NIH Public Access Author Manuscript Science. Author manuscript; available in PMC 2011 September 1.
|
|
|
- Dennis West
- 5 years ago
- Views:
Transcription
1 NIH Public Access Author Manuscript Published in final edited form as: Science February 26; 327(5969): doi: /science Cortical Plasticity Induced by Inhibitory Neuron Transplantation Derek G. Southwell 1, Robert C. Froemke 2, Arturo Alvarez-Buylla 1,*, Michael P. Stryker 3,*, and Sunil P. Gandhi 3,* 1 Department of Neurological Surgery and the Eli and Edythe Broad Center of Regenerative Medicine and Stem Cell Research, University of California, San Francisco, 513 Parnassus Avenue, San Francisco, CA 94143, USA. 2 Department of Otolaryngology, University of California, San Francisco, 513 Parnassus Avenue, San Francisco, CA 94143, USA. 3 Department of Physiology, University of California, San Francisco, 513 Parnassus Avenue, San Francisco, CA 94143, USA. Abstract Critical periods are times of pronounced brain plasticity. During a critical period in the postnatal development of the visual cortex, the occlusion of one eye triggers a rapid reorganization of neuronal responses, a process known as ocular dominance plasticity. We have shown that the transplantation of inhibitory neurons induces ocular dominance plasticity after the critical period. Transplanted inhibitory neurons receive excitatory synapses, make inhibitory synapses onto host cortical neurons, and promote plasticity when they reach a cellular age equivalent to that of endogenous inhibitory neurons during the normal critical period. These findings suggest that ocular dominance plasticity is regulated by the execution of a maturational program intrinsic to inhibitory neurons. By inducing plasticity, inhibitory neuron transplantation may facilitate brain repair. Once in life, a critical period for ocular dominance plasticity is initiated by the development of intracortical inhibitory synaptic transmission (1). Reduction of inhibitory transmission disrupts ocular dominance plasticity (2), whereas the early enhancement of inhibitory transmission promotes a precocious period of ocular dominance plasticity (3 6). After the critical period has passed, however, direct pharmacological augmentation of inhibitory transmission does not induce plasticity (7). Cortical inhibitory neurons are produced in the medial and caudal ganglionic eminences of the embryonic ventral forebrain (8 10). When transplanted into the brains of older animals, embryonic inhibitory neuron precursors disperse widely (11) and develop the characteristics of mature cortical inhibitory neurons (12). We have used repeated optical imaging of intrinsic signals (13, 14) to examine whether inhibitory neuron transplantation produces ocular dominance plasticity after the critical period (fig. S1). In mice, ocular dominance plasticity reaches a peak in the fourth postnatal week, when cortical inhibitory neurons are ~33 to 35 days old (3, 10) (Fig. 1A). At this age, monocular visual deprivation shifts neuronal responses in the binocular visual cortex away from the Copyright 2010 by the American Association for the Advancement of Science; all rights reserved. * To whom correspondence should be addressed. abuylla@stemcell.ucsf.edu (A.A.B.); stryker@phy.ucsf.edu (M.P.S.); sunil@phy.ucsf.edu (S.P.G.). Supporting Online Material Materials and Methods Figs. S1 to S9 References
2 Southwell et al. Page 2 deprived eye and toward the nondeprived eye. Throughout this study, we have quantified the balance of cortical responses to the two eyes by calculating an ocular dominance index (ODI). An ODI value of 1 indicates responses dominated by the ipsilateral eye, a value of 1 represents responses dominated by the contralateral eye, and a value of 0 represents equal binocular responses. In untreated mice at the peak of the critical period [postnatal day 28 (P28)], the binocular visual cortex responded more to the contralateral eye, with a mean ODI of 0.22 (Fig. 1B, open black circles). After four days of visual deprivation of the contralateral eye, cortical responses were shifted toward the ipsilateral eye, with a mean ODI value of 0.00 (Fig. 1B, filled black circles). We first examined whether inhibitory neuron transplantation induced ocular dominance plasticity 14 to 18 days after the critical period, at P42 to 46. We transplanted cells from the embryonic day 13.5 to 14.5 (E13.5 to 14.5) medial ganglionic eminence (MGE) into sites flanking the host primary visual cortex at two ages, P0 to 2 and P9 to 11, respectively (Fig. 1A). Host mice that received transplants at P9 to 11 were thus studied 33 to 35 days after transplantation (DAT), whereas hosts that received transplants at P0 to 2 were studied 43 to 46 DAT. Transplantation did not alter the absolute magnitudes of visual responses in the host binocular visual cortex (fig. S2). Before monocular deprivation, host cortex responded more to the contralateral eye (Fig. 1B; P9 to 11 hosts, 33 to 35 DAT ODI mean ± SD = 0.23 ± 0.02, open green squares; P0 to 2 hosts, 43 to 46 DAT ODI = 0.24 ± 0.01, open red diamonds). Monocular deprivation shifted visual responses toward the nondeprived eye 33 to 35 DAT into P9 to 11 hosts (Fig. 1B, ODI = 0.05 ± 0.06, solid green squares). By contrast, monocular deprivation produced much weaker effects 43 to 46 DAT into P0 to 2 animals (Fig. 1B, ODI = 0.16 ± 0.04, solid red diamonds, Mann-Whitney test, U = 1, P = 0.005), consistent with earlier descriptions of the normal decline of ocular dominance plasticity after the critical period (15, 16). These findings demonstrate that transplantation into P9 to 11 animals induces ocular dominance plasticity 33 to 35 DAT, 14 to 18 days after the peak of the critical period. It remained unclear why transplantation into P0 to 2 hosts did not produce plasticity. We reasoned that the effects of transplantation may have been determined by the age of the host at transplantation (P0 to 2 versus P9 to 11) or by the cellular age of the transplanted population at the time of monocular deprivation (43 to 46 DAT versus 33 to 35 DAT). To distinguish between these possibilities, we transplanted into P0 to 2 and P9 to 11 hosts and then studied plasticity at P33 to 37, shortly after the critical period. Mice transplanted at P0 to 2 were thus studied 33 to 35 DAT, whereas animals transplanted at P9 to 11 were studied 25 to 27 DAT. Monocular deprivation elicited a strong effect 33 to 35 DAT into P0 to 2 hosts (Fig.1B; ODI = 0.04 ± 0.05, solid green diamonds) but produced a much weaker effect 25 to 27 DAT to P9 to 11 hosts (Fig. 1B; ODI = 0.13 ± 0.07, solid blue squares; U = 13, P = 0.035). Thus, transplantation into both P0 to 2 and P9 to 11 hosts could produce plasticity, depending on the age of the transplanted cells at the time of monocular deprivation: Transplantation was effective when the cells were 33 to 35 days old but not when the cells were 25 to 27 or 43 to 46 days old. These results showed that inhibitory neuron transplantation induced plasticity after the critical period, but they did not establish whether transplantation altered the normal plasticity at the peak of the critical period. We therefore studied plasticity 17 DAT into P11 animals (Fig. 1B, solid purple squares). At this age, monocular deprivation produced similar effects in hosts and age-matched, untreated animals (17 DAT ODI = 0.04 ± 0.01; untreated P28 control ODI = 0.00 ± 0.01; U = 1, P = 0.057). This finding, taken together with the results observed 25 to 27 and 33 to 35 DAT, indicates that transplantation produces a novel period of ocular dominance plasticity, rather than a delay or lengthening of normal critical period plasticity.
3 Southwell et al. Page 3 To determine whether the effects of transplantation were specific to the introduction of inhibitory precursors from the MGE, we transplanted cells from the lateral ganglionic eminence (LGE), an embryonic ventral forebrain region that produces olfactory bulb inhibitory neurons and striatal medium spiny neurons (17) (fig. S3). The transplantation of E13.5 LGE cells into P9 to 11 hosts did not produce a significant effect on ocular dominance plasticity 33 to 35 days later (Fig. 1B; before monocular deprivation, ODI = 0.24 ± 0.02, open green squares with red crosses; after monocular deprivation, ODI = 0.22 ± 0.03, solid green squares with red crosses; U = 4, P = 0.343). The transplantation of freeze-thawed, dead MGE cells also produced little plasticity (fig. S4; before monocular deprivation, ODI = 0.24 ± 0.01; after monocular deprivation, ODI = 0.19 ± 0.03), further suggesting that the effects of transplantation were specifically caused by live MGE cells (live cell 33 to 35 DAT versus dead cell ODI after monocular deprivation, U = 0, P = 0.006). Many of our results were obtained by measuring visual responses in the same host before and after monocular deprivation. For each animal, we calculated an ocular dominance shift, the difference between ODIs measured before and after monocular deprivation (Fig. 1C). The ocular dominance shift observed 33 to 35 DAT into P9 to 11 hosts (green) was 77% of that observed in untreated animals during the critical period (black). The effects of transplantation 33 to 35 DAT into P9 to 11 hosts were more than double those produced 25 to 27 DAT (blue) and 43 to 46 DAT (red). These findings indicate that transplantation has its strongest effect on plasticity when the transplanted cells reach a cellular age of 33 to 35 days. This age matches the age of endogenous inhibitory neurons at the peak of the normal critical period (10, 15). In most of our experiments, cells expressing a red fluorescent protein (DsRed) (18) were transplanted into hosts containing endogenous inhibitory neurons expressing green fluorescent protein (GAD67-GFP) (19). In other experiments, cells expressing GFP (20) were transplanted into wild-type hosts. The transplantation of DsRed cells into GAD67-GFP hosts was done to facilitate the identification of the transplanted and host inhibitory cells in electrophysiological recordings. Transplantation of GFP donor cells into wild-type hosts permitted the detailed histological characterization of the transplanted cells. The effects of monocular deprivation in wild-type host animals 33 to 35 DAT were similar to those observed in GAD67-GFP hosts (fig. S5; wild-type ODI = 0.04 ± 0.03; GAD67-GFP ODI = 0.05 ± 0.06). We next examined whether transplanted MGE cells migrated into the primary visual cortex and developed into inhibitory neurons. After the optical imaging experiments, we labeled the borders of host binocular visual cortex with DiI injections and studied the morphologies, molecular pheno-types, spatial distributions, and densities of transplanted cells within the DiI-labeled binocular visual cortex. Across all experimental groups, transplanted cells had migrated into all layers of the host visual cortex (Fig. 2A). Essentially all transplanted cells studied (17 DAT to 43 to 46 DAT) developed morphologies of mature inhibitory neurons (99.8%, Fig. 2B), whereas a very small fraction developed morphologies of glia (0.2%). In P9 to 11 hosts studied 33 to 35 DAT, nearly half of the transplanted neurons (49.5 ± 2.0%) expressed somatostatin, whereas over one-quarter expressed parvalbumin (27.7 ± 1.4%) (Fig. 2, C and D) (21, 22). Smaller fractions of the transplanted neurons expressed calretinin (17.5 ± 1.6%), calbindin (22.5 ± 1.2%), and neuropeptide Y(10.3 ± 1.7%). Awide range of transplanted cell densities were sufficient to induce plasticity 33 to 35 DAT (fig. S6). Variations in the total transplanted cell densities and spatial distributions (fig. S7) could not account for why transplant-induced plasticity was greatest 33 to 35 DAT. Because of evidence indicating that parvalbumin-expressing inhibitory neurons may regulate critical period ocular dominance plasticity (1, 5, 6), we examined whether the groups studied
4 Southwell et al. Page 4 at 17, 25 to 27, and 43 to 46 DAT had also received transplanted populations that included parvalbumin-expressing cells (fig. S8). At all ages studied, parvalbumin-expressing cells made up a fraction of the transplanted populations and the fraction of parvalbuminexpressing cells increased with the cellular age of the transplanted population. Thus, the lack of plasticity at 25 to 27 DAT and 43 to 46 DAT cannot be attributed to an absence of parvalbumin-expressing transplanted cells in these groups. Instead, the cellular age of the transplanted population determines the effects on cortical plasticity. We next examined whether transplanted neurons provided new inhibition to host visual cortex by making whole-cell current-clamp recordings from transplanted and host neurons (12, 23). Transplantation was performed into P0 to 2 or P9 to 11 hosts, and recordings were made from host brain slices at 36 ± 3 DAT (mean ± SD; host ages P43 ± 4), when transplant-induced plasticity was strongest. Transplanted inhibitory neurons (I trans ) received excitatory synapses and made inhibitory synapses with host excitatory neurons (E host ) (Fig. 3A). We compared the connection probabilities and synaptic strengths of transplanted neurons with those of adjacent host inhibitory neurons in the same slices (I host ; P45 ± 6; 38 ± 4 DAT) (Fig. 3B). Compared with host inhibitory neurons, transplanted inhibitory neurons were three times as likely to receive synapses from host excitatory neurons, and were also three times as likely to make inhibitory synapses onto host excitatory neurons (Fig. 3C; connection probability E host -to-i trans = 10/35 or 28.6%; E host -to-i host = 4/47 or 8.5%, P < 0.04; I trans -to-e host = 7/24 or 29.2%; I host -to-e host = 4/43 or 9.3%, P < 0.05). The synapses received and made by transplanted inhibitory neurons were about one-third as strong as host-to-host synapses [Fig. 3D; synaptic strength, E host -to-i trans EPSPs (excitatory postsynaptic potentials) = 0.34 ± 0.07 mv; E host -to-i host EPSPs = 1.1 ± 0.24 mv, P < 0.05; I trans -to-e host IPSPs (inhibitory postsynaptic potentials) = 0.24 ± 0.06 mv; I host -to-e host IPSPs = 0.56 ± 0.09, P < 0.03]. These results demonstrate that transplanted inhibitory neurons form weak but numerous synaptic connections with neighboring excitatory neurons, widely modifying inhibitory signaling in host cortex. The presence of numerous connections between transplanted inhibitory neurons and the host brain may explain why lower transplanted cell densities (fig. S6) were sufficient to induce plasticity. Our results show that inhibitory neuron transplantation induces ocular dominance plasticity after the normal critical period. Thirty-three to thirty-five days after transplantation, monocular deprivation produced a shift in visual responses comparable to that observed during the normal critical period (Fig. 1C). However, at shorter and longer intervals after transplantation (25 to 27 and 43 to 46 DAT, respectively), the effect of monocular deprivation was much smaller (Fig. 1C). Thus, transplant-induced plasticity resembles critical period plasticity in two ways: First, transplant-induced and critical period plasticity both peak when the transplanted and endogenous inhibitory neurons reach cellular ages of 33 to 35 days, respectively. Second, both transplant-induced and critical period plasticity are of short duration. Within the binocular visual cortex, the vast majority of transplanted cells expressed the morphological and molecular characteristics of mature inhibitory neurons. Transplanted inhibitory neurons received excitatory synapses from host neurons and made inhibitory synapses onto host neurons. These synaptic contacts were individually weaker but more numerous than those made by host inhibitory neurons. At a host age when transplanted inhibitory neurons induce plasticity (P42 to 46), the direct pharmacological enhancement of inhibition does not (7). How then, might inhibitory neuron transplantation produce a new period of cortical plasticity? One plausible explanation is that transplanted inhibitory neurons reorganize the cortical circuitry by introducing a new set of weak inhibitory synapses (6), rather than simply augmenting the strength of the endogenous, mature inhibitory connections. This pattern of numerous, weak connections is consistent
5 Southwell et al. Page 5 with the form of developing inhibition predicted to enhance Hebbian plasticity mechanisms during the critical period (24). Cortical plasticity can be produced after the critical period by the activation of neuromodulatory systems (25, 26) and the manipulation of molecules that promote structural plasticity (27). Each of these manipulations was associated with an accompanying reorganization of inhibitory circuits. Perhaps these experimental manipulations stimulate endogenous inhibitory neurons to form numerous, weak inhibitory synapses similar to those produced by transplanted inhibitory neurons. It is remarkable that transplanted inhibitory neurons induce plasticity when they reach a cellular age similar to that of endogenous inhibitory neurons during the critical period. This finding suggests (i) that the critical period is regulated by the execution of a developmental program intrinsic to inhibitory neurons and (ii) that embryonic inhibitory neuron precursors retain and execute this program when transplanted into the postnatal cortex. Inhibitory neuron transplantation provides a new experimental preparation for the study of cortical plasticity. Moreover, by promoting cortical plasticity, inhibitory neuron transplantation may facilitate the restoration of normal function to the diseased brain. Supplementary Material Acknowledgments Refer to Web version on PubMed Central for supplementary material. 28. We thank A. Kriegstein, P. Parker, and members of the Kriegstein laboratory for help with the electrophysiological recordings, R. Romero for assistance with histology, and P. McQuillen for sharing histology equipment. This work was funded by grants R01 NS (A.A.-B.) and P50 MH (M.P.S.) from the National Institutes of Health, grant P from the Dana Foundation (M.P.S.), and a grant from the Adelson Medical Research Foundation (M.P.S.). D.G.S. was supported by a predoctoral training grant from the California Institute for Regenerative Medicine. S.P.G. was supported by postdoctoral fellowship EY from the National Institutes of Health. R.C.F. is a recipient of a NIDCD K99/R00 Career Award. References and Notes 1. Hensch TK. Annu. Rev. Neurosci. 2004; 27:549. [PubMed: ] 2. Hensch TK, et al. Science. 1998; 282:1504. [PubMed: ] 3. Huang ZJ, et al. Cell. 1999; 98:739. [PubMed: ] 4. Hanover JL, Huang ZJ, Tonegawa S, Stryker MP. J. Neurosci. 1999; 19:RC40. [PubMed: ] 5. Fagiolini M, et al. Science. 2004; 303:1681. [PubMed: ] 6. Sugiyama S, et al. Cell. 2008; 134:508. [PubMed: ] 7. Fagiolini M, Hensch TK. Nature. 2000; 404:183. [PubMed: ] 8. Anderson SA, Eisenstat DD, Shi L, Rubenstein JL. Science. 1997; 278:474. [PubMed: ] 9. Nery S, Fishell G, Corbin JG. Nat. Neurosci. 2002; 5:1279. [PubMed: ] 10. Wonders CP, Anderson SA. Nat. Rev. Neurosci. 2006; 7:687. [PubMed: ] 11. Wichterle H, Garcia-Verdugo JM, Herrera DG, Alvarez-Buylla A. Nat. Neurosci. 1999; 2:461. [PubMed: ] 12. Alvarez-Dolado M, et al. J. Neurosci. 2006; 26:7380. [PubMed: ] 13. Cang J, Kalatsky VA, Löwel S, Stryker MP. Vis. Neurosci. 2005; 22:685. [PubMed: ] 14. Kaneko M, Hanover JL, England PM, Stryker MP. Nat. Neurosci. 2008; 11:497. [PubMed: ] 15. Gordon JA, Stryker MP. J. Neurosci. 1996; 16:3274. [PubMed: ] 16. Taha S, Hanover JL, Silva AJ, Stryker MP. Neuron. 2002; 36:483. [PubMed: ]
6 Southwell et al. Page Wichterle H, Turnbull DH, Nery S, Fishell G, Alvarez-Buylla A. Development. 2001; 128:3759. [PubMed: ] 18. Vintersten K, et al. Genesis. 2004; 40:241. [PubMed: ] 19. Tamamaki N, et al. J. Comp. Neurol. 2003; 467:60. [PubMed: ] 20. Hadjantonakis AK, Gertsenstein M, Ikawa M, Okabe M, Nagy A. Mech. Dev. 1998; 76:79. [PubMed: ] 21. Kawaguchi Y, Kubota Y. Cereb. Cortex. 1997; 7:476. [PubMed: ] 22. Gonchar Y, Wang Q, Burkhalter A. Front. Neuroanat. 2008; 1: Maffei A, Nataraj K, Nelson SB, Turrigiano GG. Nature. 2006; 443:81. [PubMed: ] 24. Gandhi SP, Yanagawa Y, Stryker MP. Proc. Natl. Acad. Sci. U.S.A. 2008; 105: [PubMed: ] 25. Froemke RC, Merzenich MM, Schreiner CE. Nature. 2007; 450:425. [PubMed: ] 26. Vetencourt, J. F. Maya, et al. Science. 2008; 320:385. [PubMed: ] 27. Pizzorusso T, et al. Science. 2002; 298:1248. [PubMed: ]
7 Southwell et al. Page 7 Fig. 1. Ocular dominance plasticity induced by transplantation of inhibitory neuron precursors. (A) Experimental design. Cortical inhibitory neurons are produced from E12 to 16. Mouse ocular dominance plasticity peaks at P26 to 28, when inhibitory neurons are 33 to 35 days old. Inhibitory neuron precursors were transplanted from E13.5 to 14.5 donor embryos into host animals of two ages: P0 to 2 (diamonds) and P9 to 11 (squares). Ocular dominance plasticity was assessed 17 (purple), 25 to 27 (blue), 33 to 35 (green), and 43 to 46 (red) DAT by measuring visual responses to the two eyes before (open symbols) and after (solid symbols) 4 days of monocular deprivation (MD). Visual responses were quantified using an ODI. (B) Results of plasticity studies. During the critical period (P28) in untreated mice, monocular deprivation shifted responses toward the nondeprived eye (open versus filled black circles). In P42 to 46 host animals, 14 to 18 days after the critical period, monocular deprivation produced a strong shift in responses 33 to 35 DAT (solid green squares). However, 43 to 46 DAT, monocular deprivation produced a weaker shift in responses (solid red diamonds). In P33 to 37 host animals, 5 to 9 days after the critical period, monocular deprivation produced a stronger effect 33 to 35 DAT (solid green diamonds) than at 25 to 27 DAT (solid blue squares). Transplantation did not alter the effects of monocular deprivation during the critical period (solid purple squares), as compared with untreated controls (solid black circles; Mann-Whitney test, U = 1, P = 0.057). Monocular deprivation produced an insignificant effect at 33 to 35 DAT using cells from the LGE (U = 4, P = 0.343, solid green squares with red crosses). (C) An ocular dominance shift quantified the change in ODI produced by monocular deprivation. Thirty-three to 35 DAT (green) the shift was 2.5 times as high as at 25 to 27 DAT (blue; P < 0.05) and 2.2 times as high as at 43 to 46 DAT (red; P < 0.05, Kruskal-Wallis test; H = 8.6 with Dunn s post test). The shift observed 33 to 35 DAT (green) was 77% of that observed in untreated animals during the critical period (black). Error bars represent SEM.
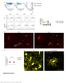
8 Southwell et al. Page 8 Fig. 2. Transplanted MGE cells migrate into binocular visual cortex and develop properties of mature inhibitory neurons. (A) Host cortex labeled for transplanted DsRed (red) and host GAD67-GFP (green) inhibitory neurons. DiI injections (red; dotted yellow line for emphasis) labeled the boundaries of binocular visual cortex, as identified by intrinsic signal imaging. Images are oriented with dorsal at top and lateral at left. (B) Detailed view of transplanted DsRed cell morphology. (C) To characterize the expression of inhibitory neuron molecular markers in the transplanted population, E13.5 to 14.5 MGE cells expressing GFP under the beta-actin promoter were transplanted into P9 to 11 wild-type hosts. Transplanted cells were immunostained for GFP (green), calbindin (CB), calretinin (CR), neuropeptide Y (NPY), parvalbumin (PV), or somatostatin (SOM). White chevrons identify colabeled cells. (D) Quantification of marker expression (N = 6 animals). Error bars represent SEM. Scale bars: 250 μm (A), 50 μm [(B) and (C)].
9 Southwell et al. Page 9 Fig. 3. Transplanted inhibitory neurons make and receive numerous, weak synaptic connections. Whole-cell recordings made from fluorescent transplanted inhibitory cells (I trans, red), host inhibitory cells (I host, green), and nonfluorescent putative pre- and postsynaptic pyramidal cell partners (E host ). Loose-patch stimulation was sometimes used to trigger presynaptic cells. (A) Whole-cell recording from a transplanted inhibitory cell. Loose-patch stimulation of a presynaptic pyramidal neuron evoked EPSPs in the transplanted cell. Transient depolarization of the transplanted neuron evoked IPSPs in a different postsynaptic pyramidal cell. Scale bars: 25 ms, 90 mv (presynaptic); 25 ms, mv (postsynaptic). (B) Recordings from pairs of host pyramidal and host inhibitory cells. (Top) Dual whole-cell recording of a presynaptic pyramidal cell and a postsynaptic I host. (Bottom) Loose-patch stimulation of a presynaptic host inhibitory neuron and whole-cell recording of a postsynaptic pyramidal cell. Scale bars: 25 ms, 40 mv (presynaptic); 25 ms, 0.25 mv (postsynaptic). (C) Connection probabilities for host-transplanted (red) and host-host (green) cell pairs. Transplanted inhibitory neurons made and received about three times as many connections as host inhibitory neurons (P < 0.05; two-tailed Fisher s exact test). (D) Connection strengths for all connected host-transplant and host-host cell pairs. Inhibitory and excitatory synapses made and received by transplanted cells were about one-third as strong as synapses of host inhibitory cells (P < 0.05).
Structural basis for the role of inhibition in facilitating adult brain plasticity
 Structural basis for the role of inhibition in facilitating adult brain plasticity Jerry L. Chen, Walter C. Lin, Jae Won Cha, Peter T. So, Yoshiyuki Kubota & Elly Nedivi SUPPLEMENTARY FIGURES 1-6 a b M
Structural basis for the role of inhibition in facilitating adult brain plasticity Jerry L. Chen, Walter C. Lin, Jae Won Cha, Peter T. So, Yoshiyuki Kubota & Elly Nedivi SUPPLEMENTARY FIGURES 1-6 a b M
Embryonic MGE Cells as a Treatment for Epilepsy December 1, 2012
 Embryonic MGE Cells as a Treatment for Epilepsy December 1, 2012 Scott C. Baraban, PhD University of California, San Francisco American Epilepsy Society Annual Meeting Disclosure Name of Commercial Interest
Embryonic MGE Cells as a Treatment for Epilepsy December 1, 2012 Scott C. Baraban, PhD University of California, San Francisco American Epilepsy Society Annual Meeting Disclosure Name of Commercial Interest
Precocious deposition of perineuronal nets on Parvalbumin inhibitory neurons transplanted into adult visual cortex
 www.nature.com/scientificreports Received: 23 June 2017 Accepted: 23 April 2018 Published: xx xx xxxx OPEN Precocious deposition of perineuronal nets on Parvalbumin inhibitory neurons transplanted into
www.nature.com/scientificreports Received: 23 June 2017 Accepted: 23 April 2018 Published: xx xx xxxx OPEN Precocious deposition of perineuronal nets on Parvalbumin inhibitory neurons transplanted into
Supplementary Figure 1. Microglia do not show signs of classical immune activation following MD a-b. Images showing immunoreactivity for MHCII (a)
 1 Supplementary Figure 1. Microglia do not show signs of classical immune activation following MD a-b. Images showing immunoreactivity for MHCII (a) and CD45 (b) in fixed sections of binocular visual cortex
1 Supplementary Figure 1. Microglia do not show signs of classical immune activation following MD a-b. Images showing immunoreactivity for MHCII (a) and CD45 (b) in fixed sections of binocular visual cortex
File name: Supplementary Information Description: Supplementary Figures, Supplementary Table and Supplementary References
 File name: Supplementary Information Description: Supplementary Figures, Supplementary Table and Supplementary References File name: Supplementary Data 1 Description: Summary datasheets showing the spatial
File name: Supplementary Information Description: Supplementary Figures, Supplementary Table and Supplementary References File name: Supplementary Data 1 Description: Summary datasheets showing the spatial
Optical imaging of the intrinsic signal as a measure of cortical plasticity in the mouse
 Visual Neuroscience ~2005!, 22, 685 691. Printed in the USA. Copyright 2005 Cambridge University Press 0952-5238005 $16.00 DOI: 10.10170S0952523805225178 Optical imaging of the intrinsic signal as a measure
Visual Neuroscience ~2005!, 22, 685 691. Printed in the USA. Copyright 2005 Cambridge University Press 0952-5238005 $16.00 DOI: 10.10170S0952523805225178 Optical imaging of the intrinsic signal as a measure
mir-132, an experience-dependent microrna, is essential for visual cortex plasticity
 mir-132, an experience-dependent microrna, is essential for visual cortex plasticity The MIT Faculty has made this article openly available. Please share how this access benefits you. Your story matters.
mir-132, an experience-dependent microrna, is essential for visual cortex plasticity The MIT Faculty has made this article openly available. Please share how this access benefits you. Your story matters.
Unique functional properties of somatostatin-expressing GABAergic neurons in mouse barrel cortex
 Supplementary Information Unique functional properties of somatostatin-expressing GABAergic neurons in mouse barrel cortex Luc Gentet, Yves Kremer, Hiroki Taniguchi, Josh Huang, Jochen Staiger and Carl
Supplementary Information Unique functional properties of somatostatin-expressing GABAergic neurons in mouse barrel cortex Luc Gentet, Yves Kremer, Hiroki Taniguchi, Josh Huang, Jochen Staiger and Carl
Astrocyte signaling controls spike timing-dependent depression at neocortical synapses
 Supplementary Information Astrocyte signaling controls spike timing-dependent depression at neocortical synapses Rogier Min and Thomas Nevian Department of Physiology, University of Berne, Bern, Switzerland
Supplementary Information Astrocyte signaling controls spike timing-dependent depression at neocortical synapses Rogier Min and Thomas Nevian Department of Physiology, University of Berne, Bern, Switzerland
Nature Neuroscience: doi: /nn Supplementary Figure 1. ACx plasticity is required for fear conditioning.
 Supplementary Figure 1 ACx plasticity is required for fear conditioning. (a) Freezing time of conditioned and control mice before CS presentation and during CS presentation in a new context. Student s
Supplementary Figure 1 ACx plasticity is required for fear conditioning. (a) Freezing time of conditioned and control mice before CS presentation and during CS presentation in a new context. Student s
Dep. Control Time (min)
 aa Control Dep. RP 1s 1 mv 2s 1 mv b % potentiation of IPSP 2 15 1 5 Dep. * 1 2 3 4 Time (min) Supplementary Figure 1. Rebound potentiation of IPSPs in PCs. a, IPSPs recorded with a K + gluconate pipette
aa Control Dep. RP 1s 1 mv 2s 1 mv b % potentiation of IPSP 2 15 1 5 Dep. * 1 2 3 4 Time (min) Supplementary Figure 1. Rebound potentiation of IPSPs in PCs. a, IPSPs recorded with a K + gluconate pipette
Input-speci"c adaptation in complex cells through synaptic depression
 0 0 0 0 Neurocomputing }0 (00) } Input-speci"c adaptation in complex cells through synaptic depression Frances S. Chance*, L.F. Abbott Volen Center for Complex Systems and Department of Biology, Brandeis
0 0 0 0 Neurocomputing }0 (00) } Input-speci"c adaptation in complex cells through synaptic depression Frances S. Chance*, L.F. Abbott Volen Center for Complex Systems and Department of Biology, Brandeis
ErbB4 migrazione II parte
 ErbB4 migrazione II parte Control SVZ cells prefer to migrate on the NRG1 type III substrate the substrate preference of the neuroblasts migrating out of the SVZ explant was evaluated SVZ cells had a strong
ErbB4 migrazione II parte Control SVZ cells prefer to migrate on the NRG1 type III substrate the substrate preference of the neuroblasts migrating out of the SVZ explant was evaluated SVZ cells had a strong
Ube3a is required for experience-dependent maturation of the neocortex
 Ube3a is required for experience-dependent maturation of the neocortex Koji Yashiro, Thorfinn T. Riday, Kathryn H. Condon, Adam C. Roberts, Danilo R. Bernardo, Rohit Prakash, Richard J. Weinberg, Michael
Ube3a is required for experience-dependent maturation of the neocortex Koji Yashiro, Thorfinn T. Riday, Kathryn H. Condon, Adam C. Roberts, Danilo R. Bernardo, Rohit Prakash, Richard J. Weinberg, Michael
Supplementary figure 1: LII/III GIN-cells show morphological characteristics of MC
 1 2 1 3 Supplementary figure 1: LII/III GIN-cells show morphological characteristics of MC 4 5 6 7 (a) Reconstructions of LII/III GIN-cells with somato-dendritic compartments in orange and axonal arborizations
1 2 1 3 Supplementary figure 1: LII/III GIN-cells show morphological characteristics of MC 4 5 6 7 (a) Reconstructions of LII/III GIN-cells with somato-dendritic compartments in orange and axonal arborizations
Recipes for Making Human Interneurons from Stem Cells Require Multiple Factors, Careful Timing, and Long Maturation Periods
 Current Literature In Basic Science Recipes for Making Human Interneurons from Stem Cells Require Multiple Factors, Careful Timing, and Long Maturation Periods Directed Differentiation and Functional Maturation
Current Literature In Basic Science Recipes for Making Human Interneurons from Stem Cells Require Multiple Factors, Careful Timing, and Long Maturation Periods Directed Differentiation and Functional Maturation
Mechanisms of plasticity in the developing visual cortex and how behavioral state changes cortical gain and adult plasticity
 Mechanisms of plasticity in the developing visual cortex and how behavioral state changes cortical gain and adult plasticity Michael P. Stryker Center for Integrative Neuroscience University of California,
Mechanisms of plasticity in the developing visual cortex and how behavioral state changes cortical gain and adult plasticity Michael P. Stryker Center for Integrative Neuroscience University of California,
Plasticity of Cerebral Cortex in Development
 Plasticity of Cerebral Cortex in Development Jessica R. Newton and Mriganka Sur Department of Brain & Cognitive Sciences Picower Center for Learning & Memory Massachusetts Institute of Technology Cambridge,
Plasticity of Cerebral Cortex in Development Jessica R. Newton and Mriganka Sur Department of Brain & Cognitive Sciences Picower Center for Learning & Memory Massachusetts Institute of Technology Cambridge,
During Brain Development Final Destinations for Neurons and Glia Get Separated from Germinal Niches
 During Brain Development Final Destinations for Neurons and Glia Get Separated from Germinal Niches Progenitors are Contained within Unique Domains and Tangentially Fixed. EMBRYO ADULT Migratory Behavior
During Brain Development Final Destinations for Neurons and Glia Get Separated from Germinal Niches Progenitors are Contained within Unique Domains and Tangentially Fixed. EMBRYO ADULT Migratory Behavior
Cellular Therapies December 9, 2013
 Cellular Therapies December 9, 2013 Scott C. Baraban, Ph.D. University of California, San Francisco American Epilepsy Society Annual Meeting Disclosure Neurona Therapeutics Co-founder BioCrea - Consultant
Cellular Therapies December 9, 2013 Scott C. Baraban, Ph.D. University of California, San Francisco American Epilepsy Society Annual Meeting Disclosure Neurona Therapeutics Co-founder BioCrea - Consultant
Brain-derived Neurotrophic Factor Overexpression Induces Precocious Critical Period in Mouse Visual Cortex
 The Journal of Neuroscience, 1999, Vol. 19 RC40 1of5 Brain-derived Neurotrophic Factor Overexpression Induces Precocious Critical Period in Mouse Visual Cortex Jessica L. Hanover, 1 Z. Josh Huang, 2 Susumu
The Journal of Neuroscience, 1999, Vol. 19 RC40 1of5 Brain-derived Neurotrophic Factor Overexpression Induces Precocious Critical Period in Mouse Visual Cortex Jessica L. Hanover, 1 Z. Josh Huang, 2 Susumu
SUPPLEMENTARY INFORMATION
 Supplementary Figure 1. Normal AMPAR-mediated fepsp input-output curve in CA3-Psen cdko mice. Input-output curves, which are plotted initial slopes of the evoked fepsp as function of the amplitude of the
Supplementary Figure 1. Normal AMPAR-mediated fepsp input-output curve in CA3-Psen cdko mice. Input-output curves, which are plotted initial slopes of the evoked fepsp as function of the amplitude of the
Brain Plasticity. Takao K. Hensch. Center for Brain Science Dept Molecular Cellular Biology Center on the Developing Child Harvard University
 Brain Plasticity Takao K. Hensch FM Kirby Neurobiology Center Department of Neurology Children's Hospital Boston Harvard Medical School Center for Brain Science Dept Molecular Cellular Biology Center on
Brain Plasticity Takao K. Hensch FM Kirby Neurobiology Center Department of Neurology Children's Hospital Boston Harvard Medical School Center for Brain Science Dept Molecular Cellular Biology Center on
Supplementary Figure 1. GABA depolarizes the majority of immature neurons in the
 Supplementary Figure 1. GABA depolarizes the majority of immature neurons in the upper cortical layers at P3 4 in vivo. (a b) Cell-attached current-clamp recordings illustrate responses to puff-applied
Supplementary Figure 1. GABA depolarizes the majority of immature neurons in the upper cortical layers at P3 4 in vivo. (a b) Cell-attached current-clamp recordings illustrate responses to puff-applied
Nature Neuroscience: doi: /nn Supplementary Figure 1. Trial structure for go/no-go behavior
 Supplementary Figure 1 Trial structure for go/no-go behavior a, Overall timeline of experiments. Day 1: A1 mapping, injection of AAV1-SYN-GCAMP6s, cranial window and headpost implantation. Water restriction
Supplementary Figure 1 Trial structure for go/no-go behavior a, Overall timeline of experiments. Day 1: A1 mapping, injection of AAV1-SYN-GCAMP6s, cranial window and headpost implantation. Water restriction
Modeling Depolarization Induced Suppression of Inhibition in Pyramidal Neurons
 Modeling Depolarization Induced Suppression of Inhibition in Pyramidal Neurons Peter Osseward, Uri Magaram Department of Neuroscience University of California, San Diego La Jolla, CA 92092 possewar@ucsd.edu
Modeling Depolarization Induced Suppression of Inhibition in Pyramidal Neurons Peter Osseward, Uri Magaram Department of Neuroscience University of California, San Diego La Jolla, CA 92092 possewar@ucsd.edu
Zhu et al, page 1. Supplementary Figures
 Zhu et al, page 1 Supplementary Figures Supplementary Figure 1: Visual behavior and avoidance behavioral response in EPM trials. (a) Measures of visual behavior that performed the light avoidance behavior
Zhu et al, page 1 Supplementary Figures Supplementary Figure 1: Visual behavior and avoidance behavioral response in EPM trials. (a) Measures of visual behavior that performed the light avoidance behavior
Supplementary Figure 1: Kv7 currents in neonatal CA1 neurons measured with the classic M- current voltage-clamp protocol.
 Supplementary Figures 1-11 Supplementary Figure 1: Kv7 currents in neonatal CA1 neurons measured with the classic M- current voltage-clamp protocol. (a), Voltage-clamp recordings from CA1 pyramidal neurons
Supplementary Figures 1-11 Supplementary Figure 1: Kv7 currents in neonatal CA1 neurons measured with the classic M- current voltage-clamp protocol. (a), Voltage-clamp recordings from CA1 pyramidal neurons
VISUAL CORTICAL PLASTICITY
 VISUAL CORTICAL PLASTICITY OCULAR DOMINANCE AND OTHER VARIETIES REVIEW OF HIPPOCAMPAL LTP 1 when an axon of cell A is near enough to excite a cell B and repeatedly and consistently takes part in firing
VISUAL CORTICAL PLASTICITY OCULAR DOMINANCE AND OTHER VARIETIES REVIEW OF HIPPOCAMPAL LTP 1 when an axon of cell A is near enough to excite a cell B and repeatedly and consistently takes part in firing
Loss of Sensory Input Causes Rapid Structural Changes of Inhibitory Neurons in Adult Mouse Visual Cortex
 Article Loss of Sensory Input Causes Rapid Structural Changes of Inhibitory Neurons in Adult Mouse Visual Cortex Tara Keck, 1,3 Volker Scheuss, 1 R. Irene Jacobsen, 1,3 Corette J. Wierenga, 1 Ulf T. Eysel,
Article Loss of Sensory Input Causes Rapid Structural Changes of Inhibitory Neurons in Adult Mouse Visual Cortex Tara Keck, 1,3 Volker Scheuss, 1 R. Irene Jacobsen, 1,3 Corette J. Wierenga, 1 Ulf T. Eysel,
Nature Neuroscience: doi: /nn Supplementary Figure 1. MADM labeling of thalamic clones.
 Supplementary Figure 1 MADM labeling of thalamic clones. (a) Confocal images of an E12 Nestin-CreERT2;Ai9-tdTomato brain treated with TM at E10 and stained for BLBP (green), a radial glial progenitor-specific
Supplementary Figure 1 MADM labeling of thalamic clones. (a) Confocal images of an E12 Nestin-CreERT2;Ai9-tdTomato brain treated with TM at E10 and stained for BLBP (green), a radial glial progenitor-specific
Cortical Map Plasticity. Gerald Finnerty Dept Basic and Clinical Neuroscience
 Cortical Map Plasticity Gerald Finnerty Dept Basic and Clinical Neuroscience Learning Objectives Be able to: 1. Describe the characteristics of a cortical map 2. Appreciate that the term plasticity is
Cortical Map Plasticity Gerald Finnerty Dept Basic and Clinical Neuroscience Learning Objectives Be able to: 1. Describe the characteristics of a cortical map 2. Appreciate that the term plasticity is
The molecule calcium calmodulin kinase II ( CaMKII)
 Ocular dominance plasticity is stably maintained in the absence of calcium calmodulin kinase II ( CaMKII) autophosphorylation Sharif A. Taha and Michael P. Stryker* Department of Physiology and W. M. Keck
Ocular dominance plasticity is stably maintained in the absence of calcium calmodulin kinase II ( CaMKII) autophosphorylation Sharif A. Taha and Michael P. Stryker* Department of Physiology and W. M. Keck
Activity-Dependent Development II April 25, 2007 Mu-ming Poo
 Activity-Dependent Development II April 25, 2007 Mu-ming Poo 1. The neurotrophin hypothesis 2. Maps in somatic sensory and motor cortices 3. Development of retinotopic map 4. Reorganization of cortical
Activity-Dependent Development II April 25, 2007 Mu-ming Poo 1. The neurotrophin hypothesis 2. Maps in somatic sensory and motor cortices 3. Development of retinotopic map 4. Reorganization of cortical
Benzodiazepine actions on GABAergic inhibitory postsynaptic currents (IPSCs) were
 Supplementary Online Material Materials and Methods Benzodiazepine actions on GABAergic inhibitory postsynaptic currents (IPSCs) were confirmed in slices of mouse visual cortex (C57Bl/6) prepared and maintained
Supplementary Online Material Materials and Methods Benzodiazepine actions on GABAergic inhibitory postsynaptic currents (IPSCs) were confirmed in slices of mouse visual cortex (C57Bl/6) prepared and maintained
Synaptic Plasticity and the NMDA Receptor
 Synaptic Plasticity and the NMDA Receptor Lecture 4.2 David S. Touretzky November, 2015 Long Term Synaptic Plasticity Long Term Potentiation (LTP) Reversal of LTP Long Term Depression (LTD) Reversal of
Synaptic Plasticity and the NMDA Receptor Lecture 4.2 David S. Touretzky November, 2015 Long Term Synaptic Plasticity Long Term Potentiation (LTP) Reversal of LTP Long Term Depression (LTD) Reversal of
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION doi:10.1038/nature11306 Supplementary Figures Supplementary Figure 1. Basic characterization of GFP+ RGLs in the dentate gyrus of adult nestin-gfp mice. a, Sample confocal images
SUPPLEMENTARY INFORMATION doi:10.1038/nature11306 Supplementary Figures Supplementary Figure 1. Basic characterization of GFP+ RGLs in the dentate gyrus of adult nestin-gfp mice. a, Sample confocal images
Nature Neuroscience: doi: /nn Supplementary Figure 1
 Supplementary Figure 1 Atlas representations of the midcingulate (MCC) region targeted in this study compared against the anterior cingulate (ACC) region commonly reported. Coronal sections are shown on
Supplementary Figure 1 Atlas representations of the midcingulate (MCC) region targeted in this study compared against the anterior cingulate (ACC) region commonly reported. Coronal sections are shown on
Tuning properties of individual circuit components and stimulus-specificity of experience-driven changes.
 Supplementary Figure 1 Tuning properties of individual circuit components and stimulus-specificity of experience-driven changes. (a) Left, circuit schematic with the imaged component (L2/3 excitatory neurons)
Supplementary Figure 1 Tuning properties of individual circuit components and stimulus-specificity of experience-driven changes. (a) Left, circuit schematic with the imaged component (L2/3 excitatory neurons)
Supplementary Figure 1. Basic properties of compound EPSPs at
 Supplementary Figure 1. Basic properties of compound EPSPs at hippocampal CA3 CA3 cell synapses. (a) EPSPs were evoked by extracellular stimulation of the recurrent collaterals and pharmacologically isolated
Supplementary Figure 1. Basic properties of compound EPSPs at hippocampal CA3 CA3 cell synapses. (a) EPSPs were evoked by extracellular stimulation of the recurrent collaterals and pharmacologically isolated
SUPPLEMENTARY INFORMATION
 doi:10.1038/nature10776 Supplementary Information 1: Influence of inhibition among blns on STDP of KC-bLN synapses (simulations and schematics). Unconstrained STDP drives network activity to saturation
doi:10.1038/nature10776 Supplementary Information 1: Influence of inhibition among blns on STDP of KC-bLN synapses (simulations and schematics). Unconstrained STDP drives network activity to saturation
Cell Migration II: CNS Cell Migration. Steven McLoon Department of Neuroscience University of Minnesota
 Cell Migration II: CNS Cell Migration Steven McLoon Department of Neuroscience University of Minnesota 1 Hey! The major concepts discussed relative to neural crest cell migration apply to cell migration
Cell Migration II: CNS Cell Migration Steven McLoon Department of Neuroscience University of Minnesota 1 Hey! The major concepts discussed relative to neural crest cell migration apply to cell migration
During Brain Development Final Destinations for Neurons and Glia Get Separated from Germinal Niches
 During Brain Development Final Destinations for Neurons and Glia Get Separated from Germinal Niches Two mayor forms of neuronal migration: Radial and Tangential Leber & Sanes, 95 How do young neurons actually
During Brain Development Final Destinations for Neurons and Glia Get Separated from Germinal Niches Two mayor forms of neuronal migration: Radial and Tangential Leber & Sanes, 95 How do young neurons actually
SUPPLEMENTARY INFORMATION
 doi: 1.138/nature6416 Supplementary Notes Spine Ca 2+ signals produced by glutamate uncaging We imaged uncaging-evoked [Ca 2+ ] transients in neurons loaded with a green Ca 2+ - sensitive indicator (G;
doi: 1.138/nature6416 Supplementary Notes Spine Ca 2+ signals produced by glutamate uncaging We imaged uncaging-evoked [Ca 2+ ] transients in neurons loaded with a green Ca 2+ - sensitive indicator (G;
mir-7a regulation of Pax6 in neural stem cells controls the spatial origin of forebrain dopaminergic neurons
 Supplemental Material mir-7a regulation of Pax6 in neural stem cells controls the spatial origin of forebrain dopaminergic neurons Antoine de Chevigny, Nathalie Coré, Philipp Follert, Marion Gaudin, Pascal
Supplemental Material mir-7a regulation of Pax6 in neural stem cells controls the spatial origin of forebrain dopaminergic neurons Antoine de Chevigny, Nathalie Coré, Philipp Follert, Marion Gaudin, Pascal
Early- and Late-Born Parvalbumin Basket Cell Subpopulations Exhibiting Distinct Regulation and Roles in Learning
 Article Early- and Late-Born Parvalbumin Basket Cell Subpopulations Exhibiting Distinct Regulation and Roles in Learning Highlights d PV Basket cells consist of subpopulations defined by their schedule
Article Early- and Late-Born Parvalbumin Basket Cell Subpopulations Exhibiting Distinct Regulation and Roles in Learning Highlights d PV Basket cells consist of subpopulations defined by their schedule
PAPER A Model for Ocular Dominance Plasticity Controlled by Feedforward and Feedback Inhibition
 1780 PAPER A Model for Ocular Dominance Plasticity Controlled by Feedforward and Feedback Inhibition Ichiro SAKURAI, Nonmember, Shigeru KUBOTA a), and Michio NIWANO, Members SUMMARY The maturation of inhibitory
1780 PAPER A Model for Ocular Dominance Plasticity Controlled by Feedforward and Feedback Inhibition Ichiro SAKURAI, Nonmember, Shigeru KUBOTA a), and Michio NIWANO, Members SUMMARY The maturation of inhibitory
How Nicotinic Signaling Shapes Neural Networks
 How Nicotinic Signaling Shapes Neural Networks Darwin K. Berg Division of Biological Sciences University of California, San Diego Nicotinic Cholinergic Signaling Uses the transmitter ACh to activate cation-selective
How Nicotinic Signaling Shapes Neural Networks Darwin K. Berg Division of Biological Sciences University of California, San Diego Nicotinic Cholinergic Signaling Uses the transmitter ACh to activate cation-selective
Nature Neuroscience: doi: /nn Supplementary Figure 1. Diverse anorexigenic signals induce c-fos expression in CEl PKC-δ + neurons
 Supplementary Figure 1 Diverse anorexigenic signals induce c-fos expression in CEl PKC-δ + neurons a-c. Quantification of CEl c-fos expression in mice intraperitoneal injected with anorexigenic drugs (a),
Supplementary Figure 1 Diverse anorexigenic signals induce c-fos expression in CEl PKC-δ + neurons a-c. Quantification of CEl c-fos expression in mice intraperitoneal injected with anorexigenic drugs (a),
Supplementary Figure 1. Nature Neuroscience: doi: /nn.4547
 Supplementary Figure 1 Characterization of the Microfetti mouse model. (a) Gating strategy for 8-color flow analysis of peripheral Ly-6C + monocytes from Microfetti mice 5-7 days after TAM treatment. Living
Supplementary Figure 1 Characterization of the Microfetti mouse model. (a) Gating strategy for 8-color flow analysis of peripheral Ly-6C + monocytes from Microfetti mice 5-7 days after TAM treatment. Living
Part 11: Mechanisms of Learning
 Neurophysiology and Information: Theory of Brain Function Christopher Fiorillo BiS 527, Spring 2012 042 350 4326, fiorillo@kaist.ac.kr Part 11: Mechanisms of Learning Reading: Bear, Connors, and Paradiso,
Neurophysiology and Information: Theory of Brain Function Christopher Fiorillo BiS 527, Spring 2012 042 350 4326, fiorillo@kaist.ac.kr Part 11: Mechanisms of Learning Reading: Bear, Connors, and Paradiso,
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION Human cerebral cortex development from pluripotent stem cells to functional excitatory synapses Yichen Shi 1,2, Peter Kirwan 1,2, James Smith 1,2, Hugh P.C. Robinson 3 and Frederick
SUPPLEMENTARY INFORMATION Human cerebral cortex development from pluripotent stem cells to functional excitatory synapses Yichen Shi 1,2, Peter Kirwan 1,2, James Smith 1,2, Hugh P.C. Robinson 3 and Frederick
Supplementary Materials for VAMP4 directs synaptic vesicles to a pool that selectively maintains asynchronous neurotransmission
 Supplementary Materials for VAMP4 directs synaptic vesicles to a pool that selectively maintains asynchronous neurotransmission Jesica Raingo, Mikhail Khvotchev, Pei Liu, Frederic Darios, Ying C. Li, Denise
Supplementary Materials for VAMP4 directs synaptic vesicles to a pool that selectively maintains asynchronous neurotransmission Jesica Raingo, Mikhail Khvotchev, Pei Liu, Frederic Darios, Ying C. Li, Denise
Supplementary Figure 1 Information on transgenic mouse models and their recording and optogenetic equipment. (a) 108 (b-c) (d) (e) (f) (g)
 Supplementary Figure 1 Information on transgenic mouse models and their recording and optogenetic equipment. (a) In four mice, cre-dependent expression of the hyperpolarizing opsin Arch in pyramidal cells
Supplementary Figure 1 Information on transgenic mouse models and their recording and optogenetic equipment. (a) In four mice, cre-dependent expression of the hyperpolarizing opsin Arch in pyramidal cells
NIH Public Access Author Manuscript Nat Neurosci. Author manuscript; available in PMC 2006 September 5.
 NIH Public Access Author Manuscript Published in final edited form as: Nat Neurosci. 2006 August ; 9(8): 1004 1006. Maternal presence serves as a switch between learning fear and attraction in infancy
NIH Public Access Author Manuscript Published in final edited form as: Nat Neurosci. 2006 August ; 9(8): 1004 1006. Maternal presence serves as a switch between learning fear and attraction in infancy
Dendritic BDNF Synthesis Is Required for Late-Phase Spine Maturation and Recovery of Cortical Responses Following Sensory Deprivation
 4790 The Journal of Neuroscience, April 4, 2012 32(14):4790 4802 Development/Plasticity/Repair Dendritic BDNF Synthesis Is Required for Late-Phase Spine Maturation and Recovery of Cortical Responses Following
4790 The Journal of Neuroscience, April 4, 2012 32(14):4790 4802 Development/Plasticity/Repair Dendritic BDNF Synthesis Is Required for Late-Phase Spine Maturation and Recovery of Cortical Responses Following
Dynamic Stochastic Synapses as Computational Units
 Dynamic Stochastic Synapses as Computational Units Wolfgang Maass Institute for Theoretical Computer Science Technische Universitat Graz A-B01O Graz Austria. email: maass@igi.tu-graz.ac.at Anthony M. Zador
Dynamic Stochastic Synapses as Computational Units Wolfgang Maass Institute for Theoretical Computer Science Technische Universitat Graz A-B01O Graz Austria. email: maass@igi.tu-graz.ac.at Anthony M. Zador
Spatial profiles of inhibition in piriform cortex. Anne-Marie Oswald Department of Neuroscience, University of Pittsburgh
 Spatial profiles of inhibition in piriform cortex Anne-Marie Oswald Department of Neuroscience, University of Pittsburgh amoswald@pitt.edu Spatial representation of odors Olfactory Bulb Odor Maps Olfactory
Spatial profiles of inhibition in piriform cortex Anne-Marie Oswald Department of Neuroscience, University of Pittsburgh amoswald@pitt.edu Spatial representation of odors Olfactory Bulb Odor Maps Olfactory
When cells are already maximally potentiated LTP is occluded.
 When cells are already maximally potentiated LTP is occluded. Stein, V et al., (2003) J Neurosci, 23:5503-6606. Also found in Rat Barrel Cortex Ehrlich & Malinow (2004) J. Neurosci. 24:916-927 Over-expression
When cells are already maximally potentiated LTP is occluded. Stein, V et al., (2003) J Neurosci, 23:5503-6606. Also found in Rat Barrel Cortex Ehrlich & Malinow (2004) J. Neurosci. 24:916-927 Over-expression
Nature Methods: doi: /nmeth Supplementary Figure 1. Activity in turtle dorsal cortex is sparse.
 Supplementary Figure 1 Activity in turtle dorsal cortex is sparse. a. Probability distribution of firing rates across the population (notice log scale) in our data. The range of firing rates is wide but
Supplementary Figure 1 Activity in turtle dorsal cortex is sparse. a. Probability distribution of firing rates across the population (notice log scale) in our data. The range of firing rates is wide but
Thalamo-Cortical Relationships Ultrastructure of Thalamic Synaptic Glomerulus
 Central Visual Pathways V1/2 NEUR 3001 dvanced Visual Neuroscience The Lateral Geniculate Nucleus () is more than a relay station LP SC Professor Tom Salt UCL Institute of Ophthalmology Retina t.salt@ucl.ac.uk
Central Visual Pathways V1/2 NEUR 3001 dvanced Visual Neuroscience The Lateral Geniculate Nucleus () is more than a relay station LP SC Professor Tom Salt UCL Institute of Ophthalmology Retina t.salt@ucl.ac.uk
Axon initial segment position changes CA1 pyramidal neuron excitability
 Axon initial segment position changes CA1 pyramidal neuron excitability Cristina Nigro and Jason Pipkin UCSD Neurosciences Graduate Program Abstract The axon initial segment (AIS) is the portion of the
Axon initial segment position changes CA1 pyramidal neuron excitability Cristina Nigro and Jason Pipkin UCSD Neurosciences Graduate Program Abstract The axon initial segment (AIS) is the portion of the
A Theory of the Transition to Critical Period Plasticity: Inhibition Selectively Suppresses Spontaneous Activity
 Viewpoint A Theory of the Transition to Critical Period Plasticity: Inhibition Selectively Suppresses Spontaneous Activity Taro Toyoizumi, 1,3,4, * Hiroyuki Miyamoto, 3 Yoko Yazaki-Sugiyama, 3 Nafiseh
Viewpoint A Theory of the Transition to Critical Period Plasticity: Inhibition Selectively Suppresses Spontaneous Activity Taro Toyoizumi, 1,3,4, * Hiroyuki Miyamoto, 3 Yoko Yazaki-Sugiyama, 3 Nafiseh
Supplementary Information
 Supplementary Information D-Serine regulates cerebellar LTD and motor coordination through the 2 glutamate receptor Wataru Kakegawa, Yurika Miyoshi, Kenji Hamase, Shinji Matsuda, Keiko Matsuda, Kazuhisa
Supplementary Information D-Serine regulates cerebellar LTD and motor coordination through the 2 glutamate receptor Wataru Kakegawa, Yurika Miyoshi, Kenji Hamase, Shinji Matsuda, Keiko Matsuda, Kazuhisa
Inhibitory and Excitatory Spike-Timing-Dependent Plasticity in the Auditory Cortex
 Article Inhibitory and Excitatory Spike-Timing-Dependent Plasticity in the Auditory Cortex Highlights d Spike pairing induces STDP of excitatory and inhibitory layer 5 cortical synapses d d d Inhibitory
Article Inhibitory and Excitatory Spike-Timing-Dependent Plasticity in the Auditory Cortex Highlights d Spike pairing induces STDP of excitatory and inhibitory layer 5 cortical synapses d d d Inhibitory
SUPPLEMENTARY INFORMATION
 doi:1.138/nature1139 a d Whisker angle (deg) Whisking repeatability Control Muscimol.4.3.2.1 -.1 8 4-4 1 2 3 4 Performance (d') Pole 8 4-4 1 2 3 4 5 Time (s) b Mean protraction angle (deg) e Hit rate (p
doi:1.138/nature1139 a d Whisker angle (deg) Whisking repeatability Control Muscimol.4.3.2.1 -.1 8 4-4 1 2 3 4 Performance (d') Pole 8 4-4 1 2 3 4 5 Time (s) b Mean protraction angle (deg) e Hit rate (p
An acetylcholine-activated microcircuit drives temporal dynamics of cortical activity
 An acetylcholine-activated microcircuit drives temporal dynamics of cortical activity Naiyan Chen, Hiroki Sugihara, & Mriganka Sur Nature America, nc. All rights reserved. Cholinergic modulation of cortex
An acetylcholine-activated microcircuit drives temporal dynamics of cortical activity Naiyan Chen, Hiroki Sugihara, & Mriganka Sur Nature America, nc. All rights reserved. Cholinergic modulation of cortex
Cell Migration II: CNS Cell Migration. Steven McLoon Department of Neuroscience University of Minnesota
 Cell Migration II: CNS Cell Migration Steven McLoon Department of Neuroscience University of Minnesota 1 Course News Coffee Hour Wednesday (Oct 18) 9:00-10:00am Surdyk s Café in Northrop Auditorium Stop
Cell Migration II: CNS Cell Migration Steven McLoon Department of Neuroscience University of Minnesota 1 Course News Coffee Hour Wednesday (Oct 18) 9:00-10:00am Surdyk s Café in Northrop Auditorium Stop
Beyond Vanilla LTP. Spike-timing-dependent-plasticity or STDP
 Beyond Vanilla LTP Spike-timing-dependent-plasticity or STDP Hebbian learning rule asn W MN,aSN MN Δw ij = μ x j (v i - φ) learning threshold under which LTD can occur Stimulation electrode Recording electrode
Beyond Vanilla LTP Spike-timing-dependent-plasticity or STDP Hebbian learning rule asn W MN,aSN MN Δw ij = μ x j (v i - φ) learning threshold under which LTD can occur Stimulation electrode Recording electrode
Neuroscience with Pharmacology 2 Functions and Mechanisms of Reflexes. Prof Richard Ribchester
 Neuroscience with Pharmacology 2 Functions and Mechanisms of Reflexes Prof Richard Ribchester René Descartes Cogito, ergo sum The 21st century still holds many challenges to Neuroscience and Pharmacology
Neuroscience with Pharmacology 2 Functions and Mechanisms of Reflexes Prof Richard Ribchester René Descartes Cogito, ergo sum The 21st century still holds many challenges to Neuroscience and Pharmacology
Disrupting GluA2-GAPDH Interaction Affects Axon and Dendrite Development
 Disrupting GluA2-GAPDH Interaction Affects Axon and Dendrite Development 1 Frankie Hang Fung Lee, 1 Ping Su, 1 Yu Feng Xie, 1 Kyle Ethan Wang, 2 Qi Wan and 1,3 Fang Liu 1 Campbell Research Institute, Centre
Disrupting GluA2-GAPDH Interaction Affects Axon and Dendrite Development 1 Frankie Hang Fung Lee, 1 Ping Su, 1 Yu Feng Xie, 1 Kyle Ethan Wang, 2 Qi Wan and 1,3 Fang Liu 1 Campbell Research Institute, Centre
Simulating inputs of parvalbumin inhibitory interneurons onto excitatory pyramidal cells in piriform cortex
 Simulating inputs of parvalbumin inhibitory interneurons onto excitatory pyramidal cells in piriform cortex Jeffrey E. Dahlen jdahlen@ucsd.edu and Kerin K. Higa khiga@ucsd.edu Department of Neuroscience
Simulating inputs of parvalbumin inhibitory interneurons onto excitatory pyramidal cells in piriform cortex Jeffrey E. Dahlen jdahlen@ucsd.edu and Kerin K. Higa khiga@ucsd.edu Department of Neuroscience
Supplementary Figure 1
 Supplementary Figure 1 Supplementary Figure 1 SNARE Probes for FRET/2pFLIM Analysis Used in the Present Study. mturquoise (mtq) and Venus (Ven) are in blue and yellow, respectively. The soluble N-ethylmaleimide-sensitive
Supplementary Figure 1 Supplementary Figure 1 SNARE Probes for FRET/2pFLIM Analysis Used in the Present Study. mturquoise (mtq) and Venus (Ven) are in blue and yellow, respectively. The soluble N-ethylmaleimide-sensitive
Systems Neurobiology: Plasticity in the Auditory System. Jason Middleton -
 Systems Neurobiology: Plasticity in the Auditory System Jason Middleton - jmiddlet@pitt.edu Auditory plasticity Plasticity Early development and critical period Adult plasticity and neuromodulation Brainstem
Systems Neurobiology: Plasticity in the Auditory System Jason Middleton - jmiddlet@pitt.edu Auditory plasticity Plasticity Early development and critical period Adult plasticity and neuromodulation Brainstem
OPTO 5320 VISION SCIENCE I
 OPTO 5320 VISION SCIENCE I Monocular Sensory Processes of Vision: Color Vision Mechanisms of Color Processing . Neural Mechanisms of Color Processing A. Parallel processing - M- & P- pathways B. Second
OPTO 5320 VISION SCIENCE I Monocular Sensory Processes of Vision: Color Vision Mechanisms of Color Processing . Neural Mechanisms of Color Processing A. Parallel processing - M- & P- pathways B. Second
Preferential Origin and Layer Destination of GAD65-GFP Cortical Interneurons
 Preferential Origin and Layer Destination of GAD65-GFP Cortical Interneurons Guillermina López-Bendito 1, Katherine Sturgess 1, Ferenc Erdélyi 2, Gábor Szabó 2, Zoltán Molnár 1 and Ole Paulsen 3 1 Department
Preferential Origin and Layer Destination of GAD65-GFP Cortical Interneurons Guillermina López-Bendito 1, Katherine Sturgess 1, Ferenc Erdélyi 2, Gábor Szabó 2, Zoltán Molnár 1 and Ole Paulsen 3 1 Department
THE JOURNAL OF COMPARATIVE NEUROLOGY 422: (2000)
 THE JOURNAL OF COMPARATIVE NEUROLOGY 422:652 664 (2000) Distributions of Synaptic Vesicle Proteins and GAD65 in Deprived and Nondeprived Ocular Dominance Columns in Layer IV of Kitten Primary Visual Cortex
THE JOURNAL OF COMPARATIVE NEUROLOGY 422:652 664 (2000) Distributions of Synaptic Vesicle Proteins and GAD65 in Deprived and Nondeprived Ocular Dominance Columns in Layer IV of Kitten Primary Visual Cortex
Suppl. Information Supplementary Figure 1. Strategy/latency analysis of individual mice during maze learning. a,
 Goal-oriented searching mediated by ventral hippocampus early in trial-and-error learning Ruediger, S, Spirig, D., Donato, F., Caroni, P. Suppl. Information Supplementary Figure 1. Strategy/latency analysis
Goal-oriented searching mediated by ventral hippocampus early in trial-and-error learning Ruediger, S, Spirig, D., Donato, F., Caroni, P. Suppl. Information Supplementary Figure 1. Strategy/latency analysis
SUPPLEMENTARY INFORMATION. Supplementary Figure 1
 SUPPLEMENTARY INFORMATION Supplementary Figure 1 The supralinear events evoked in CA3 pyramidal cells fulfill the criteria for NMDA spikes, exhibiting a threshold, sensitivity to NMDAR blockade, and all-or-none
SUPPLEMENTARY INFORMATION Supplementary Figure 1 The supralinear events evoked in CA3 pyramidal cells fulfill the criteria for NMDA spikes, exhibiting a threshold, sensitivity to NMDAR blockade, and all-or-none
Carl P. Wonders* and Stewart A. Anderson
 The origin and specification of cortical interneurons Carl P. Wonders* and Stewart A. Anderson Abstract GABA-containing interneurons are crucial to both the development and function of the cerebral cortex.
The origin and specification of cortical interneurons Carl P. Wonders* and Stewart A. Anderson Abstract GABA-containing interneurons are crucial to both the development and function of the cerebral cortex.
Research Article Visual Deprivation Decreases Somatic GAD65 Puncta Number on Layer 2/3 Pyramidal Neurons in Mouse Visual Cortex
 Neural Plasticity Volume 29, Article ID 415135, 7 pages doi:1.1155/29/415135 Research Article Visual Deprivation Decreases Somatic GAD65 Puncta Number on Layer 2/3 Pyramidal Neurons in Mouse Visual Cortex
Neural Plasticity Volume 29, Article ID 415135, 7 pages doi:1.1155/29/415135 Research Article Visual Deprivation Decreases Somatic GAD65 Puncta Number on Layer 2/3 Pyramidal Neurons in Mouse Visual Cortex
The neuron. Biocytin labeled pyramidal neuron recorded in piriform cortex
 The neuron Biocytin labeled pyramidal neuron recorded in piriform cortex Discovery of the neuron (A) Reticularist Doctrine (B) Neuron Doctrine Exception.GAP JUNCTIONS between neurons Neuronal shape Dendrites
The neuron Biocytin labeled pyramidal neuron recorded in piriform cortex Discovery of the neuron (A) Reticularist Doctrine (B) Neuron Doctrine Exception.GAP JUNCTIONS between neurons Neuronal shape Dendrites
Supplementary Table I Blood pressure and heart rate measurements pre- and post-stroke
 SUPPLEMENTARY INFORMATION doi:10.1038/nature09511 Supplementary Table I Blood pressure and heart rate measurements pre- and post-stroke Pre Post 7-days Systolic Diastolic BPM Systolic Diastolic BPM Systolic
SUPPLEMENTARY INFORMATION doi:10.1038/nature09511 Supplementary Table I Blood pressure and heart rate measurements pre- and post-stroke Pre Post 7-days Systolic Diastolic BPM Systolic Diastolic BPM Systolic
NEURONS COMMUNICATE WITH OTHER CELLS AT SYNAPSES 34.3
 NEURONS COMMUNICATE WITH OTHER CELLS AT SYNAPSES 34.3 NEURONS COMMUNICATE WITH OTHER CELLS AT SYNAPSES Neurons communicate with other neurons or target cells at synapses. Chemical synapse: a very narrow
NEURONS COMMUNICATE WITH OTHER CELLS AT SYNAPSES 34.3 NEURONS COMMUNICATE WITH OTHER CELLS AT SYNAPSES Neurons communicate with other neurons or target cells at synapses. Chemical synapse: a very narrow
LOCAL CIRCUIT NEURONS IN THE FRONTAL CORTICO-STRIATAL SYSTEM
 8 LOCAL CIRCUIT NEURONS IN THE FRONTAL CORTICO-STRIATAL SYSTEM Y. Kawaguchi 1. INTRODUCTION The neostriatum is the principal recipient of afferents to basal ganglia from the cerebral cortex, especially,
8 LOCAL CIRCUIT NEURONS IN THE FRONTAL CORTICO-STRIATAL SYSTEM Y. Kawaguchi 1. INTRODUCTION The neostriatum is the principal recipient of afferents to basal ganglia from the cerebral cortex, especially,
Lecture 22: A little Neurobiology
 BIO 5099: Molecular Biology for Computer Scientists (et al) Lecture 22: A little Neurobiology http://compbio.uchsc.edu/hunter/bio5099 Larry.Hunter@uchsc.edu Nervous system development Part of the ectoderm
BIO 5099: Molecular Biology for Computer Scientists (et al) Lecture 22: A little Neurobiology http://compbio.uchsc.edu/hunter/bio5099 Larry.Hunter@uchsc.edu Nervous system development Part of the ectoderm
Monocular and Binocular Mechanisms of Contrast Gain Control. Izumi Ohzawa and Ralph D. Freeman
 Monocular and Binocular Mechanisms of Contrast Gain Control Izumi Ohzawa and alph D. Freeman University of California, School of Optometry Berkeley, California 9472 E-mail: izumi@pinoko.berkeley.edu ABSTACT
Monocular and Binocular Mechanisms of Contrast Gain Control Izumi Ohzawa and alph D. Freeman University of California, School of Optometry Berkeley, California 9472 E-mail: izumi@pinoko.berkeley.edu ABSTACT
Genesis of cerebellar interneurons and the prevention of neural DNA damage require XRCC1.
 Genesis of cerebellar interneurons and the prevention of neural DNA damage require XRCC1. Youngsoo Lee, Sachin Katyal, Yang Li, Sherif F. El-Khamisy, Helen R. Russell, Keith W. Caldecott and Peter J. McKinnon.
Genesis of cerebellar interneurons and the prevention of neural DNA damage require XRCC1. Youngsoo Lee, Sachin Katyal, Yang Li, Sherif F. El-Khamisy, Helen R. Russell, Keith W. Caldecott and Peter J. McKinnon.
Reversing the Effects of Fragile X Syndrome
 CLINICAL IMPLICATIONS OF BASIC RESEARCH Paul J. Lombroso, M.D., Marilee P. Ogren, Ph.D. Assistant Editors Reversing the Effects of Fragile X Syndrome MARILEE P. OGREN, PH.D., AND PAUL J. LOMBROSO, M.D.
CLINICAL IMPLICATIONS OF BASIC RESEARCH Paul J. Lombroso, M.D., Marilee P. Ogren, Ph.D. Assistant Editors Reversing the Effects of Fragile X Syndrome MARILEE P. OGREN, PH.D., AND PAUL J. LOMBROSO, M.D.
Supplementary Figure 1
 Supplementary Figure 1 Global TeNT expression effectively impairs synaptic transmission. Injection of 100 pg tent mrna leads to a reduction of vesicle mediated synaptic transmission in the spinal cord
Supplementary Figure 1 Global TeNT expression effectively impairs synaptic transmission. Injection of 100 pg tent mrna leads to a reduction of vesicle mediated synaptic transmission in the spinal cord
Supplementary Figure 1
 Supplementary Figure 1 Localization of virus injections. (a) Schematic showing the approximate center of AAV-DIO-ChR2-YFP injection sites in the NAc of Dyn-cre mice (n=8 mice, 16 injections; caudate/putamen,
Supplementary Figure 1 Localization of virus injections. (a) Schematic showing the approximate center of AAV-DIO-ChR2-YFP injection sites in the NAc of Dyn-cre mice (n=8 mice, 16 injections; caudate/putamen,
Resonant synchronization of heterogeneous inhibitory networks
 Cerebellar oscillations: Anesthetized rats Transgenic animals Recurrent model Review of literature: γ Network resonance Life simulations Resonance frequency Conclusion Resonant synchronization of heterogeneous
Cerebellar oscillations: Anesthetized rats Transgenic animals Recurrent model Review of literature: γ Network resonance Life simulations Resonance frequency Conclusion Resonant synchronization of heterogeneous
Supporting Information
 ATP from synaptic terminals and astrocytes regulates NMDA receptors and synaptic plasticity through PSD- 95 multi- protein complex U.Lalo, O.Palygin, A.Verkhratsky, S.G.N. Grant and Y. Pankratov Supporting
ATP from synaptic terminals and astrocytes regulates NMDA receptors and synaptic plasticity through PSD- 95 multi- protein complex U.Lalo, O.Palygin, A.Verkhratsky, S.G.N. Grant and Y. Pankratov Supporting
Is Intrinsic Hyperexcitability in CA3 the Culprit for Seizures in Rett Syndrome?
 Current Literature In Basic Science Is Intrinsic Hyperexcitability in CA3 the Culprit for Seizures in Rett Syndrome? Network Hyperexcitability in Hippocampal Slices From Mecp2 Mutant Mice Revealed by Voltage-Sensitive
Current Literature In Basic Science Is Intrinsic Hyperexcitability in CA3 the Culprit for Seizures in Rett Syndrome? Network Hyperexcitability in Hippocampal Slices From Mecp2 Mutant Mice Revealed by Voltage-Sensitive
Neocortex. Cortical Structures in the Brain. Neocortex Facts. Laminar Organization. Bark-like (cortical) structures: Shepherd (2004) Chapter 12
 Neocortex Shepherd (2004) Chapter 12 Rodney Douglas, Henry Markram, and Kevan Martin Instructor: Yoonsuck Choe; CPSC 644 Cortical Networks Cortical Structures in the Brain Bark-like (cortical) structures:
Neocortex Shepherd (2004) Chapter 12 Rodney Douglas, Henry Markram, and Kevan Martin Instructor: Yoonsuck Choe; CPSC 644 Cortical Networks Cortical Structures in the Brain Bark-like (cortical) structures:
Applied Neuroscience. Conclusion of Science Honors Program Spring 2017
 Applied Neuroscience Conclusion of Science Honors Program Spring 2017 Review Circle whichever is greater, A or B. If A = B, circle both: I. A. permeability of a neuronal membrane to Na + during the rise
Applied Neuroscience Conclusion of Science Honors Program Spring 2017 Review Circle whichever is greater, A or B. If A = B, circle both: I. A. permeability of a neuronal membrane to Na + during the rise
Nature Neuroscience: doi: /nn Supplementary Figure 1
 Supplementary Figure 1 Relative expression of K IR2.1 transcript to enos was reduced 29-fold in capillaries from knockout animals. Relative expression of K IR2.1 transcript to enos was reduced 29-fold
Supplementary Figure 1 Relative expression of K IR2.1 transcript to enos was reduced 29-fold in capillaries from knockout animals. Relative expression of K IR2.1 transcript to enos was reduced 29-fold
Supplementary Figure 1. ACE robotic platform. A. Overview of the rig setup showing major hardware components of ACE (Automatic single Cell
 2 Supplementary Figure 1. ACE robotic platform. A. Overview of the rig setup showing major hardware components of ACE (Automatic single Cell Experimenter) including the MultiClamp 700B, Digidata 1440A,
2 Supplementary Figure 1. ACE robotic platform. A. Overview of the rig setup showing major hardware components of ACE (Automatic single Cell Experimenter) including the MultiClamp 700B, Digidata 1440A,
mm Distance (mm)
 b a Magnet Illumination Coverslips MPs Objective 2575 µm 1875 µm 1575 µm 1075 µm 875 µm 545 µm 20µm 2 3 0.5 0.3mm 1 1000 100 10 1 0.1 1000 100 10 1 0.1 Field Induction (Gauss) 1.5 0 5 10 15 20 Distance
b a Magnet Illumination Coverslips MPs Objective 2575 µm 1875 µm 1575 µm 1075 µm 875 µm 545 µm 20µm 2 3 0.5 0.3mm 1 1000 100 10 1 0.1 1000 100 10 1 0.1 Field Induction (Gauss) 1.5 0 5 10 15 20 Distance
SUPPLEMENTARY INFORMATION
 doi:1.138/nature9553 Supplementary Table 1. Overlap of neuronal marker and PKC- expression in CEl. Marker/PKC- PKC- Marker Gad65 87.4±4.7 5.3±12.6 CRH 1.2±1. 16.9±15.2 Dyn 1.9±1.2 4.5±2.9 Enk 42.8±7.4
doi:1.138/nature9553 Supplementary Table 1. Overlap of neuronal marker and PKC- expression in CEl. Marker/PKC- PKC- Marker Gad65 87.4±4.7 5.3±12.6 CRH 1.2±1. 16.9±15.2 Dyn 1.9±1.2 4.5±2.9 Enk 42.8±7.4
