COMMISSION REGULATION (EU) / of XXX
|
|
|
- Lorin Tyler
- 5 years ago
- Views:
Transcription
1 EUROPEAN COMMISSION Brussels, XXX SANTE/11077/2016 Rev. 1 [ ](2016) XXX draft COMMISSION REGULATION (EU) / of XXX amending Annexes II and III to Regulation (EC) No 396/2005 of the European Parliament and of the Council as regards maximum residue levels for acrinathrin, metalaxyl and thiabendazole in or on certain products (Text with EEA relevance) EN EN
2 COMMISSION REGULATION (EU) / of XXX amending Annexes II and III to Regulation (EC) No 396/2005 of the European Parliament and of the Council as regards maximum residue levels for acrinathrin, metalaxyl and thiabendazole in or on certain products (Text with EEA relevance) THE EUROPEAN COMMISSION, Having regard to the Treaty on the Functioning of the European Union, Having regard to Regulation (EC) No 396/2005 of the European Parliament and of the Council of 23 February 2005 on maximum residue levels of pesticides in or on food and feed of plant and animal origin and amending Council Directive 91/414/EEC 1, and in particular Article 14(1)(a) and Article 49(2) thereof, Whereas: (1) For acrinathrin, maximum residue levels (MRLs) were set in Part A of Annex III to Regulation (EC) No 396/2005. For metalaxyl and thiabendazole MRLs were set in Annex II and Part B of Annex III to that Regulation. (2) For acrinathrin, the European Food Safety Authority, hereinafter "the Authority", submitted a reasoned opinion on the review of the existing MRLs in accordance with Article 12(1) of Regulation (EC) No 396/ It proposed to change the residue definition. It identified concerning the MRLs for bananas, melons, peppers, watermelons, peaches and apricots a risk for consumers. It is therefore appropriate to lower these MRLs. For other products, it recommended raising or keeping the existing MRLs. It concluded that concerning the MRLs for lamb's lettuce, escarole, cress, rocket, red mustard, soya beans and all animal origin products, some information was not available and that further consideration by risk managers was required. As there is no risk for consumers, the MRLs for those products should be set in Annex II to Regulation (EC) No 396/2005 at the existing level or the level identified by the Authority. These MRLs will be reviewed; the review will take into account the information available within two years from the publication of this Regulation. The Authority concluded that regardless of the data gaps identified the critical good agriculture practice was not compliant with the approval restrictions for acrinathrin for pome fruits, apricots, cherries, peaches, plums, strawberries, bananas, garlic, onions, tomatoes, peppers, aubergines, okra (lady's fingers), cucurbits with edible peel, gherkins, courgettes, melons, pumpkins, watermelons, lettuce, beans (fresh, with pods) and soya beans and that further consideration by risk managers was required. The MRLs for these products should be set at the specific limit of determination. 1 2 OJ L 070, , p. 1. European Food Safety Authority; Reasoned opinion on the review of the existing maximum residue levels (MRLs) for acrinathrin according to Article 12 of Regulation (EC) No 396/2005. EFSA Journal 2015;13(7):4203. EN 2 EN
3 (3) For metalaxyl, the Authority submitted a reasoned opinion on the existing MRLs in accordance with Article 12(1) of Regulation (EC) No 396/ It proposed to change the residue definition and recommended lowering the MRLs for head cabbage, chinese cabbage, kohlrabi, spinach, beet leaves (chard), beans (fresh, with and without pods), peas (fresh, with and without pods), asparagus, leek, beans (dry), peas (dry), lupins (dry), linseed, poppy seed, rape seed, mustard seed, gold of pleasure, maize, swine meat and fat, bovine meat and fat, sheep meat and fat, goat meat and fat, poultry meat and fat, milk and birds eggs. It concluded that concerning the MRLs for grapefruits, oranges, lemons, limes, mandarins, apples, pears, table grapes, wine grapes, strawberries, onions, peppers, soya bean, swine liver and kidney, bovine liver and kidney, sheep liver and kidney and poultry liver some information was not available and that further consideration by risk managers was required. As there is no risk for consumers, the MRLs for those products should be set in Annex II to Regulation (EC) No 396/2005 at the existing level or the level identified by the Authority. These MRLs will be reviewed; the review will take into account the information available within two years from the publication of this Regulation. The Authority concluded that concerning the MRLs for quinces, medlar, loquat, avocados, gherkins, cotton seed, barley, buckwheat, millet, oats, rice, rye, sorghum, wheat, spices from seeds and sugar beet (root), no information was available and that further consideration by risk managers was required. The MRLs for these products should be set at the specific limit of determination. (4) For thiabendazole, the Authority submitted a reasoned opinion on the existing MRLs in accordance with Article 12(1) of Regulation (EC) No 396/ It identified concerning the MRLs for mangoes and cultivated fungi a risk for consumers. It is therefore appropriate to lower theses MRLs. The Authority proposed to change the residue definition for milk and other animal origin products. It recommended lowering the MRLs for apples, pears, potatoes, witloof/belgian endives, swine muscle and fat and poultry muscle and fat. For other products it recommended raising or keeping the existing MRLs. It concluded that concerning the MRLs for grapefruits, oranges, lemons, limes, mandarins, quinces, medlar, loquat, kumquats, avocados, bananas, papayas, potatoes, witloof/belgian endives and all animal origin products some information was not available and that further consideration by risk managers was required. As there is no risk for consumers, the MRLs for those products should be set in Annex II to Regulation (EC) No 396/2005 at the existing level or the level identified by the Authority. These MRLs will be reviewed; the review will take into account the information available within two years from the publication of this Regulation. (5) As regards products on which the use of the plant protection product concerned is not authorised, and for which no import tolerances or Codex maximum residue limits (CXLs) exist, MRLs should be set at the specific limit of determination or the default MRL should apply, as provided for in Article 18(1)(b) of Regulation (EC) No 396/ European Food Safety Authority; Reasoned opinion on combined review opinion on the review of the existing maximum residue levels (MRLs) for the active substances metalaxyl and metalaxyl-m. EFSA Journal 2015;13(4):4076. European Food Safety Authority; Reasoned opinion on the revision of the review of the existing maximum residue levels (MRLs) for thiabendazole according to Article 12 of Regulation (EC) No 396/2005. EFSA Journal 2016;14(6):4516. EN 3 EN
4 (6) The Commission consulted the European Union reference laboratories for residues of pesticides as regards the need to adapt certain limits of determination. As regards several substances, those laboratories concluded that for certain commodities technical development requires the setting of specific limits of determination. (7) Based on the reasoned opinions of the Authority and taking into account the factors relevant to the matter under consideration, the appropriate modifications to the MRLs fulfil the requirements of Article 14(2) of Regulation (EC) No 396/2005. (8) Through the World Trade Organisation, the trading partners of the Union were consulted on the new MRLs and their comments have been taken into account. (9) Regulation (EC) No 396/2005 should therefore be amended accordingly. (10) In order to allow for the normal marketing, processing and consumption of products, this Regulation should provide for a transitional arrangement for products which have been produced before the modification of the MRLs and for which information shows that a high level of consumer protection is maintained. Because a risk to consumers cannot be excluded at the current MRLs, the value of 0.01 mg/kg for acrinathrin in and on bananas, melons, peppers, watermelons, peaches and apricots and the value of 0.01 mg/kg for thiabendazole in and on mangoes and cultivated fungi should apply to all products from the date of application of this Regulation. (11) A reasonable period should be allowed to elapse before the modified MRLs become applicable in order to permit Member States, third countries and food business operators to prepare themselves to meet the new requirements which will result from the modification of the MRLs. (12) The measures provided for in this Regulation are in accordance with the opinion of the Standing Committee on Plants, Animals, Food and Feed, HAS ADOPTED THIS REGULATION: Article 1 Annexes II and III to Regulation (EC) No 396/2005 are amended in accordance with the Annex to this Regulation. Article 2 Regulation (EC) No 396/2005 as it stood before being amended by this Regulation shall continue to apply to products which were produced before [Office of Publication please insert date 6 months after entry into force of this Regulation]: (1) as regards the active substance metalaxyl in and on all products; (2) as regards the active substance achrinathrin in and all products except bananas, melons, peppers, watermelons, peaches and apricots; (3) as regards the active substance thiabendazole in and on all products except mangoes and cultivated fungi. Article 3 This Regulation shall enter into force on the twentieth day following that of its publication in the Official Journal of the European Union. EN 4 EN
5 It shall apply from [Office of Publication: please insert date 6 months after entry into force]. This Regulation shall be binding in its entirety and directly applicable in all Member States. Done at Brussels, For the Commission The President Jean-Claude JUNCKER EN 5 EN
COMMISSION REGULATION (EU) / of XXX
 EUROPEAN COMMISSION Brussels, XXX SANTE/10530/2015 Rev. 0 [ ](2015) XXX draft COMMISSION REGULATION (EU) / of XXX amending Annexes II and III to Regulation (EC) No 396/2005 of the European Parliament and
EUROPEAN COMMISSION Brussels, XXX SANTE/10530/2015 Rev. 0 [ ](2015) XXX draft COMMISSION REGULATION (EU) / of XXX amending Annexes II and III to Regulation (EC) No 396/2005 of the European Parliament and
Council of the European Union Brussels, 4 November 2015 (OR. en)
 Council of the European Union Brussels, 4 November 2015 (OR. en) 13706/15 AGRILEG 208 COVER NOTE From: European Commission date of receipt: 30 October 2015 To: No. Cion doc.: D041471/02 Subject: General
Council of the European Union Brussels, 4 November 2015 (OR. en) 13706/15 AGRILEG 208 COVER NOTE From: European Commission date of receipt: 30 October 2015 To: No. Cion doc.: D041471/02 Subject: General
COMMISSION REGULATION (EU) / of XXX
 EUROPEAN COMMISSION Brussels, XXX SANTE/11715/2017 rev.2 [ ](2018) XXX draft COMMISSION REGULATION (EU) / of XXX amending Annexes II, III and V to Regulation (EC) No 396/2005 of the European Parliament
EUROPEAN COMMISSION Brussels, XXX SANTE/11715/2017 rev.2 [ ](2018) XXX draft COMMISSION REGULATION (EU) / of XXX amending Annexes II, III and V to Regulation (EC) No 396/2005 of the European Parliament
Council of the European Union Brussels, 12 August 2014 (OR. en) Mr Uwe CORSEPIUS, Secretary-General of the Council of the European Union
 Council of the European Union Brussels, 12 August 2014 (OR. en) 12459/14 AGRILEG 168 COVER NOTE From: European Commission date of receipt: 8 August 2014 To: No. Cion doc.: D033914/02 Subject: Mr Uwe CORSEPIUS,
Council of the European Union Brussels, 12 August 2014 (OR. en) 12459/14 AGRILEG 168 COVER NOTE From: European Commission date of receipt: 8 August 2014 To: No. Cion doc.: D033914/02 Subject: Mr Uwe CORSEPIUS,
COMMISSION REGULATION (EU) / of XXX
 EUROPEAN COMMISSION Brussels, XXX SANTE/10154/2018 Rev. 2 [ ](2018) XXX draft COMMISSION REGULATION (EU) / of XXX amending Annexes II, III and V to Regulation (EC) No 396/2005 of the European Parliament
EUROPEAN COMMISSION Brussels, XXX SANTE/10154/2018 Rev. 2 [ ](2018) XXX draft COMMISSION REGULATION (EU) / of XXX amending Annexes II, III and V to Regulation (EC) No 396/2005 of the European Parliament
Council of the European Union Brussels, 5 December 2014 (OR. en) Mr Uwe CORSEPIUS, Secretary-General of the Council of the European Union
 Council of the European Union Brussels, 5 December 2014 (OR. en) 16594/14 AGRILEG 254 COVER NOTE From: European Commission date of receipt: 3 December 2014 To: No. Cion doc.: D035772/02 Subject: Mr Uwe
Council of the European Union Brussels, 5 December 2014 (OR. en) 16594/14 AGRILEG 254 COVER NOTE From: European Commission date of receipt: 3 December 2014 To: No. Cion doc.: D035772/02 Subject: Mr Uwe
COMMISSION REGULATION (EU) / of XXX
 EUROPEAN COMMISSION Brussels, XXX SANTE/10893/2018 Rev. 1 [ ](2018) XXX draft COMMISSION REGULATION (EU) / of XXX amending Annexes II, III, IV and V to Regulation (EC) No 396/2005 of the European Parliament
EUROPEAN COMMISSION Brussels, XXX SANTE/10893/2018 Rev. 1 [ ](2018) XXX draft COMMISSION REGULATION (EU) / of XXX amending Annexes II, III, IV and V to Regulation (EC) No 396/2005 of the European Parliament
COMMISSION REGULATION (EU) / of XXX
 EUROPEAN COMMISSION Brussels, XXX SANTE/12049/2017 Rev. 1 [ ](2017) XXX draft COMMISSION REGULATION (EU) / of XXX amending Annexes II, III and V to Regulation (EC) No 396/2005 of the European Parliament
EUROPEAN COMMISSION Brussels, XXX SANTE/12049/2017 Rev. 1 [ ](2017) XXX draft COMMISSION REGULATION (EU) / of XXX amending Annexes II, III and V to Regulation (EC) No 396/2005 of the European Parliament
Council of the European Union Brussels, 28 March 2018 (OR. en)
 Council of the European Union Brussels, 28 March 2018 (OR. en) 7563/18 AGRILEG 47 COVER NOTE From: European Commission date of receipt: 23 March 2018 To: No. Cion doc.: D055526/02 Subject: General Secretariat
Council of the European Union Brussels, 28 March 2018 (OR. en) 7563/18 AGRILEG 47 COVER NOTE From: European Commission date of receipt: 23 March 2018 To: No. Cion doc.: D055526/02 Subject: General Secretariat
Setting of new MRLs for fluxapyroxad (BAS 700 F) in various commodities of plant and animal origin 1
 : EFSA Journal 2011;9(6):2196 REASONED OPINION Setting of new MRLs for fluxapyroxad (BAS 700 F) in various commodities of plant and animal origin 1 European Food Safety Authority 2 European Food Safety
: EFSA Journal 2011;9(6):2196 REASONED OPINION Setting of new MRLs for fluxapyroxad (BAS 700 F) in various commodities of plant and animal origin 1 European Food Safety Authority 2 European Food Safety
Proposed Revision or Revocation of Maximum Residue Limits for Discontinued Agricultural Pest Control Products: Update 2
 Proposed Maximum Residue Limit PMRL2018-44 Proposed Revision or Revocation of Maximum Residue Limits for Discontinued Agricultural Pest Control Products: Update 2 (publié aussi en français) 8 November
Proposed Maximum Residue Limit PMRL2018-44 Proposed Revision or Revocation of Maximum Residue Limits for Discontinued Agricultural Pest Control Products: Update 2 (publié aussi en français) 8 November
COU CIL OF THE EUROPEA U IO. Brussels, 13 March 2012 (14.03) (OR. fr) 7585/12 AGRILEG 37
 COU CIL OF THE EUROPEA U IO Brussels, 13 March 2012 (14.03) (OR. fr) 7585/12 AGRILEG 37 COVER OTE from: European Commission date of receipt: 7 March 2012 to: General Secretariat of the Council No Cion
COU CIL OF THE EUROPEA U IO Brussels, 13 March 2012 (14.03) (OR. fr) 7585/12 AGRILEG 37 COVER OTE from: European Commission date of receipt: 7 March 2012 to: General Secretariat of the Council No Cion
WORKING DOCUMENT DOES NOT NECESSARILY REPRESENT THE VIEWS OF THE EUROPEAN COMMISSION SERVICES
 EUROPEAN COMMISSION Brussels, XXX SANTE/11728/2016 [ ](2016) XXX draft of XXX in certain raw apricot kernels and derived products (Text with EEA relevance) WORKING DOCUMT DOES NOT NECESSARILY REPREST THE
EUROPEAN COMMISSION Brussels, XXX SANTE/11728/2016 [ ](2016) XXX draft of XXX in certain raw apricot kernels and derived products (Text with EEA relevance) WORKING DOCUMT DOES NOT NECESSARILY REPREST THE
Delegations will find attached document D051559/03.
 Council of the European Union Brussels, 9 November 2017 (OR. en) 14153/17 DLEG 98 AGRI 614 SAN 406 COVER NOTE From: European Commission date of receipt: 8 November 2017 To: No. Cion doc.: D051559/03 Subject:
Council of the European Union Brussels, 9 November 2017 (OR. en) 14153/17 DLEG 98 AGRI 614 SAN 406 COVER NOTE From: European Commission date of receipt: 8 November 2017 To: No. Cion doc.: D051559/03 Subject:
Review of the existing maximum residue levels (MRLs) for fludioxonil according to Article 12 of Regulation (EC) No 396/2005 1
 EFSA Journal 2011;9(8):2335 REASONED OPINION Review of the existing maximum residue levels (MRLs) for fludioxonil according to Article 12 of Regulation (EC) No 396/2005 1 European Food Safety Authority
EFSA Journal 2011;9(8):2335 REASONED OPINION Review of the existing maximum residue levels (MRLs) for fludioxonil according to Article 12 of Regulation (EC) No 396/2005 1 European Food Safety Authority
Council of the European Union Brussels, 27 March 2017 (OR. en)
 Council of the European Union Brussels, 27 March 2017 (OR. en) 7686/17 DLEG 25 AGRI 162 SAN 121 COVER NOTE From: European Commission date of receipt: 24 March 2017 To: No. Cion doc.: D049176/01 Subject:
Council of the European Union Brussels, 27 March 2017 (OR. en) 7686/17 DLEG 25 AGRI 162 SAN 121 COVER NOTE From: European Commission date of receipt: 24 March 2017 To: No. Cion doc.: D049176/01 Subject:
Council of the European Union Brussels, 24 March 2015 (OR. en)
 Council of the European Union Brussels, 24 March 2015 (OR. en) 7498/15 DLEG 43 AGRI 154 SAN 84 COVER NOTE From: European Commission date of receipt: 24 March 2015 To: No. Cion doc.: D038008/02 Subject:
Council of the European Union Brussels, 24 March 2015 (OR. en) 7498/15 DLEG 43 AGRI 154 SAN 84 COVER NOTE From: European Commission date of receipt: 24 March 2015 To: No. Cion doc.: D038008/02 Subject:
Council of the European Union Brussels, 6 February 2017 (OR. en)
 Council of the European Union Brussels, 6 February 2017 (OR. en) 5966/17 AGRILEG 28 VETER 11 COVER NOTE From: European Commission date of receipt: 3 February 2017 To: General Secretariat of the Council
Council of the European Union Brussels, 6 February 2017 (OR. en) 5966/17 AGRILEG 28 VETER 11 COVER NOTE From: European Commission date of receipt: 3 February 2017 To: General Secretariat of the Council
COMMISSION REGULATION (EU) No /.. of XXX. amending Regulation (EC) No 1881/2006 as regards maximum levels of lead in certain foodstuffs
 EUROPEAN COMMISSION Brussels, XXX SANCO/10946/2014 [ ](2015) XXX COMMISSION REGULATION (EU) No /.. of XXX amending Regulation (EC) No 1881/2006 as regards maximum levels of lead in certain foodstuffs (Text
EUROPEAN COMMISSION Brussels, XXX SANCO/10946/2014 [ ](2015) XXX COMMISSION REGULATION (EU) No /.. of XXX amending Regulation (EC) No 1881/2006 as regards maximum levels of lead in certain foodstuffs (Text
Review of the existing maximum residue levels (MRLs) for pyraclostrobin according to Article 12 of Regulation (EC) No 396/2005 1
 EFSA Journal 2011;9(8):2344 REASONED OPINION Review of the existing maximum levels (MRLs) for pyraclostrobin according to Article 12 of Regulation (EC) No 396/2005 1 European Food Safety Authority 2, 3
EFSA Journal 2011;9(8):2344 REASONED OPINION Review of the existing maximum levels (MRLs) for pyraclostrobin according to Article 12 of Regulation (EC) No 396/2005 1 European Food Safety Authority 2, 3
COMMISSION DELEGATED REGULATION (EU) /... of XXX
 EUROPEAN COMMISSION Brussels, XXX SANTE/11481/2018 CIS (POOL/E1/2018/11481/11481-EN CIS.doc) [ ](2018) XXX draft COMMISSION DELEGATED REGULATION (EU) /... of XXX amending Commission Delegated Regulation
EUROPEAN COMMISSION Brussels, XXX SANTE/11481/2018 CIS (POOL/E1/2018/11481/11481-EN CIS.doc) [ ](2018) XXX draft COMMISSION DELEGATED REGULATION (EU) /... of XXX amending Commission Delegated Regulation
COMMISSION DELEGATED REGULATION (EU) /... of XXX
 EUROPEAN COMMISSION Brussels, XXX SANTE/11481/2018 CIS (POOL/E1/2018/11481/11481- CIS.doc) [ ](2018) XXX draft COMMISSION DELEGATED REGULATION (EU) /... of XXX amending Commission Delegated Regulation
EUROPEAN COMMISSION Brussels, XXX SANTE/11481/2018 CIS (POOL/E1/2018/11481/11481- CIS.doc) [ ](2018) XXX draft COMMISSION DELEGATED REGULATION (EU) /... of XXX amending Commission Delegated Regulation
COMMISSION REGULATION (EU) / of XXX
 EUROPEAN COMMISSION Brussels, XXX SANTE/11490/2017 [ ](2017) XXX draft COMMISSION REGULATION (EU) / of XXX refusing to authorise certain health claims made on foods, other than those referring to the reduction
EUROPEAN COMMISSION Brussels, XXX SANTE/11490/2017 [ ](2017) XXX draft COMMISSION REGULATION (EU) / of XXX refusing to authorise certain health claims made on foods, other than those referring to the reduction
Reasoned opinion on the review of the existing maximum residue levels (MRLs) for diquat according to Article 12 of Regulation (EC) No 396/2005 1
 EFSA Journal 2015;13(1):3972 REASONED OPINION Reasoned opinion on the review of the existing maximum residue levels (MRLs) for according to Article 12 of Regulation (EC) No 396/2005 1 European Food Safety
EFSA Journal 2015;13(1):3972 REASONED OPINION Reasoned opinion on the review of the existing maximum residue levels (MRLs) for according to Article 12 of Regulation (EC) No 396/2005 1 European Food Safety
Delegations will find attached document D042445/02.
 Council of the European Union Brussels, 11 December 2015 (OR. en) 15273/15 DLEG 164 AGRI 670 SAN 437 COVER NOTE From: European Commission date of receipt: 11 December 2015 To: No. Cion doc.: D042445/02
Council of the European Union Brussels, 11 December 2015 (OR. en) 15273/15 DLEG 164 AGRI 670 SAN 437 COVER NOTE From: European Commission date of receipt: 11 December 2015 To: No. Cion doc.: D042445/02
COMMISSION REGULATION (EU) / of XXX
 Ref. Ares(2016)5616438-28/09/2016 EUROPEAN COMMISSION Brussels, XXX [ ](2016) XXX draft COMMISSION REGULATION (EU) / of XXX amending Annex III to Directive 2008/98/EC of the European Parliament and of
Ref. Ares(2016)5616438-28/09/2016 EUROPEAN COMMISSION Brussels, XXX [ ](2016) XXX draft COMMISSION REGULATION (EU) / of XXX amending Annex III to Directive 2008/98/EC of the European Parliament and of
Delegations will find attached document D056135/03.
 Council of the European Union Brussels, 1 June 2018 (OR. en) 9586/18 DLEG 46 SAN 175 AGRI 260 COVER NOTE From: European Commission date of receipt: 30 May 2018 To: No. Cion doc.: D056135/03 Subject: General
Council of the European Union Brussels, 1 June 2018 (OR. en) 9586/18 DLEG 46 SAN 175 AGRI 260 COVER NOTE From: European Commission date of receipt: 30 May 2018 To: No. Cion doc.: D056135/03 Subject: General
COMMISSION REGULATION (EU) / of XXX
 EUROPEAN COMMISSION Brussels, XXX SANTE/11992/2017 Rev. 0 [ ](2017) XXX draft COMMISSION REGULATION (EU) / of XXX amending Annex II to Regulation (EC) No 1107/2009 by setting out scientific criteria for
EUROPEAN COMMISSION Brussels, XXX SANTE/11992/2017 Rev. 0 [ ](2017) XXX draft COMMISSION REGULATION (EU) / of XXX amending Annex II to Regulation (EC) No 1107/2009 by setting out scientific criteria for
Follow up assessment of MRLs for the active substance iprodione. European Food Safety Authority (EFSA)
 Page 1 of 24 EFSA Journal TECHNICAL REPORT APPROVED: 27 March 2018 doi:10.2903/sp.efsa.2018.en-1404 Abstract Follow up assessment of MRLs for the active substance iprodione European Food Safety Authority
Page 1 of 24 EFSA Journal TECHNICAL REPORT APPROVED: 27 March 2018 doi:10.2903/sp.efsa.2018.en-1404 Abstract Follow up assessment of MRLs for the active substance iprodione European Food Safety Authority
Official Journal of the European Union
 23.2.2018 EN L 53/69 COMMISSION IMPLEMENTING REGULATION (EU) 2018/243 of 15 February 2018 concerning the authorisation of 3-hydroxybutan-2-one, pentan-2,3-dione, 3,5-dimethyl cyclopentan-1,2-dione, hexan-3,4-dione,
23.2.2018 EN L 53/69 COMMISSION IMPLEMENTING REGULATION (EU) 2018/243 of 15 February 2018 concerning the authorisation of 3-hydroxybutan-2-one, pentan-2,3-dione, 3,5-dimethyl cyclopentan-1,2-dione, hexan-3,4-dione,
COMMISSION DELEGATED REGULATION (EU) /... of XXX
 EUROPEAN COMMISSION Brussels, XXX SANTE/11174/2016 (POOL/E1/2016/E1/11174/11174- EN.doc) [ ](2017) XXX draft COMMISSION DELEGATED REGULATION (EU) /... of XXX amending the Annex to Regulation (EU) No 609/2013
EUROPEAN COMMISSION Brussels, XXX SANTE/11174/2016 (POOL/E1/2016/E1/11174/11174- EN.doc) [ ](2017) XXX draft COMMISSION DELEGATED REGULATION (EU) /... of XXX amending the Annex to Regulation (EU) No 609/2013
(Text with EEA relevance)
 5.10.2018 L 251/13 COMMISSION REGULATION (EU) 2018/1481 of 4 October 2018 amending Annexes II and III to Regulation (EC) No 1333/2008 of the European Parliament and of the Council and the Annex to Commission
5.10.2018 L 251/13 COMMISSION REGULATION (EU) 2018/1481 of 4 October 2018 amending Annexes II and III to Regulation (EC) No 1333/2008 of the European Parliament and of the Council and the Annex to Commission
Delegations will find attached document D055983/01.
 Council of the European Union Brussels, 2 May 2018 (OR. en) 8547/18 DLEG 34 SAN 130 AGRI 208 COVER NOTE From: European Commission date of receipt: 30 April 2018 To: No. Cion doc.: D055983/01 Subject: General
Council of the European Union Brussels, 2 May 2018 (OR. en) 8547/18 DLEG 34 SAN 130 AGRI 208 COVER NOTE From: European Commission date of receipt: 30 April 2018 To: No. Cion doc.: D055983/01 Subject: General
Sample results. Actual results may vary. PATIENT INFORMATION DOB: AGE: GENDER: FASTING: Clinical Info: Test Name Result Flag Reference Range Lab
 Sample results. Actual results may vary. SPECIMEN INFORMATION SPECIMEN: REQUISITION: LAB REF NO: PATIENT INFORMATION DOB: AGE: GENDER: FASTING: REPORT STATUS: FINAL ORDERING PHYSICIAN CLIENT INFORMATION
Sample results. Actual results may vary. SPECIMEN INFORMATION SPECIMEN: REQUISITION: LAB REF NO: PATIENT INFORMATION DOB: AGE: GENDER: FASTING: REPORT STATUS: FINAL ORDERING PHYSICIAN CLIENT INFORMATION
COMMISSION REGULATION (EU) / of XXX. establishing mitigation measures and benchmark levels for the reduction of the presence of acrylamide in food
 EUROPEAN COMMISSION Brussels, XXX SANTE/11059/2016 Rev. 2 (POOL/E2/2016/11059/11059R2- EN.doc) D048379/05 [ ](2017) XXX draft COMMISSION REGULATION (EU) / of XXX establishing mitigation measures and benchmark
EUROPEAN COMMISSION Brussels, XXX SANTE/11059/2016 Rev. 2 (POOL/E2/2016/11059/11059R2- EN.doc) D048379/05 [ ](2017) XXX draft COMMISSION REGULATION (EU) / of XXX establishing mitigation measures and benchmark
COMMISSION REGULATION (EU) / of XXX. establishing mitigation measures and benchmark levels for the reduction of the presence of acrylamide in food
 EUROPEAN COMMISSION Brussels, XXX SANTE/11059/2016 Rev. 2 (POOL/E2/2016/11059/11059R2- EN.doc) D048379/05 [ ](2017) XXX draft COMMISSION REGULATION (EU) / of XXX establishing mitigation measures and benchmark
EUROPEAN COMMISSION Brussels, XXX SANTE/11059/2016 Rev. 2 (POOL/E2/2016/11059/11059R2- EN.doc) D048379/05 [ ](2017) XXX draft COMMISSION REGULATION (EU) / of XXX establishing mitigation measures and benchmark
Official Journal of the European Union
 L 79/10 30.3.2016 COMMISSION REGULATION (EU) 2016/452 of 29 March 2016 amending Annexes II and III to Regulation (EC) No 396/2005 of the European Parliament and of the Council as regards maximum residue
L 79/10 30.3.2016 COMMISSION REGULATION (EU) 2016/452 of 29 March 2016 amending Annexes II and III to Regulation (EC) No 396/2005 of the European Parliament and of the Council as regards maximum residue
COMMISSION REGULATION (EU) / of XXX. authorising a health claim made on foods and referring to the reduction of disease risk
 EUROPEAN COMMISSION Brussels, XXX SANTE/12053/2016 [ ](2016) XXX draft COMMISSION REGULATION (EU) / of XXX authorising a health claim made on foods and referring to the reduction of disease risk (Text
EUROPEAN COMMISSION Brussels, XXX SANTE/12053/2016 [ ](2016) XXX draft COMMISSION REGULATION (EU) / of XXX authorising a health claim made on foods and referring to the reduction of disease risk (Text
Official Journal of the European Union
 L 230/8 EN 25.8.2016 COMMISSION REGULATION (EU) 2016/1413 of 24 August 2016 amending Regulation (EU) No 432/2012 establishing a list of permitted health claims made on foods other than those referring
L 230/8 EN 25.8.2016 COMMISSION REGULATION (EU) 2016/1413 of 24 August 2016 amending Regulation (EU) No 432/2012 establishing a list of permitted health claims made on foods other than those referring
COMMISSION REGULATION (EU) / of XXX. authorising certain health claims made on foods and referring to children's development and health
 EUROPEAN COMMISSION Brussels, XXX SANTE/10891/2015 [ ](2015) XXX draft COMMISSION REGULATION (EU) / of XXX authorising certain health claims made on foods and referring to children's development and health
EUROPEAN COMMISSION Brussels, XXX SANTE/10891/2015 [ ](2015) XXX draft COMMISSION REGULATION (EU) / of XXX authorising certain health claims made on foods and referring to children's development and health
Delegations will find attached document D048379/05.
 Council of the European Union Brussels, 17 August 2017 (OR. en) 11651/17 AGRILEG 150 DENLEG 63 COVER NOTE From: European Commission date of receipt: 24 July 2017 To: No. Cion doc.: D048379/05 Subject:
Council of the European Union Brussels, 17 August 2017 (OR. en) 11651/17 AGRILEG 150 DENLEG 63 COVER NOTE From: European Commission date of receipt: 24 July 2017 To: No. Cion doc.: D048379/05 Subject:
Council of the European Union Brussels, 30 January 2018 (OR. en) Mr Jeppe TRANHOLM-MIKKELSEN, Secretary-General of the Council of the European Union
 Council of the European Union Brussels, 30 January 2018 (OR. en) 5777/18 DLEG 19 AGRI 55 SAN 48 COVER NOTE From: date of receipt: 29 January 2018 To: No. Cion doc.: Secretary-General of the European Commission,
Council of the European Union Brussels, 30 January 2018 (OR. en) 5777/18 DLEG 19 AGRI 55 SAN 48 COVER NOTE From: date of receipt: 29 January 2018 To: No. Cion doc.: Secretary-General of the European Commission,
COMMISSION DELEGATED REGULATION (EU).../... of XXX
 EUROPEAN COMMISSION Brussels, XXX SANTE/10355/2015 (POOL/E4/2015/10355/10355-EN. doc) [...1(2015) XXX draft COMMISSION DELEGATED REGULATION (EU).../... of XXX supplementmg Regulation (EU) No 609/2013 of
EUROPEAN COMMISSION Brussels, XXX SANTE/10355/2015 (POOL/E4/2015/10355/10355-EN. doc) [...1(2015) XXX draft COMMISSION DELEGATED REGULATION (EU).../... of XXX supplementmg Regulation (EU) No 609/2013 of
GREEN YELLOW RED PURPLE WHITE
 GREEN YELLOW RED PURPLE WHITE Broccoli, green cabbage, spinach, romaine lettuce, green peas, kiwi fruit, swiss chard Avoid cancer of the prostate and colon (large intestine) Develop and maintain healthy
GREEN YELLOW RED PURPLE WHITE Broccoli, green cabbage, spinach, romaine lettuce, green peas, kiwi fruit, swiss chard Avoid cancer of the prostate and colon (large intestine) Develop and maintain healthy
European Union comments for the. CODEX COMMITTEE ON PESTICIDE RESIDUES 44th Session. Shanghai, China, April 2012.
 - 1-16/04/2012 European Union comments for the CODEX COMMITTEE ON PESTICIDE RESIDUES 44th Session Shanghai, China, 23-28 April 2012 Agenda Item 6 a) Draft and Proposed Draft Maximum Residue Limits for
- 1-16/04/2012 European Union comments for the CODEX COMMITTEE ON PESTICIDE RESIDUES 44th Session Shanghai, China, 23-28 April 2012 Agenda Item 6 a) Draft and Proposed Draft Maximum Residue Limits for
Modification of the existing MRLs for chlorpyrifos-methyl in various crops 1
 EFSA Journal 2011;9(6):2219 REASONED OPINION Modification of the existing MRLs for chlorpyrifos-methyl in various crops 1 European Food Safety Authority 2 European Food Safety Authority (EFSA), Parma,
EFSA Journal 2011;9(6):2219 REASONED OPINION Modification of the existing MRLs for chlorpyrifos-methyl in various crops 1 European Food Safety Authority 2 European Food Safety Authority (EFSA), Parma,
(Text with EEA relevance) (2014/798/EU)
 L 330/50 COMMISSION IMPLEMTING DECISION of 13 November 2014 amending Annex F to Council Directive 64/432/EEC as regards the format of the model health certificates for intra-union trade in bovine animals
L 330/50 COMMISSION IMPLEMTING DECISION of 13 November 2014 amending Annex F to Council Directive 64/432/EEC as regards the format of the model health certificates for intra-union trade in bovine animals
13480/12 PM/ns 1 DG B 4B
 COUNCIL OF THE EUROPEAN UNION Brussels, 10 September 2012 13480/12 DENLEG 80 AGRI 553 COVER NOTE from: European Commission date of receipt: 22 August 2012 to: General Secretariat of the Council No Cion
COUNCIL OF THE EUROPEAN UNION Brussels, 10 September 2012 13480/12 DENLEG 80 AGRI 553 COVER NOTE from: European Commission date of receipt: 22 August 2012 to: General Secretariat of the Council No Cion
EVEN QUICKER,CHEAPER AND SAFER GERMAN-MADE FOR YOUR MULTIPLE METHODS
 EVEN QUICKER,CHEAPER AND SAFER GERMAN-MADE FOR YOUR MULTIPLE METHODS THE PROCESS AND ITS VARIANTS The QuEChERS method, which concerns the analysis of pesticides and other organic contaminants in foodstuffs
EVEN QUICKER,CHEAPER AND SAFER GERMAN-MADE FOR YOUR MULTIPLE METHODS THE PROCESS AND ITS VARIANTS The QuEChERS method, which concerns the analysis of pesticides and other organic contaminants in foodstuffs
APPROVED: 4 December 2015 PUBLISHED: 9 December 2015
 REASONED OPINION APPROVED: 4 December 2015 PUBLISHED: 9 December 2015 doi:10.2903/j.efsa.2015.4356 Review of the existing maximum residue levels for sodium 5-nitroguaiacolate, sodium o-nitrophenolate and
REASONED OPINION APPROVED: 4 December 2015 PUBLISHED: 9 December 2015 doi:10.2903/j.efsa.2015.4356 Review of the existing maximum residue levels for sodium 5-nitroguaiacolate, sodium o-nitrophenolate and
Review of the existing maximum residue levels (MRLs) for oxamyl according to Article 12 of Regulation (EC) No 396/2005 1
 REASONED OPINION Review of the existing maximum residue levels (MRLs) for oxamyl according to Article 12 of Regulation (EC) No 396/2005 1 European Food Safety Authority 2, 3 European Food Safety Authority
REASONED OPINION Review of the existing maximum residue levels (MRLs) for oxamyl according to Article 12 of Regulation (EC) No 396/2005 1 European Food Safety Authority 2, 3 European Food Safety Authority
COMMISSION REGULATION (EU) / of XXX
 ² EUROPEAN COMMISSION Brussels, XXX SANTE/00131/2015 REV1 [ ](2015) XXX draft COMMISSION REGULATION (EU) / of XXX amending Section E of Chapter V of Annex IV to Regulation (EC) No 999/2001 of the European
² EUROPEAN COMMISSION Brussels, XXX SANTE/00131/2015 REV1 [ ](2015) XXX draft COMMISSION REGULATION (EU) / of XXX amending Section E of Chapter V of Annex IV to Regulation (EC) No 999/2001 of the European
Fluopyram FLUOPYRAM (243)
 Fluopyram 163 5.19 FLUOPYRAM (243) RESIDUE AND ANALYTICAL ASPECTS Fluopyram, a pyridylethylamide broad spectrum fungicide was evaluated for the first time by the 2010 JMPR, where an ADI of 0 0.01 mg/kg
Fluopyram 163 5.19 FLUOPYRAM (243) RESIDUE AND ANALYTICAL ASPECTS Fluopyram, a pyridylethylamide broad spectrum fungicide was evaluated for the first time by the 2010 JMPR, where an ADI of 0 0.01 mg/kg
Pesticide Monitoring Program: Design Assessment 1
 EFSA Journal 2015;13(2):4005 ABSTRACT SCIENTIFIC REPORT OF EFSA Pesticide Monitoring Program: Design Assessment 1 European Food Safety Authority 2, 3 European Food Safety Authority (EFSA), Parma, Italy
EFSA Journal 2015;13(2):4005 ABSTRACT SCIENTIFIC REPORT OF EFSA Pesticide Monitoring Program: Design Assessment 1 European Food Safety Authority 2, 3 European Food Safety Authority (EFSA), Parma, Italy
COMMISSION REGULATION (EU) No /.. of XXX
 EUROPEAN COMMISSION Brussels, XXX [ ] [ ](2013) XXX draft COMMISSION REGULATION (EU) No /.. of XXX amending Annex III to Regulation (EC) No 1925/2006 of the European Parliament and of the Council of 20
EUROPEAN COMMISSION Brussels, XXX [ ] [ ](2013) XXX draft COMMISSION REGULATION (EU) No /.. of XXX amending Annex III to Regulation (EC) No 1925/2006 of the European Parliament and of the Council of 20
COMMISSION REGULATION (EU) / of XXX
 EUROPEAN COMMISSION Brussels, XXX SANTE/10747/2018 CIS (POOL/E2/2018/10747/10747-EN.doc) [ ](2018) XXX draft COMMISSION REGULATION (EU) / of XXX amending and correcting Regulation (EU) No 10/2011 on plastic
EUROPEAN COMMISSION Brussels, XXX SANTE/10747/2018 CIS (POOL/E2/2018/10747/10747-EN.doc) [ ](2018) XXX draft COMMISSION REGULATION (EU) / of XXX amending and correcting Regulation (EU) No 10/2011 on plastic
13267/11 PM/tl 1 DG I
 COUNCIL OF THE EUROPEAN UNION Brussels, 27 July 2011 13267/11 DLEG 109 COVER NOTE from: European Commission date of receipt: 22 July 2011 to: General Secretariat of the Council No Cion doc.: D015369/01
COUNCIL OF THE EUROPEAN UNION Brussels, 27 July 2011 13267/11 DLEG 109 COVER NOTE from: European Commission date of receipt: 22 July 2011 to: General Secretariat of the Council No Cion doc.: D015369/01
Review of the existing maximum residue levels for copper compounds according to Article 12 of Regulation (EC) No 396/2005
 REASONED OPINION ADOPTED: 1 March 2018 doi: 10.2903/j.efsa.2018.5212 Review of the existing imum residue levels for copper compounds according to Article 12 of Regulation (EC) No 396/2005 European Food
REASONED OPINION ADOPTED: 1 March 2018 doi: 10.2903/j.efsa.2018.5212 Review of the existing imum residue levels for copper compounds according to Article 12 of Regulation (EC) No 396/2005 European Food
374 Saflufenacil Short-term dietary exposure
 374 Saflufenacil Short-term dietary exposure The 2011 JMPR decided that an acute reference dose is unnecessary for saflufenacil. The Meeting therefore concluded that the short-term dietary exposure to
374 Saflufenacil Short-term dietary exposure The 2011 JMPR decided that an acute reference dose is unnecessary for saflufenacil. The Meeting therefore concluded that the short-term dietary exposure to
588G: Dietary Antigen Testing: Sensitivity and Complement 1/5. Dietary Antigen Exposure by Food Group
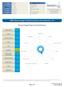 PATIENT NAME: CLINIC: DOB: SAMPLE DATE: RECEIVE DATE: REPORT DATE: R //2 /28/27 /29/27 /3/27 Dunwoody Labs 9 Dunwoody Park Suite 2 Dunwoody, GA 3338 USA Phone: 6787366374 Fax: 776747 Nine Dunwoody Park,
PATIENT NAME: CLINIC: DOB: SAMPLE DATE: RECEIVE DATE: REPORT DATE: R //2 /28/27 /29/27 /3/27 Dunwoody Labs 9 Dunwoody Park Suite 2 Dunwoody, GA 3338 USA Phone: 6787366374 Fax: 776747 Nine Dunwoody Park,
COMMISSION IMPLEMENTING DECISION. of XXX
 EUROPEAN COMMISSION Brussels, XXX SANTE/11331/2017 (POOL/E3/2017/11331/11331-EN.doc) [ ](2017) XXX draft COMMISSION IMPLEMENTING DECISION of XXX renewing the authorisation for the placing on the market
EUROPEAN COMMISSION Brussels, XXX SANTE/11331/2017 (POOL/E3/2017/11331/11331-EN.doc) [ ](2017) XXX draft COMMISSION IMPLEMENTING DECISION of XXX renewing the authorisation for the placing on the market
REQUIREMENTS FOR ACCREDITED LABORATORIES APPLYING FOR A FLEXIBLE SCOPE FOR ANALYSIS OF PESTICIDE RESIDUES IN FOOD AND FEED
 BELAC 2-104 Rev 2-2009 REQUIREMENTS FOR ACCREDITED LABORATORIES APPLYING FOR A FLEXIBLE SCOPE FOR ANALYSIS OF PESTICIDE RESIDUES IN FOOD AND FEED English translation for information only French and Dutch
BELAC 2-104 Rev 2-2009 REQUIREMENTS FOR ACCREDITED LABORATORIES APPLYING FOR A FLEXIBLE SCOPE FOR ANALYSIS OF PESTICIDE RESIDUES IN FOOD AND FEED English translation for information only French and Dutch
Official Journal of the European Union. (Non-legislative acts) REGULATIONS
 27.1.2016 L 20/1 II (Non-legislative acts) REGULATIONS COMMISSION REGULATION (EU) 2016/71 of 26 January 2016 amending Annexes II, III and V to Regulation (EC) No 396/2005 of the European Parliament and
27.1.2016 L 20/1 II (Non-legislative acts) REGULATIONS COMMISSION REGULATION (EU) 2016/71 of 26 January 2016 amending Annexes II, III and V to Regulation (EC) No 396/2005 of the European Parliament and
(Text with EEA relevance)
 L 204/36 EN COMMISSION IMPLEMENTING REGULATION (EU) 2018/1122 of 10 August 2018 authorising the placing on the market of pyrroloquinoline quinone disodium salt as a novel food under Regulation (EU) 2015/2283
L 204/36 EN COMMISSION IMPLEMENTING REGULATION (EU) 2018/1122 of 10 August 2018 authorising the placing on the market of pyrroloquinoline quinone disodium salt as a novel food under Regulation (EU) 2015/2283
REASONED OPINION. European Food Safety Authority 2, 3. European Food Safety Authority (EFSA), Parma, Italy
 EFSA Journal 2013;11(7):3339 REASONED OPINION Reasoned opinion on the review of the existing maximum residue levels (MRLs) for methyl bromide according to Article 12 of Regulation (EC) No 396/2005 1 European
EFSA Journal 2013;11(7):3339 REASONED OPINION Reasoned opinion on the review of the existing maximum residue levels (MRLs) for methyl bromide according to Article 12 of Regulation (EC) No 396/2005 1 European
COMMISSION IMPLEMENTING REGULATION (EU)
 EN 31.10.2013 Official Journal of the European Union L 289/33 COMMISSION IMPLEMENTING REGULATION (EU) No 1060/2013 of 29 October 2013 concerning the of bentonite as a feed additive for all animal (Text
EN 31.10.2013 Official Journal of the European Union L 289/33 COMMISSION IMPLEMENTING REGULATION (EU) No 1060/2013 of 29 October 2013 concerning the of bentonite as a feed additive for all animal (Text
Review of the existing maximum residue levels (MRLs) for cyromazine according to Article 12 of Regulation (EC) No 396/2005 1
 EFSA Journal 2011;9(7):2326 REASONED OPINION Review of the existing maximum residue levels (MRLs) for cyromazine according to Article 12 of Regulation (EC) No 396/2005 1 European Food Safety Authority
EFSA Journal 2011;9(7):2326 REASONED OPINION Review of the existing maximum residue levels (MRLs) for cyromazine according to Article 12 of Regulation (EC) No 396/2005 1 European Food Safety Authority
COMMISSION REGULATION (EU) / of XXX. authorising a health claim made on foods and referring to the reduction of disease risk
 EUROPEAN COMMISSION Brussels, XXX SANTE/12053/2016 CIS (POOL/E1/2016/12053/12053-EN CIS.doc) [ ](2017) XXX draft COMMISSION REGULATION (EU) / of XXX authorising a health claim made on foods and referring
EUROPEAN COMMISSION Brussels, XXX SANTE/12053/2016 CIS (POOL/E1/2016/12053/12053-EN CIS.doc) [ ](2017) XXX draft COMMISSION REGULATION (EU) / of XXX authorising a health claim made on foods and referring
COMMISSION REGULATION (EU)
 23.10.2010 Official Journal of the European Union L 279/3 REGULATIONS COMMISSION REGULATION (EU) No 955/2010 of 22 October 2010 amending Regulation (EC) No 798/2008 as regards the use of vaccines against
23.10.2010 Official Journal of the European Union L 279/3 REGULATIONS COMMISSION REGULATION (EU) No 955/2010 of 22 October 2010 amending Regulation (EC) No 798/2008 as regards the use of vaccines against
COMMISSION IMPLEMENTING REGULATION (EU) /... of XXX
 EUROPEAN COMMISSION Brussels, XXX [ ](2018) XXX draft COMMISSION IMPLEMENTING REGULATION (EU) /... of XXX amending Regulation (EC) No 889/2008 laying down detailed rules for the implementation of Council
EUROPEAN COMMISSION Brussels, XXX [ ](2018) XXX draft COMMISSION IMPLEMENTING REGULATION (EU) /... of XXX amending Regulation (EC) No 889/2008 laying down detailed rules for the implementation of Council
Official Journal of the European Union
 L 96/44 COMMISSION REGULATION (EU) 2017/627 of 3 April 2017 amending Annexes II, III and V to Regulation (EC) No 396/2005 of the European Parliament and of the Council as regards maximum residue levels
L 96/44 COMMISSION REGULATION (EU) 2017/627 of 3 April 2017 amending Annexes II, III and V to Regulation (EC) No 396/2005 of the European Parliament and of the Council as regards maximum residue levels
LAWS, STANDARDS & REGULATIONS
 Appendixes APPENDIX 1: LAWS, STANDARDS & REGULATIONS The United States enjoys one of the safest food supplies in the world. The laws and regulations required to achieve that safe food supply are lengthy
Appendixes APPENDIX 1: LAWS, STANDARDS & REGULATIONS The United States enjoys one of the safest food supplies in the world. The laws and regulations required to achieve that safe food supply are lengthy
Boscalid BOSCALID (221)
 Boscalid 49 5.3 BOSCALID (221) RESIDUE AND ANALYTICAL ASPECTS Boscalid is a systemic fungicide first evaluated by JMPR in 2006 for residues and toxicology as a new active substance. An ADI of 0 0.04 mg/kg
Boscalid 49 5.3 BOSCALID (221) RESIDUE AND ANALYTICAL ASPECTS Boscalid is a systemic fungicide first evaluated by JMPR in 2006 for residues and toxicology as a new active substance. An ADI of 0 0.04 mg/kg
Activity #4: Healthy Food Festival!
 Activity #4: Healthy Food Festival! Activity Overview In this activity, students apply their new learning to create fun ways to eat the rainbow during the week. Students are encouraged to consider the
Activity #4: Healthy Food Festival! Activity Overview In this activity, students apply their new learning to create fun ways to eat the rainbow during the week. Students are encouraged to consider the
Combined review of the existing maximum residue levels (MRLs) for the active substances metalaxyl and metalaxyl-m
 REASONED OPINION ADOPTED: 1 April 2015 PUBLISHED: 2 April 2015 doi:10.2903/j.efsa.2015.4076 Combined review of the existing maximum residue levels (MRLs) for the active substances metalaxyl and metalaxyl-m
REASONED OPINION ADOPTED: 1 April 2015 PUBLISHED: 2 April 2015 doi:10.2903/j.efsa.2015.4076 Combined review of the existing maximum residue levels (MRLs) for the active substances metalaxyl and metalaxyl-m
Quality Certification in Italy and in the Mediterranean countries: a way for accessing foreign markets. Davide Pierleoni
 Quality Certification in Italy and in the Mediterranean countries: a way for accessing foreign markets Davide Pierleoni Vice President Istituto Mediterraneo di Certificazione srl Presentation of IMC IMC
Quality Certification in Italy and in the Mediterranean countries: a way for accessing foreign markets Davide Pierleoni Vice President Istituto Mediterraneo di Certificazione srl Presentation of IMC IMC
COMMISSION REGULATION (EU) / of XXX
 EUROPEAN COMMISSION Brussels, XXX SANTE/10057/2017 (POOL/E5/2017/10057/10057-.doc) D049730/04 [ ](2017) XXX draft COMMISSION REGULATION (EU) / of XXX amending Annex I to Directive 2002/32/EC of the European
EUROPEAN COMMISSION Brussels, XXX SANTE/10057/2017 (POOL/E5/2017/10057/10057-.doc) D049730/04 [ ](2017) XXX draft COMMISSION REGULATION (EU) / of XXX amending Annex I to Directive 2002/32/EC of the European
COMMISSION REGULATION (EU) / of XXX
 Ref. Ares(2016)6396619-14/11/2016 EUROPEAN COMMISSION Brussels, XXX SANTE/10539/2016 CIS Rev. 3 (POOL/G4/2016/10539/10539R4-EN CIS.doc) [ ](2016) XXX draft COMMISSION REGULATION (EU) / of XXX amending
Ref. Ares(2016)6396619-14/11/2016 EUROPEAN COMMISSION Brussels, XXX SANTE/10539/2016 CIS Rev. 3 (POOL/G4/2016/10539/10539R4-EN CIS.doc) [ ](2016) XXX draft COMMISSION REGULATION (EU) / of XXX amending
National reporting 2014 Pesticide residues in food Federal Republic of Germany
 National reporting 2014 Pesticide residues in food Federal Republic of Germany Summary The report presents the results of the analysis of food for pesticide residues. In accordance with Regulation (EC)
National reporting 2014 Pesticide residues in food Federal Republic of Germany Summary The report presents the results of the analysis of food for pesticide residues. In accordance with Regulation (EC)
Official Journal of the European Union. (Non-legislative acts) REGULATIONS
 22.8.2017 EN L 216/1 II (Non-legislative acts) REGULATIONS COMMISSION IMPLEMENTING REGULATION (EU) 2017/1490 of 21 August 2017 concerning the of manganous chloride tetrahydrate, manganese (II) oxide, manganous
22.8.2017 EN L 216/1 II (Non-legislative acts) REGULATIONS COMMISSION IMPLEMENTING REGULATION (EU) 2017/1490 of 21 August 2017 concerning the of manganous chloride tetrahydrate, manganese (II) oxide, manganous
REASONED OPINION. European Food Safety Authority 2, 3. European Food Safety Authority (EFSA), Parma, Italy
 EFSA Journal 2012;10(7):2841 REASONED OPINION Reasoned opinion on the review of the existing maximum residue levels (MRLs) for paraffin oil (CAS 64742-54-7) according to Article 12 of Regulation (EC) No
EFSA Journal 2012;10(7):2841 REASONED OPINION Reasoned opinion on the review of the existing maximum residue levels (MRLs) for paraffin oil (CAS 64742-54-7) according to Article 12 of Regulation (EC) No
COMMISSION REGULATION (EU) / of XXX
 Ref. Ares(2017)4140854-23/08/2017 EUROPEAN COMMISSION Brussels, XXX SANTE/10578/2017 Rev. 2 CIS (POOL/E2/2017/10578/10578R2-EN CIS.doc) [ ](2017) XXX draft COMMISSION REGULATION (EU) / of XXX on the use
Ref. Ares(2017)4140854-23/08/2017 EUROPEAN COMMISSION Brussels, XXX SANTE/10578/2017 Rev. 2 CIS (POOL/E2/2017/10578/10578R2-EN CIS.doc) [ ](2017) XXX draft COMMISSION REGULATION (EU) / of XXX on the use
COMMISSION REGULATION (EU) / of XXX
 Ref. Ares(2017)4140854-23/08/2017 EUROPEAN COMMISSION Brussels, XXX SANTE/10578/2017 Rev. 2 CIS (POOL/E2/2017/10578/10578R2-EN CIS.doc) [ ](2017) XXX draft COMMISSION REGULATION (EU) / of XXX on the use
Ref. Ares(2017)4140854-23/08/2017 EUROPEAN COMMISSION Brussels, XXX SANTE/10578/2017 Rev. 2 CIS (POOL/E2/2017/10578/10578R2-EN CIS.doc) [ ](2017) XXX draft COMMISSION REGULATION (EU) / of XXX on the use
L 113/18 EN Official Journal of the European Union COMMISSION REGULATION (EU) 2017/752 of 28 April 2017 amending and correcting Regulation (
 L 113/18 EN 29.4.2017 COMMISSION REGULATION (EU) 2017/752 of 28 April 2017 amending and correcting Regulation (EU) No 10/2011 on plastic materials and articles intended to come into contact with food (Text
L 113/18 EN 29.4.2017 COMMISSION REGULATION (EU) 2017/752 of 28 April 2017 amending and correcting Regulation (EU) No 10/2011 on plastic materials and articles intended to come into contact with food (Text
5.20 PYRACLOSTROBIN (210)
 Pyraclostrobin 213 5.20 PYRACLOSTROBIN (210) RESIDUE AND ANALYTICAL ASPECTS Pyraclostrobin was first evaluated by JMPR in 2003 when an ADI of 0 0.03mg/kg bw and an ARfD of 0.05 mg/kg bw were established,
Pyraclostrobin 213 5.20 PYRACLOSTROBIN (210) RESIDUE AND ANALYTICAL ASPECTS Pyraclostrobin was first evaluated by JMPR in 2003 when an ADI of 0 0.03mg/kg bw and an ARfD of 0.05 mg/kg bw were established,
Review of the existing maximum residue levels (MRLs) for 2,4-D according to Article 12 of Regulation (EC) No 396/2005 1
 EFSA Journal 2011;9(11):2431 REASONED OPINION Review of the existing maximum residue levels (MRLs) for 2,4-D according to Article 12 of Regulation (EC) No 396/2005 1 European Food Safety Authority 2, 3
EFSA Journal 2011;9(11):2431 REASONED OPINION Review of the existing maximum residue levels (MRLs) for 2,4-D according to Article 12 of Regulation (EC) No 396/2005 1 European Food Safety Authority 2, 3
Du r i n g the past tw^o years there has been a decided increase
 THE VITAMIN A, VITAMIN Bi (THIAMIN), VITAMIN C (ASCORBIC ACID) AND RIBO FLA V IN CO N TEN T OF COMMON FOODS A S U M M A R Y O F R E P R E S E N T A T IV E V A L U E S H a z e l E. M u n s e l l "^ Du r
THE VITAMIN A, VITAMIN Bi (THIAMIN), VITAMIN C (ASCORBIC ACID) AND RIBO FLA V IN CO N TEN T OF COMMON FOODS A S U M M A R Y O F R E P R E S E N T A T IV E V A L U E S H a z e l E. M u n s e l l "^ Du r
REASONED OPINION. European Food Safety Authority 2, ABSTRACT. European Food Safety Authority (EFSA), Parma, Italy KEY WORDS
 EFSA Journal 2012;10(10):2920 REASONED OPINION Reasoned opinion on the modification of the existing MRL(s) for in citrus fruit, pome fruit, stone fruit, grapes, hops, strawberries, tomatoes, peppers, aubergines,
EFSA Journal 2012;10(10):2920 REASONED OPINION Reasoned opinion on the modification of the existing MRL(s) for in citrus fruit, pome fruit, stone fruit, grapes, hops, strawberries, tomatoes, peppers, aubergines,
COMMISSION REGULATION (EU) No /.. of XXX
 EUROPEAN COMMISSION Brussels, XXX SANCO/11214/2014 (POOL/E3/2014/11214/11214-EN.doc) D036822/04 [ ](2014) XXX draft COMMISSION REGULATION (EU) No /.. of XXX amending and correcting Annexes II and III to
EUROPEAN COMMISSION Brussels, XXX SANCO/11214/2014 (POOL/E3/2014/11214/11214-EN.doc) D036822/04 [ ](2014) XXX draft COMMISSION REGULATION (EU) No /.. of XXX amending and correcting Annexes II and III to
COMMISSION REGULATION (EU) No /.. of XXX. amending Regulation (EC) No 1881/2006 as regards maximum levels of cadmium in foodstuffs
 EUROPEAN COMMISSION Brussels, XXX SANCO/10617/2009 rev.09 [ ](2013) XXX draft COMMISSION REGULATION (EU) No /.. of XXX amending Regulation (EC) No 1881/2006 as regards maximum levels of cadmium in foodstuffs
EUROPEAN COMMISSION Brussels, XXX SANCO/10617/2009 rev.09 [ ](2013) XXX draft COMMISSION REGULATION (EU) No /.. of XXX amending Regulation (EC) No 1881/2006 as regards maximum levels of cadmium in foodstuffs
Modification of the existing MRLs for pyraclostrobin in various crops 1
 EFSA Journal 2011;9(3):2120 REASONED OPINION Modification of the existing MRLs for pyraclostrobin in various crops 1 European Food Safety Authority 2 European Food Safety Authority (EFSA), Parma, Italy
EFSA Journal 2011;9(3):2120 REASONED OPINION Modification of the existing MRLs for pyraclostrobin in various crops 1 European Food Safety Authority 2 European Food Safety Authority (EFSA), Parma, Italy
The Daniel Fast "Fasting For Health And Healing"
 The Daniel Fast "Fasting For Health And Healing" Starting Monday January 3, 2011 at Midnight Ending Sunday January 23, 2011 at 11:59 pm This information was primarily taken from the book "Fasting For Spiritual
The Daniel Fast "Fasting For Health And Healing" Starting Monday January 3, 2011 at Midnight Ending Sunday January 23, 2011 at 11:59 pm This information was primarily taken from the book "Fasting For Spiritual
Official Journal of the European Union. (Non-legislative acts) REGULATIONS
 29.6.2016 L 172/1 II (Non-legislative acts) REGULATIONS COMMISSION REGULATION (EU) 2016/1015 of 17 June 2016 amending Annexes II and III to Regulation (EC) No 396/2005 of the European Parliament and of
29.6.2016 L 172/1 II (Non-legislative acts) REGULATIONS COMMISSION REGULATION (EU) 2016/1015 of 17 June 2016 amending Annexes II and III to Regulation (EC) No 396/2005 of the European Parliament and of
European Community Positions for the 41 st Session of the Codex Committee on Pesticide Residues Beijing, China April 2009
 European Community Positions for the 41 st Session of the Codex Committee on Pesticide Residues Beijing, China. 20-25 April 2009 Agenda Items 4a, 4b, 5, 6 and 10 Agenda Item 4a JMPR Report 2008, Point
European Community Positions for the 41 st Session of the Codex Committee on Pesticide Residues Beijing, China. 20-25 April 2009 Agenda Items 4a, 4b, 5, 6 and 10 Agenda Item 4a JMPR Report 2008, Point
5.8 DIMETHOMORPH (225)
 Dimethomorph 107 5.8 DIMETHOMORPH (225) RESIDUE AND ANALYTICAL ASPECTS Dimethomorph is a fungicide with protective action against plant pathogenic Phytophthora species and a number of downy mildew diseases
Dimethomorph 107 5.8 DIMETHOMORPH (225) RESIDUE AND ANALYTICAL ASPECTS Dimethomorph is a fungicide with protective action against plant pathogenic Phytophthora species and a number of downy mildew diseases
A FULL SPECTRUM OF NUTRIENTS
 A FULL SPECTRUM OF NUTRIENTS All of us remember being told by our parents Eat your Vegetables. They re good for you. Fruits and vegetables are good for our Budgies too. Each color group has its own nutritional
A FULL SPECTRUM OF NUTRIENTS All of us remember being told by our parents Eat your Vegetables. They re good for you. Fruits and vegetables are good for our Budgies too. Each color group has its own nutritional
Official Journal of the European Union
 1.6.2016 L 144/27 COMMISSION REGULATION (EU) 2016/863 of 31 May 2016 amending Annexes VII and VIII to Regulation (EC) No 1907/2006 of the European Parliament and of the Council on the Registration, Evaluation,
1.6.2016 L 144/27 COMMISSION REGULATION (EU) 2016/863 of 31 May 2016 amending Annexes VII and VIII to Regulation (EC) No 1907/2006 of the European Parliament and of the Council on the Registration, Evaluation,
COMMISSION REGULATION (EU) / of XXX
 EUROPEAN COMMISSION Brussels, XXX SANTE/131/2015 CIS REV2 (POOL/G4/2015/131/131-EN CIS.doc) [ ](2015) XXX draft COMMISSION REGULATION (EU) / of XXX amending Annex III and Annex IV to Regulation (EC) No
EUROPEAN COMMISSION Brussels, XXX SANTE/131/2015 CIS REV2 (POOL/G4/2015/131/131-EN CIS.doc) [ ](2015) XXX draft COMMISSION REGULATION (EU) / of XXX amending Annex III and Annex IV to Regulation (EC) No
Council of the European Union Brussels, 12 November 2015 (OR. en)
 Council of the European Union Brussels, 12 November 2015 (OR. en) 14050/15 AGRILEG 222 DENLEG 147 VETER 102 COVER NOTE From: European Commission date of receipt: 12 November 2015 To: No. Cion doc.: D039258/04
Council of the European Union Brussels, 12 November 2015 (OR. en) 14050/15 AGRILEG 222 DENLEG 147 VETER 102 COVER NOTE From: European Commission date of receipt: 12 November 2015 To: No. Cion doc.: D039258/04
17257/13 PM/pm 1 DG B 4B
 COUNCIL OF THE EUROPEAN UNION Brussels, 3 December 2013 17257/13 DENLEG 145 SAN 503 AGRI 813 COVER NOTE from: European Commission date of receipt: 29 November 2013 to: General Secretariat of the Council
COUNCIL OF THE EUROPEAN UNION Brussels, 3 December 2013 17257/13 DENLEG 145 SAN 503 AGRI 813 COVER NOTE from: European Commission date of receipt: 29 November 2013 to: General Secretariat of the Council
