Cryptosporidium Oocysts in Fecal Specimens
|
|
|
- Chad Poole
- 5 years ago
- Views:
Transcription
1 JOURNAL OF CLINICAL MICROBIOLOGY, Nov. 1992, p /92/ $02.00/0 Copyright 1992, American Society for Microbiology Vol. 30, No. 11 Improved Stool Concentration Procedure for Detection of Cryptosporidium Oocysts in Fecal Specimens RAINER WEBER,t RALPH T. BRYAN,* AND DENNIS D. JURANEK Parasitic Diseases Branch, Division of Parasitic Diseases, National Center for Infectious Diseases, Centers for Disease Control, Atlanta, Georgia Received 18 March 1992/Accepted 25 August 1992 Epidemiologic and laboratory data suggest that coprodiagnostic methods may fail to detect Cryptosporidium oocysts in stool specimens of infected patients. To improve the efficacy of stool concentration procedures, we modified different steps of the Formalin-ethyl acetate (FEA) stool concentration technique and evaluated these modifications by examining stool samples seeded with known numbers of Cryptosporidium oocysts. Because these modifications failed to improve oocyst detection, we developed a new stool concentration technique that includes FEA sedimentation followed by layering and flotation over hypertonic sodium chloride solution to separate parasites from stool debris. Compared with the standard FEA procedure, this technique improved Cryptosporidium oocyst detection. The sensitivities of the two concentration techniques were similar for diarrheal (watery) stool specimens (100%/ of watery stool specimens seeded with 5,000 oocysts per g of stool were identified as positive by the new technique, compared with 901% of stools processed by the standard FEA technique). However, the most significant improvement in diagnosis occurred with formed stool specimens that were not fatty; 70 to 90% of formed stool specimens seeded with 5,000 oocysts were identified as positive by the new technique, compared with 01% of specimens processed by the standard FEA technique. One hundred percent of formed specimens seeded with 10,000 oocysts were correctly diagnosed by using the new technique, while 0 to 60% of specimens processed by the standard FEA technique were found positive. Similarly, only 50 to 90N% of stool specimens seeded with 50,000 oocysts were identified as positive by using the standard FEA technique, compared with a 100%o positive rate by the new technique. The new stool concentration procedure provides enhanced detection of Cryptosporidium oocysts in all stool samples. The coccidian parasite Cryptospoiidium sp. first gained attention as an etiologic agent of human diarrhea when it was identified in patients infected with the human immunodeficiency virus. Subsequently, this organism has also been recognized as an important cause of diarrhea in immunocompetent persons (1, 7, 8, 13-18, 20, 26, 29-31, 35). Various stool concentration techniques, staining methods, and antigen detection assays for the coprodiagnosis of cryptosporidiosis have been developed (2, 4, 9, 10, 20, 23, 27, 30, 32, 38). The Formalin-ethyl acetate (FEA) procedure to concentrate stool specimens and the modified cold Kinyoun acid-fast (AF) and the fluorescein-tagged monoclonal antibody (immunofluorescence [IF]) techniques to stain Cryptosporidium oocysts are among the most popular methods currently in use in diagnostic laboratories. Recent epidemiologic data suggest that coprodiagnostic methods may fail to detect Cryptosporidium oocysts in stool specimens of infected patients: in two studies of human immunodeficiency virus-infected patients with chronic diarrhea and negative stool examinations, Cryptosporidium oocysts were detected in 4.5 and 11.6% of patients undergoing small-bowel biopsy (12, 19). Similar research with immunocompetent patients without diarrhea has shown that stool examinations for Cryptosporidium oocysts may be negative in up to 53% of patients whose duodenal aspirates are positive for oocysts (26). By examining human stool samples seeded with known numbers of Cryptosporidium oocysts, we have previously * Corresponding author. t Present address: Division of Infectious Diseases, Department of Medicine, University Hospital, CH-8091, Zurich, Switzerland shown that the minimum number of oocysts that can be detected by using FEA stool concentration with AF- or IF-staining techniques is unexpectedly high. In addition, by analyzing different steps of the FEA stool concentration technique, we found that rather than actually concentrating oocysts, this procedure resulted in a net loss of detectable oocysts (34). To develop a stool concentration procedure that would improve the sensitivity of Cryptosporidium oocyst detection in stool specimens, we modified different steps of the FEA procedure and evaluated these modifications by examining stool samples seeded with known numbers of Cryptosporidium oocysts. These efforts led to the development of a new stool concentration technique, which we then compared with the standard FEA procedure for efficacy of oocyst recovery. MATERIALS AND METHODS Cryptosporidium oocysts. Viable Cryptosporidium parvum oocysts were recovered from fecal material of calves (3) and fixed with 10% Formalin. The number of oocysts in the Formalin stock solution was determined by using a cellcounting chamber (Spencer Bright Line hemocytometer; American Optical Co., Buffalo, N.Y.). All human stool specimens were seeded with 0.5 ml of the oocyst suspension. The number of Cryptosporidium oocysts in the dosage suspension was prepared by adjusting the stock solution by adding or removing Formalin. Human stool specimens. Feces-Formalin suspensions that were free of ova and parasites were prepared by fixing fresh fecal material with 10% Formalin in a 1:3 stool-formalin ratio. For experiments attempting to improve the FEA
2 2870 WEBER ET AL. concentration procedure, specimens from one volunteer with formed stools were used. For experiments evaluating the new stool concentration technique, samples (150 g each) of fresh fecal material from four volunteers with formed stools and of one fresh watery diarrheal specimen from a patient with AIDS were separately prepared. Portions of 20-ml stool-formalin suspensions containing 5 g of fecal material were placed in stool collection vials and individually seeded with a 0.5-ml suspension containing the number of Cryptosponidium oocysts necessary to yield the following concentrations: 5,000, 10,000, 50,000, and 1,000,000 oocysts per g of stool. Standard stool concentration procedures. The modified Ritchie FEA stool concentration procedure was performed as previously described (25, 36). Briefly, 4 ml of the Formalin-fixed stool suspension was washed with water through wet gauze into 15-ml conical centrifuge tubes. The sediment, which was collected by centrifugation at 500 x g for 2 min, was resuspended in 9 ml of 10% Formalin and 3 ml of ethyl acetate and shaken vigorously for 30 s. The second centrifugation step at 500 x g for 2 min resulted in the following four layers (from top to bottom): ethyl acetate, plug of debris, Formalin, and sediment. The top three layers were decanted, and slides were prepared by using 10-,ul aliquots of the sediment. Sheather's sugar and zinc sulfate centrifugal flotation techniques were also performed as previously described (22, 28) Ṁodification of the FEA stool concentration procedure. We evaluated the following modifications of the FEA stool concentration procedure. (i) Variable centrifugation times. The first and second centrifugation steps were varied between 1 and 10 min and compared with the standard centrifugation time of 2 min. (ii) Single centrifugation step with variation in centrifugation times. Only one centrifugation step was performed. Four milliliters of the Formalin-fixed stool suspension was washed with approximately 6 ml of 10% Formalin through wet gauze into 15-ml conical centrifuge tubes, mixed with 3 ml of ethyl acetate by shaking for 30 s, and centrifuged at 500 x g. Centrifugation times of 2, 5, and 10 min were compared. (iii) Nongauze filters. We used a 600-,um metallic filter and a commercially available plastic filter (fecal concentrator kit; Remel, Lenexa, Kans.) instead of wet gauze in the crude filtration of the stool suspension. (iv) Use of a surfactant. Three drops of 20% Triton X-100 were added to the centrifugation tube and mixed with the Formalin-fixed stool suspension before the first centrifugation step in an attempt to reduce the adherence of stool particles. (v) Variable g forces. Centrifugation at the relative centrifugal force of 100 x g or 1,000 x g was compared with standard centrifugation at 500 x g. Slides were prepared by using 10-,ul aliquots of the resulting sediments. Cryptosporidium oocysts were counted in 50 random microscopic fields at a magnification of x400. Three different specimens seeded with 1,000,000 oocysts per g of stool were examined for each experiment. The same specimens, processed by the standard FEA concentration method, served as controls. New stool concentration method (Fig. 1). Four milliliters of the Formalin-fixed stool suspension, 6 ml of 10% Formalin, and 3 ml of ethyl acetate were placed into a 15-ml conical glass centrifuge tube, mixed by shaking vigorously for 30 s, and centrifuged at 500 x g for 5 min. This centrifugation resulted in the following four layers (from top to bottom): FIG. 1. Flow diagram of the new stool concentration method. ethyl acetate, plug of debris, Formalin, and sediment. The top three layers were decanted. The sediment was resuspended in 5 ml of deionized water, layered over 5 ml of saturated sodium chloride (specific gravity, 1.20) by using a disposable plastic bulb pipette, and centrifuged at 500 x g for 10 min. This second centrifugation step resulted in the following three layers (from top to bottom): deionized water containing a small amount of fecal debris and Cryptosporidium oocysts, which were just above the surface of the sodium chloride layer; saturated sodium chloride; and a pellet containing most of the fecal debris. The uppermost 3.5 to 4 ml of the top layer was removed by using a disposable plastic bulb pipette and discarded. The remainder of the top layer (containing the parasites) and approximately 0.5 ml of the top portion of the sodium chloride layer were removed with the same pipette and washed in approximately 13 ml of deionized water by centrifugation at 500 x g for 10 min. Slides were prepared by using 10-,ul aliquots of the resulting sediment. Ten samples per oocyst concentration per person were processed by the new stool concentration procedure and by the standard FEA technique. Slides were stained and completely scanned by using fluorescent microscopy at a magnification of x400. Staining of slides. All slides were stained by using the indirect immunofluorescence detection procedure (Merifluor Cryptosporidium; Meridian Diagnostics, Cincinnati, Ohio) (4, 9, 27) according to the instructions of the manufacturer. RESULTS J. CLIN. MICROBIOL. Modifications of the FEA stool concentration procedure. Modifications of the FEA concentration technique offered no improvement in Cryptosporidium oocyst recovery compared with that by the standard FEA procedure. Increasing the centrifugation time did not result in higher numbers of detected oocysts (Table 1) and made slides more difficult to read than those prepared from sediments obtained by shorter centrifugation times. Slides prepared from specimens centrifuged for longer times had fecal debris that was more compact than that of control slides; the compact nature of the material on the slide resulted in increased background fluorescence, which reduced contrast between the applegreen fluorescence of Cryptosporidium oocysts and fecal debris. The single-step centrifugation procedure was significantly
3 VOL. 30, 1992 TABLE 1. Comparison of different centrifugation times in FEA stool concentration procedure Centrifugation time (min) Mean no. (range) of Step 1 Step 2 detected oocystsa (58-86) 2C ND 31 (24-40) 5 ND 29 (25-31) 10 ND 32 (21-52) (26-31) (45-59) (59-86) (53-74) (39-48) a Oocysts were counted in 50 random microscopic fields at a magnification of x400. Three specimens seeded with 1,000,000 Cryptosporidiwn oocysts per g of stool were examined for each combination of centrifugation times. b The standard FEA technique was used. The single centrifugation stool concentration procedure as described in Materials and Methods was used. STOOL CONCENTRATION PROCEDURES 2871 less efficacious than the standard two-step FEA technique (P, <0.001). Neither using metallic or plastic filters instead of wet gauze to filter stool suspensions nor adding a surfactant to the stool suspension improved oocyst recovery. Centrifugation at a relative centrifugal force of 100 x g led to less concentration of oocysts, and centrifugation at 1,000 x g resulted in oocyst detection similar to that obtained with standard centrifugation at 500 x g. However, increasing the relative centrifugal force adversely affected the reading of slides in a manner similar to that of increasing the centrifugation times; compaction of fecal elements by high-speed centrifugation resulted in more background fluorescence. Flotation methods. Using Sheather's sugar or zinc sulfate centrifugal flotation techniques, we detected no oocysts in 10 specimens of formed stool seeded with 50,000 Cryptosporidium oocysts per g of stool, whereas the standard FEA procedure showed 9 oocyst-positive slides of 10. New stool concentration method. Cryptosporidium oocyst detection was improved by our new stool concentration method compared with the FEA procedure (Tables 2 and 3). The threshold of detection of Cryptosporidium oocysts differed from one specimen to the next because stool specimens from different volunteers had different characteristics. Scanning smears from sediments obtained by the new stool concentration technique, we observed a substantially better separation of fecal debris and parasites than we obtained with the standard FEA technique. The slides had considerably less fecal debris and background fluorescence. Fat in the stool reduced recovery of oocysts by both the new technique and the standard FEA technique; nevertheless, the new technique gave better results than the standard FEA technique with the fatty stool. DISCUSSION We sought to improve stool concentration procedures on the basis of our previous finding that the threshold of detection of Cryptosporidium oocysts (i.e., the minimum number of oocysts that can be detected in stool specimens) with the FEA concentration technique and AF- and IFstaining methods is unexpectedly high (34). Others also have reported that the standard stool concentration method results in the loss of large numbers of oocysts (5, 6). In addition, epidemiologic studies reporting the presence of Cryptospondium oocysts in small-bowel biopsies or duodenal aspirates of patients whose stool examinations were negative indicated that new or modified coprodiagnostic techniques were needed (12, 19). On the basis of coprodiagnostic experiences with Giardia spp., one might expect that nonmicroscopic methods for detecting Cryptosporidium antigen in stool specimens would be more sensitive than traditional microscopic techniques for oocyst detection. However, recent reports indicate that available Cryptosporidium antigen detection methods are not more sensitive than the combination of FEA processing and microscopy for oocyst detection (2, 32). Anusz et al. found that the monoclonal antibody capture enzyme-linked immunosorbent assay was less sensitive than the IF procedure for detecting Cryptosporidium oocysts (2). Similarly, the indirect, double-antibody enzyme-linked immunosorbent assay described by Ungar (32) failed to detect Cryptosporidium antigen in all stool specimens in which oocysts were detected by light microscopy. Until the sensitivity of antigen detection methods improves, better stool concentration methods may provide a useful adjunct to existing techniques for the diagnosis of cryptosporidiosis. On the basis of the results of others who have found the IF detection procedure to have better sensitivity than the AF-staining procedure in epidemiologic studies (4, 9, 27), and on the basis of our laboratory findings showing that the yield by the AF-staining technique was significantly worse than that by the IF technique (34), we exclusively used the IF technique to evaluate the stool concentration methods. Because our analysis of the FEA stool concentration technique showed high numbers of Cryptosponidium oocysts in the discarded elements of the FEA procedure (i.e., in the TABLE 2. Comparison between new stool concentration technique and standard FEA stool concentration procedure No. of specimens positive/no. examineda cooncynbt Stool 1 (formed) Stool 2 (formed) Stool 3 (formed) Stool 4S (watery) concys (formed/fatty) Sol5(aey FEAC Newd FEA New FEA New FEA New FEA New 1,000 0/10 0/10 0/10 0/10 0/10 0/10 0/10 0/10 0/10 0/10 5,000 0/10 9/10 0/10 8/10 0/10 7/10 0/10 0/10 9/10 10/10 10,000 6/10 10/10 0/10 10/10 4/10 10/10 0/10 3/10 10/10 10/10 50,000 9/10 10/10 5/10 10/10 6/10 10/10 2/10 7/10 10/10 10/10 a Slides were scanned completely. b Number of oocysts per gram of stool. c FEA, FEA stool concentration procedure. d New, new stool concentration procedure.
4 2872 WEBER ET AL. J. CLIN. MICROBIOL. TABLE 3. Comparison between new stool concentration technique and standard FEA stool concentration procedure Mean no. (range) of detected oocystsa in: concnb Stool 1 (formed) Stool 2 (formed) Stool 3 (formed) Stool 4 (formed/fatty) Stool 5 (watery) FEAC Newd FEA New FEA New FEA New FEA New 1, , (0-6) (0-5) (0-3) (0-6) 4.1 (1-9) 10, (0-3) 2.6 (1-6) (1-7) 0.4 (0-1) 4.9 (1-7) (0-3) 12.2 (4-16) 18.3 (6-22) 50,000 e 2.8 (0-5) 40.9 (19-64) 1.0 (0-3) 12.6 (2-21) 0.7 (0-2) 19.5 (10-37) 0.3 (0-3) 2.4 (0-5) e a Slides were scanned completely. b Number of oocysts per gram of stool. c FEA, FEA stool concentration procedure. d New, new stool concentration procedure. e, all 10 specimens examined were positive; oocysts were not counted. gauze that was used for stool filtration and in the supernatants from both the first and second centrifugation steps), we attempted to modify the FEA procedure (i.e., by using alternative filter material or varying the relative centrifugal force or centrifugation times) to improve oocyst recovery (34). However, these modifications offered no improvement and, in some cases, impaired oocyst detection. Separation of parasites from other fecal elements is a crucial step in coprodiagnostic procedures and appears to be enhanced by centrifugal flotation techniques, such as Sheather's sugar and zinc sulfate. In our experience, these procedures provide clean preparations of oocysts but are inferior to other stool-processing techniques for concentrating oocysts. Similarly, epidemiologic data do not support the superiority of Sheather's sugar centrifugal flotation for detection of Cryptosporidium spp. (21). In our study, Sheather's sugar and zinc sulfate centrifugal flotation techniques had a lower sensitivity for oocyst recovery than did the FEA procedure. The new stool concentration procedure described here provided excellent separation of parasites from stool debris and enhanced detection of Cryptosporidium oocysts in all stool samples. The greatest improvement in oocyst recovery occurred when formed stool specimens were processed. In the first step of the procedure, stool fat and mucus were removed by Formalin-ethyl acetate sedimentation. In the next step, parasites were separated from most fecal debris. In the third step, fecal material containing Cryptosporidium oocysts was concentrated to obtain the sediment that was used to prepare the slides. The new method was technically easy to perform but involved longer centrifugation times and additional pipetting. This work load can be reduced, however, by processing stool specimens in batches. Moreover, whereas slides of FEA concentrations required microscopic examination at a magnification of x400 to distinguish oocysts from fluorescing background fecal debris, we were able to scan slides rapidly at a magnification of x200, using the new technique without affecting the sensitivity of oocyst detection because these slides had much less background debris. Improved detection of Cryptospordium oocysts in formed stools has important implications for both clinical and epidemiologic studies when the diagnosis of early or asymptomatic infections is needed. Indeed, asymptomatic carriage of Cryptosporidium spp. has been described repeatedly (11, 15, 16, 24, 26, 33, 37), and a high rate of asymptomatic carriage (12.7%) has been documented in one group of immunocompetent patients undergoing endoscopy (26). Sensitive stool detection techniques are also required to monitor responses to new therapeutic agents for cryptosporidiosis. As therapeutic agents become available, early detection could allow for earlier treatment and possible prevention of the lifethreatening diarrhea often seen in immunocompromised patients. Available epidemiologic data (12, 19, 26) as well as our findings suggest that in patients who are stool negative but are strongly suspected of harboring a Cryptosporidium infection, examination of intestinal biopsies and duodenal aspirates may be indicated since current stool detection methods will not detect infections in patients who are excreting fewer than 5,000 to 10,000 Cryptosporidium oocysts per g of stool. Further validation of the new stool concentration technique as an epidemiologic or clinical tool is needed. In addition, further study is needed to assess the ability of this technique to concentrate helminth eggs and protozoa other than Cryptosporidium spp. The absence of a "gold standard" for the diagnosis of Cryptosporidium infection continues to be an obstacle to the development and evaluation of new coprodiagnostic techniques. The techniques described here (i.e., with stool specimens seeded with known numbers of C. parvum oocysts) maybe useful to others who wish to pursue this objective. ACKNOWLEDGMENTS We thank Susanne P. Wahlquist and Henry S. Bishop for technical assistance, Michael J. Arrowood for providing Cryptosporidium oocysts, and Phyllis Moir for help in editing the manuscript. This work was supported in part by the Swiss National Science Foundation, Zurich, Switzerland (scholarship to R.W.). REFERENCES 1. Alpert, G., L. M. Bell, C. E. Kirkpatrick, L. D. Budnick, J. M. Campos, H. M. Friedman, and S. A. Plotldn Outbreak of cryptosporidiosis in a day-care center. Pediatrics 77: Anusz, K. Z., P. H. Mason, M. W. Riggs, and L. E. Perryman Detection of Cryptosponidium parvum oocysts in bovine feces by monoclonal antibody capture enzyme-linked immunosorbent assay. J. Clin. Microbiol. 28: Arrowood, M. J., and C. R. Sterling Isolation of Cryptosporidium oocysts and sporozoites using discontinuous sucrose and isopycnic percoll gradients. J. Parasitol. 73: Arrowood, M. J., and C. R. Sterling Comparison of conventional staining methods and monoclonal antibody-based methods for Cryptosporidium oocyst detection. J. Clin. Microbiol. 27: Casemore, D. P Laboratory methods for diagnosing cryptosporidiosis. J. Clin. Pathol. 44: Casemore, D. P., M. Armstrong, and R. L. Sands Laboratory diagnosis of crytosporidiosis. J. Clin. Pathol. 38:
5 VOL. 30, 1992 STOOL CONCENTRATION PROCEDURES Current, W. L., N. C. Reese, J. V. Ernst, W. S. Bailey, M. B. Heyman, and W. M. Weinstein Human cryptosporidiosis in immunocompetent and immunodeficient persons. N. Engl. J. Med. 308: D'Antonio, R. G., R. E. Winn, J. P. Taylor, T. L. Gustafson, W. L. Current, M. M. Rhodes, G. W. Gary, and R. A. Zajac A waterborne outbreak of cryptosporidiosis in normal hosts. Ann. Intern. Med. 103: Garcia, L. S., T. C. Brewer, and D. A. Bruckner Fluorescence detection of Cryptosporidium oocysts in human fecal specimens by using monoclonal antibodies. J. Clin. Microbiol. 25: Garcia, L. S., D. A. Bruckner, T. C. Brewer, and R. Y. Shimizu Techniques for the recovery and identification of Cryptosporidium oocysts from stool specimens. J. Clin. Microbiol. 18: Gentile, G., A. Caprioli, G. Donelli, M. Venditti, F. Mandeili, and P. Martino Asymptomatic carriage of Cryptosporidium in two patients with leukemia. Am. J. Infect. Control 18: Greenson, J. K., P. C. Belitsos, J. H. Yardley, and J. G. Batlett AIDS enteropathy: occult enteric infections and duodenal mucosal alterations in chronic diarrhea. Ann. Intern. Med. 114: Hayes, E. B., T. D. Matte, T. R. O'Brien, T. W. McKinley, G. S. Logsdon, J. B. Rose, B. L. P. Ungar, D. M. Word, P. F. Pinsky, M. L. Cummings, M. A. Wilson, E. G. Long, E. S. Hurwitz, and D. D. Juranek Large community outbreak of cryptosporidiosis due to contamination of a ifitered public water supply. N. Engl. J. Med. 320: Holley, H. P., and C. Dover Cryptosporidium: a common cause of parasitic diarrhea in otherwise healthy individuals. J. Infect. Dis. 153: Janoff, E. N., C. Limas, and R. L. Gebhard Cryptosporidial carriage without symptoms in the acquired immunodeficiency syndrome (AIDS). Ann. Intern. Med. 112: Joidpii, L., and A. M. Jokipii Timing of symptoms of oocyst excretion in human cryptosporidiosis. N. Engl. J. Med. 315: Jokipii, L., S. Pohojola, and A. M. Jokipii Cryptosporidiosis associated with traveling and giardiasis. Gastroenterology 89: Koch, K. L., D. J. Philipps, R. C. Aber, and W. L. Current Cryptosporidiosis in hospital personnel. Ann. Intern. Med. 102: Kotler, P. D., A. Francisco, F. Clayton, J. V. Scholes, and J. M. Orenstein Small intestinal injury and parasitic diseases in AIDS. Ann. Intern. Med. 113: Ma, P., and R. Soave Three-step examination for cryptosporidiosis in 10 homosexual men with protracted watery diarrhea. J. Infect. Dis. 147: McNabb, S. J., D. M. Hensel, D. F. Welch, H. Heibel, G. L. McKee, and G. R. Istre Comparison of sedimentation and flotation techniques for identification of Cryptosporidium sp. oocysts in a large outbreak of human diarrhea. J. Clin. Microbiol. 22: Paik, G., and M. T. Suggs Reagents, stains, and miscellaneous test procedures, p In E. H. Lennette, E. H. Spaulding, and J. P. Truant (ed.), Manual of clinical microbiology, 2nd ed. American Society for Microbiology, Washington, D.C. 23. Perry, J. L., J. S. Matthews, and G. R. Miller Parasite detection efficiencies of five stool concentration systems. J. Clin. Microbiol. 28: Ravn, P., J. D. Lundgren, P. Kjaeldgaard, W. Holten-Anderson, N. Hjlyng, J. 0. Nielson, and J. Gaub Nosocomial outbreak of cryptosporidiosis in AIDS patients. Br. Med. J. 302: Ritchie, L. S An ether sedimentation technique for routine stool examinations. Bull. U.S. Army Med. Dept. 8: Roberts, W. G., P. H. R. Green, J. Ma, M. Carr, and A. M. Ginsberg Prevalence of cryptosporidiosis in patients undergoing endoscopy: evidence for an asymptomatic carrier state. Am. J. Med. 87: Rusnak, J., T. L. Hadfield, M. M. Rhodes, and J. K. Gaines Detection of Cryptosporidium oocysts in human fecal specimens by an indirect immunofluorescence assay with monoclonal antibodies. J. Clin. Microbiol. 27: Sheather, A. L The detection of intestinal protozoan and mange parasites by a flotation technique. J. Comp. Pathol. 36: Sheperd, R. C., C. L. Reed, and G. P. Sinha Shedding of oocysts of Cryptosporidium in immunocompetent patients. J. Clin. Pathol. 41: Soave, R., and W. D. Johnson Cryptosporidium and Isospora belli infection. J. Infect. Dis. 157: Soave, R., and P. Ma Cryptosporidiosis. Traveler's diarrhea in two families. Arch. Intern. Med. 145: Ungar, B. L. P Enzyme-linked immunoassay for detection of Cryptosporidium antigens in fecal specimens. J. Clin. Microbiol. 28: Vajro, P., L. de Martino, S. Scotti, C. Barbati, A. Fontanella, and M. Pettoello Montovani Intestinal Cryptosporidium carriage in two liver-transplanted children. J. Pediatr. Gastroenterol. Nutr. 12: Weber, R., R. T. Bryan, H. S. Bishop, S. P. Wahlquist, J. J. Sullivan, and D. D. Juranek Threshold of detection of Cryptosporidium oocysts in human stool specimens: evidence for low sensitivity of current diagnostic methods. J. Clin. Microbiol. 29: Wolfson, J. S., J. M. Richter, M. A. Waldron, D. J. Weber, D. M. McCarthy, and C. C. Hopkins Cryptosporidiosis in immunocompetent patients. N. Engl. J. Med. 312: Young, KL H., S. L. Bullock, D. M. Melvin, and C. L. Spruill Ethyl acetate as a substitute for diethyl ether in the Formalin-ether sedimentation technique. J. Clin. Microbiol. 10: Zar, F., P. J. Geiseler, and V. A. Brown Asymptomatic carriage of Cryptosporidium in the stool of a patient with acquired immunodeficiency syndrome. J. Infect. Dis. 151: Zierdt, W. S Concentration and identification of Cryptosporidium sp. by use of a parasite concentrator. J. Clin. Microbiol. 20:
Specimens: Evidence for Low Sensitivity of Current Diagnostic Methods
 JOURNAL OF CLINICAL MICROBIOLOGY, July 1991, p. 1323-1327 0095-1137/91/071323-05$02.00/0 Copyright D 1991, American Society for Microbiology Vol. 29, No. 7 Threshold of Detection of Cryptosporidium Oocysts
JOURNAL OF CLINICAL MICROBIOLOGY, July 1991, p. 1323-1327 0095-1137/91/071323-05$02.00/0 Copyright D 1991, American Society for Microbiology Vol. 29, No. 7 Threshold of Detection of Cryptosporidium Oocysts
Origination Date: 25 Mar 2004 Total Pages: Effective Date: 18 Sep 2012 SOP Number LTC-SOP-17 v2.0. Network Name, Title Signature Date
 Title: Stool Processing and Cryptosporidium/Giardia Origination Date: 25 Mar 2004 Total Pages: Effective Date: 18 Sep 2012 SOP Number LTC-SOP-17 v2.0 Prepared By: Bill Kabat and Nancy Isham of the Supersedes
Title: Stool Processing and Cryptosporidium/Giardia Origination Date: 25 Mar 2004 Total Pages: Effective Date: 18 Sep 2012 SOP Number LTC-SOP-17 v2.0 Prepared By: Bill Kabat and Nancy Isham of the Supersedes
Cryptosporidium Veterinary
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
Crypto / Giardia Ag Combo (Fecal) ELISA
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
PARA-TECT Cryptosporidium/Giardia Direct Fluorescent Assay Directions For Use For In Vitro Diagnostic Use
 Medical Chemical Corp., 19430 Van Ness Avenue, Torrance, California 90501 (310)787-6800, Fax (310)787-4464 Catalog # MCC-C/G - DFA, 75 Test PARA-TECT Cryptosporidium/Giardia Direct Fluorescent Assay Directions
Medical Chemical Corp., 19430 Van Ness Avenue, Torrance, California 90501 (310)787-6800, Fax (310)787-4464 Catalog # MCC-C/G - DFA, 75 Test PARA-TECT Cryptosporidium/Giardia Direct Fluorescent Assay Directions
Enzyme-Linked Immunoassay for Detection of Cryptosporidium Antigens in Fecal Specimens
 JOURNAL OF CLINICAL MICROBIOLOGY, Nov. 1990, p. 2491-2495 0095-1137/90/112491-05$02.00/0 Copyright C 1990, American Society for Microbiology Vol. 28, No. 11 Enzyme-Linked Immunoassay for Detection of Cryptosporidium
JOURNAL OF CLINICAL MICROBIOLOGY, Nov. 1990, p. 2491-2495 0095-1137/90/112491-05$02.00/0 Copyright C 1990, American Society for Microbiology Vol. 28, No. 11 Enzyme-Linked Immunoassay for Detection of Cryptosporidium
Cryptosporidiosis: Multiattribute Evaluation of Six
 JOURNAL OF CLINICAL MICROBIOLOGY, Feb. 1993, P. 198-202 0095-1137/93/020198-05$02.00/0 Copyright 1993, American Society for Microbiology Vol. 31, No. 2 Cryptosporidiosis: Multiattribute Evaluation of Six
JOURNAL OF CLINICAL MICROBIOLOGY, Feb. 1993, P. 198-202 0095-1137/93/020198-05$02.00/0 Copyright 1993, American Society for Microbiology Vol. 31, No. 2 Cryptosporidiosis: Multiattribute Evaluation of Six
American Association of Bioanalysts 5615 Kirby Drive, Suite 870 Houston, TX
 Q3 2018 Parasitology American Association of Bioanalysts 5615 Kirby Drive, Suite 870 Houston, TX 77005 800-234-5315 281-436-5357 Specimen 1 Referees Extent 1 Extent 2 Total Few to 534 Giardia lamblia Many
Q3 2018 Parasitology American Association of Bioanalysts 5615 Kirby Drive, Suite 870 Houston, TX 77005 800-234-5315 281-436-5357 Specimen 1 Referees Extent 1 Extent 2 Total Few to 534 Giardia lamblia Many
Comparison of Polyvinyl Alcohol Fixative with Three Less Hazardous Fixatives for Detection and Identification of Intestinal Parasites
 JOURNAL OF CLINICAL MICROBIOLOGY, Apr. 2000, p. 1592 1598 Vol. 38, No. 4 0095-1137/00/$04.00 0 Comparison of Polyvinyl Alcohol Fixative with Three Less Hazardous Fixatives for Detection and Identification
JOURNAL OF CLINICAL MICROBIOLOGY, Apr. 2000, p. 1592 1598 Vol. 38, No. 4 0095-1137/00/$04.00 0 Comparison of Polyvinyl Alcohol Fixative with Three Less Hazardous Fixatives for Detection and Identification
Parasitology. Helminthology (Helminths)
 Parasitology Protozoology (Protozoa) Helminthology (Helminths) Entomology (Arthropodes) Platyhelminthes (flat worms) Nematheminthes (round worms) Trematodes Nematodes Cestodes Collection of the specimens
Parasitology Protozoology (Protozoa) Helminthology (Helminths) Entomology (Arthropodes) Platyhelminthes (flat worms) Nematheminthes (round worms) Trematodes Nematodes Cestodes Collection of the specimens
Comparison of Formol-Ether, Direct Smear and Nigrosine Methylene Blue for the Diagnosis of Human Intestinal Parasites
 Journal of Microbiology Research and Reviews Vol. (): -, August, www.resjournals.org/jmr Comparison of Formol-Ether, Direct Smear and Nigrosine Methylene Blue for the Diagnosis of Human Intestinal Parasites
Journal of Microbiology Research and Reviews Vol. (): -, August, www.resjournals.org/jmr Comparison of Formol-Ether, Direct Smear and Nigrosine Methylene Blue for the Diagnosis of Human Intestinal Parasites
Evaluation of a Commercially Available Enzyme-Linked Immunosorbent Assay for Giardia lamblia Antigen in Stool
 JOURNAL OF CLINICAL MICROBIOLOGY, June 1991, p. 1137-1142 0095-1137/91/061137-06$02.00/0 Copyright C 1991, American Society for Microbiology Vol. 29, No. 6 Evaluation of a Commercially Available Enzyme-Linked
JOURNAL OF CLINICAL MICROBIOLOGY, June 1991, p. 1137-1142 0095-1137/91/061137-06$02.00/0 Copyright C 1991, American Society for Microbiology Vol. 29, No. 6 Evaluation of a Commercially Available Enzyme-Linked
Research Method Validation
 Evaluation of the FECPAK Sheep Method (Moredun Institute, Scotland) General Observations The microscope included in the kit had a "floating stage"; samples would rapidly fade out of focus making counting
Evaluation of the FECPAK Sheep Method (Moredun Institute, Scotland) General Observations The microscope included in the kit had a "floating stage"; samples would rapidly fade out of focus making counting
Giardia lamblia (flagellates)
 Giardia lamblia (flagellates) Dr. Hala Al Daghistani Giardia lamblia (Giardia duodenalis or Giardia intestinalis) is the causative agent of giardiasis and is the only common pathogenic protozoan found
Giardia lamblia (flagellates) Dr. Hala Al Daghistani Giardia lamblia (Giardia duodenalis or Giardia intestinalis) is the causative agent of giardiasis and is the only common pathogenic protozoan found
PARASITOLOGY CASE HISTORY 15 (HISTOLOGY) (Lynne S. Garcia)
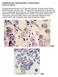 PARASITOLOGY CASE HISTORY 15 (HISTOLOGY) (Lynne S. Garcia) A biopsy was performed on a 27-year-old man with no known travel history, presenting with a perianal ulcer. The specimen was preserved in formalin
PARASITOLOGY CASE HISTORY 15 (HISTOLOGY) (Lynne S. Garcia) A biopsy was performed on a 27-year-old man with no known travel history, presenting with a perianal ulcer. The specimen was preserved in formalin
PROTOZOAN ENTERIC INFECTION IN AIDS RELATED DIARRHEA IN THAILAND
 PROTOZOAN ENTERIC INFECTION IN AIDS RELATED DIARRHEA IN THAILAND Duangdao Waywa 1, Siriporn Kongkriengdaj 1, Suparp Chaidatch 1, Surapee Tiengrim 1 Boonchai Kowadisaiburana 2, Suchada Chaikachonpat 1,
PROTOZOAN ENTERIC INFECTION IN AIDS RELATED DIARRHEA IN THAILAND Duangdao Waywa 1, Siriporn Kongkriengdaj 1, Suparp Chaidatch 1, Surapee Tiengrim 1 Boonchai Kowadisaiburana 2, Suchada Chaikachonpat 1,
Prevalence of Intestinal Parasites in HIV Seropositive Patients with and without Diarrhoea and its Correlation with CD4 Counts
 International Journal of Current Microbiology and Applied Sciences ISSN: 2319-7706 Volume 5 Number 10 (2016) pp. 527-532 Journal homepage: http://www.ijcmas.com Original Research Article http://dx.doi.org/10.20546/ijcmas.2016.510.058
International Journal of Current Microbiology and Applied Sciences ISSN: 2319-7706 Volume 5 Number 10 (2016) pp. 527-532 Journal homepage: http://www.ijcmas.com Original Research Article http://dx.doi.org/10.20546/ijcmas.2016.510.058
PARATEST STABILITY STUDY, SYSTEM USED FOR DIAGNOSIS OF INTESTINAL PARASITES
 PARATEST STABILITY STUDY, SYSTEM USED FOR DIAGNOSIS OF INTESTINAL PARASITES Rev.00 January 10, 2011 1. Introduction Intestinal parasitic diseases represent a serious public health issue in underdeveloped
PARATEST STABILITY STUDY, SYSTEM USED FOR DIAGNOSIS OF INTESTINAL PARASITES Rev.00 January 10, 2011 1. Introduction Intestinal parasitic diseases represent a serious public health issue in underdeveloped
Catalog # OKDA00123 SUMMARY AND EXPLANATION
 Aviva Systems Biology 5754 Pacific Center Blvd., Suite 201, San Diego, CA 92121, USA Tel: 858-552-6979 Fax: 858-552-6975 www.avivasysbio.com Email: info@avivasysbio.com Giardia lamblia ELISA Kit Catalog
Aviva Systems Biology 5754 Pacific Center Blvd., Suite 201, San Diego, CA 92121, USA Tel: 858-552-6979 Fax: 858-552-6975 www.avivasysbio.com Email: info@avivasysbio.com Giardia lamblia ELISA Kit Catalog
Practice Guidelines for Ordering Stool Ova and Parasite Testing in a Pediatric Population
 CLINICAL MICROBIOLOGY AND INFECTIOUS DISEASE Original Article Practice Guidelines for Ordering Stool Ova and Parasite Testing in a Pediatric Population AMIN KABANI, MD, FRCPC, GISELE CADRAIN, RT, CYNTHIA
CLINICAL MICROBIOLOGY AND INFECTIOUS DISEASE Original Article Practice Guidelines for Ordering Stool Ova and Parasite Testing in a Pediatric Population AMIN KABANI, MD, FRCPC, GISELE CADRAIN, RT, CYNTHIA
Sure-Vue Signature Cryptosporidium/Giardia Test
 SAMPLE PROCEDURE This procedure is provided to Fisher Healthcare customers to assist with the development of laboratory procedures. This document was derived from, and was current with, the instructions
SAMPLE PROCEDURE This procedure is provided to Fisher Healthcare customers to assist with the development of laboratory procedures. This document was derived from, and was current with, the instructions
Dr. Jabar Etaby Lecture GIARDIASIS(lambliasis) Etiology: Giardia lamblia (flagellate)
 Dr. Jabar Etaby Lecture Two GIARDIASIS(lambliasis) Etiology: Giardia lamblia (flagellate) Epidemiology: It has worldwide distribution and is not uncommon in South Carolina. It is the most frequent protozoan
Dr. Jabar Etaby Lecture Two GIARDIASIS(lambliasis) Etiology: Giardia lamblia (flagellate) Epidemiology: It has worldwide distribution and is not uncommon in South Carolina. It is the most frequent protozoan
Giardiasis. Table of Contents
 Table of Contents Case Definition... Error! Bookmark not defined. Reporting Requirements... 2 Etiology... Error! Bookmark not defined. Clinical Presentation... Error! Bookmark not defined. Diagnosis...
Table of Contents Case Definition... Error! Bookmark not defined. Reporting Requirements... 2 Etiology... Error! Bookmark not defined. Clinical Presentation... Error! Bookmark not defined. Diagnosis...
Comparison of Three Collection-Preservation Methods for
 JOURNAL OF CLINICAL MICROBIOLOGY, Dec. 1981, p. 656-660 0095-1 137/81/120656-05$02.00/0 Vol. 14, No. 6 Comparison of Three Collection-Preservation Methods for Detection of Intestinal Parasites DONALD L.
JOURNAL OF CLINICAL MICROBIOLOGY, Dec. 1981, p. 656-660 0095-1 137/81/120656-05$02.00/0 Vol. 14, No. 6 Comparison of Three Collection-Preservation Methods for Detection of Intestinal Parasites DONALD L.
ccess safe drinking wa r is everyone s right Protozoans that cause diarrheal disease
 ccess safe drinking wa r is everyone s right Protozoa: Protozoans that cause diarrheal disease 1. Giardia lamblia 2. Entameba histolytica 3. Cryptosporidium parvum 4. Cyclospora cayetanensis 1 Giardia
ccess safe drinking wa r is everyone s right Protozoa: Protozoans that cause diarrheal disease 1. Giardia lamblia 2. Entameba histolytica 3. Cryptosporidium parvum 4. Cyclospora cayetanensis 1 Giardia
Prevalence of Intestinal Parasitic Infections in HIV-Positive Patients
 ISSN: 2319-7706 Volume 4 Number 5 (2015) pp. 269-273 http://www.ijcmas.com Original Research Article Prevalence of Intestinal Parasitic Infections in HIV-Positive Patients Vasundhara*, Haris M.Khan, Harekrishna
ISSN: 2319-7706 Volume 4 Number 5 (2015) pp. 269-273 http://www.ijcmas.com Original Research Article Prevalence of Intestinal Parasitic Infections in HIV-Positive Patients Vasundhara*, Haris M.Khan, Harekrishna
Giardiasis Surveillance Protocol
 Provider Responsibilities 1. Report all cases to your local health department by completing the provider section of the WVEDSS form within the timeframe indicated: Sporadic case of - should be reported
Provider Responsibilities 1. Report all cases to your local health department by completing the provider section of the WVEDSS form within the timeframe indicated: Sporadic case of - should be reported
EPIDEMIOLOGY OF GIARDIASIS AND CRYPTOSPORIDIOSIS IN JAMAICA
 Am. J. Trop. Med. Hyg., 59(5), 1998, pp. 717 721 Copyright 1998 by The American Society of Tropical Medicine and Hygiene EPIDEMIOLOGY OF GIARDIASIS AND CRYPTOSPORIDIOSIS IN JAMAICA JOHN F. LINDO, VALERIE
Am. J. Trop. Med. Hyg., 59(5), 1998, pp. 717 721 Copyright 1998 by The American Society of Tropical Medicine and Hygiene EPIDEMIOLOGY OF GIARDIASIS AND CRYPTOSPORIDIOSIS IN JAMAICA JOHN F. LINDO, VALERIE
Prevalence of Giardiasis in Patients Attending Tertiary Care Hospital in Northern India
 ISSN: 2319-7706 Volume 4 Number 5 (2015) pp. 339-344 http://www.ijcmas.com Original Research Article Prevalence of Giardiasis in Patients Attending Tertiary Care Hospital in Northern India Sangram Singh
ISSN: 2319-7706 Volume 4 Number 5 (2015) pp. 339-344 http://www.ijcmas.com Original Research Article Prevalence of Giardiasis in Patients Attending Tertiary Care Hospital in Northern India Sangram Singh
Concurrent response to challenge infection with Cryptosporidium parvum in
 J. Vet. Sci. (2006),G7(1), 47 51 J O U R N A L O F Veterinary Science Concurrent response to challenge infection with Cryptosporidium parvum in immunosuppressed C57BL/6N mice Chan-Gu Surl 1, Hyeon-Cheol
J. Vet. Sci. (2006),G7(1), 47 51 J O U R N A L O F Veterinary Science Concurrent response to challenge infection with Cryptosporidium parvum in immunosuppressed C57BL/6N mice Chan-Gu Surl 1, Hyeon-Cheol
Cryptosporidium is a protozoa in the Phylum Apicomplexa Cryptosporidium Parvum genotype 1
 September, 2010 Cryptosporidium is a protozoa in the Phylum Apicomplexa Cryptosporidium Parvum genotype 1 Livestock not commonly infected but can occur through contamination of feeds by other species,
September, 2010 Cryptosporidium is a protozoa in the Phylum Apicomplexa Cryptosporidium Parvum genotype 1 Livestock not commonly infected but can occur through contamination of feeds by other species,
Hot Topics in Infectious Diseases: Enteric Infections in the Arctic
 Hot Topics in Infectious Diseases: Enteric Infections in the Arctic Tobey Audcent MD, FRCPC Department of Pediatrics Children s Hospital of Eastern Ontario 6 th International Meeting on Indigenous Child
Hot Topics in Infectious Diseases: Enteric Infections in the Arctic Tobey Audcent MD, FRCPC Department of Pediatrics Children s Hospital of Eastern Ontario 6 th International Meeting on Indigenous Child
MORPHOLOGY AND FREQUENCY DISTRIBUTION OF PROTOZOAN BLASTOCYSTIS HOMINIS
 MORPHOLOGY AND FREQUENCY DISTRIBUTION OF PROTOZOAN BLASTOCYSTIS HOMINIS Abstract Pages with reference to book, From 322 To 324 M. Naqi Zafar ( Zafar Research and Diagnostic Centre, 7/14, Riznpa Plaza,
MORPHOLOGY AND FREQUENCY DISTRIBUTION OF PROTOZOAN BLASTOCYSTIS HOMINIS Abstract Pages with reference to book, From 322 To 324 M. Naqi Zafar ( Zafar Research and Diagnostic Centre, 7/14, Riznpa Plaza,
Cryptosporidium spp. ELISA Kit
 Cryptosporidium spp. ELISA Kit Catalog Number KA2094 96 assays Version: 03 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
Cryptosporidium spp. ELISA Kit Catalog Number KA2094 96 assays Version: 03 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
News and Notes. Parasitology Comprehensive 5 November 2013
 NEW YORK STATE Parasitology Proficiency Testing Program News and Notes Beginning with the May 2013 event a separate set of 3 samples were supplied for laboratories performing antigen detection. For the
NEW YORK STATE Parasitology Proficiency Testing Program News and Notes Beginning with the May 2013 event a separate set of 3 samples were supplied for laboratories performing antigen detection. For the
District NTD Training module 9 Learners Guide
 District NTD Training module 9 Learners Guide Session 3: Diagnostic and laboratory tools for filarial worms, Soil- Transmitted Helminths and Schistosomiasis Key message addressed to communities: Community
District NTD Training module 9 Learners Guide Session 3: Diagnostic and laboratory tools for filarial worms, Soil- Transmitted Helminths and Schistosomiasis Key message addressed to communities: Community
Cryptosporidiosis in Wisconsin: A case-control study of
 Epidemiol. Infect. (1996), 117, 297-304 Copyright ( 1996 Cambridge University Press Cryptosporidiosis in Wisconsin: A case-control study of post-outbreak transmission P. OSEWE', D. G. ADDISS2*, K. A. BLAIR3,
Epidemiol. Infect. (1996), 117, 297-304 Copyright ( 1996 Cambridge University Press Cryptosporidiosis in Wisconsin: A case-control study of post-outbreak transmission P. OSEWE', D. G. ADDISS2*, K. A. BLAIR3,
IMMUNOFLUORESCENT STAINING FOR RESPIRATORY VIRUS DETECTION: INFLUENZA A, INFLUENZA B AND RESPIRATORY SYNCYTIAL VIRUS
 IMMUNOFLUORESCENT STAINING FOR RESPIRATORY VIRUS DETECTION: INFLUENZA A, INFLUENZA B AND RESPIRATORY SYNCYTIAL VIRUS A. Introduction Influenza A, Influenza B and RSV are responsible for serious respiratory
IMMUNOFLUORESCENT STAINING FOR RESPIRATORY VIRUS DETECTION: INFLUENZA A, INFLUENZA B AND RESPIRATORY SYNCYTIAL VIRUS A. Introduction Influenza A, Influenza B and RSV are responsible for serious respiratory
Human Influenza A (Swine Flu) Rapid test
 Human Influenza A (Swine Flu) Rapid test Cat.No: DTSXY-Z9 Lot. No. (See product label) Size 20T Intended use The Influenza A (Swine Flu) test is a rapid chromatographic immunoassay for the qualitative
Human Influenza A (Swine Flu) Rapid test Cat.No: DTSXY-Z9 Lot. No. (See product label) Size 20T Intended use The Influenza A (Swine Flu) test is a rapid chromatographic immunoassay for the qualitative
Incidence And Risk Factors Of Cryptosporidium Spp. Infection In Water Buffaloes Confined In A Communal Management System In The Philippines
 ISPUB.COM The Internet Journal of Veterinary Medicine Volume 8 Number 1 Incidence And Risk Factors Of Cryptosporidium Spp. Infection In Water Buffaloes Confined In A Communal Management System In The Philippines
ISPUB.COM The Internet Journal of Veterinary Medicine Volume 8 Number 1 Incidence And Risk Factors Of Cryptosporidium Spp. Infection In Water Buffaloes Confined In A Communal Management System In The Philippines
For Vets General Information Prevalence and Risk Factors Humans
 For Vets General Information Cryptosporidium spp. are intestinal protozoal parasites of animals and humans that cause the disease cryptosporidiosis. The primary clinical sign of infection is diarrhea,
For Vets General Information Cryptosporidium spp. are intestinal protozoal parasites of animals and humans that cause the disease cryptosporidiosis. The primary clinical sign of infection is diarrhea,
Alberta Health and Wellness Public Health Notifiable Disease Management Guidelines August 2011
 August 2011 Giardiasis Revision Dates Case Definition Reporting Requirements Remainder of the Guideline (i.e., Etiology to References sections inclusive) Case Definition August 2011 August 2011 October
August 2011 Giardiasis Revision Dates Case Definition Reporting Requirements Remainder of the Guideline (i.e., Etiology to References sections inclusive) Case Definition August 2011 August 2011 October
TYPES OF ORGANISM RELATIONSHIPS
 TYPES OF ORGANISM RELATIONSHIPS Normal Flora. Normal flora consists of microorganisms that are normally and consistently found in or on the body in the absence of disease. Symbiosis. This is the close
TYPES OF ORGANISM RELATIONSHIPS Normal Flora. Normal flora consists of microorganisms that are normally and consistently found in or on the body in the absence of disease. Symbiosis. This is the close
Evaluation of a New Rapid Plasma Reagin Card Test as a Screening Test for Syphilis
 JOURNAL OF CLINICAL MICROBIOLOGY, Aug. 1982, p. 286-290 0095-1 137/82/080286-05$02.00/0 Vol. 16, No. 2 Evaluation of a New Rapid Plasma Reagin Card Test as a Screening Test for Syphilis MARY W. PERRYMAN,'*
JOURNAL OF CLINICAL MICROBIOLOGY, Aug. 1982, p. 286-290 0095-1 137/82/080286-05$02.00/0 Vol. 16, No. 2 Evaluation of a New Rapid Plasma Reagin Card Test as a Screening Test for Syphilis MARY W. PERRYMAN,'*
PREVALENCE OF DIFFERENT PROTOZOAN PARASITES IN PATIENTS VISITING AT ICDDR B HOSPITAL, DHAKA
 J. Asiat. Soc. Bangladesh, Sci. 39(1): 117-123, June 213 PREVALENCE OF DIFFERENT PROTOZOAN PARASITES IN PATIENTS VISITING AT ICDDR B HOSPITAL, DHAKA SHAHELA ALAM, HAMIDA KHANUM 1, RIMI FARHANA ZAMAN AND
J. Asiat. Soc. Bangladesh, Sci. 39(1): 117-123, June 213 PREVALENCE OF DIFFERENT PROTOZOAN PARASITES IN PATIENTS VISITING AT ICDDR B HOSPITAL, DHAKA SHAHELA ALAM, HAMIDA KHANUM 1, RIMI FARHANA ZAMAN AND
E. Histolytica IgG ELISA Kit
 E. Histolytica IgG ELISA Kit Catalog Number KA3193 96 assays Version: 01 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of
E. Histolytica IgG ELISA Kit Catalog Number KA3193 96 assays Version: 01 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of
Washington State Department of Health (DOH) Cryptosporidium in Drinking Water Position Paper. Purpose
 Washington State Department of Health (DOH) Cryptosporidium in Drinking Water Position Paper Purpose Cryptosporidium is a micro-organism which has gained increased public health significance as a result
Washington State Department of Health (DOH) Cryptosporidium in Drinking Water Position Paper Purpose Cryptosporidium is a micro-organism which has gained increased public health significance as a result
Rotavirus Test Kit. Instructions For Use. Format: Cassette Specimen: Fecal Extract Catalog Number: VEL-001-ROTA
 Rotavirus Test Kit Instructions For Use Format: Cassette Specimen: Fecal Extract Catalog Number: VEL-001-ROTA * Please read the instructions carefully before use INTENDED USE Velotest Rotavirus Test is
Rotavirus Test Kit Instructions For Use Format: Cassette Specimen: Fecal Extract Catalog Number: VEL-001-ROTA * Please read the instructions carefully before use INTENDED USE Velotest Rotavirus Test is
Outline EP1201. NEHA 2012 AEC June 2012
 Food and Water Borne Enteric Protozoa: Environmental Health Perspectives Stephanie M. Fletcher, PhD (c) NEHA AEC & Exhibition San Diego June 30, 2012 Outline Introduction Overview of the Epidemiology of
Food and Water Borne Enteric Protozoa: Environmental Health Perspectives Stephanie M. Fletcher, PhD (c) NEHA AEC & Exhibition San Diego June 30, 2012 Outline Introduction Overview of the Epidemiology of
Prof. Mahmoud Rushdi. Assiut University Egypt.
 By Prof. Mahmoud Rushdi Faculty of Veterinary Mdii Medicine Assiut University Egypt Precautions to be taken during collection and preservation of fecal sample 2 1 Precautions during collection of fecal
By Prof. Mahmoud Rushdi Faculty of Veterinary Mdii Medicine Assiut University Egypt Precautions to be taken during collection and preservation of fecal sample 2 1 Precautions during collection of fecal
Giardia lamblia/ Cryptosporidium spp. ELISA Kit
 Giardia lamblia/ Cryptosporidium spp. ELISA Kit Catalog Number KA2095 96 assays Version: 02 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background...
Giardia lamblia/ Cryptosporidium spp. ELISA Kit Catalog Number KA2095 96 assays Version: 02 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background...
Effect of Nitazoxanide in Diarrhea and Enteritis Caused by Cryptosporidium Species
 CLINICAL GASTROENTEROLOGY AND HEPATOLOGY 2006;4:320 324 Effect of Nitazoxanide in Diarrhea and Enteritis Caused by Cryptosporidium Species JEAN FRANÇOIS ROSSIGNOL,* SAMIR M. KABIL, YEHIA EL GOHARY, and
CLINICAL GASTROENTEROLOGY AND HEPATOLOGY 2006;4:320 324 Effect of Nitazoxanide in Diarrhea and Enteritis Caused by Cryptosporidium Species JEAN FRANÇOIS ROSSIGNOL,* SAMIR M. KABIL, YEHIA EL GOHARY, and
Cryptosporidiosis. By: Nikole Stewart
 Cryptosporidiosis By: Nikole Stewart Cryptosporidiosis ("Crypto"); Etiological agent- Cryptosporidium (1) Transmission: Transmission occurs via the fecal-oral route when individuals ingest water or food
Cryptosporidiosis By: Nikole Stewart Cryptosporidiosis ("Crypto"); Etiological agent- Cryptosporidium (1) Transmission: Transmission occurs via the fecal-oral route when individuals ingest water or food
Public Health Microbiology. CE421/521 Lecture Prof. Tim Ellis
 Public Health Microbiology CE421/521 Lecture 10-03-06 Prof. Tim Ellis Pathogens and parasites Epidemiology Definitions epidemiology = study of spread of d in populations infectious disease = disease that
Public Health Microbiology CE421/521 Lecture 10-03-06 Prof. Tim Ellis Pathogens and parasites Epidemiology Definitions epidemiology = study of spread of d in populations infectious disease = disease that
cytoplasm contains two 2 nuclei and two parabasal bodies (Figure 7).
 Dr. Jabar Etaby Lecture one GIARDIASIS (lambliasis) Etiology: Giardia lamblia (flagellate) Epidemiology: It has worldwide distribution and is not uncommon in South Carolina. It is the most frequent protozoan
Dr. Jabar Etaby Lecture one GIARDIASIS (lambliasis) Etiology: Giardia lamblia (flagellate) Epidemiology: It has worldwide distribution and is not uncommon in South Carolina. It is the most frequent protozoan
PREVALENCE OF INTESTINAL PARASITES IN HIV-POSITIVE/AIDS PATIENTS. O.O Oguntibeju
 Malaysian Journal of Medical Sciences, Vol. 13, No. 1, January 2006 (68-73) ORIGINAL ARTICLE PREVALENCE OF INTESTINAL PARASITES IN HIV-POSITIVE/AIDS PATIENTS O.O Oguntibeju School of Health Technology,
Malaysian Journal of Medical Sciences, Vol. 13, No. 1, January 2006 (68-73) ORIGINAL ARTICLE PREVALENCE OF INTESTINAL PARASITES IN HIV-POSITIVE/AIDS PATIENTS O.O Oguntibeju School of Health Technology,
Recent Diagnostic Methods for Intestinal Parasitic Infections
 Recent Diagnostic Methods for Intestinal Parasitic Infections By Dr. Doaa Abdel Badie Salem Lecturer of Medical Parasitology, Mansoura Faculty of Medicine Agenda Intestinal parasites. Traditional Diagnostic
Recent Diagnostic Methods for Intestinal Parasitic Infections By Dr. Doaa Abdel Badie Salem Lecturer of Medical Parasitology, Mansoura Faculty of Medicine Agenda Intestinal parasites. Traditional Diagnostic
Veterinary Parasitology and Parasitic Diseases Department of Pathology and Animal Health Faculty of Veterinary Medicine, University of Naples
 Veterinary Parasitology and Parasitic Diseases Department of Pathology and Animal Health Faculty of Veterinary Medicine, University of Naples Federico II Via della Veterinaria, 1-80137 Naples, Italy www.flotac.unina.it
Veterinary Parasitology and Parasitic Diseases Department of Pathology and Animal Health Faculty of Veterinary Medicine, University of Naples Federico II Via della Veterinaria, 1-80137 Naples, Italy www.flotac.unina.it
Comparison of Lateral Flow Immunoassays to Current Stool Evaluation Methods for the Detection of Giardia and Cryptosporidium. Laurianne T.
 Comparison of Lateral Flow Immunoassays to Current Stool Evaluation Methods for the Detection of Giardia and Cryptosporidium Laurianne T. Mullinax A thesis submitted in partial fulfillment of the requirements
Comparison of Lateral Flow Immunoassays to Current Stool Evaluation Methods for the Detection of Giardia and Cryptosporidium Laurianne T. Mullinax A thesis submitted in partial fulfillment of the requirements
RIDA QUICK Cryptosporidium/Giardia/Entamoeba Combi
 RIDA QUICK Cryptosporidium/Giardia/Entamoeba Combi Article no: N1723 R-Biopharm AG, An der neuen Bergstraße 17, D-64297 Darmstadt, Germany Phone: +49 (0) 61 51 81 02-0 / Fax: +49 (0) 61 51 81 02-20 1.
RIDA QUICK Cryptosporidium/Giardia/Entamoeba Combi Article no: N1723 R-Biopharm AG, An der neuen Bergstraße 17, D-64297 Darmstadt, Germany Phone: +49 (0) 61 51 81 02-0 / Fax: +49 (0) 61 51 81 02-20 1.
Overview IMPORTANCE CLASSIFICATION SPECIMEN COLLECTION PROTOZOA WORMS BLOOD PARASITES ARTHROPODS DELUSIONAL PARASITOSIS QUIZZES GROSS
 IMPORTANCE Overview CLASSIFICATION SPECIMEN COLLECTION PROTOZOA WORMS BLOOD PARASITES ARTHROPODS DELUSIONAL PARASITOSIS QUIZZES GROSS - Of 6.6 billion people on Earth, 2.7 billion live on
IMPORTANCE Overview CLASSIFICATION SPECIMEN COLLECTION PROTOZOA WORMS BLOOD PARASITES ARTHROPODS DELUSIONAL PARASITOSIS QUIZZES GROSS - Of 6.6 billion people on Earth, 2.7 billion live on
Rapid-VIDITEST Swine Flu
 Rapid-VIDITEST Swine Flu One Step Influenza type A Antigen Card test. Instruction manual Producer: VIDIA spol. s r.o., Nad Safinou II 365, 252 50 Vestec, Czech Republic, Tel.: +420 261 090 565, www.vidia.cz
Rapid-VIDITEST Swine Flu One Step Influenza type A Antigen Card test. Instruction manual Producer: VIDIA spol. s r.o., Nad Safinou II 365, 252 50 Vestec, Czech Republic, Tel.: +420 261 090 565, www.vidia.cz
Fecal H. pylori Antigen Rapid Test (Strip)
 Fecal H. pylori Antigen Rapid Test (Strip) Cat. No.:DTS526 Pkg.Size: Intended use This H. pylori Antigen Rapid Test is intended for the direct qualitative detection of the presence of H. pylori antigen
Fecal H. pylori Antigen Rapid Test (Strip) Cat. No.:DTS526 Pkg.Size: Intended use This H. pylori Antigen Rapid Test is intended for the direct qualitative detection of the presence of H. pylori antigen
Veterinary Parasitology and Parasitic Diseases Department of Pathology and Animal Health Faculty of Veterinary Medicine, University of Naples
 Veterinary Parasitology and Parasitic Diseases Department of Pathology and Animal Health Faculty of Veterinary Medicine, University of Naples Federico II Via della Veterinaria, 1-80137 Naples, Italy www.flotac.unina.it
Veterinary Parasitology and Parasitic Diseases Department of Pathology and Animal Health Faculty of Veterinary Medicine, University of Naples Federico II Via della Veterinaria, 1-80137 Naples, Italy www.flotac.unina.it
News and Notes. Parasitology Comprehensive 5 February Sample Preparation and Quality Control. 13 A Helminthes Only
 NEW YORK STATE Parasitology Proficiency Testing Program News and Notes Recent reports in the literature have indicated a high rate of Cryptosporidium sp. false positive associated with Rapid Cartridge
NEW YORK STATE Parasitology Proficiency Testing Program News and Notes Recent reports in the literature have indicated a high rate of Cryptosporidium sp. false positive associated with Rapid Cartridge
Anton van Leeuwenhoek. Protozoa: This is what he saw in his own stool sample. Morphology 10/14/2009. Protozoans that cause diarrheal disease
 Access to safe drinking water is everyone s right Anton van Leeuwenhoek Protozoa: Protozoans that cause diarrheal disease This is what he saw in his own stool sample 1. Giardia lamblia 2. Entameba histolytica
Access to safe drinking water is everyone s right Anton van Leeuwenhoek Protozoa: Protozoans that cause diarrheal disease This is what he saw in his own stool sample 1. Giardia lamblia 2. Entameba histolytica
ADVL0411 study. (temozolomide in patients with leukemia)
 ADVL0411 study (temozolomide in patients with leukemia) I. Overview of study: We are interested in determining the level of MGMT activity and MRS mutations in leukemic samples (plasma and lymphoblasts)
ADVL0411 study (temozolomide in patients with leukemia) I. Overview of study: We are interested in determining the level of MGMT activity and MRS mutations in leukemic samples (plasma and lymphoblasts)
Access to safe drinking water is everyone s right. Protozoans that cause diarrheal disease
 Access to safe drinking water is everyone s right Protozoa: Protozoans that cause diarrheal disease 1. Giardia lamblia 2. Entameba histolytica 3. Cryptosporidium parvum 4. Cyclospora cayetanensis 1 Giardia
Access to safe drinking water is everyone s right Protozoa: Protozoans that cause diarrheal disease 1. Giardia lamblia 2. Entameba histolytica 3. Cryptosporidium parvum 4. Cyclospora cayetanensis 1 Giardia
PARASITE CONCENTRATOR FOR THE LABORATORY DIAGNOSIS OF INTESTINAL PARASITISM
 EVALUATION OF MINI PARASEP SF FAECAL PARASITE CONCENTRATOR FOR THE LABORATORY DIAGNOSIS OF INTESTINAL PARASITISM 1 IKEH, E.I. AND 2 ELUJOLA, M. 1 Department of Medical Microbiology/Medical Laboratory Science
EVALUATION OF MINI PARASEP SF FAECAL PARASITE CONCENTRATOR FOR THE LABORATORY DIAGNOSIS OF INTESTINAL PARASITISM 1 IKEH, E.I. AND 2 ELUJOLA, M. 1 Department of Medical Microbiology/Medical Laboratory Science
Instructions. Fuse-It-Color. Overview. Specifications
 Membrane fusion is a novel and highly superior method for incorporating various molecules and particles into mammalian cells. Cargo-specific liposomal carriers are able to attach and rapidly fuse with
Membrane fusion is a novel and highly superior method for incorporating various molecules and particles into mammalian cells. Cargo-specific liposomal carriers are able to attach and rapidly fuse with
FREQUENTLY ASKED QUESTIONS ABOUT DIAGNOSTIC PARASITOLOGY (LYNNE S. GARCIA:
 FREQUENTLY ASKED QUESTIONS ABOUT DIAGNOSTIC PARASITOLOGY (LYNNE S. GARCIA: LYNNEGARCIA2@VERIZON.NET) As laboratory personnel become more widely cross-trained, the availability of people who have expertise
FREQUENTLY ASKED QUESTIONS ABOUT DIAGNOSTIC PARASITOLOGY (LYNNE S. GARCIA: LYNNEGARCIA2@VERIZON.NET) As laboratory personnel become more widely cross-trained, the availability of people who have expertise
DEVELOPMENTAL VALIDATION OF SPERM HY-LITER EXPRESS Jennifer Old, Chris Martersteck, Anna Kalinina, Independent Forensics, Lombard IL
 DEVELOPMENTAL VALIDATION OF SPERM HY-LITER EXPRESS Jennifer Old, Chris Martersteck, Anna Kalinina, Independent Forensics, Lombard IL Background and Introduction SPERM HY-LITER is the first immunofluorescent
DEVELOPMENTAL VALIDATION OF SPERM HY-LITER EXPRESS Jennifer Old, Chris Martersteck, Anna Kalinina, Independent Forensics, Lombard IL Background and Introduction SPERM HY-LITER is the first immunofluorescent
Rapid-VIDITEST Enterovirus
 Rapid-VIDITEST Enterovirus A rapid one step Enterovirus Card test for the qualitative detection of Enterovirus antigens in human feces. Instruction manual Producer: VIDIA spol. s r.o., Nad Safinou II 365,
Rapid-VIDITEST Enterovirus A rapid one step Enterovirus Card test for the qualitative detection of Enterovirus antigens in human feces. Instruction manual Producer: VIDIA spol. s r.o., Nad Safinou II 365,
ab CytoPainter Golgi/ER Staining Kit
 ab139485 CytoPainter Golgi/ER Staining Kit Instructions for Use Designed to detect Golgi bodies and endoplasmic reticulum by microscopy This product is for research use only and is not intended for diagnostic
ab139485 CytoPainter Golgi/ER Staining Kit Instructions for Use Designed to detect Golgi bodies and endoplasmic reticulum by microscopy This product is for research use only and is not intended for diagnostic
Diagnostic tests for Cryptosporidium
 Diagnostic tests for Cryptosporidium Dr Rachel Chalmers Director, UK Cryptosporidium Reference Unit Public Health Wales Microbiology Singleton Hospital Swansea SA2 8QA What is Cryptosporidium? Protozoan,
Diagnostic tests for Cryptosporidium Dr Rachel Chalmers Director, UK Cryptosporidium Reference Unit Public Health Wales Microbiology Singleton Hospital Swansea SA2 8QA What is Cryptosporidium? Protozoan,
Recognizing African swine fever 23. Diagnosis of ASF
 Recognizing African swine fever 23 Diagnosis of ASF When large numbers of pigs of all ages die and the clinical signs and post mortem lesions look like those of ASF, that is the first disease that should
Recognizing African swine fever 23 Diagnosis of ASF When large numbers of pigs of all ages die and the clinical signs and post mortem lesions look like those of ASF, that is the first disease that should
CRYPTOSPORIDIUM PARVUM is a coccidian parasite
 Vol. 332 No. 13 INFECTIVITY OF CRYPTOSPORIDIUM PARVUM IN HEALTHY VOLUNTEERS 855 THE INFECTIVITY OF CRYPTOSPORIDIUM PARVUM IN HEALTHY VOLUNTEERS HERBERT L. DUPONT, M.D., CYNTHIA L. CHAPPELL, PH.D., CHARLES
Vol. 332 No. 13 INFECTIVITY OF CRYPTOSPORIDIUM PARVUM IN HEALTHY VOLUNTEERS 855 THE INFECTIVITY OF CRYPTOSPORIDIUM PARVUM IN HEALTHY VOLUNTEERS HERBERT L. DUPONT, M.D., CYNTHIA L. CHAPPELL, PH.D., CHARLES
Physician Use of Parasite Tests in the United States from 1997 to 2006 and in a Utah Cryptosporidium Outbreak in 2007
 JOURNAL OF CLINICAL MICROBIOLOGY, Feb. 2011, p. 591 596 Vol. 49, No. 2 0095-1137/11/$12.00 doi:10.1128/jcm.01806-10 Copyright 2011, American Society for Microbiology. All Rights Reserved. Physician Use
JOURNAL OF CLINICAL MICROBIOLOGY, Feb. 2011, p. 591 596 Vol. 49, No. 2 0095-1137/11/$12.00 doi:10.1128/jcm.01806-10 Copyright 2011, American Society for Microbiology. All Rights Reserved. Physician Use
Stool Collection Guidelines
 Stool Collection Guidelines Your child s stool (bowel movement) must be tested so we can plan the treatment for your child. You will need to collect the stool specimen at home. Bring it to a Laboratory
Stool Collection Guidelines Your child s stool (bowel movement) must be tested so we can plan the treatment for your child. You will need to collect the stool specimen at home. Bring it to a Laboratory
The following protocol describes the isolation of nuclei from tissue. Item. Catalog No Manufacturer
 SOP: Nuclei isolation from tissue and DNaseI treatment Date modified: 090923 Modified by: P. Sabo. (UW) The following protocol describes the isolation of nuclei from tissue. Ordering Information Item.
SOP: Nuclei isolation from tissue and DNaseI treatment Date modified: 090923 Modified by: P. Sabo. (UW) The following protocol describes the isolation of nuclei from tissue. Ordering Information Item.
Detection of gastrointestinal protozoa by microscopic examination
 Clinical and Analytical Evaluation of a Single-Vial Stool Collection Device with Formalin-Free Fixative for Improved Processing and Comprehensive Detection of Gastrointestinal Parasites Brianne A. Couturier,
Clinical and Analytical Evaluation of a Single-Vial Stool Collection Device with Formalin-Free Fixative for Improved Processing and Comprehensive Detection of Gastrointestinal Parasites Brianne A. Couturier,
DIAGNOSTIC MICROBIOLOGY
 DIAGNOSTIC MICROBIOLOGY Libuše Kolářová Václava Adámková Institute for Immunology and Microbiology, 1st. Fac.Med.,Charles Univ. in Prague Czech Republic Which factors should precipitate testing? CLINICAL
DIAGNOSTIC MICROBIOLOGY Libuše Kolářová Václava Adámková Institute for Immunology and Microbiology, 1st. Fac.Med.,Charles Univ. in Prague Czech Republic Which factors should precipitate testing? CLINICAL
Detection of Helminth Ova and Larvae in Trichrome-Stained Stool Smears
 JOURNAL OF CLINICAL MICROBIOIOGY, Dec. 1982. p. 1137-1144 Vol. 16. No. 6 0095-1137/82/121137-08$02.00/0 Copyright 1982, American Society for Microbiology Detection of Helminth Ova and Larvae in Trichrome-Stained
JOURNAL OF CLINICAL MICROBIOIOGY, Dec. 1982. p. 1137-1144 Vol. 16. No. 6 0095-1137/82/121137-08$02.00/0 Copyright 1982, American Society for Microbiology Detection of Helminth Ova and Larvae in Trichrome-Stained
MICROBIOLOGY SPECIMEN COLLECTION MANUAL
 Lee Memorial Health System Lee County, FL CLINICAL LABORATORY MICROBIOLOGY SPECIMEN COLLECTION MANUAL ACID FAST CULTURE Specimen Type see Specimen Chart ACID FAST STAIN see Specimen Chart Acid Fast stain
Lee Memorial Health System Lee County, FL CLINICAL LABORATORY MICROBIOLOGY SPECIMEN COLLECTION MANUAL ACID FAST CULTURE Specimen Type see Specimen Chart ACID FAST STAIN see Specimen Chart Acid Fast stain
for Syphilis visualize the antigen-antibody reaction. Procedures
 JOURNAL OF CLINICAL MICROBIOLOGY, Nov. 1983, p. 1141-1145 0095-1137/83/111141-05$02.00/0 Copyright X 1983, American Society for Microbiology Vol. 18, No. 5 Toluidine Red Unheated Serum Test, a Nontreponemal
JOURNAL OF CLINICAL MICROBIOLOGY, Nov. 1983, p. 1141-1145 0095-1137/83/111141-05$02.00/0 Copyright X 1983, American Society for Microbiology Vol. 18, No. 5 Toluidine Red Unheated Serum Test, a Nontreponemal
Rapid-VIDITEST. Astrovirus Card
 Rapid-VIDITEST Astrovirus Card One step Astrovirus Card Test. Instruction manual INTENDED USE: The Rapid-VIDITEST Astrovirus Card is a one step coloured chromatographic immunoassay for the qualitative
Rapid-VIDITEST Astrovirus Card One step Astrovirus Card Test. Instruction manual INTENDED USE: The Rapid-VIDITEST Astrovirus Card is a one step coloured chromatographic immunoassay for the qualitative
Microscopic Examination of Urine
 Download http://www.vetlab.com/kova.htm Definition of urine sediment: all solid materials suspended in the urine - a semiquantative evaluation of the urine sediment Significance of formed elements in the
Download http://www.vetlab.com/kova.htm Definition of urine sediment: all solid materials suspended in the urine - a semiquantative evaluation of the urine sediment Significance of formed elements in the
A New Multiplex Real-time PCR Assay For Detection Of Intestinal Parasites
 A New Multiplex Real-time PCR Assay For Detection Of Intestinal Parasites Dr. Andreas Simons Worldwide provider of diagnostic assay solutions Offers a variety of test kit methodologies R-Biopharm Headquarters
A New Multiplex Real-time PCR Assay For Detection Of Intestinal Parasites Dr. Andreas Simons Worldwide provider of diagnostic assay solutions Offers a variety of test kit methodologies R-Biopharm Headquarters
Fecal Me This, Fecal Me That Where Are We Now With Giardia and Cryptosporidium Diagnostics?
 Fecal Me This, Fecal Me That Where Are We Now With Giardia and Cryptosporidium Diagnostics? Shelley Miller, PhD, D(ABMM) Clinical Instructor Dept of Pathology & Lab Medicine UCLA Objectives Discuss giardiasis
Fecal Me This, Fecal Me That Where Are We Now With Giardia and Cryptosporidium Diagnostics? Shelley Miller, PhD, D(ABMM) Clinical Instructor Dept of Pathology & Lab Medicine UCLA Objectives Discuss giardiasis
Instructions. Fuse-It-mRNA easy. Shipping and Storage. Overview. Kit Contents. Specifications. Note: Important Guidelines
 Membrane fusion is a highly efficient method for transfecting various molecules and particles into mammalian cells, even into sensitive and primary cells. The Fuse-It reagents are cargo-specific liposomal
Membrane fusion is a highly efficient method for transfecting various molecules and particles into mammalian cells, even into sensitive and primary cells. The Fuse-It reagents are cargo-specific liposomal
NEW YORK STATE Parasitology Proficiency Testing Program. Parasitology (General) 02 February Sample Preparation and Quality Control
 NEW YORK STATE Parasitology Proficiency Testing Program Parasitology (General) 02 February 2010 The purpose of the New York State Proficiency Testing Program in the category of Parasitology (General) is
NEW YORK STATE Parasitology Proficiency Testing Program Parasitology (General) 02 February 2010 The purpose of the New York State Proficiency Testing Program in the category of Parasitology (General) is
Coral sperm collection for genome sequencing protocol, prepared by Shini Sunagawa. Centrifuge (e.g., Sorvall Biofuge Primo R, Rotor: Heraeus #7588)
 Medina Lab, Penn State University Coral sperm collection for genome sequencing protocol, prepared by Shini Sunagawa Last updated: 09/14/09, Version 1.2 Materials needed: Centrifuge (e.g., Sorvall Biofuge
Medina Lab, Penn State University Coral sperm collection for genome sequencing protocol, prepared by Shini Sunagawa Last updated: 09/14/09, Version 1.2 Materials needed: Centrifuge (e.g., Sorvall Biofuge
Enteric Illness. Shigellosis
 Section 3 Page 1 of 7 Notification Timeline: From Lab/Practitioner to Public Health: Immediately. From Public Health to Saskatchewan Health: Within 72 hours. Public Health Follow-up Timeline: Initiate
Section 3 Page 1 of 7 Notification Timeline: From Lab/Practitioner to Public Health: Immediately. From Public Health to Saskatchewan Health: Within 72 hours. Public Health Follow-up Timeline: Initiate
What is cryptosporidiosis? How is cryptosporidiosis spread?
 What is cryptosporidiosis? Cryptosporidiosis is a diarrheal disease caused by microscopic parasites of the genus Cryptosporidium. Once an animal or person is infected, the parasite lives in the intestine
What is cryptosporidiosis? Cryptosporidiosis is a diarrheal disease caused by microscopic parasites of the genus Cryptosporidium. Once an animal or person is infected, the parasite lives in the intestine
Indian Journal of Basic and Applied Medical Research; September 2014: Vol.-3, Issue- 4, P
 Original article: Comparison of stool concentration methods for detection of prevalence of enteroparasitic infection in rural tertiary care teaching hospital of Maharashtra 1 Dr. Jaishree Puri, 2 Dr. S.
Original article: Comparison of stool concentration methods for detection of prevalence of enteroparasitic infection in rural tertiary care teaching hospital of Maharashtra 1 Dr. Jaishree Puri, 2 Dr. S.
E. Histolytica IgG (Amebiasis)
 DIAGNOSTIC AUTOMATION, INC. 21250 Califa Street, Suite 102 and 116, Woodland hills, CA 91367 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com
DIAGNOSTIC AUTOMATION, INC. 21250 Califa Street, Suite 102 and 116, Woodland hills, CA 91367 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com
Cyclospora Infection among School Children in Kathmandu, Nepal: Prevalence and Associated Risk Factors
 Tropical Medicine and Health Vol. 43 No.4, 2015, 211 216 doi: 10.2149/tmh.2015-25 Copyright 2015 by The Japanese Society of Tropical Medicine 211 Original Papers Cyclospora Infection among School Children
Tropical Medicine and Health Vol. 43 No.4, 2015, 211 216 doi: 10.2149/tmh.2015-25 Copyright 2015 by The Japanese Society of Tropical Medicine 211 Original Papers Cyclospora Infection among School Children
