Chapter 21 The Chemistry of Carboxylic Acid Deriva7ves
|
|
|
- Rebecca Shanna Armstrong
- 5 years ago
- Views:
Transcription
1 Organic Chemistry, 5th ed. Marc Loudon Chapter 21 The Chemistry of Carboxylic Acid Deriva7ves Eric J. Kantorowski California Polytechnic State University San Luis Obispo, CA
2 Chapter 21 Overview 21.1 Nomenclature and Classifica5on of Carboxylic Acid Deriva5ves 21.2 Structures of Carboxylic Acid Deriva5ves 21.3 Physical Proper5es of Carboxylic Acid Deriva5ves 21.4 Spectroscopy of Carboxylic Acid Deriva5ves 21.5 Basicity of Carboxylic Acid Deriva5ves 21.6 Introduc5on to Reac5ons of Carboxylic Acid Deriva5ves 21.7 Hydrolysis of Carboxylic Acid Deriva5ves 21.8 Reac5ons of Carboxylic Acid Deriva5ves with Nucleophiles 21.9 Reduc5on of Carboxylic Acid Deriva5ves 2
3 Chapter 21 Overview Reac5ons of Carboxylic Acid Deriva5ves with Organometallic Reagents Synthesis of Carboxylic Acid Deriva5ves Use and Occurrence of Carboxylic Acids and Their Deriva5ves 3
4 Carboxylic Acid Deriva8ves A carboxylic acid deriva8ve is a compound that can be hydrolyzed under acidic of basic condi5ons to give the related carboxylic acid 21.1 Nomenclature and Classifica7on of Carboxylic Acid Deriva7ves 4
5 Structures of Carboxylic Acid Deriva8ves 21.1 Nomenclature and Classifica7on of Carboxylic Acid Deriva7ves 5
6 Esters and Lactones 21.1 Nomenclature and Classifica7on of Carboxylic Acid Deriva7ves 6
7 Esters and Lactones Cyclic esters are called lactones The name is derived from the acid with the same number of carbons in the principle chain The ring size is denoted by a Greek leuer indica5ng the point of auachment 21.1 Nomenclature and Classifica7on of Carboxylic Acid Deriva7ves 7
8 Acid Halides Replace the ic ending with yl followed by the name of the halide 21.1 Nomenclature and Classifica7on of Carboxylic Acid Deriva7ves 8
9 Anhydrides The name is the parent acid is followed by anhydride 21.1 Nomenclature and Classifica7on of Carboxylic Acid Deriva7ves 9
10 Nitriles Drop the ic or oic ending and add onitrile The nitrile carbon counts as part of the chain length 21.1 Nomenclature and Classifica7on of Carboxylic Acid Deriva7ves 10
11 Amides, Lactams, and Imides Replace the ic or oic and add amide 21.1 Nomenclature and Classifica7on of Carboxylic Acid Deriva7ves 11
12 Amides, Lactams, and Imides Amides are classified according to the number of hydrogens on the amide nitrogen Use N for subs5tuents on the nitrogen 21.1 Nomenclature and Classifica7on of Carboxylic Acid Deriva7ves 12
13 Amides, Lactams, and Imides Cyclic amides are called lactams The nomenclature follows that of lactones 21.1 Nomenclature and Classifica7on of Carboxylic Acid Deriva7ves 13
14 Amides, Lactams, and Imides Imides are the nitrogen analogs of anhydrides Cyclic imides are of greater importance than open chain imides 21.1 Nomenclature and Classifica7on of Carboxylic Acid Deriva7ves 14
15 Nomenclature of Subs8tuent Groups The priori5es for ci5ng principle groups in a carboxylic acid deriva5ve are: 21.1 Nomenclature and Classifica7on of Carboxylic Acid Deriva7ves 15
16 Nomenclature of Subs8tuent Groups 21.1 Nomenclature and Classifica7on of Carboxylic Acid Deriva7ves 16
17 Carbonic Acid Deriva8ves Esters of carbonic acid are named like any other ester Other important deriva5ves include: 21.1 Nomenclature and Classifica7on of Carboxylic Acid Deriva7ves 17
18 Structures of Carboxylic Acid Deriva8ves The deriva5ves are very similar to other carbonyl compounds (bond lengths, angles) 21.2 Structures of Carboxylic Acid Deriva7ves 18
19 Structures of Carboxylic Acid Deriva8ves Because of the trigonal planar geometry at nitrogen, E and Z conformers are possible The interconversion is rela5vely rapid at room temperature (~10 s 1, E barrier 71 kj mol 1 ) 21.2 Structures of Carboxylic Acid Deriva7ves 19
20 Esters Polar; H bond acceptors only Typically vola5le, fragrant liquids Most are insoluble in water 21.3 Physical Proper7es of Carboxylic Acid Deriva7ves 20
21 Anhydrides and Acid Chlorides Lower anhydrides and acid chlorides are dense, water insoluble liquids Typically have acrid, piercing odors 21.3 Physical Proper7es of Carboxylic Acid Deriva7ves 21
22 Nitriles Highly polar (3.4 D for H 3 C C N) Nitriles larger than propionitrile are waterinsoluble Acetonitrile is a useful polar, apro5c solvent 21.3 Physical Proper7es of Carboxylic Acid Deriva7ves 22
23 Amides Polar with high boiling points Lower amides are water soluble Tend to aggregate due to H bonding 21.3 Physical Proper7es of Carboxylic Acid Deriva7ves 23
24 IR Spectroscopy 21.4 Spectroscopy of Carboxylic Acid Deriva7ves 24
25 IR Spectroscopy: Esters C=O stretch: cm 1 Readily dis5nguishes them from carboxylic acids, aldehydes, and ketones 21.4 Spectroscopy of Carboxylic Acid Deriva7ves 25
26 IR Spectroscopy: Anhydrides Two C=O stretches (also common for acid chlorides) Due to symmetric and asymmetric stretching 21.4 Spectroscopy of Carboxylic Acid Deriva7ves 26
27 IR Spectroscopy: Amides C=O stretch: cm 1 Much lower than other C=O stretches 21.4 Spectroscopy of Carboxylic Acid Deriva7ves 27
28 Primary, secondary, and ter5ary amides may also be differen5ated IR Spectroscopy: Amides 21.4 Spectroscopy of Carboxylic Acid Deriva7ves 28
29 IR Spectroscopy: Nitriles C N stretch: cm 1 Normally much stronger than C C stretch due to the large bond dipole 21.4 Spectroscopy of Carboxylic Acid Deriva7ves 29
30 1 H NMR Spectroscopy α Hydrogens next to carbonyl: δ Hydrogens on carbon next to alkoxy O: ~+0.6 ppm more than for that of alcohols and ethers 21.4 Spectroscopy of Carboxylic Acid Deriva7ves 30
31 1 H NMR Spectroscopy Amides display non equivalency for the two groups auached to the nitrogen They are diastereotopic Although these rotate at ~10 s 1, on the NMR 5me scale these groups appear frozen in posi5on 21.4 Spectroscopy of Carboxylic Acid Deriva7ves 31
32 C=O: δ C NMR Spectroscopy C N: δ Spectroscopy of Carboxylic Acid Deriva7ves 32
33 Basicity of Carboxylic Acid Deriva8ves Like carboxylic acids, their deriva5ves are also weakly basic Protona5on occurs on the carbonyl oxygen 21.5 Basicity of Carboxylic Acid Deriva7ves 33
34 Basicity of Carboxylic Acid Deriva8ves Amides are considerably more basic Nitriles are extremely weak bases 21.5 Basicity of Carboxylic Acid Deriva7ves 34
35 Reac8ons of Carboxylic Acid Deriva8ves There are three general categories: 1. Reac5ons at the carbonyl (or cyano) group a. at the C=O oxygen or C N nitrogen b. at the C=O carbon or C N carbon 2. Reac5ons involving the α carbon 3. Reac5ons at the nitrogen of amides 21.6 Introduc7on to the Reac7ons of Carboxylic Acid Deriva7ves 35
36 Reac8ons of Carboxylic Acid Deriva8ves Acyl subs8tu8on: Subs5tu5on at the carbonyl Addi7on is a typical reac5on of nitriles 21.6 Introduc7on to the Reac7ons of Carboxylic Acid Deriva7ves 36
37 Saponifica8on Hydrolysis of Esters Saponifica5on is effec5vely irreversible Classified as a nucleophilic acyl subs8tu8on 21.7 Hydrolysis of Carboxylic Acid Deriva7ves 37
38 Hydrolysis of Esters 21.7 Hydrolysis of Carboxylic Acid Deriva7ves 38
39 Hydrolysis of Esters Acid catalyzed hydrolysis The reac5on is slow and requires an excess of water 21.7 Hydrolysis of Carboxylic Acid Deriva7ves 39
40 Hydrolysis of Esters Classified as a nucleophilic acyl subs8tu8on 21.7 Hydrolysis of Carboxylic Acid Deriva7ves 40
41 Hydrolysis and Forma8on of Lactones 21.7 Hydrolysis of Carboxylic Acid Deriva7ves 41
42 Hydrolysis of Amides 21.7 Hydrolysis of Carboxylic Acid Deriva7ves 42
43 Hydrolysis of Nitriles More extreme condi5ons are required compared to those of esters and amides 21.7 Hydrolysis of Carboxylic Acid Deriva7ves 43
44 Hydrolysis of Nitriles Imidic acids can be viewed as the nitrogenanalogs of enols 21.7 Hydrolysis of Carboxylic Acid Deriva7ves 44
45 Hydrolysis of Nitriles Base promoted hydrolysis is also possible 21.7 Hydrolysis of Carboxylic Acid Deriva7ves 45
46 Hydrolysis of Acid Chlorides and Anhydrides Both react rapidly with water 21.7 Hydrolysis of Carboxylic Acid Deriva7ves 46
47 Nucleophilic Acyl Subs8tu8on Reac8ons The condi7ons for hydrolysis for these deriva5ves can differ considerably 21.7 Hydrolysis of Carboxylic Acid Deriva7ves 47
48 Nucleophilic Acyl Subs8tu8on Reac8ons 21.7 Hydrolysis of Carboxylic Acid Deriva7ves 48
49 Nucleophilic Acyl Subs8tu8on Reac8ons Energies in the reac5on coordinate diagram are affected by: Stability of the carbonyl compound The leaving group ability of X: 21.7 Hydrolysis of Carboxylic Acid Deriva7ves 49
50 Nucleophilic Acyl Subs8tu8on Reac8ons 21.7 Hydrolysis of Carboxylic Acid Deriva7ves 50
51 Reac8ons of Acid Chlorides with Nucleophiles 21.8 Reac7ons of Carboxylic Acid Deriva7ves with Nucleophiles 51
52 Reac8ons of Acid Chlorides with Nucleophiles 21.8 Reac7ons of Carboxylic Acid Deriva7ves with Nucleophiles 52
53 Reac8ons of Acid Chlorides with Nucleophiles Related reac5ons of sulfonyl chlorides 21.8 Reac7ons of Carboxylic Acid Deriva7ves with Nucleophiles 53
54 Reac8ons of Anhydrides with Nucleophiles 21.8 Reac7ons of Carboxylic Acid Deriva7ves with Nucleophiles 54
55 Reac8ons of Esters with Nucleophiles Transesterifica8on: 21.8 Reac7ons of Carboxylic Acid Deriva7ves with Nucleophiles 55
56 Reduc8on of Esters to Primary Alcohols 21.9 Reduc7on of Carboxylic Acid Deriva7ves 56
57 Reduc8on of Amides to Amines Note that a protonolysis step with aqueous acid follows the LiAlH 4 reduc5on This is followed by base to free the amine 21.9 Reduc7on of Carboxylic Acid Deriva7ves 57
58 Reduc8on of Amides to Amines Note how the amide reduc5on differs from that of ester reduc5on 21.9 Reduc7on of Carboxylic Acid Deriva7ves 58
59 Reduc8on of Amides to Amines 21.9 Reduc7on of Carboxylic Acid Deriva7ves 59
60 Reduc8on of Amides to Amines 21.9 Reduc7on of Carboxylic Acid Deriva7ves 60
61 Reduc8on of Nitriles to Primary Amines Two successive nucleophilic addi5ons occur 21.9 Reduc7on of Carboxylic Acid Deriva7ves 61
62 Reduc8on of Nitriles to Primary Amines 21.9 Reduc7on of Carboxylic Acid Deriva7ves 62
63 Reduc8on of Acid Chlorides to Aldehydes The Rosenmund reduc8on: 21.9 Reduc7on of Carboxylic Acid Deriva7ves 63
64 Reduc8on of Acid Chlorides to Aldehydes Use of deriva5ves of LiAlH 4 : 21.9 Reduc7on of Carboxylic Acid Deriva7ves 64
65 Rela8ve Reac8vi8es 21.9 Reduc7on of Carboxylic Acid Deriva7ves 65
66 Reac8ons of Esters with Grignard Reagents Reac7ons of Carboxylic Acid Deriva7ves with Organometallic Reagents 66
67 Reac8ons of Acid Chlorides with R 2 CuLi Dialkylcuprates are less reac5ve than Grignard reagents Reac7ons of Carboxylic Acid Deriva7ves with Organometallic Reagents 67
68 Summary of C.A. Deriva8ve Prepara8on Synthesis of esters: Acid catalyzed esterifica5on of RCO 2 H Alkyla5on of RCO 2 H or RCO 2 Acid chlorides or anhydrides with ROH Transesterifica5on Synthesis of acid chlorides: RCO 2 H with SOCl 2 or PCl Synthesis of Carboxylic Acid Deriva7ves 68
69 Summary of C.A. Deriva8ve Prepara8on Synthesis of anhydrides: RCO 2 H with dehydra5ng agents Acid chlorides with RCO 2 Synthesis of amides: Acid chlorides, anhydrides, or esters with amines Synthesis of Carboxylic Acid Deriva7ves 69
70 Summary of C.A. Deriva8ve Prepara8on Synthesis of nitriles: Cyanohydrin forma5on S N 2 reac5on of cyanide ion with alkyl halides or sulfonate esters Synthesis of Carboxylic Acid Deriva7ves 70
71 Nylon and Polyesters Nylon is a generic name given to polyamides Use and Occurrence of Carboxylic Acids and Their Deriva7ves 71
72 Nylon and Polyesters Polyesters are condensa8on polymers derived from diols and dicarboxylic acids Use and Occurrence of Carboxylic Acids and Their Deriva7ves 72
73 Waxes, Fats, and Phospholipids A wax is an ester of a fauy acid and a fauy alcohol A fat is derived form a molecule of glycerol and three molecules of fauy acid Use and Occurrence of Carboxylic Acids and Their Deriva7ves 73
74 Waxes, Fats, and Phospholipids Fats with no double bonds are called saturated fats Fats containing double bonds are called unsaturated fats Treatment of fats with KOH or NaOH gives glycerol and the salt of the fauy acid (a soap) This reac5on is the origin of the term saponifica8on Use and Occurrence of Carboxylic Acids and Their Deriva7ves 74
75 Waxes, Fats, and Phospholipids Phospholipids are also esters of glycerol The key structural difference is the presence of a polar phosphoric acid deriva5ve Use and Occurrence of Carboxylic Acids and Their Deriva7ves 75
Carboxylic Acids and Carboxylic Acid Deriva3ves. Nucleophilic Acyl Subs0tu0on (Addi0on- Elimina0on)
 Carboxylic Acids and Carboxylic Acid Deriva3ves Nucleophilic Acyl Subs0tu0on (Addi0on- Elimina0on) 1 Carboxylic Compounds Acyl group bonded to X, an electronega3ve atom or leaving group Includes: X = halide
Carboxylic Acids and Carboxylic Acid Deriva3ves Nucleophilic Acyl Subs0tu0on (Addi0on- Elimina0on) 1 Carboxylic Compounds Acyl group bonded to X, an electronega3ve atom or leaving group Includes: X = halide
Chapter 18 Carboxylic Acids and Their Derivatives. Nucleophilic Addition- Elimination at the Acyl Carbon
 Chapter 18 Carboxylic Acids and Their Derivatives. Nucleophilic Addition- Elimination at the Acyl Carbon Introduction The carboxyl group (-CO 2 H) is the parent group of a family of compounds called acyl
Chapter 18 Carboxylic Acids and Their Derivatives. Nucleophilic Addition- Elimination at the Acyl Carbon Introduction The carboxyl group (-CO 2 H) is the parent group of a family of compounds called acyl
Chapter 10. Carboxylic Acids and Derivatives. Naming Carboxylic Acids and Derivatives. Carboxylic Acids: RCOOH (RCO 2 H)
 Chapter 10 Carboxylic Acids and Derivatives Naming Carboxylic Acids and Derivatives Carboxylic Acids: RCH (RC 2 H) The functional group of a carboxylic acid is a carboxyl group (carbonyl & hydroxyl group)
Chapter 10 Carboxylic Acids and Derivatives Naming Carboxylic Acids and Derivatives Carboxylic Acids: RCH (RC 2 H) The functional group of a carboxylic acid is a carboxyl group (carbonyl & hydroxyl group)
1/3/2011. Chapter 17 Carboxylic Acids and Their Derivatives. Nucleophilic Addition- Elimination at the Acyl Carbon
 Introduction The carboxyl group (-CO 2 H) is the parent group of a family of compounds called acyl compounds or carboxylic acid derivatives Chapter 17 Carboxylic Acids and Their Derivatives. Nucleophilic
Introduction The carboxyl group (-CO 2 H) is the parent group of a family of compounds called acyl compounds or carboxylic acid derivatives Chapter 17 Carboxylic Acids and Their Derivatives. Nucleophilic
Chapter 18. Carboxylic Acids and Their Derivatives. Nucleophilic Addition-Elimination at the Acyl Carbon
 Chapter 18 Carboxylic Acids and Their Derivatives. Nucleophilic Addition-Elimination at the Acyl Carbon Carboxylic Acids Organic compounds characterized by their acidity Contains COOH group (must be at
Chapter 18 Carboxylic Acids and Their Derivatives. Nucleophilic Addition-Elimination at the Acyl Carbon Carboxylic Acids Organic compounds characterized by their acidity Contains COOH group (must be at
Carboxylic Acid Derivatives: Nucleophilic Acyl Substitution
 Carboxylic Acid Derivatives: Nucleophilic Acyl Substitution Carboxylic Acid Derivatives Carboxylic acid derivatives. Acyl chloride Acid anhydride Ester Amide Nucleophilic acyl substitution 19.1 Nomenclature
Carboxylic Acid Derivatives: Nucleophilic Acyl Substitution Carboxylic Acid Derivatives Carboxylic acid derivatives. Acyl chloride Acid anhydride Ester Amide Nucleophilic acyl substitution 19.1 Nomenclature
CARBOXYLIC ACIDS AND THEIR DERIVATIVES: NUCLEOPHILIC ADDITION-ELIMINATION AT THE ACYL CARBON
 CARBOXYLIC ACIDS AND THEIR DERIVATIVES: NUCLEOPHILIC ADDITION-ELIMINATION AT THE ACYL CARBON RED ANT WAS SOURCE OF FORMIC ACID (RCOOH) Lecture 8 ORGANIC CHEMISTRY 2 Introduction The carboxyl group (-CO
CARBOXYLIC ACIDS AND THEIR DERIVATIVES: NUCLEOPHILIC ADDITION-ELIMINATION AT THE ACYL CARBON RED ANT WAS SOURCE OF FORMIC ACID (RCOOH) Lecture 8 ORGANIC CHEMISTRY 2 Introduction The carboxyl group (-CO
Carboxylic Acid Derivatives Reading Study Problems Key Concepts and Skills Lecture Topics: Structures and reactivity of carboxylic acid derivatives
 Carboxylic Acid Derivatives Reading: Wade chapter 21, sections 21-1- 21-16 Study Problems: 21-45, 21-46, 21-48, 21-49, 21-50, 21-53, 21-56, 21-58, 21-63 Key Concepts and Skills: Interpret the spectra of
Carboxylic Acid Derivatives Reading: Wade chapter 21, sections 21-1- 21-16 Study Problems: 21-45, 21-46, 21-48, 21-49, 21-50, 21-53, 21-56, 21-58, 21-63 Key Concepts and Skills: Interpret the spectra of
Fundamentals of Organic Chemistry CHEM 109 For Students of Health Colleges Credit hrs.: (2+1)
 Fundamentals of Organic Chemistry CHEM 109 For Students of Health Colleges Credit hrs.: (2+1) King Saud University College of Science, Chemistry Department CHEM 109 CHAPTER 7. CARBOXYLIC ACIDS AND THEIR
Fundamentals of Organic Chemistry CHEM 109 For Students of Health Colleges Credit hrs.: (2+1) King Saud University College of Science, Chemistry Department CHEM 109 CHAPTER 7. CARBOXYLIC ACIDS AND THEIR
Loudon Chapter 21 Review: Carboxylic Acid Derivatives Jacquie Richardson, CU Boulder Last updated 3/20/2018
 Loudon Chapter 21 eview: Carboxylic Acid Derivatives Jacquie ichardson, CU Boulder Last updated 3/20/2018 We learned how to make a lot of carboxylic acid derivatives from acids in Ch. 20, but now we ll
Loudon Chapter 21 eview: Carboxylic Acid Derivatives Jacquie ichardson, CU Boulder Last updated 3/20/2018 We learned how to make a lot of carboxylic acid derivatives from acids in Ch. 20, but now we ll
Carboxylic Acids. The Importance of Carboxylic Acids (RCO 2 H)
 Carboxylic Acids The Importance of Carboxylic Acids (RCO 2 H) Starting materials for acyl derivatives (esters, amides, and acid chlorides) Abundant in nature from oxidation of aldehydes and alcohols in
Carboxylic Acids The Importance of Carboxylic Acids (RCO 2 H) Starting materials for acyl derivatives (esters, amides, and acid chlorides) Abundant in nature from oxidation of aldehydes and alcohols in
10. CARBOXYLIC ACIDS AND THEIR DERIVATIVES 10.1 Nomenclature of Carboxylic Acids 10.2 Physical Properties of Carboxylic Acids 10.
 BOOKS 1) Organic Chemistry Structure and Function, K. Peter C. Vollhardt, Neil Schore, 6th Edition 2) Organic Chemistry, T. W. Graham Solomons, Craig B. Fryhle 3) Organic Chemistry: A Short Course, H.
BOOKS 1) Organic Chemistry Structure and Function, K. Peter C. Vollhardt, Neil Schore, 6th Edition 2) Organic Chemistry, T. W. Graham Solomons, Craig B. Fryhle 3) Organic Chemistry: A Short Course, H.
Chapter 20: Carboxylic Acids and Nitriles شیمی آلی 2
 Chapter 20: Carboxylic Acids and Nitriles شیمی آلی 2 Dr M. Mehrdad University of Guilan, Department of Chemistry, Rasht, Iran m-mehrdad@guilan.ac.ir Based on McMurry s Organic Chemistry, 7 th edition The
Chapter 20: Carboxylic Acids and Nitriles شیمی آلی 2 Dr M. Mehrdad University of Guilan, Department of Chemistry, Rasht, Iran m-mehrdad@guilan.ac.ir Based on McMurry s Organic Chemistry, 7 th edition The
Carboxylic Acids and Nitriles. Chapters 20, 21 Organic Chemistry, 8th Edition John McMurry
 Carboxylic Acids and Nitriles Chapters 20, 21 Organic Chemistry, 8th Edition John McMurry 1 Carboxylic Acid Derivatives 2 Carboxylic Acid Derivatives nitrile R = CH 3 acetonitrile 3 Structure and Bonding
Carboxylic Acids and Nitriles Chapters 20, 21 Organic Chemistry, 8th Edition John McMurry 1 Carboxylic Acid Derivatives 2 Carboxylic Acid Derivatives nitrile R = CH 3 acetonitrile 3 Structure and Bonding
Chapter 19: Carboxylic Acid Derivatives: Nucleophilic Acyl Substitution 19.1: Nomenclature of Carboxylic Acid Derivatives (please read)
 problem 18.33b - = 128.7 123.9 179.7 146.8 147.4 45.3 18.0 161 hapter 19: arboxylic Acid Derivatives: ucleophilic Acyl Substitution 19.1: omenclature of arboxylic Acid Derivatives (please read) carboxylic
problem 18.33b - = 128.7 123.9 179.7 146.8 147.4 45.3 18.0 161 hapter 19: arboxylic Acid Derivatives: ucleophilic Acyl Substitution 19.1: omenclature of arboxylic Acid Derivatives (please read) carboxylic
Carboxylic Acid Derivatives: Nucleophilic Acyl Substitution Reactions
 Carboxylic Acid Derivatives: Nucleophilic Acyl Substitution Reactions Dr. Ayad Kareem Department of Pharmaceutical Chemistry, Collage of Pharmacy Al-Mustansiriyah University (2017-2018). Closely related
Carboxylic Acid Derivatives: Nucleophilic Acyl Substitution Reactions Dr. Ayad Kareem Department of Pharmaceutical Chemistry, Collage of Pharmacy Al-Mustansiriyah University (2017-2018). Closely related
Chapter 20: Carboxylic Acid Derivatives: Nucleophilic Acyl Substitution
 hapter 20: arboxylic Acid Derivatives: ucleophilic Acyl Substitution 20.1: omenclature of arboxylic Acid Derivatives (please read) carboxylic acid -oic acid ' ester -oate ' lactone cyclic ester l acid
hapter 20: arboxylic Acid Derivatives: ucleophilic Acyl Substitution 20.1: omenclature of arboxylic Acid Derivatives (please read) carboxylic acid -oic acid ' ester -oate ' lactone cyclic ester l acid
Esters of Carboxylic Acids These are derivatives of carboxylic acids where the hydroxyl group is replaced by an alkoxy group.
 Carboxylic acid Derivatives Carboxylic acid derivatives are described as compounds that can be converted to carboxylic acids via simple acidic or basic hydrolysis. The most important acid derivatives are
Carboxylic acid Derivatives Carboxylic acid derivatives are described as compounds that can be converted to carboxylic acids via simple acidic or basic hydrolysis. The most important acid derivatives are
Carboxylic Acids and Their Derivatives. Chapter 17. Carboxylic Acids and Their Derivatives
 Chapter 17 Carboxylic Acids and Their Derivatives Chapter 17 suggested problems: 36, 38, 40, 42, 44, 52, 54, 56, 62, 64, 66, 70 Class Notes I. Carboxylic acids (organic acids) and their derivatives A.
Chapter 17 Carboxylic Acids and Their Derivatives Chapter 17 suggested problems: 36, 38, 40, 42, 44, 52, 54, 56, 62, 64, 66, 70 Class Notes I. Carboxylic acids (organic acids) and their derivatives A.
R O R' Acid anhydride. Acid halide. Carboxylic acid. Ester O O O O. Nitrile Acyl phosphate Thioester. Amide
 Chapter 10. Carboxylic Acids and Derivatives Carboxylic acid X Acid halide ' Acid anhydride Ester ' P N 2 C N S' Amide Nitrile Acyl phosphate Thioester The common structural feature of all these compounds
Chapter 10. Carboxylic Acids and Derivatives Carboxylic acid X Acid halide ' Acid anhydride Ester ' P N 2 C N S' Amide Nitrile Acyl phosphate Thioester The common structural feature of all these compounds
Lecture 19. Nucleophilic Acyl Substitution Y - + X - Y X R C X. April 2, Chemistry 328N
 Lecture 19 Nucleophilic Acyl Substitution X Y - - Y X X - Y April 2, 2019 hemistry 328N Acid-catalyzed Esterification (also called Fischer esterification) H H 3 H H H 2 H 3 Please study the mechanism hemistry
Lecture 19 Nucleophilic Acyl Substitution X Y - - Y X X - Y April 2, 2019 hemistry 328N Acid-catalyzed Esterification (also called Fischer esterification) H H 3 H H H 2 H 3 Please study the mechanism hemistry
Alehydes, Ketones and Carboxylic Acid
 Alehydes, Ketones and Carboxylic Acid Aldehydes and Ketones: Introduction Aldedydes and ketones are organic compounds that contain carbon-oxygen doule bonds. The general formula for aldehydes is O C R
Alehydes, Ketones and Carboxylic Acid Aldehydes and Ketones: Introduction Aldedydes and ketones are organic compounds that contain carbon-oxygen doule bonds. The general formula for aldehydes is O C R
Lecture 20. Herman Emil Fischer Nobel Prize 1902 Sugars, Esters and Purines. April 4, Chemistry 328N
 Lecture 20 April 4, 2019 Herman Emil Fischer 1852-1919 Nobel Prize 1902 Sugars, Esters and Purines Acid-catalyzed Esterification (also called Fischer esterification) CH CH 3 H H H 2 CCH 3 Please study
Lecture 20 April 4, 2019 Herman Emil Fischer 1852-1919 Nobel Prize 1902 Sugars, Esters and Purines Acid-catalyzed Esterification (also called Fischer esterification) CH CH 3 H H H 2 CCH 3 Please study
Chapter 20 Carboxylic Acids. Introduction
 hapter 20 arboxylic Acids Introduction arbonyl (-=) and hydroxyl (-H) on the same carbon is carboxyl group. arboxyl group is usually written -H or 2 H. Aliphatic acids have an alkyl group bonded to -H.
hapter 20 arboxylic Acids Introduction arbonyl (-=) and hydroxyl (-H) on the same carbon is carboxyl group. arboxyl group is usually written -H or 2 H. Aliphatic acids have an alkyl group bonded to -H.
Carboxylic Acids and their Derivatives I
 2302272 Org Chem II Part I Lecture 5 Carboxylic Acids and their Derivatives I Instructor: Dr. Tanatorn Khotavivattana E-mail: tanatorn.k@chula.ac.th Recommended Textbook: Chapter 20 in Organic Chemistry,
2302272 Org Chem II Part I Lecture 5 Carboxylic Acids and their Derivatives I Instructor: Dr. Tanatorn Khotavivattana E-mail: tanatorn.k@chula.ac.th Recommended Textbook: Chapter 20 in Organic Chemistry,
Chapter 17 Carboxylic Acids, Esters, and Amides Prepared by Andrea D. Leonard University of Louisiana at Lafayette
 Chapter 17 Carboxylic Acids, Esters, and Amides Prepared by Andrea D. Leonard University of Louisiana at Lafayette Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display.
Chapter 17 Carboxylic Acids, Esters, and Amides Prepared by Andrea D. Leonard University of Louisiana at Lafayette Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display.
Carboxylic Acid Derivatives
 arboxylic Acid Derivatives The most important derivatives of carboxylic acids are l " ' ' acid halide acid anhydride an ester an amide Although not direct derivatives, nitriles, -, are related to carboxylic
arboxylic Acid Derivatives The most important derivatives of carboxylic acids are l " ' ' acid halide acid anhydride an ester an amide Although not direct derivatives, nitriles, -, are related to carboxylic
EXPERIMENT 8 (Organic Chemistry II) Carboxylic Acids Reactions and Derivatives
 EXPERIMENT 8 (rganic Chemistry II) Carboxylic Acids Reactions and Derivatives Pahlavan/Cherif Materials Medium test tubes (6) Test tube rack Beakers (50, 150, 400 ml) Ice Hot plate Graduated cylinders
EXPERIMENT 8 (rganic Chemistry II) Carboxylic Acids Reactions and Derivatives Pahlavan/Cherif Materials Medium test tubes (6) Test tube rack Beakers (50, 150, 400 ml) Ice Hot plate Graduated cylinders
13. Carboxylic Acids (text )
 2009, Department of Chemistry, The University of Western ntario 13.1 13. Carboxylic Acids (text 14.1 14.9) A. Structure and Nomenclature The carboxylic acid functional group results from the connection
2009, Department of Chemistry, The University of Western ntario 13.1 13. Carboxylic Acids (text 14.1 14.9) A. Structure and Nomenclature The carboxylic acid functional group results from the connection
Chapters 13/14: Carboxylic Acids and Carboxylic Acid Derivatives
 CHM 201 (Elements of Organic Chemistry) Dr. Virgil Lee Cal Poly Pomona Chapters 13/14: Carboxylic Acids and Carboxylic Acid Derivatives resonance stabilized OH group donates electron density to carbonyl
CHM 201 (Elements of Organic Chemistry) Dr. Virgil Lee Cal Poly Pomona Chapters 13/14: Carboxylic Acids and Carboxylic Acid Derivatives resonance stabilized OH group donates electron density to carbonyl
Physical properties: C L = L. Cl, NH 2, OCH 3, OH, OCR O O O NH 2 CH 3 N(CH 3 ) 2. Sol. in H 2 O
 Lecture Notes hem 51 S. King hapter 22 arboxylic Acids and their Derivatives: Nucleophilic Acyl Substitution I. Structure and Physical Properties: Type 2 carbonyl compounds (carboxylic acids and derivatives)
Lecture Notes hem 51 S. King hapter 22 arboxylic Acids and their Derivatives: Nucleophilic Acyl Substitution I. Structure and Physical Properties: Type 2 carbonyl compounds (carboxylic acids and derivatives)
Paper 9: ORGANIC CHEMISTRY-III (Reaction Mechanism-2) Module17: Reduction by Metal hydrides Part-II CHEMISTRY
 Subject Chemistry Paper No and Title Module No and Title Module Tag 9: ORGANIC -III (Reaction Mechanism-2) 17: Reduction by Metal hydrides Part-1I CHE_P9_M17 Table of Contents 1. Learning Outcomes 2. Introduction
Subject Chemistry Paper No and Title Module No and Title Module Tag 9: ORGANIC -III (Reaction Mechanism-2) 17: Reduction by Metal hydrides Part-1I CHE_P9_M17 Table of Contents 1. Learning Outcomes 2. Introduction
Carboxylic acid derivatives
 Carboxylic acid derivatives Nucleophilic acyl substitution reaction Among the most important reactions of carboxylic acids are those that convert the carboxyl group into other acid derivatives by a nucleophilic
Carboxylic acid derivatives Nucleophilic acyl substitution reaction Among the most important reactions of carboxylic acids are those that convert the carboxyl group into other acid derivatives by a nucleophilic
REACTIONS OF CARBOXYLIC ACID DERIVATIVES WITH NUCLEOPHILES A. Reactions of Acid Chlorides with Nucleophiles
 1016 CHAPTER 1 THE CHEMITRY F CARBXYLIC ACID DERIVATIVE 1.8 REACTI F CARBXYLIC ACID DERIVATIVE WITH UCLEPHILE ection 1.7 showed that all carboxylic acid derivatives hydrolyze to carboxylic acids. Water
1016 CHAPTER 1 THE CHEMITRY F CARBXYLIC ACID DERIVATIVE 1.8 REACTI F CARBXYLIC ACID DERIVATIVE WITH UCLEPHILE ection 1.7 showed that all carboxylic acid derivatives hydrolyze to carboxylic acids. Water
Ch 21 Carboxylic Acid Derivatives and Nu Acyl Subst n
 Ch 21 Carboxylic Acid Derivatives and Nu Acyl Subst n Acid Derivatives and their Names - Acid Halides have a Cl or Br instead of OH. Replace ic acid with yl halide, such as propionyl chloride (a common
Ch 21 Carboxylic Acid Derivatives and Nu Acyl Subst n Acid Derivatives and their Names - Acid Halides have a Cl or Br instead of OH. Replace ic acid with yl halide, such as propionyl chloride (a common
Identifying Functional Groups. (Chapter 2 in the Klein text)
 Identifying Functional Groups (Chapter 2 in the Klein text) Basic Ideas A functional group is a substructure within a molecule that will have the potential to undergo chemical change, i.e. the group has
Identifying Functional Groups (Chapter 2 in the Klein text) Basic Ideas A functional group is a substructure within a molecule that will have the potential to undergo chemical change, i.e. the group has
Ch. 21: CARBOXYLIC ACID DERIVATIVES AND NUCLEOPHILIC ACYL SUBSTITUTION REACTIONS Nomenclature of Carboxylic Acid Derivatives:
 h. 21: ABXYLI AID DEIVATIVES AND NULEPILI AYL SUBSTITUTIN EATINS Nomenclature of arboxylic Acid Derivatives: arboxylic acids "-oic acid" Examples: 3 2 Propanoic acid yclohexanecarboxylic acid 1 arboxylate
h. 21: ABXYLI AID DEIVATIVES AND NULEPILI AYL SUBSTITUTIN EATINS Nomenclature of arboxylic Acid Derivatives: arboxylic acids "-oic acid" Examples: 3 2 Propanoic acid yclohexanecarboxylic acid 1 arboxylate
Functional Derivatives of Carboxylic Acids
 Functional Derivatives of Carboxylic Acids Derivatives of Carboxylic Acids are compounds in which the OH of a carboxyl group has been replaced by CI, OOCR, NH2, or OR'to convert acid chlorides,anhydrides,
Functional Derivatives of Carboxylic Acids Derivatives of Carboxylic Acids are compounds in which the OH of a carboxyl group has been replaced by CI, OOCR, NH2, or OR'to convert acid chlorides,anhydrides,
Chemistry Chapter 19
 hemistry 2100 hapter 19 arboxyl Derivatives In this chapter, we study three classes of compounds derived from carboxylic acids; anhydrides, esters, and amides. Each is related to a carboxyl group by loss
hemistry 2100 hapter 19 arboxyl Derivatives In this chapter, we study three classes of compounds derived from carboxylic acids; anhydrides, esters, and amides. Each is related to a carboxyl group by loss
Chapter 21. Carboxylic Acid Derivatives: Nucleophilic Acyl Substitution Reactions شیمی آلی 2
 Chapter 21. Carboxylic Acid Derivatives: Nucleophilic Acyl Substitution Reactions شیمی آلی 2 Dr M. Mehrdad University of Guilan, Department of Chemistry, Rasht, Iran m-mehrdad@guilan.ac.ir Based on McMurry
Chapter 21. Carboxylic Acid Derivatives: Nucleophilic Acyl Substitution Reactions شیمی آلی 2 Dr M. Mehrdad University of Guilan, Department of Chemistry, Rasht, Iran m-mehrdad@guilan.ac.ir Based on McMurry
Carboxylic Acids and Esters
 24 Carboxylic Acids and Esters The sour tang in fruit juice comes from carboxylic acids. Introduction to General, Organic, and Biochemistry, 10e John Wiley & Sons, Inc Morris Hein, Scott Pattison, and
24 Carboxylic Acids and Esters The sour tang in fruit juice comes from carboxylic acids. Introduction to General, Organic, and Biochemistry, 10e John Wiley & Sons, Inc Morris Hein, Scott Pattison, and
DERIVATIVES OF CARBOXYLIC ACIDS
 13 Rl RH RNH 2 RR RR DERIVATIVES F ARBXYLI AIDS HAPTER SUMMARY 13.1 Structure and Nomenclature of arboxylic Acid Derivatives A. Structure arboxylic acids and their derivatives can be expressed as variations
13 Rl RH RNH 2 RR RR DERIVATIVES F ARBXYLI AIDS HAPTER SUMMARY 13.1 Structure and Nomenclature of arboxylic Acid Derivatives A. Structure arboxylic acids and their derivatives can be expressed as variations
Chapter 16 and GHW#6 Questions. Carboxylic Acids, Esters, and Other Acid Derivatives
 Chapter 16 and GHW#6 Questions Carboxylic Acids, Esters, and Other Acid Derivatives Bonding Characteristics of Carboxylic Acids A carboxylic acid has functional a carboxyl group. A carboxyl group is a
Chapter 16 and GHW#6 Questions Carboxylic Acids, Esters, and Other Acid Derivatives Bonding Characteristics of Carboxylic Acids A carboxylic acid has functional a carboxyl group. A carboxyl group is a
Chem 263 Nov 26, 2013 O R' alkyl. acid. ethyl. acetic acid. ethyl acetate ethyl ethanoate
 hem 263 ov 26, 2013 arboxylic Acids and Derivatives omenclature Esters Systematic names for esters are derived by first giving the name of the alkyl group attached to the oxygen, and then identifying the
hem 263 ov 26, 2013 arboxylic Acids and Derivatives omenclature Esters Systematic names for esters are derived by first giving the name of the alkyl group attached to the oxygen, and then identifying the
Alcohol aldehydes cetones and carboxylic acids
 Alcohol aldehydes cetones and carboxylic acids 1 Classes of organic compounds 2 Alcohols Alcohols are organic compounds containing hydroxyl (-OH) group attached to C atom. In an alcohol, -OH group replaces
Alcohol aldehydes cetones and carboxylic acids 1 Classes of organic compounds 2 Alcohols Alcohols are organic compounds containing hydroxyl (-OH) group attached to C atom. In an alcohol, -OH group replaces
Carboxylic Acids, Esters and Acyl Chlorides
 R hemistry A 432 arboxylic Acids, Esters and Acyl hlorides arboxylic Acids, Esters and Acyl hlorides arboxylic acids contain the functional group, attached to an alkyl stem. They are widely found in nature,
R hemistry A 432 arboxylic Acids, Esters and Acyl hlorides arboxylic Acids, Esters and Acyl hlorides arboxylic acids contain the functional group, attached to an alkyl stem. They are widely found in nature,
IR Spectroscopy Part II
 IR Spectroscopy Part II Carbonyl - compounds For simple aldehydes and ketones, the stretching vibration of the carbonyl group is a strong infrared absorption beetwen 1710 and 1740 cm -1. Alkyl substituents
IR Spectroscopy Part II Carbonyl - compounds For simple aldehydes and ketones, the stretching vibration of the carbonyl group is a strong infrared absorption beetwen 1710 and 1740 cm -1. Alkyl substituents
A carboxylic acid is an organic compound that contains a carboxyl group, COOH
 1.6 Carboxylic Acids, Esters and Fats Carboxylic Acids A carboxylic acid is an organic compound that contains a carboxyl group, COOH These compounds are weak acids. Citrus fruits, crabapples, rhubarb,
1.6 Carboxylic Acids, Esters and Fats Carboxylic Acids A carboxylic acid is an organic compound that contains a carboxyl group, COOH These compounds are weak acids. Citrus fruits, crabapples, rhubarb,
Chapter 15. Alcohols, Diols, and Thiols. B. Sources: there are two principal sources of simple aliphatic alcohols
 Chapter 15 Alcohols, Diols, and Thiols Chapter 15 suggested problems: 17, 19, 24, 28, 37, 39 I. Introduction A. The relevance of alcohols (from M&B: 497): "If an organic chemist were allowed to choose
Chapter 15 Alcohols, Diols, and Thiols Chapter 15 suggested problems: 17, 19, 24, 28, 37, 39 I. Introduction A. The relevance of alcohols (from M&B: 497): "If an organic chemist were allowed to choose
Chapter 21. Carboxylic Acid Derivatives. and Nucleophilic Acyl Substitution. Reactions. - many carboxylic acid derivatives are known:
 hapter 21 arboxylic Acid Derivatives and ucleophilic Acyl Substitution eactions - many carboxylic acid derivatives are known: X ' carboxylic acid acid halide (X = F, l, Br, I) acid anhydride ' 2 ester
hapter 21 arboxylic Acid Derivatives and ucleophilic Acyl Substitution eactions - many carboxylic acid derivatives are known: X ' carboxylic acid acid halide (X = F, l, Br, I) acid anhydride ' 2 ester
H O. rapidly reduces. They dissolve. because they can hydrogen bond to the water molecules.
 3.9 arboxylic Acids and Derivatives Naming arboxylic acids These have the ending oic acid but no number is necessary for the acid group as it must always be at the end of the chain. The numbering always
3.9 arboxylic Acids and Derivatives Naming arboxylic acids These have the ending oic acid but no number is necessary for the acid group as it must always be at the end of the chain. The numbering always
Carbon s unique bonding pattern arises from the hybridization of the electrons.
 Unit 8 Neptune, the 8 th planet of our solar system Organic Chemistry Organic: compound containing carbon, excluding oxides and carbonates Carbon is an allotrope, meaning it has different bonding patterns.
Unit 8 Neptune, the 8 th planet of our solar system Organic Chemistry Organic: compound containing carbon, excluding oxides and carbonates Carbon is an allotrope, meaning it has different bonding patterns.
CH 3. Lipids CHAPTER SUMMARY
 H 3 C H 3 C 15 H 3 C H Views of Cholesterol APTER SUMMARY 15.1 The Nature of can best be defined as biomolecules which are soluble to a great extent in solvents. In contrast to carbohydrates, proteins
H 3 C H 3 C 15 H 3 C H Views of Cholesterol APTER SUMMARY 15.1 The Nature of can best be defined as biomolecules which are soluble to a great extent in solvents. In contrast to carbohydrates, proteins
Carboxylic Acids. Carboxylic acid groups are always terminal groups with a carbonyl carbon also bound to a hydroxyl For example:
 Carboxylic Acids The functional group of carboxylic acids consists of a C=O with -OH bonded to the same carbon. Structure of Carboxyl Carbon is sp 2 hybridized. Bond angles are close to 120. O-H eclipsed
Carboxylic Acids The functional group of carboxylic acids consists of a C=O with -OH bonded to the same carbon. Structure of Carboxyl Carbon is sp 2 hybridized. Bond angles are close to 120. O-H eclipsed
6/9/2015. Unit 15: Organic Chemistry Lesson 15.2: Substituted Hydrocarbons & Functional Groups
 1-chloropropane 2-methylpropane 1-iodobutane Ethanoic Acid Unit 15: Organic Chemistry Lesson 15.2: Substituted Hydrocarbons & Functional Groups 43 It Ain t Just Hydrocarbons There are all sorts of organic
1-chloropropane 2-methylpropane 1-iodobutane Ethanoic Acid Unit 15: Organic Chemistry Lesson 15.2: Substituted Hydrocarbons & Functional Groups 43 It Ain t Just Hydrocarbons There are all sorts of organic
ORGANIC SYNTHESIS VIA ENOLATES
 1 ORGANIC SYNTHESIS VIA ENOLATES Aldehydes and ketones undergo nucleophilic addition reaction at the carbonyl group. Further, α-hydrogen containing compounds are acidic in nature. In addition to carbonyl
1 ORGANIC SYNTHESIS VIA ENOLATES Aldehydes and ketones undergo nucleophilic addition reaction at the carbonyl group. Further, α-hydrogen containing compounds are acidic in nature. In addition to carbonyl
Carboxylic Acids. Seminar_7
 Seminar_7 1. Nomenclature of Carboxylic Acids 2. Preparation of Carboxylic Acids 3. Reactivity 4. Soaps and Detergents 5. Fats and Oils TEST Carboxylic acides, esters and fats. Give the names Carboxylic
Seminar_7 1. Nomenclature of Carboxylic Acids 2. Preparation of Carboxylic Acids 3. Reactivity 4. Soaps and Detergents 5. Fats and Oils TEST Carboxylic acides, esters and fats. Give the names Carboxylic
Chemistry 212 Fall Semester 1996 Examination #2
 Chemistry 212 Fall Semester 1996 Examination #2 University of Missouri Columbia Prof. Rainer Glaser Wednesday, October 16, 1996 103 Schlundt Hall, 8:40-9:30 featuring Carboxylic Acids and Carboxylic Acid
Chemistry 212 Fall Semester 1996 Examination #2 University of Missouri Columbia Prof. Rainer Glaser Wednesday, October 16, 1996 103 Schlundt Hall, 8:40-9:30 featuring Carboxylic Acids and Carboxylic Acid
where R doesn t have to equal R or R
 hem 263 Nov 24, 2016 arboxylic Acids and Derivatives arboxylic acids are very important compounds in nature and serve as building blocks for preparing related derivatives such as esters and amides. The
hem 263 Nov 24, 2016 arboxylic Acids and Derivatives arboxylic acids are very important compounds in nature and serve as building blocks for preparing related derivatives such as esters and amides. The
Name the ester produced when methanol and pentanoic acid react. methyl pentanoate. Name the type of reaction used to make an ester
 1 Name the ester produced when methanol and pentanoic acid react methyl pentanoate 2 Name the type of reaction used to make an ester condensation reaction 3 Name the by-product of the reaction used to
1 Name the ester produced when methanol and pentanoic acid react methyl pentanoate 2 Name the type of reaction used to make an ester condensation reaction 3 Name the by-product of the reaction used to
Lecture Notes Chemistry Mukund P. Sibi Lecture 31 Reactions at the Alpha-Carbon of Carbonyl Compounds
 Lecture Notes hemistry 342-2008 Mukund P. Sibi eactions at the Alpha-arbon of arbonyl ompounds Enolates are nucleophilic and undergo reaction with electrophiles. For example, one can do halogenation under
Lecture Notes hemistry 342-2008 Mukund P. Sibi eactions at the Alpha-arbon of arbonyl ompounds Enolates are nucleophilic and undergo reaction with electrophiles. For example, one can do halogenation under
Chemistry B11 Chapters 14 Amines, aldehydes, ketones and carboxylic acids
 Chapters 4 Amines, aldehydes, ketones and carboxylic acids Amines: are derivatives from ammonia ( 3 ). Aliphatic amines: an amine in which nitrogen is bonded only to alkyl group or hydrogens. Aromatic
Chapters 4 Amines, aldehydes, ketones and carboxylic acids Amines: are derivatives from ammonia ( 3 ). Aliphatic amines: an amine in which nitrogen is bonded only to alkyl group or hydrogens. Aromatic
H 3 C OCH 3 3 C N(CH 3 ) 2 H 3 C H H 3 C CH 3. ketone. pk a = 9 H H. 1,3-keto ester pk a = 11
 hapter 21: Ester Enolates 21.1: Ester α ydrogens and Their pk a s. The α-protons of s are less acidic that ketones and aldehydes. Typical pk a s of carbonyl compounds (α-protons): aldehydes 17 ketones
hapter 21: Ester Enolates 21.1: Ester α ydrogens and Their pk a s. The α-protons of s are less acidic that ketones and aldehydes. Typical pk a s of carbonyl compounds (α-protons): aldehydes 17 ketones
Chapter 2 Part 3: Organic and Inorganic Compounds
 Chapter 2 Part 3: Organic and Inorganic Compounds Objectives: 1) List the major groups of inorganic chemicals common in cells. 2) Describe the functions of various types of inorganic chemicals in cells.
Chapter 2 Part 3: Organic and Inorganic Compounds Objectives: 1) List the major groups of inorganic chemicals common in cells. 2) Describe the functions of various types of inorganic chemicals in cells.
Carboxylic Acids and Their Derivatives
 arboxylic Acids and Their Derivatives Families ontaining the arbonyl Group Family Y Z Y Z aldehyde or ketone carboxylic acid or -- ester or -- acid halide or -F,-l,-Br,-I acid anhydride or amide or -N
arboxylic Acids and Their Derivatives Families ontaining the arbonyl Group Family Y Z Y Z aldehyde or ketone carboxylic acid or -- ester or -- acid halide or -F,-l,-Br,-I acid anhydride or amide or -N
Carbonyl Chemistry VI + C O C. 1pm In Geology Room 112. Exam is Monday 11am-1pm. Chemistry /06/02
 arbonyl hemistry VI Ō - + hemistry 391 11/06/02 Exam is Monday 11am-1pm 1pm In Geology Room 112 The Dibasic Acids h - My - Such - hemistry 391 11/06/02 Good- Apple- Pie- Fischer Esterification Esters can
arbonyl hemistry VI Ō - + hemistry 391 11/06/02 Exam is Monday 11am-1pm 1pm In Geology Room 112 The Dibasic Acids h - My - Such - hemistry 391 11/06/02 Good- Apple- Pie- Fischer Esterification Esters can
Chemistry 1120 Exam 1 Study Guide
 Chemistry 1120 Exam 1 Study Guide Chapter 3 3.1 a) Know that alcohols contain a hydroxy (-OH) group. Determine the IUPAC name for a given structure by determining the longest chain. b) Determine the number
Chemistry 1120 Exam 1 Study Guide Chapter 3 3.1 a) Know that alcohols contain a hydroxy (-OH) group. Determine the IUPAC name for a given structure by determining the longest chain. b) Determine the number
Patrick, An Introduction to Medicinal Chemistry 5e Chapter 1 Drugs and drug targets: an overview
 Patrick, An Introduction to dicinal hemistry 5e Answers to end-of-chapter questions 1) The ability of a molecule to cross the fatty cell membrane has little to do with its size, but more with its hydrophobic
Patrick, An Introduction to dicinal hemistry 5e Answers to end-of-chapter questions 1) The ability of a molecule to cross the fatty cell membrane has little to do with its size, but more with its hydrophobic
REVIEW IN CARBOXYLIC ACIDS AND ITS DERIVATIVES
 IASET: International Journal of Agricultural & Bio-Chemical Science (IASET: IJABS) ISSN(P): Applied; ISSN(E): Applied Vol. 1, Issue 1, Jan - Jun 2017, 49-66 IASET REVIEW IN CARBXYLIC ACIDS AND ITS DERIVATIVES
IASET: International Journal of Agricultural & Bio-Chemical Science (IASET: IJABS) ISSN(P): Applied; ISSN(E): Applied Vol. 1, Issue 1, Jan - Jun 2017, 49-66 IASET REVIEW IN CARBXYLIC ACIDS AND ITS DERIVATIVES
Chapter 24. The Chemistry of Life: Organic and Biological Chemistry. Lecture Presentation. James F. Kirby Quinnipiac University Hamden, CT
 Lecture Presentation Chapter 24 The of Life: James F. Kirby Quinnipiac University Hamden, CT Organic and Biochemistry Chapter focus: the molecules that bridge chemistry & biology Most common elements:
Lecture Presentation Chapter 24 The of Life: James F. Kirby Quinnipiac University Hamden, CT Organic and Biochemistry Chapter focus: the molecules that bridge chemistry & biology Most common elements:
Organic Chemistry. Chapter 23. Hill, Petrucci, McCreary & Perry 4 th. Ed. Alkane to Substituent Group methane CH 4 methyl CH 3
 hapter 23 rganic hemistry ill, Petrucci, Mcreary & Perry 4 th Ed. Alkane to Substituent Group methane 4 methyl 3 ethane 3 3 ethyl 3 2 propane 3 2 3 propyl 3 2 2 isopropyl ( 3 ) 2 or 3 3 butyl 3 2 2 2 butane
hapter 23 rganic hemistry ill, Petrucci, Mcreary & Perry 4 th Ed. Alkane to Substituent Group methane 4 methyl 3 ethane 3 3 ethyl 3 2 propane 3 2 3 propyl 3 2 2 isopropyl ( 3 ) 2 or 3 3 butyl 3 2 2 2 butane
ORGANIC CHEMISTRY Serkan SAYINER, DVM PhD. Assist. Prof.
 ORGANIC CHEMISTRY Serkan SAYINER, DVM PhD. Assist. Prof. Near East University, Faculty of Veterinary Medicine, Department of Biochemistry serkan.sayiner@neu.edu.tr Compounds Containing a C=O (Carbonyl)
ORGANIC CHEMISTRY Serkan SAYINER, DVM PhD. Assist. Prof. Near East University, Faculty of Veterinary Medicine, Department of Biochemistry serkan.sayiner@neu.edu.tr Compounds Containing a C=O (Carbonyl)
Revision Sheet Final Exam Term
 Revision Sheet Final Exam Term-1 2018-2019 Name: Subject: Chemistry Grade: 12 A, B, C Required Materials: Chapter: 22 Section: 1,2,3,4 (Textbook pg. 669-697) Chapter: 23 Section: 1,2 (Textbook pg. 707-715)
Revision Sheet Final Exam Term-1 2018-2019 Name: Subject: Chemistry Grade: 12 A, B, C Required Materials: Chapter: 22 Section: 1,2,3,4 (Textbook pg. 669-697) Chapter: 23 Section: 1,2 (Textbook pg. 707-715)
Esters An Introduction To Organic Chemistry Reactions
 We have made it easy for you to find a PDF Ebooks without any digging. And by having access to our ebooks online or by storing it on your computer, you have convenient answers with esters an introduction
We have made it easy for you to find a PDF Ebooks without any digging. And by having access to our ebooks online or by storing it on your computer, you have convenient answers with esters an introduction
Carboxylic Acids and Esters
 arboxylic Acids and Esters N Goalby hemrevise.org - absorption IR Spectrum for arboxylic acids Butanoic acid 1 Solubility in Water The smaller carboxylic (up to 4) acids dissolve in water in all proportions
arboxylic Acids and Esters N Goalby hemrevise.org - absorption IR Spectrum for arboxylic acids Butanoic acid 1 Solubility in Water The smaller carboxylic (up to 4) acids dissolve in water in all proportions
6. The catalytic mechanism of arylsulfatase A and its theoretical investigation
 6. The catalytic mechanism of arylsulfatase A and its theoretical investigation When the crystal structure of arylsulfatase A was solved, a remarkable structural analogy to another hydrolytic enzyme, the
6. The catalytic mechanism of arylsulfatase A and its theoretical investigation When the crystal structure of arylsulfatase A was solved, a remarkable structural analogy to another hydrolytic enzyme, the
Reading. Learning Objectives. How are macromolecules assembled? 8. Macromolecules I. Contents
 Contents 1 Reading 2 Learning Objectives 3 How are macromolecules assembled? 4 Carbohydrates 4.1 Structural Carbohydrates 5 Lipids 5.1 Fats/Triglycerides 5.1.1 Saturated versus Unsaturated fats 5.2 Phospholipids
Contents 1 Reading 2 Learning Objectives 3 How are macromolecules assembled? 4 Carbohydrates 4.1 Structural Carbohydrates 5 Lipids 5.1 Fats/Triglycerides 5.1.1 Saturated versus Unsaturated fats 5.2 Phospholipids
BIOB111_CHBIO - Tutorial activity for Session 12
 BIOB111_CHBIO - Tutorial activity for Session 12 General topic for week 6 Session 12 Lipids Useful Links: 1. Animations on Cholesterol (its synthesis, lifestyle factors, LDL) http://www.wiley.com/college/boyer/0470003790/animations/cholesterol/cholesterol.htm
BIOB111_CHBIO - Tutorial activity for Session 12 General topic for week 6 Session 12 Lipids Useful Links: 1. Animations on Cholesterol (its synthesis, lifestyle factors, LDL) http://www.wiley.com/college/boyer/0470003790/animations/cholesterol/cholesterol.htm
A. Carboxylic acid functional groups contain the carboxyl structural feature. 1. Features of the carboxyl group
 Chapter 17 Carboxylic Acids and Their Derivatives Chem 306 Roper I. Overview A. Carboxylic acid functional groups contain the carboxyl structural feature. 1. Features of the carboxyl group 2. The reactivity
Chapter 17 Carboxylic Acids and Their Derivatives Chem 306 Roper I. Overview A. Carboxylic acid functional groups contain the carboxyl structural feature. 1. Features of the carboxyl group 2. The reactivity
Alkenes. Isomerism in the alkenes
 Alkenes Alkenes are a family of hydrocarbons (compounds containing carbon and hydrogen only) containing a carbon-carbon double bond. The first two are: ethene 2 4 propene 3 6 You can work out the formula
Alkenes Alkenes are a family of hydrocarbons (compounds containing carbon and hydrogen only) containing a carbon-carbon double bond. The first two are: ethene 2 4 propene 3 6 You can work out the formula
Chemistry 304B Spring 1999 Lecture 24 1
 Chemistry 304B Spring 1999 Lecture 24 1 Exam: Monday evening, 7:30-10 pm, McCosh 50 pen book, etc. eview sessions: Today: 5-6 pm m 124 Frick Sunday: 8-9:30 pm m 324 Frick Monday lecture 9 am, m 120 Frick
Chemistry 304B Spring 1999 Lecture 24 1 Exam: Monday evening, 7:30-10 pm, McCosh 50 pen book, etc. eview sessions: Today: 5-6 pm m 124 Frick Sunday: 8-9:30 pm m 324 Frick Monday lecture 9 am, m 120 Frick
Lecture'11:'February'21,'2013 Reac&ons*of*Deriva&ves*( )
 CM'224' 'rganic'chemistry'ii Spring'2013,'Des'Plaines' 'Prof.'Chad'Landrie Vegetable il (C 2 ) 7 (C 2 ) 7 (C 2 ) 7 LL (a triacylglyceride in soybean oil) (1) transesterification ac 3 Biodiesel 3 C 3 C
CM'224' 'rganic'chemistry'ii Spring'2013,'Des'Plaines' 'Prof.'Chad'Landrie Vegetable il (C 2 ) 7 (C 2 ) 7 (C 2 ) 7 LL (a triacylglyceride in soybean oil) (1) transesterification ac 3 Biodiesel 3 C 3 C
Oxoacids of Phosphorus *
 OpenStax-CNX module: m35001 1 Oxoacids of Phosphorus * Andrew R. Barron This work is produced by OpenStax-CNX and licensed under the Creative Commons Attribution License 3.0 Phosphorous pentoxide (P 2
OpenStax-CNX module: m35001 1 Oxoacids of Phosphorus * Andrew R. Barron This work is produced by OpenStax-CNX and licensed under the Creative Commons Attribution License 3.0 Phosphorous pentoxide (P 2
Organic Chemistry Diversity of Carbon Compounds
 Organic Chemistry Diversity of Carbon Compounds Hydrocarbons The Alkanes The Alkenes The Alkynes Naming Hydrocarbons Cyclic Hydrocarbons Alkyl Groups Aromatic Hydrocarbons Naming Complex Hydrocarbons Chemical
Organic Chemistry Diversity of Carbon Compounds Hydrocarbons The Alkanes The Alkenes The Alkynes Naming Hydrocarbons Cyclic Hydrocarbons Alkyl Groups Aromatic Hydrocarbons Naming Complex Hydrocarbons Chemical
Chem 263 Nov 21, 2013
 hem 263 Nov 21, 2013 arbohydrates- emiacetal Formation You know from previous lectures that carbonyl compounds react with all kinds of nucleophiles. ydration and hemiacetal formation are typical examples.
hem 263 Nov 21, 2013 arbohydrates- emiacetal Formation You know from previous lectures that carbonyl compounds react with all kinds of nucleophiles. ydration and hemiacetal formation are typical examples.
TOPIC 4. CARBOXYLIC ACIDS AND THEIR DERIVATES: NUCLEOPHILIC ADDITION-ELIMINATION AT THE ACYL CARBON (Chapter 17)
 L TPI 4. ABXYLI AIDS AND TEI DEIVATES: NULEPILI ADDITIN-ELIMINATIN AT TE AYL ABN (hapter 17) BJETIVES 1. Name carboxylic acids and acid derivatives: acyl chlorides, anhydrides, esters, amides and nitriles
L TPI 4. ABXYLI AIDS AND TEI DEIVATES: NULEPILI ADDITIN-ELIMINATIN AT TE AYL ABN (hapter 17) BJETIVES 1. Name carboxylic acids and acid derivatives: acyl chlorides, anhydrides, esters, amides and nitriles
Nu: - Addition or Nu: - Acyl Substitution?
 12. Apply nucleophilic addition and elimination concepts to nucleophilic acyl substution reactions of acids and derivatives (focus on esters and amides) In Class problems: 1. The reactive site of aldehydes,
12. Apply nucleophilic addition and elimination concepts to nucleophilic acyl substution reactions of acids and derivatives (focus on esters and amides) In Class problems: 1. The reactive site of aldehydes,
15.1 Lipids 15.2 Fatty Acids. Copyright 2009 by Pearson Education, Inc.
 Chapter 15 Lipids 15.1 Lipids 15.2 Fatty Acids Copyright 2009 by Pearson Education, Inc. 1 Lipids Lipids are biomolecules that contain fatty acids or a steroid nucleus. soluble in organic solvents, but
Chapter 15 Lipids 15.1 Lipids 15.2 Fatty Acids Copyright 2009 by Pearson Education, Inc. 1 Lipids Lipids are biomolecules that contain fatty acids or a steroid nucleus. soluble in organic solvents, but
Chap 7: Alcohols, Phenols, & Thiols
 Chap 7: Alcohols, Phenols, & Thiols Objectives: Chap 7: Alcohols, Phenols, & Thiols (Chapter 7 and pages 283-285 & 296-297, A-1 & A-2 in lab manual) 1. Identify molecules as an alcohol, phenol, glycol,
Chap 7: Alcohols, Phenols, & Thiols Objectives: Chap 7: Alcohols, Phenols, & Thiols (Chapter 7 and pages 283-285 & 296-297, A-1 & A-2 in lab manual) 1. Identify molecules as an alcohol, phenol, glycol,
Experiment 12 Lipids. Structures of Common Fatty Acids Name Number of carbons
 Experiment 12 Lipids Lipids are a class of biological molecules that are insoluble in water and soluble in nonpolar solvents. There are many different categories of lipids and each category has different
Experiment 12 Lipids Lipids are a class of biological molecules that are insoluble in water and soluble in nonpolar solvents. There are many different categories of lipids and each category has different
Infrared Spectroscopy
 Carbonyl Compounds Cl H H N 2 1810 cm -1 (band 1) 1800 cm -1 1760 cm -1 both present (band 2) 1735 cm -1 1725 cm -1 1715 cm -1 1710 cm -1 1690 cm -1 Inductive Effects esonance Effects stronger bond W W
Carbonyl Compounds Cl H H N 2 1810 cm -1 (band 1) 1800 cm -1 1760 cm -1 both present (band 2) 1735 cm -1 1725 cm -1 1715 cm -1 1710 cm -1 1690 cm -1 Inductive Effects esonance Effects stronger bond W W
Experiment 15: Fischer Esterification and Combinatorial Chemistry Phill Rasnick Introduction
 28 November 2012 Experiment 15: Fischer Esterification and Combinatorial Chemistry Phill Rasnick Introduction Esterification reactions, which produce esters, have proven to have great importance in organic
28 November 2012 Experiment 15: Fischer Esterification and Combinatorial Chemistry Phill Rasnick Introduction Esterification reactions, which produce esters, have proven to have great importance in organic
(iii). Decarboxylation (iv)kolbe Electrolysis
 ALKANOIC ACID/CARBOXYLIC ACID Contains carboxyl functional group COOH Two functional groups are contained in carboxyl- carbonyl (C=O)and hydroxyl (-OH) Saturated aliphatic alkanoic acids have general formula
ALKANOIC ACID/CARBOXYLIC ACID Contains carboxyl functional group COOH Two functional groups are contained in carboxyl- carbonyl (C=O)and hydroxyl (-OH) Saturated aliphatic alkanoic acids have general formula
Carboxylic Acids and Derivatives. Decarboxylation R H + CO 2. R OH Reaction type: Elimination. H H Malonic acid. Mechanism:
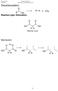 rganic hemistry arboxylic Acids and Derivatives Decarboxylation eaction type: Elimination 2 Malonic acid Mechanism: 235 rganic hemistry arboxylic Acids and Derivatives ucleophilic Acyl Substitution u Two
rganic hemistry arboxylic Acids and Derivatives Decarboxylation eaction type: Elimination 2 Malonic acid Mechanism: 235 rganic hemistry arboxylic Acids and Derivatives ucleophilic Acyl Substitution u Two
Chem 263 B6 Notes March 30, 2006 Demo-In-Class: O
 hem 263 B6 otes March 30, 2006 Demo-In-lass: + 2 carbon dioxide carbonic acid arbon dioxide ( 2 ) is a solid at -78. It is dry ice. When it is added to water, we made carbonated water (as in soda pop).
hem 263 B6 otes March 30, 2006 Demo-In-lass: + 2 carbon dioxide carbonic acid arbon dioxide ( 2 ) is a solid at -78. It is dry ice. When it is added to water, we made carbonated water (as in soda pop).
CHAPTER 3. Carbon & the Molecular Diversity of Life
 CHAPTER 3 Carbon & the Molecular Diversity of Life Carbon: The Organic Element Compounds that are synthesized by cells and contain carbon are organic So what is inorganic? Why are carbon compounds so prevalent?
CHAPTER 3 Carbon & the Molecular Diversity of Life Carbon: The Organic Element Compounds that are synthesized by cells and contain carbon are organic So what is inorganic? Why are carbon compounds so prevalent?
Chapter 3. Table of Contents. Section 1 Carbon Compounds. Section 2 Molecules of Life. Biochemistry
 Biochemistry Table of Contents Section 1 Carbon Compounds Section 2 Molecules of Life Section 1 Carbon Compounds Objectives Distinguish between organic and inorganic compounds. Explain the importance of
Biochemistry Table of Contents Section 1 Carbon Compounds Section 2 Molecules of Life Section 1 Carbon Compounds Objectives Distinguish between organic and inorganic compounds. Explain the importance of
Alcohols, Phenols, Ethers And Thiols Lec:3
 Alcohols, Phenols, Ethers And Thiols Lec:3 The word alcohol refers to a class of compounds that contain an group called a hydroxyl or hydroxyl group, bounded to an alkyl group. Alcohols can be viewed as
Alcohols, Phenols, Ethers And Thiols Lec:3 The word alcohol refers to a class of compounds that contain an group called a hydroxyl or hydroxyl group, bounded to an alkyl group. Alcohols can be viewed as
Bio 100 Serine Proteases 9/26/11
 Assigned Reading: 4th ed. 6.4.1 The Chymotrypsin Mechanism Involves Acylation And Deacylation Of A Ser Residue p. 213 BOX 20-1 Penicillin and β-lactamase p. 779 6.5.7 Some Enzymes Are Regulated By Proteolytic
Assigned Reading: 4th ed. 6.4.1 The Chymotrypsin Mechanism Involves Acylation And Deacylation Of A Ser Residue p. 213 BOX 20-1 Penicillin and β-lactamase p. 779 6.5.7 Some Enzymes Are Regulated By Proteolytic
