MEDICAID QUANTITY LIMIT DRUG LIST
|
|
|
- Eric Clark
- 5 years ago
- Views:
Transcription
1 MEDICAID QUANTITY LIMIT DRUG LIST PH51-R
2 Brand Name Generic Name Dosage Form Tier Quantity Limit Details Cambia Diclofenac Potassium PACK Tier 2 QL: 9 per 30 days Fentanyl (12 Mcg/Hr, 25 Mcg/ Hr, 50 Mcg/Hr, 75 Mcg/Hr, 100 Mcg/Hr) Fentanyl PATCH TD72 Tier 1 QL: 15 per 30 days; Cumulative Opioid Nucynta ER Tapentadol TB12 Tier 2 QL: 120 per 30 days; Cumulative Opioid Nucynta Er Tapentadol TB12 Tier 2 QL: 60 per 30 days; Cumulative Opioid Nucynta Tapentadol TABLET Tier 2 QL: 60 per 30 days; Cumulative Opioid Buprenorphine Buprenorphine PATCH TDWK Tier 2 QL: 4 per 28 days; Cumulative Opioid Butrans Buprenorphine PATCH TDWK Tier 2 QL: 4 per 28 days; Cumulative Opioid Buprenorphine Buprenorphine TAB SUBL Tier 1 QL: 90 per 30 days; Cumulative Opioid Suboxone (12 Mg - 3 Mg) Buprenorphine / FILM Tier 2 QL: 60 per 30 days; Cumulative Opioid Naloxone Suboxone (2 Mg Mg, 4 Mg Buprenorphine / FILM Tier 2 QL: 90 per 30 days; Cumulative Opioid - 1 Mg, 8 Mg - 2 Mg) Naloxone Buprenorphine-Naloxone Buprenorphine / TAB SUBL Tier 1 QL: 90 per 30 days; Cumulative Opioid Naloxone Narcan Naloxone SPRAY Tier 2 QL: 2 per 365 days Azasite Azithromycin SOLN Tier 2 QL: 5 per 30 days Almotriptan Malate Almotriptan Malate TABLET Tier 1
3 Eletriptan HBR Eletriptan HBR TABLET Tier 1 Frovatriptan Succinate Frovatriptan Succinate TABLET Tier 1 Naratriptan Naratriptan TABLET Tier 1 Rizatriptan Rizatriptan Benzoate TABLET Tier 1 Rizatriptan Rizatriptan Benzoate TBDP Tier 1 Sumatriptan Sumatriptan SPRAY Tier 1 Onzetra Xsail Sumatriptan Succinate AER POW BA Tier 2 Sumatriptan Succinate Sumatriptan Succinate CARTRIDGE Tier 1 Imitrex (6 Mg) Sumatriptan Succinate PEN INJCTR Tier 2 Sumatriptan Succinate Sumatriptan Succinate PEN INJCTR Tier 1 Sumatriptan Succinate Sumatriptan Succinate SOCT Tier 1 Sumatriptan Succinate Sumatriptan Succinate SOLN Tier 1 Sumatriptan Succinate Sumatriptan Succinate SYRINGE Tier 1 Sumatriptan Succinate Sumatriptan Succinate TABLET Tier 1 Zomig Zolmitriptan SOLN Tier 2 Zomig Zolmitriptan SPRAY Tier 2 QL: 16 per 30 days, ST (total for all triptans
4 Zolmitriptan ODT Zolmitriptan TAB RAPDIS Tier 1 Zolmitriptan Zolmitriptan TABLET Tier 1 Tagrisso Osimertinib Mesylate TABLET Tier 2 QL: 30 per 30 days Neupro Rotigotine PATCH TD24 Tier 2 QL: 30 per 30 days Neupro Rotigotine PT24 Tier 2 QL: 30 per 30 days Vraylar Cariprazine CAPSULE Tier 2 QL: 30 per 30 days, ST Invokamet Canagliflozin/Metformin TABLET Tier 2 QL: 60 per 30 days Xigduo Xr (5 Mg Mg) Dapagliflozin/Metformin TAB BP 24H Tier 2 QL: 60 per 30 days Xigduo Xr (5 Mg Mg, 10 Dapagliflozin/Metformin Mg Mg, 10 Mg Mg) TAB BP 24H Tier 2 QL: 30 per 30 days Synjardy XR Empagliflozin/Metformin TAB BP 24H Tier 2 QL: 30 per 30 days Synjardy Empagliflozin/Metformin TABLET Tier 2 QL: 60 per 30 days Bydureon Pen Exenatide Microspheres PEN INJCTR Tier 2 QL: 4 per 28 days Bydureon Exenatide Microspheres SRER Tier 2 QL: 4 per 28 days Xarelto (10 Mg) Rivaroxaban TABLET Tier 2 QL: 94 per 365 days Clonidine Clonidine PATCH TDWK Tier 1 QL: 4 per 28 days Nitro-Dur (0.3 Mg/Hr, 0.8 Mg/ Hr) Nitroglycerin PATCH TD24 Tier 2 QL: 30 per 30 days Nitroglycerin Patch Nitroglycerin PATCH TD24 Tier 1 QL: 30 per 30 days Minitran Nitroglycerin PT24 Tier 1 QL: 30 per 30 days Wide Seal Diaphragm Diaphragms, Wide Seal DIAPHRAGM $0 Copay Durable Medical Equipment; QL: 3 per 365 days Oxytrol Oxybutynin PATCH TDSW Tier 2 QL: 8 per 28 days Cialis (2.5 Mg, 5 Mg) Tadalafil TABLET Tier 2 QL: 30 per 30 days Caverject Alprostadil KIT Tier 2 QL: 6 per 30 days
5 Edex Alprostadil KIT Tier 2 QL: 6 per 30 days Caverject Alprostadil VIAL Tier 2 QL: 6 per 30 days Stendra Avanafil TABLET Tier 2 QL: 8 per 30 days Cialis (10 Mg, 20 Mg) Tadalafil TABLET Tier 2 QL: 8 per 30 days Staxyn Vardenafil HCl TAB RAPDIS Tier 2 QL: 8 per 30 days Levitra Vardenafil TABLET Tier 2 QL: 8 per 30 days Androderm Testosterone PATCH TD24 Tier 2 QL: 30 per 30 days Testosterone Testosterone SOL MD PMP Tier 1 QL: 180 per 30 days Alora Estradiol PATCH TDSW Tier 2 QL: 8 per 28 days Estradiol Estradiol PATCH TDSW Tier 1 QL: 8 per 28 days Minivelle Estradiol PATCH TDSW Tier 2 QL: 8 per 28 days Climara Estradiol PATCH TDWK Tier 2 QL: 4 per 28 days Estradiol Estradiol PATCH TDWK Tier 1 QL: 4 per 28 days Menostar Estradiol PATCH TDWK Tier 2 QL: 4 per 28 days Estring Estradiol VAG RING Tier 2 QL: 1 per 90 days Femring Estradiol Acetate VAG RING Tier 2 QL: 1 per 90 days Climara Pro Estradiol/Levonorgestrel PATCH TDWK Tier 2 QL: 4 per 28 days Combipatch Estradiol/Norethindrone Acet PTTW Tier 2 QL: 8 per 28 days Nuvaring Etonogestrel/Ethinyl Estradiol VAG RING $0 Copay QL: 1 per 28 days Ortho Evra Norelgestromin/Ethin. Estradiol PATCH TDWK Tier 2 QL: 3 per 28 days Xulane Norelgestromin/Ethin. Estradiol PATCH TDWK $0 Copay QL: 3 per 28 days Kyleena Levonorgestrel IUD $0 Copay Liletta Levonorgestrel IUD $0 Copay Mirena Levonorgestrel IUD $0 Copay
6 Skyla Levonorgestrel IUD $0 Copay Paragard T 380-A Copper IUD $0 Copay Alendronate Sodium Alendronate Sodium SOLUTION Tier 1 QL: 300 per 28 days Alendronate Sodium (35 Mg, 70 Mg) Alendronate Sodium TABLET Tier 1 QL: 4 per 28 days Fosamax Plus D Alendronate Sodium/ Vitamin D3 TABLET Tier 2 QL: 4 per 28 days Boniva Ibandronate Sodium SOLN Tier 3 Medical Injectable QL: 3 per 90 days Ibandronate Sodium Ibandronate Sodium SYRINGE Tier 3 Medical Injectable QL: 3 per 90 days Ibandronate Sodium Ibandronate Sodium TABLET Tier 1 QL: 1 per 28 days Risedronate Sodium (150 Mg) Risedronate Sodium TABLET Tier 1 QL: 1 per 28 days Risedronate Sodium (35 Mg) Risedronate Sodium TABLET Tier 1 QL: 4 per 28 days Restasis Cyclosporine EMUL Tier 2 QL: 60 per 30 days Lacrisert Hydroxypropyl Cellulose INSERT Tier 2 Specialty QL: 60 per 30 days Tobramycin-Dexamethasone Tobramycin/ Dexamethasone DROPS SUSP Tier 1 QL: 20 per 30 days Olopatadine Olopatadine DROPS Tier 1 QL: 5 per 30 days Olopatadine Olopatadine DROPS Tier 1 QL: 5 per 31 days Betaxolol Betaxolol DROPS Tier 1 QL: 5 per 30 days Betoptic-S Betaxolol SUSP Tier 2 QL: 10 per 30 days Alphagan P (0.1%) Brimonidine Tartrate DROPS Tier 2 QL: 10 per 30 days Brimonidine Tartrate Brimonidine Tartrate DROPS Tier 1 QL: 10 per 30 days Combigan Brimonidine Tartrate/ Timolol DROPS Tier 2 QL: 10 per 30 days Azopt Brinzolamide DROPS SUSP Tier 2 QL: 10 per 30 days Carteolol Carteolol DROPS Tier 1 QL: 5 per 30 days Dorzolamide-Timolol Dorzolamide Hcl/Timolol Maleat SOLN Tier 1 QL: 10 per 30 days Cosopt PF Dorzolamide/Timolol/PF SOLN Tier 2 QL: 60 per 30 days
7 Levobunolol Levobunolol SOLN Tier 1 QL: 5 per 30 days Pilocarpine Pilocarpine DROPS Tier 1 QL: 15 per 30 days Pilocarpine Pilocarpine SOLN Tier 1 QL: 15 per 30 days Betimol Timolol SOLN Tier 2 QL: 10 per 30 days Timolol Maleate (0.5%) Timolol Maleate DROPS Tier 1 QL: 5 per 30 days Timolol Maleate Timolol Maleate SOL-GEL Tier 1 QL: 5 per 30 days Timolol Male Sol 0.5% Timolol Maleate SOLN Tier 1 QL: 5 per 30 days Diclofenac Sodium (0.1%) Diclofenac Sodium DROPS Tier 1 QL: 5 per 30 days Lumigan Bimatoprost DROPS Tier 2 QL: 5 per 60 days Latanoprost Latanoprost DROPS Tier 1 QL: 2.5 per 30 days Zioptan Tafluprost/PF SOLN Tier 2 QL: 30 per 30 days Travatan Z Travoprost DROPS Tier 2 QL: 5 per 60 days Azelastine Azelastine SPRAY/PUMP Tier 1 QL: 60 per 30 days Qvar Beclomethasone Dipropionate AER W/ADAP Tier 2 QL: 17.4 per 30 days Qvar Beclomethasone Dipropionate AERS Tier 2 QL: 17.4 per 30 days Beconase AQ Beclomethasone Dipropionate SPRAY Tier 2 QL: 50 per 30 days Pulmicort Flexhaler Budesonide AER POW BA Tier 2 QL: 2 per 30 days Alvesco Ciclesonide HFA AER AD Tier 2 QL: 12.2 per 30 days Flunisolide Flunisolide SPRAY Tier 1 QL: 50 per 30 days Arnuity Ellipta Fluticasone Furoate BLST W/DEV Tier 2 QL: 30 per 30 days Flovent HFA (44 Mcg) Fluticasone Propionate AER W/ADAP Tier 2 QL: 21.2 per 30 days Flovent Diskus Fluticasone Propionate BLST W/DEV Tier 2 QL: 120 per 30 days Asmanex Twisthaler 14 Metered Doses Mometasone Furoate AEPB Tier 2 QL: 1 per 30 days Asmanex (7, 14, 30, 60) Mometasone Furoate AER POW BA Tier 2 QL: 1 per 30 days Asmanex (120) Mometasone Furoate AER POW BA Tier 2 QL: 1 per 60 days Asmanex HFA Mometasone Furoate HFA AER AD Tier 2 QL: 13 per 30 days Mometasone Furoate Mometasone Furoate SPRAY/PUMP Tier 1 QL: 34 per 30 days Triamcinolone Acetonide Triamcinolone Acetonide SPRAY Tier 1 QL: 33 per 30 days Seebri Neohaler Glycopyrrolate CAP W/DEV Tier 2 QL: 60 per 30 days
8 Atrovent HFA Ipratropium Bromide HFA AER AD Tier 2 QL: 25.8 per 30 days Incruse Ellipta Umeclidinium Bromide BLST W/DEV Tier 2 QL: 30 per 30 days Proair Respiclick Albuterol Sulfate AER POW BA Tier 2 QL: 2 per 30 days Proair HFA Albuterol Sulfate AERS Tier 2 QL: 17 per 30 days Ventolin HFA Albuterol Sulfate AERS Tier 2 QL: 36 per 30 days Proventil HFA Albuterol Sulfate HFA AER AD Tier 2 QL: 13.4 per 30 days Ventolin HFA Albuterol Sulfate HFA AER AD Tier 2 QL: 36 per 30 days Arcapta Neohaler Indacaterol Maleate CAP W/DEV Tier 2 QL: 30 per 30 days Xopenex HFA Levalbuterol Tartrate HFA AER AD Tier 2 QL: 30 per 30 days Striverdi Respimat Olodaterol MIST INHAL Tier 2 QL: 4 per 30 days Serevent Diskus Salmeterol Xinafoate AEPB Tier 2 QL: 60 per 30 days Serevent Diskus Salmeterol Xinafoate BLST W/DEV Tier 2 QL: 60 per 30 days Symbicort Budesonide/Formoterol Fumarate HFA AER AD Tier 2 QL: 10.2 per 30 days Advair Diskus Fluticasone/Salmeterol AEPB Tier 2 QL: 60 per 30 days, ST Advair Diskus Fluticasone/Salmeterol BLST W/DEV Tier 2 QL: 60 per 30 days, ST Advair HFA Fluticasone/Salmeterol HFA AER AD Tier 2 QL: 12 per 30 days, ST Breo Ellipta Fluticasone/Vilanterol BLST W/DEV Tier 2 QL: 60 per 30 days Utibron Neohaler Indacaterol/Glycopyrrolate CAP W/DEV Tier 2 QL: 60 per 30 days Dulera Mometasone/Formoterol HFA AER AD Tier 2 QL: 13 per 30 days Combivent Respimat Ipratropium/Albuterol Sulfate MIST INHAL Tier 2 QL: 8 per 30 days Stiolto Respimat Tiotropium Br/Olodaterol MIST INHAL Tier 2 QL: 4 per 30 days
University System of Georgia Prior Authorization, Step Therapy and Quantity Limit List (Updated 1/1/2016)
 University System of Georgia, Step Therapy and Quantity Limit List (Updated 1/1/2016) (PA) Your doctor will need to obtain a prior authorization for the drugs listed below, before your prescription drug
University System of Georgia, Step Therapy and Quantity Limit List (Updated 1/1/2016) (PA) Your doctor will need to obtain a prior authorization for the drugs listed below, before your prescription drug
Select Inhaled Respiratory Agents
 Applies to all products administered or underwritten by Blue Cross and Blue Shield of Louisiana and its subsidiary, HMO Louisiana, Inc.(collectively referred to as the Company ), unless otherwise provided
Applies to all products administered or underwritten by Blue Cross and Blue Shield of Louisiana and its subsidiary, HMO Louisiana, Inc.(collectively referred to as the Company ), unless otherwise provided
TRELEGY ELLIPTA (fluticasone-umeclidinium-vilanterol) aerosol powder
 TRELEGY ELLIPTA (fluticasone-umeclidinium-vilanterol) aerosol powder Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific
TRELEGY ELLIPTA (fluticasone-umeclidinium-vilanterol) aerosol powder Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific
The Medical Letter. on Drugs and Therapeutics
 The Medical Letter publications are protected by US and international copyright laws. Forwarding, copying or any other distribution of this material is strictly prohibited. For further information call:
The Medical Letter publications are protected by US and international copyright laws. Forwarding, copying or any other distribution of this material is strictly prohibited. For further information call:
Diagnosis and Management of Asthma
 Supporting Evidence: Diagnosis and Management of Asthma The subdivision of this section is: Appendix B Tables Copyright 2016 by 1 Eleventh Edition/December 2016 Appendix B Asthma Summary Tables Class:
Supporting Evidence: Diagnosis and Management of Asthma The subdivision of this section is: Appendix B Tables Copyright 2016 by 1 Eleventh Edition/December 2016 Appendix B Asthma Summary Tables Class:
2014 Quantity Limits (QL) Criteria
 2014 Quantity Limits (QL) Criteria Certain drugs covered through your EmblemHealth Medicare HMO/PPO Medicare Plan are covered for only a limited quantity. We do this to ensure compliance with the US Food
2014 Quantity Limits (QL) Criteria Certain drugs covered through your EmblemHealth Medicare HMO/PPO Medicare Plan are covered for only a limited quantity. We do this to ensure compliance with the US Food
A Visual Approach to Simplifying Respiratory Drug Regimens
 A Visual Approach to Simplifying Respiratory Drug Regimens Stephanie Cheng, PharmD, MPH, BCGP 3 Main Categories Inhaled Respiratory Drugs Binds to beta-2 receptors Relaxation of smooth muscles in the lung
A Visual Approach to Simplifying Respiratory Drug Regimens Stephanie Cheng, PharmD, MPH, BCGP 3 Main Categories Inhaled Respiratory Drugs Binds to beta-2 receptors Relaxation of smooth muscles in the lung
Pain Oral-Intranasal Fentanyl (Abstral, Actiq, Fentora, Lazanda, Onsolis, Subsys)
 Pennsylvania Employees Benefit Trust Fund (PEBTF) and n- Medicare Eligible Retired Employees Health Program (REHP), Step Therapy and Quantity Limit List Your doctor needs to get prior authorization for
Pennsylvania Employees Benefit Trust Fund (PEBTF) and n- Medicare Eligible Retired Employees Health Program (REHP), Step Therapy and Quantity Limit List Your doctor needs to get prior authorization for
REVISED RESPIRATORY MEDICATION USE QUESTIONNAIRE
 REVISED RESPIRATORY MEDICATION USE QUESTIONNAIRE ID NUMBER: 0a) Date of Collection / / 0b) Staff Code Instructions: This form should be completed during the participant s clinic visit. 1) Are you regularly
REVISED RESPIRATORY MEDICATION USE QUESTIONNAIRE ID NUMBER: 0a) Date of Collection / / 0b) Staff Code Instructions: This form should be completed during the participant s clinic visit. 1) Are you regularly
A Visual Approach to Simplifying Respiratory Drug Regimens
 A Visual Approach to Simplifying Respiratory Drug Regimens Stephanie Cheng, PharmD, MPH, BCGP October 23, 2017 Learning Objectives Be able to list at least 3 major adverse effects of inhaled medications
A Visual Approach to Simplifying Respiratory Drug Regimens Stephanie Cheng, PharmD, MPH, BCGP October 23, 2017 Learning Objectives Be able to list at least 3 major adverse effects of inhaled medications
A Visual Approach to Simplifying Respiratory Drug Regimens
 Adverse Effects of Inhaled Medications A Visual Approach to Simplifying Respiratory Drug Regimens Stephanie Cheng, PharmD, MPH, BCGP June 28, 2017 Drug Category Beta 2 agonists antagonists Adverse Effects
Adverse Effects of Inhaled Medications A Visual Approach to Simplifying Respiratory Drug Regimens Stephanie Cheng, PharmD, MPH, BCGP June 28, 2017 Drug Category Beta 2 agonists antagonists Adverse Effects
Inhaled bronchodilators relax constricted airways and treat the noisy part of asthma: coughing, wheezing, choking and shortness of breath.
 Inhaled bronchodilators relax constricted airways and treat the noisy part of asthma: coughing, wheezing, choking and shortness of breath. AccuNeb inhalation 0.021% solution: 0.63mg/3mL 3-4 times solution
Inhaled bronchodilators relax constricted airways and treat the noisy part of asthma: coughing, wheezing, choking and shortness of breath. AccuNeb inhalation 0.021% solution: 0.63mg/3mL 3-4 times solution
Ferris State University College of Pharmacy MPA CE Symposium 2016 Paul Thill, PharmD, BCPS
 Ferris State University College of Pharmacy MPA CE Symposium 2016 Paul Thill, PharmD, BCPS Objectives Categorize the new asthma and COPD inhalers in to existing or newly created categories Discuss the
Ferris State University College of Pharmacy MPA CE Symposium 2016 Paul Thill, PharmD, BCPS Objectives Categorize the new asthma and COPD inhalers in to existing or newly created categories Discuss the
AIRDUO RESPICLICK (fluticasone-salmeterol) aerosol DULERA (mometasone furoate and formoterol fumarate dihydrate) aerosol
 DULERA (mometasone furoate and formoterol fumarate dihydrate) aerosol Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific
DULERA (mometasone furoate and formoterol fumarate dihydrate) aerosol Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific
Data Class: Internal. 1 inhaler (30 blisters OR 14 blisters institutional pack) per presciption
 To help make the use of prescription drugs safer and more affordable, our plan is now using a Drug Quantity Management program. That is, for certain medications, you can receive an amount to last you a
To help make the use of prescription drugs safer and more affordable, our plan is now using a Drug Quantity Management program. That is, for certain medications, you can receive an amount to last you a
TEST ANTICONVULSANT THERAPY. Products Affected. Step 2: Network Health Insurance Corporation NetworkCares Step Therapy Criteria Last Updated 11/2018
 TEST Network Health Insurance Corporation NetworkCares Step Therapy Last Updated 11/2018 ANTICONVULSANT THERAPY Aptiom Banzel Briviact Celontin Dilantin 30 Mg Capsule Equetro Fycompa 0.5 Mg/ml Oral Susp
TEST Network Health Insurance Corporation NetworkCares Step Therapy Last Updated 11/2018 ANTICONVULSANT THERAPY Aptiom Banzel Briviact Celontin Dilantin 30 Mg Capsule Equetro Fycompa 0.5 Mg/ml Oral Susp
Drug Effectiveness Review Project Summary Report
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Inhaled Corticosteroids Drug Class Prior Authorization Protocol
 Inhaled Corticosteroids Drug Class Prior Authorization Protocol Line of Business: Medicaid P&T Approval Date: February 21, 2018 Effective Date: April 1, 2018 This policy has been developed through review
Inhaled Corticosteroids Drug Class Prior Authorization Protocol Line of Business: Medicaid P&T Approval Date: February 21, 2018 Effective Date: April 1, 2018 This policy has been developed through review
ALLERGIC RHINITIS-NASAL
 ALLERGIC RHINITIS-NASAL FLUNISOLIDE Patient needs to have paid claims for any one of the following Step 1 drugs: NasaCort OTC, fluticasone Rx, fluticasone OTC, Budesonide OTC. Prior to filling the Step
ALLERGIC RHINITIS-NASAL FLUNISOLIDE Patient needs to have paid claims for any one of the following Step 1 drugs: NasaCort OTC, fluticasone Rx, fluticasone OTC, Budesonide OTC. Prior to filling the Step
COPD Medicine. No one ever showed me how to use this. Wendy Happel; RRT, COPD Educator Krystal Fedoris; RRT-NPS, BA, COPD Educator
 Medicine. No one ever showed me how to use this. Wendy Happel; RRT, Educator Krystal Fedoris; RRT-NPS, BA, Educator 1 Taking prescriptions correctly Taking prescriptions can be a challenge Busy schedules
Medicine. No one ever showed me how to use this. Wendy Happel; RRT, Educator Krystal Fedoris; RRT-NPS, BA, Educator 1 Taking prescriptions correctly Taking prescriptions can be a challenge Busy schedules
First to Market or 505 (b)2 CMC Considerations IPAC-RS/UF Orlando Inhalation Conference Orlando, Florida
 First to Market or 505 (b)2 CMC Considerations IPAC-RS/UF Orlando Inhalation Conference Orlando, Florida Prasad Peri, Ph.D., Branch Chief, ONDQA, FDA March 19, 2014 1 Topics for discussion Introduction
First to Market or 505 (b)2 CMC Considerations IPAC-RS/UF Orlando Inhalation Conference Orlando, Florida Prasad Peri, Ph.D., Branch Chief, ONDQA, FDA March 19, 2014 1 Topics for discussion Introduction
Inhaled Corticosteroids Drug Class Prior Authorization Protocol
 Inhaled Corticosteroids Drug Class Prior Authorization Protocol Line of Business: Medi-Cal P&T Approval Date: February 21, 2018 Effective Date: April 1, 2018 This policy has been developed through review
Inhaled Corticosteroids Drug Class Prior Authorization Protocol Line of Business: Medi-Cal P&T Approval Date: February 21, 2018 Effective Date: April 1, 2018 This policy has been developed through review
Drug Class Monograph
 Drug Class Monograph Class: Inhaled Corticosteroids Drugs: Aerospan (flunisolide), Advair Diskus, Advair HFA (fluticasone/salmeterol), Alvesco (ciclesonide), Arnuity Ellipta (fluticasone furoate), Asmanex
Drug Class Monograph Class: Inhaled Corticosteroids Drugs: Aerospan (flunisolide), Advair Diskus, Advair HFA (fluticasone/salmeterol), Alvesco (ciclesonide), Arnuity Ellipta (fluticasone furoate), Asmanex
Pequot Health Care Smart Quantity Program*
 Pequot Health Care 1 Annie George Drive Mashantucket, CT 06338 Phone: 1-888-779-6638 Fax: 1-860-396-6494 Pequot Health Care Smart Quantity Program* Updated January 2018 *Quantity Program limits apply to
Pequot Health Care 1 Annie George Drive Mashantucket, CT 06338 Phone: 1-888-779-6638 Fax: 1-860-396-6494 Pequot Health Care Smart Quantity Program* Updated January 2018 *Quantity Program limits apply to
MEDICAL COVERAGE GUIDELINES ORIGINAL EFFECTIVE DATE: 07/05/18 SECTION: DRUGS LAST REVIEW DATE: LAST CRITERIA REVISION DATE: ARCHIVE DATE:
 CINQAIR (reslizumab) Non-Discrimination Statement and Multi-Language Interpreter Services information are located at the end of this document. Coverage for services, procedures, medical devices and drugs
CINQAIR (reslizumab) Non-Discrimination Statement and Multi-Language Interpreter Services information are located at the end of this document. Coverage for services, procedures, medical devices and drugs
MDI Bonanza. Dwayne Griffin, DO
 MDI Bonanza Dwayne Griffin, DO Bonanza 3. A MDI costing $200 - $500 per month SISYPHUS MDI Griffin Mountain Evolution of Deliver Systems for COPD in the US 2003 2009 2011 2013 2004 2012 2014 Prescribing
MDI Bonanza Dwayne Griffin, DO Bonanza 3. A MDI costing $200 - $500 per month SISYPHUS MDI Griffin Mountain Evolution of Deliver Systems for COPD in the US 2003 2009 2011 2013 2004 2012 2014 Prescribing
ATYPICAL ANTIPSYCHOTICS
 Step Therapy CareOregon 2018 Last Updated: 07/27/2018 ATYPICAL ANTIPSYCHOTICS Fanapt Fanapt Titration Pack Paliperidone Er Vraylar The following criteria applies to members who newly start on the drug:
Step Therapy CareOregon 2018 Last Updated: 07/27/2018 ATYPICAL ANTIPSYCHOTICS Fanapt Fanapt Titration Pack Paliperidone Er Vraylar The following criteria applies to members who newly start on the drug:
Impact of a Comprehensive COPD Therapeutic Interchange Program on 30-Day Readmission Rates in Hospitalized Patients
 Impact of a Comprehensive COPD Therapeutic Interchange Program on 30-Day Readmission Rates in Hospitalized Patients Maren A. McGurran, PharmD, BCPS; Lisa M. Richter, PharmD, BCPS, BCCCP; Nathan D. Leedahl,
Impact of a Comprehensive COPD Therapeutic Interchange Program on 30-Day Readmission Rates in Hospitalized Patients Maren A. McGurran, PharmD, BCPS; Lisa M. Richter, PharmD, BCPS, BCCCP; Nathan D. Leedahl,
WVCH Formulary Additions Effective 01/01/2016 Name Strength Dosage Form Route Formulary Restrictions
 WVCH Formulary Additions Effective 01/01/2016 Name Strength Dosage Form Route Formulary Restrictions ANORO ELLIPTA 62.5-25MCG BLST W/DEV INHALATION ARCAPTA NEOHALER 75 MCG CAP W/DEV INHALATION CALCIPOTRIENE
WVCH Formulary Additions Effective 01/01/2016 Name Strength Dosage Form Route Formulary Restrictions ANORO ELLIPTA 62.5-25MCG BLST W/DEV INHALATION ARCAPTA NEOHALER 75 MCG CAP W/DEV INHALATION CALCIPOTRIENE
Correct Use of Inhaler Devices
 PL Detail-Document #300206 This PL Detail-Document gives subscribers additional insight related to the Recommendations published in PHARMACIST S LETTER / PRESCRIBER S LETTER February 2014 Correct Use of
PL Detail-Document #300206 This PL Detail-Document gives subscribers additional insight related to the Recommendations published in PHARMACIST S LETTER / PRESCRIBER S LETTER February 2014 Correct Use of
FASENRA (benralizumab)
 FASENRA (benralizumab) Non-Discrimination Statement and Multi-Language Interpreter Services information are located at the end of this document. Coverage for services, procedures, medical devices and drugs
FASENRA (benralizumab) Non-Discrimination Statement and Multi-Language Interpreter Services information are located at the end of this document. Coverage for services, procedures, medical devices and drugs
Diabetes Fortamet (metformin ER), Glumetza (metformin ER) generic of Glucophage XR (metformin ER) preferred
 Pennsylvania Employees Benefit Trust Fund (PEBTF) and n-medicare Eligible Retired Employees Health Program (REHP), Step Therapy and Quantity Limit List Your doctor needs to get prior authorization for
Pennsylvania Employees Benefit Trust Fund (PEBTF) and n-medicare Eligible Retired Employees Health Program (REHP), Step Therapy and Quantity Limit List Your doctor needs to get prior authorization for
Step Therapy Medications
 Step Therapy Medications Step Therapy (ST PA ) is an automated form of prior authorization. It encourages the use of therapies that should be tried first, before other treatments are covered, based on
Step Therapy Medications Step Therapy (ST PA ) is an automated form of prior authorization. It encourages the use of therapies that should be tried first, before other treatments are covered, based on
ANTICONVULSANT THERAPY
 Network Health Insurance Corporation NetworkCares Step Therapy Last Updated: 7/2017 ANTICONVULSANT THERAPY Aptiom 200 mg tablet Aptiom 400 mg tablet Aptiom 600 mg tablet Aptiom 800 mg tablet Banzel 200
Network Health Insurance Corporation NetworkCares Step Therapy Last Updated: 7/2017 ANTICONVULSANT THERAPY Aptiom 200 mg tablet Aptiom 400 mg tablet Aptiom 600 mg tablet Aptiom 800 mg tablet Banzel 200
Mercy Care Plan. Acyclovir Ointment. Products Affected. acyclovir ointment 5 % external Details. Criteria. Requires use of oral Acyclovir
 Acyclovir Ointment Mercy Care Plan acyclovir ointment 5 % external Requires use of oral Acyclovir 1 Adcirca ADCIRCA TABLET 20 MG ORAL Requires use of Sildenafil 2 Albenza ALBENZA TABLET 200 MG ORAL Requires
Acyclovir Ointment Mercy Care Plan acyclovir ointment 5 % external Requires use of oral Acyclovir 1 Adcirca ADCIRCA TABLET 20 MG ORAL Requires use of Sildenafil 2 Albenza ALBENZA TABLET 200 MG ORAL Requires
MEDICATION COVERAGE POLICY PHARMACY AND THERAPEUTICS ADVISORY COMMITTEE POLICY: Asthma/COPD P&T DATE 12/14/2017 CLASS:
 MEDICATION COVERAGE POLICY PHARMACY AND THERAPEUTICS ADVISORY COMMITTEE POLICY: Asthma/COPD P&T DATE 12/14/2017 CLASS: LOB: Respiratory Disorders Medi-Cal REVIEW HISTORY (MONTH/YEAR) 12/17,12/16, 5/15,
MEDICATION COVERAGE POLICY PHARMACY AND THERAPEUTICS ADVISORY COMMITTEE POLICY: Asthma/COPD P&T DATE 12/14/2017 CLASS: LOB: Respiratory Disorders Medi-Cal REVIEW HISTORY (MONTH/YEAR) 12/17,12/16, 5/15,
BREEZHALER. Medications available: Onbrez (indacaterol maleate) Seebri (glycopyrronium bromide) Ultibro (glycopyrronium bromide/ (indacaterol maleate)
 Medications available: Onbrez (indacaterol maleate) Seebri (glycopyrronium bromide) Ultibro (glycopyrronium bromide/ (indacaterol maleate) BREEZHALER Please date initial after you have directly observed
Medications available: Onbrez (indacaterol maleate) Seebri (glycopyrronium bromide) Ultibro (glycopyrronium bromide/ (indacaterol maleate) BREEZHALER Please date initial after you have directly observed
QUANTITY LIMIT CRITERIA. BROVANA (arformoterol tartrate) SEREVENT DISKUS (salmeterol) STRIVERDI RESPIMAT (olodaterol)
 Carelirst. +.V Family of health care plans DRUG CLASS COMBINATIONS QUANTITY LIMIT CRITERIA LONG ACTING BETA2-ADRENERGIC AGONIST, ORAL INHALATION BRAND NAME (generic) LONG-ACTING BETA2-ADRENERGIC AGONISTS:
Carelirst. +.V Family of health care plans DRUG CLASS COMBINATIONS QUANTITY LIMIT CRITERIA LONG ACTING BETA2-ADRENERGIC AGONIST, ORAL INHALATION BRAND NAME (generic) LONG-ACTING BETA2-ADRENERGIC AGONISTS:
BREEZHALER. Medications available: Onbrez (indacaterol maleate) Seebri (glycopyrronium bromide) Ultibro (glycopyrronium bromide/ (indacaterol maleate)
 Medications available: Onbrez (indacaterol maleate) Seebri (glycopyrronium bromide) Ultibro (glycopyrronium bromide/ (indacaterol maleate) BREEZHALER Please date initial after you have directly observed
Medications available: Onbrez (indacaterol maleate) Seebri (glycopyrronium bromide) Ultibro (glycopyrronium bromide/ (indacaterol maleate) BREEZHALER Please date initial after you have directly observed
BREEZHALER. Medications available: Onbrez (indacaterol maleate) Seebri (glycopyrronium bromide) Ultibro (glycopyrronium bromide/ (indacaterol maleate)
 Medications available: Onbrez (indacaterol maleate) Seebri (glycopyrronium bromide) Ultibro (glycopyrronium bromide/ (indacaterol maleate) BREEZHALER Please date initial after you have directly observed
Medications available: Onbrez (indacaterol maleate) Seebri (glycopyrronium bromide) Ultibro (glycopyrronium bromide/ (indacaterol maleate) BREEZHALER Please date initial after you have directly observed
Health Partners Medicare Prime 2019 Formulary Changes
 Health Partners Medicare Prime 2019 Formulary Changes Changes occur, for example, because new drugs come on the market, a drug is moved to a different cost-sharing level (tier), or a generic version becomes
Health Partners Medicare Prime 2019 Formulary Changes Changes occur, for example, because new drugs come on the market, a drug is moved to a different cost-sharing level (tier), or a generic version becomes
STRIVERDI RESPIMAT (olodaterol hcl) aerosol
 STRIVERDI RESPIMAT (olodaterol hcl) aerosol Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This Pharmacy
STRIVERDI RESPIMAT (olodaterol hcl) aerosol Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This Pharmacy
COPD Medications Coverage Summary Non-Insured Health Benefits Coverage SABA Bricanyl turbuhaler Yes Yes
 COPD Medications Coverage Summary Drug Non-Insured Health Benefits Coverage SABA Bricanyl turbuhaler Yes Yes Ventolin MDI + generics Yes Yes Ventolin Diskus NO NO Yukon Pharmacare/Chronic Disease Program
COPD Medications Coverage Summary Drug Non-Insured Health Benefits Coverage SABA Bricanyl turbuhaler Yes Yes Ventolin MDI + generics Yes Yes Ventolin Diskus NO NO Yukon Pharmacare/Chronic Disease Program
Asthma/COPD Update with Inhaler Workshop
 Asthma/COPD Update with Inhaler Workshop October 8, 2017 Nathan Samsa, DO, Pharm D, RPh, FACOI None Disclosures Agenda Asthma Updates COPD Updates Inhaler Workshop Medication Acronyms SABA: Short acting
Asthma/COPD Update with Inhaler Workshop October 8, 2017 Nathan Samsa, DO, Pharm D, RPh, FACOI None Disclosures Agenda Asthma Updates COPD Updates Inhaler Workshop Medication Acronyms SABA: Short acting
Quarterly pharmacy formulary change notice
 Quarterly pharmacy formulary change notice Summary of change: The Pharmacy and Therapeutics Committee (P&T) reviewed and approved the formulary changes listed in the table below on March 29, 2016. What
Quarterly pharmacy formulary change notice Summary of change: The Pharmacy and Therapeutics Committee (P&T) reviewed and approved the formulary changes listed in the table below on March 29, 2016. What
reslizumab (Cinqair )
 Applies to all products administered or underwritten by Blue Cross and Blue Shield of Louisiana and its subsidiary, HMO Louisiana, Inc.(collectively referred to as the Company ), unless otherwise provided
Applies to all products administered or underwritten by Blue Cross and Blue Shield of Louisiana and its subsidiary, HMO Louisiana, Inc.(collectively referred to as the Company ), unless otherwise provided
Your prescription benefit updates Formulary Updates - Effective January 1, 2019
 Your prescription benefit updates Formulary Updates - Effective January 1, 2019 Medications are grouped by the conditions they treat. Each medication is placed in a tier that shows the amount you will
Your prescription benefit updates Formulary Updates - Effective January 1, 2019 Medications are grouped by the conditions they treat. Each medication is placed in a tier that shows the amount you will
Blue Cross and Blue Shield of Minnesota GenRx Formulary Updates
 Blue Cross and Blue Shield of Minnesota GenRx Formulary Updates July 2018 TRADE NAME (generic name) or generic name ADVAIR DISKUS (fluticasone-salmeterol aer powder ba 100-50 mcg/dose) Brand Addition ADVAIR
Blue Cross and Blue Shield of Minnesota GenRx Formulary Updates July 2018 TRADE NAME (generic name) or generic name ADVAIR DISKUS (fluticasone-salmeterol aer powder ba 100-50 mcg/dose) Brand Addition ADVAIR
TABLE OF CONTENTS (Click on a link below to view the section.)
 Follow the links below to access the complete formularies for Plans: Health Plan Acne Allergy Allergic Anaphylactic Reaction Allergic Conjunctivitis Allergic Rhinitis Asthma Atopic Dermatitis Behavioral
Follow the links below to access the complete formularies for Plans: Health Plan Acne Allergy Allergic Anaphylactic Reaction Allergic Conjunctivitis Allergic Rhinitis Asthma Atopic Dermatitis Behavioral
APPENDIX 1 Printable point-of-care tables Asthma Action Plan Yellow Zone Formulation Table Region: Europe
 APPENDIX 1 Printable point-of-care tables Asthma Action Plan Yellow Zone Formulation Table Region: Europe Instructions: Print on 8.5 x14 (216 x 279 mm) paper (Legal size) Medication in Green Zone Change
APPENDIX 1 Printable point-of-care tables Asthma Action Plan Yellow Zone Formulation Table Region: Europe Instructions: Print on 8.5 x14 (216 x 279 mm) paper (Legal size) Medication in Green Zone Change
AVMED 4 TIER AND 5 TIER FORMULARY QUANTITY LIMIT TABLE
 Effective 4/1/2018 AVMED 4 TIER AND 5 TIER FORMULARY QUANTITY LIMIT TABLE Generic Name Brand Name Quantity Limit Description Comments ABACAVIR SOLN 20 MG/ML ZIAGEN 900ml every 30 days ABACAVIR TAB 300
Effective 4/1/2018 AVMED 4 TIER AND 5 TIER FORMULARY QUANTITY LIMIT TABLE Generic Name Brand Name Quantity Limit Description Comments ABACAVIR SOLN 20 MG/ML ZIAGEN 900ml every 30 days ABACAVIR TAB 300
Mercy Care ALBENDAZOLE. Products Affected. ALBENZA TABLET 200 MG ORAL Details. Criteria. Refer to PA Guideline for approval criteria
 ALBENDAZOLE Mercy Care ALBENZA TABLET 200 MG ORAL Refer to PA Guideline for approval criteria 1 BRIMONIDINE-TIMOLOL COMBIGAN SOLUTION 0.2-0.5 % OPHTHALMIC Requires use of separate ingredients for at least
ALBENDAZOLE Mercy Care ALBENZA TABLET 200 MG ORAL Refer to PA Guideline for approval criteria 1 BRIMONIDINE-TIMOLOL COMBIGAN SOLUTION 0.2-0.5 % OPHTHALMIC Requires use of separate ingredients for at least
THERAPEUTIC AREA NAME STRENGTH DOSAGE FORM
 Value Based Tier Drugs are selected for the management of Asthma, Diabetes, Hypertension and Hyperlipidemia. These drugs are covered at no charge or at a reduced cost share. Medications are under continual
Value Based Tier Drugs are selected for the management of Asthma, Diabetes, Hypertension and Hyperlipidemia. These drugs are covered at no charge or at a reduced cost share. Medications are under continual
COPD Update. Plus New and Improved Products for Inhaled Therapy. Catherine Bourg Rebitch, PharmD, BCACP Clinical Associate Professor
 COPD Update Plus New and Improved Products for Inhaled Therapy Catherine Bourg Rebitch, PharmD, BCACP Clinical Associate Professor Disclosure The presenter has nothing to disclose concerning possible financial
COPD Update Plus New and Improved Products for Inhaled Therapy Catherine Bourg Rebitch, PharmD, BCACP Clinical Associate Professor Disclosure The presenter has nothing to disclose concerning possible financial
Step Therapy Requirements
 An Independent Licensee of the Blue Cross and Blue Shield Association Step Therapy Requirements Effective: 12/01/2017 Updated 11/2017 H0302_2_2014 CMS Accepted 05/05/2014 1 ABILIFY Abilify 10 mg tablet
An Independent Licensee of the Blue Cross and Blue Shield Association Step Therapy Requirements Effective: 12/01/2017 Updated 11/2017 H0302_2_2014 CMS Accepted 05/05/2014 1 ABILIFY Abilify 10 mg tablet
JANUVIA 50 MG TABLET BYDUREON 2 MG/0.65 ML JARDIANCE 10 MG TABLET SUBCUTANEOUS PEN INJECTOR JARDIANCE 25 MG TABLET BYDUREON BCISE 2 MG/0.
 ANTI DIABETICS BYDUREON 2 MG SUBCUTANEOUS JANUVIA 25 MG TABLET EXTENDED RELEASE SUSPENSION JANUVIA 50 MG TABLET BYDUREON 2 MG/0.65 ML JARDIANCE 10 MG TABLET SUBCUTANEOUS PEN INJECTOR JARDIANCE 25 MG TABLET
ANTI DIABETICS BYDUREON 2 MG SUBCUTANEOUS JANUVIA 25 MG TABLET EXTENDED RELEASE SUSPENSION JANUVIA 50 MG TABLET BYDUREON 2 MG/0.65 ML JARDIANCE 10 MG TABLET SUBCUTANEOUS PEN INJECTOR JARDIANCE 25 MG TABLET
EFFECTIVE 01/04/2019. pimecrolimus 1 % cream (g) - Added to Tier 1 - ST Added: TOPICAL IMMUNOMODULATORS
 EFFECTIVE 01/04/2019 pimecrolimus 1 % cream (g) - ST Added: TOPICAL IMMUNOMODULATORS PAGE 1 LAST UPDATED 03/2019 EFFECTIVE 01/05/2019 LORBRENA 100 MG TABLET lorlatinib LORBRENA 25 MG TABLET lorlatinib
EFFECTIVE 01/04/2019 pimecrolimus 1 % cream (g) - ST Added: TOPICAL IMMUNOMODULATORS PAGE 1 LAST UPDATED 03/2019 EFFECTIVE 01/05/2019 LORBRENA 100 MG TABLET lorlatinib LORBRENA 25 MG TABLET lorlatinib
Commissioner for the Department for Medicaid Services Selections for Preferred Products
 Commissioner for the Department for Medicaid Services Selections for Preferred Products This is a summary of the final Preferred Drug List (PDL) selections made by the Commissioner for the Department for
Commissioner for the Department for Medicaid Services Selections for Preferred Products This is a summary of the final Preferred Drug List (PDL) selections made by the Commissioner for the Department for
Three s Company - The role of triple therapy in chronic obstructive pulmonary
 Three s Company - The role of triple therapy in chronic obstructive pulmonary disease (COPD) October 26 th, 2018 Zahava Picado, PharmD PGY1 Pharmacy Resident Central Texas Veterans Healthcare System Zahava.Picado@va.gov
Three s Company - The role of triple therapy in chronic obstructive pulmonary disease (COPD) October 26 th, 2018 Zahava Picado, PharmD PGY1 Pharmacy Resident Central Texas Veterans Healthcare System Zahava.Picado@va.gov
A Patient s Guide to Aerosol Medication Delivery
 A Patient s Guide to Aerosol Medication Delivery 3rd Edition Prepared by: Tim Op t Holt, EdD, RRT, AE-C, FAARC Kimberly Wiles, RRT, CPFT Ellen Becker, PhD, RRT, RRT-NPS, RPFT, AE-C, FAARC Edited by: Timothy
A Patient s Guide to Aerosol Medication Delivery 3rd Edition Prepared by: Tim Op t Holt, EdD, RRT, AE-C, FAARC Kimberly Wiles, RRT, CPFT Ellen Becker, PhD, RRT, RRT-NPS, RPFT, AE-C, FAARC Edited by: Timothy
TennCare Program TN MAC Price Change List As of: 03/30/2017
 1 TN List Run : 03/30/17 Old PRAZOSIN HCL 5 MG CAPSULE ORAL 03/29/2017 1.11209 1.12560 ( 1.2) CAPTOPRIL 12.5 MG TABLET ORAL 07/07/2015 1.07191 1.10416 ( 2.9) ISOSORBIDE DINITRATE 5 MG TABLET ORAL 03/29/2017
1 TN List Run : 03/30/17 Old PRAZOSIN HCL 5 MG CAPSULE ORAL 03/29/2017 1.11209 1.12560 ( 1.2) CAPTOPRIL 12.5 MG TABLET ORAL 07/07/2015 1.07191 1.10416 ( 2.9) ISOSORBIDE DINITRATE 5 MG TABLET ORAL 03/29/2017
Up in FLAMES: Stable Chronic Obstructive Pulmonary Disease (COPD) Management. Colleen Sakon, PharmD BCPS September 27, 2018
 Up in FLAMES: Stable Chronic Obstructive Pulmonary Disease (COPD) Management Colleen Sakon, PharmD BCPS September 27, 2018 Disclosures I have no actual or potential conflicts of interest 2 Objectives Summarize
Up in FLAMES: Stable Chronic Obstructive Pulmonary Disease (COPD) Management Colleen Sakon, PharmD BCPS September 27, 2018 Disclosures I have no actual or potential conflicts of interest 2 Objectives Summarize
Step Therapy Requirements
 An Independent Licensee of the Blue Cross and Blue Shield Association Step Therapy Requirements Effective: 05/01/2018 Updated 4/2018 H0302_2_2014 CMS Accepted 05/05/2014 1 BETA-BLOCKERS BYSTOLIC 10 MG
An Independent Licensee of the Blue Cross and Blue Shield Association Step Therapy Requirements Effective: 05/01/2018 Updated 4/2018 H0302_2_2014 CMS Accepted 05/05/2014 1 BETA-BLOCKERS BYSTOLIC 10 MG
Reference Guide for Caring for Pediatric Patients with Asthma
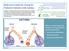 Reference Guide for Caring for Pediatric Patients with Asthma Co-Chair: Nancy Cantey Banasiak, DNP, PPCNP-BC, APRN Co-Chair: Deborah Hickman, DNP, APRN-CNP, CPNP-PC, NNP-BC Asthma and Allergy SIG Members
Reference Guide for Caring for Pediatric Patients with Asthma Co-Chair: Nancy Cantey Banasiak, DNP, PPCNP-BC, APRN Co-Chair: Deborah Hickman, DNP, APRN-CNP, CPNP-PC, NNP-BC Asthma and Allergy SIG Members
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: (Seebri Neohaler) Reference Number: CP.CPA.150 Effective Date: 11.16.16 Last Review Date: 11.17 Line of Business: Commercial Revision Log See Important Reminder at the end of this policy
Clinical Policy: (Seebri Neohaler) Reference Number: CP.CPA.150 Effective Date: 11.16.16 Last Review Date: 11.17 Line of Business: Commercial Revision Log See Important Reminder at the end of this policy
Inhaled Corticosteroid Dose Comparison in Asthma
 This Clinical Resource gives subscribers additional insight related to the Recommendations published in April 2017 ~ Resource #330402 Inhaled Corticosteroid Dose Comparison in Asthma The chart below provides
This Clinical Resource gives subscribers additional insight related to the Recommendations published in April 2017 ~ Resource #330402 Inhaled Corticosteroid Dose Comparison in Asthma The chart below provides
Michigan Department of Community Health Quantity Limitations
 Abstral (fentanyl) sl tab all strength Acetaminophen Actonel Actonel 35mg Adderall XR 5mg, 10mg, 15mg 240 per 34 days 3 gm/day 2 every 28 days 4 every 28 days Advair Diskus. No more than 180 every 30 days
Abstral (fentanyl) sl tab all strength Acetaminophen Actonel Actonel 35mg Adderall XR 5mg, 10mg, 15mg 240 per 34 days 3 gm/day 2 every 28 days 4 every 28 days Advair Diskus. No more than 180 every 30 days
Asthma/COPD Update with Inhaler Workshop
 Asthma/COPD Update with Inhaler Workshop October 8, 2017 Nathan Samsa, DO, Pharm D, RPh, FACOI None Disclosures Agenda Asthma Updates COPD Updates Inhaler Workshop Asthma Updates Asthma Updates SMART Trial
Asthma/COPD Update with Inhaler Workshop October 8, 2017 Nathan Samsa, DO, Pharm D, RPh, FACOI None Disclosures Agenda Asthma Updates COPD Updates Inhaler Workshop Asthma Updates Asthma Updates SMART Trial
12:00 Autonomic Drugs 12:00. Autonomic Drugs
 Autonomic Drugs Autonomic Drugs 12:04 PARASYMPATHOMIMETIC (CHOLINERGIC) AGENTS PILOCARPINE HCL 5 MG ORAL TABLET 00002216345 SALAGEN 1.4298 PYRIDOSTIGMINE BROMIDE 60 MG ORAL TABLET 00000869961 MESTINON
Autonomic Drugs Autonomic Drugs 12:04 PARASYMPATHOMIMETIC (CHOLINERGIC) AGENTS PILOCARPINE HCL 5 MG ORAL TABLET 00002216345 SALAGEN 1.4298 PYRIDOSTIGMINE BROMIDE 60 MG ORAL TABLET 00000869961 MESTINON
Clinical Policy: Roflumilast (Daliresp) Reference Number: CP.PMN.46 Effective Date: Last Review Date: 08.18
 Clinical Policy: (Daliresp) Reference Number: CP.PMN.46 Effective Date: 11.01.11 Last Review Date: 08.18 Line of Business: Medicaid Revision Log See Important Reminder at the end of this policy for important
Clinical Policy: (Daliresp) Reference Number: CP.PMN.46 Effective Date: 11.01.11 Last Review Date: 08.18 Line of Business: Medicaid Revision Log See Important Reminder at the end of this policy for important
Pharmacy Updates Summary
 All of the following changes were reviewed and approved by the SFHP Pharmacy & Therapeutics (P&T) Committee on 7/16/2014 Effective date: 8/15/2014 Therapeutic Classes reviewed: Hepatitis C Ophthalmic Prostaglandins
All of the following changes were reviewed and approved by the SFHP Pharmacy & Therapeutics (P&T) Committee on 7/16/2014 Effective date: 8/15/2014 Therapeutic Classes reviewed: Hepatitis C Ophthalmic Prostaglandins
Quarterly pharmacy formulary change notice
 Provider Bulletin June 24, 2016 Summary of change The Pharmacy and Therapeutics Committee reviewed and approved the formulary changes listed in the table below on March 29, 2016. What this means to you
Provider Bulletin June 24, 2016 Summary of change The Pharmacy and Therapeutics Committee reviewed and approved the formulary changes listed in the table below on March 29, 2016. What this means to you
2014 Step Therapy Criteria (List of Step Therapy Criteria)
 Criteria Last Updated: November 1, 2014 2014 Step Therapy Criteria (List of Step Therapy Criteria) PLEASE READ CAREFULLY: IEHP MEDICARE DUALCHOICE (HMO SNP) REQUIRES YOU TO FIRST TRY CERTAIN DRUGS TO TREAT
Criteria Last Updated: November 1, 2014 2014 Step Therapy Criteria (List of Step Therapy Criteria) PLEASE READ CAREFULLY: IEHP MEDICARE DUALCHOICE (HMO SNP) REQUIRES YOU TO FIRST TRY CERTAIN DRUGS TO TREAT
Question I was one of the first dry power devices available in the US Flovent, Serevent and Advair are all available in this device
 What Device am I Class Side Effects History Potpourri Monitoring Tools 10 10 10 10 10 20 20 20 20 20 30 30 30 30 30 40 40 40 40 40 50 50 50 50 50 WHAT KIND OF DEVICE AM I? I was one of the first dry power
What Device am I Class Side Effects History Potpourri Monitoring Tools 10 10 10 10 10 20 20 20 20 20 30 30 30 30 30 40 40 40 40 40 50 50 50 50 50 WHAT KIND OF DEVICE AM I? I was one of the first dry power
THERAPEUTIC AREA NAME STRENGTH DOSAGE FORM
 Value Based Tier Drugs are selected for the management of Asthma, Diabetes, Hypertension and Hyperlipidemia. These drugs are covered at no charge or at a reduced cost share. Medications are under continual
Value Based Tier Drugs are selected for the management of Asthma, Diabetes, Hypertension and Hyperlipidemia. These drugs are covered at no charge or at a reduced cost share. Medications are under continual
CRITERIA Trial of two generic formulary products from the following: atomoxetine or ADHD stimulant medication.
 ADHD STIMULANTS ATOMOXETINE HCL, DEXEDRINE 10 MG TABLET, DEXEDRINE 5 MG TABLET, DEXMETHYLPHENIDATE HCL, DEXMETHYLPHENIDATE HCL ER, DEXTROAMPHETAMINE 10 MG TAB, DEXTROAMPHETAMINE 5 MG TAB, DEXTROAMPHETAMINE
ADHD STIMULANTS ATOMOXETINE HCL, DEXEDRINE 10 MG TABLET, DEXEDRINE 5 MG TABLET, DEXMETHYLPHENIDATE HCL, DEXMETHYLPHENIDATE HCL ER, DEXTROAMPHETAMINE 10 MG TAB, DEXTROAMPHETAMINE 5 MG TAB, DEXTROAMPHETAMINE
Prescription benefit updates Individual/small group
 Prescription benefit updates Individual/small group Moda Health s prescription program is a pharmacy benefit that offers members a choice of safe and effective medication treatments. The program also helps
Prescription benefit updates Individual/small group Moda Health s prescription program is a pharmacy benefit that offers members a choice of safe and effective medication treatments. The program also helps
HEALTH SHARE/PROVIDENCE (OHP)
 HEALTH SHARE/PROVIDENCE (OHP) STEP THERAPY This is a complete list of drugs that have written coverage determination policies. Drugs on this list do not indicate that this particular drug will be covered
HEALTH SHARE/PROVIDENCE (OHP) STEP THERAPY This is a complete list of drugs that have written coverage determination policies. Drugs on this list do not indicate that this particular drug will be covered
March 2018 P & T Updates
 March 2018 P & T Updates Commercial Triple Tier 4th Tier Applicable Traditional Prior Auth AURYXIA 3 2 12 tablets per BAXDELA TABLETS 3 2 2 tablets per Depending on your specific benefits and in which
March 2018 P & T Updates Commercial Triple Tier 4th Tier Applicable Traditional Prior Auth AURYXIA 3 2 12 tablets per BAXDELA TABLETS 3 2 2 tablets per Depending on your specific benefits and in which
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: (Fasenra) Reference Number: CP.PHAR.## Effective Date: 01.16.18 Last Review Date: 05.18 Line of Business: Commercial, Medicaid Revision Log See Important Reminder at the end of this policy
Clinical Policy: (Fasenra) Reference Number: CP.PHAR.## Effective Date: 01.16.18 Last Review Date: 05.18 Line of Business: Commercial, Medicaid Revision Log See Important Reminder at the end of this policy
Acyclovir Ointment. Aetna Better Health Pennsylvania. Products Affected. acyclovir ointment 5 % external Details. Criteria
 Medications that require Step Therapy (ST) require trial and failure of preferred formulary agents prior to their authorization. If the prerequisite medications have been filled within the specified time
Medications that require Step Therapy (ST) require trial and failure of preferred formulary agents prior to their authorization. If the prerequisite medications have been filled within the specified time
12/18/2017. Disclosures. Asthma Management Updates: A Focus on Long-acting Muscarinic Antagonists and Intermittent Inhaled Corticosteroid Dosing
 Asthma Management Updates: A Focus on Long-acting Muscarinic Antagonists and Intermittent Inhaled Corticosteroid Dosing Diana M. Sobieraj, PharmD, BCPS Assistant Professor University of Connecticut School
Asthma Management Updates: A Focus on Long-acting Muscarinic Antagonists and Intermittent Inhaled Corticosteroid Dosing Diana M. Sobieraj, PharmD, BCPS Assistant Professor University of Connecticut School
AGGRENOX. Products Affected. Details. Open 1 Last Updated: 10/01/2018. Aggrenox
 Open 1 Last Updated: 10/01/2018 AGGRENOX Aggrenox A documented trial of one month of formulary generic aspirin/dipyridamole capsules. NR_0009_3742 09/2014 Formulary ID: 19076: version 7 1 ANTIDEPRESSANTS
Open 1 Last Updated: 10/01/2018 AGGRENOX Aggrenox A documented trial of one month of formulary generic aspirin/dipyridamole capsules. NR_0009_3742 09/2014 Formulary ID: 19076: version 7 1 ANTIDEPRESSANTS
Global Strategy for the Diagnosis, Management and Prevention of COPD 2016 Clinical Practice Guideline. MedStar Health
 Global Strategy for the Diagnosis, Management and Prevention of COPD 2016 Clinical Practice Guideline MedStar Health These guidelines are provided to assist physicians and other clinicians in making decisions
Global Strategy for the Diagnosis, Management and Prevention of COPD 2016 Clinical Practice Guideline MedStar Health These guidelines are provided to assist physicians and other clinicians in making decisions
STEP THERAPY CRITERIA
 STEP THERAPY This is a complete list of drugs that have written coverage determination policies. Drugs on this list do not indicate that this particular drug will be covered under your medical or prescription
STEP THERAPY This is a complete list of drugs that have written coverage determination policies. Drugs on this list do not indicate that this particular drug will be covered under your medical or prescription
Using Inhaled Corticosteroids as Needed for Asthma: giving patients relief or leaving them breathless?
 Using Inhaled Corticosteroids as Needed for Asthma: giving patients relief or leaving them breathless? Lindsay Thomas, Pharm.D. PGY2 Ambulatory Care Resident Department of Pharmacotherapy and Pharmacy
Using Inhaled Corticosteroids as Needed for Asthma: giving patients relief or leaving them breathless? Lindsay Thomas, Pharm.D. PGY2 Ambulatory Care Resident Department of Pharmacotherapy and Pharmacy
Oregon Health Plan prescription benefit updates
 Oregon Health Plan prescription benefit updates EOCCO s prescription program is a pharmacy benefit that offers members a choice of safe and effective medication treatments. The program also helps you save
Oregon Health Plan prescription benefit updates EOCCO s prescription program is a pharmacy benefit that offers members a choice of safe and effective medication treatments. The program also helps you save
Asthma & COPD Medication Review. Hutchison Disclosures 2/16/2017. Objectives
 Asthma & COPD Medication Review Anna Meador, PharmD, BCACP Assistant Professor/ Pharmacy Director McWhorter School of Pharmacy/ Christ Health Center Amber Hutchison, PharmD, BCPS Assistant Clinical Professor
Asthma & COPD Medication Review Anna Meador, PharmD, BCACP Assistant Professor/ Pharmacy Director McWhorter School of Pharmacy/ Christ Health Center Amber Hutchison, PharmD, BCPS Assistant Clinical Professor
Adjustment of Inhaled Controller Therapy of Asthma in the Yellow Zone, Based on the Inhaler Product Used in the Green Zone Age 12 Years and Older
 Adjustment of Inhaled Controller Therapy of Asthma in the Yellow Zone, Based on the Inhaler Product Used in the Green Zone Age 12 Years and Older The Canadian Thacic Society recommends a tempary, greater
Adjustment of Inhaled Controller Therapy of Asthma in the Yellow Zone, Based on the Inhaler Product Used in the Green Zone Age 12 Years and Older The Canadian Thacic Society recommends a tempary, greater
Medicare Part D 2012 Formulary Changes Service To Senior and Total Fit
 Medicare Part D 2012 Formulary s Service To Senior and Total Fit Inter Valley Health Plan may add or remove drugs from our formulary during the year. If we remove a drug from our formulary, add prior authorization,
Medicare Part D 2012 Formulary s Service To Senior and Total Fit Inter Valley Health Plan may add or remove drugs from our formulary during the year. If we remove a drug from our formulary, add prior authorization,
ABILIFY ABILIFY DISCMELT ACTONEL ACTOPLUS MET ACTOPLUS MET XR ACTOS ADCIRCA ADVAIR DISKUS ADVAIR HFA
 Quantity Limits Paramount Medicare Formulary 2012 Formulary ID 12112, Version 22. CMS Approved 10-23-2012. ABILIFY Abilify TABS ABILIFY DISCMELT Abilify Discmelt ACTONEL Actonel TABS 150MG Actonel TABS
Quantity Limits Paramount Medicare Formulary 2012 Formulary ID 12112, Version 22. CMS Approved 10-23-2012. ABILIFY Abilify TABS ABILIFY DISCMELT Abilify Discmelt ACTONEL Actonel TABS 150MG Actonel TABS
The Latest Medications A Pharmacological Update for RTs
 The Latest Medications A Pharmacological Update for RTs Douglas S. Gardenhire, EdD, RRT-NPS, FAARC Associate Professor and Chairman Department of Respiratory Therapy Georgia State University Objectives
The Latest Medications A Pharmacological Update for RTs Douglas S. Gardenhire, EdD, RRT-NPS, FAARC Associate Professor and Chairman Department of Respiratory Therapy Georgia State University Objectives
CAMPER APPLICATION PACKET
 CAMPER APPLICATION PACKET Monday- Friday June 12-16, 2017 Rockport, Texas DEADLINE FOR SUBMITTING ALL FORMS: THE IMPORTANCE OF COMPLETING ALL CAMP FORMS Although it may seem like a lot of paperwork, the
CAMPER APPLICATION PACKET Monday- Friday June 12-16, 2017 Rockport, Texas DEADLINE FOR SUBMITTING ALL FORMS: THE IMPORTANCE OF COMPLETING ALL CAMP FORMS Although it may seem like a lot of paperwork, the
What You Need to Know about Metered-Dose Inhalers and the HFA Propellant
 What You Need to Know about Metered- Inhalers and the HFA Propellant There are a number ways to deliver inhaled medication. They include: Metered-dose inhaler () Metered-dose inhaler with spacer/holding
What You Need to Know about Metered- Inhalers and the HFA Propellant There are a number ways to deliver inhaled medication. They include: Metered-dose inhaler () Metered-dose inhaler with spacer/holding
End Stage COPD Guidance Document
 End Stage COPD Guidance Document Suggested Guidelines for the Determination of Hospice Eligibility A patient with severe chronic pulmonary disease that meets the following criteria may be eligible for
End Stage COPD Guidance Document Suggested Guidelines for the Determination of Hospice Eligibility A patient with severe chronic pulmonary disease that meets the following criteria may be eligible for
Quantity Limit Drug List
 Quantity Limit Drug List Quantity limits define the maximum amount of medication covered by your drug benefit for one prescription or copayment. This means your benefit covers a limited number of doses
Quantity Limit Drug List Quantity limits define the maximum amount of medication covered by your drug benefit for one prescription or copayment. This means your benefit covers a limited number of doses
Pharmacy Updates Summary
 All of the following changes were reviewed and approved by the SFHP Pharmacy & Therapeutics (P&T) Committee on 4/16/2014 Effective date: 5/15/2014 Therapeutic Classes reviewed: ADHD Ophthalmic antihistamines
All of the following changes were reviewed and approved by the SFHP Pharmacy & Therapeutics (P&T) Committee on 4/16/2014 Effective date: 5/15/2014 Therapeutic Classes reviewed: ADHD Ophthalmic antihistamines
Asthma Management Updates: A Focus on Long-acting Muscarinic Antagonists and Intermittent Inhaled Corticosteroid Dosing
 Asthma Management Updates: A Focus on Long-acting Muscarinic Antagonists and Intermittent Inhaled Corticosteroid Dosing Diana M. Sobieraj, PharmD, BCPS Assistant Professor University of Connecticut School
Asthma Management Updates: A Focus on Long-acting Muscarinic Antagonists and Intermittent Inhaled Corticosteroid Dosing Diana M. Sobieraj, PharmD, BCPS Assistant Professor University of Connecticut School
Allergies and Asthma 5/21/2013. Objectives. Allergic Rhinitis (AR): Risk Factor for ASTHMA. Rhinitis and Asthma
 Allergies and Asthma Presented By: Dr. Fadwa Gillanders, Pharm.D Clinical Pharmacy Specialist May 2013 Objectives Understand the relationship between asthma and allergic rhinitis Understand what is going
Allergies and Asthma Presented By: Dr. Fadwa Gillanders, Pharm.D Clinical Pharmacy Specialist May 2013 Objectives Understand the relationship between asthma and allergic rhinitis Understand what is going
Michigan Department of Community Health Co-pay and Quantity Limitations
 Michigan Department of Community Health Co-pay and Quantity Limitations Benefit Plan Co-pay Information Group ID Coverage Co-pay INCARCE Incarcerated Medicaid No coverage No coverage patients SHPDUAL CSHCSCAID
Michigan Department of Community Health Co-pay and Quantity Limitations Benefit Plan Co-pay Information Group ID Coverage Co-pay INCARCE Incarcerated Medicaid No coverage No coverage patients SHPDUAL CSHCSCAID
