9410 CARROLL PARK DRIVE, SAN DIEGO, CA P: (858) F:
|
|
|
- Andra Morris
- 6 years ago
- Views:
Transcription
1 About Prometheus Financial Information Website Address Management Team Employees Irritable Bowel Syndrome Inflammatory Bowel Disease (IBD) COMPANY FACT SHEET Prometheus Laboratories Inc. is committed to improving lives through the development and commercialization of novel pharmaceutical and diagnostic products that enable physicians to provide greater individualized patient care. Prometheus is a leader in applying the principles of personalized medicine to the diagnosis and treatment of gastrointestinal diseases and is also applying these principles to oncology. By integrating therapeutics and diagnostics, Prometheus can provide physicians with more targeted solutions to optimize care for their patients. Corporate offices in San Diego, California The Company reported 2010 earnings before interest, taxes, depreciation and amortization (EBITDA) of $138.9 million and net income of $48.2 million. The Company reported a 26% increase in net sales to $519.0 million for 2010, compared to $341.5 million in For the five-year period 2005 through 2010, the Company s net sales have grown by a compounded annual growth rate (CAGR) of 30%. Joseph Limber, President and Chief Executive Officer Mark Spring, Senior Vice President and Chief Financial Officer William Franzblau, Vice President, Legal Affairs Ron Rocca, General Manager, GI Products Tony Goosmann, General Manager, Oncology Toni Wayne, Vice President, Human Resources Prometheus currently has approximately 500 employees across the United States including over 300 employees in its San Diego headquarters. Markets We Serve According to the National Institute of Diabetes and Digestive and Kidney Diseases up to 60 million (20%) of the U.S. population may have symptoms of IBS, making it one of the most common disorders diagnosed by physicians. IBS is characterized most commonly by cramping, abdominal pain, bloating, constipation and diarrhea. Twice as many women as men are affected by IBS, of which more than 35% have diarrhea-predominant IBS (IBS-d). Inflammatory bowel disease refers to two chronic diseases that cause inflammation of the intestines: ulcerative colitis and Crohn's disease. IBD is estimated to affect approximately 1.2 million Americans, with roughly 60% of patients having ulcerative colitis and 40% having Crohn s Disease. Ulcerative colitis is an inflammatory disease of the large intestine, or colon. In ulcerative colitis, the inner lining (mucosa) of the intestine becomes inflamed and develops ulcers. Ulcerative colitis is often the most severe in the rectal area, which can cause frequent diarrhea. Mucus and blood often appear in the stool if the lining of the colon is damaged. Crohn's disease differs from ulcerative colitis in the areas of the bowel it involves; it most commonly affects the last part of the small intestine (called
2 Celiac Disease Prometheus IBD Serology 7 Prometheus Crohn s Prognostic Prometheus Celiac Serology & Genetics Prometheus Thiopurine Management MyCeliacID Lotronex the terminal ileum) and parts of the large intestine. However, it isn't limited to these areas and can attack any part of the digestive tract. Crohn's disease causes inflammation that extends much deeper into the layers of the intestinal wall and generally tends to involve the entire bowel wall, whereas ulcerative colitis affects only the lining of the bowel. Celiac disease is an immune-mediated digestive disease that is triggered by the consumption of gluten and damages the small intestine, thus interfering with the absorption of nutrients from food. Symptoms of celiac disease may vary significantly from person to person and include, but are not limited to, diarrhea, anemia, abdominal pain, skin rash and weight loss. The prevalence in the US is projected to be approximately 1% and is expected to increase significantly over the next several years, driven by broad-based educational campaigns, National Institutes of Health research funding, and growing awareness of the need for testing. The vast majority of celiac patients are currently undiagnosed. Our Diagnostic Products PROMETHEUS IBD Serology 7, the most comprehensive diagnostic test for inflammatory bowel disease, offers proprietary markers to help physicians diagnose inflammatory bowel disease (IBD) and to differentiate the subtypes - Crohn s disease and ulcerative colitis. The PROMTHEUS Crohn s Prognostic test combines six serologic markers and three genetic mutation markers to provide physicians with a personalized serogenetic profile for their patients. This test helps enable physicians to quantify a patient s risk of developing disease complications over time and assists physicians in determining optimal management strategies for their Crohn s patients. Prometheus is the leader in celiac testing and offers the most comprehensive test that includes a serology and genetic test to help physicians diagnose the disease. PROMETHEUS Thiopurine Metabolites helps keep patients drug metabolite levels within an optimal therapeutic range, thereby increasing the probability of a positive response and potentially reducing the need for stronger medications. Thiopurine metabolites testing helps identify some of the reasons for treatment failure and monitors for potential toxicity before it occurs. PROMETHEUS TPMT Enzyme and TPMT Genetics help physicians choose appropriate patients and guides dosing of Thiopurine therapy. MyCeliacID is the first do it yourself, saliva-based genetic test that identifies distinct genetic sequences associated with celiac disease. MyCeliacID provides individuals with their risk of developing the disease relative to the general population and, with a negative result, can virtually rule out celiac disease in a person s lifetime. Our Pharmaceutical Products Lotronex (alosetron HCI) Tablets is the only FDA-approved medicine to offer
3 Entocort EC multi-symptom relief for some women who suffer from severe IBS-d. Lotronex sales have almost tripled since Prometheus acquired the product in late Important Safety Information Lotronex is indicated for use only for women with severe diarrheapredominant IBS who have chronic IBS symptoms (generally lasting 6 months or longer), had anatomic or biochemical abnormalities of the gastrointestinal tract excluded, and not responded adequately to conventional therapy. Diarrhea-predominant IBS is severe if it includes diarrhea and one or more of the following: frequent and severe abdominal pain/discomfort, frequent bowel urgency or fecal incontinence, disability or restriction of daily activities due to IBS. Because of infrequent but serious gastrointestinal adverse reactions associated with LOTRONEX, the indication is restricted to those patients for whom the benefit-to-risk balance is most favorable. Clinical studies have not been performed to adequately confirm the benefits of LOTRONEX in men. Safety and effectiveness in pediatric patients have not been established. Use of LOTRONEX is not recommended in the pediatric population, based upon the risk of serious complications of constipation and ischemic colitis in adults. LOTRONEX has a boxed warning regarding serious gastrointestinal adverse events that have been reported with the use of LOTRONEX. These events, including ischemic colitis and serious complications of constipation, have resulted in hospitalizations, blood transfusions, surgery and fatalities. LOTRONEX should be discontinued immediately in patients who develop constipation or symptoms of ischemic colitis. LOTRONEX should not be resumed in patients who develop ischemic colitis. Prescribers should instruct patients who report constipation to immediately contact them if the constipation does not resolve after discontinuation of LOTRONEX. Patients with resolved constipation should resume LOTRONEX only on the advice of their treating prescriber. Contraindications to LOTRONEX include constipation, history of severe bowel or hepatic disorders, lack of understanding of the patient acknowledgement form and concomitant use of fluvoxamine. Only physicians who have enrolled in the Prometheus Prescribing Program for Lotronex, based on their understanding of the benefits and risks, should prescribe LOTRONEX. The Prescribing Program for Lotronex was implemented to help reduce risks of serious gastrointestinal adverse events. Please see full Prescribing Information for Lotronex. Entocort EC is the only FDA-approved drug for the induction and maintenance of clinical remission in mild to moderate Crohn s disease involving the ileum and/or the ascending colon. Entocort EC sales have grown almost 15-fold since Prometheus took over commercialization in Important Safety Information Since ENTOCORT EC (budesonide) Capsules is a glucocorticosteroid (GCS), general warnings about GCSs should be followed. GCSs can reduce the response of the hypothalamuspituitaryadrenal axis to stress.
4 Supplementation with a systemic GCS is recommended before surgery or other stress situations. Adrenocortical function monitoring may be required in patients being transferred to ENTOCORT EC from a systemic GCS, and the dose of the systemic GCS should be reduced cautiously. Patients on drugs that suppress the immune system are more susceptible to infections, which may be more severe, and should avoid exposure to infections such as chicken pox or measles. Caution should be taken in patients with tuberculosis, hypertension, diabetes mellitus, osteoporosis, peptic ulcer, glaucoma or cataracts, or with a family history of diabetes or glaucoma, or with any other condition where GCSs may have unwanted effects. Reduced liver function affects the elimination of GCSs, and increased systemic availability of oral budesonide has been observed in patients with liver cirrhosis. Patients with moderate to severe liver disease and patients who are concomitantly taking ketoconazole or any other CYP3A4 inhibitor should be closely monitored for increased signs and/or symptoms of hypercorticism. Reduction in the dose of ENTOCORT EC should be considered in these patients. Patients should be advised to avoid consuming grapefruits and grapefruit juice while being treated with ENTOCORT EC. Safety and effectiveness in pediatric, geriatric, and pregnant patients have not been established. ENTOCORT EC should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus. Hypoadrenalism may occur in infants born of mothers receiving corticosteroids during pregnancy. Budesonide is secreted in human milk. A decision should be made whether to discontinue nursing or to discontinue ENTOCORT EC, taking into account the clinical importance of ENTOCORT EC to the mother and the potential for serious adverse reactions in the nursing infant. The adverse event profile of ENTOCORT EC in 6 mg once daily clinical trial treatment (52-week) was similar to that of 9 mg daily clinical trial treatment (8-week). The most frequently reported adverse events in clinical trials with ENTOCORT EC were headache, respiratory infection, nausea, and symptoms of hypercorticism. Please see full Prescribing Information for ENTOCORT EC. Our Gastroenterology Pipeline Prometheus plans to launch a new and improved version of their IBD Serology 7 towards the end of This updated version of the market leading IBD diagnostic test may have improved accuracy thus adding clarity to the physician s diagnosis of Crohn s disease or ulcerative colitis. In 2012, Prometheus plans to launch a new test for detecting drug and antibody levels for the approximate 100,000 IBD patients treated with biologic therapy. This novel test will help allow physicians to make better treatment and dosing decisions to optimize care. Prometheus and Tarrot Laboratories, a business unit of Cedars-Sinai Medical Center, are collaborating on identifying genetic or serologic markers associated with clinical responses to anti-tnf therapies such as Cimzia, Humira and Remicade utilized in the treatment of IBD. Prometheus and The Regents of the University of California, Los Angeles
5 Proleukin RENCAREX CEER campus, are collaborating on the identification of biological markers of mucosal healing in patients with IBD. Mucosal healing, specifically repair of the damaged areas of the bowel in IBD patients, may be considered to be the ultimate goal of IBD therapy. Our Oncology Products Proleukin (aldesleukin) for injection is a recombinant human interleukin-2 for treatment in adults with metastatic melanoma and metastatic kidney cancer. Proleukin is a form of immunotherapy that enhances the body's natural immune system to help fight these types of cancer. Important Safety Information Therapy with Proleukin (aldesleukin) should be restricted to patients with normal cardiac and pulmonary functions as defined by thallium stress testing and formal pulmonary function testing. Extreme caution should be used in patients with a normal thallium stress test and a normal pulmonary function test who have a history of cardiac or pulmonary disease. Proleukin should be administered in a hospital setting under the supervision of a qualified physician experienced in the use of anticancer agents. An intensive care facility and specialists skilled in cardiopulmonary or intensive care medicine must be available. Proleukin administration has been associated with capillary leak syndrome (CLS) which is characterized by a loss of vascular tone and extravasation of plasma proteins and fluid into the extravascular space. CLS results in hypotension and reduced organ perfusion which may be severe and can result in death. CLS may be associated with cardiac arrhythmias (supraventricular and ventricular), angina, myocardial infarction, respiratory insufficiency requiring intubation, gastrointestinal bleeding or infarction, renal insufficiency, edema, and mental status changes. Proleukin treatment is associated with impaired neutrophil function (reduced chemotaxis) and with an increased risk of disseminated infection, including sepsis and bacterial endocarditis. Consequently, preexisting bacterial infections should be adequately treated prior to initiation of Proleukin therapy. Patients with indwelling central lines are particularly at risk for infection with gram positive microorganisms. Antibiotic prophylaxis with oxacillin, nafcillin, ciprofloxacin, or vancomycin has been associated with a reduced prevalence of staphylococcal infections. Proleukin administration should be withheld in patients developing moderate to severe lethargy or somnolence; continued administration may result in coma. Please see full Prescribing Information for Proleukin. Our Oncology Pipeline RENCAREX (girentuximab), which is being developed by Wilex AG, a biopharmaceutical company based in Munich Germany, is a Phase III product candidate for adjuvant use in non-metastatic clear-cell renal cell cancer (ccrcc). Prometheus has the commercialization rights in the United States for RENCAREX. Approximately 208,500 new cases of kidney cancer are diagnosed each year worldwide and the most prevalent form is ccrcc.
6 S, LOTRONEX, MyCeliacID and CEER are trademarks or registered trademarks of Prometheus Laboratories Inc. PROLEUKIN is a registered trademark of Novartis. RENCAREX is a registered trademark of Wilex AG ENTOCORT is a registered trademark of the AstraZeneca group of companies. CIMZIA is a registered trademark of the UCB group of companies. HUMIRA is a registered trademark of Abbott Laboratories. There is no adjuvant treatment currently approved by the FDA or EMA for patients after surgery. Prometheus proprietary CEER platform measures the expression and activation of specific cancer pathways with high levels of sensitivity and specificity using tissue or blood samples. The CEER platform has shown great promise to improve the selection and evaluation of the efficacy of drugs in development and in the clinical setting to enable real-time molecular profiling and monitoring of drug effectiveness. Prometheus and Bayer Schering Pharma AG are collaborating on multiple projects that partner the CEER platform with certain compounds in Bayer s broad oncology pipeline in an effort to stratify patients to appropriate drug candidates and potentially accelerate the development of novel oncology therapeutic products. REMICADE is a registered trademark of Centocor Ortho Biotech Inc. PROMETHEU
Understanding the Benefits and Risks
 LOTRONEX and its authorized generic alosetron hydrochloride: Understanding the Benefits and Risks The LOTRONEX REMS Program Prescriber Education Slide Deck LOTRONEX is a registered trademark of Prometheus
LOTRONEX and its authorized generic alosetron hydrochloride: Understanding the Benefits and Risks The LOTRONEX REMS Program Prescriber Education Slide Deck LOTRONEX is a registered trademark of Prometheus
LOTRONEX and its authorized generic alosetron hydrochloride:
 LOTRONEX and its authorized generic alosetron hydrochloride: Understanding the Benefits and Risks The Prescribing Program for LOTRONEX TM Prescriber Education Slide Deck PROMETHEUS and the Link Design
LOTRONEX and its authorized generic alosetron hydrochloride: Understanding the Benefits and Risks The Prescribing Program for LOTRONEX TM Prescriber Education Slide Deck PROMETHEUS and the Link Design
Clinical Policy: Alosetron (Lotronex) Reference Number: CP.CPA.65 Effective Date: Last Review Date: Line of Business: Medicaid Medi-Cal
 Clinical Policy: (Lotronex) Reference Number: CP.CPA.65 Effective Date: 11.16.16 Last Review Date: 11.17 Line of Business: Medicaid Medi-Cal Revision Log See Important Reminder at the end of this policy
Clinical Policy: (Lotronex) Reference Number: CP.CPA.65 Effective Date: 11.16.16 Last Review Date: 11.17 Line of Business: Medicaid Medi-Cal Revision Log See Important Reminder at the end of this policy
Xifaxan, Lotronex and Viberzi Prior Authorization and Quantity Limit Program Summary
 Xifaxan, Lotronex and Viberzi Prior Authorization and Quantity Limit Program Summary FDA APPROVED INDICATIONS DOSAGE 1,2 Lotronex (alosetron) a Indication For women with severe diarrheapredominant irritable
Xifaxan, Lotronex and Viberzi Prior Authorization and Quantity Limit Program Summary FDA APPROVED INDICATIONS DOSAGE 1,2 Lotronex (alosetron) a Indication For women with severe diarrheapredominant irritable
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: (Lotronex) Reference Number: CP.PMN.153 Effective Date: 11.16.16 Last Review Date: 11.18 Line of Business: Commercial, Medicaid Revision Log See Important Reminder at the end of this policy
Clinical Policy: (Lotronex) Reference Number: CP.PMN.153 Effective Date: 11.16.16 Last Review Date: 11.18 Line of Business: Commercial, Medicaid Revision Log See Important Reminder at the end of this policy
WARNING, CONTRAINDICATIONS, WARNINGS AND PRECAUTIONS,
 Celgene Corporation 86 Morris Avenue Summit, New Jersey 07901 Tel 908-673-9000 Fax 908-673-9001 October 2012 NEW Indication Announcement for ABRAXANE for Injectable Suspension (paclitaxel protein-bound
Celgene Corporation 86 Morris Avenue Summit, New Jersey 07901 Tel 908-673-9000 Fax 908-673-9001 October 2012 NEW Indication Announcement for ABRAXANE for Injectable Suspension (paclitaxel protein-bound
FERRING PHARMACEUTICALS. Enjoy The simple COR/939/2014/CH3
 Enjoy The simple pleasures of life COR/939/2014/CH3 Ulcerative colitis disrupts the simple pleasures in life Patients with ulcerative colitis may live with a considerable symptom burden despite medical
Enjoy The simple pleasures of life COR/939/2014/CH3 Ulcerative colitis disrupts the simple pleasures in life Patients with ulcerative colitis may live with a considerable symptom burden despite medical
REFERENCE CODE GDHC519DFR PUBLICAT ION DATE DECEMBER 2014 LOTRONEX (IRRITABLE BOWEL SYNDROME) FORECAST AND MARKET ANALYSIS TO 2023
 REFERENCE CODE GDHC519DFR PUBLICAT ION DATE DECEMBER 2014 LOTRONEX (IRRITABLE BOWEL SYNDROME) Executive Summary The Table below presents the key metrics of Prometheus Lotronex for irritable bowel syndrome
REFERENCE CODE GDHC519DFR PUBLICAT ION DATE DECEMBER 2014 LOTRONEX (IRRITABLE BOWEL SYNDROME) Executive Summary The Table below presents the key metrics of Prometheus Lotronex for irritable bowel syndrome
Treatment of Inflammatory Bowel Disease. Michael Weiss MD, FACG
 Treatment of Inflammatory Bowel Disease Michael Weiss MD, FACG What is IBD? IBD is an immune-mediated chronic intestinal disorder, characterized by chronic or relapsing inflammation within the GI tract.
Treatment of Inflammatory Bowel Disease Michael Weiss MD, FACG What is IBD? IBD is an immune-mediated chronic intestinal disorder, characterized by chronic or relapsing inflammation within the GI tract.
Certain genes passed on from parent to child increase the risk of developing Crohn's disease, if the right trigger occurs.
 Topic Page: Crohn's disease Definition: Crohn's disease from Benders' Dictionary of Nutrition and Food Technology Chronic inflammatory disease of the bowel, commonly the terminal ileum, of unknown aetiology,
Topic Page: Crohn's disease Definition: Crohn's disease from Benders' Dictionary of Nutrition and Food Technology Chronic inflammatory disease of the bowel, commonly the terminal ileum, of unknown aetiology,
Cimzia shows duration of response up to week 26 in moderate to severe Crohn s patients who failed the intravenous infusion treatment infliximab.
 C3073-0409 CIMZIA (certolizumab pegol) PROVIDES LONG-TERM REMISSION AND RESPONSE RATES IN INFLIXIMAB-REFRACTORY CROHN S PATIENTS Cimzia shows duration of response up to week 26 in moderate to severe Crohn
C3073-0409 CIMZIA (certolizumab pegol) PROVIDES LONG-TERM REMISSION AND RESPONSE RATES IN INFLIXIMAB-REFRACTORY CROHN S PATIENTS Cimzia shows duration of response up to week 26 in moderate to severe Crohn
(212) (347)
 EMBARGOED FOR MONDAY, JUNE 21, 2010: 3:00 P.M. EST For immediate release: June 21, 2010 Media Contact: Curtis Allen (212) 733-2096 (347) 443-5252 Investors Contact: Suzanne Harnett (212) 733-8009 Pfizer
EMBARGOED FOR MONDAY, JUNE 21, 2010: 3:00 P.M. EST For immediate release: June 21, 2010 Media Contact: Curtis Allen (212) 733-2096 (347) 443-5252 Investors Contact: Suzanne Harnett (212) 733-8009 Pfizer
REFERENCE CODE GDHC518DFR PUBLICAT ION DATE DECEMBER 2014 LINZESS (IRRITABLE BOWEL SYNDROME) - FORECAST AND MARKET ANALYSIS TO 2023
 REFERENCE CODE GDHC518DFR PUBLICAT ION DATE DECEMBER 2014 LINZESS (IRRITABLE BOWEL SYNDROME) - Executive Summary The Table below presents the key metrics of Ironwood/Actavis/Almirall/Astellas Linzess for
REFERENCE CODE GDHC518DFR PUBLICAT ION DATE DECEMBER 2014 LINZESS (IRRITABLE BOWEL SYNDROME) - Executive Summary The Table below presents the key metrics of Ironwood/Actavis/Almirall/Astellas Linzess for
Clinically proven to quickly relieve symptoms of common gastrointestinal disorders. TERRAGASTRO - Good health starts in the gut
 Clinically proven to quickly relieve symptoms of common gastrointestinal disorders GASTROINTESTINAL DISEASE Referred to as gastrointestinal diseases, they are common disorders which affect the esophagus,
Clinically proven to quickly relieve symptoms of common gastrointestinal disorders GASTROINTESTINAL DISEASE Referred to as gastrointestinal diseases, they are common disorders which affect the esophagus,
FDA Approves ABRAXANE for the First-Line Treatment of Advanced Non-Small Cell Lung Cancer
 October 12, 2012 FDA Approves ABRAXANE for the First-Line Treatment of Advanced Non-Small Cell Lung Cancer Approval Based on Significantly Improved Overall Response Rates in all Patients Regardless of
October 12, 2012 FDA Approves ABRAXANE for the First-Line Treatment of Advanced Non-Small Cell Lung Cancer Approval Based on Significantly Improved Overall Response Rates in all Patients Regardless of
How do I choose amongst medicines for inflammatory bowel disease. Maria T. Abreu, MD
 How do I choose amongst medicines for inflammatory bowel disease Maria T. Abreu, MD Overview of IBD Pathogenesis Bacterial Products Moderately Acutely Inflamed Chronic Inflammation = IBD Normal Gut Mildly
How do I choose amongst medicines for inflammatory bowel disease Maria T. Abreu, MD Overview of IBD Pathogenesis Bacterial Products Moderately Acutely Inflamed Chronic Inflammation = IBD Normal Gut Mildly
William Chey, MD University of Michigan Ann Arbor, MI
 Lin Chang, MD David Geffen School of Medicine at UCLA Los Angeles, CA William Chey, MD University of Michigan Ann Arbor, MI Mark Pimentel, MD Cedars-Sinai Medical Center Los Angeles, CA Accredited by Jointly
Lin Chang, MD David Geffen School of Medicine at UCLA Los Angeles, CA William Chey, MD University of Michigan Ann Arbor, MI Mark Pimentel, MD Cedars-Sinai Medical Center Los Angeles, CA Accredited by Jointly
Chapter 5: Common Digestive Problems from The Kansas State University Human Nutrition (HN 400) Flexbook by Brian Lindshield is in the public domain
 Chapter 5: Common Digestive Problems from The Kansas State University Human Nutrition (HN 400) Flexbook by Brian Lindshield is in the public domain in the United States. 5 Common Digestive Problems Before
Chapter 5: Common Digestive Problems from The Kansas State University Human Nutrition (HN 400) Flexbook by Brian Lindshield is in the public domain in the United States. 5 Common Digestive Problems Before
REFERENCE CODE GDHC1042FPR PUBLICAT ION DATE DECEMBER 2014 IRRITABLE BOWEL SYNDROME CURRENT AND FUTURE PLAYERS
 REFERENCE CODE GDHC1042FPR PUBLICAT ION DATE DECEMBER 2014 IRRITABLE BOWEL SYNDROME Executive Summary Sales of Irritable Bowel Syndrome Market The Irritable Bowel Syndrome (IBS) market in the 7MM (US,
REFERENCE CODE GDHC1042FPR PUBLICAT ION DATE DECEMBER 2014 IRRITABLE BOWEL SYNDROME Executive Summary Sales of Irritable Bowel Syndrome Market The Irritable Bowel Syndrome (IBS) market in the 7MM (US,
REFERENCE CODE GDHC525DFR PUBLICAT ION DATE DECEMBER 2014 TENAPANOR (IRRITABLE BOWEL SYNDROME) FORECAST AND MARKET ANALYSIS TO 2023
 REFERENCE CODE GDHC525DFR PUBLICAT ION DATE DECEMBER 2014 TENAPANOR (IRRITABLE BOWEL SYNDROME) Executive Summary The Table below presents the key metrics of AstraZeneca/Ardelyx s tenapanor for Irritable
REFERENCE CODE GDHC525DFR PUBLICAT ION DATE DECEMBER 2014 TENAPANOR (IRRITABLE BOWEL SYNDROME) Executive Summary The Table below presents the key metrics of AstraZeneca/Ardelyx s tenapanor for Irritable
Diarrhea may be: Acute (short-term, usually lasting several days), which is usually related to bacterial or viral infections.
 Pediatric Gastroenterology Conditions Evaluated and Treated Having a child suffer with abdominal pain, chronic eating problems, or other gastrointestinal disorders can be a very trying time for a parent.
Pediatric Gastroenterology Conditions Evaluated and Treated Having a child suffer with abdominal pain, chronic eating problems, or other gastrointestinal disorders can be a very trying time for a parent.
IBD Tools to Aid in the Accurate Diagnosis of Inflammatory Bowel Disease
 IBD Tools to Aid in the Accurate Diagnosis of Inflammatory Bowel Disease Inflammatory Bowel Disease Experts in Autoimmunity Inova Diagnostics offers a complete array of methods to aid in the diagnosis
IBD Tools to Aid in the Accurate Diagnosis of Inflammatory Bowel Disease Inflammatory Bowel Disease Experts in Autoimmunity Inova Diagnostics offers a complete array of methods to aid in the diagnosis
REFERENCE CODE GDHC521DFR PUBLICAT ION DATE DECEMBER 2014 ELUXADOLINE (IRRITABLE BOWEL SYNDROME) FORECAST AND MARKET ANALYSIS TO 2023
 REFERENCE CODE GDHC521DFR PUBLICAT ION DATE DECEMBER 2014 ELUXADOLINE (IRRITABLE BOWEL SYNDROME) Executive Summary The Table below presents the key metrics of Furiex (Actavis) s eluxadoline for Irritable
REFERENCE CODE GDHC521DFR PUBLICAT ION DATE DECEMBER 2014 ELUXADOLINE (IRRITABLE BOWEL SYNDROME) Executive Summary The Table below presents the key metrics of Furiex (Actavis) s eluxadoline for Irritable
Outline. Biologic Drugs in Inflammatory Bowel Disease Dr. Jason Etzel MD The Vancouver Clinic. Biologic Drugs. Biologic Drugs. Biologic Drugs Anti-TNF
 Outline Biologic Drugs in Inflammatory Bowel Disease Dr. Jason Etzel MD The Vancouver Clinic Types of biologic drugs How do they work? How effective are they? Safety/Toxicity concerns with biologics Biologic
Outline Biologic Drugs in Inflammatory Bowel Disease Dr. Jason Etzel MD The Vancouver Clinic Types of biologic drugs How do they work? How effective are they? Safety/Toxicity concerns with biologics Biologic
INFLAMMATORY BOWEL DISEASE. Jean-Paul Achkar, MD Center for Inflammatory Bowel Disease Cleveland Clinic
 INFLAMMATORY BOWEL DISEASE Jean-Paul Achkar, MD Center for Inflammatory Bowel Disease Cleveland Clinic WHAT IS INFLAMMATORY BOWEL DISEASE (IBD)? Chronic inflammation of the intestinal tract Two related
INFLAMMATORY BOWEL DISEASE Jean-Paul Achkar, MD Center for Inflammatory Bowel Disease Cleveland Clinic WHAT IS INFLAMMATORY BOWEL DISEASE (IBD)? Chronic inflammation of the intestinal tract Two related
I B D. etter than this. isease UNDERSTANDING INFLAMMATORY BOWEL DISEASES
 I B D m etter than this isease UNDERSTANDING INFLAMMATORY BOWEL DISEASES What types of people have learned how to manage their IBD? Athletes Musicians Firefighters DOCTORS HEROES Artists Presidents Actors
I B D m etter than this isease UNDERSTANDING INFLAMMATORY BOWEL DISEASES What types of people have learned how to manage their IBD? Athletes Musicians Firefighters DOCTORS HEROES Artists Presidents Actors
DIGESTIVE SYSTEM. Unit 2: Cells & Systems
 DIGESTIVE SYSTEM Unit 2: Cells & Systems CRACKER DIGESTION OBSERVATION Write down your observations: 1) What does it feel like initially (at the beginning)? 2) What does it feel like after 1 minutes in
DIGESTIVE SYSTEM Unit 2: Cells & Systems CRACKER DIGESTION OBSERVATION Write down your observations: 1) What does it feel like initially (at the beginning)? 2) What does it feel like after 1 minutes in
Digestion: Small and Large Intestines Pathology
 Digestion: Small and Large Intestines Pathology Dr. Ritamarie Loscalzo Medical Disclaimer: The information in this presentation is not intended to replace a one onone relationship with a qualified health
Digestion: Small and Large Intestines Pathology Dr. Ritamarie Loscalzo Medical Disclaimer: The information in this presentation is not intended to replace a one onone relationship with a qualified health
Fact Sheet. Zohydro ER (hydrocodone bitartrate) Extended-Release Capsules, CII
 Zohydro ER (hydrocodone bitartrate) Extended-Release Capsules, CII Fact Sheet Zohydro ER (hydrocodone bitartrate) Extended-Release Capsule, CII, is a long-acting (extendedrelease) type of pain medication
Zohydro ER (hydrocodone bitartrate) Extended-Release Capsules, CII Fact Sheet Zohydro ER (hydrocodone bitartrate) Extended-Release Capsule, CII, is a long-acting (extendedrelease) type of pain medication
WARNING: RISK OF SERIOUS DEHYDRATION IN PEDIATRIC PATIENTS
 June 5, 2018 Synergy Pharmaceuticals Highlights New Data at Digestive Disease Week (DDW) 2018 Linking Uroguanylin Deficiency to Chronic Idiopathic Constipation (CIC) and Irritable Bowel Syndrome with Constipation
June 5, 2018 Synergy Pharmaceuticals Highlights New Data at Digestive Disease Week (DDW) 2018 Linking Uroguanylin Deficiency to Chronic Idiopathic Constipation (CIC) and Irritable Bowel Syndrome with Constipation
Efficacy and Safety of Treatment for Pediatric IBD
 Efficacy and Safety of Treatment for Pediatric IBD Andrew B. Grossman MD Co-Director, Center for Pediatric Inflammatory Bowel Disease Associate Professor of Clinical Pediatrics Division of Gastroenterology,
Efficacy and Safety of Treatment for Pediatric IBD Andrew B. Grossman MD Co-Director, Center for Pediatric Inflammatory Bowel Disease Associate Professor of Clinical Pediatrics Division of Gastroenterology,
Crohn's Disease. What causes Crohn s disease? What are the symptoms?
 Crohn's Disease Crohn s disease is an ongoing disorder that causes inflammation of the digestive tract, also referred to as the gastrointestinal (GI) tract. Crohn s disease can affect any area of the GI
Crohn's Disease Crohn s disease is an ongoing disorder that causes inflammation of the digestive tract, also referred to as the gastrointestinal (GI) tract. Crohn s disease can affect any area of the GI
Medical Therapy for Pediatric IBD: Efficacy and Safety
 Medical Therapy for Pediatric IBD: Efficacy and Safety Betsy Maxwell, MD Assistant Professor of Clinical Pediatrics Division of Gastroenterology, Hepatology, and Nutrition Pediatric IBD: Defining Remission
Medical Therapy for Pediatric IBD: Efficacy and Safety Betsy Maxwell, MD Assistant Professor of Clinical Pediatrics Division of Gastroenterology, Hepatology, and Nutrition Pediatric IBD: Defining Remission
Ulcerative Colitis. ulcerative colitis usually only affects the colon.
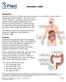 Ulcerative Colitis Introduction Ulcerative colitis is an inflammatory bowel disease. It is one of the 2 most common inflammatory bowel diseases. The other one is Crohn s disease. Ulcerative colitis and
Ulcerative Colitis Introduction Ulcerative colitis is an inflammatory bowel disease. It is one of the 2 most common inflammatory bowel diseases. The other one is Crohn s disease. Ulcerative colitis and
PEDIATRIC INFLAMMATORY BOWEL DISEASE
 PEDIATRIC INFLAMMATORY BOWEL DISEASE Alexis Rodriguez, MD Pediatric Gastroenterology Advocate Children s Hospital Disclosers Abbott Nutrition - Speaker Inflammatory Bowel Disease Chronic inflammatory disease
PEDIATRIC INFLAMMATORY BOWEL DISEASE Alexis Rodriguez, MD Pediatric Gastroenterology Advocate Children s Hospital Disclosers Abbott Nutrition - Speaker Inflammatory Bowel Disease Chronic inflammatory disease
DRAFT. Remission rates, calculated using observed case (OC) analyses were as follows: Year 1 Year 2 Year 3 Year 4 All patients 62.
 DRAFT New Efficacy Data Shows Cimzia (certolizumab pegol) Provides Long-Term Remission of Moderate to Severe Crohn s Disease Regardless of Prior Anti-TNF Exposure, According to Data Presented at DDW Oral
DRAFT New Efficacy Data Shows Cimzia (certolizumab pegol) Provides Long-Term Remission of Moderate to Severe Crohn s Disease Regardless of Prior Anti-TNF Exposure, According to Data Presented at DDW Oral
ALUNBRIG (brigatinib) Dosing Guide
 ALUNBRIG (brigatinib) Dosing Guide INDICATION ALUNBRIG (brigatinib) is indicated for the treatment of patients with anaplastic lymphoma kinase (ALK)-positive metastatic non-small cell lung cancer (NSCLC)
ALUNBRIG (brigatinib) Dosing Guide INDICATION ALUNBRIG (brigatinib) is indicated for the treatment of patients with anaplastic lymphoma kinase (ALK)-positive metastatic non-small cell lung cancer (NSCLC)
GLEEVEC PATIENT RESOURCES
 GLEEVEC TALKING WITH YOUR DOCTOR ABOUT KIT+ GIST AND ITS TREATMENT When you have KIT-positive gastrointestinal stromal tumor (KIT+ GIST), you will likely have many questions and may, at times, feel overwhelmed
GLEEVEC TALKING WITH YOUR DOCTOR ABOUT KIT+ GIST AND ITS TREATMENT When you have KIT-positive gastrointestinal stromal tumor (KIT+ GIST), you will likely have many questions and may, at times, feel overwhelmed
REVLIMID IN COMBINATION WITH DEXAMETHASONE snda GRANTED APPROVAL BY FDA FOR TREATMENT OF MULTIPLE MYELOMA
 Contact: Robert J. Hugin Brian P. Gill President and COO Senior Director, PR/IR Celgene Corporation Celgene Corporation (908) 673-9102 (908) 673-9530 REVLIMID IN COMBINATION WITH DEXAMETHASONE snda GRANTED
Contact: Robert J. Hugin Brian P. Gill President and COO Senior Director, PR/IR Celgene Corporation Celgene Corporation (908) 673-9102 (908) 673-9530 REVLIMID IN COMBINATION WITH DEXAMETHASONE snda GRANTED
Iroko Pharmaceuticals Announces Acceptance for Filing of ZORVOLEX snda for the Treatment of Osteoarthritis Pain in Adults
 Iroko Pharmaceuticals Announces Acceptance for Filing of ZORVOLEX snda for the Treatment of Osteoarthritis Pain in Adults First Lower Dose NSAID Using SoluMatrix Fine Particle Technology to be Reviewed
Iroko Pharmaceuticals Announces Acceptance for Filing of ZORVOLEX snda for the Treatment of Osteoarthritis Pain in Adults First Lower Dose NSAID Using SoluMatrix Fine Particle Technology to be Reviewed
(+1) Matti Ojanen (+44) Investor Contact: Jennifer M. Davis (+1)
 For immediate release: October 24, 2012 Media Contact: Victoria Davis (+1) 347-558-3455 Matti Ojanen (+44) 7557-202-394 Investor Contact: Jennifer M. Davis (+1) 212-733-0717 Pfizer s XALKORI Receives Conditional
For immediate release: October 24, 2012 Media Contact: Victoria Davis (+1) 347-558-3455 Matti Ojanen (+44) 7557-202-394 Investor Contact: Jennifer M. Davis (+1) 212-733-0717 Pfizer s XALKORI Receives Conditional
Testimony Submitted by: Eric Zuckerman, DO Bloomfield Hill, MI Board Chairman, Pediatric IBD Foundation
 Testimony Submitted by: Carlo Di Lorenzo, MD Nationwide Children s Hospital, Columbus, OH President, North American Society for Pediatric Gastroenterology, Hepatology and Nutrition Benjamin Gold, MD, Children's
Testimony Submitted by: Carlo Di Lorenzo, MD Nationwide Children s Hospital, Columbus, OH President, North American Society for Pediatric Gastroenterology, Hepatology and Nutrition Benjamin Gold, MD, Children's
SER-287 Phase 1b topline study results in patients with mild-to-moderate Ulcerative Colitis October 2, 2017
 SER-287 Phase 1b topline study results in patients with mild-to-moderate Ulcerative Colitis October 2, 2017 Leading the Microbiome Revolution Forward Looking Statements Some of the statements in this presentation
SER-287 Phase 1b topline study results in patients with mild-to-moderate Ulcerative Colitis October 2, 2017 Leading the Microbiome Revolution Forward Looking Statements Some of the statements in this presentation
OTEZLA (Apremilast) Showed Meaningful Improvements in Clinical and Quality of Life Measures of Psoriasis Beyond Those Captured by Assessing Skin Alone
 OTEZLA (Apremilast) Showed Meaningful Improvements in Clinical and Quality of Life Measures of Psoriasis Beyond Those Captured by Assessing Skin Alone Patients with moderate to severe plaque psoriasis
OTEZLA (Apremilast) Showed Meaningful Improvements in Clinical and Quality of Life Measures of Psoriasis Beyond Those Captured by Assessing Skin Alone Patients with moderate to severe plaque psoriasis
Efficacy and Safety of Treatment for Pediatric IBD
 Efficacy and Safety of Treatment for Pediatric IBD Andrew B. Grossman MD Co-Director, Center for Pediatric Inflammatory Bowel Disease Assistant Professor of Clinical Pediatrics Division of Gastroenterology,
Efficacy and Safety of Treatment for Pediatric IBD Andrew B. Grossman MD Co-Director, Center for Pediatric Inflammatory Bowel Disease Assistant Professor of Clinical Pediatrics Division of Gastroenterology,
POMALYST (pomalidomide) for Previously Treated Multiple Myeloma
 POMALYST (pomalidomide) for Previously Treated What is POMALYST? POMALYST (pomalidomide) capsule is an oral immunomodulatory therapy (a thalidomide analogue) indicated for patients with multiple myeloma
POMALYST (pomalidomide) for Previously Treated What is POMALYST? POMALYST (pomalidomide) capsule is an oral immunomodulatory therapy (a thalidomide analogue) indicated for patients with multiple myeloma
Agreed Core Safety Profile for Budesonide nasal spray suspension and Budesonide nasal powder
 CSP Drug Budesonide Substance Date 13 Oct 2011 rev 11Nov Supersedes 18 Aug 2011 Agreed Core Safety Profile for DK/H/PSUR/0041/001 TABLE OF CONTENTS PAGE TITLE PAGE... 1 TABLE OF CONTENTS... 2 Introduction...
CSP Drug Budesonide Substance Date 13 Oct 2011 rev 11Nov Supersedes 18 Aug 2011 Agreed Core Safety Profile for DK/H/PSUR/0041/001 TABLE OF CONTENTS PAGE TITLE PAGE... 1 TABLE OF CONTENTS... 2 Introduction...
Medical therapies and IBD
 Medical therapies and IBD Although there is no cure for IBD, there are many treatment options available. There is no standard treatment for IBD that is effective in all situations or for all patients,
Medical therapies and IBD Although there is no cure for IBD, there are many treatment options available. There is no standard treatment for IBD that is effective in all situations or for all patients,
Phase III ARISER trial data ASCO 2013 RENCAREX - Unique targeted antibody therapy for adjuvant treatment of non-metastatic clear cell renal cell
 Phase III ARISER trial data ASCO 2013 RENCAREX - Unique targeted antibody therapy for adjuvant treatment of non-metastatic clear cell renal cell carcinoma patients June 2013 Carbonic Anhydrase IX (CAIX):
Phase III ARISER trial data ASCO 2013 RENCAREX - Unique targeted antibody therapy for adjuvant treatment of non-metastatic clear cell renal cell carcinoma patients June 2013 Carbonic Anhydrase IX (CAIX):
TIVORBEX Now Available in U.S. Pharmacies for the Treatment of Acute Pain
 TIVORBEX Now Available in U.S. Pharmacies for the Treatment of Acute Pain Second Low-Dose SoluMatrix NSAID from Iroko Now Available by Prescription PHILADELPHIA, June 29, 2015 Iroko Pharmaceuticals, LLC,
TIVORBEX Now Available in U.S. Pharmacies for the Treatment of Acute Pain Second Low-Dose SoluMatrix NSAID from Iroko Now Available by Prescription PHILADELPHIA, June 29, 2015 Iroko Pharmaceuticals, LLC,
Top 10 Things you need to know about IBD. Suresh Pola, MD Kaiser San Diego
 Top 10 Things you need to know about IBD Suresh Pola, MD Kaiser San Diego Top 10 Things to Know: IBD What you can eat How to treat the pain Not all diarrhea is a flare Ways to reduce your risk of getting
Top 10 Things you need to know about IBD Suresh Pola, MD Kaiser San Diego Top 10 Things to Know: IBD What you can eat How to treat the pain Not all diarrhea is a flare Ways to reduce your risk of getting
GLEEVEC (imatinib mesylate) PATIENT RESOURCES
 Talking with your doctor about Ph+ CML and its treatment When you have Philadelphia chromosome-positive chronic myeloid leukemia (Ph+ CML), it s important to take an active role in your care. You will
Talking with your doctor about Ph+ CML and its treatment When you have Philadelphia chromosome-positive chronic myeloid leukemia (Ph+ CML), it s important to take an active role in your care. You will
Approval based on the successful BFORE Phase 3 study conducted by Avillion under a collaborative development agreement with Pfizer
 Avillion Announces US Approval of Pfizer s BOSULIF (bosutinib) for the Treatment of Patients with Newly-Diagnosed Ph+ Chronic Myelogenous Leukemia (CML) Approval based on the successful BFORE Phase 3 study
Avillion Announces US Approval of Pfizer s BOSULIF (bosutinib) for the Treatment of Patients with Newly-Diagnosed Ph+ Chronic Myelogenous Leukemia (CML) Approval based on the successful BFORE Phase 3 study
Metformin Hydrochloride
 Metformin Hydrochloride 500 mg, 850 mg, 500 mg LA and 750 mg LA Tablet Description Informet is a preparation of metformin hydrochloride that belongs to a biguanide class of oral antidiabetic drugs. Metformin
Metformin Hydrochloride 500 mg, 850 mg, 500 mg LA and 750 mg LA Tablet Description Informet is a preparation of metformin hydrochloride that belongs to a biguanide class of oral antidiabetic drugs. Metformin
THERAPIES AND BIOMARKERS FOR INFLAMMATORY BOWEL DISEASE
 THERAPIES AND BIOMARKERS FOR INFLAMMATORY BOWEL DISEASE PHM125A January 2013 Willem Westra Project Analyst ISBN: 0-89336-715-X BCC Research 49 Walnut Park, Building 2 Wellesley, MA 02481 866-285-7215,
THERAPIES AND BIOMARKERS FOR INFLAMMATORY BOWEL DISEASE PHM125A January 2013 Willem Westra Project Analyst ISBN: 0-89336-715-X BCC Research 49 Walnut Park, Building 2 Wellesley, MA 02481 866-285-7215,
Children Enteric coated tablet : 1-3 mg/kg per day in divided doses.
 Ultrafen Tablet/SR Tablet/Suppository/Gel Description Ultrafen is a preparation of Diclofenac is a non-steroidal antiinflammatory agent with marked analgesic, anti-inflammatory and antipyretic properties.
Ultrafen Tablet/SR Tablet/Suppository/Gel Description Ultrafen is a preparation of Diclofenac is a non-steroidal antiinflammatory agent with marked analgesic, anti-inflammatory and antipyretic properties.
NEW ZEALAND DATA SHEET
 NEW ZEALAND DATA SHEET 1 PRODUCT NAME (strength pharmaceutical form) ENTOCORT 3 mg capsules 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each capsule contains 3 mg budesonide For the full list of excipients,
NEW ZEALAND DATA SHEET 1 PRODUCT NAME (strength pharmaceutical form) ENTOCORT 3 mg capsules 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each capsule contains 3 mg budesonide For the full list of excipients,
Celgene Receives Positive CHMP Opinion for ABRAXANE in Combination with Gemcitabine as Treatment for Patients with Metastatic Pancreatic Cancer
 November 22, 2013 Celgene Receives Positive CHMP Opinion for ABRAXANE in Combination with Gemcitabine as Treatment for Patients with Metastatic Pancreatic Cancer BOUDRY, Switzerland--(BUSINESS WIRE)--Celgene
November 22, 2013 Celgene Receives Positive CHMP Opinion for ABRAXANE in Combination with Gemcitabine as Treatment for Patients with Metastatic Pancreatic Cancer BOUDRY, Switzerland--(BUSINESS WIRE)--Celgene
Outcomes from treating bile acid malabsorption (BAM) using a multidisciplinary approach
 Outcomes from treating bile acid malabsorption (BAM) using a multidisciplinary approach Gupta A et al. Support Care Cancer. 2015 Oct;23(10):2881-90 Prescribing information can be found at the end of this
Outcomes from treating bile acid malabsorption (BAM) using a multidisciplinary approach Gupta A et al. Support Care Cancer. 2015 Oct;23(10):2881-90 Prescribing information can be found at the end of this
Barbara G. Wells, PharmD, FASHP, FCCP, BCPP Dean and Professor School of Pharmacy, The University of Mississippi Oxford, Mississippi
 Barbara G. Wells, PharmD, FASHP, FCCP, BCPP Dean and Professor School of Pharmacy, The University of Mississippi Oxford, Mississippi Joseph T. DiPiro, PharmD, FCCP Panoz Professor of Pharmacy, College
Barbara G. Wells, PharmD, FASHP, FCCP, BCPP Dean and Professor School of Pharmacy, The University of Mississippi Oxford, Mississippi Joseph T. DiPiro, PharmD, FCCP Panoz Professor of Pharmacy, College
Labeled Uses: Treatment of Clostiridum Difficile associated diarrhea (CDAD)
 Brand Name: Dificid Generic Name: fidaxomicin Manufacturer 1,2,3,4,5 : Optimer Pharmaceuticals, Inc. Drug Class 1,2,3,4,5 : Macrolide Antibiotic Uses 1,2,3,4,5 : Labeled Uses: Treatment of Clostiridum
Brand Name: Dificid Generic Name: fidaxomicin Manufacturer 1,2,3,4,5 : Optimer Pharmaceuticals, Inc. Drug Class 1,2,3,4,5 : Macrolide Antibiotic Uses 1,2,3,4,5 : Labeled Uses: Treatment of Clostiridum
DOSING GUIDE. Indications. Important Safety Information. Enable the immune system. RECOGNIZE. RESPOND.
 DOSING GUIDE For patients with unresectable Stage III NSCLC following concurrent CRT For patients with locally advanced or metastatic UC previously treated with platinum-based therapy Enable the immune
DOSING GUIDE For patients with unresectable Stage III NSCLC following concurrent CRT For patients with locally advanced or metastatic UC previously treated with platinum-based therapy Enable the immune
Phase 3 Top-Line Results Show IBRANCE in Combination with Fulvestrant Meets Progression-Free Survival (PFS) Primary Endpoint
 For immediate release: April 15, 2015 Media Contact: Sally Beatty (212) 733-6566 Investor Contact: Ryan Crowe (212) 733-8160 Pfizer Announces PALOMA-3 Trial for IBRANCE (Palbociclib) Stopped Early Due
For immediate release: April 15, 2015 Media Contact: Sally Beatty (212) 733-6566 Investor Contact: Ryan Crowe (212) 733-8160 Pfizer Announces PALOMA-3 Trial for IBRANCE (Palbociclib) Stopped Early Due
Inflammatory Bowel Disease Medical Exam Questionnaire
 Patient Name: MR: Date: Name DOB / / Age Marital Status Race Gender M / F Height Present Weight Usual Weight Insurance Managed Care Self referral Yes No Yes No Yes No Primary Care Physician Referring Physician
Patient Name: MR: Date: Name DOB / / Age Marital Status Race Gender M / F Height Present Weight Usual Weight Insurance Managed Care Self referral Yes No Yes No Yes No Primary Care Physician Referring Physician
Iroko Pharmaceuticals Receives FDA Approval for VIVLODEX - First Low Dose SoluMatrix Meloxicam for Osteoarthritis Pain
 Iroko Pharmaceuticals Receives FDA Approval for VIVLODEX - First Low Dose SoluMatrix Meloxicam for Osteoarthritis Pain VIVLODEX Developed to Align with FDA NSAID Recommendations Proven Efficacy at Low
Iroko Pharmaceuticals Receives FDA Approval for VIVLODEX - First Low Dose SoluMatrix Meloxicam for Osteoarthritis Pain VIVLODEX Developed to Align with FDA NSAID Recommendations Proven Efficacy at Low
IBD 101. Ronen Stein, MD Assistant Professor of Clinical Pediatrics Division of Gastroenterology, Hepatology, and Nutrition
 IBD 101 Ronen Stein, MD Assistant Professor of Clinical Pediatrics Division of Gastroenterology, Hepatology, and Nutrition Objectives Identify factors involved in the development of inflammatory bowel
IBD 101 Ronen Stein, MD Assistant Professor of Clinical Pediatrics Division of Gastroenterology, Hepatology, and Nutrition Objectives Identify factors involved in the development of inflammatory bowel
Immunocore and MedImmune announce new collaboration to conduct immuno-oncology combination trials in melanoma
 PRESS RELEASE IMMUNOCORE LIMITED Immunocore and MedImmune announce new collaboration to conduct immuno-oncology combination trials in melanoma (Oxford, UK, 16 April 2015) Immunocore Limited, a world-leading
PRESS RELEASE IMMUNOCORE LIMITED Immunocore and MedImmune announce new collaboration to conduct immuno-oncology combination trials in melanoma (Oxford, UK, 16 April 2015) Immunocore Limited, a world-leading
FURAMIST Nasal Spray (Fluticasone furoate )
 Published on: 21 Jan 2016 FURAMIST Nasal Spray (Fluticasone furoate ) Composition Each spray contains: Fluticasone furoate 27.5 mcg Dosage Form Aqueous intranasal spray Pharmacology Pharmacodynamics Fluticasone
Published on: 21 Jan 2016 FURAMIST Nasal Spray (Fluticasone furoate ) Composition Each spray contains: Fluticasone furoate 27.5 mcg Dosage Form Aqueous intranasal spray Pharmacology Pharmacodynamics Fluticasone
There is no single IBD diet
 Nutrition and IBD There is no single IBD diet Nutrition plays an important role in health, during times of IBD disease activity as well as during remission. Although diet does not cause or cure IBD, the
Nutrition and IBD There is no single IBD diet Nutrition plays an important role in health, during times of IBD disease activity as well as during remission. Although diet does not cause or cure IBD, the
taking charge of your options For adult patients with metastatic melanoma
 For adult patients with metastatic melanoma taking charge of your options Proleukin (aldesleukin) is indicated for the treatment of adults with metastatic renal cell carcinoma or metastatic melanoma Complete
For adult patients with metastatic melanoma taking charge of your options Proleukin (aldesleukin) is indicated for the treatment of adults with metastatic renal cell carcinoma or metastatic melanoma Complete
IBD 101. Ronen Stein, MD Assistant Professor of Clinical Pediatrics Division of Gastroenterology, Hepatology, and Nutrition
 IBD 101 Ronen Stein, MD Assistant Professor of Clinical Pediatrics Division of Gastroenterology, Hepatology, and Nutrition Objectives Identify factors involved in the development of inflammatory bowel
IBD 101 Ronen Stein, MD Assistant Professor of Clinical Pediatrics Division of Gastroenterology, Hepatology, and Nutrition Objectives Identify factors involved in the development of inflammatory bowel
Factsheet LINACLOTIDE (Constella ) Irritable Bowel Syndrome constipation predominant (IBS-C)
 North Central London Joint Formulary Committee Factsheet LINACLOTIDE (Constella ) Irritable Bowel Syndrome constipation predominant (IBS-C) Start date: September 2018 Review date: September 2021 Document
North Central London Joint Formulary Committee Factsheet LINACLOTIDE (Constella ) Irritable Bowel Syndrome constipation predominant (IBS-C) Start date: September 2018 Review date: September 2021 Document
Treating Crohn s and Colitis in the ASC
 Treating Crohn s and Colitis in the ASC Kimberly M Persley, MD Texas Digestive Disease consultants TASC Meeting Outline IBD 101 Diagnosis Treatment Burden of Disease Role of ASC Inflammatory Bowel Disease
Treating Crohn s and Colitis in the ASC Kimberly M Persley, MD Texas Digestive Disease consultants TASC Meeting Outline IBD 101 Diagnosis Treatment Burden of Disease Role of ASC Inflammatory Bowel Disease
Increased colonic bile acid exposure: a relevant factor for symptoms and treatment in irritable bowel syndrome (IBS)
 Increased colonic bile acid exposure: a relevant factor for symptoms and treatment in irritable bowel syndrome (IBS) Bajor A et al. Gut 2015; 64: 84-92. Prescribing information can be found at the end
Increased colonic bile acid exposure: a relevant factor for symptoms and treatment in irritable bowel syndrome (IBS) Bajor A et al. Gut 2015; 64: 84-92. Prescribing information can be found at the end
vedolizumab (Entyvio )
 vedolizumab (Entyvio ) Applies to all products administered or underwritten by Blue Cross and Blue Shield of Louisiana and its subsidiary, HMO Louisiana, Inc.(collectively referred to as the Company ),
vedolizumab (Entyvio ) Applies to all products administered or underwritten by Blue Cross and Blue Shield of Louisiana and its subsidiary, HMO Louisiana, Inc.(collectively referred to as the Company ),
Genentech Statement on Counterfeit Drug Labeled as Avastin (bevacizumab) in the United States
 Genentech Statement on Counterfeit Drug Labeled as Avastin (bevacizumab) in the United States SOUTH SAN FRANCISCO, Calif. February 14, 2012 Roche and Genentech have been informed that a counterfeit product,
Genentech Statement on Counterfeit Drug Labeled as Avastin (bevacizumab) in the United States SOUTH SAN FRANCISCO, Calif. February 14, 2012 Roche and Genentech have been informed that a counterfeit product,
Western Locality Shared care information ~ Azathioprine and Mercaptopurine
 The following guidelines are currently under review. In the interim, the guidelines remain valid; if GPs have any specific concerns or questions, they should seek advice from the specialist with whom they
The following guidelines are currently under review. In the interim, the guidelines remain valid; if GPs have any specific concerns or questions, they should seek advice from the specialist with whom they
Algorithms & Information Sheets
 Minor Ailment Scheme Algorithms & Information Sheets Acute Diarrhoea Diarrhoea is an increased frequency, fluidity or volume of the bowel movements with the passage of soft and watery stools as compared
Minor Ailment Scheme Algorithms & Information Sheets Acute Diarrhoea Diarrhoea is an increased frequency, fluidity or volume of the bowel movements with the passage of soft and watery stools as compared
TARCEVA NEARLY DOUBLED THE TIME PEOPLE WITH A GENETICALLY DISTINCT TYPE OF LUNG CANCER LIVED WITHOUT THEIR DISEASE GETTING WORSE
 NEWS RELEASE Genentech Contacts: Media Contact: Charlotte Arnold (650) 467-6800 Advocacy Contact: Jen Mills (650) 467-6722 Investor Contacts: Thomas Kudsk Larsen (650) 467-2016 Karl Mahler 011 41 61 687
NEWS RELEASE Genentech Contacts: Media Contact: Charlotte Arnold (650) 467-6800 Advocacy Contact: Jen Mills (650) 467-6722 Investor Contacts: Thomas Kudsk Larsen (650) 467-2016 Karl Mahler 011 41 61 687
U.S. FDA Approves New Indication for the Use of XTANDI (enzalutamide) Capsules for Patients with Metastatic Castration-Resistant Prostate Cancer
 Astellas Contact: Medivation Contact: Tyler Marciniak Rick Bierly Director, Communications and Advocacy Chief Financial Officer (847) 736-7145 (415) 543-3470 tyler.marciniak@astellas.com Anne Bowdidge
Astellas Contact: Medivation Contact: Tyler Marciniak Rick Bierly Director, Communications and Advocacy Chief Financial Officer (847) 736-7145 (415) 543-3470 tyler.marciniak@astellas.com Anne Bowdidge
FDA APPROVES TARCEVA (ERLOTINIB) TABLETS AND COBAS EGFR MUTATION TEST FOR SPECIFIC TYPE OF LUNG CANCER
 Genentech Contacts: Media: Holli Dickson (650) 467-6800 Advocacy: Jen Mills (650) 467-6722 Investors: Thomas Kudsk Larsen (650) 467-2016 Karl Mahler 011 41 61 687 8503 Astellas Contacts: Media: Jenny Kite
Genentech Contacts: Media: Holli Dickson (650) 467-6800 Advocacy: Jen Mills (650) 467-6722 Investors: Thomas Kudsk Larsen (650) 467-2016 Karl Mahler 011 41 61 687 8503 Astellas Contacts: Media: Jenny Kite
Emflaza. (deflazacort) New Product Slideshow
 Emflaza (deflazacort) New Product Slideshow Introduction Brand name: Emflaza Generic name: Deflazacort Pharmacological class: Corticosteroid Strength and Formulation: 6mg, 18mg, 30mg, 36mg tablets; 22.75mg/mL
Emflaza (deflazacort) New Product Slideshow Introduction Brand name: Emflaza Generic name: Deflazacort Pharmacological class: Corticosteroid Strength and Formulation: 6mg, 18mg, 30mg, 36mg tablets; 22.75mg/mL
Abiraterone Acetate is an antiandrogen used in the treatment of Castration-Resistant Prostate Cancer(CRPC).
 For the use only of an Oncologist or a Hospital or a Laboratory ABIRATERONE ACETATE TABLETS Zabiteron-250 COMPOSITION Abiraterone Acetate Tablets 250mg Each uncoated tablets contains: Abiraterone Acetate
For the use only of an Oncologist or a Hospital or a Laboratory ABIRATERONE ACETATE TABLETS Zabiteron-250 COMPOSITION Abiraterone Acetate Tablets 250mg Each uncoated tablets contains: Abiraterone Acetate
SUCRALFATE TABLETS, USP
 1234567890 10 210002-01 SUCRALFATE TABLETS, USP DESCRIPTION Sucralfate is an -D-glucopyranoside, -D-fructofuranosyl-, octakis-(hydrogen sulfate), aluminum complex. It has the following structural formula:
1234567890 10 210002-01 SUCRALFATE TABLETS, USP DESCRIPTION Sucralfate is an -D-glucopyranoside, -D-fructofuranosyl-, octakis-(hydrogen sulfate), aluminum complex. It has the following structural formula:
Available at American Society of Clinical Oncology: 3
 Addition of Tarceva (erlotinib) to Nexavar (sorafenib) did not Provide Additional Benefit to Patients with Unresectable Liver Cancer Versus Nexavar alone in Phase 3 Trial Wayne, NJ, South San Francisco,
Addition of Tarceva (erlotinib) to Nexavar (sorafenib) did not Provide Additional Benefit to Patients with Unresectable Liver Cancer Versus Nexavar alone in Phase 3 Trial Wayne, NJ, South San Francisco,
Rydapt. Summary of the Risk Management Plan (RMP) for Rydapt (midostaurin)
 Regulatory Affairs Rydapt 25 mg soft capsules Summary of the Risk Management Plan (RMP) for Rydapt (midostaurin) Document version: 01 Document status: Document Date: Final 02-June-2017 1 of 6 Summary of
Regulatory Affairs Rydapt 25 mg soft capsules Summary of the Risk Management Plan (RMP) for Rydapt (midostaurin) Document version: 01 Document status: Document Date: Final 02-June-2017 1 of 6 Summary of
Immodium / loprarmide
 Immodium / loprarmide IMODIUM (loperamide hydrochloride) is indicated for the control and symptomatic relief of acute nonspecific diarrhea and of chronic diarrhea associated with inflammatory bowel disease.
Immodium / loprarmide IMODIUM (loperamide hydrochloride) is indicated for the control and symptomatic relief of acute nonspecific diarrhea and of chronic diarrhea associated with inflammatory bowel disease.
FIRST RESULTS OF NEW DATA OF ABRAXANE IN COMBINATION WITH ATEZOLIZUMAB PRESENTED AT ESMO 2018
 FIRST RESULTS OF NEW DATA OF ABRAXANE IN COMBINATION WITH ATEZOLIZUMAB PRESENTED AT ESMO 2018 IMpassion130 reports first positive Phase III study results for a chemotherapy/immunotherapy (ABRAXANE plus
FIRST RESULTS OF NEW DATA OF ABRAXANE IN COMBINATION WITH ATEZOLIZUMAB PRESENTED AT ESMO 2018 IMpassion130 reports first positive Phase III study results for a chemotherapy/immunotherapy (ABRAXANE plus
Drafting a Coverage Authorization Request Letter
 Drafting a Coverage Authorization Request Letter The following information is presented for informational purposes only and is not intended to provide reimbursement or legal advice. Laws, regulations,
Drafting a Coverage Authorization Request Letter The following information is presented for informational purposes only and is not intended to provide reimbursement or legal advice. Laws, regulations,
Iroko Pharmaceuticals Gains Additional Patents for ZORVOLEX and TIVORBEX TM
 Iroko Pharmaceuticals Gains Additional Patents for ZORVOLEX and TIVORBEX TM PHILADELPHIA, April 8, 2015 Iroko Pharmaceuticals, LLC, a global specialty pharmaceutical company dedicated to advancing the
Iroko Pharmaceuticals Gains Additional Patents for ZORVOLEX and TIVORBEX TM PHILADELPHIA, April 8, 2015 Iroko Pharmaceuticals, LLC, a global specialty pharmaceutical company dedicated to advancing the
Pharmacotherapy Handbook
 Pharmacotherapy Handbook Eighth Edition Barbara G. Wells, PharmD, HP, FCCP, BCPP Dean and Professor Executive Director, Research Institute of Pharmaceutical Sciences School of Pharmacy, The University
Pharmacotherapy Handbook Eighth Edition Barbara G. Wells, PharmD, HP, FCCP, BCPP Dean and Professor Executive Director, Research Institute of Pharmaceutical Sciences School of Pharmacy, The University
Crohn's disease CAUSES COURSE OF CROHN'S DISEASE TREATMENT. Sulfasalazine
 Crohn's disease Crohn's disease is an inflammatory condition of the digestive tract that affects children and adults. Common features of Crohn's disease include mouth sores, diarrhea, abdominal pain, weight
Crohn's disease Crohn's disease is an inflammatory condition of the digestive tract that affects children and adults. Common features of Crohn's disease include mouth sores, diarrhea, abdominal pain, weight
(212) Investors Contact: Ryan Crowe (212)
 For immediate release: February 5, 2014 Media Contact: Sally Beatty (212) 733-6566 Investors Contact: Ryan Crowe (212) 733-8160 Pfizer And Merck To Collaborate On Innovative Anti-Cancer Combination Studies
For immediate release: February 5, 2014 Media Contact: Sally Beatty (212) 733-6566 Investors Contact: Ryan Crowe (212) 733-8160 Pfizer And Merck To Collaborate On Innovative Anti-Cancer Combination Studies
Media Contacts: Amy Rose Investor Contact: Graeme Bell (908) (908)
 News Release FOR IMMEDIATE RELEASE Media Contacts: Amy Rose Investor Contact: Graeme Bell (908) 423-6537 (908) 423-5185 Tracy Ogden (267) 305-0960 FDA Approves Once-Daily JANUVIA, the First and Only DPP-4
News Release FOR IMMEDIATE RELEASE Media Contacts: Amy Rose Investor Contact: Graeme Bell (908) 423-6537 (908) 423-5185 Tracy Ogden (267) 305-0960 FDA Approves Once-Daily JANUVIA, the First and Only DPP-4
Patient Name Date of Birth Age. Other phone ( ) . Other
 GASTROINTESTINAL & MINIMALLY INVASIVE SURGERY HEALTH HISTORY QUESTIONNAIRE Date Patient Name _ Date of Birth Age Daytime phone ( ) Other phone ( ) Email How did you hear about us? My doctor Yellow pages
GASTROINTESTINAL & MINIMALLY INVASIVE SURGERY HEALTH HISTORY QUESTIONNAIRE Date Patient Name _ Date of Birth Age Daytime phone ( ) Other phone ( ) Email How did you hear about us? My doctor Yellow pages
Understanding Your Benefits and Risks
 IBD Treatment Options Understanding Your Benefits and Risks benefit risk assessment tool Understand your options. Improve your health... and life. Inflammatory bowel disease (IBD) is two, separate disorders
IBD Treatment Options Understanding Your Benefits and Risks benefit risk assessment tool Understand your options. Improve your health... and life. Inflammatory bowel disease (IBD) is two, separate disorders
- Amendment accelerates anticipated PROSPER top-line results by two years -
 Pfizer Contacts: For Media Dean Mastrojohn (212) 733-6944 dean.mastrojohn@pfizer.com For Investors Ryan Crowe (212) 733-8160 ryan.crowe@pfizer.com Astellas Contact: For Media Tyler Marciniak (847) 736-7145
Pfizer Contacts: For Media Dean Mastrojohn (212) 733-6944 dean.mastrojohn@pfizer.com For Investors Ryan Crowe (212) 733-8160 ryan.crowe@pfizer.com Astellas Contact: For Media Tyler Marciniak (847) 736-7145
For topical use only. Not for oral, ophthalmic, or intravaginal use.
 DESOXIMETASONE Ointment USP, 0.25% For topical use only. Not for oral, ophthalmic, or intravaginal use. Rx only DESCRIPTION Desoximetasone ointment USP, 0.25% contains the active synthetic corticosteroid
DESOXIMETASONE Ointment USP, 0.25% For topical use only. Not for oral, ophthalmic, or intravaginal use. Rx only DESCRIPTION Desoximetasone ointment USP, 0.25% contains the active synthetic corticosteroid
The National Association of Crohn s and Colitis of Trinidad and Tobago CROHN S DISEASE AND ULCERATIVE COLITIS GENERAL PATIENT INFORMATION
 The National Association of Crohn s and Colitis of Trinidad and Tobago CROHN S DISEASE AND ULCERATIVE COLITIS GENERAL PATIENT INFORMATION You are reading this pamphlet because you or someone you known
The National Association of Crohn s and Colitis of Trinidad and Tobago CROHN S DISEASE AND ULCERATIVE COLITIS GENERAL PATIENT INFORMATION You are reading this pamphlet because you or someone you known
Core Safety Profile. Pharmaceutical form(s)/strength: Dry Powder Inhaler, Nebuliser Suspension, pressurised Metered Dose Inhaler (pmdi)
 Core Safety Profile Active substance: Budesonide Pharmaceutical form(s)/strength: Dry Powder Inhaler, Nebuliser Suspension, pressurised Metered Dose Inhaler (pmdi) P - RMS: DK/H/PSUR/0041/001 Date of FAR:
Core Safety Profile Active substance: Budesonide Pharmaceutical form(s)/strength: Dry Powder Inhaler, Nebuliser Suspension, pressurised Metered Dose Inhaler (pmdi) P - RMS: DK/H/PSUR/0041/001 Date of FAR:
