During infections, innate cytokines direct the development
|
|
|
- Felicity Welch
- 5 years ago
- Views:
Transcription
1 IFN- Is Not Sufficient to Drive Th1 Development Due to Lack of Stable T-bet Expression 1 Hilario J. Ramos, 2 * Ann M. Davis, 2 * Thaddeus C. George, and J. David Farrar 3 * During inflammatory immune responses, the innate cytokine IL-12 promotes CD4 Th-1 development through the activation of the second messenger STAT4 and the subsequent expression of T-bet. In addition, type I IFN (IFN- ), secreted primarily during viral and intracellular bacterial infections, can promote STAT4 activation in human CD4 T cells. However, the role of IFN- in regulating Th1 development is controversial, and previous studies have suggested a species-specific pathway leading to Th1 development in human but not mouse CD4 T cells. In this study, we found that although both IFN- and IL-12 can promote STAT4 activation, IFN- failed to promote Th1 commitment in human CD4 T cells. The difference between these innate signaling pathways lies with the ability of IL-12 to promote sustained STAT4 tyrosine phosphorylation, which correlated with stable T-bet expression in committed Th1 cells. IFN- did not promote Th1 development in human CD4 T cells because of attenuated STAT4 phosphorylation, which was insufficient to induce stable expression of T-bet. Further, the defect in IFN- driven Th1 development was corrected by ectopic expression of T-bet within primary naive human CD4 T cells. These results indicate that IL-12 remains unique in its ability to drive Th1 development in human CD4 T cells and that IFN- lacks this activity due to its inability to promote sustained T-bet expression. The Journal of Immunology, 2007, 179: During infections, innate cytokines direct the development of CD4 T cells toward effector Th subsets that secrete distinct cytokines. This process is initiated by pathogenassociated molecular patterns (PAMPs) 4 that are recognized by specific TLRs expressed within professional APC subsets (1, 2). In cases of bacterial infections, PAMPs such as peptidoglycan and LPS are sensed by TLRs 2 and 4, which are expressed predominantly by monocyte-derived dendritic cells (3). As a consequence, monocyte-derived dendritic cells secrete high levels of IL-12, which directly promotes CD4 Th1 development (4, 5). In contrast, virus-derived PAMPs such as double-stranded and singlestranded RNA signal through TLRs 3 and 7, respectively, both of which are potent inducers of type I IFN (IFN- ) (1, 6). Thus, IL-12 and IFN- represent two critical innate cytokines that provide instructive cues for adaptive T cell responses (7). *Department of Immunology and Department of Molecular Biology, University of Texas Southwestern Medical Center, Dallas, TX 75390; and Amnis Corporation, Seattle, WA Received for publication October 26, Accepted for publication July 12, The costs of publication of this article were defrayed in part by the payment of page charges. This article must therefore be hereby marked advertisement in accordance with 18 U.S.C. Section 1734 solely to indicate this fact. 1 This study was supported by a grant from the National Institutes of Health/National Institute of Allergy and Infectious Diseases (AI56222) awarded to J.D.F. H.J.R. was supported by a predoctoral fellowship from the National Institutes of Health/ National Institute of Allergy and Infectious Diseases (AI68622) and A.M.D. was supported by a training grant from the National Institutes of Health (GM ). 2 H.J.R. and A.M.D. contributed equally to the present work. 3 Address correspondence and reprint requests to Dr. J. David Farrar, Department of Immunology, University of Texas Southwestern Medical Center, 5323 Harry Hines Boulevard, Dallas, TX address: David.Farrar@ UTSouthwestern.edu 4 Abbreviations used in this paper: PAMP, pathogen-associated molecular pattern; cimdm, complete IMDM; GFPRV, retrovirus vector expressing GFP; h, human (prefix); IFNAR, IFN- receptor; LCMV, lymphocytic choriomeningitis virus; P-Y, tyrosine phosphorylated; qpcr, quantitative real-time PCR; rh, recombinant human (prefix); SOCS, suppressor of cytokine signaling. Copyright 2007 by The American Association of Immunologists, Inc /07/$2.00 A role for IL-12 in promoting Th1 development has been well established (5). IL-12 signaling promotes the phosphorylation of STAT4 (8, 9), which is required for Th1 commitment (10). Further, IL-12-dependent STAT4 activation is conserved across species, and disruptive mutations in this signaling pathway significantly impair Th1 responses in both mice (10, 11) and humans (12). Likewise, early reports demonstrated a potential role for IFN- in promoting Th1 development in human (h)pbmcs (13, 14). These initial studies were followed by findings that IFN-, like IL-12, could promote STAT4 activation in human lymphocytes (15 17). These early reports concluded that IFN- could directly regulate Th1 commitment in CD4 T cells through the activation of STAT4 (18). However, unlike IL-12, the role of IFN- in promoting Th1 development has been met with considerable controversy. Initial reports demonstrating IFN- -dependent STAT4 activation and Th1 development were performed with hpbmcs or with human T and NK cell lines (13 15). In parallel studies with murine T cells, IFN- was insufficient to promote Th1 development (17, 19) or activate STAT4 when compared with human CD4 T cells (16, 17), indicating a potential species-specific role for IFN- in regulating Th1 development. Further, human STAT2 was implicated in facilitating STAT4 recruitment to the human IFN- receptor (IFNAR) complex in a species-specific manner (16, 20). These findings seemed to explain the presumed ability of IFN- to promote STAT4 activation and Th1 development in human but not in murine CD4 T cells. However, CD4 T cells from STAT2 knock-in mice that expressed a humanized Stat2 gene failed to exhibit either STAT4 phosphorylation or Th1 commitment in response to IFN- activation (21). These results suggested that although human STAT2 was required for STAT4 activation in human cells, it was not sufficient to restore this pathway when expressed in the context of the murine IFNAR. Recent studies have called this species-specific pathway into question (22, 23). Biron and colleagues demonstrated that IFN- could promote STAT4 phosphorylation in murine T cells, although this effect was more pronounced in CD8 than in CD4 T cells (22). A further examination comparing various subtypes of IFN-
2 demonstrated that although murine IFN- could indeed induce weak STAT4 phosphorylation, it was insufficient to drive IFN- production and Th1 development in murine CD4 T cells (24). In a related study, Hilkens and colleagues demonstrated that even in human T cells IFN- -driven STAT4 activation was attenuated compared with the effects of IL-12 (25). This study suggested that IL-12 was more efficient than IFN- at promoting Th1 development in human CD4 T cells. One explanation was that IL-12 stimulation was able to maintain STAT4 phosphorylation over a longer period of time compared with IFN-. We have recently confirmed this observation in murine CD4 T cells (26); however, a molecular explanation for this difference in signaling between IFN- and IL-12 has not been examined. At the core of this issue is whether IL-12 and IFN- share redundant roles in Th commitment and whether there truly is a species-specific pathway that operates in human but not in murine T cells. In this study, we have addressed this central issue by comparing the ability of IL-12 and IFN- to direct the commitment of primary human CD4 T cells toward the Th1 fate. We found that IFN- does not promote Th1 development from naive human CD4 T cells, and we have linked this developmental defect to the inability of IFN- to induce sustained T-bet expression. Further, we demonstrate that in primary naive human CD4 T cells, T-bet expression is sufficient to drive Th1 commitment in a cytokineindependent manner. Thus, in contrast to previous reports, IFN- does not promote Th1 development in the absence of IL-12 because of the lack of sustained T-bet expression, and this effect is consistent across species. Materials and Methods Human subjects Peripheral blood ( ml) was obtained from healthy adults by venipuncture. Informed consent was obtained from each donor according to guidelines established by the Internal Review Board (University of Texas Southwestern Medical Center, Dallas, TX). Mice DO11.10 TCR transgenic mice were housed in specific pathogen-free conditions in accordance with guidelines established by the Institutional Animal Care and Use Committee (University of Texas Southwestern Medical Center). Cytokines, Abs, and reagents Recombinant human IL-12, IFN- A, IFN-, and anti-human IL-4 were purchased from R&D Systems. Recombinant human IL-18 was purchased from BioSource International. Recombinant human IL-2 was a generous gift from M. Bennett (University of Texas Southwestern Med. Center). Neutralizing anti-human IFNAR2 was purchased from Calbiochem. The PE- and TRI-COLOR-conjugated anti-human CD4, anti-human IFN-, antimurine-cd4, and anti-murine-ifn- were purchased from Caltag Laboratories. FITC-conjugated anti-human CD45RA Ab and PE-conjugated antihuman IL-12R 2 Abs were purchased from BD Pharmingen. Polyclonal rabbit anti-human STAT4, anti-human STAT1, and anti-human T-bet Abs were purchased from Santa Cruz Biotechnology. Anti-phospho-STAT4 was purchased from Zymed Laboratories, and anti-phospho-stat1 was purchased from Upstate Biotechnology. HRP-conjugated goat anti-rabbit Ig antiserum was purchased from Jackson ImmunoResearch Laboratories. Purification of hpbmcs and naive CD4 T cells from peripheral blood Human PBMCs (hpbmcs) were isolated by Ficoll density centrifugation using lymphocyte separation medium (Cellgro) and cultured in complete IMDM supplemented with 10% FBS (Valley Biomedical) (cimdm). hpbmcs used for purification of naive cells were stained with anti-human CD45RA-FITC and either anti-human CD4-PE or anti-human CD4-TRI- COLOR. Cells were sorted on a MoFlo cytometer (DakoCytomation) to 93% purity. Both hpbmcs and naive CD4 T cells were activated for 3 days at cells/ml in cimdm containing U/ml recombinant human (rh)il-2 using either 1 g/ml PHA (Calbiochem) or culture plates coated with 5 g/ml anti-cd3 (OKT3) and 5 g/ml anti-cd28 (clone 37.51). Cytokines and neutralizing anti-cytokine Abs were added at the following concentrations as indicated in the figures: rhil-12, 10 ng/ml; rhifn- A, 1,000 U/ml; rhifn-, 10 ng/ml; rhil-4, 10 ng/ml; anti-human IL-12 (clone 20C2), 5 g/ml; anti-human IFNAR2, 2 5 g/ml; anti-human IFN- (clone 4SB3), 5 g/ml; and anti-human IL-4, 5 g/ml. On day 3, cells were split into cimdm containing 50 U/ml rhil-2 and rested until day 7. Restimulation was performed using either 0.8 g/ml PMA (A.G. Scientific) plus 1 M ionomycin (Sigma-Aldrich) or plates coated with 5 g/ml anti-cd3 (OKT3). Intracellular cytokine and phospho-stat4 staining For intracellular cytokine staining, resting Th cells were restimulated with 0.8 g/ml PMA plus 1 M ionomycin for 4 h. Brefeldin A (1 g/ml; Epicentre Technologies) was added during the last 2 h of stimulation. For Th1 effector function assays, cells were stimulated with either PMA plus ionomycin, 10 ng/ml rhil-12, 1,000 U/ml IFN- A, 50 ng/ml IL-18, or a combination of IL-12/IL-18 or IFN- A/IL-18. After stimulation, cells were harvested and intracellular cytokine staining was performed as previously described (21). Intracellular tyrosine-phosphorylated (P-Y) STAT4 was detected by intracellular staining as described (27). Freshly isolated PBMCs were activated with cytokines as indicated in the text and were then incubated in 5% formalin/pbs followed by fixation in cold 100% methanol. Cells were washed extensively in staining buffer (PBS, 0.5% BSA, and 1 mm NaVO 4 ) followed by permeabilization in staining buffer containing 0.1% saponin. Cells were stained with 0.5 g of anti-stat4 (clone SC-486; Santa Cruz Biotechnology) or with anti-phosphotyrosine STAT4 (Zymed Laboratories). A goat-anti-mouse Ig-biotin (Jackson ImmunoResearch Laboratories) was used for secondary detection followed by staining with streptavidin- PE-Cy5. Cells were also stained with anti-hcd4-pe and anti-hcd45ra- FITC. Cells were analyzed on a FACScan (BD Biosciences) and the data were processed through CellQuest software. Analysis of cytoplasmic and nuclear STAT4 hpbmcs activated with IL-12 or IFN- for various times were stained for CD4, CD45RA, and phosphotyrosine-stat4 as described above and counterstained with 20 ng/ml propidium iodide. Alternatively, the biotinylated phosphotyrosine-stat4 was stained with streptavidin-fitc and the nucleus was counterstained with 5 M DRAQ5 (Axxora). These cells were also stained with anti-hcd4-pe. Image files of 5,000 10,000 events were collected for each sample using the ImageStream imaging flow cytometer (Amnis) and analyzed using IDEAS software (Amnis). In-focus single cells were identified by gating on propidium iodide or DRAQ5-positive events with high nuclear aspect ratios (minor to major axis ratio, a measure of circularity) and high nuclear contrast (as measured by the Gradient Max feature). CD4 CD45RA lymphocytes (low side scatter/low area cells) were then gated. Nuclear localization of phosphotyrosine-stat4 within these cells was measured using the similarity score, which quantifies the correlation of pixel values of the nuclear and phosphotyrosine-stat4 images on a per-cell basis (28). If the transcription factor is nuclear, the two images will be similar and have large positive values. If it is cytoplasmic, the two images will be anti-similar and have large negative values. Events with positive values 1 had visually apparent nuclear distributions of transcription factor and were gated to quantify the percentage of cells within the CD4 CD45RA lymphocyte population with nuclear-localized phosphotyrosine-stat4. Quantitative real-time PCR (qpcr) analysis 3793 Total RNA was extracted using the RNeasy Mini Kit (Qiagen) according to the manufacturer s instructions followed by reverse transcription. The resultant cdna was subjected to qpcr analysis with SYBR Green Master Mix (Stratagene) on a ABI7300 real-time thermocycler (Applied Biosystems). Human GAPDH was used as a reference. Relative changes in mrna expression were calculated by the 2 CT method (29), and all treatment groups were referenced to the unstimulated controls. The primers used to detect mrna transcripts are as follows: hifn-, 5 -TGATTACA AGGCTTTATCTCAGGG-3 (forward) and 5 -GGCAGTAACTGGATAG TATCACTTCAC-3 (reverse);hil-12r 2,5 -ACCTTCCCACCCATGAT GGC-3 (forward) and 5 -GAAAACAGAAAGGGAGATGTGCTG-3 (reverse); ht-bet, 5 -CGTCCAACAATGTGACCCAG-3 (forward) and 5 -GCAGTCACGGCAATGAACTG-3 (reverse); and hgapdh, 5 -AC ATCGCTCAGACACCATGG-3 (forward) and 5 -CATGTAGTTGAGGT CAATGAAGGG-3 (reverse).
3 3794 IFN- DOES NOT SUSTAIN T-BET EXPRESSION Cytometric bead array and ELISA analyses of human cytokines Naive human CD4 T cells were activated for 7 days as described above and then restimulated at cells/ml for 24 h on anti-cd3-coated plates. Supernatants were analyzed for IL-4, IL-5, and IFN- cytokine concentrations by either cytometric bead arrays (BD Pharmingen) or ELISAs (ebioscience) according to the manufacturer s protocols. Immunoprecipitation and immunoblotting hpbmcs were stimulated and expanded for two consecutive weeks with PHA under Th1 conditions (10 ng/ml rhil-12). On day 14, cells were washed in cimdm and rested at cells/ml for 30 min at 37 C in 5% CO 2. Cells were stimulated with either 10 ng/ml rhil-12 or 1,000 U/ml rhifn- A for the indicated periods of time at 37 C in 5% CO 2. Lysis was performed for 1 h at 4 C in radioimmune precipitation assay buffer containing proteinase inhibitors. Samples were precleared using rabbit preimmune serum, and immunoprecipitation of human STAT4 was performed using 3 g per sample of rabbit anti-human STAT4 bound to protein G- Sepharose beads (Amersham Biosciences). Immunoprecipitates were resolved by electrophoresis with 7% SDS-polyacrylamide gel and transferred to polyvinylidene difluoride membranes (Bio-Rad). Western blotting was performed using rabbit anti-phosphorylated STAT4 with an HRP-conjugated goat anti-rabbit secondary Ab. Detection was performed using ECL detection reagents followed by exposure to Hyperfilm ECL (Amersham Biosciences). The membranes were then stripped and reprobed with antihuman STAT4 polyclonal antiserum followed by HRP-conjugated goat anti-rabbit Ig. Retrovirus cdna expression constructs The full-length murine IFNAR2 cdna was amplified from a cdna clone (clone identifier: ; Invitrogen Life Technologies) with the primers 5 -AAAAAGATCTAGCTGAGCAGGATGCG-TTCACGG-3 (forward) and 5 -AAAAAGATCTCTTTGGAGTCATCTCATGATGTAGCC-3 (reverse). The PCR product was digested with BglII and cloned into the BamHI site of a retrovirus vector expressing GFP (GFPRV). A full-length human T-bet cdna clone (GenBank accession no. BC039739) was purchased from American Type Culture Collection. The open reading of this cdna clone was amplified by PCR with the primers 5 -AAAACTCGAGCC CGGATGGGCATCGTGGAG-3 (forward) and 5 -AAAACTCGAGCTGC TCAGTTGGGAAAATAGTTATAAAACTGTCC-3 (reverse). The PCR product was digested with XhoI and ligated into the XhoI site within the retrovirus vector GFPRV (30), and all constructs were confirmed by DNA sequencing. Retroviral transduction of primary naive human CD4 T cells Retroviral supernatants were generated by transfection of the Phoenix amphotropic cell line as described (31). Naive CD4 CD45RA human T cells were purified by cell sorting as described above. Purified cells were activated with plate-bound anti-cd3/anti-cd28 in complete medium with 600 U/ml hil-2 for 24 h. The cells were subjected to two rounds of retroviral transduction by spinning the cells at 1,000 g for 90 min in the presence of retroviral supernatants containing 5 g/ml Polybrene. Twentyfour hours after the first round of retroviral transduction, cytokines or anticytokine Abs were added to the cultures as indicated in the text and figure legends. Cells were expanded on day 7 by restimulating the cells on anti- CD3 coated plates. The cells were rested until day 14 before intracellular cytokine analysis was conducted. Retroviral transduction was performed in murine CD4 T cells as previously described (31). Briefly, infectious retrovirus supernatants were generated in the Phoenix ecotropic packaging cell line transfected with retrovirus plasmids. DO11.10 splenic T cells were transduced with retrovirus supernatants supplemented with 2 mg/ml protamine sulfate and 10 ng/ml IL-12 to generate polarized Th1 cells. Transduced T cells were purified on day 7 after activation by flow cytometric sorting based on GFP expression. Purified cells were expanded by restimulation with OVA peptide and irradiated BALB/c splenocytes. Results IFN- does not promote Th1 development in the absence of IL-12 We began this study by first assessing the ability of IFN- to promote Th1 development within highly purified naive CD4 T cells. For these experiments, human CD4 CD45RA cells were sorted to 95% purity from peripheral blood obtained from healthy adult donors. Sorted cells were then immediately activated on plate-bound anti-cd3/anti-cd28-coated plates in the presence of either purified cytokines or neutralizing anti-cytokine mabs. Seven days following primary activation, resting cells were restimulated with either PMA plus ionomycin (Fig. 1, A and D) or platebound anti-cd3 (Fig. 1, B and C) and assessed for cytokine protein or mrna expression. As shown in Fig. 1A, IL-12 was capable of enhancing IFN- expression, and this effect was more pronounced when IL-4 was neutralized during primary stimulation (Fig. 1A, condition 3). Further, IFN- did not inhibit the ability of IL-12 to induce IFN- expression either in the absence or presence of neutralizing anti-il-4 Ab (Fig. 1A, condition 4). Stimulation with IFN- alone did not promote Th1 development (Fig. 1A, condition 5), as expected and as previously demonstrated (19, 32). However, in contrast to previous reports (17, 22, 23), IFN- was also ineffective at promoting Th1 development in the absence of IL-12, and the percentage of cells capable of secreting IFN- was less in IFN- -treated cells compared with neutralizing conditions (Fig. 1A, compare conditions 1 and 2). Further, cells primed with IFN- did not induce IFN- mrna in response to anti-cd3 stimulation (Fig. 1B, condition 3), indicating a general defect in Th1 commitment. We considered the possibility that IFN- was promoting Th2 development. To address this, purified naive human CD4 T cells were activated under polarizing conditions as described above, and IFN-, IL-4, and IL-5 secretion was measured by cytometric bead arrays (Fig. 1C). IL-12 stimulation enhanced IFN- secretion (Fig. 1C, upper panel, compare conditions 1 and 2) and did not promote IL-4 or IL-5 secretion compared with neutralizing conditions (Fig. 1C, middle and lower panels, compare conditions 1 and 2). As expected, stimulation of cells in the presence of IL-4 inhibited IFN- secretion while enhancing IL-4 and IL-5 secretion (Fig. 1C, condition 3). Although IFN- did not inhibit the ability of IL-12 to promote IFN- secretion (Fig. 1C, upper panel, compare conditions 2 and 5), in the absence of IL-12 or IL-4 IFN- did not promote the secretion of either IFN- or IL-4 and IL-5 (Fig. 1C, condition 4). Thus, in the absence of IL-12 or IL-4, IFN- did not promote either Th1 or Th2 development. Each experiment described above was repeated at least three times. We observed heterogeneity in the overall percentages of cells that were capable of secreting IFN- from one donor to the next. This level of heterogeneity in IFN- expression is depicted in Fig. 1D. Here, naive CD4 CD45RA cells were purified from three separate healthy human donors and activated under neutralizing conditions (anti-il-12 plus anti-ifnar2; Control in Fig. 1D), or with IL-12 plus anti-ifnar2 ( IL-12 in Fig. 1D) or IFN- A plus anti-il-12 ( IFN- A in Fig. 1D). The percentage of IFN- -secreting CD4 T cells that developed in the absence of IL-12 or IFN- varied significantly from each of the three donors. However, in all cases IL-12 stimulation increased while IFN- decreased the percentage of cells capable of secreting IFN- compared with cells activated under neutralizing conditions. Early reports demonstrating a role for IFN- in Th1 development used mixed populations of peripheral or cord blood lymphocytes (13, 14). Thus, it was possible that IFN- was acting indirectly on other populations of cells to promote Th1 development in CD4 T cells. Therefore, we addressed this issue by attempting to reproduce those early studies. For these experiments, nonfractionated human PBMCs were activated with plate-bound anti-cd3 plus anti-cd28 in the presence of cytokines or neutralizing anticytokine Abs as indicated in Fig. 2A. Cells were expanded in culture for 7 days followed by restimulation with PMA and ionomycin to assess IFN- expression by intracellular cytokine staining. IL-12 was sufficient to increase the percentage of CD4 T cells capable of secreting IFN- as expected (Fig. 2A, condition 3), and
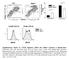
4 3795 FIGURE 1. IL-12, but not IFN-, promotes Th1 development in highly purified naive human CD4 T cells. Purified human CD4 CD45RA T cells were activated with plate-bound anti-cd3 and anti-cd28 in the presence of IL-2, anti-il-4 ( -IL-4), and the indicated cytokines and neutralizing Abs, where indicates the addition of a cytokine and indicates the addition of a neutralizing anti-cytokine Ab. On day 3, cells were diluted into fresh medium containing IL-2 and rested until day 7. A, Parallel cultures were stimulated in the absence (open bars) or presence (closed bars) of neutralizing anti-hil-4 Ab. Cells were restimulated for 4hinthepresence of PMA plus ionomycin. Intracellular cytokine staining was performed with Abs specific for hcd4 and hifn-. Data were gated on live cell populations and expressed as a percentage of CD4 IFN- cells. B, Total RNA was isolated from cells that were resting (open bars) or restimulated (closed bars) with plate-bound anti-cd3 for 2 h. Analysis of IFN- transcript levels was performed by qpcr, and transcript levels were normalized to GAPDH. Data were normalized relative to nonstimulated (control) cells activated under neutralizing conditions. C, Cells were rested (open bars) or restimulated for 24 h with plate-bound anti-cd3 (closed bars). Cell culture supernatants were analyzed for the presence of IFN- (upper panel), IL-4 (middle panel), and IL-5 (lower panel) by cytometric bead array. D, Purified CD4 CD45RA T cells from 3 separate donors (D1, D2, and D3) were stimulated as in C under neutralizing conditions ( Control ) or with IL-12 plus anti-ifnar2 ( IL-12 ) or anti-il-12 plus IFN- A ( IFN- A ). On day 7, cells were restimulated and stained for CD4 and intracellular IFN- as described above. Data were gated on live cell populations. the absence or presence of neutralizing anti-il-4 Ab did not significantly enhance this effect. However, differentiation of human PBMCs in the presence of IFN- alone was not sufficient to expand IFN- -secreting CD4 T cells compared with neutralizing conditions (Fig. 2A, compare conditions 1 and 2). Although IFN- failed to enhance Th1 development, it did not inhibit the ability of IL-12 to expand a population of IFN- -secreting CD4 T cells (Fig. 2A, condition 4). The differences observed between our data and previous reports could have resulted from our strict control of endogenous IL-12 levels with the use of neutralizing anti-il-12 Abs with cells cultured in the presence of IFN-. In some cases, previous studies did not neutralize IL-12 during the primary activation (13, 14). To address this, hpbmcs were activated in the presence of IL-12 or IFN- in the absence or presence of neutralizing Abs for 7 days (Fig. 2B). IFN- expression from CD4 T cells was assessed as described above. Even in the absence of neutralizing Abs against IL-12, IFN- alone was still unable to induce Th1 development in CD4 T cells (Fig. 2B, compare conditions 6 and 7). We considered the possibility that subtypes of IFN- other that IFN- A may differentially regulate Th1 commitment. To address this, naive human CD4 T cells were activated in parallel cultures with IFN- B2, IFN- D, IFN- A/D, and IFN-. Consistent with results obtained with IFN- A (Figs. 1 and 2) and the other IFN- subtypes (data not shown), IFN- (Fig. 3A) failed to promote Th1 development. In each case, these cultures were activated with 1,000 U/ml IFN-. Previous reports demonstrating a role for IFN- in murine Th1 development used IFN- at concentrations ranging from 50,000 to 100,000 U/ml (22, 23). Thus, it was possible that extremely high concentrations of IFN- were required to promote Th1 development. However, we titrated IFN- A in primary stimulation cultures and found that IFN- A at concentrations up to 100,000 U/ml was still incapable of driving Th1 development in human CD4 T cells (Fig. 3B). Studies of in vitro activated murine T cells suggest that repeated stimulation under Th1-polarizing conditions tends to reinforce Th1 commitment as measured by increased IFN- secretion and the inability of these cells to be redirected to the Th2 phenotype (33).
5 3796 IFN- DOES NOT SUSTAIN T-BET EXPRESSION FIGURE 2. IL-12, but not IFN-, promotes Th1 development in human PBMC cultures. A, Human PBMCs were stimulated with plate-bound anti- CD3 plus anti-cd28 in the presence of IL-2 and the indicated cytokines and/or neutralizing Abs, where indicates the addition of a cytokine, and indicates the addition of a neutralizing anti-cytokine Ab. Parallel cultures were stimulated in the absence (open bars) or presence (closed bars) of neutralizing anti-hil-4 Ab. IL-4, anti-il-4. B, Human PBMCs were activated with plate-bound anti-cd3 and anti-cd28 in the presence of IL-2 and the indicated cytokines and neutralizing Abs, where indicates the addition of a cytokine, indicates the addition of a neutralizing anti-cytokine Ab, and O indicates that the cytokine was not manipulated. On day 3, cells were diluted 1:8 into fresh medium containing IL-2 and rested to day 7. Cells were restimulated for 4hinthepresence of PMA plus ionomycin. Intracellular cytokine staining was performed with Abs specific for hcd4 and hifn-. Data were gated on live cell populations and expressed as a percentage of CD4 IFN- cells. Based on these observations, we wished to determine whether repeated IFN- stimulation could eventually lead to Th1 commitment. Naive human CD4 CD45RA T cells were purified and activated for 1 wk with plate-bound anti-cd3 plus anti-cd28 in the presence of cytokines or neutralizing anti-cytokine Abs as indicated in Fig. 3C (primary (1 ) conditions). These cells were then restimulated for a second week with either additional cytokines or neutralizing anti-cytokine Abs (secondary (2 ) conditions). IFN- mrna was quantified from cells that were restimulated with either medium or PMA plus ionomycin. Cells that were stimulated for the first week with IL-12 displayed 3- to 4-fold induction of IFN- mrna compared with cells that were differentiated under neutralizing conditions during the primary and secondary stimulations (Fig. 3C, compare conditions 1 and 2). However, cells that received IFN- during the primary stimulation alone (Fig. 3C, condition 4) or during both the primary and secondary stimulations (Fig. 3C, condition 5) failed to induce IFN- mrna compared with cells polarized under neutralizing conditions (Fig. 3C, condition 1). Although IFN- failed to promote Th1 development in the absence of IL-12, it did not significantly inhibit IFN- expression from cells differentiated in the presence of IL-12 during the primary stimulation (Fig. 3C, compare conditions 2 and 3; p FIGURE 3. Type I IFN does not promote Th1 commitment. A, Purified naive CD4 T cells were stimulated with plate-bound anti-cd3/anti-cd28 in the presence of IL-12 or IFN- (1,000 U/ml) as indicated in the figure. On day 7, cells were restimulated with PMA plus ionomycin and IFN- expression was measured by intracellular staining. B, Purified naive CD4 T cells were stimulated with IFN- at the concentrations indicated in the figure, and IFN- was measured by intracellular staining as describe above. C, Purified naive CD4 T cells were activated as described above for 7 days (primary (1 ) conditions) where indicates the addition of a cytokine and indicates the addition of a neutralizing anti-cytokine Ab. These cells were then restimulated with plate-bound anti-cd3 under the cytokine conditions indicated in the figure for an additional 7 days (secondary (2 ) conditions). Cells were then restimulated with either medium alone or PMA plus ionomycin for 1 h. Total RNA was isolated from these cells, and IFN- mrna induction was measured by qpcr. All data are referenced to the unstimulated control in condition ). Collectively, these data demonstrate that IFN- is not sufficient to promote Th1 development in human CD4 T cells. Cytokine-driven STAT4 phosphorylation and acute IFN- expression IL-12 drives Th1 development in CD4 T cells by efficiently promoting STAT4 tyrosine phosphorylation (9, 34). In addition, IL- 12, in synergy with IL-18, induces sustained secretion of IFN-
6 from Th1 cells that, in mice, has been shown to be dependent upon STAT4 (35 37). Thus, IL-12-dependent STAT4 activation plays significant roles in both Th1 development and effector function. Although both IL-12 and IFN- can promote STAT4 phosphorylation in human CD4 T cells (38, 39), several reports have suggested that either the magnitude or the kinetics of IFN- -induced STAT4 phosphorylation is decreased compared with IL-12 signaling (25, 38). Based on these observations, we wished to determine which aspects of Th1 development and effector function could be directly regulated by IFN- through STAT4 activation. First, the kinetics of IL-12- and IFN- -dependent STAT4 tyrosine phosphorylation were assessed in human PBMCs by immunoblotting (Fig. 4, A and B). IL-12 stimulation of Th1-polarized PBMCs promoted STAT4 phosphorylation that was sustained up to 24 h poststimulation (Fig. 4A, upper panel, lanes 2 7). In contrast, IFN- A-dependent STAT4 phosphorylation was attenuated (Fig. 4A, upper panel, lanes 8 13), peaking at h and extinguished by 6 h (Fig. 4B, lanes 8 13). In addition, IFN- -dependent STAT1 phosphorylation was sustained up to 6 h poststimulation (Fig. 4A, lower panel, lanes 8 13), indicating that the rate at which STAT4 and STAT1 are dephosphorylated may be regulated through different mechanisms. We wished to determine whether the kinetics of IFN- -dependent STAT4 tyrosine phosphorylation differed between fully polarized Th1 cells and uncommitted naive CD4 T cell progenitors. To address this, we assessed the kinetics of STAT4 tyrosine phosphorylation in naive CD4 CD45RA T cells within freshly isolated human PBMCs (Fig. 4, C G). For these experiments, both total STAT4 protein and P-Y STAT4 were measured by intracellular staining from cells activated with either IL-12 or IFN- between 0 and 24 h. The overall expression of STAT4 protein in CD4 CD45RA cells was not altered by cytokine treatment (Fig. 4C) and remained constant throughout the 24 h time course (data not shown). In naive CD4 CD45RA T cells, IL-12 did not increase the percentage of cells displaying elevated P-Y STAT4 compared with unstimulated cells (Fig. 4D), presumably due to the lack of IL-12R 2 expression (32). However, IFN- stimulation promoted STAT4 phosphorylation in naive CD4 T cells (Fig. 4D). Further, consistent with our observations in Th1 cells (Fig. 4, A and B), we found that IFN- -driven STAT4 phosphorylation was rapidly induced by 30 min and then extinguished by 6 h poststimulation in naive CD4 T cells (Fig. 4, E and G). Further, the kinetics of IFN- -dependent STAT4 phosphorylation was similar in the CD45RA and CD45RA populations (Fig. 4, F and G). We also observed that IL-12 promoted STAT4 phosphorylation in 15 20% of the CD45RA cells (Fig. 4F), indicating that these cells were responsive to IL-12. Further, IL-12-dependent STAT4 phosphorylation was maintained in this population through the entire 24-h time course. Taken together, these data indicate that unlike IL-12, IFN- does not promote sustained STAT4 tyrosine phosphorylation in naive human CD4 T cells. It was possible that the reason IFN- failed to promote Th1 development was due to lack of sustained STAT4 phosphorylation. However, it was also possible that phosphorylated STAT4 was not efficiently translocated to the nucleus in response to IFN-. This possibility was addressed by visualizing the relative nuclear accumulation of phosphorylated STAT4 within naive human CD4 T cells (Fig. 4, H and I). We used a novel technology that combines multiparametric flow cytometry with single cell microscopy. For these experiments, freshly isolated human PBMCs were stimulated with either IL-12 or IFN- followed by intracellular staining for CD4, CD45RA, and P-Y STAT4 as described above. Cells were imaged with the use of an ImageStream flow cytometer. Single cell images were processed through the live, CD4, and CD45RA 3797 gates. Cells that exhibited an increase in phosphorylated STAT4 signal were divided into populations that displayed either P-Y STAT4 cytoplasmic accumulation (Fig. 4H, left panel) or nuclear accumulation (Fig. 4H, right panel). As shown in Fig. 4I, 50% of the CD4 CD45RA P-Y STAT4 cells displayed accumulation of P-Y STAT4 in the nuclei in response to IFN-. Although the level of P-Y STAT4 was significantly diminished by 6 h (Fig. 4G), the residual amount of P-Y STAT4 within those cells was still retained within the nucleus (Fig. 4I). Thus, these data suggest that although IFN- -induced STAT4 phosphorylation was attenuated compared with IL-12, STAT4 nuclear translocation remained intact in naive human CD4 T cells. We wished to correlate the duration of IFN- -dependent STAT4 phosphorylation with Th1 effector function. IFN- secretion by Th1 cells is regulated independently by both Ag activation and innate cytokines (36, 40, 41). In the case of innate cytokines, IL-12 synergizes with IL-18 to promote IFN- secretion from fully polarized Th1 cells in a STAT4-dependent manner. Although IFN- plus IL-18 stimulation was reported to induce IFN- secretion from human T cells (39), we suspected that IFN- and IL-12 differentially regulated acute IFN- secretion because of the altered kinetics of STAT4 activation observed above. To test this possibility, human CD4 T cells were activated under Th1-inducing conditions (IL-12 plus -IL-4) to generate a population of cells capable of responding to IL-12 and secreting IFN- upon restimulation with innate cytokines. Human Th1 cells restimulated for 4 h with either IL-12 plus IL-18 or IFN- A plus IL-18 secreted IFN- to equivalent levels (Fig. 5, A and B). Stimulation with each cytokine alone failed to promote IFN- secretion (Fig. 5B) as previously reported (36, 38). However, if cells were stimulated for 24 h, the level of IFN- secreted into the culture supernatants was significantly lower in response to IFN- plus IL-18 as compared with IL-12 plus IL-18 (Fig. 5C). These data correlate with the attenuated activation of STAT4 in response to IFN- observed above. Ectopic IFNAR2 expression enhances IFN- -dependent STAT4 phosphorylation and acute IFN- secretion Several mechanisms could account for the differential activation of STAT4 by IL-12 vs IFN-. First, IFN- signaling may induce the expression of negative regulators of JAK/STAT signaling such as suppressor of cytokine signaling (SOCS) proteins or Ubp43. However, we found that SOCS mrna induction was equivalent in cells activated with either IL-12 or IFN- (data not shown). In addition to SOCS proteins, Ubp43 has recently been shown to specifically inhibit JAK kinase activation by the IFNAR (42), and Ubp43- deficient mice display enhanced antiviral and antibacterial responses to IFN- signaling (43, 44). We examined STAT4 activation and IFN- secretion in Ubp43-deficient T cells but detected no differences in these parameters compared with strain-matched wild-type control T cells (data not shown). Previous studies have suggested that acute IFN- signaling negatively regulates expression of the human IFNAR1 subunit, thereby extinguishing downstream signaling events (25). However, Berenson et al. found that the murine IFNAR1 subunit was stably expressed on murine CD4 T cells following IFN- activation (26). This recent study concluded that, as in human T cells, STAT4 tyrosine phosphorylation was attenuated in response to IFN- compared with IL-12. Further, we recently demonstrated that the IFNAR2 subunit plays an important role in STAT4 activation (45). In that study, we found that the STAT4 N-domain interacts with the cytoplasmic domain of the IFNAR2 subunit. Based on these collective observations, we wished to determine whether ectopic overexpression of either the IFNAR1 or IFNAR2
7 3798 IFN- DOES NOT SUSTAIN T-BET EXPRESSION FIGURE 4. IL-12 and IFN- differentially regulate the kinetics of STAT4 tyrosine phosphorylation in human CD4 T cells. A and B, Human PBMCs were activated and expanded for two consecutive weeks with PHA in the presence of IL-2 and IL-12. On day 14, cells were rested in fresh medium for 30 min and stimulated with either IL-12 or IFN- A for the indicated periods of time. Immunoprecipitation (IP) was performed using polyclonal antisera for human STAT4 or STAT1, and Western blotting was performed for phosphorylated STAT4 or STAT1, as indicated. Membranes were then stripped and reprobed for total STAT4 or STAT1. A, Time course of STAT4 and STAT1 phosphorylation in human Th1 PBMCs stimulated with IL-12 (lanes 2 7) or IFN- A (lanes 8 13) for 0 24 h. B, Time course of STAT4 phosphorylation in human Th1 PBMCs stimulated with IL-12 (lanes 2 7) or IFN- A (lanes 8 13) for 0 6 h. C G, Freshly isolated PBMCs were activated with medium alone, IL-12, or IFN- at various time points up to 24 h. Cells were stained for CD4, CD45RA, and intracellular STAT4 and P-Y STAT4 as described in the Materials and Methods. C and D, Total STAT4 (C) and P-Y STAT4 (D) were measured from cells activated for 30 min by flow cytometry. Data are gated on live, CD4, and CD45RA cells. Black line, Unstimulated; green line, IL-12 stimulated; red line, IFN- stimulated; gray shaded, nonimmune rabbit Ig control. E, Gating scheme is shown for the analysis of CD4, CD45RA (R2), and CD45RA (R3) cells. F and G, CD45RA (F) and CD45RA (G) gated cells are represented as a percentage of cells that display increased P-Y STAT4 staining over a 24 h period., Unstimulated;, IL-12 stimulated; E, IFN- stimulated. H and I, hpbmcs were activated and stained as described above and analyzed on the ImageStream imaging flow cytometer. Single cell images were gated on the live, CD4, and CD45RA populations. H, Cells displaying positive staining for P-Y STAT4 were categorized for either low similarity (left panel) or high similarity (right panel) of colocalization of P-Y STAT4 within the nucleus based on nuclear dye staining (Draq-5). I, Cells displaying elevated P-Y STAT4 staining were quantified for colocalization of P-Y STAT4 within the nucleus of cells treated with IL-12 (f) or IFN- (Œ). subunit could restore IFN- -dependent STAT4 activation in murine T cells. To test this, we expressed the full length murine IF- NAR1 and IFNAR2 subunits in murine Th1 cells and tested both the kinetics of STAT4 phosphorylation and acute IFN- secretion in response to IFN-. Retroviral expression of the IFNAR1 did not alter either the kinetics of STAT4 phosphorylation or the secretion
8 FIGURE 5. Cytokine-dependent IFN- secretion from fully polarized human Th1 cells. Purified human CD4 CD45RA T cells were activated with plate-bound anti-cd3 and anti-cd28 in the presence of IL-2, anti- IL-4, anti-ifnar2, and IL-12 for 3 days (Th1-inducing conditions). Cells were diluted into fresh medium and rested to day 7. A and B, Cells were restimulated for 4hinthepresence of IL-12, IFN- A, IL-18, or a combination of these cytokines. PMA plus ionomycin (Iono) was used as a positive control. Intracellular cytokine staining was performed with Abs specific for hcd4 and hifn-. Data were gated on live cell populations. A, Representative dot plots showing unstimulated cells and cells stimulated in the presence of IL-12 plus IL-18 or IFN- A plus IL-18. B, Graphical representation of the proportion of CD4 IFN- cells. C, Cells were restimulated for 24 h in the presence of the indicated cytokines or with platebound anti-cd3 Ab ( -CD3) as described above. Cell culture supernatants were harvested and analyzed for the presence of IFN- protein by ELISA. of IFN- in response to IFN- plus IL-18 stimulation (data not shown). However, as shown in Fig. 6A, we found that expression of the IFNAR2 subunit increased both the magnitude and duration of IFN- -dependent STAT4 tyrosine phosphorylation. In contrast, IL-12-induced STAT4 activation was not altered by expression of the IFNAR2 subunit. The increased duration of STAT4 activation correlated well with enhanced induction of IFN- in response to IFN- plus IL-18 stimulation (Fig. 6B). Here, purified transduced cells were activated for 24 h with either IL-12 plus IL-18 or with IFN- plus IL-18 to assess IFN- secretion. IL-12 plus IL-18 activated cells displayed 60% IFN- cells, and this effect was independent of the expression of the IFNAR2 subunit. However, we found that expression of the IFNAR2 subunit increased the IFN- population by 10-fold in response to IFN- plus IL-18 activation (Fig. 6, B and C). Differential induction of T-bet expression by IL-12 and IFN- The differential kinetics of STAT4 phosphorylation observed in response to IL-12 and IFN- could result in differences in the 3799 commitment of naive human CD4 T cells to the Th1 phenotype. One aspect of Th1 commitment is the acquisition of IL-12 responsiveness mediated through induction of the IL-12R 2 subunit (46, 47). Because both IL-12 and IFN- have been reported to induce IL-12R 2 expression (32), we examined whether IL-12 and IFN- differed in their induction of IL-12R 2 in developing human CD4 T cells. For these experiments, naive human CD4 CD45RA cells were purified from human peripheral blood and activated with plate-bound anti-cd3 plus anti-cd28 with the cytokines or neutralizing anti-cytokine Abs indicated in Fig. 7. When analyzed at 48 h poststimulation, both IL-12 and IFN- were able to promote expression of IL-12R 2 cell surface protein (Fig. 7A) and mrna transcripts (Fig. 7B, upper panel) to similar levels. In addition, IL-12 plus IFN- acted synergistically to promote enhanced IL-12R 2 expression at 48 h (Fig. 7, A, and B, upper panel). However, IFN- failed to maintain IL-12R 2 expression in cells activated for 7 days (Fig. 7B). In mice, T-bet is known to be involved in the regulation of IL-12R 2 (46, 48) and also acts downstream of IL-12 signaling to promote Th1 development (49 51). In addition, previous studies have demonstrated that IFN- induces expression of both IL-12 2 (32) and T-bet (52) in human CD4 T cells. Thus, it has been assumed that IL-12 and IFN- play redundant roles in Th1 commitment in human T cells through the induction of T-bet. However, given that IFN- failed to induce Th1 development (Figs. 1 3), we re-examined the ability of IL-12 and IFN- to regulate T-bet expression during the early phases of Th1 commitment and in fully polarized Th1 cells. Similar to the regulation of IL-12R 2, both IL-12 and IFN- independently induced expression of T-bet at 48 h, and IL-12 plus IFN- costimulation further enhanced T- bet expression (Fig. 7B, lower panel). However, T-bet expression was not maintained in response to IFN- alone in fully polarized cells analyzed on day 7 (Fig. 7B, lower panel). The low expression of T-bet mrna in response to IFN- correlated well with decreased T-bet protein accumulation in these cells (Fig. 7C). Based on these observations, we considered the possibility that IFN- failed to promote Th1 development because of the lack of stable T-bet expression within polarized Th cells. If this hypothesis is correct, then Th1 development should be restored by ectopic T-bet expression regardless of initial polarizing conditions. To test this hypothesis, we expressed T-bet by retroviral transduction of primary naive human CD4 T cells (Fig. 8). For these experiments, purified CD4 CD45RA T cells were activated with platebound anti-cd3 plus anti-cd28 and transduced with retrovirus constructs expressing GFP only (GFPRV) or human T-bet and GFP (ht-bet-gfp). Upon retroviral transduction, polarizing cytokine conditions were imposed with the addition of cytokines or neutralizing anti-cytokine Abs as indicated in Fig. 8. These cells were expanded on day 7 with plate-bound anti-cd3, and IFN- expression was assessed on day 14 by intracellular cytokine staining. We were able to achieve 15 20% transduction efficiency as measured by GFP expression, and the non-gfp expressing cells served as an additional internal negative control. As shown in Fig. 8A, cells transduced with the ht-bet-gfp construct were gated on GFP and GFP populations. Within the GFP- population, IL-12 increased the frequency of IFN- cells whereas IFN- did not promote IFN- expression, as expected. However, expression of T-bet within the GFP population increased the percentage of cells capable of secreting IFN-, and this effect was independent of initial cytokine conditions (Fig. 8A, compare left and right panels). Expression of GFP alone from the GFPRV had little effect on IFN- expression (Fig. 8B). Further, T-bet was sufficient to promote IFN- expression even when cells were polarized under
9 3800 IFN- DOES NOT SUSTAIN T-BET EXPRESSION FIGURE 6. Ectopic IFNAR2 expression promotes sustained STAT4 phosphorylation and IFN- secretion in response to IFN-. A, Spleen and lymph node cells from DO11.10 mice were activated with OVA peptide under Th1- inducing conditions and transduced with retrovirus vectors expressing GFP alone (GFPRV) or the full-length mifnar2 subunit. Cells were sorted on day 7 based on GFP expression and restimulated with irradiated BALB/c splenocytes and OVA peptide. Following expansion for an additional 7 days, resting cells were activated with either IL-12 or IFN- for the times indicated in the figure. Cells were then stained and analyzed for intracellular P-Y STAT4 as described in Fig. 5. B and C, Day 14 transduced Th1 cells were activated with either IL-12 plus IL-18 or IFN- plus IL-18 or with the individual cytokines FIGURE 7. IFN- does not promote stable T-bet expression in human CD4 T cells. Purified human CD4 CD45RA T cells were activated with plate-bound anti-cd3 plus anti-cd28, IL-2, and anti-il-4 and with anti- IFNAR2 and anti-il-12 ( Ctl ), anti-ifnar2 and IL-12 ( IL-12 ), IFN- A and anti-il-12 ( IFN- A ), or with IFN- A and IL-12 ( IL-12 IFN- A ). A, After 72 h, cells were stained for surface expression of IL-12R 2; filled histogram, Neutralizing Abs alone; dashed line, IL-12 plus anti-ifnar2; dotted line, IFN- A plus anti-il-12; solid line, IL-12 plus IFN- A. B, Total RNA was isolated from cells harvested 48 h or 7 days after activation. Analysis of IL-12R 2 and T-bet transcript levels was performed by qpcr using the primers listed in Materials and Methods. Transcript levels for each condition were normalized to GAPDH, and the data were further normalized relative to cells activated under neutralizing conditions. C, Whole-cell lysates were prepared from day 7 activated cells and assessed for expression of T-bet protein by Western blotting (upper panel). Blots were stripped and re-probed for lamin protein expression (lower panel). Th2-inducing conditions (Fig. 8, A, bottom panels, and B, condition 8). Thus, these data place T-bet downstream of STAT4 and suggest that T-bet is sufficient to mediate Th1 development in indicated in the figure for 24 h. Brefeldin A was added during the last 4 h of stimulation. Cells were then stained for murine CD4 and IFN- and analyzed by flow cytometry. Data were gated on live cells and GFP expression.
Supplementary Information
 Supplementary Information Supplementary Figure 1. CD4 + T cell activation and lack of apoptosis after crosslinking with anti-cd3 + anti-cd28 + anti-cd160. (a) Flow cytometry of anti-cd160 (5D.10A11) binding
Supplementary Information Supplementary Figure 1. CD4 + T cell activation and lack of apoptosis after crosslinking with anti-cd3 + anti-cd28 + anti-cd160. (a) Flow cytometry of anti-cd160 (5D.10A11) binding
Supplementary Figure 1 Role of Raf-1 in TLR2-Dectin-1-mediated cytokine expression
 Supplementary Figure 1 Supplementary Figure 1 Role of Raf-1 in TLR2-Dectin-1-mediated cytokine expression. Quantitative real-time PCR of indicated mrnas in DCs stimulated with TLR2-Dectin-1 agonist zymosan
Supplementary Figure 1 Supplementary Figure 1 Role of Raf-1 in TLR2-Dectin-1-mediated cytokine expression. Quantitative real-time PCR of indicated mrnas in DCs stimulated with TLR2-Dectin-1 agonist zymosan
Optimizing Intracellular Flow Cytometry:
 Optimizing Intracellular Flow Cytometry: Simultaneous Detection of Cytokines and Transcription Factors An encore presentation by Jurg Rohrer, PhD, BD Biosciences 10.26.10 Outline Introduction Cytokines
Optimizing Intracellular Flow Cytometry: Simultaneous Detection of Cytokines and Transcription Factors An encore presentation by Jurg Rohrer, PhD, BD Biosciences 10.26.10 Outline Introduction Cytokines
Cover Page. The handle holds various files of this Leiden University dissertation.
 Cover Page The handle http://hdl.handle.net/1887/23854 holds various files of this Leiden University dissertation. Author: Marel, Sander van der Title: Gene and cell therapy based treatment strategies
Cover Page The handle http://hdl.handle.net/1887/23854 holds various files of this Leiden University dissertation. Author: Marel, Sander van der Title: Gene and cell therapy based treatment strategies
Supplemental Information. T Cells Enhance Autoimmunity by Restraining Regulatory T Cell Responses via an Interleukin-23-Dependent Mechanism
 Immunity, Volume 33 Supplemental Information T Cells Enhance Autoimmunity by Restraining Regulatory T Cell Responses via an Interleukin-23-Dependent Mechanism Franziska Petermann, Veit Rothhammer, Malte
Immunity, Volume 33 Supplemental Information T Cells Enhance Autoimmunity by Restraining Regulatory T Cell Responses via an Interleukin-23-Dependent Mechanism Franziska Petermann, Veit Rothhammer, Malte
Supplementary Figure 1. Normal T lymphocyte populations in Dapk -/- mice. (a) Normal thymic development in Dapk -/- mice. Thymocytes from WT and Dapk
 Supplementary Figure 1. Normal T lymphocyte populations in Dapk -/- mice. (a) Normal thymic development in Dapk -/- mice. Thymocytes from WT and Dapk -/- mice were stained for expression of CD4 and CD8.
Supplementary Figure 1. Normal T lymphocyte populations in Dapk -/- mice. (a) Normal thymic development in Dapk -/- mice. Thymocytes from WT and Dapk -/- mice were stained for expression of CD4 and CD8.
Supplementary Materials for
 immunology.sciencemag.org/cgi/content/full/2/16/eaan6049/dc1 Supplementary Materials for Enzymatic synthesis of core 2 O-glycans governs the tissue-trafficking potential of memory CD8 + T cells Jossef
immunology.sciencemag.org/cgi/content/full/2/16/eaan6049/dc1 Supplementary Materials for Enzymatic synthesis of core 2 O-glycans governs the tissue-trafficking potential of memory CD8 + T cells Jossef
Supplementary Figure 1 CD4 + T cells from PKC-θ null mice are defective in NF-κB activation during T cell receptor signaling. CD4 + T cells were
 Supplementary Figure 1 CD4 + T cells from PKC-θ null mice are defective in NF-κB activation during T cell receptor signaling. CD4 + T cells were isolated from wild type (PKC-θ- WT) or PKC-θ null (PKC-θ-KO)
Supplementary Figure 1 CD4 + T cells from PKC-θ null mice are defective in NF-κB activation during T cell receptor signaling. CD4 + T cells were isolated from wild type (PKC-θ- WT) or PKC-θ null (PKC-θ-KO)
MATERIALS AND METHODS. Neutralizing antibodies specific to mouse Dll1, Dll4, J1 and J2 were prepared as described. 1,2 All
 MATERIALS AND METHODS Antibodies (Abs), flow cytometry analysis and cell lines Neutralizing antibodies specific to mouse Dll1, Dll4, J1 and J2 were prepared as described. 1,2 All other antibodies used
MATERIALS AND METHODS Antibodies (Abs), flow cytometry analysis and cell lines Neutralizing antibodies specific to mouse Dll1, Dll4, J1 and J2 were prepared as described. 1,2 All other antibodies used
SUPPLEMENTARY INFORMATION
 Complete but curtailed T-cell response to very-low-affinity antigen Dietmar Zehn, Sarah Y. Lee & Michael J. Bevan Supp. Fig. 1: TCR chain usage among endogenous K b /Ova reactive T cells. C57BL/6 mice
Complete but curtailed T-cell response to very-low-affinity antigen Dietmar Zehn, Sarah Y. Lee & Michael J. Bevan Supp. Fig. 1: TCR chain usage among endogenous K b /Ova reactive T cells. C57BL/6 mice
ECM1 controls T H 2 cell egress from lymph nodes through re-expression of S1P 1
 ZH, Li et al, page 1 ECM1 controls T H 2 cell egress from lymph nodes through re-expression of S1P 1 Zhenhu Li 1,4,Yuan Zhang 1,4, Zhiduo Liu 1, Xiaodong Wu 1, Yuhan Zheng 1, Zhiyun Tao 1, Kairui Mao 1,
ZH, Li et al, page 1 ECM1 controls T H 2 cell egress from lymph nodes through re-expression of S1P 1 Zhenhu Li 1,4,Yuan Zhang 1,4, Zhiduo Liu 1, Xiaodong Wu 1, Yuhan Zheng 1, Zhiyun Tao 1, Kairui Mao 1,
Supplementary data Supplementary Figure 1 Supplementary Figure 2
 Supplementary data Supplementary Figure 1 SPHK1 sirna increases RANKL-induced osteoclastogenesis in RAW264.7 cell culture. (A) RAW264.7 cells were transfected with oligocassettes containing SPHK1 sirna
Supplementary data Supplementary Figure 1 SPHK1 sirna increases RANKL-induced osteoclastogenesis in RAW264.7 cell culture. (A) RAW264.7 cells were transfected with oligocassettes containing SPHK1 sirna
Detailed step-by-step operating procedures for NK cell and CTL degranulation assays
 Supplemental methods Detailed step-by-step operating procedures for NK cell and CTL degranulation assays Materials PBMC isolated from patients, relatives and healthy donors as control K562 cells (ATCC,
Supplemental methods Detailed step-by-step operating procedures for NK cell and CTL degranulation assays Materials PBMC isolated from patients, relatives and healthy donors as control K562 cells (ATCC,
Supplemental Figure 1
 Supplemental Figure 1 1a 1c PD-1 MFI fold change 6 5 4 3 2 1 IL-1α IL-2 IL-4 IL-6 IL-1 IL-12 IL-13 IL-15 IL-17 IL-18 IL-21 IL-23 IFN-α Mut Human PD-1 promoter SBE-D 5 -GTCTG- -1.2kb SBE-P -CAGAC- -1.kb
Supplemental Figure 1 1a 1c PD-1 MFI fold change 6 5 4 3 2 1 IL-1α IL-2 IL-4 IL-6 IL-1 IL-12 IL-13 IL-15 IL-17 IL-18 IL-21 IL-23 IFN-α Mut Human PD-1 promoter SBE-D 5 -GTCTG- -1.2kb SBE-P -CAGAC- -1.kb
PBMC from each patient were suspended in AIM V medium (Invitrogen) with 5% human
 Anti-CD19-CAR transduced T-cell preparation PBMC from each patient were suspended in AIM V medium (Invitrogen) with 5% human AB serum (Gemini) and 300 international units/ml IL-2 (Novartis). T cell proliferation
Anti-CD19-CAR transduced T-cell preparation PBMC from each patient were suspended in AIM V medium (Invitrogen) with 5% human AB serum (Gemini) and 300 international units/ml IL-2 (Novartis). T cell proliferation
Blocking antibodies and peptides. Rat anti-mouse PD-1 (29F.1A12, rat IgG2a, k), PD-
 Supplementary Methods Blocking antibodies and peptides. Rat anti-mouse PD-1 (29F.1A12, rat IgG2a, k), PD- L1 (10F.9G2, rat IgG2b, k), and PD-L2 (3.2, mouse IgG1) have been described (24). Anti-CTLA-4 (clone
Supplementary Methods Blocking antibodies and peptides. Rat anti-mouse PD-1 (29F.1A12, rat IgG2a, k), PD- L1 (10F.9G2, rat IgG2b, k), and PD-L2 (3.2, mouse IgG1) have been described (24). Anti-CTLA-4 (clone
Nature Medicine: doi: /nm.2109
 HIV 1 Infects Multipotent Progenitor Cells Causing Cell Death and Establishing Latent Cellular Reservoirs Christoph C. Carter, Adewunmi Onafuwa Nuga, Lucy A. M c Namara, James Riddell IV, Dale Bixby, Michael
HIV 1 Infects Multipotent Progenitor Cells Causing Cell Death and Establishing Latent Cellular Reservoirs Christoph C. Carter, Adewunmi Onafuwa Nuga, Lucy A. M c Namara, James Riddell IV, Dale Bixby, Michael
Supporting Information
 Supporting Information van der Windt et al. 10.1073/pnas.1221740110 SI Materials and Methods Mice and Reagents. C57BL/6 and major histocompatibility complex class I-restricted OVA-specific T-cell receptor
Supporting Information van der Windt et al. 10.1073/pnas.1221740110 SI Materials and Methods Mice and Reagents. C57BL/6 and major histocompatibility complex class I-restricted OVA-specific T-cell receptor
HEK293FT cells were transiently transfected with reporters, N3-ICD construct and
 Supplementary Information Luciferase reporter assay HEK293FT cells were transiently transfected with reporters, N3-ICD construct and increased amounts of wild type or kinase inactive EGFR. Transfections
Supplementary Information Luciferase reporter assay HEK293FT cells were transiently transfected with reporters, N3-ICD construct and increased amounts of wild type or kinase inactive EGFR. Transfections
SUPPLEMENTARY INFORMATION
 Supplemental Figure 1. Furin is efficiently deleted in CD4 + and CD8 + T cells. a, Western blot for furin and actin proteins in CD4cre-fur f/f and fur f/f Th1 cells. Wild-type and furin-deficient CD4 +
Supplemental Figure 1. Furin is efficiently deleted in CD4 + and CD8 + T cells. a, Western blot for furin and actin proteins in CD4cre-fur f/f and fur f/f Th1 cells. Wild-type and furin-deficient CD4 +
Optimizing Intracellular Flow Cytometry
 Optimizing Intracellular Flow Cytometry Detection of Cytokines, Transcription Factors, and Phosphoprotein by Flow Cytometry Presented by Erika O Donnell, PhD, BD Biosciences 23-14876-00 Outline Basic principles
Optimizing Intracellular Flow Cytometry Detection of Cytokines, Transcription Factors, and Phosphoprotein by Flow Cytometry Presented by Erika O Donnell, PhD, BD Biosciences 23-14876-00 Outline Basic principles
Supplementary Figure 1. Efficient DC depletion in CD11c.DOG transgenic mice
 Supplementary Figure 1. Efficient DC depletion in CD11c.DOG transgenic mice (a) CD11c.DOG transgenic mice (tg) were treated with 8 ng/g body weight (b.w.) diphtheria toxin (DT) i.p. on day -1 and every
Supplementary Figure 1. Efficient DC depletion in CD11c.DOG transgenic mice (a) CD11c.DOG transgenic mice (tg) were treated with 8 ng/g body weight (b.w.) diphtheria toxin (DT) i.p. on day -1 and every
Supplementary Figure 1 IL-27 IL
 Tim-3 Supplementary Figure 1 Tc0 49.5 0.6 Tc1 63.5 0.84 Un 49.8 0.16 35.5 0.16 10 4 61.2 5.53 10 3 64.5 5.66 10 2 10 1 10 0 31 2.22 10 0 10 1 10 2 10 3 10 4 IL-10 28.2 1.69 IL-27 Supplementary Figure 1.
Tim-3 Supplementary Figure 1 Tc0 49.5 0.6 Tc1 63.5 0.84 Un 49.8 0.16 35.5 0.16 10 4 61.2 5.53 10 3 64.5 5.66 10 2 10 1 10 0 31 2.22 10 0 10 1 10 2 10 3 10 4 IL-10 28.2 1.69 IL-27 Supplementary Figure 1.
Supplementary Table; Supplementary Figures and legends S1-S21; Supplementary Materials and Methods
 Silva et al. PTEN posttranslational inactivation and hyperactivation of the PI3K/Akt pathway sustain primary T cell leukemia viability Supplementary Table; Supplementary Figures and legends S1-S21; Supplementary
Silva et al. PTEN posttranslational inactivation and hyperactivation of the PI3K/Akt pathway sustain primary T cell leukemia viability Supplementary Table; Supplementary Figures and legends S1-S21; Supplementary
Supplementary Data Table of Contents:
 Supplementary Data Table of Contents: - Supplementary Methods - Supplementary Figures S1(A-B) - Supplementary Figures S2 (A-B) - Supplementary Figures S3 - Supplementary Figures S4(A-B) - Supplementary
Supplementary Data Table of Contents: - Supplementary Methods - Supplementary Figures S1(A-B) - Supplementary Figures S2 (A-B) - Supplementary Figures S3 - Supplementary Figures S4(A-B) - Supplementary
Primary Adult Naïve CD4+ CD45RA+ Cells. Prepared by: David Randolph at University of Alabama, Birmingham
 Primary Adult Naïve CD4+ CD45RA+ Cells Prepared by: David Randolph (drdrdr@uab.edu) at University of Alabama, Birmingham Goal: To obtain large numbers of highly pure primary CD4+ CD45RO- CD25- cells from
Primary Adult Naïve CD4+ CD45RA+ Cells Prepared by: David Randolph (drdrdr@uab.edu) at University of Alabama, Birmingham Goal: To obtain large numbers of highly pure primary CD4+ CD45RO- CD25- cells from
Recombinant Protein Expression Retroviral system
 Recombinant Protein Expression Retroviral system Viruses Contains genome DNA or RNA Genome encased in a protein coat or capsid. Some viruses have membrane covering protein coat enveloped virus Ø Essential
Recombinant Protein Expression Retroviral system Viruses Contains genome DNA or RNA Genome encased in a protein coat or capsid. Some viruses have membrane covering protein coat enveloped virus Ø Essential
Supplementary Figure S1: Alignment of CD28H. (a) Alignment of human CD28H with other known B7 receptors. (b) Alignment of CD28H orthologs.
 Supplementary Figure S1: Alignment of CD28H. (a) Alignment of human CD28H with other known B7 receptors. (b) Alignment of CD28H orthologs. Predicted CD28H protein sequences from human, chimpanzee (pan
Supplementary Figure S1: Alignment of CD28H. (a) Alignment of human CD28H with other known B7 receptors. (b) Alignment of CD28H orthologs. Predicted CD28H protein sequences from human, chimpanzee (pan
Optimizing Intracellular Flow Cytometry:
 Optimizing Intracellular Flow Cytometry: Simultaneous Detection of Cytokines and Transcription Factors Presented by Jurg Rohrer, PhD, BD Biosciences 23-10780-00 Outline Introduction Cytokines Transcription
Optimizing Intracellular Flow Cytometry: Simultaneous Detection of Cytokines and Transcription Factors Presented by Jurg Rohrer, PhD, BD Biosciences 23-10780-00 Outline Introduction Cytokines Transcription
Nature Medicine: doi: /nm.3922
 Title: Glucocorticoid-induced tumor necrosis factor receptor-related protein co-stimulation facilitates tumor regression by inducing IL-9-producing helper T cells Authors: Il-Kyu Kim, Byung-Seok Kim, Choong-Hyun
Title: Glucocorticoid-induced tumor necrosis factor receptor-related protein co-stimulation facilitates tumor regression by inducing IL-9-producing helper T cells Authors: Il-Kyu Kim, Byung-Seok Kim, Choong-Hyun
SUPPLEMENTARY INFORMATION
 doi: 1.138/nature775 4 O.D. (595-655) 3 1 -ζ no antibody isotype ctrl Plated Soluble 1F6 397 7H11 Supplementary Figure 1 Soluble and plated anti- Abs induce -! signalling. B3Z cells stably expressing!
doi: 1.138/nature775 4 O.D. (595-655) 3 1 -ζ no antibody isotype ctrl Plated Soluble 1F6 397 7H11 Supplementary Figure 1 Soluble and plated anti- Abs induce -! signalling. B3Z cells stably expressing!
The autoimmune disease-associated PTPN22 variant promotes calpain-mediated Lyp/Pep
 SUPPLEMENTARY INFORMATION The autoimmune disease-associated PTPN22 variant promotes calpain-mediated Lyp/Pep degradation associated with lymphocyte and dendritic cell hyperresponsiveness Jinyi Zhang, Naima
SUPPLEMENTARY INFORMATION The autoimmune disease-associated PTPN22 variant promotes calpain-mediated Lyp/Pep degradation associated with lymphocyte and dendritic cell hyperresponsiveness Jinyi Zhang, Naima
Cell isolation. Spleen and lymph nodes (axillary, inguinal) were removed from mice
 Supplementary Methods: Cell isolation. Spleen and lymph nodes (axillary, inguinal) were removed from mice and gently meshed in DMEM containing 10% FBS to prepare for single cell suspensions. CD4 + CD25
Supplementary Methods: Cell isolation. Spleen and lymph nodes (axillary, inguinal) were removed from mice and gently meshed in DMEM containing 10% FBS to prepare for single cell suspensions. CD4 + CD25
MTC-TT and TPC-1 cell lines were cultured in RPMI medium (Gibco, Breda, The Netherlands)
 Supplemental data Materials and Methods Cell culture MTC-TT and TPC-1 cell lines were cultured in RPMI medium (Gibco, Breda, The Netherlands) supplemented with 15% or 10% (for TPC-1) fetal bovine serum
Supplemental data Materials and Methods Cell culture MTC-TT and TPC-1 cell lines were cultured in RPMI medium (Gibco, Breda, The Netherlands) supplemented with 15% or 10% (for TPC-1) fetal bovine serum
Supporting Information Table of Contents
 Supporting Information Table of Contents Supporting Information Figure 1 Page 2 Supporting Information Figure 2 Page 4 Supporting Information Figure 3 Page 5 Supporting Information Figure 4 Page 6 Supporting
Supporting Information Table of Contents Supporting Information Figure 1 Page 2 Supporting Information Figure 2 Page 4 Supporting Information Figure 3 Page 5 Supporting Information Figure 4 Page 6 Supporting
Supplementary Figure 1
 Supplementary Figure 1 Identification of IFN-γ-producing CD8 + and CD4 + T cells with naive phenotype by alternative gating and sample-processing strategies. a. Contour 5% probability plots show definition
Supplementary Figure 1 Identification of IFN-γ-producing CD8 + and CD4 + T cells with naive phenotype by alternative gating and sample-processing strategies. a. Contour 5% probability plots show definition
Supplementary Fig. 1 p38 MAPK negatively regulates DC differentiation. (a) Western blot analysis of p38 isoform expression in BM cells, immature DCs
 Supplementary Fig. 1 p38 MAPK negatively regulates DC differentiation. (a) Western blot analysis of p38 isoform expression in BM cells, immature DCs (idcs) and mature DCs (mdcs). A myeloma cell line expressing
Supplementary Fig. 1 p38 MAPK negatively regulates DC differentiation. (a) Western blot analysis of p38 isoform expression in BM cells, immature DCs (idcs) and mature DCs (mdcs). A myeloma cell line expressing
Supplemental Figure 1. Signature gene expression in in vitro differentiated Th0, Th1, Th2, Th17 and Treg cells. (A) Naïve CD4 + T cells were cultured
 Supplemental Figure 1. Signature gene expression in in vitro differentiated Th0, Th1, Th2, Th17 and Treg cells. (A) Naïve CD4 + T cells were cultured under Th0, Th1, Th2, Th17, and Treg conditions. mrna
Supplemental Figure 1. Signature gene expression in in vitro differentiated Th0, Th1, Th2, Th17 and Treg cells. (A) Naïve CD4 + T cells were cultured under Th0, Th1, Th2, Th17, and Treg conditions. mrna
Suppl Video: Tumor cells (green) and monocytes (white) are seeded on a confluent endothelial
 Supplementary Information Häuselmann et al. Monocyte induction of E-selectin-mediated endothelial activation releases VE-cadherin junctions to promote tumor cell extravasation in the metastasis cascade
Supplementary Information Häuselmann et al. Monocyte induction of E-selectin-mediated endothelial activation releases VE-cadherin junctions to promote tumor cell extravasation in the metastasis cascade
Human Immunodeficiency Virus Type-1 Myeloid Derived Suppressor Cells Inhibit Cytomegalovirus Inflammation through Interleukin-27 and B7-H4
 Human Immunodeficiency Virus Type-1 Myeloid Derived Suppressor Cells Inhibit Cytomegalovirus Inflammation through Interleukin-27 and B7-H4 Ankita Garg, Rodney Trout and Stephen A. Spector,,* Department
Human Immunodeficiency Virus Type-1 Myeloid Derived Suppressor Cells Inhibit Cytomegalovirus Inflammation through Interleukin-27 and B7-H4 Ankita Garg, Rodney Trout and Stephen A. Spector,,* Department
% of live splenocytes. STAT5 deletion. (open shapes) % ROSA + % floxed
 Supp. Figure 1. a 14 1 1 8 6 spleen cells (x1 6 ) 16 % of live splenocytes 5 4 3 1 % of live splenocytes 8 6 4 b 1 1 c % of CD11c + splenocytes (closed shapes) 8 6 4 8 6 4 % ROSA + (open shapes) % floxed
Supp. Figure 1. a 14 1 1 8 6 spleen cells (x1 6 ) 16 % of live splenocytes 5 4 3 1 % of live splenocytes 8 6 4 b 1 1 c % of CD11c + splenocytes (closed shapes) 8 6 4 8 6 4 % ROSA + (open shapes) % floxed
In vitro human regulatory T cell expansion
 - 1 - Human CD4 + CD25 + regulatory T cell isolation, Workflow in vitro expansion and analysis In vitro human regulatory T cell expansion Introduction Regulatory T (Treg) cells are a subpopulation of T
- 1 - Human CD4 + CD25 + regulatory T cell isolation, Workflow in vitro expansion and analysis In vitro human regulatory T cell expansion Introduction Regulatory T (Treg) cells are a subpopulation of T
T H 1, T H 2 and T H 17 polarization of naïve CD4 + mouse T cells
 A complete workflow for cell preparation, isolation, polarization and analysis T H 1, T H 2 and T H 17 polarization of naïve CD4 + mouse T cells Introduction Workflow CD4 + T helper (T H) cells play a
A complete workflow for cell preparation, isolation, polarization and analysis T H 1, T H 2 and T H 17 polarization of naïve CD4 + mouse T cells Introduction Workflow CD4 + T helper (T H) cells play a
Supplementary Figures
 Inhibition of Pulmonary Anti Bacterial Defense by IFN γ During Recovery from Influenza Infection By Keer Sun and Dennis W. Metzger Supplementary Figures d a Ly6G Percentage survival f 1 75 5 1 25 1 5 1
Inhibition of Pulmonary Anti Bacterial Defense by IFN γ During Recovery from Influenza Infection By Keer Sun and Dennis W. Metzger Supplementary Figures d a Ly6G Percentage survival f 1 75 5 1 25 1 5 1
CD4 + T cells recovered in Rag2 / recipient ( 10 5 ) Heart Lung Pancreas
 a CD4 + T cells recovered in Rag2 / recipient ( 1 5 ) Heart Lung Pancreas.5 1 2 4 6 2 4 6 Ctla4 +/+ Ctla4 / Ctla4 / Lung Ctla4 / Pancreas b Heart Lung Pancreas Ctla4 +/+ Ctla4 / Ctla4 / Lung Ctla4 / Pancreas
a CD4 + T cells recovered in Rag2 / recipient ( 1 5 ) Heart Lung Pancreas.5 1 2 4 6 2 4 6 Ctla4 +/+ Ctla4 / Ctla4 / Lung Ctla4 / Pancreas b Heart Lung Pancreas Ctla4 +/+ Ctla4 / Ctla4 / Lung Ctla4 / Pancreas
Peli1 negatively regulates T-cell activation and prevents autoimmunity
 Peli1 negatively regulates T-cell activation and prevents autoimmunity Mikyoung Chang 1,*, Wei Jin 1,5,*, Jae-Hoon Chang 1, Yi-chuan Xiao 1, George Brittain 1, Jiayi Yu 1, Xiaofei Zhou 1, Yi-Hong Wang
Peli1 negatively regulates T-cell activation and prevents autoimmunity Mikyoung Chang 1,*, Wei Jin 1,5,*, Jae-Hoon Chang 1, Yi-chuan Xiao 1, George Brittain 1, Jiayi Yu 1, Xiaofei Zhou 1, Yi-Hong Wang
Reviewers' comments: Reviewer #1 Expert in EAE and IL-17a (Remarks to the Author):
 Reviewers' comments: Reviewer #1 Expert in EAE and IL-17a (Remarks to the Author): This study shows that the inducible camp early repressor (ICER) is involved in development of Th17 cells that are pathogenic
Reviewers' comments: Reviewer #1 Expert in EAE and IL-17a (Remarks to the Author): This study shows that the inducible camp early repressor (ICER) is involved in development of Th17 cells that are pathogenic
Supplementary Figures
 MiR-29 controls innate and adaptive immune responses against intracellular bacterial infection by targeting IFN-γ Feng Ma 1,2,5, Sheng Xu 1,5, Xingguang Liu 1, Qian Zhang 1, Xiongfei Xu 1, Mofang Liu 3,
MiR-29 controls innate and adaptive immune responses against intracellular bacterial infection by targeting IFN-γ Feng Ma 1,2,5, Sheng Xu 1,5, Xingguang Liu 1, Qian Zhang 1, Xiongfei Xu 1, Mofang Liu 3,
Supplementary Figure 1. IL-12 serum levels and frequency of subsets in FL patients. (A) IL-12
 1 Supplementary Data Figure legends Supplementary Figure 1. IL-12 serum levels and frequency of subsets in FL patients. (A) IL-12 serum levels measured by multiplex ELISA (Luminex) in FL patients before
1 Supplementary Data Figure legends Supplementary Figure 1. IL-12 serum levels and frequency of subsets in FL patients. (A) IL-12 serum levels measured by multiplex ELISA (Luminex) in FL patients before
Commercially available HLA Class II tetramers (Beckman Coulter) conjugated to
 Class II tetramer staining Commercially available HLA Class II tetramers (Beckman Coulter) conjugated to PE were combined with dominant HIV epitopes (DRB1*0101-DRFYKTLRAEQASQEV, DRB1*0301- PEKEVLVWKFDSRLAFHH,
Class II tetramer staining Commercially available HLA Class II tetramers (Beckman Coulter) conjugated to PE were combined with dominant HIV epitopes (DRB1*0101-DRFYKTLRAEQASQEV, DRB1*0301- PEKEVLVWKFDSRLAFHH,
The encephalitogenicity of TH17 cells is dependent on IL-1- and IL-23- induced production of the cytokine GM-CSF
 CORRECTION NOTICE Nat.Immunol. 12, 568 575 (2011) The encephalitogenicity of TH17 cells is dependent on IL-1- and IL-23- induced production of the cytokine GM-CSF Mohamed El-Behi, Bogoljub Ciric, Hong
CORRECTION NOTICE Nat.Immunol. 12, 568 575 (2011) The encephalitogenicity of TH17 cells is dependent on IL-1- and IL-23- induced production of the cytokine GM-CSF Mohamed El-Behi, Bogoljub Ciric, Hong
Supplementary Figure S1. Flow cytometric analysis of the expression of Thy1 in NH cells. Flow cytometric analysis of the expression of T1/ST2 and
 Supplementary Figure S1. Flow cytometric analysis of the expression of Thy1 in NH cells. Flow cytometric analysis of the expression of T1/ST2 and Thy1 in NH cells derived from the lungs of naïve mice.
Supplementary Figure S1. Flow cytometric analysis of the expression of Thy1 in NH cells. Flow cytometric analysis of the expression of T1/ST2 and Thy1 in NH cells derived from the lungs of naïve mice.
SUPPLEMENTARY INFORMATION
 doi: 1.138/nature89 IFN- (ng ml ) 5 4 3 1 Splenocytes NS IFN- (ng ml ) 6 4 Lymph node cells NS Nfkbiz / Nfkbiz / Nfkbiz / Nfkbiz / IL- (ng ml ) 3 1 Splenocytes IL- (ng ml ) 1 8 6 4 *** ** Lymph node cells
doi: 1.138/nature89 IFN- (ng ml ) 5 4 3 1 Splenocytes NS IFN- (ng ml ) 6 4 Lymph node cells NS Nfkbiz / Nfkbiz / Nfkbiz / Nfkbiz / IL- (ng ml ) 3 1 Splenocytes IL- (ng ml ) 1 8 6 4 *** ** Lymph node cells
In vitro human regulatory T cell expansion
 - 1 - Human CD4 + CD25 + CD127 dim/- regulatory T cell Workflow isolation, in vitro expansion and analysis In vitro human regulatory T cell expansion Introduction Regulatory T (Treg) cells are a subpopulation
- 1 - Human CD4 + CD25 + CD127 dim/- regulatory T cell Workflow isolation, in vitro expansion and analysis In vitro human regulatory T cell expansion Introduction Regulatory T (Treg) cells are a subpopulation
Interferon γ regulates idiopathic pneumonia syndrome, a. Th17 + CD4 + T-cell-mediated GvH disease
 Interferon γ regulates idiopathic pneumonia syndrome, a Th17 + CD4 + T-cell-mediated GvH disease Nora Mauermann, Julia Burian, Christophe von Garnier, Stefan Dirnhofer, Davide Germano, Christine Schuett,
Interferon γ regulates idiopathic pneumonia syndrome, a Th17 + CD4 + T-cell-mediated GvH disease Nora Mauermann, Julia Burian, Christophe von Garnier, Stefan Dirnhofer, Davide Germano, Christine Schuett,
Supplementary Data 1. Alanine substitutions and position variants of APNCYGNIPL. Applied in
 Supplementary Data 1. Alanine substitutions and position variants of APNCYGNIPL. Applied in Supplementary Fig. 2 Substitution Sequence Position variant Sequence original APNCYGNIPL original APNCYGNIPL
Supplementary Data 1. Alanine substitutions and position variants of APNCYGNIPL. Applied in Supplementary Fig. 2 Substitution Sequence Position variant Sequence original APNCYGNIPL original APNCYGNIPL
Supplementary Materials for
 www.sciencesignaling.org/cgi/content/full/3/114/ra23/dc1 Supplementary Materials for Regulation of Zap70 Expression During Thymocyte Development Enables Temporal Separation of CD4 and CD8 Repertoire Selection
www.sciencesignaling.org/cgi/content/full/3/114/ra23/dc1 Supplementary Materials for Regulation of Zap70 Expression During Thymocyte Development Enables Temporal Separation of CD4 and CD8 Repertoire Selection
W/T Itgam -/- F4/80 CD115. F4/80 hi CD115 + F4/80 + CD115 +
 F4/8 % in the peritoneal lavage 6 4 2 p=.15 n.s p=.76 CD115 F4/8 hi CD115 + F4/8 + CD115 + F4/8 hi CD115 + F4/8 + CD115 + MHCII MHCII Supplementary Figure S1. CD11b deficiency affects the cellular responses
F4/8 % in the peritoneal lavage 6 4 2 p=.15 n.s p=.76 CD115 F4/8 hi CD115 + F4/8 + CD115 + F4/8 hi CD115 + F4/8 + CD115 + MHCII MHCII Supplementary Figure S1. CD11b deficiency affects the cellular responses
sequences of a styx mutant reveals a T to A transversion in the donor splice site of intron 5
 sfigure 1 Styx mutant mice recapitulate the phenotype of SHIP -/- mice. (A) Analysis of the genomic sequences of a styx mutant reveals a T to A transversion in the donor splice site of intron 5 (GTAAC
sfigure 1 Styx mutant mice recapitulate the phenotype of SHIP -/- mice. (A) Analysis of the genomic sequences of a styx mutant reveals a T to A transversion in the donor splice site of intron 5 (GTAAC
Supplementary Figure 1: si-craf but not si-braf sensitizes tumor cells to radiation.
 Supplementary Figure 1: si-craf but not si-braf sensitizes tumor cells to radiation. (a) Embryonic fibroblasts isolated from wildtype (WT), BRAF -/-, or CRAF -/- mice were irradiated (6 Gy) and DNA damage
Supplementary Figure 1: si-craf but not si-braf sensitizes tumor cells to radiation. (a) Embryonic fibroblasts isolated from wildtype (WT), BRAF -/-, or CRAF -/- mice were irradiated (6 Gy) and DNA damage
BD CBA on the BD Accuri C6: Bringing Multiplexed Cytokine Detection to the Benchtop
 BD CBA on the BD Accuri C6: Bringing Multiplexed Cytokine Detection to the Benchtop Maria Dinkelmann, PhD Senior Marketing Applications Specialist BD Biosciences, Ann Arbor, MI 23-14380-00 Cellular Communication
BD CBA on the BD Accuri C6: Bringing Multiplexed Cytokine Detection to the Benchtop Maria Dinkelmann, PhD Senior Marketing Applications Specialist BD Biosciences, Ann Arbor, MI 23-14380-00 Cellular Communication
Bead Based Assays for Cytokine Detection
 Bead Based Assays for Cytokine Detection September 27, 2014 6 th EFIS-EJI South East European Immunology School SEEIS 2014 Timisoara, Romania The Cells of the Immune System The Immune Reaction (Th2) (Th1)
Bead Based Assays for Cytokine Detection September 27, 2014 6 th EFIS-EJI South East European Immunology School SEEIS 2014 Timisoara, Romania The Cells of the Immune System The Immune Reaction (Th2) (Th1)
Supporting Information
 Supporting Information Valkenburg et al. 10.1073/pnas.1403684111 SI Materials and Methods ELISA and Microneutralization. Sera were treated with Receptor Destroying Enzyme II (RDE II, Accurate) before ELISA
Supporting Information Valkenburg et al. 10.1073/pnas.1403684111 SI Materials and Methods ELISA and Microneutralization. Sera were treated with Receptor Destroying Enzyme II (RDE II, Accurate) before ELISA
Supplementary Data. Treg phenotype
 Supplementary Data Additional Experiment An additional experiment was performed using cryopreserved peripheral blood mononuclear cells (PBMC) derived from five renal cell carcinoma (RCC) patients [see
Supplementary Data Additional Experiment An additional experiment was performed using cryopreserved peripheral blood mononuclear cells (PBMC) derived from five renal cell carcinoma (RCC) patients [see
Scott Abrams, Ph.D. Professor of Oncology, x4375 Kuby Immunology SEVENTH EDITION
 Scott Abrams, Ph.D. Professor of Oncology, x4375 scott.abrams@roswellpark.org Kuby Immunology SEVENTH EDITION CHAPTER 11 T-Cell Activation, Differentiation, and Memory Copyright 2013 by W. H. Freeman and
Scott Abrams, Ph.D. Professor of Oncology, x4375 scott.abrams@roswellpark.org Kuby Immunology SEVENTH EDITION CHAPTER 11 T-Cell Activation, Differentiation, and Memory Copyright 2013 by W. H. Freeman and
Title of file for HTML: Supplementary Information Description: Supplementary Figures and Supplementary Table
 Title of file for HTML: Supplementary Information Description: Supplementary Figures and Supplementary Table Title of file for HTML: Peer Review File Description: Innate Scavenger Receptor-A regulates
Title of file for HTML: Supplementary Information Description: Supplementary Figures and Supplementary Table Title of file for HTML: Peer Review File Description: Innate Scavenger Receptor-A regulates
<10. IL-1β IL-6 TNF + _ TGF-β + IL-23
 3 ns 25 ns 2 IL-17 (pg/ml) 15 1 ns ns 5 IL-1β IL-6 TNF
3 ns 25 ns 2 IL-17 (pg/ml) 15 1 ns ns 5 IL-1β IL-6 TNF
Intracellular MHC class II molecules promote TLR-triggered innate. immune responses by maintaining Btk activation
 Intracellular MHC class II molecules promote TLR-triggered innate immune responses by maintaining Btk activation Xingguang Liu, Zhenzhen Zhan, Dong Li, Li Xu, Feng Ma, Peng Zhang, Hangping Yao and Xuetao
Intracellular MHC class II molecules promote TLR-triggered innate immune responses by maintaining Btk activation Xingguang Liu, Zhenzhen Zhan, Dong Li, Li Xu, Feng Ma, Peng Zhang, Hangping Yao and Xuetao
Ex vivo Human Antigen-specific T Cell Proliferation and Degranulation Willemijn Hobo 1, Wieger Norde 1 and Harry Dolstra 2*
 Ex vivo Human Antigen-specific T Cell Proliferation and Degranulation Willemijn Hobo 1, Wieger Norde 1 and Harry Dolstra 2* 1 Department of Laboratory Medicine - Laboratory of Hematology, Radboud University
Ex vivo Human Antigen-specific T Cell Proliferation and Degranulation Willemijn Hobo 1, Wieger Norde 1 and Harry Dolstra 2* 1 Department of Laboratory Medicine - Laboratory of Hematology, Radboud University
Islet viability assay and Glucose Stimulated Insulin Secretion assay RT-PCR and Western Blot
 Islet viability assay and Glucose Stimulated Insulin Secretion assay Islet cell viability was determined by colorimetric (3-(4,5-dimethylthiazol-2-yl)-2,5- diphenyltetrazolium bromide assay using CellTiter
Islet viability assay and Glucose Stimulated Insulin Secretion assay Islet cell viability was determined by colorimetric (3-(4,5-dimethylthiazol-2-yl)-2,5- diphenyltetrazolium bromide assay using CellTiter
Nature Immunology: doi: /ni Supplementary Figure 1. Huwe1 has high expression in HSCs and is necessary for quiescence.
 Supplementary Figure 1 Huwe1 has high expression in HSCs and is necessary for quiescence. (a) Heat map visualizing expression of genes with a known function in ubiquitin-mediated proteolysis (KEGG: Ubiquitin
Supplementary Figure 1 Huwe1 has high expression in HSCs and is necessary for quiescence. (a) Heat map visualizing expression of genes with a known function in ubiquitin-mediated proteolysis (KEGG: Ubiquitin
Figure S1. Generation of inducible PTEN deficient mice and the BMMCs (A) B6.129 Pten loxp/loxp mice were mated with B6.
 Figure S1. Generation of inducible PTEN deficient mice and the BMMCs (A) B6.129 Pten loxp/loxp mice were mated with B6.129-Gt(ROSA)26Sor tm1(cre/ert2)tyj /J mice. To induce deletion of the Pten locus,
Figure S1. Generation of inducible PTEN deficient mice and the BMMCs (A) B6.129 Pten loxp/loxp mice were mated with B6.129-Gt(ROSA)26Sor tm1(cre/ert2)tyj /J mice. To induce deletion of the Pten locus,
The toll-like receptor 4 ligands Mrp8 and Mrp14 play a critical role in the development of autoreactive CD8 + T cells
 1 SUPPLEMENTARY INFORMATION The toll-like receptor 4 ligands Mrp8 and Mrp14 play a critical role in the development of autoreactive CD8 + T cells Karin Loser 1,2,6, Thomas Vogl 2,3, Maik Voskort 1, Aloys
1 SUPPLEMENTARY INFORMATION The toll-like receptor 4 ligands Mrp8 and Mrp14 play a critical role in the development of autoreactive CD8 + T cells Karin Loser 1,2,6, Thomas Vogl 2,3, Maik Voskort 1, Aloys
of whole cell cultures in U-bottomed wells of a 96-well plate are shown. 2
 Supplementary online material Supplementary figure legends Supplementary Figure 1 Exposure to T reg cells causes loss of T resp cells in co-cultures. T resp cells were stimulated with CD3+CD28 alone or
Supplementary online material Supplementary figure legends Supplementary Figure 1 Exposure to T reg cells causes loss of T resp cells in co-cultures. T resp cells were stimulated with CD3+CD28 alone or
SUPPLEMENTARY MATERIAL
 SUPPLEMENTARY MATERIAL IL-1 signaling modulates activation of STAT transcription factors to antagonize retinoic acid signaling and control the T H 17 cell it reg cell balance Rajatava Basu 1,5, Sarah K.
SUPPLEMENTARY MATERIAL IL-1 signaling modulates activation of STAT transcription factors to antagonize retinoic acid signaling and control the T H 17 cell it reg cell balance Rajatava Basu 1,5, Sarah K.
Sipper BK Experimental Animal Co. (Shanghai, China) and bred in a specific. pathogen-free environment. The animal study protocol was approved by the
 Supplementary information, Data S1 Materials and Methods Mice, Ad vectors and reagents Female C57BL/6 mice, 8-10 weeks of age, were purchased from Joint Ventures Sipper BK Experimental Animal Co. (Shanghai,
Supplementary information, Data S1 Materials and Methods Mice, Ad vectors and reagents Female C57BL/6 mice, 8-10 weeks of age, were purchased from Joint Ventures Sipper BK Experimental Animal Co. (Shanghai,
Supplemental Methods In vitro T cell assays Inhibition of perforin- and FasL-mediated cytotoxicity Flow Cytometry
 Supplemental Methods In vitro T cell assays Cell lines Jurkat (ATCC #TIB-152), CCRF-CEM (ATCC #CCL-119), MOLT-4 (ATCC #CRL- 1582), Hut 78 (ATCC #TIB-161), SupT1 (ATCC #CRL-1942), Raji (ATCC #CCL-86) and
Supplemental Methods In vitro T cell assays Cell lines Jurkat (ATCC #TIB-152), CCRF-CEM (ATCC #CCL-119), MOLT-4 (ATCC #CRL- 1582), Hut 78 (ATCC #TIB-161), SupT1 (ATCC #CRL-1942), Raji (ATCC #CCL-86) and
(a) Significant biological processes (upper panel) and disease biomarkers (lower panel)
 Supplementary Figure 1. Functional enrichment analyses of secretomic proteins. (a) Significant biological processes (upper panel) and disease biomarkers (lower panel) 2 involved by hrab37-mediated secretory
Supplementary Figure 1. Functional enrichment analyses of secretomic proteins. (a) Significant biological processes (upper panel) and disease biomarkers (lower panel) 2 involved by hrab37-mediated secretory
Supplemental Figure 1 ELISA scheme to measure plasma total, mature and furin-cleaved
 1 Supplemental Figure Legends Supplemental Figure 1 ELISA scheme to measure plasma total, mature and furin-cleaved PCSK9 concentrations. 4 Plasma mature and furin-cleaved PCSK9s were measured by a sandwich
1 Supplemental Figure Legends Supplemental Figure 1 ELISA scheme to measure plasma total, mature and furin-cleaved PCSK9 concentrations. 4 Plasma mature and furin-cleaved PCSK9s were measured by a sandwich
Hepatitis B Antiviral Drug Development Multi-Marker Screening Assay
 Hepatitis B Antiviral Drug Development Multi-Marker Screening Assay Background ImQuest BioSciences has developed and qualified a single-plate method to expedite the screening of antiviral agents against
Hepatitis B Antiviral Drug Development Multi-Marker Screening Assay Background ImQuest BioSciences has developed and qualified a single-plate method to expedite the screening of antiviral agents against
MicroRNA sponges: competitive inhibitors of small RNAs in mammalian cells
 MicroRNA sponges: competitive inhibitors of small RNAs in mammalian cells Margaret S Ebert, Joel R Neilson & Phillip A Sharp Supplementary figures and text: Supplementary Figure 1. Effect of sponges on
MicroRNA sponges: competitive inhibitors of small RNAs in mammalian cells Margaret S Ebert, Joel R Neilson & Phillip A Sharp Supplementary figures and text: Supplementary Figure 1. Effect of sponges on
Peer review correspondence
 Highly polarized Th17 cells induce EAE via a T-bet-independent mechanism Heather M. Grifka-Walk, Stephen J. Lalor and Benjamin M. Segal Corresponding author: Benjamin Segal, Holtom-Garrett Program in Neuroimmunology
Highly polarized Th17 cells induce EAE via a T-bet-independent mechanism Heather M. Grifka-Walk, Stephen J. Lalor and Benjamin M. Segal Corresponding author: Benjamin Segal, Holtom-Garrett Program in Neuroimmunology
CONTRACTING ORGANIZATION: Johns Hopkins University School of Medicine Baltimore, MD 21205
 AD Award Number: DAMD7---7 TITLE: Development of Artificial Antigen Presenting Cells for Prostate Cancer Immunotherapy PRINCIPAL INVESTIGATOR: Jonathan P. Schneck, M.D., Ph.D. Mathias Oelke, Ph.D. CONTRACTING
AD Award Number: DAMD7---7 TITLE: Development of Artificial Antigen Presenting Cells for Prostate Cancer Immunotherapy PRINCIPAL INVESTIGATOR: Jonathan P. Schneck, M.D., Ph.D. Mathias Oelke, Ph.D. CONTRACTING
well for 2 h at rt. Each dot represents an individual mouse and bar is the mean ±
 Supplementary data: Control DC Blimp-1 ko DC 8 6 4 2-2 IL-1β p=.5 medium 8 6 4 2 IL-2 Medium p=.16 8 6 4 2 IL-6 medium p=.3 5 4 3 2 1-1 medium IL-1 n.s. 25 2 15 1 5 IL-12(p7) p=.15 5 IFNγ p=.65 4 3 2 1
Supplementary data: Control DC Blimp-1 ko DC 8 6 4 2-2 IL-1β p=.5 medium 8 6 4 2 IL-2 Medium p=.16 8 6 4 2 IL-6 medium p=.3 5 4 3 2 1-1 medium IL-1 n.s. 25 2 15 1 5 IL-12(p7) p=.15 5 IFNγ p=.65 4 3 2 1
Therapeutic PD L1 and LAG 3 blockade rapidly clears established blood stage Plasmodium infection
 Supplementary Information Therapeutic PD L1 and LAG 3 blockade rapidly clears established blood stage Plasmodium infection Noah S. Butler, Jacqueline Moebius, Lecia L. Pewe, Boubacar Traore, Ogobara K.
Supplementary Information Therapeutic PD L1 and LAG 3 blockade rapidly clears established blood stage Plasmodium infection Noah S. Butler, Jacqueline Moebius, Lecia L. Pewe, Boubacar Traore, Ogobara K.
Supporting Online Material for
 www.sciencemag.org/cgi/content/full/1175194/dc1 Supporting Online Material for A Vital Role for Interleukin-21 in the Control of a Chronic Viral Infection John S. Yi, Ming Du, Allan J. Zajac* *To whom
www.sciencemag.org/cgi/content/full/1175194/dc1 Supporting Online Material for A Vital Role for Interleukin-21 in the Control of a Chronic Viral Infection John S. Yi, Ming Du, Allan J. Zajac* *To whom
Electron micrograph of phosphotungstanic acid-stained exosomes derived from murine
 1 SUPPLEMENTARY INFORMATION SUPPLEMENTARY FIGURES Supplementary Figure 1. Physical properties of murine DC-derived exosomes. a, Electron micrograph of phosphotungstanic acid-stained exosomes derived from
1 SUPPLEMENTARY INFORMATION SUPPLEMENTARY FIGURES Supplementary Figure 1. Physical properties of murine DC-derived exosomes. a, Electron micrograph of phosphotungstanic acid-stained exosomes derived from
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION FOR Liver X Receptor α mediates hepatic triglyceride accumulation through upregulation of G0/G1 Switch Gene 2 (G0S2) expression I: SUPPLEMENTARY METHODS II: SUPPLEMENTARY FIGURES
SUPPLEMENTARY INFORMATION FOR Liver X Receptor α mediates hepatic triglyceride accumulation through upregulation of G0/G1 Switch Gene 2 (G0S2) expression I: SUPPLEMENTARY METHODS II: SUPPLEMENTARY FIGURES
Supplementary Figure S1. PTPN2 levels are not altered in proliferating CD8+ T cells. Lymph node (LN) CD8+ T cells from C57BL/6 mice were stained with
 Supplementary Figure S1. PTPN2 levels are not altered in proliferating CD8+ T cells. Lymph node (LN) CD8+ T cells from C57BL/6 mice were stained with CFSE and stimulated with plate-bound α-cd3ε (10µg/ml)
Supplementary Figure S1. PTPN2 levels are not altered in proliferating CD8+ T cells. Lymph node (LN) CD8+ T cells from C57BL/6 mice were stained with CFSE and stimulated with plate-bound α-cd3ε (10µg/ml)
Supplementary Figures
 Supplementary Figures Supplementary Figure 1 Characterization of stable expression of GlucB and sshbira in the CT26 cell line (a) Live cell imaging of stable CT26 cells expressing green fluorescent protein
Supplementary Figures Supplementary Figure 1 Characterization of stable expression of GlucB and sshbira in the CT26 cell line (a) Live cell imaging of stable CT26 cells expressing green fluorescent protein
Increased IL-12 induced STAT-4 signaling in CD8 T cells. from aged mice
 Increased IL-2 induced STAT-4 signaling in CD8 T cells from aged mice Erin Rottinghaus * Abstract: Aging is associated with poor immune function leading to increased susceptibility to infectious diseases
Increased IL-2 induced STAT-4 signaling in CD8 T cells from aged mice Erin Rottinghaus * Abstract: Aging is associated with poor immune function leading to increased susceptibility to infectious diseases
D CD8 T cell number (x10 6 )
 IFNγ Supplemental Figure 1. CD T cell number (x1 6 ) 18 15 1 9 6 3 CD CD T cells CD6L C CD5 CD T cells CD6L D CD8 T cell number (x1 6 ) 1 8 6 E CD CD8 T cells CD6L F Log(1)CFU/g Feces 1 8 6 p
IFNγ Supplemental Figure 1. CD T cell number (x1 6 ) 18 15 1 9 6 3 CD CD T cells CD6L C CD5 CD T cells CD6L D CD8 T cell number (x1 6 ) 1 8 6 E CD CD8 T cells CD6L F Log(1)CFU/g Feces 1 8 6 p
Supplementary Materials and Methods
 Supplementary Materials and Methods Immunoblotting Immunoblot analysis was performed as described previously (1). Due to high-molecular weight of MUC4 (~ 950 kda) and MUC1 (~ 250 kda) proteins, electrophoresis
Supplementary Materials and Methods Immunoblotting Immunoblot analysis was performed as described previously (1). Due to high-molecular weight of MUC4 (~ 950 kda) and MUC1 (~ 250 kda) proteins, electrophoresis
and follicular helper T cells is Egr2-dependent. (a) Diagrammatic representation of the
 Supplementary Figure 1. LAG3 + Treg-mediated regulation of germinal center B cells and follicular helper T cells is Egr2-dependent. (a) Diagrammatic representation of the experimental protocol for the
Supplementary Figure 1. LAG3 + Treg-mediated regulation of germinal center B cells and follicular helper T cells is Egr2-dependent. (a) Diagrammatic representation of the experimental protocol for the
SUPPORTING MATREALS. Methods and Materials
 SUPPORTING MATREALS Methods and Materials Cell Culture MC3T3-E1 (subclone 4) cells were maintained in -MEM with 10% FBS, 1% Pen/Strep at 37ºC in a humidified incubator with 5% CO2. MC3T3 cell differentiation
SUPPORTING MATREALS Methods and Materials Cell Culture MC3T3-E1 (subclone 4) cells were maintained in -MEM with 10% FBS, 1% Pen/Strep at 37ºC in a humidified incubator with 5% CO2. MC3T3 cell differentiation
Supporting Information
 Supporting Information Pang et al. 10.1073/pnas.1322009111 SI Materials and Methods ELISAs. These assays were performed as previously described (1). ELISA plates (MaxiSorp Nunc; Thermo Fisher Scientific)
Supporting Information Pang et al. 10.1073/pnas.1322009111 SI Materials and Methods ELISAs. These assays were performed as previously described (1). ELISA plates (MaxiSorp Nunc; Thermo Fisher Scientific)
Supplementary Appendix
 Supplementary Appendix This appendix has been provided by the authors to give readers additional information about their work. Supplement to: Nair S, Branagan AR, Liu J, Boddupalli CS, Mistry PK, Dhodapkar
Supplementary Appendix This appendix has been provided by the authors to give readers additional information about their work. Supplement to: Nair S, Branagan AR, Liu J, Boddupalli CS, Mistry PK, Dhodapkar
Supplementary Figure 1. ETBF activate Stat3 in B6 and Min mice colons
 Supplementary Figure 1 ETBF activate Stat3 in B6 and Min mice colons a pstat3 controls Pos Neg ETBF 1 2 3 4 b pstat1 pstat2 pstat3 pstat4 pstat5 pstat6 Actin Figure Legend: (a) ETBF induce predominantly
Supplementary Figure 1 ETBF activate Stat3 in B6 and Min mice colons a pstat3 controls Pos Neg ETBF 1 2 3 4 b pstat1 pstat2 pstat3 pstat4 pstat5 pstat6 Actin Figure Legend: (a) ETBF induce predominantly
SUPPLEMENTARY INFORMATION. Supp. Fig. 1. Autoimmunity. Tolerance APC APC. T cell. T cell. doi: /nature06253 ICOS ICOS TCR CD28 TCR CD28
 Supp. Fig. 1 a APC b APC ICOS ICOS TCR CD28 mir P TCR CD28 P T cell Tolerance Roquin WT SG Icos mrna T cell Autoimmunity Roquin M199R SG Icos mrna www.nature.com/nature 1 Supp. Fig. 2 CD4 + CD44 low CD4
Supp. Fig. 1 a APC b APC ICOS ICOS TCR CD28 mir P TCR CD28 P T cell Tolerance Roquin WT SG Icos mrna T cell Autoimmunity Roquin M199R SG Icos mrna www.nature.com/nature 1 Supp. Fig. 2 CD4 + CD44 low CD4
B6/COLODR/SPL/11C/83/LAP/#2.006 B6/COLODR/SPL/11C/86/LAP/#2.016 CD11C B6/COLODR/SPL/11C/80/LAP/#2.011 CD11C
 CD3-specific antibody-induced immune tolerance and suppression of autoimmune encephalomyelitis involves TGF-β production through phagocytes digesting apoptotic T cells Sylvain Perruche 1,3, Pin Zhang 1,
CD3-specific antibody-induced immune tolerance and suppression of autoimmune encephalomyelitis involves TGF-β production through phagocytes digesting apoptotic T cells Sylvain Perruche 1,3, Pin Zhang 1,
