Reversal of tolerance to the antitransit e ects of morphine during acute intestinal in ammation in mice
|
|
|
- Randolf Marshall
- 5 years ago
- Views:
Transcription
1 British Journal of Pharmacology (1997) 122, 1216 ± Stockton Press All rights reserved 0007 ± 1188/97 $12.00 Reversal of tolerance to the antitransit e ects of morphine during acute intestinal in ammation in mice Olga Pol & 1 Margarita M. Puig Anesthesiology Research Unit, IMIM, Department of Anesthesiology, Hospital Universitario del Mar, Barcelona, Spain 1 The aim of investigation was to establish and compare the reversibility of tolerance to the antitransit e ects of morphine by three di erent procedures: (a) acute in ammation of the gut, (b) lorglumide a cholecystokinin A (CCK A ) receptor antagonist, or (c) MK-801, an N-methyl-D-aspartate (NMDA) receptor ion channel blocker. The type of interaction between morphine and lorglumide or MK-801 on the inhibition of gastrointestinal transit (GIT) in naive animals was also evaluated. 2 Male Swiss CD-1 mice were implanted with 75 mg of morphine base or placebo pellets. Gastrointestinal transit was assessed with a charcoal meal and results expressed as % inhibition of GIT. In ammation was induced by the intragastric (p.o.) administration of croton oil (CO), while controls received castor oil (CA) or saline (SS). Morphine was administered by subcutaneous (s.c.) or intracerebroventricular (i.c.v.) injection, to naive and tolerant animals treated with CO, CA or SS. Doseresponse curves for s.c. morphine were also perforrmed in naive and tolerant mice receiving 5.2 or 7.4 nmol (s.c.) lorglumide or MK-801, respectively. 3 The ED 50 values for inhibition of GIT by s.c. morphine were: and nmol in naive and tolerant animals, respectively, demonstrating a ve fold decrease in the potency of morphine. In naive animals, in ammation (CO) decreased the ED 50 of morphine three times ( nmol). However, no tolerance to s.c. morphine (ED nmol) was manifested during intestinal in ammation. After i.c.v. administration, a similar degree of tolerance to morphine was observed (4.8 fold decrease in potency). Intestinal in ammation had no e ect on the ED 50 values of i.c.v. morphine in naive and tolerant animals, showing that reversal of tolerance is related to local mechanism/s. Mean values for intestinal ph were and in SS and CO treated mice, respectively. In addition, morphine was 74 times more potent by the i.c.v. than by the s.c. route (naive-ss). 4 Morphine and lorglumide interacted synergistically in naive animals; in addition, the administration of lorglumide reversed tolerance to s.c. morphine. No interaction (additivity) was observed in naive animals when morphine and MK-801 were administered in combination. However, the drug completely reversed tolerance to the antitransit e ects of morphine. 5 The present investigation shows that acute in ammation of the gut reverses tolerance to the antitransit e ects of s.c. morphine by a peripheral mechanism. Qualitatively similar results were obtained after the administration of lorglumide or MK-801. Our results suggest that a local decrease in ph could play an important role during in ammation, while antagonism of endogenous compensatory systems would explain the reversal of tolerance induced by lorglumide or MK-801. Keywords: Morphine tolerance; opioids; gastrointestinal transit; intestinal in ammation; cholecystokinin-a receptor antagonist; N-methyl-D-aspartate receptor channel blocker Introduction 1 Author for correspondence at: Department of Anesthesiology, Hospital Universitario del Mar, Paseo MarõÂ timo 22, Barcelona, Spain. Opioids are extensively used as analgesics in the treatment of pain of moderate to severe intensity. Chronic administration of morphine induces tolerance to the analgesic and other pharmacological e ects, including constipation. However, the rate and magnitude of the development of tolerance to the di erent e ects is not completely clear (Ja e, 1990). Tolerance to opioids is manifested by a decrease in the intensity of the response to a constant dose (Foley, 1991) and a shift to the right of the dose-response curve to the agonists. Tolerance to m opioid receptor agonists has been widely investigated, but the mechanisms involved are not well understood. At present, an increase in adenylate cyclase activity (Nestler, 1992), uncoupling of the m opioid receptors to G-proteins (Tao et al., 1993), and/or sequestration of the m opioid receptors in the presence of an unaltered m opioid receptor gene expression (Buzas et al., 1996), are the most likely mechanisms involved. The intestine has been widely used to evaluate the pharmacological e ects of opioids and ileus/constipation constitute one of the major side e ects after acute and chronic administration. The characteristics of the development of tolerance to the intestinal e ects of opioids are not well characterized in animals or man. However, in experimental models of nociception, di erent groups of drugs have been shown to prevent/ reverse tolerance to the antinociceptive e ect of opioids; among them, calcium channel blockers (Dierssen et al., 1990) and cholecystokinin (CCK) or N-methyl-D-aspartate (NMDA) receptor antagonists (Trujillo & Akil, 1991; Xu et al., 1992) seem to be most e ective. In addition, other investigators have shown a lack of tolerance to the antinociceptive e ects of morphine in di erent models of chronic in ammation (Neil et al., 1990; Stein et al., 1996). However, the impact or in uence of the above mentioned drugs and/or intestinal in ammation, on the reversal of tolerance to the antitransit e ects of morphine have not been investigated. We have previously shown that acute intestinal in ammation increases approximately three times the potency of s.c. morphine to inhibit gastrointestinal transit (GIT) (Pol et al., 1994) by a peripheral mechanism. In the present investigation, our working hypothesis was that local events occurring at the in amed site such as `sensitization' of m opioid receptors, could restore the response to morphine in tolerant animals. Among the local events occurring during acute in ammation, a decrease in ph is usually present, a phenomena that has been shown to increase the e cacy of opioids (Selley et al., 1993).
2 Thus in our study, we measured ph changes in the di erent experimental conditions. Since the e ects of morphine on GIT are mediated by m opioid receptors located in the brain and the gut, we assessed tolerance (at peripheral and central sites) after the s.c. and i.c.v. administration of morphine. In addition, we examined the reversibility of tolerance to the antitransit e ects of morphine after the administration of lorglumide, a CCK-A antagonist (Van der Bent et al., 1994) or MK-801, an NMDA receptor ion channel blocker (Trujillo & Akil, 1991). The type of interaction between morphine and lorglumide or MK-801 on the inhibition of GIT was also evaluated. Preliminary results have been presented at the XXVI International Narcotics Research Conference (Scotland, U.K., July 9 ± 13, 1995). Methods Animals Experiments were performed on male Swiss CD-1 mice, weighing 20 ± 25 g. Animals were housed under 12 hour light/ 12 hour dark conditions in a room with controlled temperature (228C) and humidity (66%). Mice had free access to food and water and were used after a minimum of four days acclimatization to the housing conditions. All experiments were conducted between 9 h 00 min and 14 h 00 min. The International Association for the Study of Pain guidelines on ethical standards for investigations in animals were followed. Gastrointestinal transit (GIT) GIT was measured according to procedures used in our laboratory (Pol et al., 1995; Puig et al., 1996). Brie y, food was removed 18 h before the experiment but animals had free access to water. At this time, a charcoal meal (0.25 ml of 10% charcoal in 5% gum acacia) was administered intragastrically and GIT was evaluated 20 min later. Animals were killed and the small intestine separated from the omentum avoiding stretching. The length of intestine from the pyloric sphincter to the ileocecal junction and the distance travelled by the charcoal meal were measured. For each animal, GIT was calculated as the percentage (%) of distance travelled by the charcoal, relative to the total length of the small intestine (% of GIT). The inhibitory e ects of drugs on GIT are expressed as a percentage of inhibition of the transit in a drug-treated animal (test GIT) when compared with the mean transit measured in a group of vehicle-treated mice (n=20). % inhibition=[(vehicle GIT ± test GIT)/(vehicle GIT)]6100. In ammation of the gut Acute in ammation was induced by the intragastric administration of 0.05 ml of croton oil (CO) according to the method described previously (Pol et al., 1994; 1996b). Two other groups of animals receiving the same volume of saline (SS) or castor oil (CA) served as controls (Pol et al., 1996c). All groups of animals were weighed and placed into separate cages. After 3 h, the animals were again weighed and GIT was measured with a charcoal meal. Histological examination O. Pol & M.M. Puig Animals were killed three hours after p.o. administration of CO, CA or SS, and the small intestine rapidly excised. Samples of the proximal jejunum were xed with 2.5% glutaraldehyde in phosphate bu er (200 ± 400 mosm; ph: 7.2 ± 7.4) for 24 h and processed either for light or electron microscopic examination. For light microscopic studies, the samples were embedded in para n and 5 m thick longitudinal and radial sections were obtained with a sliding microtome; dewaxed sections were stained with haematoxylin and eosin. For electron microscopic studies, small blocks were washed in phosphate bu er and post xed with 2% osmium tetroxide for 2 h; Tolerance and intestinal inflammation 1217 blocks were embedded in araldite and ultrathin sections were obtained with an ultramicrotome; the sections were stained with uranyl acetate and lead citrate. Five animals treated with CO, CA or SS were used for histological studies. Measurement of ph Local changes in ph induced by in ammation were measured in the small intestine (25 cm down from the pyloric sphincter) of mice treated with CO, CA or SS (10 animals per group). Animals were killed three h after treatment and ph recorded with a microph 2001 (Crison, Barcelona, Spain) and a glass electrode, calibrated just before the study with standard solutions (ph 4.0 and 7.0). Induction of tolerance to morphine Under light ether anaesthesia, mice were implanted with 75 mg morphine base or placebo pellets, at the nape of the neck. GIT was determined 72 h after pellet implantation. Dependence on morphine was demonstrated in SS, CA and CO treated animals after the s.c. administration of naloxone (68.7 nmol) which induced a withdrawal syndrome characterized by increased spontaneous activity, tremours, jumping, and in some animals, spontaneous vocalization (Brown et al., 1988). Dose-response curves for morphine Dose-response curves for morphine were obtained after s.c. or i.c.v. injection, in naive and tolerant animals treated with CO, CA or SS. Subcutaneous morphine was administered 30 min before and i.c.v. morphine immediately before the charcoal. In addition, dose-response curves for s.c. morphine were recorded in naive and tolerant mice (in the absence of in ammation, SS) that had received, 40 min before morphine, one of the following drugs: lorglumide (CCK A antagonist), MK-801 (NMDA receptor ion channel blocker) or vehicle. Drugs were administered intraperitoneally (i.p.). In order to be able to compare the relative potency of morphine when administered by the s.c. and i.c.v. routes, all doses of morphine are expressed in nmol. Drugs The drugs used were: morphine sulphate (Alcaiber S.A., Madrid, Spain), lorglumide (ICN Ibe rica, S.A., Barcelona, Spain), MK-801 ((+)-5-methyl-10,11-dihydro-5H-dibenzyl[a,d]cyclohepten-5,10-imine hydrogen maleate; dizocilpine, Research Biochemicals Incorporated, U.S.A.) and naloxone hydrochloride (Sigma Chemical Co., St Louis, Mo., U.S.A.). Drugs were dissolved in pyrogen-free 0.9% sodium chloride just before use and injected s.c. (at the nape of the neck) or i.p., in a nal volume of 10 ml kg 71. For the i.c.v. injections, drugs were delivered in a volume of 5 ml, by a Hamilton syringe (Microdispenser Socorex, PANREAC S.A.) tted with a 26- gauge needle, according to the method of Haley & McCormick (1957). The site of injection was 2 mm caudal and 2 mm lateral to bregma, and 3 mm in depth from the skull surface. Data analysis The data are expressed as group means+s.e. All statistical calculations were performed as described by Tallarida & Murray (1986). ED 50 (dose which produced a 50% e ect) values were determined by linear regression analysis of doseresponse relations based on at least ten mice per dose. Tests for parallelism and validity of the tests were estimated by use of parallel line assays. Statistical analysis for signi cant differences between two groups were obtained by Student's t test; when multiple groups were compared, one or two way analysis of variance (ANOVA) followed by Student-Newman-Keuls test was used. A value of P50.05 was considered signi cant.
3 1218 O. Pol & M.M. Puig Results E ect of castor (CA) or croton oil (CO) on the gut The intragastric administration of CA or CO to fasted mice induced (3 h after treatment): (a) weight loss of 9.8 and 10% in CA and CO treated mice, respectively, while SS animals lost 4% (P50.05); (b) an increase in GIT of 12% (CA) and 20% (CO) that was not statistically signi cant between groups, and (c) electron microscopic evidence of in ammation in CO but not in CA animals. The main morphological changes included an increased number of clear vesicles in the cytoplasm of jejunal epithelial cells and enlarged spaces lled with ne granular material in the extravascular compartment. Since these ndings have been published previously (Pol et al., 1994; 1996c), only ve animals per group were used to corroborate the characteristics of the model. Thus, our results show that only animals treated with CO presented intestinal in ammation. Dose-response curves for morphine during in ammation in naive and tolerant mice Dose-response curves for s.c. morphine were recorded in naive and tolerant mice treated with CO (in ammation), CA or SS. Similar experiments were carried out after the i.c.v. administration of morphine. For each treatment and route of administration, linear parallel dose-response curves were obtained and their ED 50 values calculated. Table 1 shows the ED 50 values of morphine after s.c. administration. In SS animals implanted with a morphine pellet, the dose-response curve for morphine was shifted to the right resulting in a 5.4 fold increase of the ED 50, thus demonstrating tolerance to morphine. In tolerant mice treated with CA (without in ammation) the potency of morphine decreased 4.6 times; no statistical di erences were observed when SS and CA were compared. In the presence of in ammation (CO) the ED 50 of morphine was the same in naive and morphine implanted (pellet) animals, showing that intestinal in ammation restores the potency of morphine in tolerant animals. The results also show that in ammation of the gut (CO) increases the potency of morphine 3.1 and 15.2 times in naive and tolerant animals respectively. The antitransit e ects of morphine in naive and tolerant animals were also assessed after i.c.v. administration. These experiments were performed in SS and CO treated animals, since CA did not induce signi cant di erences from the control (SS) when morphine was given s.c. Parallel dose-response curves for i.c.v. morphine were obtained in naive and tolerant animals treated with SS or CO (Figure 1). In SS animals (no in ammation), tolerance was manifested by a shift to the right of the dose-response curve for morphine. In addition, the presence of intestinal in ammation (CO) did not alter the dose-response curves for i.c.v. morphine in naive or tolerant animals. From the data, ED 50 values were obtained in the four Tolerance and intestinal inflammation study groups (Table 2). The results show that in the absence of in ammation (SS), the potency of morphine in tolerant mice was 4.8 lower than in naive mice; the same decrease was observed during in ammation. In addition, CO treatment did not alter the ED 50 of morphine in naive or tolerant mice. The results show that morphine is 74 times more potent when administered by the i.c.v. than by the s.c. route in naive SS animals. Mean values of intestinal ph (n=5 for each group) measured in tolerant SS, CA and CO treated animals were (SS), (CA), and (CO). The di erences were statistically signi cant between the CO and the CA or SS groups (P50.01; Student-Newman-Keuls test). E ects of lorglumide and MK-801 on the inhibition of GIT induced by morphine in naive mice Dose-response curves for s.c. morphine were recorded in naive animals receiving 5.2 nmol of lorglumide or 7.4 nmol of MK- 801 (Figure 2). The administration of 5.2 nmol lorglumide alone induced a % inhibition of GIT. Pretreatment with this dose of lorglumide induced a shift to the left of the dose-response curve for morphine with ED 50 values of and nmol for morphine alone and morphine+lorglumide, respectively (Table 3). The interaction be- Inhibition of gastrointestinal transit (%) Naive-SS Naive-CO Tolerant-SS Tolerant-CO Table 1 ED 50 values for e ect of subcutaneous morphine (nmol), on the % inhibition of GIT in naive and tolerant mice, treated with saline (SS), croton (CO) or castor oil (CA) Treatment (p.o) Naive Tolerant SS CO CA a b a c b c Ratio (tolerant/naive) Values shown are mean ED 50 +s.e. (in nmol). For naive and tolerant mice treated with SS, CO or CA, identical letters (aa, bb, or cc) indicate that there are no signi cant di erences between groups, while di erent letters (ab, ac or bc) show signi cant di erences (P<0.05; Student-Newman- Keuls test) Dose (nmol) Figure 1 Inhibition of GIT induced by i.c.v. morphine in naive and tolerant animals treated with SS or CO. Gastrointestinal transit was evaluated 20 min after administration of charcoal. Each point represents the mean of 10 or more mice; vertical lines show s.e. *P50.05, when naive and tolerant animals were compared (Student- Newman-Keuls test).
4 Table 2 ED 50 values for e ect of intracerebroventricular morphine (nmol), on the % inhibition of GIT in naive and tolerant mice, treated with saline (SS) or croton oil (CO) Treatment Ratio (p.o.) Naive Tolerant (tolerant/naive) SS CO Ratio SS/CO a a b b Values shown are mean ED 50 +s.e. (nmol). For naive and tolerant mice treated with SS or CO, identical letters (aa or bb) indicate that there are no signi cant di erences between groups, while di erent letters (ab) show signi cant di erences (P<0.05; Student-Newman-Keuls test). Inhibition of gastrointestinal transit (%) Morphine + saline Morphine + MK-801 Morphine + lorglumide O. Pol & M.M. Puig Dose (nmol) Figure 2 Inhibition of GIT induced by s.c. morphine in naive animals receiving (i.p.) SS, lorglumide or MK-801. Gastrointestinal transit was evaluated 20 min after administration of charcoal. Each point represents the mean of 10 or more mice; vertical lines show s.e. *P50.05, when morphine+lorglumide was compared to morphine alone (Student-Newman-Keuls test). Tolerance and intestinal inflammation 1219 tween morphine and lorglumide appears to be synergistic based on the following evidence: (a) when the dose responsecurves for morphine and morphine+lorglumide were analysed by two way ANOVA, signi cant e ects of the treatment (morphine, morphine+lorglumide), the dose and their interaction (P50.02; n=10 for each point) were obtained. Further comparison with one-way ANOVA showed that the e ect of treatment was related to lorglumide, since the combination signi cantly increased the antitransit e ects of morphine (P50.001; n=10 for each dose). (b) The non-parallel displacement to the left of the dose-response curve of the combination, although lorglumide alone did not show a signi cant e ect. (c) The sum of the e ects produced by each drug alone was at each dose of morphine tested, signi cantly lower than the observed e ects of the combination. MK-801 alone at a dose of 7.4 nmol, produced a % inhibition of GIT. In the presence of MK-801, the dose-response curve for morphine was slightly (but not signi cantly) shifted to the left (Figure 2). ED 50 values for morphine alone and combined with MK-801 were and nmol, respectively. When data were analysed by two-way ANOVA, a signi cant e ect of the dose (P50.001; n=10 for each point), but not of the drugs or their interaction (drugs6doses) was observed. In addition, when the e ect of MK-801 individually was added to the e ect produced by each dose of morphine alone, the expected e ects were not di erent from the observed e ects, demonstrating additivity. The results show that both the NMDA and CCK A receptor antagonists have a modest inhibitory e ect on GIT. However, only the latter enhanced the antitransit e ects of morphine. E ects of lorglumide and MK-801 on the inhibition of GIT induced by morphine in tolerant mice The e ects of 5.2 nmol of lorglumide or 7.4 nnol of MK-801 on the dose-response curves for morphine were also evaluated in tolerant mice; in these animals, the inhibitory e ect of each drug alone was unaltered. The resulting dose-response curves were superimposed and shifted to the left when compared to the curve with morphine alone (Figure 3). Similar results were obtained in tolerant animals treated with CO, indicating that lorglumide, MK-801 and intestinal in ammation can reverse tolerance to the antitransit e ects of morphine. In tolerant animals, the e ects of treatment (morphine, morphine+lorglumide, morphine+mk-801 or morphine- CO) and dose were analysed by 2-way ANOVA. Each factor (treatment and doses) as well as their interaction had a signi cant e ect (P50.001; n=10 for each point). Comparison with one-way ANOVA showed that the e ect of treatment was related to lorglumide, MK-801 or CO, since their combination with morphine signi cantly increased the inhibition of GIT when compared to morphine alone (P50.001; n=10 for each point). Table 3 shows the ED 50 values for morphine in naive and tolerant animals. Our results demonstrate that tolerance to morphine is reversed by the administration of lorglumide or MK-801 or by acute in ammation. In naive animals the interaction of morphine+lorglumide was synergistic and had an ED 50 of ; in tolerant animals, the ED 50 was signi cantly increased to nmol demonstrating the reversal of tolerance and the absence of an interaction between morphine and lorglumide (additivity). The ED 50 s of animals treated with MK-801 or CO remained unaltered in tolerant animals. Results were compared by one-way ANOVA. Table 3 ED 50 (nmol) of morphine+ss, morphine+lorglumide, morphine+mk-801 and morphine during in ammation (morphine+co), on % inhibition of GIT in naive and tolerant mice Drugs Naive Tolerant Morphine+SS Morphine+lorglumide Morphine+MK-801 Morphine+CO a b a b c a a b Values shown are mean ED 50 +s.e. (nmol). For naive and tolerant mice treated with morphine+ss, morphine+lorglumide, morphine+mk-801 or morphine+co identical letters (aa or bb) indicate that there are no signi cant di erences between groups, while di erent letters (ab, ac or bc) show signi cant di erences (P<0.05; Students-Newman- Keuls test).
5 O. Pol & M.M. Puig Inhibition of gastrointestinal transit (%) Discussion Morphine + lorglumide Morphine + SS Morphine + CO Morphine + MK The present investigation shows for the rst time, that intestinal in ammation reverses tolerance to the antitransit e ect of peripheral morphine. A qualitatively similar e ect was observed after the administration of lorglumide or MK-801, although the underlying mechanisms are likely to be di erent. We have recently characterized and validated (Pol et al., 1994; 1995; 1996a) a model of acute intestinal in ammation in mice, induced by the intragastric administration of CO. Acute in ammation (substantiated by electron microscopy) increased the potency of s.c. morphine approximately three times. In the present study we have also used CA, an agent that induces hypersecretory diarrhoea without in ammation (Pol et al., 1996c). When the antitransit e ects of morphine were compared in the groups treated with CO or CA, the potency of morphine increased only in the CO group, demonstrating that in ammation is required in order to enhance the antitransit e ects of morphine. Since the e ects of s.c. morphine in SS and CA treated animals were indistinguishable, all subsequent experiments were performed in SS treated animals as control. The relative potency of morphine when administered by central (i.c.v.) and systemic (s.c.) routes was also assessed. In these experiments, we converted all units (mg ml 71 and mg kg 71 ) to nmol, in order to be able to compare the ED 50 values for morphine; the results show that i.c.v. morphine is 74 times more potent in inhibiting GIT than s.c. morphine. This nding probably involves di erent factors such as receptor density, local morphine concentration and kinetics, and has been previously described in the literature (Shook et al., 1987). The development of tolerance to the gastrointestinal e ects of opioids has been shown to follow a di erent pattern than 100 Dose (nmol) Figure 3 Inhibition of GIT induced by s.c. morphine in tolerant animals treated with SS, lorglumide, MK-801, or CO. Gastrointestinal transit was evaluated 20 min after administration of charcoal. Each point represents the mean of 10 or more mice; vertical lines show s.e. *P50.001, when morphine alone (control group, SS) was compared to the three other groups (Student-Newman-Keuls test) Tolerance and intestinal inflammation other pharmacological e ects. In our model, tolerance was manifested by a decrease of approximately ve times in the potency of morphine, supporting the results obtained by other investigators in small rodents (Wong et al., 1980; Brown et al., 1988). Our results show a similar degree or level of tolerance to morphine after s.c. and i.c.v. administration (ratios tolerant/ naive were 5.4 and 4.8 for the s.c. and i.c.v. routes, respectively), demonstrating that brain and gut m opioid receptors mediating the antitransit e ects of morphine behave in a similar manner when chronically exposed to opioids. The potency of morphine administered by the s.c. and i.c.v. routes was evaluated in the presence of intestinal in ammation (CO). As previously found, the potency of s.c. morphine increased three times during in ammation. However, after i.c.v. administration, the potency of morphine was analogous in SS and CO mice, demonstrating that the enhanced e ects of morphine during in ammation are mediated by intestinal m opioid receptors. As mentioned above, tolerance was substantiated by a ve fold decrease in the potency of s.c. or i.c.v. morphine. In ammation of the gut did not alter the response to i.c.v. morphine in tolerant animals. However, after s.c. administration no tolerance (as de ned by a shift to the right of the dose-response curve) to morphine was observed. Since the decrease in the potency of i.c.v. morphine was comparable in SS and CO animals, we postulate that local events induced by the in ammatory process could be responsible for the reversal of tolerance to morphine. Among other events, peripheral in ammation induces local acidosis; in our model we were able to document a signi cnat decrease in ph of 0.7 in CO-treated mice. In preparations of isolated neuronal membranes, acidosis increased the e cacy of opioid agonists to inhibit adenylate cyclase (Selley et al., 1993), probably by enhancing the interaction of the m opioid receptors with guanine-nucleotide-binding proteins (G-proteins). Since chronic exposure to opioids has been shown to uncouple the m opioid receptors from G-proteins (Tao et al., 1993), local acidosis could restore the potency of morphine in tolerant animals by this mechanism. In addition, tolerance induces sequestration of m opioid receptors (Zadina et al., 1995) and it is possible that tissue acidosis would disrupt the perineurum and facilitate the access of morphine to `sequestered' m opioid receptors in the gut (Antonijevic et al., 1995). Thus, the enhanced e ects of opioids during intestinal in ammation could be partially explained on the basis of a local decrease in ph. Accepting that chronic exposure to opioids induces changes in the con guration of the m opioid receptors or the receptor-g protein complex, we hypothesize that local acidosis and/or other events occurring during intestinal in ammation could be responsible for the reversal of tolerance to morphine. In addition to the events occurring at the receptor-complex level, other endogenous systems that apparently function as compensatory mechanisms could be involved in the development of tolerance to opioids (Han, 1995); among them, CCK and excitatory amino acids (EAAs), both implicated in primary sensory transmission, are likely to have a prominent role. In the present experiments, we have used two receptor-speci c antagonists, lorglumide and MK-801, in order to block these endogenous compensatory mechanisms. The antagonists were selected because there is strong evidence in the literature for their e ectiveness and selectivity for CCK A and NMDA receptors, respectively (Makovec et al., 1987; Woodru et al., 1987). The doses of the antagonists were used on the basis of previous data that evaluated their e ects on intestinal motility (D'Amato et al., 1991; Campbell et al., 1991) or nociception (Kellstein & Mayer, 1991; Bilsky et al., 1996). In addition, we performed pilot experiments with di erent doses of lorglumide (0.52, 5.2 and 52 nmol); a maximal increase in the antitransit e ect of a xed dose of morphine was obtained with 5.2 nmol of lorglumide, and thus, this dose was used in the study. Since MK-801 (at doses of 0.74, 7.4 and 74 nmol) did not induce changes in the e ect of morphine, we employed 7.4 nmol, a dose that has been demonstrated to block NMDA receptors in di ferent experimental conditions.
6 O. Pol & M.M. Puig In tolerant rats, an enhanced expression of the gene encoding CCK has been observed (Zhou et al., 1992) and CCK antagonists have been shown to prevent the development of tolerance to the antinociceptive e ects of morphine (Xu et al., 1992). In the gut, the CCK family of peptides have an important role in the neural control of intestinal motility (Meyer et al., 1989). We have used lorglumide, a CCK A receptor antagonist, in order to evaluate the reversal of tolerance to the antitransit e ects of morphine. Our results show that lorglumide by itself has a negligible antitransit e ect, but that at the dose-ratios used, interacts synergistically with morphine; interestingly, synergism is no longer present in tolerant animals, a phenomenon that has been previously observed when clonidine and morphine are combined (Roerig, 1995). The administration of a xed dose of lorglumide reversed the tolerance to morphine, supporting a negative feedback control mechanism between opioids and CCK in the gut. The interaction between lorglumide and morphine on the inhibition of GIT in naive and tolerant animals could be of future importance if combinations of CCK antagonists and opioids are used in the treatment of pain. Excitatory amino acid (EAA) antagonists such as MK-801 have been shown to produce analgesia (Dunbar & Yaksh, 1996), enhance the e ects of morphine (Advokat & Rhein, 1995) and block or attenuate tolerance to opioids (Trujillo & Akil, 1994). Some of these results have been di cult to replicate and controversy still exists regarding the interaction of opioids and EAA antagonists on nociception. In the gut, in situ hybridization studies have identi ed myenteric neurones which express mrna for the NMDA receptor (Broussard et al., 1994), suggesting that glutamate could have a neurotransmitter role in the enteric nervous system. However, our results show that at the dose used in the study, MK-801 alone did not Tolerance and intestinal inflammation 1221 alter GIT in control (SS) naive animals. When MK-801 was administered in combination with morphine, no interaction could be observed (additivity) but the drug completely reversed tolerance to morphine. Thus, although CCK A and NMDA antagonists reversed morphine tolerance in our experimental conditions, the type of interaction between morphine and the antagonists lorglumide and MK-801 was di erent. In the present investigation we showed that acute in ammation of the gut reverses tolerance to the antitransit e ects of morphine by a peripheral mechanism. The observed e ect was qualitatively similar to the reversal of tolerance observed after the administration of either a CCK A antagonist or NMDA receptor ion channel blocker, but their mechanism/s appear to be distinct. We hypothesize that the decrease in local ph plays an important role during acute in ammation, while antagonisms of endogenous compensatory systems would explain the reversal of tolerance induced by lorglumide or MK-801. In addition, intestinal in ammation and acidosis could also alter the local regulation/expression of other `compensatory systems' including CCK and glutamate. Undoubtedly, the problem of tolerance to opioids during acute and chronic in ammation involves other systems and complex cellular events that cannot be explained by a modest change in ph. However, tissue acidosis could play a role in the reversal of tolerance to the antitransit e ects of morphine during acute in ammation of the gut. The authors wish to thank Mr Sergi Leanez for his excellent technical assistance. The work was partially supported by grants from Fondo de Investigaciones Sanitarias, Ministerio de Sanidad, 94/1380 and 96/0719, Madrid, Spain. References ADVOKAT, C. & RHEIN, F.Q. (1995). Potentiation of morphineinduced antinociception in acute spinal rats by the NMDA antagonist dextrorphan. Brain Res., 699, 157 ± 160. ANTONIJEVIC, I., MOUSA, S.A., SCHAFER, M. & STEIN, C. (1995). Perineural defect and peripheral opioid analgesia in in ammation. J. Neurosci., 15, 165 ± 172. BILSKY, E.J., INTURRISI, C.E., SADEÂ E,W.,HRUBY,V.J.&PORRECA, F. (1996). Competitive and non-competitive NMDA antagonists block the development of antinociceptive tolerance to morphine, but not to selective m or d opioid agonists in mice. Pain, 68, 229 ± 237. BROUSSARD, D.L., LI, X., PRITCHETT, D.B., LYNCH, D., BANNER- MANN, G.C. & PLEASURE, D. (1994). The expression of a NMDA receptor gene in guinea-pig myenteric plexus. NeuroReport, 5, 973 ± 976. BROWN, N.J., COUPAR, I.M. & RUMSEY, R.D.E. (1988). The e ect of acute and chronic administration of morphine and morphine withdrawal on intestinal transit time in the rat. J. Pharm. Pharmacol., 40, 844 ± 848. BUZAS, B., ROSENBERGER, J. & COX, B.M. (1996). m and d opioid receptor gene expression after chronic treatment with opioid agonist. NeuroReport, 7, 1505 ± CAMPBELL, B.G., COUCEYRO, P., KEANA, J.F.W. & WEBER, E. (1991). N-methyl-D-aspartate receptor-mediated contractions of the guinea pig ileum longitudinal muscle/myenteric plexus preparation: modulation by phencyclidine and glycine receptors. J. Pharmacol. Exp. Ther., 257, 754 ± 766. D'AMATO, M., STAMFORD, I.F. & BENNETT, A. (1991). Studies of three non-peptide cholecystokinin antagonists (devazepide, lorglumide and loxiglumide) in human isolated alimentary muscle and guinea-pig ileium. Br.J.Pharmacol.,102, 391 ± 395. DIERSSEN, M., FLOREZ, J. & HURLE, M.A. (1990). Calcium channel modulation by dihydropyridine modi es sulfentanil-induced antinociception in acute and tolerant conditions. Naunyn- Schmiedeberg's Arch. Pharmacol., 342, 559 ± 565. DUNBAR, S. & YAKSH, T.L. (1996). Concurrent spinal infusion of MK-801 blocks spinal tolerance and dependence induced by chronic intrathecal morphine in the rat. Anesthesiology, 84, 1177 ± FOLEY, K.M. (1991). Clinical tolerance to opioids. In Towards a New Pharmacotherapy of Pain. pp. 181 ± 203. Chichester: John Wiley & Sons. HALLEY, T.J. & MCCORNICK, W.G. (1957). Pharmacological e ects produced by intracerebral injection of drugs in the conscious mice. Br. J. Pharmacol. Chemother., 12, 12 ± 15. HAN, J.S. (1995). Cholecystokinin octapeptide (CCK-8): a negative feedback control mechanism for opioid analgesia. In Progress in Brain Research.ed.Yu,A.,Eng,L.,McMahan,V.,Shulman,H., Shooter, S. & Stadlin, A. pp. 263 ± 271, China: Elsevier Science BV. JAFFE, J.H. (1990). Drug addiction and drug abuse. In The Pharmacological Basis of Therapeutics. ed. Goodman, A., Rall, T.W., Nies, A.S. & Taylor, P. pp. 522 ± 573, New York: Pergamon Press. KELLSTEIN, D.E. & MAYER, D.J. (1991). Spinal co-administration of cholecystokinin antagonists with morphine prevents the development of opioid tolerance. Pain, 47, 221 ± 229. MAKOVEC, F., BANI, M., CEREDA, R., CHISTE, R., PACINI, M.A., REVEL, L., ROVATI, L.C. & SETNIKAR, I. (1987). Pharmacological properties of lorglumide as a member of a new class of cholecystokinin antagonists. Arzneimittelforschung, 37, 1265 ± MEYER, B.M., WERTH, B.A., BEGLINGER, C., HILDEBRAND, P., JANSEN, J.B., ZACH, D., ROVATI, L.C. & STADLER, G.A. (1989) Role of cholecystokinin regulation of gastrointestinal motor functions. Lancet, 2, 12 ± 15. NEIL, A., KAYSER, V., CHEN, Y.L. & GUILBAUD, G. (1990). Repeated low doses of morphine do not induce tolerance but increase the opioid antinociceptive e ect in rats with a peripheral neuropathy. Brain Res., 552, 140 ± 143.
7 1222 O. Pol & M.M. Puig NESTLER, E.J. (1992). Molecular mechanisms of drug addiction. J. Neurosci., 12, 2439 ± POL, O., FERRER, I. & PUIG, M.M. (1994). Diarrhea associated with intestinal in ammation increases the potency of m and d opioids on the inhibition of gastrointestinal transit in mice. J. Pharmacol. Exp. Ther., 270, 386 ± 391. POL, O., PLANAS, E. & PUIG, M.M. (1995). Peripheral e ects of naloxone in mice with acute diarrhea associated with intestinal in ammation. J. Pharmacol. Exp. Ther., 272, 1271 ± POL, O., PLANAS, E. & PUIG, M.M. (1996a). E ects of morphine and liposomal morphine in a model of intestinal in ammation in mice. Pharmacology, 53, 180 ± 189. POL, O., SANCHEZ, B. & PUIG, M.M. (1996b). Peripheral e ects of opioids in a model of intestinal in ammation in mice. Pharmacology, 53, 340 ± 350. POL, O., VALLE, L.L., FERRER, I. & PUIG, M.M. (1996c). The inhibitory e ects of a 2 -adrenoceptor agonists on gastrointestinal transit during croton oil-induced intestinal in ammation. Br. J. Pharmacol., 119, 1649 ± PUIG, M.M., POL, O. & WARNER, W. (1996). Interaction of morphine and clonidine on gastrointestinal transit in mice. Anesthesiology, 85, 1403 ± ROERIG, S.C. (1995). Decreased spinal morphine/clonidine antinociception synergism in morphine-tolerant mice. Life Sci., 56, 115 ± 122. SELLEY, D.E., BREIVOGEL, C.S. & CHILDERS, S.R. (1993). Modi cation of opioid receptor-g-protein function by low ph pretreatment of membranes from NG cells: Increase in opioid agonist e cacy by decreased inactivation of G-proteins. Mol. Pharmacol., 44, 731 ± 741. SHOOK, J.E., PELTON, J.T., HRUBY, V.J. & BURKS, T.F. (1987). Peptide opioid antagonists separate peripheral and central opioid antitransit e ects. J. Pharmacol. Exp. Ther., 243, 492 ± 500. STEIN, C., PFLUÈ GER, M., YASSOURIDIS, A., HOELZL, J., LEHRBER- GER, K., WELTE, C. & HASSAN, A.H.S. (1996). No tolerance to peripheral morphine analgesia in presence of opioid expression in in amed synovia. J. Clin. Invest., 98, 793 ± 799. Tolerance and intestinal inflammation TALLARIDA, R.J. & MURRAY, R.B. (1986). Manual of Pharmacologic Calculations with Computer Programs. New York: Springer- Verlag. TAO, P.L., LEE, C.R., LAW, P.Y. & LOH, H.H. (1993). The interaction of the m-opioid receptor and G protein is altered after chronic morphine treatment in rats. Naunyn-Schmiedeberg's Arch. Pharmacol., 348, 504 ± 508. TRUJILLO, K.A. & AKIL, H. (1991). Inhibition of morphine tolerance and dependence by the NMDA receptor antagonist MK-801. Science, 251, 85 ± 87. TRUJILLO, K.A. & AKIL, H. (1994). Inhibition of opiate tolerance by non-competitive N-methyl-D-aspartate receptor antagonists. Brain Res., 633, 178 ± 188. VAN DER BENT, A., IJZERMAN, A.P. & SOUDIJN, W. (1994). Molecular-modelling of CCK-A receptors. Drug Des. Discov., 12, 129 ± 148. WONG, C.L., ROBERTS, M.B. & WAI, M.K. (1980). E ect of morphine and naloxone on intestinal transit in mice. Eur. J. Pharmacol., 64, 289 ± 295. WOODRUFF, G.N., FOSTER, A.C., GILL, R., KEMP, J.A., WONG, E.H. &IVERSEN,L.L.(1987). The interaction between MK-801 and receptors for N-methyl-D-aspartate: functional consequences. Neuropharmacology, 26, 903 ± 909. XU, X.J., WIESENFELD-HALLIN, Z., HUGHES, J., HORWELL, D.C. & HOKFELT, T.C. (1992). A selective antagonist of cholecystokinin- B receptors, prevents morphine tolerance in the rat. Br. J. Pharmacol., 105, 591 ± 596. ZADINA, J.E., KASTIN, A.J., HARRISON, L.M., GE, L.J. & CHANG, S.L. (1995) Opiate receptor changes after chronic exposure to agonists and antagonists. Ann. New York Acad. Sci., 757, 353 ± 361. ZHOU, Y., SUN, Y.H., ZHANG, Z.W. & HAN, J.S. (1992). Accelerated expression of cholecystokinin gene in the brain of rats rendered tolerant to morphine. NeuroReport, 3, 1121 ± (Received March 12, 1997 Revised June 26, 1997 Accepted July 31, 1997)
Peripheral Effects of Opioids in a Model of Chronic Intestinal Inflammation in Mice 1,2
 0022-3565/98/2873-1068$03.00/0 THE JOURNAL OF PHARMACOLOGY AND EXPERIMENTAL THERAPEUTICS Vol. 287, No. 3 Copyright 1998 by The American Society for Pharmacology and Experimental Therapeutics Printed in
0022-3565/98/2873-1068$03.00/0 THE JOURNAL OF PHARMACOLOGY AND EXPERIMENTAL THERAPEUTICS Vol. 287, No. 3 Copyright 1998 by The American Society for Pharmacology and Experimental Therapeutics Printed in
NMDA-Receptor Antagonists and Opioid Receptor Interactions as Related to Analgesia and Tolerance
 Vol. 19 No. 1(Suppl.) January 2000 Journal of Pain and Symptom Management S7 Proceedings Supplement NDMA-Receptor Antagonists: Evolving Role in Analgesia NMDA-Receptor Antagonists and Opioid Receptor Interactions
Vol. 19 No. 1(Suppl.) January 2000 Journal of Pain and Symptom Management S7 Proceedings Supplement NDMA-Receptor Antagonists: Evolving Role in Analgesia NMDA-Receptor Antagonists and Opioid Receptor Interactions
A comparison of e ects measured with isotonic and isometric recording: II. Concentration-e ect curves for physiological antagonists
 British Journal of Pharmacology (2001) 133, 1087 ± 1095 ã 2001 Nature Publishing Group All rights reserved 0007 ± 1188/01 $15.00 A comparison of e ects measured with isotonic and isometric recording: II.
British Journal of Pharmacology (2001) 133, 1087 ± 1095 ã 2001 Nature Publishing Group All rights reserved 0007 ± 1188/01 $15.00 A comparison of e ects measured with isotonic and isometric recording: II.
Blockade and reversal of spinal morphine tolerance by peptide and non-peptide calcitonin gene-related peptide receptor antagonists
 British Journal of Pharmacology (2000) 131, 875 ± 884 ã 2000 Macmillan Publishers Ltd All rights reserved 0007 ± 1188/00 $15.00 www.nature.com/bjp Blockade and reversal of spinal morphine tolerance by
British Journal of Pharmacology (2000) 131, 875 ± 884 ã 2000 Macmillan Publishers Ltd All rights reserved 0007 ± 1188/00 $15.00 www.nature.com/bjp Blockade and reversal of spinal morphine tolerance by
The Neurotransmitters
 The Neurotransmitters Thursday September 7, 2017 Notes By: Jeffrey Vocabulary Pharmacokinetics The study of the movement of administered substances within the body Psychopharmacology The study of how drugs
The Neurotransmitters Thursday September 7, 2017 Notes By: Jeffrey Vocabulary Pharmacokinetics The study of the movement of administered substances within the body Psychopharmacology The study of how drugs
TRAMADOL, A CENTRALLY ACTING OPIOID : ANTICONVULSANT EFFECT AGAINST MAXIMAL ELECTROSHOCK SEIZURE IN MICE
 Indian J Physiol Pharmacol 1998; 42 (3) : 407-411 TRAMADOL, A CENTRALLY ACTING OPIOID : ANTICONVULSANT EFFECT AGAINST MAXIMAL ELECTROSHOCK SEIZURE IN MICE ANSHU MANOCHA, KRISHNA K. SHARMA* AND PRAMOD K.
Indian J Physiol Pharmacol 1998; 42 (3) : 407-411 TRAMADOL, A CENTRALLY ACTING OPIOID : ANTICONVULSANT EFFECT AGAINST MAXIMAL ELECTROSHOCK SEIZURE IN MICE ANSHU MANOCHA, KRISHNA K. SHARMA* AND PRAMOD K.
Introduction SHORT COMMUNICATION. Irma V. Belozertseva Anton Yu. Bespalov
 Naunyn-Schmiedeberg s Arch Pharmacol (1998) 358:270 274 Springer-Verlag 1998 SHORT COMMUNICATION Irma V. Belozertseva Anton Yu. Bespalov Effects of NMDA receptor channel blockers, dizocilpine and memantine,
Naunyn-Schmiedeberg s Arch Pharmacol (1998) 358:270 274 Springer-Verlag 1998 SHORT COMMUNICATION Irma V. Belozertseva Anton Yu. Bespalov Effects of NMDA receptor channel blockers, dizocilpine and memantine,
INTERACTION DRUG BODY
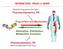 INTERACTION DRUG BODY What the drug does to the body What the body does to the drug Receptors - intracellular receptors - membrane receptors - Channel receptors - G protein-coupled receptors - Tyrosine-kinase
INTERACTION DRUG BODY What the drug does to the body What the body does to the drug Receptors - intracellular receptors - membrane receptors - Channel receptors - G protein-coupled receptors - Tyrosine-kinase
Spinal amino acid release and repeated withdrawal in spinal morphine tolerant rats
 British Journal of Pharmacology (2003) 138, 689 ± 697 ã 2003 Nature Publishing Group All rights reserved 0007 ± 1188/03 $25.00 Spinal amino acid release and repeated withdrawal in spinal morphine tolerant
British Journal of Pharmacology (2003) 138, 689 ± 697 ã 2003 Nature Publishing Group All rights reserved 0007 ± 1188/03 $25.00 Spinal amino acid release and repeated withdrawal in spinal morphine tolerant
Anesthesiology, V 92, No 3, Mar 2000
 786 Anesthesiology 2000; 92:786 94 2000 American Society of Anesthesiologists, Inc. Lippincott Williams & Wilkins, Inc. Complete Prevention but Stimulus-dependent Reversion of Morphine Tolerance by the
786 Anesthesiology 2000; 92:786 94 2000 American Society of Anesthesiologists, Inc. Lippincott Williams & Wilkins, Inc. Complete Prevention but Stimulus-dependent Reversion of Morphine Tolerance by the
PAIN & ANALGESIA. often accompanied by clinical depression. fibromyalgia, chronic fatigue, etc. COX 1, COX 2, and COX 3 (a variant of COX 1)
 Pain - subjective experience associated with detection of tissue damage ( nociception ) acute - serves as a warning chronic - nociception gone bad often accompanied by clinical depression fibromyalgia,
Pain - subjective experience associated with detection of tissue damage ( nociception ) acute - serves as a warning chronic - nociception gone bad often accompanied by clinical depression fibromyalgia,
EFFECT OF ANTIMUSCARINIC AGENTS ON THE CONTRACTILE
 Br. J. Pharmac. (1981), 73,829-835 EFFECT OF ANTIMUSCARINIC AGENTS ON THE CONTRACTILE RESPONSES TO CHOLINOMIMETICS IN THE RAT ANOCOCCYGEUS MUSCLE SHEILA A. DOGGRELL Department of Pharmacology & Clinical
Br. J. Pharmac. (1981), 73,829-835 EFFECT OF ANTIMUSCARINIC AGENTS ON THE CONTRACTILE RESPONSES TO CHOLINOMIMETICS IN THE RAT ANOCOCCYGEUS MUSCLE SHEILA A. DOGGRELL Department of Pharmacology & Clinical
Nervous System Communication. Nervous System Communication. The First Nerve Cells 1/2/11
 Nervous System Communication Nervous System Communication Process information Transfer information to other neurons Generate behavior and experience The First Nerve Cells Developed in primitive animals
Nervous System Communication Nervous System Communication Process information Transfer information to other neurons Generate behavior and experience The First Nerve Cells Developed in primitive animals
Special Issue on Pain and Itch
 Special Issue on Pain and Itch Title: Recent Progress in Understanding the Mechanisms of Pain and Itch Guest Editor of the Special Issue: Ru-Rong Ji, PhD Chronic pain is a major health problem world-wide.
Special Issue on Pain and Itch Title: Recent Progress in Understanding the Mechanisms of Pain and Itch Guest Editor of the Special Issue: Ru-Rong Ji, PhD Chronic pain is a major health problem world-wide.
Class 5 the neurotransmitters (drugs)
 Victoria September 7th 2017 Class 5 the neurotransmitters (drugs) psychopharmacology: study of the effects of a drug on behavior pharmacokinetics: study of the fate / movement of substances administered
Victoria September 7th 2017 Class 5 the neurotransmitters (drugs) psychopharmacology: study of the effects of a drug on behavior pharmacokinetics: study of the fate / movement of substances administered
Analgesic Drugs PHL-358-PHARMACOLOGY AND THERAPEUTICS-I. Mr.D.Raju,M.pharm, Lecturer
 Analgesic Drugs PHL-358-PHARMACOLOGY AND THERAPEUTICS-I Mr.D.Raju,M.pharm, Lecturer Mechanisms of Pain and Nociception Nociception is the mechanism whereby noxious peripheral stimuli are transmitted to
Analgesic Drugs PHL-358-PHARMACOLOGY AND THERAPEUTICS-I Mr.D.Raju,M.pharm, Lecturer Mechanisms of Pain and Nociception Nociception is the mechanism whereby noxious peripheral stimuli are transmitted to
In uence of the CB 1 receptor antagonist, AM 251, on the regional haemodynamic e ects of WIN or HU 210 in conscious rats
 British Journal of Pharmacology (2002) 136, 581 ± 587 ã 2002 Nature Publishing Group All rights reserved 0007 ± 1188/02 $25.00 www.nature.com/bjp In uence of the CB 1 receptor antagonist, AM 251, on the
British Journal of Pharmacology (2002) 136, 581 ± 587 ã 2002 Nature Publishing Group All rights reserved 0007 ± 1188/02 $25.00 www.nature.com/bjp In uence of the CB 1 receptor antagonist, AM 251, on the
Buprenorphine Blocks - and -Opioid Receptor-Mediated Antinociception in the Mouse
 0022-3565/03/3061-394 400$7.00 THE JOURNAL OF PHARMACOLOGY AND EXPERIMENTAL THERAPEUTICS Vol. 306, No. 1 Copyright 2003 by The American Society for Pharmacology and Experimental Therapeutics 48835/1076304
0022-3565/03/3061-394 400$7.00 THE JOURNAL OF PHARMACOLOGY AND EXPERIMENTAL THERAPEUTICS Vol. 306, No. 1 Copyright 2003 by The American Society for Pharmacology and Experimental Therapeutics 48835/1076304
Co-administration of calcium gluconate and magnesium acetate effectively blocks the signs of morphine withdrawal in mice
 Magnesium Research 212; 25 (1): 4-8 ORIGINAL ARTICLE Co-administration of calcium gluconate and magnesium acetate effectively blocks the signs of morphine withdrawal in mice Mohammed Rabbani, Valiollah
Magnesium Research 212; 25 (1): 4-8 ORIGINAL ARTICLE Co-administration of calcium gluconate and magnesium acetate effectively blocks the signs of morphine withdrawal in mice Mohammed Rabbani, Valiollah
Morphine analgesic tolerance in 129P3/J and 129S6/SvEv mice
 Pharmacology, Biochemistry and Behavior 85 (2006) 769 779 www.elsevier.com/locate/pharmbiochembeh Morphine analgesic tolerance in 129P3/J and 129S6/SvEv mice Camron D. Bryant a,b,c,, Kristofer W. Roberts
Pharmacology, Biochemistry and Behavior 85 (2006) 769 779 www.elsevier.com/locate/pharmbiochembeh Morphine analgesic tolerance in 129P3/J and 129S6/SvEv mice Camron D. Bryant a,b,c,, Kristofer W. Roberts
The effects of a new opioid analgesic, meptazinol, on the
 Br. J. Pharmac. (1985), 85, 25-211 The effects of a new opioid analgesic, meptazinol, on the respiration of the conscious rat I.S. Cowlrick' & N.B. Shepperson2 Wyeth Laboratories, Huntercombe Lane South,
Br. J. Pharmac. (1985), 85, 25-211 The effects of a new opioid analgesic, meptazinol, on the respiration of the conscious rat I.S. Cowlrick' & N.B. Shepperson2 Wyeth Laboratories, Huntercombe Lane South,
Pharmacological characterization of buprenorphine, a mixed agonist antagonist with k analgesia
 Brain Research 744 1997 41 46 Research report Pharmacological characterization of buprenorphine, a mixed agonist antagonist with k analgesia Chaim G. Pick a,), Yakov Peter a, Shaul Schreiber b, Ronit Weizman
Brain Research 744 1997 41 46 Research report Pharmacological characterization of buprenorphine, a mixed agonist antagonist with k analgesia Chaim G. Pick a,), Yakov Peter a, Shaul Schreiber b, Ronit Weizman
Non-opioidergic mechanism mediating morphine-induced. antianalgesia in the mouse spinal cord. Hsiang-En Wu, Jonathan Thompson, Han-Sen Sun,
 JPET This Fast article Forward. has not been Published copyedited on and formatted. March 3, The 2004 final version as DOI:10.1124/jpet.104.065334 may differ from this version. JPET #65334 Non-opioidergic
JPET This Fast article Forward. has not been Published copyedited on and formatted. March 3, The 2004 final version as DOI:10.1124/jpet.104.065334 may differ from this version. JPET #65334 Non-opioidergic
Overview of Pharmacodynamics. Psyc 472 Pharmacology of Psychoactive Drugs. Pharmacodynamics. Effects on Target Binding Site.
 Pharmacodynamics Overview of Pharmacodynamics Psychology 472: Pharmacology of Psychoactive Drugs Generally is defined as effects of drugs on a systems Can be associated with any system Neural, Heart, Liver,
Pharmacodynamics Overview of Pharmacodynamics Psychology 472: Pharmacology of Psychoactive Drugs Generally is defined as effects of drugs on a systems Can be associated with any system Neural, Heart, Liver,
2018 Learning Outcomes
 I. Pain Physiology and Anatomy (20%) A. Describe the basic anatomy of the nervous system. B. Describe the physiological mechanisms of neuronal function (eg- action potentials). C. Review the nociceptive
I. Pain Physiology and Anatomy (20%) A. Describe the basic anatomy of the nervous system. B. Describe the physiological mechanisms of neuronal function (eg- action potentials). C. Review the nociceptive
Mk-801 Administration in Adolescent Male Rats and Cocaine Conditioned Place
 Mk-801 Administration in Adolescent Male Rats and Cocaine Conditioned Place Preference Stephanie Willis, Jonnique Adjmul, Shabaaz Sandhu, Antoniette M. Maldonado-Devincci, Cheryl Kirsten ABSTRACT The present
Mk-801 Administration in Adolescent Male Rats and Cocaine Conditioned Place Preference Stephanie Willis, Jonnique Adjmul, Shabaaz Sandhu, Antoniette M. Maldonado-Devincci, Cheryl Kirsten ABSTRACT The present
International Journal of Cell Cloning 6: (1988) Research Laboratory of Blood Physiology, Hunan Medical College, Changsha, Hunan, PRC
 Original Paper International Journal of Cell Cloning 6:290-295 (1988) The Influence of Histamine at Various Concentrations on the Cell Cycle State of Hematopoietic Stem Cells (CF'U-s) I"i Shounan, XU You-Heng
Original Paper International Journal of Cell Cloning 6:290-295 (1988) The Influence of Histamine at Various Concentrations on the Cell Cycle State of Hematopoietic Stem Cells (CF'U-s) I"i Shounan, XU You-Heng
A novel role for vitamin D: modulation of expression and function of the local renin angiotensin system in mouse pancreatic islets
 Diabetologia () 5:77 DOI.7/s5--- SHORT COMMUNICATION A novel role for vitamin D: modulation of expression and function of the local renin angiotensin system in mouse pancreatic islets Q. Cheng & Y. C.
Diabetologia () 5:77 DOI.7/s5--- SHORT COMMUNICATION A novel role for vitamin D: modulation of expression and function of the local renin angiotensin system in mouse pancreatic islets Q. Cheng & Y. C.
Gastric, intestinal and colonic absorption of metoprolol in
 Br. J. clin. Pharmac. (1985), 19, 85S-89S Gastric, intestinal and colonic absorption of metoprolol in the rat J. DOMENECH', M. ALBA', J. M. MORERA', R. OBACH' & J. M. PLA DELFINA2 'Department of Pharmaceutics,
Br. J. clin. Pharmac. (1985), 19, 85S-89S Gastric, intestinal and colonic absorption of metoprolol in the rat J. DOMENECH', M. ALBA', J. M. MORERA', R. OBACH' & J. M. PLA DELFINA2 'Department of Pharmaceutics,
Zinc and water intake in rats: investigation of adrenergic and opiatergic central mechanisms
 Brazilian Journal of Medical and Biological Research (1999) 32: 1217-1222 Opioid and zinc interplay in isoproterenol-induced drinking ISSN 0100-879X Short Communication 1217 Zinc and water intake in rats:
Brazilian Journal of Medical and Biological Research (1999) 32: 1217-1222 Opioid and zinc interplay in isoproterenol-induced drinking ISSN 0100-879X Short Communication 1217 Zinc and water intake in rats:
Basics of Pharmacology
 Basics of Pharmacology Pekka Rauhala Transmed 2013 What is pharmacology? Pharmacology may be defined as the study of the effects of drugs on the function of living systems Pharmacodynamics The mechanism(s)
Basics of Pharmacology Pekka Rauhala Transmed 2013 What is pharmacology? Pharmacology may be defined as the study of the effects of drugs on the function of living systems Pharmacodynamics The mechanism(s)
Prevention of Development of Tolerance and Dependence to Opiate in Mice by BR-16A (Mentat) A Herbal Psychotropic Preparation
 [Indian Journal of Experimental Biology (1992): (30), 885] Prevention of Development of Tolerance and Dependence to Opiate in Mice by BR-16A (Mentat) A Herbal Psychotropic Preparation Kulkarni, S.K. and
[Indian Journal of Experimental Biology (1992): (30), 885] Prevention of Development of Tolerance and Dependence to Opiate in Mice by BR-16A (Mentat) A Herbal Psychotropic Preparation Kulkarni, S.K. and
Receptor Sites and Drug Design
 Receptor Sites and Drug Design Case Study: piates use for Anesthesia & analgesia 1 PAI 2 The functional groups and their placement in three-dimensional space determines to a large degree a molecule s biological
Receptor Sites and Drug Design Case Study: piates use for Anesthesia & analgesia 1 PAI 2 The functional groups and their placement in three-dimensional space determines to a large degree a molecule s biological
The Use of Analgesics in Rodents and Rabbits Deborah M. Mook, DVM
 The Use of Analgesics in Rodents and Rabbits Deborah M. Mook, DVM (Last updated August, 2005) Rationale. Well-established studies have shown repeatedly that effective analgesia enhances locomotion, increases
The Use of Analgesics in Rodents and Rabbits Deborah M. Mook, DVM (Last updated August, 2005) Rationale. Well-established studies have shown repeatedly that effective analgesia enhances locomotion, increases
Intrathecal Infusion Therapy for Chronic Pain: Challenges, Lessons and Opportunities
 June 14-15, 2016 Mult imodal Therapies for Brain Disorders: A Workshop Hosted by: Forum on Neuroscience and Nervous System Disorders Intrathecal Infusion Therapy for Chronic Pain: Challenges, Lessons and
June 14-15, 2016 Mult imodal Therapies for Brain Disorders: A Workshop Hosted by: Forum on Neuroscience and Nervous System Disorders Intrathecal Infusion Therapy for Chronic Pain: Challenges, Lessons and
Section: Chapter 5: Multiple Choice. 1. The structure of synapses is best viewed with a(n):
 Section: Chapter 5: Multiple Choice 1. The structure of synapses is best viewed with a(n): p.155 electron microscope. light microscope. confocal microscope. nissle-stained microscopic procedure. 2. Electron
Section: Chapter 5: Multiple Choice 1. The structure of synapses is best viewed with a(n): p.155 electron microscope. light microscope. confocal microscope. nissle-stained microscopic procedure. 2. Electron
d-methadone Is Antinociceptive in the Rat Formalin Test 1
 0022-3565/97/2832-0648$03.00/0 THE JOURNAL OF PHARMACOLOGY AND EXPERIMENTAL THERAPEUTICS Vol. 283, No. 2 Copyright 1997 by The American Society for Pharmacology and Experimental Therapeutics Printed in
0022-3565/97/2832-0648$03.00/0 THE JOURNAL OF PHARMACOLOGY AND EXPERIMENTAL THERAPEUTICS Vol. 283, No. 2 Copyright 1997 by The American Society for Pharmacology and Experimental Therapeutics Printed in
Tsutomu Fujimura, Kimie Murayama, Shunsuke Kawamura, Takumi Sato, Chikai Sakurada,
 JPET Fast This Forward. article has not Published been copyedited on and November formatted. The 23, final 2004 version as may DOI:10.1124/jpet.104.075697 differ from this version. JPET #75697 Title A
JPET Fast This Forward. article has not Published been copyedited on and November formatted. The 23, final 2004 version as may DOI:10.1124/jpet.104.075697 differ from this version. JPET #75697 Title A
9.98 Neuropharmacology January (IAP) 2009
 MIT OpenCourseWare http://ocw.mit.edu 9.98 Neuropharmacology January (IAP) 2009 For information about citing these materials or our Terms of Use, visit: http://ocw.mit.edu/terms. Neuropharmacology: The
MIT OpenCourseWare http://ocw.mit.edu 9.98 Neuropharmacology January (IAP) 2009 For information about citing these materials or our Terms of Use, visit: http://ocw.mit.edu/terms. Neuropharmacology: The
The peripheral antinociceptive e ect induced by morphine is associated with ATP-sensitive K + channels
 (2000) 129, 110 ± 114 ã 2000 Macmillan Publishers Ltd All rights reserved 0007 ± 1188/00 $15.00 www.nature.com/bjp The peripheral antinociceptive e ect induced by morphine is associated with ATP-sensitive
(2000) 129, 110 ± 114 ã 2000 Macmillan Publishers Ltd All rights reserved 0007 ± 1188/00 $15.00 www.nature.com/bjp The peripheral antinociceptive e ect induced by morphine is associated with ATP-sensitive
Acute Pain Management in the Opioid Tolerant Patient. Objectives. Opioids. The participant will be able to define opioid tolerance
 Acute Pain Management in the Opioid Tolerant Patient Kathleen M. Colfer, MSN, RN-BC Clinical Nurse Specialist Acute Pain Management Service Department of Anesthesiology Thomas Jefferson University Hospital
Acute Pain Management in the Opioid Tolerant Patient Kathleen M. Colfer, MSN, RN-BC Clinical Nurse Specialist Acute Pain Management Service Department of Anesthesiology Thomas Jefferson University Hospital
Opioid Receptor Trafficking in Pain States
 Encyclopedia of Pain Springer-Verlag Berlin Heidelberg 2007 10.1007/978-3-540-29805-2_2962 Robert F. Schmidt and William D. Willis Opioid Receptor Trafficking in Pain States Catherine M. Cahill 3, Anne
Encyclopedia of Pain Springer-Verlag Berlin Heidelberg 2007 10.1007/978-3-540-29805-2_2962 Robert F. Schmidt and William D. Willis Opioid Receptor Trafficking in Pain States Catherine M. Cahill 3, Anne
Role of ion channel modifiers in reversal of morphine induced gastrointestinal inertia by prokinetic agents in mice
 Indian Journal of Experimental Biology Vol. 46, January 2008, pp. 60-65 Role of ion channel modifiers in reversal of morphine induced gastrointestinal inertia by prokinetic agents in mice Selvarajan Sandhiya,
Indian Journal of Experimental Biology Vol. 46, January 2008, pp. 60-65 Role of ion channel modifiers in reversal of morphine induced gastrointestinal inertia by prokinetic agents in mice Selvarajan Sandhiya,
Analgesics, pain and tolerance: The St John s discussion
 EDITORIAL Analgesics, pain and tolerance: The St John s discussion Four of the articles in the present issue of Pain Research & Management discuss preclinical issues relevant to understanding the changes
EDITORIAL Analgesics, pain and tolerance: The St John s discussion Four of the articles in the present issue of Pain Research & Management discuss preclinical issues relevant to understanding the changes
Review of Neurochemistry What are neurotransmitters?
 Review of Neurochemistry What are neurotransmitters? In molecular terms, neurotransmitters are molecules that ( ) and of neurons by, for example, increasing or decreasing enzymatic activity or altering
Review of Neurochemistry What are neurotransmitters? In molecular terms, neurotransmitters are molecules that ( ) and of neurons by, for example, increasing or decreasing enzymatic activity or altering
A Comparative study of the Anti convulsant effect of Nimodipine and Ketamine combination with standard anticonvulsant drug in Rodents
 Original Article A Comparative study of the Anti convulsant effect of Nimodipine and Ketamine combination with standard anticonvulsant drug in Rodents Prasanand S 1, Pushpalatha C 2, Mohsin MD 3, Sam Pavan
Original Article A Comparative study of the Anti convulsant effect of Nimodipine and Ketamine combination with standard anticonvulsant drug in Rodents Prasanand S 1, Pushpalatha C 2, Mohsin MD 3, Sam Pavan
Journal of Chemical and Pharmaceutical Research
 Available online www.jocpr.com Journal of Chemical and Pharmaceutical Research ISSN No: 0975-7384 CODEN(USA): JCPRC5 J. Chem. Pharm. Res., 2011, 3(5):468-472 Study on the anticonvulsant activity of Pentazocine
Available online www.jocpr.com Journal of Chemical and Pharmaceutical Research ISSN No: 0975-7384 CODEN(USA): JCPRC5 J. Chem. Pharm. Res., 2011, 3(5):468-472 Study on the anticonvulsant activity of Pentazocine
Science & Technologies COMPARISON THE EFFECTS OF TACRINE AND GALANTAMINE ON ACTIVE AVOIDANCE TEST IN RATS WITH DIAZEPAM-AMNESIA MODEL
 COMPARISON THE EFFECTS OF TACRINE AND GALANTAMINE ON ACTIVE AVOIDANCE TEST IN RATS WITH DIAZEPAM-AMNESIA MODEL Darinka Dimitrova, Damianka Getova Medical University Plovdiv, Medical Faculty, 4002 Plovdiv,
COMPARISON THE EFFECTS OF TACRINE AND GALANTAMINE ON ACTIVE AVOIDANCE TEST IN RATS WITH DIAZEPAM-AMNESIA MODEL Darinka Dimitrova, Damianka Getova Medical University Plovdiv, Medical Faculty, 4002 Plovdiv,
What are the 6 types of neuroglia and their functions?!
 Warm Up! Take out your 11C Notes What are the 6 types of neuroglia and their functions?! Astrocytes Microglia Ependymal Cells Satellite Cells Schwann Cells Oligodendrocytes Support, brace, & nutrient transfer
Warm Up! Take out your 11C Notes What are the 6 types of neuroglia and their functions?! Astrocytes Microglia Ependymal Cells Satellite Cells Schwann Cells Oligodendrocytes Support, brace, & nutrient transfer
Suven Microdialysis Services
 Suven Microdialysis Services In-Vivo Brain Microdialysis Studies in Rodents for Monitoring Changes in Neurotransmitters Acetylcholine Histamine and Metabolite GABA and Glutamate Monoamines (NE, DA, 5HT
Suven Microdialysis Services In-Vivo Brain Microdialysis Studies in Rodents for Monitoring Changes in Neurotransmitters Acetylcholine Histamine and Metabolite GABA and Glutamate Monoamines (NE, DA, 5HT
C81ADD Psychology of Addiction. Alcohol. Ethyl alcohol (ethanol) School of Psychology. Tobias Bast.
 C81ADD Psychology of Addiction Alcohol Ethyl alcohol (ethanol) Tobias Bast School of Psychology tobias.bast@nottingham.ac.uk 1 Selected aspects of the psychopharmacology of alcohol (ethanol) Primary neuropharmacological
C81ADD Psychology of Addiction Alcohol Ethyl alcohol (ethanol) Tobias Bast School of Psychology tobias.bast@nottingham.ac.uk 1 Selected aspects of the psychopharmacology of alcohol (ethanol) Primary neuropharmacological
THE OPIUM POPPY OPIOID PHARMACOLOGY 2/18/16. PCTH 300/305 Andrew Horne, PhD MEDC 309. Papaver somniferum. Poppy Seeds Opiates
 OPIOID PHARMACOLOGY PCTH 300/305 Andrew Horne, PhD andrew.horne@ubc.ca MEDC 309 THE OPIUM POPPY Papaver somniferum Sleep-bringing poppy Poppy Seeds Opiates Opium Poppy Straw 1 OPIATES VS. OPIOIDS Opiates:
OPIOID PHARMACOLOGY PCTH 300/305 Andrew Horne, PhD andrew.horne@ubc.ca MEDC 309 THE OPIUM POPPY Papaver somniferum Sleep-bringing poppy Poppy Seeds Opiates Opium Poppy Straw 1 OPIATES VS. OPIOIDS Opiates:
Myocardial Opiate Receptors
 Gen. Physiol. Biophys. 1982, 1, 447-452 Myocardial Opiate Receptors M. E. SAXON, G. R. IVANITSKY, F. F. BELOYARTSEV, V. G. SAFRONOVA, YU. M. KOKOZ and A. A. FREYDIN Institute of Biological Physics, Academy
Gen. Physiol. Biophys. 1982, 1, 447-452 Myocardial Opiate Receptors M. E. SAXON, G. R. IVANITSKY, F. F. BELOYARTSEV, V. G. SAFRONOVA, YU. M. KOKOZ and A. A. FREYDIN Institute of Biological Physics, Academy
JUHANA J. IDÄNPÄÄN-HEIKKILÄ, GISÈLE GUILBAUD and VALÉRIE KAYSER
 0022-3565/97/2823-1366$03.00/0 THE JOURNAL OF PHARMACOLOGY AND EXPERIMENTAL THERAPEUTICS Vol. 282, No. 3 Copyright 1997 by The American Society for Pharmacology and Experimental Therapeutics Printed in
0022-3565/97/2823-1366$03.00/0 THE JOURNAL OF PHARMACOLOGY AND EXPERIMENTAL THERAPEUTICS Vol. 282, No. 3 Copyright 1997 by The American Society for Pharmacology and Experimental Therapeutics Printed in
Local administration of 9 -tetrahydrocannabinol attenuates capsaicin-induced thermal nociception in rhesus monkeys: a peripheral cannabinoid action
 Psychopharmacology (1999) 143:322 326 Springer-Verlag 1999 RAPID COMMUNICATION Mei-Chuan Ko James H. Woods Local administration of 9 -tetrahydrocannabinol attenuates capsaicin-induced thermal nociception
Psychopharmacology (1999) 143:322 326 Springer-Verlag 1999 RAPID COMMUNICATION Mei-Chuan Ko James H. Woods Local administration of 9 -tetrahydrocannabinol attenuates capsaicin-induced thermal nociception
38 Noble ] mm. Acta Physiologica Sinica Tsinghua Tongfang Optical Disc Co., Ltd. All rights reserved. ; icv M ; icv H 1 (250 , NT
![38 Noble ] mm. Acta Physiologica Sinica Tsinghua Tongfang Optical Disc Co., Ltd. All rights reserved. ; icv M ; icv H 1 (250 , NT 38 Noble ] mm. Acta Physiologica Sinica Tsinghua Tongfang Optical Disc Co., Ltd. All rights reserved. ; icv M ; icv H 1 (250 , NT](/thumbs/91/105226125.jpg) 140, 1999 4, 51 (2), 140 146 Acta Physiologica Sinica 1998202219 1998206205 (, 210029) Sprague2Dawley, (112 g/ kg) (icv) (NT) (10, 20 g), icv 1 (115 g/ 3 l), NT ; icv M (215 g/ 3 l), NT ; icv H 1 (50 g/
140, 1999 4, 51 (2), 140 146 Acta Physiologica Sinica 1998202219 1998206205 (, 210029) Sprague2Dawley, (112 g/ kg) (icv) (NT) (10, 20 g), icv 1 (115 g/ 3 l), NT ; icv M (215 g/ 3 l), NT ; icv H 1 (50 g/
Role of camp-dependent Protein Kinase (PKA) in Opioid Agonist-Induced -Opioid Receptor Downregulation and Tolerance in Mice
 SYNAPSE 38:322 327 (2000) Role of camp-dependent Protein Kinase (PKA) in Opioid Agonist-Induced -Opioid Receptor Downregulation and Tolerance in Mice JI SHEN, A. BENEDICT GOMES, ANNEMARIE GALLAGHER, KRISTI
SYNAPSE 38:322 327 (2000) Role of camp-dependent Protein Kinase (PKA) in Opioid Agonist-Induced -Opioid Receptor Downregulation and Tolerance in Mice JI SHEN, A. BENEDICT GOMES, ANNEMARIE GALLAGHER, KRISTI
Abuse Potential of Morphine/ Dextromethorphan Combinations
 S26 Journal of Pain and Symptom Management Vol. 19 No. 1(Suppl.) January 2000 Proceedings Supplement NMDA-Receptor Antagonists: Evolving Role in Analgesia Abuse Potential of Morphine/ Dextromethorphan
S26 Journal of Pain and Symptom Management Vol. 19 No. 1(Suppl.) January 2000 Proceedings Supplement NMDA-Receptor Antagonists: Evolving Role in Analgesia Abuse Potential of Morphine/ Dextromethorphan
Insulin Attenuates Formalin-Induced Nociceptive Response in Mice through a Mechanism that Is Deranged by Diabetes Mellitus
 0022-3565/97/2811-0315$03.00/0 THE JOURNAL OF PHARMACOLOGY AND EXPERIMENTAL THERAPEUTICS Vol. 281, No. 1 Copyright 1997 by The American Society for Pharmacology and Experimental Therapeutics Printed in
0022-3565/97/2811-0315$03.00/0 THE JOURNAL OF PHARMACOLOGY AND EXPERIMENTAL THERAPEUTICS Vol. 281, No. 1 Copyright 1997 by The American Society for Pharmacology and Experimental Therapeutics Printed in
Ligand-Gated Ion Channels
 Ligand-Gated Ion Channels The Other Machines That Make It Possible... Topics I Introduction & Electrochemical Gradients Passive Membrane Properties Action Potentials Voltage-Gated Ion Channels Topics II
Ligand-Gated Ion Channels The Other Machines That Make It Possible... Topics I Introduction & Electrochemical Gradients Passive Membrane Properties Action Potentials Voltage-Gated Ion Channels Topics II
Action Potentials and Synaptic Transmission. BIO 219 Napa Valley College Dr. Adam Ross
 Action Potentials and Synaptic Transmission BIO 219 Napa Valley College Dr. Adam Ross Review of action potentials Nodes of Ranvier Nucleus Dendrites Cell body In saltatory conduction, the nerve impulses
Action Potentials and Synaptic Transmission BIO 219 Napa Valley College Dr. Adam Ross Review of action potentials Nodes of Ranvier Nucleus Dendrites Cell body In saltatory conduction, the nerve impulses
Cholecystokinin as a factor in the enhanced potency of spinal morphine following carrageenin inflammation
 Br. J. Pharmacol. (1993), 18, 967-973 191" Macmillan Press Ltd, 1993 Cholecystokinin as a factor in the enhanced potency of spinal morphine following carrageenin inflammation 'Louise C. Stanfa & Anthony
Br. J. Pharmacol. (1993), 18, 967-973 191" Macmillan Press Ltd, 1993 Cholecystokinin as a factor in the enhanced potency of spinal morphine following carrageenin inflammation 'Louise C. Stanfa & Anthony
Pharmacology of intravenous induction agents
 Pharmacology of intravenous induction agents Ákos Csomós MD, PhD Professor, Head of Department Medical Centre, Hungarian Defence Force, Budapest What do we have in the market? Thiopental Metohexital Etomidate
Pharmacology of intravenous induction agents Ákos Csomós MD, PhD Professor, Head of Department Medical Centre, Hungarian Defence Force, Budapest What do we have in the market? Thiopental Metohexital Etomidate
Case Study: Opiates use for Anesthesia & analgesia
 rganic Lecture Series Receptor Sites and Drug Design Case Study: piates use for Anesthesia & analgesia 52 rganic Lecture Series PAI 53 Drug interactions at the cellular level rganic Lecture Series The
rganic Lecture Series Receptor Sites and Drug Design Case Study: piates use for Anesthesia & analgesia 52 rganic Lecture Series PAI 53 Drug interactions at the cellular level rganic Lecture Series The
Section Coordinator: Jerome W. Breslin, PhD, Assistant Professor of Physiology, MEB 7208, ,
 IDP Biological Systems Gastrointestinal System Section Coordinator: Jerome W. Breslin, PhD, Assistant Professor of Physiology, MEB 7208, 504-568-2669, jbresl@lsuhsc.edu Overall Learning Objectives 1. Characterize
IDP Biological Systems Gastrointestinal System Section Coordinator: Jerome W. Breslin, PhD, Assistant Professor of Physiology, MEB 7208, 504-568-2669, jbresl@lsuhsc.edu Overall Learning Objectives 1. Characterize
MEDICAL POLICY SUBJECT: KETAMINE INFUSION THERAPY FOR THE TREATMENT OF CHRONIC PAIN SYNDROMES POLICY NUMBER: CATEGORY: Technology Assessment
 Clinical criteria used to make utilization review decisions are based on credible scientific evidence published in peer reviewed medical literature generally recognized by the medical community. Guidelines
Clinical criteria used to make utilization review decisions are based on credible scientific evidence published in peer reviewed medical literature generally recognized by the medical community. Guidelines
PHRM20001: Pharmacology - How Drugs Work!
 PHRM20001: Pharmacology - How Drugs Work Drug: a chemical that affects physiological function in a specific way. Endogenous substances: hormones, neurotransmitters, antibodies, genes. Exogenous substances:
PHRM20001: Pharmacology - How Drugs Work Drug: a chemical that affects physiological function in a specific way. Endogenous substances: hormones, neurotransmitters, antibodies, genes. Exogenous substances:
Neurotransmitter Systems II Receptors. Reading: BCP Chapter 6
 Neurotransmitter Systems II Receptors Reading: BCP Chapter 6 Neurotransmitter Systems Normal function of the human brain requires an orderly set of chemical reactions. Some of the most important chemical
Neurotransmitter Systems II Receptors Reading: BCP Chapter 6 Neurotransmitter Systems Normal function of the human brain requires an orderly set of chemical reactions. Some of the most important chemical
agonistic Summation: additive Potentiation synergism :
 25 Two common types of agonistic drug interactions are : 1. Summation: When two drugs with similar mechanisms are given together, they typically produce additive effects. 2. Potentiation or synergism :
25 Two common types of agonistic drug interactions are : 1. Summation: When two drugs with similar mechanisms are given together, they typically produce additive effects. 2. Potentiation or synergism :
Extended therapeutic validation of an anti-mc4 receptor antibody in an animal model of anorexia nervosa
 Extended therapeutic validation of an anti-mc4 receptor antibody in an animal model of anorexia nervosa (project no. 04-12B) Authors Guus Akkermans, Martien Kas Preliminary data report Introduction In
Extended therapeutic validation of an anti-mc4 receptor antibody in an animal model of anorexia nervosa (project no. 04-12B) Authors Guus Akkermans, Martien Kas Preliminary data report Introduction In
Brief Communication. The Journal of Neuroscience, March 23, (12):
 The Journal of Neuroscience, March 23, 2005 25(12):3229 3233 3229 Brief Communication In Vivo Activation of a Mutant -Opioid Receptor by Naltrexone Produces a Potent Analgesic Effect But No Tolerance:
The Journal of Neuroscience, March 23, 2005 25(12):3229 3233 3229 Brief Communication In Vivo Activation of a Mutant -Opioid Receptor by Naltrexone Produces a Potent Analgesic Effect But No Tolerance:
Close to site of release (at synapse); binds to receptors in
 Chapter 18: The Endocrine System Chemical Messengers 1. Neural 2. Endocrine 3. Neuroendocrine 4. Paracrine 5. Autocrine Endocrine System --Endocrine and nervous systems work together --Endocrine vs. Nervous
Chapter 18: The Endocrine System Chemical Messengers 1. Neural 2. Endocrine 3. Neuroendocrine 4. Paracrine 5. Autocrine Endocrine System --Endocrine and nervous systems work together --Endocrine vs. Nervous
General principles of gastrointestinal motility
 General principles of gastrointestinal motility OBJECTIVES Physiological anatomy General Principles Circulation of blood through the GIT organs Control of all GIT functions by local, nervous, and hormonal
General principles of gastrointestinal motility OBJECTIVES Physiological anatomy General Principles Circulation of blood through the GIT organs Control of all GIT functions by local, nervous, and hormonal
Graduate School of Acupuncture and Moxibustion, Meiji University of Oriental Medicine , Japan
 The American Journal of Chinese Medicine, Vol. 32, No. 2, 269 279 2004 World Scientific Publishing Company Institute for Advanced Research in Asian Science and Medicine Corticotropin-Releasing Factor and
The American Journal of Chinese Medicine, Vol. 32, No. 2, 269 279 2004 World Scientific Publishing Company Institute for Advanced Research in Asian Science and Medicine Corticotropin-Releasing Factor and
Neurochemical mechanisms of stimulation-produced analgesia: Comparison of tests involving tonic and phasic pain
 Physiological Psychology 1984, Vol. 12 (4), 280-284 Neurochemical mechanisms of stimulation-produced analgesia: Comparison of tests involving tonic and phasic pain BEVERLY E. THORN and KENNETH E. PLOTKIN
Physiological Psychology 1984, Vol. 12 (4), 280-284 Neurochemical mechanisms of stimulation-produced analgesia: Comparison of tests involving tonic and phasic pain BEVERLY E. THORN and KENNETH E. PLOTKIN
ALVIMOPAN 0.0 OVERVIEW
 ALVIMOPAN 0.0 OVERVIEW A. Alvimopan is a peripherally restricted mu-opioid receptor antagonist. B. DOSING INFORMATION : For the treatment of opioid bowel dysfunction, oral alvimopan doses between 0.5 milligrams
ALVIMOPAN 0.0 OVERVIEW A. Alvimopan is a peripherally restricted mu-opioid receptor antagonist. B. DOSING INFORMATION : For the treatment of opioid bowel dysfunction, oral alvimopan doses between 0.5 milligrams
DBL NALOXONE HYDROCHLORIDE INJECTION USP
 Name of medicine Naloxone hydrochloride Data Sheet New Zealand DBL NALXNE HYDRCHLRIDE INJECTIN USP Presentation DBL Naloxone Hydrochloride Injection USP is a sterile, clear, colourless solution, free from
Name of medicine Naloxone hydrochloride Data Sheet New Zealand DBL NALXNE HYDRCHLRIDE INJECTIN USP Presentation DBL Naloxone Hydrochloride Injection USP is a sterile, clear, colourless solution, free from
E ect of chronic clonidine treatment on transmitter release from sympathetic varicosities of the guinea-pig vas deferens
 British Journal of Pharmacology (2001) 134, 1480 ± 1486 ã 2001 Nature Publishing Group All rights reserved 0007 ± 1188/01 $15.00 E ect of chronic clonidine treatment on transmitter release from sympathetic
British Journal of Pharmacology (2001) 134, 1480 ± 1486 ã 2001 Nature Publishing Group All rights reserved 0007 ± 1188/01 $15.00 E ect of chronic clonidine treatment on transmitter release from sympathetic
Fundamentals of the Nervous System and Nervous Tissue: Part C
 PowerPoint Lecture Slides prepared by Janice Meeking, Mount Royal College C H A P T E R 11 Fundamentals of the Nervous System and Nervous Tissue: Part C Warm Up What is a neurotransmitter? What is the
PowerPoint Lecture Slides prepared by Janice Meeking, Mount Royal College C H A P T E R 11 Fundamentals of the Nervous System and Nervous Tissue: Part C Warm Up What is a neurotransmitter? What is the
University of Buea. Faculty of Health Sciences. Programme in Medicine
 Faculty of Health Sciences University of Buea Wednesday, 28 th January 2009 Time: 8 00-10 00 Programme in Medicine MED 303 (Gastrointestinal Physiology) EXAMS (2008-2009) Identify the letter of the choice
Faculty of Health Sciences University of Buea Wednesday, 28 th January 2009 Time: 8 00-10 00 Programme in Medicine MED 303 (Gastrointestinal Physiology) EXAMS (2008-2009) Identify the letter of the choice
NEURONS COMMUNICATE WITH OTHER CELLS AT SYNAPSES 34.3
 NEURONS COMMUNICATE WITH OTHER CELLS AT SYNAPSES 34.3 NEURONS COMMUNICATE WITH OTHER CELLS AT SYNAPSES Neurons communicate with other neurons or target cells at synapses. Chemical synapse: a very narrow
NEURONS COMMUNICATE WITH OTHER CELLS AT SYNAPSES 34.3 NEURONS COMMUNICATE WITH OTHER CELLS AT SYNAPSES Neurons communicate with other neurons or target cells at synapses. Chemical synapse: a very narrow
Goals for sedation during mechanical ventilation
 New Uses of Old Medications Gina Riggi, PharmD, BCCCP, BCPS Clinical Pharmacist Trauma ICU Jackson Memorial Hospital Disclosure I do not have anything to disclose Objectives Describe the use of ketamine
New Uses of Old Medications Gina Riggi, PharmD, BCCCP, BCPS Clinical Pharmacist Trauma ICU Jackson Memorial Hospital Disclosure I do not have anything to disclose Objectives Describe the use of ketamine
Leon F. Tseng* Keywords: Endomorphin-1, Endomorphin-2, Antinociception, -Opioid receptor, Descending pain control system
 Jpn. J. Pharmacol. 89, 216 220 (2002) FORUM MINIREVIEW Recent Advances in the Search for the -Opioidergic System The Antinociceptive Properties of Endomorphin-1 and Endomorphin-2 in the Mouse Leon F. Tseng*
Jpn. J. Pharmacol. 89, 216 220 (2002) FORUM MINIREVIEW Recent Advances in the Search for the -Opioidergic System The Antinociceptive Properties of Endomorphin-1 and Endomorphin-2 in the Mouse Leon F. Tseng*
SUPPLEMENTARY MATERIAL. Sample preparation for light microscopy
 SUPPLEMENTARY MATERIAL Sample preparation for light microscopy To characterize the granulocytes and melanomacrophage centers, cross sections were prepared for light microscopy, as described in Material
SUPPLEMENTARY MATERIAL Sample preparation for light microscopy To characterize the granulocytes and melanomacrophage centers, cross sections were prepared for light microscopy, as described in Material
THE EFFECT OF THE CHOLINERGIC SYSTEM ON PLANTAR PAIN RESPONSE IN RATS
 ARTICLE THE EFFECT OF THE CHOLINERGIC SYSTEM ON PLANTAR PAIN RESPONSE IN RATS Mohammad Radmehr 1, Mohammad Pourahmadi 2, Hossein Kargar Jahromi 3, Nazanin Shafeie 4, Saeid Mahmoudi Teimourabad 5, Ali Eslamifar
ARTICLE THE EFFECT OF THE CHOLINERGIC SYSTEM ON PLANTAR PAIN RESPONSE IN RATS Mohammad Radmehr 1, Mohammad Pourahmadi 2, Hossein Kargar Jahromi 3, Nazanin Shafeie 4, Saeid Mahmoudi Teimourabad 5, Ali Eslamifar
A NEW TYPE OF DRUG ENHANCEMENT: INCREASED MAXIMUM RESPONSE TO CUMULATIVE NORADREN- ALINE IN THE ISOLATED RAT VAS DEFERENS
 Br. J. Pharmac. Chemother. (1968), 33, 171-176. A NEW TYPE OF DRUG ENHANCEMENT: NCREASED MAXMUM RESPONSE TO CUMULATVE NORADREN- ALNE N THE SOLATED RAT VAS DEFERENS BY A. BARNETT, D. D. GREENHOUSE AND R..
Br. J. Pharmac. Chemother. (1968), 33, 171-176. A NEW TYPE OF DRUG ENHANCEMENT: NCREASED MAXMUM RESPONSE TO CUMULATVE NORADREN- ALNE N THE SOLATED RAT VAS DEFERENS BY A. BARNETT, D. D. GREENHOUSE AND R..
D. Nishizawa 1, N. Gajya 2 and K. Ikeda 1, * Global Research & Development, Nagoya Laboratories, Pfizer Japan Inc, Nagoya, Japan
 Current Neuropharmacology, 2011, 9, 113-117 113 Identification of Selective Agonists and Antagonists to G Protein-Activated Inwardly Rectifying Potassium Channels: Candidate Medicines for Drug Dependence
Current Neuropharmacology, 2011, 9, 113-117 113 Identification of Selective Agonists and Antagonists to G Protein-Activated Inwardly Rectifying Potassium Channels: Candidate Medicines for Drug Dependence
Chapter 24 Chemical Communications Neurotransmitters & Hormones
 Chapter 24 Chemical Communications Neurotransmitters & Hormones 1 Chemical Communication Terms and definitions: Neuron: A nerve cell. Neurotransmitter: A chemical messenger between a neuron and another
Chapter 24 Chemical Communications Neurotransmitters & Hormones 1 Chemical Communication Terms and definitions: Neuron: A nerve cell. Neurotransmitter: A chemical messenger between a neuron and another
Abbott s TRPV1 Antagonist Program
 Abbott s TRPV1 Antagonist Program Connie R. Faltynek, Ph.D. Sr. Director, Neuroscience and Pain Discovery American Pain Society Annual Meeting Basic Science Dinner Symposium May 9, 2008 Early History of
Abbott s TRPV1 Antagonist Program Connie R. Faltynek, Ph.D. Sr. Director, Neuroscience and Pain Discovery American Pain Society Annual Meeting Basic Science Dinner Symposium May 9, 2008 Early History of
Minimal conditions for the creation of a unit relationship: the social bond between birthdaymates
 European Journal of Social Psychology Eur. J. Soc. Psychol. 28, 475±481 (1998) Minimal conditions for the creation of a unit relationship: the social bond between birthdaymates DALE T. MILLER*, JULIE S.
European Journal of Social Psychology Eur. J. Soc. Psychol. 28, 475±481 (1998) Minimal conditions for the creation of a unit relationship: the social bond between birthdaymates DALE T. MILLER*, JULIE S.
Effects of a Novel Fentanyl Derivative on Drug Discrimination and Learning in Rhesus Monkeys
 PII S0091-3057(99)00058-1 Pharmacology Biochemistry and Behavior, Vol. 64, No. 2, pp. 367 371, 1999 1999 Elsevier Science Inc. Printed in the USA. All rights reserved 0091-3057/99/$ see front matter Effects
PII S0091-3057(99)00058-1 Pharmacology Biochemistry and Behavior, Vol. 64, No. 2, pp. 367 371, 1999 1999 Elsevier Science Inc. Printed in the USA. All rights reserved 0091-3057/99/$ see front matter Effects
Antidiarrheals Antidiarrheal
 Antidiarrheals Major factors in diarrhea Increased motility of the GI tract. Decreased absorption of fluid. Antidiarrheal drugs include: Antimotility agents. Adsorbents. Drugs that modify fluid and electrolyte
Antidiarrheals Major factors in diarrhea Increased motility of the GI tract. Decreased absorption of fluid. Antidiarrheal drugs include: Antimotility agents. Adsorbents. Drugs that modify fluid and electrolyte
Drug Receptor Interactions and Pharmacodynamics
 Drug Receptor Interactions and Pharmacodynamics Dr. Raz Mohammed MSc Pharmacology School of Pharmacy 22.10.2017 Lec 6 Pharmacodynamics definition Pharmacodynamics describes the actions of a drug on the
Drug Receptor Interactions and Pharmacodynamics Dr. Raz Mohammed MSc Pharmacology School of Pharmacy 22.10.2017 Lec 6 Pharmacodynamics definition Pharmacodynamics describes the actions of a drug on the
Refractory Central Neurogenic Pain in Spinal Cord Injury. Case Presentation
 Refractory Central Neurogenic Pain in Spinal Cord Injury Case Presentation Edwin B. George, MD, PhD Wayne State University John D. Dingell VAMC 2012 Disclosures This continuing education activity is managed
Refractory Central Neurogenic Pain in Spinal Cord Injury Case Presentation Edwin B. George, MD, PhD Wayne State University John D. Dingell VAMC 2012 Disclosures This continuing education activity is managed
The Role of Ketamine in the Management of Complex Acute Pain
 The Role of Ketamine in the Management of Complex Acute Pain Dr James Bennett Consultant Anaesthetist Consultant Lead for Inpatient Pain Service East Sussex Healthcare NHS Trust STAPG Committee Member
The Role of Ketamine in the Management of Complex Acute Pain Dr James Bennett Consultant Anaesthetist Consultant Lead for Inpatient Pain Service East Sussex Healthcare NHS Trust STAPG Committee Member
A comparison of the sensitivities of innervated and denervated rat vasa deferentia to agonist drugs
 Br. J. Pharmac. (1970), 39, 748-754. A comparison of the sensitivities of innervated and denervated rat vasa deferentia to agonist drugs A. T. BIRMINGHAM*, G. PATRSON AND J. W6JCICKIt Department of Pharmacology,
Br. J. Pharmac. (1970), 39, 748-754. A comparison of the sensitivities of innervated and denervated rat vasa deferentia to agonist drugs A. T. BIRMINGHAM*, G. PATRSON AND J. W6JCICKIt Department of Pharmacology,
Effects of nalbuphine, pentazocine and U50488H on gastric emptying and gastrointestinal transit in the rat
 British Journal of Anaesthesia 1998; 80: 814 819 Effects of nalbuphine, pentazocine and U50488H on gastric emptying and gastrointestinal transit in the rat T. ASAI, W. W. MAPLESON, I. POWER Summary We
British Journal of Anaesthesia 1998; 80: 814 819 Effects of nalbuphine, pentazocine and U50488H on gastric emptying and gastrointestinal transit in the rat T. ASAI, W. W. MAPLESON, I. POWER Summary We
BIPN100 F15 Human Physiology 1 Lecture 3. Synaptic Transmission p. 1
 BIPN100 F15 Human Physiology 1 Lecture 3. Synaptic Transmission p. 1 Terms you should know: synapse, neuromuscular junction (NMJ), pre-synaptic, post-synaptic, synaptic cleft, acetylcholine (ACh), acetylcholine
BIPN100 F15 Human Physiology 1 Lecture 3. Synaptic Transmission p. 1 Terms you should know: synapse, neuromuscular junction (NMJ), pre-synaptic, post-synaptic, synaptic cleft, acetylcholine (ACh), acetylcholine
Acute Pain NETP: SEPTEMBER 2013 COHORT
 Acute Pain NETP: SEPTEMBER 2013 COHORT Pain & Suffering an unpleasant sensory & emotional experience associated with actual or potential tissue damage, or described in terms of such damage International
Acute Pain NETP: SEPTEMBER 2013 COHORT Pain & Suffering an unpleasant sensory & emotional experience associated with actual or potential tissue damage, or described in terms of such damage International
At a Glance. Background Information. Lesson 3 Drugs Change the Way Neurons Communicate
 Lesson 3 Drugs Change the Way Neurons Communicate Overview Students build upon their understanding of neurotransmission by learning how different drugs of abuse disrupt communication between neurons. Students
Lesson 3 Drugs Change the Way Neurons Communicate Overview Students build upon their understanding of neurotransmission by learning how different drugs of abuse disrupt communication between neurons. Students
