Biliary Sterol Secretion Is Not Required for Macrophage Reverse Cholesterol Transport
|
|
|
- George Carr
- 5 years ago
- Views:
Transcription
1 Short Article Biliary Sterol Secretion Is Not Required for Macrophage Reverse Cholesterol Transport Ryan E. Temel, 1 Janet K. Sawyer, 1 Liqing Yu, 1 Caleb Lord, 1 Chiara Degirolamo, 1 Allison McDaniel, 1 Stephanie Marshall, 1 Nanping Wang, 2 Ramesh Shah, 1 Lawrence L. Rudel, 1 and J. Mark Brown 1, * 1 Section on Lipid Sciences, Department of Pathology, Wake Forest University School of Medicine, Winston-Salem, NC 27157, USA 2 Department of Preventative Medicine, School of Medicine, China Three Gorges University, Hubei , China *Correspondence: mabrown@wfubmc.edu DOI /j.cmet SUMMARY Recent evidence suggests that the intestine may play a direct facilitative role in reverse cholesterol transport (RCT), independent of hepatobiliary secretion. In order to understand the nonbiliary pathway for RCT, we created both genetic and surgical models of biliary cholesterol insufficiency. To genetically inhibit biliary cholesterol secretion, we generated mice in which Niemann-Pick C1-Like 1 (NPC1L1) was overexpressed in the liver. Compared to controls, NPC1L1 Liver-Tg mice exhibit a >90% decrease in biliary cholesterol secretion, yet mass fecal sterol loss and macrophage RCT are normal. To surgically inhibit biliary emptying into the intestine, we have established an acute biliary diversion model. Strikingly, macrophage RCT persists in mice surgically lacking the ability to secrete bile into the intestine. Collectively, these studies demonstrate that mass fecal sterol loss and macrophage RCT can proceed in the absence of biliary sterol secretion, challenging the obligate role of bile in RCT. INTRODUCTION Nearly 40 years ago, John Glomset (Glomset, 1968) presented the seminal framework of reverse cholesterol transport (RCT), which was described as a process by which peripheral cholesterol is returned to the liver via high-density lipoproteins (HDLs) for secretion into bile and excretion through the feces. In the context of atherosclerosis, RCT is thought to involve HDL-mediated efflux of cholesterol from the arterial wall, specifically from cholesterol-laden macrophages (Wang and Rader, 2007). In the current view of macrophage RCT, HDL-mediated delivery of peripheral cholesterol to the liver directly promotes biliary and fecal excretion (Lewis and Rader, 2005). Given this model, plasma HDL levels should accurately predict both biliary sterol secretion and fecal sterol loss. However, in mice with extremely low HDL levels, biliary and fecal sterol loss is normal (Xie et al., 2009; Groen et al., 2001; Jolley et al., 1998). In addition, biliary sterol levels do not accurately predict fecal sterol loss in several mouse models of altered hepatic cholesterol metabolism (Brown et al., 2008; Plosch et al., 2002; Yu et al., 2002). Collectively, this has led us to believe that fecal cholesterol loss likely originates from two distinct excretory routes: (1) the classic hepatobiliary route, and (2) a nonbiliary liver / plasma lipoprotein / small intestine / feces route (Brown et al., 2008). From a therapeutic standpoint, exploiting the nonbiliary route for RCT is a much more attractive option, since excessive augmentation of biliary cholesterol concentrations can promote cholesterol gallstone formation in humans (Cooper, 1991). Preceding Glomset s (Glomset, 1968) initial model of RCT by 40 years, the existence of a nonbiliary pathway for fecal sterol loss was proposed first by Warren Sperry (Sperry, 1927). Sperry s experiments demonstrated that compared to intact enterohepatic circulation, chronic surgical biliary diversion paradoxically increased fecal neutral sterol loss in dogs. Nearly a half-century later, Pertsemlidis and colleagues confirmed the results of Sperry by demonstrating that biliary diversion resulted in complete loss of fecal acidic sterol output, yet fecal neutral sterol output actually increased 7-fold (Pertsemlidis et al., 1973). Based on this observation, the authors speculated that the increased output of fecal neutral steroids could be the result of transfer of plasma cholesterol across the gut wall or due to increased synthesis in the gut. Similar results have been seen under conditions of biliary diversion in rats (Bandsma et al., 1998) and in patients with familial hypercholesterolemia (Deckelbaum et al., 1977). Actually, as early as 1959, it was suggested that nondietary fecal sterol loss in humans consists of two distinct fractions: (1) the traditional fraction coming from hepatobiliary secretion, and (2) an elusive fraction directly secreted by the intestine (Cheng and Stanley, 1959). Unfortunately, many of these early observations have been largely ignored, and the theory that fecal sterol loss derives solely from a biliary origin has become well accepted. However, strong evidence for nonbiliary fecal sterol loss continues to come to light in the age of genetically modified mice. In a powerful example of nonbiliary fecal sterol loss in mice (Kruit et al., 2005), it was shown that mice lacking the cannicular phospholipid transporter Mdr2, which secondarily have no biliary cholesterol secretion, have normal fecal sterol loss. Furthermore, activation of the liver X receptor (LXR) in Mdr2 / mice resulted in large increases in fecal sterol output, supporting the presence of an LXR-inducible nonbiliary pathway for fecal sterol loss (Kruit et al., 2005). Several studies from this same group have subsequently demonstrated the existence of a nuclear hormone receptor-inducible pathway for nonbiliary fecal sterol loss using pioneering approaches such as in situ intestinal perfusion and stable isotope methodologies (van der Veen et al., 2009; Vrins et al., 2009; van der Velde et al., 2007, 2008). However, the relative contribution of biliary and nonbiliary 96 Cell Metabolism 12, , July 7, 2010 ª2010 Elsevier Inc.
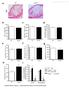
2 pathways to the specific process of macrophage RCT is not currently known. Therefore, the purpose of these studies was to determine the direct role of the intestine in macrophage RCT by using novel genetic and surgical mouse models where bile contributes minimally to fecal sterol loss. RESULTS NPC1L1- LiverTg Mice Serve as an Excellent Model to Study Nonbiliary Fecal Sterol Loss In order to characterize the role of Niemann-Pick C1-Like 1 (NPC1L1) in hepatobiliary sterol secretion and ezetimibe action, our group previously generated two independent lines of mice transgenically overexpressing NPC1L1 in hepatocytes (Temel et al., 2007). As previously reported, NPC1L1 overexpression in the liver results in a >90% reduction in gall bladder bile cholesterol concentrations (Figure 1A), without affecting biliary phospholipid or bile acid secretion (data not shown; Temel et al., 2007). However, in the face of severely diminished biliary sterol loss, fecal neutral sterol excretion in NPC1L1- LiverTg mice is normal (Figure 1B). This disparity indicates that the nonbiliary pathway must completely compensate for biliary sterol insufficiency in NPC1L1- LiverTg mice to maintain normal levels of fecal sterol loss. It is important to note that fractional cholesterol absorption (Figure 1C) and the mrna expression of cholesterol synthetic genes (see Figure S1A available online) in the liver and intestine were normal in NPC1L1- LiverTg mice. Macrophage RCT Persists in Genetically Modified Mice with Disrupted Biliary Cholesterol Secretion (NPC1L1- LiverTg ) In the current view of macrophage RCT, macrophage-derived cholesterol is delivered to the liver by HDL, which shunts this sterol cargo preferentially into the bile for subsequent fecal excretion (Lewis and Rader, 2005; degoma et al., 2008). We set out to test the specific role of bile in this framework using mice genetically lacking the ability to secrete cholesterol into bile (NPC1L1- LiverTg ). In these studies, mice were treated with the LXR agonist T , since it is known that LXR activation promotes fecal sterol loss and macrophage RCT (Naik et al., 2006) primarily by augmenting the nonbiliary pathway (Kruit et al., 2005; van der Veen et al., 2009). We have previously seen that NPC1L1- LiverTg mice are mildly hypercholesterolemic when fed synthetic diets (Temel et al., 2007). However, when young mice (6 8 weeks old) are maintained on standard rodent chow, NPC1L1- LiverTg mice have similar plasma cholesterol levels as those seen in WT littermates (Figure 2A), and the majority of cholesterol is found in HDL (Figure 2B). When treated with the LXR agonist T , both WT and NPC1L1- LiverTg mice exhibit mild hypercholesterolemia (164 and 166 mg/dl, respectively), with the majority of the cholesterol elevation seen in large HDL particles (Figure 2B), as has been previously described (Grefhorst et al., 2002). Following [ 3 H]-cholesterollabeled macrophage injection, the plasma [ 3 H]-cholesterol recovery was significantly higher in T treated mice (Figure 2C). As expected, the [ 3 H]-cholesterol distribution in plasma (Figure 2D) tracked closely with the cholesterol mass distribution (Figure 2B). Importantly, in WT mice, there was LXR-inducible biliary [ 3 H]-cholesterol secretion (Figure 2E), Figure 1. Mice Genetically Lacking the Ability to Secrete Cholesterol into Bile Have Normal Fecal Sterol Loss Wild-type mice (WT) or littermates with moderate (NPC1L1-Tg-Mod) or high-level (NPC1L1-Tg-High) overexpression of hepatic NPC1L1 were fed diets containing 0.015% or 0.2% cholesterol (wt/wt) for 6 weeks. (A) The concentration of cholesterol in gall bladder bile was determined by gas liquid chromatography. (B) Fecal neutral sterol excretion was determined by gas liquid chromatography. (C) Fractional cholesterol absorption was determined using the dual fecal isotope method. Data represent the means ± SEM from four to seven mice per group, **, significantly different than WT within each diet group, p < whereas NPC1L1- LiverTg mice had barely detectable biliary [ 3 H]-cholesterol levels (Figure 2E). However, [ 3 H]-bile acid secretion into bile was not different between WT and NPC1L1- LiverTg mice in the absence or presence of LXR agonist (Figure 2F), which coincides with mass measurements (Temel et al., 2007; data not shown). However, there was no difference in [ 3 H]- cholesterol recovery in the liver between WT and NPC1L1- LiverTg mice on either treatment (Figure 2G). In stark contrast to the biliary [ 3 H]-cholesterol recovery (Figure 2E), fecal [ 3 H]-cholesterol recovery in WT and NPC1L1- LiverTg was identical following treatment with vehicle or the LXR agonist (Figure 2H). As previously reported (Naik et al., 2006), LXR activation did increase fecal [ 3 H]-cholesterol recovery, but it did so to the same extent in mice with normal enterohepatic circulation (WT) and those Cell Metabolism 12, , July 7, 2010 ª2010 Elsevier Inc. 97
3 Figure 2. Macrophage RCT Is Normal in Mice Genetically Engineered to Lack the Ability to Secrete Cholesterol into Bile Wild-type mice (- in bar graphs) or NPC1L1- LiverTg (, in bar graphs, L1-Tg) littermates were maintained on a standard chow diet in the absence (Vehicle) or presence of the LXR agonist T (25 mg/kg per day) for 7 days. During the last 48 hr, mice were singly housed, and macrophage to feces RCT was measured as described in the Experimental Procedures. (A) Total plasma cholesterol (TPC) levels. (B) Mass cholesterol distribution of pooled plasma (n = 4 per pool). (C) Time course of [ 3 H]-cholesterol recovery in plasma; *, significantly different than WT vehicle group within each time point, p < (D) [ 3 H]-cholesterol distribution of pooled plasma (n = 4 per pool). (E) [ 3 H]-cholesterol recovery in newly secreted bile. (F) [ 3 H]-bile acid recovery in newly secreted bile. (G) [ 3 H]-cholesterol recovery in the liver. (H) [ 3 H]-cholesterol recovery in the feces. (I) [ 3 H]-bile acids recovery in the feces. (J) [ 3 H]-cholesterol recovery in the SI wall. (K) Mass biliary cholesterol concentrations in newly secreted bile. (L) Mass fecal neutral sterol loss. Results are combined from two independent experiments. Data in (A), (C), (E), (F), (G), (H), (I), and (J) represent the means ± SEM from seven to eleven mice per group, and data in (K) and (L) represent means ± SEM from five to nine mice per group. Means not sharing a common superscript differ significantly, p < genetically lacking the ability to secrete cholesterol into bile (NPC1L1- LiverTg )(Figure 2H). This effect seems to be specific to neutral sterol loss, since fecal [ 3 H]-bile acid recovery was not significantly different between any of the groups (Figure 2I). Interestingly, the [ 3 H]-cholesterol tracer recovery in the small intestinal (SI) wall (Figure 2J) was as high as levels seen in the liver (3% 5% of the dose; Figure 2G). The recovery in the SI wall was not significantly different between WT and NPC1L1- LiverTg mice but was modestly increased by LXR activation (Figure 2J). When examining cholesterol mass, vehicletreated NPC1L1- LiverTg mice compared to WT mice had a 78% reduction in cholesterol concentration in bile (Figure 2K), yet fecal neutral sterol loss was similar between the two genotypes (Figure 2L). LXR activation did result in significant increases in biliary cholesterol in both genotypes (Figure 2K), but the levels seen in NPC1L1- LiverTg mice only reached that of vehicle-treated WT mice (Figure 2K). In contrast, fecal neutral sterol loss in T treated WT and NPC1L1- LiverTg mice was significantly higher than that of vehicle-treated counterparts (Figure 2L), further supporting the disconnect between biliary sterol secretion and fecal neutral sterol loss in these mice. Surgical Biliary Diversion Reveals the Existence of a Nonbiliary Pathway for Macrophage RCT Although NPC1L1- LiverTg mice serve as an excellent model where nonbiliary sterol loss predominates, there is a small residual amount of biliary sterol loss in this genetic model (Figures 1A and 2K). In order to more definitively test whether bile is required for macrophage RCT, we set out to create a surgical model where there would be no biliary emptying into the small intestine, without obstructing bile flow. To do this, we surgically diverted bile flow away from the small intestine (Figure 3A). Once this biliary diversion, or an appropriate sham surgery, was complete, mice were allowed to recover from anesthesia and received a[ 3 H]-cholesterol-labeled macrophage foam cell dose via an externalized port in order to avoid accidental injection directly into the intestine. Quite strikingly, the appearance of [ 3 H]-cholesterol in the intestinal lumen was no different between shamoperated and surgically diverted mice (Figure 3B). Furthermore, LXR activation resulted in significant increases in the lumenal appearance of [ 3 H]-cholesterol in both sham and diverted animals (Figure 3B), further supporting the notion that LXR activation promotes primarily the nonbiliary pathway for fecal sterol loss (Kruit et al., 2005; van der Veen et al., 2009). Importantly, the lumenal appearance of [ 3 H]-bile acids was completely blocked by surgical biliary diversion (Figure 3C), which agrees with previous mass data in dogs (Sperry, 1927; Pertsemlidis et al., 1973). These data support the notion that bile is the only route for hepatic bile acids to reach the SI lumen, while cholesterol delivery into the intestine and feces can persist in the absence of biliary return (Sperry, 1927; Pertsemlidis et al., 1973; Dietschy, 1968; Figure 3). Interestingly, at this early 8 hr time point after 98 Cell Metabolism 12, , July 7, 2010 ª2010 Elsevier Inc.
4 Figure 3. Surgical Biliary Diversion Reveals the Existence of a Nonbiliary Pathway for Macrophage RCT Wild-type C57BL/6N mice were maintained on a standard chow diet in the absence (Vehicle) or presence of the LXR agonist T (25 mg/kg per day) for 7 days. (A) The experimental design is as follows: following 7 days of vehicle or LXR agonist treatment, mice were either sham operated or underwent complete surgical bile diversion as described in the Experimental Procedures. Thereafter, mice received a [ 3 H]-cholesterol-labeled macrophage (MF) dose via an externalized peritoneal port in order to ensure no dose was accidentally injected into the intestine. After 8 hr, the SI lumenal contents and wall were collected and extracted to separate [ 3 H]-cholesterol and [ 3 H]-bile acids. (B) [ 3 H]-cholesterol recovered in the SI lumenal contents. (C) [ 3 H]-bile acids recovered in the SI lumenal contents. (D) [ 3 H]-cholesterol recovered in the SI wall. Data represent the means ± SEM from five to nine mice per group, and means not sharing a common superscript differ significantly, p < [ 3 H]-cholesterol-labeled macrophage injection, 2% 4% of the dose can be found in the SI wall (Figure 3D). There was no difference in the SI wall recovery between sham and diverted animals, yet LXR activation did significantly increase [ 3 H]-cholesterol recovery in the SI wall under conditions of intact and diverted enterohepatic circulation (Figure 3D). Collectively, these data support the notion that macrophage RCT can persist in the complete absence of biliary contributions. DISCUSSION Although it is generally accepted that macrophage RCT depends entirely on the ability of the liver to secrete HDL-delivered cholesterol into bile (Lewis and Rader, 2005), results from this study suggest that the current conceptual framework of macrophage RCT requires significant modification. The major findings of the current study are the following: (1) mice genetically lacking the ability to adequately secrete cholesterol into bile (NPC1L1- LiverTg mice) have normal LXR-inducible macrophage RCT, and (2) mice surgically lacking biliary contributions to the intestine have normal LXR-inducible macrophage RCT. Importantly, the genetic and surgical models defined in this work should continue to serve as useful tools to further interrogate the molecular mechanisms mediating nonbiliary fecal sterol loss and macrophage RCT. The relative contribution of biliary versus nonbiliary pathways to fecal sterol loss has not been fully defined. Using an intestinal perfusion system, it was estimated that 44% of total fecal sterol output originated from nonbiliary sources in humans (Simmonds et al., 1967). In normal chow-fed C57BL/6J mice, the nonbiliary route accounts for 33% (76 mmol/kg/day) of total fecal neutral sterol loss (van der Veen et al., 2009) and roughly 20% of total fecal sterol loss in FVB mice (Kruit et al., 2005). However, it is important to point out that nonbiliary fecal sterol loss is quite sensitive to pharmacological manipulation. To this end, LXR activation can dramatically augment nonbiliary macrophage RCT (Figures 2 and 3) and increase mass fecal neutral sterol loss (Kruit et al., 2005; van der Veen et al., 2009). In fact, LXR activation in C57BL/6J increases the contribution of nonbiliary fecal sterol loss from 33% of the total (76 mmol/kg/day) in vehicle-treated mice to 63% of the total (442 mmol/kg/day) in T treated mice (van der Veen et al., 2009). Furthermore, activation of the peroxisome proliferator-activated receptor d (PPAR-d) promotes nonbiliary fecal sterol loss in mice (Vrins et al., 2009). Importantly, now in three independent genetically modified mouse models (ABCG5/G8 /, Mdr2 /, and NPC1L1- LiverTg ) that have severely diminished biliary cholesterol secretion, fecal sterol loss is only modestly decreased (Yu et al., 2002) or not altered at all (Kruit et al., 2005; Figure 1). This clearly indicates that the nonbiliary pathway must be able to adequately compensate for biliary insufficiency to maintain normal fecal sterol loss in rodents. Collectively, these data support the idea that fecal sterol loss is a mixture of dietary, biliary, and intestinally derived sterols, and the origins of the latter source likely originate from the plasma compartment (Brown et al., 2008; Kruit et al., 2005; van der Veen et al., 2009). Given the plasma source of intestinally derived fecal sterols, and the central role of the liver in lipoprotein metabolism, one must consider the liver as a potential site of organization for nonbiliary fecal sterol loss. The classic view of RCT involves the delivery of peripheral cholesterol via HDL to the liver for secretion into bile (Lewis and Rader, 2005; degoma et al., 2008). In parallel, we believe that the liver also plays a gatekeeper role for nonbiliary fecal sterol loss by repackaging peripheral cholesterol into nascent plasma lipoproteins that are destined for subsequent intestinal delivery (Brown et al., 2008). Probably not coincidentally, all of the mouse models described in which nonbiliary fecal sterol loss is apparent (Figure 1; Brown et al., 2008; Kruit et al., 2005; Temel et al., 2007) represent conditions where free cholesterol Cell Metabolism 12, , July 7, 2010 ª2010 Elsevier Inc. 99
5 could potentially accumulate in the liver due to defects in normal elimination pathways. It remains possible that under conditions where hepatic free cholesterol burden becomes too excessive for disposal through esterification or biliary secretion (i.e., ACAT2 ASO treatment [Brown et al., 2008], NPC1L1- Liver-Tg mice [Figure 1; Temel et al., 2007], or Mdr2 / mice [Kruit et al., 2005]), an alternative plasma-based route for direct intestinal secretion and fecal disposal is utilized. In previous work, we were able to show that the liver can secrete lipoprotein particles that preferentially deliver cholesterol to the proximal small intestine for fecal excretion (Brown et al., 2008). However, whether these liver-derived lipoproteins represent nascent VLDL particles, nascent HDL particles, or some novel lipoprotein remains to be addressed. Interestingly, unlike the genetic models of biliary cholesterol insufficiency (ABCG5/G8 /, Mdr2 /, and NPC1L1- LiverTg ), acute surgical diversion of bile is a situation in which normal elimination pathways such as esterification or biliary secretion are not obviously impaired, yet de novo cholesterol synthesis is augmented (Figure S1; Dietschy, 1968; Deckelbaum et al., 1977; Pertsemlidis et al., 1973; Bandsma et al., 1998). In this case it has been assumed that the origin of fecal sterols in bile-diverted animals comes from the enhanced de novo synthesis (Dietschy, 1968; Deckelbaum et al., 1977; Pertsemlidis et al., 1973; Bandsma et al., 1998). However, our data suggest that a small portion of the fecal sterols present in bile-diverted mice is derived from direct intestinal secretion of macrophage-derived cholesterol. This is supported by the fact that we have added an intact [ 3 H]-cholesterol molecule delivered via J774 macrophages, excluding any contribution from endogenous synthesis. Although macrophage RCT is agreed to be key to the regression of atherosclerosis, it is important to discuss the quantitative importance of macrophages in RCT and centripetal flux of cholesterol into the feces. To put macrophage RCT in perspective to the total process of centripetal cholesterol flux, it has been estimated that there are only macrophages in the adult mouse (Lee et al., 1985). Hence, the number of macrophages in the whole body represents only a small fraction of the other cell types present, making the relative contribution of macrophages to mass fecal neutral sterol loss extremely small. Even though macrophages are a minor contributor to mass fecal neutral sterol loss, macrophage-specific RCT is likely highly relevant to atherosclerosis regression in the artery wall (Lewis and Rader, 2005). Therefore, it is also important to discuss whether the macrophage RCT assay used in this work truly recapitulates the process thought to occur in the artery wall. Since the original description (Zhang et al., 2003) of the assay used in our studies, there have been over 30 independent studies described in the literature utilizing this method. Collectively, these studies indicate that once the [ 3 H]-cholesterol-labeled J774 macrophages are injected, these cells remain primarily within the peritoneal cavity, and the appearance of [ 3 H]-cholesterol in the plasma and tissues is not likely due to the direct transport of the tracer by intact J774 cells (Zhang et al., 2003; Naik et al., 2006; Wang et al., 2007; Rader et al., 2009). Instead, the bulk of evidence suggests that a small fraction of [ 3 H]-cholesterol tracer is removed from the J774 macrophages by traditional efflux mechanisms, and once effluxed this tracer is metabolized in a manner comparable to endogenous cholesterol mass (Zhang et al., 2003; Naik et al., 2006; Wang et al., 2007; Rader et al., 2009). It is important to note that the seemingly modest fecal recovery (1% 4%) of injected [ 3 H]-cholesterol (Figure 2H) may actually be substantial, given that only 1% 5% was found in the plasma over the 48 hr period (Figure 2C). Whether this assay truly recapitulates the process of macrophage RCT from the artery wall is still a matter of debate, but there is good evidence that determining the rate of macrophage RCT using this assay accurately predicts atherosclerosis burden in mice (Rader et al., 2009). However, given the assay limitations, and the relatively small contributions of macrophages to fecal sterol loss, it remains critically important for investigators to quantify mass fecal sterol loss to more accurately determine wholebody centripetal cholesterol flux in future studies. It has been over 80 years since Warren Sperry speculated that a nonbiliary route for fecal neutral sterol loss must exist in dogs (Sperry, 1927). Unfortunately, this alternative pathway has been largely ignored, and very little progress has been made to identify the molecular mechanisms that define it. There is now strong evidence in mice (Figures 1 3; Kruit et al., 2005; van der Velde et al., 2007, 2008; van der Veen et al., 2009; Vrins et al., 2009; Brown et al., 2008; Plosch et al., 2002), and mounting evidence in man (Simmonds et al., 1967; Cheng and Stanley, 1959; Deckelbaum et al., 1977), that a nonbiliary pathway for fecal sterol loss indeed exists. In order to more fully understand this pathway, the following challenges will need to be addressed: (1) establishing quantitative methods to measure nonbiliary fecal sterol loss in primates and man, (2) identifying the intestinal transport proteins/receptors involved, (3) characterizing the lipoprotein metabolism that is requisite for nonbiliary fecal sterol loss, and (4) identifying bona fide drug targets to specifically modulate the intestinal component of RCT. Advancement in these areas has the potential to open new therapeutic opportunities targeting the intestine as an inducible sterol excretory organ. EXPERIMENTAL PROCEDURES In Vivo Macrophage RCT Studies in NPC1L1- LiverTg Mice In vivo measurement of macrophage RCT was conducted essentially as described by Rader and colleagues (Zhang et al., 2003; Naik et al., 2006), with minor modifications. Extensive descriptions of the mouse models used, cell culture protocols, and RCT method are included in the Supplemental Information. In Vivo Macrophage RCT Studies with Acute Surgical Biliary Diversion C57BL/6N mice maintained on standard rodent chow were gavaged with either vehicle or 25 mg/kg T daily for 7 consecutive days prior to surgical biliary diversion. The surgical procedure used to acutely divert biliary emptying into the intestine and macrophage RCT protocol is included in the Supplemental Information. Plasma Lipoprotein Distribution Analysis Detailed description of plasma lipid analyses is included in the Supplemental Information. Quantitative Real-Time PCR RNA extraction and quantitative real-time PCR (qpcr) were conducted as previously described (Brown et al., 2008) using the DD-CT method. Primers used for qpcr are available upon request. 100 Cell Metabolism 12, , July 7, 2010 ª2010 Elsevier Inc.
6 Statistical Analysis Data are expressed as the mean ± standard error of the mean (SEM). All data were analyzed using two-way analysis of variance (ANOVA) followed by Student s t tests for post hoc analysis. Differences were considered significant at p < All analyses were performed using JMP version (SAS Institute; Cary, NC) software. SUPPLEMENTAL INFORMATION Supplemental Information includes Supplemental Experimental Procedures and one figure and can be found with this article online at doi: / j.cmet ACKNOWLEDGMENTS We sincerely thank Paul Dawson (Wake Forest University) for critical reading of this manuscript and providing meaningful input into these studies. We also thank George Rothblat (The Children s Hospital of Philadelphia) for providing J774 macrophages. This work was supported by the National Heart, Lung, and Blood Institute through a pathway to independence grant (1K99- HL to J.M.B.) and a program project grant (5P01HL to L.L.R.). L.Y. is supported by a Scientist Development Grant (# N) from the American Heart Association. L.L.R. is a member of the Merck speaker s bureau. Received: January 7, 2010 Revised: April 7, 2010 Accepted: May 13, 2010 Published: July 6, 2010 REFERENCES Bandsma, R.H.J., Stellaard, F., Vonk, R.J., Nagel, G.T., Neese, R.A., Hellerstein, M.K., and Kuipers, F. (1998). Contribution of newly synthesized cholesterol to rat plasma and bile determined by mass isotopomer distribution analysis: bile-salt flux promotes secretion of newly synthesized cholesterol into bile. Biochem. J. 329, Brown, J.M., Bell, T.A., III, Alger, H.M., Sawyer, J.K., Smith, T.L., Kelley, K., Shah, R., Wilson, M.D., Davis, M.A., Lee, R.G., et al. (2008). Targeted depletion of hepatic ACAT2-driven cholesterol esterification reveals a nonbiliary route for fecal neutral sterol loss. J. Biol. Chem. 283, Cheng, S.H., and Stanley, M.M. (1959). Secretion of cholesterol by intestinal mucosa in patients with complete common bile duct obstruction. Proc. Soc. Exp. Biol. Med. 101, Cooper, A.D. (1991). Metabolic basis of cholesterol gallstone disease. Gastroenterol. Clin. North Am. 20, Deckelbaum, R.J., Lee, R.S., Small, D.M., Hedberg, S.E., and Grundy, S.M. (1977). Failure of complete bile diversion and oral bile acid therapy in the treatment of homozygous familial hypercholesterolemia. N. Engl. J. Med. 296, degoma, E.M., degoma, R.L., and Rader, D.J. (2008). Beyond high-density lipoprotein cholesterol levels evaluating high-density lipoprotein function as influenced by novel therapeutic approaches. J. Am. Coll. Cardiol. 51, Dietschy, J.M. (1968). The role of bile salts in controlling the rate of intestinal cholesterogenesis. J. Clin. Invest. 47, Glomset, J.A. (1968). The plasma lecithin:cholesterol acyltransferase reaction. J. Lipid Res. 9, Grefhorst, A., Elzinga, B.M., Voshol, P.J., Plosch, T., Kok, T., Bloks, V.W., van der Sluijs, F.H., Havekes, L.M., Romijn, J.A., Verkade, H.J., and Kuipers, F. (2002). Stimulation of lipogenesis by pharmacological activation of the liver X receptor leads to production of large, triglyceride-rich very low density lipoprotein particles. J. Biol. Chem. 277, Groen, A.K., Bloks, V.W., Bandsma, R.H., Ottenhoff, R., Chimini, G., and Kuipers, F. (2001). Hepatobiliary cholesterol transport is not impaired in Abca1-null mice lacking HDL. J. Clin. Invest. 108, Jolley, C.D., Woollett, L.A., Turley, S.D., and Dietschy, J.M. (1998). Centripetal cholesterol flux to the liver is dictated by events in the peripheral organs and not by the plasma high density lipoprotein or apolipoprotein A-I concentration. J. Lipid Res. 39, Kruit, J.K., Plosch, T., Havinga, R., Boverhof, R., Groot, P.H., Groen, A.K., and Kuipers, F. (2005). Increased fecal neutral sterol loss upon liver X receptor activation is independent of biliary sterol secretion in mice. Gastroenterology 128, Lee, S.H., Starkey, P.M., and Gordon, S. (1985). Quantitative analysis of total macrophage content in adult mouse tissues. Immunohistochemical studies with monoclonal antibody F4/80. J. Exp. Med. 161, Lewis, G.F., and Rader, D.J. (2005). New insights into the regulation of HDL metabolism and reverse cholesterol transport. Circ. Res. 96, Naik, S.U., Wang, X., Da Silva, J.S., Jaye, M., Macphee, C.H., Reilly, M.P., Billheimer, J.T., Rothblat, G.H., and Rader, D.J. (2006). Pharmacological activation of liver X receptors promotes reverse cholesterol transport in vivo. Circulation 113, Pertsemlidis, D., Kirchman, E.H., and Ahrens, E.H., Jr. (1973). Regulation of cholesterol metabolism in the dog I. Effects of complete bile diversion and of cholesterol feeding on absorption, synthesis, accumulation, and excretion rates measured during life. J. Clin. Invest. 52, Plosch, T., Kok, T., Bloks, V.W., Smit, M.J., Havinga, R., Chimini, G., Groen, A.K., and Kuipers, F. (2002). Increased heptobiliary and fecal cholesterol excretion upon activation of the liver X receptor is independent of ABCA1. J. Biol. Chem. 277, Rader, D.J., Alexander, E.T., Weibel, G.L., Billheimer, J., and Rothblat, G.H. (2009). The role of reverse cholesterol transport in animals and humans and relationship to atherosclerosis. J. Lipid Res. 50 (Suppl.), S189 S194. Simmonds, W.J., Hofmann, A.F., and Theodor, E. (1967). Absorption of cholesterol from a micellar solution: intestinal perfusion studies in man. J. Clin. Invest. 46, Sperry, W.M. (1927). Lipid Excretion IV. A study of the relationship of the bile to the fecal lipids with special reference to certain problems of sterol metabolism. J. Biol. Chem. 71, Temel, R.E., Tang, W., Ma, Y., Rudel, L.L., Willingham, M.C., Ioannou, Y.A., Davies, J.P., Nilsson, L.M., and Yu, L. (2007). Hepatic Niemann-Pick C1-like 1 regulates biliary cholesterol concentrations and is a target of ezetimibe. J. Clin. Invest. 117, van der Velde, A.E., Vrins, C.L., van den Oever, K., Kunne, C., Oude Elferink, R.P., Kuipers, F., and Groen, A.K. (2007). Direct intestinal cholesterol secretion contributes significantly to total fecal neutral sterol excretion in mice. Gastroenterology 133, van der Velde, A.E., Vrins, C.L., Van den Oever, K., Seemann, I., Oude Elferink, R.P., Van Eck, M., Kuipers, F., and Groen, A.K. (2008). Regulation of direct transintestinal cholesterol excretion in mice. Am. J. Physiol. Gastrointest. Liver Physiol. 295, G203 G208. van der Veen, J.N., Van Dijk, T.H., Vrins, C.L., Van Meer, H., Havinga, R., Bijsterveld, K., Tietge, U.J., Groen, A.K., and Kuipers, F. (2009). Activation of the liver X receptor stimulates trans-intestinal excretion of plasma cholesterol. J. Biol. Chem. 284, Vrins, C.L., Van der Velde, A.E., Van den Oever, K., Levels, J.H., Huet, S., Oude Elferink, R.P., Kuipers, F., and Groen, A.K. (2009). PPARd activation leads to increased trans intestinal cholesterol efflux. J. Lipid Res. Published online May 13, /jlr.M JLR200. Wang, X., and Rader, D.J. (2007). Molecular regulation of macrophage reverse cholesterol transport. Curr. Opin. Cardiol. 22, Wang, X., Collins, H.L., Ranalletta, M., Fuki, I.V., Billheimer, J.T., Rothblat, G.H., Tall, A.R., and Rader, D.J. (2007). Macrophage ABCA1 and ABCG1, but not SR-BI, promote macrophage reverse cholesterol transport in vivo. J. Clin. Invest. 117, Cell Metabolism 12, , July 7, 2010 ª2010 Elsevier Inc. 101
7 Xie, C., Turley, S.D., and Dietschy, J.M. (2009). ABCA1 plays no role in the centripetal movement of cholesterol from peripheral tissues to the liver and intestine in the mouse. J. Lipid Res. 50, Yu, L., Hammer, R.E., Li-Hawkins, J., Von Bergmann, K., Lutjohann, D., Cohen, J.C., and Hobbs, H.H. (2002). Disruption of Abcg5 and Abcg8 in mice reveals their crucial role in biliary cholesterol secretion. Proc. Natl. Acad. Sci. USA 99, Zhang, Y., Zanotti, I., Reilly, M.P., Glick, J.M., Rothblat, G.H., and Rader, D.J. (2003). Overexpression of apolipoprotein A-I promotes reverse transport of cholesterol from macrophages to feces in vivo. Circulation 108, Cell Metabolism 12, , July 7, 2010 ª2010 Elsevier Inc.
* This work was supported by Grant from the Netherlands Organization for Scientific Research. S
 THE JOURNAL OF BIOLOGICAL CHEMISTRY VOL. 284, NO. 29, pp. 19211 19219, July 17, 2009 2009 by The American Society for Biochemistry and Molecular Biology, Inc. Printed in the U.S.A. Activation of the Liver
THE JOURNAL OF BIOLOGICAL CHEMISTRY VOL. 284, NO. 29, pp. 19211 19219, July 17, 2009 2009 by The American Society for Biochemistry and Molecular Biology, Inc. Printed in the U.S.A. Activation of the Liver
Biliary Sterol Secretion Is Required for Functional In Vivo Reverse Cholesterol Transport in Mice
 GASTROENTEROLOGY 2011;140:1043 1051 Biliary Sterol Secretion Is Required for Functional In Vivo Reverse Cholesterol Transport in Mice NIELS NIJSTAD,* THOMAS GAUTIER,* FRANÇOIS BRIAND, DANIEL J. RADER,
GASTROENTEROLOGY 2011;140:1043 1051 Biliary Sterol Secretion Is Required for Functional In Vivo Reverse Cholesterol Transport in Mice NIELS NIJSTAD,* THOMAS GAUTIER,* FRANÇOIS BRIAND, DANIEL J. RADER,
Commentary. New mechanisms by which statins lower plasma cholesterol. Henri Brunengraber
 Commentary New mechanisms by which statins lower plasma cholesterol Henri Brunengraber Department of Nutrition Case Western Reserve University Cleveland, OH 44109 Phone: 216 368 6429 Fax: 216 368 6560
Commentary New mechanisms by which statins lower plasma cholesterol Henri Brunengraber Department of Nutrition Case Western Reserve University Cleveland, OH 44109 Phone: 216 368 6429 Fax: 216 368 6560
Intestinal cholesterol secretion: future clinical implications
 REVIEW Intestinal cholesterol secretion: future clinical implications L. Jakulj 1 *, J. Besseling 1, E.S.G. Stroes, 1 A.K. Groen 2 1 Department of Vascular Medicine, Academic Medical Centre, Amsterdam,
REVIEW Intestinal cholesterol secretion: future clinical implications L. Jakulj 1 *, J. Besseling 1, E.S.G. Stroes, 1 A.K. Groen 2 1 Department of Vascular Medicine, Academic Medical Centre, Amsterdam,
Summary and concluding remarks
 Summary and concluding remarks This thesis is focused on the role and interaction of different cholesterol and phospholipid transporters. Cholesterol homeostasis is accomplished via a tightly regulated
Summary and concluding remarks This thesis is focused on the role and interaction of different cholesterol and phospholipid transporters. Cholesterol homeostasis is accomplished via a tightly regulated
It is generally accepted that lipoproteins carry cholesterol
 Red Blood Cells Play a Role in Reverse Cholesterol Transport Kimberly T. Hung, Stela Z. Berisha, Brian M. Ritchey, Jennifer Santore, Jonathan D. Smith Objective Reverse cholesterol transport (RCT) involves
Red Blood Cells Play a Role in Reverse Cholesterol Transport Kimberly T. Hung, Stela Z. Berisha, Brian M. Ritchey, Jennifer Santore, Jonathan D. Smith Objective Reverse cholesterol transport (RCT) involves
Lipoprotein Formation, Structure and Metabolism: Cholesterol Balance and the Regulation of Plasma Lipid Levels
 Lipoprotein Formation, Structure and Metabolism: Balance and the Regulation of Plasma Lipid Levels David E. Cohen, MD, PhD Director of Hepatology, Gastroenterology Division, Brigham and Women s Hospital
Lipoprotein Formation, Structure and Metabolism: Balance and the Regulation of Plasma Lipid Levels David E. Cohen, MD, PhD Director of Hepatology, Gastroenterology Division, Brigham and Women s Hospital
Direct Intestinal Cholesterol Secretion Contributes Significantly to Total Fecal Neutral Sterol Excretion in Mice
 GASTROENTEROLOGY 2007;133:967 975 Direct Intestinal Cholesterol Secretion Contributes Significantly to Total Fecal Neutral Sterol Excretion in Mice ASTRID E. VAN DER VELDE,* CARLOS L. J. VRINS,* KARIN
GASTROENTEROLOGY 2007;133:967 975 Direct Intestinal Cholesterol Secretion Contributes Significantly to Total Fecal Neutral Sterol Excretion in Mice ASTRID E. VAN DER VELDE,* CARLOS L. J. VRINS,* KARIN
Trans-intestinal cholesterol efflux is not mediated through high density lipoprotein
 Trans-intestinal cholesterol efflux is not mediated through high density lipoprotein Carlos L. J. Vrins, *, Roelof Ottenhoff, * Karin van den Oever, Dirk R. de Waart, J. Kar Kruyt, Ying Zhao, Theo J. C.
Trans-intestinal cholesterol efflux is not mediated through high density lipoprotein Carlos L. J. Vrins, *, Roelof Ottenhoff, * Karin van den Oever, Dirk R. de Waart, J. Kar Kruyt, Ying Zhao, Theo J. C.
We are IntechOpen, the world s leading publisher of Open Access books Built by scientists, for scientists. International authors and editors
 We are IntechOpen, the world s leading publisher of Open Access books Built by scientists, for scientists 4,000 116,000 120M Open access books available International authors and editors Downloads Our
We are IntechOpen, the world s leading publisher of Open Access books Built by scientists, for scientists 4,000 116,000 120M Open access books available International authors and editors Downloads Our
Lipid accumulation in macrophages, leading to formation
 ATVB in Focus HDL Structure, Function, Therapeutics, and Imaging Series Editor: Stanley Hazen Reverse Cholesterol Transport Revisited Contribution of Biliary Versus Intestinal Cholesterol Excretion Gemma
ATVB in Focus HDL Structure, Function, Therapeutics, and Imaging Series Editor: Stanley Hazen Reverse Cholesterol Transport Revisited Contribution of Biliary Versus Intestinal Cholesterol Excretion Gemma
Acute Sterol O-Acyltransferase 2 (SOAT2) Knockdown Rapidly Mobilizes Hepatic Cholesterol for Fecal Excretion
 University of Kentucky UKnowledge Saha Cardiovascular Research Center Faculty Publications Cardiovascular Research 6-5-2014 Acute Sterol O-Acyltransferase 2 (SOAT2) Knockdown Rapidly Mobilizes Hepatic
University of Kentucky UKnowledge Saha Cardiovascular Research Center Faculty Publications Cardiovascular Research 6-5-2014 Acute Sterol O-Acyltransferase 2 (SOAT2) Knockdown Rapidly Mobilizes Hepatic
High density lipoprotein metabolism
 High density lipoprotein metabolism Lipoprotein classes and atherosclerosis Chylomicrons, VLDL, and their catabolic remnants Pro-atherogenic LDL HDL Anti-atherogenic Plasma lipid transport Liver VLDL FC
High density lipoprotein metabolism Lipoprotein classes and atherosclerosis Chylomicrons, VLDL, and their catabolic remnants Pro-atherogenic LDL HDL Anti-atherogenic Plasma lipid transport Liver VLDL FC
2.5. AMPK activity
 Supplement Fig. A 3 B phos-ampk 2.5 * Control AICAR AMPK AMPK activity (Absorbance at 45 nm) 2.5.5 Control AICAR Supplement Fig. Effects of AICAR on AMPK activation in macrophages. J774. macrophages were
Supplement Fig. A 3 B phos-ampk 2.5 * Control AICAR AMPK AMPK activity (Absorbance at 45 nm) 2.5.5 Control AICAR Supplement Fig. Effects of AICAR on AMPK activation in macrophages. J774. macrophages were
Cholesterol absorption is mainly regulated by the jejunal and ileal ATP-binding cassette sterol efflux transporters Abcg5 and Abcg8 in mice 1
 Cholesterol absorption is mainly regulated by the jejunal and ileal ATP-binding cassette sterol efflux transporters Abcg5 and Abcg8 in mice 1 Li-Ping Duan, Helen H. Wang, and David Q-H. Wang 2 Department
Cholesterol absorption is mainly regulated by the jejunal and ileal ATP-binding cassette sterol efflux transporters Abcg5 and Abcg8 in mice 1 Li-Ping Duan, Helen H. Wang, and David Q-H. Wang 2 Department
Regulating Hepatic Cellular Cholesterol
 Under circumstances of cholesterol deficiency, Sterol Regulatory Element Binding Proteins (SREBPs) via binding to DNA nuclear response elements set off genomic production of proteins and enzymes that induce
Under circumstances of cholesterol deficiency, Sterol Regulatory Element Binding Proteins (SREBPs) via binding to DNA nuclear response elements set off genomic production of proteins and enzymes that induce
The new guidelines issued in PRESENTATIONS... Future Outlook: Changing Perspectives on Best Practice
 ... PRESENTATIONS... Future Outlook: Changing Perspectives on Best Practice Based on a presentation by Daniel J. Rader, MD Presentation Summary The guidelines recently released by the National Cholesterol
... PRESENTATIONS... Future Outlook: Changing Perspectives on Best Practice Based on a presentation by Daniel J. Rader, MD Presentation Summary The guidelines recently released by the National Cholesterol
Plasma cholesterol has been established as a predictor of
 Dietary Fat Induced Alterations in Atherosclerosis Are Abolished by ACAT2-Deficiency in ApoB100 Only, LDLr / Mice Thomas A. Bell III, Kathryn Kelley, Martha D. Wilson, Janet K. Sawyer, Lawrence L. Rudel
Dietary Fat Induced Alterations in Atherosclerosis Are Abolished by ACAT2-Deficiency in ApoB100 Only, LDLr / Mice Thomas A. Bell III, Kathryn Kelley, Martha D. Wilson, Janet K. Sawyer, Lawrence L. Rudel
UvA-DARE (Digital Academic Repository) Novel insights in cholesterol excretion van der Velde, A.E. Link to publication
 UvA-DARE (Digital Academic Repository) Novel insights in cholesterol excretion van der Velde, A.E. Link to publication Citation for published version (APA): van der Velde, A. E. (2009). Novel insights
UvA-DARE (Digital Academic Repository) Novel insights in cholesterol excretion van der Velde, A.E. Link to publication Citation for published version (APA): van der Velde, A. E. (2009). Novel insights
Ebrahim Abbasi Oshaghi 1,2
 1 2 Flaxseed normalized antioxidant status and also changed ABCG5 and ABCG8 genes expression in diabetic rat Fatemeh Mirzaei 1,Mona Pourjafarr 1, Seyyed Alireza Vafaei 1, Rezvan Mostoli 1, Ebrahim Abbasi
1 2 Flaxseed normalized antioxidant status and also changed ABCG5 and ABCG8 genes expression in diabetic rat Fatemeh Mirzaei 1,Mona Pourjafarr 1, Seyyed Alireza Vafaei 1, Rezvan Mostoli 1, Ebrahim Abbasi
Increased Fecal Neutral Sterol Loss Upon Liver X Receptor Activation Is Independent of Biliary Sterol Secretion in Mice
 GASTROENTEROLOGY 2005;128:147 156 Increased Fecal Neutral Sterol Loss Upon Liver X Receptor Activation Is Independent of Biliary Sterol Secretion in Mice JANINE K. KRUIT,* TORSTEN PLÖSCH,* RICK HAVINGA,*
GASTROENTEROLOGY 2005;128:147 156 Increased Fecal Neutral Sterol Loss Upon Liver X Receptor Activation Is Independent of Biliary Sterol Secretion in Mice JANINE K. KRUIT,* TORSTEN PLÖSCH,* RICK HAVINGA,*
body inhibition of MTP on cholesterol metabolism and RCT in mice
 Differential impact of hepatic deficiency and total body inhibition of MTP on cholesterol metabolism and RCT in mice Arne Dikkers, * Wijtske Annema, *, Jan Freark de Boer, * Jahangir Iqbal, M. Mahmood
Differential impact of hepatic deficiency and total body inhibition of MTP on cholesterol metabolism and RCT in mice Arne Dikkers, * Wijtske Annema, *, Jan Freark de Boer, * Jahangir Iqbal, M. Mahmood
Intestinal cholesterol absorption is substantially reduced in mice deficient in both ABCA1 and ACAT2
 Intestinal cholesterol absorption is substantially reduced in mice deficient in both ABCA1 and ACAT2 Ryan E. Temel, Richard G. Lee, Kathryn L. Kelley, Matthew A. Davis, Ramesh Shah, Janet K. Sawyer, Martha
Intestinal cholesterol absorption is substantially reduced in mice deficient in both ABCA1 and ACAT2 Ryan E. Temel, Richard G. Lee, Kathryn L. Kelley, Matthew A. Davis, Ramesh Shah, Janet K. Sawyer, Martha
Cholesterol Metabolism
 Cholesterol Metabolism Lippincott s Illustrated Review Chapter 18 Steroid Nucleus 1 2 Cholesterol was isolated from gall bladder stones in 1774 3 Sources and Elimination of Cholesterol Synthesis: 1000
Cholesterol Metabolism Lippincott s Illustrated Review Chapter 18 Steroid Nucleus 1 2 Cholesterol was isolated from gall bladder stones in 1774 3 Sources and Elimination of Cholesterol Synthesis: 1000
Integrative Physiology
 Integrative Physiology Acute Psychological Stress Accelerates Reverse Cholesterol Transport via Corticosterone-Dependent Inhibition of Intestinal Cholesterol Absorption Reija Silvennoinen, Joan Carles
Integrative Physiology Acute Psychological Stress Accelerates Reverse Cholesterol Transport via Corticosterone-Dependent Inhibition of Intestinal Cholesterol Absorption Reija Silvennoinen, Joan Carles
Hepatic Niemann-Pick C1 like 1 regulates biliary cholesterol concentration and is a target of ezetimibe
 Research article Hepatic Niemann-Pick C1 like 1 regulates biliary cholesterol concentration and is a target of ezetimibe Ryan E. Temel, 1 Weiqing Tang, 1 Yinyan Ma, 1 Lawrence L. Rudel, 1 Mark C. Willingham,
Research article Hepatic Niemann-Pick C1 like 1 regulates biliary cholesterol concentration and is a target of ezetimibe Ryan E. Temel, 1 Weiqing Tang, 1 Yinyan Ma, 1 Lawrence L. Rudel, 1 Mark C. Willingham,
The TMAO-Generating Enzyme Flavin Monooxygenase 3 Is a Central Regulator of Cholesterol Balance
 Report The TMAO-Generating Enzyme Flavin Monooxygenase 3 Is a Central Regulator of Cholesterol Balance Graphical Abstract Authors Manya Warrier, Diana M. Shih,..., Ryan E. Temel, J. Mark Brown Correspondence
Report The TMAO-Generating Enzyme Flavin Monooxygenase 3 Is a Central Regulator of Cholesterol Balance Graphical Abstract Authors Manya Warrier, Diana M. Shih,..., Ryan E. Temel, J. Mark Brown Correspondence
Supplemental Material. Results
 Supplemental Material Results Fractionation of mouse plasma by high-resolution SEC. APOA1 eluted as a single major peak in fractions 16 of plasma (the apparent size of mature, lipidated HDL) when it was
Supplemental Material Results Fractionation of mouse plasma by high-resolution SEC. APOA1 eluted as a single major peak in fractions 16 of plasma (the apparent size of mature, lipidated HDL) when it was
The role of reverse cholesterol transport in animals and humans and relationship to atherosclerosis
 The role of reverse cholesterol transport in animals and humans and relationship to atherosclerosis Daniel J. Rader, 1, *, Eric T. Alexander,*, Ginny L. Weibel, Jeffrey Billheimer,* and George H. Rothblat
The role of reverse cholesterol transport in animals and humans and relationship to atherosclerosis Daniel J. Rader, 1, *, Eric T. Alexander,*, Ginny L. Weibel, Jeffrey Billheimer,* and George H. Rothblat
Lipoproteins Metabolism Reference: Campbell Biochemistry and Lippincott s Biochemistry
 Lipoproteins Metabolism Reference: Campbell Biochemistry and Lippincott s Biochemistry Learning Objectives 1. Define lipoproteins and explain the rationale of their formation in blood. 2. List different
Lipoproteins Metabolism Reference: Campbell Biochemistry and Lippincott s Biochemistry Learning Objectives 1. Define lipoproteins and explain the rationale of their formation in blood. 2. List different
Cholesterol and Lipoprotein Metabolism and Atherosclerosis: Recent Advances in Reverse Cholesterol Transport
 Cholesterol and Lipoprotein Metabolism and Atherosclerosis. 27 November, Vol. 16 (Suppl. 1), 2017: s27-s42 The Official Journal of the Mexican Association of Hepatology, the Latin-American Association
Cholesterol and Lipoprotein Metabolism and Atherosclerosis. 27 November, Vol. 16 (Suppl. 1), 2017: s27-s42 The Official Journal of the Mexican Association of Hepatology, the Latin-American Association
Liver X receptor activation promotes macrophage-to-feces reverse cholesterol transport in a dyslipidemic hamster model
 Liver X receptor activation promotes macrophage-to-feces reverse cholesterol transport in a dyslipidemic hamster model François Briand, 1, * Morgan Tréguier, Agnès André, *, Didier Grillot, Marc Issandou,
Liver X receptor activation promotes macrophage-to-feces reverse cholesterol transport in a dyslipidemic hamster model François Briand, 1, * Morgan Tréguier, Agnès André, *, Didier Grillot, Marc Issandou,
Lipid Digestion. An Introduction to Lipid Transport and Digestion with consideration of High Density and Low Density Lipoproteins.
 Digestion An Introduction to Transport and Digestion with consideration of High Density and Low Density Lipoproteins By Noel Ways Suspension and Nutralization of Chyme ph Boli containing lipids enters
Digestion An Introduction to Transport and Digestion with consideration of High Density and Low Density Lipoproteins By Noel Ways Suspension and Nutralization of Chyme ph Boli containing lipids enters
It is well-known that nutritional factors influence the
 Fish Oil Promotes Macrophage Reverse Cholesterol Transport in Mice Tomoyuki Nishimoto, Michael A. Pellizzon, Masakazu Aihara, Ioannis M. Stylianou, Jeffery T. Billheimer, George Rothblat, Daniel J. Rader
Fish Oil Promotes Macrophage Reverse Cholesterol Transport in Mice Tomoyuki Nishimoto, Michael A. Pellizzon, Masakazu Aihara, Ioannis M. Stylianou, Jeffery T. Billheimer, George Rothblat, Daniel J. Rader
Investigations on the mechanism of hypercholesterolemia observed in copper deficiency in rats
 J. Biosci., Vol. 12, Number 2, June 1987, pp. 137 142. Printed in India. Investigations on the mechanism of hypercholesterolemia observed in copper deficiency in rats P. VALSALA and P. A. KURUP Department
J. Biosci., Vol. 12, Number 2, June 1987, pp. 137 142. Printed in India. Investigations on the mechanism of hypercholesterolemia observed in copper deficiency in rats P. VALSALA and P. A. KURUP Department
 Acetyl CoA HMG CoA Mevalonate (C6) Dimethylallyl Pyrophosphate isopentenyl Pyrophosphate (C5) Geranyl Pyrophosphate (C10) FarnesylPyrophosphate (C15) Squalene (C30) Lanosterol (C30) 7 Dehydrocholesterol
Acetyl CoA HMG CoA Mevalonate (C6) Dimethylallyl Pyrophosphate isopentenyl Pyrophosphate (C5) Geranyl Pyrophosphate (C10) FarnesylPyrophosphate (C15) Squalene (C30) Lanosterol (C30) 7 Dehydrocholesterol
Nature Medicine doi: /nm.3150
 Nature Medicine doi:10.1038/nm.3150 Supplementary Table 1 Primer sequences used for q-pcr analysis Gene Forward primer Reverse primer Abca1 CAGCTTCCATCCTCCTTGTC CCACATCCACAACTGTCTGG Abcg1 GTACCATGACATCGCTGGTG
Nature Medicine doi:10.1038/nm.3150 Supplementary Table 1 Primer sequences used for q-pcr analysis Gene Forward primer Reverse primer Abca1 CAGCTTCCATCCTCCTTGTC CCACATCCACAACTGTCTGG Abcg1 GTACCATGACATCGCTGGTG
Hepatic SR-BI, not endothelial lipase, expression determines biliary cholesterol secretion in mice 1
 Hepatic SR-BI, not endothelial lipase, expression determines biliary cholesterol secretion in mice 1 Harmen Wiersma, 1 Alberto Gatti, Niels Nijstad, Folkert Kuipers, and Uwe J. F. Tietge 2 Center for Liver,
Hepatic SR-BI, not endothelial lipase, expression determines biliary cholesterol secretion in mice 1 Harmen Wiersma, 1 Alberto Gatti, Niels Nijstad, Folkert Kuipers, and Uwe J. F. Tietge 2 Center for Liver,
Is it really that simple? Alyssa Hasty, PhD Associate Professor Molecular Physiology and Biophysics
 Alyssa Hasty, PhD Associate Professor Molecular Physiology and Biophysics Why we care about hepatic lipogenesis Control of lipid synthesis What can go wrong in humans Animal models dlto study lipoprotein
Alyssa Hasty, PhD Associate Professor Molecular Physiology and Biophysics Why we care about hepatic lipogenesis Control of lipid synthesis What can go wrong in humans Animal models dlto study lipoprotein
Reverse Transport of Cholesterol Is the Reason for Resistance to Development of Atherosclerosis in Prague Hereditary Hypercholesterolemic (PHHC) Rat
 Physiol. Res. 63: 591-596, 2014 Reverse Transport of Cholesterol Is the Reason for Resistance to Development of Atherosclerosis in Prague Hereditary Hypercholesterolemic (PHHC) Rat M. SCHMIEDTOVA 1, M.
Physiol. Res. 63: 591-596, 2014 Reverse Transport of Cholesterol Is the Reason for Resistance to Development of Atherosclerosis in Prague Hereditary Hypercholesterolemic (PHHC) Rat M. SCHMIEDTOVA 1, M.
Lipid Digestion. and Human Nutrition. An Introduction to Lipid Transport and Digestion with consideration of High Density and Low Density Lipoproteins
 Digestion and Human Nutrition An Introduction to Transport and Digestion with consideration of High Density and Low Density Lipoproteins By Noel Ways Emulsification of s and release of Pancreatic Lipase
Digestion and Human Nutrition An Introduction to Transport and Digestion with consideration of High Density and Low Density Lipoproteins By Noel Ways Emulsification of s and release of Pancreatic Lipase
Lipids digestion and absorption, Biochemistry II
 Lipids digestion and absorption, blood plasma lipids, lipoproteins Biochemistry II Lecture 1 2008 (J.S.) Triacylglycerols (as well as free fatty acids and both free and esterified cholesterol) are very
Lipids digestion and absorption, blood plasma lipids, lipoproteins Biochemistry II Lecture 1 2008 (J.S.) Triacylglycerols (as well as free fatty acids and both free and esterified cholesterol) are very
Cholesterol metabolism. Function Biosynthesis Transport in the organism Hypercholesterolemia
 Cholesterol metabolism Function Biosynthesis Transport in the organism Hypercholesterolemia - component of all cell membranes - precursor of bile acids steroid hormones vitamin D Cholesterol Sources: dietary
Cholesterol metabolism Function Biosynthesis Transport in the organism Hypercholesterolemia - component of all cell membranes - precursor of bile acids steroid hormones vitamin D Cholesterol Sources: dietary
LIVER PHYSIOLOGY AND DISEASE
 GASTROENTEROLOGY 64: 298-303, 1973 Copyright 1973 by The Williams & Wilkins Co. Vol. 64, No.2 Printed in U.S.A. LIVER PHYSIOLOGY AND DISEASE BILE ACID METABOLISM IN CIRRHOSIS III. Biliary lipid secretion
GASTROENTEROLOGY 64: 298-303, 1973 Copyright 1973 by The Williams & Wilkins Co. Vol. 64, No.2 Printed in U.S.A. LIVER PHYSIOLOGY AND DISEASE BILE ACID METABOLISM IN CIRRHOSIS III. Biliary lipid secretion
Hydrophobic Surfactant Treatment Prevents Atherosclerosis in the Rabbit
 Hydrophobic Surfactant Treatment Prevents Atherosclerosis in the Rabbit RAPID PUBLICATIONS J. B. RODGERS, E. C. KYRIAKIDES, B. KAPUSCINSKA, S. K. PENG, and W. J. BOCHENEK, Department of Medicine, Albany
Hydrophobic Surfactant Treatment Prevents Atherosclerosis in the Rabbit RAPID PUBLICATIONS J. B. RODGERS, E. C. KYRIAKIDES, B. KAPUSCINSKA, S. K. PENG, and W. J. BOCHENEK, Department of Medicine, Albany
Cholesterol feeding strongly reduces hepatic VLDL-triglyceride production in mice lacking the liver X receptor a
 Cholesterol feeding strongly reduces hepatic VLDL-triglyceride production in mice lacking the liver X receptor a Jelske N. van der Veen, 1, * Rick Havinga,* Vincent W. Bloks,* Albert K. Groen, and Folkert
Cholesterol feeding strongly reduces hepatic VLDL-triglyceride production in mice lacking the liver X receptor a Jelske N. van der Veen, 1, * Rick Havinga,* Vincent W. Bloks,* Albert K. Groen, and Folkert
Lipid Metabolism Prof. Dr. rer physiol. Dr.h.c. Ulrike Beisiegel
 Lipid Metabolism Department of Biochemistry and Molecular Biology II Medical Center Hamburg-ppendorf 1 Lipids. visceral fat. nutritional lipids 0 1.5 3 4.5 9 h. serum lipids. lipid accumulation in the
Lipid Metabolism Department of Biochemistry and Molecular Biology II Medical Center Hamburg-ppendorf 1 Lipids. visceral fat. nutritional lipids 0 1.5 3 4.5 9 h. serum lipids. lipid accumulation in the
The LXR agonist T promotes the reverse cholesterol transport from macrophages by increasing plasma efflux potential
 The LXR agonist T0901317 promotes the reverse cholesterol transport from macrophages by increasing plasma efflux potential Ilaria Zanotti,* Francesco Potì,* Matteo Pedrelli,* Elda Favari,* Elsa Moleri,
The LXR agonist T0901317 promotes the reverse cholesterol transport from macrophages by increasing plasma efflux potential Ilaria Zanotti,* Francesco Potì,* Matteo Pedrelli,* Elda Favari,* Elsa Moleri,
Emerging roles of the intestine in control of cholesterol metabolism Kruit, Janine K.; Groen, Albert K.; van Berkel, Theo J.
 University of Groningen Emerging roles of the intestine in control of cholesterol metabolism Kruit, Janine K.; Groen, Albert K.; van Berkel, Theo J.; Kuipers, Folkert Published in: World Journal of Gastroenterology
University of Groningen Emerging roles of the intestine in control of cholesterol metabolism Kruit, Janine K.; Groen, Albert K.; van Berkel, Theo J.; Kuipers, Folkert Published in: World Journal of Gastroenterology
Reduced cholesterol absorption upon PPAR activation coincides with decreased intestinal expression of NPC1L1
 Reduced cholesterol absorption upon PPAR activation coincides with decreased intestinal expression of NPC1L1 Jelske N. van der Veen, 1,2, * Janine K. Kruit, 2, * Rick Havinga,* Julius F. W. Baller,* Giovanna
Reduced cholesterol absorption upon PPAR activation coincides with decreased intestinal expression of NPC1L1 Jelske N. van der Veen, 1,2, * Janine K. Kruit, 2, * Rick Havinga,* Julius F. W. Baller,* Giovanna
Lipid/Lipoprotein Structure and Metabolism (Overview)
 Lipid/Lipoprotein Structure and Metabolism (Overview) Philip Barter President, International Atherosclerosis Society Centre for Vascular Research University of New South Wales Sydney, Australia Disclosures
Lipid/Lipoprotein Structure and Metabolism (Overview) Philip Barter President, International Atherosclerosis Society Centre for Vascular Research University of New South Wales Sydney, Australia Disclosures
Potential Atheroprotective Effects of Ixmyelocel-T Cellular Therapy. Kelly J. Ledford, Nikki Murphy, Frank Zeigler, Ronnda L.
 Potential Atheroprotective Effects of Ixmyelocel-T Cellular Therapy Kelly J. Ledford, Nikki Murphy, Frank Zeigler, Ronnda L. Bartel 1 Ixmyelocel-T, an expanded, autologous multicellular therapy cultured
Potential Atheroprotective Effects of Ixmyelocel-T Cellular Therapy Kelly J. Ledford, Nikki Murphy, Frank Zeigler, Ronnda L. Bartel 1 Ixmyelocel-T, an expanded, autologous multicellular therapy cultured
A model for cholesterol absorption: isotope vs. mass; single dose vs. constant infusion
 A model for cholesterol absorption: isotope vs. mass; single dose vs. constant infusion Donald B. Zilversmit' Division of Nutritional Sciences and Section of Biochemistry, Molecular and Cell Biology, Division
A model for cholesterol absorption: isotope vs. mass; single dose vs. constant infusion Donald B. Zilversmit' Division of Nutritional Sciences and Section of Biochemistry, Molecular and Cell Biology, Division
Antihyperlipidemic Drugs
 Antihyperlipidemic Drugs Hyperlipidemias. Hyperlipoproteinemias. Hyperlipemia. Hypercholestrolemia. Direct relationship with acute pancreatitis and atherosclerosis Structure Lipoprotein Particles Types
Antihyperlipidemic Drugs Hyperlipidemias. Hyperlipoproteinemias. Hyperlipemia. Hypercholestrolemia. Direct relationship with acute pancreatitis and atherosclerosis Structure Lipoprotein Particles Types
Incorporating HDL interaction in an ODE model of Atherosclerosis
 Incorporating HDL interaction in an ODE model of Atherosclerosis Diego Lopez Texas A&M University July 29, 2015 Diego Lopez (TAMU) July 29, 2015 1 / 10 Introduction Atherosclerosis A disease of the arteries
Incorporating HDL interaction in an ODE model of Atherosclerosis Diego Lopez Texas A&M University July 29, 2015 Diego Lopez (TAMU) July 29, 2015 1 / 10 Introduction Atherosclerosis A disease of the arteries
Role of reverse cholesterol transport in animals and humans and relationship to atherosclerosis
 Role of reverse cholesterol transport in animals and humans and relationship to atherosclerosis Daniel J. Rader 1,2, Eric T. Alexander 1,2, Ginny L. Weibel 2, Jeffrey Billheimer 1, George H. Rothblat 2
Role of reverse cholesterol transport in animals and humans and relationship to atherosclerosis Daniel J. Rader 1,2, Eric T. Alexander 1,2, Ginny L. Weibel 2, Jeffrey Billheimer 1, George H. Rothblat 2
Lipid Metabolism in Familial Hypercholesterolemia
 Lipid Metabolism in Familial Hypercholesterolemia Khalid Al-Rasadi, BSc, MD, FRCPC Head of Biochemistry Department, SQU Head of Lipid and LDL-Apheresis Unit, SQUH President of Oman society of Lipid & Atherosclerosis
Lipid Metabolism in Familial Hypercholesterolemia Khalid Al-Rasadi, BSc, MD, FRCPC Head of Biochemistry Department, SQU Head of Lipid and LDL-Apheresis Unit, SQUH President of Oman society of Lipid & Atherosclerosis
Podcast (Video Recorded Lecture Series): Lipoprotein Metabolism and Lipid Therapy for the USMLE Step One Exam
 Podcast (Video Recorded Lecture Series): Lipoprotein Metabolism and Lipid Therapy for the USMLE Step One Exam Howard J. Sachs, MD www.12daysinmarch.com Email: Howard@12daysinmarch.com Podcast (Video Recorded
Podcast (Video Recorded Lecture Series): Lipoprotein Metabolism and Lipid Therapy for the USMLE Step One Exam Howard J. Sachs, MD www.12daysinmarch.com Email: Howard@12daysinmarch.com Podcast (Video Recorded
Chapter VIII: Dr. Sameh Sarray Hlaoui
 Chapter VIII: Dr. Sameh Sarray Hlaoui Lipoproteins a Lipids are insoluble in plasma. In order to be transported they are combined with specific proteins to form lipoproteins: Clusters of proteins and lipids.
Chapter VIII: Dr. Sameh Sarray Hlaoui Lipoproteins a Lipids are insoluble in plasma. In order to be transported they are combined with specific proteins to form lipoproteins: Clusters of proteins and lipids.
Transintestinal transport of the anti-inflammatory drug 4F and the modulation of transintestinal cholesterol efflux
 Transintestinal transport of the anti-inflammatory drug 4F and the modulation of transintestinal cholesterol efflux David Meriwether,*, Dawoud Sulaiman,*, Alan Wagner,* Victor Grijalva,* Izumi Kaji,* Kevin
Transintestinal transport of the anti-inflammatory drug 4F and the modulation of transintestinal cholesterol efflux David Meriwether,*, Dawoud Sulaiman,*, Alan Wagner,* Victor Grijalva,* Izumi Kaji,* Kevin
Reviewers' comments: Reviewer #1 (Remarks to the Author):
 Reviewers' comments: Reviewer #1 (Remarks to the Author): Bempedoic acid (ETC-1002) is an ATP-citrate lyase (ACL) inhibitor that in clinical studies has been shown to decrease plasma LDL cholesterol apparently
Reviewers' comments: Reviewer #1 (Remarks to the Author): Bempedoic acid (ETC-1002) is an ATP-citrate lyase (ACL) inhibitor that in clinical studies has been shown to decrease plasma LDL cholesterol apparently
Liver X receptor-mediated activation of reverse cholesterol. transport from macrophages to feces in vivo requires ATPbinding
 Liver X receptor-mediated activation of reverse cholesterol transport from macrophages to feces in vivo requires ATPbinding cassette (ABC) G5/G8 Laura Calpe-Berdiel a, Noemí Rotllan a, Catherine Fiévet
Liver X receptor-mediated activation of reverse cholesterol transport from macrophages to feces in vivo requires ATPbinding cassette (ABC) G5/G8 Laura Calpe-Berdiel a, Noemí Rotllan a, Catherine Fiévet
CETP inhibition: pros and cons. Philip Barter The Heart Research Institute Sydney, Australia
 CETP inhibition: pros and cons Philip Barter The Heart Research Institute Sydney, Australia Philip Barter Disclosures Received honorariums for lectures, consultancies or membership of advisory boards from:
CETP inhibition: pros and cons Philip Barter The Heart Research Institute Sydney, Australia Philip Barter Disclosures Received honorariums for lectures, consultancies or membership of advisory boards from:
Lipoproteins Metabolism
 Lipoproteins Metabolism LEARNING OBJECTIVES By the end of this Lecture, the student should be able to describe: What are Lipoproteins? Describe Lipoprotein Particles. Composition of Lipoproteins. The chemical
Lipoproteins Metabolism LEARNING OBJECTIVES By the end of this Lecture, the student should be able to describe: What are Lipoproteins? Describe Lipoprotein Particles. Composition of Lipoproteins. The chemical
The inhibition of CETP: From simply raising HDL-c to promoting cholesterol efflux and lowering of atherogenic lipoproteins Prof Dr J Wouter Jukema
 The inhibition of CETP: From simply raising HDL-c to promoting cholesterol efflux and lowering of atherogenic lipoproteins Prof Dr J Wouter Jukema Dept Cardiology, Leiden University Medical Center, Leiden,
The inhibition of CETP: From simply raising HDL-c to promoting cholesterol efflux and lowering of atherogenic lipoproteins Prof Dr J Wouter Jukema Dept Cardiology, Leiden University Medical Center, Leiden,
Treatment of Atherosclerosis in 2007
 Treatment of Atherosclerosis in 2007 Szilard Voros, M.D. Medical Director Cardiovascular MR and CT Piedmont Hospital, Piedmont Hospital Our Paradigm Genotype Phenotype Environment Atherosclerotic Disease
Treatment of Atherosclerosis in 2007 Szilard Voros, M.D. Medical Director Cardiovascular MR and CT Piedmont Hospital, Piedmont Hospital Our Paradigm Genotype Phenotype Environment Atherosclerotic Disease
Cooperation between hepatic cholesteryl ester hydrolase. and scavenger receptor BI for hydrolysis of HDL-CE
 Cooperation between hepatic cholesteryl ester hydrolase and scavenger receptor BI for hydrolysis of HDL-CE Quan Yuan, Jinghua Bie, Jing Wang, Siddhartha S. Ghosh, and Shobha Ghosh 1 Department of Internal
Cooperation between hepatic cholesteryl ester hydrolase and scavenger receptor BI for hydrolysis of HDL-CE Quan Yuan, Jinghua Bie, Jing Wang, Siddhartha S. Ghosh, and Shobha Ghosh 1 Department of Internal
The central role of the liver in regulating the production
 LIVER BIOLOGY AND PATHOBIOLOGY BIOLOGY ACAT2 Deficiency Limits Cholesterol Absorption in the Cholesterol-Fed Mouse: Impact on Hepatic Cholesterol Homeostasis Joyce J. Repa, 1,2 Kimberly K. Buhman, 3 Robert
LIVER BIOLOGY AND PATHOBIOLOGY BIOLOGY ACAT2 Deficiency Limits Cholesterol Absorption in the Cholesterol-Fed Mouse: Impact on Hepatic Cholesterol Homeostasis Joyce J. Repa, 1,2 Kimberly K. Buhman, 3 Robert
Mechanisms of high density lipoprotein-mediated efflux of cholesterol from cell plasma membranes
 Atherosclerosis 137 Suppl. (1998) S13 S17 Mechanisms of high density lipoprotein-mediated efflux of cholesterol from cell plasma membranes Michael C. Phillips *, Kristin L. Gillotte, M. Page Haynes, William
Atherosclerosis 137 Suppl. (1998) S13 S17 Mechanisms of high density lipoprotein-mediated efflux of cholesterol from cell plasma membranes Michael C. Phillips *, Kristin L. Gillotte, M. Page Haynes, William
Citation for published version (APA): Lukovac, S. (2010). Essential fatty acid deficiency and the small intestine Groningen: s.n.
 University of Groningen Essential fatty acid deficiency and the small intestine Lukovac, Sabina IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you wish to cite
University of Groningen Essential fatty acid deficiency and the small intestine Lukovac, Sabina IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you wish to cite
Role of apolipoprotein B-containing lipoproteins in the development of atherosclerosis Jan Borén MD, PhD
 Role of apolipoprotein B-containing lipoproteins in the development of atherosclerosis Jan Borén MD, PhD Our laboratory focuses on the role of apolipoprotein (apo) B- containing lipoproteins in normal
Role of apolipoprotein B-containing lipoproteins in the development of atherosclerosis Jan Borén MD, PhD Our laboratory focuses on the role of apolipoprotein (apo) B- containing lipoproteins in normal
Diabetes Contributes to Cholesterol Metabolism Regardless of Obesity
 Clinical Care/Education/Nutrition O R I G I N A L A R T I C L E Diabetes Contributes to Cholesterol Metabolism Regardless of Obesity PIIA P. SIMONEN, MD 1 HELENA K. GYLLING, MD 2 TATU A. MIETTINEN, MD
Clinical Care/Education/Nutrition O R I G I N A L A R T I C L E Diabetes Contributes to Cholesterol Metabolism Regardless of Obesity PIIA P. SIMONEN, MD 1 HELENA K. GYLLING, MD 2 TATU A. MIETTINEN, MD
thoracic lymph duct cannulation
 Cholesterol esterification by ACAT2 is essential for efficient intestinal cholesterol absorption: evidence from thoracic lymph duct cannulation Tam M. Nguyen, 1 Janet K. Sawyer, Kathryn L. Kelley, Matthew
Cholesterol esterification by ACAT2 is essential for efficient intestinal cholesterol absorption: evidence from thoracic lymph duct cannulation Tam M. Nguyen, 1 Janet K. Sawyer, Kathryn L. Kelley, Matthew
Effects of plant sterols and stanols on intestinal cholesterol metabolism: Suggested mechanisms from past to present
 1058 Mol. Nutr. Food Res. 2012, 56, 1058 1072 DOI 10.1002/mnfr.201100722 REVIEW Effects of plant sterols and stanols on intestinal cholesterol metabolism: Suggested mechanisms from past to present Els
1058 Mol. Nutr. Food Res. 2012, 56, 1058 1072 DOI 10.1002/mnfr.201100722 REVIEW Effects of plant sterols and stanols on intestinal cholesterol metabolism: Suggested mechanisms from past to present Els
Pathophysiology of Lipid Disorders
 Pathophysiology of Lipid Disorders Henry Ginsberg, M.D. Division of Preventive Medicine and Nutrition CHD in the United States CHD is the single largest killer of men and women 12 million have history
Pathophysiology of Lipid Disorders Henry Ginsberg, M.D. Division of Preventive Medicine and Nutrition CHD in the United States CHD is the single largest killer of men and women 12 million have history
ANSC/NUTR 618 LIPIDS & LIPID METABOLISM Lipoprotein Metabolism
 ANSC/NUTR 618 LIPIDS & LIPID METABOLISM Lipoprotein Metabolism I. Chylomicrons (exogenous pathway) A. 83% triacylglycerol, 2% protein, 8% cholesterol plus cholesterol esters, 7% phospholipid (esp. phosphatidylcholine)
ANSC/NUTR 618 LIPIDS & LIPID METABOLISM Lipoprotein Metabolism I. Chylomicrons (exogenous pathway) A. 83% triacylglycerol, 2% protein, 8% cholesterol plus cholesterol esters, 7% phospholipid (esp. phosphatidylcholine)
The Unique Acyl Chain Specificity of Biliary Phosphatidylcholines in Mice Is Independent of Their Biosynthetic Origin in the Liver
 The Unique Acyl Chain Specificity of Biliary Phosphatidylcholines in Mice Is Independent of Their Biosynthetic Origin in the Liver LUIS B. AGELLON, 1 CHRISTOPHER J. WALKEY, 1 DENNIS E. VANCE, 1 FOLKERT
The Unique Acyl Chain Specificity of Biliary Phosphatidylcholines in Mice Is Independent of Their Biosynthetic Origin in the Liver LUIS B. AGELLON, 1 CHRISTOPHER J. WALKEY, 1 DENNIS E. VANCE, 1 FOLKERT
activity through redistribution of intracellular cholesterol pools
 Proc. Natl. Acad. Sci. USA Vol. 89, pp. 1797-181, November 1992 Medical Sciences Fatty acids regulate hepatic low density lipoprotein receptor activity through redistribution of intracellular cholesterol
Proc. Natl. Acad. Sci. USA Vol. 89, pp. 1797-181, November 1992 Medical Sciences Fatty acids regulate hepatic low density lipoprotein receptor activity through redistribution of intracellular cholesterol
ORIGINS OF INTESTINAL ABCA1-MEDIATED HDL CHOLESTEROL. F. Jeffrey Field*, Kim Watt, Satya N. Mathur
 ORIGINS OF INTESTINAL ABCA1-MEDIATED HDL CHOLESTEROL F. Jeffrey Field, Kim Watt, Satya N. Mathur Department of Internal Medicine, University of Iowa, Iowa City, IA 52242. Running head: Intestinal HDL cholesterol
ORIGINS OF INTESTINAL ABCA1-MEDIATED HDL CHOLESTEROL F. Jeffrey Field, Kim Watt, Satya N. Mathur Department of Internal Medicine, University of Iowa, Iowa City, IA 52242. Running head: Intestinal HDL cholesterol
Transintestinal Cholesterol Transport Is Active in Mice and Humans and Controls Ezetimibe-Induced Fecal Neutral Sterol Excretion
 Clinical and Translational Report Transintestinal Cholesterol Transport Is Active in Mice and Humans and Controls Ezetimibe-Induced Fecal Neutral Sterol Excretion Graphical Abstract Authors Lily Jakulj,
Clinical and Translational Report Transintestinal Cholesterol Transport Is Active in Mice and Humans and Controls Ezetimibe-Induced Fecal Neutral Sterol Excretion Graphical Abstract Authors Lily Jakulj,
Hepatic Mttp deletion reverses gallstone susceptibility in L-Fabp knockout mice
 Hepatic Mttp deletion reverses gallstone susceptibility in L-Fabp knockout mice Yan Xie, * Ho Yee Joyce Fung, * Elizabeth P. Newberry, * Susan Kennedy, * Jianyang Luo, * Rosanne M. Crooke, Mark J. Graham,
Hepatic Mttp deletion reverses gallstone susceptibility in L-Fabp knockout mice Yan Xie, * Ho Yee Joyce Fung, * Elizabeth P. Newberry, * Susan Kennedy, * Jianyang Luo, * Rosanne M. Crooke, Mark J. Graham,
Pathophysiology of Bile Secretion
 Pathophysiology of Bile Secretion Martin C. Carey, D.Sc., M.D. Division of Gastroenterology, Brigham and Women s Hospital and Department of Medicine, Harvard Medical School Boston, MA, U.S.A. Functions
Pathophysiology of Bile Secretion Martin C. Carey, D.Sc., M.D. Division of Gastroenterology, Brigham and Women s Hospital and Department of Medicine, Harvard Medical School Boston, MA, U.S.A. Functions
1Why lipids cannot be transported in blood alone? 2How we transport Fatty acids and steroid hormones?
 1Why lipids cannot be transported in blood alone? 2How we transport Fatty acids and steroid hormones? 3How are dietary lipids transported? 4How lipids synthesized in the liver are transported? 5 Lipoprotien
1Why lipids cannot be transported in blood alone? 2How we transport Fatty acids and steroid hormones? 3How are dietary lipids transported? 4How lipids synthesized in the liver are transported? 5 Lipoprotien
Cellular control of cholesterol. Peter Takizawa Department of Cell Biology
 Cellular control of cholesterol Peter Takizawa Department of Cell Biology Brief overview of cholesterol s biological role Regulation of cholesterol synthesis Dietary and cellular uptake of cholesterol
Cellular control of cholesterol Peter Takizawa Department of Cell Biology Brief overview of cholesterol s biological role Regulation of cholesterol synthesis Dietary and cellular uptake of cholesterol
Effect of Ezetimibe on the Prevention and Dissolution of Cholesterol Gallstones
 GASTROENTEROLOGY 2008;134:2101 2110 Effect of Ezetimibe on the Prevention and Dissolution of Cholesterol Gallstones HELEN H. WANG,* PIERO PORTINCASA, NAHUM MENDEZ SANCHEZ, MISAEL URIBE, and DAVID Q. H.
GASTROENTEROLOGY 2008;134:2101 2110 Effect of Ezetimibe on the Prevention and Dissolution of Cholesterol Gallstones HELEN H. WANG,* PIERO PORTINCASA, NAHUM MENDEZ SANCHEZ, MISAEL URIBE, and DAVID Q. H.
Liver LXRα expression is crucial for whole body cholesterol homeostasis and reverse cholesterol transport in mice
 Research article Liver LXRα expression is crucial for whole body cholesterol homeostasis and reverse cholesterol transport in mice Yuan Zhang, 1 Sarah R. Breevoort, 2 Jerry Angdisen, 2 Mingui Fu, 1 Daniel
Research article Liver LXRα expression is crucial for whole body cholesterol homeostasis and reverse cholesterol transport in mice Yuan Zhang, 1 Sarah R. Breevoort, 2 Jerry Angdisen, 2 Mingui Fu, 1 Daniel
DRUG ELIMINATION II BILIARY EXCRETION MAMMARY, SALIVARY AND PULMONARY EXCRETION
 DRUG ELIMINATION II BILIARY EXCRETION MAMMARY, SALIVARY AND PULMONARY EXCRETION ROUTE OF DRUG ADMINISTRATION AND EXTRAHEPATIC DRUG METABOLISM The decline in plasma concentration after drug administration
DRUG ELIMINATION II BILIARY EXCRETION MAMMARY, SALIVARY AND PULMONARY EXCRETION ROUTE OF DRUG ADMINISTRATION AND EXTRAHEPATIC DRUG METABOLISM The decline in plasma concentration after drug administration
Supersaturated bile is an essential prerequisite for
 AMERICAN ASSOCIATION FOR THE STUDY OFLIVERD I S E ASES HEPATOLOGY, VOL. 64, NO. 3, 2016 Evidence That the Adenosine Triphosphate-Binding Cassette G5/G8-Independent Pathway Plays a Determinant Role in Cholesterol
AMERICAN ASSOCIATION FOR THE STUDY OFLIVERD I S E ASES HEPATOLOGY, VOL. 64, NO. 3, 2016 Evidence That the Adenosine Triphosphate-Binding Cassette G5/G8-Independent Pathway Plays a Determinant Role in Cholesterol
The clathrin adaptor Numb regulates intestinal cholesterol. absorption through dynamic interaction with NPC1L1
 The clathrin adaptor Numb regulates intestinal cholesterol absorption through dynamic interaction with NPC1L1 Pei-Shan Li 1, Zhen-Yan Fu 1,2, Ying-Yu Zhang 1, Jin-Hui Zhang 1, Chen-Qi Xu 1, Yi-Tong Ma
The clathrin adaptor Numb regulates intestinal cholesterol absorption through dynamic interaction with NPC1L1 Pei-Shan Li 1, Zhen-Yan Fu 1,2, Ying-Yu Zhang 1, Jin-Hui Zhang 1, Chen-Qi Xu 1, Yi-Tong Ma
Inhibition of PCSK9: The Birth of a New Therapy
 Inhibition of PCSK9: The Birth of a New Therapy Prof. John J.P. Kastelein, MD PhD FESC Dept. of Vascular Medicine Academic Medical Center / University of Amsterdam The Netherlands Disclosures Dr. Kastelein
Inhibition of PCSK9: The Birth of a New Therapy Prof. John J.P. Kastelein, MD PhD FESC Dept. of Vascular Medicine Academic Medical Center / University of Amsterdam The Netherlands Disclosures Dr. Kastelein
ANSC/NUTR 618 LIPIDS & LIPID METABOLISM The LDL Receptor, LDL Uptake, and the Free Cholesterol Pool
 ANSC/NUTR 618 LIPIDS & LIPID METABOLISM The, LDL Uptake, and the Free Cholesterol Pool I. Michael Brown and Joseph Goldstein A. Studied families with familial hypercholesterolemia. B. Defined the relationship
ANSC/NUTR 618 LIPIDS & LIPID METABOLISM The, LDL Uptake, and the Free Cholesterol Pool I. Michael Brown and Joseph Goldstein A. Studied families with familial hypercholesterolemia. B. Defined the relationship
Peroxisome proliferator-activated receptor delta activation leads to increased transintestinal cholesterol efflux
 Peroxisome proliferator-activated receptor delta activation leads to increased transintestinal cholesterol efflux Carlos L. J. Vrins, 1,2, *, Astrid E. van der Velde, 1, Karin van den Oever, Johannes H.
Peroxisome proliferator-activated receptor delta activation leads to increased transintestinal cholesterol efflux Carlos L. J. Vrins, 1,2, *, Astrid E. van der Velde, 1, Karin van den Oever, Johannes H.
Ezetimibe: a selective inhibitor of cholesterol absorption
 European Heart Journal Supplements (2001) 3 (Supplement E), E6 E10 Ezetimibe: a selective inhibitor of cholesterol absorption Dipartimento di Scienze Farmacologiche, Universita degli Studi di Milano, Milano,
European Heart Journal Supplements (2001) 3 (Supplement E), E6 E10 Ezetimibe: a selective inhibitor of cholesterol absorption Dipartimento di Scienze Farmacologiche, Universita degli Studi di Milano, Milano,
Ezetimibe: a selective inhibitor of cholesterol absorption
 European Heart Journal Supplements (2001) 3 (Supplement E), E6 E10 Ezetimibe: a selective inhibitor of cholesterol absorption Dipartimento di Scienze Farmacologiche, Universita degli Studi di Milano, Milano,
European Heart Journal Supplements (2001) 3 (Supplement E), E6 E10 Ezetimibe: a selective inhibitor of cholesterol absorption Dipartimento di Scienze Farmacologiche, Universita degli Studi di Milano, Milano,
G. Chinetti-Gbaguidi and B. Staels, UR 545 INSERM, Institut Pasteur de Lille and Université de Lille 2, Lille, France
 LIVER X RECEPTORS (LXRS): TRANSCRIPTIONAL REGULATORS OF MACROPHAGE CHOLESTEROL METABOLISM G. Chinetti-Gbaguidi and B. Staels, UR 545 INSERM, Institut Pasteur de Lille and Université de Lille 2, Lille,
LIVER X RECEPTORS (LXRS): TRANSCRIPTIONAL REGULATORS OF MACROPHAGE CHOLESTEROL METABOLISM G. Chinetti-Gbaguidi and B. Staels, UR 545 INSERM, Institut Pasteur de Lille and Université de Lille 2, Lille,
Niacin Metabolism: Effects on Cholesterol
 Niacin Metabolism: Effects on Cholesterol By Julianne R. Edwards For Dr. William R. Proulx, PhD, RD Associate Professor of Nutrition and Dietetics In partial fulfillments for the requirements of NUTR342
Niacin Metabolism: Effects on Cholesterol By Julianne R. Edwards For Dr. William R. Proulx, PhD, RD Associate Professor of Nutrition and Dietetics In partial fulfillments for the requirements of NUTR342
ANTIHYPERLIPIDEMIA. Darmawan,dr.,M.Kes,Sp.PD
 ANTIHYPERLIPIDEMIA Darmawan,dr.,M.Kes,Sp.PD Plasma lipids consist mostly of lipoproteins Spherical complexes of lipids and specific proteins (apolipoproteins). The clinically important lipoproteins, listed
ANTIHYPERLIPIDEMIA Darmawan,dr.,M.Kes,Sp.PD Plasma lipids consist mostly of lipoproteins Spherical complexes of lipids and specific proteins (apolipoproteins). The clinically important lipoproteins, listed
Lysosomal unesterified cholesterol content correlates with liver cell death in murine Niemann-Pick type C disease
 Lysosomal unesterified cholesterol content correlates with liver cell death in murine Niemann-Pick type C disease Eduardo P. Beltroy, Benny Liu, John M. Dietschy, and Stephen D. Turley 1 Department of
Lysosomal unesterified cholesterol content correlates with liver cell death in murine Niemann-Pick type C disease Eduardo P. Beltroy, Benny Liu, John M. Dietschy, and Stephen D. Turley 1 Department of
Citation for published version (APA): Minich, D. M. (1999). Essential fatty acid absorption and metabolism Groningen: s.n.
 University of Groningen Essential fatty acid absorption and metabolism Minich, Deanna Marie IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you wish to cite from
University of Groningen Essential fatty acid absorption and metabolism Minich, Deanna Marie IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you wish to cite from
PCSK9 RNAi Therapeutics. Kevin FitzGerald
 PCSK9 RNAi Therapeutics Kevin FitzGerald Presenter Disclosure Information PSCK9 RNAi Therapeutics The following relationships exist related to this presentation:» Kevin Fitzgerald and Alnylam team members:
PCSK9 RNAi Therapeutics Kevin FitzGerald Presenter Disclosure Information PSCK9 RNAi Therapeutics The following relationships exist related to this presentation:» Kevin Fitzgerald and Alnylam team members:
