive study on complications of endoscopic sinus surgery
 International Journal of Otorhinolaryngology and Head and Neck Surgery Shyras JAD et al. Int J Otorhinolaryngol Head Neck Surg. 2017 Jul;3(3):472-477 http://www.ijorl.com pissn 2454-5929 eissn 2454-5937
International Journal of Otorhinolaryngology and Head and Neck Surgery Shyras JAD et al. Int J Otorhinolaryngol Head Neck Surg. 2017 Jul;3(3):472-477 http://www.ijorl.com pissn 2454-5929 eissn 2454-5937
Sinusitis: Medical and Surgical Management. R. Jonathan Lara, DO, FAOCO. Sonoran Ear Nose & Throat
 Sinusitis: Medical and Surgical Management R. Jonathan Lara, DO, FAOCO Sonoran Ear Nose & Throat 27 th Annual Southwestern Conference on Medicine April 26, 2018 What are sinuses? Air filled cavities of
Sinusitis: Medical and Surgical Management R. Jonathan Lara, DO, FAOCO Sonoran Ear Nose & Throat 27 th Annual Southwestern Conference on Medicine April 26, 2018 What are sinuses? Air filled cavities of
Chronic Sinusitis. Acute Sinusitis. Sinusitis. Anatomy of the Paranasal Sinuses. Sinusitis. Medical Topics - Sinusitis
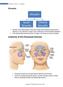 1 Acute Chronic is the inflammation of the inner lining of the parnasal sinuses due to infection or non-infectious causes such as allergies or environmental pullutants. If the inflammation lasts more than
1 Acute Chronic is the inflammation of the inner lining of the parnasal sinuses due to infection or non-infectious causes such as allergies or environmental pullutants. If the inflammation lasts more than
7/28/2017. Disclaimer. AOA Practice Builder Session: Coding & Coverage of Drug eluting Sinus Implants. Introduction and Agenda
 AOA Practice Builder Session: Coding & Coverage of Drug eluting Sinus Implants Betsy Gross, RN, BSN, CPC, CENTC Vice President, Health Economics & Reimbursement DELIVERING INNOVATION. Intersect ENT WHERE
AOA Practice Builder Session: Coding & Coverage of Drug eluting Sinus Implants Betsy Gross, RN, BSN, CPC, CENTC Vice President, Health Economics & Reimbursement DELIVERING INNOVATION. Intersect ENT WHERE
MEDICAL POLICY SUBJECT: COMPUTER ASSISTED NAVIGATION FOR KNEE AND HIP ARTHROPLASTY
 MEDICAL POLICY SUBJECT: COMPUTER ASSISTED PAGE: 1 OF: 5 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial product, including an
MEDICAL POLICY SUBJECT: COMPUTER ASSISTED PAGE: 1 OF: 5 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial product, including an
Hitting the Jackpot Sinuses, Scopes, and Surgery
 Hitting the Jackpot Sinuses, Scopes, and Surgery Candice Fenildo holds an Associates Degree in Health Sciences and is Certified in Healthcare Compliance (CHC), a Certified Professional Coder (CPC), a Certified
Hitting the Jackpot Sinuses, Scopes, and Surgery Candice Fenildo holds an Associates Degree in Health Sciences and is Certified in Healthcare Compliance (CHC), a Certified Professional Coder (CPC), a Certified
Retrospective Analysis of Patients with Allergy Sinusitis
 Original article: Retrospective Analysis of Patients with Allergy Sinusitis G.S. Thalor Senior Specialist (MS) (department of Oto Rhino Laryngology), Govt. S.K. Hospital, Sikar, Rajasthan, India. Corresponding
Original article: Retrospective Analysis of Patients with Allergy Sinusitis G.S. Thalor Senior Specialist (MS) (department of Oto Rhino Laryngology), Govt. S.K. Hospital, Sikar, Rajasthan, India. Corresponding
Sphenoid rhinosinusitis associated with abducens nerve palsy Case report
 Romanian Journal of Rhinology, Volume 8, No. 30, April-June 2018 CASE REPORT Sphenoid rhinosinusitis associated with abducens nerve palsy Case report Lucian Lapusneanu 1, Marlena Radulescu 1, Florin Ghita
Romanian Journal of Rhinology, Volume 8, No. 30, April-June 2018 CASE REPORT Sphenoid rhinosinusitis associated with abducens nerve palsy Case report Lucian Lapusneanu 1, Marlena Radulescu 1, Florin Ghita
Sinusitis: Medical and Surgical Management. R. Jonathan Lara, DO, FAOCO. Sonoran Ear Nose & Throat
 Sinusitis: Medical and Surgical Management R. Jonathan Lara, DO, FAOCO Sonoran Ear Nose & Throat 27 th Annual Southwestern Conference on Medicine April 26, 2018 What are sinuses? Air filled cavities of
Sinusitis: Medical and Surgical Management R. Jonathan Lara, DO, FAOCO Sonoran Ear Nose & Throat 27 th Annual Southwestern Conference on Medicine April 26, 2018 What are sinuses? Air filled cavities of
treating chronic sinusitis
 NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Medical technology consultation document XprESS multi-sinus dilation system for The National Institute for Health and Care Excellence (NICE) is producing
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Medical technology consultation document XprESS multi-sinus dilation system for The National Institute for Health and Care Excellence (NICE) is producing
Pediatric Endoscopic Sinus Surgery in a Tertiary Government Hospital: Patient Profile and Surgical Indications
 Philippine Journal Of Otolaryngology-Head And Neck Surgery Vol. 4 No. January June ichael Joseph C. David, D Gil. Vicente, D, Antonio H. Chua, D, Jose R. Reyes emorial edical Center St. Luke s edical Center
Philippine Journal Of Otolaryngology-Head And Neck Surgery Vol. 4 No. January June ichael Joseph C. David, D Gil. Vicente, D, Antonio H. Chua, D, Jose R. Reyes emorial edical Center St. Luke s edical Center
