Quarterly Reports - Message to Our Shareholders
|
|
|
- Patience Ross
- 5 years ago
- Views:
Transcription
1 Quarterly Reports - Message to Our Shareholders The Company is pleased to report record sales and earnings results for the third quarter of fiscal year Biomet has also announced its intent to acquire Interpore Cross International for approximately $280 million. Biomet will finance the purchase price using cash reserves and short-term debt. Interpore's 2003 net sales were approximately $67.5 million. Excluding one-time acquisition-related charges, it is anticipated the acquisition will be neutral to Biomet's diluted earnings for fiscal year ending May 31, 2005 and accretive thereafter. The transaction has been approved by the Boards of both companies and is expected to close during the second calendar quarter of 2004, subject to customary closing conditions. The planned merger will bring together a wealth of complementary products and services, providing a host of spinal solutions for our spinal surgeons and neurosurgeons worldwide. Biomet previously announced it had reached an understanding with Merck KGaA, Darmstadt, Germany, to acquire Merck's 50% interest in the Biomet Merck Joint Venture. The Joint Venture was established in 1998 and created a market-leading European orthopedic company, with current annualized sales of approximately $330 million. The aggregate purchase price offered for Merck's 50% interest was $300 million in cash to be paid from Biomet's cash reserves. It is anticipated that the acquisition will be neutral to Biomet's earnings during the fourth quarter of fiscal year 2004 and the full fiscal year 2005, and will be slightly accretive thereafter. On March 22, 2004, Biomet announced the successful completion of the Biomet Merck Joint Venture acquisition, subject only to the receipt of clearance from the competition authorities in certain European jurisdictions. Biomet's net sales increased 16% during the third quarter to $410,185,000. Gross profit increased 19% to $294,193,000 during the quarter, representing 71.7% of sales. Selling, general and administrative expenses increased 14% to $145,712,000 and represented 35.5% of sales, improving 70 basis points over the third quarter of last year. Operating income increased 28% to $132,589,000 with operating margins at 32.3% of sales. Net income increased 19% to $86,600,000 during the third quarter, while diluted earnings increased 21% to $0.34 per share. Excluding foreign currency, which increased sales by approximately $18 million, net sales during the third quarter increased 11%. Domestic sales increased 11% to $271,520,000 during the quarter and international sales increased 11% on a constant currency basis to $138,665,000. Biomet's direct sales presence in Japan continues to experience excellent market penetration with third quarter revenues of $5.2 million, up 107% over last year's third quarter. During the third quarter of 2004, reconstructive device sales increased 22% to $270,203,000. Worldwide reconstructive device sales increased 15%, constant currency. Knee sales increased 18% worldwide and 15% in the United States during the third quarter. Worldwide knee sales increased 12%, constant currency. Knee products driving growth during the third quarter were the Ascent Primary and Revision Systems, the Biomet OSS (Orthopaedic Salvage System) utilized for oncology and revision procedures, and the minimally invasive Vanguard Unicompartmental Knee System. Following the Company's market-leading role in minimally invasive unicompartmental knees, Biomet continued its pioneering efforts by becoming the first company to introduce minimally invasive total knee instruments to the orthopedic market. During fiscal year 2004, approximately 250 sets of Biomet's Microplasty Minimally Invasive Total Knee Instruments have been introduced, with more than 100 additional sets scheduled for release during the fourth quarter. The proprietary Microplasty Total Knee Instruments are designed to provide the patient with a more rapid recovery by allowing for a substantially reduced incision size along with minimal muscle disruption. Approximately 50 surgeons completed training on the Microplasty Total Knee Instrumentation during the third quarter, bringing the total number of surgeons trained on this specific technique to more than 200 surgeons during this fiscal year. The first live webcast of a Microplasty Total Knee Replacement took place on March 23, James W. Scott,
2 M.D. of Tifton, Georgia performed the procedure, which can be viewed by visiting the website. During the third quarter, Biomet initiated the general launch of the Company's newest and most comprehensive knee system, the Vanguard Complete Knee System. The Vanguard System includes fixedbearing cruciate-retaining and posterior-stabilized versions, with numerous options in development, including mobile-bearing and revision components, as well as a patello-femoral replacement option. During the fourth quarter, Biomet is planning to launch the first 50 sets of Microplasty Vanguard Complete Knee Instruments. Hip sales increased 14% worldwide during the third quarter and 9% in the United States. Worldwide hip sales increased 8%, constant currency. Biomet's hip implant sales faced another tough comparison with last year's third quarter worldwide hip growth of 23%. Leading hip sales, Biomet's metal-on-metal articulation systems and the Company's broad line of complementary cementless stems, particularly the Taperloc, BiMetric and Integral components, continued to experience strong growth. Domestic sales of Biomet's metal-on-metal systems increased 18% during the third quarter and represented 33% of the Company's hip articulation unit sales in the United States. Designed for improved joint stability and increased range of motion, Biomet's M2a -38 hip cup continued to exp erience excellent market acceptance with a sales increase of 29% compared to last year's third quarter. Additionally, the enrollment for the C2a Ringloc ceramic -on-ceramic study is currently nearing completion. Biomet's Microplasty Minimally Invasive Hip Programs (Posterior and Anterior/Lateral Approaches) continued to experience excellent market acceptance and all training labs are overflowing. Approximately 350 sets of Microplasty Hip Instrument sets have been placed during fiscal year During the third quarter, Biomet trained more than 200 surgeons on the Microplasty Minimally Invasive Hip Programs at the Biomet Learning Center in Warsaw and the Orthopedic Learning Center in Chicago. An additional 250 surgeons are scheduled for training during the fourth quarter. Development of the Microplasty Anterior/Supine Approach with related implants and instrumentation will continue during the fourth quarter. Revision hip sales continued to experience significant growth during the third quarter. Biomet's Freedom Constrained Liner, indicated for patients with chronic hip dislocation tendencies, experienced strong market acceptance during the third quarter. With its unique patented design, the Freedom Constrained Liner allows for increased range of motion of up to 110. The MaxTi Modular and Mallory-Head Protrusio Cages, designed for difficult revision cases with substantial bone defects, continue to experience excellent market acceptance. Additionally, the Mallory-Head One-Piece Calcar and Modular Reach Hip Systems are experiencing very strong growth. During the fourth quarter, Biomet will release the ReCap Femoral Resurfacing System, a bone-conserving hemi-hip system indicated for use with patients in the early stages of osteoarthritis. In Europe, the ReCap Total Resurfacing Hip System is currently in its initial launch. Extremity sales increased 27% worldwide during the third quarter and 25% in the United States. Worldwide extremity sales increased 21%, constant currency. Growth in the extremity segment during the third quarter was driven by strong demand for the Copeland Humeral (shoulder) Resurfacing Head and the Liverpool Radial Head Replacement. The Copeland Shoulder is a bone-conserving cementless resurfacing system with excellent long-term clinical results. The Liverpool Radial Head Replacement was developed to restore the anatomy of the natural elbow joint and is used in cases of trauma, post-traumatic deformity and elbow instability. Dental reconstructive implant sales increased 29% worldwide during the third quarter and 19% in the United States. On a constant currency basis, revenues of dental reconstructive implants increased 22% worldwide. The global rollout of 3i's first internal-connection dental reconstructive implant system, the OSSEOTITE Certain Internal Connection Implant System, took place during the second and third quarters of this fiscal year. A primary advantage of 3i's OSSEOTITE Certain System is the 30º incremental rotational adjustment capabilities it offers clinicians, while the competing systems allow for only 60º incremental adjustments. The success of the OSSEOTITE Certain Implant System, along with solid broadbased market growth during the quarter, resulted in another outstanding quarter for 3i.
3 Sales of bone cements and accessories increased 43% worldwide during the third quarter and increased 85% in the United States, further securing Biomet's number two position in the estimated $205 million domestic market. Sales of bone cements and accessories increased 26% worldwide, excluding the effects of foreign currency. Palacos Bone Cement and the complementary Optivac Vacuum Mixing and Delivery System continued to experience excellent growth rates. Additionally, Palacos G antibiotic-loaded bone cement received strong market acceptance during the short time period since the U.S. Food and Drug Administration clearance of this product during the third quarter. Palacos G is comprised of standard Palacos Bone Cement with the addition of Gentamicin antibiotic and is cleared for use in the second stage of a two-stage revision. Palacos bone cement with Gentamicin has been the standard of care in Europe for nearly 35 years. Now, domestic surgeons also have access to the world's most clinically proven antibioticloaded bone cement. The proprietary Generation 4 Bone Cement with VacPac self-contained mixing and delivery system continues to experience excellent market acceptance. The benefits of the Generation 4 System include the elimination of fume exposure during the mixing process, improved consistency and quality of the cement, and reduced operating room time due to the ease of mixing. The GPS (Gravitational Platelet Separation) System continued to experience excellent market acceptance during the third quarter, contributing to the Company's reconstructive sales growth. The GPS System, distributed by Biomet's Cell Factor Technologies Division, requires minimal operator intervention and consistently produces the highest quality concentrate of any platelet system on the market. The GPS System possesses broad potential applications in the reconstructive, plastic and spine segments of the market and promotes more rapid healing of wounds, such as diabetic ulcers and general surgical incisions. Worldwide fixation sales increased 6% to $62,460,000 during the third quarter of fiscal year Lorenz Surgical's craniomaxillofacial fixation sales increased 19% worldwide and 13% in the United States during the quarter. Lorenz is currently introducing several new products to the market, including the RapidFlap SpinDown System for cranial flap fixation and the Titanium RapidFire Screw Delivery System, an innovative product that effectively reduces operating room time. Electrical stimulation sales increased 9% worldwide and 8% in the United States during the third quarter. With the recent approval from the U.S. Food and Drug Administration for the Mechanism of Action expanded label claim, the EBI Bone Healing System and the Pulsed Electromagnetic Field (PEMF) technology can now be more clearly differentiated from competitive systems. EBI's technology has been demonstrated to increase the production of a cascade of bone morphogenic proteins ("BMPs") and other osteopromotive growth factors, which are known to facilitate the healing of fractures and spinal fusions. This is in stark contrast to commercially available BMPs, which deliver a single short-lived growth factor to the fracture or fusion site. External fixation sales were flat worldwide and decreased 2% in the United States during the third quarter. On a constant currency basis, external fixation sales decreased 1% worldwide. Several new external fixation products are currently being introduced, augmenting the DynaFix Vision product line, including MRI compatible clamps and the DynaFix Vision Radiolucent Unilateral Fixator. Internal fixation sales decreased 11% worldwide during the third quarter and decreased 5% in the United States. Worldwide internal fixation sales decreased 15%, constant currency. The Company continues to focus on execution of the transition of the internal fixation business to the EBI business unit. Additionally, EBI is currently developing internal fixation line extensions, including minimally invasive plating options, which are scheduled for launch during the fourth quarter. Worldwide spinal product sales increased 9% to $39,322,000 and increased 8% in the United States during the third quarter of Domestic sales of spinal implant and orthobiological products for the spine increased 20% during the quarter. Sales of spinal stimulation systems increased 4% in the United States. The SpinalPak-II Stimulation System was recently introduced to the market. The SpinalPak-II System is
4 smaller in size and uses rechargeable batteries, eliminating the need for the patient to change batteries on a daily basis, thus providing for uninterrupted treatment contributing to optimal patient compliance. The Array Top-Loading Universal Spinal System was introduced on a limited basis during the third quarter and was extremely well received by EBI's customers. The Array System is EBI's first top-loading system, which opens the door to the deformity segment of the spinal products market. Also, the EBI VuePASS Minimally Invasive Fusion System was in a limited launch during the quarter and OsteoStim Demineralized Bone Matrix was launched with positive results. The development of the lumbar and cervical versions of Regain, EBI's one-piece pyrocarbon artificial disc nucleus replacement system, continues on schedule. The pyrocarbon material has a high level of strength, is biocompatible and extremely resistant to wear. Additionally, EBI is developing lumbar and cervical versions of Rescue, a total disc replacement product. During the third quarter of 2004, sales of the Company's "other products" increased 4% worldwide to $38,200,000 and decreased 2% in the United States. On a constant currency basis, "other product" sales increased 2% worldwide. Arthroscopy sales increased 10% worldwide during the quarter and 7% in the United States. Numerous Arthrotek products received 510(k) clearance from the U.S. Food and Drug Administration during the third quarter, including the resorbable CentraLoc Tibial Fixation device, a cannulated version of the ArthroRivet, the SpeedPass Suture Passer and several items from the MaxBraid product line. Sales of softgoods and bracing products increased 3% worldwide and 2% in the United States during the third quarter. Several new products were recently introduced to the market, including the Alliance Knee Brace, the EBI Gravity Ankle System and the EBI Ulnar Styloid Wrist Brace. We are pleased with the Company's record sales and earnings results during the third quarter and first nine months of fiscal year Strong performance of the Co mpany's reconstructive, spinal hardware and orthobiologics products led revenue growth during the quarter. Adding to Biomet's broad base of products and technologies with proven long-term clinical results, the Company introduced more than 410 new products to the musculoskeletal market during the past 5 years, expanding the availability of new treatment options for surgeons and their patients. Respectfully, Dane A. Miller, Ph.D. President and Chief Executive Officer Niles L. Noblitt Chairman of the Board Quarterly Reports - Featured Market Segment Bone Cements and Accessories The 2004 bone cements and accessories market in the United States is currently estimated to be $205 million with market growth currently in
5 the range of 13-15%. The market is comprised of bone cements at $120 million and accessories at $85 million. This market is experiencing renewed growth as a result of the recent introduction of antibioticloaded bone cements to the domestic market. Biomet entered the domestic bone cements and accessories market in June of 2000, with Palacos Bone Cement and the Optivac Vacuum Mixing and Delivery System. Within Biomet's first year of market introduction, the Company quickly moved into the second largest domestic market share position and continues to capture market share. The Company's antibiotic-loaded bone cement, Palacos G, first became available in the United States during the third quarter of fiscal year 2004 with the United States Food and Drug Administration clearance of the product. Palacos G bone cement is a combination of standard Palacos Bone Cement and Gentamicin antibiotic and is cleared for use in the second stage of a two-stage revision procedure. Palacos bone cement with Gentamicin has been the standard of care in Europe for nearly 35 years. Palacos G bone cement is the most clinically proven antibiotic-loaded bone cement available in the world. The Company's sales of bone cements and accessories increased 85% in the United States and 43% worldwide during the third quarter of fiscal year Generation 4 Bone Cement with the VacPac Delivery System continues to experience excellent market acceptance due to its key differentiating factors. The proprietary, self-contained VacPac System is designed to promote consistency and integrity of the cement, eliminate exposure to fumes during mixing, and reduce operating room time due to the ease of the mixing and delivery process. Generation 4 Bone Cement with the VacPac Delivery System was first introduced to the market during fiscal year The Company distributes bone cements and accessories in the United States through its orthopedic reconstructive device salesforce of approximately 550 representatives and throughout Europe and the rest of the world through a salesforce in excess of 600 representatives. Biomet is committed to offering surgeons a broad spectrum of products and technologies, as is evidenced by the Company's more than 410 new products which have been introduced to the market during the past five years. Quarterly Reports - Consolidated Statements of Income Fiscal Year rd Quarter For the periods ended February 29, 2004 and 2003 (in thousands, except per share amounts) Nine Months Three Months Net sales 1,168,065 1,013, , ,042 Cost of sales 330, , , ,636 Gross profit 837, , , ,406 Selling, general and administrative expenses 414, , , ,990 Research and development expense 46,450 40,262 15,892 14,555
6 Operating income 376, , , ,861 Other income, net 10,260 15,947 3,570 9,145 Income before income taxes and minority interest 386, , , ,006 Provision for income taxes 134, ,307 47,430 39,169 Income before minority interest 252, ,964 88,729 73,837 Minority interest 6,273 5,010 2,129 1,243 Net Income 245, ,954 86,600 72,594 Earnings per share: Basic $.96 $.80 $.34 $.28 Diluted $.95 $.80 $.34 $.28 Shares used in the computation of earnings per share: Basic 255, , , ,929 Diluted 257, , , ,824 Cash dividends per common share $.15 $ Quarterly Reports - Sales Analysis Fiscal Year rd Quarter Quarter Ended February 29, 2004 For the three months ended February 29, 2004 and 2003 (in thousands) Domestic/Foreign Sales U.S. sales $ 271,520 $ 244,838 Foreign sales 138, ,204 Segment Sales Reconstructive $ 270,203 $ 222,253 Fixation 62,460 59,061 Spinal products 39,322 36,157 Other 38,200 36,571 Quarterly Reports - Market Value of Common Shares Fiscal Year rd Quarter Quarter Ended February 29, 2004 The following table shows the quarterly range of high and low sale prices for the Company s Common Shares as reported by Nasdaq Stock Market (Biomet s fiscal year commences on June 1 and ends on May 31.) Stock Symbol: BMET. The amount reflects inter-dealer prices, without retail mark-up, mark-down or commission. Record holders of outstanding Common Shares February 29, st Qtr. 2nd Qtr. 3rd Qtr. 4th Qtr.
7 2004 High Low High Low High Low Quarterly Reports - Consolidated Balance Sheets and Consolidated Statements of Cash Flow Fiscal Year rd Quarter Quarter Ended February 29, 2004 Assets 02/29/ /31/2003 Current assets: Cash and cash equivalents $376,923 $225,650 Investments 13,945 37,337 Accounts and notes receivable, net 468, ,095 Inventories 383, ,270 Deferred income taxes 56,642 54,262 Prepaid expenses and other 24,987 20,141 Total current assets 1,322,194 1,111,755 Property, plant and equipment, at cost 525, ,965 Less, Accumulated depreciation 258, ,519 Property, plant and equipment, net 267, ,446 Investments 72, ,607 Goodwill, net 130, ,706 Intangible assets, net 10,033 10,874 Other assets 15,089 13,781 Total assets $1,817,925 $1,672,169 Liabilities and Shareholders Equity Current liabilities: Short-term borrowings $122,465 $114,120 Accounts payable 38,769 42,106 Accrued income taxes 17,746 12,453 Accrued wages and commissions 45,964 12,453 Other accrued expenses 63,598 54,260 Total current liabilities 288, ,654 Long-term liabilities: Deferred federal income taxes 7,455 7,031
8 Other liabilities Total liabilities 295, ,147 Minority interest 118, ,888 Contingencies (Note 0) Shareholders equity: Common shares 161, ,931 Additional paid-in capital 54,155 54,081 Retained earnings 1,159,125 1,100,462 Accumulated other comprehensive loss 29,328 (10,340) Total shareholders equity 1,403,767 1,286,134 Total liabilities and $1,817,925 $1,672,169 shareholders equity Consolidated Statements of Cash Flow For the nine months ended February 29, 2004 and 2003 (in thousands) Cash flows from (used in) investing activities: Proceeds from sales and maturities 215, ,717 of investments Purchases of investments (106,694) ( 43,899) Capital expenditures ( 42,594) ( 41,766) Other ( 1,587) ( 4,178) Net cash from (used in) investing activities 64,411 14,874 Cash flows from (used in) financing activities: Increase (decrease) in short-term (2,254) 6,655 borrowings, net Issuance of common shares 21,874 13,897 Cash dividends (38,604) (26,431) Purchase of common shares (152,020) (187,115) Net cash used in financing activities (171,004) (192,994) Effect of exchange rate changes on cash 3,773 7,222 Increase in cash and cash equivalents 151,273 13,480 Cash and cash equivalents, beginning 225, ,297 of year Cash and cash equivalents, end of period $376,923 $167,777 Quarterly Reports - Quarterly Graphs Fiscal Year rd Quarter - Quarter Ended February 29, 2004
9
First Albany Orthopedics Conference Marina Village, San Diego February 13, 2007 Orthofix International
 First Albany Orthopedics Conference Marina Village, San Diego February 13, 2007 Orthofix International Tom Hein, CFO Safe Harbor Statement Except for historical information contained herein, the statements
First Albany Orthopedics Conference Marina Village, San Diego February 13, 2007 Orthofix International Tom Hein, CFO Safe Harbor Statement Except for historical information contained herein, the statements
Interim Report 1 January September 2017
 2014-01-01 2014-12 Interim Report 1 January 2017 30 September 2017 Cantargia AB 556791-6019 Cantargia refers to Cantargia AB (publ), corporate ID number 556791-6019. Clinical trial initiated Highlights
2014-01-01 2014-12 Interim Report 1 January 2017 30 September 2017 Cantargia AB 556791-6019 Cantargia refers to Cantargia AB (publ), corporate ID number 556791-6019. Clinical trial initiated Highlights
Argos Therapeutics Reports Fourth Quarter and Year-End 2013 Financial Results
 March 27, 2014 Argos Therapeutics Reports Fourth Quarter and Year-End 2013 Financial Results - AGS-003 Phase 3 ADAPT Trial on Track to Complete Enrollment This Year - - AGS-004-Induced Immune Responses
March 27, 2014 Argos Therapeutics Reports Fourth Quarter and Year-End 2013 Financial Results - AGS-003 Phase 3 ADAPT Trial on Track to Complete Enrollment This Year - - AGS-004-Induced Immune Responses
Orthopaedics Business. Dave Illingworth, President February
 Orthopaedics Business Dave Illingworth, President February 23 2005 1 Smith & Nephew Orthopaedics Attractive market Strong performance Momentum 2 Attractive market Demographics - 2x Growth by 2020 Active
Orthopaedics Business Dave Illingworth, President February 23 2005 1 Smith & Nephew Orthopaedics Attractive market Strong performance Momentum 2 Attractive market Demographics - 2x Growth by 2020 Active
ST. JUDE MEDICAL / THORATEC TRANSACTION HIGHLIGHTS July 22, 2015
 ST. JUDE MEDICAL / THORATEC TRANSACTION HIGHLIGHTS July 22, 2015 1 FORWARD LOOKING STATEMENT This presentation contains forward-looking statements within the meaning of the Private Securities Litigation
ST. JUDE MEDICAL / THORATEC TRANSACTION HIGHLIGHTS July 22, 2015 1 FORWARD LOOKING STATEMENT This presentation contains forward-looking statements within the meaning of the Private Securities Litigation
ObsEva Reports Third Quarter 2017 Financial Results and Provides Business Update
 ObsEva Reports Third Quarter 2017 Financial Results and Provides Business Update - All Three Development Compounds Progressing with Key Clinical Milestones Over the Next 12 Months - Geneva, Switzerland
ObsEva Reports Third Quarter 2017 Financial Results and Provides Business Update - All Three Development Compounds Progressing with Key Clinical Milestones Over the Next 12 Months - Geneva, Switzerland
Q Investor Kit JANUARY-MARCH 2014
 Q1 2014 Investor Kit JANUARY-MARCH 2014 Swedish Match reporting segments Snus and moist snuff Snus (Scandinavia and US) Moist snuff (US) SMPM International Other tobacco products Cigars (US) Chewing tobacco
Q1 2014 Investor Kit JANUARY-MARCH 2014 Swedish Match reporting segments Snus and moist snuff Snus (Scandinavia and US) Moist snuff (US) SMPM International Other tobacco products Cigars (US) Chewing tobacco
Oncolytics Biotech Inc. Announces Third Quarter 2010 Results
 November 10, 2010 Oncolytics Biotech Announces Third Quarter 2010 Results CALGARY, Nov. 10 /PRNewswire-FirstCall/ - Oncolytics Biotech (TSX:ONC, NASDAQ:ONCY) ("Oncolytics" or the "Company") today announced
November 10, 2010 Oncolytics Biotech Announces Third Quarter 2010 Results CALGARY, Nov. 10 /PRNewswire-FirstCall/ - Oncolytics Biotech (TSX:ONC, NASDAQ:ONCY) ("Oncolytics" or the "Company") today announced
Emisphere Technologies, Inc. Announces 2008 Second Quarter Financial Results
 August 11, 2008 Emisphere Technologies, Inc. Announces 2008 Second Quarter Financial Results Conference Call/Webcast to be Held Monday, August 11 at 10:00 AM EDT CEDAR KNOLLS, N.J., Aug 11, 2008 /PRNewswire-FirstCall
August 11, 2008 Emisphere Technologies, Inc. Announces 2008 Second Quarter Financial Results Conference Call/Webcast to be Held Monday, August 11 at 10:00 AM EDT CEDAR KNOLLS, N.J., Aug 11, 2008 /PRNewswire-FirstCall
Q Investor Kit JANUARY-JUNE 2014
 Q2 2014 Investor Kit JANUARY-JUNE 2014 Swedish Match reporting segments Snus and moist snuff Snus (Scandinavia and US) Moist snuff (US) SMPM International Other tobacco products Cigars (US) Chewing tobacco
Q2 2014 Investor Kit JANUARY-JUNE 2014 Swedish Match reporting segments Snus and moist snuff Snus (Scandinavia and US) Moist snuff (US) SMPM International Other tobacco products Cigars (US) Chewing tobacco
HALOZYME REPORTS SECOND QUARTER 2018 RESULTS
 Contacts: Robert H. Uhl Managing Director Westwicke Partners, LLC 858-356-5932 robert.uhl@westwicke.com Laurie Stelzer 858-704-8222 ir@halozyme.com FOR IMMEDIATE RELEASE HALOZYME REPORTS SECOND QUARTER
Contacts: Robert H. Uhl Managing Director Westwicke Partners, LLC 858-356-5932 robert.uhl@westwicke.com Laurie Stelzer 858-704-8222 ir@halozyme.com FOR IMMEDIATE RELEASE HALOZYME REPORTS SECOND QUARTER
For personal use only
 HY18 Result Presentation 13 February 2018 Dig Howitt Brent Cubis CEO & President CFO HY18 Result highlights Strong momentum across developed markets continues Developed market unit growth up 12% Strengthening
HY18 Result Presentation 13 February 2018 Dig Howitt Brent Cubis CEO & President CFO HY18 Result highlights Strong momentum across developed markets continues Developed market unit growth up 12% Strengthening
January 30, 2018 Dow Wilson President and Chief Executive Officer
 Acquisition of SIRTeX January 30, 2018 Dow Wilson President and Chief Executive Officer This presentation is intended exclusively for investors. It is not intended for use in Sales or Marketing. Forward-Looking
Acquisition of SIRTeX January 30, 2018 Dow Wilson President and Chief Executive Officer This presentation is intended exclusively for investors. It is not intended for use in Sales or Marketing. Forward-Looking
FY2007 Consolidated Financial Overview
 FY2007 Consolidated Financial Overview CHUGAI PHARMACEUTICAL CO., LTD. Executive Vice President and CFO Ryuzo Kodama January 30/31, 2008 Forward-Looking Statements This presentation may include forward-looking
FY2007 Consolidated Financial Overview CHUGAI PHARMACEUTICAL CO., LTD. Executive Vice President and CFO Ryuzo Kodama January 30/31, 2008 Forward-Looking Statements This presentation may include forward-looking
THIRD QUARTER REPORT 2011 (UNAUDITED)
 THIRD QUARTER REPORT 2011 (UNAUDITED) Kronprinsesse Märthas pl. 1, Vika, 0166 Oslo, Norway. Tel: + 47 23 01 09 60, www.bionorpharma.com Page 1 THIRD QUARTER REPORT 2011 HIGHLIGHTS On the 25th August Bionor
THIRD QUARTER REPORT 2011 (UNAUDITED) Kronprinsesse Märthas pl. 1, Vika, 0166 Oslo, Norway. Tel: + 47 23 01 09 60, www.bionorpharma.com Page 1 THIRD QUARTER REPORT 2011 HIGHLIGHTS On the 25th August Bionor
Agreement to Acquire Spinal Kinetics, Inc. March 15, 2018
 Agreement to Acquire Spinal Kinetics, Inc. 1 March 15, 2018 2 SAFE HARBOR STATEMENT AND USE OF NON GAAP MEASURES WHY SPINAL KINETICS? The Spinal Kinetics M6 artificial discs further round out Orthofix
Agreement to Acquire Spinal Kinetics, Inc. 1 March 15, 2018 2 SAFE HARBOR STATEMENT AND USE OF NON GAAP MEASURES WHY SPINAL KINETICS? The Spinal Kinetics M6 artificial discs further round out Orthofix
MAKO Surgical Corp. January MAKO Surgical Corp
 MAKO Surgical Corp. January 2012 MAKO Surgical Corp. 2012 1 Forward Looking Statement This presentation contains forward-looking statements regarding, among other things, statements related to expectations,
MAKO Surgical Corp. January 2012 MAKO Surgical Corp. 2012 1 Forward Looking Statement This presentation contains forward-looking statements regarding, among other things, statements related to expectations,
Hyundai Motor Company 1 st Quarter 2011 Business Results. April 28, 2011
 Hyundai Motor Company 1 st Quarter 2011 Business Results April 28, 2011 Cautionary Statement with Respect to Forward-Looking Statements In the presentation that follows and in related comments by Hyundai
Hyundai Motor Company 1 st Quarter 2011 Business Results April 28, 2011 Cautionary Statement with Respect to Forward-Looking Statements In the presentation that follows and in related comments by Hyundai
Q Investor Kit January December 2014
 Q4 2014 Investor Kit January December 2014 Highlights for Q4 Sales up for all product areas and operating profit from product areas up by 8% Scandinavian hoarding for snus prior to tax increase in January
Q4 2014 Investor Kit January December 2014 Highlights for Q4 Sales up for all product areas and operating profit from product areas up by 8% Scandinavian hoarding for snus prior to tax increase in January
1Q Fornebu, April 29, 2015 Luis Araujo and Svein Stoknes
 1Q 2015 Fornebu, April 29, 2015 Luis Araujo and Svein Stoknes Forward-Looking Statements and Copyright This Presentation includes and is based, inter alia, on forward-looking information and statements
1Q 2015 Fornebu, April 29, 2015 Luis Araujo and Svein Stoknes Forward-Looking Statements and Copyright This Presentation includes and is based, inter alia, on forward-looking information and statements
Q Investor Kit JANUARY-JUNE 2013
 Q2 2013 Investor Kit JANUARY-JUNE 2013 1 Swedish Match reporting segments Snus and snuff - Swedish snus (Scandinavia and US) - Moist snuff (US) - SMPM International Other tobacco products - Cigars (US)
Q2 2013 Investor Kit JANUARY-JUNE 2013 1 Swedish Match reporting segments Snus and snuff - Swedish snus (Scandinavia and US) - Moist snuff (US) - SMPM International Other tobacco products - Cigars (US)
Clovis Oncology Announces Q Operating Results and Corporate Update. November 3, :05 PM ET
 Clovis Oncology Announces Q3 2016 Operating Results and Corporate Update November 3, 2016 4:05 PM ET Rucaparib New Drug Application (NDA) accepted for Priority Review in the treatment of advanced BRCA-mutant
Clovis Oncology Announces Q3 2016 Operating Results and Corporate Update November 3, 2016 4:05 PM ET Rucaparib New Drug Application (NDA) accepted for Priority Review in the treatment of advanced BRCA-mutant
Table of Contents: JULY TH EDITION
 Table of Contents: Table of Contents of the worldwide Orthopaedic Contract manufacturing Market 2016-2021 & Top 100 player profiles 275+ pages 450+ exhibits July 2017 5 th edition I- Worldwide orthopaedic
Table of Contents: Table of Contents of the worldwide Orthopaedic Contract manufacturing Market 2016-2021 & Top 100 player profiles 275+ pages 450+ exhibits July 2017 5 th edition I- Worldwide orthopaedic
NewLink Genetics Corporation Provides Operational Update and Reports Second Quarter 2015 Financial Results
 July 31, 2015 NewLink Genetics Corporation Provides Operational Update and Reports Second Quarter 2015 Financial Results -Management to Host Conference Call Today at 8:30 a.m. ET- AMES, Iowa, July 31,
July 31, 2015 NewLink Genetics Corporation Provides Operational Update and Reports Second Quarter 2015 Financial Results -Management to Host Conference Call Today at 8:30 a.m. ET- AMES, Iowa, July 31,
METVIX PDT ON THE MARKET IN GERMANY AND UK
 METVIX PDT ON THE MARKET IN GERMANY AND UK PhotoCure ASA First Quarter Report 2003 Highlights: The launches of Metvix PDT by Galderma in February in Germany and in May in the UK, have triggered milestone
METVIX PDT ON THE MARKET IN GERMANY AND UK PhotoCure ASA First Quarter Report 2003 Highlights: The launches of Metvix PDT by Galderma in February in Germany and in May in the UK, have triggered milestone
SPHERIX ANNOUNCES FIRST QUARTER 2010 FINANCIAL RESULTS
 SPHERIX Investor Relations Phone: (301) 897-2564 Email: info@spherix.com SPHERIX ANNOUNCES FIRST QUARTER 2010 FINANCIAL RESULTS BETHESDA, MD, May 21, 2010 - Spherix Incorporated (NASDAQ CM: SPEX), an innovator
SPHERIX Investor Relations Phone: (301) 897-2564 Email: info@spherix.com SPHERIX ANNOUNCES FIRST QUARTER 2010 FINANCIAL RESULTS BETHESDA, MD, May 21, 2010 - Spherix Incorporated (NASDAQ CM: SPEX), an innovator
For personal use only
 Cochlear Limited Results for the half year ended 31 December 2011 (H1 F12) Chris Roberts, CEO Neville Mitchell, CFO Cochlear Overview Cochlear Limited (ASX:COH): global leader in implantable devices for
Cochlear Limited Results for the half year ended 31 December 2011 (H1 F12) Chris Roberts, CEO Neville Mitchell, CFO Cochlear Overview Cochlear Limited (ASX:COH): global leader in implantable devices for
FY2009 First Quarter Financial Results. SUZUKI MOTOR CORPORATION August 3, 2009
 FY2009 First Quarter Financial Results SUZUKI MOTOR CORPORATION August 3, 2009 Consolidated: Financial Summary Page 2 FY2009 1Q FY2008 4Q 09/4-6 09/1-3 Change FY2008 1Q 08/4-6 Change (Billion Yen) (A)
FY2009 First Quarter Financial Results SUZUKI MOTOR CORPORATION August 3, 2009 Consolidated: Financial Summary Page 2 FY2009 1Q FY2008 4Q 09/4-6 09/1-3 Change FY2008 1Q 08/4-6 Change (Billion Yen) (A)
Telekom Austria Group Results for the 1st Quarter May 27, 2003
 Telekom Austria Group Results for the 1st Quarter 2003 May 27, 2003 1 Cautionary Statement This presentation contains certain forward-looking statements. Actual results may differ materially from those
Telekom Austria Group Results for the 1st Quarter 2003 May 27, 2003 1 Cautionary Statement This presentation contains certain forward-looking statements. Actual results may differ materially from those
ESC. Enhanced Stability Liners. Design Rationale & Surgical Technique
 ESC Enhanced Stability Liners Design Rationale & Surgical Technique Choice Without Compromise DePuy Synthes PINNACLE Hip Solutions are designed with a wide range of acetabular cup options, biological and
ESC Enhanced Stability Liners Design Rationale & Surgical Technique Choice Without Compromise DePuy Synthes PINNACLE Hip Solutions are designed with a wide range of acetabular cup options, biological and
Summary of Results for the First Half of FY2015/3
 Summary of Results for the First Half of FY2015/3 November 10, 2014 Tokyu Corporation (9005) http://www.tokyu.co.jp/ Contents Ⅰ.Executive Summary 2 Ⅱ.Conditions in Each Business 5 Ⅲ.Details of Financial
Summary of Results for the First Half of FY2015/3 November 10, 2014 Tokyu Corporation (9005) http://www.tokyu.co.jp/ Contents Ⅰ.Executive Summary 2 Ⅱ.Conditions in Each Business 5 Ⅲ.Details of Financial
ObsEva Reports First Quarter 2018 Financial Results and Provides Business Update
 ObsEva Reports First Quarter 2018 Financial Results and Provides Business Update -Key First Quarter 2018 Clinical Milestones Achieved Patient randomization completed in Phase 2b EDELWEISS trial of OBE2109
ObsEva Reports First Quarter 2018 Financial Results and Provides Business Update -Key First Quarter 2018 Clinical Milestones Achieved Patient randomization completed in Phase 2b EDELWEISS trial of OBE2109
HOSPITAL "A" (2007) ORTHOPEDIC JOINT IMPLANT COMPONENT MATRIX
 HOSPITAL "A" (2007) ORTHOPEDIC JOINT IMPLANT COMPONENT MATRIX HIP IMPLANTS TOTAL HIPS Ultra High Demand (metal on metal) Femur, ingrowth Femoral head, metal Acetabular shell, ingrowth Acetabular liner,
HOSPITAL "A" (2007) ORTHOPEDIC JOINT IMPLANT COMPONENT MATRIX HIP IMPLANTS TOTAL HIPS Ultra High Demand (metal on metal) Femur, ingrowth Femoral head, metal Acetabular shell, ingrowth Acetabular liner,
Financial Presentation
 Financial Presentation Certain of the statements that will be made during this meeting, as well as statements contained in our 2006 Annual Report to Shareholders and Annual Report on Form 10-K filed with
Financial Presentation Certain of the statements that will be made during this meeting, as well as statements contained in our 2006 Annual Report to Shareholders and Annual Report on Form 10-K filed with
Regenerex Porous Titanium Construct. Knees Hips Extremities Cement and Accessories PMI Trauma Technology
 Regenerex Porous Titanium Construct Knees Hips Extremities Cement and Accessories PMI Trauma Technology One Surgeon. One Patient. Over 1 million times per year, Biomet helps one surgeon provide personalized
Regenerex Porous Titanium Construct Knees Hips Extremities Cement and Accessories PMI Trauma Technology One Surgeon. One Patient. Over 1 million times per year, Biomet helps one surgeon provide personalized
Endesa 1Q 2017 Results 09/05/2017
 Endesa 1Q 2017 Results 09/05/2017 1. Highlights and key financial figures 2. Endesa s performance in 1Q 2017 market context 3. Financial results 4. Final remarks 2 1. Highlights and key financial figures
Endesa 1Q 2017 Results 09/05/2017 1. Highlights and key financial figures 2. Endesa s performance in 1Q 2017 market context 3. Financial results 4. Final remarks 2 1. Highlights and key financial figures
ObsEva Reports Third Quarter 2018 Financial Results and Provides Business Update
 ObsEva Reports Third Quarter 2018 Financial Results and Provides Business Update IMPLANT 4 trial of nolasiban in IVF starting in Q4 2018, European MAA filing expected late 2019 24-week data from Phase
ObsEva Reports Third Quarter 2018 Financial Results and Provides Business Update IMPLANT 4 trial of nolasiban in IVF starting in Q4 2018, European MAA filing expected late 2019 24-week data from Phase
SAMPLE. South Korea Orthopedic Procedures Outlook to Reference Code: GDMEMC0008PDB. Publication Date: March 2014
 South Korea Orthopedic Procedures Outlook to 2020 Reference Code: GDMEMC0008PDB Publication Date: March 2014 Page 1 1 Table of Contents 1 Table of Contents... 2 1.1 List of Tables... 4 1.2 List of Figures...
South Korea Orthopedic Procedures Outlook to 2020 Reference Code: GDMEMC0008PDB Publication Date: March 2014 Page 1 1 Table of Contents 1 Table of Contents... 2 1.1 List of Tables... 4 1.2 List of Figures...
Slide 1. Investor presentation. London 5 February 2019
 Slide Investor presentation London 5 February 209 Slide 2 Forward-looking statements Novo Nordisk s reports filed with or furnished to the US Securities and Exchange Commission (SEC), including this presentation
Slide Investor presentation London 5 February 209 Slide 2 Forward-looking statements Novo Nordisk s reports filed with or furnished to the US Securities and Exchange Commission (SEC), including this presentation
Uroplasty, Inc. Investor Update Canaccord Genuity Conference December 6, 2011
 Uroplasty, Inc. Investor Update Canaccord Genuity Conference December 6, 2011 Forward Looking Statement This presentation includes forward-looking statements, including financial projections, relating
Uroplasty, Inc. Investor Update Canaccord Genuity Conference December 6, 2011 Forward Looking Statement This presentation includes forward-looking statements, including financial projections, relating
Consolidated: Financial Summary
 FY2010 First Quarter Financial Results SUZUKI MOTOR CORPORATION 3 August 2010 Consolidated: Financial Summary Page2 FY2010 1Q FY2009 1Q (2010/4-6) (2009/4-6) Change Net sales 656.3 577.1 +79.2 +13.7% Operating
FY2010 First Quarter Financial Results SUZUKI MOTOR CORPORATION 3 August 2010 Consolidated: Financial Summary Page2 FY2010 1Q FY2009 1Q (2010/4-6) (2009/4-6) Change Net sales 656.3 577.1 +79.2 +13.7% Operating
Solutions For The Aging Spine
 Solutions For The Aging Spine NASDAQ: ATEC February 2009 www.alphatecspine.com Slide 0 5818 El Camino Real, Carlsbad, CA 92008 www.alphatecspine.com Forward Looking Statements This presentation and our
Solutions For The Aging Spine NASDAQ: ATEC February 2009 www.alphatecspine.com Slide 0 5818 El Camino Real, Carlsbad, CA 92008 www.alphatecspine.com Forward Looking Statements This presentation and our
Walgreen Co. Reports Second Quarter 2010 Earnings Per Diluted Share of 68 Cents; Results Include 2 Cents Per Diluted Share of Restructuring Costs
 Walgreen Co. Reports Second Quarter 2010 Earnings Per Diluted Share of 68 Cents; Results Include 2 Cents Per Diluted Share of Restructuring Costs Second quarter sales up 3.1 percent to record $17.0 billion
Walgreen Co. Reports Second Quarter 2010 Earnings Per Diluted Share of 68 Cents; Results Include 2 Cents Per Diluted Share of Restructuring Costs Second quarter sales up 3.1 percent to record $17.0 billion
Sunesis Pharmaceuticals Reports Second Quarter 2011 Financial Results
 Investor and Media Inquiries: David Pitts Argot Partners 212-600-1902 Eric Bjerkholt Sunesis Pharmaceuticals Inc. 650-266-3717 Sunesis Pharmaceuticals Reports Second Quarter 2011 Financial Results SOUTH
Investor and Media Inquiries: David Pitts Argot Partners 212-600-1902 Eric Bjerkholt Sunesis Pharmaceuticals Inc. 650-266-3717 Sunesis Pharmaceuticals Reports Second Quarter 2011 Financial Results SOUTH
Operations included in the National Joint Registry (NJR) Quick links, go to: Hips > Knees > Ankles > Elbows > Shoulders > Trauma >
 Operations included in the National Joint Registry (NJR) Quick links, go to: Hips > Knees > Ankles > Elbows > Shoulders > Trauma > Version 5 December 2013 1 Version control Version number Date Amendments
Operations included in the National Joint Registry (NJR) Quick links, go to: Hips > Knees > Ankles > Elbows > Shoulders > Trauma > Version 5 December 2013 1 Version control Version number Date Amendments
Aerpio Reports Fourth Quarter and Full Year 2017 Financial Results and Provides Business Update
 Aerpio Reports Fourth Quarter and Full Year 2017 Financial Results and Provides Business Update TIME-2b Clinical Trial of AKB-9778 in Patients with Diabetic Retinopathy Remains on Track Conference Call
Aerpio Reports Fourth Quarter and Full Year 2017 Financial Results and Provides Business Update TIME-2b Clinical Trial of AKB-9778 in Patients with Diabetic Retinopathy Remains on Track Conference Call
Spinal Surgical Robots: Market Shares, Strategies, and Forecasts, Worldwide,
 Spinal Surgical Robots: Market Shares, Strategies, and Forecasts, Worldwide, 2016-2022 Table of Contents Spinal Surgical Robot: Executive Summary The study is designed to give a comprehensive overview
Spinal Surgical Robots: Market Shares, Strategies, and Forecasts, Worldwide, 2016-2022 Table of Contents Spinal Surgical Robot: Executive Summary The study is designed to give a comprehensive overview
CENTRALE. Monetary. compared. domestic. million.
 CENTRALE BANK VAN ARUBAA Statistical News Release Second Quarter 2012 Date: November 7, 2012 Monetary and financial developments Money and credit In the second quarterr of 2012, the money supply decreased
CENTRALE BANK VAN ARUBAA Statistical News Release Second Quarter 2012 Date: November 7, 2012 Monetary and financial developments Money and credit In the second quarterr of 2012, the money supply decreased
The Leader in Orthopaedic Innovation
 The Leader in Orthopaedic Innovation Wright is a leading international manufacturer and distributor of superior, easy to use, and innovative orthopaedic implants and instrumentation. For over 50 years,
The Leader in Orthopaedic Innovation Wright is a leading international manufacturer and distributor of superior, easy to use, and innovative orthopaedic implants and instrumentation. For over 50 years,
Second Quarter and First Half 2011 Conference Call. 29 July, SORIN GROUP Presentation 1
 Second Quarter and First Half 2011 Conference Call 29 July, 2011 SORIN GROUP Presentation 1 DISCLAIMER This presentation contains management preliminary estimates and forward-looking statements, including
Second Quarter and First Half 2011 Conference Call 29 July, 2011 SORIN GROUP Presentation 1 DISCLAIMER This presentation contains management preliminary estimates and forward-looking statements, including
Presentation of. Burgdorf, 1 st November 2012
 Presentation of Semi-Annual Results 2012/13 Burgdorf, 1 st November 2012 Important Notice The information contained in this document has not been independently verified and no representation or warranty,
Presentation of Semi-Annual Results 2012/13 Burgdorf, 1 st November 2012 Important Notice The information contained in this document has not been independently verified and no representation or warranty,
ThromboGenics Business Update H1 2018
 Press release 6 September 2018 Regulated Information ThromboGenics Business Update H1 2018 Positive Topline Results for Diabetic Eye Disease Portfolio Shareholder approval for company name change to Oxurion
Press release 6 September 2018 Regulated Information ThromboGenics Business Update H1 2018 Positive Topline Results for Diabetic Eye Disease Portfolio Shareholder approval for company name change to Oxurion
Q Investor Kit JANUARY-MARCH 2013
 Q1 2013 Investor Kit JANUARY-MARCH 2013 1 Swedish Match reporting segments Snus and snuff - Swedish snus (Scandinavia and US) - Moist snuff (US) - SMPM International Other tobacco products - Cigars (US)
Q1 2013 Investor Kit JANUARY-MARCH 2013 1 Swedish Match reporting segments Snus and snuff - Swedish snus (Scandinavia and US) - Moist snuff (US) - SMPM International Other tobacco products - Cigars (US)
Operations included in the National Joint Registry (NJR)
 Operations included in the National Joint Registry (NJR) Quick links, go to: Hips > Knees > Ankles > Elbows > Shoulders > Trauma > Version 8 June 2018 1 Version control Version number Date Amendments 1
Operations included in the National Joint Registry (NJR) Quick links, go to: Hips > Knees > Ankles > Elbows > Shoulders > Trauma > Version 8 June 2018 1 Version control Version number Date Amendments 1
HILLENBRAND INDUSTRIES INC
 HILLENBRAND INDUSTRIES INC FORM 8-K (Unscheduled Material Events) Filed 2/17/2004 For Period Ending 2/13/2004 Address 700 STATE ROUTE 46 E BATESVILLE, Indiana 47006-8835 Telephone 812-934-7000 CIK 0000047518
HILLENBRAND INDUSTRIES INC FORM 8-K (Unscheduled Material Events) Filed 2/17/2004 For Period Ending 2/13/2004 Address 700 STATE ROUTE 46 E BATESVILLE, Indiana 47006-8835 Telephone 812-934-7000 CIK 0000047518
China Orthopedics Implants Medical Devices Market Brief
 China Orthopedics Implants Medical Devices Market Brief By: PIM LTD. August 2017 reproduced for distribution outside the client organization without prior written approval from PIM Ltd. 1 Definition and
China Orthopedics Implants Medical Devices Market Brief By: PIM LTD. August 2017 reproduced for distribution outside the client organization without prior written approval from PIM Ltd. 1 Definition and
Transparency Market Research
 Transparency Market Research IGBT and Super Junction MOSFET Market - Global Industry Analysis, Size, Share, Growth, Trends and Forecast, 2013-2019 Single User License: USD 4595 Multi User License: USD
Transparency Market Research IGBT and Super Junction MOSFET Market - Global Industry Analysis, Size, Share, Growth, Trends and Forecast, 2013-2019 Single User License: USD 4595 Multi User License: USD
Slide 1. Financial update and closing remarks. Jesper Brandgaard EVP and CFO. RAFAEL DE JESÚS FLORES, Mexico Rafael has haemophilia A
 Slide 1 Financial update and closing remarks Jesper Brandgaard EVP and CFO RAFAEL DE JESÚS FLORES, Mexico Rafael has haemophilia A Slide 2 Forward-looking statements Novo Nordisk s reports filed with or
Slide 1 Financial update and closing remarks Jesper Brandgaard EVP and CFO RAFAEL DE JESÚS FLORES, Mexico Rafael has haemophilia A Slide 2 Forward-looking statements Novo Nordisk s reports filed with or
AstraZeneca to acquire MedImmune for $58 per share in a fully recommended, all-cash transaction with a total enterprise value of $15.
 [NOT FOR RELEASE, PUBLICATION OR DISTRIBUTION IN WHOLE OR IN PART, IN, INTO OR FROM AUSTRALIA, CANADA OR JAPAN OR ANY OTHER JURISDICTION WHERE TO DO SO WOULD CONSTITUTE A VIOLATION OF THE RELEVANT LAWS
[NOT FOR RELEASE, PUBLICATION OR DISTRIBUTION IN WHOLE OR IN PART, IN, INTO OR FROM AUSTRALIA, CANADA OR JAPAN OR ANY OTHER JURISDICTION WHERE TO DO SO WOULD CONSTITUTE A VIOLATION OF THE RELEVANT LAWS
Evofem Biosciences Reports Second Quarter 2018 Financial Results and Provides Corporate Update
 Evofem Biosciences Reports Second Quarter 2018 Financial Results and Provides Corporate Update Phase 3 Trial of Amphora for Hormone-free Contraception Continues to Move Ahead of Schedule; Top Line Data
Evofem Biosciences Reports Second Quarter 2018 Financial Results and Provides Corporate Update Phase 3 Trial of Amphora for Hormone-free Contraception Continues to Move Ahead of Schedule; Top Line Data
ALLERGAN TO ACQUIRE ZELTIQ FOR $2.47B BEST-IN-CLASS COMPANY IN FAST GROWING BODY CONTOURING SEGMENT
 ALLERGAN TO ACQUIRE ZELTIQ FOR $2.47B BEST-IN-CLASS COMPANY IN FAST GROWING BODY CONTOURING SEGMENT ALLERGAN CAUTIONARY STATEMENT REGARDING FORWARD-LOOKING STATEMENTS Statements contained in this presentation
ALLERGAN TO ACQUIRE ZELTIQ FOR $2.47B BEST-IN-CLASS COMPANY IN FAST GROWING BODY CONTOURING SEGMENT ALLERGAN CAUTIONARY STATEMENT REGARDING FORWARD-LOOKING STATEMENTS Statements contained in this presentation
PhotoCure ASA. Presentation. Results 1 Quarter 2004
 PhotoCure ASA Presentation Results 1 Quarter 2004 7 th of May 2004 Progress towards sustained profitability Continued product roll out Metvix European sales continue to grow Final US approval for Actinic
PhotoCure ASA Presentation Results 1 Quarter 2004 7 th of May 2004 Progress towards sustained profitability Continued product roll out Metvix European sales continue to grow Final US approval for Actinic
Q2 INVESTOR KIT JANUARY-JUNE 2011
 Q2 INVESTOR KIT JANUARY-JUNE 2011 Swedish Match reporting segments Reporting segments following the transaction with STG on October 1, 2010 Snus and snuff - Swedish snus in Scandinavia and US - US moist
Q2 INVESTOR KIT JANUARY-JUNE 2011 Swedish Match reporting segments Reporting segments following the transaction with STG on October 1, 2010 Snus and snuff - Swedish snus in Scandinavia and US - US moist
DIEBOLD NIXDORF AG AT GERMANY EQUITY FORUM
 November 28, 2017 DIEBOLD NIXDORF AG AT GERMANY EQUITY FORUM Dr. Jürgen Wunram Agenda Diebold Nixdorf AG History and shareholder structure Market trends, strategy and progress of integration Diebold Nixdorf
November 28, 2017 DIEBOLD NIXDORF AG AT GERMANY EQUITY FORUM Dr. Jürgen Wunram Agenda Diebold Nixdorf AG History and shareholder structure Market trends, strategy and progress of integration Diebold Nixdorf
National Joint Replacement Registry. Lay Summary 2015 Annual Report Hip and Knee Replacement
 National Joint Replacement Registry Lay Summary 2015 Annual Report Hip and Knee Replacement SUPPLEMENTARY REPORT 2015 TABLE OF CONTENTS Introduction... 1 A brief history of the Registry origins... 1 The
National Joint Replacement Registry Lay Summary 2015 Annual Report Hip and Knee Replacement SUPPLEMENTARY REPORT 2015 TABLE OF CONTENTS Introduction... 1 A brief history of the Registry origins... 1 The
Acasti Pharma Provides Business Update for the Third Quarter of Fiscal 2019
 Acasti Pharma Provides Business Update for the Third Quarter of Fiscal 2019 Planned Enrollment targets achieved in both TRILOGY studies with over 74% of patients randomized at more than 150 clinical sites
Acasti Pharma Provides Business Update for the Third Quarter of Fiscal 2019 Planned Enrollment targets achieved in both TRILOGY studies with over 74% of patients randomized at more than 150 clinical sites
BALCHEM CORPORATION. Q Investor Relations Presentation
 BALCHEM CORPORATION Q4 2017 Investor Relations Presentation Balchem Corporation Balchem develops, manufactures, and markets specialty ingredients that improve and enhance the health and well being of life
BALCHEM CORPORATION Q4 2017 Investor Relations Presentation Balchem Corporation Balchem develops, manufactures, and markets specialty ingredients that improve and enhance the health and well being of life
Visionary Private Equity Group is Pleased to Announce its Investment in MEDITE Cancer Diagnostics
 Dear VPEG Limited Partner, We hope this investor update finds you well. We're pleased to share with you an important update below on the following: Visionary Private Equity Group is Pleased to Announce
Dear VPEG Limited Partner, We hope this investor update finds you well. We're pleased to share with you an important update below on the following: Visionary Private Equity Group is Pleased to Announce
Flexion Therapeutics Reports First-Quarter 2018 Financial Results and Recent Business Highlights
 May 8, 2018 Flexion Therapeutics Reports First-Quarter 2018 Financial Results and Recent Business Highlights Company booked net ZILRETTA sales of $2.2 million in Q1 Positive developments in Medicare reimbursement
May 8, 2018 Flexion Therapeutics Reports First-Quarter 2018 Financial Results and Recent Business Highlights Company booked net ZILRETTA sales of $2.2 million in Q1 Positive developments in Medicare reimbursement
ONO PHARMACEUTICAL CO., LTD.
 ONO PHARMACEUTICAL CO., LTD. February 4, 2005 Ono Pharmaceutical Co., Ltd. has announced its consolidated financial results for nine months ended December 31,. This First-Third Quarter Flash Report 2005
ONO PHARMACEUTICAL CO., LTD. February 4, 2005 Ono Pharmaceutical Co., Ltd. has announced its consolidated financial results for nine months ended December 31,. This First-Third Quarter Flash Report 2005
ONO PHARMACEUTICAL CO., LTD.
 ONO PHARMACEUTICAL CO., LTD. February 6, 2006 Ono Pharmaceutical Co., Ltd. has announced its consolidated financial results for nine months ended December 31,. This First-Third Quarter Flash Report 2006
ONO PHARMACEUTICAL CO., LTD. February 6, 2006 Ono Pharmaceutical Co., Ltd. has announced its consolidated financial results for nine months ended December 31,. This First-Third Quarter Flash Report 2006
Forward Looking Information
 Vital Signs, Inc. Forward Looking Information Except for historical information discussed, the statements made today are forward-looking statements that involve risks and uncertainties. Investors are cautioned
Vital Signs, Inc. Forward Looking Information Except for historical information discussed, the statements made today are forward-looking statements that involve risks and uncertainties. Investors are cautioned
Value creation through profitable growth Handelsbanken Nordic Large Cap Seminar, Stockholm 2016 Anders Lonning-Skovgaard, CFO
 Value creation through profitable growth Handelsbanken Nordic Large Cap Seminar, Stockholm 2016 Anders Lonning-Skovgaard, CFO Coloplast has four business areas all with global sales presence Group revenue
Value creation through profitable growth Handelsbanken Nordic Large Cap Seminar, Stockholm 2016 Anders Lonning-Skovgaard, CFO Coloplast has four business areas all with global sales presence Group revenue
Infinity Highlights Promising Preclinical Hedgehog Data and Announces First Quarter Financial Results
 Infinity Highlights Promising Preclinical Hedgehog Data and Announces First Quarter Financial Results Heat Shock Protein 90 Program Continues to Advance Toward Potential Registration Trial CAMBRIDGE, Mass.,
Infinity Highlights Promising Preclinical Hedgehog Data and Announces First Quarter Financial Results Heat Shock Protein 90 Program Continues to Advance Toward Potential Registration Trial CAMBRIDGE, Mass.,
CPH Chemie + Papier Holding AG. Investor Day. Perlen, 13 th September 2018
 CPH Chemie + Papier Holding AG Investor Day Perlen, 13 th September 2018 Agenda 2.00 pm Welcome 2.05 pm Presentation Course of Business 2018 2.50 pm Break 3.00 pm Presentation Perlen Packaging 3.30 pm
CPH Chemie + Papier Holding AG Investor Day Perlen, 13 th September 2018 Agenda 2.00 pm Welcome 2.05 pm Presentation Course of Business 2018 2.50 pm Break 3.00 pm Presentation Perlen Packaging 3.30 pm
Coloplast A/S. Investor presentation 1H 2005/06
 Coloplast A/S Investor presentation 1H 2005/06 2 Coloplast Coloplast products and services help patients achieve greater independence from medical challenges in 4 areas: Ostomy care, continence care, wound
Coloplast A/S Investor presentation 1H 2005/06 2 Coloplast Coloplast products and services help patients achieve greater independence from medical challenges in 4 areas: Ostomy care, continence care, wound
Over 20 Years of Proven Clinical Success. Zimmer Natural-Knee II System
 Over 20 Years of Proven Clinical Success Zimmer Natural-Knee II System CSTi Porous Coating Structurally similar to human bone CSTi porous coating combines the excellent biocompatibility of titanium with
Over 20 Years of Proven Clinical Success Zimmer Natural-Knee II System CSTi Porous Coating Structurally similar to human bone CSTi porous coating combines the excellent biocompatibility of titanium with
LifeHealthcare Investor Day 5 October 2016
 LifeHealthcare Investor Day 5 October 2016 Agenda 1. Lunch and Welcome 2. LifeHealthcare Overview and Update on Strategic Priorities 3. Spine Overview 4. Dr Hsu Presentation 5. Orthopaedics Overview 6.
LifeHealthcare Investor Day 5 October 2016 Agenda 1. Lunch and Welcome 2. LifeHealthcare Overview and Update on Strategic Priorities 3. Spine Overview 4. Dr Hsu Presentation 5. Orthopaedics Overview 6.
DARA Reports Year-End 2012 Financial Results
 April 1, 2013 DARA Reports Year-End 2012 Financial Results Company Provides a Commercial Portfolio and Development Pipeline Business Update RALEIGH, NC -- (MARKETWIRE) -- 04/01/13 -- DARA BioSciences,
April 1, 2013 DARA Reports Year-End 2012 Financial Results Company Provides a Commercial Portfolio and Development Pipeline Business Update RALEIGH, NC -- (MARKETWIRE) -- 04/01/13 -- DARA BioSciences,
Sumitomo Dainippon Pharma to Acquire Tolero Pharmaceuticals, Inc. (US Biotechnology Company)
 December 21, 2016 Sumitomo Dainippon Pharma Co., Ltd. Sumitomo Dainippon Pharma to Acquire Tolero Pharmaceuticals, Inc. (US Biotechnology Company) Sumitomo Dainippon Pharma Co., Ltd. (Head office: Osaka,
December 21, 2016 Sumitomo Dainippon Pharma Co., Ltd. Sumitomo Dainippon Pharma to Acquire Tolero Pharmaceuticals, Inc. (US Biotechnology Company) Sumitomo Dainippon Pharma Co., Ltd. (Head office: Osaka,
Q Investor Kit January December 2015
 Q4 2015 Investor Kit January December 2015 Summary and Overview 2 Q4 and full year 2015: Headline numbers Sales for the full year increased by 9 percent and for the fourth quarter by 5 percent. In local
Q4 2015 Investor Kit January December 2015 Summary and Overview 2 Q4 and full year 2015: Headline numbers Sales for the full year increased by 9 percent and for the fourth quarter by 5 percent. In local
Sumitomo Pharmaceuticals reaches a basic agreement to merge with Dainippon Pharmaceutical
 For immediate release November 25, 2004 Sumitomo Pharmaceuticals reaches a basic agreement to merge with Dainippon Pharmaceutical Sumitomo Chemical Co., Ltd. announced today that its subsidiary, Sumitomo
For immediate release November 25, 2004 Sumitomo Pharmaceuticals reaches a basic agreement to merge with Dainippon Pharmaceutical Sumitomo Chemical Co., Ltd. announced today that its subsidiary, Sumitomo
REATA PHARMACEUTICALS, INC. ANNOUNCES SECOND QUARTER 2018 FINANCIAL RESULTS AND AN UPDATE ON DEVELOPMENT PROGRAMS
 REATA PHARMACEUTICALS, INC. ANNOUNCES SECOND QUARTER 2018 FINANCIAL RESULTS AND AN UPDATE ON DEVELOPMENT PROGRAMS IRVING, Texas August 8, 2018 Reata Pharmaceuticals, Inc. (Nasdaq: RETA), a clinical-stage
REATA PHARMACEUTICALS, INC. ANNOUNCES SECOND QUARTER 2018 FINANCIAL RESULTS AND AN UPDATE ON DEVELOPMENT PROGRAMS IRVING, Texas August 8, 2018 Reata Pharmaceuticals, Inc. (Nasdaq: RETA), a clinical-stage
Hip and Knee Orthopedic Surgical Implants Market Growth, Research & Trend to Radiant Insights, Inc
 Hip and Knee Orthopedic Surgical Implants Market Growth, Research & Trend to 2016-2022 Radiant Insights, Inc Orthopedic surgical implants help replace deteriorated joints. This deterioration could be due
Hip and Knee Orthopedic Surgical Implants Market Growth, Research & Trend to 2016-2022 Radiant Insights, Inc Orthopedic surgical implants help replace deteriorated joints. This deterioration could be due
Interim report Second quarter 2017 and subsequent events (Unaudited)
 SERODUS ASA Interim report Second quarter 2017 and subsequent events (Unaudited) August 2017 2017-08-15 Q2-2017 report ABOUT SERODUS Serodus is a private biotech company with a focus on diabetic comorbidities
SERODUS ASA Interim report Second quarter 2017 and subsequent events (Unaudited) August 2017 2017-08-15 Q2-2017 report ABOUT SERODUS Serodus is a private biotech company with a focus on diabetic comorbidities
SWEDISH MATCH INVESTOR KIT Q4 2016
 SWEDISH MATCH INVESTOR KIT Q4 2016 A look at the quarter 2 Q4 HEADLINE BULLETS Sales increased by 6 percent to 3,957 MSEK (3,719) for the fourth quarter and by 7 percent to 15,551 MSEK (14,486) for the
SWEDISH MATCH INVESTOR KIT Q4 2016 A look at the quarter 2 Q4 HEADLINE BULLETS Sales increased by 6 percent to 3,957 MSEK (3,719) for the fourth quarter and by 7 percent to 15,551 MSEK (14,486) for the
RAI reports strong 2Q16 performance and positive outlook; Accelerates returns to shareholders
 Contact: Investor Relations: Bob Bannon (336) 741-3359 Media: Jane Seccombe (336) 741-5068 Reynolds American Inc. P.O. Box 2990 Winston-Salem, NC 27102-2990 RAI 2016-22 RAI reports strong 2Q16 performance
Contact: Investor Relations: Bob Bannon (336) 741-3359 Media: Jane Seccombe (336) 741-5068 Reynolds American Inc. P.O. Box 2990 Winston-Salem, NC 27102-2990 RAI 2016-22 RAI reports strong 2Q16 performance
Acquisition of Novartis Influenza Vaccines Business. 27 th October 2014
 1 Acquisition of Novartis Influenza Vaccines Business 27 th October 2014 2 Legal Notice Forward looking statements The materials in this presentation speak only as of the date of these materials, and include
1 Acquisition of Novartis Influenza Vaccines Business 27 th October 2014 2 Legal Notice Forward looking statements The materials in this presentation speak only as of the date of these materials, and include
ASL MARINE. DBS Vickers Pulse of Asia Roadshow 10 January 2012
 ASL MARINE DBS Vickers Pulse of Asia Roadshow 10 January 2012 Presentation Outline Group Overview 1Q FY2012 Financial Review (3 months ended 30 September 2011) Operations Review - Shipbuilding - Shiprepair
ASL MARINE DBS Vickers Pulse of Asia Roadshow 10 January 2012 Presentation Outline Group Overview 1Q FY2012 Financial Review (3 months ended 30 September 2011) Operations Review - Shipbuilding - Shiprepair
Global Spine Market with Focus on Spinal Fusion: Industry Analysis & Outlook ( )
 Industry Research by Koncept Analytics Global Spine Market with Focus on Spinal Fusion: Industry Analysis & Outlook ----------------------------------------- (2016-2020) December 2016 1 Executive Summary
Industry Research by Koncept Analytics Global Spine Market with Focus on Spinal Fusion: Industry Analysis & Outlook ----------------------------------------- (2016-2020) December 2016 1 Executive Summary
RULES OF CONDUCT OF INSIDERS RESPECTING
 T RULES OF CONDUCT OF INSIDERS RESPECTING RADING O F SECURITIES OF TFI International lnc. Amended and restated July 2015 Executive Summary As an insider of TFI International Inc. ( TFI International )
T RULES OF CONDUCT OF INSIDERS RESPECTING RADING O F SECURITIES OF TFI International lnc. Amended and restated July 2015 Executive Summary As an insider of TFI International Inc. ( TFI International )
Patrick J. Sullivan Chief Executive Officer. January 9, 2018 Investor Presentation
 Patrick J. Sullivan Chief Executive Officer January 9, 2018 Investor Presentation Forward Looking Statement This presentation may contain forward-looking statements concerning Insulet's expectations, anticipations,
Patrick J. Sullivan Chief Executive Officer January 9, 2018 Investor Presentation Forward Looking Statement This presentation may contain forward-looking statements concerning Insulet's expectations, anticipations,
Genomic Health. Kim Popovits, Chairman, CEO and President
 Genomic Health Kim Popovits, Chairman, CEO and President Safe Harbor Statement Various remarks that we make in this presentation that are not historical, including those about our future financial and
Genomic Health Kim Popovits, Chairman, CEO and President Safe Harbor Statement Various remarks that we make in this presentation that are not historical, including those about our future financial and
Sunesis Pharmaceuticals Reports Third Quarter 2014 Financial Results and Recent Highlights
 Sunesis Pharmaceuticals Reports Third Quarter 2014 Financial Results and Recent Highlights November 10, 2014 7:00 AM ET Announces Submission of Letter of Intent to File MAA for Vosaroxin in Relapsed or
Sunesis Pharmaceuticals Reports Third Quarter 2014 Financial Results and Recent Highlights November 10, 2014 7:00 AM ET Announces Submission of Letter of Intent to File MAA for Vosaroxin in Relapsed or
A Patient s Guide to Adult Forearm Fractures
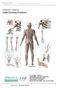 A Patient s Guide to Adult Forearm Fractures Orthopedic and Sports Medicine 825 South 8th Street, #550 Minneapolis, MN 55404 Phone: 612-333-5000 Fax: 612-333-6922 1 DISCLAIMER: The information in this
A Patient s Guide to Adult Forearm Fractures Orthopedic and Sports Medicine 825 South 8th Street, #550 Minneapolis, MN 55404 Phone: 612-333-5000 Fax: 612-333-6922 1 DISCLAIMER: The information in this
Presentation First quarter 2006
 Immunity for Life TM Presentation First quarter 2006 CEO Gunnar Rørstad CFO Finn Samuelsen 10 May 2006 Highlights - operations Inclusion of patients almost completed in the the phase II clinical trial
Immunity for Life TM Presentation First quarter 2006 CEO Gunnar Rørstad CFO Finn Samuelsen 10 May 2006 Highlights - operations Inclusion of patients almost completed in the the phase II clinical trial
Arcos Modular Femoral Revision System
 Arcos Modular Femoral Revision System Arcos System Simplify the Complex The Arcos Modular Femoral Revision System meets the demands of complex hip revision surgery by offering surgeons and OR staff the
Arcos Modular Femoral Revision System Arcos System Simplify the Complex The Arcos Modular Femoral Revision System meets the demands of complex hip revision surgery by offering surgeons and OR staff the
Oral semaglutide and production expansion. Henrik Wulff EVP Product Supply. Peter Kristensen SVP Global Development
 YASMIN FIEDLER Germany Yasmin has type 1 diabetes Slide 1 Oral semaglutide and production expansion Henrik Wulff EVP Product Supply Peter Kristensen SVP Global Development Slide 2 Forward-looking statements
YASMIN FIEDLER Germany Yasmin has type 1 diabetes Slide 1 Oral semaglutide and production expansion Henrik Wulff EVP Product Supply Peter Kristensen SVP Global Development Slide 2 Forward-looking statements
Knee Replacement Implants
 Knee Replacement Implants During knee replacement surgery, an orthopaedic surgeon will resurface your damaged knee with artificial components, called implants. There are many different types of implants.
Knee Replacement Implants During knee replacement surgery, an orthopaedic surgeon will resurface your damaged knee with artificial components, called implants. There are many different types of implants.
DELICA D:5 ACTIVE GEAR 0
 DELICA D:5 ACTIVE GEAR 0 1 1. FY2017 1Q Results 2. FY2017 1Q Results by Region 3. FY2017 Forecast 4. Initiatives for the Future FY2017 1Q Results Summary (vs. FY2016 1Q) (Apr-Jun 2016) (Apr-Jun 2017) (billion
DELICA D:5 ACTIVE GEAR 0 1 1. FY2017 1Q Results 2. FY2017 1Q Results by Region 3. FY2017 Forecast 4. Initiatives for the Future FY2017 1Q Results Summary (vs. FY2016 1Q) (Apr-Jun 2016) (Apr-Jun 2017) (billion
