Supplementary Table 1. Data collection and refinement statistics (molecular replacement).
|
|
|
- Milton Scott
- 5 years ago
- Views:
Transcription
1 Supplementary Table 1. Data collection and refinement statistics (molecular replacement). Data set statistics HLA A*0201- ALWGPDPAAA PPI TCR PPI TCR/A2- ALWGPDPAAA PPI TCR/A2- ALWGPDPAAA Space Group P2 1 C2 1 P1 P2 1 a=53.1, b=81.15, a=192, b=43.26, a=41.48, b=98.44, Unit Cell a=93.68, b=84.59, c=56.55, c=124.71, c=121.30, α=97.27, parameters (Å, ) c=126.28, β=90.03 β= β = β=98.15, γ=93.37 Radiation Source DIAMOND I03 DIAMOND I24 DIAMOND I03 DIAMOND I03 Wavelength (Å) Resolution (Å) ( ) ( ) ( ) ( ) Unique reflections (3594) (2358) (5876) (3894) Completeness (%) 98.1 (94.8) 99.6 (99.8) 96.9 (96.5) 99.4 (99.4) Multiplicity 4.0 (4.0) 3.7 (3.6) 2.1 (2.2) 4.1 (4.2) I/Sigma(I) 16.2 (2.2) 9.3 (1.8) (2.48) 11.1 (2.1) Rmerge (%) 5.0 (57.0) 8.2 (78.2) 5.4 (36.1) 8.5 (70.6) Refinement statistics (highest resolution shell in parenthesis) MR Input Model 2P5E (A,B,C) P1 model 2P5E & 3HG1 P1 model No reflections used No reflections in Rfree set Rcryst (no cutoff) (%) Rfree (%) RMSD from ideal geometry (target values in parenthesis) Bond lengths (Å) (0.021) (0.022) (0.021) Bond Angles ( ) (1.922) (1.954) (1.935) Mean B value (Å 2 ) n/m 52.5 Wilson B-factor (Å 2 ) Outliers Ramachandran plot (%) Overall ESU based on Maximum Likelihood (Å) One crystal was used for data collection. n/m: not measured *Values in parentheses are for highest-resolution shell.
2 Supplementary Figure 1: Flow cytometric analysis of MHCI up-regulation after treatment of islet cells purified from 3 HLA-A*0201+ donors and treated with IL-1, TNF and IFN-. (a-c; organs donors 1-3, respectively) Staining with the PE-conjugated pan-class I mab W6/32 is shown for islet cells before (solid line) and after (dashed line) MHCI upregulation. Supplementary Figure 2. Binding affinity of the 1E6-A2-ALW interaction at 5, 25 and 37 C. Ten serial dilutions of the 1E6 TCR were measured in triplicate at each temperature; representative data from these experiments are plotted. (a-c). The equilibrium binding constant (K D ) values were calculated using a nonlinear curve fit (y= (P 1 x)/p 2 + x)); mean plus SD values are shown. In order to calculate each response, the 1E6 TCR was also injected over a control sample (HLA-A*0201 in complex with ELAGIGILTV peptide) that was deducted from the experimental data (shown in the inset).
3 Supplementary Figure 3: 2Fo-Fc electron density maps for the 1E6-A2-ALW complex, uncomplexed A2-ALW and uncomplexed 1E6 TCR models. All maps shown are within 2Å from the atoms to which they relate. (a) 1E6-A2-ALW at the interface between the TCR (density shown in grey) and the GPD peptide (density shown in blue) motif in copy 1 contoured at 1.5σ. (b) 1E6-A2-ALW at the interface between the TCR (density shown in orange) and the GPD peptide (density shown in blue) motif in copy 2 contoured at 1.5σ. (c) Uncomplexed A2-ALW looking down on top of ALW (density shown in blue) contoured at 1σ. (d) Electron density around the CDR3α and β loops of the uncomplexed 1E6 TCR (density shown in blue) contoured at 1σ.
4 Supplementary Figure 4: Conformation of A2-ALW in complex with the 1E6 TCR versus A2-ALW uncomplexed. (a) Conformation of A2-ALW in complex with the 1E6 TCR (1E6 TCR omitted). (b) Orientation looking from above at the conformation of the bound ALW peptide (yellow sticks) with the MHC shown as surface (grey). (c) Conformation of free A2- ALW. (d) Orientation looking from above at the conformation of the free ALW peptide (red sticks) with the MHC shown as surface (grey). (e) Superposition of the free (red) and complexed (yellow) ALW peptides in the context of HLA A*0201. (f) Orientation looking from above at the conformation of the free (red) and complexed (yellow) ALW peptides superposed. Some small conformational changes could be observed at Pro5, Asp6 and Pro7 (circled). Structure of uncomplexed A2-ALW: Previous investigations have shown that both the peptide 1 and the MHC 2 can undergo conformational changes during TCR binding. The structure of A2-ALW was solved at 1.67Å and refinement gave a model with 100% of residues in the preferred and allowed regions of the Ramachandran plot and geometry consistent with the data resolution (Supplementary Table 1, Supplementary Fig. 2c). The crystallographic R/Rfree factors were 19.5% and 24.4% respectively, and fell in the expected ratio range 3. Superposition of the 1E6 TCR complexed A2-ALW molecule (Supplementary Fig. 3a and Fig. 3b) and the free A2-ALW (Supplementary Fig. 3c and Fig. 3d) models showed that the overall conformations were virtually identical. Some small movements could be observed in the solvent exposed side chains of Pro5, Asp6 and Pro7 (Supplementary Fig. 3e and Fig. 3f), but these relatively minor changes were unlikely to govern TCR binding.
5 Supplementary Figure 5: Comparison of the conformation of the 1E6 TCR CDR1, CDR2 and CDR3 loops in the 1E6-A2-ALW complex versus 1E6 TCR uncomplexed. (a) Superposition of the free (red and cyan cartoon) and complexed (blue and green cartoon) 1E6 TCR. (b) Superposition of the free (red and cyan lines) and complexed (blue and green lines) 1E6 TCR looking down on the peptide (yellow sticks). (c) Superposition of the free (red cartoon) and complexed (blue cartoon) 1E6 TCR CDR1α loop. (d) Superposition of the free (red cartoon) and complexed (blue cartoon) 1E6 TCR CDR2α loop. (e) Superposition of the free (red cartoon) and complexed (blue cartoon) 1E6 TCR CDR3α loop. (f) Superposition of the free (cyan cartoon) and complexed (green cartoon) 1E6 TCR CDR1β loop. (g) Superposition of the free (cyan cartoon) and complexed (green cartoon) 1E6 TCR CDR2β loop. (h) Superposition of the free (cyan cartoon) and complexed (green cartoon) 1E6 TCR CDR3β loop.
6 Structure of the uncomplexed 1E6 TCR: The flexible TCR CDR-loops have been shown to undergo numerous, and sometimes large, conformational changes upon pmhc binding 4. To date, there exist only 5 examples of both free and complexed human TCRs 4, 5 providing a rare insight into the mechanism that governs TCR antigen recognition and cross-reactivity. We solved the structure of the 1E6 TCR at 2.57Å (Supplementary Table 1, Supplementary Fig. 2d). Refinement resulted in a model with 98.2% of residues in the preferred and allowed regions of the Ramachandran plot and geometry consistent with the data resolution. The crystallographic R/Rfree factors were 22.9% and 29.1%, consistent with the expected ratio range 3. Superposition of the free and the bound 1E6 TCR showed that the overall conformations were virtually identical (Supplementary Fig. 4a). Similarly, the overall positions of the TCR CDR-loops were conserved before and after binding to A2-ALW (Supplementary Fig. 4b). Closer inspection of the TCR CDR loops showed that, in accordance with previously published work, the CDR1 and 2 loops exhibited only modest changes in position compared with the CDR3 loops before and after pmhc docking 4. The CDR1 and CDR2 loops underwent a small rigid body shift of 1Å (Supplementary Fig. 4c, Fig. 4d, Fig. 4f and Fig. 4g). Additionally, the CDR2 loops exhibited a small degree of hinge bending. Both of the CDR3 loops displayed a larger degree of conformational change including an average hinge movement of ~1.5Å (Supplementary Fig. 4e and Fig. 4h). These changes were relatively minor compared to other structural studies (JM22 6, 7 exhibited the largest loop movement of 5.6Å). Overall, the 1E6 TCR requires only a moderate level of CDR loop plasticity in order to dock with A2- ALW. References: 1. Tynan, F.E. et al. A T cell receptor flattens a bulged antigenic peptide presented by a major histocompatibility complex class I molecule. Nat Immunol 8, (2007). 2. Borbulevych, O.Y. et al. T cell receptor cross-reactivity directed by antigendependent tuning of peptide-mhc molecular flexibility. Immunity 31, (2009). 3. Tickle, I.J., Laskowski, R.A. & Moss, D.S. Rfree and the rfree ratio. II. Calculation Of the expected values and variances of cross-validation statistics in macromolecular least-squares refinement. Acta Crystallogr D Biol Crystallogr 56, (2000). 4. Armstrong, K.M., Piepenbrink, K.H. & Baker, B.M. Conformational changes and flexibility in T-cell receptor recognition of peptide-mhc complexes. Biochem J 415, (2008). 5. Archbold, J.K. et al. Natural micropolymorphism in human leukocyte antigens provides a basis for genetic control of antigen recognition. J Exp Med 206, (2009). 6. Kjer-Nielsen, L. et al. The 1.5 A crystal structure of a highly selected antiviral T cell receptor provides evidence for a structural basis of immunodominance. Structure (Camb) 10, (2002). 7. Stewart-Jones, G.B., McMichael, A.J., Bell, J.I., Stuart, D.I. & Jones, E.Y. A structural basis for immunodominant human T cell receptor recognition. Nat Immunol 4, (2003).
T Cell Receptor Optimized Peptide Skewing of the T-Cell Repertoire Can Enhance Antigen Targeting
 T Cell Receptor Optimized Peptide Skewing of the T-Cell Repertoire Can Enhance Antigen Targeting Julia Ekeruche-Makinde 1*, Mathew Clement 1*, David K Cole 1*, Emily S J Edwards 1, Kristin Ladell 1, John
T Cell Receptor Optimized Peptide Skewing of the T-Cell Repertoire Can Enhance Antigen Targeting Julia Ekeruche-Makinde 1*, Mathew Clement 1*, David K Cole 1*, Emily S J Edwards 1, Kristin Ladell 1, John
Atypical Natural Killer T-cell receptor recognition of CD1d-lipid antigens supplementary Information.
 Atypical Natural Killer T-cell receptor recognition of CD1d-lipid antigens supplementary Information. Supplementary Figure 1. Phenotypic analysis of TRBV25-1 + and TRBV25-1 - CD1d-α-GalCerreactive cells.
Atypical Natural Killer T-cell receptor recognition of CD1d-lipid antigens supplementary Information. Supplementary Figure 1. Phenotypic analysis of TRBV25-1 + and TRBV25-1 - CD1d-α-GalCerreactive cells.
Table S1. X-ray data collection and refinement statistics
 Table S1. X-ray data collection and refinement statistics Data collection H7.167 Fab-Sh2/H7 complex Beamline SSRL 12-2 Wavelength (Å) 0.97950 Space group I2 1 3 Unit cell parameters (Å, º) a = b = c=207.3,
Table S1. X-ray data collection and refinement statistics Data collection H7.167 Fab-Sh2/H7 complex Beamline SSRL 12-2 Wavelength (Å) 0.97950 Space group I2 1 3 Unit cell parameters (Å, º) a = b = c=207.3,
Detergent solubilised 5 TMD binds pregnanolone at the Q245 neurosteroid potentiation site.
 Supplementary Figure 1 Detergent solubilised 5 TMD binds pregnanolone at the Q245 neurosteroid potentiation site. (a) Gel filtration profiles of purified 5 TMD samples at 100 nm, heated beforehand for
Supplementary Figure 1 Detergent solubilised 5 TMD binds pregnanolone at the Q245 neurosteroid potentiation site. (a) Gel filtration profiles of purified 5 TMD samples at 100 nm, heated beforehand for
Supplementary Figure 1 (previous page). EM analysis of full-length GCGR. (a) Exemplary tilt pair images of the GCGR mab23 complex acquired for Random
 S1 Supplementary Figure 1 (previous page). EM analysis of full-length GCGR. (a) Exemplary tilt pair images of the GCGR mab23 complex acquired for Random Conical Tilt (RCT) reconstruction (left: -50,right:
S1 Supplementary Figure 1 (previous page). EM analysis of full-length GCGR. (a) Exemplary tilt pair images of the GCGR mab23 complex acquired for Random Conical Tilt (RCT) reconstruction (left: -50,right:
FCC2 5CY7 FCC1 5CY8. Actinonin 5CVQ
 Table S1. Data collection and refinement statistics Data collection Apo 5E5D MA 5CPD MAS 5CP0 Actinonin 5CVQ FCC1 5CY8 FCC2 5CY7 FCC 5CWY FCC4 5CVK FCC5 5CWX FCC6 5CVP X-ray source 17A-KEK PAL4A PAL4A
Table S1. Data collection and refinement statistics Data collection Apo 5E5D MA 5CPD MAS 5CP0 Actinonin 5CVQ FCC1 5CY8 FCC2 5CY7 FCC 5CWY FCC4 5CVK FCC5 5CWX FCC6 5CVP X-ray source 17A-KEK PAL4A PAL4A
Supplementary Information Janssen et al.
 Supplementary Information Janssen et al. Insights into complement convertase formation based on the structure of the factor B CVF complex Bert J.C. Janssen 1, Lucio Gomes 1, Roman I. Koning 2, Dmitri I.
Supplementary Information Janssen et al. Insights into complement convertase formation based on the structure of the factor B CVF complex Bert J.C. Janssen 1, Lucio Gomes 1, Roman I. Koning 2, Dmitri I.
Supporting Information
 Supporting Information Guan et al. 10.1073/pnas.1217609110 Fig. S1. Three patterns of reactivity for CD4-induced (CD4i) mabs. The following representative ELISAs show three patterns of reactivity for CD4i
Supporting Information Guan et al. 10.1073/pnas.1217609110 Fig. S1. Three patterns of reactivity for CD4-induced (CD4i) mabs. The following representative ELISAs show three patterns of reactivity for CD4i
Supplementary Information
 Supplementary Information Two common structural motifs for TCR recognition by staphylococcal enterotoxins Karin Erica Johanna Rödström 1, Paulina Regenthal 1, Christopher Bahl 2, Alex Ford 2, David Baker
Supplementary Information Two common structural motifs for TCR recognition by staphylococcal enterotoxins Karin Erica Johanna Rödström 1, Paulina Regenthal 1, Christopher Bahl 2, Alex Ford 2, David Baker
Supporting Information
 Supporting Information McCullough et al. 10.1073/pnas.0801567105 A α10 α8 α9 N α7 α6 α5 C β2 β1 α4 α3 α2 α1 C B N C Fig. S1. ALIX Bro1 in complex with the C-terminal CHMP4A helix. (A) Ribbon diagram showing
Supporting Information McCullough et al. 10.1073/pnas.0801567105 A α10 α8 α9 N α7 α6 α5 C β2 β1 α4 α3 α2 α1 C B N C Fig. S1. ALIX Bro1 in complex with the C-terminal CHMP4A helix. (A) Ribbon diagram showing
SUPPLEMENTARY INFORMATION
 doi:10.1038/nature10913 Supplementary Figure 1 2F o -F c electron density maps of cognate and near-cognate trna Leu 2 in the A site of the 70S ribosome. The maps are contoured at 1.2 sigma and some of
doi:10.1038/nature10913 Supplementary Figure 1 2F o -F c electron density maps of cognate and near-cognate trna Leu 2 in the A site of the 70S ribosome. The maps are contoured at 1.2 sigma and some of
Structural Analysis of TCRpMHC Complexes Using Computational Tools. Feroze Mohideen Briarcliff High School
 Structural Analysis of TCRpMHC Complexes Using Computational Tools Feroze Mohideen Briarcliff High School TCR-pMHC Complexes Peptide Structure of TCR-pMHC complex PDB AO7 Crossreactivity is the ability
Structural Analysis of TCRpMHC Complexes Using Computational Tools Feroze Mohideen Briarcliff High School TCR-pMHC Complexes Peptide Structure of TCR-pMHC complex PDB AO7 Crossreactivity is the ability
(B D) Three views of the final refined 2Fo-Fc electron density map of the Vpr (red)-ung2 (green) interacting region, contoured at 1.4σ.
 Supplementary Figure 1 Overall structure of the DDB1 DCAF1 Vpr UNG2 complex. (A) The final refined 2Fo-Fc electron density map, contoured at 1.4σ of Vpr, illustrating well-defined side chains. (B D) Three
Supplementary Figure 1 Overall structure of the DDB1 DCAF1 Vpr UNG2 complex. (A) The final refined 2Fo-Fc electron density map, contoured at 1.4σ of Vpr, illustrating well-defined side chains. (B D) Three
Nature Structural & Molecular Biology: doi: /nsmb Supplementary Figure 1
 Supplementary Figure 1 The UBL and RING1 interface remains associated in the complex structures of Parkin and pub. a) Asymmetric Unit of crystal structure of UBLR0RBR and pub complex showing UBL (green),
Supplementary Figure 1 The UBL and RING1 interface remains associated in the complex structures of Parkin and pub. a) Asymmetric Unit of crystal structure of UBLR0RBR and pub complex showing UBL (green),
Supporting Information
 Supporting Information Mechanism of inactivation of -aminobutyric acid aminotransferase by (1S,3S)-3-amino-4-difluoromethylenyl-1- cyclopentanoic acid (CPP-115) Hyunbeom Lee, 1, Emma H. Doud, 1,2 Rui Wu,
Supporting Information Mechanism of inactivation of -aminobutyric acid aminotransferase by (1S,3S)-3-amino-4-difluoromethylenyl-1- cyclopentanoic acid (CPP-115) Hyunbeom Lee, 1, Emma H. Doud, 1,2 Rui Wu,
Transient β-hairpin Formation in α-synuclein Monomer Revealed by Coarse-grained Molecular Dynamics Simulation
 Transient β-hairpin Formation in α-synuclein Monomer Revealed by Coarse-grained Molecular Dynamics Simulation Hang Yu, 1, 2, a) Wei Han, 1, 3, b) Wen Ma, 1, 2 1, 2, 3, c) and Klaus Schulten 1) Beckman
Transient β-hairpin Formation in α-synuclein Monomer Revealed by Coarse-grained Molecular Dynamics Simulation Hang Yu, 1, 2, a) Wei Han, 1, 3, b) Wen Ma, 1, 2 1, 2, 3, c) and Klaus Schulten 1) Beckman
Supplementary Material
 Supplementary Material Materials and methods Enzyme assay The enzymatic activity of -glucosidase toward salicin was measured with the Miller method (Miller, 1959) using glucose as the standard. A total
Supplementary Material Materials and methods Enzyme assay The enzymatic activity of -glucosidase toward salicin was measured with the Miller method (Miller, 1959) using glucose as the standard. A total
Supplementary Materials for
 www.sciencemag.org/cgi/content/full/science.aal4326/dc1 Supplementary Materials for Structure of a eukaryotic voltage-gated sodium channel at near-atomic resolution Huaizong Shen, Qiang Zhou, Xiaojing
www.sciencemag.org/cgi/content/full/science.aal4326/dc1 Supplementary Materials for Structure of a eukaryotic voltage-gated sodium channel at near-atomic resolution Huaizong Shen, Qiang Zhou, Xiaojing
SUPPLEMENTARY INFORMATION
 doi: 10.1038/nature07422 SUPPLEMENTARY INFRMATIN K S(P) R S I M Q(L4) R M 7 6 Sp Q I K R 5 4 3 2 1 L(L0) L L S E 0 +1 +2 +3 Figure S1a Difference electron density (mfo DFc) for the peptide (Qpeptide),
doi: 10.1038/nature07422 SUPPLEMENTARY INFRMATIN K S(P) R S I M Q(L4) R M 7 6 Sp Q I K R 5 4 3 2 1 L(L0) L L S E 0 +1 +2 +3 Figure S1a Difference electron density (mfo DFc) for the peptide (Qpeptide),
Surface plasmon resonance (SPR) analysis
 Surface plasmon resonance (SPR) analysis Soluble CD8αα and was manufactured as described previously. 1,2 The C12C heterodimeric TCR was produced using an engineered disulfide bridge between the constant
Surface plasmon resonance (SPR) analysis Soluble CD8αα and was manufactured as described previously. 1,2 The C12C heterodimeric TCR was produced using an engineered disulfide bridge between the constant
SUPPLEMENTARY INFORMATION (SI) FIGURES AND TABLES
 SUPPLEMENTARY INFORMATION (SI) FIGURES AND TABLES 1 Title: Discovery of a junctional epitope antibody that stabilizes IL-6 and gp80 protein:protein interaction and modulates its downstream signaling Authors:
SUPPLEMENTARY INFORMATION (SI) FIGURES AND TABLES 1 Title: Discovery of a junctional epitope antibody that stabilizes IL-6 and gp80 protein:protein interaction and modulates its downstream signaling Authors:
Nature Immunology: doi: /ni Supplementary Figure 1
 Supplementary Figure 1 A β-strand positions consistently places the residues at CDR3β P6 and P7 within human and mouse TCR-peptide-MHC interfaces. (a) E8 TCR containing V β 13*06 carrying with an 11mer
Supplementary Figure 1 A β-strand positions consistently places the residues at CDR3β P6 and P7 within human and mouse TCR-peptide-MHC interfaces. (a) E8 TCR containing V β 13*06 carrying with an 11mer
Figure S1. HP1α localizes to centromeres in mitosis and interacts with INCENP. (A&B) HeLa
 SUPPLEMENTARY FIGURES Figure S1. HP1α localizes to centromeres in mitosis and interacts with INCENP. (A&B) HeLa tet-on cells that stably express HP1α-CFP, HP1β-CFP, or HP1γ-CFP were monitored with livecell
SUPPLEMENTARY FIGURES Figure S1. HP1α localizes to centromeres in mitosis and interacts with INCENP. (A&B) HeLa tet-on cells that stably express HP1α-CFP, HP1β-CFP, or HP1γ-CFP were monitored with livecell
SUPPLEMENTARY INFORMATION FOR. (R)-Profens Are Substrate-Selective Inhibitors of Endocannabinoid Oxygenation. by COX-2
 SUPPLEMENTARY INFORMATION FOR (R)-Profens Are Substrate-Selective Inhibitors of Endocannabinoid Oxygenation by COX-2 Kelsey C. Duggan, Daniel J. Hermanson, Joel Musee, Jeffery J. Prusakiewicz, Jami L.
SUPPLEMENTARY INFORMATION FOR (R)-Profens Are Substrate-Selective Inhibitors of Endocannabinoid Oxygenation by COX-2 Kelsey C. Duggan, Daniel J. Hermanson, Joel Musee, Jeffery J. Prusakiewicz, Jami L.
Supplementary Figure 1 Preparation, crystallization and structure determination of EpEX. (a), Purified EpEX and EpEX analyzed on homogenous 12.
 Supplementary Figure 1 Preparation, crystallization and structure determination of EpEX. (a), Purified EpEX and EpEX analyzed on homogenous 12.5 % SDS-PAGE gel under reducing and non-reducing conditions.
Supplementary Figure 1 Preparation, crystallization and structure determination of EpEX. (a), Purified EpEX and EpEX analyzed on homogenous 12.5 % SDS-PAGE gel under reducing and non-reducing conditions.
Supplementary Materials for
 advances.sciencemag.org/cgi/content/full/4/3/eaaq0762/dc1 Supplementary Materials for Structures of monomeric and oligomeric forms of the Toxoplasma gondii perforin-like protein 1 Tao Ni, Sophie I. Williams,
advances.sciencemag.org/cgi/content/full/4/3/eaaq0762/dc1 Supplementary Materials for Structures of monomeric and oligomeric forms of the Toxoplasma gondii perforin-like protein 1 Tao Ni, Sophie I. Williams,
Supplementary Figures
 Supplementary Figures Supplementary Figure 1: Predicted structure of the most stable {110} antiphase boundary defect in magnetite (model APB-I). a) The same structure as that shown in Fig. 1b (main text)
Supplementary Figures Supplementary Figure 1: Predicted structure of the most stable {110} antiphase boundary defect in magnetite (model APB-I). a) The same structure as that shown in Fig. 1b (main text)
Supplementary Table 1. Functional avidities of survivin-specific T-cell clones against LML-peptide
 Supplementary Table 1. Functional avidities of survivin-specific T-cell clones against LML-peptide pulsed T2 cells. clone avidity by 4-hour 51 Cr-release assay 50% lysis at E:T 10:1 [LML peptide, M] #24
Supplementary Table 1. Functional avidities of survivin-specific T-cell clones against LML-peptide pulsed T2 cells. clone avidity by 4-hour 51 Cr-release assay 50% lysis at E:T 10:1 [LML peptide, M] #24
Structure of the measles virus hemagglutinin bound to the CD46 receptor. César Santiago, María L. Celma, Thilo Stehle and José M.
 Supporting Figures and Table for Structure of the measles virus hemagglutinin bound to the CD46 receptor César Santiago, María L. Celma, Thilo Stehle and José M. Casasnovas This PDF file includes: Supplementary
Supporting Figures and Table for Structure of the measles virus hemagglutinin bound to the CD46 receptor César Santiago, María L. Celma, Thilo Stehle and José M. Casasnovas This PDF file includes: Supplementary
SUPPLEMENTAL MATERIAL. UNC119 is required for G protein trafficking in sensory neurons
 1 SUPPLEMENTAL MATERIAL UNC119 is required for G protein trafficking in sensory neurons Houbin Zhang, Ryan N. Constantine, Sergey Vorobiev, Yang Chen, Jayaraman Seetharaman, Yuanpeng Janet Huang, Rong
1 SUPPLEMENTAL MATERIAL UNC119 is required for G protein trafficking in sensory neurons Houbin Zhang, Ryan N. Constantine, Sergey Vorobiev, Yang Chen, Jayaraman Seetharaman, Yuanpeng Janet Huang, Rong
A rt i c l e s Nature America, Inc. All rights reserved.
 Structural basis for the killing of human beta cells by CD8 + T cells in type 1 diabetes Anna M Bulek 1,9, David K Cole 1,9, Ania Skowera 2,3,9, Garry Dolton 1, Stephanie Gras 4, Florian Madura 1, Anna
Structural basis for the killing of human beta cells by CD8 + T cells in type 1 diabetes Anna M Bulek 1,9, David K Cole 1,9, Ania Skowera 2,3,9, Garry Dolton 1, Stephanie Gras 4, Florian Madura 1, Anna
Supplementary Figure 1. Using DNA barcode-labeled MHC multimers to generate TCR fingerprints
 Supplementary Figure 1 Using DNA barcode-labeled MHC multimers to generate TCR fingerprints (a) Schematic overview of the workflow behind a TCR fingerprint. Each peptide position of the original peptide
Supplementary Figure 1 Using DNA barcode-labeled MHC multimers to generate TCR fingerprints (a) Schematic overview of the workflow behind a TCR fingerprint. Each peptide position of the original peptide
SUPPLEMENTARY INFORMATION. Computational Assay of H7N9 Influenza Neuraminidase Reveals R292K Mutation Reduces Drug Binding Affinity
 SUPPLEMENTARY INFORMATION Computational Assay of H7N9 Influenza Neuraminidase Reveals R292K Mutation Reduces Drug Binding Affinity Christopher Woods 1, Maturos Malaisree 1, Ben Long 2, Simon McIntosh-Smith
SUPPLEMENTARY INFORMATION Computational Assay of H7N9 Influenza Neuraminidase Reveals R292K Mutation Reduces Drug Binding Affinity Christopher Woods 1, Maturos Malaisree 1, Ben Long 2, Simon McIntosh-Smith
Protein Secondary Structure
 Protein Secondary Structure Reading: Berg, Tymoczko & Stryer, 6th ed., Chapter 2, pp. 37-45 Problems in textbook: chapter 2, pp. 63-64, #1,5,9 Directory of Jmol structures of proteins: http://www.biochem.arizona.edu/classes/bioc462/462a/jmol/routines/routines.html
Protein Secondary Structure Reading: Berg, Tymoczko & Stryer, 6th ed., Chapter 2, pp. 37-45 Problems in textbook: chapter 2, pp. 63-64, #1,5,9 Directory of Jmol structures of proteins: http://www.biochem.arizona.edu/classes/bioc462/462a/jmol/routines/routines.html
Supplementary Information
 Supplementary Information Structural Basis of Sialidase in Complex with Geranylated Flavonoids as Potent Natural Inhibitors Youngjin Lee, a,b Young Bae Ryu, d Hyung-Seop Youn, a,b Jung Keun Cho, e Young
Supplementary Information Structural Basis of Sialidase in Complex with Geranylated Flavonoids as Potent Natural Inhibitors Youngjin Lee, a,b Young Bae Ryu, d Hyung-Seop Youn, a,b Jung Keun Cho, e Young
Chapter 6. X-ray structure analysis of D30N tethered HIV-1 protease. dimer/saquinavir complex
 Chapter 6 X-ray structure analysis of D30N tethered HIV-1 protease dimer/saquinavir complex 6.1 Introduction: The arrival of HIV protease inhibitors (PIs) in late 1995 marked the beginning of an important
Chapter 6 X-ray structure analysis of D30N tethered HIV-1 protease dimer/saquinavir complex 6.1 Introduction: The arrival of HIV protease inhibitors (PIs) in late 1995 marked the beginning of an important
Crystal structure of the neutralizing antibody HK20 in complex with its gp41 antigen
 Crystal structure of the neutralizing antibody HK20 in complex with its gp41 antigen David Lutje Hulsik Unit for Virus Host Cell Interaction UMI 3265 University Joseph Fourier-EMBL-CNRS, Grenoble Env catalyzed
Crystal structure of the neutralizing antibody HK20 in complex with its gp41 antigen David Lutje Hulsik Unit for Virus Host Cell Interaction UMI 3265 University Joseph Fourier-EMBL-CNRS, Grenoble Env catalyzed
Data are contained in multiple tabs in Excel spreadsheets and in CSV files.
 Contents Overview Curves Methods Measuring enzymatic activity (figure 2) Enzyme characterisation (Figure S1, S2) Enzyme kinetics (Table 3) Effect of ph on activity (figure 3B) Effect of metals and inhibitors
Contents Overview Curves Methods Measuring enzymatic activity (figure 2) Enzyme characterisation (Figure S1, S2) Enzyme kinetics (Table 3) Effect of ph on activity (figure 3B) Effect of metals and inhibitors
Chapter VI. Increased affinity in the complex of yeast cytochrome c and cytochrome c peroxidase
 Chapter VI Increased affinity in the complex of yeast cytochrome c and cytochrome c peroxidase Chapter VI Abstract We report the study of T12A Cyt c CcP complex using isothermal titration calorimetry (ITC),
Chapter VI Increased affinity in the complex of yeast cytochrome c and cytochrome c peroxidase Chapter VI Abstract We report the study of T12A Cyt c CcP complex using isothermal titration calorimetry (ITC),
Nature Structural and Molecular Biology: doi: /nsmb Supplementary Figure 1
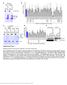 Supplementary Figure 1 Mutational analysis of the SA2-Scc1 interaction in vitro and in human cells. (a) Autoradiograph (top) and Coomassie stained gel (bottom) of 35 S-labeled Myc-SA2 proteins (input)
Supplementary Figure 1 Mutational analysis of the SA2-Scc1 interaction in vitro and in human cells. (a) Autoradiograph (top) and Coomassie stained gel (bottom) of 35 S-labeled Myc-SA2 proteins (input)
List of Figures. List of Tables
 Supporting Information for: Signaling Domain of Sonic Hedgehog as Cannibalistic Calcium-Regulated Zinc-Peptidase Rocio Rebollido-Rios 1, Shyam Bandari 3, Christoph Wilms 1, Stanislav Jakuschev 1, Andrea
Supporting Information for: Signaling Domain of Sonic Hedgehog as Cannibalistic Calcium-Regulated Zinc-Peptidase Rocio Rebollido-Rios 1, Shyam Bandari 3, Christoph Wilms 1, Stanislav Jakuschev 1, Andrea
Supplementary Materials for
 advances.sciencemag.org/cgi/content/full/2/9/e1600292/dc1 Supplementary Materials for Native phasing of x-ray free-electron laser data for a G protein coupled receptor Alexander Batyuk, Lorenzo Galli,
advances.sciencemag.org/cgi/content/full/2/9/e1600292/dc1 Supplementary Materials for Native phasing of x-ray free-electron laser data for a G protein coupled receptor Alexander Batyuk, Lorenzo Galli,
Antigen Receptor Structures October 14, Ram Savan
 Antigen Receptor Structures October 14, 2016 Ram Savan savanram@uw.edu 441 Lecture #8 Slide 1 of 28 Three lectures on antigen receptors Part 1 (Today): Structural features of the BCR and TCR Janeway Chapter
Antigen Receptor Structures October 14, 2016 Ram Savan savanram@uw.edu 441 Lecture #8 Slide 1 of 28 Three lectures on antigen receptors Part 1 (Today): Structural features of the BCR and TCR Janeway Chapter
KDM2A. Reactions. containing. Reactions
 Supplementary Figure 1 KDM2A catalyses only lysine demethylation.. MALDI-TOF MS of demethylation of the shown variant histone peptides as catalysed by recombinant KDM2A. Reactions containing enzyme are
Supplementary Figure 1 KDM2A catalyses only lysine demethylation.. MALDI-TOF MS of demethylation of the shown variant histone peptides as catalysed by recombinant KDM2A. Reactions containing enzyme are
and Jamie Rossjohn 1,2,4 *
 Understanding the drivers of MHC restriction of T cell receptors Nicole L. La Gruta 1,2 *, Stephanie Gras 1,2, Stephen R. Daley 1, Paul G. Thomas 3 and Jamie Rossjohn 1,2,4 * Abstract T cell discrimination
Understanding the drivers of MHC restriction of T cell receptors Nicole L. La Gruta 1,2 *, Stephanie Gras 1,2, Stephen R. Daley 1, Paul G. Thomas 3 and Jamie Rossjohn 1,2,4 * Abstract T cell discrimination
Supporting Information Identification of Amino Acids with Sensitive Nanoporous MoS 2 : Towards Machine Learning-Based Prediction
 Supporting Information Identification of Amino Acids with Sensitive Nanoporous MoS 2 : Towards Machine Learning-Based Prediction Amir Barati Farimani, Mohammad Heiranian, Narayana R. Aluru 1 Department
Supporting Information Identification of Amino Acids with Sensitive Nanoporous MoS 2 : Towards Machine Learning-Based Prediction Amir Barati Farimani, Mohammad Heiranian, Narayana R. Aluru 1 Department
Supplementary Materials for
 advances.sciencemag.org/cgi/content/full/2/4/e1500980/dc1 Supplementary Materials for The crystal structure of human dopamine -hydroxylase at 2.9 Å resolution Trine V. Vendelboe, Pernille Harris, Yuguang
advances.sciencemag.org/cgi/content/full/2/4/e1500980/dc1 Supplementary Materials for The crystal structure of human dopamine -hydroxylase at 2.9 Å resolution Trine V. Vendelboe, Pernille Harris, Yuguang
How much can a T-cell antigen receptor adapt to structurally distinct antigenic peptides?
 The EMBO Journal (2007) 26, 1972 1983 & 2007 European Molecular Biology Organization All Rights Reserved 0261-4189/07 www.embojournal.org How much can a T-cell antigen receptor adapt to structurally distinct
The EMBO Journal (2007) 26, 1972 1983 & 2007 European Molecular Biology Organization All Rights Reserved 0261-4189/07 www.embojournal.org How much can a T-cell antigen receptor adapt to structurally distinct
Immune surveillance: The immune system can recognize and destroy nascent malignant cells
 Immune surveillance: The immune system can recognize and destroy nascent malignant cells Control Escape APC T C T H B NKT NK Innate Tumor T cells are believed to play a major role in controlling tumor
Immune surveillance: The immune system can recognize and destroy nascent malignant cells Control Escape APC T C T H B NKT NK Innate Tumor T cells are believed to play a major role in controlling tumor
Nature Structural & Molecular Biology: doi: /nsmb.1933
 The structural basis of open channel block in a prokaryotic pentameric ligand-gated ion channel Ricarda J. C. Hilf, Carlo Bertozzi, Iwan Zimmermann, Alwin Reiter, Dirk Trauner and Raimund Dutzler a GLIC
The structural basis of open channel block in a prokaryotic pentameric ligand-gated ion channel Ricarda J. C. Hilf, Carlo Bertozzi, Iwan Zimmermann, Alwin Reiter, Dirk Trauner and Raimund Dutzler a GLIC
This exam consists of two parts. Part I is multiple choice. Each of these 25 questions is worth 2 points.
 MBB 407/511 Molecular Biology and Biochemistry First Examination - October 1, 2002 Name Social Security Number This exam consists of two parts. Part I is multiple choice. Each of these 25 questions is
MBB 407/511 Molecular Biology and Biochemistry First Examination - October 1, 2002 Name Social Security Number This exam consists of two parts. Part I is multiple choice. Each of these 25 questions is
Supplementary Data 1. Alanine substitutions and position variants of APNCYGNIPL. Applied in
 Supplementary Data 1. Alanine substitutions and position variants of APNCYGNIPL. Applied in Supplementary Fig. 2 Substitution Sequence Position variant Sequence original APNCYGNIPL original APNCYGNIPL
Supplementary Data 1. Alanine substitutions and position variants of APNCYGNIPL. Applied in Supplementary Fig. 2 Substitution Sequence Position variant Sequence original APNCYGNIPL original APNCYGNIPL
T cell Receptor. Chapter 9. Comparison of TCR αβ T cells
 Chapter 9 The αβ TCR is similar in size and structure to an antibody Fab fragment T cell Receptor Kuby Figure 9-3 The αβ T cell receptor - Two chains - α and β - Two domains per chain - constant (C) domain
Chapter 9 The αβ TCR is similar in size and structure to an antibody Fab fragment T cell Receptor Kuby Figure 9-3 The αβ T cell receptor - Two chains - α and β - Two domains per chain - constant (C) domain
Nature Immunology: doi: /ni Supplementary Figure 1. Cytokine pattern in skin in response to urushiol.
 Supplementary Figure 1 Cytokine pattern in skin in response to urushiol. Wild-type (WT) and CD1a-tg mice (n = 3 per group) were sensitized and challenged with urushiol (uru) or vehicle (veh). Quantitative
Supplementary Figure 1 Cytokine pattern in skin in response to urushiol. Wild-type (WT) and CD1a-tg mice (n = 3 per group) were sensitized and challenged with urushiol (uru) or vehicle (veh). Quantitative
Crystallization-grade After D After V3 cocktail. Time (s) Time (s) Time (s) Time (s) Time (s) Time (s)
 Ligand Type Name 6 Crystallization-grade After 447-52D After V3 cocktail Receptor CD4 Resonance Units 5 1 5 1 5 1 Broadly neutralizing antibodies 2G12 VRC26.9 Resonance Units Resonance Units 3 1 15 1 5
Ligand Type Name 6 Crystallization-grade After 447-52D After V3 cocktail Receptor CD4 Resonance Units 5 1 5 1 5 1 Broadly neutralizing antibodies 2G12 VRC26.9 Resonance Units Resonance Units 3 1 15 1 5
Structure and functional analysis of the IGF-II/IGF2R interaction
 Structure and functional analysis of the IGF-II/IGF2R interaction James Brown 1, Carlie Delaine 2, Oliver J. Zaccheo 3, Christian Siebold 1, Robert J. Gilbert 1, Gijs van Boxel 1, Adam Denley 2, John C.
Structure and functional analysis of the IGF-II/IGF2R interaction James Brown 1, Carlie Delaine 2, Oliver J. Zaccheo 3, Christian Siebold 1, Robert J. Gilbert 1, Gijs van Boxel 1, Adam Denley 2, John C.
Antigen Presentation to T lymphocytes
 Antigen Presentation to T lymphocytes Immunology 441 Lectures 6 & 7 Chapter 6 October 10 & 12, 2016 Jessica Hamerman jhamerman@benaroyaresearch.org Office hours by arrangement Antigen processing: How are
Antigen Presentation to T lymphocytes Immunology 441 Lectures 6 & 7 Chapter 6 October 10 & 12, 2016 Jessica Hamerman jhamerman@benaroyaresearch.org Office hours by arrangement Antigen processing: How are
Supplementary Figure S1. Supplementary Figure S1. Chemical structure of CH
 Supplementary Figure S1 Supplementary Figure S1. Chemical structure of CH7057288. Supplementary Figure S2 Kinase % competition TRKA 99 TRKC 95 TRKB 92 MEK5 87 AXL 84 CLK1 82 MERTK 82 GCN2 (Kin.Dom.2,S808G)
Supplementary Figure S1 Supplementary Figure S1. Chemical structure of CH7057288. Supplementary Figure S2 Kinase % competition TRKA 99 TRKC 95 TRKB 92 MEK5 87 AXL 84 CLK1 82 MERTK 82 GCN2 (Kin.Dom.2,S808G)
ATP-independent reversal of a membrane protein aggregate by a chloroplast SRP
 ATP-independent reversal of a membrane protein aggregate by a chloroplast SRP Peera Jaru-Ampornpan 1, Kuang Shen 1,3, Vinh Q. Lam 1,3, Mona Ali 2, Sebastian Doniach 2, Tony Z. Jia 1, Shu-ou Shan 1 Supplementary
ATP-independent reversal of a membrane protein aggregate by a chloroplast SRP Peera Jaru-Ampornpan 1, Kuang Shen 1,3, Vinh Q. Lam 1,3, Mona Ali 2, Sebastian Doniach 2, Tony Z. Jia 1, Shu-ou Shan 1 Supplementary
Supplementary Figure 1 Overall structure of the transmembrane pore of FraC. a, Crystal packing of the pore along the y-z, b, the z-x, and c, the y-x
 Supplementary Figure 1 Overall structure of the transmembrane pore of FraC. a, Crystal packing of the pore along the y-z, b, the z-x, and c, the y-x planes. The N-terminal and the β-core regions are depicted
Supplementary Figure 1 Overall structure of the transmembrane pore of FraC. a, Crystal packing of the pore along the y-z, b, the z-x, and c, the y-x planes. The N-terminal and the β-core regions are depicted
NIH Public Access Author Manuscript J Am Chem Soc. Author manuscript; available in PMC 2008 September 29.
 NIH Public Access Author Manuscript Published in final edited form as: J Am Chem Soc. 2006 March 8; 128(9): 2812 2813. doi:10.1021/ja058211x. HIV-1 protease flaps spontaneously close to the correct structure
NIH Public Access Author Manuscript Published in final edited form as: J Am Chem Soc. 2006 March 8; 128(9): 2812 2813. doi:10.1021/ja058211x. HIV-1 protease flaps spontaneously close to the correct structure
SUPPORTING INFORMATION FOR. A Computational Approach to Enzyme Design: Using Docking and MM- GBSA Scoring
 SUPPRTING INFRMATIN FR A Computational Approach to Enzyme Design: Predicting ω- Aminotransferase Catalytic Activity Using Docking and MM- GBSA Scoring Sarah Sirin, 1 Rajesh Kumar, 2 Carlos Martinez, 2
SUPPRTING INFRMATIN FR A Computational Approach to Enzyme Design: Predicting ω- Aminotransferase Catalytic Activity Using Docking and MM- GBSA Scoring Sarah Sirin, 1 Rajesh Kumar, 2 Carlos Martinez, 2
Modeling holo-acp:dh and holo-acp:kr complexes of modular polyketide synthases: a docking and molecular dynamics study
 Anand and Mohanty BMC Structural Biology 2012, 12:10 RESEARCH ARTICLE Open Access Modeling holo-acp:dh and holo-acp:kr complexes of modular polyketide synthases: a docking and molecular dynamics study
Anand and Mohanty BMC Structural Biology 2012, 12:10 RESEARCH ARTICLE Open Access Modeling holo-acp:dh and holo-acp:kr complexes of modular polyketide synthases: a docking and molecular dynamics study
Supplementary Materials. High affinity binding of phosphatidylinositol-4-phosphate. by Legionella pneumophila DrrA
 Supplementary Materials High affinity binding of phosphatidylinositol-4-phosphate by Legionella pneumophila DrrA Running title: Molecular basis of PtdIns(4)P-binding by DrrA Stefan Schoebel, Wulf Blankenfeldt,
Supplementary Materials High affinity binding of phosphatidylinositol-4-phosphate by Legionella pneumophila DrrA Running title: Molecular basis of PtdIns(4)P-binding by DrrA Stefan Schoebel, Wulf Blankenfeldt,
SUPPLEMENTARY INFORMATION
 Supplementary discussion of the role of C05 HCDR1 in antigen binding. Deletion of the 5 amino-acid insertion in HCDR1 del(fgest) and scanning mutagenesis of the tip of the loop had differing effects on
Supplementary discussion of the role of C05 HCDR1 in antigen binding. Deletion of the 5 amino-acid insertion in HCDR1 del(fgest) and scanning mutagenesis of the tip of the loop had differing effects on
Rapid Characterization of Allosteric Networks. with Ensemble Normal Mode Analysis
 Supporting Information Rapid Characterization of Allosteric Networks with Ensemble Normal Mode Analysis Xin-Qiu Yao 1, Lars Skjærven 2, and Barry J. Grant 1* 1 Department of Computational Medicine and
Supporting Information Rapid Characterization of Allosteric Networks with Ensemble Normal Mode Analysis Xin-Qiu Yao 1, Lars Skjærven 2, and Barry J. Grant 1* 1 Department of Computational Medicine and
MHC class I MHC class II Structure of MHC antigens:
 MHC class I MHC class II Structure of MHC antigens: MHC class I antigens consist of a transmembrane heavy chain (α chain) that is non-covalently associated with β2- microglobulin. Membrane proximal domain
MHC class I MHC class II Structure of MHC antigens: MHC class I antigens consist of a transmembrane heavy chain (α chain) that is non-covalently associated with β2- microglobulin. Membrane proximal domain
Excerpt from J. Mol. Biol. (2002) 320, :
 Excerpt from J. Mol. Biol. (2002) 320, 1095 1108: Crystal Structure of the Ternary Complex of the Catalytic Domain of Human Phenylalanine Hydroxylase with Tetrahydrobiopterin and 3-(2-Thienyl)-L-alanine,
Excerpt from J. Mol. Biol. (2002) 320, 1095 1108: Crystal Structure of the Ternary Complex of the Catalytic Domain of Human Phenylalanine Hydroxylase with Tetrahydrobiopterin and 3-(2-Thienyl)-L-alanine,
Antigen Recognition by T cells
 Antigen Recognition by T cells TCR only recognize foreign Ags displayed on cell surface These Ags can derive from pathogens, which replicate within cells or from pathogens or their products that cells
Antigen Recognition by T cells TCR only recognize foreign Ags displayed on cell surface These Ags can derive from pathogens, which replicate within cells or from pathogens or their products that cells
Characterizing the mesophase behavior of hydrated 9.9 MAG at 20 C and at increasing concentrations of DDM by SAXS.
 Supplementary Figure 1 Characterizing the mesophase behavior of hydrated 9.9 MAG at 20 C and at increasing concentrations of DDM by SAXS. Individual panels show I/2 SAXS profiles at the specified concentration
Supplementary Figure 1 Characterizing the mesophase behavior of hydrated 9.9 MAG at 20 C and at increasing concentrations of DDM by SAXS. Individual panels show I/2 SAXS profiles at the specified concentration
Thermal shift binding experiments were carried out using Thermofluor 384 ELS system. Protein
 Supplementary Methods Thermal shift assays Thermal shift binding experiments were carried out using Thermofluor 384 ELS system. Protein unfolding was examined by monitoring the fluorescence of ANS (1-anilinonaphthalene-8-
Supplementary Methods Thermal shift assays Thermal shift binding experiments were carried out using Thermofluor 384 ELS system. Protein unfolding was examined by monitoring the fluorescence of ANS (1-anilinonaphthalene-8-
Supplementary Material to Oxidative properties of FeO2+: electronic structure and solvation effects.
 Supplementary Material to Oxidative properties of FeO2+: electronic structure and solvation effects. Manuel J. Louwerse and Evert Jan Baerends Theoretical Chemistry, Vrije Universiteit Amsterdam, De Boelelaan
Supplementary Material to Oxidative properties of FeO2+: electronic structure and solvation effects. Manuel J. Louwerse and Evert Jan Baerends Theoretical Chemistry, Vrije Universiteit Amsterdam, De Boelelaan
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION doi:10.1038/nature22394 Supplementary Table 1 Observed intermolecular interactions within the GLP-1:GLP-1R TMD interface. Superscripts refer to the Wootten residue numbering system
SUPPLEMENTARY INFORMATION doi:10.1038/nature22394 Supplementary Table 1 Observed intermolecular interactions within the GLP-1:GLP-1R TMD interface. Superscripts refer to the Wootten residue numbering system
CPM (x 10-3 ) Tregs +Teffs. Tregs alone ICOS CLTA-4
 A 2,5 B 4 Number of cells (x 1-6 ) 2, 1,5 1, 5 CPM (x 1-3 ) 3 2 1 5 1 15 2 25 3 Days of culture 1/1 1/2 1/4 1/8 1/16 1/32 Treg/Teff ratio C alone alone alone alone CD25 FoxP3 GITR CD44 ICOS CLTA-4 CD127
A 2,5 B 4 Number of cells (x 1-6 ) 2, 1,5 1, 5 CPM (x 1-3 ) 3 2 1 5 1 15 2 25 3 Days of culture 1/1 1/2 1/4 1/8 1/16 1/32 Treg/Teff ratio C alone alone alone alone CD25 FoxP3 GITR CD44 ICOS CLTA-4 CD127
The structural organization of substrate loading in iterative polyketide synthases
 SUPPLEMENTARY INFORMATION Articles https://doi.org/10.1038/s41589-018-0026-3 In the format provided by the authors and unedited. The structural organization of substrate loading in iterative polyketide
SUPPLEMENTARY INFORMATION Articles https://doi.org/10.1038/s41589-018-0026-3 In the format provided by the authors and unedited. The structural organization of substrate loading in iterative polyketide
Table S1: Kinetic parameters of drug and substrate binding to wild type and HIV-1 protease variants. Data adapted from Ref. 6 in main text.
 Dynamical Network of HIV-1 Protease Mutants Reveals the Mechanism of Drug Resistance and Unhindered Activity Rajeswari Appadurai and Sanjib Senapati* BJM School of Biosciences and Department of Biotechnology,
Dynamical Network of HIV-1 Protease Mutants Reveals the Mechanism of Drug Resistance and Unhindered Activity Rajeswari Appadurai and Sanjib Senapati* BJM School of Biosciences and Department of Biotechnology,
Supplementary Figure 1. Heavy chain sequences of 2G1 and 8M2 aligned with V H 1-69
 Supplementary Figure 1. Heavy chain sequences of 2G1 and 8M2 aligned with V H 1-69 germline gene sequence. 2G1 and 8M2 acquired 14 and 18 mutations from the germline gene sequence, respectively. In 2G1,
Supplementary Figure 1. Heavy chain sequences of 2G1 and 8M2 aligned with V H 1-69 germline gene sequence. 2G1 and 8M2 acquired 14 and 18 mutations from the germline gene sequence, respectively. In 2G1,
The role of Ca² + ions in the complex assembling of protein Z and Z-dependent protease inhibitor: A structure and dynamics investigation
 www.bioinformation.net Hypothesis Volume 8(9) The role of Ca² + ions in the complex assembling of protein Z and Z-dependent protease inhibitor: A structure and dynamics investigation Zahra Karimi 1 *,
www.bioinformation.net Hypothesis Volume 8(9) The role of Ca² + ions in the complex assembling of protein Z and Z-dependent protease inhibitor: A structure and dynamics investigation Zahra Karimi 1 *,
obtained for the simulations of the E2 conformation of SERCA in a pure POPC lipid bilayer (blue) and in a
 Supplementary Figure S1. Distribution of atoms along the bilayer normal. Normalized density profiles obtained for the simulations of the E2 conformation of SERCA in a pure POPC lipid bilayer (blue) and
Supplementary Figure S1. Distribution of atoms along the bilayer normal. Normalized density profiles obtained for the simulations of the E2 conformation of SERCA in a pure POPC lipid bilayer (blue) and
Mechanisms of antagonism of HIVspecific CD4+ T cell responses BSRI
 Mechanisms of antagonism of HIVspecific CD4+ T cell responses BSRI Problems Virus escape from immune recognition Antagonism of T cell responses Peptide-MHC-TCR interaction T cell antagonism Variants of
Mechanisms of antagonism of HIVspecific CD4+ T cell responses BSRI Problems Virus escape from immune recognition Antagonism of T cell responses Peptide-MHC-TCR interaction T cell antagonism Variants of
Supplementary Figure 1. BMS enhances human T cell activation in vitro in a
 Supplementary Figure 1. BMS98662 enhances human T cell activation in vitro in a concentration-dependent manner. Jurkat T cells were activated with anti-cd3 and anti-cd28 antibody in the presence of titrated
Supplementary Figure 1. BMS98662 enhances human T cell activation in vitro in a concentration-dependent manner. Jurkat T cells were activated with anti-cd3 and anti-cd28 antibody in the presence of titrated
ABSTRACT. A diverse T cell receptor (TCR) repertoire is a prerequisite for effective viral
 ABSTRACT Title of Thesis: STRUCTURAL BASIS FOR CLONAL DIVERSITY OF THE PUBLIC T CELL RESPONSE TO DOMINANT EPITOPES FROM CYTOMEGALOVIRUS AND INFLUENZA Xinbo Yang, Doctor of Philosophy, 2016 Thesis Directed
ABSTRACT Title of Thesis: STRUCTURAL BASIS FOR CLONAL DIVERSITY OF THE PUBLIC T CELL RESPONSE TO DOMINANT EPITOPES FROM CYTOMEGALOVIRUS AND INFLUENZA Xinbo Yang, Doctor of Philosophy, 2016 Thesis Directed
Supplementary Information
 Supplementary Information Using the pimeloyl-coa synthetase adenylation fold to synthesise fatty acid thioesters Menglu Wang 1a ; Lucile Moynié 2a, Peter J. Harrison 1, Van Kelly 1, Andrew Piper 1, James
Supplementary Information Using the pimeloyl-coa synthetase adenylation fold to synthesise fatty acid thioesters Menglu Wang 1a ; Lucile Moynié 2a, Peter J. Harrison 1, Van Kelly 1, Andrew Piper 1, James
Crystal Structure of the Subtilisin Carlsberg: OMTKY3 Complex
 John Clizer & Greg Ralph Crystal Structure of the Subtilisin Carlsberg: OMTKY3 Complex The turkey ovomucoid third domain (OMTKY3) is considered to be one of the most studied protein inhibitors. 1 Ovomucin
John Clizer & Greg Ralph Crystal Structure of the Subtilisin Carlsberg: OMTKY3 Complex The turkey ovomucoid third domain (OMTKY3) is considered to be one of the most studied protein inhibitors. 1 Ovomucin
Probing the catalytic mechanism of an antifibrotic copper metallodrug
 Probing the catalytic mechanism of an antifibrotic copper metallodrug 4 December 2017 Lizelle Lubbe Supervisor: Prof Ed Sturrock Transition metals offer benefit of designing compounds with complex architectures,
Probing the catalytic mechanism of an antifibrotic copper metallodrug 4 December 2017 Lizelle Lubbe Supervisor: Prof Ed Sturrock Transition metals offer benefit of designing compounds with complex architectures,
Principles of Adaptive Immunity
 Principles of Adaptive Immunity Chapter 3 Parham Hans de Haard 17 th of May 2010 Agenda Recognition molecules of adaptive immune system Features adaptive immune system Immunoglobulins and T-cell receptors
Principles of Adaptive Immunity Chapter 3 Parham Hans de Haard 17 th of May 2010 Agenda Recognition molecules of adaptive immune system Features adaptive immune system Immunoglobulins and T-cell receptors
Supplementary Figure-1. SDS PAGE analysis of purified designed carbonic anhydrase enzymes. M1-M4 shown in lanes 1-4, respectively, with molecular
 Supplementary Figure-1. SDS PAGE analysis of purified designed carbonic anhydrase enzymes. M1-M4 shown in lanes 1-4, respectively, with molecular weight markers (M). Supplementary Figure-2. Overlay of
Supplementary Figure-1. SDS PAGE analysis of purified designed carbonic anhydrase enzymes. M1-M4 shown in lanes 1-4, respectively, with molecular weight markers (M). Supplementary Figure-2. Overlay of
BIRKBECK COLLEGE (University of London)
 BIRKBECK COLLEGE (University of London) SCHOOL OF BIOLOGICAL SCIENCES M.Sc. EXAMINATION FOR INTERNAL STUDENTS ON: Postgraduate Certificate in Principles of Protein Structure MSc Structural Molecular Biology
BIRKBECK COLLEGE (University of London) SCHOOL OF BIOLOGICAL SCIENCES M.Sc. EXAMINATION FOR INTERNAL STUDENTS ON: Postgraduate Certificate in Principles of Protein Structure MSc Structural Molecular Biology
Structural analysis of fungus-derived FAD glucose dehydrogenase
 Structural analysis of fungus-derived FAD glucose dehydrogenase Hiromi Yoshida 1, Genki Sakai 2, Kazushige Mori 3, Katsuhiro Kojima 3, Shigehiro Kamitori 1, and Koji Sode 2,3,* 1 Life Science Research
Structural analysis of fungus-derived FAD glucose dehydrogenase Hiromi Yoshida 1, Genki Sakai 2, Kazushige Mori 3, Katsuhiro Kojima 3, Shigehiro Kamitori 1, and Koji Sode 2,3,* 1 Life Science Research
More powerful virus inhibitors from structure-based analysis of
 More powerful virus inhibitors from structure-based analysis of HEV71 capsid-binding molecules Luigi De Colibus, Xiangxi Wang, John A. B. Spyrou, James Kelly, Jingshan Ren, Jonathan Grimes, Gerhard Puerstinger,
More powerful virus inhibitors from structure-based analysis of HEV71 capsid-binding molecules Luigi De Colibus, Xiangxi Wang, John A. B. Spyrou, James Kelly, Jingshan Ren, Jonathan Grimes, Gerhard Puerstinger,
Computer Simulation of T-Cell Activation
 ISIM1 Computational Immunology Computer Simulation of T-Cell Activation W. Schreiner, M. Cibena, U. Omasits, M. Berger, R. Kobler, M. Neumann, O. Steinhauser Section for Biomedical Computersimulation and
ISIM1 Computational Immunology Computer Simulation of T-Cell Activation W. Schreiner, M. Cibena, U. Omasits, M. Berger, R. Kobler, M. Neumann, O. Steinhauser Section for Biomedical Computersimulation and
Bioinformatics for molecular biology
 Bioinformatics for molecular biology Structural bioinformatics tools, predictors, and 3D modeling Structural Biology Review Dr Research Scientist Department of Microbiology, Oslo University Hospital -
Bioinformatics for molecular biology Structural bioinformatics tools, predictors, and 3D modeling Structural Biology Review Dr Research Scientist Department of Microbiology, Oslo University Hospital -
CS612 - Algorithms in Bioinformatics
 Spring 2016 Protein Structure February 7, 2016 Introduction to Protein Structure A protein is a linear chain of organic molecular building blocks called amino acids. Introduction to Protein Structure Amine
Spring 2016 Protein Structure February 7, 2016 Introduction to Protein Structure A protein is a linear chain of organic molecular building blocks called amino acids. Introduction to Protein Structure Amine
SDS-Assisted Protein Transport Through Solid-State Nanopores
 Supplementary Information for: SDS-Assisted Protein Transport Through Solid-State Nanopores Laura Restrepo-Pérez 1, Shalini John 2, Aleksei Aksimentiev 2 *, Chirlmin Joo 1 *, Cees Dekker 1 * 1 Department
Supplementary Information for: SDS-Assisted Protein Transport Through Solid-State Nanopores Laura Restrepo-Pérez 1, Shalini John 2, Aleksei Aksimentiev 2 *, Chirlmin Joo 1 *, Cees Dekker 1 * 1 Department
172R 172K TAM-2/172R TAM-2/172K. AZT concentration [nm] AZT concentration [nm] MgCl 2 2.5K 2.5K 5K 2.5K 5K 2.5K K 5K 2.5K 5K 2.5K 50 2.
![172R 172K TAM-2/172R TAM-2/172K. AZT concentration [nm] AZT concentration [nm] MgCl 2 2.5K 2.5K 5K 2.5K 5K 2.5K K 5K 2.5K 5K 2.5K 50 2. 172R 172K TAM-2/172R TAM-2/172K. AZT concentration [nm] AZT concentration [nm] MgCl 2 2.5K 2.5K 5K 2.5K 5K 2.5K K 5K 2.5K 5K 2.5K 50 2.](/thumbs/82/85138523.jpg) 5 5 5 5 A MgCl 2 172R 172K TAM-2/172R TAM-2/172K AZT concentration [nm] B 172R 172K TAM-2/172R TAM-2/172K AZT concentration [nm] ATP + ATP - Supplemental Figure 1. Primer extension of HIV-1 RT polymorphisms
5 5 5 5 A MgCl 2 172R 172K TAM-2/172R TAM-2/172K AZT concentration [nm] B 172R 172K TAM-2/172R TAM-2/172K AZT concentration [nm] ATP + ATP - Supplemental Figure 1. Primer extension of HIV-1 RT polymorphisms
Experimental. Crystal data. C 22 H 26 N 2 O 3 M r = Triclinic, P1 a = (3) Å b = (4) Å c = (4) Å = (6) = 100.
 Acta Crystallographica Section E Structure Reports Online ISSN 1600-5368 15-Methoxy-14,15-dihydroandranginine Dian-Lei Wang, a Xiang-Hai Cai, b Peng Huang a * and He-Ping Huang a * a School of Pharmacy,
Acta Crystallographica Section E Structure Reports Online ISSN 1600-5368 15-Methoxy-14,15-dihydroandranginine Dian-Lei Wang, a Xiang-Hai Cai, b Peng Huang a * and He-Ping Huang a * a School of Pharmacy,
Supplementary Figure 1
 Supplementary Figure 1 Identification of IFN-γ-producing CD8 + and CD4 + T cells with naive phenotype by alternative gating and sample-processing strategies. a. Contour 5% probability plots show definition
Supplementary Figure 1 Identification of IFN-γ-producing CD8 + and CD4 + T cells with naive phenotype by alternative gating and sample-processing strategies. a. Contour 5% probability plots show definition
Significance of the MHC
 CHAPTER 7 Major Histocompatibility Complex (MHC) What is is MHC? HLA H-2 Minor histocompatibility antigens Peter Gorer & George Sneell (1940) Significance of the MHC role in immune response role in organ
CHAPTER 7 Major Histocompatibility Complex (MHC) What is is MHC? HLA H-2 Minor histocompatibility antigens Peter Gorer & George Sneell (1940) Significance of the MHC role in immune response role in organ
Introduction to proteins and protein structure
 Introduction to proteins and protein structure The questions and answers below constitute an introduction to the fundamental principles of protein structure. They are all available at [link]. What are
Introduction to proteins and protein structure The questions and answers below constitute an introduction to the fundamental principles of protein structure. They are all available at [link]. What are
SUPPLEMENTARY INFORMATION
 Supplementary Notes 1: accuracy of prediction algorithms for peptide binding affinities to HLA and Mamu alleles For each HLA and Mamu allele we have analyzed the accuracy of four predictive algorithms
Supplementary Notes 1: accuracy of prediction algorithms for peptide binding affinities to HLA and Mamu alleles For each HLA and Mamu allele we have analyzed the accuracy of four predictive algorithms
