Updates on the Progress of the. Harmonization Teams (GBC-HT) for Small & Large Molecules
|
|
|
- Allyson Blair
- 5 years ago
- Views:
Transcription
1 Updates on the Progress of the Global BioanalyticalConsortium Harmonization Teams (GBC-HT) for Small & Large Molecules Dr. Fabio Garofolo for GBC AAPS Annual Meeting October Washington DC -USA
2 Agenda Brief History Review GBC structure Harmonization Teams Activities New insights developed at GBC-SC meetings
3 History : Loose discussions in multiple BA communities contemplating on the need and added value of harmonized BA guidelines Dec EBF Conference (Barcelona, Spain) Formal request for harmonization from Bioanalytical community Acknowledgement by Regulatory Agencies present (FDA & EMA) Discussion among international pharmaceutical scientific organizations with a strong stake in bioanalysis: AAPS, APA, CVG and EBF Request Health Authorities to initiate a harmonization process Offer support to Health Authorities for such a process Letter sent to FDA and EMA in February 2010 Publication as Open letter in April 2010 issue of Bioanalysis Entertain initial idea of forming a Global Bioanalysis Consortium
4 History Apr th CVG Workshop (Montreal, Canada) Consensus reached among panelists, 5 regulatory agencies and international attendees on how to proceed with the Global Harmonization of Bioanalytical Guidances: institution of a Global Bioanalytical Consortium Agreement on the main characteristic of a Global Bioanalytical Guidance: Should be science driven Should include rationale behind each requirement to prevent box checking Should look at global picture, not local issues Should NOT be a prescriptive guidance Must get buy-in from all the countries
5 Organization Chart Steering Committee (GBC-SC) Scientific Leadership Team (GBC-SLT) Harmonization Team # 1 Harmonization Team # 2 Harmonization Team # n
6 Operating committees: GBC-SC North America (US + Canada) Mark Arnold (AAPS) Binodh DeSilva (AAPS) Fabio Garofolo (CVG) Latin America (South America + Mexico) Rafael Barrientos (AcBio) Asia Pacific (Asia + Pacific area) Shinobu Kudoh (JBF) Shrinivas Savale (APA-India) Daniel Tang (SBDG&BBDG) Europe (Europe + Africa/Middle East) Peter van Amsterdam (EBF) Michaela Golob (EBF) Philip Timmerman (EBF)
7 Harmonization Teams A Topics Common to All Molecules L Large Molecules Ligand Binding Assay S Small Molecules Chromatographic Assay A1 Scope and regulations A2 Tiered approaches for method validation L1 Large molecule specific run acceptance S1 Small molecule specific run acceptance A3 Method Transfer, partial/cross validations A4 Reference standards and reagents L2 Large molecule specific assay operation S2 Small molecule specific assay operation A5 Sample Management A6 Stability L3 Assay formats S3 Chromatographic Run Quality Assessment A7 Repeat analysis and ISR A8 Documentation L4 Reagents and their stability Link with tiered approach A9 Analytical Instrument Qualification A10 New Frontiers L5 Automation practices in LM bioanalysis A11 Biomarkers L6 Immunogenicity (effect on PK).
8 Harmonization Teams Team Leaders SC Sponsor Team Leaders SC Sponsor A1: Surendra Bansal A2: Steve Lowes A4: Joseph Bower A6: Nico van de Merbel A11: TBD Philip Timmerman Daniel Tang Shinobu Kudoh L1: Marian Kelley L2: Lauren Stevenson L3: Sherri Dudal L4: Lindsay King L5: Scott Davis L6: Jeff Sailstad A11: TBD L6: Jeff Sailstad Michaela Golob Fabio Garofolo Binodh DeSilva A3: Ray Briggs A5: Mike Redrup A7: Eric Fluhler A8: Tom Verhaeghe Peter van Amsterdam Shrinivas Savale A9: Chad Briscoe A10: Bob Bethem S1: Douglas Fast S2: Eric Woolf S3: Stuart McDougall Rafael Barrientos Mark Arnold
9 S2: Small Molecule Specific Assay Operation Team members: Team lead Eric Woolf NA Other members Abhishek Sharma APAC Barbara Duncan NA Berthold Lausecker EU Gabriel Marcelín LA Kazutaka Togashi APAC Miguel Vago- LA Pat Bennett NA Ravi Kumar Trivedi APAC Roger Hayes NA Steve White- EU Interdependencies with other teams: Team A6 (re: stability) Sample reinjection API Salt / Counter-ion changes Team A9 (re: system suitability) System Equilibration In scope Carryover and contamination methodology to assess acceptance criteria impact of sample analysis sequence Sensitivity One off std. curve range changes Specificity - selectivity impact of co. meds/metabolites Matrix Effects assessment methodology effect of hemolyzed/hyperlipidemic plasma Recovery assessment methodology & acceptance criteria IS evaluation addition methodology response variability assessment & acceptance criteria System equilibration use of study samples Sample reinjections Reporting of failed runs Impact of salt form/counter ion changes of analyte Preparation of calibrators organic solvent content Out of scope stability criteria
10 L1: Large Molecule Specific Run Acceptance Team members: Team lead Marian Kelley NA Other members Paula Kaminski, NA Daniella Stollner, EU Ross Bamford, EU Muruganandam Arumugam, APAC Ravi Trivedi, APAC Samantha Little, EU Lauren Stevenson, NA Dongbei Li, APAC Chris Beaver, NA In scope: Non-Linearity, of standard curve Makeup of standard curve Standard Curve editing Selection of best curve fit Quality Controls Assay range definition Accuracy, precision, total error Individual runs and overall run acceptance during validation Individual runs acceptance during samples analysis Inter-dependencies with other teams S1 Small Molecule Run Acceptance L2 Specific LBA Operation Out of scope: Stability of QCs long term during sample analysis: If it fails what do you compare to? Nominal, fresh? Two fold question: technical vs. stability. Will be addressed in L2 team
11 NEW insights developed at GBC-SC meetings Desire for increased engagement, input and contribution from the different regions The current team dynamics and composition may not sufficiently engage current non contributors in the broader scientific community Open discussion Desire to provide opportunity for regular updates on GBC progress in an open format The current process may lead to a significant period of radio silence Prevent that all GBC-proposals come as one avalanche at the global meeting, which may be too much to manage if not previewed Provide regulators a chance to get better understanding of activities of GBC
12 Proposal - How? Shift GBC Global Meeting from Q2 to Q Use appropriate regional meetings in all 4 regions (best attended and most affordable in each region, >1x region) to give a flavor of the progress we are making. If the regional meeting can accommodate, include a GBC session in those meetings to provide update and allow input Invite 4-5 topic HT-L (or a regional representative from those teams) to present the progress of their teams and to share. Stimulate HT-SC(s) to present high level progress on other topics, with input from other HT Engage with meeting organizers how to optimize GBC visibility during the meeting Publish outcome as a rapid communication to ensure all regions connect (GBC website or Bioanalysis ) Inviting organizations to provide travel assistance
13 Potential win-win Connect GBC better with the regions Reconnection with supporting organizations as our day to day supporters All regions get expanded opportunity to be involved Engage and inform a broader scientific community in advance of the global meeting Allow BA community to comment within the comfort zone of their region Allow BA community to comment to their regional organizations Provide the opportunity to publish a summary of thinking in advance of the global meeting Allow participants to know what s coming Be more engaged in the global meeting and not be caught by surprise Create visibility, recognition and connectivity in regions for HT-L and HT members for SC members Create flexibility to present on topics in need of influencing current thinking of regulators or on emerging guidelines
14 Proposed way forward Start up phase HT- L identification HT identification HT working on content working close with SLT SLT f-2-f Consolidation and joint discussion of all topics in preparation of 1st Global Meeting Global Meeting 3-day Conference in Rome (Italy) in week of Sept. 24, today Identified regional meetings Invite 4-5 topics to present the progress of their teams and to share. Present high level progress on other topics Get input
15 In practice With AAPS meeting being too soon on the calendar after our strategy update, 4th EBF meeting Barcelona (16-18 November) is first meeting where we will put this strategy in practice Session Global Harmonization on day 1: 4 teams volunteered to give update on progress and get input A1 A6 L6 S1 In addition, session includes round table, moderated by SC members from all 4 regions From then onwards, other meetings to follow depending on their ability to include GBC sessions (see next slide for meetings potentially qualifying)
16 In practice Identified meetings qualifying for inclusion GBC session Fit with respect to timing (italics= post GBC meeting) Fit with respect to willingness of organizers to include GBC session Meetings potentially qualifying further discussion with meeting organizers needed NA: EU: Mar. 2012: 6 th WRIB (CVG) San Antonio, USA May 2012: NBC (AAPS) - San Diego, USA May 2012: ASMS Vancouver, Canada Jul. 2012: Land O Lakes - Wisconsin, USA Sep. 2012: APA USA (BSAT) Boston, USA Other regional meetings (e.g., DVDMG) Nov. 2011: 4 th EBF Open Symposium Barcelona, Spain Jun. 2012: EBF Focus meeting Brussels, Belgium Nov. 2012: 5 th EBF Open Symposium Barcelona, Spain Other regional meetings (e.g., Fabian, French GLP,..) APAC: LA: Feb 2012: APA India (BSAT) Ahmedabad, India Mar. 2012: JBF, Tokyo Japan Nov. 2012:- 2 nd APBC (CVG) - Shanghai, China Other regional meetings May 2012 ACBio Sao Paulo, Brazil Other regional meetings
17 Acknowledgment Mark Arnold (AAPS) SC & FM Surendra Bansal (AAPS) - FM Rafael Barrientos (AcBio) - SC Binodh DeSilva (AAPS) - SC Douglas Fast (BSAT) - FM Fabio Garofolo (CVG) SC & FM Michaela Golob (EBF) SC Shinobu Kudoh (JBF) SC Steve Lowes (AAPS) - FM Shrinivas Savale (APA-India) - SC Daniel Tang (SBDG&BBDG) - SC Philip Timmerman (EBF) SC & FM Peter van Amsterdam (EBF) SC & FM Eric Woolf (BSAT) FM SC = Steering Committee FM = Founding Member
GLOBAL BIOANALYSIS CONSORTIUM
 GLOBAL BIOANALYSIS CONSORTIUM Regulated Bioanalysis - A Proposed Global HarmonizaDon Process presented by Peter Van Amsterdam on behalf of GBC at 3 rd EBF Focus Mee=ng June 13 th 2012 - Brussels - BELGIUM
GLOBAL BIOANALYSIS CONSORTIUM Regulated Bioanalysis - A Proposed Global HarmonizaDon Process presented by Peter Van Amsterdam on behalf of GBC at 3 rd EBF Focus Mee=ng June 13 th 2012 - Brussels - BELGIUM
GLOBAL BIOANALYSIS CONSORTIUM
 GLOBAL BIOANALYSIS CONSORTIUM Regulated Bioanalysis - A Proposed Global Harmonization Process Presented by Peter van Amsterdam for GBC SoFAQ International Seminar "Quality Assurance and Electronic Data"
GLOBAL BIOANALYSIS CONSORTIUM Regulated Bioanalysis - A Proposed Global Harmonization Process Presented by Peter van Amsterdam for GBC SoFAQ International Seminar "Quality Assurance and Electronic Data"
Current Developments in Guidance for Regulatory Bioanalysis
 Current Developments in Guidance for Regulatory Bioanalysis Dr Gerry McGuire Resolution Analytical Consultancy Ltd Edinburgh, Scotland UK gerry@resolutioneurope.com 1st MENA Regulatory Conference on Bioequivalence,
Current Developments in Guidance for Regulatory Bioanalysis Dr Gerry McGuire Resolution Analytical Consultancy Ltd Edinburgh, Scotland UK gerry@resolutioneurope.com 1st MENA Regulatory Conference on Bioequivalence,
AAPS Views on Bioanalytical Method Validation Harmonization (on Behalf of AAPS Bioanalytical Community)
 AAPS Views on Bioanalytical Method Validation Harmonization (on Behalf of AAPS Bioanalytical Community) Faye Vazvaei, Roche Innovation Center New York The 8th JBF Meeting, 8-9 February 2017 Background
AAPS Views on Bioanalytical Method Validation Harmonization (on Behalf of AAPS Bioanalytical Community) Faye Vazvaei, Roche Innovation Center New York The 8th JBF Meeting, 8-9 February 2017 Background
AAPS/PSCW 2010 APQ Open Forum
 AAPS/PSCW 2010 APQ Open Forum Harmonization of Global Bioanalytical Regulations Scientific Perspective and Update on Developments 18 November 2010 Steve Lowes, Ph.D. Advion BioServices Inc Ithaca, NY Introduction
AAPS/PSCW 2010 APQ Open Forum Harmonization of Global Bioanalytical Regulations Scientific Perspective and Update on Developments 18 November 2010 Steve Lowes, Ph.D. Advion BioServices Inc Ithaca, NY Introduction
GLOBAL BIOANALYSIS CONSORTIUM
 GLOBAL BIOANALYSIS CONSORTIUM Scope and Regulations Harmonization Team: A1 (Work in Progress) John Smeraglia on behalf of the HT-A1 A1: Scope and regulations Team members: Team lead Region Expertise Surendra
GLOBAL BIOANALYSIS CONSORTIUM Scope and Regulations Harmonization Team: A1 (Work in Progress) John Smeraglia on behalf of the HT-A1 A1: Scope and regulations Team members: Team lead Region Expertise Surendra
Regulated bioanalysis status in Japan and notable points of the draft Japanese guideline
 Regulated bioanalysis status in Japan and notable points of the draft Japanese guideline EBF 5th Open Meeting Old Battles, New Horizons, November, 16, 2012, Barcelona Noriko Katori, PhD National Institute
Regulated bioanalysis status in Japan and notable points of the draft Japanese guideline EBF 5th Open Meeting Old Battles, New Horizons, November, 16, 2012, Barcelona Noriko Katori, PhD National Institute
JBF Annual Report FY2014
 JBF Annual Report FY2014 (From 1 st April 2014 to 31 st March 2015) 1. Overview It has been passed three years and a half since the establishment of Japan Bioanalysis Forum (JBF). We aim to facilitate
JBF Annual Report FY2014 (From 1 st April 2014 to 31 st March 2015) 1. Overview It has been passed three years and a half since the establishment of Japan Bioanalysis Forum (JBF). We aim to facilitate
Progress to Date in A3: Method Transfer, Partial Validation and Cross validation
 Progress to Date in A3: Method Transfer, Partial Validation and Cross validation A3: Method Transfer, partial and cross validation Team members: Team lead Ray Briggs EU Other members Richard Abbott-EU
Progress to Date in A3: Method Transfer, Partial Validation and Cross validation A3: Method Transfer, partial and cross validation Team members: Team lead Ray Briggs EU Other members Richard Abbott-EU
The 6 th JBF Symposium Program Challenge of Regulated Bioanalysis
 25 th February (Wed.) The 6 th JBF Symposium Program Challenge of Regulated Bioanalysis Date: 25 th -26 th February 2015 Venue: Tower Hall Funabori, Tokyo, Japan (Oral presentation:small Hall on 5F, Poster
25 th February (Wed.) The 6 th JBF Symposium Program Challenge of Regulated Bioanalysis Date: 25 th -26 th February 2015 Venue: Tower Hall Funabori, Tokyo, Japan (Oral presentation:small Hall on 5F, Poster
Standardised Lab Manual for bioanalytical support in clinical studies : Introducing the harmonised Lab Manual. Introduction to the Workshop
 Standardised Lab Manual for bioanalytical support in clinical studies : Introducing the harmonised Lab Manual Introduction to the Workshop Philip Timmerman (for EBF) 05 February 2014 NH Sablon Hotel, Brussels
Standardised Lab Manual for bioanalytical support in clinical studies : Introducing the harmonised Lab Manual Introduction to the Workshop Philip Timmerman (for EBF) 05 February 2014 NH Sablon Hotel, Brussels
The regulatory landscape Stephen White on behalf of the EBF
 The regulatory landscape Stephen White on behalf of the EBF http://www.europeanbioanalysisforum.eu Overview 1. MIST and DDI Guideline 2. Regulatory landscape for BA 3. Tiered Approach on BA of metabolites
The regulatory landscape Stephen White on behalf of the EBF http://www.europeanbioanalysisforum.eu Overview 1. MIST and DDI Guideline 2. Regulatory landscape for BA 3. Tiered Approach on BA of metabolites
The 6 th JBF Symposium Program (draft) Challenge of Regulated Bioanalysis
 The 6 th JBF Symposium Program (draft) Challenge of Regulated Bioanalysis Date: 25 th -26 th February 2015 Venue: Tower Hall Funabori, Tokyo, Japan (Oral presentation:small Hall on 5F, Poster session :
The 6 th JBF Symposium Program (draft) Challenge of Regulated Bioanalysis Date: 25 th -26 th February 2015 Venue: Tower Hall Funabori, Tokyo, Japan (Oral presentation:small Hall on 5F, Poster session :
Update on ANVISA Bioanalytical Method Validation (BMV) Guideline
 Av. Andrade Neves, 295 - Sala 184 - Ed. Torre São Paulo - Centro - Campinas/SP CEP: 13013-160 - F:(+55-19)3231 8146 acbio@acbio.org.br - www.acbio.org.br Update on ANVISA Bioanalytical Method Validation
Av. Andrade Neves, 295 - Sala 184 - Ed. Torre São Paulo - Centro - Campinas/SP CEP: 13013-160 - F:(+55-19)3231 8146 acbio@acbio.org.br - www.acbio.org.br Update on ANVISA Bioanalytical Method Validation
Biomarker classification: a philosophical introduction.
 Biomarker classification: a philosophical introduction. Philip Timmerman, on behalf of the EBF biomarker team (TT-14) 16 November 2011, Barcelona, Spain Background A few no-brainers to start the conference
Biomarker classification: a philosophical introduction. Philip Timmerman, on behalf of the EBF biomarker team (TT-14) 16 November 2011, Barcelona, Spain Background A few no-brainers to start the conference
Global Harmonization: Latin America Overview and Perspective
 Av. Andrade Neves, 295 - Sala 184 - Ed. Torre São Paulo - Centro - Campinas/SP CEP: 13013-160 - F:(+55-19)3231 8146 secretaria@acbio.org.br - www.acbio.org.br Global Harmonization: Latin America Overview
Av. Andrade Neves, 295 - Sala 184 - Ed. Torre São Paulo - Centro - Campinas/SP CEP: 13013-160 - F:(+55-19)3231 8146 secretaria@acbio.org.br - www.acbio.org.br Global Harmonization: Latin America Overview
Bioanalytical Method Validation in Japan and activities of the Japan Bioanalysis Forum
 Bioanalytical Method Validation in Japan and activities of the Japan Bioanalysis Forum BMVJBF Noriko Katori, Ph.D. National Institute of Health Sciences Today s Topics 1. Recent History of BMV in Japan
Bioanalytical Method Validation in Japan and activities of the Japan Bioanalysis Forum BMVJBF Noriko Katori, Ph.D. National Institute of Health Sciences Today s Topics 1. Recent History of BMV in Japan
Bioanalytical Issues when. Brigitte Pellerin Bioanalytical Associate Director
 Bioanalytical Issues when Dealing with Phase II/III Studies Brigitte Pellerin Bioanalytical Associate Director 17-NOV-2015 Agenda Different phases of clinical trials Phase I to IV Challenges with Phase
Bioanalytical Issues when Dealing with Phase II/III Studies Brigitte Pellerin Bioanalytical Associate Director 17-NOV-2015 Agenda Different phases of clinical trials Phase I to IV Challenges with Phase
EBF Tiered approach final. Validation criteria. Philip Timmerman, on behalf of the EBF 8 th EBF Open Symposium
 EBF Tiered approach final recommendation of Scientific Validation criteria Philip Timmerman, on behalf of the EBF 8 th EBF Open Symposium - 2015 The problem statement 1990 CC-I 2001 FDA Further refinement,
EBF Tiered approach final recommendation of Scientific Validation criteria Philip Timmerman, on behalf of the EBF 8 th EBF Open Symposium - 2015 The problem statement 1990 CC-I 2001 FDA Further refinement,
t 14,081 FOLLOWERS f 28,855 FANS l 2,879 FOLLOWERS
 7 OVERVIEW 8,8 MEMBERS REPRESENTING 4 COUNTRIES,68,7 UNIQUE VISITS TO ASN WEBSITE,96 KIDNEY WEEK PARTICIPANTS IMPACT FACTORS 8.97 JASN 9,78 KIDNEY NEWS CIRCULATION NONMEMBER - 9,8 4.78 CJASN Latest available
7 OVERVIEW 8,8 MEMBERS REPRESENTING 4 COUNTRIES,68,7 UNIQUE VISITS TO ASN WEBSITE,96 KIDNEY WEEK PARTICIPANTS IMPACT FACTORS 8.97 JASN 9,78 KIDNEY NEWS CIRCULATION NONMEMBER - 9,8 4.78 CJASN Latest available
GUIDANCE DOCUMENTS CONTAINING THE COMMON PROVISIONS ON THE CONDUCT OF GCP INSPECTIONS BY COMPETENT AUTHORITIES OF THE DIFFERENT MEMBER STATES
 EUROPEAN COMMISSION ENTERPRISE AND INDUSTRY DIRECTORATE-GENERAL Consumer goods Pharmaceuticals Brussels, 5 November 2008 ENTR/F/2/SF D(2008) 34957 GUIDANCE DOCUMENTS CONTAINING THE COMMON PROVISIONS ON
EUROPEAN COMMISSION ENTERPRISE AND INDUSTRY DIRECTORATE-GENERAL Consumer goods Pharmaceuticals Brussels, 5 November 2008 ENTR/F/2/SF D(2008) 34957 GUIDANCE DOCUMENTS CONTAINING THE COMMON PROVISIONS ON
The potential of South East Asia and India as the hub for vaccine studies
 The potential of South East Asia and India as the hub for vaccine studies Angela Chen Therapeutic Strategy Lead 16 November 2017 Copyright 2017 IQVIA. All rights reserved. The potential of South East Asia
The potential of South East Asia and India as the hub for vaccine studies Angela Chen Therapeutic Strategy Lead 16 November 2017 Copyright 2017 IQVIA. All rights reserved. The potential of South East Asia
White Paper. For reprint orders, please contact
 White Paper For reprint orders, please contact reprints@future-science.com The European Bioanalysis Forum community s evaluation, interpretation and implementation of the European Medicines Agency guideline
White Paper For reprint orders, please contact reprints@future-science.com The European Bioanalysis Forum community s evaluation, interpretation and implementation of the European Medicines Agency guideline
THE MARKET-LEADING RESOURCE FOR THE GLOBAL BIOANALYTICAL COMMUNITY BIOANALYSIS ZONE 2018 MEDIA PACK
 THE MARKET-LEADING RESOURCE FOR THE GLOBAL BIOANALYTICAL COMMUNITY BIOANALYSIS ZONE 2018 MEDIA PACK Sarah Mayes: s.mayes@future-science.com Dionne Murray: d.murray@future-science.com +44 (0)20 8371 6090
THE MARKET-LEADING RESOURCE FOR THE GLOBAL BIOANALYTICAL COMMUNITY BIOANALYSIS ZONE 2018 MEDIA PACK Sarah Mayes: s.mayes@future-science.com Dionne Murray: d.murray@future-science.com +44 (0)20 8371 6090
EPIDEMIOLOGY. Accurate, in-depth information for understanding and assessing targeted markets
 EPIDEMIOLOGY Accurate, in-depth information for understanding and assessing targeted markets WHY ACCURATE MARKET FORECASTS Start with accurate understanding Epidemiology is the study of disease patterns
EPIDEMIOLOGY Accurate, in-depth information for understanding and assessing targeted markets WHY ACCURATE MARKET FORECASTS Start with accurate understanding Epidemiology is the study of disease patterns
Short Range Outlook for Steel Demand Autumn OECD Steel Committee, Paris, 10 December Short Range Outlook overview
 Short Range Outlook for Steel Demand Autumn 29 OECD Steel Committee, Paris, 1 December 29 Content Short Range Outlook overview Background of the Forecast Regional overviews Conclusion 2 1 Short Range Outlook
Short Range Outlook for Steel Demand Autumn 29 OECD Steel Committee, Paris, 1 December 29 Content Short Range Outlook overview Background of the Forecast Regional overviews Conclusion 2 1 Short Range Outlook
Sample logistics: problems and solutions in multi-center clinical trials Proposed Generic EBF Lab Manual
 Sample logistics: problems and solutions in multi-center clinical trials Proposed Generic EBF Lab Manual Rebecca Sleigh on behalf of TT-12 6 th EBF Open Symposium 20-22 November 2013 Hesperia Tower, Barcelona
Sample logistics: problems and solutions in multi-center clinical trials Proposed Generic EBF Lab Manual Rebecca Sleigh on behalf of TT-12 6 th EBF Open Symposium 20-22 November 2013 Hesperia Tower, Barcelona
WHAT IS THE DECISION MAKING PROCESS?
 5841 Cedar Lake Road, Suite 204 Minneapolis, MN 55416 USA Tel: 952.646.2029 Fax: 952.545.6073 www.humanbrainmapping.org GUIDE AND GUIDE AGREEMENT TO SUBMITTING TO SUBMITTING A A LETTER BID OF INTEREST
5841 Cedar Lake Road, Suite 204 Minneapolis, MN 55416 USA Tel: 952.646.2029 Fax: 952.545.6073 www.humanbrainmapping.org GUIDE AND GUIDE AGREEMENT TO SUBMITTING TO SUBMITTING A A LETTER BID OF INTEREST
Global E-Cigarette & Vaporizer Market Research Report - Forecast to 2027
 Report Information More information from: https://www.marketresearchfuture.com/reports/788 Global E-Cigarette & Vaporizer Market Research Report - Forecast to 2027 Report / Search Code: MRFR/CR/0304-CRR
Report Information More information from: https://www.marketresearchfuture.com/reports/788 Global E-Cigarette & Vaporizer Market Research Report - Forecast to 2027 Report / Search Code: MRFR/CR/0304-CRR
High-Throughput, Cost-Efficient LC-MS/MS Forensic Method for Measuring Buprenorphine and Norbuprenorphine in Urine
 High-Throughput, Cost-Efficient LC-MS/MS Forensic Method for Measuring and in Urine Xiaolei Xie, Joe DiBussolo, Marta Kozak; Thermo Fisher Scientific, San Jose, CA Application Note 627 Key Words, norbuprenorphine,
High-Throughput, Cost-Efficient LC-MS/MS Forensic Method for Measuring and in Urine Xiaolei Xie, Joe DiBussolo, Marta Kozak; Thermo Fisher Scientific, San Jose, CA Application Note 627 Key Words, norbuprenorphine,
Towards a Sustainable Global Infrastructure for Medical Countermeasures
 Towards a Sustainable Global Infrastructure for Medical Countermeasures Institute of Medicine The Public Health Emergency Medical Countermeasures Enterprise: Innovative Strategies to Enhance Products from
Towards a Sustainable Global Infrastructure for Medical Countermeasures Institute of Medicine The Public Health Emergency Medical Countermeasures Enterprise: Innovative Strategies to Enhance Products from
PPD S EXPERT HEMATOLOGY AND ONCOLOGY TEAM
 HEMATOLOGY ONCOLOGY PPD S EXPERT HEMATOLOGY AND ONCOLOGY TEAM COMMITTED TO ADVANCING DRUG DEVELOPMENT IN ONCOLOGY $ MARKETPLACE COMPLEXITIES increasingly competitive marketplace and rising cost pressures
HEMATOLOGY ONCOLOGY PPD S EXPERT HEMATOLOGY AND ONCOLOGY TEAM COMMITTED TO ADVANCING DRUG DEVELOPMENT IN ONCOLOGY $ MARKETPLACE COMPLEXITIES increasingly competitive marketplace and rising cost pressures
Bioanalytical Method Development and Validation: from the USFDA 2001 to the USFDA 2018 Guidance for Industry
 Journal of Applied Bioanalysis http://dx.doi.org/10.17145/jab.18.010 COMMENTARY Bioanalytical Method Development and Validation: from the USFDA 2001 to the USFDA 2018 Guidance for Industry Roland J.W.
Journal of Applied Bioanalysis http://dx.doi.org/10.17145/jab.18.010 COMMENTARY Bioanalytical Method Development and Validation: from the USFDA 2001 to the USFDA 2018 Guidance for Industry Roland J.W.
MARKET NEWS for pig meat
 MARKET NEWS for pig meat Market analysis 17 December 2018 Week 51 MARKET SITUATION Europe: Trading in legs and other cuts is at unchanged or slightly falling prices. UK: Prices are reported as stable.
MARKET NEWS for pig meat Market analysis 17 December 2018 Week 51 MARKET SITUATION Europe: Trading in legs and other cuts is at unchanged or slightly falling prices. UK: Prices are reported as stable.
Assay Cross Validation
 Assay Cross Validation Recent experiences in transferring bioanalytical assays from sponsor to CRO partners and between CROs Stephen White EBF Open Symposium Barcelona 20 th Nov 2013 Assay Cross Validation
Assay Cross Validation Recent experiences in transferring bioanalytical assays from sponsor to CRO partners and between CROs Stephen White EBF Open Symposium Barcelona 20 th Nov 2013 Assay Cross Validation
Gender Statistics Programme of the United Nations Economic Commission for Europe
 United Nations Statistical Commission 7 February 2011 Forty-second session New York, 22-25 February 2011 Room paper Item 3b of the provisional agenda Available in English only Programme review: gender
United Nations Statistical Commission 7 February 2011 Forty-second session New York, 22-25 February 2011 Room paper Item 3b of the provisional agenda Available in English only Programme review: gender
European Patients Academy on Therapeutic Innovation
 European Patients Academy on Therapeutic Innovation http://www.patientsacademy.eu info@patientsacademy.eu The project is receiving support from the Innovative Medicines Initiative Joint Undertaking under
European Patients Academy on Therapeutic Innovation http://www.patientsacademy.eu info@patientsacademy.eu The project is receiving support from the Innovative Medicines Initiative Joint Undertaking under
Supplementary Data of Consolidated Financial Statements for the third quarter ended December 31, 2016
 Supplementary Data of Financial Statements for the third quarter ended December 31, 2016 January 31, 2017 NITTO DENKO CORPORATION CONTENTS Page Business 1 Segment Information 2 Segment Information By Geographic
Supplementary Data of Financial Statements for the third quarter ended December 31, 2016 January 31, 2017 NITTO DENKO CORPORATION CONTENTS Page Business 1 Segment Information 2 Segment Information By Geographic
Tandem Diabetes Care, Inc. (TNDM) - Medical Equipment - Deals and Alliances Profile
 Tandem Diabetes Care, Inc. (TNDM) - Medical Equipment - Deals and Alliances Profile Tandem Diabetes Care, Inc. (TNDM) - Medical Equipment - Deals and Alliances Profile BioPortfolio has been marketing business
Tandem Diabetes Care, Inc. (TNDM) - Medical Equipment - Deals and Alliances Profile Tandem Diabetes Care, Inc. (TNDM) - Medical Equipment - Deals and Alliances Profile BioPortfolio has been marketing business
SEVENTH INTERNATIONAL WORKSHOP ON DOSIMETRY FOR RADIATION PROCESSING
 INTRODUCTION ASTM Subcommittee E10.01, Radiation Processing: Dosimetry and Applications, is proud to present the seventh edition of our International Workshop on Dosimetry for Radiation Processing. This
INTRODUCTION ASTM Subcommittee E10.01, Radiation Processing: Dosimetry and Applications, is proud to present the seventh edition of our International Workshop on Dosimetry for Radiation Processing. This
IAEA Activities in the Area of Fast Neutron Systems
 Worrksshop on Teecchnollogy aand Applliiccaattiionss off Acccceelleerraattorr Drriiveen Sysstteemss ((ADS)) 17 -- 28 Occttobeerr 2005 1677/10 IAEA Activities in the Area of Fast Neutron Systems A. Stanculescu
Worrksshop on Teecchnollogy aand Applliiccaattiionss off Acccceelleerraattorr Drriiveen Sysstteemss ((ADS)) 17 -- 28 Occttobeerr 2005 1677/10 IAEA Activities in the Area of Fast Neutron Systems A. Stanculescu
MEETING PROSPECTUS. International Workshop on NASH Biomarkers
 International Workshop on NASH Biomarkers 2018 18-19 May 2018 Washington, D.C., USA MEETING PROSPECTUS www.expertmedicaleducation.com www.expertmedicalevents.com ADVANCING HEALTH GLOBALLY THROUGH INNOVATIVE
International Workshop on NASH Biomarkers 2018 18-19 May 2018 Washington, D.C., USA MEETING PROSPECTUS www.expertmedicaleducation.com www.expertmedicalevents.com ADVANCING HEALTH GLOBALLY THROUGH INNOVATIVE
WCO Strategic Initiatives for Trade Facilitation
 International Seminar on Trade Facilitation in North East Asia 13 December 2016 Incheon, Republic of Korea WCO Strategic Initiatives for Trade Facilitation Ms. Alpha WONG World Customs Organization (WCO)
International Seminar on Trade Facilitation in North East Asia 13 December 2016 Incheon, Republic of Korea WCO Strategic Initiatives for Trade Facilitation Ms. Alpha WONG World Customs Organization (WCO)
Global Trade in Lightweight Coated Writing Paper TradeData International Pty Ltd (www.tradedata.net) Page 1 5/18/2015
 Page 1 5/18/2015 An Analysis of Global Trade in Lightweight paper, coated with inorganic substances, used for writing etc., of which more than 10% by weight of total fibre content consists of fibres obtained
Page 1 5/18/2015 An Analysis of Global Trade in Lightweight paper, coated with inorganic substances, used for writing etc., of which more than 10% by weight of total fibre content consists of fibres obtained
PRA International Early Development Services Bioanalytical Laboratory
 PRA International Early Development Services Bioanalytical Laboratory Bioanalytical Laboratories: Retain profitability in a competitive market A View from a Major CRO Houten October 8, 2009 Peter Ketelaar
PRA International Early Development Services Bioanalytical Laboratory Bioanalytical Laboratories: Retain profitability in a competitive market A View from a Major CRO Houten October 8, 2009 Peter Ketelaar
Implementation of estimands in Novo Nordisk
 Implementation of estimands in Novo Nordisk Søren Andersen Helle Lynggaard Biostatistics, Novo Nordisk A/S DSBS meeting 26 October 2017 2 Agenda Overview of implementation process Cross-functional working
Implementation of estimands in Novo Nordisk Søren Andersen Helle Lynggaard Biostatistics, Novo Nordisk A/S DSBS meeting 26 October 2017 2 Agenda Overview of implementation process Cross-functional working
Slide 1. Winning with GLP-1. Lars Fruergaard Jørgensen President and CEO. ALEXANDRE DE GREGORIO, Brazil Alexandre has type 2 diabetes
 Slide 1 Lars Fruergaard Jørgensen President and CEO ALEXANDRE DE GREGORIO, Brazil Alexandre has type 2 diabetes Slide 2 Forward-looking statements Novo Nordisk s reports filed with or furnished to the
Slide 1 Lars Fruergaard Jørgensen President and CEO ALEXANDRE DE GREGORIO, Brazil Alexandre has type 2 diabetes Slide 2 Forward-looking statements Novo Nordisk s reports filed with or furnished to the
t 11,028 FOLLOWERS f 19,240 FANS l 2,517 FOLLOWERS
 216 OVERVIEW 16,824 MEMBERS REPRESENTING 124 COUNTRIES 4,594,81 UNIQUE VISITS TO ASN WEBSITE 13,351 KIDNEY WEEK PARTICIPANTS IMPACT FACTORS 8.49 JASN 19,337 KIDNEY NEWS CIRCULATION 4.7 CJASN Latest available
216 OVERVIEW 16,824 MEMBERS REPRESENTING 124 COUNTRIES 4,594,81 UNIQUE VISITS TO ASN WEBSITE 13,351 KIDNEY WEEK PARTICIPANTS IMPACT FACTORS 8.49 JASN 19,337 KIDNEY NEWS CIRCULATION 4.7 CJASN Latest available
Challenges in Performing a Scientifically Meaningful Lipemic Plasma Test in Bioanalytical Method Validation
 Challenges in Performing a Scientifically Meaningful Lipemic Plasma Test in Bioanalytical Method Validation Laurence Mayrand-Provencher, Milton Furtado, Jean- Nicholas Mess, Josée Michon, Annik Bergeron,
Challenges in Performing a Scientifically Meaningful Lipemic Plasma Test in Bioanalytical Method Validation Laurence Mayrand-Provencher, Milton Furtado, Jean- Nicholas Mess, Josée Michon, Annik Bergeron,
Update from FDA Office of Regulatory Affairs
 Update from FDA Office of Regulatory Affairs Ellen F. Morrison Assistant Commissioner for Medical Products and Tobacco Operations Office of Regulatory Affairs U.S. Food and Drug Administration 1 The Impact
Update from FDA Office of Regulatory Affairs Ellen F. Morrison Assistant Commissioner for Medical Products and Tobacco Operations Office of Regulatory Affairs U.S. Food and Drug Administration 1 The Impact
Setting the scene Background of the Radiation Protection Culture initiative
 IRPA IOMP WHO Workshop Radiation Protection Culture in Medicine Geneva, 30.11. 02.11.2015 Setting the scene Background of the Radiation Protection Culture initiative Bernard Le Guen, IRPA Executive Officer
IRPA IOMP WHO Workshop Radiation Protection Culture in Medicine Geneva, 30.11. 02.11.2015 Setting the scene Background of the Radiation Protection Culture initiative Bernard Le Guen, IRPA Executive Officer
Bio-Rad Laboratories HEMOGLOBIN Testing Bio-Rad A1c. VARIANT II TURBO Link. Fully-Automated HbA 1c Testing
 Bio-Rad Laboratories HEMOGLOBIN Testing Bio-Rad A1c VARIANT II TURBO Link Fully-Automated HbA 1c Testing Bio-Rad Laboratories HEMOGLOBIN Testing VARIANT II TURBO Link Efficient streamlined process Powerful
Bio-Rad Laboratories HEMOGLOBIN Testing Bio-Rad A1c VARIANT II TURBO Link Fully-Automated HbA 1c Testing Bio-Rad Laboratories HEMOGLOBIN Testing VARIANT II TURBO Link Efficient streamlined process Powerful
Economic and Social Council
 United Nations E/CN.3/2018/22 Economic and Social Council Distr.: General 14 December 2017 Original: English Statistical Commission Forty-ninth session 6 9 March 2018 Item 4 (b) of the provisional agenda*
United Nations E/CN.3/2018/22 Economic and Social Council Distr.: General 14 December 2017 Original: English Statistical Commission Forty-ninth session 6 9 March 2018 Item 4 (b) of the provisional agenda*
ICH M4E(R2): Revised Guideline on Common Technical Document -- Efficacy
 ICH M4E(R2): Revised Guideline on Common Technical Document -- Efficacy International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use 1 Background Regulatory authorities
ICH M4E(R2): Revised Guideline on Common Technical Document -- Efficacy International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use 1 Background Regulatory authorities
Study Endpoint Considerations: Final PRO Guidance and Beyond
 Study Endpoint Considerations: Final PRO Guidance and Beyond Laurie Burke Associate Director for Study Endpoints and Labeling OND/CDER/FDA Presented at: FIRST ANNUAL PATIENT REPORTED OUTCOMES (PRO) CONSORTIUM
Study Endpoint Considerations: Final PRO Guidance and Beyond Laurie Burke Associate Director for Study Endpoints and Labeling OND/CDER/FDA Presented at: FIRST ANNUAL PATIENT REPORTED OUTCOMES (PRO) CONSORTIUM
ILyte TM Automated electrolyte analyzers
 ILyte TM Automated electrolyte analyzers The IL program for electrolyte analysis. Worry Free, Easy to Use Maintenance Free Electrodes ILyte Na +, K + analyzer ILyte Na +, K +, Cl - analyzer ILyte Na +,
ILyte TM Automated electrolyte analyzers The IL program for electrolyte analysis. Worry Free, Easy to Use Maintenance Free Electrodes ILyte Na +, K + analyzer ILyte Na +, K +, Cl - analyzer ILyte Na +,
Global Pediatric Development: We Are Making Progress. PMDA Perspective. Junko Sato, PhD PMDA
 Global Pediatric Development: We Are Making Progress PMDA Perspective Junko Sato, PhD PMDA Disclaimer The views and opinions expressed in the following PowerPoint slides are those of the individual presenter
Global Pediatric Development: We Are Making Progress PMDA Perspective Junko Sato, PhD PMDA Disclaimer The views and opinions expressed in the following PowerPoint slides are those of the individual presenter
First Quarter 2006 Revenues. Paris, May 3, 2006
 First Quarter 2006 Revenues Paris, May 3, 2006 1Q 2006 Revenues ( millions ) 1Q 2005 1Q 2006 888 994 Organic growth : + 12 % + 6.3 % Little change to the scope of consolidation Positive exchange rate effects
First Quarter 2006 Revenues Paris, May 3, 2006 1Q 2006 Revenues ( millions ) 1Q 2005 1Q 2006 888 994 Organic growth : + 12 % + 6.3 % Little change to the scope of consolidation Positive exchange rate effects
Newspaper Content Analysis Monthly Update January 2010 Prepared by: Michael Devanney, economist, NSDA
 Newspaper Content Analysis Monthly Update January 21 Prepared by: Michael Devanney, economist, NSDA devannm@gov.ns.ca The approach of the Atlantic Canadian seal hunt resulted in high media exposure for
Newspaper Content Analysis Monthly Update January 21 Prepared by: Michael Devanney, economist, NSDA devannm@gov.ns.ca The approach of the Atlantic Canadian seal hunt resulted in high media exposure for
Country Coverage. Company Coverage
 Global Heart Valve Devices Market: Analysis By Procedure (Replacement & Repair), Replacement Procedure By Technique (Mechanical, Bioprosthetic & TAVR), Repair Procedure By Technique (Surgical, Balloon
Global Heart Valve Devices Market: Analysis By Procedure (Replacement & Repair), Replacement Procedure By Technique (Mechanical, Bioprosthetic & TAVR), Repair Procedure By Technique (Surgical, Balloon
Drug development of highly potent therapeutic peptides Regulated microdose Bioanalysis
 Drug development of highly potent therapeutic peptides Regulated microdose Bioanalysis Magnus Knutsson, Director, Bioanalysis LC-MS/MS Ferring Pharmaceuticals A/S Copenhagen, Denmark Presented at EBF Open
Drug development of highly potent therapeutic peptides Regulated microdose Bioanalysis Magnus Knutsson, Director, Bioanalysis LC-MS/MS Ferring Pharmaceuticals A/S Copenhagen, Denmark Presented at EBF Open
Safety Pharmacology Post Meeting Survey 2014
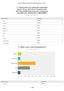 Q1 Please enter your personal information below. (If you want to be included in the 2014 Annual Meeting survey for a chance to win $200, this information is REQUIRED) Answered: 132 Skipped: 14 Name: Company:
Q1 Please enter your personal information below. (If you want to be included in the 2014 Annual Meeting survey for a chance to win $200, this information is REQUIRED) Answered: 132 Skipped: 14 Name: Company:
REGULATION AND HTA OF MEDICAL DEVICES IN EU: WHAT CAN WE LEARN?
 REGULATION AND HTA OF MEDICAL DEVICES IN EU: WHAT CAN WE LEARN? Rosanna Tarricone, PhD Associate Dean, Government Health and Non Profit Division Issue Panel Opportunities and challenges in international
REGULATION AND HTA OF MEDICAL DEVICES IN EU: WHAT CAN WE LEARN? Rosanna Tarricone, PhD Associate Dean, Government Health and Non Profit Division Issue Panel Opportunities and challenges in international
Global E-Cigarette & Vaporizer Market Research Report - Forecast 2011 to 2021
 Report Information More information from: https://www.marketresearchfuture.com/reports/949 Global E-Cigarette & Vaporizer Market Research Report - Forecast 2011 to 2021 Report / Search Code: MRFR/F-B &
Report Information More information from: https://www.marketresearchfuture.com/reports/949 Global E-Cigarette & Vaporizer Market Research Report - Forecast 2011 to 2021 Report / Search Code: MRFR/F-B &
Strategy to move from accelerated burden reduction to malaria elimination in the GMS by 2030
 Strategy to move from accelerated burden reduction to malaria elimination in the GMS by 2030 Dr Walter M Kazadi Coordinator Regional Hub Emergency Response to Artemisinin Resistance Regional Hub GMS MMV
Strategy to move from accelerated burden reduction to malaria elimination in the GMS by 2030 Dr Walter M Kazadi Coordinator Regional Hub Emergency Response to Artemisinin Resistance Regional Hub GMS MMV
Bio-Rad Laboratories HEMOGLOBIN Testing Bio-Rad A1c. VARIANT II TURBO Link System. Fully-Automated HbA 1c Testing
 Bio-Rad Laboratories HEMOGLOBIN Testing Bio-Rad A1c VARIANT II TURBO Link System Fully-Automated HbA 1c Testing Bio-Rad Laboratories HEMOGLOBIN Testing VARIANT II TURBO Link System Efficient streamlined
Bio-Rad Laboratories HEMOGLOBIN Testing Bio-Rad A1c VARIANT II TURBO Link System Fully-Automated HbA 1c Testing Bio-Rad Laboratories HEMOGLOBIN Testing VARIANT II TURBO Link System Efficient streamlined
Challenges in Development and Validation of an Intracellular Cytokine Staining assay
 Challenges in Development and Validation of an Intracellular Cytokine Staining assay Jenny Hendriks, Crucell Hatching @ EBF, Brussels, June 202 www.crucell.com Vaccines vs Protein therapeutics Protein
Challenges in Development and Validation of an Intracellular Cytokine Staining assay Jenny Hendriks, Crucell Hatching @ EBF, Brussels, June 202 www.crucell.com Vaccines vs Protein therapeutics Protein
How a Signs of Safety approach is changing practice in Norfolk. Andrea Brown Principal Social Worker Community Care- Live Tuesday 10 th May 2016
 How a Signs of Safety approach is changing practice in Norfolk Andrea Brown Principal Social Worker Community Care- Live Tuesday 10 th May 2016 The Norfolk Context 2014 OFSTED What did we need to do? Address
How a Signs of Safety approach is changing practice in Norfolk Andrea Brown Principal Social Worker Community Care- Live Tuesday 10 th May 2016 The Norfolk Context 2014 OFSTED What did we need to do? Address
Patient and Public Involvement in JPND Research
 Patient and Public Involvement in JPND Research A user-friendly guide for applicants to JPND Calls for proposals January 5 2015 Created in conjunction with DenDRoN, United Kingdom Purpose The purpose of
Patient and Public Involvement in JPND Research A user-friendly guide for applicants to JPND Calls for proposals January 5 2015 Created in conjunction with DenDRoN, United Kingdom Purpose The purpose of
Clinical Trials in Third Countries
 Clinical Trials in Third Countries meeting, 29 January 2015 Presented by Fergus Sweeney European Medicines Agency An agency of the European Union Overview Distribution of clinical trials submitted in support
Clinical Trials in Third Countries meeting, 29 January 2015 Presented by Fergus Sweeney European Medicines Agency An agency of the European Union Overview Distribution of clinical trials submitted in support
AAM Fall Tech Conference November 6-8, 2017 Rockville, MD
 AAM Fall Tech Conference 2017 November 6-8, 2017 Rockville, MD Excipients Breakout Session Highlights of FDA-USP workshop on Critical Importance of Excipients in Drug Development Why Excipients are Important
AAM Fall Tech Conference 2017 November 6-8, 2017 Rockville, MD Excipients Breakout Session Highlights of FDA-USP workshop on Critical Importance of Excipients in Drug Development Why Excipients are Important
April 17, 2018 City Council Workshop
 April 17, 2018 City Council Workshop Community Engagement Plan 5TH AVENUE DEVELOPMENT COMMUNITY ENGAGEMENT OUTREACH & INPUT OPPORTUNITIES AS OF 12/19 OUTREACH & INPUT OPPORTUNITIES AS OF 4/17 254 ENEWSLETTERS
April 17, 2018 City Council Workshop Community Engagement Plan 5TH AVENUE DEVELOPMENT COMMUNITY ENGAGEMENT OUTREACH & INPUT OPPORTUNITIES AS OF 12/19 OUTREACH & INPUT OPPORTUNITIES AS OF 4/17 254 ENEWSLETTERS
Academic Insights for Biomarker Priorities and Candidate Pilot Project(s)
 Academic Panel Session Academic Insights for Biomarker Priorities and Candidate Pilot Project(s) Moderators: Dr. Chirag Parikh (Yale) Dr. Kumar Sharma (UCSD) Panelists: Dr. Ronald Perrone (Tufts Medical
Academic Panel Session Academic Insights for Biomarker Priorities and Candidate Pilot Project(s) Moderators: Dr. Chirag Parikh (Yale) Dr. Kumar Sharma (UCSD) Panelists: Dr. Ronald Perrone (Tufts Medical
Health Equity Workgroup. January 18, 2018
 Health Equity Workgroup January 18, 2018 Health Equity Workgroup Co-Chairs Fleda Mask Jackson, PhD Founder, Save 100 Babies President and CEO, Majaica, LLC University Affiliate, Columbia University Arthur
Health Equity Workgroup January 18, 2018 Health Equity Workgroup Co-Chairs Fleda Mask Jackson, PhD Founder, Save 100 Babies President and CEO, Majaica, LLC University Affiliate, Columbia University Arthur
MARKET NEWS for pig meat
 MARKET NEWS for pig meat Market analysis 3 December Week 49 MARKET SITUATION Europe: Trading in legs and other cuts is at unchanged prices this week. UK: Prices are reported as stable. Demand is reported
MARKET NEWS for pig meat Market analysis 3 December Week 49 MARKET SITUATION Europe: Trading in legs and other cuts is at unchanged prices this week. UK: Prices are reported as stable. Demand is reported
The Investigation of Factors Contributing to Immunosuppressant Drugs Response Variability in LC-MS/MS Analysis
 The Investigation of Factors Contributing to Immunosuppressant Drugs Variability in LC-MS/MS Analysis Joseph Herman, Dayana Argoti, and Sarah Fair Thermo Fisher Scientific, Franklin, MA, USA Overview Purpose:
The Investigation of Factors Contributing to Immunosuppressant Drugs Variability in LC-MS/MS Analysis Joseph Herman, Dayana Argoti, and Sarah Fair Thermo Fisher Scientific, Franklin, MA, USA Overview Purpose:
Regional Activities Department
 Regional Activities Department Annual Meeting of the OIE Regional and Sub-Regional Representations PARIS, OIE Headquarters, 22 25 October 2013 Dr François CAYA, Head of the Department RAD Team (Oct.2013)
Regional Activities Department Annual Meeting of the OIE Regional and Sub-Regional Representations PARIS, OIE Headquarters, 22 25 October 2013 Dr François CAYA, Head of the Department RAD Team (Oct.2013)
Assessing Change with IIS. Steve Robison Oregon Immunization Program
 Assessing Change with IIS Steve Robison Oregon Immunization Program Assessing Change with IIS Steve Robison Oregon Immunization Program Overview IIS Use in Public Health Static vs Dynamic Assessment Working
Assessing Change with IIS Steve Robison Oregon Immunization Program Assessing Change with IIS Steve Robison Oregon Immunization Program Overview IIS Use in Public Health Static vs Dynamic Assessment Working
AAPS Advances in the Pharmaceutical Sciences Series
 AAPS Advances in the Pharmaceutical Sciences Series Volume 26 Editors-in-chief Daan J.A. Crommelin, Utrecht University, Utrecht, Utrecht, The Netherlands Robert A. Lipper, Back Cove Pharma, LLC, Waldoboro,
AAPS Advances in the Pharmaceutical Sciences Series Volume 26 Editors-in-chief Daan J.A. Crommelin, Utrecht University, Utrecht, Utrecht, The Netherlands Robert A. Lipper, Back Cove Pharma, LLC, Waldoboro,
White Paper Timmerman, White, Cobb, de Vries, Thomas & van Baar
 White Paper Mini Focus Issue: Dried blood spots For reprint orders, please contact reprints@future-science.com Update of the EBF recommendation for the use of DBS in regulated bioana lysis integrating
White Paper Mini Focus Issue: Dried blood spots For reprint orders, please contact reprints@future-science.com Update of the EBF recommendation for the use of DBS in regulated bioana lysis integrating
Slide 1. International Operations update. Mike Doustdar EVP International Operations. YASMIN FIEDLER, Germany Yasmin has type 1 diabetes
 Slide 1 International Operations update Mike Doustdar EVP International Operations YASMIN FIEDLER, Germany Yasmin has type 1 diabetes Slide 2 Forward-looking statements Novo Nordisk s reports filed with
Slide 1 International Operations update Mike Doustdar EVP International Operations YASMIN FIEDLER, Germany Yasmin has type 1 diabetes Slide 2 Forward-looking statements Novo Nordisk s reports filed with
Strengthening Veterinary Services in Asia
 Strengthening Veterinary Services in Asia 4 th Steering Committee Highly Pathogenic Emerging and Re-Emerging Diseases (HPED) Programme Session 3 Progress on the OIE component Tokyo (Japan) 16 July 2013
Strengthening Veterinary Services in Asia 4 th Steering Committee Highly Pathogenic Emerging and Re-Emerging Diseases (HPED) Programme Session 3 Progress on the OIE component Tokyo (Japan) 16 July 2013
February 23, Q4 and Year-End 2016 Financial Results
 February 23, 2017 Q4 and Year-End 2016 Financial Results 2 RETHINKING CNS Agenda Today s Speakers Paul Cox, Senior Director, Investor Relations Jeff Jonas, M.D., Chief Executive Officer Jim Doherty, Ph.D.,
February 23, 2017 Q4 and Year-End 2016 Financial Results 2 RETHINKING CNS Agenda Today s Speakers Paul Cox, Senior Director, Investor Relations Jeff Jonas, M.D., Chief Executive Officer Jim Doherty, Ph.D.,
2007 Multiple Primary and Histology Coding Rules
 National Cancer Institute 2007 Multiple Primary and Histology Coding Rules NCI SEER for NAACCR 2006 Conference U.S. DEPARTMENT OF HEALTH AND HUMAN SERVICES Regina, Saskatchewan June 15, 2006 National Institutes
National Cancer Institute 2007 Multiple Primary and Histology Coding Rules NCI SEER for NAACCR 2006 Conference U.S. DEPARTMENT OF HEALTH AND HUMAN SERVICES Regina, Saskatchewan June 15, 2006 National Institutes
CONTROL CHART METHODOLOGY
 Center for Performance Sciences, 2001 CONTROL CHART METHODOLOGY Statistical Process Control (SPC) Analysis Statistical process control is a tool (e.g., control chart) used to help understand any process
Center for Performance Sciences, 2001 CONTROL CHART METHODOLOGY Statistical Process Control (SPC) Analysis Statistical process control is a tool (e.g., control chart) used to help understand any process
ADULT IMMUNIZATION QUALITY IMPROVEMENT BEST PRACTICES COLLABORATIVE Applications are due by close of business (5:30pm, PDT), October 24, 2014
 ADULT IMMUNIZATION QUALITY IMPROVEMENT BEST PRACTICES COLLABORATIVE Applications are due by close of business (5:30pm, PDT), October 24, 2014 The American Medical Group Foundation (AMGF), AMGA s Anceta
ADULT IMMUNIZATION QUALITY IMPROVEMENT BEST PRACTICES COLLABORATIVE Applications are due by close of business (5:30pm, PDT), October 24, 2014 The American Medical Group Foundation (AMGF), AMGA s Anceta
Essential Oil & Aromatherapy Market Research Report- Forecast to 2023
 Report Information More information from: https://www.marketresearchfuture.com/reports/3319 Essential Oil & Aromatherapy Market Research Report- Forecast to 2023 Report / Search Code: MRFR/F-B & N/2386-HCRR
Report Information More information from: https://www.marketresearchfuture.com/reports/3319 Essential Oil & Aromatherapy Market Research Report- Forecast to 2023 Report / Search Code: MRFR/F-B & N/2386-HCRR
Discussion Meeting for MCP-Mod Qualification Opinion Request. Novartis 10 July 2013 EMA, London, UK
 Discussion Meeting for MCP-Mod Qualification Opinion Request Novartis 10 July 2013 EMA, London, UK Attendees Face to face: Dr. Frank Bretz Global Statistical Methodology Head, Novartis Dr. Björn Bornkamp
Discussion Meeting for MCP-Mod Qualification Opinion Request Novartis 10 July 2013 EMA, London, UK Attendees Face to face: Dr. Frank Bretz Global Statistical Methodology Head, Novartis Dr. Björn Bornkamp
Global Invisible Braces Market: Trends, Opportunities and Forecasts ( )
 Global Invisible Braces Market: Trends, Opportunities and Forecasts (2016-2021) By Types Clear Aligners, Ceramic, Lingual By Region Americas, Europe, Middle East, Africa, Asia-Pacific By Country- USA,
Global Invisible Braces Market: Trends, Opportunities and Forecasts (2016-2021) By Types Clear Aligners, Ceramic, Lingual By Region Americas, Europe, Middle East, Africa, Asia-Pacific By Country- USA,
MARKET NEWS for pig meat
 MARKET NEWS for pig meat Market analysis 24th September 2018 Week 39 MARKET SITUATION Europe: Trading in fresh legs and other cuts is at unchanged prices. UK: Prices are reported as stable. Sales activity
MARKET NEWS for pig meat Market analysis 24th September 2018 Week 39 MARKET SITUATION Europe: Trading in fresh legs and other cuts is at unchanged prices. UK: Prices are reported as stable. Sales activity
Role of the UN agencies in the Prevention and control of NCDs Dr Jacob Kumaresan Executive Director WHO office at the United Nations
 Role of the UN agencies in the Prevention and control of NCDs Dr Jacob Kumaresan Executive Director WHO office at the United Nations 27 February 2013, SEA Regional Consultation on NCDs New perspectives
Role of the UN agencies in the Prevention and control of NCDs Dr Jacob Kumaresan Executive Director WHO office at the United Nations 27 February 2013, SEA Regional Consultation on NCDs New perspectives
Activities of the OIE Regional Commission for Asia, the Far East and Oceania
 Activities of the OIE Regional Commission for Asia, the Far East and Oceania 28 th Conference of the OIE Regional Commission for Asia, the Far East and Oceania Cebu, 19 November 2013 Dr Zhang Zhongqui
Activities of the OIE Regional Commission for Asia, the Far East and Oceania 28 th Conference of the OIE Regional Commission for Asia, the Far East and Oceania Cebu, 19 November 2013 Dr Zhang Zhongqui
Joint FAO/WHO Food Standards Programme Food and Agriculture Organization of the United Nations Lusaka, Zambia, 3-5 July 2002
 FAO/WHO CODEX ALIMENTARIUS COMMISSION (CODEX) Joint FAO/WHO Food Standards Programme Food and Agriculture Organization of the United Nations Lusaka, Zambia, 3-5 July 2002 1. Introducing Codex Alimentarius
FAO/WHO CODEX ALIMENTARIUS COMMISSION (CODEX) Joint FAO/WHO Food Standards Programme Food and Agriculture Organization of the United Nations Lusaka, Zambia, 3-5 July 2002 1. Introducing Codex Alimentarius
World Renderers Organisation Report
 World Renderers Organisation Report Tim Juzefowicz, 1st vice President NRA 81 st National Convention Palm Springs October 22nd 2014 WRO Representing the Animal by-product processing Sector around the world
World Renderers Organisation Report Tim Juzefowicz, 1st vice President NRA 81 st National Convention Palm Springs October 22nd 2014 WRO Representing the Animal by-product processing Sector around the world
IAPP KnowledgeNet Chapter Chair Manual 2018
 IAPP KnowledgeNet Chapter Chair Manual 2018 CHAIR ROLES & EXPECTATIONS IAPP RESPONSIBILITIES PLANNING A CHAPTER MEETING INFORMATION & TIPS FOR HOST VENUE INFORMATION & TIPS FOR SPEAKERS OTHER CHAPTER VOLUNTEERS
IAPP KnowledgeNet Chapter Chair Manual 2018 CHAIR ROLES & EXPECTATIONS IAPP RESPONSIBILITIES PLANNING A CHAPTER MEETING INFORMATION & TIPS FOR HOST VENUE INFORMATION & TIPS FOR SPEAKERS OTHER CHAPTER VOLUNTEERS
New Annex 15 Updated Requirements & Approach to Validation. Presented by Ashley Isbel 10 th August 2015
 New Annex 15 Updated Requirements & Approach to Validation Presented by Ashley Isbel 10 th August 2015 EU GMP Guide Annex 15 Qualification & Validation 2001 Current version of Annex 15 published Nov 2012
New Annex 15 Updated Requirements & Approach to Validation Presented by Ashley Isbel 10 th August 2015 EU GMP Guide Annex 15 Qualification & Validation 2001 Current version of Annex 15 published Nov 2012
GLP Update and Hot Topics
 GLP Update and Hot Topics RQA Annual Conference Mark Goodwin 9 Nov 2016 Introduction International GLP Updates OECD including MAD Agreement EC/ EMA US FDA & US EPA UK GLP Update MHRA MHRA GLP Deficiencies
GLP Update and Hot Topics RQA Annual Conference Mark Goodwin 9 Nov 2016 Introduction International GLP Updates OECD including MAD Agreement EC/ EMA US FDA & US EPA UK GLP Update MHRA MHRA GLP Deficiencies
NICOLE s Shared vision on Sustainable Remediation
 NICOLE s Shared vision on Sustainable Remediation Green Remediation conference 09 November 2009 Copenhagen www.nicole.org 1 Agenda Methodology followed Work completed since Oct 2008 Main lessons learned
NICOLE s Shared vision on Sustainable Remediation Green Remediation conference 09 November 2009 Copenhagen www.nicole.org 1 Agenda Methodology followed Work completed since Oct 2008 Main lessons learned
Placement of Biomarker Analysis in a CLIA or Bioanalytical Laboratory
 Placement of Biomarker Analysis in a CLIA or Bioanalytical Laboratory SQA National Meeting 29Mar2017 Carolyn Eberhardt, Principal Consultant, QC2 Mark Stiles, Senior Consultant, QC2 Comparison of Quality
Placement of Biomarker Analysis in a CLIA or Bioanalytical Laboratory SQA National Meeting 29Mar2017 Carolyn Eberhardt, Principal Consultant, QC2 Mark Stiles, Senior Consultant, QC2 Comparison of Quality
4/10/2017. Placement of Biomarker Analysis in a CLIA or Bioanalytical Laboratory
 Placement of Biomarker Analysis in a CLIA or Bioanalytical Laboratory SQA National Meeting 29Mar2017 Carolyn Eberhardt, Principal Consultant, QC2 Mark Stiles, Senior Consultant, QC2 Comparison of Quality
Placement of Biomarker Analysis in a CLIA or Bioanalytical Laboratory SQA National Meeting 29Mar2017 Carolyn Eberhardt, Principal Consultant, QC2 Mark Stiles, Senior Consultant, QC2 Comparison of Quality
