A Harmonized Segmentation Protocol for Hippocampal and Parahippocampal Subregions: Why Do We Need One and What Are the Key Goals?
|
|
|
- Randell Dorsey
- 6 years ago
- Views:
Transcription
1 HIPPOCAMPUS 27:3 11 (2017) COMMENTARY A Harmonized Segmentation Protocol for Hippocampal and Parahippocampal Subregions: Why Do We Need One and What Are the Key Goals? Laura E.M. Wisse, 1 * Ana M. Daugherty, 2 Rosanna K. Olsen, 3 David Berron, 4 Valerie A. Carr, 5,6 Craig E.L. Stark, 7 Robert S.C. Amaral, 8,9,10 Katrin Amunts, 11,12,13 Jean C. Augustinack, 14 Andrew R. Bender, 15 Jeffrey D. Bernstein, 16 Marina Boccardi, 17 Martina Bocchetta, 18 Alison Burggren, 19 M. Mallar Chakravarty, 8,9 Marie Chupin, 20 Arne Ekstrom, 21,22 Robin de Flores, 23 Ricardo Insausti, 24 Prabesh Kanel, 25 Olga Kedo, 11 Kristen M. Kennedy, 26 Geoffrey A. Kerchner, 16 Karen F. LaRocque, 5 Xiuwen Liu, 25 Anne Maass, 27 Nicolai Malykhin, 28,29,30 Susanne G. Mueller, 31,32 Noa Ofen, 33,34 Daniela J. Palombo, 35 Mansi B. Parekh, 36 John B. Pluta, 1 Jens C. Pruessner, 37,38 Naftali Raz, 33,34 Karen M. Rodrigue, 26 Dorothee Schoemaker, 37,38 Andrea T. Shafer, 33,34 Trevor A. Steve, 39 Nanthia Suthana, 19,40 Lei Wang, 41 Julie L. Winterburn, 8,9 Michael A. Yassa, 7,42 Paul A. Yushkevich, 1 Renaud la Joie, 23 for the Hippocampal Subfields Group ABSTRACT: The advent of high-resolution magnetic resonance imaging (MRI) has enabled in vivo research in a variety of populations and diseases on the structure and function of hippocampal subfields and subdivisions of the parahippocampal gyrus. Because of the many extant and highly discrepant segmentation protocols, comparing results across studies is difficult. To overcome this barrier, the Hippocampal Subfields Group was formed as an international collaboration with the aim of developing a 1 Penn Image Computing and Science Laboratory, Department of Radiology, University of Pennsylvania, Philadelphia; 2 Beckman Institute for Advanced Science and Technology, University of Illinois Urbana-Champaign, Champaign, Illinois; 3 Baycrest Health Sciences, Rotman Research Institute, Toronto, Ontario, Canada; 4 Institute of Cognitive Neurology and Dementia Research, Otto-von-Guericke University, Magdeburg, Germany; 5 Department of Psychology, Stanford University, Palo Alto, California; 6 Department of Psychology, San Jose State University, San Jose, California; 7 Department of Neurobiology and Behavior, University of California Irvine, Irvine,, California; 8 Cerebral Imaging Centre, Douglas Mental Health University Institute, McGill University, Montreal, Quebec, Canada; 9 Departments of Psychiatry and Biological and Biomedical Engineering, McGill University, Montreal, Quebec, Canada; 10 Integrated Program in Neuroscience McGill University, Montreal, Quebec, Canada; 11 Institute of Neuroscience and Medicine, INM-1, Research Center J ulich, J ulich, Germany; 12 JARA-BRAIN J ulich-aachen Research Alliance, J ulich, Germany; 13 C. and O. Vogt Institute for Brain Research, Heinrich Heine University D usseldorf, D usseldorf, Germany; 14 Department of Radiology, AA Martinos Center for Biomedical Imaging, Harvard Medical School, Massachusetts General Hospital, Boston, Massachusetts; 15 Center for Lifespan Psychology, Max Planck Institute for Human Development, Berlin, Germany; 16 Department of Neurology and Neurological Sciences, Stanford University School of Medicine, Palo Alto, California; 17 LANVIE Laboratory of Neuroimaging of Aging, University of Geneva, Geneva, Switzerland; 18 Dementia Research Centre, Department of Neurodegenerative Disease, UCL Institute of Neurology, Queen Square, London, United Kingdom; 19 Department of Psychiatry and Biobehavioural Sciences, University of California Los Angeles, Los Angeles, California; 20 INSERM, CNRS, UMR-S975, Institut du Cerveau et de la Moelle Epinière (ICM), Paris, France; 21 Center for Neuroscience, University of California Davis, Davis, California; 22 Department of Psychology, University of California Davis, Davis, California; 23 INSERM U1077, Universite de Caen Normandie, UMR-S1077, Ecole Pratique des Hautes Etudes, Centre Hospitalier Universitaire de Caen, Caen, France; 24 Human Neuroanatomy Laboratory and C.R.I.B School of Medicine, University of Castilla-La Mancha, Albacete, Spain; 25 Department of Computer Science, Florida State University, Tallahassee, Florida; 26 School of Behavioral and Brain Sciences, Center for Vital Longevity, University of Texas at Dallas, Dallas, Texas; 27 School of Public Health and Helen Wills Neuroscience Institute, University of California Berkeley, Berkeley, California; 28 Department of Biomedical Engineering, University of Alberta, Edmonton, Canada; 29 The Neuroscience and Mental Health Institute, University of Alberta, Edmonton, Canada; 30 Department of Psychiatry, University of Alberta, Edmonton, Canada; 31 Department of Radiology, University of California, San Francisco, California; 32 Center for Imaging of Neurodegenerative Diseases, San Francisco VA Medical Center, San Francisco, California; 33 Psychology Department, Wayne State University, Detroit, Michigan; 34 Institute of Gerontology, Wayne State University, Detroit, Michigan; 35 VA Boston Healthcare System, Boston, Massachusetts; 36 Department of Radiology, Stanford University, Palo Alto, California; 37 Faculty of Medicine, McGill Centre for Studies in Aging, McGill University, Montreal, Quebec, Canada; 38 Department of Psychology, McGill University, Montreal, Canada; 39 Division of Neurology, Department of Medicine, University of Alberta, Edmonton, Alberta, Canada; 40 Department of Neurosurgery, University of California, Los Angeles, California; 41 Feinberg School of Medicine, Northwestern University, Chicago, Michigan; 42 Department of Neurology, University of California, Irvine, California VC 2016 WILEY PERIODICALS, INC.
2 4 WISSE ET AL. harmonized protocol for manual segmentation of hippocampal and parahippocampal subregions on high-resolution MRI. In this commentary we discuss the goals for this protocol and the associated key challenges involved in its development. These include differences among existing anatomical reference materials, striking the right balance between reliability of measurements and anatomical validity, and the development of a versatile protocol that can be adopted for the study of populations varying in age and health. The commentary outlines these key challenges, as well as the proposed solution of each, with concrete examples from our working plan. Finally, with two examples, we illustrate how the harmonized protocol, once completed, is expected to impact the field by producing measurements that are quantitatively comparable across labs and by facilitating the synthesis of findings across different studies. VC 2016 Wiley Periodicals, Inc. KEY WORDS: hippocampus; segmentation; harmonization; MRI; parahippocampal gyrus INTRODUCTION The medial temporal lobe (MTL) in general, and the hippocampus in particular, have attracted extensive interest in in vivo human studies of a wide range of diseases (Small et al., 2011), normal life-span development (Daugherty et al., 2016) and cognitive abilities (Squire et al., 2004;Van Petten, 2004; Carr et al., 2010; Kyle et al., 2015a) involving structural (Mueller et al., 2008) as well as functional magnetic resonance imaging (MRI) (Yassa et al., 2010). The MTL includes the hippocampal formation and several cortical regions within the parahippocampal gyrus, namely the entorhinal, perirhinal, and parahippocampal cortices. The hippocampal formation itself is a complex structure that is composed of distinct subfields the subicular complex (including prosubiculum, subiculum proper, presubiculum and parasubiculum), Cornu ammonis (CA1- CA4), and the dentate gyrus (DG) 1 (Duvernoy et al., 2005; 1 Note that the terminology for MTL structures differs per neuroanatomy laboratory. For example, some neuroanatomists prefer the term CA4 (Duvernoy et al., 2005), whereas others refer to this region as hilus (West and Gundersen, 1990) or part of CA3 (Insausti and Amaral, 2012). Insausti and Amaral, 2012). In the early 2000 s, in vivo measurement of hippocampal subfields first became possible through advancements in neuroimaging and analysis techniques (Small et al., 1999, 2000; Zeineh et al., 2000, 2001). Since then, over 20 manual protocols (Insausti et al., 1998; Small et al., 1999; Zeineh et al., 2001; Kirwan et al., 2007; Mueller et al., 2007; Ekstrom et al., 2009; Olsen et al., 2009; Kerchner et al., 2010; La Joie et al., 2010; Malykhin et al., 2010; Preston et al., 2010; Yassa et al., 2010; Bonnici et al., 2012; Libby et al., 2012; Wisse et al., 2012; Bender et al., 2013; Palombo et al., 2013; Winterburn et al., 2013; Suthana et al., 2015); see also (Yushkevich et al., 2015a) and several automatic procedures (Fischl et al., 2009; Van Leemput et al., 2009; Yushkevich et al., 2010; Augustinack et al., 2013; Pipitone et al., 2014; Iglesias et al., 2015; Yushkevich et al., 2015b) have been developed for in vivo segmentation on MR images of hippocampal and parahippocampal subregions. These manual and automatic segmentation protocols are highly discrepant from one another and often employ different terminology and definitions of the regional boundaries (Yushkevich et al., 2015a). This variability in segmentation protocols produces widely inconsistent results, even when studying similar populations and phenomena (de Flores et al., 2015a; Wisse et al., 2015). This variability complicates meaningful comparison of results between studies (de Flores et al., 2015a) and hampers the ability to draw broader theoretical conclusions. Thus, there is a strong need for a harmonized protocol that can be employed consistently across laboratories. Because of the wide variability in segmentation protocols and the lack of consensus in the field, no single protocol can be readily adopted as a common standard. Instead, it is necessary to develop a new manual segmentation protocol via consensus from the scientific community that resolves the various discrepancies and consolidates the commonalities across procedures and neuroanatomical reference sources. Although automated methods are appealing for efficiency and ease of adoption across research groups, the current automated protocols also differ widely from one another (Yushkevich et al., 2015a) and, when pitted against the gold standard of manual segmentation protocols, show limited Additional Supporting Information may be found in the online version of this article. Grant sponsor: NIH; Grant number: R ; Grant sponsor: NIH/NINDS; Grant number: NS076856, NS093052; Grant sponsor: Eisai and the Canadian League Against Epilepsy (CLAE); Grant sponsor: NIDA; Grant number: K01 DA034728; Grant sponsor: NIH/NIA; Grant number: R00 AG ; Grant sponsor: Alzheimer Association; Grant number: ADNI ; Grant number: GI ; Grant sponsor: US NIA; Grant number: P50 AG05146; Grant sponsor: NIA; Grant number: R21 AG049220; Grant sponsor: NIMH; Grant number: R01 MH102392; Grant sponsor: National Sciences and Engineering Research Council Canada, Canadian Institutes of Health Research, Weston Brian Institute, Alzheimer s Association, Michael J. Fox Foundation for Parkinson s Research, and Fonds de recherche saute Quebec; Grant sponsor: NIA; Grant number: R00 AG-36848; Grant sponsor: NIA; Grant number: R01 AG037376; Grant sponsor: NIBIB; Grant number: R01 EB017255; Grant sponsor: Fondation Therèse et Rene Planiol pour l etude du Cerveau. *Correspondence to: Laura E.M. Wisse, Richards Building 6th Floor, 3700 Hamilton Walk, PA Philadelphia. laura. wisse@uphs.upenn.edu L.E.M.W. and A.M.D. contributed equally to this work. Received 6 May 2016; Revised 6 October 2016; Accepted for publication 17 October DOI /hipo Published online 24 October 2016 in Wiley Online Library (wileyonlinelibrary.com).
3 A HARMONIZED HIPPOCAMPAL SUBFIELD PROTOCOL: KEY GOALS AND IMPACT 5 FIGURE 1. Comparison of 21 segmentation protocols in a coronal section in the hippocampal body of one subject. Figure from Yushkevich et al., NeuroImage, 2015a; reprinted with permission from Elsevier. Alv, Alveus; CA, cornu ammonis; CSF, cerebrospinal fluid; DG, dentate gyrus; DG:H, dentate gyrus Hilar region; Fim, fimbria; GCL, granular cell layer; H, hippocampus; Para, parasubiculum; PHC, parahippocampal cortex; Pre, presubiculum; Sub, subiculum; SP, stratum pyramidale; SRLM, stratum radiatum lacunosum moleculare. [Color figure can be viewed at wileyonlinelibrary.com] concurrent validity, especially for smaller subfields. Automation of subfield segmentation therefore remains a hopeful future goal, but the first necessary step in its development is to establish a harmonized protocol for manual segmentation. Inspired by the European Alzheimer s Disease Consortium (EADC) Alzheimer s Disease Neuroimaging Initiative (ADNI) Harmonized Protocol (HarP) project (Boccardi et al., 2015; Frisoni et al., 2015), which successfully developed a reliable harmonized segmentation protocol for labeling the whole hippocampal formation as a single structure on 1 mm 3 T1-weighted images, the Hippocampal Subfields Group (HSG) was formed in 2013 with the aim of developing a harmonized protocol for the segmentation of hippocampal subfields and the adjacent entorhinal, perirhinal, and parahippocampal cortices (Kivisaari et al., 2013) for high-resolution T2-weighted 3 and 7 tesla (T) MRI data. T2-weighted imaging is the most commonly used for subfield segmentation because the stratum moleculare laconosum radiatum that is visualized as a dark band is used to separate the CA and the DG; this band is less visible on T1-weighted images. As one of the first steps towards harmonization, 21 segmentation protocols for hippocampal and parahippocampal subregions were compared directly and revealed a range of similarities and differences between protocols (Yushkevich et al., 2015a). The greatest disagreement between protocols in the hippocampal body, for example, was in the demarcation of the CA1-subicular complex boundary (Fig. 1). This comparison underscores the urgent need for a harmonized protocol. Since publication of this comparison paper in 2015, the HSG has developed a working plan for creating a harmonized protocol for high-resolution T2-weighted MRIs. In short, it includes: (1) localizing subfield boundaries on histology in a reference set of multiple ex vivo specimens, (2) deriving specific rules for placing boundaries on in vivo MRI using this histological reference material, (3) sharing the protocol with the larger community to solicit feedback, and (4) performing a formal reliability analysis on the agreed manual segmentation protocol. These steps are illustrated in Figure 2. This working plan reflects three key goals: attaining content (anatomical) validity, establishing measurement reliability, and enabling application to study normative and pathological changes across the lifespan. These key goals are
4 6 WISSE ET AL. FIGURE 2. Overview of the concrete steps involved in the harmonization project. In Step 1 in a set of three tissue samples, the boundaries on histology will be annotated by three anatomists. In Step 2 the boundaries on MRI will be derived based on the anatomical reference set in Step 1. These boundaries are divided into two sets: the outer boundaries with surrounding structures and the boundaries between the subfields. Note for Step 2 that for both protocols an initial reliability test will be performed and the protocols will be shared with the Boundary Working Group (BWG). In case the reliability criteria are not met or in case of considerable critique, the protocol will be adjusted. This will be an iterative process. In Step 3 feedback will be elicited from the larger Hippocampal Subfields Group (HSG). In Step 4 a formal reliability analysis will be performed by six raters from different labs. discussed below in the context of the unique challenges posed in this venture and the proposed working plan for achieving each goal. In addition, we elaborate on the expected impact of this harmonized protocol on the field. KEY GOALS AND CHALLENGES IN THE DEVELOPMENT OF A HARMONIZED SEGMENTATION PROTOCOL FOR HIPPOCAMPAL AND PARAHIPPOCAMPAL SUBREGIONS Content Validity: Agreement with Anatomy The harmonized protocol must achieve content validity for anatomy confirmed by various histological reference materials. Although extant segmentation protocols adhere to printed neuroanatomical atlases (Duvernoy et al., 2005; Mai et al., 2008; Insausti and Amaral, 2012), they largely rely on different reference materials (Yushkevich et al., 2015a), which may have, in part, contributed to the variability amongst protocols. In addition to simply providing different images and different anatomical definitions, most references depict a limited number of slices along the anterior posterior length of the hippocampus. Furthermore, the angulation of histology samples commonly differs from the orientation of MRI, thereby limiting their usefulness as the anchors of validation. Moreover, printed anatomical atlases often show only a limited number of specimens, making approximation of individual differences in anatomical landmarks and variability of subfield boundaries along the anterior posterior axis of the hippocampus difficult to assess. Approach: To resolve the outlined challenges, at least three neuroanatomy laboratories (e.g., KA, RI, and JCA) will provide a specimen, processed according to the standards in the specific labs, and all three labs will annotate each sample (Step 1) thus creating a variety of samples whilst allowing direct comparison of anatomical boundary demarcation between neuroanatomists. Notably, subfield boundary placement may differ between neuroanatomists, potentially arising from differences in processing methods or their own subfield definitions. While resolving these potential discrepancies is beyond the scope of our effort, we will use all available information and characterize the range in which the boundaries may fall to develop a protocol for MRI. Moreover, the use of different processing methods will broaden the applicability of the harmonized protocol. Critically, the samples will include multiple slices spanning the anterior posterior length and sectioning will be oriented similar to common neuroimaging protocols [i.e., perpendicular to the long axis of the hippocampus (Mueller et al., 2007; Yushkevich et al., 2015a)]. This reference set will be the most comprehensive dataset to date and will be a good starting point, used in addition to canonical atlases, to develop a protocol for in vivo MRI segmentation. Although unique and comprehensive, this atlas set only includes three samples because of practical constraints e.g., the labor intensive nature of annotating histological samples, the available time of the expert neuroanatomists and the availability of data either still intact or cut perpendicular to the long axis of the hippocampus, which is not a common neuroanatomical procedure. Therefore, existing histological references materials will also be used throughout the process and the neuroanatomists and MRI groups will continue to consult throughout the protocol development, particularly as it relates to individual differences in anatomical features. Optimizing Measurement Reliability While Maintaining Anatomical Validity As important as the content validity vis-a-vis the anatomy is, the harmonized protocol must also produce reliable volumetric measures. High inter-rater reliability is particularly essential for this protocol as it is meant to be adopted and applied
5 A HARMONIZED HIPPOCAMPAL SUBFIELD PROTOCOL: KEY GOALS AND IMPACT 7 uniformly across groups. Because many of the features used to determine hippocampal subfield boundaries ex vivo (e.g., cell shape, size, or density) cannot be visualized on high-resolution MRI, attempts to directly replicate anatomical boundaries commonly result in unreliable measures. An example is the endfolial pathway (Lim et al., 1997) in the stratum oriens of CA3, composed of loosely packed cells, which forms the inferior border of the CA3 curving into the DG. Although this border has been reported to be discernable on 7T MRI (Parekh et al., 2015), this border cannot be consistently observed on the typically-used lower-resolution 3T scans and might therefore lead to unreliable measurements. The use of a geometrical rule for this border on 3T data may therefore be necessary. Given the limitations of MRI resolution and quality, some small compromises in neuroanatomical precision are to be expected as boundaries must be reliably reproduced. Thus, the HSG endeavors to create a protocol that strives to maximize the approximation of the MTL anatomy while affording high reliability of its measurement. Approach: Toward this end, we will develop a manual segmentation protocol for T2-weighted MRI based on the comprehensive anatomical reference set, while testing reliability at multiple stages. In Step 2, we will derive heuristic rules from the anatomical reference set and, via comparison to MRI, determine landmarks available in the vast majority of in vivo T2-weighted MR images that are commonly used for hippocampal subfield segmentation. The draft protocol will be tested for reliability and may be subsequently retested until sufficient reliability is attained (Fig. 2), based upon criteria reported in the literature and if needed, by majority voting. Additionally, following critical feedback from the larger community (Step 3), we will perform a formal reliability analysis of the final protocol in Step 4 with six raters drawn from across laboratories, which will ensure that the protocol can be adopted by different research groups. Broad Application of the Harmonized Protocol A final key goal is to develop a harmonized protocol that can be adopted widely for the study of populations across the lifespan and across several diseases. There are several practical challenges to achieving this goal: differences in scanning platform and acquisition protocols, variable scan quality (e.g., common motion artifacts in MRI scans of children or patients with Alzheimer s disease), segmentation goals of a particular study (e.g., the number of hippocampal subfields or cortical regions of interest) and potential differences in the hippocampal subfield boundary locations due to alterations in the internal composition of the hippocampus in certain populations. Approach: We have incorporated a number of solutions in our working plan to ensure that the harmonized protocol meets the requirements and needs of different research groups. First, to allow for the highest level of adoption across research groups, we aim for the harmonized protocol to include definitions to separately segment the following subfields: subicular complex, CA1, CA2, CA3, and DG (including Fascia dentata and CA4), and the parahippocampal, perirhinal, and entorhinal cortices. Such delineation will be contingent upon the achievement of high reliability and validity for each region. The protocol is intended to remain flexible for the user to decide, which subfields to potentially collapse into a single measurement (e.g., the common inclusion of CA3 with the DG) depending on the particular research goals. Of note, we deliberately limit our protocol to the selected subfields in an effort to maintain high reliability of all measures, as the amount of detail on in vivo MR images is insufficient, in our opinion, to segment additional structures at 3T. Developing a protocol for 3T data is the first focus of the group as it is most common in the field. Upon completion of the 3T harmonized protocol, it will be adjusted for other applications, such as 7T structural images, which may involve adding more fine-grained demarcations to the 3T protocol. Second, we will develop and test our protocol in two different datasets consisting of scans from different research groups and reflecting the different ages and diseases most commonly studied in relation to these MTL regions. More specifically, it will include the age range from 7 to 100 years, and scans from healthy subjects as well as patients with hypertension, epilepsy and Alzheimer s disease. Importantly, these images also reflect the different types of 3T scanner manufacturers and sequences used in the field, as well as commonly observed imaging artifacts. Relatedly, by using anatomical features that are observable on these MRI scans and rules that can fit hippocampi with varying shapes, the protocol will be able to accommodate variability in hippocampal morphometry that occurs in development and diseases such as epilepsy and Alzheimer s disease, as has been shown to be feasible by previous segmentation work in these populations (Mueller et al., 2010; Daugherty et al., 2016; Santyr et al., 2016). However, our MRI dataset does not include all possible MTL abnormalities that can occur due to injury or disease [e.g., encephalitis or prenatal/postnatal abnormalities that may affect the folding of the hippocampus (Rosenbaum et al., 2014)]. We will therefore additionally leverage the collective experience of the large research community contributing to this effort, including members who have firsthand experience with segmentation in special cases of MTL abnormalities. Additionally, it should be noted that we currently only have access to histological material from older adults, but not to histological material from younger age groups or some of the diseases of interest in our effort. This may limit our ability to confirm anatomical validity of our protocol when applied to various pathological conditions that may plausibly alter the internal composition and shape of the hippocampus. When evaluating the validity of the protocol, we will therefore complement our histological dataset of healthy brains with histological reference material from pathology (Insausti et al., 2010) whenever feasible. In the application to the study of development, we have a reasonable expectation that a similar protocol can be applied to samples of children and adults. Although hippocampal structure and morphometry continue to develop into adolescence (Insausti et al., 2010), the cytoarchitectonic differences that define the subfields as observed in the
6 8 WISSE ET AL. adult brain can be seen as early as mid-gestation (Humphrey, 1967;Arnold and Trojanowski, 1996) and are almost adult-like from the first postnatal year on (Insausti et al., 2010). Additionally, some cells in the hippocampal formation take on adult-like qualities between the 2nd and the 8th year (Seress et al., 2001; Seress, 2007). However, in the future, if more histological datasets become available with sectioning perpendicular to the long axis of the hippocampus, this may allow the validation of this protocol in younger age groups or certain disease populations and potentially necessitate updating the protocol. Third, the HSG is a large, international collaborative group (150 members from more than 15 countries) that represents all levels of expertize and experience with different types of data, and different research interests. The HSG community will be asked for input in two stages. In Step 2, the boundary working group members (30) that are not involved in developing the in vivo definitions will be asked for initial feedback. In Step 3 feedback will be solicited from the larger community via an on-line questionnaire, similar to the Delphi procedure used in the HarP project (Boccardi et al., 2015). To further increase the versatility and expertize of the HSG, the authors invite other researchers to join this open effort and to provide input ( IMPACT OF A HARMONIZED SEGMENTATION PROTOCOL FOR HIPPOCAMPAL AND PARAHIPPOCAMPAL SUBREGIONS We expect that this harmonized protocol will have a significant impact in the research community as its use will enable direct comparison of results between studies, thereby better accommodating the replication of results and the pooling of data for meta-analyses. Existing data could potentially be reanalyzed with the harmonized protocol and we encourage researchers to consider this approach as an avenue for reconciling current discrepancies in the literature. Additionally, capitalizing on a uniform segmentation protocol adopted across research groups, the field can gain further insight into lifespan developmental trajectories and related diseases on hippocampal and parahippocampal structure and function. We provide two compelling examples of this in the study of aging and of pattern separation and completion for which the harmonized protocol could specifically be of added value. Example 1: Age-related hippocampal subfield atrophy Characterizing effects of advanced age on hippocampal subfield volumes is of great importance for understanding typical cognitive decline and departure from normal trajectories in the course of disease. Unfortunately, so far results pertaining to the association of age with hippocampal subfield volumes are inconclusive, with studies finding an effect of ageing on virtually all possible combinations of subfields (Mueller and Weiner, 2009; Shing et al., 2011; Pereira et al., 2014; Wisse et al., 2014; de Flores et al., 2015b; Daugherty et al., 2016), for a review see (de Flores et al., 2015a). These discrepancies most likely reflect, at least in part, differences between segmentation protocols. For example, the seemingly disparate findings of age differences in CA1 or subicular volume, might in fact pertain to the same region as these labels often overlap between segmentation protocols. Hypothetically, new and existing data could be analyzed with the harmonized protocol and not only accommodate direct comparisons of age effects, but also evaluate differences in population characteristics and health [e.g., age-related decline in cardiovascular health (Shing et al., 2011; Bender et al., 2013)] that might account for variability among studies. In addition, a harmonized protocol will enable the synthesis and integration of results across studies that have included different age groups, thus allowing for a characterization of hippocampal volume across the lifespan without a single lab having to collect data from each age group. Example 2: Pattern separation and completion High-resolution fmri studies of hippocampal subfields aim to elucidate their functional role across various cognitive tasks. Two important mechanisms thought to be involved in human memory and spatial navigation are pattern separation and pattern completion (Marr, 1971; McClelland et al., 1995; Yassa and Stark, 2011). Whereas it is proposed that pattern separation plays an important role whenever similar memories have to be encoded in a distinct fashion, pattern completion is critical for recalling episodes based on partial or degraded cues (Yassa et al., 2010; Hunsaker and Kesner, 2013). Anatomical evidence from nonhuman animal work, suggests that pattern separation relies on the DG (Leutgeb et al., 2007), while pattern completion appears to be associated with a network of recurrent collaterals in CA3 (Neunuebel and Knierim, 2014). So far, most human neuroimaging studies of pattern separation and completion mechanisms combine CA2, CA3, and DG into one region, due to limited spatial resolution currently available at 3T (Bakker et al., 2008; Yassa et al., 2010; Dudukovic et al., 2011; Kyle et al., 2015b; Stokes et al., 2015). With new technological advancements at 3T and increasing availability of high-resolution 7T imaging (Feinberg and Yacoub, 2012; Todd et al., 2016), it will become increasingly feasible to functionally separate these subregions. However, most extant segmentation protocols propose different locations of the CA3 boundaries and as a result variable numbers of CA3 voxels are counted towards DG (Wisse et al., 2012; Winterburn et al., 2013; Iglesias et al., 2015; Yushkevich et al., 2015b). This results in high anatomical variability in DG and CA3 masks among protocols. A harmonized protocol will therefore be critical to better understand the specific functional roles of DG and CA3 in pattern separation and completion using human in vivo imaging. SUMMARY In summary, we are proposing to develop a harmonized segmentation protocol for in vivo T2-weighted MR images for
7 A HARMONIZED HIPPOCAMPAL SUBFIELD PROTOCOL: KEY GOALS AND IMPACT 9 hippocampal and parahippocampal subregions that maximally represent the underlying MTL anatomy while affording high reliability of its measurement and that can be applied in different study populations and on scans acquired in different research groups with different scanners. The development of this harmonized protocol for the currently available 3T and 7T data is crucial in the immediate future as the heterogeneity in protocols is greatly hampering progress of research related to these MTL regions. We believe the current plan will allow us to achieve our main aim of harmonization while also attaining acceptable anatomical validity. However, in the future, the protocol may be updated based upon advances in imaging and analysis techniques, as well as more comprehensive histology reference sets from different populations as they become available via ongoing research endeavors. Additionally, in the future, it would be helpful to obtain a common understanding of the parcellation at the microscopic scale, in which discrepancies in labeling are also an issue, e.g., with CA4 part of some histological segmentations but not others (Duvernoy et al., 2005; Insausti and Amaral, 2012). We have started our effort by developing a manual segmentation protocol of the hippocampal subfields within the hippocampal body for T2-weighted 3T images because the body is included in all existing segmentation protocols (Yushkevich et al., 2015a) and is more uniform than the head and tail regions of the hippocampus. We plan to extend the protocol to the head and tail portions. We aim to finalize and publish the harmonized protocol for the hippocampal body in the near future to allow for its more immediate adoption while continuing the efforts for procedures in the remainder of the hippocampus, as well as the adjacent cortical regions. To facilitate wide adoption of the harmonized protocol by new users, we plan to provide training resources, for example an instructional video or an example dataset, alongside the segmentation protocol. Additionally, we plan to implement it into (semi-)automated segmentation algorithms. All extant (semi- )automated segmentation procedures are atlas-based and can be updated with the harmonized segmentation protocol. This step will therefore follow after the initial development of a manual harmonized segmentation protocol. When finished, this manual protocol will be made available to the research groups involved in the development of automated subfield segmentation algorithms (Pipitone et al., 2014; Iglesias et al., 2015; Yushkevich et al., 2015b), several of whom are part of this collaborative effort. ACKNOWLEDGMENTS The authors thank the members of the larger Hippocampal Subfields Group for their input on the harmonization effort and insightful comments over the years. They acknowledge the work of Ms. Mercedes Iniguez de Onzono for magnificent technical work. None of the authors has any disclosures, except GAK, who is an employee of Genentech, Inc. See Supplementary Material for a list of collaborators in the Hippocampal Subfields Groups. REFERENCES Arnold SE, Trojanowski JQ Human fetal hippocampal development: I. Cytoarchitecture, myeloarchitecture, and neuronal morphologic features. J Comp Neurol 367: Augustinack JC, Huber KE, Stevens AA, Roy M, Frosch MP, van der Kouwe AJ, Wald LL, Van Leemput K, McKee AC, Fischl B, Alzheimer s Disease Neuroimaging Initiative Predicting the location of human perirhinal cortex, Brodmann s area 35, from MRI. Neuroimage 64: Bakker A, Kirwan CB, Miller M, Stark CE Pattern separation in the human hippocampal CA3 and dentate gyrus. Science 319: Bender AR, Daugherty AM, Raz N Vascular risk moderates associations between hippocampal subfield volumes and memory. J Cogn Neurosci 25: Boccardi M, Bocchetta M, Apostolova LG, Barnes J, Bartzokis G, Corbetta G, DeCarli C, detoledo-morrell L, Firbank M, Ganzola R, Gerritsen L, Henneman W, Killiany RJ, Malykhin N, Pasqualetti P, Pruessner JC, Redolfi A, Robitaille N, Soininen H, Tolomeo D, Wang L, Watson C, Wolf H, Duvernoy H, Duchesne S, Jack CR Jr, Frisoni GB, EADC-ADNI Working Group on the Harmonized Protocol for Manual Hippocampal Segmentation Delphi definition of the EADC-ADNI harmonized protocol for hippocampal segmentation on magnetic resonance. Alzheimers Dement 11: Bonnici HM, Chadwick MJ, Kumaran D, Hassabis D, Weiskopf N, Maguire EA Multi-voxel pattern analysis in human hippocampal subfields. Front Hum Neurosci 6:290. Carr VA, Rissman J, Wagner AD Imaging the human medial temporal lobe with high-resolution fmri. Neuron 65: Daugherty AM, Bender AR, Raz N, Ofen N Age differences in hippocampal subfield volumes from childhood to late adulthood. 26: de Flores R, La Joie R, Chetelat G. 2015a. Structural imaging of hippocampal subfields in healthy aging and Alzheimer s disease. Neuroscience 309: de Flores R, La Joie R, Landeau B, Perrotin A, Mezenge F, de La Sayette V, Eustache F, Desgranges B, Chetelat G. 2015b. Effects of age and Alzheimer s disease on hippocampal subfields: Comparison between manual and FreeSurfer volumetry. Hum Brain Mapp 36: Dudukovic NM, Preston AR, Archie JJ, Glover GH, Wagner AD High-resolution fmri reveals match enhancement and attentional modulation in the human medial temporal lobe. J Cogn Neurosci 23: Duvernoy HM, Cattin E, Naidich T, Fatterpekar GM, Raybaud C, Risold PY, Sakvolini U, Scarabino T The human hippocampus. Berlin, Heidelberg, Germany: Springer Verlag. p Ekstrom AD, Bazih AJ, Suthana NA, Al-Hakim R, Ogura K, Zeineh M, Burggren AC, Bookheimer SY Advances in highresolution imaging and computational unfolding of the human hippocampus. Neuroimage 47: Feinberg DA, Yacoub E The rapid development of high speed, resolution and precision in fmri. Neuroimage 62: Fischl B, Stevens AA, Rajendran N, Yeo BT, Greve DN, Van Leemput K, Polimeni JR, Kakunoori S, Buckner RL, Pacheco J, Salat DH, Melcher J, Frosch MP, Hyman BT, Grant PE, Rosen BR, van der Kouwe AJ, Wiggins GC, Wald LL, Augustinack JC Predicting the location of entorhinal cortex from MRI. Neuroimage 47: Frisoni GB, Jack CR, Jr, Bocchetta M, Bauer C, Frederiksen KS, Liu Y, Preboske G, Swihart T, Blair M, Cavedo E, Grothe MJ, Lanfredi M, Martinez O, Nishikawa M, Portegies M, Stoub T,
8 10 WISSE ET AL. Ward C, Apostolova LG, Ganzola R, Wolf D, Barkhof F, Bartzokis G, DeCarli C, Csernansky JG, detoledo-morrell L, Geerlings MI, Kaye J, Killiany RJ, Lehericy S, Matsuda H, O Brien J, Silbert LC, Scheltens P, Soininen H, Teipel S, Waldemar G, Fellgiebel A, Barnes J, Firbank M, Gerritsen L, Henneman W, Malykhin N, Pruessner JC, Wang L, Watson C, Wolf H, deleon M, Pantel J, Ferrari C, Bosco P, Pasqualetti P, Duchesne S, Duvernoy H, Boccardi M, EADC-ADNI Working Group on The Harmonized Protocol for Manual Hippocampal Volumetry of the Alzheimer s Disease Neuroimaging Initiative The EADC-ADNI harmonized protocol for manual hippocampal segmentation on magnetic resonance: Evidence of validity. Alzheimers Dement 11: Humphrey T The development of the human hippocampal fissure. J Anat 101: Hunsaker MR, Kesner RP The operation of pattern separation and pattern completion processes associated with different attributes or domains of memory. Neurosci Biobehav Rev 37: Iglesias JE, Augustinack JC, Nguyen K, Player CM, Player A, Wright M, Roy N, Frosch MP, McKee AC, Wald LL, Fischl B, Van Leemput K, Alzheimer s Disease Neuroimaging Initiative A computational atlas of the hippocampal formation using ex vivo, ultra-high resolution MRI: Application to adaptive segmentation of in vivo MRI. Neuroimage 115: Insausti R, Cebaza-Sanchez S, Marcos P Postnatal Developmentof the Human Hippocampal Formation. Frankfurt: Springer. Insausti R, Amaral DG Hippocampal Formation. In: Mai JK, Paxinos G, editors. The Human Nervous System. San Diego: Elsevier Academic Press. Insausti R, Juottonen K, Soininen H, Insausti AM, Partanen K, Vainio P, Laakso MP, Pitkanen A MR volumetric analysis of the human entorhinal, perirhinal, and temporopolar cortices. AJNR Am J Neuroradiol 19: Kerchner GA, Hess CP, Hammond-Rosenbluth KE, Xu D, Rabinovici GD, Kelley DA, Vigneron DB, Nelson SJ, Miller BL Hippocampal CA1 apical neuropil atrophy in mild Alzheimer disease visualized with 7-T MRI. Neurology 75: Kirwan CB, Jones CK, Miller MI, Stark CE High-resolution fmri investigation of the medial temporal lobe. Hum Brain Mapp 28: Kivisaari SL, Probst A, Taylor KI The perirhinal, entorhinal, and parahippocampal cortices and hippocampus: An overview of functional anatomy and protocol for their segmentation in MR images. In: Ulmer S, Jansen O, editors. fmri - Basics and Clinical Applications. Berlin Heidelberg: Springer-Verlag. p 239. Kyle CT, Smuda DN, Hassan AS, Ekstrom AD. 2015a. Roles of human hippocampal subfields in retrieval of spatial and temporal context. Behav Brain Res 278: Kyle CT, Stokes JD, Lieberman JS, Hassan AS, Ekstrom AD. 2015b. Successful retrieval of competing spatial environments in humans involves hippocampal pattern separation mechanisms. Elife 4: /eLife La Joie R, Fouquet M, Mezenge F, Landeau B, Villain N, Mevel K, Pelerin A, Eustache F, Desgranges B, Chetelat G Differential effect of age on hippocampal subfields assessed using a new high-resolution 3T MR sequence. Neuroimage 53: Leutgeb JK, Leutgeb S, Moser MB, Moser EI Pattern separation in the dentate gyrus and CA3 of the hippocampus. Science 315: Libby LA, Ekstrom AD, Ragland JD, Ranganath C Differential connectivity of perirhinal and parahippocampal cortices within human hippocampal subregions revealed by high-resolution functional imaging. J Neurosci 32: Lim C, Mufson EJ, Kordower JH, Blume HW, Madsen JR, Saper CB Connections of the hippocampal formation in humans: II. The endfolial fiber pathway. J Comp Neurol 385: Mai JK, Paxinos G, Voss T Atlas of the Human Brain. New York: Academic Press, Elsevier. pp Malykhin NV, Lebel RM, Coupland NJ, Wilman AH, Carter R In vivo quantification of hippocampal subfields using 4.7 T fast spin echo imaging. Neuroimage 49: Marr D Simple memory: A theory for archicortex. Philos Trans R Soc Lond B: Biol Sci 262: McClelland JL, McNaughton BL, O Reilly RC Why there are complementary learning systems in the hippocampus and neocortex: Insights from the successes and failures of connectionist models of learning and memory. Psychol Rev 102: Mueller SG, Weiner MW Selective effect of age, Apo e4, and Alzheimer s disease on hippocampal subfields. 19: Mueller SG, Schuff N, Raptentsetsang S, Elman J, Weiner MW Selective effect of Apo e4 on CA3 and dentate in normal aging and Alzheimer s disease using high resolution MRI at 4 T. Neuroimage 42: Mueller SG, Schuff N, Yaffe K, Madison C, Miller B, Weiner MW Hippocampal atrophy patterns in mild cognitive impairment and Alzheimer s disease. Hum Brain Mapp 31: Mueller SG, Stables L, Du AT, Schuff N, Truran D, Cashdollar N, Weiner MW Measurement of hippocampal subfields and age-related changes with high resolution MRI at 4T. Neurobiol Aging 28: Neunuebel JP, Knierim JJ CA3 retrieves coherent representations from degraded input: direct evidence for CA3 pattern completion and dentate gyrus pattern separation. Neuron 81: Olsen RK, Nichols EA, Chen J, Hunt JF, Glover GH, Gabrieli JD, Wagner AD Performance-related sustained and anticipatory activity in human medial temporal lobe during delayed match-tosample. J Neurosci 29: Palombo DJ, Amaral RS, Olsen RK, Muller DJ, Todd RM, Anderson AK, Levine B KIBRA polymorphism is associated with individual differences in hippocampal subregions: Evidence from anatomical segmentation using high-resolution MRI. J Neurosci 33: Parekh MB, Rutt BK, Purcell R, Chen Y, Zeineh MM Ultrahigh resolution in-vivo 7.0T structural imaging of the human hippocampus reveals the endfolial pathway. Neuroimage 112:1 6. Pereira JB, Valls-Pedret C, Ros E, Palacios E, Falcon C, Bargallo N, Bartres-Faz D, Wahlund LO, Westman E, Junque C Regional vulnerability of hippocampal subfields to aging measured by structural and diffusion MRI. 24: Pipitone J, Park MT, Winterburn J, Lett TA, Lerch JP, Pruessner JC, Lepage M, Voineskos AN, Chakravarty MM, Alzheimer s Disease Neuroimaging Initiative Multi-atlas segmentation of the whole hippocampus and subfields using multiple automatically generated templates. Neuroimage 101: Preston AR, Bornstein AM, Hutchinson JB, Gaare ME, Glover GH, Wagner AD High-resolution fmri of content-sensitive subsequent memory responses in human medial temporal lobe. J Cogn Neurosci 22: Rosenbaum RS, Gao F, Honjo K, Raybaud C, Olsen RK, Palombo DJ, Levine B, Black SE Congenital absence of the mammillary bodies: A novel finding in a well-studied case of developmental amnesia. Neuropsychologia 65: Santyr BG, Goubran M, Lau JC, Kwan BY, Salehi F, Lee DH, Mirsattari SM, Burneo JG, Steven DA, Parrent AG, de Ribaupierre S, Hammond RR, Peters TM, Khan AR Investigation of hippocampal substructures in focal temporal lobe epilepsy with and without hippocampal sclerosis at 7T. J Magn Reson Imaging. epub ahead of print. Seress L Comparative anatomy of the hippocampal dentate gyrus in adult and developing rodents, non-human primates and humans. Prog Brain Res 163: Seress L, Abraham H, Tornoczky T, Kosztolanyi G Cell formation in the human hippocampal formation from mid-gestation to the late postnatal period. Neuroscience 105:
9 A HARMONIZED HIPPOCAMPAL SUBFIELD PROTOCOL: KEY GOALS AND IMPACT 11 Shing YL, Rodrigue KM, Kennedy KM, Fandakova Y, Bodammer N, Werkle-Bergner M, Lindenberger U, Raz N Hippocampal subfield volumes: age, vascular risk, and correlation with associative memory. Front Aging Neurosci 3:2. Small SA, Perera GM, DeLaPaz R, Mayeux R, Stern Y Differential regional dysfunction of the hippocampal formation among elderly with memory decline and Alzheimer s disease. Ann Neurol 45: Small SA, Wu EX, Bartsch D, Perera GM, Lacefield CO, DeLaPaz R, Mayeux R, Stern Y, Kandel ER Imaging physiologic dysfunction of individual hippocampal subregions in humans and genetically modified mice. Neuron 28: Small SA, Schobel SA, Buxton RB, Witter MP, Barnes CA A pathophysiological framework of hippocampal dysfunction in ageing and disease. Nat Rev Neurosci 12: Squire LR, Stark CE, Clark RE The medial temporal lobe. Annu Rev Neurosci 27: Stokes J, Kyle C, Ekstrom AD Complementary roles of human hippocampal subfields in differentiation and integration of spatial context. J Cogn Neurosci 27: Suthana NA, Donix M, Wozny DR, Bazih A, Jones M, Heidemann RM, Trampel R, Ekstrom AD, Scharf M, Knowlton B, Turner R, Bookheimer SY High-resolution 7T fmri of human hippocampal subfields during associative learning. J Cogn Neurosci 27: Todd N, Moeller S, Auerbach EJ, Yacoub E, Flandin G, Weiskopf N Evaluation of 2D multiband EPI imaging for highresolution, whole-brain, task-based fmri studies at 3T: Sensitivity and slice leakage artifacts. Neuroimage 124: Van Leemput K, Bakkour A, Benner T, Wiggins G, Wald LL, Augustinack J, Dickerson BC, Golland P, Fischl B Automated segmentation of hippocampal subfields from ultra-high resolution in vivo MRI. 19: Van Petten C Relationship between hippocampal volume and memory ability in healthy individuals across the lifespan: Review and meta-analysis. Neuropsychologia 42: West MJ, Gundersen HJ Unbiased stereological estimation of the number of neurons in the human hippocampus. J Comp Neurol 296:1 22. Winterburn JL, Pruessner JC, Chavez S, Schira MM, Lobaugh NJ, Voineskos AN, Chakravarty MM A novel in vivo atlas of human hippocampal subfields using high-resolution 3 T magnetic resonance imaging. Neuroimage 74: Wisse LE, Biessels GJ, Heringa SM, Kuijf HJ, Koek DH, Luijten PR, Geerlings MI, Utrecht Vascular Cognitive Impairment (VCI.), Study Group Hippocampal subfield volumes at 7T in early Alzheimer s disease and normal aging. Neurobiol Aging 35: Wisse LE, Biessels GJ, Stegenga BT, Kooistra M, van der Veen PH, Zwanenburg JJ, van der Graaf Y, Geerlings MI Major depressive episodes over the course of 7 years and hippocampal subfield volumes at 7 tesla MRI: the PREDICT-MR study. J Affect Disord 175:1 7. Wisse LEM, Gerritsen L, Zwanenburg JJ, Kuijf HJ, Luijten PR, Biessels GJ, Geerlings MI Subfields of the hippocampal formation at 7 T MRI: In vivo volumetric assessment. Neuroimage 61: Yassa MA, Stark CE Pattern separation in the hippocampus. Trends Neurosci 34: Yassa MA, Stark SM, Bakker A, Albert MS, Gallagher M, Stark CE High-resolution structural and functional MRI of hippocampal CA3 and dentate gyrus in patients with amnestic Mild Cognitive Impairment. Neuroimage 51: Yushkevich PA, Wang H, Pluta J, Das SR, Craige C, Avants BB, Weiner MW, Mueller S Nearly automatic segmentation of hippocampal subfields in in vivo focal T2-weighted MRI. Neuroimage 53: Yushkevich PA, Amaral RS, Augustinack JC, Bender AR, Bernstein JD, Boccardi M, Bocchetta M, Burggren AC, Carr VA, Chakravarty MM, Chetelat G, Daugherty AM, Davachi L, Ding SL, Ekstrom A, Geerlings MI, Hassan A, Huang Y, Iglesias JE, La Joie R, Kerchner GA, LaRocque KF, Libby LA, Malykhin N, Mueller SG, Olsen RK, Palombo DJ, Parekh MB, Pluta JB, Preston AR, Pruessner JC, Ranganath C, Raz N, Schlichting ML, Schoemaker D, Singh S, Stark CE, Suthana N, Tompary A, Turowski MM, Van Leemput K, Wagner AD, Wang L, Winterburn JL, Wisse LE, Yassa MA, Zeineh MM, Hippocampal Subfields Group (HSG). 2015a. Quantitative comparison of 21 protocols for labeling hippocampal subfields and parahippocampal subregions in in vivo MRI: Towards a harmonized segmentation protocol. Neuroimage 1: Yushkevich PA, Pluta JB, Wang H, Xie L, Ding SL, Gertje EC, Mancuso L, Kliot D, Das SR, Wolk DA. 2015b. Automated volumetry and regional thickness analysis of hippocampal subfields and medial temporal cortical structures in mild cognitive impairment. Hum Brain Mapp 36: Zeineh MM, Engel SA, Bookheimer SY Application of cortical unfolding techniques to functional MRI of the human hippocampal region. Neuroimage 11: Zeineh MM, Engel SA, Thompson PM, Bookheimer SY Unfolding the human hippocampus with high resolution structural and functional MRI. Anat Rec 265:
University of California, Los Angeles B.S., Neuroscience Honors Thesis: Circadian Regulation of Long-term Potentiation
 Nanthia A. Suthana, Ph.D. Assistant Professor-in-Residence Department of Psychiatry & Biobehavioral Sciences Department of Neurosurgery Department of Psychology 300 Stein Plaza, Suite 562 University of
Nanthia A. Suthana, Ph.D. Assistant Professor-in-Residence Department of Psychiatry & Biobehavioral Sciences Department of Neurosurgery Department of Psychology 300 Stein Plaza, Suite 562 University of
HHS Public Access Author manuscript Neuroimage. Author manuscript; available in PMC 2016 May 01.
 Quantitative Comparison of 21 Protocols for Labeling Hippocampal Subfields and Parahippocampal Subregions in In Vivo MRI: Towards a Harmonized Segmentation Protocol Paul A. Yushkevich a,1, Robert S. C.
Quantitative Comparison of 21 Protocols for Labeling Hippocampal Subfields and Parahippocampal Subregions in In Vivo MRI: Towards a Harmonized Segmentation Protocol Paul A. Yushkevich a,1, Robert S. C.
University of California, Los Angeles B.S., Neuroscience Honors Thesis: Circadian Regulation of Long-term Potentiation
 Nanthia A. Suthana, Ph.D. Assistant Professor-in-Residence Department of Psychiatry & Biobehavioral Sciences Department of Neurosurgery Department of Psychology 300 Stein Plaza, Suite 562 University of
Nanthia A. Suthana, Ph.D. Assistant Professor-in-Residence Department of Psychiatry & Biobehavioral Sciences Department of Neurosurgery Department of Psychology 300 Stein Plaza, Suite 562 University of
ADNI-related initiatives in Europe. E-ADNI neugrid/outgrid EADC/ADNI harmonization of hippo manual segmentation
 ADNI-related initiatives in Europe E-ADNI neugrid/outgrid EADC/ADNI harmonization of hippo manual segmentation Pharma-Cog AIM: To develop new markers homologous in animal and humans sensitive to symptomatic
ADNI-related initiatives in Europe E-ADNI neugrid/outgrid EADC/ADNI harmonization of hippo manual segmentation Pharma-Cog AIM: To develop new markers homologous in animal and humans sensitive to symptomatic
Characterizing Anatomical Variability And Alzheimer s Disease Related Cortical Thinning in the Medial Temporal Lobe
 Characterizing Anatomical Variability And Alzheimer s Disease Related Cortical Thinning in the Medial Temporal Lobe Long Xie, Laura Wisse, Sandhitsu Das, Ranjit Ittyerah, Jiancong Wang, David Wolk, Paul
Characterizing Anatomical Variability And Alzheimer s Disease Related Cortical Thinning in the Medial Temporal Lobe Long Xie, Laura Wisse, Sandhitsu Das, Ranjit Ittyerah, Jiancong Wang, David Wolk, Paul
Alzheimer disease is the most common cause of dementia in
 ORIGINAL RESEARCH BRAIN Automated Segmentation of Hippocampal Subfields in Drug-Naïve Patients with Alzheimer Disease H.K. Lim, S.C. Hong, W.S. Jung, K.J. Ahn, W.Y. Won, C. Hahn, I.S. Kim, and C.U. Lee
ORIGINAL RESEARCH BRAIN Automated Segmentation of Hippocampal Subfields in Drug-Naïve Patients with Alzheimer Disease H.K. Lim, S.C. Hong, W.S. Jung, K.J. Ahn, W.Y. Won, C. Hahn, I.S. Kim, and C.U. Lee
Dissociable Profiles of Generalization/Discrimination in the Human Hippocampus During Associative Retrieval
 HIPPOCAMPUS 27:115 121 (2017) RAPID COMMUNICATION Dissociable Profiles of Generalization/Discrimination in the Human During Associative Retrieval Natalie G. De Shetler 1 and Jesse Rissman 1,2,3,4 * ABSTRACT:
HIPPOCAMPUS 27:115 121 (2017) RAPID COMMUNICATION Dissociable Profiles of Generalization/Discrimination in the Human During Associative Retrieval Natalie G. De Shetler 1 and Jesse Rissman 1,2,3,4 * ABSTRACT:
Unravelling The Subfields Of The Hippocampal Head Using 7-Tesla Structural MRI
 Western University Scholarship@Western Electronic Thesis and Dissertation Repository August 2016 Unravelling The Subfields Of The Hippocampal Head Using 7-Tesla Structural MRI Jordan M. K. DeKraker The
Western University Scholarship@Western Electronic Thesis and Dissertation Repository August 2016 Unravelling The Subfields Of The Hippocampal Head Using 7-Tesla Structural MRI Jordan M. K. DeKraker The
Dissociations Within Human Hippocampal Subregions During Encoding and Retrieval of Spatial Information
 HIPPOCAMPUS 21:694 701 (2011) RAPID COMMUNICATION Dissociations Within Human Hippocampal Subregions During Encoding and Retrieval of Spatial Information Nanthia Suthana, 1,2 Arne Ekstrom, 3 Saba Moshirvaziri,
HIPPOCAMPUS 21:694 701 (2011) RAPID COMMUNICATION Dissociations Within Human Hippocampal Subregions During Encoding and Retrieval of Spatial Information Nanthia Suthana, 1,2 Arne Ekstrom, 3 Saba Moshirvaziri,
C. Brock Kirwan, Ph.D.
 , Ph.D. Department of Psychology & Neuroscience Center Brigham Young University 1052 Kimball Tower Provo, UT 84602 Phone: (801) 422-2532 kirwan@byu.edu ACADEMIC & RESEARCH POSITIONS Assistant Professor:
, Ph.D. Department of Psychology & Neuroscience Center Brigham Young University 1052 Kimball Tower Provo, UT 84602 Phone: (801) 422-2532 kirwan@byu.edu ACADEMIC & RESEARCH POSITIONS Assistant Professor:
Curriculum Vitae. Personal information. Work experience. First name / Surname Martina BOCCHETTA
 Curriculum Vitae Personal information First name / Surname Martina BOCCHETTA E-mail mbocchetta@fatebenefratelli.it Nationality Italian Place and Date of birth Bergamo (Italy), May, 9 th 1986 Gender female
Curriculum Vitae Personal information First name / Surname Martina BOCCHETTA E-mail mbocchetta@fatebenefratelli.it Nationality Italian Place and Date of birth Bergamo (Italy), May, 9 th 1986 Gender female
SUPPLEMENTARY INFORMATION In format provided by Frank et al. (JULY 2010)
 Table 1 Imaging bios for Alzheimer s Visual rating High correlation with Multicenter studies have Accuracy for longitudinal hippocampus volume (R 2 been performed, but changes only at chance about 0.9,
Table 1 Imaging bios for Alzheimer s Visual rating High correlation with Multicenter studies have Accuracy for longitudinal hippocampus volume (R 2 been performed, but changes only at chance about 0.9,
Neural Activity in the Hippocampus and Perirhinal Cortex during Encoding Is Associated with the Durability of Episodic Memory
 Neural Activity in the Hippocampus and Perirhinal Cortex during Encoding Is Associated with the Durability of Episodic Memory Valerie A. Carr*, Indre V. Viskontas*, Stephen A. Engel, and Barbara J. Knowlton
Neural Activity in the Hippocampus and Perirhinal Cortex during Encoding Is Associated with the Durability of Episodic Memory Valerie A. Carr*, Indre V. Viskontas*, Stephen A. Engel, and Barbara J. Knowlton
Remembering the Past to Imagine the Future: A Cognitive Neuroscience Perspective
 MILITARY PSYCHOLOGY, 21:(Suppl. 1)S108 S112, 2009 Copyright Taylor & Francis Group, LLC ISSN: 0899-5605 print / 1532-7876 online DOI: 10.1080/08995600802554748 Remembering the Past to Imagine the Future:
MILITARY PSYCHOLOGY, 21:(Suppl. 1)S108 S112, 2009 Copyright Taylor & Francis Group, LLC ISSN: 0899-5605 print / 1532-7876 online DOI: 10.1080/08995600802554748 Remembering the Past to Imagine the Future:
Overcoming interference: An fmri investigation of pattern separation in the medial temporal lobe
 Research Overcoming interference: An fmri investigation of pattern separation in the medial temporal lobe C. Brock Kirwan 1 and Craig E.L. Stark 1,2,3 1 Department of Psychological and Brain Sciences,
Research Overcoming interference: An fmri investigation of pattern separation in the medial temporal lobe C. Brock Kirwan 1 and Craig E.L. Stark 1,2,3 1 Department of Psychological and Brain Sciences,
WWADNI MRI Core Boston July 2013
 WWADNI MRI Core Boston July 2013 Bret Borowski - Mayo Matt Bernstein - Mayo Jeff Gunter Mayo Clifford Jack - Mayo David Jones - Mayo Kejal Kantarci - Mayo Denise Reyes Mayo Matt Senjem Mayo Prashanthi
WWADNI MRI Core Boston July 2013 Bret Borowski - Mayo Matt Bernstein - Mayo Jeff Gunter Mayo Clifford Jack - Mayo David Jones - Mayo Kejal Kantarci - Mayo Denise Reyes Mayo Matt Senjem Mayo Prashanthi
Detailed segmentation of human hippocampal and subicular subfields using a combined approach
 RESEARCH HIGHLIGHT Detailed segmentation of human hippocampal and subicular subfields using a combined approach Song-Lin Ding Allen Institute for Brain Science, Seattle, WA 98103, USA Correspondence: Song-Lin
RESEARCH HIGHLIGHT Detailed segmentation of human hippocampal and subicular subfields using a combined approach Song-Lin Ding Allen Institute for Brain Science, Seattle, WA 98103, USA Correspondence: Song-Lin
Investigating the impact of midlife obesity on Alzheimer s disease (AD) pathology in a mouse model of AD
 Brain@McGill Prize for Neuroscience Undergraduate Research Colleen Rollins Supervisor: Dr. Mallar Chakravarty Revised: August 8, 2017 Investigating the impact of midlife obesity on Alzheimer s disease
Brain@McGill Prize for Neuroscience Undergraduate Research Colleen Rollins Supervisor: Dr. Mallar Chakravarty Revised: August 8, 2017 Investigating the impact of midlife obesity on Alzheimer s disease
Structural And Functional Integration: Why all imaging requires you to be a structural imager. David H. Salat
 Structural And Functional Integration: Why all imaging requires you to be a structural imager David H. Salat salat@nmr.mgh.harvard.edu Salat:StructFunct:HST.583:2015 Structural Information is Critical
Structural And Functional Integration: Why all imaging requires you to be a structural imager David H. Salat salat@nmr.mgh.harvard.edu Salat:StructFunct:HST.583:2015 Structural Information is Critical
The Functional Role of Hippocampal Subregions and Subfields: A High-Resolution fmri Study of Memory. Melanie Carol MacGillivray.
 The Functional Role of Hippocampal Subregions and Subfields: A High-Resolution fmri Study of Memory by Melanie Carol MacGillivray A thesis submitted in partial fulfillment of the requirements for the degree
The Functional Role of Hippocampal Subregions and Subfields: A High-Resolution fmri Study of Memory by Melanie Carol MacGillivray A thesis submitted in partial fulfillment of the requirements for the degree
Delineation of hippocampal subregions using T1-weighted magnetic resonance images at 3 Tesla
 Journal Articles Donald and Barbara Zucker School of Medicine Academic Works 2014 Delineation of hippocampal subregions using T1-weighted magnetic resonance images at 3 Tesla K. Rhindress Northwell Health
Journal Articles Donald and Barbara Zucker School of Medicine Academic Works 2014 Delineation of hippocampal subregions using T1-weighted magnetic resonance images at 3 Tesla K. Rhindress Northwell Health
Fully-automated volumetric MRI with normative ranges: Translation to clinical practice
 Behavioural Neurology 21 (2009) 21 28 21 DOI 10.3233/BEN-2009-0226 IOS Press Fully-automated volumetric MRI with normative ranges: Translation to clinical practice J.B. Brewer Department of Radiology and
Behavioural Neurology 21 (2009) 21 28 21 DOI 10.3233/BEN-2009-0226 IOS Press Fully-automated volumetric MRI with normative ranges: Translation to clinical practice J.B. Brewer Department of Radiology and
Four Tissue Segmentation in ADNI II
 Four Tissue Segmentation in ADNI II Charles DeCarli, MD, Pauline Maillard, PhD, Evan Fletcher, PhD Department of Neurology and Center for Neuroscience, University of California at Davis Summary Table of
Four Tissue Segmentation in ADNI II Charles DeCarli, MD, Pauline Maillard, PhD, Evan Fletcher, PhD Department of Neurology and Center for Neuroscience, University of California at Davis Summary Table of
Marina Boccardi Thomson Reuter Researcher ID: B ORCID ID: Update: May 2014
 Marina Boccardi Thomson Reuter Researcher ID: B-2733-2010 ORCID ID: 0000-0001-6744-8246 Update: May 2014 Education: Degree in Experimental Psychology Psychobiology, 110/110 cum laude, University of Padua-
Marina Boccardi Thomson Reuter Researcher ID: B-2733-2010 ORCID ID: 0000-0001-6744-8246 Update: May 2014 Education: Degree in Experimental Psychology Psychobiology, 110/110 cum laude, University of Padua-
Harvard Medical School Curriculum Vitae
 Harvard Medical School Curriculum Vitae Date Prepared: August 9, 2017 Name: Office Address: Corinna Bauer Department of Ophthalmology Massachusetts Eye & Ear Infirmary 20 Staniford Street 2W-257 Boston,
Harvard Medical School Curriculum Vitae Date Prepared: August 9, 2017 Name: Office Address: Corinna Bauer Department of Ophthalmology Massachusetts Eye & Ear Infirmary 20 Staniford Street 2W-257 Boston,
Morphometric MRI Analysis of the Parahippocampal Region in Temporal Lobe Epilepsy
 Morphometric MRI Analysis of the Parahippocampal Region in Temporal Lobe Epilepsy NEDA BERNASCONI, a ANDREA BERNASCONI, ZOGRAFOS CARAMANOS, FREDERICK ANDERMANN, FRANÇOIS DUBEAU, AND DOUGLAS L. ARNOLD Department
Morphometric MRI Analysis of the Parahippocampal Region in Temporal Lobe Epilepsy NEDA BERNASCONI, a ANDREA BERNASCONI, ZOGRAFOS CARAMANOS, FREDERICK ANDERMANN, FRANÇOIS DUBEAU, AND DOUGLAS L. ARNOLD Department
NIH Public Access Author Manuscript Proc SPIE. Author manuscript; available in PMC 2014 February 07.
 NIH Public Access Author Manuscript Published in final edited form as: Proc SPIE. 2007 March 5; 6512: 651236. doi:10.1117/12.708950. Semi-Automatic Parcellation of the Corpus Striatum Ramsey Al-Hakim a,
NIH Public Access Author Manuscript Published in final edited form as: Proc SPIE. 2007 March 5; 6512: 651236. doi:10.1117/12.708950. Semi-Automatic Parcellation of the Corpus Striatum Ramsey Al-Hakim a,
Human Brain Mapping. Heritability of hippocampal subfield volumes using a twin and non-twin siblings design
 Heritability of hippocampal subfield volumes using a twin and non-twin siblings design Journal: Manuscript ID HBM--0.R Wiley - Manuscript type: Research Article Date Submitted by the Author: n/a Complete
Heritability of hippocampal subfield volumes using a twin and non-twin siblings design Journal: Manuscript ID HBM--0.R Wiley - Manuscript type: Research Article Date Submitted by the Author: n/a Complete
Discriminative Analysis for Image-Based Studies
 Discriminative Analysis for Image-Based Studies Polina Golland 1, Bruce Fischl 2, Mona Spiridon 3, Nancy Kanwisher 3, Randy L. Buckner 4, Martha E. Shenton 5, Ron Kikinis 6, Anders Dale 2, and W. Eric
Discriminative Analysis for Image-Based Studies Polina Golland 1, Bruce Fischl 2, Mona Spiridon 3, Nancy Kanwisher 3, Randy L. Buckner 4, Martha E. Shenton 5, Ron Kikinis 6, Anders Dale 2, and W. Eric
The Low Sensitivity of Fluid-Attenuated Inversion-Recovery MR in the Detection of Multiple Sclerosis of the Spinal Cord
 The Low Sensitivity of Fluid-Attenuated Inversion-Recovery MR in the Detection of Multiple Sclerosis of the Spinal Cord Mark D. Keiper, Robert I. Grossman, John C. Brunson, and Mitchell D. Schnall PURPOSE:
The Low Sensitivity of Fluid-Attenuated Inversion-Recovery MR in the Detection of Multiple Sclerosis of the Spinal Cord Mark D. Keiper, Robert I. Grossman, John C. Brunson, and Mitchell D. Schnall PURPOSE:
Representations of Recent and Remote Autobiographical Memories in Hippocampal Subfields
 HIPPOCAMPUS 23:849 854 (2013) RAPID COMMUNICATION Representations of Recent and Remote Autobiographical Memories in Hippocampal Subfields Heidi M. Bonnici, Martin J. Chadwick, and Eleanor A. Maguire* ABSTRACT:
HIPPOCAMPUS 23:849 854 (2013) RAPID COMMUNICATION Representations of Recent and Remote Autobiographical Memories in Hippocampal Subfields Heidi M. Bonnici, Martin J. Chadwick, and Eleanor A. Maguire* ABSTRACT:
Automated detection of abnormal changes in cortical thickness: A tool to help diagnosis in neocortical focal epilepsy
 Automated detection of abnormal changes in cortical thickness: A tool to help diagnosis in neocortical focal epilepsy 1. Introduction Epilepsy is a common neurological disorder, which affects about 1 %
Automated detection of abnormal changes in cortical thickness: A tool to help diagnosis in neocortical focal epilepsy 1. Introduction Epilepsy is a common neurological disorder, which affects about 1 %
Multi-template approaches for segmenting the hippocampus: the case of the SACHA software
 Multi-template approaches for segmenting the hippocampus: the case of the SACHA software Ludovic Fillon, Olivier Colliot, Dominique Hasboun, Bruno Dubois, Didier Dormont, Louis Lemieux, Marie Chupin To
Multi-template approaches for segmenting the hippocampus: the case of the SACHA software Ludovic Fillon, Olivier Colliot, Dominique Hasboun, Bruno Dubois, Didier Dormont, Louis Lemieux, Marie Chupin To
Automatic pathology classification using a single feature machine learning - support vector machines
 Automatic pathology classification using a single feature machine learning - support vector machines Fernando Yepes-Calderon b,c, Fabian Pedregosa e, Bertrand Thirion e, Yalin Wang d,* and Natasha Leporé
Automatic pathology classification using a single feature machine learning - support vector machines Fernando Yepes-Calderon b,c, Fabian Pedregosa e, Bertrand Thirion e, Yalin Wang d,* and Natasha Leporé
Curriculum Vitae. Education 2007 B.Mus.A. Piano Performance University of Western Ontario
 Curriculum Vitae Date Prepared: October 4, 2018 Name:, Ph.D. Education 2007 B.Mus.A. Piano Performance University of Western Ontario 2007 B.Sc. Physiology and Psychology University of Western Ontario 2008
Curriculum Vitae Date Prepared: October 4, 2018 Name:, Ph.D. Education 2007 B.Mus.A. Piano Performance University of Western Ontario 2007 B.Sc. Physiology and Psychology University of Western Ontario 2008
Neuro-Imaging in dementia: using MRI in routine work-up Prof. Philip Scheltens
 Neuro-Imaging in dementia: Philip Scheltens Alzheimer Center VU University Medical Center Amsterdam The Netherlands 1 Outline of talk Current guidelines Imaging used to exclude disease Specific patterns
Neuro-Imaging in dementia: Philip Scheltens Alzheimer Center VU University Medical Center Amsterdam The Netherlands 1 Outline of talk Current guidelines Imaging used to exclude disease Specific patterns
Anatomy of the Hippocampus
 Anatomy of the Hippocampus Lecture 3.2 David S. Touretzky September, 2015 Human Hippocampus 2 Human Hippocampus 3 Hippocampus Means Seahorse Dissected human hippocampus next to a specimen of hippocampus
Anatomy of the Hippocampus Lecture 3.2 David S. Touretzky September, 2015 Human Hippocampus 2 Human Hippocampus 3 Hippocampus Means Seahorse Dissected human hippocampus next to a specimen of hippocampus
Chapter 5. Summary and Conclusions! 131
 ! Chapter 5 Summary and Conclusions! 131 Chapter 5!!!! Summary of the main findings The present thesis investigated the sensory representation of natural sounds in the human auditory cortex. Specifically,
! Chapter 5 Summary and Conclusions! 131 Chapter 5!!!! Summary of the main findings The present thesis investigated the sensory representation of natural sounds in the human auditory cortex. Specifically,
Henry Molaison. Biography. From Wikipedia, the free encyclopedia
 Henry Molaison From Wikipedia, the free encyclopedia Henry Gustav Molaison (February 26, 1926 December 2, 2008), known widely as H.M., was an American memory disorder patient who had a bilateral medial
Henry Molaison From Wikipedia, the free encyclopedia Henry Gustav Molaison (February 26, 1926 December 2, 2008), known widely as H.M., was an American memory disorder patient who had a bilateral medial
HHS Public Access Author manuscript Alzheimers Dement. Author manuscript; available in PMC 2016 February 01.
 HHS Public Access Author manuscript Published in final edited form as: Alzheimers Dement. 2015 February ; 11(2): 126 138. doi:10.1016/j.jalz.2014.02.009. Delphi definition of the EADC-ADNI Harmonized Protocol
HHS Public Access Author manuscript Published in final edited form as: Alzheimers Dement. 2015 February ; 11(2): 126 138. doi:10.1016/j.jalz.2014.02.009. Delphi definition of the EADC-ADNI Harmonized Protocol
Inter-method Reliability of Brainstem Volume Segmentation Algorithms in Preschoolers with ASD
 Bosco, Paolo and Giuliano, Alessia and Delafield-Butt, Jonathan and Muratori, Filippo and Calderoni, Sara and Retico, Alessandra (2017) Inter-method reliability of brainstem volume segmentation algorithms
Bosco, Paolo and Giuliano, Alessia and Delafield-Butt, Jonathan and Muratori, Filippo and Calderoni, Sara and Retico, Alessandra (2017) Inter-method reliability of brainstem volume segmentation algorithms
ORIGINAL CONTRIBUTION. Application of Automated Medial Temporal Lobe Atrophy Scale to Alzheimer Disease
 ORIGINAL CONTRIBUTION Application of Automated Medial Temporal Lobe Atrophy Scale to Alzheimer Disease Basil H. Ridha, MRCP; Josephine Barnes, MA, PhD; Laura A. van de Pol, MD; Jonathan M. Schott, MD,
ORIGINAL CONTRIBUTION Application of Automated Medial Temporal Lobe Atrophy Scale to Alzheimer Disease Basil H. Ridha, MRCP; Josephine Barnes, MA, PhD; Laura A. van de Pol, MD; Jonathan M. Schott, MD,
MRI Hippocampal Volume for Enrichment
 MRI Hippocampal Volume for Enrichment Derek Hill 1,2 & Jerry Novak 3, Pat Cole 4, Diane Stephenson 5 1. IXICO plc 2. UCL, London, UK 3. Johnson and Johnson 4. Takeda 5. CAMD, Critical Path Institute 1
MRI Hippocampal Volume for Enrichment Derek Hill 1,2 & Jerry Novak 3, Pat Cole 4, Diane Stephenson 5 1. IXICO plc 2. UCL, London, UK 3. Johnson and Johnson 4. Takeda 5. CAMD, Critical Path Institute 1
Nicole M. Dudukovic, Ph.D. Department of Psychology 1227 University of Oregon Eugene, OR Phone:
 Nicole M. Dudukovic, Ph.D. Department of Psychology 1227 University of Oregon Eugene, OR 97403 Email: ndudukov@uoregon.edu Phone: 541-346-7225 EDUCATION 2007 Stanford University, Ph.D., Psychology (Cognitive
Nicole M. Dudukovic, Ph.D. Department of Psychology 1227 University of Oregon Eugene, OR 97403 Email: ndudukov@uoregon.edu Phone: 541-346-7225 EDUCATION 2007 Stanford University, Ph.D., Psychology (Cognitive
City, University of London Institutional Repository
 City Research Online City, University of London Institutional Repository Citation: Worker, A., Dima, D., Combes, A., Crum, W. R., Streffer, J., Einstein, S., Mehta, M. A., Barker, G. J., Williams, S. C.
City Research Online City, University of London Institutional Repository Citation: Worker, A., Dima, D., Combes, A., Crum, W. R., Streffer, J., Einstein, S., Mehta, M. A., Barker, G. J., Williams, S. C.
High-resolution fmri of Content-sensitive Subsequent Memory Responses in Human Medial Temporal Lobe
 High-resolution fmri of Content-sensitive Subsequent Memory Responses in Human Medial Temporal Lobe Alison R. Preston 1,2, Aaron M. Bornstein 1, J. Benjamin Hutchinson 1, Meghan E. Gaare 1, Gary H. Glover
High-resolution fmri of Content-sensitive Subsequent Memory Responses in Human Medial Temporal Lobe Alison R. Preston 1,2, Aaron M. Bornstein 1, J. Benjamin Hutchinson 1, Meghan E. Gaare 1, Gary H. Glover
Investigation of hippocampal substructures in focal temporal lobe epilepsy with and without hippocampal sclerosis at 7T.
 Western University Scholarship@Western Robarts Imaging Publications Robarts Research Institute 2016 Investigation of hippocampal substructures in focal temporal lobe epilepsy with and without hippocampal
Western University Scholarship@Western Robarts Imaging Publications Robarts Research Institute 2016 Investigation of hippocampal substructures in focal temporal lobe epilepsy with and without hippocampal
ARTICLE IN PRESS. Neurobiology of Aging xxx (2007) xxx xxx
 Neurobiology of Aging xxx (2007) xxx xxx An MRI-based method for measuring volume, thickness and surface area of entorhinal, perirhinal, and posterior parahippocampal cortex Eric Feczko b, Jean C. Augustinack
Neurobiology of Aging xxx (2007) xxx xxx An MRI-based method for measuring volume, thickness and surface area of entorhinal, perirhinal, and posterior parahippocampal cortex Eric Feczko b, Jean C. Augustinack
Establishing a Relationship Between Activity Reduction in Human Perirhinal Cortex and Priming
 HIPPOCAMPUS 19:773 778 (2009) RAPID COMMUNICATION Establishing a Relationship Between Activity Reduction in Human Perirhinal Cortex and Priming Joel L. Voss, 1,2,3 * Katherina K.Y. Hauner, 1 and Ken A.
HIPPOCAMPUS 19:773 778 (2009) RAPID COMMUNICATION Establishing a Relationship Between Activity Reduction in Human Perirhinal Cortex and Priming Joel L. Voss, 1,2,3 * Katherina K.Y. Hauner, 1 and Ken A.
J. Benjamin Hutchinson
 J. Benjamin Hutchinson Department of Psychology Nightingale Hall Northeastern University Boston, MA, USA, 02115 Phone: 617-373-4196 Email: b.hutchinson@northeastern.edu http://www.northeastern.edu/hutchinsonlab
J. Benjamin Hutchinson Department of Psychology Nightingale Hall Northeastern University Boston, MA, USA, 02115 Phone: 617-373-4196 Email: b.hutchinson@northeastern.edu http://www.northeastern.edu/hutchinsonlab
Supplementary online data
 THELANCETNEUROLOGY-D-07-00083 Supplementary online data MRI assessments MRI at each site included a volumetric spoiled gradient echo (T1-weighted) sequence with slice partition thickness of 1 5 mm or less
THELANCETNEUROLOGY-D-07-00083 Supplementary online data MRI assessments MRI at each site included a volumetric spoiled gradient echo (T1-weighted) sequence with slice partition thickness of 1 5 mm or less
Hippocampal maturity promotes memory distinctiveness in childhood and adolescence
 Hippocampal maturity promotes memory distinctiveness in childhood and adolescence Attila Keresztes a, Andrew R. Bender a, Nils C. Bodammer a, Ulman Lindenberger a,b, Yee Lee Shing a,c,1, and Markus Werkle-Bergner
Hippocampal maturity promotes memory distinctiveness in childhood and adolescence Attila Keresztes a, Andrew R. Bender a, Nils C. Bodammer a, Ulman Lindenberger a,b, Yee Lee Shing a,c,1, and Markus Werkle-Bergner
LAURA A LIBBY. University of California, Davis 1544 Newton Court, Davis, CA (530)
 LAURA A LIBBY University of California, Davis 1544 Newton Court, Davis, CA 95618 lalibby@ucdavis.edu (530) 757-8865 http://lauraannelibby.github.io ACADEMIC HISTORY University of California, Davis Postdoctoral
LAURA A LIBBY University of California, Davis 1544 Newton Court, Davis, CA 95618 lalibby@ucdavis.edu (530) 757-8865 http://lauraannelibby.github.io ACADEMIC HISTORY University of California, Davis Postdoctoral
Fully-Automated, Multi-Stage Hippocampus Mapping in Very Mild Alzheimer Disease
 Fully-Automated, Multi-Stage Hippocampus Mapping in Very Mild Alzheimer Disease Lei Wang 1, Ali Khan 2, John G. Csernansky 1, Bruce Fischl 3, Michael I. Miller 4,5, John C. Morris 6,7, M. Faisal Beg 2
Fully-Automated, Multi-Stage Hippocampus Mapping in Very Mild Alzheimer Disease Lei Wang 1, Ali Khan 2, John G. Csernansky 1, Bruce Fischl 3, Michael I. Miller 4,5, John C. Morris 6,7, M. Faisal Beg 2
University, Montreal, Quebec, Canada
 Volumetry of Temporopolar, Perirhinal, Entorhinal and Parahippocampal Cortex from High-resolution MR Images: Considering the Variability of the Collateral Sulcus Jens C. Pruessner 1,2, Stefan Köhler 3,
Volumetry of Temporopolar, Perirhinal, Entorhinal and Parahippocampal Cortex from High-resolution MR Images: Considering the Variability of the Collateral Sulcus Jens C. Pruessner 1,2, Stefan Köhler 3,
Automated Whole Brain Segmentation Using FreeSurfer
 Automated Whole Brain Segmentation Using FreeSurfer https://surfer.nmr.mgh.harvard.edu/ FreeSurfer (FS) is a free software package developed at the Martinos Center for Biomedical Imaging used for three
Automated Whole Brain Segmentation Using FreeSurfer https://surfer.nmr.mgh.harvard.edu/ FreeSurfer (FS) is a free software package developed at the Martinos Center for Biomedical Imaging used for three
QIBA/NIBIB Final Progress Report
 QIBA/NIBIB Final Progress Report Amyloid Profile Continued Support with Brain Phantom Development Larry Pierce, David Haynor, John Sunderland, Paul Kinahan August 29, 2015 Title: Amyloid Profile Continued
QIBA/NIBIB Final Progress Report Amyloid Profile Continued Support with Brain Phantom Development Larry Pierce, David Haynor, John Sunderland, Paul Kinahan August 29, 2015 Title: Amyloid Profile Continued
Cover Page. The handle holds various files of this Leiden University dissertation
 Cover Page The handle http://hdl.handle.net/1887/26921 holds various files of this Leiden University dissertation Author: Doan, Nhat Trung Title: Quantitative analysis of human brain MR images at ultrahigh
Cover Page The handle http://hdl.handle.net/1887/26921 holds various files of this Leiden University dissertation Author: Doan, Nhat Trung Title: Quantitative analysis of human brain MR images at ultrahigh
Halle R. Dimsdale-Zucker 1544 Newton Court UC Davis Center for Neuroscience Davis, CA Address:
 Halle R. Dimsdale-Zucker 1544 Newton Court UC Davis Center for Neuroscience Davis, CA 95618 E-Mail Address: hrzucker@ucdavis.edu Education 2013-current Ph.D. in Cognitive Neuroscience Advanced to Candidacy,
Halle R. Dimsdale-Zucker 1544 Newton Court UC Davis Center for Neuroscience Davis, CA 95618 E-Mail Address: hrzucker@ucdavis.edu Education 2013-current Ph.D. in Cognitive Neuroscience Advanced to Candidacy,
Prior Knowledge and Memory Consolidation Expanding Competitive Trace Theory
 Prior Knowledge and Memory Consolidation Expanding Competitive Trace Theory Anna Smith Outline 1. 2. 3. 4. 5. Background in Memory Models Models of Consolidation The Hippocampus Competitive Trace Theory
Prior Knowledge and Memory Consolidation Expanding Competitive Trace Theory Anna Smith Outline 1. 2. 3. 4. 5. Background in Memory Models Models of Consolidation The Hippocampus Competitive Trace Theory
Distinct Ensemble Codes in Hippocampal Areas CA3 and CA1
 Distinct Ensemble Codes in Hippocampal Areas CA3 and CA1 Stefan Leutgeb, 1 Jill K. Leutgeb, 1 Alessandro Treves, 1,2 May-Britt Moser, 1 Edvard I. Moser 1* 1 Centre for the Biology of Memory, MTFS, Norwegian
Distinct Ensemble Codes in Hippocampal Areas CA3 and CA1 Stefan Leutgeb, 1 Jill K. Leutgeb, 1 Alessandro Treves, 1,2 May-Britt Moser, 1 Edvard I. Moser 1* 1 Centre for the Biology of Memory, MTFS, Norwegian
KATHERINE D DUNCAN. Ph.D. Psychology, New York University, New York, NY, USA Advisor: Lila Davachi
 University of Toronto Department of Psychology 100 St. George. St Toronto, ON, M5S 3G3 duncan@psych.utoronto.ca KATHERINE D DUNCAN EDUCATION Ph.D. Psychology, New York University, New York, NY, USA 2006-2011
University of Toronto Department of Psychology 100 St. George. St Toronto, ON, M5S 3G3 duncan@psych.utoronto.ca KATHERINE D DUNCAN EDUCATION Ph.D. Psychology, New York University, New York, NY, USA 2006-2011
Space and brain: a tabula rasa for the sense of direction. Paul A. Dudchenko
 Space and brain: a tabula rasa for the sense of direction Paul A. Dudchenko University of Stirling Psychology, School of Natural Sciences Stirling, FK9 4LA United Kingdom Phone: +44 131 650 3531 p.a.dudchenko@stir.ac.uk
Space and brain: a tabula rasa for the sense of direction Paul A. Dudchenko University of Stirling Psychology, School of Natural Sciences Stirling, FK9 4LA United Kingdom Phone: +44 131 650 3531 p.a.dudchenko@stir.ac.uk
Subject Index. Band of Giacomini 22 Benton Visual Retention Test 66 68
 Subject Index Adams, R.D. 4 Addenbrooke s Cognitive Examination 101 Alzheimer s disease clinical assessment histological imaging 104 neuroimaging 101 104 neuropsychological assessment 101 clinical presentation
Subject Index Adams, R.D. 4 Addenbrooke s Cognitive Examination 101 Alzheimer s disease clinical assessment histological imaging 104 neuroimaging 101 104 neuropsychological assessment 101 clinical presentation
Cortico-Striatal Connections Predict Control over Speed and Accuracy in Perceptual Decision Making
 Cortico-Striatal Connections Predict Control over Speed and Accuracy in Perceptual Decision Making Birte U. Forstmann 1,*, Andreas Schäfer 2, Alfred Anwander 2, Jane Neumann 2, Scott Brown 3, Eric-Jan
Cortico-Striatal Connections Predict Control over Speed and Accuracy in Perceptual Decision Making Birte U. Forstmann 1,*, Andreas Schäfer 2, Alfred Anwander 2, Jane Neumann 2, Scott Brown 3, Eric-Jan
Visual Rating Scale Reference Material. Lorna Harper Dementia Research Centre University College London
 Visual Rating Scale Reference Material Lorna Harper Dementia Research Centre University College London Background The reference materials included in this document were compiled and used in relation to
Visual Rating Scale Reference Material Lorna Harper Dementia Research Centre University College London Background The reference materials included in this document were compiled and used in relation to
Characterizing the role of the hippocampus during episodic simulation and encoding
 Received: 15 April 2017 Revised: 21 August 2017 Accepted: 22 August 2017 DOI: 10.1002/hipo.22796 RESEARCH ARTICLE Characterizing the role of the hippocampus during episodic simulation and encoding Preston
Received: 15 April 2017 Revised: 21 August 2017 Accepted: 22 August 2017 DOI: 10.1002/hipo.22796 RESEARCH ARTICLE Characterizing the role of the hippocampus during episodic simulation and encoding Preston
Lag-Sensitive Repetition Suppression Effects in the Anterior Parahippocampal Gyrus
 HIPPOCAMPUS 15:557 561 (2005) RAPID COMMUNICATION Lag-Sensitive Repetition Suppression Effects in the Anterior Parahippocampal Gyrus Craig J. Brozinsky,* Andrew P. Yonelinas, Neal E.A. Kroll, and Charan
HIPPOCAMPUS 15:557 561 (2005) RAPID COMMUNICATION Lag-Sensitive Repetition Suppression Effects in the Anterior Parahippocampal Gyrus Craig J. Brozinsky,* Andrew P. Yonelinas, Neal E.A. Kroll, and Charan
Visualization strategies for major white matter tracts identified by diffusion tensor imaging for intraoperative use
 International Congress Series 1281 (2005) 793 797 www.ics-elsevier.com Visualization strategies for major white matter tracts identified by diffusion tensor imaging for intraoperative use Ch. Nimsky a,b,
International Congress Series 1281 (2005) 793 797 www.ics-elsevier.com Visualization strategies for major white matter tracts identified by diffusion tensor imaging for intraoperative use Ch. Nimsky a,b,
THE HUMAN DENTATE GYRUS PLAYS A NECESSARY ROLE IN DISCRIMINATING NEW MEMORIES STEVENSON W. BAKER
 THE HUMAN DENTATE GYRUS PLAYS A NECESSARY ROLE IN DISCRIMINATING NEW MEMORIES STEVENSON W. BAKER A THESIS SUBMITTED TO THE FACULTY OF GRADUATE STUDIES IN PARTIAL FULFILLMENT OF THE REQUIREMENTS FOR THE
THE HUMAN DENTATE GYRUS PLAYS A NECESSARY ROLE IN DISCRIMINATING NEW MEMORIES STEVENSON W. BAKER A THESIS SUBMITTED TO THE FACULTY OF GRADUATE STUDIES IN PARTIAL FULFILLMENT OF THE REQUIREMENTS FOR THE
Supplementary Information Methods Subjects The study was comprised of 84 chronic pain patients with either chronic back pain (CBP) or osteoarthritis
 Supplementary Information Methods Subjects The study was comprised of 84 chronic pain patients with either chronic back pain (CBP) or osteoarthritis (OA). All subjects provided informed consent to procedures
Supplementary Information Methods Subjects The study was comprised of 84 chronic pain patients with either chronic back pain (CBP) or osteoarthritis (OA). All subjects provided informed consent to procedures
USE OF BIOMARKERS TO DISTINGUISH SUBTYPES OF DEMENTIA. SGEC Webinar Handouts 1/18/2013
 Please visit our website for more information http://sgec.stanford.edu/ SGEC Webinar Handouts 1/18/2013 2013 WEBINAR SERIES STATE OF THE SCIENCE: DEMENTIA EVALUATION AND MANAGEMENT AMONG DIVERSE OLDER
Please visit our website for more information http://sgec.stanford.edu/ SGEC Webinar Handouts 1/18/2013 2013 WEBINAR SERIES STATE OF THE SCIENCE: DEMENTIA EVALUATION AND MANAGEMENT AMONG DIVERSE OLDER
Performance-Related Sustained and Anticipatory Activity in Human Medial Temporal Lobe during Delayed Match-to-Sample
 11880 The Journal of Neuroscience, September 23, 2009 29(38):11880 11890 Behavioral/Systems/Cognitive Performance-Related Sustained and Anticipatory Activity in Human Medial Temporal Lobe during Delayed
11880 The Journal of Neuroscience, September 23, 2009 29(38):11880 11890 Behavioral/Systems/Cognitive Performance-Related Sustained and Anticipatory Activity in Human Medial Temporal Lobe during Delayed
Brain tissue and white matter lesion volume analysis in diabetes mellitus type 2
 Brain tissue and white matter lesion volume analysis in diabetes mellitus type 2 C. Jongen J. van der Grond L.J. Kappelle G.J. Biessels M.A. Viergever J.P.W. Pluim On behalf of the Utrecht Diabetic Encephalopathy
Brain tissue and white matter lesion volume analysis in diabetes mellitus type 2 C. Jongen J. van der Grond L.J. Kappelle G.J. Biessels M.A. Viergever J.P.W. Pluim On behalf of the Utrecht Diabetic Encephalopathy
Experimental Assessment of Infarct Lesion Growth in Mice using Time-Resolved T2* MR Image Sequences
 Experimental Assessment of Infarct Lesion Growth in Mice using Time-Resolved T2* MR Image Sequences Nils Daniel Forkert 1, Dennis Säring 1, Andrea Eisenbeis 2, Frank Leypoldt 3, Jens Fiehler 2, Heinz Handels
Experimental Assessment of Infarct Lesion Growth in Mice using Time-Resolved T2* MR Image Sequences Nils Daniel Forkert 1, Dennis Säring 1, Andrea Eisenbeis 2, Frank Leypoldt 3, Jens Fiehler 2, Heinz Handels
AnalyzePro HIPPOCAMPAL VOLUME ASSESSMENT
 AnalyzePro HIPPOCAMPAL VOLUME ASSESSMENT HIPPOCAMPAL VOLUME ASSESSMENT GUIDE Introduction page 1 Loading Data page 4 Preprocessing Steps page 5 Manual AC-PC Alignment of Brain Data page 6 Cropping and
AnalyzePro HIPPOCAMPAL VOLUME ASSESSMENT HIPPOCAMPAL VOLUME ASSESSMENT GUIDE Introduction page 1 Loading Data page 4 Preprocessing Steps page 5 Manual AC-PC Alignment of Brain Data page 6 Cropping and
Computational approach to the schizophrenia: disconnection syndrome and dynamical pharmacology
 Computational approach to the schizophrenia: disconnection syndrome and dynamical pharmacology Péter Érdi1,2, Brad Flaugher 1, Trevor Jones 1, Balázs Ujfalussy 2, László Zalányi 2 and Vaibhav Diwadkar
Computational approach to the schizophrenia: disconnection syndrome and dynamical pharmacology Péter Érdi1,2, Brad Flaugher 1, Trevor Jones 1, Balázs Ujfalussy 2, László Zalányi 2 and Vaibhav Diwadkar
Application of Cortical Unfolding Techniques to Functional MRI of the Human Hippocampal Region
 NeuroImage 11, 668 683 (2000) doi:10.1006/nimg.2000.0561, available online at http://www.idealibrary.com on Application of Cortical Unfolding Techniques to Functional MRI of the Human Hippocampal Region
NeuroImage 11, 668 683 (2000) doi:10.1006/nimg.2000.0561, available online at http://www.idealibrary.com on Application of Cortical Unfolding Techniques to Functional MRI of the Human Hippocampal Region
Use of Multimodal Neuroimaging Techniques to Examine Age, Sex, and Alcohol-Related Changes in Brain Structure Through Adolescence and Young Adulthood
 American Psychiatric Association San Diego, CA 24 May 2017 Use of Multimodal Neuroimaging Techniques to Examine Age, Sex, and Alcohol-Related Changes in Brain Structure Through Adolescence and Young Adulthood
American Psychiatric Association San Diego, CA 24 May 2017 Use of Multimodal Neuroimaging Techniques to Examine Age, Sex, and Alcohol-Related Changes in Brain Structure Through Adolescence and Young Adulthood
Resistance to forgetting associated with hippocampus-mediated. reactivation during new learning
 Resistance to Forgetting 1 Resistance to forgetting associated with hippocampus-mediated reactivation during new learning Brice A. Kuhl, Arpeet T. Shah, Sarah DuBrow, & Anthony D. Wagner Resistance to
Resistance to Forgetting 1 Resistance to forgetting associated with hippocampus-mediated reactivation during new learning Brice A. Kuhl, Arpeet T. Shah, Sarah DuBrow, & Anthony D. Wagner Resistance to
Structural lesion analysis: applications
 Structural lesion analysis: applications Dagmar Timmann Department of Neurology University of Duisburg-Essen, Germany Human cerebellar lesion conditions 1. Focal cerebellar lesions a. Stroke Pro s: - acute
Structural lesion analysis: applications Dagmar Timmann Department of Neurology University of Duisburg-Essen, Germany Human cerebellar lesion conditions 1. Focal cerebellar lesions a. Stroke Pro s: - acute
HST.583 Functional Magnetic Resonance Imaging: Data Acquisition and Analysis Fall 2006
 MIT OpenCourseWare http://ocw.mit.edu HST.583 Functional Magnetic Resonance Imaging: Data Acquisition and Analysis Fall 2006 For information about citing these materials or our Terms of Use, visit: http://ocw.mit.edu/terms.
MIT OpenCourseWare http://ocw.mit.edu HST.583 Functional Magnetic Resonance Imaging: Data Acquisition and Analysis Fall 2006 For information about citing these materials or our Terms of Use, visit: http://ocw.mit.edu/terms.
Supplementary Material for The neural basis of rationalization: Cognitive dissonance reduction during decision-making. Johanna M.
 Supplementary Material for The neural basis of rationalization: Cognitive dissonance reduction during decision-making Johanna M. Jarcho 1,2 Elliot T. Berkman 3 Matthew D. Lieberman 3 1 Department of Psychiatry
Supplementary Material for The neural basis of rationalization: Cognitive dissonance reduction during decision-making Johanna M. Jarcho 1,2 Elliot T. Berkman 3 Matthew D. Lieberman 3 1 Department of Psychiatry
Morris water maze: standard test for spatial memory in rodents
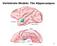 Vertebrate Models: The Hippocampus 34 Vertebrate Models: The Hippocampus 35 Vertebrate Models: The Hippocampus 36 Vertebrate Models: The Hippocampus 37 Animal Models of Learning (Vertebrates) Morris water
Vertebrate Models: The Hippocampus 34 Vertebrate Models: The Hippocampus 35 Vertebrate Models: The Hippocampus 36 Vertebrate Models: The Hippocampus 37 Animal Models of Learning (Vertebrates) Morris water
ANA MARIE DAUGHERTY, Ph.D. curriculum vitae
 ANA MARIE DAUGHERTY, Ph.D. curriculum vitae Office 8307.1 Maccabees 5057 Woodward Ave. 8 th Floor Detroit, MI 48202 ana.daugherty@wayne.edu Research Lab 1111 Integrative Biosciences Center 6135 Woodward
ANA MARIE DAUGHERTY, Ph.D. curriculum vitae Office 8307.1 Maccabees 5057 Woodward Ave. 8 th Floor Detroit, MI 48202 ana.daugherty@wayne.edu Research Lab 1111 Integrative Biosciences Center 6135 Woodward
Early identification of neurobiological markers of remission. Michael Bodnar, PhD Ashok K. Malla, MD Martin Lepage, PhD
 Early identification of neurobiological markers of remission Michael Bodnar, PhD Ashok K. Malla, MD Martin Lepage, PhD Outline Why study remission? Defining remission Data collection Results neurocognition
Early identification of neurobiological markers of remission Michael Bodnar, PhD Ashok K. Malla, MD Martin Lepage, PhD Outline Why study remission? Defining remission Data collection Results neurocognition
Reproducibility of Visual Activation During Checkerboard Stimulation in Functional Magnetic Resonance Imaging at 4 Tesla
 Reproducibility of Visual Activation During Checkerboard Stimulation in Functional Magnetic Resonance Imaging at 4 Tesla Atsushi Miki*, Grant T. Liu*, Sarah A. Englander, Jonathan Raz, Theo G. M. van Erp,
Reproducibility of Visual Activation During Checkerboard Stimulation in Functional Magnetic Resonance Imaging at 4 Tesla Atsushi Miki*, Grant T. Liu*, Sarah A. Englander, Jonathan Raz, Theo G. M. van Erp,
Preserved Hippocampal Novelty Responses Following Anterior Temporal-Lobe Resection That Impairs Familiarity But Spares Recollection
 HIPPOCAMPUS 21:847 854 (2011) Preserved Hippocampal Novelty Responses Following Anterior Temporal-Lobe Resection That Impairs Familiarity But Spares Recollection Ben Bowles, 1 Edward B. O Neil, 1 Seyed
HIPPOCAMPUS 21:847 854 (2011) Preserved Hippocampal Novelty Responses Following Anterior Temporal-Lobe Resection That Impairs Familiarity But Spares Recollection Ben Bowles, 1 Edward B. O Neil, 1 Seyed
Evidence for a specific role of the anterior hippocampal region in successful associative encoding
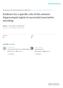 See discussions, stats, and author profiles for this publication at: http://www.researchgate.net/publication/6233976 Evidence for a specific role of the anterior hippocampal region in successful associative
See discussions, stats, and author profiles for this publication at: http://www.researchgate.net/publication/6233976 Evidence for a specific role of the anterior hippocampal region in successful associative
Dentate imaging: methods and applications
 Dentate imaging: methods and applications Dagmar Timmann Department of Neurology University of Duisburg-Essen, Germany Cerebellar output via deep nuclei Folie 2 Brodal 2010 Cerebellar nuclei Fastigial
Dentate imaging: methods and applications Dagmar Timmann Department of Neurology University of Duisburg-Essen, Germany Cerebellar output via deep nuclei Folie 2 Brodal 2010 Cerebellar nuclei Fastigial
Unfolding the Human Hippocampus With High Resolution Structural and Functional MRI
 THE ANATOMICAL RECORD (NEW ANAT.) 265:111 120, 2001 TUTORIAL Unfolding the Human Hippocampus With High Resolution Structural and Functional MRI MICHAEL M. ZEINEH, STEPHEN A. ENGEL, PAUL M. THOMPSON, AND
THE ANATOMICAL RECORD (NEW ANAT.) 265:111 120, 2001 TUTORIAL Unfolding the Human Hippocampus With High Resolution Structural and Functional MRI MICHAEL M. ZEINEH, STEPHEN A. ENGEL, PAUL M. THOMPSON, AND
Neuroimaging biomarkers and predictors of motor recovery: implications for PTs
 Neuroimaging biomarkers and predictors of motor recovery: implications for PTs 2018 Combined Sections Meeting of the American Physical Therapy Association New Orleans, LA February 21-24, 2018 Presenters:
Neuroimaging biomarkers and predictors of motor recovery: implications for PTs 2018 Combined Sections Meeting of the American Physical Therapy Association New Orleans, LA February 21-24, 2018 Presenters:
Modeling of Hippocampal Behavior
 Modeling of Hippocampal Behavior Diana Ponce-Morado, Venmathi Gunasekaran and Varsha Vijayan Abstract The hippocampus is identified as an important structure in the cerebral cortex of mammals for forming
Modeling of Hippocampal Behavior Diana Ponce-Morado, Venmathi Gunasekaran and Varsha Vijayan Abstract The hippocampus is identified as an important structure in the cerebral cortex of mammals for forming
MR Volumetric Analysis of the Human Entorhinal, Perirhinal, and Temporopolar Cortices
 AJNR Am J Neuroradiol 19:659 671, April 1998 MR Volumetric Analysis of the Human Entorhinal, Perirhinal, and Temporopolar Cortices Ricardo Insausti, Kirsi Juottonen, Hilkka Soininen, Ana Maria Insausti,
AJNR Am J Neuroradiol 19:659 671, April 1998 MR Volumetric Analysis of the Human Entorhinal, Perirhinal, and Temporopolar Cortices Ricardo Insausti, Kirsi Juottonen, Hilkka Soininen, Ana Maria Insausti,
A systems neuroscience approach to memory
 A systems neuroscience approach to memory Critical brain structures for declarative memory Relational memory vs. item memory Recollection vs. familiarity Recall vs. recognition What about PDs? R-K paradigm
A systems neuroscience approach to memory Critical brain structures for declarative memory Relational memory vs. item memory Recollection vs. familiarity Recall vs. recognition What about PDs? R-K paradigm
Assessing Brain Volumes Using MorphoBox Prototype
 MAGNETOM Flash (68) 2/207 33 Assessing Brain Volumes Using MorphoBox Prototype Alexis Roche,2,3 ; Bénédicte Maréchal,2,3 ; Tobias Kober,2,3 ; Gunnar Krueger 4 ; Patric Hagmann ; Philippe Maeder ; Reto
MAGNETOM Flash (68) 2/207 33 Assessing Brain Volumes Using MorphoBox Prototype Alexis Roche,2,3 ; Bénédicte Maréchal,2,3 ; Tobias Kober,2,3 ; Gunnar Krueger 4 ; Patric Hagmann ; Philippe Maeder ; Reto
Automated Volumetric Cardiac Ultrasound Analysis
 Whitepaper Automated Volumetric Cardiac Ultrasound Analysis ACUSON SC2000 Volume Imaging Ultrasound System Bogdan Georgescu, Ph.D. Siemens Corporate Research Princeton, New Jersey USA Answers for life.
Whitepaper Automated Volumetric Cardiac Ultrasound Analysis ACUSON SC2000 Volume Imaging Ultrasound System Bogdan Georgescu, Ph.D. Siemens Corporate Research Princeton, New Jersey USA Answers for life.
Intracranial Studies Of Human Epilepsy In A Surgical Setting
 Intracranial Studies Of Human Epilepsy In A Surgical Setting Department of Neurology David Geffen School of Medicine at UCLA Presentation Goals Epilepsy and seizures Basics of the electroencephalogram
Intracranial Studies Of Human Epilepsy In A Surgical Setting Department of Neurology David Geffen School of Medicine at UCLA Presentation Goals Epilepsy and seizures Basics of the electroencephalogram
arxiv: v2 [cs.cv] 19 Dec 2017
![arxiv: v2 [cs.cv] 19 Dec 2017 arxiv: v2 [cs.cv] 19 Dec 2017](/thumbs/89/97557782.jpg) An Ensemble of Deep Convolutional Neural Networks for Alzheimer s Disease Detection and Classification arxiv:1712.01675v2 [cs.cv] 19 Dec 2017 Jyoti Islam Department of Computer Science Georgia State University
An Ensemble of Deep Convolutional Neural Networks for Alzheimer s Disease Detection and Classification arxiv:1712.01675v2 [cs.cv] 19 Dec 2017 Jyoti Islam Department of Computer Science Georgia State University
