New trick from an old dog: The toxic lipid and oxidative stress mediator 4-hydroxynonenal activates peroxisome proliferator-activated receptor-β/δ
|
|
|
- Blanche Green
- 6 years ago
- Views:
Transcription
1 R E P R T S New trick from an old dog: The toxic lipid and oxidative stress mediator 4-hydroxynonenal activates peroxisome proliferator-activated receptor-β/δ Jeffrey D. Coleman 1, K. Sandeep Prabhu 1, Jerry T. Thompson 1, P. Sreenivasula Reddy 1, Jeffrey M. Peters 1, Blake R. Peterson 2, Channa Reddy 1, and John P. Vanden Heuvel 1 Chemical-induced hepatotoxicity often results from generation of reactive intermediates and lipid peroxidation, with the cytotoxic aldehyde 4-hydroxynonenal (4-HNE) being a model molecule of oxidative stress. In the absence of the nuclear receptor PPARβ/δ, the mouse liver becomes more susceptible to pro-oxidative toxicants. We hypothesized this resulted from decreased feed-back regulation of metabolism by reactive lipids via PPARβ/δ. In support, oxidized VLDL and constituents including 4-HNE are PPARβ/δ agonists. Hepatocytes with diminished PPARβ/δ activity were more susceptible to 4-HNE-induced gene expression and cytotoxicity. This suggests PPARβ/δ agonists may be utilized to prevent or treat liver disease associated with oxidative damage. Liver insufficiency and damage is a major cause of death and disease worldwide and may result from a variety of stimuli, including exposure to environmental toxicants, specific combinations or dosages of pharmaceuticals, and microbial metabolites. Greater than 2.2 million hospitalized Americans suffer adverse drug reactions each year, with liver toxicity presenting as the most common adverse effect, and approximately 1, individuals die unintentionally from administration of medications (1-3). Many additional cases of liver failure occur due to acute, chronic and degenerative disease processes, including those related to acetaminophen overdose, 1 Department of Veterinary and Biomedical Sciences and Center for Molecular Toxicology and Carcinogenesis, and 2 Department of Chemistry, The Pennsylvania State University, University Park, PA These authors contributed equally to this work To whom correspondence should be addressed: 325 Life Sciences Building, The Pennsylvania State University, University Park, PA 1682 alcohol consumption and solvent exposures. Reactive oxygen intermediates (RI) and other prooxidant agents elicit oxidative decomposition of polyunsaturated fatty acids (i.e, lipid peroxidation), an initiating event of many hepatotoxicants. Lipid peroxidation leads to the formation of a complex mixture of aldehydic end-products, including malonyldialdehyde (MDA), 4-hydroxy-2,3-nonenal (4-HNE), and other alkenals. These aldehydic molecules have been considered the ultimate mediators of toxic effects elicited by oxidative stress but may also affect cellular function at nontoxic levels via signal transduction, gene expression and cell proliferation. Although the overt toxicity caused by aldehydic endproducts is due primarily to covalent binding to cellular macromolecules, the effects on signal transduction are less well known. The peroxisome proliferator-activated receptors (PPARs) are nuclear receptors that exist as three subtypes (α, β and γ), which exhibit tissue-specific expression, preferential ligand recognition, and distinct biological functions (4-9). Although important as targets of pharmaceutical intervention, there is increasing evidence that the biological niche occupied by the PPARs is that of receptor for fatty acid and their metabolites. f the different PPARs, the PPARb/d isoform is the least well studied in terms of its biological role and endogenous ligands. PPARβ/δ plays an important role in differentiation of epithelial tissues, fatty acid catabolism in skeletal muscle, improvement of insulin sensitivity, attenuated weight gain and elevated HDL levels (1). Emerging evidence suggests the presence of this receptor is important in ameliorating the effects on hepatotoxicants. For example, histological examination of liver and analysis of markers of overt damage to this organ (serum GPT) with xenobiotics azoxymethane (AM), carbon tetrachloride, arsenic and acetaminophen (APAP) demonstrated that the extent of liver toxicity in PPARβ/β-null mice was more severe as compared to wildtype mice. While necrotic cells were found in AM-treated wild-type mice, bile duct hyperplasia, areas of regenerative hyperplasia and early adenomas were found exclusively in PPARβ/δ-null mice and markers of liver damage were seen earlier than in wildtype animals. Subchronic administration of CCl4 caused central lobular granular degeneration with necrosis in both genotypes, however lipid deposition surrounding lobules was only observed in PPARβ/δ-null mouse liver and overt damage to the liver was preferentially found in the mice lacking PPARβ/δ. Exacerbated overt toxicity with bile duct hyperplasia is found in PPARβ/δ-null mouse liver in response to exposure to both APAP and arsenic. While it is remotely possible that the metabolic fate of some of these hepatotoxicants could be influenced by PPARβ/δ, it is more likely that regulation of oxidative stress/inflammation could underlie the protective role of this receptor in liver. These chemicals share a common mechanism of overt toxicity via production of RI and oxidized lipid intermediates. For example, CCl4 affects eicosanoid pathways (11, 12) and increase circulating prostaglandin E2 (PGE2) levels (13) 1 SCIENCE
2 R E P R T S 2 1 mpparα mpparβ/δ mpparγ R.L.U (S)HDE released (ng) xvldl (µg) CuS 4 DMS + control LPL VLDL oxvldl VLDL+LPL oxvldl+lpl CuS LpL-treated xvldl ( µg/ml) EtH TTA 13HDE 9HDE 9HETE 12HETE 15HETE 5HETE 12HpDE 5HpETE 15HpETE 5,15diHpETE 5,6diHETE Figure 1. xidation of VLDL leads to increased activation of PPARβ. VLDL (1µg/ml) was oxidized with CuS4 (1µM) for 72 h, or pretreated with LpL for 1 h at 35 oc, and the mixture was added to 3T3-L1 cells transfected with GAL4-LBD of each of the PPAR isoforms. Following treatment the cells were lysed and relative luciferase activity was determined. Results were corrected for DMS. Figure 1 Dose-dependent activation of PPARβ by LPL-treated oxvldl. 3T3-L1 preadipocytes were transfected with Gal4-LBD-mPPARβ and treated overnight with oxidized VLDL incubated for 1 h with LpL. The cells were lysed and relative luciferase activity was determined. N = 8. Figure 1 xidized 13(S)-HDE is a strong PPARβ agonist. Chemicals were obtained as described in Methods. UV-Visible spectroscopy was used to determine their respective concentrations, and the original solvent was evaporated under a stream of N2 and the chemical reconstituted in ethanol to 5 µm. xidation products were obtained via incubation with CuS4 (1 µm) for 72 h. 3T3-L1 preadipocytes were transfected with Gal4-LBD-mPPARβ and treated with compound overnight. The cells were lysed and relative luciferase activity was determined. Results were normalized to EtH/H2. N = 3. Significantly different than the EtH control; +Significantly different than the non-cus4-treated counterpart. Inset: xidation of VLDL releases 13(S)-HDE. VLDL was oxidized using 1 µm CuS4 for 72 h. xidized VLDL was then incubated with LpL for 1 h. An ELISA specific for 13(S)-HDE (Assay Designs) was performed to quantify the amount hydrolyzed from oxvldl. Samples were prepared by diluting 1, and 1 µl of oxvldl with standard diluent to a final volume of 1 µl (N = 2). resulting in an enhanced production of 4-HNE and 4-HNE-protein adducts (14-16). Similarly, APAP-treated rats also produced greater quantities of PGE2, thromboxane A2 and leukotriene C4 from normal liver (17) accompanied by an increased 4-HNE and MDA formation in rat liver (18). ne possible explanation for the increased susceptibility of PPARβ/δ null mice to hepatotoxicity is that agents such as CCl4 and APAP increase the production of an endogenous ligand for PPARβ/δ, which would in turn stimulate lipid metabolism and degradation of lipid peroxidation intermediates. In the absence of this receptor, the signaling cascade would be disrupted and accumulation of toxic lipids such as 4-HNE would result. If our hypothesis were correct, endogenous ligands of PPARβ/δ should include oxidized lipids, in particular those derived from fatty acids. The VLDL particle, which is naturally generated in the liver, is a mixture of free and esterified cholesterols, triglycerides (formed from various fatty acids) and apolipoproteins. As seen in Fig. 1A, the oxidation of VLDL with CuS4 (oxvldl) failed to activate any of the PPAR isoforms, including PPARβ/δ. However, incubation of oxvldl with Lipoprotein Lipase (LpL) greatly increased PPARβ /δ activity specifically, with minimal increases in activity of the other two PPAR subtypes. Furthermore, in an effort to establish the concentration at which PPARβ activity is maximally stimulated by oxvldl, a doseresponse relationship was established (Fig. 1B) with a half maximal concentration (EC5) of approximately 2 µg/ml. To determine which, if any, fatty acids released by oxvldl are playing a role in the enhancement of PPARβ/δ activity, we conducted a screen study involving 11 fatty acids and their CuS4 oxidation products. In Fig. 1C we show that, while 13(S)-HDE alone activates PPARβ/δ, its oxidation product increases activity roughly two-fold higher. No significant enhancement of PPARβ/δ was observed with any of the other fatty acids tested, except 15-HETE, which also activated the PPARβ/δ isoforms. Furthermore, we hypothesized that the polyunsaturated fatty acid 13(S)- HDE, an oxidation product of the 15-lipoxygenase metabolic pathway, is hydrolyzed upon incubation with LpL to the free fatty acid which, ultimately, induces PPARβ/δ activity. Both 13(S)-HDE and its hydroperoxy derivative, 13(S)- HpDE, were equally potent in their ability to activate PPARβ/δ (data not shown). To examine if 13(S)-HDE was released from oxidized VLDL treated with LpL, we used an enzyme-linked immunosorbent assay specific for the S-enantiomer of 13- HDE. ur results show that the release of 13(S)-HDE increased correspondingly with the amount of oxvldl used (Fig. 1C, Inset). Based on the EIA, we estimated that 1 µg of LpL-treated oxidized VLDL contains approximately 37.5 ng of 13(S)- HDE. Interestingly, while CuS4 oxidation of 13(S)-HDE also generated a PPARβ/δ agonist with greater affinity for the subtype than the fatty acid itself, oxidation of 15- HETE in the same manner did not have any effect on its ability to increase receptor activity in our reporter assay. Together, these results indicated that receptor activation by products of lipid peroxidation were likely to be endogenous ligands for the PPARβ/δ protein. 2 SCIENCE
3 R E P R T S H H H 4-hydroxy-Hexenal 4-hydroxy-2-Nonenal 4-hydroperoxy-2-Nonenal 4-oxo-2-Nonenal Ethanol TTA 4-HpNE 4-HNE Figure 2. Structures of the test molecules. Figure 2 PPARβ/δ is activated in a dose-by 4-HNE in 3T3-L1 preadipocytes. The cells were transfected with Gal4-LBD-mPPARβ/δ followed by treatment with each of the lipid peroxidation products or vehicle control for 12 h. Following the treatment, cells were lysed and luciferase activity was determined and normalized to EtH. Treatment was with with 25 µm 4-HHE, 4-HNE, 4-NE and trans-4,5-epoxy-2e-decenal (Decenal; Cayman) overnight. Cells were treated with 25 µm 4-HpNE or 4-HNE for 12 h. D. Dose-response of PPARβ/δ activation by 4-HNE. PPARb activity is increased with addition of 25 µm 4-HNE, while cellular toxicity is observed at higher concentrations. N = 8. In order to determine which specific type of aldehydic product induces PPARβ/δ activity, a structure-activity relationship was established using a representative compound from each family of lipid peroxidation product, 2- alkenals, 4-hydroxy-2-alkenals, and ketoaldehydes (Fig. 2). 2-Alkenals were represented by trans-4,5-epoxy- 2(E)-decenal, ketoaldehydes were trans-4,5-epoxy-2(e)-decenal 2 mpparα mpparβ/δ mpparγ 1 EtH D. + control 4-HHE represented by 4-NE, and 4- hydroxy-2-alkenals were represented by both 4-HHE and 4-HNE, aldehydes which differ only in their carbon chain length (Fig. 2A). Reporter assays clearly showed that 4-HNE alone enhanced the activity of all three PPAR subtypes, with the greatest augmentation afforded to the PPARβ/δ subtype (Figure 2B). n 4-HNE 4-NE Decenal log [4-HNE] (µm) the other hand, 4-HHE, 4-NE, and trans-4,5-epoxy-2(e)-decenal did not activate any of the PPARs. Periods of oxidative stress initiate free-radical mediated oxidative damage of lipids, such as 13(S)-HDE. The breakdown pathway from lipid to 4- HNE includes a hydroperoxyderivative, 4-hydroperoxynonenal (4- HpNE), which is rapidly reduced to 4- HNE by cellular peroxidases. In fact, 4-HNE, the resultant hydroxyderivative formed from 4-HpNE, activated PPARβ/δ to a greater extent than its precursor (Fig. 2C). Furthermore, a dose-dependent activation of PPARβ/δ was also seen with 4-HNE (Fig. 3D), where maximal receptor activation was observed around 25 µm. Cellular toxicity was observed at concentrations higher than 3 µm and biological concentration of 4-HNE are known to be within this range. In order to analyze if 4-HNE affected gene expression or toxicity in a PPARβ/δ-dependent manner, studies were performed on murine SV-4 immortalized hepatocytes prepared from wild-type (MuSH WT) and PPARβ/δ-/- (MuSHβ/δ-/-) mice. Such hepatocytes were treated with vehicle control (DMS), a known PPARβ/δ agonist tetradecylthioacetic acid (TTA), 13(S)-HDE and 4-HNE. The expression of a PPARβ/δ-regulated gene, adipose differentiation related protein (ADRP), was up-regulated in a PPARβ/δ-dependent manner in cells treated with 25µM each of 13 (S)-HDE and 4-HNE (Fig. 3A). mrna ADRP/mRNA β-actin DMS TTA MuSH WT MuSH β/δ -/- 13-HDE 4-HNE Viability MuSH WT MuSHβ/δ -/ log[4-hne] Viability DMS Inhibitor HNE (um) Figure 3. 4-HNE regulates gene expression in a PPARβ/δ -dependent manner. MuSH WT and MuSHβ/δ -/- cells were treated with 13(S)-HDE and 4-HNE overnight and the cells were lysed. Total RNA was extracted using Tri-Reagent and RT-PCR performed for ADRP and corrected for b- actin expression. N = 3. Figure 3 PPARβ is crucial in ameliorating the toxic effects of 4-HNE. MuSHWT and MuSHβ-/- hepatocytes were treated with,.1,.3, 1, 3, 1 or 25 µm 4-HNE. The data is normalized for % viable cells, shown on the y-axis. N = 8. Representative of six independent experiments. Figure 3 Inhibition of PPARβ/δ leads to increased susceptibility to oxidative damage. MuSHWT cells were pre-treated with vehicle or GW9662 for 6 h prior to treatment with 4-HNE. and Significantly different than the DMS counterpart. N = 5. 3 SCIENCE
4 R E P R T S Figure 4.A: X-ray structure of PPARβ/δ bound to eicosapentaenoic acid. Figure 4B: A molecular model of PPARβ/δ bound to (S)-4- hydroxynonenal (CPK model) in a conformation similar to the natural ligand. Figure 4C: Visualization of the putative hydrogen-bonding interaction between His413 (stick model) and the 4-hydroxyl group of the ligand (CPK model). Figure 4D. 4- HNE activates PPARβ/δ via a direct and non-covalent interaction. Mouse PPARβ/δ LBD was expressed using an in vitro transcriptiontranslation system (Roche) and incubated with 4-HNE. Immunoblot analysis was used to determine 4-HNE-protein adducts. Lane 1 PPARβ/δ-LBD alone; Lane 2, PPARβ/δ-LBD plus EtH; Lane 3, PPARβ/δ-LBD plus 4-HNE (1 µm); Lane 4, BAS plus 4-HNE (1 µm). Figure 4E. Identical incubation as performed above with immunoblot analysis of PPARβ/δ. Importantly, the cells lacking PPARβ/ δ were more susceptible to 4-HNE cytotoxicity than the wildtype cells (Fig. 3B) and treating MuSH WT cells with a chemical antagonist of PPARβ/ δ increased sensitivity to this toxic lipid (Fig. 3C). Molecular modeling was employed using the coordinates of PPARβ/δ bound to eicosapentaenoic acid (PDB ID: 3GWX, Figure 4A) and was examined for the putative association with 4-HNE. PPARβ/δ bound to (S)-4-hydroxynonenal (CPK model) in a conformation similar to the natural ligand (Figure 4B). However, a putative hydrogenbonding interaction between His413 (stick model) and the 4-hydroxyl group of the 4-HNE ligand (CPK model) occurs (Figure 4C), suggesting the association is in some ways unique. Knowing that 4-HNE interacts with a wide variety of proteins via the formation of covalent adducts with cysteinyl, lysyl, and histidyl residues, we used a mixed polyclonal antibody towards 4-HNE protein adducts to detect the presence of such covalent products with the mouse PPARβ/δ isoform (Fig. 4D). However, we were unable to show covalent interactions between 4-HNE and PPARβ/δ strongly suggesting that the association with PPARβ/δ is reversible. The last nine carbons of arachidonic and linoleic acid are represented in 4-HNE and the precursor to 13(S)-HDE, 13(S)- HPDE may be further oxidized by a Hock Cleavage mechanism to yield 4-HNE. The arachidonic and linoleic acid oxidation leads to the formation of a highly unstable hydroperoxide and racemic derivative, 4-HpNE, which can then be metabolized by cellular peroxidases to 4-HNE and its isomer, 4-NE. Consistent with this idea, treatment of 4-HpNE also activated PPARβ/δ in the reporter while treatment of cells with its reduced product, 4-HNE, led to a greater increase in the PPARβ/δ reporter activity. In order to rule out the possibility that 4-HNE-treatment resulted in an oxidative-stress mediated increase in other endogenous ligands, we used 4-NE as a control to show that a related lipid peroxidation product, which differs from 4-HNE in having a carbonyl rather than a hydroxyl group at 4-C, was ineffective in activating the receptor. The results unequivocally confirm that PPARβ/δ activation by this class of molecule is specifically effected by 4-HNE treatment. Furthermore, even other lipid peroxidation products such as 4- HHE and decenal were not capable of activating PPARβ/δ isoform. In addition to being controls, these assays also shed some light on the possible structural and spatial requirements of an endogenous ligand for PPARβ/δ in that a nine carbon chain length with a hydroxyl group at 4-C seems essential for ligand-dependent activation. Based on the molecular modeling information, it is possible that an association between the hydroxyl group and Histidine-413 is necessary for optimal receptor activation. PPARβ/δ is the least understood of the three PPAR subtypes in many respects, including the identification of endogenous ligands. This receptor is ubiquitously expressed and is often found in higher abundance than PPARα or γ in brain, adipose tissue and skin. Examination of the PPARβ/δ null mice has shown a role for PPARβ/δ in development, myelination of the corpus callosum, lipid metabolism, and epidermal cell proliferation. Few high affinity ligands for PPARβ/δ are known, either xenobiotic or endogenous. However, fatty acids are weak activators of this receptor with roughly the same preference as PPARα. Interestingly, similar to PPARα and γ, incubation of triglyceride rich lipoproteins with LPL results in the production of PPARβ ligands. ne of the potential products from the oxvldl particular that affects PPARβ / δ is 13-Shydroperoxyoctadecadienoic acid (13 (S)-HDE). In addition to being the primary product of 15-lipoxygenase-1 with linoleic acid as the substrate, this molecule may be an indicator of potential lipid peroxidation produced from VLDL. There is a wealth of information on 4-HNE as being a marker of lipid peroxidation as well as a mediator of hepatotoxicity and a variety of other pathophysiological 4 SCIENCE
5 R E P R T S conditions including cancer, Alzheimer s disease and atherosclerosis (19). Being a lipophilic molecule, 4-HNE can cross subcellular compartments and ultimately react with the variety of different cell proteins. Target molecules that are inhibited by 4- HNE include the glucose transporter GLUT3, tau, the proteasome and IκB kinase (19). While 4-HNE formed covalent adducts with all the three above mentioned proteins, results from our studies show that the interaction of 4-HNE with PPARβ/δ did not proceed through such a Michael addition mechanism. In summary, we have shown that the lipid peroxidation product 4- HNE, which was regarded to be a mutagen for all these years, has now been demonstrated to have a nuclear receptor binding activity. This receptor, PPARβ/δ is thereby activated and provides a feed-back regulation of gene expression that ameliorates toxicity to this tissue. The discovery of 4-HNE as an endogenous nuclear receptor ligand also rescues PPARβ/δ from the orphanage as well as reveals a crosstalk of PPARβ/δ in cellular events involved in inflammation and energy homeostasis. Given the large number of individuals hospitalized due to chemically-induced liver damage, it is attractive to speculate that this knowledge may be exploited for more effective means of protecting or treating hepatotoxicity as well as other diseases in which 4- HNE plays a causative role. 7. J. P. Vanden Heuvel, J. M. Peters, Comments Toxicol. 7, 233 (21). 8. J. P. Vanden Heuvel, Toxicol Sci 47, 1 (1999). 9. J. P. Vanden Heuvel, J Nutr 129, 575S (1999). 1. D. Burdick, D. J. Kim, M. Peraza, F. J. Gonzalez, J. M. Peters, Cell Signal (Aug 15, 25). 11. S. Basu, Toxicology 189, 113 (Jul 15, 23). 12. N. Kawada, Y. Mizoguchi, K. Kobayashi, S. Yamamoto, S. Morisawa, Gastroenterol Jpn 25, 363 (Jun, 199). 13. M. Lopez-Parra et al., Br J Pharmacol 135, 891 (Feb, 22). 14. D. P. Hartley, K. L. Kolaja, J. Reichard, D. R. Petersen, Toxicol Appl Pharmacol 161, 23 (Nov 15, 1999). 15. M. Comporti, Free Radic Res 28, 623 (Jun, 1998). 16. D. P. Hartley, D. J. Kroll, D. R. Petersen, Chem Res Toxicol 1, 895 (Aug, 1997). 17. F. Culo et al., Eur J Gastroenterol Hepatol 7, 757 (Aug, 1995). 18. J. Liu et al., Hepatology 37, 324 (Feb, 23). 19. K. Uchida, Prog Lipid Res 42, 318 (Jul, 23). Atheroscler Rep 6, (Nov, 1. W. M. Lee, J. R. Senior, Toxicol Pathol 33, 155 (25). 2. W. M. Lee, N Engl J Med 349, 474 (Jul 31, 23). 3. W. M. Lee, Semin Liver Dis 23, 217 (Aug, 23). 4. J. P. Vanden Heuvel, Curr Atheroscler Rep 6, 432 (Nov, 24). 5. S. Khan, J. P. Vanden Heuvel, J Nutr Biochem 14, 554 (ct, 23). 6. J. M. Peters, J. P. Vanden Heuvel, in Cellular and Molecular Toxicology J. P. Vanden Heuvel, G. H. Perdew, W. Mattes, W. F. Greenlee, Eds. (Elsevier Science, Amsterdam, The Netherlands, 22), vol. 14, pp SCIENCE
Detection of Lipid Peroxidation Products From Free Radical and Enzymatic Processes. Jason D. Morrow M.D. Vanderbilt University School of Medicine
 Detection of Lipid Peroxidation Products From Free Radical and Enzymatic Processes Jason D. Morrow M.D. Vanderbilt University School of Medicine Question? Which one of the following is the most accurate
Detection of Lipid Peroxidation Products From Free Radical and Enzymatic Processes Jason D. Morrow M.D. Vanderbilt University School of Medicine Question? Which one of the following is the most accurate
3-Thia Fatty Acids A New Generation of Functional Lipids?
 Conference on Food Structure and Food Quality 3-Thia Fatty Acids A New Generation of Functional Lipids? Rolf K. Berge rolf.berge@med.uib.no Fatty acids- Essential cellular metabolites Concentrations must
Conference on Food Structure and Food Quality 3-Thia Fatty Acids A New Generation of Functional Lipids? Rolf K. Berge rolf.berge@med.uib.no Fatty acids- Essential cellular metabolites Concentrations must
OXIDIZED LIPIDS FORMED NON-ENZYMATICALLY BY REACTIVE OXYGEN SPECIES
 JBC Papers in Press. Published on February 19, 2008 as Manuscript R800006200 The latest version is at http://www.jbc.org/cgi/doi/10.1074/jbc.r800006200 OXIDIZED LIPIDS FORMED NON-ENZYMATICALLY BY REACTIVE
JBC Papers in Press. Published on February 19, 2008 as Manuscript R800006200 The latest version is at http://www.jbc.org/cgi/doi/10.1074/jbc.r800006200 OXIDIZED LIPIDS FORMED NON-ENZYMATICALLY BY REACTIVE
LOOKING FOR LIPID PEROXIDATION IN VITRO AND IN VIVO: IS SEEING BELIEVING? Vanderbilt University School of Medicine Jason D.
 LOOKING FOR LIPID PEROXIDATION IN VITRO AND IN VIVO: IS SEEING BELIEVING? Vanderbilt University School of Medicine Jason D. Morrow MD Which of the following assays of lipid peroxidation may be useful and
LOOKING FOR LIPID PEROXIDATION IN VITRO AND IN VIVO: IS SEEING BELIEVING? Vanderbilt University School of Medicine Jason D. Morrow MD Which of the following assays of lipid peroxidation may be useful and
Reactivity and Biological Functions of Oxidized Lipids
 Reactivity and Biological Functions of Oxidized Lipids Matthew J. Picklo, Sr. Outline 1. Lipid Oxidation Mechanisms and Products 2. Biological Mechanisms 3. n-3 vs n-6 PUFA Oxidation 1. Fatty acids 2.
Reactivity and Biological Functions of Oxidized Lipids Matthew J. Picklo, Sr. Outline 1. Lipid Oxidation Mechanisms and Products 2. Biological Mechanisms 3. n-3 vs n-6 PUFA Oxidation 1. Fatty acids 2.
2.5. AMPK activity
 Supplement Fig. A 3 B phos-ampk 2.5 * Control AICAR AMPK AMPK activity (Absorbance at 45 nm) 2.5.5 Control AICAR Supplement Fig. Effects of AICAR on AMPK activation in macrophages. J774. macrophages were
Supplement Fig. A 3 B phos-ampk 2.5 * Control AICAR AMPK AMPK activity (Absorbance at 45 nm) 2.5.5 Control AICAR Supplement Fig. Effects of AICAR on AMPK activation in macrophages. J774. macrophages were
Enzymatic oxidation of lipids: mechanisms and functions.
 Enzymatic oxidation of lipids: mechanisms and functions. Valerie B. O Donnell, PhD. Cardiff University. Enzymatic lipid oxidation: involves an enzyme catalyst, and gives very specific stereo- and regiospecific
Enzymatic oxidation of lipids: mechanisms and functions. Valerie B. O Donnell, PhD. Cardiff University. Enzymatic lipid oxidation: involves an enzyme catalyst, and gives very specific stereo- and regiospecific
Cellular functions of protein degradation
 Protein Degradation Cellular functions of protein degradation 1. Elimination of misfolded and damaged proteins: Environmental toxins, translation errors and genetic mutations can damage proteins. Misfolded
Protein Degradation Cellular functions of protein degradation 1. Elimination of misfolded and damaged proteins: Environmental toxins, translation errors and genetic mutations can damage proteins. Misfolded
Supporting Information Table of content
 Supporting Information Table of content Supporting Information Fig. S1 Supporting Information Fig. S2 Supporting Information Fig. S3 Supporting Information Fig. S4 Supporting Information Fig. S5 Supporting
Supporting Information Table of content Supporting Information Fig. S1 Supporting Information Fig. S2 Supporting Information Fig. S3 Supporting Information Fig. S4 Supporting Information Fig. S5 Supporting
GPR120 *** * * Liver BAT iwat ewat mwat Ileum Colon. UCP1 mrna ***
 a GPR120 GPR120 mrna/ppia mrna Arbitrary Units 150 100 50 Liver BAT iwat ewat mwat Ileum Colon b UCP1 mrna Fold induction 20 15 10 5 - camp camp SB202190 - - - H89 - - - - - GW7647 Supplementary Figure
a GPR120 GPR120 mrna/ppia mrna Arbitrary Units 150 100 50 Liver BAT iwat ewat mwat Ileum Colon b UCP1 mrna Fold induction 20 15 10 5 - camp camp SB202190 - - - H89 - - - - - GW7647 Supplementary Figure
Removal of Unreacted Dinitrophenyl Hydrazine from Carbonyl Derivatives. Amira Barkal Mentored by Dr. Gary Merrill Biochemistry and Biophysics
 Removal of Unreacted Dinitrophenyl Hydrazine from Carbonyl Derivatives Amira Barkal Mentored by Dr. Gary Merrill Biochemistry and Biophysics Carbonyls and Disease Alzheimer s Disease Degenerative brain
Removal of Unreacted Dinitrophenyl Hydrazine from Carbonyl Derivatives Amira Barkal Mentored by Dr. Gary Merrill Biochemistry and Biophysics Carbonyls and Disease Alzheimer s Disease Degenerative brain
Dean J. Tuma, Ph.D., and Carol A. Casey, Ph.D.
 Dangerous Byproducts of Alcohol Breakdown Focus on Adducts Dean J. Tuma, Ph.D., and Carol A. Casey, Ph.D. Alcohol breakdown in the liver results in the generation of the reactive molecule acetaldehyde
Dangerous Byproducts of Alcohol Breakdown Focus on Adducts Dean J. Tuma, Ph.D., and Carol A. Casey, Ph.D. Alcohol breakdown in the liver results in the generation of the reactive molecule acetaldehyde
David Clark, MD Chief Medical Officer Aldeyra Therapeutics, Lexington, MA. ARVO: April 30th, 2018
 A Randomized, Double-Masked, Parallel-Group, Phase 2a Dry Eye Disease Clinical Trial to Evaluate the Safety and Efficacy of Topical Ocular ADX-102 (Reproxalap), a Novel Aldehyde Sequestering Agent David
A Randomized, Double-Masked, Parallel-Group, Phase 2a Dry Eye Disease Clinical Trial to Evaluate the Safety and Efficacy of Topical Ocular ADX-102 (Reproxalap), a Novel Aldehyde Sequestering Agent David
Human Oxidized LDL ELISA Kit (MDA-LDL Quantitation), General
 Human Oxidized LDL ELISA Kit (MDA-LDL Quantitation), General For the detection and quantitation of human OxLDL in plasma, serum or other biological fluid samples Cat. No. KT-959 For Research Use Only.
Human Oxidized LDL ELISA Kit (MDA-LDL Quantitation), General For the detection and quantitation of human OxLDL in plasma, serum or other biological fluid samples Cat. No. KT-959 For Research Use Only.
OBJECTIVE. Lipids are largely hydrocarbon derivatives and thus represent
 Paper 4. Biomolecules and their interactions Module 20: Saturated and unsaturated fatty acids, Nomenclature of fatty acids and Essential and non-essential fatty acids OBJECTIVE The main aim of this module
Paper 4. Biomolecules and their interactions Module 20: Saturated and unsaturated fatty acids, Nomenclature of fatty acids and Essential and non-essential fatty acids OBJECTIVE The main aim of this module
OxiSelect HNE Adduct Competitive ELISA Kit
 Product Manual OxiSelect HNE Adduct Competitive ELISA Kit Catalog Number STA-838 STA-838-5 96 assays 5 x 96 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Lipid peroxidation
Product Manual OxiSelect HNE Adduct Competitive ELISA Kit Catalog Number STA-838 STA-838-5 96 assays 5 x 96 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Lipid peroxidation
General Laboratory methods Plasma analysis: Gene Expression Analysis: Immunoblot analysis: Immunohistochemistry:
 General Laboratory methods Plasma analysis: Plasma insulin (Mercodia, Sweden), leptin (duoset, R&D Systems Europe, Abingdon, United Kingdom), IL-6, TNFα and adiponectin levels (Quantikine kits, R&D Systems
General Laboratory methods Plasma analysis: Plasma insulin (Mercodia, Sweden), leptin (duoset, R&D Systems Europe, Abingdon, United Kingdom), IL-6, TNFα and adiponectin levels (Quantikine kits, R&D Systems
Steroid Hormones Synthesis
 *I ll try my best to incorporate the Slides in this Sheet; you don t need to study the slides if you study this sheet. Steroid Hormones Synthesis - The figure to the right is the Steroid nucleus, it has
*I ll try my best to incorporate the Slides in this Sheet; you don t need to study the slides if you study this sheet. Steroid Hormones Synthesis - The figure to the right is the Steroid nucleus, it has
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION FOR Liver X Receptor α mediates hepatic triglyceride accumulation through upregulation of G0/G1 Switch Gene 2 (G0S2) expression I: SUPPLEMENTARY METHODS II: SUPPLEMENTARY FIGURES
SUPPLEMENTARY INFORMATION FOR Liver X Receptor α mediates hepatic triglyceride accumulation through upregulation of G0/G1 Switch Gene 2 (G0S2) expression I: SUPPLEMENTARY METHODS II: SUPPLEMENTARY FIGURES
: Overview of EFA metabolism
 Figure 1 gives an overview of the metabolic fate of the EFAs when consumed in the diet. The n-6 and n-3 PUFAs when consumed in the form of dietary triglyceride from various food sources undergoes digestion
Figure 1 gives an overview of the metabolic fate of the EFAs when consumed in the diet. The n-6 and n-3 PUFAs when consumed in the form of dietary triglyceride from various food sources undergoes digestion
Radicals. Structure and Stability of Radicals. Radicals are formed from covalent bonds by adding energy in the form of heat (Δ) or light (hν).
 Radicals Chapter 15 A small but significant group of reactions involve radical intermediates. A radical is a reactive intermediate with a single unpaired electron, formed by homolysis of a covalent bond.
Radicals Chapter 15 A small but significant group of reactions involve radical intermediates. A radical is a reactive intermediate with a single unpaired electron, formed by homolysis of a covalent bond.
Oxidation of Long Chain Fatty Acids
 Oxidation of Long Chain Fatty Acids Dr NC Bird Oxidation of long chain fatty acids is the primary source of energy supply in man and animals. Hibernating animals utilise fat stores to maintain body heat,
Oxidation of Long Chain Fatty Acids Dr NC Bird Oxidation of long chain fatty acids is the primary source of energy supply in man and animals. Hibernating animals utilise fat stores to maintain body heat,
OxiSelect Human Oxidized LDL ELISA Kit (MDA- LDL Quantitation)
 Product Manual OxiSelect Human Oxidized LDL ELISA Kit (MDA- LDL Quantitation) Catalog Number STA- 369 96 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Lipoproteins are
Product Manual OxiSelect Human Oxidized LDL ELISA Kit (MDA- LDL Quantitation) Catalog Number STA- 369 96 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Lipoproteins are
Role of metabolism in Drug-Induced Liver Injury (DILI) Drug Metab Rev. 2007;39(1):
 Role of metabolism in Drug-Induced Liver Injury (DILI) Drug Metab Rev. 2007;39(1):159-234 Drug Metab Rev. 2007;39(1):159-234 Drug Metab Rev. 2007;39(1):159-234 A schematic representation of the most relevant
Role of metabolism in Drug-Induced Liver Injury (DILI) Drug Metab Rev. 2007;39(1):159-234 Drug Metab Rev. 2007;39(1):159-234 Drug Metab Rev. 2007;39(1):159-234 A schematic representation of the most relevant
Fatty Acid Degradation. Catabolism Overview. TAG and FA 11/11/2015. Chapter 27, Stryer Short Course. Lipids as a fuel source diet Beta oxidation
 Fatty Acid Degradation Chapter 27, Stryer Short Course Catabolism verview Lipids as a fuel source diet Beta oxidation saturated Unsaturated dd chain Ketone bodies as fuel Physiology High energy More reduced
Fatty Acid Degradation Chapter 27, Stryer Short Course Catabolism verview Lipids as a fuel source diet Beta oxidation saturated Unsaturated dd chain Ketone bodies as fuel Physiology High energy More reduced
OxiSelect HNE-His Adduct ELISA Kit
 Product Manual OxiSelect HNE-His Adduct ELISA Kit Catalog Number STA-334 96 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Lipid peroxidation is a well-defined mechanism
Product Manual OxiSelect HNE-His Adduct ELISA Kit Catalog Number STA-334 96 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Lipid peroxidation is a well-defined mechanism
CHAPTER 28 LIPIDS SOLUTIONS TO REVIEW QUESTIONS
 28 09/16/2013 17:44:40 Page 415 APTER 28 LIPIDS SLUTINS T REVIEW QUESTINS 1. The lipids, which are dissimilar substances, are arbitrarily classified as a group on the basis of their solubility in fat solvents
28 09/16/2013 17:44:40 Page 415 APTER 28 LIPIDS SLUTINS T REVIEW QUESTINS 1. The lipids, which are dissimilar substances, are arbitrarily classified as a group on the basis of their solubility in fat solvents
Org/Biochem Final Lec Form, Spring 2012 Page 1 of 6
 Page 1 of 6 Missing Complete Protein and Question #45 Key Terms: Fill in the blank in the following 25 statements with one of the key terms in the table. Each key term may only be used once. Print legibly.
Page 1 of 6 Missing Complete Protein and Question #45 Key Terms: Fill in the blank in the following 25 statements with one of the key terms in the table. Each key term may only be used once. Print legibly.
OxiSelect Human Oxidized LDL ELISA Kit (OxPL-LDL Quantitation)
 Product Manual OxiSelect Human Oxidized LDL ELISA Kit (OxPL-LDL Quantitation) Catalog Number STA-358 96 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Lipoproteins are submicroscopic
Product Manual OxiSelect Human Oxidized LDL ELISA Kit (OxPL-LDL Quantitation) Catalog Number STA-358 96 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Lipoproteins are submicroscopic
Lipids Definition. Definition: Water insoluble No common structure (though generally large R groups)
 Lipids Definition Definition: Water insoluble No common structure (though generally large R groups) Water Solubility (Hydrophilic) What makes molecules water soluble (hydrophilic)? Like dissolves like
Lipids Definition Definition: Water insoluble No common structure (though generally large R groups) Water Solubility (Hydrophilic) What makes molecules water soluble (hydrophilic)? Like dissolves like
Supplementary Figure 1.
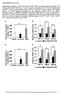 Supplementary Figure 1. FGF21 does not exert direct effects on hepatic glucose production. The liver explants from C57BL/6J mice (A, B) or primary rat hepatocytes (C, D) were incubated with rmfgf21 (2
Supplementary Figure 1. FGF21 does not exert direct effects on hepatic glucose production. The liver explants from C57BL/6J mice (A, B) or primary rat hepatocytes (C, D) were incubated with rmfgf21 (2
Lipids Types, Food Sources, Functions
 Lipids Types, Food Sources, Functions What Are Lipids? Lipids Diverse group of molecules that are insoluble in water Fats The lipid content of diets and foods 1 Lipids in Body Cells and Tissues Types of
Lipids Types, Food Sources, Functions What Are Lipids? Lipids Diverse group of molecules that are insoluble in water Fats The lipid content of diets and foods 1 Lipids in Body Cells and Tissues Types of
Prostaglandins And Other Biologically Active Lipids
 Prostaglandins And Other Biologically Active Lipids W. M. Grogan, Ph.D. OBJECTIVES After studying the material of this lecture, the student will: 1. Draw the structure of a prostaglandin, name the fatty
Prostaglandins And Other Biologically Active Lipids W. M. Grogan, Ph.D. OBJECTIVES After studying the material of this lecture, the student will: 1. Draw the structure of a prostaglandin, name the fatty
Life History of A Drug
 DRUG ACTION & PHARMACODYNAMIC M. Imad Damaj, Ph.D. Associate Professor Pharmacology and Toxicology Smith 652B, 828-1676, mdamaj@hsc.vcu.edu Life History of A Drug Non-Specific Mechanims Drug-Receptor Interaction
DRUG ACTION & PHARMACODYNAMIC M. Imad Damaj, Ph.D. Associate Professor Pharmacology and Toxicology Smith 652B, 828-1676, mdamaj@hsc.vcu.edu Life History of A Drug Non-Specific Mechanims Drug-Receptor Interaction
PPAR history of research
 PPAR Rubens, 1640 PPAR history of research number of publications 3000 2000 1000 0 till now: : 16 296 publications 1985 1990 1995 2000 2005 year liver, brown adipocytes, kidney, heart, skeletal muscles,
PPAR Rubens, 1640 PPAR history of research number of publications 3000 2000 1000 0 till now: : 16 296 publications 1985 1990 1995 2000 2005 year liver, brown adipocytes, kidney, heart, skeletal muscles,
Males- Western Diet WT KO Age (wks) Females- Western Diet WT KO Age (wks)
 Relative Arv1 mrna Adrenal 33.48 +/- 6.2 Skeletal Muscle 22.4 +/- 4.93 Liver 6.41 +/- 1.48 Heart 5.1 +/- 2.3 Brain 4.98 +/- 2.11 Ovary 4.68 +/- 2.21 Kidney 3.98 +/-.39 Lung 2.15 +/-.6 Inguinal Subcutaneous
Relative Arv1 mrna Adrenal 33.48 +/- 6.2 Skeletal Muscle 22.4 +/- 4.93 Liver 6.41 +/- 1.48 Heart 5.1 +/- 2.3 Brain 4.98 +/- 2.11 Ovary 4.68 +/- 2.21 Kidney 3.98 +/-.39 Lung 2.15 +/-.6 Inguinal Subcutaneous
Human LDL ELISA Kit. Innovative Research, Inc.
 Human LDL ELISA Kit Catalog No: IRKTAH2582 Lot No: SAMPLE INTRODUCTION Human low-density lipoprotein (LDL) transports cholesterol from the liver to tissues where it is incorporated into cell membranes.
Human LDL ELISA Kit Catalog No: IRKTAH2582 Lot No: SAMPLE INTRODUCTION Human low-density lipoprotein (LDL) transports cholesterol from the liver to tissues where it is incorporated into cell membranes.
Lipids digestion and absorption, Biochemistry II
 Lipids digestion and absorption, blood plasma lipids, lipoproteins Biochemistry II Lecture 1 2008 (J.S.) Triacylglycerols (as well as free fatty acids and both free and esterified cholesterol) are very
Lipids digestion and absorption, blood plasma lipids, lipoproteins Biochemistry II Lecture 1 2008 (J.S.) Triacylglycerols (as well as free fatty acids and both free and esterified cholesterol) are very
Chemically Reactive Drug Metabolites in Drug Discovery and Development Detection, Evaluation, and Risk Assessment
 Chemically Reactive Drug Metabolites in Drug Discovery and Development Detection, Evaluation, and Risk Assessment Pacific Northwest Bio Meeting Seattle, WA, August 14, 2012 Thomas A. Baillie, PhD, DSc
Chemically Reactive Drug Metabolites in Drug Discovery and Development Detection, Evaluation, and Risk Assessment Pacific Northwest Bio Meeting Seattle, WA, August 14, 2012 Thomas A. Baillie, PhD, DSc
Source Variation in Antioxidant Capacity of Cranberries from Eight U.S. Cultivars
 33 Source Variation in Antioxidant Capacity of Cranberries from Eight U.S. Cultivars Peter J. Schaaf Faculty Sponsors: Margaret A. Maher and Ted Wilson, Departments of Biology/Microbiology ABSTRACT Antioxidants
33 Source Variation in Antioxidant Capacity of Cranberries from Eight U.S. Cultivars Peter J. Schaaf Faculty Sponsors: Margaret A. Maher and Ted Wilson, Departments of Biology/Microbiology ABSTRACT Antioxidants
Catalysts of Lipid Oxidation
 Catalysts of Lipid Oxidation Iron The most important nonenzymic catalyst for initiation of lipid peroxidation The most abundant transitional metal in biological systems Possibility of various oxidation
Catalysts of Lipid Oxidation Iron The most important nonenzymic catalyst for initiation of lipid peroxidation The most abundant transitional metal in biological systems Possibility of various oxidation
CHAPTER 28 LIPIDS SOLUTIONS TO REVIEW QUESTIONS
 HAPTER 28 LIPIDS SLUTINS T REVIEW QUESTINS 1. The lipids, which are dissimilar substances, are arbitrarily classified as a group on the basis of their solubility in fat solvents and their insolubility
HAPTER 28 LIPIDS SLUTINS T REVIEW QUESTINS 1. The lipids, which are dissimilar substances, are arbitrarily classified as a group on the basis of their solubility in fat solvents and their insolubility
ANSC/NUTR 618 LIPIDS & LIPID METABOLISM Lipoprotein Metabolism
 ANSC/NUTR 618 LIPIDS & LIPID METABOLISM Lipoprotein Metabolism I. Chylomicrons (exogenous pathway) A. 83% triacylglycerol, 2% protein, 8% cholesterol plus cholesterol esters, 7% phospholipid (esp. phosphatidylcholine)
ANSC/NUTR 618 LIPIDS & LIPID METABOLISM Lipoprotein Metabolism I. Chylomicrons (exogenous pathway) A. 83% triacylglycerol, 2% protein, 8% cholesterol plus cholesterol esters, 7% phospholipid (esp. phosphatidylcholine)
Integration Of Metabolism
 Integration Of Metabolism Metabolism Consist of Highly Interconnected Pathways The basic strategy of catabolic metabolism is to form ATP, NADPH, and building blocks for biosyntheses. 1. ATP is the universal
Integration Of Metabolism Metabolism Consist of Highly Interconnected Pathways The basic strategy of catabolic metabolism is to form ATP, NADPH, and building blocks for biosyntheses. 1. ATP is the universal
Antihyperlipidemic Drugs
 Antihyperlipidemic Drugs Hyperlipidemias. Hyperlipoproteinemias. Hyperlipemia. Hypercholestrolemia. Direct relationship with acute pancreatitis and atherosclerosis Structure Lipoprotein Particles Types
Antihyperlipidemic Drugs Hyperlipidemias. Hyperlipoproteinemias. Hyperlipemia. Hypercholestrolemia. Direct relationship with acute pancreatitis and atherosclerosis Structure Lipoprotein Particles Types
The Pennsylvania State University. The Graduate School. The Huck Institutes of the Life Sciences ROLES OF PEROXISOME PROLIFERATOR-ACTIVATED RECEPTOR-
 The Pennsylvania State University The Graduate School The Huck Institutes of the Life Sciences ROLES OF PEROXISOME PROLIFERATOR-ACTIVATED RECEPTOR- BETA/DELTA AND B-CELL LYMPHOMA 6 IN PANCREATIC CANCER
The Pennsylvania State University The Graduate School The Huck Institutes of the Life Sciences ROLES OF PEROXISOME PROLIFERATOR-ACTIVATED RECEPTOR- BETA/DELTA AND B-CELL LYMPHOMA 6 IN PANCREATIC CANCER
Chem 60 Takehome Test 2 Student Section
 Multiple choice: 1 point each. Mark only one answer for each question. 1. are composed primarily of carbon and hydrogen, but may also include oxygen, nitrogen, sulfur, phosphorus, and a few other elements.
Multiple choice: 1 point each. Mark only one answer for each question. 1. are composed primarily of carbon and hydrogen, but may also include oxygen, nitrogen, sulfur, phosphorus, and a few other elements.
RayBio Human PPAR-alpha Transcription Factor Activity Assay Kit
 RayBio Human PPAR-alpha Transcription Factor Activity Assay Kit Catalog #: TFEH-PPARa User Manual Jan 5, 2018 3607 Parkway Lane, Suite 200 Norcross, GA 30092 Tel: 1-888-494-8555 (Toll Free) or 770-729-2992,
RayBio Human PPAR-alpha Transcription Factor Activity Assay Kit Catalog #: TFEH-PPARa User Manual Jan 5, 2018 3607 Parkway Lane, Suite 200 Norcross, GA 30092 Tel: 1-888-494-8555 (Toll Free) or 770-729-2992,
The antiparasitic drug ivermectin is a novel FXR ligand that regulates metabolism
 Supplementary Information The antiparasitic drug ivermectin is a novel FXR ligand that regulates metabolism Address correspondence to Yong Li (yongli@xmu.edu.cn, Tel: 86-592-218151) GW464 CDCA Supplementary
Supplementary Information The antiparasitic drug ivermectin is a novel FXR ligand that regulates metabolism Address correspondence to Yong Li (yongli@xmu.edu.cn, Tel: 86-592-218151) GW464 CDCA Supplementary
Status of LDL Oxidation and antioxidant potential of LDL in Type II Diabetes Mellitus
 Biomedical Research 2010; 21 (4): 416-418 Status of LDL Oxidation and antioxidant potential of LDL in Type II Singh N, Singh N, Singh S K, Singh A K, Bhargava V. Department of Biochemistry, G. R. Medical
Biomedical Research 2010; 21 (4): 416-418 Status of LDL Oxidation and antioxidant potential of LDL in Type II Singh N, Singh N, Singh S K, Singh A K, Bhargava V. Department of Biochemistry, G. R. Medical
Definition: Water insoluble No common structure (though generally large R groups)
 Lipids Definition Definition: Water insoluble No common structure (though generally large R groups) Water Solubility (Hydrophilic) What makes molecules water soluble (hydrophilic)? Like dissolves like
Lipids Definition Definition: Water insoluble No common structure (though generally large R groups) Water Solubility (Hydrophilic) What makes molecules water soluble (hydrophilic)? Like dissolves like
Supplementary Figure 1. Characterization of human carotid plaques. (a) Flash-frozen human plaques were separated into vulnerable (V) and stable (S),
 Supplementary Figure 1. Characterization of human carotid plaques. (a) Flash-frozen human plaques were separated into vulnerable (V) and stable (S), regions which were then quantified for mean fluorescence
Supplementary Figure 1. Characterization of human carotid plaques. (a) Flash-frozen human plaques were separated into vulnerable (V) and stable (S), regions which were then quantified for mean fluorescence
D. Vitamin K -- Group of naphthoquinones having antihemorrhagic activity.
 D. Vitamin K -- Group of naphthoquinones having antihemorrhagic activity. 1. Structures VITAMIN K1; Phytonadione; 2-Methyl-3-phytyl-1,4-naphthoquinone VITAMIN K2; Menaquinone-6 and 7; N=6,7 n 2. Function
D. Vitamin K -- Group of naphthoquinones having antihemorrhagic activity. 1. Structures VITAMIN K1; Phytonadione; 2-Methyl-3-phytyl-1,4-naphthoquinone VITAMIN K2; Menaquinone-6 and 7; N=6,7 n 2. Function
Mechanistic Toxicology
 SECOND EDITION Mechanistic Toxicology The Molecular Basis of How Chemicals Disrupt Biological Targets URS A. BOELSTERLI CRC Press Tavlor & France Croup CRC Press is an imp^t o* :H Taylor H Francn C'r,,jpi
SECOND EDITION Mechanistic Toxicology The Molecular Basis of How Chemicals Disrupt Biological Targets URS A. BOELSTERLI CRC Press Tavlor & France Croup CRC Press is an imp^t o* :H Taylor H Francn C'r,,jpi
Multiple choice: Circle the best answer on this exam. There are 12 multiple choice questions, each question is worth 3 points.
 CHEM 4420 Exam 4 Spring 2015 Dr. Stone Page 1 of 6 Name Use complete sentences when requested. There are 120 possible points on this exam. Therefore there are 20 bonus points. Multiple choice: Circle the
CHEM 4420 Exam 4 Spring 2015 Dr. Stone Page 1 of 6 Name Use complete sentences when requested. There are 120 possible points on this exam. Therefore there are 20 bonus points. Multiple choice: Circle the
PROGRESS TOWARDS OBJECTIVES, SIGNIFICANT RESULTS, DEVIATIONS, REASONS FOR FAILING, CORRECTIVE ACTIONS
 FINAL REPORT Grant Agreement PIEF-GA-2011-303197 Obesity and Light PROGRESS TOWARDS OBJECTIVES, SIGNIFICANT RESULTS, DEVIATIONS, REASONS FOR FAILING, CORRECTIVE ACTIONS For the total period (2 years) three
FINAL REPORT Grant Agreement PIEF-GA-2011-303197 Obesity and Light PROGRESS TOWARDS OBJECTIVES, SIGNIFICANT RESULTS, DEVIATIONS, REASONS FOR FAILING, CORRECTIVE ACTIONS For the total period (2 years) three
Product Manual. Human LDLR ELISA Kit. Catalog Number. FOR RESEARCH USE ONLY Not for use in diagnostic procedures
 Product Manual Human LDLR ELISA Kit Catalog Number STA-386 96 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Cholesterol is an essential component of cellular membranes,
Product Manual Human LDLR ELISA Kit Catalog Number STA-386 96 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Cholesterol is an essential component of cellular membranes,
Factors to Consider in the Study of Biomolecules
 Factors to Consider in the Study of Biomolecules What are the features of the basic building blocks? (ex: monosaccharides, alcohols, fatty acids, amino acids) 1) General structure and functional groups
Factors to Consider in the Study of Biomolecules What are the features of the basic building blocks? (ex: monosaccharides, alcohols, fatty acids, amino acids) 1) General structure and functional groups
INTERACTION DRUG BODY
 INTERACTION DRUG BODY What the drug does to the body What the body does to the drug Receptors - intracellular receptors - membrane receptors - Channel receptors - G protein-coupled receptors - Tyrosine-kinase
INTERACTION DRUG BODY What the drug does to the body What the body does to the drug Receptors - intracellular receptors - membrane receptors - Channel receptors - G protein-coupled receptors - Tyrosine-kinase
OxiSelect Malondialdehyde (MDA) Immunoblot Kit
 Product Manual OxiSelect Malondialdehyde (MDA) Immunoblot Kit Catalog Number STA- 331 10 blots FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Lipid peroxidation is a well-defined
Product Manual OxiSelect Malondialdehyde (MDA) Immunoblot Kit Catalog Number STA- 331 10 blots FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Lipid peroxidation is a well-defined
Eicosapentaenoic Acid and Docosahexaenoic Acid: Are They Different?
 Eicosapentaenoic Acid and Docosahexaenoic Acid: Are They Different? Trevor A Mori, Ph.D., Professor, School of Medicine and Pharmacology, Royal Perth Hospital Unit, University of Western Australia, Perth,
Eicosapentaenoic Acid and Docosahexaenoic Acid: Are They Different? Trevor A Mori, Ph.D., Professor, School of Medicine and Pharmacology, Royal Perth Hospital Unit, University of Western Australia, Perth,
ab Lipid Hydroperoxide (LPO) Assay Kit
 ab133085 Lipid Hydroperoxide (LPO) Assay Kit Instructions for Use For the measurement of hydroperoxides directly utilizing the redox reactions with ferrous ions. This product is for research use only and
ab133085 Lipid Hydroperoxide (LPO) Assay Kit Instructions for Use For the measurement of hydroperoxides directly utilizing the redox reactions with ferrous ions. This product is for research use only and
Nature Immunology: doi: /ni Supplementary Figure 1. IC261 inhibits a virus-induced type I interferon response.
 Supplementary Figure 1 IC261 inhibits a virus-induced type I interferon response. (a) HEK293T cells were cultured in 384 wells and transiently transfected with 50 ng of the IFN-β promoter-luc construct
Supplementary Figure 1 IC261 inhibits a virus-induced type I interferon response. (a) HEK293T cells were cultured in 384 wells and transiently transfected with 50 ng of the IFN-β promoter-luc construct
Title. YOON, Seokjoo; MARUYAMA, Yutaka; KA FUJITA, Shoichi. Author(s) Issue Date /jjvr
 Title Application of FT-IR and ESR spectr study of CCl_4-induced peroxidation Author(s) YOON, Seokjoo; MARUYAMA, Yutaka; KA FUJITA, Shoichi Citation Japanese Journal of Veterinary Rese Issue Date 2000-02-29
Title Application of FT-IR and ESR spectr study of CCl_4-induced peroxidation Author(s) YOON, Seokjoo; MARUYAMA, Yutaka; KA FUJITA, Shoichi Citation Japanese Journal of Veterinary Rese Issue Date 2000-02-29
Metabolism of cardiac muscle. Dr. Mamoun Ahram Cardiovascular system, 2013
 Metabolism of cardiac muscle Dr. Mamoun Ahram Cardiovascular system, 2013 References This lecture Mark s Basic Medical Biochemistry, 4 th ed., p. 890-891 Hand-out Why is this topic important? Heart failure
Metabolism of cardiac muscle Dr. Mamoun Ahram Cardiovascular system, 2013 References This lecture Mark s Basic Medical Biochemistry, 4 th ed., p. 890-891 Hand-out Why is this topic important? Heart failure
Figure 1. Effects of FGF21 on adipose tissue. (A) Representative histological. findings of epididymal adipose tissue (B) mrna expression of
 SUPPLEMENTAL MATERIAL EN-12-2276 Figure 1. Effects of FGF21 on adipose tissue. (A) Representative histological findings of epididymal adipose tissue (B) mrna expression of adipocytokines in adipose tissue.
SUPPLEMENTAL MATERIAL EN-12-2276 Figure 1. Effects of FGF21 on adipose tissue. (A) Representative histological findings of epididymal adipose tissue (B) mrna expression of adipocytokines in adipose tissue.
UCLA Nutrition Noteworthy
 UCLA Nutrition Noteworthy Title Effects of Omega-6 and Omega-3 Polyunsaturated Fatty Acids on Breast Cancer Permalink https://escholarship.org/uc/item/12g6398b Journal Nutrition Noteworthy, 2(1) ISSN 1556-1895
UCLA Nutrition Noteworthy Title Effects of Omega-6 and Omega-3 Polyunsaturated Fatty Acids on Breast Cancer Permalink https://escholarship.org/uc/item/12g6398b Journal Nutrition Noteworthy, 2(1) ISSN 1556-1895
Crosstalk between Adiponectin and IGF-IR in breast cancer. Prof. Young Jin Suh Department of Surgery The Catholic University of Korea
 Crosstalk between Adiponectin and IGF-IR in breast cancer Prof. Young Jin Suh Department of Surgery The Catholic University of Korea Obesity Chronic, multifactorial disorder Hypertrophy and hyperplasia
Crosstalk between Adiponectin and IGF-IR in breast cancer Prof. Young Jin Suh Department of Surgery The Catholic University of Korea Obesity Chronic, multifactorial disorder Hypertrophy and hyperplasia
Lipoproteins Metabolism Reference: Campbell Biochemistry and Lippincott s Biochemistry
 Lipoproteins Metabolism Reference: Campbell Biochemistry and Lippincott s Biochemistry Learning Objectives 1. Define lipoproteins and explain the rationale of their formation in blood. 2. List different
Lipoproteins Metabolism Reference: Campbell Biochemistry and Lippincott s Biochemistry Learning Objectives 1. Define lipoproteins and explain the rationale of their formation in blood. 2. List different
2. List routes of exposure in the order of most rapid response.
 Practice Test questions: 1. What are the two areas of toxicology that a regulatory toxicologist must integrate in order to determine the "safety" of any chemical? 2. List routes of exposure in the order
Practice Test questions: 1. What are the two areas of toxicology that a regulatory toxicologist must integrate in order to determine the "safety" of any chemical? 2. List routes of exposure in the order
Introduction to the Study of Lipids
 Introduction to the Study of Lipids Factors to Consider in the Study of Biomolecules What are the features of the basic building blocks? (ex: monosaccharides, alcohols, fatty acids, amino acids) 1) General
Introduction to the Study of Lipids Factors to Consider in the Study of Biomolecules What are the features of the basic building blocks? (ex: monosaccharides, alcohols, fatty acids, amino acids) 1) General
General Biochemistry-1 BCH 202
 General Biochemistry-1 BCH 202 1 I would like to acknowledge Dr. Farid Ataya for his valuable input & help in this course. 2 Outline Lipids Definition, function, fatty acids, classification: simple lipids:
General Biochemistry-1 BCH 202 1 I would like to acknowledge Dr. Farid Ataya for his valuable input & help in this course. 2 Outline Lipids Definition, function, fatty acids, classification: simple lipids:
DM, NAFLD, and conjugated linoleic acid (omega 6); what is the link
 DM, NAFLD, and conjugated linoleic acid (omega 6); what is the link Mona Hegazy Professor of Internal Medicine Hepatology Department Cairo University Egypt Agenda Congugated lionleic fatty acid NAFLD
DM, NAFLD, and conjugated linoleic acid (omega 6); what is the link Mona Hegazy Professor of Internal Medicine Hepatology Department Cairo University Egypt Agenda Congugated lionleic fatty acid NAFLD
Unit IV Problem 3 Biochemistry: Cholesterol Metabolism and Lipoproteins
 Unit IV Problem 3 Biochemistry: Cholesterol Metabolism and Lipoproteins - Cholesterol: It is a sterol which is found in all eukaryotic cells and contains an oxygen (as a hydroxyl group OH) on Carbon number
Unit IV Problem 3 Biochemistry: Cholesterol Metabolism and Lipoproteins - Cholesterol: It is a sterol which is found in all eukaryotic cells and contains an oxygen (as a hydroxyl group OH) on Carbon number
Taylor Yohe. Project Advisor: Dr. Martha A. Belury. Department of Human Nutrition at the Ohio State University
 Atherosclerosis Development and the Inflammatory Response of Hepatocytes to Sesame Oil Supplementation Taylor Yohe Project Advisor: Dr. Martha A. Belury Department of Human Nutrition at the Ohio State
Atherosclerosis Development and the Inflammatory Response of Hepatocytes to Sesame Oil Supplementation Taylor Yohe Project Advisor: Dr. Martha A. Belury Department of Human Nutrition at the Ohio State
Biologic Oxidation BIOMEDICAL IMPORTAN
 Biologic Oxidation BIOMEDICAL IMPORTAN Chemically, oxidation is defined as the removal of electrons and reduction as the gain of electrons. Thus, oxidation is always accompanied by reduction of an electron
Biologic Oxidation BIOMEDICAL IMPORTAN Chemically, oxidation is defined as the removal of electrons and reduction as the gain of electrons. Thus, oxidation is always accompanied by reduction of an electron
14.15 Lecture Protein-bound lipid peroxidation products: structures and functions K. Uchida (Japan)
 PROGRAM FRIDAY 16TH JUNE 2006 14.00 Opening Remarks U.M. Marinari, G. Poli Session I LIPID PEROXIDATION PRODUCTS AND ANALYTICAL PROCEDURES Chairmen: P. Eckl, C. Spickett 14.15 Protein-bound lipid peroxidation
PROGRAM FRIDAY 16TH JUNE 2006 14.00 Opening Remarks U.M. Marinari, G. Poli Session I LIPID PEROXIDATION PRODUCTS AND ANALYTICAL PROCEDURES Chairmen: P. Eckl, C. Spickett 14.15 Protein-bound lipid peroxidation
By: Dr Mehrnoosh Shanaki
 Resveratrol could partly improve the crosstalk between canonical β-catenin/wnt and FOXO pathways in coronary artery disease patients with metabolic syndrome: A case control study By: Dr Mehrnoosh Shanaki
Resveratrol could partly improve the crosstalk between canonical β-catenin/wnt and FOXO pathways in coronary artery disease patients with metabolic syndrome: A case control study By: Dr Mehrnoosh Shanaki
LDL (Human) ELISA Kit
 LDL (Human) ELISA Kit Cat. No.:DEIA3864 Pkg.Size:96T Intended use This immunoassay kit allows for the specific measurement of human low density lipoprotein, LDL concentrations in cell culture supernates,
LDL (Human) ELISA Kit Cat. No.:DEIA3864 Pkg.Size:96T Intended use This immunoassay kit allows for the specific measurement of human low density lipoprotein, LDL concentrations in cell culture supernates,
Nafith Abu Tarboush DDS, MSc, PhD
 Nafith Abu Tarboush DDS, MSc, PhD natarboush@ju.edu.jo www.facebook.com/natarboush Lipids (cholesterol, cholesterol esters, phospholipids & triacylglycerols) combined with proteins (apolipoprotein) in
Nafith Abu Tarboush DDS, MSc, PhD natarboush@ju.edu.jo www.facebook.com/natarboush Lipids (cholesterol, cholesterol esters, phospholipids & triacylglycerols) combined with proteins (apolipoprotein) in
Biochemistry: A Short Course
 Tymoczko Berg Stryer Biochemistry: A Short Course Second Edition CHAPTER 28 Fatty Acid Synthesis 2013 W. H. Freeman and Company Chapter 28 Outline 1. The first stage of fatty acid synthesis is transfer
Tymoczko Berg Stryer Biochemistry: A Short Course Second Edition CHAPTER 28 Fatty Acid Synthesis 2013 W. H. Freeman and Company Chapter 28 Outline 1. The first stage of fatty acid synthesis is transfer
Chapter 26 Biochemistry 5th edition. phospholipids. Sphingolipids. Cholesterol. db=books&itool=toolbar
 http://www.ncbi.nlm.nih.gov/sites/entrez? db=books&itool=toolbar 1 The surface of a soap bubble is a bilayer formed by detergent molecules 2 Chapter 26 Biochemistry 5th edition phospholipids Sphingolipids
http://www.ncbi.nlm.nih.gov/sites/entrez? db=books&itool=toolbar 1 The surface of a soap bubble is a bilayer formed by detergent molecules 2 Chapter 26 Biochemistry 5th edition phospholipids Sphingolipids
Alpha Lipoic Acid Snapshot Monograph
 vitamins minerals nutrients Alpha Lipoic Acid Snapshot Monograph Alpha lipoic Acid Most Frequent Reported Uses: - Antioxidant - Peripheral neuropathy - Improves insulin signaling and regulation of appetite
vitamins minerals nutrients Alpha Lipoic Acid Snapshot Monograph Alpha lipoic Acid Most Frequent Reported Uses: - Antioxidant - Peripheral neuropathy - Improves insulin signaling and regulation of appetite
Bringing metabolic profiling into clinical practice. Linda Mustelin, MD, PhD, MPH Senior Medical Scientist Nightingale Health
 Bringing metabolic profiling into clinical practice Linda Mustelin, MD, PhD, MPH Senior Medical Scientist Nightingale Health Nightingale Health Ltd. Finnish biotech company specialized in comprehensive
Bringing metabolic profiling into clinical practice Linda Mustelin, MD, PhD, MPH Senior Medical Scientist Nightingale Health Nightingale Health Ltd. Finnish biotech company specialized in comprehensive
shehab Moh Tarek ... ManarHajeer
 3 shehab Moh Tarek... ManarHajeer In the previous lecture we discussed the accumulation of oxygen- derived free radicals as a mechanism of cell injury, we covered their production and their pathologic
3 shehab Moh Tarek... ManarHajeer In the previous lecture we discussed the accumulation of oxygen- derived free radicals as a mechanism of cell injury, we covered their production and their pathologic
Porphyrins: Chemistry and Biology
 Porphyrins: Chemistry and Biology 20.109 Lecture 6 24 February, 2011 Goals Explore some essential roles of heme in biology Appreciate how ature has used the same cofactor to achieve diverse functions Gain
Porphyrins: Chemistry and Biology 20.109 Lecture 6 24 February, 2011 Goals Explore some essential roles of heme in biology Appreciate how ature has used the same cofactor to achieve diverse functions Gain
The Orphan Nuclear Receptors: Unrecognized Targets of Botanical Medicines?
 The Orphan Nuclear Receptors: Unrecognized Targets of Botanical Medicines? Kevin Spelman, PhD, MCPP These notes are designed as a primer for the lecture and do not necessarily represent lecture content.
The Orphan Nuclear Receptors: Unrecognized Targets of Botanical Medicines? Kevin Spelman, PhD, MCPP These notes are designed as a primer for the lecture and do not necessarily represent lecture content.
THE EFFECT OF VITAMIN-C THERAPY ON HYPERGLYCEMIA, HYPERLIPIDEMIA AND NON HIGH DENSITY LIPOPROTEIN LEVEL IN TYPE 2 DIABETES
 Int. J. LifeSc. Bt & Pharm. Res. 2013 Varikasuvu Seshadri Reddy et al., 2013 Review Article ISSN 2250-3137 www.ijlbpr.com Vol. 2, No. 1, January 2013 2013 IJLBPR. All Rights Reserved THE EFFECT OF VITAMIN-C
Int. J. LifeSc. Bt & Pharm. Res. 2013 Varikasuvu Seshadri Reddy et al., 2013 Review Article ISSN 2250-3137 www.ijlbpr.com Vol. 2, No. 1, January 2013 2013 IJLBPR. All Rights Reserved THE EFFECT OF VITAMIN-C
BIOL2171 ANU TCA CYCLE
 TCA CYCLE IMPORTANCE: Oxidation of 2C Acetyl Co-A 2CO 2 + 3NADH + FADH 2 (8e-s donated to O 2 in the ETC) + GTP (energy) + Heat OVERVIEW: Occurs In the mitochondrion matrix. 1. the acetyl portion of acetyl-coa
TCA CYCLE IMPORTANCE: Oxidation of 2C Acetyl Co-A 2CO 2 + 3NADH + FADH 2 (8e-s donated to O 2 in the ETC) + GTP (energy) + Heat OVERVIEW: Occurs In the mitochondrion matrix. 1. the acetyl portion of acetyl-coa
Lipid Hydroperoxide (LPO) Assay Kit
 Lipid Hydroperoxide (LPO) Assay Kit Catalog Number KA1328 96 assays Version: 05 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Background... 3 Principle of the Assay...
Lipid Hydroperoxide (LPO) Assay Kit Catalog Number KA1328 96 assays Version: 05 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Background... 3 Principle of the Assay...
Sphingolipids. Sphingolipids are an additional type of membrane lipids, after glycerophospholipids, galactolipids and sulfolipids
 Lipids 2 Steven E. Massey, Ph.D. Assistant Professor of Bioinformatics Department of Biology and Environmental Sciences University of Puerto Rico Río Piedras Office & Lab: Bioinformatics Lab NCN#343B Tel:
Lipids 2 Steven E. Massey, Ph.D. Assistant Professor of Bioinformatics Department of Biology and Environmental Sciences University of Puerto Rico Río Piedras Office & Lab: Bioinformatics Lab NCN#343B Tel:
OxiSelect MDA Adduct ELISA Kit
 Revised Protocol Product Manual OxiSelect MDA Adduct ELISA Kit Catalog Number STA-332 96 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Lipid peroxidation is a well-defined
Revised Protocol Product Manual OxiSelect MDA Adduct ELISA Kit Catalog Number STA-332 96 assays FOR RESEARCH USE ONLY Not for use in diagnostic procedures Introduction Lipid peroxidation is a well-defined
Estimation of glucose in blood serum
 Estimation of glucose in blood serum Enzymatic estimation of glucose uses a reagent containing two enzymes and a chromogen. Glucose oxidase catalyses the oxidation of glucose to gluconolactone with the
Estimation of glucose in blood serum Enzymatic estimation of glucose uses a reagent containing two enzymes and a chromogen. Glucose oxidase catalyses the oxidation of glucose to gluconolactone with the
Metabolism of acylglycerols and sphingolipids. Martina Srbová
 Metabolism of acylglycerols and sphingolipids Martina Srbová Types of glycerolipids and sphingolipids 1. Triacylglycerols function as energy reserves adipose tissue (storage of triacylglycerol), lipoproteins
Metabolism of acylglycerols and sphingolipids Martina Srbová Types of glycerolipids and sphingolipids 1. Triacylglycerols function as energy reserves adipose tissue (storage of triacylglycerol), lipoproteins
Supporting Information
 Supporting Information Chlorinated Polyfluorinated Ether Sulfonates Exhibit Higher Activity towards Peroxisome Proliferator-Activated Receptors Signaling Pathways than Perfluorooctane Sulfonate Chuan-Hai
Supporting Information Chlorinated Polyfluorinated Ether Sulfonates Exhibit Higher Activity towards Peroxisome Proliferator-Activated Receptors Signaling Pathways than Perfluorooctane Sulfonate Chuan-Hai
Iron Chelates and Unwanted Biological Oxidations
 The Virtual Free Radical School Iron Chelates and Unwanted Biological Oxidations Kevin D. Welch and Steven D. Aust Department of Chemistry and Biochemistry Biotechnology Center Utah State University Logan,
The Virtual Free Radical School Iron Chelates and Unwanted Biological Oxidations Kevin D. Welch and Steven D. Aust Department of Chemistry and Biochemistry Biotechnology Center Utah State University Logan,
Glutathione / Thioredoxin Nrf2 & Hyperoxia
 Glutathione / Thioredoxin Nrf2 & Hyperoxia Trent E. Tipple, MD Principal Investigator, Center for Perinatal Research The Research Institute at Nationwide Children s Hospital Assistant Professor of Pediatrics,
Glutathione / Thioredoxin Nrf2 & Hyperoxia Trent E. Tipple, MD Principal Investigator, Center for Perinatal Research The Research Institute at Nationwide Children s Hospital Assistant Professor of Pediatrics,
Plasma lipoproteins & atherosclerosis by. Prof.Dr. Maha M. Sallam
 Biochemistry Department Plasma lipoproteins & atherosclerosis by Prof.Dr. Maha M. Sallam 1 1. Recognize structures,types and role of lipoproteins in blood (Chylomicrons, VLDL, LDL and HDL). 2. Explain
Biochemistry Department Plasma lipoproteins & atherosclerosis by Prof.Dr. Maha M. Sallam 1 1. Recognize structures,types and role of lipoproteins in blood (Chylomicrons, VLDL, LDL and HDL). 2. Explain
Biosynthesis of Triacylglycerides (TG) in liver. Mobilization of stored fat and oxidation of fatty acids
 Biosynthesis of Triacylglycerides (TG) in liver Mobilization of stored fat and oxidation of fatty acids Activation of hormone sensitive lipase This enzyme is activated when phosphorylated (3,5 cyclic AMPdependent
Biosynthesis of Triacylglycerides (TG) in liver Mobilization of stored fat and oxidation of fatty acids Activation of hormone sensitive lipase This enzyme is activated when phosphorylated (3,5 cyclic AMPdependent
