REFLEX TESTING LIST March 2012
|
|
|
- Sharleen Charles
- 5 years ago
- Views:
Transcription
1 Aeromonas/Plesiomonas Positive findings Organism ID & Susceptibility April 2011 Culture *AFB Culture and Stain Positive findings Organism ID & Susceptibility Nov *AFB Culture Only Positive findings Organism ID & Susceptibility Nov AFB Stain Positive findings PCR testing to determine if July 2010 Mycobacteria tuberculosis complex vs. nontuberculosis Mycobacteria AFP Amniotic Fluid Abnormal AFP Acetylcholinesterase and Fetal July 1998 Hemoglobin ANA Panel 1 Positive ANA ANA Titer and Pattern (IFA), March 2002 DNA, ENA Antibody Panel (RNP, SSA, SSB, Centromere, Jo1, Ribosomal RNP, Smith, Scleroderma IgG and Histone Antibodies) DNA Positive: Crithidia will be performed ANA Panel 2 Positive ANA ANA Titer Nov ANA Panel 3 Positive ANA DNA and ENA Antibody Panel March 2002 (RNP, SSA, SSB, Centromere, Jo1, Ribosomal RNP, Smith, Scleroderma IgG and Histone Antibodies) DNA Positive: Crithidia will be performed *Anaerobe Culture and Stain Positive findings Organism ID Nov Anti Platelet Factor 4 Antibody OD > Platelet Antibody / Heparin July 2002 *Blood Culture Positive findings Organism ID & Susceptibility Nov Blood Culture Positive for gram-positive Staphylococcus aureus FISH Nov 2007 cocci in clusters Blood Culture Yeast Yeast FISH Nov 2008 *Blood Culture, Special Positive findings Organism ID & Susceptibility Nov Nov *Campylobacter Culture Positive findings Organism ID *Complete Blood Count WBC > 20.0 KuL or WBC < Differential Nov KuL or Platelet counts > 1,000 KuL Complete Blood Count (BECBC) At Beachwood Family Health Center: When % CBC with Differential (performed on Main Campus) Jan Page 1 of 6
2 granulocytes = **** (unable to calculate) Cryoglobulin with Identification Cryoglobulin > 500 Cryoglobulin Identification Nov micrograms/ml *CSF Culture and Stain Positive findings Organism ID & Susceptibility Nov Cystic Fibrosis Carrier Screen If the RII7H mutation is Cystic Fibrosis Poly (T) Variant Jan detected DNA Antibody with DNA > 74 IU/mL Crithidia Lucillae Nov Confirmation Endomysial Antibody Positive findings Titer May 2003 FISH for HER2 Amplification CEP17 mean > 3.0 Reflex FISH for HER2 Amplification (D17S122) FISH for HER2 If HER2 is equivocal HER2(4B5) by Immunohistochemistry FISH for Plasma Cell If IGH break-apart is positive FISH for t(11;14), t(4;14), and Myeloma t(14;16) February 2011 July 2007 January 2012 *Fungal Blood Culture Positive findings Organism ID & Susceptibility Nov *Fungal CSF Culture/CAD Positive findings Organism ID Nov *Fungal Culture Positive findings Organism ID Nov *Fungus Screen Positive findings Organism ID Nov Gram Stain Hb Screen by HPLC Beaded, gram-positive filamentous and branching bacteria are present Abnormal hemoglobin or abnormal distribution of hemoglobin Partial acid fast stain and Nocardia culture, unless these have already been ordered. Alkaline Electrophoresis, Acid Agar, sickle solubility, Acid Globin Chains, Unstable Hgb, Hb H, Professional Interpretation April 2009 May, 2009 Helicobacter pylori Culture Positive findings Organism ID Aug Hepatitis B Surface Antigen Positive findings Hepatitis B Surface Antigen Sept 2003 confirmation Hepatitis C Antibody EIA Positive findings Qualitative Hepatitis C RNA Sept 2003 HER2 (4B5) by IHC for GI Equivocal FISH for HER2 August 2011 cases Herpes Type Culture Presumptive positive Immunofluorescence stain April 2001 *HIV-1/2 Antibody EIA Positive screen If the HIV-1 Western Blot HIV-1 Western Blot Dec Page 2 of 6
3 negative or indeterminate Positive screen *HIV Screen for Post Exposure EIA HIV-1 Western Blot April 2002, Modified If the HIV-1 Western Blot January 2006 negative or indeterminate HLA-B27 Indeterminant HLA-B27 PCR March 2010 Insulin and Insulin Antibody Positive Insulin Antibody Free & Total Insulin Nov Lamellar Body Count LBC = 15,000 40, 000 L/S Ratio with Phosphatidyl Oct 2011 Gycerol (PG) *Legionella Culture and PCR Positive findings Organism ID Nov *Legionella Culture Positive findings Organism ID Nov *Lipid Panel, Basic Triglyceride > 400 Direct LDL Nov *Miscellaneous Culture Positive findings Organism ID & Susceptibility Nov *Miscellaneous Culture and Positive findings Organism ID & Susceptibility Nov Stain *Miscellaneous GC Screen Positive findings Organism ID Nov *Miscellaneous Screen Positive findings Organism ID & Susceptibility Nov Mitochondrial Antibody Screen Positive Findings Titer Feb *Mycoplasma hominis Culture Positive findings Organism ID Nov Neutrophil Cytoplasmic Positive or questionable P or C-ANCA and P-ANCA by EIA Oct, 2011 Antibody C ANCA by IFA *Nocardia Culture Only Positive findings Organism ID Nov *Nocardia Culture and Stain Positive findings Organism ID Nov Orthopedic Implant Culture Positive findings Organism ID & Susceptibility Nov Parainfluenzae 1, 2, 3 Culture Presumptive positive Immunofluorescence stain April 2001 Parietal Cell Antibody Positive findings Titer Feb Protein Electrophoresis, Serum, with IFE Protein Electrophoresis, Urine, with IFE If M protein is identified If M protein is identified Monoclonal protein analysis by IFE (Immunofixation Electrophoresis) Monoclonal protein analysis by IFE (Immunofixation Electrophoresis) July 2002 July 2002 Page 3 of 6
4 *Respiratory Culture and Stain Positive findings Organism ID & Susceptibility Nov *Respiratory Culture Positive findings Organism ID & Susceptibility Nov *Respiratory Culture Screen Positive findings Organism ID & Susceptibility Nov *Respiratory Culture, Special Positive findings Organism ID & Susceptibility Nov *Respiratory GC Screen Positive findings Organism ID Nov RPR Result of reactive RPR Quantitative June 2002 RPR with Titer (RPRT) Result of reactive RPR Quantitative Nov 2009 *Sickle Cell Preparation Positive findings Hb Screen by HPLC, Cellulose Nov Acetate Screen and Acid Agar May 2009 Smooth Muscle Antibody Positive findings Titer Feb *Stool Culture Positive findings Organism ID & Susceptibility Nov Syphilis IgG with Confirmation Reactive findings RPR with titer, if RPRT is Dec 2010 nonreactive, FTA Antibody test. *Throat Culture, Routine Positive findings Organism ID Nov *Tissue Culture and Stain Positive findings Organism ID & Susceptibility Nov Type and Screen Type and Screen and SCR (30D) Type and Screen, Prenatal Type and Screen, Predelivery ABO/RH Indirect Antiglobulin Test When certain clinical conditions are met: Conditions: Patients for whom prophylactic phenotype matching may be indicated, including sickle cell anemia, thalassemia, and any other medical condition requiring long-term Red Cell transfusion support clinically significant alloantibodies for whom standard serological typing cannot be performed, e.g., due to positive direct Human Erythrocyte Antigen Nov 2011 Page 4 of 6
5 antiglobulin test or recent transfusion serological reactivity that interferes with exclusion of clinically significant antibodies, including autoantibodies, high titer low avidity antibodies, and selected cases of antibody reactivity with no apparent specificity suspected antibody to a high frequency antigen, or other antigen for which typing sera are not readily available discrepancies on serological typing Ureaplasma sp. Culture Positive findings Organism ID Feb 2008 *Urinalysis Only Positive dipstick for Microscopic Nov hemoglobin/blood, leukocyte esterase and/or protein *Urine Culture Positive findings Organism ID & Susceptibility Nov *Urine Culture/Special Positive findings Organism ID & Susceptibility Nov Vibrio Culture Positive findings Organism ID & Susceptibility April 2011 Yersinia Culture Positive findings Organism ID & Susceptibility April 2011 Page 5 of 6
6 The following panels contain tests that, under certain conditions, may result in an order for an additional test(s) placed by and at the discretion of the Clinical Pathologist. Test Flow Cytometric Immunophenotyping (Acute Leukemia Markers) Immunoglobulin Gene Rearrangement (PCR) Biomed2 T-cell receptor gene rearrangement (PCR) Biomed 2 Hemoglobin Screen by HPLC Hypercoagulation Diagnostic Interpretive Panel Fibrinolysis Diagnostic Interpretive Panel Von Willebrand Diagnostic Interpretive Panel Lupus Anticoagulant Diagnostic Interpretive Panel Prothrombin Time Elevation Diagnostic Panel PTT Elevation Diagnostic Panel Prothrombin Time and PTT Elevation Diagnostic Panel Normal Prothrombin Time and PTT Bleeding Panel Flow Cytometric Immunophenotyping (Low Grade Leukemia/Lymphoma markers) Flow Cytometric Immunophenotyping (Lymphoma, Tissue) Flow Cytometric Immunophenotyping (Individual Markers) Flow Cytometric Immunophenotyping (TdT) Flow Cytometric Immunophenotyping (Surface/Cytoplasmic IgM) Flow Cytometric Immunophenotyping (TCR V-beta) Page 6 of 6
Reflex Test Protocols
 UCH Clinical Laboratory Reflex Test Protocols Blood Bank Prepare RBCs for Transfusion (aka Crossmatch) N/A No Antibody Screen ordered Antibody Screen Direct Antiglobulin Test (DAT) Polyspecific DAT Positive
UCH Clinical Laboratory Reflex Test Protocols Blood Bank Prepare RBCs for Transfusion (aka Crossmatch) N/A No Antibody Screen ordered Antibody Screen Direct Antiglobulin Test (DAT) Polyspecific DAT Positive
Reflex Test Protocols
 Blood Bank Prepare RBCs for Transfusion (aka Crossmatch) N/A No Antibody Screen ordered Antibody Screen Direct Antiglobulin Test (DAT) Polyspecific DAT Positive polyspecific DAT IgG DAT, C3 DAT Fetal Cell
Blood Bank Prepare RBCs for Transfusion (aka Crossmatch) N/A No Antibody Screen ordered Antibody Screen Direct Antiglobulin Test (DAT) Polyspecific DAT Positive polyspecific DAT IgG DAT, C3 DAT Fetal Cell
COMPLIANCE. Individual Highlights: Advance Beneficiary Notices Medical Necessity IL Requisition Reflex Test List Panel Test. September 2018.
 September 2018 COMPLIANCE Dear Physician: Inova Laboratories (IL) is proud to serve the Northern Virginia community as the only full-service reference laboratory. Each year we disclose information about
September 2018 COMPLIANCE Dear Physician: Inova Laboratories (IL) is proud to serve the Northern Virginia community as the only full-service reference laboratory. Each year we disclose information about
Reflex Test Protocols
 University of Colorado Hospital Clinical Laboratory Reflex Test Protocols Blood Bank Prepare RBCs for Transfusion (aka Crossmatch) N/A No Antibody Screen ordered Antibody Screen Direct Antiglobulin Test
University of Colorado Hospital Clinical Laboratory Reflex Test Protocols Blood Bank Prepare RBCs for Transfusion (aka Crossmatch) N/A No Antibody Screen ordered Antibody Screen Direct Antiglobulin Test
Recommendations for Celiac Disease Testing. IgA & ttg IgA
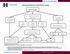 Recommendations for Celiac Disease Testing IgA & ttg IgA IgA Normal ttg IgA Negative IgA >10 mg/dl, but less than age matched range IgA
Recommendations for Celiac Disease Testing IgA & ttg IgA IgA Normal ttg IgA Negative IgA >10 mg/dl, but less than age matched range IgA
A written order to request the performance of a laboratory test can be initiated by the following parties:
 Requests/Reporting Requests for Laboratory Services A written order to request the performance of a laboratory test can be initiated by the following parties: Staff physicians and dentists who have been
Requests/Reporting Requests for Laboratory Services A written order to request the performance of a laboratory test can be initiated by the following parties: Staff physicians and dentists who have been
Clinical Laboratory. 14:41:00 Complement Component 3 50 mg/dl Oct-18
 Clinical Laboratory Procedure Result Units Ref Interval Accession Collected Received Thyroid Peroxidase (TPO) Antibody 5.0 IU/mL [0.0-9.0] 18-289-900139 16-Oct-18 Complement Component 3 50 mg/dl 18-289-900139
Clinical Laboratory Procedure Result Units Ref Interval Accession Collected Received Thyroid Peroxidase (TPO) Antibody 5.0 IU/mL [0.0-9.0] 18-289-900139 16-Oct-18 Complement Component 3 50 mg/dl 18-289-900139
Case Workshop of Society for Hematopathology and European Association for Haematopathology
 Case 148 2007 Workshop of Society for Hematopathology and European Association for Haematopathology Robert P Hasserjian Department of Pathology Massachusetts General Hospital Boston, MA Clinical history
Case 148 2007 Workshop of Society for Hematopathology and European Association for Haematopathology Robert P Hasserjian Department of Pathology Massachusetts General Hospital Boston, MA Clinical history
Clinical Laboratory. [None
 Clinical Laboratory Procedure Result Units Ref Interval Accession Collected Received Double-Stranded DNA (dsdna) Ab IgG ELISA Detected * [None 18-289-900151 Detected] Double-Stranded DNA (dsdna) Ab IgG
Clinical Laboratory Procedure Result Units Ref Interval Accession Collected Received Double-Stranded DNA (dsdna) Ab IgG ELISA Detected * [None 18-289-900151 Detected] Double-Stranded DNA (dsdna) Ab IgG
Specific Panels. Celiac disease panel. Pancreas Panel:
 Specific Panels Celiac disease panel Anti Endomysium IgA Anti Endomysium IgG Anti Gliadin IgA Anti Gliadin IgG Anti Transglutaminase IgA Anti Transglutaminase IgG Total IgA Total IgG Stool analysis +Sudan
Specific Panels Celiac disease panel Anti Endomysium IgA Anti Endomysium IgG Anti Gliadin IgA Anti Gliadin IgG Anti Transglutaminase IgA Anti Transglutaminase IgG Total IgA Total IgG Stool analysis +Sudan
1. Purpose 1.1. To define testing locations, schedule and order priority for each test performed in the core laboratory.
 Department Of Pathology GEN.1017.03 Integrated Test Schedule Version# 5 Department Specimen Processing POLICY NO. 1938 PAGE NO. 1 OF 6 Printed copies are for reference only. Please refer to the electronic
Department Of Pathology GEN.1017.03 Integrated Test Schedule Version# 5 Department Specimen Processing POLICY NO. 1938 PAGE NO. 1 OF 6 Printed copies are for reference only. Please refer to the electronic
Accredited entity according to ČSN EN ISO 15189:2013 Laboratoře Mikrochem a.s. Mikrochem Laboratories Nezvalova 984/2, Hodolany, Olomouc
 Medical laboratory locations: 1. Olomouc Nezvalova 984/2, 779 00 Olomouc 2. Přerov Čechova 8, 750 02 Přerov 3. Šumperk Nerudova 34, 787 01 Šumperk 4. Valašské Meziříčí Vsetínská 854, 757 01 Valašské Meziříčí
Medical laboratory locations: 1. Olomouc Nezvalova 984/2, 779 00 Olomouc 2. Přerov Čechova 8, 750 02 Přerov 3. Šumperk Nerudova 34, 787 01 Šumperk 4. Valašské Meziříčí Vsetínská 854, 757 01 Valašské Meziříčí
p. 111 p. 185 p. 197 p. 238
 Editorial Board Contributors Preface General Methods p. 1 General Methods: Antibody-Based and Molecular p. 3 Introduction p. 5 Antibody-Based Methods p. 6 p. xiii p. xv p. xxv Introduction to Molecular
Editorial Board Contributors Preface General Methods p. 1 General Methods: Antibody-Based and Molecular p. 3 Introduction p. 5 Antibody-Based Methods p. 6 p. xiii p. xv p. xxv Introduction to Molecular
Current September 2017
 1 Lab Service PRICE 87077 PATHOGEN ID 6 87088 PRESUMP ID, URINE CULTUR 6 87140 HSV CULTURE TYPING 13 87186 MIC (MIN INHIB CONC) 7 AMYLASE, SERUM 3 ANA SCREEN/RFX TITER AND DSDNA 4 ANA SCREEN/RFX TITER/COMPREHEN
1 Lab Service PRICE 87077 PATHOGEN ID 6 87088 PRESUMP ID, URINE CULTUR 6 87140 HSV CULTURE TYPING 13 87186 MIC (MIN INHIB CONC) 7 AMYLASE, SERUM 3 ANA SCREEN/RFX TITER AND DSDNA 4 ANA SCREEN/RFX TITER/COMPREHEN
Microbiology EQA Product Portfolio
 Labquality EQAS Microbiology EQA Product Portfolio Clinically relevant external quality assessment program for microbiology Bacterial serology Bacteriology Mycology Parasitology Preanalytics Virology Labquality
Labquality EQAS Microbiology EQA Product Portfolio Clinically relevant external quality assessment program for microbiology Bacterial serology Bacteriology Mycology Parasitology Preanalytics Virology Labquality
Test Name Results Units Bio. Ref. Interval
 135091662 Age 45 Years Gender Male 29/8/2017 120000AM 29/8/2017 100215AM 29/8/2017 110825AM Ref By Final RHEUMATOID AUTOIMMUNE COMREHENSIVE ANEL ANTI NUCLEAR ANTIBODY / FACTOR (ANA/ANF), SERUM ----- 20-60
135091662 Age 45 Years Gender Male 29/8/2017 120000AM 29/8/2017 100215AM 29/8/2017 110825AM Ref By Final RHEUMATOID AUTOIMMUNE COMREHENSIVE ANEL ANTI NUCLEAR ANTIBODY / FACTOR (ANA/ANF), SERUM ----- 20-60
July Monthly Update, Quest Diagnostics Nichols Institute, Valencia
 NEW TESTS Please Note: Not all test codes assigned to each assay are listed in the table of contents. Please refer to the complete listing on the page numbers indicated. Test Name Effective Date Page #
NEW TESTS Please Note: Not all test codes assigned to each assay are listed in the table of contents. Please refer to the complete listing on the page numbers indicated. Test Name Effective Date Page #
Overseas Referred Tests 2014
 OVERSEAS REFERRALS Listed below are some of the tests currently being offered by CML via Quest Diagnostics and Quest Nichols Institute The fees are quoted in US$ and a JA$ handling charge is also applicable.
OVERSEAS REFERRALS Listed below are some of the tests currently being offered by CML via Quest Diagnostics and Quest Nichols Institute The fees are quoted in US$ and a JA$ handling charge is also applicable.
H. PYLORI AB, IGG HPYL
 H. PYLORI AB, IGG HPYL Specimen Required: 3 ml blood, Serum gel tube Analytical Time: 1 day CPT Code: 86677 HAPTOGLOBIN HAP Specimen Required: 1.5 ml blood, gel tube Reference Range: 30-200 mg/dl CPT Code:
H. PYLORI AB, IGG HPYL Specimen Required: 3 ml blood, Serum gel tube Analytical Time: 1 day CPT Code: 86677 HAPTOGLOBIN HAP Specimen Required: 1.5 ml blood, gel tube Reference Range: 30-200 mg/dl CPT Code:
Test Name Results Units Bio. Ref. Interval
 LL - LL-ROHINI (NATIONAL REFERENCE 135091593 Age 25 Years Gender Male 30/8/2017 91600AM 30/8/2017 93946AM 31/8/2017 84826AM Ref By Final COLLAGEN DISEASES ANTIBODY ANEL ANTI NUCLEAR ANTIBODY / FACTOR (ANA/ANF),
LL - LL-ROHINI (NATIONAL REFERENCE 135091593 Age 25 Years Gender Male 30/8/2017 91600AM 30/8/2017 93946AM 31/8/2017 84826AM Ref By Final COLLAGEN DISEASES ANTIBODY ANEL ANTI NUCLEAR ANTIBODY / FACTOR (ANA/ANF),
TEST REQUEST INFORMATION- VIROLOGY
 Chlamydia/ Gonorrhea Nucleic Acid Amplification VC75 Qualitative Nucleic Acid Amplification SPECIMEN: Genital swab or first catch urine CONTAINER:GEN-PROBE APTIMA 2 Combo swab transport tube or urine transport
Chlamydia/ Gonorrhea Nucleic Acid Amplification VC75 Qualitative Nucleic Acid Amplification SPECIMEN: Genital swab or first catch urine CONTAINER:GEN-PROBE APTIMA 2 Combo swab transport tube or urine transport
Schedule of Accreditation issued by United Kingdom Accreditation Service 2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK
 2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK Luton and Dunstable University Hospital NHS Foundation Trust Lewsey Road Luton Bedfordshire LU4 0DZ Contact: Pauline Philip Tel: +44 (0)1582
2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK Luton and Dunstable University Hospital NHS Foundation Trust Lewsey Road Luton Bedfordshire LU4 0DZ Contact: Pauline Philip Tel: +44 (0)1582
Clinical Laboratory. 14:42:00 SSA-52 (Ro52) (ENA) Antibody, IgG 1 AU/mL [0-40] Oct-18
![Clinical Laboratory. 14:42:00 SSA-52 (Ro52) (ENA) Antibody, IgG 1 AU/mL [0-40] Oct-18 Clinical Laboratory. 14:42:00 SSA-52 (Ro52) (ENA) Antibody, IgG 1 AU/mL [0-40] Oct-18](/thumbs/95/125595183.jpg) Clinical Laboratory Procedure Result Units Ref Interval Accession Collected Received Rheumatoid Factor
Clinical Laboratory Procedure Result Units Ref Interval Accession Collected Received Rheumatoid Factor
Autoantibodies panel ANA
 Autoantibodies panel ANA Anti-nuclear antibodies, ANA screening General: Anti-nuclear antibodies (ANA) contain all kinds of autoantibodies against nuclear antigens. Their targets are cell components in
Autoantibodies panel ANA Anti-nuclear antibodies, ANA screening General: Anti-nuclear antibodies (ANA) contain all kinds of autoantibodies against nuclear antigens. Their targets are cell components in
Limitations - CEA Limitations Beta HCG
 Frequency Limitations often determine the coverage a patient receives for certain tests. The following information is obtained directly from the NCD/LCD policies for your convenience. Limitations : Glycated
Frequency Limitations often determine the coverage a patient receives for certain tests. The following information is obtained directly from the NCD/LCD policies for your convenience. Limitations : Glycated
AccuSet Autoimmune Performance Panel
 PACKAGE INSERT 0840-0001 INTENDED USE The 0840-0001 is intended for use by diagnostic manufacturers, researchers, and clinical laboratories to develop, evaluate, or troubleshoot autoimmune related test
PACKAGE INSERT 0840-0001 INTENDED USE The 0840-0001 is intended for use by diagnostic manufacturers, researchers, and clinical laboratories to develop, evaluate, or troubleshoot autoimmune related test
Controls & Calibrators. Disease Quality Controls
 Controls & Calibrators Infectious Disease Quality Controls Infectious Disease Quality Controls A broad selection of controls designed to monitor assay precision of hepatitis, retrovirus, sexually transmitted
Controls & Calibrators Infectious Disease Quality Controls Infectious Disease Quality Controls A broad selection of controls designed to monitor assay precision of hepatitis, retrovirus, sexually transmitted
BID ANNUAL CONTRACT FOR LAB TESTING
 1 ACID FAST BACILLISTAIN 100 10.48 1,048.00 8.00 800.00 2 AFP, TUMOR (CHIRON) 10 9.04 90.40 7.00 70.00 3 AMYLASE 160 3.13 500.80 2.75 440.00 4 BASIC METAB PNL 1,800 1.44 2,592.00 1.95 3,510.00 5 BZV IGGAB
1 ACID FAST BACILLISTAIN 100 10.48 1,048.00 8.00 800.00 2 AFP, TUMOR (CHIRON) 10 9.04 90.40 7.00 70.00 3 AMYLASE 160 3.13 500.80 2.75 440.00 4 BASIC METAB PNL 1,800 1.44 2,592.00 1.95 3,510.00 5 BZV IGGAB
Medical Laboratory Accreditation Programme
 Client Client Number 121 Address PO Box 6064, Dunedin North, Dunedin, 9059 Plunket House, 472 George Street, North Dunedin, Dunedin, 9016 Telephone 03 477-6981 Fax 03 477-9160 Authorised Representative
Client Client Number 121 Address PO Box 6064, Dunedin North, Dunedin, 9059 Plunket House, 472 George Street, North Dunedin, Dunedin, 9016 Telephone 03 477-6981 Fax 03 477-9160 Authorised Representative
Laboratory diagnosis of congenital infections
 Laboratory diagnosis of congenital infections Laboratory diagnosis of HSV Direct staining Tzanck test Immunostaining HSV isolation Serology PCR Tzanck test Cell scrape from base of the lesion smear on
Laboratory diagnosis of congenital infections Laboratory diagnosis of HSV Direct staining Tzanck test Immunostaining HSV isolation Serology PCR Tzanck test Cell scrape from base of the lesion smear on
BLOOD. Dr. Vedat Evren
 BLOOD Dr. Vedat Evren Blood Liquid suspension of formed elements Blood = Blood cells + plasma Plasma = Coagulation factors + serum Cells = Erythrocytes + Leukocytes + Thrombocytes 8 % of the total body
BLOOD Dr. Vedat Evren Blood Liquid suspension of formed elements Blood = Blood cells + plasma Plasma = Coagulation factors + serum Cells = Erythrocytes + Leukocytes + Thrombocytes 8 % of the total body
Is it Autoimmune or NOT! Presented to AONP! October 2015!
 Is it Autoimmune or NOT! Presented to AONP! October 2015! Four main jobs of immune system Detects Contains and eliminates Self regulates Protects Innate Immune System! Epithelial cells, phagocytic cells
Is it Autoimmune or NOT! Presented to AONP! October 2015! Four main jobs of immune system Detects Contains and eliminates Self regulates Protects Innate Immune System! Epithelial cells, phagocytic cells
Hematology Revision. By Dr.AboRashad . Mob
 1 1- Hb A2 is consisting of: a) 3 ά chains and 2 γ chains b) 2 ά chains and 2 β chains c) 2 ά chains and 2 δ chains** d) 2 ά chains and 3 δ chains e) 3 ά chains and 2 δ chains 2- The main (most) Hb found
1 1- Hb A2 is consisting of: a) 3 ά chains and 2 γ chains b) 2 ά chains and 2 β chains c) 2 ά chains and 2 δ chains** d) 2 ά chains and 3 δ chains e) 3 ά chains and 2 δ chains 2- The main (most) Hb found
In Office Lab Testing
 Effective January 1, 201, the lab services below can be performed and reimbursed in an office setting. All other office-based lab services must be submitted through our contracted laboratory providers.
Effective January 1, 201, the lab services below can be performed and reimbursed in an office setting. All other office-based lab services must be submitted through our contracted laboratory providers.
Transfusion Reactions. Directed by M-azad March 2012
 Transfusion Reactions Directed by M-azad March 2012 Transfusion Reactions are Adverse reactions associated with the transfusion of blood and its components Transfusion reactions Non-threatening to fatal
Transfusion Reactions Directed by M-azad March 2012 Transfusion Reactions are Adverse reactions associated with the transfusion of blood and its components Transfusion reactions Non-threatening to fatal
PILOT STUDY OF ANTIGEN MATCHING FOR AUTOIMMUNE HEMOLYTIC ANEMIA
 PILOT STUDY OF ANTIGEN MATCHING FOR AUTOIMMUNE HEMOLYTIC ANEMIA Sharon Rice & Fred Plapp Saint Luke s Hospital Kansas City, MO CENTRALIZED TRANSFUSION SERVICE Antibody identification Antibody titer Antigen
PILOT STUDY OF ANTIGEN MATCHING FOR AUTOIMMUNE HEMOLYTIC ANEMIA Sharon Rice & Fred Plapp Saint Luke s Hospital Kansas City, MO CENTRALIZED TRANSFUSION SERVICE Antibody identification Antibody titer Antigen
In-OfficeLabTesting. Effective date: August 1, 2017
 Effective date: August 1, 2017 In-OfficeLabTesting The lab services below can be performed and reimbursed in an office setting. All other office-based lab services must be submitted through our contracted
Effective date: August 1, 2017 In-OfficeLabTesting The lab services below can be performed and reimbursed in an office setting. All other office-based lab services must be submitted through our contracted
Chapter 17 Worksheet Code It
 Class: Date: Chapter 17 Worksheet 3 2 1 Code It True/False Indicate whether the statement is true or false. 1. CPT laboratory codes include collection of the specimen. 2. A dipstick is a small piece of
Class: Date: Chapter 17 Worksheet 3 2 1 Code It True/False Indicate whether the statement is true or false. 1. CPT laboratory codes include collection of the specimen. 2. A dipstick is a small piece of
SPECTRA EAST, INC. Rockleigh, NJ
 A2LA has accredited SPECTRA EAST, INC. Rockleigh, NJ for technical competence in the field of Medical Testing This laboratory is accredited in accordance with the recognized International Standard ISO/IEC
A2LA has accredited SPECTRA EAST, INC. Rockleigh, NJ for technical competence in the field of Medical Testing This laboratory is accredited in accordance with the recognized International Standard ISO/IEC
Technical Article Series Scleroderma ANA and Antibody Testing
 Technical Article Series Scleroderma ANA and Antibody Testing ANA Testing Basics The long-standing way of doing ANA testing is a method called indirect immunofluorescence (IFA). It is a time consuming,
Technical Article Series Scleroderma ANA and Antibody Testing ANA Testing Basics The long-standing way of doing ANA testing is a method called indirect immunofluorescence (IFA). It is a time consuming,
Schedule of Accreditation issued by United Kingdom Accreditation Service 2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK
 2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK Department of Microbiology Princess Alexandra Hospital Hamstel Road Harlow CM20 1QX Contact: D Orriss Tel: +44 01279 827138 E-Mail: Debbie.Orriss@pah.nhs.uk
2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK Department of Microbiology Princess Alexandra Hospital Hamstel Road Harlow CM20 1QX Contact: D Orriss Tel: +44 01279 827138 E-Mail: Debbie.Orriss@pah.nhs.uk
Chapter 3 Diseases of the Blood and Bloodforming Organs and Certain Disorders Involving the Immune Mechanism D50-D89
 Chapter 3 Diseases of the Blood and Bloodforming Organs and Certain Disorders Involving the Immune Mechanism D50-D89 Presented by Jennifer Kurkulonis 1 FOUR MAJOR TYPES OF BLOOD CELLS White blood cells
Chapter 3 Diseases of the Blood and Bloodforming Organs and Certain Disorders Involving the Immune Mechanism D50-D89 Presented by Jennifer Kurkulonis 1 FOUR MAJOR TYPES OF BLOOD CELLS White blood cells
Laboratory Values, Critical. ThedaCare. Date Last Reviewed: 4/5/2018 Reviewing Body(s): Quality; Laboratory Leadership
 Laboratory Values, Critical ThedaCare Policy & Procedure Policy Title: Laboratory Values, Critical Policy Number: 694 Location(s): All ThedaCare Department(s): Laboratory Date Last Reviewed: 4/5/2018 Reviewing
Laboratory Values, Critical ThedaCare Policy & Procedure Policy Title: Laboratory Values, Critical Policy Number: 694 Location(s): All ThedaCare Department(s): Laboratory Date Last Reviewed: 4/5/2018 Reviewing
DEPARTMENT OF MICROBIOLOGY IMPORTANT NOTICE TO USERS Turnaround Times (TATs) for Microbiology Investigations
 Dear User, ISSUE: M008 DEPARTMENT OF MICROBIOLOGY IMPORTANT NOTICE TO USERS Turnaround Times (TATs) for Microbiology Investigations In order to comply with national quality guidance and as part of our
Dear User, ISSUE: M008 DEPARTMENT OF MICROBIOLOGY IMPORTANT NOTICE TO USERS Turnaround Times (TATs) for Microbiology Investigations In order to comply with national quality guidance and as part of our
ISO 15189:2012 Internationally-Recognized Accredited Laboratory. SPECTRA EAST, INC. Rockleigh, NJ
 ISO 15189:2012 Internationally-Recognized Accredited Laboratory A2LA has accredited SPECTRA EAST, INC. Rockleigh, NJ for technical competence in the field of Clinical Testing This laboratory is accredited
ISO 15189:2012 Internationally-Recognized Accredited Laboratory A2LA has accredited SPECTRA EAST, INC. Rockleigh, NJ for technical competence in the field of Clinical Testing This laboratory is accredited
Result Electrophoresis Tests
 Result Electrophoresis Tests Result a Serum Protein Electrophoresis Test... 2 Result a Urine Protein Electrophoresis Test... 7 Result a Hemoglobin Electrophoresis Test... 8 Request and Result an IFE test...
Result Electrophoresis Tests Result a Serum Protein Electrophoresis Test... 2 Result a Urine Protein Electrophoresis Test... 7 Result a Hemoglobin Electrophoresis Test... 8 Request and Result an IFE test...
Inspector's Accreditation Unit Activity Menu
 01/12/20XX 15:58:57 Laboratory Accreditation Program Page 1 of 9 CHEMISTRY 1501 ALT, serum/plasma 1502 Albumin, serum/plasma 1504 Alkaline phosphatase, serum/plasma 1506 Amylase, serum/plasma 1508 Bilirubin,
01/12/20XX 15:58:57 Laboratory Accreditation Program Page 1 of 9 CHEMISTRY 1501 ALT, serum/plasma 1502 Albumin, serum/plasma 1504 Alkaline phosphatase, serum/plasma 1506 Amylase, serum/plasma 1508 Bilirubin,
PERIPHERAL NERVE WORKUP FORM (PNW)
 PERIPHERAL NERVE WORKUP FORM (PNW) The Peripheral Nerve Workup Form (PNW) has to be filled out at the time a new patient is enrolled into the PNRR study. Only testing conducted within 36 months of visit
PERIPHERAL NERVE WORKUP FORM (PNW) The Peripheral Nerve Workup Form (PNW) has to be filled out at the time a new patient is enrolled into the PNRR study. Only testing conducted within 36 months of visit
PROFILES MENU. Page no. 9 Page no. 8 Page no. 7 Page no. 6 Page no. 5. Page no. 2. Page no. 4 Page no. 3
 Health Profiles PROFILES MENU Page no. 1 Page no. 2 Page no. 4 Page no. 3 1 Basic Metabolic Panel (BMP) 2 Liver Function Tests (LFT) 3 Comprehensive Metabolic Panel (CMP) 4 Lipid Profile 5 Chem 7 Panel
Health Profiles PROFILES MENU Page no. 1 Page no. 2 Page no. 4 Page no. 3 1 Basic Metabolic Panel (BMP) 2 Liver Function Tests (LFT) 3 Comprehensive Metabolic Panel (CMP) 4 Lipid Profile 5 Chem 7 Panel
M.D.IPA, M.D.IPA Preferred, Optimum Choice and Optimum Choice Preferred STAT Laboratory List Revised Jan. 5, 2017
 M.D.IPA, M.D.IPA Preferred, Optimum Choice and Optimum Choice Preferred STAT Laboratory List Revised Jan. 5, 2017 If laboratory results are required on a STAT basis, the designated commercial medical laboratory
M.D.IPA, M.D.IPA Preferred, Optimum Choice and Optimum Choice Preferred STAT Laboratory List Revised Jan. 5, 2017 If laboratory results are required on a STAT basis, the designated commercial medical laboratory
Epic Labs Orderable As STAT PRIORITY As of 06/22/2016
 ABG+HB(CORDARTERIAL) - BABY A ABG+HB(CORD ARTERIAL)- BABY B ABG+HB(CORD ARTERIAL)- BABY C ACETAMINOPHEN LEVEL ALANINE AMINOTRANSFERASE (ALT) ALBUMIN, FLUID ALBUMIN, PLEURAL FLUID ALBUMIN, SYNOVIAL FLUID
ABG+HB(CORDARTERIAL) - BABY A ABG+HB(CORD ARTERIAL)- BABY B ABG+HB(CORD ARTERIAL)- BABY C ACETAMINOPHEN LEVEL ALANINE AMINOTRANSFERASE (ALT) ALBUMIN, FLUID ALBUMIN, PLEURAL FLUID ALBUMIN, SYNOVIAL FLUID
Viral Hepatitis Diagnosis and Management
 Viral Hepatitis Diagnosis and Management CLINICAL BACKGROUND Viral hepatitis is a relatively common disease (25 per 100,000 individuals in the United States) caused by a diverse group of hepatotropic agents
Viral Hepatitis Diagnosis and Management CLINICAL BACKGROUND Viral hepatitis is a relatively common disease (25 per 100,000 individuals in the United States) caused by a diverse group of hepatotropic agents
Susanne Norris Zanto, MPH, MLS (ASCP) CM, SM Montana Public Health Laboratory
 Susanne Norris Zanto, MPH, MLS (ASCP) CM, SM Montana Public Health Laboratory Describe the challenges in syphilis diagnostics Present two testing algorithms Non-treponemal test as initial screen Treponemal
Susanne Norris Zanto, MPH, MLS (ASCP) CM, SM Montana Public Health Laboratory Describe the challenges in syphilis diagnostics Present two testing algorithms Non-treponemal test as initial screen Treponemal
Disclosures. Rheumatological Approaches to Differential Diagnosis, Physical Examination, and Interpretation of Studies. None
 Rheumatological Approaches to Differential Diagnosis, Physical Examination, and Interpretation of Studies Sarah Goglin MD Assistant Professor of Medicine Division of Rheumatology Disclosures None 1 [footer
Rheumatological Approaches to Differential Diagnosis, Physical Examination, and Interpretation of Studies Sarah Goglin MD Assistant Professor of Medicine Division of Rheumatology Disclosures None 1 [footer
DCO Registration. If you have any questions contact the DCO help desk at:
 DCO Registration Please register using the two simple steps below: 1. Log-in or create a CME account: https://tiny.army.mil/r/zb8a/cme **Tip: If your facility is not listed as an option on the registration
DCO Registration Please register using the two simple steps below: 1. Log-in or create a CME account: https://tiny.army.mil/r/zb8a/cme **Tip: If your facility is not listed as an option on the registration
Form 2033 R3.0: Wiskott-Aldrich Syndrome Pre-HSCT Data
 Key Fields Sequence Number: Date Received: - - CIBMTR Center Number: CIBMTR Recipient ID: Has this patient's data been previously reported to USIDNET? USIDNET ID: Today's Date: - - Date of HSCT for which
Key Fields Sequence Number: Date Received: - - CIBMTR Center Number: CIBMTR Recipient ID: Has this patient's data been previously reported to USIDNET? USIDNET ID: Today's Date: - - Date of HSCT for which
Action Item for 2019 Review of Tool. Maintain (add include oral cavity) Maintain. Archive. Archive. 12 creatinine)
 NEWLY DIAGNOSED/ NEW TO CARE PROGRAM SITE: REVIEWER(S): REVIEW DATE: CORE SERVICES Outpatient/Ambulatory Health Services Tool - 2018 (OLD) SECTION 1: CHART REVIEW Review for newly diagnosed HIV patients
NEWLY DIAGNOSED/ NEW TO CARE PROGRAM SITE: REVIEWER(S): REVIEW DATE: CORE SERVICES Outpatient/Ambulatory Health Services Tool - 2018 (OLD) SECTION 1: CHART REVIEW Review for newly diagnosed HIV patients
Laboratory Testing for the Primary Care Optometrist
 General lab principles Indications for testing - Confirm a clinical diagnosis - Rule out a diagnosis - Manage a disease - Screen for a disease Sensitivity is the ability to show a positive result in a
General lab principles Indications for testing - Confirm a clinical diagnosis - Rule out a diagnosis - Manage a disease - Screen for a disease Sensitivity is the ability to show a positive result in a
VASCULITIS PRODUCT HIGHLIGHTS
 VASCULITIS PRODUCT HIGHLIGHTS AESKU.DIAGNOSTICS offers a comprehensive and complete diagnostic portfolio in the field of vasculitis diagnostics. Not only are screening and profiling s available but also
VASCULITIS PRODUCT HIGHLIGHTS AESKU.DIAGNOSTICS offers a comprehensive and complete diagnostic portfolio in the field of vasculitis diagnostics. Not only are screening and profiling s available but also
Test Name Results Units Bio. Ref. Interval
 135091660 Age 44 Years Gender Male 29/8/2017 120000AM 29/8/2017 100219AM 29/8/2017 105510AM Ref By Final EXTRACTABLENUCLEAR ANTIGENS (ENA), QUANTITATIVE ROFILE CENTROMERE ANTIBODY, SERUM 20-30 Weak ositive
135091660 Age 44 Years Gender Male 29/8/2017 120000AM 29/8/2017 100219AM 29/8/2017 105510AM Ref By Final EXTRACTABLENUCLEAR ANTIGENS (ENA), QUANTITATIVE ROFILE CENTROMERE ANTIBODY, SERUM 20-30 Weak ositive
Assays. New. New. Combinations. Possibilities. Patents: EP , AU
 Assays Patents: EP 2362222, AU 2011217190 New Combinations New Possibilities Technology Classical Handling of Autoimmune Diagnostics 2-Step Diagnostics 1 st Screening 2 nd Confirmation Cell based IFA ELISA
Assays Patents: EP 2362222, AU 2011217190 New Combinations New Possibilities Technology Classical Handling of Autoimmune Diagnostics 2-Step Diagnostics 1 st Screening 2 nd Confirmation Cell based IFA ELISA
Chain of Infection Agent Mode of transmission Contact (direct, indirect, droplet spread) Airborne Common-vehicle spread Host
 Goals Microbiology of Healthcare-associated Infections William A. Rutala, Ph.D., M.P.H. Director, Statewide Program for Infection Control and Epidemiology and Research Professor of Medicine, University
Goals Microbiology of Healthcare-associated Infections William A. Rutala, Ph.D., M.P.H. Director, Statewide Program for Infection Control and Epidemiology and Research Professor of Medicine, University
ALPS Screen Order Set B-Cell Panel Order Set (% and absolute #) (% and Absolute #) ALPS (CD3+/CD4-/CD8-/TCR αβ+)
 All test indicated in a Panel or are available individually. Version 1/1/2018 ALPS Screen B-Cell Panel (% and absolute #) (% and Absolute #) ALPS (/CD4-/CD8-/TCR αβ+) CD5+/ ANA Profile * Anti-Nuclear Ab*
All test indicated in a Panel or are available individually. Version 1/1/2018 ALPS Screen B-Cell Panel (% and absolute #) (% and Absolute #) ALPS (/CD4-/CD8-/TCR αβ+) CD5+/ ANA Profile * Anti-Nuclear Ab*
Budsakorn Darawankul, MD. Maharat Nakhon Ratchasima Hospital
 Budsakorn Darawankul, MD. Maharat Nakhon Ratchasima Hospital Outline What is ANA? How to detect ANA? Clinical application Common autoantibody in ANA diseases Outline What is ANA? How to detect ANA? Clinical
Budsakorn Darawankul, MD. Maharat Nakhon Ratchasima Hospital Outline What is ANA? How to detect ANA? Clinical application Common autoantibody in ANA diseases Outline What is ANA? How to detect ANA? Clinical
PENN STATE MILTON S. HERSHEY MEDICAL CENTER TABLE OF CRITICAL LAB VALUES CURRENT AS OF: 1/2015
 PENN STATE MILTON S. HERSHEY MEDICAL CENTER TABLE OF CRITICAL LAB VALUES CURRENT AS OF: 1/2015 Section: Chemistry For inpatients, once 3 consecutive critical values for the same analyte (test result) have
PENN STATE MILTON S. HERSHEY MEDICAL CENTER TABLE OF CRITICAL LAB VALUES CURRENT AS OF: 1/2015 Section: Chemistry For inpatients, once 3 consecutive critical values for the same analyte (test result) have
GUIDELINES FOR THE TRANSFUSION OF BLOOD COMPONENTS
 CHILDREN S HOSPITALS AND CLINICS OF MINNESOTA Introduction: GUIDELINES FOR THE TRANSFUSION OF BLOOD COMPONENTS These guidelines have been developed in conjunction with the hospital Transfusion Committee.
CHILDREN S HOSPITALS AND CLINICS OF MINNESOTA Introduction: GUIDELINES FOR THE TRANSFUSION OF BLOOD COMPONENTS These guidelines have been developed in conjunction with the hospital Transfusion Committee.
Overview of changing happening to laboratory testing at Texas Department of State Health Services (DSHS), effective September 1, 2013
 Overview of changing happening to laboratory testing at Texas Department of State Health Services (DSHS), effective September 1, 2013 Testing Effective September 1, 2013 the tests listed below are no longer
Overview of changing happening to laboratory testing at Texas Department of State Health Services (DSHS), effective September 1, 2013 Testing Effective September 1, 2013 the tests listed below are no longer
Case #1. Robert J. Glinert, M.D. David C. Fisher, M.D. Dana Farber Cancer Institute
 Case #1 Robert J. Glinert, M.D. David C. Fisher, M.D. Dana Farber Cancer Institute Patient History Part I 76 year-old man 1997 diagnosed with MGUS (biclonal) during evaluation of (self-limited) anemia.
Case #1 Robert J. Glinert, M.D. David C. Fisher, M.D. Dana Farber Cancer Institute Patient History Part I 76 year-old man 1997 diagnosed with MGUS (biclonal) during evaluation of (self-limited) anemia.
Medicaid Family Planning Waiver Services CPT Codes and ICD-10 Diagnosis Codes
 CPT Code Description of Covered Codes Evaluation and Management 99384FP 99385FP Family planning new visit 99386FP 99394FP 99395FP Family planning established visit 99396FP 99401FP HIV counseling (pre-test)
CPT Code Description of Covered Codes Evaluation and Management 99384FP 99385FP Family planning new visit 99386FP 99394FP 99395FP Family planning established visit 99396FP 99401FP HIV counseling (pre-test)
TEST NAME/ PSYCHE CODE COLLECTION GUIDELINES REFERENCE VALUES ROOM TEMP. REFRIGERATED FROZEN
 TEST NAME/ PSYCHE CODE COLLECTION GUIDELINES REFERENCE VALUES ROOM TEMP. REFRIGERATED FROZEN Clinical Utility CA19-9 (CA-GI) 8 hours 48 hours UNDETERMINED CA 19-9 may be ordered along with other tests,
TEST NAME/ PSYCHE CODE COLLECTION GUIDELINES REFERENCE VALUES ROOM TEMP. REFRIGERATED FROZEN Clinical Utility CA19-9 (CA-GI) 8 hours 48 hours UNDETERMINED CA 19-9 may be ordered along with other tests,
MLAB 1235 Immunology/Serology Course Objectives and Course Ooutlinee Fall 2003
 MLAB 1235 Immunology/Serology Course Objectives and Course Ooutlinee Fall 2003 1. Nature of the Immune System I. Historical Concepts 1. Define immunology and immunity. 2. Describe the process and purpose
MLAB 1235 Immunology/Serology Course Objectives and Course Ooutlinee Fall 2003 1. Nature of the Immune System I. Historical Concepts 1. Define immunology and immunity. 2. Describe the process and purpose
LPS CD40 + IL-4. Vorinostat (24 Hours) Vorinostat (24 Hours) Panobinostat (24 Hours) Panobinostat (24 Hours) Romidepsin (48 Hours)
 A) CD + IL- B) LPS ( Hours) ( Hours) Cell number (x1-3 ) 1 1 3.7 M 1. M. M.1 M Cell number (x1 - ) 1 1 3. M 1. M.7 M.38 M Cell number (x1-3 ) Cell number (x1-3 ) 3 1 1 1 ( Hours) 7.nM.nM 1.7nM.nM Romidepsin
A) CD + IL- B) LPS ( Hours) ( Hours) Cell number (x1-3 ) 1 1 3.7 M 1. M. M.1 M Cell number (x1 - ) 1 1 3. M 1. M.7 M.38 M Cell number (x1-3 ) Cell number (x1-3 ) 3 1 1 1 ( Hours) 7.nM.nM 1.7nM.nM Romidepsin
Test Result Reference Range Flag
 Date of Last Result Test Result Reference Range Flag Dec 07, 2016 25-Hydroxy Vitamin D Total 53 ng/ml 30-100 ng/ml Activated Partial Thromboplast Time Alanine Aminotransferase (ALT/SGPT) 25 sec 24-35 sec
Date of Last Result Test Result Reference Range Flag Dec 07, 2016 25-Hydroxy Vitamin D Total 53 ng/ml 30-100 ng/ml Activated Partial Thromboplast Time Alanine Aminotransferase (ALT/SGPT) 25 sec 24-35 sec
If a critical result is obtained for a clinic patient after the clinic has closed, we will notify the individual on-call for the clinic.
 UCSF Clinical Laboratories Critical ('Panic') and Subcritical Values Policy (Last updated 11/3/17): In collaboration with the clinical services, the following results have been determined to represent
UCSF Clinical Laboratories Critical ('Panic') and Subcritical Values Policy (Last updated 11/3/17): In collaboration with the clinical services, the following results have been determined to represent
Supplementary Online Content
 Supplementary Online Content Peters PJ, Westheimer E, Cohen S, et al. Screening yield of HIV antigen/antibody combination and pooled HIV RNA testing for acute HIV infection in a high-prevalence population.
Supplementary Online Content Peters PJ, Westheimer E, Cohen S, et al. Screening yield of HIV antigen/antibody combination and pooled HIV RNA testing for acute HIV infection in a high-prevalence population.
Collection (Specimen Source Required on all tests) Sputum: >5 ml required. First morning specimen preferred.
 Type Acid Fast (Mycobacteria) Sputum: >5 ml required. First morning specimen preferred. For blood, sodium heparin tube preferred. Lithium heparin acceptable. Do not centrifuge.. delay. Swabs are not appropriate
Type Acid Fast (Mycobacteria) Sputum: >5 ml required. First morning specimen preferred. For blood, sodium heparin tube preferred. Lithium heparin acceptable. Do not centrifuge.. delay. Swabs are not appropriate
Brrrr, It s Cold In Here
 Brrrr, It s Cold In Here Kate Grogan, MD Transfusion Medicine Fellow Physician, BloodworksNW 4/24/15 Patient 20 year old female Viral illness Presents one week later with severe hemolysis Hgb 5.6 Increased
Brrrr, It s Cold In Here Kate Grogan, MD Transfusion Medicine Fellow Physician, BloodworksNW 4/24/15 Patient 20 year old female Viral illness Presents one week later with severe hemolysis Hgb 5.6 Increased
2014 Notice to Physicians
 2014 Notice to Physicians August 20, 2014 Dear Physician, will only pay for tests that are deemed medically necessary. These regulations impact physicians who order the tests as well as the laboratories
2014 Notice to Physicians August 20, 2014 Dear Physician, will only pay for tests that are deemed medically necessary. These regulations impact physicians who order the tests as well as the laboratories
University Medical Center at Brackenridge. Gastroenterology Clinic Worksheet
 Gastroenterology Clinic Worksheet 1. GI Bleeding (occult or symptomatic) a. CBC b. Iron, Ferritin b. Medication history 2. Iron Deficiency Anemia and no evident source (if no iron deficiency consider hematological
Gastroenterology Clinic Worksheet 1. GI Bleeding (occult or symptomatic) a. CBC b. Iron, Ferritin b. Medication history 2. Iron Deficiency Anemia and no evident source (if no iron deficiency consider hematological
Basic Metabolic Panel
 Basic Metabolic Panel Order Name: CHEM 8 Test Number: 2028100 REV DATE:2/5/2008 Glucose Urea Nitrogen, Blood (BUN) Creatinine Sodium Potassium Serum/Plasma Chloride Bicarbonate Calcium Anion Gap Calculated
Basic Metabolic Panel Order Name: CHEM 8 Test Number: 2028100 REV DATE:2/5/2008 Glucose Urea Nitrogen, Blood (BUN) Creatinine Sodium Potassium Serum/Plasma Chloride Bicarbonate Calcium Anion Gap Calculated
Let`s go for the diagnosis! Yazeed Toukan, MD Pediatric Pulmonary Institute, Ruth Rappaport Children`s Hospital July 2016
 Let`s go for the diagnosis! Yazeed Toukan, MD Pediatric Pulmonary Institute, Ruth Rappaport Children`s Hospital July 2016 Case report 20 months old girl Israeli Arab Muslim family, consanguineous marriage
Let`s go for the diagnosis! Yazeed Toukan, MD Pediatric Pulmonary Institute, Ruth Rappaport Children`s Hospital July 2016 Case report 20 months old girl Israeli Arab Muslim family, consanguineous marriage
Medical Immunology Practice Questions-2016 Autoimmunity + Case Studies
 Medical Immunology Practice Questions-2016 Autoimmunity + Case Studies Directions: Each of the numbered items or incomplete statements in this section is followed by answers or by completions of the statement.
Medical Immunology Practice Questions-2016 Autoimmunity + Case Studies Directions: Each of the numbered items or incomplete statements in this section is followed by answers or by completions of the statement.
Confirmatory Testing Algorithm. Routine Donor Testing Algorithm
 Confirmatory Testing Algorithm Blood Confirmatory ABO/Rh Beckman Coulter PK-ABO/Rh Immucor NEO ABO/Rh Immucor NEO ABO/Rh Immucor Manual ABO/Rh Typing Immucor Manual ABO/Rh Typing Discrepancy Resolution
Confirmatory Testing Algorithm Blood Confirmatory ABO/Rh Beckman Coulter PK-ABO/Rh Immucor NEO ABO/Rh Immucor NEO ABO/Rh Immucor Manual ABO/Rh Typing Immucor Manual ABO/Rh Typing Discrepancy Resolution
LIST OF INVESTIGATIONS
 Karyotyping: K001 K002 LIST OF INVESTIGATIONS SAMPLE CONTAINER TYPE cells For Karyotyping [Single] cells For Karyotyping [Couple] Vacutainer Vacutainer 7-8 7-8 K003 Fetal Blood Sample For Karyotyping Vacutainer
Karyotyping: K001 K002 LIST OF INVESTIGATIONS SAMPLE CONTAINER TYPE cells For Karyotyping [Single] cells For Karyotyping [Couple] Vacutainer Vacutainer 7-8 7-8 K003 Fetal Blood Sample For Karyotyping Vacutainer
Unit II Problem 2 Microbiology Lab: Pneumonia
 Unit II Problem 2 Microbiology Lab: Pneumonia - What are the steps needed to obtain a proper sputum specimen? You need the following: A wide-mouth labeled container. Gloves. Water. Mouth wash + tissues.
Unit II Problem 2 Microbiology Lab: Pneumonia - What are the steps needed to obtain a proper sputum specimen? You need the following: A wide-mouth labeled container. Gloves. Water. Mouth wash + tissues.
All abnormal results for this test will automatically have the DNA test reflexed at an additional cost.
 2211 Michigan Avenue Santa Monica, CA 90404 800 421 4449 Dear Colleague: May-June 2004 REVISED 6-15-04 At Specialty we continue to focus on working with you to reduce the problems that can result in tests
2211 Michigan Avenue Santa Monica, CA 90404 800 421 4449 Dear Colleague: May-June 2004 REVISED 6-15-04 At Specialty we continue to focus on working with you to reduce the problems that can result in tests
HIV Diagnostic Testing
 In The name of God HIV Diagnostic Testing By : Dr. Shahzamani PhD of Medical virology Purpose of HIV Testing To identify asymptomatic individuals To diagnose HIV infection in those who practice high risk
In The name of God HIV Diagnostic Testing By : Dr. Shahzamani PhD of Medical virology Purpose of HIV Testing To identify asymptomatic individuals To diagnose HIV infection in those who practice high risk
CRITICAL VALUES, SIGNIFICANT FINDINGS LIST and STAT RESULTS
 Page 1 of 6 Affected Sites: X Enterprise Chandler Good Samaritan CRITICAL VALUES: s are defined as an analytic result that suggests a clinical condition that may be lifethreatening and may require immediate
Page 1 of 6 Affected Sites: X Enterprise Chandler Good Samaritan CRITICAL VALUES: s are defined as an analytic result that suggests a clinical condition that may be lifethreatening and may require immediate
ISTITUTO DI RICERCHE FARMACOLOGICHE MARIO NEGRI CLINICAL RESEARCH CENTER ALDO E FOR CELE RARE DACCO DISEASES ALDO E CELE DACCO
 ISTITUTO DI RICERCHE FARMACOLOGICHE MARIO NEGRI CENTRO MARIO DI NEGRI RICERCHE INSTITUTE CLINICHE FOR PHARMACOLOGICAL PER LE MALATTIE RESEARCH RARE CLINICAL RESEARCH CENTER ALDO E FOR CELE RARE DACCO DISEASES
ISTITUTO DI RICERCHE FARMACOLOGICHE MARIO NEGRI CENTRO MARIO DI NEGRI RICERCHE INSTITUTE CLINICHE FOR PHARMACOLOGICAL PER LE MALATTIE RESEARCH RARE CLINICAL RESEARCH CENTER ALDO E FOR CELE RARE DACCO DISEASES
Schedule of Accreditation issued by United Kingdom Accreditation Service 2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK
 2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK Clinical Microbiology QMC Campus Derby Road Nottingham NG7 2UH Contact: Jil Bowskill Tel: +44 (0) 1905 760192 Ext 30659 E-Mail: jilean.bowskill@nuh.nhs.uk
2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK Clinical Microbiology QMC Campus Derby Road Nottingham NG7 2UH Contact: Jil Bowskill Tel: +44 (0) 1905 760192 Ext 30659 E-Mail: jilean.bowskill@nuh.nhs.uk
Mandatory Reflex Testing Approved by Medical Executive Committee (MEC) October 2018
 Mandatory Reflex Testing Approved by Medical Executive Committee (MEC) October 2018 INITIAL TEST and RESULT All newly diagnosed patients with Acute Myeloid Leukemia under age 80, unless otherwise specified
Mandatory Reflex Testing Approved by Medical Executive Committee (MEC) October 2018 INITIAL TEST and RESULT All newly diagnosed patients with Acute Myeloid Leukemia under age 80, unless otherwise specified
Schedule of Accreditation issued by United Kingdom Accreditation Service 2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK
 2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK Royal Liverpool & Broadgreen University Hospitals NHS Trust Department of Virology Liverpool Clinical Laboratories Royal Liverpool and Broadgreen
2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK Royal Liverpool & Broadgreen University Hospitals NHS Trust Department of Virology Liverpool Clinical Laboratories Royal Liverpool and Broadgreen
Specimen Collection Requirements
 The following is a job aid listing the specimen collection requirements for laboratory testing at Colchester East Hants Health Center. Specimens must be accompanied by the Patient Information Form G09.
The following is a job aid listing the specimen collection requirements for laboratory testing at Colchester East Hants Health Center. Specimens must be accompanied by the Patient Information Form G09.
MICROBIOLOGY SPECIMEN COLLECTION MANUAL
 Lee Memorial Health System Lee County, FL CLINICAL LABORATORY MICROBIOLOGY SPECIMEN COLLECTION MANUAL ACID FAST CULTURE Specimen Type see Specimen Chart ACID FAST STAIN see Specimen Chart Acid Fast stain
Lee Memorial Health System Lee County, FL CLINICAL LABORATORY MICROBIOLOGY SPECIMEN COLLECTION MANUAL ACID FAST CULTURE Specimen Type see Specimen Chart ACID FAST STAIN see Specimen Chart Acid Fast stain
Specimen Collection Requirements
 The following is a job aid listing the specimen collection requirements for laboratory testing at Colchester East Hants Health Center. Specimens must be accompanied by the Patient Information Form G09.
The following is a job aid listing the specimen collection requirements for laboratory testing at Colchester East Hants Health Center. Specimens must be accompanied by the Patient Information Form G09.
Update on Lyme Disease Surveillance in Wisconsin for Providers and Laboratories
 Update on Lyme Disease Surveillance in Wisconsin for Providers and Laboratories Christopher Steward Division of Public Health Wisconsin Department of Health Services 04/10/14 Protecting and promoting the
Update on Lyme Disease Surveillance in Wisconsin for Providers and Laboratories Christopher Steward Division of Public Health Wisconsin Department of Health Services 04/10/14 Protecting and promoting the
Patient Information Specimen Information Client Information. Specimen: EN255254W. Requisition: TSENG, JUSTINA J DOB: 10/26/1955 AGE: 59
 Report Status: Final Patient Information Specimen Information Client Information Specimen: EN255254W Client #: 44123510 MAIL001 Requisition: 0014895 Lab Ref #: PRO76199 GREENVILLE RANCHERIA 1425 MONTGOMERY
Report Status: Final Patient Information Specimen Information Client Information Specimen: EN255254W Client #: 44123510 MAIL001 Requisition: 0014895 Lab Ref #: PRO76199 GREENVILLE RANCHERIA 1425 MONTGOMERY
e-figure 1. The design of the study.
 e-figure 1. The design of the study. NDM, no diabetes; DM, diabetes; TB, tuberculosis; SN, sputum smear negative; SP, sputum smear positive; AFB, acid fast bacilli. e-figure 2. Representative H&E (A) and
e-figure 1. The design of the study. NDM, no diabetes; DM, diabetes; TB, tuberculosis; SN, sputum smear negative; SP, sputum smear positive; AFB, acid fast bacilli. e-figure 2. Representative H&E (A) and
Clinical Microbiology CLS 2019 Clinical Microbiology Laboratory Scheme Application Form
 Laboratory Scheme Application Form complete all sections below and return to LGC Standards Proficiency Testing by email, fax or post. Returning customer Lab ID: Purchase order no.: (compulsory) Round Despatch
Laboratory Scheme Application Form complete all sections below and return to LGC Standards Proficiency Testing by email, fax or post. Returning customer Lab ID: Purchase order no.: (compulsory) Round Despatch
Test Critical Low Critical High Alerting Category
 Test Critical Low Critical High Alerting CHEMISTRY Amylase > 400 U/L Emergency Dept only Amylase, Pancreatic >300 U/L Emergency Dept only Bicarb (Carbon Dioxide) 40 mmol/l Bilirubin, Total
Test Critical Low Critical High Alerting CHEMISTRY Amylase > 400 U/L Emergency Dept only Amylase, Pancreatic >300 U/L Emergency Dept only Bicarb (Carbon Dioxide) 40 mmol/l Bilirubin, Total
