Tranexamic acid for intracerebral haemorrhage: (TICH-2) trial. Nikola Sprigg On behalf of the TICH-2 investigators
|
|
|
- Bruno Smith
- 5 years ago
- Views:
Transcription
1 Tranexamic acid for intracerebral haemorrhage: (TICH-2) trial Nikola Sprigg On behalf of the TICH-2 investigators
2 Disclosures TICH-2 funded by National Institute of Health Research Health Technology Assessment (NIHR HTA project code 11_129_109), and Swiss Heart Foundation. The views expressed are those of the authors and not necessarily those of the NHS, the NIHR or the Department of Health and Social Care.
3 TICH-2 would not have been possible TICH-2 participants and families Nottingham Stroke Research Partnership Group Christine Knott and Malcolm Jarvis Prof Philip Bath TICH-2 Deputy CI TICH-2 Trials team in Nottingham Sally Utton, Diane Havard, Hayley Foster, Pauline Hyman- Taylor Sponsor's representative: Angela Shone (University of Nottingham) Protocol development - Alexandra Erven, Gillian Bumphrey, Diane Whitham (Nottingham Clinical Trials Unit) Independent Steering Committee Prof Colin Baigent (Chair, Oxford), Prof Yvo Roos (Amsterdam), Prof Matthew Walters (Glasgow), Christine Knott (Nottingham) Data Monitoring Committee Data Monitoring Committee John Bamford (Chair, Leeds), Martin Bland (York), Graham Venables (Sheffield), Wei Tan Events (outcome, SAE) Adjudicators Timothy England (Derby), Amit Mistri (Nottingham) Neuroimaging Adjudicators Lead Robert Dineen (Nottingham), Alessandro Adami (Italy), Lesley Cala (Australia), Ana Casado (Edinburgh), Rebecca Gallagher (Leicester) TICH-2 Collaborators in UK Ass Prof Tim England, Ass Prof Rob Dineen, Prof David Whynes, Prof Leila Duley, Prof Tom Robinson, Prof Stuart J Pocock, Prof Ian Roberts Prof Christine Roffe, Prof David Werring, Rustam Al-Shahi Salman TICH-2 International Advisory Committee Denmark Prof Christensen, Georgia Prof Beridze, Hungary Prof Bereczki, Italy Prof Ciccone, Malaysia J Thanabalan, Poland Prof Czlonkowska, Republic of Ireland R Collins, Spain Prof Egea-Guerrero, Switzerland Prof Lyrer, Turkey Prof Ozturk The TICH-2 sites THANK YOU TO EVERYONE
4 Intracerebral haemorrhage Significant early mortality and morbidity Haematoma expansion occurs early and is associated with bad outcome Preventing haematoma expansion may improve outcome Spot sign may predict haematoma expansion 1 but not routinely performed Haemostatic approaches: Recombinant Factor VIIa no evidence of benefit on functional outcome despite reduction in haematoma expansion 2 Tranexamic acid - anti-fibrinolytic reduces death due to bleeding in trauma and post partum haemorrhage 3 Intensive BP lowering improved functional outcome despite no significant effect on haematoma expansion [1] Demchuck et al. Lancet Neurol 2012 [2] Al Shahi-Salman et al. Cochrane 2018 [3] Gayet-Ageron et al. Lancet 2018
5 Methods
6 Design: Participants Multi-centre, double-blind, blinded-outcome, placebo-controlled trial Inclusion: Adult no upper age limit Spontaneous intracerebral haemorrhage <8 hours symptom onset Exclusion: Secondary ICH (e.g. anticoagulation, AVM, tumour, trauma, SAH) Contra-indication to tranexamic acid (VTE) Pregnancy (do pregnancy test if unsure) Pre-morbid disability (mrs >4) Glasgow coma scale <5 Life expectancy < 3 months due to other disease (e.g. advanced metastatic cancer) Consent: Written informed consent from each participant if they had capacity If participants could not give consent, a relative or representative gave proxy consent in accordance with ethics approval in each country
7 Study procedures Intervention: Tranexamic acid 1g/10 mins,1g/8 hours iv (Sharp plc) Comparator: Placebo identical regime Primary outcome: Primary outcome: modified Rankin Scale at day 90 (central telephone) Pre-specified secondary outcomes: Radiological Haematoma expansion, change in haematoma volume Haematoma expansion defined >6mls or 33% change Safety Early death, serious adverse events, safety outcomes (DVT, PE, MI, ACS, Seizures) Function Barthel index, cognition, mood, HRQOL all day 90 Treatment allocation was concealed from all staff involved in the trial Adopted in UK by National Institute Health Research Clinical Research Network, and registered as ISRCTN
8 Statistical methods Sample size 2000 participants 1,2 : assuming significance of 5%, power of 90%, ordinal odds ratio of 0 79 Randomisation 1:1 by internet: stratification by country and minimisation on key prognostic factors: age; sex; time since onset; systolic blood pressure; stroke severity (NIHSS); IVH; known history of antiplatelet treatment. Analysis of primary outcome: shift in modified Rankin Scale at day 90 using ordinal logistic regression, with adjustment for stratification and minimisation criteria Analyses followed the intention-to-treat (ITT) principle Pre-specified subgroups: age, time, systolic blood pressure, NIHSS score, presence of IVH, history of antiplatelet treatment, spot sign on CT angiography, ICH location and ethnicity [1] Protocol paper TICH-2 Sprigg et al. IJS 2016; [2] Statistical analysis plan Flaherty et al. Trials 2017
9 Results
10 Recruitment Date trial started 01 MAR 2013 Date recruitment started 14 MAR 2013 Date recruitment closed 30 SEP 2017 Number of patients randomised 2325 Number of centres 124 Number of countries 12 Country Total recruited UK 1910 Georgia 141 Italy 96 Malaysia 46 Switzerland 46 Denmark 39 Republic of Ireland 17 Hungary 9 Turkey 9 Sweden 8 Poland 3 Spain 1
11 Trial flow diagram Enrollment Randomised (n= 2325) Allocated to tranexamic acid (n= 1161) Received allocated intervention (n= 1150) Did not receive allocated intervention: No treatment given (n= 9) Not known if treatment given (n= 2) Allocation Allocated to placebo (n= 1164) Received allocated intervention (n= 1157) Did not receive allocated intervention: No treatment given (n= 6) Not known if treatment given (n= 1) Day 2 assessment completed (n= 1161) Lost to follow-up (n= 0) Died by day 2 (n= 40) Follow-Up, day 2 Day 2 assessment completed (n= 1164) Lost to follow-up (n= 0) Died by day 2 (n= 57) Day 7 assessment completed (n= 1161) Lost to follow-up (n= 0) Died by day 7 (n= 101) Follow-Up, day 7 Day 7 assessment completed (n= 1164) Lost to follow-up (n= 0) Died by day 7 (n= 123) Analysed (n= 1152) Died by day 90 (n= 250) Excluded from analysis: Withdrawn (n= 6) Lost to follow-up (n= 3) Analysis, day 90 Analysed (n= 1155) Died by day 90 (n= 249) Excluded from analysis: Withdrawn (n= 3) Lost to follow-up (n= 6)
12 Baseline characteristics Groups well matched at baseline Mean time to enrollment 3.6 hours Moderate stroke severity Predominance of deep haematoma location CTA angiography performed in only 10% Baseline variable TXA Placebo Participants randomised n=1161 n=1164 Age (years)* 69 1 (13 7) 68 7 (13 9) Sex*, male (%) 642 (55 3%) 659 (56 6%) White (%) 986 (85%) 992 (85 2%) Onset to randomisation (hours)* 3 6 [2 6, 5 1] 3 7 [2 6, 5 0] 3 hours (%) 421 (36 3%) 412 (35 4%) 4.5 hours (%) 779 (67 1%) 796 (68 4%) Previous antiplatelet therapy* 316 (27 2%) 295 (25 3%) Pre-stroke mrs, 0 [0, 1] 0 [0, 1] Glasgow coma scale, 13 (2 2) 14 (2 1) NIHSS score*, 13 (7 5) 13 (7 5) Systolic blood pressure* 172 (27 5) 174 (26 8) Haematoma location Supra-tentorial lobar haematoma (%) 379 (32 6%) 359 (30 8%) Supra-tentorial deep haematoma (%) 675 (58 1%) 696 (59 8%) Infra-tentorial haematoma (%) 73 (6 3%) 76 (6 5%) Haematoma volume (ml), 14 1 [5 9, 32 4] 12 5 [5 1, 31 9] Intraventricular haemorrhage*, yes (%) 382 (32 9%) 363 (31 2%) CT angiography performed,(%) 121 (10 6%) 128 (11 3%) Spot positive (%) 24 (19 8%) 32 (25 0%) Spot negative (%) 97 (80 2%) 96 (75 0%) *minimisation criteria, Mean (SD),Median [IQR]
13 Baseline characteristics Groups well matched at baseline Mean time to enrollment 3.6 hours Moderate stroke severity Predominance of deep haematoma location CTA angiography performed in only 10% Baseline variable TXA Placebo Participants randomised n=1161 n=1164 Age (years)* 69 1 (13 7) 68 7 (13 9) Sex*, male (%) 642 (55 3%) 659 (56 6%) White (%) 986 (85%) 992 (85 2%) Onset to randomisation (hours)* 3 6 [2 6, 5 1] 3 7 [2 6, 5 0] 3 hours (%) 421 (36 3%) 412 (35 4%) 4.5 hours (%) 779 (67 1%) 796 (68 4%) Previous antiplatelet therapy* 316 (27 2%) 295 (25 3%) Pre-stroke mrs, 0 [0, 1] 0 [0, 1] Glasgow coma scale, 13 (2 2) 14 (2 1) NIHSS score*, 13 (7 5) 13 (7 5) Systolic blood pressure* 172 (27 5) 174 (26 8) Haematoma location Supra-tentorial lobar haematoma (%) 379 (32 6%) 359 (30 8%) Supra-tentorial deep haematoma (%) 675 (58 1%) 696 (59 8%) Infra-tentorial haematoma (%) 73 (6 3%) 76 (6 5%) Haematoma volume (ml), 14 1 [5 9, 32 4] 12 5 [5 1, 31 9] Intraventricular haemorrhage*, yes (%) 382 (32 9%) 363 (31 2%) CT angiography performed,(%) 121 (10 6%) 128 (11 3%) Spot positive (%) 24 (19 8%) 32 (25 0%) Spot negative (%) 97 (80 2%) 96 (75 0%) *minimisation criteria, Mean (SD),Median [IQR]
14 Baseline characteristics Groups well matched at baseline Mean time to enrollment 3.6 hours Moderate stroke severity Predominance of deep haematoma location CTA angiography performed in only 10% Baseline variable TXA Placebo Participants randomised n=1161 n=1164 Age (years)* 69 1 (13 7) 68 7 (13 9) Sex*, male (%) 642 (55 3%) 659 (56 6%) White (%) 986 (85%) 992 (85 2%) Onset to randomisation (hours)* 3 6 [2 6, 5 1] 3 7 [2 6, 5 0] 3 hours (%) 421 (36 3%) 412 (35 4%) 4.5 hours (%) 779 (67 1%) 796 (68 4%) Previous antiplatelet therapy* 316 (27 2%) 295 (25 3%) Pre-stroke mrs, 0 [0, 1] 0 [0, 1] Glasgow coma scale, 13 (2 2) 14 (2 1) NIHSS score*, 13 (7 5) 13 (7 5) Systolic blood pressure* 172 (27 5) 174 (26 8) Haematoma location Supra-tentorial lobar haematoma (%) 379 (32 6%) 359 (30 8%) Supra-tentorial deep haematoma (%) 675 (58 1%) 696 (59 8%) Infra-tentorial haematoma (%) 73 (6 3%) 76 (6 5%) Haematoma volume (ml), 14 1 [5 9, 32 4] 12 5 [5 1, 31 9] Intraventricular haemorrhage*, yes (%) 382 (32 9%) 363 (31 2%) CT angiography performed,(%) 121 (10 6%) 128 (11 3%) Spot positive (%) 24 (19 8%) 32 (25 0%) Spot negative (%) 97 (80 2%) 96 (75 0%) *minimisation criteria, Mean (SD),Median [IQR]
15 Baseline characteristics Groups well matched at baseline Mean time to enrollment 3.6 hours Moderate stroke severity Predominance of deep haematoma location CTA angiography performed in only 10% Baseline variable TXA Placebo Participants randomised n=1161 n=1164 Age (years)* 69 1 (13 7) 68 7 (13 9) Sex*, male (%) 642 (55 3%) 659 (56 6%) White (%) 986 (85%) 992 (85 2%) Onset to randomisation (hours)* 3 6 [2 6, 5 1] 3 7 [2 6, 5 0] 3 hours (%) 421 (36 3%) 412 (35 4%) 4.5 hours (%) 779 (67 1%) 796 (68 4%) Previous antiplatelet therapy* 316 (27 2%) 295 (25 3%) Pre-stroke mrs, 0 [0, 1] 0 [0, 1] Glasgow coma scale, 13 (2 2) 14 (2 1) NIHSS score*, 13 (7 5) 13 (7 5) Systolic blood pressure* 172 (27 5) 174 (26 8) Haematoma location Supra-tentorial lobar haematoma (%) 379 (32 6%) 359 (30 8%) Supra-tentorial deep haematoma (%) 675 (58 1%) 696 (59 8%) Infra-tentorial haematoma (%) 73 (6 3%) 76 (6 5%) Haematoma volume (ml), 14 1 [5 9, 32 4] 12 5 [5 1, 31 9] Intraventricular haemorrhage*, yes (%) 382 (32 9%) 363 (31 2%) CT angiography performed,(%) 121 (10 6%) 128 (11 3%) Spot positive (%) 24 (19 8%) 32 (25 0%) Spot negative (%) 97 (80 2%) 96 (75 0%) *minimisation criteria, Mean (SD),Median [IQR]
16 Baseline characteristics Groups well matched at baseline Mean time to enrollment 3.6 hours Moderate stroke severity Predominance of deep haematoma location CTA angiography performed in only 10% Baseline variable TXA Placebo Participants randomised n=1161 n=1164 Age (years)* 69 1 (13 7) 68 7 (13 9) Sex*, male (%) 642 (55 3%) 659 (56 6%) White (%) 986 (85%) 992 (85 2%) Onset to randomisation (hours)* 3 6 [2 6, 5 1] 3 7 [2 6, 5 0] 3 hours (%) 421 (36 3%) 412 (35 4%) 4.5 hours (%) 779 (67 1%) 796 (68 4%) Previous antiplatelet therapy* 316 (27 2%) 295 (25 3%) Pre-stroke mrs, 0 [0, 1] 0 [0, 1] Glasgow coma scale, 13 (2 2) 14 (2 1) NIHSS score*, 13 (7 5) 13 (7 5) Systolic blood pressure* 172 (27 5) 174 (26 8) Haematoma location Supra-tentorial lobar haematoma (%) 379 (32 6%) 359 (30 8%) Supra-tentorial deep haematoma (%) 675 (58 1%) 696 (59 8%) Infra-tentorial haematoma (%) 73 (6 3%) 76 (6 5%) Haematoma volume (ml), 14 1 [5 9, 32 4] 12 5 [5 1, 31 9] Intraventricular haemorrhage*, yes (%) 382 (32 9%) 363 (31 2%) CT angiography performed,(%) 121 (10 6%) 128 (11 3%) Spot positive (%) 24 (19 8%) 32 (25 0%) Spot negative (%) 97 (80 2%) 96 (75 0%) *minimisation criteria, Mean (SD),Median [IQR]
17 Baseline characteristics Groups well matched at baseline Mean time to enrollment 3.6 hours Moderate stroke severity Predominance of deep haematoma location CTA angiography performed in only 10% Baseline variable TXA Placebo Participants randomised n=1161 n=1164 Age (years)* 69 1 (13 7) 68 7 (13 9) Sex*, male (%) 642 (55 3%) 659 (56 6%) White (%) 986 (85%) 992 (85 2%) Onset to randomisation (hours)* 3 6 [2 6, 5 1] 3 7 [2 6, 5 0] 3 hours (%) 421 (36 3%) 412 (35 4%) 4.5 hours (%) 779 (67 1%) 796 (68 4%) Previous antiplatelet therapy* 316 (27 2%) 295 (25 3%) Pre-stroke mrs, 0 [0, 1] 0 [0, 1] Glasgow coma scale, 13 (2 2) 14 (2 1) NIHSS score*, 13 (7 5) 13 (7 5) Systolic blood pressure* 172 (27 5) 174 (26 8) Haematoma location Supra-tentorial lobar haematoma (%) 379 (32 6%) 359 (30 8%) Supra-tentorial deep haematoma (%) 675 (58 1%) 696 (59 8%) Infra-tentorial haematoma (%) 73 (6 3%) 76 (6 5%) Haematoma volume (ml), 14 1 [5 9, 32 4] 12 5 [5 1, 31 9] Intraventricular haemorrhage*, yes (%) 382 (32 9%) 363 (31 2%) CT angiography performed,(%) 121 (10 6%) 128 (11 3%) Spot positive (%) 24 (19 8%) 32 (25 0%) Spot negative (%) 97 (80 2%) 96 (75 0%) *minimisation criteria, Mean (SD),Median [IQR]
18 Adherence to treatment Good adherence to treatment 95% received all allocated treatment 98% received at least some treatment Mean time to treatment 21 minutes TXA Placebo p Number of participants randomised All randomised treatment received as intended, n (%) 1101 (94 8%) 1106 (95 0%) 0 96 Some randomised treatment received, n (%) 49 (4 2%) 51 (4 4%) 0 66 No randomised treatment received, n (%) 9 (0 8%) 6 (0 5%) 0 48 Not known what treatment received, n (%) 2 (0 2%) 1 (0 1%) 0 54 Protocol violation submitted for incorrect drug administration*, n (%) Time until first dose (minutes), median [IQR] {range} 31 (2.7%) 26 (2.2%) [13, 33] {1, 167} 21 [13, 32] {1, 196} 0 49
19 Primary outcome: shift analysis mrs day 90 No significant difference in primary outcome OR 0.88 ( ) p=0.11 No significant difference in binary outcome mrs >3 OR 0.82 ( ) p=0.08
20 Secondary outcomes Significant reduction in haematoma change and haematoma expansion By day 7 fewer patients had died in the TXA group No significant differences in any of the functional outcomes at day 90 Radiological TXA Placebo OR/MD* (95% CI) p * Adjusted for minimisation factors Change in haematoma volume from baseline to 24 hours 3 72 (15 9) 4 90 (16 0) MD: (-2 71, -0 04) Participants with haematoma expansion 265 (25 1%) 304 (28 7%) OR: 0 80 (0 66, 0 98) Day 7 Death by day 7, n (%) 101 (8 7%) 123 (10 6%) OR: 0 73 (0 53, 0 99) NIHSS day (8 3) (8 3) MD: (-0 94, 0 09) 0 10 Day 90 Death by day 90, n (%) 250 (21 5%) 249 (21 4%) HR: 0 92 (0 77, 1 10) 0 37 Discharge Length of stay in hospital (47 1) (48 1) MD: 1 09 (0 97, 1 24) 0 16 Disposition at discharge Home 465 (40 1%) 453 (38 9%) OR: 1 14 (0 93, 1 40) 0 20 Institution 505 (43 5%) 506 (43 5%) OR: 0 99 (0 83, 1 18) 0 90
21 Secondary outcomes Significant reduction in haematoma change and haematoma expansion By day 7 fewer patients had died in the TXA group No significant differences in any of the functional outcomes at day 90 Radiological TXA Placebo OR/MD* (95% CI) p * Adjusted for minimisation factors Change in haematoma volume from baseline to 24 hours 3 72 (15 9) 4 90 (16 0) MD: (-2 71, -0 04) Participants with haematoma expansion 265 (25 1%) 304 (28 7%) OR: 0 80 (0 66, 0 98) Day 7 Death by day 7, n (%) 101 (8 7%) 123 (10 6%) OR: 0 73 (0 53, 0 99) NIHSS day (8 3) (8 3) MD: (-0 94, 0 09) 0 10 Day 90 Death by day 90, n (%) 250 (21 5%) 249 (21 4%) HR: 0 92 (0 77, 1 10) 0 37 Discharge Length of stay in hospital (47 1) (48 1) MD: 1 09 (0 97, 1 24) 0 16 Disposition at discharge Home 465 (40 1%) 453 (38 9%) OR: 1 14 (0 93, 1 40) 0 20 Institution 505 (43 5%) 506 (43 5%) OR: 0 99 (0 83, 1 18) 0 90
22 Secondary outcomes Significant reduction in haematoma change and haematoma expansion By day 7 fewer patients had died in the TXA group No significant differences in any of the functional outcomes at day 90 Radiological TXA Placebo OR/MD* (95% CI) p * Adjusted for minimisation factors Change in haematoma volume from baseline to 24 hours 3 72 (15 9) 4 90 (16 0) MD: (-2 71, -0 04) Participants with haematoma expansion 265 (25 1%) 304 (28 7%) OR: 0 80 (0 66, 0 98) Day 7 Death by day 7, n (%) 101 (8 7%) 123 (10 6%) OR: 0 73 (0 53, 0 99) NIHSS day (8 3) (8 3) MD: (-0 94, 0 09) 0 10 Day 90 Death by day 90, n (%) 250 (21 5%) 249 (21 4%) HR: 0 92 (0 77, 1 10) 0 37 Discharge Length of stay in hospital (47 1) (48 1) MD: 1 09 (0 97, 1 24) 0 16 Disposition at discharge Home 465 (40 1%) 453 (38 9%) OR: 1 14 (0 93, 1 40) 0 20 Institution 505 (43 5%) 506 (43 5%) OR: 0 99 (0 83, 1 18) 0 90
23 Cumulative mortality No significant difference in cumulative mortality Adjusted HR 0.92 ( p=0.37)
24 Sub-groups Pre-specified sub-groups: Participants BP < 170mmHg favoured TXA 0.73 ( ) p= Participants onset < 4.5 hours favoured TXA 0.84 ( ) p=0.28 Exploratory post hoc: Participants haematoma size 30-60mls favoured TXA 0.66 ( ) p=0.84
25 Serious adverse events Participants in the TXA groups had fewer safety outcomes and SAEs There was no increase in venous thromboembolism Commonest SAEs were nervous system disorders and infections Median days to event [IQR] TXA 1161 Placebo 1164 p By day (32 6%) 417 (35 8%) By day (39 3%) 497 (42 7%) By day (44 9%) 556 (47 8%) Safety outcomes Death 1 [0, 15] 250 (21 5%) 249 (21 4%) 0 60 ACS or MI 5 [1, 53] 11 (0 9%) 6 (0 5%) 0 24 Deep vein thrombosis (DVT) 15 [10, 34] 19 (1 6%) 14 (1 2%) 0 41 Pulmonary embolism (PE) 16 [8, 29] 20 (1 7%) 23 (2 0%) 0 58 VTE (combined DVT/PE) 16 [10, 30] 39 (3 4%) 37 (3 2%) 0 98 Seizure / convulsions 1 [0, 3] 77 (6 6%) 85 (7 3%) 0 44 Ischaemic stroke or TIA 9 [2, 52] 16 (1 4%) 11 (0 9%) 0 27 SAEs by subcategory Infections and infestations 2 [1, 3] 98 (8 4%) 116 (10 0%) 0 14 Nervous system disorders 1 [0, 1] 149 (12 8%) 163 (14 0%) 0 33
26 Serious adverse events Participants in the TXA groups had fewer safety outcomes and SAEs There was no increase in venous thromboembolism Commonest SAEs were nervous system disorders and infections Median days to event [IQR] TXA 1161 Placebo 1164 p By day (32 6%) 417 (35 8%) By day (39 3%) 497 (42 7%) By day (44 9%) 556 (47 8%) Safety outcomes Death 1 [0, 15] 250 (21 5%) 249 (21 4%) 0 60 ACS or MI 5 [1, 53] 11 (0 9%) 6 (0 5%) 0 24 Deep vein thrombosis (DVT) 15 [10, 34] 19 (1 6%) 14 (1 2%) 0 41 Pulmonary embolism (PE) 16 [8, 29] 20 (1 7%) 23 (2 0%) 0 58 VTE (combined DVT/PE) 16 [10, 30] 39 (3 4%) 37 (3 2%) 0 98 Seizure / convulsions 1 [0, 3] 77 (6 6%) 85 (7 3%) 0 44 Ischaemic stroke or TIA 9 [2, 52] 16 (1 4%) 11 (0 9%) 0 27 SAEs by subcategory Infections and infestations 2 [1, 3] 98 (8 4%) 116 (10 0%) 0 14 Nervous system disorders 1 [0, 1] 149 (12 8%) 163 (14 0%) 0 33
27 Serious adverse events Participants in the TXA groups had fewer safety outcomes and SAEs There was no increase in venous thromboembolism Commonest SAEs were nervous system disorders and infections Median days to event [IQR] TXA 1161 Placebo 1164 p By day (32 6%) 417 (35 8%) By day (39 3%) 497 (42 7%) By day (44 9%) 556 (47 8%) Safety outcomes Death 1 [0, 15] 250 (21 5%) 249 (21 4%) 0 60 ACS or MI 5 [1, 53] 11 (0 9%) 6 (0 5%) 0 24 Deep vein thrombosis (DVT) 15 [10, 34] 19 (1 6%) 14 (1 2%) 0 41 Pulmonary embolism (PE) 16 [8, 29] 20 (1 7%) 23 (2 0%) 0 58 VTE (combined DVT/PE) 16 [10, 30] 39 (3 4%) 37 (3 2%) 0 98 Seizure / convulsions 1 [0, 3] 77 (6 6%) 85 (7 3%) 0 44 Ischaemic stroke or TIA 9 [2, 52] 16 (1 4%) 11 (0 9%) 0 27 SAEs by subcategory Infections and infestations 2 [1, 3] 98 (8 4%) 116 (10 0%) 0 14 Nervous system disorders 1 [0, 1] 149 (12 8%) 163 (14 0%) 0 33
28 Strengths and weaknesses Strengths: Largest RCT of haemostatic agent in ICH Broad inclusion criteria Good allocation concealment Minimal loss to follow-up (<1%) Weaknesses: Although 12 countries, participants mostly recruited from UK Broad inclusion led to heterogenous population Greater proportion of severe stroke compared to other ICH studies Despite efforts to include patients rapidly mean time to enrolment 3.6 hours with only 30% enrolled < 3 hours Caution needed interpreting the sub-group analysis
29 Discussion No significant effect on primary outcome at 90 days despite early benefits Possible that the anticipated treatment effect OR 0.79 was too large and the trial was unable to detect this Other TXA studies in trauma (CRASH-2) and post partum haemorrhage (WOMAN) much larger to detect modest effect sizes OR 0.81 & OR 0.85 Subsequent meta-analysis in 40,138 participants demonstrated that treatment needed to given < 3 hours to benefit [1] Possible that effect on attenuating haematoma growth (mean 1.5mls) was too modest to translate into improved outcome [1] Lancet Jan 13;391(10116):
30 Conclusion No significant difference in primary outcome mrs at day 90 (shift analysis) Evidence of antifibrinolytic effect reduced haematoma expansion, fewer early deaths No safety concerns in this population which was older with more comorbidities than previous tranexamic acid studies Results of several on-going studies awaited but likely larger randomised trials are needed More research needed to explore which sub-groups of patients may benefit Future work should focus on enrolling and treating patients earlier
31 Thank you
32 Hear more about TICH-2: Nottingham Trials Stand Exhibition Hall ICH and Hyperglycaemia 17 th May Hall H TICH-2 MRI Sub-study methods poster 17 th May TICH-2 MRI Sub -study Results 18 th May Hall C
Review of the TICH-2 Trial
 Review of the TICH-2 Trial Mikaela Hofer, PharmD PGY-1 Pharmacy Resident Pharmacy Grand Rounds September 18, 2018 2018 MFMER slide-1 Objectives Review the pharmacologic options to limit hematoma expansion
Review of the TICH-2 Trial Mikaela Hofer, PharmD PGY-1 Pharmacy Resident Pharmacy Grand Rounds September 18, 2018 2018 MFMER slide-1 Objectives Review the pharmacologic options to limit hematoma expansion
Tranexamic acid for hyperacute primary IntraCerebral Haemorrhage (TICH-2): an international randomised, placebo-controlled, phase 3 superiority trial
 Tranexamic acid for hyperacute primary IntraCerebral Haemorrhage (TICH-2): an international randomised, placebo-controlled, phase 3 superiority trial Nikola Sprigg, Katie Flaherty, Jason P Appleton, Rustam
Tranexamic acid for hyperacute primary IntraCerebral Haemorrhage (TICH-2): an international randomised, placebo-controlled, phase 3 superiority trial Nikola Sprigg, Katie Flaherty, Jason P Appleton, Rustam
Contents. Version 1.0: 01/02/2010 Protocol# ISRCTN Page 1 of 7
 Contents 1. INTRODUCTION... 2 2. STUDY SYNOPSIS... 2 3. STUDY OBJECTIVES... 2 3.1. Primary Objective... 2 3.2. Secondary Objectives... 2 3.3. Assessment of Objectives... 3 3.4. Change the Primary Objective
Contents 1. INTRODUCTION... 2 2. STUDY SYNOPSIS... 2 3. STUDY OBJECTIVES... 2 3.1. Primary Objective... 2 3.2. Secondary Objectives... 2 3.3. Assessment of Objectives... 3 3.4. Change the Primary Objective
FVIIa for Acute Hemorrhagic Stroke Administered at Earliest Time (FASTEST) Trial. Joseph P. Broderick, MD James Grotta, MD Jordan Elm, PhD
 FVIIa for Acute Hemorrhagic Stroke Administered at Earliest Time (FASTEST) Trial Joseph P. Broderick, MD James Grotta, MD Jordan Elm, PhD Background Intracerebral hemorrhage (ICH) accounts for more than
FVIIa for Acute Hemorrhagic Stroke Administered at Earliest Time (FASTEST) Trial Joseph P. Broderick, MD James Grotta, MD Jordan Elm, PhD Background Intracerebral hemorrhage (ICH) accounts for more than
FOCUS: Fluoxetine Or Control Under Supervision Results. Martin Dennis on behalf of the FOCUS collaborators
 FOCUS: Fluoxetine Or Control Under Supervision Results Martin Dennis on behalf of the FOCUS collaborators Background Pre clinical and imaging studies had suggested benefits from fluoxetine (and other SSRIs)
FOCUS: Fluoxetine Or Control Under Supervision Results Martin Dennis on behalf of the FOCUS collaborators Background Pre clinical and imaging studies had suggested benefits from fluoxetine (and other SSRIs)
Katharine Ker, David Prieto-Merino, Nikola Sprigg, Abda Mahmood, Philip Bath 4, Zhe Kang Law 4, Katie Flaherty 4, Ian Roberts 1
 STUDY PROTOCOL The effectiveness and safety of antifibrinolytics in patients with acute intracranial haemorrhage: statistical analysis plan for an individual patient data meta-analysis [version 2; referees:
STUDY PROTOCOL The effectiveness and safety of antifibrinolytics in patients with acute intracranial haemorrhage: statistical analysis plan for an individual patient data meta-analysis [version 2; referees:
What can we learn from the AVERT trial (so far)?
 South West Stroke Network Event, 29 th April, 2015 What can we learn from the AVERT trial (so far)? Peter Langhorne, Professor of stroke care, Glasgow University Disclosure PL was AVERT investigator and
South West Stroke Network Event, 29 th April, 2015 What can we learn from the AVERT trial (so far)? Peter Langhorne, Professor of stroke care, Glasgow University Disclosure PL was AVERT investigator and
A Very Early Rehabilitation Trial (AVERT): What we know, what we think and what s to come
 A Very Early Rehabilitation Trial (AVERT): What we know, what we think and what s to come The AVERT Trial Collaboration group Joshua Kwant, Blinded Assessor 17 th May 2016 NIMAST Nothing to disclose Disclosure
A Very Early Rehabilitation Trial (AVERT): What we know, what we think and what s to come The AVERT Trial Collaboration group Joshua Kwant, Blinded Assessor 17 th May 2016 NIMAST Nothing to disclose Disclosure
Bath, Philip M.W. and England, Timothy J. (2009) Thighlength compression stockings and DVT after stroke. Lancet. ISSN (In Press)
 Bath, Philip M.W. and England, Timothy J. (2009) Thighlength compression stockings and DVT after stroke. Lancet. ISSN 0140-6736 (In Press) Access from the University of Nottingham repository: http://eprints.nottingham.ac.uk/1087/1/lancet_clots_1_20090522_4.pdf
Bath, Philip M.W. and England, Timothy J. (2009) Thighlength compression stockings and DVT after stroke. Lancet. ISSN 0140-6736 (In Press) Access from the University of Nottingham repository: http://eprints.nottingham.ac.uk/1087/1/lancet_clots_1_20090522_4.pdf
Outlook for intracerebral haemorrhage after a MISTIE spell
 Outlook for intracerebral haemorrhage after a MISTIE spell David J Werring PhD FRCP Stroke Research Centre, Department of Brain Repair and Rehabilitation, UCL Institute of Neurology, National Hospital
Outlook for intracerebral haemorrhage after a MISTIE spell David J Werring PhD FRCP Stroke Research Centre, Department of Brain Repair and Rehabilitation, UCL Institute of Neurology, National Hospital
A common clinical dilemma. Ischaemic stroke or TIA with atrial fibrillation MRI scan with blood-sensitive imaging shows cerebral microbleeds
 Cerebral microbleeds and intracranial haemorrhage risk in patients with atrial fibrillation after acute ischaemic stroke or transient ischaemic attack: multicentre observational cohort study D. Wilson,
Cerebral microbleeds and intracranial haemorrhage risk in patients with atrial fibrillation after acute ischaemic stroke or transient ischaemic attack: multicentre observational cohort study D. Wilson,
Journal Club. 1. Develop a PICO (Population, Intervention, Comparison, Outcome) question for this study
 Journal Club Articles for Discussion Tissue plasminogen activator for acute ischemic stroke. The National Institute of Neurological Disorders and Stroke rt-pa Stroke Study Group. N Engl J Med. 1995 Dec
Journal Club Articles for Discussion Tissue plasminogen activator for acute ischemic stroke. The National Institute of Neurological Disorders and Stroke rt-pa Stroke Study Group. N Engl J Med. 1995 Dec
ICSS Safety Results NOT for PUBLICATION. June 2009 ICSS ICSS ICSS ICSS. International Carotid Stenting Study: Main Inclusion Criteria
 Safety Results NOT for The following slides were presented to the Investigators Meeting on 22/05/09 and most of them were also presented at the European Stroke Conference on 27/05/09 They are NOT for in
Safety Results NOT for The following slides were presented to the Investigators Meeting on 22/05/09 and most of them were also presented at the European Stroke Conference on 27/05/09 They are NOT for in
BACKGROUND AND SCIENTIFIC RATIONALE. Protocol Code: ISRCTN V 1.0 date 30 Jan 2012
 BACKGROUND AND SCIENTIFIC RATIONALE Protocol Code: ISRCTN15088122 V 1.0 date 30 Jan 2012 Traumatic Brain Injury 10 million killed or hospitalised every year 90% in low and middle income countries Mostly
BACKGROUND AND SCIENTIFIC RATIONALE Protocol Code: ISRCTN15088122 V 1.0 date 30 Jan 2012 Traumatic Brain Injury 10 million killed or hospitalised every year 90% in low and middle income countries Mostly
Thrombolysis-WAKE UP Intra-arterial interventions DEFUSE 3 Haemorrhagic Stroke - TICH 2 Secondary Prevention CROMIS 2 Secondary Prevention NAVIGATE
 Thrombolysis-WAKE UP Intra-arterial interventions DEFUSE 3 Haemorrhagic Stroke - TICH 2 Secondary Prevention CROMIS 2 Secondary Prevention NAVIGATE ESUS Progression of haematoma Anticoagulation Large ICH
Thrombolysis-WAKE UP Intra-arterial interventions DEFUSE 3 Haemorrhagic Stroke - TICH 2 Secondary Prevention CROMIS 2 Secondary Prevention NAVIGATE ESUS Progression of haematoma Anticoagulation Large ICH
ARTEMIS. ARixtra (fondaparinux) for ThromboEmbolism prevention in. a Medical Indications Study. NV Organon Protocol 63129
 ARTEMIS ARixtra (fondaparinux) for ThromboEmbolism prevention in NV Organon Protocol 63129 a Medical Indications Study Objective To demonstrate efficacy and to assess safety of oncedaily subcutaneous (SC)
ARTEMIS ARixtra (fondaparinux) for ThromboEmbolism prevention in NV Organon Protocol 63129 a Medical Indications Study Objective To demonstrate efficacy and to assess safety of oncedaily subcutaneous (SC)
An international, double-blind, phase III randomized trial. Main Results
 An international, double-blind, phase III randomized trial Main Results Robert Hart on behalf of the NAVIGATE ESUS Steering Committee and Investigators Sponsorship & Disclosures NAVIGATE ESUS was sponsored
An international, double-blind, phase III randomized trial Main Results Robert Hart on behalf of the NAVIGATE ESUS Steering Committee and Investigators Sponsorship & Disclosures NAVIGATE ESUS was sponsored
London School of Hygiene and Tropical Medicine, Keppel Street, London, UK 2
 CRASH-2 (Clinical Randomisation of an Antifibrinolytic in Significant Haemorrhage) intracranial bleeding study: the effect of tranexamic acid in traumatic brain injury a nested, randomised, placebo-controlled
CRASH-2 (Clinical Randomisation of an Antifibrinolytic in Significant Haemorrhage) intracranial bleeding study: the effect of tranexamic acid in traumatic brain injury a nested, randomised, placebo-controlled
Blood Pressure Reduction Among Acute Stroke Patients A Randomized Controlled Clinical Trial
 Blood Pressure Reduction Among Acute Stroke Patients A Randomized Controlled Clinical Trial Jiang He, Yonghong Zhang, Tan Xu, Weijun Tong, Shaoyan Zhang, Chung-Shiuan Chen, Qi Zhao, Jing Chen for CATIS
Blood Pressure Reduction Among Acute Stroke Patients A Randomized Controlled Clinical Trial Jiang He, Yonghong Zhang, Tan Xu, Weijun Tong, Shaoyan Zhang, Chung-Shiuan Chen, Qi Zhao, Jing Chen for CATIS
Drug Class Review Newer Oral Anticoagulant Drugs
 Drug Class Review Newer Oral Anticoagulant Drugs Final Original Report May 2016 The purpose of reports is to make available information regarding the comparative clinical effectiveness and harms of different
Drug Class Review Newer Oral Anticoagulant Drugs Final Original Report May 2016 The purpose of reports is to make available information regarding the comparative clinical effectiveness and harms of different
The Effect of Graduated Compression Stockings on Long-term Outcomes After Stroke The CLOTS Trials 1 and 2
 The Effect of Graduated Compression on Long-term Outcomes After Stroke The CLOTS Trials 1 and 2 The CLOTS Trials Collaboration; Martin Dennis, MD; Peter Sandercock, DM; John Reid, MBChB; Catriona Graham,
The Effect of Graduated Compression on Long-term Outcomes After Stroke The CLOTS Trials 1 and 2 The CLOTS Trials Collaboration; Martin Dennis, MD; Peter Sandercock, DM; John Reid, MBChB; Catriona Graham,
Modern Management of ICH
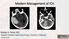 Modern Management of ICH Bradley A. Gross, MD Assistant Professor, Dept of Neurosurgery, University of Pittsburgh October 2018 ICH Background Assessment & Diagnosis Medical Management Surgical Management
Modern Management of ICH Bradley A. Gross, MD Assistant Professor, Dept of Neurosurgery, University of Pittsburgh October 2018 ICH Background Assessment & Diagnosis Medical Management Surgical Management
Does intermittent pneumatic compression reduce the risk of post stroke deep vein thrombosis? The CLOTS 3 trial: statistical analysis plan
 Dennis et al. Trials 2013, 14:66 TRIALS UPDATE Open Access Does intermittent pneumatic compression reduce the risk of post stroke deep vein thrombosis? The CLOTS 3 trial: statistical analysis plan Martin
Dennis et al. Trials 2013, 14:66 TRIALS UPDATE Open Access Does intermittent pneumatic compression reduce the risk of post stroke deep vein thrombosis? The CLOTS 3 trial: statistical analysis plan Martin
Clinical Study Synopsis
 Clinical Study Synopsis This Clinical Study Synopsis is provided for patients and healthcare professionals to increase the transparency of Bayer's clinical research. This document is not intended to replace
Clinical Study Synopsis This Clinical Study Synopsis is provided for patients and healthcare professionals to increase the transparency of Bayer's clinical research. This document is not intended to replace
INTRACEREBRAL HAEMORRHAGE:
 INTRACEREBRAL HAEMORRHAGE: WHAT IS THE CAUSE? Prof. Charlotte Cordonnier Head, Department of neurology & stroke centre Director, Lille haemorrhagic stroke research program Lille University Hospital France
INTRACEREBRAL HAEMORRHAGE: WHAT IS THE CAUSE? Prof. Charlotte Cordonnier Head, Department of neurology & stroke centre Director, Lille haemorrhagic stroke research program Lille University Hospital France
Klinikum Frankfurt Höchst
 Blood pressure management in hemorrhagic stroke Blood pressure in acute ICH Do we need additional trials after INTERACT2 and ATTACH-II? Focus.de Department of Neurology,, Germany Department of Neurology,
Blood pressure management in hemorrhagic stroke Blood pressure in acute ICH Do we need additional trials after INTERACT2 and ATTACH-II? Focus.de Department of Neurology,, Germany Department of Neurology,
Recombinant Factor VIIa for Intracerebral Hemorrhage
 Recombinant Factor VIIa for Intracerebral Hemorrhage January 24, 2006 Justin Lee Pharmacy Resident University Health Network Outline 1. Introduction to patient case 2. Overview of intracerebral hemorrhage
Recombinant Factor VIIa for Intracerebral Hemorrhage January 24, 2006 Justin Lee Pharmacy Resident University Health Network Outline 1. Introduction to patient case 2. Overview of intracerebral hemorrhage
GSK Medicine: Study Number: Title: Rationale: Phase: Study Period: Study Design: Centres: Indication: Treatment: Objectives:
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
Early mobilization after stroke What do we know (so far)?
 NICIS Neurosciences in Critical Care International Symposium 19 th June, 2015 Early mobilization after stroke What do we know (so far)? Peter Langhorne, Professor of stroke care, Glasgow University Acknowledgements
NICIS Neurosciences in Critical Care International Symposium 19 th June, 2015 Early mobilization after stroke What do we know (so far)? Peter Langhorne, Professor of stroke care, Glasgow University Acknowledgements
Deep vein thrombosis and its prevention in critically ill adults Attia J, Ray J G, Cook D J, Douketis J, Ginsberg J S, Geerts W H
 Deep vein thrombosis and its prevention in critically ill adults Attia J, Ray J G, Cook D J, Douketis J, Ginsberg J S, Geerts W H Authors' objectives To systematically review the incidence of deep vein
Deep vein thrombosis and its prevention in critically ill adults Attia J, Ray J G, Cook D J, Douketis J, Ginsberg J S, Geerts W H Authors' objectives To systematically review the incidence of deep vein
Improving the Analysis and Interpretation of Vascular Prevention Trials
 Improving the Analysis and Interpretation of Vascular Prevention Trials Philip M W Bath Stroke, Division of Clinical Neuroscience University of Nottingham Work with Stuart Pocock, Chamila Geeganage, Lisa
Improving the Analysis and Interpretation of Vascular Prevention Trials Philip M W Bath Stroke, Division of Clinical Neuroscience University of Nottingham Work with Stuart Pocock, Chamila Geeganage, Lisa
Antifibrinolytic drugs for acute traumatic injury(review)
 Cochrane Database of Systematic Reviews Antifibrinolytic drugs for acute traumatic injury(review) KerK,RobertsI,ShakurH,CoatsTJ KerK,RobertsI,ShakurH,CoatsTJ. Antifibrinolytic drugs for acute traumatic
Cochrane Database of Systematic Reviews Antifibrinolytic drugs for acute traumatic injury(review) KerK,RobertsI,ShakurH,CoatsTJ KerK,RobertsI,ShakurH,CoatsTJ. Antifibrinolytic drugs for acute traumatic
Articles. Funding National Institutes of Health Research Health Technology Assessment Programme, British Heart Foundation.
 Antiplatelet therapy with aspirin, clopidogrel, and dipyridamole versus clopidogrel alone or aspirin and dipyridamole in patients with acute cerebral ischaemia (TARDIS): a randomised, open-label, phase
Antiplatelet therapy with aspirin, clopidogrel, and dipyridamole versus clopidogrel alone or aspirin and dipyridamole in patients with acute cerebral ischaemia (TARDIS): a randomised, open-label, phase
Triple versus guideline antiplatelet therapy to prevent recurrence after acute ischaemic stroke or transient ischaemic attack: the TARDIS RCT
 Triple versus guideline antiplatelet therapy to prevent recurrence after acute ischaemic stroke or transient ischaemic attack: the TARDIS RCT Philip M Bath, 1,2 * Lisa J Woodhouse, 1 Jason P Appleton,
Triple versus guideline antiplatelet therapy to prevent recurrence after acute ischaemic stroke or transient ischaemic attack: the TARDIS RCT Philip M Bath, 1,2 * Lisa J Woodhouse, 1 Jason P Appleton,
Controversies in Hemorrhagic Stroke Management. Sarah L. Livesay, DNP, RN, ACNP-BC, ACNS-BC Associate Professor Rush University
 Controversies in Hemorrhagic Stroke Management Sarah L. Livesay, DNP, RN, ACNP-BC, ACNS-BC Associate Professor Rush University Disclosures AHA/ASA Outline Blood pressure VTE Coagulopathy Early mobilization
Controversies in Hemorrhagic Stroke Management Sarah L. Livesay, DNP, RN, ACNP-BC, ACNS-BC Associate Professor Rush University Disclosures AHA/ASA Outline Blood pressure VTE Coagulopathy Early mobilization
Acute Stroke Care: the Nuts and Bolts of it. ECASS I and II ATLANTIS. Chris V. Fanale, MD Colorado Neurological Institute Swedish Medical Center
 Acute Stroke Care: the Nuts and Bolts of it Chris V. Fanale, MD Colorado Neurological Institute Swedish Medical Center ECASS I and II tpa for patients presenting
Acute Stroke Care: the Nuts and Bolts of it Chris V. Fanale, MD Colorado Neurological Institute Swedish Medical Center ECASS I and II tpa for patients presenting
Hypertensive Haemorrhagic Stroke. Dr Philip Lam Thuon Mine
 Hypertensive Haemorrhagic Stroke Dr Philip Lam Thuon Mine Intracerebral Haemorrhage Primary ICH Spontaneous rupture of small vessels damaged by HBP Basal ganglia, thalamus, pons and cerebellum Amyloid
Hypertensive Haemorrhagic Stroke Dr Philip Lam Thuon Mine Intracerebral Haemorrhage Primary ICH Spontaneous rupture of small vessels damaged by HBP Basal ganglia, thalamus, pons and cerebellum Amyloid
Restart or stop antithrombotics after intracerebral haemorrhage (ICH)?
 Restart or stop antithrombotics after intracerebral haemorrhage (ICH)? Rustam Al-Shahi Salman professor of clinical neurology & honorary consultant neurologist www.rush.ed.ac.uk @BleedingStroke /bleedingstroke
Restart or stop antithrombotics after intracerebral haemorrhage (ICH)? Rustam Al-Shahi Salman professor of clinical neurology & honorary consultant neurologist www.rush.ed.ac.uk @BleedingStroke /bleedingstroke
Cover Page. The handle holds various files of this Leiden University dissertation.
 Cover Page The handle http://hdl.handle.net/188/20915 holds various files of this Leiden University dissertation. Author: Flinterman, Linda Elisabeth Title: Risk factors for a first and recurrent venous
Cover Page The handle http://hdl.handle.net/188/20915 holds various files of this Leiden University dissertation. Author: Flinterman, Linda Elisabeth Title: Risk factors for a first and recurrent venous
Stroke Update. Lacunar 19% Thromboembolic 6% SAH 13% ICH 13% Unknown 32% Hemorrhagic 26% Ischemic 71% Other 3% Cardioembolic 14%
 Stroke Update Michel Torbey, MD, MPH, FAHA, FNCS Medical Director, Neurovascular Stroke Center Professor Department of Neurology and Neurosurgery The Ohio State University Wexner Medical Center Objectives
Stroke Update Michel Torbey, MD, MPH, FAHA, FNCS Medical Director, Neurovascular Stroke Center Professor Department of Neurology and Neurosurgery The Ohio State University Wexner Medical Center Objectives
Lothian Audit of the Treatment of Cerebral Haemorrhage (LATCH)
 1. INTRODUCTION Stroke physicians, emergency department doctors, and neurologists are often unsure about which patients they should refer for neurosurgical intervention. Early neurosurgical evacuation
1. INTRODUCTION Stroke physicians, emergency department doctors, and neurologists are often unsure about which patients they should refer for neurosurgical intervention. Early neurosurgical evacuation
Canadian Best Practice Recommendations for Stroke Care 3.7 Acute Intracerebral Hemorrhage
 Last Updated: May 21st, 2013 Canadian Best Practice Recommendations for Stroke Care 3.7 Acute Intracerebral Hemorrhage Canadian Best Practice Recommendations for Stroke Care 2012-2013 Update Contents Search
Last Updated: May 21st, 2013 Canadian Best Practice Recommendations for Stroke Care 3.7 Acute Intracerebral Hemorrhage Canadian Best Practice Recommendations for Stroke Care 2012-2013 Update Contents Search
Efficacy of beta-blockers in heart failure patients with atrial fibrillation: An individual patient data meta-analysis
 Efficacy of beta-blockers in heart failure patients with atrial fibrillation: An individual patient data meta-analysis Dipak Kotecha, MD PhD on behalf of the Selection of slides presented at the European
Efficacy of beta-blockers in heart failure patients with atrial fibrillation: An individual patient data meta-analysis Dipak Kotecha, MD PhD on behalf of the Selection of slides presented at the European
Technology appraisal guidance Published: 26 August 2015 nice.org.uk/guidance/ta354
 Edoxaban for treating and for preventingenting deep vein thrombosis and pulmonary embolism Technology appraisal guidance Published: 26 August 2015 nice.org.uk/guidance/ta354 NICE 2017. All rights reserved.
Edoxaban for treating and for preventingenting deep vein thrombosis and pulmonary embolism Technology appraisal guidance Published: 26 August 2015 nice.org.uk/guidance/ta354 NICE 2017. All rights reserved.
Clinical Study Synopsis
 Clinical Study Synopsis This Clinical Study Synopsis is provided for patients and healthcare professionals to increase the transparency of Bayer's clinical research. This document is not intended to replace
Clinical Study Synopsis This Clinical Study Synopsis is provided for patients and healthcare professionals to increase the transparency of Bayer's clinical research. This document is not intended to replace
COMMITTEE FOR PROPRIETARY MEDICINAL PRODUCTS (CPMP)
 The European Agency for the Evaluation of Medicinal Products Evaluation of Medicines for Human Use London, 16 December 1999 COMMITTEE FOR PROPRIETARY MEDICINAL PRODUCTS (CPMP) NOTE FOR GUIDANCE ON CLINICAL
The European Agency for the Evaluation of Medicinal Products Evaluation of Medicines for Human Use London, 16 December 1999 COMMITTEE FOR PROPRIETARY MEDICINAL PRODUCTS (CPMP) NOTE FOR GUIDANCE ON CLINICAL
MRI carotid plaque imaging predicts future stroke in patients with mild to moderate stenosis ICAD study
 MRI carotid plaque imaging predicts future stroke in patients with mild to moderate stenosis ICAD study Richard Simpson, Senior Clinical Vascular Scientist and Stroke Association Junior Research Training
MRI carotid plaque imaging predicts future stroke in patients with mild to moderate stenosis ICAD study Richard Simpson, Senior Clinical Vascular Scientist and Stroke Association Junior Research Training
Updates in venous thromboembolism. Cecilia Becattini University of Perugia
 Updates in venous thromboembolism Cecilia Becattini University of Perugia News for VTE Diagnosis Treatment the acute phase the agents Pulmonary embolism: diagnosis Vein ultrasonography Meta-analysis 15
Updates in venous thromboembolism Cecilia Becattini University of Perugia News for VTE Diagnosis Treatment the acute phase the agents Pulmonary embolism: diagnosis Vein ultrasonography Meta-analysis 15
Clinical Study Synopsis
 Clinical Study Synopsis This Clinical Study Synopsis is provided for patients and healthcare professionals to increase the transparency of Bayer's clinical research. This document is not intended to replace
Clinical Study Synopsis This Clinical Study Synopsis is provided for patients and healthcare professionals to increase the transparency of Bayer's clinical research. This document is not intended to replace
Kenneth W. Mahaffey, MD and Keith AA Fox, MB ChB
 Once-daily oral direct factor Xa inhibition Compared with vitamin K antagonism for prevention of stroke and Embolism Trial in Atrial Fibrillation Kenneth W. Mahaffey, MD and Keith AA Fox, MB ChB on behalf
Once-daily oral direct factor Xa inhibition Compared with vitamin K antagonism for prevention of stroke and Embolism Trial in Atrial Fibrillation Kenneth W. Mahaffey, MD and Keith AA Fox, MB ChB on behalf
Efficient analysis of ordinal functional outcome scales
 Efficient analysis of ordinal functional outcome scales Gordon D Murray University of Edinburgh Outline of presentation Functional outcome scales Ordinal analysis Case study: SCAST Results Points to consider
Efficient analysis of ordinal functional outcome scales Gordon D Murray University of Edinburgh Outline of presentation Functional outcome scales Ordinal analysis Case study: SCAST Results Points to consider
Treatment of Acute Hemorrhagic Stroke 5th QSVS Neurovascular Conference Dar Dowlatshahi MD PhD FRCPC Sept 14, 2012
 Treatment of Acute Hemorrhagic Stroke 5th QSVS Neurovascular Conference Dar Dowlatshahi MD PhD FRCPC Sept 14, 2012 Disclosure of potential conflicts of interest Quebec Society of Vascular Sciences presents
Treatment of Acute Hemorrhagic Stroke 5th QSVS Neurovascular Conference Dar Dowlatshahi MD PhD FRCPC Sept 14, 2012 Disclosure of potential conflicts of interest Quebec Society of Vascular Sciences presents
New Frontiers in Intracerebral Hemorrhage
 New Frontiers in Intracerebral Hemorrhage Ryan Hakimi, DO, MS Director, Neuro ICU Director, Inpatient Neurology Services Greenville Health System Clinical Associate Professor Department of Medicine (Neurology)
New Frontiers in Intracerebral Hemorrhage Ryan Hakimi, DO, MS Director, Neuro ICU Director, Inpatient Neurology Services Greenville Health System Clinical Associate Professor Department of Medicine (Neurology)
EAST MULTICENTER STUDY DATA DICTIONARY
 EAST MULTICENTER STUDY DATA DICTIONARY Does the Addition of Daily Aspirin to Standard Deep Venous Thrombosis Prophylaxis Reduce the Rate of Venous Thromboembolic Events? Data Entry Points and appropriate
EAST MULTICENTER STUDY DATA DICTIONARY Does the Addition of Daily Aspirin to Standard Deep Venous Thrombosis Prophylaxis Reduce the Rate of Venous Thromboembolic Events? Data Entry Points and appropriate
An introduction to Quality by Design. Dr Martin Landray University of Oxford
 An introduction to Quality by Design Dr Martin Landray University of Oxford Criteria for a good trial Ask an IMPORTANT question Answer it RELIABLY Quality Quality is the absence of errors that matter to
An introduction to Quality by Design Dr Martin Landray University of Oxford Criteria for a good trial Ask an IMPORTANT question Answer it RELIABLY Quality Quality is the absence of errors that matter to
Apixaban for stroke prevention in atrial fibrillation. August 2010
 Apixaban for stroke prevention in atrial fibrillation August 2010 This technology summary is based on information available at the time of research and a limited literature search. It is not intended to
Apixaban for stroke prevention in atrial fibrillation August 2010 This technology summary is based on information available at the time of research and a limited literature search. It is not intended to
Life after ARUBA: Management of Unruptured Brain Arteriovenous Malformations (AVMs)
 Life after ARUBA: Management of Unruptured Brain Arteriovenous Malformations (AVMs) Eric L. Zager, MD University of Pennsylvania Department of Neurosurgery No Disclosures Brain AVMs Incidence ~1 in 100,000
Life after ARUBA: Management of Unruptured Brain Arteriovenous Malformations (AVMs) Eric L. Zager, MD University of Pennsylvania Department of Neurosurgery No Disclosures Brain AVMs Incidence ~1 in 100,000
Boehringer Ingelheim Page 6 of 178 Study report for non-interventional studies based on existing data BI Study Number
 Page 6 of 178 report for non-interventional studies based on existing data BI Number 1160.144 c14462719-01 International GmbH or one or more of its affiliated companies 1. ABSTRACT Title of study: Keywords:
Page 6 of 178 report for non-interventional studies based on existing data BI Number 1160.144 c14462719-01 International GmbH or one or more of its affiliated companies 1. ABSTRACT Title of study: Keywords:
Definition พ.ญ.ส ธ ดา เย นจ นทร. Epidemiology. Definition 5/25/2016. Seizures after stroke Can we predict? Poststroke seizure
 Seizures after stroke Can we predict? พ.ญ.ส ธ ดา เย นจ นทร PMK Epilepsy Annual Meeting 2016 Definition Poststroke seizure : single or multiple convulsive episode(s) after stroke and thought to be related
Seizures after stroke Can we predict? พ.ญ.ส ธ ดา เย นจ นทร PMK Epilepsy Annual Meeting 2016 Definition Poststroke seizure : single or multiple convulsive episode(s) after stroke and thought to be related
AVERT Trial Debate: Implications for Practice Have we AVERT ed the real message?
 AVERT Trial Debate: Implications for Practice Have we AVERT ed the real message? Jackie Bosch, PhD, OT Reg(Ont) School of Rehabilitation Science McMaster University Vince DePaul PT PhD School of Rehabilitation
AVERT Trial Debate: Implications for Practice Have we AVERT ed the real message? Jackie Bosch, PhD, OT Reg(Ont) School of Rehabilitation Science McMaster University Vince DePaul PT PhD School of Rehabilitation
Critical Thinking A tour through the science of neuroscience
 Critical Thinking A tour through the science of neuroscience NEBM 10032/5 Publication Bias Emily S Sena Centre for Clinical Brain Sciences, University of Edinburgh slides at @CAMARADES_ Bias Biases in
Critical Thinking A tour through the science of neuroscience NEBM 10032/5 Publication Bias Emily S Sena Centre for Clinical Brain Sciences, University of Edinburgh slides at @CAMARADES_ Bias Biases in
Position statement: Anti-coagulants and Risk Assessment
 Position statement: Anti-coagulants and Risk Assessment Document information Protective marking: NOT PROTECTVELY MARKED Author: Matt Johnston Force/Organisation: College of Policing NPCC Coordination Committee
Position statement: Anti-coagulants and Risk Assessment Document information Protective marking: NOT PROTECTVELY MARKED Author: Matt Johnston Force/Organisation: College of Policing NPCC Coordination Committee
CADTH CANADIAN DRUG EXPERT COMMITTEE FINAL RECOMMENDATION
 CADTH CANADIAN DRUG EXPERT COMMITTEE FINAL RECOMMENDATION Edoxaban (Lixiana SERVIER Canada Inc.) Indication: Prevention of Stroke and Systemic Embolic Events in Patients With Nonvalvular Atrial Fibrillation
CADTH CANADIAN DRUG EXPERT COMMITTEE FINAL RECOMMENDATION Edoxaban (Lixiana SERVIER Canada Inc.) Indication: Prevention of Stroke and Systemic Embolic Events in Patients With Nonvalvular Atrial Fibrillation
Experiences with interim trial monitoring, particularly with early stopped trials
 Experiences with interim trial monitoring, particularly with early stopped trials 1 Robert J Glynn, ScD Divisions of Preventive Medicine and Pharmacoepidemiology & Pharmacoeconomics, Brigham & Women s
Experiences with interim trial monitoring, particularly with early stopped trials 1 Robert J Glynn, ScD Divisions of Preventive Medicine and Pharmacoepidemiology & Pharmacoeconomics, Brigham & Women s
TXA: NOT JUST A ONE TRICK PONY TERRY MCGOVERN DO, MPH VITUITY, ST JOHN S REGIONAL MEDICAL CENTER, OXNARD, CA
 TXA: NOT JUST A ONE TRICK PONY TERRY MCGOVERN DO, MPH VITUITY, ST JOHN S REGIONAL MEDICAL CENTER, OXNARD, CA DISCLOSURES I have no relevant financial relationships with the manufacturer(s) of any commercial
TXA: NOT JUST A ONE TRICK PONY TERRY MCGOVERN DO, MPH VITUITY, ST JOHN S REGIONAL MEDICAL CENTER, OXNARD, CA DISCLOSURES I have no relevant financial relationships with the manufacturer(s) of any commercial
Boehringer-Ingelheim satellite symposium Ligue cardiologique belge 13/05/2017
 Boehringer-Ingelheim satellite symposium Ligue cardiologique belge 13/05/2017 Dr André Peeters Service de Neurologie Cliniques Universitaires Saint-Luc / U.C.L. 1200 BRUXELLES Introduction 1. Aim NOACS
Boehringer-Ingelheim satellite symposium Ligue cardiologique belge 13/05/2017 Dr André Peeters Service de Neurologie Cliniques Universitaires Saint-Luc / U.C.L. 1200 BRUXELLES Introduction 1. Aim NOACS
Protocol. This trial protocol has been provided by the authors to give readers additional information about their work.
 Protocol This trial protocol has been provided by the authors to give readers additional information about their work. Protocol for: Anderson CS, Robinson T, Lindley RI, et al. Low-dose versus standard-dose
Protocol This trial protocol has been provided by the authors to give readers additional information about their work. Protocol for: Anderson CS, Robinson T, Lindley RI, et al. Low-dose versus standard-dose
FOCUS: Fluoxetine Or Control Under Supervision. Gillian Mead and Martin Dennis Co-chief investigators, on behalf of the FOCUS team
 FOCUS: Fluoxetine Or Control Under Supervision Gillian Mead and Martin Dennis Co-chief investigators, on behalf of the FOCUS team Background Several small trials suggest a benefit of SSRI in prevention
FOCUS: Fluoxetine Or Control Under Supervision Gillian Mead and Martin Dennis Co-chief investigators, on behalf of the FOCUS team Background Several small trials suggest a benefit of SSRI in prevention
REVEAL: Randomized placebo-controlled trial of anacetrapib in 30,449 patients with atherosclerotic vascular disease
 REVEAL: Randomized placebo-controlled trial of anacetrapib in 30,449 patients with atherosclerotic vascular disease Martin Landray and Louise Bowman on behalf of the HPS 3 / TIMI 55 - REVEAL Collaborative
REVEAL: Randomized placebo-controlled trial of anacetrapib in 30,449 patients with atherosclerotic vascular disease Martin Landray and Louise Bowman on behalf of the HPS 3 / TIMI 55 - REVEAL Collaborative
Anticoagulation Therapy in SELECTeD Cancer Patients at Risk of Recurrence of Venous Thromboembolism
 Anticoagulation Therapy in SELECTeD Cancer Patients at Risk of Recurrence of Venous Thromboembolism Annie Young PhD Professor of Nursing, University of Warwick, UK on behalf of the select-d Collaborative
Anticoagulation Therapy in SELECTeD Cancer Patients at Risk of Recurrence of Venous Thromboembolism Annie Young PhD Professor of Nursing, University of Warwick, UK on behalf of the select-d Collaborative
Dr Ben Turner. Consultant Neurologist and Honorary Senior Lecturer Barts and The London NHS Trust London Bridge Hospital
 Stroke Management Dr Ben Turner Consultant Neurologist and Honorary Senior Lecturer Barts and The London NHS Trust London Bridge Hospital Introduction Stroke is the major cause of disability in the developed
Stroke Management Dr Ben Turner Consultant Neurologist and Honorary Senior Lecturer Barts and The London NHS Trust London Bridge Hospital Introduction Stroke is the major cause of disability in the developed
Technology appraisal guidance Published: 17 December 2014 nice.org.uk/guidance/ta327
 Dabigatran an etexilate for the treatment and secondary prevention ention of deep vein thrombosis and/or pulmonary embolism Technology appraisal guidance Published: 17 December 2014 nice.org.uk/guidance/ta327
Dabigatran an etexilate for the treatment and secondary prevention ention of deep vein thrombosis and/or pulmonary embolism Technology appraisal guidance Published: 17 December 2014 nice.org.uk/guidance/ta327
Rivaroxaban (Xarelto) in the management of stroke and DVT
 Rivaroxaban (Xarelto) in the management of stroke and DVT Steve Chaplin MSc, MRPharmS, Victoria Haunton BM, DGM, MRCP, Thompson Robinson BMedSci, MD, FRCP and Catherine Bagot MD, FRCPath KEY POINTS is
Rivaroxaban (Xarelto) in the management of stroke and DVT Steve Chaplin MSc, MRPharmS, Victoria Haunton BM, DGM, MRCP, Thompson Robinson BMedSci, MD, FRCP and Catherine Bagot MD, FRCPath KEY POINTS is
Stroke in the ED. Dr. William Whiteley. Scottish Senior Clinical Fellow University of Edinburgh Consultant Neurologist NHS Lothian
 Stroke in the ED Dr. William Whiteley Scottish Senior Clinical Fellow University of Edinburgh Consultant Neurologist NHS Lothian 2016 RCP Guideline for Stroke RCP guidelines for acute ischaemic stroke
Stroke in the ED Dr. William Whiteley Scottish Senior Clinical Fellow University of Edinburgh Consultant Neurologist NHS Lothian 2016 RCP Guideline for Stroke RCP guidelines for acute ischaemic stroke
RBWH ICU Journal Club February 2018 Adam Simpson
 RBWH ICU Journal Club February 2018 Adam Simpson 3 THROMBOLYSIS Reperfusion therapy has become the mainstay of therapy for ischaemic stroke. Thrombolysis is now well accepted within 4.5 hours. - Improved
RBWH ICU Journal Club February 2018 Adam Simpson 3 THROMBOLYSIS Reperfusion therapy has become the mainstay of therapy for ischaemic stroke. Thrombolysis is now well accepted within 4.5 hours. - Improved
DVT PROPHYLAXIS IN HOSPITALIZED MEDICAL PATIENTS SAURABH MAJI SR (PULMONARY,MEDICINE)
 DVT PROPHYLAXIS IN HOSPITALIZED MEDICAL PATIENTS SAURABH MAJI SR (PULMONARY,MEDICINE) Introduction VTE (DVT/PE) is an important complication in hospitalized patients Hospitalization for acute medical illness
DVT PROPHYLAXIS IN HOSPITALIZED MEDICAL PATIENTS SAURABH MAJI SR (PULMONARY,MEDICINE) Introduction VTE (DVT/PE) is an important complication in hospitalized patients Hospitalization for acute medical illness
Articles. Introduction Each year, about 22 million people have a stroke worldwide, 1,2 of whom 4 million reside in high-income
 The benefits and harms of intravenous thrombolysis with recombinant tissue plasminogen activator within 6 h of acute ischaemic stroke (the third international stroke trial []): a randomised controlled
The benefits and harms of intravenous thrombolysis with recombinant tissue plasminogen activator within 6 h of acute ischaemic stroke (the third international stroke trial []): a randomised controlled
HORMONE REPLACEMENT THERAPY
 TRIALS OF HR RUTH (Barrett- Connor et al 29 ) JULY 2006 (Country) mean ± sd, range International trial 67.5 an Placebo component in 67.5 ± 6.7 women with Raloxifene or multiple 67.5 ± 6.6 risk factors
TRIALS OF HR RUTH (Barrett- Connor et al 29 ) JULY 2006 (Country) mean ± sd, range International trial 67.5 an Placebo component in 67.5 ± 6.7 women with Raloxifene or multiple 67.5 ± 6.6 risk factors
Critical Appraisal. Dave Abbott Senior Medicines Information Pharmacist
 Critical Appraisal Dave Abbott Senior Medicines Information Pharmacist Aims Identify key components of clinical trial design and apply these to a critical appraisal of the literature Be able to work out
Critical Appraisal Dave Abbott Senior Medicines Information Pharmacist Aims Identify key components of clinical trial design and apply these to a critical appraisal of the literature Be able to work out
The tpa Cage Match. Disclosures. Cage Match. Cage Match 1/27/2014. January 8, Advisory Boards
 The tpa Cage Match January 8, 2014 Andy Jagoda, MD, FACEP Professor and Chair of Emergency Medicine Mount Sinai School of Medicine New York, New York Advisory Boards Disclosures Astra Zeneca, Pfizer, CSL
The tpa Cage Match January 8, 2014 Andy Jagoda, MD, FACEP Professor and Chair of Emergency Medicine Mount Sinai School of Medicine New York, New York Advisory Boards Disclosures Astra Zeneca, Pfizer, CSL
a. Ischemic stroke An acute focal infarction of the brain or retina (and does not include anterior ischemic optic neuropathy (AION)).
 12.0 Outcomes 12.1 Definitions 12.1.1 Neurologic Outcome Events a. Ischemic stroke An acute focal infarction of the brain or retina (and does not include anterior ischemic optic neuropathy (AION)). Criteria:
12.0 Outcomes 12.1 Definitions 12.1.1 Neurologic Outcome Events a. Ischemic stroke An acute focal infarction of the brain or retina (and does not include anterior ischemic optic neuropathy (AION)). Criteria:
The spectrum of clinical outcome of PE
 Practical treatment approach for patients with PE Cecilia Becattini University of Perugia The spectrum of clinical presentation of PE PE-related shock Mild clinical symptoms The spectrum of clinical outcome
Practical treatment approach for patients with PE Cecilia Becattini University of Perugia The spectrum of clinical presentation of PE PE-related shock Mild clinical symptoms The spectrum of clinical outcome
Gastrointestinal haemorrhage
 Gastrointestinal haemorrhage A common emergency Important cause of mortality and morbidity Case fatality is high (10 20% in the UK) Rockall TA et al. BMJ, 1995. 311(6999): p. 222-6. Williams JG et al.
Gastrointestinal haemorrhage A common emergency Important cause of mortality and morbidity Case fatality is high (10 20% in the UK) Rockall TA et al. BMJ, 1995. 311(6999): p. 222-6. Williams JG et al.
Content 1. Relevance 2. Principles 3. Manangement
 Intracranial haemorrhage and anticoagulation Department of Neurology,, Germany Department of Neurology, Heidelberg University Hospital, Germany Department of Clinical Medicine Copenhagen University, Denmark
Intracranial haemorrhage and anticoagulation Department of Neurology,, Germany Department of Neurology, Heidelberg University Hospital, Germany Department of Clinical Medicine Copenhagen University, Denmark
Transfusion Requirements and Management in Trauma RACHEL JACK
 Transfusion Requirements and Management in Trauma RACHEL JACK Overview Haemostatic resuscitation Massive Transfusion Protocol Overview of NBA research guidelines Haemostatic resuscitation Permissive hypotension
Transfusion Requirements and Management in Trauma RACHEL JACK Overview Haemostatic resuscitation Massive Transfusion Protocol Overview of NBA research guidelines Haemostatic resuscitation Permissive hypotension
The current place of tranexamic acid in the management of bleeding
 Anaesthesia 2015, 70 (Suppl. 1), 50 53 Review Article doi:10.1111/anae.12910 The current place of tranexamic acid in the management of B. J. Hunt 1,2 1 Professor of Thrombosis & Haemostasis, Kings College
Anaesthesia 2015, 70 (Suppl. 1), 50 53 Review Article doi:10.1111/anae.12910 The current place of tranexamic acid in the management of B. J. Hunt 1,2 1 Professor of Thrombosis & Haemostasis, Kings College
Stroke: What did we learn in the last year?
 Stroke: What did we learn in the last year? J. Claude Hemphill III, MD, MAS Associate Professor of Clinical Neurology and Neurological Surgery University of California, San Francisco Director, Neurocritical
Stroke: What did we learn in the last year? J. Claude Hemphill III, MD, MAS Associate Professor of Clinical Neurology and Neurological Surgery University of California, San Francisco Director, Neurocritical
Scottish Stroke Improvement Plan. Prof Martin Dennis Chair of National Advisory Committee
 Scottish Stroke Improvement Plan Prof Martin Dennis Chair of National Advisory Committee Examples where Stroke Nurses are key to improvement Implementation of Intermittent Pneumatic Compression to reduce
Scottish Stroke Improvement Plan Prof Martin Dennis Chair of National Advisory Committee Examples where Stroke Nurses are key to improvement Implementation of Intermittent Pneumatic Compression to reduce
Session 7: The Sliding Dichotomy 7.1 Background 7.2 Principles 7.3 Hypothetical example 7.4 Implementation 7.5 Example: CRASH Trial
 Session 7: The Sliding Dichotomy 7.1 Background 7.2 Principles 7.3 Hypothetical example 7.4 Implementation 7.5 Example: CRASH Trial Session 7 1 7.1 The Sliding Dichotomy: Background The sliding dichotomy
Session 7: The Sliding Dichotomy 7.1 Background 7.2 Principles 7.3 Hypothetical example 7.4 Implementation 7.5 Example: CRASH Trial Session 7 1 7.1 The Sliding Dichotomy: Background The sliding dichotomy
When and How to Use the Newly Approved Oral Anticoagulants to Treat Acute Deep Vein Thrombosis (DVT) and Pulmonary Embolism (PE) Ian del Conde, MD
 When and How to Use the Newly Approved Oral Anticoagulants to Treat Acute Deep Vein Thrombosis (DVT) and Pulmonary Embolism (PE) Ian del Conde, MD December 12, 2015 Disclosures CONSULTANT Merck; New Haven
When and How to Use the Newly Approved Oral Anticoagulants to Treat Acute Deep Vein Thrombosis (DVT) and Pulmonary Embolism (PE) Ian del Conde, MD December 12, 2015 Disclosures CONSULTANT Merck; New Haven
ENCHANTED Era: Is it time to rethink treatment of acute ischemic stroke? Kristin J. Scherber, PharmD, BCPS Emergency Medicine Clinical Pharmacist
 ENCHANTED Era: Is it time to rethink treatment of acute ischemic stroke? Kristin J. Scherber, PharmD, BCPS Emergency Medicine Clinical Pharmacist Pharmacy Grand Rounds 26 July 2016 2015 MFMER slide-1 Learning
ENCHANTED Era: Is it time to rethink treatment of acute ischemic stroke? Kristin J. Scherber, PharmD, BCPS Emergency Medicine Clinical Pharmacist Pharmacy Grand Rounds 26 July 2016 2015 MFMER slide-1 Learning
New Study Presented at American Heart Association (AHA) Scientific Sessions 2016:
 Investor News Not intended for U.S. and UK Media Bayer AG Investor Relations 51368 Leverkusen Germany www.investor.bayer.com New Study Presented at American Heart Association (AHA) Scientific Sessions
Investor News Not intended for U.S. and UK Media Bayer AG Investor Relations 51368 Leverkusen Germany www.investor.bayer.com New Study Presented at American Heart Association (AHA) Scientific Sessions
Simon Everett. Consultant Gastroenterologist, SJUH, Leeds. if this is what greets you in the morning, you probably need to go see a doctor
 Simon Everett Consultant Gastroenterologist, SJUH, Leeds if this is what greets you in the morning, you probably need to go see a doctor Presentation Audit data and mortality NICE guidance Risk assessment
Simon Everett Consultant Gastroenterologist, SJUH, Leeds if this is what greets you in the morning, you probably need to go see a doctor Presentation Audit data and mortality NICE guidance Risk assessment
Alteplase for the treatment of acute ischaemic stroke (review of technology appraisal 122)
 Alteplase for the treatment of acute ischaemic stroke (review of technology appraisal 122) Produced by School of Health and Related Research (ScHARR), The University of Sheffield Authors Sarah Davis, ScHARR,
Alteplase for the treatment of acute ischaemic stroke (review of technology appraisal 122) Produced by School of Health and Related Research (ScHARR), The University of Sheffield Authors Sarah Davis, ScHARR,
Professor DA Fitzmaurice Primary Care Clinical Sciences University of Birmingham
 New Guidelines for SPAF Professor DA Fitzmaurice Primary Care Clinical Sciences University of Birmingham Stroke prevention and atrial fibrillation Epidemiology of atrial fibrillation How common is it?
New Guidelines for SPAF Professor DA Fitzmaurice Primary Care Clinical Sciences University of Birmingham Stroke prevention and atrial fibrillation Epidemiology of atrial fibrillation How common is it?
Diagnosis and Treatment of Deep Venous Thrombosis and Pulmonary Embolism
 Agency for Healthcare Research and Quality Evidence Report/Technology Assessment Diagnosis and Treatment of Deep Venous Thrombosis and Pulmonary Embolism Summary Number 68 Overview Venous thromboembolism
Agency for Healthcare Research and Quality Evidence Report/Technology Assessment Diagnosis and Treatment of Deep Venous Thrombosis and Pulmonary Embolism Summary Number 68 Overview Venous thromboembolism
VTE Risk Assessment. Challenges of Hemostasis in Cancer Patients. Cihan Ay, MD Associate Professor
 Challenges of Hemostasis in Cancer Patients VTE Risk Assessment Cihan Ay, MD Associate Professor Clinical Division of Haematology and Haemostaseology Department of Medicine I, Comprehensive Cancer Center
Challenges of Hemostasis in Cancer Patients VTE Risk Assessment Cihan Ay, MD Associate Professor Clinical Division of Haematology and Haemostaseology Department of Medicine I, Comprehensive Cancer Center
Blood Pressure Management in Acute Ischemic Stroke
 Blood Pressure Management in Acute Ischemic Stroke Kimberly Clark, PharmD, BCCCP Clinical Pharmacy Specialist Critical Care, Greenville Health System Adjunct Assistant Professor, South Carolina College
Blood Pressure Management in Acute Ischemic Stroke Kimberly Clark, PharmD, BCCCP Clinical Pharmacy Specialist Critical Care, Greenville Health System Adjunct Assistant Professor, South Carolina College
TITLE: Tranexamic Acid for the Management of Bleeding: A Review of the Clinical Effectiveness and Guidelines
 TITLE: Tranexamic Acid for the Management of Bleeding: A Review of the Clinical Effectiveness and Guidelines DATE: 24 June 2013 CONTEXT AND POLICY ISSUES Bleeding or hemorrhage indicates a blood loss from
TITLE: Tranexamic Acid for the Management of Bleeding: A Review of the Clinical Effectiveness and Guidelines DATE: 24 June 2013 CONTEXT AND POLICY ISSUES Bleeding or hemorrhage indicates a blood loss from
