Drug Use Evaluation: Low Dose Quetiapine
|
|
|
- Arnold Goodwin
- 5 years ago
- Views:
Transcription
1 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon Phone Fax Drug Use Evaluation: Low Dose Quetiapine Research Questions: Does the current safety edit appropriately prevent prescribing in at risk populations (very old or very young) for off-label uses? Conclusions: The majority of low-dose quetiapine claims (85%, n=3482) were for adult patients years of age. There were 324 patients (7.9%) with claims for low-dose quetiapine who were less than 18 years of age and 292 patients (7.1%) over 60 years of age with low-dose quetiapine claims. Eighty-five percent of patients who met criteria for the safety edit had a paid claim for low-dose quetiapine within 90 days. Fifteen percent of patients (n=621) had an initial denied claim without subsequent follow-up. The majority of patients without a paid low-dose quetiapine claim had denied claims with a daily dose less than 50 mg (68%) or did not have an FDAindicated diagnosis (65%). Recommendations: Recommend modification of safety edit to apply to only patients with a daily dose less than 50 mg (see Appendix 1). Background: Quetiapine is a second generation antipsychotic which is been approved by the Food and Drug Administration (FDA) for bipolar disorder, schizophrenia and as adjunct treatment for depression. 1 Doses for these conditions typically range from 300 mg to 800 mg daily. Low-dose quetiapine (<150 mg) may be used offlabel for a variety of conditions including insomnia, anxiety, obsessive compulsive disorder and dementia. 1 However, quetiapine has been associated with significant safety concerns including increased mortality in elderly patients with dementia-related psychosis and suicidal thoughts and behaviors in children and adolescents. 1 A drug use evaluation in 2010 demonstrated that a significant proportion of the Oregon Health Plan (OHP) population (~56%) had claims for low-dose quetiapine without an FDA approved diagnosis. 2 In January 2011, a safety edit for any claims less than 150 mg was implemented to discourage off-label use. 2 Patients with diagnoses of schizophrenia, bipolar disorder, history for use for a second generation antipsychotic, or prescriptions written by a mental health specialist could be automatically approved. In the past few years, the majority of requests submitted for low-dose quetiapine have been subsequently approved. This brief drug use evaluation reassesses need for the low-dose quetiapine safety edit and explores how criteria can be modified to more appropriately target at risk populations. Methods: The patient population included current Medicaid patients with a fee for service (FFS) claim for quetiapine in one year period from 7/1/2016 to 06/30/2017. The index event was defined as the first paid or denied claim for quetiapine in the reporting period prescribed at a daily dose of 150 mg or less. Patients were stratified by age, quetiapine dose, and diagnosis. Patients with diagnoses for schizophrenia, bipolar disorders, and major depressive disorder within the last year Author: Sarah Servid, PharmD Date: November 2017
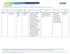
2 were identified based on ICD-10 diagnosis codes (Appendix 2). Denied claim were defined as claims with Explanation of Benefit (EOB) codes of 1056 PA required, 4506 non-preferred drug, 0030 Quantity per day limit exceeded or 0148 Quantity dispensed exceed max allowed without any of the following EOB codes: 0154 Bill part D plan, 1109 drug covered by Medicare Part D, 0003 recipient not eligible, 91 non-covered service, 1048 prescribing provider not on file. Patients with claims for concurrent antidepressants, mood stabilizers (lithium or divalproex), or history of claims for other second generation antipsychotics were also identified. Patients were excluded if they had Medicare part D coverage or had Medicaid eligibility for less than 75% of time in the year prior to the index event. Results: Results for patients with paid and denied claims are reported in Table 1. Over the course of the year, approximately 4100 patients had claims for low-dose quetiapine. The majority of these claims (85%, n=3482) were for patients years of age. Eighty-five percent of patients who met criteria for the safety edit had a paid claim within 90 days. Only 1 patient had a request which was submitted and denied, but 15% of patients (n=621) had an initial denied claim without subsequent follow-up. Of these patients without follow-up, the majority were for a daily dose of 50 mg or less (68% of patients) or for patients without an FDA indicated diagnosis (65% of patients). Because data are based on a retrospective analysis of claims history, diagnoses may not accurately reflect true patient diagnoses or correlate with actual medication adherence. Of the patients with no subsequent claim within 90 days, 45 patients (7%) had other insurance on file, which may explain the lack of follow-up for these particular patients. Claim initially paid OR paid within 1 day of denial Claim paid within 2-30 days of the initial denial Claim paid within days of the initial denial Request submitted and denied No request submitted AND no paid claim within 90 days of the initial denial* N % N % N % N % N % N Total 2,777 68% % 151 4% 1 0% % 4,098 Age % 5 11% 3 7% 5 11% % 35 13% 11 4% 27 10% ,344 67% % 135 4% 1 0% % 3, % 33 11% 2 1% 62 21% 292 Dose 50 mg/day % % 82 5% % 1, mg/day 1,056 73% % 52 4% 1 0% % 1, mg/day % 68 8% 17 2% 28 3% 863 Diagnoses % 13 6% 4 2% 9 4% 215 Bipolar Disorder % % 30 3% 84 8% 1,055 MDD diagnoses AND a claim for an antidepressant within % % 52 4% % 1, days before the index event Pts with a claim for lithium or divalproex (HSNs , % 47 12% 10 3% 29 7% , or ) within 45 days before the index event Pts with a claim for a second generation antipsychotic the 6 months prior to the index event 2,702 99% 18 1% 4 0% 7 0% 2,731 Total
3 None of the above 37 5% % 69 9% 1 0% % 750 *Patients are only included in this category if they had continuous enrollment 90 days post index event. Table 1. Status for patients with an index event in the last year. Diagnoses are not mutually exclusive. References: 1. Micromedex Healthcare Series [internet database]. Greenwood Village, CO: Truven Health Analytics, Inc. Updated periodically. Accessed October 23, Meeker A. Oregon Medicaid FFS Drug Class List. Policy Evaluation: Low Dose Quetiapine Safety Edit. September 2015; Accessed October 23, Appendix 1. Proposed Safety Edit Low Dose Quetiapine Goal(s): To promote and ensure use of quetiapine that is supported by the medical literature. To discourage off-label use for insomnia. Promote the use of non-pharmacologic alternatives for chronic insomnia. Initiative: Low dose quetiapine (Seroquel and Seroquel XR ) Length of Authorization: Up to 12 months (criteria-specific) Requires PA: Quetiapine (HSN = 14015) doses <150 mg/day Auto PA approvals for : o Patients with a claim for a second generation antipsychotic in the last 6 months o Patients with prior claims evidence of schizophrenia or bipolar disorder o Prescriptions identified as being written by a mental health provider Covered Alternatives:
4 Preferred alternatives listed at Zolpidem and benzodiazepine sedatives areis available for short-term use (15 doses/30 days) without PA. Table 1. Adult (age 18 years) FDA-approved Indications for Quetiapine Bipolar Disorder Major Depressive Disorder Bipolar Mania Bipolar Depression F3010; F302; F3160-F3164; F ; F319 F ; F ; F329; F ; F339; F3130 F205; F209; F2081; F2089 F3010; F339; F3110-F3113; F312 F3130 For Seroquel XR only, Adjunctive therapy with antidepressants for Major Depressive Disorder Table 2. Pediatric FDA-approved indications Bipolar Mania Adolescents (13-17 years) Children and Adolescents (10 to 17 years) Monotherapy Approval Criteria 1. What diagnosis is being treated? Record ICD10 code. Do not proceed and deny if diagnosis is not listed in Table 1 or Table 2 above (medical appropriateness) 2. Is the prescription for quetiapine less than 150 mg/day? (verify days supply is accurate) Yes: Go to #3 No: Trouble-shoot claim processing with the pharmacy. 3. Is planned duration of therapy longer than 90 days? Yes: Go to #4 No: Approve for titration up to maintenance dose (60 days).
5 Approval Criteria 4. Is reason for dose <150 mg/day due to any of the following: low dose needed due to debilitation from a medical condition or age; unable to tolerate higher doses; stable on current dose; or impaired drug clearance? any diagnosis in table 1 or 2 above? Yes: Approve for up to 12 months No: Pass to RPh. Deny for medical appropriateness. Note: may approve up to 6 months to allow taper. P&T/DUR Review: 9/15 (KK); 9/10; 5/10 Implementation: 10/15; 1/1/11 Appendix 2. Quetiapine GSN codes and diagnosis codes GSN Generic QUETIAPINE FUMARATE QUETIAPINE FUMARATE QUETIAPINE FUMARATE QUETIAPINE FUMARATE QUETIAPINE FUMARATE QUETIAPINE FUMARATE QUETIAPINE FUMARATE QUETIAPINE FUMARATE QUETIAPINE FUMARATE QUETIAPINE FUMARATE QUETIAPINE FUMARATE QUETIAPINE FUMARATE Condition Bipolar Disorders MDD ICD-10 Diagnosis Code F205; F209; F2081; F2089 F3010; F302; F3160-F3164; F ; F319 ; F3110-F3113; F312 ; F3130 F ; F ; F329; F ; F339
Policy Evaluation: Low Dose Quetiapine Safety Edit
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University 500 Summer Street NE, E35, Salem, Oregon 97301 1079 Phone 503 947 5220 Fax 503
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University 500 Summer Street NE, E35, Salem, Oregon 97301 1079 Phone 503 947 5220 Fax 503
Rexulti (brexpiprazole)
 Market DC Rexulti (brexpiprazole) Override(s) Approval Duration Prior Authorization 1 year Quantity Limit *Indiana see State Specific Mandates below *Maryland see State Specific Mandates below *Virginia
Market DC Rexulti (brexpiprazole) Override(s) Approval Duration Prior Authorization 1 year Quantity Limit *Indiana see State Specific Mandates below *Maryland see State Specific Mandates below *Virginia
Prior Authorization Update: Nusinersen
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Pharmacy Medical Necessity Guidelines: Antipsychotic Medications
 Pharmacy Medical Necessity Guidelines: Antipsychotic Medications Effective: July. 1, 2016 Prior Authorization Required Type of Review Care Management Not Covered Type of Review Clinical Review Pharmacy
Pharmacy Medical Necessity Guidelines: Antipsychotic Medications Effective: July. 1, 2016 Prior Authorization Required Type of Review Care Management Not Covered Type of Review Clinical Review Pharmacy
Policy Evaluation: Substance Use Disorders
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
STEP THERAPY CRITERIA
 DRUG CLASS PRODUCTS) BRAND NAME (BRAND ONLY) (generic) STEP THERAPY CRITERIA ATYPICAL ANTIPSYCHOTICS (BRAND ONLY ABILIFY (AL TABLET & AL SOLUTION ONLY) (aripiprazole) FANAPT (BRAND ONLY) (iloperidone)
DRUG CLASS PRODUCTS) BRAND NAME (BRAND ONLY) (generic) STEP THERAPY CRITERIA ATYPICAL ANTIPSYCHOTICS (BRAND ONLY ABILIFY (AL TABLET & AL SOLUTION ONLY) (aripiprazole) FANAPT (BRAND ONLY) (iloperidone)
Prior Authorization Criteria Update: Androgens, Topical and Parenteral
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Antipsychotic Medications Age and Step Therapy
 Market DC *- Florida Healthy Kids Antipsychotic Medications Age and Step Therapy Override(s) Approval Duration Prior Authorization 1 year Quantity Limit *Virginia Medicaid See State Specific Mandates *Indiana
Market DC *- Florida Healthy Kids Antipsychotic Medications Age and Step Therapy Override(s) Approval Duration Prior Authorization 1 year Quantity Limit *Virginia Medicaid See State Specific Mandates *Indiana
Policy Evaluation: Step Therapy Prior Authorization of Combination Inhaled Corticosteroid / Long-Acting Beta-Agonists
 Drug Use Research & Management Program OHA Division of Medical Assistance Programs 500 Summer Street NE, E35; Salem, OR 97301-1079 Phone 503-947-5220 Fax 503-947-1119 Policy Evaluation: Step Therapy Prior
Drug Use Research & Management Program OHA Division of Medical Assistance Programs 500 Summer Street NE, E35; Salem, OR 97301-1079 Phone 503-947-5220 Fax 503-947-1119 Policy Evaluation: Step Therapy Prior
IOWA MEDICAID DRUG UTILIZATION REVIEW COMMISSION 100 Army Post Road (515)
 IOWA MEDICAID DRUG UTILIZATION REVIEW COMMISSION 100 Army Post Road (515) 974-3131 -866-626-0216 Brett Faine, Pharm.D. Larry Ambroson, R.Ph. Casey Clor, M.D. Professional Staff: Mark Graber, M.D., FACEP
IOWA MEDICAID DRUG UTILIZATION REVIEW COMMISSION 100 Army Post Road (515) 974-3131 -866-626-0216 Brett Faine, Pharm.D. Larry Ambroson, R.Ph. Casey Clor, M.D. Professional Staff: Mark Graber, M.D., FACEP
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: (Rexulti) Reference Number: CP.PMN.68 Effective Date: 11.05.15 Last Review Date: 02.18 Line of Business: Commercial, Health Insurance Marketplace, Medicaid Revision Log See Important Reminder
Clinical Policy: (Rexulti) Reference Number: CP.PMN.68 Effective Date: 11.05.15 Last Review Date: 02.18 Line of Business: Commercial, Health Insurance Marketplace, Medicaid Revision Log See Important Reminder
Table of Contents. 1.0 Policy Statement...1
 Division of Medical Assistance General Clinical Policy No. A-6 Table of Contents 1.0 Policy Statement...1 2.0 Policy Guidelines...1 2.1 Eligible Recipients...1 2.1.1 General Provisions...1 2.1.2 EPSDT
Division of Medical Assistance General Clinical Policy No. A-6 Table of Contents 1.0 Policy Statement...1 2.0 Policy Guidelines...1 2.1 Eligible Recipients...1 2.1.1 General Provisions...1 2.1.2 EPSDT
Author: K. Ketchum Date: July 2015
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
It is the policy of health plans affiliated with Centene Corporation that Seroquel XR is medically necessary when the following criteria are met:
 Clinical Policy: (Seroquel XR) Reference Number: CP.PMN.64 Effective Date: 12.01.14 Last Review Date: 02.18 Line of Business: Commercial, Health Insurance Marketplace, Medicaid Revision Log See Important
Clinical Policy: (Seroquel XR) Reference Number: CP.PMN.64 Effective Date: 12.01.14 Last Review Date: 02.18 Line of Business: Commercial, Health Insurance Marketplace, Medicaid Revision Log See Important
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: (Saphris) Reference Number: CP.PMN.15 Effective Date: 12.01.14 Last Review Date: 02.18 Line of Business: Commercial, Health Insurance Marketplace, Medicaid Revision Log See Important Reminder
Clinical Policy: (Saphris) Reference Number: CP.PMN.15 Effective Date: 12.01.14 Last Review Date: 02.18 Line of Business: Commercial, Health Insurance Marketplace, Medicaid Revision Log See Important Reminder
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: Reference Number: HIM.PA.59 Effective Date: 12/14 Last Review Date: 08/17 Line of Business: Health Insurance Marketplace Coding Implications Revision Log See Important Reminder at the
Clinical Policy: Reference Number: HIM.PA.59 Effective Date: 12/14 Last Review Date: 08/17 Line of Business: Health Insurance Marketplace Coding Implications Revision Log See Important Reminder at the
The Louis de la Parte Florida Mental Health Institute
 Data Brief December 2003 Mary Rose Murrin, M.A. Kelley Dhont, M.S. David Thornton, M.A. The Louis de la Parte Florida Mental Health Institute Children s Psychotropic Medication Use by Age and Diagnostic
Data Brief December 2003 Mary Rose Murrin, M.A. Kelley Dhont, M.S. David Thornton, M.A. The Louis de la Parte Florida Mental Health Institute Children s Psychotropic Medication Use by Age and Diagnostic
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: (Invega) Reference Number: CP. PMA.10.11.19 Effective Date: 10.06.16 Last Review Date: 04.18 Line of Business: CenpaticoMedicaid Revision Log See Important Reminder at the end of this
Clinical Policy: (Invega) Reference Number: CP. PMA.10.11.19 Effective Date: 10.06.16 Last Review Date: 04.18 Line of Business: CenpaticoMedicaid Revision Log See Important Reminder at the end of this
Maryland Department of Health and Mental Hygiene /Office of Systems, Operations and Pharmacy. Date Prescription will Start Denying at the
 Maryland Medicaid Pharmacy Program & News ViewsNovember 2013 Maryland Department of Health and Mental Hygiene /Office of Systems, Operations and Pharmacy The Antipsychotic Peer Review Program is Expanding!
Maryland Medicaid Pharmacy Program & News ViewsNovember 2013 Maryland Department of Health and Mental Hygiene /Office of Systems, Operations and Pharmacy The Antipsychotic Peer Review Program is Expanding!
Molina Healthcare of Texas
 Texas Standard Prior Authorization Form Addendum Molina Healthcare of Texas Ingrezza (Marketplace) This fax machine is located in a secure location as required by HIPAA Regulations. Complete / Review information,
Texas Standard Prior Authorization Form Addendum Molina Healthcare of Texas Ingrezza (Marketplace) This fax machine is located in a secure location as required by HIPAA Regulations. Complete / Review information,
Policy Evaluation: Proton Pump Inhibitors (PPIs)
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: (Rexulti) Reference Number: NE.PMN.68 Effective Date: 01/01/2017 Last Review Date: Line of Business: Medicaid Coding Implications Revision Log See Important Reminder at the end of this
Clinical Policy: (Rexulti) Reference Number: NE.PMN.68 Effective Date: 01/01/2017 Last Review Date: Line of Business: Medicaid Coding Implications Revision Log See Important Reminder at the end of this
Clinical Policy: Olanzapine Orally Disintegrating Tablet (Zyprexa Zydis) Reference Number: CP.PMN.29 Effective Date: Last Review Date: 02.
 Clinical Policy: (Zyprexa Zydis) Reference Number: CP.PMN.29 Effective Date: 08.01.15 Last Review Date: 02.19 Line of Business: Medicaid See Important Reminder at the end of this policy for important regulatory
Clinical Policy: (Zyprexa Zydis) Reference Number: CP.PMN.29 Effective Date: 08.01.15 Last Review Date: 02.19 Line of Business: Medicaid See Important Reminder at the end of this policy for important regulatory
Opioid Analgesics. Recommended starting dose for opioid-naïve patients
 Opioid Analgesics Goals: Restrict use of opioid analgesics to OHP-funded conditions with documented sustained improvement in pain and function and with routine monitoring for opioid misuse and abuse. Promote
Opioid Analgesics Goals: Restrict use of opioid analgesics to OHP-funded conditions with documented sustained improvement in pain and function and with routine monitoring for opioid misuse and abuse. Promote
Literature Scan: Parenteral Antipsychotics
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
2. Did the member receive this medication during a recent hospitalization? Y N
 Pharmacy Prior Authorization AETA BETTER HEALTH PESLVAIA & AETA BETTER HEALTH KIDS Antipsychotics (Medicaid) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review
Pharmacy Prior Authorization AETA BETTER HEALTH PESLVAIA & AETA BETTER HEALTH KIDS Antipsychotics (Medicaid) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review
2. Did the patient receive this medication during a recent hospitalization? Y N
 Pharmacy Prior Authorization AETA BETTER HEALTH PESLVAIA & AETA BETTER HEALTH KIDS Antipsychotics (Medicaid) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review
Pharmacy Prior Authorization AETA BETTER HEALTH PESLVAIA & AETA BETTER HEALTH KIDS Antipsychotics (Medicaid) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review
Criteria for Child Psychiatrist on the Use of Selected Psychotropic Medications in Children & Adolescents
 Criteria for Child Psychiatrist on the Use of Selected Psychotropic Medications in Children & Adolescents DRUG NAME INDICATIONS / ACCEPTABLE USES PRIOR STIMULANT/ADHD DRUGS Daytrana (methylphenidate) ADHD
Criteria for Child Psychiatrist on the Use of Selected Psychotropic Medications in Children & Adolescents DRUG NAME INDICATIONS / ACCEPTABLE USES PRIOR STIMULANT/ADHD DRUGS Daytrana (methylphenidate) ADHD
Idaho DUR Board Meeting Minutes. Committee Member Present: Matthew Hyde, Pharm.D., Perry Brown M.D., Paul Cady, Pharm.D., Ryan Heyborne M.D.
 Date: February 16, 2017 Time: 9am-12:15pm Idaho DUR Board Meeting Minutes Location: Idaho Medicaid, 3232 Elder Street, Boise, Idaho, Conference Room D-West Moderator: Perry Brown, M.D. Committee Member
Date: February 16, 2017 Time: 9am-12:15pm Idaho DUR Board Meeting Minutes Location: Idaho Medicaid, 3232 Elder Street, Boise, Idaho, Conference Room D-West Moderator: Perry Brown, M.D. Committee Member
CELEXA (CITALOPRAM) UTILIZATION AND DOSING MANAGEMENT
 BACKGROUND CELEXA (CITALOPRAM) UTILIZATION AND DOSING MANAGEMENT In May 2007, the FDA issued a notice that the agency was updating the black box warning for antidepressants to include warnings about increased
BACKGROUND CELEXA (CITALOPRAM) UTILIZATION AND DOSING MANAGEMENT In May 2007, the FDA issued a notice that the agency was updating the black box warning for antidepressants to include warnings about increased
2013 Virtual AD/HD Conference 1
 Medication for & Coexisting Conditions Part 2 Dr. Kenny Handelman Child, Adolescent & Adult Psychiatrist Halton Healthcare Adjunct Professor of Psychiatry, University of Western Ontario www.drkenny.com
Medication for & Coexisting Conditions Part 2 Dr. Kenny Handelman Child, Adolescent & Adult Psychiatrist Halton Healthcare Adjunct Professor of Psychiatry, University of Western Ontario www.drkenny.com
Note: Mandatory measures are those measures that are a requirement of accreditation and must be reported to URAC on an annual basis.
 Note: Mandatory measures are those measures that are a requirement of accreditation and must be reported to URAC on an annual basis. DESCRIPTION NUMERATOR DENOMINATOR DM2012-12 Portion of Days Covered
Note: Mandatory measures are those measures that are a requirement of accreditation and must be reported to URAC on an annual basis. DESCRIPTION NUMERATOR DENOMINATOR DM2012-12 Portion of Days Covered
Note: Mandatory measures are those measures that are a requirement of accreditation and must be reported to URAC on an annual basis.
 COMMUNITY PHARMACY V1.1 MANDATORY S Note: Mandatory measures are those measures that are a requirement of accreditation and must be reported to URAC on an annual basis. # NAME DESCRIPTION NUMERATOR DENOMINATOR
COMMUNITY PHARMACY V1.1 MANDATORY S Note: Mandatory measures are those measures that are a requirement of accreditation and must be reported to URAC on an annual basis. # NAME DESCRIPTION NUMERATOR DENOMINATOR
Pharmacy Medical Necessity Guidelines: Atypical Antipsychotic Medications. Effective: February 20, 2017
 Pharmacy Medical Necessity Guidelines: Effective: February 20, 2017 Prior Authorization Required Type of Review Care Management Not Covered Type of Review Clinical Review Pharmacy (RX) or Medical (MED)
Pharmacy Medical Necessity Guidelines: Effective: February 20, 2017 Prior Authorization Required Type of Review Care Management Not Covered Type of Review Clinical Review Pharmacy (RX) or Medical (MED)
OHSU Drug Effectiveness Review Project Summary Report Long-Acting Insulins
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-2596
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-2596
Pediatric Psychopharmacology
 Pediatric Psychopharmacology General issues to consider. Pharmacokinetic differences Availability of Clinical Data Psychiatric Disorders can be common in childhood. Early intervention may prevent disorders
Pediatric Psychopharmacology General issues to consider. Pharmacokinetic differences Availability of Clinical Data Psychiatric Disorders can be common in childhood. Early intervention may prevent disorders
3. Has the patient had a sustained improvement in Pain or Function (e.g. PEG scale with a 30 percent response from baseline)?
 Pharmacy Prior Authorization AETA BETTER HEALTH KETUCK Opioids Long-Acting and Short-Acting (Medicaid) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review
Pharmacy Prior Authorization AETA BETTER HEALTH KETUCK Opioids Long-Acting and Short-Acting (Medicaid) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review
A Retrospective Claims Analysis of Medication Adherence and. Persistence Among Patients Taking Antidepressants
 A Retrospective Claims Analysis of Medication Adherence and Persistence Among Patients Taking Antidepressants for the Treatment of Major Depressive Disorder (MDD) Katelyn R. Keyloun A thesis submitted
A Retrospective Claims Analysis of Medication Adherence and Persistence Among Patients Taking Antidepressants for the Treatment of Major Depressive Disorder (MDD) Katelyn R. Keyloun A thesis submitted
Pharmacy Medical Necessity Guidelines: Atypical Antipsychotic Medications. Effective: December 12, 2017
 Pharmacy Medical Necessity Guidelines: Effective: December 12, 2017 Prior Authorization Required Type of Review Care Management Not Covered Type of Review Clinical Review Pharmacy (RX) or Medical (MED)
Pharmacy Medical Necessity Guidelines: Effective: December 12, 2017 Prior Authorization Required Type of Review Care Management Not Covered Type of Review Clinical Review Pharmacy (RX) or Medical (MED)
be administered three times daily.
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use SEROQUEL safely and effectively. See full prescribing information for SEROQUEL. SEROQUEL (quetiapine
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use SEROQUEL safely and effectively. See full prescribing information for SEROQUEL. SEROQUEL (quetiapine
REXULTI (brexpiprazole) oral tablet
 REXULTI (brexpiprazole) oral tablet Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This Pharmacy
REXULTI (brexpiprazole) oral tablet Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This Pharmacy
Use of Anti-Psychotic Agents in Irish Long Term Care Residents with Dementia
 Use of Anti-Psychotic Agents in Irish Long Term Care Residents with Dementia Aine Leen, Kieran Walsh, David O Sullivan, Denis O Mahony, Stephen Byrne, Margaret Bermingham Pharmaceutical Care Research Group,
Use of Anti-Psychotic Agents in Irish Long Term Care Residents with Dementia Aine Leen, Kieran Walsh, David O Sullivan, Denis O Mahony, Stephen Byrne, Margaret Bermingham Pharmaceutical Care Research Group,
Hepatitis C Policy Discussion
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: (Invega Sustenna, Invega Trinza) Reference Number: CP.PHAR.291 Effective Date: 12.01.16 Last Review Date: 08.18 Line of Business: Medicaid See Important Reminder at the end of this policy
Clinical Policy: (Invega Sustenna, Invega Trinza) Reference Number: CP.PHAR.291 Effective Date: 12.01.16 Last Review Date: 08.18 Line of Business: Medicaid See Important Reminder at the end of this policy
Literature Scan: Erythropoiesis Stimulating Agents
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
MEDICAL ASSISTANCE BULLETIN
 ISSUE DATE January 6, 2016 SUBJECT EFFECTIVE DATE January 20, 2016 MEDICAL ASSISTANCE BULLETIN NUMBER *See below BY Prior Authorization of COPD Agents Pharmacy Service Leesa M. Allen, Deputy Secretary
ISSUE DATE January 6, 2016 SUBJECT EFFECTIVE DATE January 20, 2016 MEDICAL ASSISTANCE BULLETIN NUMBER *See below BY Prior Authorization of COPD Agents Pharmacy Service Leesa M. Allen, Deputy Secretary
Page 1 of RECENT MAJOR CHANGES Warnings and Precautions, Falls (5.
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use QUETIAPINE FUMARATE EXTENDED-RELEASE TABLETS safely and effectively. See full prescribing information
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use QUETIAPINE FUMARATE EXTENDED-RELEASE TABLETS safely and effectively. See full prescribing information
Prior Authorization Update: Botulinum Toxins
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: (Vraylar) Reference Number: CP.PMN.91 Effective Date: 11.16.16 Last Review Date: 02.18 Line of Business: Commercial, Medicaid Revision Log See Important Reminder at the end of this policy
Clinical Policy: (Vraylar) Reference Number: CP.PMN.91 Effective Date: 11.16.16 Last Review Date: 02.18 Line of Business: Commercial, Medicaid Revision Log See Important Reminder at the end of this policy
2. Is this request for a preferred medication? Y N
 Pharmacy Prior Authorization AETA BETTER HEALTH EW JERSE (MEDICAID) Opioids Long-Acting and Short-Acting (Medicaid) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review
Pharmacy Prior Authorization AETA BETTER HEALTH EW JERSE (MEDICAID) Opioids Long-Acting and Short-Acting (Medicaid) This fax machine is located in a secure location as required by HIPAA regulations. Complete/review
Kelly E. Williams, Pharm.D. PGY2 Psychiatric Pharmacy Resident April 16,2009
 Kelly E. Williams, Pharm.D. PGY2 Psychiatric Pharmacy Resident April 16,2009 List the antipsychotics most often prescribed Compare and contrast the use and adverse effects experienced in the pediatric
Kelly E. Williams, Pharm.D. PGY2 Psychiatric Pharmacy Resident April 16,2009 List the antipsychotics most often prescribed Compare and contrast the use and adverse effects experienced in the pediatric
Major Depressive Disorder (MDD) in Children under Age 6
 in Children under Age 6 Level 0 Comprehensive assessment. Refer to Principles of Practice on page 6. Level 1 Psychotherapeutic intervention (e.g., dyadic therapy) for 6 to 9 months; assessment of parent/guardian
in Children under Age 6 Level 0 Comprehensive assessment. Refer to Principles of Practice on page 6. Level 1 Psychotherapeutic intervention (e.g., dyadic therapy) for 6 to 9 months; assessment of parent/guardian
Class Update: Oral Antipsychotics
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Literature Scan: Alzheimer s Drugs
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Antidepressant Medication Therapy in Primary Care July 25, 2013
 New York State Collaborative Care Initiative Antidepressant Medication Therapy in Primary Care July 25, 2013 http://uwaims.org Presenter Building on 25 years of Research and Practice in Integrated Mental
New York State Collaborative Care Initiative Antidepressant Medication Therapy in Primary Care July 25, 2013 http://uwaims.org Presenter Building on 25 years of Research and Practice in Integrated Mental
Nuplazid. Nuplazid (pimavanserin) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.60.03 Subject: Nuplazid Page: 1 of 4 Last Review Date: June 22, 2018 Nuplazid Description Nuplazid (pimavanserin)
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.60.03 Subject: Nuplazid Page: 1 of 4 Last Review Date: June 22, 2018 Nuplazid Description Nuplazid (pimavanserin)
FL Medicaid Drug Therapy Management Program for Behavioral Health Monitoring for Safety and Quality
 FL Medicaid Drug Therapy Management Program for Behavioral Health Monitoring for Safety and Quality April 23, 2014 Pensacola, FL Presentation Objectives To briefly describe the program and how its components
FL Medicaid Drug Therapy Management Program for Behavioral Health Monitoring for Safety and Quality April 23, 2014 Pensacola, FL Presentation Objectives To briefly describe the program and how its components
Alabama Medicaid Pharmacy Override
 Alabama Medicaid Pharmacy Override Therapeutic Duplication, Early Refill, Maximum Unit, Prescription Limit Switchover, Dispense as Written, Accumulation Edit, Maintenance Supply Opt Out, and Maximum Cost
Alabama Medicaid Pharmacy Override Therapeutic Duplication, Early Refill, Maximum Unit, Prescription Limit Switchover, Dispense as Written, Accumulation Edit, Maintenance Supply Opt Out, and Maximum Cost
Impact of Florida s Medicaid Reform on Recipients of Mental Health Services
 Impact of Florida s Medicaid Reform on Recipients of Mental Health Services Jeffrey Harman, PhD John Robst, PhD Lilliana Bell, MHA The Quality of Behavioral Healthcare : A Drive for Change Through Research
Impact of Florida s Medicaid Reform on Recipients of Mental Health Services Jeffrey Harman, PhD John Robst, PhD Lilliana Bell, MHA The Quality of Behavioral Healthcare : A Drive for Change Through Research
The STOP Measure. Safe and Transparent Opioid Prescribing to Promote Patient Safety and Reduced Risk of Opioid Misuse FEBRUARY 2018
 The STOP Measure Safe and Transparent Opioid Prescribing to Promote Patient Safety and Reduced Risk of Opioid Misuse FEBRUARY 2018 AHIP s Safe, Transparent Opioid Prescribing (STOP) Initiative Methodology
The STOP Measure Safe and Transparent Opioid Prescribing to Promote Patient Safety and Reduced Risk of Opioid Misuse FEBRUARY 2018 AHIP s Safe, Transparent Opioid Prescribing (STOP) Initiative Methodology
Curbing the High Rates of Psychotropic Medication Prescriptions among Children and Youth in Foster Care
 Curbing the High Rates of Psychotropic Medication Prescriptions among Children and Youth in Foster Care Appendices Appendix A Psych Meds Data Indicators by State The data elements being collected by the
Curbing the High Rates of Psychotropic Medication Prescriptions among Children and Youth in Foster Care Appendices Appendix A Psych Meds Data Indicators by State The data elements being collected by the
Pediatric Behavioral Health Medication Initiative Prior Authorization (PA) Request Form
 Pediatric Behavioral Health Medication Initiative Prior Authorization (PA) Request Form Please fax form to 617.673.0988 or mail to Tufts Health Plan, 705 Mount Auburn Street, Watertown, MA 02472, Attn:
Pediatric Behavioral Health Medication Initiative Prior Authorization (PA) Request Form Please fax form to 617.673.0988 or mail to Tufts Health Plan, 705 Mount Auburn Street, Watertown, MA 02472, Attn:
PA Update: Oral Cystic Fibrosis Modulators
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Clinical Policy: Clozapine orally disintegrating tablet (Fazaclo) Reference Number: CP.PMN.12 Effective Date: Last Review Date: 02.
 Clinical Policy: Clozapine orally disintegrating tablet (Fazaclo) Reference Number: CP.PMN.12 Effective Date: 08.01.15 Last Review Date: 02.18 Line of Business: Medicaid Revision Log See Important Reminder
Clinical Policy: Clozapine orally disintegrating tablet (Fazaclo) Reference Number: CP.PMN.12 Effective Date: 08.01.15 Last Review Date: 02.18 Line of Business: Medicaid Revision Log See Important Reminder
Idaho DUR Board Meeting Minutes
 Idaho DUR Board Meeting Minutes Date: July 20, 2017 Time: 9am-12:30pm Location: Holiday Inn Boise Airport 2970 West Elder Street, Boise, Idaho, 83705 Moderator: David Agler, M.D. Committee Member Present:
Idaho DUR Board Meeting Minutes Date: July 20, 2017 Time: 9am-12:30pm Location: Holiday Inn Boise Airport 2970 West Elder Street, Boise, Idaho, 83705 Moderator: David Agler, M.D. Committee Member Present:
Idaho DUR Board Meeting Minutes
 Idaho DUR Board Meeting Minutes Date: October 16, 2014 Time: 9am-2pm Location: Idaho Medicaid, 3232 Elder Street, Boise, Idaho, Conference Room D-West Moderator: Mark Turner, M.D. Committee Member Present:
Idaho DUR Board Meeting Minutes Date: October 16, 2014 Time: 9am-2pm Location: Idaho Medicaid, 3232 Elder Street, Boise, Idaho, Conference Room D-West Moderator: Mark Turner, M.D. Committee Member Present:
Idaho DUR Board Meeting Minutes. Committee Member Present: David Agler, M.D., Dawn Berheim, Pharm.D., Perry Brown, M.D., Matthew Hyde, Pharm.D.
 Idaho DUR Board Meeting Minutes Date: January 25, 2018 Time: 9am-12:00pm Location: Idaho Medicaid, 3232 Elder Street, Boise, Idaho, Conference Room D-West Moderator: David Agler, M.D. Committee Member
Idaho DUR Board Meeting Minutes Date: January 25, 2018 Time: 9am-12:00pm Location: Idaho Medicaid, 3232 Elder Street, Boise, Idaho, Conference Room D-West Moderator: David Agler, M.D. Committee Member
Promoting and Monitoring Evidenced-Based Antipsychotic Prescribing Practices in Children and Adolescents: Florida Medicaid Initiatives
 Promoting and Monitoring Evidenced-Based Antipsychotic Prescribing Practices in Children and Adolescents: Florida Medicaid Initiatives Mary Elizabeth Jones, Pharm BSc, RPh Senior Pharmacist AHCA Pharmacy
Promoting and Monitoring Evidenced-Based Antipsychotic Prescribing Practices in Children and Adolescents: Florida Medicaid Initiatives Mary Elizabeth Jones, Pharm BSc, RPh Senior Pharmacist AHCA Pharmacy
Antipsychotic Prior Authorization Request
 Antipsychotic Prior Authorization Request Commonwealth of Massachusetts MassHealth Drug Utilization Review Program P.O. Box 2586, Worcester, MA 01613-2586 Fax: 1-877-208-7428 Phone: 1-800-745-7318 MassHealth
Antipsychotic Prior Authorization Request Commonwealth of Massachusetts MassHealth Drug Utilization Review Program P.O. Box 2586, Worcester, MA 01613-2586 Fax: 1-877-208-7428 Phone: 1-800-745-7318 MassHealth
ProviderNews FEBRUARY
 ProviderNews FEBRUARY 2017 Reminder: decimal billing required on time-based therapy codes for BadgerCare Plus members In accordance with Forward Health guidelines, Security Health Plan requires decimal
ProviderNews FEBRUARY 2017 Reminder: decimal billing required on time-based therapy codes for BadgerCare Plus members In accordance with Forward Health guidelines, Security Health Plan requires decimal
Mail Service Pharmacy
 Mail Service s At A Glance Disclaimer: reserves the right to update its measures and measure sets to maintain measure relevancy and to remedy any unintended consequences that may arise during implementation.
Mail Service s At A Glance Disclaimer: reserves the right to update its measures and measure sets to maintain measure relevancy and to remedy any unintended consequences that may arise during implementation.
FULL PRESCRIBING INFORMATION: CONTENTS*
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use SEROQUEL XR safely and effectively. See full prescribing information for SEROQUEL XR. SEROQUEL XR
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use SEROQUEL XR safely and effectively. See full prescribing information for SEROQUEL XR. SEROQUEL XR
New patients approved for the Novo Nordisk PAP may only be eligible for insulin vials. For a full list of available products, please visit:
 The Novo Nordisk Diabetes Patient Assistance Program (PAP) provides medication to qualifying applicants at no charge. If the applicant qualifies under the Novo Nordisk Diabetes PAP guidelines, a 120-day
The Novo Nordisk Diabetes Patient Assistance Program (PAP) provides medication to qualifying applicants at no charge. If the applicant qualifies under the Novo Nordisk Diabetes PAP guidelines, a 120-day
Exhibit I-1 Performance Measures. Numerator (general description only)
 # Priority Type Performance Measure Core Measures (implement 9/1/09) 1 C OE Hospital readmissions within 7, 30 and 90 days postdischarge 2 C OE Percent of Members prescribed redundant or duplicated antipsychotic
# Priority Type Performance Measure Core Measures (implement 9/1/09) 1 C OE Hospital readmissions within 7, 30 and 90 days postdischarge 2 C OE Percent of Members prescribed redundant or duplicated antipsychotic
Clinical Policy: Olanzapine Long-Acting Injection (Zyprexa Relprevv) Reference Number: CP.PHAR.292 Effective Date: Last Review Date: 08.
 Clinical Policy: (Zyprexa Relprevv) Reference Number: CP.PHAR.292 Effective Date: 12.01.16 Last Review Date: 08.18 Line of Business: Medicaid See Important Reminder at the end of this policy for important
Clinical Policy: (Zyprexa Relprevv) Reference Number: CP.PHAR.292 Effective Date: 12.01.16 Last Review Date: 08.18 Line of Business: Medicaid See Important Reminder at the end of this policy for important
Current Non-Preferred Agents
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35, Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35, Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Issues for Part D Compliance
 HCCA- MEDICARE PRESCRIPTION DRUG PART D COMPLIANCE CONFERENCE Issues for Part D Compliance Craig Miner, RPh, JD Division of Drug Plan Policy Babette S. Edgar, Pharm.D., MBA Division of Finance and Operations
HCCA- MEDICARE PRESCRIPTION DRUG PART D COMPLIANCE CONFERENCE Issues for Part D Compliance Craig Miner, RPh, JD Division of Drug Plan Policy Babette S. Edgar, Pharm.D., MBA Division of Finance and Operations
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: Non-Benzodiazepine Insomnia Reference Number: CP.PMA_10.11.23 Effective Date: 07.16 Last Review Date: 04.18 Line of Business: Medicaid- Arizona (AHCS, CIC) Revision Log See Important Reminder
Clinical Policy: Non-Benzodiazepine Insomnia Reference Number: CP.PMA_10.11.23 Effective Date: 07.16 Last Review Date: 04.18 Line of Business: Medicaid- Arizona (AHCS, CIC) Revision Log See Important Reminder
MEDICATION GUIDE. Quetiapine (kwe-tye-a-peen) Tablets USP
 MEDICATION GUIDE Quetiapine (kwe-tye-a-peen) Tablets USP Read this Medication Guide before you start taking quetiapine tablets and each time you get a refill. There may be new information. This information
MEDICATION GUIDE Quetiapine (kwe-tye-a-peen) Tablets USP Read this Medication Guide before you start taking quetiapine tablets and each time you get a refill. There may be new information. This information
MEDICATION GUIDE Quetiapine Fumarate Extended-Release Tablets (Kwe-TYE-a-peen FUE-ma-rate)
 MEDICATION GUIDE Quetiapine Fumarate Extended-Release Tablets (Kwe-TYE-a-peen FUE-ma-rate) Read this Medication Guide before you start taking quetiapine fumarate extended-release tablets and each time
MEDICATION GUIDE Quetiapine Fumarate Extended-Release Tablets (Kwe-TYE-a-peen FUE-ma-rate) Read this Medication Guide before you start taking quetiapine fumarate extended-release tablets and each time
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: Policy Reference Number: CP.PMN.53 Effective Date: 09.12.17 Last Review Date: 11.18 Line of Business: Medicaid, HIM-Medical Benefit Revision Log See Important Reminder at the end of this
Clinical Policy: Policy Reference Number: CP.PMN.53 Effective Date: 09.12.17 Last Review Date: 11.18 Line of Business: Medicaid, HIM-Medical Benefit Revision Log See Important Reminder at the end of this
Integrating INTERACT into Interim Pharmacist Reviews
 Integrating INTERACT into Interim Pharmacist Reviews Chad R. Worz, Pharm.D. President, Medication Managers, LLC Adjunct Assistant Professor of Pharmacy Practice, University of Cincinnati, College of Pharmacy
Integrating INTERACT into Interim Pharmacist Reviews Chad R. Worz, Pharm.D. President, Medication Managers, LLC Adjunct Assistant Professor of Pharmacy Practice, University of Cincinnati, College of Pharmacy
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: (Abilify Maintena, Aristada, Aristada Initio) Reference Number: CP.PHAR.290 Effective Date: 12.01.16 Last Review Date: 08.18 Line of Business: Medicaid Coding Implications Revision Log
Clinical Policy: (Abilify Maintena, Aristada, Aristada Initio) Reference Number: CP.PHAR.290 Effective Date: 12.01.16 Last Review Date: 08.18 Line of Business: Medicaid Coding Implications Revision Log
DURATION LIMIT WITH QUANTITY LIMIT AND POST LIMIT PRIOR AUTHORIZATION CRITERIA IMMEDIATE-RELEASE OPIOID ANALGESICS (BRAND AND GENERIC)*
 Carelirst. +.V Family of health care plans cvs caremarktm DURATION LIMIT WITH QUANTITY LIMIT AND POST LIMIT PRIOR AUTHORIZATION CRITERIA DRUG CLASS generic name, dosage form IMMEDIATE-RELEASE OPIOID ANALGESICS
Carelirst. +.V Family of health care plans cvs caremarktm DURATION LIMIT WITH QUANTITY LIMIT AND POST LIMIT PRIOR AUTHORIZATION CRITERIA DRUG CLASS generic name, dosage form IMMEDIATE-RELEASE OPIOID ANALGESICS
Archived SECTION 15 - BILLING INSTRUCTIONS. Section 15 - Billing Instructions
 SECTION 15 - BILLING INSTRUCTIONS 15.1 ELECTRONIC DATA INTERCHANGE... 2 15.2 INTERNET ELECTRONIC CLAIM SUBMISSION... 2 15.3 DENTAL CLAIM FORM... 3 15.4 PROVIDER RELATIONS COMMUNICATION UNIT... 3 15.5 RESUBMISSION
SECTION 15 - BILLING INSTRUCTIONS 15.1 ELECTRONIC DATA INTERCHANGE... 2 15.2 INTERNET ELECTRONIC CLAIM SUBMISSION... 2 15.3 DENTAL CLAIM FORM... 3 15.4 PROVIDER RELATIONS COMMUNICATION UNIT... 3 15.5 RESUBMISSION
Major Depressive Disorder (MDD) in Children under Age 6
 in Children under Age 6 Level 0 Comprehensive assessment. Refer to Principles of Practice on page 5. Level 1 Psychotherapeutic intervention (e.g., dyadic therapy) for 6 to 9 months; assessment of parent/guardian
in Children under Age 6 Level 0 Comprehensive assessment. Refer to Principles of Practice on page 5. Level 1 Psychotherapeutic intervention (e.g., dyadic therapy) for 6 to 9 months; assessment of parent/guardian
MEASURE STEWARD Pharmacy Quality Alliance (PQA) D ATA SOURCE Enrollment; U R A C DOMAIN Engagement & Experience of Care
 MANDATORY S Note: Mandatory measures are those measures that are a requirement of accreditation and must be reported to URAC on an annual basis. DM2012-12 DTM2015-01 Portion of Days Covered (PDC) Adherence
MANDATORY S Note: Mandatory measures are those measures that are a requirement of accreditation and must be reported to URAC on an annual basis. DM2012-12 DTM2015-01 Portion of Days Covered (PDC) Adherence
ZURAMPIC (lesinurad) oral tablet
 ZURAMPIC (lesinurad) oral tablet Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This Pharmacy Coverage
ZURAMPIC (lesinurad) oral tablet Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This Pharmacy Coverage
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: Non-Benzodiazepine Insomnia Medications Reference Number: CP.HNMC.265 Effective Date: 11.16.16 Last Review Date: 11.17 Line of Business: Medicaid Medi-Cal Revision Log See Important Reminder
Clinical Policy: Non-Benzodiazepine Insomnia Medications Reference Number: CP.HNMC.265 Effective Date: 11.16.16 Last Review Date: 11.17 Line of Business: Medicaid Medi-Cal Revision Log See Important Reminder
Geodon for anxiety and depression
 When drugs like carbamazepine or cisapride are taken with Geodon, drug interactions can potentially occur. This emedtv page lists other drugs that may lead to Geodon. 8-2-2017 Find a comprehensive guide
When drugs like carbamazepine or cisapride are taken with Geodon, drug interactions can potentially occur. This emedtv page lists other drugs that may lead to Geodon. 8-2-2017 Find a comprehensive guide
Drug Therapy Management
 Measures At A Glance Updated: 11/01/2018 2018 Page 1 of 9 Mandatory Measures (10) Note: Mandatory measures are those measures that are a requirement of accreditation and must be reported to on an annual
Measures At A Glance Updated: 11/01/2018 2018 Page 1 of 9 Mandatory Measures (10) Note: Mandatory measures are those measures that are a requirement of accreditation and must be reported to on an annual
LOUISIANA MEDICAID PROGRAM ISSUED: 02/01/12 REPLACED: 02/01/94 CHAPTER 5: PROFESSIONAL SERVICES SECTION 5.1: COVERED SERVICES PAGE(S) 5
 Immunizations Vaccine Codes Providers should refer to the Immunization Fee Schedules to determine covered vaccines and any restriction to the use of the vaccine codes. (See Appendix A for information on
Immunizations Vaccine Codes Providers should refer to the Immunization Fee Schedules to determine covered vaccines and any restriction to the use of the vaccine codes. (See Appendix A for information on
Opioid Analgesic Treatment Worksheet
 Opioid Analgesic Treatment Worksheet Aetna Better Health of Louisiana Fax: 1 844 699 2889 www.aetnabetterhealth.com/louisiana/providers/pharmacy LA Legacy Fee for Service (FFS) Medicaid Fax: 1 866 797
Opioid Analgesic Treatment Worksheet Aetna Better Health of Louisiana Fax: 1 844 699 2889 www.aetnabetterhealth.com/louisiana/providers/pharmacy LA Legacy Fee for Service (FFS) Medicaid Fax: 1 866 797
ADMINISTRATIVE POLICY AND PROCEDURE
 Page 1 of 6 SECTION: Medical SUBJECT: Electroconvulsive Therapy (ECT) DATE OF ORIGIN: 10/1/96 REVIEW DATES: 7/1/98, 10/1/99, 7/1/02, 7/1/04, 10/1/05, 5/1/09, 1/3/13, 7/1/15, 8/1/16 EFFECTIVE DATE: 3/24/17
Page 1 of 6 SECTION: Medical SUBJECT: Electroconvulsive Therapy (ECT) DATE OF ORIGIN: 10/1/96 REVIEW DATES: 7/1/98, 10/1/99, 7/1/02, 7/1/04, 10/1/05, 5/1/09, 1/3/13, 7/1/15, 8/1/16 EFFECTIVE DATE: 3/24/17
Bipolar Disorder Or Drugs True Stories Of Life In A Psychiatric Hospital Book 5
 Bipolar Disorder Or Drugs True Stories Of Life In A Psychiatric Hospital Book 5 We have made it easy for you to find a PDF Ebooks without any digging. And by having access to our ebooks online or by storing
Bipolar Disorder Or Drugs True Stories Of Life In A Psychiatric Hospital Book 5 We have made it easy for you to find a PDF Ebooks without any digging. And by having access to our ebooks online or by storing
Pharmacy Prior Authorization GMH/SA and Non-Title 19/21 SMI Non-Formulary and Prior Authorization Guidelines
 Non-Formulary Behavioral Health Medications ADHD medications for children under The patient must have a diagnosis for which the requested medication is: o Approved based on FDA indication and limits; OR
Non-Formulary Behavioral Health Medications ADHD medications for children under The patient must have a diagnosis for which the requested medication is: o Approved based on FDA indication and limits; OR
MEDICAL ASSISTANCE BULLETIN
 ISSUE DATE September 4, 2015 SUBJECT EFFECTIVE DATE September 9, 2015 MEDICAL ASSISTANCE BULLETIN NUMBER *See below BY Prior Authorization of Analgesics, Narcotic Long Acting and Analgesics, Narcotic Short
ISSUE DATE September 4, 2015 SUBJECT EFFECTIVE DATE September 9, 2015 MEDICAL ASSISTANCE BULLETIN NUMBER *See below BY Prior Authorization of Analgesics, Narcotic Long Acting and Analgesics, Narcotic Short
Seroquel XR. quetiapine fumarate APPROVED PRODUCT INFORMATION
 Seroquel XR quetiapine fumarate APPROVED PRODUCT INFORMATION NAME OF THE MEDICINE Quetiapine fumarate Chemical Name: Bis[2-(2-[4-(dibenzo[b,f][1,4]-thiazepin-11-yl)piperazin-1-yl] ethoxy) ethanol] fumarate.
Seroquel XR quetiapine fumarate APPROVED PRODUCT INFORMATION NAME OF THE MEDICINE Quetiapine fumarate Chemical Name: Bis[2-(2-[4-(dibenzo[b,f][1,4]-thiazepin-11-yl)piperazin-1-yl] ethoxy) ethanol] fumarate.
METHODS RESULTS. Supported by funding from Ortho-McNeil Janssen Scientific Affairs, LLC
 PREDICTORS OF MEDICATION ADHERENCE AMONG PATIENTS WITH SCHIZOPHRENIC DISORDERS TREATED WITH TYPICAL AND ATYPICAL ANTIPSYCHOTICS IN A LARGE STATE MEDICAID PROGRAM S.P. Lee 1 ; K. Lang 2 ; J. Jackel 2 ;
PREDICTORS OF MEDICATION ADHERENCE AMONG PATIENTS WITH SCHIZOPHRENIC DISORDERS TREATED WITH TYPICAL AND ATYPICAL ANTIPSYCHOTICS IN A LARGE STATE MEDICAID PROGRAM S.P. Lee 1 ; K. Lang 2 ; J. Jackel 2 ;
OHSU Drug Effectiveness Review Project Summary Report Benzodiazepines
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
