Immunochromatographic Test for Detection of Cryptosporidium in Malignant Children Before and During Chemotherapy in Basra
|
|
|
- Alexander Patrick
- 5 years ago
- Views:
Transcription
1 International Journal of Innovative Biochemistry & Microbiology Research 4(1):1-9, Jan.-Mar SEAHI PUBLICATIONS, ISSN: Immunochromatographic Test for Detection of Cryptosporidium in Malignant Children Before and During Chemotherapy in Basra Sabeeha M. Abdul-Hussein¹, Abdulmohsin H. Jassim², Jinan G. Hassan³ Department of Medical Microbiology, College of Medicine, Basra University, Basra, Iraq. ABSTRACT Immunochromatographic (IC) tests may play an important role in the future diagnosis of parasitic diseases because of their speed and simplicity of use. A recently developed test to detect Cryptosporidium. Microscopy the gold standard reference techniques and the results of this IC test were compared with those obtained with ELISA and IC single test for the parasite. One hundred stool samples were assayed from malignant children aged 2 month to 14 years before and during (6 to 8 weeks) chemotherapy. Using microscopy,9 sample before chemotherapy and 15 during chemotherapy were diagnosed as positive for Cryptosporidium. Results of IC tests show 13 positive sample before chemotherapy and 33 during chemotherapy sensitivities of 100% and specificities were of 95.9 % before chemotherapy, when,results of IC tests show sensitivities of 100% and specificities were of 80.2 % during chemotherapy when compare with staining method. Keywords: Cryptosporidiosis, Immunochromatographic test (IC), Cryptosporidium, ELISA, INTRODUCTION Cryptosporidiosis is a parasitic disease caused by an apicomplexan protozoa of the genus Cryptosporidium ; Cryptosporidium parvum is the specific infective agent in human (Ikechukwu et al.,2011).cryptosporidium are waterborne, obligate intracellular protozoan parasite,(mosallanejad et al.,2010). Ernest Edward Tyzzer in 1907 was the first person successively to identify the oocyst of Cryptosporidium parasite (Fayer,2004). The first case of human cryptosporidiosis was reported in 1976(Nime et al., 1976). Cryptosporidiosis, long considered to be a veterinary disease, has emerged as a serious human health problem, being the most frequent secondary diagnosis in people with AIDS and significantly contributing to their mortality. Human Cryptosporidium parvum associated disease is the result of zoonotic or anthroponotic transmission of the parasite s infectious stages, the oocysts. The parasite is transmitted via a fecal - oral route and very frequently via contaminated water and food ( Graczyk T.K., et al., 2003 ). Diarrheal caused by Cryptosporidium was estimated in immunocompetent individuals between 1%-10% of diarrheal disease worldwide. It is also found in 6% of all patients with acquired immunodeficiency syndrome (AIDS) and in 21% of AIDS patients with diarrhea (Ayalew,2006). The life cycle of Cryptosporidium is complex(casemore,1990). In this study determining the most sensitive and specific method for diagnosing the parasite to be a guidative for diagnostic laboratories. SUBJECTS AND METHODS A total of 100 fecal specimens were collected from malignant children aged 2 month to 14 years before and during (6 to 8 weeks) chemotherapy in Pediatric Oncology Center at Basrah children s specialty Hospital in Basrah Province during May to November 2015; 100 stool specimen from symptomatic and asymptomatic hospitalized children (including males and females).stool samples were collected and examined using Formalin-Ethyl Acetate Sedimentation method, a modified acid fast staining method,immunochromatographic test (IC) and ELISA. Stool samples must be collected in clean containers 1
2 without any additives and stored at 2 8 C. If stored for more than 3 days, the sample must be frozen at 20 C. In this case, the sample must be completely thawed out and brought to room temperature before testing begins. 1. Concentration of Fecal Samples :- Stool samples were concentrated using the Formalin-Ethyl Acetate Sedimentation technique to achieve a better result in the a modified acid fast staining method( Garcia and Bruckner,1993). 2. Staining Method:- Two thin smears were prepared from concentrated materials, dried in room air and fixed, then stained with Kinyoun s Acid-Fast and Modified Ziehl-Neelsen acid fast staining method,then examined under a microscope using the high-power, using oil immersion(garcia and Bruckner,1993 ). The oocysts of Cryptosporidium spp. were described pink-red Oocysts are 4 to 6 µm in diameter, and four sporozoites may be present internally. The background should stain uniformly blue(garcia and Bruckner,1993). 3.Immunochromatographic test (IC) :- The RIDA Quick Cryptosporidium test (R-Biopharm AG, Darmstadt, Germany) was used. This test uses monoclonal antibodies directed against specific membrane proteins of Cryptosporidium. The method is a one-step lateral flow immunochromatographic assay containing specific antibodies labelled to coloured latex particles (blue for Cryptosporidium) to generate a visual signal. Latex labelled specific antibodies are applied into a porous filter affixed to a membrane. Capture specific antibodies are also applied at different levels on the membrane. Capture specific antibodies are also applied at different levels on the membrane. To perform the test, a faecal specimen is suspended in antigen extraction buffer and added to the porous filter containing latex labelled antibodies. If the target antigens are present in the specimens, these antibodies specifically bind to the antigens. The resulting latex labelled antigen/antibody complexes flow by capillarity action from the filter through the membrane. Here they are bound and immobilized by the redeposited capture antibodies, which are specific against the same protozoan antigens or against the latex labelled antibodies (as an internal control). This allows colour particles to concentrate and form a visible line. Indeed, after binding, immune complexes become visible and permit visual inspection of positive reactions. Change of colour on the control line is necessary to validate the test and its non-appearance, with or without colour changes on the test lines, invalidates the test. For the accomplishment of the method, reagents were stored at room temperature (20-25 C) until use. The tests were conducted at room temperature following the recommended manufacturer s instructions. Briefly, 1 ml of extraction buffer was placed into a test tube. Approximately 50 mg of stool sample were added, homogenized by a vortex mixer, and allowed to settle for 3 minutes. Then, four drops of the clear supernatant were placed into the round inlet opening of the test cassette. After 5 minutes, results were visually read. A negative result was represented by the appearance of a single blue band in the control area of the cassette. A positive result was indicated by the presence of a red(cryptosporidium positive ) band in the test areas of the cassette. 3. Enzyme Linked Immunosorbent Assay (ELISA) :- Enzyme Linked Immunoassay (ELISA) for the detection of C. parvum in faecal samples. RIDIASCREEN test (C 1201). (Art. No.: C GmbH, Darmstadt, Germany) The test was used for in vitro diagnosis of Cryptosporidium. This test is enzyme immunoassay for the qualitative determination of C. parvum in faecal samples and performed according to instructions of the kits. Microwell plate and reagents were brought to room temperature (20-25 ºC). The washing buffer was diluted with distilled water 1:10, followed by dilution the faecal sample with the sample dilution buffer diluent 1:11. The required micro well strips were placed in the frame followed by addition of 2 drops or 100 µl positive control (control +), negative control (diluent), or sample. After that 2 drops or 100 µl conjugate were added. Incubation was at room temperature (20-25ºC) for 60 min followed by Wash for five times with 300 µl diluted wash buffer followed by addition of 2 drops or 100 µl substrate. The faecal sample was incubated again at room temperature (20-25ºC) in the dark for 15 min followed by adding 1 drop or 50 µl stop reagent. Finally, photometric measurement was carried out at 450 nm (optional reference wave length > 600 nm). As indicated by the manufacturer instruction s, samples were considered positive when their extinction was more than 10% above the calculated cut-off, equivocal when their extinctions were within ± 10% of the cut-off and negative when their extinctions were 10% below the calculated cut-off. 2
3 Statistical methods: Statistical package of social science (SPSS) version 20 was used to analyze data, Chi-square (ᵡ²) test was used to assess the significance of difference between groups and variable, P-value less than 0.05 was considered to be statistically significant. RESULTS The results showed that the total infectivity rate of Cryptosporidium spp. in children was (31.1%) during chemotherapy in comparison with(12.3%) before chemotherapy ( Table 1). The number of positive samples were 33 out of 106 patients during chemotherapy and 13 out of 106 before chemotherapy. A total of 106 stool samples were microscopically examined for the presence of Cryptosporidium,9 were positive for Cryptosporidium before chemotherapy and 15 were positive for Cryptosporidium during chemotherapy, but were examined by immunochromatographic test (IC) and ELISA shows 13 positive for Cryptosporidium before chemotherapy and 33 positive for Cryptosporidium during chemotherapy. In( Table 2 )show Results of IC tests 13positive sample before chemotherapy and 33 during chemotherapy sensitivities of 100% and specificities were of 96.7 % before chemotherapy, when,results of IC tests show sensitivities of 100% and specificities were of 81.2 % during chemotherapy when compare with staining method. Table (1) Cryptosporidium by staining method,ic test and ELISA before and during chemotherapy Patients group Staining method ᵡ² P value Positive Negative No. % No. % Before chemotherapy During chemotherapy Patients group IC test &ELISA ᵡ² P value Positive Negative No. % No. % Before chemotherapy During chemotherapy
4 Figure (1) Cryptosporidium by staining method before and during chemotherapy Figure (2) Cryptosporidium by IC&ELISA before and during chemotherapy 4
5 Table(2) Diagnosis of Cryptosporidium by Immunochromatographic test (IC) and ELISA in comparison with staining method ( standard method) before and during chemotherapy Before chemotherapy Test Result Staining Method No. Patients Total Positive Negative Ic Test Positive (100) Negative (95.9) ELISA Positive (100) Negative 0 93 (95.9) 93 During chemotherapy Test Result Staining Method No. Patients Total Positive Negative IC Test Positive (100) Negative (80.2) ELISA Positive (100) Negative 0 73 (80.2) 73 5
6 Figure (3) Comparison between staining method, Immunochromatographic test (IC) and ELISA before and during chemotherapy In Table (2) A total of 106 stool samples were microscopically examined for the presence of Cryptosporidium,9 were positive for Cryptosporidium before chemotherapy and 15 were positive for Cryptosporidium during chemotherapy, but were examined by Immunochromatographic test (IC) and ELISA shows 13 positive for Cryptosporidium before chemotherapy and 33 positive for Cryptosporidium during chemotherapy. Also in Table (2) show Results of Immunochromatographic test (IC) and ELISA (13)positive sample before chemotherapy and (33) during chemotherapy sensitivities of (100% )and specificities were of (95.9 %) before chemotherapy, when,results of Immunochromatographic test (IC) and ELISA show sensitivities of (100%) and specificities were of (80.2 % )during chemotherapy when compare with staining method. In table (3) From among hundred and six patients with malignant diseases before chemotherapy 13 (12.26 %) were found to excrete Cryptosporidium oocyst in their stool compared to 33(31.13%) of patients with malignant diseases during chemotherapy,while among hundred and six patients with malignant diseases before chemotherapy 66( 62.3 % ) were found to be positive for various species of intestinal parasites to 40 ( 37.7 %) of patients with malignant diseases during chemotherapy (Table 4-2). Statistically, the difference was very highly significant ( ᵡ² = , df =2, p <0.05). Table (3) Parasitic and Cryptosporidium infections among patients with malignant disease before and during chemotherapy Patients group No. examined Parasitic infections Cryptosporidium infections +ve % +ve % Before chemotherapy During chemotherapy X 2 =15.598, df =2, p value=
7 DISCUSSION Comparison of the results of the new test with Staining Method for detection of Cryptosporidium infection in all samples from children, as well as detection of Immunochromatographic Test and ELISA Specificity & sensitivity were summarized in Table(1 and 2). Immunochromatographic Test allows the reliable detection of oocysts in one step does not require any special skill and is simple to use. Among several authors who performed Antigen detection by immunoassays, Agnamey et al. (2011) concluded that these methods have become a well-established aid to microscopic examination for the diagnosis of cryptosporidiosis. Good sensitivities and specificities have been reported for some of these tests in their comparative study. Cryptosporidium was found in (12.3%) in different types of malignancy in patients before chemotherapy and (31.1%) in different types of malignancy in patients during chemotherapy. These results are differently to some studies, as in in Basra city which found cryptosporidiosis among cancer patients to be (8%)( Mahdi et al.,2002),also in Basra by Mahdi (9.9%)( Mahdi et al.,1998) and in Baghdad (14.78%) by Al-Warid et al.(2012 ).In Kuwait found Cryptosporidium about (3.4%) ( Iqbal et al.,2011). In Egypt, cryptosporidiosis in immunocompromised children about (4.8%)( El-Mahallawy et al.,2014 ). In Iran found in children suffered from hematopoietic malignancy about (4.2%)( Dehkordy et al., 2010). In Ethiopia about (10.4%) ( Adamu,2010), in German, cryptosporidium was found in (12.6% )of patients with colorectal cancer (Sulżyc- Bielicka et al., 2012). In India found Cryptosporidium in (3.8%) (Sharma et al., 2013). explanation the differences and disparities in rates of Cryptosporidium infection among children of cancer patients because the patients have a lack of interaction between nutrition and immunity of the patient due to exposure to chemotherapy and malnutrition one recurring infected reasons, also patients who are exposed to chemotherapy weakens the cells and spread the parasite to the weakness of the patient's immunity and exposure and contact with infected animals.( Rudrapantna et al.,1997; Berenji et al.,2007 ) But did not report in previous studies to know the preparation of Cryptosporidium infection during chemotherapy.while was found intestinal parasite in (62.3%) different types of malignancy in children before chemotherapy and (37.7 %) in different types of malignancy in children during chemotherapy, this significant reflect two major changing reduction in the all-percent per dent and increasing in Cryptosporidium infection. These results are differently to some studies, as in Basra was found (49.5%) in different types of malignancy(mahdi et al.,1998),in Baghdad was found parasitic infections about (71.4%)(Hussein et al., 2011). In Egypt found parasitic intestinal about (12.9%)( Rivera et al., 1989), Botero et al. in 2003 reported about (32.9%) of intestinal parasites found in leukemia patients(botero et al., 2003).In Iran Zabolinejad et al. found Intestinal Parasites in Children with Lymphohematopoietic Malignancy in (35.9% )( Zabolinejad et al.,2013). In this study we addressed several diagnosed Cryptosporidium ways and comparison between them and the standard method, the results were the emergence of positive cases in the examination IC and ELISA and negative in the staining method for the same patient. This result is due to the number of oocysts, which take during the examination where in the way staining method need oocysts about 500,000 oocysts per gram of stool(weber B et al.,1991),but in ELISA needed about 500 oocysts per gram in stool (Chappell C L et al., 1999) and in IC needed about 1000 oocysts per gram in stool (Llorente T M et al., 2002). So observed proportion of positive cases in Immunochromatographic test and ELISA more than in staining method because in staining method needed high number of oocysts,also it needs to be versed examiner to detect oocysts due to the small size of the oocysts by microscopic examination((mchardy I H et al.,2014). CONCLUSION We can conclude that Immunochromatographic Test is a specific, sensitive and easy to use test for diagnostic of Cryptosporidium oocysts in stools. To our knowledge, this is the first study in IRAQ that uses both RIDASCREEN Cryptosporidium ELISA coproantigenic test and RIDA Quick Cryptosporidium test to detect Cryptosporidium infection in humans and compare their sensitivity and specificity in malignant children before and during chemotherapy. 7
8 REFERENCES 1. Ikechukwu D, Benjamin N, Uchechukwu C. Cryptosporidiosis in Imo State, Nigeria. Journal of Rural and Tropical Public Health, 2011; 10: Mosallanejad B, Hamidinejat H, Avizeh R, Ghorbanpoor N M, Razi M H. Antigenic detection of Cryptosporidium parvum in urban and rural dogs in Ahvaz district, southwestern Iran. Iranian Journal of Veterinary Research, 2010; 11(3): Fayer R. Cryptosporidium: a water-borne zoonotic parasite. Veterinary Parasitology, 2004; 126: doi: /j.vetpar Nime FA,Burek JD,Page DL,Holscher MA,Yardley JH.Acute enterocolitis in a human being infected with the protozoan Cryptosporidium. Gastroenterology,1976;70(4): Graczyk T K, Grimes B H, Knight R, Da Sliva A J, Pieniazek NJ, Veal D A. Detection of Cryptosporidium parvum and Giardia lamblia carried by synanthropic flies by combined fluorescent in situ hybridization and a monoclonal antibody. American Journal Tropical Medicine Hygiene,2003; 68(2): Ayalew D.Association of Cryptosporidium parvum, Giardia lamblia and Entamoeba histolytica /dispar INFECTION WITH DRINKING WATER SOURCES AMONG CHILDREN IN RURAL PART OF DIRE- DAWA EASTERN ETHIOPIA. MSc. Thesis University of Addis Ababa, July Casemore DP. Epidemiological Aspects Of Human Cryptosporidiosis. Epidemiology Infection journal.,1990, 104, Garcia LS And Bruckner DA. Diagnostic medical parasitology, 2nd edition, Washington, D.C. United States of America, 1993: Mahdi NK, Ali NH.Cryptosporidiosis among animal handlers and their livestock in Basrah, Iraq. East African Medical Journal,2002; 79(10):550_ Mahdi NK, Al-Sadoon IA, Mohamed AT.First report of cryptosporidiosis among Iraqi children. Medical Journal of Basra University,1997; 2(1): Iqbal J, Khalid N, Hira P R. Cryptosporidiosis in Kuwaiti children: association of clinical characteristics with Cryptosporidium species and subtypes. Journal of Medical Microbiology,2011; 60: El-Mahallawy H, Noussa R. El Basha, Mayssa M. Zaki, Maha El-Arousy,Shaadi F. Elswaifi, E. M. Abo-hashem. A comparative study on enteric parasitic infections in immunocompetent and immunosuppressed children in Egypt. Comparative Clinical Pathology,2014;23(5): Dehkordy A B, Rafiei A, Alavi S M, Latifi S M.Prevalence of Cryptosporidium Infection in Immunocompromised Patients, In South-West of Iran. Iranian J Parasitol, 2010 ; 5(4) : Sulżyc-Bielicka V, Kołodziejczyk L, Jaczewska S, Bielicki D, Kładny J. Prevalence of Cryptosporidium sp. In patients with colorectal cancer. Polski Przegląd Chirurgiczny,2012;84(7); Sharma P, Sharma A, Sehgal R, Malla N, Khurana S. Genetic diversity of Cryptosporidium isolates from patients in North India. International Journal of Infectious Diseases,2013; Berenji F, Zabolinejad N, Kianifar H, Badeii Z, Banihashem A, Hiradfar A.Cryptosporidium Infection in Pediatric Patients with Lymphohematopoietic Malignancies. Iran J Pediatr,2007 ; 17 (3) : Rudrapantna JS, Kumar V, Sridhar H. Intestinal parasitic infections in patients with malignant diseases. J. Diarrhoea Dis. Res.,1997; 15(2): Hussein R A, Shaker M J, Majeed H A. Prevalence of Intestinal Parasitic Infections among Children in Baghdad City,2011; Rivera L R, Cardos R, Martínez-Guerra G, Ayón A, Leal C, Rivera-Ortegón F. Childhood acute leukemia and intestinal parasitosis. Europe PMC,1989;3(11): Botero J H, Castaño A, Montoya M N, Ocampo N E, Hurtado M I, Lopera M M. A Preliminary Study Of The Prevalence Of Intestinal Parasites Inimmunocompromised Patients With And Without Gastrointestinal Manifestations. Rev. Inst. Med. trop. S. Paulo,2003; 45(4): Zabolinejad N, Berenji F, Eshkaftaki E B, Badeii Z, Banihashem A, Afzalaqaei M. Intestinal Parasites in Children with Lymphohematopoietic Malignancy in Iran, Mashhad. Jundishapur Journal of Microbiology,2013;6(6). 8
9 22. Weber B, Bryan R T, Bishop H S, Wahlquist S P, Sullivan J J, Juranek D. Threshold of Detection of Cryptosporidium Oocysts in Human Stool Specimens: Evidence for Low Sensitivity of Current Diagnostic Methods. Journal Of Clinical Microbiology, 1991;29(7): Chappell C L, Okhuysen P O, Sterling C R, Wang C, Jakubowski W,Dupont H L. INFECTIVITY OF CRYPTOSPORIDIUM PARVUM IN HEALTHY ADULTS WITH PRE-EXISTING ANTI-C. PARVUM SERUM IMMUNOGLOBULIN. Am. J. Trop. Med. Hyg.,1999; 60(1): Llorente T M, Clavel A, Varea M, Olivera S, Castillo F J, Sahagún J, Rubio M C, Gómez-Lus R.Evaluation of an Immunochromatographic Dip-Strip Test for the Detection of Cryptosporidium Oocysts in Stool Specimens. Eur. J. Clin. Microbiol. Infect Dis.,2002; 21: McHardy I H, Wu M, Shimizu-Cohen R, Couturier M R, Humphriesa R M. Detection of Intestinal Protozoa in the Clinical Laboratory. Journal of Clinical Microbiology,2014; 52(3) :
Cryptosporidium Veterinary
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
JMSCR Vol 06 Issue 09 Page September 2018
 www.jmscr.igmpublication.org Impact Factor (SJIF): 6.379 Index Copernicus Value: 79.54 ISSN (e)-2347-176x ISSN (p) 2455-0450 DOI: https://dx.doi.org/10.18535/jmscr/v6i9.91 A review for Cryptosporidiosis
www.jmscr.igmpublication.org Impact Factor (SJIF): 6.379 Index Copernicus Value: 79.54 ISSN (e)-2347-176x ISSN (p) 2455-0450 DOI: https://dx.doi.org/10.18535/jmscr/v6i9.91 A review for Cryptosporidiosis
Cryptosporidium spp. ELISA Kit
 Cryptosporidium spp. ELISA Kit Catalog Number KA2094 96 assays Version: 03 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
Cryptosporidium spp. ELISA Kit Catalog Number KA2094 96 assays Version: 03 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
RIDA QUICK Rotavirus. Article no: N0902
 RIDA QUICK Rotavirus Article no: N0902 R-Biopharm AG, An der neuen Bergstraße 17, D-64297 Darmstadt, Germany Phone: +49 (0) 61 51 81 02-0 / Fax: +49 (0) 61 51 81 02-20 1. Intended use For in vitro diagnostic
RIDA QUICK Rotavirus Article no: N0902 R-Biopharm AG, An der neuen Bergstraße 17, D-64297 Darmstadt, Germany Phone: +49 (0) 61 51 81 02-0 / Fax: +49 (0) 61 51 81 02-20 1. Intended use For in vitro diagnostic
RIDA QUICK Cryptosporidium/Giardia/Entamoeba Combi
 RIDA QUICK Cryptosporidium/Giardia/Entamoeba Combi Article no: N1723 R-Biopharm AG, An der neuen Bergstraße 17, D-64297 Darmstadt, Germany Phone: +49 (0) 61 51 81 02-0 / Fax: +49 (0) 61 51 81 02-20 1.
RIDA QUICK Cryptosporidium/Giardia/Entamoeba Combi Article no: N1723 R-Biopharm AG, An der neuen Bergstraße 17, D-64297 Darmstadt, Germany Phone: +49 (0) 61 51 81 02-0 / Fax: +49 (0) 61 51 81 02-20 1.
PREVALENCE OF DIFFERENT PROTOZOAN PARASITES IN PATIENTS VISITING AT ICDDR B HOSPITAL, DHAKA
 J. Asiat. Soc. Bangladesh, Sci. 39(1): 117-123, June 213 PREVALENCE OF DIFFERENT PROTOZOAN PARASITES IN PATIENTS VISITING AT ICDDR B HOSPITAL, DHAKA SHAHELA ALAM, HAMIDA KHANUM 1, RIMI FARHANA ZAMAN AND
J. Asiat. Soc. Bangladesh, Sci. 39(1): 117-123, June 213 PREVALENCE OF DIFFERENT PROTOZOAN PARASITES IN PATIENTS VISITING AT ICDDR B HOSPITAL, DHAKA SHAHELA ALAM, HAMIDA KHANUM 1, RIMI FARHANA ZAMAN AND
E. Histolytica IgG ELISA Kit
 E. Histolytica IgG ELISA Kit Catalog Number KA3193 96 assays Version: 01 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of
E. Histolytica IgG ELISA Kit Catalog Number KA3193 96 assays Version: 01 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of
Outline EP1201. NEHA 2012 AEC June 2012
 Food and Water Borne Enteric Protozoa: Environmental Health Perspectives Stephanie M. Fletcher, PhD (c) NEHA AEC & Exhibition San Diego June 30, 2012 Outline Introduction Overview of the Epidemiology of
Food and Water Borne Enteric Protozoa: Environmental Health Perspectives Stephanie M. Fletcher, PhD (c) NEHA AEC & Exhibition San Diego June 30, 2012 Outline Introduction Overview of the Epidemiology of
Crypto / Giardia Ag Combo (Fecal) ELISA
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
RIDA QUICK Entamoeba. Article no: N1702
 RIDA QUICK Entamoeba Article no: N1702 R-Biopharm AG, An der neuen Bergstraße 17, D-64297 Darmstadt, Germany Phone: +49 (0) 61 51 81 02-0 / Fax: +49 (0) 61 51 81 02-20 1. Intended use For in vitro diagnostic
RIDA QUICK Entamoeba Article no: N1702 R-Biopharm AG, An der neuen Bergstraße 17, D-64297 Darmstadt, Germany Phone: +49 (0) 61 51 81 02-0 / Fax: +49 (0) 61 51 81 02-20 1. Intended use For in vitro diagnostic
Giardia lamblia/ Cryptosporidium spp. ELISA Kit
 Giardia lamblia/ Cryptosporidium spp. ELISA Kit Catalog Number KA2095 96 assays Version: 02 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background...
Giardia lamblia/ Cryptosporidium spp. ELISA Kit Catalog Number KA2095 96 assays Version: 02 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background...
RIDA QUICK Entamoeba. Article no: N 1703
 RIDA QUICK Entamoeba Article no: N 1703 R-Biopharm AG, Landwehrstr. 54, D-64293 Darmstadt, Germany Tel: +49 (0) 61 51 81 02-0 / Fax: +49 (0) 61 51 81 02-20 1. Intended use For in vitro diagnostic use.
RIDA QUICK Entamoeba Article no: N 1703 R-Biopharm AG, Landwehrstr. 54, D-64293 Darmstadt, Germany Tel: +49 (0) 61 51 81 02-0 / Fax: +49 (0) 61 51 81 02-20 1. Intended use For in vitro diagnostic use.
Fecal H. pylori Antigen Rapid Test (Strip)
 Fecal H. pylori Antigen Rapid Test (Strip) Cat. No.:DTS526 Pkg.Size: Intended use This H. pylori Antigen Rapid Test is intended for the direct qualitative detection of the presence of H. pylori antigen
Fecal H. pylori Antigen Rapid Test (Strip) Cat. No.:DTS526 Pkg.Size: Intended use This H. pylori Antigen Rapid Test is intended for the direct qualitative detection of the presence of H. pylori antigen
PARA-TECT Cryptosporidium/Giardia Direct Fluorescent Assay Directions For Use For In Vitro Diagnostic Use
 Medical Chemical Corp., 19430 Van Ness Avenue, Torrance, California 90501 (310)787-6800, Fax (310)787-4464 Catalog # MCC-C/G - DFA, 75 Test PARA-TECT Cryptosporidium/Giardia Direct Fluorescent Assay Directions
Medical Chemical Corp., 19430 Van Ness Avenue, Torrance, California 90501 (310)787-6800, Fax (310)787-4464 Catalog # MCC-C/G - DFA, 75 Test PARA-TECT Cryptosporidium/Giardia Direct Fluorescent Assay Directions
Giardia lamblia ELISA Kit
 Giardia lamblia ELISA Kit Catalog Number KA2093 96 assay Version: 01 Intend for research use only www.abnova.com Introduction and Background A. Intended Use Giardia lamblia ELISA Kit is an Enzyme-Linked
Giardia lamblia ELISA Kit Catalog Number KA2093 96 assay Version: 01 Intend for research use only www.abnova.com Introduction and Background A. Intended Use Giardia lamblia ELISA Kit is an Enzyme-Linked
CoproELISA Giardia/Cryptosporidium
 CoproELISA Giardia/Cryptosporidium Enzyme-Linked Immunosorbent Assay (ELISA) for the detection of Giardia lamblia and Cryptosporidium spp. antigens in human feces Instruction Manual Test kit for 96 determinations
CoproELISA Giardia/Cryptosporidium Enzyme-Linked Immunosorbent Assay (ELISA) for the detection of Giardia lamblia and Cryptosporidium spp. antigens in human feces Instruction Manual Test kit for 96 determinations
Prevalence of Intestinal Parasitic Infections in HIV-Positive Patients
 ISSN: 2319-7706 Volume 4 Number 5 (2015) pp. 269-273 http://www.ijcmas.com Original Research Article Prevalence of Intestinal Parasitic Infections in HIV-Positive Patients Vasundhara*, Haris M.Khan, Harekrishna
ISSN: 2319-7706 Volume 4 Number 5 (2015) pp. 269-273 http://www.ijcmas.com Original Research Article Prevalence of Intestinal Parasitic Infections in HIV-Positive Patients Vasundhara*, Haris M.Khan, Harekrishna
Rotavirus Test Kit. Instructions For Use. Format: Cassette Specimen: Fecal Extract Catalog Number: VEL-001-ROTA
 Rotavirus Test Kit Instructions For Use Format: Cassette Specimen: Fecal Extract Catalog Number: VEL-001-ROTA * Please read the instructions carefully before use INTENDED USE Velotest Rotavirus Test is
Rotavirus Test Kit Instructions For Use Format: Cassette Specimen: Fecal Extract Catalog Number: VEL-001-ROTA * Please read the instructions carefully before use INTENDED USE Velotest Rotavirus Test is
Catalog # OKDA00123 SUMMARY AND EXPLANATION
 Aviva Systems Biology 5754 Pacific Center Blvd., Suite 201, San Diego, CA 92121, USA Tel: 858-552-6979 Fax: 858-552-6975 www.avivasysbio.com Email: info@avivasysbio.com Giardia lamblia ELISA Kit Catalog
Aviva Systems Biology 5754 Pacific Center Blvd., Suite 201, San Diego, CA 92121, USA Tel: 858-552-6979 Fax: 858-552-6975 www.avivasysbio.com Email: info@avivasysbio.com Giardia lamblia ELISA Kit Catalog
Cryptosporidiosis. By: Nikole Stewart
 Cryptosporidiosis By: Nikole Stewart Cryptosporidiosis ("Crypto"); Etiological agent- Cryptosporidium (1) Transmission: Transmission occurs via the fecal-oral route when individuals ingest water or food
Cryptosporidiosis By: Nikole Stewart Cryptosporidiosis ("Crypto"); Etiological agent- Cryptosporidium (1) Transmission: Transmission occurs via the fecal-oral route when individuals ingest water or food
Rapid-VIDITEST Shigella dysenteriae
 Rapid-VIDITEST Shigella dysenteriae One step Shigella dysenteriae test for the qualitative detection of Shigella dysenteriae in faeces. Instruction manual Producer: VIDIA spol. s r.o., Nad Safinou II 365,
Rapid-VIDITEST Shigella dysenteriae One step Shigella dysenteriae test for the qualitative detection of Shigella dysenteriae in faeces. Instruction manual Producer: VIDIA spol. s r.o., Nad Safinou II 365,
Rapid-VIDITEST. Astrovirus Card
 Rapid-VIDITEST Astrovirus Card One step Astrovirus Card Test. Instruction manual INTENDED USE: The Rapid-VIDITEST Astrovirus Card is a one step coloured chromatographic immunoassay for the qualitative
Rapid-VIDITEST Astrovirus Card One step Astrovirus Card Test. Instruction manual INTENDED USE: The Rapid-VIDITEST Astrovirus Card is a one step coloured chromatographic immunoassay for the qualitative
Study of Intestinal Protozoan Parasites in Rural Inhabitants of Mazandaran Province, Northern Iran
 Original Article Study of Intestinal Protozoan Parasites in Rural Inhabitants of Mazandaran Province, Northern Iran EB Kia 1, M Hosseini 2, MR Nilforoushan 1, AR Meamar 3, *M Rezaeian 1 1 Dept. of Medical
Original Article Study of Intestinal Protozoan Parasites in Rural Inhabitants of Mazandaran Province, Northern Iran EB Kia 1, M Hosseini 2, MR Nilforoushan 1, AR Meamar 3, *M Rezaeian 1 1 Dept. of Medical
PARASITOLOGY CASE HISTORY 15 (HISTOLOGY) (Lynne S. Garcia)
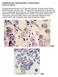 PARASITOLOGY CASE HISTORY 15 (HISTOLOGY) (Lynne S. Garcia) A biopsy was performed on a 27-year-old man with no known travel history, presenting with a perianal ulcer. The specimen was preserved in formalin
PARASITOLOGY CASE HISTORY 15 (HISTOLOGY) (Lynne S. Garcia) A biopsy was performed on a 27-year-old man with no known travel history, presenting with a perianal ulcer. The specimen was preserved in formalin
PREVALENCE OF INTESTINAL PARASITES IN HIV-POSITIVE/AIDS PATIENTS. O.O Oguntibeju
 Malaysian Journal of Medical Sciences, Vol. 13, No. 1, January 2006 (68-73) ORIGINAL ARTICLE PREVALENCE OF INTESTINAL PARASITES IN HIV-POSITIVE/AIDS PATIENTS O.O Oguntibeju School of Health Technology,
Malaysian Journal of Medical Sciences, Vol. 13, No. 1, January 2006 (68-73) ORIGINAL ARTICLE PREVALENCE OF INTESTINAL PARASITES IN HIV-POSITIVE/AIDS PATIENTS O.O Oguntibeju School of Health Technology,
Washington State Department of Health (DOH) Cryptosporidium in Drinking Water Position Paper. Purpose
 Washington State Department of Health (DOH) Cryptosporidium in Drinking Water Position Paper Purpose Cryptosporidium is a micro-organism which has gained increased public health significance as a result
Washington State Department of Health (DOH) Cryptosporidium in Drinking Water Position Paper Purpose Cryptosporidium is a micro-organism which has gained increased public health significance as a result
E. Histolytica IgG (Amebiasis)
 DIAGNOSTIC AUTOMATION, INC. 21250 Califa Street, Suite 102 and 116, Woodland hills, CA 91367 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com
DIAGNOSTIC AUTOMATION, INC. 21250 Califa Street, Suite 102 and 116, Woodland hills, CA 91367 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com
Prevalence of Intestinal Protozoan among Students Visiting Wollega University Students Clinic
 Research article Prevalence of Intestinal Protozoan among Students Visiting Wollega University Students Clinic 1 Asegid Abebe and 2 Desalegn Amenu 1 College of Natural and Computational Science, Wollega
Research article Prevalence of Intestinal Protozoan among Students Visiting Wollega University Students Clinic 1 Asegid Abebe and 2 Desalegn Amenu 1 College of Natural and Computational Science, Wollega
CRYPTOSPORIDIUM SPECIE AS A CAUSATIVE AGENT OF DIARRHOEA IN UNIVERSITY OF MAIDUGURI TEACHING HOSPITAL,MAIDUGURI.
 CRYPTOSPORIDIUM SPECIE AS A CAUSATIVE AGENT OF DIARRHOEA IN UNIVERSITY OF MAIDUGURI TEACHING HOSPITAL,MAIDUGURI 1 BALLA, HJ, 1 MM ASKIRA 1 Department of medical laboratory science, college of medicine,
CRYPTOSPORIDIUM SPECIE AS A CAUSATIVE AGENT OF DIARRHOEA IN UNIVERSITY OF MAIDUGURI TEACHING HOSPITAL,MAIDUGURI 1 BALLA, HJ, 1 MM ASKIRA 1 Department of medical laboratory science, college of medicine,
Origination Date: 25 Mar 2004 Total Pages: Effective Date: 18 Sep 2012 SOP Number LTC-SOP-17 v2.0. Network Name, Title Signature Date
 Title: Stool Processing and Cryptosporidium/Giardia Origination Date: 25 Mar 2004 Total Pages: Effective Date: 18 Sep 2012 SOP Number LTC-SOP-17 v2.0 Prepared By: Bill Kabat and Nancy Isham of the Supersedes
Title: Stool Processing and Cryptosporidium/Giardia Origination Date: 25 Mar 2004 Total Pages: Effective Date: 18 Sep 2012 SOP Number LTC-SOP-17 v2.0 Prepared By: Bill Kabat and Nancy Isham of the Supersedes
Sure-Vue Signature Cryptosporidium/Giardia Test
 SAMPLE PROCEDURE This procedure is provided to Fisher Healthcare customers to assist with the development of laboratory procedures. This document was derived from, and was current with, the instructions
SAMPLE PROCEDURE This procedure is provided to Fisher Healthcare customers to assist with the development of laboratory procedures. This document was derived from, and was current with, the instructions
Rapid-VIDITEST Enterovirus
 Rapid-VIDITEST Enterovirus A rapid one step Enterovirus Card test for the qualitative detection of Enterovirus antigens in human feces. Instruction manual Producer: VIDIA spol. s r.o., Nad Safinou II 365,
Rapid-VIDITEST Enterovirus A rapid one step Enterovirus Card test for the qualitative detection of Enterovirus antigens in human feces. Instruction manual Producer: VIDIA spol. s r.o., Nad Safinou II 365,
Chlamydia trachomatis (CHLa)Test Kit
 Chlamydia trachomatis (CHLa)Test Kit Instructions For Use Format: Cassette Specimen: Urethral/Genital Swab Catalog Number: VEL-001-CHLa * Please read the instructions carefully before use INTENDED USE
Chlamydia trachomatis (CHLa)Test Kit Instructions For Use Format: Cassette Specimen: Urethral/Genital Swab Catalog Number: VEL-001-CHLa * Please read the instructions carefully before use INTENDED USE
Rapid-VIDITEST. Rota-Adeno. One Step Rotavirus and Adenovirus Antigen Blister test. Instruction manual
 Rapid-VIDITEST Rota-Adeno One Step Rotavirus and Adenovirus Antigen Blister test. Instruction manual Producer: VIDIA spol. s r.o., Nad Safinou II 365, Vestec, 252 42 Jesenice, Czech Republic, Tel.: +420
Rapid-VIDITEST Rota-Adeno One Step Rotavirus and Adenovirus Antigen Blister test. Instruction manual Producer: VIDIA spol. s r.o., Nad Safinou II 365, Vestec, 252 42 Jesenice, Czech Republic, Tel.: +420
Rapid-VIDITEST C. difficile Ag (GDH) Card/Blister
 Li StarFish S.r.l. Via Cavour, 35-20063 Cernusco S/N (MI), Italy Tel. +39-02-92150794 - Fax. +39-02-92157285 info@listarfish.it -www.listarfish.it Rapid-VIDITEST C. difficile Ag (GDH) Card/Blister One
Li StarFish S.r.l. Via Cavour, 35-20063 Cernusco S/N (MI), Italy Tel. +39-02-92150794 - Fax. +39-02-92157285 info@listarfish.it -www.listarfish.it Rapid-VIDITEST C. difficile Ag (GDH) Card/Blister One
A New Multiplex Real-time PCR Assay For Detection Of Intestinal Parasites
 A New Multiplex Real-time PCR Assay For Detection Of Intestinal Parasites Dr. Andreas Simons Worldwide provider of diagnostic assay solutions Offers a variety of test kit methodologies R-Biopharm Headquarters
A New Multiplex Real-time PCR Assay For Detection Of Intestinal Parasites Dr. Andreas Simons Worldwide provider of diagnostic assay solutions Offers a variety of test kit methodologies R-Biopharm Headquarters
Comparison of two Methods in the Detection of Cryptosporidium in Pigs in Ogun State, Nigeria
 : G Veterinary Science and Veterinary Medicine Volume 15 Issue 1 Version 1.0 Year 2015 Type: Double Blind Peer Reviewed International Research Journal Publisher: Global Journals Inc. (USA) Online ISSN:
: G Veterinary Science and Veterinary Medicine Volume 15 Issue 1 Version 1.0 Year 2015 Type: Double Blind Peer Reviewed International Research Journal Publisher: Global Journals Inc. (USA) Online ISSN:
MALARIA P. FALCIPARUM / P. VIVAX
 MALARIA P. FALCIPARUM / P. VIVAX 1. EXPLANATION OF THE TEST: Malaria is a serious, sometimes fatal, parasitic disease characterized by fever, chills, and anemia and is caused by a parasite that is transmitted
MALARIA P. FALCIPARUM / P. VIVAX 1. EXPLANATION OF THE TEST: Malaria is a serious, sometimes fatal, parasitic disease characterized by fever, chills, and anemia and is caused by a parasite that is transmitted
PARASITIC INFECTIONS AMONG ORANG ASLI (ABORIGINE) IN THE CAMERON HIGHLANDS, MALAYSIA
 PARASITIC INFECTIONS AMONG ORANG ASLI (ABORIGINE) IN THE CAMERON HIGHLANDS, MALAYSIA S Lokman Hakim 1, CC Gan 1, K Malkit 1, My Noor Azian 1, CK Chong 2, N Shaari 2, W Zainuddin 3, CN Chin 2, Y Sara 2
PARASITIC INFECTIONS AMONG ORANG ASLI (ABORIGINE) IN THE CAMERON HIGHLANDS, MALAYSIA S Lokman Hakim 1, CC Gan 1, K Malkit 1, My Noor Azian 1, CK Chong 2, N Shaari 2, W Zainuddin 3, CN Chin 2, Y Sara 2
Specimens: Evidence for Low Sensitivity of Current Diagnostic Methods
 JOURNAL OF CLINICAL MICROBIOLOGY, July 1991, p. 1323-1327 0095-1137/91/071323-05$02.00/0 Copyright D 1991, American Society for Microbiology Vol. 29, No. 7 Threshold of Detection of Cryptosporidium Oocysts
JOURNAL OF CLINICAL MICROBIOLOGY, July 1991, p. 1323-1327 0095-1137/91/071323-05$02.00/0 Copyright D 1991, American Society for Microbiology Vol. 29, No. 7 Threshold of Detection of Cryptosporidium Oocysts
Human Influenza A (Swine Flu) Rapid test
 Human Influenza A (Swine Flu) Rapid test Cat.No: DTSXY-Z9 Lot. No. (See product label) Size 20T Intended use The Influenza A (Swine Flu) test is a rapid chromatographic immunoassay for the qualitative
Human Influenza A (Swine Flu) Rapid test Cat.No: DTSXY-Z9 Lot. No. (See product label) Size 20T Intended use The Influenza A (Swine Flu) test is a rapid chromatographic immunoassay for the qualitative
Rapid-VIDITEST Swine Flu
 Rapid-VIDITEST Swine Flu One Step Influenza type A Antigen Card test. Instruction manual Producer: VIDIA spol. s r.o., Nad Safinou II 365, 252 50 Vestec, Czech Republic, Tel.: +420 261 090 565, www.vidia.cz
Rapid-VIDITEST Swine Flu One Step Influenza type A Antigen Card test. Instruction manual Producer: VIDIA spol. s r.o., Nad Safinou II 365, 252 50 Vestec, Czech Republic, Tel.: +420 261 090 565, www.vidia.cz
Treponema Pallidum (TP) Antibody Test
 Treponema Pallidum (TP) Antibody Test Instructions For Use Format: Cassette For: Catalog Number: VEL-001-TP Specimen: Serum/Plasma * Please read the instructions carefully before use INTENDED USE Velotest
Treponema Pallidum (TP) Antibody Test Instructions For Use Format: Cassette For: Catalog Number: VEL-001-TP Specimen: Serum/Plasma * Please read the instructions carefully before use INTENDED USE Velotest
Prevalence of Cryptosporidium parvum among children in Iraq
 American Journal of Life Sciences 2013; 1(6): 256-260 Published online December 10, 2013 (http://www.sciencepublishinggroup.com/j/ajls) doi: 10.11648/j.ajls.20130106.13 Prevalence of Cryptosporidium parvum
American Journal of Life Sciences 2013; 1(6): 256-260 Published online December 10, 2013 (http://www.sciencepublishinggroup.com/j/ajls) doi: 10.11648/j.ajls.20130106.13 Prevalence of Cryptosporidium parvum
American Association of Bioanalysts 5615 Kirby Drive, Suite 870 Houston, TX
 Q3 2018 Parasitology American Association of Bioanalysts 5615 Kirby Drive, Suite 870 Houston, TX 77005 800-234-5315 281-436-5357 Specimen 1 Referees Extent 1 Extent 2 Total Few to 534 Giardia lamblia Many
Q3 2018 Parasitology American Association of Bioanalysts 5615 Kirby Drive, Suite 870 Houston, TX 77005 800-234-5315 281-436-5357 Specimen 1 Referees Extent 1 Extent 2 Total Few to 534 Giardia lamblia Many
Journal of Nepal Health Research Council Vol. 2 No. 1 April 2004
 Enteric Parasitosis in Patients with Human Immunodeficiency Virus (HIV) Infection and Acquired Immunodeficiency Syndrome (AIDS) in Nepal Sapkota D a, Ghimire P a and Manandhar S a Introduction Objectives
Enteric Parasitosis in Patients with Human Immunodeficiency Virus (HIV) Infection and Acquired Immunodeficiency Syndrome (AIDS) in Nepal Sapkota D a, Ghimire P a and Manandhar S a Introduction Objectives
Rapid-VIDITEST. Rota-Adeno Blister. One Step Rotavirus and Adenovirus Antigen Blister test. Instruction manual
 Rapid-VIDITEST Rota-Adeno Blister One Step Rotavirus and Adenovirus Antigen Blister test. Instruction manual INTENDED USE: The Rapid-VIDITEST Rota-Adeno Blister test is a rapid chromatographic immunoassay
Rapid-VIDITEST Rota-Adeno Blister One Step Rotavirus and Adenovirus Antigen Blister test. Instruction manual INTENDED USE: The Rapid-VIDITEST Rota-Adeno Blister test is a rapid chromatographic immunoassay
Rapid-VIDITEST FOB Blister
 Rapid-VIDITEST FOB Blister One Step Fecal Occult Blood Blister test. Instruction manual Producer: VIDIA spol. s r.o., Nad Safinou II 365, Vestec, 252 42 Jesenice, Czech Republic, Tel.: +420 261 090 565,
Rapid-VIDITEST FOB Blister One Step Fecal Occult Blood Blister test. Instruction manual Producer: VIDIA spol. s r.o., Nad Safinou II 365, Vestec, 252 42 Jesenice, Czech Republic, Tel.: +420 261 090 565,
ccess safe drinking wa r is everyone s right Protozoans that cause diarrheal disease
 ccess safe drinking wa r is everyone s right Protozoa: Protozoans that cause diarrheal disease 1. Giardia lamblia 2. Entameba histolytica 3. Cryptosporidium parvum 4. Cyclospora cayetanensis 1 Giardia
ccess safe drinking wa r is everyone s right Protozoa: Protozoans that cause diarrheal disease 1. Giardia lamblia 2. Entameba histolytica 3. Cryptosporidium parvum 4. Cyclospora cayetanensis 1 Giardia
Amebiasis Ab E. histolytica IgG
 Code: HP003 96 Test distribuito da: Amebiasis Ab E. histolytica IgG Amebiasis E. histolytica lgg test is an enzyme immunoassay for the detection of specific lgg antibodies against Anti - Entamoeba histolytica
Code: HP003 96 Test distribuito da: Amebiasis Ab E. histolytica IgG Amebiasis E. histolytica lgg test is an enzyme immunoassay for the detection of specific lgg antibodies against Anti - Entamoeba histolytica
Prevalence of Giardiasis in Patients Attending Tertiary Care Hospital in Northern India
 ISSN: 2319-7706 Volume 4 Number 5 (2015) pp. 339-344 http://www.ijcmas.com Original Research Article Prevalence of Giardiasis in Patients Attending Tertiary Care Hospital in Northern India Sangram Singh
ISSN: 2319-7706 Volume 4 Number 5 (2015) pp. 339-344 http://www.ijcmas.com Original Research Article Prevalence of Giardiasis in Patients Attending Tertiary Care Hospital in Northern India Sangram Singh
Recent Diagnostic Methods for Intestinal Parasitic Infections
 Recent Diagnostic Methods for Intestinal Parasitic Infections By Dr. Doaa Abdel Badie Salem Lecturer of Medical Parasitology, Mansoura Faculty of Medicine Agenda Intestinal parasites. Traditional Diagnostic
Recent Diagnostic Methods for Intestinal Parasitic Infections By Dr. Doaa Abdel Badie Salem Lecturer of Medical Parasitology, Mansoura Faculty of Medicine Agenda Intestinal parasites. Traditional Diagnostic
IVD. DRG Toxocara canis ELISA (EIA-3518) Revised 12 Feb rm (Vers. 6.1)
 Please use only the valid version of the package insert provided with the kit. Intended Use For the qualitative screening of serum IgG antibodies to Toxocara using an Enzyme-Linked Immunosorbent Assay
Please use only the valid version of the package insert provided with the kit. Intended Use For the qualitative screening of serum IgG antibodies to Toxocara using an Enzyme-Linked Immunosorbent Assay
Toxoplasma gondii IgM (Toxo IgM)
 DIAGNOSTIC AUTOMATION, INC. 21250 Califa Street, Suite 102 and116, Woodland Hills, CA 91367 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com
DIAGNOSTIC AUTOMATION, INC. 21250 Califa Street, Suite 102 and116, Woodland Hills, CA 91367 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com
Hepatitis C Virus (HCV) Antibody Test
 Hepatitis C Virus (HCV) Antibody Test Instructions For Use Format: Cassette For: Catalog Number: VEL-001-HCV Specimen: Serum/Plasma * Please read the instructions carefully before use INTENDED USE Hepatitis
Hepatitis C Virus (HCV) Antibody Test Instructions For Use Format: Cassette For: Catalog Number: VEL-001-HCV Specimen: Serum/Plasma * Please read the instructions carefully before use INTENDED USE Hepatitis
EPIDEMIOLOGY OF GIARDIASIS AND CRYPTOSPORIDIOSIS IN JAMAICA
 Am. J. Trop. Med. Hyg., 59(5), 1998, pp. 717 721 Copyright 1998 by The American Society of Tropical Medicine and Hygiene EPIDEMIOLOGY OF GIARDIASIS AND CRYPTOSPORIDIOSIS IN JAMAICA JOHN F. LINDO, VALERIE
Am. J. Trop. Med. Hyg., 59(5), 1998, pp. 717 721 Copyright 1998 by The American Society of Tropical Medicine and Hygiene EPIDEMIOLOGY OF GIARDIASIS AND CRYPTOSPORIDIOSIS IN JAMAICA JOHN F. LINDO, VALERIE
Prevalence of Intestinal Parasites in HIV Seropositive Patients with and without Diarrhoea and its Correlation with CD4 Counts
 International Journal of Current Microbiology and Applied Sciences ISSN: 2319-7706 Volume 5 Number 10 (2016) pp. 527-532 Journal homepage: http://www.ijcmas.com Original Research Article http://dx.doi.org/10.20546/ijcmas.2016.510.058
International Journal of Current Microbiology and Applied Sciences ISSN: 2319-7706 Volume 5 Number 10 (2016) pp. 527-532 Journal homepage: http://www.ijcmas.com Original Research Article http://dx.doi.org/10.20546/ijcmas.2016.510.058
Dengue IgG/IgM/NS1 Combo Rapid Test Device
 Dengue IgG/IgM/NS1 Combo Rapid Test Device Cat.No: DTSXY-L2 Lot. No. (See product label) Intended Use The Dengue IgG/IgM/NS1 Combo Rapid Test Device is a lateral flow chromatographic immunoassay for the
Dengue IgG/IgM/NS1 Combo Rapid Test Device Cat.No: DTSXY-L2 Lot. No. (See product label) Intended Use The Dengue IgG/IgM/NS1 Combo Rapid Test Device is a lateral flow chromatographic immunoassay for the
Indian Journal of Basic and Applied Medical Research; September 2014: Vol.-3, Issue- 4, P
 Original article: Comparison of stool concentration methods for detection of prevalence of enteroparasitic infection in rural tertiary care teaching hospital of Maharashtra 1 Dr. Jaishree Puri, 2 Dr. S.
Original article: Comparison of stool concentration methods for detection of prevalence of enteroparasitic infection in rural tertiary care teaching hospital of Maharashtra 1 Dr. Jaishree Puri, 2 Dr. S.
ELISA TEST FOR DETECTION OF BLASTOCYSTIS SPP. IN HUMAN FAECES: COMPARISON OF THREE METHODS
 ELISA TEST FOR DETECTION OF BLASTOCYSTIS SPP. IN HUMAN FAECES: COMPARISON OF THREE METHODS István Kucsera, Mónika Molnár, Eszter Gályász, József Danka, Erika Orosz National Center for Epidemiology, Department
ELISA TEST FOR DETECTION OF BLASTOCYSTIS SPP. IN HUMAN FAECES: COMPARISON OF THREE METHODS István Kucsera, Mónika Molnár, Eszter Gályász, József Danka, Erika Orosz National Center for Epidemiology, Department
PARATEST STABILITY STUDY, SYSTEM USED FOR DIAGNOSIS OF INTESTINAL PARASITES
 PARATEST STABILITY STUDY, SYSTEM USED FOR DIAGNOSIS OF INTESTINAL PARASITES Rev.00 January 10, 2011 1. Introduction Intestinal parasitic diseases represent a serious public health issue in underdeveloped
PARATEST STABILITY STUDY, SYSTEM USED FOR DIAGNOSIS OF INTESTINAL PARASITES Rev.00 January 10, 2011 1. Introduction Intestinal parasitic diseases represent a serious public health issue in underdeveloped
Parasitic Protozoa, Helminths, and Arthropod Vectors
 PowerPoint Lecture Slides for MICROBIOLOGY ROBERT W. BAUMAN Chapter 23 Parasitic Protozoa, Helminths, and Arthropod Vectors Parasitic Diseases Protozoan and helminthic parasites are emerging as serious
PowerPoint Lecture Slides for MICROBIOLOGY ROBERT W. BAUMAN Chapter 23 Parasitic Protozoa, Helminths, and Arthropod Vectors Parasitic Diseases Protozoan and helminthic parasites are emerging as serious
Comparism of Various Staining Techniques in the Diagnosis of Coccidian Parasitosis in HIV Infection
 Original Article Comparism of Various Staining Techniques in the Diagnosis of Coccidian Parasitosis in HIV Infection 1 2 Joseph A.A,* Popoola G.O 1. Department of Medical Microbiology and Parasitology,
Original Article Comparism of Various Staining Techniques in the Diagnosis of Coccidian Parasitosis in HIV Infection 1 2 Joseph A.A,* Popoola G.O 1. Department of Medical Microbiology and Parasitology,
Effect of Nitazoxanide in Diarrhea and Enteritis Caused by Cryptosporidium Species
 CLINICAL GASTROENTEROLOGY AND HEPATOLOGY 2006;4:320 324 Effect of Nitazoxanide in Diarrhea and Enteritis Caused by Cryptosporidium Species JEAN FRANÇOIS ROSSIGNOL,* SAMIR M. KABIL, YEHIA EL GOHARY, and
CLINICAL GASTROENTEROLOGY AND HEPATOLOGY 2006;4:320 324 Effect of Nitazoxanide in Diarrhea and Enteritis Caused by Cryptosporidium Species JEAN FRANÇOIS ROSSIGNOL,* SAMIR M. KABIL, YEHIA EL GOHARY, and
CURRENT SITUATION OF GIARDIA AND CRYPTOSPORIDIUM AMONG ORANG ASLI (ABORIGINAL) COMMUNITIES IN PAHANG, MALAYSIA
 CURRENT SITUATION OF GIARDIA AND CRYPTOSPORIDIUM AMONG ORANG ASLI (ABORIGINAL) COMMUNITIES IN PAHANG, MALAYSIA AK Mohammed Mahdy, Johari Surin, YAL Lim and MS Hesham AI-Mekhlafi Department of Parasitology,
CURRENT SITUATION OF GIARDIA AND CRYPTOSPORIDIUM AMONG ORANG ASLI (ABORIGINAL) COMMUNITIES IN PAHANG, MALAYSIA AK Mohammed Mahdy, Johari Surin, YAL Lim and MS Hesham AI-Mekhlafi Department of Parasitology,
Evaluation and comparison of PCR, Coproantigen ELISA and microscopy for diagnosis of Cryptosporidium in human diarrhoeic specimens.
 Evaluation and comparison of PCR, Coproantigen ELISA and microscopy for diagnosis of Cryptosporidium in human diarrhoeic specimens. Magda A.El-Settawy and Ghada M. Fathy. Department of Medical Parasitology,
Evaluation and comparison of PCR, Coproantigen ELISA and microscopy for diagnosis of Cryptosporidium in human diarrhoeic specimens. Magda A.El-Settawy and Ghada M. Fathy. Department of Medical Parasitology,
Leishmania Canine IgG -ELISA
 Leishmania Canine IgG -ELISA GWB-521229 www.genwaybio.com FOR RESEARCH USE ONLY Enzyme immunoassay for the qualitative detection of IgG-class antibodies against Leishmania in canine serum 1. INTRODUCTION
Leishmania Canine IgG -ELISA GWB-521229 www.genwaybio.com FOR RESEARCH USE ONLY Enzyme immunoassay for the qualitative detection of IgG-class antibodies against Leishmania in canine serum 1. INTRODUCTION
Hot Topics in Infectious Diseases: Enteric Infections in the Arctic
 Hot Topics in Infectious Diseases: Enteric Infections in the Arctic Tobey Audcent MD, FRCPC Department of Pediatrics Children s Hospital of Eastern Ontario 6 th International Meeting on Indigenous Child
Hot Topics in Infectious Diseases: Enteric Infections in the Arctic Tobey Audcent MD, FRCPC Department of Pediatrics Children s Hospital of Eastern Ontario 6 th International Meeting on Indigenous Child
See external label. 2 C-30 C for test card and Sample tube Cat.# OneStep Rotavirus Antigen RapiCard InstaTest.
 CORTEZ DIAGNOSTICS, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 USA Tel: (818) 591-3030 Fax: (818) 591-8383 E-mail: onestep@rapidtest.com Web site: www.rapidtest.com See external label
CORTEZ DIAGNOSTICS, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 USA Tel: (818) 591-3030 Fax: (818) 591-8383 E-mail: onestep@rapidtest.com Web site: www.rapidtest.com See external label
Enzyme-Linked Immunoassay for Detection of Cryptosporidium Antigens in Fecal Specimens
 JOURNAL OF CLINICAL MICROBIOLOGY, Nov. 1990, p. 2491-2495 0095-1137/90/112491-05$02.00/0 Copyright C 1990, American Society for Microbiology Vol. 28, No. 11 Enzyme-Linked Immunoassay for Detection of Cryptosporidium
JOURNAL OF CLINICAL MICROBIOLOGY, Nov. 1990, p. 2491-2495 0095-1137/90/112491-05$02.00/0 Copyright C 1990, American Society for Microbiology Vol. 28, No. 11 Enzyme-Linked Immunoassay for Detection of Cryptosporidium
Rapid-VIDITEST. Strep A Blister. One step Strep A Blister for the detection of Group A Streptococcal antigen from throat swabs or culture.
 Rapid-VIDITEST Strep A Blister One step Strep A Blister for the detection of Group A Streptococcal antigen from throat swabs or culture. Instruction manual Producer: VIDIA spol. s r.o., Nad Safinou II
Rapid-VIDITEST Strep A Blister One step Strep A Blister for the detection of Group A Streptococcal antigen from throat swabs or culture. Instruction manual Producer: VIDIA spol. s r.o., Nad Safinou II
IMMUNOGLOBULIN LEVELS IN SERUM AND CERVICOVAGINAL SECRETIONS OF PATIENTS INFECTED WITH TRICHOMONAS VAGINALIS
 Journal of Al-Nahrain University Vol13 (2), June, 2010, pp147-151 Science IMMUNOGLOBULIN LEVELS IN SERUM AND CERVICOVAGINAL SECRETIONS OF PATIENTS INFECTED WITH TRICHOMONAS VAGINALIS Ekhlas Mushrif *,
Journal of Al-Nahrain University Vol13 (2), June, 2010, pp147-151 Science IMMUNOGLOBULIN LEVELS IN SERUM AND CERVICOVAGINAL SECRETIONS OF PATIENTS INFECTED WITH TRICHOMONAS VAGINALIS Ekhlas Mushrif *,
Campylobacter Antigen ELISA Kit
 Campylobacter Antigen ELISA Kit Catalog Number KA3204 96 assays Version: 01 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
Campylobacter Antigen ELISA Kit Catalog Number KA3204 96 assays Version: 01 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
H.pylori IgA Cat #
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
District NTD Training module 9 Learners Guide
 District NTD Training module 9 Learners Guide Session 3: Diagnostic and laboratory tools for filarial worms, Soil- Transmitted Helminths and Schistosomiasis Key message addressed to communities: Community
District NTD Training module 9 Learners Guide Session 3: Diagnostic and laboratory tools for filarial worms, Soil- Transmitted Helminths and Schistosomiasis Key message addressed to communities: Community
RIDASCREEN Risk Material 10/5
 RIDASCREEN Risk Material 10/5 Enzymimmunoassay zur quantitativen Bestimmung von Risikomaterial (ZNS) in / auf rohem Fleisch sowie auf kontaminierten Oberflächen Enzyme immunoassay for the quantitative
RIDASCREEN Risk Material 10/5 Enzymimmunoassay zur quantitativen Bestimmung von Risikomaterial (ZNS) in / auf rohem Fleisch sowie auf kontaminierten Oberflächen Enzyme immunoassay for the quantitative
Toxoplasma gondii IgM ELISA Kit
 Toxoplasma gondii IgM ELISA Kit Catalog Number KA0226 96 assays Version: 01 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
Toxoplasma gondii IgM ELISA Kit Catalog Number KA0226 96 assays Version: 01 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
Toxocara. Cat # Toxocara ELISA. Enzyme Linked Immunosorbent Assay. ELISA-Indirect; Antigen Coated Plate
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
Toxoplasma gondii IgM ELISA Kit
 Toxoplasma gondii IgM ELISA Kit Catalog Number KA0226 96 assays Version: 02 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
Toxoplasma gondii IgM ELISA Kit Catalog Number KA0226 96 assays Version: 02 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
Please use only the valid version of the package insert provided with the kit.
 Please use only the valid version of the package insert provided with the kit. Intended Use For the qualitative screening of serum IgG antibodies to Trichinella using an Enzyme Linked Immunoabsorbant Assay
Please use only the valid version of the package insert provided with the kit. Intended Use For the qualitative screening of serum IgG antibodies to Trichinella using an Enzyme Linked Immunoabsorbant Assay
HAV IgG/IgM Rapid Test
 HAV IgG/IgM Rapid Test Cat. No.:DTS649 Pkg.Size: Intended use CD HAV IgG/IgM Rapid Test is a solid phase immunochromatographic assay for the rapid, qualitative and differential detection of IgG and IgM
HAV IgG/IgM Rapid Test Cat. No.:DTS649 Pkg.Size: Intended use CD HAV IgG/IgM Rapid Test is a solid phase immunochromatographic assay for the rapid, qualitative and differential detection of IgG and IgM
EPIDEMIOLOGICAL STUDY: LABORATORY DATA MINING IN SOUTH OF JORDAN
 American Journal of Infectious Diseases 10 (3): 137-141, 2014 ISSN: 1553-6203 2014 H. Nawafleh et al., This open access article is distributed under a Creative Commons Attribution (CC-BY) 3.0 license doi:10.3844/ajidsp.2014.137.141
American Journal of Infectious Diseases 10 (3): 137-141, 2014 ISSN: 1553-6203 2014 H. Nawafleh et al., This open access article is distributed under a Creative Commons Attribution (CC-BY) 3.0 license doi:10.3844/ajidsp.2014.137.141
Chlamydia Trachomatis IgG ELISA Kit
 Chlamydia Trachomatis IgG ELISA Kit Catalog Number KA0959 96 assays Version: 03 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
Chlamydia Trachomatis IgG ELISA Kit Catalog Number KA0959 96 assays Version: 03 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
25(OH) Vitamin D ELISA (BD-220BA), 192 Tests
 INTENDED USE The 25-hydroxy (25-OH) Vitamin D ELISA is intended for the quantitative determination of total 25-OH Vitamin D in human serum and Plasma. SUMMARY AND EXPLANATION Vitamin D is a steroid hormone
INTENDED USE The 25-hydroxy (25-OH) Vitamin D ELISA is intended for the quantitative determination of total 25-OH Vitamin D in human serum and Plasma. SUMMARY AND EXPLANATION Vitamin D is a steroid hormone
Chlamydia Trachomatis IgM ELISA Kit
 Chlamydia Trachomatis IgM ELISA Kit Catalog Number KA0960 96 assays Version: 03 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
Chlamydia Trachomatis IgM ELISA Kit Catalog Number KA0960 96 assays Version: 03 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
Rapid-VIDITEST. Strep A Card. One step Strep A Card for the detection of Group A Streptococcal antigen from throat swabs or culture.
 Rapid-VIDITEST Strep A Card One step Strep A Card for the detection of Group A Streptococcal antigen from throat swabs or culture. Instruction manual Producer: VIDIA spol. s r.o., Nad Safinou II 365, Vestec,
Rapid-VIDITEST Strep A Card One step Strep A Card for the detection of Group A Streptococcal antigen from throat swabs or culture. Instruction manual Producer: VIDIA spol. s r.o., Nad Safinou II 365, Vestec,
For Vets General Information Prevalence and Risk Factors Humans
 For Vets General Information Cryptosporidium spp. are intestinal protozoal parasites of animals and humans that cause the disease cryptosporidiosis. The primary clinical sign of infection is diarrhea,
For Vets General Information Cryptosporidium spp. are intestinal protozoal parasites of animals and humans that cause the disease cryptosporidiosis. The primary clinical sign of infection is diarrhea,
IBD EZ VUE I. PRINCIPLE
 IBD EZ VUE INTENDED USE The IBD EZ VUE test is an immunochromatographic test for the qualitative detection of elevated levels of lactoferrin, a marker for fecal leukocytes and an indicator of intestinal
IBD EZ VUE INTENDED USE The IBD EZ VUE test is an immunochromatographic test for the qualitative detection of elevated levels of lactoferrin, a marker for fecal leukocytes and an indicator of intestinal
Anton van Leeuwenhoek. Protozoa: This is what he saw in his own stool sample. Morphology 10/14/2009. Protozoans that cause diarrheal disease
 Access to safe drinking water is everyone s right Anton van Leeuwenhoek Protozoa: Protozoans that cause diarrheal disease This is what he saw in his own stool sample 1. Giardia lamblia 2. Entameba histolytica
Access to safe drinking water is everyone s right Anton van Leeuwenhoek Protozoa: Protozoans that cause diarrheal disease This is what he saw in his own stool sample 1. Giardia lamblia 2. Entameba histolytica
H.Pylori IgG
 DIAGNOSTIC AUTOMATION, INC. 21250 Califa Street, Suite 102 and116, Woodland Hills, CA 91367 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com
DIAGNOSTIC AUTOMATION, INC. 21250 Califa Street, Suite 102 and116, Woodland Hills, CA 91367 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com
CDIA TM Rubella IgG/IgM Rapid Test Kit
 CDIA TM Rubella IgG/IgM Rapid Test Kit Cat.No: DTSJZ024 Lot. No. (See product label) Intended Use The CDIA TM Rubella IgG/IgM Rapid Test Kit is a rapid chromatographic immunoassay for the qualitative detection
CDIA TM Rubella IgG/IgM Rapid Test Kit Cat.No: DTSJZ024 Lot. No. (See product label) Intended Use The CDIA TM Rubella IgG/IgM Rapid Test Kit is a rapid chromatographic immunoassay for the qualitative detection
Access to safe drinking water is everyone s right. Protozoans that cause diarrheal disease
 Access to safe drinking water is everyone s right Protozoa: Protozoans that cause diarrheal disease 1. Giardia lamblia 2. Entameba histolytica 3. Cryptosporidium parvum 4. Cyclospora cayetanensis 1 Giardia
Access to safe drinking water is everyone s right Protozoa: Protozoans that cause diarrheal disease 1. Giardia lamblia 2. Entameba histolytica 3. Cryptosporidium parvum 4. Cyclospora cayetanensis 1 Giardia
Molecular Epidemiology of Cryptosporidiosis in Iranian Children, Tehran, Iran
 Tehran University of Medical Sciences Publication http:// tums.ac.ir Original Article Iranian J Parasitol Open access Journal at http:// ijpa.tums.ac.ir Iranian Society of Parasitology http:// isp.tums.ac.ir
Tehran University of Medical Sciences Publication http:// tums.ac.ir Original Article Iranian J Parasitol Open access Journal at http:// ijpa.tums.ac.ir Iranian Society of Parasitology http:// isp.tums.ac.ir
H.Pylori IgG Cat # 1503Z
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
Influenza A IgG ELISA
 Influenza A IgG ELISA For the qualitative determination of IgG-class antibodies against Influenza A virus in human serum or plasma (citrate, heparin). For Research Use Only. Not For Use In Diagnostic Procedures.
Influenza A IgG ELISA For the qualitative determination of IgG-class antibodies against Influenza A virus in human serum or plasma (citrate, heparin). For Research Use Only. Not For Use In Diagnostic Procedures.
PROTOZOAN ENTERIC INFECTION IN AIDS RELATED DIARRHEA IN THAILAND
 PROTOZOAN ENTERIC INFECTION IN AIDS RELATED DIARRHEA IN THAILAND Duangdao Waywa 1, Siriporn Kongkriengdaj 1, Suparp Chaidatch 1, Surapee Tiengrim 1 Boonchai Kowadisaiburana 2, Suchada Chaikachonpat 1,
PROTOZOAN ENTERIC INFECTION IN AIDS RELATED DIARRHEA IN THAILAND Duangdao Waywa 1, Siriporn Kongkriengdaj 1, Suparp Chaidatch 1, Surapee Tiengrim 1 Boonchai Kowadisaiburana 2, Suchada Chaikachonpat 1,
Amantadine ELISA Test Kit
 Amantadine ELISA Test Kit Catalog No. LSY-10052 1. Principle This test kit is based on the competitive enzyme immunoassay for the detection of Amantadine. The coupling antigen is pre-coated on the micro-well
Amantadine ELISA Test Kit Catalog No. LSY-10052 1. Principle This test kit is based on the competitive enzyme immunoassay for the detection of Amantadine. The coupling antigen is pre-coated on the micro-well
