Determinants of Topical 5-Aminosalicylic Acid Use in Veterans with Ulcerative Colitis
|
|
|
- Megan Hodge
- 5 years ago
- Views:
Transcription
1 Research Article imedpub Journals Abstract Determinants of Topical 5-Aminosalicylic Acid Use in Veterans with Ulcerative Colitis Aims: The aim of our study was to identify the determinants of topical 5-ASA therapy in a large ulcerative colitis cohort. Methods: We performed a cross sectional study of patients receiving care through the Veterans Administration (VA) from 2003 to Patients with UC were identified using ICD-9 codes. Patient demographic and prescription data were obtained from national VA datasets. Univariate and multivariate logistic regression were used to identify the effect of age, gender and race on topical 5-ASA therapy use. Results: We identified 20,259 UC patients for inclusion in the analysis. The prevalence of topical 5-ASA therapy use was 27%. With univariate logistic regression, female gender and non-caucasian race were associated with a higher odds of topical 5-ASA therapy use. Increasing age was associated with lower odds of topical 5-ASA therapy use. On multivariate logistic regression, Blacks and Hispanics remained more likely to use topical 5-ASA therapies compared to Caucasians and increasing age remained associated with a lower odds of topical 5-ASA therapy; however, other demographic features lost significance. Conclusions: In a large, national cohort of patients with UC, Blacks and Hispanics were more likely to use topical 5-ASA therapies compared to patients of other ethnicities. Keywords: Inflammatory bowel disease; Ulcerative colitis; 5-Aminosalicylic acid Abbreviations: Inflammatory bowel disease (IBD); Ulcerative colitis (UC); 5-Aminosalicylates (5-ASA); Veterans Administration (VA); Decision Support Systems (DSS) Rajesh Shah 1, Jason Hou 1,2, Hoda Malaty 2,3, Shubhada Sansgiry 2,3 1 Department of Internal Medicine, Baylor College of Medicine, Houston, TX 77030,USA 2 Center of Excellence, HSR&D, Houston VA,USA 3 Michael E. DeBakey Veterans Affairs Medical Center, Houston, Texas 77030,USA Corresponding author: Rajesh Shah rajeshs@bcm.edu 6620 Main Street, #11d.32.5 Houston,USA Tel: Fax: Introduction Topical 5-aminosalicylate therapies (5-ASAs) are efficacious, safe and cost effective treatments for ulcerative colitis (UC). Current practice guidelines from the American College of Gastroenterology recommend topical 5-ASA therapy use in mild to moderate distal UC [1]. Compared to placebo, 5-ASA therapy can improve the probability of symptomatic, endoscopic and histologic remission [2]. Topical 5-ASA therapy combined with oral therapy is superior to either alone. The addition of 5-ASA enemas to oral 5-ASA medications raises the percent reaching clinical remission from 43% to 64% [3]. Despite documented efficacy, the use of topical 5-ASA therapies is variable. Data from the United States suggest approximately 53% of newly diagnosed patients with UC were prescribed a topical 5-ASA therapy [4]. Studies from Europe have reported a wide range of topical 5-ASA therapy use. From Spain, a survey based study found gastroenterologists commonly recommended topical therapies (74-81%) [5]. However, they did not report the prevalence of patients who actually followed these recommendations. In contrast, a study from a Swiss IBD cohort found only 7.8% of 1
2 patients with UC were prescribed topical 5-ASA therapies [6]. These studies highlight the wide variation in topical 5-ASA therapy usage, but do not address possible determinants of topical 5-ASA therapy use. The aims of our study were to determine the prevalence of topical 5-ASA use and the effect of age, gender and race as determinants for their use in a large United States cohort of patients. Materials and Methods Data Sources We performed a cross-sectional study using the Veterans Administration (VA) National Patient Care Database from fiscal years The study was approved by the IRB of Baylor College of Medicine. Specifically, we used the Inpatient and Outpatient Medical SAS datasets, which contain patient-level data. The data within these datasets include demographics (age, gender, race/ethnicity), encounters (date and frequency) and diagnosis codes [International Classification of, 9th Revision, (ICD-9)]. Race/ethnicity was designated as Caucasian (non-hispanic), Black, Hispanic, and other. The VA Decision Support Systems (DSS) database was used to obtain pharmacy utilization data of filled prescriptions, fill dates, and days of medication dispensed. UC case definition VA users with UC were identified using a previously validated ICD-9 diagnosis code algorithm [7] of at least two VA encounters for UC (556.x) with at least one UC-associated VA outpatient encounter from the fiscal years Patients with any ICD-9 codes for Crohn s disease (555.x) were excluded. In order to evaluate only patients who received care through the VA, patients with no filled prescriptions for an IBD medication (5- ASA, prednisone, azathioprine, 6-mercaptopurine, methotrexate, infliximab, adalimumab, or certolizumab pegol) during the study period were excluded. Patients with missing information for gender or race were excluded. Determination of 5-aminosalicylic acid therapy use Data for filled prescriptions of 5-ASA from the VA were obtained from DSS. Oral 5-ASA therapy was classified as a filled prescription for any oral formulation of mesalamine, sulfasalazine, or balsalazide. Topical 5-ASA therapy was classified as either suppository or enema. Statistical analysis The crude period prevalence of 5-ASA therapy usage over the study period was determined for topical and oral 5-ASA separately. For the study period, the total number of patients with a filled prescription for topical 5-ASA therapy was used as the numerator and the total number of UC patients included in the analyses was used as the denominator. Similarly, the total number of patients with a filled prescription for oral 5-ASA therapy was used as the numerator and the total number of UC patients included in the analyses was used as the denominator. Determinants of topical 5-ASA therapy use were identified using univariate and multivariate analysis. Variables with a p-value <0.05 were included in the multivariate logistic regression model. Among patients prescribed both formulations of topical 5-ASA therapy, the sequence and timing of transition were analyzed. Statistical analysis was performed using STATA version 13 software (College Station, TX). Results Prevalence of topical and oral 5-ASA therapies A total of 26,242 patients with UC were initially identified using the ICD-9 code case finding algorithm described. Of these patients, 5,713 patients were excluded because they did not have either gender or race information; 20,259 patients were included in the analysis. The prevalence of any filled prescription for 5-ASA (oral or topical) therapy was 81%; 77% for any oral and 27% for any topical. Among the study population, the prevalence of filled prescriptions for suppositories and enemas was 14% and 19%, respectively. (Table 1) shows the baseline characteristics for the groups who filled topical 5-ASA and those who did not. Of the patients who filled topical therapies, their mean age was 56 years and the majority were Caucasian (81%) men (93%). Effects of Age, Gender and Race on use of topical 5-ASA therapies Patients who filled topical therapies were significantly younger Table 1 Baseline Demographics by Filled Topical 5-ASA therapy prescriptions. Topical 5-ASA (n=5,604) No Topical 5-ASA (n=14,925) P value Age at diagnosis (yrs), mean (SD) 56 (16) 61 (14) <0.05 Gender, n (%) Male 5,212 (93) 14,158 (95) <0.05 Female 392 (7) 767 (5) <0.05 Female 392 (7) 767 (5) <0.05 Race Caucasian 4,543 (81) 13,093 (88) <0.05 Black 647 (12) 1009 (7) <0.05 Hispanic 286 (5) 542 (4) <0.05 Other 128 (2) 281 (1) <0.05 2
3 than those who did not fill prescriptions (OR 0.978, 95% CI ). A univariate logistic regression analysis showed that females were more likely to fill topical 5-ASA therapies than males (OR 1.39, 95% CI ). Similarly, Blacks, Hispanics and other non-caucasian races were more likely to fill topical 5-ASA therapies compared to Caucasians (Table 2). Age, gender and race were tested for interactions using a multivariate logistic regression model. Age (OR 0.980, 95% CI ), Black race (OR 1.53, 95% CI ) and Hispanic ethnicity (OR 1.33, 95% CI ) remained significant variables associated with the use of topical 5-ASA therapies. However, female (OR 0.99, 95% CI ) and other races (OR 1.18, 95% CI ) lost their significance. Sequence and switching among topical 5-ASA therapies A total of 1,310 patients filled prescriptions for both suppository and enema 5-ASA formulations during the study period. Of these patients, 58.5% were initially prescribed an enema before using suppositories, 34.6% were initially prescribed suppositories before using enemas and 6.95% were provided both topical medications at the time of diagnosis. When patients were switched between medications, the majority (25-49%) switched after 90 days (Table 3). The results revealed a higher percentage of people switched from suppositories to enemas in less than 30 days (5.5%) compared to those changing from enemas to suppositories (4.81%) (p=0.45). Discussion We performed a national cross sectional study to determine the prevalence of topical 5-ASA use and associations between topical 5-ASA use and age, gender, and race among patients with UC. We found a low prevalence of topical 5-ASA use compared to prior studies. Despite guideline recommendations for the use of topical or combination oral and topical 5-ASA therapy in mild to moderate UC, topical therapy is rarely used. We found only 27% of UC patients had filled prescriptions for topical 5-ASA therapies. Our findings are in sharp contrast with prior studies from Europe, where 74-81% of physicians recommended treating mild to moderate ulcerative proctitis with topical 5-ASA therapy [5]. Possible reasons our observed prevalence is markedly lower than the European studies is that we analyzed filled prescriptions and included all patients with UC regardless of disease extent. Our observed prevalence is similar to other U.S. based studies, ranging from %. [4, 8] We observed no difference in 5-ASA use by gender, similar to that reported in prior studies. Data from the University of Manitoba IBD Database showed no gender-specific differences in oral 5-ASA therapy use and did not report the gender specific results for topical therapies [9]. Similar findings were reported from a German study exploring differences in IBD treatment. They found no significant difference in 5-ASA therapy use, but did find women were less likely to receive immunosuppressive medications (azathioprine, methotrexate, 6-mercaptopurine, cyclosporine, tacrolimus) compared to men [10]. Differences in topical 5-ASA use among patients with UC separated by race have not previously been described. We observed Blacks and Hispanics were more likely than Caucasians to use topical 5-ASA therapy. A study (184 patients) from Houston, TX reported the treatment patterns among different racial groups (40% Caucasian, 37% Black, 20% Hispanic and 3% Asian). They found no significant difference in use of 5-ASA therapies, but topical therapies were not specifically examined [11]. Sewell et al. performed a retrospective study of patients from San Francisco General Hospital and found any 5-ASA use was less common among Blacks and Hispanics [12]. They, however, did not separately analyze oral and topical 5-ASA therapies. These differences may be explained by our focus on topical therapies, compared to those studies analyzing overall 5-ASA therapy use. Another potential reason for differences in topical 5-ASA use by race is differences in UC extent. Blacks (17-23%) and Hispanics (4-56%) have a higher proportion of proctitis compared to non- Table 2 Univariate and Multivariate Logistic Regression Analysis for Age, Gender and Race as Determinants of Topical 5-ASA therapy use. Univariate Multivariate OR 95% CI OR 95% CI Age Gender Male Ref - Ref - Female Race Caucasian Ref - Ref - Black Hispanic Other Table3 Frequency of Changing Topical 5-ASA among Patients Prescribed both Formulations. Suppositories to Enemas, n=453(%) Enemas to Suppositories, n=766 (%) P value <30 days 72 (5.50%) 63 (4.81%) days 38 (2.90%) 34 (2.60%) days 21 (1.60%) 22 (1.68%) 0.92 >90 days 322 (24.58%) 647 (49.39%) <0.05 Under License of Creative Commons Attribution 3.0 License 3
4 Hispanic Caucasians (6-10%) [11, 13-15]. Based on this, we would assume Blacks and Hispanics should receive topical 5-ASA therapies more often than Caucasians, which is in agreement with our findings. Almost of quarter of patients prescribed topical 5-ASA had filled prescriptions for both enema and suppository 5-ASA formulations throughout our study period. We observed nearly twice as many patients were switched from enemas to suppositories compared to switching from suppositories to enemas. We hypothesize this change in usage pattern of topical 5-ASA therapy is related to improved tolerance of suppositories compared to enemas; however, further studies considering disease extent are needed to confirm this observation. Our study has several strengths, most notably the large sample size and accuracy of VA national pharmacy databases and use of filled prescriptions rather than prescribed medications or physician self-report. Furthermore, VA care centers are community based and less likely to have referral bias frequently seen in data from tertiary care centers. Limitations of our study include inherent limitations from utilizing administrative data. The VA population is predominantly male; therefore, we may have been underpowered to assess gender differences. Additionally, we included all patients treated through the VA system, without distinguishing incident and prevalent cases, therefore prevalent cases may have received topical therapies prior to seeking treatment at the VA. Regarding definitions for disease extent, ICD-9 codes accounting for disease extent exist, but they are inaccurate [16]. Further studies accurately accounting for disease extent will be needed. In conclusion, prevalence of topical 5-ASA therapy use is low. Black and Hispanic patients are more likely to receive filled prescriptions of 5-ASA compared to non-hispanic Caucasians. Further studies are needed to explore confounding factors, such as disease extent, and potential differences in outcomes related to topical 5-ASA therapy use. Funding This work was supported by an investigator-initiated grant from Aptalis Pharmaceuticals. The sponsor provided funding but was not involved in the study design, analysis, or drafting or editing of the manuscript. 4
5 References 1 Kornbluth A, Sachar DB, Practice Parameters Committee of the American College of Gastroenterology (2010) Ulcerative colitis practice guidelines in adults: American College Of Gastroenterology, Practice Parameters Committee. The American journal of gastroenterology 105: Marshall JK, Thabane M, Steinhart AH, Newman JR, Anand, A et al. (2010) Rectal 5-aminosalicylic acid for induction of remission in ulcerative colitis. The Cochrane database of systematic reviews. 3 Marteau P, Probert CS, Lindgren S, Gassul M, Tan TG, et al. (2005) Combined oral and enema treatment with Pentasa (mesalazine) is superior to oral therapy alone in patients with extensive mild/moderate active ulcerative colitis: a randomised, double blind, placebo controlled study. Gut 54: Khan NH, Almukhtar RM, Cole EB, Abbas AM (2014) Early corticosteroids requirement after the diagnosis of ulcerative colitis diagnosis can predict a more severe long-term course of the disease - a nationwide study of 1035 patients. Alimentary pharmacology & therapeutics 40: Gisbert JP, Gomollon F, Hinojosa J, Lopez San Roman A (2010) Adherence of gastroenterologists to European Crohn's and Colitis Organisation consensus on ulcerative colitis: a real-life survey in Spain. Journal of Crohn's & colitis 4: Seibold F, Fournier N, Beglinger C, Mottet C, Pittet V, et.al (2014) Topical therapy is underused in patients with ulcerative colitis. Journal of Crohn's & colitis 8: Hou JK, Tan M, Stidham RW, Colozzi J, Adams D, et al. (2014) Accuracy of diagnostic codes for identifying patients with ulcerative colitis and Crohn's disease in the Veterans Affairs Health Care System. Digestive diseases and sciences 59: Reddy SI, Friedman S, Telford JJ, Strate L, Ookubo R, et al. (2005) Are patients with inflammatory bowel disease receiving optimal care?. The American journal of gastroenterology 100: Metge CJ, Blanchard JF, Peterson S, Bernstein CN (2001) Use of pharmaceuticals by inflammatory bowel disease patients: a population-based study. The American journal of gastroenterology 96: Blumenstein I, Herrmann E, Filmann N, Zosel C, Tacke W, et al. (2011) Female patients suffering from inflammatory bowel diseases are treated less frequently with immunosuppressive medication and have a higher disease activity: a subgroup analysis of a large multi-centre, prospective, internet-based study. Journal of Crohn's & colitis 5: Basu D, Lopez I, Kulkarni A, Sellin JH (2005) Impact of race and ethnicity on inflammatory bowel disease. The American journal of gastroenterology 100: Sewell JL, Inadomi JM, Hal F. Yee Jr (2010) Race and inflammatory bowel disease in an urban healthcare system. Digestive diseases and sciences 55: Nguyen GC, Torres EA, Regueiro M, Bromfield G, Bitton A, et al. (2006) Inflammatory bowel disease characteristics among African Americans, Hispanics, and non-hispanic Whites: characterization of a large North American cohort. The American journal of gastroenterology 101: Flasar MH, Quezada S, Bijpuria P, Cross RK (2008) Racial differences in disease extent and severity in patients with ulcerative colitis: a retrospective cohort study. Digestive diseases and sciences 53: Hou JK, El-Serag H, Thirumurthi S (2009) Distribution and manifestations of inflammatory bowel disease in Asians, Hispanics, and African Americans: a systematic review. The American journal of gastroenterology 104: Thirumurthi S, Chowdhury R, Richardson P, Abraham NS (2010) Validation of ICD-9-CM diagnostic codes for inflammatory bowel disease among veterans. Digestive diseases and sciences 55: Under License of Creative Commons Attribution 3.0 License 5
Topical therapy is underused in patients with ulcerative colitis
 Journal of Crohn's and Colitis (2014) 8, 56 63 Available online at www.sciencedirect.com ScienceDirect Topical therapy is underused in patients with ulcerative colitis F. Seibold a, f,, N. Fournier b,
Journal of Crohn's and Colitis (2014) 8, 56 63 Available online at www.sciencedirect.com ScienceDirect Topical therapy is underused in patients with ulcerative colitis F. Seibold a, f,, N. Fournier b,
DENOMINATOR: All patients aged 18 and older with a diagnosis of inflammatory bowel disease
 Measure #270: Inflammatory Bowel Disease (IBD): Preventive Care: Corticosteroid Sparing Therapy National Quality Strategy Domain: Effective Clinical Care 2016 PQRS OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY
Measure #270: Inflammatory Bowel Disease (IBD): Preventive Care: Corticosteroid Sparing Therapy National Quality Strategy Domain: Effective Clinical Care 2016 PQRS OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY
Doncaster & Bassetlaw Medicines Formulary
 Doncaster & Bassetlaw Medicines Formulary Section 1.5 Chronic Bowel Disorders (including IBD) Aminosalicylates: Mesalazine 400mg and 800mg MR Tablets (Octasa) Mesalazine 1.2g MR Tablets (Mezavant XL) Mesalazine
Doncaster & Bassetlaw Medicines Formulary Section 1.5 Chronic Bowel Disorders (including IBD) Aminosalicylates: Mesalazine 400mg and 800mg MR Tablets (Octasa) Mesalazine 1.2g MR Tablets (Mezavant XL) Mesalazine
The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION. 20 October 2010
 The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 20 October 2010 MEZAVANT LP 1200 mg, prolonged-release gastro-resistant tablets B/60 (CIP code: 378 689-2) Applicant
The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 20 October 2010 MEZAVANT LP 1200 mg, prolonged-release gastro-resistant tablets B/60 (CIP code: 378 689-2) Applicant
Once Daily Dosing for Induction and Maintenance of Remission in Ulcerative Colitis
 Once Daily Dosing for Induction and Maintenance of Remission in Ulcerative Colitis John K. Marshall MD MSc FRCPC AGAF Division of Gastroenterology McMaster University JKM 2014 Svartz N. Acta Med Scand
Once Daily Dosing for Induction and Maintenance of Remission in Ulcerative Colitis John K. Marshall MD MSc FRCPC AGAF Division of Gastroenterology McMaster University JKM 2014 Svartz N. Acta Med Scand
Month/Year of Review: September 2012 Date of Last Review: September 2010
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35, Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35, Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
5-ASA for the treatment of Crohn s disease DR. STEPHEN HANAUER FEINBERG SCHOOL OF MEDICINE, NORTHWESTERN UNIVERSITY, CHICAGO, IL, USA
 5-ASA for the treatment of Crohn s disease DR. STEPHEN HANAUER FEINBERG SCHOOL OF MEDICINE, NORTHWESTERN UNIVERSITY, CHICAGO, IL, USA Background RCTs investigating the efficacy of aminosalicylates for
5-ASA for the treatment of Crohn s disease DR. STEPHEN HANAUER FEINBERG SCHOOL OF MEDICINE, NORTHWESTERN UNIVERSITY, CHICAGO, IL, USA Background RCTs investigating the efficacy of aminosalicylates for
Dr. Elmer Schabel, MD. Bundesinstitut für Arzneimittel und Medizinprodukte, Bonn, Germany (No conflicts of interest)
 EMA workshop on the development of new medicinal products for the treatment of ulcerative colitis and Crohn s disease Overview of authorised medicines for IBD in Europe - previous regulatory positions
EMA workshop on the development of new medicinal products for the treatment of ulcerative colitis and Crohn s disease Overview of authorised medicines for IBD in Europe - previous regulatory positions
Moderately to severely active ulcerative colitis
 Adalimumab in the Treatment of Moderate-to-Severe Ulcerative Colitis: ULTRA 2 Trial Results Sandborn WJ, van Assche G, Reinisch W, et al. Adalimumab induces and maintains clinical remission in patients
Adalimumab in the Treatment of Moderate-to-Severe Ulcerative Colitis: ULTRA 2 Trial Results Sandborn WJ, van Assche G, Reinisch W, et al. Adalimumab induces and maintains clinical remission in patients
IBD Understanding Your Medications. Thomas V. Aguirre, MD Santa Barbara GI Consultants
 IBD Understanding Your Medications Thomas V. Aguirre, MD Santa Barbara GI Consultants IBD Understanding Your Medications (& Your Doctor) Thomas V. Aguirre, MD Santa Barbara GI Consultants Disclosure I
IBD Understanding Your Medications Thomas V. Aguirre, MD Santa Barbara GI Consultants IBD Understanding Your Medications (& Your Doctor) Thomas V. Aguirre, MD Santa Barbara GI Consultants Disclosure I
The Harvard community has made this article openly available. Please share how this access benefits you. Your story matters. doi:10.
 Oral 5-Aminosalicylate, Suppository, and Enema as Initial Therapy for Ulcerative Proctitis in Clinical Practice with Quality of Care Implications The Harvard community has made this article openly available.
Oral 5-Aminosalicylate, Suppository, and Enema as Initial Therapy for Ulcerative Proctitis in Clinical Practice with Quality of Care Implications The Harvard community has made this article openly available.
To help protect your privacy, PowerPoint prevented this external picture from being automatically downloaded. To download and display this picture,
 To help protect your privacy, PowerPoint prevented this external picture from being automatically downloaded. To download and display this picture, click Options in the Message Bar, and then click Enable
To help protect your privacy, PowerPoint prevented this external picture from being automatically downloaded. To download and display this picture, click Options in the Message Bar, and then click Enable
Mild-moderate Ulcerative Colitis Sequential & Combined treatments need to be tested. Philippe Marteau, Paris, France
 Mild-moderate Ulcerative Colitis Sequential & Combined treatments need to be tested Philippe Marteau, Paris, France Sequential vs combined treatments When should one switch? Sequential vs combined treatments
Mild-moderate Ulcerative Colitis Sequential & Combined treatments need to be tested Philippe Marteau, Paris, France Sequential vs combined treatments When should one switch? Sequential vs combined treatments
Definitions. Clinical remission: Resolution of symptoms (stool frequency 3/day, no bleeding and no urgency)
 CROHN S DISEASE Definitions Clinical remission: Resolution of symptoms (stool frequency 3/day, no bleeding and no urgency) Recurrence: The reappearance of lesions after surgical resection Endoscopic remission:
CROHN S DISEASE Definitions Clinical remission: Resolution of symptoms (stool frequency 3/day, no bleeding and no urgency) Recurrence: The reappearance of lesions after surgical resection Endoscopic remission:
Review article: induction therapy for patients with active ulcerative colitis
 Alimentary Pharmacology & Therapeutics Review article: induction therapy for patients with active ulcerative colitis S. P. L. TRAVIS John Radcliffe Hospital and Linacre College, Oxford, UK Correspondence
Alimentary Pharmacology & Therapeutics Review article: induction therapy for patients with active ulcerative colitis S. P. L. TRAVIS John Radcliffe Hospital and Linacre College, Oxford, UK Correspondence
Treatment of ulcerative colitis with adalimumab or infliximab: long-term follow-up of a single-centre cohort
 Alimentary Pharmacology and Therapeutics Treatment of ulcerative colitis with adalimumab or infliximab: long-term follow-up of a single-centre cohort N. Gies, K. I. Kroeker, K. Wong & R. N. Fedorak Division
Alimentary Pharmacology and Therapeutics Treatment of ulcerative colitis with adalimumab or infliximab: long-term follow-up of a single-centre cohort N. Gies, K. I. Kroeker, K. Wong & R. N. Fedorak Division
Retention in HIV care predicts subsequent retention and predicts survival well after the first year of care: a national study of US Veterans
 Retention in HIV care predicts subsequent retention and predicts survival well after the first year of care: a national study of US Veterans Thomas P. Giordano, MD, MPH, Jessica A. Davila, PhD, Christine
Retention in HIV care predicts subsequent retention and predicts survival well after the first year of care: a national study of US Veterans Thomas P. Giordano, MD, MPH, Jessica A. Davila, PhD, Christine
Predictors of DEXA Use and Guideline Performance for the Detection of Low Bone Mineral Density in Inflammatory Bowel Disease
 Predictors of DEXA Use and Guideline Performance for the Detection of Low Bone Mineral Density in Inflammatory Bowel Disease Jason Etzel Resident Research Forum Seattle VAMC 6/13/08 Background Increased
Predictors of DEXA Use and Guideline Performance for the Detection of Low Bone Mineral Density in Inflammatory Bowel Disease Jason Etzel Resident Research Forum Seattle VAMC 6/13/08 Background Increased
13-year mortality trends among hospitalized patients with inflammatory bowel disease
 Sewell and Yee BMC Gastroenterology 212, 12:79 RESEARCH ARTICLE Open Access 13-year mortality trends among hospitalized patients with inflammatory bowel disease Justin L Sewell * and Hal F Yee Jr Abstract
Sewell and Yee BMC Gastroenterology 212, 12:79 RESEARCH ARTICLE Open Access 13-year mortality trends among hospitalized patients with inflammatory bowel disease Justin L Sewell * and Hal F Yee Jr Abstract
Primary & Secondary Care Inflammatory Bowel Disease Pathway February 2018
 South East London Area Prescribing Committee: Primary & Secondary Care Inflammatory Bowel Disease Pathway February 2018 Developed by: South East London IBD Pathway Development Group Approved: February
South East London Area Prescribing Committee: Primary & Secondary Care Inflammatory Bowel Disease Pathway February 2018 Developed by: South East London IBD Pathway Development Group Approved: February
How to Optimize Induction and Maintenance Responses: Definitions and Dosing Advances in Inflammatory Bowel Disease December 6, 2009
 How to Optimize Induction and Maintenance Responses: Definitions and Dosing 2009 Advances in Inflammatory Bowel Disease December 6, 2009 Fernando Velayos MD MPH University of California, San Francisco
How to Optimize Induction and Maintenance Responses: Definitions and Dosing 2009 Advances in Inflammatory Bowel Disease December 6, 2009 Fernando Velayos MD MPH University of California, San Francisco
Full title of guideline: Ulcerative colitis: the management of ulcerative colitis
 Economic Plan This document identifies the priorities for economic analysis and the proposed methods for addressing these questions as described in section 7.1.3 of the Guidelines Manual (2009). 1 Guideline
Economic Plan This document identifies the priorities for economic analysis and the proposed methods for addressing these questions as described in section 7.1.3 of the Guidelines Manual (2009). 1 Guideline
Significant racial, ethnic, and socioeconomic disparities in
 ORIGINAL ARTICLE Race Differences in Initial Presentation, Early Treatment, and 1-year Outcomes of Pediatric Crohn s Disease: Results from the ImproveCareNow Network Jennifer L. Dotson, MD, MPH,*, Michele
ORIGINAL ARTICLE Race Differences in Initial Presentation, Early Treatment, and 1-year Outcomes of Pediatric Crohn s Disease: Results from the ImproveCareNow Network Jennifer L. Dotson, MD, MPH,*, Michele
Regulatory Status FDA-approved indications: Entyvio is an α4β7integrin receptor antagonist indicated for: (1)
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.50.12 Subject: Entyvio Page: 1 of 7 Last Review Date: September 20, 2018 Entyvio Description Entyvio
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.50.12 Subject: Entyvio Page: 1 of 7 Last Review Date: September 20, 2018 Entyvio Description Entyvio
Perianal and Fistulizing Crohn s Disease: Tough Management Decisions. Jean-Paul Achkar, M.D. Kenneth Rainin Chair for IBD Research Cleveland Clinic
 Perianal and Fistulizing Crohn s Disease: Tough Management Decisions Jean-Paul Achkar, M.D. Kenneth Rainin Chair for IBD Research Cleveland Clinic Talk Overview Background Assessment and Classification
Perianal and Fistulizing Crohn s Disease: Tough Management Decisions Jean-Paul Achkar, M.D. Kenneth Rainin Chair for IBD Research Cleveland Clinic Talk Overview Background Assessment and Classification
Epidemiology and Treatment of Colonic Angiodysplasia; a Population-Based Study. Naomi G. Diggs, MD Lisa L. Strate, MD MPH March 2, 2010
 Epidemiology and Treatment of Colonic Angiodysplasia; a Population-Based Study. Naomi G. Diggs, MD Lisa L. Strate, MD MPH March 2, 2010 Background Angiodysplasia is an important cause of occult and acute
Epidemiology and Treatment of Colonic Angiodysplasia; a Population-Based Study. Naomi G. Diggs, MD Lisa L. Strate, MD MPH March 2, 2010 Background Angiodysplasia is an important cause of occult and acute
National Institute for Health and Care Excellence
 National Institute for Health and Care Excellence 4-year surveillance (2017) Ulcerative colitis: management (2013) NICE guideline CG166 Appendix A.2: Summary of new evidence from surveillance Patient information
National Institute for Health and Care Excellence 4-year surveillance (2017) Ulcerative colitis: management (2013) NICE guideline CG166 Appendix A.2: Summary of new evidence from surveillance Patient information
New treatment options in UC. Rob Bryant IBD Consultant Royal Adelaide Hospital
 New treatment options in UC Rob Bryant IBD Consultant Royal Adelaide Hospital Talk Outline 1. Raising expectations 2. Optimising UC therapy 3. Clinical trials 4. What s new on the PBS? 5. Questions 1.
New treatment options in UC Rob Bryant IBD Consultant Royal Adelaide Hospital Talk Outline 1. Raising expectations 2. Optimising UC therapy 3. Clinical trials 4. What s new on the PBS? 5. Questions 1.
Carefirst.+.V Family of health care plans
 Carefirst.+.V Family of health care plans CVS care mark POLICY Document for ENTYVIO The overall objective of this policy is to support the appropriate and cost effective use of the medication, specific
Carefirst.+.V Family of health care plans CVS care mark POLICY Document for ENTYVIO The overall objective of this policy is to support the appropriate and cost effective use of the medication, specific
There is a well-established association between inflammatory
 CLINICAL GASTROENTEROLOGY AND HEPATOLOGY 2006;4:1346 1350 Aminosalicylate Therapy in the Prevention of Dysplasia and Colorectal Cancer in Ulcerative Colitis DAVID T. RUBIN, ANDELKA LOSAVIO, NICOLE YADRON,
CLINICAL GASTROENTEROLOGY AND HEPATOLOGY 2006;4:1346 1350 Aminosalicylate Therapy in the Prevention of Dysplasia and Colorectal Cancer in Ulcerative Colitis DAVID T. RUBIN, ANDELKA LOSAVIO, NICOLE YADRON,
September 12, 2015 Millie D. Long MD, MPH, FACG
 Update on Biologic Therapy in 2015 September 12, 2015 Millie D. Long MD, MPH, FACG Assistant Professor of Medicine Inflammatory Bowel Disease Center University of North Carolina-Chapel Hill Outline Crohn
Update on Biologic Therapy in 2015 September 12, 2015 Millie D. Long MD, MPH, FACG Assistant Professor of Medicine Inflammatory Bowel Disease Center University of North Carolina-Chapel Hill Outline Crohn
Ashish Malhotra 1,2,3*, K.C. Mandip 3, Aasma Shaukat 1,2,3 and Thomas Rector 2,3
 Malhotra et al. Military Medical Research (2016) 3:28 DOI 10.1186/s40779-016-0098-x RESEARCH Open Access All-cause hospitalizations for inflammatory bowel diseases: Can the reason for admission provide
Malhotra et al. Military Medical Research (2016) 3:28 DOI 10.1186/s40779-016-0098-x RESEARCH Open Access All-cause hospitalizations for inflammatory bowel diseases: Can the reason for admission provide
Understanding Inflammatory Bowel Diseases (IBD):
 Understanding Inflammatory Bowel Diseases (IBD): What Every Patient Needs to Know William H Holderman, MD Digestive Health Specialists Tacoma, WA Today s Objectives Define IBD, its potential causes and
Understanding Inflammatory Bowel Diseases (IBD): What Every Patient Needs to Know William H Holderman, MD Digestive Health Specialists Tacoma, WA Today s Objectives Define IBD, its potential causes and
Ali Keshavarzian MD Rush University Medical Center
 Treatment: Step Up or Top Down? Ali Keshavarzian MD Rush University Medical Center Questions What medication should IBD be treated with? Can we predict which patients with IBD are high risk? Is starting
Treatment: Step Up or Top Down? Ali Keshavarzian MD Rush University Medical Center Questions What medication should IBD be treated with? Can we predict which patients with IBD are high risk? Is starting
Medical therapies and IBD
 Medical therapies and IBD Although there is no cure for IBD, there are many treatment options available. There is no standard treatment for IBD that is effective in all situations or for all patients,
Medical therapies and IBD Although there is no cure for IBD, there are many treatment options available. There is no standard treatment for IBD that is effective in all situations or for all patients,
Evidence review for Surrey Prescribing Clinical Network SUMMARY
 East Surrey CCG, Guildford & Waverley CCG, North West Surrey CCG, Surrey Downs CCG, Surrey Heath CCG, Crawley CCG, Horsham & Mid-Sussex CCG Evidence review for Surrey Prescribing Clinical Network Medicine
East Surrey CCG, Guildford & Waverley CCG, North West Surrey CCG, Surrey Downs CCG, Surrey Heath CCG, Crawley CCG, Horsham & Mid-Sussex CCG Evidence review for Surrey Prescribing Clinical Network Medicine
Accepted Manuscript. Frank I. Scott, Siddharth Singh. S (19) DOI: Reference: YJCGH 56282
 Accepted Manuscript Bridging Gaps in Evidence-based Clinical Practice in Inflammatory Bowel Diseases: Observational Comparative Effectiveness Research for the Win Frank I. Scott, Siddharth Singh PII: S1542-3565(19)30020-5
Accepted Manuscript Bridging Gaps in Evidence-based Clinical Practice in Inflammatory Bowel Diseases: Observational Comparative Effectiveness Research for the Win Frank I. Scott, Siddharth Singh PII: S1542-3565(19)30020-5
INFLIXIMAB FOR PREVENTION OF POST-OPERATIVE CROHN S DISEASE RECURRENCE: THE PREVENT TRIAL
 INFLIXIMAB FOR PREVENTION OF POST-OPERATIVE CROHN S DISEASE RECURRENCE: THE PREVENT TRIAL A. Hillary Steinhart, MD MSc FRCP(C) Medical Lead, Mount Sinai Hospital IBD Centre Professor of Medicine University
INFLIXIMAB FOR PREVENTION OF POST-OPERATIVE CROHN S DISEASE RECURRENCE: THE PREVENT TRIAL A. Hillary Steinhart, MD MSc FRCP(C) Medical Lead, Mount Sinai Hospital IBD Centre Professor of Medicine University
Review article: the long-term management of ulcerative colitis
 Aliment Pharmacol Ther 2004; 20 (Suppl. 4): 97 101. Review article: the long-term management of ulcerative colitis S. B. HANAUER Section of Gastroenterology, University of Chicago, Chicago, IL, USA SUMMARY
Aliment Pharmacol Ther 2004; 20 (Suppl. 4): 97 101. Review article: the long-term management of ulcerative colitis S. B. HANAUER Section of Gastroenterology, University of Chicago, Chicago, IL, USA SUMMARY
Infliximab (Remicade) for paediatric ulcerative colitis - second line
 Infliximab (Remicade) for paediatric ulcerative colitis - second line September 2011 This technology summary is based on information available at the time of research and a limited literature search. It
Infliximab (Remicade) for paediatric ulcerative colitis - second line September 2011 This technology summary is based on information available at the time of research and a limited literature search. It
Position of Biologics in IBD Circa 2006: Top Down vs. Step Up Therapy
 Position of Biologics in IBD Circa 2006: Top Down vs. Step Up Therapy Stephen B. Hanauer, MD University of Chicago Potential Conflicts: Centocor/Schering, Abbott, UCB, Elan, Berlex, PDL Goals of Treatment
Position of Biologics in IBD Circa 2006: Top Down vs. Step Up Therapy Stephen B. Hanauer, MD University of Chicago Potential Conflicts: Centocor/Schering, Abbott, UCB, Elan, Berlex, PDL Goals of Treatment
Guideline Ulcerative colitis: management
 NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Guideline Ulcerative colitis: management Draft for consultation, December 0 This guideline covers the care and treatment of adults, children and young
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Guideline Ulcerative colitis: management Draft for consultation, December 0 This guideline covers the care and treatment of adults, children and young
Title: Author: Journal:
 IMPORTANT COPYRIGHT NOTICE: This electronic article is provided to you by courtesy of Ferring Pharmaceuticals. The document is provided for personal usage only. Further reproduction and/or distribution
IMPORTANT COPYRIGHT NOTICE: This electronic article is provided to you by courtesy of Ferring Pharmaceuticals. The document is provided for personal usage only. Further reproduction and/or distribution
HARVARD PILGRIM HEALTH CARE RECOMMENDED MEDICATION REQUEST GUIDELINES HUMIRA PEDIATRIC
 Generic Brand HICL GCN Exception/Other ADALIMUMAB HUMIRA 24800 HUMIRA PEDIATRIC GUIDELINES FOR USE INITIAL CRITERIA (NOTE: FOR RENEWAL CRITERIA SEE BELOW) 1. Is the patient currently taking Humira? If
Generic Brand HICL GCN Exception/Other ADALIMUMAB HUMIRA 24800 HUMIRA PEDIATRIC GUIDELINES FOR USE INITIAL CRITERIA (NOTE: FOR RENEWAL CRITERIA SEE BELOW) 1. Is the patient currently taking Humira? If
IBD :- a new era of diagnostics and therapy Dr Martyn Dibb Consultant Luminal Gastroenterologist Royal Liverpool University Hospital
 IBD :- a new era of diagnostics and therapy Dr Martyn Dibb Consultant Luminal Gastroenterologist Royal Liverpool University Hospital Aims To understand the aetiology of IBD To understand the impact that
IBD :- a new era of diagnostics and therapy Dr Martyn Dibb Consultant Luminal Gastroenterologist Royal Liverpool University Hospital Aims To understand the aetiology of IBD To understand the impact that
IBD in teenagers Biological and Transition
 IBD in teenagers Biological and Transition Dr Warren Hyer Consultant Paediatric Gastroenterologist St Mark s Hospital Chelsea and Westminster Hospital Conflict of Interest None to declare Fee for presentation
IBD in teenagers Biological and Transition Dr Warren Hyer Consultant Paediatric Gastroenterologist St Mark s Hospital Chelsea and Westminster Hospital Conflict of Interest None to declare Fee for presentation
Inflammatory bowel disease (IBD) Overview of the Paediatric investigation plans. Presented by: Richard Veselý. An agency of the European Union
 Inflammatory bowel disease (IBD) Overview of the Paediatric investigation plans Presented by: Richard Veselý An agency of the European Union Adalimumab - Crohn s disease Indication: Treatment of severe,
Inflammatory bowel disease (IBD) Overview of the Paediatric investigation plans Presented by: Richard Veselý An agency of the European Union Adalimumab - Crohn s disease Indication: Treatment of severe,
C. Assess clinical response after the first three months of treatment.
 Government Health Plan (GHP) of Puerto Rico Authorization Criteria Tumor Necrosis Factor Alpha (TNFα) Adalimumab (Humira ) Managed by MCO Section I. Prior Authorization Criteria A. Physician must submit
Government Health Plan (GHP) of Puerto Rico Authorization Criteria Tumor Necrosis Factor Alpha (TNFα) Adalimumab (Humira ) Managed by MCO Section I. Prior Authorization Criteria A. Physician must submit
Effective Health Care Program
 Comparative Effectiveness Review Number 131 Effective Health Care Program Pharmacologic Therapies for the Management of Crohn s Disease: Comparative Effectiveness Executive Summary Description of Crohn
Comparative Effectiveness Review Number 131 Effective Health Care Program Pharmacologic Therapies for the Management of Crohn s Disease: Comparative Effectiveness Executive Summary Description of Crohn
Efficacy and Safety of Treatment for Pediatric IBD
 Efficacy and Safety of Treatment for Pediatric IBD Andrew B. Grossman MD Co-Director, Center for Pediatric Inflammatory Bowel Disease Associate Professor of Clinical Pediatrics Division of Gastroenterology,
Efficacy and Safety of Treatment for Pediatric IBD Andrew B. Grossman MD Co-Director, Center for Pediatric Inflammatory Bowel Disease Associate Professor of Clinical Pediatrics Division of Gastroenterology,
Pharmacotherapy of Inflammatory Bowel Disorder
 PHARMACY / MEDICAL POLICY 5.01.563 Pharmacotherapy of Inflammatory Bowel Disorder Effective Date: Feb. 14, 2018 Last Revised: April 1, 2018 Replaces: Extracted from 5.01.550 RELATED MEDICAL POLICIES: 11.01.523
PHARMACY / MEDICAL POLICY 5.01.563 Pharmacotherapy of Inflammatory Bowel Disorder Effective Date: Feb. 14, 2018 Last Revised: April 1, 2018 Replaces: Extracted from 5.01.550 RELATED MEDICAL POLICIES: 11.01.523
Systematic review and meta-analysis of third-line salvage therapy with infliximab or cyclosporine in severe ulcerative colitis
 Systematic review and meta-analysis of third-line salvage therapy with infliximab or cyclosporine in severe ulcerative colitis The Harvard community has made this article openly available. Please share
Systematic review and meta-analysis of third-line salvage therapy with infliximab or cyclosporine in severe ulcerative colitis The Harvard community has made this article openly available. Please share
C olorectal cancer (CRC) is one of the most feared
 1573 INFLAMMATORY BOWEL DISEASE 5-Aminosalicylate use and colorectal cancer risk in inflammatory bowel disease: a large epidemiological study T P van Staa, T Card, R F Logan, H G M Leufkens... See end
1573 INFLAMMATORY BOWEL DISEASE 5-Aminosalicylate use and colorectal cancer risk in inflammatory bowel disease: a large epidemiological study T P van Staa, T Card, R F Logan, H G M Leufkens... See end
5-ASA Therapy, Steroids and Antibiotics in Inflammatory Bowel Disease
 5-ASA Therapy, Steroids and Antibiotics in Inflammatory Bowel Disease David T. Rubin, MD Associate Professor of Medicine Co-Director, Inflammatory Bowel Disease Center University it of Chicago Medical
5-ASA Therapy, Steroids and Antibiotics in Inflammatory Bowel Disease David T. Rubin, MD Associate Professor of Medicine Co-Director, Inflammatory Bowel Disease Center University it of Chicago Medical
INFLAMMATORY BOWEL DISEASE
 1. Medical Condition INFLAMMATORY BOWEL DISEASE (IBD) specifically includes Crohn s disease (CD) and ulcerative colitis (UC) but also includes IBD unclassified (IBDu), seen in about 10% of cases. These
1. Medical Condition INFLAMMATORY BOWEL DISEASE (IBD) specifically includes Crohn s disease (CD) and ulcerative colitis (UC) but also includes IBD unclassified (IBDu), seen in about 10% of cases. These
This information explains the advice about Crohn's disease that is set out in NICE guideline CG152.
 Information for the public Published: 1 October 2012 nice.org.uk About this information NICE guidelines provide advice on the care and support that should be offered to people who use health and care services.
Information for the public Published: 1 October 2012 nice.org.uk About this information NICE guidelines provide advice on the care and support that should be offered to people who use health and care services.
WHAT IS ULCERATIVE COLITIS?
 235 60th Street, West New York, NJ 07093 T: (201) 854-4646 F: (201) 854-4647 810 Main Street, Hackensack, NJ 07601 T: (201) 488-0095 Ulcerative Colitis WHAT IS ULCERATIVE COLITIS? Ulcerative colitis is
235 60th Street, West New York, NJ 07093 T: (201) 854-4646 F: (201) 854-4647 810 Main Street, Hackensack, NJ 07601 T: (201) 488-0095 Ulcerative Colitis WHAT IS ULCERATIVE COLITIS? Ulcerative colitis is
Endpoints for Stopping Treatment in UC
 Endpoints for Stopping Treatment in UC Jana G. Hashash, MD Assistant Professor of Medicine Inflammatory Bowel Disease Center Division of Gastroenterology, Hepatology, and Nutrition University of Pittsburgh
Endpoints for Stopping Treatment in UC Jana G. Hashash, MD Assistant Professor of Medicine Inflammatory Bowel Disease Center Division of Gastroenterology, Hepatology, and Nutrition University of Pittsburgh
Bridges to excellence quality indicators in inflammatory bowel disease (IBD): differences between IBD and non-ibd gastroenterologists
 ORIGINAL ARTICLE Annals of Gastroenterology (2017) 30, 1-5 Bridges to excellence quality indicators in inflammatory bowel disease (IBD): differences between IBD and non-ibd gastroenterologists Mohammad
ORIGINAL ARTICLE Annals of Gastroenterology (2017) 30, 1-5 Bridges to excellence quality indicators in inflammatory bowel disease (IBD): differences between IBD and non-ibd gastroenterologists Mohammad
Treating Crohn s and Colitis in the ASC
 Treating Crohn s and Colitis in the ASC Kimberly M Persley, MD Texas Digestive Disease consultants TASC Meeting Outline IBD 101 Diagnosis Treatment Burden of Disease Role of ASC Inflammatory Bowel Disease
Treating Crohn s and Colitis in the ASC Kimberly M Persley, MD Texas Digestive Disease consultants TASC Meeting Outline IBD 101 Diagnosis Treatment Burden of Disease Role of ASC Inflammatory Bowel Disease
Garrick Brown, MD. Digestive Health Specialists Tacoma Gig Harbor
 Garrick Brown, MD Digestive Health Specialists Tacoma Gig Harbor Today s Objectives Define IBD, its potential causes and diagnosis Discuss management and treatment Discuss complementary and alternative
Garrick Brown, MD Digestive Health Specialists Tacoma Gig Harbor Today s Objectives Define IBD, its potential causes and diagnosis Discuss management and treatment Discuss complementary and alternative
Indications for use of Infliximab
 Indications for use of Infliximab Moscow, June 10 th 2006 Prof. Dr. Dr. Gerhard Rogler Klinik und Poliklinik für Innere Medizin I Universität Regensburg Case report 1989: Diagnosis of Crohn s disease of
Indications for use of Infliximab Moscow, June 10 th 2006 Prof. Dr. Dr. Gerhard Rogler Klinik und Poliklinik für Innere Medizin I Universität Regensburg Case report 1989: Diagnosis of Crohn s disease of
Azathioprine for Induction and Maintenance of Remission in Crohn s Disease
 Azathioprine for Induction and Maintenance of Remission in Crohn s Disease William J. Sandborn, MD Chief, Division of Gastroenterology Director, UCSD IBD Center Objectives Azathioprine as induction and
Azathioprine for Induction and Maintenance of Remission in Crohn s Disease William J. Sandborn, MD Chief, Division of Gastroenterology Director, UCSD IBD Center Objectives Azathioprine as induction and
Lessons to learn from Crohn's disease clinical trials: implications for ulcerative colitis
 Lessons to learn from Crohn's disease clinical trials: implications for ulcerative colitis Aránzazu Jáuregui Amézaga, Elena Ricart, Julián Panés Department of Gastroenterology, Hospital Clínic de Barcelona,
Lessons to learn from Crohn's disease clinical trials: implications for ulcerative colitis Aránzazu Jáuregui Amézaga, Elena Ricart, Julián Panés Department of Gastroenterology, Hospital Clínic de Barcelona,
Diagnostic Ionizing Radiation Exposure in a Population-Based Cohort of Children with Inflammatory Bowel Disease
 Diagnostic Ionizing Radiation Exposure in a Population-Based Cohort of Children with Inflammatory Bowel Disease Lena B. Palmer MD, Carol Q. Porter, Michael D. Kappelman MD MPH No relevant financial disclosures
Diagnostic Ionizing Radiation Exposure in a Population-Based Cohort of Children with Inflammatory Bowel Disease Lena B. Palmer MD, Carol Q. Porter, Michael D. Kappelman MD MPH No relevant financial disclosures
Overall and Comparative Risk of Herpes Zoster With Pharmacotherapy for Inflammatory Bowel Diseases: A Nationwide Cohort Study
 Accepted Manuscript Overall and Comparative Risk of Herpes Zoster With Pharmacotherapy for Inflammatory Bowel Diseases: A Nationwide Cohort Study Nabeel Khan, Dhruvan Patel, Chinmay Trivedi, Yash Shah,
Accepted Manuscript Overall and Comparative Risk of Herpes Zoster With Pharmacotherapy for Inflammatory Bowel Diseases: A Nationwide Cohort Study Nabeel Khan, Dhruvan Patel, Chinmay Trivedi, Yash Shah,
Budesonide Use and Hospitalization Rate in Crohn s Disease: Results From a Cohort at a Tertiary Care IBD Referral Center
 Elmer ress Original Article J Clin Med Res. 2016;8(10):705-709 Budesonide Use and Hospitalization Rate in Crohn s Disease: Results From a Cohort at a Tertiary Care IBD Referral Center Jordan Orr a, d,
Elmer ress Original Article J Clin Med Res. 2016;8(10):705-709 Budesonide Use and Hospitalization Rate in Crohn s Disease: Results From a Cohort at a Tertiary Care IBD Referral Center Jordan Orr a, d,
Efficacy and Safety of Treatment for Pediatric IBD
 Efficacy and Safety of Treatment for Pediatric IBD Andrew B. Grossman MD Co-Director, Center for Pediatric Inflammatory Bowel Disease Assistant Professor of Clinical Pediatrics Division of Gastroenterology,
Efficacy and Safety of Treatment for Pediatric IBD Andrew B. Grossman MD Co-Director, Center for Pediatric Inflammatory Bowel Disease Assistant Professor of Clinical Pediatrics Division of Gastroenterology,
Clinical Study Clinical Study of the Relation between Mucosal Healing and Long-Term Outcomes in Ulcerative Colitis
 Hindawi Publishing Corporation Gastroenterology Research and Practice Volume 2013, Article ID 192794, 6 pages http://dx.doi.org/10.1155/2013/192794 Clinical Study Clinical Study of the Relation between
Hindawi Publishing Corporation Gastroenterology Research and Practice Volume 2013, Article ID 192794, 6 pages http://dx.doi.org/10.1155/2013/192794 Clinical Study Clinical Study of the Relation between
Treatment Goals. Current Therapeutic Pyramids Crohn s Disease Ulcerative Colitis 11/14/10
 Current Management of IBD: From Conventional Agents to Biologics Stephen B. Hanauer, M.D. University of Chicago Treatment Goals Induce and maintain response/ remission Prevent complications Improve quality
Current Management of IBD: From Conventional Agents to Biologics Stephen B. Hanauer, M.D. University of Chicago Treatment Goals Induce and maintain response/ remission Prevent complications Improve quality
Pediatric Inflammatory Bowel Diseases
 Pediatric Inflammatory Bowel Diseases Michael Stephens, M.D. Denny Sanford Pediatric Symposium November 21 2014, Sioux Falls, SD 2014 slide-1 Disclosures I have nothing to disclose 2014 slide-2 Objectives
Pediatric Inflammatory Bowel Diseases Michael Stephens, M.D. Denny Sanford Pediatric Symposium November 21 2014, Sioux Falls, SD 2014 slide-1 Disclosures I have nothing to disclose 2014 slide-2 Objectives
What is ulcerative colitis?
 What is ulcerative colitis? Ulcerative colitis is a disease that causes inflammation and sores, called ulcers, in the lining of the rectum and colon. Ulcers form where inflammation has killed the cells
What is ulcerative colitis? Ulcerative colitis is a disease that causes inflammation and sores, called ulcers, in the lining of the rectum and colon. Ulcers form where inflammation has killed the cells
During the past 2 decades, an increase in the ageadjusted
 CLINICAL GASTROENTEROLOGY AND HEPATOLOGY 2006;4:104 110 Racial Differences in Survival of Hepatocellular Carcinoma in the United States: A Population-Based Study JESSICA A. DAVILA* and HASHEM B. EL SERAG*,
CLINICAL GASTROENTEROLOGY AND HEPATOLOGY 2006;4:104 110 Racial Differences in Survival of Hepatocellular Carcinoma in the United States: A Population-Based Study JESSICA A. DAVILA* and HASHEM B. EL SERAG*,
Circle Yes or No Y N. [If no, skip to question 7.] 2. Does the patient have a diagnosis of ulcerative colitis? Y N. [If no, skip to question 4.
![Circle Yes or No Y N. [If no, skip to question 7.] 2. Does the patient have a diagnosis of ulcerative colitis? Y N. [If no, skip to question 4. Circle Yes or No Y N. [If no, skip to question 7.] 2. Does the patient have a diagnosis of ulcerative colitis? Y N. [If no, skip to question 4.](/thumbs/85/91758188.jpg) 06/01/2016 Prior Authorization AETA BETTER HEALTH OF MICHIGA (MEDICAID) Humira (MI88) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review information, sign
06/01/2016 Prior Authorization AETA BETTER HEALTH OF MICHIGA (MEDICAID) Humira (MI88) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review information, sign
Technology appraisal guidance Published: 26 August 2015 nice.org.uk/guidance/ta352
 Vedolizumab for treating moderately to severely erely active Crohn's disease after prior therapy Technology appraisal guidance Published: 26 August 2015 nice.org.uk/guidance/ta352 NICE 2017. All rights
Vedolizumab for treating moderately to severely erely active Crohn's disease after prior therapy Technology appraisal guidance Published: 26 August 2015 nice.org.uk/guidance/ta352 NICE 2017. All rights
Clinical Policy: Vedolizumab (Entyvio) Reference Number: CP.PHAR.265 Effective Date: Last Review Date: Line of Business: Medicaid
 Clinical Policy: (Entyvio) Reference Number: CP.PHAR.265 Effective Date: 07.16 Last Review Date: 11.18 Line of Business: Medicaid Coding Implications Revision Log See Important Reminder at the end of this
Clinical Policy: (Entyvio) Reference Number: CP.PHAR.265 Effective Date: 07.16 Last Review Date: 11.18 Line of Business: Medicaid Coding Implications Revision Log See Important Reminder at the end of this
IBD Updates. Themes in IBD IBD management journey. New tools for therapeutic monitoring. First-line treatment in IBD
 IBD Updates Maria T. Abreu, MD University of Miami Miller School of Medicine Miami, Florida Themes in IBD 213 First-line treatment in IBD New tools for therapeutic monitoring Biologic therapy for CD and
IBD Updates Maria T. Abreu, MD University of Miami Miller School of Medicine Miami, Florida Themes in IBD 213 First-line treatment in IBD New tools for therapeutic monitoring Biologic therapy for CD and
Of Treatment For Inflammatory Bowel Diseases
 Balancing The Risks And Benefits Of Treatment For Inflammatory Bowel Diseases Corey A. Siegel, MD Assistant Professor of Medicine Dartmouth Medical School Director, Inflammatory Bowel Diseases Center Dartmouth-Hitchcock
Balancing The Risks And Benefits Of Treatment For Inflammatory Bowel Diseases Corey A. Siegel, MD Assistant Professor of Medicine Dartmouth Medical School Director, Inflammatory Bowel Diseases Center Dartmouth-Hitchcock
Scottish Medicines Consortium
 Scottish Medicines Consortium infliximab 100mg powder for intravenous infusion (Remicade ) No. (364/07) Schering-Plough UK Ltd 6 April 2007 The Scottish Medicines Consortium (SMC) has completed its assessment
Scottish Medicines Consortium infliximab 100mg powder for intravenous infusion (Remicade ) No. (364/07) Schering-Plough UK Ltd 6 April 2007 The Scottish Medicines Consortium (SMC) has completed its assessment
Clinical Study Low Body Mass Index Can Identify Majority of Osteoporotic Inflammatory Bowel Disease Patients Missed by Current Guidelines
 The Scientific World Journal Volume 2012, Article ID 807438, 6 pages doi:10.1100/2012/807438 The cientificworldjournal Clinical Study Low Body Mass Index Can Identify Majority of Osteoporotic Inflammatory
The Scientific World Journal Volume 2012, Article ID 807438, 6 pages doi:10.1100/2012/807438 The cientificworldjournal Clinical Study Low Body Mass Index Can Identify Majority of Osteoporotic Inflammatory
CCFA. Crohns Disease vs UC: What is the best treatment for me? November
 CCFA Crohns Disease vs UC: What is the best treatment for me? November 8 2009 Ellen J. Scherl,, MD, FACP,AGAF Roberts Inflammatory Bowel Disease Center Weill Medical College Cornell University New York
CCFA Crohns Disease vs UC: What is the best treatment for me? November 8 2009 Ellen J. Scherl,, MD, FACP,AGAF Roberts Inflammatory Bowel Disease Center Weill Medical College Cornell University New York
IBD Case Studies. David Rowbotham. Clinical Director & Consultant Gastroenterologist Dept of Gastroenterology & Hepatology Auckland City Hospital
 IBD Case Studies David Rowbotham Clinical Director & Consultant Gastroenterologist Dept of Gastroenterology & Hepatology Auckland City Hospital Dr David Rowbotham The Leeds Teaching Hospitals NHS Trust
IBD Case Studies David Rowbotham Clinical Director & Consultant Gastroenterologist Dept of Gastroenterology & Hepatology Auckland City Hospital Dr David Rowbotham The Leeds Teaching Hospitals NHS Trust
ULCERATIVE COLITIS. Sean Lynch, MD and Richard Bloomfeld, MD Wake Forest University School of Medicine Winston-Salem, NC
 ULCERATIVE COLITIS Sean Lynch, MD and Richard Bloomfeld, MD Wake Forest University School of Medicine Winston-Salem, NC What is Ulcerative Colitis? Ulcerative colitis (UC) is a disease marked by inflammation
ULCERATIVE COLITIS Sean Lynch, MD and Richard Bloomfeld, MD Wake Forest University School of Medicine Winston-Salem, NC What is Ulcerative Colitis? Ulcerative colitis (UC) is a disease marked by inflammation
Pharmacotherapy of Inflammatory Bowel Disorder
 PHARMACY / MEDICAL POLICY 5.01.563 Pharmacotherapy of Inflammatory Bowel Disorder Effective Date: June 9, 2019* Last Revised: Feb. 12, 2019 Replaces: Extracted from 5.01.550 RELATED MEDICAL POLICIES: 11.01.523
PHARMACY / MEDICAL POLICY 5.01.563 Pharmacotherapy of Inflammatory Bowel Disorder Effective Date: June 9, 2019* Last Revised: Feb. 12, 2019 Replaces: Extracted from 5.01.550 RELATED MEDICAL POLICIES: 11.01.523
Novel anti-inflammatory compounds for ulcerative colitis and rheumatoid arthritis therapy
 Novel anti-inflammatory compounds for ulcerative colitis and rheumatoid arthritis therapy JANSA, P. 1, HOLÝ, A. 1, ZÍDEK, Z. 2, JANEBA, Z. 1, KOLMAN, V. 1 1 Institute of Organic Chemistry and Biochemistry
Novel anti-inflammatory compounds for ulcerative colitis and rheumatoid arthritis therapy JANSA, P. 1, HOLÝ, A. 1, ZÍDEK, Z. 2, JANEBA, Z. 1, KOLMAN, V. 1 1 Institute of Organic Chemistry and Biochemistry
David Y. Graham, MD Baylor College of Medicine, Houston TX, USA
 Phase III Randomized, Double Blind, Placebo- Controlled, Multicenter, Parallel Group Study to Assess the Efficacy and Safety of Add-On Fixed-Dose Anti- Mycobacterial Therapy (RHB-104) in Moderately to
Phase III Randomized, Double Blind, Placebo- Controlled, Multicenter, Parallel Group Study to Assess the Efficacy and Safety of Add-On Fixed-Dose Anti- Mycobacterial Therapy (RHB-104) in Moderately to
Choosing and Positioning Biologic Therapy for Crohn s Disease: (Still) Looking for the Crystal Ball
 Choosing and Positioning Biologic Therapy for Crohn s Disease: (Still) Looking for the Crystal Ball Siddharth Singh, MD, MS Assistant Professor of Medicine Division of Gastroenterology Division of Biomedical
Choosing and Positioning Biologic Therapy for Crohn s Disease: (Still) Looking for the Crystal Ball Siddharth Singh, MD, MS Assistant Professor of Medicine Division of Gastroenterology Division of Biomedical
Prevalence of Nonadherence With Maintenance Mesalamine in Quiescent Ulcerative Colitis
 THE AMERICAN JOURNAL OF GASTROENTEROLOGY Vol. 96, No. 10, 2001 2001 by Am. Coll. of Gastroenterology ISSN 0002-9270/01/$20.00 Published by Elsevier Science Inc. PII S0002-9270(01)03245-2 Prevalence of
THE AMERICAN JOURNAL OF GASTROENTEROLOGY Vol. 96, No. 10, 2001 2001 by Am. Coll. of Gastroenterology ISSN 0002-9270/01/$20.00 Published by Elsevier Science Inc. PII S0002-9270(01)03245-2 Prevalence of
Disparities in Epilepsy Care: What is Known/Not Known December 4, 2011
 Disparities in Epilepsy Care: What is Known/Not Known December 4, 2011 Charles E. Begley, Ph.D. University of Texas School of Public Health Disclosure Name of Commercial Interest Type of Financial Relationship
Disparities in Epilepsy Care: What is Known/Not Known December 4, 2011 Charles E. Begley, Ph.D. University of Texas School of Public Health Disclosure Name of Commercial Interest Type of Financial Relationship
Chronic Pain and Opioid Use in Women Veterans: Is there an Association with Menopause?
 Chronic Pain and Opioid Use in Women Veterans: Is there an Association with Menopause? Carolyn Gibson, PhD, MPH San Francisco VA Health Care System and University of California, San Francisco NAMS Annual
Chronic Pain and Opioid Use in Women Veterans: Is there an Association with Menopause? Carolyn Gibson, PhD, MPH San Francisco VA Health Care System and University of California, San Francisco NAMS Annual
-2002: Rectal blood loss, UC? (no definite diagnosis) rectal mesalazine. -June 2008: Recurrence of rectal blood loss and urgency
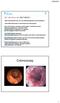 SD, male 40 yrs. old. (680718M467.) -2002: Rectal blood loss, UC? (no definite diagnosis) rectal mesalazine -June 2008: Recurrence of rectal blood loss and urgency Total colonoscopy: ulcerative rectitis,
SD, male 40 yrs. old. (680718M467.) -2002: Rectal blood loss, UC? (no definite diagnosis) rectal mesalazine -June 2008: Recurrence of rectal blood loss and urgency Total colonoscopy: ulcerative rectitis,
1 P a g e. Systemic Juvenile Idiopathic Arthritis (SJIA) (1.3) Patients 2 years of age and older with active systemic juvenile idiopathic arthritis.
 LENGTH OF AUTHORIZATION: Initial: 3 months for Crohn s or Ulcerative Colitis; 1 year for all other indications. Renewal: 1 year dependent upon medical records supporting response to therapy and review
LENGTH OF AUTHORIZATION: Initial: 3 months for Crohn s or Ulcerative Colitis; 1 year for all other indications. Renewal: 1 year dependent upon medical records supporting response to therapy and review
The following slides are provided as presented by the author during the live educa7onal ac7vity and are intended for reference purposes only.
 The following slides are provided as presented by the author during the live educa7onal ac7vity and are intended for reference purposes only. If you have any ques7ons, please contact Imedex via email at:
The following slides are provided as presented by the author during the live educa7onal ac7vity and are intended for reference purposes only. If you have any ques7ons, please contact Imedex via email at:
The 2017 CMS Merit-based Incentive Payment System includes 2 inflammatory bowel disease (IBD) measures.
 The 2017 CMS Merit-based Incentive Payment System includes 2 inflammatory bowel disease (IBD) measures. Measure Specifications for Registry Reporting MIPS #271: Inflammatory Bowel Disease (IBD): Preventive
The 2017 CMS Merit-based Incentive Payment System includes 2 inflammatory bowel disease (IBD) measures. Measure Specifications for Registry Reporting MIPS #271: Inflammatory Bowel Disease (IBD): Preventive
Center for Evidence-based Policy
 P&T Committee Brief Targeted Immune Modulators: Comparative Drug Class Review Alison Little, MD Center for Evidence-based Policy Oregon Health & Science University 3455 SW US Veterans Hospital Road, SN-4N
P&T Committee Brief Targeted Immune Modulators: Comparative Drug Class Review Alison Little, MD Center for Evidence-based Policy Oregon Health & Science University 3455 SW US Veterans Hospital Road, SN-4N
The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION. 3 October 2012
 The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 3 October 2012 REMICADE 100 mg, powder for concentrate for solution for infusion B/1 vial (CIP code: 562 070-1) Applicant:
The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 3 October 2012 REMICADE 100 mg, powder for concentrate for solution for infusion B/1 vial (CIP code: 562 070-1) Applicant:
ENTYVIO (VEDOLIZUMAB)
 ENTYVIO (VEDOLIZUMAB) UnitedHealthcare Community Plan Medical Benefit Drug Policy Policy Number: CS2017D0053F Effective Date: July 1, 2017 Table of Contents Page INSTRUCTIONS FOR USE... 1 BENEFIT CONSIDERATIONS...
ENTYVIO (VEDOLIZUMAB) UnitedHealthcare Community Plan Medical Benefit Drug Policy Policy Number: CS2017D0053F Effective Date: July 1, 2017 Table of Contents Page INSTRUCTIONS FOR USE... 1 BENEFIT CONSIDERATIONS...
Highlights of DDW 2015: Crohn s disease
 Highlights of DDW 2015: Crohn s disease Mark S. Silverberg, MD, PhD, FRCPC Associate Professor of Medicine, University of Toronto Staff Gastroenterologist, Mount Sinai Hospital Senior Investigator, Lunenfeld-Tanenbaum
Highlights of DDW 2015: Crohn s disease Mark S. Silverberg, MD, PhD, FRCPC Associate Professor of Medicine, University of Toronto Staff Gastroenterologist, Mount Sinai Hospital Senior Investigator, Lunenfeld-Tanenbaum
CLINICAL MEDICAL POLICY
 Policy Name: Policy Number: Responsible Department(s): CLINICAL MEDICAL POLICY Tysabri (natalizumab) MP-042-MD-WV Provider Notice Date: 10/01/2017 Original Effective Date: 11/01/2017 Annual Approval Date:
Policy Name: Policy Number: Responsible Department(s): CLINICAL MEDICAL POLICY Tysabri (natalizumab) MP-042-MD-WV Provider Notice Date: 10/01/2017 Original Effective Date: 11/01/2017 Annual Approval Date:
