Coenzyme Q 10 Improves Developmental Competence of Mice Pre-antral Follicle Derived From Vitrified Ovary
|
|
|
- Augusta Wilson
- 5 years ago
- Views:
Transcription
1 Coenzyme Q 10 Improves Developmental Competence of Mice Pre-antral Follicle Derived From Vitrified Ovary Elmira Hosseinzadeh 1, Saeed Zavareh 1, 2,*, Taghi Lashkarboluki 1,2 1 School of Biology, Damghan University, Damghan, Iran. 2 Institute of biological sciences, Damghan University, Damghan, Iran *Corresponding Author: address: Zavareh.S@du.ac.ir (S. Zavareh) ABSTRACT The aim of this study was to investigate the in vitro development of isolated pre-antral follicles derived from vitrified ovaries in the presence of coenzyme Q 10 (CoQ 10 ). Mice pre-antral follicles derived from fresh and vitrified-warmed ovarian tissues were cultured individually in α-mem medium supplemented with or without CoQ 10, followed by adding human Chorionic Gonadotropin (hcg) to induce ovulation. The follicle development parameters and ovulated oocyte maturation were assessed. The developmental parameters of pre-antral follicles and oocyte maturation rates were significantly higher in CoQ 10 pretreatment groups of both vitrified and fresh samples compared to the respective CoQ 10 free groups. CoQ 10 improves the in vitro development of pre-antral follicles derived from fresh and vitrified warmed ovaries. Keywords: Vitrification, Ovary, Pre-antral follicles, Coenzyme Q 10. INTRODUCTION The Ovarian tissue cryopreservation is a good strategy to preserve fertility of women who are at risk of premature ovarian failure or need chemo and radiation therapy. Ovarian tissue cryopreservation is wrapped methods because it can be done in several protocols with many variable [1-3]. However, vitrification as simple, quick and economical method is more efficient than other approaches [4]. Vitrification as a part of assisted reproduction technique (ART) induces several destructive phenomena which among them, oxidative Stress (OS) is particularly important [5, 6]. OS occur as a result of an imbalance between the systemic production of reactive oxygen species (ROS) and its removal [7] which leading to cell toxicity, DNA damage and apoptosis [8]. The development of OS is one of the main reasons for the poor development of gametes and embryos in ART [5]. In in vivo condition, ROS constantly be neutralized by enzymatic and non-enzymatic antioxidant agents. However, a small amount of ROS is required for setting various vital functions of gametes development, as a result, the activity of ROS must be kept at a normal balance [5, 7-9]. ROS generation following in vitro condition is unavoidable [5, 6, 9]. Therefore, supplemented culture media with antioxidants were usually used. However the use and potential benefits of antioxidants during in vitro culture is still debate [9, 10]. Coenzyme Q 10 (CoQ 10 ) are well known for antioxidant properties in both oxidized and reduced form to eliminate free-radicals [11, 12]. CoQ 10 is a fat-soluble vitamin like agent involved in mitochondrial respiratory chain [9, 13]. It has been shown that, there is a direct correlation between seminal plasma CoQ 10 concentration and semen parameters [14]. Also, the previous results showed that administration of CoQ 10 in the infertile women had beneficial effects [12, 13, 15]. Presence of CoQ 10 in the human follicular fluid has been demonstrated [13]. Although the actions of CoQ 10 in the follicular fluid is unclear, total CoQ 10 levels were higher in follicular fluids correlated with mature oocyte and high quality embryos, suggesting a possible role in mechanisms of control and growth of follicles and protective role against OS [13]. 65
2 Although, several antioxidants have been investigated on in vitro development of preantral follicles, it has not yet been determined whether adding the CoQ 10 in culture medium affect the developmental competence of preantral follicles derived from fresh and vitrified warmed ovaries. Therefore, the aim of this study was to investigate the rates of developmental competence of isolated mice pre-antral follicles during in vitro culture in the presence of CoQ 10. MATERIALS AND METHODS Reagents All reagents were purchased from Sigma- Aldrich UK, unless otherwise stated and all media were made with Mili-Q water. Animals and experimental Groups In this experimental study, Female Naval Medical Research Institute (NMRI) mice were purchased from Razi Vaccine and Serum Research Institute and were cared for and used according to the Animal Ethics Committee. The mice were housed and bred in the Animal House under light and temperature-controlled conditions (12 h of light and 12 h of darkness; and 24ᵒC) and they provided with food and water ad libitum. For all experiments, mice aged days were sacrificed by cervical dislocation and their ovaries were removed and placed in α-minimal essential medium (α-mem; Gibco, UK) supplemented with 10% fetal bovine serum (FBS; Gibco, UK), 2.2 g/l sodium bicarbonate, 25 mm HEPES, 100 IU/ml penicillin and 75 µg/ml streptomycin. The ovaries were randomly divided into two main groups: (1) Fresh (control) and (2) vitrified groups. Isolated pre-antral follicles of each groups were cultured in the presence or absence of CoQ 10 for evaluation of development parameters. Ovarian tissue cryopreservation and thawing The vitrification procedure was based on the method described previously [16] with some modifications. The ovary was placed in an equilibrium solution containing 7.5%v/v ethylene glycol (EG), 7.5% v/v dimethyl sulfoxide (DMSO) and 20%FBS in Dulbecco's phosphate-buffered saline (DPBS) medium for 10 min and then they were placed for 2min in the vitrification solution containing 15% v/v EG, 15% v/v DMSO, 20% FBS and 0.5M sucrose in DPBS medium. Then ovaries were placed in a Cryovial carrier and transferred to liquid nitrogen (LN2) immediately. All equilibration and vitrification steps were carried out at room temperature. For warming, the ovaries were left in the warming solution (1M sucrose in DPBS) for 5 min and transferred into droplets of DPBS medium containing 0.5 and 0.25M sucrose at an interval of 5 min at room temperature. Finally, ovaries were placed into fresh α-mem medium supplemented with 10% FBS at 37 C in CO 2 incubator for another 20 min before submitted to isolation of their pre-antral follicle. Pre-antral follicle isolation Pre-antral follicles derived from fresh and vitrified-warmed ovaries were isolated mechanically as described previously [17] by using a 29 gauge needle under a stereomicroscope (Nikon, Japan) at 10 magnification. Isolated follicles were selected according to the following criteria: rounded structure with µm diameters, containing visible centrally located oocyte surrounded with intact several layers of granulosa cells, intact basement membrane, at least one layer of theca cells and without antral cavity. During the operating procedures the culture medium was always kept at 37 C. In vitro cultured and assessment of developmental parameters: Isolated pre-antral follicles from fresh and vitrified ovaries were cultured in 25 μl drops of α-mem medium supplemented with 5% FBS, 100mIU/ml recombinant human follicle stimulating hormone (rhfsh), 1% Insulintransferin-selenium (ITS), 10ng/ml Epidermal growth factor (EGF), 100 IU/ml penicillin, 75 µg/ml streptomycin, 2.2g/l sodium bicarbonate and 50µM CoQ 10 under mineral oil in a humidified atmosphere of 5% CO2 in air at 37 C in Falcon Petri dishes (60 x 15 mm) for up to 12 days. At 48h intervals, 15µl of culture medium from each drop was replaced by fresh medium. Assessment of developmental and morphological parameters The survival rate of cultured pre-antral follicles was assessed microscopically based on the morphology of the pre-antral follicle every 66
3 other day as described previously [18]. Briefly, follicles were considered as normal which had a central oocyte and several layers of granulose and theca cells and intact basement membrane without any signs of degeneration, such as dark oocyte or cumulus cells. Visible lucent area in the granulosa cell mass around the oocyte considered as antral-like cavity formation. Follicles diameter was measured only in healthy follicles on day 2 and 4 of culturing period by measuring and averaging two perpendicular cross sectional diameters of each follicle at 100 magnification with pre-calibrated ocular micrometer under inverted microscope (Nikon, Tokyo, Japan). Induction of ovulation At twelfth day of cultivation period, ovulation was induced by changing culture medium containing 1.5 IU/ml human chorionic gonadotropin (hcg) h after that, ovulation was assessed and released oocyte were classified as germinal vesicle (GV), germinal vesicle breakdown (GVBD) when the GV was absent, and metaphase II oocytes (MII) when the first polar body was extruded. Statistical analysis Statistical analysis was performed by using SPSS-ver.16 software package (SPSS Inc., Chicago, IL, USA). Data were statically analyzed by one-way analysis of variance (ANOVA) and Tukey's HSD was used as post hoc test. Significant differences were considered at level of P < Each experiment was carried out at least three replicates. RESULTS The mean diameter and rates of developmental parameters of isolated pre-antral follicles derived from vitrified and non-vitrified ovaries with or without pretreatment of CoQ 10 are summarized in Tables I and II. Pre-antral follicles at different stage of development can be seen in Fig1. Every other day, the cultured follicles were morphologically evaluated. During culture period, pre-antral follicles initially became attached to the culture dish through proliferation of their granulosa and theca cells. After the fourth day, the follicles were out from the symmetric mode and form an irregular and diffuse appearance so that it was impossible to measure its diameter. Phenomenon analogous to ovulation occurred at hours after hcg administration (Figure 1). There were no significant difference among the mean diameter of pre-antral follicles of all groups (p<0.05) at the initial time of culturing. While, the diameter of pre-antral follicle is increased progressively during the culture period. As shown in table I, the diameter of isolated pre-antral follicle derived from fresh ovaries (Control) on 2 th and 4 th day of culture period (229 and 290 µm respectively) was significantly higher than those of isolated pre-antral follicles derived from vitrified-warmed ovaries (161 and 178 µm respectively; P<0.05). The mean diameter of the fresh pre-antral follicles in the presence of CoQ 10 on day 2 and 4 (280 and 345µm respectively) was significantly higher in comparison with other groups (P<0.05). The mean diameter of isolated pre-antral follicles derived from vitrified-warmed ovaries with pre-treatment of CoQ 10 at 2 th and 4 th day (187 and 223 µm respectively) were significantly greater than those of isolated preantral follicles derived from vitrified ovaries in the absence of CoQ 10 (178 and 161 µm respectively) while, these were significantly lower than those of pre-antral follicle derived from fresh ovaries (P<0.05). The rates of survival (75%), antrum formation (62%) and ovulation (40%) of the fresh pre-antral follicles were significantly higher than those of pre-antral follicles derived from vitrified-warmed ovaries (48%, 58% and 31% respectively; P<0.05). Whereas, these rates of pre-antral follicles derived from vitrified-warmed ovaries which were cultured in the presence of CoQ 10 (Survival rate; 65%, antrum formation; 58% and ovulation 31%) were similar to those of fresh preantral follicles (P<0.05). Also the highest rates of survival (91%), antrum formation (86%) and ovulation (71%) were obtained from fresh preantral follicles with pre-treatment of CoQ 10 (Table 2). Percentage of MII oocytes resulting from fresh pre-antral follicles (30%) was significantly higher than those of pre-antral follicles derived from vitrified-warmed ovaries (14.0%; P<0.05), although both were statistically lower than that of fresh pre-antral follicles which were cultured in the presence of CoQ 10 (48%; P<0.05). The percentages of MII oocytes were 67
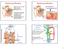
4 similar between pre-antral follicles derived from vitrified-warmed ovaries with CoQ 10 pretreatment (26%) and that of control groups (30%; P<0.05). Table 1. Diameter of cultured pre-antral follicles derived from fresh and vitrified ovaries in the presence of CoQ 10 Groups NO. of Follicles Follicle Diameter (um ± SD) 0 th day 2 th day 4 th day Fresh (Control) ± ±53.1 a 290.6±93.7 a Fresh +CoQ ± ±31.4 b 345.7±56.0 b Vitrified ± ±14.2 c 178.7±17.7 c Vitrified +CoQ ± ±21.6 d 223.7±27.1 d Different superscripts in the same column reflect different levels of significant difference (P<0.05) CoQ 10 : Coenzyme Q 10 Figure1. Graphic representation of the in vitro cultured mouse pre-antral follicles Pre-antral follicle on initial day (A), day 8 (B), day 12 (C), The MII oocyte ovulated after hcg stimulus on day 12 (D) Antral-like cavities are indicated by black arrow. Ovulated MII oocyte indicated by white arrow. Table 2. Maturation rates of cultured pre-antral follicles derived from fresh and vitrified ovaries in the presence of CoQ 10 Antrum Ovulated NO. of Survived Degenerated MI oocytes Groups Formation Follicles Follicles (% ± SD) (% ± SD) (% ± SD) (% ± SD) (% ± SD) MII Oocytes (% ± SD) Fresh (Control) 78 (75.78±3.88) a (24.22±3.88) a (62.00±8.69) a (40.89±5.12) a (11.55±2.89) a (30.67±3.65) a Fresh +CoQ (91.11±3.85) b (8.89±3.85) b (86.67±6.67) b (71.11±7.70) b (22.22±3.85) b (48.89±3.85) b Vitrified 87 (48.80±7.45) c (51.20±7.45) c (44.64±3.90) c (20.77±4.57) c (5.94±3.99) a (14.82±1.49) c Vitrified+CoQ (65.00±6.38) a (35.00±6.38) a (58.33±6.39) a (31.67±11.39) a (5.00±6.38) a (26.67±4.44) a Different superscripts in the same column reflect different levels of significant difference (P<0.05) CoQ 10 : Coenzyme Q 10 ; MI: metaphase I oocyte; MII: metaphase II oocyte. 68
5 DISCUSSION Cryopreservation of ovarian tissue is a promising technology for fertility preservation. But there are many difficulties ahead in this approach. Cryopreservation change the cell membrane, cytoskeletal structure and mitochondrial distribution and depolarization which in turn lead to increasing the production of ROS and inducing OS [19, 20]. It has been shown that mitochondrial defects are accompanying with failures in development of oocyte and embryos [21, 22]. Also, it has been shown that the ROS production of pre-antral follicles derived from vitrified and non-vitrified ovaries during in vitro culture periods had a tendency to be increased, whereas, ROS concentration of fresh pre-antral follicles were significantly lower than those of vitrified samples [18, 23]. It was suggested that mitochondrial electron-transport chain is a major source of ROS production [24]. Production of ROS in the in vitro culture is an unavoidable event, because exogenous factors such as visible light, oxygen concentration, handling and etc. as well as endogenous sources of ROS originating from cellular metabolism induce production ROS [9, 25]. Both enzymatic and non-enzymatic antioxidants form a front line of defense against cell damage caused by ROS [26]. It has been shown that the addition of various natural or synthetic antioxidants prevent or attenuate the OS in in vitro culture condition [10, 17, 18, 23, 27, 28]. In this regard, it has been demonstrated that supplemented medium with sodium selenite improved the rates of survival and developmental competence the pre-antral follicles via reduction of ROS [10]. Also, results from another studies indicated that the presence of alpha lipoic acid (ALA) in pre antral culture medium could enhance the rates of developmental parameters [17, 18]. These observations are in agreement with our results that showed CoQ 10 as a potent antioxidant could improve the rates of survival, antrum formation and MII oocytes of pre-antral follicles derived from vitrified and fresh ovaries after longterm in vitro culture. CoQ 10 as a major cellular antioxidant is located in the mitochondrial respiratory chain [29] which transports electrons from complexes I and II to complex III which in turn lead to the stability of complex III and subsequent production of energy [22]. Whereas, not all aspects of CoQ 10 s mechanisms are completely understood. It has been shown that CoQ 10 can act as a superior antioxidant against OS damages [30]. CoQ 10 plays an important role in prevention of lipid peroxidation. Also CoQ 10 reacts with superoxide and reduce to ubisemiquinone. Other ROS also react with CoQ 10, while the biological importance of these reactions is unclear [30]. The previous results showed that CoQ 10 caused progress of oocyte maturation through changing in pattern of mitochondrial distribution and polarization, the polarity of the membrane and expression of genes that involved in the electron transport chain [15]. It has been demonstrated that administration of CoQ 10 for aged mice leads to increasing the number of ovulated oocytes via improvement of oocyte mitochondrial function [31]. In another study showed that CoQ 10 supplementation in infertile men, increased fertilization rates in the subsequent in vitro fertilization cycle [14]. It has been also found that supplemented media with CoQ 10 increased spermatozoa motility [14]. CoQ 10 intake in men who have idiopathic infertility improved acrosome reaction [32] and fertility rates [33]. CoQ 10 may acts as a potent gene regulator and exert its effects through the induction of gene transcription involved in cell signaling and metabolism [34]. Also, it has been shown that CoQ 10 is directly involved in apoptotic pathway and attributed it to its antioxidant properties [35]. In conclusion, present study demonstrate that supplementation of culture medium with CoQ 10 has positive effects on development of mice preantral follicle. These effects may be due to at least in part to antioxidant properties of CoQ 10. Although more studies are needed to be continue. ACKNOWLEDGEMENTS This study was supported in part by grants from the Damghan University and institute of biological sciences of Damghan University. The authors are grateful for Mr. Abutaleb Koosha for their technical assistance. REFERENCES 69
6 1. Oktay K and Oktem O. Ovarian cryopreservation and transplantation for fertility preservation for medical indications: report of an ongoing experience. Fertility and sterility. 2010;93(3): Shaw J, Oranratnachai A and Trounson A. Fundamental cryobiology of mammalian oocytes and ovarian tissue. Theriogenology. 2000;53(1): Hovatta O. Methods for cryopreservation of human ovarian tissue. Reproductive biomedicine online. 2005;10(6): Bagchi A, Woods EJ and Critser JK. Cryopreservation and vitrification: recent advances in fertility preservation technologies Agarwal A, Said TM, Bedaiwy MA, Banerjee J and Alvarez JG. Oxidative stress in an assisted reproductive techniques setting. Fertility and sterility. 2006;86(3): Gupta S, Sekhon L, Kim Y and Agarwal A. The role of oxidative stress and antioxidants in assisted reproduction. Current Women's Health Reviews. 2010;6(3): Saalu LC. The incriminating role of reactive oxygen species in idiopathic male infertility: an evidence based evaluation. Pak J Biol Sci. 2010;13(9): Hammadeh M, Al Hasani S, Rosenbaum P, Schmidt W and Hammadeh CF. Reactive oxygen species, total antioxidant concentration of seminal plasma and their effect on sperm parameters and outcome of IVF/ICSI patients. Archives of gynecology and obstetrics. 2008;277(6): Agarwal A, Durairajanayagam D and du Plessis SS. Utility of antioxidants during assisted reproductive techniques: an evidence based review. Reprod Biol Endocrinol. 2014;12: Abedelahi A, Salehnia M, Allameh A and Davoodi D. Sodium selenite improves the in vitro follicular development by reducing the reactive oxygen species level and increasing the total antioxidant capacity and glutathione peroxide activity. Human reproduction. 2010;25(4): Frei B, Kim MC and Ames BN. Ubiquinol-10 is an effective lipid-soluble antioxidant at physiological concentrations. Proceedings of the National Academy of Sciences. 1990;87(12): Crane F, Sun I, Crowe R, Alcain F and Löw H. Coenzyme Q 10, plasma membrane oxidase and growth control. Molecular aspects of medicine. 1994;15:s1-s Turi A, Giannubilo SR, Brugè F, Principi F, Battistoni S, Santoni F, et al. Coenzyme Q10 content in follicular fluid and its relationship with oocyte fertilization and embryo grading. Archives of gynecology and obstetrics. 2012;285(4): Lewin A and Lavon H. The effect of coenzyme Q10 on sperm motility and function. Mol Aspects Med. 1997;18 Suppl:S Stojkovic M, Westesen K, Zakhartchenko V, Stojkovic P, Boxhammer K and Wolf E. Coenzyme Q10 in submicron-sized dispersion improves development, hatching, cell proliferation, and adenosine triphosphate content of in vitro-produced bovine embryos. Biology of reproduction. 1999;61(2): Chen SU, Chien CL, Wu MY, Chen TH, Lai SM, Lin CW, et al. Novel direct cover vitrification for cryopreservation of ovarian tissues increases follicle viability and pregnancy capability in mice. Hum Reprod. 2006;21(11): Hatami S, Zavareh S, Salehnia M, Lashkarbolouki T, Ghorbanian MT and Karimi I. The impact of alpha lipoic acid on developmental competence of mouse vitrified pre-antral follicles in comparison to those isolated from vitrified ovaries. Iranian journal of reproductive medicine. 2014;12(1): Talebi A, Zavareh S, Kashani MH, Lashgarbluki T and Karimi I. The effect of alpha lipoic acid on the developmental competence of mouse isolated preantral follicles. J Assist Reprod Genet. 2012;29(2): Zhao XM, Du WH, Wang D, Hao HS, Liu Y, Qin T, et al. Recovery of mitochondrial function and endogenous antioxidant systems in vitrified bovine oocytes during extended in vitro culture. Mol Reprod Dev. 2011;78(12): Zhao XM, Fu XW, Hou YP, Yan CL, Suo L, Wang YP, et al. Effect of vitrification on mitochondrial distribution and membrane potential in mouse two pronuclear (2-PN) 70
7 embryos. Mol Reprod Dev. 2009;76(11): Manipalviratn S, Tong ZB, Stegmann B, Widra E, Carter J and DeCherney A. Effect of vitrification and thawing on human oocyte ATP concentration. Fertil Steril. 2011;95(5): Santos TA, El Shourbagy S and St John JC. Mitochondrial content reflects oocyte variability and fertilization outcome. Fertil Steril. 2006;85(3): Hatami S, Zavareh S, Salehnia M, Lashkarbolouki T and Karimi I. Comparison of oxidative status of mouse pre-antral follicles derived from vitrified whole ovarian tissue and vitrified pre-antral follicles in the presence of alpha lipoic acid. J Obstet Gynaecol Res. 2014;40(6): Lesnefsky EJ, Moghaddas S, Tandler B, Kerner J and Hoppel CL. Mitochondrial dysfunction in cardiac disease: ischemia-- reperfusion, aging, and heart failure. Journal of molecular and cellular cardiology. 2001;33(6): Combelles CM, Gupta S and Agarwal A. Could oxidative stress influence the in-vitro maturation of oocytes? Reproductive biomedicine online. 2009;18(6): Shafaq N. An overview of oxidative stress and antioxidant defensive system Hatami S, Zavareh S, Salehnia M, Lashkarbolouki T, Ghorbanian MT and Karimi I. Total oxidative status of mouse vitrified preantral follicles with pre-treatment of alpha lipoic acid. Iran Biomed J. 2014;18(3): Rahnama A, Zavareh S, Ghorbanian MT and Karimi I. The Effects of camp-elevating Agents and Alpha Lipoic Acid on In Vitro Maturation of Mouse Germinal Vesicle Oocytes. J Reprod Infertil. 2013;14(4): Bentinger M, Brismar K and Dallner G. The antioxidant role of coenzyme Q. Mitochondrion. 2007;7 Suppl:S James AM, Smith RA and Murphy MP. Antioxidant and prooxidant properties of mitochondrial Coenzyme Q. Arch Biochem Biophys. 2004;423(1): Burstein E, Perumalsamy A, Bentov Y, Esfandiari N, Jurisicova A and Casper RF. Coenzyme Q10 supplementation improves ovarian response and mitochondrial function in aged mice. Fertility and Sterility.92(3):S Safarinejad MR. Efficacy of coenzyme Q10 on semen parameters, sperm function and reproductive hormones in infertile men. J Urol. 2009;182(1): Safarinejad MR, Safarinejad S, Shafiei N and Safarinejad S. Effects of the reduced form of coenzyme Q10 (ubiquinol) on semen parameters in men with idiopathic infertility: a double-blind, placebo controlled, randomized study. J Urol. 2012;188(2): Groneberg DA, Kindermann B, Althammer M, Klapper M, Vormann J, Littarru GP, et al. Coenzyme Q10 affects expression of genes involved in cell signalling, metabolism and transport in human CaCo-2 cells. Int J Biochem Cell Biol. 2005;37(6): Crane FL. Biochemical functions of coenzyme Q10. J Am Coll Nutr. 2001;20(6):
Total Oxidative status of Mouse Vitrified Pre-Antral Follicles with Pre-Treatment of Alpha Lipoic Acid
 Iranian Biomedical Journal 18 (3): 181-188 (July 2014) DOI: 10.6091/ibj.12582.2014 Total Oxidative status of Mouse Vitrified Pre-Antral Follicles with Pre-Treatment of Alpha Lipoic Acid Sahar Hatami 1,
Iranian Biomedical Journal 18 (3): 181-188 (July 2014) DOI: 10.6091/ibj.12582.2014 Total Oxidative status of Mouse Vitrified Pre-Antral Follicles with Pre-Treatment of Alpha Lipoic Acid Sahar Hatami 1,
Name: Xueming Zhao. Professional Title: Professor. Animal embryo biotechnology, mainly including in vitro maturation (IVM), in vitro fertilization
 Name: Xueming Zhao Professional Title: Professor Telephone:86-010-62815892 Fax:86-010-62895971 E-mail: zhaoxueming@caas.cn Website: http://www.iascaas.net.cn/yjspy/dsjj/sssds/dwyzyzypz1/62040.htm Research
Name: Xueming Zhao Professional Title: Professor Telephone:86-010-62815892 Fax:86-010-62895971 E-mail: zhaoxueming@caas.cn Website: http://www.iascaas.net.cn/yjspy/dsjj/sssds/dwyzyzypz1/62040.htm Research
Rejuvenation of Gamete Cells; Past, Present and Future
 Rejuvenation of Gamete Cells; Past, Present and Future Denny Sakkas PhD Scientific Director, Boston IVF Waltham, MA, USA Conflict of Interest I have no conflict of interest related to this presentation.
Rejuvenation of Gamete Cells; Past, Present and Future Denny Sakkas PhD Scientific Director, Boston IVF Waltham, MA, USA Conflict of Interest I have no conflict of interest related to this presentation.
Effect of Bovine Follicular Fluid Added to the Maturation Medium on Sperm Penetration in Pig Oocytes Matured In Vitro
 Article Effect of Bovine Follicular Fluid Added to the Maturation Medium on Sperm Penetration in Pig Oocytes Matured In Vitro Abstract Naoki ISOBE Research Associate Graduate School for International Development
Article Effect of Bovine Follicular Fluid Added to the Maturation Medium on Sperm Penetration in Pig Oocytes Matured In Vitro Abstract Naoki ISOBE Research Associate Graduate School for International Development
A comparison of the effects of estrus cow. nuclear maturation of bovine oocytes
 A comparison of the effects of estrus cow serum and fetal calf serum on in vitro nuclear maturation of bovine oocytes J Spiropoulos, SE Long University of Bristol, School of Veterinary Science, Department
A comparison of the effects of estrus cow serum and fetal calf serum on in vitro nuclear maturation of bovine oocytes J Spiropoulos, SE Long University of Bristol, School of Veterinary Science, Department
In Vitro Growth of Mouse Ovarian Preantral Follicles and the Capacity of Their Oocytes to Develop to the Blastocyst Stage
 FULL PAPER Theriogenology In Vitro Growth of Mouse Ovarian Preantral Follicles and the Capacity of Their Oocytes to Develop to the Blastocyst Stage Christopher BISHONGA 1), Yoshiyuki TAKAHASHI 1)*, Seiji
FULL PAPER Theriogenology In Vitro Growth of Mouse Ovarian Preantral Follicles and the Capacity of Their Oocytes to Develop to the Blastocyst Stage Christopher BISHONGA 1), Yoshiyuki TAKAHASHI 1)*, Seiji
CARD HyperOva (Superovulation Reagent for mouse)
 Product manual (Superovulation Reagent for mouse) Cat. No. KYD-010-EX -X5 Size: 5 1 ML Origin Serum of goat, Horse-derived villus gonatropin. Composition 1. Inhibin antiserum (Goat). 2. Equine chorionic
Product manual (Superovulation Reagent for mouse) Cat. No. KYD-010-EX -X5 Size: 5 1 ML Origin Serum of goat, Horse-derived villus gonatropin. Composition 1. Inhibin antiserum (Goat). 2. Equine chorionic
Effects of Preservation of Porcine Oocytes by Dibutyryl Cyclic AMP on in vitro Maturation, Fertilization and Development
 JARQ 45 (3), 295 300 (2011) http://www.jircas.affrc.go.jp of Porcine Oocytes Using dbcamp Effects of of Porcine Oocytes by Dibutyryl Cyclic AMP on in vitro Maturation, Fertilization and Development Dai-ichiro
JARQ 45 (3), 295 300 (2011) http://www.jircas.affrc.go.jp of Porcine Oocytes Using dbcamp Effects of of Porcine Oocytes by Dibutyryl Cyclic AMP on in vitro Maturation, Fertilization and Development Dai-ichiro
The Cytotoxic Effect of Cryoprotective Agents on in vitro Fertilization Rates of Mammalian Oocytes
 Cean A. et al./scientific Papers: Animal Science and Biotechnologies, 2013, 46 (2) The Cytotoxic Effect of Cryoprotective Agents on in vitro Fertilization Rates of Mammalian Oocytes Ada Cean 1,2,*, Ivan
Cean A. et al./scientific Papers: Animal Science and Biotechnologies, 2013, 46 (2) The Cytotoxic Effect of Cryoprotective Agents on in vitro Fertilization Rates of Mammalian Oocytes Ada Cean 1,2,*, Ivan
The use of mitochondrial nutrients to improve IVF. Robert F Casper MD
 The use of mitochondrial nutrients to improve IVF Robert F Casper MD Reproductive Aging Women in the developed world are delaying childbirth for many reasons In the last 10 years, first births for: Women
The use of mitochondrial nutrients to improve IVF Robert F Casper MD Reproductive Aging Women in the developed world are delaying childbirth for many reasons In the last 10 years, first births for: Women
Downloaded from ismj.bpums.ac.ir at 22: on Friday March 22nd 2019
 - ( ) - Bcl-2 Bax * Bcl-2 Bax :.. : α-mem (VNA) (NVA) (NVNA). Bcl-2 Bax (VA) /±/ /±/ /±/ VA VNA NVA NVNA :. /±/ /±/ /±/ /±/ /±/.(p=/ p=/ ) VNA NVNA NVA NVA Bax.(p=/).(p=/). in vitro :. Bax Bcl-2 Bax :
- ( ) - Bcl-2 Bax * Bcl-2 Bax :.. : α-mem (VNA) (NVA) (NVNA). Bcl-2 Bax (VA) /±/ /±/ /±/ VA VNA NVA NVNA :. /±/ /±/ /±/ /±/ /±/.(p=/ p=/ ) VNA NVNA NVA NVA Bax.(p=/).(p=/). in vitro :. Bax Bcl-2 Bax :
IVM in PCOS patients. Introduction (1) Introduction (2) Michael Grynberg René Frydman
 IVM in PCOS patients Michael Grynberg René Frydman Department of Obstetrics and Gynecology A. Beclere Hospital, Clamart, France Maribor, Slovenia, 27-28 February 2009 Introduction (1) IVM could be a major
IVM in PCOS patients Michael Grynberg René Frydman Department of Obstetrics and Gynecology A. Beclere Hospital, Clamart, France Maribor, Slovenia, 27-28 February 2009 Introduction (1) IVM could be a major
Supplementation of culture media with vitamin E improves mouse antral follicle maturation and embryo development from vitrified ovarian tissue
 doi:10.1111/jog.12933 J. Obstet. Gynaecol. Res. Vol. 42, No. 5: 526 535, May 2016 Supplementation of culture media with vitamin E improves mouse antral follicle maturation and embryo development from vitrified
doi:10.1111/jog.12933 J. Obstet. Gynaecol. Res. Vol. 42, No. 5: 526 535, May 2016 Supplementation of culture media with vitamin E improves mouse antral follicle maturation and embryo development from vitrified
Relationship between Energy Expenditure Related Factors and Oxidative Stress in Follicular Fluid
 Original Article Relationship between Energy Expenditure Related Factors and Oxidative Stress in Follicular Fluid Abstract This study evaluated the impact of body mass index (BMI), total calorie intake
Original Article Relationship between Energy Expenditure Related Factors and Oxidative Stress in Follicular Fluid Abstract This study evaluated the impact of body mass index (BMI), total calorie intake
In Vitro Culture of Mouse Preantral Follicles Using Membrane Inserts and Developmental Competence of In Vitro Ovulated Oocytes
 Journal of Reproduction and Development, Vol. 50, No. 5, 2004 Research Note In Vitro Culture of Mouse Preantral Follicles Using Membrane Inserts and Developmental Competence of In Vitro Ovulated Oocytes
Journal of Reproduction and Development, Vol. 50, No. 5, 2004 Research Note In Vitro Culture of Mouse Preantral Follicles Using Membrane Inserts and Developmental Competence of In Vitro Ovulated Oocytes
Boram Kim and Sanghoon Lee Department of Obstetrics and Gynecology, Korea University College of Medicine, Seoul, Korea
 Comparison of slow freezing versus vitrification for human ovarian tissue cryopreservation and xenotransplantation Boram Kim and Sanghoon Lee Department of Obstetrics and Gynecology, Korea University College
Comparison of slow freezing versus vitrification for human ovarian tissue cryopreservation and xenotransplantation Boram Kim and Sanghoon Lee Department of Obstetrics and Gynecology, Korea University College
Faculty of Biological Sciences, Tarbiat Modares University, Tehran, Iran. Received 23 January 2013; revised 10 April 2013; accepted 13 April 2013
 Iranian Biomedical Journal 17 (3): 123-128 (July 2013) DOI: 10.6091/ibj.1199.2013 The Effect of Vitrification and in vitro Culture on the Adenosine Triphosphate Content and Mitochondrial Distribution of
Iranian Biomedical Journal 17 (3): 123-128 (July 2013) DOI: 10.6091/ibj.1199.2013 The Effect of Vitrification and in vitro Culture on the Adenosine Triphosphate Content and Mitochondrial Distribution of
Cryotop Vitrification Affects Oocyte Quality and Embryo Developmental Potential
 Cronicon OPEN ACCESS Ling Jia*, Bo Xu*, Yu-sheng Liu and Xian-hong Tong Center for Reproductive Medicine, Anhui Provincial Hospital Affiliated to Anhui Medical University, China *These authors contributed
Cronicon OPEN ACCESS Ling Jia*, Bo Xu*, Yu-sheng Liu and Xian-hong Tong Center for Reproductive Medicine, Anhui Provincial Hospital Affiliated to Anhui Medical University, China *These authors contributed
Microinsemination (Intracytoplasmic Sperm Injection) Microinsemination schedule. 1. Preparation of mediums
 Microinsemination (Intracytoplasmic Sperm Injection) Masumi Hirabayashi Section of Mammalian Transgenesis, Center for Genetic Analysis of Behavior, National Institute for Physiological Sciences, National
Microinsemination (Intracytoplasmic Sperm Injection) Masumi Hirabayashi Section of Mammalian Transgenesis, Center for Genetic Analysis of Behavior, National Institute for Physiological Sciences, National
Protocol for embryo vitrification using open pulled straws. Introduction. Reagents. Materials
 Protocol for embryo vitrification using open pulled straws Froylan Sosa and Peter J Hansen Department of Animal Sciences, University of Florida Introduction This vitrification protocol is a slight modification
Protocol for embryo vitrification using open pulled straws Froylan Sosa and Peter J Hansen Department of Animal Sciences, University of Florida Introduction This vitrification protocol is a slight modification
RLI Mouse Vitrification Media Kit
 RLI Mouse Vitrification Media Kit Product Description RLI Vitrification Media Kit (Catalog#: RLI Vitri-Cooling 01, RLI Vitri-Warming 01, RLI Vitri Complete Kit 01) enables ultra-rapid cooling and recovery
RLI Mouse Vitrification Media Kit Product Description RLI Vitrification Media Kit (Catalog#: RLI Vitri-Cooling 01, RLI Vitri-Warming 01, RLI Vitri Complete Kit 01) enables ultra-rapid cooling and recovery
Article Effect of gonadotrophin priming on in-vitro maturation of oocytes collected from women at risk of OHSS
 RBMOnline - Vol 13. No 3. 2006 340 348 Reproductive BioMedicine Online; www.rbmonline.com/article/2328 on web 12 June 2006 Article Effect of gonadotrophin priming on in-vitro maturation of oocytes collected
RBMOnline - Vol 13. No 3. 2006 340 348 Reproductive BioMedicine Online; www.rbmonline.com/article/2328 on web 12 June 2006 Article Effect of gonadotrophin priming on in-vitro maturation of oocytes collected
Effect of the Well of the Well (WOW) System on In Vitro Culture for Porcine Embryos after Intracytoplasmic Sperm Injection
 Journal of Reproduction and Development, Vol. 51, No. 4, 2005 Technical Note Effect of the Well of the Well (WOW) System on In Vitro Culture for Porcine Embryos after Intracytoplasmic Sperm Injection Mikiko
Journal of Reproduction and Development, Vol. 51, No. 4, 2005 Technical Note Effect of the Well of the Well (WOW) System on In Vitro Culture for Porcine Embryos after Intracytoplasmic Sperm Injection Mikiko
Effect of sucrose and propylene glycol on the vitrification of sheep oocytes
 Journal of Cell and Animal Biology Vol. 7 (3), pp. 25-30, March 2013 Available online at http://www.academicjournals.org/jcab DOI: 10.5897/JCAB12.033 ISSN 1996-0867 2013 Academic Journals Full Length Research
Journal of Cell and Animal Biology Vol. 7 (3), pp. 25-30, March 2013 Available online at http://www.academicjournals.org/jcab DOI: 10.5897/JCAB12.033 ISSN 1996-0867 2013 Academic Journals Full Length Research
Journal of Ningxia Medical University DMEM F12 GIBCO. 2mm mm 4mm 2mm vitrification group 10mm. 4mm 2mm VG 3
 34 9 2012 9 Journal of Ningxia Medical University 869 1674-6309 2012 09-0869 - 03 FSH 1 2 1 1 1 1 1 1 3 1. 750004 2. 054000 3. 750001 FSH FSH 20 5mm 4mm 2mm 10mm 4mm 2mm 10μg FSH ml - 1 4. 5h FSH 1 2 -
34 9 2012 9 Journal of Ningxia Medical University 869 1674-6309 2012 09-0869 - 03 FSH 1 2 1 1 1 1 1 1 3 1. 750004 2. 054000 3. 750001 FSH FSH 20 5mm 4mm 2mm 10mm 4mm 2mm 10μg FSH ml - 1 4. 5h FSH 1 2 -
Analysis of post-warming degeneration & apoptosis following porcine ovarian tissue vitrification using the ohio-cryo device
 Analysis of post-warming degeneration & apoptosis following porcine ovarian tissue vitrification using the ohio-cryo device e-poster: 363 Congress: ESHRE 2008 Type: Scientific poster Topic: ART, laboratory:
Analysis of post-warming degeneration & apoptosis following porcine ovarian tissue vitrification using the ohio-cryo device e-poster: 363 Congress: ESHRE 2008 Type: Scientific poster Topic: ART, laboratory:
Effect of In Vitro Maturation Technique and Alpha Lipoic Acid Supplementation on Oocyte Maturation Rate: Focus on Oxidative Status of Oocytes
 Original Article Effect of In Vitro Maturation Technique and Alpha Lipoic Acid Supplementation on Oocyte Maturation Rate: Focus on Oxidative Status of Oocytes Saeed Zavareh, Ph.D. 1, 2 *, Isaac Karimi,
Original Article Effect of In Vitro Maturation Technique and Alpha Lipoic Acid Supplementation on Oocyte Maturation Rate: Focus on Oxidative Status of Oocytes Saeed Zavareh, Ph.D. 1, 2 *, Isaac Karimi,
In-vitro maturation of camel oocytes using different media and sera
 In-vitro maturation of camel oocytes using different media and sera A. E. B. Zeidan 1, M. E. Hammad 2, Sh. A. Gabr 2, M. H. Farouk 3*, Sh. M. Shamiah 1, E. A. A. Ahmadi 1 and W. M. A. Nagy 1. 1 Dept. Camel
In-vitro maturation of camel oocytes using different media and sera A. E. B. Zeidan 1, M. E. Hammad 2, Sh. A. Gabr 2, M. H. Farouk 3*, Sh. M. Shamiah 1, E. A. A. Ahmadi 1 and W. M. A. Nagy 1. 1 Dept. Camel
INFRAFRONTIER-I3 - Cryopreservation training course. Hosted by the Frozen Embryo and Sperm Archive, MRC - Harwell
 Hosted by the Frozen Embryo and Sperm Archive, MRC - Harwell IVF recovery procedure incorporting methyl-β-cyclodextrin and reduced glutathione This protocol is based on the work published by Takeo et al.,
Hosted by the Frozen Embryo and Sperm Archive, MRC - Harwell IVF recovery procedure incorporting methyl-β-cyclodextrin and reduced glutathione This protocol is based on the work published by Takeo et al.,
Toxic Effect of Cryoprotectants on Embryo Development in a Murine Model
 : 31 1 2004 Kor J Fertil Steril, Vol 31, No 1, 2004, 3 1 2,, 1 2 3 3 3 3 3 3 3* Toxic Effect of Cryoprotectants on Embryo Development in a Murine Model Kwan Cheal Yang 1, Hee-Gyoo Kang 2,Hoi-ChangLee 3,
: 31 1 2004 Kor J Fertil Steril, Vol 31, No 1, 2004, 3 1 2,, 1 2 3 3 3 3 3 3 3* Toxic Effect of Cryoprotectants on Embryo Development in a Murine Model Kwan Cheal Yang 1, Hee-Gyoo Kang 2,Hoi-ChangLee 3,
Adenosine Triphosphate Content in Human Unfertilized Oocytes, Undivided Zygotes and Embryos Unsuitable for Transfer or Cryopreservation
 The Journal of International Medical Research 2012; 40: 734 739 Adenosine Triphosphate Content in Human Unfertilized Oocytes, Undivided Zygotes and Embryos Unsuitable for Transfer or Cryopreservation J
The Journal of International Medical Research 2012; 40: 734 739 Adenosine Triphosphate Content in Human Unfertilized Oocytes, Undivided Zygotes and Embryos Unsuitable for Transfer or Cryopreservation J
Cetrorelix preserves follicular viability in Cyclophosphamid-induced ovarian toxicity
 International Journal of Research in Applied and Basic Medical Sciences 2015; 1(1): 56-60 Cetrorelix preserves follicular viability in Cyclophosphamid-induced ovarian toxicity Mohammadnejad D 1, Tayefi-nasrabadi
International Journal of Research in Applied and Basic Medical Sciences 2015; 1(1): 56-60 Cetrorelix preserves follicular viability in Cyclophosphamid-induced ovarian toxicity Mohammadnejad D 1, Tayefi-nasrabadi
Effect of Warming on the Survivability and Fertilizability of Vitrified Matured Bovine Oocytes
 International Journal of Agricultural Technology 2014 Vol. 10(1):49-58 Available online http://www.ijat-aatsea.com ISSN 2630-0192 (Online) Fungal Diversity Effect of Warming on the Survivability and Fertilizability
International Journal of Agricultural Technology 2014 Vol. 10(1):49-58 Available online http://www.ijat-aatsea.com ISSN 2630-0192 (Online) Fungal Diversity Effect of Warming on the Survivability and Fertilizability
OOPLASM CRYOPRESERVATION: OVARIAN FRAGMENTS VERSUS OOCYTES ALONE
 2018 NPPC ISSN 1337-9984 OOPLASM CRYOPRESERVATION: OVARIAN FRAGMENTS VERSUS OOCYTES ALONE A. V. MAKAREVICH*, L. OLEXIKOVA, M. FÖLDEŠIOVÁ, J. PIVKO, E. KUBOVIČOVÁ NPPC Research Institute for Animal Production
2018 NPPC ISSN 1337-9984 OOPLASM CRYOPRESERVATION: OVARIAN FRAGMENTS VERSUS OOCYTES ALONE A. V. MAKAREVICH*, L. OLEXIKOVA, M. FÖLDEŠIOVÁ, J. PIVKO, E. KUBOVIČOVÁ NPPC Research Institute for Animal Production
Ultrarapid freezing of early cleavage stage human embryos and eight-cell mouse embryos*
 FERTILITY AND STERILITY Copyright 1988 The American Fertility Society Printed in U.S.A. Ultrarapid freezing of early cleavage stage human embryos and eight-cell mouse embryos* Alan Trounson, Ph.D.t:!:
FERTILITY AND STERILITY Copyright 1988 The American Fertility Society Printed in U.S.A. Ultrarapid freezing of early cleavage stage human embryos and eight-cell mouse embryos* Alan Trounson, Ph.D.t:!:
Vitrification of reproductive cells: The next breakthrough in ART? Department of Obstetrics and Gynaecology University of Aberdeen
 Vitrification of reproductive cells: The next breakthrough in ART? Maureen J Wood PhD Department of Obstetrics and Gynaecology University of Aberdeen breakthrough Some signal achievement in scientific
Vitrification of reproductive cells: The next breakthrough in ART? Maureen J Wood PhD Department of Obstetrics and Gynaecology University of Aberdeen breakthrough Some signal achievement in scientific
DMBA acts on cumulus cells to desynchronize nuclear and cytoplasmic maturation of pig oocytes
 DMBA acts on cumulus cells to desynchronize nuclear and cytoplasmic maturation of pig oocytes Zhi-Qiang Song 1, Xuan Li 1, Yan-Kui Wang 1, Zhi-Qiang Du 1,2*, Cai-Xia Yang 1,2* Supplementary information
DMBA acts on cumulus cells to desynchronize nuclear and cytoplasmic maturation of pig oocytes Zhi-Qiang Song 1, Xuan Li 1, Yan-Kui Wang 1, Zhi-Qiang Du 1,2*, Cai-Xia Yang 1,2* Supplementary information
Iranian Journal of Reproductive Medicine Vol.7. No.2. pp: 45-52, Spring 2009
 Iranian Journal of Reproductive Medicine Vol.7. No.2. pp: 45-52, Spring 2009 Comparison of ultrastructure and morphology of mouse ovarian follicles after conventional and direct cover vitrification using
Iranian Journal of Reproductive Medicine Vol.7. No.2. pp: 45-52, Spring 2009 Comparison of ultrastructure and morphology of mouse ovarian follicles after conventional and direct cover vitrification using
How to Use ivitri - Straw and ivitri - EZ system? Manufacture Instruction
 How to Use ivitri - Straw and ivitri - EZ system? Manufacture Instruction The Magic Lotus Leaf - Nature's Nanotechnology ivitri design http://www.p2i.com/blogs/articles /magic_lotus_leaf_natures_nano technology
How to Use ivitri - Straw and ivitri - EZ system? Manufacture Instruction The Magic Lotus Leaf - Nature's Nanotechnology ivitri design http://www.p2i.com/blogs/articles /magic_lotus_leaf_natures_nano technology
How to Use ivitri - Straw system? Manufacture Instruction
 How to Use ivitri - Straw system? Manufacture Instruction Information of ivitri -Straw ivitri is individual Vitrification Device, called ivitri. ivitri -Straw is US patent and patent pending, and registered
How to Use ivitri - Straw system? Manufacture Instruction Information of ivitri -Straw ivitri is individual Vitrification Device, called ivitri. ivitri -Straw is US patent and patent pending, and registered
Use of in vitro maturation for fertility preservation
 Use of in vitro maturation for fertility preservation G. Arroyo Servei de Medicina de la Reproducció Departament d Obstetrícia, Ginecologia i Reproducció INSTITUT UNIVERSITARI DEXEUS MEDICAL STRATEGY TO
Use of in vitro maturation for fertility preservation G. Arroyo Servei de Medicina de la Reproducció Departament d Obstetrícia, Ginecologia i Reproducció INSTITUT UNIVERSITARI DEXEUS MEDICAL STRATEGY TO
Introduction. Key Message AOGS MAIN RESEARCH ARTICLE HELLE N. WILKEN-JENSEN, STINE G. KRISTENSEN, JANNI V. JEPPESEN & CLAUS YDING ANDERSEN.
 A C TA Obstetricia et Gynecologica AOGS MAIN RESEARCH ARTICLE Developmental competence of oocytes isolated from surplus medulla tissue in connection with cryopreservation of ovarian tissue for fertility
A C TA Obstetricia et Gynecologica AOGS MAIN RESEARCH ARTICLE Developmental competence of oocytes isolated from surplus medulla tissue in connection with cryopreservation of ovarian tissue for fertility
LOW RESPONDERS. Poor Ovarian Response, Por
 LOW RESPONDERS Poor Ovarian Response, Por Patients with a low number of retrieved oocytes despite adequate ovarian stimulation during fertility treatment. Diagnosis Female About Low responders In patients
LOW RESPONDERS Poor Ovarian Response, Por Patients with a low number of retrieved oocytes despite adequate ovarian stimulation during fertility treatment. Diagnosis Female About Low responders In patients
FERTILITY PRESERVATION. Juergen Eisermann, M.D., F.A.C.O.G South Florida Institute for Reproductive Medicine South Miami Florida
 FERTILITY PRESERVATION Juergen Eisermann, M.D., F.A.C.O.G South Florida Institute for Reproductive Medicine South Miami Florida 1 2 3 4 Oocyte Cryopreservation Experimental option Offer to single cancer
FERTILITY PRESERVATION Juergen Eisermann, M.D., F.A.C.O.G South Florida Institute for Reproductive Medicine South Miami Florida 1 2 3 4 Oocyte Cryopreservation Experimental option Offer to single cancer
Masashi NAGANO 1, 2, Eufrocina P. ATABAY 1, 3, Edwin C ATABAY 1, 3, Mitsugu HISHINUMA 2, Seiji KATAGIRI 1. and Yoshiyuki TAKAHASHI 1 1
 Biomedical Research 28 (3) 53-60, 2007 Effects of isolation method and pre-treatment with ethylene glycol or raffinose before vitrification on in vitro viability of mouse preantral follicles Masashi NAGANO,
Biomedical Research 28 (3) 53-60, 2007 Effects of isolation method and pre-treatment with ethylene glycol or raffinose before vitrification on in vitro viability of mouse preantral follicles Masashi NAGANO,
Rescue IVF protocol for legacy stock
 Rescue IVF protocol for legacy stock Sperm thawing/ivf protocol for MTG sperm samples (80ul per straw) from straw and conventional CPA from Vial (100ml per vial) This protocol is based on methods developed
Rescue IVF protocol for legacy stock Sperm thawing/ivf protocol for MTG sperm samples (80ul per straw) from straw and conventional CPA from Vial (100ml per vial) This protocol is based on methods developed
Improved human oocyte development after vitrification: a comparison of thawing methods
 FERTILITY AND STERILITY VOL. 72, NO. 1, JULY 1999 Copyright 1999 American Society for Reproductive Medicine Published by Elsevier Science Inc. Printed on acid-free paper in U.S.A. Improved human oocyte
FERTILITY AND STERILITY VOL. 72, NO. 1, JULY 1999 Copyright 1999 American Society for Reproductive Medicine Published by Elsevier Science Inc. Printed on acid-free paper in U.S.A. Improved human oocyte
In vitro growth of mouse preantral follicles: effect of animal age and stem cell factor/insulin-like growth factor supplementation
 ORIGINAL ARTICLE pissn 2233-8233 eissn 2233-8241 Clin Exp Reprod Med 2012;39(3):107-113 In vitro growth of mouse preantral follicles: effect of animal age and stem cell factor/insulin-like growth factor
ORIGINAL ARTICLE pissn 2233-8233 eissn 2233-8241 Clin Exp Reprod Med 2012;39(3):107-113 In vitro growth of mouse preantral follicles: effect of animal age and stem cell factor/insulin-like growth factor
Support for Fertility and Infertility Treatment
 Support for Fertility and Infertility Treatment Obesity causes an alteration to the hormones associated with women fertility Many different studies have concluded that obesity is related to impaired fertility.
Support for Fertility and Infertility Treatment Obesity causes an alteration to the hormones associated with women fertility Many different studies have concluded that obesity is related to impaired fertility.
Induction of the human sperm acrosome reaction by human oocytes*
 FERTILITY AND STERILITY Copyright C> 1988 The American Fertility Society Vol. 50, No.6, December 1988 Printed in U.S.A. Induction of the human sperm acrosome reaction by human oocytes* Christopher J. De
FERTILITY AND STERILITY Copyright C> 1988 The American Fertility Society Vol. 50, No.6, December 1988 Printed in U.S.A. Induction of the human sperm acrosome reaction by human oocytes* Christopher J. De
Are all-freeze cycles & frozen-thawed embryo transfers improving IVF outcomes?
 Are all-freeze cycles & frozen-thawed embryo transfers improving IVF outcomes? Andrea Weghofer Foundation for Reproductive Medicine 2017 New York, November 16-19 Conflict of interest No relevant financial
Are all-freeze cycles & frozen-thawed embryo transfers improving IVF outcomes? Andrea Weghofer Foundation for Reproductive Medicine 2017 New York, November 16-19 Conflict of interest No relevant financial
2013 Sep.; 24(3):
 Journal of Reproduction & Contraception doi: 10.7669/j.issn.1001-7844.2013.03.0151 2013 Sep.; 24(3):151-158 E-mail: randc_journal@163.com Reducing the Trigger Dose of Human Chorionic Gonadotrophin Does
Journal of Reproduction & Contraception doi: 10.7669/j.issn.1001-7844.2013.03.0151 2013 Sep.; 24(3):151-158 E-mail: randc_journal@163.com Reducing the Trigger Dose of Human Chorionic Gonadotrophin Does
Role of Antioxidants in Assisted Reproductive Techniques
 pissn: 2287-4208 / eissn: 2287-4690 World J Mens Health 2017 August 35(2): 77-93 https://doi.org/10.5534/wjmh.2017.35.2.77 Review Article Role of Antioxidants in Assisted Reproductive Techniques Ashok
pissn: 2287-4208 / eissn: 2287-4690 World J Mens Health 2017 August 35(2): 77-93 https://doi.org/10.5534/wjmh.2017.35.2.77 Review Article Role of Antioxidants in Assisted Reproductive Techniques Ashok
To describe the procedure used for piezo-activated mouse intracellular sperm injection (ICSI) in mice.
 1.0 Purpose: To describe the procedure used for piezo-activated mouse intracellular sperm injection (ICSI) in mice. Useful References: Kimura, Y & Yanagimuach1 R (1995) Intracytoplasmic sperm injection
1.0 Purpose: To describe the procedure used for piezo-activated mouse intracellular sperm injection (ICSI) in mice. Useful References: Kimura, Y & Yanagimuach1 R (1995) Intracytoplasmic sperm injection
Effects of Centrifugation and Lipid Removal on the Cryopreservation of in Vitro Produced Bovine Embryos at the Eight-Cell Stage
 CRYOBIOLOGY 36, 206 212 (1998) ARTICLE NO. CY982077 Effects of Centrifugation and Lipid Removal on the Cryopreservation of in Vitro Produced Bovine Embryos at the Eight-Cell Stage M. Murakami,* T. Otoi,
CRYOBIOLOGY 36, 206 212 (1998) ARTICLE NO. CY982077 Effects of Centrifugation and Lipid Removal on the Cryopreservation of in Vitro Produced Bovine Embryos at the Eight-Cell Stage M. Murakami,* T. Otoi,
I. ART PROCEDURES. A. In Vitro Fertilization (IVF)
 DFW Fertility Associates ASSISTED REPRODUCTIVE TECHNOLOGY (ART) Welcome to DFW Fertility Associates/ Presbyterian-Harris Methodist Hospital ARTS program. This document provides an overview of treatment
DFW Fertility Associates ASSISTED REPRODUCTIVE TECHNOLOGY (ART) Welcome to DFW Fertility Associates/ Presbyterian-Harris Methodist Hospital ARTS program. This document provides an overview of treatment
Evaluation of ovarian response prediction according to age and serum AMH levels in IVF cycles: a retrospective analysis
 International Journal of Reproduction, Contraception, Obstetrics and Gynecology Pillai SM et al. Int J Reprod Contracept Obstet Gynecol. 2017 Aug;6(8):3306-3310 www.ijrcog.org DOI: http://dx.doi.org/10.18203/2320-1770.ijrcog20173190
International Journal of Reproduction, Contraception, Obstetrics and Gynecology Pillai SM et al. Int J Reprod Contracept Obstet Gynecol. 2017 Aug;6(8):3306-3310 www.ijrcog.org DOI: http://dx.doi.org/10.18203/2320-1770.ijrcog20173190
Ovarian Tissue Vitrification as a Method for Ovarian Preservation in Women with Cancer: An Analysis of Granulose Cell Apoptosis
 The 6th Congress of the Asia Pacific Initiative on Reproduction (ASPIRE 2016), Volume 2016 Conference Paper Ovarian Tissue Vitrification as a Method for Ovarian Preservation in Women with Cancer: An Analysis
The 6th Congress of the Asia Pacific Initiative on Reproduction (ASPIRE 2016), Volume 2016 Conference Paper Ovarian Tissue Vitrification as a Method for Ovarian Preservation in Women with Cancer: An Analysis
EFFECT OF CoQ10 ADDITION TO MATURATION MEDIUM AFTER VITRIFICATION OF BOVINE OOCYTES
 I.J.S.N., VOL.6 (1) 215: 4-44 ISSN 2229 6441 EFFECT OF CoQ1 ADDITION TO MATURATION MEDIUM AFTER VITRIFICATION OF BOVINE OOCYTES 1 Abdulhasan, M.A.K., 2 Fakhrildin, M.B.M.R. & 3 Shubber, M.H. 1 College
I.J.S.N., VOL.6 (1) 215: 4-44 ISSN 2229 6441 EFFECT OF CoQ1 ADDITION TO MATURATION MEDIUM AFTER VITRIFICATION OF BOVINE OOCYTES 1 Abdulhasan, M.A.K., 2 Fakhrildin, M.B.M.R. & 3 Shubber, M.H. 1 College
Maturation and Freezing of Bovine Oocytes
 Maturation and Freezing of Bovine Oocytes D. Mapes and M. E. Wells Story in Brief Immature bovine oocytes were aspirated from small to medium size follicles of bovine ovaries by needle and syringe. The
Maturation and Freezing of Bovine Oocytes D. Mapes and M. E. Wells Story in Brief Immature bovine oocytes were aspirated from small to medium size follicles of bovine ovaries by needle and syringe. The
Effects of Cryopreservation on the Ultrastructure of Human Testicular Sperm
 Journal of Reproduction & Contraception (2005) 16 (4):195-200 ORIGINAL PAPER Effects of Cryopreservation on the Ultrastructure of Human Testicular Sperm Xin-qiang LAI 1, Wei-jie ZHU 2, Jing LI 3, Fu-xing
Journal of Reproduction & Contraception (2005) 16 (4):195-200 ORIGINAL PAPER Effects of Cryopreservation on the Ultrastructure of Human Testicular Sperm Xin-qiang LAI 1, Wei-jie ZHU 2, Jing LI 3, Fu-xing
FERTIUP PM 1 ml / 0.5 ml - CARD MEDIUM Set
 Product manual FERTIUP PM 1 ml / 0.5 ml - CARD MEDIUM Set Cat. No. KYD-004-EX Size: 1 SET KYD-005-EX 1 SET Department of Reproductive Engineering, Center for Animal Resources and Development, Kumamoto
Product manual FERTIUP PM 1 ml / 0.5 ml - CARD MEDIUM Set Cat. No. KYD-004-EX Size: 1 SET KYD-005-EX 1 SET Department of Reproductive Engineering, Center for Animal Resources and Development, Kumamoto
THE EFFECT OF VITRIFICATION OF OOCYTES FOR MICE AND EXAMINING SURVIVED OOCYTE PERCENTAGE
 : 376-390 ISSN: 2277 4998 THE EFFECT OF VITRIFICATION OF OOCYTES FOR MICE AND EXAMINING SURVIVED OOCYTE PERCENTAGE FATEMEH FAZELI SHORAKI Fazali101.f@gmail.com MOHAMMAD MEHDI AKHONDI Akhondi@avicenna.ac.ir
: 376-390 ISSN: 2277 4998 THE EFFECT OF VITRIFICATION OF OOCYTES FOR MICE AND EXAMINING SURVIVED OOCYTE PERCENTAGE FATEMEH FAZELI SHORAKI Fazali101.f@gmail.com MOHAMMAD MEHDI AKHONDI Akhondi@avicenna.ac.ir
Relation between Level of Total Antioxidant Capacity and Semen Quality in Male Infertility
 Relation between Level of Total Antioxidant Capacity and Semen Quality in Male Infertility Ahmed M. A. Tahoun, Tarek M. Emran, Amr M. Al-Kharasawy and Ahmed A. Abo Nar Department of Clinical Pathology,
Relation between Level of Total Antioxidant Capacity and Semen Quality in Male Infertility Ahmed M. A. Tahoun, Tarek M. Emran, Amr M. Al-Kharasawy and Ahmed A. Abo Nar Department of Clinical Pathology,
ORIGINAL ARTICLE. Introduction. Min Kyung Kim 1,2, Hyun Sun Kong 2,3, Hye Won Youm 2, Byung Chul Jee 2,3
 ORIGINAL ARTICLE pissn 2233-8233 eissn 2233-8241 Clin Exp Reprod Med 2017;44(1):8-14 Effects of supplementation with antifreeze proteins on the follicular integrity of vitrified-warmed mouse ovaries: Comparison
ORIGINAL ARTICLE pissn 2233-8233 eissn 2233-8241 Clin Exp Reprod Med 2017;44(1):8-14 Effects of supplementation with antifreeze proteins on the follicular integrity of vitrified-warmed mouse ovaries: Comparison
Mouse sperm extraction:
 Mouse sperm extraction: This method of extraction is used for acrosome reaction assays, immunocytochemistry and biochemical assays. Collect two cauda epidydimus from one male, cut them 5 times and place
Mouse sperm extraction: This method of extraction is used for acrosome reaction assays, immunocytochemistry and biochemical assays. Collect two cauda epidydimus from one male, cut them 5 times and place
Interpreting follicular Progesterone: Late follicular Progesterone to Estradiol ratio is not influenced by protocols or gonadotropins used
 Interpreting follicular Progesterone: Late follicular Progesterone to Estradiol ratio is not influenced by protocols or gonadotropins used Ellenbogen A., M.D., Shalom-Paz E., M.D, Asalih N., M.D, Samara
Interpreting follicular Progesterone: Late follicular Progesterone to Estradiol ratio is not influenced by protocols or gonadotropins used Ellenbogen A., M.D., Shalom-Paz E., M.D, Asalih N., M.D, Samara
Sperm production. Sperm production. Meiosis. Mitosis. The cells of Leydig in testes secrete
 Sperm production Ductus deferens Epididymis The cells of Leydig in testes secrete Seminiferous testosterone (T) tubules T secreted at puberty produces 2 o sex characteristics, spermatogenesis, & maintain
Sperm production Ductus deferens Epididymis The cells of Leydig in testes secrete Seminiferous testosterone (T) tubules T secreted at puberty produces 2 o sex characteristics, spermatogenesis, & maintain
Sperm production. Sperm production. Controlling sperm production. Meiosis. Mitosis. The cells of Leydig in testes secrete
 Ductus deferens Sperm production Epididymis The cells of Leydig in testes secrete Seminiferous testosterone (T) tubules T secreted at puberty produces 2 o sex characteristics, spermatogenesis, & maintain
Ductus deferens Sperm production Epididymis The cells of Leydig in testes secrete Seminiferous testosterone (T) tubules T secreted at puberty produces 2 o sex characteristics, spermatogenesis, & maintain
IVF: PAST, PRESENT AND FUTURE
 IVF: PAST, PRESENT AND FUTURE Mark Larman Chief Scientific Officer 1 HISTORY OF IVF IVF first achieved with rabbits in 1959 IVF with human gametes - pioneered by Robert Edwards and Patrick Steptoe during
IVF: PAST, PRESENT AND FUTURE Mark Larman Chief Scientific Officer 1 HISTORY OF IVF IVF first achieved with rabbits in 1959 IVF with human gametes - pioneered by Robert Edwards and Patrick Steptoe during
Evaluation of Serial Thawing-Refreezing on Human Spermatozoa Resistance Using Cryovials and Straws
 Original Article Evaluation of Serial Thawing-Refreezing on Human Spermatozoa Resistance Using Cryovials and Straws Fatemeh Ghasemian, M.Sc. 1, 2, Roya Faraji, M.D. 2, Mohaddese Mohammadi Sardoo, M.Sc.
Original Article Evaluation of Serial Thawing-Refreezing on Human Spermatozoa Resistance Using Cryovials and Straws Fatemeh Ghasemian, M.Sc. 1, 2, Roya Faraji, M.D. 2, Mohaddese Mohammadi Sardoo, M.Sc.
IN VITRO MATURATION OF HUMAN OOCYTES
 REVIEW ARTICLE IN VITRO MATURATION OF HUMAN OOCYTES Yu-Hung Lin 1,2, Jiann-Loung Hwang 1,3 * 1 Department of Obstetrics and Gynecology, Shin Kong Wu Ho-Su Memorial Hospital, 2 School of Medicine, Fu Jen
REVIEW ARTICLE IN VITRO MATURATION OF HUMAN OOCYTES Yu-Hung Lin 1,2, Jiann-Loung Hwang 1,3 * 1 Department of Obstetrics and Gynecology, Shin Kong Wu Ho-Su Memorial Hospital, 2 School of Medicine, Fu Jen
Oocyte maturation. A.Trounson 1 ' 3, C.Anderiesz 1, G.MJones 1, A.Kausche 1, N.Lolatgis 2 and C.Wood 2
 A.Trounson 1 ' 3, C.Anderiesz 1, G.MJones 1, A.Kausche 1, N.Lolatgis 2 and C.Wood 2 Centre for Early Human Development, Institute of Reproduction and Development, Monash University, Monash Medical Centre,
A.Trounson 1 ' 3, C.Anderiesz 1, G.MJones 1, A.Kausche 1, N.Lolatgis 2 and C.Wood 2 Centre for Early Human Development, Institute of Reproduction and Development, Monash University, Monash Medical Centre,
OVARY The surface of the ovary is covered with surface epithelium
 OVARY Cow The ovary, or female gonad, is: 1. an exocrine gland, producing oocytes 2. an endocrine gland, secreting hormones, i.e., estrogen and progesterone OVARY OVARY The surface of the ovary is covered
OVARY Cow The ovary, or female gonad, is: 1. an exocrine gland, producing oocytes 2. an endocrine gland, secreting hormones, i.e., estrogen and progesterone OVARY OVARY The surface of the ovary is covered
DEVELOPMENT OF A COMMERCIAL EMBRYO
 DEVELOPMENT OF A COMMERCIAL EMBRYO FREEZING PROTOCOL FOR THE CONSERVATION, TRANSPORTATION AND IMPORTATION OF PIG GENETICS 2E-105 Report prepared for the Co-operative Research Centre for an Internationally
DEVELOPMENT OF A COMMERCIAL EMBRYO FREEZING PROTOCOL FOR THE CONSERVATION, TRANSPORTATION AND IMPORTATION OF PIG GENETICS 2E-105 Report prepared for the Co-operative Research Centre for an Internationally
RapiDVIT & rapidwarm oocyte. Specialised media for oocyte vitrification.
 RapiDVIT & rapidwarm oocyte Specialised media for oocyte vitrification. Special media for A unique cell Cryopreservation of oocytes requires care. Some preservation techniques cause premature oocyte activation
RapiDVIT & rapidwarm oocyte Specialised media for oocyte vitrification. Special media for A unique cell Cryopreservation of oocytes requires care. Some preservation techniques cause premature oocyte activation
A Tale of Three Hormones: hcg, Progesterone and AMH
 A Tale of Three Hormones: hcg, Progesterone and AMH Download the Ferring AR ipad/iphone app from the Apple Store: http://bit.ly/1okk74m Human Ovarian Steroidogenesis and Gonadotrophin Stimulation Johan
A Tale of Three Hormones: hcg, Progesterone and AMH Download the Ferring AR ipad/iphone app from the Apple Store: http://bit.ly/1okk74m Human Ovarian Steroidogenesis and Gonadotrophin Stimulation Johan
Damage of embryo development caused by peroxidized mineral oil and its association with albumin in culture
 Damage of embryo development caused by peroxidized and its association with albumin in culture Junko Otsuki, Ph.D., a,b Yasushi Nagai, M.D., a and Kazuyoshi Chiba, Ph.D. b a Nagai Clinic, Saitama; and
Damage of embryo development caused by peroxidized and its association with albumin in culture Junko Otsuki, Ph.D., a,b Yasushi Nagai, M.D., a and Kazuyoshi Chiba, Ph.D. b a Nagai Clinic, Saitama; and
The Effects of camp-elevating Agents and Alpha Lipoic Acid on In Vitro Maturation of Mouse Germinal Vesicle Oocytes
 Original Article The Effects of camp-elevating Agents and Alpha Lipoic Acid on In Vitro Maturation of Mouse Germinal Vesicle Oocytes Ali Rahnama 1, Saeed Zavareh 1, 2*, Mohammad Taghi Ghorbanian 1,2, Isaac
Original Article The Effects of camp-elevating Agents and Alpha Lipoic Acid on In Vitro Maturation of Mouse Germinal Vesicle Oocytes Ali Rahnama 1, Saeed Zavareh 1, 2*, Mohammad Taghi Ghorbanian 1,2, Isaac
Effect of Leukemia Inhibiton Factor (LIF) on in vitro maturation and fertilization of matured cattle oocytes
 Theriogenology Insight: 4(3): 17-111, December, 214 DOI Number: 1.98/2277-3371.214.74.2 Effect of Leukemia Inhibiton Factor (LIF) on in vitro maturation and fertilization of matured cattle oocytes K M
Theriogenology Insight: 4(3): 17-111, December, 214 DOI Number: 1.98/2277-3371.214.74.2 Effect of Leukemia Inhibiton Factor (LIF) on in vitro maturation and fertilization of matured cattle oocytes K M
Results and Discussion
 The AMH and Inhibin B in serum and follicular fluid was a very good predictive marker for assessment of oocyte quantity but did not correlate with the maturity of the oocyte or fertilization or reproductive
The AMH and Inhibin B in serum and follicular fluid was a very good predictive marker for assessment of oocyte quantity but did not correlate with the maturity of the oocyte or fertilization or reproductive
Progesterone and clinical outcomes
 Synchronization of Slowly Developing Embryos Restores Implantation Success Richard T. Scott, Jr, MD, HCLD Clinical and Scientific Director, Reproductive Medicine Associates of New Jersey Professor and
Synchronization of Slowly Developing Embryos Restores Implantation Success Richard T. Scott, Jr, MD, HCLD Clinical and Scientific Director, Reproductive Medicine Associates of New Jersey Professor and
Understanding eggs, sperm and embryos. Marta Jansa Perez Wolfson Fertility Centre
 Understanding eggs, sperm and embryos Marta Jansa Perez Wolfson Fertility Centre What does embryology involve? Aims of the embryology laboratory Creation of a large number of embryos and supporting their
Understanding eggs, sperm and embryos Marta Jansa Perez Wolfson Fertility Centre What does embryology involve? Aims of the embryology laboratory Creation of a large number of embryos and supporting their
IVF (,, ) : (HP-hMG) - (IVF- ET) : GnRH, HP-hMG (HP-hMG )57, (rfsh )140, (Gn)
 34 11 Vol.34 No.11 2014 11 Nov. 2014 Reproduction & Contraception doi: 10.7669/j.issn.0253-3X.2014.11.0892 E-mail: randc_journal@163.com IVF ( 710003) : (H-hMG) - (IVF- ET) : GnRH H-hMG (H-hMG ) (rfsh
34 11 Vol.34 No.11 2014 11 Nov. 2014 Reproduction & Contraception doi: 10.7669/j.issn.0253-3X.2014.11.0892 E-mail: randc_journal@163.com IVF ( 710003) : (H-hMG) - (IVF- ET) : GnRH H-hMG (H-hMG ) (rfsh
EFFECT OF VITRIFICATION AND CRYOSTORAGE LENGTH ON VIABILITY OF RABBIT EMBRYOS AFTER THAWING
 World Rabbit Science Association Proceedings 10 th World Rabbit Congress September 3-6, 2012 Sharm El- Sheikh Egypt, 303-307 EFFECT OF VITRIFICATION AND CRYOSTORAGE LENGTH ON VIABILITY OF RABBIT EMBRYOS
World Rabbit Science Association Proceedings 10 th World Rabbit Congress September 3-6, 2012 Sharm El- Sheikh Egypt, 303-307 EFFECT OF VITRIFICATION AND CRYOSTORAGE LENGTH ON VIABILITY OF RABBIT EMBRYOS
VERGE 3 Lundeberg 1. Dependence of fertilization in sea urchins, Strongylocentrotus purpuratus, on microfilament
 VERGE 3 Lundeberg 1 Dependence of fertilization in sea urchins, Strongylocentrotus purpuratus, on microfilament formation and internal calcium concentration Megan Lundeberg Amy Ruggerio and Amy Isaacson
VERGE 3 Lundeberg 1 Dependence of fertilization in sea urchins, Strongylocentrotus purpuratus, on microfilament formation and internal calcium concentration Megan Lundeberg Amy Ruggerio and Amy Isaacson
Patrick Quinn IVF PROTOKOL FOR SINGLE EMBRYO CULTURE
 1. With cumulus-free oocytes and embryos up to Day (D) 3, use 275-300 um diameter pipette tips to minimize medium transfer between drops; transfer volume should be < 1 ul. DAY -1 2. At ~ 4.00 pm on the
1. With cumulus-free oocytes and embryos up to Day (D) 3, use 275-300 um diameter pipette tips to minimize medium transfer between drops; transfer volume should be < 1 ul. DAY -1 2. At ~ 4.00 pm on the
Curriculum Vitae. Personal Information. In the name of God. First name: Zahra Last name: Shams Mofarahe
 In the name of God Curriculum Vitae Personal Information First name: Zahra Last name: Shams Mofarahe Status: Assistant Professor of Department Biology and Anatomical Sciences Gender: Female Marital status:
In the name of God Curriculum Vitae Personal Information First name: Zahra Last name: Shams Mofarahe Status: Assistant Professor of Department Biology and Anatomical Sciences Gender: Female Marital status:
Ovarian Follicular Development in the Untreated and
 Ovarian Follicular Development in the Untreated and PMSG-treated Cyclic Rat Hajime MIYAMOTO, Goro KATSUURA and Takehiko ISHIBASHI Department of Animal Science, College of Agriculture, Kyoto University,
Ovarian Follicular Development in the Untreated and PMSG-treated Cyclic Rat Hajime MIYAMOTO, Goro KATSUURA and Takehiko ISHIBASHI Department of Animal Science, College of Agriculture, Kyoto University,
Effect of Equilibration Temperature on In vitro Viability and Subsequent Embryo Development of Vitrified-Warmed Immature Bovine Oocytes
 American Journal of Animal and Veterinary Sciences 5 (2): 71-75, 2010 ISSN 1557-4555 2010 Science Publications Effect of Equilibration Temperature on In vitro Viability and Subsequent Embryo Development
American Journal of Animal and Veterinary Sciences 5 (2): 71-75, 2010 ISSN 1557-4555 2010 Science Publications Effect of Equilibration Temperature on In vitro Viability and Subsequent Embryo Development
Original Article. Abstract
 Original Article Comparison of Different Vitrification Procedures on Developmental Competence of Mouse Germinal Vesicle Oocytes in the Presence or Absence of Cumulus Cells Saeed Zavareh, D.V.M. 1, Mojdeh
Original Article Comparison of Different Vitrification Procedures on Developmental Competence of Mouse Germinal Vesicle Oocytes in the Presence or Absence of Cumulus Cells Saeed Zavareh, D.V.M. 1, Mojdeh
Simple, efficient and successful vitrification of bovine blastocysts using electron microscope grids
 Human Reproduction vol.14 no.11 pp.838-843, 1999 Simple, efficient and successful vitrification of bovine blastocysts using electron microscope grids Se-Pill Park, Eun Young Kim, Deok Im Kim, Noh Hyung
Human Reproduction vol.14 no.11 pp.838-843, 1999 Simple, efficient and successful vitrification of bovine blastocysts using electron microscope grids Se-Pill Park, Eun Young Kim, Deok Im Kim, Noh Hyung
The serum estradiol/oocyte ratio in patients with breast cancer undergoing ovarian stimulation with letrozole and gonadotropins
 Original Article Obstet Gynecol Sci 2018;61(2):242-246 https://doi.org/10.5468/ogs.2018.61.2.242 pissn 2287-8572 eissn 2287-8580 The serum estradiol/oocyte ratio in patients with breast cancer undergoing
Original Article Obstet Gynecol Sci 2018;61(2):242-246 https://doi.org/10.5468/ogs.2018.61.2.242 pissn 2287-8572 eissn 2287-8580 The serum estradiol/oocyte ratio in patients with breast cancer undergoing
Female Reproductive Physiology. Dr Raelia Lew CREI, FRANZCOG, PhD, MMed, MBBS Fertility Specialist, Melbourne IVF
 Female Reproductive Physiology Dr Raelia Lew CREI, FRANZCOG, PhD, MMed, MBBS Fertility Specialist, Melbourne IVF REFERENCE Lew, R, Natural History of ovarian function including assessment of ovarian reserve
Female Reproductive Physiology Dr Raelia Lew CREI, FRANZCOG, PhD, MMed, MBBS Fertility Specialist, Melbourne IVF REFERENCE Lew, R, Natural History of ovarian function including assessment of ovarian reserve
Effect of excess (within tolerable range) and excessive iodine for different durations on functional status of testis with possible mechanism
 Section III. Chapter 4 Effect of excess (within tolerable range) and excessive iodine for different durations on functional status of testis with possible mechanism Introduction Thyroid hormones regulate
Section III. Chapter 4 Effect of excess (within tolerable range) and excessive iodine for different durations on functional status of testis with possible mechanism Introduction Thyroid hormones regulate
SHORT COMMUNICATION SEOUL, SOUTH KOREA
 ( C 2006) DOI: 10.1007/s10815-005-9006-0 SHORT COMMUNICATION SEOUL, SOUTH KOREA Optimization of a Dilution Method for Human Expanded Blastocysts Vitrified Using EM Grids After Artificial Shrinkage Submitted
( C 2006) DOI: 10.1007/s10815-005-9006-0 SHORT COMMUNICATION SEOUL, SOUTH KOREA Optimization of a Dilution Method for Human Expanded Blastocysts Vitrified Using EM Grids After Artificial Shrinkage Submitted
Assisted Reproductive Technologies. Simpler Processes. Less Stress. Better Results. Embryo Culture Media. Protein Supplements and Oil.
 Assisted Reproductive Technologies Simpler Processes. Less Stress. Better Results. Embryo Culture Media Protein Supplements and Oil Vitrification Visit us at www.irvinesci.com for more information To order
Assisted Reproductive Technologies Simpler Processes. Less Stress. Better Results. Embryo Culture Media Protein Supplements and Oil Vitrification Visit us at www.irvinesci.com for more information To order
Clinical ICSI in the horse:
 Clinical ICSI in the horse: differences and similarities to human in an in vitro maturation-based system Katrin Hinrichs College of Veterinary Medicine & Biomedical Sciences Texas A&M University Standard
Clinical ICSI in the horse: differences and similarities to human in an in vitro maturation-based system Katrin Hinrichs College of Veterinary Medicine & Biomedical Sciences Texas A&M University Standard
IN VITRO FERTILIZATION OF RABBIT EGGS IN OVIDUCT SECRETIONS FROM DIFFERENT DAYS BEFORE AND AFTER OVULATION*
 FERTILITY AND STERILITY Copyright~ 1975 The American Fertility Society Vol. 26, No.7, July 1975 Printed in U.SA. IN VITRO FERTILIZATION OF RABBIT EGGS IN OVIDUCT SECRETIONS FROM DIFFERENT DAYS BEFORE AND
FERTILITY AND STERILITY Copyright~ 1975 The American Fertility Society Vol. 26, No.7, July 1975 Printed in U.SA. IN VITRO FERTILIZATION OF RABBIT EGGS IN OVIDUCT SECRETIONS FROM DIFFERENT DAYS BEFORE AND
A Case of Pregnancy Using Recombinant Follicle Stimulating Hormone and Gonadotropin Releasing Hormone Antagonist
 1 *, ** * * * ** A Case of Pregnancy Using Recombinant Follicle Stimulating Hormone and Gonadotropin Releasing Hormone Antagonist Yoon Sung Nam, Nam Keun Kim*, Eun Kyung Kim**, Hyung Min Chung** and Kwang
1 *, ** * * * ** A Case of Pregnancy Using Recombinant Follicle Stimulating Hormone and Gonadotropin Releasing Hormone Antagonist Yoon Sung Nam, Nam Keun Kim*, Eun Kyung Kim**, Hyung Min Chung** and Kwang
