The Conditions of Sale apply to the written text in this Catalogue superseding earlier texts related to such conditions.
|
|
|
- Ashlee Briggs
- 5 years ago
- Views:
Transcription
1
2 Conditions of Sale Validity Intention of Use The Conditions of Sale apply to the written text in this Catalogue superseding earlier texts related to such conditions. Our products are intended for research purposes only. Prices Delivery See under Order Information. All prices in this catalogue are net prices in Euro, ex works. Taxes, shipping costs or other external costs demanded by the buyer are invoiced. See under Order Information shipping terms. Payment terms Payment terms are normally net 30 days. Deductions are not accepted unless we have issued a credit note. We accept payment by credit card (Visa/ Mastercard), bank transfer (wiring) or by cheque. If payment by cheque we will add a bank fee to our invoice. Complaints Insurance Delays Warranty and Compensation Claims Acceptance Complaints about a product or products must be made inside 30 days from the invoice date. All claims must specify batch (lot) and invoice numbers. Return of goods will not be accepted unless authorized by us. Insurance will not be made unless otherwise instructed. We cannot accept compensation claims due to delays or non-deliveries. We reserve us the right to withdraw from delivery due to long term shortage of starting materials, production breakdown or other circumstances beyond our control. All products in this catalogue are warranted to be free of defects and in accordance with given specifications. If this warranty does not comply with specifications, any indemnities will be limited to not exceed the price paid for the goods. Placing of an order implies acceptance of our conditions of sale. We accept credit cards (Visa/ Mastercard) info@larodan.se
3 Ordering Information You can easily order from Larodan please contact our local distributor or us by phone, , fax or letter. Visiting & Mailing Larodan Fine Chemicals AB Phone nat address Limhamnsgårdens allé int SE Malmö info@larodan.se Sweden Fax nat web site Placing your Order To speed up handling of orders, please indicate the following Product catalogue number Description Size. EU Vat number if you are located inside the EU. Order acknowledgement Prices Shipping terms Normally we do not acknowledge incoming orders. If you want us to acknowledge your order please indicate that. All prices are quoted in Euro and are subject to change without notice. However, if the price of any item has been changed by more than 10%, we will notify you before processing your order. Delivery terms are FOB Malmö. We ship by courier, registered mail or parcel post if not otherwise instructed. If not shipped collect we prepay the freight charge and add it to our invoice. 2 info@larodan.se
4 Contents Alphabetical Index Page no Acyl-L-Carnitines Native Deuterium Labelled Bile Acids & Methyl Esters 132 Cholesterol & Esters 96 Coenzyme A & Fatty Acid CoA Esters Conjugated Linoleic Acids (CLA) Analytical Standards 18 FFA/ Esters/ Triglycerides in bulk Dicarboxylic Acids & Dimethyl Esters Dolichols & Phosphates 130 Eicosanoids Fatty Acids & Methyl Esters straight chain Saturated & Unsaturated 6-18 EPA & DHA Esters in bulk 26 Fatty Acids - Unusual / Methyl Esters Conjugated Polyene Odd-Numbered Polyunsaturated Other Unusual Fatty Acids & Methyl Esters methyl branched/ cyclopropyl Phytanic & Pristanic Native and Deuterium labelled 57 Iso & Anteiso methyl Branched 62 Cyclopropyl 62 Fatty Acids & Methyl Esters Oxygenated/ Oxylipin related Hydroxy Dihydroxy 48 Epoxy 47 Oxo Fatty Acids & Methyl Esters Hydroxy (synthetic) Hydroxy Hydroxy Omega-Hydroxy Other Microbiological 68 info@larodan.se
5 Fatty Acids Deuterium labelled Perdeuterated α-deuterated ω-deuterated Polyunsaturated 90 Miscellaneous 90 Fatty Acids 13C labelled 92 Fatty Alcohols Gangliosides & Derivatives Glycerides Monoglycerides - Saturated/ Unsaturated Diglycerides Single/ Mixed Fatty Acids Triglycerides - Single/ Mixed Fatty Acids Triglycerides 13C-Labelled 93 Hydrocarbons Aliphatic Isoprenoid 135 Kits Fatty Acids/ Methyl Esters Glycerides 140 Aliphatic Hydrocarbons 140 Phosphoglycerides 141 Phospholipids 141 Plant Sterols 141 Larodan Reference Mixtures Glycoshingolipid Mixtures FAME Mixtures 142 Oil Mixtures Methyl Branched FAME Mixtures Glyceride Mixtures 156 Mixed Lipid Mixtures Polar Lipids Mixture 159 Polyprenol Mixtures 158 Free Fatty Acid Mixtures 153 n-paraffin Mixture 159 Fish Oil FAME Mixture 157 Metabolic Research Products Long Chain Fatty Acids Deuteriumlabelled 91 Acylglycines/ Miscellaneous 13C/ Deuteriumlabelled 93 Oligoprenyl Phosphates info@larodan.se
6 Oxylipines Hydroperoxides Hydroxides Ketones Epoxides Diols Allene-Oxide-Synthase- -derived Divinyl-Ether-Synthase- -derived Epoxy Alcohol Synthase / Peroxygenase-derived Lyase-derived 44 Others 44 D-myo-Phosphatidylinositol Phosphates Phospholipids Natural PC, PE, PI, PA, PG, CL, PS, LPC, LPE, LPI, LPG Semisynthetic GPC, PC, PE, PA, PG, PS, LPC, LPG Deuterium Labelled 90 Di-O-Alkyl 114 Plant Leaf Lipids 126 Plant Sterols & Steryl Glucosides 126 Plasmalogens 109 Polyprenols & Monophosphates Sialic Acid & Derivatives 124 Sphingolipids Sphingosines Ceramides Cerebrosides Sphingomyelins 121 Fluorescent 122 Wax Esters C12-C info@larodan.se
7 Nomenclature Geometrical Isomers In this catalogue we use two systems to descibe the geometry of structures having carbon-carbon double bonds (geometric isomers). Cis/ trans denomination is used to indicate C=C configuration of common unsaturated fatty acids of natural origin, i.e. from vegetable and animal fats and oils. A more distinct nomenclature, the Z/E-system, was introduced in the 1950 s by Cahn, Ingold and Prelog. For fatty acids and derivatives cis is equivalent to Z and trans to E, which is not always the case for other kinds of molecules. Fatty Acids & Derivatives Saturated n-fatty Acids (n-carboxylic) Bulk discounts are available on larger quantities. Other esters, such as ethyl esters can be offered on request Saturated and unsaturated fatty acids occur, as triglycerides, mainly in plants and animals, such as in oil bearing seeds and fruits, tallow, lard and marine oils. In natural fats and oils, even numbered fatty acids are the predominant ones - odd numbered are rare. Larodan provides a growing range of high purity fatty acids and esters for research and analytical purposes. In this catalogue you will find new lines of fatty acids and metabolites under special headings. Please visit our web site for the latest news Propionic Acid No. of C: 3:0 Mw: Documentation: GC Purity: 99+% Size Order no EUR 100 mg g g Butyric Acid No. of C: 4:0 Mw: Documentation: GC Purity: 99+% Pentanoic Acid No. of C: 5:0 Mw: Documentation: GC Purity: 99+% Hexanoic Acid (Caproic Acid) No. of C: 6:0 Mw: Documentation: GC Purity: 99+% Heptanoic Acid No. of C: 7:0 Mw: Documentation: GC Purity: 99+% Octanoic Acid (Caprylic Acid) No. of C: 8:0 Mw: Documentation: GC Purity: 99+% 100 mg g g mg g g mg g g mg g g mg g g info@larodan.se
8 Nonanoic Acid (Pelargonic Acid) No. of C: 9:0 Mw: Documentation: GC Purity: 99+% Decanoic Acid (Capric Acid) No. of C: 10:0 Mw: Documentation: GC Purity: 99+% Undecanoic Acid No. of C: 11:0 Mw: Documentation: GC Purity: 99+% Dodecanoic Acid (Lauric Acid) No. of C: 12:0 Mw: Documentation: GC Purity: 99+% Tridecanoic Acid No. of C: 13:0 Mw: Documentation: GC Purity: 99+% Tetradecanoic Acid (Myristic Acid) No. of C: 14:0 Mw: Documentation: GC Purity: 99+% Pentadecanoic Acid No. of C: 15:0 Mw: Documentation: GC Purity: 99+% Hexadecanoic Acid (Palmitic Acid) No. of C: 16:0 Mw: Documentation: GC Purity: 99+% Heptadecanoic Acid (Margaric Acid) No. of C: 17:0 Mw: Documentation: GC Purity: 99+% Octadecanoic Acid (Stearic Acid) No. of C: 18:0 Mw: Documentation: GC Purity: 99+% Nonadecanoic Acid No. of C: 19:0 Mw: Documentation: GC Purity: 99+% 100 mg g g mg g g mg g g mg g g mg g g mg g g mg g g mg g g mg g g mg g g mg g g info@larodan.se
9 Eicosanoic Acid(Arachidic Acid) No. of C: 20:0 Mw: Documentation: GC Purity: 99+% Heneicosanoic Acid No. of C: 21:0 Mw: Documentation: GC Purity: 99+% Docosanoic Acid (Behenic Acid) No. of C: 22:0 Mw: Documentation: GC Purity: 99+% Tricosanoic Acid No. of C: 23:0 Mw: Documentation: GC Purity: 99+% Tetracosanoic Acid (Lignoceric Acid) No. of C: 24:0 Mw: Documentation: GC Purity: 99+% Pentacosanoic Acid No. of C: 25:0 Mw: Documentation: GC Purity: 99+% Hexacosanoic Acid (Cerotic Acid) No. of C: 26:0 Mw: Documentation: GC Purity: 99+% Heptacosanoic Acid No. of C: 27:0 Mw: Documentation: GC Purity: 97+% Octacosanoic Acid (Montanic Acid) No. of C: 28:0 Mw: Documentation: GC Purity: 99+% Nonacosanoic Acid No. of C: 29:0 Mw: Documentation: GC Purity: 99+% Triacontanoic Acid(Melissic Acid) No. of C: 30:0 Mw: Documentation: GC Purity: 98+% Hentriacontanoic Acid No. of C: 31:0 Mw: Documentation: GC Purity: 99+% 100 mg g g mg mg g mg g g mg mg g mg mg g mg mg mg mg g mg mg mg mg mg mg mg mg Inquire 8 info@larodan.se
10 Saturated n-fatty Acid Methyl Esters Methyl Propionate No. of C: 3:0 Documentation: GC Purity: 99+% Methyl Butyrate No. of C: 4:0 Documentation: GC Purity: 99+% Methyl Pentanoate No. of C: 5:0 Documentation: GC Purity: 99+% Methyl Hexanoate No. of C: 6:0 Documentation: GC Purity: 99+% Methyl Heptanoate No. of C: 7:0 Documentation: GC Purity: 99+% Methyl Octanoate No. of C: 8:0 Documentation: GC Purity: 99+% Methyl Nonanoate No. of C: 9:0 Documentation: GC Purity: 99+% Methyl Decanoate No. of C: 10:0 Documentation: GC Purity: 99+% Methyl Undecanoate No. of C: 11:0 Documentation: GC Purity: 99+% Methyl Dodecanoate No. of C: 12:0 Documentation: GC Purity: 99+% Methyl Tridecanoate No. of C: 13:0 Documentation: GC Purity: 99+% 100 mg g g mg g g mg g g mg g g mg g g mg g g mg g g mg g g mg g g mg g g mg g g info@larodan.se
11 Methyl Tetradecanoate No. of C: 14:0 Documentation: GC Purity: 99+% Methyl Pentadecanoate No. of C: 15:0 Documentation: GC Purity: 99+% Methyl Hexadecanoate No. of C: 16:0 Documentation: GC Purity: 99+% Methyl Heptadecanoate No. of C: 17:0 Documentation: GC Purity: 99+% Methyl Octadecanoate No. of C: 18:0 Documentation: GC Purity: 99+% Methyl Nonadecanoate No. of C: 19:0 Documentation: GC Purity: 99+% Methyl Eicosanoate No. of C: 20:0 Documentation: GC Purity: 99+% Methyl Heneicosanoate No. of C: 21:0 Documentation: GC Purity: 99+% Methyl Docosanoate No. of C: 22:0 Documentation: GC Purity: 99+% Methyl Tricosanoate No. of C: 23:0 Documentation: GC Purity: 99+% Methyl Tetracosanoate No. of C: 24:0 Documentation: GC Purity: 99+% 100 mg g g mg g g mg g g mg g g mg g g mg g g mg g g mg mg g mg g g mg mg g mg mg g info@larodan.se
12 Methyl Pentacosanoate No. of C: 25:0 Documentation: GC Purity: 99+% Methyl Hexacosanoate No. of C: 26:0 Documentation: GC Purity: 99+% Methyl Heptacosanoate No. of C: 27:0 Documentation: GC Purity: 99+% Methyl Octacosanoate No. of C: 28:0 Documentation: GC Purity: 99+% Methyl Nonacosanoate No. of C: 29:0 Documentation: GC Purity: 99+% Methyl Triacontanoate No. of C: 30:0 Documentation: GC Purity: 98% Methyl Hentriacontanoate No. of C: 31:0 Documentation: GC Purity: 99+% 25 mg mg mg mg g mg mg mg mg mg mg mg mg mg mg info@larodan.se
13 Unsaturated n-fatty Acids 10-Hydroxy-2tr-Decenoic Acid (Royal Jelly Acid) No. of C: 10:1 Mw: Documentation: Not included Purity: 98% 10c-Undecenoic Acid No. of C: 11:1 Mw: Documentation: GC Purity: 99+% 11c-Dodecenoic Acid No. of C: 12:1 Mw: Documentation: GC Purity: 99+% 12c-Tridecenoic Acid No. of C: 13:1 Mw: Documentation: GC Purity: 99+% Myristoleic Acid (9c) No. of C: 14:1 Mw: Documentation: GC Purity: 99+% Myristelaidic Acid (9tr) No. of C: 14:1 Mw: Documentation: GC Purity: 99+% 10c-Pentadecenoic Acid No. of C: 15:1 Mw: Documentation: GC Purity: 99+% 10tr-Pentadecenoic Acid No. of C: 15:1 Mw: Documentation: GC Purity: 99+% Palmitoleic Acid (9c) No. of C: 16:1 Mw: Documentation: GC Purity: 99+% Palmitelaidic Acid (9tr) No. of C: 16:1 Mw: Documentation: GC Purity: 99+% 2tr-Hexadecenoic Acid No. of C: 16:1 Mw: Documentation: Not included Purity: 98% 10c-Heptadecenoic Acid No. of C: 17:1 Mw: Documentation: GC Purity: 99+% 10tr-Heptadecenoic Acid No. of C: 17:1 Mw: Documentation: GC Purity: 99+% 50 mg mg x 100 mg mg x 100 mg mg x 100 mg mg x 100 mg mg x 100 mg mg x 100 mg mg mg mg x 100 mg mg x 100 mg Inquire 100 mg x 100 mg mg x 25 mg info@larodan.se
14 Oleic Acid (9c) No. of C: 18:1 Mw: Documentation: GC Purity: 99+% Elaidic Acid (9tr) No. of C: 18:1 Mw: Documentation: GC Purity: 99+% Petroselinic Acid (6c) No. of C: 18:1 Mw: Documentation: GC Purity: 99+% Vaccenic Acid (11c) No. of C: 18:1 Mw: Documentation: GC Purity: 99+% Transvaccenic Acid (11tr) No. of C: 18:1 Mw: Documentation: GC Purity: 99+% Ricinoleic Acid (9c, 12OH) No. of C: 18:1 Mw: Documentation: GC Purity: 99+% Ricinelaidic Acid (9tr, 12OH) No. of C: 18:1 Mw: Documentation: GC Purity: 99+% Linoleic Acid (9c, 12c) No. of C: 18:2 Mw: Documentation: GC Purity: 99+% Linoelaidic Acid (9tr, 12tr)) No. of C: 18:2 Mw: Documentation: GC Purity: 99+% Linolenic Acid (9c, 12c, 15c) No. of C: 18:3 Mw: Documentation: GC Purity: 99+% Linolenic Acid (9c, 12c, 15c) No. of C: 18:3 Mw: Documentation: GC Purity: min. 80% Gamma Linolenic Acid (6c, 9c, 12c) No. of C: 18:3 Mw: Documentation: GC Purity: 99+% 6c,9c,12c,15c-Octadecatetraenoic Acid (Stearidonic acid) No. of C: 18:4 Mw: c-Eicosenoic Acid No. of C: 20:1 Mw: Documentation: GC Purity: 99+% 1 g g g x 1 g mg x 100 mg mg x 100 mg mg x 100 mg mg x 100 mg mg x 100 mg g g mg x 100 mg mg x 100 mg kg mg x 100 mg mg mg x 100 mg info@larodan.se
15 11tr-Eicosenoic Acid No. of C: 20:1 Mw: Documentation: GC Purity: 99+% 11c,14c-Eicosadienoic Acid No. of C: 20:2 Mw: Documentation: GC Purity: 99+% 11c,14c,17c-Eicosatrienoic Acid No. of C: 20:3 Mw: Documentation: GC Purity: 99+% Homogamma Linolenic Acid (8c,11c,14c) No. of C: 20:3 Mw: Documentation: GC Purity: 99+% Arachidonic Acid (5c,8c,11c,14c) No. of C: 20:4 Mw: Documentation: GC Purity: 99+% Omega3-Arachidonic Acid (8c,11c,14c,17c)) No. of C: 20:4 Mw: Documentation: CoA Purity: 98% 5c,8c,11c,14c,17c-Eicosapentaenoic Acid No. of C: 20:5 Mw: Documentation: GC Purity: 99+% Erucic Acid (13c-Docosenoic Acid) No. of C: 22:1 Mw: Documentation: GC Purity: 99+% Brassidic Acid (13tr-Docosenoic Acid) No. of C: 22:1 Mw: Documentation: GC Purity: 99+% 13c,16c-Docosadienoic Acid No. of C: 22:2 Mw: Documentation: Not included Purity: 99+% 13c,16c,19c-Docosatrienoic Acid No. of C: 22:3 Mw: Documentation: Not included Purity: 99+% 7c,10c,13c,16c-Docosatetraenoic Acid No. of C: 22:4 Mw: Documentation: Not included Purity: 99+% 4c,7c,10c,13c,16c-Docosapentaenoic Acid No. of C: 22:5 Mw: Documentation: GC Purity: 99+% 7c,10,13c,16c,19c-Docosapentaenoic Acid No. of C: 22:5 Mw: Documentation: GC Purity: 99+% 25 mg x 25 mg mg x 100 mg mg x 100 mg mg mg mg mg mg mg mg x 100 mg mg x 100 mg mg x 100 mg mg x 25 mg mg x 25 mg mg x 25 mg mg mg x 100 mg info@larodan.se
16 4c,7c,10c,13c,16c,19c-Docosahexaenoic Acid No. of C: 22:6 Mw: Documentation: GC Purity: 99+% Nervonic Acid (15c) No. of C: 24:1 Mw: Documentation: GC Purity: 99+% 9c, 12c, 15c, 18c, 21c-Tetracosapentaenoic Acid No. of C: 24:5 Mw: c, 9c, 12c, 15c, 18c, 21c-Tetracosahexaenoic Acid No. of C: 24:6 Mw: Unsaturated n-fatty Acid Methyl Esters Methyl 10c-Undecenoate No. of C: 11:1 Documentation: GC Purity: 99+% Methyl 11c-Dodecenoate No. of C: 12:1 Documentation: GC Purity: 99+% Methyl 12c-Tridecenoate No. of C: 13:1 Documentation: GC Purity: 99+% Methyl Myristoleate (9c) No. of C: 14:1 Documentation: GC Purity: 99+% Methyl Myristelaidate (9tr) No. of C: 14:1 Documentation: GC Purity: 99+% Methyl 10c-Pentadecenoate No. of C: 15:1 Documentation: GC Purity: 99+% Methyl -10tr-Pentadecenoate No. of C: 15:1 Documentation: GC Purity: 99+% Methyl Palmitoleate (9c) No. of C: 16:1 Documentation: GC Purity: 99+% Methyl Palmitelaidate (9tr) No. of C: 16:1 Documentation: GC Purity: 99+% 100 mg x 100 mg mg x 100 mg mg mg mg x 100 mg mg x 100 mg mg x 100 mg mg x 100 mg mg x 100 mg mg x 100 mg mg mg mg x 100 mg mg x 100 mg info@larodan.se
17 Methyl 10c-Heptadecenoate No. of C: 17:1 Documentation: GC Purity: 99+% Methyl 10tr-Heptadecenoate No. of C: 17:1 Documentation: GC Purity: 99+% Methyl Oleate (9c) No. of C: 18:1 Documentation: GC Purity: 99+% Methyl Elaidate (9tr) No. of C: 18:1 Documentation: GC Purity: 99+% Methyl Petroselinoate (6c) No. of C: 18:1 Documentation: GC Purity: 99+% Methyl Vaccenate (11c) No. of C: 18:1 Documentation: GC Purity: 99+% Methyl Transvaccenate (11tr) No. of C: 18:1 Documentation: GC Purity: 99+% Methyl Ricinoleate ( 9c, 12OH) No. of C: 18:1 Documentation: GC Purity: 99+% Methyl Ricinelaidate ( 9tr, 12OH) No. of C: 18:1 Documentation: GC Purity: 99+% Methyl Linoleate (9c, 12c) No. of C: 18:2 Documentation: GC Purity: 99+% Methyl Linoelaidate (9tr, 12tr) No. of C: 18:2 Documentation: GC Purity: 99+% Methyl Linolenate (9c, 12c, 15c) No. of C: 18:3 Documentation: GC Purity: 99+% Methyl Gamma Linolenate (6c, 9c, 12c) No. of C: 18:3 Documentation: GC Purity: 99+% Methyl Octadecatetraenoate (6c, 9c, 12c, 15c) (Methyl Stearidonate) No. of C: 18:4 100 mg x 100 mg mg x 25 mg g g g x 1 g mg x 100 mg mg x 100 mg mg x 100 mg mg x 100 mg mg x 100 mg g g mg x 100 mg mg x 100 mg mg x 100 mg mg info@larodan.se
18 Methyl 11c-Eicosenoate No. of C: 20:1 Documentation: GC Purity: 99+% Methyl 11tr-Eicosenoate No. of C: 20:1 Documentation: GC Purity: 99+% Methyl 11c,14c- Eicosadienoate No. of C: 20:2 Documentation: GC Purity: 99+% Methyl Eicosatrienoate (11c, 14c, 17c) No. of C: 20:3 Documentation: GC Purity: 99+% Methyl Hommo-gammalinolenate (8c, 11c, 14c) No. of C: 20:3 Documentation: GC Purity: 99+% Methyl Arachidonate (5c, 8c, 11c, 14c) No. of C: 20:4 Documentation: GC Purity: 99+% Omega3-Methyl Arachidonate (8c, 11c,14c, 17c ) No. of C: 20:4 Documentation: CoA Purity: 98% Methyl Eicosapentaenoate (5c, 8c, 11c, 14c, 17c) No. of C: 20:5 Documentation: GC Purity: 99+% Methyl Erucate (13c) No. of C: 22:1 Documentation: GC Purity: 99+% Methyl Brassidate ( 13tr) No. of C: 22:1 Documentation: GC Purity: 99+% Methyl Docosadienoate (13c, 16c) No. of C: 22:2 Documentation: Not included Purity: 99+% Methyl Docosatrienoate(13c, 16c, 19c) No. of C: 22:3 Documentation: Not included Purity: 99+% Methyl Docosatetraenoate (7c, 10c, 13c, 16c) No. of C: 22:4 Documentation: Not included Purity: 99+% Methyl Docosapentaenoate (4c,7c,10c,13c,16c) No. of C: 22:5 Documentation: GC Purity: 99+% 100 mg x 100 mg mg x 25 mg mg x 100 mg mg x 100 mg mg mg mg mg mg mg mg x 100 mg mg x 100 mg mg x 100 mg mg x 25 mg mg x 25 mg mg x 25 mg mg info@larodan.se
19 Methyl Docosapentaenoate ( 7c, 10c, 13c, 16c, 19c) No. of C: 22:5 Documentation: GC Purity: 99+% 100 mg x 100 mg Methyl Docosahexaenoate ( 4c, 7c, 10c, 13c, 16c, 19c) No. of C: 22:6 Documentation: GC Purity: 99+% Methyl Nervonate ( 15c) No. of C: 24:1 Documentation: GC Purity: 99+% Methyl (9c, 12c, 15c, 18c, 21c)-Tetracosapentaenoate No. of C: 24:5 Methyl (6c, 9c, 12c, 15c, 18c, 21c)-Tetracosahexaenoate No. of C: 24:6 100 mg x 100 mg mg x 100 mg mg mg CLAs Analytical Standards 9c,11tr-Octadecadienoic Acid 9c,11c-Octadecadienoic Acid Documentation: Not included Purity: 96+% 9tr,11tr-Octadecadienoic Acid Documentation: Not included Purity: 98% 10tr,12c-Octadecadienoic Acid Methyl 9c,11tr-Octadecadienoate Methyl 9c,11c-Octadecadienoate Documentation: Not included Purity: 96+% Methyl 9tr,11tr-Octadecadienoate Documentation: Not included Purity: 98% Methyl 10tr,12c-Octadecadienoate 25 mg g g mg g mg mg g g mg mg mg mg info@larodan.se
20 Unusual Fatty Acids Fatty acid biosynthesis by de novo pathways or by pathways involving elongation of preformed molecules takes place by successive addition of two carbons from either malonyl-coa or acetyl-coa, and this is the reason why the overwhelming part of naturally occurring fatty acids have even-numbered acyl chains. Furthermore, because of the specificities of acyl desaturases, most polyunsaturated fatty acids of natural origin possess two or more copies of the Z- configured element (-CH=CH-CH 2 -) n. For this reason even the simplest of the naturally occurring polyunsaturated fatty acids, i.e. linoleic acid (n = 2), will possess a bis-allylic methylene group located between double bonds: (-CH=CH- CH 2 -CH=CH-CH 2 -). The lowered carbon-hydrogen bond dissociation energy of this methylene group makes it unusually reactive and explains the susceptibility of polyunsaturated fatty acids to e.g. autoxidation and base-catalyzed isomerization as well as their conversions by enzymes like lipoxygenases, cyclooxygenases and conjugases. In addition to the dioxygenase and monooxygenase classes of enzymes, which introduce molecular oxygen into fatty acids to produce the oxylipin family of compounds, acyl chains can be modified by a large number of unorthodox desaturases. Such enzymes are especially common in the plant kingdom and are frequently active during seed development. Examples are the desaturases which introduce the 5(Z) double bond in 5(Z),9(Z)-octadecadienoic acid (taxoleic acid) and 5(Z),9(Z),12(Z)-octadecatrienoic acid (pinolenic acid), the desaturase which convert linoleic acid into 9(Z)-octadecen-12-ynoic acid (crepenynic acid), and the conjugase-type of desaturases which catalyze the dehydrogenation of linoleic acid into α-eleostearic, α-calendic and punicic acids. It should be noted that the fatty acids in this category are non-oxygenated, occur in esterified form in plant lipids, and are as a rule isolated together with conventional fatty acids. Larodan takes special interest in the isolation of uncommon fatty acids from natural origin and also applies synthetic methods for modification of existing fatty acids. In this way we can provide a collection of unusual fatty acids, most of which are not available from other commercial sources. Conjugated Polyene Fatty Acids / Methyl Esters Recent research has revealed new biological roles of Conjugated PUFAs, e.g. Conjugated Linoleic acid and alpha- Parinaric acid act as high affinity binding ligands and activators of peroxisomal proliferator-activated receptors (PPAR). Others (trienoic and tetraenoic acids) display potent cytotoxic activity in cell cultures. A review article on Conjugated PUFAs is available on our web site. 8(E),10(E)-Octadecadienoic Acid 8(E),10(E)-Octadecadienoic Acid Methyl Ester 11(E),13(E)-Octadecadienoic Acid 2 mg mg (E),13(E)-Octadecadienoic Acid Methyl Ester Documentation: GS-MS / HPLC Purity: 97+% 2 mg mg info@larodan.se
21 9(Z),11(E),13(E)-Octadecatrienoic Acid (alpha-eleostearic) 9(E),11(E),13(E)-Octadecatrienoic Acid (beta-eleostearic) 25 mg (E),10(E),12(Z)-Octadecatrienoic Acid (alpha-calendic) 25 mg (E),10(E),12(E)-Octadecatrienoic Acid (beta-calendic) 25 mg (Z),11(E),13(Z)-Octadecatrienoic Acid (Punicic) 25 mg (Z),10(E),12(Z)-Octadecatrienoic Acid (Jacaric) 25 mg (E),11(E),13(Z)-Octadecatrienoic Acid (Catalpic) 25 mg (Z),11(E),13(E),15(Z)-Octadecatetraenoic Acid (alpha-parinaric) 5 mg mg info@larodan.se
22 Odd-Numbered PUFAs Odd-numbered fatty acids are formed inter alia by the alpha-oxidation pathway. 8(Z),11(Z),14(Z)-Heptadecatrienoic Acid 10(Z),13(Z)-Nonadecadienoic acid 5 mg (Z),10(Z),13(Z)-Nonadecatrienoic Acid 5 mg (Z),13(Z),16(Z)-Nonadecatrienoic Acid 5 mg (Z),15(Z),18(Z)-Heneicosatrienoic Acid 5 mg (Z),12(Z),15(Z)-Heneicosatrienoic Acid 5 mg (Z),9(Z),12(Z),15(Z)-Heneicosatetraenoic Acid 5 mg (Z),9(Z),12(Z),15(Z),18(Z)-Heneicosapentaenoic Acid 5 mg (Z),11(Z),14(Z),17(Z),20(Z)-Tricosapentaenoic Acid 5 mg (Z),8(Z),11(Z),14(Z),17(Z),20(Z)-Tricosahexaenoic Acid 5 mg mg info@larodan.se
23 Odd-Numbered PUFA Methyl Esters 8(Z),11(Z),14(Z)-Heptadecatrienoic acid Methyl Ester 10(Z),13(Z)-Nonadecadienoic acid Methyl Ester 7(Z),10(Z),13(Z)-Nonadecatrienoic acid Methyl Ester 10(Z),13(Z),16(Z)-Nonadecatrienoic acid Methyl Ester 12(Z),15(Z),18(Z)-Heneicosatrienoic acid Methyl Ester 9(Z),12(Z),15(Z)-Heneicosatrienoic acid Methyl Ester 6(Z),9(Z),12(Z),15(Z)-Heneicosatetraenoic acid Methyl Ester 6(Z),9(Z),12(Z),15(Z),18(Z)-Heneicosapentaenoic acid Methyl Ester 8(Z),11(Z),14(Z),17(Z),20(Z)-Tricosapentaenoic acid Methyl Ester 5(Z),8(Z),11(Z),14(Z),17(Z),20(Z)-Tricosahexaenoic acid Methyl Ester 5 mg mg mg mg mg mg mg mg mg mg Other Unusual PUFAs 9(Z),12(Z)-Hexadecadienoic acid 5 mg (Z), 10(Z), 13(Z)-Hexadecatrienoic Acid 2(E)-Octadecenoic Acid 5 mg mg info@larodan.se
24 9(Z)-Octadecen-12-ynoic Acid (Crepenynic) 11(E)-Octadecen-9-ynoic Acid (Santalbic or Ximenynic) 25 mg (Z),12(E)-Octadecadienoic Acid 25 mg (E),12(Z)-Octadecadienoic Acid 5 mg b (Z),9(Z)-Octadecadienoic Acid (Taxoleic) 5 mg c (E),9(Z),12(Z)-Octadecatrienoic Acid (Columbinic) 5 mg (Z),9(Z),12(Z)-Octadecatrienoic Acid (Pinolenic) 5 mg Oxalinolenic Acid 5 mg (Z),9(Z)-Octadecadienoic Acid 2 mg (Z)-Eicosenoic Acid (Gadoleic) 2 mg (E),8(Z),11(Z),14(Z)-Eicosatetraenoic Acid 5 mg mg info@larodan.se
25 5(E),8(Z),11(Z),14(Z)-Eicosatetraenoic Acid (5(E)-Arachidonic) 5(Z),8Z),11(Z)-Eicosatrien-1,20-dioic acid 1 mg (Z),8(Z),11(Z)-Eicosatrienoic acid (Mead acid) Documentation: GC-MS / HPLC Purity: 98% 2 mg ,7-Eicosadiynoic acid 2 mg (E),10(Z),13(Z),16(Z)-Docosatetraenoic Acid 5 mg mg (E),7(Z),10(Z),13(Z),16(Z)-Docosapentaenoic Acid 5 mg (E),7(Z),10(Z),13(Z),16(Z),19(Z)-Docosahexaenoic Acid 5 mg (E),7(Z),10(Z),13(Z),16(Z),19(Z)-Docosahexaenoic Acid 15(E)-Tetracosenoic Acid (trans-nervonic) 1 mg mg (E),6(Z), 9(Z), 12(Z), 15(Z), 18(Z), 21(Z)-Tetracosaheptaenoic Acid 5 mg info@larodan.se
26 Other Unusual PUFA Methyl Esters Methyl 9(Z),12(Z)-Hexadecadienoate Methyl (7(Z), 10(Z), 13(Z))-Hexadecatrienoate 2(E)-Octadecenoic acid Methyl Ester 9(Z)-Octadecen-12-ynoic acid (Crepenynic) Methyl Ester 11(E)-Octadecen-9-ynoic acid (Santalbic or Ximenynic) Methyl Ester 9(Z),12(E)-Octadecadienoic acid Methyl Ester Documentation: GC-MS/HPLC Purity: 97+% 9(E),12(Z)-Octadecadienoic acid Methyl Ester 5(Z),9(Z)-Octadecadienoic acid (Taxoleic) Methyl Ester 5(E),9(Z),12(Z)-Octadecatrienoic acid (Columbinic) Methyl Ester 5(Z),9(Z),12(Z)-Octadecatrienoic acid (Pinolenic) Methyl Ester 6(Z),9(Z)-Octadecadienoic acid Methyl Ester 9(Z)-Eicosenoic acid (Gadoleic) Methyl Ester 2(E),8(Z),11(Z),14(Z)-Eicosatetraenoic acid Methyl Ester 5(E),8(Z),11(Z),14(Z)-Eicosatetraenoic acid (5(E)-Arachidonic) Methyl Ester 5(Z),8(Z),11(Z)-Eicosatrien-1,20-dioic acid Methyl Ester Documentation: GC-MS - HPLC Purity: 97+% 5(Z),8(Z),11(Z)-Eicosatrienoic acid (Mead acid) Methyl Ester Documentation: GC-MS / HPLC Purity: 98% 2(E),10(Z),13(Z),16(Z)-Docosatetraenoic acid Methyl Ester 5 mg mg mg mg mg mg b mg c mg mg mg mg mg mg mg mg mg mg info@larodan.se
27 2(E),7(Z),10(Z),13(Z),16(Z)-Docosapentaenoic acid Methyl Ester 2(E),7(Z),10(Z),13(Z),16(Z),19(Z)-Docosahexaenoic acid Methyl Ester 4(E),7(Z),10(Z),13(Z),16(Z),19(Z)-Docosahexaenoic acid Methyl Ester 15(E)-Tetracosenoic acid Methyl Ester (trans-nervonic) 2(E),6(Z), 9(Z), 12(Z), 15(Z),18(Z),21(Z)-Tetracosaheptaenoic Acid Methyl Ester 5 mg mg mg mg mg Omega-3 Fish Oil Fatty Acid Products EPA (5c,8c,11c,14c,17c) EPA Methyl Ester EPA Ethyl Ester Documentation: GC Purity: 90% Documentation: GC Purity: 90% Documentation: GC Purity: 90% DHA (4c,7c,10c,13c,16c,19c) Documentation: GC Purity: 90% DHA Methyl Ester DHA Ethyl Ester Documentation: GC Purity: 90% Documentation: GC Purity: 90% 1 g g g g g g g g g g g g g g g g g g info@larodan.se
28 CLAs Conjugated Linoleic Acids / Bulk Linoleic acids occur naturally as natural Octadienoic acid (9c,12c) and related isomers, so called Conjugated Linoleic Acid (CLA) having double bonds in 9,11 or 10,12 positions (cis/trans). CLA isomers have been shown to be biologically active, e.g. suppressing cancer diseases and inhibiting development of atherosclerosis. Larodan offers selected CLAs for analytical and larger scale experiments (bulk). The analytical standards are listed in the Fatty Acid section. CLAs in bulk Free Fatty Acids Methyl/ Ethyl Esters Triglycerides CLA 9c,11tr Free Fatty Acid Documentation: Data sheet Purity: 89-94% CLA 9c,11tr Methyl Ester CLA 9c,11tr Ethyl Ester CLA 9c,11tr Triglyceride Documentation: Data sheet Purity: 89-94% Documentation: Data sheet Purity: 89-94% Documentation: Data sheet Purity: 89-94% CLA 10tr,12c Free Fatty Acid Documentation: Data sheet Purity: 89-94% CLA 10tr,12c Methyl Ester Documentation: Data sheet Purity: 89-94% 1 g g g g g g g g g g g g g g g g g g g g g g g g g g g g g g info@larodan.se
29 CLA 10tr,12c Ethyl Ester CLA 10tr,12c Triglyceride Documentation: Data sheet Purity: 89-94% Documentation: Data sheet Purity: 89-94% 1 g g g g g g g g g g Oxylipins Fatty acid hydroperoxides can be enzymatically produced in plants by lipoxygenase- or α-dioxygenase-catalyzed oxygenation. Further conversion by secondary enzymes results in an array of oxygenated derivatives belonging to the oxylipin family of compounds. The name "oxylipin" was introduced in 1990 as an encompassing term for fatty acid derivatives of all chain lengths formed by mono- or dioxygenase-catalyzed oxygenations. Although it is a general term which is unspecific for lengths of acyl chains, it has so far found most use to denote linoleic and linolenic acid-derived products in the plant fatty acid field. Interest in oxylipins to a large part stems from their involvement in plant development and their role as defensive agents in plants attacked by fungi, insects and bacteria. The Larodan company has supplied oxylipins for a multitude of studies on plants oxylipins and is actively involved in research aimed at elucidating the antimicrobial effects of oxylipins as well as more specific projects dealing e.g. with the structures of complex lipids containing 12- oxophytodienoic acid and with the mechanism of the hydroperoxide lyase reaction. Many oxylipins are unstable and/or rapidly inactivated by metabolic degradation. Research at Larodan with the aim of obtaining oxylipin analogs with enhanced metabolic stability has recently resulted in a series of β-oxidation-stable 3-oxa oxylipins, and limited quantities of the precursor of such analogs, i.e. 3-oxalinolenic acid, is now available from Larodan. The almost hundred different oxylipins provided by Larodan range from fatty acid hydroperoxides produced by lipoxygenases (LOX) to a multitude of derivatives generated from hydroperoxides by chemical transformations or by secondary plant enzymes such as allene oxide synthase (AOS), divinyl ether synthase (DES), hydroperoxide lyase (HPL), epoxy alcohol synthase (EAS) and peroxygenase (POX). References and further information about Larodan s oxylipins are available at our website info@larodan.se
30 COOH DH or OX HO b COOH COOH POX or PX CHO O c 9-LOX HPL OHC COOH 9-LOX COOH HOO a Linoleic acid AOS EAS DES COOH O COOH HO O b + O HO a COOH + COOH O O + OH COOH HOOC O COOH HO O EH a a Oxygenation of linoleic acid by the 9-lipoxygenase pathway OH COOH 9-LOX, 9-lipoxygenase AOS, allene oxide synthase DES, divinyl ether synthase HPL, hydroperoxide lyase EAS, epoxy alcohol synthase DH, dehydrogenase OX, oxidase POX, peroxygenase PX, peroxidase EH, epoxide hydrolase HO HO OH + HO COOH OH info@larodan.se
31 COOH DH or OX OH COOH b O COOH 13-LOX POX or PX HPL CHO OHC COOH 13-LOX COOH OOH Linolenic acid AOS α-dox DES OOH COOH COOH O OH a + O COOH Spontaneous COOH HO O O COOH CHO COOH OH O a COOH Oxygenation of linolenic acid by the 13-lipoxygenase and α-dioxygenase pathways a 13-LOX, 13-lipoxygenase AOS, allene oxide synthase DES, divinyl ether synthase HPL, hydroperoxide lyase DH, dehydrogenase OX, oxidase POX, peroxygenase PX, peroxidase α-dox, α-dioxygenase 30 info@larodan.se
32 Hydroperoxides 13(S)-HPOD [13(S)-hydroperoxy-9(Z),11(E)-octadecadienoic acid] 1 mg a x 1 mg a LOX product of linoleic acid. Serves as the substrate for secondary enzymes like AOS, HPL, DES and POX. λ max (EtOH) 234 nm, ε 27,000 13(R,S)-Hydroperoxy-9(Z),11(E)-octadecadienoic Acid 100 microgram b Primary autoxidation product of linoleic acid. λ max (EtOH) 234 nm, ε 27,000 13(R,S)-Hydroperoxy-9(E),11(E)-octadecadienoic Acid 100 microgram c Primary autoxidation product of linoleic acid. λ max (EtOH) 230 nm. 9(S)-HPOD [9(S)-hydroperoxy-10(E),12(Z)-octadecadienoic acid] 1 mg a x 1 mg a LOX product of linoleic acid. Serves as the substrate for secondary enzymes like AOS, HPL, DES and POX. λ max (EtOH) 234 nm, ε = 27,000 9(R,S)-Hydroperoxy-10(E),12(Z)-octadecadienoic Acid 100 microgram c Primary autoxidation product of linoleic acid. λ max (EtOH) 234 nm, ε 27,000 9(R,S)-Hydroperoxy-10(E),12(E)-octadecadienoic Acid 100 microgram d Primary autoxidation product of linoleic acid. λ max (EtOH) 230 nm. info@larodan.se
33 13(S)-HPOT [13(S)-hydroperoxy-9(Z),11(E),15(Z)-octadecatrienoic acid] 1 mg x 1 mg LOX product of linolenic acid. Serves as the substrate for secondary enzymes like AOS, HPL, DES and POX. λ max (EtOH) 235 nm, ε 27,000 9(S)-HPOT [9(S)-hydroperoxy-10(E),12(Z),15(Z)-octadecatrienoic acid] 1 mg a x 1 mg a LOX product of linolenic acid. Serves as the substrate for secondary enzymes like AOS, HPL, DES and POX. λ max (EtOH) 235 nm, ε 27,000 11(S)-Hydroperoxy-7(Z),9(E),13(Z)-hexadecatrienoic Acid 100 microgram x 100 microgram LOX product of 7,10,13-hexadecatrienoic acid. λ max (EtOH) 235 nm, ε 27,000 2(R,S)-Hydroperoxy-9(Z)-octadecenoic Acid 100 microgram c Chemical oxygenation product of oleic acid. 2(R,S)-Hydroperoxyhexadecanoic acid 1mg Chemical oxygenation product of palmitic acid Hydroxides 13(S)-HOD [13(S)-hydroxy-9(Z),11(E)-octadecadienoic acid] 1 mg a x 1 mg a Chemical or enzymatical reduction product of the corresponding hydroperoxide. λ max (EtOH) 234 nm, ε 27, info@larodan.se
34 13(R,S)-Hydroxy-9(Z),11(E)-octadecadienoic acid 100 microgram f Chemical or enzymatical reduction product of the corresponding hydroperoxide. λ max (EtOH) 234 nm, ε 27,000 13(R,S)-Hydroxy-9(E),11(E)-octadecadienoic acid 100 microgram g Chemical or enzymatical reduction product of the corresponding hydroperoxide. λ max (EtOH) 230 nm 9(S)-HOD [9(S)-hydroxy-10(E),12(Z)-octadecadienoic acid] 1 mg b x 1 mg b Chemical or enzymatical reduction product of the corresponding hydroperoxide. λ max (EtOH) 234 nm, ε = 27,000 9(R,S)-Hydroxy-10(E),12(Z)-octadecadienoic acid 100 microgram e Chemical or enzymatical reduction product of the corresponding hydroperoxide. λ max (EtOH) 234 nm, ε 27,000 9(R,S)-Hydroxy-10(E),12(E)-octadecadienoic acid 100 microgram f Chemical or enzymatical reduction product of the corresponding hydroperoxide. λ max (EtOH) 230 nm 10(R,S)-Hydroxy-8(E),12(Z)-octadecadienoic Acid 100 microgram b Reduction product of hydroperoxide formed on singlet oxygen oxygenation of linoleic acid. 11(R)-Hydroxy-9(Z),12(Z)-octadecadienoic acid 100 microgram b Enzymatic bis-allylic oxidation product of linoleic acid. The methyl ester is supplied. info@larodan.se
35 11(R,S)-Hydroxy-9(Z),12(Z)-octadecadienoic acid 100 microgram c Nonenzymatic bis-allylic oxidation product of linoleic acid. The methyl ester is supplied. 12(R,S)-Hydroxy-9(Z),13(E)-octadecadienoic Acid 100 microgram c Reduction product of hydroperoxide formed on singlet oxygen oxygenation of linoleic acid. 15(R)-Hydroxy-9(Z),12(Z)-octadecadienoic Acid (Avenoleic) 100 microgram d x 100 microgram d Monooxygenase product of linoleic acid 15(R,S)-Hydroxy-9(Z),12(Z)-octadecadienoic acid 100 microgram e Reduction product of 15-oxolinoleic acid 13(S)-HOT [13(S)-hydroxy-9(Z),11(E),15(Z)-octadecatrienoic acid] 1 mg x 1 mg Chemical or enzymatical reduction product of the corresponding hydroperoxide. λ max (EtOH) 234 nm, ε 27,000 9(S)-HOT [9(S)-hydroxy-10(E),12(Z),15(Z)-octadecatrienoic acid] 1 mg b x 1 mg b Chemical or enzymatical reduction product of the corresponding hydroperoxide. λ max (EtOH) 235 nm, ε 27,000 13(S)-Hydroxy-6(Z),9(Z),11(E)-octadecatrienoic acid [13(S)-HOTω6] 1 mg Chemical or enzymatical reduction product of the 13(S)-hydroperoxide derivative of γ-linolenic acid. λ max (EtOH) 235 nm, ε 27, info@larodan.se
36 11(S)-Hydroxy-7(Z),9(E),13(Z)-hexadecatrienoic Acid 100 microgram x 100 microgram Chemical or enzymatical reduction product of the corresponding hydroperoxide. λ max (EtOH) 234 nm, ε 27,000 12(S)-Hydroxy-5(Z),8(E),10(E)-heptadecatrienoic acid [12(S)-HHT] 100 microgram x 100 microgram Degradation product of prostaglandin H 2. λ max (EtOH) 231 nm 2(R,S)-Hydroxy-9(Z)-octadecenoic acid 100 microgram d Chemical or enzymatical reduction product of the corresponding hydroperoxide. 2(R)-Hydroxy-9(Z),12(Z),15(Z)-octadecatrienoic acid 5 mg a Secondary α-dioxygenase product of linolenic acid 2(R,S)-Hydroxy-9(Z),12(Z),15(Z)-octadecatrienoic acid 1 mg b Reduction product of 2-oxolinolenic acid Ketones 13-Oxo-9(Z),11(E)-octadecadienoic Acid [13-KOD] 1 mg b x 1 mg b Secondary 13-LOX product of linoleic acid. λ max (EtOH) 278 nm, ε 24,000 9-Oxo-10(E),12(Z)-octadecadienoic Acid [ 9-KOD] 1 mg c x 1 mg c Secondary 9-LOX product of linoleic acid λ max (EtOH) 278 nm, ε 24,000 info@larodan.se
37 13-Oxo-9(Z),11(E),15(Z)-octadecatrienoic Acid [13-KOT] 1 mg b Secondary 13-LOX product of linolenic acid. λ max (EtOH) 280 nm, ε 24,000 9-Oxo-10(E),12(Z),15(Z)-octadecatrienoic Acid [9-KOT] 1 mg c x 1 mg c Secondary 9-LOX product of linolenic acid. λ max (EtOH) 278 nm, ε 24,000 2-Oxo-9(Z),12(Z),15(Z)-octadecatrienoic acid 1 mg Secondary α-dioxygenase product of linolenic acid. 15-Oxo-9(Z),12(Z)-octadecadienoic acid 100 microgram Dehydrogenation product of avenoleic acid. Epoxides (±)-cis-12,13-epoxy-9(z)-octadecenoic Acid ((±)-Vernolic) 1 mg a Epoxidation product of linoleic acid 12(S),13(R)-Epoxy-9(Z)-octadecenoic Acid ((+)-Vernolic) 25 mg b Methyl ester 25 mg b Epoxygenase/peroxygenase product of linoleic acid info@larodan.se
38 12(R),13(S)-Epoxy-9(Z)-octadecenoic Acid ((-)-Vernolic) 5 mg c Epoxygenase/peroxygenase product of linoleic acid. (±)-cis-9,10-epoxy-12(z)-octadecenoic Acid ((±)-Coronaric) 1 mg d Epoxidation product of linoleic acid. 9(R),10(S)-Epoxy-12(Z)-octadecenoic Acid ((+)-Coronaric) 5 mg e Epoxygenase/peroxygenase product of linoleic acid. (±)-cis-12,13-epoxy-9(z),15(z)-octadecadienoic Acid 1 mg a Epoxidation product of linolenic acid. (±)-trans-5,6-epoxy-9(z),12(z)-octadecadienoic Acid 1 mg b Epoxidation product of columbinic acid. (±)-cis-5,6-epoxy-9(z),12(z)-octadecadienoic acid 1 mg c Epoxidation product of pinolenic acid. (±)-cis-11,12-epoxy-14(z)-eicosenoic Acid 1mg Epoxidation product of 11,14-eicosadienoic acid. info@larodan.se
39 (±)-cis-14,15-epoxy-11(z)-eicosenoic Acid ((±)-Alchornic) 1 mg Epoxidation product of 11,14-eicosadienoic acid. (±)-cis-4,5-epoxy-7(z),10(z),13(z),16(z),19(z)-docosapentaenoic acid 1 mg Epoxidation product of docosahexaenoic acid. The methyl ester is supplied. 10,13-epoxy-10,12-octadecadienoic Acid 1 mg Nonenzymatical degradation product of many oxylipins. Diols (±)-threo-9,10-dihydroxy-12(z)-octadecenoic Acid 5 mg a Chemical or enzymatical hydrolysis product of coronaric acid. (±)-threo-12,13-dihydroxy-9(z)-octadecenoic Acid 5 mg b Chemical or enzymatical hydrolysis product of vernolic acid. (±)-threo-12,13-dihydroxy-9(z),15(z)-octadecadienoic Acid 1 mg Chemical or enzymatical hydrolysis product of cis-12,13-epoxy-9(z),15(z)-octadecadienoic acid info@larodan.se
Tel. +49(0) # Fax +49(0) # Katalog mit den Produkten der Firma Larodan, die wir vertreten.
 Katalog mit den Produkten der Firma Larodan, die wir vertreten. Inhalt Alphabetischer Index Seitennummer Acyl-L-Carnitines Native 50 Deuterium Labelled 52 Bile Acids & Methyl Esters 152 Cholesterol & Esters
Katalog mit den Produkten der Firma Larodan, die wir vertreten. Inhalt Alphabetischer Index Seitennummer Acyl-L-Carnitines Native 50 Deuterium Labelled 52 Bile Acids & Methyl Esters 152 Cholesterol & Esters
Fats and Lipids (Ans570)
 Fats and Lipids (Ans570) Outlines Fats and Lipids Structure, nomenclature Phospholipids, Sterols, and Lipid Derivatives Lipid Oxidation Roles of fat in food processing and dietary fat Lipid and fat analysis:
Fats and Lipids (Ans570) Outlines Fats and Lipids Structure, nomenclature Phospholipids, Sterols, and Lipid Derivatives Lipid Oxidation Roles of fat in food processing and dietary fat Lipid and fat analysis:
CERTIFICATE OF ANALYSIS AR-15-QD
 Eurofins Sample Code: 464-2015-09040561 Sample Description: Fermented Cod Liver Oil Client Sample Code: 29442 PO Number: Credit Card on file Client Code: QD0006557 attn: Dr. Jie Zhang Nutrition Analysis
Eurofins Sample Code: 464-2015-09040561 Sample Description: Fermented Cod Liver Oil Client Sample Code: 29442 PO Number: Credit Card on file Client Code: QD0006557 attn: Dr. Jie Zhang Nutrition Analysis
Highly Reproducible Detailed cis/trans FAMEs Analysis Ensured by New Optimized Rt-2560 Column Manufacturing and Application-Specific QC Test
 Highly Reproducible Detailed cis/trans FAMEs Analysis Ensured by New Optimized Rt-2560 Manufacturing and Application-Specific QC Test By Kristi Sellers and Rebecca Stevens Restek s Rt-2560 GC column is
Highly Reproducible Detailed cis/trans FAMEs Analysis Ensured by New Optimized Rt-2560 Manufacturing and Application-Specific QC Test By Kristi Sellers and Rebecca Stevens Restek s Rt-2560 GC column is
MPS Advanced Plant Biochemistry Course
 MPS 587 - Advanced Plant Biochemistry Course Lipids section guest lecture: Philip Bates Post-doc Browse lab Office: 453 Clark Hall Email: phil_bates@wsu.edu Lecture 9 Lipids I 1. What is a lipid? 2. Fatty
MPS 587 - Advanced Plant Biochemistry Course Lipids section guest lecture: Philip Bates Post-doc Browse lab Office: 453 Clark Hall Email: phil_bates@wsu.edu Lecture 9 Lipids I 1. What is a lipid? 2. Fatty
Trans FAT LABELING an Industry Perspective
 Trans FAT LABELING an Industry Perspective National Nutrition Database Conference 28 th Edition June 24-26, 2004 Iowa City, IA Trans FAT LABELING an Industry Perspective Christine Wold QRO Product Labeling
Trans FAT LABELING an Industry Perspective National Nutrition Database Conference 28 th Edition June 24-26, 2004 Iowa City, IA Trans FAT LABELING an Industry Perspective Christine Wold QRO Product Labeling
NEW! 200 m GC Columns for Detailed Analysis of cis/trans FAME Isomers
 Order: 00--00 (U.S.) -- (Global) NEW! 00 m GC Columns for Detailed Analysis of cis/trans FAME Isomers Leonard M. Sidisky, R&D Manager; and Michael D. Buchanan, Product Manager mike.buchanan@sial.com Over
Order: 00--00 (U.S.) -- (Global) NEW! 00 m GC Columns for Detailed Analysis of cis/trans FAME Isomers Leonard M. Sidisky, R&D Manager; and Michael D. Buchanan, Product Manager mike.buchanan@sial.com Over
Biological role of lipids
 Lipids Lipids Organic compounds present in living organisms, insoluble in water but able to be extracted by organic solvents such as: chloroform, acetone, benzene. Extraction = the action of taking out
Lipids Lipids Organic compounds present in living organisms, insoluble in water but able to be extracted by organic solvents such as: chloroform, acetone, benzene. Extraction = the action of taking out
ANSC 619 PHYSIOLOGICAL CHEMISTRY OF LIVESTOCK SPECIES. Lipid Chemistry NO. OF CARBONS COMMON NAME GENEVA NAME STRUCTURE
 ANSC 619 PHYSIOLOGICAL CHEMISTRY OF LIVESTOCK SPECIES I. Common Saturated Fatty Acids NO. OF CARBONS COMMON NAME GENEVA NAME STRUCTURE 4 Butyric Tetranoic CH 3 (CH 2 ) 2 COOH 6 Caproic Hexanoic CH 3 (CH
ANSC 619 PHYSIOLOGICAL CHEMISTRY OF LIVESTOCK SPECIES I. Common Saturated Fatty Acids NO. OF CARBONS COMMON NAME GENEVA NAME STRUCTURE 4 Butyric Tetranoic CH 3 (CH 2 ) 2 COOH 6 Caproic Hexanoic CH 3 (CH
Column Selection for the Analysis of Fatty Acid Methyl Esters Application
 Column Selection for the Analysis of Fatty Acid Methyl Esters Application Food Analysis Authors Frank David Research Institute for Chromatography President Kennedy Park B- Kortrijk, Belgium Pat Sandra
Column Selection for the Analysis of Fatty Acid Methyl Esters Application Food Analysis Authors Frank David Research Institute for Chromatography President Kennedy Park B- Kortrijk, Belgium Pat Sandra
Erythrocyte Essential Fatty Acids
 Biolab reference: PHWN\ABCD\A12 Referred by: Dr.. Patient: Mr Sample Patient Date of birth: 16/01/969 Your reference: Date: 04/01/2012 Sex: Male Sample date: 03/01/2012 Erythrocyte Essential Fatty Acids
Biolab reference: PHWN\ABCD\A12 Referred by: Dr.. Patient: Mr Sample Patient Date of birth: 16/01/969 Your reference: Date: 04/01/2012 Sex: Male Sample date: 03/01/2012 Erythrocyte Essential Fatty Acids
Optimising. membranes
 Optimising Neuronal membranes Lipids are classified as 1. Simple lipids oils and fats 2. Complex lipids a) Phospholipids b) Glycosphingolipids containing a fatty acid, sphingosine and a CHO c) Lipoproteins
Optimising Neuronal membranes Lipids are classified as 1. Simple lipids oils and fats 2. Complex lipids a) Phospholipids b) Glycosphingolipids containing a fatty acid, sphingosine and a CHO c) Lipoproteins
Introduction to the Study of Lipids
 Introduction to the Study of Lipids Factors to Consider in the Study of Biomolecules What are the features of the basic building blocks? (ex: monosaccharides, alcohols, fatty acids, amino acids) 1) General
Introduction to the Study of Lipids Factors to Consider in the Study of Biomolecules What are the features of the basic building blocks? (ex: monosaccharides, alcohols, fatty acids, amino acids) 1) General
Certificate of Analysis Standard Reference Material 3275
 National Institute of Standards & Technology Certificate of Analysis Standard Reference Material 3275 Omega-3 and Omega-6 Fatty Acids in Fish Oil This Standard Reference Material (SRM) is intended primarily
National Institute of Standards & Technology Certificate of Analysis Standard Reference Material 3275 Omega-3 and Omega-6 Fatty Acids in Fish Oil This Standard Reference Material (SRM) is intended primarily
Chapter 8. Functions of Lipids. Structural Nature of Lipids. BCH 4053 Spring 2001 Chapter 8 Lecture Notes. Slide 1. Slide 2.
 BCH 4053 Spring 2001 Chapter 8 Lecture Notes 1 Chapter 8 Lipids 2 Functions of Lipids Energy Storage Thermal Insulation Structural Components of Membranes Protective Coatings of Plants and Insects Hormonal
BCH 4053 Spring 2001 Chapter 8 Lecture Notes 1 Chapter 8 Lipids 2 Functions of Lipids Energy Storage Thermal Insulation Structural Components of Membranes Protective Coatings of Plants and Insects Hormonal
Analysis of FAMEs Using Cold EI GC/MS for Enhanced Molecular Ion Selectivity
 APPLICATION NOTE Gas Chromatography/ Mass Spectrometry Author: Adam Patkin PerkinElmer, Inc. Shelton, CT Analysis of FAMEs Using GC/MS for Enhanced Molecular Ion Selectivity Introduction Characterization
APPLICATION NOTE Gas Chromatography/ Mass Spectrometry Author: Adam Patkin PerkinElmer, Inc. Shelton, CT Analysis of FAMEs Using GC/MS for Enhanced Molecular Ion Selectivity Introduction Characterization
ANSC 689 PHYSIOLOGICAL CHEMISTRY OF LIVESTOCK SPECIDS General Chemistry of Fatty Acids and Triacylglycerols
 ANSC 689 PHYSIOLOGICAL CHEMISTRY OF LIVESTOCK SPECIDS General Chemistry of Fatty Acids and Triacylglycerols I. Common Saturated Fatty Acids NO. OF CARBONS COMMON NAME GENEVA NAME STRUCTURE 4 Butyric Tetranoic
ANSC 689 PHYSIOLOGICAL CHEMISTRY OF LIVESTOCK SPECIDS General Chemistry of Fatty Acids and Triacylglycerols I. Common Saturated Fatty Acids NO. OF CARBONS COMMON NAME GENEVA NAME STRUCTURE 4 Butyric Tetranoic
Structure SMILES IUPAC name Name MW Plate description CAS Catalog number Concentration Solvent Plate part number
 H CCCCCCCCCC()= decanoic acid Decanoic acid 1723 Fatty Acid Library 334-48-5 BL-135 10mM DMS 2803 H CCCCCCCCCCC()= undecanoic acid Undecanoic acid 1863 Fatty Acid Library 112-37-8 BL-136 10mM DMS 2803
H CCCCCCCCCC()= decanoic acid Decanoic acid 1723 Fatty Acid Library 334-48-5 BL-135 10mM DMS 2803 H CCCCCCCCCCC()= undecanoic acid Undecanoic acid 1863 Fatty Acid Library 112-37-8 BL-136 10mM DMS 2803
DET REPORT NO. 69 JUNE 2015
 1.) THINKING BEYOND THE NPD BOX - INEXPENSIVE CONVERSION OF NPD EQUIPMENT TO MULTIPLE MODES OF SELECTIVE GC DETECTION. 2.) GC-CCID DIFFERENTIATION BETWEEN SATURATE VS. UNSATURATE AND MONO-UNSATURATE VS.
1.) THINKING BEYOND THE NPD BOX - INEXPENSIVE CONVERSION OF NPD EQUIPMENT TO MULTIPLE MODES OF SELECTIVE GC DETECTION. 2.) GC-CCID DIFFERENTIATION BETWEEN SATURATE VS. UNSATURATE AND MONO-UNSATURATE VS.
Improving the Analysis of 37 Fatty Acid Methyl Esters
 Application Note Food Testing Improving the Analysis of 37 Fatty Acid Methyl Esters Using Three Types of Capillary GC columns Authors Yun Zou Agilent Technologies (Shanghai) Co.Ltd, Shanghai 200131 P.R.China
Application Note Food Testing Improving the Analysis of 37 Fatty Acid Methyl Esters Using Three Types of Capillary GC columns Authors Yun Zou Agilent Technologies (Shanghai) Co.Ltd, Shanghai 200131 P.R.China
OBJECTIVE. Lipids are largely hydrocarbon derivatives and thus represent
 Paper 4. Biomolecules and their interactions Module 20: Saturated and unsaturated fatty acids, Nomenclature of fatty acids and Essential and non-essential fatty acids OBJECTIVE The main aim of this module
Paper 4. Biomolecules and their interactions Module 20: Saturated and unsaturated fatty acids, Nomenclature of fatty acids and Essential and non-essential fatty acids OBJECTIVE The main aim of this module
Lipids are broadly classified in to simple, complex and derived, which are further subdivided into different groups.
 Paper No. 01 Paper Title: Food Chemistry Module -9: Classification of lipids Lipids are organic substances which are insoluble in water and soluble in organic solvents. Lipids are not polymers and exist
Paper No. 01 Paper Title: Food Chemistry Module -9: Classification of lipids Lipids are organic substances which are insoluble in water and soluble in organic solvents. Lipids are not polymers and exist
Lipids and Classification:
 Lipids and Classification: Lipids: Biological lipids are a chemically diverse group of organic compounds which are insoluble or only poorly soluble in water. They are readily soluble in non-polar solvents
Lipids and Classification: Lipids: Biological lipids are a chemically diverse group of organic compounds which are insoluble or only poorly soluble in water. They are readily soluble in non-polar solvents
High-Resolution GC Analyses of Fatty Acid Methyl Esters (FAMEs)
 High-Resolution GC Analyses of Fatty Acid Methyl Esters (s) Fatty acid methyl esters (s) analysis is an important tool both for characterizing fats and oils and for determining the total fat content in
High-Resolution GC Analyses of Fatty Acid Methyl Esters (s) Fatty acid methyl esters (s) analysis is an important tool both for characterizing fats and oils and for determining the total fat content in
FOOD TECHNOLOGY FACT SHEET
 FAPC-196 Robert M. Kerr Food & Agricultural Products Center Nurhan Turgut Dunford FAPC Oil/oilseed Specialist FOOD TECHNOLOGY FACT SHEET Adding Value to OKLAHOMA 405-744-6071 www.fapc.biz fapc@okstate.edu
FAPC-196 Robert M. Kerr Food & Agricultural Products Center Nurhan Turgut Dunford FAPC Oil/oilseed Specialist FOOD TECHNOLOGY FACT SHEET Adding Value to OKLAHOMA 405-744-6071 www.fapc.biz fapc@okstate.edu
Food Analysis Standards
 Food Chemists routinely use AccuStandard's comprehensive program of Analytical Reference Materials for their Food Analysis. These include, Vitamin Standards, Preservative Standards and Antimicrobial Standards.
Food Chemists routinely use AccuStandard's comprehensive program of Analytical Reference Materials for their Food Analysis. These include, Vitamin Standards, Preservative Standards and Antimicrobial Standards.
Separation of 37 Fatty Acid Methyl Esters Utilizing a High-Efficiency 10 m Capillary GC Column with Optimization in Three Carrier Gases
 APPLICATION NOTE Separation of 37 Fatty Acid Methyl Esters Utilizing a High-Efficiency 10 m Capillary GC Column with Optimization in Three Carrier Gases No. 21557 Aaron L. Lamb Thermo Fisher Scientific,
APPLICATION NOTE Separation of 37 Fatty Acid Methyl Esters Utilizing a High-Efficiency 10 m Capillary GC Column with Optimization in Three Carrier Gases No. 21557 Aaron L. Lamb Thermo Fisher Scientific,
Dr. Nafith Abu Tarboush
 4 Dr. Nafith Abu Tarboush June 24 th 2013 Ahmad Moayd 1 Definition and general properties refer to slide no. 2 Lipids: macromolecules made from Alcohol and Fatty acid bonded by ester linkage. Amphipathic
4 Dr. Nafith Abu Tarboush June 24 th 2013 Ahmad Moayd 1 Definition and general properties refer to slide no. 2 Lipids: macromolecules made from Alcohol and Fatty acid bonded by ester linkage. Amphipathic
FAMEs Analyses High-Resolution GC Analyses of Fatty Acid Methyl Esters
 APPLICATIONS NOTE FAMEs Analyses High-Resolution GC Analyses of Fatty Acid Methyl Esters Fatty acid methyl esters (FAMEs) analysis is an important tool both for characterizing fats and oils and for determining
APPLICATIONS NOTE FAMEs Analyses High-Resolution GC Analyses of Fatty Acid Methyl Esters Fatty acid methyl esters (FAMEs) analysis is an important tool both for characterizing fats and oils and for determining
3 Lipids. 3.1 Foreword
 3 Lipids 3.1 Foreword Lipids are formed from structural units with a pronounced hydrophobicity. This solubility characteristic, rather than a common structural feature, is unique for this class of compounds.
3 Lipids 3.1 Foreword Lipids are formed from structural units with a pronounced hydrophobicity. This solubility characteristic, rather than a common structural feature, is unique for this class of compounds.
AOAC Official Method Determination of Labeled Fatty Acids Content in Milk Products and Infant Formula
 AOAC Official Method 2012.13 Determination of Labeled Fatty Acids Content in Milk Products and Infant Formula Capillary Gas Chromatography First Action 2012 A. Scope The method involves the quantification
AOAC Official Method 2012.13 Determination of Labeled Fatty Acids Content in Milk Products and Infant Formula Capillary Gas Chromatography First Action 2012 A. Scope The method involves the quantification
Physical properties. Carboxylic acids. Boiling points of Carboxylic Acids 3/19/2012
 R H Carboxylic acid Carboxylic acids Ionization R _ Carboxylate ion + H (proton) 1 Physical properties Carboxylic acids are polar, and form hydrogen bonds with each other. At high temperatures, in vapour
R H Carboxylic acid Carboxylic acids Ionization R _ Carboxylate ion + H (proton) 1 Physical properties Carboxylic acids are polar, and form hydrogen bonds with each other. At high temperatures, in vapour
Definition: Water insoluble No common structure (though generally large R groups)
 Lipids Definition Definition: Water insoluble No common structure (though generally large R groups) Water Solubility (Hydrophilic) What makes molecules water soluble (hydrophilic)? Like dissolves like
Lipids Definition Definition: Water insoluble No common structure (though generally large R groups) Water Solubility (Hydrophilic) What makes molecules water soluble (hydrophilic)? Like dissolves like
Biosynthesis of Triacylglycerides (TG) in liver. Mobilization of stored fat and oxidation of fatty acids
 Biosynthesis of Triacylglycerides (TG) in liver Mobilization of stored fat and oxidation of fatty acids Activation of hormone sensitive lipase This enzyme is activated when phosphorylated (3,5 cyclic AMPdependent
Biosynthesis of Triacylglycerides (TG) in liver Mobilization of stored fat and oxidation of fatty acids Activation of hormone sensitive lipase This enzyme is activated when phosphorylated (3,5 cyclic AMPdependent
Classification, functions and structure
 Classification, functions and structure Elena Rivneac PhD, Associate Professor Department of Biochemistry and Clinical Biochemistry State University of Medicine and Pharmacy "Nicolae Testemitanu" Lipids
Classification, functions and structure Elena Rivneac PhD, Associate Professor Department of Biochemistry and Clinical Biochemistry State University of Medicine and Pharmacy "Nicolae Testemitanu" Lipids
Experiment 12 Lipids. Structures of Common Fatty Acids Name Number of carbons
 Experiment 12 Lipids Lipids are a class of biological molecules that are insoluble in water and soluble in nonpolar solvents. There are many different categories of lipids and each category has different
Experiment 12 Lipids Lipids are a class of biological molecules that are insoluble in water and soluble in nonpolar solvents. There are many different categories of lipids and each category has different
ANSC/NUTR 618 Lipids & Lipid Metabolism
 I. Overall concepts A. Definitions ANC/NUTR 618 Lipids & Lipid Metabolism 1. De novo synthesis = synthesis from non-fatty acid precursors a. Carbohydrate precursors (glucose, lactate, and pyruvate) b.
I. Overall concepts A. Definitions ANC/NUTR 618 Lipids & Lipid Metabolism 1. De novo synthesis = synthesis from non-fatty acid precursors a. Carbohydrate precursors (glucose, lactate, and pyruvate) b.
CHEMISTRY OF OILS AND FATS. Albert J. Dijkstra
 CHEMISTRY F ILS AND FATS Albert J. Dijkstra Production vegetable oil (m tonnes) Seven oil seeds 2008/09 2009/10 2010/11 2011/12 total 396.7 444.1 455.7 441.4 Soya bean 211.6 260.2 263.6 238.7 ther 185.1
CHEMISTRY F ILS AND FATS Albert J. Dijkstra Production vegetable oil (m tonnes) Seven oil seeds 2008/09 2009/10 2010/11 2011/12 total 396.7 444.1 455.7 441.4 Soya bean 211.6 260.2 263.6 238.7 ther 185.1
Lipids and Membranes
 Lipids and Membranes Presented by Dr. Mohammad Saadeh The requirements for the Pharmaceutical Biochemistry I Philadelphia University Faculty of pharmacy Lipids and Membranes I. overview Lipids are related
Lipids and Membranes Presented by Dr. Mohammad Saadeh The requirements for the Pharmaceutical Biochemistry I Philadelphia University Faculty of pharmacy Lipids and Membranes I. overview Lipids are related
ANSC/NUTR 618 LIPIDS & LIPID METABOLISM. Fatty Acid Elongation and Desaturation
 ANSC/NUTR 618 LIPIDS & LIPID METABOLISM I. Fatty acid elongation A. General 1. At least 60% of fatty acids in triacylglycerols are C18. 2. Free palmitic acid (16:0) synthesized in cytoplasm is elongated
ANSC/NUTR 618 LIPIDS & LIPID METABOLISM I. Fatty acid elongation A. General 1. At least 60% of fatty acids in triacylglycerols are C18. 2. Free palmitic acid (16:0) synthesized in cytoplasm is elongated
Fats Can Be Good For You!
 Fats Can Be Good For You! Fat Quiz (True or False) 1. Excess fat in the diet is a major cause of heart disease. 2. Saturated fats raise cholesterol levels. 3. The saturated fats in butter and coconut oil
Fats Can Be Good For You! Fat Quiz (True or False) 1. Excess fat in the diet is a major cause of heart disease. 2. Saturated fats raise cholesterol levels. 3. The saturated fats in butter and coconut oil
FATTY ACIDS IN PLASMA BY GC/MS - Code GC75010
 FATTY ACIDS IN PLASMA BY GC/MS - Code GC75010 BIOCHEMISTRY The term fatty acids (abbreviation FA, English Fatty Acids) are indicated aliphatic monocarboxylic acids. They are, with few exceptions, long
FATTY ACIDS IN PLASMA BY GC/MS - Code GC75010 BIOCHEMISTRY The term fatty acids (abbreviation FA, English Fatty Acids) are indicated aliphatic monocarboxylic acids. They are, with few exceptions, long
Lipids ON HPLC COLUMNS
 Lipids N HPLC CLUMNS No.TI17E cis - trans Isomers of 9-octadecenoic acid 2 1 1 2 mau 5 1 15 2 min Cadenza CD-C18, 15 x 4.6 mm ACN / water / formic acid = 9 / 1 /.5.8 ml/min, 37 C UV at 215 nm, 3.7 MPa
Lipids N HPLC CLUMNS No.TI17E cis - trans Isomers of 9-octadecenoic acid 2 1 1 2 mau 5 1 15 2 min Cadenza CD-C18, 15 x 4.6 mm ACN / water / formic acid = 9 / 1 /.5.8 ml/min, 37 C UV at 215 nm, 3.7 MPa
Lipids. Dr. Mamoun Ahram Summer semester, Resources This lecture Campbell and Farrell s Biochemistry, Chapter 8
 Lipids Dr. Mamoun Ahram Summer semester, 2017-2018 Resources This lecture Campbell and Farrell s Biochemistry, Chapter 8 Lipids Lipids are a heterogeneous class of naturally occurring organic compounds
Lipids Dr. Mamoun Ahram Summer semester, 2017-2018 Resources This lecture Campbell and Farrell s Biochemistry, Chapter 8 Lipids Lipids are a heterogeneous class of naturally occurring organic compounds
Introduction to Lipid Chemistry
 Introduction to Lipid Chemistry Benjamin Schwartz Ontario SCC Education Day September 18, 2018 Lipid knowledge for the personal care industry What is a Lipid? Lipids are fatty acids and their derivatives,
Introduction to Lipid Chemistry Benjamin Schwartz Ontario SCC Education Day September 18, 2018 Lipid knowledge for the personal care industry What is a Lipid? Lipids are fatty acids and their derivatives,
N a t i o n a l M e a s u r e m e n t I n s t i t u t e
 Page: 1 of 5 Client : KIMBERLEY WILD GUBINGE Job No. : KIMB10/170116 MUNGET ABORIGINAL CORPORATION Quote No. : QT-02094 PENDER BAY Order No. : DAMPIER PENINSULA WA 6725 Date Sampled : Date Received : 16-JAN-2017
Page: 1 of 5 Client : KIMBERLEY WILD GUBINGE Job No. : KIMB10/170116 MUNGET ABORIGINAL CORPORATION Quote No. : QT-02094 PENDER BAY Order No. : DAMPIER PENINSULA WA 6725 Date Sampled : Date Received : 16-JAN-2017
Supplementary Information
 Supplementary Information Serum and synovial fluid lipidomic profiles predict obesity-associated osteoarthritis, synovitis, and wound repair Chia-Lung Wu 1,2, Kelly A. Kimmerling 1,2, Dianne Little 3,
Supplementary Information Serum and synovial fluid lipidomic profiles predict obesity-associated osteoarthritis, synovitis, and wound repair Chia-Lung Wu 1,2, Kelly A. Kimmerling 1,2, Dianne Little 3,
Chem 5 PAL Worksheet Lipids Smith text Chapter 15
 Chem 5 PAL Worksheet Lipids Smith text Chapter 15 Principle: Fatty acids are carboxylic acids with long (usually > 14) carbon chains which can be saturated (no carbon-carbon double bonds) are unsaturated
Chem 5 PAL Worksheet Lipids Smith text Chapter 15 Principle: Fatty acids are carboxylic acids with long (usually > 14) carbon chains which can be saturated (no carbon-carbon double bonds) are unsaturated
INTEGRATIVE MEDICINE BLOOD - EDTA Result Range Units
 D Collected : 00-00-0000 INTEGRATIVE MEDICINE BLOOD - EDTA Result Range Units RED CELL FATTY ACID PROFILE Red Cell Fatty Acid Summary Saturated Fats, Total 36.99 19.30-39.40 Monounsaturated Fats, Total
D Collected : 00-00-0000 INTEGRATIVE MEDICINE BLOOD - EDTA Result Range Units RED CELL FATTY ACID PROFILE Red Cell Fatty Acid Summary Saturated Fats, Total 36.99 19.30-39.40 Monounsaturated Fats, Total
REP15/FO Appendix III 1. PROPOSED DRAFT CODEX STANDARD FOR FISH OILS (at Step 5 of the Procedure)
 REP15/FO Appendix III 1 PROPOSED DRAFT CODEX STANDARD FOR FISH OILS (at Step 5 of the Procedure) Appendix III 1. Scope This Standard applies to the fish oils described in section 2 that are presented in
REP15/FO Appendix III 1 PROPOSED DRAFT CODEX STANDARD FOR FISH OILS (at Step 5 of the Procedure) Appendix III 1. Scope This Standard applies to the fish oils described in section 2 that are presented in
Chapter 28: Lipids In a slightly different order than in your textbook Fatty Acids
 common name hapter 28: Lipids In a slightly different order than in your textbook Fatty Acids shorthand notations #of : # of = #of, location of = line structure comments butyric acid 4:0 in butterfat lauric
common name hapter 28: Lipids In a slightly different order than in your textbook Fatty Acids shorthand notations #of : # of = #of, location of = line structure comments butyric acid 4:0 in butterfat lauric
Lecture: 26 OXIDATION OF FATTY ACIDS
 Lecture: 26 OXIDATION OF FATTY ACIDS Fatty acids obtained by hydrolysis of fats undergo different oxidative pathways designated as alpha ( ), beta ( ) and omega ( ) pathways. -oxidation -Oxidation of fatty
Lecture: 26 OXIDATION OF FATTY ACIDS Fatty acids obtained by hydrolysis of fats undergo different oxidative pathways designated as alpha ( ), beta ( ) and omega ( ) pathways. -oxidation -Oxidation of fatty
FATTY ACIDS (FAs) SIMPLE AND COMPLEX LIPIDS
 FATTY ACIDS (FAs) SIMPLE AND COMPLEX LIPIDS Dicarboxylic acids, ketone bodies. Department of General Chemistry Structure and classification of lipids Lipids can be divided into five categories, on the
FATTY ACIDS (FAs) SIMPLE AND COMPLEX LIPIDS Dicarboxylic acids, ketone bodies. Department of General Chemistry Structure and classification of lipids Lipids can be divided into five categories, on the
Factors to Consider in the Study of Biomolecules
 Factors to Consider in the Study of Biomolecules What are the features of the basic building blocks? (ex: monosaccharides, alcohols, fatty acids, amino acids) 1) General structure and functional groups
Factors to Consider in the Study of Biomolecules What are the features of the basic building blocks? (ex: monosaccharides, alcohols, fatty acids, amino acids) 1) General structure and functional groups
Chapter 11: Lipids. Voet & Voet: Pages
 Chapter 11: Lipids Voet & Voet: Pages 380-394 Slide 1 Lipids Lipids are distinguished by their high solubility in non polar solvents and low solubility in H2O Diverse group of compounds including Fats,
Chapter 11: Lipids Voet & Voet: Pages 380-394 Slide 1 Lipids Lipids are distinguished by their high solubility in non polar solvents and low solubility in H2O Diverse group of compounds including Fats,
LIPID METABOLISM. Sri Widia A Jusman Department of Biochemistry & Molecular Biology FMUI
 LIPID METABOLISM Sri Widia A Jusman Department of Biochemistry & Molecular Biology FMUI Lipid metabolism is concerned mainly with fatty acids cholesterol Source of fatty acids from dietary fat de novo
LIPID METABOLISM Sri Widia A Jusman Department of Biochemistry & Molecular Biology FMUI Lipid metabolism is concerned mainly with fatty acids cholesterol Source of fatty acids from dietary fat de novo
Dr. Nafith Abu Tarboush
 5 Dr. Nafith Abu Tarboush June 25 th 2013 Mohammad Abu Dosh Sheet 5.. Lipids ( Dr. Nafith ) : Classification of fatty acids : - they are classified depending on the existence of double bonds to : 1) Saturated
5 Dr. Nafith Abu Tarboush June 25 th 2013 Mohammad Abu Dosh Sheet 5.. Lipids ( Dr. Nafith ) : Classification of fatty acids : - they are classified depending on the existence of double bonds to : 1) Saturated
Recap: A little chemistry helps to understand a lot of biology
 Recap: A little chemistry helps to understand a lot of biology Covalent Bonds Polar and Non-Polar Electronegativity is key! Non-covalent bonds: Intra and inter molecular interactions Hydrogen Bonds Ionic
Recap: A little chemistry helps to understand a lot of biology Covalent Bonds Polar and Non-Polar Electronegativity is key! Non-covalent bonds: Intra and inter molecular interactions Hydrogen Bonds Ionic
Lipids Definition. Definition: Water insoluble No common structure (though generally large R groups)
 Lipids Definition Definition: Water insoluble No common structure (though generally large R groups) Water Solubility (Hydrophilic) What makes molecules water soluble (hydrophilic)? Like dissolves like
Lipids Definition Definition: Water insoluble No common structure (though generally large R groups) Water Solubility (Hydrophilic) What makes molecules water soluble (hydrophilic)? Like dissolves like
MCQS ON LIPIDS. Dr. RUCHIKA YADU
 MCQS ON LIPIDS Dr. RUCHIKA YADU Q1. THE FATS AND OILS ARE RESPECTIVELY RICH IN a) Unsaturated fatty acids b) Saturated fatty acids c) Saturated and unsaturated fatty acids d) None of these Q2. ESSENTIAL
MCQS ON LIPIDS Dr. RUCHIKA YADU Q1. THE FATS AND OILS ARE RESPECTIVELY RICH IN a) Unsaturated fatty acids b) Saturated fatty acids c) Saturated and unsaturated fatty acids d) None of these Q2. ESSENTIAL
I. Structure and Properties of Lipids
 I. Structure and Properties of Lipids Lipids: A diverse group of compounds characterized by their low solubility in water and a high solubility in organic solvents such as chloroform and methanol. Nonpolar
I. Structure and Properties of Lipids Lipids: A diverse group of compounds characterized by their low solubility in water and a high solubility in organic solvents such as chloroform and methanol. Nonpolar
STANDARD FOR FISH OILS CODEX STAN Adopted in 2017.
 STANDARD FOR FISH OILS CODEX STAN 329-2017 Adopted in 2017. CODEX STAN 329-2017 2 1. SCOPE This Standard applies to the fish oils described in section 2 that are presented in a state for human consumption.
STANDARD FOR FISH OILS CODEX STAN 329-2017 Adopted in 2017. CODEX STAN 329-2017 2 1. SCOPE This Standard applies to the fish oils described in section 2 that are presented in a state for human consumption.
ESSENTIAL FATTY ACIDS - RED CELL
 Healthscope Functional Pathology 1868 Dandenong Road, Clayton, Victoria 3168, Australia www.functionalpathology.com.au ABN 62 006 823 089 T 1300 55 44 80 F 03 8540 5555 infofp@healthscope.com.au LAB No:
Healthscope Functional Pathology 1868 Dandenong Road, Clayton, Victoria 3168, Australia www.functionalpathology.com.au ABN 62 006 823 089 T 1300 55 44 80 F 03 8540 5555 infofp@healthscope.com.au LAB No:
Nafith Abu Tarboush DDS, MSc, PhD
 Nafith Abu Tarboush DDS, MSc, PhD natarboush@ju.edu.jo www.facebook.com/natarboush A heterogeneous class of naturally occurring organic compounds Formed mainly from alcohol & fatty acids combined together
Nafith Abu Tarboush DDS, MSc, PhD natarboush@ju.edu.jo www.facebook.com/natarboush A heterogeneous class of naturally occurring organic compounds Formed mainly from alcohol & fatty acids combined together
Lipids. Lipids: a Diverse group of chemicals. Storage Lipids: derivatives of fatty acids. 11/21/10
 1 Lipids Lehninger 3 rd ed. Chapter 11 (For biosynthesis see Chapter 21) 2 Lipids: a Diverse group of chemicals Insolubility in water. Fats and oils: energy stores. Phospholipids and sterols: structural
1 Lipids Lehninger 3 rd ed. Chapter 11 (For biosynthesis see Chapter 21) 2 Lipids: a Diverse group of chemicals Insolubility in water. Fats and oils: energy stores. Phospholipids and sterols: structural
ANNEX. to the. COMMISSION REGULATION (EU) No /..
 EUROPEAN COMMISSION Brussels, XXX SANCO/10387/2013 Rev. 1 ANNEX (POOL/E3/2013/10387/10387R1-EN ANNEX.doc) D030733/02 [ ](2014) XXX draft ANNEX 1 ANNEX to the COMMISSION REGULATION (EU) No /.. granting
EUROPEAN COMMISSION Brussels, XXX SANCO/10387/2013 Rev. 1 ANNEX (POOL/E3/2013/10387/10387R1-EN ANNEX.doc) D030733/02 [ ](2014) XXX draft ANNEX 1 ANNEX to the COMMISSION REGULATION (EU) No /.. granting
Biosynthesis of Fatty Acids
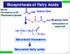 Biosynthesis of Fatty Acids CH 511 ource b-oxidation of FA Glycolysis of glucose n H CoA Acetyl CoA CoA Malonyl CoA Carboxylation of acetyl CoA H 3 C CH 2 CH 2 n-1 CoA aturated thioesters aturated fatty
Biosynthesis of Fatty Acids CH 511 ource b-oxidation of FA Glycolysis of glucose n H CoA Acetyl CoA CoA Malonyl CoA Carboxylation of acetyl CoA H 3 C CH 2 CH 2 n-1 CoA aturated thioesters aturated fatty
Saba Al Fayoumi. Nour Hamdan. Bann Khraisat. Dr. Mamoun Ahram
 9 Saba Al Fayoumi Nour Hamdan Bann Khraisat Dr. Mamoun Ahram Proteoglycans and glycoproteins have been previously discussed and the differences between them have been noted. Protein glycosylation (protein-linked
9 Saba Al Fayoumi Nour Hamdan Bann Khraisat Dr. Mamoun Ahram Proteoglycans and glycoproteins have been previously discussed and the differences between them have been noted. Protein glycosylation (protein-linked
Fatty acids and cardiovascular health: current evidence and next steps
 Fatty acids and cardiovascular health: current evidence and next steps Emanuele Di Angelantonio, MD, PhD Department of Public Health and Primary Care NICE guidelines on fatty acids Eliminate the use of
Fatty acids and cardiovascular health: current evidence and next steps Emanuele Di Angelantonio, MD, PhD Department of Public Health and Primary Care NICE guidelines on fatty acids Eliminate the use of
By: Dr Hadi Mozafari 1
 Biological lipids are a chemically diverse group of compounds, the common and defining feature of which is their insolubility in water. By: Dr Hadi Mozafari 1 Fats and oils are the principal stored forms
Biological lipids are a chemically diverse group of compounds, the common and defining feature of which is their insolubility in water. By: Dr Hadi Mozafari 1 Fats and oils are the principal stored forms
Part 4. Polyunsaturated Fatty Acids
 MASS SPECTRA OF PYRROLIDIE DERIVATIVES Part 4. Polyunsaturated Fatty Acids As was described for dienes, the interpretation of mass spectra of pyrrolidides is similar to that for monoenes in that it is
MASS SPECTRA OF PYRROLIDIE DERIVATIVES Part 4. Polyunsaturated Fatty Acids As was described for dienes, the interpretation of mass spectra of pyrrolidides is similar to that for monoenes in that it is
UNIVERSITY OF CAMBRIDGE. Fatty acids and heart disease. Kay-Tee Khaw
 UNIVERSITY OF CAMBRIDGE Fatty acids and heart disease Kay-Tee Khaw Bristol 3 October 2013 Yerushalmy J Hilleboe HE NY State J Med 1957 Coronary heart disease death rates in relation to blood cholesterol
UNIVERSITY OF CAMBRIDGE Fatty acids and heart disease Kay-Tee Khaw Bristol 3 October 2013 Yerushalmy J Hilleboe HE NY State J Med 1957 Coronary heart disease death rates in relation to blood cholesterol
Summary of fatty acid synthesis
 Lipid Metabolism, part 2 1 Summary of fatty acid synthesis 8 acetyl CoA + 14 NADPH + 14 H+ + 7 ATP palmitic acid (16:0) + 8 CoA + 14 NADP + + 7 ADP + 7 Pi + 7 H20 1. The major suppliers of NADPH for fatty
Lipid Metabolism, part 2 1 Summary of fatty acid synthesis 8 acetyl CoA + 14 NADPH + 14 H+ + 7 ATP palmitic acid (16:0) + 8 CoA + 14 NADP + + 7 ADP + 7 Pi + 7 H20 1. The major suppliers of NADPH for fatty
Fatty acids, cardiovascular disease and diabetes
 Fatty acids, cardiovascular disease and diabetes Rajiv Chowdhury, Nita Forouhi 28 th October 2015 Workshop on nutritional biomarkers A joint PHRI/MRC Epidemiology Unit Initiative Population nutrient goal
Fatty acids, cardiovascular disease and diabetes Rajiv Chowdhury, Nita Forouhi 28 th October 2015 Workshop on nutritional biomarkers A joint PHRI/MRC Epidemiology Unit Initiative Population nutrient goal
Test Bank for Lehninger Principles of Biochemistry 5th Edition by Nelson
 Test Bank for Lehninger Principles of Biochemistry 5th Edition by Nelson Link download full: http://testbankair.com/download/test-bank-forlehninger-principles-of-biochemistry-5th-edition-by-nelson/ Chapter
Test Bank for Lehninger Principles of Biochemistry 5th Edition by Nelson Link download full: http://testbankair.com/download/test-bank-forlehninger-principles-of-biochemistry-5th-edition-by-nelson/ Chapter
Figure 1 Separation of Ginger oil by (a) Cold Maceration (b) Hot Extraction (c) Liquid- Liquid Extraction -
 Effect of Methods of Separation on Ginger Oil Method of extractions affects how many components of a sample is successfully extracted from the sample. Figure one shows the chromatogram obtained for ginger
Effect of Methods of Separation on Ginger Oil Method of extractions affects how many components of a sample is successfully extracted from the sample. Figure one shows the chromatogram obtained for ginger
Nebal Al - Gallab. Shatha Al - Jabri. Mamoon Ahram
 10 Nebal Al - Gallab Shatha Al - Jabri Mamoon Ahram Note: the doctor showed extra examples, they were in the slides, you can refer to them... Naming of Fatty Acids - 1 st Method ( IUPAC system ) We start
10 Nebal Al - Gallab Shatha Al - Jabri Mamoon Ahram Note: the doctor showed extra examples, they were in the slides, you can refer to them... Naming of Fatty Acids - 1 st Method ( IUPAC system ) We start
CERTIFICATE OF ANALYSIS
 CERTIFICATE OF ANALYSIS Schizochytrium DHA-Rich Algal Oil T=0, Lot#BY120306 Date received: 05/09/2012 NDI#: 11072012 Analysis for: Source-Omega LLC Attention: Scott Doughman 11312 US 15-501 North, Suite
CERTIFICATE OF ANALYSIS Schizochytrium DHA-Rich Algal Oil T=0, Lot#BY120306 Date received: 05/09/2012 NDI#: 11072012 Analysis for: Source-Omega LLC Attention: Scott Doughman 11312 US 15-501 North, Suite
: Overview of EFA metabolism
 Figure 1 gives an overview of the metabolic fate of the EFAs when consumed in the diet. The n-6 and n-3 PUFAs when consumed in the form of dietary triglyceride from various food sources undergoes digestion
Figure 1 gives an overview of the metabolic fate of the EFAs when consumed in the diet. The n-6 and n-3 PUFAs when consumed in the form of dietary triglyceride from various food sources undergoes digestion
26.1 Acetyl Coenzyme A
 Chapter 26 Lipids Lipids Lipids are naturally occurring substances grouped together on the basis of a common property they they are more soluble in nonpolar solvents than in water. Some of the most important
Chapter 26 Lipids Lipids Lipids are naturally occurring substances grouped together on the basis of a common property they they are more soluble in nonpolar solvents than in water. Some of the most important
2-more complex molecules (fatty acyl esters) as triacylglycerols.
 ** Fatty acids exist in two forms:- 1-free fatty acids (unesterified) 2-more complex molecules (fatty acyl esters) as triacylglycerols. ** most tissues might use fatty acids as source of energy during
** Fatty acids exist in two forms:- 1-free fatty acids (unesterified) 2-more complex molecules (fatty acyl esters) as triacylglycerols. ** most tissues might use fatty acids as source of energy during
General Chemistry. Ch. 10
 General Chemistry Ch. 10 Essentials of Organic Chemistry Most biological important molecules are composed of organic compounds. These are mostly produced by biological systems. Organic molecules contain
General Chemistry Ch. 10 Essentials of Organic Chemistry Most biological important molecules are composed of organic compounds. These are mostly produced by biological systems. Organic molecules contain
CHAPTER 28 LIPIDS SOLUTIONS TO REVIEW QUESTIONS
 28 09/16/2013 17:44:40 Page 415 APTER 28 LIPIDS SLUTINS T REVIEW QUESTINS 1. The lipids, which are dissimilar substances, are arbitrarily classified as a group on the basis of their solubility in fat solvents
28 09/16/2013 17:44:40 Page 415 APTER 28 LIPIDS SLUTINS T REVIEW QUESTINS 1. The lipids, which are dissimilar substances, are arbitrarily classified as a group on the basis of their solubility in fat solvents
Lecture 25: Polymers and Biopolymers
 Lecture 25: Polymers and Biopolymers In the previous lecture we learned that by using carbon as the primary backbone, we can create a seemingly endless array of molecules built on covelent bonds to carbon.
Lecture 25: Polymers and Biopolymers In the previous lecture we learned that by using carbon as the primary backbone, we can create a seemingly endless array of molecules built on covelent bonds to carbon.
2013 W. H. Freeman and Company. 10 Lipids
 2013 W. H. Freeman and Company 10 Lipids Storage lipids: TG lipid 의기능 : 1 Energy source 3 Electrical insulator 2 Thermal insulator 4 Membrane 의구성성분, 방수, 부력, cofactor, signaling 등 지방대사이상 : obesity, atherosclerosis,
2013 W. H. Freeman and Company 10 Lipids Storage lipids: TG lipid 의기능 : 1 Energy source 3 Electrical insulator 2 Thermal insulator 4 Membrane 의구성성분, 방수, 부력, cofactor, signaling 등 지방대사이상 : obesity, atherosclerosis,
Lipids are used to store and excess energy from extra carbohydrates in animals
 Lipids Lipids are a major source of energy used by cells, however lipids are more difficult for your body to break down. They produce nearly twice the amount of energy than proteins or carbohydrates. Lipids
Lipids Lipids are a major source of energy used by cells, however lipids are more difficult for your body to break down. They produce nearly twice the amount of energy than proteins or carbohydrates. Lipids
BCM 221 LECTURES OJEMEKELE O.
 BCM 221 LECTURES BY OJEMEKELE O. OUTLINE INTRODUCTION TO LIPID CHEMISTRY STORAGE OF ENERGY IN ADIPOCYTES MOBILIZATION OF ENERGY STORES IN ADIPOCYTES KETONE BODIES AND KETOSIS PYRUVATE DEHYDROGENASE COMPLEX
BCM 221 LECTURES BY OJEMEKELE O. OUTLINE INTRODUCTION TO LIPID CHEMISTRY STORAGE OF ENERGY IN ADIPOCYTES MOBILIZATION OF ENERGY STORES IN ADIPOCYTES KETONE BODIES AND KETOSIS PYRUVATE DEHYDROGENASE COMPLEX
Lipid Structure and Function
 Lipid Structure and Function ommon Physical Properties of Lipids Soluble in non-polar organic solvents ontain,, O Sometimes N & P Includes fats and oils mostly triglycerides Fat: solid at room temperature
Lipid Structure and Function ommon Physical Properties of Lipids Soluble in non-polar organic solvents ontain,, O Sometimes N & P Includes fats and oils mostly triglycerides Fat: solid at room temperature
General Biochemistry-1 BCH 202
 General Biochemistry-1 BCH 202 1 I would like to acknowledge Dr. Farid Ataya for his valuable input & help in this course. 2 Outline Lipids Definition, function, fatty acids, classification: simple lipids:
General Biochemistry-1 BCH 202 1 I would like to acknowledge Dr. Farid Ataya for his valuable input & help in this course. 2 Outline Lipids Definition, function, fatty acids, classification: simple lipids:
BY: RASAQ NURUDEEN OLAJIDE
 BY: RASAQ NURUDEEN OLAJIDE LECTURE CONTENT INTRODUCTION CLASSIFICATION OF LIPIDS PROPERTIES OF LIPIDS REACTIONS OF LIPIDS (CHEMICAL PROPERTIES) SOME QUANTITATIVE TESTS FOR LIPIDS CHEMISTRY AND PROPERTIES
BY: RASAQ NURUDEEN OLAJIDE LECTURE CONTENT INTRODUCTION CLASSIFICATION OF LIPIDS PROPERTIES OF LIPIDS REACTIONS OF LIPIDS (CHEMICAL PROPERTIES) SOME QUANTITATIVE TESTS FOR LIPIDS CHEMISTRY AND PROPERTIES
Non-Conjugated Double Bonds
 1 H-NMR Spectroscopy of Fatty Acids and Their Derivatives Non-Conjugated Double Bonds The introduction of one double bond gives rise to several peaks in the NMR spectrum compared to the saturated chains
1 H-NMR Spectroscopy of Fatty Acids and Their Derivatives Non-Conjugated Double Bonds The introduction of one double bond gives rise to several peaks in the NMR spectrum compared to the saturated chains
Dr. Dicken Weatherby's "Foundations of Functional Diagnosis" Training INSIDER S GUIDE. Dicken Weatherby, N.D.
 Dr. Dicken Weatherby's "Foundations of Functional Diagnosis" Training INSIDER S GUIDE Functional Diagnosis of Essential fatty acids By http://www.fmtrainingcenter.com Limits of Liability & Disclaimer of
Dr. Dicken Weatherby's "Foundations of Functional Diagnosis" Training INSIDER S GUIDE Functional Diagnosis of Essential fatty acids By http://www.fmtrainingcenter.com Limits of Liability & Disclaimer of
BIOB111_CHBIO - Tutorial activity for Session 12
 BIOB111_CHBIO - Tutorial activity for Session 12 General topic for week 6 Session 12 Lipids Useful Links: 1. Animations on Cholesterol (its synthesis, lifestyle factors, LDL) http://www.wiley.com/college/boyer/0470003790/animations/cholesterol/cholesterol.htm
BIOB111_CHBIO - Tutorial activity for Session 12 General topic for week 6 Session 12 Lipids Useful Links: 1. Animations on Cholesterol (its synthesis, lifestyle factors, LDL) http://www.wiley.com/college/boyer/0470003790/animations/cholesterol/cholesterol.htm
WHAT IS A LIPID? OBJECTIVE The objective of this worksheet is to understand the structure and function of lipids
 WHAT IS A LIPID? OBJECTIVE The objective of this worksheet is to understand the structure and function of lipids PART A: Understanding Lipids Lipids are more commonly known as fats and include triglycerides,
WHAT IS A LIPID? OBJECTIVE The objective of this worksheet is to understand the structure and function of lipids PART A: Understanding Lipids Lipids are more commonly known as fats and include triglycerides,
4 Types of Organic Polar Reactions
 Objective 10 Apply Reactivity Principles to Elimination Reactions: identify structural features (alpha C, H on beta C, LG) Use curved arrows to predict product. Compare E1 vs. E2 mechanisms. 4 Types of
Objective 10 Apply Reactivity Principles to Elimination Reactions: identify structural features (alpha C, H on beta C, LG) Use curved arrows to predict product. Compare E1 vs. E2 mechanisms. 4 Types of
Table S1. Identified metabolites in rats plasma
 Electronic Supplementary Material (ESI) for Molecular BioSystems. This journal is The Royal Society of Chemistry 2015 Table S1. Identified metabolites in rats plasma NO Retention time Metabolites Level
Electronic Supplementary Material (ESI) for Molecular BioSystems. This journal is The Royal Society of Chemistry 2015 Table S1. Identified metabolites in rats plasma NO Retention time Metabolites Level
Fatty acids synthesis
 Fatty acids synthesis The synthesis start from Acetyl COA the first step requires ATP + reducing power NADPH! even though the oxidation and synthesis are different pathways but from chemical part of view
Fatty acids synthesis The synthesis start from Acetyl COA the first step requires ATP + reducing power NADPH! even though the oxidation and synthesis are different pathways but from chemical part of view
Fatty Acid Mass Spectrometry Protocol Updated 10/11/2007 By Daren Stephens
 Fatty Acid Mass Spectrometry Protocol Updated 10/11/2007 By Daren Stephens Synopsis: This protocol describes the standard method for extracting and quantifying free fatty acids found in cells and media
Fatty Acid Mass Spectrometry Protocol Updated 10/11/2007 By Daren Stephens Synopsis: This protocol describes the standard method for extracting and quantifying free fatty acids found in cells and media
Use natural fish oil with 2g daily dose? Yes Use other omega-3? Replicate test? Date of birth
 Fatty Acid Analysis NORSAN Fatty Acid Analysis Analyse-ID RL44UA52 Date of analysis 31.03.2017 Country GB Sex Male Use natural fish oil with 2g daily dose? Yes Use other omega-3? No Replicate test? Yes
Fatty Acid Analysis NORSAN Fatty Acid Analysis Analyse-ID RL44UA52 Date of analysis 31.03.2017 Country GB Sex Male Use natural fish oil with 2g daily dose? Yes Use other omega-3? No Replicate test? Yes
Comparison of Nutrients, Nutrient Ratios and Other Food Components in NDSR and the ASA24
 Comparison of Nutrients, Nutrient Ratios and Other Food Components in and the Category Primary Energy Sources Alcohol Total Fat Total Protein Energy (kilocalories) Total Carbohydrate Energy (kilojoules)
Comparison of Nutrients, Nutrient Ratios and Other Food Components in and the Category Primary Energy Sources Alcohol Total Fat Total Protein Energy (kilocalories) Total Carbohydrate Energy (kilojoules)
