b-nerve growth factor is a major component of alpaca seminal plasma and induces ovulation in female alpacas
|
|
|
- Austin Sherman Nichols
- 5 years ago
- Views:
Transcription
1 CSIRO PUBLISHING Reproduction, Fertility and Development, 2012, 24, b-nerve growth factor is a major component of alpaca seminal plasma and induces ovulation in female alpacas C. M. Kershaw-Young A,D, X. Druart B, J. Vaughan C and W. M. C. Maxwell A A Faculty of Veterinary Science, The University of Sydney, Camperdown, NSW 2006, Australia. B UMR 6175, Inra-Cnrs-Université de Tours-Haras Nationaux, 37380, Nouzilly, France. C Cria Genesis, PO Box 406, Ocean Grove, Vic. 3226, Australia. D Corresponding author. claire.kershawyoung@yahoo.co.uk Abstract. Ovulation in camelids is induced by an unidentified protein in the seminal plasma of the male termed ovulation-inducing factor. This protein has been reported to be a 14-kDa protein under reducing conditions, which, when purified from seminal plasma, induces ovulation in llamas. The identification of this protein and investigation of its potential to induce ovulation in camelids may aid the development of protocols for the induction of ovulation. In the present study, alpaca seminal plasma proteins were separated using one-dimensional sodium dodecyl sulfate polyacrylamide gel electrophoresis and the most abundant protein of 14 kda was identified as b-nerve growth factor (b-ngf) by liquid chromatography mass spectrometry. Female alpacas (n ¼ 5 per group) were given intramuscular injections of: (1) 1 ml of 0.9% saline; (2) 4 mg buserelin, a gonadotrophin-releasing hormone agonist; (3) 2 ml alpaca seminal plasma; or (4) 1 mg human b-ngf. Ovulation was detected by transrectal ultrasonography 8 days after treatment and confirmed by plasma progesterone concentrations. Ovulation occurred in 0%, 80%, 80% and 80% of animals treated with saline, buserelin, seminal plasma and b-ngf, respectively. Treatment type did not affect the diameter of the corpus luteum, but plasma progesterone concentrations were lower in saline-treated animals than in the other treatment groups owing to the lack of a corpus luteum. The present study is the first to identify the ovulation-inducing factor protein in alpacas. b-ngf successfully induces ovulation in alpacas and this finding may lead to new methods for the induction of ovulation in camelids. Additional keywords: camelid, ovulation inducing factor. Received 14 February 2012, accepted 10 March 2012, published online 24 April 2012 Introduction The use of assisted reproductive technologies such as AI is hindered in camelids by a lack of understanding of ovulation induction, the optimal time of AI and the optimal concentration of spermatozoa required for successful fertilisation. In natural mating, camelids are induced to ovulate after copulation. Consequently, for AI it is necessary to induce ovulation before semen deposition. The current method for the induction of ovulation in camelids involves transrectal ultrasonography of the ovaries to detect a dominant follicle of suitable size, followed by administration of the gonadotrophin-releasing hormone (GnRH) analogue buserelin. In camels, this requires a follicle of mm diameter with 20 mg buserelin administered intravenously (Skidmore 2011). In alpacas, 4 8 mg buserelin administered intramuscularly (i.m.) is sufficient to ovulate follicles ranging in size from 6 to 10 mm (Vaughan et al. 2003). Semen is generally deposited h after the induction of ovulation (Bravo et al. 2000). Although buserelin is successful in inducing ovulation, ultrasonography is time consuming, requires technical ability and is difficult in llamas and alpacas owing to the restricted size of the rectum. Moreover, as Journal compilation Ó CSIRO 2012 farming practices move to a clean, green and ethical approach to animal husbandry, there are requirements for a reduction in the use of synthetic hormones for controlled breeding programs. In camelids, ovulation is induced by a factor present in the seminal plasma of the male, termed ovulation-inducing factor. Intrauterine administration of seminal plasma induced ovulation in 87% of camels (Chen et al. 1985) and 41% of alpacas (Ratto et al. 2005), whereas i.m. administration of seminal plasma induced ovulation in 93% and 90% 100% of alpacas and llamas, respectively (Adams et al. 2005; Ratto et al. 2005). Ovulation-inducing factor has been characterised as a protein that is different from GnRH, LH, human chorionic gonadotrophin, pregnant mare s serum gonadotropin and prostaglandin F 2a (Paolicchi et al. 1999; Pan et al. 2001). Although initial observations indicated that the ovulation-inducing factor protein was.30 kda in size (Ratto et al. 2010), recently a protein of 14 kda identified under reducing conditions has been purified from llama seminal plasma and used to induce ovulation in 90% of llamas (Ratto et al. 2011; Tanco et al. 2011). The presence of an ovulation-inducing factor in other species, including those that are spontaneous and induced
2 1094 Reproduction, Fertility and Development C. M. Kershaw-Young et al. ovulators, has also been investigated. The seminal plasma of the bull (Ratto et al. 2006), horse, pig (Bogle et al. 2011) and rabbit (Silva et al. 2011) induced ovulation in 26%, 29%, 18% and 100% of llamas, respectively. These findings suggest that the ovulation-inducing factor protein is present in the seminal plasma of many livestock species, but that it is more abundant or most potent in induced ovulators, such as the rabbit. Despite comprehensive research describing the effect of seminal plasma or purified ovulation-inducing factor on ovulation, the protein remains uncharacterised. Therefore, the identification of the protein and determination of its ability to induce ovulation may provide new methods for the induction of ovulation in camelids and have wider application in other induced ovulators as well as on spontaneously ovulating species. This could reduce the need for synthetic hormones in controlled breeding programs, thereby meeting consumer demand for cleaner and greener approaches to farming. In addition, the use of the protein, as opposed to buserelin, may improve ovulation rates and better synchronise the timing of ovulation, thereby improving pregnancy rates after AI in camelids. In the present study a major protein component of alpaca seminal plasma was identified and its ability to induce ovulation in female alpacas investigated. Materials and methods Two experiments were performed under authorisation from the University of Sydney Animal Ethics Committee. Experiment 1: identification of protein This experiment was performed between September 2009 and May 2010 using six male alpacas.3 years of age with a body condition score $3 (out of 5), weighing.70 kg and with testes.3 cm in length. By 3 years of age, all male alpacas lose their preputial adhesions and the size of the testes reaches a maximum, such that this is the recommended age for breeding (Tibary and Vaughan 2006). Seminal plasma Semen was collected from the male alpacas (three samples per male) using an artificial vagina fitted inside a mannequin, as described previously (Morton et al. 2009). Within 3 min of collection, the ejaculates were centrifuged at g for 30 min, the seminal plasma decanted and the sperm pellet discarded. The seminal plasma was then centrifuged again at g for 30 min to ensure all spermatozoa were removed. The twice-centrifuged seminal plasma was stored at 808C until further analysis. One-dimensional sodium dodecyl sulfate polyacrylamide gel electrophoresis and mass spectrometry Seminal plasma samples from all male alpacas were pooled and the protein concentration was determined using the Pierce BCA protein quantification assay (Pierce, Rockford, IL, USA) according to the manufacturer s instructions. Seminal plasma protein (50 mg) was reduced in Laemmli buffer (62.5 mm TRIS- HCl, ph 6.8 (Sigma-Aldrich, St Louis, MO, USA), 10% (v/v) glycerol (Sigma), 2% (v/v) sodium dodecyl sulfate (SDS; Sigma), 5% b-mercaptoethanol (Sigma) and 0.2% bromophenol blue at 1008C for 15 min and separated on a mini protean TGX 4 20% precast SDS polyacrylamide gel electrophoresis (PAGE) gel (BioRad, Hercules, CA, USA) for 90 min at 125 V. The gel was stained with Coomassie blue and the proteins visualised on a GS-800 scanner (BioRad). The highly abundant 14 kda protein was excised from the gel, destained with 50 mm ammonium bicarbonate in 40% acetonitrile, dried, rehydrated with 15 ml modified sequencing grade trypsin (12 ng ml 1 ; Promega, Sydney, NSW, Australia) at 48C for 1 h, then incubated in 20 ml of 50 mm ammonium bicarbonate overnight at 378C. The sample was analysed using reverse-phase liquid chromatography (LC) tandem mass spectrometry (MS/MS) on a Q-STAR Elite mass spectrometer (Applied Biosystems, Foster City, CA, USA). The peptides were separated on an Agilent 1100 HPLC system (Mulgrave, Vic., Australia) using a 30 min gradient of acetonitrile (5% 70% in 0.1% formic acid) and the eluted peptides were analysed with Analyst QS 1.1 software (Applied Biosystems). The LC MS/MS data were analysed with ProteinPilot 3.0 software (Applied Biosystems) using the uniprot-taxonomy-mammalia SwissProt database ( accessed February 2011). Only proteins with 95% confidence and at least two unique peptides were accepted. Matches to keratin and porcine trypsin and were automatically excluded. Experiment 2: induction of ovulation Animals This study was performed from October to November 2011 using 20 female alpacas. The female alpacas were housed in pens in groups of five with pasture hay and water provided ad libitum. All female alpacas had previously had at least one successful pregnancy, were.4 years of age (mean months) and were reproductively active. Treatments Each female alpaca was injected with 200 mg, i.m., cloprostenol (250 mgml 1 ; Estrumate; Intervet Australia, Bendigo East, Vic., Australia) to remove any corpora lutea (CL) from the ovaries, followed by 4 mg, i.m., buserelin (4 mgml 1 ; Receptal; Intervet Australia) 24 h later to induce ovulation and synchronise the emergence of the next ovarian follicular wave. Luteolysis was induced 11 days after the induction of ovulation using 200 mg, i.m., cloprostenol. To determine the presence of a dominant follicle, female alpacas were restrained in sternal recumbency and ovaries were examined by transrectal ultrasonography using a MyLab Five Vet scanner with a 7.5-MHz linear array transducer (Esaote Group, Genova, Italy). Female alpacas with a newly emerged dominant follicle of 6 10 mm in diameter were randomly assigned to receive one of four treatments: (1) 1 ml of 0.9% saline injected i.m. (n ¼ 5); (2) 4 mg, i.m., buserelin (n ¼ 5); (3) 2 ml seminal plasma injected i.m., collected as described above for Experiment 1 (n ¼ 5); or (4) 1 mg, i.m., recombinant active human b-nerve growth factor (b-ngf) protein suspended in 0.9% saline (Sino Biological Inc., Beijing, China; n ¼ 5).
3 b-ngf induces ovulation in alpacas Reproduction, Fertility and Development β-ngf 14 kda Fig. 1. One-dimensional sodium dodecyl sulfate polyacrylamide gel electrophoresis of alpaca seminal plasma. A total of 50 mg protein was separated on a 4% 20% acrylamide gel under reducing conditions. The arrow indicates the highly abundant kDa protein. b-ngf, b-nerve growth factor. Each female alpaca was examined by transrectal ultrasonography h after treatment to detect ovulation. Ovulation was defined as the absence of the dominant follicle that was observed during the previous scan at the time of treatment. Ultrasonography was also repeated 8 days after treatment to confirm ovulation and to determine the size of the CL. Blood samples and hormone analysis Jugular venous blood samples (5 ml) were collected on Day 8 at the time of ultrasonography for determination of plasma progesterone. Samples were placed in heparinised tubes, centrifuged at 2000g for 10 min and the plasma decanted and stored at 208C until analysis. Plasma progesterone concentrations were determined using a commercially available double-antibody radioimmunoassay kit (Coat-a-Count Progesterone kit; Diagnostic Products, Los Angeles, CA, USA) previously validated in alpacas (Sumar et al. 1988; Adams et al. 2005). The sensitivity of the assay was 0.02 ng ml 1 and the intraassay CV was 9.7%. Statistical analysis Data were analysed using ANOVA in GENSTAT ver. 11 (VSN International, Hemel Hempstead, UK) with post hoc comparisons using the least significant difference (l.s.d.) test where appropriate. For all analyses P, 0.05 was considered significant. To determine differences in plasma progesterone concentrations between treatments, animals in the buserelin, seminal plasma and b-ngf treatment groups that did not ovulate were excluded from the analysis. Results One-dimensional SDS-PAGE of alpaca seminal plasma identified a highly abundant protein of approximately kda (Fig. 1). Using a protein cut-off of 95% confidence and at least two unique peptides, mass spectrometry identified one protein in the 14-kDa protein band: b-ngf precursor (Table 1). When scanned for the presence or absence of the dominant follicle h after treatment with buserelin, seminal plasma and b-ngf, the dominant follicle was absent in four of five, three of five and four of five female alpacas, respectively. However, in saline-treated alpacas, the dominant follicle was present in all five animals, indicating that ovulation had not occurred. On Day 8 following treatment, CL were detected in none, four, four and four (of five) female alpacas treated with saline, buserelin, seminal plasma and b-ngf, respectively. The size of the dominant follicle on the day of treatment (P ¼ 0.904) and of the CL on Day 8 after treatment (P ¼ 0.818) did not differ between treatment groups (Table 2). Plasma progesterone concentrations differed significantly between treatment groups (P ¼ 0.003) and were higher in alpacas treated with buserelin, seminal plasma and b-ngf compared with those treated with saline (Table 2). Discussion The present study is the first to identify the ovulation-inducing factor protein in alpacas as b-ngf. This protein was extremely abundant in alpaca seminal plasma and induced ovulation in four of five (80%) female alpacas following the administration of 1 mg, i.m. b-nerve growth factor was the most abundant protein in alpaca seminal plasma. The total protein content of alpaca seminal plasma is 40 mg ml 1 (Garnica et al. 1993) and the average ejaculate in alpacas and llamas is 1 2 ml (Lichtenwalner et al. 1996; Bravo et al. 1997; Vaughan et al. 2003). In llamas, each ejaculate contains approximately 12 mg ovulationinducing factor protein (Tanco et al. 2011); consequently, b-ngf contributes to 15% 30% of total protein in camelid seminal plasma. Furthermore, b-ngf is a 27-kDa homodimer that is reduced to two dimers of approximately 14 kda under reducing conditions, such as SDS-PAGE. Supporting our hypothesis that the ovulation-inducing factor in camelids is b-ngf, and in agreement with the present study, llama seminal plasma proteins of $30 kda induced ovulation in nine of nine llamas when administered i.m. (Ratto et al. 2010). Although a 14-kDa protein has also been identified as ovulation-inducing factor (Ratto et al. 2011), the protein size was determined under reducing conditions (as in the present study). Consequently, it is likely that the identified 14-kDa protein is the b-ngf homodimer. Dissen et al. (1996b) implicated NGF in the control of ovarian function, and its receptor trka is present in the follicle of the rat (Dissen et al. 1996a). Furthermore, NGF acts through this receptor on human granulosa cells to stimulate the expression of FSH receptors and the secretion of oestradiol (Salas et al. 2006). Although these findings suggest a direct effect of b-ngf on the ovary and follicle to induce ovulation, in other studies camelid seminal plasma (Adams et al. 2005) and purified ovulation-inducing factor (Tanco et al. 2011) initiate an LH surge within 8 h of treatment. Alpaca seminal plasma has been reported to induce the release of LH from mice pituitary cells in a dose-dependent manner and the use of an anti-gnrh antibody did not hinder this response (Paolicchi et al. 1999). Consequently, it is likely that the mode of action of seminal plasma b-ngf is at
4 1096 Reproduction, Fertility and Development C. M. Kershaw-Young et al. Table 1. Proteins identified in alpaca seminal plasma by liquid chromatography tandem mass spectrometry of the 13]14-kDa band A search of SwissProt database ( accessed February 2011) was performed using ProteinPilot software (Applied Biosystems, Foster City, CA, USA). Only proteins with 95% confidence and at least two unique peptides were accepted Uniprot accession no. Name Peptide sequences P13600 b-nerve growth factor precursor GKEVMVLGEVNINNSVFKQYFFETK ALTMDGKQAAWR HWNSYCTTTHTFVK IRIDTACVCVLSRKTGQR LDTALRRARSAP GEFSVCDSISVWVGDK Table 2. Follicle diameter before treatment, and corpus luteum diameter and plasma progesterone concentrations on Day 8 after treatment in female alpacas injected i.m. with 1 ml of 0.9% saline, 4 lg buserelin, 2 ml alpaca seminal plasma or 1 mg human b-nerve growth factor in 1 ml 0.9% saline Data are the mean s.e.m. Values within a column with different superscript letters differ significantly (P, 0.05). Mean values for plasma progesterone concentrations do not include data from animals that did not ovulate, except in the case of saline-treated animals. CL, corpus luteum; b-ngf, b-nerve growth factor Treatment group Follicle diameter (mm) CL diameter (mm) Progesterone (ng ml 1 ) Saline a None present a Buserelin a a b Seminal plasma a a b b-ngf a a b the level of the hypothalamo pituitary axis, inducing ovulation by stimulating the secretion of LH. This endocrine effect of b- NGF may explain why the proportion of llamas ovulating is higher when seminal plasma is administered i.m. than when it is deposited in the uterus (Ratto et al. 2005). In the present study, the proportion of alpacas ovulating did not differ between the groups treated with seminal plasma, buserelin and b-ngf. This is in agreement with other studies in which seminal plasma, GnRH agonists and LH have been reported to induce ovulation with equal success (Chen et al. 1985; Adams et al. 2005). Moreover, the type of treatment did not affect the size of the CL 8 days after treatment in the present study, suggesting that all treatments initiated a similar ovulatory effect. This is in contrast with previous findings in which the diameter of the CL and the concentration of plasma progesterone were higher in llamas treated with seminal plasma compared with GnRH (Adams et al. 2005). This apparent discrepancy may be due to the different GnRH agonists and concentrations used in the present study compared with the study of Adams et al. (2005). Ovulation-inducing factor purified from llama seminal plasma induces ovulation in llamas in a dose-dependant manner, whereby higher concentrations of ovulation-inducing factor increase the proportion of female llamas ovulating, the diameter of CL and the concentrations of plasma progesterone (Tanco et al. 2011). It is possible that the concentration of b-ngf in the alpaca seminal plasma used in the present study was lower than that occurring naturally in the llama seminal plasma used in the study of Tanco et al. (2011). The lower plasma progesterone concentrations in saline-treated animals compared with other treatment groups in the present study was expected because of the absence of a CL in saline-treated alpacas. The source of seminal plasma b-ngf in camelids is unknown. It has been reported that b-ngf mrna is expressed predominantly in the vas deferens of the mouse and rat reproductive tract (MacGrogan et al. 1991), whereas in the guinea-pig (Harper et al. 1979; MacGrogan et al. 1991) and bull and rabbit (Harper and Theonen 1980) NGF is mostly expressed in the prostate. Both b-ngf protein and mrna are expressed in the testis of the alpaca and are thought to be involved in spermatogenesis (Wang et al. 2011), although it is not known whether b-ngf from the testes is secreted in the seminal plasma. In Bactrian camels, intrauterine administration of seminal plasma from a vasectomised male induced ovulation in 100% of females (Chen et al. 1985), suggesting that ovulation-inducing factor is derived from the accessory sex glands and not the testes. Therefore, it is likely that seminal plasma b-ngf in camelids is secreted by the prostate, although this requires further investigation. Although the presence of b-ngf has not been reported in camelid seminal plasma, or identified elsewhere as ovulationinducing factor, NGF has been purified from bovine seminal plasma as a protein of approximately 15 kda under reducing conditions (Harper et al. 1982). Concentrations of NGF in bovine seminal plasma are approximately 0.7 mg ml 1 semen, much higher than in sheep, goat, human and pig seminal plasma (Harper et al. 1982). In contrast, llama seminal plasma contains approximately 12 mg ovulation-inducing factor per ejaculate (Tanco et al. 2011). Intramuscular administration of bovine seminal plasma has been reported to induce ovulation in 26% of llamas (Ratto et al. 2006) and we propose that this was due to the b-ngf present in bull seminal plasma. The presence of b-ngf in other livestock species has not been reported. However, horse and pig seminal plasma induced ovulation in 29% and 18% of llamas, respectively (Bogle et al. 2011), and rabbit seminal plasma induced ovulation in 100% of llamas (Silva et al. 2011) when administered i.m. These findings imply that b-ngf is relatively well conserved among species and that seminal plasma b-ngf may be active in spontaneously ovulating species.
5 b-ngf induces ovulation in alpacas Reproduction, Fertility and Development 1097 In conclusion, the findings of the present study suggest that the previously unidentified ovulation-inducing factor in camelid seminal plasma is b-ngf. The ability of b-ngf to induce ovulation in alpacas is apparent and equally effective compared with the GnRH agonist buserelin and seminal plasma. Thus, b-ngf may provide an alternative mechanism for the induction of ovulation in alpacas, reduce the need for synthetic hormones and possibly improve fertility in combination with AI. The ability of b-ngf to induce ovulation in other livestock species warrants investigation, especially because NGF is present in the seminal plasma of non-camelid species and the seminal plasma from these species can induce ovulation in camelids. Acknowledgements The authors thank Julienne Gelber (Bumble Hill Alpaca Stud, Orange, NSW, Australia) for provision of animals, facilities and generous hospitality, Kim Heasman for progesterone analysis, Dr Svetlana Mactier and Phillipa Kohnke for assistance with mass spectrometry and Byron Biffin and Keith Tribe for maintenance of the male alpacas. This work was funded by the Rural Industries Research and Development Corporation of Australia. References Adams, G. P., Ratto, M. H., Huanca, W., and Singh, J. (2005). Ovulationinducing factor in the seminal plasma of alpacas and llamas. Biol. Reprod. 73, doi: /biolreprod Bogle, O. A., Ratto, M. H., and Adams, G. P. (2011). Evidence for the conservation of biological activity of ovulation-inducing factor in seminal plasma. Reproduction 142, doi: /rep Bravo, P. W., Flores, U., Garnica, J., and Ordonez, C. (1997). Collection of semen and artificial insemination of alpacas. Theriogenology 47, doi: /s x(97) Bravo, P. W., Skidmore, J. A., and Zhao, X. X. (2000). Reproductive aspects and storage of semen in camelidae. Anim. Reprod. Sci. 62, doi: /s (00) Chen, B. X., Yuen, Z. X., and Pan, G. W. (1985). Semen-induced ovulation in the bactrian camel (Camelus bactrianus). J. Reprod. Fertil. 74, doi: /jrf Dissen, G. A., Hill, D. F., Costa, M. E., Dees, W. L., Lara, H. E., and Ojeda, S. R. (1996a). A role for trka nerve growth factor receptors in mammalian ovulation. Endocrinology 137, doi: / EN Dissen, G. A., Mayerhofer, A., and Ojeda, S. R. (1996b). Participation of nerve growth factor in the regulation of ovarian function. Zygote 4, doi: /s Garnica, J., Achata, R., and Bravo, P. W. (1993). Physical and biochemical characteristics of alpaca semen. Anim. Reprod. Sci. 32, doi: / (93)90059-z Harper, G. P., and Theonen, H. (1980). The distribution of nerve growth factor in the male sex organs of mammals. J. Neurochem. 34, doi: /j tb09663.x Harper, G. P., Barde, Y. A., Burnstock, G., Carstairs, J. R., Dennison, M. E., Suda, K., and Vernon, C. A. (1979). Guinea pig prostate is a rich source of nerve growth factor. Nature 279, doi: /279160a0 Harper, G. P., Glanville, R. W., and Thoenen, H. (1982). The purification of nerve growth factor from bovine seminal plasma. Biochemical characterization and partial amino acid sequence. J. Biol. Chem. 257, Lichtenwalner, A. B., Woods, G. L., and Weber, J. A. (1996). Seminal collection, seminal charateristics and pattern of ejaculation in llamas. Theriogenology 46, doi: / x(96) MacGrogan, D., Despres, G., Romand, R., and Dicou, E. (1991). Expression of the b-nerve growth factor gene in male sex organs of the mouse, rat, and guinea pig. J. Neurosci. Res. 28, doi: /jnr Morton, K. M., Gibb, Z., Bertoldo, M., and Maxwell, W. M. C. (2009). Effect of diluent, dilution rate and storage temperature on longevity and functional integrity of liquid stored alpaca (Vicugna pacos) semen. J. Camelid Sci. 2, Pan, G., Chen, Z., Liu, X., Li, D., Xie, Q., Ling, F., and Fang, L. (2001). Isolation and purification of the ovulation-inducing factor from seminal plasma in the bactrian camel (Camelus bactrianus). Theriogenology 55, doi: /s x(01) Paolicchi, F., Urquieta, B., Del Valle, L., and Bustos-Obregon, E. (1999). Biological activity of the seminal plasma of alpacas: stimulus for the production of LH by pituitary cells. Anim. Reprod. Sci. 54, doi: /s (98)00150-x Ratto, M. H., Huanca, W., Singh, J., and Adams, G. P. (2005). Local versus systemic effect of ovulation-inducing factor in the seminal plasma of alpacas. Reprod. Biol. Endocrinol. 3, 29. doi: / Ratto, M. H., Huanca, W., Singh, J., and Adams, G. P. (2006). Comparison of the effect of ovulation-inducing factor (OIF) in the seminal plasma of llamas, alpacas, and bulls. Theriogenology 66, doi: / J.THERIOGENOLOGY Ratto, M. H., Huanca, W., and Adams, G. P. (2010). Ovulation-inducing factor: a protein component of llama seminal plasma. Reprod. Biol. Endocrinol. 8, doi: / Ratto, M. H., Delbaere, L. T. J., Leduc, Y. A., Pierson, R. A., and Adams, G. P. (2011). Biochemical isolation and purification of ovulation-inducing factor (OIF) in seminal plasma of llamas. Reprod. Biol. Endocrinol. 9, doi: / Salas, C., Julio-Pieper, M., Valladares, M., Pommer, R., Vega, M., Mastronardi, C., Kerr, B., Ojeda, S. R., Lara, H. E., and Romero, C. (2006). Nerve growth factor-dependent activation of trka receptors in the human ovary results in synthesis of follicle-stimulating hormone receptors and estrogen secretion. J. Clin. Endocrinol. Metab. 91, doi: /jc Silva, M., Nino, A., Guerra, M., Letelier, C., Valderrama, X. P., Adams, G. P., and Ratto, M. H. (2011). Is an ovulation-inducing factor (OIF) present in the seminal plasma of rabbits? Anim. Reprod. Sci. 127, doi: /j.anireprosci Skidmore, J. A. (2011). Reproductive physiology in female Old World camelids. Anim. Reprod. Sci. 124, doi: /j.anire PROSCI Sumar, J., Fredriksson, G., Alarcon, V., Kindahl, H., and Edqvist, L. (1988). Levels of 15-keto-13,14-dihydro-pgf2a, progesterone and estradiol-17- beta after induced ovulations in llamas and alpacas. Acta Vet. Scand. 29, Tanco, V. M., Ratto, M. H., Lazzarotta, M., and Adams, G. P. (2011). Dose response of female llamas to ovulation-inducing factor from seminal plasma. Biol. Reprod. 85, doi: /biolreprod Tibary, J., and Vaughan, J. (2006). Reproductive infertility and physiology in male South American camelids: a review and clinical observations. Small Rumin. Res. 61, doi: /j.smallrumres Vaughan, J., Galloway, D., and Hopkins, D. (2003). Artificial Insemination in Alpacas (Lama pacos). (Rural Industries Research and Development Corporation: Kingston, ACT.) Wang, H., Dong, Y., Chen, W., Hei, J., and Dong, C. (2011). Expression and localization of nerve growth factor (NGF) in the testis of alpaca (Llama pacos). Folia Histochem. Cytobiol. 49,
Semen-induced ovulation in the bactrian camel (Camelus bactrianus)
 Semen-induced ovulation in the bactrian camel (Camelus bactrianus) B. X. Chen, Z. X. Yuen and G. W. Pan Department of Veterinary Medicine, Gansu Agricultural University, Wuwei, Gansu and *Haixi Institute
Semen-induced ovulation in the bactrian camel (Camelus bactrianus) B. X. Chen, Z. X. Yuen and G. W. Pan Department of Veterinary Medicine, Gansu Agricultural University, Wuwei, Gansu and *Haixi Institute
The continued development of artificial insemination technologies in alpacas
 The continued development of artificial insemination technologies in alpacas Katherine M. Morton and W.M. Chis Maxwell 1 1 Centre for Advanced Technologies in Animal Genetics and Reproduction (ReproGen),
The continued development of artificial insemination technologies in alpacas Katherine M. Morton and W.M. Chis Maxwell 1 1 Centre for Advanced Technologies in Animal Genetics and Reproduction (ReproGen),
WBC / ICAR 2008 Satellite Meeting on Camelid Reproduction
 Close window to return to IVIS www.ivis.org WBC / ICAR 2008 Satellite Meeting on Camelid Reproduction 12-13 July, 2008, Budapest, Hungary Program and Extended stracts Peter Nagy; Gyula Huszenicza; Judit
Close window to return to IVIS www.ivis.org WBC / ICAR 2008 Satellite Meeting on Camelid Reproduction 12-13 July, 2008, Budapest, Hungary Program and Extended stracts Peter Nagy; Gyula Huszenicza; Judit
The effect of LH and GnRH analogues on induction of ovulation in Bactrian camel (Camelus bactrianus)
 The effect of LH and GnRH analogues on induction of ovulation in Bactrian camel (Camelus bactrianus) Moghiseh, A. 1 ; Niasari-Naslaji, A. 1* ; Nikjou, D. 1 ; Gerami, A. 2 ; Razavi, K. 3 and Mostafaey,
The effect of LH and GnRH analogues on induction of ovulation in Bactrian camel (Camelus bactrianus) Moghiseh, A. 1 ; Niasari-Naslaji, A. 1* ; Nikjou, D. 1 ; Gerami, A. 2 ; Razavi, K. 3 and Mostafaey,
Concentrations of Luteinizing Hormone and Ovulatory Responses in Dairy Cows Before Timed Artificial Insemination
 Concentrations of Luteinizing Hormone and Ovulatory Responses in Dairy Cows Before Timed Artificial Insemination S. L. Pulley, D. H. Keisler, S. L. Hill, and J. S. Stevenson Summary The objective of this
Concentrations of Luteinizing Hormone and Ovulatory Responses in Dairy Cows Before Timed Artificial Insemination S. L. Pulley, D. H. Keisler, S. L. Hill, and J. S. Stevenson Summary The objective of this
REPRODUCCIÓN. La idea fija. Copyright 2004 Pearson Education, Inc., publishing as Benjamin Cummings
 REPRODUCCIÓN La idea fija How male and female reproductive systems differentiate The reproductive organs and how they work How gametes are produced and fertilized Pregnancy, stages of development, birth
REPRODUCCIÓN La idea fija How male and female reproductive systems differentiate The reproductive organs and how they work How gametes are produced and fertilized Pregnancy, stages of development, birth
Treatment 3 Days After Ovulation In Mares
 Luteal Regression And Follicle Development Following Prostaglandin-F 2α Treatment 3 Days After Ovulation In Mares D.R. Bergfelt a, R.A. Pierson b, and O.J. Ginther a a University of Wisconsin, Madison,
Luteal Regression And Follicle Development Following Prostaglandin-F 2α Treatment 3 Days After Ovulation In Mares D.R. Bergfelt a, R.A. Pierson b, and O.J. Ginther a a University of Wisconsin, Madison,
Outline. Male Reproductive System Testes and Sperm Hormonal Regulation
 Outline Male Reproductive System Testes and Sperm Hormonal Regulation Female Reproductive System Genital Tract Hormonal Levels Uterine Cycle Fertilization and Pregnancy Control of Reproduction Infertility
Outline Male Reproductive System Testes and Sperm Hormonal Regulation Female Reproductive System Genital Tract Hormonal Levels Uterine Cycle Fertilization and Pregnancy Control of Reproduction Infertility
Advanced Non-Cycling Program. Health
 Advanced Non-Cycling Program Health Why Treat Non-Cycling Cows? Treating cows that have not been detected in oestrus ( non-cycling ) prior to the planned start of mating with DIB-Synch provides a return
Advanced Non-Cycling Program Health Why Treat Non-Cycling Cows? Treating cows that have not been detected in oestrus ( non-cycling ) prior to the planned start of mating with DIB-Synch provides a return
Small Ruminant Reproductive Management Workshop
 Small Ruminant Reproductive Management Workshop Animal Nutrition and Physiology Center, North Dakota State University Sponsors: American Sheep and Goat Center, North Dakota State University, University
Small Ruminant Reproductive Management Workshop Animal Nutrition and Physiology Center, North Dakota State University Sponsors: American Sheep and Goat Center, North Dakota State University, University
M. Irfan-ur-Rehman Khan, M. A. Rana and N. Ahmad. Department of Theriogenology, University of Veterinary and Animal Sciences, Lahore, Pakistan
 82 ULTRASONIC MONITORING OF FOLLICLES AND CORPORA LUTEA DURING SYNCHRONIZATION IN SUMMER ANOESTROUS NILI RAVI BUFFALOES AND THEIR SUBSEQUENT SUPEROVULATORY RESPONSE M. Irfan-ur-Rehman Khan, M. A. Rana
82 ULTRASONIC MONITORING OF FOLLICLES AND CORPORA LUTEA DURING SYNCHRONIZATION IN SUMMER ANOESTROUS NILI RAVI BUFFALOES AND THEIR SUBSEQUENT SUPEROVULATORY RESPONSE M. Irfan-ur-Rehman Khan, M. A. Rana
Concentrations of luteinizing hormone and ovulatory responses in dairy cows before timed artificial insemination
 Kansas Agricultural Experiment Station Research Reports Volume 0 Issue Dairy Research (98-0) Article 8 0 Concentrations of luteinizing hormone and ovulatory responses in dairy cows before timed artificial
Kansas Agricultural Experiment Station Research Reports Volume 0 Issue Dairy Research (98-0) Article 8 0 Concentrations of luteinizing hormone and ovulatory responses in dairy cows before timed artificial
Chapter 14 Reproduction Review Assignment
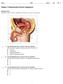 Date: Mark: _/45 Chapter 14 Reproduction Review Assignment Multiple Choice Identify the choice that best completes the statement or answers the question. 1. Use the diagram above to answer the next question.
Date: Mark: _/45 Chapter 14 Reproduction Review Assignment Multiple Choice Identify the choice that best completes the statement or answers the question. 1. Use the diagram above to answer the next question.
Ovarian Characteristics, Serum Hormone Concentrations, and Fertility in Lactating Dairy Cows in Response to Equine Chorionic Gonadotropin
 Ovarian Characteristics, Serum Hormone Concentrations, and Fertility in Lactating Dairy Cows in Response to quine Chorionic Gonadotropin S. L. Pulley, L. D. Wallace, H. I. Mellieon, and J. S. Stevenson
Ovarian Characteristics, Serum Hormone Concentrations, and Fertility in Lactating Dairy Cows in Response to quine Chorionic Gonadotropin S. L. Pulley, L. D. Wallace, H. I. Mellieon, and J. S. Stevenson
Male Reproduction Organs. 1. Testes 2. Epididymis 3. Vas deferens 4. Urethra 5. Penis 6. Prostate 7. Seminal vesicles 8. Bulbourethral glands
 Outline Terminology Human Reproduction Biol 105 Lecture Packet 21 Chapter 17 I. Male Reproduction A. Reproductive organs B. Sperm development II. Female Reproduction A. Reproductive organs B. Egg development
Outline Terminology Human Reproduction Biol 105 Lecture Packet 21 Chapter 17 I. Male Reproduction A. Reproductive organs B. Sperm development II. Female Reproduction A. Reproductive organs B. Egg development
REPRODUCTION & GENETICS. Hormones
 REPRODUCTION & GENETICS Hormones http://www.youtube.com/watch?v=np0wfu_mgzo Objectives 2 Define what hormones are; Compare and contrast the male and female hormones; Explain what each hormone in the mail
REPRODUCTION & GENETICS Hormones http://www.youtube.com/watch?v=np0wfu_mgzo Objectives 2 Define what hormones are; Compare and contrast the male and female hormones; Explain what each hormone in the mail
Female Reproductive System. Lesson 10
 Female Reproductive System Lesson 10 Learning Goals 1. What are the five hormones involved in the female reproductive system? 2. Understand the four phases of the menstrual cycle. Human Reproductive System
Female Reproductive System Lesson 10 Learning Goals 1. What are the five hormones involved in the female reproductive system? 2. Understand the four phases of the menstrual cycle. Human Reproductive System
Buserelin animedica mg/ml Injektionslösung für Rinder, Pferde, Kaninchen
 1. NAME OF THE VETERINARY MEDICINAL PRODUCT Buserelin animedica 0,004 mg/ml Injektionslösung für Rinder, Pferde, Kaninchen (Germany) Buserelin animedica 0.004 mg/ml Injektionslösung für Rinder, Pferde,
1. NAME OF THE VETERINARY MEDICINAL PRODUCT Buserelin animedica 0,004 mg/ml Injektionslösung für Rinder, Pferde, Kaninchen (Germany) Buserelin animedica 0.004 mg/ml Injektionslösung für Rinder, Pferde,
GONADOTROPHIN (LUTEINISING)- RELEASING HORMONE AND ANALOGUES (GnRH OR LHRH)
 GONADOTROPHIN (LUTEINISING)- RELEASING HORMONE AND ANALOGUES (GnRH OR LHRH) Naturally occurring hormone, produced by the hypothalamus and transferred to the anterior pituitary gland in the hypophyseal
GONADOTROPHIN (LUTEINISING)- RELEASING HORMONE AND ANALOGUES (GnRH OR LHRH) Naturally occurring hormone, produced by the hypothalamus and transferred to the anterior pituitary gland in the hypophyseal
Time / days. Explain how the release of FSH is controlled by negative feedback.
 1. The graph shows the changes in concentration of the hormones responsible for controlling the menstrual cycle. A Hormone concentration Oestrogen B C 0 14 28 Time / days WD Phillips and TJ Chilton A Level
1. The graph shows the changes in concentration of the hormones responsible for controlling the menstrual cycle. A Hormone concentration Oestrogen B C 0 14 28 Time / days WD Phillips and TJ Chilton A Level
SISTEMA REPRODUCTOR (LA IDEA FIJA) Copyright 2004 Pearson Education, Inc., publishing as Benjamin Cummings
 SISTEMA REPRODUCTOR (LA IDEA FIJA) How male and female reproductive systems differentiate The reproductive organs and how they work How gametes are produced and fertilized Pregnancy, stages of development,
SISTEMA REPRODUCTOR (LA IDEA FIJA) How male and female reproductive systems differentiate The reproductive organs and how they work How gametes are produced and fertilized Pregnancy, stages of development,
Reproductive Endocrinology. Isabel Hwang Department of Physiology Faculty of Medicine University of Hong Kong Hong Kong May2007
 Reproductive Endocrinology Isabel Hwang Department of Physiology Faculty of Medicine University of Hong Kong Hong Kong May2007 isabelss@hkucc.hku.hk A 3-hormone chain of command controls reproduction with
Reproductive Endocrinology Isabel Hwang Department of Physiology Faculty of Medicine University of Hong Kong Hong Kong May2007 isabelss@hkucc.hku.hk A 3-hormone chain of command controls reproduction with
Animal Reproduction Science
 Animal Reproduction Science 136 (2013) 194 204 Contents lists available at SciVerse ScienceDirect Animal Reproduction Science journa l h omepa g e: www.elsevier.com/locate/anireprosci Factors influencing
Animal Reproduction Science 136 (2013) 194 204 Contents lists available at SciVerse ScienceDirect Animal Reproduction Science journa l h omepa g e: www.elsevier.com/locate/anireprosci Factors influencing
THE EFFECT OF OVULATION-INDUCING FACTOR (OIF) IN BOVINE SEMINAL PLASMA ON OVARIAN FUNCTION IN CATTLE
 THE EFFECT OF OVULATION-INDUCING FACTOR (OIF) IN BOVINE SEMINAL PLASMA ON OVARIAN FUNCTION IN CATTLE A thesis submitted to the College of Graduate Studies and Research, in partial fulfillment of the requirements
THE EFFECT OF OVULATION-INDUCING FACTOR (OIF) IN BOVINE SEMINAL PLASMA ON OVARIAN FUNCTION IN CATTLE A thesis submitted to the College of Graduate Studies and Research, in partial fulfillment of the requirements
10.7 The Reproductive Hormones
 10.7 The Reproductive Hormones December 10, 2013. Website survey?? QUESTION: Who is more complicated: men or women? The Female Reproductive System ovaries: produce gametes (eggs) produce estrogen (steroid
10.7 The Reproductive Hormones December 10, 2013. Website survey?? QUESTION: Who is more complicated: men or women? The Female Reproductive System ovaries: produce gametes (eggs) produce estrogen (steroid
Sow Reproduction and Seasonal Infertility. Darlington Pig Discussion Group 13 th March 2014 Richard Bull
 Sow Reproduction and Seasonal Infertility Darlington Pig Discussion Group 13 th March 2014 Richard Bull Richard Bull Taurus Concepts Ltd Sow Reproduction Endogenous Hormones Gland Hormone Function Hypothalamus
Sow Reproduction and Seasonal Infertility Darlington Pig Discussion Group 13 th March 2014 Richard Bull Richard Bull Taurus Concepts Ltd Sow Reproduction Endogenous Hormones Gland Hormone Function Hypothalamus
Papain and its inhibitor E-64 reduce camelid semen viscosity without impairing sperm function and improve post-thaw motility rates
 Papain and its inhibitor E-64 reduce camelid semen viscosity without impairing sperm function and improve post-thaw motility rates by Kershaw, C.M., Evans, G., Rodney, R. and Maxwell, W.M.C. Copyright,
Papain and its inhibitor E-64 reduce camelid semen viscosity without impairing sperm function and improve post-thaw motility rates by Kershaw, C.M., Evans, G., Rodney, R. and Maxwell, W.M.C. Copyright,
ONTARIO PORK RESEARCH PROPOSAL FINAL REPORT
 PROPOSAL # 04/16 ONTARIO PORK RESEARCH PROPOSAL FINAL REPORT Project Leader: R.N. Kirkwood/ R.M. Friendship Project Title: Effect of addition of seminal plasma to thawed semen on sow fertility Objectives
PROPOSAL # 04/16 ONTARIO PORK RESEARCH PROPOSAL FINAL REPORT Project Leader: R.N. Kirkwood/ R.M. Friendship Project Title: Effect of addition of seminal plasma to thawed semen on sow fertility Objectives
Ovarian Kinetics and Control of Ovulation (17 June 2000)
 In: Recent Advances in Camelid Reproduction, Skidmore J.A. and Adams G.P. (Eds.) Publisher: International Veterinary Information Service (www.ivis.org) Ovarian Kinetics and Control of Ovulation (17 June
In: Recent Advances in Camelid Reproduction, Skidmore J.A. and Adams G.P. (Eds.) Publisher: International Veterinary Information Service (www.ivis.org) Ovarian Kinetics and Control of Ovulation (17 June
Superovulation of Beef Heifers with Follicle Stimulating Hormone or Human Menopausal Gonadotropin: Acute Effects on Hormone Secretion
 Beef Research Report, 1996 Animal Science Research Reports 1997 Superovulation of Beef Heifers with Follicle Stimulating Hormone or Human Menopausal Gonadotropin: Acute Effects on Hormone Secretion Acacia
Beef Research Report, 1996 Animal Science Research Reports 1997 Superovulation of Beef Heifers with Follicle Stimulating Hormone or Human Menopausal Gonadotropin: Acute Effects on Hormone Secretion Acacia
Animal and Veterinary Science Department University of Idaho. REGULATION OF REPRODUCTION AVS 222 (Instructor: Dr. Amin Ahmadzadeh) Chapter 5
 Animal and Veterinary Science Department University of Idaho REGULATION OF REPRODUCTION AVS 222 (Instructor: Dr. Amin Ahmadzadeh) Chapter 5 I. DEFINITIONS A. Endocrine Gland B. Hormone Chemical messenger
Animal and Veterinary Science Department University of Idaho REGULATION OF REPRODUCTION AVS 222 (Instructor: Dr. Amin Ahmadzadeh) Chapter 5 I. DEFINITIONS A. Endocrine Gland B. Hormone Chemical messenger
BIOLOGICAL CHARACTERIZATION OF OVULATION-INDUCING FACTOR (OIF) IN LLAMA SEMINAL PLASMA
 BIOLOGICAL CHARACTERIZATION OF OVULATION-INDUCING FACTOR (OIF) IN LLAMA SEMINAL PLASMA A thesis submitted to the College of Graduate Studies and Research, University of Saskatchewan in partial fulfillment
BIOLOGICAL CHARACTERIZATION OF OVULATION-INDUCING FACTOR (OIF) IN LLAMA SEMINAL PLASMA A thesis submitted to the College of Graduate Studies and Research, University of Saskatchewan in partial fulfillment
Superovulation of Beef Heifers with Follicle Stimulating Hormone or Human Menopausal Gonadotropin: Acute Effects on Hormone Secretion
 Superovulation of Beef Heifers with Follicle Stimulating Hormone or Human Menopausal Gonadotropin: Acute Effects on Hormone Secretion A.S. Leaflet R1362 Acacia A. Alcivar, graduate research assistant,
Superovulation of Beef Heifers with Follicle Stimulating Hormone or Human Menopausal Gonadotropin: Acute Effects on Hormone Secretion A.S. Leaflet R1362 Acacia A. Alcivar, graduate research assistant,
Chapter 28: REPRODUCTIVE SYSTEM: MALE
 Chapter 28: REPRODUCTIVE SYSTEM: MALE I. FUNCTIONAL ANATOMY (Fig. 28.1) A. Testes: glands which produce male gametes, as well as glands producing testosterone 2. Seminiferous tubules (Fig.28.3; 28.5) a.
Chapter 28: REPRODUCTIVE SYSTEM: MALE I. FUNCTIONAL ANATOMY (Fig. 28.1) A. Testes: glands which produce male gametes, as well as glands producing testosterone 2. Seminiferous tubules (Fig.28.3; 28.5) a.
Sample Provincial exam Q s: Reproduction
 Sample Provincial exam Q s: Reproduction 11. Functions Testosterone Makes the male sex organs function normally, and also inhibits hypothalamus s release of GnRH and thus LH & FSH and thus testosterone
Sample Provincial exam Q s: Reproduction 11. Functions Testosterone Makes the male sex organs function normally, and also inhibits hypothalamus s release of GnRH and thus LH & FSH and thus testosterone
OVARIAN RESPONSES AND CONCEPTION RATES IN RESPONSE TO GnRH, hcg, AND PROGESTERONE 1
 Dairy Research 2006 OVARIAN RESPONSES AND CONCEPTION RATES IN RESPONSE TO GnRH, hcg, AND PROGESTERONE 1 J. S. Stevenson, M. A. Portaluppi, D. E. Tenhouse, A. Lloyd, D. R. Eborn, S. Kacuba 2 and J. M. DeJarnette
Dairy Research 2006 OVARIAN RESPONSES AND CONCEPTION RATES IN RESPONSE TO GnRH, hcg, AND PROGESTERONE 1 J. S. Stevenson, M. A. Portaluppi, D. E. Tenhouse, A. Lloyd, D. R. Eborn, S. Kacuba 2 and J. M. DeJarnette
Basic Reproduction & Genetics. Steve Pritchard UNL Extension Educator Boone-Nance Counties
 Basic Reproduction & Genetics Steve Pritchard UNL Extension Educator Boone-Nance Counties Hormonal Regulation of the Estrous Cycle Several hormones regulate the estrous cycle Changes in the concentrations
Basic Reproduction & Genetics Steve Pritchard UNL Extension Educator Boone-Nance Counties Hormonal Regulation of the Estrous Cycle Several hormones regulate the estrous cycle Changes in the concentrations
Semen Characteristics and Artificial Insemination in the Bactrian Camel (25 Sep 2000)
 In: Recent Advances in Camelid Reproduction, Skidmore J.A. and Adams G.P. (Eds.) Publisher: International Veterinary Information Service (www.ivis.org) Semen Characteristics and Artificial Insemination
In: Recent Advances in Camelid Reproduction, Skidmore J.A. and Adams G.P. (Eds.) Publisher: International Veterinary Information Service (www.ivis.org) Semen Characteristics and Artificial Insemination
Dr. Julio Giordano. Ovulation. Follicle. Corpus Luteum. GnRH
 Dr. Julio Giordano Follicle Corpus Luteum LH FSH E2 Hypothalamic hormones Gonadotropin releasing hormone () Hormone Concentration CL LH (ng/ml) 12 10 8 6 4 2 LH Response Cows Treated with 28 h (22-36)
Dr. Julio Giordano Follicle Corpus Luteum LH FSH E2 Hypothalamic hormones Gonadotropin releasing hormone () Hormone Concentration CL LH (ng/ml) 12 10 8 6 4 2 LH Response Cows Treated with 28 h (22-36)
Ultrasonographic Observation of Follicular and Luteal Dynamics during the Estrous Cycle in Shiba Goats
 Journal of Reproduction and Development, Vol. 46, No. 1, 2000 Ultrasonographic Observation of Follicular and Luteal Dynamics during the Estrous Cycle in Shiba Goats Jun ORITA, Tomomi TANAKA, Hideo KAMOMAE
Journal of Reproduction and Development, Vol. 46, No. 1, 2000 Ultrasonographic Observation of Follicular and Luteal Dynamics during the Estrous Cycle in Shiba Goats Jun ORITA, Tomomi TANAKA, Hideo KAMOMAE
LIFE SCIENCES Grade 12 REPRODUCTION 30 JUNE 2014
 REPRODUCTION 30 JUNE 2014 Checklist Make sure you Can describe different reproductive strategies of vertebrates Are able to identify the structure and function of the male and female reproductive organs
REPRODUCTION 30 JUNE 2014 Checklist Make sure you Can describe different reproductive strategies of vertebrates Are able to identify the structure and function of the male and female reproductive organs
Chapter 36 Active Reading Guide Reproduction and Development
 Name: AP Biology Mr. Croft Chapter 36 Active Reading Guide Reproduction and Development Section 1 1. Distinguish between sexual reproduction and asexual reproduction. 2. Which form of reproduction: a.
Name: AP Biology Mr. Croft Chapter 36 Active Reading Guide Reproduction and Development Section 1 1. Distinguish between sexual reproduction and asexual reproduction. 2. Which form of reproduction: a.
SUMMARY OF PRODUCT CHARACTERISTICS. Active Ingredient Buserelin acetate (INN)
 1 (5) SUMMARY OF PRODUCT CHARACTERISTICS 1. Name of the veterinary medicinal product Receptal ad us. vet. 2. Qualitative and quantitative composition 1 ml contains Active Ingredient Buserelin acetate (INN)
1 (5) SUMMARY OF PRODUCT CHARACTERISTICS 1. Name of the veterinary medicinal product Receptal ad us. vet. 2. Qualitative and quantitative composition 1 ml contains Active Ingredient Buserelin acetate (INN)
Chapter 14 The Reproductive System
 Biology 12 Name: Reproductive System Per: Date: Chapter 14 The Reproductive System Complete using BC Biology 12, page 436-467 14. 1 Male Reproductive System pages 440-443 1. Distinguish between gametes
Biology 12 Name: Reproductive System Per: Date: Chapter 14 The Reproductive System Complete using BC Biology 12, page 436-467 14. 1 Male Reproductive System pages 440-443 1. Distinguish between gametes
REPRODUCTION The diagram below shows a section through seminiferous tubules in a testis.
 1. The diagram below shows a section through seminiferous tubules in a testis. Which cell produces testosterone? 2. A function of the interstitial cells in the testes is to produce A sperm B testosterone
1. The diagram below shows a section through seminiferous tubules in a testis. Which cell produces testosterone? 2. A function of the interstitial cells in the testes is to produce A sperm B testosterone
Synchronization of Ovulation and Fixed-Time Insemination for Improvement of Conception Rate in Dairy Herds with Poor Estrus Detection Efficiency
 Journal of Reproduction and Development, Vol. 45, No. 1, 1999 Synchronization of Ovulation and Fixed-Time Insemination for Improvement of Conception Rate in Dairy Herds with Poor Estrus Detection Efficiency
Journal of Reproduction and Development, Vol. 45, No. 1, 1999 Synchronization of Ovulation and Fixed-Time Insemination for Improvement of Conception Rate in Dairy Herds with Poor Estrus Detection Efficiency
Prostaglandin F 2α. J. S. Stevenson, S. L. Pulley, and H. I. Mellieon, Jr.
 Prostaglandin F 2α and GnRH Administration Improved Progesterone tatus, Luteal Number, and Proportion of Ovular and Anovular Dairy Cows with Corpora Lutea efore a Timed Artificial Insemination Program
Prostaglandin F 2α and GnRH Administration Improved Progesterone tatus, Luteal Number, and Proportion of Ovular and Anovular Dairy Cows with Corpora Lutea efore a Timed Artificial Insemination Program
Chapter 22 The Reproductive System (I)
 Chapter 22 The Reproductive System (I) An Overview of Reproductive Physiology o The Male Reproductive System o The Female Reproductive System 22.1 Reproductive System Overview Reproductive system = all
Chapter 22 The Reproductive System (I) An Overview of Reproductive Physiology o The Male Reproductive System o The Female Reproductive System 22.1 Reproductive System Overview Reproductive system = all
Testes (male gonads) -Produce sperm -Produce sex hormones -Found in a sac called the scrotum -Suspended outside of the body cavity for temperature
 REPRODUCTION Testes (male gonads) -Produce sperm -Produce sex hormones -Found in a sac called the scrotum -Suspended outside of the body cavity for temperature reduction -Testes wall made of fibrous connective
REPRODUCTION Testes (male gonads) -Produce sperm -Produce sex hormones -Found in a sac called the scrotum -Suspended outside of the body cavity for temperature reduction -Testes wall made of fibrous connective
Theriogenology Department, Faculty of Veterinary Medicine, Beni Suef University, Egypt 2
 Theriogenology Insight: 3(1):11-16. April, 2013 Ultrasonic monitoring and biometry of ovaries and ovarian structures during superovulation following transvagianl follicle ablation in Murrah buffaloes S.M.
Theriogenology Insight: 3(1):11-16. April, 2013 Ultrasonic monitoring and biometry of ovaries and ovarian structures during superovulation following transvagianl follicle ablation in Murrah buffaloes S.M.
Five-day Resynch Programs in Dairy Cows Including Controlled Internal Drug Release at Two Stages Post- Artificial Insemination
 Five-day Resynch Programs in Dairy Cows Including Controlled Internal Drug Release at Two Stages Post- Artificial Insemination S. L. Pulley, S. L. Hill, and J. S. Stevenson Summary Two experiments were
Five-day Resynch Programs in Dairy Cows Including Controlled Internal Drug Release at Two Stages Post- Artificial Insemination S. L. Pulley, S. L. Hill, and J. S. Stevenson Summary Two experiments were
The beginning of puberty is marked by the progressive increase in the production of sex hormones.
 Puberty is characterized by the changes that prepare the human body for the ability to reproduce. This stage generally occurs between the ages of 10 and 14 years old. The beginning of puberty is marked
Puberty is characterized by the changes that prepare the human body for the ability to reproduce. This stage generally occurs between the ages of 10 and 14 years old. The beginning of puberty is marked
Influence of large follicles on oestrus induction and ovulation after embryo collection in superovulated Japanese Black cows
 J. Reprod. Engineer. 2015; 17: 1 5. http://sreprod.jp/contents.htm = Original Article = Journal of REPRODUCTION ENGINEERING Influence of large follicles on oestrus induction and ovulation after embryo
J. Reprod. Engineer. 2015; 17: 1 5. http://sreprod.jp/contents.htm = Original Article = Journal of REPRODUCTION ENGINEERING Influence of large follicles on oestrus induction and ovulation after embryo
Cryopreservation of epididymal alpaca (Vicugna pacos) sperm: a comparison of citrate-, Tris- and lactose-based diluents and pellets and straws
 CSIRO PUBLISHING Reproduction, Fertility and Development, 2007, 19, 792 796 www.publish.csiro.au/journals/rfd Cryopreservation of epididymal alpaca (Vicugna pacos) sperm: a comparison of citrate-, Tris-
CSIRO PUBLISHING Reproduction, Fertility and Development, 2007, 19, 792 796 www.publish.csiro.au/journals/rfd Cryopreservation of epididymal alpaca (Vicugna pacos) sperm: a comparison of citrate-, Tris-
Alpaca semen quality in relation to different diets
 CSIRO PUBLISHING Reproduction, Fertility and Development http://dx.doi.org/10.1071/rd12050 Alpaca semen quality in relation to different diets N. S. Juyena A, J. Vencato A, G. Pasini B, I. Vazzana C and
CSIRO PUBLISHING Reproduction, Fertility and Development http://dx.doi.org/10.1071/rd12050 Alpaca semen quality in relation to different diets N. S. Juyena A, J. Vencato A, G. Pasini B, I. Vazzana C and
Effects of Buserelin acetate and semen extension on fertility in rabbit does under artificial insemination
 Nig. J. Anim. Prod. 2017 44(1):130-135 Nigerian Society for Animal Production Nigerian Journal of Animal Production Effects of Buserelin acetate and semen extension on fertility in rabbit does under artificial
Nig. J. Anim. Prod. 2017 44(1):130-135 Nigerian Society for Animal Production Nigerian Journal of Animal Production Effects of Buserelin acetate and semen extension on fertility in rabbit does under artificial
Management of recipients for an embryo transfer program in dromedary camels (Camelus dromedarius)
 Management of recipients for an embryo transfer program in dromedary camels (Camelus dromedarius) J.A. Skidmore* and M. Billah Camel Reproduction Centre, P.O. Box 79914, Dubai, United Arab Emirates Summary
Management of recipients for an embryo transfer program in dromedary camels (Camelus dromedarius) J.A. Skidmore* and M. Billah Camel Reproduction Centre, P.O. Box 79914, Dubai, United Arab Emirates Summary
Effects of Label-Dose Permethrin Administration on Reproductive Function and Embryo Quality on Superovulated Beef Heifers
 Animal Industry Report AS 662 ASL R3050 2016 Effects of Label-Dose Permethrin Administration on Reproductive Function and Embryo Quality on Superovulated Beef Heifers Tyler M. Dohlman Iowa State University,
Animal Industry Report AS 662 ASL R3050 2016 Effects of Label-Dose Permethrin Administration on Reproductive Function and Embryo Quality on Superovulated Beef Heifers Tyler M. Dohlman Iowa State University,
Study Guide Answer Key Reproductive System
 Biology 12 Human Biology Textbook: BC Biology 12 Study Guide Answer Key Reproductive System 1. Distinguish between a gamete and a gonad using specific examples from the male and female systems. Gonads
Biology 12 Human Biology Textbook: BC Biology 12 Study Guide Answer Key Reproductive System 1. Distinguish between a gamete and a gonad using specific examples from the male and female systems. Gonads
CASE 41. What is the pathophysiologic cause of her amenorrhea? Which cells in the ovary secrete estrogen?
 CASE 41 A 19-year-old woman presents to her gynecologist with complaints of not having had a period for 6 months. She reports having normal periods since menarche at age 12. She denies sexual activity,
CASE 41 A 19-year-old woman presents to her gynecologist with complaints of not having had a period for 6 months. She reports having normal periods since menarche at age 12. She denies sexual activity,
TREATMENT OUTCOMES IN POSTPARTUM ANOESTRUS COWS GUIDED BY TRANSRECTAL ULTRASONOGRAPHY ABSTRACT
 Progress. Agric. 24(1 & 2): 93 100, 2013 ISSN 1017-8139 TREATMENT OUTCOMES IN POSTPARTUM ANOESTRUS COWS GUIDED BY TRANSRECTAL ULTRASONOGRAPHY M. R. Islam, N. S. Juyena 1 *, M. M. U. Bhuiyan, M. M. Rahman
Progress. Agric. 24(1 & 2): 93 100, 2013 ISSN 1017-8139 TREATMENT OUTCOMES IN POSTPARTUM ANOESTRUS COWS GUIDED BY TRANSRECTAL ULTRASONOGRAPHY M. R. Islam, N. S. Juyena 1 *, M. M. U. Bhuiyan, M. M. Rahman
Endocrine System Hormones & Homeostasis. Regents Biology
 Endocrine System Hormones & Homeostasis 2009-2010 Homeostasis Homeostasis maintaining internal balance in the body organism must keep internal conditions stable even if environment changes also called
Endocrine System Hormones & Homeostasis 2009-2010 Homeostasis Homeostasis maintaining internal balance in the body organism must keep internal conditions stable even if environment changes also called
Fertility Diagnostics
 Fertility Diagnostics Fertility hormones measured on PATHFAST For internal use only Diagnostics PATHFAST Chemiluminescence-immuno-analyzer 1 Content: page 1. Fertility hormones - general aspects 1.1 Reproductive
Fertility Diagnostics Fertility hormones measured on PATHFAST For internal use only Diagnostics PATHFAST Chemiluminescence-immuno-analyzer 1 Content: page 1. Fertility hormones - general aspects 1.1 Reproductive
MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question.
 MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1) Which of the following hormones controls the release of anterior pituitary gonadotropins? A) LH
MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1) Which of the following hormones controls the release of anterior pituitary gonadotropins? A) LH
DATE: NAME: CLASS: Chapter 14 Test
 Multiple Choice Questions Decide which of the choices best completes the statement or answers the question. Locate that question number on the separate answer sheet provided. Use the procedure described
Multiple Choice Questions Decide which of the choices best completes the statement or answers the question. Locate that question number on the separate answer sheet provided. Use the procedure described
Unit B Understanding Animal Body Systems. Lesson 6 Anatomy and Physiology of Animal Reproduction Systems
 Unit B Understanding Animal Body Systems Lesson 6 Anatomy and Physiology of Animal Reproduction Systems 1 Terms Alimentary canal Bladder Cervix Clitoris Cloaca Copulation Cowper s gland Epididymis Fallopian
Unit B Understanding Animal Body Systems Lesson 6 Anatomy and Physiology of Animal Reproduction Systems 1 Terms Alimentary canal Bladder Cervix Clitoris Cloaca Copulation Cowper s gland Epididymis Fallopian
Available online at Theriogenology xxx (2009) xxx xxx
 Available online at www.sciencedirect.com 1 2 3 4 5 6 7 8 9 10 11 The effect of hormone treatments (hcg and cloprostenol) and season on the incidence of hemorrhagic anovulatory follicles in the mare: A
Available online at www.sciencedirect.com 1 2 3 4 5 6 7 8 9 10 11 The effect of hormone treatments (hcg and cloprostenol) and season on the incidence of hemorrhagic anovulatory follicles in the mare: A
Animal Reproduction Chapter 46. Fission. Budding. Parthenogenesis. Fragmentation 11/27/2017
 Animal Reproduction Chapter 46 Both asexual and sexual reproduction occur in the animal kingdom Sexual reproduction is the creation of an offspring by fusion of a male gamete (sperm) and female gamete
Animal Reproduction Chapter 46 Both asexual and sexual reproduction occur in the animal kingdom Sexual reproduction is the creation of an offspring by fusion of a male gamete (sperm) and female gamete
INDUCTIONS OF SUPEROVULATION USING SEVERAL FSH REGIMENS IN HOLSTEIN-FRIESIAN HEIFERS
 lpn. l. Vet. Res., 33, 45-50 (1985) INDUCTIONS OF SUPEROVULATION USING SEVERAL FSH REGIMENS IN HOLSTEIN-FRIESIAN HEIFERS Yoshiyuki TAKAHASHI and Hiroshi KANAGAWA (Received for publication, February 2,
lpn. l. Vet. Res., 33, 45-50 (1985) INDUCTIONS OF SUPEROVULATION USING SEVERAL FSH REGIMENS IN HOLSTEIN-FRIESIAN HEIFERS Yoshiyuki TAKAHASHI and Hiroshi KANAGAWA (Received for publication, February 2,
Concentrations of Circulating Gonadotropins During. Various Reproductive States in Mares
 BIOLOGY OF REPRODUCTION, 744-75 (19) Concentrations of Circulating Gonadotropins During Various Reproductive States in Mares KURT F. MILLER, S. L. BERG, D. C. SHARP and. J. GINTHER Department of Veterinary
BIOLOGY OF REPRODUCTION, 744-75 (19) Concentrations of Circulating Gonadotropins During Various Reproductive States in Mares KURT F. MILLER, S. L. BERG, D. C. SHARP and. J. GINTHER Department of Veterinary
MULTIPLE CHOICE: match the term(s) or description with the appropriate letter of the structure.
 Chapter 27 Exam Due NLT Thursday, July 31, 2015 Name MULTIPLE CHOICE: match the term(s) or description with the appropriate letter of the structure. Figure 27.1 Using Figure 27.1, match the following:
Chapter 27 Exam Due NLT Thursday, July 31, 2015 Name MULTIPLE CHOICE: match the term(s) or description with the appropriate letter of the structure. Figure 27.1 Using Figure 27.1, match the following:
Chapter 46 ~ Animal Reproduction
 Chapter 46 ~ Animal Reproduction Overview Asexual (one parent) fission (parent separation) budding (corals) fragmentation & regeneration (inverts) parthenogenesis Sexual (fusion of haploid gametes) gametes
Chapter 46 ~ Animal Reproduction Overview Asexual (one parent) fission (parent separation) budding (corals) fragmentation & regeneration (inverts) parthenogenesis Sexual (fusion of haploid gametes) gametes
List of Equipment, Tools, Supplies, and Facilities:
 Unit B: Understanding Animal Body Systems Lesson 6: Anatomy and Physiology of Animal Reproductive Systems Student Learning Objectives: Instruction in this lesson should result in students achieving the
Unit B: Understanding Animal Body Systems Lesson 6: Anatomy and Physiology of Animal Reproductive Systems Student Learning Objectives: Instruction in this lesson should result in students achieving the
1. During the follicular phase of the ovarian cycle, the hypothalamus releases GnRH.
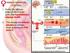 1. During the follicular phase of the ovarian cycle, the hypothalamus releases GnRH. 2. This causes the anterior pituitary to secrete small quantities of FSH and LH. 3. At this time, the follicles in the
1. During the follicular phase of the ovarian cycle, the hypothalamus releases GnRH. 2. This causes the anterior pituitary to secrete small quantities of FSH and LH. 3. At this time, the follicles in the
The reproductive lifespan
 The reproductive lifespan Reproductive potential Ovarian cycles Pregnancy Lactation Male Female Puberty Menopause Age Menstruation is an external indicator of ovarian events controlled by the hypothalamicpituitary
The reproductive lifespan Reproductive potential Ovarian cycles Pregnancy Lactation Male Female Puberty Menopause Age Menstruation is an external indicator of ovarian events controlled by the hypothalamicpituitary
THE APPLICATION OF EQUINE CHORIONIC GONADOTROPIN (ecg) AND PROSTAGLANDIN F2α TO INCREASE THE RATE OF PREGNANCY IN BALI CATTLE AT BULELENG, BALI
 The Veterinary Medicine International Conference 2017 Volume 2017 Conference Paper THE APPLICATION OF EQUINE CHORIONIC GONADOTROPIN (ecg) AND PROSTAGLANDIN F2α TO INCREASE THE RATE OF PREGNANCY IN BALI
The Veterinary Medicine International Conference 2017 Volume 2017 Conference Paper THE APPLICATION OF EQUINE CHORIONIC GONADOTROPIN (ecg) AND PROSTAGLANDIN F2α TO INCREASE THE RATE OF PREGNANCY IN BALI
Course: Animal Production. Instructor: Ms. Hutchinson. Objectives: After completing this unit of instruction, students will be able to:
 Course: Animal Production Unit Title: Hormones TEKS: 130.3 (C)(6)(A) Instructor: Ms. Hutchinson Objectives: After completing this unit of instruction, students will be able to: A. Define what hormones
Course: Animal Production Unit Title: Hormones TEKS: 130.3 (C)(6)(A) Instructor: Ms. Hutchinson Objectives: After completing this unit of instruction, students will be able to: A. Define what hormones
Human Reproductive Anatomy The female anatomy first just the reproductive parts:
 Human Reproduction: Structures, Functions, and Hormones Human reproduction differs from the mechanisms in lower vertebrates and many invertebrates. We depend on internal fertilization sperm are deposited
Human Reproduction: Structures, Functions, and Hormones Human reproduction differs from the mechanisms in lower vertebrates and many invertebrates. We depend on internal fertilization sperm are deposited
9.4 Regulating the Reproductive System
 9.4 Regulating the Reproductive System The Reproductive System to unite a single reproductive cell from a female with a single reproductive cell from a male Both male and female reproductive systems include
9.4 Regulating the Reproductive System The Reproductive System to unite a single reproductive cell from a female with a single reproductive cell from a male Both male and female reproductive systems include
PHARMACOLOGY AND TOXICOLOGY:
 ANADA200-541, Approved by FDA Equivalent to 100 mcg gonadorelin/ml Sterile solution For the treatment of cystic ovaries in dairy cattle. For use with cloprostenol sodium to synchronize estrous cycles to
ANADA200-541, Approved by FDA Equivalent to 100 mcg gonadorelin/ml Sterile solution For the treatment of cystic ovaries in dairy cattle. For use with cloprostenol sodium to synchronize estrous cycles to
are associated with low fertility in dairy cows
 J. Dairy Sci. 95 :2355 2361 http://dx.doi.org/ 10.3168/jds.2011-4325 American Dairy Science Association, 2012. Open access under CC BY-NC-ND license. are associated with low fertility in dairy cows F.
J. Dairy Sci. 95 :2355 2361 http://dx.doi.org/ 10.3168/jds.2011-4325 American Dairy Science Association, 2012. Open access under CC BY-NC-ND license. are associated with low fertility in dairy cows F.
4-7 July 2000 Valencia Spain
 Proceedings of the 4-7 July 2000 Valencia Spain These proceedings were printed as a special issue of WORLD RABBIT SCIENCE, the journal of the World Rabbit Science Association, Volume 8, supplement 1 ISSN
Proceedings of the 4-7 July 2000 Valencia Spain These proceedings were printed as a special issue of WORLD RABBIT SCIENCE, the journal of the World Rabbit Science Association, Volume 8, supplement 1 ISSN
What are the main functions of the male reproductive system? 1. Produce sperm 2. Deposit sperm into the female 3. Provide a pathway for the removal
 What are the main functions of the male reproductive system? 1. Produce sperm 2. Deposit sperm into the female 3. Provide a pathway for the removal of urine Where is sperm produced? -In the 2 testes What
What are the main functions of the male reproductive system? 1. Produce sperm 2. Deposit sperm into the female 3. Provide a pathway for the removal of urine Where is sperm produced? -In the 2 testes What
Effect of reducing the period of follicle dominance in a timed artificial insemination protocol on reproduction of dairy cows
 J. Dairy Sci. 93 :2976 2988 doi: 10.3168/jds.2009-2870 American Dairy Science Association, 2010. Effect of reducing the period of follicle dominance in a timed artificial insemination protocol on reproduction
J. Dairy Sci. 93 :2976 2988 doi: 10.3168/jds.2009-2870 American Dairy Science Association, 2010. Effect of reducing the period of follicle dominance in a timed artificial insemination protocol on reproduction
Female Reproductive System. Justin D. Vidal
 Female Reproductive System Justin D. Vidal If you cannot identify the tissue, then it is probably part of the female reproductive system! Introduction The female reproductive system is constantly changing,
Female Reproductive System Justin D. Vidal If you cannot identify the tissue, then it is probably part of the female reproductive system! Introduction The female reproductive system is constantly changing,
VM 744 THERIOGENOLOGY Spring 2013
 VM 744 THERIOGENOLOGY Spring 2013 Credits-3 Coordinator: Patrick M., DVM, PhD, Diplomate ACT Department of Clinical Sciences Office: Equine Reproduction Laboratory Phone: 491-8626 email: pmccue@colostate.edu
VM 744 THERIOGENOLOGY Spring 2013 Credits-3 Coordinator: Patrick M., DVM, PhD, Diplomate ACT Department of Clinical Sciences Office: Equine Reproduction Laboratory Phone: 491-8626 email: pmccue@colostate.edu
I. ART PROCEDURES. A. In Vitro Fertilization (IVF)
 DFW Fertility Associates ASSISTED REPRODUCTIVE TECHNOLOGY (ART) Welcome to DFW Fertility Associates/ Presbyterian-Harris Methodist Hospital ARTS program. This document provides an overview of treatment
DFW Fertility Associates ASSISTED REPRODUCTIVE TECHNOLOGY (ART) Welcome to DFW Fertility Associates/ Presbyterian-Harris Methodist Hospital ARTS program. This document provides an overview of treatment
Abstracts for the KSAR and JSAR Joint Symposium. Fertility control in female domestic animals: From basic understanding to application
 Abstracts for the KSAR and JSAR Joint Symposium Fertility control in female domestic animals: From basic understanding to application Current Research Orientation in Livestock Reproduction in Korea Choong-Saeng
Abstracts for the KSAR and JSAR Joint Symposium Fertility control in female domestic animals: From basic understanding to application Current Research Orientation in Livestock Reproduction in Korea Choong-Saeng
Manipulation of Ovarian Function for the Reproductive Management of Dairy Cows
 Veterinary Research Communications,28(2004) 111 119 2004 Kluwer Academic Publishers. Printed in the Netherlands Manipulation of Ovarian Function for the Reproductive Management of Dairy Cows W.W. Thatcher1*,
Veterinary Research Communications,28(2004) 111 119 2004 Kluwer Academic Publishers. Printed in the Netherlands Manipulation of Ovarian Function for the Reproductive Management of Dairy Cows W.W. Thatcher1*,
Student Academic Learning Services Page 1 of 5 Reproductive System Practice
 Student Academic Learning Services Page 1 of 5 Reproductive System Practice Fill in the blanks using the words listed: accessory glands, vas deferens, penis, scrotum, fallopian tube, testes, urethra, vagina,
Student Academic Learning Services Page 1 of 5 Reproductive System Practice Fill in the blanks using the words listed: accessory glands, vas deferens, penis, scrotum, fallopian tube, testes, urethra, vagina,
INDUCING DOUBLE OVULATIONS IN BEEF CATTLE VIA SIMULTANEOUS LUTEAL REGRESSION AND FOLLICLE WAVE EMERGENCE IN A LOW PROGESTERONE ENVIRONMENT
 INDUCING DOUBLE OVULATIONS IN BEEF CATTLE VIA SIMULTANEOUS LUTEAL REGRESSION AND FOLLICLE WAVE EMERGENCE IN A LOW PROGESTERONE ENVIRONMENT E M M A M. J I N K S Honors Research Thesis Research Advisor:
INDUCING DOUBLE OVULATIONS IN BEEF CATTLE VIA SIMULTANEOUS LUTEAL REGRESSION AND FOLLICLE WAVE EMERGENCE IN A LOW PROGESTERONE ENVIRONMENT E M M A M. J I N K S Honors Research Thesis Research Advisor:
Female Reproductive Physiology. Dr Raelia Lew CREI, FRANZCOG, PhD, MMed, MBBS Fertility Specialist, Melbourne IVF
 Female Reproductive Physiology Dr Raelia Lew CREI, FRANZCOG, PhD, MMed, MBBS Fertility Specialist, Melbourne IVF REFERENCE Lew, R, Natural History of ovarian function including assessment of ovarian reserve
Female Reproductive Physiology Dr Raelia Lew CREI, FRANZCOG, PhD, MMed, MBBS Fertility Specialist, Melbourne IVF REFERENCE Lew, R, Natural History of ovarian function including assessment of ovarian reserve
Effect of repeated laparoscopic ovum pick-up on yield and quality of oocytes in goats
 Effect of repeated laparoscopic ovum pick-up on yield and quality of oocytes in goats Nor Farizah, A.H., Rahman, M.M., Wan Khadijah, W.E. and Abdullah, R.B.* Animal Biotechnology-Embryo Laboratory (ABEL),
Effect of repeated laparoscopic ovum pick-up on yield and quality of oocytes in goats Nor Farizah, A.H., Rahman, M.M., Wan Khadijah, W.E. and Abdullah, R.B.* Animal Biotechnology-Embryo Laboratory (ABEL),
Reproductive FSH. Analyte Information
 Reproductive FSH Analyte Information 1 Follicle-stimulating hormone Introduction Follicle-stimulating hormone (FSH, also known as follitropin) is a glycoprotein hormone secreted by the anterior pituitary
Reproductive FSH Analyte Information 1 Follicle-stimulating hormone Introduction Follicle-stimulating hormone (FSH, also known as follitropin) is a glycoprotein hormone secreted by the anterior pituitary
Investigation: The Human Menstrual Cycle Research Question: How do hormones control the menstrual cycle?
 Investigation: The Human Menstrual Cycle Research Question: How do hormones control the menstrual cycle? Introduction: The menstrual cycle (changes within the uterus) is an approximately 28-day cycle that
Investigation: The Human Menstrual Cycle Research Question: How do hormones control the menstrual cycle? Introduction: The menstrual cycle (changes within the uterus) is an approximately 28-day cycle that
Rodrigo Carrasco, Jaswant Singh and Gregg P. Adams *
 Carrasco et al. Reproductive Biology and Endocrinology (2016) 14:47 DOI 10.1186/s12958-016-0182-9 RESEARCH Open Access The dynamics of trka expression in the bovine ovary are associated with a luteotrophic
Carrasco et al. Reproductive Biology and Endocrinology (2016) 14:47 DOI 10.1186/s12958-016-0182-9 RESEARCH Open Access The dynamics of trka expression in the bovine ovary are associated with a luteotrophic
Web Activity: Simulation Structures of the Female Reproductive System
 differentiate. The epididymis is a coiled tube found along the outer edge of the testis where the sperm mature. 3. Testosterone is a male sex hormone produced in the interstitial cells of the testes. It
differentiate. The epididymis is a coiled tube found along the outer edge of the testis where the sperm mature. 3. Testosterone is a male sex hormone produced in the interstitial cells of the testes. It
Hormonal Control of Human Reproduction
 Hormonal Control of Human Reproduction Bởi: OpenStaxCollege The human male and female reproductive cycles are controlled by the interaction of hormones from the hypothalamus and anterior pituitary with
Hormonal Control of Human Reproduction Bởi: OpenStaxCollege The human male and female reproductive cycles are controlled by the interaction of hormones from the hypothalamus and anterior pituitary with
Key note presentation: Methods to improve the reproductive of camel Julian A. Skidmore Camel Reproduction Centre, Dubai, United Arab Emirates
 Key note presentation: Methods to improve the reproductive of camel Julian A. Skidmore Camel Reproduction Centre, Dubai, United Arab Emirates ABSTRACT The reproductive performance of camels is generally
Key note presentation: Methods to improve the reproductive of camel Julian A. Skidmore Camel Reproduction Centre, Dubai, United Arab Emirates ABSTRACT The reproductive performance of camels is generally
AP Biology Ch ANIMAL REPRODUCTION. Using only what you already know (you cannot look up anything) complete the chart below.
 AP Biology Ch. 46 - ANIMAL REPRODUCTION Using only what you already know (you cannot look up anything) complete the chart below. I. Overview of Animal Reproduction A. Both asexual and sexual reproduction
AP Biology Ch. 46 - ANIMAL REPRODUCTION Using only what you already know (you cannot look up anything) complete the chart below. I. Overview of Animal Reproduction A. Both asexual and sexual reproduction
