GSK Medicine: salmeterol, Salmeterol+Fluticasone proprionate, fluticasone propionate, beclomethasone
|
|
|
- Bridget Blake
- 5 years ago
- Views:
Transcription
1 GSK Medicine: salmeterol, Salmeterol+Fluticasone proprionate, fluticasone propionate, beclomethasone Study No.: WWE111984/WEUSRTP2640/EPI40528 Title: The Asthma Death Case Control Study (ADCCS): Association between Recent Medication Use and Asthma Mortality Rationale: Two clinical trials reported evidence of an increased risk of asthma-related death in patients treated with salmeterol, a long-acting beta2-agonist (LABA) [Castle, 1993; Nelson, 2006]; however, observational studies conducted to date have not reported this association. The largest observational study conducted to date is the British Asthma Death Case-Control Study (ADCCS), which consisted of 532 asthma deaths (cases) and 532 controls with hospital admissions for asthma (controls) [Anderson, 2005]. Based on mentions in the primary care record, this original ADCCS study investigated use of LABA in the 0 to 3 months, >3 to 12 months or >1 to 5 years prior to index date (date of death for cases; date of asthma admission for controls). However, comparisons between cases and controls in the period immediately leading up to the index date may have been differentially biased, as medication information in the primary care notes was not as complete in the cases relative to the controls. Objectives: After adjusting for previously identified sources of bias in the 2005 Anderson study, this study was conducted using ADCCS data to investigate the association between asthma death and recent mention of LABA in the primary care record including potential effect modification by inhaled corticosteroids (ICS). The primary objective of the current study was to examine: current use of LABA ( 2 months of index date) and the risk of asthma death including effect modification by the concurrent use of ICS (within one month either side of LABA). Secondary objectives were to examine 1) Recent use of LABA (>2 to 6 months prior to index date) and the risk of asthma death including effect modification by the concurrent use of ICS, 2) Use of other medications and the risk of asthma death (oral corticosteroids, inhaled corticosteroids, inhaled short-acting beta2-agonists and antibiotics), and 3) Effect modification by age and by deprivation rank. Indication: asthma Study Investigators/Centers: H. Ross Anderson, St. George s, University of London. Research Methods: Data Source: The original ADCSS study extracted data from anonymized photocopies of primary care records covering the 5 years prior to the index date. Primary care records could include the general practitioner s notes, computer printouts, referral letters, hospital discharge, and outpatient letters. Data for the original analysis were extracted in a blinded fashion to patient case-control status from the paper records with details described previously [Anderson, 2005]. As a continuation of the ADCCS, more detailed information was extracted on asthma medications and antibiotics mentioned in the 7 months leading up to the index event. In addition, information on COPD and other chronic lung diseases was re-extracted in an attempt to improve data quality. The new information required for this study was extracted twice in a blinded fashion and resulting data was entered into the preexisting ADCCS study database. 1
2 Study Design: Case-control study. This was a retrospective case-control study of asthma patients in Britain. The ADCCS consisted of 532 asthma deaths (cases) between 1994 and 1998 and 532 hospital admissions with a primary discharge diagnosis of asthma (controls). The index date was the date of death for cases and the date of hospital admission for the controls. Cases and controls were matched on hospital, index date and age. Study Population: Asthma patients in this study were younger than 65 years of age living within specific areas of England, the populated central belt of Scotland, and all of Wales. Study areas in England included East Anglia, North Essex, West Midlands, Northampton and Kettering, Cumbria and districts north of the Tyne. Asthma deaths (cases) were identified between for England and Wales, and in Scotland between 1996 and All patients were considered to have severe asthma as controls experienced an asthma-related hospitalization and cases died due to asthma. Study Exposures, Outcomes: Exposure: Exposures were defined as prescribed asthma medications according to the primary care record. The primary exposure of interest was long-acting beta2-agonists (LABA) prescription in the 0-2 months or >2 to 6 months prior to index date, with and without concurrent inhaled corticosteroids (ICS) prescription. Outcome: asthma-related death per death certificate Data Analysis Methods: The analysis was based on 532 cases and 532 controls analyzed in STATA 9 and10 using conditional logistic regression with case-control status as the dependent variable. Cases and controls were matched 1:1 on hospital, date of index event within 6 months, and age within 5 years. In terms of matching by hospital, the control was selected from the same or next nearest hospital to its case. If the case died in the community, the hospital to which the patient would have been admitted was chosen. For each medication class of interest, conditional logistic regression models were fitted to examine the effect of adjustment for important confounders. Unadjusted and adjusted model estimates were presented. The primary analysis adjusted for all confounders including age of asthma onset, mention of ever being obese, sex, number of previous hospital admissions for asthma in the past year and the >1 to 5 years prior, mention of COPD in the past 5 years, mention of other chronic lung disease in the past 5 years, mention of prescription for other asthma medication classes, index of deprivation rank, and country. Deprivation scores and their ranks were based on government indices calculated by each country and were not directly comparable. Variables in the model that were not dichotomous were entered as continuous variables, after assessing linearity using fractional polynomials. Interactions were examined in the model to identify potential effect modification. Effect modification of the association between LABA and risk of asthma death by concurrent ICS was evaluated. Effect modification was also examined with respect to age (< 45 years and 45 to 64 years) and deprivation (England less deprived, England more deprived, Scotland less deprived, Scotland more deprived, Wales less deprived and Wales more deprived), having categorised deprivation rank within country according to being above or below the country-specific median rank. The associations between medication class and asthma death and interaction terms in the model were assessed at the 5% significance level using the likelihood ratio test. 2
3 Limitations: This study utilized paper records from the general practice notes, including 5 years prior to the index date (death among cases or asthma hospitalization among controls). The analysis was based on mentions of patient demographics, asthma history, and asthma medication in the paper record. Mentions of medication and/or references to prescriptions in the paper record may not correspond to actual use of medications. In addition, information in the records may not have been as detailed or complete for some patients relative to others. There was the potential for residual confounding by severity despite matching cases and controls and having a control group that had been hospitalized for asthma Cases were significantly different with respect to earlier age of asthma onset and a higher proportion with mention of COPD and other chronic lung diseases in the last 5 years. While these factors were adjusted for in the analysis, cases and controls could be different in other ways that could not be accounted for in the analysis. Asthma management and treatment guidelines have changed since this study period ( ), which was before the approval of treatment alternatives including ICS/LABA combination therapy in a single device; therefore, the use of ICS and LABA in a single device could not be evaluated. The modifying effect of ICS use with LABA used in two devices could not be fully evaluated due to low power, as 92% of patients with mention of LABA in the past 6 months also had a mention of ICS concurrently (within one month in the patient record). Despite these limitations, these data provide important information on the safety of LABA including salmeterol. 3
4 Study Results: Median age was 53 years (interquartile range 40 to 59) for both cases and controls, who were also similar with respect to their proportion of males and females. The case series included 262 community deaths and 270 deaths in hospital with 140 dying on the day of admission. Compared to controls, cases had a significantly earlier age of asthma onset (p=0.013), higher proportions with mention of COPD (p=0.010) and other chronic lung disease (p=0.046) in the last 5 years, and a higher proportion of obesity (p=0.037) (Table1 ). Primary Objective: There was no evidence of an overall association between asthma death and mention of LABA in the primary care record in the 0-2 months prior to index date (odds ratio = 0.89 (0.61 to 1.30)) (Table 2). The odds ratios for LABA with and without a concurrent mention of inhaled corticosteroids were 0.92 (0.62 to 1.37) and 0.75 (0.32 to 1.77), respectively. Most controls with mention of LABA within the past 6 months also had a concurrent mention of ICS (92%), limiting the statistical power to detect any potential modifying effects of ICS. Secondary Objectives: There was no evidence of an overall association between asthma death and mention of LABA in the primary care record in the >2-6 months prior to index date (odds ratio = 1.08 (0.76 to 1.53)); although odds ratios differed significantly when comparing the under 45 year age group (0.63 (0.34 to 1.15) and the year age group (1.35 (0.90 to 2.03)). The odds ratios for LABA with and without a concurrent mention of inhaled corticosteroids were 1.05 (0.73 to 1.51) and 1.39 (0.53 to 3.65), respectively. Regarding medications in the two month period prior to the index date, mention of oral corticosteroids, injected corticosteroids or antibiotics were each significantly associated with a lower risk of asthma death. There was no evidence of an association of asthma death with either methylzanthines or inhaled corticosteroids. Atimuscarinics were associated with a statistically significant greater risk of asthma death, which was persistent in magnitude but was not statistically significant after full adjustment. In the period >2-6 months prior to the index date, there was no evidence of an association between asthma death and mention of corticosteroids (oral, inhaled, or injected), methylzanthines, antimuscarinics or antibiotics. During this period, short-acting beta2-agonists (SABA) were associated with a statistically significant increased risk of asthma death that persisted in magnitude but was not statistically significant after full adjustment. The odds ratios for SABA differed significantly across the 6 categories (i.e. England less deprived, England more deprived, Scotland less deprived, Scotland more deprived, Wales less deprived, Wales more deprived) formed by country and deprivation rank (OR=2.72 (1.48 to 5.00); OR=1.56 (0.97 to 2.52); OR=1.04 (0.28 to 3.83); OR=0.87 (0.39 to 1.93); OR=0.48 (0.14 to 1.64); OR=0.72 (0.33 to 1.55)) respectively. This pattern of results by SABA and region was unexpected and difficult to interpret. Table 1: Demographics/Baseline Characteristics Characteristic Cases (n = 532) Controls (n = 532) % % Median age (yrs) (interquartile range) 53 (40-59) 53 (40-59) Country England
5 Scotland Wales Females Median age at onset (yrs) 30 (10-47) 33 (13-49) COPD mentioned in the last 5 years Other chronic lung disease in the last 5 years # Obesity mentioned ever Death Information (Cases Only) Community death 49.2 NA Hospital death relative to admission Death on day of admission 29.7 NA 1-7 days after admission 12.2 NA 8-14 days after admission 3.0 NA days after admission 3.2 NA days after admission 1.3 NA >52 days after admission 1.3 NA Hospital admissions for asthma In the last year >1 to 5 years Median No. of hospital admissions for asthma in 2 (1-3) 2 (1-3) the last year (interquartile range) Median No. of hospital admissions for asthma in the last >1 to 5 years (interquartile range) 2 (1-5) 2 (1-5) # Other chronic lung diseases include bronchiectasis, aspergillosis, pulmonary TB and pulmonary fibrosis Information on age of onset missing for 39 cases and 30 controls. Includes 18 patients with missing information on length of stay. Figures take account of length of index admission etc. through censoring 5
6 Table 2: Primary and Secondary Outcome(s): Risk of asthma-related death associated with asthma medication use in time periods prior to index date Drug class Unadjusted Model Adjusted Model OR (95% CI) OR (95% CI) Mention in prior 0-2 months (n=1032) Short acting 2-agonists All to to 1.93 Long acting 2-agonists All to to 1.30 Salmeterol to to 1.20 Antimuscarinics All 1.34* 1.01 to to 1.95 Corticosteroids All to *** 0.36 to 0.73 Inhaled to to 1.22 Oral to ** 0.49 to 0.88 Injected 0.41* 0.18 to * 0.14 to 0.81 Methylzanthines All to to 1.73 Antibiotics All to * 0.50 to 0.92 Drug class Unadjusted Model Adjusted Model OR (95% CI) OR (95% CI) Mention in the >2 months to 6 months prior (n=1052) Short acting 2-agonists All 1.35* 1.05 to to 1.99 Long acting 2-agonists All to to 1.53 Salmeterol to to 1.45 Antimuscarinics All to to 1.55 Corticosteroids All to to 1.43 Inhaled to to 1.20 Oral to to 1.33 Injected to to 2.58 Methylzanthines All to to 1.38 Antibiotics All to to 1.26 Adjusted for matching and sex only Adjusted for matching, sex, age of asthma onset, mention of obesity ever, number of hospital admissions for asthma in the past year and >1 to 5 years prior, mention of COPD in the past 5 years, mention of other chronic lung disease in the past 5 years, mention / prescription of other asthma drug classes (as appropriate), and deprivation rank within country (Sources of deprivation data: National Statistics, Ordinance Survey, Office of the Deputy Priminister, Welsh Assembly Government, The Scottish Government). * p<0.05, ** p<0.01, *** p<0.001 Conclusion: In this British population-based case-control study, there was no evidence of an overall association between asthma death and recent prescriptions of LABA either in the 0-2 months prior or the >2-6 months prior to the index date after adjusting for measured confounders. In the 6-month period prior to the index date, most controls with LABA mentioned in their primary care record also had a mention of concurrent ICS (92%), limiting the statistical power to detect any modifying effects of ICS when used with LABA. In the 0-2 month period prior to the index date, mention of oral corticosteroids, injected corticosteroids and antibiotics were each significantly associated with a lower risk of asthma death, which may reflect appropriate asthma treatment and/or health care utilization. 6
Outcomes: Initially, our primary definitions of pneumonia was severe pneumonia, where the subject was hospitalized
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
Online supplementary material
 Online supplementary material Add-on long-acting β2-agonist (LABA) in a separate inhaler as asthma step-up therapy versus increased dose of inhaled corticosteroid (ICS) or ICS/LABA combination inhaler
Online supplementary material Add-on long-acting β2-agonist (LABA) in a separate inhaler as asthma step-up therapy versus increased dose of inhaled corticosteroid (ICS) or ICS/LABA combination inhaler
Optum Research Database Proportion of asthma patients with prescription fills for rescue and controller medications by year,
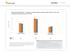 ASTHMA CHART 1 Optum Research Database Proportion of asthma patients with prescription fills for rescue and controller medications by year, 215-216 7 6 5 4 3 2 1 1 Controller medication 1 Rescue medication
ASTHMA CHART 1 Optum Research Database Proportion of asthma patients with prescription fills for rescue and controller medications by year, 215-216 7 6 5 4 3 2 1 1 Controller medication 1 Rescue medication
Policy Evaluation: Step Therapy Prior Authorization of Combination Inhaled Corticosteroid / Long-Acting Beta-Agonists
 Drug Use Research & Management Program OHA Division of Medical Assistance Programs 500 Summer Street NE, E35; Salem, OR 97301-1079 Phone 503-947-5220 Fax 503-947-1119 Policy Evaluation: Step Therapy Prior
Drug Use Research & Management Program OHA Division of Medical Assistance Programs 500 Summer Street NE, E35; Salem, OR 97301-1079 Phone 503-947-5220 Fax 503-947-1119 Policy Evaluation: Step Therapy Prior
Kirthi Gunasekera MD Respiratory Physician National Hospital of Sri Lanka Colombo,
 Kirthi Gunasekera MD Respiratory Physician National Hospital of Sri Lanka Colombo, BRONCHODILATORS: Beta Adrenoreceptor Agonists Actions Adrenoreceptor agonists have many of the same actions as epinephrine/adrenaline,
Kirthi Gunasekera MD Respiratory Physician National Hospital of Sri Lanka Colombo, BRONCHODILATORS: Beta Adrenoreceptor Agonists Actions Adrenoreceptor agonists have many of the same actions as epinephrine/adrenaline,
Research in Real Life
 Research in Real Life Study 1: Exploratory study - identifying the benefits of pmdi versus Diskus for delivering fluticasone/salmeterol combination therapy in patients with chronic obstructive pulmonary
Research in Real Life Study 1: Exploratory study - identifying the benefits of pmdi versus Diskus for delivering fluticasone/salmeterol combination therapy in patients with chronic obstructive pulmonary
Inhaled Corticosteroids for the Treatment of Chronic Asthma in Adults & Adolescents aged 12 years & over
 Manufacturer Submission To The National Institute for Health and Clinical Excellence By GlaxoSmithKline UK Inhaled Corticosteroids for the Treatment of Chronic Asthma in Adults & Adolescents aged 12 years
Manufacturer Submission To The National Institute for Health and Clinical Excellence By GlaxoSmithKline UK Inhaled Corticosteroids for the Treatment of Chronic Asthma in Adults & Adolescents aged 12 years
Surveillance report Published: 6 April 2016 nice.org.uk. NICE All rights reserved.
 Surveillance report 2016 Chronic obstructive pulmonary disease in over 16s: diagnosis and management (2010) NICE guideline CG101 Surveillance report Published: 6 April 2016 nice.org.uk NICE 2016. All rights
Surveillance report 2016 Chronic obstructive pulmonary disease in over 16s: diagnosis and management (2010) NICE guideline CG101 Surveillance report Published: 6 April 2016 nice.org.uk NICE 2016. All rights
Inhaled Corticosteroid vs. Add-On Long-Acting Beta-Agonist for Step-Up Therapy in Asthma
 Online Data Supplement Inhaled Corticosteroid vs. Add-On Long-Acting Beta-Agonist for Step-Up Therapy in Asthma Elliot Israel, Nicolas Roche, Richard J. Martin, Gene Colice, Paul M. Dorinsky, Dirkje S.
Online Data Supplement Inhaled Corticosteroid vs. Add-On Long-Acting Beta-Agonist for Step-Up Therapy in Asthma Elliot Israel, Nicolas Roche, Richard J. Martin, Gene Colice, Paul M. Dorinsky, Dirkje S.
Asthma Treatment Guideline for Adults (aged 17 and over)
 Asthma Treatment Guideline for Adults (aged 17 and over) Sharon Andrew MLCSU January 2019 (Review date 0 January 2022) VERSION CONTROL. Please access via the LMMG website to ensure that the correct version
Asthma Treatment Guideline for Adults (aged 17 and over) Sharon Andrew MLCSU January 2019 (Review date 0 January 2022) VERSION CONTROL. Please access via the LMMG website to ensure that the correct version
Comprehensive Research Plan: Inhaled corticosteroids + long-acting beta agonists (ICS+LABA) for the treatment of asthma
 Comprehensive Research Plan: Inhaled corticosteroids + long-acting beta agonists (ICS+LABA) for the treatment of asthma Pharmacoepidemiology Unit July 10, 2014 30 Bond Street, Toronto ON, M5B 1W8 www.odprn.ca
Comprehensive Research Plan: Inhaled corticosteroids + long-acting beta agonists (ICS+LABA) for the treatment of asthma Pharmacoepidemiology Unit July 10, 2014 30 Bond Street, Toronto ON, M5B 1W8 www.odprn.ca
Research in real life ltd study protocol for Teva Pharmaceutical Industries Ltd.
 Research in real life ltd study protocol for Teva Pharmaceutical Industries Ltd. The use of a spacer in the delivery of large (Fluticasone Propionate) and small particle FP and Qvar Spacer vs Non-Spacer
Research in real life ltd study protocol for Teva Pharmaceutical Industries Ltd. The use of a spacer in the delivery of large (Fluticasone Propionate) and small particle FP and Qvar Spacer vs Non-Spacer
Design - Multicentre prospective cohort study. Setting UK Community Pharmacies within one CCG area within the UK
 Enabling Patient Health Improvements through COPD (EPIC) Medicines Optimisation within Community Pharmacy: a prospective cohort study Abstract Objectives To improve patients ability to manage their own
Enabling Patient Health Improvements through COPD (EPIC) Medicines Optimisation within Community Pharmacy: a prospective cohort study Abstract Objectives To improve patients ability to manage their own
Adherence to asthma controller medication regimens
 Respiratory Medicine (2005) 99, 1263 1267 Adherence to asthma controller medication regimens D.A. Stempel a,, S.W. Stoloff b, J.R. Carranza Rosenzweig c, R.H. Stanford c, K.L. Ryskina d, A.P. Legorreta
Respiratory Medicine (2005) 99, 1263 1267 Adherence to asthma controller medication regimens D.A. Stempel a,, S.W. Stoloff b, J.R. Carranza Rosenzweig c, R.H. Stanford c, K.L. Ryskina d, A.P. Legorreta
Evaluation of Asthma Management in Middle EAst North Africa Adult population
 STUDY REPORT SUMMARY Evaluation of Asthma Management in Middle EAst North Africa Adult population Descriptive study on the management of asthma in an asthmatic Middle East Africa adult population Background/Rationale:
STUDY REPORT SUMMARY Evaluation of Asthma Management in Middle EAst North Africa Adult population Descriptive study on the management of asthma in an asthmatic Middle East Africa adult population Background/Rationale:
BEDFORDSHIRE AND LUTON JOINT PRESCRIBING COMMITTEE
 BEDFORDSHIRE AND LUTON JOINT PRESCRIBING COMMITTEE December 2014 Review Date: December 2017 Bulletin 206 : DuoResp Spiromax 160 / 4.5 and 320 / 9 budesonide & formoterol dry powder inhaler JPC Recommendations
BEDFORDSHIRE AND LUTON JOINT PRESCRIBING COMMITTEE December 2014 Review Date: December 2017 Bulletin 206 : DuoResp Spiromax 160 / 4.5 and 320 / 9 budesonide & formoterol dry powder inhaler JPC Recommendations
NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE SCOPE
 NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE 1 Guideline title SCOPE Chronic obstructive pulmonary disease: the management of adults with chronic obstructive pulmonary disease in primary and secondary
NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE 1 Guideline title SCOPE Chronic obstructive pulmonary disease: the management of adults with chronic obstructive pulmonary disease in primary and secondary
Effects of age-at-diagnosis and duration of diabetes on GADA and IA-2A positivity
 Effects of age-at-diagnosis and duration of diabetes on GADA and IA-2A positivity Duration of diabetes was inversely correlated with age-at-diagnosis (ρ=-0.13). However, as backward stepwise regression
Effects of age-at-diagnosis and duration of diabetes on GADA and IA-2A positivity Duration of diabetes was inversely correlated with age-at-diagnosis (ρ=-0.13). However, as backward stepwise regression
NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE SCOPE
 DRAFT NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE 1 Guideline title SCOPE Chronic obstructive pulmonary disease: the management of adults with chronic obstructive pulmonary disease in primary
DRAFT NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE 1 Guideline title SCOPE Chronic obstructive pulmonary disease: the management of adults with chronic obstructive pulmonary disease in primary
Type of intervention Treatment. Economic study type Cost-effectiveness analysis.
 Cost-effectiveness of salmeterol/fluticasone propionate combination product 50/250 micro g twice daily and budesonide 800 micro g twice daily in the treatment of adults and adolescents with asthma Lundback
Cost-effectiveness of salmeterol/fluticasone propionate combination product 50/250 micro g twice daily and budesonide 800 micro g twice daily in the treatment of adults and adolescents with asthma Lundback
Choosing an inhaler for COPD made simple. Dr Simon Hart Castle Hill Hospital
 Choosing an inhaler for COPD made simple Dr Simon Hart Castle Hill Hospital 1 Declaration of interests I have received speaker fees, sponsorship to attend conferences, and funding for research from companies
Choosing an inhaler for COPD made simple Dr Simon Hart Castle Hill Hospital 1 Declaration of interests I have received speaker fees, sponsorship to attend conferences, and funding for research from companies
Include patients: with a confirmed diagnosis of asthma who have been free of asthma symptoms for 3 months or more.
 Corby Clinical Commissioning Group Kettering General Hospital NHS Trust Nene Clinical Commissioning Group rthampton General Hospital NHS Trust rthamptonshire Healthcare Foundation Trust Stepping down asthma
Corby Clinical Commissioning Group Kettering General Hospital NHS Trust Nene Clinical Commissioning Group rthampton General Hospital NHS Trust rthamptonshire Healthcare Foundation Trust Stepping down asthma
Anna Hansell, Jen Hollowell, Tom Nichols, Rosie McNiece, David Strachan
 Thorax 1999;54:413 419 413 Department of Public Health Sciences, St George s Hospital Medical School, Cranmer Terrace, London SW17 RE, UK A Hansell T Nichols D Strachan General Practice Research Database,
Thorax 1999;54:413 419 413 Department of Public Health Sciences, St George s Hospital Medical School, Cranmer Terrace, London SW17 RE, UK A Hansell T Nichols D Strachan General Practice Research Database,
Supplementary materials
 Supplementary materials Table S1 Patient comorbidities by diagnosis Total Asthma n (%) 5348 (148.4) COPD n (%) 4563 (143.4) ACOS n (%) 469 (197.9) No other comorbidities 95 (26.4) 614 (19.3) 27 (11.4)
Supplementary materials Table S1 Patient comorbidities by diagnosis Total Asthma n (%) 5348 (148.4) COPD n (%) 4563 (143.4) ACOS n (%) 469 (197.9) No other comorbidities 95 (26.4) 614 (19.3) 27 (11.4)
Three s Company - The role of triple therapy in chronic obstructive pulmonary disease (COPD)
 Three s Company - The role of triple therapy in chronic obstructive pulmonary disease (COPD) Zahava Picado, PharmD PGY1 Pharmacy Practice Resident Central Texas Veterans Healthcare System Temple, TX October
Three s Company - The role of triple therapy in chronic obstructive pulmonary disease (COPD) Zahava Picado, PharmD PGY1 Pharmacy Practice Resident Central Texas Veterans Healthcare System Temple, TX October
Not available 100/6mcg 2 BD formoterol (Fostair MDI) 100/6mcg 33
 COMMISSIONING POLICY RECOMMENDATION TREATMENT ADVISORY GROUP FLUTICASONE FUROATE/VILANTEROL COMBINATION INHALER - ASTHMA Policy agreed by Vale of York CCG (date) Drug, Treatment, Device name Fluticasone
COMMISSIONING POLICY RECOMMENDATION TREATMENT ADVISORY GROUP FLUTICASONE FUROATE/VILANTEROL COMBINATION INHALER - ASTHMA Policy agreed by Vale of York CCG (date) Drug, Treatment, Device name Fluticasone
THE USE OF LONG-ACTING β 2 -AGONISTS AS MONOTHERAPY IN CHILDREN AND ADULTS
 THE USE OF LONG-ACTING β 2 -AGONISTS AS MONOTHERAPY IN CHILDREN AND ADULTS Pat G Camp 1,2, Tingting Zhang 3, M Anne Smith 3, Bruce C Carleton 3 1 UBC James Hogg Research Centre and the Institute for Heart
THE USE OF LONG-ACTING β 2 -AGONISTS AS MONOTHERAPY IN CHILDREN AND ADULTS Pat G Camp 1,2, Tingting Zhang 3, M Anne Smith 3, Bruce C Carleton 3 1 UBC James Hogg Research Centre and the Institute for Heart
Effects on Resource Utilization of Adding Salmeterol in Combination or Separately to Inhaled Corticosteroids
 RESEARCH Effects on Resource Utilization of Adding Salmeterol in Combination or Separately to Inhaled Corticosteroids JAMES CHAN, PharmD, PhD; RITA L. HUI, PharmD, MS; and MICHELE M. SPENCE, PhD ABSTRACT
RESEARCH Effects on Resource Utilization of Adding Salmeterol in Combination or Separately to Inhaled Corticosteroids JAMES CHAN, PharmD, PhD; RITA L. HUI, PharmD, MS; and MICHELE M. SPENCE, PhD ABSTRACT
Journal of the COPD Foundation. Journal Club. Chronic Obstructive Pulmonary Diseases:
 85 Journal Club Chronic Obstructive Pulmonary Diseases: Journal of the COPD Foundation Journal Club Ron Balkissoon, MD, MSc, DIH, FRCPC 1 Abbreviations: inhaled corticosteroid, ICS; long acting β 2 -agonist,
85 Journal Club Chronic Obstructive Pulmonary Diseases: Journal of the COPD Foundation Journal Club Ron Balkissoon, MD, MSc, DIH, FRCPC 1 Abbreviations: inhaled corticosteroid, ICS; long acting β 2 -agonist,
Study No.: Title: Rationale: Phase: Study Period: Study Design: Centres: Indication: Treatment: Objectives: Co-Primary Outcomes/Efficacy Variables:
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
Ivax Pharmaceuticals UK Sponsor Submission to the National Institute for Health and Clinical Excellence
 Ivax Pharmaceuticals UK Sponsor Submission to the National Institute for Health and Clinical Excellence Clinical and cost-effectiveness of QVAR for the treatment of chronic asthma in adults and children
Ivax Pharmaceuticals UK Sponsor Submission to the National Institute for Health and Clinical Excellence Clinical and cost-effectiveness of QVAR for the treatment of chronic asthma in adults and children
This is the publisher s version. This version is defined in the NISO recommended practice RP
 Journal Article Version This is the publisher s version. This version is defined in the NISO recommended practice RP-8-2008 http://www.niso.org/publications/rp/ Suggested Reference Chong, J., Karner, C.,
Journal Article Version This is the publisher s version. This version is defined in the NISO recommended practice RP-8-2008 http://www.niso.org/publications/rp/ Suggested Reference Chong, J., Karner, C.,
Drug prescriptions (Pharm) Exposure (36/48 months)
 ANNEX SECTION PART A - Study design: Figure 1 overview of the study design Drug prescriptions (Pharm) 2006-2010 Exposure (36/48 months) flexible time windows (e.g. 90 days) time index date (hospital discharge)
ANNEX SECTION PART A - Study design: Figure 1 overview of the study design Drug prescriptions (Pharm) 2006-2010 Exposure (36/48 months) flexible time windows (e.g. 90 days) time index date (hospital discharge)
C aring for patients with interstitial lung disease is an
 980 INTERSTITIAL LUNG DISEASE Incidence and mortality of idiopathic pulmonary fibrosis and sarcoidosis in the UK J Gribbin, R B Hubbard, I Le Jeune, C J P Smith, J West, L J Tata... See end of article
980 INTERSTITIAL LUNG DISEASE Incidence and mortality of idiopathic pulmonary fibrosis and sarcoidosis in the UK J Gribbin, R B Hubbard, I Le Jeune, C J P Smith, J West, L J Tata... See end of article
ESM1 for Glucose, blood pressure and cholesterol levels and their relationships to clinical outcomes in type 2 diabetes: a retrospective cohort study
 ESM1 for Glucose, blood pressure and cholesterol levels and their relationships to clinical outcomes in type 2 diabetes: a retrospective cohort study Statistical modelling details We used Cox proportional-hazards
ESM1 for Glucose, blood pressure and cholesterol levels and their relationships to clinical outcomes in type 2 diabetes: a retrospective cohort study Statistical modelling details We used Cox proportional-hazards
Drug Class Monograph
 Drug Class Monograph Class: Inhaled Corticosteroids Drugs: Aerospan (flunisolide), Advair Diskus, Advair HFA (fluticasone/salmeterol), Alvesco (ciclesonide), Arnuity Ellipta (fluticasone furoate), Asmanex
Drug Class Monograph Class: Inhaled Corticosteroids Drugs: Aerospan (flunisolide), Advair Diskus, Advair HFA (fluticasone/salmeterol), Alvesco (ciclesonide), Arnuity Ellipta (fluticasone furoate), Asmanex
KewKM,KarnerC,MindusSM,FerraraG. Cochrane Database of Systematic Reviews.
 Cochrane Database of Systematic Reviews Combination formoterol and budesonide as maintenance and reliever therapy versus combination inhaler maintenance for chronic asthma in adults and children(review)
Cochrane Database of Systematic Reviews Combination formoterol and budesonide as maintenance and reliever therapy versus combination inhaler maintenance for chronic asthma in adults and children(review)
RightBreathe to help with inhaler prescribing and technique
 www.rightbreathe.com RightBreathe to help with inhaler prescribing and technique Dr Azhar Saleem GP Respiratory Lead, Lambeth CCG GP Specialist NHS London Procurement Partnership Clinical Lead RightBreathe
www.rightbreathe.com RightBreathe to help with inhaler prescribing and technique Dr Azhar Saleem GP Respiratory Lead, Lambeth CCG GP Specialist NHS London Procurement Partnership Clinical Lead RightBreathe
April 10 th, Bond Street, Toronto ON, M5B 1W8
 Comprehensive Research Plan: Inhaled long-acting muscarinic antagonists (LAMAs; long-acting anticholinergics) for the treatment of chronic obstructive pulmonary disease (COPD) April 10 th, 2014 30 Bond
Comprehensive Research Plan: Inhaled long-acting muscarinic antagonists (LAMAs; long-acting anticholinergics) for the treatment of chronic obstructive pulmonary disease (COPD) April 10 th, 2014 30 Bond
Roflumilast (Daxas) for chronic obstructive pulmonary disease
 Roflumilast (Daxas) for chronic obstructive pulmonary disease August 2009 This technology summary is based on information available at the time of research and a limited literature search. It is not intended
Roflumilast (Daxas) for chronic obstructive pulmonary disease August 2009 This technology summary is based on information available at the time of research and a limited literature search. It is not intended
Algorithm for the use of inhaled therapies in COPD Version 2 May 2017
 Algorithm for the use of inhaled therapies in COPD This document has been revised by the Berkshire West Respiratory Network to support clinicians in selecting the most appropriate, cost effective treatments
Algorithm for the use of inhaled therapies in COPD This document has been revised by the Berkshire West Respiratory Network to support clinicians in selecting the most appropriate, cost effective treatments
GSK Medicine: Study Number: Title: Rationale: Study Period: Objectives: Indication: Study Investigators/Centers: Research Methods: Data Source
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The impacts of cognitive impairment on acute exacerbations of chronic obstructive pulmonary disease
 The impacts of cognitive impairment on acute exacerbations of chronic obstructive pulmonary disease Dr. Lo Iek Long Department of Respiratory Medicine C.H.C.S.J. Chronic Obstructive Pulmonary Disease (COPD)
The impacts of cognitive impairment on acute exacerbations of chronic obstructive pulmonary disease Dr. Lo Iek Long Department of Respiratory Medicine C.H.C.S.J. Chronic Obstructive Pulmonary Disease (COPD)
Decline In lung-function Among Patients with chronic obstructive Lung disease On maintenance therapy (DIAPLO)
 Version V1.4 Decline In lung-function Among Patients with chronic obstructive Lung disease On maintenance therapy (DIAPLO) An observational study evaluating the benefits of early intervention with maintenance
Version V1.4 Decline In lung-function Among Patients with chronic obstructive Lung disease On maintenance therapy (DIAPLO) An observational study evaluating the benefits of early intervention with maintenance
Algorithm for the use of inhaled therapies in COPD
 Berkshire West Integrated Care System Representing Berkshire West Clinical Commisioning Group Royal Berkshire NHS Foundation Trust Berkshire Healthcare NHS Foundation Trust Berkshire West Primary Care
Berkshire West Integrated Care System Representing Berkshire West Clinical Commisioning Group Royal Berkshire NHS Foundation Trust Berkshire Healthcare NHS Foundation Trust Berkshire West Primary Care
Long Term Care Formulary RS -29
 RESTRICTED USE Asthma/COPD Management 1 of 6 PROTOCOL: Asthma Glossary of Medication Acronyms: SABA: short-acting beta agonist (e.g. salbutamol) SABD: short-acting bronchodilator (e.g. ipratropium or SABA)
RESTRICTED USE Asthma/COPD Management 1 of 6 PROTOCOL: Asthma Glossary of Medication Acronyms: SABA: short-acting beta agonist (e.g. salbutamol) SABD: short-acting bronchodilator (e.g. ipratropium or SABA)
Proposal on subsidy changes for some respiratory inhalation products and access restrictions to combination inhalers.
 30 August 2011 Proposal on changes for some respiratory products and access restrictions to combination inhalers. As notified on 16 June 2011, PHARMAC has been considering options for managing the funding
30 August 2011 Proposal on changes for some respiratory products and access restrictions to combination inhalers. As notified on 16 June 2011, PHARMAC has been considering options for managing the funding
ASTHMA TREATMENT GUIDE (ADULTS)
 ASTHMA TREATMENT GUIDE (ADULTS) The BTS/SIGN guideline provides a wide range of information and guidance on the treatment of patients with asthma. https://www.brit-thoracic.org.uk/document-library/clinical-information/asthma/btssign-asthma-guideline-2016/
ASTHMA TREATMENT GUIDE (ADULTS) The BTS/SIGN guideline provides a wide range of information and guidance on the treatment of patients with asthma. https://www.brit-thoracic.org.uk/document-library/clinical-information/asthma/btssign-asthma-guideline-2016/
Study No.: Title: Rationale: Phase: Study Period: Study Design: Centres: Indication: Treatment: Objectives: Primary Outcome/Efficacy Variable:
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
fluticasone furoate / vilanterol 92/22, 184/22 micrograms inhalation powder (Relvar Ellipta ) SMC No. (966/14) GlaxoSmithKline UK
 fluticasone furoate / vilanterol 92/22, 184/22 micrograms inhalation powder (Relvar Ellipta ) SMC No. (966/14) GlaxoSmithKline UK 09 May 2014 The Scottish Medicines Consortium (SMC) has completed its assessment
fluticasone furoate / vilanterol 92/22, 184/22 micrograms inhalation powder (Relvar Ellipta ) SMC No. (966/14) GlaxoSmithKline UK 09 May 2014 The Scottish Medicines Consortium (SMC) has completed its assessment
Disclosure and Conflict of Interest 8/15/2017. Pharmacist Objectives. At the conclusion of this program, the pharmacist will be able to:
 Digging for GOLD Rebecca Young, PharmD, BCACP, Roosevelt University College of Pharmacy Assistant Professor of Clinical Sciences Practice Site Advocate Medical Group-Nesset Pavilion Disclosure and Conflict
Digging for GOLD Rebecca Young, PharmD, BCACP, Roosevelt University College of Pharmacy Assistant Professor of Clinical Sciences Practice Site Advocate Medical Group-Nesset Pavilion Disclosure and Conflict
Patient characteristics Intervention Comparison Length of followup
 ORAL MUCOLYTICS Ref ID: 2511 Bachh AA, Shah NN, Bhargava R et al. Effect oral N- in COPD - A randomised controlled trial. JK Practitioner. 2007; 14(1):12-16. Ref ID: 2511 RCT Single blind; unclear allocation
ORAL MUCOLYTICS Ref ID: 2511 Bachh AA, Shah NN, Bhargava R et al. Effect oral N- in COPD - A randomised controlled trial. JK Practitioner. 2007; 14(1):12-16. Ref ID: 2511 RCT Single blind; unclear allocation
Global Initiative for Asthma (GINA) What s new in GINA 2016?
 Global Initiative for Asthma (GINA) What s new in GINA 2016? GINA Global Strategy for Asthma Management and Prevention GINA: A Brief History Established in 1993 Collaboration between NHLBI and WHO Multiple
Global Initiative for Asthma (GINA) What s new in GINA 2016? GINA Global Strategy for Asthma Management and Prevention GINA: A Brief History Established in 1993 Collaboration between NHLBI and WHO Multiple
Optimum COPD Care in 2010 Why Not Now? David E. Taylor, M.D. Pulmonary/Critical Care Ochnser Medical Center
 Optimum COPD Care in 2010 Why Not Now? David E. Taylor, M.D. Pulmonary/Critical Care Ochnser Medical Center dtaylor@ochsner.org Observations from Yesterday EPIC is epidemic No EMR No Way!!! Accountability/Benchmarking
Optimum COPD Care in 2010 Why Not Now? David E. Taylor, M.D. Pulmonary/Critical Care Ochnser Medical Center dtaylor@ochsner.org Observations from Yesterday EPIC is epidemic No EMR No Way!!! Accountability/Benchmarking
LAMA Products for the Treatment of COPD
 FINAL REPORT LAMA Products for the Treatment of COPD Pharmacoepidemiology Unit: Censored Final Report Tara Gomes, Andrea Gershon, Matthew Stanbrook, Ximena Camacho, Diana Martins, Samantha Singh, Zhan
FINAL REPORT LAMA Products for the Treatment of COPD Pharmacoepidemiology Unit: Censored Final Report Tara Gomes, Andrea Gershon, Matthew Stanbrook, Ximena Camacho, Diana Martins, Samantha Singh, Zhan
Medicines Optimisation Team Standard Operating Procedure for Audit: High Dose Inhaled Corticosteroids
 Cannock Chase Clinical Commissioning Group South East Staffordshire and Seisdon Peninsula Clinical Commissioning Group Stafford and Surrounds Clinical Commissioning Group East Staffordshire Clinical Commissioning
Cannock Chase Clinical Commissioning Group South East Staffordshire and Seisdon Peninsula Clinical Commissioning Group Stafford and Surrounds Clinical Commissioning Group East Staffordshire Clinical Commissioning
Peak Expiratory Flow Rate (PEFR) for ED Management of Acute Asthma Exacerbation
 Peak Expiratory Flow Rate (PEFR) for ED Management of Acute Asthma Exacerbation PI: Brian Driver, MD Checklist Reviewed Inclusion and Exclusion Criteria Confirm pertinent exclusion criteria with PMP Engage
Peak Expiratory Flow Rate (PEFR) for ED Management of Acute Asthma Exacerbation PI: Brian Driver, MD Checklist Reviewed Inclusion and Exclusion Criteria Confirm pertinent exclusion criteria with PMP Engage
at a younger age than white populations,
 Research Alice Martin, Ellena Badrick, Rohini Mathur and Sally Hull Effect of ethnicity on the prevalence, severity, and management of COPD in general practice Abstract Background Chronic obstructive pulmonary
Research Alice Martin, Ellena Badrick, Rohini Mathur and Sally Hull Effect of ethnicity on the prevalence, severity, and management of COPD in general practice Abstract Background Chronic obstructive pulmonary
Secondary Outcome/Efficacy Variable(s):
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
(PLACE PATIENT LABEL HERE) Date: Time: Assessment nurse: Sign: STOP!
 ASTHMA BEST CARE BUNDLE P A T H W A Y ADULT ASTHMA Date: Time: Assessment nurse: Sign: INCLUSION CRITERIA Known asthmatic Shortness of breath and / or wheeze EXCLUSION CRITERIA Chronic lung disease other
ASTHMA BEST CARE BUNDLE P A T H W A Y ADULT ASTHMA Date: Time: Assessment nurse: Sign: INCLUSION CRITERIA Known asthmatic Shortness of breath and / or wheeze EXCLUSION CRITERIA Chronic lung disease other
Study Exposures, Outcomes:
 GSK Medicine: Coreg IR, Coreg CR, and InnoPran Study No.: WWE111944/WEUSRTP3149 Title: A nested case-control study of the association between Coreg IR and Coreg CR and hypersensitivity reactions: anaphylactic
GSK Medicine: Coreg IR, Coreg CR, and InnoPran Study No.: WWE111944/WEUSRTP3149 Title: A nested case-control study of the association between Coreg IR and Coreg CR and hypersensitivity reactions: anaphylactic
CHARM ASTHMA TREATMENT GUIDELINE
 NHS City and Hackney Prescribing Guidelines Adults ( 12 years of age) CHARM ASTHMA TREATMENT GUIDELINE Written by: Hetal Dhruve (Specialist Respiratory Pharmacist, City and Hackney CCG) Checked by: Prof
NHS City and Hackney Prescribing Guidelines Adults ( 12 years of age) CHARM ASTHMA TREATMENT GUIDELINE Written by: Hetal Dhruve (Specialist Respiratory Pharmacist, City and Hackney CCG) Checked by: Prof
NIH Public Access Author Manuscript J Asthma. Author manuscript; available in PMC 2013 June 07.
 NIH Public Access Author Manuscript Published in final edited form as: J Asthma. 2012 June ; 49(5): 450 455. doi:10.3109/02770903.2012.677894. Long-Acting Beta-Agonists And The Risk of Intensive Care Unit
NIH Public Access Author Manuscript Published in final edited form as: J Asthma. 2012 June ; 49(5): 450 455. doi:10.3109/02770903.2012.677894. Long-Acting Beta-Agonists And The Risk of Intensive Care Unit
glycopyrronium 44 micrograms hard capsules of inhalation powder (Seebri Breezhaler ) SMC No. (829/12) Novartis Pharmaceuticals Ltd.
 glycopyrronium 44 micrograms hard capsules of inhalation powder (Seebri Breezhaler ) SMC No. (829/12) Novartis Pharmaceuticals Ltd. 07 December 2012 The Scottish Medicines Consortium (SMC) has completed
glycopyrronium 44 micrograms hard capsules of inhalation powder (Seebri Breezhaler ) SMC No. (829/12) Novartis Pharmaceuticals Ltd. 07 December 2012 The Scottish Medicines Consortium (SMC) has completed
Scottish Medicines Consortium
 Scottish Medicines Consortium montelukast 10mg tablets (Singulair ) No. (185/05) Merck, Sharp & Dohme Ltd (MSD) New indication: for asthmatic patients in whom montelukast is indicated in asthma, montelukast
Scottish Medicines Consortium montelukast 10mg tablets (Singulair ) No. (185/05) Merck, Sharp & Dohme Ltd (MSD) New indication: for asthmatic patients in whom montelukast is indicated in asthma, montelukast
Rapid Effects of Inhaled Corticosteroids in Acute Asthma Gustavo J. Rodrigo, MD.
 Rapid Effects of Inhaled Corticosteroids in Acute Asthma Gustavo J. Rodrigo, MD WWW.chestjournal.org Background: Current reviews on the use of inhaled corticosteroids (ICS) for acute asthma underestimated
Rapid Effects of Inhaled Corticosteroids in Acute Asthma Gustavo J. Rodrigo, MD WWW.chestjournal.org Background: Current reviews on the use of inhaled corticosteroids (ICS) for acute asthma underestimated
aclidinium 322 micrograms inhalation powder (Eklira Genuair ) SMC No. (810/12) Almirall S.A.
 aclidinium 322 micrograms inhalation powder (Eklira Genuair ) SMC No. (810/12) Almirall S.A. 05 October 2012 The Scottish Medicines Consortium (SMC) has completed its assessment of the above product and
aclidinium 322 micrograms inhalation powder (Eklira Genuair ) SMC No. (810/12) Almirall S.A. 05 October 2012 The Scottish Medicines Consortium (SMC) has completed its assessment of the above product and
Lead team presentation: Roflumilast for treating chronic obstructive pulmonary disease [ID984]
![Lead team presentation: Roflumilast for treating chronic obstructive pulmonary disease [ID984] Lead team presentation: Roflumilast for treating chronic obstructive pulmonary disease [ID984]](/thumbs/85/91889043.jpg) Lead team presentation: Roflumilast for treating chronic obstructive pulmonary disease [ID984] 1 st Appraisal Committee meeting Background & Clinical Effectiveness John McMurray 11 th January 2016 For
Lead team presentation: Roflumilast for treating chronic obstructive pulmonary disease [ID984] 1 st Appraisal Committee meeting Background & Clinical Effectiveness John McMurray 11 th January 2016 For
Asthma Population Management: Identifying Persistent Asthma, Defining High Risk Asthma, and Measuring Quality of Asthma Care
 Asthma Population Management: Identifying Persistent Asthma, Defining High Risk Asthma, and Measuring Quality of Asthma Care Michael Schatz, MD, MS Allergy Department Kaiser Permanente, San Diego, CA Constructs
Asthma Population Management: Identifying Persistent Asthma, Defining High Risk Asthma, and Measuring Quality of Asthma Care Michael Schatz, MD, MS Allergy Department Kaiser Permanente, San Diego, CA Constructs
Interventions to improve adherence to inhaled steroids for asthma. Respiratory department
 Interventions to improve adherence to inhaled steroids for asthma Respiratory department Content Overview Research References Overview Asthma is a chronic breathing condition that affects more than 300
Interventions to improve adherence to inhaled steroids for asthma Respiratory department Content Overview Research References Overview Asthma is a chronic breathing condition that affects more than 300
NHS Dumfries & Galloway Triple therapy in COPD patients over 16 years
 Title of Project: NHS Dumfries & Galloway Triple therapy in COPD patients over 16 years 1 Reason for the review Respiratory prescribing is long term and can be costly. Appropriate choice and use of inhaled
Title of Project: NHS Dumfries & Galloway Triple therapy in COPD patients over 16 years 1 Reason for the review Respiratory prescribing is long term and can be costly. Appropriate choice and use of inhaled
Stepping-down combination ICS/LABA asthma inhaler therapy: Adults 18yrs
 Step Down guidance Stepping-down combination ICS/LABA asthma inhaler therapy: Adults 18yrs Important Complete asthma control needs to be achieved for at least 12 weeks before attempting to step patients
Step Down guidance Stepping-down combination ICS/LABA asthma inhaler therapy: Adults 18yrs Important Complete asthma control needs to be achieved for at least 12 weeks before attempting to step patients
Relvar Ellipta (fluticasone furoate/vilanterol [as trifenatate]) for the treatment of patients with asthma
![Relvar Ellipta (fluticasone furoate/vilanterol [as trifenatate]) for the treatment of patients with asthma Relvar Ellipta (fluticasone furoate/vilanterol [as trifenatate]) for the treatment of patients with asthma](/thumbs/89/99956827.jpg) Relvar Ellipta (fluticasone furoate/vilanterol [as trifenatate]) for the treatment of patients with asthma Evidence Summary to support formulary decision making and guideline development Prescribing and
Relvar Ellipta (fluticasone furoate/vilanterol [as trifenatate]) for the treatment of patients with asthma Evidence Summary to support formulary decision making and guideline development Prescribing and
Setting The setting was the community. The economic study was carried out in New Jersey, USA.
 Asthma rescue and allergy medication use among asthmatic children with prior allergy prescriptions who initiated asthma controller therapy Luskin A, Bukstein D, Kocevar V S, Yin D D Record Status This
Asthma rescue and allergy medication use among asthmatic children with prior allergy prescriptions who initiated asthma controller therapy Luskin A, Bukstein D, Kocevar V S, Yin D D Record Status This
ONLINE SUPPLEMENTARY MATERIAL
 Is chlamydia screening and testing in Britain reaching young adults at risk of infection? Findings from the third National Survey of Sexual ONLINE SUPPLEMENTARY MATERIAL Supplementary Figure S1: Flow chart
Is chlamydia screening and testing in Britain reaching young adults at risk of infection? Findings from the third National Survey of Sexual ONLINE SUPPLEMENTARY MATERIAL Supplementary Figure S1: Flow chart
REACH II: Stage 2. Final Report
 Study protocol: R02813 REACH II stage 2 31 st August 2017 Final Report REACH II: Stage 2 Examining real-life outcomes for UK patients with COPD after initiating Fostair pmdi according to its licensed indication
Study protocol: R02813 REACH II stage 2 31 st August 2017 Final Report REACH II: Stage 2 Examining real-life outcomes for UK patients with COPD after initiating Fostair pmdi according to its licensed indication
Study No.: SAM40012 Title: A multicentre, randomised, double-blind, double-dummy, parallel group comparison of three treatments : 1)
 Study No.: SAM40012 Title: A multicentre, randomised, double-blind, double-dummy, parallel group comparison of three treatments : 1) salmeterol/fluticasone propionate () (mcg strength) bd via DISKUS/ACCUHALER
Study No.: SAM40012 Title: A multicentre, randomised, double-blind, double-dummy, parallel group comparison of three treatments : 1) salmeterol/fluticasone propionate () (mcg strength) bd via DISKUS/ACCUHALER
Why Asthma Still Kills The National Review of Asthma Deaths (NRAD)
 APPROVED FINAL VERSION NHS Protect Why Asthma Still Kills The National Review of Asthma Deaths (NRAD) Summary of Recommendations for GP Practices and Community Pharmacies Author: Anne Henry Contact: anne.henry@nhs.net
APPROVED FINAL VERSION NHS Protect Why Asthma Still Kills The National Review of Asthma Deaths (NRAD) Summary of Recommendations for GP Practices and Community Pharmacies Author: Anne Henry Contact: anne.henry@nhs.net
THE COPD PRESCRIBING TOOL
 THE COPD PRESCRIBING TOOL Revised edition, 2017 www.bpac.org.nz/copd CLASSIFICATION The COPD prescribing tool This tool provides pharmacological treatment options for patients with COPD based on their
THE COPD PRESCRIBING TOOL Revised edition, 2017 www.bpac.org.nz/copd CLASSIFICATION The COPD prescribing tool This tool provides pharmacological treatment options for patients with COPD based on their
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
Cite this article as: BMJ, doi: /bmj f (published 23 December 2004) Bronchodilator treatment and deaths from asthma: case-control
 Cite this article as: BMJ, doi:10.1136/bmj.38316.729907.8f (published 23 December 2004) Bronchodilator treatment and deaths from asthma: case-control study H Ross Anderson, Jon G Ayres, Patricia M Sturdy,
Cite this article as: BMJ, doi:10.1136/bmj.38316.729907.8f (published 23 December 2004) Bronchodilator treatment and deaths from asthma: case-control study H Ross Anderson, Jon G Ayres, Patricia M Sturdy,
OPAT FOR INFECTION IN BRONCHIECTASIS
 OPAT FOR INFECTION IN BRONCHIECTASIS AN AUDIT EVALUATING THE USAGE OF OUTPATIENT ANTIBIOTIC THERAPY FOR INFECTIVE EXACERBATIONS OF BRONCHIECTASIS AGAINST CURRENT BRITISH THORACIC SOCIETY GUIDELINES Dr
OPAT FOR INFECTION IN BRONCHIECTASIS AN AUDIT EVALUATING THE USAGE OF OUTPATIENT ANTIBIOTIC THERAPY FOR INFECTIVE EXACERBATIONS OF BRONCHIECTASIS AGAINST CURRENT BRITISH THORACIC SOCIETY GUIDELINES Dr
Bronchodilator Delivery and Nebuliser Trials in Adults
 Bronchodilator Delivery and Nebuliser Trials in Adults Acute Management Favour the use of MDI (+/- Spacer) If considering nebuliser Short term treatment Approx. < 3 weeks See optimisation of inhaled bronchodilators
Bronchodilator Delivery and Nebuliser Trials in Adults Acute Management Favour the use of MDI (+/- Spacer) If considering nebuliser Short term treatment Approx. < 3 weeks See optimisation of inhaled bronchodilators
This is the author s final accepted version.
 Smart, R., Carter, B., McGovern, J., Luckman, S., Connelly, A., Hewitt, J., Quasim, T. and Moug, S. (2017) Frailty exists in younger adults admitted as surgical emergency leading to adverse outcomes. Journal
Smart, R., Carter, B., McGovern, J., Luckman, S., Connelly, A., Hewitt, J., Quasim, T. and Moug, S. (2017) Frailty exists in younger adults admitted as surgical emergency leading to adverse outcomes. Journal
COPD: Current Medical Therapy
 COPD: Current Medical Therapy Angela Golden, DNP, FNP-C, FAANP Owner, NP from Home, LLC Outcomes As a result of this activity, learners will be able to: 1. List the appropriate classes of medications for
COPD: Current Medical Therapy Angela Golden, DNP, FNP-C, FAANP Owner, NP from Home, LLC Outcomes As a result of this activity, learners will be able to: 1. List the appropriate classes of medications for
Stopping long-acting beta 2 -agonists (LABA) for adults with asthma well controlled by LABA and inhaled corticosteroids (Review)
 Stopping long-acting beta 2 -agonists (LABA) for adults with asthma well controlled by LABA and inhaled corticosteroids (Review) Ahmad S, Kew KM, Normansell R This is a reprint of a Cochrane review, prepared
Stopping long-acting beta 2 -agonists (LABA) for adults with asthma well controlled by LABA and inhaled corticosteroids (Review) Ahmad S, Kew KM, Normansell R This is a reprint of a Cochrane review, prepared
THIS TALK STATINS REDUCE: Immortal time bias Immeasurable time bias Time-window bias TIME-RELATED BIASES IN PHARMACOEPIDEMIOLOGY
 TIME-RELATED BIASES IN PHARMACOEPIDEMIOLOGY Samy Suissa McGill University and Jewish General Hospital Montreal, Canada ISPE mid-year meeting, Miami April 22, 2012 STATINS REDUCE: JAMA 2000: Fracture rate
TIME-RELATED BIASES IN PHARMACOEPIDEMIOLOGY Samy Suissa McGill University and Jewish General Hospital Montreal, Canada ISPE mid-year meeting, Miami April 22, 2012 STATINS REDUCE: JAMA 2000: Fracture rate
Statistical Analysis Plan
 Drug utilisation study of new users of fluticasone furoate / vilanterol (FF/VI) in the primary care setting: UK Clinical Practice Research Datalink (CPRD) study Statistical Analysis Plan Subject: 205052:
Drug utilisation study of new users of fluticasone furoate / vilanterol (FF/VI) in the primary care setting: UK Clinical Practice Research Datalink (CPRD) study Statistical Analysis Plan Subject: 205052:
Up in FLAMES: Stable Chronic Obstructive Pulmonary Disease (COPD) Management. Colleen Sakon, PharmD BCPS September 27, 2018
 Up in FLAMES: Stable Chronic Obstructive Pulmonary Disease (COPD) Management Colleen Sakon, PharmD BCPS September 27, 2018 Disclosures I have no actual or potential conflicts of interest 2 Objectives Summarize
Up in FLAMES: Stable Chronic Obstructive Pulmonary Disease (COPD) Management Colleen Sakon, PharmD BCPS September 27, 2018 Disclosures I have no actual or potential conflicts of interest 2 Objectives Summarize
The National Heart Failure Audit 2010/2011
 The National Heart Failure Audit 2010/2011 Project Steering Group; TA McDonagh, JG Cleland, HJ Dargie, S Hardman,P Mitchell, A Cunningham 85% NHS Trusts submitting data (133/156) 36,504 admissions 70%
The National Heart Failure Audit 2010/2011 Project Steering Group; TA McDonagh, JG Cleland, HJ Dargie, S Hardman,P Mitchell, A Cunningham 85% NHS Trusts submitting data (133/156) 36,504 admissions 70%
your breathing problems worsen quickly. you use your rescue inhaler, but it does not relieve your breathing problems.
 MEDICATION GUIDE ADVAIR DISKUS [ad vair disk us] (fluticasone propionate and salmeterol inhalation powder) for oral inhalation What is the most important information I should know about ADVAIR DISKUS?
MEDICATION GUIDE ADVAIR DISKUS [ad vair disk us] (fluticasone propionate and salmeterol inhalation powder) for oral inhalation What is the most important information I should know about ADVAIR DISKUS?
Aclidinium bromide/formoterol Endpoint category. RR Endpoint. [95% CI 2 ] Study. with event
![Aclidinium bromide/formoterol Endpoint category. RR Endpoint. [95% CI 2 ] Study. with event Aclidinium bromide/formoterol Endpoint category. RR Endpoint. [95% CI 2 ] Study. with event](/thumbs/90/102875145.jpg) Resolution by the Federal Joint Committee on an amendment to the Pharmaceutical Directive (AM-RL): Appendix XII Resolutions on the benefit assessment of pharmaceuticals with new active ingredients, in
Resolution by the Federal Joint Committee on an amendment to the Pharmaceutical Directive (AM-RL): Appendix XII Resolutions on the benefit assessment of pharmaceuticals with new active ingredients, in
The clinical effectiveness and costeffectiveness. treatment of chronic asthma in children under the age of 12 years
 The clinical effectiveness and costeffectiveness of corticosteroids for the treatment of chronic asthma in children under the age of 12 years Submission of evidence from AstraZeneca UK Ltd regarding the
The clinical effectiveness and costeffectiveness of corticosteroids for the treatment of chronic asthma in children under the age of 12 years Submission of evidence from AstraZeneca UK Ltd regarding the
TORCH: Salmeterol and Fluticasone Propionate and Survival in COPD
 TORCH: and Propionate and Survival in COPD April 19, 2007 Justin Lee Pharmacy Resident University Health Network Outline Overview of COPD Pathophysiology Pharmacological Treatment Overview of the TORCH
TORCH: and Propionate and Survival in COPD April 19, 2007 Justin Lee Pharmacy Resident University Health Network Outline Overview of COPD Pathophysiology Pharmacological Treatment Overview of the TORCH
Pharmacotherapy for COPD
 10/3/2017 Topics to be covered Pharmacotherapy for chronic treatment Pharmacotherapy for COPD Dr. W C Yu 3rd September 2017 Commonly used drugs Guidelines for their use Inhaled corticosteroids (ICS) in
10/3/2017 Topics to be covered Pharmacotherapy for chronic treatment Pharmacotherapy for COPD Dr. W C Yu 3rd September 2017 Commonly used drugs Guidelines for their use Inhaled corticosteroids (ICS) in
ASTHMA & RESPIRATORY FOUNDATION NZ ADULT ASTHMA GUIDELINES: A QUICK REFERENCE GUIDE 1
 ASTHMA & RESPIRATORY FOUNDATION NZ ADULT ASTHMA GUIDELINES: A QUICK REFERENCE GUIDE 1 1. Richard Beasley, Bob Hancox, Matire Harwood, Kyle Perrin, Betty Poot, Janine Pilcher, Jim Reid, Api Talemaitoga,
ASTHMA & RESPIRATORY FOUNDATION NZ ADULT ASTHMA GUIDELINES: A QUICK REFERENCE GUIDE 1 1. Richard Beasley, Bob Hancox, Matire Harwood, Kyle Perrin, Betty Poot, Janine Pilcher, Jim Reid, Api Talemaitoga,
RESEARCH. Predicting risk of type 2 diabetes in England and Wales: prospective derivation and validation of QDScore
 Predicting risk of type 2 diabetes in England and Wales: prospective derivation and validation of QDScore Julia Hippisley-Cox, professor of clinical epidemiology and general practice, 1 Carol Coupland,
Predicting risk of type 2 diabetes in England and Wales: prospective derivation and validation of QDScore Julia Hippisley-Cox, professor of clinical epidemiology and general practice, 1 Carol Coupland,
WEBINAR. Difficult-to-treat and severe asthma: changing the paradigm
 WEBINAR Difficult-to-treat and severe asthma: changing the paradigm A multidisciplinary discussion on new therapies, and how to identify and manage difficult-to-treat and severe asthma DIFFICULT-TO-TREAT
WEBINAR Difficult-to-treat and severe asthma: changing the paradigm A multidisciplinary discussion on new therapies, and how to identify and manage difficult-to-treat and severe asthma DIFFICULT-TO-TREAT
T he effect of systemic corticosteroid use on the risk of
 1247 EXTENDED REPORT A population based case-control study of cataract and inhaled corticosteroids L Smeeth, M Boulis, R Hubbard, A E Fletcher... See end of article for authors affiliations... Correspondence
1247 EXTENDED REPORT A population based case-control study of cataract and inhaled corticosteroids L Smeeth, M Boulis, R Hubbard, A E Fletcher... See end of article for authors affiliations... Correspondence
