Clinical Study Pulmonary Effects of Neonatal Hydrocortisone Treatment in Ventilator-Dependent Preterm Infants
|
|
|
- Emery Nicholson
- 5 years ago
- Views:
Transcription
1 International Pediatrics Volume 2011, Article ID , 7 pages doi: /2011/ Clinical Study Pulmonary Effects of Neonatal Hydrocortisone Treatment in Ventilator-Dependent Preterm Infants Sandra E. A. de Jong, Floris Groenendaal, Frank van Bel, and Karin J. Rademaker Department of Neonatology, University Medical Center Utrecht/Wilhelmina Children s Hospital, P.O. Box 85090, 3508 AB Utrecht, The Netherlands Correspondence should be addressed to Karin J. Rademaker, k.rademaker@umcutrecht.nl Received 24 June 2011; Accepted 21 September 2011 Academic Editor: Praveen Kumar Copyright 2011 Sandra E. A. de Jong et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. Background/Objective. Hydrocortisone, administered to ventilated preterm neonates to facilitate extubation, has no adverse longterm effects, but short-term pulmonary effects have not been described previously. In the present study, we analyzed effects of hydrocortisone on ventilator settings and FiO 2 in ventilator-dependent preterm infants. Patients and Methods. Fifty-five preterm children were included in this retrospective cohort study. Hydrocortisone was administered at a postnatal age of > 7 days to treat chronic lung disease (CLD). Ventilator settings before and after hydrocortisone administration were recorded as well as FiO 2 at 36 weeks gestational age. Presence of cerebral palsy was assessed at a mean corrected age of 24.1 months. Results. Hydrocortisone administered at a median postnatal age of 14 days significantly reduced FiO 2 from a median of 0.39 to 0.30, mean airway pressure (MAP) from a median of 10.0 cm H 2 Oto7.6cmH 2 O, and PaCO 2 from a median of 53.5 mmhg to 47 mmhg. Extubation was achieved in all patients. CLD at 36 weeks was present in 11 of the 52 patients (21.1%). None developed cerebral palsy. Conclusions. Hydrocortisone was effective in reducing the FiO 2,MAP,andPaCO 2 and facilitated extubation. Hydrocortisone was not associated with cerebral palsy. 1. Introduction Bronchopulmonary dysplasia (BPD) or chronic lung disease (CLD) was first described in 1967 as a severe form of lung disease in premature infants with severe respiratory distress syndrome, who received prolonged mechanical ventilation and high concentrations of oxygen [1]. Improved mechanically ventilatory strategies, and the introduction of antenatal glucocorticosteroids and postnatal surfactant treatment, have greatly reduced postnatal lung injury and increased survival of premature infants, resulting in a reported incidence of CLD of 8% 35% [2, 3]. Still, CLD remains a significant problem in neonatal intensive care because of the significant cause of mortality, morbidity, and prolonged hospitalization [3]. Since inflammation plays an important role in the pathogenesis of CLD [3, 4], glucocorticosteroids, mainly dexamethasone, have been widely used to reduce CLD. Numerous studies show that dexamethasone improves short-term respiratory function, leading to a reduction in supplementary oxygen requirements and earlier extubation [5 7]. Despite its positive effect on neonatal lung function, dexamethasone therapy exerts several acute adverse effects, such as hyperglycemia, hypertension, and left ventricular hypertrophy. Concerns about the long-term benefits of this steroid in ventilator-dependent infants arose in the 1970s when it became clear that there was an increased risk of cerebral palsy after postnatal dexamethasone exposure [8]. In recent years, scientific evidence has accumulated on the longterm negative effects of neonatal dexamethasone treatment [9 11]. In contrast to other neonatal intensive care units (NICUs) in the Netherlands, the Wilhelmina Children s Hospital has historically always used the corticosteroid hydrocortisone instead of the more potent dexamethasone to treat ventilator-dependent preterm infants at risk for CLD
2 2 International Pediatrics because of fewer adverse effects and the apparently similar clinical benefits [12, 13].However, until now no studies have described the short-term effects of hydrocortisone treatment on respiratory function. The purpose of this retrospective study is to analyze the immediate effect of hydrocortisone therapy on mechanical ventilation settings in ventilator-dependent preterm infants. 2. Patients and Methods The study population presented in this retrospective cohort study consists of patients who were born after a gestational age of 32 weeks and/or a birth weight of 1500 grams between January 1, 2003 and December 31, These patients were consecutively admitted soon after birth over a period of five years to the Neonatal Intensive Care Unit of the Wilhelmina Children s Hospital, a tertiary referral centre. One hundred and twenty-seven children born after a pregnancy of 32 weeks and/or with a birth weight of 1500 g had been treated postnatally with corticosteroids for several reasons. Nine patients received besides hydrocortisone also dexamethasone (two during part of their treatment in another hospital and seven prior to extubation) and were excluded to avoid contamination of hydrocortisone treatment effects. One hundred and eighteen infants born after a pregnancy of 32 weeks and/or with a birth weight of 1500 g received hydrocortisone treatment. Two patients received hydrocortisone as a replacement therapy for their congenital adrenal hyperplasia. Fifty-three patients received hydrocortisone to treat hypotension. Sixty-eight patients were treated with hydrocortisone to facilitate extubation. In one case, the attending neonatological staff decided to stop treatment after one day of hydrocortisone treatment because of severe respiratory insufficiency and the poor prognosis based on pulmonary hypoplasia as a result of PPROM. In twelve children treated with nasal continuous positive airway pressure (ncpap) after having been ventilated, hydrocortisone was administered in order to avoid re-intubation. These thirteen children were excluded. Four infants in whom hydrocortisone treatment was started while being ventilated died in neonatal period on day 13, 18, 22, and 63; one of bullous emphysema and three of severe chronic lung injury with in one case in combination with a metabolic disorder of unspecified origin. These four children were not excluded. Fifty-five out of the 118 children (46.6%) received hydrocortisone treatment for prolonged ventilator dependency Eligibility Criteria. Infants were eligible for the study if they met the following criteria: (1) gestational age 32 weeks and/or birth weight 1500 g, (2) ventilator dependency based on signs of chronic lung disease, (3) hydrocortisone treatment, and (4) availability of data as defined in the study protocol. Infants who were treated with dexamethasone, for example, after referral from other NICUs, were excluded as well as infants who were not ventilator dependent but had CPAP for respiratory support Study Protocol Demographic Variables. The following characteristics were retrospectively collected: gestational age (weeks), birth weight (grams), gender, small for gestational age <p2.3 (SGA), administration of antenatal steroids, premature prolonged rupture of membranes (>24 hours, PPROM), Apgar scores, and surfactant therapy Ventilator Settings. The charts of all preterm infants born after a pregnancy of 32 weeks and/or a birth weight 1500 g receiving hydrocortisone treatment were systematically reviewed for ventilation dependency. It was clear that in several days before the start of hydrocortisone treatment, no positive changes were seen in ventilator settings. Data on ventilator settings were recorded within an hour before the start of hydrocortisone therapy and again one day and two days later: inspiratory oxygen fraction (FiO 2 ), mean airway pressure (MAP; cm H 2 O), and arterial PaCO 2 (mm Hg). Duration of mechanical ventilation and oxygen requirements at postmenstrual age of 36 weeks were documented Ventilation Policy. The targeted level of oxygen saturation and PaCO 2 was 88 93% and mm Hg, respectively Hydrocortisone Treatment. Generally, hydrocortisone was started when the postnatal age was at least more than 7 days and the child was ventilator-dependent with increasing oxygen requirements (FiO 2 more than 0.40), prolonged dependency of extra oxygen (FiO 2 of more than 0.40) based on chronic lung disease as evidenced by repeated pulmonary X-rays. Echocardiography was performed in all patients to exclude a hemodynamically significant patent ductus arteriosus before considering hydrocortisone. Hydrocortisone was given at a starting dose of 5 mg/kg/day tapering off to 1.25 mg/kg/day over a 22-day period. Depending on the response to therapy, steroid treatment was either prolonged or repeated. Weaning from the ventilator occurred on the basis of clinical condition and blood gases. Postnatal age at start of treatment, number of days treated with hydrocortisone, and hydrocortisone dose (mg/ kg mean weight; mean weight: weight at the end of the treatment plus weight at the start of treatment divided by two) over the treatment period were recorded Neurological Outcome. Infants were evaluated with a standardized neurologic examination at corrected ages of 0, 6, 15, and 24 months using items from the methods of Amiel-Tison and Grenier [14], Touwen [15], and a Griffiths assessment [16]. Each child was examined for the presence of cerebral palsy. If present, neuromotor abnormalities including tone abnormalities/presence and type of cerebral palsy were recorded. Cerebral palsy was defined as a nonprogressive central nervous system disorder characterized by abnormal muscle tone in at least one extremity and abnormal control of movement and posture [8, 17].
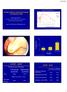
3 International Pediatrics 3 Table 1: Patient characteristics. Characteristics Total (n = 55) % Gestational age (wk) a 27.3 ± 1.4 Birth weight (g) a 924 ± 231 Gender c Female : male 17 : 38 SGA (<p2.3) c Antenatal steroids c PROM > 24 h c Apgar score b At 1 min 6 (1 10) At 5 min 8 (1 10) Mortality c 4 Postnatal age at death (days) 13, 18, 22, 63 Inotropes c Patent ductus arteriosus c Indometacin Surgery 12 # 38.7 NEC c NEC surgery requirement c IVH c Grades I-II 14 Grades III-IV 7 Cystic PVL c (grade II) 1 Data are shown as a mean ± SD, b median (range) or c n. SGA: small for gestational age, PROM: premature rupture of membranes, NEC: necrotizing enterocolitis, IVH: intraventricular haemorrhage, and PVL: periventricular leukomalacia. #In one case indometacin therapy was contraindicated because of IVH Study Endpoints. Primary outcome measures were ventilator settings after day 1 and day 2 of treatment, duration of mechanical ventilation, successful extubation after start of hydrocortisone, CLD at postmenstrual age of 36 weeks, mortality, and development of cerebral palsy Statistical Analysis. Data are summarized as means ± SD or median and ranges. Data on respiratory function of day 0, day 1, and day 2, that is, FiO2, MAP, and PaCO 2 were pairwise compared using Wilcoxon rank sum. A P-value < 0.05 was considered statistically significant. All analyses were performed using SPSS version for windows (SPSS, Chicago, Il, USA). 3. Results Fifty-five ventilation-dependent children fulfilled the inclusion criteria and were included in the present retrospective study. Table 1 shows the most important patient characteristics whereastable 2 shows respiratory characteristics. Hydrocortisone was initiated at a median age of 11 days (Table 2 and Figure 1). Median duration of hydrocortisone treatment was 21 days. In 52 out of 55 hydrocortisone-treated patients, the cumulative dose/kg mean weight could be calculated (mean Number of patients >27 Age at start hydrocortisone (days) Figure 1: Age at start treatment with hydrocortisone. Table 2: Respiratory characteristics. Characteristics Total (n = 55) % Duration of ventilation (days) b 15.0 (5 69) HFOV before treatment c Postnatal age at start of HC (days) b 11 (7 44) Duration of HC (days) d 21.0 (9.0) HC dose (mg/kg mean weight) d 56.9 (28.2) Extubation after start HC (days) d 4.0 (4.0) IRDS c Grades I-II Grades III-IV Surfactant c No Yes Pneumothorax c Data are shown as b median (range), c n or d median (interquartile range). HFOV: high frequency oscillation ventilation; HC: hydrocortisone; IRDS: infant respiratory distress syndrome. In one case it took 69 days to wean the child from ventilator because of ongoing sepsis. weight: weight at the end of treatment plus weight at the start of treatment divided by two). Median cumulative hydrocortisone dose was 56.9 mg/kg mean weight Respiratory Effects. All 55 patients, who received hydrocortisone while being ventilated, could be successfully extubated at a median age of 4 days. In one case, it was hard to wean the child from the ventilator because of intercurrent periods of Staphylococcus epidermidis sepsis. This child was eventually weaned after 69 days. Treatment with hydrocortisone was potent in reducing the need for extra oxygen (Figure 2). There was a significantly reduced requirement for oxygen after day 2 (P-value = 0.001) following start of hydrocortisone treatment and between day 1 and day 2 after start of hydrocortisone (P-value = 0.001).
4 4 International Pediatrics FiO2 (fraction) NS PaCO2 (mmhg) NS 0.2 Day 0 Day 1 Day 2 Figure 2: Values of oxygen requirement (inspirational oxygen fraction (FiO 2 )) on day 0 (baseline), day 1, and day 2 following treatment with hydrocortisone (HC). P < 0.05 versus day 0 (baseline) and day 1. NS = non significant. FiO 2 significantly decreases after day 2 compared with day 0 (baseline) and between day 1 and day 2. Mean airway pressure (cm H2O) Day 0 Day 1 Day 2 Figure 3: Values of Mean Airway Pressure (cm H 2 O) on day 0 (baseline), day 1, and day 2 following treatment with hydrocortisone (HC). P < 0.05 versus day 0 (baseline) and day 1. Mean airway pressure significantly decreases on day 1 and day 2 compared with day 0 (baseline). No significant reduced oxygen requirement was seen after one day (P-value = 0.006). Maximal effect of hydrocortisone treatment on MAP was seen after day 1 after start treatment (Figure 3). This effectwassignificant witha P-value of Furthermore, a significant effect was also seen after day 2 after start treatment (P-value < 0.001) and between day 1 and day 2 (P-value < 0.001). Hydrocortisone treatment had a maximal effect on PaCO 2 one day after start treatment (P-value < 0.001) (Figure 4). No significant effect on PaCO 2 was seen between day 1 and 2 after start hydrocortisone (P-value = 0.453). After a gestational age of 36 weeks, 11 children (21.1%) required supplemental oxygen. Four children died before 30 Day 0 Day 1 Day 2 Figure 4: Values of PaCO 2 (mmhg) on day 0 (baseline), day 1, and day 2 following treatment with hydrocortisone (HC). P < 0.05 versus day 0 (baseline) and day 1. NS = non significant. PaCO 2 significantly decreases on day 1 and day 2 compared with day 0 (baseline). gestational age of 36 weeks, and in one surviving case supplemental oxygen requirement was not noted Neurodevelopmental Followup. Fifty-five ventilationdependent children were included in this retrospective study. Of these, four were known to have died during neonatal period, leaving fifty-one patients available for followup at 24 months of corrected age (mean 24.1 ± 0.27 months). Standardized neurologic examination revealed that none of the 51 patients developed cerebral palsy. 4. Discussion Our findings suggest that moderately early hydrocortisone therapy, in the dose mentioned, leads to a significant reduction in oxygen requirement (FiO 2 ), PaCO 2,andmean airway pressure in ventilator-dependent preterm infants more than 7 days old who were at risk for CLD. Several studies indicate that low-dose dexamethasone administration after the first week of life significantly reduces the short-term pulmonary effects as failure to extubation and days of mechanical ventilation in ventilator-dependent preterm infants [5, 7, 18]. The study of Doyle et al. reported that dexamethasone (0.89 mg/kg/10 days) given at a median treatment age of 23 days to ventilator-dependent extremely low birthweight infants improved ventilator and oxygen requirements substantially and shortened the duration of intubation. There was little evidence for a reduction in either the mortality rate or the rate of oxygen dependency at 36 weeks [5]. Another study found that moderately early low-dose dexamethasone therapy for 7 days (tapering course, 0.2 to 0.1 mg/kg) had comparable beneficial effects to a high dose regimen. In both groups of infants (gestational age weeks; birth weight g), FiO 2 and MAP were significantly reduced [18]. A recent systematic review of 16 randomized, placebocontrolled trials (RTCs) including 1136 randomly assigned ventilated preterm infants more than 7 days of age assessed the effects of different cumulative dexamethasone doses on
5 International Pediatrics 5 CLD and neurodevelopmental outcome. This paper con cluded that higher cumulative dexamethasone doses ( 4mg/ kg) administered after the first week of life may decrease the relative risk for the combined outcome CLD or mortality. The risk of mortality or cerebral palsy was decreased by 6.2% for each incremental mg/kg cumulative dexamethasone dose when administered between 7 14 days of live. Furthermore, a significant reduction in short-term pulmonary effects as failure to extubation on day 3 and 7 and days of mechanical ventilation was seen in patients treated with dexamethasone after 7 days of live. These outcomes were independent of the cumulative dexamethasone dose [19]. Over the past years, concerns on the long-term neurodevelopmental consequences after dexamethasone therapy arose, when followup of a randomized controlled trial revealed a marked increase in neurodevelopmental dysfunction in neonates treated with dexamethasone compared with controls [11]. Since then, more reports of long-term negative neurodevelopmental sequelae have been published [10, 20, 21], and as a result The American Academy of Pediatrics stated in 2002 [22] that postnatal steroid administration, outside clinical trials, should be limited to exceptional clinical circumstances. In addition, a revised statement was issued by The American Academy of Pediatrics stating that current evidence is insufficient to make a recommendation regarding treatment with other glucocorticosteroids [23]. The long-term negative effects of dexamethasone necessitate a search for alternatives in the treatment of neonates with chronic lung disease. One of the possibilities frequently suggested in recent literature is the glucocorticosteroid hydrocortisone. Few studies [12, 24 26] have investigated the use of hydrocortisone for treatment of CLD in premature infants and described long-term neurological outcomes, reviewed by Rademaker et al. [13]. Van der Heide-Jalving et al. retrospectively studied in a group of 25 preterm infants (mean gestational age 28.3 weeks; mean birth weight 1040 g) the effect of highdose hydrocortisone on neurodevelopmental outcome. No significant differences were detected between the matched control group and hydrocortisone group with respect to neurological outcome, psychomotor development, and school performance at age 5 7 years. However, children treated with dexamethasone had a significantly less favourable school performance than their controls [12]. Another retrospective matched cohort study, Karemaker et al., compared behavioural outcome and motor skills in 192 preterm infants who neonatally either received dexamethasone or hydrocortisone for chronic lung disease. Dexamethasone- and hydrocortisone-treated groups were compared with a reference group and a group treated only antenatally with betamethasone. The groups were matched for gestational age, birth weight, gender, rate of respiratory distress syndrome, and rate of peri/intraventricular haemorrhage. At school age (7 10 years), the children treated with dexamethasone had a worse neurodevelopmental outcome than the reference group and betamethasone group, whereas the outcome of hydrocortisone-treated children did not differ from these two groups [24]. A large cohort study on long-term neurodevelopmental outcome in children following neonatal hydrocortisone treatment was reported in 2007 [25]. In this study, 226 preterm children (62 had been treated postnatally with hydrocortisone for CLD and 164 had not been treated with steroids) were followed up for 8 years. As this study was nonrandomized and treated children were generally sicker than untreated children, adjustments were made for gestational age, birth weight, gender, need for mechanical ventilation and small-for-gestational age. No differences were found in neurocognitive and motor performance between the two groups. Incidence of cerebral palsy and brain lesions on MRI was also similar. Watterberg et al. reported that early low-dose hydrocortisone therapy for prophylaxis of early adrenal insufficiency to prevent bronchopulmonary dysplasia did not increase the incidence of cerebral palsy in extremely low birth weight infants. Treated infants had indicators of improved developmental outcome at 18 to 22 months corrected age [26]. Recently, a prospective study was performed to evaluate the brain growth at term in preterm infants who did receive neonatal hydrocortisone for CLD. Thirty-eight preterm infants (n = 19 hydrocortisone, n = 19 controls) were matched for gestational age at birth. There were no differences in cerebral tissue volumes between the two groups at term equivalent age. In conclusion, no effect on brain growth was shown after treatment with hydrocortisone for CLD [27]. Our study supports the suggestion that hydrocortisone is not associated with cerebral palsy at 24 months adjusted age. According to findings of a recent study, we would have expected to find 2 5 cases of CP in our patients [28]. In 2010, Doyle et al. reviewed systematically the data from eight randomised controlled trials in newborn infants where hydrocortisone was compared with placebo to establish the balance between risks and benefits of hydrocortisone in newborn infants in the prevention or treatment of CLD [29]. In all eight trials, treatment was started in the first week of life; there were no trials of treatment started in infants who were chronically ventilated-dependent after the first week of life. A meta-analysis demonstrated little evidence to support the use of hydrocortisone (ranging in dose from 5 to 15 mg/kg) to prevent CLD; there were no improvements in important outcomes of mortality, or of rates in CLD or home oxygen dependence. To our knowledge, no studies are performed that assess the short-term pulmonary effects following hydrocortisone treatment. Our study provides the first evidence that, besides dexamethasone, hydrocortisone also facilitates extubation, reduces oxygen and ventilator requirements. Since shortterm pulmonary effects of dexamethasone and hydrocortisone are clinical comparable, and dexamethasone has proven long-term negative adverse effects, studies to use hydrocortisone in preterm ventilator-dependent infants at risk for CLD are warranted.
6 6 International Pediatrics 4.1. Limitations of the Study. The present study has also limitations. The primary limitation of our study is the lack of a control group. This lack is mainly related to its retrospective nature. Furthermore, the data obtained in this study were limited by the fact that we performed a retrospective cohort study and not a randomized prospective study. The sample size of our study population is small. The fifty-five patients in this study were born with a gestational age 32 weeks and/or birthweight of 1500 grams admitted over a period of five years to our NICU. Only a relatively small group fulfilled our inclusion criteria, a large group received hydrocortisone for other reasons than weaning from the ventilator. Bearing in mind the above considerations, the results of this study still provides useful information. We are convinced that this is the first step towards considering hydrocortisone as a therapy for prolonged ventilatordependent children at risk for CLD. Finally, the results of the present study urge for a large double-blind randomized controlled trial on hydrocortisone versus placebo treatment for ventilator-dependent children at risk for CLD to clarify the discussion about which corticosteroid (dexamethasone or hydrocortisone) is safer for treatment of preterm infants with chronic lung injury. References [1] W.H.Northway,R.C.Rosan,andD.Y.Porter, Pulmonary disease following respirator therapy of hyaline-membrane disease. Bronchopulmonary dysplasia, New England Journal of Medicine, vol. 276, no. 7, pp , [2] J. Hentschel, T. M. Berger, A. Tschopp, M. Müller, M. Adams, and H. U. Bucher, Population-based study of bronchopulmonary dysplasia in very low birth weight infants in Switzerland, European Pediatrics, vol. 164, no. 5, pp , [3] J. A. Lemons, C. R. Bauer, W. Oh et al., Very low birth weight outcomes of the National Institute of Child health and human development neonatal research network, January 1995 through December NICHD Neonatal Research Network, Pediatrics, vol. 107, no. 1, p. E1, [4] C. P. Speer, Pulmonary inflammation and bronchopulmonary dysplasia, Perinatology, vol. 26, supplement 1, pp. S57 S62, [5] L. W. Doyle, P. G. Davis, C. J. Morley et al., Low-dose dexamethasone facilitates extubation among chronically ventilatordependent infants: a multicenter, international, randomized, controlled trial, Pediatrics, vol. 117, no. 1, pp , [6] D. G. Grier and H. L. Halliday, Corticosteroids in the prevention and management of bronchopulmonary dysplasia, Seminars in Neonatology, vol. 8, no. 1, pp , [7] G. Suske, K. Oestreich, V. Varnholt, P. Lasch, and W. Kachel, Influence of early postnatal dexamethasone therapy on ventilator dependency in surfactant-substituted preterm infants, Acta Paediatrica, vol. 85, no. 6, pp , [8] B. Hagberg, G. Hagberg, and I. Olow, The changing panorama of cerebral palsy in Sweden II. Analysis of the various syndromes, Acta Paediatrica Scandinavica, vol. 64, no. 2, pp , [9] E. C. Eichenwald and A. R. Stark, Are postnatal steroids ever justified to treat severe bronchopulmonary dysplasia? Archives of Disease in Childhood, vol. 92, no. 5, pp. F334 F337, [10] E. S. Shinwell, M. Karplus, D. Reich et al., Early postnatal dexamethasone treatment and increased incidence of cerebral palsy, Archives of Disease in Childhood, vol. 83, no. 3, pp. F177 F181, [11] T. F. Yeh, Y. J. Lin, C. C. Huang et al., Early dexamethasone therapy in preterm infants: a follow-up study, Pediatrics, vol. 101, no. 5, p. E7, [12] M. Van Der Heide-Jalving, P. J. G. H. Kamphuis, M. J. Van Der Laan et al., Short- and long-term effects of neonatal glucocorticoid therapy: is hydrocortisone an alternative to dexamethasone? Acta Paediatrica, vol. 92, no. 7, pp , [13] K. J. Rademaker, L. S. De Vries, C. S. P. M. Uiterwaal, F. Groenendaal, D. E. Grobbee, and F. Van Bel, Postnatal hydrocortisone treatment for chronic lung disease in the preterm newborn and long-term neurodevelopmental followup, Archives of Disease in Childhood, vol. 93, no. 1, pp. F58 F63, [14] C. Amiel-Tison and A. Grenier, Evaluation Neurologique du Nouveau-ne et du Nourrisson, Masson, Paris, France, [15] B. C. L. Touwen, Examination of the Child with Minor Neurological Dysfunction, no. 71, Clinics in Developmental Medicine, SIMP/Heinemann, London, UK, [16] R. Griffiths, The Abilities of Babies: A Study in Mental Meas urement, Association of Research in Infant and Child Development, Amersham, Bucks, UK, [17] L. Mutch, E. Alberman, B. Hagberg, K. Kodama, and M. V. Perat, Cerebral palsy epidemiology: where are we now and where are we going? Developmental Medicine and Child Neurology, vol. 34, no. 6, pp , [18] M. Durand, M. E. Mendoza, P. Tantivit, A. Kugelman, and C. McEvoy, A randomized trial of moderately early lowdose dexamethasone therapy in very low birth weight infants: dynamic pulmonary mechanics, oxygenation, and ventilation, Pediatrics, vol. 109, no. 2 I, pp , [19] W. Onland, M. Offringa, A. P. D. Jaegere, and A. H. Van Kaam, Finding the optimal postnatal dexamethasone regimen for preterm infants at risk of bronchopulmonary dysplasia: a systematic review of placebo-controlled trials, Pediatrics, vol. 123, no. 1, pp , [20] T. M. O Shea, J. M. Kothadia, K. L. Klinepeter et al., Randomized placebo-controlled trial of a 42-day tapering course of dexamethasone to reduce the duration of ventilator dependency in very low birth weight infants: outcome of study participants at 1-year adjusted age, Pediatrics, vol. 104, no. 1 I, pp , [21] T. F. Yeh, Y. J. Lin, H. C. Lin et al., Outcomes at school age after postnatal dexamethasone therapy for lung disease of prematurity, New England Medicine, vol. 350, no. 13, pp , [22] L. R. Blackmon, E. F. Bell, W. A. Engle et al., Postnatal corticosteroids to treat or prevent chronic lung disease in preterm infants, Pediatrics, vol. 109, no. 2, pp , [23] K. L. Watterberg, L.-A. Papile, D. H. Adamkin et al., Postnatal corticosteroids to prevent or treat bronchopulmonary dysplasia, Pediatrics, vol. 126, no. 4, pp , [24] R. Karemaker, C. J. Heijnen, S. Veen et al., Differences in behavioral outcome and motor development at school age after
7 International Pediatrics 7 neonatal treatment for chronic lung disease with dexamethasone versus hydrocortisone, Pediatric Research, vol. 60, no. 6, pp , [25] K. J. Rademaker, C. S. P. M. Uiterwaal, F. Groenendaal et al., Neonatal hydrocortisone treatment: neurodevelopmental outcome and MRI at school age in preterm-born children, Pediatrics, vol. 150, no. 4, pp , [26] K. L. Watterberg, M. L. Shaffer, M. J. Mishefske et al., Growth and neurodevelopmental outcomes after early lowdose hydrocortisone treatment in extremely low birth weight infants, Pediatrics, vol. 120, no. 1, pp , [27] M. J. N. L. Benders, F. Groenendaal, F. Van Bel et al., Brain development of the preterm neonate after neonatal hydrocortisone treatment for chronic lung disease, Pediatric Research, vol. 66, no. 5, pp , [28] F. Groenendaal, J. Termote, M. Van Der Heide-Jalving, I. Van Haastert, and L. De Vries, Complications affecting preterm neonates from 1991 to 2006: what have we gained? Acta Paediatrica, vol. 99, no. 3, pp , [29] L. W. Doyle, R. A. Ehrenkranz, and H. L. Halliday, Postnatal hydrocortisone for preventing or treating bronchopulmonary dysplasia in preterm infants: a systematic review, Neonatology, vol. 98, no. 2, pp , 2010.
8 MEDIATORS of INFLAMMATION The Scientific World Journal Gastroenterology Research and Practice Diabetes Research International Endocrinology Immunology Research Disease Markers Submit your manuscripts at BioMed Research International PPAR Research Obesity Ophthalmology Evidence-Based Complementary and Alternative Medicine Stem Cells International Oncology Parkinson s Disease Computational and Mathematical Methods in Medicine AIDS Behavioural Neurology Research and Treatment Oxidative Medicine and Cellular Longevity
Noah Hillman M.D. IPOKRaTES Conference Guadalajaira, Mexico August 23, 2018
 Postnatal Steroids Use for Bronchopulmonary Dysplasia in 2018 + = Noah Hillman M.D. IPOKRaTES Conference Guadalajaira, Mexico August 23, 2018 AAP Policy Statement - 2002 This statement is intended for
Postnatal Steroids Use for Bronchopulmonary Dysplasia in 2018 + = Noah Hillman M.D. IPOKRaTES Conference Guadalajaira, Mexico August 23, 2018 AAP Policy Statement - 2002 This statement is intended for
Hazards and Benefits of Postnatal Steroids. David J. Burchfield, MD Professor and Chief, Neonatology University of Florida
 Hazards and Benefits of Postnatal Steroids David J. Burchfield, MD Professor and Chief, Neonatology University of Florida Disclosures I have no financial affiliations or relationships to disclose. I will
Hazards and Benefits of Postnatal Steroids David J. Burchfield, MD Professor and Chief, Neonatology University of Florida Disclosures I have no financial affiliations or relationships to disclose. I will
ROLE OF EARLY POSTNATAL DEXAMETHASONE IN RESPIRATORY DISTRESS SYNDROME
 INDIAN PEDIATRICS VOLUME 35-FEBRUAKY 1998 ROLE OF EARLY POSTNATAL DEXAMETHASONE IN RESPIRATORY DISTRESS SYNDROME Kanya Mukhopadhyay, Praveen Kumar and Anil Narang From the Division of Neonatology, Department
INDIAN PEDIATRICS VOLUME 35-FEBRUAKY 1998 ROLE OF EARLY POSTNATAL DEXAMETHASONE IN RESPIRATORY DISTRESS SYNDROME Kanya Mukhopadhyay, Praveen Kumar and Anil Narang From the Division of Neonatology, Department
Research Article Timing of Caffeine Therapy and Neonatal Outcomes in Preterm Infants: A Retrospective Study
 International Pediatrics Volume 2016, Article ID 9478204, 6 pages http://dx.doi.org/10.1155/2016/9478204 Research Article Timing of Caffeine Therapy and Neonatal Outcomes in Preterm Infants: A Retrospective
International Pediatrics Volume 2016, Article ID 9478204, 6 pages http://dx.doi.org/10.1155/2016/9478204 Research Article Timing of Caffeine Therapy and Neonatal Outcomes in Preterm Infants: A Retrospective
Name and title of the investigators responsible for conducting the research: Dr Anna Lavizzari, Dr Mariarosa Colnaghi
 Protocol title: Heated, Humidified High-Flow Nasal Cannula vs Nasal CPAP for Respiratory Distress Syndrome of Prematurity. Protocol identifying number: Clinical Trials.gov NCT02570217 Name and title of
Protocol title: Heated, Humidified High-Flow Nasal Cannula vs Nasal CPAP for Respiratory Distress Syndrome of Prematurity. Protocol identifying number: Clinical Trials.gov NCT02570217 Name and title of
Update on mangement of patent ductus arteriosus in preterm infants. Dr. Trinh Thi Thu Ha
 Update on mangement of patent ductus arteriosus in preterm infants Dr. Trinh Thi Thu Ha Outline 1. Overview of PDA 2. Timing of screening PDA? 3. When to treat PDA? Timing of ductal closure Prenatal
Update on mangement of patent ductus arteriosus in preterm infants Dr. Trinh Thi Thu Ha Outline 1. Overview of PDA 2. Timing of screening PDA? 3. When to treat PDA? Timing of ductal closure Prenatal
1
 1 2 3 RIFAI 5 6 Dublin cohort, retrospective review. Milrinone was commenced at an initial dose of 0.50 μg/kg/minute up to 0.75 μg/kg/minute and was continued depending on clinical response. No loading
1 2 3 RIFAI 5 6 Dublin cohort, retrospective review. Milrinone was commenced at an initial dose of 0.50 μg/kg/minute up to 0.75 μg/kg/minute and was continued depending on clinical response. No loading
Patent Ductus Arteriosus: Philosophy or Pathology?
 Patent Ductus Arteriosus: Philosophy or Pathology? Disclosure Ray Sato, MD is a speaker for Prolacta Biosciences, Inc. This presentation will discuss off-label uses of acetaminophen and ibuprofen. RAY
Patent Ductus Arteriosus: Philosophy or Pathology? Disclosure Ray Sato, MD is a speaker for Prolacta Biosciences, Inc. This presentation will discuss off-label uses of acetaminophen and ibuprofen. RAY
Kristi Watterberg, MD University of New Mexico
 Kristi Watterberg, MD University of New Mexico Dr. Watterberg has no financial or other conflicts of interest to disclose She will be discussing studies of glucocorticoids in preterm infants History how
Kristi Watterberg, MD University of New Mexico Dr. Watterberg has no financial or other conflicts of interest to disclose She will be discussing studies of glucocorticoids in preterm infants History how
USE OF INHALED NITRIC OXIDE IN THE NICU East Bay Newborn Specialists Guideline Prepared by P Joe, G Dudell, A D Harlingue Revised 7/9/2014
 USE OF INHALED NITRIC OXIDE IN THE NICU East Bay Newborn Specialists Guideline Prepared by P Joe, G Dudell, A D Harlingue Revised 7/9/2014 ino for Late Preterm and Term Infants with Severe PPHN Background:
USE OF INHALED NITRIC OXIDE IN THE NICU East Bay Newborn Specialists Guideline Prepared by P Joe, G Dudell, A D Harlingue Revised 7/9/2014 ino for Late Preterm and Term Infants with Severe PPHN Background:
Early (< 8 days) postnatal corticosteroids for preventing chronic lung disease in preterm infants (Review)
 Early (< 8 days) postnatal corticosteroids for preventing chronic lung disease in preterm infants (Review) Halliday HL, Ehrenkranz RA, Doyle LW This is a reprint of a Cochrane review, prepared and maintained
Early (< 8 days) postnatal corticosteroids for preventing chronic lung disease in preterm infants (Review) Halliday HL, Ehrenkranz RA, Doyle LW This is a reprint of a Cochrane review, prepared and maintained
Short-Term Outcome Of Different Treatment Modalities Of Patent Ductus Arteriosus In Preterm Infants. Five Years Experiences In Qatar
 ISPUB.COM The Internet Journal of Cardiovascular Research Volume 7 Number 2 Short-Term Outcome Of Different Treatment Modalities Of Patent Ductus Arteriosus In Preterm Infants. Five Years Experiences In
ISPUB.COM The Internet Journal of Cardiovascular Research Volume 7 Number 2 Short-Term Outcome Of Different Treatment Modalities Of Patent Ductus Arteriosus In Preterm Infants. Five Years Experiences In
PEDIATRIC PHARMACOTHERAPY
 PEDIATRIC PHARMACOTHERAPY Volume 22 Number 12 December 2016 A The Role of Hydrocortisone in the Management of Bronchopulmonary Dysplasia Emily Monds, PharmD lthough the clinical landscape of bronchopulmonary
PEDIATRIC PHARMACOTHERAPY Volume 22 Number 12 December 2016 A The Role of Hydrocortisone in the Management of Bronchopulmonary Dysplasia Emily Monds, PharmD lthough the clinical landscape of bronchopulmonary
PEDIATRIC NEWBORN MEDICINE CLINICAL PRACTICE GUIDELINES Pharmacologic Strategies for the Prevention of Bronchopulmonary Dysplasia
 PEDIATRIC NEWBORN MEDICINE CLINICAL PRACTICE GUIDELINES Pharmacologic Strategies for the Prevention of Bronchopulmonary Dysplasia Clinical Practice Guideline: Pharmacologic Strategies for the Prevention
PEDIATRIC NEWBORN MEDICINE CLINICAL PRACTICE GUIDELINES Pharmacologic Strategies for the Prevention of Bronchopulmonary Dysplasia Clinical Practice Guideline: Pharmacologic Strategies for the Prevention
NEONATAL NEWS Here s Some More Good Poop
 NEONATALNEWS Here ssomemoregoodpoop WINTEREDITION2010 THISNEWSLETTERISPUBLISHEDPERIODICALLYBYTHENEONATOLOGISTSOF ASSOCIATESINNEWBORNMEDICINETOCONVEYNEWANDUPDATEDPOLICIES ANDGUIDELINESANDPROVIDEGENERALEDUCATIONTONICUCARETAKERSAT
NEONATALNEWS Here ssomemoregoodpoop WINTEREDITION2010 THISNEWSLETTERISPUBLISHEDPERIODICALLYBYTHENEONATOLOGISTSOF ASSOCIATESINNEWBORNMEDICINETOCONVEYNEWANDUPDATEDPOLICIES ANDGUIDELINESANDPROVIDEGENERALEDUCATIONTONICUCARETAKERSAT
Original Policy Date
 MP 8.01.17 Inhaled Nitric Oxide Medical Policy Section Therapy Issue 12/2013 Original Policy Date 12/2013 Last Review Status/Date Reviewed with literature search/12/2013 Return to Medical Policy Index
MP 8.01.17 Inhaled Nitric Oxide Medical Policy Section Therapy Issue 12/2013 Original Policy Date 12/2013 Last Review Status/Date Reviewed with literature search/12/2013 Return to Medical Policy Index
TEXAS CHILDREN S HOSPITAL EVIDENCE-BASED OUTCOMES CENTER Respiratory Management of the Preterm Infant Evidence Summary
 TEXAS CHILDREN S HOSPITAL EVIDENCE-BASED OUTCOMES CENTER Respiratory Management of the Preterm Infant Evidence Summary DATE: April 2016 Critically Analyze the Evidence The GRADE criteria were used to evaluate
TEXAS CHILDREN S HOSPITAL EVIDENCE-BASED OUTCOMES CENTER Respiratory Management of the Preterm Infant Evidence Summary DATE: April 2016 Critically Analyze the Evidence The GRADE criteria were used to evaluate
Rescue Therapies in Neonatology. February 2014 Bill Walsh Monroe Carell Jr Children s Vanderbilt
 Rescue Therapies in Neonatology February 2014 Bill Walsh Monroe Carell Jr Children s Vanderbilt Off Label Use-Disclaimer All discussion in this talk concerns off-label use of nitric oxide, steroids, and
Rescue Therapies in Neonatology February 2014 Bill Walsh Monroe Carell Jr Children s Vanderbilt Off Label Use-Disclaimer All discussion in this talk concerns off-label use of nitric oxide, steroids, and
Clinical Study Incidence of Retinopathy of Prematurity in Extremely Premature Infants
 ISRN Pediatrics, Article ID 134347, 4 pages http://dx.doi.org/10.1155/2014/134347 Clinical Study Incidence of Retinopathy of Prematurity in Extremely Premature Infants Alparslan Fahin, Muhammed Fahin,
ISRN Pediatrics, Article ID 134347, 4 pages http://dx.doi.org/10.1155/2014/134347 Clinical Study Incidence of Retinopathy of Prematurity in Extremely Premature Infants Alparslan Fahin, Muhammed Fahin,
Hyaline membrane disease. By : Dr. Ch Sarishma Peadiatric Pg
 Hyaline membrane disease By : Dr. Ch Sarishma Peadiatric Pg Also called Respiratory distress syndrome. It occurs primarily in premature infants; its incidence is inversely related to gestational age and
Hyaline membrane disease By : Dr. Ch Sarishma Peadiatric Pg Also called Respiratory distress syndrome. It occurs primarily in premature infants; its incidence is inversely related to gestational age and
Kugelman A, Riskin A, Said W, Shoris I, Mor F, Bader D.
 Heated, Humidified High-Flow Nasal Cannula (HHHFNC) vs. Nasal Intermittent Positive Pressure Ventilation (NIPPV) for the Primary Treatment of RDS, A Randomized, Controlled, Prospective, Pilot Study Kugelman
Heated, Humidified High-Flow Nasal Cannula (HHHFNC) vs. Nasal Intermittent Positive Pressure Ventilation (NIPPV) for the Primary Treatment of RDS, A Randomized, Controlled, Prospective, Pilot Study Kugelman
Postnatal Corticosteroids for the Prevention and Treatment of Chronic Lung Disease in Preterm Infants
 Postnatal Corticosteroids for the Prevention and Treatment of Chronic Lung Disease in Preterm Infants Clinical trial evidence for the effectiveness of corticosteroids in the prevention of CLD is strong
Postnatal Corticosteroids for the Prevention and Treatment of Chronic Lung Disease in Preterm Infants Clinical trial evidence for the effectiveness of corticosteroids in the prevention of CLD is strong
The New England Journal of Medicine A MULTICENTER TRIAL OF TWO DEXAMETHASONE REGIMENS IN VENTILATOR-DEPENDENT PREMATURE INFANTS
 A MULTICENTER TRIAL OF TWO REGIMENS IN VENTILATOR-DEPENDENT PREMATURE INFANTS LU-ANN PAPILE, M.D., JON E. TYSON, M.D., BARBARA J. STOLL, M.D., LINDA L. WRIGHT, M.D., EDWARD F. DONOVAN, M.D., CHARLES R.
A MULTICENTER TRIAL OF TWO REGIMENS IN VENTILATOR-DEPENDENT PREMATURE INFANTS LU-ANN PAPILE, M.D., JON E. TYSON, M.D., BARBARA J. STOLL, M.D., LINDA L. WRIGHT, M.D., EDWARD F. DONOVAN, M.D., CHARLES R.
Randomized study of nasal continuous positive airway pressure in the preterm infant with respiratory distress syndrome
 Acta Pñdiatr 92: 1±6. 2003 Randomized study of nasal continuous positive airway pressure in the preterm infant with respiratory distress syndrome J Tooley and M Dyke Department of Paediatrics, Norfolk
Acta Pñdiatr 92: 1±6. 2003 Randomized study of nasal continuous positive airway pressure in the preterm infant with respiratory distress syndrome J Tooley and M Dyke Department of Paediatrics, Norfolk
Postnatal Steroid (PNS) Administration: Rationale
 Postnatal Steroid (PNS) Administration: Rationale Background: Postnatal steroid use has been subjected to intense scrutiny in recent years, the subject of several metaanalyses and most recently been the
Postnatal Steroid (PNS) Administration: Rationale Background: Postnatal steroid use has been subjected to intense scrutiny in recent years, the subject of several metaanalyses and most recently been the
Review Article Postnatal Corticosteroids for Prevention and Treatment of Chronic Lung Disease in the Preterm Newborn
 International Journal of Pediatrics Volume 2012, Article ID 315642, 12 pages doi:10.1155/2012/315642 Review Article Postnatal Corticosteroids for Prevention and Treatment of Chronic Lung Disease in the
International Journal of Pediatrics Volume 2012, Article ID 315642, 12 pages doi:10.1155/2012/315642 Review Article Postnatal Corticosteroids for Prevention and Treatment of Chronic Lung Disease in the
Chronic Lung Disease Of Prematurity. Dr Jo Harrison
 Chronic Lung Disease Of Prematurity Dr Jo Harrison 9.9.14 Chronic Neonatal Lung Disease Bronchopulmonary dysplasia (BPD) first described in 1967 by Northway Defined as O 2 dependence at 28 days post birth
Chronic Lung Disease Of Prematurity Dr Jo Harrison 9.9.14 Chronic Neonatal Lung Disease Bronchopulmonary dysplasia (BPD) first described in 1967 by Northway Defined as O 2 dependence at 28 days post birth
Admission/Discharge Form for Infants Born in Please DO NOT mail or fax this form to the CPQCC Data Center. This form is for internal use ONLY.
 Selection Criteria Admission/Discharge Form for Infants Born in 2016 To be eligible, you MUST answer YES to at least one of the possible criteria (A-C) A. 401 1500 grams o Yes B. GA range 22 0/7 31 6/7
Selection Criteria Admission/Discharge Form for Infants Born in 2016 To be eligible, you MUST answer YES to at least one of the possible criteria (A-C) A. 401 1500 grams o Yes B. GA range 22 0/7 31 6/7
Bubble CPAP for Respiratory Distress Syndrome in Preterm Infants
 R E S E A R C H P A P E R Bubble CPAP for Respiratory Distress Syndrome in Preterm Infants JAGDISH KOTI*, SRINIVAS MURKI, PRAMOD GADDAM, ANUPAMA REDDY AND M DASARADHA RAMI REDDY From Fernandez Hospital
R E S E A R C H P A P E R Bubble CPAP for Respiratory Distress Syndrome in Preterm Infants JAGDISH KOTI*, SRINIVAS MURKI, PRAMOD GADDAM, ANUPAMA REDDY AND M DASARADHA RAMI REDDY From Fernandez Hospital
FANNP 28TH NATIONAL NNP SYMPOSIUM: CLINICAL UPDATE AND REVIEW OCTOBER 17-21, 2017
 Pulse Oximetry in the Delivery Room: Principles and Practice GS2 3 Jonathan P. Mintzer, MD, FAAP Assistant Professor of Pediatrics Stony Brook Children s Hospital, Division of Neonatal-Perinatal Medicine,
Pulse Oximetry in the Delivery Room: Principles and Practice GS2 3 Jonathan P. Mintzer, MD, FAAP Assistant Professor of Pediatrics Stony Brook Children s Hospital, Division of Neonatal-Perinatal Medicine,
Surfactant Administration
 Approved by: Surfactant Administration Gail Cameron Senior Director Operations, Maternal, Neonatal & Child Health Programs Dr. Paul Byrne Medical Director, Neonatology Neonatal Policy & Procedures Manual
Approved by: Surfactant Administration Gail Cameron Senior Director Operations, Maternal, Neonatal & Child Health Programs Dr. Paul Byrne Medical Director, Neonatology Neonatal Policy & Procedures Manual
SAMPLE. V.12.1 Special Report: Very Low Birthweight Neonates. I. Introduction
 I. Introduction V.12.1 Special Report: Very Low Birthweight Neonates The delivery of a very low birth weight infant continues to present many challenges to families and health care providers in spite of
I. Introduction V.12.1 Special Report: Very Low Birthweight Neonates The delivery of a very low birth weight infant continues to present many challenges to families and health care providers in spite of
AMERICAN ACADEMY OF PEDIATRICS CANADIAN PAEDIATRIC SOCIETY. Postnatal Corticosteroids to Treat or Prevent Chronic Lung Disease in Preterm Infants
 AMERICAN ACADEMY OF PEDIATRICS Committee on Fetus and Newborn CANADIAN PAEDIATRIC SOCIETY Fetus and Newborn Committee Postnatal Corticosteroids to Treat or Prevent Chronic Lung Disease in Preterm Infants
AMERICAN ACADEMY OF PEDIATRICS Committee on Fetus and Newborn CANADIAN PAEDIATRIC SOCIETY Fetus and Newborn Committee Postnatal Corticosteroids to Treat or Prevent Chronic Lung Disease in Preterm Infants
PATENT DUCTUS ARTERIOSUS IN THE PRETERM INFANT EVIDENCE FOR & AGAINST TREATMENT
 PATENT DUCTUS ARTERIOSUS IN THE PRETERM INFANT EVIDENCE FOR & AGAINST TREATMENT Dr. Youssef Abou Zanouna, FRCPI, FACC Consultant Pediatric Cardiologist King Fahd Military Medical Complex Dhahran Introduction
PATENT DUCTUS ARTERIOSUS IN THE PRETERM INFANT EVIDENCE FOR & AGAINST TREATMENT Dr. Youssef Abou Zanouna, FRCPI, FACC Consultant Pediatric Cardiologist King Fahd Military Medical Complex Dhahran Introduction
Usefulness of DuoPAP in the treatment of very low birth weight preterm infants with neonatal respiratory distress syndrome
 European Review for Medical and Pharmacological Sciences 2015; 19: 573-577 Usefulness of DuoPAP in the treatment of very low birth weight preterm infants with neonatal respiratory distress syndrome B.
European Review for Medical and Pharmacological Sciences 2015; 19: 573-577 Usefulness of DuoPAP in the treatment of very low birth weight preterm infants with neonatal respiratory distress syndrome B.
SWISS SOCIETY OF NEONATOLOGY. Preterm infant with. pulmonary hypertension and hypopituitarism
 SWISS SOCIETY OF NEONATOLOGY Preterm infant with pulmonary hypertension and hypopituitarism November 2007 2 Pilgrim S, Stocker M, Neonatal and Pediatric Intensiv Care Unit, Children s Hospital of Lucerne,
SWISS SOCIETY OF NEONATOLOGY Preterm infant with pulmonary hypertension and hypopituitarism November 2007 2 Pilgrim S, Stocker M, Neonatal and Pediatric Intensiv Care Unit, Children s Hospital of Lucerne,
Minimizing Lung Damage During Respiratory Support
 Minimizing Lung Damage During Respiratory Support University of Miami Jackson Memorial Medical Center Care of the Sick Newborn 15 Eduardo Bancalari MD University of Miami Miller School of Medicine Jackson
Minimizing Lung Damage During Respiratory Support University of Miami Jackson Memorial Medical Center Care of the Sick Newborn 15 Eduardo Bancalari MD University of Miami Miller School of Medicine Jackson
An Overview of Bronchopulmonary Dysplasia and Chronic Lung Disease in Infancy
 An Overview of Bronchopulmonary Dysplasia and Chronic Lung Disease in Infancy Housekeeping: I have no financial disclosures Learning objectives: Develop an understanding of bronchopulmonary dysplasia (BPD)
An Overview of Bronchopulmonary Dysplasia and Chronic Lung Disease in Infancy Housekeeping: I have no financial disclosures Learning objectives: Develop an understanding of bronchopulmonary dysplasia (BPD)
Incidence of Bronchopulmonary Dysplasia in Korea
 ORIGINAL ARTICLE Pediatrics http://dx.doi.org/1.3346/jkms.12.27.8.914 J Korean Med Sci 12; 27: 914-921 Incidence of Bronchopulmonary Dysplasia in Korea Chang Won Choi 1,2, Beyong Il Kim 1,2, Ee-Kyung Kim
ORIGINAL ARTICLE Pediatrics http://dx.doi.org/1.3346/jkms.12.27.8.914 J Korean Med Sci 12; 27: 914-921 Incidence of Bronchopulmonary Dysplasia in Korea Chang Won Choi 1,2, Beyong Il Kim 1,2, Ee-Kyung Kim
Research Roundtable Summary
 Research Roundtable Summary 10 TENTH in a Series of Seminars on MCHB-funded Research Projects Early Cortisol Deficiency and Bronchopulmonary Dysplasia October 18, 1995 Parklawn Building Potomac Conference
Research Roundtable Summary 10 TENTH in a Series of Seminars on MCHB-funded Research Projects Early Cortisol Deficiency and Bronchopulmonary Dysplasia October 18, 1995 Parklawn Building Potomac Conference
COMPARISON OF THE EFFICIENCY OF CAFFEINE VERSUS AMINOPHYLLINE FOR THE TREATMENT OF APNOEA OF PREMATURITY
 CASE STUDIES COMPARISON OF THE EFFICIENCY OF CAFFEINE VERSUS AMINOPHYLLINE FOR THE TREATMENT OF APNOEA OF PREMATURITY Gabriela Ildiko Zonda 1, Andreea Avasiloaiei 1, Mihaela Moscalu 2, Maria Stamatin 1
CASE STUDIES COMPARISON OF THE EFFICIENCY OF CAFFEINE VERSUS AMINOPHYLLINE FOR THE TREATMENT OF APNOEA OF PREMATURITY Gabriela Ildiko Zonda 1, Andreea Avasiloaiei 1, Mihaela Moscalu 2, Maria Stamatin 1
CPAP failure in preterm infants: incidence, predictors and consequences
 CPAP failure in preterm infants: incidence, predictors and consequences SUPPLEMENTAL TEXT METHODS Study setting The Royal Hobart Hospital has an 11-bed combined Neonatal and Paediatric Intensive Care Unit
CPAP failure in preterm infants: incidence, predictors and consequences SUPPLEMENTAL TEXT METHODS Study setting The Royal Hobart Hospital has an 11-bed combined Neonatal and Paediatric Intensive Care Unit
The Basics. Editorial Team. Editorial Team 9/28/2017
 Title of Program: What has Cochrane Neonatal Done For Babies? Speakers/Moderators: Roger F. Soll, MD Planning Committee: Jeffery D. Horbar, MD, Madge E. Buus-Frank, RN, MS, APRN-BC, FAAN, Roger F. Soll,
Title of Program: What has Cochrane Neonatal Done For Babies? Speakers/Moderators: Roger F. Soll, MD Planning Committee: Jeffery D. Horbar, MD, Madge E. Buus-Frank, RN, MS, APRN-BC, FAAN, Roger F. Soll,
Planning Committee: Jeffery D. Horbar, MD, Madge E. Buus-Frank, RN, MS, APRN-BC, FAAN, Roger F. Soll, MD
 Title of Program: What has Cochrane Neonatal Done For Babies? Speakers/Moderators: Roger F. Soll, MD Planning Committee: Jeffery D. Horbar, MD, Madge E. Buus-Frank, RN, MS, APRN-BC, FAAN, Roger F. Soll,
Title of Program: What has Cochrane Neonatal Done For Babies? Speakers/Moderators: Roger F. Soll, MD Planning Committee: Jeffery D. Horbar, MD, Madge E. Buus-Frank, RN, MS, APRN-BC, FAAN, Roger F. Soll,
SWISS SOCIETY OF NEONATOLOGY. Spontaneous intestinal perforation or necrotizing enterocolitis?
 SWISS SOCIETY OF NEONATOLOGY Spontaneous intestinal perforation or necrotizing enterocolitis? June 2004 2 Stocker M, Berger TM, Neonatal and Pediatric Intensive Care Unit, Children s Hospital of Lucerne,
SWISS SOCIETY OF NEONATOLOGY Spontaneous intestinal perforation or necrotizing enterocolitis? June 2004 2 Stocker M, Berger TM, Neonatal and Pediatric Intensive Care Unit, Children s Hospital of Lucerne,
Is There a Treatment for BPD?
 Is There a Treatment for BPD? Amir Kugelman, Pediatric Pulmonary Unit and Department of Neonatology Bnai Zion Medical Center, Rappaport Faculty of Medicine Haifa, Israel Conflict of Interest Our study
Is There a Treatment for BPD? Amir Kugelman, Pediatric Pulmonary Unit and Department of Neonatology Bnai Zion Medical Center, Rappaport Faculty of Medicine Haifa, Israel Conflict of Interest Our study
Prophylactic nasal continuous positive airway pressure for preventing morbidity and mortality in very preterm infants(review)
 Cochrane Database of Systematic Reviews Prophylactic nasal continuous positive airway pressure for preventing morbidity and mortality in very preterm infants (Review) SubramaniamP,HoJJ,DavisPG Subramaniam
Cochrane Database of Systematic Reviews Prophylactic nasal continuous positive airway pressure for preventing morbidity and mortality in very preterm infants (Review) SubramaniamP,HoJJ,DavisPG Subramaniam
AEROSURF Phase 2 Program Update Investor Conference Call
 AEROSURF Phase 2 Program Update Investor Conference Call November 12, 2015 Forward Looking Statement To the extent that statements in this presentation are not strictly historical, including statements
AEROSURF Phase 2 Program Update Investor Conference Call November 12, 2015 Forward Looking Statement To the extent that statements in this presentation are not strictly historical, including statements
increased incidence of cerebral
 Arch Dis Child Fetal Neonatal Ed 2000;83:F177 F181 Kaplan Medical Center, Rechovot, Israel E S Shinwell S Mogilner Soroka Medical Center, Beersheva M Karplus Ha emek Medical Center, Afula D Reich Carmel
Arch Dis Child Fetal Neonatal Ed 2000;83:F177 F181 Kaplan Medical Center, Rechovot, Israel E S Shinwell S Mogilner Soroka Medical Center, Beersheva M Karplus Ha emek Medical Center, Afula D Reich Carmel
NEONATAL CLINICAL PRACTICE GUIDELINE
 NEONATAL CLINICAL PRACTICE GUIDELINE Title: Brain Oxygen Monitoring in Newborns Using Near Infrared Spectroscopy (NIRS) Approval Date: Pages: June 2016 Approved by: Neonatal Patient Care Teams, HSC & SBH
NEONATAL CLINICAL PRACTICE GUIDELINE Title: Brain Oxygen Monitoring in Newborns Using Near Infrared Spectroscopy (NIRS) Approval Date: Pages: June 2016 Approved by: Neonatal Patient Care Teams, HSC & SBH
Objectives. Apnea Definition and Pitfalls. Pathophysiology of Apnea. Apnea of Prematurity and hypoxemia episodes 5/18/2015
 Apnea of Prematurity and hypoxemia episodes Deepak Jain MD Care of Sick Newborn Conference May 2015 Objectives Differentiating between apnea and hypoxemia episodes. Pathophysiology Diagnosis of apnea and
Apnea of Prematurity and hypoxemia episodes Deepak Jain MD Care of Sick Newborn Conference May 2015 Objectives Differentiating between apnea and hypoxemia episodes. Pathophysiology Diagnosis of apnea and
With wider acceptance of the early use of
 Weaning of nasal CPAP in preterm infants: who, when and how? a systematic review of literature Shaili Amatya, Deepa Rastogi, Alok Bhutada, Shantanu Rastogi New York, USA Background: There is increased
Weaning of nasal CPAP in preterm infants: who, when and how? a systematic review of literature Shaili Amatya, Deepa Rastogi, Alok Bhutada, Shantanu Rastogi New York, USA Background: There is increased
Pneumothorax in preterm babies has been associated with an increased risk of intraventricular hemorrhage (IVH)
 Identification of Pneumothorax in Very Preterm Infants Risha Bhatia, MB, MA, MRCPCH, Peter G. Davis, MD, FRACP, Lex W. Doyle, MD, FRACP, Connie Wong, RN, and Colin J. Morley, MD, FRACP, FRCPCH Objective
Identification of Pneumothorax in Very Preterm Infants Risha Bhatia, MB, MA, MRCPCH, Peter G. Davis, MD, FRACP, Lex W. Doyle, MD, FRACP, Connie Wong, RN, and Colin J. Morley, MD, FRACP, FRCPCH Objective
ADMISSION/DISCHARGE FORM FOR INFANTS BORN IN 2019 DO NOT mail or fax this form to the CPQCC Data Center. This form is for internal use ONLY.
 1 Any eligible inborn infant who dies in the delivery room or at any other location in your hospital within 12 hours after birth and prior to admission to the NICU is defined as a "Delivery Room Death."
1 Any eligible inborn infant who dies in the delivery room or at any other location in your hospital within 12 hours after birth and prior to admission to the NICU is defined as a "Delivery Room Death."
Rango de saturacion de oxigeno: Cual es la evidencia?
 Rango de saturacion de oxigeno: Cual es la evidencia? Wally Carlo, M.D. University of Alabama at Birmingham Department of Pediatrics Division of Neonatology wcarlo@peds.uab.edu 1 2 Stevie Wonder 4 Objectives
Rango de saturacion de oxigeno: Cual es la evidencia? Wally Carlo, M.D. University of Alabama at Birmingham Department of Pediatrics Division of Neonatology wcarlo@peds.uab.edu 1 2 Stevie Wonder 4 Objectives
Uses 1,2,3 : Labeled: Prevention of respiratory distress syndrome in premature infants
 Brand Name: Surfaxin Generic Name: lucinactant Manufacturer 1 : Discovery Laboratories, Inc. Drug Class 2,3 : Synthetic lung surfactant Uses 1,2,3 : Labeled: Prevention of respiratory distress syndrome
Brand Name: Surfaxin Generic Name: lucinactant Manufacturer 1 : Discovery Laboratories, Inc. Drug Class 2,3 : Synthetic lung surfactant Uses 1,2,3 : Labeled: Prevention of respiratory distress syndrome
Early postnatal dexamethasone administration
 Controlled Trial of Early Dexamethasone Treatment for the Prevention of Chronic Lung Disease in Preterm Infants: A 3-Year Follow-up Costantino Romagnoli, MD*; Enrico Zecca, MD*; Rita Luciano, MD*; Giulia
Controlled Trial of Early Dexamethasone Treatment for the Prevention of Chronic Lung Disease in Preterm Infants: A 3-Year Follow-up Costantino Romagnoli, MD*; Enrico Zecca, MD*; Rita Luciano, MD*; Giulia
Prematurity as a Risk Factor for ASD. Disclaimer
 Prematurity as a Risk Factor for ASD Angela M. Montgomery, MD, MSEd Assistant Professor of Pediatrics (Neonatology) Director, Yale NICU GRAD Program Suzanne L. Macari, PhD Research Scientist, Child Study
Prematurity as a Risk Factor for ASD Angela M. Montgomery, MD, MSEd Assistant Professor of Pediatrics (Neonatology) Director, Yale NICU GRAD Program Suzanne L. Macari, PhD Research Scientist, Child Study
Hummi Micro Draw Blood Transfer Device. An Important Addition to Your IVH Bundle
 Hummi Micro Draw Blood Transfer Device An Important Addition to Your IVH Bundle Hummi Micro Draw & Micro T Connector For Infec6on Control and IVH Risk Reduc6on The Next Genera6on System for Closed Micro
Hummi Micro Draw Blood Transfer Device An Important Addition to Your IVH Bundle Hummi Micro Draw & Micro T Connector For Infec6on Control and IVH Risk Reduc6on The Next Genera6on System for Closed Micro
MEDICAL POLICY I. POLICY POLICY TITLE POLICY NUMBER INHALED NITRIC OXIDE MP-4.021
 Original Issue Date (Created): August 23, 2002 Most Recent Review Date (Revised): January 28, 2014 Effective Date: April 1, 2014 I. POLICY Inhaled nitric oxide may be considered medically necessary as
Original Issue Date (Created): August 23, 2002 Most Recent Review Date (Revised): January 28, 2014 Effective Date: April 1, 2014 I. POLICY Inhaled nitric oxide may be considered medically necessary as
STOP ROP The STOP-ROP Multicenter Study Group: Pediatrics 105:295, 2000 Progression to Threshold Conventional Sat 89-94% STOP ROP
 Hrs TcPO2 > 80 nnhg (weeks 1 4) OXYGEN TARGETS: HOW GOOD ARE WE IN ACHIEVING THEM Oxygen Dependency GA wks Eduardo Bancalari MD University of Miami Miller School of Medicine Jackson Memorial Medical Center
Hrs TcPO2 > 80 nnhg (weeks 1 4) OXYGEN TARGETS: HOW GOOD ARE WE IN ACHIEVING THEM Oxygen Dependency GA wks Eduardo Bancalari MD University of Miami Miller School of Medicine Jackson Memorial Medical Center
The Turkish Journal of Pediatrics 2014; 56:
 The Turkish Journal of Pediatrics 2014; 56: 232-237 Original INSURE method (INtubation-SURfactant-Extubation) in early and late premature neonates with respiratory distress: factors affecting the outcome
The Turkish Journal of Pediatrics 2014; 56: 232-237 Original INSURE method (INtubation-SURfactant-Extubation) in early and late premature neonates with respiratory distress: factors affecting the outcome
Evaluation of the Local Incidence and Determinants of Bronchopulmonary Dysplasia and Pulmonary Morbidity in Extremely Preterm Infants
 Evaluation of the Local Incidence and Determinants of Bronchopulmonary Dysplasia and Pulmonary Morbidity in Extremely Preterm Infants by Amber Elise Reichert A thesis submitted in partial fulfilment of
Evaluation of the Local Incidence and Determinants of Bronchopulmonary Dysplasia and Pulmonary Morbidity in Extremely Preterm Infants by Amber Elise Reichert A thesis submitted in partial fulfilment of
Bronchopulmonary dysplasia: incidence and risk factors
 Original article Arch Argent Pediatr 2017;115(5):476-482 / 476 Bronchopulmonary dysplasia: incidence and risk factors Pablo H. Brener Dik, M.D. a, Yeimy M. Niño Gualdron, M.D. a, María F. Galletti, M.D.
Original article Arch Argent Pediatr 2017;115(5):476-482 / 476 Bronchopulmonary dysplasia: incidence and risk factors Pablo H. Brener Dik, M.D. a, Yeimy M. Niño Gualdron, M.D. a, María F. Galletti, M.D.
Newborn Life Support. NLS guidance.
 Kelly Harvey, ANNP NWNODN, previously Wythenshawe Hospital has shared this presentation with the understanding that it is for personal use following your attendance at the 8th Annual Senior Neonatal Nursing
Kelly Harvey, ANNP NWNODN, previously Wythenshawe Hospital has shared this presentation with the understanding that it is for personal use following your attendance at the 8th Annual Senior Neonatal Nursing
Insults to the Developing Brain & Effect on Neurodevelopmental Outcomes
 Insults to the Developing Brain & Effect on Neurodevelopmental Outcomes Ira Adams-Chapman, MD Assistant Professor of Pediatrics Director, Developmental Progress Clinic Emory University School of Medicine
Insults to the Developing Brain & Effect on Neurodevelopmental Outcomes Ira Adams-Chapman, MD Assistant Professor of Pediatrics Director, Developmental Progress Clinic Emory University School of Medicine
McMASTER NICU INHALED STEROIDS FOR EVOLVING BPD (GA < 29 WEEKS)
 McMASTER NICU INHALED STEROIDS FOR EVOLVING BPD (GA < 29 WEEKS) Developed by: Amit Mukerji, Samira Samiee-Zafarghandy, Jennifer Twiss, Ereny Bassilious, Elizabeth Vo, Shari Gray, Salhab el Helou on behalf
McMASTER NICU INHALED STEROIDS FOR EVOLVING BPD (GA < 29 WEEKS) Developed by: Amit Mukerji, Samira Samiee-Zafarghandy, Jennifer Twiss, Ereny Bassilious, Elizabeth Vo, Shari Gray, Salhab el Helou on behalf
** SURFACTANT THERAPY**
 ** SURFACTANT THERAPY** Full Title of Guideline: Surfactant Therapy Author (include email and role): Stephen Wardle (V4) Reviewed by Dushyant Batra Consultant Neonatologist Division & Speciality: Division:
** SURFACTANT THERAPY** Full Title of Guideline: Surfactant Therapy Author (include email and role): Stephen Wardle (V4) Reviewed by Dushyant Batra Consultant Neonatologist Division & Speciality: Division:
IMPAIRED CEREBRAL CORTICAL GRAY MATTER GROWTH FOLLOWING TREATMENT WITH DEXAMETHASONE FOR NEONATAL CHRONIC LUNG DISEASE
 IMPAIRED CEREBRAL CORTICAL GRAY MATTER GROWTH FOLLOWING TREATMENT WITH DEXAMETHASONE FOR NEONATAL CHRONIC LUNG DISEASE Brendan P. Murphy, MB BCh 1, Terrie E. Inder, MD 2,5, Petra S. Huppi, MD 3,5, Simon
IMPAIRED CEREBRAL CORTICAL GRAY MATTER GROWTH FOLLOWING TREATMENT WITH DEXAMETHASONE FOR NEONATAL CHRONIC LUNG DISEASE Brendan P. Murphy, MB BCh 1, Terrie E. Inder, MD 2,5, Petra S. Huppi, MD 3,5, Simon
New Zealand Data Sheet. Poractant alfa (Phospholipid fraction of porcine lung) 80 mg/ml
 CUROSURF New Zealand Data Sheet Poractant alfa (Phospholipid fraction of porcine lung) 80 mg/ml Presentation Sterile suspension in single-dose vials for intratracheal or intrabronchial administration.
CUROSURF New Zealand Data Sheet Poractant alfa (Phospholipid fraction of porcine lung) 80 mg/ml Presentation Sterile suspension in single-dose vials for intratracheal or intrabronchial administration.
Received: Jun 15, 2013; Accepted: Nov 08, 2013; First Online Available: Jan 24, 2014
 Original Article Iran J Pediatr Feb 2014; Vol 24 (No 1), Pp: 57-63 Management of Neonatal Respiratory Distress Syndrome Employing ACoRN Respiratory Sequence Protocol versus Early Nasal Continuous Positive
Original Article Iran J Pediatr Feb 2014; Vol 24 (No 1), Pp: 57-63 Management of Neonatal Respiratory Distress Syndrome Employing ACoRN Respiratory Sequence Protocol versus Early Nasal Continuous Positive
Cover Page. The handle holds various files of this Leiden University dissertation
 Cover Page The handle http://hdl.handle.net/1887/46692 holds various files of this Leiden University dissertation Author: Zanten, Henriëtte van Title: Oxygen titration and compliance with targeting oxygen
Cover Page The handle http://hdl.handle.net/1887/46692 holds various files of this Leiden University dissertation Author: Zanten, Henriëtte van Title: Oxygen titration and compliance with targeting oxygen
GS3. Understanding How to Use Statistics to Evaluate an Article. Session Summary. Session Objectives. References. Session Outline
 GS3 Understanding How to Use Statistics to Evaluate an Article Reese H. Clark, MD Director of Research Pediatrix Medical Group Neonatologist Greenville Memorial Hospital, Greenville, SC The speaker has
GS3 Understanding How to Use Statistics to Evaluate an Article Reese H. Clark, MD Director of Research Pediatrix Medical Group Neonatologist Greenville Memorial Hospital, Greenville, SC The speaker has
Back to the Future: Updated Guidelines for Evaluation and Management of Adrenal Insufficiency in the Critically Ill
 Back to the Future: Updated Guidelines for Evaluation and Management of Adrenal Insufficiency in the Critically Ill Joe Palumbo PGY-2 Critical Care Pharmacy Resident Buffalo General Medical Center Disclosures
Back to the Future: Updated Guidelines for Evaluation and Management of Adrenal Insufficiency in the Critically Ill Joe Palumbo PGY-2 Critical Care Pharmacy Resident Buffalo General Medical Center Disclosures
Late(œ 7 days) systemic postnatal corticosteroids for prevention of bronchopulmonary dysplasia in preterm infants(review)
 Cochrane Database of Systematic Reviews Late(œ 7 days) systemic postnatal corticosteroids for prevention of bronchopulmonary dysplasia in preterm infants (Review) Doyle LW, Cheong JL, Ehrenkranz RA, Halliday
Cochrane Database of Systematic Reviews Late(œ 7 days) systemic postnatal corticosteroids for prevention of bronchopulmonary dysplasia in preterm infants (Review) Doyle LW, Cheong JL, Ehrenkranz RA, Halliday
Effects of permissive hypercapnia on pulmonary and neurodevelopmental sequelae in extremely low birth weight infants: a meta analysis
 DOI 10.1186/s40064-016-2437-5 REVIEW Open Access Effects of permissive hypercapnia on pulmonary and neurodevelopmental sequelae in extremely low birth weight infants: a meta analysis Jianglin Ma 1 and
DOI 10.1186/s40064-016-2437-5 REVIEW Open Access Effects of permissive hypercapnia on pulmonary and neurodevelopmental sequelae in extremely low birth weight infants: a meta analysis Jianglin Ma 1 and
Neurodevelopmental and Behavioral Outcome of Very Low Birth Weight Babies at Corrected Age of 2 Years
 Indian J Pediatr (2010) 77:963 967 DOI 10.1007/s12098-010-0149-3 ORIGINAL ARTICLE Neurodevelopmental and Behavioral Outcome of Very Low Birth Weight Babies at Corrected Age of 2 Years Kanya Mukhopadhyay
Indian J Pediatr (2010) 77:963 967 DOI 10.1007/s12098-010-0149-3 ORIGINAL ARTICLE Neurodevelopmental and Behavioral Outcome of Very Low Birth Weight Babies at Corrected Age of 2 Years Kanya Mukhopadhyay
O) or to bi-level NCPAP (group B; n=20, lower CPAP level=4.5 cm H 2
 1 NICU, Children s Hospital, Via Castelvetro, Milan, Italy 2 Laboratory, V.Buzzi Children s Hospital ICP, Milan, Italy 3 Department of Statistics, Catholic University, Milan, Italy Correspondence to Gianluca
1 NICU, Children s Hospital, Via Castelvetro, Milan, Italy 2 Laboratory, V.Buzzi Children s Hospital ICP, Milan, Italy 3 Department of Statistics, Catholic University, Milan, Italy Correspondence to Gianluca
Postnatal corticosteroids to treat or prevent chronic lung disease in preterm infants.
 Complete Summary GUIDELINE TITLE Postnatal corticosteroids to treat or prevent chronic lung disease in preterm infants. BIBLIOGRAPHIC SOURCE(S) Postnatal corticosteroids to treat or prevent chronic lung
Complete Summary GUIDELINE TITLE Postnatal corticosteroids to treat or prevent chronic lung disease in preterm infants. BIBLIOGRAPHIC SOURCE(S) Postnatal corticosteroids to treat or prevent chronic lung
Tehran University of Medical Sciences, Tehran, Iran. Received: 22 May 2013; Received in revised form: 12 Sep. 2013; Accepted: 17 Sep.
 ORIGINAL ARTICLE Comparison of INSURE Method with Conventional Mechanical Ventilation after Surfactant Administration in Preterm Infants with Respiratory Distress Syndrome: Therapeutic Challenge Fatemeh
ORIGINAL ARTICLE Comparison of INSURE Method with Conventional Mechanical Ventilation after Surfactant Administration in Preterm Infants with Respiratory Distress Syndrome: Therapeutic Challenge Fatemeh
Research Article Factors Affecting the Weaning from Nasal CPAP in Preterm Neonates
 International Pediatrics Volume 2012, Article ID 416073, 7 pages doi:10.1155/2012/416073 Research Article Factors Affecting the Weaning from Nasal CPAP in Preterm Neonates Shantanu Rastogi, 1, 2 Hariprem
International Pediatrics Volume 2012, Article ID 416073, 7 pages doi:10.1155/2012/416073 Research Article Factors Affecting the Weaning from Nasal CPAP in Preterm Neonates Shantanu Rastogi, 1, 2 Hariprem
Permissive Hypotension in Extremely Low Birth Weight Infants ( 1000 gm)
 Original Article http://dx.doi.org/./ymj.2.3.4.7 pissn: 3-79, eissn: 97-2437 Yonsei Med J 3(4):7-77, 2 Permissive Hypotension in Extremely Low Birth Weight Infants ( gm) So Yoon Ahn, Eun Sun Kim, Jin Kyu
Original Article http://dx.doi.org/./ymj.2.3.4.7 pissn: 3-79, eissn: 97-2437 Yonsei Med J 3(4):7-77, 2 Permissive Hypotension in Extremely Low Birth Weight Infants ( gm) So Yoon Ahn, Eun Sun Kim, Jin Kyu
CONTINUOUS POSITIVE AIRWAY PRESSURE (CPAP) DEFINITION
 CONTINUOUS POSITIVE AIRWAY PRESSURE (CPAP) DEFINITION Method of maintaining low pressure distension of lungs during inspiration and expiration when infant breathing spontaneously Benefits Improves oxygenation
CONTINUOUS POSITIVE AIRWAY PRESSURE (CPAP) DEFINITION Method of maintaining low pressure distension of lungs during inspiration and expiration when infant breathing spontaneously Benefits Improves oxygenation
SWISS SOCIETY OF NEONATOLOGY. Supercarbia in an infant with meconium aspiration syndrome
 SWISS SOCIETY OF NEONATOLOGY Supercarbia in an infant with meconium aspiration syndrome January 2006 2 Wilhelm C, Frey B, Department of Intensive Care and Neonatology, University Children s Hospital Zurich,
SWISS SOCIETY OF NEONATOLOGY Supercarbia in an infant with meconium aspiration syndrome January 2006 2 Wilhelm C, Frey B, Department of Intensive Care and Neonatology, University Children s Hospital Zurich,
Benefits of Caffeine Citrate: Neurodevelopmental Outcomes of ELBW Infants
 St. Catherine University SOPHIA Master of Arts in Nursing Theses Nursing 12-2011 Benefits of Caffeine Citrate: Neurodevelopmental Outcomes of ELBW Infants Teri Johnson St. Catherine University Follow this
St. Catherine University SOPHIA Master of Arts in Nursing Theses Nursing 12-2011 Benefits of Caffeine Citrate: Neurodevelopmental Outcomes of ELBW Infants Teri Johnson St. Catherine University Follow this
Provide guidelines for the management of mechanical ventilation in infants <34 weeks gestation.
 Page 1 of 5 PURPOSE: Provide guidelines for the management of mechanical ventilation in infants
Page 1 of 5 PURPOSE: Provide guidelines for the management of mechanical ventilation in infants
BRONCHOPULMONARY DYSPLASIA
 BRONCHOPULMONARY DYSPLASIA CHRONIC NEONATAL LUNG DISEASE (CLD) 2 2 nd BERLIN NEONATOLOGY SUMMER SCHOOL September 2014 3 Mt. Scopus 4 Ein Kerem 5 BRONCHOPULMONARY DYSPLASIA 1960: Ventilation of Neonates
BRONCHOPULMONARY DYSPLASIA CHRONIC NEONATAL LUNG DISEASE (CLD) 2 2 nd BERLIN NEONATOLOGY SUMMER SCHOOL September 2014 3 Mt. Scopus 4 Ein Kerem 5 BRONCHOPULMONARY DYSPLASIA 1960: Ventilation of Neonates
Respiratory Management and Outcome of Preterm Infants
 Respiratory Management and Outcome of Preterm Infants 6 th Annual Care Of The Sick Newborn Conference Shu Wu, MD. Department of Pediatrics Division of Neonatology University of Miami School of Medicine
Respiratory Management and Outcome of Preterm Infants 6 th Annual Care Of The Sick Newborn Conference Shu Wu, MD. Department of Pediatrics Division of Neonatology University of Miami School of Medicine
Ibuprofen for the prevention of patent ductus arteriosus in preterm and/or low birth weight infants (Review)
 Ibuprofen for the prevention of patent ductus arteriosus in preterm and/or low birth weight infants (Review) Ohlsson A, Shah SS This is a reprint of a Cochrane review, prepared and maintained by The Cochrane
Ibuprofen for the prevention of patent ductus arteriosus in preterm and/or low birth weight infants (Review) Ohlsson A, Shah SS This is a reprint of a Cochrane review, prepared and maintained by The Cochrane
SURFACTANT UPDATE. George Mandy, M.D. Nationwide Children s Hospital The Ohio State University
 SURFACTANT UPDATE George Mandy, M.D. Nationwide Children s Hospital The Ohio State University Surfactant Update Objectives History Meta-analysis of surfactant therapy New synthetic surfactant Genetic disorders
SURFACTANT UPDATE George Mandy, M.D. Nationwide Children s Hospital The Ohio State University Surfactant Update Objectives History Meta-analysis of surfactant therapy New synthetic surfactant Genetic disorders
Non-invasive ventilatory strategies, such as
 R E S E A R C H P A P E R Heated Humidified High Flow Nasal Cannula versus Nasal Continuous Positive Airway Pressure as Primary Mode of Respiratory Support for Respiratory Distress in Preterm Infants DEEPARAJ
R E S E A R C H P A P E R Heated Humidified High Flow Nasal Cannula versus Nasal Continuous Positive Airway Pressure as Primary Mode of Respiratory Support for Respiratory Distress in Preterm Infants DEEPARAJ
Clinicoetiological profile and risk assessment of newborn with respiratory distress in a tertiary care centre in South India
 International Journal of Contemporary Pediatrics Sahoo MR et al. Int J Contemp Pediatr. 2015 Nov;2(4):433-439 http://www.ijpediatrics.com pissn 2349-3283 eissn 2349-3291 Research Article DOI: http://dx.doi.org/10.18203/2349-3291.ijcp20150990
International Journal of Contemporary Pediatrics Sahoo MR et al. Int J Contemp Pediatr. 2015 Nov;2(4):433-439 http://www.ijpediatrics.com pissn 2349-3283 eissn 2349-3291 Research Article DOI: http://dx.doi.org/10.18203/2349-3291.ijcp20150990
Case Report Long-Term Outcomes of Balloon Dilation for Acquired Subglottic Stenosis in Children
 Case Reports in Otolaryngology, Article ID 304593, 4 pages http://dx.doi.org/10.1155/2014/304593 Case Report Long-Term Outcomes of Balloon Dilation for Acquired Subglottic Stenosis in Children Aliye Filiz
Case Reports in Otolaryngology, Article ID 304593, 4 pages http://dx.doi.org/10.1155/2014/304593 Case Report Long-Term Outcomes of Balloon Dilation for Acquired Subglottic Stenosis in Children Aliye Filiz
Available online at ScienceDirect. journal homepage:
 Pediatrics and Neonatology (2016) 57, 19e26 Available online at www.sciencedirect.com ScienceDirect journal homepage: http://www.pediatr-neonatol.com ORIGINAL ARTICLE Bovine Surfactant Replacement Therapy
Pediatrics and Neonatology (2016) 57, 19e26 Available online at www.sciencedirect.com ScienceDirect journal homepage: http://www.pediatr-neonatol.com ORIGINAL ARTICLE Bovine Surfactant Replacement Therapy
Dr. AM MAALIM KPA 2018
 Dr. AM MAALIM KPA 2018 Journey Towards Lung protection Goals of lung protection Strategies Summary Conclusion Before 1960: Oxygen; impact assessed clinically. The 1960s:President JFK, Ventilators mortality;
Dr. AM MAALIM KPA 2018 Journey Towards Lung protection Goals of lung protection Strategies Summary Conclusion Before 1960: Oxygen; impact assessed clinically. The 1960s:President JFK, Ventilators mortality;
The Effectiveness of Surfactant Replacement Therapy for Preterm Infants with Respiratory Dis~ress Syndrome
 The Effectiveness of Surfactant Replacement Therapy for Preterm Infants with Respiratory Dis~ress Syndrome W L Lim, MRCP, C T Lim, FRCP, J K Chye, FRACP, Department of Paediatrics, U nive.rsity of Malaya,
The Effectiveness of Surfactant Replacement Therapy for Preterm Infants with Respiratory Dis~ress Syndrome W L Lim, MRCP, C T Lim, FRCP, J K Chye, FRACP, Department of Paediatrics, U nive.rsity of Malaya,
Neonatal Resuscitation Using a Nasal Cannula: A Single-Center Experience
 Original Article Neonatal Resuscitation Using a Nasal Cannula: A Single-Center Experience Pedro Paz, MD, MPH 1 Rangasamy Ramanathan, MD 1,2 Richard Hernandez, RCP 2 Manoj Biniwale, MD 1 1 Division of Neonatal
Original Article Neonatal Resuscitation Using a Nasal Cannula: A Single-Center Experience Pedro Paz, MD, MPH 1 Rangasamy Ramanathan, MD 1,2 Richard Hernandez, RCP 2 Manoj Biniwale, MD 1 1 Division of Neonatal
By: Armend Lokku Supervisor: Dr. Lucia Mirea. Maternal-Infant Care Research Center, Mount Sinai Hospital
 By: Armend Lokku Supervisor: Dr. Lucia Mirea Maternal-Infant Care Research Center, Mount Sinai Hospital Background My practicum placement was at the Maternal-Infant Care Research Center (MiCare) at Mount
By: Armend Lokku Supervisor: Dr. Lucia Mirea Maternal-Infant Care Research Center, Mount Sinai Hospital Background My practicum placement was at the Maternal-Infant Care Research Center (MiCare) at Mount
HFOV IN THE NON-RECRUITABLE LUNG
 HFOV IN THE NON-RECRUITABLE LUNG HFOV IN THE NON-RECRUITABLE LUNG PPHN Pulmonary hypoplasia after PPROM Congenital diaphragmatic hernia Pulmonary interstitial emphysema / cystic lung disease 1 30 Mean
HFOV IN THE NON-RECRUITABLE LUNG HFOV IN THE NON-RECRUITABLE LUNG PPHN Pulmonary hypoplasia after PPROM Congenital diaphragmatic hernia Pulmonary interstitial emphysema / cystic lung disease 1 30 Mean
